User login
AHS White Paper Guides Treatment of Posttraumatic Headache in Youth
The guidance document, the first of its kind, covers risk factors for prolonged recovery, along with pharmacologic and nonpharmacologic management strategies, and supports an emphasis on multidisciplinary care, lead author Carlyn Patterson Gentile, MD, PhD, attending physician in the Division of Neurology at Children’s Hospital of Philadelphia in Pennsylvania, and colleagues reported.
“There are no guidelines to inform the management of posttraumatic headache in youth, but multiple studies have been conducted over the past 2 decades,” the authors wrote in Headache. “This white paper aims to provide a thorough review of the current literature, identify gaps in knowledge, and provide a road map for [posttraumatic headache] management in youth based on available evidence and expert opinion.”
Clarity for an Underrecognized Issue
According to Russell Lonser, MD, professor and chair of neurological surgery at Ohio State University, Columbus, the white paper is important because it offers concrete guidance for health care providers who may be less familiar with posttraumatic headache in youth.
“It brings together all of the previous literature ... in a very well-written way,” Dr. Lonser said in an interview. “More than anything, it could reassure [providers] that they shouldn’t be hunting down potentially magical cures, and reassure them in symptomatic management.”
Meeryo C. Choe, MD, associate clinical professor of pediatric neurology at UCLA Health in Calabasas, California, said the paper also helps shine a light on what may be a more common condition than the public suspects.
“While the media focuses on the effects of concussion in professional sports athletes, the biggest population of athletes is in our youth population,” Dr. Choe said in a written comment. “Almost 25 million children participate in sports throughout the country, and yet we lack guidelines on how to treat posttraumatic headache which can often develop into persistent postconcussive symptoms.”
This white paper, she noted, builds on Dr. Gentile’s 2021 systematic review, introduces new management recommendations, and aligns with the latest consensus statement from the Concussion in Sport Group.
Risk Factors
The white paper first emphasizes the importance of early identification of youth at high risk for prolonged recovery from posttraumatic headache. Risk factors include female sex, adolescent age, a high number of acute symptoms following the initial injury, and social determinants of health.
“I agree that it is important to identify these patients early to improve the recovery trajectory,” Dr. Choe said.
Identifying these individuals quickly allows for timely intervention with both pharmacologic and nonpharmacologic therapies, Dr. Gentile and colleagues noted, potentially mitigating persistent symptoms. Clinicians are encouraged to perform thorough initial assessments to identify these risk factors and initiate early, personalized management plans.
Initial Management of Acute Posttraumatic Headache
For the initial management of acute posttraumatic headache, the white paper recommends a scheduled dosing regimen of simple analgesics. Ibuprofen at a dosage of 10 mg/kg every 6-8 hours (up to a maximum of 600 mg per dose) combined with acetaminophen has shown the best evidence for efficacy. Provided the patient is clinically stable, this regimen should be initiated within 48 hours of the injury and maintained with scheduled dosing for 3-10 days.
If effective, these medications can subsequently be used on an as-needed basis. Careful usage of analgesics is crucial, the white paper cautions, as overadministration can lead to medication-overuse headaches, complicating the recovery process.
Secondary Treatment Options
In cases where first-line oral medications are ineffective, the AHS white paper outlines several secondary treatment options. These include acute intravenous therapies such as ketorolac, dopamine receptor antagonists, and intravenous fluids. Nerve blocks and oral corticosteroid bridges may also be considered.
The white paper stresses the importance of individualized treatment plans that consider the specific needs and responses of each patient, noting that the evidence supporting these approaches is primarily derived from retrospective studies and case reports.
“Patient preferences should be factored in,” said Sean Rose, MD, pediatric neurologist and codirector of the Complex Concussion Clinic at Nationwide Children’s Hospital, Columbus, Ohio.
Supplements and Preventive Measures
For adolescents and young adults at high risk of prolonged posttraumatic headache, the white paper suggests the use of riboflavin and magnesium supplements. Small randomized clinical trials suggest that these supplements may aid in speeding recovery when administered for 1-2 weeks within 48 hours of injury.
If significant headache persists after 2 weeks, a regimen of riboflavin 400 mg daily and magnesium 400-500 mg nightly can be trialed for 6-8 weeks, in line with recommendations for migraine prevention. Additionally, melatonin at a dose of 3-5 mg nightly for an 8-week course may be considered for patients experiencing comorbid sleep disturbances.
Targeted Preventative Therapy
The white paper emphasizes the importance of targeting preventative therapy to the primary headache phenotype.
For instance, patients presenting with a migraine phenotype, or those with a personal or family history of migraines, may be most likely to respond to medications proven effective in migraine prevention, such as amitriptyline, topiramate, and propranolol.
“Most research evidence [for treating posttraumatic headache in youth] is still based on the treatment of migraine,” Dr. Rose pointed out in a written comment.
Dr. Gentile and colleagues recommend initiating preventive therapies 4-6 weeks post injury if headaches are not improving, occur more than 1-2 days per week, or significantly impact daily functioning.
Specialist Referrals and Physical Activity
Referral to a headache specialist is advised for patients who do not respond to first-line acute and preventive therapies. Specialists can offer advanced diagnostic and therapeutic options, the authors noted, ensuring a comprehensive approach to managing posttraumatic headache.
The white paper also recommends noncontact, sub–symptom threshold aerobic physical activity and activities of daily living after an initial 24-48 hour period of symptom-limited cognitive and physical rest. Engaging in these activities may promote faster recovery and help patients gradually return to their normal routines.
“This has been a shift in the concussion treatment approach over the last decade, and is one of the most important interventions we can recommend as physicians,” Dr. Choe noted. “This is where pediatricians and emergency department physicians seeing children acutely can really make a difference in the recovery trajectory for a child after a concussion. ‘Cocoon therapy’ has been proven not only to not work, but be detrimental to recovery.”
Nonpharmacologic Interventions
Based on clinical assessment, nonpharmacologic interventions may also be considered, according to the white paper. These interventions include cervico-vestibular therapy, which addresses neck and balance issues, and cognitive-behavioral therapy, which helps manage the psychological aspects of chronic headache. Dr. Gentile and colleagues highlighted the potential benefits of a collaborative care model that incorporates these nonpharmacologic interventions alongside pharmacologic treatments, providing a holistic approach to posttraumatic headache management.
“Persisting headaches after concussion are often driven by multiple factors,” Dr. Rose said. “Multidisciplinary concussion clinics can offer multiple treatment approaches such as behavioral, physical therapy, exercise, and medication options.”
Unmet Needs
The white paper concludes by calling for high-quality prospective cohort studies and placebo-controlled, randomized, controlled trials to further advance the understanding and treatment of posttraumatic headache in children.
Dr. Lonser, Dr. Choe, and Dr. Rose all agreed.
“More focused treatment trials are needed to gauge efficacy in children with headache after concussion,” Dr. Rose said.
Specifically, Dr. Gentile and colleagues underscored the need to standardize data collection via common elements, which could improve the ability to compare results across studies and develop more effective treatments. In addition, research into the underlying pathophysiology of posttraumatic headache is crucial for identifying new therapeutic targets and clinical and biological markers that can personalize patient care.
They also stressed the importance of exploring the impact of health disparities and social determinants on posttraumatic headache outcomes, aiming to develop interventions that are equitable and accessible to all patient populations.The white paper was approved by the AHS, and supported by the National Institutes of Health/National Institute of Neurological Disorders and Stroke K23 NS124986. The authors disclosed relationships with Eli Lilly, Pfizer, Amgen, and others. The interviewees disclosed no conflicts of interest.
The guidance document, the first of its kind, covers risk factors for prolonged recovery, along with pharmacologic and nonpharmacologic management strategies, and supports an emphasis on multidisciplinary care, lead author Carlyn Patterson Gentile, MD, PhD, attending physician in the Division of Neurology at Children’s Hospital of Philadelphia in Pennsylvania, and colleagues reported.
“There are no guidelines to inform the management of posttraumatic headache in youth, but multiple studies have been conducted over the past 2 decades,” the authors wrote in Headache. “This white paper aims to provide a thorough review of the current literature, identify gaps in knowledge, and provide a road map for [posttraumatic headache] management in youth based on available evidence and expert opinion.”
Clarity for an Underrecognized Issue
According to Russell Lonser, MD, professor and chair of neurological surgery at Ohio State University, Columbus, the white paper is important because it offers concrete guidance for health care providers who may be less familiar with posttraumatic headache in youth.
“It brings together all of the previous literature ... in a very well-written way,” Dr. Lonser said in an interview. “More than anything, it could reassure [providers] that they shouldn’t be hunting down potentially magical cures, and reassure them in symptomatic management.”
Meeryo C. Choe, MD, associate clinical professor of pediatric neurology at UCLA Health in Calabasas, California, said the paper also helps shine a light on what may be a more common condition than the public suspects.
“While the media focuses on the effects of concussion in professional sports athletes, the biggest population of athletes is in our youth population,” Dr. Choe said in a written comment. “Almost 25 million children participate in sports throughout the country, and yet we lack guidelines on how to treat posttraumatic headache which can often develop into persistent postconcussive symptoms.”
This white paper, she noted, builds on Dr. Gentile’s 2021 systematic review, introduces new management recommendations, and aligns with the latest consensus statement from the Concussion in Sport Group.
Risk Factors
The white paper first emphasizes the importance of early identification of youth at high risk for prolonged recovery from posttraumatic headache. Risk factors include female sex, adolescent age, a high number of acute symptoms following the initial injury, and social determinants of health.
“I agree that it is important to identify these patients early to improve the recovery trajectory,” Dr. Choe said.
Identifying these individuals quickly allows for timely intervention with both pharmacologic and nonpharmacologic therapies, Dr. Gentile and colleagues noted, potentially mitigating persistent symptoms. Clinicians are encouraged to perform thorough initial assessments to identify these risk factors and initiate early, personalized management plans.
Initial Management of Acute Posttraumatic Headache
For the initial management of acute posttraumatic headache, the white paper recommends a scheduled dosing regimen of simple analgesics. Ibuprofen at a dosage of 10 mg/kg every 6-8 hours (up to a maximum of 600 mg per dose) combined with acetaminophen has shown the best evidence for efficacy. Provided the patient is clinically stable, this regimen should be initiated within 48 hours of the injury and maintained with scheduled dosing for 3-10 days.
If effective, these medications can subsequently be used on an as-needed basis. Careful usage of analgesics is crucial, the white paper cautions, as overadministration can lead to medication-overuse headaches, complicating the recovery process.
Secondary Treatment Options
In cases where first-line oral medications are ineffective, the AHS white paper outlines several secondary treatment options. These include acute intravenous therapies such as ketorolac, dopamine receptor antagonists, and intravenous fluids. Nerve blocks and oral corticosteroid bridges may also be considered.
The white paper stresses the importance of individualized treatment plans that consider the specific needs and responses of each patient, noting that the evidence supporting these approaches is primarily derived from retrospective studies and case reports.
“Patient preferences should be factored in,” said Sean Rose, MD, pediatric neurologist and codirector of the Complex Concussion Clinic at Nationwide Children’s Hospital, Columbus, Ohio.
Supplements and Preventive Measures
For adolescents and young adults at high risk of prolonged posttraumatic headache, the white paper suggests the use of riboflavin and magnesium supplements. Small randomized clinical trials suggest that these supplements may aid in speeding recovery when administered for 1-2 weeks within 48 hours of injury.
If significant headache persists after 2 weeks, a regimen of riboflavin 400 mg daily and magnesium 400-500 mg nightly can be trialed for 6-8 weeks, in line with recommendations for migraine prevention. Additionally, melatonin at a dose of 3-5 mg nightly for an 8-week course may be considered for patients experiencing comorbid sleep disturbances.
Targeted Preventative Therapy
The white paper emphasizes the importance of targeting preventative therapy to the primary headache phenotype.
For instance, patients presenting with a migraine phenotype, or those with a personal or family history of migraines, may be most likely to respond to medications proven effective in migraine prevention, such as amitriptyline, topiramate, and propranolol.
“Most research evidence [for treating posttraumatic headache in youth] is still based on the treatment of migraine,” Dr. Rose pointed out in a written comment.
Dr. Gentile and colleagues recommend initiating preventive therapies 4-6 weeks post injury if headaches are not improving, occur more than 1-2 days per week, or significantly impact daily functioning.
Specialist Referrals and Physical Activity
Referral to a headache specialist is advised for patients who do not respond to first-line acute and preventive therapies. Specialists can offer advanced diagnostic and therapeutic options, the authors noted, ensuring a comprehensive approach to managing posttraumatic headache.
The white paper also recommends noncontact, sub–symptom threshold aerobic physical activity and activities of daily living after an initial 24-48 hour period of symptom-limited cognitive and physical rest. Engaging in these activities may promote faster recovery and help patients gradually return to their normal routines.
“This has been a shift in the concussion treatment approach over the last decade, and is one of the most important interventions we can recommend as physicians,” Dr. Choe noted. “This is where pediatricians and emergency department physicians seeing children acutely can really make a difference in the recovery trajectory for a child after a concussion. ‘Cocoon therapy’ has been proven not only to not work, but be detrimental to recovery.”
Nonpharmacologic Interventions
Based on clinical assessment, nonpharmacologic interventions may also be considered, according to the white paper. These interventions include cervico-vestibular therapy, which addresses neck and balance issues, and cognitive-behavioral therapy, which helps manage the psychological aspects of chronic headache. Dr. Gentile and colleagues highlighted the potential benefits of a collaborative care model that incorporates these nonpharmacologic interventions alongside pharmacologic treatments, providing a holistic approach to posttraumatic headache management.
“Persisting headaches after concussion are often driven by multiple factors,” Dr. Rose said. “Multidisciplinary concussion clinics can offer multiple treatment approaches such as behavioral, physical therapy, exercise, and medication options.”
Unmet Needs
The white paper concludes by calling for high-quality prospective cohort studies and placebo-controlled, randomized, controlled trials to further advance the understanding and treatment of posttraumatic headache in children.
Dr. Lonser, Dr. Choe, and Dr. Rose all agreed.
“More focused treatment trials are needed to gauge efficacy in children with headache after concussion,” Dr. Rose said.
Specifically, Dr. Gentile and colleagues underscored the need to standardize data collection via common elements, which could improve the ability to compare results across studies and develop more effective treatments. In addition, research into the underlying pathophysiology of posttraumatic headache is crucial for identifying new therapeutic targets and clinical and biological markers that can personalize patient care.
They also stressed the importance of exploring the impact of health disparities and social determinants on posttraumatic headache outcomes, aiming to develop interventions that are equitable and accessible to all patient populations.The white paper was approved by the AHS, and supported by the National Institutes of Health/National Institute of Neurological Disorders and Stroke K23 NS124986. The authors disclosed relationships with Eli Lilly, Pfizer, Amgen, and others. The interviewees disclosed no conflicts of interest.
The guidance document, the first of its kind, covers risk factors for prolonged recovery, along with pharmacologic and nonpharmacologic management strategies, and supports an emphasis on multidisciplinary care, lead author Carlyn Patterson Gentile, MD, PhD, attending physician in the Division of Neurology at Children’s Hospital of Philadelphia in Pennsylvania, and colleagues reported.
“There are no guidelines to inform the management of posttraumatic headache in youth, but multiple studies have been conducted over the past 2 decades,” the authors wrote in Headache. “This white paper aims to provide a thorough review of the current literature, identify gaps in knowledge, and provide a road map for [posttraumatic headache] management in youth based on available evidence and expert opinion.”
Clarity for an Underrecognized Issue
According to Russell Lonser, MD, professor and chair of neurological surgery at Ohio State University, Columbus, the white paper is important because it offers concrete guidance for health care providers who may be less familiar with posttraumatic headache in youth.
“It brings together all of the previous literature ... in a very well-written way,” Dr. Lonser said in an interview. “More than anything, it could reassure [providers] that they shouldn’t be hunting down potentially magical cures, and reassure them in symptomatic management.”
Meeryo C. Choe, MD, associate clinical professor of pediatric neurology at UCLA Health in Calabasas, California, said the paper also helps shine a light on what may be a more common condition than the public suspects.
“While the media focuses on the effects of concussion in professional sports athletes, the biggest population of athletes is in our youth population,” Dr. Choe said in a written comment. “Almost 25 million children participate in sports throughout the country, and yet we lack guidelines on how to treat posttraumatic headache which can often develop into persistent postconcussive symptoms.”
This white paper, she noted, builds on Dr. Gentile’s 2021 systematic review, introduces new management recommendations, and aligns with the latest consensus statement from the Concussion in Sport Group.
Risk Factors
The white paper first emphasizes the importance of early identification of youth at high risk for prolonged recovery from posttraumatic headache. Risk factors include female sex, adolescent age, a high number of acute symptoms following the initial injury, and social determinants of health.
“I agree that it is important to identify these patients early to improve the recovery trajectory,” Dr. Choe said.
Identifying these individuals quickly allows for timely intervention with both pharmacologic and nonpharmacologic therapies, Dr. Gentile and colleagues noted, potentially mitigating persistent symptoms. Clinicians are encouraged to perform thorough initial assessments to identify these risk factors and initiate early, personalized management plans.
Initial Management of Acute Posttraumatic Headache
For the initial management of acute posttraumatic headache, the white paper recommends a scheduled dosing regimen of simple analgesics. Ibuprofen at a dosage of 10 mg/kg every 6-8 hours (up to a maximum of 600 mg per dose) combined with acetaminophen has shown the best evidence for efficacy. Provided the patient is clinically stable, this regimen should be initiated within 48 hours of the injury and maintained with scheduled dosing for 3-10 days.
If effective, these medications can subsequently be used on an as-needed basis. Careful usage of analgesics is crucial, the white paper cautions, as overadministration can lead to medication-overuse headaches, complicating the recovery process.
Secondary Treatment Options
In cases where first-line oral medications are ineffective, the AHS white paper outlines several secondary treatment options. These include acute intravenous therapies such as ketorolac, dopamine receptor antagonists, and intravenous fluids. Nerve blocks and oral corticosteroid bridges may also be considered.
The white paper stresses the importance of individualized treatment plans that consider the specific needs and responses of each patient, noting that the evidence supporting these approaches is primarily derived from retrospective studies and case reports.
“Patient preferences should be factored in,” said Sean Rose, MD, pediatric neurologist and codirector of the Complex Concussion Clinic at Nationwide Children’s Hospital, Columbus, Ohio.
Supplements and Preventive Measures
For adolescents and young adults at high risk of prolonged posttraumatic headache, the white paper suggests the use of riboflavin and magnesium supplements. Small randomized clinical trials suggest that these supplements may aid in speeding recovery when administered for 1-2 weeks within 48 hours of injury.
If significant headache persists after 2 weeks, a regimen of riboflavin 400 mg daily and magnesium 400-500 mg nightly can be trialed for 6-8 weeks, in line with recommendations for migraine prevention. Additionally, melatonin at a dose of 3-5 mg nightly for an 8-week course may be considered for patients experiencing comorbid sleep disturbances.
Targeted Preventative Therapy
The white paper emphasizes the importance of targeting preventative therapy to the primary headache phenotype.
For instance, patients presenting with a migraine phenotype, or those with a personal or family history of migraines, may be most likely to respond to medications proven effective in migraine prevention, such as amitriptyline, topiramate, and propranolol.
“Most research evidence [for treating posttraumatic headache in youth] is still based on the treatment of migraine,” Dr. Rose pointed out in a written comment.
Dr. Gentile and colleagues recommend initiating preventive therapies 4-6 weeks post injury if headaches are not improving, occur more than 1-2 days per week, or significantly impact daily functioning.
Specialist Referrals and Physical Activity
Referral to a headache specialist is advised for patients who do not respond to first-line acute and preventive therapies. Specialists can offer advanced diagnostic and therapeutic options, the authors noted, ensuring a comprehensive approach to managing posttraumatic headache.
The white paper also recommends noncontact, sub–symptom threshold aerobic physical activity and activities of daily living after an initial 24-48 hour period of symptom-limited cognitive and physical rest. Engaging in these activities may promote faster recovery and help patients gradually return to their normal routines.
“This has been a shift in the concussion treatment approach over the last decade, and is one of the most important interventions we can recommend as physicians,” Dr. Choe noted. “This is where pediatricians and emergency department physicians seeing children acutely can really make a difference in the recovery trajectory for a child after a concussion. ‘Cocoon therapy’ has been proven not only to not work, but be detrimental to recovery.”
Nonpharmacologic Interventions
Based on clinical assessment, nonpharmacologic interventions may also be considered, according to the white paper. These interventions include cervico-vestibular therapy, which addresses neck and balance issues, and cognitive-behavioral therapy, which helps manage the psychological aspects of chronic headache. Dr. Gentile and colleagues highlighted the potential benefits of a collaborative care model that incorporates these nonpharmacologic interventions alongside pharmacologic treatments, providing a holistic approach to posttraumatic headache management.
“Persisting headaches after concussion are often driven by multiple factors,” Dr. Rose said. “Multidisciplinary concussion clinics can offer multiple treatment approaches such as behavioral, physical therapy, exercise, and medication options.”
Unmet Needs
The white paper concludes by calling for high-quality prospective cohort studies and placebo-controlled, randomized, controlled trials to further advance the understanding and treatment of posttraumatic headache in children.
Dr. Lonser, Dr. Choe, and Dr. Rose all agreed.
“More focused treatment trials are needed to gauge efficacy in children with headache after concussion,” Dr. Rose said.
Specifically, Dr. Gentile and colleagues underscored the need to standardize data collection via common elements, which could improve the ability to compare results across studies and develop more effective treatments. In addition, research into the underlying pathophysiology of posttraumatic headache is crucial for identifying new therapeutic targets and clinical and biological markers that can personalize patient care.
They also stressed the importance of exploring the impact of health disparities and social determinants on posttraumatic headache outcomes, aiming to develop interventions that are equitable and accessible to all patient populations.The white paper was approved by the AHS, and supported by the National Institutes of Health/National Institute of Neurological Disorders and Stroke K23 NS124986. The authors disclosed relationships with Eli Lilly, Pfizer, Amgen, and others. The interviewees disclosed no conflicts of interest.
FROM HEADACHE
New First-Line Therapies for Migraine Prevention
This transcript has been edited for clarity.
Today I am going to talk about the position statement from the American Headache Society (AHS) “Calcitonin gene-related peptide [CGRP]–targeting therapies are a first-line option for the prevention of migraine”. This update is of critical importance because about three fourths of people with migraine get their care from a primary care clinician, not from a neurologist or a headache specialist. CGRP-targeting therapies have transformed migraine care at the specialty level, but many in primary care are not yet familiar with this class of medicines. Until this new statement was released, CGRPs were not viewed as first-line agents for migraine. That has now changed.
Two main types of therapy for people with migraine headache are: (1) acute or abortive therapy (when a headache develops, it is treated), and (2) preventive therapy. Preventive therapy is typically used when the patient has headaches on 4 or more days per month. Preventive therapy is aimed at reducing the frequency and severity of headaches. About 40% of patients with migraine qualify for preventive therapy, but only a minority are receiving it.
The armamentarium for preventive therapy of migraines had not changed in a long time — until now. First-line preventive therapy has traditionally consisted of three classes of agents: beta-blockers, tricyclic antidepressants, and topiramate. These medicines were developed for different therapeutic purposes, yet they work for migraines. These drugs may have off-target effects that can make them difficult to tolerate.
Based on new evidence, candesartan — an angiotensin receptor blocker (ARB) — is now also a first-line drug for migraine. This is good news, because ARBs are a drug class that we have a lot of experience with, are easy to use, and could be an excellent choice for people with concomitant hypertension or chronic kidney disease. The serotonin-norepinephrine reuptake inhibitors (venlafaxine and duloxetine) are also considered first-line agents for migraine treatment.
In the AHS’s new position statement, the two main drug classes are small-molecule CGRP receptor antagonists and monoclonal antibodies.
The role of the neuropeptide CGRP in migraine was originally discovered after finding that blood levels of CGRP were elevated during migraine attacks. This led to the discovery of agents that blocked CGRP, initially for acute treatment of migraine, and then for preventive therapy. Multiple clinical studies show the CGRP targeting therapies to be as or even more effective than traditional first-line agents at decreasing the number of migraine days per month.
The efficacy and safety of these agents have been demonstrated in both randomized trials and in real-world studies. Other important positive endpoints include fewer days of migraine, reduced acute medication use, and improvements in many quality-of-life outcomes. Studies also have shown that CGRP-targeting therapies are well tolerated and safe, with very few serious adverse events.
Furthermore, studies have shown the CGRP targeting therapies are effective in individuals who have failed multiple other first-line therapies. They fit now both as first-line agents and as agents that can be used in difficult-to-treat patients as well as in patients who struggle with acute medication overuse, which is often very challenging.
To quote from the AHS statement,
Side effects are uncommon and can include hypertension, constipation, and Raynaud phenomenon.
The position statement is strong and is based on a lot of evidence and clinical experience. CGRP-targeting therapies are now first-line agents for the prevention of migraine headache. We should learn more about and begin to feel comfortable using this class of agents because they stand to benefit our patients greatly. I’d suggest looking at the table below and picking one new agent to become familiar with so that you can add that agent to your toolbox. 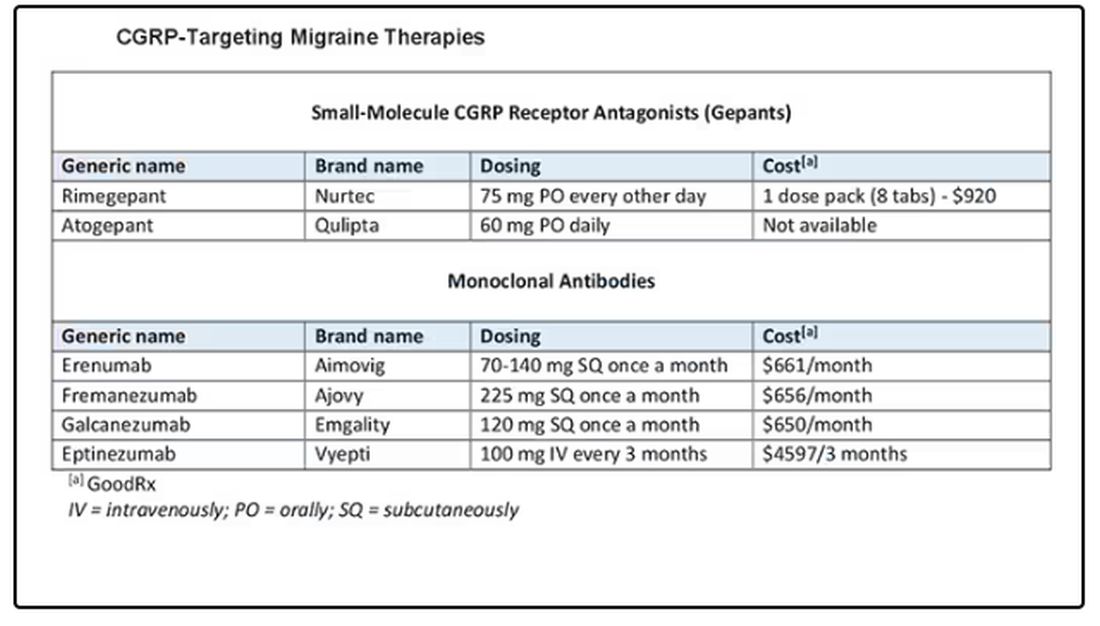
Dr. Skolnik, professor, Department of Family Medicine, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, Pennsylvania, and associate director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, Bayer, and Teva.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I am going to talk about the position statement from the American Headache Society (AHS) “Calcitonin gene-related peptide [CGRP]–targeting therapies are a first-line option for the prevention of migraine”. This update is of critical importance because about three fourths of people with migraine get their care from a primary care clinician, not from a neurologist or a headache specialist. CGRP-targeting therapies have transformed migraine care at the specialty level, but many in primary care are not yet familiar with this class of medicines. Until this new statement was released, CGRPs were not viewed as first-line agents for migraine. That has now changed.
Two main types of therapy for people with migraine headache are: (1) acute or abortive therapy (when a headache develops, it is treated), and (2) preventive therapy. Preventive therapy is typically used when the patient has headaches on 4 or more days per month. Preventive therapy is aimed at reducing the frequency and severity of headaches. About 40% of patients with migraine qualify for preventive therapy, but only a minority are receiving it.
The armamentarium for preventive therapy of migraines had not changed in a long time — until now. First-line preventive therapy has traditionally consisted of three classes of agents: beta-blockers, tricyclic antidepressants, and topiramate. These medicines were developed for different therapeutic purposes, yet they work for migraines. These drugs may have off-target effects that can make them difficult to tolerate.
Based on new evidence, candesartan — an angiotensin receptor blocker (ARB) — is now also a first-line drug for migraine. This is good news, because ARBs are a drug class that we have a lot of experience with, are easy to use, and could be an excellent choice for people with concomitant hypertension or chronic kidney disease. The serotonin-norepinephrine reuptake inhibitors (venlafaxine and duloxetine) are also considered first-line agents for migraine treatment.
In the AHS’s new position statement, the two main drug classes are small-molecule CGRP receptor antagonists and monoclonal antibodies.
The role of the neuropeptide CGRP in migraine was originally discovered after finding that blood levels of CGRP were elevated during migraine attacks. This led to the discovery of agents that blocked CGRP, initially for acute treatment of migraine, and then for preventive therapy. Multiple clinical studies show the CGRP targeting therapies to be as or even more effective than traditional first-line agents at decreasing the number of migraine days per month.
The efficacy and safety of these agents have been demonstrated in both randomized trials and in real-world studies. Other important positive endpoints include fewer days of migraine, reduced acute medication use, and improvements in many quality-of-life outcomes. Studies also have shown that CGRP-targeting therapies are well tolerated and safe, with very few serious adverse events.
Furthermore, studies have shown the CGRP targeting therapies are effective in individuals who have failed multiple other first-line therapies. They fit now both as first-line agents and as agents that can be used in difficult-to-treat patients as well as in patients who struggle with acute medication overuse, which is often very challenging.
To quote from the AHS statement,
Side effects are uncommon and can include hypertension, constipation, and Raynaud phenomenon.
The position statement is strong and is based on a lot of evidence and clinical experience. CGRP-targeting therapies are now first-line agents for the prevention of migraine headache. We should learn more about and begin to feel comfortable using this class of agents because they stand to benefit our patients greatly. I’d suggest looking at the table below and picking one new agent to become familiar with so that you can add that agent to your toolbox. 
Dr. Skolnik, professor, Department of Family Medicine, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, Pennsylvania, and associate director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, Bayer, and Teva.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Today I am going to talk about the position statement from the American Headache Society (AHS) “Calcitonin gene-related peptide [CGRP]–targeting therapies are a first-line option for the prevention of migraine”. This update is of critical importance because about three fourths of people with migraine get their care from a primary care clinician, not from a neurologist or a headache specialist. CGRP-targeting therapies have transformed migraine care at the specialty level, but many in primary care are not yet familiar with this class of medicines. Until this new statement was released, CGRPs were not viewed as first-line agents for migraine. That has now changed.
Two main types of therapy for people with migraine headache are: (1) acute or abortive therapy (when a headache develops, it is treated), and (2) preventive therapy. Preventive therapy is typically used when the patient has headaches on 4 or more days per month. Preventive therapy is aimed at reducing the frequency and severity of headaches. About 40% of patients with migraine qualify for preventive therapy, but only a minority are receiving it.
The armamentarium for preventive therapy of migraines had not changed in a long time — until now. First-line preventive therapy has traditionally consisted of three classes of agents: beta-blockers, tricyclic antidepressants, and topiramate. These medicines were developed for different therapeutic purposes, yet they work for migraines. These drugs may have off-target effects that can make them difficult to tolerate.
Based on new evidence, candesartan — an angiotensin receptor blocker (ARB) — is now also a first-line drug for migraine. This is good news, because ARBs are a drug class that we have a lot of experience with, are easy to use, and could be an excellent choice for people with concomitant hypertension or chronic kidney disease. The serotonin-norepinephrine reuptake inhibitors (venlafaxine and duloxetine) are also considered first-line agents for migraine treatment.
In the AHS’s new position statement, the two main drug classes are small-molecule CGRP receptor antagonists and monoclonal antibodies.
The role of the neuropeptide CGRP in migraine was originally discovered after finding that blood levels of CGRP were elevated during migraine attacks. This led to the discovery of agents that blocked CGRP, initially for acute treatment of migraine, and then for preventive therapy. Multiple clinical studies show the CGRP targeting therapies to be as or even more effective than traditional first-line agents at decreasing the number of migraine days per month.
The efficacy and safety of these agents have been demonstrated in both randomized trials and in real-world studies. Other important positive endpoints include fewer days of migraine, reduced acute medication use, and improvements in many quality-of-life outcomes. Studies also have shown that CGRP-targeting therapies are well tolerated and safe, with very few serious adverse events.
Furthermore, studies have shown the CGRP targeting therapies are effective in individuals who have failed multiple other first-line therapies. They fit now both as first-line agents and as agents that can be used in difficult-to-treat patients as well as in patients who struggle with acute medication overuse, which is often very challenging.
To quote from the AHS statement,
Side effects are uncommon and can include hypertension, constipation, and Raynaud phenomenon.
The position statement is strong and is based on a lot of evidence and clinical experience. CGRP-targeting therapies are now first-line agents for the prevention of migraine headache. We should learn more about and begin to feel comfortable using this class of agents because they stand to benefit our patients greatly. I’d suggest looking at the table below and picking one new agent to become familiar with so that you can add that agent to your toolbox. 
Dr. Skolnik, professor, Department of Family Medicine, Sidney Kimmel Medical College of Thomas Jefferson University, Philadelphia, Pennsylvania, and associate director, Department of Family Medicine, Abington Jefferson Health, Abington, Pennsylvania, disclosed ties with AstraZeneca, Teva, Eli Lilly, Boehringer Ingelheim, Sanofi, Sanofi Pasteur, GlaxoSmithKline, Merck, Bayer, and Teva.
A version of this article appeared on Medscape.com.
How Clinicians Can Help Patients Navigate Psychedelics/Microdosing
Peter Grinspoon, MD, has some advice for clinicians when patients ask questions about microdosing of psychedelics: Keep the lines of communication open — and don’t be judgmental.
“If you’re dismissive or critical or sound like you’re judging them, then the patients just clam up,” said Dr. Grinspoon, a professor of medicine at Harvard Medical School and a primary care physician at Massachusetts General Hospital, both in Boston.
Psychedelic drugs are still illegal in the majority of states despite the growth of public interest in and use of these substances. That growth is evidenced by a flurry of workshops, reports, law enforcement seizures, and pressure by Congressional members for the Food and Drug Administration to approve new psychedelic drugs, just in the past year.
A recent study in JAMA Health Forum showed a nearly 14-fold increase in Google searches — from 7.9 to 105.6 per 10 million nationwide — for the term “microdosing” and related wording, between 2015 and 2023.
Two states — Oregon and Colorado — have decriminalized certain psychedelic drugs and are in various stages of establishing regulations and centers for prospective clients. Almost two dozen localities, like Ann Arbor, Michigan, have decriminalized psychedelic drugs. A handful of states have active legislation to decriminalize use, while others have bills that never made it out of committee.
But no definitive studies have reported that microdosing produces positive mental effects at a higher rate than placebo, according to Dr. Grinspoon. So
“We’re in this renaissance where everybody is idealizing these medications, as opposed to 20 years ago when we were in the war on drugs and everybody was dismissing them,” Dr. Grinspoon said. “The truth is somewhere in between.”
The Science
Microdosing is defined as taking doses of 1/5 to 1/20 of the conventional recreational amount, which might include a dried psilocybin mushroom, lysergic acid diethylamide, or 3,4-methylenedioxymethamphetamine. But even that much may be neither effective nor safe.
Dr. Grinspoon said clinicians should tell patients that psychedelics may cause harm, although the drugs are relatively nontoxic and are not addictive. An illegally obtained psilocybin could cause negative reactions, especially if the drug has been adulterated with other substances and if the actual dose is higher than what was indicated by the seller.
He noted that people have different reactions to psychedelics, just as they have to prescription medications. He cited one example of a woman who microdosed and could not sleep for 2 weeks afterward. Only recently have randomized, double-blinded studies begun on benefits and harms.
Researchers have also begun investigating whether long-term microdosing of psilocybin could lead to valvular heart disease (VHD), said Kevin Yang, MD, a psychiatry resident at the University of California San Diego School of Medicine. A recent review of evidence concluded that microdosing various psychedelics over a period of months can lead to drug-induced VHD.
“It’s extremely important to emphasize with patients that not only do we not know if it works or not, we also don’t really know how safe it is,” Dr. Yang said.
Dr. Yang also said clinicians should consider referring patients to a mental health professional, and especially those that may have expertise in psychedelic therapies.
One of those experts is Rachel Yehuda, PhD, director of the Center for Psychedelic Psychotherapy and Trauma Research at Icahn School of Medicine at Mount Sinai in New York City. She said therapists should be able to assess the patient’s perceived need for microdosing and “invite reflections about why current approaches are falling short.”
“I would also not actively discourage it either but remain curious until both of you have a better understanding of the reasons for seeking this out and potential alternative strategies for obtaining more therapeutic benefits,” she said. “I think it is really important to study the effects of both micro- and macrodosing of psychedelics but not move in advance of the data.”
Navigating Legality
Recent ballot measures in Oregon and Colorado directed the states to develop regulated and licensed psilocybin-assisted therapy centers for legal “trips.” Oregon’s first center was opened in 2023, and Colorado is now developing its own licensing model.
According to the Oregon Health Authority, the centers are not medical facilities, and prescription or referral from a medical professional is not required.
The Oregon Academy of Family Physicians (OAFP) has yet to release guidance to clinicians on how to talk to their patients about these drugs or potential interest in visiting a licensed therapy center.
However, Betsy Boyd-Flynn, executive director of OAFP, said the organization is working on continuing medical education for what the average family physician needs to know if a patient asks about use.
“We suspect that many of our members have interest and want to learn more,” she said.
Dr. Grinspoon said clinicians should talk with patients about legality during these conversations.
“The big question I get is: ‘I really want to try microdosing, but how do I obtain the mushrooms?’ ” he said. “You can’t really as a physician tell them to do anything illegal. So you tell them to be safe, be careful, and to use their judgment.”
Patients who want to pursue microdosing who do not live in Oregon have two legal and safe options, Dr. Grinspoon said: Enroll in a clinical study or find a facility in a state or country — such as Oregon or Jamaica — that offers microdosing with psilocybin.
Clinicians also should warn their patients that the consequences of obtaining illicit psilocybin could exacerbate the mental health stresses they are seeking to alleviate.
“It’s going to get worse if they get tangled up with law enforcement or take something that’s contaminated and they get real sick,” he said.
Lisa Gillespie contributed reporting to this story. A version of this article appeared on Medscape.com.
Peter Grinspoon, MD, has some advice for clinicians when patients ask questions about microdosing of psychedelics: Keep the lines of communication open — and don’t be judgmental.
“If you’re dismissive or critical or sound like you’re judging them, then the patients just clam up,” said Dr. Grinspoon, a professor of medicine at Harvard Medical School and a primary care physician at Massachusetts General Hospital, both in Boston.
Psychedelic drugs are still illegal in the majority of states despite the growth of public interest in and use of these substances. That growth is evidenced by a flurry of workshops, reports, law enforcement seizures, and pressure by Congressional members for the Food and Drug Administration to approve new psychedelic drugs, just in the past year.
A recent study in JAMA Health Forum showed a nearly 14-fold increase in Google searches — from 7.9 to 105.6 per 10 million nationwide — for the term “microdosing” and related wording, between 2015 and 2023.
Two states — Oregon and Colorado — have decriminalized certain psychedelic drugs and are in various stages of establishing regulations and centers for prospective clients. Almost two dozen localities, like Ann Arbor, Michigan, have decriminalized psychedelic drugs. A handful of states have active legislation to decriminalize use, while others have bills that never made it out of committee.
But no definitive studies have reported that microdosing produces positive mental effects at a higher rate than placebo, according to Dr. Grinspoon. So
“We’re in this renaissance where everybody is idealizing these medications, as opposed to 20 years ago when we were in the war on drugs and everybody was dismissing them,” Dr. Grinspoon said. “The truth is somewhere in between.”
The Science
Microdosing is defined as taking doses of 1/5 to 1/20 of the conventional recreational amount, which might include a dried psilocybin mushroom, lysergic acid diethylamide, or 3,4-methylenedioxymethamphetamine. But even that much may be neither effective nor safe.
Dr. Grinspoon said clinicians should tell patients that psychedelics may cause harm, although the drugs are relatively nontoxic and are not addictive. An illegally obtained psilocybin could cause negative reactions, especially if the drug has been adulterated with other substances and if the actual dose is higher than what was indicated by the seller.
He noted that people have different reactions to psychedelics, just as they have to prescription medications. He cited one example of a woman who microdosed and could not sleep for 2 weeks afterward. Only recently have randomized, double-blinded studies begun on benefits and harms.
Researchers have also begun investigating whether long-term microdosing of psilocybin could lead to valvular heart disease (VHD), said Kevin Yang, MD, a psychiatry resident at the University of California San Diego School of Medicine. A recent review of evidence concluded that microdosing various psychedelics over a period of months can lead to drug-induced VHD.
“It’s extremely important to emphasize with patients that not only do we not know if it works or not, we also don’t really know how safe it is,” Dr. Yang said.
Dr. Yang also said clinicians should consider referring patients to a mental health professional, and especially those that may have expertise in psychedelic therapies.
One of those experts is Rachel Yehuda, PhD, director of the Center for Psychedelic Psychotherapy and Trauma Research at Icahn School of Medicine at Mount Sinai in New York City. She said therapists should be able to assess the patient’s perceived need for microdosing and “invite reflections about why current approaches are falling short.”
“I would also not actively discourage it either but remain curious until both of you have a better understanding of the reasons for seeking this out and potential alternative strategies for obtaining more therapeutic benefits,” she said. “I think it is really important to study the effects of both micro- and macrodosing of psychedelics but not move in advance of the data.”
Navigating Legality
Recent ballot measures in Oregon and Colorado directed the states to develop regulated and licensed psilocybin-assisted therapy centers for legal “trips.” Oregon’s first center was opened in 2023, and Colorado is now developing its own licensing model.
According to the Oregon Health Authority, the centers are not medical facilities, and prescription or referral from a medical professional is not required.
The Oregon Academy of Family Physicians (OAFP) has yet to release guidance to clinicians on how to talk to their patients about these drugs or potential interest in visiting a licensed therapy center.
However, Betsy Boyd-Flynn, executive director of OAFP, said the organization is working on continuing medical education for what the average family physician needs to know if a patient asks about use.
“We suspect that many of our members have interest and want to learn more,” she said.
Dr. Grinspoon said clinicians should talk with patients about legality during these conversations.
“The big question I get is: ‘I really want to try microdosing, but how do I obtain the mushrooms?’ ” he said. “You can’t really as a physician tell them to do anything illegal. So you tell them to be safe, be careful, and to use their judgment.”
Patients who want to pursue microdosing who do not live in Oregon have two legal and safe options, Dr. Grinspoon said: Enroll in a clinical study or find a facility in a state or country — such as Oregon or Jamaica — that offers microdosing with psilocybin.
Clinicians also should warn their patients that the consequences of obtaining illicit psilocybin could exacerbate the mental health stresses they are seeking to alleviate.
“It’s going to get worse if they get tangled up with law enforcement or take something that’s contaminated and they get real sick,” he said.
Lisa Gillespie contributed reporting to this story. A version of this article appeared on Medscape.com.
Peter Grinspoon, MD, has some advice for clinicians when patients ask questions about microdosing of psychedelics: Keep the lines of communication open — and don’t be judgmental.
“If you’re dismissive or critical or sound like you’re judging them, then the patients just clam up,” said Dr. Grinspoon, a professor of medicine at Harvard Medical School and a primary care physician at Massachusetts General Hospital, both in Boston.
Psychedelic drugs are still illegal in the majority of states despite the growth of public interest in and use of these substances. That growth is evidenced by a flurry of workshops, reports, law enforcement seizures, and pressure by Congressional members for the Food and Drug Administration to approve new psychedelic drugs, just in the past year.
A recent study in JAMA Health Forum showed a nearly 14-fold increase in Google searches — from 7.9 to 105.6 per 10 million nationwide — for the term “microdosing” and related wording, between 2015 and 2023.
Two states — Oregon and Colorado — have decriminalized certain psychedelic drugs and are in various stages of establishing regulations and centers for prospective clients. Almost two dozen localities, like Ann Arbor, Michigan, have decriminalized psychedelic drugs. A handful of states have active legislation to decriminalize use, while others have bills that never made it out of committee.
But no definitive studies have reported that microdosing produces positive mental effects at a higher rate than placebo, according to Dr. Grinspoon. So
“We’re in this renaissance where everybody is idealizing these medications, as opposed to 20 years ago when we were in the war on drugs and everybody was dismissing them,” Dr. Grinspoon said. “The truth is somewhere in between.”
The Science
Microdosing is defined as taking doses of 1/5 to 1/20 of the conventional recreational amount, which might include a dried psilocybin mushroom, lysergic acid diethylamide, or 3,4-methylenedioxymethamphetamine. But even that much may be neither effective nor safe.
Dr. Grinspoon said clinicians should tell patients that psychedelics may cause harm, although the drugs are relatively nontoxic and are not addictive. An illegally obtained psilocybin could cause negative reactions, especially if the drug has been adulterated with other substances and if the actual dose is higher than what was indicated by the seller.
He noted that people have different reactions to psychedelics, just as they have to prescription medications. He cited one example of a woman who microdosed and could not sleep for 2 weeks afterward. Only recently have randomized, double-blinded studies begun on benefits and harms.
Researchers have also begun investigating whether long-term microdosing of psilocybin could lead to valvular heart disease (VHD), said Kevin Yang, MD, a psychiatry resident at the University of California San Diego School of Medicine. A recent review of evidence concluded that microdosing various psychedelics over a period of months can lead to drug-induced VHD.
“It’s extremely important to emphasize with patients that not only do we not know if it works or not, we also don’t really know how safe it is,” Dr. Yang said.
Dr. Yang also said clinicians should consider referring patients to a mental health professional, and especially those that may have expertise in psychedelic therapies.
One of those experts is Rachel Yehuda, PhD, director of the Center for Psychedelic Psychotherapy and Trauma Research at Icahn School of Medicine at Mount Sinai in New York City. She said therapists should be able to assess the patient’s perceived need for microdosing and “invite reflections about why current approaches are falling short.”
“I would also not actively discourage it either but remain curious until both of you have a better understanding of the reasons for seeking this out and potential alternative strategies for obtaining more therapeutic benefits,” she said. “I think it is really important to study the effects of both micro- and macrodosing of psychedelics but not move in advance of the data.”
Navigating Legality
Recent ballot measures in Oregon and Colorado directed the states to develop regulated and licensed psilocybin-assisted therapy centers for legal “trips.” Oregon’s first center was opened in 2023, and Colorado is now developing its own licensing model.
According to the Oregon Health Authority, the centers are not medical facilities, and prescription or referral from a medical professional is not required.
The Oregon Academy of Family Physicians (OAFP) has yet to release guidance to clinicians on how to talk to their patients about these drugs or potential interest in visiting a licensed therapy center.
However, Betsy Boyd-Flynn, executive director of OAFP, said the organization is working on continuing medical education for what the average family physician needs to know if a patient asks about use.
“We suspect that many of our members have interest and want to learn more,” she said.
Dr. Grinspoon said clinicians should talk with patients about legality during these conversations.
“The big question I get is: ‘I really want to try microdosing, but how do I obtain the mushrooms?’ ” he said. “You can’t really as a physician tell them to do anything illegal. So you tell them to be safe, be careful, and to use their judgment.”
Patients who want to pursue microdosing who do not live in Oregon have two legal and safe options, Dr. Grinspoon said: Enroll in a clinical study or find a facility in a state or country — such as Oregon or Jamaica — that offers microdosing with psilocybin.
Clinicians also should warn their patients that the consequences of obtaining illicit psilocybin could exacerbate the mental health stresses they are seeking to alleviate.
“It’s going to get worse if they get tangled up with law enforcement or take something that’s contaminated and they get real sick,” he said.
Lisa Gillespie contributed reporting to this story. A version of this article appeared on Medscape.com.
Anxiety Linked to a Threefold Increased Risk for Dementia
TOPLINE:
, new research shows.
METHODOLOGY:
- A total of 2132 participants aged 55-85 years (mean age, 76 years) were recruited from the Hunter Community Study. Of these, 53% were women.
- Participants were assessed over three different waves, 5 years apart. Demographic and health-related data were captured at wave 1.
- Researchers used the Kessler Psychological Distress Scale (K10) to measure anxiety at two points: Baseline (wave 1) and first follow-up (wave 2), with a 5-year interval between them. Anxiety was classified as chronic if present during both waves, resolved if only present at wave 1, and new if only appearing at wave 2.
- The primary outcome, incident all-cause dementia, during the follow-up period (maximum 13 years after baseline) was identified using the International Classification of Disease-10 codes.
TAKEAWAY:
- Out of 2132 cognitively healthy participants, 64 developed dementia, with an average time to diagnosis of 10 years. Chronic anxiety was linked to a 2.8-fold increased risk for dementia, while new-onset anxiety was associated with a 3.2-fold increased risk (P = .01).
- Participants younger than 70 years with chronic anxiety had a 4.6-fold increased risk for dementia (P = .03), and those with new-onset anxiety had a 7.2 times higher risk for dementia (P = .004).
- There was no significant risk for dementia in participants with anxiety that had resolved.
- Investigators speculated that individuals with anxiety were more likely to engage in unhealthy lifestyle behaviors, such as poor diet and smoking, which can lead to cardiovascular disease — a condition strongly associated with dementia.
IN PRACTICE:
“This prospective cohort study used causal inference methods to explore the role of anxiety in promoting the development of dementia,” lead author Kay Khaing, MMed, The University of Newcastle, Australia, wrote in a press release. “The findings suggest that anxiety may be a new risk factor to target in the prevention of dementia and also indicate that treating anxiety may reduce this risk.”
SOURCE:
Kay Khaing, MMed, of The University of Newcastle, Australia, led the study, which was published online in the Journal of the American Geriatrics Society.
LIMITATIONS:
Anxiety was measured using K10, which assessed symptoms experienced in the most recent 4 weeks, raising concerns about its accuracy over the entire observation period. The authors acknowledged that despite using a combination of the total K10 score and the anxiety subscale, the overlap of anxiety and depression might not be fully disentangled, leading to residual confounding by depression. Additionally, 33% of participants were lost to follow-up, and those lost had higher anxiety rates at baseline, potentially leading to missing cases of dementia and affecting the effect estimate.
DISCLOSURES:
This study did not report any funding or conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
TOPLINE:
, new research shows.
METHODOLOGY:
- A total of 2132 participants aged 55-85 years (mean age, 76 years) were recruited from the Hunter Community Study. Of these, 53% were women.
- Participants were assessed over three different waves, 5 years apart. Demographic and health-related data were captured at wave 1.
- Researchers used the Kessler Psychological Distress Scale (K10) to measure anxiety at two points: Baseline (wave 1) and first follow-up (wave 2), with a 5-year interval between them. Anxiety was classified as chronic if present during both waves, resolved if only present at wave 1, and new if only appearing at wave 2.
- The primary outcome, incident all-cause dementia, during the follow-up period (maximum 13 years after baseline) was identified using the International Classification of Disease-10 codes.
TAKEAWAY:
- Out of 2132 cognitively healthy participants, 64 developed dementia, with an average time to diagnosis of 10 years. Chronic anxiety was linked to a 2.8-fold increased risk for dementia, while new-onset anxiety was associated with a 3.2-fold increased risk (P = .01).
- Participants younger than 70 years with chronic anxiety had a 4.6-fold increased risk for dementia (P = .03), and those with new-onset anxiety had a 7.2 times higher risk for dementia (P = .004).
- There was no significant risk for dementia in participants with anxiety that had resolved.
- Investigators speculated that individuals with anxiety were more likely to engage in unhealthy lifestyle behaviors, such as poor diet and smoking, which can lead to cardiovascular disease — a condition strongly associated with dementia.
IN PRACTICE:
“This prospective cohort study used causal inference methods to explore the role of anxiety in promoting the development of dementia,” lead author Kay Khaing, MMed, The University of Newcastle, Australia, wrote in a press release. “The findings suggest that anxiety may be a new risk factor to target in the prevention of dementia and also indicate that treating anxiety may reduce this risk.”
SOURCE:
Kay Khaing, MMed, of The University of Newcastle, Australia, led the study, which was published online in the Journal of the American Geriatrics Society.
LIMITATIONS:
Anxiety was measured using K10, which assessed symptoms experienced in the most recent 4 weeks, raising concerns about its accuracy over the entire observation period. The authors acknowledged that despite using a combination of the total K10 score and the anxiety subscale, the overlap of anxiety and depression might not be fully disentangled, leading to residual confounding by depression. Additionally, 33% of participants were lost to follow-up, and those lost had higher anxiety rates at baseline, potentially leading to missing cases of dementia and affecting the effect estimate.
DISCLOSURES:
This study did not report any funding or conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
TOPLINE:
, new research shows.
METHODOLOGY:
- A total of 2132 participants aged 55-85 years (mean age, 76 years) were recruited from the Hunter Community Study. Of these, 53% were women.
- Participants were assessed over three different waves, 5 years apart. Demographic and health-related data were captured at wave 1.
- Researchers used the Kessler Psychological Distress Scale (K10) to measure anxiety at two points: Baseline (wave 1) and first follow-up (wave 2), with a 5-year interval between them. Anxiety was classified as chronic if present during both waves, resolved if only present at wave 1, and new if only appearing at wave 2.
- The primary outcome, incident all-cause dementia, during the follow-up period (maximum 13 years after baseline) was identified using the International Classification of Disease-10 codes.
TAKEAWAY:
- Out of 2132 cognitively healthy participants, 64 developed dementia, with an average time to diagnosis of 10 years. Chronic anxiety was linked to a 2.8-fold increased risk for dementia, while new-onset anxiety was associated with a 3.2-fold increased risk (P = .01).
- Participants younger than 70 years with chronic anxiety had a 4.6-fold increased risk for dementia (P = .03), and those with new-onset anxiety had a 7.2 times higher risk for dementia (P = .004).
- There was no significant risk for dementia in participants with anxiety that had resolved.
- Investigators speculated that individuals with anxiety were more likely to engage in unhealthy lifestyle behaviors, such as poor diet and smoking, which can lead to cardiovascular disease — a condition strongly associated with dementia.
IN PRACTICE:
“This prospective cohort study used causal inference methods to explore the role of anxiety in promoting the development of dementia,” lead author Kay Khaing, MMed, The University of Newcastle, Australia, wrote in a press release. “The findings suggest that anxiety may be a new risk factor to target in the prevention of dementia and also indicate that treating anxiety may reduce this risk.”
SOURCE:
Kay Khaing, MMed, of The University of Newcastle, Australia, led the study, which was published online in the Journal of the American Geriatrics Society.
LIMITATIONS:
Anxiety was measured using K10, which assessed symptoms experienced in the most recent 4 weeks, raising concerns about its accuracy over the entire observation period. The authors acknowledged that despite using a combination of the total K10 score and the anxiety subscale, the overlap of anxiety and depression might not be fully disentangled, leading to residual confounding by depression. Additionally, 33% of participants were lost to follow-up, and those lost had higher anxiety rates at baseline, potentially leading to missing cases of dementia and affecting the effect estimate.
DISCLOSURES:
This study did not report any funding or conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article appeared on Medscape.com.
Does Headache Surgery Really Work? Neurologists Remain Unconvinced
Jeffrey E. Janis, MD, is on a mission. The professor of plastic surgery, surgery, neurosurgery, and neurology at The Ohio State University Wexner Medical Center, Columbus, Ohio, wants to convince neurologists of the safety and efficacy of nerve decompression surgery for treatment-resistant headache. However, many neurologists remain unconvinced.
There’s 24 years of evidence behind this surgical technique across hundreds of different studies with different study designs,” Dr. Janis said.
Yet this treatment approach — surgery on peripheral nerves rather than the brain or spinal cord — hasn’t garnered much support from neurologists. A scan of the agenda of topics at the recently held 2024 annual meeting of the American Headache Society showed few if any studies or presentations on this topic. And neurologists this news organization spoke to said they believe the surgery is experimental and unproven.
Experts do agree drugs don’t work for all patients with migraines. Up to 30% of patients don’t respond to the “laundry list of medications” available to treat the condition, said Dr. Janis.
Many patients have also tried, and failed, alternative treatment approaches such as massage, acupuncture, craniosacral therapy, transdermal patches, electrical stimulation, cryoablation, neurostimulation, and radiofrequency ablation.
If nothing else works, is surgery for headaches the answer?
Long-Held Theory
The idea that pinched, irritated, or compressed peripheral nerves can trigger migraine attacks has been around for nearly 25 years. Studies suggest that in addition to migraine, nerve compression can lead to other headache conditions, including occipital neuralgia, supraorbital neuralgia , and post-traumatic headaches.
This has led to the development of surgical techniques to deactivate various compression trigger sites — what Dr. Janis calls “pinch points” — which could involve muscles, bone, fascia, blood vessels, or scar tissue from prior trauma or surgery.
The procedure is predominantly performed by plastic surgeons, but to a lesser degree by neurosurgeons and ear, nose, and throat specialists.
Target nerves in surgical interventions include those in the frontal region of the head above the eye, temporal region, neck region, and nasal region. Affected areas are usually identified either through patient self-reports or by using a nerve block agent such as lidocaine or Botox at specific points, Dr. Janis noted. If pain subsides after an injection, that location is marked as a target.
One of the barriers to referring complicated patients for surgery is that neurologists evaluating migraine treatments “speak a different language” than surgeons performing the procedure, said Dr. Janis.
Neurologists tend to focus on reduction in monthly migraine days (MMD), while surgeons typically use the Migraine Headache Index that incorporates the frequency, intensity, and duration of migraine attacks.
“Rather than try to convince somebody to speak a different language, we thought, why don’t we just learn their language so we can build bridges and take down barriers,” said Dr. Janis, coauthor of a systematic review and meta-analysis published online recently in Plastic and Reconstructive Surgery.
Investigators examined 19 studies in the review, including five randomized controlled trials (RCTs), published from January 2020 to September 2023, with a total of 1603 participants who were mostly female and ranged in age from 9 to 72 years. Study follow-ups extended from 6 to 38 months. All but three studies were carried out in the United States, and six different compression sites were addressed during surgery.
Investigators found that across studies and by a number of measures, migraine frequency and severity improved after surgery.
Monthly migraine days decreased by 36%-92% and the number of overall migraine attacks per month dropped 25%-87.5%. Patients also reported decreases in attack duration of 41%-75% and intensity of 28%-82% across studies.
“Even using the neurologist-standard language of monthly migraine days, this surgery works,” said Dr. Janis. “Now this is documented both in the surgical literature and the nonsurgical literature.”
The most common complications were ecchymosis, hair loss or thinning, itching, dryness, and rhinorrhea, all of which Dr. Janis described as “fairly minor.” Major complications such as intraoperative bleeding and wound dehiscence were rare, occurring in 1% or less of participants.
‘One And Done?’
These surgeries are usually done on an outpatient basis and generally offer long-term results, Dr. Janis said.
“The idea is one and done,” he said. “The literature around this type of surgery says that whatever type of effect you get at 1 year is likely to be permanent.”
The American Society of Plastic Surgeons agrees. A 2018 position paper developed by experts and commissioned by the society reports that the intervention is safe and effective for appropriate patients, based on a comprehensive literature search and review of a large body of peer-reviewed scientific evidence.
“There is substantial, extensively replicated clinical data that demonstrates a significant reduction in [migraine headache] symptoms and frequency (even complete elimination of headache pain) following trigger site surgery,” the authors noted.
Pamela Blake, MD, a neurologist, board-certified headache specialist, and medical director at the Headache Center of River Oaks, Houston, is a proponent of what she said can be “lifesaving” headache surgery.
“If a doctor told you that you can either treat this problem with medications that you’ll need to take for the rest of your life or you can have a surgical procedure as an outpatient that has extremely low risk and has, in my experience, a 75% chance of reducing or eliminating your pain, you probably would be interested in surgery,” she said.
Continued Skepticism
However, other neurologists and clinicians appear doubtful about this intervention, including Hans-Christoph Diener, MD, PhD, professor of neurology and director, Essen Headache Centre, University of Duisburg-Essen in Germany.
During a debate on the topic a decade ago at the International Headache Congress, Dr. Diener argued that, as migraine is a complex multigene-related disorder of the brain, it doesn’t make sense that surgery would affect the epigenetics of 22 different genes.
Recently, he said that his views have not changed.
The topic remains controversial, and some neurologists are uncomfortable even openly discussing the procedure. Two clinicians who previously commented on this article later asked not to be included.
One neurologist, who asked to remain anonymous, said that Dr. Janis’s review article is “merely a review collecting 19 studies over the previous 10-plus years.”
Other limitations cited by this neurologist are the lack of consistency in procedures among the various studies and the inclusion of only four RCTs, the most recent of which was published 8 years ago, suggesting “the study was probably done closer to 9 or 10 years ago,” the neurologist said.
Dr. Blake suggested some neurologists’ reluctance could be due to limited background on the procedure, which she said isn’t widely discussed at headache meetings and is covered mostly in plastic surgery journals, not neurology literature. Access to surgery is further limited by a lack of specialists who perform the procedure and inconsistent insurance coverage.
A closer collaboration between neurologists and surgeons who perform the procedure could benefit patients, Dr. Blake noted.
“The headache doctor’s role is to identify who’s a candidate for surgery, who meets the criteria for nerve compression, and then follow that patient postoperatively, managing their medications, although usually we get them off their medications,” she added.
From Dr. Janis’s perspective, things are starting to change.
“I’m definitely seeing a greater comfort level among neurologists who are understanding where this sits in the algorithm for treatment, especially for complicated patients,” he said.
Dr. Janis receives royalties from Thieme and Springer Publishing. Dr. Blake reported no relevant conflicts. Dr. Diener received research support from the German Research Council; serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs; and has received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva, Weber & Weber, and WebMD.
A version of this article appeared on Medscape.com.
Jeffrey E. Janis, MD, is on a mission. The professor of plastic surgery, surgery, neurosurgery, and neurology at The Ohio State University Wexner Medical Center, Columbus, Ohio, wants to convince neurologists of the safety and efficacy of nerve decompression surgery for treatment-resistant headache. However, many neurologists remain unconvinced.
There’s 24 years of evidence behind this surgical technique across hundreds of different studies with different study designs,” Dr. Janis said.
Yet this treatment approach — surgery on peripheral nerves rather than the brain or spinal cord — hasn’t garnered much support from neurologists. A scan of the agenda of topics at the recently held 2024 annual meeting of the American Headache Society showed few if any studies or presentations on this topic. And neurologists this news organization spoke to said they believe the surgery is experimental and unproven.
Experts do agree drugs don’t work for all patients with migraines. Up to 30% of patients don’t respond to the “laundry list of medications” available to treat the condition, said Dr. Janis.
Many patients have also tried, and failed, alternative treatment approaches such as massage, acupuncture, craniosacral therapy, transdermal patches, electrical stimulation, cryoablation, neurostimulation, and radiofrequency ablation.
If nothing else works, is surgery for headaches the answer?
Long-Held Theory
The idea that pinched, irritated, or compressed peripheral nerves can trigger migraine attacks has been around for nearly 25 years. Studies suggest that in addition to migraine, nerve compression can lead to other headache conditions, including occipital neuralgia, supraorbital neuralgia , and post-traumatic headaches.
This has led to the development of surgical techniques to deactivate various compression trigger sites — what Dr. Janis calls “pinch points” — which could involve muscles, bone, fascia, blood vessels, or scar tissue from prior trauma or surgery.
The procedure is predominantly performed by plastic surgeons, but to a lesser degree by neurosurgeons and ear, nose, and throat specialists.
Target nerves in surgical interventions include those in the frontal region of the head above the eye, temporal region, neck region, and nasal region. Affected areas are usually identified either through patient self-reports or by using a nerve block agent such as lidocaine or Botox at specific points, Dr. Janis noted. If pain subsides after an injection, that location is marked as a target.
One of the barriers to referring complicated patients for surgery is that neurologists evaluating migraine treatments “speak a different language” than surgeons performing the procedure, said Dr. Janis.
Neurologists tend to focus on reduction in monthly migraine days (MMD), while surgeons typically use the Migraine Headache Index that incorporates the frequency, intensity, and duration of migraine attacks.
“Rather than try to convince somebody to speak a different language, we thought, why don’t we just learn their language so we can build bridges and take down barriers,” said Dr. Janis, coauthor of a systematic review and meta-analysis published online recently in Plastic and Reconstructive Surgery.
Investigators examined 19 studies in the review, including five randomized controlled trials (RCTs), published from January 2020 to September 2023, with a total of 1603 participants who were mostly female and ranged in age from 9 to 72 years. Study follow-ups extended from 6 to 38 months. All but three studies were carried out in the United States, and six different compression sites were addressed during surgery.
Investigators found that across studies and by a number of measures, migraine frequency and severity improved after surgery.
Monthly migraine days decreased by 36%-92% and the number of overall migraine attacks per month dropped 25%-87.5%. Patients also reported decreases in attack duration of 41%-75% and intensity of 28%-82% across studies.
“Even using the neurologist-standard language of monthly migraine days, this surgery works,” said Dr. Janis. “Now this is documented both in the surgical literature and the nonsurgical literature.”
The most common complications were ecchymosis, hair loss or thinning, itching, dryness, and rhinorrhea, all of which Dr. Janis described as “fairly minor.” Major complications such as intraoperative bleeding and wound dehiscence were rare, occurring in 1% or less of participants.
‘One And Done?’
These surgeries are usually done on an outpatient basis and generally offer long-term results, Dr. Janis said.
“The idea is one and done,” he said. “The literature around this type of surgery says that whatever type of effect you get at 1 year is likely to be permanent.”
The American Society of Plastic Surgeons agrees. A 2018 position paper developed by experts and commissioned by the society reports that the intervention is safe and effective for appropriate patients, based on a comprehensive literature search and review of a large body of peer-reviewed scientific evidence.
“There is substantial, extensively replicated clinical data that demonstrates a significant reduction in [migraine headache] symptoms and frequency (even complete elimination of headache pain) following trigger site surgery,” the authors noted.
Pamela Blake, MD, a neurologist, board-certified headache specialist, and medical director at the Headache Center of River Oaks, Houston, is a proponent of what she said can be “lifesaving” headache surgery.
“If a doctor told you that you can either treat this problem with medications that you’ll need to take for the rest of your life or you can have a surgical procedure as an outpatient that has extremely low risk and has, in my experience, a 75% chance of reducing or eliminating your pain, you probably would be interested in surgery,” she said.
Continued Skepticism
However, other neurologists and clinicians appear doubtful about this intervention, including Hans-Christoph Diener, MD, PhD, professor of neurology and director, Essen Headache Centre, University of Duisburg-Essen in Germany.
During a debate on the topic a decade ago at the International Headache Congress, Dr. Diener argued that, as migraine is a complex multigene-related disorder of the brain, it doesn’t make sense that surgery would affect the epigenetics of 22 different genes.
Recently, he said that his views have not changed.
The topic remains controversial, and some neurologists are uncomfortable even openly discussing the procedure. Two clinicians who previously commented on this article later asked not to be included.
One neurologist, who asked to remain anonymous, said that Dr. Janis’s review article is “merely a review collecting 19 studies over the previous 10-plus years.”
Other limitations cited by this neurologist are the lack of consistency in procedures among the various studies and the inclusion of only four RCTs, the most recent of which was published 8 years ago, suggesting “the study was probably done closer to 9 or 10 years ago,” the neurologist said.
Dr. Blake suggested some neurologists’ reluctance could be due to limited background on the procedure, which she said isn’t widely discussed at headache meetings and is covered mostly in plastic surgery journals, not neurology literature. Access to surgery is further limited by a lack of specialists who perform the procedure and inconsistent insurance coverage.
A closer collaboration between neurologists and surgeons who perform the procedure could benefit patients, Dr. Blake noted.
“The headache doctor’s role is to identify who’s a candidate for surgery, who meets the criteria for nerve compression, and then follow that patient postoperatively, managing their medications, although usually we get them off their medications,” she added.
From Dr. Janis’s perspective, things are starting to change.
“I’m definitely seeing a greater comfort level among neurologists who are understanding where this sits in the algorithm for treatment, especially for complicated patients,” he said.
Dr. Janis receives royalties from Thieme and Springer Publishing. Dr. Blake reported no relevant conflicts. Dr. Diener received research support from the German Research Council; serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs; and has received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva, Weber & Weber, and WebMD.
A version of this article appeared on Medscape.com.
Jeffrey E. Janis, MD, is on a mission. The professor of plastic surgery, surgery, neurosurgery, and neurology at The Ohio State University Wexner Medical Center, Columbus, Ohio, wants to convince neurologists of the safety and efficacy of nerve decompression surgery for treatment-resistant headache. However, many neurologists remain unconvinced.
There’s 24 years of evidence behind this surgical technique across hundreds of different studies with different study designs,” Dr. Janis said.
Yet this treatment approach — surgery on peripheral nerves rather than the brain or spinal cord — hasn’t garnered much support from neurologists. A scan of the agenda of topics at the recently held 2024 annual meeting of the American Headache Society showed few if any studies or presentations on this topic. And neurologists this news organization spoke to said they believe the surgery is experimental and unproven.
Experts do agree drugs don’t work for all patients with migraines. Up to 30% of patients don’t respond to the “laundry list of medications” available to treat the condition, said Dr. Janis.
Many patients have also tried, and failed, alternative treatment approaches such as massage, acupuncture, craniosacral therapy, transdermal patches, electrical stimulation, cryoablation, neurostimulation, and radiofrequency ablation.
If nothing else works, is surgery for headaches the answer?
Long-Held Theory
The idea that pinched, irritated, or compressed peripheral nerves can trigger migraine attacks has been around for nearly 25 years. Studies suggest that in addition to migraine, nerve compression can lead to other headache conditions, including occipital neuralgia, supraorbital neuralgia , and post-traumatic headaches.
This has led to the development of surgical techniques to deactivate various compression trigger sites — what Dr. Janis calls “pinch points” — which could involve muscles, bone, fascia, blood vessels, or scar tissue from prior trauma or surgery.
The procedure is predominantly performed by plastic surgeons, but to a lesser degree by neurosurgeons and ear, nose, and throat specialists.
Target nerves in surgical interventions include those in the frontal region of the head above the eye, temporal region, neck region, and nasal region. Affected areas are usually identified either through patient self-reports or by using a nerve block agent such as lidocaine or Botox at specific points, Dr. Janis noted. If pain subsides after an injection, that location is marked as a target.
One of the barriers to referring complicated patients for surgery is that neurologists evaluating migraine treatments “speak a different language” than surgeons performing the procedure, said Dr. Janis.
Neurologists tend to focus on reduction in monthly migraine days (MMD), while surgeons typically use the Migraine Headache Index that incorporates the frequency, intensity, and duration of migraine attacks.
“Rather than try to convince somebody to speak a different language, we thought, why don’t we just learn their language so we can build bridges and take down barriers,” said Dr. Janis, coauthor of a systematic review and meta-analysis published online recently in Plastic and Reconstructive Surgery.
Investigators examined 19 studies in the review, including five randomized controlled trials (RCTs), published from January 2020 to September 2023, with a total of 1603 participants who were mostly female and ranged in age from 9 to 72 years. Study follow-ups extended from 6 to 38 months. All but three studies were carried out in the United States, and six different compression sites were addressed during surgery.
Investigators found that across studies and by a number of measures, migraine frequency and severity improved after surgery.
Monthly migraine days decreased by 36%-92% and the number of overall migraine attacks per month dropped 25%-87.5%. Patients also reported decreases in attack duration of 41%-75% and intensity of 28%-82% across studies.
“Even using the neurologist-standard language of monthly migraine days, this surgery works,” said Dr. Janis. “Now this is documented both in the surgical literature and the nonsurgical literature.”
The most common complications were ecchymosis, hair loss or thinning, itching, dryness, and rhinorrhea, all of which Dr. Janis described as “fairly minor.” Major complications such as intraoperative bleeding and wound dehiscence were rare, occurring in 1% or less of participants.
‘One And Done?’
These surgeries are usually done on an outpatient basis and generally offer long-term results, Dr. Janis said.
“The idea is one and done,” he said. “The literature around this type of surgery says that whatever type of effect you get at 1 year is likely to be permanent.”
The American Society of Plastic Surgeons agrees. A 2018 position paper developed by experts and commissioned by the society reports that the intervention is safe and effective for appropriate patients, based on a comprehensive literature search and review of a large body of peer-reviewed scientific evidence.
“There is substantial, extensively replicated clinical data that demonstrates a significant reduction in [migraine headache] symptoms and frequency (even complete elimination of headache pain) following trigger site surgery,” the authors noted.
Pamela Blake, MD, a neurologist, board-certified headache specialist, and medical director at the Headache Center of River Oaks, Houston, is a proponent of what she said can be “lifesaving” headache surgery.
“If a doctor told you that you can either treat this problem with medications that you’ll need to take for the rest of your life or you can have a surgical procedure as an outpatient that has extremely low risk and has, in my experience, a 75% chance of reducing or eliminating your pain, you probably would be interested in surgery,” she said.
Continued Skepticism
However, other neurologists and clinicians appear doubtful about this intervention, including Hans-Christoph Diener, MD, PhD, professor of neurology and director, Essen Headache Centre, University of Duisburg-Essen in Germany.
During a debate on the topic a decade ago at the International Headache Congress, Dr. Diener argued that, as migraine is a complex multigene-related disorder of the brain, it doesn’t make sense that surgery would affect the epigenetics of 22 different genes.
Recently, he said that his views have not changed.
The topic remains controversial, and some neurologists are uncomfortable even openly discussing the procedure. Two clinicians who previously commented on this article later asked not to be included.
One neurologist, who asked to remain anonymous, said that Dr. Janis’s review article is “merely a review collecting 19 studies over the previous 10-plus years.”
Other limitations cited by this neurologist are the lack of consistency in procedures among the various studies and the inclusion of only four RCTs, the most recent of which was published 8 years ago, suggesting “the study was probably done closer to 9 or 10 years ago,” the neurologist said.
Dr. Blake suggested some neurologists’ reluctance could be due to limited background on the procedure, which she said isn’t widely discussed at headache meetings and is covered mostly in plastic surgery journals, not neurology literature. Access to surgery is further limited by a lack of specialists who perform the procedure and inconsistent insurance coverage.
A closer collaboration between neurologists and surgeons who perform the procedure could benefit patients, Dr. Blake noted.
“The headache doctor’s role is to identify who’s a candidate for surgery, who meets the criteria for nerve compression, and then follow that patient postoperatively, managing their medications, although usually we get them off their medications,” she added.
From Dr. Janis’s perspective, things are starting to change.
“I’m definitely seeing a greater comfort level among neurologists who are understanding where this sits in the algorithm for treatment, especially for complicated patients,” he said.
Dr. Janis receives royalties from Thieme and Springer Publishing. Dr. Blake reported no relevant conflicts. Dr. Diener received research support from the German Research Council; serves on the editorial boards of Cephalalgia, Lancet Neurology, and Drugs; and has received honoraria for participation in clinical trials, contribution to advisory boards, or oral presentations from AbbVie, Lilly, Lundbeck, Novartis, Pfizer, Teva, Weber & Weber, and WebMD.
A version of this article appeared on Medscape.com.
Maternal Obesity Linked to Sudden Infant Death
More than 5% of cases of sudden infant death may be linked to maternal obesity, new research showed.
“When a parent has a child that dies of sudden unexplained infant death [SUID], it’s extremely devastating,” said Jan-Marino Ramirez, PhD, the Zain Nadella Endowed Chair in Pediatric Neurosciences at the University of Washington, Seattle, and director of the Center for Integrative Brain Research at Seattle Children’s Hospital. “And the most devastating problem is that there’s no clear answer. Understanding the mechanisms will help parents understand.”
The study was published online in JAMA Pediatrics.
In the United States, approximately 3500 cases of SUID are reported yearly. After educational campaigns in the 1990s demonstrating safe infant sleep positions, rates of these fatalities dropped but have since plateaued.
Maternal Obesity During Pregnancy
Rates of maternal obesity are increasing globally, and more than half of women of reproductive age are overweight or obese.
“Maternal obesity before pregnancy affects placental development, gene expression, and has long-term implications,” said Patrick Catalano, MD, a professor in residence at the Departments of Reproductive Endocrinology and Obstetrics and Gynecology at Massachusetts General Hospital and Harvard Medical School in Boston.
Maternal obesity is a well-documented risk factor for adverse outcomes of pregnancy including stillbirth, preterm birth, and admission to the neonatal intensive care unit. Swedish researchers in 2014 reported maternal obesity was linked to an increase in infant mortality that increased with body mass index (BMI), but that study did not look specifically at SUID.
For their new study, Dr. Ramirez and colleagues looked at data from all live births in the United States from 2015 to 2019 recorded by the Centers for Disease Control and Prevention and the National Center for Health Statistics. Of the 18,857,694 live births occurring at 28 weeks of gestation or later, 16,545 infants died of a sudden, unexplained cause.
Rates of SUID in babies born to mothers with obesity increased in a statistically significant, dose-dependent manner relative to normal weight mothers. The unadjusted absolute risks for SUID were 0.74 cases per 1000 births for normal weight mothers, 0.99 cases at BMIs between 30 and 35, 1.17 cases at BMIs between 35 and 40, and 1.47 instances at BMI ≥ 40.
After adjustment for maternal age, race, ethnicity, and level of education, the adjusted odds ratio for a case of SUID was 1.39 among women with the highest levels of obesity (95% CI, 1.31-1.47), according to the researchers.
While the study revealed an association between maternal obesity and SUID, the basis for this connection remains unknown, the investigators noted. One possibility for the link is that obesity increases the risk for obstructive sleep apnea, which can result in intermittent hypoxia. That, in turn, causes oxidative stress, which may possibly have effects on the fetus causing effects that eventually lead to SUID in the infant.
An accompanying editorial by Jacqueline Maya, MD; Marie-France Hivert, MD, MMSc; and Lydia Shook, MD, from the Massachusetts General Hospital and Harvard Medical School, suggested that the SUID is unlikely directly influenced by high maternal BMI but rather by the metabolic concerns related to obesity such as inflammation, insulin resistance, and abnormal lipid metabolism. Epigenetics may also play a role.
“We believe the evidence for this study of an association between prepregnancy obesity and SUID is a call to action for the scientific and medical community to better understand the complex interplay of biological, social, and behavioral factors that may lead to SUID, a devastating complication that no family should experience,” the authors of the editorial wrote.
Dr. Ramirez stressed the importance of not initiating guilt because there are many factors in SUID such as genetics that cannot be controlled.
“We are far from saying a baby died because you were obese; that’s an important message to parents,” he said. What he sees as important, rather, is using this new research to elucidate further mechanisms that may allow for more targeted interventions: “If we discover that it’s due to, for example, sleep apnea, that’s something we can prevent.”
The researchers reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
More than 5% of cases of sudden infant death may be linked to maternal obesity, new research showed.
“When a parent has a child that dies of sudden unexplained infant death [SUID], it’s extremely devastating,” said Jan-Marino Ramirez, PhD, the Zain Nadella Endowed Chair in Pediatric Neurosciences at the University of Washington, Seattle, and director of the Center for Integrative Brain Research at Seattle Children’s Hospital. “And the most devastating problem is that there’s no clear answer. Understanding the mechanisms will help parents understand.”
The study was published online in JAMA Pediatrics.
In the United States, approximately 3500 cases of SUID are reported yearly. After educational campaigns in the 1990s demonstrating safe infant sleep positions, rates of these fatalities dropped but have since plateaued.
Maternal Obesity During Pregnancy
Rates of maternal obesity are increasing globally, and more than half of women of reproductive age are overweight or obese.
“Maternal obesity before pregnancy affects placental development, gene expression, and has long-term implications,” said Patrick Catalano, MD, a professor in residence at the Departments of Reproductive Endocrinology and Obstetrics and Gynecology at Massachusetts General Hospital and Harvard Medical School in Boston.
Maternal obesity is a well-documented risk factor for adverse outcomes of pregnancy including stillbirth, preterm birth, and admission to the neonatal intensive care unit. Swedish researchers in 2014 reported maternal obesity was linked to an increase in infant mortality that increased with body mass index (BMI), but that study did not look specifically at SUID.
For their new study, Dr. Ramirez and colleagues looked at data from all live births in the United States from 2015 to 2019 recorded by the Centers for Disease Control and Prevention and the National Center for Health Statistics. Of the 18,857,694 live births occurring at 28 weeks of gestation or later, 16,545 infants died of a sudden, unexplained cause.
Rates of SUID in babies born to mothers with obesity increased in a statistically significant, dose-dependent manner relative to normal weight mothers. The unadjusted absolute risks for SUID were 0.74 cases per 1000 births for normal weight mothers, 0.99 cases at BMIs between 30 and 35, 1.17 cases at BMIs between 35 and 40, and 1.47 instances at BMI ≥ 40.
After adjustment for maternal age, race, ethnicity, and level of education, the adjusted odds ratio for a case of SUID was 1.39 among women with the highest levels of obesity (95% CI, 1.31-1.47), according to the researchers.
While the study revealed an association between maternal obesity and SUID, the basis for this connection remains unknown, the investigators noted. One possibility for the link is that obesity increases the risk for obstructive sleep apnea, which can result in intermittent hypoxia. That, in turn, causes oxidative stress, which may possibly have effects on the fetus causing effects that eventually lead to SUID in the infant.
An accompanying editorial by Jacqueline Maya, MD; Marie-France Hivert, MD, MMSc; and Lydia Shook, MD, from the Massachusetts General Hospital and Harvard Medical School, suggested that the SUID is unlikely directly influenced by high maternal BMI but rather by the metabolic concerns related to obesity such as inflammation, insulin resistance, and abnormal lipid metabolism. Epigenetics may also play a role.
“We believe the evidence for this study of an association between prepregnancy obesity and SUID is a call to action for the scientific and medical community to better understand the complex interplay of biological, social, and behavioral factors that may lead to SUID, a devastating complication that no family should experience,” the authors of the editorial wrote.
Dr. Ramirez stressed the importance of not initiating guilt because there are many factors in SUID such as genetics that cannot be controlled.
“We are far from saying a baby died because you were obese; that’s an important message to parents,” he said. What he sees as important, rather, is using this new research to elucidate further mechanisms that may allow for more targeted interventions: “If we discover that it’s due to, for example, sleep apnea, that’s something we can prevent.”
The researchers reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
More than 5% of cases of sudden infant death may be linked to maternal obesity, new research showed.
“When a parent has a child that dies of sudden unexplained infant death [SUID], it’s extremely devastating,” said Jan-Marino Ramirez, PhD, the Zain Nadella Endowed Chair in Pediatric Neurosciences at the University of Washington, Seattle, and director of the Center for Integrative Brain Research at Seattle Children’s Hospital. “And the most devastating problem is that there’s no clear answer. Understanding the mechanisms will help parents understand.”
The study was published online in JAMA Pediatrics.
In the United States, approximately 3500 cases of SUID are reported yearly. After educational campaigns in the 1990s demonstrating safe infant sleep positions, rates of these fatalities dropped but have since plateaued.
Maternal Obesity During Pregnancy
Rates of maternal obesity are increasing globally, and more than half of women of reproductive age are overweight or obese.
“Maternal obesity before pregnancy affects placental development, gene expression, and has long-term implications,” said Patrick Catalano, MD, a professor in residence at the Departments of Reproductive Endocrinology and Obstetrics and Gynecology at Massachusetts General Hospital and Harvard Medical School in Boston.
Maternal obesity is a well-documented risk factor for adverse outcomes of pregnancy including stillbirth, preterm birth, and admission to the neonatal intensive care unit. Swedish researchers in 2014 reported maternal obesity was linked to an increase in infant mortality that increased with body mass index (BMI), but that study did not look specifically at SUID.
For their new study, Dr. Ramirez and colleagues looked at data from all live births in the United States from 2015 to 2019 recorded by the Centers for Disease Control and Prevention and the National Center for Health Statistics. Of the 18,857,694 live births occurring at 28 weeks of gestation or later, 16,545 infants died of a sudden, unexplained cause.
Rates of SUID in babies born to mothers with obesity increased in a statistically significant, dose-dependent manner relative to normal weight mothers. The unadjusted absolute risks for SUID were 0.74 cases per 1000 births for normal weight mothers, 0.99 cases at BMIs between 30 and 35, 1.17 cases at BMIs between 35 and 40, and 1.47 instances at BMI ≥ 40.
After adjustment for maternal age, race, ethnicity, and level of education, the adjusted odds ratio for a case of SUID was 1.39 among women with the highest levels of obesity (95% CI, 1.31-1.47), according to the researchers.
While the study revealed an association between maternal obesity and SUID, the basis for this connection remains unknown, the investigators noted. One possibility for the link is that obesity increases the risk for obstructive sleep apnea, which can result in intermittent hypoxia. That, in turn, causes oxidative stress, which may possibly have effects on the fetus causing effects that eventually lead to SUID in the infant.
An accompanying editorial by Jacqueline Maya, MD; Marie-France Hivert, MD, MMSc; and Lydia Shook, MD, from the Massachusetts General Hospital and Harvard Medical School, suggested that the SUID is unlikely directly influenced by high maternal BMI but rather by the metabolic concerns related to obesity such as inflammation, insulin resistance, and abnormal lipid metabolism. Epigenetics may also play a role.
“We believe the evidence for this study of an association between prepregnancy obesity and SUID is a call to action for the scientific and medical community to better understand the complex interplay of biological, social, and behavioral factors that may lead to SUID, a devastating complication that no family should experience,” the authors of the editorial wrote.
Dr. Ramirez stressed the importance of not initiating guilt because there are many factors in SUID such as genetics that cannot be controlled.
“We are far from saying a baby died because you were obese; that’s an important message to parents,” he said. What he sees as important, rather, is using this new research to elucidate further mechanisms that may allow for more targeted interventions: “If we discover that it’s due to, for example, sleep apnea, that’s something we can prevent.”
The researchers reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM JAMA PEDIATRICS
New Drugs Could Reduce AMD Treatment Burden
STOCKHOLM — Current treatments for age-related macular degeneration (AMD) have proved effective and safe. However, these lifelong therapies involve frequent ocular injections. “It can be nerve-wracking for patients about to embark on this journey,” Lisa Olmos de Koo, MD, an ophthalmologist at the University of Washington Eye Institute at Harborview, Seattle, told this news organization.
At the American Society of Retina Specialists (ASRS) 2024 annual meeting, researchers from around the world presented results from clinical studies aiming at reducing the burden of AMD treatment by:
- Identifying patients at a higher risk for degeneration and vision loss who will be more likely to respond to treatment
- Developing gene therapies that promise to drastically reduce or eliminate the need for injections
- Testing novel drugs with mechanisms of action that use different pathways than currently available medications, offering patients more options and longer-lasting treatments
“It’s exciting to see the broad range of novel approaches in AMD treatments,” Dimitra Skondra, MD, PhD, a retina specialist at the University of Chicago, told this news organization.
Whom to Treat
Anti–vascular endothelial growth factor (anti-VEGF) therapies shook the AMD treatment scene when they were introduced in the early 2000s. “It was incredible,” Dr. Olmos de Koo said. Patients with wet AMD could finally see their vision improve with each injection. “It was a great motivator to begin therapy.”
However, patients with the advanced form of dry AMD involving geographic atrophy (GA) have had less luck. Pegcetacoplan and avacincaptad pegol, the only US Food and Drug Administration (FDA)–approved treatments for GA, slow the progression of the disease but do not restore vision. In fact, vision continues to decline. “Patients want to understand if their condition is worsening and whether treatment is necessary,” Dr. Olmos de Koo said.
Researchers are developing tools to help clinicians identify lesions that are more likely to grow and reach the fovea, causing vision loss.
For example, Cleveland Clinic’s Katherine Talcott, MD, presented an analysis of the GATHER1 and GATHER2 clinical trials that showed that spectral domain optical coherence tomography can be used to examine the integrity of the ellipsoid zone for predicting GA growth and treatment response. The retina’s ellipsoid zone contains densely packed mitochondria within the inner segments of the photoreceptor cells and plays a critical role in visual function.
Dr. Talcott and her team found that more severe baseline damage of the ellipsoid zone was associated with a faster growth rate of GA.
Another analysis of the same trials, presented by Dilraj Grewal, MD, associate professor of ophthalmology, vitreoretinal surgery, and uveitis at Duke Eye Center, Durham, North Carolina, showed that intravitreal administration of avacincaptad pegol efficiently reduced GA growth whether the treated eye developed macular neovascularization or not. Avacincaptad pegol is a complement factor inhibitor that aims to reduce complement-mediated inflammation and tissue damage in the retina.
Dr. Olmos de Koo explained that clinical trials have shown that more patients develop neovascularization when treated for dry GA than they would if left untreated. This has raised the question among clinicians whether the increased risk is a valid reason to avoid treatment. “This useful analysis tells us that there is still a rationale to continue treating GA, even while you’re concurrently treating the wet component with anti-VEGF therapies,” she said.
Another biomarker of GA growth is the position of the lesion at baseline. Daniel Muth, MD, an ophthalmology consultant at the Karolinska Institutet in Stockholm, Sweden, reported the results from a long-term, retrospective analysis of fundus autofluorescence in patients with GA. His semiautomated artificial intelligence–based analysis showed that patients affected bilaterally, but whose fovea was not yet affected, exhibited a faster GA growth rate than fovea-involving patients, with an approximate 15% risk for fovea involvement.
“Those patients whose atrophy has not yet affected the very center are the most likely to benefit from preventive therapy,” Dr. Olmos de Koo said. “Left untreated, a large proportion of them will develop atrophy that does affect their central vision — that’s their reading or facial recognition ability.”
“Potential predictors of rapid growth rates guide us clinically and allow patients to make more informed decisions about whether to pursue treatments that require frequent interventions,” Dr. Olmos de Koo said.
Forecasting the side to which the cost-benefit balance of treatment will tip for each patient is a complex decision-making process, she explained. “A patient is not a statistic, but these predictive studies are one important piece of the pie.”
The Promise of Gene Therapy
The one-and-done promise of gene therapy could rattle the field once again. Trials presented at the ASRS24 showed a drastic reduction (from 85% to 95%) in the number of anti-VEGF and complement treatments needed following gene therapy injection, improving patient vision while relieving them from the stress of monthly injections.
But researchers are still debating the optimal corticosteroid regimen that is required for reducing the inflammatory response associated with the administration of gene therapies, especially those that use viral vectors. The main controversy is whether systemic immunosuppression is necessary or if local therapies, such as topical and intravitreal administration, can suffice.
Results presented at the meeting suggest that local therapies alone can be effective, potentially reducing the need for systemic immunosuppression.
The LUNA trial evaluated the efficacy and tolerability of ixoberogene soroparvovec, a therapy that delivers an anti-VEGF gene into the eye. Investigators included various prophylactic regimens, including local corticosteroids with and without oral prednisone. They found that local corticosteroid therapy alone effectively reduced inflammation.
Biopharma company 4DMT conducted the PRISM study, which examined a dual transgene therapy for neovascular AMD. Patients in this trial received a 20-week topical steroid taper. Only one patient (of 39) required a 6-week extension of steroid therapy. No patients experienced clinically significant intraocular inflammation, indicating that local corticosteroid therapy was effective in managing immune responses.
Currently, gene therapy clinical trials are designed for patients who have failed standard therapy or require frequent injections. “Once we figure out possible long-term side effects and how to deal with inflammation, [gene therapy] could reach many more patients,” Dr. Olmos de Koo said.
New Approaches Enter Pipeline
While gene therapy brings excitement to the field, it might not be for everyone, experts agreed at the ASRS24. New agents are being evaluated to offer a broader range of treatment options with longer-lasting efficacy. Results from early-phase trials presented at the meeting show favorable safety and efficacy signals.
“Finally, after a long time, we have a lot of exciting drugs for geographic atrophy in the pipeline that seem to be safe, with many studies also showing a functional outcome in addition to anatomical outcome,” Dr. Skondra told this news organization.
Current FDA-approved treatments for GA focus on inhibiting the humoral arm of the immune system through C3 and C5 inhibitors. However, a new approach targets both the humoral and cellular arms of the immune response by inhibiting macrophages that release pro-inflammatory cytokines. The goal is to convert these macrophages to a “resolution state,” potentially reducing the release of inflammatory cytokines and offering a more comprehensive treatment for wet and dry AMD, said Rishi Singh, MD, a retina surgeon at the Cole Eye Institute, Cleveland Clinic.
AVD-104, a sialic acid–coated nanoparticle developed by Aviceda Therapeutics, is a promising candidate in this approach. This 100-nm-in-diameter particle, which is only as heavy as 20 hydrogen atoms, is designed for better tissue penetration and has a pharmacokinetic profile lasting 3-4 months after a single intravitreal dose.
AVD-104 aims to repolarize macrophages into a resolution phenotype and decreases complement factor overamplification through direct binding to complement factor H, which downregulates C3 production in immune cells. This dual-action approach could offer a more effective and long-lasting treatment option.
Dr. Singh, who presented the phase 2/3 SIGLEC clinical trial assessing AVD-104, said a single dose resulted in significantly slower rates of disease progression as early as 1 month post-treatment and a notable decrease in junctional zone hyper-autofluorescence.
In addition, about 40% of patients gained vision, which was unexpected but a pleasant surprise, Dr. Singh said. “This is a small study. I don’t want anyone to walk away with the conclusion that we’ve figured out how to improve visual acuity in GA. But it’s promising.”
Other researchers are tackling GA by focusing on therapies that aim to intervene before the complement system is activated.
ONL1204 is a novel agent designed to inhibit the activation of the tumor necrosis factor FAS receptor, which is activated and upregulated in a disease state and is implicated in multiple cell death and inflammatory pathways.
Multiple preclinical models of AMD have shown that ONL1204 preserves retinal cells and inhibits inflammation by inhibiting the FAS receptor. Phase 1 trial results presented at the meeting showed that ONL1204 was safe and showed strong efficacy signals as early as 6 months after treatment initiation.
“We need to be cautiously optimistic,” Dr. Skondra said. “Larger studies will tell us if these signals are real. But it’s a very exciting time. I’m happy to see different mechanisms of action besides the complement because we can attack the disease from multiple fronts.”
Dr. Grewal declared interests with Eyepoint, Iveric Bio, Regeneron, Alumis, Apellis, DORC, and Genentech. Dr. Muth declared interests with Bayer, Canon, and Roche. Dr. Olmos de Koo declared interests with Alcon and Pixium Vision. Dr. Singh declared interests with Gyroscope, 4DMT, Aviceda, Eyepoint, Alcon, Bausch and Lomb, Novartis, and Regeneron. Dr. Skondra declared interests with Biogen, Iveric Bio, Allergan, and Trinity Health Science. Dr. Talcott declared interests with Bausch and Lomb, Eyepoint, Regeneron, REGENXBIO, Zeiss, Apellis, Genentech, Alimera, Outlook, and Iveric Bio.
A version of this article first appeared on Medscape.com.
STOCKHOLM — Current treatments for age-related macular degeneration (AMD) have proved effective and safe. However, these lifelong therapies involve frequent ocular injections. “It can be nerve-wracking for patients about to embark on this journey,” Lisa Olmos de Koo, MD, an ophthalmologist at the University of Washington Eye Institute at Harborview, Seattle, told this news organization.
At the American Society of Retina Specialists (ASRS) 2024 annual meeting, researchers from around the world presented results from clinical studies aiming at reducing the burden of AMD treatment by:
- Identifying patients at a higher risk for degeneration and vision loss who will be more likely to respond to treatment
- Developing gene therapies that promise to drastically reduce or eliminate the need for injections
- Testing novel drugs with mechanisms of action that use different pathways than currently available medications, offering patients more options and longer-lasting treatments
“It’s exciting to see the broad range of novel approaches in AMD treatments,” Dimitra Skondra, MD, PhD, a retina specialist at the University of Chicago, told this news organization.
Whom to Treat
Anti–vascular endothelial growth factor (anti-VEGF) therapies shook the AMD treatment scene when they were introduced in the early 2000s. “It was incredible,” Dr. Olmos de Koo said. Patients with wet AMD could finally see their vision improve with each injection. “It was a great motivator to begin therapy.”
However, patients with the advanced form of dry AMD involving geographic atrophy (GA) have had less luck. Pegcetacoplan and avacincaptad pegol, the only US Food and Drug Administration (FDA)–approved treatments for GA, slow the progression of the disease but do not restore vision. In fact, vision continues to decline. “Patients want to understand if their condition is worsening and whether treatment is necessary,” Dr. Olmos de Koo said.
Researchers are developing tools to help clinicians identify lesions that are more likely to grow and reach the fovea, causing vision loss.
For example, Cleveland Clinic’s Katherine Talcott, MD, presented an analysis of the GATHER1 and GATHER2 clinical trials that showed that spectral domain optical coherence tomography can be used to examine the integrity of the ellipsoid zone for predicting GA growth and treatment response. The retina’s ellipsoid zone contains densely packed mitochondria within the inner segments of the photoreceptor cells and plays a critical role in visual function.
Dr. Talcott and her team found that more severe baseline damage of the ellipsoid zone was associated with a faster growth rate of GA.
Another analysis of the same trials, presented by Dilraj Grewal, MD, associate professor of ophthalmology, vitreoretinal surgery, and uveitis at Duke Eye Center, Durham, North Carolina, showed that intravitreal administration of avacincaptad pegol efficiently reduced GA growth whether the treated eye developed macular neovascularization or not. Avacincaptad pegol is a complement factor inhibitor that aims to reduce complement-mediated inflammation and tissue damage in the retina.
Dr. Olmos de Koo explained that clinical trials have shown that more patients develop neovascularization when treated for dry GA than they would if left untreated. This has raised the question among clinicians whether the increased risk is a valid reason to avoid treatment. “This useful analysis tells us that there is still a rationale to continue treating GA, even while you’re concurrently treating the wet component with anti-VEGF therapies,” she said.
Another biomarker of GA growth is the position of the lesion at baseline. Daniel Muth, MD, an ophthalmology consultant at the Karolinska Institutet in Stockholm, Sweden, reported the results from a long-term, retrospective analysis of fundus autofluorescence in patients with GA. His semiautomated artificial intelligence–based analysis showed that patients affected bilaterally, but whose fovea was not yet affected, exhibited a faster GA growth rate than fovea-involving patients, with an approximate 15% risk for fovea involvement.
“Those patients whose atrophy has not yet affected the very center are the most likely to benefit from preventive therapy,” Dr. Olmos de Koo said. “Left untreated, a large proportion of them will develop atrophy that does affect their central vision — that’s their reading or facial recognition ability.”
“Potential predictors of rapid growth rates guide us clinically and allow patients to make more informed decisions about whether to pursue treatments that require frequent interventions,” Dr. Olmos de Koo said.
Forecasting the side to which the cost-benefit balance of treatment will tip for each patient is a complex decision-making process, she explained. “A patient is not a statistic, but these predictive studies are one important piece of the pie.”
The Promise of Gene Therapy
The one-and-done promise of gene therapy could rattle the field once again. Trials presented at the ASRS24 showed a drastic reduction (from 85% to 95%) in the number of anti-VEGF and complement treatments needed following gene therapy injection, improving patient vision while relieving them from the stress of monthly injections.
But researchers are still debating the optimal corticosteroid regimen that is required for reducing the inflammatory response associated with the administration of gene therapies, especially those that use viral vectors. The main controversy is whether systemic immunosuppression is necessary or if local therapies, such as topical and intravitreal administration, can suffice.
Results presented at the meeting suggest that local therapies alone can be effective, potentially reducing the need for systemic immunosuppression.
The LUNA trial evaluated the efficacy and tolerability of ixoberogene soroparvovec, a therapy that delivers an anti-VEGF gene into the eye. Investigators included various prophylactic regimens, including local corticosteroids with and without oral prednisone. They found that local corticosteroid therapy alone effectively reduced inflammation.
Biopharma company 4DMT conducted the PRISM study, which examined a dual transgene therapy for neovascular AMD. Patients in this trial received a 20-week topical steroid taper. Only one patient (of 39) required a 6-week extension of steroid therapy. No patients experienced clinically significant intraocular inflammation, indicating that local corticosteroid therapy was effective in managing immune responses.
Currently, gene therapy clinical trials are designed for patients who have failed standard therapy or require frequent injections. “Once we figure out possible long-term side effects and how to deal with inflammation, [gene therapy] could reach many more patients,” Dr. Olmos de Koo said.
New Approaches Enter Pipeline
While gene therapy brings excitement to the field, it might not be for everyone, experts agreed at the ASRS24. New agents are being evaluated to offer a broader range of treatment options with longer-lasting efficacy. Results from early-phase trials presented at the meeting show favorable safety and efficacy signals.
“Finally, after a long time, we have a lot of exciting drugs for geographic atrophy in the pipeline that seem to be safe, with many studies also showing a functional outcome in addition to anatomical outcome,” Dr. Skondra told this news organization.
Current FDA-approved treatments for GA focus on inhibiting the humoral arm of the immune system through C3 and C5 inhibitors. However, a new approach targets both the humoral and cellular arms of the immune response by inhibiting macrophages that release pro-inflammatory cytokines. The goal is to convert these macrophages to a “resolution state,” potentially reducing the release of inflammatory cytokines and offering a more comprehensive treatment for wet and dry AMD, said Rishi Singh, MD, a retina surgeon at the Cole Eye Institute, Cleveland Clinic.
AVD-104, a sialic acid–coated nanoparticle developed by Aviceda Therapeutics, is a promising candidate in this approach. This 100-nm-in-diameter particle, which is only as heavy as 20 hydrogen atoms, is designed for better tissue penetration and has a pharmacokinetic profile lasting 3-4 months after a single intravitreal dose.
AVD-104 aims to repolarize macrophages into a resolution phenotype and decreases complement factor overamplification through direct binding to complement factor H, which downregulates C3 production in immune cells. This dual-action approach could offer a more effective and long-lasting treatment option.
Dr. Singh, who presented the phase 2/3 SIGLEC clinical trial assessing AVD-104, said a single dose resulted in significantly slower rates of disease progression as early as 1 month post-treatment and a notable decrease in junctional zone hyper-autofluorescence.
In addition, about 40% of patients gained vision, which was unexpected but a pleasant surprise, Dr. Singh said. “This is a small study. I don’t want anyone to walk away with the conclusion that we’ve figured out how to improve visual acuity in GA. But it’s promising.”
Other researchers are tackling GA by focusing on therapies that aim to intervene before the complement system is activated.
ONL1204 is a novel agent designed to inhibit the activation of the tumor necrosis factor FAS receptor, which is activated and upregulated in a disease state and is implicated in multiple cell death and inflammatory pathways.
Multiple preclinical models of AMD have shown that ONL1204 preserves retinal cells and inhibits inflammation by inhibiting the FAS receptor. Phase 1 trial results presented at the meeting showed that ONL1204 was safe and showed strong efficacy signals as early as 6 months after treatment initiation.
“We need to be cautiously optimistic,” Dr. Skondra said. “Larger studies will tell us if these signals are real. But it’s a very exciting time. I’m happy to see different mechanisms of action besides the complement because we can attack the disease from multiple fronts.”
Dr. Grewal declared interests with Eyepoint, Iveric Bio, Regeneron, Alumis, Apellis, DORC, and Genentech. Dr. Muth declared interests with Bayer, Canon, and Roche. Dr. Olmos de Koo declared interests with Alcon and Pixium Vision. Dr. Singh declared interests with Gyroscope, 4DMT, Aviceda, Eyepoint, Alcon, Bausch and Lomb, Novartis, and Regeneron. Dr. Skondra declared interests with Biogen, Iveric Bio, Allergan, and Trinity Health Science. Dr. Talcott declared interests with Bausch and Lomb, Eyepoint, Regeneron, REGENXBIO, Zeiss, Apellis, Genentech, Alimera, Outlook, and Iveric Bio.
A version of this article first appeared on Medscape.com.
STOCKHOLM — Current treatments for age-related macular degeneration (AMD) have proved effective and safe. However, these lifelong therapies involve frequent ocular injections. “It can be nerve-wracking for patients about to embark on this journey,” Lisa Olmos de Koo, MD, an ophthalmologist at the University of Washington Eye Institute at Harborview, Seattle, told this news organization.
At the American Society of Retina Specialists (ASRS) 2024 annual meeting, researchers from around the world presented results from clinical studies aiming at reducing the burden of AMD treatment by:
- Identifying patients at a higher risk for degeneration and vision loss who will be more likely to respond to treatment
- Developing gene therapies that promise to drastically reduce or eliminate the need for injections
- Testing novel drugs with mechanisms of action that use different pathways than currently available medications, offering patients more options and longer-lasting treatments
“It’s exciting to see the broad range of novel approaches in AMD treatments,” Dimitra Skondra, MD, PhD, a retina specialist at the University of Chicago, told this news organization.
Whom to Treat
Anti–vascular endothelial growth factor (anti-VEGF) therapies shook the AMD treatment scene when they were introduced in the early 2000s. “It was incredible,” Dr. Olmos de Koo said. Patients with wet AMD could finally see their vision improve with each injection. “It was a great motivator to begin therapy.”
However, patients with the advanced form of dry AMD involving geographic atrophy (GA) have had less luck. Pegcetacoplan and avacincaptad pegol, the only US Food and Drug Administration (FDA)–approved treatments for GA, slow the progression of the disease but do not restore vision. In fact, vision continues to decline. “Patients want to understand if their condition is worsening and whether treatment is necessary,” Dr. Olmos de Koo said.
Researchers are developing tools to help clinicians identify lesions that are more likely to grow and reach the fovea, causing vision loss.
For example, Cleveland Clinic’s Katherine Talcott, MD, presented an analysis of the GATHER1 and GATHER2 clinical trials that showed that spectral domain optical coherence tomography can be used to examine the integrity of the ellipsoid zone for predicting GA growth and treatment response. The retina’s ellipsoid zone contains densely packed mitochondria within the inner segments of the photoreceptor cells and plays a critical role in visual function.
Dr. Talcott and her team found that more severe baseline damage of the ellipsoid zone was associated with a faster growth rate of GA.
Another analysis of the same trials, presented by Dilraj Grewal, MD, associate professor of ophthalmology, vitreoretinal surgery, and uveitis at Duke Eye Center, Durham, North Carolina, showed that intravitreal administration of avacincaptad pegol efficiently reduced GA growth whether the treated eye developed macular neovascularization or not. Avacincaptad pegol is a complement factor inhibitor that aims to reduce complement-mediated inflammation and tissue damage in the retina.
Dr. Olmos de Koo explained that clinical trials have shown that more patients develop neovascularization when treated for dry GA than they would if left untreated. This has raised the question among clinicians whether the increased risk is a valid reason to avoid treatment. “This useful analysis tells us that there is still a rationale to continue treating GA, even while you’re concurrently treating the wet component with anti-VEGF therapies,” she said.
Another biomarker of GA growth is the position of the lesion at baseline. Daniel Muth, MD, an ophthalmology consultant at the Karolinska Institutet in Stockholm, Sweden, reported the results from a long-term, retrospective analysis of fundus autofluorescence in patients with GA. His semiautomated artificial intelligence–based analysis showed that patients affected bilaterally, but whose fovea was not yet affected, exhibited a faster GA growth rate than fovea-involving patients, with an approximate 15% risk for fovea involvement.
“Those patients whose atrophy has not yet affected the very center are the most likely to benefit from preventive therapy,” Dr. Olmos de Koo said. “Left untreated, a large proportion of them will develop atrophy that does affect their central vision — that’s their reading or facial recognition ability.”
“Potential predictors of rapid growth rates guide us clinically and allow patients to make more informed decisions about whether to pursue treatments that require frequent interventions,” Dr. Olmos de Koo said.
Forecasting the side to which the cost-benefit balance of treatment will tip for each patient is a complex decision-making process, she explained. “A patient is not a statistic, but these predictive studies are one important piece of the pie.”
The Promise of Gene Therapy
The one-and-done promise of gene therapy could rattle the field once again. Trials presented at the ASRS24 showed a drastic reduction (from 85% to 95%) in the number of anti-VEGF and complement treatments needed following gene therapy injection, improving patient vision while relieving them from the stress of monthly injections.
But researchers are still debating the optimal corticosteroid regimen that is required for reducing the inflammatory response associated with the administration of gene therapies, especially those that use viral vectors. The main controversy is whether systemic immunosuppression is necessary or if local therapies, such as topical and intravitreal administration, can suffice.
Results presented at the meeting suggest that local therapies alone can be effective, potentially reducing the need for systemic immunosuppression.
The LUNA trial evaluated the efficacy and tolerability of ixoberogene soroparvovec, a therapy that delivers an anti-VEGF gene into the eye. Investigators included various prophylactic regimens, including local corticosteroids with and without oral prednisone. They found that local corticosteroid therapy alone effectively reduced inflammation.
Biopharma company 4DMT conducted the PRISM study, which examined a dual transgene therapy for neovascular AMD. Patients in this trial received a 20-week topical steroid taper. Only one patient (of 39) required a 6-week extension of steroid therapy. No patients experienced clinically significant intraocular inflammation, indicating that local corticosteroid therapy was effective in managing immune responses.
Currently, gene therapy clinical trials are designed for patients who have failed standard therapy or require frequent injections. “Once we figure out possible long-term side effects and how to deal with inflammation, [gene therapy] could reach many more patients,” Dr. Olmos de Koo said.
New Approaches Enter Pipeline
While gene therapy brings excitement to the field, it might not be for everyone, experts agreed at the ASRS24. New agents are being evaluated to offer a broader range of treatment options with longer-lasting efficacy. Results from early-phase trials presented at the meeting show favorable safety and efficacy signals.
“Finally, after a long time, we have a lot of exciting drugs for geographic atrophy in the pipeline that seem to be safe, with many studies also showing a functional outcome in addition to anatomical outcome,” Dr. Skondra told this news organization.
Current FDA-approved treatments for GA focus on inhibiting the humoral arm of the immune system through C3 and C5 inhibitors. However, a new approach targets both the humoral and cellular arms of the immune response by inhibiting macrophages that release pro-inflammatory cytokines. The goal is to convert these macrophages to a “resolution state,” potentially reducing the release of inflammatory cytokines and offering a more comprehensive treatment for wet and dry AMD, said Rishi Singh, MD, a retina surgeon at the Cole Eye Institute, Cleveland Clinic.
AVD-104, a sialic acid–coated nanoparticle developed by Aviceda Therapeutics, is a promising candidate in this approach. This 100-nm-in-diameter particle, which is only as heavy as 20 hydrogen atoms, is designed for better tissue penetration and has a pharmacokinetic profile lasting 3-4 months after a single intravitreal dose.
AVD-104 aims to repolarize macrophages into a resolution phenotype and decreases complement factor overamplification through direct binding to complement factor H, which downregulates C3 production in immune cells. This dual-action approach could offer a more effective and long-lasting treatment option.
Dr. Singh, who presented the phase 2/3 SIGLEC clinical trial assessing AVD-104, said a single dose resulted in significantly slower rates of disease progression as early as 1 month post-treatment and a notable decrease in junctional zone hyper-autofluorescence.
In addition, about 40% of patients gained vision, which was unexpected but a pleasant surprise, Dr. Singh said. “This is a small study. I don’t want anyone to walk away with the conclusion that we’ve figured out how to improve visual acuity in GA. But it’s promising.”
Other researchers are tackling GA by focusing on therapies that aim to intervene before the complement system is activated.
ONL1204 is a novel agent designed to inhibit the activation of the tumor necrosis factor FAS receptor, which is activated and upregulated in a disease state and is implicated in multiple cell death and inflammatory pathways.
Multiple preclinical models of AMD have shown that ONL1204 preserves retinal cells and inhibits inflammation by inhibiting the FAS receptor. Phase 1 trial results presented at the meeting showed that ONL1204 was safe and showed strong efficacy signals as early as 6 months after treatment initiation.
“We need to be cautiously optimistic,” Dr. Skondra said. “Larger studies will tell us if these signals are real. But it’s a very exciting time. I’m happy to see different mechanisms of action besides the complement because we can attack the disease from multiple fronts.”
Dr. Grewal declared interests with Eyepoint, Iveric Bio, Regeneron, Alumis, Apellis, DORC, and Genentech. Dr. Muth declared interests with Bayer, Canon, and Roche. Dr. Olmos de Koo declared interests with Alcon and Pixium Vision. Dr. Singh declared interests with Gyroscope, 4DMT, Aviceda, Eyepoint, Alcon, Bausch and Lomb, Novartis, and Regeneron. Dr. Skondra declared interests with Biogen, Iveric Bio, Allergan, and Trinity Health Science. Dr. Talcott declared interests with Bausch and Lomb, Eyepoint, Regeneron, REGENXBIO, Zeiss, Apellis, Genentech, Alimera, Outlook, and Iveric Bio.
A version of this article first appeared on Medscape.com.
FROM ASRS 2024
Too Much Coffee Linked to Accelerated Cognitive Decline
PHILADELPHIA – results from a large study suggest.
Investigators examined the impact of different amounts of coffee and tea on fluid intelligence — a measure of cognitive functions including abstract reasoning, pattern recognition, and logical thinking.
“It’s the old adage that too much of anything isn’t good. It’s all about balance, so moderate coffee consumption is okay but too much is probably not recommended,” said study investigator Kelsey R. Sewell, PhD, Advent Health Research Institute, Orlando, Florida.
The findings of the study were presented at the 2024 Alzheimer’s Association International Conference (AAIC).
One of the World’s Most Widely Consumed Beverages
Coffee is one of the most widely consumed beverages around the world. The beans contain a range of bioactive compounds, including caffeine, chlorogenic acid, and small amounts of vitamins and minerals.
Consistent evidence from observational and epidemiologic studies indicates that intake of both coffee and tea has beneficial effects on stroke, heart failure, cancers, diabetes, and Parkinson’s disease.
Several studies also suggest that coffee may reduce the risk for Alzheimer’s disease, said Dr. Sewell. However, there are limited longitudinal data on associations between coffee and tea intake and cognitive decline, particularly in distinct cognitive domains.
Dr. Sewell’s group previously published a study of cognitively unimpaired older adults that found greater coffee consumption was associated with slower cognitive decline and slower accumulation of brain beta-amyloid.
Their current study extends some of the prior findings and investigates the relationship between both coffee and tea intake and cognitive decline over time in a larger sample of older adults.
This new study included 8451 mostly female (60%) and White (97%) cognitively unimpaired adults older than 60 (mean age, 67.8 years) in the UK Biobank, a large-scale research resource containing in-depth, deidentified genetic and health information from half a million UK participants. Study subjects had a mean body mass index (BMI) of 26, and about 26% were apolipoprotein epsilon 4 (APOE e4) gene carriers.
Researchers divided coffee and tea consumption into tertiles: high, moderate, and no consumption.
For daily coffee consumption, 18% reported drinking four or more cups (high consumption), 58% reported drinking one to three cups (moderate consumption), and 25% reported that they never drink coffee. For daily tea consumption, 47% reported drinking four or more cups (high consumption), 38% reported drinking one to three cups (moderate consumption), and 15% reported that they never drink tea.
The study assessed cognitive function at baseline and at least two additional patient visits.
Researchers used linear mixed models to assess the relationships between coffee and tea intake and cognitive outcomes. The models adjusted for age, sex, Townsend deprivation index (reflecting socioeconomic status), ethnicity, APOE e4 status, and BMI.
Steeper Decline
Compared with high coffee consumption (four or more cups daily), people who never consumed coffee (beta, 0.06; standard error [SE], 0.02; P = .005) and those with moderate consumption (beta, 0.07; SE, 0.02; P = < .001) had slower decline in fluid intelligence after an average of 8.83 years of follow-up.
“We can see that those with high coffee consumption showed the steepest decline in fluid intelligence across the follow up, compared to those with moderate coffee consumption and those never consuming coffee,” said Dr. Sewell, referring to illustrative graphs.
At the same time, “our data suggest that across this time period, moderate coffee consumption can serve as some kind of protective factor against cognitive decline,” she added.
For tea, there was a somewhat different pattern. People who never drank tea had a greater decline in fluid intelligence, compared with those who had moderate consumption (beta, 0.06; SE, 0.02; P = .0090) or high consumption (beta, 0.06; SE, 0.02; P = .003).
Because this is an observational study, “we still need randomized controlled trials to better understand the neuroprotective mechanism of coffee and tea compounds,” said Dr. Sewell.
Responding later to a query from a meeting delegate about how moderate coffee drinking could be protective, Dr. Sewell said there are probably “different levels of mechanisms,” including at the molecular level (possibly involving amyloid toxicity) and the behavioral level (possibly involving sleep patterns).
Dr. Sewell said that she hopes this line of investigation will lead to new avenues of research in preventive strategies for Alzheimer’s disease.
“We hope that coffee and tea intake could contribute to the development of a safe and inexpensive strategy for delaying the onset and reducing the incidence for Alzheimer’s disease.”
A limitation of the study is possible recall bias, because coffee and tea consumption were self-reported. However, this may not be much of an issue because coffee and tea consumption “is usually quite a habitual behavior,” said Dr. Sewell.
The study also had no data on midlife coffee or tea consumption and did not compare the effect of different preparation methods or types of coffee and tea — for example, green tea versus black tea.
When asked if the study controlled for smoking, Dr. Sewell said it didn’t but added that it would be interesting to explore its impact on cognition.
Dr. Sewell reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – results from a large study suggest.
Investigators examined the impact of different amounts of coffee and tea on fluid intelligence — a measure of cognitive functions including abstract reasoning, pattern recognition, and logical thinking.
“It’s the old adage that too much of anything isn’t good. It’s all about balance, so moderate coffee consumption is okay but too much is probably not recommended,” said study investigator Kelsey R. Sewell, PhD, Advent Health Research Institute, Orlando, Florida.
The findings of the study were presented at the 2024 Alzheimer’s Association International Conference (AAIC).
One of the World’s Most Widely Consumed Beverages
Coffee is one of the most widely consumed beverages around the world. The beans contain a range of bioactive compounds, including caffeine, chlorogenic acid, and small amounts of vitamins and minerals.
Consistent evidence from observational and epidemiologic studies indicates that intake of both coffee and tea has beneficial effects on stroke, heart failure, cancers, diabetes, and Parkinson’s disease.
Several studies also suggest that coffee may reduce the risk for Alzheimer’s disease, said Dr. Sewell. However, there are limited longitudinal data on associations between coffee and tea intake and cognitive decline, particularly in distinct cognitive domains.
Dr. Sewell’s group previously published a study of cognitively unimpaired older adults that found greater coffee consumption was associated with slower cognitive decline and slower accumulation of brain beta-amyloid.
Their current study extends some of the prior findings and investigates the relationship between both coffee and tea intake and cognitive decline over time in a larger sample of older adults.
This new study included 8451 mostly female (60%) and White (97%) cognitively unimpaired adults older than 60 (mean age, 67.8 years) in the UK Biobank, a large-scale research resource containing in-depth, deidentified genetic and health information from half a million UK participants. Study subjects had a mean body mass index (BMI) of 26, and about 26% were apolipoprotein epsilon 4 (APOE e4) gene carriers.
Researchers divided coffee and tea consumption into tertiles: high, moderate, and no consumption.
For daily coffee consumption, 18% reported drinking four or more cups (high consumption), 58% reported drinking one to three cups (moderate consumption), and 25% reported that they never drink coffee. For daily tea consumption, 47% reported drinking four or more cups (high consumption), 38% reported drinking one to three cups (moderate consumption), and 15% reported that they never drink tea.
The study assessed cognitive function at baseline and at least two additional patient visits.
Researchers used linear mixed models to assess the relationships between coffee and tea intake and cognitive outcomes. The models adjusted for age, sex, Townsend deprivation index (reflecting socioeconomic status), ethnicity, APOE e4 status, and BMI.
Steeper Decline
Compared with high coffee consumption (four or more cups daily), people who never consumed coffee (beta, 0.06; standard error [SE], 0.02; P = .005) and those with moderate consumption (beta, 0.07; SE, 0.02; P = < .001) had slower decline in fluid intelligence after an average of 8.83 years of follow-up.
“We can see that those with high coffee consumption showed the steepest decline in fluid intelligence across the follow up, compared to those with moderate coffee consumption and those never consuming coffee,” said Dr. Sewell, referring to illustrative graphs.
At the same time, “our data suggest that across this time period, moderate coffee consumption can serve as some kind of protective factor against cognitive decline,” she added.
For tea, there was a somewhat different pattern. People who never drank tea had a greater decline in fluid intelligence, compared with those who had moderate consumption (beta, 0.06; SE, 0.02; P = .0090) or high consumption (beta, 0.06; SE, 0.02; P = .003).
Because this is an observational study, “we still need randomized controlled trials to better understand the neuroprotective mechanism of coffee and tea compounds,” said Dr. Sewell.
Responding later to a query from a meeting delegate about how moderate coffee drinking could be protective, Dr. Sewell said there are probably “different levels of mechanisms,” including at the molecular level (possibly involving amyloid toxicity) and the behavioral level (possibly involving sleep patterns).
Dr. Sewell said that she hopes this line of investigation will lead to new avenues of research in preventive strategies for Alzheimer’s disease.
“We hope that coffee and tea intake could contribute to the development of a safe and inexpensive strategy for delaying the onset and reducing the incidence for Alzheimer’s disease.”
A limitation of the study is possible recall bias, because coffee and tea consumption were self-reported. However, this may not be much of an issue because coffee and tea consumption “is usually quite a habitual behavior,” said Dr. Sewell.
The study also had no data on midlife coffee or tea consumption and did not compare the effect of different preparation methods or types of coffee and tea — for example, green tea versus black tea.
When asked if the study controlled for smoking, Dr. Sewell said it didn’t but added that it would be interesting to explore its impact on cognition.
Dr. Sewell reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA – results from a large study suggest.
Investigators examined the impact of different amounts of coffee and tea on fluid intelligence — a measure of cognitive functions including abstract reasoning, pattern recognition, and logical thinking.
“It’s the old adage that too much of anything isn’t good. It’s all about balance, so moderate coffee consumption is okay but too much is probably not recommended,” said study investigator Kelsey R. Sewell, PhD, Advent Health Research Institute, Orlando, Florida.
The findings of the study were presented at the 2024 Alzheimer’s Association International Conference (AAIC).
One of the World’s Most Widely Consumed Beverages
Coffee is one of the most widely consumed beverages around the world. The beans contain a range of bioactive compounds, including caffeine, chlorogenic acid, and small amounts of vitamins and minerals.
Consistent evidence from observational and epidemiologic studies indicates that intake of both coffee and tea has beneficial effects on stroke, heart failure, cancers, diabetes, and Parkinson’s disease.
Several studies also suggest that coffee may reduce the risk for Alzheimer’s disease, said Dr. Sewell. However, there are limited longitudinal data on associations between coffee and tea intake and cognitive decline, particularly in distinct cognitive domains.
Dr. Sewell’s group previously published a study of cognitively unimpaired older adults that found greater coffee consumption was associated with slower cognitive decline and slower accumulation of brain beta-amyloid.
Their current study extends some of the prior findings and investigates the relationship between both coffee and tea intake and cognitive decline over time in a larger sample of older adults.
This new study included 8451 mostly female (60%) and White (97%) cognitively unimpaired adults older than 60 (mean age, 67.8 years) in the UK Biobank, a large-scale research resource containing in-depth, deidentified genetic and health information from half a million UK participants. Study subjects had a mean body mass index (BMI) of 26, and about 26% were apolipoprotein epsilon 4 (APOE e4) gene carriers.
Researchers divided coffee and tea consumption into tertiles: high, moderate, and no consumption.
For daily coffee consumption, 18% reported drinking four or more cups (high consumption), 58% reported drinking one to three cups (moderate consumption), and 25% reported that they never drink coffee. For daily tea consumption, 47% reported drinking four or more cups (high consumption), 38% reported drinking one to three cups (moderate consumption), and 15% reported that they never drink tea.
The study assessed cognitive function at baseline and at least two additional patient visits.
Researchers used linear mixed models to assess the relationships between coffee and tea intake and cognitive outcomes. The models adjusted for age, sex, Townsend deprivation index (reflecting socioeconomic status), ethnicity, APOE e4 status, and BMI.
Steeper Decline
Compared with high coffee consumption (four or more cups daily), people who never consumed coffee (beta, 0.06; standard error [SE], 0.02; P = .005) and those with moderate consumption (beta, 0.07; SE, 0.02; P = < .001) had slower decline in fluid intelligence after an average of 8.83 years of follow-up.
“We can see that those with high coffee consumption showed the steepest decline in fluid intelligence across the follow up, compared to those with moderate coffee consumption and those never consuming coffee,” said Dr. Sewell, referring to illustrative graphs.
At the same time, “our data suggest that across this time period, moderate coffee consumption can serve as some kind of protective factor against cognitive decline,” she added.
For tea, there was a somewhat different pattern. People who never drank tea had a greater decline in fluid intelligence, compared with those who had moderate consumption (beta, 0.06; SE, 0.02; P = .0090) or high consumption (beta, 0.06; SE, 0.02; P = .003).
Because this is an observational study, “we still need randomized controlled trials to better understand the neuroprotective mechanism of coffee and tea compounds,” said Dr. Sewell.
Responding later to a query from a meeting delegate about how moderate coffee drinking could be protective, Dr. Sewell said there are probably “different levels of mechanisms,” including at the molecular level (possibly involving amyloid toxicity) and the behavioral level (possibly involving sleep patterns).
Dr. Sewell said that she hopes this line of investigation will lead to new avenues of research in preventive strategies for Alzheimer’s disease.
“We hope that coffee and tea intake could contribute to the development of a safe and inexpensive strategy for delaying the onset and reducing the incidence for Alzheimer’s disease.”
A limitation of the study is possible recall bias, because coffee and tea consumption were self-reported. However, this may not be much of an issue because coffee and tea consumption “is usually quite a habitual behavior,” said Dr. Sewell.
The study also had no data on midlife coffee or tea consumption and did not compare the effect of different preparation methods or types of coffee and tea — for example, green tea versus black tea.
When asked if the study controlled for smoking, Dr. Sewell said it didn’t but added that it would be interesting to explore its impact on cognition.
Dr. Sewell reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM AAIC 2024
Almost 50% of Global Dementia Cases May Be Preventable
PHILADELPHIA – a report from the Lancet Commission on dementia prevention, intervention, and care.
The report adds two new modifiable risk factors for dementia — high cholesterol and vision loss — to the 12 risk factors identified in the 2020 Lancet Commission report, which were linked to about 40% of all dementia cases.
The original Lancet Commission report, published in 2017, identified nine modifiable risk factors that were estimated to be responsible for one third of dementia cases.
“Our new report reveals that there is much more that can and should be done to reduce the risk of dementia. It’s never too early or too late to act, with opportunities to make an impact at any stage of life,” lead author Gill Livingston, MD, from University College London in England, said in a statement.
The 57-page report was published online in The Lancet Neurology (to coincide with its presentation at the 2024 Alzheimer’s Association International Conference (AAIC).
‘Compelling’ New Evidence
The 12 risk factors cited in the 2020 report are lower levels of education, hearing loss, hypertension, smoking, obesity, depression, physical inactivity, diabetes, excessive alcohol consumption, traumatic brain injury (TBI), air pollution, and social isolation.
According to the authors of the current report, there is “new compelling evidence” that untreated vision loss and elevated low-density lipoprotein (LDL) cholesterol are also risk factors for dementia.
These two added risk factors are associated with 9% of all dementia cases — with an estimated 7% of cases caused by high LDL cholesterol from about age 40 years, and 2% of cases caused by untreated vision loss in later life, the authors said.
Out of all 14 risk factors, those tied to the greatest proportion of dementia in the global population are hearing impairment and high LDL cholesterol (7% each), along with less education in early life, and social isolation in later life (5% each), the report estimates.
The new report also outlines 13 recommendations aimed at individuals and governments to help guard against dementia. They include preventing and treating hearing loss, vision loss, and depression; being cognitively active throughout life; using head protection in contact sports; reducing vascular risk factors (high cholesterol, diabetes, obesity, hypertension); improving air quality; and providing supportive community environments to increase social contact.
Tara Spires-Jones, PhD, president of the British Neuroscience Association, emphasized that, while this research doesn’t directly link specific factors to dementia, it supports evidence that a healthy lifestyle — encompassing education, social activities, exercise, cognitive engagement, and avoiding head injuries and harmful factors for heart and lung health — can enhance brain resilience and prevent dementia.
In an interview, Heather M. Snyder, PhD, senior vice president of medical and scientific relations, Alzheimer’s Association, said: “Our brains are complex and what happens throughout our lives may increase or decrease our risk for dementia as we age. Protecting brain health as we age requires a comprehensive approach that includes discussions on diet, exercise, heart health, hearing, and vision.”
Also weighing in on the new report, Shaheen Lakhan, MD, PhD, neurologist and researcher based in Miami, Florida, said the addition of high cholesterol is “particularly noteworthy as it reinforces the intricate connection between vascular health and brain health — a link we’ve long suspected but can now target more effectively.”
As for vision loss, “it’s not just a matter of seeing clearly; it’s a matter of thinking clearly. Untreated vision loss can lead to social isolation, reduced physical activity, and cognitive decline,” said Dr. Lakhan.
Dementia Is Not Inevitable
In his view, “the potential to prevent or delay nearly half of dementia cases by addressing these risk factors is nothing short of revolutionary. It shifts our perspective from viewing dementia as an inevitable part of aging to seeing it as a condition we can actively work to prevent,” Dr. Lakhan added.
He said the report’s emphasis on health equity is also important.
“Dementia risk factors disproportionately affect socioeconomically disadvantaged groups and low- and middle-income countries. Addressing these disparities isn’t just a matter of fairness in the fight against dementia, equality in prevention is as important as equality in treatment,” Dr. Lakhan commented.
While the report offers hope, it also presents a challenge, he said.
Implementing the recommended preventive measures requires a “coordinated effort from individuals, healthcare systems, and policymakers. The potential benefits, both in terms of quality of life and economic savings, make this effort not just worthwhile but imperative. Preventing dementia is not just a medical imperative — it’s an economic and humanitarian one,” Dr. Lakhan said.
Masud Husain, PhD, with the University of Oxford in England, agreed.
The conclusions in this report are “very important for all of us, but particularly for health policy makers and government,” he said.
“If we did simple things well such as screening for some of the factors identified in this report, with adequate resources to perform this, we have the potential to prevent dementia on a national scale. This would be far more cost effective than developing high-tech treatments, which so far have been disappointing in their impacts on people with established dementia,” Dr. Husain said.
The Lancet Commission was funded by University College London, Alzheimer’s Society, Alzheimer’s Research UK, and the Economic and Social Research Council. A complete list of author disclosures is available with the original article. Dr. Snyder, Dr. Lakhan, Dr. Husain and Dr. Spires-Jones have no relevant disclosures.
A version of this article appeared on Medscape.com.
PHILADELPHIA – a report from the Lancet Commission on dementia prevention, intervention, and care.
The report adds two new modifiable risk factors for dementia — high cholesterol and vision loss — to the 12 risk factors identified in the 2020 Lancet Commission report, which were linked to about 40% of all dementia cases.
The original Lancet Commission report, published in 2017, identified nine modifiable risk factors that were estimated to be responsible for one third of dementia cases.
“Our new report reveals that there is much more that can and should be done to reduce the risk of dementia. It’s never too early or too late to act, with opportunities to make an impact at any stage of life,” lead author Gill Livingston, MD, from University College London in England, said in a statement.
The 57-page report was published online in The Lancet Neurology (to coincide with its presentation at the 2024 Alzheimer’s Association International Conference (AAIC).
‘Compelling’ New Evidence
The 12 risk factors cited in the 2020 report are lower levels of education, hearing loss, hypertension, smoking, obesity, depression, physical inactivity, diabetes, excessive alcohol consumption, traumatic brain injury (TBI), air pollution, and social isolation.
According to the authors of the current report, there is “new compelling evidence” that untreated vision loss and elevated low-density lipoprotein (LDL) cholesterol are also risk factors for dementia.
These two added risk factors are associated with 9% of all dementia cases — with an estimated 7% of cases caused by high LDL cholesterol from about age 40 years, and 2% of cases caused by untreated vision loss in later life, the authors said.
Out of all 14 risk factors, those tied to the greatest proportion of dementia in the global population are hearing impairment and high LDL cholesterol (7% each), along with less education in early life, and social isolation in later life (5% each), the report estimates.
The new report also outlines 13 recommendations aimed at individuals and governments to help guard against dementia. They include preventing and treating hearing loss, vision loss, and depression; being cognitively active throughout life; using head protection in contact sports; reducing vascular risk factors (high cholesterol, diabetes, obesity, hypertension); improving air quality; and providing supportive community environments to increase social contact.
Tara Spires-Jones, PhD, president of the British Neuroscience Association, emphasized that, while this research doesn’t directly link specific factors to dementia, it supports evidence that a healthy lifestyle — encompassing education, social activities, exercise, cognitive engagement, and avoiding head injuries and harmful factors for heart and lung health — can enhance brain resilience and prevent dementia.
In an interview, Heather M. Snyder, PhD, senior vice president of medical and scientific relations, Alzheimer’s Association, said: “Our brains are complex and what happens throughout our lives may increase or decrease our risk for dementia as we age. Protecting brain health as we age requires a comprehensive approach that includes discussions on diet, exercise, heart health, hearing, and vision.”
Also weighing in on the new report, Shaheen Lakhan, MD, PhD, neurologist and researcher based in Miami, Florida, said the addition of high cholesterol is “particularly noteworthy as it reinforces the intricate connection between vascular health and brain health — a link we’ve long suspected but can now target more effectively.”
As for vision loss, “it’s not just a matter of seeing clearly; it’s a matter of thinking clearly. Untreated vision loss can lead to social isolation, reduced physical activity, and cognitive decline,” said Dr. Lakhan.
Dementia Is Not Inevitable
In his view, “the potential to prevent or delay nearly half of dementia cases by addressing these risk factors is nothing short of revolutionary. It shifts our perspective from viewing dementia as an inevitable part of aging to seeing it as a condition we can actively work to prevent,” Dr. Lakhan added.
He said the report’s emphasis on health equity is also important.
“Dementia risk factors disproportionately affect socioeconomically disadvantaged groups and low- and middle-income countries. Addressing these disparities isn’t just a matter of fairness in the fight against dementia, equality in prevention is as important as equality in treatment,” Dr. Lakhan commented.
While the report offers hope, it also presents a challenge, he said.
Implementing the recommended preventive measures requires a “coordinated effort from individuals, healthcare systems, and policymakers. The potential benefits, both in terms of quality of life and economic savings, make this effort not just worthwhile but imperative. Preventing dementia is not just a medical imperative — it’s an economic and humanitarian one,” Dr. Lakhan said.
Masud Husain, PhD, with the University of Oxford in England, agreed.
The conclusions in this report are “very important for all of us, but particularly for health policy makers and government,” he said.
“If we did simple things well such as screening for some of the factors identified in this report, with adequate resources to perform this, we have the potential to prevent dementia on a national scale. This would be far more cost effective than developing high-tech treatments, which so far have been disappointing in their impacts on people with established dementia,” Dr. Husain said.
The Lancet Commission was funded by University College London, Alzheimer’s Society, Alzheimer’s Research UK, and the Economic and Social Research Council. A complete list of author disclosures is available with the original article. Dr. Snyder, Dr. Lakhan, Dr. Husain and Dr. Spires-Jones have no relevant disclosures.
A version of this article appeared on Medscape.com.
PHILADELPHIA – a report from the Lancet Commission on dementia prevention, intervention, and care.
The report adds two new modifiable risk factors for dementia — high cholesterol and vision loss — to the 12 risk factors identified in the 2020 Lancet Commission report, which were linked to about 40% of all dementia cases.
The original Lancet Commission report, published in 2017, identified nine modifiable risk factors that were estimated to be responsible for one third of dementia cases.
“Our new report reveals that there is much more that can and should be done to reduce the risk of dementia. It’s never too early or too late to act, with opportunities to make an impact at any stage of life,” lead author Gill Livingston, MD, from University College London in England, said in a statement.
The 57-page report was published online in The Lancet Neurology (to coincide with its presentation at the 2024 Alzheimer’s Association International Conference (AAIC).
‘Compelling’ New Evidence
The 12 risk factors cited in the 2020 report are lower levels of education, hearing loss, hypertension, smoking, obesity, depression, physical inactivity, diabetes, excessive alcohol consumption, traumatic brain injury (TBI), air pollution, and social isolation.
According to the authors of the current report, there is “new compelling evidence” that untreated vision loss and elevated low-density lipoprotein (LDL) cholesterol are also risk factors for dementia.
These two added risk factors are associated with 9% of all dementia cases — with an estimated 7% of cases caused by high LDL cholesterol from about age 40 years, and 2% of cases caused by untreated vision loss in later life, the authors said.
Out of all 14 risk factors, those tied to the greatest proportion of dementia in the global population are hearing impairment and high LDL cholesterol (7% each), along with less education in early life, and social isolation in later life (5% each), the report estimates.
The new report also outlines 13 recommendations aimed at individuals and governments to help guard against dementia. They include preventing and treating hearing loss, vision loss, and depression; being cognitively active throughout life; using head protection in contact sports; reducing vascular risk factors (high cholesterol, diabetes, obesity, hypertension); improving air quality; and providing supportive community environments to increase social contact.
Tara Spires-Jones, PhD, president of the British Neuroscience Association, emphasized that, while this research doesn’t directly link specific factors to dementia, it supports evidence that a healthy lifestyle — encompassing education, social activities, exercise, cognitive engagement, and avoiding head injuries and harmful factors for heart and lung health — can enhance brain resilience and prevent dementia.
In an interview, Heather M. Snyder, PhD, senior vice president of medical and scientific relations, Alzheimer’s Association, said: “Our brains are complex and what happens throughout our lives may increase or decrease our risk for dementia as we age. Protecting brain health as we age requires a comprehensive approach that includes discussions on diet, exercise, heart health, hearing, and vision.”
Also weighing in on the new report, Shaheen Lakhan, MD, PhD, neurologist and researcher based in Miami, Florida, said the addition of high cholesterol is “particularly noteworthy as it reinforces the intricate connection between vascular health and brain health — a link we’ve long suspected but can now target more effectively.”
As for vision loss, “it’s not just a matter of seeing clearly; it’s a matter of thinking clearly. Untreated vision loss can lead to social isolation, reduced physical activity, and cognitive decline,” said Dr. Lakhan.
Dementia Is Not Inevitable
In his view, “the potential to prevent or delay nearly half of dementia cases by addressing these risk factors is nothing short of revolutionary. It shifts our perspective from viewing dementia as an inevitable part of aging to seeing it as a condition we can actively work to prevent,” Dr. Lakhan added.
He said the report’s emphasis on health equity is also important.
“Dementia risk factors disproportionately affect socioeconomically disadvantaged groups and low- and middle-income countries. Addressing these disparities isn’t just a matter of fairness in the fight against dementia, equality in prevention is as important as equality in treatment,” Dr. Lakhan commented.
While the report offers hope, it also presents a challenge, he said.
Implementing the recommended preventive measures requires a “coordinated effort from individuals, healthcare systems, and policymakers. The potential benefits, both in terms of quality of life and economic savings, make this effort not just worthwhile but imperative. Preventing dementia is not just a medical imperative — it’s an economic and humanitarian one,” Dr. Lakhan said.
Masud Husain, PhD, with the University of Oxford in England, agreed.
The conclusions in this report are “very important for all of us, but particularly for health policy makers and government,” he said.
“If we did simple things well such as screening for some of the factors identified in this report, with adequate resources to perform this, we have the potential to prevent dementia on a national scale. This would be far more cost effective than developing high-tech treatments, which so far have been disappointing in their impacts on people with established dementia,” Dr. Husain said.
The Lancet Commission was funded by University College London, Alzheimer’s Society, Alzheimer’s Research UK, and the Economic and Social Research Council. A complete list of author disclosures is available with the original article. Dr. Snyder, Dr. Lakhan, Dr. Husain and Dr. Spires-Jones have no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM AAIC 2024
Promising New Data Support GLP-1s for Dementia Prevention
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
PHILADELPHIA –
In the phase 2b ELAD clinical trial, adults with early-stage Alzheimer’s disease taking the GLP-1 receptor agonist liraglutide exhibited slower decline in memory and thinking and experienced less brain atrophy over 12 months, compared with placebo.
“The slower loss of brain volume suggests liraglutide protects the brain, much like statins protect the heart,” study chief Paul Edison, MD, PhD, with Imperial College London, London, England, said in a statement.
“While further research is needed, liraglutide may work through various mechanisms, such as reducing inflammation in the brain, lowering insulin resistance and the toxic effects of Alzheimer’s biomarkers amyloid beta and tau, and improving how the brain’s nerve cells communicate,” Dr. Edison said.
He presented the study results at the 2024 Alzheimer’s Association International Conference (AAIC).
Brain Benefits
Liraglutide has previously demonstrated promising neuroprotective effects in animal models of Alzheimer’s disease and epidemiologic studies.
In ELAD, 204 patients with mild to moderate Alzheimer’s disease were randomly allocated (1:1) to a daily subcutaneous injection of up to 1.8 mg of liraglutide or placebo for 12 months; 80 patients in the liraglutide group and 89 in the placebo group completed the study.
Brain MRI was performed at baseline and at 12 months, along with neuropsychometric evaluation and 18F-fludeoxyglucose PET.
The study’s primary endpoint — change in the cerebral glucose metabolic rate in the cortical regions of the brain (hippocampus, medial temporal lobe, and posterior cingulate) — was not met.
However, patients taking liraglutide experienced a significant slowing of cognitive decline, compared with placebo group (P = .01), which was a key secondary outcome, calculated as a composite score of 18 different tests of memory, comprehension, language, and spatial orientation.
Although the study was not powered to assess cognitive changes, adults taking liraglutide had an 18% slower decline in cognitive function over 12 months, compared with those on placebo, Dr. Edison reported.
In addition, patients treated with liraglutide had nearly 50% less volume loss in several areas of the brain involved in memory, language, and decision-making, including frontal, temporal, parietal, and total gray matter, as measured by MRI.
Liraglutide daily subcutaneous injections were safe and well tolerated in patients with Alzheimer’s disease, Dr. Edison reported. There were 25 serious side effects — 18 in the placebo group and 7 in the liraglutide group — and most were considered unlikely to be related to the study treatment. There were no deaths.
Promising, Preliminary
This study shows a positive effect of liraglutide on the brain in terms of “slowing down of brain atrophy and slowing down the rate of cognitive decline,” said Howard Fillit, MD, founding executive director of the Alzheimer’s Drug Discovery Foundation, who wasn’t involved in the study.
Heather Snyder, PhD, vice-president of medical and scientific relations at the Alzheimer’s Association, said it’s “interesting” to see slowing of brain volume loss and some cognitive benefit “especially as the study was not powered necessarily to see some of those changes. The fact that they did see these changes in this small study provides a window into what may happen, but we certainly need larger phase 3 studies.”
In a statement from the UK nonprofit Science Media Centre, Tara Spires-Jones, PhD, president of the British Neuroscience Association and group leader at the UK Dementia Research Institute, called the data “promising.”
“There are clear links from strong data in the field between vascular risk factors including diabetes and obesity being associated with increased risk of dementia. The GLP-1 drug should help reduce these risk factors as well as potentially directly protecting brain cells,” Dr. Spires-Jones said.
However, she said “more research in bigger trials is needed to confirm whether this type of treatment will be effective in people with Alzheimer’s disease.”
Stephen Evans, MSc, emeritus professor, London School of Hygiene and Tropical Medicine, noted that the repurposing of drugs is “an important avenue of research but there is a lot of uncertainty here.”
He cautioned that the “50% brain volume change may not translate to important cognitive effects, and reporting only on those who completed the full 52 weeks of treatment could bring bias into the results. It sounds like it is worth pursuing a larger trial, but these results cannot demonstrate that liraglutide can protect against dementia.”
The ongoing phase 3 EVOKE trial is investigating the effects of the GLP-1 receptor agonist semaglutide in early Alzheimer’s disease.
Funding for the study was provided by Alzheimer’s Society UK, Alzheimer’s Drug Discovery Foundation, Novo Nordisk, John and Lucille Van Geest Foundation, and the National Institute for Health and Care Research Biomedical Research Centre. Dr. Edison, Dr. Fillit, Dr. Snyder, Mr. Evans, and Dr. Spires-Jones had no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
FROM AAIC 2024



