User login
Double morning-after pill dose for women with obesity not effective
Emergency contraception is more likely to fail in women with obesity, but simply doubling the dose of levonorgestrel (LNG)-based contraception does not appear to be effective according to the results of a randomized, controlled trial.
Alison B. Edelman, MD, MPH, of the department of obstetrics & gynecology at Oregon Health & Science University, Portland, led the study published online in Obstetrics & Gynecology.
The researchers included healthy women ages 18-35 with regular menstrual cycles, body mass index (BMI) higher than 30 kg/m2, and weight at least 176 pounds in a randomized study.
After confirming ovulation, researchers monitored participants with transvaginal ultrasonography and blood sampling for progesterone, luteinizing hormone, and estradiol every other day until a dominant follicle 15 mm or greater was seen.
At that point the women received either LNG 1.5 mg or 3 mg and returned for daily monitoring up to 7 days.
Emergency contraception with LNG works by preventing the luteinizing hormone surge, blocking follicle rupture. The researchers had hypothesized that women with obesity might not be getting enough LNG to block the surge after oral dosing.
Previous trials had shown women with obesity had a fourfold higher risk of pregnancy, compared with women with normal BMI taking emergency contraception.
The primary outcome in this trial was whether women had follicle rupture 5 days after dosing.
The authors wrote: “The study had 80% power to detect a 30% difference in the proportion of cycles with at least a 5-day delay in follicle rupture (50% decrease).”
A total of 70 women completed study procedures. The two groups (35 women in each) had similar demographics (mean age, 28 years; BMI, 38).
No differences found between groups
“We found no difference between groups in the proportion of participants without follicle rupture,” the researchers wrote.
More than 5 days after dosing, 51.4% in the lower-dose group did not experience follicle rupture. In the double-dose group 68.6% did not experience rupture but the difference was not significant (P = .14).
Among participants with follicle rupture before 5 days, the time to rupture – the secondary endpoint – also did not differ between groups.
The researchers concluded that more research on the failures of hormonal emergency contraception in women with obesity is needed.
Eve Espey, MD, MPH, distinguished professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque, said in an interview that the study was well designed and the results “form a strong basis for clinical recommendations.”
“Providers should not recommend a higher dose of LNG emergency contraception for patients who are overweight or obese, but rather should counsel patients on the superior effectiveness of ulipristal acetate for those seeking oral emergency contraception as well as the longer time period after unprotected sex – 5 days – that ulipristal maintains its effectiveness.”
“Providers should also counsel patients on the most effective emergency contraception methods, the copper or LNG intrauterine device,” she said.
She said the unique study design of a pharmacodynamic randomized controlled trial adds weight to the findings.
She and the authors noted a limitation is the use of a surrogate outcome, ovulation delay, for ethical and feasibility reasons, instead of the outcome of interest, pregnancy.
The trial was conducted at Oregon Health & Science University and Eastern Virginia Medical School, Norfolk, from June 2017 to February 2021.
Study enrollees were compensated for their time. They were required not to be at risk for pregnancy (abstinent or using a nonhormonal method of contraception).
Dr. Edelman reported receiving honoraria and travel reimbursement from the American College of Obstetricians and Gynecologists, the World Health Organization, and Gynuity for committee activities and honoraria for peer review from the Karolinska Institute. She receives royalties from UpToDate. Several coauthors have received payments for consulting from multiple pharmaceutical companies. These companies and organizations may have a commercial or financial interest in the results of this research and technology. Another was involved in this study as a private consultant and is employed by Gilead Sciences, which was not involved in this research.
Emergency contraception is more likely to fail in women with obesity, but simply doubling the dose of levonorgestrel (LNG)-based contraception does not appear to be effective according to the results of a randomized, controlled trial.
Alison B. Edelman, MD, MPH, of the department of obstetrics & gynecology at Oregon Health & Science University, Portland, led the study published online in Obstetrics & Gynecology.
The researchers included healthy women ages 18-35 with regular menstrual cycles, body mass index (BMI) higher than 30 kg/m2, and weight at least 176 pounds in a randomized study.
After confirming ovulation, researchers monitored participants with transvaginal ultrasonography and blood sampling for progesterone, luteinizing hormone, and estradiol every other day until a dominant follicle 15 mm or greater was seen.
At that point the women received either LNG 1.5 mg or 3 mg and returned for daily monitoring up to 7 days.
Emergency contraception with LNG works by preventing the luteinizing hormone surge, blocking follicle rupture. The researchers had hypothesized that women with obesity might not be getting enough LNG to block the surge after oral dosing.
Previous trials had shown women with obesity had a fourfold higher risk of pregnancy, compared with women with normal BMI taking emergency contraception.
The primary outcome in this trial was whether women had follicle rupture 5 days after dosing.
The authors wrote: “The study had 80% power to detect a 30% difference in the proportion of cycles with at least a 5-day delay in follicle rupture (50% decrease).”
A total of 70 women completed study procedures. The two groups (35 women in each) had similar demographics (mean age, 28 years; BMI, 38).
No differences found between groups
“We found no difference between groups in the proportion of participants without follicle rupture,” the researchers wrote.
More than 5 days after dosing, 51.4% in the lower-dose group did not experience follicle rupture. In the double-dose group 68.6% did not experience rupture but the difference was not significant (P = .14).
Among participants with follicle rupture before 5 days, the time to rupture – the secondary endpoint – also did not differ between groups.
The researchers concluded that more research on the failures of hormonal emergency contraception in women with obesity is needed.
Eve Espey, MD, MPH, distinguished professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque, said in an interview that the study was well designed and the results “form a strong basis for clinical recommendations.”
“Providers should not recommend a higher dose of LNG emergency contraception for patients who are overweight or obese, but rather should counsel patients on the superior effectiveness of ulipristal acetate for those seeking oral emergency contraception as well as the longer time period after unprotected sex – 5 days – that ulipristal maintains its effectiveness.”
“Providers should also counsel patients on the most effective emergency contraception methods, the copper or LNG intrauterine device,” she said.
She said the unique study design of a pharmacodynamic randomized controlled trial adds weight to the findings.
She and the authors noted a limitation is the use of a surrogate outcome, ovulation delay, for ethical and feasibility reasons, instead of the outcome of interest, pregnancy.
The trial was conducted at Oregon Health & Science University and Eastern Virginia Medical School, Norfolk, from June 2017 to February 2021.
Study enrollees were compensated for their time. They were required not to be at risk for pregnancy (abstinent or using a nonhormonal method of contraception).
Dr. Edelman reported receiving honoraria and travel reimbursement from the American College of Obstetricians and Gynecologists, the World Health Organization, and Gynuity for committee activities and honoraria for peer review from the Karolinska Institute. She receives royalties from UpToDate. Several coauthors have received payments for consulting from multiple pharmaceutical companies. These companies and organizations may have a commercial or financial interest in the results of this research and technology. Another was involved in this study as a private consultant and is employed by Gilead Sciences, which was not involved in this research.
Emergency contraception is more likely to fail in women with obesity, but simply doubling the dose of levonorgestrel (LNG)-based contraception does not appear to be effective according to the results of a randomized, controlled trial.
Alison B. Edelman, MD, MPH, of the department of obstetrics & gynecology at Oregon Health & Science University, Portland, led the study published online in Obstetrics & Gynecology.
The researchers included healthy women ages 18-35 with regular menstrual cycles, body mass index (BMI) higher than 30 kg/m2, and weight at least 176 pounds in a randomized study.
After confirming ovulation, researchers monitored participants with transvaginal ultrasonography and blood sampling for progesterone, luteinizing hormone, and estradiol every other day until a dominant follicle 15 mm or greater was seen.
At that point the women received either LNG 1.5 mg or 3 mg and returned for daily monitoring up to 7 days.
Emergency contraception with LNG works by preventing the luteinizing hormone surge, blocking follicle rupture. The researchers had hypothesized that women with obesity might not be getting enough LNG to block the surge after oral dosing.
Previous trials had shown women with obesity had a fourfold higher risk of pregnancy, compared with women with normal BMI taking emergency contraception.
The primary outcome in this trial was whether women had follicle rupture 5 days after dosing.
The authors wrote: “The study had 80% power to detect a 30% difference in the proportion of cycles with at least a 5-day delay in follicle rupture (50% decrease).”
A total of 70 women completed study procedures. The two groups (35 women in each) had similar demographics (mean age, 28 years; BMI, 38).
No differences found between groups
“We found no difference between groups in the proportion of participants without follicle rupture,” the researchers wrote.
More than 5 days after dosing, 51.4% in the lower-dose group did not experience follicle rupture. In the double-dose group 68.6% did not experience rupture but the difference was not significant (P = .14).
Among participants with follicle rupture before 5 days, the time to rupture – the secondary endpoint – also did not differ between groups.
The researchers concluded that more research on the failures of hormonal emergency contraception in women with obesity is needed.
Eve Espey, MD, MPH, distinguished professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque, said in an interview that the study was well designed and the results “form a strong basis for clinical recommendations.”
“Providers should not recommend a higher dose of LNG emergency contraception for patients who are overweight or obese, but rather should counsel patients on the superior effectiveness of ulipristal acetate for those seeking oral emergency contraception as well as the longer time period after unprotected sex – 5 days – that ulipristal maintains its effectiveness.”
“Providers should also counsel patients on the most effective emergency contraception methods, the copper or LNG intrauterine device,” she said.
She said the unique study design of a pharmacodynamic randomized controlled trial adds weight to the findings.
She and the authors noted a limitation is the use of a surrogate outcome, ovulation delay, for ethical and feasibility reasons, instead of the outcome of interest, pregnancy.
The trial was conducted at Oregon Health & Science University and Eastern Virginia Medical School, Norfolk, from June 2017 to February 2021.
Study enrollees were compensated for their time. They were required not to be at risk for pregnancy (abstinent or using a nonhormonal method of contraception).
Dr. Edelman reported receiving honoraria and travel reimbursement from the American College of Obstetricians and Gynecologists, the World Health Organization, and Gynuity for committee activities and honoraria for peer review from the Karolinska Institute. She receives royalties from UpToDate. Several coauthors have received payments for consulting from multiple pharmaceutical companies. These companies and organizations may have a commercial or financial interest in the results of this research and technology. Another was involved in this study as a private consultant and is employed by Gilead Sciences, which was not involved in this research.
FROM OBSTETRICS & GYNECOLOGY
PCOS comes with high morbidity, medication use into late 40s
Women with polycystic ovary syndrome (PCOS) have an increased risk for several diseases and symptoms, many independent of body mass index (BMI), new research indicates.
Some diseases are linked for the first time to PCOS in this study, the authors wrote.
Researchers, led by Linda Kujanpää, MD, of the research unit for pediatrics, dermatology, clinical genetics, obstetrics, and gynecology at University of Oulu (Finland), found the morbidity risk is evident through the late reproductive years.
The paper was published online in Acta Obstetricia et Gynecologica Scandinavica.
This population-based follow-up study investigated comorbidities and medication and health care services use among women with PCOS in Finland at age 46 years via answers to a questionnaire.
The whole PCOS population (n = 280) consisted of women who reported both hirsutism and oligo/amenorrhea at age 31 (4.1%) and/or polycystic ovary morphology/PCOS at age 46 (3.1%), of which 246 replied to the 46-year questionnaire. They were compared with a control group of 1,573 women without PCOS.
Overall morbidity risk was 35% higher than for women without PCOS (risk ratio, 1.35; 95% confidence interval, 1.16-1.57). Medication use was 27% higher (RR, 1.27; 95% CI, 1.08-1.50), and the risk remained after adjusting for BMI.
Diagnoses with increased prevalence in women with PCOS were osteoarthritis, migraine, hypertension, tendinitis, and endometriosis. PCOS was also associated with autoimmune diseases and recurrent upper respiratory tract infections.
“BMI seems not to be solely responsible for the increased morbidity,” the researchers found. The average morbidity score of women with PCOS with a BMI of 25 kg/m2 or higher was similar to that of women with PCOS and lower BMI.
Mindy Christianson, MD, medical director at Johns Hopkins Fertility Center and associate professor of gynecology and obstetrics at Johns Hopkins University, both in Baltimore, said in an interview that the links to diseases independent of BMI are interesting because there’s so much focus on counseling women with PCOS to lose weight.
While that message is still important, it’s important to realize that some related diseases and conditions – such as autoimmune diseases and migraine – are not driven by BMI.
“It really drives home the point that polycystic ovary syndrome is really a chronic medical condition and puts patients at risk for a number of health conditions,” she said. “Having a good primary care physician is important to help them with their overall health.”
Women with PCOS said their health was poor or very poor almost three times more often than did women in the control group.
Surprisingly few studies have looked at overall comorbidity in women with PCOS, the authors wrote.
“This should be of high priority given the high cost to society resulting from PCOS-related morbidity,” they added. As an example, they pointed out that PCOS-related type 2 diabetes alone costs an estimated $1.77 billion in the United States and £237 million ($310 million) each year in the United Kingdom.
Additionally, the focus in previous research has typically been on women in their early or mid-reproductive years, and morbidity burden data in late reproductive years are scarce.
The study population was pulled from the longitudinal Northern Finland Birth Cohort 1966 and included all pregnancies with estimated date of delivery during 1966 in two provinces of Finland (5,889 women).
Dr. Christianson said she hopes this study will spur more research on PCOS, which has been severely underfunded, especially in the United States.
Part of the reason for that is there is a limited number of subspecialists in the country who work with patients with PCOS and do research in the area. PCOS often gets lost in the research priorities of infertility, diabetes, and thyroid disease.
The message in this study that PCOS is not just a fertility issue or an obesity issue but an overall health issue with a substantial cost to the health system may help raise awareness, Dr. Christianson said.
This study was supported by grants from The Finnish Medical Foundation, The Academy of Finland, The Sigrid Juselius Foundation, The Finnish Cultural Foundation, The Jalmari and Rauha Ahokas Foundation, The Päivikki and Sakari Sohlberg Foundation, Genesis Research Trust, The Medical Research Council, University of Oulu, Oulu University Hospital, Ministry of Health and Social Affairs, National Institute for Health and Welfare, Regional Institute of Occupational Health, and the European Regional Development Fund. The Study authors and Dr. Christianson reported no relevant financial relationships.
Women with polycystic ovary syndrome (PCOS) have an increased risk for several diseases and symptoms, many independent of body mass index (BMI), new research indicates.
Some diseases are linked for the first time to PCOS in this study, the authors wrote.
Researchers, led by Linda Kujanpää, MD, of the research unit for pediatrics, dermatology, clinical genetics, obstetrics, and gynecology at University of Oulu (Finland), found the morbidity risk is evident through the late reproductive years.
The paper was published online in Acta Obstetricia et Gynecologica Scandinavica.
This population-based follow-up study investigated comorbidities and medication and health care services use among women with PCOS in Finland at age 46 years via answers to a questionnaire.
The whole PCOS population (n = 280) consisted of women who reported both hirsutism and oligo/amenorrhea at age 31 (4.1%) and/or polycystic ovary morphology/PCOS at age 46 (3.1%), of which 246 replied to the 46-year questionnaire. They were compared with a control group of 1,573 women without PCOS.
Overall morbidity risk was 35% higher than for women without PCOS (risk ratio, 1.35; 95% confidence interval, 1.16-1.57). Medication use was 27% higher (RR, 1.27; 95% CI, 1.08-1.50), and the risk remained after adjusting for BMI.
Diagnoses with increased prevalence in women with PCOS were osteoarthritis, migraine, hypertension, tendinitis, and endometriosis. PCOS was also associated with autoimmune diseases and recurrent upper respiratory tract infections.
“BMI seems not to be solely responsible for the increased morbidity,” the researchers found. The average morbidity score of women with PCOS with a BMI of 25 kg/m2 or higher was similar to that of women with PCOS and lower BMI.
Mindy Christianson, MD, medical director at Johns Hopkins Fertility Center and associate professor of gynecology and obstetrics at Johns Hopkins University, both in Baltimore, said in an interview that the links to diseases independent of BMI are interesting because there’s so much focus on counseling women with PCOS to lose weight.
While that message is still important, it’s important to realize that some related diseases and conditions – such as autoimmune diseases and migraine – are not driven by BMI.
“It really drives home the point that polycystic ovary syndrome is really a chronic medical condition and puts patients at risk for a number of health conditions,” she said. “Having a good primary care physician is important to help them with their overall health.”
Women with PCOS said their health was poor or very poor almost three times more often than did women in the control group.
Surprisingly few studies have looked at overall comorbidity in women with PCOS, the authors wrote.
“This should be of high priority given the high cost to society resulting from PCOS-related morbidity,” they added. As an example, they pointed out that PCOS-related type 2 diabetes alone costs an estimated $1.77 billion in the United States and £237 million ($310 million) each year in the United Kingdom.
Additionally, the focus in previous research has typically been on women in their early or mid-reproductive years, and morbidity burden data in late reproductive years are scarce.
The study population was pulled from the longitudinal Northern Finland Birth Cohort 1966 and included all pregnancies with estimated date of delivery during 1966 in two provinces of Finland (5,889 women).
Dr. Christianson said she hopes this study will spur more research on PCOS, which has been severely underfunded, especially in the United States.
Part of the reason for that is there is a limited number of subspecialists in the country who work with patients with PCOS and do research in the area. PCOS often gets lost in the research priorities of infertility, diabetes, and thyroid disease.
The message in this study that PCOS is not just a fertility issue or an obesity issue but an overall health issue with a substantial cost to the health system may help raise awareness, Dr. Christianson said.
This study was supported by grants from The Finnish Medical Foundation, The Academy of Finland, The Sigrid Juselius Foundation, The Finnish Cultural Foundation, The Jalmari and Rauha Ahokas Foundation, The Päivikki and Sakari Sohlberg Foundation, Genesis Research Trust, The Medical Research Council, University of Oulu, Oulu University Hospital, Ministry of Health and Social Affairs, National Institute for Health and Welfare, Regional Institute of Occupational Health, and the European Regional Development Fund. The Study authors and Dr. Christianson reported no relevant financial relationships.
Women with polycystic ovary syndrome (PCOS) have an increased risk for several diseases and symptoms, many independent of body mass index (BMI), new research indicates.
Some diseases are linked for the first time to PCOS in this study, the authors wrote.
Researchers, led by Linda Kujanpää, MD, of the research unit for pediatrics, dermatology, clinical genetics, obstetrics, and gynecology at University of Oulu (Finland), found the morbidity risk is evident through the late reproductive years.
The paper was published online in Acta Obstetricia et Gynecologica Scandinavica.
This population-based follow-up study investigated comorbidities and medication and health care services use among women with PCOS in Finland at age 46 years via answers to a questionnaire.
The whole PCOS population (n = 280) consisted of women who reported both hirsutism and oligo/amenorrhea at age 31 (4.1%) and/or polycystic ovary morphology/PCOS at age 46 (3.1%), of which 246 replied to the 46-year questionnaire. They were compared with a control group of 1,573 women without PCOS.
Overall morbidity risk was 35% higher than for women without PCOS (risk ratio, 1.35; 95% confidence interval, 1.16-1.57). Medication use was 27% higher (RR, 1.27; 95% CI, 1.08-1.50), and the risk remained after adjusting for BMI.
Diagnoses with increased prevalence in women with PCOS were osteoarthritis, migraine, hypertension, tendinitis, and endometriosis. PCOS was also associated with autoimmune diseases and recurrent upper respiratory tract infections.
“BMI seems not to be solely responsible for the increased morbidity,” the researchers found. The average morbidity score of women with PCOS with a BMI of 25 kg/m2 or higher was similar to that of women with PCOS and lower BMI.
Mindy Christianson, MD, medical director at Johns Hopkins Fertility Center and associate professor of gynecology and obstetrics at Johns Hopkins University, both in Baltimore, said in an interview that the links to diseases independent of BMI are interesting because there’s so much focus on counseling women with PCOS to lose weight.
While that message is still important, it’s important to realize that some related diseases and conditions – such as autoimmune diseases and migraine – are not driven by BMI.
“It really drives home the point that polycystic ovary syndrome is really a chronic medical condition and puts patients at risk for a number of health conditions,” she said. “Having a good primary care physician is important to help them with their overall health.”
Women with PCOS said their health was poor or very poor almost three times more often than did women in the control group.
Surprisingly few studies have looked at overall comorbidity in women with PCOS, the authors wrote.
“This should be of high priority given the high cost to society resulting from PCOS-related morbidity,” they added. As an example, they pointed out that PCOS-related type 2 diabetes alone costs an estimated $1.77 billion in the United States and £237 million ($310 million) each year in the United Kingdom.
Additionally, the focus in previous research has typically been on women in their early or mid-reproductive years, and morbidity burden data in late reproductive years are scarce.
The study population was pulled from the longitudinal Northern Finland Birth Cohort 1966 and included all pregnancies with estimated date of delivery during 1966 in two provinces of Finland (5,889 women).
Dr. Christianson said she hopes this study will spur more research on PCOS, which has been severely underfunded, especially in the United States.
Part of the reason for that is there is a limited number of subspecialists in the country who work with patients with PCOS and do research in the area. PCOS often gets lost in the research priorities of infertility, diabetes, and thyroid disease.
The message in this study that PCOS is not just a fertility issue or an obesity issue but an overall health issue with a substantial cost to the health system may help raise awareness, Dr. Christianson said.
This study was supported by grants from The Finnish Medical Foundation, The Academy of Finland, The Sigrid Juselius Foundation, The Finnish Cultural Foundation, The Jalmari and Rauha Ahokas Foundation, The Päivikki and Sakari Sohlberg Foundation, Genesis Research Trust, The Medical Research Council, University of Oulu, Oulu University Hospital, Ministry of Health and Social Affairs, National Institute for Health and Welfare, Regional Institute of Occupational Health, and the European Regional Development Fund. The Study authors and Dr. Christianson reported no relevant financial relationships.
FROM ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA
Antipsychotic tied to dose-related weight gain, higher cholesterol
new research suggests.
Investigators analyzed 1-year data for more than 400 patients who were taking risperidone and/or its metabolite paliperidone (Invega). Results showed increments of 1 mg of risperidone-equivalent doses were associated with an increase of 0.25% of weight within a year of follow-up.
“Although our findings report a positive and statistically significant dose-dependence of weight gain and cholesterol, both total and LDL [cholesterol], the size of the predicted changes of metabolic effects is clinically nonrelevant,” lead author Marianna Piras, PharmD, Centre for Psychiatric Neuroscience, Lausanne (Switzerland) University Hospital, said in an interview.
“Therefore, dose lowering would not have a beneficial effect on attenuating weight gain or cholesterol increases and could lead to psychiatric decompensation,” said Ms. Piras, who is also a PhD candidate in the unit of pharmacogenetics and clinical psychopharmacology at the University of Lausanne.
However, she added that because dose increments could increase risk for significant weight gain in the first month of treatment – the dose can be increased typically in a range of 1-10 grams – and strong dose increments could contribute to metabolic worsening over time, “risperidone minimum effective doses should be preferred.”
The findings were published online in the Journal of Clinical Psychiatry.
‘Serious public health issue’
Compared with the general population, patients with mental illness present with a greater prevalence of metabolic disorders. In addition, several psychotropic medications, including antipsychotics, can induce metabolic alterations such as weight gain, the investigators noted.
Antipsychotic-induced metabolic adverse effects “constitute a serious public health issue” because they are risk factors for cardiovascular diseases such as obesity and/or dyslipidemia, “which have been associated with a 10-year reduced life expectancy in the psychiatric population,” Ms. Piras said.
“The dose-dependence of metabolic adverse effects is a debated subject that needs to be assessed for each psychotropic drug known to induce weight gain,” she added.
Several previous studies have examined whether there is a dose-related effect of antipsychotics on metabolic parameters, “with some results suggesting that [weight gain] seems to develop even when low off-label doses are prescribed,” Ms. Piras noted.
She and her colleagues had already studied dose-related metabolic effects of quetiapine (Seroquel) and olanzapine (Zyprexa).
Risperidone is an antipsychotic with a “medium to high metabolic risk profile,” the researchers note, and few studies have examined the impact of risperidone on metabolic parameters other than weight gain.
For the current analysis, they analyzed data from a longitudinal study that included 438 patients (mean age, 40.7 years; 50.7% men) who started treatment with risperidone and/or paliperidone between 2007 and 2018.
The participants had diagnoses of schizophrenia, schizoaffective disorder, bipolar disorder, depression, “other,” or “unknown.”
Clinical follow-up periods were up to a year, but were no shorter than 3 weeks. The investigators also assessed the data at different time intervals at 1, 3, 6, and 12 months “to appreciate the evolution of the metabolic parameters.”
In addition, they collected demographic and clinical information, such as comorbidities, and measured patients’ weight, height, waist circumference, blood pressure, plasma glucose, and lipids at baseline and at 1, 3, and 12 months and then annually. Weight, waist circumference, and BP were also assessed at 2 and 6 months.
Doses of paliperidone were converted into risperidone-equivalent doses.
Significant weight gain over time
The mean duration of follow-up for the participants, of whom 374 were being treated with risperidone and 64 with paliperidone, was 153 days. Close to half (48.2%) were taking other psychotropic medications known to be associated with some degree of metabolic risk.
Patients were divided into two cohorts based on their daily dose intake (DDI): less than 3 mg/day (n = 201) and at least 3 mg/day (n = 237).
In the overall cohort, a “significant effect of time on weight change was found for each time point,” the investigators reported.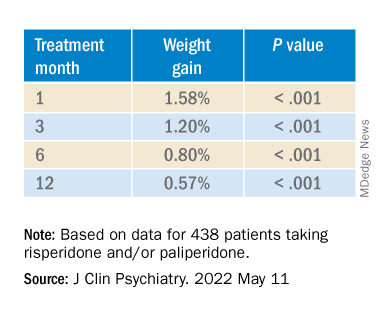
When the researchers looked at the changes according to DDI, they found that each 1-mg dose increase was associated with incremental weight gain at each time point.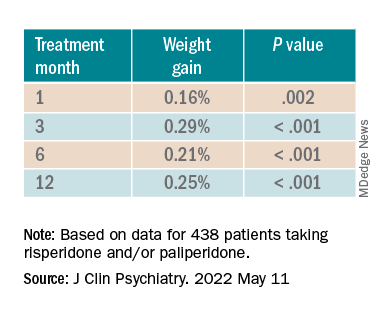
Patients who had 5% or greater weight gain in the first month continued to gain weight more than patients who did not reach that threshold, leading the researchers to call that early threshold a “strong predictor of important weight gain in the long term.” There was a weight gain of 6.68% at 3 months, of 7.36% at 6 months, and of 7.7% at 12 months.
After the patients were stratified by age, there were differences in the effect of DDI on various age groups at different time points.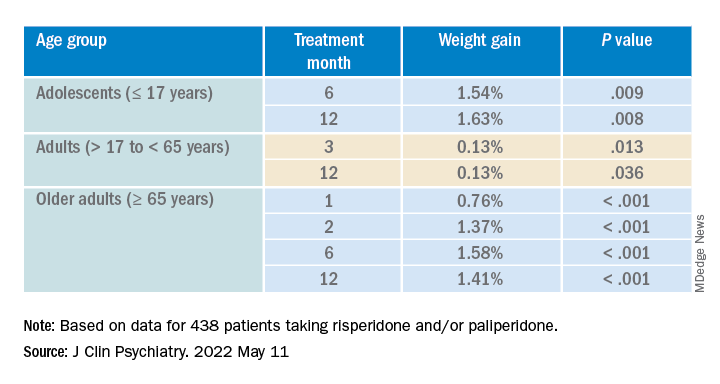
Dose was shown to have a significant effect on weight gain for women at all four time points (P ≥ .001), but for men only at 3 months (P = .003).
For each additional 1-mg dose, there was a 0.05 mmol/L (1.93 mg/dL) increase in total cholesterol (P = .018) after 1 year and a 0.04 mmol/L (1.54 mg/dL) increase in LDL cholesterol (P = .011).
There were no significant effects of time or DDI on triglycerides, HDL cholesterol, glucose levels, and systolic BP, and there was a negative effect of DDI on diastolic BP (P = .001).
The findings “provide evidence for a small dose effect of risperidone” on weight gain and total and LDL cholesterol levels, the investigators note.
Ms. Piras added that because each antipsychotic differs in its metabolic risk profile, “further analyses on other antipsychotics are ongoing in our laboratory, so far confirming our findings.”
Small increases, big changes
Commenting on the study, Erika Nurmi, MD, PhD, associate professor in the department of psychiatry and biobehavioral sciences at the Semel Institute for Neuroscience, University of California, Los Angeles, said the study is “unique in the field.”
It “leverages real-world data from a large patient registry to ask a long-unanswered question: Are weight and metabolic adverse effects proportional to dose? Big data approaches like these are very powerful, given the large number of participants that can be included,” said Dr. Nurmi, who was not involved with the research.
However, she cautioned, the “biggest drawback [is that] these data are by nature much more complex and prone to confounding effects.”
In this case, a “critical confounder” for the study was that the majority of individuals taking higher risperidone doses were also taking other drugs known to cause weight gain, whereas the majority of those on lower risperidone doses were not. “This difference may explain the dose relationship observed,” she said.
Because real-world, big data are “valuable but also messy, conclusions drawn from them must be interpreted with caution,” Dr. Nurmi said.
She added that it is generally wise to use the lowest effective dose possible.
“Clinicians should appreciate that even small doses of antipsychotics can cause big changes in weight. Risks and benefits of medications must be carefully considered in clinical practice,” Dr. Nurmi said.
The research was funded in part by the Swiss National Research Foundation. Piras reports no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Nurmi reported no relevant financial relationships, but she is an unpaid member of the Tourette Association of America’s medical advisory board and of the Myriad Genetics scientific advisory board.
A version of this article first appeared on Medscape.com.
new research suggests.
Investigators analyzed 1-year data for more than 400 patients who were taking risperidone and/or its metabolite paliperidone (Invega). Results showed increments of 1 mg of risperidone-equivalent doses were associated with an increase of 0.25% of weight within a year of follow-up.
“Although our findings report a positive and statistically significant dose-dependence of weight gain and cholesterol, both total and LDL [cholesterol], the size of the predicted changes of metabolic effects is clinically nonrelevant,” lead author Marianna Piras, PharmD, Centre for Psychiatric Neuroscience, Lausanne (Switzerland) University Hospital, said in an interview.
“Therefore, dose lowering would not have a beneficial effect on attenuating weight gain or cholesterol increases and could lead to psychiatric decompensation,” said Ms. Piras, who is also a PhD candidate in the unit of pharmacogenetics and clinical psychopharmacology at the University of Lausanne.
However, she added that because dose increments could increase risk for significant weight gain in the first month of treatment – the dose can be increased typically in a range of 1-10 grams – and strong dose increments could contribute to metabolic worsening over time, “risperidone minimum effective doses should be preferred.”
The findings were published online in the Journal of Clinical Psychiatry.
‘Serious public health issue’
Compared with the general population, patients with mental illness present with a greater prevalence of metabolic disorders. In addition, several psychotropic medications, including antipsychotics, can induce metabolic alterations such as weight gain, the investigators noted.
Antipsychotic-induced metabolic adverse effects “constitute a serious public health issue” because they are risk factors for cardiovascular diseases such as obesity and/or dyslipidemia, “which have been associated with a 10-year reduced life expectancy in the psychiatric population,” Ms. Piras said.
“The dose-dependence of metabolic adverse effects is a debated subject that needs to be assessed for each psychotropic drug known to induce weight gain,” she added.
Several previous studies have examined whether there is a dose-related effect of antipsychotics on metabolic parameters, “with some results suggesting that [weight gain] seems to develop even when low off-label doses are prescribed,” Ms. Piras noted.
She and her colleagues had already studied dose-related metabolic effects of quetiapine (Seroquel) and olanzapine (Zyprexa).
Risperidone is an antipsychotic with a “medium to high metabolic risk profile,” the researchers note, and few studies have examined the impact of risperidone on metabolic parameters other than weight gain.
For the current analysis, they analyzed data from a longitudinal study that included 438 patients (mean age, 40.7 years; 50.7% men) who started treatment with risperidone and/or paliperidone between 2007 and 2018.
The participants had diagnoses of schizophrenia, schizoaffective disorder, bipolar disorder, depression, “other,” or “unknown.”
Clinical follow-up periods were up to a year, but were no shorter than 3 weeks. The investigators also assessed the data at different time intervals at 1, 3, 6, and 12 months “to appreciate the evolution of the metabolic parameters.”
In addition, they collected demographic and clinical information, such as comorbidities, and measured patients’ weight, height, waist circumference, blood pressure, plasma glucose, and lipids at baseline and at 1, 3, and 12 months and then annually. Weight, waist circumference, and BP were also assessed at 2 and 6 months.
Doses of paliperidone were converted into risperidone-equivalent doses.
Significant weight gain over time
The mean duration of follow-up for the participants, of whom 374 were being treated with risperidone and 64 with paliperidone, was 153 days. Close to half (48.2%) were taking other psychotropic medications known to be associated with some degree of metabolic risk.
Patients were divided into two cohorts based on their daily dose intake (DDI): less than 3 mg/day (n = 201) and at least 3 mg/day (n = 237).
In the overall cohort, a “significant effect of time on weight change was found for each time point,” the investigators reported.
When the researchers looked at the changes according to DDI, they found that each 1-mg dose increase was associated with incremental weight gain at each time point.
Patients who had 5% or greater weight gain in the first month continued to gain weight more than patients who did not reach that threshold, leading the researchers to call that early threshold a “strong predictor of important weight gain in the long term.” There was a weight gain of 6.68% at 3 months, of 7.36% at 6 months, and of 7.7% at 12 months.
After the patients were stratified by age, there were differences in the effect of DDI on various age groups at different time points.
Dose was shown to have a significant effect on weight gain for women at all four time points (P ≥ .001), but for men only at 3 months (P = .003).
For each additional 1-mg dose, there was a 0.05 mmol/L (1.93 mg/dL) increase in total cholesterol (P = .018) after 1 year and a 0.04 mmol/L (1.54 mg/dL) increase in LDL cholesterol (P = .011).
There were no significant effects of time or DDI on triglycerides, HDL cholesterol, glucose levels, and systolic BP, and there was a negative effect of DDI on diastolic BP (P = .001).
The findings “provide evidence for a small dose effect of risperidone” on weight gain and total and LDL cholesterol levels, the investigators note.
Ms. Piras added that because each antipsychotic differs in its metabolic risk profile, “further analyses on other antipsychotics are ongoing in our laboratory, so far confirming our findings.”
Small increases, big changes
Commenting on the study, Erika Nurmi, MD, PhD, associate professor in the department of psychiatry and biobehavioral sciences at the Semel Institute for Neuroscience, University of California, Los Angeles, said the study is “unique in the field.”
It “leverages real-world data from a large patient registry to ask a long-unanswered question: Are weight and metabolic adverse effects proportional to dose? Big data approaches like these are very powerful, given the large number of participants that can be included,” said Dr. Nurmi, who was not involved with the research.
However, she cautioned, the “biggest drawback [is that] these data are by nature much more complex and prone to confounding effects.”
In this case, a “critical confounder” for the study was that the majority of individuals taking higher risperidone doses were also taking other drugs known to cause weight gain, whereas the majority of those on lower risperidone doses were not. “This difference may explain the dose relationship observed,” she said.
Because real-world, big data are “valuable but also messy, conclusions drawn from them must be interpreted with caution,” Dr. Nurmi said.
She added that it is generally wise to use the lowest effective dose possible.
“Clinicians should appreciate that even small doses of antipsychotics can cause big changes in weight. Risks and benefits of medications must be carefully considered in clinical practice,” Dr. Nurmi said.
The research was funded in part by the Swiss National Research Foundation. Piras reports no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Nurmi reported no relevant financial relationships, but she is an unpaid member of the Tourette Association of America’s medical advisory board and of the Myriad Genetics scientific advisory board.
A version of this article first appeared on Medscape.com.
new research suggests.
Investigators analyzed 1-year data for more than 400 patients who were taking risperidone and/or its metabolite paliperidone (Invega). Results showed increments of 1 mg of risperidone-equivalent doses were associated with an increase of 0.25% of weight within a year of follow-up.
“Although our findings report a positive and statistically significant dose-dependence of weight gain and cholesterol, both total and LDL [cholesterol], the size of the predicted changes of metabolic effects is clinically nonrelevant,” lead author Marianna Piras, PharmD, Centre for Psychiatric Neuroscience, Lausanne (Switzerland) University Hospital, said in an interview.
“Therefore, dose lowering would not have a beneficial effect on attenuating weight gain or cholesterol increases and could lead to psychiatric decompensation,” said Ms. Piras, who is also a PhD candidate in the unit of pharmacogenetics and clinical psychopharmacology at the University of Lausanne.
However, she added that because dose increments could increase risk for significant weight gain in the first month of treatment – the dose can be increased typically in a range of 1-10 grams – and strong dose increments could contribute to metabolic worsening over time, “risperidone minimum effective doses should be preferred.”
The findings were published online in the Journal of Clinical Psychiatry.
‘Serious public health issue’
Compared with the general population, patients with mental illness present with a greater prevalence of metabolic disorders. In addition, several psychotropic medications, including antipsychotics, can induce metabolic alterations such as weight gain, the investigators noted.
Antipsychotic-induced metabolic adverse effects “constitute a serious public health issue” because they are risk factors for cardiovascular diseases such as obesity and/or dyslipidemia, “which have been associated with a 10-year reduced life expectancy in the psychiatric population,” Ms. Piras said.
“The dose-dependence of metabolic adverse effects is a debated subject that needs to be assessed for each psychotropic drug known to induce weight gain,” she added.
Several previous studies have examined whether there is a dose-related effect of antipsychotics on metabolic parameters, “with some results suggesting that [weight gain] seems to develop even when low off-label doses are prescribed,” Ms. Piras noted.
She and her colleagues had already studied dose-related metabolic effects of quetiapine (Seroquel) and olanzapine (Zyprexa).
Risperidone is an antipsychotic with a “medium to high metabolic risk profile,” the researchers note, and few studies have examined the impact of risperidone on metabolic parameters other than weight gain.
For the current analysis, they analyzed data from a longitudinal study that included 438 patients (mean age, 40.7 years; 50.7% men) who started treatment with risperidone and/or paliperidone between 2007 and 2018.
The participants had diagnoses of schizophrenia, schizoaffective disorder, bipolar disorder, depression, “other,” or “unknown.”
Clinical follow-up periods were up to a year, but were no shorter than 3 weeks. The investigators also assessed the data at different time intervals at 1, 3, 6, and 12 months “to appreciate the evolution of the metabolic parameters.”
In addition, they collected demographic and clinical information, such as comorbidities, and measured patients’ weight, height, waist circumference, blood pressure, plasma glucose, and lipids at baseline and at 1, 3, and 12 months and then annually. Weight, waist circumference, and BP were also assessed at 2 and 6 months.
Doses of paliperidone were converted into risperidone-equivalent doses.
Significant weight gain over time
The mean duration of follow-up for the participants, of whom 374 were being treated with risperidone and 64 with paliperidone, was 153 days. Close to half (48.2%) were taking other psychotropic medications known to be associated with some degree of metabolic risk.
Patients were divided into two cohorts based on their daily dose intake (DDI): less than 3 mg/day (n = 201) and at least 3 mg/day (n = 237).
In the overall cohort, a “significant effect of time on weight change was found for each time point,” the investigators reported.
When the researchers looked at the changes according to DDI, they found that each 1-mg dose increase was associated with incremental weight gain at each time point.
Patients who had 5% or greater weight gain in the first month continued to gain weight more than patients who did not reach that threshold, leading the researchers to call that early threshold a “strong predictor of important weight gain in the long term.” There was a weight gain of 6.68% at 3 months, of 7.36% at 6 months, and of 7.7% at 12 months.
After the patients were stratified by age, there were differences in the effect of DDI on various age groups at different time points.
Dose was shown to have a significant effect on weight gain for women at all four time points (P ≥ .001), but for men only at 3 months (P = .003).
For each additional 1-mg dose, there was a 0.05 mmol/L (1.93 mg/dL) increase in total cholesterol (P = .018) after 1 year and a 0.04 mmol/L (1.54 mg/dL) increase in LDL cholesterol (P = .011).
There were no significant effects of time or DDI on triglycerides, HDL cholesterol, glucose levels, and systolic BP, and there was a negative effect of DDI on diastolic BP (P = .001).
The findings “provide evidence for a small dose effect of risperidone” on weight gain and total and LDL cholesterol levels, the investigators note.
Ms. Piras added that because each antipsychotic differs in its metabolic risk profile, “further analyses on other antipsychotics are ongoing in our laboratory, so far confirming our findings.”
Small increases, big changes
Commenting on the study, Erika Nurmi, MD, PhD, associate professor in the department of psychiatry and biobehavioral sciences at the Semel Institute for Neuroscience, University of California, Los Angeles, said the study is “unique in the field.”
It “leverages real-world data from a large patient registry to ask a long-unanswered question: Are weight and metabolic adverse effects proportional to dose? Big data approaches like these are very powerful, given the large number of participants that can be included,” said Dr. Nurmi, who was not involved with the research.
However, she cautioned, the “biggest drawback [is that] these data are by nature much more complex and prone to confounding effects.”
In this case, a “critical confounder” for the study was that the majority of individuals taking higher risperidone doses were also taking other drugs known to cause weight gain, whereas the majority of those on lower risperidone doses were not. “This difference may explain the dose relationship observed,” she said.
Because real-world, big data are “valuable but also messy, conclusions drawn from them must be interpreted with caution,” Dr. Nurmi said.
She added that it is generally wise to use the lowest effective dose possible.
“Clinicians should appreciate that even small doses of antipsychotics can cause big changes in weight. Risks and benefits of medications must be carefully considered in clinical practice,” Dr. Nurmi said.
The research was funded in part by the Swiss National Research Foundation. Piras reports no relevant financial relationships. The other investigators’ disclosures are listed in the original article. Dr. Nurmi reported no relevant financial relationships, but she is an unpaid member of the Tourette Association of America’s medical advisory board and of the Myriad Genetics scientific advisory board.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL PSYCHIATRY
Excess weight may ward off infection in breast cancer treatment
– a potentially deadly outcome that can occur as a result of chemotherapy treatment.
The study was presented at the annual meeting of the American Society of Clinical Oncology.
It is known that body mass index can affect breast cancer risk and prognosis, but it is not known if BMI can affect how well endocrine therapy works in a given patient. In the PALLAS clinical trial, Georg Pfeiler, MD, Medical University of Vienna, and colleagues, conducted an analysis of 5,698 patients with early hormone receptor–positive breast cancer receiving endocrine therapy with or without palbociclib. Dr. Pfeiler found that women who are overweight or obese had significantly less frequent and less severe cases of neutropenia. With fewer or less severe cases of neutropenia, there were also fewer interruptions in palbociclib treatment.
“One explanation for the lower discontinuation rates may be that the distributional volume of the drug is higher in overweight and obese patients leading to lower serum drug concentrations. It could also be influenced by differences in pharmacokinetics with respect to hyperinsulinemia,” said Dr. Pfeiler, who leads the Oncological Breast Outpatient Clinic and Bone Health Outpatient Clinic at the Medical University of Vienna.
The PALLAS trial compared the combination of palbociclib and adjuvant endocrine therapy with endocrine therapy alone in 5,698 women with early breast cancer. Patients were categorized according to BMI as underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5-24.9), overweight (BMI, 25-29.9), and obese (> 30). The investigators analyzed differences in adverse events, time to early discontinuation of palbociclib, and in time to invasive disease-free survival.
At baseline, of 5,698 patients, 68 (1.2%) were underweight, 2,082 (36.5%) were of normal weight, 1,818 (31.9%) were overweight, and, 1,730 (30.4%) were obese. In patients treated with palbociclib, neutropenia was the primary toxicity leading to treatment discontinuation with rates of 21.1% in normal-weight patients, 14.0% of overweight patients, and 5.9% of obese patients, respectively.
Significantly different rates of all-grade neutropenia were observed in normal weight, overweight, and obese participants with regard to total (88.5%, 85.7% and 74.7%), as well as grade 3 (64.1%, 62.0% and 43.9%) and grade 4 neutropenia (7.0%, 3.6% and 2.0%), respectively. The lower frequency and severity of neutropenia observed in overweight and obese patients was associated with a significantly lower treatment discontinuation rate over time when compared with normal-weight patients (overweight vs. normal weight: HR, 0.73; 95% CI 0.63-0.84; P < .0001, and obese vs. normal weight: HR, 0.65; 95% CI, 0.56-0.75; P < .0001). There was, however, despite these observations, no statistically significant improvement in invasive disease survival with the addition of palbociclib to endocrine therapy in any weight category (normal weight: HR, 0.84; 95% CI 0.63-1.12; overweight: HR, 1.10; 95% CI 0.82-1.49; and obese: HR, 0.95; 95% CI, 0.69-1.30).
“This is an early analysis, and should be interpreted with caution, especially with regard to disease outcomes. The findings may reduce concerns over hematologic side effects in the overweight and obese. In the future there may be an important impact if it turns out after longer-term follow-up that palbociclib has been underdosed in the overweight and obese. We may need BMI-adapted dose management,” said Dr. Pfeiler, who is currently working on a longer-term follow-up study of the PALLAS group.
The analysis found no significant correlation between weight and occurrence of invasive disease events.
Dr. Pfeiler disclosed honoraria and grants from Pfizer. The study was funded by Alliance Foundation Trials.
– a potentially deadly outcome that can occur as a result of chemotherapy treatment.
The study was presented at the annual meeting of the American Society of Clinical Oncology.
It is known that body mass index can affect breast cancer risk and prognosis, but it is not known if BMI can affect how well endocrine therapy works in a given patient. In the PALLAS clinical trial, Georg Pfeiler, MD, Medical University of Vienna, and colleagues, conducted an analysis of 5,698 patients with early hormone receptor–positive breast cancer receiving endocrine therapy with or without palbociclib. Dr. Pfeiler found that women who are overweight or obese had significantly less frequent and less severe cases of neutropenia. With fewer or less severe cases of neutropenia, there were also fewer interruptions in palbociclib treatment.
“One explanation for the lower discontinuation rates may be that the distributional volume of the drug is higher in overweight and obese patients leading to lower serum drug concentrations. It could also be influenced by differences in pharmacokinetics with respect to hyperinsulinemia,” said Dr. Pfeiler, who leads the Oncological Breast Outpatient Clinic and Bone Health Outpatient Clinic at the Medical University of Vienna.
The PALLAS trial compared the combination of palbociclib and adjuvant endocrine therapy with endocrine therapy alone in 5,698 women with early breast cancer. Patients were categorized according to BMI as underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5-24.9), overweight (BMI, 25-29.9), and obese (> 30). The investigators analyzed differences in adverse events, time to early discontinuation of palbociclib, and in time to invasive disease-free survival.
At baseline, of 5,698 patients, 68 (1.2%) were underweight, 2,082 (36.5%) were of normal weight, 1,818 (31.9%) were overweight, and, 1,730 (30.4%) were obese. In patients treated with palbociclib, neutropenia was the primary toxicity leading to treatment discontinuation with rates of 21.1% in normal-weight patients, 14.0% of overweight patients, and 5.9% of obese patients, respectively.
Significantly different rates of all-grade neutropenia were observed in normal weight, overweight, and obese participants with regard to total (88.5%, 85.7% and 74.7%), as well as grade 3 (64.1%, 62.0% and 43.9%) and grade 4 neutropenia (7.0%, 3.6% and 2.0%), respectively. The lower frequency and severity of neutropenia observed in overweight and obese patients was associated with a significantly lower treatment discontinuation rate over time when compared with normal-weight patients (overweight vs. normal weight: HR, 0.73; 95% CI 0.63-0.84; P < .0001, and obese vs. normal weight: HR, 0.65; 95% CI, 0.56-0.75; P < .0001). There was, however, despite these observations, no statistically significant improvement in invasive disease survival with the addition of palbociclib to endocrine therapy in any weight category (normal weight: HR, 0.84; 95% CI 0.63-1.12; overweight: HR, 1.10; 95% CI 0.82-1.49; and obese: HR, 0.95; 95% CI, 0.69-1.30).
“This is an early analysis, and should be interpreted with caution, especially with regard to disease outcomes. The findings may reduce concerns over hematologic side effects in the overweight and obese. In the future there may be an important impact if it turns out after longer-term follow-up that palbociclib has been underdosed in the overweight and obese. We may need BMI-adapted dose management,” said Dr. Pfeiler, who is currently working on a longer-term follow-up study of the PALLAS group.
The analysis found no significant correlation between weight and occurrence of invasive disease events.
Dr. Pfeiler disclosed honoraria and grants from Pfizer. The study was funded by Alliance Foundation Trials.
– a potentially deadly outcome that can occur as a result of chemotherapy treatment.
The study was presented at the annual meeting of the American Society of Clinical Oncology.
It is known that body mass index can affect breast cancer risk and prognosis, but it is not known if BMI can affect how well endocrine therapy works in a given patient. In the PALLAS clinical trial, Georg Pfeiler, MD, Medical University of Vienna, and colleagues, conducted an analysis of 5,698 patients with early hormone receptor–positive breast cancer receiving endocrine therapy with or without palbociclib. Dr. Pfeiler found that women who are overweight or obese had significantly less frequent and less severe cases of neutropenia. With fewer or less severe cases of neutropenia, there were also fewer interruptions in palbociclib treatment.
“One explanation for the lower discontinuation rates may be that the distributional volume of the drug is higher in overweight and obese patients leading to lower serum drug concentrations. It could also be influenced by differences in pharmacokinetics with respect to hyperinsulinemia,” said Dr. Pfeiler, who leads the Oncological Breast Outpatient Clinic and Bone Health Outpatient Clinic at the Medical University of Vienna.
The PALLAS trial compared the combination of palbociclib and adjuvant endocrine therapy with endocrine therapy alone in 5,698 women with early breast cancer. Patients were categorized according to BMI as underweight (BMI < 18.5 kg/m2), normal weight (BMI 18.5-24.9), overweight (BMI, 25-29.9), and obese (> 30). The investigators analyzed differences in adverse events, time to early discontinuation of palbociclib, and in time to invasive disease-free survival.
At baseline, of 5,698 patients, 68 (1.2%) were underweight, 2,082 (36.5%) were of normal weight, 1,818 (31.9%) were overweight, and, 1,730 (30.4%) were obese. In patients treated with palbociclib, neutropenia was the primary toxicity leading to treatment discontinuation with rates of 21.1% in normal-weight patients, 14.0% of overweight patients, and 5.9% of obese patients, respectively.
Significantly different rates of all-grade neutropenia were observed in normal weight, overweight, and obese participants with regard to total (88.5%, 85.7% and 74.7%), as well as grade 3 (64.1%, 62.0% and 43.9%) and grade 4 neutropenia (7.0%, 3.6% and 2.0%), respectively. The lower frequency and severity of neutropenia observed in overweight and obese patients was associated with a significantly lower treatment discontinuation rate over time when compared with normal-weight patients (overweight vs. normal weight: HR, 0.73; 95% CI 0.63-0.84; P < .0001, and obese vs. normal weight: HR, 0.65; 95% CI, 0.56-0.75; P < .0001). There was, however, despite these observations, no statistically significant improvement in invasive disease survival with the addition of palbociclib to endocrine therapy in any weight category (normal weight: HR, 0.84; 95% CI 0.63-1.12; overweight: HR, 1.10; 95% CI 0.82-1.49; and obese: HR, 0.95; 95% CI, 0.69-1.30).
“This is an early analysis, and should be interpreted with caution, especially with regard to disease outcomes. The findings may reduce concerns over hematologic side effects in the overweight and obese. In the future there may be an important impact if it turns out after longer-term follow-up that palbociclib has been underdosed in the overweight and obese. We may need BMI-adapted dose management,” said Dr. Pfeiler, who is currently working on a longer-term follow-up study of the PALLAS group.
The analysis found no significant correlation between weight and occurrence of invasive disease events.
Dr. Pfeiler disclosed honoraria and grants from Pfizer. The study was funded by Alliance Foundation Trials.
FROM ASCO 2022
Obesity in adolescence raises risk for adult type 1 diabetes
NEW ORLEANS – Obesity in adolescence is linked to an increased risk for type 1 diabetes onset in adulthood, new research suggests.
These new data, from Israeli military recruits followed for over a decade, suggest that obesity may be playing a causal role in type 1 as well as type 2 diabetes.
The incidence of type 1 diabetes has been increasing by about 2%-3% annually over recent decades, but the reasons aren’t clear. The study is the first to examine the role of obesity in adolescence and type 1 diabetes in young adulthood, and also the first to examine the question of using antibody status as part of the criteria for a type 1 diagnosis.
The findings were reported at the annual scientific sessions of the American Diabetes Association by Gilad Twig, MD, PhD, professor of medicine at Sheba Medical Center, Tel HaShomer, Israel. “For people who might have a high risk for developing type 1 diabetes, these results emphasize the importance of maintaining a normal weight,” he said in an interview. He noted that, although this recommendation applies to everyone, “here it’s becoming more precise for the population – more individualized in the sense that this might specifically help you.”
Naveed Sattar, PhD, professor and honorary consultant in cardiovascular and medical sciences at the University of Glasgow, said in an interview that carrying too much weight “will make the pancreas have to work harder to make insulin to keep the sugar normal. So, if you’re stressing the system and the pancreas is already likely to fail, it will fail faster.”
Clinically, Dr. Sattar said, “Lifestyle does matter to the risk of developing type 1 diabetes. The weighting may be different [from type 2]. The major factor in type 1 is still the genetics, but if you have a family history of type 1 and your genetic potential is greater, you will minimize your risk by staying leaner.”
Study highlights that type 1 is not always ‘juvenile’
In addition to countering the long-held belief that type 1 diabetes is primarily a condition of thin individuals and unrelated to obesity, the data also reinforce the emerging recognition that type 1 diabetes isn’t always “juvenile” and in fact often arises in adulthood.
“About half of all cases of type 1 diabetes develop after age 18. By reputation, people think it’s a disease of children. But it’s begun to grow so that now 50% of cases occur after late adolescence,” noted Dr. Twig.
Dr. Sattar pointed to a UK Biobank study showing that nearly half of all cases of type 1 diabetes arise after age 30 years. “You absolutely can get type 1 in adulthood. It’s not rare.”
Direct correlation seen in otherwise healthy young people
The retrospective nationwide cohort study included 1,426,362 17-year-olds (834,050 male and 592,312 female) who underwent medical evaluation prior to military conscription starting in January 1996 and followed them through 2016. At baseline, none had a history of dysglycemia.
The data were linked with information about adult-onset type 1 diabetes in the Israeli National Diabetes Registry. In all, 777 incident type 1 diabetes cases were recorded over the study period, with a rate of 4.9 cases per 100,000 person-years.
Over a median follow-up of 11.2 years, there was a graded incidence of type 1 diabetes across BMI groups from underweight to obesity, from 3.6 to 8.4 cases per 100,000 person-years.
After adjustment for sex, birth year, age at study entry, education, and cognitive performance with BMI 5th-49th percentiles as the reference, the hazard ratios were 1.05 for the 50th-74th BMI percentiles, 1.41 for 75th-84th, 1.54 for those who were overweight, and 2.05 for those with obesity.
Every 5-unit increment in BMI corresponded to a 35% greater incidence of type 1 diabetes (adjusted hazard ratio 1.35) and every one increment was associated with a 35% greater risk (1.25), both values significant.
Sensitivity analyses resulted in similar findings for those with no other chronic health conditions at baseline. The results also didn’t change in a separate analysis of 574,720 subjects in whom autoantibody data were available to confirm the type 1 diabetes diagnosis.
Hypotheses for mechanisms
The mechanism for the association isn’t clear, but in a simultaneously published article in Diabetologia, Dr. Twig and colleagues outline several hypotheses. One relates to the growing evidence of a link between various autoimmune conditions, which point to the possibility of elevated adipokines and cytokines in obesity diminishing self-tolerance by promoting proinflammatory processes.
The authors cite data from the TrialNet Pathway to Prevention study of relatives of people with type 1 diabetes in which participants who were overweight and obese had an increased risk of islet autoantibody expression. However, not all data have supported this finding.
“Obesity is related to several other autoimmune conditions, so it’s not a complete surprise it might be related to another,” Dr. Twig noted.
Other possibilities include vitamin D deficiency, a high-fat diet, and alterations in gut microbiota.
And then there’s the “accelerator hypothesis,” suggesting that both type 1 and type 2 diabetes result from insulin resistance and genetic background that affect the rate of beta cell loss and the disease phenotype. Dr. Sattar said that the accelerator hypotheses “makes complete sense to me. Because the population is so obese, we’re seeing it more now whereas we might not have seen it 40 years ago when the BMI differentials were far less in society.”
Dr. Twig has no disclosures. Dr. Sattar has consulted for or received lecture fees from Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceutical, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, and Sanofi, and received grant support from AstraZeneca, Boehringer Ingelheim, Novartis, and Roche Diagnostics through his institution.
NEW ORLEANS – Obesity in adolescence is linked to an increased risk for type 1 diabetes onset in adulthood, new research suggests.
These new data, from Israeli military recruits followed for over a decade, suggest that obesity may be playing a causal role in type 1 as well as type 2 diabetes.
The incidence of type 1 diabetes has been increasing by about 2%-3% annually over recent decades, but the reasons aren’t clear. The study is the first to examine the role of obesity in adolescence and type 1 diabetes in young adulthood, and also the first to examine the question of using antibody status as part of the criteria for a type 1 diagnosis.
The findings were reported at the annual scientific sessions of the American Diabetes Association by Gilad Twig, MD, PhD, professor of medicine at Sheba Medical Center, Tel HaShomer, Israel. “For people who might have a high risk for developing type 1 diabetes, these results emphasize the importance of maintaining a normal weight,” he said in an interview. He noted that, although this recommendation applies to everyone, “here it’s becoming more precise for the population – more individualized in the sense that this might specifically help you.”
Naveed Sattar, PhD, professor and honorary consultant in cardiovascular and medical sciences at the University of Glasgow, said in an interview that carrying too much weight “will make the pancreas have to work harder to make insulin to keep the sugar normal. So, if you’re stressing the system and the pancreas is already likely to fail, it will fail faster.”
Clinically, Dr. Sattar said, “Lifestyle does matter to the risk of developing type 1 diabetes. The weighting may be different [from type 2]. The major factor in type 1 is still the genetics, but if you have a family history of type 1 and your genetic potential is greater, you will minimize your risk by staying leaner.”
Study highlights that type 1 is not always ‘juvenile’
In addition to countering the long-held belief that type 1 diabetes is primarily a condition of thin individuals and unrelated to obesity, the data also reinforce the emerging recognition that type 1 diabetes isn’t always “juvenile” and in fact often arises in adulthood.
“About half of all cases of type 1 diabetes develop after age 18. By reputation, people think it’s a disease of children. But it’s begun to grow so that now 50% of cases occur after late adolescence,” noted Dr. Twig.
Dr. Sattar pointed to a UK Biobank study showing that nearly half of all cases of type 1 diabetes arise after age 30 years. “You absolutely can get type 1 in adulthood. It’s not rare.”
Direct correlation seen in otherwise healthy young people
The retrospective nationwide cohort study included 1,426,362 17-year-olds (834,050 male and 592,312 female) who underwent medical evaluation prior to military conscription starting in January 1996 and followed them through 2016. At baseline, none had a history of dysglycemia.
The data were linked with information about adult-onset type 1 diabetes in the Israeli National Diabetes Registry. In all, 777 incident type 1 diabetes cases were recorded over the study period, with a rate of 4.9 cases per 100,000 person-years.
Over a median follow-up of 11.2 years, there was a graded incidence of type 1 diabetes across BMI groups from underweight to obesity, from 3.6 to 8.4 cases per 100,000 person-years.
After adjustment for sex, birth year, age at study entry, education, and cognitive performance with BMI 5th-49th percentiles as the reference, the hazard ratios were 1.05 for the 50th-74th BMI percentiles, 1.41 for 75th-84th, 1.54 for those who were overweight, and 2.05 for those with obesity.
Every 5-unit increment in BMI corresponded to a 35% greater incidence of type 1 diabetes (adjusted hazard ratio 1.35) and every one increment was associated with a 35% greater risk (1.25), both values significant.
Sensitivity analyses resulted in similar findings for those with no other chronic health conditions at baseline. The results also didn’t change in a separate analysis of 574,720 subjects in whom autoantibody data were available to confirm the type 1 diabetes diagnosis.
Hypotheses for mechanisms
The mechanism for the association isn’t clear, but in a simultaneously published article in Diabetologia, Dr. Twig and colleagues outline several hypotheses. One relates to the growing evidence of a link between various autoimmune conditions, which point to the possibility of elevated adipokines and cytokines in obesity diminishing self-tolerance by promoting proinflammatory processes.
The authors cite data from the TrialNet Pathway to Prevention study of relatives of people with type 1 diabetes in which participants who were overweight and obese had an increased risk of islet autoantibody expression. However, not all data have supported this finding.
“Obesity is related to several other autoimmune conditions, so it’s not a complete surprise it might be related to another,” Dr. Twig noted.
Other possibilities include vitamin D deficiency, a high-fat diet, and alterations in gut microbiota.
And then there’s the “accelerator hypothesis,” suggesting that both type 1 and type 2 diabetes result from insulin resistance and genetic background that affect the rate of beta cell loss and the disease phenotype. Dr. Sattar said that the accelerator hypotheses “makes complete sense to me. Because the population is so obese, we’re seeing it more now whereas we might not have seen it 40 years ago when the BMI differentials were far less in society.”
Dr. Twig has no disclosures. Dr. Sattar has consulted for or received lecture fees from Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceutical, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, and Sanofi, and received grant support from AstraZeneca, Boehringer Ingelheim, Novartis, and Roche Diagnostics through his institution.
NEW ORLEANS – Obesity in adolescence is linked to an increased risk for type 1 diabetes onset in adulthood, new research suggests.
These new data, from Israeli military recruits followed for over a decade, suggest that obesity may be playing a causal role in type 1 as well as type 2 diabetes.
The incidence of type 1 diabetes has been increasing by about 2%-3% annually over recent decades, but the reasons aren’t clear. The study is the first to examine the role of obesity in adolescence and type 1 diabetes in young adulthood, and also the first to examine the question of using antibody status as part of the criteria for a type 1 diagnosis.
The findings were reported at the annual scientific sessions of the American Diabetes Association by Gilad Twig, MD, PhD, professor of medicine at Sheba Medical Center, Tel HaShomer, Israel. “For people who might have a high risk for developing type 1 diabetes, these results emphasize the importance of maintaining a normal weight,” he said in an interview. He noted that, although this recommendation applies to everyone, “here it’s becoming more precise for the population – more individualized in the sense that this might specifically help you.”
Naveed Sattar, PhD, professor and honorary consultant in cardiovascular and medical sciences at the University of Glasgow, said in an interview that carrying too much weight “will make the pancreas have to work harder to make insulin to keep the sugar normal. So, if you’re stressing the system and the pancreas is already likely to fail, it will fail faster.”
Clinically, Dr. Sattar said, “Lifestyle does matter to the risk of developing type 1 diabetes. The weighting may be different [from type 2]. The major factor in type 1 is still the genetics, but if you have a family history of type 1 and your genetic potential is greater, you will minimize your risk by staying leaner.”
Study highlights that type 1 is not always ‘juvenile’
In addition to countering the long-held belief that type 1 diabetes is primarily a condition of thin individuals and unrelated to obesity, the data also reinforce the emerging recognition that type 1 diabetes isn’t always “juvenile” and in fact often arises in adulthood.
“About half of all cases of type 1 diabetes develop after age 18. By reputation, people think it’s a disease of children. But it’s begun to grow so that now 50% of cases occur after late adolescence,” noted Dr. Twig.
Dr. Sattar pointed to a UK Biobank study showing that nearly half of all cases of type 1 diabetes arise after age 30 years. “You absolutely can get type 1 in adulthood. It’s not rare.”
Direct correlation seen in otherwise healthy young people
The retrospective nationwide cohort study included 1,426,362 17-year-olds (834,050 male and 592,312 female) who underwent medical evaluation prior to military conscription starting in January 1996 and followed them through 2016. At baseline, none had a history of dysglycemia.
The data were linked with information about adult-onset type 1 diabetes in the Israeli National Diabetes Registry. In all, 777 incident type 1 diabetes cases were recorded over the study period, with a rate of 4.9 cases per 100,000 person-years.
Over a median follow-up of 11.2 years, there was a graded incidence of type 1 diabetes across BMI groups from underweight to obesity, from 3.6 to 8.4 cases per 100,000 person-years.
After adjustment for sex, birth year, age at study entry, education, and cognitive performance with BMI 5th-49th percentiles as the reference, the hazard ratios were 1.05 for the 50th-74th BMI percentiles, 1.41 for 75th-84th, 1.54 for those who were overweight, and 2.05 for those with obesity.
Every 5-unit increment in BMI corresponded to a 35% greater incidence of type 1 diabetes (adjusted hazard ratio 1.35) and every one increment was associated with a 35% greater risk (1.25), both values significant.
Sensitivity analyses resulted in similar findings for those with no other chronic health conditions at baseline. The results also didn’t change in a separate analysis of 574,720 subjects in whom autoantibody data were available to confirm the type 1 diabetes diagnosis.
Hypotheses for mechanisms
The mechanism for the association isn’t clear, but in a simultaneously published article in Diabetologia, Dr. Twig and colleagues outline several hypotheses. One relates to the growing evidence of a link between various autoimmune conditions, which point to the possibility of elevated adipokines and cytokines in obesity diminishing self-tolerance by promoting proinflammatory processes.
The authors cite data from the TrialNet Pathway to Prevention study of relatives of people with type 1 diabetes in which participants who were overweight and obese had an increased risk of islet autoantibody expression. However, not all data have supported this finding.
“Obesity is related to several other autoimmune conditions, so it’s not a complete surprise it might be related to another,” Dr. Twig noted.
Other possibilities include vitamin D deficiency, a high-fat diet, and alterations in gut microbiota.
And then there’s the “accelerator hypothesis,” suggesting that both type 1 and type 2 diabetes result from insulin resistance and genetic background that affect the rate of beta cell loss and the disease phenotype. Dr. Sattar said that the accelerator hypotheses “makes complete sense to me. Because the population is so obese, we’re seeing it more now whereas we might not have seen it 40 years ago when the BMI differentials were far less in society.”
Dr. Twig has no disclosures. Dr. Sattar has consulted for or received lecture fees from Amgen, AstraZeneca, Boehringer Ingelheim, Eli Lilly, Hanmi Pharmaceutical, Merck Sharp & Dohme, Novartis, Novo Nordisk, Pfizer, and Sanofi, and received grant support from AstraZeneca, Boehringer Ingelheim, Novartis, and Roche Diagnostics through his institution.
AT ADA 2022
Tirzepatide powers ‘unprecedented’ weight loss in SURMOUNT-1
NEW ORLEANS – Treatment of people with obesity but no diabetes with the dual–incretin agonist tirzepatide safely produced “unprecedented” levels of weight loss in the vast majority of patients in SURMOUNT-1, a placebo-controlled trial with more than 2,500 people with obesity or overweight plus at least one weight-related complication.
Although the pivotal trial did not directly compare weekly subcutaneous injection with the twincretin tirzepatide (at 5 mg, 10 mg, or 15 mg) with either bariatric surgery or what has been the reigning champ of weight-loss agents, a 2.4-mg/week injection of semaglutide (Wegovy), the new findings are impressive because they eclipsed semaglutide’s past performance in at least three important ways, said Ania M. Jastreboff, MD, PhD, SURMOUNT-1’s lead investigator, at the annual scientific sessions of the American Diabetes Association.
First, the highest-tested dosage of tirzepatide, 15 mg/week, for 72 weeks, produced a 5% or greater loss in baseline weight in 91%-96% of patients, an effect “not previously seen” in any prior phase 3 trial of a weight-loss agent, noted Dr. Jastreboff, an endocrinologist and director of Weight Management & Obesity Prevention at Yale University in New Haven, Conn.
Second, the average level of weight loss among the 630 people who received 15 mg/week was 22.5% in the on-treatment analysis, and 20.9% in the intention-to-treat analysis, again a magnitude of effect never before seen with any other medical intervention.
And in an exploratory analysis, 40% of people who received the highest-tested tirzepatide dose of 15 mg/week had at least a 25% loss in baseline weight in the on-treatment analysis, another example of unprecedented weight-loss achievement, said Dr. Jastreboff.
Looking at the data another way, the average baseline weight of those in the trial was 104 kg (230 lb) at the start, and the average weight loss was between 35 and 52 lbs by 72 weeks on treatment, Dr. Jastreboff said in a press conference.
She noted, however, that not everyone will respond to tirzepatide, “but if you do respond to this medicine, you will feel full earlier, you won’t want to go back for seconds, and you may eat smaller amounts more often.”
Such weight-loss agents will need to be taken chronically, in the same way that medications are for hypertension or dyslipidemia, Dr. Jastreboff stressed. “If you stop the antiobesity medication then the body fat mass set point will go back up so this necessitates long-term treatment.”
A new era: Weight loss ‘in the range of bariatric surgery’
Tirzepatide, developed by Lilly, has recently been approved in the United States for the treatment of type 2 diabetes, under the brand name Mounjaro.
SURMOUNT-1 was designed to examine the effect of the agent in overweight/obesity, and the company will be filing for the additional indication of weight loss in the future. Top-line results of SURMOUNT-1 generated much excitement when Lilly reported them back in April, including a story in The New York Times.
Semaglutide, a Novo Nordisk drug, is approved in the United States for type 2 diabetes (as Ozempic at doses of either 1 mg or 2 mg per week) and also for weight loss, as Wegovy, at the higher dose of 2.4 mg per week. When Wegovy was given the green light by the Food and Drug Administration a year ago, it too was hailed as a “game changer” for obesity.
The weight-loss results seen in SURMOUNT-1 “put tirzepatide squarely in the range of weight loss achieved with bariatric surgery,” concluded Louis J. Aronne, MD, a coinvestigator on the trial, professor at Weill-Cornell Medicine in New York, and director of the Center for Weight Management and Metabolic Clinical Research of Weill-Cornell.
The results are “amazing,” and propel the weight-loss field into “a new era of obesity treatment,” commented Lee M. Kaplan, MD, who was not involved in the study and served as designated discussant for the trial.
Despite the lack of direct comparison, the findings indicate that “tirzepatide causes more weight loss than semaglutide,” and it provides “an opportunity to meet or exceed” the weight-loss effects of bariatric surgery, added Dr. Kaplan, director of the Obesity, Metabolism and Nutrition Institute at Massachusetts General Hospital in Boston.
Simultaneously with Dr. Jastreboff’s report at the meeting, the results were published online in The New England Journal of Medicine.
An accompanying editorial agrees with Dr. Kaplan: “It is remarkable that the magnitude of weight loss with tirzepatide was similar to that with gastric bypass, which raises the potential for alternative medical approaches to the treatment of obesity.”
“The tides are shifting, and there are now more options for people with obesity to lose weight,” write Clifford J. Rosen, MD, of Tufts University, Boston, and Julie R. Ingelfinger, MD, of Harvard University and Massachusetts General Hospital, Boston.
Dual incretin agonism ‘enhances activity,’ says expert
Tirzepatide is the first agent on the U.S. market from a novel class of dual-incretin agonists, with a molecular structure engineered to activate both the glucagonlike protein-1 (GLP-1) receptor and the glucose-dependent insulinotropic polypeptide (GIP), the two predominant incretins in the human gut. This combined activity has led to the twincretin nickname for tirzepatide.
Semaglutide is a single-incretin agonist, with its activity focused exclusively on the GLP-1 receptor.
Dr. Aronne tied the apparently superior efficacy of tirzepatide relative to semaglutide directly to the added incretin activity of tirzepatide. “The dual approach enhances efficacy,” he proposed during his presentation at the meeting.
The impressive efficacy and reassuring safety profile reported from SURMOUNT-1 opens the door to a new approach to treating obesity, which in the past has often taken a back seat to treatments for dyslipidemia, hypertension, and diabetes.
“Now that we can treat obesity safely and effectively, it makes sense to treat obesity first,” Dr. Aronne recommended.
Dr. Jastreboff agreed: “Perhaps we can prevent diabetes by treating obesity head-on,” she remarked.
Weight-loss agents gain U.S. traction
There have been concerns about patient access to these newer weight-loss drugs in the United States, given that the retail cost of semaglutide for obesity exceeds $1,000/month, but Dr. Aronne reported data that painted a more optimistic picture.
His numbers showed that during the first months that semaglutide was on the U.S. market as a weight-loss agent, the number of U.S. prescriptions written for branded antiobesity medications roughly doubled, a spike that seemed mostly driven by the introduction and growing use of semaglutide.
With tirzepatide, every prespecified cardiometabolic parameter assessed in the trial showed clinically meaningful improvements, reported Dr. Jastreboff, including an average 17% reduction in waist circumference in patients on either of the highest two dosages, a 34% average drop in total fat mass, an average 0.5–percentage point cut in baseline hemoglobin A1c at the highest two dosages, substantial cuts in fasting plasma glucose and fasting insulin levels, an average 28% drop in triglyceride levels, and an average systolic blood pressure reduction of about 8 mm Hg that occurred within 24 weeks on treatment.
“I think that insurers will sign up” for tirzepatide coverage based on benefits like this, Dr. Aronne predicted.
SURMOUNT-1 randomized 2,539 patients with obesity or with overweight plus at least one weight-related complication at any of 119 sites in nine countries. They had a body mass index of 30 kg/m2 or more, or 27 kg/m2 or more and at least one weight-related complication, excluding diabetes. They were randomized in a 1:1:1:1 ratio to receive once-weekly, subcutaneous tirzepatide (5 mg, 10 mg, or 15 mg) or placebo for 72 weeks, including a 20-week dose-escalation period.
The study’s two primary endpoints were the average percentage change in body weight from entry to 72 weeks, and the percentage of participants reaching at least a 5% reduction in their baseline body weight by 72 weeks.
The most common adverse events with tirzepatide were gastrointestinal, and most were mild to moderate in severity, occurring primarily during dose escalation. Adverse events caused treatment discontinuation in 4.3%, 7.1%, 6.2%, and 2.6% of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses and placebo, respectively
The trial ran from December 2019 to April 2022, so during the peak of the COVID-19 pandemic, which Dr. Jastreboff described as an “amazing feat.”
Jamy Ard, MD, who chaired the SURMOUNT-1 session quipped, after hearing the results, “Wow; that’s exciting. If you’re not excited by the results, you’d better check your pulse.”
Dr. Ard is a professor at Wake Forest University, Winston-Salem, N.C., and codirector of the Wake Forest Baptist Health Weight Management Center in Winston-Salem.
SURMOUNT-1 was sponsored by Eli Lilly, the company that markets tirzepatide (Mounjaro). Dr. Jastreboff has been an advisor or consultant to Eli Lilly, as well as to Boehringer Ingelheim, Intellihealth, Novo Nordisk, Pfizer, Rhythm Pharmaceuticals, Scholar Rock, and Weight Watchers, and she has received research funding from Eli Lilly and Novo Nordisk. Dr. Aronne has been a consultant or advisor to, speaker on behalf of, or received research funding from Eli Lilly as well as from Altimmune, Amgen, Allurion, Intellihealth, Janssen, Novo Nordisk, Pfizer, and United Health group; he has an ownership interest in ERX, Gelesis, and Intellihealth; and he serves on the board of ERX, Jamieson Wellness, and Intellihealth. Dr. Kaplan has been a consultant to Eli Lilly, as well as to Amgen, Boehringer Ingelheim, Gelesis, Gilead, Novo Nordisk, Optum Health, Pfizer, Rhythm Pharmaceuticals, the Obesity and Nutrition Institute, and Xeno Biosciences. Dr. Ard has been a consultant to Eli Lilly, as well as to Nestle Health Sciences and Novo Nordisk, and he has received research funding from Boehringer Ingelheim, Epitomee, Medical, and United Health Group.
A version of this article first appeared on Medscape.com.
NEW ORLEANS – Treatment of people with obesity but no diabetes with the dual–incretin agonist tirzepatide safely produced “unprecedented” levels of weight loss in the vast majority of patients in SURMOUNT-1, a placebo-controlled trial with more than 2,500 people with obesity or overweight plus at least one weight-related complication.
Although the pivotal trial did not directly compare weekly subcutaneous injection with the twincretin tirzepatide (at 5 mg, 10 mg, or 15 mg) with either bariatric surgery or what has been the reigning champ of weight-loss agents, a 2.4-mg/week injection of semaglutide (Wegovy), the new findings are impressive because they eclipsed semaglutide’s past performance in at least three important ways, said Ania M. Jastreboff, MD, PhD, SURMOUNT-1’s lead investigator, at the annual scientific sessions of the American Diabetes Association.
First, the highest-tested dosage of tirzepatide, 15 mg/week, for 72 weeks, produced a 5% or greater loss in baseline weight in 91%-96% of patients, an effect “not previously seen” in any prior phase 3 trial of a weight-loss agent, noted Dr. Jastreboff, an endocrinologist and director of Weight Management & Obesity Prevention at Yale University in New Haven, Conn.
Second, the average level of weight loss among the 630 people who received 15 mg/week was 22.5% in the on-treatment analysis, and 20.9% in the intention-to-treat analysis, again a magnitude of effect never before seen with any other medical intervention.
And in an exploratory analysis, 40% of people who received the highest-tested tirzepatide dose of 15 mg/week had at least a 25% loss in baseline weight in the on-treatment analysis, another example of unprecedented weight-loss achievement, said Dr. Jastreboff.
Looking at the data another way, the average baseline weight of those in the trial was 104 kg (230 lb) at the start, and the average weight loss was between 35 and 52 lbs by 72 weeks on treatment, Dr. Jastreboff said in a press conference.
She noted, however, that not everyone will respond to tirzepatide, “but if you do respond to this medicine, you will feel full earlier, you won’t want to go back for seconds, and you may eat smaller amounts more often.”
Such weight-loss agents will need to be taken chronically, in the same way that medications are for hypertension or dyslipidemia, Dr. Jastreboff stressed. “If you stop the antiobesity medication then the body fat mass set point will go back up so this necessitates long-term treatment.”
A new era: Weight loss ‘in the range of bariatric surgery’
Tirzepatide, developed by Lilly, has recently been approved in the United States for the treatment of type 2 diabetes, under the brand name Mounjaro.
SURMOUNT-1 was designed to examine the effect of the agent in overweight/obesity, and the company will be filing for the additional indication of weight loss in the future. Top-line results of SURMOUNT-1 generated much excitement when Lilly reported them back in April, including a story in The New York Times.
Semaglutide, a Novo Nordisk drug, is approved in the United States for type 2 diabetes (as Ozempic at doses of either 1 mg or 2 mg per week) and also for weight loss, as Wegovy, at the higher dose of 2.4 mg per week. When Wegovy was given the green light by the Food and Drug Administration a year ago, it too was hailed as a “game changer” for obesity.
The weight-loss results seen in SURMOUNT-1 “put tirzepatide squarely in the range of weight loss achieved with bariatric surgery,” concluded Louis J. Aronne, MD, a coinvestigator on the trial, professor at Weill-Cornell Medicine in New York, and director of the Center for Weight Management and Metabolic Clinical Research of Weill-Cornell.
The results are “amazing,” and propel the weight-loss field into “a new era of obesity treatment,” commented Lee M. Kaplan, MD, who was not involved in the study and served as designated discussant for the trial.
Despite the lack of direct comparison, the findings indicate that “tirzepatide causes more weight loss than semaglutide,” and it provides “an opportunity to meet or exceed” the weight-loss effects of bariatric surgery, added Dr. Kaplan, director of the Obesity, Metabolism and Nutrition Institute at Massachusetts General Hospital in Boston.
Simultaneously with Dr. Jastreboff’s report at the meeting, the results were published online in The New England Journal of Medicine.
An accompanying editorial agrees with Dr. Kaplan: “It is remarkable that the magnitude of weight loss with tirzepatide was similar to that with gastric bypass, which raises the potential for alternative medical approaches to the treatment of obesity.”
“The tides are shifting, and there are now more options for people with obesity to lose weight,” write Clifford J. Rosen, MD, of Tufts University, Boston, and Julie R. Ingelfinger, MD, of Harvard University and Massachusetts General Hospital, Boston.
Dual incretin agonism ‘enhances activity,’ says expert
Tirzepatide is the first agent on the U.S. market from a novel class of dual-incretin agonists, with a molecular structure engineered to activate both the glucagonlike protein-1 (GLP-1) receptor and the glucose-dependent insulinotropic polypeptide (GIP), the two predominant incretins in the human gut. This combined activity has led to the twincretin nickname for tirzepatide.
Semaglutide is a single-incretin agonist, with its activity focused exclusively on the GLP-1 receptor.
Dr. Aronne tied the apparently superior efficacy of tirzepatide relative to semaglutide directly to the added incretin activity of tirzepatide. “The dual approach enhances efficacy,” he proposed during his presentation at the meeting.
The impressive efficacy and reassuring safety profile reported from SURMOUNT-1 opens the door to a new approach to treating obesity, which in the past has often taken a back seat to treatments for dyslipidemia, hypertension, and diabetes.
“Now that we can treat obesity safely and effectively, it makes sense to treat obesity first,” Dr. Aronne recommended.
Dr. Jastreboff agreed: “Perhaps we can prevent diabetes by treating obesity head-on,” she remarked.
Weight-loss agents gain U.S. traction
There have been concerns about patient access to these newer weight-loss drugs in the United States, given that the retail cost of semaglutide for obesity exceeds $1,000/month, but Dr. Aronne reported data that painted a more optimistic picture.
His numbers showed that during the first months that semaglutide was on the U.S. market as a weight-loss agent, the number of U.S. prescriptions written for branded antiobesity medications roughly doubled, a spike that seemed mostly driven by the introduction and growing use of semaglutide.
With tirzepatide, every prespecified cardiometabolic parameter assessed in the trial showed clinically meaningful improvements, reported Dr. Jastreboff, including an average 17% reduction in waist circumference in patients on either of the highest two dosages, a 34% average drop in total fat mass, an average 0.5–percentage point cut in baseline hemoglobin A1c at the highest two dosages, substantial cuts in fasting plasma glucose and fasting insulin levels, an average 28% drop in triglyceride levels, and an average systolic blood pressure reduction of about 8 mm Hg that occurred within 24 weeks on treatment.
“I think that insurers will sign up” for tirzepatide coverage based on benefits like this, Dr. Aronne predicted.
SURMOUNT-1 randomized 2,539 patients with obesity or with overweight plus at least one weight-related complication at any of 119 sites in nine countries. They had a body mass index of 30 kg/m2 or more, or 27 kg/m2 or more and at least one weight-related complication, excluding diabetes. They were randomized in a 1:1:1:1 ratio to receive once-weekly, subcutaneous tirzepatide (5 mg, 10 mg, or 15 mg) or placebo for 72 weeks, including a 20-week dose-escalation period.
The study’s two primary endpoints were the average percentage change in body weight from entry to 72 weeks, and the percentage of participants reaching at least a 5% reduction in their baseline body weight by 72 weeks.
The most common adverse events with tirzepatide were gastrointestinal, and most were mild to moderate in severity, occurring primarily during dose escalation. Adverse events caused treatment discontinuation in 4.3%, 7.1%, 6.2%, and 2.6% of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses and placebo, respectively
The trial ran from December 2019 to April 2022, so during the peak of the COVID-19 pandemic, which Dr. Jastreboff described as an “amazing feat.”
Jamy Ard, MD, who chaired the SURMOUNT-1 session quipped, after hearing the results, “Wow; that’s exciting. If you’re not excited by the results, you’d better check your pulse.”
Dr. Ard is a professor at Wake Forest University, Winston-Salem, N.C., and codirector of the Wake Forest Baptist Health Weight Management Center in Winston-Salem.
SURMOUNT-1 was sponsored by Eli Lilly, the company that markets tirzepatide (Mounjaro). Dr. Jastreboff has been an advisor or consultant to Eli Lilly, as well as to Boehringer Ingelheim, Intellihealth, Novo Nordisk, Pfizer, Rhythm Pharmaceuticals, Scholar Rock, and Weight Watchers, and she has received research funding from Eli Lilly and Novo Nordisk. Dr. Aronne has been a consultant or advisor to, speaker on behalf of, or received research funding from Eli Lilly as well as from Altimmune, Amgen, Allurion, Intellihealth, Janssen, Novo Nordisk, Pfizer, and United Health group; he has an ownership interest in ERX, Gelesis, and Intellihealth; and he serves on the board of ERX, Jamieson Wellness, and Intellihealth. Dr. Kaplan has been a consultant to Eli Lilly, as well as to Amgen, Boehringer Ingelheim, Gelesis, Gilead, Novo Nordisk, Optum Health, Pfizer, Rhythm Pharmaceuticals, the Obesity and Nutrition Institute, and Xeno Biosciences. Dr. Ard has been a consultant to Eli Lilly, as well as to Nestle Health Sciences and Novo Nordisk, and he has received research funding from Boehringer Ingelheim, Epitomee, Medical, and United Health Group.
A version of this article first appeared on Medscape.com.
NEW ORLEANS – Treatment of people with obesity but no diabetes with the dual–incretin agonist tirzepatide safely produced “unprecedented” levels of weight loss in the vast majority of patients in SURMOUNT-1, a placebo-controlled trial with more than 2,500 people with obesity or overweight plus at least one weight-related complication.
Although the pivotal trial did not directly compare weekly subcutaneous injection with the twincretin tirzepatide (at 5 mg, 10 mg, or 15 mg) with either bariatric surgery or what has been the reigning champ of weight-loss agents, a 2.4-mg/week injection of semaglutide (Wegovy), the new findings are impressive because they eclipsed semaglutide’s past performance in at least three important ways, said Ania M. Jastreboff, MD, PhD, SURMOUNT-1’s lead investigator, at the annual scientific sessions of the American Diabetes Association.
First, the highest-tested dosage of tirzepatide, 15 mg/week, for 72 weeks, produced a 5% or greater loss in baseline weight in 91%-96% of patients, an effect “not previously seen” in any prior phase 3 trial of a weight-loss agent, noted Dr. Jastreboff, an endocrinologist and director of Weight Management & Obesity Prevention at Yale University in New Haven, Conn.
Second, the average level of weight loss among the 630 people who received 15 mg/week was 22.5% in the on-treatment analysis, and 20.9% in the intention-to-treat analysis, again a magnitude of effect never before seen with any other medical intervention.
And in an exploratory analysis, 40% of people who received the highest-tested tirzepatide dose of 15 mg/week had at least a 25% loss in baseline weight in the on-treatment analysis, another example of unprecedented weight-loss achievement, said Dr. Jastreboff.
Looking at the data another way, the average baseline weight of those in the trial was 104 kg (230 lb) at the start, and the average weight loss was between 35 and 52 lbs by 72 weeks on treatment, Dr. Jastreboff said in a press conference.
She noted, however, that not everyone will respond to tirzepatide, “but if you do respond to this medicine, you will feel full earlier, you won’t want to go back for seconds, and you may eat smaller amounts more often.”
Such weight-loss agents will need to be taken chronically, in the same way that medications are for hypertension or dyslipidemia, Dr. Jastreboff stressed. “If you stop the antiobesity medication then the body fat mass set point will go back up so this necessitates long-term treatment.”
A new era: Weight loss ‘in the range of bariatric surgery’
Tirzepatide, developed by Lilly, has recently been approved in the United States for the treatment of type 2 diabetes, under the brand name Mounjaro.
SURMOUNT-1 was designed to examine the effect of the agent in overweight/obesity, and the company will be filing for the additional indication of weight loss in the future. Top-line results of SURMOUNT-1 generated much excitement when Lilly reported them back in April, including a story in The New York Times.
Semaglutide, a Novo Nordisk drug, is approved in the United States for type 2 diabetes (as Ozempic at doses of either 1 mg or 2 mg per week) and also for weight loss, as Wegovy, at the higher dose of 2.4 mg per week. When Wegovy was given the green light by the Food and Drug Administration a year ago, it too was hailed as a “game changer” for obesity.
The weight-loss results seen in SURMOUNT-1 “put tirzepatide squarely in the range of weight loss achieved with bariatric surgery,” concluded Louis J. Aronne, MD, a coinvestigator on the trial, professor at Weill-Cornell Medicine in New York, and director of the Center for Weight Management and Metabolic Clinical Research of Weill-Cornell.
The results are “amazing,” and propel the weight-loss field into “a new era of obesity treatment,” commented Lee M. Kaplan, MD, who was not involved in the study and served as designated discussant for the trial.
Despite the lack of direct comparison, the findings indicate that “tirzepatide causes more weight loss than semaglutide,” and it provides “an opportunity to meet or exceed” the weight-loss effects of bariatric surgery, added Dr. Kaplan, director of the Obesity, Metabolism and Nutrition Institute at Massachusetts General Hospital in Boston.
Simultaneously with Dr. Jastreboff’s report at the meeting, the results were published online in The New England Journal of Medicine.
An accompanying editorial agrees with Dr. Kaplan: “It is remarkable that the magnitude of weight loss with tirzepatide was similar to that with gastric bypass, which raises the potential for alternative medical approaches to the treatment of obesity.”
“The tides are shifting, and there are now more options for people with obesity to lose weight,” write Clifford J. Rosen, MD, of Tufts University, Boston, and Julie R. Ingelfinger, MD, of Harvard University and Massachusetts General Hospital, Boston.
Dual incretin agonism ‘enhances activity,’ says expert
Tirzepatide is the first agent on the U.S. market from a novel class of dual-incretin agonists, with a molecular structure engineered to activate both the glucagonlike protein-1 (GLP-1) receptor and the glucose-dependent insulinotropic polypeptide (GIP), the two predominant incretins in the human gut. This combined activity has led to the twincretin nickname for tirzepatide.
Semaglutide is a single-incretin agonist, with its activity focused exclusively on the GLP-1 receptor.
Dr. Aronne tied the apparently superior efficacy of tirzepatide relative to semaglutide directly to the added incretin activity of tirzepatide. “The dual approach enhances efficacy,” he proposed during his presentation at the meeting.
The impressive efficacy and reassuring safety profile reported from SURMOUNT-1 opens the door to a new approach to treating obesity, which in the past has often taken a back seat to treatments for dyslipidemia, hypertension, and diabetes.
“Now that we can treat obesity safely and effectively, it makes sense to treat obesity first,” Dr. Aronne recommended.
Dr. Jastreboff agreed: “Perhaps we can prevent diabetes by treating obesity head-on,” she remarked.
Weight-loss agents gain U.S. traction
There have been concerns about patient access to these newer weight-loss drugs in the United States, given that the retail cost of semaglutide for obesity exceeds $1,000/month, but Dr. Aronne reported data that painted a more optimistic picture.
His numbers showed that during the first months that semaglutide was on the U.S. market as a weight-loss agent, the number of U.S. prescriptions written for branded antiobesity medications roughly doubled, a spike that seemed mostly driven by the introduction and growing use of semaglutide.
With tirzepatide, every prespecified cardiometabolic parameter assessed in the trial showed clinically meaningful improvements, reported Dr. Jastreboff, including an average 17% reduction in waist circumference in patients on either of the highest two dosages, a 34% average drop in total fat mass, an average 0.5–percentage point cut in baseline hemoglobin A1c at the highest two dosages, substantial cuts in fasting plasma glucose and fasting insulin levels, an average 28% drop in triglyceride levels, and an average systolic blood pressure reduction of about 8 mm Hg that occurred within 24 weeks on treatment.
“I think that insurers will sign up” for tirzepatide coverage based on benefits like this, Dr. Aronne predicted.
SURMOUNT-1 randomized 2,539 patients with obesity or with overweight plus at least one weight-related complication at any of 119 sites in nine countries. They had a body mass index of 30 kg/m2 or more, or 27 kg/m2 or more and at least one weight-related complication, excluding diabetes. They were randomized in a 1:1:1:1 ratio to receive once-weekly, subcutaneous tirzepatide (5 mg, 10 mg, or 15 mg) or placebo for 72 weeks, including a 20-week dose-escalation period.
The study’s two primary endpoints were the average percentage change in body weight from entry to 72 weeks, and the percentage of participants reaching at least a 5% reduction in their baseline body weight by 72 weeks.
The most common adverse events with tirzepatide were gastrointestinal, and most were mild to moderate in severity, occurring primarily during dose escalation. Adverse events caused treatment discontinuation in 4.3%, 7.1%, 6.2%, and 2.6% of participants receiving 5-mg, 10-mg, and 15-mg tirzepatide doses and placebo, respectively
The trial ran from December 2019 to April 2022, so during the peak of the COVID-19 pandemic, which Dr. Jastreboff described as an “amazing feat.”
Jamy Ard, MD, who chaired the SURMOUNT-1 session quipped, after hearing the results, “Wow; that’s exciting. If you’re not excited by the results, you’d better check your pulse.”
Dr. Ard is a professor at Wake Forest University, Winston-Salem, N.C., and codirector of the Wake Forest Baptist Health Weight Management Center in Winston-Salem.
SURMOUNT-1 was sponsored by Eli Lilly, the company that markets tirzepatide (Mounjaro). Dr. Jastreboff has been an advisor or consultant to Eli Lilly, as well as to Boehringer Ingelheim, Intellihealth, Novo Nordisk, Pfizer, Rhythm Pharmaceuticals, Scholar Rock, and Weight Watchers, and she has received research funding from Eli Lilly and Novo Nordisk. Dr. Aronne has been a consultant or advisor to, speaker on behalf of, or received research funding from Eli Lilly as well as from Altimmune, Amgen, Allurion, Intellihealth, Janssen, Novo Nordisk, Pfizer, and United Health group; he has an ownership interest in ERX, Gelesis, and Intellihealth; and he serves on the board of ERX, Jamieson Wellness, and Intellihealth. Dr. Kaplan has been a consultant to Eli Lilly, as well as to Amgen, Boehringer Ingelheim, Gelesis, Gilead, Novo Nordisk, Optum Health, Pfizer, Rhythm Pharmaceuticals, the Obesity and Nutrition Institute, and Xeno Biosciences. Dr. Ard has been a consultant to Eli Lilly, as well as to Nestle Health Sciences and Novo Nordisk, and he has received research funding from Boehringer Ingelheim, Epitomee, Medical, and United Health Group.
A version of this article first appeared on Medscape.com.
AT ADA 2022
Neoadjuvant denosumab ineffective in breast cancer
Results from a new phase IIb, 2x2 randomized, open-label trial (GeparSEPTO) showed no improvement in pathologic complete response (pCR) rates when the RANKL inhibitor denosumab was added to anthracycline/taxane-based neoadjuvant chemotherapy in the treatment of primary breast cancer. The study found that a weekly anthracycline/taxane-based neoadjuvant chemotherapy led to improved pCR compared to an every 3-week schedule both overall and in patients with triple-negative breast cancer (TNBC), though it was associated with greater toxicity.
“Currently, I do not see a place for antiosteolytic agents as part of the neoadjuvant therapy in breast cancer,” said lead author Sibylle Loibl, MD, PhD, who is a breast cancer researcher at Goethe (Germany) University. The researchers can’t exclude the possibility of a long-term benefit, and patients will be followed for disease-free and overall survival.
, prompting optimism that the agents might improve pCR rates and improve survival rates in the neoadjuvant setting.
The failure may be because of the shorter treatment duration, and it’s possible that pCR is not the best endpoint to study for a drug that has long-acting potential, according to Dr. Loibl.
Patients were randomized to 120 mg denosumab every 4 weeks for six cycles or to receive no supplementary treatment. Patients with or without denosumab either received nab-paclitaxel 125 mg/m2 weekly for 12 weeks or on days 1 and 8 every 3 weeks, over four cycles (eight total doses), then epirubicin/cyclophosphamide, 90/600 mg/m2 (every 2 and 3 weeks, respectively). Patients with triple-negative breast cancer also received carboplatin. Patients with ERBB2-positive breast cancer received the trastuzumab biosimilar ABP980 plus pertuzumab.
The study included 780 patients (1 male), with a median age of 49.0 years. There was no difference in pCR among denosumab recipients and nonrecipients (41.0% versus 42.8%; P = .58). Weekly nab-paclitaxel led to a higher pCR rate than a schedule of days 1 and 8 every 3 weeks (44.9% versus 39.0%; P = .06, significance level of alpha = .10). Among subgroups, there was only a difference in pCR rates among those with TNBC (60.4% versus 50.0%; P = .06). Grade 3-4 toxic effects were similar regardless of denosumab exposure, but nonhematologic grade 3-4 toxicity was higher with weekly nab-paclitaxel (33.7% versus 24.1%; P = .004).
“The overall pCR difference (with nab-paclitaxel as neoadjuvant chemotherapy) seems small, but looking at the data from the GeparSEPTO study we would expect a transformation into a better invasive disease-free survival. Looking specifically into patients with TNBC, there is clear pCR increase by using the weekly regimen. With just over 60%, this is the highest pCR rate reported so far for a chemotherapy-only regimen. I would prefer to use nab-paclitaxel also in early breast cancer and would use the weekly regimen for women with TNBC who are at high risk,” Dr. Loibl said.
The study was limited by an imbalance of tumor subtypes between the treatment groups. The authors wrote that the results should guide further research, but nab-paclitaxel should not currently be viewed as a standard neoadjuvant treatment of breast cancer.
The study was funded by the German Breast Group, Bristol Myers Squibb, and Amgen. Dr. Loibl has served on the advisory boards or given lectures for AbbVie, Amgen, Bayer, Celgene, EirGenix, GSK, Lilly, Merck, Puma, Seagen, AstraZeneca, Daiichi Sankyo, Novartis, Pierre Fabre, Prime/Medscape, Bristol Myers Squibb, Chugai, Ipsen, Roche, and Samsung. She also holds a related patent.
Results from a new phase IIb, 2x2 randomized, open-label trial (GeparSEPTO) showed no improvement in pathologic complete response (pCR) rates when the RANKL inhibitor denosumab was added to anthracycline/taxane-based neoadjuvant chemotherapy in the treatment of primary breast cancer. The study found that a weekly anthracycline/taxane-based neoadjuvant chemotherapy led to improved pCR compared to an every 3-week schedule both overall and in patients with triple-negative breast cancer (TNBC), though it was associated with greater toxicity.
“Currently, I do not see a place for antiosteolytic agents as part of the neoadjuvant therapy in breast cancer,” said lead author Sibylle Loibl, MD, PhD, who is a breast cancer researcher at Goethe (Germany) University. The researchers can’t exclude the possibility of a long-term benefit, and patients will be followed for disease-free and overall survival.
, prompting optimism that the agents might improve pCR rates and improve survival rates in the neoadjuvant setting.
The failure may be because of the shorter treatment duration, and it’s possible that pCR is not the best endpoint to study for a drug that has long-acting potential, according to Dr. Loibl.
Patients were randomized to 120 mg denosumab every 4 weeks for six cycles or to receive no supplementary treatment. Patients with or without denosumab either received nab-paclitaxel 125 mg/m2 weekly for 12 weeks or on days 1 and 8 every 3 weeks, over four cycles (eight total doses), then epirubicin/cyclophosphamide, 90/600 mg/m2 (every 2 and 3 weeks, respectively). Patients with triple-negative breast cancer also received carboplatin. Patients with ERBB2-positive breast cancer received the trastuzumab biosimilar ABP980 plus pertuzumab.
The study included 780 patients (1 male), with a median age of 49.0 years. There was no difference in pCR among denosumab recipients and nonrecipients (41.0% versus 42.8%; P = .58). Weekly nab-paclitaxel led to a higher pCR rate than a schedule of days 1 and 8 every 3 weeks (44.9% versus 39.0%; P = .06, significance level of alpha = .10). Among subgroups, there was only a difference in pCR rates among those with TNBC (60.4% versus 50.0%; P = .06). Grade 3-4 toxic effects were similar regardless of denosumab exposure, but nonhematologic grade 3-4 toxicity was higher with weekly nab-paclitaxel (33.7% versus 24.1%; P = .004).
“The overall pCR difference (with nab-paclitaxel as neoadjuvant chemotherapy) seems small, but looking at the data from the GeparSEPTO study we would expect a transformation into a better invasive disease-free survival. Looking specifically into patients with TNBC, there is clear pCR increase by using the weekly regimen. With just over 60%, this is the highest pCR rate reported so far for a chemotherapy-only regimen. I would prefer to use nab-paclitaxel also in early breast cancer and would use the weekly regimen for women with TNBC who are at high risk,” Dr. Loibl said.
The study was limited by an imbalance of tumor subtypes between the treatment groups. The authors wrote that the results should guide further research, but nab-paclitaxel should not currently be viewed as a standard neoadjuvant treatment of breast cancer.
The study was funded by the German Breast Group, Bristol Myers Squibb, and Amgen. Dr. Loibl has served on the advisory boards or given lectures for AbbVie, Amgen, Bayer, Celgene, EirGenix, GSK, Lilly, Merck, Puma, Seagen, AstraZeneca, Daiichi Sankyo, Novartis, Pierre Fabre, Prime/Medscape, Bristol Myers Squibb, Chugai, Ipsen, Roche, and Samsung. She also holds a related patent.
Results from a new phase IIb, 2x2 randomized, open-label trial (GeparSEPTO) showed no improvement in pathologic complete response (pCR) rates when the RANKL inhibitor denosumab was added to anthracycline/taxane-based neoadjuvant chemotherapy in the treatment of primary breast cancer. The study found that a weekly anthracycline/taxane-based neoadjuvant chemotherapy led to improved pCR compared to an every 3-week schedule both overall and in patients with triple-negative breast cancer (TNBC), though it was associated with greater toxicity.
“Currently, I do not see a place for antiosteolytic agents as part of the neoadjuvant therapy in breast cancer,” said lead author Sibylle Loibl, MD, PhD, who is a breast cancer researcher at Goethe (Germany) University. The researchers can’t exclude the possibility of a long-term benefit, and patients will be followed for disease-free and overall survival.
, prompting optimism that the agents might improve pCR rates and improve survival rates in the neoadjuvant setting.
The failure may be because of the shorter treatment duration, and it’s possible that pCR is not the best endpoint to study for a drug that has long-acting potential, according to Dr. Loibl.
Patients were randomized to 120 mg denosumab every 4 weeks for six cycles or to receive no supplementary treatment. Patients with or without denosumab either received nab-paclitaxel 125 mg/m2 weekly for 12 weeks or on days 1 and 8 every 3 weeks, over four cycles (eight total doses), then epirubicin/cyclophosphamide, 90/600 mg/m2 (every 2 and 3 weeks, respectively). Patients with triple-negative breast cancer also received carboplatin. Patients with ERBB2-positive breast cancer received the trastuzumab biosimilar ABP980 plus pertuzumab.
The study included 780 patients (1 male), with a median age of 49.0 years. There was no difference in pCR among denosumab recipients and nonrecipients (41.0% versus 42.8%; P = .58). Weekly nab-paclitaxel led to a higher pCR rate than a schedule of days 1 and 8 every 3 weeks (44.9% versus 39.0%; P = .06, significance level of alpha = .10). Among subgroups, there was only a difference in pCR rates among those with TNBC (60.4% versus 50.0%; P = .06). Grade 3-4 toxic effects were similar regardless of denosumab exposure, but nonhematologic grade 3-4 toxicity was higher with weekly nab-paclitaxel (33.7% versus 24.1%; P = .004).
“The overall pCR difference (with nab-paclitaxel as neoadjuvant chemotherapy) seems small, but looking at the data from the GeparSEPTO study we would expect a transformation into a better invasive disease-free survival. Looking specifically into patients with TNBC, there is clear pCR increase by using the weekly regimen. With just over 60%, this is the highest pCR rate reported so far for a chemotherapy-only regimen. I would prefer to use nab-paclitaxel also in early breast cancer and would use the weekly regimen for women with TNBC who are at high risk,” Dr. Loibl said.
The study was limited by an imbalance of tumor subtypes between the treatment groups. The authors wrote that the results should guide further research, but nab-paclitaxel should not currently be viewed as a standard neoadjuvant treatment of breast cancer.
The study was funded by the German Breast Group, Bristol Myers Squibb, and Amgen. Dr. Loibl has served on the advisory boards or given lectures for AbbVie, Amgen, Bayer, Celgene, EirGenix, GSK, Lilly, Merck, Puma, Seagen, AstraZeneca, Daiichi Sankyo, Novartis, Pierre Fabre, Prime/Medscape, Bristol Myers Squibb, Chugai, Ipsen, Roche, and Samsung. She also holds a related patent.
REPORTING FROM JAMA ONCOLOGY
Coffee drinkers – even those with a sweet tooth – live longer
Among more than 170,000 people in the United Kingdom, those who drank about two to four cups of coffee a day, with or without sugar, had a lower rate of death than those who didn’t drink coffee, reported lead author Dan Liu, MD, of the department of epidemiology at Southern Medical University, Guangzhou, China.
“Previous observational studies have suggested an association between coffee intake and reduced risk for death, but they did not distinguish between coffee consumed with sugar or artificial sweeteners and coffee consumed without,” Dr. Liu, who is also of the department of public health and preventive medicine, Jinan University, Guangzhou, China, and colleagues wrote in Annals of Internal Medicine.
To learn more, the investigators turned to the UK Biobank, which recruited approximately half a million participants in the United Kingdom between 2006 and 2010 to undergo a variety of questionnaires, interviews, physical measurements, and medical tests. Out of this group, 171,616 participants completed at least one dietary questionnaire and met the criteria for the present study, including lack of cancer or cardiovascular disease upon enrollment.
Results from these questionnaires showed that 55.4% of participants drank coffee without any sweetener, 14.3% drank coffee with sugar, 6.1% drank coffee with artificial sweetener, and 24.2% did not drink coffee at all. Coffee drinkers were further sorted into groups based on how many cups of coffee they drank per day.
Coffee drinkers were significantly less likely to die from any cause
Over the course of about 7 years, 3,177 of the participants died, including 1,725 who died from cancer and 628 who died from cardiovascular disease.
After accounting for other factors that might impact risk of death, like lifestyle choices, the investigators found that coffee drinkers were significantly less likely to die from any cause, cardiovascular disease, or cancer, than those who didn’t drink coffee at all. This benefit was observed across types of coffee, including ground, instant, and decaffeinated varieties. The protective effects of coffee were most apparent in people who drank about two to four cups a day, among whom death was about 30% less likely, regardless of whether they added sugar to their coffee or not. Individuals who drank coffee with artificial sweetener did not live significantly longer than those who drank no coffee at all; however, the investigators suggested that this result may have been skewed by higher rates of negative health factors, such as obesity and hypertension, in the artificial sweetener group.
Dr. Liu and colleagues noted that their findings align with previous studies linking coffee consumption with survival. Like those other studies, the present data revealed a “U-shaped” benefit curve, in which moderate coffee consumption was associated with longer life, whereas low or no consumption and high consumption were not.
Experts caution against drinking sweetened beverages despite new findings
Although the present findings suggested that adding sugar did not eliminate the health benefits of coffee, Dr. Liu and colleagues still cautioned against sweetened beverages, citing widely known associations between sugar consumption and poor health.
In an accompanying editorial, Christina C. Wee, MD, MPH, deputy editor of Annals of Internal Medicine, pointed out a key detail from the data: the amount of sugar added to coffee in the U.K. study may be dwarfed by the amount consumed by some coffee drinkers across the pond.
“The average dose of added sugar per cup of sweetened coffee [in the study] was only a little over a teaspoon, or about 4 grams,” Dr. Wee wrote. “This is a far cry from the 15 grams of sugar in an 8-ounce cup of caramel macchiato at a popular U.S. coffee chain.”
Still, Dr. Wee, an associate professor of medicine at Harvard Medical School, Boston, and director of the obesity research program in the division of general medicine at Beth Israel Deaconess Medical Center, Boston, suggested that your typical coffee drinker can feel safe in their daily habit.
“The evidence does not suggest a need for most coffee drinkers – particularly those who drink it with no or modest amounts of sugar – to eliminate coffee,” she wrote. “So drink up – but it would be prudent to avoid too many caramel macchiatos while more evidence brews.”
Estefanía Toledo, MD, MPH, PhD, of the department of preventive medicine and public health at the University of Navarra, Pamplona, Spain, offered a similar takeaway.
“For those who enjoy drinking coffee, are not pregnant or lactating, and do not have special health conditions, coffee consumption could be considered part of a healthy lifestyle,” Dr. Toledo said in a written comment. “I would recommend adding as little sugar as possible to coffee until more evidence has been accrued.”
Dr. Toledo, who previously published a study showing a link between coffee and extended survival, noted that moderate coffee consumption has “repeatedly” been associated with lower rates of “several chronic diseases” and death, but there still isn’t enough evidence to recommend coffee for those who don’t already drink it.
More long-term research is needed, Dr. Toledo said, ideally with studies comparing changes in coffee consumption and health outcomes over time. These may not be forthcoming, however, as such trials are “not easy and feasible to conduct.”
David Kao, MD, assistant professor of medicine-cardiology and medical director of the school of medicine at the University of Colorado at Denver, Aurora, said that the study conducted by Dr. Liu and colleagues is a “very well-executed analysis” that strengthens our confidence in the safety of long-term coffee consumption, even for patients with heart disease.
Dr. Kao, who recently published an analysis showing that higher coffee intake is associated with a lower risk of heart failure, refrained from advising anyone to up their coffee quota.
“I remain cautious about stating too strongly that people should increase coffee intake purely to improve survival,” Dr. Kao said in a written comment. “That said, it does not appear harmful to increase it some, until you drink consistently more than six to seven cups per day.”
The study was supported by the National Natural Science Foundation of China, the Young Elite Scientist Sponsorship Program by CAST, the Guangdong Basic and Applied Basic Research Foundation, and others. Dr. Toledo and Dr. Kao disclosed no relevant conflicts of interest.
Among more than 170,000 people in the United Kingdom, those who drank about two to four cups of coffee a day, with or without sugar, had a lower rate of death than those who didn’t drink coffee, reported lead author Dan Liu, MD, of the department of epidemiology at Southern Medical University, Guangzhou, China.
“Previous observational studies have suggested an association between coffee intake and reduced risk for death, but they did not distinguish between coffee consumed with sugar or artificial sweeteners and coffee consumed without,” Dr. Liu, who is also of the department of public health and preventive medicine, Jinan University, Guangzhou, China, and colleagues wrote in Annals of Internal Medicine.
To learn more, the investigators turned to the UK Biobank, which recruited approximately half a million participants in the United Kingdom between 2006 and 2010 to undergo a variety of questionnaires, interviews, physical measurements, and medical tests. Out of this group, 171,616 participants completed at least one dietary questionnaire and met the criteria for the present study, including lack of cancer or cardiovascular disease upon enrollment.
Results from these questionnaires showed that 55.4% of participants drank coffee without any sweetener, 14.3% drank coffee with sugar, 6.1% drank coffee with artificial sweetener, and 24.2% did not drink coffee at all. Coffee drinkers were further sorted into groups based on how many cups of coffee they drank per day.
Coffee drinkers were significantly less likely to die from any cause
Over the course of about 7 years, 3,177 of the participants died, including 1,725 who died from cancer and 628 who died from cardiovascular disease.
After accounting for other factors that might impact risk of death, like lifestyle choices, the investigators found that coffee drinkers were significantly less likely to die from any cause, cardiovascular disease, or cancer, than those who didn’t drink coffee at all. This benefit was observed across types of coffee, including ground, instant, and decaffeinated varieties. The protective effects of coffee were most apparent in people who drank about two to four cups a day, among whom death was about 30% less likely, regardless of whether they added sugar to their coffee or not. Individuals who drank coffee with artificial sweetener did not live significantly longer than those who drank no coffee at all; however, the investigators suggested that this result may have been skewed by higher rates of negative health factors, such as obesity and hypertension, in the artificial sweetener group.
Dr. Liu and colleagues noted that their findings align with previous studies linking coffee consumption with survival. Like those other studies, the present data revealed a “U-shaped” benefit curve, in which moderate coffee consumption was associated with longer life, whereas low or no consumption and high consumption were not.
Experts caution against drinking sweetened beverages despite new findings
Although the present findings suggested that adding sugar did not eliminate the health benefits of coffee, Dr. Liu and colleagues still cautioned against sweetened beverages, citing widely known associations between sugar consumption and poor health.
In an accompanying editorial, Christina C. Wee, MD, MPH, deputy editor of Annals of Internal Medicine, pointed out a key detail from the data: the amount of sugar added to coffee in the U.K. study may be dwarfed by the amount consumed by some coffee drinkers across the pond.
“The average dose of added sugar per cup of sweetened coffee [in the study] was only a little over a teaspoon, or about 4 grams,” Dr. Wee wrote. “This is a far cry from the 15 grams of sugar in an 8-ounce cup of caramel macchiato at a popular U.S. coffee chain.”
Still, Dr. Wee, an associate professor of medicine at Harvard Medical School, Boston, and director of the obesity research program in the division of general medicine at Beth Israel Deaconess Medical Center, Boston, suggested that your typical coffee drinker can feel safe in their daily habit.
“The evidence does not suggest a need for most coffee drinkers – particularly those who drink it with no or modest amounts of sugar – to eliminate coffee,” she wrote. “So drink up – but it would be prudent to avoid too many caramel macchiatos while more evidence brews.”
Estefanía Toledo, MD, MPH, PhD, of the department of preventive medicine and public health at the University of Navarra, Pamplona, Spain, offered a similar takeaway.
“For those who enjoy drinking coffee, are not pregnant or lactating, and do not have special health conditions, coffee consumption could be considered part of a healthy lifestyle,” Dr. Toledo said in a written comment. “I would recommend adding as little sugar as possible to coffee until more evidence has been accrued.”
Dr. Toledo, who previously published a study showing a link between coffee and extended survival, noted that moderate coffee consumption has “repeatedly” been associated with lower rates of “several chronic diseases” and death, but there still isn’t enough evidence to recommend coffee for those who don’t already drink it.
More long-term research is needed, Dr. Toledo said, ideally with studies comparing changes in coffee consumption and health outcomes over time. These may not be forthcoming, however, as such trials are “not easy and feasible to conduct.”
David Kao, MD, assistant professor of medicine-cardiology and medical director of the school of medicine at the University of Colorado at Denver, Aurora, said that the study conducted by Dr. Liu and colleagues is a “very well-executed analysis” that strengthens our confidence in the safety of long-term coffee consumption, even for patients with heart disease.
Dr. Kao, who recently published an analysis showing that higher coffee intake is associated with a lower risk of heart failure, refrained from advising anyone to up their coffee quota.
“I remain cautious about stating too strongly that people should increase coffee intake purely to improve survival,” Dr. Kao said in a written comment. “That said, it does not appear harmful to increase it some, until you drink consistently more than six to seven cups per day.”
The study was supported by the National Natural Science Foundation of China, the Young Elite Scientist Sponsorship Program by CAST, the Guangdong Basic and Applied Basic Research Foundation, and others. Dr. Toledo and Dr. Kao disclosed no relevant conflicts of interest.
Among more than 170,000 people in the United Kingdom, those who drank about two to four cups of coffee a day, with or without sugar, had a lower rate of death than those who didn’t drink coffee, reported lead author Dan Liu, MD, of the department of epidemiology at Southern Medical University, Guangzhou, China.
“Previous observational studies have suggested an association between coffee intake and reduced risk for death, but they did not distinguish between coffee consumed with sugar or artificial sweeteners and coffee consumed without,” Dr. Liu, who is also of the department of public health and preventive medicine, Jinan University, Guangzhou, China, and colleagues wrote in Annals of Internal Medicine.
To learn more, the investigators turned to the UK Biobank, which recruited approximately half a million participants in the United Kingdom between 2006 and 2010 to undergo a variety of questionnaires, interviews, physical measurements, and medical tests. Out of this group, 171,616 participants completed at least one dietary questionnaire and met the criteria for the present study, including lack of cancer or cardiovascular disease upon enrollment.
Results from these questionnaires showed that 55.4% of participants drank coffee without any sweetener, 14.3% drank coffee with sugar, 6.1% drank coffee with artificial sweetener, and 24.2% did not drink coffee at all. Coffee drinkers were further sorted into groups based on how many cups of coffee they drank per day.
Coffee drinkers were significantly less likely to die from any cause
Over the course of about 7 years, 3,177 of the participants died, including 1,725 who died from cancer and 628 who died from cardiovascular disease.
After accounting for other factors that might impact risk of death, like lifestyle choices, the investigators found that coffee drinkers were significantly less likely to die from any cause, cardiovascular disease, or cancer, than those who didn’t drink coffee at all. This benefit was observed across types of coffee, including ground, instant, and decaffeinated varieties. The protective effects of coffee were most apparent in people who drank about two to four cups a day, among whom death was about 30% less likely, regardless of whether they added sugar to their coffee or not. Individuals who drank coffee with artificial sweetener did not live significantly longer than those who drank no coffee at all; however, the investigators suggested that this result may have been skewed by higher rates of negative health factors, such as obesity and hypertension, in the artificial sweetener group.
Dr. Liu and colleagues noted that their findings align with previous studies linking coffee consumption with survival. Like those other studies, the present data revealed a “U-shaped” benefit curve, in which moderate coffee consumption was associated with longer life, whereas low or no consumption and high consumption were not.
Experts caution against drinking sweetened beverages despite new findings
Although the present findings suggested that adding sugar did not eliminate the health benefits of coffee, Dr. Liu and colleagues still cautioned against sweetened beverages, citing widely known associations between sugar consumption and poor health.
In an accompanying editorial, Christina C. Wee, MD, MPH, deputy editor of Annals of Internal Medicine, pointed out a key detail from the data: the amount of sugar added to coffee in the U.K. study may be dwarfed by the amount consumed by some coffee drinkers across the pond.
“The average dose of added sugar per cup of sweetened coffee [in the study] was only a little over a teaspoon, or about 4 grams,” Dr. Wee wrote. “This is a far cry from the 15 grams of sugar in an 8-ounce cup of caramel macchiato at a popular U.S. coffee chain.”
Still, Dr. Wee, an associate professor of medicine at Harvard Medical School, Boston, and director of the obesity research program in the division of general medicine at Beth Israel Deaconess Medical Center, Boston, suggested that your typical coffee drinker can feel safe in their daily habit.
“The evidence does not suggest a need for most coffee drinkers – particularly those who drink it with no or modest amounts of sugar – to eliminate coffee,” she wrote. “So drink up – but it would be prudent to avoid too many caramel macchiatos while more evidence brews.”
Estefanía Toledo, MD, MPH, PhD, of the department of preventive medicine and public health at the University of Navarra, Pamplona, Spain, offered a similar takeaway.
“For those who enjoy drinking coffee, are not pregnant or lactating, and do not have special health conditions, coffee consumption could be considered part of a healthy lifestyle,” Dr. Toledo said in a written comment. “I would recommend adding as little sugar as possible to coffee until more evidence has been accrued.”
Dr. Toledo, who previously published a study showing a link between coffee and extended survival, noted that moderate coffee consumption has “repeatedly” been associated with lower rates of “several chronic diseases” and death, but there still isn’t enough evidence to recommend coffee for those who don’t already drink it.
More long-term research is needed, Dr. Toledo said, ideally with studies comparing changes in coffee consumption and health outcomes over time. These may not be forthcoming, however, as such trials are “not easy and feasible to conduct.”
David Kao, MD, assistant professor of medicine-cardiology and medical director of the school of medicine at the University of Colorado at Denver, Aurora, said that the study conducted by Dr. Liu and colleagues is a “very well-executed analysis” that strengthens our confidence in the safety of long-term coffee consumption, even for patients with heart disease.
Dr. Kao, who recently published an analysis showing that higher coffee intake is associated with a lower risk of heart failure, refrained from advising anyone to up their coffee quota.
“I remain cautious about stating too strongly that people should increase coffee intake purely to improve survival,” Dr. Kao said in a written comment. “That said, it does not appear harmful to increase it some, until you drink consistently more than six to seven cups per day.”
The study was supported by the National Natural Science Foundation of China, the Young Elite Scientist Sponsorship Program by CAST, the Guangdong Basic and Applied Basic Research Foundation, and others. Dr. Toledo and Dr. Kao disclosed no relevant conflicts of interest.
FROM ANNALS OF INTERNAL MEDICINE
Case study: Managing venous thromboembolism in the cancer patient
He is admitted and started on enoxaparin 1 mg/kg subcutaneously every 12 hours.
By the next morning, he is feeling better and wants to discuss discharge to home and follow-up plans.
Two months ago he presented with abdominal pain and evaluation revealed he had a pancreatic head mass with liver metastases. A liver biopsy was positive for adenocarcinoma consistent with pancreas primary. CA 19-9 level was 1,200 U/mL and he was started on FOLFIRINOX chemotherapy – which he has tolerated well thus far. CA 19-9 and follow-up CT scan show early response to chemotherapy.
Of course, this case raises many questions. Given how successful some directed biomarker-positive therapies are now, you would want to know his microsatellite instability (MSI)/progressive death–ligand 1 (PD-L1) and BRCA mutation status. A high PD-L1 positivity or MSI deficiency would suggest immunoantibody therapy and a BRCA mutation might suggest a poly (ADP-ribose) polymerase inhibitor could play a role.
However, let’s use this case to discuss his venous thromboembolism (VTE) .
Studies show that metastatic cancer patients on chemotherapy might experience a VTE episode of deep vein thrombosis (DVT) or pulmonary embolism (PE) or both as high as 20% of the time during their cancer course and therapy. This patient would be among those who experience the highest incidence of VTE because of the liver metastasis from the pancreatic adenocarcinoma.
So, what to do? Standard treatment of his pulmonary emboli would include either enoxaparin therapeutic dosing 1 mg/kg subcutaneously q12H or 1.5 mg/kg q24H for 3 months. At 3 months, repeat a CT chest scan to show resolution of pulmonary emboli and/or DVT or both, and repeat D-dimer, which should now be well under 1.
But then, there is a second decision to make: Can you stop anticoagulation if his clots have resolved? The answer is yes. If the clots were provoked and the provoking feature is gone you can stop anticoagulation. Patients with pregnancy, on a birth control pill, or on a long trip where immobilization occurred for a extended time (such as driving or flying) can have anticoagulation stopped because the provoking feature is gone, but this is not true in this case. This patient’s pancreas cancer and chemotherapy are ongoing and he will be at increased risk to clot once again if anticoagulation is stopped.
Should this patient have a hypercoagulable workup which might include protein C, protein S, and antithrombin levels? Remember this is quite rare and patients with these deficiencies usually present in their teens or 20s with increased clotting issues. The more common hypercoagulable workup would include checking for factor V Leiden and prothrombin G20210A mutations, as well as acquired antiphospholipid antibodies such as beta2 glycoprotein I, anticardiolipin, and the lupus inhibitor. However, in this 75-year-old cancer patient, these are not necessary or even relevant since his VTE was clearly provoked by metastatic cancer on chemotherapy.
Unfortunately, with metastatic active cancer, anticoagulation would need to be continued at full or possibly half therapeutic dose. Of course, enoxaparin injections can get tiresome for the patient and data suggest the same result can be achieved either with initial management or by continuing anticoagulation management using either rivaroxaban or apixaban.
Wouldn’t it have been better if this patient had never experienced VTE in the first place? Is that possible?
Yes, data suggest that it is. Higher-risk patients like this one could benefit from prophylactic anticoagulation. The Khorana predictive model gives us a simple clinical means to evaluate this and decide who might be at highest VTE risk and who could benefit from low-dose preventive anticoagulation.
In summary, cancer patients undergoing treatment for metastatic disease are at increased risk for symptomatic VTE. Once diagnosed, therapy is usually very effective, but may need to be prolonged as long as the cancer is still active or else, the VTE could recur. Preventive therapy for high-risk patients would be reasonable.
Dr. Henry is a medical oncologist with the Abramson Cancer Center at the University of Pennsylvania, Philadelphia.
He is admitted and started on enoxaparin 1 mg/kg subcutaneously every 12 hours.
By the next morning, he is feeling better and wants to discuss discharge to home and follow-up plans.
Two months ago he presented with abdominal pain and evaluation revealed he had a pancreatic head mass with liver metastases. A liver biopsy was positive for adenocarcinoma consistent with pancreas primary. CA 19-9 level was 1,200 U/mL and he was started on FOLFIRINOX chemotherapy – which he has tolerated well thus far. CA 19-9 and follow-up CT scan show early response to chemotherapy.
Of course, this case raises many questions. Given how successful some directed biomarker-positive therapies are now, you would want to know his microsatellite instability (MSI)/progressive death–ligand 1 (PD-L1) and BRCA mutation status. A high PD-L1 positivity or MSI deficiency would suggest immunoantibody therapy and a BRCA mutation might suggest a poly (ADP-ribose) polymerase inhibitor could play a role.
However, let’s use this case to discuss his venous thromboembolism (VTE) .
Studies show that metastatic cancer patients on chemotherapy might experience a VTE episode of deep vein thrombosis (DVT) or pulmonary embolism (PE) or both as high as 20% of the time during their cancer course and therapy. This patient would be among those who experience the highest incidence of VTE because of the liver metastasis from the pancreatic adenocarcinoma.
So, what to do? Standard treatment of his pulmonary emboli would include either enoxaparin therapeutic dosing 1 mg/kg subcutaneously q12H or 1.5 mg/kg q24H for 3 months. At 3 months, repeat a CT chest scan to show resolution of pulmonary emboli and/or DVT or both, and repeat D-dimer, which should now be well under 1.
But then, there is a second decision to make: Can you stop anticoagulation if his clots have resolved? The answer is yes. If the clots were provoked and the provoking feature is gone you can stop anticoagulation. Patients with pregnancy, on a birth control pill, or on a long trip where immobilization occurred for a extended time (such as driving or flying) can have anticoagulation stopped because the provoking feature is gone, but this is not true in this case. This patient’s pancreas cancer and chemotherapy are ongoing and he will be at increased risk to clot once again if anticoagulation is stopped.
Should this patient have a hypercoagulable workup which might include protein C, protein S, and antithrombin levels? Remember this is quite rare and patients with these deficiencies usually present in their teens or 20s with increased clotting issues. The more common hypercoagulable workup would include checking for factor V Leiden and prothrombin G20210A mutations, as well as acquired antiphospholipid antibodies such as beta2 glycoprotein I, anticardiolipin, and the lupus inhibitor. However, in this 75-year-old cancer patient, these are not necessary or even relevant since his VTE was clearly provoked by metastatic cancer on chemotherapy.
Unfortunately, with metastatic active cancer, anticoagulation would need to be continued at full or possibly half therapeutic dose. Of course, enoxaparin injections can get tiresome for the patient and data suggest the same result can be achieved either with initial management or by continuing anticoagulation management using either rivaroxaban or apixaban.
Wouldn’t it have been better if this patient had never experienced VTE in the first place? Is that possible?
Yes, data suggest that it is. Higher-risk patients like this one could benefit from prophylactic anticoagulation. The Khorana predictive model gives us a simple clinical means to evaluate this and decide who might be at highest VTE risk and who could benefit from low-dose preventive anticoagulation.
In summary, cancer patients undergoing treatment for metastatic disease are at increased risk for symptomatic VTE. Once diagnosed, therapy is usually very effective, but may need to be prolonged as long as the cancer is still active or else, the VTE could recur. Preventive therapy for high-risk patients would be reasonable.
Dr. Henry is a medical oncologist with the Abramson Cancer Center at the University of Pennsylvania, Philadelphia.
He is admitted and started on enoxaparin 1 mg/kg subcutaneously every 12 hours.
By the next morning, he is feeling better and wants to discuss discharge to home and follow-up plans.
Two months ago he presented with abdominal pain and evaluation revealed he had a pancreatic head mass with liver metastases. A liver biopsy was positive for adenocarcinoma consistent with pancreas primary. CA 19-9 level was 1,200 U/mL and he was started on FOLFIRINOX chemotherapy – which he has tolerated well thus far. CA 19-9 and follow-up CT scan show early response to chemotherapy.
Of course, this case raises many questions. Given how successful some directed biomarker-positive therapies are now, you would want to know his microsatellite instability (MSI)/progressive death–ligand 1 (PD-L1) and BRCA mutation status. A high PD-L1 positivity or MSI deficiency would suggest immunoantibody therapy and a BRCA mutation might suggest a poly (ADP-ribose) polymerase inhibitor could play a role.
However, let’s use this case to discuss his venous thromboembolism (VTE) .
Studies show that metastatic cancer patients on chemotherapy might experience a VTE episode of deep vein thrombosis (DVT) or pulmonary embolism (PE) or both as high as 20% of the time during their cancer course and therapy. This patient would be among those who experience the highest incidence of VTE because of the liver metastasis from the pancreatic adenocarcinoma.
So, what to do? Standard treatment of his pulmonary emboli would include either enoxaparin therapeutic dosing 1 mg/kg subcutaneously q12H or 1.5 mg/kg q24H for 3 months. At 3 months, repeat a CT chest scan to show resolution of pulmonary emboli and/or DVT or both, and repeat D-dimer, which should now be well under 1.
But then, there is a second decision to make: Can you stop anticoagulation if his clots have resolved? The answer is yes. If the clots were provoked and the provoking feature is gone you can stop anticoagulation. Patients with pregnancy, on a birth control pill, or on a long trip where immobilization occurred for a extended time (such as driving or flying) can have anticoagulation stopped because the provoking feature is gone, but this is not true in this case. This patient’s pancreas cancer and chemotherapy are ongoing and he will be at increased risk to clot once again if anticoagulation is stopped.
Should this patient have a hypercoagulable workup which might include protein C, protein S, and antithrombin levels? Remember this is quite rare and patients with these deficiencies usually present in their teens or 20s with increased clotting issues. The more common hypercoagulable workup would include checking for factor V Leiden and prothrombin G20210A mutations, as well as acquired antiphospholipid antibodies such as beta2 glycoprotein I, anticardiolipin, and the lupus inhibitor. However, in this 75-year-old cancer patient, these are not necessary or even relevant since his VTE was clearly provoked by metastatic cancer on chemotherapy.
Unfortunately, with metastatic active cancer, anticoagulation would need to be continued at full or possibly half therapeutic dose. Of course, enoxaparin injections can get tiresome for the patient and data suggest the same result can be achieved either with initial management or by continuing anticoagulation management using either rivaroxaban or apixaban.
Wouldn’t it have been better if this patient had never experienced VTE in the first place? Is that possible?
Yes, data suggest that it is. Higher-risk patients like this one could benefit from prophylactic anticoagulation. The Khorana predictive model gives us a simple clinical means to evaluate this and decide who might be at highest VTE risk and who could benefit from low-dose preventive anticoagulation.
In summary, cancer patients undergoing treatment for metastatic disease are at increased risk for symptomatic VTE. Once diagnosed, therapy is usually very effective, but may need to be prolonged as long as the cancer is still active or else, the VTE could recur. Preventive therapy for high-risk patients would be reasonable.
Dr. Henry is a medical oncologist with the Abramson Cancer Center at the University of Pennsylvania, Philadelphia.
ESG’s cardiometabolic benefits last 5 years
SAN DIEGO – Endoscopic sleeve gastroplasty (ESG) led to sustained weight loss and a reduction of cardiometabolic syndrome comorbidities at 5 years, according to a new retrospective analysis of prospectively collected data.
Improved cardiometabolic outcomes following bariatric surgery have been well documented, but ESG is relatively new, so its outcomes haven’t been as well described. The outcomes are encouraging, though not as good as those of bariatric surgery. “It’s still better, but only one percent of the patients undergo the surgery, even though they’re candidates,” said Donevan Westerveld, MD, who presented the study at the annual Digestive Disease Week® (DDW).
Improvements included weight, HbA1c percentage, hypertension, and low-density lipoprotein. “I was surprised that the LDL decreased numerically, not so much HbA1c and hypertension. I knew [those] would come down with weight loss,” said Dr. Westerveld, a second-year fellow at Weill Cornell Medicine, New York.
He also called for guidelines for ESG. “Given the fact there’s an improvement of comorbid conditions, it’s something we should look at,” said Dr. Westerveld.
“It’s fascinating because it tells us two important things about endoscopic sleeve gastroplasty. One, [the benefit] in the majority of cases lasts at least 5 years. The weight loss is durable. And then it tells us that there’s improvement in all the cardiometabolic factors that matter, and those effects are seen all the way up to 5 years. So very important findings that support the benefits of the endoscopic gastroplasty in obesity and cardiometabolic risks and metabolic syndrome,” said Andres Acosta, MD, PhD, a comoderator of the session where the study was presented. He is assistant professor of medicine and a consultant in gastroenterology and hepatology at Mayo Clinic in Rochester, Minn.
The findings should also encourage more innovation. “Doing these endoscopic procedures, having successful results that hold for 5 years, opens the path for new and better procedures, so we have better weight loss,” said Dr. Acosta.
Previous work by Dr. Westerveld’s group found benefits of ESG at 12 months, including improvements in mean HbA1c levels in all patients (6.1%-5.5%; P = .05) and those with diabetes or prediabetes (6.6%-5.6%; P = .02), reduction in mean waist circumference (119.66-92.75 cm; P < .001), reduction in systolic blood pressure (129.02-122.23 mg/dL; P = .023), triglycerides (131.84-92.36 mg/dL; P = .017), and alanine aminotransferase (ALT, 32.26-20.68 mg/dL; P < .001).
In the new study, the group followed 255 patients at 1, 3, and 5 years post procedure who were treated consecutively at Weill Cornell Medicine from 2013 to 2021. Among the patients were those who had failed weight loss measures and were either not candidates for surgery or had refused surgery.
The mean age was 45.5 years, 69% were female, and the mean body mass index was 38.6. Overall, 40.3% had prediabetes or diabetes, 26.7% had hypertension, 60.8% had low-density lipoprotein (LDL) above 100 mg/dL, and 29.3% had elevated ALT. Sixty-six percent had been followed up at 1 year, 78% at 3 years, and 87% at 5 years.
Weight loss averaged 15.7% at 1 year and 15.3% at year 5, and the values were statistically significant. Among patients with diabetes and prediabetes, HbA1c percentage dropped from a baseline value of 6.4% to 5.7% at year 1, 6.1% at year 3, and 5.8% at year 5 (P < .05 for all). For all patients, the value dropped from 5.8% at baseline to 5.6% at year 1, 5.7% at year 3, and 5.4% at year 5. These changes were not statistically significant.
Systolic blood pressure went down among patients with stage 1 hypertension, from 135 mm Hg at baseline to 122 at year 1 and 121 at year 3 (P < .05 or both), but the mean value increased to 129 at year 5 and was not statistically significant. LDL among all patients declined from 136 mg/dL at baseline to 125 at year 1 (nonsignificant), 115 at year 3 (P < .05), and 109 at year 5 (P < .05). Alanine transaminase values declined from about 29 at baseline to 25 at year 1, 26 at year 3, and 24 at year 5 (P < .05 for all).
Serious adverse events were rare, occurring in just two cases (< 1%).
The study was limited by lack of a sham control, and its retrospective data may have included bias because many of the procedures were not paid for by insurance, leading to high rates of self-pay.
Dr. Westerveld has no relevant financial disclosures. Dr. Acosta is a founder of Gila Therapeutics and Phenomix Sciences. Dr. Acosta consults for Amgen, Gila Therapeutics, Rhythm Pharmaceuticals, and General Mills. He has received funding from Rhythm, Novo Nordisk, Apollo Endosurgery, and USGI Medical.
SAN DIEGO – Endoscopic sleeve gastroplasty (ESG) led to sustained weight loss and a reduction of cardiometabolic syndrome comorbidities at 5 years, according to a new retrospective analysis of prospectively collected data.
Improved cardiometabolic outcomes following bariatric surgery have been well documented, but ESG is relatively new, so its outcomes haven’t been as well described. The outcomes are encouraging, though not as good as those of bariatric surgery. “It’s still better, but only one percent of the patients undergo the surgery, even though they’re candidates,” said Donevan Westerveld, MD, who presented the study at the annual Digestive Disease Week® (DDW).
Improvements included weight, HbA1c percentage, hypertension, and low-density lipoprotein. “I was surprised that the LDL decreased numerically, not so much HbA1c and hypertension. I knew [those] would come down with weight loss,” said Dr. Westerveld, a second-year fellow at Weill Cornell Medicine, New York.
He also called for guidelines for ESG. “Given the fact there’s an improvement of comorbid conditions, it’s something we should look at,” said Dr. Westerveld.
“It’s fascinating because it tells us two important things about endoscopic sleeve gastroplasty. One, [the benefit] in the majority of cases lasts at least 5 years. The weight loss is durable. And then it tells us that there’s improvement in all the cardiometabolic factors that matter, and those effects are seen all the way up to 5 years. So very important findings that support the benefits of the endoscopic gastroplasty in obesity and cardiometabolic risks and metabolic syndrome,” said Andres Acosta, MD, PhD, a comoderator of the session where the study was presented. He is assistant professor of medicine and a consultant in gastroenterology and hepatology at Mayo Clinic in Rochester, Minn.
The findings should also encourage more innovation. “Doing these endoscopic procedures, having successful results that hold for 5 years, opens the path for new and better procedures, so we have better weight loss,” said Dr. Acosta.
Previous work by Dr. Westerveld’s group found benefits of ESG at 12 months, including improvements in mean HbA1c levels in all patients (6.1%-5.5%; P = .05) and those with diabetes or prediabetes (6.6%-5.6%; P = .02), reduction in mean waist circumference (119.66-92.75 cm; P < .001), reduction in systolic blood pressure (129.02-122.23 mg/dL; P = .023), triglycerides (131.84-92.36 mg/dL; P = .017), and alanine aminotransferase (ALT, 32.26-20.68 mg/dL; P < .001).
In the new study, the group followed 255 patients at 1, 3, and 5 years post procedure who were treated consecutively at Weill Cornell Medicine from 2013 to 2021. Among the patients were those who had failed weight loss measures and were either not candidates for surgery or had refused surgery.
The mean age was 45.5 years, 69% were female, and the mean body mass index was 38.6. Overall, 40.3% had prediabetes or diabetes, 26.7% had hypertension, 60.8% had low-density lipoprotein (LDL) above 100 mg/dL, and 29.3% had elevated ALT. Sixty-six percent had been followed up at 1 year, 78% at 3 years, and 87% at 5 years.
Weight loss averaged 15.7% at 1 year and 15.3% at year 5, and the values were statistically significant. Among patients with diabetes and prediabetes, HbA1c percentage dropped from a baseline value of 6.4% to 5.7% at year 1, 6.1% at year 3, and 5.8% at year 5 (P < .05 for all). For all patients, the value dropped from 5.8% at baseline to 5.6% at year 1, 5.7% at year 3, and 5.4% at year 5. These changes were not statistically significant.
Systolic blood pressure went down among patients with stage 1 hypertension, from 135 mm Hg at baseline to 122 at year 1 and 121 at year 3 (P < .05 or both), but the mean value increased to 129 at year 5 and was not statistically significant. LDL among all patients declined from 136 mg/dL at baseline to 125 at year 1 (nonsignificant), 115 at year 3 (P < .05), and 109 at year 5 (P < .05). Alanine transaminase values declined from about 29 at baseline to 25 at year 1, 26 at year 3, and 24 at year 5 (P < .05 for all).
Serious adverse events were rare, occurring in just two cases (< 1%).
The study was limited by lack of a sham control, and its retrospective data may have included bias because many of the procedures were not paid for by insurance, leading to high rates of self-pay.
Dr. Westerveld has no relevant financial disclosures. Dr. Acosta is a founder of Gila Therapeutics and Phenomix Sciences. Dr. Acosta consults for Amgen, Gila Therapeutics, Rhythm Pharmaceuticals, and General Mills. He has received funding from Rhythm, Novo Nordisk, Apollo Endosurgery, and USGI Medical.
SAN DIEGO – Endoscopic sleeve gastroplasty (ESG) led to sustained weight loss and a reduction of cardiometabolic syndrome comorbidities at 5 years, according to a new retrospective analysis of prospectively collected data.
Improved cardiometabolic outcomes following bariatric surgery have been well documented, but ESG is relatively new, so its outcomes haven’t been as well described. The outcomes are encouraging, though not as good as those of bariatric surgery. “It’s still better, but only one percent of the patients undergo the surgery, even though they’re candidates,” said Donevan Westerveld, MD, who presented the study at the annual Digestive Disease Week® (DDW).
Improvements included weight, HbA1c percentage, hypertension, and low-density lipoprotein. “I was surprised that the LDL decreased numerically, not so much HbA1c and hypertension. I knew [those] would come down with weight loss,” said Dr. Westerveld, a second-year fellow at Weill Cornell Medicine, New York.
He also called for guidelines for ESG. “Given the fact there’s an improvement of comorbid conditions, it’s something we should look at,” said Dr. Westerveld.
“It’s fascinating because it tells us two important things about endoscopic sleeve gastroplasty. One, [the benefit] in the majority of cases lasts at least 5 years. The weight loss is durable. And then it tells us that there’s improvement in all the cardiometabolic factors that matter, and those effects are seen all the way up to 5 years. So very important findings that support the benefits of the endoscopic gastroplasty in obesity and cardiometabolic risks and metabolic syndrome,” said Andres Acosta, MD, PhD, a comoderator of the session where the study was presented. He is assistant professor of medicine and a consultant in gastroenterology and hepatology at Mayo Clinic in Rochester, Minn.
The findings should also encourage more innovation. “Doing these endoscopic procedures, having successful results that hold for 5 years, opens the path for new and better procedures, so we have better weight loss,” said Dr. Acosta.
Previous work by Dr. Westerveld’s group found benefits of ESG at 12 months, including improvements in mean HbA1c levels in all patients (6.1%-5.5%; P = .05) and those with diabetes or prediabetes (6.6%-5.6%; P = .02), reduction in mean waist circumference (119.66-92.75 cm; P < .001), reduction in systolic blood pressure (129.02-122.23 mg/dL; P = .023), triglycerides (131.84-92.36 mg/dL; P = .017), and alanine aminotransferase (ALT, 32.26-20.68 mg/dL; P < .001).
In the new study, the group followed 255 patients at 1, 3, and 5 years post procedure who were treated consecutively at Weill Cornell Medicine from 2013 to 2021. Among the patients were those who had failed weight loss measures and were either not candidates for surgery or had refused surgery.
The mean age was 45.5 years, 69% were female, and the mean body mass index was 38.6. Overall, 40.3% had prediabetes or diabetes, 26.7% had hypertension, 60.8% had low-density lipoprotein (LDL) above 100 mg/dL, and 29.3% had elevated ALT. Sixty-six percent had been followed up at 1 year, 78% at 3 years, and 87% at 5 years.
Weight loss averaged 15.7% at 1 year and 15.3% at year 5, and the values were statistically significant. Among patients with diabetes and prediabetes, HbA1c percentage dropped from a baseline value of 6.4% to 5.7% at year 1, 6.1% at year 3, and 5.8% at year 5 (P < .05 for all). For all patients, the value dropped from 5.8% at baseline to 5.6% at year 1, 5.7% at year 3, and 5.4% at year 5. These changes were not statistically significant.
Systolic blood pressure went down among patients with stage 1 hypertension, from 135 mm Hg at baseline to 122 at year 1 and 121 at year 3 (P < .05 or both), but the mean value increased to 129 at year 5 and was not statistically significant. LDL among all patients declined from 136 mg/dL at baseline to 125 at year 1 (nonsignificant), 115 at year 3 (P < .05), and 109 at year 5 (P < .05). Alanine transaminase values declined from about 29 at baseline to 25 at year 1, 26 at year 3, and 24 at year 5 (P < .05 for all).
Serious adverse events were rare, occurring in just two cases (< 1%).
The study was limited by lack of a sham control, and its retrospective data may have included bias because many of the procedures were not paid for by insurance, leading to high rates of self-pay.
Dr. Westerveld has no relevant financial disclosures. Dr. Acosta is a founder of Gila Therapeutics and Phenomix Sciences. Dr. Acosta consults for Amgen, Gila Therapeutics, Rhythm Pharmaceuticals, and General Mills. He has received funding from Rhythm, Novo Nordisk, Apollo Endosurgery, and USGI Medical.
At DDW 2022













