User login
Gene plays crucial role in cancer development, team says
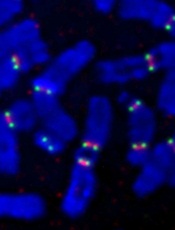
Credit: Beth A. Sullivan
New research suggests DNA ligase 3 is crucial for the evolutionary processes that drive cancer.
“We have identified a gene that, as cells age, seems to regulate whether the cells become cancerous or not,” said Eric A. Hendrickson, PhD, of the University of Minnesota in Minneapolis.
“This gene has never been identified before in this role, so this makes it a potentially very important therapeutic target.”
Dr Hendrickson and his colleagues recounted this discovery in Cell Reports.
The researchers noted that short, dysfunctional telomeres can fuse, thereby generating dicentric chromosomes and initiating breakage-fusion-bridge cycles. The cells that manage to escape the subsequent crisis have genomic rearrangements that drive clonal evolution and malignant progression.
The team wanted to determine exactly what allows these malignant cells to escape telomere-driven crisis and avoid death.
To find out, the group disabled certain genes in human cells and then studied the impact this had on telomere fusion.
They found that cells escaped death when ligase 3 was active but not when its action, which appears to promote fusion within like chromosomes rather than between different chromosomes, was inhibited.
“Telomere dysfunction has been identified in many human cancers,” said study author Duncan Baird, PhD, of Cardiff University in the UK.
“And, as we have shown previously, short telomeres can predict the outcome of patients with [chronic lymphocytic leukemia] and probably many other tumor types. Thus, the discovery that ligase 3 is required for this process is fundamentally important.”
This research was made possible by a chance meeting between Dr Baird and Dr Hendrickson at an international conference. The pair discovered they were both looking at the role of ligase 3 in cancer and decided to collaborate.
“The collaboration paid off, as we were able to uncover something that neither one of us could have done on our own,” Dr Hendrickson said.
Additional studies are already underway. The researchers are investigating the discovery that the reliance on ligase 3 appears to be dependent upon the activity of another key DNA repair gene, p53.
“Since p53 is the most commonly mutated gene in human cancer, it now behooves us to discover how these two genes are interacting and to see if we can’t use that information to develop synergistic treatment modalities,” Dr Hendrickson concluded. ![]()

Credit: Beth A. Sullivan
New research suggests DNA ligase 3 is crucial for the evolutionary processes that drive cancer.
“We have identified a gene that, as cells age, seems to regulate whether the cells become cancerous or not,” said Eric A. Hendrickson, PhD, of the University of Minnesota in Minneapolis.
“This gene has never been identified before in this role, so this makes it a potentially very important therapeutic target.”
Dr Hendrickson and his colleagues recounted this discovery in Cell Reports.
The researchers noted that short, dysfunctional telomeres can fuse, thereby generating dicentric chromosomes and initiating breakage-fusion-bridge cycles. The cells that manage to escape the subsequent crisis have genomic rearrangements that drive clonal evolution and malignant progression.
The team wanted to determine exactly what allows these malignant cells to escape telomere-driven crisis and avoid death.
To find out, the group disabled certain genes in human cells and then studied the impact this had on telomere fusion.
They found that cells escaped death when ligase 3 was active but not when its action, which appears to promote fusion within like chromosomes rather than between different chromosomes, was inhibited.
“Telomere dysfunction has been identified in many human cancers,” said study author Duncan Baird, PhD, of Cardiff University in the UK.
“And, as we have shown previously, short telomeres can predict the outcome of patients with [chronic lymphocytic leukemia] and probably many other tumor types. Thus, the discovery that ligase 3 is required for this process is fundamentally important.”
This research was made possible by a chance meeting between Dr Baird and Dr Hendrickson at an international conference. The pair discovered they were both looking at the role of ligase 3 in cancer and decided to collaborate.
“The collaboration paid off, as we were able to uncover something that neither one of us could have done on our own,” Dr Hendrickson said.
Additional studies are already underway. The researchers are investigating the discovery that the reliance on ligase 3 appears to be dependent upon the activity of another key DNA repair gene, p53.
“Since p53 is the most commonly mutated gene in human cancer, it now behooves us to discover how these two genes are interacting and to see if we can’t use that information to develop synergistic treatment modalities,” Dr Hendrickson concluded. ![]()

Credit: Beth A. Sullivan
New research suggests DNA ligase 3 is crucial for the evolutionary processes that drive cancer.
“We have identified a gene that, as cells age, seems to regulate whether the cells become cancerous or not,” said Eric A. Hendrickson, PhD, of the University of Minnesota in Minneapolis.
“This gene has never been identified before in this role, so this makes it a potentially very important therapeutic target.”
Dr Hendrickson and his colleagues recounted this discovery in Cell Reports.
The researchers noted that short, dysfunctional telomeres can fuse, thereby generating dicentric chromosomes and initiating breakage-fusion-bridge cycles. The cells that manage to escape the subsequent crisis have genomic rearrangements that drive clonal evolution and malignant progression.
The team wanted to determine exactly what allows these malignant cells to escape telomere-driven crisis and avoid death.
To find out, the group disabled certain genes in human cells and then studied the impact this had on telomere fusion.
They found that cells escaped death when ligase 3 was active but not when its action, which appears to promote fusion within like chromosomes rather than between different chromosomes, was inhibited.
“Telomere dysfunction has been identified in many human cancers,” said study author Duncan Baird, PhD, of Cardiff University in the UK.
“And, as we have shown previously, short telomeres can predict the outcome of patients with [chronic lymphocytic leukemia] and probably many other tumor types. Thus, the discovery that ligase 3 is required for this process is fundamentally important.”
This research was made possible by a chance meeting between Dr Baird and Dr Hendrickson at an international conference. The pair discovered they were both looking at the role of ligase 3 in cancer and decided to collaborate.
“The collaboration paid off, as we were able to uncover something that neither one of us could have done on our own,” Dr Hendrickson said.
Additional studies are already underway. The researchers are investigating the discovery that the reliance on ligase 3 appears to be dependent upon the activity of another key DNA repair gene, p53.
“Since p53 is the most commonly mutated gene in human cancer, it now behooves us to discover how these two genes are interacting and to see if we can’t use that information to develop synergistic treatment modalities,” Dr Hendrickson concluded. ![]()
Ofatumumab prompts fatal reaction in CLL patient

Credit: Bill Branson
Health Canada and GlaxoSmithKline (GSK) have reported a fatal infusion reaction in a patient receiving the monoclonal antibody ofatumumab (Arzerra) to treat
chronic lymphocytic leukemia (CLL).
The patient had no known history of cardiac disease.
Ofatumumab’s product monograph is being updated to include a warning about the potential for fatal infusion reactions.
In Canada, ofatumumab is approved to treat patients with CLL that is refractory to fludarabine and alemtuzumab.
The drug received this marketing authorization with conditions, pending the results of trials to verify its clinical benefit.
In light of the fatal infusion reaction, GSK and Health Canada are reminding healthcare professionals that ofatumumab should be administered under the supervision of a physician experienced in the use of cancer therapy. The drug should be given in an environment where facilities to adequately monitor and treat infusion reactions are available.
Prior to infusion, patients should always receive the appropriate premedication, as outlined in the product label. However, serious infusion reactions may occur despite premedication.
If you suspect a severe infusion reaction, stop the infusion immediately and provide symptomatic treatment. Signs and symptoms of an infusion reaction may include swelling of the face or mouth, fever, chills, difficulty breathing, tightness of the chest and/or throat, light headedness, nausea, diarrhea, and rash.
These symptoms can occur during or shortly after the infusion, predominantly with the first 2 infusions. So ensure patients are closely monitored, especially those with heart conditions. And inform patients about the risk of fatal infusion reactions associated with ofatumumab.
GSK has sent a letter to healthcare professionals detailing the risk of fatal infusion reactions. The information is also available on the Canadian website of GSK and the Health Canada website.
Any case of serious hypersensitivity, infusion reactions, or other serious or unexpected side effects in patients receiving ofatumumab should be reported to GSK or Health Canada.
Ofatumumab is also known to pose a risk of hepatitis B virus reactivation, progressive multifocal leukoencephalopathy, serious and/or fatal cardiovascular events, and serious and/or fatal infections (bacterial, fungal, and viral).
Ofatumumab recently received approval in the European Union to be used in combination with chlorambucil or bendamustine for untreated CLL patients who are not eligible for fludarabine-based therapy. The drug previously received conditional approval in Europe as monotherapy to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Ofatumumab is approved for both of these indications in the US as well. ![]()

Credit: Bill Branson
Health Canada and GlaxoSmithKline (GSK) have reported a fatal infusion reaction in a patient receiving the monoclonal antibody ofatumumab (Arzerra) to treat
chronic lymphocytic leukemia (CLL).
The patient had no known history of cardiac disease.
Ofatumumab’s product monograph is being updated to include a warning about the potential for fatal infusion reactions.
In Canada, ofatumumab is approved to treat patients with CLL that is refractory to fludarabine and alemtuzumab.
The drug received this marketing authorization with conditions, pending the results of trials to verify its clinical benefit.
In light of the fatal infusion reaction, GSK and Health Canada are reminding healthcare professionals that ofatumumab should be administered under the supervision of a physician experienced in the use of cancer therapy. The drug should be given in an environment where facilities to adequately monitor and treat infusion reactions are available.
Prior to infusion, patients should always receive the appropriate premedication, as outlined in the product label. However, serious infusion reactions may occur despite premedication.
If you suspect a severe infusion reaction, stop the infusion immediately and provide symptomatic treatment. Signs and symptoms of an infusion reaction may include swelling of the face or mouth, fever, chills, difficulty breathing, tightness of the chest and/or throat, light headedness, nausea, diarrhea, and rash.
These symptoms can occur during or shortly after the infusion, predominantly with the first 2 infusions. So ensure patients are closely monitored, especially those with heart conditions. And inform patients about the risk of fatal infusion reactions associated with ofatumumab.
GSK has sent a letter to healthcare professionals detailing the risk of fatal infusion reactions. The information is also available on the Canadian website of GSK and the Health Canada website.
Any case of serious hypersensitivity, infusion reactions, or other serious or unexpected side effects in patients receiving ofatumumab should be reported to GSK or Health Canada.
Ofatumumab is also known to pose a risk of hepatitis B virus reactivation, progressive multifocal leukoencephalopathy, serious and/or fatal cardiovascular events, and serious and/or fatal infections (bacterial, fungal, and viral).
Ofatumumab recently received approval in the European Union to be used in combination with chlorambucil or bendamustine for untreated CLL patients who are not eligible for fludarabine-based therapy. The drug previously received conditional approval in Europe as monotherapy to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Ofatumumab is approved for both of these indications in the US as well. ![]()

Credit: Bill Branson
Health Canada and GlaxoSmithKline (GSK) have reported a fatal infusion reaction in a patient receiving the monoclonal antibody ofatumumab (Arzerra) to treat
chronic lymphocytic leukemia (CLL).
The patient had no known history of cardiac disease.
Ofatumumab’s product monograph is being updated to include a warning about the potential for fatal infusion reactions.
In Canada, ofatumumab is approved to treat patients with CLL that is refractory to fludarabine and alemtuzumab.
The drug received this marketing authorization with conditions, pending the results of trials to verify its clinical benefit.
In light of the fatal infusion reaction, GSK and Health Canada are reminding healthcare professionals that ofatumumab should be administered under the supervision of a physician experienced in the use of cancer therapy. The drug should be given in an environment where facilities to adequately monitor and treat infusion reactions are available.
Prior to infusion, patients should always receive the appropriate premedication, as outlined in the product label. However, serious infusion reactions may occur despite premedication.
If you suspect a severe infusion reaction, stop the infusion immediately and provide symptomatic treatment. Signs and symptoms of an infusion reaction may include swelling of the face or mouth, fever, chills, difficulty breathing, tightness of the chest and/or throat, light headedness, nausea, diarrhea, and rash.
These symptoms can occur during or shortly after the infusion, predominantly with the first 2 infusions. So ensure patients are closely monitored, especially those with heart conditions. And inform patients about the risk of fatal infusion reactions associated with ofatumumab.
GSK has sent a letter to healthcare professionals detailing the risk of fatal infusion reactions. The information is also available on the Canadian website of GSK and the Health Canada website.
Any case of serious hypersensitivity, infusion reactions, or other serious or unexpected side effects in patients receiving ofatumumab should be reported to GSK or Health Canada.
Ofatumumab is also known to pose a risk of hepatitis B virus reactivation, progressive multifocal leukoencephalopathy, serious and/or fatal cardiovascular events, and serious and/or fatal infections (bacterial, fungal, and viral).
Ofatumumab recently received approval in the European Union to be used in combination with chlorambucil or bendamustine for untreated CLL patients who are not eligible for fludarabine-based therapy. The drug previously received conditional approval in Europe as monotherapy to treat CLL patients who are refractory to fludarabine and alemtuzumab.
Ofatumumab is approved for both of these indications in the US as well. ![]()
CHMP recommends antifungal agent

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for intravenous (IV) posaconazole (Noxafil), an antifungal agent.
If the European Commission affirms the CHMP opinion, IV posaconazole will be authorized for use in the European Union, Iceland, Liechtenstein, and Norway.
The commission previously granted marketing authorization for posaconazole delayed-release tablets and oral suspension.
Posaconazole is used to prevent invasive fungal infections in severely immunocompromised patients, such as hematopoietic stem cell transplant recipients with graft-vs-host disease or patients with hematologic malignancies and prolonged neutropenia from chemotherapy.
The drug is also used to treat fungal diseases—invasive aspergillosis, fusariosis, chromoblastomycosis, mycetoma, and coccidioidomycosis—when other antifungal agents—amphotericin B, itraconazole, or fluconazole—cannot be tolerated or have failed.
And posaconazole oral suspension is used as a first-line treatment for thrush, a fungal infection of the mouth and throat due to Candida.
Posaconazole injection is administered with a loading dose of 300 mg twice a day on the first day of therapy, then 300 mg once a day thereafter. It is given through a central venous line by IV infusion over approximately 90 minutes.
Once combined with a mixture of IV solution (150 mL of 5% dextrose in water or sodium chloride 0.9%), posaconazole should be administered immediately. If not used immediately, the solution can be stored up to 24 hours if refrigerated at 2°-8° C (36°-46° F).
The safety and effectiveness of IV posaconazole in patients younger than 18 years has not been established. IV posaconazole should not be used in pediatric patients because of non-clinical safety concerns.
Co-administration of drugs that can decrease the plasma concentration of posaconazole should be avoided unless the benefit outweighs the risk. If such drugs are necessary, patients should be monitored closely for breakthrough fungal infections.
Patients with known hypersensitivity to posaconazole or other azole antifungal medicines should not receive posaconazole. The drug should not be given with sirolimus, pimozide, quinidine, atorvastatin, lovastatin, simvastatin, or ergot alkaloids.
Drugs such as cyclosporine and tacrolimus require dose adjustments and frequent blood monitoring when administered with posaconazole. Serious side effects, including nephrotoxicity, leukoencephalopathy, and death, have been reported in patients with increased cyclosporine or tacrolimus blood levels.
Healthcare professionals should use caution when administering posaconazole to patients at risk of developing an irregular heart rhythm, as the drug has been shown to prolong the QT interval, and cases of potentially fatal irregular heart rhythm (torsades de pointes) have been reported in patients taking posaconazole.
Hepatic reactions have been reported as well. This includes mild to moderate elevations in ALT, AST, alkaline phosphatase, total bilirubin, and/or clinical hepatitis. Monitoring or discontinuation may be necessary in patients with hepatic reactions to posaconazole.
IV posaconazole should be avoided in patients with moderate or severe renal impairment (estimated glomerular filtration rate <50 mL/min), unless an assessment of the benefit/risk to the patient justifies the use of posaconazole.
In clinical trials, the adverse events associated with IV posaconazole were generally similar to those in trials of posaconazole oral suspension. The most frequently reported events were diarrhea (32%), hypokalemia (22%), fever (21%), and nausea (19%).
IV posaconazole is under development by MSD (known as Merck in the US and Canada). ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for intravenous (IV) posaconazole (Noxafil), an antifungal agent.
If the European Commission affirms the CHMP opinion, IV posaconazole will be authorized for use in the European Union, Iceland, Liechtenstein, and Norway.
The commission previously granted marketing authorization for posaconazole delayed-release tablets and oral suspension.
Posaconazole is used to prevent invasive fungal infections in severely immunocompromised patients, such as hematopoietic stem cell transplant recipients with graft-vs-host disease or patients with hematologic malignancies and prolonged neutropenia from chemotherapy.
The drug is also used to treat fungal diseases—invasive aspergillosis, fusariosis, chromoblastomycosis, mycetoma, and coccidioidomycosis—when other antifungal agents—amphotericin B, itraconazole, or fluconazole—cannot be tolerated or have failed.
And posaconazole oral suspension is used as a first-line treatment for thrush, a fungal infection of the mouth and throat due to Candida.
Posaconazole injection is administered with a loading dose of 300 mg twice a day on the first day of therapy, then 300 mg once a day thereafter. It is given through a central venous line by IV infusion over approximately 90 minutes.
Once combined with a mixture of IV solution (150 mL of 5% dextrose in water or sodium chloride 0.9%), posaconazole should be administered immediately. If not used immediately, the solution can be stored up to 24 hours if refrigerated at 2°-8° C (36°-46° F).
The safety and effectiveness of IV posaconazole in patients younger than 18 years has not been established. IV posaconazole should not be used in pediatric patients because of non-clinical safety concerns.
Co-administration of drugs that can decrease the plasma concentration of posaconazole should be avoided unless the benefit outweighs the risk. If such drugs are necessary, patients should be monitored closely for breakthrough fungal infections.
Patients with known hypersensitivity to posaconazole or other azole antifungal medicines should not receive posaconazole. The drug should not be given with sirolimus, pimozide, quinidine, atorvastatin, lovastatin, simvastatin, or ergot alkaloids.
Drugs such as cyclosporine and tacrolimus require dose adjustments and frequent blood monitoring when administered with posaconazole. Serious side effects, including nephrotoxicity, leukoencephalopathy, and death, have been reported in patients with increased cyclosporine or tacrolimus blood levels.
Healthcare professionals should use caution when administering posaconazole to patients at risk of developing an irregular heart rhythm, as the drug has been shown to prolong the QT interval, and cases of potentially fatal irregular heart rhythm (torsades de pointes) have been reported in patients taking posaconazole.
Hepatic reactions have been reported as well. This includes mild to moderate elevations in ALT, AST, alkaline phosphatase, total bilirubin, and/or clinical hepatitis. Monitoring or discontinuation may be necessary in patients with hepatic reactions to posaconazole.
IV posaconazole should be avoided in patients with moderate or severe renal impairment (estimated glomerular filtration rate <50 mL/min), unless an assessment of the benefit/risk to the patient justifies the use of posaconazole.
In clinical trials, the adverse events associated with IV posaconazole were generally similar to those in trials of posaconazole oral suspension. The most frequently reported events were diarrhea (32%), hypokalemia (22%), fever (21%), and nausea (19%).
IV posaconazole is under development by MSD (known as Merck in the US and Canada). ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended approval for intravenous (IV) posaconazole (Noxafil), an antifungal agent.
If the European Commission affirms the CHMP opinion, IV posaconazole will be authorized for use in the European Union, Iceland, Liechtenstein, and Norway.
The commission previously granted marketing authorization for posaconazole delayed-release tablets and oral suspension.
Posaconazole is used to prevent invasive fungal infections in severely immunocompromised patients, such as hematopoietic stem cell transplant recipients with graft-vs-host disease or patients with hematologic malignancies and prolonged neutropenia from chemotherapy.
The drug is also used to treat fungal diseases—invasive aspergillosis, fusariosis, chromoblastomycosis, mycetoma, and coccidioidomycosis—when other antifungal agents—amphotericin B, itraconazole, or fluconazole—cannot be tolerated or have failed.
And posaconazole oral suspension is used as a first-line treatment for thrush, a fungal infection of the mouth and throat due to Candida.
Posaconazole injection is administered with a loading dose of 300 mg twice a day on the first day of therapy, then 300 mg once a day thereafter. It is given through a central venous line by IV infusion over approximately 90 minutes.
Once combined with a mixture of IV solution (150 mL of 5% dextrose in water or sodium chloride 0.9%), posaconazole should be administered immediately. If not used immediately, the solution can be stored up to 24 hours if refrigerated at 2°-8° C (36°-46° F).
The safety and effectiveness of IV posaconazole in patients younger than 18 years has not been established. IV posaconazole should not be used in pediatric patients because of non-clinical safety concerns.
Co-administration of drugs that can decrease the plasma concentration of posaconazole should be avoided unless the benefit outweighs the risk. If such drugs are necessary, patients should be monitored closely for breakthrough fungal infections.
Patients with known hypersensitivity to posaconazole or other azole antifungal medicines should not receive posaconazole. The drug should not be given with sirolimus, pimozide, quinidine, atorvastatin, lovastatin, simvastatin, or ergot alkaloids.
Drugs such as cyclosporine and tacrolimus require dose adjustments and frequent blood monitoring when administered with posaconazole. Serious side effects, including nephrotoxicity, leukoencephalopathy, and death, have been reported in patients with increased cyclosporine or tacrolimus blood levels.
Healthcare professionals should use caution when administering posaconazole to patients at risk of developing an irregular heart rhythm, as the drug has been shown to prolong the QT interval, and cases of potentially fatal irregular heart rhythm (torsades de pointes) have been reported in patients taking posaconazole.
Hepatic reactions have been reported as well. This includes mild to moderate elevations in ALT, AST, alkaline phosphatase, total bilirubin, and/or clinical hepatitis. Monitoring or discontinuation may be necessary in patients with hepatic reactions to posaconazole.
IV posaconazole should be avoided in patients with moderate or severe renal impairment (estimated glomerular filtration rate <50 mL/min), unless an assessment of the benefit/risk to the patient justifies the use of posaconazole.
In clinical trials, the adverse events associated with IV posaconazole were generally similar to those in trials of posaconazole oral suspension. The most frequently reported events were diarrhea (32%), hypokalemia (22%), fever (21%), and nausea (19%).
IV posaconazole is under development by MSD (known as Merck in the US and Canada). ![]()
Protein-targeting drug could treat cancers, other diseases

surrounding fused cells
Credit: IRB Barcelona
Experiments in mice suggest the mitochondrial chaperone TRAP-1 is involved in the development of cancers and age-related diseases.
Previous research showed that TRAP-1 is overexpressed in leukemia, lymphoma, and many other cancers.
The new research, published in Cell Reports, clarifies TRAP-1’s role in cancers and age-related conditions. It also suggests gamitrinib, a novel agent targeting TRAP-1, could prove useful in treating these diseases.
TRAP-1 is a member of the heat shock protein 90 (HSP90) family, chaperone proteins that guide the physical formation of other proteins and serve a regulatory function within mitochondria. Tumors use HSP90 proteins like TRAP-1 to help survive therapeutic attack.
To further investigate the effects of TRAP-1, researchers bred TRAP-1 knockout mice. The team found the mice compensate for losing the protein by switching to alternative cellular mechanisms for making energy.
“We see this astounding change in TRAP-1 knockout mice, where they show fewer signs of aging and are less likely to develop cancers,” said Dario C. Altieri, MD, of The Wistar Institute in Philadelphia, Pennsylvania.
“Our findings provide an unexpected explanation for how TRAP-1 and related proteins regulate metabolism within our cells. We usually link the reprogramming of metabolic pathways with human diseases, such as cancer. What we didn’t expect to see were healthier mice with fewer tumors.”
Dr Altieri and his colleagues created the TRAP-1 knockout mice as part of their ongoing investigation into their novel drug, gamitrinib, which targets TRAP-1 in the mitochondria of tumor cells.
“In tumors, the loss of TRAP-1 is devastating, triggering a host of catastrophic defects, including metabolic problems that ultimately result in the death of the tumor cells,” Dr Altieri said. ”Mice that lack TRAP-1 from the start, however, have 3 weeks in the womb to compensate for the loss of the protein.”
The researchers found that, in the knockout mice, the loss of TRAP-1 causes mitochondrial proteins to misfold, which triggers a compensatory response that causes cells to consume more oxygen and metabolize more sugar. This prompts the mitochondria to produce deregulated levels of ATP.
This increased mitochondrial activity actually creates a moderate boost in oxidative stress (free radical damage) and the associated DNA damage. While DNA damage may seem counterproductive to longevity and good health, the low level of DNA damage actually reduces cell proliferation, slowing growth to allow the cell’s natural repair mechanisms to take effect.
According to Dr Altieri, his group’s observations provide a mechanistic foundation for the role of chaperone molecules like HSP90 in the regulation of bioenergetics in mitochondria—how cells produce and use the chemical energy they need to survive and grow.
Their results explain some contradictory findings in the scientific literature regarding the regulation of bioenergetics and show how compensatory mechanisms can arise when these chaperone molecules are taken out of the equation.
“Our findings strengthen the case for targeting HSP90 in tumor cells, but they also open up a fascinating array of questions that may have implications for metabolism and longevity,” Dr Altieri said. “I predict that the TRAP-1 knockout mouse will be a valuable tool for answering these questions.” ![]()

surrounding fused cells
Credit: IRB Barcelona
Experiments in mice suggest the mitochondrial chaperone TRAP-1 is involved in the development of cancers and age-related diseases.
Previous research showed that TRAP-1 is overexpressed in leukemia, lymphoma, and many other cancers.
The new research, published in Cell Reports, clarifies TRAP-1’s role in cancers and age-related conditions. It also suggests gamitrinib, a novel agent targeting TRAP-1, could prove useful in treating these diseases.
TRAP-1 is a member of the heat shock protein 90 (HSP90) family, chaperone proteins that guide the physical formation of other proteins and serve a regulatory function within mitochondria. Tumors use HSP90 proteins like TRAP-1 to help survive therapeutic attack.
To further investigate the effects of TRAP-1, researchers bred TRAP-1 knockout mice. The team found the mice compensate for losing the protein by switching to alternative cellular mechanisms for making energy.
“We see this astounding change in TRAP-1 knockout mice, where they show fewer signs of aging and are less likely to develop cancers,” said Dario C. Altieri, MD, of The Wistar Institute in Philadelphia, Pennsylvania.
“Our findings provide an unexpected explanation for how TRAP-1 and related proteins regulate metabolism within our cells. We usually link the reprogramming of metabolic pathways with human diseases, such as cancer. What we didn’t expect to see were healthier mice with fewer tumors.”
Dr Altieri and his colleagues created the TRAP-1 knockout mice as part of their ongoing investigation into their novel drug, gamitrinib, which targets TRAP-1 in the mitochondria of tumor cells.
“In tumors, the loss of TRAP-1 is devastating, triggering a host of catastrophic defects, including metabolic problems that ultimately result in the death of the tumor cells,” Dr Altieri said. ”Mice that lack TRAP-1 from the start, however, have 3 weeks in the womb to compensate for the loss of the protein.”
The researchers found that, in the knockout mice, the loss of TRAP-1 causes mitochondrial proteins to misfold, which triggers a compensatory response that causes cells to consume more oxygen and metabolize more sugar. This prompts the mitochondria to produce deregulated levels of ATP.
This increased mitochondrial activity actually creates a moderate boost in oxidative stress (free radical damage) and the associated DNA damage. While DNA damage may seem counterproductive to longevity and good health, the low level of DNA damage actually reduces cell proliferation, slowing growth to allow the cell’s natural repair mechanisms to take effect.
According to Dr Altieri, his group’s observations provide a mechanistic foundation for the role of chaperone molecules like HSP90 in the regulation of bioenergetics in mitochondria—how cells produce and use the chemical energy they need to survive and grow.
Their results explain some contradictory findings in the scientific literature regarding the regulation of bioenergetics and show how compensatory mechanisms can arise when these chaperone molecules are taken out of the equation.
“Our findings strengthen the case for targeting HSP90 in tumor cells, but they also open up a fascinating array of questions that may have implications for metabolism and longevity,” Dr Altieri said. “I predict that the TRAP-1 knockout mouse will be a valuable tool for answering these questions.” ![]()

surrounding fused cells
Credit: IRB Barcelona
Experiments in mice suggest the mitochondrial chaperone TRAP-1 is involved in the development of cancers and age-related diseases.
Previous research showed that TRAP-1 is overexpressed in leukemia, lymphoma, and many other cancers.
The new research, published in Cell Reports, clarifies TRAP-1’s role in cancers and age-related conditions. It also suggests gamitrinib, a novel agent targeting TRAP-1, could prove useful in treating these diseases.
TRAP-1 is a member of the heat shock protein 90 (HSP90) family, chaperone proteins that guide the physical formation of other proteins and serve a regulatory function within mitochondria. Tumors use HSP90 proteins like TRAP-1 to help survive therapeutic attack.
To further investigate the effects of TRAP-1, researchers bred TRAP-1 knockout mice. The team found the mice compensate for losing the protein by switching to alternative cellular mechanisms for making energy.
“We see this astounding change in TRAP-1 knockout mice, where they show fewer signs of aging and are less likely to develop cancers,” said Dario C. Altieri, MD, of The Wistar Institute in Philadelphia, Pennsylvania.
“Our findings provide an unexpected explanation for how TRAP-1 and related proteins regulate metabolism within our cells. We usually link the reprogramming of metabolic pathways with human diseases, such as cancer. What we didn’t expect to see were healthier mice with fewer tumors.”
Dr Altieri and his colleagues created the TRAP-1 knockout mice as part of their ongoing investigation into their novel drug, gamitrinib, which targets TRAP-1 in the mitochondria of tumor cells.
“In tumors, the loss of TRAP-1 is devastating, triggering a host of catastrophic defects, including metabolic problems that ultimately result in the death of the tumor cells,” Dr Altieri said. ”Mice that lack TRAP-1 from the start, however, have 3 weeks in the womb to compensate for the loss of the protein.”
The researchers found that, in the knockout mice, the loss of TRAP-1 causes mitochondrial proteins to misfold, which triggers a compensatory response that causes cells to consume more oxygen and metabolize more sugar. This prompts the mitochondria to produce deregulated levels of ATP.
This increased mitochondrial activity actually creates a moderate boost in oxidative stress (free radical damage) and the associated DNA damage. While DNA damage may seem counterproductive to longevity and good health, the low level of DNA damage actually reduces cell proliferation, slowing growth to allow the cell’s natural repair mechanisms to take effect.
According to Dr Altieri, his group’s observations provide a mechanistic foundation for the role of chaperone molecules like HSP90 in the regulation of bioenergetics in mitochondria—how cells produce and use the chemical energy they need to survive and grow.
Their results explain some contradictory findings in the scientific literature regarding the regulation of bioenergetics and show how compensatory mechanisms can arise when these chaperone molecules are taken out of the equation.
“Our findings strengthen the case for targeting HSP90 in tumor cells, but they also open up a fascinating array of questions that may have implications for metabolism and longevity,” Dr Altieri said. “I predict that the TRAP-1 knockout mouse will be a valuable tool for answering these questions.” ![]()
Study reveals why HSCs falter with age

in the bone marrow
A new study helps explain how blood production declines with age and why older individuals are not suitable donors for hematopoietic stem cell (HSC) transplant.
The research also reveals a potential approach for mitigating
the negative effects of aging on the blood, which can lead to anemia,
bone marrow failure, and myeloid malignancies.
The study, conducted in mice, suggests HSCs falter with age because they lose the ability to replicate their DNA accurately and efficiently during cell division.
Emmanuelle Passegué, PhD, of the University of California San Francisco, and her colleagues reported this discovery in Nature.
The researchers analyzed old HSCs in mice and found a scarcity of protein components needed to form the mini-chromosome maintenance helicase. This molecular machine unwinds double-stranded DNA so the cell’s genetic material can be duplicated and allocated to daughter cells later in cell division.
The HSCs were stressed by the loss of this machine’s activity. As a result, they had an increased risk for DNA damage and death when forced to divide.
On the other hand, the cells tended to survive unless they were confronted with a “strong replication challenge” like transplantation.
The researchers also discovered that even after the stress associated with DNA replication, old HSCs retained molecular tags on histones, a feature often associated with DNA damage.
However, these old survivors could repair induced DNA damage as efficiently as young stem cells.
“Old stem cells are not just sitting there with damaged DNA ready to develop cancer, as it has long been postulated,” Dr Passegué said.
Of course, not all was well in the old, surviving HSCs. The molecular tags accumulated on genes needed to make ribosomes.
Dr Passegué said she will further explore the consequences of reduced protein production as part of her ongoing research. She hopes it might be possible to prevent declining stem cell populations by developing a drug to prevent the loss of the helicase components needed to unwind and replicate DNA, thereby avoiding immune system failure. ![]()

in the bone marrow
A new study helps explain how blood production declines with age and why older individuals are not suitable donors for hematopoietic stem cell (HSC) transplant.
The research also reveals a potential approach for mitigating
the negative effects of aging on the blood, which can lead to anemia,
bone marrow failure, and myeloid malignancies.
The study, conducted in mice, suggests HSCs falter with age because they lose the ability to replicate their DNA accurately and efficiently during cell division.
Emmanuelle Passegué, PhD, of the University of California San Francisco, and her colleagues reported this discovery in Nature.
The researchers analyzed old HSCs in mice and found a scarcity of protein components needed to form the mini-chromosome maintenance helicase. This molecular machine unwinds double-stranded DNA so the cell’s genetic material can be duplicated and allocated to daughter cells later in cell division.
The HSCs were stressed by the loss of this machine’s activity. As a result, they had an increased risk for DNA damage and death when forced to divide.
On the other hand, the cells tended to survive unless they were confronted with a “strong replication challenge” like transplantation.
The researchers also discovered that even after the stress associated with DNA replication, old HSCs retained molecular tags on histones, a feature often associated with DNA damage.
However, these old survivors could repair induced DNA damage as efficiently as young stem cells.
“Old stem cells are not just sitting there with damaged DNA ready to develop cancer, as it has long been postulated,” Dr Passegué said.
Of course, not all was well in the old, surviving HSCs. The molecular tags accumulated on genes needed to make ribosomes.
Dr Passegué said she will further explore the consequences of reduced protein production as part of her ongoing research. She hopes it might be possible to prevent declining stem cell populations by developing a drug to prevent the loss of the helicase components needed to unwind and replicate DNA, thereby avoiding immune system failure. ![]()

in the bone marrow
A new study helps explain how blood production declines with age and why older individuals are not suitable donors for hematopoietic stem cell (HSC) transplant.
The research also reveals a potential approach for mitigating
the negative effects of aging on the blood, which can lead to anemia,
bone marrow failure, and myeloid malignancies.
The study, conducted in mice, suggests HSCs falter with age because they lose the ability to replicate their DNA accurately and efficiently during cell division.
Emmanuelle Passegué, PhD, of the University of California San Francisco, and her colleagues reported this discovery in Nature.
The researchers analyzed old HSCs in mice and found a scarcity of protein components needed to form the mini-chromosome maintenance helicase. This molecular machine unwinds double-stranded DNA so the cell’s genetic material can be duplicated and allocated to daughter cells later in cell division.
The HSCs were stressed by the loss of this machine’s activity. As a result, they had an increased risk for DNA damage and death when forced to divide.
On the other hand, the cells tended to survive unless they were confronted with a “strong replication challenge” like transplantation.
The researchers also discovered that even after the stress associated with DNA replication, old HSCs retained molecular tags on histones, a feature often associated with DNA damage.
However, these old survivors could repair induced DNA damage as efficiently as young stem cells.
“Old stem cells are not just sitting there with damaged DNA ready to develop cancer, as it has long been postulated,” Dr Passegué said.
Of course, not all was well in the old, surviving HSCs. The molecular tags accumulated on genes needed to make ribosomes.
Dr Passegué said she will further explore the consequences of reduced protein production as part of her ongoing research. She hopes it might be possible to prevent declining stem cell populations by developing a drug to prevent the loss of the helicase components needed to unwind and replicate DNA, thereby avoiding immune system failure. ![]()
Approach could improve treatment of lymphoma, other cancers
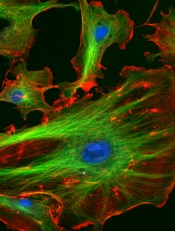
Credit: NIH
Targeting a molecule in endothelial cells could make cancer therapies significantly more effective, preclinical research suggests.
The researchers found that a molecule called focal adhesion kinase (FAK) can help protect cancer cells from the damaging effects of chemotherapy and radiotherapy.
But deleting FAK can enhance the effects of treatment directed against melanoma, lung cancer, and lymphoma.
The team recounted these findings in Nature.
“This work shows that sensitivity to cancer treatment is related to our own body mistakenly trying to shield the cancer from cell-killing effects caused by radiotherapy and chemotherapy,” said study author Bernardo Tavora, PhD, of Rockefeller University in New York.
“Although taking out FAK from blood vessels won’t destroy the cancer by itself, it can remove the barrier cancer uses to protect itself from treatment.”
Dr Tavora and his colleagues removed FAK from endothelial cells in mouse models of melanoma, lung cancer, and lymphoma. This had no effect on tumor growth in untreated mice.
However, the loss of endothelial-cell FAK did aid the effects of doxorubicin and radiotherapy. It increased apoptosis and decreased proliferation within perivascular tumor-cell compartments, thereby extending survival in the mice.
The researchers also studied samples from lymphoma patients. And they found that patients with low levels of FAK were more likely to achieve a complete remission after treatment.
Investigation into the mechanism behind these effects revealed that endothelial-cell FAK is required for the production of cytokines and for NF-κB activation induced by DNA damage.
So the loss of endothelial-cell FAK reduces DNA-damage-induced cytokine production, thereby increasing cancer cells’ sensitivity to DNA-damaging therapies in vitro and in vivo.
Taken together, these results suggest that developing drugs to target FAK could help improve the efficacy of cancer treatments and potentially prevent relapse in a number of malignancies. ![]()

Credit: NIH
Targeting a molecule in endothelial cells could make cancer therapies significantly more effective, preclinical research suggests.
The researchers found that a molecule called focal adhesion kinase (FAK) can help protect cancer cells from the damaging effects of chemotherapy and radiotherapy.
But deleting FAK can enhance the effects of treatment directed against melanoma, lung cancer, and lymphoma.
The team recounted these findings in Nature.
“This work shows that sensitivity to cancer treatment is related to our own body mistakenly trying to shield the cancer from cell-killing effects caused by radiotherapy and chemotherapy,” said study author Bernardo Tavora, PhD, of Rockefeller University in New York.
“Although taking out FAK from blood vessels won’t destroy the cancer by itself, it can remove the barrier cancer uses to protect itself from treatment.”
Dr Tavora and his colleagues removed FAK from endothelial cells in mouse models of melanoma, lung cancer, and lymphoma. This had no effect on tumor growth in untreated mice.
However, the loss of endothelial-cell FAK did aid the effects of doxorubicin and radiotherapy. It increased apoptosis and decreased proliferation within perivascular tumor-cell compartments, thereby extending survival in the mice.
The researchers also studied samples from lymphoma patients. And they found that patients with low levels of FAK were more likely to achieve a complete remission after treatment.
Investigation into the mechanism behind these effects revealed that endothelial-cell FAK is required for the production of cytokines and for NF-κB activation induced by DNA damage.
So the loss of endothelial-cell FAK reduces DNA-damage-induced cytokine production, thereby increasing cancer cells’ sensitivity to DNA-damaging therapies in vitro and in vivo.
Taken together, these results suggest that developing drugs to target FAK could help improve the efficacy of cancer treatments and potentially prevent relapse in a number of malignancies. ![]()

Credit: NIH
Targeting a molecule in endothelial cells could make cancer therapies significantly more effective, preclinical research suggests.
The researchers found that a molecule called focal adhesion kinase (FAK) can help protect cancer cells from the damaging effects of chemotherapy and radiotherapy.
But deleting FAK can enhance the effects of treatment directed against melanoma, lung cancer, and lymphoma.
The team recounted these findings in Nature.
“This work shows that sensitivity to cancer treatment is related to our own body mistakenly trying to shield the cancer from cell-killing effects caused by radiotherapy and chemotherapy,” said study author Bernardo Tavora, PhD, of Rockefeller University in New York.
“Although taking out FAK from blood vessels won’t destroy the cancer by itself, it can remove the barrier cancer uses to protect itself from treatment.”
Dr Tavora and his colleagues removed FAK from endothelial cells in mouse models of melanoma, lung cancer, and lymphoma. This had no effect on tumor growth in untreated mice.
However, the loss of endothelial-cell FAK did aid the effects of doxorubicin and radiotherapy. It increased apoptosis and decreased proliferation within perivascular tumor-cell compartments, thereby extending survival in the mice.
The researchers also studied samples from lymphoma patients. And they found that patients with low levels of FAK were more likely to achieve a complete remission after treatment.
Investigation into the mechanism behind these effects revealed that endothelial-cell FAK is required for the production of cytokines and for NF-κB activation induced by DNA damage.
So the loss of endothelial-cell FAK reduces DNA-damage-induced cytokine production, thereby increasing cancer cells’ sensitivity to DNA-damaging therapies in vitro and in vivo.
Taken together, these results suggest that developing drugs to target FAK could help improve the efficacy of cancer treatments and potentially prevent relapse in a number of malignancies. ![]()
Overcoming ibrutinib resistance in MCL

Credit: Rhoda Baer
Investigators have identified drug combinations that may overcome resistance to ibrutinib in patients with mantle cell lymphoma (MCL).
The group discovered a mutation in Bruton’s tyrosine kinase (BTK) that confers resistance to the drug.
They also found that high levels of PI3K-AKT and CDK4 signaling could explain innate resistance to ibrutinib, so combining a CDK4 inhibitor and a PI3K inhibitor might be effective in patients who don’t respond to ibrutinib.
Selina Chen-Kiang, PhD, of Weill Cornell Medical College in New York, and her colleagues detailed these findings in Cancer Discovery. Some of Dr Chen-Kiang’s colleagues reported relationships with Janssen and Pharmacyclics, the companies developing ibrutinib.
“[Ibrutinib] doesn’t work for about 32% of patients, and their lymphomas are said to have primary resistance to ibrutinib,” Dr Chen-Kiang noted. “We are also learning that most patients whose lymphomas respond to ibrutinib eventually relapse because their tumors acquire resistance to the drug.”
“The knowledge that we gained from longitudinal RNA and genomic sequencing of mantle cell lymphomas with primary and acquired resistance to ibrutinib allowed us to identify rational drug combinations that may overcome resistance in these 2 settings.”
Dr Chen-Kiang and her colleagues conducted whole-exome and whole-transcriptome analyses of 5 serial biopsies from a patient with MCL who initially responded to ibrutinib before progressing.
After comparing these data with results from an analysis of healthy tissues from the same patient, the investigators found that a mutation in BTK, the C481S mutation, appeared at relapse.
The researchers found the same mutation at relapse in a second MCL patient with acquired resistance to ibrutinib but not in any patients with primary resistance to the drug.
Further analyses revealed the consequences of the relapse-specific BTK C481S mutation, including activation of the PI3K and CDK4 signaling pathways, which promote cell survival and proliferation.
Inhibiting CDK4 with the investigational anticancer drug palbociclib made ibrutinib-resistant lymphoma cells carrying the BTK C481S mutation sensitive to investigational drugs that inhibit PI3K.
And palbociclib made ibrutinib-resistant lymphoma cells harboring normal BTK sensitive to both ibrutinib and investigational drugs that inhibit PI3K. The researchers recently opened a clinical trial to test ibrutinib and palbociclib in combination (NCT02159755).
“We are very excited to have generated data . . . that may be meaningful for patients,” Dr Chen-Kiang said. “It is also exciting because CDK4 is a new kind of drug target. It controls the cell cycle, which is a central cancer pathway. As such, it is not just important for mantle cell lymphoma but for many forms of cancer.” ![]()

Credit: Rhoda Baer
Investigators have identified drug combinations that may overcome resistance to ibrutinib in patients with mantle cell lymphoma (MCL).
The group discovered a mutation in Bruton’s tyrosine kinase (BTK) that confers resistance to the drug.
They also found that high levels of PI3K-AKT and CDK4 signaling could explain innate resistance to ibrutinib, so combining a CDK4 inhibitor and a PI3K inhibitor might be effective in patients who don’t respond to ibrutinib.
Selina Chen-Kiang, PhD, of Weill Cornell Medical College in New York, and her colleagues detailed these findings in Cancer Discovery. Some of Dr Chen-Kiang’s colleagues reported relationships with Janssen and Pharmacyclics, the companies developing ibrutinib.
“[Ibrutinib] doesn’t work for about 32% of patients, and their lymphomas are said to have primary resistance to ibrutinib,” Dr Chen-Kiang noted. “We are also learning that most patients whose lymphomas respond to ibrutinib eventually relapse because their tumors acquire resistance to the drug.”
“The knowledge that we gained from longitudinal RNA and genomic sequencing of mantle cell lymphomas with primary and acquired resistance to ibrutinib allowed us to identify rational drug combinations that may overcome resistance in these 2 settings.”
Dr Chen-Kiang and her colleagues conducted whole-exome and whole-transcriptome analyses of 5 serial biopsies from a patient with MCL who initially responded to ibrutinib before progressing.
After comparing these data with results from an analysis of healthy tissues from the same patient, the investigators found that a mutation in BTK, the C481S mutation, appeared at relapse.
The researchers found the same mutation at relapse in a second MCL patient with acquired resistance to ibrutinib but not in any patients with primary resistance to the drug.
Further analyses revealed the consequences of the relapse-specific BTK C481S mutation, including activation of the PI3K and CDK4 signaling pathways, which promote cell survival and proliferation.
Inhibiting CDK4 with the investigational anticancer drug palbociclib made ibrutinib-resistant lymphoma cells carrying the BTK C481S mutation sensitive to investigational drugs that inhibit PI3K.
And palbociclib made ibrutinib-resistant lymphoma cells harboring normal BTK sensitive to both ibrutinib and investigational drugs that inhibit PI3K. The researchers recently opened a clinical trial to test ibrutinib and palbociclib in combination (NCT02159755).
“We are very excited to have generated data . . . that may be meaningful for patients,” Dr Chen-Kiang said. “It is also exciting because CDK4 is a new kind of drug target. It controls the cell cycle, which is a central cancer pathway. As such, it is not just important for mantle cell lymphoma but for many forms of cancer.” ![]()

Credit: Rhoda Baer
Investigators have identified drug combinations that may overcome resistance to ibrutinib in patients with mantle cell lymphoma (MCL).
The group discovered a mutation in Bruton’s tyrosine kinase (BTK) that confers resistance to the drug.
They also found that high levels of PI3K-AKT and CDK4 signaling could explain innate resistance to ibrutinib, so combining a CDK4 inhibitor and a PI3K inhibitor might be effective in patients who don’t respond to ibrutinib.
Selina Chen-Kiang, PhD, of Weill Cornell Medical College in New York, and her colleagues detailed these findings in Cancer Discovery. Some of Dr Chen-Kiang’s colleagues reported relationships with Janssen and Pharmacyclics, the companies developing ibrutinib.
“[Ibrutinib] doesn’t work for about 32% of patients, and their lymphomas are said to have primary resistance to ibrutinib,” Dr Chen-Kiang noted. “We are also learning that most patients whose lymphomas respond to ibrutinib eventually relapse because their tumors acquire resistance to the drug.”
“The knowledge that we gained from longitudinal RNA and genomic sequencing of mantle cell lymphomas with primary and acquired resistance to ibrutinib allowed us to identify rational drug combinations that may overcome resistance in these 2 settings.”
Dr Chen-Kiang and her colleagues conducted whole-exome and whole-transcriptome analyses of 5 serial biopsies from a patient with MCL who initially responded to ibrutinib before progressing.
After comparing these data with results from an analysis of healthy tissues from the same patient, the investigators found that a mutation in BTK, the C481S mutation, appeared at relapse.
The researchers found the same mutation at relapse in a second MCL patient with acquired resistance to ibrutinib but not in any patients with primary resistance to the drug.
Further analyses revealed the consequences of the relapse-specific BTK C481S mutation, including activation of the PI3K and CDK4 signaling pathways, which promote cell survival and proliferation.
Inhibiting CDK4 with the investigational anticancer drug palbociclib made ibrutinib-resistant lymphoma cells carrying the BTK C481S mutation sensitive to investigational drugs that inhibit PI3K.
And palbociclib made ibrutinib-resistant lymphoma cells harboring normal BTK sensitive to both ibrutinib and investigational drugs that inhibit PI3K. The researchers recently opened a clinical trial to test ibrutinib and palbociclib in combination (NCT02159755).
“We are very excited to have generated data . . . that may be meaningful for patients,” Dr Chen-Kiang said. “It is also exciting because CDK4 is a new kind of drug target. It controls the cell cycle, which is a central cancer pathway. As such, it is not just important for mantle cell lymphoma but for many forms of cancer.”
How lymphoma affects male fertility
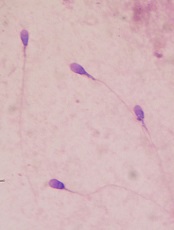
for semen quality testing
New research has shown that lymphoma and its treatment can impact a number of sperm characteristics, thereby reducing fertility in males.
Results also indicated that most patients eventually experience semen recovery, but the degree and timing of that recovery may depend on the patient’s diagnosis and treatment.
In this study, recovery was more likely among patients with Hodgkin lymphoma than those with non-Hodgkin lymphoma.
And recovery was both quicker and more likely among patients who did not receive alkylating chemotherapy.
On the other hand, multivariate analyses suggested that only a patient’s pre-treatment total sperm count was related to recovery.
Louis Bujan, MD, PhD, of Université de Toulouse in France, and colleagues reported these results in Fertility and Sterility.
The study included 75 patients—57 with Hodgkin lymphoma and 18 with non-Hodgkin lymphoma. The researchers collected sperm samples before patients began cancer treatment and again at later intervals: 3 months, 6 months, 12 months, and 24 months post-treatment.
The team compared patients’ sperm characteristics to those of a control group consisting of 257 healthy, fertile men.
Results revealed that lymphoma patients had impaired sperm quality even before they began treatment. Compared to fertile controls, patients had higher levels of sperm chromatin alterations and DNA fragmentation, with the only risk factor being their cancer diagnosis.
However, between 3 months and 6 months post-treatment, patients’ levels of sperm DNA fragmentation and chromatin structure damage improved. The damage level decreased relative to a patient’s own pre-treatment level of damage, while still remaining higher than damage levels in the control group.
After treatment, patients’ sperm density, total count, motility, and vitality decreased, with the lowest values seen at the 3- and 6-month marks.
Alkylating chemotherapy was more detrimental to spermatogenesis than non-alkylating drugs. Patients who received alkylating chemotherapy were more likely to cease sperm production entirely or take longer to resume sperm production than patients receiving non-alkylating chemotherapy.
Twelve months after treatment, mean sperm counts recovered to pre-treatment values for patients who had received doxorubicin, bleomycin, vinblastine, and darcarbacine (ABVD) or ABVD and radiotherapy.
But this was not the case for patients who received doxorubicin, cyclophosphamide, vincristine, and prednisone (CHOP) or mechlorethamine, oncovin, procarbazine, and prednisone (MOPP).
At the 24-month mark, 7% of patients remained azoospermic. Kaplan Meir estimates suggested that, after 24 months, most patients would recover normal sperm counts.
Recovery was projected for 92% of patients who received ABVD and radiotherapy, 90% of patients who received ABVD alone, and 61% of CHOP-treated patients. (There was no estimate for MOPP therapy, perhaps due to a low number of patients.)
A patient’s type of lymphoma appeared to impact sperm count recovery as well. Estimates suggested that, after 24 months, 86% of Hodgkin lymphoma patients would experience recovery, compared to 73% of non-Hodgkin lymphoma patients.
“While many men can look forward to their fertility returning after treatment is over, not all will be so fortunate,” said Rebecca Sokol, MD, MPH, President of the American Society for Reproductive Medicine.
“It is imperative that, prior to the initiation of therapy, counseling and sperm preservation be made available to all lymphoma patients and their partners who may want to have children in the future.”

for semen quality testing
New research has shown that lymphoma and its treatment can impact a number of sperm characteristics, thereby reducing fertility in males.
Results also indicated that most patients eventually experience semen recovery, but the degree and timing of that recovery may depend on the patient’s diagnosis and treatment.
In this study, recovery was more likely among patients with Hodgkin lymphoma than those with non-Hodgkin lymphoma.
And recovery was both quicker and more likely among patients who did not receive alkylating chemotherapy.
On the other hand, multivariate analyses suggested that only a patient’s pre-treatment total sperm count was related to recovery.
Louis Bujan, MD, PhD, of Université de Toulouse in France, and colleagues reported these results in Fertility and Sterility.
The study included 75 patients—57 with Hodgkin lymphoma and 18 with non-Hodgkin lymphoma. The researchers collected sperm samples before patients began cancer treatment and again at later intervals: 3 months, 6 months, 12 months, and 24 months post-treatment.
The team compared patients’ sperm characteristics to those of a control group consisting of 257 healthy, fertile men.
Results revealed that lymphoma patients had impaired sperm quality even before they began treatment. Compared to fertile controls, patients had higher levels of sperm chromatin alterations and DNA fragmentation, with the only risk factor being their cancer diagnosis.
However, between 3 months and 6 months post-treatment, patients’ levels of sperm DNA fragmentation and chromatin structure damage improved. The damage level decreased relative to a patient’s own pre-treatment level of damage, while still remaining higher than damage levels in the control group.
After treatment, patients’ sperm density, total count, motility, and vitality decreased, with the lowest values seen at the 3- and 6-month marks.
Alkylating chemotherapy was more detrimental to spermatogenesis than non-alkylating drugs. Patients who received alkylating chemotherapy were more likely to cease sperm production entirely or take longer to resume sperm production than patients receiving non-alkylating chemotherapy.
Twelve months after treatment, mean sperm counts recovered to pre-treatment values for patients who had received doxorubicin, bleomycin, vinblastine, and darcarbacine (ABVD) or ABVD and radiotherapy.
But this was not the case for patients who received doxorubicin, cyclophosphamide, vincristine, and prednisone (CHOP) or mechlorethamine, oncovin, procarbazine, and prednisone (MOPP).
At the 24-month mark, 7% of patients remained azoospermic. Kaplan Meir estimates suggested that, after 24 months, most patients would recover normal sperm counts.
Recovery was projected for 92% of patients who received ABVD and radiotherapy, 90% of patients who received ABVD alone, and 61% of CHOP-treated patients. (There was no estimate for MOPP therapy, perhaps due to a low number of patients.)
A patient’s type of lymphoma appeared to impact sperm count recovery as well. Estimates suggested that, after 24 months, 86% of Hodgkin lymphoma patients would experience recovery, compared to 73% of non-Hodgkin lymphoma patients.
“While many men can look forward to their fertility returning after treatment is over, not all will be so fortunate,” said Rebecca Sokol, MD, MPH, President of the American Society for Reproductive Medicine.
“It is imperative that, prior to the initiation of therapy, counseling and sperm preservation be made available to all lymphoma patients and their partners who may want to have children in the future.”

for semen quality testing
New research has shown that lymphoma and its treatment can impact a number of sperm characteristics, thereby reducing fertility in males.
Results also indicated that most patients eventually experience semen recovery, but the degree and timing of that recovery may depend on the patient’s diagnosis and treatment.
In this study, recovery was more likely among patients with Hodgkin lymphoma than those with non-Hodgkin lymphoma.
And recovery was both quicker and more likely among patients who did not receive alkylating chemotherapy.
On the other hand, multivariate analyses suggested that only a patient’s pre-treatment total sperm count was related to recovery.
Louis Bujan, MD, PhD, of Université de Toulouse in France, and colleagues reported these results in Fertility and Sterility.
The study included 75 patients—57 with Hodgkin lymphoma and 18 with non-Hodgkin lymphoma. The researchers collected sperm samples before patients began cancer treatment and again at later intervals: 3 months, 6 months, 12 months, and 24 months post-treatment.
The team compared patients’ sperm characteristics to those of a control group consisting of 257 healthy, fertile men.
Results revealed that lymphoma patients had impaired sperm quality even before they began treatment. Compared to fertile controls, patients had higher levels of sperm chromatin alterations and DNA fragmentation, with the only risk factor being their cancer diagnosis.
However, between 3 months and 6 months post-treatment, patients’ levels of sperm DNA fragmentation and chromatin structure damage improved. The damage level decreased relative to a patient’s own pre-treatment level of damage, while still remaining higher than damage levels in the control group.
After treatment, patients’ sperm density, total count, motility, and vitality decreased, with the lowest values seen at the 3- and 6-month marks.
Alkylating chemotherapy was more detrimental to spermatogenesis than non-alkylating drugs. Patients who received alkylating chemotherapy were more likely to cease sperm production entirely or take longer to resume sperm production than patients receiving non-alkylating chemotherapy.
Twelve months after treatment, mean sperm counts recovered to pre-treatment values for patients who had received doxorubicin, bleomycin, vinblastine, and darcarbacine (ABVD) or ABVD and radiotherapy.
But this was not the case for patients who received doxorubicin, cyclophosphamide, vincristine, and prednisone (CHOP) or mechlorethamine, oncovin, procarbazine, and prednisone (MOPP).
At the 24-month mark, 7% of patients remained azoospermic. Kaplan Meir estimates suggested that, after 24 months, most patients would recover normal sperm counts.
Recovery was projected for 92% of patients who received ABVD and radiotherapy, 90% of patients who received ABVD alone, and 61% of CHOP-treated patients. (There was no estimate for MOPP therapy, perhaps due to a low number of patients.)
A patient’s type of lymphoma appeared to impact sperm count recovery as well. Estimates suggested that, after 24 months, 86% of Hodgkin lymphoma patients would experience recovery, compared to 73% of non-Hodgkin lymphoma patients.
“While many men can look forward to their fertility returning after treatment is over, not all will be so fortunate,” said Rebecca Sokol, MD, MPH, President of the American Society for Reproductive Medicine.
“It is imperative that, prior to the initiation of therapy, counseling and sperm preservation be made available to all lymphoma patients and their partners who may want to have children in the future.”
Obinutuzumab approved for CLL in Europe
The European Commission has approved the anti-CD20 monoclonal antibody obinutuzumab for use in the European Union (EU).
Obinutuzumab can now be used in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia (CLL) who have comorbidities that make them ineligible to receive fludarabine-based therapy.
Obinutuzumab is already approved for this indication in the US.
Obinutuzumab is marketed as Gazyvaro in the EU and Switzerland but as Gazyva in the US and the rest of the world.
The European Commission’s approval follows a positive opinion granted by The European Medicine Agency’s Committee for Medicinal Products for Human Use in May.
The approval is based on results of the phase 3 CLL11 study, which showed that obinutuzumab plus chlorambucil improved progression-free survival (PFS), when compared to chlorambucil alone or in combination with rituximab.
This 2-stage study included 781 previously untreated CLL patients with comorbidities. In stage 1 (n=589), researchers compared obinutuzumab plus chlorambucil to chlorambucil alone and rituximab plus chlorambucil to chlorambucil alone.
Stage 2 (n=663) was a direct comparison of obinutuzumab plus chlorambucil and rituximab plus chlorambucil.
Stage 1 results were presented at ASCO 2013, stage 2 results were presented at ASH 2013, and the complete results were published in NEJM last March.
Obinutuzumab plus chlorambucil improved PFS when compared to chlorambucil alone. The median PFS was 26.7 months and 11.1 months, respectively (P<0.001).
Obinutuzumab plus chlorambucil also improved PFS when compared to rituximab plus chlorambucil. The median PFS was 26.7 months and 16.3 months, respectively (P<0.001).
Infusion-related reactions and neutropenia were more common in the obinutuzumab arm than in the rituximab arm. But obinutuzumab-treated patients did not have an increased risk of infection.
Obinutuzumab is being developed by Roche. The company said it expects to begin launching the drug in a number of European countries this year.
The European Commission has approved the anti-CD20 monoclonal antibody obinutuzumab for use in the European Union (EU).
Obinutuzumab can now be used in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia (CLL) who have comorbidities that make them ineligible to receive fludarabine-based therapy.
Obinutuzumab is already approved for this indication in the US.
Obinutuzumab is marketed as Gazyvaro in the EU and Switzerland but as Gazyva in the US and the rest of the world.
The European Commission’s approval follows a positive opinion granted by The European Medicine Agency’s Committee for Medicinal Products for Human Use in May.
The approval is based on results of the phase 3 CLL11 study, which showed that obinutuzumab plus chlorambucil improved progression-free survival (PFS), when compared to chlorambucil alone or in combination with rituximab.
This 2-stage study included 781 previously untreated CLL patients with comorbidities. In stage 1 (n=589), researchers compared obinutuzumab plus chlorambucil to chlorambucil alone and rituximab plus chlorambucil to chlorambucil alone.
Stage 2 (n=663) was a direct comparison of obinutuzumab plus chlorambucil and rituximab plus chlorambucil.
Stage 1 results were presented at ASCO 2013, stage 2 results were presented at ASH 2013, and the complete results were published in NEJM last March.
Obinutuzumab plus chlorambucil improved PFS when compared to chlorambucil alone. The median PFS was 26.7 months and 11.1 months, respectively (P<0.001).
Obinutuzumab plus chlorambucil also improved PFS when compared to rituximab plus chlorambucil. The median PFS was 26.7 months and 16.3 months, respectively (P<0.001).
Infusion-related reactions and neutropenia were more common in the obinutuzumab arm than in the rituximab arm. But obinutuzumab-treated patients did not have an increased risk of infection.
Obinutuzumab is being developed by Roche. The company said it expects to begin launching the drug in a number of European countries this year.
The European Commission has approved the anti-CD20 monoclonal antibody obinutuzumab for use in the European Union (EU).
Obinutuzumab can now be used in combination with chlorambucil to treat patients with previously untreated chronic lymphocytic leukemia (CLL) who have comorbidities that make them ineligible to receive fludarabine-based therapy.
Obinutuzumab is already approved for this indication in the US.
Obinutuzumab is marketed as Gazyvaro in the EU and Switzerland but as Gazyva in the US and the rest of the world.
The European Commission’s approval follows a positive opinion granted by The European Medicine Agency’s Committee for Medicinal Products for Human Use in May.
The approval is based on results of the phase 3 CLL11 study, which showed that obinutuzumab plus chlorambucil improved progression-free survival (PFS), when compared to chlorambucil alone or in combination with rituximab.
This 2-stage study included 781 previously untreated CLL patients with comorbidities. In stage 1 (n=589), researchers compared obinutuzumab plus chlorambucil to chlorambucil alone and rituximab plus chlorambucil to chlorambucil alone.
Stage 2 (n=663) was a direct comparison of obinutuzumab plus chlorambucil and rituximab plus chlorambucil.
Stage 1 results were presented at ASCO 2013, stage 2 results were presented at ASH 2013, and the complete results were published in NEJM last March.
Obinutuzumab plus chlorambucil improved PFS when compared to chlorambucil alone. The median PFS was 26.7 months and 11.1 months, respectively (P<0.001).
Obinutuzumab plus chlorambucil also improved PFS when compared to rituximab plus chlorambucil. The median PFS was 26.7 months and 16.3 months, respectively (P<0.001).
Infusion-related reactions and neutropenia were more common in the obinutuzumab arm than in the rituximab arm. But obinutuzumab-treated patients did not have an increased risk of infection.
Obinutuzumab is being developed by Roche. The company said it expects to begin launching the drug in a number of European countries this year.
Healthy habits can cut risk of metabolic syndrome in childhood cancer survivors

patient and her father
Credit: Rhoda Baer
Following a healthy lifestyle can decrease the risk of metabolic syndrome in childhood cancer survivors, according to a study published in Cancer.
Unfortunately, only about a quarter of the survivors studied actually practiced healthy lifestyle habits, such as engaging in moderate physical activity; eating the recommended daily serving of fruits, vegetables, and complex carbohydrates; and consuming red meat, alcohol, and sodium in moderation.
Childhood cancer survivors are known to have an increased risk of developing metabolic syndrome.
The syndrome is actually a number of conditions—high blood pressure, increased body fat, and abnormal cholesterol and glucose levels—that, when they occur together, increase a person’s risk of heart disease, stroke, and diabetes.
Kirsten Ness, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues wanted to determine if lifestyle habits might affect the risk of metabolic syndrome among childhood cancer survivors.
So the team analyzed 1598 survivors who were cancer-free for at least 10 years. They had a median age of 32.7 years (range, 18.9 to 60).
The analysis showed that failure to follow healthy lifestyle guidelines roughly doubled the survivors’ risk of developing metabolic syndrome. Women had a 2.4-times greater risk, and men had a 2.2-times greater risk of the syndrome if they did not follow the guidelines.
Metabolic syndrome was present in 31.8% of the participants—32.5% of males and 31% of females.
The researchers considered a subject to have metabolic syndrome if he had or received treatment for 3 or more of the following:
- Abdominal obesity (waist circumference of > 102 cm in males and > 88 cm in females)
- Triglycerides ≥ 150 mg/dL
- High-density lipoprotein cholesterol (< 40 mg/dL in males and < 50 mg/dL in females)
- Hypertension (systolic pressure ≥ 130 mm Hg or diastolic pressure ≥ 85 mm Hg)
- Fasting plasma glucose ≥ 100 mg/dL.
Questionnaires and tests helped the researchers assess whether participants followed healthy lifestyle recommendations issued by the World Cancer Research Fund and American Institute for Cancer Research.
The recommendations include:
- Having a body mass index of 25 or lower
- Engaging in moderate physical activity for 150 minutes each week
- Eating 5 or more servings of fruits and vegetables each day
- Consuming 400 g or more of complex carbohydrates daily
- Eating less than 80 g of red meat each day
- Consuming less than 2400 mg of sodium each day
- Low daily alcohol consumption (less than 14 g for females and less than 28 g for males).
Subjects who met at least 4 of these 7 criteria were classified as following the guidelines. And 27% of the participants—25.2% of males and 28.8% of females—were classified as such.
“These findings are important because they indicate that adults who were treated for cancer as children have the opportunity to influence their own health outcomes,” Dr Ness said.
“[A]dopting a lifestyle that includes maintaining a healthy body weight, regular physical activity, and a diet that includes fruits and vegetables and that limits refined sugars, excessive alcohol, red meat, and salt has potential to prevent development of metabolic syndrome.”

patient and her father
Credit: Rhoda Baer
Following a healthy lifestyle can decrease the risk of metabolic syndrome in childhood cancer survivors, according to a study published in Cancer.
Unfortunately, only about a quarter of the survivors studied actually practiced healthy lifestyle habits, such as engaging in moderate physical activity; eating the recommended daily serving of fruits, vegetables, and complex carbohydrates; and consuming red meat, alcohol, and sodium in moderation.
Childhood cancer survivors are known to have an increased risk of developing metabolic syndrome.
The syndrome is actually a number of conditions—high blood pressure, increased body fat, and abnormal cholesterol and glucose levels—that, when they occur together, increase a person’s risk of heart disease, stroke, and diabetes.
Kirsten Ness, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues wanted to determine if lifestyle habits might affect the risk of metabolic syndrome among childhood cancer survivors.
So the team analyzed 1598 survivors who were cancer-free for at least 10 years. They had a median age of 32.7 years (range, 18.9 to 60).
The analysis showed that failure to follow healthy lifestyle guidelines roughly doubled the survivors’ risk of developing metabolic syndrome. Women had a 2.4-times greater risk, and men had a 2.2-times greater risk of the syndrome if they did not follow the guidelines.
Metabolic syndrome was present in 31.8% of the participants—32.5% of males and 31% of females.
The researchers considered a subject to have metabolic syndrome if he had or received treatment for 3 or more of the following:
- Abdominal obesity (waist circumference of > 102 cm in males and > 88 cm in females)
- Triglycerides ≥ 150 mg/dL
- High-density lipoprotein cholesterol (< 40 mg/dL in males and < 50 mg/dL in females)
- Hypertension (systolic pressure ≥ 130 mm Hg or diastolic pressure ≥ 85 mm Hg)
- Fasting plasma glucose ≥ 100 mg/dL.
Questionnaires and tests helped the researchers assess whether participants followed healthy lifestyle recommendations issued by the World Cancer Research Fund and American Institute for Cancer Research.
The recommendations include:
- Having a body mass index of 25 or lower
- Engaging in moderate physical activity for 150 minutes each week
- Eating 5 or more servings of fruits and vegetables each day
- Consuming 400 g or more of complex carbohydrates daily
- Eating less than 80 g of red meat each day
- Consuming less than 2400 mg of sodium each day
- Low daily alcohol consumption (less than 14 g for females and less than 28 g for males).
Subjects who met at least 4 of these 7 criteria were classified as following the guidelines. And 27% of the participants—25.2% of males and 28.8% of females—were classified as such.
“These findings are important because they indicate that adults who were treated for cancer as children have the opportunity to influence their own health outcomes,” Dr Ness said.
“[A]dopting a lifestyle that includes maintaining a healthy body weight, regular physical activity, and a diet that includes fruits and vegetables and that limits refined sugars, excessive alcohol, red meat, and salt has potential to prevent development of metabolic syndrome.”

patient and her father
Credit: Rhoda Baer
Following a healthy lifestyle can decrease the risk of metabolic syndrome in childhood cancer survivors, according to a study published in Cancer.
Unfortunately, only about a quarter of the survivors studied actually practiced healthy lifestyle habits, such as engaging in moderate physical activity; eating the recommended daily serving of fruits, vegetables, and complex carbohydrates; and consuming red meat, alcohol, and sodium in moderation.
Childhood cancer survivors are known to have an increased risk of developing metabolic syndrome.
The syndrome is actually a number of conditions—high blood pressure, increased body fat, and abnormal cholesterol and glucose levels—that, when they occur together, increase a person’s risk of heart disease, stroke, and diabetes.
Kirsten Ness, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee, and her colleagues wanted to determine if lifestyle habits might affect the risk of metabolic syndrome among childhood cancer survivors.
So the team analyzed 1598 survivors who were cancer-free for at least 10 years. They had a median age of 32.7 years (range, 18.9 to 60).
The analysis showed that failure to follow healthy lifestyle guidelines roughly doubled the survivors’ risk of developing metabolic syndrome. Women had a 2.4-times greater risk, and men had a 2.2-times greater risk of the syndrome if they did not follow the guidelines.
Metabolic syndrome was present in 31.8% of the participants—32.5% of males and 31% of females.
The researchers considered a subject to have metabolic syndrome if he had or received treatment for 3 or more of the following:
- Abdominal obesity (waist circumference of > 102 cm in males and > 88 cm in females)
- Triglycerides ≥ 150 mg/dL
- High-density lipoprotein cholesterol (< 40 mg/dL in males and < 50 mg/dL in females)
- Hypertension (systolic pressure ≥ 130 mm Hg or diastolic pressure ≥ 85 mm Hg)
- Fasting plasma glucose ≥ 100 mg/dL.
Questionnaires and tests helped the researchers assess whether participants followed healthy lifestyle recommendations issued by the World Cancer Research Fund and American Institute for Cancer Research.
The recommendations include:
- Having a body mass index of 25 or lower
- Engaging in moderate physical activity for 150 minutes each week
- Eating 5 or more servings of fruits and vegetables each day
- Consuming 400 g or more of complex carbohydrates daily
- Eating less than 80 g of red meat each day
- Consuming less than 2400 mg of sodium each day
- Low daily alcohol consumption (less than 14 g for females and less than 28 g for males).
Subjects who met at least 4 of these 7 criteria were classified as following the guidelines. And 27% of the participants—25.2% of males and 28.8% of females—were classified as such.
“These findings are important because they indicate that adults who were treated for cancer as children have the opportunity to influence their own health outcomes,” Dr Ness said.
“[A]dopting a lifestyle that includes maintaining a healthy body weight, regular physical activity, and a diet that includes fruits and vegetables and that limits refined sugars, excessive alcohol, red meat, and salt has potential to prevent development of metabolic syndrome.”