User login
Histone variant may contribute to lymphoma

Image by Eric Smith
Researchers say they have identified histone chaperones that play an important role in the structure of chromatin.
The team believes this finding, published in Molecular Cell, could lead to a better understanding of lymphomas and other cancers.
“Maintaining an appropriate chromatin structure is essential for normal development, and, not surprisingly, defects in chromatin components can lead to several diseases,” said study author François Robert, PhD, of Institut de Recherches Cliniques de Montréal in Québec, Canada.
In studying chromatin, Dr Robert and his colleagues have been interested in a histone variant called H2A.Z.
The researchers knew that H2A.Z is incorporated into promoter regions of the gene by SWR-C-related chromatin remodeling complexes, but they wanted to determine if H2A.Z is actively excluded from non-promoter regions.
“With this study, we discovered that 2 other proteins, FACT and Spt6, play an important role in the location of H2A.Z,” said Célia Jeronimo, PhD, a research associate in Dr Robert’s lab.
The team found that FACT and SPt6 both help keep H2A.Z from accumulating in intragenic regions. When either histone chaperone is absent, H2A.Z is mislocalized, which alters chromatin composition and contributes to cryptic transcription.
“Inappropriate H2A.Z localization has previously been observed in cancer cells, but little was understood about the consequences of this phenomenon,” Dr Robert said.
“Although our study was performed in yeast cells, it suggests that mislocalization of H2A.Z may lead to cryptic transcription in some types of cancer such as lymphoma, and this may contribute to the disease. Our next step is therefore to investigate the possible role of H2A.Z and its associated gene expression defects in cancer cells.” ![]()

Image by Eric Smith
Researchers say they have identified histone chaperones that play an important role in the structure of chromatin.
The team believes this finding, published in Molecular Cell, could lead to a better understanding of lymphomas and other cancers.
“Maintaining an appropriate chromatin structure is essential for normal development, and, not surprisingly, defects in chromatin components can lead to several diseases,” said study author François Robert, PhD, of Institut de Recherches Cliniques de Montréal in Québec, Canada.
In studying chromatin, Dr Robert and his colleagues have been interested in a histone variant called H2A.Z.
The researchers knew that H2A.Z is incorporated into promoter regions of the gene by SWR-C-related chromatin remodeling complexes, but they wanted to determine if H2A.Z is actively excluded from non-promoter regions.
“With this study, we discovered that 2 other proteins, FACT and Spt6, play an important role in the location of H2A.Z,” said Célia Jeronimo, PhD, a research associate in Dr Robert’s lab.
The team found that FACT and SPt6 both help keep H2A.Z from accumulating in intragenic regions. When either histone chaperone is absent, H2A.Z is mislocalized, which alters chromatin composition and contributes to cryptic transcription.
“Inappropriate H2A.Z localization has previously been observed in cancer cells, but little was understood about the consequences of this phenomenon,” Dr Robert said.
“Although our study was performed in yeast cells, it suggests that mislocalization of H2A.Z may lead to cryptic transcription in some types of cancer such as lymphoma, and this may contribute to the disease. Our next step is therefore to investigate the possible role of H2A.Z and its associated gene expression defects in cancer cells.” ![]()

Image by Eric Smith
Researchers say they have identified histone chaperones that play an important role in the structure of chromatin.
The team believes this finding, published in Molecular Cell, could lead to a better understanding of lymphomas and other cancers.
“Maintaining an appropriate chromatin structure is essential for normal development, and, not surprisingly, defects in chromatin components can lead to several diseases,” said study author François Robert, PhD, of Institut de Recherches Cliniques de Montréal in Québec, Canada.
In studying chromatin, Dr Robert and his colleagues have been interested in a histone variant called H2A.Z.
The researchers knew that H2A.Z is incorporated into promoter regions of the gene by SWR-C-related chromatin remodeling complexes, but they wanted to determine if H2A.Z is actively excluded from non-promoter regions.
“With this study, we discovered that 2 other proteins, FACT and Spt6, play an important role in the location of H2A.Z,” said Célia Jeronimo, PhD, a research associate in Dr Robert’s lab.
The team found that FACT and SPt6 both help keep H2A.Z from accumulating in intragenic regions. When either histone chaperone is absent, H2A.Z is mislocalized, which alters chromatin composition and contributes to cryptic transcription.
“Inappropriate H2A.Z localization has previously been observed in cancer cells, but little was understood about the consequences of this phenomenon,” Dr Robert said.
“Although our study was performed in yeast cells, it suggests that mislocalization of H2A.Z may lead to cryptic transcription in some types of cancer such as lymphoma, and this may contribute to the disease. Our next step is therefore to investigate the possible role of H2A.Z and its associated gene expression defects in cancer cells.” ![]()
Dexrazoxane Tx did not affect overall survival in pediatric leukemia and lymphoma
Exposure to dexrazoxane among pediatric patients with leukemia or lymphoma did not affect overall mortality during a median follow-up period of 12.6 years, according to a report published online in the Journal of Clinical Oncology.
Aggregated data from three Children’s Oncology Group trials showed that among 1,008 pediatric patients who received treatment with doxorubicin with or without dexrazoxane (DRZ) from 1996 to 2001, exposure to DRZ was not associated with an increased risk of relapse (HR, 0.81; 95% CI, 0.60-1.08) or death (HR, 1.03; 0.73-1.45). Comparing DRZ with non-DRZ treatment groups at 10 years, the cumulative incidence of relapse was 16.1% vs. 19.1% (difference, – 3.0%; 95% CI, – 7.9% to 0.2%) and overall mortality was 12.8% vs. 12.2% (difference, – 0.6%; 95% CI, – 3.5% to 4.7%). The three trials (P9404, P9425, and P9426) evaluated individually likewise did not show significant differences in relapse or mortality rates.
Although studies in adults show a positive effect of DRZ on heart failure rates after anthracycline therapy, concern over DRZ interference with cancer therapies and a possible link to second cancers have limited its use in children and prompted Dr. Eric Chow of the Fred Hutchinson Cancer Research Center, Seattle, and his colleagues to assess the effect of DRZ on mortality.
The investigators wrote that DRZ “does not appear to interfere with cancer treatment efficacy, in terms of original cancer mortality or overall risk of relapse. Although the risk for secondary cancer mortality (mainly as a result of AML/MDS [acute myeloid leukemia/myelodysplastic syndrome]) was greater among those exposed to DRZ, the overall number of events was small, and the differences were not statistically significant,” the investigators said. (J. Clin. Oncol. 2015 May 26 [doi:10.1200/JCO.2014.59.4473])
Aggregated data from the three trials shows that the 10-year mortality rate of AML/MDS was 1.4% for those treated with DRZ (seven patients), compared with 0.8% for those treated without DRZ (five patients).
The beneficial effects of DRZ in decreasing the risk of heart failure have been observed in trials of adult patients, but the results for survivors of childhood cancers have been inconclusive because heart failure may develop over a longer time period in children. With the median age of survivors in this study of 24 years, significant differences in cardiac mortality due to DRZ use are not detectable. To evaluate DRZ as a cardioprotectant, a new Children’s Oncology Group study (Effects of Dexrazoxane Hydrochloride on Biomarkers Associated With Cardiomyopathy and Heart Failure After Cancer Treatment [HEART]) will determine the cardiovascular health of individuals in the three trials P9404, P9425, and P9426.
“Given that second cancers and symptomatic cardiac disease appear to be by far the two most common categories of serious late effects (in terms of both absolute and relative risks) among long-term childhood cancer survivors as a group … with cumulative incidences of each approaching 20% by age 50 years, any strategy that offers the promise of reduced cardiotoxicity without being offset by second cancers is highly attractive,” Dr. Chow and his associates wrote.
The study was supported by the National Institutes of Health, St. Baldrick’s Foundation, and the Leukemia and Lymphoma Society. Dr. Chow reported having no relevant financial conflicts. Three of his coauthors reported having financial relationships with industry.
Exposure to dexrazoxane among pediatric patients with leukemia or lymphoma did not affect overall mortality during a median follow-up period of 12.6 years, according to a report published online in the Journal of Clinical Oncology.
Aggregated data from three Children’s Oncology Group trials showed that among 1,008 pediatric patients who received treatment with doxorubicin with or without dexrazoxane (DRZ) from 1996 to 2001, exposure to DRZ was not associated with an increased risk of relapse (HR, 0.81; 95% CI, 0.60-1.08) or death (HR, 1.03; 0.73-1.45). Comparing DRZ with non-DRZ treatment groups at 10 years, the cumulative incidence of relapse was 16.1% vs. 19.1% (difference, – 3.0%; 95% CI, – 7.9% to 0.2%) and overall mortality was 12.8% vs. 12.2% (difference, – 0.6%; 95% CI, – 3.5% to 4.7%). The three trials (P9404, P9425, and P9426) evaluated individually likewise did not show significant differences in relapse or mortality rates.
Although studies in adults show a positive effect of DRZ on heart failure rates after anthracycline therapy, concern over DRZ interference with cancer therapies and a possible link to second cancers have limited its use in children and prompted Dr. Eric Chow of the Fred Hutchinson Cancer Research Center, Seattle, and his colleagues to assess the effect of DRZ on mortality.
The investigators wrote that DRZ “does not appear to interfere with cancer treatment efficacy, in terms of original cancer mortality or overall risk of relapse. Although the risk for secondary cancer mortality (mainly as a result of AML/MDS [acute myeloid leukemia/myelodysplastic syndrome]) was greater among those exposed to DRZ, the overall number of events was small, and the differences were not statistically significant,” the investigators said. (J. Clin. Oncol. 2015 May 26 [doi:10.1200/JCO.2014.59.4473])
Aggregated data from the three trials shows that the 10-year mortality rate of AML/MDS was 1.4% for those treated with DRZ (seven patients), compared with 0.8% for those treated without DRZ (five patients).
The beneficial effects of DRZ in decreasing the risk of heart failure have been observed in trials of adult patients, but the results for survivors of childhood cancers have been inconclusive because heart failure may develop over a longer time period in children. With the median age of survivors in this study of 24 years, significant differences in cardiac mortality due to DRZ use are not detectable. To evaluate DRZ as a cardioprotectant, a new Children’s Oncology Group study (Effects of Dexrazoxane Hydrochloride on Biomarkers Associated With Cardiomyopathy and Heart Failure After Cancer Treatment [HEART]) will determine the cardiovascular health of individuals in the three trials P9404, P9425, and P9426.
“Given that second cancers and symptomatic cardiac disease appear to be by far the two most common categories of serious late effects (in terms of both absolute and relative risks) among long-term childhood cancer survivors as a group … with cumulative incidences of each approaching 20% by age 50 years, any strategy that offers the promise of reduced cardiotoxicity without being offset by second cancers is highly attractive,” Dr. Chow and his associates wrote.
The study was supported by the National Institutes of Health, St. Baldrick’s Foundation, and the Leukemia and Lymphoma Society. Dr. Chow reported having no relevant financial conflicts. Three of his coauthors reported having financial relationships with industry.
Exposure to dexrazoxane among pediatric patients with leukemia or lymphoma did not affect overall mortality during a median follow-up period of 12.6 years, according to a report published online in the Journal of Clinical Oncology.
Aggregated data from three Children’s Oncology Group trials showed that among 1,008 pediatric patients who received treatment with doxorubicin with or without dexrazoxane (DRZ) from 1996 to 2001, exposure to DRZ was not associated with an increased risk of relapse (HR, 0.81; 95% CI, 0.60-1.08) or death (HR, 1.03; 0.73-1.45). Comparing DRZ with non-DRZ treatment groups at 10 years, the cumulative incidence of relapse was 16.1% vs. 19.1% (difference, – 3.0%; 95% CI, – 7.9% to 0.2%) and overall mortality was 12.8% vs. 12.2% (difference, – 0.6%; 95% CI, – 3.5% to 4.7%). The three trials (P9404, P9425, and P9426) evaluated individually likewise did not show significant differences in relapse or mortality rates.
Although studies in adults show a positive effect of DRZ on heart failure rates after anthracycline therapy, concern over DRZ interference with cancer therapies and a possible link to second cancers have limited its use in children and prompted Dr. Eric Chow of the Fred Hutchinson Cancer Research Center, Seattle, and his colleagues to assess the effect of DRZ on mortality.
The investigators wrote that DRZ “does not appear to interfere with cancer treatment efficacy, in terms of original cancer mortality or overall risk of relapse. Although the risk for secondary cancer mortality (mainly as a result of AML/MDS [acute myeloid leukemia/myelodysplastic syndrome]) was greater among those exposed to DRZ, the overall number of events was small, and the differences were not statistically significant,” the investigators said. (J. Clin. Oncol. 2015 May 26 [doi:10.1200/JCO.2014.59.4473])
Aggregated data from the three trials shows that the 10-year mortality rate of AML/MDS was 1.4% for those treated with DRZ (seven patients), compared with 0.8% for those treated without DRZ (five patients).
The beneficial effects of DRZ in decreasing the risk of heart failure have been observed in trials of adult patients, but the results for survivors of childhood cancers have been inconclusive because heart failure may develop over a longer time period in children. With the median age of survivors in this study of 24 years, significant differences in cardiac mortality due to DRZ use are not detectable. To evaluate DRZ as a cardioprotectant, a new Children’s Oncology Group study (Effects of Dexrazoxane Hydrochloride on Biomarkers Associated With Cardiomyopathy and Heart Failure After Cancer Treatment [HEART]) will determine the cardiovascular health of individuals in the three trials P9404, P9425, and P9426.
“Given that second cancers and symptomatic cardiac disease appear to be by far the two most common categories of serious late effects (in terms of both absolute and relative risks) among long-term childhood cancer survivors as a group … with cumulative incidences of each approaching 20% by age 50 years, any strategy that offers the promise of reduced cardiotoxicity without being offset by second cancers is highly attractive,” Dr. Chow and his associates wrote.
The study was supported by the National Institutes of Health, St. Baldrick’s Foundation, and the Leukemia and Lymphoma Society. Dr. Chow reported having no relevant financial conflicts. Three of his coauthors reported having financial relationships with industry.
FROM JOURNAL OF CLINICAL ONCOLOGY
Key clinical point: Treatment with dexrazoxane was not associated with an increased risk for cancer relapse or death.
Major finding: For pediatric patients with leukemia and lymphoma, the cumulative incidence of relapse at 10 years was 16.1% with DRZ, compared with 19.1% without DRZ (difference, – 3.0%; 95% CI, – 7.9% to 0.2%); overall mortality was 12.8% with DRZ vs. 12.2% without DRZ (difference, – 0.6%; 95% CI, – 3.5% to 4.7%).
Data source: Aggregated Children’s Oncology Group trials enrolling 1,008 pediatric patients with leukemia or lymphoma who were randomized to receive doxorubicin with or without DRZ from 1996 to 2001.
Disclosures: The study was supported by the National Institutes of Health, St. Baldrick’s Foundation, and the Leukemia and Lymphoma Society. Dr. Chow reported having no relevant financial conflicts. Three of his coauthors reported having financial relationships with industry.
Herbs reduce fatigue in cancer patients

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()

Photo by Alexander Baxevanis
An herbal mixture used in traditional Chinese medicine can reduce fatigue in cancer patients, results of a phase 1/2 study suggest.
The mixture, Ren Shen Yangrong Tang (RSYRT), is a soup containing 12 herbs.
In the study, cancer patients suffering from moderate to severe fatigue reported significantly less fatigue after taking RSYRT for 2 to 3 weeks.
Researchers reported these results in the Journal of Alternative and Complementary Medicine.
Yichen Xu, MD, of Beijing Cancer Hospital & Institute in China, and colleagues evaluated RSYRT in 33 patients who had completed cancer treatment. The patients had stable disease and no anemia.
Eleven patients had moderate fatigue (a score of 4-6 on a 0-10 scale), and 22 had severe fatigue (a score of 7-10). All patients had experienced fatigue for at least 4 months.
Patients took RSYRT twice a day for 6 weeks and experienced a significant decrease in fatigue severity. The mean fatigue score decreased from 7.06 at baseline to 3.30 at the 6-week mark (P<0.001).
The fatigue category also changed significantly (P=0.024). Among the 22 patients who had severe fatigue before RSYRT, half had mild fatigue after therapy, and half had moderate fatigue.
Among the 11 patients who had moderate fatigue at baseline, only 1 still had moderate fatigue after receiving RSYRT. The rest had mild fatigue.
All of the patients said they felt better after taking RSYRT for 4 weeks.
There were no “uncomfortable events” related to RSYRT, such as gastrointestinal upset, insomnia, headache, or rash. None of the patients required a dose reduction or dose interruption.
None of the patients had blood chemistry abnormalities or abnormal liver/kidney function. Two patients who had a change in ST segment before RSYRT had normal electrocardiogram results after treatment. ![]()
Inhibitor promotes chemosensitization in CLL

PHILADELPHIA—A DNA-dependent protein kinase (DNA-PK) inhibitor can sensitize chronic lymphocytic leukemia (CLL) cells to chemotherapy, according to
preclinical research.
The inhibitor, NDD0004, sensitized CLL cells—even those from patients with high-risk cytogenetics—to treatment with mitoxantrone.
However, not all CLL samples were sensitive to treatment, so researchers are now trying to determine which patients might derive benefit from DNA-PK inhibitors.
Gesa Junge, a PhD student at Newcastle University in the UK, and her colleagues conducted this research and presented the results at the AACR Annual Meeting 2015 (abstract 3624*). The work was supported by AstraZeneca.
The researchers’ goal was to validate that DNA-PK inhibition is a valid approach to chemosensitization in CLL. So the team tested NU7441—a compound that inhibits DNA-PK and PI3 kinase—and NDD0004—a more selective DNA-PK inhibitor.
The team isolated CLL cells from patients’ peripheral blood, cultured the cells, and treated them with mitoxantrone and/or 1μM of NDD0004 or 1μM of NU7441.
Junge and her colleagues found that NDD0004 sensitized cells to mitoxantrone more effectively than NU7441. Sensitization was 202-fold higher with NDD004 plus mitoxantrone than with mitoxantrone alone and 69-fold higher with NU7441 plus mitoxantrone than with mitoxantrone alone (P=0.02).
However, sensitization varied between CLL samples, and the researchers have yet to determine why. Their experiments showed that variability was not a result of DNA-PK levels.
Still, the team found that CLL cells from patients with poor prognostic markers were sensitive to DNA-PK inhibition.
Sensitization with NU7441 plus mitoxantrone was 69-fold higher than mitoxantrone alone in CLL samples with del(13q), 25-fold higher in samples with del(11q), 12-fold higher in samples with TP53 mutation, and 16-fold higher in samples with ATM dysfunction.
Sensitization with NDD0004 plus mitoxantrone was 201-fold higher than mitoxantrone alone in CLL samples with del(13q), 314-fold higher in samples with del(11q), 27-fold higher in samples with TP53 mutation, and 18-fold higher in samples with ATM dysfunction.
To confirm that sensitization was a result of DNA-PK inhibition, Junge and her colleagues tested NDD0004 in an isogenic pair of DNA-PK-deficient and DNA-PK-proficient HCT116 cells. They found that HCT116 cells lacking DNA-PK were not sensitive to NDD0004, but cells with DNA-PK were sensitive.
The researchers also investigated the mechanism of NDD0004. Their results suggest the drug works by inhibiting the repair of DNA double-strand breaks.
“What we think is happening is that we are inducing DNA damage with mitoxantrone, and that gets repaired by 24 hours,” Junge said. “But if the DNA-PK inhibitor is there, the damage persists, and that seems to translate quite nicely into an apoptosis response.”
To further this research, Junge and her colleagues are hoping to identify biomarkers that can help them determine which CLL patients are likely to respond to DNA-PK inhibitors. ![]()
*Information in the abstract differs from that presented at the meeting.

PHILADELPHIA—A DNA-dependent protein kinase (DNA-PK) inhibitor can sensitize chronic lymphocytic leukemia (CLL) cells to chemotherapy, according to
preclinical research.
The inhibitor, NDD0004, sensitized CLL cells—even those from patients with high-risk cytogenetics—to treatment with mitoxantrone.
However, not all CLL samples were sensitive to treatment, so researchers are now trying to determine which patients might derive benefit from DNA-PK inhibitors.
Gesa Junge, a PhD student at Newcastle University in the UK, and her colleagues conducted this research and presented the results at the AACR Annual Meeting 2015 (abstract 3624*). The work was supported by AstraZeneca.
The researchers’ goal was to validate that DNA-PK inhibition is a valid approach to chemosensitization in CLL. So the team tested NU7441—a compound that inhibits DNA-PK and PI3 kinase—and NDD0004—a more selective DNA-PK inhibitor.
The team isolated CLL cells from patients’ peripheral blood, cultured the cells, and treated them with mitoxantrone and/or 1μM of NDD0004 or 1μM of NU7441.
Junge and her colleagues found that NDD0004 sensitized cells to mitoxantrone more effectively than NU7441. Sensitization was 202-fold higher with NDD004 plus mitoxantrone than with mitoxantrone alone and 69-fold higher with NU7441 plus mitoxantrone than with mitoxantrone alone (P=0.02).
However, sensitization varied between CLL samples, and the researchers have yet to determine why. Their experiments showed that variability was not a result of DNA-PK levels.
Still, the team found that CLL cells from patients with poor prognostic markers were sensitive to DNA-PK inhibition.
Sensitization with NU7441 plus mitoxantrone was 69-fold higher than mitoxantrone alone in CLL samples with del(13q), 25-fold higher in samples with del(11q), 12-fold higher in samples with TP53 mutation, and 16-fold higher in samples with ATM dysfunction.
Sensitization with NDD0004 plus mitoxantrone was 201-fold higher than mitoxantrone alone in CLL samples with del(13q), 314-fold higher in samples with del(11q), 27-fold higher in samples with TP53 mutation, and 18-fold higher in samples with ATM dysfunction.
To confirm that sensitization was a result of DNA-PK inhibition, Junge and her colleagues tested NDD0004 in an isogenic pair of DNA-PK-deficient and DNA-PK-proficient HCT116 cells. They found that HCT116 cells lacking DNA-PK were not sensitive to NDD0004, but cells with DNA-PK were sensitive.
The researchers also investigated the mechanism of NDD0004. Their results suggest the drug works by inhibiting the repair of DNA double-strand breaks.
“What we think is happening is that we are inducing DNA damage with mitoxantrone, and that gets repaired by 24 hours,” Junge said. “But if the DNA-PK inhibitor is there, the damage persists, and that seems to translate quite nicely into an apoptosis response.”
To further this research, Junge and her colleagues are hoping to identify biomarkers that can help them determine which CLL patients are likely to respond to DNA-PK inhibitors. ![]()
*Information in the abstract differs from that presented at the meeting.

PHILADELPHIA—A DNA-dependent protein kinase (DNA-PK) inhibitor can sensitize chronic lymphocytic leukemia (CLL) cells to chemotherapy, according to
preclinical research.
The inhibitor, NDD0004, sensitized CLL cells—even those from patients with high-risk cytogenetics—to treatment with mitoxantrone.
However, not all CLL samples were sensitive to treatment, so researchers are now trying to determine which patients might derive benefit from DNA-PK inhibitors.
Gesa Junge, a PhD student at Newcastle University in the UK, and her colleagues conducted this research and presented the results at the AACR Annual Meeting 2015 (abstract 3624*). The work was supported by AstraZeneca.
The researchers’ goal was to validate that DNA-PK inhibition is a valid approach to chemosensitization in CLL. So the team tested NU7441—a compound that inhibits DNA-PK and PI3 kinase—and NDD0004—a more selective DNA-PK inhibitor.
The team isolated CLL cells from patients’ peripheral blood, cultured the cells, and treated them with mitoxantrone and/or 1μM of NDD0004 or 1μM of NU7441.
Junge and her colleagues found that NDD0004 sensitized cells to mitoxantrone more effectively than NU7441. Sensitization was 202-fold higher with NDD004 plus mitoxantrone than with mitoxantrone alone and 69-fold higher with NU7441 plus mitoxantrone than with mitoxantrone alone (P=0.02).
However, sensitization varied between CLL samples, and the researchers have yet to determine why. Their experiments showed that variability was not a result of DNA-PK levels.
Still, the team found that CLL cells from patients with poor prognostic markers were sensitive to DNA-PK inhibition.
Sensitization with NU7441 plus mitoxantrone was 69-fold higher than mitoxantrone alone in CLL samples with del(13q), 25-fold higher in samples with del(11q), 12-fold higher in samples with TP53 mutation, and 16-fold higher in samples with ATM dysfunction.
Sensitization with NDD0004 plus mitoxantrone was 201-fold higher than mitoxantrone alone in CLL samples with del(13q), 314-fold higher in samples with del(11q), 27-fold higher in samples with TP53 mutation, and 18-fold higher in samples with ATM dysfunction.
To confirm that sensitization was a result of DNA-PK inhibition, Junge and her colleagues tested NDD0004 in an isogenic pair of DNA-PK-deficient and DNA-PK-proficient HCT116 cells. They found that HCT116 cells lacking DNA-PK were not sensitive to NDD0004, but cells with DNA-PK were sensitive.
The researchers also investigated the mechanism of NDD0004. Their results suggest the drug works by inhibiting the repair of DNA double-strand breaks.
“What we think is happening is that we are inducing DNA damage with mitoxantrone, and that gets repaired by 24 hours,” Junge said. “But if the DNA-PK inhibitor is there, the damage persists, and that seems to translate quite nicely into an apoptosis response.”
To further this research, Junge and her colleagues are hoping to identify biomarkers that can help them determine which CLL patients are likely to respond to DNA-PK inhibitors. ![]()
*Information in the abstract differs from that presented at the meeting.
CHMP recommends drug for WM

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) is recommending that ibrutinib (Imbruvica) be approved to treat Waldenström’s macroglobulinemia (WM).
The CHMP is recommending the drug for use in WM patients who have received at least 1 prior therapy as well as previously untreated WM patients who are not suitable candidates for chemo-immunotherapy.
The European Commission will review this recommendation and should make a decision later this year.
Ibrutinib is already approved to treat WM in the US. The drug is also approved in the European Union, the US, and other countries to treat chronic lymphocytic leukemia and mantle cell lymphoma.
Janssen-Cilag International NV (Janssen) holds the marketing authorization for ibrutinib in Europe, and its affiliates market the drug in Europe and the rest of the world. In the US, ibrutinib is under joint development by Pharmacyclics and Janssen Biotech, Inc.
Phase 2 study
The CHMP’s recommendation for ibrutinib was based on a multicenter, phase 2 study in which researchers tested the drug in 63 patients with previously treated WM. Initial data showed an overall response rate of 87.3% in patients who received the drug for a median of 11.7 months.
Updated results from the study were published in NEJM in April. After a median treatment duration of 19.1 months, the overall response rate was 91%.
At 24 months, the estimated rate of progression-free survival was 69%, and the estimated rate of overall survival was 95%.
The most common grade 2-4 adverse events were neutropenia (22%) and thrombocytopenia (14%). Ibrutinib-related neutropenia and thrombocytopenia were reversible but required a dose reduction in 3 patients and treatment discontinuation in 4 patients.
Grade 2 or higher bleeding events occurred in 4 patients, and there were 15 infections considered possibly related to ibrutinib.
Treatment-related atrial fibrillation (AFib) occurred in 3 patients, all of whom had a prior history of paroxysmal AFib. AFib resolved when treatment was withheld, and all 3 patients were able to continue on therapy per protocol without an additional event. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) is recommending that ibrutinib (Imbruvica) be approved to treat Waldenström’s macroglobulinemia (WM).
The CHMP is recommending the drug for use in WM patients who have received at least 1 prior therapy as well as previously untreated WM patients who are not suitable candidates for chemo-immunotherapy.
The European Commission will review this recommendation and should make a decision later this year.
Ibrutinib is already approved to treat WM in the US. The drug is also approved in the European Union, the US, and other countries to treat chronic lymphocytic leukemia and mantle cell lymphoma.
Janssen-Cilag International NV (Janssen) holds the marketing authorization for ibrutinib in Europe, and its affiliates market the drug in Europe and the rest of the world. In the US, ibrutinib is under joint development by Pharmacyclics and Janssen Biotech, Inc.
Phase 2 study
The CHMP’s recommendation for ibrutinib was based on a multicenter, phase 2 study in which researchers tested the drug in 63 patients with previously treated WM. Initial data showed an overall response rate of 87.3% in patients who received the drug for a median of 11.7 months.
Updated results from the study were published in NEJM in April. After a median treatment duration of 19.1 months, the overall response rate was 91%.
At 24 months, the estimated rate of progression-free survival was 69%, and the estimated rate of overall survival was 95%.
The most common grade 2-4 adverse events were neutropenia (22%) and thrombocytopenia (14%). Ibrutinib-related neutropenia and thrombocytopenia were reversible but required a dose reduction in 3 patients and treatment discontinuation in 4 patients.
Grade 2 or higher bleeding events occurred in 4 patients, and there were 15 infections considered possibly related to ibrutinib.
Treatment-related atrial fibrillation (AFib) occurred in 3 patients, all of whom had a prior history of paroxysmal AFib. AFib resolved when treatment was withheld, and all 3 patients were able to continue on therapy per protocol without an additional event. ![]()

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) is recommending that ibrutinib (Imbruvica) be approved to treat Waldenström’s macroglobulinemia (WM).
The CHMP is recommending the drug for use in WM patients who have received at least 1 prior therapy as well as previously untreated WM patients who are not suitable candidates for chemo-immunotherapy.
The European Commission will review this recommendation and should make a decision later this year.
Ibrutinib is already approved to treat WM in the US. The drug is also approved in the European Union, the US, and other countries to treat chronic lymphocytic leukemia and mantle cell lymphoma.
Janssen-Cilag International NV (Janssen) holds the marketing authorization for ibrutinib in Europe, and its affiliates market the drug in Europe and the rest of the world. In the US, ibrutinib is under joint development by Pharmacyclics and Janssen Biotech, Inc.
Phase 2 study
The CHMP’s recommendation for ibrutinib was based on a multicenter, phase 2 study in which researchers tested the drug in 63 patients with previously treated WM. Initial data showed an overall response rate of 87.3% in patients who received the drug for a median of 11.7 months.
Updated results from the study were published in NEJM in April. After a median treatment duration of 19.1 months, the overall response rate was 91%.
At 24 months, the estimated rate of progression-free survival was 69%, and the estimated rate of overall survival was 95%.
The most common grade 2-4 adverse events were neutropenia (22%) and thrombocytopenia (14%). Ibrutinib-related neutropenia and thrombocytopenia were reversible but required a dose reduction in 3 patients and treatment discontinuation in 4 patients.
Grade 2 or higher bleeding events occurred in 4 patients, and there were 15 infections considered possibly related to ibrutinib.
Treatment-related atrial fibrillation (AFib) occurred in 3 patients, all of whom had a prior history of paroxysmal AFib. AFib resolved when treatment was withheld, and all 3 patients were able to continue on therapy per protocol without an additional event. ![]()
Team reports new method to identify immune cells

Photo by Graham Colm
A new method for identifying immune cells could pave the way for rapid detection of hematologic malignancies from a small blood sample, according to researchers.
The team found they could use wavelength modulated Raman spectroscopy (WMRS) to identify subsets of T cells, natural killer cells, and dendritic cells.
Traditional methods of identifying these cells usually involve labeling them with fluorescent or magnetically labeled antibodies.
Using WMRS, the researchers were able to identify immune cells with no labeling at all, thus permitting rapid identification and further analysis to take place with no potential alteration to the cells.
Simon Powis, PhD, of the University of St Andrews in Fife, Scotland, and his colleagues described this work in PLOS ONE.
Raman scattering refers to light scattering from molecules in a sample where the light energy can be shifted up or down and recorded as a “molecular fingerprint” that can be used for identification. Normally, this process is very weak and further hampered by other background light (eg, fluorescence).
WMRS subtly changes the incident laser light that, in turn, results in a modulation of the Raman signal, allowing it to be extracted from any (stationary) interfering signal.
Using WMRS, Dr Powis and his colleagues found they could identify CD4+ T cells, CD8+ T cells, CD56+ natural killer cells, CD303+ lymphoid/plasmacytoid dendritic cells, and CD1c+ myeloid dendritic cells.
“Under a normal light microscope, these immune cells essentially all look identical,” Dr Powis said. “With this new method, we can identify key cell types without any labeling.”
“Our next goal is to make a full catalogue of all the normal cell types of the immune system that can be detected in the bloodstream. Once we have this completed, we can then collaborate with our clinical colleagues to start identifying when these immune cells are altered, in conditions such as leukemia and lymphoma, potentially providing a rapid detection system from just a small blood sample.” ![]()

Photo by Graham Colm
A new method for identifying immune cells could pave the way for rapid detection of hematologic malignancies from a small blood sample, according to researchers.
The team found they could use wavelength modulated Raman spectroscopy (WMRS) to identify subsets of T cells, natural killer cells, and dendritic cells.
Traditional methods of identifying these cells usually involve labeling them with fluorescent or magnetically labeled antibodies.
Using WMRS, the researchers were able to identify immune cells with no labeling at all, thus permitting rapid identification and further analysis to take place with no potential alteration to the cells.
Simon Powis, PhD, of the University of St Andrews in Fife, Scotland, and his colleagues described this work in PLOS ONE.
Raman scattering refers to light scattering from molecules in a sample where the light energy can be shifted up or down and recorded as a “molecular fingerprint” that can be used for identification. Normally, this process is very weak and further hampered by other background light (eg, fluorescence).
WMRS subtly changes the incident laser light that, in turn, results in a modulation of the Raman signal, allowing it to be extracted from any (stationary) interfering signal.
Using WMRS, Dr Powis and his colleagues found they could identify CD4+ T cells, CD8+ T cells, CD56+ natural killer cells, CD303+ lymphoid/plasmacytoid dendritic cells, and CD1c+ myeloid dendritic cells.
“Under a normal light microscope, these immune cells essentially all look identical,” Dr Powis said. “With this new method, we can identify key cell types without any labeling.”
“Our next goal is to make a full catalogue of all the normal cell types of the immune system that can be detected in the bloodstream. Once we have this completed, we can then collaborate with our clinical colleagues to start identifying when these immune cells are altered, in conditions such as leukemia and lymphoma, potentially providing a rapid detection system from just a small blood sample.” ![]()

Photo by Graham Colm
A new method for identifying immune cells could pave the way for rapid detection of hematologic malignancies from a small blood sample, according to researchers.
The team found they could use wavelength modulated Raman spectroscopy (WMRS) to identify subsets of T cells, natural killer cells, and dendritic cells.
Traditional methods of identifying these cells usually involve labeling them with fluorescent or magnetically labeled antibodies.
Using WMRS, the researchers were able to identify immune cells with no labeling at all, thus permitting rapid identification and further analysis to take place with no potential alteration to the cells.
Simon Powis, PhD, of the University of St Andrews in Fife, Scotland, and his colleagues described this work in PLOS ONE.
Raman scattering refers to light scattering from molecules in a sample where the light energy can be shifted up or down and recorded as a “molecular fingerprint” that can be used for identification. Normally, this process is very weak and further hampered by other background light (eg, fluorescence).
WMRS subtly changes the incident laser light that, in turn, results in a modulation of the Raman signal, allowing it to be extracted from any (stationary) interfering signal.
Using WMRS, Dr Powis and his colleagues found they could identify CD4+ T cells, CD8+ T cells, CD56+ natural killer cells, CD303+ lymphoid/plasmacytoid dendritic cells, and CD1c+ myeloid dendritic cells.
“Under a normal light microscope, these immune cells essentially all look identical,” Dr Powis said. “With this new method, we can identify key cell types without any labeling.”
“Our next goal is to make a full catalogue of all the normal cell types of the immune system that can be detected in the bloodstream. Once we have this completed, we can then collaborate with our clinical colleagues to start identifying when these immune cells are altered, in conditions such as leukemia and lymphoma, potentially providing a rapid detection system from just a small blood sample.” ![]()
CTLs captured on video destroying cancer cells
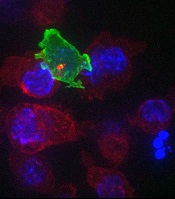
cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()

cancer cells (blue)
Image courtesy of Gillian
Griffiths and Jonny Settle
New research has illuminated the behavior of cytotoxic T cells (CTLs) as they hunt down and eliminate cancer cells.
Investigators used novel imaging techniques to capture the process on film and described their findings in an article published in Immunity.
The team captured the footage through high-resolution, 3D, time-lapse, multi-color imaging, making use of both spinning disk confocal microscopy and lattice light sheet microscopy.
These techniques involve capturing “slices” of an object and “stitching” them together to provide the final 3D images across the whole cell.
These approaches allowed the investigators to determine the order of events leading to the lethal “hit” CTLs deliver to cancer cells.
“Inside all of us lurks an army of ‘serial killers’ whose primary function is to kill again and again,” said study author Gillian Griffiths, PhD, of the Cambridge Institute for Medical Research in the UK.
“These [CTLs] patrol our bodies, identifying and destroying virally infected and cancer cells, and they do so with remarkable precision and efficiency.”
The CTLs, seen in the video as orange or green amorphous “blobs,” move around rapidly, investigating their environment as they travel.
When a CTL finds a cancer cell (blue), membrane protrusions rapidly explore the surface of the cell, checking for tell-tale signs that this is an uninvited guest.
The CTL binds to the cancer cell and injects cytotoxins (red) down microtubules to the interface between the T cell and the cancer cell, before puncturing the surface of the cancer cell and delivering its deadly cargo.
“In our bodies, where cells are packed together, it’s essential that the T cell focuses the lethal hit on its target,” Dr Griffiths explained. “Otherwise, it will cause collateral damage to neighboring, healthy cells.”
“Once the cytotoxins are injected into the cancer cell, its fate is sealed, and we can watch as it withers and dies. The T cell then moves on, hungry to find another victim.”
The investigators’ video is available on YouTube. ![]()
Combo targets AML, BL in the same way

Image by Ed Uthman
Combining a cholesterol-lowering drug and a contraceptive steroid could be a safe, effective treatment for leukemias, lymphomas, and other malignancies, according to researchers.
Their work helps explain how this combination treatment, bezafibrate and medroxyprogesterone acetate (BaP), kills cancer cells.
The team discovered that BaP’s mechanism of action is the same in acute myeloid leukemia (AML) and Burkitt lymphoma (BL) cells.
The findings have been published in Cancer Research.
BaP has been shown to prolong survival in early stage trials of elderly AML patients, when compared to standard palliative care. BaP has also been used alongside chemotherapy to successfully treat children with BL.
However, it was unclear whether BaP’s activity against these 2 very different malignancies was mediated by a common mechanism or by different effects in each cancer.
To gain some insight, Andrew Southam, PhD, of the University of Birmingham in the UK, and his colleagues investigated the drugs’ effects on the metabolism and chemical make-up of AML and BL cells.
They found that, in both cell types, BaP blocks stearoyl CoA desaturase, an enzyme crucial to the production of fatty acids, which cancer cells need to grow and multiply. The team also showed that BaP’s ability to deactivate stearoyl CoA desaturase was what prompted the cancer cells to die.
“Developing drugs to target the fatty-acid building blocks of cancer cells has been a promising area of research in recent years,” Dr Southam said. “It is very exciting we have identified these non-toxic drugs already sitting on pharmacy shelves.”
He and his colleagues believe these findings also open up the possibility that BaP could be used to treat other cancers that rely on high levels of stearoyl CoA desaturase to grow, including chronic lymphocytic leukemia and some types of non-Hodgkin lymphoma, as well as prostate, colon, and esophageal cancers.
“This drug combination shows real promise,” said Chris Bunce, PhD, also of the University of Birmingham.
“Affordable, effective, non-toxic treatments that extend survival, while offering a good quality of life, are in demand for almost all types of cancer.” ![]()

Image by Ed Uthman
Combining a cholesterol-lowering drug and a contraceptive steroid could be a safe, effective treatment for leukemias, lymphomas, and other malignancies, according to researchers.
Their work helps explain how this combination treatment, bezafibrate and medroxyprogesterone acetate (BaP), kills cancer cells.
The team discovered that BaP’s mechanism of action is the same in acute myeloid leukemia (AML) and Burkitt lymphoma (BL) cells.
The findings have been published in Cancer Research.
BaP has been shown to prolong survival in early stage trials of elderly AML patients, when compared to standard palliative care. BaP has also been used alongside chemotherapy to successfully treat children with BL.
However, it was unclear whether BaP’s activity against these 2 very different malignancies was mediated by a common mechanism or by different effects in each cancer.
To gain some insight, Andrew Southam, PhD, of the University of Birmingham in the UK, and his colleagues investigated the drugs’ effects on the metabolism and chemical make-up of AML and BL cells.
They found that, in both cell types, BaP blocks stearoyl CoA desaturase, an enzyme crucial to the production of fatty acids, which cancer cells need to grow and multiply. The team also showed that BaP’s ability to deactivate stearoyl CoA desaturase was what prompted the cancer cells to die.
“Developing drugs to target the fatty-acid building blocks of cancer cells has been a promising area of research in recent years,” Dr Southam said. “It is very exciting we have identified these non-toxic drugs already sitting on pharmacy shelves.”
He and his colleagues believe these findings also open up the possibility that BaP could be used to treat other cancers that rely on high levels of stearoyl CoA desaturase to grow, including chronic lymphocytic leukemia and some types of non-Hodgkin lymphoma, as well as prostate, colon, and esophageal cancers.
“This drug combination shows real promise,” said Chris Bunce, PhD, also of the University of Birmingham.
“Affordable, effective, non-toxic treatments that extend survival, while offering a good quality of life, are in demand for almost all types of cancer.” ![]()

Image by Ed Uthman
Combining a cholesterol-lowering drug and a contraceptive steroid could be a safe, effective treatment for leukemias, lymphomas, and other malignancies, according to researchers.
Their work helps explain how this combination treatment, bezafibrate and medroxyprogesterone acetate (BaP), kills cancer cells.
The team discovered that BaP’s mechanism of action is the same in acute myeloid leukemia (AML) and Burkitt lymphoma (BL) cells.
The findings have been published in Cancer Research.
BaP has been shown to prolong survival in early stage trials of elderly AML patients, when compared to standard palliative care. BaP has also been used alongside chemotherapy to successfully treat children with BL.
However, it was unclear whether BaP’s activity against these 2 very different malignancies was mediated by a common mechanism or by different effects in each cancer.
To gain some insight, Andrew Southam, PhD, of the University of Birmingham in the UK, and his colleagues investigated the drugs’ effects on the metabolism and chemical make-up of AML and BL cells.
They found that, in both cell types, BaP blocks stearoyl CoA desaturase, an enzyme crucial to the production of fatty acids, which cancer cells need to grow and multiply. The team also showed that BaP’s ability to deactivate stearoyl CoA desaturase was what prompted the cancer cells to die.
“Developing drugs to target the fatty-acid building blocks of cancer cells has been a promising area of research in recent years,” Dr Southam said. “It is very exciting we have identified these non-toxic drugs already sitting on pharmacy shelves.”
He and his colleagues believe these findings also open up the possibility that BaP could be used to treat other cancers that rely on high levels of stearoyl CoA desaturase to grow, including chronic lymphocytic leukemia and some types of non-Hodgkin lymphoma, as well as prostate, colon, and esophageal cancers.
“This drug combination shows real promise,” said Chris Bunce, PhD, also of the University of Birmingham.
“Affordable, effective, non-toxic treatments that extend survival, while offering a good quality of life, are in demand for almost all types of cancer.”
How mAbs produce lasting antitumor effects

Photo by Linda Bartlett
Results of preclinical research help explain how antitumor monoclonal antibodies (mAbs) fight lymphoma.
Researchers uncovered a 2-step process that revolves around 2 antibody-binding receptors found on different types of immune cells.
Experiments suggested that these Fc receptors are needed to eradicate lymphoma and ensure it doesn’t return.
The researchers reported these findings in an article published in Cell.
“These findings suggests ways current anticancer antibody treatments might be improved, as well as combined with other immune-system-stimulating therapies to help cancer patients,” said study author Jeffrey Ravetch, MD, PhD, of The Rockefeller University in New York, New York.
Previous research has shown that antitumor mAbs bind to Fc receptors on activated immune cells, prompting those immune cells to kill tumors.
However, it was unclear which Fc receptors are involved or how the tumor killing led the immune system to generate memory T cells against these same antigens, in case the tumor producing them should return.
Dr Ravetch and David DiLillo, PhD, also of The Rockefeller University, investigated this process by injecting CD20-expressing lymphoma cells into mice with immune systems engineered to contain human Fc receptors, treating the mice with anti-CD20 mAbs, and then re-introducing lymphoma.
Wild-type C57BL/6 mice received syngeneic EL4 lymphoma cells expressing human CD20 (EL4-hCD20 cells). When these mice received treatment with an mIgG2a isotype anti-hCD20 mAb, they all survived.
Ninety days later, after the mAb had been cleared from their systems, the mice were re-challenged with EL4-hCD20 tumor cells, at a dose 10-fold greater than the initial challenge, but they did not receive any additional mAb.
All of these mice survived, but tumor/mAb-primed mice that were re-challenged with EL4-wild-type cells, which don’t express hCD20, had poor survival. Results were similar with a different anti-hCD20 mAb, clone 2B8.
The researchers also re-challenged tumor/mAb-primed mice with B6BL lymphoma cells that expressed either hCD20 or an irrelevant antigen, mCD20. All of the mice re-challenged with B6BL-mCD20 cells had died by day 31, but 80% of the mice re-challenged with B6BL-hCD20 cells survived at least 90 days.
Drs Ravetch and DiLillo said these results suggest an anti-hCD20 immune response is generated after the initial FcγR-mediated clearance of tumor cells by antibody-dependent cellular cytotoxicity.
The researchers then took a closer look at the role of Fc receptors, keeping in mind that different types of immune cells can express different receptors.
Based on the cells the researchers thought were involved, they looked to the Fc receptors expressed by cytotoxic immune cells that carried out the initial attack on tumors, as well as the Fc receptors found on dendritic cells, which are crucial to the formation of memory T cells.
To test the involvement of these receptors, the pair altered the mAbs delivered to the lymphoma-infected mice so as to change their affinity for the receptors. Then, they looked for changes in the survival rate of the mice after the first and second challenges with lymphoma cells.
When they dissected this process, the researchers uncovered 2 steps. The Fc receptor FcγRIIIA, which is found on macrophages, responded to mAbs and prompted the macrophages to engulf and destroy the antibody-laden tumor cells.
These same antibodies, still attached to tumor antigens, activated a second receptor, FcγRIIA, on dendritic cells, which used the antigen to prime T cells. The result was the generation of a T-cell memory response that protected the mice against future lymphoma cells expressing CD20.
“By engineering the antibodies so as to increase their affinity for both FcγRIIIA and FcγRIIA, we were able to optimize both steps in this process,” Dr DiLillo said.
“Current antibody therapies are only engineered to improve the immediate killing of tumor cells but not the formation of immunological memory. We are proposing that an ideal antibody therapy would be engineered to take full advantage of both steps.”

Photo by Linda Bartlett
Results of preclinical research help explain how antitumor monoclonal antibodies (mAbs) fight lymphoma.
Researchers uncovered a 2-step process that revolves around 2 antibody-binding receptors found on different types of immune cells.
Experiments suggested that these Fc receptors are needed to eradicate lymphoma and ensure it doesn’t return.
The researchers reported these findings in an article published in Cell.
“These findings suggests ways current anticancer antibody treatments might be improved, as well as combined with other immune-system-stimulating therapies to help cancer patients,” said study author Jeffrey Ravetch, MD, PhD, of The Rockefeller University in New York, New York.
Previous research has shown that antitumor mAbs bind to Fc receptors on activated immune cells, prompting those immune cells to kill tumors.
However, it was unclear which Fc receptors are involved or how the tumor killing led the immune system to generate memory T cells against these same antigens, in case the tumor producing them should return.
Dr Ravetch and David DiLillo, PhD, also of The Rockefeller University, investigated this process by injecting CD20-expressing lymphoma cells into mice with immune systems engineered to contain human Fc receptors, treating the mice with anti-CD20 mAbs, and then re-introducing lymphoma.
Wild-type C57BL/6 mice received syngeneic EL4 lymphoma cells expressing human CD20 (EL4-hCD20 cells). When these mice received treatment with an mIgG2a isotype anti-hCD20 mAb, they all survived.
Ninety days later, after the mAb had been cleared from their systems, the mice were re-challenged with EL4-hCD20 tumor cells, at a dose 10-fold greater than the initial challenge, but they did not receive any additional mAb.
All of these mice survived, but tumor/mAb-primed mice that were re-challenged with EL4-wild-type cells, which don’t express hCD20, had poor survival. Results were similar with a different anti-hCD20 mAb, clone 2B8.
The researchers also re-challenged tumor/mAb-primed mice with B6BL lymphoma cells that expressed either hCD20 or an irrelevant antigen, mCD20. All of the mice re-challenged with B6BL-mCD20 cells had died by day 31, but 80% of the mice re-challenged with B6BL-hCD20 cells survived at least 90 days.
Drs Ravetch and DiLillo said these results suggest an anti-hCD20 immune response is generated after the initial FcγR-mediated clearance of tumor cells by antibody-dependent cellular cytotoxicity.
The researchers then took a closer look at the role of Fc receptors, keeping in mind that different types of immune cells can express different receptors.
Based on the cells the researchers thought were involved, they looked to the Fc receptors expressed by cytotoxic immune cells that carried out the initial attack on tumors, as well as the Fc receptors found on dendritic cells, which are crucial to the formation of memory T cells.
To test the involvement of these receptors, the pair altered the mAbs delivered to the lymphoma-infected mice so as to change their affinity for the receptors. Then, they looked for changes in the survival rate of the mice after the first and second challenges with lymphoma cells.
When they dissected this process, the researchers uncovered 2 steps. The Fc receptor FcγRIIIA, which is found on macrophages, responded to mAbs and prompted the macrophages to engulf and destroy the antibody-laden tumor cells.
These same antibodies, still attached to tumor antigens, activated a second receptor, FcγRIIA, on dendritic cells, which used the antigen to prime T cells. The result was the generation of a T-cell memory response that protected the mice against future lymphoma cells expressing CD20.
“By engineering the antibodies so as to increase their affinity for both FcγRIIIA and FcγRIIA, we were able to optimize both steps in this process,” Dr DiLillo said.
“Current antibody therapies are only engineered to improve the immediate killing of tumor cells but not the formation of immunological memory. We are proposing that an ideal antibody therapy would be engineered to take full advantage of both steps.”

Photo by Linda Bartlett
Results of preclinical research help explain how antitumor monoclonal antibodies (mAbs) fight lymphoma.
Researchers uncovered a 2-step process that revolves around 2 antibody-binding receptors found on different types of immune cells.
Experiments suggested that these Fc receptors are needed to eradicate lymphoma and ensure it doesn’t return.
The researchers reported these findings in an article published in Cell.
“These findings suggests ways current anticancer antibody treatments might be improved, as well as combined with other immune-system-stimulating therapies to help cancer patients,” said study author Jeffrey Ravetch, MD, PhD, of The Rockefeller University in New York, New York.
Previous research has shown that antitumor mAbs bind to Fc receptors on activated immune cells, prompting those immune cells to kill tumors.
However, it was unclear which Fc receptors are involved or how the tumor killing led the immune system to generate memory T cells against these same antigens, in case the tumor producing them should return.
Dr Ravetch and David DiLillo, PhD, also of The Rockefeller University, investigated this process by injecting CD20-expressing lymphoma cells into mice with immune systems engineered to contain human Fc receptors, treating the mice with anti-CD20 mAbs, and then re-introducing lymphoma.
Wild-type C57BL/6 mice received syngeneic EL4 lymphoma cells expressing human CD20 (EL4-hCD20 cells). When these mice received treatment with an mIgG2a isotype anti-hCD20 mAb, they all survived.
Ninety days later, after the mAb had been cleared from their systems, the mice were re-challenged with EL4-hCD20 tumor cells, at a dose 10-fold greater than the initial challenge, but they did not receive any additional mAb.
All of these mice survived, but tumor/mAb-primed mice that were re-challenged with EL4-wild-type cells, which don’t express hCD20, had poor survival. Results were similar with a different anti-hCD20 mAb, clone 2B8.
The researchers also re-challenged tumor/mAb-primed mice with B6BL lymphoma cells that expressed either hCD20 or an irrelevant antigen, mCD20. All of the mice re-challenged with B6BL-mCD20 cells had died by day 31, but 80% of the mice re-challenged with B6BL-hCD20 cells survived at least 90 days.
Drs Ravetch and DiLillo said these results suggest an anti-hCD20 immune response is generated after the initial FcγR-mediated clearance of tumor cells by antibody-dependent cellular cytotoxicity.
The researchers then took a closer look at the role of Fc receptors, keeping in mind that different types of immune cells can express different receptors.
Based on the cells the researchers thought were involved, they looked to the Fc receptors expressed by cytotoxic immune cells that carried out the initial attack on tumors, as well as the Fc receptors found on dendritic cells, which are crucial to the formation of memory T cells.
To test the involvement of these receptors, the pair altered the mAbs delivered to the lymphoma-infected mice so as to change their affinity for the receptors. Then, they looked for changes in the survival rate of the mice after the first and second challenges with lymphoma cells.
When they dissected this process, the researchers uncovered 2 steps. The Fc receptor FcγRIIIA, which is found on macrophages, responded to mAbs and prompted the macrophages to engulf and destroy the antibody-laden tumor cells.
These same antibodies, still attached to tumor antigens, activated a second receptor, FcγRIIA, on dendritic cells, which used the antigen to prime T cells. The result was the generation of a T-cell memory response that protected the mice against future lymphoma cells expressing CD20.
“By engineering the antibodies so as to increase their affinity for both FcγRIIIA and FcγRIIA, we were able to optimize both steps in this process,” Dr DiLillo said.
“Current antibody therapies are only engineered to improve the immediate killing of tumor cells but not the formation of immunological memory. We are proposing that an ideal antibody therapy would be engineered to take full advantage of both steps.”
Company stops phase 3 PTCL trial

for use in a clinical trial
Photo by Esther Dyson
Takeda Pharmaceutical Company Limited has announced its decision to discontinue its phase 3 trial of the aurora A kinase inhibitor alisertib (MLN8237) in patients with relapsed or refractory peripheral T-cell lymphoma (PTCL).
Results of a pre-specified interim analysis indicated that alisertib was unlikely to meet the study’s primary endpoint: providing superior progression-free survival over the standard of care for PTCL.
Takeda said patients enrolled in the trial may continue to receive alisertib if they are thought to be benefitting from it and no safety concerns are present.
The company is encouraging patients to consult their study investigators to address any questions and before making any changes to their medication.
Takeda is working with trial investigators and local regulatory authorities to ensure that PTCL patients who participated in the study receive appropriate care.
The company is still investigating alisertib for use in small-cell lung cancer.
“While we are disappointed that alisertib will not be further investigated for relapsed or refractory peripheral T-cell lymphoma, we are optimistic about alisertib’s clinical development program in small-cell lung cancer,” said Michael Vasconcelles, MD, global head of the Takeda Oncology Therapeutic Unit.
“The randomized, phase 2 study of alisertib in small-cell lung cancer will continue as planned and is currently underway. Takeda also continues to support investigator-initiated research with alisertib and will evaluate its potential use in other oncology indications going forward.”

for use in a clinical trial
Photo by Esther Dyson
Takeda Pharmaceutical Company Limited has announced its decision to discontinue its phase 3 trial of the aurora A kinase inhibitor alisertib (MLN8237) in patients with relapsed or refractory peripheral T-cell lymphoma (PTCL).
Results of a pre-specified interim analysis indicated that alisertib was unlikely to meet the study’s primary endpoint: providing superior progression-free survival over the standard of care for PTCL.
Takeda said patients enrolled in the trial may continue to receive alisertib if they are thought to be benefitting from it and no safety concerns are present.
The company is encouraging patients to consult their study investigators to address any questions and before making any changes to their medication.
Takeda is working with trial investigators and local regulatory authorities to ensure that PTCL patients who participated in the study receive appropriate care.
The company is still investigating alisertib for use in small-cell lung cancer.
“While we are disappointed that alisertib will not be further investigated for relapsed or refractory peripheral T-cell lymphoma, we are optimistic about alisertib’s clinical development program in small-cell lung cancer,” said Michael Vasconcelles, MD, global head of the Takeda Oncology Therapeutic Unit.
“The randomized, phase 2 study of alisertib in small-cell lung cancer will continue as planned and is currently underway. Takeda also continues to support investigator-initiated research with alisertib and will evaluate its potential use in other oncology indications going forward.”

for use in a clinical trial
Photo by Esther Dyson
Takeda Pharmaceutical Company Limited has announced its decision to discontinue its phase 3 trial of the aurora A kinase inhibitor alisertib (MLN8237) in patients with relapsed or refractory peripheral T-cell lymphoma (PTCL).
Results of a pre-specified interim analysis indicated that alisertib was unlikely to meet the study’s primary endpoint: providing superior progression-free survival over the standard of care for PTCL.
Takeda said patients enrolled in the trial may continue to receive alisertib if they are thought to be benefitting from it and no safety concerns are present.
The company is encouraging patients to consult their study investigators to address any questions and before making any changes to their medication.
Takeda is working with trial investigators and local regulatory authorities to ensure that PTCL patients who participated in the study receive appropriate care.
The company is still investigating alisertib for use in small-cell lung cancer.
“While we are disappointed that alisertib will not be further investigated for relapsed or refractory peripheral T-cell lymphoma, we are optimistic about alisertib’s clinical development program in small-cell lung cancer,” said Michael Vasconcelles, MD, global head of the Takeda Oncology Therapeutic Unit.
“The randomized, phase 2 study of alisertib in small-cell lung cancer will continue as planned and is currently underway. Takeda also continues to support investigator-initiated research with alisertib and will evaluate its potential use in other oncology indications going forward.”