User login
New anticancer drugs linked to increased costs, life expectancy
New anticancer drugs are often expensive and have been accompanied by large increases in the cost of medical treatment, but they also are associated with gains in life expectancy, according to an analysis of Medicare data published online.
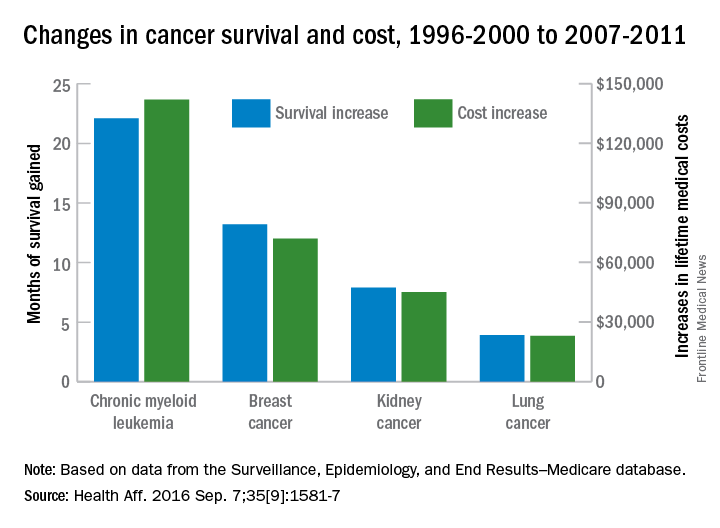
Investigators looked at four different types of cancer – breast, kidney, lung, and chronic myeloid leukemia (CML) – over two time periods: 1996-2000 and 2007-2011. Patients treated for CML during 2007-2011 had the largest increases in both average lifetime medical cost ($142,000) and months of life gained (22.1) over those treated during 1996-2000, reported David H. Howard, PhD, of Emory University, Atlanta, and his associates.
Breast cancer patients had the next-largest increases: 13.2 months of life expectancy and $72,000 in lifetime medical cost for those who received physician-administered intravenous drugs. For breast cancer patients who received only oral drugs, the increases were 2 months of life and $9,000 in lifetime cost, they noted.
Patients with kidney cancer had an average life-expectancy increase of 7.9 months and a cost increase of $45,000, but those estimates don’t fully reflect the effect of several oral drugs that were introduced after 2007 but did not come into widespread use during the entire study period, Dr. Howard and his associates noted (Health Aff. 2016 Sep 7;35[9]:1581-7).
Lung cancer patients experienced the smallest changes between the two time periods, with an increase in life expectancy of 3.9 months for those who received physician-administered anticancer drugs and a lifetime medical cost increase of $23,000. Patients with lung cancer who did not receive such drugs had increases of 0.7 months of life expectancy and $4,000 in lifetime medical costs.
The researchers used data from the Surveillance, Epidemiology, and End Results–Medicare database, and all costs are adjusted to 2012 dollars. Data collection was supported by the California Department of Health and funding for the study was provided by Pfizer. Three of Dr. Howard’s five coinvestigators are Pfizer employees.
New anticancer drugs are often expensive and have been accompanied by large increases in the cost of medical treatment, but they also are associated with gains in life expectancy, according to an analysis of Medicare data published online.
Investigators looked at four different types of cancer – breast, kidney, lung, and chronic myeloid leukemia (CML) – over two time periods: 1996-2000 and 2007-2011. Patients treated for CML during 2007-2011 had the largest increases in both average lifetime medical cost ($142,000) and months of life gained (22.1) over those treated during 1996-2000, reported David H. Howard, PhD, of Emory University, Atlanta, and his associates.
Breast cancer patients had the next-largest increases: 13.2 months of life expectancy and $72,000 in lifetime medical cost for those who received physician-administered intravenous drugs. For breast cancer patients who received only oral drugs, the increases were 2 months of life and $9,000 in lifetime cost, they noted.
Patients with kidney cancer had an average life-expectancy increase of 7.9 months and a cost increase of $45,000, but those estimates don’t fully reflect the effect of several oral drugs that were introduced after 2007 but did not come into widespread use during the entire study period, Dr. Howard and his associates noted (Health Aff. 2016 Sep 7;35[9]:1581-7).
Lung cancer patients experienced the smallest changes between the two time periods, with an increase in life expectancy of 3.9 months for those who received physician-administered anticancer drugs and a lifetime medical cost increase of $23,000. Patients with lung cancer who did not receive such drugs had increases of 0.7 months of life expectancy and $4,000 in lifetime medical costs.
The researchers used data from the Surveillance, Epidemiology, and End Results–Medicare database, and all costs are adjusted to 2012 dollars. Data collection was supported by the California Department of Health and funding for the study was provided by Pfizer. Three of Dr. Howard’s five coinvestigators are Pfizer employees.
New anticancer drugs are often expensive and have been accompanied by large increases in the cost of medical treatment, but they also are associated with gains in life expectancy, according to an analysis of Medicare data published online.
Investigators looked at four different types of cancer – breast, kidney, lung, and chronic myeloid leukemia (CML) – over two time periods: 1996-2000 and 2007-2011. Patients treated for CML during 2007-2011 had the largest increases in both average lifetime medical cost ($142,000) and months of life gained (22.1) over those treated during 1996-2000, reported David H. Howard, PhD, of Emory University, Atlanta, and his associates.
Breast cancer patients had the next-largest increases: 13.2 months of life expectancy and $72,000 in lifetime medical cost for those who received physician-administered intravenous drugs. For breast cancer patients who received only oral drugs, the increases were 2 months of life and $9,000 in lifetime cost, they noted.
Patients with kidney cancer had an average life-expectancy increase of 7.9 months and a cost increase of $45,000, but those estimates don’t fully reflect the effect of several oral drugs that were introduced after 2007 but did not come into widespread use during the entire study period, Dr. Howard and his associates noted (Health Aff. 2016 Sep 7;35[9]:1581-7).
Lung cancer patients experienced the smallest changes between the two time periods, with an increase in life expectancy of 3.9 months for those who received physician-administered anticancer drugs and a lifetime medical cost increase of $23,000. Patients with lung cancer who did not receive such drugs had increases of 0.7 months of life expectancy and $4,000 in lifetime medical costs.
The researchers used data from the Surveillance, Epidemiology, and End Results–Medicare database, and all costs are adjusted to 2012 dollars. Data collection was supported by the California Department of Health and funding for the study was provided by Pfizer. Three of Dr. Howard’s five coinvestigators are Pfizer employees.
FROM HEALTH AFFAIRS
Study reveals barriers to accessing palliative care services

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—Patients may face challenges when trying to access palliative and supportive care services at cancer centers, a new study suggests.
Researchers took a “mystery shopper” approach and placed calls to cancer centers inquiring about palliative and supportive care services for a family member.
The callers sometimes had difficulty obtaining information about these services, even though all of the centers offer them.
“It’s sobering to hear that such services are not readily accessible at many centers,” said study investigator Kathryn Hutchins, a medical student at Duke University in Durham, North Carolina.
“However, it provides an opportunity for cancer centers to empower their front-line staff, as well as the oncology care team, through education and training so that the entire enterprise has a common understanding of palliative care and how to access it.”
Hutchins and her colleagues presented this research at the 2016 Palliative Care in Oncology Symposium (abstract 122).
The researchers placed 160 calls to 40 major cancer centers. The team chose to focus on National Cancer Institute-designated cancer centers because they all provide palliative care services along with other supportive care services.
The researchers used the same script for every call, asking about services for a 58-year-old female who was recently diagnosed with inoperable liver cancer. The team called each center 4 times on different days.
In 38.2% of the calls, the researchers were not able to receive complete information about supportive care services.
In 9.5% of calls, cancer center staff gave an answer other than “yes” regarding the availability of palliative care services, even though such services were available.
The answers varied and included responses such as:
- Palliative care was for end-of-life patients only (n=2)
- No physicians specialized in symptom management (n=3)
- A medical record review would be needed first (n=2).
In addition, 10 staff members said they were unsure about the availability of palliative care, and 2 were unfamiliar with the term.
Overall, 37.6% of the callers were told that all 7 supportive care services they inquired about were offered.
“As oncologists, we like to believe that, when we refer patients to our institution’s helpline, they will get connected to the services they need, but that doesn’t always happen,” said study investigator Arif Kamal, MD, of Duke Cancer Institute.
“It’s important for oncologists to be aware of these barriers and to work to eliminate them.” ![]()

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—Patients may face challenges when trying to access palliative and supportive care services at cancer centers, a new study suggests.
Researchers took a “mystery shopper” approach and placed calls to cancer centers inquiring about palliative and supportive care services for a family member.
The callers sometimes had difficulty obtaining information about these services, even though all of the centers offer them.
“It’s sobering to hear that such services are not readily accessible at many centers,” said study investigator Kathryn Hutchins, a medical student at Duke University in Durham, North Carolina.
“However, it provides an opportunity for cancer centers to empower their front-line staff, as well as the oncology care team, through education and training so that the entire enterprise has a common understanding of palliative care and how to access it.”
Hutchins and her colleagues presented this research at the 2016 Palliative Care in Oncology Symposium (abstract 122).
The researchers placed 160 calls to 40 major cancer centers. The team chose to focus on National Cancer Institute-designated cancer centers because they all provide palliative care services along with other supportive care services.
The researchers used the same script for every call, asking about services for a 58-year-old female who was recently diagnosed with inoperable liver cancer. The team called each center 4 times on different days.
In 38.2% of the calls, the researchers were not able to receive complete information about supportive care services.
In 9.5% of calls, cancer center staff gave an answer other than “yes” regarding the availability of palliative care services, even though such services were available.
The answers varied and included responses such as:
- Palliative care was for end-of-life patients only (n=2)
- No physicians specialized in symptom management (n=3)
- A medical record review would be needed first (n=2).
In addition, 10 staff members said they were unsure about the availability of palliative care, and 2 were unfamiliar with the term.
Overall, 37.6% of the callers were told that all 7 supportive care services they inquired about were offered.
“As oncologists, we like to believe that, when we refer patients to our institution’s helpline, they will get connected to the services they need, but that doesn’t always happen,” said study investigator Arif Kamal, MD, of Duke Cancer Institute.
“It’s important for oncologists to be aware of these barriers and to work to eliminate them.” ![]()

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—Patients may face challenges when trying to access palliative and supportive care services at cancer centers, a new study suggests.
Researchers took a “mystery shopper” approach and placed calls to cancer centers inquiring about palliative and supportive care services for a family member.
The callers sometimes had difficulty obtaining information about these services, even though all of the centers offer them.
“It’s sobering to hear that such services are not readily accessible at many centers,” said study investigator Kathryn Hutchins, a medical student at Duke University in Durham, North Carolina.
“However, it provides an opportunity for cancer centers to empower their front-line staff, as well as the oncology care team, through education and training so that the entire enterprise has a common understanding of palliative care and how to access it.”
Hutchins and her colleagues presented this research at the 2016 Palliative Care in Oncology Symposium (abstract 122).
The researchers placed 160 calls to 40 major cancer centers. The team chose to focus on National Cancer Institute-designated cancer centers because they all provide palliative care services along with other supportive care services.
The researchers used the same script for every call, asking about services for a 58-year-old female who was recently diagnosed with inoperable liver cancer. The team called each center 4 times on different days.
In 38.2% of the calls, the researchers were not able to receive complete information about supportive care services.
In 9.5% of calls, cancer center staff gave an answer other than “yes” regarding the availability of palliative care services, even though such services were available.
The answers varied and included responses such as:
- Palliative care was for end-of-life patients only (n=2)
- No physicians specialized in symptom management (n=3)
- A medical record review would be needed first (n=2).
In addition, 10 staff members said they were unsure about the availability of palliative care, and 2 were unfamiliar with the term.
Overall, 37.6% of the callers were told that all 7 supportive care services they inquired about were offered.
“As oncologists, we like to believe that, when we refer patients to our institution’s helpline, they will get connected to the services they need, but that doesn’t always happen,” said study investigator Arif Kamal, MD, of Duke Cancer Institute.
“It’s important for oncologists to be aware of these barriers and to work to eliminate them.” ![]()
Cancer patients’ caregivers may carry greater burden

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—New research suggests caring for a loved one with cancer may be more burdensome than caring for a loved one with a different condition.
Researchers analyzed data from “Caregiving in the U.S. 2015,” an online panel study of unpaid adult caregivers.
The team compared cancer and non-cancer caregivers to determine similarities and differences in characteristics and experiences.
The findings were presented at the 2016 Palliative Care in Oncology Symposium (abstract 4).
The study included 1248 caregivers, age 18 and older at the time they were surveyed, who provided care to an adult patient. Seven percent of these caregivers were looking after patients with cancer.
Cancer caregivers reported spending more hours per week providing care than non-cancer caregivers—32.9 and 23.9 hours, respectively.
In addition, cancer caregivers were more likely than other caregivers to communicate with healthcare professionals (82% and 62%, respectively), monitor and adjust patients’ care (76% and 66%, respectively), and advocate on behalf of patients (62% and 49%, respectively).
Despite high levels of engagement with providers, cancer caregivers were nearly twice as likely as non-cancer caregivers to report needing more help and information with making end-of-life decisions—40% and 21%, respectively.
“Our research demonstrates the ripple effect that cancer has on families and patient support systems,” said study investigator Erin Kent, PhD, of the National Cancer Institute in Rockville, Maryland.
“Caregiving can be extremely stressful and demanding—physically, emotionally, and financially. The data show we need to do a better job of supporting these individuals, as their wellbeing is essential to the patient’s quality of life and outcomes.”
Dr Kent emphasized the cyclical nature of cancer care, often requiring short, highly intense periods of time where patients undergo active treatment as a possible reason for the increased intensity in caregiving. She noted that such intensity is also associated with increased caregiver stress and depression.
“Based on our findings, it’s clear we need additional research on caregiving to better understand at what point providers and clinicians should intervene to assess the wellbeing of caregivers,” Dr Kent said.
“Technology, combined with use of a clinical distress rating system, could be promising in the future as a means to ensure caregivers are being supported in a meaningful way.” ![]()

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—New research suggests caring for a loved one with cancer may be more burdensome than caring for a loved one with a different condition.
Researchers analyzed data from “Caregiving in the U.S. 2015,” an online panel study of unpaid adult caregivers.
The team compared cancer and non-cancer caregivers to determine similarities and differences in characteristics and experiences.
The findings were presented at the 2016 Palliative Care in Oncology Symposium (abstract 4).
The study included 1248 caregivers, age 18 and older at the time they were surveyed, who provided care to an adult patient. Seven percent of these caregivers were looking after patients with cancer.
Cancer caregivers reported spending more hours per week providing care than non-cancer caregivers—32.9 and 23.9 hours, respectively.
In addition, cancer caregivers were more likely than other caregivers to communicate with healthcare professionals (82% and 62%, respectively), monitor and adjust patients’ care (76% and 66%, respectively), and advocate on behalf of patients (62% and 49%, respectively).
Despite high levels of engagement with providers, cancer caregivers were nearly twice as likely as non-cancer caregivers to report needing more help and information with making end-of-life decisions—40% and 21%, respectively.
“Our research demonstrates the ripple effect that cancer has on families and patient support systems,” said study investigator Erin Kent, PhD, of the National Cancer Institute in Rockville, Maryland.
“Caregiving can be extremely stressful and demanding—physically, emotionally, and financially. The data show we need to do a better job of supporting these individuals, as their wellbeing is essential to the patient’s quality of life and outcomes.”
Dr Kent emphasized the cyclical nature of cancer care, often requiring short, highly intense periods of time where patients undergo active treatment as a possible reason for the increased intensity in caregiving. She noted that such intensity is also associated with increased caregiver stress and depression.
“Based on our findings, it’s clear we need additional research on caregiving to better understand at what point providers and clinicians should intervene to assess the wellbeing of caregivers,” Dr Kent said.
“Technology, combined with use of a clinical distress rating system, could be promising in the future as a means to ensure caregivers are being supported in a meaningful way.” ![]()

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—New research suggests caring for a loved one with cancer may be more burdensome than caring for a loved one with a different condition.
Researchers analyzed data from “Caregiving in the U.S. 2015,” an online panel study of unpaid adult caregivers.
The team compared cancer and non-cancer caregivers to determine similarities and differences in characteristics and experiences.
The findings were presented at the 2016 Palliative Care in Oncology Symposium (abstract 4).
The study included 1248 caregivers, age 18 and older at the time they were surveyed, who provided care to an adult patient. Seven percent of these caregivers were looking after patients with cancer.
Cancer caregivers reported spending more hours per week providing care than non-cancer caregivers—32.9 and 23.9 hours, respectively.
In addition, cancer caregivers were more likely than other caregivers to communicate with healthcare professionals (82% and 62%, respectively), monitor and adjust patients’ care (76% and 66%, respectively), and advocate on behalf of patients (62% and 49%, respectively).
Despite high levels of engagement with providers, cancer caregivers were nearly twice as likely as non-cancer caregivers to report needing more help and information with making end-of-life decisions—40% and 21%, respectively.
“Our research demonstrates the ripple effect that cancer has on families and patient support systems,” said study investigator Erin Kent, PhD, of the National Cancer Institute in Rockville, Maryland.
“Caregiving can be extremely stressful and demanding—physically, emotionally, and financially. The data show we need to do a better job of supporting these individuals, as their wellbeing is essential to the patient’s quality of life and outcomes.”
Dr Kent emphasized the cyclical nature of cancer care, often requiring short, highly intense periods of time where patients undergo active treatment as a possible reason for the increased intensity in caregiving. She noted that such intensity is also associated with increased caregiver stress and depression.
“Based on our findings, it’s clear we need additional research on caregiving to better understand at what point providers and clinicians should intervene to assess the wellbeing of caregivers,” Dr Kent said.
“Technology, combined with use of a clinical distress rating system, could be promising in the future as a means to ensure caregivers are being supported in a meaningful way.” ![]()
Improving communication between cancer pts and docs

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—Results of the VOICE study showed that training advanced cancer patients and their oncologists on how to communicate resulted in more clinically meaningful discussions between the parties.
However, these discussions did not significantly improve patients’ understanding of their prognosis, have a significant impact on their quality of life or end-of-life care, or significantly improve the
patient-physician relationship.
Ronald Epstein, MD, of the University of Rochester in New York, and his colleagues reported results from this study in JAMA Oncology and at the 2016 Palliative Care in Oncology Symposium (abstract 2).
The VOICE study included 265 patients with stage 3 or 4 cancer, 130 of whom received communication training. As part of the training, patients received a booklet Dr Epstein’s team wrote called “My Cancer Care: What Now? What Next? What I Prefer.”
The patients and their caregivers also met with social workers or nurses to discuss commonly asked questions and how to express their fears, for example, or how to be assertive and state their preferences.
Of the 38 oncologists studied, 19 received communication training. This included mock office sessions with actors (known as standardized patients), video training, and individualized feedback.
Later, the researchers audio-recorded real sessions between the oncologists and patients, then asked both groups to fill out questionnaires. The team coded the interactions and matched the scores to the goals of the training.
Results
The study’s primary endpoint was a composite of 4 communication measures—engaging patients in consultations, responding to emotions, informing patients about prognosis and treatment choices, and balanced framing of information.
The researchers found that communication training resulted in a significant improvement in this endpoint (P=0.02).
“We have shown, in the first large study of its kind, that it is possible to change the conversation in advanced cancer,” Dr Epstein said. “This is a huge first step.”
However, when Dr Epstein and his colleagues looked at the individual components of the endpoint, only the engaging measure was significantly different between the intervention and control groups.
Communication training had no significant effect on the patient-physician relationship, patients’ quality of life, or healthcare utilization at the end of life.
Likewise, communication training had no significant effect on patients’ understanding of their prognosis, which was assessed by the discordance in 2-year survival estimates and curability estimates between patients and physicians.
“We need to try harder to communicate well so that it’s harder to miscommunicate,” Dr Epstein said. “Simply having the conversation is not enough. The quality of the conversation will influence a mutual understanding between patients and their oncologists.”
The researchers said a limitation of this study may have been the timing of the training, which was only provided once and not timed to key decision points during patients’ trajectories. The effects of the training may have waned over the months, especially as the cancer progressed.
“We need to embed communication interventions into the fabric of everyday clinical care,” Dr Epstein said. “This does not take a lot of time, but, in our audio-recordings, there was precious little dialogue that reaffirmed the human experience and the needs of patients. The next step is to make good communication the rule, not the exception, so that cancer patients’ voices can be heard.” ![]()

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—Results of the VOICE study showed that training advanced cancer patients and their oncologists on how to communicate resulted in more clinically meaningful discussions between the parties.
However, these discussions did not significantly improve patients’ understanding of their prognosis, have a significant impact on their quality of life or end-of-life care, or significantly improve the
patient-physician relationship.
Ronald Epstein, MD, of the University of Rochester in New York, and his colleagues reported results from this study in JAMA Oncology and at the 2016 Palliative Care in Oncology Symposium (abstract 2).
The VOICE study included 265 patients with stage 3 or 4 cancer, 130 of whom received communication training. As part of the training, patients received a booklet Dr Epstein’s team wrote called “My Cancer Care: What Now? What Next? What I Prefer.”
The patients and their caregivers also met with social workers or nurses to discuss commonly asked questions and how to express their fears, for example, or how to be assertive and state their preferences.
Of the 38 oncologists studied, 19 received communication training. This included mock office sessions with actors (known as standardized patients), video training, and individualized feedback.
Later, the researchers audio-recorded real sessions between the oncologists and patients, then asked both groups to fill out questionnaires. The team coded the interactions and matched the scores to the goals of the training.
Results
The study’s primary endpoint was a composite of 4 communication measures—engaging patients in consultations, responding to emotions, informing patients about prognosis and treatment choices, and balanced framing of information.
The researchers found that communication training resulted in a significant improvement in this endpoint (P=0.02).
“We have shown, in the first large study of its kind, that it is possible to change the conversation in advanced cancer,” Dr Epstein said. “This is a huge first step.”
However, when Dr Epstein and his colleagues looked at the individual components of the endpoint, only the engaging measure was significantly different between the intervention and control groups.
Communication training had no significant effect on the patient-physician relationship, patients’ quality of life, or healthcare utilization at the end of life.
Likewise, communication training had no significant effect on patients’ understanding of their prognosis, which was assessed by the discordance in 2-year survival estimates and curability estimates between patients and physicians.
“We need to try harder to communicate well so that it’s harder to miscommunicate,” Dr Epstein said. “Simply having the conversation is not enough. The quality of the conversation will influence a mutual understanding between patients and their oncologists.”
The researchers said a limitation of this study may have been the timing of the training, which was only provided once and not timed to key decision points during patients’ trajectories. The effects of the training may have waned over the months, especially as the cancer progressed.
“We need to embed communication interventions into the fabric of everyday clinical care,” Dr Epstein said. “This does not take a lot of time, but, in our audio-recordings, there was precious little dialogue that reaffirmed the human experience and the needs of patients. The next step is to make good communication the rule, not the exception, so that cancer patients’ voices can be heard.” ![]()

©ASCO/Todd Buchanan 2016
SAN FRANCISCO—Results of the VOICE study showed that training advanced cancer patients and their oncologists on how to communicate resulted in more clinically meaningful discussions between the parties.
However, these discussions did not significantly improve patients’ understanding of their prognosis, have a significant impact on their quality of life or end-of-life care, or significantly improve the
patient-physician relationship.
Ronald Epstein, MD, of the University of Rochester in New York, and his colleagues reported results from this study in JAMA Oncology and at the 2016 Palliative Care in Oncology Symposium (abstract 2).
The VOICE study included 265 patients with stage 3 or 4 cancer, 130 of whom received communication training. As part of the training, patients received a booklet Dr Epstein’s team wrote called “My Cancer Care: What Now? What Next? What I Prefer.”
The patients and their caregivers also met with social workers or nurses to discuss commonly asked questions and how to express their fears, for example, or how to be assertive and state their preferences.
Of the 38 oncologists studied, 19 received communication training. This included mock office sessions with actors (known as standardized patients), video training, and individualized feedback.
Later, the researchers audio-recorded real sessions between the oncologists and patients, then asked both groups to fill out questionnaires. The team coded the interactions and matched the scores to the goals of the training.
Results
The study’s primary endpoint was a composite of 4 communication measures—engaging patients in consultations, responding to emotions, informing patients about prognosis and treatment choices, and balanced framing of information.
The researchers found that communication training resulted in a significant improvement in this endpoint (P=0.02).
“We have shown, in the first large study of its kind, that it is possible to change the conversation in advanced cancer,” Dr Epstein said. “This is a huge first step.”
However, when Dr Epstein and his colleagues looked at the individual components of the endpoint, only the engaging measure was significantly different between the intervention and control groups.
Communication training had no significant effect on the patient-physician relationship, patients’ quality of life, or healthcare utilization at the end of life.
Likewise, communication training had no significant effect on patients’ understanding of their prognosis, which was assessed by the discordance in 2-year survival estimates and curability estimates between patients and physicians.
“We need to try harder to communicate well so that it’s harder to miscommunicate,” Dr Epstein said. “Simply having the conversation is not enough. The quality of the conversation will influence a mutual understanding between patients and their oncologists.”
The researchers said a limitation of this study may have been the timing of the training, which was only provided once and not timed to key decision points during patients’ trajectories. The effects of the training may have waned over the months, especially as the cancer progressed.
“We need to embed communication interventions into the fabric of everyday clinical care,” Dr Epstein said. “This does not take a lot of time, but, in our audio-recordings, there was precious little dialogue that reaffirmed the human experience and the needs of patients. The next step is to make good communication the rule, not the exception, so that cancer patients’ voices can be heard.” ![]()
Risk factors for early death in older pts with DLBCL

Photo courtesy of CDC
A retrospective study has uncovered potential risk factors for early death in older patients with diffuse large B-cell lymphoma (DLBCL).
Researchers examined electronic health record data for roughly 5500 DLBCL patients over the age of 65 who received contemporary immunochemotherapy.
This revealed 7 factors that were significantly associated with the risk of death within 30 days of treatment initiation.
“The first month of treatment, when patients are compromised both by active lymphoma and toxicities of chemotherapy, is a period of particular concern, as nearly 1 in 4 patients were hospitalized during that time,” said study author Adam J. Olszewski, MD, of Rhode Island Hospital in Providence.
“While comprehensive geriatric assessment remains the gold standard for risk assessment, our study suggests that readily available data from electronic medical records can help identify the high-risk factors in practice.”
Dr Olszewski and his colleagues described their study in JNCCN.
The researchers looked at Medicare claims linked to Surveillance, Epidemiology and End Results registry data for 5530 DLBCL patients who had a median age of 76.
The patients were treated with rituximab, cyclophosphamide, and vincristine in combination with doxorubicin, mitoxantrone, or etoposide from 2003 to 2012.
The cumulative incidence of death at 30 days was 2.2%. The most common causes of death were lymphoma (72%), heart disease (9%), septicemia (3%), and cerebrovascular events (3%).
The researchers created a prediction model based on 7 factors that were significantly associated with early death in multivariate analysis. These include:
- B symptoms
- Chronic kidney disease
- Poor performance status
- Prior use of walking aids or wheelchairs
- Prior hospitalization within the past 12 months
- Upper endoscopy within the past 12 months
- Age 75 or older.
Patients with 0 to 1 of these risk factors were considered low-risk, with a 0.6% chance of early death. Fifty-six percent of the patients studied fit this definition.
Patients with 2 to 3 of the risk factors were intermediate-risk, with a 3.2% chance of early death. Thirty-eight percent of the patients studied fell into this category.
Only 6% of the patients studied were considered high-risk. These patients had 4 or more risk factors and an 8.3% chance of early death.
The researchers also found the administration of prophylactic granulocyte-colony stimulating factor (G-CSF) was associated with lower probability of early death in the high-risk group.
They noted that prophylactic G-CSF was given to 66% of patients in this study, which suggests an opportunity for preventing early deaths.
“It is equally important to realize that a majority of older patients without risk factors can safely receive curative immunochemotherapy,” Dr Olszewski said. “Enhanced supportive care and monitoring should be provided for high-risk groups.” ![]()

Photo courtesy of CDC
A retrospective study has uncovered potential risk factors for early death in older patients with diffuse large B-cell lymphoma (DLBCL).
Researchers examined electronic health record data for roughly 5500 DLBCL patients over the age of 65 who received contemporary immunochemotherapy.
This revealed 7 factors that were significantly associated with the risk of death within 30 days of treatment initiation.
“The first month of treatment, when patients are compromised both by active lymphoma and toxicities of chemotherapy, is a period of particular concern, as nearly 1 in 4 patients were hospitalized during that time,” said study author Adam J. Olszewski, MD, of Rhode Island Hospital in Providence.
“While comprehensive geriatric assessment remains the gold standard for risk assessment, our study suggests that readily available data from electronic medical records can help identify the high-risk factors in practice.”
Dr Olszewski and his colleagues described their study in JNCCN.
The researchers looked at Medicare claims linked to Surveillance, Epidemiology and End Results registry data for 5530 DLBCL patients who had a median age of 76.
The patients were treated with rituximab, cyclophosphamide, and vincristine in combination with doxorubicin, mitoxantrone, or etoposide from 2003 to 2012.
The cumulative incidence of death at 30 days was 2.2%. The most common causes of death were lymphoma (72%), heart disease (9%), septicemia (3%), and cerebrovascular events (3%).
The researchers created a prediction model based on 7 factors that were significantly associated with early death in multivariate analysis. These include:
- B symptoms
- Chronic kidney disease
- Poor performance status
- Prior use of walking aids or wheelchairs
- Prior hospitalization within the past 12 months
- Upper endoscopy within the past 12 months
- Age 75 or older.
Patients with 0 to 1 of these risk factors were considered low-risk, with a 0.6% chance of early death. Fifty-six percent of the patients studied fit this definition.
Patients with 2 to 3 of the risk factors were intermediate-risk, with a 3.2% chance of early death. Thirty-eight percent of the patients studied fell into this category.
Only 6% of the patients studied were considered high-risk. These patients had 4 or more risk factors and an 8.3% chance of early death.
The researchers also found the administration of prophylactic granulocyte-colony stimulating factor (G-CSF) was associated with lower probability of early death in the high-risk group.
They noted that prophylactic G-CSF was given to 66% of patients in this study, which suggests an opportunity for preventing early deaths.
“It is equally important to realize that a majority of older patients without risk factors can safely receive curative immunochemotherapy,” Dr Olszewski said. “Enhanced supportive care and monitoring should be provided for high-risk groups.” ![]()

Photo courtesy of CDC
A retrospective study has uncovered potential risk factors for early death in older patients with diffuse large B-cell lymphoma (DLBCL).
Researchers examined electronic health record data for roughly 5500 DLBCL patients over the age of 65 who received contemporary immunochemotherapy.
This revealed 7 factors that were significantly associated with the risk of death within 30 days of treatment initiation.
“The first month of treatment, when patients are compromised both by active lymphoma and toxicities of chemotherapy, is a period of particular concern, as nearly 1 in 4 patients were hospitalized during that time,” said study author Adam J. Olszewski, MD, of Rhode Island Hospital in Providence.
“While comprehensive geriatric assessment remains the gold standard for risk assessment, our study suggests that readily available data from electronic medical records can help identify the high-risk factors in practice.”
Dr Olszewski and his colleagues described their study in JNCCN.
The researchers looked at Medicare claims linked to Surveillance, Epidemiology and End Results registry data for 5530 DLBCL patients who had a median age of 76.
The patients were treated with rituximab, cyclophosphamide, and vincristine in combination with doxorubicin, mitoxantrone, or etoposide from 2003 to 2012.
The cumulative incidence of death at 30 days was 2.2%. The most common causes of death were lymphoma (72%), heart disease (9%), septicemia (3%), and cerebrovascular events (3%).
The researchers created a prediction model based on 7 factors that were significantly associated with early death in multivariate analysis. These include:
- B symptoms
- Chronic kidney disease
- Poor performance status
- Prior use of walking aids or wheelchairs
- Prior hospitalization within the past 12 months
- Upper endoscopy within the past 12 months
- Age 75 or older.
Patients with 0 to 1 of these risk factors were considered low-risk, with a 0.6% chance of early death. Fifty-six percent of the patients studied fit this definition.
Patients with 2 to 3 of the risk factors were intermediate-risk, with a 3.2% chance of early death. Thirty-eight percent of the patients studied fell into this category.
Only 6% of the patients studied were considered high-risk. These patients had 4 or more risk factors and an 8.3% chance of early death.
The researchers also found the administration of prophylactic granulocyte-colony stimulating factor (G-CSF) was associated with lower probability of early death in the high-risk group.
They noted that prophylactic G-CSF was given to 66% of patients in this study, which suggests an opportunity for preventing early deaths.
“It is equally important to realize that a majority of older patients without risk factors can safely receive curative immunochemotherapy,” Dr Olszewski said. “Enhanced supportive care and monitoring should be provided for high-risk groups.” ![]()
Optimizing CAR T-cell therapy in NHL

Photo from Fred Hutchinson
Cancer Research Center
Results from a phase 1 study have provided insights that may help researchers optimize treatment with JCAR014, a chimeric antigen receptor (CAR) T-cell therapy, in patients with advanced non-Hodgkin lymphoma (NHL).
Researchers said they identified a lymphodepleting regimen that improved the likelihood of complete response (CR) to JCAR014.
Although the regimen also increased the risk of severe cytokine release syndrome (CRS) and neurotoxicity, the researchers discovered biomarkers that might allow them to identify patients who have a high risk of these events and could potentially benefit from early interventions.
The researchers reported these findings in Science Translational Medicine. The trial (NCT01865617) was funded, in part, by Juno Therapeutics, the company developing JCAR014.
Previous results from this trial, in patients with acute lymphoblastic leukemia, were published in The Journal of Clinical Investigation.
About JCAR014
JCAR014 is a CD19-directed CAR T-cell therapy in which CD4+ and CD8+ cells are administered in equal proportions.
“The idea . . . is that by [controlling the ratio of T cells], we would get more reproducible data around the effects of the cells—both beneficial effects against the cancer and also any side effects they might cause the patient,” said study author Stanley Riddell, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
“And then, by adjusting the dose, we could improve what we call the therapeutic index—the benefit against the tumor—without too much toxicity.”
Patients and treatment
Dr Riddell and his colleagues reported results with JCAR014, following lymphodepleting chemotherapy, in 32 patients with NHL who had a median age of 58 (range, 36-70).
The patients had de novo large B-cell lymphoma (n=11), transformed de novo large B-cell lymphoma (n=11), mantle cell lymphoma (n=4), and follicular lymphoma (n=6).
They had received a median of 5 prior treatment regimens (range, 2 to 11), including autologous
(n=14) and allogeneic (n=4) transplant. All patients had measurable disease (≥ 2 cm) in the lymph nodes or other extramedullary sites at baseline.
The patients received JCAR014 at 1 of 3 dose levels, given 36 to 96 hours after lymphodepleting chemotherapy.
Twelve patients received cyclophosphamide (Cy) alone or Cy and etoposide (E), and 20 received Cy plus fludarabine (Flu). Five patients received 2 × 105 CAR T cells/kg, 18 received 2 × 106 CAR T cells/kg, and 9 received 2 × 107 CAR T cells/kg.
Efficacy
In the 30 evaluable patients, the overall response rate (ORR) was 63% (n=19), and the CR rate was 33% (n=10).
Among patients who received Cy or Cy/E, the ORR was 50% (n=6), and the CR rate was 8% (n=1). Among patients who received Cy/Flu, the ORR was 72% (n=13), and the CR rate was 50% (n=9).
The patients who received Cy/Flu had better CAR T-cell expansion and persistence than patients who received Cy or Cy/E, which was reflected in the higher response rates.
Higher response rates were also observed in patients who received 2 × 106 CAR T cells/kg rather than the other 2 dose levels.
The researchers noted that, although follow-up is short, patients who received CAR T cells at ≤ 2 × 106/kg after Cy/Flu had better progression-free survival (P=0.008) than patients who received CAR T cells at ≤ 2 × 106/kg after Cy or Cy/E.
Of the 9 patients who achieved a CR after Cy/Flu and JCAR014, 1 has relapsed, with follow-up ranging from 2.3 months to 11.2 months. (Seven of these 9 patients had received 2 × 106 CAR T cells/kg, and 1 patient each had received the other 2 doses.)
“The main message is that you can treat patients with non-Hodgkin’s lymphoma with CAR T cells and get very good response rates with optimization of the CAR T-cell dose and lymphodepletion,” said study author Cameron Turtle, MBBS, PhD, of the Fred Hutchinson Cancer Research Center.
“Strategies like modifying the lymphodepletion in conjunction with suitable CAR T-cell dosing can have a big impact on clinical outcome.”
Safety
Two patients who were treated with the highest CAR T-cell dose (2 × 107 cells/kg) died—1 of pontine hemorrhage and 1 of gastrointestinal hemorrhage associated with a known gastrointestinal invasive lymphomatous mass.
This dose was also associated with an increased risk of severe CRS and neurotoxicity, as was the Cy/Flu regimen.
Twenty patients (62.5%) developed CRS, and 4 (12.5%) had severe CRS. All 4 of these patients had received Cy/Flu.
Nine patients (28%) developed severe neurotoxicity (grade 3 or higher), 7 of whom had received Cy/Flu.
Three of the 6 patients (50%) treated at 2 × 107 CAR-T cells/kg after Cy/Flu developed severe CRS, and 4 of the 6 patients (67%) developed severe neurotoxicity.
The researchers looked for biomarkers of toxicity in serum collected 1 day after CAR T-cell infusion.
They found that high IL-6, IL-8, IL-10, IL-15, and IFN-γ concentrations were associated with subsequent severe CRS and neurotoxicity, and low TGF-Β concentration was associated with neurotoxicity.
The team said these findings provide an opportunity for studying the use of serum cytokine concentrations to identify patients at the highest risk of toxicity who might benefit from early interventions. ![]()

Photo from Fred Hutchinson
Cancer Research Center
Results from a phase 1 study have provided insights that may help researchers optimize treatment with JCAR014, a chimeric antigen receptor (CAR) T-cell therapy, in patients with advanced non-Hodgkin lymphoma (NHL).
Researchers said they identified a lymphodepleting regimen that improved the likelihood of complete response (CR) to JCAR014.
Although the regimen also increased the risk of severe cytokine release syndrome (CRS) and neurotoxicity, the researchers discovered biomarkers that might allow them to identify patients who have a high risk of these events and could potentially benefit from early interventions.
The researchers reported these findings in Science Translational Medicine. The trial (NCT01865617) was funded, in part, by Juno Therapeutics, the company developing JCAR014.
Previous results from this trial, in patients with acute lymphoblastic leukemia, were published in The Journal of Clinical Investigation.
About JCAR014
JCAR014 is a CD19-directed CAR T-cell therapy in which CD4+ and CD8+ cells are administered in equal proportions.
“The idea . . . is that by [controlling the ratio of T cells], we would get more reproducible data around the effects of the cells—both beneficial effects against the cancer and also any side effects they might cause the patient,” said study author Stanley Riddell, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
“And then, by adjusting the dose, we could improve what we call the therapeutic index—the benefit against the tumor—without too much toxicity.”
Patients and treatment
Dr Riddell and his colleagues reported results with JCAR014, following lymphodepleting chemotherapy, in 32 patients with NHL who had a median age of 58 (range, 36-70).
The patients had de novo large B-cell lymphoma (n=11), transformed de novo large B-cell lymphoma (n=11), mantle cell lymphoma (n=4), and follicular lymphoma (n=6).
They had received a median of 5 prior treatment regimens (range, 2 to 11), including autologous
(n=14) and allogeneic (n=4) transplant. All patients had measurable disease (≥ 2 cm) in the lymph nodes or other extramedullary sites at baseline.
The patients received JCAR014 at 1 of 3 dose levels, given 36 to 96 hours after lymphodepleting chemotherapy.
Twelve patients received cyclophosphamide (Cy) alone or Cy and etoposide (E), and 20 received Cy plus fludarabine (Flu). Five patients received 2 × 105 CAR T cells/kg, 18 received 2 × 106 CAR T cells/kg, and 9 received 2 × 107 CAR T cells/kg.
Efficacy
In the 30 evaluable patients, the overall response rate (ORR) was 63% (n=19), and the CR rate was 33% (n=10).
Among patients who received Cy or Cy/E, the ORR was 50% (n=6), and the CR rate was 8% (n=1). Among patients who received Cy/Flu, the ORR was 72% (n=13), and the CR rate was 50% (n=9).
The patients who received Cy/Flu had better CAR T-cell expansion and persistence than patients who received Cy or Cy/E, which was reflected in the higher response rates.
Higher response rates were also observed in patients who received 2 × 106 CAR T cells/kg rather than the other 2 dose levels.
The researchers noted that, although follow-up is short, patients who received CAR T cells at ≤ 2 × 106/kg after Cy/Flu had better progression-free survival (P=0.008) than patients who received CAR T cells at ≤ 2 × 106/kg after Cy or Cy/E.
Of the 9 patients who achieved a CR after Cy/Flu and JCAR014, 1 has relapsed, with follow-up ranging from 2.3 months to 11.2 months. (Seven of these 9 patients had received 2 × 106 CAR T cells/kg, and 1 patient each had received the other 2 doses.)
“The main message is that you can treat patients with non-Hodgkin’s lymphoma with CAR T cells and get very good response rates with optimization of the CAR T-cell dose and lymphodepletion,” said study author Cameron Turtle, MBBS, PhD, of the Fred Hutchinson Cancer Research Center.
“Strategies like modifying the lymphodepletion in conjunction with suitable CAR T-cell dosing can have a big impact on clinical outcome.”
Safety
Two patients who were treated with the highest CAR T-cell dose (2 × 107 cells/kg) died—1 of pontine hemorrhage and 1 of gastrointestinal hemorrhage associated with a known gastrointestinal invasive lymphomatous mass.
This dose was also associated with an increased risk of severe CRS and neurotoxicity, as was the Cy/Flu regimen.
Twenty patients (62.5%) developed CRS, and 4 (12.5%) had severe CRS. All 4 of these patients had received Cy/Flu.
Nine patients (28%) developed severe neurotoxicity (grade 3 or higher), 7 of whom had received Cy/Flu.
Three of the 6 patients (50%) treated at 2 × 107 CAR-T cells/kg after Cy/Flu developed severe CRS, and 4 of the 6 patients (67%) developed severe neurotoxicity.
The researchers looked for biomarkers of toxicity in serum collected 1 day after CAR T-cell infusion.
They found that high IL-6, IL-8, IL-10, IL-15, and IFN-γ concentrations were associated with subsequent severe CRS and neurotoxicity, and low TGF-Β concentration was associated with neurotoxicity.
The team said these findings provide an opportunity for studying the use of serum cytokine concentrations to identify patients at the highest risk of toxicity who might benefit from early interventions. ![]()

Photo from Fred Hutchinson
Cancer Research Center
Results from a phase 1 study have provided insights that may help researchers optimize treatment with JCAR014, a chimeric antigen receptor (CAR) T-cell therapy, in patients with advanced non-Hodgkin lymphoma (NHL).
Researchers said they identified a lymphodepleting regimen that improved the likelihood of complete response (CR) to JCAR014.
Although the regimen also increased the risk of severe cytokine release syndrome (CRS) and neurotoxicity, the researchers discovered biomarkers that might allow them to identify patients who have a high risk of these events and could potentially benefit from early interventions.
The researchers reported these findings in Science Translational Medicine. The trial (NCT01865617) was funded, in part, by Juno Therapeutics, the company developing JCAR014.
Previous results from this trial, in patients with acute lymphoblastic leukemia, were published in The Journal of Clinical Investigation.
About JCAR014
JCAR014 is a CD19-directed CAR T-cell therapy in which CD4+ and CD8+ cells are administered in equal proportions.
“The idea . . . is that by [controlling the ratio of T cells], we would get more reproducible data around the effects of the cells—both beneficial effects against the cancer and also any side effects they might cause the patient,” said study author Stanley Riddell, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
“And then, by adjusting the dose, we could improve what we call the therapeutic index—the benefit against the tumor—without too much toxicity.”
Patients and treatment
Dr Riddell and his colleagues reported results with JCAR014, following lymphodepleting chemotherapy, in 32 patients with NHL who had a median age of 58 (range, 36-70).
The patients had de novo large B-cell lymphoma (n=11), transformed de novo large B-cell lymphoma (n=11), mantle cell lymphoma (n=4), and follicular lymphoma (n=6).
They had received a median of 5 prior treatment regimens (range, 2 to 11), including autologous
(n=14) and allogeneic (n=4) transplant. All patients had measurable disease (≥ 2 cm) in the lymph nodes or other extramedullary sites at baseline.
The patients received JCAR014 at 1 of 3 dose levels, given 36 to 96 hours after lymphodepleting chemotherapy.
Twelve patients received cyclophosphamide (Cy) alone or Cy and etoposide (E), and 20 received Cy plus fludarabine (Flu). Five patients received 2 × 105 CAR T cells/kg, 18 received 2 × 106 CAR T cells/kg, and 9 received 2 × 107 CAR T cells/kg.
Efficacy
In the 30 evaluable patients, the overall response rate (ORR) was 63% (n=19), and the CR rate was 33% (n=10).
Among patients who received Cy or Cy/E, the ORR was 50% (n=6), and the CR rate was 8% (n=1). Among patients who received Cy/Flu, the ORR was 72% (n=13), and the CR rate was 50% (n=9).
The patients who received Cy/Flu had better CAR T-cell expansion and persistence than patients who received Cy or Cy/E, which was reflected in the higher response rates.
Higher response rates were also observed in patients who received 2 × 106 CAR T cells/kg rather than the other 2 dose levels.
The researchers noted that, although follow-up is short, patients who received CAR T cells at ≤ 2 × 106/kg after Cy/Flu had better progression-free survival (P=0.008) than patients who received CAR T cells at ≤ 2 × 106/kg after Cy or Cy/E.
Of the 9 patients who achieved a CR after Cy/Flu and JCAR014, 1 has relapsed, with follow-up ranging from 2.3 months to 11.2 months. (Seven of these 9 patients had received 2 × 106 CAR T cells/kg, and 1 patient each had received the other 2 doses.)
“The main message is that you can treat patients with non-Hodgkin’s lymphoma with CAR T cells and get very good response rates with optimization of the CAR T-cell dose and lymphodepletion,” said study author Cameron Turtle, MBBS, PhD, of the Fred Hutchinson Cancer Research Center.
“Strategies like modifying the lymphodepletion in conjunction with suitable CAR T-cell dosing can have a big impact on clinical outcome.”
Safety
Two patients who were treated with the highest CAR T-cell dose (2 × 107 cells/kg) died—1 of pontine hemorrhage and 1 of gastrointestinal hemorrhage associated with a known gastrointestinal invasive lymphomatous mass.
This dose was also associated with an increased risk of severe CRS and neurotoxicity, as was the Cy/Flu regimen.
Twenty patients (62.5%) developed CRS, and 4 (12.5%) had severe CRS. All 4 of these patients had received Cy/Flu.
Nine patients (28%) developed severe neurotoxicity (grade 3 or higher), 7 of whom had received Cy/Flu.
Three of the 6 patients (50%) treated at 2 × 107 CAR-T cells/kg after Cy/Flu developed severe CRS, and 4 of the 6 patients (67%) developed severe neurotoxicity.
The researchers looked for biomarkers of toxicity in serum collected 1 day after CAR T-cell infusion.
They found that high IL-6, IL-8, IL-10, IL-15, and IFN-γ concentrations were associated with subsequent severe CRS and neurotoxicity, and low TGF-Β concentration was associated with neurotoxicity.
The team said these findings provide an opportunity for studying the use of serum cytokine concentrations to identify patients at the highest risk of toxicity who might benefit from early interventions. ![]()
Pneumonitis with nivolumab treatment shows common radiographic patterns
A study of cancer patients enrolled in trials of the programmed cell death-1 inhibiting medicine nivolumab found that among a minority who developed pneumonitis during treatment, distinct radiographic patterns were significantly associated with the level of pneumonitis severity.
Investigators found that cryptic organizing pneumonia pattern (COP) was the most common, though not the most severe. Led by Mizuki Nishino, MD, of Brigham and Women’s Hospital, Boston, the researchers looked at the 20 patients out of a cohort of 170 (11.8%) who had developed pneumonitis, and found that radiologic patterns indicating acute interstitial pneumonia/acute respiratory distress syndrome (n = 2) had the highest severity grade on a scale of 1-5 (median 3), followed by those with COP pattern (n = 13, median grade 2), hypersensitivity pneumonitis (n = 2, median grade 1), and nonspecific interstitial pneumonia (n = 3, median grade 1). The pattern was significantly associated with severity (P = .0006).
The study cohort included patients being treated with nivolumab for lung cancer, melanoma, and lymphoma; the COP patten was the most common across tumor types and observed in patients receiving monotherapy and combination therapy alike. Therapy with nivolumab was suspended for all 20 pneumonitis patients, and most (n = 17) received treatment for pneumonitis with corticosteroids with or without infliximab, for a median treatment time of 6 weeks. Seven patients were able to restart nivolumab, though pneumonitis recurred in two, the investigators reported (Clin Cancer Res. 2016 Aug 17. doi: 10.1158/1078-0432.CCR-16-1320).
“Time from initiation of therapy to the development of pneumonitis had a wide range (0.5-11.5 months), indicating an importance of careful observation and follow-up for signs and symptoms of pneumonitis throughout treatment,” Dr. Nishino and colleagues wrote in their analysis, adding that shorter times were observed for lung cancer patients, possibly because of their higher pulmonary burden, a lower threshold for performing chest scans in these patients, or both. “In most patients, clinical and radiographic improvements were noted after treatment, indicating that [PD-1 inhibitor-related pneumonitis], although potentially serious, is treatable if diagnosed and managed appropriately. The observation emphasizes the importance of timely recognition, accurate diagnosis, and early intervention.”
The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
A study of cancer patients enrolled in trials of the programmed cell death-1 inhibiting medicine nivolumab found that among a minority who developed pneumonitis during treatment, distinct radiographic patterns were significantly associated with the level of pneumonitis severity.
Investigators found that cryptic organizing pneumonia pattern (COP) was the most common, though not the most severe. Led by Mizuki Nishino, MD, of Brigham and Women’s Hospital, Boston, the researchers looked at the 20 patients out of a cohort of 170 (11.8%) who had developed pneumonitis, and found that radiologic patterns indicating acute interstitial pneumonia/acute respiratory distress syndrome (n = 2) had the highest severity grade on a scale of 1-5 (median 3), followed by those with COP pattern (n = 13, median grade 2), hypersensitivity pneumonitis (n = 2, median grade 1), and nonspecific interstitial pneumonia (n = 3, median grade 1). The pattern was significantly associated with severity (P = .0006).
The study cohort included patients being treated with nivolumab for lung cancer, melanoma, and lymphoma; the COP patten was the most common across tumor types and observed in patients receiving monotherapy and combination therapy alike. Therapy with nivolumab was suspended for all 20 pneumonitis patients, and most (n = 17) received treatment for pneumonitis with corticosteroids with or without infliximab, for a median treatment time of 6 weeks. Seven patients were able to restart nivolumab, though pneumonitis recurred in two, the investigators reported (Clin Cancer Res. 2016 Aug 17. doi: 10.1158/1078-0432.CCR-16-1320).
“Time from initiation of therapy to the development of pneumonitis had a wide range (0.5-11.5 months), indicating an importance of careful observation and follow-up for signs and symptoms of pneumonitis throughout treatment,” Dr. Nishino and colleagues wrote in their analysis, adding that shorter times were observed for lung cancer patients, possibly because of their higher pulmonary burden, a lower threshold for performing chest scans in these patients, or both. “In most patients, clinical and radiographic improvements were noted after treatment, indicating that [PD-1 inhibitor-related pneumonitis], although potentially serious, is treatable if diagnosed and managed appropriately. The observation emphasizes the importance of timely recognition, accurate diagnosis, and early intervention.”
The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
A study of cancer patients enrolled in trials of the programmed cell death-1 inhibiting medicine nivolumab found that among a minority who developed pneumonitis during treatment, distinct radiographic patterns were significantly associated with the level of pneumonitis severity.
Investigators found that cryptic organizing pneumonia pattern (COP) was the most common, though not the most severe. Led by Mizuki Nishino, MD, of Brigham and Women’s Hospital, Boston, the researchers looked at the 20 patients out of a cohort of 170 (11.8%) who had developed pneumonitis, and found that radiologic patterns indicating acute interstitial pneumonia/acute respiratory distress syndrome (n = 2) had the highest severity grade on a scale of 1-5 (median 3), followed by those with COP pattern (n = 13, median grade 2), hypersensitivity pneumonitis (n = 2, median grade 1), and nonspecific interstitial pneumonia (n = 3, median grade 1). The pattern was significantly associated with severity (P = .0006).
The study cohort included patients being treated with nivolumab for lung cancer, melanoma, and lymphoma; the COP patten was the most common across tumor types and observed in patients receiving monotherapy and combination therapy alike. Therapy with nivolumab was suspended for all 20 pneumonitis patients, and most (n = 17) received treatment for pneumonitis with corticosteroids with or without infliximab, for a median treatment time of 6 weeks. Seven patients were able to restart nivolumab, though pneumonitis recurred in two, the investigators reported (Clin Cancer Res. 2016 Aug 17. doi: 10.1158/1078-0432.CCR-16-1320).
“Time from initiation of therapy to the development of pneumonitis had a wide range (0.5-11.5 months), indicating an importance of careful observation and follow-up for signs and symptoms of pneumonitis throughout treatment,” Dr. Nishino and colleagues wrote in their analysis, adding that shorter times were observed for lung cancer patients, possibly because of their higher pulmonary burden, a lower threshold for performing chest scans in these patients, or both. “In most patients, clinical and radiographic improvements were noted after treatment, indicating that [PD-1 inhibitor-related pneumonitis], although potentially serious, is treatable if diagnosed and managed appropriately. The observation emphasizes the importance of timely recognition, accurate diagnosis, and early intervention.”
The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
FROM CLINICAL CANCER RESEARCH
Key clinical point: Pneumonitis related to treatment with PD-1 inhibitors showed distinct radiographic patterns associated with severity; most cases resolved with corticosteroid treatment.
Major finding: Of 20 patients in nivolumab trials who developed pneumonitis, a COP pattern was seen in 13, and other patterns in 7; different patterns were significantly associated with pneumonitis severity (P = .006).
Data source: 170 patients with melanoma, lung cancer or lymphoma enrolled in single-site open-label clinical trial of nivolumab.
Disclosures: The lead author and several coauthors disclosed funding from Bristol-Myers Squibb, which sponsored the trial, as well as from other manufacturers.
HSCT may age T cells as much as 30 years

Photo by Chad McNeeley
New research suggests hematopoietic stem cell transplant (HSCT) may increase the molecular age of peripheral blood T cells.
The study showed an increase in peripheral blood T-cell senescence in patients with hematologic malignancies who were treated with autologous (auto-) or allogeneic (allo-) HSCT.
The patients had elevated levels of p16INK4a, a known marker of cellular senescence.
Auto-HSCT in particular had a strong effect on p16INK4a, increasing the expression of this marker to a degree comparable to 30 years of chronological aging.
Researchers reported these findings in EBioMedicine.
“We know that transplant is life-prolonging, and, in many cases, it’s life-saving for many patients with blood cancers and other disorders,” said study author William Wood, MD, of the University of North Carolina School of Medicine in Chapel Hill.
“At the same time, we’re increasingly recognizing that survivors of transplant are at risk for long-term health problems, and so there is interest in determining what markers may exist to help predict risk for long-term health problems or even in helping choose which patients are best candidates for transplantation.”
With this in mind, Dr Wood and his colleagues looked at levels of p16INK4a in 63 patients who underwent auto- or allo-HSCT to treat myeloma, lymphoma, or leukemia. The researchers assessed p16INK4a expression in T cells before HSCT and 6 months after.
Among auto-HSCT recipients, there were no baseline characteristics associated with pre-transplant p16INK4a expression.
However, allo-HSCT recipients had significantly higher pre-transplant p16INK4a levels the more cycles of chemotherapy they received before transplant (P=0.003), if they had previously undergone auto-HSCT (P=0.01), and if they had been exposed to alkylating agents (P=0.01).
After transplant, allo-HSCT recipients had a 1.93-fold increase in p16INK4a expression (P=0.0004), and auto-HSCT recipients had a 3.05-fold increase (P=0.002).
The researchers said the measured change in p16INK4a from pre- to post-HSCT in allogeneic recipients likely underestimates the age-promoting effects of HSCT, given that the pre-HSCT levels were elevated in the recipients from prior therapeutic exposure.
The researchers also pointed out that this study does not show a clear connection between changes in p16INK4a levels and the actual function of peripheral blood T cells, but they did say that p16INK4a is “arguably one of the best in vivo markers of cellular senescence and is directly associated with age-related deterioration.”
So the results of this research suggest the forced bone marrow repopulation associated with HSCT accelerates the molecular aging of peripheral blood T cells.
“Many oncologists would not be surprised by the finding that stem cell transplant accelerates aspects of aging,” said study author Norman Sharpless, MD, of the University of North Carolina School of Medicine.
“We know that, years after a curative transplant, stem cell transplant survivors are at increased risk for blood problems that can occur with aging, such as reduced immunity, increased risk for bone marrow failure, and increased risk of blood cancers. What is important about this work, however, is that it allows us to quantify the effect of stem cell transplant on molecular age.” ![]()

Photo by Chad McNeeley
New research suggests hematopoietic stem cell transplant (HSCT) may increase the molecular age of peripheral blood T cells.
The study showed an increase in peripheral blood T-cell senescence in patients with hematologic malignancies who were treated with autologous (auto-) or allogeneic (allo-) HSCT.
The patients had elevated levels of p16INK4a, a known marker of cellular senescence.
Auto-HSCT in particular had a strong effect on p16INK4a, increasing the expression of this marker to a degree comparable to 30 years of chronological aging.
Researchers reported these findings in EBioMedicine.
“We know that transplant is life-prolonging, and, in many cases, it’s life-saving for many patients with blood cancers and other disorders,” said study author William Wood, MD, of the University of North Carolina School of Medicine in Chapel Hill.
“At the same time, we’re increasingly recognizing that survivors of transplant are at risk for long-term health problems, and so there is interest in determining what markers may exist to help predict risk for long-term health problems or even in helping choose which patients are best candidates for transplantation.”
With this in mind, Dr Wood and his colleagues looked at levels of p16INK4a in 63 patients who underwent auto- or allo-HSCT to treat myeloma, lymphoma, or leukemia. The researchers assessed p16INK4a expression in T cells before HSCT and 6 months after.
Among auto-HSCT recipients, there were no baseline characteristics associated with pre-transplant p16INK4a expression.
However, allo-HSCT recipients had significantly higher pre-transplant p16INK4a levels the more cycles of chemotherapy they received before transplant (P=0.003), if they had previously undergone auto-HSCT (P=0.01), and if they had been exposed to alkylating agents (P=0.01).
After transplant, allo-HSCT recipients had a 1.93-fold increase in p16INK4a expression (P=0.0004), and auto-HSCT recipients had a 3.05-fold increase (P=0.002).
The researchers said the measured change in p16INK4a from pre- to post-HSCT in allogeneic recipients likely underestimates the age-promoting effects of HSCT, given that the pre-HSCT levels were elevated in the recipients from prior therapeutic exposure.
The researchers also pointed out that this study does not show a clear connection between changes in p16INK4a levels and the actual function of peripheral blood T cells, but they did say that p16INK4a is “arguably one of the best in vivo markers of cellular senescence and is directly associated with age-related deterioration.”
So the results of this research suggest the forced bone marrow repopulation associated with HSCT accelerates the molecular aging of peripheral blood T cells.
“Many oncologists would not be surprised by the finding that stem cell transplant accelerates aspects of aging,” said study author Norman Sharpless, MD, of the University of North Carolina School of Medicine.
“We know that, years after a curative transplant, stem cell transplant survivors are at increased risk for blood problems that can occur with aging, such as reduced immunity, increased risk for bone marrow failure, and increased risk of blood cancers. What is important about this work, however, is that it allows us to quantify the effect of stem cell transplant on molecular age.” ![]()

Photo by Chad McNeeley
New research suggests hematopoietic stem cell transplant (HSCT) may increase the molecular age of peripheral blood T cells.
The study showed an increase in peripheral blood T-cell senescence in patients with hematologic malignancies who were treated with autologous (auto-) or allogeneic (allo-) HSCT.
The patients had elevated levels of p16INK4a, a known marker of cellular senescence.
Auto-HSCT in particular had a strong effect on p16INK4a, increasing the expression of this marker to a degree comparable to 30 years of chronological aging.
Researchers reported these findings in EBioMedicine.
“We know that transplant is life-prolonging, and, in many cases, it’s life-saving for many patients with blood cancers and other disorders,” said study author William Wood, MD, of the University of North Carolina School of Medicine in Chapel Hill.
“At the same time, we’re increasingly recognizing that survivors of transplant are at risk for long-term health problems, and so there is interest in determining what markers may exist to help predict risk for long-term health problems or even in helping choose which patients are best candidates for transplantation.”
With this in mind, Dr Wood and his colleagues looked at levels of p16INK4a in 63 patients who underwent auto- or allo-HSCT to treat myeloma, lymphoma, or leukemia. The researchers assessed p16INK4a expression in T cells before HSCT and 6 months after.
Among auto-HSCT recipients, there were no baseline characteristics associated with pre-transplant p16INK4a expression.
However, allo-HSCT recipients had significantly higher pre-transplant p16INK4a levels the more cycles of chemotherapy they received before transplant (P=0.003), if they had previously undergone auto-HSCT (P=0.01), and if they had been exposed to alkylating agents (P=0.01).
After transplant, allo-HSCT recipients had a 1.93-fold increase in p16INK4a expression (P=0.0004), and auto-HSCT recipients had a 3.05-fold increase (P=0.002).
The researchers said the measured change in p16INK4a from pre- to post-HSCT in allogeneic recipients likely underestimates the age-promoting effects of HSCT, given that the pre-HSCT levels were elevated in the recipients from prior therapeutic exposure.
The researchers also pointed out that this study does not show a clear connection between changes in p16INK4a levels and the actual function of peripheral blood T cells, but they did say that p16INK4a is “arguably one of the best in vivo markers of cellular senescence and is directly associated with age-related deterioration.”
So the results of this research suggest the forced bone marrow repopulation associated with HSCT accelerates the molecular aging of peripheral blood T cells.
“Many oncologists would not be surprised by the finding that stem cell transplant accelerates aspects of aging,” said study author Norman Sharpless, MD, of the University of North Carolina School of Medicine.
“We know that, years after a curative transplant, stem cell transplant survivors are at increased risk for blood problems that can occur with aging, such as reduced immunity, increased risk for bone marrow failure, and increased risk of blood cancers. What is important about this work, however, is that it allows us to quantify the effect of stem cell transplant on molecular age.” ![]()
Study reveals higher risk of injuries pre- and post-cancer diagnosis

Photo courtesy of the CDC
Cancer patients have a heightened risk of injuries 16 weeks before and after their diagnosis, according to a large study.
This includes injuries arising from medical complications and treatments, such as infections or bleeding after invasive treatment, and other types of injuries, such as bruising or fractures from self-harm and accidents.
Fang Fang, MD, PhD, of Karolinska Institutet in Stockholm, Sweden, and her colleagues reported these findings in The BMJ.
The researchers analyzed all injury-related hospital admissions in Swedish patients with cancer between 1990 and 2010. The team compared a diagnostic period—16 weeks before and after diagnosis—with a control period the year before diagnosis.
Among 720,901 patients, there were 7306 injuries from medical complications and drug treatments and 8331 injuries from accidents and self-harm that resulted in hospital admission during the diagnostic period.
Patients with central nervous system cancers had the highest risk of medical-related injuries—a 14.7-fold higher risk during the diagnostic period than the control period.
Patients with lymphatic or hematopoietic cancers had a 4-fold higher risk of such injuries during the diagnostic period than during the control period.
Patients who were younger, were cohabiting, had a higher socioeconomic status or education, and had no pre-existing psychiatric disorder had a higher risk of medical-related injuries during the diagnostic period than other groups of patients.
The risk of other types of injuries from self-harm and accidents was also higher during the diagnostic period. There was a 5.3-fold increased risk during the 2 weeks before diagnosis. The researchers said this suggests that psychological stress is high when patients are expecting a diagnosis.
Patients with lymphatic or hematopoietic cancers and patients with central nervous system cancers had the highest risk of self-harm and accidental injuries—a 2.8-fold increased risk during the diagnostic period compared to the control period (for both groups).
Older patients and those with lower socioeconomic status or education had slightly greater increases in the risk of self-harm and accidental injuries compared to other groups.
The researchers said the estimates of risk in this study are conservative because the team did not account for injuries that failed to result in a hospital admission or for those that were fatal.
Furthermore, this was an observational study, so no firm conclusions about cause and effect can be made.
Still, the researchers said this study sheds light on which patients might be at an increased risk of injuries, providing evidence for clinicians and policy makers to develop targeted prevention strategies. ![]()

Photo courtesy of the CDC
Cancer patients have a heightened risk of injuries 16 weeks before and after their diagnosis, according to a large study.
This includes injuries arising from medical complications and treatments, such as infections or bleeding after invasive treatment, and other types of injuries, such as bruising or fractures from self-harm and accidents.
Fang Fang, MD, PhD, of Karolinska Institutet in Stockholm, Sweden, and her colleagues reported these findings in The BMJ.
The researchers analyzed all injury-related hospital admissions in Swedish patients with cancer between 1990 and 2010. The team compared a diagnostic period—16 weeks before and after diagnosis—with a control period the year before diagnosis.
Among 720,901 patients, there were 7306 injuries from medical complications and drug treatments and 8331 injuries from accidents and self-harm that resulted in hospital admission during the diagnostic period.
Patients with central nervous system cancers had the highest risk of medical-related injuries—a 14.7-fold higher risk during the diagnostic period than the control period.
Patients with lymphatic or hematopoietic cancers had a 4-fold higher risk of such injuries during the diagnostic period than during the control period.
Patients who were younger, were cohabiting, had a higher socioeconomic status or education, and had no pre-existing psychiatric disorder had a higher risk of medical-related injuries during the diagnostic period than other groups of patients.
The risk of other types of injuries from self-harm and accidents was also higher during the diagnostic period. There was a 5.3-fold increased risk during the 2 weeks before diagnosis. The researchers said this suggests that psychological stress is high when patients are expecting a diagnosis.
Patients with lymphatic or hematopoietic cancers and patients with central nervous system cancers had the highest risk of self-harm and accidental injuries—a 2.8-fold increased risk during the diagnostic period compared to the control period (for both groups).
Older patients and those with lower socioeconomic status or education had slightly greater increases in the risk of self-harm and accidental injuries compared to other groups.
The researchers said the estimates of risk in this study are conservative because the team did not account for injuries that failed to result in a hospital admission or for those that were fatal.
Furthermore, this was an observational study, so no firm conclusions about cause and effect can be made.
Still, the researchers said this study sheds light on which patients might be at an increased risk of injuries, providing evidence for clinicians and policy makers to develop targeted prevention strategies. ![]()

Photo courtesy of the CDC
Cancer patients have a heightened risk of injuries 16 weeks before and after their diagnosis, according to a large study.
This includes injuries arising from medical complications and treatments, such as infections or bleeding after invasive treatment, and other types of injuries, such as bruising or fractures from self-harm and accidents.
Fang Fang, MD, PhD, of Karolinska Institutet in Stockholm, Sweden, and her colleagues reported these findings in The BMJ.
The researchers analyzed all injury-related hospital admissions in Swedish patients with cancer between 1990 and 2010. The team compared a diagnostic period—16 weeks before and after diagnosis—with a control period the year before diagnosis.
Among 720,901 patients, there were 7306 injuries from medical complications and drug treatments and 8331 injuries from accidents and self-harm that resulted in hospital admission during the diagnostic period.
Patients with central nervous system cancers had the highest risk of medical-related injuries—a 14.7-fold higher risk during the diagnostic period than the control period.
Patients with lymphatic or hematopoietic cancers had a 4-fold higher risk of such injuries during the diagnostic period than during the control period.
Patients who were younger, were cohabiting, had a higher socioeconomic status or education, and had no pre-existing psychiatric disorder had a higher risk of medical-related injuries during the diagnostic period than other groups of patients.
The risk of other types of injuries from self-harm and accidents was also higher during the diagnostic period. There was a 5.3-fold increased risk during the 2 weeks before diagnosis. The researchers said this suggests that psychological stress is high when patients are expecting a diagnosis.
Patients with lymphatic or hematopoietic cancers and patients with central nervous system cancers had the highest risk of self-harm and accidental injuries—a 2.8-fold increased risk during the diagnostic period compared to the control period (for both groups).
Older patients and those with lower socioeconomic status or education had slightly greater increases in the risk of self-harm and accidental injuries compared to other groups.
The researchers said the estimates of risk in this study are conservative because the team did not account for injuries that failed to result in a hospital admission or for those that were fatal.
Furthermore, this was an observational study, so no firm conclusions about cause and effect can be made.
Still, the researchers said this study sheds light on which patients might be at an increased risk of injuries, providing evidence for clinicians and policy makers to develop targeted prevention strategies.
FDA approves new indication for ofatumumab in CLL

Photo courtesy of GSK
The US Food and Drug Administration (FDA) has approved the use of ofatumumab (Arzerra®) in combination with fludarabine and cyclophosphamide to treat patients with relapsed chronic lymphocytic leukemia (CLL).
Ofatumumab was previously approved by the FDA for use in combination with chlorambucil to treat previously untreated CLL patients who cannot receive fludarabine-based therapy, as monotherapy for CLL that is refractory to fludarabine and alemtuzumab, and as maintenance therapy for patients who are in complete or partial response after receiving at least 2 lines of therapy for recurrent or progressive CLL.
Ofatumumab is a monoclonal antibody designed to target CD20.
The drug’s prescribing information includes a boxed warning noting that hepatitis B virus reactivation can occur in patients receiving CD20-directed cytolytic antibodies, including ofatumumab. In some cases, this results in fulminant hepatitis, hepatic failure, and death.
The boxed warning also states that progressive multifocal leukoencephalopathy, resulting in death, can occur in patients receiving CD20-directed cytolytic antibodies, including ofatumumab.
Ofatumumab is marketed under a collaboration agreement between Genmab and Novartis.
COMPLEMENT 2 trial
The FDA’s latest approval for ofatumumab is based on results of the phase 3 COMPLEMENT 2 trial. Novartis reported top-line results from this study in April.
The trial enrolled 365 patients with relapsed CLL. The patients were randomized 1:1 to receive up to 6 cycles of ofatumumab in combination with fludarabine and cyclophosphamide or up to 6 cycles of fludarabine and cyclophosphamide alone.
The primary endpoint was progression-free survival, as assessed by an independent review committee.
The median progression-free survival was 28.9 months for patients receiving ofatumumab plus fludarabine and cyclophosphamide, compared to 18.8 months for patients receiving fludarabine and cyclophosphamide alone (hazard ratio=0.67, P=0.0032).
Novartis said the safety profile observed in this study was consistent with other trials of ofatumumab, and no new safety signals were observed.

Photo courtesy of GSK
The US Food and Drug Administration (FDA) has approved the use of ofatumumab (Arzerra®) in combination with fludarabine and cyclophosphamide to treat patients with relapsed chronic lymphocytic leukemia (CLL).
Ofatumumab was previously approved by the FDA for use in combination with chlorambucil to treat previously untreated CLL patients who cannot receive fludarabine-based therapy, as monotherapy for CLL that is refractory to fludarabine and alemtuzumab, and as maintenance therapy for patients who are in complete or partial response after receiving at least 2 lines of therapy for recurrent or progressive CLL.
Ofatumumab is a monoclonal antibody designed to target CD20.
The drug’s prescribing information includes a boxed warning noting that hepatitis B virus reactivation can occur in patients receiving CD20-directed cytolytic antibodies, including ofatumumab. In some cases, this results in fulminant hepatitis, hepatic failure, and death.
The boxed warning also states that progressive multifocal leukoencephalopathy, resulting in death, can occur in patients receiving CD20-directed cytolytic antibodies, including ofatumumab.
Ofatumumab is marketed under a collaboration agreement between Genmab and Novartis.
COMPLEMENT 2 trial
The FDA’s latest approval for ofatumumab is based on results of the phase 3 COMPLEMENT 2 trial. Novartis reported top-line results from this study in April.
The trial enrolled 365 patients with relapsed CLL. The patients were randomized 1:1 to receive up to 6 cycles of ofatumumab in combination with fludarabine and cyclophosphamide or up to 6 cycles of fludarabine and cyclophosphamide alone.
The primary endpoint was progression-free survival, as assessed by an independent review committee.
The median progression-free survival was 28.9 months for patients receiving ofatumumab plus fludarabine and cyclophosphamide, compared to 18.8 months for patients receiving fludarabine and cyclophosphamide alone (hazard ratio=0.67, P=0.0032).
Novartis said the safety profile observed in this study was consistent with other trials of ofatumumab, and no new safety signals were observed.

Photo courtesy of GSK
The US Food and Drug Administration (FDA) has approved the use of ofatumumab (Arzerra®) in combination with fludarabine and cyclophosphamide to treat patients with relapsed chronic lymphocytic leukemia (CLL).
Ofatumumab was previously approved by the FDA for use in combination with chlorambucil to treat previously untreated CLL patients who cannot receive fludarabine-based therapy, as monotherapy for CLL that is refractory to fludarabine and alemtuzumab, and as maintenance therapy for patients who are in complete or partial response after receiving at least 2 lines of therapy for recurrent or progressive CLL.
Ofatumumab is a monoclonal antibody designed to target CD20.
The drug’s prescribing information includes a boxed warning noting that hepatitis B virus reactivation can occur in patients receiving CD20-directed cytolytic antibodies, including ofatumumab. In some cases, this results in fulminant hepatitis, hepatic failure, and death.
The boxed warning also states that progressive multifocal leukoencephalopathy, resulting in death, can occur in patients receiving CD20-directed cytolytic antibodies, including ofatumumab.
Ofatumumab is marketed under a collaboration agreement between Genmab and Novartis.
COMPLEMENT 2 trial
The FDA’s latest approval for ofatumumab is based on results of the phase 3 COMPLEMENT 2 trial. Novartis reported top-line results from this study in April.
The trial enrolled 365 patients with relapsed CLL. The patients were randomized 1:1 to receive up to 6 cycles of ofatumumab in combination with fludarabine and cyclophosphamide or up to 6 cycles of fludarabine and cyclophosphamide alone.
The primary endpoint was progression-free survival, as assessed by an independent review committee.
The median progression-free survival was 28.9 months for patients receiving ofatumumab plus fludarabine and cyclophosphamide, compared to 18.8 months for patients receiving fludarabine and cyclophosphamide alone (hazard ratio=0.67, P=0.0032).
Novartis said the safety profile observed in this study was consistent with other trials of ofatumumab, and no new safety signals were observed.