User login
‘Obesity paradox’ in AFib challenged as mortality climbs with BMI
The relationship between body mass index (BMI) and all-cause mortality in patients with atrial fibrillation (AFib) is U-shaped, with the risk highest in those who are underweight or severely obese and lowest in patients defined simply as obese, a registry analysis suggests. It also showed a similar relationship between BMI and risk for new or worsening heart failure (HF).
Mortality bottomed out at a BMI of about 30-35 kg/m2, which suggests that mild obesity was protective, compared even with “normal-weight” or “overweight” BMI. Still, mortality went up sharply from there with rising BMI.
But higher BMI, a surrogate for obesity, apparently didn’t worsen outcomes by itself. The risk for death from any cause at higher obesity levels was found to depend a lot on related risk factors and comorbidities when the analysis controlled for conditions such as diabetes and hypertension.
The findings suggest an inverse relationship between BMI and all-cause mortality in AFib only for patients with BMI less than about 30. They therefore argue against any “obesity paradox” in AFib that posits consistently better survival with increasing levels of obesity, say researchers, based on their analysis of patients with new-onset AFib in the GARFIELD-AF registry.
“It’s common practice now for clinicians to discuss weight within a clinic setting when they’re talking to their AFib patients,” observed Christian Fielder Camm, BM, BCh, University of Oxford (England), and Royal Berkshire NHS Foundation Trust, Reading, England. So studies suggesting an inverse association between BMI and AFib-related risk can be a concern.
Such studies “seem to suggest that once you’ve got AFib, maintaining a high or very high BMI may in some way be protective – which is contrary to what would seem to make sense and certainly contrary to what our results have shown,” Dr. Camm told this news organization.
“I think that having further evidence now to suggest, actually, that greater BMI is associated with a greater risk of all-cause mortality and heart failure helps reframe that discussion at the physician-patient interaction level more clearly, and ensures that we’re able to talk to our patients appropriately about risks associated with BMI and atrial fibrillation,” said Dr. Camm, who is lead author on the analysis published in Open Heart.
“Obesity is a cause of most cardiovascular diseases, but [these] data would support that being overweight or having mild obesity does not increase the risk,” observed Carl J. Lavie, MD, of the John Ochsner Heart and Vascular Institute, New Orleans, La., and the Ochsner Clinical School at the University of Queensland, Brisbane, Australia.
“At a BMI of 40, it’s very important for them to lose weight for their long-term prognosis,” Dr. Lavie noted, but “at a BMI of 30, the important thing would be to prevent further weight gain. And if they could keep their BMI of 30, they should have a good prognosis. Their prognosis would be particularly good if they didn’t gain weight and put themselves in a more extreme obesity class that is associated with worse risk.”
The current analysis, Dr. Lavie said, “is way better than the AFFIRM study,” which yielded an obesity-paradox report on its patients with AFib about a dozen years ago. “It’s got more data, more numbers, more statistical power,” and breaks BMI into more categories.
That previous analysis based on the influential AFFIRM randomized trial separated its 4,060 patients with AFib into normal (BMI, 18.5-25), overweight (BMI, 25-30), and obese (BMI, > 30) categories, per the convention at the time. It concluded that “obese patients with atrial fibrillation appear to have better long-term outcomes than nonobese patients.”
Bleeding risk on oral anticoagulants
Also noteworthy in the current analysis, variation in BMI didn’t seem to affect mortality or risk for major bleeding or nonhemorrhagic stroke according to choice of oral anticoagulant – whether a new oral anticoagulant (NOAC) or a vitamin K antagonist (VKA).
“We saw that even in the obese and extremely obese group, all-cause mortality was lower in the group taking NOACs, compared with taking warfarin,” Dr. Camm observed, “which goes against the idea that we would need any kind of dose adjustments for increased BMI.”
Indeed, the report notes, use of NOACs, compared with VKA, was associated with a 23% drop in risk for death among patients who were either normal weight or overweight and also in those who were obese or extremely obese.
Those findings “are basically saying that the NOACs look better than warfarin regardless of weight,” agreed Dr. Lavie. “The problem is that the study is not very powered.”
Whereas the benefits of NOACs, compared to VKA, seem similar for patients with a BMI of 30 or 34, compared with a BMI of 23, for example, “none of the studies has many people with 50 BMI.” Many clinicians “feel uncomfortable giving the same dose of NOAC to somebody who has a 60 BMI,” he said. At least with warfarin, “you can check the INR [international normalized ratio].”
The current analysis included 40,482 patients with recently diagnosed AFib and at least one other stroke risk factor from among the registry’s more than 50,000 patients from 35 countries, enrolled from 2010 to 2016. They were followed for 2 years.
The 703 patients with BMI under 18.5 at AFib diagnosis were classified per World Health Organization definitions as underweight; the 13,095 with BMI 18.5-25 as normal weight; the 15,043 with BMI 25-30 as overweight; the 7,560 with BMI 30-35 as obese; and the 4,081 with BMI above 35 as extremely obese. Their ages averaged 71 years, and 55.6% were men.
BMI effects on different outcomes
Relationships between BMI and all-cause mortality and between BMI and new or worsening HF emerged as U-shaped, the risk climbing with both increasing and decreasing BMI. The nadir BMI for risk was about 30 in the case of mortality and about 25 for new or worsening HF.
The all-cause mortality risk rose by 32% for every 5 BMI points lower than a BMI of 30, and by 16% for every 5 BMI points higher than 30, in a partially adjusted analysis. The risk for new or worsening HF rose significantly with increasing but not decreasing BMI, and the reverse was observed for the endpoint of major bleeding.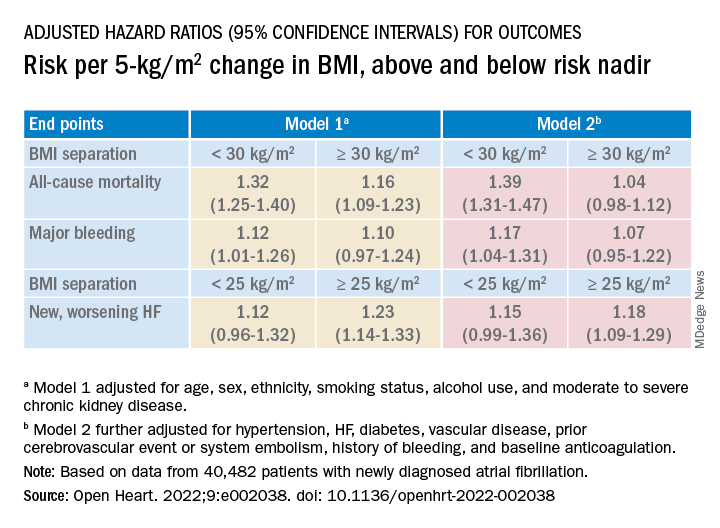
The effect of BMI on all-cause mortality was “substantially attenuated” when the analysis was further adjusted with “likely mediators of any association between BMI and outcomes,” including hypertension, diabetes, HF, cerebrovascular events, and history of bleeding, Dr. Camm said.
That blunted BMI-mortality relationship, he said, “suggests that a lot of the effect is mediated through relatively traditional risk factors like hypertension and diabetes.”
The 2010 AFFIRM analysis by BMI, Dr. Lavie noted, “didn’t even look at the underweight; they actually threw them out.” Yet, such patients with AFib, who tend to be extremely frail or have chronic diseases or conditions other than the arrhythmia, are common. A take-home of the current study is that “the underweight with atrial fibrillation have a really bad prognosis.”
That message isn’t heard as much, he observed, “but is as important as saying that BMI 30 has the best prognosis. The worst prognosis is with the underweight or the really extreme obese.”
Dr. Camm discloses research funding from the British Heart Foundation. Disclosures for the other authors are in the report. Dr. Lavie has previously disclosed serving as a speaker and consultant for PAI Health and DSM Nutritional Products and is the author of “The Obesity Paradox: When Thinner Means Sicker and Heavier Means Healthier” (Avery, 2014).
A version of this article first appeared on Medscape.com.
The relationship between body mass index (BMI) and all-cause mortality in patients with atrial fibrillation (AFib) is U-shaped, with the risk highest in those who are underweight or severely obese and lowest in patients defined simply as obese, a registry analysis suggests. It also showed a similar relationship between BMI and risk for new or worsening heart failure (HF).
Mortality bottomed out at a BMI of about 30-35 kg/m2, which suggests that mild obesity was protective, compared even with “normal-weight” or “overweight” BMI. Still, mortality went up sharply from there with rising BMI.
But higher BMI, a surrogate for obesity, apparently didn’t worsen outcomes by itself. The risk for death from any cause at higher obesity levels was found to depend a lot on related risk factors and comorbidities when the analysis controlled for conditions such as diabetes and hypertension.
The findings suggest an inverse relationship between BMI and all-cause mortality in AFib only for patients with BMI less than about 30. They therefore argue against any “obesity paradox” in AFib that posits consistently better survival with increasing levels of obesity, say researchers, based on their analysis of patients with new-onset AFib in the GARFIELD-AF registry.
“It’s common practice now for clinicians to discuss weight within a clinic setting when they’re talking to their AFib patients,” observed Christian Fielder Camm, BM, BCh, University of Oxford (England), and Royal Berkshire NHS Foundation Trust, Reading, England. So studies suggesting an inverse association between BMI and AFib-related risk can be a concern.
Such studies “seem to suggest that once you’ve got AFib, maintaining a high or very high BMI may in some way be protective – which is contrary to what would seem to make sense and certainly contrary to what our results have shown,” Dr. Camm told this news organization.
“I think that having further evidence now to suggest, actually, that greater BMI is associated with a greater risk of all-cause mortality and heart failure helps reframe that discussion at the physician-patient interaction level more clearly, and ensures that we’re able to talk to our patients appropriately about risks associated with BMI and atrial fibrillation,” said Dr. Camm, who is lead author on the analysis published in Open Heart.
“Obesity is a cause of most cardiovascular diseases, but [these] data would support that being overweight or having mild obesity does not increase the risk,” observed Carl J. Lavie, MD, of the John Ochsner Heart and Vascular Institute, New Orleans, La., and the Ochsner Clinical School at the University of Queensland, Brisbane, Australia.
“At a BMI of 40, it’s very important for them to lose weight for their long-term prognosis,” Dr. Lavie noted, but “at a BMI of 30, the important thing would be to prevent further weight gain. And if they could keep their BMI of 30, they should have a good prognosis. Their prognosis would be particularly good if they didn’t gain weight and put themselves in a more extreme obesity class that is associated with worse risk.”
The current analysis, Dr. Lavie said, “is way better than the AFFIRM study,” which yielded an obesity-paradox report on its patients with AFib about a dozen years ago. “It’s got more data, more numbers, more statistical power,” and breaks BMI into more categories.
That previous analysis based on the influential AFFIRM randomized trial separated its 4,060 patients with AFib into normal (BMI, 18.5-25), overweight (BMI, 25-30), and obese (BMI, > 30) categories, per the convention at the time. It concluded that “obese patients with atrial fibrillation appear to have better long-term outcomes than nonobese patients.”
Bleeding risk on oral anticoagulants
Also noteworthy in the current analysis, variation in BMI didn’t seem to affect mortality or risk for major bleeding or nonhemorrhagic stroke according to choice of oral anticoagulant – whether a new oral anticoagulant (NOAC) or a vitamin K antagonist (VKA).
“We saw that even in the obese and extremely obese group, all-cause mortality was lower in the group taking NOACs, compared with taking warfarin,” Dr. Camm observed, “which goes against the idea that we would need any kind of dose adjustments for increased BMI.”
Indeed, the report notes, use of NOACs, compared with VKA, was associated with a 23% drop in risk for death among patients who were either normal weight or overweight and also in those who were obese or extremely obese.
Those findings “are basically saying that the NOACs look better than warfarin regardless of weight,” agreed Dr. Lavie. “The problem is that the study is not very powered.”
Whereas the benefits of NOACs, compared to VKA, seem similar for patients with a BMI of 30 or 34, compared with a BMI of 23, for example, “none of the studies has many people with 50 BMI.” Many clinicians “feel uncomfortable giving the same dose of NOAC to somebody who has a 60 BMI,” he said. At least with warfarin, “you can check the INR [international normalized ratio].”
The current analysis included 40,482 patients with recently diagnosed AFib and at least one other stroke risk factor from among the registry’s more than 50,000 patients from 35 countries, enrolled from 2010 to 2016. They were followed for 2 years.
The 703 patients with BMI under 18.5 at AFib diagnosis were classified per World Health Organization definitions as underweight; the 13,095 with BMI 18.5-25 as normal weight; the 15,043 with BMI 25-30 as overweight; the 7,560 with BMI 30-35 as obese; and the 4,081 with BMI above 35 as extremely obese. Their ages averaged 71 years, and 55.6% were men.
BMI effects on different outcomes
Relationships between BMI and all-cause mortality and between BMI and new or worsening HF emerged as U-shaped, the risk climbing with both increasing and decreasing BMI. The nadir BMI for risk was about 30 in the case of mortality and about 25 for new or worsening HF.
The all-cause mortality risk rose by 32% for every 5 BMI points lower than a BMI of 30, and by 16% for every 5 BMI points higher than 30, in a partially adjusted analysis. The risk for new or worsening HF rose significantly with increasing but not decreasing BMI, and the reverse was observed for the endpoint of major bleeding.
The effect of BMI on all-cause mortality was “substantially attenuated” when the analysis was further adjusted with “likely mediators of any association between BMI and outcomes,” including hypertension, diabetes, HF, cerebrovascular events, and history of bleeding, Dr. Camm said.
That blunted BMI-mortality relationship, he said, “suggests that a lot of the effect is mediated through relatively traditional risk factors like hypertension and diabetes.”
The 2010 AFFIRM analysis by BMI, Dr. Lavie noted, “didn’t even look at the underweight; they actually threw them out.” Yet, such patients with AFib, who tend to be extremely frail or have chronic diseases or conditions other than the arrhythmia, are common. A take-home of the current study is that “the underweight with atrial fibrillation have a really bad prognosis.”
That message isn’t heard as much, he observed, “but is as important as saying that BMI 30 has the best prognosis. The worst prognosis is with the underweight or the really extreme obese.”
Dr. Camm discloses research funding from the British Heart Foundation. Disclosures for the other authors are in the report. Dr. Lavie has previously disclosed serving as a speaker and consultant for PAI Health and DSM Nutritional Products and is the author of “The Obesity Paradox: When Thinner Means Sicker and Heavier Means Healthier” (Avery, 2014).
A version of this article first appeared on Medscape.com.
The relationship between body mass index (BMI) and all-cause mortality in patients with atrial fibrillation (AFib) is U-shaped, with the risk highest in those who are underweight or severely obese and lowest in patients defined simply as obese, a registry analysis suggests. It also showed a similar relationship between BMI and risk for new or worsening heart failure (HF).
Mortality bottomed out at a BMI of about 30-35 kg/m2, which suggests that mild obesity was protective, compared even with “normal-weight” or “overweight” BMI. Still, mortality went up sharply from there with rising BMI.
But higher BMI, a surrogate for obesity, apparently didn’t worsen outcomes by itself. The risk for death from any cause at higher obesity levels was found to depend a lot on related risk factors and comorbidities when the analysis controlled for conditions such as diabetes and hypertension.
The findings suggest an inverse relationship between BMI and all-cause mortality in AFib only for patients with BMI less than about 30. They therefore argue against any “obesity paradox” in AFib that posits consistently better survival with increasing levels of obesity, say researchers, based on their analysis of patients with new-onset AFib in the GARFIELD-AF registry.
“It’s common practice now for clinicians to discuss weight within a clinic setting when they’re talking to their AFib patients,” observed Christian Fielder Camm, BM, BCh, University of Oxford (England), and Royal Berkshire NHS Foundation Trust, Reading, England. So studies suggesting an inverse association between BMI and AFib-related risk can be a concern.
Such studies “seem to suggest that once you’ve got AFib, maintaining a high or very high BMI may in some way be protective – which is contrary to what would seem to make sense and certainly contrary to what our results have shown,” Dr. Camm told this news organization.
“I think that having further evidence now to suggest, actually, that greater BMI is associated with a greater risk of all-cause mortality and heart failure helps reframe that discussion at the physician-patient interaction level more clearly, and ensures that we’re able to talk to our patients appropriately about risks associated with BMI and atrial fibrillation,” said Dr. Camm, who is lead author on the analysis published in Open Heart.
“Obesity is a cause of most cardiovascular diseases, but [these] data would support that being overweight or having mild obesity does not increase the risk,” observed Carl J. Lavie, MD, of the John Ochsner Heart and Vascular Institute, New Orleans, La., and the Ochsner Clinical School at the University of Queensland, Brisbane, Australia.
“At a BMI of 40, it’s very important for them to lose weight for their long-term prognosis,” Dr. Lavie noted, but “at a BMI of 30, the important thing would be to prevent further weight gain. And if they could keep their BMI of 30, they should have a good prognosis. Their prognosis would be particularly good if they didn’t gain weight and put themselves in a more extreme obesity class that is associated with worse risk.”
The current analysis, Dr. Lavie said, “is way better than the AFFIRM study,” which yielded an obesity-paradox report on its patients with AFib about a dozen years ago. “It’s got more data, more numbers, more statistical power,” and breaks BMI into more categories.
That previous analysis based on the influential AFFIRM randomized trial separated its 4,060 patients with AFib into normal (BMI, 18.5-25), overweight (BMI, 25-30), and obese (BMI, > 30) categories, per the convention at the time. It concluded that “obese patients with atrial fibrillation appear to have better long-term outcomes than nonobese patients.”
Bleeding risk on oral anticoagulants
Also noteworthy in the current analysis, variation in BMI didn’t seem to affect mortality or risk for major bleeding or nonhemorrhagic stroke according to choice of oral anticoagulant – whether a new oral anticoagulant (NOAC) or a vitamin K antagonist (VKA).
“We saw that even in the obese and extremely obese group, all-cause mortality was lower in the group taking NOACs, compared with taking warfarin,” Dr. Camm observed, “which goes against the idea that we would need any kind of dose adjustments for increased BMI.”
Indeed, the report notes, use of NOACs, compared with VKA, was associated with a 23% drop in risk for death among patients who were either normal weight or overweight and also in those who were obese or extremely obese.
Those findings “are basically saying that the NOACs look better than warfarin regardless of weight,” agreed Dr. Lavie. “The problem is that the study is not very powered.”
Whereas the benefits of NOACs, compared to VKA, seem similar for patients with a BMI of 30 or 34, compared with a BMI of 23, for example, “none of the studies has many people with 50 BMI.” Many clinicians “feel uncomfortable giving the same dose of NOAC to somebody who has a 60 BMI,” he said. At least with warfarin, “you can check the INR [international normalized ratio].”
The current analysis included 40,482 patients with recently diagnosed AFib and at least one other stroke risk factor from among the registry’s more than 50,000 patients from 35 countries, enrolled from 2010 to 2016. They were followed for 2 years.
The 703 patients with BMI under 18.5 at AFib diagnosis were classified per World Health Organization definitions as underweight; the 13,095 with BMI 18.5-25 as normal weight; the 15,043 with BMI 25-30 as overweight; the 7,560 with BMI 30-35 as obese; and the 4,081 with BMI above 35 as extremely obese. Their ages averaged 71 years, and 55.6% were men.
BMI effects on different outcomes
Relationships between BMI and all-cause mortality and between BMI and new or worsening HF emerged as U-shaped, the risk climbing with both increasing and decreasing BMI. The nadir BMI for risk was about 30 in the case of mortality and about 25 for new or worsening HF.
The all-cause mortality risk rose by 32% for every 5 BMI points lower than a BMI of 30, and by 16% for every 5 BMI points higher than 30, in a partially adjusted analysis. The risk for new or worsening HF rose significantly with increasing but not decreasing BMI, and the reverse was observed for the endpoint of major bleeding.
The effect of BMI on all-cause mortality was “substantially attenuated” when the analysis was further adjusted with “likely mediators of any association between BMI and outcomes,” including hypertension, diabetes, HF, cerebrovascular events, and history of bleeding, Dr. Camm said.
That blunted BMI-mortality relationship, he said, “suggests that a lot of the effect is mediated through relatively traditional risk factors like hypertension and diabetes.”
The 2010 AFFIRM analysis by BMI, Dr. Lavie noted, “didn’t even look at the underweight; they actually threw them out.” Yet, such patients with AFib, who tend to be extremely frail or have chronic diseases or conditions other than the arrhythmia, are common. A take-home of the current study is that “the underweight with atrial fibrillation have a really bad prognosis.”
That message isn’t heard as much, he observed, “but is as important as saying that BMI 30 has the best prognosis. The worst prognosis is with the underweight or the really extreme obese.”
Dr. Camm discloses research funding from the British Heart Foundation. Disclosures for the other authors are in the report. Dr. Lavie has previously disclosed serving as a speaker and consultant for PAI Health and DSM Nutritional Products and is the author of “The Obesity Paradox: When Thinner Means Sicker and Heavier Means Healthier” (Avery, 2014).
A version of this article first appeared on Medscape.com.
FROM OPEN HEART
Ultrasound on par with CT for evaluating sarcopenia in patients with cirrhosis
Using ultrasound (US) to evaluate sarcopenic obesity in patients with cirrhosis may offer accuracy on par with computed tomography (CT), according to investigators.
US-based assessment presents a more affordable point-of-care strategy that limits radiation exposure, which enables sequential monitoring, reported lead author Sukhpal Dhariwal, MBBS, MD, of the Postgraduate Institute of Medical Education and Research, Chandigarh, India.
“Preliminary data in patients with liver disease ... suggest that US muscle assessment–derived indices, especially thigh muscle thickness, identify sarcopenia CT-skeletal muscle index (SMI) and also predict hospitalization and mortality,” the investigators wrote in Journal of Clinical Gastroenterology. “However, the applicability of US-based techniques to measure muscle mass in the high-risk group of patients with cirrhosis and sarcopenic obesity has not been evaluated.”
To address this knowledge gap, the investigators performed both US- and CT-based muscle assessments in 52 patients with obesity and evidence of cirrhosis; 40 patients were male and the mean age was 50.9 years. In all, 20 (38.5%) were diagnosed with sarcopenia based on CT-determined SMI scores of less than 39 cm2/m2 for women and 50 cm2/m2 for men.
US showed that it was similarly capable of categorizing patients. The modality significantly differentiated individuals with or without sarcopenia based on high area under the curve values in four muscle indices: quadriceps muscle thickness (0.98), quadriceps muscle feather index (0.95), forearm muscle thickness (0.85), and forearm feather index (0.80).
Direct comparison of US-based assessment against CT-based SMI revealed positive correlations, with significant r values ranging from 0.40 to 0.58. These correlations were stronger in a male-only subgroup analysis, in which r values ranged from 0.52 to 0.70. R values were not calculated in the female subgroup because of the small sample size (n = 12).
The investigators adjusted indices for height, which may pose bias for overestimating muscle mass. Another limitation is the small sample size.
“US-based assessment of sarcopenia has excellent diagnostic accuracy and correlates highly with cross-sectional imaging-based SMI in cirrhosis patients with sarcopenic obesity,” the investigators concluded. “US may serve as an easy-to-use, point-of-care tool for assessing sarcopenia in sarcopenic obesity with the advantage of repeated sequential assessment.”
According to Jamile Wakim-Fleming, MD, of the Cleveland Clinic, “US-based muscle mass assessment seems to be reliable, reproducible, and simple to perform and should be encouraged along with nutrition assessments in all patients with cirrhosis and obesity.”
In a written comment, Dr. Wakim-Fleming noted the importance of timely monitoring and intervention in this patient population.
“Considering the morbidity and the poor outcomes associated with sarcopenic obesity and its frequency in cirrhosis, it is important to make early diagnosis and institute a management plan to improve muscle mass and function,” she said.
The study was supported the Patient-Centered Outcomes Research Institute, the American Medical Association, and the American Heart Association. The investigators disclosed additional relationships with Pfizer, Bristol Myers Squibb, and Novartis. Dr. Wakim-Fleming reported no relevant conflicts of interest.
Using ultrasound (US) to evaluate sarcopenic obesity in patients with cirrhosis may offer accuracy on par with computed tomography (CT), according to investigators.
US-based assessment presents a more affordable point-of-care strategy that limits radiation exposure, which enables sequential monitoring, reported lead author Sukhpal Dhariwal, MBBS, MD, of the Postgraduate Institute of Medical Education and Research, Chandigarh, India.
“Preliminary data in patients with liver disease ... suggest that US muscle assessment–derived indices, especially thigh muscle thickness, identify sarcopenia CT-skeletal muscle index (SMI) and also predict hospitalization and mortality,” the investigators wrote in Journal of Clinical Gastroenterology. “However, the applicability of US-based techniques to measure muscle mass in the high-risk group of patients with cirrhosis and sarcopenic obesity has not been evaluated.”
To address this knowledge gap, the investigators performed both US- and CT-based muscle assessments in 52 patients with obesity and evidence of cirrhosis; 40 patients were male and the mean age was 50.9 years. In all, 20 (38.5%) were diagnosed with sarcopenia based on CT-determined SMI scores of less than 39 cm2/m2 for women and 50 cm2/m2 for men.
US showed that it was similarly capable of categorizing patients. The modality significantly differentiated individuals with or without sarcopenia based on high area under the curve values in four muscle indices: quadriceps muscle thickness (0.98), quadriceps muscle feather index (0.95), forearm muscle thickness (0.85), and forearm feather index (0.80).
Direct comparison of US-based assessment against CT-based SMI revealed positive correlations, with significant r values ranging from 0.40 to 0.58. These correlations were stronger in a male-only subgroup analysis, in which r values ranged from 0.52 to 0.70. R values were not calculated in the female subgroup because of the small sample size (n = 12).
The investigators adjusted indices for height, which may pose bias for overestimating muscle mass. Another limitation is the small sample size.
“US-based assessment of sarcopenia has excellent diagnostic accuracy and correlates highly with cross-sectional imaging-based SMI in cirrhosis patients with sarcopenic obesity,” the investigators concluded. “US may serve as an easy-to-use, point-of-care tool for assessing sarcopenia in sarcopenic obesity with the advantage of repeated sequential assessment.”
According to Jamile Wakim-Fleming, MD, of the Cleveland Clinic, “US-based muscle mass assessment seems to be reliable, reproducible, and simple to perform and should be encouraged along with nutrition assessments in all patients with cirrhosis and obesity.”
In a written comment, Dr. Wakim-Fleming noted the importance of timely monitoring and intervention in this patient population.
“Considering the morbidity and the poor outcomes associated with sarcopenic obesity and its frequency in cirrhosis, it is important to make early diagnosis and institute a management plan to improve muscle mass and function,” she said.
The study was supported the Patient-Centered Outcomes Research Institute, the American Medical Association, and the American Heart Association. The investigators disclosed additional relationships with Pfizer, Bristol Myers Squibb, and Novartis. Dr. Wakim-Fleming reported no relevant conflicts of interest.
Using ultrasound (US) to evaluate sarcopenic obesity in patients with cirrhosis may offer accuracy on par with computed tomography (CT), according to investigators.
US-based assessment presents a more affordable point-of-care strategy that limits radiation exposure, which enables sequential monitoring, reported lead author Sukhpal Dhariwal, MBBS, MD, of the Postgraduate Institute of Medical Education and Research, Chandigarh, India.
“Preliminary data in patients with liver disease ... suggest that US muscle assessment–derived indices, especially thigh muscle thickness, identify sarcopenia CT-skeletal muscle index (SMI) and also predict hospitalization and mortality,” the investigators wrote in Journal of Clinical Gastroenterology. “However, the applicability of US-based techniques to measure muscle mass in the high-risk group of patients with cirrhosis and sarcopenic obesity has not been evaluated.”
To address this knowledge gap, the investigators performed both US- and CT-based muscle assessments in 52 patients with obesity and evidence of cirrhosis; 40 patients were male and the mean age was 50.9 years. In all, 20 (38.5%) were diagnosed with sarcopenia based on CT-determined SMI scores of less than 39 cm2/m2 for women and 50 cm2/m2 for men.
US showed that it was similarly capable of categorizing patients. The modality significantly differentiated individuals with or without sarcopenia based on high area under the curve values in four muscle indices: quadriceps muscle thickness (0.98), quadriceps muscle feather index (0.95), forearm muscle thickness (0.85), and forearm feather index (0.80).
Direct comparison of US-based assessment against CT-based SMI revealed positive correlations, with significant r values ranging from 0.40 to 0.58. These correlations were stronger in a male-only subgroup analysis, in which r values ranged from 0.52 to 0.70. R values were not calculated in the female subgroup because of the small sample size (n = 12).
The investigators adjusted indices for height, which may pose bias for overestimating muscle mass. Another limitation is the small sample size.
“US-based assessment of sarcopenia has excellent diagnostic accuracy and correlates highly with cross-sectional imaging-based SMI in cirrhosis patients with sarcopenic obesity,” the investigators concluded. “US may serve as an easy-to-use, point-of-care tool for assessing sarcopenia in sarcopenic obesity with the advantage of repeated sequential assessment.”
According to Jamile Wakim-Fleming, MD, of the Cleveland Clinic, “US-based muscle mass assessment seems to be reliable, reproducible, and simple to perform and should be encouraged along with nutrition assessments in all patients with cirrhosis and obesity.”
In a written comment, Dr. Wakim-Fleming noted the importance of timely monitoring and intervention in this patient population.
“Considering the morbidity and the poor outcomes associated with sarcopenic obesity and its frequency in cirrhosis, it is important to make early diagnosis and institute a management plan to improve muscle mass and function,” she said.
The study was supported the Patient-Centered Outcomes Research Institute, the American Medical Association, and the American Heart Association. The investigators disclosed additional relationships with Pfizer, Bristol Myers Squibb, and Novartis. Dr. Wakim-Fleming reported no relevant conflicts of interest.
FROM JAMA INTERNAL MEDICINE
Obesity drug shortage triggers frustrations, workarounds
The glucagon-like peptide-1 (GLP-1) agonist semaglutide formulated for treating obesity (Wegovy) had a roaring takeoff a little more than a year ago, with surging patient demand after the U.S. Food and Drug Administration approved it in June 2021. But starting doses of the Wegovy form of semaglutide went missing in action starting late 2021 and continue to date, frustrating patients and their health care providers.
The arrival of Wegovy last year was hailed by obesity medicine specialists and others as a “game changer” for treating people with obesity because of semaglutide’s proven safety and efficacy at the subcutaneous dose of 2.4 mg delivered once a week to produce at least 15% weight loss in half the people who received it, as documented last year in results from one of the drug’s pivotal clinical trials.
But during the months following semaglutide’s approval for treating obesity (it also received an FDA marketing nod in late 2017 as Ozempic for treating type 2 diabetes), a worldwide shortage of Wegovy, including in the United States, emerged.
A manufacturing glitch shut down the primary location for production of U.S.-bound Wegovy injector pens for several months starting in late 2021, according to a December report from Novo Nordisk, the company that makes and markets the agent. (The Wegovy production issue appears to have had a very modest impact, especially in U.S. pharmacies, on the supply of semaglutide formulated as Ozempic, also marketed by Novo Nordisk, although Wegovy supply and demand have dramatically limited Ozempic availability in Australia.)
‘Unprecedented demand’ for Wegovy derailed when plant went offline
The supply side for Wegovy became so hopelessly broken that just months after U.S. sales began and immediately skyrocketed, Novo Nordisk made the remarkable decision to pull starting doses of Wegovy from the market to make it much harder to initiate patients (semaglutide and other GLP-1 agonists require gradual dose ramp-up to avoid gastrointestinal side effects), and the company publicly implored clinicians to not start new patients on the agent, which is where the status remains as of early August 2022.
Novo Nordisk’s financial report for the second quarter of 2022, released on Aug. 3, said the company “expects to make all Wegovy dose strengths available in the United States towards the end of 2022.”
A Dear Health Care Provider letter that Novo Nordisk posted on its U.S. Wegovy website last spring cited “unprecedented demand” that exceeded every prior product launch in the company’s history. It forced Novo Nordisk to pull the plug on all U.S. promotion of Wegovy and compelled the company to ask U.S. clinicians to halt new patient starts.
“I stopped offering Wegovy to new patients” since about the beginning of 2022, says Lauren D. Oshman, MD, a family and obesity medicine specialist at the University of Michigan, Ann Arbor. “It’s very frustrating to not have patients [with obesity] receive the optimal treatment available.” Although she adds that she tries to match obesity treatments to each patient’s clinical needs, and a GLP-1 agonist is not the first choice for every person with obesity.
“It was a disastrous rollout,” says Catherine W. Varney, DO, a family and obesity medicine specialist at the University of Virginia, Charlottesville. “It’s frustrating to know that the treatment is there but not being able to use it,” she said in an interview.
“I had about 800 patients on Wegovy” when the supply dropped earlier this year, and “I couldn’t handle the volume of messages that I got from patients,” recalls Angela Fitch, MD, associate director of the Massachusetts General Hospital Weight Center, Boston. “It was painful,” she said in an interview.
“Frustrating and chaotic,” is the description from Ivania M. Rizo, MD, director of obesity medicine at Boston Medical Center.
The liraglutide/Saxenda workaround
The upshot is that people with obesity and their health care providers have been busy devising workarounds to try to meet the intense demand for this drug-assisted approach to appetite control and weight loss. Their tactics run a wide gamut based on the crazy-quilt diversity of health insurance coverage across America.
Because the bottleneck for starting Wegovy resulted from unavailable starting doses (dosing starts at 0.25 mg delivered subcutaneously once a week, eventually ramping up to a maximum of 2.4 mg weekly), one option was to start patients on a different GLP-1 agonist, such as liraglutide (Saxenda, approved for obesity).
Starting a patient on liraglutide involves the same sort of up-titration and acclimation to a GLP-1 agonist that semaglutide requires, and transition between these agents seems feasible for at least some. It also means daily injections of liraglutide rather than the weekly schedule for semaglutide, although some patients prefer maintaining a daily dosing schedule. Another limitation of liraglutide is that evidence shows it is not nearly as effective for weight loss as semaglutide.
Results from the head-to-head STEP 8 trial, published in JAMA, showed an average weight loss from baseline of about 16% with semaglutide and about 6% with liraglutide (and about 2% with placebo).
A ‘reasonable’ evidence base, but more work
Changing from Saxenda to Wegovy, or from Wegovy to Saxenda, “would be reasonably evidence-based medicine,” said Dr. Oshman in an interview. She has managed a Wegovy-to-Saxenda switch for a “handful” of patients to deal with Wegovy shortages, but she has not yet moved anyone to Wegovy after a Saxenda initiation.
“No prospective study has looked at this transition,” but dose equivalence tables exist based on expert opinion, noted Dr. Oshman, as in this 2020 report.
Dr. Varney has several patients on the Saxenda-to-Wegovy track. She up-titrates patients on Saxenda to the maximum daily dose of 3.0 mg and then switches them to the 1.7 mg weekly dose of Wegovy, one of the “destination” Wegovy doses that has remained generally available during the shortage. But Dr. Varney’s experience is that only half of her patients made the changeover smoothly, with the others having “severe gastrointestinal distress,” including vomiting, she notes.
Dr. Fitch has also successfully used this Saxenda-to-Wegovy approach for some of her patients, but it hasn’t been easy.
“It’s more work and more prior authorizations. It’s harder and adds a layer of stress,” but, Dr. Fitch adds, “people are willing to work on it because the weight loss is worth it.”
The liraglutide to semaglutide shuffle is “doable,” says Dr. Rizo, “but I’m looking forward to not having to do it and being able to just start Wegovy.”
The tirzepatide coupon program works ‘off label’ for obesity
Another workaround depends on the FDA approval in May for tirzepatide (Mounjaro) for type 2 diabetes. Tirzepatide is a related GLP-1 agonist that also adds a second incretin-like agonist activity that mimics the glucose-dependent insulinotropic polypeptide.
Soon after approval, Lilly, the company that markets tirzepatide, started a U.S. coupon program geared exclusively to people with commercial insurance. Within certain refill and dollar limits, the program lets patients buy tirzepatide at pharmacies at an out-of-pocket cost of $25 for a 4-week supply (tirzepatide is also dosed by weekly subcutaneous injections). The program will extend into 2023.
Novo Nordisk offered U.S. patients with commercial insurance a similar discount when Wegovy first hit the U.S. market in 2021, but the program closed down once the supply shortage began.
Despite tirzepatide’s current approval only for type 2 diabetes, Dr. Varney has been successfully prescribing it to patients without diabetes off-label for weight loss.
“The coupons still work even when tirzepatide is used off-label,” she notes. And while the drug’s rollout is still only a couple of months old, so far, it’s gone “beautifully” with no hints of supply issues, she says.
But a major drawback to relying on an introductory coupon program that makes these agents affordable to patients is their ability to maintain treatment once the discounts inevitably end.
“We try to only prescribe agents that patients can continue to access,” says Dr. Fitch, who has had some patients with commercial insurance start on Wegovy with coupon discounts only to later lose access.
Many commercial U.S. insurers do not cover obesity treatments, a decision often driven by the employers who sponsor the coverage, she notes.
Study results have documented that when people with obesity stop taking a GLP-1 agonist their lost weight rebounds, as in a study that tracked people who stopped taking semaglutide.
Dr. Fitch has had success prescribing tirzepatide to patients with obesity but without diabetes who have certain types of Medicare drug coverage policies, which often do not deny off-label drug coverage. That approach works until patients reach the “donut hole” in their drug coverage and are faced with a certain level of out-of-pocket costs that can balloon to several thousand dollars.
Even more workarounds
Other approaches patients have used to acquire Wegovy include purchasing it in other countries, such as Canada or Brazil, says Dr. Fitch. But prices outside the United States, while substantially lower, can still be a barrier for many patients, notes Dr. Oshman.
Semaglutide in Canada goes for about $300 for a 4-week supply, roughly a quarter the U.S. price, she says, but is “still too high for many of my patients.”
Intense patient demand sometimes bordering on desperation has prompted some to seek semaglutide from private compounding pharmacies, a step clinicians regard as downright dangerous.
“Semaglutide from compounding pharmacies is not known to be safe. We feel strongly that it’s not something that people should do,” says Dr. Fitch.
“Compounding pharmacies have no FDA regulation. People don’t know what they’re getting. It’s dangerous,” agrees Dr. Varney. Physicians who refer people for privately compounded semaglutide “are taking advantage of desperate people,” she adds.
Although it seems likely that Novo Nordisk will soon sort out the supply problems and Wegovy will once again become more widely available, some of the issues patients have had with access to the weight loss medication stem from more systemic issues in the United States health insurance landscape: an unwillingness by payers to cover the costs of weight loss medications, a shortcoming that also exists for Medicare and Medicaid.
“We need to make obesity treatment a standard benefit, and not something that can be carved out,” says Dr. Fitch. People with obesity “deserve access to effective treatments for their disease,” she declares.
Dr. Oshman, Dr. Varney, and Dr. Rizo have reported no relevant financial relationships. Dr. Fitch has reported being an advisor to Jenny Craig.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) agonist semaglutide formulated for treating obesity (Wegovy) had a roaring takeoff a little more than a year ago, with surging patient demand after the U.S. Food and Drug Administration approved it in June 2021. But starting doses of the Wegovy form of semaglutide went missing in action starting late 2021 and continue to date, frustrating patients and their health care providers.
The arrival of Wegovy last year was hailed by obesity medicine specialists and others as a “game changer” for treating people with obesity because of semaglutide’s proven safety and efficacy at the subcutaneous dose of 2.4 mg delivered once a week to produce at least 15% weight loss in half the people who received it, as documented last year in results from one of the drug’s pivotal clinical trials.
But during the months following semaglutide’s approval for treating obesity (it also received an FDA marketing nod in late 2017 as Ozempic for treating type 2 diabetes), a worldwide shortage of Wegovy, including in the United States, emerged.
A manufacturing glitch shut down the primary location for production of U.S.-bound Wegovy injector pens for several months starting in late 2021, according to a December report from Novo Nordisk, the company that makes and markets the agent. (The Wegovy production issue appears to have had a very modest impact, especially in U.S. pharmacies, on the supply of semaglutide formulated as Ozempic, also marketed by Novo Nordisk, although Wegovy supply and demand have dramatically limited Ozempic availability in Australia.)
‘Unprecedented demand’ for Wegovy derailed when plant went offline
The supply side for Wegovy became so hopelessly broken that just months after U.S. sales began and immediately skyrocketed, Novo Nordisk made the remarkable decision to pull starting doses of Wegovy from the market to make it much harder to initiate patients (semaglutide and other GLP-1 agonists require gradual dose ramp-up to avoid gastrointestinal side effects), and the company publicly implored clinicians to not start new patients on the agent, which is where the status remains as of early August 2022.
Novo Nordisk’s financial report for the second quarter of 2022, released on Aug. 3, said the company “expects to make all Wegovy dose strengths available in the United States towards the end of 2022.”
A Dear Health Care Provider letter that Novo Nordisk posted on its U.S. Wegovy website last spring cited “unprecedented demand” that exceeded every prior product launch in the company’s history. It forced Novo Nordisk to pull the plug on all U.S. promotion of Wegovy and compelled the company to ask U.S. clinicians to halt new patient starts.
“I stopped offering Wegovy to new patients” since about the beginning of 2022, says Lauren D. Oshman, MD, a family and obesity medicine specialist at the University of Michigan, Ann Arbor. “It’s very frustrating to not have patients [with obesity] receive the optimal treatment available.” Although she adds that she tries to match obesity treatments to each patient’s clinical needs, and a GLP-1 agonist is not the first choice for every person with obesity.
“It was a disastrous rollout,” says Catherine W. Varney, DO, a family and obesity medicine specialist at the University of Virginia, Charlottesville. “It’s frustrating to know that the treatment is there but not being able to use it,” she said in an interview.
“I had about 800 patients on Wegovy” when the supply dropped earlier this year, and “I couldn’t handle the volume of messages that I got from patients,” recalls Angela Fitch, MD, associate director of the Massachusetts General Hospital Weight Center, Boston. “It was painful,” she said in an interview.
“Frustrating and chaotic,” is the description from Ivania M. Rizo, MD, director of obesity medicine at Boston Medical Center.
The liraglutide/Saxenda workaround
The upshot is that people with obesity and their health care providers have been busy devising workarounds to try to meet the intense demand for this drug-assisted approach to appetite control and weight loss. Their tactics run a wide gamut based on the crazy-quilt diversity of health insurance coverage across America.
Because the bottleneck for starting Wegovy resulted from unavailable starting doses (dosing starts at 0.25 mg delivered subcutaneously once a week, eventually ramping up to a maximum of 2.4 mg weekly), one option was to start patients on a different GLP-1 agonist, such as liraglutide (Saxenda, approved for obesity).
Starting a patient on liraglutide involves the same sort of up-titration and acclimation to a GLP-1 agonist that semaglutide requires, and transition between these agents seems feasible for at least some. It also means daily injections of liraglutide rather than the weekly schedule for semaglutide, although some patients prefer maintaining a daily dosing schedule. Another limitation of liraglutide is that evidence shows it is not nearly as effective for weight loss as semaglutide.
Results from the head-to-head STEP 8 trial, published in JAMA, showed an average weight loss from baseline of about 16% with semaglutide and about 6% with liraglutide (and about 2% with placebo).
A ‘reasonable’ evidence base, but more work
Changing from Saxenda to Wegovy, or from Wegovy to Saxenda, “would be reasonably evidence-based medicine,” said Dr. Oshman in an interview. She has managed a Wegovy-to-Saxenda switch for a “handful” of patients to deal with Wegovy shortages, but she has not yet moved anyone to Wegovy after a Saxenda initiation.
“No prospective study has looked at this transition,” but dose equivalence tables exist based on expert opinion, noted Dr. Oshman, as in this 2020 report.
Dr. Varney has several patients on the Saxenda-to-Wegovy track. She up-titrates patients on Saxenda to the maximum daily dose of 3.0 mg and then switches them to the 1.7 mg weekly dose of Wegovy, one of the “destination” Wegovy doses that has remained generally available during the shortage. But Dr. Varney’s experience is that only half of her patients made the changeover smoothly, with the others having “severe gastrointestinal distress,” including vomiting, she notes.
Dr. Fitch has also successfully used this Saxenda-to-Wegovy approach for some of her patients, but it hasn’t been easy.
“It’s more work and more prior authorizations. It’s harder and adds a layer of stress,” but, Dr. Fitch adds, “people are willing to work on it because the weight loss is worth it.”
The liraglutide to semaglutide shuffle is “doable,” says Dr. Rizo, “but I’m looking forward to not having to do it and being able to just start Wegovy.”
The tirzepatide coupon program works ‘off label’ for obesity
Another workaround depends on the FDA approval in May for tirzepatide (Mounjaro) for type 2 diabetes. Tirzepatide is a related GLP-1 agonist that also adds a second incretin-like agonist activity that mimics the glucose-dependent insulinotropic polypeptide.
Soon after approval, Lilly, the company that markets tirzepatide, started a U.S. coupon program geared exclusively to people with commercial insurance. Within certain refill and dollar limits, the program lets patients buy tirzepatide at pharmacies at an out-of-pocket cost of $25 for a 4-week supply (tirzepatide is also dosed by weekly subcutaneous injections). The program will extend into 2023.
Novo Nordisk offered U.S. patients with commercial insurance a similar discount when Wegovy first hit the U.S. market in 2021, but the program closed down once the supply shortage began.
Despite tirzepatide’s current approval only for type 2 diabetes, Dr. Varney has been successfully prescribing it to patients without diabetes off-label for weight loss.
“The coupons still work even when tirzepatide is used off-label,” she notes. And while the drug’s rollout is still only a couple of months old, so far, it’s gone “beautifully” with no hints of supply issues, she says.
But a major drawback to relying on an introductory coupon program that makes these agents affordable to patients is their ability to maintain treatment once the discounts inevitably end.
“We try to only prescribe agents that patients can continue to access,” says Dr. Fitch, who has had some patients with commercial insurance start on Wegovy with coupon discounts only to later lose access.
Many commercial U.S. insurers do not cover obesity treatments, a decision often driven by the employers who sponsor the coverage, she notes.
Study results have documented that when people with obesity stop taking a GLP-1 agonist their lost weight rebounds, as in a study that tracked people who stopped taking semaglutide.
Dr. Fitch has had success prescribing tirzepatide to patients with obesity but without diabetes who have certain types of Medicare drug coverage policies, which often do not deny off-label drug coverage. That approach works until patients reach the “donut hole” in their drug coverage and are faced with a certain level of out-of-pocket costs that can balloon to several thousand dollars.
Even more workarounds
Other approaches patients have used to acquire Wegovy include purchasing it in other countries, such as Canada or Brazil, says Dr. Fitch. But prices outside the United States, while substantially lower, can still be a barrier for many patients, notes Dr. Oshman.
Semaglutide in Canada goes for about $300 for a 4-week supply, roughly a quarter the U.S. price, she says, but is “still too high for many of my patients.”
Intense patient demand sometimes bordering on desperation has prompted some to seek semaglutide from private compounding pharmacies, a step clinicians regard as downright dangerous.
“Semaglutide from compounding pharmacies is not known to be safe. We feel strongly that it’s not something that people should do,” says Dr. Fitch.
“Compounding pharmacies have no FDA regulation. People don’t know what they’re getting. It’s dangerous,” agrees Dr. Varney. Physicians who refer people for privately compounded semaglutide “are taking advantage of desperate people,” she adds.
Although it seems likely that Novo Nordisk will soon sort out the supply problems and Wegovy will once again become more widely available, some of the issues patients have had with access to the weight loss medication stem from more systemic issues in the United States health insurance landscape: an unwillingness by payers to cover the costs of weight loss medications, a shortcoming that also exists for Medicare and Medicaid.
“We need to make obesity treatment a standard benefit, and not something that can be carved out,” says Dr. Fitch. People with obesity “deserve access to effective treatments for their disease,” she declares.
Dr. Oshman, Dr. Varney, and Dr. Rizo have reported no relevant financial relationships. Dr. Fitch has reported being an advisor to Jenny Craig.
A version of this article first appeared on Medscape.com.
The glucagon-like peptide-1 (GLP-1) agonist semaglutide formulated for treating obesity (Wegovy) had a roaring takeoff a little more than a year ago, with surging patient demand after the U.S. Food and Drug Administration approved it in June 2021. But starting doses of the Wegovy form of semaglutide went missing in action starting late 2021 and continue to date, frustrating patients and their health care providers.
The arrival of Wegovy last year was hailed by obesity medicine specialists and others as a “game changer” for treating people with obesity because of semaglutide’s proven safety and efficacy at the subcutaneous dose of 2.4 mg delivered once a week to produce at least 15% weight loss in half the people who received it, as documented last year in results from one of the drug’s pivotal clinical trials.
But during the months following semaglutide’s approval for treating obesity (it also received an FDA marketing nod in late 2017 as Ozempic for treating type 2 diabetes), a worldwide shortage of Wegovy, including in the United States, emerged.
A manufacturing glitch shut down the primary location for production of U.S.-bound Wegovy injector pens for several months starting in late 2021, according to a December report from Novo Nordisk, the company that makes and markets the agent. (The Wegovy production issue appears to have had a very modest impact, especially in U.S. pharmacies, on the supply of semaglutide formulated as Ozempic, also marketed by Novo Nordisk, although Wegovy supply and demand have dramatically limited Ozempic availability in Australia.)
‘Unprecedented demand’ for Wegovy derailed when plant went offline
The supply side for Wegovy became so hopelessly broken that just months after U.S. sales began and immediately skyrocketed, Novo Nordisk made the remarkable decision to pull starting doses of Wegovy from the market to make it much harder to initiate patients (semaglutide and other GLP-1 agonists require gradual dose ramp-up to avoid gastrointestinal side effects), and the company publicly implored clinicians to not start new patients on the agent, which is where the status remains as of early August 2022.
Novo Nordisk’s financial report for the second quarter of 2022, released on Aug. 3, said the company “expects to make all Wegovy dose strengths available in the United States towards the end of 2022.”
A Dear Health Care Provider letter that Novo Nordisk posted on its U.S. Wegovy website last spring cited “unprecedented demand” that exceeded every prior product launch in the company’s history. It forced Novo Nordisk to pull the plug on all U.S. promotion of Wegovy and compelled the company to ask U.S. clinicians to halt new patient starts.
“I stopped offering Wegovy to new patients” since about the beginning of 2022, says Lauren D. Oshman, MD, a family and obesity medicine specialist at the University of Michigan, Ann Arbor. “It’s very frustrating to not have patients [with obesity] receive the optimal treatment available.” Although she adds that she tries to match obesity treatments to each patient’s clinical needs, and a GLP-1 agonist is not the first choice for every person with obesity.
“It was a disastrous rollout,” says Catherine W. Varney, DO, a family and obesity medicine specialist at the University of Virginia, Charlottesville. “It’s frustrating to know that the treatment is there but not being able to use it,” she said in an interview.
“I had about 800 patients on Wegovy” when the supply dropped earlier this year, and “I couldn’t handle the volume of messages that I got from patients,” recalls Angela Fitch, MD, associate director of the Massachusetts General Hospital Weight Center, Boston. “It was painful,” she said in an interview.
“Frustrating and chaotic,” is the description from Ivania M. Rizo, MD, director of obesity medicine at Boston Medical Center.
The liraglutide/Saxenda workaround
The upshot is that people with obesity and their health care providers have been busy devising workarounds to try to meet the intense demand for this drug-assisted approach to appetite control and weight loss. Their tactics run a wide gamut based on the crazy-quilt diversity of health insurance coverage across America.
Because the bottleneck for starting Wegovy resulted from unavailable starting doses (dosing starts at 0.25 mg delivered subcutaneously once a week, eventually ramping up to a maximum of 2.4 mg weekly), one option was to start patients on a different GLP-1 agonist, such as liraglutide (Saxenda, approved for obesity).
Starting a patient on liraglutide involves the same sort of up-titration and acclimation to a GLP-1 agonist that semaglutide requires, and transition between these agents seems feasible for at least some. It also means daily injections of liraglutide rather than the weekly schedule for semaglutide, although some patients prefer maintaining a daily dosing schedule. Another limitation of liraglutide is that evidence shows it is not nearly as effective for weight loss as semaglutide.
Results from the head-to-head STEP 8 trial, published in JAMA, showed an average weight loss from baseline of about 16% with semaglutide and about 6% with liraglutide (and about 2% with placebo).
A ‘reasonable’ evidence base, but more work
Changing from Saxenda to Wegovy, or from Wegovy to Saxenda, “would be reasonably evidence-based medicine,” said Dr. Oshman in an interview. She has managed a Wegovy-to-Saxenda switch for a “handful” of patients to deal with Wegovy shortages, but she has not yet moved anyone to Wegovy after a Saxenda initiation.
“No prospective study has looked at this transition,” but dose equivalence tables exist based on expert opinion, noted Dr. Oshman, as in this 2020 report.
Dr. Varney has several patients on the Saxenda-to-Wegovy track. She up-titrates patients on Saxenda to the maximum daily dose of 3.0 mg and then switches them to the 1.7 mg weekly dose of Wegovy, one of the “destination” Wegovy doses that has remained generally available during the shortage. But Dr. Varney’s experience is that only half of her patients made the changeover smoothly, with the others having “severe gastrointestinal distress,” including vomiting, she notes.
Dr. Fitch has also successfully used this Saxenda-to-Wegovy approach for some of her patients, but it hasn’t been easy.
“It’s more work and more prior authorizations. It’s harder and adds a layer of stress,” but, Dr. Fitch adds, “people are willing to work on it because the weight loss is worth it.”
The liraglutide to semaglutide shuffle is “doable,” says Dr. Rizo, “but I’m looking forward to not having to do it and being able to just start Wegovy.”
The tirzepatide coupon program works ‘off label’ for obesity
Another workaround depends on the FDA approval in May for tirzepatide (Mounjaro) for type 2 diabetes. Tirzepatide is a related GLP-1 agonist that also adds a second incretin-like agonist activity that mimics the glucose-dependent insulinotropic polypeptide.
Soon after approval, Lilly, the company that markets tirzepatide, started a U.S. coupon program geared exclusively to people with commercial insurance. Within certain refill and dollar limits, the program lets patients buy tirzepatide at pharmacies at an out-of-pocket cost of $25 for a 4-week supply (tirzepatide is also dosed by weekly subcutaneous injections). The program will extend into 2023.
Novo Nordisk offered U.S. patients with commercial insurance a similar discount when Wegovy first hit the U.S. market in 2021, but the program closed down once the supply shortage began.
Despite tirzepatide’s current approval only for type 2 diabetes, Dr. Varney has been successfully prescribing it to patients without diabetes off-label for weight loss.
“The coupons still work even when tirzepatide is used off-label,” she notes. And while the drug’s rollout is still only a couple of months old, so far, it’s gone “beautifully” with no hints of supply issues, she says.
But a major drawback to relying on an introductory coupon program that makes these agents affordable to patients is their ability to maintain treatment once the discounts inevitably end.
“We try to only prescribe agents that patients can continue to access,” says Dr. Fitch, who has had some patients with commercial insurance start on Wegovy with coupon discounts only to later lose access.
Many commercial U.S. insurers do not cover obesity treatments, a decision often driven by the employers who sponsor the coverage, she notes.
Study results have documented that when people with obesity stop taking a GLP-1 agonist their lost weight rebounds, as in a study that tracked people who stopped taking semaglutide.
Dr. Fitch has had success prescribing tirzepatide to patients with obesity but without diabetes who have certain types of Medicare drug coverage policies, which often do not deny off-label drug coverage. That approach works until patients reach the “donut hole” in their drug coverage and are faced with a certain level of out-of-pocket costs that can balloon to several thousand dollars.
Even more workarounds
Other approaches patients have used to acquire Wegovy include purchasing it in other countries, such as Canada or Brazil, says Dr. Fitch. But prices outside the United States, while substantially lower, can still be a barrier for many patients, notes Dr. Oshman.
Semaglutide in Canada goes for about $300 for a 4-week supply, roughly a quarter the U.S. price, she says, but is “still too high for many of my patients.”
Intense patient demand sometimes bordering on desperation has prompted some to seek semaglutide from private compounding pharmacies, a step clinicians regard as downright dangerous.
“Semaglutide from compounding pharmacies is not known to be safe. We feel strongly that it’s not something that people should do,” says Dr. Fitch.
“Compounding pharmacies have no FDA regulation. People don’t know what they’re getting. It’s dangerous,” agrees Dr. Varney. Physicians who refer people for privately compounded semaglutide “are taking advantage of desperate people,” she adds.
Although it seems likely that Novo Nordisk will soon sort out the supply problems and Wegovy will once again become more widely available, some of the issues patients have had with access to the weight loss medication stem from more systemic issues in the United States health insurance landscape: an unwillingness by payers to cover the costs of weight loss medications, a shortcoming that also exists for Medicare and Medicaid.
“We need to make obesity treatment a standard benefit, and not something that can be carved out,” says Dr. Fitch. People with obesity “deserve access to effective treatments for their disease,” she declares.
Dr. Oshman, Dr. Varney, and Dr. Rizo have reported no relevant financial relationships. Dr. Fitch has reported being an advisor to Jenny Craig.
A version of this article first appeared on Medscape.com.
Weight-loss surgery has a big effect on marriage
Kristal was only in her mid-30s when she decided to have surgery. Her doctor said it was too early. But the Oregon mom of three had found herself in the hospital twice for obesity-related lung complications before her 35th birthday. So she got the gastric sleeve.
And at first it seemed like the best decision for her and her family. She was losing weight – 100 pounds in 16 months – and so was her husband. The whole family was more active and seemed to have more energy. But then her husband’s weight began to creep back up.
While she joined a running group and signed up for half-marathons, her husband’s depression and drinking worsened. The healthier lifestyle they’d shared was now an unspoken wedge between them.
And the added attention Kristal was getting from men and women because of her thinner size only added to the tension. After 30 years together and 22 years of marriage, the high school sweethearts divorced in June 2021. Kristal’s weight loss wasn’t the only problem, but she and her ex-husband believe it was the beginning of the end.
An unexpected outcome?
New research from the University of Pittsburgh found that Kristal’s experience is a common one. The study looked at data from 1,441 bariatric surgery patients and found that never-married patients were over 50% more likely to get married, and married patients were more than twice as likely to get divorced, compared to the general U.S. population.
This U.S. data follows two Scandinavian studies from 2018 and 2020 that found similar relationship changes after bariatric surgery. But the postsurgery divorce rate in the United States was only about half that found in the Danish and Swedish studies, according to the new study published in Annals of Surgery.
It’s important to note that even with an increase in the divorce rate, most marriages in the study were unchanged, said epidemiologist and lead author Wendy King, PhD. In fact, 81% of couples were still married 5 years after surgery. But where the U.S. population has a divorce rate of 3.5%, bariatric patients in the study had an 8% divorce rate. Likewise, those who’d never been married before the surgery had a marriage rate of 18%, compared to 7% in the U.S. population.
Surgery certainly isn’t a death sentence for a patient’s love life. But the uptick in marriage and divorce suggests bariatric surgery significantly impacts how people engage in relationships.
“It makes sense,” said clinical psychologist Rachel Goldman, PhD, who specializes in health and wellness issues and bariatric surgery cases in New York City. “People are changing their lifestyle.” And those changes don’t start or stop the day of surgery, they begin as soon as someone decides to have surgery and continue as a lifelong process, she said.
For some patients, these healthy habits may offer a “new lease on life,” said Dr. King. According to the study, patients who had better physical health after surgery were more likely to get married.
But the continual lifestyle changes can dramatically impact the rituals of existing relationships, said Dr. Goldman. Maybe a couple loved to go out and enjoy an extravagant meal before surgery, or they had ice cream and watched a movie every Friday. The habit changes that come with bariatric surgery can require one partner to focus less on those rituals.
These sorts of changes may leave one or both people feeling like their partner is turning away from them, said Don Cole, DMin, a relationship therapist and clinical director at the Gottman Institute in Seattle, a think tank focused on the science of relationships. The person who had surgery may feel unsupported in the new journey if the partner keeps advocating for unhealthy habits, he said. And the person who didn’t have surgery may feel cast aside by the partner’s new health priorities.
Changes, even those that are positive and healthy, create a kind of crisis for relationships, Dr. Cole said. It’s not just bariatric surgery. Bringing a baby into the home, infertility treatments, and substance abuse recovery are all considered positive changes that are also predictors of relationship dissatisfaction and divorce, he said.
A couple could have a range of emotions after one partner gets bariatric surgery, Dr. Cole said. Unfortunately, “my experience as a therapist says they aren’t that good [at talking about it],” he said.
But bariatric surgery isn’t the only thing at play in these relationship changes, according to the study. Married patients had a much lower chance of separation or divorce (13%) than patients who were unmarried but living together (44%) by 5 years after surgery. Similarly, most people who were already separated either got divorced or resumed being married. It’s as if the surgery and lifestyle changes served as a catalyst for people who already had one foot out of (or in) the door, Dr. Goldman said.
A high sexual desire after surgery was also a predictor of divorce. In fact, there were more things before surgery that impacted divorce than surgery-related changes. It’s possible that many of these patients are “on the path toward change already,” Dr. King said. “Who knows how much the surgery had to do with it.”
Dr. Goldman recalled a patient who, before surgery, had very low self-worth. She wasn’t satisfied with her relationship but admitted to staying because she didn’t believe she could do any better than her current partner. After surgery, her perspective radically changed. She started to get healthier, invested in her education, and changed jobs. And when her partner refused to join her in making changes, she left. Maybe some of these patients “were already thinking about leaving but just didn’t have the confidence,” Dr. Goldman said.
Still, it’s critical that patients receive more counseling on how choosing to have bariatric surgery can impact their relationship before and after their weight loss procedure, Dr. King said. It should be the standard of care.
Currently, relationship-specific counseling isn’t required, Dr. Goldman said. Most programs do require a psychosocial evaluation before surgery, “but they are quite varied.” And even in programs where relationships are mentioned, there often isn’t a psychologist or licensed mental health professional on the team.
Since Dr. King’s previous research on substance abuse after bariatric surgery changed common practice in the field, Dr. Goldman said she hopes the new data will have a similar influence and relationship counseling will become the norm.
Dr. Cole actually had bariatric surgery. He recalled potential relationship issues were briefly mentioned. Someone at the clinic said if his marriage felt challenged, he should seek help from a professional, and that was it.
For Dr. Cole, there were unexpected negative feelings of shame and disappointment after surgery. He felt the extreme weight loss was all his colleagues could talk about and was very disappointed when there was no change in his chronic pain, a primary reason he had the procedure.
Fortunately, he could talk to his wife – also is a relationship therapist at Gottman – about the range of emotions. “One of the things that we know that creates a deep sense of trust is [when] I know my partner is there for me when I’m not well,” Dr. Cole said.
But these negative emotions can be the very things that feel most difficult to talk about or hear from a partner. It’s hard to share our own negative feelings and to hear someone else’s, Dr. Cole said.
He advises creating a new “ritual of connection: moments in time when you plan to turn toward one another.”
That could be a daily walk, where you intentionally talk about the surgery-related changes that both of you have had. Dr. Cole said to ask yourself, “Are we intentional about turning toward one another in those [challenging] moments?”
A version of this article first appeared on WebMD.com.
Kristal was only in her mid-30s when she decided to have surgery. Her doctor said it was too early. But the Oregon mom of three had found herself in the hospital twice for obesity-related lung complications before her 35th birthday. So she got the gastric sleeve.
And at first it seemed like the best decision for her and her family. She was losing weight – 100 pounds in 16 months – and so was her husband. The whole family was more active and seemed to have more energy. But then her husband’s weight began to creep back up.
While she joined a running group and signed up for half-marathons, her husband’s depression and drinking worsened. The healthier lifestyle they’d shared was now an unspoken wedge between them.
And the added attention Kristal was getting from men and women because of her thinner size only added to the tension. After 30 years together and 22 years of marriage, the high school sweethearts divorced in June 2021. Kristal’s weight loss wasn’t the only problem, but she and her ex-husband believe it was the beginning of the end.
An unexpected outcome?
New research from the University of Pittsburgh found that Kristal’s experience is a common one. The study looked at data from 1,441 bariatric surgery patients and found that never-married patients were over 50% more likely to get married, and married patients were more than twice as likely to get divorced, compared to the general U.S. population.
This U.S. data follows two Scandinavian studies from 2018 and 2020 that found similar relationship changes after bariatric surgery. But the postsurgery divorce rate in the United States was only about half that found in the Danish and Swedish studies, according to the new study published in Annals of Surgery.
It’s important to note that even with an increase in the divorce rate, most marriages in the study were unchanged, said epidemiologist and lead author Wendy King, PhD. In fact, 81% of couples were still married 5 years after surgery. But where the U.S. population has a divorce rate of 3.5%, bariatric patients in the study had an 8% divorce rate. Likewise, those who’d never been married before the surgery had a marriage rate of 18%, compared to 7% in the U.S. population.
Surgery certainly isn’t a death sentence for a patient’s love life. But the uptick in marriage and divorce suggests bariatric surgery significantly impacts how people engage in relationships.
“It makes sense,” said clinical psychologist Rachel Goldman, PhD, who specializes in health and wellness issues and bariatric surgery cases in New York City. “People are changing their lifestyle.” And those changes don’t start or stop the day of surgery, they begin as soon as someone decides to have surgery and continue as a lifelong process, she said.
For some patients, these healthy habits may offer a “new lease on life,” said Dr. King. According to the study, patients who had better physical health after surgery were more likely to get married.
But the continual lifestyle changes can dramatically impact the rituals of existing relationships, said Dr. Goldman. Maybe a couple loved to go out and enjoy an extravagant meal before surgery, or they had ice cream and watched a movie every Friday. The habit changes that come with bariatric surgery can require one partner to focus less on those rituals.
These sorts of changes may leave one or both people feeling like their partner is turning away from them, said Don Cole, DMin, a relationship therapist and clinical director at the Gottman Institute in Seattle, a think tank focused on the science of relationships. The person who had surgery may feel unsupported in the new journey if the partner keeps advocating for unhealthy habits, he said. And the person who didn’t have surgery may feel cast aside by the partner’s new health priorities.
Changes, even those that are positive and healthy, create a kind of crisis for relationships, Dr. Cole said. It’s not just bariatric surgery. Bringing a baby into the home, infertility treatments, and substance abuse recovery are all considered positive changes that are also predictors of relationship dissatisfaction and divorce, he said.
A couple could have a range of emotions after one partner gets bariatric surgery, Dr. Cole said. Unfortunately, “my experience as a therapist says they aren’t that good [at talking about it],” he said.
But bariatric surgery isn’t the only thing at play in these relationship changes, according to the study. Married patients had a much lower chance of separation or divorce (13%) than patients who were unmarried but living together (44%) by 5 years after surgery. Similarly, most people who were already separated either got divorced or resumed being married. It’s as if the surgery and lifestyle changes served as a catalyst for people who already had one foot out of (or in) the door, Dr. Goldman said.
A high sexual desire after surgery was also a predictor of divorce. In fact, there were more things before surgery that impacted divorce than surgery-related changes. It’s possible that many of these patients are “on the path toward change already,” Dr. King said. “Who knows how much the surgery had to do with it.”
Dr. Goldman recalled a patient who, before surgery, had very low self-worth. She wasn’t satisfied with her relationship but admitted to staying because she didn’t believe she could do any better than her current partner. After surgery, her perspective radically changed. She started to get healthier, invested in her education, and changed jobs. And when her partner refused to join her in making changes, she left. Maybe some of these patients “were already thinking about leaving but just didn’t have the confidence,” Dr. Goldman said.
Still, it’s critical that patients receive more counseling on how choosing to have bariatric surgery can impact their relationship before and after their weight loss procedure, Dr. King said. It should be the standard of care.
Currently, relationship-specific counseling isn’t required, Dr. Goldman said. Most programs do require a psychosocial evaluation before surgery, “but they are quite varied.” And even in programs where relationships are mentioned, there often isn’t a psychologist or licensed mental health professional on the team.
Since Dr. King’s previous research on substance abuse after bariatric surgery changed common practice in the field, Dr. Goldman said she hopes the new data will have a similar influence and relationship counseling will become the norm.
Dr. Cole actually had bariatric surgery. He recalled potential relationship issues were briefly mentioned. Someone at the clinic said if his marriage felt challenged, he should seek help from a professional, and that was it.
For Dr. Cole, there were unexpected negative feelings of shame and disappointment after surgery. He felt the extreme weight loss was all his colleagues could talk about and was very disappointed when there was no change in his chronic pain, a primary reason he had the procedure.
Fortunately, he could talk to his wife – also is a relationship therapist at Gottman – about the range of emotions. “One of the things that we know that creates a deep sense of trust is [when] I know my partner is there for me when I’m not well,” Dr. Cole said.
But these negative emotions can be the very things that feel most difficult to talk about or hear from a partner. It’s hard to share our own negative feelings and to hear someone else’s, Dr. Cole said.
He advises creating a new “ritual of connection: moments in time when you plan to turn toward one another.”
That could be a daily walk, where you intentionally talk about the surgery-related changes that both of you have had. Dr. Cole said to ask yourself, “Are we intentional about turning toward one another in those [challenging] moments?”
A version of this article first appeared on WebMD.com.
Kristal was only in her mid-30s when she decided to have surgery. Her doctor said it was too early. But the Oregon mom of three had found herself in the hospital twice for obesity-related lung complications before her 35th birthday. So she got the gastric sleeve.
And at first it seemed like the best decision for her and her family. She was losing weight – 100 pounds in 16 months – and so was her husband. The whole family was more active and seemed to have more energy. But then her husband’s weight began to creep back up.
While she joined a running group and signed up for half-marathons, her husband’s depression and drinking worsened. The healthier lifestyle they’d shared was now an unspoken wedge between them.
And the added attention Kristal was getting from men and women because of her thinner size only added to the tension. After 30 years together and 22 years of marriage, the high school sweethearts divorced in June 2021. Kristal’s weight loss wasn’t the only problem, but she and her ex-husband believe it was the beginning of the end.
An unexpected outcome?
New research from the University of Pittsburgh found that Kristal’s experience is a common one. The study looked at data from 1,441 bariatric surgery patients and found that never-married patients were over 50% more likely to get married, and married patients were more than twice as likely to get divorced, compared to the general U.S. population.
This U.S. data follows two Scandinavian studies from 2018 and 2020 that found similar relationship changes after bariatric surgery. But the postsurgery divorce rate in the United States was only about half that found in the Danish and Swedish studies, according to the new study published in Annals of Surgery.
It’s important to note that even with an increase in the divorce rate, most marriages in the study were unchanged, said epidemiologist and lead author Wendy King, PhD. In fact, 81% of couples were still married 5 years after surgery. But where the U.S. population has a divorce rate of 3.5%, bariatric patients in the study had an 8% divorce rate. Likewise, those who’d never been married before the surgery had a marriage rate of 18%, compared to 7% in the U.S. population.
Surgery certainly isn’t a death sentence for a patient’s love life. But the uptick in marriage and divorce suggests bariatric surgery significantly impacts how people engage in relationships.
“It makes sense,” said clinical psychologist Rachel Goldman, PhD, who specializes in health and wellness issues and bariatric surgery cases in New York City. “People are changing their lifestyle.” And those changes don’t start or stop the day of surgery, they begin as soon as someone decides to have surgery and continue as a lifelong process, she said.
For some patients, these healthy habits may offer a “new lease on life,” said Dr. King. According to the study, patients who had better physical health after surgery were more likely to get married.
But the continual lifestyle changes can dramatically impact the rituals of existing relationships, said Dr. Goldman. Maybe a couple loved to go out and enjoy an extravagant meal before surgery, or they had ice cream and watched a movie every Friday. The habit changes that come with bariatric surgery can require one partner to focus less on those rituals.
These sorts of changes may leave one or both people feeling like their partner is turning away from them, said Don Cole, DMin, a relationship therapist and clinical director at the Gottman Institute in Seattle, a think tank focused on the science of relationships. The person who had surgery may feel unsupported in the new journey if the partner keeps advocating for unhealthy habits, he said. And the person who didn’t have surgery may feel cast aside by the partner’s new health priorities.
Changes, even those that are positive and healthy, create a kind of crisis for relationships, Dr. Cole said. It’s not just bariatric surgery. Bringing a baby into the home, infertility treatments, and substance abuse recovery are all considered positive changes that are also predictors of relationship dissatisfaction and divorce, he said.
A couple could have a range of emotions after one partner gets bariatric surgery, Dr. Cole said. Unfortunately, “my experience as a therapist says they aren’t that good [at talking about it],” he said.
But bariatric surgery isn’t the only thing at play in these relationship changes, according to the study. Married patients had a much lower chance of separation or divorce (13%) than patients who were unmarried but living together (44%) by 5 years after surgery. Similarly, most people who were already separated either got divorced or resumed being married. It’s as if the surgery and lifestyle changes served as a catalyst for people who already had one foot out of (or in) the door, Dr. Goldman said.
A high sexual desire after surgery was also a predictor of divorce. In fact, there were more things before surgery that impacted divorce than surgery-related changes. It’s possible that many of these patients are “on the path toward change already,” Dr. King said. “Who knows how much the surgery had to do with it.”
Dr. Goldman recalled a patient who, before surgery, had very low self-worth. She wasn’t satisfied with her relationship but admitted to staying because she didn’t believe she could do any better than her current partner. After surgery, her perspective radically changed. She started to get healthier, invested in her education, and changed jobs. And when her partner refused to join her in making changes, she left. Maybe some of these patients “were already thinking about leaving but just didn’t have the confidence,” Dr. Goldman said.
Still, it’s critical that patients receive more counseling on how choosing to have bariatric surgery can impact their relationship before and after their weight loss procedure, Dr. King said. It should be the standard of care.
Currently, relationship-specific counseling isn’t required, Dr. Goldman said. Most programs do require a psychosocial evaluation before surgery, “but they are quite varied.” And even in programs where relationships are mentioned, there often isn’t a psychologist or licensed mental health professional on the team.
Since Dr. King’s previous research on substance abuse after bariatric surgery changed common practice in the field, Dr. Goldman said she hopes the new data will have a similar influence and relationship counseling will become the norm.
Dr. Cole actually had bariatric surgery. He recalled potential relationship issues were briefly mentioned. Someone at the clinic said if his marriage felt challenged, he should seek help from a professional, and that was it.
For Dr. Cole, there were unexpected negative feelings of shame and disappointment after surgery. He felt the extreme weight loss was all his colleagues could talk about and was very disappointed when there was no change in his chronic pain, a primary reason he had the procedure.
Fortunately, he could talk to his wife – also is a relationship therapist at Gottman – about the range of emotions. “One of the things that we know that creates a deep sense of trust is [when] I know my partner is there for me when I’m not well,” Dr. Cole said.
But these negative emotions can be the very things that feel most difficult to talk about or hear from a partner. It’s hard to share our own negative feelings and to hear someone else’s, Dr. Cole said.
He advises creating a new “ritual of connection: moments in time when you plan to turn toward one another.”
That could be a daily walk, where you intentionally talk about the surgery-related changes that both of you have had. Dr. Cole said to ask yourself, “Are we intentional about turning toward one another in those [challenging] moments?”
A version of this article first appeared on WebMD.com.
FROM ANNALS OF SURGERY
Young adults who learn how to cook eat more veggies
Obesity remains a significant risk factor for numerous diseases, and is often a problem in young adults, who often fall back on fast food and other less-healthy meals associated with a lower quality diet, lead author Carol S. O’Neal, PhD, of the University of Louisville (Ky.), said in an interview.
Previous research involving Social Cognitive Theory and goal-setting to promote self-efficacy and behavior changes has shown success in improving eating habits in young adults, but adding video technology for an additional education element has not been well studied, Dr. O’Neal and colleagues wrote in the Journal of Nutrition Education and Behavior.
Methods and results
In the study, 138 college students aged 18-40 years participated in a 15-week pilot intervention course at a large, metropolitan university. The course included lectures on a topic, such as carbohydrates, and included skill-based activities, such as how to read an ingredient list, and discussion of how these skills could improve healthier eating and meet nutrition goals, such as eating more whole grains.
A total of 77 completed the study in person, and 61 participated online. The majority (59%) were college sophomores, 74% were White, and 82% were female.
The course engaged the students in weekly food challenges to apply their knowledge and develop better eating habits and behaviors. The challenges were accompanied by cooking videos related to each week’s topic, such as how to make overnight oats for the healthy carbohydrates/whole grains week.
Students also selected two goals each week, such as choosing whole grain foods to increase fiber consumption, from a list of 10-15 goals, and were required to write weekly reflections to track their progress toward these goals. Goal-setting was based on the strategy of creating goals that are specific, measurable, attainable, realistic, and time-bound (the SMART method).
The main outcomes were increased consumption of fruits and vegetables, improved skills in cooking and healthy eating, and improved attitudes about healthy cooking and eating. The researchers surveyed the students to determine whether these outcomes were met.
Students participating in the study indicated that they met the goal of eating at least five servings of fruits and vegetables per day more often after the course than before, the researchers wrote.
By the course’s end, the students showed significant increases in consumption of fruits and vegetables (P < .001 for both), and in the self-efficacy related to consumption of produce (P = .004); cooking (P = .002;, and using more fruits, vegetables, and seasonings rather than salt in cooking (P = .001).
A review of the students’ written reflections illustrated positive behavior changes such as planning meals before shopping, preparing meals in advance on weekends, taking lunch to school, and using herbs and spices, the researchers noted.
“Self-directed SMART goals set you up for success by making goals specific, measurable, achievable, realistic, and timely,” Dr. O’Neal said in an interview. “The SMART method helps push you further, gives you a sense of direction, and helps you organize and reach your goals,” but self-monitoring and social support are also needed for success. The takeaway message for clinicians is that use of a self-directed goal-setting strategy may be more effective at changing dietary behaviors and promoting self-efficacy than a traditional dietary prescription.
In addition, “this model could be used to address a variety of health outcomes in dietetics, health education and community health programs,” said Dr. O’Neal. “I think the key components of this intervention are teaching SMART goal setting, self-monitoring, and social support of successes. I see time as a main barrier, but this barrier could be reduced for populations who are able to use online learning. Our intervention was successful for in-person and online learning.”
Other areas for future research include evaluation of progress that combines quantitative data and qualitative reflections, she said.
Real-world applications
“Clinicians have limited time to address behavioral counseling, and this study offers an opportunity to reach patients not only in class sessions, but virtually,” M. Susan Jay, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview.
Although the findings from the study are not new, the knowledge can be used by clinicians to help promote behavior change. The study also showcased the use of additional tools, such as weekly food challenges, to impact college students who often consume high-fat diets in nonmedical settings, Dr. Jay said.
For consumers, the real-world implications are exciting, Dr. Jay said.
“People are increasingly attempting to “eat healthy” and despite clinicians wanting to impact healthy eating, limited office visits may not be conducive to behavioral change,” she said.
The current study was important as a way to identify tactics to improve the diet and nutrition of young adults, Margaret Thew, DNP, FNP-BC, medical director of adolescent medicine at the University of Wisconsin–Madison, said in an interview.
The study findings of increased fruit and vegetable consumption were not surprising, as the study population may have been more highly motivated to improve their diets, Dr. Thew said. However, she was surprised to see the significant improvement in cooking attitudes and cooking self-efficiency after the intervention. “This tells me that we need to offer more opportunities to educate young adults on how to cook to improve diet outcomes.”
The message for clinicians is to encourage and support young adults to learn cooking skills to promote healthier eating, said Dr. Thew.
“When patients have confidence in their ability to cook, they will explore more food options and consequently improve their diets,” she emphasized. “As clinicians, we need to advocate for nutrition education and promote cooking classes that are accessible to all if we hope to reduce obesity and improve our patients’ diets.”
Limitations
The study findings were limited by several factors including the use of a convenience sample that might not represent all college students, the reliance on self-reports, the inability to account for the impact of demographic factors, and the lack of a control group, the researchers wrote.
“Larger prospective studies are needed,” given the limitations of the pilot design and short study period, Dr. Jay noted.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Jay and Dr. Thew had no financial conflicts to disclose.
Obesity remains a significant risk factor for numerous diseases, and is often a problem in young adults, who often fall back on fast food and other less-healthy meals associated with a lower quality diet, lead author Carol S. O’Neal, PhD, of the University of Louisville (Ky.), said in an interview.
Previous research involving Social Cognitive Theory and goal-setting to promote self-efficacy and behavior changes has shown success in improving eating habits in young adults, but adding video technology for an additional education element has not been well studied, Dr. O’Neal and colleagues wrote in the Journal of Nutrition Education and Behavior.
Methods and results
In the study, 138 college students aged 18-40 years participated in a 15-week pilot intervention course at a large, metropolitan university. The course included lectures on a topic, such as carbohydrates, and included skill-based activities, such as how to read an ingredient list, and discussion of how these skills could improve healthier eating and meet nutrition goals, such as eating more whole grains.
A total of 77 completed the study in person, and 61 participated online. The majority (59%) were college sophomores, 74% were White, and 82% were female.
The course engaged the students in weekly food challenges to apply their knowledge and develop better eating habits and behaviors. The challenges were accompanied by cooking videos related to each week’s topic, such as how to make overnight oats for the healthy carbohydrates/whole grains week.
Students also selected two goals each week, such as choosing whole grain foods to increase fiber consumption, from a list of 10-15 goals, and were required to write weekly reflections to track their progress toward these goals. Goal-setting was based on the strategy of creating goals that are specific, measurable, attainable, realistic, and time-bound (the SMART method).
The main outcomes were increased consumption of fruits and vegetables, improved skills in cooking and healthy eating, and improved attitudes about healthy cooking and eating. The researchers surveyed the students to determine whether these outcomes were met.
Students participating in the study indicated that they met the goal of eating at least five servings of fruits and vegetables per day more often after the course than before, the researchers wrote.
By the course’s end, the students showed significant increases in consumption of fruits and vegetables (P < .001 for both), and in the self-efficacy related to consumption of produce (P = .004); cooking (P = .002;, and using more fruits, vegetables, and seasonings rather than salt in cooking (P = .001).
A review of the students’ written reflections illustrated positive behavior changes such as planning meals before shopping, preparing meals in advance on weekends, taking lunch to school, and using herbs and spices, the researchers noted.
“Self-directed SMART goals set you up for success by making goals specific, measurable, achievable, realistic, and timely,” Dr. O’Neal said in an interview. “The SMART method helps push you further, gives you a sense of direction, and helps you organize and reach your goals,” but self-monitoring and social support are also needed for success. The takeaway message for clinicians is that use of a self-directed goal-setting strategy may be more effective at changing dietary behaviors and promoting self-efficacy than a traditional dietary prescription.
In addition, “this model could be used to address a variety of health outcomes in dietetics, health education and community health programs,” said Dr. O’Neal. “I think the key components of this intervention are teaching SMART goal setting, self-monitoring, and social support of successes. I see time as a main barrier, but this barrier could be reduced for populations who are able to use online learning. Our intervention was successful for in-person and online learning.”
Other areas for future research include evaluation of progress that combines quantitative data and qualitative reflections, she said.
Real-world applications
“Clinicians have limited time to address behavioral counseling, and this study offers an opportunity to reach patients not only in class sessions, but virtually,” M. Susan Jay, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview.
Although the findings from the study are not new, the knowledge can be used by clinicians to help promote behavior change. The study also showcased the use of additional tools, such as weekly food challenges, to impact college students who often consume high-fat diets in nonmedical settings, Dr. Jay said.
For consumers, the real-world implications are exciting, Dr. Jay said.
“People are increasingly attempting to “eat healthy” and despite clinicians wanting to impact healthy eating, limited office visits may not be conducive to behavioral change,” she said.
The current study was important as a way to identify tactics to improve the diet and nutrition of young adults, Margaret Thew, DNP, FNP-BC, medical director of adolescent medicine at the University of Wisconsin–Madison, said in an interview.
The study findings of increased fruit and vegetable consumption were not surprising, as the study population may have been more highly motivated to improve their diets, Dr. Thew said. However, she was surprised to see the significant improvement in cooking attitudes and cooking self-efficiency after the intervention. “This tells me that we need to offer more opportunities to educate young adults on how to cook to improve diet outcomes.”
The message for clinicians is to encourage and support young adults to learn cooking skills to promote healthier eating, said Dr. Thew.
“When patients have confidence in their ability to cook, they will explore more food options and consequently improve their diets,” she emphasized. “As clinicians, we need to advocate for nutrition education and promote cooking classes that are accessible to all if we hope to reduce obesity and improve our patients’ diets.”
Limitations
The study findings were limited by several factors including the use of a convenience sample that might not represent all college students, the reliance on self-reports, the inability to account for the impact of demographic factors, and the lack of a control group, the researchers wrote.
“Larger prospective studies are needed,” given the limitations of the pilot design and short study period, Dr. Jay noted.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Jay and Dr. Thew had no financial conflicts to disclose.
Obesity remains a significant risk factor for numerous diseases, and is often a problem in young adults, who often fall back on fast food and other less-healthy meals associated with a lower quality diet, lead author Carol S. O’Neal, PhD, of the University of Louisville (Ky.), said in an interview.
Previous research involving Social Cognitive Theory and goal-setting to promote self-efficacy and behavior changes has shown success in improving eating habits in young adults, but adding video technology for an additional education element has not been well studied, Dr. O’Neal and colleagues wrote in the Journal of Nutrition Education and Behavior.
Methods and results
In the study, 138 college students aged 18-40 years participated in a 15-week pilot intervention course at a large, metropolitan university. The course included lectures on a topic, such as carbohydrates, and included skill-based activities, such as how to read an ingredient list, and discussion of how these skills could improve healthier eating and meet nutrition goals, such as eating more whole grains.
A total of 77 completed the study in person, and 61 participated online. The majority (59%) were college sophomores, 74% were White, and 82% were female.
The course engaged the students in weekly food challenges to apply their knowledge and develop better eating habits and behaviors. The challenges were accompanied by cooking videos related to each week’s topic, such as how to make overnight oats for the healthy carbohydrates/whole grains week.
Students also selected two goals each week, such as choosing whole grain foods to increase fiber consumption, from a list of 10-15 goals, and were required to write weekly reflections to track their progress toward these goals. Goal-setting was based on the strategy of creating goals that are specific, measurable, attainable, realistic, and time-bound (the SMART method).
The main outcomes were increased consumption of fruits and vegetables, improved skills in cooking and healthy eating, and improved attitudes about healthy cooking and eating. The researchers surveyed the students to determine whether these outcomes were met.
Students participating in the study indicated that they met the goal of eating at least five servings of fruits and vegetables per day more often after the course than before, the researchers wrote.
By the course’s end, the students showed significant increases in consumption of fruits and vegetables (P < .001 for both), and in the self-efficacy related to consumption of produce (P = .004); cooking (P = .002;, and using more fruits, vegetables, and seasonings rather than salt in cooking (P = .001).
A review of the students’ written reflections illustrated positive behavior changes such as planning meals before shopping, preparing meals in advance on weekends, taking lunch to school, and using herbs and spices, the researchers noted.
“Self-directed SMART goals set you up for success by making goals specific, measurable, achievable, realistic, and timely,” Dr. O’Neal said in an interview. “The SMART method helps push you further, gives you a sense of direction, and helps you organize and reach your goals,” but self-monitoring and social support are also needed for success. The takeaway message for clinicians is that use of a self-directed goal-setting strategy may be more effective at changing dietary behaviors and promoting self-efficacy than a traditional dietary prescription.
In addition, “this model could be used to address a variety of health outcomes in dietetics, health education and community health programs,” said Dr. O’Neal. “I think the key components of this intervention are teaching SMART goal setting, self-monitoring, and social support of successes. I see time as a main barrier, but this barrier could be reduced for populations who are able to use online learning. Our intervention was successful for in-person and online learning.”
Other areas for future research include evaluation of progress that combines quantitative data and qualitative reflections, she said.
Real-world applications
“Clinicians have limited time to address behavioral counseling, and this study offers an opportunity to reach patients not only in class sessions, but virtually,” M. Susan Jay, MD, of the Medical College of Wisconsin, Milwaukee, said in an interview.
Although the findings from the study are not new, the knowledge can be used by clinicians to help promote behavior change. The study also showcased the use of additional tools, such as weekly food challenges, to impact college students who often consume high-fat diets in nonmedical settings, Dr. Jay said.
For consumers, the real-world implications are exciting, Dr. Jay said.
“People are increasingly attempting to “eat healthy” and despite clinicians wanting to impact healthy eating, limited office visits may not be conducive to behavioral change,” she said.
The current study was important as a way to identify tactics to improve the diet and nutrition of young adults, Margaret Thew, DNP, FNP-BC, medical director of adolescent medicine at the University of Wisconsin–Madison, said in an interview.
The study findings of increased fruit and vegetable consumption were not surprising, as the study population may have been more highly motivated to improve their diets, Dr. Thew said. However, she was surprised to see the significant improvement in cooking attitudes and cooking self-efficiency after the intervention. “This tells me that we need to offer more opportunities to educate young adults on how to cook to improve diet outcomes.”
The message for clinicians is to encourage and support young adults to learn cooking skills to promote healthier eating, said Dr. Thew.
“When patients have confidence in their ability to cook, they will explore more food options and consequently improve their diets,” she emphasized. “As clinicians, we need to advocate for nutrition education and promote cooking classes that are accessible to all if we hope to reduce obesity and improve our patients’ diets.”
Limitations
The study findings were limited by several factors including the use of a convenience sample that might not represent all college students, the reliance on self-reports, the inability to account for the impact of demographic factors, and the lack of a control group, the researchers wrote.
“Larger prospective studies are needed,” given the limitations of the pilot design and short study period, Dr. Jay noted.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Jay and Dr. Thew had no financial conflicts to disclose.
FROM THE JOURNAL OF NUTRITION EDUCATION AND BEHAVIOR
Federal Health Care Data Trends 2022
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Vaccinations
- Mental Health and Related Disorders
- LGBTQ+ Veterans
- Military Sexual Trauma
- Sleep Disorders
- Respiratory Illnesses
- HIV Care in the VA
- Rheumatologic Diseases
- The Cancer-Obesity Connection
- Skin Health for Active-Duty Personnel
- Contraception
- Chronic Kidney Disease
- Cardiovascular Diseases
- Neurologic Disorders
- Hearing, Vision, and Balance
Federal Practitioner would like to thank the following experts for their review of content and helpful guidance in developing this issue:
Kelvin N.V. Bush, MD, FACC, CCDS; Sonya Borrero, MD, MS; Kenneth L. Cameron, PhD, MPH, ATC, FNATA; Jason DeViva, PhD; Ellen Lockard Edens, MD; Leonard E. Egede, MD, MS; Amy Justice, MD, PhD; Stephanie Knudson, MD; Willis H. Lyford, MD; Sarah O. Meadows, PhD; Tamara Schult, PhD, MPH; Eric L. Singman, MD, PhD; Art Wallace, MD, PhD; Elizabeth Waterhouse, MD, FAAN
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Vaccinations
- Mental Health and Related Disorders
- LGBTQ+ Veterans
- Military Sexual Trauma
- Sleep Disorders
- Respiratory Illnesses
- HIV Care in the VA
- Rheumatologic Diseases
- The Cancer-Obesity Connection
- Skin Health for Active-Duty Personnel
- Contraception
- Chronic Kidney Disease
- Cardiovascular Diseases
- Neurologic Disorders
- Hearing, Vision, and Balance
Federal Practitioner would like to thank the following experts for their review of content and helpful guidance in developing this issue:
Kelvin N.V. Bush, MD, FACC, CCDS; Sonya Borrero, MD, MS; Kenneth L. Cameron, PhD, MPH, ATC, FNATA; Jason DeViva, PhD; Ellen Lockard Edens, MD; Leonard E. Egede, MD, MS; Amy Justice, MD, PhD; Stephanie Knudson, MD; Willis H. Lyford, MD; Sarah O. Meadows, PhD; Tamara Schult, PhD, MPH; Eric L. Singman, MD, PhD; Art Wallace, MD, PhD; Elizabeth Waterhouse, MD, FAAN
Federal Health Care Data Trends (click to view the digital edition) is a special supplement to Federal Practitioner highlighting the latest research and study outcomes related to the health of veteran and active-duty populations.
In this issue:
- Vaccinations
- Mental Health and Related Disorders
- LGBTQ+ Veterans
- Military Sexual Trauma
- Sleep Disorders
- Respiratory Illnesses
- HIV Care in the VA
- Rheumatologic Diseases
- The Cancer-Obesity Connection
- Skin Health for Active-Duty Personnel
- Contraception
- Chronic Kidney Disease
- Cardiovascular Diseases
- Neurologic Disorders
- Hearing, Vision, and Balance
Federal Practitioner would like to thank the following experts for their review of content and helpful guidance in developing this issue:
Kelvin N.V. Bush, MD, FACC, CCDS; Sonya Borrero, MD, MS; Kenneth L. Cameron, PhD, MPH, ATC, FNATA; Jason DeViva, PhD; Ellen Lockard Edens, MD; Leonard E. Egede, MD, MS; Amy Justice, MD, PhD; Stephanie Knudson, MD; Willis H. Lyford, MD; Sarah O. Meadows, PhD; Tamara Schult, PhD, MPH; Eric L. Singman, MD, PhD; Art Wallace, MD, PhD; Elizabeth Waterhouse, MD, FAAN
MERIT: Endoscopic sleeve gastroplasty shows ‘very impressive’ outcomes in randomized clinical trial
In a randomized, controlled trial, endoscopic sleeve gastroplasty (ESG) combined with lifestyle modifications was safe and effective for weight loss among individuals with class I and class II obesity, compared with lifestyle modifications alone.
“Lifestyle modifications and pharmacological therapy have several limitations, and the use of bariatric surgery is hampered by its invasive nature and patient perceptions,” the study authors wrote. ESG is a minimally invasive, reversible, organ-sparing bariatric procedure that might be able to fill those care gaps, they explained.
Previous retrospective studies have suggested that ESG is effective, and a meta-analysis of 1,772 patients found an average total body weight loss of 15.1% at 6 months (95% confidence interval, 14.3%-16.0%) and 16.5% at 12 months (95% CI, 15.2%-17.8%). However, according to the authors of the current study, known as MERIT and published in the Lancet, there have been no randomized clinical trials investigating ESG's efficacy to date.
“[This is] the kind of study that we have been looking forward to. The outcomes were very impressive,” said Danny Issa, MD, who was asked to comment on the study. He is a clinical assistant professor of medicine at the University of California, Los Angeles. meta-analysis of 1,772 patients found an average total body weight loss of 15.1% at 6 months (95% confidence interval, 14.3%-16.0%) and 16.5% at 12 months (95% CI, 15.2%-17.8%).
Understanding the study and its results
Between December 2017 and June 2019, the researchers randomized 209 participants to ESG plus lifestyle modification or lifestyle modification only, which served as the control. The mean age was 47.3 in the ESG group (88% female) and 45.7 in the control group (84% female). The mean body mass index (BMI) was 35.5 kg/m2 in the ESG group and 35.7 among controls.
After 1 year, the intervention group had a mean percentage of excess weight loss (EWL) of 49.2% , compared with 3.2% for the control group (P < .0001). The mean percentage of total body weight lost was 13.6% in the ESG group and 0.8% in the control group (P < .0001). After adjustment for age, sex, type 2 diabetes, hypertension, and baseline BMI, the ESG group had a mean difference of excess weight loss of 44.7% (95% CI, 37.5%-51.9%) and a mean difference of total weight loss of 12.6% (95% CI, 10.7%-14.5%), compared with the control group at 52 weeks. At 52 weeks, 77% of the ESG group had at least a 25% excess weight loss, which was the secondary endpoint, compared with 12% of the control group (P < .0001).
Overall, 80% of the ESG group had an improvement in at least one metabolic comorbidity, while 12% experienced a worsening. Among the control group, 45% had an improvement and 50% worsened. Among 27 patients in the treatment group with diabetes, 93% experienced an improvement in hemoglobin A1c levels, compared with 15% of patients with diabetes in the control group. Similarly among patients with hypertension, 60% in the intervention group had an improvement, compared with 40% of controls. Of those with metabolic syndrome, 83% improved after undergoing surgery, compared with 35% of controls.
At 2 years, 68% of the ESG group who achieved a 25% EWL continued to have at least 25% EWL; 2% in the treatment group had a serious ESG-related adverse event, but there was no mortality or need for intensive care or follow-up surgery.
Aiming for level I evidence
“The results are very encouraging, so I think it’s good news for the field of bariatric endoscopy. I think it’s going to provide more confidence to patients and physicians, and for new trainees who are interested in this field, I think it’s going to inspire them,” said Shailendra Singh, MD, who was asked to comment on the study. Dr. Singh is an associate professor of medicine and director of bariatric medicine at West Virginia University, Morgantown.
The study could also improve insurance coverage of the procedure, said Dr. Singh. “I think this study will help us reach out to the payers and give them the data behind this because they always look for level I evidence. ESG is a relatively new endoscopic procedure; I think this is a step forward in that direction,” he said.
The study underlines the applicability of the procedure to patients who don’t want more invasive surgery, or who can’t tolerate some of the higher efficacy medications that are increasingly available.
It is also just one of various options for obesity treatment, which are increasingly being used in combination, according to Avlin Imaeda, MD. “Just like we see in hypertension, where you progressively add more and more medications, I think we’re going to see obesity treatment go that way too. I see this as adding choice for patients and adding to this potentially multimodal approach,” said Dr. Imaeda, an associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study.
The study authors report various financial relationships, including some with Apollo Endosurgery, which funded this study. Dr. Issa and Dr. Imaeda have no relevant financial disclosures. Dr. Singh is a consultant for Apollo Endosurgery.
This article was updated Aug. 18, 2022.
In a randomized, controlled trial, endoscopic sleeve gastroplasty (ESG) combined with lifestyle modifications was safe and effective for weight loss among individuals with class I and class II obesity, compared with lifestyle modifications alone.
“Lifestyle modifications and pharmacological therapy have several limitations, and the use of bariatric surgery is hampered by its invasive nature and patient perceptions,” the study authors wrote. ESG is a minimally invasive, reversible, organ-sparing bariatric procedure that might be able to fill those care gaps, they explained.
Previous retrospective studies have suggested that ESG is effective, and a meta-analysis of 1,772 patients found an average total body weight loss of 15.1% at 6 months (95% confidence interval, 14.3%-16.0%) and 16.5% at 12 months (95% CI, 15.2%-17.8%). However, according to the authors of the current study, known as MERIT and published in the Lancet, there have been no randomized clinical trials investigating ESG's efficacy to date.
“[This is] the kind of study that we have been looking forward to. The outcomes were very impressive,” said Danny Issa, MD, who was asked to comment on the study. He is a clinical assistant professor of medicine at the University of California, Los Angeles. meta-analysis of 1,772 patients found an average total body weight loss of 15.1% at 6 months (95% confidence interval, 14.3%-16.0%) and 16.5% at 12 months (95% CI, 15.2%-17.8%).
Understanding the study and its results
Between December 2017 and June 2019, the researchers randomized 209 participants to ESG plus lifestyle modification or lifestyle modification only, which served as the control. The mean age was 47.3 in the ESG group (88% female) and 45.7 in the control group (84% female). The mean body mass index (BMI) was 35.5 kg/m2 in the ESG group and 35.7 among controls.
After 1 year, the intervention group had a mean percentage of excess weight loss (EWL) of 49.2% , compared with 3.2% for the control group (P < .0001). The mean percentage of total body weight lost was 13.6% in the ESG group and 0.8% in the control group (P < .0001). After adjustment for age, sex, type 2 diabetes, hypertension, and baseline BMI, the ESG group had a mean difference of excess weight loss of 44.7% (95% CI, 37.5%-51.9%) and a mean difference of total weight loss of 12.6% (95% CI, 10.7%-14.5%), compared with the control group at 52 weeks. At 52 weeks, 77% of the ESG group had at least a 25% excess weight loss, which was the secondary endpoint, compared with 12% of the control group (P < .0001).
Overall, 80% of the ESG group had an improvement in at least one metabolic comorbidity, while 12% experienced a worsening. Among the control group, 45% had an improvement and 50% worsened. Among 27 patients in the treatment group with diabetes, 93% experienced an improvement in hemoglobin A1c levels, compared with 15% of patients with diabetes in the control group. Similarly among patients with hypertension, 60% in the intervention group had an improvement, compared with 40% of controls. Of those with metabolic syndrome, 83% improved after undergoing surgery, compared with 35% of controls.
At 2 years, 68% of the ESG group who achieved a 25% EWL continued to have at least 25% EWL; 2% in the treatment group had a serious ESG-related adverse event, but there was no mortality or need for intensive care or follow-up surgery.
Aiming for level I evidence
“The results are very encouraging, so I think it’s good news for the field of bariatric endoscopy. I think it’s going to provide more confidence to patients and physicians, and for new trainees who are interested in this field, I think it’s going to inspire them,” said Shailendra Singh, MD, who was asked to comment on the study. Dr. Singh is an associate professor of medicine and director of bariatric medicine at West Virginia University, Morgantown.
The study could also improve insurance coverage of the procedure, said Dr. Singh. “I think this study will help us reach out to the payers and give them the data behind this because they always look for level I evidence. ESG is a relatively new endoscopic procedure; I think this is a step forward in that direction,” he said.
The study underlines the applicability of the procedure to patients who don’t want more invasive surgery, or who can’t tolerate some of the higher efficacy medications that are increasingly available.
It is also just one of various options for obesity treatment, which are increasingly being used in combination, according to Avlin Imaeda, MD. “Just like we see in hypertension, where you progressively add more and more medications, I think we’re going to see obesity treatment go that way too. I see this as adding choice for patients and adding to this potentially multimodal approach,” said Dr. Imaeda, an associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study.
The study authors report various financial relationships, including some with Apollo Endosurgery, which funded this study. Dr. Issa and Dr. Imaeda have no relevant financial disclosures. Dr. Singh is a consultant for Apollo Endosurgery.
This article was updated Aug. 18, 2022.
In a randomized, controlled trial, endoscopic sleeve gastroplasty (ESG) combined with lifestyle modifications was safe and effective for weight loss among individuals with class I and class II obesity, compared with lifestyle modifications alone.
“Lifestyle modifications and pharmacological therapy have several limitations, and the use of bariatric surgery is hampered by its invasive nature and patient perceptions,” the study authors wrote. ESG is a minimally invasive, reversible, organ-sparing bariatric procedure that might be able to fill those care gaps, they explained.
Previous retrospective studies have suggested that ESG is effective, and a meta-analysis of 1,772 patients found an average total body weight loss of 15.1% at 6 months (95% confidence interval, 14.3%-16.0%) and 16.5% at 12 months (95% CI, 15.2%-17.8%). However, according to the authors of the current study, known as MERIT and published in the Lancet, there have been no randomized clinical trials investigating ESG's efficacy to date.
“[This is] the kind of study that we have been looking forward to. The outcomes were very impressive,” said Danny Issa, MD, who was asked to comment on the study. He is a clinical assistant professor of medicine at the University of California, Los Angeles. meta-analysis of 1,772 patients found an average total body weight loss of 15.1% at 6 months (95% confidence interval, 14.3%-16.0%) and 16.5% at 12 months (95% CI, 15.2%-17.8%).
Understanding the study and its results
Between December 2017 and June 2019, the researchers randomized 209 participants to ESG plus lifestyle modification or lifestyle modification only, which served as the control. The mean age was 47.3 in the ESG group (88% female) and 45.7 in the control group (84% female). The mean body mass index (BMI) was 35.5 kg/m2 in the ESG group and 35.7 among controls.
After 1 year, the intervention group had a mean percentage of excess weight loss (EWL) of 49.2% , compared with 3.2% for the control group (P < .0001). The mean percentage of total body weight lost was 13.6% in the ESG group and 0.8% in the control group (P < .0001). After adjustment for age, sex, type 2 diabetes, hypertension, and baseline BMI, the ESG group had a mean difference of excess weight loss of 44.7% (95% CI, 37.5%-51.9%) and a mean difference of total weight loss of 12.6% (95% CI, 10.7%-14.5%), compared with the control group at 52 weeks. At 52 weeks, 77% of the ESG group had at least a 25% excess weight loss, which was the secondary endpoint, compared with 12% of the control group (P < .0001).
Overall, 80% of the ESG group had an improvement in at least one metabolic comorbidity, while 12% experienced a worsening. Among the control group, 45% had an improvement and 50% worsened. Among 27 patients in the treatment group with diabetes, 93% experienced an improvement in hemoglobin A1c levels, compared with 15% of patients with diabetes in the control group. Similarly among patients with hypertension, 60% in the intervention group had an improvement, compared with 40% of controls. Of those with metabolic syndrome, 83% improved after undergoing surgery, compared with 35% of controls.
At 2 years, 68% of the ESG group who achieved a 25% EWL continued to have at least 25% EWL; 2% in the treatment group had a serious ESG-related adverse event, but there was no mortality or need for intensive care or follow-up surgery.
Aiming for level I evidence
“The results are very encouraging, so I think it’s good news for the field of bariatric endoscopy. I think it’s going to provide more confidence to patients and physicians, and for new trainees who are interested in this field, I think it’s going to inspire them,” said Shailendra Singh, MD, who was asked to comment on the study. Dr. Singh is an associate professor of medicine and director of bariatric medicine at West Virginia University, Morgantown.
The study could also improve insurance coverage of the procedure, said Dr. Singh. “I think this study will help us reach out to the payers and give them the data behind this because they always look for level I evidence. ESG is a relatively new endoscopic procedure; I think this is a step forward in that direction,” he said.
The study underlines the applicability of the procedure to patients who don’t want more invasive surgery, or who can’t tolerate some of the higher efficacy medications that are increasingly available.
It is also just one of various options for obesity treatment, which are increasingly being used in combination, according to Avlin Imaeda, MD. “Just like we see in hypertension, where you progressively add more and more medications, I think we’re going to see obesity treatment go that way too. I see this as adding choice for patients and adding to this potentially multimodal approach,” said Dr. Imaeda, an associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study.
The study authors report various financial relationships, including some with Apollo Endosurgery, which funded this study. Dr. Issa and Dr. Imaeda have no relevant financial disclosures. Dr. Singh is a consultant for Apollo Endosurgery.
This article was updated Aug. 18, 2022.
FROM THE LANCET
Early time-restricted eating ups weight loss, but jury still out
, new findings suggest.
Previous studies have produced mixed results regarding the weight-loss potential for intermittent fasting, the practice of alternating eating with extended fasting, and the “time-restricted eating” format, where eating is restricted to a specific, often 10-hour, time window during the day.
In a new randomized clinical trial of 90 people with obesity in which that time window was 7 AM through 3 PM, so 8 hours long, researchers report that “eTRE was more effective for losing weight and lowering diastolic blood pressure than was eating over a period of 12 or more hours at 14 weeks. The eTRE intervention may therefore be an effective treatment for both obesity and hypertension.” The study, by Humaira Jamshed, PhD, of the department of nutrition sciences, University of Alabama at Birmingham, and colleagues, was published in JAMA Internal Medicine.
In an accompanying invited commentary, Shalender Bhasin, MBBS, points out that the study findings differ from those of a previous trial published in April of 139 adults conducted in China, which did not find a significant weight loss benefit with TRE versus ad lib eating.
“The scientific premise and the preclinical data of the effects of TRE are promising, but the inconsistency among studies renders it difficult to draw strong inferences from these well-conducted but relatively small trials,” notes Dr. Bhasin, of Harvard Medical School, Boston.
Need for larger and longer trials of TRE
Dr. Bhasin says – and the study authors also acknowledge – that much larger randomized clinical trials of longer duration are needed “to comprehensively evaluate the hypothesized benefits and risks of long-term TRE of calorically restricted diets in adults.”
Commenting on the study for the U.K. Science Media Centre, Simon Steenson, PhD, nutrition scientist, British Nutrition Foundation, said “one of the strengths of this new study is the trial design and the number of people who were recruited compared to many of the previous trials to date.”
However, Dr. Steenson also pointed to the prior Chinese research as evidence that the inconsistencies across studies highlight the need for larger and longer trials, with cardiovascular as well as weight-loss endpoints.
Still, Dr. Steenson said, “For individuals who may find that this pattern of eating fits better with their lifestyle and preferences, time-restricted feeding is one option for reducing overall calorie intake that might be a suitable approach for some. Ultimately, it is about finding the best approach to moderate calorie intake that works for each person, as successful and sustained weight loss is about ensuring the diet is feasible to follow in the long-term.”
Differences in weight loss, diastolic BP, but not all measures
The study population included 90 adults seen at the Weight Loss Medicine clinic at the University of Alabama at Birmingham between August 2018 and December 2019. Participants had a body mass index of 30-60 kg/m2, and none had diabetes.
They were randomized to eTRE with the 7 AM to PM eating window or a control schedule with eating across 12 hours or more, mimicking U.S. median mealtimes, at least 6 days a week. All participants received 30-minute weight-loss counseling sessions at baseline and at weeks 2, 6, and 10 and were advised to follow a diet of 500 kcal/day below their resting energy expenditure and exercise 75-150 minutes per week.
The eTRE group adhered with their schedule a mean of 6 days per week, lower than the 6.3 days among controls (P = .03), and adherence declined by about 0.4 days per week in the eTRE group over the 14 weeks (P = .001).
At 14 weeks, both the eTRE group and controls had lost clinically meaningful amounts of weight, at –6.3 kg and –4.0 kg, respectively, but the –2.3 kg difference was significant (P = .002).
However, there was no difference in absolute fat loss (P = .09) or ratio of fat loss to weight loss (P = .43). There were also no significant differences in changes in other body composition parameters, including visceral fat and waist circumference.
Diastolic blood pressure was lowered by an additional 4 mmHg in the eTRE group, compared with controls at 14 weeks (P = .04), but there were no significant differences in systolic blood pressure, heart rate, glucose, A1c levels, insulin levels, measures of insulin resistance, or plasma lipids.
There were no differences between the two groups in self-reported physical activity, energy intake, or dietary macronutrient composition either. However, weight-loss modeling in 77 participants with at least two weight measurements indicated that the eTRE group reduced their intake by about 214 kcal/day, compared with controls (P = .04).
Those in the eTRE group also showed greater improvements in measures of mood disturbance, vigor-activity, fatigue-inertia, and depression-dejection. Other mood and sleep endpoints were similar between groups.
In a secondary analysis of just the 59 participants who completed the study, eTRE was also more effective at reducing body fat (P = .047) and trunk fat (P = .03).
About 41% of the eTRE completers planned to continue the practice after the study concluded.
The study was supported by grants from the National Center for Advancing Translational Sciences of the National Institutes of Health and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Jamshed has reported no relevant financial relationships. Dr. Bhasin has reported receiving grants to his institution for research on which Dr. Bhasin is the principal investigator from AbbVie and MIB, receiving personal fees from OPKO and Aditum and holding equity interest in FPT and XYOne. Dr. Steenson has declared funding in support of the British Nutrition Foundation that comes from a range of sources.
A version of this article first appeared on Medscape.com.
, new findings suggest.
Previous studies have produced mixed results regarding the weight-loss potential for intermittent fasting, the practice of alternating eating with extended fasting, and the “time-restricted eating” format, where eating is restricted to a specific, often 10-hour, time window during the day.
In a new randomized clinical trial of 90 people with obesity in which that time window was 7 AM through 3 PM, so 8 hours long, researchers report that “eTRE was more effective for losing weight and lowering diastolic blood pressure than was eating over a period of 12 or more hours at 14 weeks. The eTRE intervention may therefore be an effective treatment for both obesity and hypertension.” The study, by Humaira Jamshed, PhD, of the department of nutrition sciences, University of Alabama at Birmingham, and colleagues, was published in JAMA Internal Medicine.
In an accompanying invited commentary, Shalender Bhasin, MBBS, points out that the study findings differ from those of a previous trial published in April of 139 adults conducted in China, which did not find a significant weight loss benefit with TRE versus ad lib eating.
“The scientific premise and the preclinical data of the effects of TRE are promising, but the inconsistency among studies renders it difficult to draw strong inferences from these well-conducted but relatively small trials,” notes Dr. Bhasin, of Harvard Medical School, Boston.
Need for larger and longer trials of TRE
Dr. Bhasin says – and the study authors also acknowledge – that much larger randomized clinical trials of longer duration are needed “to comprehensively evaluate the hypothesized benefits and risks of long-term TRE of calorically restricted diets in adults.”
Commenting on the study for the U.K. Science Media Centre, Simon Steenson, PhD, nutrition scientist, British Nutrition Foundation, said “one of the strengths of this new study is the trial design and the number of people who were recruited compared to many of the previous trials to date.”
However, Dr. Steenson also pointed to the prior Chinese research as evidence that the inconsistencies across studies highlight the need for larger and longer trials, with cardiovascular as well as weight-loss endpoints.
Still, Dr. Steenson said, “For individuals who may find that this pattern of eating fits better with their lifestyle and preferences, time-restricted feeding is one option for reducing overall calorie intake that might be a suitable approach for some. Ultimately, it is about finding the best approach to moderate calorie intake that works for each person, as successful and sustained weight loss is about ensuring the diet is feasible to follow in the long-term.”
Differences in weight loss, diastolic BP, but not all measures
The study population included 90 adults seen at the Weight Loss Medicine clinic at the University of Alabama at Birmingham between August 2018 and December 2019. Participants had a body mass index of 30-60 kg/m2, and none had diabetes.
They were randomized to eTRE with the 7 AM to PM eating window or a control schedule with eating across 12 hours or more, mimicking U.S. median mealtimes, at least 6 days a week. All participants received 30-minute weight-loss counseling sessions at baseline and at weeks 2, 6, and 10 and were advised to follow a diet of 500 kcal/day below their resting energy expenditure and exercise 75-150 minutes per week.
The eTRE group adhered with their schedule a mean of 6 days per week, lower than the 6.3 days among controls (P = .03), and adherence declined by about 0.4 days per week in the eTRE group over the 14 weeks (P = .001).
At 14 weeks, both the eTRE group and controls had lost clinically meaningful amounts of weight, at –6.3 kg and –4.0 kg, respectively, but the –2.3 kg difference was significant (P = .002).
However, there was no difference in absolute fat loss (P = .09) or ratio of fat loss to weight loss (P = .43). There were also no significant differences in changes in other body composition parameters, including visceral fat and waist circumference.
Diastolic blood pressure was lowered by an additional 4 mmHg in the eTRE group, compared with controls at 14 weeks (P = .04), but there were no significant differences in systolic blood pressure, heart rate, glucose, A1c levels, insulin levels, measures of insulin resistance, or plasma lipids.
There were no differences between the two groups in self-reported physical activity, energy intake, or dietary macronutrient composition either. However, weight-loss modeling in 77 participants with at least two weight measurements indicated that the eTRE group reduced their intake by about 214 kcal/day, compared with controls (P = .04).
Those in the eTRE group also showed greater improvements in measures of mood disturbance, vigor-activity, fatigue-inertia, and depression-dejection. Other mood and sleep endpoints were similar between groups.
In a secondary analysis of just the 59 participants who completed the study, eTRE was also more effective at reducing body fat (P = .047) and trunk fat (P = .03).
About 41% of the eTRE completers planned to continue the practice after the study concluded.
The study was supported by grants from the National Center for Advancing Translational Sciences of the National Institutes of Health and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Jamshed has reported no relevant financial relationships. Dr. Bhasin has reported receiving grants to his institution for research on which Dr. Bhasin is the principal investigator from AbbVie and MIB, receiving personal fees from OPKO and Aditum and holding equity interest in FPT and XYOne. Dr. Steenson has declared funding in support of the British Nutrition Foundation that comes from a range of sources.
A version of this article first appeared on Medscape.com.
, new findings suggest.
Previous studies have produced mixed results regarding the weight-loss potential for intermittent fasting, the practice of alternating eating with extended fasting, and the “time-restricted eating” format, where eating is restricted to a specific, often 10-hour, time window during the day.
In a new randomized clinical trial of 90 people with obesity in which that time window was 7 AM through 3 PM, so 8 hours long, researchers report that “eTRE was more effective for losing weight and lowering diastolic blood pressure than was eating over a period of 12 or more hours at 14 weeks. The eTRE intervention may therefore be an effective treatment for both obesity and hypertension.” The study, by Humaira Jamshed, PhD, of the department of nutrition sciences, University of Alabama at Birmingham, and colleagues, was published in JAMA Internal Medicine.
In an accompanying invited commentary, Shalender Bhasin, MBBS, points out that the study findings differ from those of a previous trial published in April of 139 adults conducted in China, which did not find a significant weight loss benefit with TRE versus ad lib eating.
“The scientific premise and the preclinical data of the effects of TRE are promising, but the inconsistency among studies renders it difficult to draw strong inferences from these well-conducted but relatively small trials,” notes Dr. Bhasin, of Harvard Medical School, Boston.
Need for larger and longer trials of TRE
Dr. Bhasin says – and the study authors also acknowledge – that much larger randomized clinical trials of longer duration are needed “to comprehensively evaluate the hypothesized benefits and risks of long-term TRE of calorically restricted diets in adults.”
Commenting on the study for the U.K. Science Media Centre, Simon Steenson, PhD, nutrition scientist, British Nutrition Foundation, said “one of the strengths of this new study is the trial design and the number of people who were recruited compared to many of the previous trials to date.”
However, Dr. Steenson also pointed to the prior Chinese research as evidence that the inconsistencies across studies highlight the need for larger and longer trials, with cardiovascular as well as weight-loss endpoints.
Still, Dr. Steenson said, “For individuals who may find that this pattern of eating fits better with their lifestyle and preferences, time-restricted feeding is one option for reducing overall calorie intake that might be a suitable approach for some. Ultimately, it is about finding the best approach to moderate calorie intake that works for each person, as successful and sustained weight loss is about ensuring the diet is feasible to follow in the long-term.”
Differences in weight loss, diastolic BP, but not all measures
The study population included 90 adults seen at the Weight Loss Medicine clinic at the University of Alabama at Birmingham between August 2018 and December 2019. Participants had a body mass index of 30-60 kg/m2, and none had diabetes.
They were randomized to eTRE with the 7 AM to PM eating window or a control schedule with eating across 12 hours or more, mimicking U.S. median mealtimes, at least 6 days a week. All participants received 30-minute weight-loss counseling sessions at baseline and at weeks 2, 6, and 10 and were advised to follow a diet of 500 kcal/day below their resting energy expenditure and exercise 75-150 minutes per week.
The eTRE group adhered with their schedule a mean of 6 days per week, lower than the 6.3 days among controls (P = .03), and adherence declined by about 0.4 days per week in the eTRE group over the 14 weeks (P = .001).
At 14 weeks, both the eTRE group and controls had lost clinically meaningful amounts of weight, at –6.3 kg and –4.0 kg, respectively, but the –2.3 kg difference was significant (P = .002).
However, there was no difference in absolute fat loss (P = .09) or ratio of fat loss to weight loss (P = .43). There were also no significant differences in changes in other body composition parameters, including visceral fat and waist circumference.
Diastolic blood pressure was lowered by an additional 4 mmHg in the eTRE group, compared with controls at 14 weeks (P = .04), but there were no significant differences in systolic blood pressure, heart rate, glucose, A1c levels, insulin levels, measures of insulin resistance, or plasma lipids.
There were no differences between the two groups in self-reported physical activity, energy intake, or dietary macronutrient composition either. However, weight-loss modeling in 77 participants with at least two weight measurements indicated that the eTRE group reduced their intake by about 214 kcal/day, compared with controls (P = .04).
Those in the eTRE group also showed greater improvements in measures of mood disturbance, vigor-activity, fatigue-inertia, and depression-dejection. Other mood and sleep endpoints were similar between groups.
In a secondary analysis of just the 59 participants who completed the study, eTRE was also more effective at reducing body fat (P = .047) and trunk fat (P = .03).
About 41% of the eTRE completers planned to continue the practice after the study concluded.
The study was supported by grants from the National Center for Advancing Translational Sciences of the National Institutes of Health and the National Institute of Diabetes and Digestive and Kidney Diseases. Dr. Jamshed has reported no relevant financial relationships. Dr. Bhasin has reported receiving grants to his institution for research on which Dr. Bhasin is the principal investigator from AbbVie and MIB, receiving personal fees from OPKO and Aditum and holding equity interest in FPT and XYOne. Dr. Steenson has declared funding in support of the British Nutrition Foundation that comes from a range of sources.
A version of this article first appeared on Medscape.com.
FROM JAMA INTERNAL MEDICINE
‘Staggering’ CVD rise projected in U.S., especially in minorities
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
A new analysis projects steep increases by 2060 in the prevalence of cardiovascular (CV) risk factors and disease that will disproportionately affect non-White populations who have limited access to health care.
The study by Reza Mohebi, MD, Massachusetts General Hospital and Harvard Medical School, both in Boston, and colleagues was published in the Journal of the American College of Cardiology.
“Even though several assumptions underlie these projections, the importance of this work cannot be overestimated,” Andreas P. Kalogeropoulos, MD, MPH, PhD, and Javed Butler, MD, MPH, MBA, wrote in an accompanying editorial. “The absolute numbers are staggering.”
From 2025 to 2060, the number of people with any one of four CV risk factors – type 2 diabetes, hypertension, dyslipidemia, and obesity – is projected to increase by 15.4 million, to 34.7 million.
And the number of people with of any one of four CV disease types – ischemic heart disease, heart failure, MI, and stroke – is projected to increase by 3.2 million, to 6.8 million.
Although the model predicts that the prevalence of CV risk factors will gradually decrease among White Americans, the highest prevalence of CV risk factors will be among the White population because of its overall size.
Conversely, the projected prevalence of CV risk factors is expected to increase in Black, Hispanic, Asian, and other race/ethnicity populations.
In parallel, the prevalence of CV disease is projected to decrease in the White population and increase among all other race/ethnicities, particularly in the Black and Hispanic populations.
“Our results project a worrisome increase with a particularly ominous increase in risk factors and disease in our most vulnerable patients, including Blacks and Hispanics,” senior author James L. Januzzi Jr., MD, summarized in a video issued by the society.
“The steep rise in CV risk factors and disease reflects the generally higher prevalence in populations projected to increase in the United States, owing to immigration and growth, including Black or Hispanic individuals,” Dr. Januzzi, also from Massachusetts General and Harvard, said in an interview.
“The disproportionate size of the risk is expected in a sense, as minority populations are disproportionately disadvantaged with respect to their health care,” he said. “But whether it is expected or not, the increase in projected prevalence is, nonetheless, concerning and a call to action.”
This study identifies “areas of opportunity for change in the U.S. health care system,” he continued. “Business as usual will result in us encountering a huge number of individuals with CV risk factors and diseases.”
The results from the current analysis assume there will be no modification in health care policies or changes in access to care for at-risk populations, Dr. Mohebi and colleagues noted.
To “stem the rising tide of CV disease in at-risk individuals,” would require strategies such as “emphasis on education regarding CV risk factors, improving access to quality healthcare, and facilitating lower-cost access to effective therapies for treatment of CV risk factors,” according to the researchers.
“Such advances need to be applied in a more equitable way throughout the United States, however,” they cautioned.
Census plus NHANES data
The researchers used 2020 U.S. census data and projected growth and 2013-2018 U.S. National Health and Nutrition Survey data to estimate the number of people with CV risk factors and CV disease from 2025 to 2060.
The estimates are based on a growing population and a fixed frequency.
The projected changes in CV risk factors and disease over time were similar in men and women.
The researchers acknowledge that study limitations include the assumption that the prevalence patterns for CV risk factors and disease will be stable.
“To the extent the frequency of risk factors and disease are not likely to remain static, that assumption may reduce the accuracy of the projections,” Dr. Januzzi said. “However, we would point out that the goals of our analysis were to set general trends, and not to seek to project exact figures.”
Also, they did not take into account the effect of COVID-19. CV diseases were also based on self-report and CV risk factors could have been underestimated in minority populations that do not access health care.
Changing demographic landscape
It is “striking” that the numbers of non-White individuals with CV risk factors is projected to surpass the number of White individuals over time, and the number of non-White individuals with CV disease will be almost as many as White individuals by the year 2060, the editorialists noted.
“From a policy perspective, this means that unless appropriate, targeted action is taken, disparities in the burden of cardiovascular disease are only going to be exacerbated over time,” wrote Dr. Kalogeropoulos, from Stony Brook (N.Y.) University, and Dr. Butler, from Baylor College of Medicine, Dallas.
“On the positive side,” they continued, “the absolute increase in the percent prevalence of cardiovascular risk factors and conditions is projected to lie within a manageable range,” assuming that specific prevention policies are implemented.
“This is an opportunity for professional societies, including the cardiovascular care community, to re-evaluate priorities and strategies, for both training and practice, to best match the growing demands of a changing demographic landscape in the United States,” Dr. Kalogeropoulos and Dr. Butler concluded.
Dr. Mohebi is supported by the Barry Fellowship. Dr. Januzzi is supported by the Hutter Family Professorship; is a Trustee of the American College of Cardiology; is a board member of Imbria Pharmaceuticals; has received grant support from Abbott Diagnostics, Applied Therapeutics, Innolife, and Novartis; has received consulting income from Abbott Diagnostics, Boehringer Ingelheim, Janssen, Novartis, and Roche Diagnostics; and participates in clinical endpoint committees/data safety monitoring boards for AbbVie, Siemens, Takeda, and Vifor. Dr. Kalogeropoulos has received research funding from the National Heart, Lung, and Blood Institute; the American Heart Association; and the Centers for Disease Control and Prevention. Dr. Butler has been a consultant for numerous pharmaceutical companies.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF AMERICAN COLLEGE OF CARDIOLOGY
Many saw ‘meaningful’ weight loss from 12-week online program
developed by researchers at Brown University.
Primary care doctors offered the free obesity treatment program during routine care. Each week, people reported weight changes and activity and calorie consumption; attended online lessons; and received personalized feedback.
The 464 people who took part for at least 1 week lost an average of 5% of their body weight. And those who followed the plan all 12 weeks lost an average of 7%.
The researchers point out this short-term weight loss was achieved without any face-to-face counseling, which can limit weight management in busy primary care settings.
“Obesity is a highly stigmatized condition,” says lead investigator J. Graham Thomas, PhD.
People take part in the Rx Weight Loss program in the privacy of their own homes. He says this not only makes it more convenient but could be an advantage for people who feel uncomfortable managing their weight around others.
Ideally, health care providers could offer the online program as an opportunity to patients “as opposed to something punitive,” says Dr. Thomas, a researcher at the Weight Control and Diabetes Research Center at Miriam Hospital in Providence, R.I.
The study was published online in the journal Obesity.
In three previous controlled clinical trials led by the same research team, the weight-loss program was linked to average weight losses of 4.2% to 5.8%. In the current study, the researchers were not directly involved, and Dr. Thomas says he was encouraged that the doctor-led initiative led to similar results.
About 11 pounds lost
Patients were offered the program during routine care by doctors in the Rhode Island Primary Care Physicians Corporation, which includes 100 doctors at 60 sites. To be eligible, people had to be 18-75 years old, have Internet access, be fluent in English, and have a body mass index (BMI) of 25 kg/m2 or greater.
The average age of the people in the study was 53, 70% were women, and the average BMI was 36.2.
A BMI of 25 or above means you are overweight, while those with a BMI of 30 or higher are considered obese.
The average 5.1% decrease in body weight at 12 weeks translated to just more than 11 pounds of average weight loss.
‘Very encouraging’
The results of the study are “very encouraging,” says Gareth R. Dutton, PhD, who was not affiliated with the study.
Previous strategies had limits, he says.
“Fully automated interventions that have no staff contact with participants often achieve modest weight loss,” says Dr. Dutton, a professor of medicine and investigator in the Nutrition Obesity Research Center at the University of Alabama at Birmingham.
Weight-loss programs recommended by primary care doctors have often performed even worse, he says.
“Weight-loss interventions delivered through primary care are challenging because of many barriers, including limited resources and time,” says Dr. Dutton, who is also lead investigator of a study that aims to enroll 400 primary care patients to compare daily self-weighing with standard care.
Letting doctors and their staff refer patients to an evidence-based weight-loss program has great potential, he says.
Looking to improve uptake
The Rx Weight Loss program was offered to 1,721 primary care patients overall.
When asked why only 26% of people offered the program agreed to participate, Dr. Thomas replied, “No matter how good the program is, it’s just never going to be the right time for a lot of people to add this to their lives, particularly given the last couple of years where folks are experiencing a lot of challenges and a lot of stressors.”
“Even though it’s an online program, addressing obesity always involves making substantial changes to eating and activity patterns,” he said.
Future steps
The investigators plan to look into ways to get more people to take part in the program.
It is not yet available for widespread use by others, but that’s the goal. Dr. Thomas said they learned ways during the study to make the fully automated, online program easier for others to adopt.
Measuring any effect on weight loss at 1 year is the primary aim of the study. “I think we expect to find something similar to what we see in previous studies, which is that a certain amount of weight regain will be the norm” at 1 year, Dr. Thomas said.
“But a certain amount of weight loss and associated health benefits will persist, making it worthwhile even if, on average, some gradual regain occurs.”A version of this article first appeared on WebMD.com.
developed by researchers at Brown University.
Primary care doctors offered the free obesity treatment program during routine care. Each week, people reported weight changes and activity and calorie consumption; attended online lessons; and received personalized feedback.
The 464 people who took part for at least 1 week lost an average of 5% of their body weight. And those who followed the plan all 12 weeks lost an average of 7%.
The researchers point out this short-term weight loss was achieved without any face-to-face counseling, which can limit weight management in busy primary care settings.
“Obesity is a highly stigmatized condition,” says lead investigator J. Graham Thomas, PhD.
People take part in the Rx Weight Loss program in the privacy of their own homes. He says this not only makes it more convenient but could be an advantage for people who feel uncomfortable managing their weight around others.
Ideally, health care providers could offer the online program as an opportunity to patients “as opposed to something punitive,” says Dr. Thomas, a researcher at the Weight Control and Diabetes Research Center at Miriam Hospital in Providence, R.I.
The study was published online in the journal Obesity.
In three previous controlled clinical trials led by the same research team, the weight-loss program was linked to average weight losses of 4.2% to 5.8%. In the current study, the researchers were not directly involved, and Dr. Thomas says he was encouraged that the doctor-led initiative led to similar results.
About 11 pounds lost
Patients were offered the program during routine care by doctors in the Rhode Island Primary Care Physicians Corporation, which includes 100 doctors at 60 sites. To be eligible, people had to be 18-75 years old, have Internet access, be fluent in English, and have a body mass index (BMI) of 25 kg/m2 or greater.
The average age of the people in the study was 53, 70% were women, and the average BMI was 36.2.
A BMI of 25 or above means you are overweight, while those with a BMI of 30 or higher are considered obese.
The average 5.1% decrease in body weight at 12 weeks translated to just more than 11 pounds of average weight loss.
‘Very encouraging’
The results of the study are “very encouraging,” says Gareth R. Dutton, PhD, who was not affiliated with the study.
Previous strategies had limits, he says.
“Fully automated interventions that have no staff contact with participants often achieve modest weight loss,” says Dr. Dutton, a professor of medicine and investigator in the Nutrition Obesity Research Center at the University of Alabama at Birmingham.
Weight-loss programs recommended by primary care doctors have often performed even worse, he says.
“Weight-loss interventions delivered through primary care are challenging because of many barriers, including limited resources and time,” says Dr. Dutton, who is also lead investigator of a study that aims to enroll 400 primary care patients to compare daily self-weighing with standard care.
Letting doctors and their staff refer patients to an evidence-based weight-loss program has great potential, he says.
Looking to improve uptake
The Rx Weight Loss program was offered to 1,721 primary care patients overall.
When asked why only 26% of people offered the program agreed to participate, Dr. Thomas replied, “No matter how good the program is, it’s just never going to be the right time for a lot of people to add this to their lives, particularly given the last couple of years where folks are experiencing a lot of challenges and a lot of stressors.”
“Even though it’s an online program, addressing obesity always involves making substantial changes to eating and activity patterns,” he said.
Future steps
The investigators plan to look into ways to get more people to take part in the program.
It is not yet available for widespread use by others, but that’s the goal. Dr. Thomas said they learned ways during the study to make the fully automated, online program easier for others to adopt.
Measuring any effect on weight loss at 1 year is the primary aim of the study. “I think we expect to find something similar to what we see in previous studies, which is that a certain amount of weight regain will be the norm” at 1 year, Dr. Thomas said.
“But a certain amount of weight loss and associated health benefits will persist, making it worthwhile even if, on average, some gradual regain occurs.”A version of this article first appeared on WebMD.com.
developed by researchers at Brown University.
Primary care doctors offered the free obesity treatment program during routine care. Each week, people reported weight changes and activity and calorie consumption; attended online lessons; and received personalized feedback.
The 464 people who took part for at least 1 week lost an average of 5% of their body weight. And those who followed the plan all 12 weeks lost an average of 7%.
The researchers point out this short-term weight loss was achieved without any face-to-face counseling, which can limit weight management in busy primary care settings.
“Obesity is a highly stigmatized condition,” says lead investigator J. Graham Thomas, PhD.
People take part in the Rx Weight Loss program in the privacy of their own homes. He says this not only makes it more convenient but could be an advantage for people who feel uncomfortable managing their weight around others.
Ideally, health care providers could offer the online program as an opportunity to patients “as opposed to something punitive,” says Dr. Thomas, a researcher at the Weight Control and Diabetes Research Center at Miriam Hospital in Providence, R.I.
The study was published online in the journal Obesity.
In three previous controlled clinical trials led by the same research team, the weight-loss program was linked to average weight losses of 4.2% to 5.8%. In the current study, the researchers were not directly involved, and Dr. Thomas says he was encouraged that the doctor-led initiative led to similar results.
About 11 pounds lost
Patients were offered the program during routine care by doctors in the Rhode Island Primary Care Physicians Corporation, which includes 100 doctors at 60 sites. To be eligible, people had to be 18-75 years old, have Internet access, be fluent in English, and have a body mass index (BMI) of 25 kg/m2 or greater.
The average age of the people in the study was 53, 70% were women, and the average BMI was 36.2.
A BMI of 25 or above means you are overweight, while those with a BMI of 30 or higher are considered obese.
The average 5.1% decrease in body weight at 12 weeks translated to just more than 11 pounds of average weight loss.
‘Very encouraging’
The results of the study are “very encouraging,” says Gareth R. Dutton, PhD, who was not affiliated with the study.
Previous strategies had limits, he says.
“Fully automated interventions that have no staff contact with participants often achieve modest weight loss,” says Dr. Dutton, a professor of medicine and investigator in the Nutrition Obesity Research Center at the University of Alabama at Birmingham.
Weight-loss programs recommended by primary care doctors have often performed even worse, he says.
“Weight-loss interventions delivered through primary care are challenging because of many barriers, including limited resources and time,” says Dr. Dutton, who is also lead investigator of a study that aims to enroll 400 primary care patients to compare daily self-weighing with standard care.
Letting doctors and their staff refer patients to an evidence-based weight-loss program has great potential, he says.
Looking to improve uptake
The Rx Weight Loss program was offered to 1,721 primary care patients overall.
When asked why only 26% of people offered the program agreed to participate, Dr. Thomas replied, “No matter how good the program is, it’s just never going to be the right time for a lot of people to add this to their lives, particularly given the last couple of years where folks are experiencing a lot of challenges and a lot of stressors.”
“Even though it’s an online program, addressing obesity always involves making substantial changes to eating and activity patterns,” he said.
Future steps
The investigators plan to look into ways to get more people to take part in the program.
It is not yet available for widespread use by others, but that’s the goal. Dr. Thomas said they learned ways during the study to make the fully automated, online program easier for others to adopt.
Measuring any effect on weight loss at 1 year is the primary aim of the study. “I think we expect to find something similar to what we see in previous studies, which is that a certain amount of weight regain will be the norm” at 1 year, Dr. Thomas said.
“But a certain amount of weight loss and associated health benefits will persist, making it worthwhile even if, on average, some gradual regain occurs.”A version of this article first appeared on WebMD.com.
FROM OBESITY







