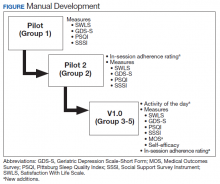
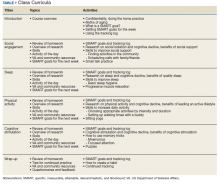
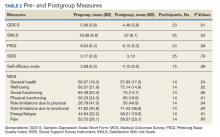
User login
Still no clear answer on intranasal insulin for MCI and Alzheimer’s disease
The randomized trial of nearly 300 patients showed that, although one insulin administration device produced marked benefit in terms of change in mean score on the Alzheimer Disease Assessment Scale–Cognitive Subscale 12 (ADAS-cog-12) over 12 months, reliability was inconsistent. A second device, used on the majority of patients in the study’s intention-to-treat population, showed no difference in these measures between patients who did and those who did not receive intranasal insulin.
“The primary analysis of the study showed no benefit of intranasal insulin on any measures of cognition or cerebrospinal fluid Alzheimer’s disease biomarkers when using the new device,” said principal investigator Suzanne Craft, PhD.
“But when we looked at our planned secondary analysis with the original device – which has been successful in previous studies – we saw quite a different picture,” added Dr. Craft, director of the Alzheimer’s Disease Research Center at Wake Forest University, Winston-Salem, N.C.
“We found a pronounced benefit with that device, such that after 18 months of administration, participants who had been receiving insulin from the beginning of the study had a large and clinically significant advantage in the primary outcome measure.”
Dr. Craft described the findings as complex. “The primary results were negative,” she added. “But the secondary results replicated those of several earlier studies when we used the same device that was used in those.”
The study was published online June 22 in JAMA Neurology.
Important for brain function
Insulin has been shown to play several important roles in brain function. The hormone is associated with a variety of cognitive functions, including memory. Through its association with vasoreactivity, lipid metabolism, and inflammation, insulin also plays an important role in vascular function.
“In the normal brain in healthy individuals, insulin is very important for synaptic function and viability. Insulin also promotes dendritic growth and facilitates synaptic health. Through this role, it plays an important part in memory,” said Dr. Craft. Given these connections, it is not surprising that reduced insulin levels or activity in brain and cerebrospinal fluid have been documented in some, but not all, studies of Alzheimer’s disease. Markers of insulin resistance also have been detected in both neuronally derived exosomes and brain tissue from adults with Alzheimer’s disease.
In light of the several important roles that insulin plays in the brain – coupled with the evidence connecting dysregulation of brain insulin and AD pathology – restoring brain insulin function may offer therapeutic benefit for adults suffering either Alzheimer’s disease or MCI. “There are a number of ways to do this,” said Dr. Craft. “But one of the approaches that we’ve focused on is providing insulin directly to the brain through intranasal administration. “By doing this, you circumvent potential issues if you administered insulin systemically.”
Previous research has shown that through this mode of administration, insulin can bypass the blood-brain barrier and reach the brain through olfactory and trigeminal perivascular channels, with little effect on peripheral insulin or blood glucose levels.
As previously reported, an earlier pilot study, also conducted by Dr. Craft and her team, showed that 4 months of daily intranasal administration of 20 IU or 40 IU of insulin preserved cognitive performance in individuals with Alzheimer’s disease or MCI.
Deeper dive
In the current investigation, the researchers wanted to broaden these findings in a larger, longer, randomized double-blinded clinical trial. The investigators assessed the efficacy of intranasal insulin on cognition, function, and biomarkers of Alzheimer’s disease, as well as the safety and feasibility of the delivery method. The multicenter trial was conducted from 2014 to 2018 and included 27 sites.
Study participants were between the ages of 55 and 85 years and had been diagnosed with amnestic MCI or Alzheimer’s disease on the basis of National Institute on Aging–Alzheimer Association criteria, a score of 20 or higher on the Mini–Mental State Examination, a clinical dementia rating of 0.5 or 1.0, or a delayed logical memory score within a specified range.
In total, 289 participants were randomly assigned to receive 40 IU of insulin or placebo for 12 months, followed by a 6-month open-label extension phase. The first 49 participants (32 men; mean age, 71.9 years) underwent insulin administration with the same device the investigators used in previous trials.
Of these, 45 completed the blinded phase, and 42 completed the open-label extension. When this device, which uses an electronic nebulizer-like delivery system, proved unreliable, the researchers switched to a second device, which uses a liquid hydrofluoroalkane propellant to deliver a metered dose of insulin through a nose tip without electronic assistance. Device 2 was used for the remaining 240 participants (123 men; mean age, 70.8 years). These patients became the study’s primary intention-to-treat population.
The study’s primary outcome was the mean change in score on the Alzheimer Disease Assessment Scale–Cognitive Subscale 12 (ADAS-cog-12), which was evaluated at 3-month intervals.
Secondary clinical outcomes were assessed at 6-month intervals. These included the mean change in scores for the Alzheimer Disease Cooperative Study Activities of Daily Living Scale for Mild Cognitive Impairment and the Clinical Dementia Rating Scale Sum of Boxes.
Safety and adherence were also assessed during each study visit. Physical and neurologic examinations were performed at baseline and at months 6, 12, and 18.
Of the primary intention-to-treat population of 240 patients, 121 were randomly assigned to receive intranasal insulin. The remaining 119 received placebo and served as controls. The two groups were demographically comparable.
Better cognitive performance
A total of 215 participants completed the blinded phase; 198 participants completed the open-label extension. Discontinuation rates were comparable in both arms. The researchers found no differences between groups with respect to mean change in ADAS-cog-12 score from baseline to month 12 (0.0258 points; 95% confidence interval, –1.771 to 1.822 points; P = .98). The two groups also proved comparable in terms of performance on all other cognitive tests.
The open-label portion yielded similar results. Participants originally assigned to the insulin arm and their counterparts in the placebo arm did not differ with respect to mean score change on the ADAS-cog-12 test (or any other outcome) at either month 15 or 18.
Cerebrospinal fluid insulin levels were unchanged between groups, as were blood glucose and hemoglobin A1c values. Indeed, levels of A-beta42, A-beta40, total tau protein, and tau p-181 were comparable for the patients who received intranasal insulin and those who received placebo.
The most common adverse events were infections, injuries, respiratory disorders, and nervous system disorders, though these did not differ between groups. In addition, there were no differences between groups with respect to severity of adverse events; most were rated as mild.
In contrast with the intention-to-treat population, the study’s secondary analysis – using data from the original administration device – yielded markedly different results. In the blinded phase, patients who received insulin had better ADAS-cog-12 performance at 12 months (−2.81 points; 95% CI, −6.09 to 0.45 points; P = .09) and nominally significant effects at 6 months (−3.78 points; 95% CI, −6.79 to −0.78 points; P = .01).
Device type critical
These effects persisted in the open-label analyses. Patients who received intranasal insulin had superior ADAS-cog-12 scores at month 15 (−5.70 points; 95% CI, −9.62 to −1.79 points; P = .004) and month 18 (−5.78 points; 95% CI, −10.55 to −1.01 points; P = .02), compared with their counterparts who received insulin via the second device. This part of the study also showed that, although individual biomarkers did not differ significantly between the two arms, the ratios of A-beta42 to A-beta40 (P = .01) and A-beta42 to total tau (P = .03) increased with use of the first device. The number, type, and severity of adverse events were comparable between the insulin and placebo groups in this arm of the study.
The mixed results revealed by the trial demonstrate that the device used for intranasal insulin administration is paramount in determining the therapy’s potential efficacy. “Our take-home message is that the device is a very important factor for these studies and that one needs to validate their ability to effectively deliver insulin to the CNS,” said Dr. Craft.
“We were quite confident that the first device was able to do that. On the other hand, the second device has never been tested in that way, and we still don’t know whether or not that device was able to successfully deliver insulin,” she said.
The investigators recognize the need for more research in the field. Such studies, Dr. Craft noted, will utilize administration devices that have been previously verified to have the ability to deliver insulin to the central nervous system. “We’re currently testing several devices,” she noted. “We’re using a protocol where we administer insulin with the devices and then conduct a lumbar puncture about 30 minutes later to verify that it is actually raising insulin levels in the cerebrospinal fluid.”
Not a failure
Commenting on the findings, Samuel E. Gandy, MD, PhD, who was not involved in the study, said the research illustrates the challenge when a new therapy, a new delivery device, and a cohort of cognitively impaired patients collide. “The result is not quite a slam dunk but is also by no means a failure,” commented Dr. Gandy, Mount Sinai Chair in Alzheimer’s Research at Mount Sinai Medical Center, New York.
“One looks forward to future iterations of the Craft et al. approach, wherein the trialists tweak the ligand and/or the delivery schedule and/or the device and/or the disease and/or the disease stage,” Dr. Gandy added. “Another ligand, VGF, also holds promise for intranasal delivery, based on work from Steve Salton, Michelle Ehrlich, and Eric Schadt, all from Mount Sinai. Perhaps the nose knows!”
For Dr. Craft, the potential upside of intranasal insulin for these patients is significant and warrants further investigation. “I understand why people who are not familiar with prior research in this area might be skeptical of our enthusiasm, given the results in the intention-to-treat population,” she said. “But those of us who have been working along with this for a while now, we feel like we’ve got to do the next study. But we need to have a device that we know works,” Dr. Craft added.
“If this is real, then there may be a very large clinical benefit in symptomatic patients, and there’s nothing so far that has really improved symptomatic disease.”
The study was supported by the National Institute on Aging. Eli Lilly provided diluent placebo for the blinded phase and insulin for the open-label phase of the clinical trial at no cost. Dr. Craft received grants from the National Institute on Aging and nonfinancial support from Eli Lilly during the conduct of the study and personal fees from T3D Therapeutics and vTv Therapeutics outside the submitted work.
A version of this article originally appeared on Medscape.com.
The randomized trial of nearly 300 patients showed that, although one insulin administration device produced marked benefit in terms of change in mean score on the Alzheimer Disease Assessment Scale–Cognitive Subscale 12 (ADAS-cog-12) over 12 months, reliability was inconsistent. A second device, used on the majority of patients in the study’s intention-to-treat population, showed no difference in these measures between patients who did and those who did not receive intranasal insulin.
“The primary analysis of the study showed no benefit of intranasal insulin on any measures of cognition or cerebrospinal fluid Alzheimer’s disease biomarkers when using the new device,” said principal investigator Suzanne Craft, PhD.
“But when we looked at our planned secondary analysis with the original device – which has been successful in previous studies – we saw quite a different picture,” added Dr. Craft, director of the Alzheimer’s Disease Research Center at Wake Forest University, Winston-Salem, N.C.
“We found a pronounced benefit with that device, such that after 18 months of administration, participants who had been receiving insulin from the beginning of the study had a large and clinically significant advantage in the primary outcome measure.”
Dr. Craft described the findings as complex. “The primary results were negative,” she added. “But the secondary results replicated those of several earlier studies when we used the same device that was used in those.”
The study was published online June 22 in JAMA Neurology.
Important for brain function
Insulin has been shown to play several important roles in brain function. The hormone is associated with a variety of cognitive functions, including memory. Through its association with vasoreactivity, lipid metabolism, and inflammation, insulin also plays an important role in vascular function.
“In the normal brain in healthy individuals, insulin is very important for synaptic function and viability. Insulin also promotes dendritic growth and facilitates synaptic health. Through this role, it plays an important part in memory,” said Dr. Craft. Given these connections, it is not surprising that reduced insulin levels or activity in brain and cerebrospinal fluid have been documented in some, but not all, studies of Alzheimer’s disease. Markers of insulin resistance also have been detected in both neuronally derived exosomes and brain tissue from adults with Alzheimer’s disease.
In light of the several important roles that insulin plays in the brain – coupled with the evidence connecting dysregulation of brain insulin and AD pathology – restoring brain insulin function may offer therapeutic benefit for adults suffering either Alzheimer’s disease or MCI. “There are a number of ways to do this,” said Dr. Craft. “But one of the approaches that we’ve focused on is providing insulin directly to the brain through intranasal administration. “By doing this, you circumvent potential issues if you administered insulin systemically.”
Previous research has shown that through this mode of administration, insulin can bypass the blood-brain barrier and reach the brain through olfactory and trigeminal perivascular channels, with little effect on peripheral insulin or blood glucose levels.
As previously reported, an earlier pilot study, also conducted by Dr. Craft and her team, showed that 4 months of daily intranasal administration of 20 IU or 40 IU of insulin preserved cognitive performance in individuals with Alzheimer’s disease or MCI.
Deeper dive
In the current investigation, the researchers wanted to broaden these findings in a larger, longer, randomized double-blinded clinical trial. The investigators assessed the efficacy of intranasal insulin on cognition, function, and biomarkers of Alzheimer’s disease, as well as the safety and feasibility of the delivery method. The multicenter trial was conducted from 2014 to 2018 and included 27 sites.
Study participants were between the ages of 55 and 85 years and had been diagnosed with amnestic MCI or Alzheimer’s disease on the basis of National Institute on Aging–Alzheimer Association criteria, a score of 20 or higher on the Mini–Mental State Examination, a clinical dementia rating of 0.5 or 1.0, or a delayed logical memory score within a specified range.
In total, 289 participants were randomly assigned to receive 40 IU of insulin or placebo for 12 months, followed by a 6-month open-label extension phase. The first 49 participants (32 men; mean age, 71.9 years) underwent insulin administration with the same device the investigators used in previous trials.
Of these, 45 completed the blinded phase, and 42 completed the open-label extension. When this device, which uses an electronic nebulizer-like delivery system, proved unreliable, the researchers switched to a second device, which uses a liquid hydrofluoroalkane propellant to deliver a metered dose of insulin through a nose tip without electronic assistance. Device 2 was used for the remaining 240 participants (123 men; mean age, 70.8 years). These patients became the study’s primary intention-to-treat population.
The study’s primary outcome was the mean change in score on the Alzheimer Disease Assessment Scale–Cognitive Subscale 12 (ADAS-cog-12), which was evaluated at 3-month intervals.
Secondary clinical outcomes were assessed at 6-month intervals. These included the mean change in scores for the Alzheimer Disease Cooperative Study Activities of Daily Living Scale for Mild Cognitive Impairment and the Clinical Dementia Rating Scale Sum of Boxes.
Safety and adherence were also assessed during each study visit. Physical and neurologic examinations were performed at baseline and at months 6, 12, and 18.
Of the primary intention-to-treat population of 240 patients, 121 were randomly assigned to receive intranasal insulin. The remaining 119 received placebo and served as controls. The two groups were demographically comparable.
Better cognitive performance
A total of 215 participants completed the blinded phase; 198 participants completed the open-label extension. Discontinuation rates were comparable in both arms. The researchers found no differences between groups with respect to mean change in ADAS-cog-12 score from baseline to month 12 (0.0258 points; 95% confidence interval, –1.771 to 1.822 points; P = .98). The two groups also proved comparable in terms of performance on all other cognitive tests.
The open-label portion yielded similar results. Participants originally assigned to the insulin arm and their counterparts in the placebo arm did not differ with respect to mean score change on the ADAS-cog-12 test (or any other outcome) at either month 15 or 18.
Cerebrospinal fluid insulin levels were unchanged between groups, as were blood glucose and hemoglobin A1c values. Indeed, levels of A-beta42, A-beta40, total tau protein, and tau p-181 were comparable for the patients who received intranasal insulin and those who received placebo.
The most common adverse events were infections, injuries, respiratory disorders, and nervous system disorders, though these did not differ between groups. In addition, there were no differences between groups with respect to severity of adverse events; most were rated as mild.
In contrast with the intention-to-treat population, the study’s secondary analysis – using data from the original administration device – yielded markedly different results. In the blinded phase, patients who received insulin had better ADAS-cog-12 performance at 12 months (−2.81 points; 95% CI, −6.09 to 0.45 points; P = .09) and nominally significant effects at 6 months (−3.78 points; 95% CI, −6.79 to −0.78 points; P = .01).
Device type critical
These effects persisted in the open-label analyses. Patients who received intranasal insulin had superior ADAS-cog-12 scores at month 15 (−5.70 points; 95% CI, −9.62 to −1.79 points; P = .004) and month 18 (−5.78 points; 95% CI, −10.55 to −1.01 points; P = .02), compared with their counterparts who received insulin via the second device. This part of the study also showed that, although individual biomarkers did not differ significantly between the two arms, the ratios of A-beta42 to A-beta40 (P = .01) and A-beta42 to total tau (P = .03) increased with use of the first device. The number, type, and severity of adverse events were comparable between the insulin and placebo groups in this arm of the study.
The mixed results revealed by the trial demonstrate that the device used for intranasal insulin administration is paramount in determining the therapy’s potential efficacy. “Our take-home message is that the device is a very important factor for these studies and that one needs to validate their ability to effectively deliver insulin to the CNS,” said Dr. Craft.
“We were quite confident that the first device was able to do that. On the other hand, the second device has never been tested in that way, and we still don’t know whether or not that device was able to successfully deliver insulin,” she said.
The investigators recognize the need for more research in the field. Such studies, Dr. Craft noted, will utilize administration devices that have been previously verified to have the ability to deliver insulin to the central nervous system. “We’re currently testing several devices,” she noted. “We’re using a protocol where we administer insulin with the devices and then conduct a lumbar puncture about 30 minutes later to verify that it is actually raising insulin levels in the cerebrospinal fluid.”
Not a failure
Commenting on the findings, Samuel E. Gandy, MD, PhD, who was not involved in the study, said the research illustrates the challenge when a new therapy, a new delivery device, and a cohort of cognitively impaired patients collide. “The result is not quite a slam dunk but is also by no means a failure,” commented Dr. Gandy, Mount Sinai Chair in Alzheimer’s Research at Mount Sinai Medical Center, New York.
“One looks forward to future iterations of the Craft et al. approach, wherein the trialists tweak the ligand and/or the delivery schedule and/or the device and/or the disease and/or the disease stage,” Dr. Gandy added. “Another ligand, VGF, also holds promise for intranasal delivery, based on work from Steve Salton, Michelle Ehrlich, and Eric Schadt, all from Mount Sinai. Perhaps the nose knows!”
For Dr. Craft, the potential upside of intranasal insulin for these patients is significant and warrants further investigation. “I understand why people who are not familiar with prior research in this area might be skeptical of our enthusiasm, given the results in the intention-to-treat population,” she said. “But those of us who have been working along with this for a while now, we feel like we’ve got to do the next study. But we need to have a device that we know works,” Dr. Craft added.
“If this is real, then there may be a very large clinical benefit in symptomatic patients, and there’s nothing so far that has really improved symptomatic disease.”
The study was supported by the National Institute on Aging. Eli Lilly provided diluent placebo for the blinded phase and insulin for the open-label phase of the clinical trial at no cost. Dr. Craft received grants from the National Institute on Aging and nonfinancial support from Eli Lilly during the conduct of the study and personal fees from T3D Therapeutics and vTv Therapeutics outside the submitted work.
A version of this article originally appeared on Medscape.com.
The randomized trial of nearly 300 patients showed that, although one insulin administration device produced marked benefit in terms of change in mean score on the Alzheimer Disease Assessment Scale–Cognitive Subscale 12 (ADAS-cog-12) over 12 months, reliability was inconsistent. A second device, used on the majority of patients in the study’s intention-to-treat population, showed no difference in these measures between patients who did and those who did not receive intranasal insulin.
“The primary analysis of the study showed no benefit of intranasal insulin on any measures of cognition or cerebrospinal fluid Alzheimer’s disease biomarkers when using the new device,” said principal investigator Suzanne Craft, PhD.
“But when we looked at our planned secondary analysis with the original device – which has been successful in previous studies – we saw quite a different picture,” added Dr. Craft, director of the Alzheimer’s Disease Research Center at Wake Forest University, Winston-Salem, N.C.
“We found a pronounced benefit with that device, such that after 18 months of administration, participants who had been receiving insulin from the beginning of the study had a large and clinically significant advantage in the primary outcome measure.”
Dr. Craft described the findings as complex. “The primary results were negative,” she added. “But the secondary results replicated those of several earlier studies when we used the same device that was used in those.”
The study was published online June 22 in JAMA Neurology.
Important for brain function
Insulin has been shown to play several important roles in brain function. The hormone is associated with a variety of cognitive functions, including memory. Through its association with vasoreactivity, lipid metabolism, and inflammation, insulin also plays an important role in vascular function.
“In the normal brain in healthy individuals, insulin is very important for synaptic function and viability. Insulin also promotes dendritic growth and facilitates synaptic health. Through this role, it plays an important part in memory,” said Dr. Craft. Given these connections, it is not surprising that reduced insulin levels or activity in brain and cerebrospinal fluid have been documented in some, but not all, studies of Alzheimer’s disease. Markers of insulin resistance also have been detected in both neuronally derived exosomes and brain tissue from adults with Alzheimer’s disease.
In light of the several important roles that insulin plays in the brain – coupled with the evidence connecting dysregulation of brain insulin and AD pathology – restoring brain insulin function may offer therapeutic benefit for adults suffering either Alzheimer’s disease or MCI. “There are a number of ways to do this,” said Dr. Craft. “But one of the approaches that we’ve focused on is providing insulin directly to the brain through intranasal administration. “By doing this, you circumvent potential issues if you administered insulin systemically.”
Previous research has shown that through this mode of administration, insulin can bypass the blood-brain barrier and reach the brain through olfactory and trigeminal perivascular channels, with little effect on peripheral insulin or blood glucose levels.
As previously reported, an earlier pilot study, also conducted by Dr. Craft and her team, showed that 4 months of daily intranasal administration of 20 IU or 40 IU of insulin preserved cognitive performance in individuals with Alzheimer’s disease or MCI.
Deeper dive
In the current investigation, the researchers wanted to broaden these findings in a larger, longer, randomized double-blinded clinical trial. The investigators assessed the efficacy of intranasal insulin on cognition, function, and biomarkers of Alzheimer’s disease, as well as the safety and feasibility of the delivery method. The multicenter trial was conducted from 2014 to 2018 and included 27 sites.
Study participants were between the ages of 55 and 85 years and had been diagnosed with amnestic MCI or Alzheimer’s disease on the basis of National Institute on Aging–Alzheimer Association criteria, a score of 20 or higher on the Mini–Mental State Examination, a clinical dementia rating of 0.5 or 1.0, or a delayed logical memory score within a specified range.
In total, 289 participants were randomly assigned to receive 40 IU of insulin or placebo for 12 months, followed by a 6-month open-label extension phase. The first 49 participants (32 men; mean age, 71.9 years) underwent insulin administration with the same device the investigators used in previous trials.
Of these, 45 completed the blinded phase, and 42 completed the open-label extension. When this device, which uses an electronic nebulizer-like delivery system, proved unreliable, the researchers switched to a second device, which uses a liquid hydrofluoroalkane propellant to deliver a metered dose of insulin through a nose tip without electronic assistance. Device 2 was used for the remaining 240 participants (123 men; mean age, 70.8 years). These patients became the study’s primary intention-to-treat population.
The study’s primary outcome was the mean change in score on the Alzheimer Disease Assessment Scale–Cognitive Subscale 12 (ADAS-cog-12), which was evaluated at 3-month intervals.
Secondary clinical outcomes were assessed at 6-month intervals. These included the mean change in scores for the Alzheimer Disease Cooperative Study Activities of Daily Living Scale for Mild Cognitive Impairment and the Clinical Dementia Rating Scale Sum of Boxes.
Safety and adherence were also assessed during each study visit. Physical and neurologic examinations were performed at baseline and at months 6, 12, and 18.
Of the primary intention-to-treat population of 240 patients, 121 were randomly assigned to receive intranasal insulin. The remaining 119 received placebo and served as controls. The two groups were demographically comparable.
Better cognitive performance
A total of 215 participants completed the blinded phase; 198 participants completed the open-label extension. Discontinuation rates were comparable in both arms. The researchers found no differences between groups with respect to mean change in ADAS-cog-12 score from baseline to month 12 (0.0258 points; 95% confidence interval, –1.771 to 1.822 points; P = .98). The two groups also proved comparable in terms of performance on all other cognitive tests.
The open-label portion yielded similar results. Participants originally assigned to the insulin arm and their counterparts in the placebo arm did not differ with respect to mean score change on the ADAS-cog-12 test (or any other outcome) at either month 15 or 18.
Cerebrospinal fluid insulin levels were unchanged between groups, as were blood glucose and hemoglobin A1c values. Indeed, levels of A-beta42, A-beta40, total tau protein, and tau p-181 were comparable for the patients who received intranasal insulin and those who received placebo.
The most common adverse events were infections, injuries, respiratory disorders, and nervous system disorders, though these did not differ between groups. In addition, there were no differences between groups with respect to severity of adverse events; most were rated as mild.
In contrast with the intention-to-treat population, the study’s secondary analysis – using data from the original administration device – yielded markedly different results. In the blinded phase, patients who received insulin had better ADAS-cog-12 performance at 12 months (−2.81 points; 95% CI, −6.09 to 0.45 points; P = .09) and nominally significant effects at 6 months (−3.78 points; 95% CI, −6.79 to −0.78 points; P = .01).
Device type critical
These effects persisted in the open-label analyses. Patients who received intranasal insulin had superior ADAS-cog-12 scores at month 15 (−5.70 points; 95% CI, −9.62 to −1.79 points; P = .004) and month 18 (−5.78 points; 95% CI, −10.55 to −1.01 points; P = .02), compared with their counterparts who received insulin via the second device. This part of the study also showed that, although individual biomarkers did not differ significantly between the two arms, the ratios of A-beta42 to A-beta40 (P = .01) and A-beta42 to total tau (P = .03) increased with use of the first device. The number, type, and severity of adverse events were comparable between the insulin and placebo groups in this arm of the study.
The mixed results revealed by the trial demonstrate that the device used for intranasal insulin administration is paramount in determining the therapy’s potential efficacy. “Our take-home message is that the device is a very important factor for these studies and that one needs to validate their ability to effectively deliver insulin to the CNS,” said Dr. Craft.
“We were quite confident that the first device was able to do that. On the other hand, the second device has never been tested in that way, and we still don’t know whether or not that device was able to successfully deliver insulin,” she said.
The investigators recognize the need for more research in the field. Such studies, Dr. Craft noted, will utilize administration devices that have been previously verified to have the ability to deliver insulin to the central nervous system. “We’re currently testing several devices,” she noted. “We’re using a protocol where we administer insulin with the devices and then conduct a lumbar puncture about 30 minutes later to verify that it is actually raising insulin levels in the cerebrospinal fluid.”
Not a failure
Commenting on the findings, Samuel E. Gandy, MD, PhD, who was not involved in the study, said the research illustrates the challenge when a new therapy, a new delivery device, and a cohort of cognitively impaired patients collide. “The result is not quite a slam dunk but is also by no means a failure,” commented Dr. Gandy, Mount Sinai Chair in Alzheimer’s Research at Mount Sinai Medical Center, New York.
“One looks forward to future iterations of the Craft et al. approach, wherein the trialists tweak the ligand and/or the delivery schedule and/or the device and/or the disease and/or the disease stage,” Dr. Gandy added. “Another ligand, VGF, also holds promise for intranasal delivery, based on work from Steve Salton, Michelle Ehrlich, and Eric Schadt, all from Mount Sinai. Perhaps the nose knows!”
For Dr. Craft, the potential upside of intranasal insulin for these patients is significant and warrants further investigation. “I understand why people who are not familiar with prior research in this area might be skeptical of our enthusiasm, given the results in the intention-to-treat population,” she said. “But those of us who have been working along with this for a while now, we feel like we’ve got to do the next study. But we need to have a device that we know works,” Dr. Craft added.
“If this is real, then there may be a very large clinical benefit in symptomatic patients, and there’s nothing so far that has really improved symptomatic disease.”
The study was supported by the National Institute on Aging. Eli Lilly provided diluent placebo for the blinded phase and insulin for the open-label phase of the clinical trial at no cost. Dr. Craft received grants from the National Institute on Aging and nonfinancial support from Eli Lilly during the conduct of the study and personal fees from T3D Therapeutics and vTv Therapeutics outside the submitted work.
A version of this article originally appeared on Medscape.com.
FROM JAMA NEUROLOGY
Repetitive hits to the head tied to depression, poor cognition in later life
A history of repetitive hits to the head (RHI), even without noticeable symptoms, is linked to a significantly increased risk of depression and poorer cognition later in life, new research shows.
“We found that a history of exposure to [repetitive hits to the head] from contact sports, military service, or physical abuse, as well as a history of TBI (traumatic brain injury), corresponded to more symptoms of later life depression and worse cognitive function,” lead author Michael Alosco, PhD, associate professor of neurology and codirector of the Boston University Alzheimer’s Disease Center Clinical Core, told Medscape Medical News.
He added that the findings underscore the importance of assessing repetitive head impacts (RHI).
The study was published online June 26 in Neurology.
Largest study to date
It is well known that sustaining a TBI is associated with worse later life cognition or mood problems, said Alosco. However, in the current research the investigators hypothesized that RHI may be a key driver of some of these outcomes, Alosco said.
Previous studies have been small or have only examined male former football players.
“What’s unique about our study is that we focused on a history of RHIs, and it is the largest study of its kind, incorporating over 30,000 males and females with different types of exposure to these RHIs.”
The researchers used data from the Brain Health Registry, an internet-based registry that longitudinally monitors cognition and functioning of participants (age 40 years and older).
Participants completed the Ohio State University TBI Identification Method (OSU TBI-ID) and answered a yes/no question: “Have you ever had a period of time in which you experienced multiple, repeated impacts to your head (eg, history of abuse, contact sports, military duty)?”
Participants also completed the Geriatric Depression Scale (GDS-15), the CogState Battery (CBB), and the Lumos Labs NeuroCognitive Performance Tests (NCPT). Demographic information included age, sex, race/ethnicity, and level of education.
Negative synergistic effect
Of the total sample (N = 13,323, mean age 62 years, 72.5% female, 88.6% White) 725 participants (5%) reported exposure to RHI, with contact sports as the most common cause, followed by physical abuse and then military duty; about 55% (7277 participants) reported TBI.
The researchers noted that 44.4% of those exposed to RHI and 70.3% of those who reported TBI were female. However, those with a history of contact sports were predominantly male and those reporting a history of abuse were predominantly women.
Among study participants who completed the GDS-15, 16.4% reported symptoms of depression, similar to rates reported among community-dwelling older adults.
Compared to the unexposed group, participants who reported TBI with loss of consciousness (LOC) and participants who reported TBI without LOC both had higher scores on the GDS-15 (beta = 0.75 [95% CI, 0.59-0.91] and beta = 0.43 [95% CI, 0.31-0.54], respectively).
A history of RHI was associated with an even higher depression score (beta = 1.24 [95% CI, 0.36-2.12).
Depression increased in tandem with increased exposure, with the lowest GDS-15 scores found in the unexposed group and subsequent increases in scores as exposure to RHI was introduced and TBI severity increased. The GDS scores were highest in those who had RHI plus TBI with LOC.
Participants with a history of RHI and/or TBI also had worse scores on tests of memory, learning, processing speed, and reaction time, compared with unexposed participants.
In particular, TBI with LOC had the most neuropsychological associations.
TBI without LOC had a negative effect on CogState tests measuring Identification and processing speed (beta = 0.004 [95% CI, 0-0.01] and beta = 0.004 [95% CI, 0.0002-0.01], respectively), whereas RHI predicted a worse processing speed score (beta = .02 [95% CI, 0.01-0.05]).
The presence of both RHI and TBI (with or without LOC) had a “synergistic negative effect” on neuropsychological performance, with a “consistent statistically significant finding” for worse neuropsychological test performance for those who had RHI and TBI with LOC, compared with those who had not sustained RHI.
Alosco said the findings highlight the need for clinicians to educate and inform parents/guardians of kids playing (or considering playing) contact sports about the research and potential risks associated with these activities.
If we want to prevent long-term problems, one way is not to expose [people] to these hits. Everyone takes risks in life with everything, but the more we can understand and mitigate the risks, the better,” Alosco said.
“A significant contribution”
Commenting on the findings for Medscape Medical News, Temitayo Oyegbile-Chidi, MD, PhD, a pediatric neurologist with Health Peak Inc, McLean, Virginia, and a member of the American Academy of Neurology, said the study “makes a significant contribution to the literature, as neurologists who specialized in TBI have long yearned to understand the long-term effects of repeated head impact on the brain and cognition.”
Clinicians should “inquire about a history of prior head impacts on all our patients, regardless of age, especially if they are experiencing or showing signs of unexpected cognitive dysfunction or mental health concerns,” said Oyegbile-Chidi, who was not involved with the study.
For those who have sustained single or repeated head impacts with or without associated LOC in the past, “it is important … to keep in mind that depression and cognitive dysfunction may persist or present even many years after the impact was sustained,” she added.
The study was supported by a grant from the National Institutes of Health. Alosco has disclosed no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Oyegbile-Chidi has disclosed no relevant financial relationships.
A history of repetitive hits to the head (RHI), even without noticeable symptoms, is linked to a significantly increased risk of depression and poorer cognition later in life, new research shows.
“We found that a history of exposure to [repetitive hits to the head] from contact sports, military service, or physical abuse, as well as a history of TBI (traumatic brain injury), corresponded to more symptoms of later life depression and worse cognitive function,” lead author Michael Alosco, PhD, associate professor of neurology and codirector of the Boston University Alzheimer’s Disease Center Clinical Core, told Medscape Medical News.
He added that the findings underscore the importance of assessing repetitive head impacts (RHI).
The study was published online June 26 in Neurology.
Largest study to date
It is well known that sustaining a TBI is associated with worse later life cognition or mood problems, said Alosco. However, in the current research the investigators hypothesized that RHI may be a key driver of some of these outcomes, Alosco said.
Previous studies have been small or have only examined male former football players.
“What’s unique about our study is that we focused on a history of RHIs, and it is the largest study of its kind, incorporating over 30,000 males and females with different types of exposure to these RHIs.”
The researchers used data from the Brain Health Registry, an internet-based registry that longitudinally monitors cognition and functioning of participants (age 40 years and older).
Participants completed the Ohio State University TBI Identification Method (OSU TBI-ID) and answered a yes/no question: “Have you ever had a period of time in which you experienced multiple, repeated impacts to your head (eg, history of abuse, contact sports, military duty)?”
Participants also completed the Geriatric Depression Scale (GDS-15), the CogState Battery (CBB), and the Lumos Labs NeuroCognitive Performance Tests (NCPT). Demographic information included age, sex, race/ethnicity, and level of education.
Negative synergistic effect
Of the total sample (N = 13,323, mean age 62 years, 72.5% female, 88.6% White) 725 participants (5%) reported exposure to RHI, with contact sports as the most common cause, followed by physical abuse and then military duty; about 55% (7277 participants) reported TBI.
The researchers noted that 44.4% of those exposed to RHI and 70.3% of those who reported TBI were female. However, those with a history of contact sports were predominantly male and those reporting a history of abuse were predominantly women.
Among study participants who completed the GDS-15, 16.4% reported symptoms of depression, similar to rates reported among community-dwelling older adults.
Compared to the unexposed group, participants who reported TBI with loss of consciousness (LOC) and participants who reported TBI without LOC both had higher scores on the GDS-15 (beta = 0.75 [95% CI, 0.59-0.91] and beta = 0.43 [95% CI, 0.31-0.54], respectively).
A history of RHI was associated with an even higher depression score (beta = 1.24 [95% CI, 0.36-2.12).
Depression increased in tandem with increased exposure, with the lowest GDS-15 scores found in the unexposed group and subsequent increases in scores as exposure to RHI was introduced and TBI severity increased. The GDS scores were highest in those who had RHI plus TBI with LOC.
Participants with a history of RHI and/or TBI also had worse scores on tests of memory, learning, processing speed, and reaction time, compared with unexposed participants.
In particular, TBI with LOC had the most neuropsychological associations.
TBI without LOC had a negative effect on CogState tests measuring Identification and processing speed (beta = 0.004 [95% CI, 0-0.01] and beta = 0.004 [95% CI, 0.0002-0.01], respectively), whereas RHI predicted a worse processing speed score (beta = .02 [95% CI, 0.01-0.05]).
The presence of both RHI and TBI (with or without LOC) had a “synergistic negative effect” on neuropsychological performance, with a “consistent statistically significant finding” for worse neuropsychological test performance for those who had RHI and TBI with LOC, compared with those who had not sustained RHI.
Alosco said the findings highlight the need for clinicians to educate and inform parents/guardians of kids playing (or considering playing) contact sports about the research and potential risks associated with these activities.
If we want to prevent long-term problems, one way is not to expose [people] to these hits. Everyone takes risks in life with everything, but the more we can understand and mitigate the risks, the better,” Alosco said.
“A significant contribution”
Commenting on the findings for Medscape Medical News, Temitayo Oyegbile-Chidi, MD, PhD, a pediatric neurologist with Health Peak Inc, McLean, Virginia, and a member of the American Academy of Neurology, said the study “makes a significant contribution to the literature, as neurologists who specialized in TBI have long yearned to understand the long-term effects of repeated head impact on the brain and cognition.”
Clinicians should “inquire about a history of prior head impacts on all our patients, regardless of age, especially if they are experiencing or showing signs of unexpected cognitive dysfunction or mental health concerns,” said Oyegbile-Chidi, who was not involved with the study.
For those who have sustained single or repeated head impacts with or without associated LOC in the past, “it is important … to keep in mind that depression and cognitive dysfunction may persist or present even many years after the impact was sustained,” she added.
The study was supported by a grant from the National Institutes of Health. Alosco has disclosed no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Oyegbile-Chidi has disclosed no relevant financial relationships.
A history of repetitive hits to the head (RHI), even without noticeable symptoms, is linked to a significantly increased risk of depression and poorer cognition later in life, new research shows.
“We found that a history of exposure to [repetitive hits to the head] from contact sports, military service, or physical abuse, as well as a history of TBI (traumatic brain injury), corresponded to more symptoms of later life depression and worse cognitive function,” lead author Michael Alosco, PhD, associate professor of neurology and codirector of the Boston University Alzheimer’s Disease Center Clinical Core, told Medscape Medical News.
He added that the findings underscore the importance of assessing repetitive head impacts (RHI).
The study was published online June 26 in Neurology.
Largest study to date
It is well known that sustaining a TBI is associated with worse later life cognition or mood problems, said Alosco. However, in the current research the investigators hypothesized that RHI may be a key driver of some of these outcomes, Alosco said.
Previous studies have been small or have only examined male former football players.
“What’s unique about our study is that we focused on a history of RHIs, and it is the largest study of its kind, incorporating over 30,000 males and females with different types of exposure to these RHIs.”
The researchers used data from the Brain Health Registry, an internet-based registry that longitudinally monitors cognition and functioning of participants (age 40 years and older).
Participants completed the Ohio State University TBI Identification Method (OSU TBI-ID) and answered a yes/no question: “Have you ever had a period of time in which you experienced multiple, repeated impacts to your head (eg, history of abuse, contact sports, military duty)?”
Participants also completed the Geriatric Depression Scale (GDS-15), the CogState Battery (CBB), and the Lumos Labs NeuroCognitive Performance Tests (NCPT). Demographic information included age, sex, race/ethnicity, and level of education.
Negative synergistic effect
Of the total sample (N = 13,323, mean age 62 years, 72.5% female, 88.6% White) 725 participants (5%) reported exposure to RHI, with contact sports as the most common cause, followed by physical abuse and then military duty; about 55% (7277 participants) reported TBI.
The researchers noted that 44.4% of those exposed to RHI and 70.3% of those who reported TBI were female. However, those with a history of contact sports were predominantly male and those reporting a history of abuse were predominantly women.
Among study participants who completed the GDS-15, 16.4% reported symptoms of depression, similar to rates reported among community-dwelling older adults.
Compared to the unexposed group, participants who reported TBI with loss of consciousness (LOC) and participants who reported TBI without LOC both had higher scores on the GDS-15 (beta = 0.75 [95% CI, 0.59-0.91] and beta = 0.43 [95% CI, 0.31-0.54], respectively).
A history of RHI was associated with an even higher depression score (beta = 1.24 [95% CI, 0.36-2.12).
Depression increased in tandem with increased exposure, with the lowest GDS-15 scores found in the unexposed group and subsequent increases in scores as exposure to RHI was introduced and TBI severity increased. The GDS scores were highest in those who had RHI plus TBI with LOC.
Participants with a history of RHI and/or TBI also had worse scores on tests of memory, learning, processing speed, and reaction time, compared with unexposed participants.
In particular, TBI with LOC had the most neuropsychological associations.
TBI without LOC had a negative effect on CogState tests measuring Identification and processing speed (beta = 0.004 [95% CI, 0-0.01] and beta = 0.004 [95% CI, 0.0002-0.01], respectively), whereas RHI predicted a worse processing speed score (beta = .02 [95% CI, 0.01-0.05]).
The presence of both RHI and TBI (with or without LOC) had a “synergistic negative effect” on neuropsychological performance, with a “consistent statistically significant finding” for worse neuropsychological test performance for those who had RHI and TBI with LOC, compared with those who had not sustained RHI.
Alosco said the findings highlight the need for clinicians to educate and inform parents/guardians of kids playing (or considering playing) contact sports about the research and potential risks associated with these activities.
If we want to prevent long-term problems, one way is not to expose [people] to these hits. Everyone takes risks in life with everything, but the more we can understand and mitigate the risks, the better,” Alosco said.
“A significant contribution”
Commenting on the findings for Medscape Medical News, Temitayo Oyegbile-Chidi, MD, PhD, a pediatric neurologist with Health Peak Inc, McLean, Virginia, and a member of the American Academy of Neurology, said the study “makes a significant contribution to the literature, as neurologists who specialized in TBI have long yearned to understand the long-term effects of repeated head impact on the brain and cognition.”
Clinicians should “inquire about a history of prior head impacts on all our patients, regardless of age, especially if they are experiencing or showing signs of unexpected cognitive dysfunction or mental health concerns,” said Oyegbile-Chidi, who was not involved with the study.
For those who have sustained single or repeated head impacts with or without associated LOC in the past, “it is important … to keep in mind that depression and cognitive dysfunction may persist or present even many years after the impact was sustained,” she added.
The study was supported by a grant from the National Institutes of Health. Alosco has disclosed no relevant financial relationships. The other authors’ disclosures are listed on the original paper. Oyegbile-Chidi has disclosed no relevant financial relationships.
Move over supplements, here come medical foods
As the Food and Drug Administration focuses on other issues, companies, both big and small, are looking to boost physician and consumer interest in their “medical foods” – products that fall somewhere between drugs and supplements and promise to mitigate symptoms, or even address underlying pathologies, of a range of diseases.
Manufacturers now market an array of medical foods, ranging from powders and capsules for Alzheimer disease to low-protein spaghetti for chronic kidney disease (CKD). The FDA has not been completely absent; it takes a narrow view of what medical conditions qualify for treatment with food products and has warned some manufacturers that their misbranded products are acting more like unapproved drugs.
By the FDA’s definition, medical food is limited to products that provide crucial therapy for patients with inborn errors of metabolism (IEM). An example is specialized baby formula for infants with phenylketonuria. Unlike supplements, medical foods are supposed to be used under the supervision of a physician. This has prompted some sales reps to turn up in the clinic, and most manufacturers have online approval forms for doctors to sign. Manufacturers, advisers, and regulators were interviewed for a closer look at this burgeoning industry.
The market
The global market for medical foods – about $18 billion in 2019 – is expected to grow steadily in the near future. It is drawing more interest, especially in Europe, where medical foods are more accepted by physicians and consumers, Meghan Donnelly, MS, RDN, said in an interview. She is a registered dietitian who conducts physician outreach in the United States for Flavis, a division of Dr. Schär. That company, based in northern Italy, started out targeting IEMs but now also sells gluten-free foods for celiac disease and low-protein foods for CKD.
It is still a niche market in the United States – and isn’t likely to ever approach the size of the supplement market, according to Marcus Charuvastra, the managing director of Targeted Medical Pharma, which markets Theramine capsules for pain management, among many other products. But it could still be a big win for a manufacturer if they get a small slice of a big market, such as for Alzheimer disease.
Defining medical food
According to an update of the Orphan Drug Act in 1988, a medical food is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” The FDA issued regulations to accompany that law in 1993 but has since only issued a guidance document that is not legally binding.
Medical foods are not drugs and they are not supplements (the latter are intended only for healthy people). The FDA doesn’t require formal approval of a medical food, but, by law, the ingredients must be generally recognized as safe, and manufacturers must follow good manufacturing practices. However, the agency has taken a narrow view of what conditions require medical foods.
Policing medical foods hasn’t been a priority for the FDA, which is why there has been a proliferation of products that don’t meet the FDA’s view of the statutory definition of medical foods, according to Miriam Guggenheim, a food and drug law attorney in Washington, D.C. The FDA usually takes enforcement action when it sees a risk to the public’s health.
The agency’s stance has led to confusion – among manufacturers, physicians, consumers, and even regulators – making the market a kind of Wild West, according to Paul Hyman, a Washington, D.C.–based attorney who has represented medical food companies.
George A. Burdock, PhD, an Orlando-based regulatory consultant who has worked with medical food makers, believes the FDA will be forced to expand their narrow definition. He foresees a reconsideration of many medical food products in light of an October 2019 White House executive order prohibiting federal agencies from issuing guidance in lieu of rules.
Manufacturers and the FDA differ
One example of a product about which regulators and manufacturers differ is Theramine, which is described as “specially designed to supply the nervous system with the fuel it needs to meet the altered metabolic requirements of chronic pain and inflammatory disorders.”
It is not considered a medical food by the FDA, and the company has had numerous discussions with the agency about their diverging views, according to Mr. Charuvastra. “We’ve had our warning letters and we’ve had our sit downs, and we just had an inspection.”
Targeted Medical Pharma continues to market its products as medical foods but steers away from making any claims that they are like drugs, he said.
Confusion about medical foods has been exposed in the California Workers’ Compensation System by Leslie Wilson, PhD, and colleagues at the University of California, San Francisco. They found that physicians regularly wrote medical food prescriptions for non–FDA-approved uses and that the system reimbursed the majority of the products at a cost of $15.5 million from 2011 to 2013. More than half of these prescriptions were for Theramine.
Dr. Wilson reported that, for most products, no evidence supported effectiveness, and they were frequently mislabeled – for all 36 that were studied, submissions for reimbursement were made using a National Drug Code, an impossibility because medical foods are not drugs, and 14 were labeled “Rx only.”
Big-name companies joining in
The FDA does not keep a list of approved medical foods or manufacturers. Both small businesses and big food companies like Danone, Nestlé, and Abbott are players. Most products are sold online.
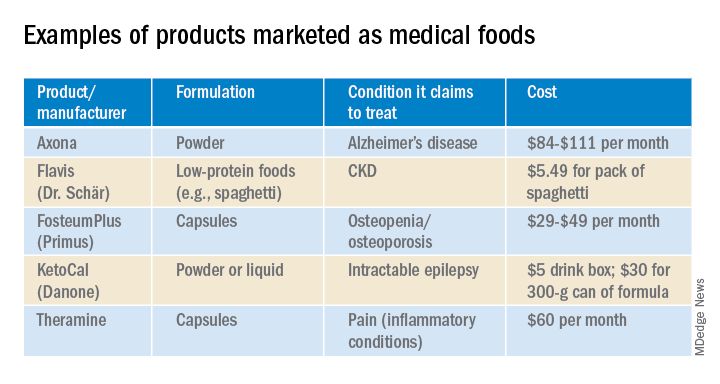
In the United States, Danone’s Nutricia division sells formulas and low-protein foods for IEMs. They also sell Ketocal, a powder or ready-to-drink liquid that is pitched as a balanced medical food to simplify and optimize the ketogenic diet for children with intractable epilepsy. Yet the FDA does not include epilepsy among the conditions that medical foods can treat.
Nestlé sells traditional medical foods for IEMs and also markets a range of what it calls nutritional therapies for such conditions as irritable bowel syndrome and dysphagia.
Nestlé is a minority shareholder in Axona, a product originally developed by Accera (Cerecin as of 2018). Jacquelyn Campo, senior director of global communications at Nestlé Health Sciences, said that the company is not actively involved in the operations management of Cerecin. However, on its website, Nestlé touts Axona, which is only available in the United States, as a “medical food” that “is intended for the clinical dietary management of mild to moderate Alzheimer disease.” The Axona site claims that the main ingredient, caprylic triglyceride, is broken down into ketones that provide fuel to treat cerebral hypometabolism, a precursor to Alzheimer disease. In a 2009 study, daily dosing of a preliminary formulation was associated with improved cognitive performance compared with placebo in patients with mild to moderate Alzheimer disease.
In 2013, the FDA warned Accera that it was misbranding Axona as a medical food and that the therapeutic claims the company was making would make the product an unapproved drug. Ms. Campo said Nestlé is aware of the agency’s warning, but added, “to our knowledge, Cerecin provided answers to the issues raised by the FDA.”
With the goal of getting drug approval, Accera went on to test a tweaked formulation in a 400-patient randomized, placebo-controlled trial called NOURISH AD that ultimately failed. Nevertheless, Axona is still marketed as a medical food. It costs about $100 for a month’s supply.
Repeated requests for comment from Cerecin were not answered. Danielle Schor, an FDA spokesperson, said the agency will not discuss the status of individual products.
More disputes and insurance coverage
Mary Ann DeMarco, executive director of sales and marketing for the Scottsdale, Ariz.–based medical food maker Primus Pharmaceuticals, said the company believes its products fit within the FDA’s medical foods rubric.
These include Fosteum Plus capsules, which it markets “for the clinical dietary management of the metabolic processes of osteopenia and osteoporosis.” The capsules contain a combination of genistein, zinc, calcium, phosphate, vitamin K2, and vitamin D. As proof of effectiveness, the company cites clinical data on some of the ingredients – not the product itself.
Primus has run afoul of the FDA before when it similarly positioned another product, called Limbrel, as a medical food for osteoarthritis. From 2007 to 2017, the FDA received 194 adverse event reports associated with Limbrel, including reports of drug-induced liver injury, pancreatitis, and hypersensitivity pneumonitis. In December 2017, the agency urged Primus to recall Limbrel, a move that it said was “necessary to protect the public health and welfare.” Primus withdrew the product but laid out a defense of Limbrel on a devoted website.
The FDA would not comment any further, said Ms. Schor. Ms. DeMarco said that Primus is working with the FDA to bring Limbrel back to market.
A lack of insurance coverage – even for approved medical foods for IEMs – has frustrated advocates, parents, and manufacturers. They are putting their weight behind the Medical Nutrition Equity Act, which would mandate public and private payer coverage of medical foods for IEMs and digestive conditions such as Crohn disease. That 2019 House bill has 56 cosponsors; there is no Senate companion bill.
“If you can get reimbursement, it really makes the market,” for Primus and the other manufacturers, Mr. Hyman said.
Primus Pharmaceuticals has launched its own campaign, Cover My Medical Foods, to enlist consumers and others to the cause.
Partnering with advocates
Although its low-protein breads, pastas, and baking products are not considered medical foods by the FDA, Dr. Schär is marketing them as such in the United States. They are trying to make a mark in CKD, according to Ms. Donnelly. She added that Dr. Schär has been successful in Europe, where nutrition therapy is more integrated in the health care system.
In 2019, Flavis and the National Kidney Foundation joined forces to raise awareness of nutritional interventions and to build enthusiasm for the Flavis products. The partnership has now ended, mostly because Flavis could no longer afford it, according to Ms. Donnelly.
“Information on diet and nutrition is the most requested subject matter from the NKF,” said Anthony Gucciardo, senior vice president of strategic partnerships at the foundation. The partnership “has never been necessarily about promoting their products per se; it’s promoting a healthy diet and really a diet specific for CKD.”
The NKF developed cobranded materials on low-protein foods for physicians and a teaching tool they could use with patients. Consumers could access nutrition information and a discount on Flavis products on a dedicated webpage. The foundation didn’t describe the low-protein products as medical foods, said Mr. Gucciardo, even if Flavis promoted them as such.
In patients with CKD, dietary management can help prevent the progression to end-stage renal disease. Although Medicare covers medical nutrition therapy – in which patients receive personalized assessments and dietary advice – uptake is abysmally low, according to a 2018 study.
Dr. Burdock thinks low-protein foods for CKD do meet the FDA’s criteria for a medical food but that the agency might not necessarily agree with him. The FDA would not comment.
Physician beware
When it comes to medical foods, the FDA has often looked the other way because the ingredients may already have been proven safe and the danger to an individual or to the public’s health is relatively low, according to Dr. Burdock and Mr. Hyman.
However, if the agency “feels that a medical food will prevent people from seeking medical care or there is potential to defraud the public, it is justified in taking action against the company,” said Dr. Burdock.
According to Dr. Wilson, the pharmacist who reported on the inappropriate medical food prescriptions in the California system, the FDA could help by creating a list of approved medical foods. Physicians should take time to learn about the difference between medical foods and supplements, she said, adding that they should also not hesitate to “question the veracity of the claims for them.”
Ms. Guggenheim believed doctors need to know that, for the most part, these are not FDA-approved products. She emphasized the importance of evaluating the products and looking at the data of their impact on a disease or condition.
“Many of these companies strongly believe that the products work and help people, so clinicians need to be very data driven,” she said.
A version of this article originally appeared on Medscape.com.
As the Food and Drug Administration focuses on other issues, companies, both big and small, are looking to boost physician and consumer interest in their “medical foods” – products that fall somewhere between drugs and supplements and promise to mitigate symptoms, or even address underlying pathologies, of a range of diseases.
Manufacturers now market an array of medical foods, ranging from powders and capsules for Alzheimer disease to low-protein spaghetti for chronic kidney disease (CKD). The FDA has not been completely absent; it takes a narrow view of what medical conditions qualify for treatment with food products and has warned some manufacturers that their misbranded products are acting more like unapproved drugs.
By the FDA’s definition, medical food is limited to products that provide crucial therapy for patients with inborn errors of metabolism (IEM). An example is specialized baby formula for infants with phenylketonuria. Unlike supplements, medical foods are supposed to be used under the supervision of a physician. This has prompted some sales reps to turn up in the clinic, and most manufacturers have online approval forms for doctors to sign. Manufacturers, advisers, and regulators were interviewed for a closer look at this burgeoning industry.
The market
The global market for medical foods – about $18 billion in 2019 – is expected to grow steadily in the near future. It is drawing more interest, especially in Europe, where medical foods are more accepted by physicians and consumers, Meghan Donnelly, MS, RDN, said in an interview. She is a registered dietitian who conducts physician outreach in the United States for Flavis, a division of Dr. Schär. That company, based in northern Italy, started out targeting IEMs but now also sells gluten-free foods for celiac disease and low-protein foods for CKD.
It is still a niche market in the United States – and isn’t likely to ever approach the size of the supplement market, according to Marcus Charuvastra, the managing director of Targeted Medical Pharma, which markets Theramine capsules for pain management, among many other products. But it could still be a big win for a manufacturer if they get a small slice of a big market, such as for Alzheimer disease.
Defining medical food
According to an update of the Orphan Drug Act in 1988, a medical food is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” The FDA issued regulations to accompany that law in 1993 but has since only issued a guidance document that is not legally binding.
Medical foods are not drugs and they are not supplements (the latter are intended only for healthy people). The FDA doesn’t require formal approval of a medical food, but, by law, the ingredients must be generally recognized as safe, and manufacturers must follow good manufacturing practices. However, the agency has taken a narrow view of what conditions require medical foods.
Policing medical foods hasn’t been a priority for the FDA, which is why there has been a proliferation of products that don’t meet the FDA’s view of the statutory definition of medical foods, according to Miriam Guggenheim, a food and drug law attorney in Washington, D.C. The FDA usually takes enforcement action when it sees a risk to the public’s health.
The agency’s stance has led to confusion – among manufacturers, physicians, consumers, and even regulators – making the market a kind of Wild West, according to Paul Hyman, a Washington, D.C.–based attorney who has represented medical food companies.
George A. Burdock, PhD, an Orlando-based regulatory consultant who has worked with medical food makers, believes the FDA will be forced to expand their narrow definition. He foresees a reconsideration of many medical food products in light of an October 2019 White House executive order prohibiting federal agencies from issuing guidance in lieu of rules.
Manufacturers and the FDA differ
One example of a product about which regulators and manufacturers differ is Theramine, which is described as “specially designed to supply the nervous system with the fuel it needs to meet the altered metabolic requirements of chronic pain and inflammatory disorders.”
It is not considered a medical food by the FDA, and the company has had numerous discussions with the agency about their diverging views, according to Mr. Charuvastra. “We’ve had our warning letters and we’ve had our sit downs, and we just had an inspection.”
Targeted Medical Pharma continues to market its products as medical foods but steers away from making any claims that they are like drugs, he said.
Confusion about medical foods has been exposed in the California Workers’ Compensation System by Leslie Wilson, PhD, and colleagues at the University of California, San Francisco. They found that physicians regularly wrote medical food prescriptions for non–FDA-approved uses and that the system reimbursed the majority of the products at a cost of $15.5 million from 2011 to 2013. More than half of these prescriptions were for Theramine.
Dr. Wilson reported that, for most products, no evidence supported effectiveness, and they were frequently mislabeled – for all 36 that were studied, submissions for reimbursement were made using a National Drug Code, an impossibility because medical foods are not drugs, and 14 were labeled “Rx only.”
Big-name companies joining in
The FDA does not keep a list of approved medical foods or manufacturers. Both small businesses and big food companies like Danone, Nestlé, and Abbott are players. Most products are sold online.

In the United States, Danone’s Nutricia division sells formulas and low-protein foods for IEMs. They also sell Ketocal, a powder or ready-to-drink liquid that is pitched as a balanced medical food to simplify and optimize the ketogenic diet for children with intractable epilepsy. Yet the FDA does not include epilepsy among the conditions that medical foods can treat.
Nestlé sells traditional medical foods for IEMs and also markets a range of what it calls nutritional therapies for such conditions as irritable bowel syndrome and dysphagia.
Nestlé is a minority shareholder in Axona, a product originally developed by Accera (Cerecin as of 2018). Jacquelyn Campo, senior director of global communications at Nestlé Health Sciences, said that the company is not actively involved in the operations management of Cerecin. However, on its website, Nestlé touts Axona, which is only available in the United States, as a “medical food” that “is intended for the clinical dietary management of mild to moderate Alzheimer disease.” The Axona site claims that the main ingredient, caprylic triglyceride, is broken down into ketones that provide fuel to treat cerebral hypometabolism, a precursor to Alzheimer disease. In a 2009 study, daily dosing of a preliminary formulation was associated with improved cognitive performance compared with placebo in patients with mild to moderate Alzheimer disease.
In 2013, the FDA warned Accera that it was misbranding Axona as a medical food and that the therapeutic claims the company was making would make the product an unapproved drug. Ms. Campo said Nestlé is aware of the agency’s warning, but added, “to our knowledge, Cerecin provided answers to the issues raised by the FDA.”
With the goal of getting drug approval, Accera went on to test a tweaked formulation in a 400-patient randomized, placebo-controlled trial called NOURISH AD that ultimately failed. Nevertheless, Axona is still marketed as a medical food. It costs about $100 for a month’s supply.
Repeated requests for comment from Cerecin were not answered. Danielle Schor, an FDA spokesperson, said the agency will not discuss the status of individual products.
More disputes and insurance coverage
Mary Ann DeMarco, executive director of sales and marketing for the Scottsdale, Ariz.–based medical food maker Primus Pharmaceuticals, said the company believes its products fit within the FDA’s medical foods rubric.
These include Fosteum Plus capsules, which it markets “for the clinical dietary management of the metabolic processes of osteopenia and osteoporosis.” The capsules contain a combination of genistein, zinc, calcium, phosphate, vitamin K2, and vitamin D. As proof of effectiveness, the company cites clinical data on some of the ingredients – not the product itself.
Primus has run afoul of the FDA before when it similarly positioned another product, called Limbrel, as a medical food for osteoarthritis. From 2007 to 2017, the FDA received 194 adverse event reports associated with Limbrel, including reports of drug-induced liver injury, pancreatitis, and hypersensitivity pneumonitis. In December 2017, the agency urged Primus to recall Limbrel, a move that it said was “necessary to protect the public health and welfare.” Primus withdrew the product but laid out a defense of Limbrel on a devoted website.
The FDA would not comment any further, said Ms. Schor. Ms. DeMarco said that Primus is working with the FDA to bring Limbrel back to market.
A lack of insurance coverage – even for approved medical foods for IEMs – has frustrated advocates, parents, and manufacturers. They are putting their weight behind the Medical Nutrition Equity Act, which would mandate public and private payer coverage of medical foods for IEMs and digestive conditions such as Crohn disease. That 2019 House bill has 56 cosponsors; there is no Senate companion bill.
“If you can get reimbursement, it really makes the market,” for Primus and the other manufacturers, Mr. Hyman said.
Primus Pharmaceuticals has launched its own campaign, Cover My Medical Foods, to enlist consumers and others to the cause.
Partnering with advocates
Although its low-protein breads, pastas, and baking products are not considered medical foods by the FDA, Dr. Schär is marketing them as such in the United States. They are trying to make a mark in CKD, according to Ms. Donnelly. She added that Dr. Schär has been successful in Europe, where nutrition therapy is more integrated in the health care system.
In 2019, Flavis and the National Kidney Foundation joined forces to raise awareness of nutritional interventions and to build enthusiasm for the Flavis products. The partnership has now ended, mostly because Flavis could no longer afford it, according to Ms. Donnelly.
“Information on diet and nutrition is the most requested subject matter from the NKF,” said Anthony Gucciardo, senior vice president of strategic partnerships at the foundation. The partnership “has never been necessarily about promoting their products per se; it’s promoting a healthy diet and really a diet specific for CKD.”
The NKF developed cobranded materials on low-protein foods for physicians and a teaching tool they could use with patients. Consumers could access nutrition information and a discount on Flavis products on a dedicated webpage. The foundation didn’t describe the low-protein products as medical foods, said Mr. Gucciardo, even if Flavis promoted them as such.
In patients with CKD, dietary management can help prevent the progression to end-stage renal disease. Although Medicare covers medical nutrition therapy – in which patients receive personalized assessments and dietary advice – uptake is abysmally low, according to a 2018 study.
Dr. Burdock thinks low-protein foods for CKD do meet the FDA’s criteria for a medical food but that the agency might not necessarily agree with him. The FDA would not comment.
Physician beware
When it comes to medical foods, the FDA has often looked the other way because the ingredients may already have been proven safe and the danger to an individual or to the public’s health is relatively low, according to Dr. Burdock and Mr. Hyman.
However, if the agency “feels that a medical food will prevent people from seeking medical care or there is potential to defraud the public, it is justified in taking action against the company,” said Dr. Burdock.
According to Dr. Wilson, the pharmacist who reported on the inappropriate medical food prescriptions in the California system, the FDA could help by creating a list of approved medical foods. Physicians should take time to learn about the difference between medical foods and supplements, she said, adding that they should also not hesitate to “question the veracity of the claims for them.”
Ms. Guggenheim believed doctors need to know that, for the most part, these are not FDA-approved products. She emphasized the importance of evaluating the products and looking at the data of their impact on a disease or condition.
“Many of these companies strongly believe that the products work and help people, so clinicians need to be very data driven,” she said.
A version of this article originally appeared on Medscape.com.
As the Food and Drug Administration focuses on other issues, companies, both big and small, are looking to boost physician and consumer interest in their “medical foods” – products that fall somewhere between drugs and supplements and promise to mitigate symptoms, or even address underlying pathologies, of a range of diseases.
Manufacturers now market an array of medical foods, ranging from powders and capsules for Alzheimer disease to low-protein spaghetti for chronic kidney disease (CKD). The FDA has not been completely absent; it takes a narrow view of what medical conditions qualify for treatment with food products and has warned some manufacturers that their misbranded products are acting more like unapproved drugs.
By the FDA’s definition, medical food is limited to products that provide crucial therapy for patients with inborn errors of metabolism (IEM). An example is specialized baby formula for infants with phenylketonuria. Unlike supplements, medical foods are supposed to be used under the supervision of a physician. This has prompted some sales reps to turn up in the clinic, and most manufacturers have online approval forms for doctors to sign. Manufacturers, advisers, and regulators were interviewed for a closer look at this burgeoning industry.
The market
The global market for medical foods – about $18 billion in 2019 – is expected to grow steadily in the near future. It is drawing more interest, especially in Europe, where medical foods are more accepted by physicians and consumers, Meghan Donnelly, MS, RDN, said in an interview. She is a registered dietitian who conducts physician outreach in the United States for Flavis, a division of Dr. Schär. That company, based in northern Italy, started out targeting IEMs but now also sells gluten-free foods for celiac disease and low-protein foods for CKD.
It is still a niche market in the United States – and isn’t likely to ever approach the size of the supplement market, according to Marcus Charuvastra, the managing director of Targeted Medical Pharma, which markets Theramine capsules for pain management, among many other products. But it could still be a big win for a manufacturer if they get a small slice of a big market, such as for Alzheimer disease.
Defining medical food
According to an update of the Orphan Drug Act in 1988, a medical food is “a food which is formulated to be consumed or administered enterally under the supervision of a physician and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” The FDA issued regulations to accompany that law in 1993 but has since only issued a guidance document that is not legally binding.
Medical foods are not drugs and they are not supplements (the latter are intended only for healthy people). The FDA doesn’t require formal approval of a medical food, but, by law, the ingredients must be generally recognized as safe, and manufacturers must follow good manufacturing practices. However, the agency has taken a narrow view of what conditions require medical foods.
Policing medical foods hasn’t been a priority for the FDA, which is why there has been a proliferation of products that don’t meet the FDA’s view of the statutory definition of medical foods, according to Miriam Guggenheim, a food and drug law attorney in Washington, D.C. The FDA usually takes enforcement action when it sees a risk to the public’s health.
The agency’s stance has led to confusion – among manufacturers, physicians, consumers, and even regulators – making the market a kind of Wild West, according to Paul Hyman, a Washington, D.C.–based attorney who has represented medical food companies.
George A. Burdock, PhD, an Orlando-based regulatory consultant who has worked with medical food makers, believes the FDA will be forced to expand their narrow definition. He foresees a reconsideration of many medical food products in light of an October 2019 White House executive order prohibiting federal agencies from issuing guidance in lieu of rules.
Manufacturers and the FDA differ
One example of a product about which regulators and manufacturers differ is Theramine, which is described as “specially designed to supply the nervous system with the fuel it needs to meet the altered metabolic requirements of chronic pain and inflammatory disorders.”
It is not considered a medical food by the FDA, and the company has had numerous discussions with the agency about their diverging views, according to Mr. Charuvastra. “We’ve had our warning letters and we’ve had our sit downs, and we just had an inspection.”
Targeted Medical Pharma continues to market its products as medical foods but steers away from making any claims that they are like drugs, he said.
Confusion about medical foods has been exposed in the California Workers’ Compensation System by Leslie Wilson, PhD, and colleagues at the University of California, San Francisco. They found that physicians regularly wrote medical food prescriptions for non–FDA-approved uses and that the system reimbursed the majority of the products at a cost of $15.5 million from 2011 to 2013. More than half of these prescriptions were for Theramine.
Dr. Wilson reported that, for most products, no evidence supported effectiveness, and they were frequently mislabeled – for all 36 that were studied, submissions for reimbursement were made using a National Drug Code, an impossibility because medical foods are not drugs, and 14 were labeled “Rx only.”
Big-name companies joining in
The FDA does not keep a list of approved medical foods or manufacturers. Both small businesses and big food companies like Danone, Nestlé, and Abbott are players. Most products are sold online.

In the United States, Danone’s Nutricia division sells formulas and low-protein foods for IEMs. They also sell Ketocal, a powder or ready-to-drink liquid that is pitched as a balanced medical food to simplify and optimize the ketogenic diet for children with intractable epilepsy. Yet the FDA does not include epilepsy among the conditions that medical foods can treat.
Nestlé sells traditional medical foods for IEMs and also markets a range of what it calls nutritional therapies for such conditions as irritable bowel syndrome and dysphagia.
Nestlé is a minority shareholder in Axona, a product originally developed by Accera (Cerecin as of 2018). Jacquelyn Campo, senior director of global communications at Nestlé Health Sciences, said that the company is not actively involved in the operations management of Cerecin. However, on its website, Nestlé touts Axona, which is only available in the United States, as a “medical food” that “is intended for the clinical dietary management of mild to moderate Alzheimer disease.” The Axona site claims that the main ingredient, caprylic triglyceride, is broken down into ketones that provide fuel to treat cerebral hypometabolism, a precursor to Alzheimer disease. In a 2009 study, daily dosing of a preliminary formulation was associated with improved cognitive performance compared with placebo in patients with mild to moderate Alzheimer disease.
In 2013, the FDA warned Accera that it was misbranding Axona as a medical food and that the therapeutic claims the company was making would make the product an unapproved drug. Ms. Campo said Nestlé is aware of the agency’s warning, but added, “to our knowledge, Cerecin provided answers to the issues raised by the FDA.”
With the goal of getting drug approval, Accera went on to test a tweaked formulation in a 400-patient randomized, placebo-controlled trial called NOURISH AD that ultimately failed. Nevertheless, Axona is still marketed as a medical food. It costs about $100 for a month’s supply.
Repeated requests for comment from Cerecin were not answered. Danielle Schor, an FDA spokesperson, said the agency will not discuss the status of individual products.
More disputes and insurance coverage
Mary Ann DeMarco, executive director of sales and marketing for the Scottsdale, Ariz.–based medical food maker Primus Pharmaceuticals, said the company believes its products fit within the FDA’s medical foods rubric.
These include Fosteum Plus capsules, which it markets “for the clinical dietary management of the metabolic processes of osteopenia and osteoporosis.” The capsules contain a combination of genistein, zinc, calcium, phosphate, vitamin K2, and vitamin D. As proof of effectiveness, the company cites clinical data on some of the ingredients – not the product itself.
Primus has run afoul of the FDA before when it similarly positioned another product, called Limbrel, as a medical food for osteoarthritis. From 2007 to 2017, the FDA received 194 adverse event reports associated with Limbrel, including reports of drug-induced liver injury, pancreatitis, and hypersensitivity pneumonitis. In December 2017, the agency urged Primus to recall Limbrel, a move that it said was “necessary to protect the public health and welfare.” Primus withdrew the product but laid out a defense of Limbrel on a devoted website.
The FDA would not comment any further, said Ms. Schor. Ms. DeMarco said that Primus is working with the FDA to bring Limbrel back to market.
A lack of insurance coverage – even for approved medical foods for IEMs – has frustrated advocates, parents, and manufacturers. They are putting their weight behind the Medical Nutrition Equity Act, which would mandate public and private payer coverage of medical foods for IEMs and digestive conditions such as Crohn disease. That 2019 House bill has 56 cosponsors; there is no Senate companion bill.
“If you can get reimbursement, it really makes the market,” for Primus and the other manufacturers, Mr. Hyman said.
Primus Pharmaceuticals has launched its own campaign, Cover My Medical Foods, to enlist consumers and others to the cause.
Partnering with advocates
Although its low-protein breads, pastas, and baking products are not considered medical foods by the FDA, Dr. Schär is marketing them as such in the United States. They are trying to make a mark in CKD, according to Ms. Donnelly. She added that Dr. Schär has been successful in Europe, where nutrition therapy is more integrated in the health care system.
In 2019, Flavis and the National Kidney Foundation joined forces to raise awareness of nutritional interventions and to build enthusiasm for the Flavis products. The partnership has now ended, mostly because Flavis could no longer afford it, according to Ms. Donnelly.
“Information on diet and nutrition is the most requested subject matter from the NKF,” said Anthony Gucciardo, senior vice president of strategic partnerships at the foundation. The partnership “has never been necessarily about promoting their products per se; it’s promoting a healthy diet and really a diet specific for CKD.”
The NKF developed cobranded materials on low-protein foods for physicians and a teaching tool they could use with patients. Consumers could access nutrition information and a discount on Flavis products on a dedicated webpage. The foundation didn’t describe the low-protein products as medical foods, said Mr. Gucciardo, even if Flavis promoted them as such.
In patients with CKD, dietary management can help prevent the progression to end-stage renal disease. Although Medicare covers medical nutrition therapy – in which patients receive personalized assessments and dietary advice – uptake is abysmally low, according to a 2018 study.
Dr. Burdock thinks low-protein foods for CKD do meet the FDA’s criteria for a medical food but that the agency might not necessarily agree with him. The FDA would not comment.
Physician beware
When it comes to medical foods, the FDA has often looked the other way because the ingredients may already have been proven safe and the danger to an individual or to the public’s health is relatively low, according to Dr. Burdock and Mr. Hyman.
However, if the agency “feels that a medical food will prevent people from seeking medical care or there is potential to defraud the public, it is justified in taking action against the company,” said Dr. Burdock.
According to Dr. Wilson, the pharmacist who reported on the inappropriate medical food prescriptions in the California system, the FDA could help by creating a list of approved medical foods. Physicians should take time to learn about the difference between medical foods and supplements, she said, adding that they should also not hesitate to “question the veracity of the claims for them.”
Ms. Guggenheim believed doctors need to know that, for the most part, these are not FDA-approved products. She emphasized the importance of evaluating the products and looking at the data of their impact on a disease or condition.
“Many of these companies strongly believe that the products work and help people, so clinicians need to be very data driven,” she said.
A version of this article originally appeared on Medscape.com.
Blood biomarker detects concussion, shows severity, predicts recovery
(TBI), new research indicates.
“Blood NfL may be used to aid in the diagnosis of patients with concussion or mild TBI [and] to identify individuals at increased risk of developing persistent postconcussive symptoms following TBI,” said lead author Pashtun Shahim, MD, PhD, National Institutes of Health Clinical Center, Bethesda, Md.
“This study is the first to do a detailed assessment of serum NfL chain and advanced brain imaging in multiple cohorts, brain injury severities, and time points after injury. The cohorts included professional athletes and nonathletes, and over time up to 5 years after TBI,” Dr. Shahim added.
The study was published online July 8 in Neurology.
Rapid indicator of neuronal damage
The researchers studied two cohorts of patients with head injuries. In the first, they determined serum and CSF NfL chain levels in professional Swedish ice hockey players (median age, 27 years), including 45 with acute concussion, 31 with repetitive concussions and persistent post-concussive symptoms (PCS), 28 who contributed samples during preseason with no recent concussion, and 14 healthy nonathletes.
CSF and serum NfL concentrations were closely correlated (r = 0.71; P < .0001). Serum NfL distinguished players with persistent PCS due to repetitive concussions from preseason concussion-free players, with an area under the receiver operating characteristic curve of 0.97. Higher CSF and serum NfL levels were associated with a higher number of concussions and severity of PCS after 1 year.
The second cohort involved 230 clinic-based adults (mean age, 43 years), including 162 with TBI and 68 healthy controls. In this cohort, patients with TBI had increased serum NfL concentrations compared with controls for up to 5 years, and these concentrations were able to distinguish between mild, moderate, and severe TBI. Serum NfL also correlated with measures of functional outcome, MRI brain atrophy, and diffusion tensor imaging estimates of traumatic axonal injury.
“Our findings suggest that NfL concentrations in serum offer rapid and accessible means of assessing and predicting neuronal damage in patients with TBI,” the investigators wrote.
What’s needed going forward, said Dr. Shahim, is “validation in larger cohorts for determining what levels of NfL in blood may be associated with a specific type of TBI, and what the levels are in healthy individuals of different ages.”
Not ready for prime time
In an accompanying editorial, Christopher Filley, MD, University of Colorado at Denver, Aurora, noted that NfL “may prove useful in identifying TBI patients at risk for prolonged symptoms and in enabling more focused treatment for these individuals.”
“These reports are richly laden with acute and longitudinal data that not only support the use of NfL as a convenient diagnostic test for TBI, but plausibly correlate with the neuropathology of TBI that is thought to play a major role in immediate and lasting cognitive disability,” he wrote.
Although the origin of TBI-induced cognitive decline is not entirely explained by traumatic axonal injury, “NfL appears to have much promise as a blood test that relates directly to the ubiquitous white matter damage of TBI, revealing a great deal about not only whether a TBI occurred, but also the extent of injury sustained, and how this injury may affect patient outcome for years thereafter,” Dr. Filley wrote.
However, he cautioned more research is needed before the blood test can be routinely applied to TBI diagnosis in clinical practice. “Among the hurdles still ahead are the standardization of measurement techniques across analytical platforms, and the determination of precise cutoffs between normal and abnormal values in different ages groups and at varying levels of TBI severity,” Dr. Filley noted.
The research was supported by the National Institutes of Health, the Department of Defense, the Center for Neuroscience and Regenerative Medicine at the Uniformed Services University, and the Swedish Research Council. Dr. Shahim and Dr. Filley have reported no relevant financial relationships.
This article first appeared on Medscape.com.
(TBI), new research indicates.
“Blood NfL may be used to aid in the diagnosis of patients with concussion or mild TBI [and] to identify individuals at increased risk of developing persistent postconcussive symptoms following TBI,” said lead author Pashtun Shahim, MD, PhD, National Institutes of Health Clinical Center, Bethesda, Md.
“This study is the first to do a detailed assessment of serum NfL chain and advanced brain imaging in multiple cohorts, brain injury severities, and time points after injury. The cohorts included professional athletes and nonathletes, and over time up to 5 years after TBI,” Dr. Shahim added.
The study was published online July 8 in Neurology.
Rapid indicator of neuronal damage
The researchers studied two cohorts of patients with head injuries. In the first, they determined serum and CSF NfL chain levels in professional Swedish ice hockey players (median age, 27 years), including 45 with acute concussion, 31 with repetitive concussions and persistent post-concussive symptoms (PCS), 28 who contributed samples during preseason with no recent concussion, and 14 healthy nonathletes.
CSF and serum NfL concentrations were closely correlated (r = 0.71; P < .0001). Serum NfL distinguished players with persistent PCS due to repetitive concussions from preseason concussion-free players, with an area under the receiver operating characteristic curve of 0.97. Higher CSF and serum NfL levels were associated with a higher number of concussions and severity of PCS after 1 year.
The second cohort involved 230 clinic-based adults (mean age, 43 years), including 162 with TBI and 68 healthy controls. In this cohort, patients with TBI had increased serum NfL concentrations compared with controls for up to 5 years, and these concentrations were able to distinguish between mild, moderate, and severe TBI. Serum NfL also correlated with measures of functional outcome, MRI brain atrophy, and diffusion tensor imaging estimates of traumatic axonal injury.
“Our findings suggest that NfL concentrations in serum offer rapid and accessible means of assessing and predicting neuronal damage in patients with TBI,” the investigators wrote.
What’s needed going forward, said Dr. Shahim, is “validation in larger cohorts for determining what levels of NfL in blood may be associated with a specific type of TBI, and what the levels are in healthy individuals of different ages.”
Not ready for prime time
In an accompanying editorial, Christopher Filley, MD, University of Colorado at Denver, Aurora, noted that NfL “may prove useful in identifying TBI patients at risk for prolonged symptoms and in enabling more focused treatment for these individuals.”
“These reports are richly laden with acute and longitudinal data that not only support the use of NfL as a convenient diagnostic test for TBI, but plausibly correlate with the neuropathology of TBI that is thought to play a major role in immediate and lasting cognitive disability,” he wrote.
Although the origin of TBI-induced cognitive decline is not entirely explained by traumatic axonal injury, “NfL appears to have much promise as a blood test that relates directly to the ubiquitous white matter damage of TBI, revealing a great deal about not only whether a TBI occurred, but also the extent of injury sustained, and how this injury may affect patient outcome for years thereafter,” Dr. Filley wrote.
However, he cautioned more research is needed before the blood test can be routinely applied to TBI diagnosis in clinical practice. “Among the hurdles still ahead are the standardization of measurement techniques across analytical platforms, and the determination of precise cutoffs between normal and abnormal values in different ages groups and at varying levels of TBI severity,” Dr. Filley noted.
The research was supported by the National Institutes of Health, the Department of Defense, the Center for Neuroscience and Regenerative Medicine at the Uniformed Services University, and the Swedish Research Council. Dr. Shahim and Dr. Filley have reported no relevant financial relationships.
This article first appeared on Medscape.com.
(TBI), new research indicates.
“Blood NfL may be used to aid in the diagnosis of patients with concussion or mild TBI [and] to identify individuals at increased risk of developing persistent postconcussive symptoms following TBI,” said lead author Pashtun Shahim, MD, PhD, National Institutes of Health Clinical Center, Bethesda, Md.
“This study is the first to do a detailed assessment of serum NfL chain and advanced brain imaging in multiple cohorts, brain injury severities, and time points after injury. The cohorts included professional athletes and nonathletes, and over time up to 5 years after TBI,” Dr. Shahim added.
The study was published online July 8 in Neurology.
Rapid indicator of neuronal damage
The researchers studied two cohorts of patients with head injuries. In the first, they determined serum and CSF NfL chain levels in professional Swedish ice hockey players (median age, 27 years), including 45 with acute concussion, 31 with repetitive concussions and persistent post-concussive symptoms (PCS), 28 who contributed samples during preseason with no recent concussion, and 14 healthy nonathletes.
CSF and serum NfL concentrations were closely correlated (r = 0.71; P < .0001). Serum NfL distinguished players with persistent PCS due to repetitive concussions from preseason concussion-free players, with an area under the receiver operating characteristic curve of 0.97. Higher CSF and serum NfL levels were associated with a higher number of concussions and severity of PCS after 1 year.
The second cohort involved 230 clinic-based adults (mean age, 43 years), including 162 with TBI and 68 healthy controls. In this cohort, patients with TBI had increased serum NfL concentrations compared with controls for up to 5 years, and these concentrations were able to distinguish between mild, moderate, and severe TBI. Serum NfL also correlated with measures of functional outcome, MRI brain atrophy, and diffusion tensor imaging estimates of traumatic axonal injury.
“Our findings suggest that NfL concentrations in serum offer rapid and accessible means of assessing and predicting neuronal damage in patients with TBI,” the investigators wrote.
What’s needed going forward, said Dr. Shahim, is “validation in larger cohorts for determining what levels of NfL in blood may be associated with a specific type of TBI, and what the levels are in healthy individuals of different ages.”
Not ready for prime time
In an accompanying editorial, Christopher Filley, MD, University of Colorado at Denver, Aurora, noted that NfL “may prove useful in identifying TBI patients at risk for prolonged symptoms and in enabling more focused treatment for these individuals.”
“These reports are richly laden with acute and longitudinal data that not only support the use of NfL as a convenient diagnostic test for TBI, but plausibly correlate with the neuropathology of TBI that is thought to play a major role in immediate and lasting cognitive disability,” he wrote.
Although the origin of TBI-induced cognitive decline is not entirely explained by traumatic axonal injury, “NfL appears to have much promise as a blood test that relates directly to the ubiquitous white matter damage of TBI, revealing a great deal about not only whether a TBI occurred, but also the extent of injury sustained, and how this injury may affect patient outcome for years thereafter,” Dr. Filley wrote.
However, he cautioned more research is needed before the blood test can be routinely applied to TBI diagnosis in clinical practice. “Among the hurdles still ahead are the standardization of measurement techniques across analytical platforms, and the determination of precise cutoffs between normal and abnormal values in different ages groups and at varying levels of TBI severity,” Dr. Filley noted.
The research was supported by the National Institutes of Health, the Department of Defense, the Center for Neuroscience and Regenerative Medicine at the Uniformed Services University, and the Swedish Research Council. Dr. Shahim and Dr. Filley have reported no relevant financial relationships.
This article first appeared on Medscape.com.
New insight into neurobehavioral effects of legalized cannabis
Researchers have published one of the first studies to characterize the association between the consumption of legal cannabis and subsequent pharmacologic and neurobehavioral outcomes, with somewhat surprising results.
The study showed that, although cannabis consumption did not affect most short-term neurobehavioral measures, it delayed recall memory and impaired balance.
The investigation also showed that users of much more potent cannabis concentrates actually demonstrated similar or lower levels of subjective drug intoxication and short-term impairment than did their counterparts who used lower-potency forms of the cannabis flower.
“It does not appear that the potency being used matters that much,” senior investigator Kent E. Hutchison, PhD, said in an interview. “People seem to be titrating to a certain level of intoxication or a certain level of feeling high. And for some people that requires a lot of drug, and for other people not as much.”
added Dr. Hutchison, a professor of psychology and neuroscience at the University of Colorado Boulder.
The study was published online June 10 in JAMA Psychiatry.
Widespread availability, little research
Recreational cannabis is now legal in 11 states and the District of Columbia, while medical cannabis is legal in 33. However, despite growing popularity of cannabis, there is little research on its potential health and biobehavioral risks, largely because of federal restrictions on cannabis research.
Cannabis users typically consume various forms of the cannabis flower, which can boast concentrations of the psychoactive cannabinoid delta-9-tetrahydrocannabinol (THC) of up to 30%. However, use of concentrated forms of cannabis – which are made by extracting plant cannabinoids into a different form – is increasing.
Such formulations can boast THC concentrations as high as 90%. Nevertheless, data regarding the relative risks of these higher-strength products are limited.
Previous research has shown a variety of negative short-term and long-term neurobehavioral effects associated with cannabis use, including harmful cognitive and motor effects. Extended exposure to THC may also negatively affect brain regions that are associated with the control of coordinated movement, and create brain-activation deficits in motor control regions that persist well beyond the effects of short-term intoxication.
Despite such findings, Dr. Hutchison said the existing literature on the subject does not yield a real-world view of current cannabis use because it tends to focus on low-THC products that are increasingly less common in today’s legal market.
Given such shortcomings, the investigators wanted to address persistent questions surrounding the neurobehavioral effects of legal cannabis flower products (16% or 24% THC) and cannabis concentrate products (70% or 90% THC). In doing so, they examined three primary topics:
- The association between short-term use of these products and THC plasma levels; subjective intoxication; and mood, cognitive performance, and balance
- Differences in such associations between users of cannabis flower and concentrate products
- Potential variations in these associations by THC potency
High- versus low-potency varieties
The study included 133 individuals (aged 21-70 years), who were designated as either cannabis flower users or cannabis concentrate users. Participants had all used cannabis at least four times in the previous month with no adverse reaction and were not receiving treatment for a psychotic disorder or bipolar disorder.
Participants were randomly assigned to consume either higher-potency or lower-potency products that had been purchased from a local dispensary. Flower users were randomized to purchase 3 g of either a 16% THC or 24% THC product, while concentrate users were randomized to purchase 1g of either 70% THC or 90% THC.
Participants completed a series of four assessments, one at baseline and three others at a mobile laboratory. The mobile laboratory assessments occurred before, immediately after, and one hour after participants had all consumed their cannabis ad libitum.
Of the original cohort of 133 participants, 55 flower cannabis users (mean age, 28.8 years; 46% women) and 66 concentrate cannabis users (mean age, 28.3 years; 45% women) complied with the study’s instructions and had complete data.
The study’s primary outcome measures included plasma cannabinoids, subjective drug intoxication and mood, and neurobehavioral outcomes such as attention, memory, inhibitory control, and balance.
Mixed results
With respect to cannabis concentrations, results showed that users of concentrate exhibited higher levels of both plasma THC and the active metabolite of THC (11-hydroxy-delta9-THC) across all points than did their counterparts who used cannabis flower products.
Specifically, mean plasma THC levels were 1,016 ± 1,380 mcg/ml in concentrate users and 455±503 mcg/mL in flower users after ad libitum cannabis consumption. Nevertheless, self-reported levels of intoxication were no different between users of cannabis flower or concentrate products.
Although results also showed that most neurobehavioral measures were not altered by short-term cannabis consumption, there were some notable exceptions. There was a negative linear effect with delayed verbal recall errors, suggesting poorer performance after cannabis use (F1, 203 = 32.31; P < .001).
On the other hand, investigators found a positive linear effect with inhibitory control and working memory, which actually suggests better performance after cannabis use. This finding, the researchers note, may be the result of a practice effect. Cannabis flower users performed better across all inhibitory control assessments.
The researchers also tested participants’ balance with their eyes open and closed. In the eyes-open condition, they found a trend toward impaired balance after cannabis use, though this normalized within an hour. When subjects closed their eyes, however, researchers observed a significant short-term increase in sway after cannabis use, which fell back to pre-use levels one hour after use (F1, 203 = 18.88; P < .001).
Of note, outcomes did not differ between groups according to the type of cannabis product consumed or its relative potency.
The study yielded several surprising findings, beginning with self-reported intoxication levels, which were not statistically significant between different cannabis flower and concentrate users, despite significantly different plasma THC levels between the two groups.
Dr. Hutchison explained that this may be the result of greater THC tolerance among concentrate users, THC saturation of cannabinoid receptors, or interindividual differences among users with respect to cannabis metabolism or sensitivity.
“I thought for sure that high-potency users would be much more compromised,” he said. “I guess it just goes to show we have a lot to learn about how these things work.”
Additionally, there were virtually no significant changes in acute performance after cannabis use, with the exception of delayed verbal recall. In fact, the most marked change observed in the study was the effect of cannabis on balance immediately after drug use, though these changes seemed to abate within an hour.
Nevertheless, the study highlights several potential public health implications of cannabis consumption, Dr. Hutchison added. “What happens when people with high blood concentrations decide to quit?” he asked. “Do they have trouble quitting? Do they have withdrawal symptoms?”
The long-term effects of cannabis use is another important question that still needs to be answered, he added.
Finally, Dr. Hutchison noted that, although the study showed little difference between users of cannabis flower and concentrates, study participants were all experienced users.
“There is certainly the potential for harm when a naive person uses cannabis concentrate,” he said. “Suddenly they have way more THC than they thought they were going to get, and that’s where a lot of people get into trouble with cannabis.”
Pitfalls and hurdles
In an accompanying editorial, Margaret Haney, PhD, of Columbia University Irving Medical Center, New York, explained that cannabis’ awkward position as simultaneously legal and illegal, medical and recreational, has hampered researchers’ ability to study its effects as comprehensively as they would otherwise like.
“With a federally illegal drug legalized in individual states, scientists constrained, and federal agencies somewhat silent, clinicians have none of the data that guide their decisions for other medications (eg, which indication, product, cannabinoid ratio, dose, or route of administration; what risks for individual patients [eg, pregnant, adolescent, psychiatric?]),” Dr. Haney wrote.
These pitfalls are compounded by the significant regulatory hurdles.
“The FDA is appropriately cautious about what it allows scientists to test in patients, and none of the products available in dispensaries or online have undergone the safety and manufacturing procedures needed for FDA approval,” she continued. “How then to conduct the studies so needed?”
Yet as Haney noted, giving cannabinoid researchers a Schedule I exemption may help address many of the barriers facing these scientists. Such a move, she said, would increase the number of randomized controlled trials being performed, “and thereby begin to breach the divide between the use of these products and empirical evidence.”
Dr. Hutchison has disclosed no relevant financial relationships. Dr. Haney disclosed funding from the US National Institute on Drug Abuse and from the Thompson Family Foundation Initiative. The study was funded by the NIH and Colorado Department of Public Health and Environment.
A version of this article originally appeared on Medscape.com.
Researchers have published one of the first studies to characterize the association between the consumption of legal cannabis and subsequent pharmacologic and neurobehavioral outcomes, with somewhat surprising results.
The study showed that, although cannabis consumption did not affect most short-term neurobehavioral measures, it delayed recall memory and impaired balance.
The investigation also showed that users of much more potent cannabis concentrates actually demonstrated similar or lower levels of subjective drug intoxication and short-term impairment than did their counterparts who used lower-potency forms of the cannabis flower.
“It does not appear that the potency being used matters that much,” senior investigator Kent E. Hutchison, PhD, said in an interview. “People seem to be titrating to a certain level of intoxication or a certain level of feeling high. And for some people that requires a lot of drug, and for other people not as much.”
added Dr. Hutchison, a professor of psychology and neuroscience at the University of Colorado Boulder.
The study was published online June 10 in JAMA Psychiatry.
Widespread availability, little research
Recreational cannabis is now legal in 11 states and the District of Columbia, while medical cannabis is legal in 33. However, despite growing popularity of cannabis, there is little research on its potential health and biobehavioral risks, largely because of federal restrictions on cannabis research.
Cannabis users typically consume various forms of the cannabis flower, which can boast concentrations of the psychoactive cannabinoid delta-9-tetrahydrocannabinol (THC) of up to 30%. However, use of concentrated forms of cannabis – which are made by extracting plant cannabinoids into a different form – is increasing.
Such formulations can boast THC concentrations as high as 90%. Nevertheless, data regarding the relative risks of these higher-strength products are limited.
Previous research has shown a variety of negative short-term and long-term neurobehavioral effects associated with cannabis use, including harmful cognitive and motor effects. Extended exposure to THC may also negatively affect brain regions that are associated with the control of coordinated movement, and create brain-activation deficits in motor control regions that persist well beyond the effects of short-term intoxication.
Despite such findings, Dr. Hutchison said the existing literature on the subject does not yield a real-world view of current cannabis use because it tends to focus on low-THC products that are increasingly less common in today’s legal market.
Given such shortcomings, the investigators wanted to address persistent questions surrounding the neurobehavioral effects of legal cannabis flower products (16% or 24% THC) and cannabis concentrate products (70% or 90% THC). In doing so, they examined three primary topics:
- The association between short-term use of these products and THC plasma levels; subjective intoxication; and mood, cognitive performance, and balance
- Differences in such associations between users of cannabis flower and concentrate products
- Potential variations in these associations by THC potency
High- versus low-potency varieties
The study included 133 individuals (aged 21-70 years), who were designated as either cannabis flower users or cannabis concentrate users. Participants had all used cannabis at least four times in the previous month with no adverse reaction and were not receiving treatment for a psychotic disorder or bipolar disorder.
Participants were randomly assigned to consume either higher-potency or lower-potency products that had been purchased from a local dispensary. Flower users were randomized to purchase 3 g of either a 16% THC or 24% THC product, while concentrate users were randomized to purchase 1g of either 70% THC or 90% THC.
Participants completed a series of four assessments, one at baseline and three others at a mobile laboratory. The mobile laboratory assessments occurred before, immediately after, and one hour after participants had all consumed their cannabis ad libitum.
Of the original cohort of 133 participants, 55 flower cannabis users (mean age, 28.8 years; 46% women) and 66 concentrate cannabis users (mean age, 28.3 years; 45% women) complied with the study’s instructions and had complete data.
The study’s primary outcome measures included plasma cannabinoids, subjective drug intoxication and mood, and neurobehavioral outcomes such as attention, memory, inhibitory control, and balance.
Mixed results
With respect to cannabis concentrations, results showed that users of concentrate exhibited higher levels of both plasma THC and the active metabolite of THC (11-hydroxy-delta9-THC) across all points than did their counterparts who used cannabis flower products.
Specifically, mean plasma THC levels were 1,016 ± 1,380 mcg/ml in concentrate users and 455±503 mcg/mL in flower users after ad libitum cannabis consumption. Nevertheless, self-reported levels of intoxication were no different between users of cannabis flower or concentrate products.
Although results also showed that most neurobehavioral measures were not altered by short-term cannabis consumption, there were some notable exceptions. There was a negative linear effect with delayed verbal recall errors, suggesting poorer performance after cannabis use (F1, 203 = 32.31; P < .001).
On the other hand, investigators found a positive linear effect with inhibitory control and working memory, which actually suggests better performance after cannabis use. This finding, the researchers note, may be the result of a practice effect. Cannabis flower users performed better across all inhibitory control assessments.
The researchers also tested participants’ balance with their eyes open and closed. In the eyes-open condition, they found a trend toward impaired balance after cannabis use, though this normalized within an hour. When subjects closed their eyes, however, researchers observed a significant short-term increase in sway after cannabis use, which fell back to pre-use levels one hour after use (F1, 203 = 18.88; P < .001).
Of note, outcomes did not differ between groups according to the type of cannabis product consumed or its relative potency.
The study yielded several surprising findings, beginning with self-reported intoxication levels, which were not statistically significant between different cannabis flower and concentrate users, despite significantly different plasma THC levels between the two groups.
Dr. Hutchison explained that this may be the result of greater THC tolerance among concentrate users, THC saturation of cannabinoid receptors, or interindividual differences among users with respect to cannabis metabolism or sensitivity.
“I thought for sure that high-potency users would be much more compromised,” he said. “I guess it just goes to show we have a lot to learn about how these things work.”
Additionally, there were virtually no significant changes in acute performance after cannabis use, with the exception of delayed verbal recall. In fact, the most marked change observed in the study was the effect of cannabis on balance immediately after drug use, though these changes seemed to abate within an hour.
Nevertheless, the study highlights several potential public health implications of cannabis consumption, Dr. Hutchison added. “What happens when people with high blood concentrations decide to quit?” he asked. “Do they have trouble quitting? Do they have withdrawal symptoms?”
The long-term effects of cannabis use is another important question that still needs to be answered, he added.
Finally, Dr. Hutchison noted that, although the study showed little difference between users of cannabis flower and concentrates, study participants were all experienced users.
“There is certainly the potential for harm when a naive person uses cannabis concentrate,” he said. “Suddenly they have way more THC than they thought they were going to get, and that’s where a lot of people get into trouble with cannabis.”
Pitfalls and hurdles
In an accompanying editorial, Margaret Haney, PhD, of Columbia University Irving Medical Center, New York, explained that cannabis’ awkward position as simultaneously legal and illegal, medical and recreational, has hampered researchers’ ability to study its effects as comprehensively as they would otherwise like.
“With a federally illegal drug legalized in individual states, scientists constrained, and federal agencies somewhat silent, clinicians have none of the data that guide their decisions for other medications (eg, which indication, product, cannabinoid ratio, dose, or route of administration; what risks for individual patients [eg, pregnant, adolescent, psychiatric?]),” Dr. Haney wrote.
These pitfalls are compounded by the significant regulatory hurdles.
“The FDA is appropriately cautious about what it allows scientists to test in patients, and none of the products available in dispensaries or online have undergone the safety and manufacturing procedures needed for FDA approval,” she continued. “How then to conduct the studies so needed?”
Yet as Haney noted, giving cannabinoid researchers a Schedule I exemption may help address many of the barriers facing these scientists. Such a move, she said, would increase the number of randomized controlled trials being performed, “and thereby begin to breach the divide between the use of these products and empirical evidence.”
Dr. Hutchison has disclosed no relevant financial relationships. Dr. Haney disclosed funding from the US National Institute on Drug Abuse and from the Thompson Family Foundation Initiative. The study was funded by the NIH and Colorado Department of Public Health and Environment.
A version of this article originally appeared on Medscape.com.
Researchers have published one of the first studies to characterize the association between the consumption of legal cannabis and subsequent pharmacologic and neurobehavioral outcomes, with somewhat surprising results.
The study showed that, although cannabis consumption did not affect most short-term neurobehavioral measures, it delayed recall memory and impaired balance.
The investigation also showed that users of much more potent cannabis concentrates actually demonstrated similar or lower levels of subjective drug intoxication and short-term impairment than did their counterparts who used lower-potency forms of the cannabis flower.
“It does not appear that the potency being used matters that much,” senior investigator Kent E. Hutchison, PhD, said in an interview. “People seem to be titrating to a certain level of intoxication or a certain level of feeling high. And for some people that requires a lot of drug, and for other people not as much.”
added Dr. Hutchison, a professor of psychology and neuroscience at the University of Colorado Boulder.
The study was published online June 10 in JAMA Psychiatry.
Widespread availability, little research
Recreational cannabis is now legal in 11 states and the District of Columbia, while medical cannabis is legal in 33. However, despite growing popularity of cannabis, there is little research on its potential health and biobehavioral risks, largely because of federal restrictions on cannabis research.
Cannabis users typically consume various forms of the cannabis flower, which can boast concentrations of the psychoactive cannabinoid delta-9-tetrahydrocannabinol (THC) of up to 30%. However, use of concentrated forms of cannabis – which are made by extracting plant cannabinoids into a different form – is increasing.
Such formulations can boast THC concentrations as high as 90%. Nevertheless, data regarding the relative risks of these higher-strength products are limited.
Previous research has shown a variety of negative short-term and long-term neurobehavioral effects associated with cannabis use, including harmful cognitive and motor effects. Extended exposure to THC may also negatively affect brain regions that are associated with the control of coordinated movement, and create brain-activation deficits in motor control regions that persist well beyond the effects of short-term intoxication.
Despite such findings, Dr. Hutchison said the existing literature on the subject does not yield a real-world view of current cannabis use because it tends to focus on low-THC products that are increasingly less common in today’s legal market.
Given such shortcomings, the investigators wanted to address persistent questions surrounding the neurobehavioral effects of legal cannabis flower products (16% or 24% THC) and cannabis concentrate products (70% or 90% THC). In doing so, they examined three primary topics:
- The association between short-term use of these products and THC plasma levels; subjective intoxication; and mood, cognitive performance, and balance
- Differences in such associations between users of cannabis flower and concentrate products
- Potential variations in these associations by THC potency
High- versus low-potency varieties
The study included 133 individuals (aged 21-70 years), who were designated as either cannabis flower users or cannabis concentrate users. Participants had all used cannabis at least four times in the previous month with no adverse reaction and were not receiving treatment for a psychotic disorder or bipolar disorder.
Participants were randomly assigned to consume either higher-potency or lower-potency products that had been purchased from a local dispensary. Flower users were randomized to purchase 3 g of either a 16% THC or 24% THC product, while concentrate users were randomized to purchase 1g of either 70% THC or 90% THC.
Participants completed a series of four assessments, one at baseline and three others at a mobile laboratory. The mobile laboratory assessments occurred before, immediately after, and one hour after participants had all consumed their cannabis ad libitum.
Of the original cohort of 133 participants, 55 flower cannabis users (mean age, 28.8 years; 46% women) and 66 concentrate cannabis users (mean age, 28.3 years; 45% women) complied with the study’s instructions and had complete data.
The study’s primary outcome measures included plasma cannabinoids, subjective drug intoxication and mood, and neurobehavioral outcomes such as attention, memory, inhibitory control, and balance.
Mixed results
With respect to cannabis concentrations, results showed that users of concentrate exhibited higher levels of both plasma THC and the active metabolite of THC (11-hydroxy-delta9-THC) across all points than did their counterparts who used cannabis flower products.
Specifically, mean plasma THC levels were 1,016 ± 1,380 mcg/ml in concentrate users and 455±503 mcg/mL in flower users after ad libitum cannabis consumption. Nevertheless, self-reported levels of intoxication were no different between users of cannabis flower or concentrate products.
Although results also showed that most neurobehavioral measures were not altered by short-term cannabis consumption, there were some notable exceptions. There was a negative linear effect with delayed verbal recall errors, suggesting poorer performance after cannabis use (F1, 203 = 32.31; P < .001).
On the other hand, investigators found a positive linear effect with inhibitory control and working memory, which actually suggests better performance after cannabis use. This finding, the researchers note, may be the result of a practice effect. Cannabis flower users performed better across all inhibitory control assessments.
The researchers also tested participants’ balance with their eyes open and closed. In the eyes-open condition, they found a trend toward impaired balance after cannabis use, though this normalized within an hour. When subjects closed their eyes, however, researchers observed a significant short-term increase in sway after cannabis use, which fell back to pre-use levels one hour after use (F1, 203 = 18.88; P < .001).
Of note, outcomes did not differ between groups according to the type of cannabis product consumed or its relative potency.
The study yielded several surprising findings, beginning with self-reported intoxication levels, which were not statistically significant between different cannabis flower and concentrate users, despite significantly different plasma THC levels between the two groups.
Dr. Hutchison explained that this may be the result of greater THC tolerance among concentrate users, THC saturation of cannabinoid receptors, or interindividual differences among users with respect to cannabis metabolism or sensitivity.
“I thought for sure that high-potency users would be much more compromised,” he said. “I guess it just goes to show we have a lot to learn about how these things work.”
Additionally, there were virtually no significant changes in acute performance after cannabis use, with the exception of delayed verbal recall. In fact, the most marked change observed in the study was the effect of cannabis on balance immediately after drug use, though these changes seemed to abate within an hour.
Nevertheless, the study highlights several potential public health implications of cannabis consumption, Dr. Hutchison added. “What happens when people with high blood concentrations decide to quit?” he asked. “Do they have trouble quitting? Do they have withdrawal symptoms?”
The long-term effects of cannabis use is another important question that still needs to be answered, he added.
Finally, Dr. Hutchison noted that, although the study showed little difference between users of cannabis flower and concentrates, study participants were all experienced users.
“There is certainly the potential for harm when a naive person uses cannabis concentrate,” he said. “Suddenly they have way more THC than they thought they were going to get, and that’s where a lot of people get into trouble with cannabis.”
Pitfalls and hurdles
In an accompanying editorial, Margaret Haney, PhD, of Columbia University Irving Medical Center, New York, explained that cannabis’ awkward position as simultaneously legal and illegal, medical and recreational, has hampered researchers’ ability to study its effects as comprehensively as they would otherwise like.
“With a federally illegal drug legalized in individual states, scientists constrained, and federal agencies somewhat silent, clinicians have none of the data that guide their decisions for other medications (eg, which indication, product, cannabinoid ratio, dose, or route of administration; what risks for individual patients [eg, pregnant, adolescent, psychiatric?]),” Dr. Haney wrote.
These pitfalls are compounded by the significant regulatory hurdles.
“The FDA is appropriately cautious about what it allows scientists to test in patients, and none of the products available in dispensaries or online have undergone the safety and manufacturing procedures needed for FDA approval,” she continued. “How then to conduct the studies so needed?”
Yet as Haney noted, giving cannabinoid researchers a Schedule I exemption may help address many of the barriers facing these scientists. Such a move, she said, would increase the number of randomized controlled trials being performed, “and thereby begin to breach the divide between the use of these products and empirical evidence.”
Dr. Hutchison has disclosed no relevant financial relationships. Dr. Haney disclosed funding from the US National Institute on Drug Abuse and from the Thompson Family Foundation Initiative. The study was funded by the NIH and Colorado Department of Public Health and Environment.
A version of this article originally appeared on Medscape.com.
FDA expands Dysport use for cerebral palsy–related spasticity
– for patients as young as 2 years and older, according to manufacturer Ipsen Biopharmaceuticals.
When Dysport (abobotulinumtoxinA) initially was approved for treating pediatric lower limb spasticity by the FDA in 2016, Ipsen was granted Orphan Drug exclusivity for children whose lower-limb spasticity was caused by cerebral palsy. In 2019, Dysport was approved by the FDA for treating of upper-limb spasticity in children 2 years older. But if that spasticity was caused by cerebral palsy, Dysport could be used to treat it only through Orphan Drug exclusivity granted to another manufacturer, according to an Ipsen press release.
“The proactive step to resolve the uncertainty created by the previous CP [cerebral palsy] carve out enables us as physicians to prescribe consistent therapy for pediatric patients experiencing both upper- and lower-limb spasticity,” Sarah Helen Evans, MD, division chief of rehabilitation medicine in the department of pediatrics at the Children’s Hospital of Philadelphia, said in the press release.
The most common adverse effects among children with lower-limb spasticity treated with Dysport were nasopharyngitis, cough, and pyrexia. Among children with upper-limb spasticity, the most common effects associated with Dysport treatment were upper respiratory tract infection and pharyngitis.
The press release also included a warning of the distant spread of the botulinum toxin from the area of injection hours to weeks afterward, causing symptoms including blurred vision, generalized muscle weakness, and swallowing and breathing difficulties that can be life threatening; there have been reports of death.
Suspected adverse effects can be reported to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
– for patients as young as 2 years and older, according to manufacturer Ipsen Biopharmaceuticals.
When Dysport (abobotulinumtoxinA) initially was approved for treating pediatric lower limb spasticity by the FDA in 2016, Ipsen was granted Orphan Drug exclusivity for children whose lower-limb spasticity was caused by cerebral palsy. In 2019, Dysport was approved by the FDA for treating of upper-limb spasticity in children 2 years older. But if that spasticity was caused by cerebral palsy, Dysport could be used to treat it only through Orphan Drug exclusivity granted to another manufacturer, according to an Ipsen press release.
“The proactive step to resolve the uncertainty created by the previous CP [cerebral palsy] carve out enables us as physicians to prescribe consistent therapy for pediatric patients experiencing both upper- and lower-limb spasticity,” Sarah Helen Evans, MD, division chief of rehabilitation medicine in the department of pediatrics at the Children’s Hospital of Philadelphia, said in the press release.
The most common adverse effects among children with lower-limb spasticity treated with Dysport were nasopharyngitis, cough, and pyrexia. Among children with upper-limb spasticity, the most common effects associated with Dysport treatment were upper respiratory tract infection and pharyngitis.
The press release also included a warning of the distant spread of the botulinum toxin from the area of injection hours to weeks afterward, causing symptoms including blurred vision, generalized muscle weakness, and swallowing and breathing difficulties that can be life threatening; there have been reports of death.
Suspected adverse effects can be reported to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
– for patients as young as 2 years and older, according to manufacturer Ipsen Biopharmaceuticals.
When Dysport (abobotulinumtoxinA) initially was approved for treating pediatric lower limb spasticity by the FDA in 2016, Ipsen was granted Orphan Drug exclusivity for children whose lower-limb spasticity was caused by cerebral palsy. In 2019, Dysport was approved by the FDA for treating of upper-limb spasticity in children 2 years older. But if that spasticity was caused by cerebral palsy, Dysport could be used to treat it only through Orphan Drug exclusivity granted to another manufacturer, according to an Ipsen press release.
“The proactive step to resolve the uncertainty created by the previous CP [cerebral palsy] carve out enables us as physicians to prescribe consistent therapy for pediatric patients experiencing both upper- and lower-limb spasticity,” Sarah Helen Evans, MD, division chief of rehabilitation medicine in the department of pediatrics at the Children’s Hospital of Philadelphia, said in the press release.
The most common adverse effects among children with lower-limb spasticity treated with Dysport were nasopharyngitis, cough, and pyrexia. Among children with upper-limb spasticity, the most common effects associated with Dysport treatment were upper respiratory tract infection and pharyngitis.
The press release also included a warning of the distant spread of the botulinum toxin from the area of injection hours to weeks afterward, causing symptoms including blurred vision, generalized muscle weakness, and swallowing and breathing difficulties that can be life threatening; there have been reports of death.
Suspected adverse effects can be reported to the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Influenza vaccine efficacy called undiminished in MS
, Jackie Nguyen reported at the virtual annual meeting of the Consortium of Multiple Sclerosis Centers (CMSC). She presented a systematic review and meta-analysis of nine published cohort studies including 417 MS patients and more than 500 healthy controls, all of whom received inactivated seasonal influenza vaccine.
The impetus for this project was a recognition that the great majority of the research on the impact of influenza vaccine in patients with MS has focused on safety and MS relapse rates. In contrast, the nine studies included in the meta-analysis contained data on influenza vaccine efficacy as reflected in the ability to mount an adequate immune response. This was defined in standard fashion either by seroconversion, which required at least a fourfold increase in antibody titers following vaccination, or seroprotection, with a postvaccination antihemagglutination immunoglobulin G titer of at least 40. The analysis included patients with MS irrespective of disease duration or severity or treatment regimen, noted Ms. Nguyen, a third-year medical student at Nova Southeastern University College of Allopathic Medicine in Davie, Fla.
The researchers found that there was no significant difference between patients with MS and healthy controls in the rates of an adequate immune response for influenza H1N1, H3N2, or influenza B virus. “The vaccine should thus continue to be recommended for MS patients, as the data shows it to be efficacious,” she said.
Her conclusion is consistent with guidance provided in the American Academy of Neurology’s 2019 practice guideline update on immunization in MS, highlighted elsewhere at CMSC 2020 in a presentation by Marijean Buhse, PhD, of Stony Brook University in New York.
The guideline, updated for the first time in 17 years, states that all MS patients should be advised to receive influenza vaccine annually: “With known risks of exacerbation and other morbidity with influenza infection and no identified risks of exacerbation with influenza vaccines, benefits of influenza vaccination outweigh the risks in most scenarios. The exception involves the relatively few MS patients having a specific contraindication to the influenza vaccine, such as a previous severe reaction, noted Dr. Buhse, who wasn’t involved in developing the evidence-based guidelines.
The available evidence indicates that some but not all disease-modifying therapies for MS reduce the effectiveness of vaccination against influenza.
According to the guideline, “it is possible” that persons with MS being treated with glatiramer acetate have a reduced likelihood of seroprotection from influenza vaccine, a conclusion the guidelines committee drew with “low confidence in the evidence.” Further, the guideline states that “it is probable” MS patients on fingolimod have a lower likelihood of obtaining seroprotection from influenza vaccine than patients not on the drug, with moderate confidence in the evidence. Also, it is deemed probable that patients with MS who are taking mitoxantrone have a reduced likelihood of response to influenza vaccination, compared with healthy controls. But it is probable that patients with MS who are receiving interferon-beta have no diminution in the likelihood of seroprotection. According to the guideline, there is insufficient evidence to say whether patients with MS who are on natalizumab, teriflunomide, or methotrexate have a diminished response to influenza vaccination.
Dr. Buhse noted that rituximab is off-label therapy for MS, so there are no data available regarding the likelihood of seroprotection in response to influenza vaccination in that setting. However, rituximab profoundly decreases the immunogenicity of influenza and pneumococcal vaccines in rheumatoid arthritis patients. It is therefore recommended that inactivated influenza vaccine be given to patients with MS at least 2 weeks prior to starting rituximab or 6 months after the last dose in order to optimize the humoral results. Ms. Nguyen reported having no financial conflicts regarding her presentation. Dr. Buhse reported having received honoraria from Genzyme and Biogen.
, Jackie Nguyen reported at the virtual annual meeting of the Consortium of Multiple Sclerosis Centers (CMSC). She presented a systematic review and meta-analysis of nine published cohort studies including 417 MS patients and more than 500 healthy controls, all of whom received inactivated seasonal influenza vaccine.
The impetus for this project was a recognition that the great majority of the research on the impact of influenza vaccine in patients with MS has focused on safety and MS relapse rates. In contrast, the nine studies included in the meta-analysis contained data on influenza vaccine efficacy as reflected in the ability to mount an adequate immune response. This was defined in standard fashion either by seroconversion, which required at least a fourfold increase in antibody titers following vaccination, or seroprotection, with a postvaccination antihemagglutination immunoglobulin G titer of at least 40. The analysis included patients with MS irrespective of disease duration or severity or treatment regimen, noted Ms. Nguyen, a third-year medical student at Nova Southeastern University College of Allopathic Medicine in Davie, Fla.
The researchers found that there was no significant difference between patients with MS and healthy controls in the rates of an adequate immune response for influenza H1N1, H3N2, or influenza B virus. “The vaccine should thus continue to be recommended for MS patients, as the data shows it to be efficacious,” she said.
Her conclusion is consistent with guidance provided in the American Academy of Neurology’s 2019 practice guideline update on immunization in MS, highlighted elsewhere at CMSC 2020 in a presentation by Marijean Buhse, PhD, of Stony Brook University in New York.
The guideline, updated for the first time in 17 years, states that all MS patients should be advised to receive influenza vaccine annually: “With known risks of exacerbation and other morbidity with influenza infection and no identified risks of exacerbation with influenza vaccines, benefits of influenza vaccination outweigh the risks in most scenarios. The exception involves the relatively few MS patients having a specific contraindication to the influenza vaccine, such as a previous severe reaction, noted Dr. Buhse, who wasn’t involved in developing the evidence-based guidelines.
The available evidence indicates that some but not all disease-modifying therapies for MS reduce the effectiveness of vaccination against influenza.
According to the guideline, “it is possible” that persons with MS being treated with glatiramer acetate have a reduced likelihood of seroprotection from influenza vaccine, a conclusion the guidelines committee drew with “low confidence in the evidence.” Further, the guideline states that “it is probable” MS patients on fingolimod have a lower likelihood of obtaining seroprotection from influenza vaccine than patients not on the drug, with moderate confidence in the evidence. Also, it is deemed probable that patients with MS who are taking mitoxantrone have a reduced likelihood of response to influenza vaccination, compared with healthy controls. But it is probable that patients with MS who are receiving interferon-beta have no diminution in the likelihood of seroprotection. According to the guideline, there is insufficient evidence to say whether patients with MS who are on natalizumab, teriflunomide, or methotrexate have a diminished response to influenza vaccination.
Dr. Buhse noted that rituximab is off-label therapy for MS, so there are no data available regarding the likelihood of seroprotection in response to influenza vaccination in that setting. However, rituximab profoundly decreases the immunogenicity of influenza and pneumococcal vaccines in rheumatoid arthritis patients. It is therefore recommended that inactivated influenza vaccine be given to patients with MS at least 2 weeks prior to starting rituximab or 6 months after the last dose in order to optimize the humoral results. Ms. Nguyen reported having no financial conflicts regarding her presentation. Dr. Buhse reported having received honoraria from Genzyme and Biogen.
, Jackie Nguyen reported at the virtual annual meeting of the Consortium of Multiple Sclerosis Centers (CMSC). She presented a systematic review and meta-analysis of nine published cohort studies including 417 MS patients and more than 500 healthy controls, all of whom received inactivated seasonal influenza vaccine.
The impetus for this project was a recognition that the great majority of the research on the impact of influenza vaccine in patients with MS has focused on safety and MS relapse rates. In contrast, the nine studies included in the meta-analysis contained data on influenza vaccine efficacy as reflected in the ability to mount an adequate immune response. This was defined in standard fashion either by seroconversion, which required at least a fourfold increase in antibody titers following vaccination, or seroprotection, with a postvaccination antihemagglutination immunoglobulin G titer of at least 40. The analysis included patients with MS irrespective of disease duration or severity or treatment regimen, noted Ms. Nguyen, a third-year medical student at Nova Southeastern University College of Allopathic Medicine in Davie, Fla.
The researchers found that there was no significant difference between patients with MS and healthy controls in the rates of an adequate immune response for influenza H1N1, H3N2, or influenza B virus. “The vaccine should thus continue to be recommended for MS patients, as the data shows it to be efficacious,” she said.
Her conclusion is consistent with guidance provided in the American Academy of Neurology’s 2019 practice guideline update on immunization in MS, highlighted elsewhere at CMSC 2020 in a presentation by Marijean Buhse, PhD, of Stony Brook University in New York.
The guideline, updated for the first time in 17 years, states that all MS patients should be advised to receive influenza vaccine annually: “With known risks of exacerbation and other morbidity with influenza infection and no identified risks of exacerbation with influenza vaccines, benefits of influenza vaccination outweigh the risks in most scenarios. The exception involves the relatively few MS patients having a specific contraindication to the influenza vaccine, such as a previous severe reaction, noted Dr. Buhse, who wasn’t involved in developing the evidence-based guidelines.
The available evidence indicates that some but not all disease-modifying therapies for MS reduce the effectiveness of vaccination against influenza.
According to the guideline, “it is possible” that persons with MS being treated with glatiramer acetate have a reduced likelihood of seroprotection from influenza vaccine, a conclusion the guidelines committee drew with “low confidence in the evidence.” Further, the guideline states that “it is probable” MS patients on fingolimod have a lower likelihood of obtaining seroprotection from influenza vaccine than patients not on the drug, with moderate confidence in the evidence. Also, it is deemed probable that patients with MS who are taking mitoxantrone have a reduced likelihood of response to influenza vaccination, compared with healthy controls. But it is probable that patients with MS who are receiving interferon-beta have no diminution in the likelihood of seroprotection. According to the guideline, there is insufficient evidence to say whether patients with MS who are on natalizumab, teriflunomide, or methotrexate have a diminished response to influenza vaccination.
Dr. Buhse noted that rituximab is off-label therapy for MS, so there are no data available regarding the likelihood of seroprotection in response to influenza vaccination in that setting. However, rituximab profoundly decreases the immunogenicity of influenza and pneumococcal vaccines in rheumatoid arthritis patients. It is therefore recommended that inactivated influenza vaccine be given to patients with MS at least 2 weeks prior to starting rituximab or 6 months after the last dose in order to optimize the humoral results. Ms. Nguyen reported having no financial conflicts regarding her presentation. Dr. Buhse reported having received honoraria from Genzyme and Biogen.
REPORTING FROM CMSC 2020
Healthy Aging Project-Brain: A Psychoeducational and Motivational Group for Older Veterans
With a rapidly growing older adult population, increased attention has been given to cognitive changes that occur with age, with a focus on optimizing the cognitive health of aging individuals.1 Given the absence of pharmaceutical treatments to prevent cognitive decline, there is an increased need for health care systems to offer alternative or behavioral interventions that can mitigate the effects of cognitive decline in aging.
Notably, many individuals are able to maintain or even improve cognitive functioning throughout their lifespan, with some research implicating health behaviors as an important factor for promoting brain health with age. Specifically, sleep, exercise, eating habits, social engagement, and cognitive stimulation have been linked to improved cognitive functioning.2-8 In addition to the potential benefits for brain health, there is evidence that greater investment in attaining health goals is associated with subjective reports of higher well-being, fewer mental health symptoms, lower physical health stresses, decreased caregiver burden, and increased functional independence linked with longer independent living.9 The latter has a substantial financial impact, such that the positive consequence of increased independence is likely staving off the need for admission to assisted living and adult family homes, which can be costly.
Despite the role of health behaviors in brain aging and overall health and functioning, research indicates that only a small number of older adults (12.8%) follow recommended guidelines for healthy lifestyle factors.10 Education has been identified as one factor associated with the likelihood of engaging in positive health behaviors, prompting the delivery of health-education interventions. Most psychoeducational interventions have traditionally focused on one aspect of behavior change at a time (eg, sleep); however, Gross and colleaguesconducted a meta-analysis of cognitive interventions and in addition to the overall positive benefits (effect size 0.38), they also found suggestive evidence that interventions that combined multiple training strategies were associated with larger training gains (P = .04) after adjusting for multiple comparisons.11 For example, Miller and colleagues found a significant improvement on both subjective and objective measures of memory following a multicomponent approach that combined training in memory skills, stress reduction, nutrition, and physical activity.12
In addition to the potential positive impacts of health behaviors on brain health, findings suggest that targeted emphasis on health behavior change may have the potential to stave off mild cognitiveimpairment (MCI) or dementia even if for a short time. Given the increasing prevalence rates of MCI with age (6.7% in adults aged 60-64 years, reaching 25.2% in adults aged 80-84 years13) and dementia (prevalence of MCI converting to dementia is 18-40%14), as well as the corresponding emotional, financial, and family-oriented consequences (eg, impact on the well-being of family caregivers), the need for behavioral interventions that seek to optimize brain health is becoming increasingly apparent.
More than 9 million veterans are now aged ≥ 65 years.15 In addition to representing nearly half of all veterans and a sizable portion of aging adults in the US, older veterans are at increased risk of frailty, mortality, and high rates of chronic medical/mental health conditions that can lead to accelerated cognitive aging.6-17 Together, these conditions highlight the importance of developing comprehensive psychoeducational and behavioral interventions in this population. To address this need, we developed a novel psychoeducation and behavior change group called the Healthy Aging Project-Brain (HAP-B, pronounced “happy”). The HAP-B intervention was designed to promote healthy brain aging by using empirically supported health behavior change strategies, including education, personalized goal setting, and community support. The primary aim of this project was to develop and implement an intervention that was feasible and acceptable (eg, could be implemented in our setting, was appropriate for a veteran population) and to determine any positive outcomes/preliminary effects on overall health and well-being.
Methods
We recruited veterans aged ≥ 50 years through primary care clinics and self-referrals via flyers in the US Department of Veterans Affairs (VA) Puget Sound Health Care System (VAPSHCS), Seattle Division hospital. We targeted the “worried well” and welcomed veterans with MCI and mental health diagnoses. Notably, if there were significant mental health and/or substance use concerns, we encouraged veterans to seek focused care and stabilization prior to or concurrent with group participation. Exclusion criteria included presence of suicidality/homicidality, untreated or unstable substance use disorder, or a diagnosis of dementia. Exclusion criteria were assessed by the referring health care providers (HCPs), when appropriate, and through a health record review. Group facilitators used their clinical judgment to monitor participants if they began experiencing more severe cognitive impairment or acute mental health concerns. Although we did not encounter any of these instances, facilitators were prepared to discuss any concerns with the veteran and their referring HCP. Participants sampled were from 1 of 5 groups offered between January 2018 and March 2019. A waiver from the institutional review board was obtained after meeting criteria for quality improvement/quality assurance (QI/QA) for this study.
Procedures
At the initial stages of development, our team conducted a needs assessment to identify health-related areas where HCPs felt veterans would benefit from additional education and support. The needs assessment was conducted across primary care, geriatric extended care, and the Geriatric Research, Education, and Clinical Center (GRECC) at VAPSHCS. Combining the needs assessment results with the available research base, we identified sleep, physical activity, social engagement, and cognitive stimulation as areas for focus. Notably, although nutrition has been identified as an important factor in cognitive aging, a diet and nutrition class was already available to older veterans at the Seattle VA; hence, we chose to limit overlap by not covering this topic in our group.
The group was offered on a quarterly basis as six 90-minute psychoeducational classes to allow time for didactics, discussion, and practice without overloading participants with information. Each group consisted of 4 to 9 veterans led by 2 cofacilitators. Group structure allowed for feedback and ideas from group members as well as accountability for engaging in behavior change. Cognitive functioning was not formally evaluated. Attendees were asked but not required to complete questionnaires before the classes began and again at completion. In addition at the completion of each group, feedback was collected from veterans and used to modify group content (Figure).
Two pilot groups were implemented in early and mid-2018 with iterative changes after each group. Then we revised the assessment battery and implemented the current version (v1.0), which was first offered in the fall of 2018 and was used with the final 3 groups. Noteworthy changes included weekly check-ins to assess use of health behavior logs and progress toward individual goals, additional pre-and postgroup measures, and in vivo skills practice relevant to the topic being discussed that day.
Each session began with a check-in, which included a review of daily logs and SMART (specific, measurable, attainable, relevant/realistic, and timebound) goals from the previous week.18 This allowed for praise/reinforcement of health behaviors as well as discussion of potential barriers. Second, an overview of research focusing on the relationship between aging, brain health, and the topic of the day was presented. As an example, in the discussion of social engagement, research was presented about the link between social isolation and cognitive decline; the indirect benefits of social support (eg, social support is linked to improved physical and mental health, which, in turn, is associated with less cognitive decline); and the direct benefits of social support (eg, high levels of emotional support are associated with better cognitive function) (Table 1).6
Next, facilitators reviewed skills and strategies to improve functioning in the topic of discussion. During the social engagement group, for example, facilitators discussed tips to improve social skills (eg, asking open-ended questions) and how to build social support into a daily routine (eg, scheduling weekly phone calls with family and friends). Following this discussion of skills, an activity was practiced, reinforcing learned material. During the social engagement group, veterans were invited to use small talk strategies with fellow group members. Finally, group sessions ended with each participant identifying a SMART goal for the coming week and troubleshooting potential barriers to success. SMART goals were kept broad, so veterans could choose a goal related to the topic discussed at the group that day (eg, scheduling a phone call with a friend twice in the coming week during the social engagement-focused group) or choose any other goal to focus on (eg, a sleep-related goal). Similarly, goals could change week to week, or could remain the same throughout the 6-week classes.
Measures
The questionnaires used for QI/QA analyses included the Satisfaction with Life Scale (SWLS); Geriatric Depression Scale-Short Form (GDS-S); Social Support Survey Instrument (SSSI); Pittsburg Sleep Quality Index (PSQI); Medical Outcomes Survey-Short Form (MOS-36 SF); and a self-efficacy scale (adapted from Huckans and colleagues for traumatic brain injury).19-24 Written feedback was collected at the end of the last group to assess perception of progress, self-perceived behavior change, what was helpful or unhelpful, and how likely the participants were to recommend the group to other veterans (0 to 3, very unlikely to very likely).
To promote consistency with other health and behavior change interventions at the VA, HAP-B used resources from the Whole Health model SMART goals. Research supports the use of self-monitoring techniques like SMART goals for behavior change.25
To facilitate skills practice and self-monitoring between classes, veterans were asked to complete 2 homework assignments. First, at the end of each group, each veteran identified a specific SMART goal to focus on and track in the coming week. Goals were unique to each veteran and allowed to change from week to week. Group discussion around SMART goals involved plans for how to address potential barriers; progress toward goals was discussed at the beginning of the following group. Second, veterans were asked to complete a worksheet used to track progress toward the weekly SMART goal and the specific health behaviors related to the 4 domains targeted by HAP-B. For example, when tracking sleep behaviors, veterans noted bedtime, waketime, number of times they woke up during the night, and length of daytime naps if applicable. Tracking logs were provided at the end of each class for personal purposes only. We asked veterans to rate themselves each week on whether they used the tracking sheet to monitor health behaviors; and how successful they were at accomplishing their previously identified SMART goal. We recorded responses on a 0 to 2 scale (0, not good; 1, fair; 2, good). This rating system was developed and implemented in later groups to promote self-monitoring, accountability, and discussion of potential barriers. However, due to the small sample that completed these ratings and the absence of objective corroborating data, these ratings were not included in the current analyses.
Every participant received a manual in binder format, which provided the didactic information for each group session, skills and strategies discussed in each session, and relevant resources in both the VA and community. For example, social engagement resources included information about volunteer opportunities, VA groups that focus on developing interpersonal skills, and recommendations from past group members on social events (eg, dance lessons at a senior center). We also developed a facilitator version of the manual in which we added comments and guidance on topics for discussion. Materials were developed with the goal of optimizing the ease of dissemination to other sites.
Results
Across the 5 groups, 31 veterans enrolled as participants and completed the initial intake measures, with an average of 6 participants per group (range 4-9). The majority (80%) attended at least 5 of the 6 classes. The mean
At the start of the class, the mean (SD) reports of participants were mild depressive symptoms 5.96 (3.8) on the GDS scale, moderate levels of self-efficacy 3.69 (0.5) on the self-efficacy scale, and moderate levels of satisfaction with life 18.08 (6.8) on the SWLS scale (Table 2). Data from 25 of 31 veterans who completed both pregroup and postgroup surveys were analyzed and paired samples t tests without corrections indicated a reduction in depressive symptoms (P = .01), improved self-efficacy (P = .08), and improved satisfaction with life (P = .03). There were no significant differences in self-reported sleep quality or perceived social support from pregroup to postgroup evaluations. Because the sample size was smaller for the MOS-36, which was not used until group 3, and the subscales are composed of few items each, we conducted exploratory analyses of the 8 MOS-36 subscales and found that well-being, physical functioning, role limitations due to physical and emotional functioning, and energy/fatigue significantly improved over time (Ps < .04).
Twenty-eight veterans provided written feedback following the final session. Qualitative feedback received at the completion of the group focused on participants’ desire for increased number of classes, longer sessions (eg, 2 participants recommended lengthening the group to 2 hours), and integrating mindfulness-based activities into each class. Participants rated themselves somewhat likely to very likely to recommend this group to other veterans (mean, 2.9 [SD, 0.4]).
Discussion
The ability and need to promote brain health with age is an emerging priority as our aging population grows. A growing body of evidence supports the role of health behaviors in healthy brain aging. Education and skills training in a group setting provides a supportive, cost-effective approach for increasing overall health in aging adults. Yet older adults are statistically less likely to engage in these behaviors on a regular basis. The current investigation provides preliminary support for a model of care that uses a comprehensive, experiential psychoeducational approach to facilitate behavior change in older adults. Our aim was to develop and implement an intervention that was feasible and acceptable to our older veterans and to determine any positive outcomes/preliminary effects on overall health and well-being.
Participants indicated that they enjoyed the group, learned new skills (per participant feedback and facilitator observation), and experienced improvements in mood, self-efficacy, and life satisfaction. Given the participants’ positive response to the group and its content, as well as continued referrals by HCPs to this group and low difficulty with ongoing recruitment, this program was deemed both feasible and acceptable in our veteran health care setting. Questions remain about the extent to which participants modified their health behaviors given that we did not collect objective measurements of behaviors (eg, time spent exercising), the duration of behavior change (ie, how long during and after the group were behaviors maintained), and the role of premorbid or concurrent characteristics that may moderate the effect of the intervention on health-related outcomes (eg, sleep quality, perceived social support, overall functioning, concurrent interventions, medications).
Strengths and Limitations
This study had a limited sample size and no control group. However, evidence of significant improvements in depressive symptoms, self-efficacy, and life satisfaction in the development groups without a control group is encouraging. This is particularly noteworthy given that older veterans as a group have higher rates of frailty and mortality than do other similarly aged counterparts.17An additional weakness is the absence of a brief cognitive assessment or other formal assessment as part of the inclusion/exclusion criteria. However, this program development project provides data from a realistic condition (recruited broadly and with few exclusions, offered in similar format as other VA classes), thus adding strength to the interpretation and possibly the generalizability of these findings.
Conclusions
Future directions include disseminating HAP-B materials and procedures across a variety of sites, both VA and non-VA. In line with this goal, we hope to increase sample size and sample diversity while optimizing protocol integrity during the exportation phase. With a greater sample size and power, we aim to examine the role of self-efficacy and other premorbid factors (eg, cognitive functioning at baseline) as mediators for observed changes in pre-/postmeasures and outcomes. We also hope to incorporate objective measures of behavior change, such as fitness trackers, heart rate/pulse monitors, and actigraphy for monitoring sleep. Finally, we are interested in conducting follow-up with past and future participants to detect changes that may occur with learning new skills following the completion of the group (eg, changes in sleep behavior that take time to take effect) and the extent to which participants continue to use the health behavior skills and strategies to maintain or enhance progress in behavioral goals. Finally, although this intervention was initially designed for use with older veterans receiving health care through the VA, we believe the concepts and work products described here can be used with older adults across a wide range of health care settings. Providers interested in trialing HAP-B at their local site are encouraged to contact the authors.
1. Jacobsen LA, Kent M, Lee M, Mather M. America’s aging population. Popul Bull. 2011;66(1):1-20.
2. Cappuccio FP, D’Elia L, Strazzullo P, Miller MA. Sleep duration and all-cause mortality: A systematic review and meta-analysis of prospective studies. Sleep. 2010;33(5):85-592. doi:10.1093/sleep/33.5.585
3. Kelly ME, Loughrey D, Lawlor BA, Robertson IH, Walsh C, Brennan S. The impact of exercise on the cognitive functioning of healthy older adults: a systematic review and meta-analysis. Ageing Res Rev. 2014;16:12-31. doi:10.1016/j.arr.2014.05.002
4. Middleton LE, Manini TM, Simonsick EM, et al. Activity energy expenditure and incident cognitive impairment in older adults. Arch Intern Med. 2011;171(14):1251-1257. doi:10.1001/archinternmed.2011.277
5. World Health Organization. Interventions on diet and physical activity: what works. https://www.who.int/dietphysicalactivity/whatworks/en/. Published 2009. Accessed June 19, 2020.
6. Seeman TE, Lusignolo TM, Albert M, Berkman L. Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur studies of successful aging. Health Psychol. 2001;20(4):243-255. doi:10.1037//0278-6133.20.4.243
7. La Rue A. Healthy brain aging: role of cognitive reserve, cognitive stimulation and cognitive exercises. Clin Geriatr Med. 2010;26(1):99-111. doi:10.1016/j.cger.2009.11.003
8. Salthouse TA, Berish DE, Miles JD. The role of cognitive stimulation on the relations between age and cognitive functioning. Psychol Aging. 2002;17(4):548-557. doi:10.1037//0882-7974.17.4.548
9. Wrosch C, Schulz R, Heckhausen J. Health stresses and depressive symptomatology in the elderly: the importance of health engagement control strategies. Health Psychol. 2002;21(4):340-348. doi:10.1037//0278-6133.21.4.340
10. Pronk NP, Anderson LH, Crain AL, et al. Meeting recommendations for multiple healthy lifestyle factors: prevalence, clustering, and predictors among adolescent, adult, and senior health plan members. Am J Prev Med. 2004;27(suppl 2):25-33. doi:10.1016/j.amepre.2004.04.022
11. Gross AL, Parisi JM, Spira AP, et al. Memory training interventions for older adults: a meta-analysis. Aging Ment Health. 2012;16(6):722-734. doi:10.1080/13607863.2012.667783
12. Miller KJ, Siddarth P, Gaines JM, et al. The memory fitness program: cognitive effects of a healthy aging intervention. Am J Geriat Psychiatry. 2012;20(6):514-523. doi:10.1097/JGP.0b013e318227f821
13. Petersen RC, Lopez O, Armstrong MJ, et al. Practice guideline update summary: mild cognitive impairment: report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology. 2018;90(3):126-135. doi:10.1212/WNL.0000000000004826
14. Gauthier S, Reisberg B, Zaudig M, et al. Mild cognitive impairment. Lancet. 2006;367(9518):1262-1270. doi:10.1016/S0140-6736(06)68542-5
15. US Department of Veteran Affairs, National Center for Veteran Analysis and Statistics.Veteran population. 2020. https://www.va.gov/vetdata/Veteran_Population.asp. Updated May 21, 2020 . Accessed June 17, 2020.
16. Eibner C, Krull H, Brown K, et al. Current and projected characteristics and unique healthcare needs of the patient population served by the Department of Veterans Affairs. RAND Health Q. 2016;5(4):13.
17. Orkaby AR, Nussbaum L, Ho Y, et al. The burden of frailty among U.S. Veterans and its association with mortality, 2002-2012. J Gerontol A Biol Med Sci. 2019;74(8):1257-1264. doi:10.1093/gerona/gly232
18. Doran GT. There’s a S.M.A.R.T. way to write management’s goals and objectives. Manag Rev. 1981;70(11):35-36.
19. Diener E, Emmons RA, Larsen RJ, Griffin S. The satisfaction with life scale. J Pers Assess. 1985;49(1):71-75. doi:10.1207/s15327752jpa4901-13
20. Sheikh JI, Yesavage JA. Geriatric Depression Scale (GDS): recent evidence and development of a shorter version. Clin Gerontol. 1986;5(1-2):165-173. doi:10.1300/J018v05n01_09
21. Sherbourne CD, Stewart AL. The MOS social support survey. Soc Sci Med. 1991;32(6):705-714. doi:10.1016/0277-9536(91)90150-b
22. Buysse DJ, Reynolds CF III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193-213. doi:10.1016/0165-1781(89)90047-4
23. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483.
24. Huckans M, Pavawalla S, Demadura T, et al. A pilot study examining effects of group-based cognitive strategy training treatment on self-reported cognitive problems, psychiatric symptoms, functioning, and compensatory strategy use in OIF/OEF combat veterans with persistent mild cognitive disorder and history of traumatic brain injury. J Rehabil Res Dev. 2010;47(1):43-60. doi:10.1682/jrrd.2009.02.0019
25. Pearson ES. Goal setting as a health behavior change strategy in overweight and obese adults: a systematic literature review examining intervention components. Patient Educ Couns. 2012;87(1):32-42. doi:10.1016/j.pec.2011.07.018
With a rapidly growing older adult population, increased attention has been given to cognitive changes that occur with age, with a focus on optimizing the cognitive health of aging individuals.1 Given the absence of pharmaceutical treatments to prevent cognitive decline, there is an increased need for health care systems to offer alternative or behavioral interventions that can mitigate the effects of cognitive decline in aging.
Notably, many individuals are able to maintain or even improve cognitive functioning throughout their lifespan, with some research implicating health behaviors as an important factor for promoting brain health with age. Specifically, sleep, exercise, eating habits, social engagement, and cognitive stimulation have been linked to improved cognitive functioning.2-8 In addition to the potential benefits for brain health, there is evidence that greater investment in attaining health goals is associated with subjective reports of higher well-being, fewer mental health symptoms, lower physical health stresses, decreased caregiver burden, and increased functional independence linked with longer independent living.9 The latter has a substantial financial impact, such that the positive consequence of increased independence is likely staving off the need for admission to assisted living and adult family homes, which can be costly.
Despite the role of health behaviors in brain aging and overall health and functioning, research indicates that only a small number of older adults (12.8%) follow recommended guidelines for healthy lifestyle factors.10 Education has been identified as one factor associated with the likelihood of engaging in positive health behaviors, prompting the delivery of health-education interventions. Most psychoeducational interventions have traditionally focused on one aspect of behavior change at a time (eg, sleep); however, Gross and colleaguesconducted a meta-analysis of cognitive interventions and in addition to the overall positive benefits (effect size 0.38), they also found suggestive evidence that interventions that combined multiple training strategies were associated with larger training gains (P = .04) after adjusting for multiple comparisons.11 For example, Miller and colleagues found a significant improvement on both subjective and objective measures of memory following a multicomponent approach that combined training in memory skills, stress reduction, nutrition, and physical activity.12
In addition to the potential positive impacts of health behaviors on brain health, findings suggest that targeted emphasis on health behavior change may have the potential to stave off mild cognitiveimpairment (MCI) or dementia even if for a short time. Given the increasing prevalence rates of MCI with age (6.7% in adults aged 60-64 years, reaching 25.2% in adults aged 80-84 years13) and dementia (prevalence of MCI converting to dementia is 18-40%14), as well as the corresponding emotional, financial, and family-oriented consequences (eg, impact on the well-being of family caregivers), the need for behavioral interventions that seek to optimize brain health is becoming increasingly apparent.
More than 9 million veterans are now aged ≥ 65 years.15 In addition to representing nearly half of all veterans and a sizable portion of aging adults in the US, older veterans are at increased risk of frailty, mortality, and high rates of chronic medical/mental health conditions that can lead to accelerated cognitive aging.6-17 Together, these conditions highlight the importance of developing comprehensive psychoeducational and behavioral interventions in this population. To address this need, we developed a novel psychoeducation and behavior change group called the Healthy Aging Project-Brain (HAP-B, pronounced “happy”). The HAP-B intervention was designed to promote healthy brain aging by using empirically supported health behavior change strategies, including education, personalized goal setting, and community support. The primary aim of this project was to develop and implement an intervention that was feasible and acceptable (eg, could be implemented in our setting, was appropriate for a veteran population) and to determine any positive outcomes/preliminary effects on overall health and well-being.
Methods
We recruited veterans aged ≥ 50 years through primary care clinics and self-referrals via flyers in the US Department of Veterans Affairs (VA) Puget Sound Health Care System (VAPSHCS), Seattle Division hospital. We targeted the “worried well” and welcomed veterans with MCI and mental health diagnoses. Notably, if there were significant mental health and/or substance use concerns, we encouraged veterans to seek focused care and stabilization prior to or concurrent with group participation. Exclusion criteria included presence of suicidality/homicidality, untreated or unstable substance use disorder, or a diagnosis of dementia. Exclusion criteria were assessed by the referring health care providers (HCPs), when appropriate, and through a health record review. Group facilitators used their clinical judgment to monitor participants if they began experiencing more severe cognitive impairment or acute mental health concerns. Although we did not encounter any of these instances, facilitators were prepared to discuss any concerns with the veteran and their referring HCP. Participants sampled were from 1 of 5 groups offered between January 2018 and March 2019. A waiver from the institutional review board was obtained after meeting criteria for quality improvement/quality assurance (QI/QA) for this study.
Procedures
At the initial stages of development, our team conducted a needs assessment to identify health-related areas where HCPs felt veterans would benefit from additional education and support. The needs assessment was conducted across primary care, geriatric extended care, and the Geriatric Research, Education, and Clinical Center (GRECC) at VAPSHCS. Combining the needs assessment results with the available research base, we identified sleep, physical activity, social engagement, and cognitive stimulation as areas for focus. Notably, although nutrition has been identified as an important factor in cognitive aging, a diet and nutrition class was already available to older veterans at the Seattle VA; hence, we chose to limit overlap by not covering this topic in our group.
The group was offered on a quarterly basis as six 90-minute psychoeducational classes to allow time for didactics, discussion, and practice without overloading participants with information. Each group consisted of 4 to 9 veterans led by 2 cofacilitators. Group structure allowed for feedback and ideas from group members as well as accountability for engaging in behavior change. Cognitive functioning was not formally evaluated. Attendees were asked but not required to complete questionnaires before the classes began and again at completion. In addition at the completion of each group, feedback was collected from veterans and used to modify group content (Figure).
Two pilot groups were implemented in early and mid-2018 with iterative changes after each group. Then we revised the assessment battery and implemented the current version (v1.0), which was first offered in the fall of 2018 and was used with the final 3 groups. Noteworthy changes included weekly check-ins to assess use of health behavior logs and progress toward individual goals, additional pre-and postgroup measures, and in vivo skills practice relevant to the topic being discussed that day.
Each session began with a check-in, which included a review of daily logs and SMART (specific, measurable, attainable, relevant/realistic, and timebound) goals from the previous week.18 This allowed for praise/reinforcement of health behaviors as well as discussion of potential barriers. Second, an overview of research focusing on the relationship between aging, brain health, and the topic of the day was presented. As an example, in the discussion of social engagement, research was presented about the link between social isolation and cognitive decline; the indirect benefits of social support (eg, social support is linked to improved physical and mental health, which, in turn, is associated with less cognitive decline); and the direct benefits of social support (eg, high levels of emotional support are associated with better cognitive function) (Table 1).6
Next, facilitators reviewed skills and strategies to improve functioning in the topic of discussion. During the social engagement group, for example, facilitators discussed tips to improve social skills (eg, asking open-ended questions) and how to build social support into a daily routine (eg, scheduling weekly phone calls with family and friends). Following this discussion of skills, an activity was practiced, reinforcing learned material. During the social engagement group, veterans were invited to use small talk strategies with fellow group members. Finally, group sessions ended with each participant identifying a SMART goal for the coming week and troubleshooting potential barriers to success. SMART goals were kept broad, so veterans could choose a goal related to the topic discussed at the group that day (eg, scheduling a phone call with a friend twice in the coming week during the social engagement-focused group) or choose any other goal to focus on (eg, a sleep-related goal). Similarly, goals could change week to week, or could remain the same throughout the 6-week classes.
Measures
The questionnaires used for QI/QA analyses included the Satisfaction with Life Scale (SWLS); Geriatric Depression Scale-Short Form (GDS-S); Social Support Survey Instrument (SSSI); Pittsburg Sleep Quality Index (PSQI); Medical Outcomes Survey-Short Form (MOS-36 SF); and a self-efficacy scale (adapted from Huckans and colleagues for traumatic brain injury).19-24 Written feedback was collected at the end of the last group to assess perception of progress, self-perceived behavior change, what was helpful or unhelpful, and how likely the participants were to recommend the group to other veterans (0 to 3, very unlikely to very likely).
To promote consistency with other health and behavior change interventions at the VA, HAP-B used resources from the Whole Health model SMART goals. Research supports the use of self-monitoring techniques like SMART goals for behavior change.25
To facilitate skills practice and self-monitoring between classes, veterans were asked to complete 2 homework assignments. First, at the end of each group, each veteran identified a specific SMART goal to focus on and track in the coming week. Goals were unique to each veteran and allowed to change from week to week. Group discussion around SMART goals involved plans for how to address potential barriers; progress toward goals was discussed at the beginning of the following group. Second, veterans were asked to complete a worksheet used to track progress toward the weekly SMART goal and the specific health behaviors related to the 4 domains targeted by HAP-B. For example, when tracking sleep behaviors, veterans noted bedtime, waketime, number of times they woke up during the night, and length of daytime naps if applicable. Tracking logs were provided at the end of each class for personal purposes only. We asked veterans to rate themselves each week on whether they used the tracking sheet to monitor health behaviors; and how successful they were at accomplishing their previously identified SMART goal. We recorded responses on a 0 to 2 scale (0, not good; 1, fair; 2, good). This rating system was developed and implemented in later groups to promote self-monitoring, accountability, and discussion of potential barriers. However, due to the small sample that completed these ratings and the absence of objective corroborating data, these ratings were not included in the current analyses.
Every participant received a manual in binder format, which provided the didactic information for each group session, skills and strategies discussed in each session, and relevant resources in both the VA and community. For example, social engagement resources included information about volunteer opportunities, VA groups that focus on developing interpersonal skills, and recommendations from past group members on social events (eg, dance lessons at a senior center). We also developed a facilitator version of the manual in which we added comments and guidance on topics for discussion. Materials were developed with the goal of optimizing the ease of dissemination to other sites.
Results
Across the 5 groups, 31 veterans enrolled as participants and completed the initial intake measures, with an average of 6 participants per group (range 4-9). The majority (80%) attended at least 5 of the 6 classes. The mean
At the start of the class, the mean (SD) reports of participants were mild depressive symptoms 5.96 (3.8) on the GDS scale, moderate levels of self-efficacy 3.69 (0.5) on the self-efficacy scale, and moderate levels of satisfaction with life 18.08 (6.8) on the SWLS scale (Table 2). Data from 25 of 31 veterans who completed both pregroup and postgroup surveys were analyzed and paired samples t tests without corrections indicated a reduction in depressive symptoms (P = .01), improved self-efficacy (P = .08), and improved satisfaction with life (P = .03). There were no significant differences in self-reported sleep quality or perceived social support from pregroup to postgroup evaluations. Because the sample size was smaller for the MOS-36, which was not used until group 3, and the subscales are composed of few items each, we conducted exploratory analyses of the 8 MOS-36 subscales and found that well-being, physical functioning, role limitations due to physical and emotional functioning, and energy/fatigue significantly improved over time (Ps < .04).
Twenty-eight veterans provided written feedback following the final session. Qualitative feedback received at the completion of the group focused on participants’ desire for increased number of classes, longer sessions (eg, 2 participants recommended lengthening the group to 2 hours), and integrating mindfulness-based activities into each class. Participants rated themselves somewhat likely to very likely to recommend this group to other veterans (mean, 2.9 [SD, 0.4]).
Discussion
The ability and need to promote brain health with age is an emerging priority as our aging population grows. A growing body of evidence supports the role of health behaviors in healthy brain aging. Education and skills training in a group setting provides a supportive, cost-effective approach for increasing overall health in aging adults. Yet older adults are statistically less likely to engage in these behaviors on a regular basis. The current investigation provides preliminary support for a model of care that uses a comprehensive, experiential psychoeducational approach to facilitate behavior change in older adults. Our aim was to develop and implement an intervention that was feasible and acceptable to our older veterans and to determine any positive outcomes/preliminary effects on overall health and well-being.
Participants indicated that they enjoyed the group, learned new skills (per participant feedback and facilitator observation), and experienced improvements in mood, self-efficacy, and life satisfaction. Given the participants’ positive response to the group and its content, as well as continued referrals by HCPs to this group and low difficulty with ongoing recruitment, this program was deemed both feasible and acceptable in our veteran health care setting. Questions remain about the extent to which participants modified their health behaviors given that we did not collect objective measurements of behaviors (eg, time spent exercising), the duration of behavior change (ie, how long during and after the group were behaviors maintained), and the role of premorbid or concurrent characteristics that may moderate the effect of the intervention on health-related outcomes (eg, sleep quality, perceived social support, overall functioning, concurrent interventions, medications).
Strengths and Limitations
This study had a limited sample size and no control group. However, evidence of significant improvements in depressive symptoms, self-efficacy, and life satisfaction in the development groups without a control group is encouraging. This is particularly noteworthy given that older veterans as a group have higher rates of frailty and mortality than do other similarly aged counterparts.17An additional weakness is the absence of a brief cognitive assessment or other formal assessment as part of the inclusion/exclusion criteria. However, this program development project provides data from a realistic condition (recruited broadly and with few exclusions, offered in similar format as other VA classes), thus adding strength to the interpretation and possibly the generalizability of these findings.
Conclusions
Future directions include disseminating HAP-B materials and procedures across a variety of sites, both VA and non-VA. In line with this goal, we hope to increase sample size and sample diversity while optimizing protocol integrity during the exportation phase. With a greater sample size and power, we aim to examine the role of self-efficacy and other premorbid factors (eg, cognitive functioning at baseline) as mediators for observed changes in pre-/postmeasures and outcomes. We also hope to incorporate objective measures of behavior change, such as fitness trackers, heart rate/pulse monitors, and actigraphy for monitoring sleep. Finally, we are interested in conducting follow-up with past and future participants to detect changes that may occur with learning new skills following the completion of the group (eg, changes in sleep behavior that take time to take effect) and the extent to which participants continue to use the health behavior skills and strategies to maintain or enhance progress in behavioral goals. Finally, although this intervention was initially designed for use with older veterans receiving health care through the VA, we believe the concepts and work products described here can be used with older adults across a wide range of health care settings. Providers interested in trialing HAP-B at their local site are encouraged to contact the authors.
With a rapidly growing older adult population, increased attention has been given to cognitive changes that occur with age, with a focus on optimizing the cognitive health of aging individuals.1 Given the absence of pharmaceutical treatments to prevent cognitive decline, there is an increased need for health care systems to offer alternative or behavioral interventions that can mitigate the effects of cognitive decline in aging.
Notably, many individuals are able to maintain or even improve cognitive functioning throughout their lifespan, with some research implicating health behaviors as an important factor for promoting brain health with age. Specifically, sleep, exercise, eating habits, social engagement, and cognitive stimulation have been linked to improved cognitive functioning.2-8 In addition to the potential benefits for brain health, there is evidence that greater investment in attaining health goals is associated with subjective reports of higher well-being, fewer mental health symptoms, lower physical health stresses, decreased caregiver burden, and increased functional independence linked with longer independent living.9 The latter has a substantial financial impact, such that the positive consequence of increased independence is likely staving off the need for admission to assisted living and adult family homes, which can be costly.
Despite the role of health behaviors in brain aging and overall health and functioning, research indicates that only a small number of older adults (12.8%) follow recommended guidelines for healthy lifestyle factors.10 Education has been identified as one factor associated with the likelihood of engaging in positive health behaviors, prompting the delivery of health-education interventions. Most psychoeducational interventions have traditionally focused on one aspect of behavior change at a time (eg, sleep); however, Gross and colleaguesconducted a meta-analysis of cognitive interventions and in addition to the overall positive benefits (effect size 0.38), they also found suggestive evidence that interventions that combined multiple training strategies were associated with larger training gains (P = .04) after adjusting for multiple comparisons.11 For example, Miller and colleagues found a significant improvement on both subjective and objective measures of memory following a multicomponent approach that combined training in memory skills, stress reduction, nutrition, and physical activity.12
In addition to the potential positive impacts of health behaviors on brain health, findings suggest that targeted emphasis on health behavior change may have the potential to stave off mild cognitiveimpairment (MCI) or dementia even if for a short time. Given the increasing prevalence rates of MCI with age (6.7% in adults aged 60-64 years, reaching 25.2% in adults aged 80-84 years13) and dementia (prevalence of MCI converting to dementia is 18-40%14), as well as the corresponding emotional, financial, and family-oriented consequences (eg, impact on the well-being of family caregivers), the need for behavioral interventions that seek to optimize brain health is becoming increasingly apparent.
More than 9 million veterans are now aged ≥ 65 years.15 In addition to representing nearly half of all veterans and a sizable portion of aging adults in the US, older veterans are at increased risk of frailty, mortality, and high rates of chronic medical/mental health conditions that can lead to accelerated cognitive aging.6-17 Together, these conditions highlight the importance of developing comprehensive psychoeducational and behavioral interventions in this population. To address this need, we developed a novel psychoeducation and behavior change group called the Healthy Aging Project-Brain (HAP-B, pronounced “happy”). The HAP-B intervention was designed to promote healthy brain aging by using empirically supported health behavior change strategies, including education, personalized goal setting, and community support. The primary aim of this project was to develop and implement an intervention that was feasible and acceptable (eg, could be implemented in our setting, was appropriate for a veteran population) and to determine any positive outcomes/preliminary effects on overall health and well-being.
Methods
We recruited veterans aged ≥ 50 years through primary care clinics and self-referrals via flyers in the US Department of Veterans Affairs (VA) Puget Sound Health Care System (VAPSHCS), Seattle Division hospital. We targeted the “worried well” and welcomed veterans with MCI and mental health diagnoses. Notably, if there were significant mental health and/or substance use concerns, we encouraged veterans to seek focused care and stabilization prior to or concurrent with group participation. Exclusion criteria included presence of suicidality/homicidality, untreated or unstable substance use disorder, or a diagnosis of dementia. Exclusion criteria were assessed by the referring health care providers (HCPs), when appropriate, and through a health record review. Group facilitators used their clinical judgment to monitor participants if they began experiencing more severe cognitive impairment or acute mental health concerns. Although we did not encounter any of these instances, facilitators were prepared to discuss any concerns with the veteran and their referring HCP. Participants sampled were from 1 of 5 groups offered between January 2018 and March 2019. A waiver from the institutional review board was obtained after meeting criteria for quality improvement/quality assurance (QI/QA) for this study.
Procedures
At the initial stages of development, our team conducted a needs assessment to identify health-related areas where HCPs felt veterans would benefit from additional education and support. The needs assessment was conducted across primary care, geriatric extended care, and the Geriatric Research, Education, and Clinical Center (GRECC) at VAPSHCS. Combining the needs assessment results with the available research base, we identified sleep, physical activity, social engagement, and cognitive stimulation as areas for focus. Notably, although nutrition has been identified as an important factor in cognitive aging, a diet and nutrition class was already available to older veterans at the Seattle VA; hence, we chose to limit overlap by not covering this topic in our group.
The group was offered on a quarterly basis as six 90-minute psychoeducational classes to allow time for didactics, discussion, and practice without overloading participants with information. Each group consisted of 4 to 9 veterans led by 2 cofacilitators. Group structure allowed for feedback and ideas from group members as well as accountability for engaging in behavior change. Cognitive functioning was not formally evaluated. Attendees were asked but not required to complete questionnaires before the classes began and again at completion. In addition at the completion of each group, feedback was collected from veterans and used to modify group content (Figure).
Two pilot groups were implemented in early and mid-2018 with iterative changes after each group. Then we revised the assessment battery and implemented the current version (v1.0), which was first offered in the fall of 2018 and was used with the final 3 groups. Noteworthy changes included weekly check-ins to assess use of health behavior logs and progress toward individual goals, additional pre-and postgroup measures, and in vivo skills practice relevant to the topic being discussed that day.
Each session began with a check-in, which included a review of daily logs and SMART (specific, measurable, attainable, relevant/realistic, and timebound) goals from the previous week.18 This allowed for praise/reinforcement of health behaviors as well as discussion of potential barriers. Second, an overview of research focusing on the relationship between aging, brain health, and the topic of the day was presented. As an example, in the discussion of social engagement, research was presented about the link between social isolation and cognitive decline; the indirect benefits of social support (eg, social support is linked to improved physical and mental health, which, in turn, is associated with less cognitive decline); and the direct benefits of social support (eg, high levels of emotional support are associated with better cognitive function) (Table 1).6
Next, facilitators reviewed skills and strategies to improve functioning in the topic of discussion. During the social engagement group, for example, facilitators discussed tips to improve social skills (eg, asking open-ended questions) and how to build social support into a daily routine (eg, scheduling weekly phone calls with family and friends). Following this discussion of skills, an activity was practiced, reinforcing learned material. During the social engagement group, veterans were invited to use small talk strategies with fellow group members. Finally, group sessions ended with each participant identifying a SMART goal for the coming week and troubleshooting potential barriers to success. SMART goals were kept broad, so veterans could choose a goal related to the topic discussed at the group that day (eg, scheduling a phone call with a friend twice in the coming week during the social engagement-focused group) or choose any other goal to focus on (eg, a sleep-related goal). Similarly, goals could change week to week, or could remain the same throughout the 6-week classes.
Measures
The questionnaires used for QI/QA analyses included the Satisfaction with Life Scale (SWLS); Geriatric Depression Scale-Short Form (GDS-S); Social Support Survey Instrument (SSSI); Pittsburg Sleep Quality Index (PSQI); Medical Outcomes Survey-Short Form (MOS-36 SF); and a self-efficacy scale (adapted from Huckans and colleagues for traumatic brain injury).19-24 Written feedback was collected at the end of the last group to assess perception of progress, self-perceived behavior change, what was helpful or unhelpful, and how likely the participants were to recommend the group to other veterans (0 to 3, very unlikely to very likely).
To promote consistency with other health and behavior change interventions at the VA, HAP-B used resources from the Whole Health model SMART goals. Research supports the use of self-monitoring techniques like SMART goals for behavior change.25
To facilitate skills practice and self-monitoring between classes, veterans were asked to complete 2 homework assignments. First, at the end of each group, each veteran identified a specific SMART goal to focus on and track in the coming week. Goals were unique to each veteran and allowed to change from week to week. Group discussion around SMART goals involved plans for how to address potential barriers; progress toward goals was discussed at the beginning of the following group. Second, veterans were asked to complete a worksheet used to track progress toward the weekly SMART goal and the specific health behaviors related to the 4 domains targeted by HAP-B. For example, when tracking sleep behaviors, veterans noted bedtime, waketime, number of times they woke up during the night, and length of daytime naps if applicable. Tracking logs were provided at the end of each class for personal purposes only. We asked veterans to rate themselves each week on whether they used the tracking sheet to monitor health behaviors; and how successful they were at accomplishing their previously identified SMART goal. We recorded responses on a 0 to 2 scale (0, not good; 1, fair; 2, good). This rating system was developed and implemented in later groups to promote self-monitoring, accountability, and discussion of potential barriers. However, due to the small sample that completed these ratings and the absence of objective corroborating data, these ratings were not included in the current analyses.
Every participant received a manual in binder format, which provided the didactic information for each group session, skills and strategies discussed in each session, and relevant resources in both the VA and community. For example, social engagement resources included information about volunteer opportunities, VA groups that focus on developing interpersonal skills, and recommendations from past group members on social events (eg, dance lessons at a senior center). We also developed a facilitator version of the manual in which we added comments and guidance on topics for discussion. Materials were developed with the goal of optimizing the ease of dissemination to other sites.
Results
Across the 5 groups, 31 veterans enrolled as participants and completed the initial intake measures, with an average of 6 participants per group (range 4-9). The majority (80%) attended at least 5 of the 6 classes. The mean
At the start of the class, the mean (SD) reports of participants were mild depressive symptoms 5.96 (3.8) on the GDS scale, moderate levels of self-efficacy 3.69 (0.5) on the self-efficacy scale, and moderate levels of satisfaction with life 18.08 (6.8) on the SWLS scale (Table 2). Data from 25 of 31 veterans who completed both pregroup and postgroup surveys were analyzed and paired samples t tests without corrections indicated a reduction in depressive symptoms (P = .01), improved self-efficacy (P = .08), and improved satisfaction with life (P = .03). There were no significant differences in self-reported sleep quality or perceived social support from pregroup to postgroup evaluations. Because the sample size was smaller for the MOS-36, which was not used until group 3, and the subscales are composed of few items each, we conducted exploratory analyses of the 8 MOS-36 subscales and found that well-being, physical functioning, role limitations due to physical and emotional functioning, and energy/fatigue significantly improved over time (Ps < .04).
Twenty-eight veterans provided written feedback following the final session. Qualitative feedback received at the completion of the group focused on participants’ desire for increased number of classes, longer sessions (eg, 2 participants recommended lengthening the group to 2 hours), and integrating mindfulness-based activities into each class. Participants rated themselves somewhat likely to very likely to recommend this group to other veterans (mean, 2.9 [SD, 0.4]).
Discussion
The ability and need to promote brain health with age is an emerging priority as our aging population grows. A growing body of evidence supports the role of health behaviors in healthy brain aging. Education and skills training in a group setting provides a supportive, cost-effective approach for increasing overall health in aging adults. Yet older adults are statistically less likely to engage in these behaviors on a regular basis. The current investigation provides preliminary support for a model of care that uses a comprehensive, experiential psychoeducational approach to facilitate behavior change in older adults. Our aim was to develop and implement an intervention that was feasible and acceptable to our older veterans and to determine any positive outcomes/preliminary effects on overall health and well-being.
Participants indicated that they enjoyed the group, learned new skills (per participant feedback and facilitator observation), and experienced improvements in mood, self-efficacy, and life satisfaction. Given the participants’ positive response to the group and its content, as well as continued referrals by HCPs to this group and low difficulty with ongoing recruitment, this program was deemed both feasible and acceptable in our veteran health care setting. Questions remain about the extent to which participants modified their health behaviors given that we did not collect objective measurements of behaviors (eg, time spent exercising), the duration of behavior change (ie, how long during and after the group were behaviors maintained), and the role of premorbid or concurrent characteristics that may moderate the effect of the intervention on health-related outcomes (eg, sleep quality, perceived social support, overall functioning, concurrent interventions, medications).
Strengths and Limitations
This study had a limited sample size and no control group. However, evidence of significant improvements in depressive symptoms, self-efficacy, and life satisfaction in the development groups without a control group is encouraging. This is particularly noteworthy given that older veterans as a group have higher rates of frailty and mortality than do other similarly aged counterparts.17An additional weakness is the absence of a brief cognitive assessment or other formal assessment as part of the inclusion/exclusion criteria. However, this program development project provides data from a realistic condition (recruited broadly and with few exclusions, offered in similar format as other VA classes), thus adding strength to the interpretation and possibly the generalizability of these findings.
Conclusions
Future directions include disseminating HAP-B materials and procedures across a variety of sites, both VA and non-VA. In line with this goal, we hope to increase sample size and sample diversity while optimizing protocol integrity during the exportation phase. With a greater sample size and power, we aim to examine the role of self-efficacy and other premorbid factors (eg, cognitive functioning at baseline) as mediators for observed changes in pre-/postmeasures and outcomes. We also hope to incorporate objective measures of behavior change, such as fitness trackers, heart rate/pulse monitors, and actigraphy for monitoring sleep. Finally, we are interested in conducting follow-up with past and future participants to detect changes that may occur with learning new skills following the completion of the group (eg, changes in sleep behavior that take time to take effect) and the extent to which participants continue to use the health behavior skills and strategies to maintain or enhance progress in behavioral goals. Finally, although this intervention was initially designed for use with older veterans receiving health care through the VA, we believe the concepts and work products described here can be used with older adults across a wide range of health care settings. Providers interested in trialing HAP-B at their local site are encouraged to contact the authors.
1. Jacobsen LA, Kent M, Lee M, Mather M. America’s aging population. Popul Bull. 2011;66(1):1-20.
2. Cappuccio FP, D’Elia L, Strazzullo P, Miller MA. Sleep duration and all-cause mortality: A systematic review and meta-analysis of prospective studies. Sleep. 2010;33(5):85-592. doi:10.1093/sleep/33.5.585
3. Kelly ME, Loughrey D, Lawlor BA, Robertson IH, Walsh C, Brennan S. The impact of exercise on the cognitive functioning of healthy older adults: a systematic review and meta-analysis. Ageing Res Rev. 2014;16:12-31. doi:10.1016/j.arr.2014.05.002
4. Middleton LE, Manini TM, Simonsick EM, et al. Activity energy expenditure and incident cognitive impairment in older adults. Arch Intern Med. 2011;171(14):1251-1257. doi:10.1001/archinternmed.2011.277
5. World Health Organization. Interventions on diet and physical activity: what works. https://www.who.int/dietphysicalactivity/whatworks/en/. Published 2009. Accessed June 19, 2020.
6. Seeman TE, Lusignolo TM, Albert M, Berkman L. Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur studies of successful aging. Health Psychol. 2001;20(4):243-255. doi:10.1037//0278-6133.20.4.243
7. La Rue A. Healthy brain aging: role of cognitive reserve, cognitive stimulation and cognitive exercises. Clin Geriatr Med. 2010;26(1):99-111. doi:10.1016/j.cger.2009.11.003
8. Salthouse TA, Berish DE, Miles JD. The role of cognitive stimulation on the relations between age and cognitive functioning. Psychol Aging. 2002;17(4):548-557. doi:10.1037//0882-7974.17.4.548
9. Wrosch C, Schulz R, Heckhausen J. Health stresses and depressive symptomatology in the elderly: the importance of health engagement control strategies. Health Psychol. 2002;21(4):340-348. doi:10.1037//0278-6133.21.4.340
10. Pronk NP, Anderson LH, Crain AL, et al. Meeting recommendations for multiple healthy lifestyle factors: prevalence, clustering, and predictors among adolescent, adult, and senior health plan members. Am J Prev Med. 2004;27(suppl 2):25-33. doi:10.1016/j.amepre.2004.04.022
11. Gross AL, Parisi JM, Spira AP, et al. Memory training interventions for older adults: a meta-analysis. Aging Ment Health. 2012;16(6):722-734. doi:10.1080/13607863.2012.667783
12. Miller KJ, Siddarth P, Gaines JM, et al. The memory fitness program: cognitive effects of a healthy aging intervention. Am J Geriat Psychiatry. 2012;20(6):514-523. doi:10.1097/JGP.0b013e318227f821
13. Petersen RC, Lopez O, Armstrong MJ, et al. Practice guideline update summary: mild cognitive impairment: report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology. 2018;90(3):126-135. doi:10.1212/WNL.0000000000004826
14. Gauthier S, Reisberg B, Zaudig M, et al. Mild cognitive impairment. Lancet. 2006;367(9518):1262-1270. doi:10.1016/S0140-6736(06)68542-5
15. US Department of Veteran Affairs, National Center for Veteran Analysis and Statistics.Veteran population. 2020. https://www.va.gov/vetdata/Veteran_Population.asp. Updated May 21, 2020 . Accessed June 17, 2020.
16. Eibner C, Krull H, Brown K, et al. Current and projected characteristics and unique healthcare needs of the patient population served by the Department of Veterans Affairs. RAND Health Q. 2016;5(4):13.
17. Orkaby AR, Nussbaum L, Ho Y, et al. The burden of frailty among U.S. Veterans and its association with mortality, 2002-2012. J Gerontol A Biol Med Sci. 2019;74(8):1257-1264. doi:10.1093/gerona/gly232
18. Doran GT. There’s a S.M.A.R.T. way to write management’s goals and objectives. Manag Rev. 1981;70(11):35-36.
19. Diener E, Emmons RA, Larsen RJ, Griffin S. The satisfaction with life scale. J Pers Assess. 1985;49(1):71-75. doi:10.1207/s15327752jpa4901-13
20. Sheikh JI, Yesavage JA. Geriatric Depression Scale (GDS): recent evidence and development of a shorter version. Clin Gerontol. 1986;5(1-2):165-173. doi:10.1300/J018v05n01_09
21. Sherbourne CD, Stewart AL. The MOS social support survey. Soc Sci Med. 1991;32(6):705-714. doi:10.1016/0277-9536(91)90150-b
22. Buysse DJ, Reynolds CF III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193-213. doi:10.1016/0165-1781(89)90047-4
23. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483.
24. Huckans M, Pavawalla S, Demadura T, et al. A pilot study examining effects of group-based cognitive strategy training treatment on self-reported cognitive problems, psychiatric symptoms, functioning, and compensatory strategy use in OIF/OEF combat veterans with persistent mild cognitive disorder and history of traumatic brain injury. J Rehabil Res Dev. 2010;47(1):43-60. doi:10.1682/jrrd.2009.02.0019
25. Pearson ES. Goal setting as a health behavior change strategy in overweight and obese adults: a systematic literature review examining intervention components. Patient Educ Couns. 2012;87(1):32-42. doi:10.1016/j.pec.2011.07.018
1. Jacobsen LA, Kent M, Lee M, Mather M. America’s aging population. Popul Bull. 2011;66(1):1-20.
2. Cappuccio FP, D’Elia L, Strazzullo P, Miller MA. Sleep duration and all-cause mortality: A systematic review and meta-analysis of prospective studies. Sleep. 2010;33(5):85-592. doi:10.1093/sleep/33.5.585
3. Kelly ME, Loughrey D, Lawlor BA, Robertson IH, Walsh C, Brennan S. The impact of exercise on the cognitive functioning of healthy older adults: a systematic review and meta-analysis. Ageing Res Rev. 2014;16:12-31. doi:10.1016/j.arr.2014.05.002
4. Middleton LE, Manini TM, Simonsick EM, et al. Activity energy expenditure and incident cognitive impairment in older adults. Arch Intern Med. 2011;171(14):1251-1257. doi:10.1001/archinternmed.2011.277
5. World Health Organization. Interventions on diet and physical activity: what works. https://www.who.int/dietphysicalactivity/whatworks/en/. Published 2009. Accessed June 19, 2020.
6. Seeman TE, Lusignolo TM, Albert M, Berkman L. Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur studies of successful aging. Health Psychol. 2001;20(4):243-255. doi:10.1037//0278-6133.20.4.243
7. La Rue A. Healthy brain aging: role of cognitive reserve, cognitive stimulation and cognitive exercises. Clin Geriatr Med. 2010;26(1):99-111. doi:10.1016/j.cger.2009.11.003
8. Salthouse TA, Berish DE, Miles JD. The role of cognitive stimulation on the relations between age and cognitive functioning. Psychol Aging. 2002;17(4):548-557. doi:10.1037//0882-7974.17.4.548
9. Wrosch C, Schulz R, Heckhausen J. Health stresses and depressive symptomatology in the elderly: the importance of health engagement control strategies. Health Psychol. 2002;21(4):340-348. doi:10.1037//0278-6133.21.4.340
10. Pronk NP, Anderson LH, Crain AL, et al. Meeting recommendations for multiple healthy lifestyle factors: prevalence, clustering, and predictors among adolescent, adult, and senior health plan members. Am J Prev Med. 2004;27(suppl 2):25-33. doi:10.1016/j.amepre.2004.04.022
11. Gross AL, Parisi JM, Spira AP, et al. Memory training interventions for older adults: a meta-analysis. Aging Ment Health. 2012;16(6):722-734. doi:10.1080/13607863.2012.667783
12. Miller KJ, Siddarth P, Gaines JM, et al. The memory fitness program: cognitive effects of a healthy aging intervention. Am J Geriat Psychiatry. 2012;20(6):514-523. doi:10.1097/JGP.0b013e318227f821
13. Petersen RC, Lopez O, Armstrong MJ, et al. Practice guideline update summary: mild cognitive impairment: report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology. 2018;90(3):126-135. doi:10.1212/WNL.0000000000004826
14. Gauthier S, Reisberg B, Zaudig M, et al. Mild cognitive impairment. Lancet. 2006;367(9518):1262-1270. doi:10.1016/S0140-6736(06)68542-5
15. US Department of Veteran Affairs, National Center for Veteran Analysis and Statistics.Veteran population. 2020. https://www.va.gov/vetdata/Veteran_Population.asp. Updated May 21, 2020 . Accessed June 17, 2020.
16. Eibner C, Krull H, Brown K, et al. Current and projected characteristics and unique healthcare needs of the patient population served by the Department of Veterans Affairs. RAND Health Q. 2016;5(4):13.
17. Orkaby AR, Nussbaum L, Ho Y, et al. The burden of frailty among U.S. Veterans and its association with mortality, 2002-2012. J Gerontol A Biol Med Sci. 2019;74(8):1257-1264. doi:10.1093/gerona/gly232
18. Doran GT. There’s a S.M.A.R.T. way to write management’s goals and objectives. Manag Rev. 1981;70(11):35-36.
19. Diener E, Emmons RA, Larsen RJ, Griffin S. The satisfaction with life scale. J Pers Assess. 1985;49(1):71-75. doi:10.1207/s15327752jpa4901-13
20. Sheikh JI, Yesavage JA. Geriatric Depression Scale (GDS): recent evidence and development of a shorter version. Clin Gerontol. 1986;5(1-2):165-173. doi:10.1300/J018v05n01_09
21. Sherbourne CD, Stewart AL. The MOS social support survey. Soc Sci Med. 1991;32(6):705-714. doi:10.1016/0277-9536(91)90150-b
22. Buysse DJ, Reynolds CF III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28(2):193-213. doi:10.1016/0165-1781(89)90047-4
23. Ware JE Jr, Sherbourne CD. The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med Care. 1992;30(6):473-483.
24. Huckans M, Pavawalla S, Demadura T, et al. A pilot study examining effects of group-based cognitive strategy training treatment on self-reported cognitive problems, psychiatric symptoms, functioning, and compensatory strategy use in OIF/OEF combat veterans with persistent mild cognitive disorder and history of traumatic brain injury. J Rehabil Res Dev. 2010;47(1):43-60. doi:10.1682/jrrd.2009.02.0019
25. Pearson ES. Goal setting as a health behavior change strategy in overweight and obese adults: a systematic literature review examining intervention components. Patient Educ Couns. 2012;87(1):32-42. doi:10.1016/j.pec.2011.07.018
Does moderate drinking slow cognitive decline?
new research suggests. However, at least one expert urges caution in interpreting the findings.
Investigators found that consuming 10-14 alcoholic drinks per week had the strongest cognitive benefit. The findings “add more weight” to the growing body of research identifying beneficial cognitive effects of moderate alcohol consumption, said lead author, Ruiyuan Zhang, MD, of the department of epidemiology and biostatistics at the University of Georgia, Athens. However, Dr. Zhang emphasized that nondrinkers should not take up drinking to protect brain function, as alcohol can have negative effects.
The study was published online in JAMA Network Open.
Slower cognitive decline
The observational study was a secondary analysis of data from the Health and Retirement Study, a nationally representative U.S. survey of middle-aged and older adults. The survey, which began in 1992, is conducted every 2 years and collects health and economic data.
The current analysis used data from 1996 to 2008 and included information from individuals who participated in at least three surveys. The study included 19,887 participants, with a mean age 61.8 years. Most (60.1%) were women and white (85.2%). Mean follow-up was 9.1 years.
Researchers measured cognitive domains of mental status, word recall, and vocabulary. They also calculated a total cognition score, with higher scores indicating better cognitive abilities.
For each cognitive function measure, researchers categorized participants into a consistently low–trajectory group in which cognitive test scores from baseline through follow-up were consistently low or a consistently high–trajectory group, where cognitive test scores from baseline through follow-up were consistently high.
Based on self-reports, the investigators categorized participants as never drinkers (41.8%), former drinkers (39.5%), or current drinkers (18.7%). For current drinkers, researchers determined the number of drinking days per week and number of drinks per day. They further categorized these participants as low to moderate drinkers or heavy drinkers.
One drink was defined as a 12-ounce bottle of beer, a 5-ounce glass of wine, or a 1.5-ounce shot of spirits, said Dr. Zhang.
Women who consumed 8 or more drinks per week and men who drank 15 or more drinks per week were considered heavy drinkers. Other current drinkers were deemed low to moderate drinkers. Most current drinkers (85.2%) were low to moderate drinkers.
Other covariates included age, sex, race/ethnicity, years of education, marital status, tobacco smoking status, and body mass index.
Results showed moderate drinking was associated with relatively high cognitive test scores. After controlling for all covariates, compared with never drinkers, current low to moderate drinkers were significantly less likely to have consistently low trajectories for total cognitive score (odds ratio, 0.66; 95% confidence interval, 0.59-0.74), mental status (OR, 0.71; 95% CI, 0.63-0.81), word recall (OR, 0.74; 95% CI, 0.69-0.80), and vocabulary (OR, 0.64; 95% CI, 0.56-0.74) (all P < .001).
Former drinkers also had better cognitive outcomes for all cognitive domains. Heavy drinkers had lower odds of being in the consistently low trajectory group only for the vocabulary test.
Heavy drinking ‘risky’
Because few participants were deemed to be heavy drinkers, the power to identify an association between heavy drinking and cognitive function was limited. Dr. Zhang acknowledged, though he noted that heavy drinking is “risky.”
“We found that, after the drinking dosage passes the moderate level, the risk of low cognitive function increases very fast, which indicates that heavy drinking may harm cognitive function.” Limiting alcohol consumption “is still very important,” he said.
The associations of alcohol and cognitive functions differed by race/ethnicity. Low to moderate drinking was significantly associated with a lower odds of having a consistently low trajectory for all four cognitive function measures only among white participants.
A possible reason for this is that the study had so few African Americans (who made up only 14.8% of the sample), which limited the ability to identify relationships between alcohol intake and cognitive function, said Dr. Zhang. “Another reason is that the sensitivity to alcohol may be different between white and African American subjects.”
There was a significant U-shaped association between weekly amounts of alcohol and the odds of being in the consistently low–trajectory group for all cognitive functions. Depending on the function tested, the optimal number of weekly drinks ranged from 10-14.
Dr. Zhang noted that, when women were examined separately, alcohol consumption had a significant U-shaped relationship only with word recall, with the optimal dosage being around eight drinks.
U-shaped relationship an ‘important finding’
The U-shaped relationship is “an important finding,” said Dr. Zhang. “It shows that the human body may act differently to low and high doses of alcohol. Knowing why and how this happens is very important as it would help us understand how alcohol affects the function of the human body.”
Sensitivity analyses among participants with no chronic diseases showed the U-shaped association was still significant for scores of total word recall and vocabulary, but not for mental status or total cognition score.
The authors noted that 77.2% of participants had at least one chronic disease. They maintained that the association between alcohol consumption and cognitive function may be applicable both to healthy people and to those with a chronic disease.
The study also found that low to moderate drinkers had slower rates of cognitive decline over time for all cognition domains.
Although the mechanisms underlying the cognitive benefits of alcohol consumption are unclear, the authors believe it may be via cerebrovascular and cardiovascular pathways.
Alcohol may increase levels of brain-derived neurotrophic factor, a key regulator of neuronal plasticity and development in the dorsal striatum, they noted.
Balancing act
However, there’s also evidence that drinking, especially heavy drinking, increases the risk of hypertension, stroke, liver damage, and some cancers. “We think the role of alcohol drinking in cognitive function may be a balance of its beneficial and harmful effects on the cardiovascular system,” said Dr. Zhang.
“For the low to moderate drinker, the beneficial effects may outweigh the harmful effects on the small blood vessels in the brain. In this way, it could preserve cognition,” he added.
Dr. Zhang also noted that the study focused on middle-aged and older adults. “We can’t say whether or not moderate alcohol could benefit younger people” because they may have different characteristics, he said.
The findings of other studies examining the effects of alcohol on cognitive function are mixed. While studies have identified a beneficial effect, others have uncovered no, minimal, or adverse effects. This could be due to the use of different tests of cognitive function or different study populations, said Dr. Zhang.
A limitation of the current study was that assessment of alcohol consumption was based on self-report, which might have introduced recall bias. In addition, because individuals tend to underestimate their alcohol consumption, heavy drinkers could be misclassified as low to moderate drinkers, and low to moderate drinkers as former drinkers.
“This may make our study underestimate the association between low to moderate drinking and cognitive function,” said Dr. Zhang. In addition, alcohol consumption tended to change with time, and this change may be associated with other factors that led to changes in cognitive function, the authors noted.
Interpret with caution
Commenting on the study, Brent P. Forester, MD, chief of the Center of Excellence in Geriatric Psychiatry at McLean Hospital in Belmont, Mass., associate professor of psychiatry at Harvard Medical School, Boston, and a member of the American Psychiatric Association Council on Geriatric Psychiatry, said he views the study with some trepidation.
“As a clinician taking care of older adults, I would be very cautious about overinterpreting the beneficial effects of alcohol before we understand the mechanism better,” he said.
He noted that all of the risk factors associated with heart attack and stroke are also risk factors for Alzheimer’s disease and cognitive decline more broadly. “One of the issues here is how in the world does alcohol reduce cardiovascular and cerebrovascular risks, if you know it increases the risk of hypertension and stroke, regardless of dose.”
With regard to the possible impact of alcohol on brain-derived neurotrophic factor, Dr. Forester said, “it’s an interesting idea” but the actual mechanism is still unclear.
Even with dietary studies, such as those on the Mediterranean diet that include red wine, showing cognitive benefit, Dr. Forester said he’s still concerned about the adverse effects of alcohol on older people. These can include falls and sleep disturbances in addition to cognitive issues, and these effects can increase with age.
He was somewhat surprised at the level of alcohol that the study determined was beneficial. “Essentially, what they’re saying here is that, for men, it’s two drinks a day.” This could be “problematic” as two drinks per day can quickly escalate as individuals build tolerance.
He also pointed out that the study does not determine cause and effect, noting that it’s only an association.
Dr. Forester said the study raises a number of questions, including the type of alcohol study participants consumed and whether this has any impact on cognitive benefit. He also questioned whether the mediating effects of alcohol were associated with something that wasn’t measured, such as socioeconomic status.
Another question, he said, is what factors in individuals’ medical or psychiatric history determine whether they are more or less likely to benefit from low to moderate alcohol intake.
Perhaps alcohol should be recommended only for “select subpopulations” – for example, those who are healthy and have a family history of cognitive decline –but not for those with a history of substance abuse, including alcohol abuse, said Dr. Forester.
“For this population, the last thing you want to do is recommend alcohol to reduce risk of cognitive decline,” he cautioned.
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. The investigators and Dr. Forester have reported no relevant financial disclosures.
A version of this story originally appeared on Medscape.com.
new research suggests. However, at least one expert urges caution in interpreting the findings.
Investigators found that consuming 10-14 alcoholic drinks per week had the strongest cognitive benefit. The findings “add more weight” to the growing body of research identifying beneficial cognitive effects of moderate alcohol consumption, said lead author, Ruiyuan Zhang, MD, of the department of epidemiology and biostatistics at the University of Georgia, Athens. However, Dr. Zhang emphasized that nondrinkers should not take up drinking to protect brain function, as alcohol can have negative effects.
The study was published online in JAMA Network Open.
Slower cognitive decline
The observational study was a secondary analysis of data from the Health and Retirement Study, a nationally representative U.S. survey of middle-aged and older adults. The survey, which began in 1992, is conducted every 2 years and collects health and economic data.
The current analysis used data from 1996 to 2008 and included information from individuals who participated in at least three surveys. The study included 19,887 participants, with a mean age 61.8 years. Most (60.1%) were women and white (85.2%). Mean follow-up was 9.1 years.
Researchers measured cognitive domains of mental status, word recall, and vocabulary. They also calculated a total cognition score, with higher scores indicating better cognitive abilities.
For each cognitive function measure, researchers categorized participants into a consistently low–trajectory group in which cognitive test scores from baseline through follow-up were consistently low or a consistently high–trajectory group, where cognitive test scores from baseline through follow-up were consistently high.
Based on self-reports, the investigators categorized participants as never drinkers (41.8%), former drinkers (39.5%), or current drinkers (18.7%). For current drinkers, researchers determined the number of drinking days per week and number of drinks per day. They further categorized these participants as low to moderate drinkers or heavy drinkers.
One drink was defined as a 12-ounce bottle of beer, a 5-ounce glass of wine, or a 1.5-ounce shot of spirits, said Dr. Zhang.
Women who consumed 8 or more drinks per week and men who drank 15 or more drinks per week were considered heavy drinkers. Other current drinkers were deemed low to moderate drinkers. Most current drinkers (85.2%) were low to moderate drinkers.
Other covariates included age, sex, race/ethnicity, years of education, marital status, tobacco smoking status, and body mass index.
Results showed moderate drinking was associated with relatively high cognitive test scores. After controlling for all covariates, compared with never drinkers, current low to moderate drinkers were significantly less likely to have consistently low trajectories for total cognitive score (odds ratio, 0.66; 95% confidence interval, 0.59-0.74), mental status (OR, 0.71; 95% CI, 0.63-0.81), word recall (OR, 0.74; 95% CI, 0.69-0.80), and vocabulary (OR, 0.64; 95% CI, 0.56-0.74) (all P < .001).
Former drinkers also had better cognitive outcomes for all cognitive domains. Heavy drinkers had lower odds of being in the consistently low trajectory group only for the vocabulary test.
Heavy drinking ‘risky’
Because few participants were deemed to be heavy drinkers, the power to identify an association between heavy drinking and cognitive function was limited. Dr. Zhang acknowledged, though he noted that heavy drinking is “risky.”
“We found that, after the drinking dosage passes the moderate level, the risk of low cognitive function increases very fast, which indicates that heavy drinking may harm cognitive function.” Limiting alcohol consumption “is still very important,” he said.
The associations of alcohol and cognitive functions differed by race/ethnicity. Low to moderate drinking was significantly associated with a lower odds of having a consistently low trajectory for all four cognitive function measures only among white participants.
A possible reason for this is that the study had so few African Americans (who made up only 14.8% of the sample), which limited the ability to identify relationships between alcohol intake and cognitive function, said Dr. Zhang. “Another reason is that the sensitivity to alcohol may be different between white and African American subjects.”
There was a significant U-shaped association between weekly amounts of alcohol and the odds of being in the consistently low–trajectory group for all cognitive functions. Depending on the function tested, the optimal number of weekly drinks ranged from 10-14.
Dr. Zhang noted that, when women were examined separately, alcohol consumption had a significant U-shaped relationship only with word recall, with the optimal dosage being around eight drinks.
U-shaped relationship an ‘important finding’
The U-shaped relationship is “an important finding,” said Dr. Zhang. “It shows that the human body may act differently to low and high doses of alcohol. Knowing why and how this happens is very important as it would help us understand how alcohol affects the function of the human body.”
Sensitivity analyses among participants with no chronic diseases showed the U-shaped association was still significant for scores of total word recall and vocabulary, but not for mental status or total cognition score.
The authors noted that 77.2% of participants had at least one chronic disease. They maintained that the association between alcohol consumption and cognitive function may be applicable both to healthy people and to those with a chronic disease.
The study also found that low to moderate drinkers had slower rates of cognitive decline over time for all cognition domains.
Although the mechanisms underlying the cognitive benefits of alcohol consumption are unclear, the authors believe it may be via cerebrovascular and cardiovascular pathways.
Alcohol may increase levels of brain-derived neurotrophic factor, a key regulator of neuronal plasticity and development in the dorsal striatum, they noted.
Balancing act
However, there’s also evidence that drinking, especially heavy drinking, increases the risk of hypertension, stroke, liver damage, and some cancers. “We think the role of alcohol drinking in cognitive function may be a balance of its beneficial and harmful effects on the cardiovascular system,” said Dr. Zhang.
“For the low to moderate drinker, the beneficial effects may outweigh the harmful effects on the small blood vessels in the brain. In this way, it could preserve cognition,” he added.
Dr. Zhang also noted that the study focused on middle-aged and older adults. “We can’t say whether or not moderate alcohol could benefit younger people” because they may have different characteristics, he said.
The findings of other studies examining the effects of alcohol on cognitive function are mixed. While studies have identified a beneficial effect, others have uncovered no, minimal, or adverse effects. This could be due to the use of different tests of cognitive function or different study populations, said Dr. Zhang.
A limitation of the current study was that assessment of alcohol consumption was based on self-report, which might have introduced recall bias. In addition, because individuals tend to underestimate their alcohol consumption, heavy drinkers could be misclassified as low to moderate drinkers, and low to moderate drinkers as former drinkers.
“This may make our study underestimate the association between low to moderate drinking and cognitive function,” said Dr. Zhang. In addition, alcohol consumption tended to change with time, and this change may be associated with other factors that led to changes in cognitive function, the authors noted.
Interpret with caution
Commenting on the study, Brent P. Forester, MD, chief of the Center of Excellence in Geriatric Psychiatry at McLean Hospital in Belmont, Mass., associate professor of psychiatry at Harvard Medical School, Boston, and a member of the American Psychiatric Association Council on Geriatric Psychiatry, said he views the study with some trepidation.
“As a clinician taking care of older adults, I would be very cautious about overinterpreting the beneficial effects of alcohol before we understand the mechanism better,” he said.
He noted that all of the risk factors associated with heart attack and stroke are also risk factors for Alzheimer’s disease and cognitive decline more broadly. “One of the issues here is how in the world does alcohol reduce cardiovascular and cerebrovascular risks, if you know it increases the risk of hypertension and stroke, regardless of dose.”
With regard to the possible impact of alcohol on brain-derived neurotrophic factor, Dr. Forester said, “it’s an interesting idea” but the actual mechanism is still unclear.
Even with dietary studies, such as those on the Mediterranean diet that include red wine, showing cognitive benefit, Dr. Forester said he’s still concerned about the adverse effects of alcohol on older people. These can include falls and sleep disturbances in addition to cognitive issues, and these effects can increase with age.
He was somewhat surprised at the level of alcohol that the study determined was beneficial. “Essentially, what they’re saying here is that, for men, it’s two drinks a day.” This could be “problematic” as two drinks per day can quickly escalate as individuals build tolerance.
He also pointed out that the study does not determine cause and effect, noting that it’s only an association.
Dr. Forester said the study raises a number of questions, including the type of alcohol study participants consumed and whether this has any impact on cognitive benefit. He also questioned whether the mediating effects of alcohol were associated with something that wasn’t measured, such as socioeconomic status.
Another question, he said, is what factors in individuals’ medical or psychiatric history determine whether they are more or less likely to benefit from low to moderate alcohol intake.
Perhaps alcohol should be recommended only for “select subpopulations” – for example, those who are healthy and have a family history of cognitive decline –but not for those with a history of substance abuse, including alcohol abuse, said Dr. Forester.
“For this population, the last thing you want to do is recommend alcohol to reduce risk of cognitive decline,” he cautioned.
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. The investigators and Dr. Forester have reported no relevant financial disclosures.
A version of this story originally appeared on Medscape.com.
new research suggests. However, at least one expert urges caution in interpreting the findings.
Investigators found that consuming 10-14 alcoholic drinks per week had the strongest cognitive benefit. The findings “add more weight” to the growing body of research identifying beneficial cognitive effects of moderate alcohol consumption, said lead author, Ruiyuan Zhang, MD, of the department of epidemiology and biostatistics at the University of Georgia, Athens. However, Dr. Zhang emphasized that nondrinkers should not take up drinking to protect brain function, as alcohol can have negative effects.
The study was published online in JAMA Network Open.
Slower cognitive decline
The observational study was a secondary analysis of data from the Health and Retirement Study, a nationally representative U.S. survey of middle-aged and older adults. The survey, which began in 1992, is conducted every 2 years and collects health and economic data.
The current analysis used data from 1996 to 2008 and included information from individuals who participated in at least three surveys. The study included 19,887 participants, with a mean age 61.8 years. Most (60.1%) were women and white (85.2%). Mean follow-up was 9.1 years.
Researchers measured cognitive domains of mental status, word recall, and vocabulary. They also calculated a total cognition score, with higher scores indicating better cognitive abilities.
For each cognitive function measure, researchers categorized participants into a consistently low–trajectory group in which cognitive test scores from baseline through follow-up were consistently low or a consistently high–trajectory group, where cognitive test scores from baseline through follow-up were consistently high.
Based on self-reports, the investigators categorized participants as never drinkers (41.8%), former drinkers (39.5%), or current drinkers (18.7%). For current drinkers, researchers determined the number of drinking days per week and number of drinks per day. They further categorized these participants as low to moderate drinkers or heavy drinkers.
One drink was defined as a 12-ounce bottle of beer, a 5-ounce glass of wine, or a 1.5-ounce shot of spirits, said Dr. Zhang.
Women who consumed 8 or more drinks per week and men who drank 15 or more drinks per week were considered heavy drinkers. Other current drinkers were deemed low to moderate drinkers. Most current drinkers (85.2%) were low to moderate drinkers.
Other covariates included age, sex, race/ethnicity, years of education, marital status, tobacco smoking status, and body mass index.
Results showed moderate drinking was associated with relatively high cognitive test scores. After controlling for all covariates, compared with never drinkers, current low to moderate drinkers were significantly less likely to have consistently low trajectories for total cognitive score (odds ratio, 0.66; 95% confidence interval, 0.59-0.74), mental status (OR, 0.71; 95% CI, 0.63-0.81), word recall (OR, 0.74; 95% CI, 0.69-0.80), and vocabulary (OR, 0.64; 95% CI, 0.56-0.74) (all P < .001).
Former drinkers also had better cognitive outcomes for all cognitive domains. Heavy drinkers had lower odds of being in the consistently low trajectory group only for the vocabulary test.
Heavy drinking ‘risky’
Because few participants were deemed to be heavy drinkers, the power to identify an association between heavy drinking and cognitive function was limited. Dr. Zhang acknowledged, though he noted that heavy drinking is “risky.”
“We found that, after the drinking dosage passes the moderate level, the risk of low cognitive function increases very fast, which indicates that heavy drinking may harm cognitive function.” Limiting alcohol consumption “is still very important,” he said.
The associations of alcohol and cognitive functions differed by race/ethnicity. Low to moderate drinking was significantly associated with a lower odds of having a consistently low trajectory for all four cognitive function measures only among white participants.
A possible reason for this is that the study had so few African Americans (who made up only 14.8% of the sample), which limited the ability to identify relationships between alcohol intake and cognitive function, said Dr. Zhang. “Another reason is that the sensitivity to alcohol may be different between white and African American subjects.”
There was a significant U-shaped association between weekly amounts of alcohol and the odds of being in the consistently low–trajectory group for all cognitive functions. Depending on the function tested, the optimal number of weekly drinks ranged from 10-14.
Dr. Zhang noted that, when women were examined separately, alcohol consumption had a significant U-shaped relationship only with word recall, with the optimal dosage being around eight drinks.
U-shaped relationship an ‘important finding’
The U-shaped relationship is “an important finding,” said Dr. Zhang. “It shows that the human body may act differently to low and high doses of alcohol. Knowing why and how this happens is very important as it would help us understand how alcohol affects the function of the human body.”
Sensitivity analyses among participants with no chronic diseases showed the U-shaped association was still significant for scores of total word recall and vocabulary, but not for mental status or total cognition score.
The authors noted that 77.2% of participants had at least one chronic disease. They maintained that the association between alcohol consumption and cognitive function may be applicable both to healthy people and to those with a chronic disease.
The study also found that low to moderate drinkers had slower rates of cognitive decline over time for all cognition domains.
Although the mechanisms underlying the cognitive benefits of alcohol consumption are unclear, the authors believe it may be via cerebrovascular and cardiovascular pathways.
Alcohol may increase levels of brain-derived neurotrophic factor, a key regulator of neuronal plasticity and development in the dorsal striatum, they noted.
Balancing act
However, there’s also evidence that drinking, especially heavy drinking, increases the risk of hypertension, stroke, liver damage, and some cancers. “We think the role of alcohol drinking in cognitive function may be a balance of its beneficial and harmful effects on the cardiovascular system,” said Dr. Zhang.
“For the low to moderate drinker, the beneficial effects may outweigh the harmful effects on the small blood vessels in the brain. In this way, it could preserve cognition,” he added.
Dr. Zhang also noted that the study focused on middle-aged and older adults. “We can’t say whether or not moderate alcohol could benefit younger people” because they may have different characteristics, he said.
The findings of other studies examining the effects of alcohol on cognitive function are mixed. While studies have identified a beneficial effect, others have uncovered no, minimal, or adverse effects. This could be due to the use of different tests of cognitive function or different study populations, said Dr. Zhang.
A limitation of the current study was that assessment of alcohol consumption was based on self-report, which might have introduced recall bias. In addition, because individuals tend to underestimate their alcohol consumption, heavy drinkers could be misclassified as low to moderate drinkers, and low to moderate drinkers as former drinkers.
“This may make our study underestimate the association between low to moderate drinking and cognitive function,” said Dr. Zhang. In addition, alcohol consumption tended to change with time, and this change may be associated with other factors that led to changes in cognitive function, the authors noted.
Interpret with caution
Commenting on the study, Brent P. Forester, MD, chief of the Center of Excellence in Geriatric Psychiatry at McLean Hospital in Belmont, Mass., associate professor of psychiatry at Harvard Medical School, Boston, and a member of the American Psychiatric Association Council on Geriatric Psychiatry, said he views the study with some trepidation.
“As a clinician taking care of older adults, I would be very cautious about overinterpreting the beneficial effects of alcohol before we understand the mechanism better,” he said.
He noted that all of the risk factors associated with heart attack and stroke are also risk factors for Alzheimer’s disease and cognitive decline more broadly. “One of the issues here is how in the world does alcohol reduce cardiovascular and cerebrovascular risks, if you know it increases the risk of hypertension and stroke, regardless of dose.”
With regard to the possible impact of alcohol on brain-derived neurotrophic factor, Dr. Forester said, “it’s an interesting idea” but the actual mechanism is still unclear.
Even with dietary studies, such as those on the Mediterranean diet that include red wine, showing cognitive benefit, Dr. Forester said he’s still concerned about the adverse effects of alcohol on older people. These can include falls and sleep disturbances in addition to cognitive issues, and these effects can increase with age.
He was somewhat surprised at the level of alcohol that the study determined was beneficial. “Essentially, what they’re saying here is that, for men, it’s two drinks a day.” This could be “problematic” as two drinks per day can quickly escalate as individuals build tolerance.
He also pointed out that the study does not determine cause and effect, noting that it’s only an association.
Dr. Forester said the study raises a number of questions, including the type of alcohol study participants consumed and whether this has any impact on cognitive benefit. He also questioned whether the mediating effects of alcohol were associated with something that wasn’t measured, such as socioeconomic status.
Another question, he said, is what factors in individuals’ medical or psychiatric history determine whether they are more or less likely to benefit from low to moderate alcohol intake.
Perhaps alcohol should be recommended only for “select subpopulations” – for example, those who are healthy and have a family history of cognitive decline –but not for those with a history of substance abuse, including alcohol abuse, said Dr. Forester.
“For this population, the last thing you want to do is recommend alcohol to reduce risk of cognitive decline,” he cautioned.
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. The investigators and Dr. Forester have reported no relevant financial disclosures.
A version of this story originally appeared on Medscape.com.
Epilepsy after TBI linked to worse 12-month outcomes
findings from an analysis of a large, prospective database suggest. “We found that patients essentially have a 10-times greater risk of developing posttraumatic epilepsy and seizures at 12 months [post injury] if the presenting Glasgow Coma Scale GCS) is less than 8,” said lead author John F. Burke, MD, PhD, University of California, San Francisco, in presenting the findings as part of the virtual annual meeting of the American Association of Neurological Surgeons.
Assessing risk factors
While posttraumatic epilepsy represents an estimated 20% of all cases of symptomatic epilepsy, many questions remain on those most at risk and on the long-term effects of posttraumatic epilepsy on TBI outcomes. To probe those issues, Dr. Burke and colleagues turned to the multicenter TRACK-TBI database, which has prospective, longitudinal data on more than 2,700 patients with traumatic brain injuries and is considered the largest source of prospective data on posttraumatic epilepsy.
Using the criteria of no previous epilepsy and having 12 months of follow-up, the team identified 1,493 patients with TBI. In addition, investigators identified 182 orthopedic controls (included and prospectively followed because they have injuries but not specifically head trauma) and 210 controls who are friends of the patients and who do not have injuries but allow researchers to control for socioeconomic and environmental factors.
Of the 1,493 patients with TBI, 41 (2.7%) were determined to have posttraumatic epilepsy, assessed according to a National Institute of Neurological Disorders and Stroke epilepsy screening questionnaire, which is designed to identify patients with posttraumatic epilepsy symptoms. There were no reports of epilepsy symptoms using the screening tool among the controls. Dr. Burke noted that the 2.7% was in agreement with historical reports.
In comparing patients with TBI who did and did not have posttraumatic epilepsy, no differences were observed in the groups in terms of gender, although there was a trend toward younger age among those with PTE (mean age, 35.4 years with posttraumatic injury vs. 41.5 without; P = .05).
A major risk factor for the development of posttraumatic epilepsy was presenting GCS scores. Among those with scores of less than 8, indicative of severe injury, the rate of posttraumatic epilepsy was 6% at 6 months and 12.5% at 12 months. In contrast, those with TBI presenting with GCS scores between 13 and 15, indicative of minor injury, had an incidence of posttraumatic epilepsy of 0.9% at 6 months and 1.4% at 12 months.
Imaging findings in the two groups showed that hemorrhage detected on CT imaging was associated with a significantly higher risk for posttraumatic epilepsy (P < .001).
“The main takeaway is that any hemorrhage in the brain is a major risk factor for developing seizures,” Dr. Burke said. “Whether it is subdural, epidural blood, subarachnoid or contusion, any blood confers a very [high] risk for developing seizures.”
Posttraumatic epilepsy was linked to poorer longer-term outcomes even for patients with lesser injury: Among those with TBI and GCS of 13-15, the mean Glasgow Outcome Scale Extended (GOSE) score at 12 months among those without posttraumatic epilepsy was 7, indicative of a good recovery with minor defects, whereas the mean GOSE score for those with PTE was 4.6, indicative of moderate to severe disability (P < .001).
“It was surprising to us that PTE-positive patients had a very significant decrease in GOSE, compared to PTE-negative patients,” Dr. Burke said. “There was a nearly 2-point drop in the GOSE and that was extremely significant.”
A multivariate analysis showed there was still a significant independent risk for a poor GOSE score with posttraumatic epilepsy after controlling for GCS score, head CT findings, and age (P < .001).
The authors also looked at mood outcomes using the Brief Symptom Inventory–18, which showed significant worse effect in those with posttraumatic epilepsy after multivariate adjustment (P = .01). Additionally, a highly significant worse effect in cognitive outcomes on the Rivermead cognitive metric was observed with posttraumatic epilepsy (P = .001).
“On all metrics tested, posttraumatic epilepsy worsened outcomes,” Dr. Burke said.
He noted that the study has some key limitations, including the 12-month follow-up. A previous study showed a linear increase in posttraumatic follow-up up to 30 years. “The fact that we found 41 patients at 12 months indicates there are probably more that are out there who are going to develop seizures, but because we don’t have the follow-up we can’t look at that.”
Although the screening questionnaires are effective, “the issue is these people are not being seen by an epileptologist or having scalp EEG done, and we need a more accurate way to do this,” he said. A new study, TRACK-TBI EPI, will address those limitations and a host of other issues with a 5-year follow-up.
Capturing the nuances of brain injury
Commenting on the study as a discussant, neurosurgeon Uzma Samadani, MD, PhD, of the Minneapolis Veterans Affairs Medical Center and CentraCare in Minneapolis, suggested that the future work should focus on issues including the wide-ranging mechanisms that could explain the seizure activity.
“For example, it’s known that posttraumatic epilepsy or seizures can be triggered by abnormal conductivity due to multiple different mechanisms associated with brain injury, such as endocrine dysfunction, cortical-spreading depression, and many others,” said Dr. Samadani, who has been a researcher on the TRACK-TBI study.
Factors ranging from genetic differences to comorbid conditions such as alcoholism can play a role in brain injury susceptibility, Dr. Samadani added. Furthermore, outcome measures currently available simply may not capture the unknown nuances of brain injury.
“We have to ask, are these an all-or-none phenomena, or is aberrant electrical activity after brain injury a continuum of dysfunction?” Dr. Samadani speculated.
“I would caution that we are likely underestimating the non–easily measurable consequences of brain injury,” she said. “And the better we can quantitate susceptibility, classify the nature of injury and target acute management, the less posttraumatic epilepsy/aberrant electrical activity our patients will have.”
Dr. Burke and Dr. Samadani disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
findings from an analysis of a large, prospective database suggest. “We found that patients essentially have a 10-times greater risk of developing posttraumatic epilepsy and seizures at 12 months [post injury] if the presenting Glasgow Coma Scale GCS) is less than 8,” said lead author John F. Burke, MD, PhD, University of California, San Francisco, in presenting the findings as part of the virtual annual meeting of the American Association of Neurological Surgeons.
Assessing risk factors
While posttraumatic epilepsy represents an estimated 20% of all cases of symptomatic epilepsy, many questions remain on those most at risk and on the long-term effects of posttraumatic epilepsy on TBI outcomes. To probe those issues, Dr. Burke and colleagues turned to the multicenter TRACK-TBI database, which has prospective, longitudinal data on more than 2,700 patients with traumatic brain injuries and is considered the largest source of prospective data on posttraumatic epilepsy.
Using the criteria of no previous epilepsy and having 12 months of follow-up, the team identified 1,493 patients with TBI. In addition, investigators identified 182 orthopedic controls (included and prospectively followed because they have injuries but not specifically head trauma) and 210 controls who are friends of the patients and who do not have injuries but allow researchers to control for socioeconomic and environmental factors.
Of the 1,493 patients with TBI, 41 (2.7%) were determined to have posttraumatic epilepsy, assessed according to a National Institute of Neurological Disorders and Stroke epilepsy screening questionnaire, which is designed to identify patients with posttraumatic epilepsy symptoms. There were no reports of epilepsy symptoms using the screening tool among the controls. Dr. Burke noted that the 2.7% was in agreement with historical reports.
In comparing patients with TBI who did and did not have posttraumatic epilepsy, no differences were observed in the groups in terms of gender, although there was a trend toward younger age among those with PTE (mean age, 35.4 years with posttraumatic injury vs. 41.5 without; P = .05).
A major risk factor for the development of posttraumatic epilepsy was presenting GCS scores. Among those with scores of less than 8, indicative of severe injury, the rate of posttraumatic epilepsy was 6% at 6 months and 12.5% at 12 months. In contrast, those with TBI presenting with GCS scores between 13 and 15, indicative of minor injury, had an incidence of posttraumatic epilepsy of 0.9% at 6 months and 1.4% at 12 months.
Imaging findings in the two groups showed that hemorrhage detected on CT imaging was associated with a significantly higher risk for posttraumatic epilepsy (P < .001).
“The main takeaway is that any hemorrhage in the brain is a major risk factor for developing seizures,” Dr. Burke said. “Whether it is subdural, epidural blood, subarachnoid or contusion, any blood confers a very [high] risk for developing seizures.”
Posttraumatic epilepsy was linked to poorer longer-term outcomes even for patients with lesser injury: Among those with TBI and GCS of 13-15, the mean Glasgow Outcome Scale Extended (GOSE) score at 12 months among those without posttraumatic epilepsy was 7, indicative of a good recovery with minor defects, whereas the mean GOSE score for those with PTE was 4.6, indicative of moderate to severe disability (P < .001).
“It was surprising to us that PTE-positive patients had a very significant decrease in GOSE, compared to PTE-negative patients,” Dr. Burke said. “There was a nearly 2-point drop in the GOSE and that was extremely significant.”
A multivariate analysis showed there was still a significant independent risk for a poor GOSE score with posttraumatic epilepsy after controlling for GCS score, head CT findings, and age (P < .001).
The authors also looked at mood outcomes using the Brief Symptom Inventory–18, which showed significant worse effect in those with posttraumatic epilepsy after multivariate adjustment (P = .01). Additionally, a highly significant worse effect in cognitive outcomes on the Rivermead cognitive metric was observed with posttraumatic epilepsy (P = .001).
“On all metrics tested, posttraumatic epilepsy worsened outcomes,” Dr. Burke said.
He noted that the study has some key limitations, including the 12-month follow-up. A previous study showed a linear increase in posttraumatic follow-up up to 30 years. “The fact that we found 41 patients at 12 months indicates there are probably more that are out there who are going to develop seizures, but because we don’t have the follow-up we can’t look at that.”
Although the screening questionnaires are effective, “the issue is these people are not being seen by an epileptologist or having scalp EEG done, and we need a more accurate way to do this,” he said. A new study, TRACK-TBI EPI, will address those limitations and a host of other issues with a 5-year follow-up.
Capturing the nuances of brain injury
Commenting on the study as a discussant, neurosurgeon Uzma Samadani, MD, PhD, of the Minneapolis Veterans Affairs Medical Center and CentraCare in Minneapolis, suggested that the future work should focus on issues including the wide-ranging mechanisms that could explain the seizure activity.
“For example, it’s known that posttraumatic epilepsy or seizures can be triggered by abnormal conductivity due to multiple different mechanisms associated with brain injury, such as endocrine dysfunction, cortical-spreading depression, and many others,” said Dr. Samadani, who has been a researcher on the TRACK-TBI study.
Factors ranging from genetic differences to comorbid conditions such as alcoholism can play a role in brain injury susceptibility, Dr. Samadani added. Furthermore, outcome measures currently available simply may not capture the unknown nuances of brain injury.
“We have to ask, are these an all-or-none phenomena, or is aberrant electrical activity after brain injury a continuum of dysfunction?” Dr. Samadani speculated.
“I would caution that we are likely underestimating the non–easily measurable consequences of brain injury,” she said. “And the better we can quantitate susceptibility, classify the nature of injury and target acute management, the less posttraumatic epilepsy/aberrant electrical activity our patients will have.”
Dr. Burke and Dr. Samadani disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
findings from an analysis of a large, prospective database suggest. “We found that patients essentially have a 10-times greater risk of developing posttraumatic epilepsy and seizures at 12 months [post injury] if the presenting Glasgow Coma Scale GCS) is less than 8,” said lead author John F. Burke, MD, PhD, University of California, San Francisco, in presenting the findings as part of the virtual annual meeting of the American Association of Neurological Surgeons.
Assessing risk factors
While posttraumatic epilepsy represents an estimated 20% of all cases of symptomatic epilepsy, many questions remain on those most at risk and on the long-term effects of posttraumatic epilepsy on TBI outcomes. To probe those issues, Dr. Burke and colleagues turned to the multicenter TRACK-TBI database, which has prospective, longitudinal data on more than 2,700 patients with traumatic brain injuries and is considered the largest source of prospective data on posttraumatic epilepsy.
Using the criteria of no previous epilepsy and having 12 months of follow-up, the team identified 1,493 patients with TBI. In addition, investigators identified 182 orthopedic controls (included and prospectively followed because they have injuries but not specifically head trauma) and 210 controls who are friends of the patients and who do not have injuries but allow researchers to control for socioeconomic and environmental factors.
Of the 1,493 patients with TBI, 41 (2.7%) were determined to have posttraumatic epilepsy, assessed according to a National Institute of Neurological Disorders and Stroke epilepsy screening questionnaire, which is designed to identify patients with posttraumatic epilepsy symptoms. There were no reports of epilepsy symptoms using the screening tool among the controls. Dr. Burke noted that the 2.7% was in agreement with historical reports.
In comparing patients with TBI who did and did not have posttraumatic epilepsy, no differences were observed in the groups in terms of gender, although there was a trend toward younger age among those with PTE (mean age, 35.4 years with posttraumatic injury vs. 41.5 without; P = .05).
A major risk factor for the development of posttraumatic epilepsy was presenting GCS scores. Among those with scores of less than 8, indicative of severe injury, the rate of posttraumatic epilepsy was 6% at 6 months and 12.5% at 12 months. In contrast, those with TBI presenting with GCS scores between 13 and 15, indicative of minor injury, had an incidence of posttraumatic epilepsy of 0.9% at 6 months and 1.4% at 12 months.
Imaging findings in the two groups showed that hemorrhage detected on CT imaging was associated with a significantly higher risk for posttraumatic epilepsy (P < .001).
“The main takeaway is that any hemorrhage in the brain is a major risk factor for developing seizures,” Dr. Burke said. “Whether it is subdural, epidural blood, subarachnoid or contusion, any blood confers a very [high] risk for developing seizures.”
Posttraumatic epilepsy was linked to poorer longer-term outcomes even for patients with lesser injury: Among those with TBI and GCS of 13-15, the mean Glasgow Outcome Scale Extended (GOSE) score at 12 months among those without posttraumatic epilepsy was 7, indicative of a good recovery with minor defects, whereas the mean GOSE score for those with PTE was 4.6, indicative of moderate to severe disability (P < .001).
“It was surprising to us that PTE-positive patients had a very significant decrease in GOSE, compared to PTE-negative patients,” Dr. Burke said. “There was a nearly 2-point drop in the GOSE and that was extremely significant.”
A multivariate analysis showed there was still a significant independent risk for a poor GOSE score with posttraumatic epilepsy after controlling for GCS score, head CT findings, and age (P < .001).
The authors also looked at mood outcomes using the Brief Symptom Inventory–18, which showed significant worse effect in those with posttraumatic epilepsy after multivariate adjustment (P = .01). Additionally, a highly significant worse effect in cognitive outcomes on the Rivermead cognitive metric was observed with posttraumatic epilepsy (P = .001).
“On all metrics tested, posttraumatic epilepsy worsened outcomes,” Dr. Burke said.
He noted that the study has some key limitations, including the 12-month follow-up. A previous study showed a linear increase in posttraumatic follow-up up to 30 years. “The fact that we found 41 patients at 12 months indicates there are probably more that are out there who are going to develop seizures, but because we don’t have the follow-up we can’t look at that.”
Although the screening questionnaires are effective, “the issue is these people are not being seen by an epileptologist or having scalp EEG done, and we need a more accurate way to do this,” he said. A new study, TRACK-TBI EPI, will address those limitations and a host of other issues with a 5-year follow-up.
Capturing the nuances of brain injury
Commenting on the study as a discussant, neurosurgeon Uzma Samadani, MD, PhD, of the Minneapolis Veterans Affairs Medical Center and CentraCare in Minneapolis, suggested that the future work should focus on issues including the wide-ranging mechanisms that could explain the seizure activity.
“For example, it’s known that posttraumatic epilepsy or seizures can be triggered by abnormal conductivity due to multiple different mechanisms associated with brain injury, such as endocrine dysfunction, cortical-spreading depression, and many others,” said Dr. Samadani, who has been a researcher on the TRACK-TBI study.
Factors ranging from genetic differences to comorbid conditions such as alcoholism can play a role in brain injury susceptibility, Dr. Samadani added. Furthermore, outcome measures currently available simply may not capture the unknown nuances of brain injury.
“We have to ask, are these an all-or-none phenomena, or is aberrant electrical activity after brain injury a continuum of dysfunction?” Dr. Samadani speculated.
“I would caution that we are likely underestimating the non–easily measurable consequences of brain injury,” she said. “And the better we can quantitate susceptibility, classify the nature of injury and target acute management, the less posttraumatic epilepsy/aberrant electrical activity our patients will have.”
Dr. Burke and Dr. Samadani disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM AANS 2020