User login
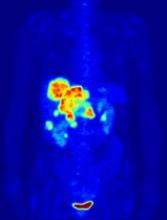
PET-guided treatment didn’t improve outcomes
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
In the PETAL trial, treatment intensification based on results of an interim positron emission tomography (PET) scan did not improve survival outcomes for patients with aggressive lymphomas.
PET-positive patients did not benefit by switching from R-CHOP to a more intensive chemotherapy regimen.
PET-negative patients did not benefit from 2 additional cycles of rituximab after R-CHOP.
These results were published in the Journal of Clinical Oncology.
PETAL was a randomized trial of patients with newly diagnosed T- or B-cell lymphomas.
Patients received 2 cycles of CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone)—plus rituximab (R-CHOP) in CD20-positive lymphomas—followed by a PET scan.
PET-positive patients were randomized to receive 6 additional cycles of R-CHOP or 6 blocks of an intensive protocol used to treat Burkitt lymphoma. This protocol consisted of high-dose methotrexate, cytarabine, hyperfractionated cyclophosphamide and ifosfamide, split-dose doxorubicin and etoposide, vincristine, vindesine, and dexamethasone.
PET-negative patients with CD20-positive lymphomas were randomized to receive 4 additional cycles of R-CHOP or 4 additional cycles of R-CHOP followed by 2 more doses of rituximab.
Among patients with T-cell lymphomas, only PET-positive individuals underwent randomization. PET-negative patients received CHOP. Patients with CD20-positive T-cell lymphomas also received rituximab.
PET-positive results
Of the PET-positive patients (108/862), 52 were randomized to receive 6 additional cycles of R-CHOP, and 56 were randomized to 6 cycles of the Burkitt protocol.
In general, survival rates were similar regardless of treatment. The 2-year overall survival (OS) rate was 63.6% for patients who received R-CHOP and 55.4% for those who received the more intensive protocol.
Two-year progression-free survival (PFS) rates were 49.4% and 43.1%, respectively. Two-year event-free survival (EFS) rates were 42.0% and 31.6%, respectively.
Among patients with diffuse large B-cell lymphoma (DLBCL), the OS rate was 64.8% for patients who received R-CHOP and 47.1% for those on the Burkitt protocol. PFS rates were 55.5% and 41.4%, respectively.
There was a significant difference in EFS rates among the DLBCL patients—52.4% in the R-CHOP arm and 28.3% in the intensive arm (P=0.0186).
Among T-cell lymphoma patients, the OS rate was 22.2% in the R-CHOP arm and 30.0% in the intensive arm. The PFS rates were 12.7% and 30%, respectively. The EFS rates were the same as the PFS rates.
Overall, patients who received the Burkitt protocol had significantly higher rates of grade 3/4 hematologic toxicities, infection, and mucositis.
PET-negative results
Of 754 PET-negative patients, 697 had CD20-positive lymphomas, and 255 of those patients (all with B-cell lymphomas) underwent randomization.
There were 129 patients who were randomized to receive 6 cycles of R-CHOP (2 before and 4 after randomization) and 126 who were randomized to receive 6 cycles of R-CHOP plus 2 additional cycles of rituximab.
Again, survival rates were similar regardless of treatment.
The 2-year OS was 88.2% for patients who received only R-CHOP and 87.2% for those with additional rituximab exposure. PFS rates were 82.0% and 77.5%, respectively. EFS rates were 76.4% and 73.5%, respectively.
In the DLBCL patients, the OS rate was 88.5% in the R-CHOP arm and 85.8% in the intensive arm. PFS rates were 82.3% and 77.7%, respectively. EFS rates were 72.6% and 78.9%, respectively.
As increasing the dose of rituximab did not improve outcomes, the investigators concluded that 6 cycles of R-CHOP should be the standard of care for these patients.
The team also said interim PET scanning is “a powerful tool” for identifying chemotherapy-resistant lymphomas, and PET-positive patients may be candidates for immunologic treatment approaches.
Mutations linked to higher risk of SNs in CCSs
New research has shown that childhood cancer survivors (CCSs) with certain germline mutations have higher relative rates (RRs) of secondary neoplasms (SNs) later in life.
Mutation carriers had significantly higher rates of breast cancer and sarcoma if they had received radiation to treat their initial cancer.
Among CCSs who did not receive radiation, the mutations were associated with increased rates of any SN, breast cancer, nonmelanoma skin cancer, and 2 or more histologically distinct SNs.
These findings were reported in the Journal of Clinical Oncology.
Researchers sequenced samples from 3006 CCSs who were at least 5 years from their initial cancer diagnosis as well as 341 samples from cancer-free control subjects.
All subjects were participants in the St. Jude Lifetime Cohort Study, a retrospective study with prospective clinical follow-up.
Thirty-five percent of the CCSs had survived leukemia, and 19% had survived lymphoma.
The CCS’s median age at childhood cancer diagnosis was 7.1 years, and the median follow-up was 28 years. The controls had a median age of 36.4 at follow-up.
Results
There were 1120 SNs diagnosed in 439 CCSs (14.6%). Ninety-one CCSs developed 2 or more histologically distinct SNs. The median time to SN diagnosis was 25.6 years
Non-melanoma skin cancer (580 in 159 CCSs), meningiomas (233 in 102 CCSs), thyroid cancer (67 in 67 CCSs), and breast cancer (60 in 53 CCSs) were among the SNs reported.
There were 15 neoplasms recorded in the control group—14 cases of non-melanoma skin cancer and 1 meningioma.
Pathogenic or likely pathogenic (P/LP) mutations in 32 genes were reported in 175 CCSs. The prevalence in CCSs (5.8%) was nearly 10-fold higher than in controls (0.6%).
The most commonly mutated genes in CCSs were RB1 (n=43), NF1 (n=22), BRCA2 (n=14), BRCA1 (n=12), and TP53 (n=10).
In a multivariable analysis adjusted for sex, age at primary cancer diagnosis, and treatment, P/LP mutation carriers had a significantly higher rate of any SN (RR=1.8).
The rate of subsequent breast cancer was significantly increased among females with a P/LP mutation (RR= 9.4), recipients of chest radiation (RR=7.9), and those with higher anthracycline exposure (RR=2.4).
The rate of subsequent sarcoma was significantly increased for mutation carriers (RR=10.9) and CCSs with greater exposure to alkylating agents (RR=3.8).
Among irradiated CCSs, P/LP mutations were associated with significantly increased rates of breast cancer (RR=13.9) and sarcoma (RR=10.6)
Among non-irradiated CCSs, P/LP mutations were associated with significantly increased rates of any SN (RR=4.7), breast cancer (RR=7.7), nonmelanoma skin cancer (RR=11.0), and 2 or more histologically distinct SNs (RR=18.6).
The researchers said the higher risk of SNs in CCSs with P/LP mutations suggests all CCSs should be referred for genetic counseling.
New research has shown that childhood cancer survivors (CCSs) with certain germline mutations have higher relative rates (RRs) of secondary neoplasms (SNs) later in life.
Mutation carriers had significantly higher rates of breast cancer and sarcoma if they had received radiation to treat their initial cancer.
Among CCSs who did not receive radiation, the mutations were associated with increased rates of any SN, breast cancer, nonmelanoma skin cancer, and 2 or more histologically distinct SNs.
These findings were reported in the Journal of Clinical Oncology.
Researchers sequenced samples from 3006 CCSs who were at least 5 years from their initial cancer diagnosis as well as 341 samples from cancer-free control subjects.
All subjects were participants in the St. Jude Lifetime Cohort Study, a retrospective study with prospective clinical follow-up.
Thirty-five percent of the CCSs had survived leukemia, and 19% had survived lymphoma.
The CCS’s median age at childhood cancer diagnosis was 7.1 years, and the median follow-up was 28 years. The controls had a median age of 36.4 at follow-up.
Results
There were 1120 SNs diagnosed in 439 CCSs (14.6%). Ninety-one CCSs developed 2 or more histologically distinct SNs. The median time to SN diagnosis was 25.6 years
Non-melanoma skin cancer (580 in 159 CCSs), meningiomas (233 in 102 CCSs), thyroid cancer (67 in 67 CCSs), and breast cancer (60 in 53 CCSs) were among the SNs reported.
There were 15 neoplasms recorded in the control group—14 cases of non-melanoma skin cancer and 1 meningioma.
Pathogenic or likely pathogenic (P/LP) mutations in 32 genes were reported in 175 CCSs. The prevalence in CCSs (5.8%) was nearly 10-fold higher than in controls (0.6%).
The most commonly mutated genes in CCSs were RB1 (n=43), NF1 (n=22), BRCA2 (n=14), BRCA1 (n=12), and TP53 (n=10).
In a multivariable analysis adjusted for sex, age at primary cancer diagnosis, and treatment, P/LP mutation carriers had a significantly higher rate of any SN (RR=1.8).
The rate of subsequent breast cancer was significantly increased among females with a P/LP mutation (RR= 9.4), recipients of chest radiation (RR=7.9), and those with higher anthracycline exposure (RR=2.4).
The rate of subsequent sarcoma was significantly increased for mutation carriers (RR=10.9) and CCSs with greater exposure to alkylating agents (RR=3.8).
Among irradiated CCSs, P/LP mutations were associated with significantly increased rates of breast cancer (RR=13.9) and sarcoma (RR=10.6)
Among non-irradiated CCSs, P/LP mutations were associated with significantly increased rates of any SN (RR=4.7), breast cancer (RR=7.7), nonmelanoma skin cancer (RR=11.0), and 2 or more histologically distinct SNs (RR=18.6).
The researchers said the higher risk of SNs in CCSs with P/LP mutations suggests all CCSs should be referred for genetic counseling.
New research has shown that childhood cancer survivors (CCSs) with certain germline mutations have higher relative rates (RRs) of secondary neoplasms (SNs) later in life.
Mutation carriers had significantly higher rates of breast cancer and sarcoma if they had received radiation to treat their initial cancer.
Among CCSs who did not receive radiation, the mutations were associated with increased rates of any SN, breast cancer, nonmelanoma skin cancer, and 2 or more histologically distinct SNs.
These findings were reported in the Journal of Clinical Oncology.
Researchers sequenced samples from 3006 CCSs who were at least 5 years from their initial cancer diagnosis as well as 341 samples from cancer-free control subjects.
All subjects were participants in the St. Jude Lifetime Cohort Study, a retrospective study with prospective clinical follow-up.
Thirty-five percent of the CCSs had survived leukemia, and 19% had survived lymphoma.
The CCS’s median age at childhood cancer diagnosis was 7.1 years, and the median follow-up was 28 years. The controls had a median age of 36.4 at follow-up.
Results
There were 1120 SNs diagnosed in 439 CCSs (14.6%). Ninety-one CCSs developed 2 or more histologically distinct SNs. The median time to SN diagnosis was 25.6 years
Non-melanoma skin cancer (580 in 159 CCSs), meningiomas (233 in 102 CCSs), thyroid cancer (67 in 67 CCSs), and breast cancer (60 in 53 CCSs) were among the SNs reported.
There were 15 neoplasms recorded in the control group—14 cases of non-melanoma skin cancer and 1 meningioma.
Pathogenic or likely pathogenic (P/LP) mutations in 32 genes were reported in 175 CCSs. The prevalence in CCSs (5.8%) was nearly 10-fold higher than in controls (0.6%).
The most commonly mutated genes in CCSs were RB1 (n=43), NF1 (n=22), BRCA2 (n=14), BRCA1 (n=12), and TP53 (n=10).
In a multivariable analysis adjusted for sex, age at primary cancer diagnosis, and treatment, P/LP mutation carriers had a significantly higher rate of any SN (RR=1.8).
The rate of subsequent breast cancer was significantly increased among females with a P/LP mutation (RR= 9.4), recipients of chest radiation (RR=7.9), and those with higher anthracycline exposure (RR=2.4).
The rate of subsequent sarcoma was significantly increased for mutation carriers (RR=10.9) and CCSs with greater exposure to alkylating agents (RR=3.8).
Among irradiated CCSs, P/LP mutations were associated with significantly increased rates of breast cancer (RR=13.9) and sarcoma (RR=10.6)
Among non-irradiated CCSs, P/LP mutations were associated with significantly increased rates of any SN (RR=4.7), breast cancer (RR=7.7), nonmelanoma skin cancer (RR=11.0), and 2 or more histologically distinct SNs (RR=18.6).
The researchers said the higher risk of SNs in CCSs with P/LP mutations suggests all CCSs should be referred for genetic counseling.
CAR T Therapy: From Bench to Bedside and Back
Release Date: July 15, 2018
Expiration Date: July 14, 2019
Note: This activity is no longer available for credit
Introductory Comments: (Duration: 9 minutes)
Aaron P. Rapoport, MD
Bone Marrow Transplant Program
University of Maryland School of Medicine
Baltimore, MD
Presentation: (Duration: 39 minutes)
Carl H. June, MD
Richard W. Vague Professor in Immunotherapy
Perelman School of Medicine
University of Pennsylvania
Philadelphia, PA
Provided by:

Learning Objectives
• Review clinical data and individual case studies to determine where CAR T-cell therapy might be appropriate in the treatment of adult and pediatric patients with leukemia, lymphoma, and multiple myeloma.
• Discuss the management of cytotoxicity of CAR T-cell therapy.
Target Audience
Hematologists, oncologists, and other members of the healthcare team who treat or manage patients with hematologic malignancies.
Statement of Need
It is critical that clinicians managing patients with acute leukemia and other hematologic malignancies are cognizant of exciting breakthroughs and are also able to integrate recent progress into practice. However, given the overwhelming influx of data, it is no surprise that many hematology professionals face difficulties in identifying the most relevant findings for clinical practice. Hematologists are unable to stay abreast of the latest evidence on investigational agents. Educational programs are thus crucial to address this important professional practice gap.
Faculty
Carl H. June, MD
Richard W. Vague Professor in Immunotherapy
Perelman School of Medicine
University of Pennsylvania
Philadelphia, PA
Disclosures: Consultant: Novartis; Grant/Research support and royalties/IPR: Novartis
Stockholder: Tmunity Therapeutics, Inc.
Aaron P. Rapoport, MD
Bone Marrow Transplant Program
University of Maryland School of Medicine
Baltimore, Maryland
Disclosures: No relevant financial relationships with a commercial supporter
Permissions
- Slide 3: Complex tumor, host and environmental factors govern the strength and timing of anti-cancer immune responses
- Reprinted from Immunity, Vol 39/No 1, Chen DS, Mellman I, Oncology meets immunology: the cancer-immunity cycle, pp 1-10, 2013, with permission from Elsevier
- Slide 9: Genes differentially expressed in CART19 cellular infusion products from CLL patients
- From Fraietta JA, Lacey SF, Orlando EJ, . . . June CH, Melenhorst JJ. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 2018; 24:563-571
- Slide 10: Characterization of CLL CAR T cells in NSG CLL model
- Same as slide 9
- Slide 15: First adult ALL patient
- Photos originally published in Kaiser Health News/Photo courtesy of Dr Keith Eaton. Available at: https://khn.org/news/cascade-of-costs-could-push-new-gene-therapy-above-1-million-per-patient/
- Slide 21: Efficient trafficking of CTL019 T Cells to CNS in ALL
- From N Engl J Med, Grupp SA, Kalos M, Barrett D, . . V. June CH, Chimeric antigen receptor-modified T cells for acute lymphoid leukemia, Volume No 368, pp 1509-1518. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
- Slide 26: Long-term persistence and expression of CTL019 is associated with durable remission in leukemia: Predictive Biomarker
- From Porter DL, Hwang WT, Frey NV . . . June CH. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med 2015; 7(303):303ra139. Reprinted with permission from AAAS.
- Slide 28: Rapid massive expansion of clonal CART cell population in patient #10
- Initially published in Fraietta JA, Nobles CL, Sammons MA, . . . June CH, Melenhors JJ. Disruption of TET2 promotes the therapeutic efficacy of CD19-targeted T cells. Nature 2018; 558(7709):307-312
- Slide 29: Mapping CAR integration site in Pt #10
- Same as slide 28.
- Slide 31: Long-term stable persistence of TET2-deficient CAR T cells in Pt #10
- Same as slide 28
- Slide 32: Epigenetic and genetic changes uncovered by ATAC-seq in Pt #10
- Same as slide 28.
- Slide 33: TET2 knock down in healthy donor T cells
- Same as slide 28.
- Slide 34: TET2 knock down in healthy donor T cells
- Same as slide 28.
- Slide 36: CAR T for myeloma: BCMA
- From Rickert RC, Jellusova J, Miletic AV. Signaling by the tumor necrosis factor receptor superfamily in B-cell biology and disease. Immunol Rev 2011; 244(1):115-33. Reprinted with permission from John Wiley and Sons.
- Slide 38: CAR T for myeloma: Patient #1
- Photo originally published by UT Southwestern Medical Center. Available at: https://www.utsouthwestern.edu/newsroom/articles/year-2018/wright-car-t.html
- Slide 39: Autoimmunity is the “Achilles’ Heel” of immunotherapy
- First published in June CH, Warshauer JT, and Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med 2017;23(5):540-7
- Slide 41: Multiplex CRISPR /Cas9 editing: Universal T cells TCR, HLA, PD-1, CTLA-4 and Fas
- From Ren J, Zhang X, Liu X, Fang C, Jiang S, June CH, Zhao Y. A versatile system for rapid multiplex genome-edited CAR T cell generation. Oncotarget 2017; 8:17002-17011.
- Slide 45: CAR T-cell trials for cancer are now global
- From June CH, O’Connor RS, Kawalekar OU, Ghassemi S, Milone MC. CAR T cell immunotherapy for human cancer. Science 2018; 359:1361-1365. Reprinted with permission from AAAS.
Disclaimer
The content and views presented in this educational activity are those of the author and do not necessarily reflect those of Hemedicus or Frontline Medical Communications. This material is prepared based upon a review of multiple sources of information, but it is not exhaustive of the subject matter. Therefore, healthcare professionals and other individuals should review and consider other publications and materials on the subject matter before relying solely upon the information contained within this educational activity.
Release Date: July 15, 2018
Expiration Date: July 14, 2019
Note: This activity is no longer available for credit
Introductory Comments: (Duration: 9 minutes)
Aaron P. Rapoport, MD
Bone Marrow Transplant Program
University of Maryland School of Medicine
Baltimore, MD
Presentation: (Duration: 39 minutes)
Carl H. June, MD
Richard W. Vague Professor in Immunotherapy
Perelman School of Medicine
University of Pennsylvania
Philadelphia, PA
Provided by:

Learning Objectives
• Review clinical data and individual case studies to determine where CAR T-cell therapy might be appropriate in the treatment of adult and pediatric patients with leukemia, lymphoma, and multiple myeloma.
• Discuss the management of cytotoxicity of CAR T-cell therapy.
Target Audience
Hematologists, oncologists, and other members of the healthcare team who treat or manage patients with hematologic malignancies.
Statement of Need
It is critical that clinicians managing patients with acute leukemia and other hematologic malignancies are cognizant of exciting breakthroughs and are also able to integrate recent progress into practice. However, given the overwhelming influx of data, it is no surprise that many hematology professionals face difficulties in identifying the most relevant findings for clinical practice. Hematologists are unable to stay abreast of the latest evidence on investigational agents. Educational programs are thus crucial to address this important professional practice gap.
Faculty
Carl H. June, MD
Richard W. Vague Professor in Immunotherapy
Perelman School of Medicine
University of Pennsylvania
Philadelphia, PA
Disclosures: Consultant: Novartis; Grant/Research support and royalties/IPR: Novartis
Stockholder: Tmunity Therapeutics, Inc.
Aaron P. Rapoport, MD
Bone Marrow Transplant Program
University of Maryland School of Medicine
Baltimore, Maryland
Disclosures: No relevant financial relationships with a commercial supporter
Permissions
- Slide 3: Complex tumor, host and environmental factors govern the strength and timing of anti-cancer immune responses
- Reprinted from Immunity, Vol 39/No 1, Chen DS, Mellman I, Oncology meets immunology: the cancer-immunity cycle, pp 1-10, 2013, with permission from Elsevier
- Slide 9: Genes differentially expressed in CART19 cellular infusion products from CLL patients
- From Fraietta JA, Lacey SF, Orlando EJ, . . . June CH, Melenhorst JJ. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 2018; 24:563-571
- Slide 10: Characterization of CLL CAR T cells in NSG CLL model
- Same as slide 9
- Slide 15: First adult ALL patient
- Photos originally published in Kaiser Health News/Photo courtesy of Dr Keith Eaton. Available at: https://khn.org/news/cascade-of-costs-could-push-new-gene-therapy-above-1-million-per-patient/
- Slide 21: Efficient trafficking of CTL019 T Cells to CNS in ALL
- From N Engl J Med, Grupp SA, Kalos M, Barrett D, . . V. June CH, Chimeric antigen receptor-modified T cells for acute lymphoid leukemia, Volume No 368, pp 1509-1518. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
- Slide 26: Long-term persistence and expression of CTL019 is associated with durable remission in leukemia: Predictive Biomarker
- From Porter DL, Hwang WT, Frey NV . . . June CH. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med 2015; 7(303):303ra139. Reprinted with permission from AAAS.
- Slide 28: Rapid massive expansion of clonal CART cell population in patient #10
- Initially published in Fraietta JA, Nobles CL, Sammons MA, . . . June CH, Melenhors JJ. Disruption of TET2 promotes the therapeutic efficacy of CD19-targeted T cells. Nature 2018; 558(7709):307-312
- Slide 29: Mapping CAR integration site in Pt #10
- Same as slide 28.
- Slide 31: Long-term stable persistence of TET2-deficient CAR T cells in Pt #10
- Same as slide 28
- Slide 32: Epigenetic and genetic changes uncovered by ATAC-seq in Pt #10
- Same as slide 28.
- Slide 33: TET2 knock down in healthy donor T cells
- Same as slide 28.
- Slide 34: TET2 knock down in healthy donor T cells
- Same as slide 28.
- Slide 36: CAR T for myeloma: BCMA
- From Rickert RC, Jellusova J, Miletic AV. Signaling by the tumor necrosis factor receptor superfamily in B-cell biology and disease. Immunol Rev 2011; 244(1):115-33. Reprinted with permission from John Wiley and Sons.
- Slide 38: CAR T for myeloma: Patient #1
- Photo originally published by UT Southwestern Medical Center. Available at: https://www.utsouthwestern.edu/newsroom/articles/year-2018/wright-car-t.html
- Slide 39: Autoimmunity is the “Achilles’ Heel” of immunotherapy
- First published in June CH, Warshauer JT, and Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med 2017;23(5):540-7
- Slide 41: Multiplex CRISPR /Cas9 editing: Universal T cells TCR, HLA, PD-1, CTLA-4 and Fas
- From Ren J, Zhang X, Liu X, Fang C, Jiang S, June CH, Zhao Y. A versatile system for rapid multiplex genome-edited CAR T cell generation. Oncotarget 2017; 8:17002-17011.
- Slide 45: CAR T-cell trials for cancer are now global
- From June CH, O’Connor RS, Kawalekar OU, Ghassemi S, Milone MC. CAR T cell immunotherapy for human cancer. Science 2018; 359:1361-1365. Reprinted with permission from AAAS.
Disclaimer
The content and views presented in this educational activity are those of the author and do not necessarily reflect those of Hemedicus or Frontline Medical Communications. This material is prepared based upon a review of multiple sources of information, but it is not exhaustive of the subject matter. Therefore, healthcare professionals and other individuals should review and consider other publications and materials on the subject matter before relying solely upon the information contained within this educational activity.
Release Date: July 15, 2018
Expiration Date: July 14, 2019
Note: This activity is no longer available for credit
Introductory Comments: (Duration: 9 minutes)
Aaron P. Rapoport, MD
Bone Marrow Transplant Program
University of Maryland School of Medicine
Baltimore, MD
Presentation: (Duration: 39 minutes)
Carl H. June, MD
Richard W. Vague Professor in Immunotherapy
Perelman School of Medicine
University of Pennsylvania
Philadelphia, PA
Provided by:

Learning Objectives
• Review clinical data and individual case studies to determine where CAR T-cell therapy might be appropriate in the treatment of adult and pediatric patients with leukemia, lymphoma, and multiple myeloma.
• Discuss the management of cytotoxicity of CAR T-cell therapy.
Target Audience
Hematologists, oncologists, and other members of the healthcare team who treat or manage patients with hematologic malignancies.
Statement of Need
It is critical that clinicians managing patients with acute leukemia and other hematologic malignancies are cognizant of exciting breakthroughs and are also able to integrate recent progress into practice. However, given the overwhelming influx of data, it is no surprise that many hematology professionals face difficulties in identifying the most relevant findings for clinical practice. Hematologists are unable to stay abreast of the latest evidence on investigational agents. Educational programs are thus crucial to address this important professional practice gap.
Faculty
Carl H. June, MD
Richard W. Vague Professor in Immunotherapy
Perelman School of Medicine
University of Pennsylvania
Philadelphia, PA
Disclosures: Consultant: Novartis; Grant/Research support and royalties/IPR: Novartis
Stockholder: Tmunity Therapeutics, Inc.
Aaron P. Rapoport, MD
Bone Marrow Transplant Program
University of Maryland School of Medicine
Baltimore, Maryland
Disclosures: No relevant financial relationships with a commercial supporter
Permissions
- Slide 3: Complex tumor, host and environmental factors govern the strength and timing of anti-cancer immune responses
- Reprinted from Immunity, Vol 39/No 1, Chen DS, Mellman I, Oncology meets immunology: the cancer-immunity cycle, pp 1-10, 2013, with permission from Elsevier
- Slide 9: Genes differentially expressed in CART19 cellular infusion products from CLL patients
- From Fraietta JA, Lacey SF, Orlando EJ, . . . June CH, Melenhorst JJ. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 2018; 24:563-571
- Slide 10: Characterization of CLL CAR T cells in NSG CLL model
- Same as slide 9
- Slide 15: First adult ALL patient
- Photos originally published in Kaiser Health News/Photo courtesy of Dr Keith Eaton. Available at: https://khn.org/news/cascade-of-costs-could-push-new-gene-therapy-above-1-million-per-patient/
- Slide 21: Efficient trafficking of CTL019 T Cells to CNS in ALL
- From N Engl J Med, Grupp SA, Kalos M, Barrett D, . . V. June CH, Chimeric antigen receptor-modified T cells for acute lymphoid leukemia, Volume No 368, pp 1509-1518. Copyright © 2013 Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.
- Slide 26: Long-term persistence and expression of CTL019 is associated with durable remission in leukemia: Predictive Biomarker
- From Porter DL, Hwang WT, Frey NV . . . June CH. Chimeric antigen receptor T cells persist and induce sustained remissions in relapsed refractory chronic lymphocytic leukemia. Sci Transl Med 2015; 7(303):303ra139. Reprinted with permission from AAAS.
- Slide 28: Rapid massive expansion of clonal CART cell population in patient #10
- Initially published in Fraietta JA, Nobles CL, Sammons MA, . . . June CH, Melenhors JJ. Disruption of TET2 promotes the therapeutic efficacy of CD19-targeted T cells. Nature 2018; 558(7709):307-312
- Slide 29: Mapping CAR integration site in Pt #10
- Same as slide 28.
- Slide 31: Long-term stable persistence of TET2-deficient CAR T cells in Pt #10
- Same as slide 28
- Slide 32: Epigenetic and genetic changes uncovered by ATAC-seq in Pt #10
- Same as slide 28.
- Slide 33: TET2 knock down in healthy donor T cells
- Same as slide 28.
- Slide 34: TET2 knock down in healthy donor T cells
- Same as slide 28.
- Slide 36: CAR T for myeloma: BCMA
- From Rickert RC, Jellusova J, Miletic AV. Signaling by the tumor necrosis factor receptor superfamily in B-cell biology and disease. Immunol Rev 2011; 244(1):115-33. Reprinted with permission from John Wiley and Sons.
- Slide 38: CAR T for myeloma: Patient #1
- Photo originally published by UT Southwestern Medical Center. Available at: https://www.utsouthwestern.edu/newsroom/articles/year-2018/wright-car-t.html
- Slide 39: Autoimmunity is the “Achilles’ Heel” of immunotherapy
- First published in June CH, Warshauer JT, and Bluestone JA. Is autoimmunity the Achilles’ heel of cancer immunotherapy? Nat Med 2017;23(5):540-7
- Slide 41: Multiplex CRISPR /Cas9 editing: Universal T cells TCR, HLA, PD-1, CTLA-4 and Fas
- From Ren J, Zhang X, Liu X, Fang C, Jiang S, June CH, Zhao Y. A versatile system for rapid multiplex genome-edited CAR T cell generation. Oncotarget 2017; 8:17002-17011.
- Slide 45: CAR T-cell trials for cancer are now global
- From June CH, O’Connor RS, Kawalekar OU, Ghassemi S, Milone MC. CAR T cell immunotherapy for human cancer. Science 2018; 359:1361-1365. Reprinted with permission from AAAS.
Disclaimer
The content and views presented in this educational activity are those of the author and do not necessarily reflect those of Hemedicus or Frontline Medical Communications. This material is prepared based upon a review of multiple sources of information, but it is not exhaustive of the subject matter. Therefore, healthcare professionals and other individuals should review and consider other publications and materials on the subject matter before relying solely upon the information contained within this educational activity.
New guideline for managing MCL
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
Rituximab should be included in first-line chemotherapy when treating mantle cell lymphoma, according to a new management guideline from the British Society for Haematology.
The best outcome data is for the R-CHOP regimen (rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisolone) followed by maintenance treatment with rituximab, wrote Pamela McKay, MD, of Beatson West of Scotland Cancer Centre in Glasgow, and her colleagues. The report was published in the British Journal of Haematology. But the combination of rituximab and bendamustine is also effective and a more favorable safety profile, according to the guideline. Single agent rituximab is not recommended.
At relapse, the guideline calls on physicians to take an individualized approach based on age, comorbidities, performance status, and response to prior therapy. Some options to consider include ibrutinib as a single agent or rituximab plus chemotherapy. The authors cautioned that there is little evidence to support maintenance rituximab after relapse treatment.
The guideline also explores the role of autologous stem cell transplantation (ASCT) and allogeneic SCT (alloSCT). The authors recommend that ASCT be considered as consolidation of first-line therapy for patients who are fit for intensive therapy. AlloSCT is a viable option in second remission among fit patients who have an appropriate donor and it may also be effective as a rescue therapy for patients who relapse after ASCT. But alloSCT is appropriate only as a first-line therapy for high-risk patients and is best used as part of a clinical trial, according to the recommendations.
The British Society of Haematology previously issued guidance on mantle cell lymphoma in 2012, but the updated document includes new drug therapeutic options and transplant data. The guideline includes a therapeutic algorithm to assist physicians in choosing first-line therapy, options after first relapse, and management in the case of higher relapse.
The guideline authors reported having no conflicts of interest.
SOURCE: McKay P et al. Br J Haematol. 2018 Jul;182(1):46-62.
FROM THE BRITISH JOURNAL OF HAEMATOLOGY
Promising phase 3 results for ixazomib in multiple myeloma
who had responded to high-dose therapy and autologous stem cell transplant.
The drug’s sponsor, Takeda, announced that the oral proteasome inhibitor had met the primary endpoint – progression-free survival versus placebo – in the randomized, phase 3 TOURMALINE-MM3 study. They also reported that adverse events were consistent with previously reported results for single-agent use of ixazomib and that there were no new safety signals.
Full study results will be presented at the annual meeting of the American Society of Hematology. Company officials plan to submit the trial data to the Food and Drug Administration and regulatory agencies around the world to gain approval of ixazomib as a single-agent maintenance therapy, according to a Takeda announcement.
The TOURMALINE-MM3 study is a double-blind study of 656 patients with multiple myeloma who have had complete response, very good partial response, or partial response to induction therapy followed by high-dose therapy and autologous stem cell transplant. In addition to progression-free survival, the trial assessed overall survival.
who had responded to high-dose therapy and autologous stem cell transplant.
The drug’s sponsor, Takeda, announced that the oral proteasome inhibitor had met the primary endpoint – progression-free survival versus placebo – in the randomized, phase 3 TOURMALINE-MM3 study. They also reported that adverse events were consistent with previously reported results for single-agent use of ixazomib and that there were no new safety signals.
Full study results will be presented at the annual meeting of the American Society of Hematology. Company officials plan to submit the trial data to the Food and Drug Administration and regulatory agencies around the world to gain approval of ixazomib as a single-agent maintenance therapy, according to a Takeda announcement.
The TOURMALINE-MM3 study is a double-blind study of 656 patients with multiple myeloma who have had complete response, very good partial response, or partial response to induction therapy followed by high-dose therapy and autologous stem cell transplant. In addition to progression-free survival, the trial assessed overall survival.
who had responded to high-dose therapy and autologous stem cell transplant.
The drug’s sponsor, Takeda, announced that the oral proteasome inhibitor had met the primary endpoint – progression-free survival versus placebo – in the randomized, phase 3 TOURMALINE-MM3 study. They also reported that adverse events were consistent with previously reported results for single-agent use of ixazomib and that there were no new safety signals.
Full study results will be presented at the annual meeting of the American Society of Hematology. Company officials plan to submit the trial data to the Food and Drug Administration and regulatory agencies around the world to gain approval of ixazomib as a single-agent maintenance therapy, according to a Takeda announcement.
The TOURMALINE-MM3 study is a double-blind study of 656 patients with multiple myeloma who have had complete response, very good partial response, or partial response to induction therapy followed by high-dose therapy and autologous stem cell transplant. In addition to progression-free survival, the trial assessed overall survival.
Ibrutinib/venetoclax shows early promise in relapsed CLL
STOCKHOLM – A chemotherapy-free regimen of ibrutinib plus venetoclax was generally safe and showed promising early efficacy in patients with relapsed or refractory chronic lymphocytic leukemia, investigators reported.
A planned interim analysis performed after the first 15 patients who had received two cycles of ibrutinib plus one of ibrutinib and venetoclax showed no treatment-related deaths or treatment interruptions, and all patients had clinical responses, including 8 with complete clinical remission (CR), reported Carsten U. Niemann, MD, PhD, from Rigshospitalet in Copenhagen, and his colleagues.
The goal of the ongoing VISION/HOVEN 141 study is to evaluate whether minimal residual disease (MRD)–guided therapy with the Bruton tyrosine kinase inhibitor ibrutinib and the BCL2 inhibitor venetoclax could lead to MRD negativity and allow select patients to stop treatment, Dr. Niemann said in an interview at the annual congress of the European Hematology Association.
“It’s a 100% clinical response rate and 53% CR. Obviously these are clinical responses, so we don’t have the CT scans, we don’t have the bone marrow biopsies, but we’re very happy to see even in the relapsed/refractory setting such good response rates,” he said.
The investigators are enrolling patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic leukemia requiring treatment and starting all patients on ibrutinib 420 mg daily for the first 2 cycles, with venetoclax added in a 5-week ramp-up from 20 mg beginning with cycle 3 to a final dose of 400 mg daily for 15 total treatment cycles.
At the end of the induction phase, patients who are determined to be MRD-negative by flow cytometry at cycles 12 and 15, and by bone marrow at cycle 15, are randomized on a 1:2 basis to ibrutinib maintenance until disease progression or intolerable toxicity, or to observation until progression or loss of MRD negativity, at which time they start maintenance with ibrutinib until progression or toxicity, plus 12 months of venetoclax.
All 15 patients who were followed for 3 months had clinical responses, including 8 CRs (53%), 6 partial remissions (40%), and 1 partial remission with lymphocytosis (7%).
Three patients had ibrutinib dose reductions and two had venetoclax dose reductions, but no patients stopped treatment. Three patients had grade 2 adverse events (AEs), three had grade 3 AEs, and two had grade 4 AEs. There were no grade 5 AEs.
Two patients had serious AEs during the first two cycles with ibrutinib alone, one of which was a case of febrile neutropenia and one which was an adenocarcinoma of the lung. There were no serious AEs reported during venetoclax ramp-up. To date, there have been no cases of tumor lysis syndrome, atrial fibrillation, or bleeding events reported.
The results suggest that treatment with ibrutinib and venetoclax ramp-up is manageable in this patient population, and the study is ongoing, with further results expected to be reported at either the 2018 annual meeting of the American Society of Hematology or the 2019 annual meeting of the American Society of Clinical Oncology, Dr. Niemann said.
The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
SOURCE: Niemann CU et al. EHA Congress, Abstract PF346.
STOCKHOLM – A chemotherapy-free regimen of ibrutinib plus venetoclax was generally safe and showed promising early efficacy in patients with relapsed or refractory chronic lymphocytic leukemia, investigators reported.
A planned interim analysis performed after the first 15 patients who had received two cycles of ibrutinib plus one of ibrutinib and venetoclax showed no treatment-related deaths or treatment interruptions, and all patients had clinical responses, including 8 with complete clinical remission (CR), reported Carsten U. Niemann, MD, PhD, from Rigshospitalet in Copenhagen, and his colleagues.
The goal of the ongoing VISION/HOVEN 141 study is to evaluate whether minimal residual disease (MRD)–guided therapy with the Bruton tyrosine kinase inhibitor ibrutinib and the BCL2 inhibitor venetoclax could lead to MRD negativity and allow select patients to stop treatment, Dr. Niemann said in an interview at the annual congress of the European Hematology Association.
“It’s a 100% clinical response rate and 53% CR. Obviously these are clinical responses, so we don’t have the CT scans, we don’t have the bone marrow biopsies, but we’re very happy to see even in the relapsed/refractory setting such good response rates,” he said.
The investigators are enrolling patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic leukemia requiring treatment and starting all patients on ibrutinib 420 mg daily for the first 2 cycles, with venetoclax added in a 5-week ramp-up from 20 mg beginning with cycle 3 to a final dose of 400 mg daily for 15 total treatment cycles.
At the end of the induction phase, patients who are determined to be MRD-negative by flow cytometry at cycles 12 and 15, and by bone marrow at cycle 15, are randomized on a 1:2 basis to ibrutinib maintenance until disease progression or intolerable toxicity, or to observation until progression or loss of MRD negativity, at which time they start maintenance with ibrutinib until progression or toxicity, plus 12 months of venetoclax.
All 15 patients who were followed for 3 months had clinical responses, including 8 CRs (53%), 6 partial remissions (40%), and 1 partial remission with lymphocytosis (7%).
Three patients had ibrutinib dose reductions and two had venetoclax dose reductions, but no patients stopped treatment. Three patients had grade 2 adverse events (AEs), three had grade 3 AEs, and two had grade 4 AEs. There were no grade 5 AEs.
Two patients had serious AEs during the first two cycles with ibrutinib alone, one of which was a case of febrile neutropenia and one which was an adenocarcinoma of the lung. There were no serious AEs reported during venetoclax ramp-up. To date, there have been no cases of tumor lysis syndrome, atrial fibrillation, or bleeding events reported.
The results suggest that treatment with ibrutinib and venetoclax ramp-up is manageable in this patient population, and the study is ongoing, with further results expected to be reported at either the 2018 annual meeting of the American Society of Hematology or the 2019 annual meeting of the American Society of Clinical Oncology, Dr. Niemann said.
The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
SOURCE: Niemann CU et al. EHA Congress, Abstract PF346.
STOCKHOLM – A chemotherapy-free regimen of ibrutinib plus venetoclax was generally safe and showed promising early efficacy in patients with relapsed or refractory chronic lymphocytic leukemia, investigators reported.
A planned interim analysis performed after the first 15 patients who had received two cycles of ibrutinib plus one of ibrutinib and venetoclax showed no treatment-related deaths or treatment interruptions, and all patients had clinical responses, including 8 with complete clinical remission (CR), reported Carsten U. Niemann, MD, PhD, from Rigshospitalet in Copenhagen, and his colleagues.
The goal of the ongoing VISION/HOVEN 141 study is to evaluate whether minimal residual disease (MRD)–guided therapy with the Bruton tyrosine kinase inhibitor ibrutinib and the BCL2 inhibitor venetoclax could lead to MRD negativity and allow select patients to stop treatment, Dr. Niemann said in an interview at the annual congress of the European Hematology Association.
“It’s a 100% clinical response rate and 53% CR. Obviously these are clinical responses, so we don’t have the CT scans, we don’t have the bone marrow biopsies, but we’re very happy to see even in the relapsed/refractory setting such good response rates,” he said.
The investigators are enrolling patients with relapsed/refractory chronic lymphocytic leukemia or small lymphocytic leukemia requiring treatment and starting all patients on ibrutinib 420 mg daily for the first 2 cycles, with venetoclax added in a 5-week ramp-up from 20 mg beginning with cycle 3 to a final dose of 400 mg daily for 15 total treatment cycles.
At the end of the induction phase, patients who are determined to be MRD-negative by flow cytometry at cycles 12 and 15, and by bone marrow at cycle 15, are randomized on a 1:2 basis to ibrutinib maintenance until disease progression or intolerable toxicity, or to observation until progression or loss of MRD negativity, at which time they start maintenance with ibrutinib until progression or toxicity, plus 12 months of venetoclax.
All 15 patients who were followed for 3 months had clinical responses, including 8 CRs (53%), 6 partial remissions (40%), and 1 partial remission with lymphocytosis (7%).
Three patients had ibrutinib dose reductions and two had venetoclax dose reductions, but no patients stopped treatment. Three patients had grade 2 adverse events (AEs), three had grade 3 AEs, and two had grade 4 AEs. There were no grade 5 AEs.
Two patients had serious AEs during the first two cycles with ibrutinib alone, one of which was a case of febrile neutropenia and one which was an adenocarcinoma of the lung. There were no serious AEs reported during venetoclax ramp-up. To date, there have been no cases of tumor lysis syndrome, atrial fibrillation, or bleeding events reported.
The results suggest that treatment with ibrutinib and venetoclax ramp-up is manageable in this patient population, and the study is ongoing, with further results expected to be reported at either the 2018 annual meeting of the American Society of Hematology or the 2019 annual meeting of the American Society of Clinical Oncology, Dr. Niemann said.
The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
SOURCE: Niemann CU et al. EHA Congress, Abstract PF346.
REPORTING FROM THE EHA CONGRESS
Key clinical point:
Major finding: All of the 15 patients analyzed to date had clinical responses to the combination, including 8 complete clinical remissions.
Study details: An ongoing, open-label, phase 2, randomized trial in patients with relapsed/refractory chronic lymphocytic leukemia/small lymphocytic leukemia.
Disclosures: The study is supported by AbbVie and Janssen, which supplied the drugs and had the right to comment on the presentation. Dr. Niemann has previously disclosed consultancy fees from those companies and others.
Source: Niemann CU et al. EHA Congress, Abstract PF346
New approach to AE reporting needed, group says
A group of experts has called for improvements in reporting adverse events (AEs) that occur in patients with hematologic malignancies.
The group highlighted deficiencies in capturing chronic, cumulative, and late AEs; collecting patient-reported outcomes (PROs); reporting AEs associated with hematopoietic stem cell transplant (HSCT); assessing long-term toxicity in survivors; reporting AEs to regulatory agencies; and tracking AEs that occur in routine clinical practice.
The experts discussed these problems and made recommendations for fixing them in The Lancet Haematology.
“The success in outcomes and survival in many hematological malignancies is historically unparalleled and fueled by scientific discovery and implementation,” said author Gita Thanarajasingam, MD, of the Mayo Clinic in Rochester, Minnesota.
“Patients are now living with the challenge of managing not just their hematological malignancy but also managing chronic therapy for their illness, with new types of acute, chronic, cumulative, and late toxicities. Measures to address the broad facets of toxicity assessment must be prioritized and further developed to ultimately enhance accurate, comprehensive, patient-centered toxicity reporting that will meaningfully inform the care of patients with blood cancers.”
Dr Thanarajasingam and a group of clinicians, investigators, regulators, biostatisticians, and patient advocates analyzed the evidence on AEs and proposed recommendations to policy makers, researchers, industry, and regulators.
First, the group noted that chronic, delayed, and cumulative AEs may go unreported in patients with hematologic malignancies. Therefore, longitudinal methods for AE analysis are needed, and phase 1 trials should have longer periods for evaluating dose-limiting toxicity.
The experts also said PROs are often overlooked, but it should be standard to assess PROs in clinical trials of patients with hematologic malignancies.
Another of the group’s concerns is the “cumbersome” reporting of AEs associated with HSCT acting as a barrier to clinical trials. The experts recommended using registry data to develop a consensus on expected AEs after HSCT.
The group also highlighted deficiencies in the “description and management” of cumulative and late toxicities in survivors of hematologic malignancies. Potential solutions to this problem include developing infrastructure to collect data for adult survivors and standardizing the use and content of survivorship care plans.
The experts said “meaningful” AEs are often underreported to regulatory agencies, so better systems are needed for collecting and analyzing AE data, and the electronic submission of AEs should be simplified.
Finally, the group said AEs occurring in routine clinical practice are difficult to capture and analyze on a large scale. This suggests a need to optimize the systematic, objective collection of toxicity data at multiple points in real-world databases, according to the experts.
Additional details and recommendations are available in the full report.
A group of experts has called for improvements in reporting adverse events (AEs) that occur in patients with hematologic malignancies.
The group highlighted deficiencies in capturing chronic, cumulative, and late AEs; collecting patient-reported outcomes (PROs); reporting AEs associated with hematopoietic stem cell transplant (HSCT); assessing long-term toxicity in survivors; reporting AEs to regulatory agencies; and tracking AEs that occur in routine clinical practice.
The experts discussed these problems and made recommendations for fixing them in The Lancet Haematology.
“The success in outcomes and survival in many hematological malignancies is historically unparalleled and fueled by scientific discovery and implementation,” said author Gita Thanarajasingam, MD, of the Mayo Clinic in Rochester, Minnesota.
“Patients are now living with the challenge of managing not just their hematological malignancy but also managing chronic therapy for their illness, with new types of acute, chronic, cumulative, and late toxicities. Measures to address the broad facets of toxicity assessment must be prioritized and further developed to ultimately enhance accurate, comprehensive, patient-centered toxicity reporting that will meaningfully inform the care of patients with blood cancers.”
Dr Thanarajasingam and a group of clinicians, investigators, regulators, biostatisticians, and patient advocates analyzed the evidence on AEs and proposed recommendations to policy makers, researchers, industry, and regulators.
First, the group noted that chronic, delayed, and cumulative AEs may go unreported in patients with hematologic malignancies. Therefore, longitudinal methods for AE analysis are needed, and phase 1 trials should have longer periods for evaluating dose-limiting toxicity.
The experts also said PROs are often overlooked, but it should be standard to assess PROs in clinical trials of patients with hematologic malignancies.
Another of the group’s concerns is the “cumbersome” reporting of AEs associated with HSCT acting as a barrier to clinical trials. The experts recommended using registry data to develop a consensus on expected AEs after HSCT.
The group also highlighted deficiencies in the “description and management” of cumulative and late toxicities in survivors of hematologic malignancies. Potential solutions to this problem include developing infrastructure to collect data for adult survivors and standardizing the use and content of survivorship care plans.
The experts said “meaningful” AEs are often underreported to regulatory agencies, so better systems are needed for collecting and analyzing AE data, and the electronic submission of AEs should be simplified.
Finally, the group said AEs occurring in routine clinical practice are difficult to capture and analyze on a large scale. This suggests a need to optimize the systematic, objective collection of toxicity data at multiple points in real-world databases, according to the experts.
Additional details and recommendations are available in the full report.
A group of experts has called for improvements in reporting adverse events (AEs) that occur in patients with hematologic malignancies.
The group highlighted deficiencies in capturing chronic, cumulative, and late AEs; collecting patient-reported outcomes (PROs); reporting AEs associated with hematopoietic stem cell transplant (HSCT); assessing long-term toxicity in survivors; reporting AEs to regulatory agencies; and tracking AEs that occur in routine clinical practice.
The experts discussed these problems and made recommendations for fixing them in The Lancet Haematology.
“The success in outcomes and survival in many hematological malignancies is historically unparalleled and fueled by scientific discovery and implementation,” said author Gita Thanarajasingam, MD, of the Mayo Clinic in Rochester, Minnesota.
“Patients are now living with the challenge of managing not just their hematological malignancy but also managing chronic therapy for their illness, with new types of acute, chronic, cumulative, and late toxicities. Measures to address the broad facets of toxicity assessment must be prioritized and further developed to ultimately enhance accurate, comprehensive, patient-centered toxicity reporting that will meaningfully inform the care of patients with blood cancers.”
Dr Thanarajasingam and a group of clinicians, investigators, regulators, biostatisticians, and patient advocates analyzed the evidence on AEs and proposed recommendations to policy makers, researchers, industry, and regulators.
First, the group noted that chronic, delayed, and cumulative AEs may go unreported in patients with hematologic malignancies. Therefore, longitudinal methods for AE analysis are needed, and phase 1 trials should have longer periods for evaluating dose-limiting toxicity.
The experts also said PROs are often overlooked, but it should be standard to assess PROs in clinical trials of patients with hematologic malignancies.
Another of the group’s concerns is the “cumbersome” reporting of AEs associated with HSCT acting as a barrier to clinical trials. The experts recommended using registry data to develop a consensus on expected AEs after HSCT.
The group also highlighted deficiencies in the “description and management” of cumulative and late toxicities in survivors of hematologic malignancies. Potential solutions to this problem include developing infrastructure to collect data for adult survivors and standardizing the use and content of survivorship care plans.
The experts said “meaningful” AEs are often underreported to regulatory agencies, so better systems are needed for collecting and analyzing AE data, and the electronic submission of AEs should be simplified.
Finally, the group said AEs occurring in routine clinical practice are difficult to capture and analyze on a large scale. This suggests a need to optimize the systematic, objective collection of toxicity data at multiple points in real-world databases, according to the experts.
Additional details and recommendations are available in the full report.
Health Canada expands approval of obinutuzumab
Health Canada has expanded the approved use of obinutuzumab (Gazyva®).
The anti-CD20 monoclonal antibody is now approved for use in combination with chemotherapy to treat patients with previously untreated follicular lymphoma (FL) that is advanced (stage II bulky, stage III, or stage IV).
In patients who respond to this treatment, obinutuzumab monotherapy can be given as maintenance.
Health Canada previously approved obinutuzumab for the following indications:
- In combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia
- First in combination with bendamustine, then as monotherapy, in FL patients who relapsed after or are refractory to a rituximab-containing regimen.
Phase 3 results
Health Canada’s latest approval of obinutuzumab is based on results from the phase 3 GALLIUM study, which were published in NEJM in October 2017. The following are updated data from the product monograph.
GALLIUM included 1385 patients with previously untreated non-Hodgkin lymphoma, and 1202 of these patients had previously untreated, advanced FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), CVP (cyclophosphamide, vincristine, and prednisone), and bendamustine.
At a median observation time of 41.1 months, the overall response rate was 91% in the obinutuzumab arm and 88% in the rituximab arm. The complete response rates were 28% and 27%, respectively.
The median progression-free survival was not reached in either arm. The hazard ratio, for obinutuzumab compared to rituximab, was 0.72 (95% CI, 0.56-0.93, P=0.0118).
The estimated 3-year progression-free survival was 78.9% in the rituximab arm and 83.4% in the obinutuzumab arm.
Safety was evaluated based on all 1385 patients in the study, 86% of whom had FL and 14% of whom had marginal zone lymphoma.
Serious adverse events (AEs) occurred in 50% of patients in the obinutuzumab arm and 43% in the rituximab arm. Fatal AEs occurred in 5% and 4%, respectively. Infections and second malignancies were the leading causes of these deaths.
During the monotherapy period, the most common AEs (≥ 5%) in patients treated with obinutuzumab were cough (21%), neutropenia (19%), upper respiratory tract infection (15%), viral upper respiratory tract infection (15%), diarrhea (13%), arthralgia (10%), fatigue (9%), sinusitis (9%), infusion reactions (8%), pneumonia (8%), herpes zoster (8%), lower respiratory tract infection (7%), pyrexia (7%), back pain (6%), headache (6%), urinary tract infection (6%), nausea (6%), bronchitis (5%), and vomiting (5%).
Grade 3-4 AEs (≥1%) in patients treated with obinutuzumab included neutropenia (17%), pneumonia (3%), and febrile neutropenia (2%). There were 2 deaths due to pneumonia in the obinutuzumab arm.
Health Canada has expanded the approved use of obinutuzumab (Gazyva®).
The anti-CD20 monoclonal antibody is now approved for use in combination with chemotherapy to treat patients with previously untreated follicular lymphoma (FL) that is advanced (stage II bulky, stage III, or stage IV).
In patients who respond to this treatment, obinutuzumab monotherapy can be given as maintenance.
Health Canada previously approved obinutuzumab for the following indications:
- In combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia
- First in combination with bendamustine, then as monotherapy, in FL patients who relapsed after or are refractory to a rituximab-containing regimen.
Phase 3 results
Health Canada’s latest approval of obinutuzumab is based on results from the phase 3 GALLIUM study, which were published in NEJM in October 2017. The following are updated data from the product monograph.
GALLIUM included 1385 patients with previously untreated non-Hodgkin lymphoma, and 1202 of these patients had previously untreated, advanced FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), CVP (cyclophosphamide, vincristine, and prednisone), and bendamustine.
At a median observation time of 41.1 months, the overall response rate was 91% in the obinutuzumab arm and 88% in the rituximab arm. The complete response rates were 28% and 27%, respectively.
The median progression-free survival was not reached in either arm. The hazard ratio, for obinutuzumab compared to rituximab, was 0.72 (95% CI, 0.56-0.93, P=0.0118).
The estimated 3-year progression-free survival was 78.9% in the rituximab arm and 83.4% in the obinutuzumab arm.
Safety was evaluated based on all 1385 patients in the study, 86% of whom had FL and 14% of whom had marginal zone lymphoma.
Serious adverse events (AEs) occurred in 50% of patients in the obinutuzumab arm and 43% in the rituximab arm. Fatal AEs occurred in 5% and 4%, respectively. Infections and second malignancies were the leading causes of these deaths.
During the monotherapy period, the most common AEs (≥ 5%) in patients treated with obinutuzumab were cough (21%), neutropenia (19%), upper respiratory tract infection (15%), viral upper respiratory tract infection (15%), diarrhea (13%), arthralgia (10%), fatigue (9%), sinusitis (9%), infusion reactions (8%), pneumonia (8%), herpes zoster (8%), lower respiratory tract infection (7%), pyrexia (7%), back pain (6%), headache (6%), urinary tract infection (6%), nausea (6%), bronchitis (5%), and vomiting (5%).
Grade 3-4 AEs (≥1%) in patients treated with obinutuzumab included neutropenia (17%), pneumonia (3%), and febrile neutropenia (2%). There were 2 deaths due to pneumonia in the obinutuzumab arm.
Health Canada has expanded the approved use of obinutuzumab (Gazyva®).
The anti-CD20 monoclonal antibody is now approved for use in combination with chemotherapy to treat patients with previously untreated follicular lymphoma (FL) that is advanced (stage II bulky, stage III, or stage IV).
In patients who respond to this treatment, obinutuzumab monotherapy can be given as maintenance.
Health Canada previously approved obinutuzumab for the following indications:
- In combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia
- First in combination with bendamustine, then as monotherapy, in FL patients who relapsed after or are refractory to a rituximab-containing regimen.
Phase 3 results
Health Canada’s latest approval of obinutuzumab is based on results from the phase 3 GALLIUM study, which were published in NEJM in October 2017. The following are updated data from the product monograph.
GALLIUM included 1385 patients with previously untreated non-Hodgkin lymphoma, and 1202 of these patients had previously untreated, advanced FL.
Half of the FL patients (n=601) were randomized to receive obinutuzumab plus chemotherapy (followed by obinutuzumab maintenance for up to 2 years), and half were randomized to rituximab plus chemotherapy (followed by rituximab maintenance for up to 2 years).
The different chemotherapies used were CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), CVP (cyclophosphamide, vincristine, and prednisone), and bendamustine.
At a median observation time of 41.1 months, the overall response rate was 91% in the obinutuzumab arm and 88% in the rituximab arm. The complete response rates were 28% and 27%, respectively.
The median progression-free survival was not reached in either arm. The hazard ratio, for obinutuzumab compared to rituximab, was 0.72 (95% CI, 0.56-0.93, P=0.0118).
The estimated 3-year progression-free survival was 78.9% in the rituximab arm and 83.4% in the obinutuzumab arm.
Safety was evaluated based on all 1385 patients in the study, 86% of whom had FL and 14% of whom had marginal zone lymphoma.
Serious adverse events (AEs) occurred in 50% of patients in the obinutuzumab arm and 43% in the rituximab arm. Fatal AEs occurred in 5% and 4%, respectively. Infections and second malignancies were the leading causes of these deaths.
During the monotherapy period, the most common AEs (≥ 5%) in patients treated with obinutuzumab were cough (21%), neutropenia (19%), upper respiratory tract infection (15%), viral upper respiratory tract infection (15%), diarrhea (13%), arthralgia (10%), fatigue (9%), sinusitis (9%), infusion reactions (8%), pneumonia (8%), herpes zoster (8%), lower respiratory tract infection (7%), pyrexia (7%), back pain (6%), headache (6%), urinary tract infection (6%), nausea (6%), bronchitis (5%), and vomiting (5%).
Grade 3-4 AEs (≥1%) in patients treated with obinutuzumab included neutropenia (17%), pneumonia (3%), and febrile neutropenia (2%). There were 2 deaths due to pneumonia in the obinutuzumab arm.
ESMO, ASCO seek improved cancer services
The European Society for Medical Oncology (ESMO) and the American Society of Clinical Oncology (ASCO) have called for renewed political commitment to improve cancer services and reduce cancer deaths.
ASCO and ESMO issued a joint statement in which they asked heads of state and health ministers to attend the United Nations Civil Society Hearing on Non-communicable Diseases (NCDs) in September and reconfirm their commitment to “pass legislation and invest in actions that will reduce the burden of NCDs, including cancer.”
Specifically, ESMO and ASCO said governments should:
- Implement the 2017 World Health Assembly Cancer Resolution
- Develop and strengthen educational programs that provide lifestyle recommendations to reduce cancer risk (eg, prevent tobacco use, encourage healthy weight control, etc.)
- Develop efficient and cost-effective primary prevention measures (eg, Helicobacter pylori eradication)
- Ensure timely access to screening, early stage diagnosis, and treatment for all stages of cancer
- Strengthen health systems so they can provide cancer services to all who need them
- Provide essential secondary healthcare services that ensure an adequate number of well-trained oncology professionals who have access to necessary resources
- Aim to reduce premature mortality by 25% by 2025 and by 33% by 2030 across all NCDs.
“Recent UN and WHO reports1,2,3,4 note that, unless countries significantly scale-up their actions and investments, they will not meet agreed targets to reduce deaths from non-communicable diseases,” said ESMO President Josep Tabernero, MD, PhD.
“We are concerned that governments may find it easier to achieve their targets by reducing deaths from only some NCDs, leaving cancer patients behind. We believe there are cost-effective ways to improve cancer care and stand ready to assist countries in doing this by providing our expertise in cancer management to support implementation of the 2017 World Health Assembly Cancer Resolution.”
“We urge member states to consider our joint call and amendments to strengthen the political declaration to be approved during the UN high-level meeting on 27 September and thus change the future outlook for cancer patients worldwide.”
1. United Nations Report by the Secretary General, Document A_72_662, 21 December 2017: http://www.who.int/ncds/governance/high-level-commission/A_72_662.pdf
2. World Health Assembly Report by the WHO Director General, Document WHA 71.2, 26 May 2018: http://apps.who.int/gb/ebwha/pdf_files/WHA71/A71_R2-en.pdf
3. WHO Independent High-Level Commission on NCDs Report, Time to Deliver, 1 June 2018: http://apps.who.int/iris/bitstream/handle/10665/272710/9789241514163-eng.pdf?ua=1
4. WHO Report Saving Lives, Spending Less, 21 May 2018: http://apps.who.int/iris/bitstream/handle/10665/272534/WHO-NMH-NVI-18.8-eng.pdf
The European Society for Medical Oncology (ESMO) and the American Society of Clinical Oncology (ASCO) have called for renewed political commitment to improve cancer services and reduce cancer deaths.
ASCO and ESMO issued a joint statement in which they asked heads of state and health ministers to attend the United Nations Civil Society Hearing on Non-communicable Diseases (NCDs) in September and reconfirm their commitment to “pass legislation and invest in actions that will reduce the burden of NCDs, including cancer.”
Specifically, ESMO and ASCO said governments should:
- Implement the 2017 World Health Assembly Cancer Resolution
- Develop and strengthen educational programs that provide lifestyle recommendations to reduce cancer risk (eg, prevent tobacco use, encourage healthy weight control, etc.)
- Develop efficient and cost-effective primary prevention measures (eg, Helicobacter pylori eradication)
- Ensure timely access to screening, early stage diagnosis, and treatment for all stages of cancer
- Strengthen health systems so they can provide cancer services to all who need them
- Provide essential secondary healthcare services that ensure an adequate number of well-trained oncology professionals who have access to necessary resources
- Aim to reduce premature mortality by 25% by 2025 and by 33% by 2030 across all NCDs.
“Recent UN and WHO reports1,2,3,4 note that, unless countries significantly scale-up their actions and investments, they will not meet agreed targets to reduce deaths from non-communicable diseases,” said ESMO President Josep Tabernero, MD, PhD.
“We are concerned that governments may find it easier to achieve their targets by reducing deaths from only some NCDs, leaving cancer patients behind. We believe there are cost-effective ways to improve cancer care and stand ready to assist countries in doing this by providing our expertise in cancer management to support implementation of the 2017 World Health Assembly Cancer Resolution.”
“We urge member states to consider our joint call and amendments to strengthen the political declaration to be approved during the UN high-level meeting on 27 September and thus change the future outlook for cancer patients worldwide.”
1. United Nations Report by the Secretary General, Document A_72_662, 21 December 2017: http://www.who.int/ncds/governance/high-level-commission/A_72_662.pdf
2. World Health Assembly Report by the WHO Director General, Document WHA 71.2, 26 May 2018: http://apps.who.int/gb/ebwha/pdf_files/WHA71/A71_R2-en.pdf
3. WHO Independent High-Level Commission on NCDs Report, Time to Deliver, 1 June 2018: http://apps.who.int/iris/bitstream/handle/10665/272710/9789241514163-eng.pdf?ua=1
4. WHO Report Saving Lives, Spending Less, 21 May 2018: http://apps.who.int/iris/bitstream/handle/10665/272534/WHO-NMH-NVI-18.8-eng.pdf
The European Society for Medical Oncology (ESMO) and the American Society of Clinical Oncology (ASCO) have called for renewed political commitment to improve cancer services and reduce cancer deaths.
ASCO and ESMO issued a joint statement in which they asked heads of state and health ministers to attend the United Nations Civil Society Hearing on Non-communicable Diseases (NCDs) in September and reconfirm their commitment to “pass legislation and invest in actions that will reduce the burden of NCDs, including cancer.”
Specifically, ESMO and ASCO said governments should:
- Implement the 2017 World Health Assembly Cancer Resolution
- Develop and strengthen educational programs that provide lifestyle recommendations to reduce cancer risk (eg, prevent tobacco use, encourage healthy weight control, etc.)
- Develop efficient and cost-effective primary prevention measures (eg, Helicobacter pylori eradication)
- Ensure timely access to screening, early stage diagnosis, and treatment for all stages of cancer
- Strengthen health systems so they can provide cancer services to all who need them
- Provide essential secondary healthcare services that ensure an adequate number of well-trained oncology professionals who have access to necessary resources
- Aim to reduce premature mortality by 25% by 2025 and by 33% by 2030 across all NCDs.
“Recent UN and WHO reports1,2,3,4 note that, unless countries significantly scale-up their actions and investments, they will not meet agreed targets to reduce deaths from non-communicable diseases,” said ESMO President Josep Tabernero, MD, PhD.
“We are concerned that governments may find it easier to achieve their targets by reducing deaths from only some NCDs, leaving cancer patients behind. We believe there are cost-effective ways to improve cancer care and stand ready to assist countries in doing this by providing our expertise in cancer management to support implementation of the 2017 World Health Assembly Cancer Resolution.”
“We urge member states to consider our joint call and amendments to strengthen the political declaration to be approved during the UN high-level meeting on 27 September and thus change the future outlook for cancer patients worldwide.”
1. United Nations Report by the Secretary General, Document A_72_662, 21 December 2017: http://www.who.int/ncds/governance/high-level-commission/A_72_662.pdf
2. World Health Assembly Report by the WHO Director General, Document WHA 71.2, 26 May 2018: http://apps.who.int/gb/ebwha/pdf_files/WHA71/A71_R2-en.pdf
3. WHO Independent High-Level Commission on NCDs Report, Time to Deliver, 1 June 2018: http://apps.who.int/iris/bitstream/handle/10665/272710/9789241514163-eng.pdf?ua=1
4. WHO Report Saving Lives, Spending Less, 21 May 2018: http://apps.who.int/iris/bitstream/handle/10665/272534/WHO-NMH-NVI-18.8-eng.pdf
PET-driven chemo strategy helps reduce toxicity in Hodgkin lymphoma
CHICAGO – Positron emission tomography (PET) performed after two cycles of BEACOPP could help identify a subset of advanced-stage Hodgkin lymphoma patients who can receive de-escalated treatment without compromising disease control, results of a phase 3 randomized trial show.
Five-year progression-free survival (PFS) exceeded 85% not only for patients receiving six cycles of escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) but also for patients who were de-escalated to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) chemotherapy based on negative PET results, according to the final analysis of the AHL2011 LYSA study, presented at the annual meeting of the American Society of Clinical Oncology.
“This approach allows us to significantly reduce the treatment-related toxicity in most patients, and provides similar patient outcomes compared to standard BEACOPP escalated treatment,” said Olivier Casasnovas, MD, of the University Hospital Le Bocage and Inserm, Dijon, France.
Previous studies have shown that BEACOPP may improve PFS, compared with ABVD, but is more toxic and associated with a higher risk of infertility and myelodysplasia or acute leukemia. Dr. Casasnovas and his colleagues sought to evaluate whether upfront BEACOPP followed by de-escalation to ABVD, when warranted by negative PET results, would improve outcomes without compromising efficacy.
The AHL2011 LYSA study included 823 patients (median age, 30 years; 63% male) with previously untreated advanced classical Hodgkin lymphoma. All patients received PET at baseline, after cycle two of chemotherapy, and again after cycle four.
Patients were randomized either to six cycles of escalated BEACOPP, or to an experimental arm in which patients started with BEACOPP but were de-escalated to ABVD if PET results were negative after two or four cycles of treatment.
PET results after cycle two were negative for 87% of patients in the experimental arm, so on an intent-to-treat basis, 84% of them received two cycles of BEACOPP and four cycles of ABVD, Dr. Casasnovas reported.
PFS, with a median follow-up of 50.4 months, was not significantly different for the standard versus the experimental arm (hazard ratio, 1.084; 95% confidence interval, 0.73-1.59; P = .68). Five-year PFS was 85.7% in the experimental treatment de-escalation arm, compared with 86.2% in the standard arm.
Overall survival was likewise similar between arms, with 5-year overall survival exceeding 95% in both groups, Dr. Casasnovas said.
Although there was no significant difference overall in the incidence of adverse events, there was significantly less anemia, febrile neutropenia, thrombocytopenia, and sepsis in the PET-driven de-escalation arm. Overall, 27% of patients in the standard chemotherapy arm experienced at least one serious adverse event, compared with 17% in the PET-driven arm (P less than .002).
The incidence of second primary malignancies was numerically lower in the experimental arm (1.2% vs. 2.4%), though that finding did not reach statistical significance.
On multivariate analysis, interim PET positivity after cycle two was associated with increased risk of disease progression for patients in this study (HR, 3.316). Risk of progression was even higher for patients with PET positivity after four cycles (HR, 12.968), identifying a subset of patients with “particularly poor outcome” who could benefit from development of new treatment options, Dr. Casasnovas said.
Dr. Casasnovas reported financial ties to AbbVie, Bristol-Myers Squibb, Celgene, Gilead Sciences, Janssen, Merck, Roche/Genentech, Sanofi, and Takeda.
SOURCE: Casasnovas O et al. ASCO 2018, Abstract 7503.
CHICAGO – Positron emission tomography (PET) performed after two cycles of BEACOPP could help identify a subset of advanced-stage Hodgkin lymphoma patients who can receive de-escalated treatment without compromising disease control, results of a phase 3 randomized trial show.
Five-year progression-free survival (PFS) exceeded 85% not only for patients receiving six cycles of escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) but also for patients who were de-escalated to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) chemotherapy based on negative PET results, according to the final analysis of the AHL2011 LYSA study, presented at the annual meeting of the American Society of Clinical Oncology.
“This approach allows us to significantly reduce the treatment-related toxicity in most patients, and provides similar patient outcomes compared to standard BEACOPP escalated treatment,” said Olivier Casasnovas, MD, of the University Hospital Le Bocage and Inserm, Dijon, France.
Previous studies have shown that BEACOPP may improve PFS, compared with ABVD, but is more toxic and associated with a higher risk of infertility and myelodysplasia or acute leukemia. Dr. Casasnovas and his colleagues sought to evaluate whether upfront BEACOPP followed by de-escalation to ABVD, when warranted by negative PET results, would improve outcomes without compromising efficacy.
The AHL2011 LYSA study included 823 patients (median age, 30 years; 63% male) with previously untreated advanced classical Hodgkin lymphoma. All patients received PET at baseline, after cycle two of chemotherapy, and again after cycle four.
Patients were randomized either to six cycles of escalated BEACOPP, or to an experimental arm in which patients started with BEACOPP but were de-escalated to ABVD if PET results were negative after two or four cycles of treatment.
PET results after cycle two were negative for 87% of patients in the experimental arm, so on an intent-to-treat basis, 84% of them received two cycles of BEACOPP and four cycles of ABVD, Dr. Casasnovas reported.
PFS, with a median follow-up of 50.4 months, was not significantly different for the standard versus the experimental arm (hazard ratio, 1.084; 95% confidence interval, 0.73-1.59; P = .68). Five-year PFS was 85.7% in the experimental treatment de-escalation arm, compared with 86.2% in the standard arm.
Overall survival was likewise similar between arms, with 5-year overall survival exceeding 95% in both groups, Dr. Casasnovas said.
Although there was no significant difference overall in the incidence of adverse events, there was significantly less anemia, febrile neutropenia, thrombocytopenia, and sepsis in the PET-driven de-escalation arm. Overall, 27% of patients in the standard chemotherapy arm experienced at least one serious adverse event, compared with 17% in the PET-driven arm (P less than .002).
The incidence of second primary malignancies was numerically lower in the experimental arm (1.2% vs. 2.4%), though that finding did not reach statistical significance.
On multivariate analysis, interim PET positivity after cycle two was associated with increased risk of disease progression for patients in this study (HR, 3.316). Risk of progression was even higher for patients with PET positivity after four cycles (HR, 12.968), identifying a subset of patients with “particularly poor outcome” who could benefit from development of new treatment options, Dr. Casasnovas said.
Dr. Casasnovas reported financial ties to AbbVie, Bristol-Myers Squibb, Celgene, Gilead Sciences, Janssen, Merck, Roche/Genentech, Sanofi, and Takeda.
SOURCE: Casasnovas O et al. ASCO 2018, Abstract 7503.
CHICAGO – Positron emission tomography (PET) performed after two cycles of BEACOPP could help identify a subset of advanced-stage Hodgkin lymphoma patients who can receive de-escalated treatment without compromising disease control, results of a phase 3 randomized trial show.
Five-year progression-free survival (PFS) exceeded 85% not only for patients receiving six cycles of escalated BEACOPP (bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone) but also for patients who were de-escalated to ABVD (doxorubicin, bleomycin, vinblastine, dacarbazine) chemotherapy based on negative PET results, according to the final analysis of the AHL2011 LYSA study, presented at the annual meeting of the American Society of Clinical Oncology.
“This approach allows us to significantly reduce the treatment-related toxicity in most patients, and provides similar patient outcomes compared to standard BEACOPP escalated treatment,” said Olivier Casasnovas, MD, of the University Hospital Le Bocage and Inserm, Dijon, France.
Previous studies have shown that BEACOPP may improve PFS, compared with ABVD, but is more toxic and associated with a higher risk of infertility and myelodysplasia or acute leukemia. Dr. Casasnovas and his colleagues sought to evaluate whether upfront BEACOPP followed by de-escalation to ABVD, when warranted by negative PET results, would improve outcomes without compromising efficacy.
The AHL2011 LYSA study included 823 patients (median age, 30 years; 63% male) with previously untreated advanced classical Hodgkin lymphoma. All patients received PET at baseline, after cycle two of chemotherapy, and again after cycle four.
Patients were randomized either to six cycles of escalated BEACOPP, or to an experimental arm in which patients started with BEACOPP but were de-escalated to ABVD if PET results were negative after two or four cycles of treatment.
PET results after cycle two were negative for 87% of patients in the experimental arm, so on an intent-to-treat basis, 84% of them received two cycles of BEACOPP and four cycles of ABVD, Dr. Casasnovas reported.
PFS, with a median follow-up of 50.4 months, was not significantly different for the standard versus the experimental arm (hazard ratio, 1.084; 95% confidence interval, 0.73-1.59; P = .68). Five-year PFS was 85.7% in the experimental treatment de-escalation arm, compared with 86.2% in the standard arm.
Overall survival was likewise similar between arms, with 5-year overall survival exceeding 95% in both groups, Dr. Casasnovas said.
Although there was no significant difference overall in the incidence of adverse events, there was significantly less anemia, febrile neutropenia, thrombocytopenia, and sepsis in the PET-driven de-escalation arm. Overall, 27% of patients in the standard chemotherapy arm experienced at least one serious adverse event, compared with 17% in the PET-driven arm (P less than .002).
The incidence of second primary malignancies was numerically lower in the experimental arm (1.2% vs. 2.4%), though that finding did not reach statistical significance.
On multivariate analysis, interim PET positivity after cycle two was associated with increased risk of disease progression for patients in this study (HR, 3.316). Risk of progression was even higher for patients with PET positivity after four cycles (HR, 12.968), identifying a subset of patients with “particularly poor outcome” who could benefit from development of new treatment options, Dr. Casasnovas said.
Dr. Casasnovas reported financial ties to AbbVie, Bristol-Myers Squibb, Celgene, Gilead Sciences, Janssen, Merck, Roche/Genentech, Sanofi, and Takeda.
SOURCE: Casasnovas O et al. ASCO 2018, Abstract 7503.
REPORTING FROM ASCO 2018
Key clinical point:
Major finding: With a median follow-up of 50.4 months, PFS was not significantly different for escalated BEACOPP vs. the experimental arm (HR, 1.084; 95% CI, 0.73-1.59; P = .68).
Study details: Final analysis of AHL2011 LYSA, a randomized phase 3 study including 823 patients with advanced-stage Hodgkin lymphoma.
Disclosures: Dr. Casasnovas reported financial ties to AbbVie, Bristol-Myers Squibb, Celgene, Gilead Sciences, Janssen, Merck, Roche/Genentech, Sanofi, and Takeda.
Source: Casasnovas O et al. ASCO 2018, Abstract 7503.