User login
FDA places SB-generated CAR T-cell therapy on clinical hold
The US Food and Drug Administrated (FDA) placed a clinical hold on the phase 1 trial of the Sleeping Beauty (SB)-generated CAR T-cell therapy in relapsed or refractory leukemia and lymphoma patients.
The Sleeping Beauty platform was designed to very rapidly manufacture CD19-specific CAR T cells at the point of care.
All SB-CAR T-cell processing is planned to take place within 2 days at the healthcare facility, thus eliminating shipping cells from hospitals to production sites and back again.
The FDA is requesting more chemistry, manufacturing, and control (CMC) information before allowing the trial to proceed.
The Sleeping Beauty technology, a non-viral transposon/transposase system, has the potential to reduce the costs and complexity associated with recombinant viral vector-based immunotherapy, according to developers.
Ziopharm Oncology, Precigen, Inc, a wholly owned subsidiary of Intrexon Corporation, and the University of Texas MD Anderson Cancer Center, are developing the Sleeping Beauty CAR T cell therapy.
“We know what is needed to address the hold issues and are looking forward to responding to the agency in a timely manner,” said Laurence Cooper, MD, PhD, chief executive officer of Ziopharm, in a corporate release.
“We are undertaking cutting-edge science and are on the verge of a paradigm shift based on our approach to very rapidly manufacture CD19-specific T cells within 2 days using our non-viral approach to CAR-T therapy based on the Sleeping Beauty platform.”
The phase 1 trial in question is a third-generation trial in which the CAR T cells are designed to co-express CD19-specific CAR, membrane-bound interleukin 15, and a safety switch.
The findings from earlier generation phase 1 trials have been previously reported in The Journal of Clinical Investigation.
The US Food and Drug Administrated (FDA) placed a clinical hold on the phase 1 trial of the Sleeping Beauty (SB)-generated CAR T-cell therapy in relapsed or refractory leukemia and lymphoma patients.
The Sleeping Beauty platform was designed to very rapidly manufacture CD19-specific CAR T cells at the point of care.
All SB-CAR T-cell processing is planned to take place within 2 days at the healthcare facility, thus eliminating shipping cells from hospitals to production sites and back again.
The FDA is requesting more chemistry, manufacturing, and control (CMC) information before allowing the trial to proceed.
The Sleeping Beauty technology, a non-viral transposon/transposase system, has the potential to reduce the costs and complexity associated with recombinant viral vector-based immunotherapy, according to developers.
Ziopharm Oncology, Precigen, Inc, a wholly owned subsidiary of Intrexon Corporation, and the University of Texas MD Anderson Cancer Center, are developing the Sleeping Beauty CAR T cell therapy.
“We know what is needed to address the hold issues and are looking forward to responding to the agency in a timely manner,” said Laurence Cooper, MD, PhD, chief executive officer of Ziopharm, in a corporate release.
“We are undertaking cutting-edge science and are on the verge of a paradigm shift based on our approach to very rapidly manufacture CD19-specific T cells within 2 days using our non-viral approach to CAR-T therapy based on the Sleeping Beauty platform.”
The phase 1 trial in question is a third-generation trial in which the CAR T cells are designed to co-express CD19-specific CAR, membrane-bound interleukin 15, and a safety switch.
The findings from earlier generation phase 1 trials have been previously reported in The Journal of Clinical Investigation.
The US Food and Drug Administrated (FDA) placed a clinical hold on the phase 1 trial of the Sleeping Beauty (SB)-generated CAR T-cell therapy in relapsed or refractory leukemia and lymphoma patients.
The Sleeping Beauty platform was designed to very rapidly manufacture CD19-specific CAR T cells at the point of care.
All SB-CAR T-cell processing is planned to take place within 2 days at the healthcare facility, thus eliminating shipping cells from hospitals to production sites and back again.
The FDA is requesting more chemistry, manufacturing, and control (CMC) information before allowing the trial to proceed.
The Sleeping Beauty technology, a non-viral transposon/transposase system, has the potential to reduce the costs and complexity associated with recombinant viral vector-based immunotherapy, according to developers.
Ziopharm Oncology, Precigen, Inc, a wholly owned subsidiary of Intrexon Corporation, and the University of Texas MD Anderson Cancer Center, are developing the Sleeping Beauty CAR T cell therapy.
“We know what is needed to address the hold issues and are looking forward to responding to the agency in a timely manner,” said Laurence Cooper, MD, PhD, chief executive officer of Ziopharm, in a corporate release.
“We are undertaking cutting-edge science and are on the verge of a paradigm shift based on our approach to very rapidly manufacture CD19-specific T cells within 2 days using our non-viral approach to CAR-T therapy based on the Sleeping Beauty platform.”
The phase 1 trial in question is a third-generation trial in which the CAR T cells are designed to co-express CD19-specific CAR, membrane-bound interleukin 15, and a safety switch.
The findings from earlier generation phase 1 trials have been previously reported in The Journal of Clinical Investigation.
Peripheral blood MRD correlates with treatment benefit in CLL
CHICAGO—Minimal residual disease (MRD) kinetics confirms the high, durable MRD-negativity with venetoclax plus rituximab in relapsed/refractory chronic lymphocytic leukemia (CLL), according to a further examination of the phase 3 MURANO study.
Undetectable MRD-negativity is associated with extended progression-free survival (PFS) and overall survival in patients receiving chemoimmunotherapy for CLL.
“Attainment of MRD-negativity in relapsed/refractory CLL is also a desired trial endpoint due to the subjectivity of complete response definition regarding pathologic lymph node size,” said Peter Hillmen, MD, of St James’s University Hospital, Leeds, United Kingdom, at the 2018 ASCO Annual Meeting.
Dr Hillmen reported new data on MRD response in cytogenetic and molecular risk groups, MRD sustainability and kinetics, and MRD conversion in the MURANO trial (abstract 7508).
MURANO trial (NCT02005471)
In the trial, venetoclax-rituximab showed superior PFS and peripheral blood and bone marrow MRD-negativity as compared to bendamustine plus rituximab (BR) in relapsed/refractory CLL patients.
Patients were randomized to venetoclax-rituximab for 6 months, followed by single-agent venetoclax for up to 1.5 years, or BR for 6 months. Peripheral blood samples were serially collected and bone marrow was collected at the end of combination treatment or at best response.
MRD findings
The new results show higher concordance in MRD-negativity between bone marrow and peripheral blood in venetoclax-rituximab (45 of 50 patients, 90%) vs BR (3 of 10 patients, 30%) in paired samples.
Focusing on peripheral blood MRD, Dr Hillmen said the best MRD-negativity rates were higher with venetoclax-rituximab (84%) than BR (23%). These results were independent of high-risk factors—such as del 17p, IGVH unmutated, and mutated TP53—only for venetoclax-rituximab treated patients.
“The superior peripheral blood MRD response with venetoclax-rituximab was consistent across subgroups at the end of completion of treatment,” Dr Hillmen said. “Most patients who achieved peripheral blood MRD-negativity on venetoclax-rituximab remained MRD-negative and were progression-free.”
Among 121 of 194 (62%) patients on venetoclax-rituximab who achieved MRD-negativity at the end of combination therapy, 100 (83%) patients maintained MRD-negativity and were progression-free at a median follow-up of 13.8 months. Two patients developed progressive disease and 2 patients died (unrelated to CLL).
Two patients developed Richter’s disease (with one MRD-positive directly before therapy) and 15 (12%) patients converted to confirmed MRD-positive at a median MRD-positive follow-up of 5.6 months.
“High peripheral blood MRD-negativity at the end of combination treatment and concordance with bone marrow MRD with venetoclax-rituximab,” Dr Hillmen said, “confirms the value of peripheral blood MRD for evaluation of treatment benefit in relapsed/refractory CLL patients. The high rate of peripheral blood MRD-negativity at end of combination treatment with venetoclax-rituximab was attained regardless of risk features.”
Some conversion to MRD-positivity occurred only in a small proportion of patients. Most cases were of intermediate level and remained progression-free, he said.
“MRD kinetics indicate that peripheral blood MRD-negativity with venetoclax-rituximab occurs early and is maintained over time with current follow-up,” Dr Hillmen added. The MRD data now provide a framework for designing response adaptive therapy.
The US Food and Drug Administration recently approved venetoclax-rituximab for CLL or small lymphocytic lymphoma for patients with or without del 17p.
Venetoclax is being developed by Genentech and Abbvie.
CHICAGO—Minimal residual disease (MRD) kinetics confirms the high, durable MRD-negativity with venetoclax plus rituximab in relapsed/refractory chronic lymphocytic leukemia (CLL), according to a further examination of the phase 3 MURANO study.
Undetectable MRD-negativity is associated with extended progression-free survival (PFS) and overall survival in patients receiving chemoimmunotherapy for CLL.
“Attainment of MRD-negativity in relapsed/refractory CLL is also a desired trial endpoint due to the subjectivity of complete response definition regarding pathologic lymph node size,” said Peter Hillmen, MD, of St James’s University Hospital, Leeds, United Kingdom, at the 2018 ASCO Annual Meeting.
Dr Hillmen reported new data on MRD response in cytogenetic and molecular risk groups, MRD sustainability and kinetics, and MRD conversion in the MURANO trial (abstract 7508).
MURANO trial (NCT02005471)
In the trial, venetoclax-rituximab showed superior PFS and peripheral blood and bone marrow MRD-negativity as compared to bendamustine plus rituximab (BR) in relapsed/refractory CLL patients.
Patients were randomized to venetoclax-rituximab for 6 months, followed by single-agent venetoclax for up to 1.5 years, or BR for 6 months. Peripheral blood samples were serially collected and bone marrow was collected at the end of combination treatment or at best response.
MRD findings
The new results show higher concordance in MRD-negativity between bone marrow and peripheral blood in venetoclax-rituximab (45 of 50 patients, 90%) vs BR (3 of 10 patients, 30%) in paired samples.
Focusing on peripheral blood MRD, Dr Hillmen said the best MRD-negativity rates were higher with venetoclax-rituximab (84%) than BR (23%). These results were independent of high-risk factors—such as del 17p, IGVH unmutated, and mutated TP53—only for venetoclax-rituximab treated patients.
“The superior peripheral blood MRD response with venetoclax-rituximab was consistent across subgroups at the end of completion of treatment,” Dr Hillmen said. “Most patients who achieved peripheral blood MRD-negativity on venetoclax-rituximab remained MRD-negative and were progression-free.”
Among 121 of 194 (62%) patients on venetoclax-rituximab who achieved MRD-negativity at the end of combination therapy, 100 (83%) patients maintained MRD-negativity and were progression-free at a median follow-up of 13.8 months. Two patients developed progressive disease and 2 patients died (unrelated to CLL).
Two patients developed Richter’s disease (with one MRD-positive directly before therapy) and 15 (12%) patients converted to confirmed MRD-positive at a median MRD-positive follow-up of 5.6 months.
“High peripheral blood MRD-negativity at the end of combination treatment and concordance with bone marrow MRD with venetoclax-rituximab,” Dr Hillmen said, “confirms the value of peripheral blood MRD for evaluation of treatment benefit in relapsed/refractory CLL patients. The high rate of peripheral blood MRD-negativity at end of combination treatment with venetoclax-rituximab was attained regardless of risk features.”
Some conversion to MRD-positivity occurred only in a small proportion of patients. Most cases were of intermediate level and remained progression-free, he said.
“MRD kinetics indicate that peripheral blood MRD-negativity with venetoclax-rituximab occurs early and is maintained over time with current follow-up,” Dr Hillmen added. The MRD data now provide a framework for designing response adaptive therapy.
The US Food and Drug Administration recently approved venetoclax-rituximab for CLL or small lymphocytic lymphoma for patients with or without del 17p.
Venetoclax is being developed by Genentech and Abbvie.
CHICAGO—Minimal residual disease (MRD) kinetics confirms the high, durable MRD-negativity with venetoclax plus rituximab in relapsed/refractory chronic lymphocytic leukemia (CLL), according to a further examination of the phase 3 MURANO study.
Undetectable MRD-negativity is associated with extended progression-free survival (PFS) and overall survival in patients receiving chemoimmunotherapy for CLL.
“Attainment of MRD-negativity in relapsed/refractory CLL is also a desired trial endpoint due to the subjectivity of complete response definition regarding pathologic lymph node size,” said Peter Hillmen, MD, of St James’s University Hospital, Leeds, United Kingdom, at the 2018 ASCO Annual Meeting.
Dr Hillmen reported new data on MRD response in cytogenetic and molecular risk groups, MRD sustainability and kinetics, and MRD conversion in the MURANO trial (abstract 7508).
MURANO trial (NCT02005471)
In the trial, venetoclax-rituximab showed superior PFS and peripheral blood and bone marrow MRD-negativity as compared to bendamustine plus rituximab (BR) in relapsed/refractory CLL patients.
Patients were randomized to venetoclax-rituximab for 6 months, followed by single-agent venetoclax for up to 1.5 years, or BR for 6 months. Peripheral blood samples were serially collected and bone marrow was collected at the end of combination treatment or at best response.
MRD findings
The new results show higher concordance in MRD-negativity between bone marrow and peripheral blood in venetoclax-rituximab (45 of 50 patients, 90%) vs BR (3 of 10 patients, 30%) in paired samples.
Focusing on peripheral blood MRD, Dr Hillmen said the best MRD-negativity rates were higher with venetoclax-rituximab (84%) than BR (23%). These results were independent of high-risk factors—such as del 17p, IGVH unmutated, and mutated TP53—only for venetoclax-rituximab treated patients.
“The superior peripheral blood MRD response with venetoclax-rituximab was consistent across subgroups at the end of completion of treatment,” Dr Hillmen said. “Most patients who achieved peripheral blood MRD-negativity on venetoclax-rituximab remained MRD-negative and were progression-free.”
Among 121 of 194 (62%) patients on venetoclax-rituximab who achieved MRD-negativity at the end of combination therapy, 100 (83%) patients maintained MRD-negativity and were progression-free at a median follow-up of 13.8 months. Two patients developed progressive disease and 2 patients died (unrelated to CLL).
Two patients developed Richter’s disease (with one MRD-positive directly before therapy) and 15 (12%) patients converted to confirmed MRD-positive at a median MRD-positive follow-up of 5.6 months.
“High peripheral blood MRD-negativity at the end of combination treatment and concordance with bone marrow MRD with venetoclax-rituximab,” Dr Hillmen said, “confirms the value of peripheral blood MRD for evaluation of treatment benefit in relapsed/refractory CLL patients. The high rate of peripheral blood MRD-negativity at end of combination treatment with venetoclax-rituximab was attained regardless of risk features.”
Some conversion to MRD-positivity occurred only in a small proportion of patients. Most cases were of intermediate level and remained progression-free, he said.
“MRD kinetics indicate that peripheral blood MRD-negativity with venetoclax-rituximab occurs early and is maintained over time with current follow-up,” Dr Hillmen added. The MRD data now provide a framework for designing response adaptive therapy.
The US Food and Drug Administration recently approved venetoclax-rituximab for CLL or small lymphocytic lymphoma for patients with or without del 17p.
Venetoclax is being developed by Genentech and Abbvie.
DLBCL survival improved with novel antibody-drug conjugate
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
STOCKHOLM, SWEDEN – Adding an experimental antibody-drug conjugate to bendamustine and rituximab more than doubled overall survival over bendamustine/rituximab alone in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL), investigators reported.
Among 80 transplant-ineligible patients with relapsed or refractory DLBCL in a phase 2 trial, the combination of the antibody-drug conjugate (ADC) polatuzumab vedotin plus bendamustine/rituximab (BR) was associated with a 40% complete response rate, compared with 15% for BR alone.
More importantly, the ADC was associated with 6.7 months median progression-free survival (PFS) versus 2 months for BR, and 11.8 months median overall survival (OS), versus 4.7 months for BR alone, reported Laurie Sehn, MD, from BC Cancer in Vancouver, British Columbia, Canada.
“This randomized phase 2 trial really is, so far, the only head-to-head comparison of a novel targeted agent against a standard therapy in this patient population that’s ineligible for transplant, and it demonstrated that the combination polatuzumab vedotin with BR significantly improved the response rates and progression-free survival, as well as overall survival,” she said at the annual congress of the European Hematology Association.
However, in a separate cohort of patients with follicular lymphoma in the same trial, there was no difference in either PFS or OS during follow-up to date, Dr. Sehn reported.
Polatuzumab vedotin consists of an antibody targeted against CD79b, an antigenic protein expressed on the surface of normal B cells, as well as DLBCL and follicular lymphoma cells.
Dr. Sehn and her colleagues enrolled 80 patients with DLBCL for whom first-line chemoimmunotherapy had failed and who were ineligible for stem cell transplant due to age and/or comorbidities.
A second cohort included 80 patients with follicular lymphoma. In this group, median PFS with polatuzumab vedotin/BR was 17 months versus 17.3 months for BR alone, and median overall survival had not been reached in either arm at the time of the data cutoff.
In the DLBCL cohort, patients were randomized to receive polatuzumab vedotin 1.8 mg/kg plus bendamustine 90mg/m2 for 2 days and rituximab 375mg/m2) or BR alone for six 21-day cycles.
The complete response rate by PET scan – the primary endpoint – was significantly higher with polatuzmab/BR at 40% versus 15% for BR alone (P = .012). Respective overall response rates were 45% versus 18% (P = .008). Also, median PFS with the polatuzmab/BR therapy was 6.7 months versus 2.0 months for BR alone, translating into a hazard ratio of 0.31 (P less than .0001).
Respective median overall survival was 11.8 versus 4.7 months, translating into a hazard ratio for the polatuzmab/BR combination of 0.35 (P = .0008).
The PET complete response rates were higher with polatuzmab/BR regardless of prior lines of therapy or refractory status, Dr. Sehn noted.
“In terms of the safety, I think importantly in the combination there were no unexpected toxicities, so typical to what we would expect with what’s known with this drug alone,” Dr. Sehn said.
Grade 3 or greater toxicities that were higher with the polatuzmab/BR combination included cytopenias, febrile neutropenia, and infections. The single serious adverse event that had a higher incidence in the polatuzumab/BR arm was febrile neutropenia (DLBCL). In total, 12% of patients in the polatuzumab-containing arm and 11% of patients in the BR-only arm died on study. Many of the deaths were due to disease progression.
Anton Hagenbeek, MD, PhD, from the Academic Medical Center at the University of Amsterdam, the Netherlands, who moderated the briefing but was not involved in the study, said that about 20%-30% of patients with relapsed/refractory DLBCL are positive for the CD33 antigen, the target of brentuximab vedotin (Adcetris), and noted that this agent is also being tested in a phase 2 trial.
Martin Hutchings, MD, PhD, from Rigshospitalet in Copenhagen, the Netherlands, who co-moderated the oral abstract session, commented that “it’s not so often that we see significant overall survival differences in a phase 2 study with 80 patients.”
Based on the results of this trial, polatuzumab has been granted breakthrough therapy designation by the U.S. Food and Drug Administration and a PRIME (priority medicine) designation from the European Medicines Agency.
SOURCE: Sehn LH et al. EHA Congress, Abstract S802.
REPORTING FROM THE EHA CONGRESS
Key clinical point:
Major finding: The complete response rate with polatuzumab vedotin plus bendamustine/rituximab (BR) was 40%, compared with 15% for BR alone.
Study details: Randomized controlled phase 2 trial in 80 patients with relapsed/refractory DLBCL.
Disclosures: The study was funded by Hoffman-La Roche. Dr. Sehn reported ties to Roche/Genentech and others.
Source: Sehn LH et al. EHA Congress, Abstract S802.
‘Excellent’ survival with HCT despite early treatment failure in FL
Autologous and allogeneic hematopoietic stem cell transplantation (HCT) both offer excellent long-term survival in follicular lymphoma (FL) patients who experience early treatment failure, an analysis of a large transplant registry suggests.
Five-year survival rates exceeded 70% for patients who received autologous or matched sibling donor (MSD) transplants, according to the analysis of the Center for International Blood and Marrow Transplant Research (CIBMTR) database. The database included 440 patients who underwent a procedure between 2002 and 2014.
“Until better risk-stratification tools are available for FL, auto-HCT and MSD allo-HCT should be considered as effective treatment options with excellent long-term survival for high-risk patients as defined by early treatment failure,” Sonali M. Smith, MD, of the University of Chicago, and co-investigators wrote in the journal Cancer.
Early treatment failure in FL is associated with worse overall survival. In the National LymphoCare Study (NLCS), patients who received upfront R-CHOP therapy and progressed within 24 months had a 5-year overall survival of 50%, versus 90% for patients without early progression.
By contrast, survival figures in the present study are “provocatively higher” than those in the NLCS, in which only 8 out of 110 patients underwent HCT, Dr Smith and co-authors said.
Dr Smith’s study showed that with a median follow-up of 69 to 73 months, adjusted probability of 5-year overall survival was 70% for autologous and 73% for MSD HCT, versus 49% for matched unrelated donor HCT (P=0.0008).
Ryan C. Lynch, MD, and Ajay K. Gopal, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, said that the finding “convincingly demonstrates” the benefit of transplant in the setting of early treatment failure.
“Select patients (particularly younger patients) with chemoresponsive disease who understand the risk-benefit ratio in comparison with currently approved and experimental therapies still remain good candidates for autologous HCT,” Drs Lynch and Gopal said in an editorial.
“For older patients or patients with comorbidities, we would continue to recommend clinical trials or treatment with an approved PI3K inhibitor,” they added.
The study by Dr Smith and colleagues is not the first to show a benefit of HCT in this clinical scenario. In a recent NLCS/CIBMTR analysis of FL patients, 5-year overall survival was 73% for those undergoing autologous HCT done within a year of early treatment failure, versus 60% for those who did not (P=0.05).
The two studies “collectively suggest that transplantation should be considered in this high-risk group of patients with early relapse,” Dr Smith and co-authors wrote.
Autologous and allogeneic hematopoietic stem cell transplantation (HCT) both offer excellent long-term survival in follicular lymphoma (FL) patients who experience early treatment failure, an analysis of a large transplant registry suggests.
Five-year survival rates exceeded 70% for patients who received autologous or matched sibling donor (MSD) transplants, according to the analysis of the Center for International Blood and Marrow Transplant Research (CIBMTR) database. The database included 440 patients who underwent a procedure between 2002 and 2014.
“Until better risk-stratification tools are available for FL, auto-HCT and MSD allo-HCT should be considered as effective treatment options with excellent long-term survival for high-risk patients as defined by early treatment failure,” Sonali M. Smith, MD, of the University of Chicago, and co-investigators wrote in the journal Cancer.
Early treatment failure in FL is associated with worse overall survival. In the National LymphoCare Study (NLCS), patients who received upfront R-CHOP therapy and progressed within 24 months had a 5-year overall survival of 50%, versus 90% for patients without early progression.
By contrast, survival figures in the present study are “provocatively higher” than those in the NLCS, in which only 8 out of 110 patients underwent HCT, Dr Smith and co-authors said.
Dr Smith’s study showed that with a median follow-up of 69 to 73 months, adjusted probability of 5-year overall survival was 70% for autologous and 73% for MSD HCT, versus 49% for matched unrelated donor HCT (P=0.0008).
Ryan C. Lynch, MD, and Ajay K. Gopal, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, said that the finding “convincingly demonstrates” the benefit of transplant in the setting of early treatment failure.
“Select patients (particularly younger patients) with chemoresponsive disease who understand the risk-benefit ratio in comparison with currently approved and experimental therapies still remain good candidates for autologous HCT,” Drs Lynch and Gopal said in an editorial.
“For older patients or patients with comorbidities, we would continue to recommend clinical trials or treatment with an approved PI3K inhibitor,” they added.
The study by Dr Smith and colleagues is not the first to show a benefit of HCT in this clinical scenario. In a recent NLCS/CIBMTR analysis of FL patients, 5-year overall survival was 73% for those undergoing autologous HCT done within a year of early treatment failure, versus 60% for those who did not (P=0.05).
The two studies “collectively suggest that transplantation should be considered in this high-risk group of patients with early relapse,” Dr Smith and co-authors wrote.
Autologous and allogeneic hematopoietic stem cell transplantation (HCT) both offer excellent long-term survival in follicular lymphoma (FL) patients who experience early treatment failure, an analysis of a large transplant registry suggests.
Five-year survival rates exceeded 70% for patients who received autologous or matched sibling donor (MSD) transplants, according to the analysis of the Center for International Blood and Marrow Transplant Research (CIBMTR) database. The database included 440 patients who underwent a procedure between 2002 and 2014.
“Until better risk-stratification tools are available for FL, auto-HCT and MSD allo-HCT should be considered as effective treatment options with excellent long-term survival for high-risk patients as defined by early treatment failure,” Sonali M. Smith, MD, of the University of Chicago, and co-investigators wrote in the journal Cancer.
Early treatment failure in FL is associated with worse overall survival. In the National LymphoCare Study (NLCS), patients who received upfront R-CHOP therapy and progressed within 24 months had a 5-year overall survival of 50%, versus 90% for patients without early progression.
By contrast, survival figures in the present study are “provocatively higher” than those in the NLCS, in which only 8 out of 110 patients underwent HCT, Dr Smith and co-authors said.
Dr Smith’s study showed that with a median follow-up of 69 to 73 months, adjusted probability of 5-year overall survival was 70% for autologous and 73% for MSD HCT, versus 49% for matched unrelated donor HCT (P=0.0008).
Ryan C. Lynch, MD, and Ajay K. Gopal, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington, said that the finding “convincingly demonstrates” the benefit of transplant in the setting of early treatment failure.
“Select patients (particularly younger patients) with chemoresponsive disease who understand the risk-benefit ratio in comparison with currently approved and experimental therapies still remain good candidates for autologous HCT,” Drs Lynch and Gopal said in an editorial.
“For older patients or patients with comorbidities, we would continue to recommend clinical trials or treatment with an approved PI3K inhibitor,” they added.
The study by Dr Smith and colleagues is not the first to show a benefit of HCT in this clinical scenario. In a recent NLCS/CIBMTR analysis of FL patients, 5-year overall survival was 73% for those undergoing autologous HCT done within a year of early treatment failure, versus 60% for those who did not (P=0.05).
The two studies “collectively suggest that transplantation should be considered in this high-risk group of patients with early relapse,” Dr Smith and co-authors wrote.
Interim PET scans identify HL patients with better outcomes
CHICAGO—Interim PET scans can identify a subset of Hodgkin lymphoma (HL) patients with a better outcome suitable for de-escalation treatment after upfront BEACOPP without impairing disease control, according to final results of the AHL2011-LYSA study.
BEACOPP, compared to ABVD, improves progression-free survival (PFS) but not overall survival (OS) and is associated with a higher risk of myelodysplasia, acute leukemia, and infertility.
Investigators evaluated whether some patients might be able to reduce treatment intensity without compromising the effectiveness of their therapy.
Olivier Casasnovas, MD, of CHU Le Bocage Service d'Hématologie Clinique, Dijon, France, presented the final analysis at the 2018 ASCO Annual Meeting (abstract 7503).
AHL2011-LYSA study (NCT01358747)
The randomized phase 3 study compared an early PET-driven treatment de-escalation to a non-PET-monitored strategy in patients with advanced-stage HL.
The study included 823 previously untreated patients, median age 30 years (range 16 – 60), with stage III, IV, or high-risk IIB HL.
The PET-driven strategy consisted of 2 BEACOPP* cycles (PET2), followed by 4 cycles of ABVD** for PET2-negative patients, and 4 cycles of BEACOPP for PET2-positive patients.
The experimental PET-driven strategy (410 patients) was randomly compared to a standard treatment delivering 6 cycles of BEACOPP (413 patients). PFS was the primary endpoint with a hypothesis of non-inferiority of the PET-driven arm compared to the standard arm.
Patients characteristics were well balanced between the arms, Dr Casasnovas said. PET2-positivity rate was similar in both arms (experimental 13%, standard 12%).
Based on PET2 results, 346 (84%) patients received 4 cycles of ABVD and 51 (12%) patients received 4 additional cycles of BEACOPP in the experimental arm.
Results
With a median follow-up of 50 months, the 5-year PFS was similar in the standard (86.2%) and the PET-driven arms (85.7%). The 5-year PFS for PET 2-negative/PET 4-negative patients was 90.9%, for PET 2-positive/PET4-negative patients was 75.4%, and for PET 4-positive patients was 46.5%.
The 5-year OS was similar in both arms (96.4% experimental, 95.2% standard).
The treatment toxicity was significantly higher in patients receiving 6 cycles of BEACOPP as compared to those who received 2 cycles of BEACOPP plus 4 cycles of ABVD.
Those who received more cycles of BEACOPP had more frequent grade 3 or higher adverse events than those with fewer cycles, including anemia (11% vs 2%), leukopenia (85% vs 74%), thrombocytopenia (44% vs 15%), and sepsis (7% vs 3%), as well as in serious adverse events (45% vs 28%).
“After 4 cycles of chemotherapy, it [PET positivity] identifies a subset of patients with a particularly poor outcome,” Dr Casasnovas said, “encouraging researchers to develop new treatment options in these patients.”
“PET performed after 2 cycles of BEACOPP escalation can be safely used to guide subsequent treatment,” he concluded.
“This approach allows clinicians to reduce the treatment-related immediate toxicity in most patients,” he added, “and provides similar patient outcomes compared to standard BEACOPP escalation treatment.”
* Bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone
**Adriamycin (doxorubicin), bleomycin, vinblastine, dacarbazine
CHICAGO—Interim PET scans can identify a subset of Hodgkin lymphoma (HL) patients with a better outcome suitable for de-escalation treatment after upfront BEACOPP without impairing disease control, according to final results of the AHL2011-LYSA study.
BEACOPP, compared to ABVD, improves progression-free survival (PFS) but not overall survival (OS) and is associated with a higher risk of myelodysplasia, acute leukemia, and infertility.
Investigators evaluated whether some patients might be able to reduce treatment intensity without compromising the effectiveness of their therapy.
Olivier Casasnovas, MD, of CHU Le Bocage Service d'Hématologie Clinique, Dijon, France, presented the final analysis at the 2018 ASCO Annual Meeting (abstract 7503).
AHL2011-LYSA study (NCT01358747)
The randomized phase 3 study compared an early PET-driven treatment de-escalation to a non-PET-monitored strategy in patients with advanced-stage HL.
The study included 823 previously untreated patients, median age 30 years (range 16 – 60), with stage III, IV, or high-risk IIB HL.
The PET-driven strategy consisted of 2 BEACOPP* cycles (PET2), followed by 4 cycles of ABVD** for PET2-negative patients, and 4 cycles of BEACOPP for PET2-positive patients.
The experimental PET-driven strategy (410 patients) was randomly compared to a standard treatment delivering 6 cycles of BEACOPP (413 patients). PFS was the primary endpoint with a hypothesis of non-inferiority of the PET-driven arm compared to the standard arm.
Patients characteristics were well balanced between the arms, Dr Casasnovas said. PET2-positivity rate was similar in both arms (experimental 13%, standard 12%).
Based on PET2 results, 346 (84%) patients received 4 cycles of ABVD and 51 (12%) patients received 4 additional cycles of BEACOPP in the experimental arm.
Results
With a median follow-up of 50 months, the 5-year PFS was similar in the standard (86.2%) and the PET-driven arms (85.7%). The 5-year PFS for PET 2-negative/PET 4-negative patients was 90.9%, for PET 2-positive/PET4-negative patients was 75.4%, and for PET 4-positive patients was 46.5%.
The 5-year OS was similar in both arms (96.4% experimental, 95.2% standard).
The treatment toxicity was significantly higher in patients receiving 6 cycles of BEACOPP as compared to those who received 2 cycles of BEACOPP plus 4 cycles of ABVD.
Those who received more cycles of BEACOPP had more frequent grade 3 or higher adverse events than those with fewer cycles, including anemia (11% vs 2%), leukopenia (85% vs 74%), thrombocytopenia (44% vs 15%), and sepsis (7% vs 3%), as well as in serious adverse events (45% vs 28%).
“After 4 cycles of chemotherapy, it [PET positivity] identifies a subset of patients with a particularly poor outcome,” Dr Casasnovas said, “encouraging researchers to develop new treatment options in these patients.”
“PET performed after 2 cycles of BEACOPP escalation can be safely used to guide subsequent treatment,” he concluded.
“This approach allows clinicians to reduce the treatment-related immediate toxicity in most patients,” he added, “and provides similar patient outcomes compared to standard BEACOPP escalation treatment.”
* Bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone
**Adriamycin (doxorubicin), bleomycin, vinblastine, dacarbazine
CHICAGO—Interim PET scans can identify a subset of Hodgkin lymphoma (HL) patients with a better outcome suitable for de-escalation treatment after upfront BEACOPP without impairing disease control, according to final results of the AHL2011-LYSA study.
BEACOPP, compared to ABVD, improves progression-free survival (PFS) but not overall survival (OS) and is associated with a higher risk of myelodysplasia, acute leukemia, and infertility.
Investigators evaluated whether some patients might be able to reduce treatment intensity without compromising the effectiveness of their therapy.
Olivier Casasnovas, MD, of CHU Le Bocage Service d'Hématologie Clinique, Dijon, France, presented the final analysis at the 2018 ASCO Annual Meeting (abstract 7503).
AHL2011-LYSA study (NCT01358747)
The randomized phase 3 study compared an early PET-driven treatment de-escalation to a non-PET-monitored strategy in patients with advanced-stage HL.
The study included 823 previously untreated patients, median age 30 years (range 16 – 60), with stage III, IV, or high-risk IIB HL.
The PET-driven strategy consisted of 2 BEACOPP* cycles (PET2), followed by 4 cycles of ABVD** for PET2-negative patients, and 4 cycles of BEACOPP for PET2-positive patients.
The experimental PET-driven strategy (410 patients) was randomly compared to a standard treatment delivering 6 cycles of BEACOPP (413 patients). PFS was the primary endpoint with a hypothesis of non-inferiority of the PET-driven arm compared to the standard arm.
Patients characteristics were well balanced between the arms, Dr Casasnovas said. PET2-positivity rate was similar in both arms (experimental 13%, standard 12%).
Based on PET2 results, 346 (84%) patients received 4 cycles of ABVD and 51 (12%) patients received 4 additional cycles of BEACOPP in the experimental arm.
Results
With a median follow-up of 50 months, the 5-year PFS was similar in the standard (86.2%) and the PET-driven arms (85.7%). The 5-year PFS for PET 2-negative/PET 4-negative patients was 90.9%, for PET 2-positive/PET4-negative patients was 75.4%, and for PET 4-positive patients was 46.5%.
The 5-year OS was similar in both arms (96.4% experimental, 95.2% standard).
The treatment toxicity was significantly higher in patients receiving 6 cycles of BEACOPP as compared to those who received 2 cycles of BEACOPP plus 4 cycles of ABVD.
Those who received more cycles of BEACOPP had more frequent grade 3 or higher adverse events than those with fewer cycles, including anemia (11% vs 2%), leukopenia (85% vs 74%), thrombocytopenia (44% vs 15%), and sepsis (7% vs 3%), as well as in serious adverse events (45% vs 28%).
“After 4 cycles of chemotherapy, it [PET positivity] identifies a subset of patients with a particularly poor outcome,” Dr Casasnovas said, “encouraging researchers to develop new treatment options in these patients.”
“PET performed after 2 cycles of BEACOPP escalation can be safely used to guide subsequent treatment,” he concluded.
“This approach allows clinicians to reduce the treatment-related immediate toxicity in most patients,” he added, “and provides similar patient outcomes compared to standard BEACOPP escalation treatment.”
* Bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, prednisone
**Adriamycin (doxorubicin), bleomycin, vinblastine, dacarbazine
British good practice paper offers MCL diagnosis pearls
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
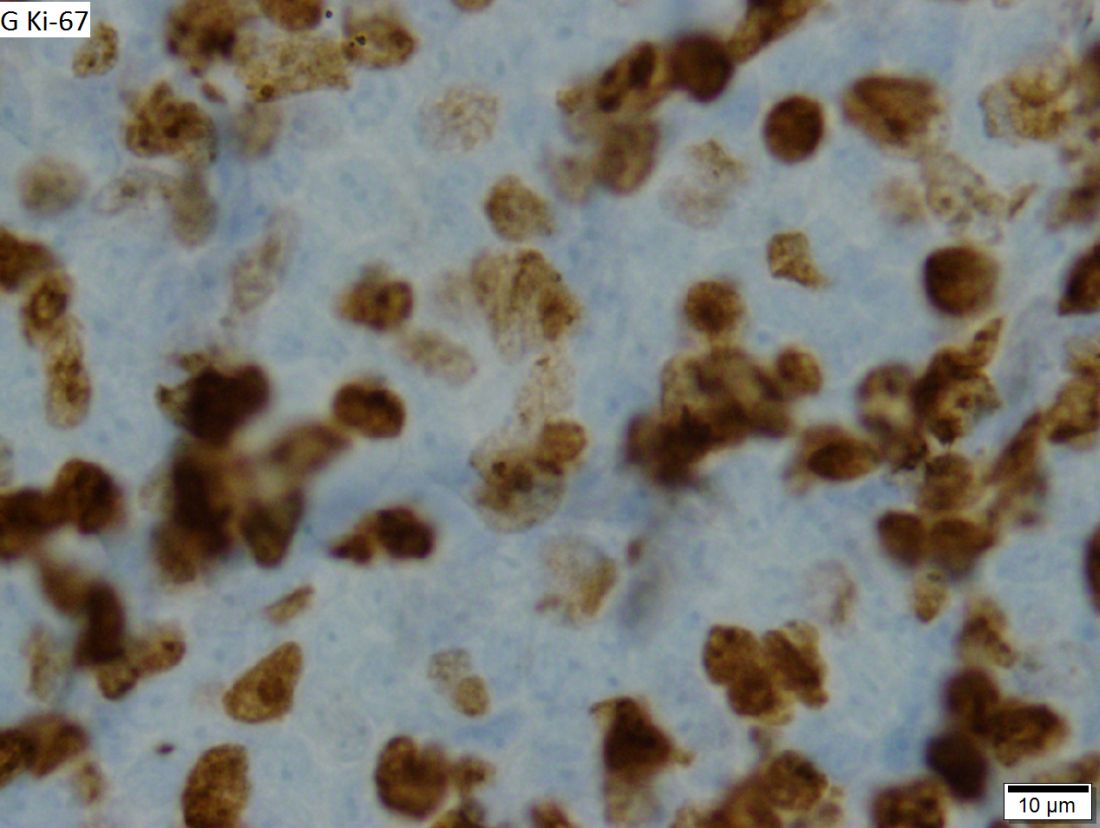
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
Immunohistochemical panels used in the diagnosis of mantle cell lymphoma should include cyclin D1 and SOX11 immunostaining, according to a good practice paper from the British Society of Haematology.
Pamela McKay, MD, of the Beatson West of Scotland Cancer Centre, Glasgow, and her colleagues provided based on a review of literature from 1980 to 2017. The good practice paper aims to offer best practice advice based on consensus where the evidence is limited. Specifically, the paper incorporates new information on molecular pathology and the use of positron emission tomography/computed tomography (PET/CT) scanning in staging of disease.
The top recommendations related to MCL diagnosis include performing lymph node excision or adequate core biopsy for diagnosis of nodal MCL. For non-nodal presentation, a tissue biopsy or peripheral blood can be used. Additionally, immunohistochemical panels should include cyclin D1 and SOX11 immunostaining.
In cases of atypical morphology, aberrant immunophenotype, equivocal cyclin D1 positivity, or unusual clinical presentation, the authors recommended fluorescence in situ hybridization (FISH) to demonstrate the presence of the t(11;14) translocation. They also recommended recording the Ki67 Proliferation Index at baseline, with an index of greater than 30% being indicative of a poorer outcome.
In terms of staging disease, Dr. McKay and her associates recommended that patients undergo staging with CT of the neck, chest, abdomen, and pelvis. They recommended against routine use of fluorodeoxyglucose PET for MCL staging, but said it could be considered if radical radiotherapy is being proposed for early-stage disease.
For cases with suspicion of central nervous system involvement, lumbar puncture with cytospin and immunophenotyping is recommended.
They recommended that all MCL patients have either their simplified or combined MCL international prognostic index score recorded at baseline.
All the authors made a declaration of interest to the British Society of Haematology and task force chairs, which may be viewed on request.
SOURCE: McKay P et al. Br J Haematol. 2018 Jun 8. doi: 10.1111/bjh.15281.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
FDA approves pembrolizumab for relapsed/refractory PMBCL
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
The immune checkpoint inhibitor in adult and pediatric patients.
The Food and Drug Administration based the accelerated approval on results from 53 patients with relapsed or refractory primary mediastinal large B-cell lymphoma in the KEYNOTE-170 trial. In the phase 2 trial, patients received 200 mg of pembrolizumab intravenously for 3 weeks until unacceptable toxicity or documented disease progression occurred. This continued for up to 24 months in patients who did not display progression. The overall response rate to pembrolizumab was 45% (95% CI, 32-60), which included both complete (11%) and partial (34%) responses. The median duration of response was not met within the follow-up period (median, 9.7 months) and the median time to first objective response was 2.8 months.
The recommended dose for pembrolizumab in adults is 200 mg every 3 weeks. It is recommended that pediatric patients receive 2 mg/kg every 3 weeks, with a maximum dose of 200 mg.
The most common adverse reactions to pembrolizumab were musculoskeletal pain, upper respiratory tract infection, pyrexia, fatigue, cough, dyspnea, diarrhea, nausea, arrhythmia, and headache. In total, a quarter of patients with adverse reactions required systemic treatment with a corticosteroid and 26% of patients had serious adverse reactions.
Pembrolizumab was approved via the FDA’s accelerated approval process, which allows for earlier approval of drugs that treat serious medical conditions and fulfill an unmet medical need. The drug was approved based on tumor response rate and durability of response, the FDA noted.
FDA grants pembrolizumab accelerated approval for PMBCL
The US Food and Drug Administration (FDA) granted accelerated approval to the anti-PD-1 therapy pembrolizumab (Keytruda) for the treatment of adult and pediatric patients with refractory primary mediastinal large B-cell lymphoma (PMBCL).
The indication also includes patients who have relapsed after 2 or more prior lines of therapy.
Pembrolizumab had received priority review for PMBCL late last year and also has orphan drug designation and breakthrough therapy designation for this indication.
The FDA based its approval on data from the KEYNOTE-170 (NCT02576990 ) trial.
Investigators enrolled 53 patients onto the multicenter, open-label, single-arm trial. Patients received pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression.
Patients whose disease did not progress received the drug for up to 24 months.
Patient characteristics
Patients were a median age of 33 years (range, 20 – 61), 43% were male, 92% white, 43% had an ECOG performance status of 0, and 57% had an ECOG performance status of 1.
Almost half (49%) had relapsed disease, and 36% had primary refractory disease.
About a quarter (26%) had undergone prior autologous hematopoietic stem cell transplant, and 32% had prior radiation therapy.
All patients had received prior rituximab.
Results
At a median follow-up of 9.7 months, the overall response rate was 45% (24 responders), including 11% complete responses and 34% partial responses.
The median duration of response was not reached during the follow-up period and ranged from a median 1.1 to 19.2 months.
Median time to first objective response was 2.8 months (range, 2.1 – 8.5). Accordingly, investigators do not recommend pembrolizumab for PMBCL patients who require urgent cytoreductive therapy.
Safety
The most common adverse events occurring in 10% or more of patients were musculoskeletal pain (30%), upper respiratory tract infection (28%), pyrexia (28%), fatigue (23%), cough (26%), dyspnea (21%), diarrhea (13%), abdominal pain (13%), nausea (11%), arrhythmia (11%), and headache (11%).
Eight percent of patients discontinued treatment, and 15% interrupted treatment due to adverse reactions.
Adverse events requiring systemic corticosteroid therapy occurred in 25% of patients.
Serious adverse events occurred in 26% and included arrhythmia (4 %), cardiac tamponade (2%), myocardial infarction (2%), pericardial effusion (2%), and pericarditis (2%).
Six (11%) patients died within 30 days of start of treatment.
The recommended pembrolizumab dose for treatment of adults with PMBCL is 200 mg every 3 weeks. The recommended dose in pediatric patients is 2 mg/kg (up to a maximum of 200 mg) every 3 weeks.
Additional indications for pembrolizumab include melanoma, non-small cell lung cancer, head and neck squamous cell cancer, classical Hodgkin lymphoma, urothelial carcinoma, microsatellite instability-high cancer, gastric cancer, and cervical cancer.
The full prescribing information is available on the FDA website.
Pembrolizumab (Keytruda) is a product of Merck & Co, Inc.
The US Food and Drug Administration (FDA) granted accelerated approval to the anti-PD-1 therapy pembrolizumab (Keytruda) for the treatment of adult and pediatric patients with refractory primary mediastinal large B-cell lymphoma (PMBCL).
The indication also includes patients who have relapsed after 2 or more prior lines of therapy.
Pembrolizumab had received priority review for PMBCL late last year and also has orphan drug designation and breakthrough therapy designation for this indication.
The FDA based its approval on data from the KEYNOTE-170 (NCT02576990 ) trial.
Investigators enrolled 53 patients onto the multicenter, open-label, single-arm trial. Patients received pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression.
Patients whose disease did not progress received the drug for up to 24 months.
Patient characteristics
Patients were a median age of 33 years (range, 20 – 61), 43% were male, 92% white, 43% had an ECOG performance status of 0, and 57% had an ECOG performance status of 1.
Almost half (49%) had relapsed disease, and 36% had primary refractory disease.
About a quarter (26%) had undergone prior autologous hematopoietic stem cell transplant, and 32% had prior radiation therapy.
All patients had received prior rituximab.
Results
At a median follow-up of 9.7 months, the overall response rate was 45% (24 responders), including 11% complete responses and 34% partial responses.
The median duration of response was not reached during the follow-up period and ranged from a median 1.1 to 19.2 months.
Median time to first objective response was 2.8 months (range, 2.1 – 8.5). Accordingly, investigators do not recommend pembrolizumab for PMBCL patients who require urgent cytoreductive therapy.
Safety
The most common adverse events occurring in 10% or more of patients were musculoskeletal pain (30%), upper respiratory tract infection (28%), pyrexia (28%), fatigue (23%), cough (26%), dyspnea (21%), diarrhea (13%), abdominal pain (13%), nausea (11%), arrhythmia (11%), and headache (11%).
Eight percent of patients discontinued treatment, and 15% interrupted treatment due to adverse reactions.
Adverse events requiring systemic corticosteroid therapy occurred in 25% of patients.
Serious adverse events occurred in 26% and included arrhythmia (4 %), cardiac tamponade (2%), myocardial infarction (2%), pericardial effusion (2%), and pericarditis (2%).
Six (11%) patients died within 30 days of start of treatment.
The recommended pembrolizumab dose for treatment of adults with PMBCL is 200 mg every 3 weeks. The recommended dose in pediatric patients is 2 mg/kg (up to a maximum of 200 mg) every 3 weeks.
Additional indications for pembrolizumab include melanoma, non-small cell lung cancer, head and neck squamous cell cancer, classical Hodgkin lymphoma, urothelial carcinoma, microsatellite instability-high cancer, gastric cancer, and cervical cancer.
The full prescribing information is available on the FDA website.
Pembrolizumab (Keytruda) is a product of Merck & Co, Inc.
The US Food and Drug Administration (FDA) granted accelerated approval to the anti-PD-1 therapy pembrolizumab (Keytruda) for the treatment of adult and pediatric patients with refractory primary mediastinal large B-cell lymphoma (PMBCL).
The indication also includes patients who have relapsed after 2 or more prior lines of therapy.
Pembrolizumab had received priority review for PMBCL late last year and also has orphan drug designation and breakthrough therapy designation for this indication.
The FDA based its approval on data from the KEYNOTE-170 (NCT02576990 ) trial.
Investigators enrolled 53 patients onto the multicenter, open-label, single-arm trial. Patients received pembrolizumab 200 mg intravenously every 3 weeks until unacceptable toxicity or documented disease progression.
Patients whose disease did not progress received the drug for up to 24 months.
Patient characteristics
Patients were a median age of 33 years (range, 20 – 61), 43% were male, 92% white, 43% had an ECOG performance status of 0, and 57% had an ECOG performance status of 1.
Almost half (49%) had relapsed disease, and 36% had primary refractory disease.
About a quarter (26%) had undergone prior autologous hematopoietic stem cell transplant, and 32% had prior radiation therapy.
All patients had received prior rituximab.
Results
At a median follow-up of 9.7 months, the overall response rate was 45% (24 responders), including 11% complete responses and 34% partial responses.
The median duration of response was not reached during the follow-up period and ranged from a median 1.1 to 19.2 months.
Median time to first objective response was 2.8 months (range, 2.1 – 8.5). Accordingly, investigators do not recommend pembrolizumab for PMBCL patients who require urgent cytoreductive therapy.
Safety
The most common adverse events occurring in 10% or more of patients were musculoskeletal pain (30%), upper respiratory tract infection (28%), pyrexia (28%), fatigue (23%), cough (26%), dyspnea (21%), diarrhea (13%), abdominal pain (13%), nausea (11%), arrhythmia (11%), and headache (11%).
Eight percent of patients discontinued treatment, and 15% interrupted treatment due to adverse reactions.
Adverse events requiring systemic corticosteroid therapy occurred in 25% of patients.
Serious adverse events occurred in 26% and included arrhythmia (4 %), cardiac tamponade (2%), myocardial infarction (2%), pericardial effusion (2%), and pericarditis (2%).
Six (11%) patients died within 30 days of start of treatment.
The recommended pembrolizumab dose for treatment of adults with PMBCL is 200 mg every 3 weeks. The recommended dose in pediatric patients is 2 mg/kg (up to a maximum of 200 mg) every 3 weeks.
Additional indications for pembrolizumab include melanoma, non-small cell lung cancer, head and neck squamous cell cancer, classical Hodgkin lymphoma, urothelial carcinoma, microsatellite instability-high cancer, gastric cancer, and cervical cancer.
The full prescribing information is available on the FDA website.
Pembrolizumab (Keytruda) is a product of Merck & Co, Inc.
Ibrutinib and venetoclax combo promising in frontline CLL
CHICAGO—Ibrutinib combined with venetoclax is showing promising clinical activity in the frontline treatment of patients with chronic lymphocytic leukemia (CLL), according to investigators for the CAPTIVATE study.
In the first 30 patients, 77% of treatment-naïve patients had undetected minimal residual disease (MRD; <10-4 cells) in the blood and 86% showed a similar response in the bone marrow.
The overall response rate (ORR) was 100% in 11 evaluable patients. The investigators reported this initial data at the 2018 Annual Meeting of the American Society of Clinical Oncology (abstract 7502).
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” said William G. Wierda, MD, PhD, of the MD Anderson Cancer Center in Houston, Texas, who presented the findings at ASCO.
Ibrutinib, a Bruton-kinase inhibitor, has already been approved for the treatment of CLL and venetoclax, a Bcl-2 inhibitor, is currently used to treat relapsed del 17p CLL.
Venetoclax in combination with rituximab was recently approved by the US Food and Drug Administration to treat patients with CLL or small lymphocytic lymphoma whether or not patients have del 17p.
With complementary mechanisms of action and preclinical studies suggesting synergy with the combination, CAPTIVATE was designed to test the efficacy of the oral combination given for 12 cycles.
Study design
CAPTIVATE (NCT02910583) is an ongoing phase 2 study that enrolled 164 patients with treatment-naïve CLL. Patients first received 3 cycles of ibrutinib monotherapy at the standard dose. This was intended to debulk the disease and reduce risk for venetoclax-associated tumor lysis syndrome (TLS).
Venetoclax 400 mg was initiated at cycle 4. After 12 cycles of the combination, patients with confirmed MRD negativity were randomized to receive ibrutinib with a placebo or to continue with the combination therapy.
In this initial report, Dr Wierda highlighted safety data for all 164 enrolled patients and efficacy data for the first 30 patients who had 6 cycles of combination therapy (MRD assessment cohort).
Dr Wierda also reported bone marrow data for the first 14 patients, who received a total of 12 cycles of the combination and represent the safety run-in cohort.
Ibrutinib and venetoclax show promising activity
Median age of patients was 58 years; about 2/3 of patients had unmutated IGHV and 1/3 had a creatine clearance of <80 mL/min.
Of 164 patients, 95% remain on therapy, with discontinuations reported for adverse events; one patient had disease progression to Richter’s transformation.
For the MRD evaluation, all 30 patients had 6 months of combination therapy and continue on treatment.
As expected, lead-in with ibrutinib monotherapy debulked the disease.
Investigators observed a reduction in the proportion of patients at high risk for TLS (24% to 3%) and an increase in the proportion of patients at low risk for TLS (12% to 29%).
A similar picture emerged for debulking of lymph node disease. No patient developed clinical TLS.
Other adverse events were consistent with the safety profile of single-agent ibrutinib and venetoclax. No new safety signals were seen.
After 6 cycles of the combination, blood MRD negativity was reported in 77% of the patients in the MRD assessment cohort.
In the safety-run in cohort of 14 patients, blood MRD negativity was reported in 86% of patients after 12 cycles and 93% of patients after 15 cycles of the combination. In these patients, bone marrow MRD negativity was achieved in 86%.
After 12 cycles of combination therapy, the objective response rate was 100% for 11 of the 14 evaluable patients from the safety run-in cohort: 6 patients showed complete remission (CR) or CR with incomplete blood count recovery (CRi) for a CR/CRi of 55%. All patients had confirmed undetectable MRD.
Investigators considered these responses promising and an assessment of the full treatment plan and durability of response are awaited.
The study was sponsored by Pharmacyclics.
CHICAGO—Ibrutinib combined with venetoclax is showing promising clinical activity in the frontline treatment of patients with chronic lymphocytic leukemia (CLL), according to investigators for the CAPTIVATE study.
In the first 30 patients, 77% of treatment-naïve patients had undetected minimal residual disease (MRD; <10-4 cells) in the blood and 86% showed a similar response in the bone marrow.
The overall response rate (ORR) was 100% in 11 evaluable patients. The investigators reported this initial data at the 2018 Annual Meeting of the American Society of Clinical Oncology (abstract 7502).
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” said William G. Wierda, MD, PhD, of the MD Anderson Cancer Center in Houston, Texas, who presented the findings at ASCO.
Ibrutinib, a Bruton-kinase inhibitor, has already been approved for the treatment of CLL and venetoclax, a Bcl-2 inhibitor, is currently used to treat relapsed del 17p CLL.
Venetoclax in combination with rituximab was recently approved by the US Food and Drug Administration to treat patients with CLL or small lymphocytic lymphoma whether or not patients have del 17p.
With complementary mechanisms of action and preclinical studies suggesting synergy with the combination, CAPTIVATE was designed to test the efficacy of the oral combination given for 12 cycles.
Study design
CAPTIVATE (NCT02910583) is an ongoing phase 2 study that enrolled 164 patients with treatment-naïve CLL. Patients first received 3 cycles of ibrutinib monotherapy at the standard dose. This was intended to debulk the disease and reduce risk for venetoclax-associated tumor lysis syndrome (TLS).
Venetoclax 400 mg was initiated at cycle 4. After 12 cycles of the combination, patients with confirmed MRD negativity were randomized to receive ibrutinib with a placebo or to continue with the combination therapy.
In this initial report, Dr Wierda highlighted safety data for all 164 enrolled patients and efficacy data for the first 30 patients who had 6 cycles of combination therapy (MRD assessment cohort).
Dr Wierda also reported bone marrow data for the first 14 patients, who received a total of 12 cycles of the combination and represent the safety run-in cohort.
Ibrutinib and venetoclax show promising activity
Median age of patients was 58 years; about 2/3 of patients had unmutated IGHV and 1/3 had a creatine clearance of <80 mL/min.
Of 164 patients, 95% remain on therapy, with discontinuations reported for adverse events; one patient had disease progression to Richter’s transformation.
For the MRD evaluation, all 30 patients had 6 months of combination therapy and continue on treatment.
As expected, lead-in with ibrutinib monotherapy debulked the disease.
Investigators observed a reduction in the proportion of patients at high risk for TLS (24% to 3%) and an increase in the proportion of patients at low risk for TLS (12% to 29%).
A similar picture emerged for debulking of lymph node disease. No patient developed clinical TLS.
Other adverse events were consistent with the safety profile of single-agent ibrutinib and venetoclax. No new safety signals were seen.
After 6 cycles of the combination, blood MRD negativity was reported in 77% of the patients in the MRD assessment cohort.
In the safety-run in cohort of 14 patients, blood MRD negativity was reported in 86% of patients after 12 cycles and 93% of patients after 15 cycles of the combination. In these patients, bone marrow MRD negativity was achieved in 86%.
After 12 cycles of combination therapy, the objective response rate was 100% for 11 of the 14 evaluable patients from the safety run-in cohort: 6 patients showed complete remission (CR) or CR with incomplete blood count recovery (CRi) for a CR/CRi of 55%. All patients had confirmed undetectable MRD.
Investigators considered these responses promising and an assessment of the full treatment plan and durability of response are awaited.
The study was sponsored by Pharmacyclics.
CHICAGO—Ibrutinib combined with venetoclax is showing promising clinical activity in the frontline treatment of patients with chronic lymphocytic leukemia (CLL), according to investigators for the CAPTIVATE study.
In the first 30 patients, 77% of treatment-naïve patients had undetected minimal residual disease (MRD; <10-4 cells) in the blood and 86% showed a similar response in the bone marrow.
The overall response rate (ORR) was 100% in 11 evaluable patients. The investigators reported this initial data at the 2018 Annual Meeting of the American Society of Clinical Oncology (abstract 7502).
“These early results show a highly active and safe treatment with 12 cycles of combined treatment with ibrutinib and venetoclax,” said William G. Wierda, MD, PhD, of the MD Anderson Cancer Center in Houston, Texas, who presented the findings at ASCO.
Ibrutinib, a Bruton-kinase inhibitor, has already been approved for the treatment of CLL and venetoclax, a Bcl-2 inhibitor, is currently used to treat relapsed del 17p CLL.
Venetoclax in combination with rituximab was recently approved by the US Food and Drug Administration to treat patients with CLL or small lymphocytic lymphoma whether or not patients have del 17p.
With complementary mechanisms of action and preclinical studies suggesting synergy with the combination, CAPTIVATE was designed to test the efficacy of the oral combination given for 12 cycles.
Study design
CAPTIVATE (NCT02910583) is an ongoing phase 2 study that enrolled 164 patients with treatment-naïve CLL. Patients first received 3 cycles of ibrutinib monotherapy at the standard dose. This was intended to debulk the disease and reduce risk for venetoclax-associated tumor lysis syndrome (TLS).
Venetoclax 400 mg was initiated at cycle 4. After 12 cycles of the combination, patients with confirmed MRD negativity were randomized to receive ibrutinib with a placebo or to continue with the combination therapy.
In this initial report, Dr Wierda highlighted safety data for all 164 enrolled patients and efficacy data for the first 30 patients who had 6 cycles of combination therapy (MRD assessment cohort).
Dr Wierda also reported bone marrow data for the first 14 patients, who received a total of 12 cycles of the combination and represent the safety run-in cohort.
Ibrutinib and venetoclax show promising activity
Median age of patients was 58 years; about 2/3 of patients had unmutated IGHV and 1/3 had a creatine clearance of <80 mL/min.
Of 164 patients, 95% remain on therapy, with discontinuations reported for adverse events; one patient had disease progression to Richter’s transformation.
For the MRD evaluation, all 30 patients had 6 months of combination therapy and continue on treatment.
As expected, lead-in with ibrutinib monotherapy debulked the disease.
Investigators observed a reduction in the proportion of patients at high risk for TLS (24% to 3%) and an increase in the proportion of patients at low risk for TLS (12% to 29%).
A similar picture emerged for debulking of lymph node disease. No patient developed clinical TLS.
Other adverse events were consistent with the safety profile of single-agent ibrutinib and venetoclax. No new safety signals were seen.
After 6 cycles of the combination, blood MRD negativity was reported in 77% of the patients in the MRD assessment cohort.
In the safety-run in cohort of 14 patients, blood MRD negativity was reported in 86% of patients after 12 cycles and 93% of patients after 15 cycles of the combination. In these patients, bone marrow MRD negativity was achieved in 86%.
After 12 cycles of combination therapy, the objective response rate was 100% for 11 of the 14 evaluable patients from the safety run-in cohort: 6 patients showed complete remission (CR) or CR with incomplete blood count recovery (CRi) for a CR/CRi of 55%. All patients had confirmed undetectable MRD.
Investigators considered these responses promising and an assessment of the full treatment plan and durability of response are awaited.
The study was sponsored by Pharmacyclics.
Chemo-free combo provides potential first-line option for FL
CHICAGO—A chemotherapy-free combination of lenalidomide plus rituximab shows similar efficacy and a different safety profile to chemotherapy plus rituximab (R-chemo) followed by rituximab maintenance in patients with previously untreated follicular lymphoma (FL).
According to investigators, the multicenter, international phase 3 RELEVANCE trial is the first to evaluate the chemo-free combination against the standard of care, R-chemo with rituximab maintenance.
“These results show that lenalidomide plus rituximab, a novel immunomodulatory approach, is a potential first-line option for patients with FL requiring treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fowler presented the results of the study at the 2018 ASCO Annual Meeting (abstract 7500).
The current standard of care in previously untreated symptomatic FL is immunochemotherapy induction followed by rituximab maintenance.
The immunomodulatory agent lenalidomide has complementary mechanisms with rituximab. Phase 2 studies of combined immunotherapy with lenalidomide and rituximab demonstrated 3-year progression-free survival (PFS) of 79%-81% in previously untreated FL, Dr Fowler said.
Phase 3 RELEVANCE trial (NCT01650701)
Investigators evaluated 1030 previously untreated grade 1-3a FL patients who required therapy.
Patients in the lenalidomide-rituximab group (n=513) received lenalidomide doses of 20 mg per day on days 2 to 22 and 28 for 6 to 12 cycles. Responders continued on therapy at 10 mg per day for a total of 18 cycles.
The rituximab dose was 375 mg/m2 weekly in cycle 1 and day 1 in cycles 2 to 6 and continued in responders for 12 additional cycles.
Patients in the R-chemo arm (n=517) received the investigator’s choice of standard rituximab-CHOP, rituximab-bendamustine, or rituximab-CVP, followed by 12 cycles of rituximab.
Most patients (72%) in the R-chemo arm received R-CHOP.
Baseline characteristics were similar in both groups, Dr Fowler said.
Co-primary endpoints were complete remission/complete remission unconfirmed (CR/Cru) at 120 weeks and PFS.
Results
At a median follow-up of 37.9 months, the superiority for lenalidomide and rituximab over rituximab-chemotherapy was not established.
For the lenalidomide-rituximab patients, the CR/Cru was 48% and 3-year PFS was 77% as compared to 53% and 78%, respectively, for rituximab-chemotherapy patients, as assessed by an independent review committee.
Overall survival was 94% in both groups.
Safety
“Important differences in safety profiles were observed between the arms,” Dr Fowler said.
Rituximab-chemotherapy patients had more frequent neutropenia, febrile neutropenia, growth factor usage, nausea, vomiting, neuropathy, and alopecia.
Lenalidomide and rituximab showed more cutaneous reactions, tumor flare, and diarrhea.
Toxicity profiles differed, with higher grade 4 neutropenia (31% vs 8%) and febrile neutropenia (7% vs 2%) with rituximab-chemotherapy compared with lenalidomide-rituximab, respectively.
More patients experienced grade 3/4 cutaneous events (7% vs 1%) with lenalidomide-rituximab.
Second primary malignancies were slightly higher with rituximab-chemotherapy (10%) than with lenalidomide-rituximab (7%). Grade 5 adverse events were 1% in both groups.
About 70% of patients completed treatment in both groups.
“Lenalidomide and rituximab was not superior to rituximab-chemotherapy based on mature CR/Cru at 120 weeks and interim PFS,” Dr Fowler said. “Both treatments showed similar efficacy results. Treatment effects on PFS were consistent across pre-specified subgroups.”
Dr Fowler presented data as of May 31, 2017. Continued follow-up on PFS and OS is ongoing.
The study is sponsored by Celgene Corporation and the Lymphoma Academic Research Organisation (LYSARC).
CHICAGO—A chemotherapy-free combination of lenalidomide plus rituximab shows similar efficacy and a different safety profile to chemotherapy plus rituximab (R-chemo) followed by rituximab maintenance in patients with previously untreated follicular lymphoma (FL).
According to investigators, the multicenter, international phase 3 RELEVANCE trial is the first to evaluate the chemo-free combination against the standard of care, R-chemo with rituximab maintenance.
“These results show that lenalidomide plus rituximab, a novel immunomodulatory approach, is a potential first-line option for patients with FL requiring treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fowler presented the results of the study at the 2018 ASCO Annual Meeting (abstract 7500).
The current standard of care in previously untreated symptomatic FL is immunochemotherapy induction followed by rituximab maintenance.
The immunomodulatory agent lenalidomide has complementary mechanisms with rituximab. Phase 2 studies of combined immunotherapy with lenalidomide and rituximab demonstrated 3-year progression-free survival (PFS) of 79%-81% in previously untreated FL, Dr Fowler said.
Phase 3 RELEVANCE trial (NCT01650701)
Investigators evaluated 1030 previously untreated grade 1-3a FL patients who required therapy.
Patients in the lenalidomide-rituximab group (n=513) received lenalidomide doses of 20 mg per day on days 2 to 22 and 28 for 6 to 12 cycles. Responders continued on therapy at 10 mg per day for a total of 18 cycles.
The rituximab dose was 375 mg/m2 weekly in cycle 1 and day 1 in cycles 2 to 6 and continued in responders for 12 additional cycles.
Patients in the R-chemo arm (n=517) received the investigator’s choice of standard rituximab-CHOP, rituximab-bendamustine, or rituximab-CVP, followed by 12 cycles of rituximab.
Most patients (72%) in the R-chemo arm received R-CHOP.
Baseline characteristics were similar in both groups, Dr Fowler said.
Co-primary endpoints were complete remission/complete remission unconfirmed (CR/Cru) at 120 weeks and PFS.
Results
At a median follow-up of 37.9 months, the superiority for lenalidomide and rituximab over rituximab-chemotherapy was not established.
For the lenalidomide-rituximab patients, the CR/Cru was 48% and 3-year PFS was 77% as compared to 53% and 78%, respectively, for rituximab-chemotherapy patients, as assessed by an independent review committee.
Overall survival was 94% in both groups.
Safety
“Important differences in safety profiles were observed between the arms,” Dr Fowler said.
Rituximab-chemotherapy patients had more frequent neutropenia, febrile neutropenia, growth factor usage, nausea, vomiting, neuropathy, and alopecia.
Lenalidomide and rituximab showed more cutaneous reactions, tumor flare, and diarrhea.
Toxicity profiles differed, with higher grade 4 neutropenia (31% vs 8%) and febrile neutropenia (7% vs 2%) with rituximab-chemotherapy compared with lenalidomide-rituximab, respectively.
More patients experienced grade 3/4 cutaneous events (7% vs 1%) with lenalidomide-rituximab.
Second primary malignancies were slightly higher with rituximab-chemotherapy (10%) than with lenalidomide-rituximab (7%). Grade 5 adverse events were 1% in both groups.
About 70% of patients completed treatment in both groups.
“Lenalidomide and rituximab was not superior to rituximab-chemotherapy based on mature CR/Cru at 120 weeks and interim PFS,” Dr Fowler said. “Both treatments showed similar efficacy results. Treatment effects on PFS were consistent across pre-specified subgroups.”
Dr Fowler presented data as of May 31, 2017. Continued follow-up on PFS and OS is ongoing.
The study is sponsored by Celgene Corporation and the Lymphoma Academic Research Organisation (LYSARC).
CHICAGO—A chemotherapy-free combination of lenalidomide plus rituximab shows similar efficacy and a different safety profile to chemotherapy plus rituximab (R-chemo) followed by rituximab maintenance in patients with previously untreated follicular lymphoma (FL).
According to investigators, the multicenter, international phase 3 RELEVANCE trial is the first to evaluate the chemo-free combination against the standard of care, R-chemo with rituximab maintenance.
“These results show that lenalidomide plus rituximab, a novel immunomodulatory approach, is a potential first-line option for patients with FL requiring treatment,” said investigator Nathan H. Fowler, MD, of the University of Texas MD Anderson Cancer Center in Houston.
Dr Fowler presented the results of the study at the 2018 ASCO Annual Meeting (abstract 7500).
The current standard of care in previously untreated symptomatic FL is immunochemotherapy induction followed by rituximab maintenance.
The immunomodulatory agent lenalidomide has complementary mechanisms with rituximab. Phase 2 studies of combined immunotherapy with lenalidomide and rituximab demonstrated 3-year progression-free survival (PFS) of 79%-81% in previously untreated FL, Dr Fowler said.
Phase 3 RELEVANCE trial (NCT01650701)
Investigators evaluated 1030 previously untreated grade 1-3a FL patients who required therapy.
Patients in the lenalidomide-rituximab group (n=513) received lenalidomide doses of 20 mg per day on days 2 to 22 and 28 for 6 to 12 cycles. Responders continued on therapy at 10 mg per day for a total of 18 cycles.
The rituximab dose was 375 mg/m2 weekly in cycle 1 and day 1 in cycles 2 to 6 and continued in responders for 12 additional cycles.
Patients in the R-chemo arm (n=517) received the investigator’s choice of standard rituximab-CHOP, rituximab-bendamustine, or rituximab-CVP, followed by 12 cycles of rituximab.
Most patients (72%) in the R-chemo arm received R-CHOP.
Baseline characteristics were similar in both groups, Dr Fowler said.
Co-primary endpoints were complete remission/complete remission unconfirmed (CR/Cru) at 120 weeks and PFS.
Results
At a median follow-up of 37.9 months, the superiority for lenalidomide and rituximab over rituximab-chemotherapy was not established.
For the lenalidomide-rituximab patients, the CR/Cru was 48% and 3-year PFS was 77% as compared to 53% and 78%, respectively, for rituximab-chemotherapy patients, as assessed by an independent review committee.
Overall survival was 94% in both groups.
Safety
“Important differences in safety profiles were observed between the arms,” Dr Fowler said.
Rituximab-chemotherapy patients had more frequent neutropenia, febrile neutropenia, growth factor usage, nausea, vomiting, neuropathy, and alopecia.
Lenalidomide and rituximab showed more cutaneous reactions, tumor flare, and diarrhea.
Toxicity profiles differed, with higher grade 4 neutropenia (31% vs 8%) and febrile neutropenia (7% vs 2%) with rituximab-chemotherapy compared with lenalidomide-rituximab, respectively.
More patients experienced grade 3/4 cutaneous events (7% vs 1%) with lenalidomide-rituximab.
Second primary malignancies were slightly higher with rituximab-chemotherapy (10%) than with lenalidomide-rituximab (7%). Grade 5 adverse events were 1% in both groups.
About 70% of patients completed treatment in both groups.
“Lenalidomide and rituximab was not superior to rituximab-chemotherapy based on mature CR/Cru at 120 weeks and interim PFS,” Dr Fowler said. “Both treatments showed similar efficacy results. Treatment effects on PFS were consistent across pre-specified subgroups.”
Dr Fowler presented data as of May 31, 2017. Continued follow-up on PFS and OS is ongoing.
The study is sponsored by Celgene Corporation and the Lymphoma Academic Research Organisation (LYSARC).