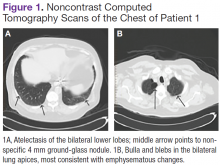
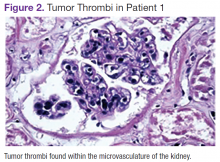
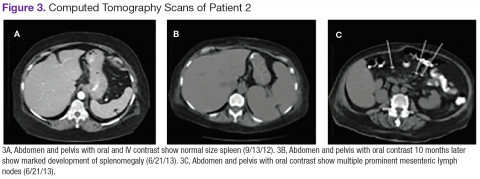
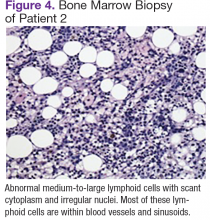
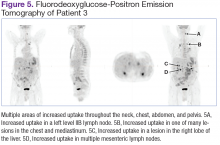
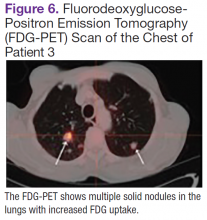
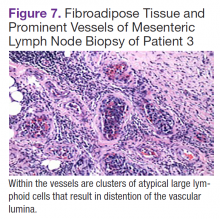
User login
Veterans don’t have higher risk of leukemia, lymphoma
People who have served in the Armed Forces do not have an increased risk of leukemia or lymphoma, according to research published in Cancer Epidemiology.
Researchers analyzed the long-term risks of developing leukemia, Hodgkin lymphoma (HL), and non-Hodgkin lymphoma (NHL) in veterans living in Scotland.
At a mean 30 years of follow-up, there were no significant differences in the risk of the aforementioned malignancies between veterans and non-veterans in Scotland.
This retrospective study included 56,205 veterans and 172,741 non-veterans.
The veterans’ earliest date of entering service was January 1960, and the latest date of leaving service was December 2012.
At a mean follow-up of 29.3 years, 294 (0.52%) veterans and 974 (0.56%) non-veterans were diagnosed with leukemia, HL, or NHL.
There were 125 (0.22%) cases of leukemia in veterans and 365 (0.21%) in non-veterans. There were 59 (0.10%) cases of HL in veterans and 182 (0.11%) in non-veterans. And there were 144 (0.26%) cases of NHL in veterans and 538 (0.31%) in non-veterans.
There was no significant difference in the risk of all 3 cancer types between the veterans and non-veterans. The unadjusted hazard ratio (HR) was 0.96 (P=0.541).
There were no significant differences in an adjusted analysis either. (The analysis was adjusted for regional deprivation, which takes into account information on income, employment, health, education, housing, crime, and access to services.)
The adjusted HR was 1.03 (P=0.773) for leukemias, 1.19 (P=0.272) for HL, and 0.86 (P=0.110) for NHL.
“This is an important study which provides reassurance that military service in the last 50 years does not increase people’s risk of leukemia overall,” said study author Beverly Bergman, PhD, of the University of Glasgow in the UK.
“The Armed Forces comply with all relevant health and safety legislation and regulations, and we can now see that their risk is no different from the general population.” ![]()
People who have served in the Armed Forces do not have an increased risk of leukemia or lymphoma, according to research published in Cancer Epidemiology.
Researchers analyzed the long-term risks of developing leukemia, Hodgkin lymphoma (HL), and non-Hodgkin lymphoma (NHL) in veterans living in Scotland.
At a mean 30 years of follow-up, there were no significant differences in the risk of the aforementioned malignancies between veterans and non-veterans in Scotland.
This retrospective study included 56,205 veterans and 172,741 non-veterans.
The veterans’ earliest date of entering service was January 1960, and the latest date of leaving service was December 2012.
At a mean follow-up of 29.3 years, 294 (0.52%) veterans and 974 (0.56%) non-veterans were diagnosed with leukemia, HL, or NHL.
There were 125 (0.22%) cases of leukemia in veterans and 365 (0.21%) in non-veterans. There were 59 (0.10%) cases of HL in veterans and 182 (0.11%) in non-veterans. And there were 144 (0.26%) cases of NHL in veterans and 538 (0.31%) in non-veterans.
There was no significant difference in the risk of all 3 cancer types between the veterans and non-veterans. The unadjusted hazard ratio (HR) was 0.96 (P=0.541).
There were no significant differences in an adjusted analysis either. (The analysis was adjusted for regional deprivation, which takes into account information on income, employment, health, education, housing, crime, and access to services.)
The adjusted HR was 1.03 (P=0.773) for leukemias, 1.19 (P=0.272) for HL, and 0.86 (P=0.110) for NHL.
“This is an important study which provides reassurance that military service in the last 50 years does not increase people’s risk of leukemia overall,” said study author Beverly Bergman, PhD, of the University of Glasgow in the UK.
“The Armed Forces comply with all relevant health and safety legislation and regulations, and we can now see that their risk is no different from the general population.” ![]()
People who have served in the Armed Forces do not have an increased risk of leukemia or lymphoma, according to research published in Cancer Epidemiology.
Researchers analyzed the long-term risks of developing leukemia, Hodgkin lymphoma (HL), and non-Hodgkin lymphoma (NHL) in veterans living in Scotland.
At a mean 30 years of follow-up, there were no significant differences in the risk of the aforementioned malignancies between veterans and non-veterans in Scotland.
This retrospective study included 56,205 veterans and 172,741 non-veterans.
The veterans’ earliest date of entering service was January 1960, and the latest date of leaving service was December 2012.
At a mean follow-up of 29.3 years, 294 (0.52%) veterans and 974 (0.56%) non-veterans were diagnosed with leukemia, HL, or NHL.
There were 125 (0.22%) cases of leukemia in veterans and 365 (0.21%) in non-veterans. There were 59 (0.10%) cases of HL in veterans and 182 (0.11%) in non-veterans. And there were 144 (0.26%) cases of NHL in veterans and 538 (0.31%) in non-veterans.
There was no significant difference in the risk of all 3 cancer types between the veterans and non-veterans. The unadjusted hazard ratio (HR) was 0.96 (P=0.541).
There were no significant differences in an adjusted analysis either. (The analysis was adjusted for regional deprivation, which takes into account information on income, employment, health, education, housing, crime, and access to services.)
The adjusted HR was 1.03 (P=0.773) for leukemias, 1.19 (P=0.272) for HL, and 0.86 (P=0.110) for NHL.
“This is an important study which provides reassurance that military service in the last 50 years does not increase people’s risk of leukemia overall,” said study author Beverly Bergman, PhD, of the University of Glasgow in the UK.
“The Armed Forces comply with all relevant health and safety legislation and regulations, and we can now see that their risk is no different from the general population.” ![]()
FDA approves pembrolizumab to treat cHL
The US Food and Drug Administration (FDA) has granted accelerated approval for pembrolizumab (Keytruda) as a treatment for adult and pediatric patients with relapsed or refractory classical Hodgkin lymphoma (cHL).
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the antitumor immune response.
The drug, which is being developed by Merck, previously received FDA approval as a treatment for melanoma, lung cancer, and head and neck cancer.
Now, pembrolizumab has received accelerated approval to treat adult and pediatric patients with refractory cHL or those with cHL who have relapsed after 3 or more prior lines of therapy.
The accelerated approval was based on tumor response rate and durability of response. Continued approval of pembrolizumab for cHL patients may be contingent upon the verification and description of clinical benefit in confirmatory trials.
In adults with cHL, pembrolizumab is administered at a fixed dose of 200 mg every 3 weeks until disease progression or unacceptable toxicity, or up to 24 months in patients without disease progression.
In pediatric patients with cHL, pembrolizumab is administered at a dose of 2 mg/kg (up to a maximum of 200 mg) every 3 weeks until disease progression or unacceptable toxicity, or up to 24 months in patients without disease progression.
Pembrolizumab trials
The FDA’s approval of pembrolizumab in adults with cHL is based on data from the phase 2 KEYNOTE-087 trial. (The following data were provided by Merck.)
The trial enrolled 210 patients who received pembrolizumab at a dose of 200 mg every 3 weeks until unacceptable toxicity or documented disease progression, or for up to 24 months in patients who did not progress.
Fifty-eight percent of patients were refractory to their last prior therapy, including 35% with primary refractory disease and 14% whose disease was refractory to all prior regimens.
Sixty-one percent of patients had undergone prior autologous hematopoietic stem cell transplant, 83% had prior brentuximab use, and 36% had prior radiation therapy.
At a median follow-up of 9.4 months, the overall response rate was 69%, and the complete response rate was 22%. The median duration of response was 11.1 months (range, 0.0+ to 11.1 months).
Five percent of patients discontinued pembrolizumab due to adverse events (AEs), and 26% had dose interruptions due to AEs. Fifteen percent of patients had an AE requiring systemic corticosteroid therapy.
The most common AEs (occurring in ≥20% of patients) were fatigue (26%), pyrexia (24%), cough (24%), musculoskeletal pain (21%), diarrhea (20%), and rash (20%).
Serious AEs occurred in 16% of patients. The most frequent serious AEs (≥1%) were pneumonia, pneumonitis, pyrexia, dyspnea, graft-vs-host disease, and herpes zoster.
Two patients died from causes other than disease progression. One death was a result of graft-vs-host disease after subsequent allogeneic transplant, and the other was from septic shock.
There is limited experience with pembrolizumab in pediatric patients. The efficacy of the drug for pediatric patients was extrapolated from the results in the adult cHL population.
However, there is safety data on pembrolizumab in pediatric patients enrolled in the phase 1/2 KEYNOTE-051 trial. (These data were also provided by Merck.)
The trial included 40 pediatric patients with advanced melanoma or PD-L1–positive advanced, relapsed, or refractory solid tumors or lymphoma. Patients in this trial received pembrolizumab for a median of 43 days (range, 1-414 days).
The safety profile in these patients was similar to the profile in adults. Toxicities that occurred at a higher rate (≥15% difference) in pediatric patients than in adults under age 65 were fatigue (45%), vomiting (38%), abdominal pain (28%), hypertransaminasemia (28%), and hyponatremia (18%). ![]()
The US Food and Drug Administration (FDA) has granted accelerated approval for pembrolizumab (Keytruda) as a treatment for adult and pediatric patients with relapsed or refractory classical Hodgkin lymphoma (cHL).
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the antitumor immune response.
The drug, which is being developed by Merck, previously received FDA approval as a treatment for melanoma, lung cancer, and head and neck cancer.
Now, pembrolizumab has received accelerated approval to treat adult and pediatric patients with refractory cHL or those with cHL who have relapsed after 3 or more prior lines of therapy.
The accelerated approval was based on tumor response rate and durability of response. Continued approval of pembrolizumab for cHL patients may be contingent upon the verification and description of clinical benefit in confirmatory trials.
In adults with cHL, pembrolizumab is administered at a fixed dose of 200 mg every 3 weeks until disease progression or unacceptable toxicity, or up to 24 months in patients without disease progression.
In pediatric patients with cHL, pembrolizumab is administered at a dose of 2 mg/kg (up to a maximum of 200 mg) every 3 weeks until disease progression or unacceptable toxicity, or up to 24 months in patients without disease progression.
Pembrolizumab trials
The FDA’s approval of pembrolizumab in adults with cHL is based on data from the phase 2 KEYNOTE-087 trial. (The following data were provided by Merck.)
The trial enrolled 210 patients who received pembrolizumab at a dose of 200 mg every 3 weeks until unacceptable toxicity or documented disease progression, or for up to 24 months in patients who did not progress.
Fifty-eight percent of patients were refractory to their last prior therapy, including 35% with primary refractory disease and 14% whose disease was refractory to all prior regimens.
Sixty-one percent of patients had undergone prior autologous hematopoietic stem cell transplant, 83% had prior brentuximab use, and 36% had prior radiation therapy.
At a median follow-up of 9.4 months, the overall response rate was 69%, and the complete response rate was 22%. The median duration of response was 11.1 months (range, 0.0+ to 11.1 months).
Five percent of patients discontinued pembrolizumab due to adverse events (AEs), and 26% had dose interruptions due to AEs. Fifteen percent of patients had an AE requiring systemic corticosteroid therapy.
The most common AEs (occurring in ≥20% of patients) were fatigue (26%), pyrexia (24%), cough (24%), musculoskeletal pain (21%), diarrhea (20%), and rash (20%).
Serious AEs occurred in 16% of patients. The most frequent serious AEs (≥1%) were pneumonia, pneumonitis, pyrexia, dyspnea, graft-vs-host disease, and herpes zoster.
Two patients died from causes other than disease progression. One death was a result of graft-vs-host disease after subsequent allogeneic transplant, and the other was from septic shock.
There is limited experience with pembrolizumab in pediatric patients. The efficacy of the drug for pediatric patients was extrapolated from the results in the adult cHL population.
However, there is safety data on pembrolizumab in pediatric patients enrolled in the phase 1/2 KEYNOTE-051 trial. (These data were also provided by Merck.)
The trial included 40 pediatric patients with advanced melanoma or PD-L1–positive advanced, relapsed, or refractory solid tumors or lymphoma. Patients in this trial received pembrolizumab for a median of 43 days (range, 1-414 days).
The safety profile in these patients was similar to the profile in adults. Toxicities that occurred at a higher rate (≥15% difference) in pediatric patients than in adults under age 65 were fatigue (45%), vomiting (38%), abdominal pain (28%), hypertransaminasemia (28%), and hyponatremia (18%). ![]()
The US Food and Drug Administration (FDA) has granted accelerated approval for pembrolizumab (Keytruda) as a treatment for adult and pediatric patients with relapsed or refractory classical Hodgkin lymphoma (cHL).
Pembrolizumab is a monoclonal antibody that binds to the PD-1 receptor and blocks its interaction with PD-L1 and PD-L2, releasing PD-1 pathway-mediated inhibition of the immune response, including the antitumor immune response.
The drug, which is being developed by Merck, previously received FDA approval as a treatment for melanoma, lung cancer, and head and neck cancer.
Now, pembrolizumab has received accelerated approval to treat adult and pediatric patients with refractory cHL or those with cHL who have relapsed after 3 or more prior lines of therapy.
The accelerated approval was based on tumor response rate and durability of response. Continued approval of pembrolizumab for cHL patients may be contingent upon the verification and description of clinical benefit in confirmatory trials.
In adults with cHL, pembrolizumab is administered at a fixed dose of 200 mg every 3 weeks until disease progression or unacceptable toxicity, or up to 24 months in patients without disease progression.
In pediatric patients with cHL, pembrolizumab is administered at a dose of 2 mg/kg (up to a maximum of 200 mg) every 3 weeks until disease progression or unacceptable toxicity, or up to 24 months in patients without disease progression.
Pembrolizumab trials
The FDA’s approval of pembrolizumab in adults with cHL is based on data from the phase 2 KEYNOTE-087 trial. (The following data were provided by Merck.)
The trial enrolled 210 patients who received pembrolizumab at a dose of 200 mg every 3 weeks until unacceptable toxicity or documented disease progression, or for up to 24 months in patients who did not progress.
Fifty-eight percent of patients were refractory to their last prior therapy, including 35% with primary refractory disease and 14% whose disease was refractory to all prior regimens.
Sixty-one percent of patients had undergone prior autologous hematopoietic stem cell transplant, 83% had prior brentuximab use, and 36% had prior radiation therapy.
At a median follow-up of 9.4 months, the overall response rate was 69%, and the complete response rate was 22%. The median duration of response was 11.1 months (range, 0.0+ to 11.1 months).
Five percent of patients discontinued pembrolizumab due to adverse events (AEs), and 26% had dose interruptions due to AEs. Fifteen percent of patients had an AE requiring systemic corticosteroid therapy.
The most common AEs (occurring in ≥20% of patients) were fatigue (26%), pyrexia (24%), cough (24%), musculoskeletal pain (21%), diarrhea (20%), and rash (20%).
Serious AEs occurred in 16% of patients. The most frequent serious AEs (≥1%) were pneumonia, pneumonitis, pyrexia, dyspnea, graft-vs-host disease, and herpes zoster.
Two patients died from causes other than disease progression. One death was a result of graft-vs-host disease after subsequent allogeneic transplant, and the other was from septic shock.
There is limited experience with pembrolizumab in pediatric patients. The efficacy of the drug for pediatric patients was extrapolated from the results in the adult cHL population.
However, there is safety data on pembrolizumab in pediatric patients enrolled in the phase 1/2 KEYNOTE-051 trial. (These data were also provided by Merck.)
The trial included 40 pediatric patients with advanced melanoma or PD-L1–positive advanced, relapsed, or refractory solid tumors or lymphoma. Patients in this trial received pembrolizumab for a median of 43 days (range, 1-414 days).
The safety profile in these patients was similar to the profile in adults. Toxicities that occurred at a higher rate (≥15% difference) in pediatric patients than in adults under age 65 were fatigue (45%), vomiting (38%), abdominal pain (28%), hypertransaminasemia (28%), and hyponatremia (18%). ![]()
Family history impacts risk of second cancer after HL
A new study suggests Hodgkin lymphoma (HL) survivors have a high risk of developing a second malignancy, particularly if they have a family history of that malignancy.
The research showed that HL survivors in Sweden were roughly 2.4 times more likely than individuals in the country’s general population to develop a second cancer.
The risk for HL survivors remained high 30 years after treatment, and the risk was even greater in HL survivors who had a family history of specific cancers.
“The vast majority of patients with Hodgkin lymphoma are cured with a combination of chemotherapy and radiotherapy,” said study author Amit Sud, MBChB, of The Institute of Cancer Research, London in the UK.
“Our research has shown that these patients are at substantially increased risk of a second cancer later in life and particularly if they have a family history of cancer.”
Dr Sud and his colleagues described this research in the Journal of Clinical Oncology.
The team analyzed data from the Swedish Family-Cancer Project Database. They identified 9522 HL patients diagnosed between 1965 and 2013. During a median follow-up of 12.6 years, there were 1215 second cancers in 1121 HL patients (12%).
Compared to the general population, the HL patients had a significantly higher risk of all second malignancies, with a standardized incident ratio (SIR) of 2.39 and an absolute excess risk of 71.2 cases per 10,000 person-years.
Cancer types
HL patients had a significantly increased risk of several malignancies. The overall SIRs were as follows:
- NHL—7.99
- Leukemia—6.46
- Connective tissue cancer—5.73
- Thyroid cancer—5.13
- Squamous cell carcinoma—4.44
- Lung cancer—3.61
- Pharyngeal cancer—3.52
- Esophageal cancer—2.62
- Brain cancer—2.58
- Breast cancer—2.52
- Colon cancer—2.21
- Pancreatic cancer—2.09
- Melanoma—2.08
- Colorectal cancer—1.85
- Stomach cancer—1.78
- Bladder cancer—1.57
- Prostate cancer—1.21.
The researchers calculated SIRs over time and found the risk for many of the cancers remained high over 30 years following HL treatment.
Family history
The researchers identified 28,277 first-degree relatives of the HL survivors. Thirty percent of HL survivors (n=2785) had 1 or more first-degree relatives with a family history of cancer.
The SIR for cancers was 1.02 in the relatives. The SIR for second cancers was 2.83 for HL survivors who had first-degree relatives with cancer and 2.16 for HL survivors who did not have any first-degree relatives with cancer.
The researchers said the increased risk of second malignancy was correlated with the number of first-degree relatives with cancer.
The SIR was 2.67 for HL patients who had a single first-degree relative with cancer and 3.40 for HL patients who had 2 or more first-degree relatives with cancer.
The SIRs for different cancer types (for HL patients with at least 1 first-degree relative with cancer and no first-degree relatives with cancer, respectively) were as follows:
- NHL—14.43 vs 7.83
- Leukemia—14.31 vs 6.37
- Squamous cell carcinoma—10.85 vs 4.30
- Lung cancer—11.24 vs 3.39
- Breast cancer—4.36 vs 2.36
- Colorectal cancer—3.71 vs 1.76.
Sex and age
The researchers found significant differences in the SIRs for second cancers between HL patients diagnosed before the age of 35 and those diagnosed after age 35.
For men, the SIRs were:
- All cancers—4.26 for <35, 2.08 for ≥ 35
- Colorectal cancer—4.07 for < 35, 1.73 for ≥35
- Lung cancer—6.16 for < 35, 3.20 for ≥35
- Breast cancer—12.60 for < 35, 4.58 for ≥35
- Squamous cell carcinoma—5.89 for < 35, 3.96 for ≥35
- NHL—15.9 for < 35, 6.93 for ≥35
- Leukemia—12.15 for < 35, 5.57 for ≥35.
For women, the SIRs were:
- All cancers—4.61 for <35, 1.73 for ≥ 35
- Colorectal cancer—1.31 for < 35, 1.65 for ≥35
- Lung cancer—8.84 for < 35, 2.50 for ≥35
- Breast cancer—6.00 for < 35, 1.14 for ≥35
- Squamous cell carcinoma—6.37 for < 35, 4.87 for ≥35
- NHL—6.23 for < 35, 6.55 for ≥35
- Leukemia—10.36 for < 35, 4.51 for ≥35.
“Younger women who have been treated with radiotherapy to the chest for Hodgkin lymphoma are already screened for breast cancer, but our study suggests that we should be looking at ways of monitoring survivors for other forms of cancer too, and potentially offering preventative interventions,” Dr Sud said.
“After patients are cured, they no longer encounter oncologists, so it’s important that other healthcare providers are aware of the increased risk to Hodgkin lymphoma survivors to improve early diagnosis of second cancers.” ![]()
A new study suggests Hodgkin lymphoma (HL) survivors have a high risk of developing a second malignancy, particularly if they have a family history of that malignancy.
The research showed that HL survivors in Sweden were roughly 2.4 times more likely than individuals in the country’s general population to develop a second cancer.
The risk for HL survivors remained high 30 years after treatment, and the risk was even greater in HL survivors who had a family history of specific cancers.
“The vast majority of patients with Hodgkin lymphoma are cured with a combination of chemotherapy and radiotherapy,” said study author Amit Sud, MBChB, of The Institute of Cancer Research, London in the UK.
“Our research has shown that these patients are at substantially increased risk of a second cancer later in life and particularly if they have a family history of cancer.”
Dr Sud and his colleagues described this research in the Journal of Clinical Oncology.
The team analyzed data from the Swedish Family-Cancer Project Database. They identified 9522 HL patients diagnosed between 1965 and 2013. During a median follow-up of 12.6 years, there were 1215 second cancers in 1121 HL patients (12%).
Compared to the general population, the HL patients had a significantly higher risk of all second malignancies, with a standardized incident ratio (SIR) of 2.39 and an absolute excess risk of 71.2 cases per 10,000 person-years.
Cancer types
HL patients had a significantly increased risk of several malignancies. The overall SIRs were as follows:
- NHL—7.99
- Leukemia—6.46
- Connective tissue cancer—5.73
- Thyroid cancer—5.13
- Squamous cell carcinoma—4.44
- Lung cancer—3.61
- Pharyngeal cancer—3.52
- Esophageal cancer—2.62
- Brain cancer—2.58
- Breast cancer—2.52
- Colon cancer—2.21
- Pancreatic cancer—2.09
- Melanoma—2.08
- Colorectal cancer—1.85
- Stomach cancer—1.78
- Bladder cancer—1.57
- Prostate cancer—1.21.
The researchers calculated SIRs over time and found the risk for many of the cancers remained high over 30 years following HL treatment.
Family history
The researchers identified 28,277 first-degree relatives of the HL survivors. Thirty percent of HL survivors (n=2785) had 1 or more first-degree relatives with a family history of cancer.
The SIR for cancers was 1.02 in the relatives. The SIR for second cancers was 2.83 for HL survivors who had first-degree relatives with cancer and 2.16 for HL survivors who did not have any first-degree relatives with cancer.
The researchers said the increased risk of second malignancy was correlated with the number of first-degree relatives with cancer.
The SIR was 2.67 for HL patients who had a single first-degree relative with cancer and 3.40 for HL patients who had 2 or more first-degree relatives with cancer.
The SIRs for different cancer types (for HL patients with at least 1 first-degree relative with cancer and no first-degree relatives with cancer, respectively) were as follows:
- NHL—14.43 vs 7.83
- Leukemia—14.31 vs 6.37
- Squamous cell carcinoma—10.85 vs 4.30
- Lung cancer—11.24 vs 3.39
- Breast cancer—4.36 vs 2.36
- Colorectal cancer—3.71 vs 1.76.
Sex and age
The researchers found significant differences in the SIRs for second cancers between HL patients diagnosed before the age of 35 and those diagnosed after age 35.
For men, the SIRs were:
- All cancers—4.26 for <35, 2.08 for ≥ 35
- Colorectal cancer—4.07 for < 35, 1.73 for ≥35
- Lung cancer—6.16 for < 35, 3.20 for ≥35
- Breast cancer—12.60 for < 35, 4.58 for ≥35
- Squamous cell carcinoma—5.89 for < 35, 3.96 for ≥35
- NHL—15.9 for < 35, 6.93 for ≥35
- Leukemia—12.15 for < 35, 5.57 for ≥35.
For women, the SIRs were:
- All cancers—4.61 for <35, 1.73 for ≥ 35
- Colorectal cancer—1.31 for < 35, 1.65 for ≥35
- Lung cancer—8.84 for < 35, 2.50 for ≥35
- Breast cancer—6.00 for < 35, 1.14 for ≥35
- Squamous cell carcinoma—6.37 for < 35, 4.87 for ≥35
- NHL—6.23 for < 35, 6.55 for ≥35
- Leukemia—10.36 for < 35, 4.51 for ≥35.
“Younger women who have been treated with radiotherapy to the chest for Hodgkin lymphoma are already screened for breast cancer, but our study suggests that we should be looking at ways of monitoring survivors for other forms of cancer too, and potentially offering preventative interventions,” Dr Sud said.
“After patients are cured, they no longer encounter oncologists, so it’s important that other healthcare providers are aware of the increased risk to Hodgkin lymphoma survivors to improve early diagnosis of second cancers.” ![]()
A new study suggests Hodgkin lymphoma (HL) survivors have a high risk of developing a second malignancy, particularly if they have a family history of that malignancy.
The research showed that HL survivors in Sweden were roughly 2.4 times more likely than individuals in the country’s general population to develop a second cancer.
The risk for HL survivors remained high 30 years after treatment, and the risk was even greater in HL survivors who had a family history of specific cancers.
“The vast majority of patients with Hodgkin lymphoma are cured with a combination of chemotherapy and radiotherapy,” said study author Amit Sud, MBChB, of The Institute of Cancer Research, London in the UK.
“Our research has shown that these patients are at substantially increased risk of a second cancer later in life and particularly if they have a family history of cancer.”
Dr Sud and his colleagues described this research in the Journal of Clinical Oncology.
The team analyzed data from the Swedish Family-Cancer Project Database. They identified 9522 HL patients diagnosed between 1965 and 2013. During a median follow-up of 12.6 years, there were 1215 second cancers in 1121 HL patients (12%).
Compared to the general population, the HL patients had a significantly higher risk of all second malignancies, with a standardized incident ratio (SIR) of 2.39 and an absolute excess risk of 71.2 cases per 10,000 person-years.
Cancer types
HL patients had a significantly increased risk of several malignancies. The overall SIRs were as follows:
- NHL—7.99
- Leukemia—6.46
- Connective tissue cancer—5.73
- Thyroid cancer—5.13
- Squamous cell carcinoma—4.44
- Lung cancer—3.61
- Pharyngeal cancer—3.52
- Esophageal cancer—2.62
- Brain cancer—2.58
- Breast cancer—2.52
- Colon cancer—2.21
- Pancreatic cancer—2.09
- Melanoma—2.08
- Colorectal cancer—1.85
- Stomach cancer—1.78
- Bladder cancer—1.57
- Prostate cancer—1.21.
The researchers calculated SIRs over time and found the risk for many of the cancers remained high over 30 years following HL treatment.
Family history
The researchers identified 28,277 first-degree relatives of the HL survivors. Thirty percent of HL survivors (n=2785) had 1 or more first-degree relatives with a family history of cancer.
The SIR for cancers was 1.02 in the relatives. The SIR for second cancers was 2.83 for HL survivors who had first-degree relatives with cancer and 2.16 for HL survivors who did not have any first-degree relatives with cancer.
The researchers said the increased risk of second malignancy was correlated with the number of first-degree relatives with cancer.
The SIR was 2.67 for HL patients who had a single first-degree relative with cancer and 3.40 for HL patients who had 2 or more first-degree relatives with cancer.
The SIRs for different cancer types (for HL patients with at least 1 first-degree relative with cancer and no first-degree relatives with cancer, respectively) were as follows:
- NHL—14.43 vs 7.83
- Leukemia—14.31 vs 6.37
- Squamous cell carcinoma—10.85 vs 4.30
- Lung cancer—11.24 vs 3.39
- Breast cancer—4.36 vs 2.36
- Colorectal cancer—3.71 vs 1.76.
Sex and age
The researchers found significant differences in the SIRs for second cancers between HL patients diagnosed before the age of 35 and those diagnosed after age 35.
For men, the SIRs were:
- All cancers—4.26 for <35, 2.08 for ≥ 35
- Colorectal cancer—4.07 for < 35, 1.73 for ≥35
- Lung cancer—6.16 for < 35, 3.20 for ≥35
- Breast cancer—12.60 for < 35, 4.58 for ≥35
- Squamous cell carcinoma—5.89 for < 35, 3.96 for ≥35
- NHL—15.9 for < 35, 6.93 for ≥35
- Leukemia—12.15 for < 35, 5.57 for ≥35.
For women, the SIRs were:
- All cancers—4.61 for <35, 1.73 for ≥ 35
- Colorectal cancer—1.31 for < 35, 1.65 for ≥35
- Lung cancer—8.84 for < 35, 2.50 for ≥35
- Breast cancer—6.00 for < 35, 1.14 for ≥35
- Squamous cell carcinoma—6.37 for < 35, 4.87 for ≥35
- NHL—6.23 for < 35, 6.55 for ≥35
- Leukemia—10.36 for < 35, 4.51 for ≥35.
“Younger women who have been treated with radiotherapy to the chest for Hodgkin lymphoma are already screened for breast cancer, but our study suggests that we should be looking at ways of monitoring survivors for other forms of cancer too, and potentially offering preventative interventions,” Dr Sud said.
“After patients are cured, they no longer encounter oncologists, so it’s important that other healthcare providers are aware of the increased risk to Hodgkin lymphoma survivors to improve early diagnosis of second cancers.” ![]()
Selinexor trials placed on partial hold
The US Food and Drug Administration (FDA) has placed a partial clinical hold on all trials of selinexor (KPT-330).
Selinexor is an inhibitor being evaluated in multiple trials of patients with relapsed and/or refractory hematologic and solid tumor malignancies.
While the partial clinical hold remains in effect, patients with stable disease or better may remain on selinexor.
However, no new patients may be enrolled in selinexor trials until the hold is lifted.
The FDA has indicated that the partial clinical hold is due to incomplete information in the existing version of the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, said it has amended the brochure, updated the informed consent documents accordingly, and submitted the documents to the FDA as requested.
As of March 10, Karyopharm had provided all requested materials to the FDA believed to be required to lift the partial clinical hold. By regulation, the FDA has 30 days from the receipt of Karyopharm’s submission to notify the company whether the partial clinical hold is lifted.
Karyopharm said it is working with the FDA to seek the release of the hold and resume enrollment in its selinexor trials as expeditiously as possible. The company believes its previously disclosed enrollment rates and timelines for its ongoing trials will remain materially unchanged.
About selinexor
Selinexor is a selective inhibitor of nuclear export (SINE) XPO1 antagonist. The drug binds with and inhibits XPO1, leading to the accumulation of tumor suppressor proteins in the cell nucleus. This reinitiates and amplifies their tumor suppressor function and is believed to induce apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor. The drug is currently being evaluated in several trials across multiple cancer indications.
One of these is the phase 2 SOPRA trial, in which selinexor is being compared to investigator’s choice of therapy (1 of 3 potential salvage therapies). The trial is enrolling patients 60 years of age or older with relapsed or refractory acute myeloid leukemia who are ineligible for standard intensive chemotherapy and/or transplant.
The SADAL study is a phase 2b trial comparing high and low doses of selinexor in patients with relapsed and/or refractory de novo diffuse large B-cell lymphoma who have no therapeutic options of demonstrated clinical benefit.
STORM is a phase 2b trial evaluating selinexor and low-dose dexamethasone in patients with heavily pretreated multiple myeloma (MM). And STOMP is a phase 1b/2 study evaluating selinexor in combination with existing therapies across the broader population in MM.
Karyopharm is also planning a randomized, phase 3 study known as BOSTON. In this trial, researchers will compare selinexor plus bortezomib and low-dose dexamethasone to bortezomib and low-dose dexamethasone in MM patients who have had 1 to 3 prior lines of therapy.
Additional phase 1, 2, and 3 studies are ongoing or currently planned.
The US Food and Drug Administration (FDA) has placed a partial clinical hold on all trials of selinexor (KPT-330).
Selinexor is an inhibitor being evaluated in multiple trials of patients with relapsed and/or refractory hematologic and solid tumor malignancies.
While the partial clinical hold remains in effect, patients with stable disease or better may remain on selinexor.
However, no new patients may be enrolled in selinexor trials until the hold is lifted.
The FDA has indicated that the partial clinical hold is due to incomplete information in the existing version of the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, said it has amended the brochure, updated the informed consent documents accordingly, and submitted the documents to the FDA as requested.
As of March 10, Karyopharm had provided all requested materials to the FDA believed to be required to lift the partial clinical hold. By regulation, the FDA has 30 days from the receipt of Karyopharm’s submission to notify the company whether the partial clinical hold is lifted.
Karyopharm said it is working with the FDA to seek the release of the hold and resume enrollment in its selinexor trials as expeditiously as possible. The company believes its previously disclosed enrollment rates and timelines for its ongoing trials will remain materially unchanged.
About selinexor
Selinexor is a selective inhibitor of nuclear export (SINE) XPO1 antagonist. The drug binds with and inhibits XPO1, leading to the accumulation of tumor suppressor proteins in the cell nucleus. This reinitiates and amplifies their tumor suppressor function and is believed to induce apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor. The drug is currently being evaluated in several trials across multiple cancer indications.
One of these is the phase 2 SOPRA trial, in which selinexor is being compared to investigator’s choice of therapy (1 of 3 potential salvage therapies). The trial is enrolling patients 60 years of age or older with relapsed or refractory acute myeloid leukemia who are ineligible for standard intensive chemotherapy and/or transplant.
The SADAL study is a phase 2b trial comparing high and low doses of selinexor in patients with relapsed and/or refractory de novo diffuse large B-cell lymphoma who have no therapeutic options of demonstrated clinical benefit.
STORM is a phase 2b trial evaluating selinexor and low-dose dexamethasone in patients with heavily pretreated multiple myeloma (MM). And STOMP is a phase 1b/2 study evaluating selinexor in combination with existing therapies across the broader population in MM.
Karyopharm is also planning a randomized, phase 3 study known as BOSTON. In this trial, researchers will compare selinexor plus bortezomib and low-dose dexamethasone to bortezomib and low-dose dexamethasone in MM patients who have had 1 to 3 prior lines of therapy.
Additional phase 1, 2, and 3 studies are ongoing or currently planned.
The US Food and Drug Administration (FDA) has placed a partial clinical hold on all trials of selinexor (KPT-330).
Selinexor is an inhibitor being evaluated in multiple trials of patients with relapsed and/or refractory hematologic and solid tumor malignancies.
While the partial clinical hold remains in effect, patients with stable disease or better may remain on selinexor.
However, no new patients may be enrolled in selinexor trials until the hold is lifted.
The FDA has indicated that the partial clinical hold is due to incomplete information in the existing version of the investigator’s brochure, including an incomplete list of serious adverse events associated with selinexor.
Karyopharm Therapeutics Inc., the company developing selinexor, said it has amended the brochure, updated the informed consent documents accordingly, and submitted the documents to the FDA as requested.
As of March 10, Karyopharm had provided all requested materials to the FDA believed to be required to lift the partial clinical hold. By regulation, the FDA has 30 days from the receipt of Karyopharm’s submission to notify the company whether the partial clinical hold is lifted.
Karyopharm said it is working with the FDA to seek the release of the hold and resume enrollment in its selinexor trials as expeditiously as possible. The company believes its previously disclosed enrollment rates and timelines for its ongoing trials will remain materially unchanged.
About selinexor
Selinexor is a selective inhibitor of nuclear export (SINE) XPO1 antagonist. The drug binds with and inhibits XPO1, leading to the accumulation of tumor suppressor proteins in the cell nucleus. This reinitiates and amplifies their tumor suppressor function and is believed to induce apoptosis in cancer cells while largely sparing normal cells.
To date, more than 1900 patients have been treated with selinexor. The drug is currently being evaluated in several trials across multiple cancer indications.
One of these is the phase 2 SOPRA trial, in which selinexor is being compared to investigator’s choice of therapy (1 of 3 potential salvage therapies). The trial is enrolling patients 60 years of age or older with relapsed or refractory acute myeloid leukemia who are ineligible for standard intensive chemotherapy and/or transplant.
The SADAL study is a phase 2b trial comparing high and low doses of selinexor in patients with relapsed and/or refractory de novo diffuse large B-cell lymphoma who have no therapeutic options of demonstrated clinical benefit.
STORM is a phase 2b trial evaluating selinexor and low-dose dexamethasone in patients with heavily pretreated multiple myeloma (MM). And STOMP is a phase 1b/2 study evaluating selinexor in combination with existing therapies across the broader population in MM.
Karyopharm is also planning a randomized, phase 3 study known as BOSTON. In this trial, researchers will compare selinexor plus bortezomib and low-dose dexamethasone to bortezomib and low-dose dexamethasone in MM patients who have had 1 to 3 prior lines of therapy.
Additional phase 1, 2, and 3 studies are ongoing or currently planned.
Drug receives orphan designation for DLBCL
The US Food and Drug Administration (FDA) has granted orphan drug designation for eFT508 to treat diffuse large B-cell lymphoma (DLBCL).
eFT508 is a highly selective inhibitor of MNK1 and MNK2, enzymes that integrate signals from several oncogenic and immune signaling pathways.
The FDA grants orphan designation to drugs or biologics intended to treat a disease or condition affecting fewer than 200,000 patients in the US.
The orphan designation for eFT508 provides several incentives for eFFECTOR Therapeutics, the company developing eFT508.
These incentives include increased access to FDA reviewers to discuss clinical trial designs, the ability to qualify for tax credits for certain clinical research costs, the ability to apply for annual grant funding, a waiver of Prescription Drug User Fee Act filing fees, and the potential for 7 years of US marketing exclusivity if eFT508 is approved.
eFFECTOR has dosed the first subject in a phase 1/2 trial of eFT508 in patients with B-cell hematologic malignancies. The study is designed to evaluate the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of eFT508.
eFFECTOR presented preclinical research of eFT508 in DLBCL at the 2015 ASH Annual Meeting. The poster is available for download from the eFFECTOR website.
The researchers reported that eFT508 demonstrated anti-proliferative activity against multiple DLBCL cell lines, including the TMD8, OCI-Ly3, and HBL1 cell lines.
eFT508 also exhibited “significant anti-tumor activity” in mouse models of TMD8 and HBL-1 ABC-DLBCL.
Finally, the researchers found that eFT508 synergized with everolimus, ibrutinib, and venetoclax both in vitro and in vivo. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation for eFT508 to treat diffuse large B-cell lymphoma (DLBCL).
eFT508 is a highly selective inhibitor of MNK1 and MNK2, enzymes that integrate signals from several oncogenic and immune signaling pathways.
The FDA grants orphan designation to drugs or biologics intended to treat a disease or condition affecting fewer than 200,000 patients in the US.
The orphan designation for eFT508 provides several incentives for eFFECTOR Therapeutics, the company developing eFT508.
These incentives include increased access to FDA reviewers to discuss clinical trial designs, the ability to qualify for tax credits for certain clinical research costs, the ability to apply for annual grant funding, a waiver of Prescription Drug User Fee Act filing fees, and the potential for 7 years of US marketing exclusivity if eFT508 is approved.
eFFECTOR has dosed the first subject in a phase 1/2 trial of eFT508 in patients with B-cell hematologic malignancies. The study is designed to evaluate the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of eFT508.
eFFECTOR presented preclinical research of eFT508 in DLBCL at the 2015 ASH Annual Meeting. The poster is available for download from the eFFECTOR website.
The researchers reported that eFT508 demonstrated anti-proliferative activity against multiple DLBCL cell lines, including the TMD8, OCI-Ly3, and HBL1 cell lines.
eFT508 also exhibited “significant anti-tumor activity” in mouse models of TMD8 and HBL-1 ABC-DLBCL.
Finally, the researchers found that eFT508 synergized with everolimus, ibrutinib, and venetoclax both in vitro and in vivo. ![]()
The US Food and Drug Administration (FDA) has granted orphan drug designation for eFT508 to treat diffuse large B-cell lymphoma (DLBCL).
eFT508 is a highly selective inhibitor of MNK1 and MNK2, enzymes that integrate signals from several oncogenic and immune signaling pathways.
The FDA grants orphan designation to drugs or biologics intended to treat a disease or condition affecting fewer than 200,000 patients in the US.
The orphan designation for eFT508 provides several incentives for eFFECTOR Therapeutics, the company developing eFT508.
These incentives include increased access to FDA reviewers to discuss clinical trial designs, the ability to qualify for tax credits for certain clinical research costs, the ability to apply for annual grant funding, a waiver of Prescription Drug User Fee Act filing fees, and the potential for 7 years of US marketing exclusivity if eFT508 is approved.
eFFECTOR has dosed the first subject in a phase 1/2 trial of eFT508 in patients with B-cell hematologic malignancies. The study is designed to evaluate the safety, pharmacokinetics, pharmacodynamics, and antitumor activity of eFT508.
eFFECTOR presented preclinical research of eFT508 in DLBCL at the 2015 ASH Annual Meeting. The poster is available for download from the eFFECTOR website.
The researchers reported that eFT508 demonstrated anti-proliferative activity against multiple DLBCL cell lines, including the TMD8, OCI-Ly3, and HBL1 cell lines.
eFT508 also exhibited “significant anti-tumor activity” in mouse models of TMD8 and HBL-1 ABC-DLBCL.
Finally, the researchers found that eFT508 synergized with everolimus, ibrutinib, and venetoclax both in vitro and in vivo. ![]()
Study confirms increased adiposity in HSCT survivors
ORLANDO, FL—A recently conducted study confirms that survivors of hematopoietic stem cell transplant (HSCT) have increased body fat mass and lower lean mass compared to normal controls. And this is despite having a comparable body mass index (BMI).
Researchers say the abnormalities in adipokine levels—leptin and adiponectin—could provide insight into the mechanisms that contribute to the metabolic syndrome and cardiovascular complications that often develop in HSCT survivors.
Leptin and adiponectin are associated with obesity, insulin secretion, insulin resistance, endothelial function, vascular homeostasis, and atherosclerosis.
“So knowing that there is a dynamic interplay between obesity and insulin resistance and cytokine and adipokine profiles and, ultimately, insulin-resistance syndrome, we sought to evaluate, as part of a larger study, how treatment effects, including high-dose chemotherapy and radiation, alter cytokine profiles as well as obesity and body composition,” said Tyler G. Ketterl, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
Dr Ketterl presented the findings at the 2017 BMT Tandem Meetings as abstract 52.*
Study design
The research team compared 151 HSCT recipients who had survived more than 2 years after transplant with 92 sibling controls.
HSCT survivors were randomly recruited from 2 centers—Fred Hutchinson Cancer Research Center and University of Minnesota Masonic Children’s Hospital—and were younger than 21 years when diagnosed.
The researchers evaluated all participants for body composition, cardiovascular risk factors, and adipokines using anthropomorphic measurements, DXA scans for muscle and fat mass, and laboratory bloodwork.
The team stratified the HSCT survivors by the preparative regimen they had received—total body irradiation (TBI) alone, TBI plus cranial radiation (CRT), and chemotherapy alone.
Study population
Males comprised more than half the study population in each arm, 58% of HSCT survivors and 54% of siblings.
Nine percent and 8% in the HSCT and sibling arms, respectively, were non-white and/or Hispanic, and the mean current ages were 24.0 (range, 10-51) for HSCT survivors and 24.2 (range, 10-48) for siblings.
The survivors’ mean age at diagnosis was 9.1 years (range, 0.4–20.6), their mean age at transplant was 11.2 years (range, 0.6–32.6), and the mean time from transplant to study participation was 13.5 years (range, 2.6–32).
Most patients received a transplant for leukemia—54 (36%) for acute myeloid leukemia, 46 (31%) for acute lymphoblastic leukemia, and 15 (10%) for chronic myeloid leukemia. Thirteen (9%) received transplants for myelodysplastic syndromes, 12 (8%) for Hodgkin lymphoma, and 10 (6%) for non-Hodgkin lymphoma.
A little more than half had TBI (85, 56%) as the preparative regimen, 31 (21%) had TBI plus CRT, and 35 (23%) had chemotherapy only.
About three-quarters (116, 77%) had an allogeneic transplant, and 35 (23%) had an autologous transplant.
Results
Overall, HSCT survivors had significantly lower adiponectin levels than siblings (P<0.001).
Survivors who received TBI with or without CRT had significantly lower adiponectin levels than siblings (P<0.001), while survivors who received chemotherapy alone did not (P=0.42).
Adiponectin is involved in insulin sensitization, hepatoprotective action, antiatherogenic action, protection against the development of diabetes, and regulation of lipid metabolism.
Overall, survivors had significantly higher leptin levels than siblings (P<0.001).
This held true regardless of conditioning regimen, although levels for patients who received chemotherapy only were not as significantly high (P=0.02) as for survivors who received TBI (P<0.001).
Leptin helps increase energy expenditure, decrease appetite and food uptake, modify insulin sensitivity on muscles and liver, prevent ectopic lipid deposition, and regulate immune function.
BMI adjusted for age, sex, and Tanner stage was not significantly different between survivors and siblings, but percent fat mass was significantly higher across all conditioning regimens for survivors compared to siblings (P<0.001).
“And this goes along with previous data,” Dr Ketterl said, “that shows sarcopenic obesity is common amongst transplant survivors.”
The researchers believe these significant differences may provide insight into the underlying risk of developing metabolic syndrome and cardiovascular complications in transplant survivors. ![]()
*Some details in the abstract differ from the presentation.
ORLANDO, FL—A recently conducted study confirms that survivors of hematopoietic stem cell transplant (HSCT) have increased body fat mass and lower lean mass compared to normal controls. And this is despite having a comparable body mass index (BMI).
Researchers say the abnormalities in adipokine levels—leptin and adiponectin—could provide insight into the mechanisms that contribute to the metabolic syndrome and cardiovascular complications that often develop in HSCT survivors.
Leptin and adiponectin are associated with obesity, insulin secretion, insulin resistance, endothelial function, vascular homeostasis, and atherosclerosis.
“So knowing that there is a dynamic interplay between obesity and insulin resistance and cytokine and adipokine profiles and, ultimately, insulin-resistance syndrome, we sought to evaluate, as part of a larger study, how treatment effects, including high-dose chemotherapy and radiation, alter cytokine profiles as well as obesity and body composition,” said Tyler G. Ketterl, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
Dr Ketterl presented the findings at the 2017 BMT Tandem Meetings as abstract 52.*
Study design
The research team compared 151 HSCT recipients who had survived more than 2 years after transplant with 92 sibling controls.
HSCT survivors were randomly recruited from 2 centers—Fred Hutchinson Cancer Research Center and University of Minnesota Masonic Children’s Hospital—and were younger than 21 years when diagnosed.
The researchers evaluated all participants for body composition, cardiovascular risk factors, and adipokines using anthropomorphic measurements, DXA scans for muscle and fat mass, and laboratory bloodwork.
The team stratified the HSCT survivors by the preparative regimen they had received—total body irradiation (TBI) alone, TBI plus cranial radiation (CRT), and chemotherapy alone.
Study population
Males comprised more than half the study population in each arm, 58% of HSCT survivors and 54% of siblings.
Nine percent and 8% in the HSCT and sibling arms, respectively, were non-white and/or Hispanic, and the mean current ages were 24.0 (range, 10-51) for HSCT survivors and 24.2 (range, 10-48) for siblings.
The survivors’ mean age at diagnosis was 9.1 years (range, 0.4–20.6), their mean age at transplant was 11.2 years (range, 0.6–32.6), and the mean time from transplant to study participation was 13.5 years (range, 2.6–32).
Most patients received a transplant for leukemia—54 (36%) for acute myeloid leukemia, 46 (31%) for acute lymphoblastic leukemia, and 15 (10%) for chronic myeloid leukemia. Thirteen (9%) received transplants for myelodysplastic syndromes, 12 (8%) for Hodgkin lymphoma, and 10 (6%) for non-Hodgkin lymphoma.
A little more than half had TBI (85, 56%) as the preparative regimen, 31 (21%) had TBI plus CRT, and 35 (23%) had chemotherapy only.
About three-quarters (116, 77%) had an allogeneic transplant, and 35 (23%) had an autologous transplant.
Results
Overall, HSCT survivors had significantly lower adiponectin levels than siblings (P<0.001).
Survivors who received TBI with or without CRT had significantly lower adiponectin levels than siblings (P<0.001), while survivors who received chemotherapy alone did not (P=0.42).
Adiponectin is involved in insulin sensitization, hepatoprotective action, antiatherogenic action, protection against the development of diabetes, and regulation of lipid metabolism.
Overall, survivors had significantly higher leptin levels than siblings (P<0.001).
This held true regardless of conditioning regimen, although levels for patients who received chemotherapy only were not as significantly high (P=0.02) as for survivors who received TBI (P<0.001).
Leptin helps increase energy expenditure, decrease appetite and food uptake, modify insulin sensitivity on muscles and liver, prevent ectopic lipid deposition, and regulate immune function.
BMI adjusted for age, sex, and Tanner stage was not significantly different between survivors and siblings, but percent fat mass was significantly higher across all conditioning regimens for survivors compared to siblings (P<0.001).
“And this goes along with previous data,” Dr Ketterl said, “that shows sarcopenic obesity is common amongst transplant survivors.”
The researchers believe these significant differences may provide insight into the underlying risk of developing metabolic syndrome and cardiovascular complications in transplant survivors. ![]()
*Some details in the abstract differ from the presentation.
ORLANDO, FL—A recently conducted study confirms that survivors of hematopoietic stem cell transplant (HSCT) have increased body fat mass and lower lean mass compared to normal controls. And this is despite having a comparable body mass index (BMI).
Researchers say the abnormalities in adipokine levels—leptin and adiponectin—could provide insight into the mechanisms that contribute to the metabolic syndrome and cardiovascular complications that often develop in HSCT survivors.
Leptin and adiponectin are associated with obesity, insulin secretion, insulin resistance, endothelial function, vascular homeostasis, and atherosclerosis.
“So knowing that there is a dynamic interplay between obesity and insulin resistance and cytokine and adipokine profiles and, ultimately, insulin-resistance syndrome, we sought to evaluate, as part of a larger study, how treatment effects, including high-dose chemotherapy and radiation, alter cytokine profiles as well as obesity and body composition,” said Tyler G. Ketterl, MD, of the Fred Hutchinson Cancer Research Center in Seattle, Washington.
Dr Ketterl presented the findings at the 2017 BMT Tandem Meetings as abstract 52.*
Study design
The research team compared 151 HSCT recipients who had survived more than 2 years after transplant with 92 sibling controls.
HSCT survivors were randomly recruited from 2 centers—Fred Hutchinson Cancer Research Center and University of Minnesota Masonic Children’s Hospital—and were younger than 21 years when diagnosed.
The researchers evaluated all participants for body composition, cardiovascular risk factors, and adipokines using anthropomorphic measurements, DXA scans for muscle and fat mass, and laboratory bloodwork.
The team stratified the HSCT survivors by the preparative regimen they had received—total body irradiation (TBI) alone, TBI plus cranial radiation (CRT), and chemotherapy alone.
Study population
Males comprised more than half the study population in each arm, 58% of HSCT survivors and 54% of siblings.
Nine percent and 8% in the HSCT and sibling arms, respectively, were non-white and/or Hispanic, and the mean current ages were 24.0 (range, 10-51) for HSCT survivors and 24.2 (range, 10-48) for siblings.
The survivors’ mean age at diagnosis was 9.1 years (range, 0.4–20.6), their mean age at transplant was 11.2 years (range, 0.6–32.6), and the mean time from transplant to study participation was 13.5 years (range, 2.6–32).
Most patients received a transplant for leukemia—54 (36%) for acute myeloid leukemia, 46 (31%) for acute lymphoblastic leukemia, and 15 (10%) for chronic myeloid leukemia. Thirteen (9%) received transplants for myelodysplastic syndromes, 12 (8%) for Hodgkin lymphoma, and 10 (6%) for non-Hodgkin lymphoma.
A little more than half had TBI (85, 56%) as the preparative regimen, 31 (21%) had TBI plus CRT, and 35 (23%) had chemotherapy only.
About three-quarters (116, 77%) had an allogeneic transplant, and 35 (23%) had an autologous transplant.
Results
Overall, HSCT survivors had significantly lower adiponectin levels than siblings (P<0.001).
Survivors who received TBI with or without CRT had significantly lower adiponectin levels than siblings (P<0.001), while survivors who received chemotherapy alone did not (P=0.42).
Adiponectin is involved in insulin sensitization, hepatoprotective action, antiatherogenic action, protection against the development of diabetes, and regulation of lipid metabolism.
Overall, survivors had significantly higher leptin levels than siblings (P<0.001).
This held true regardless of conditioning regimen, although levels for patients who received chemotherapy only were not as significantly high (P=0.02) as for survivors who received TBI (P<0.001).
Leptin helps increase energy expenditure, decrease appetite and food uptake, modify insulin sensitivity on muscles and liver, prevent ectopic lipid deposition, and regulate immune function.
BMI adjusted for age, sex, and Tanner stage was not significantly different between survivors and siblings, but percent fat mass was significantly higher across all conditioning regimens for survivors compared to siblings (P<0.001).
“And this goes along with previous data,” Dr Ketterl said, “that shows sarcopenic obesity is common amongst transplant survivors.”
The researchers believe these significant differences may provide insight into the underlying risk of developing metabolic syndrome and cardiovascular complications in transplant survivors. ![]()
*Some details in the abstract differ from the presentation.
Cell-free DNA mutational analysis in AITL
SAN FRANCISCO—An exploratory study suggests cell-free DNA from peripheral blood may be a viable alternative to tumor DNA for mutational profiling in angioimmunoblastic T-cell lymphoma (AITL).
Investigators sequenced cell-free DNA and tumor DNA collected from 13 patients with AITL and found that, in 85% of cases, there was concordance between the tumor mutational profile and the cell-free DNA mutational profile.
“The cell-free DNA mutational analysis seems to mirror the mutational analysis of the tumor in the majority of cases,” said Neha Mehta-Shah, MD, of Memorial Sloan Kettering Cancer Center in New York, New York.
“This may be particularly interesting when we don’t have very much tumor DNA available for sequencing, as occurs frequently in this patient population.”
Dr Mehta-Shah presented these findings at the 9th Annual T-cell Lymphoma Forum.
She said the primary objective of this study was to evaluate whether somatic mutation analysis of cell-free DNA for IDH2 correlates with tumor mutational profiling for IDH2.
The secondary objective was to evaluate whether somatic mutational analysis of cell-free DNA mirrors the mutational profile of the tumor.
So Dr Mehta-Shah and her colleagues sequenced various samples from 14 patients with AITL (4 who were newly diagnosed and 10 with relapsed disease).
The investigators compared cell-free DNA obtained from peripheral blood samples with genomic DNA obtained from primary tumor biopsies and with DNA from peripheral blood mononuclear cells (for germline comparison). One of the 14 patients didn’t have tumor tissue available, so 13 patients were included in the analysis.
The team performed targeted next-generation sequencing using Memorial Sloan Kettering Cancer Center’s IMPACT platform, which sequences 410 genes known to be recurrently mutated in cancer.
The investigators observed concordance between the tumor mutational profile and the cell-free DNA profile in 85% of cases (11/13).
Identical alterations in TET2, RHOA, IDH2, DNMT3A, and ROS1 were detected in cell-free DNA from peripheral blood and tumor genomic DNA, with a similar variant allele frequency.
On the other hand, mutational analysis of cell-free DNA obtained from urine samples from 2 of the patients did not correlate with somatic mutations from tumor DNA.
This research also revealed that some samples had multiple mutations in TET2, which are indicative of subclonal populations.
And sequential samples from 2 patients, collected at the start of salvage therapy and at subsequent relapse, showed the disappearance of mutations in RHOA, TET2, and IDH2. This observation has been attributed to clonal evolution and/or changes in overall disease burden. ![]()
SAN FRANCISCO—An exploratory study suggests cell-free DNA from peripheral blood may be a viable alternative to tumor DNA for mutational profiling in angioimmunoblastic T-cell lymphoma (AITL).
Investigators sequenced cell-free DNA and tumor DNA collected from 13 patients with AITL and found that, in 85% of cases, there was concordance between the tumor mutational profile and the cell-free DNA mutational profile.
“The cell-free DNA mutational analysis seems to mirror the mutational analysis of the tumor in the majority of cases,” said Neha Mehta-Shah, MD, of Memorial Sloan Kettering Cancer Center in New York, New York.
“This may be particularly interesting when we don’t have very much tumor DNA available for sequencing, as occurs frequently in this patient population.”
Dr Mehta-Shah presented these findings at the 9th Annual T-cell Lymphoma Forum.
She said the primary objective of this study was to evaluate whether somatic mutation analysis of cell-free DNA for IDH2 correlates with tumor mutational profiling for IDH2.
The secondary objective was to evaluate whether somatic mutational analysis of cell-free DNA mirrors the mutational profile of the tumor.
So Dr Mehta-Shah and her colleagues sequenced various samples from 14 patients with AITL (4 who were newly diagnosed and 10 with relapsed disease).
The investigators compared cell-free DNA obtained from peripheral blood samples with genomic DNA obtained from primary tumor biopsies and with DNA from peripheral blood mononuclear cells (for germline comparison). One of the 14 patients didn’t have tumor tissue available, so 13 patients were included in the analysis.
The team performed targeted next-generation sequencing using Memorial Sloan Kettering Cancer Center’s IMPACT platform, which sequences 410 genes known to be recurrently mutated in cancer.
The investigators observed concordance between the tumor mutational profile and the cell-free DNA profile in 85% of cases (11/13).
Identical alterations in TET2, RHOA, IDH2, DNMT3A, and ROS1 were detected in cell-free DNA from peripheral blood and tumor genomic DNA, with a similar variant allele frequency.
On the other hand, mutational analysis of cell-free DNA obtained from urine samples from 2 of the patients did not correlate with somatic mutations from tumor DNA.
This research also revealed that some samples had multiple mutations in TET2, which are indicative of subclonal populations.
And sequential samples from 2 patients, collected at the start of salvage therapy and at subsequent relapse, showed the disappearance of mutations in RHOA, TET2, and IDH2. This observation has been attributed to clonal evolution and/or changes in overall disease burden. ![]()
SAN FRANCISCO—An exploratory study suggests cell-free DNA from peripheral blood may be a viable alternative to tumor DNA for mutational profiling in angioimmunoblastic T-cell lymphoma (AITL).
Investigators sequenced cell-free DNA and tumor DNA collected from 13 patients with AITL and found that, in 85% of cases, there was concordance between the tumor mutational profile and the cell-free DNA mutational profile.
“The cell-free DNA mutational analysis seems to mirror the mutational analysis of the tumor in the majority of cases,” said Neha Mehta-Shah, MD, of Memorial Sloan Kettering Cancer Center in New York, New York.
“This may be particularly interesting when we don’t have very much tumor DNA available for sequencing, as occurs frequently in this patient population.”
Dr Mehta-Shah presented these findings at the 9th Annual T-cell Lymphoma Forum.
She said the primary objective of this study was to evaluate whether somatic mutation analysis of cell-free DNA for IDH2 correlates with tumor mutational profiling for IDH2.
The secondary objective was to evaluate whether somatic mutational analysis of cell-free DNA mirrors the mutational profile of the tumor.
So Dr Mehta-Shah and her colleagues sequenced various samples from 14 patients with AITL (4 who were newly diagnosed and 10 with relapsed disease).
The investigators compared cell-free DNA obtained from peripheral blood samples with genomic DNA obtained from primary tumor biopsies and with DNA from peripheral blood mononuclear cells (for germline comparison). One of the 14 patients didn’t have tumor tissue available, so 13 patients were included in the analysis.
The team performed targeted next-generation sequencing using Memorial Sloan Kettering Cancer Center’s IMPACT platform, which sequences 410 genes known to be recurrently mutated in cancer.
The investigators observed concordance between the tumor mutational profile and the cell-free DNA profile in 85% of cases (11/13).
Identical alterations in TET2, RHOA, IDH2, DNMT3A, and ROS1 were detected in cell-free DNA from peripheral blood and tumor genomic DNA, with a similar variant allele frequency.
On the other hand, mutational analysis of cell-free DNA obtained from urine samples from 2 of the patients did not correlate with somatic mutations from tumor DNA.
This research also revealed that some samples had multiple mutations in TET2, which are indicative of subclonal populations.
And sequential samples from 2 patients, collected at the start of salvage therapy and at subsequent relapse, showed the disappearance of mutations in RHOA, TET2, and IDH2. This observation has been attributed to clonal evolution and/or changes in overall disease burden. ![]()
Chlormethine gel approved to treat mycosis fungoides
The European Commission has granted marketing authorization for chlormethine gel (Ledaga®) as a treatment for adults with mycosis fungoides (MF), but the product is not expected to be available until next year.
Ledaga is a hybrid medicine of Caryolysine, which has been approved for use in the European Union since 1946.
Hybrid medicines have a different strength, are administered differently, or have a different indication from the reference medicine.
Ledaga contains the same active substance as Caryolysine—chlormethine, a bifunctional alkylating agent that inhibits rapidly proliferating cells—but Ledaga is a gel intended for cutaneous use.
Actelion Pharmaceuticals Ltd, the company developing Ledaga, has agreed to fulfill a list of post-approval measures for the product proposed by the European Medicines Agency’s Committee for Medicinal Products for Human Use.
Subject to fulfilling the agreed commitments and achieving market access in various countries, a potential first European launch of Ledaga is not expected before January 2018. When it is launched, Ledaga will be available as a 160 μg/g gel.
Phase 2 study
The authorization of hybrid medicines depends partly on the results of tests on the reference medicine and partly on new data from clinical trials.
The marketing authorization for Ledaga is based, in part, on results of a multicenter, randomized, observer-blinded, active-controlled study of patients with stage I and IIA MF. Results from this phase 2 study were published in JAMA Dermatology.
The study enrolled 260 MF patients who were randomized 1:1 to receive topical treatment with 0.02% chlormethine gel (Ledaga) or compounded control—0.02% chlormethine compounded in Aquaphor® ointment—once daily for up to 12 months.
A response was defined as at least a 50% improvement in the baseline Composite Assessment of Index Lesion Severity score.
In the intent-to-treat population, 59% (76/130) of patients who received Ledaga achieved a clinical response, compared to 48% (62/130) of patients treated with the compounded control. The rate of complete response was 14% (n=18) and 12% (n=15), respectively.
Patients who were treated for at least 6 months were included in the efficacy-evaluable population. In this population, 77% (69/90) of patients who received Ledaga achieved a clinical response, compared to 59% (56/95) of patients treated with the compounded control. The rate of complete response was 19% (n=17) and 15% (n=14), respectively.
Reductions in mean lesion severity were seen as early as 4 weeks into the study, with further reductions observed with continuing therapy. The time to first confirmed response favored Ledaga.
The most frequent adverse reactions reported with Ledaga were skin-related—dermatitis (55%; eg, skin irritation, erythema, rash, urticaria, skin-burning sensation, skin pain), pruritus (20%), skin infections (12%), skin ulceration and blistering (6%), and skin hyperpigmentation (6%).
No systemic absorption of chlormethine was detected with treatment. ![]()
The European Commission has granted marketing authorization for chlormethine gel (Ledaga®) as a treatment for adults with mycosis fungoides (MF), but the product is not expected to be available until next year.
Ledaga is a hybrid medicine of Caryolysine, which has been approved for use in the European Union since 1946.
Hybrid medicines have a different strength, are administered differently, or have a different indication from the reference medicine.
Ledaga contains the same active substance as Caryolysine—chlormethine, a bifunctional alkylating agent that inhibits rapidly proliferating cells—but Ledaga is a gel intended for cutaneous use.
Actelion Pharmaceuticals Ltd, the company developing Ledaga, has agreed to fulfill a list of post-approval measures for the product proposed by the European Medicines Agency’s Committee for Medicinal Products for Human Use.
Subject to fulfilling the agreed commitments and achieving market access in various countries, a potential first European launch of Ledaga is not expected before January 2018. When it is launched, Ledaga will be available as a 160 μg/g gel.
Phase 2 study
The authorization of hybrid medicines depends partly on the results of tests on the reference medicine and partly on new data from clinical trials.
The marketing authorization for Ledaga is based, in part, on results of a multicenter, randomized, observer-blinded, active-controlled study of patients with stage I and IIA MF. Results from this phase 2 study were published in JAMA Dermatology.
The study enrolled 260 MF patients who were randomized 1:1 to receive topical treatment with 0.02% chlormethine gel (Ledaga) or compounded control—0.02% chlormethine compounded in Aquaphor® ointment—once daily for up to 12 months.
A response was defined as at least a 50% improvement in the baseline Composite Assessment of Index Lesion Severity score.
In the intent-to-treat population, 59% (76/130) of patients who received Ledaga achieved a clinical response, compared to 48% (62/130) of patients treated with the compounded control. The rate of complete response was 14% (n=18) and 12% (n=15), respectively.
Patients who were treated for at least 6 months were included in the efficacy-evaluable population. In this population, 77% (69/90) of patients who received Ledaga achieved a clinical response, compared to 59% (56/95) of patients treated with the compounded control. The rate of complete response was 19% (n=17) and 15% (n=14), respectively.
Reductions in mean lesion severity were seen as early as 4 weeks into the study, with further reductions observed with continuing therapy. The time to first confirmed response favored Ledaga.
The most frequent adverse reactions reported with Ledaga were skin-related—dermatitis (55%; eg, skin irritation, erythema, rash, urticaria, skin-burning sensation, skin pain), pruritus (20%), skin infections (12%), skin ulceration and blistering (6%), and skin hyperpigmentation (6%).
No systemic absorption of chlormethine was detected with treatment. ![]()
The European Commission has granted marketing authorization for chlormethine gel (Ledaga®) as a treatment for adults with mycosis fungoides (MF), but the product is not expected to be available until next year.
Ledaga is a hybrid medicine of Caryolysine, which has been approved for use in the European Union since 1946.
Hybrid medicines have a different strength, are administered differently, or have a different indication from the reference medicine.
Ledaga contains the same active substance as Caryolysine—chlormethine, a bifunctional alkylating agent that inhibits rapidly proliferating cells—but Ledaga is a gel intended for cutaneous use.
Actelion Pharmaceuticals Ltd, the company developing Ledaga, has agreed to fulfill a list of post-approval measures for the product proposed by the European Medicines Agency’s Committee for Medicinal Products for Human Use.
Subject to fulfilling the agreed commitments and achieving market access in various countries, a potential first European launch of Ledaga is not expected before January 2018. When it is launched, Ledaga will be available as a 160 μg/g gel.
Phase 2 study
The authorization of hybrid medicines depends partly on the results of tests on the reference medicine and partly on new data from clinical trials.
The marketing authorization for Ledaga is based, in part, on results of a multicenter, randomized, observer-blinded, active-controlled study of patients with stage I and IIA MF. Results from this phase 2 study were published in JAMA Dermatology.
The study enrolled 260 MF patients who were randomized 1:1 to receive topical treatment with 0.02% chlormethine gel (Ledaga) or compounded control—0.02% chlormethine compounded in Aquaphor® ointment—once daily for up to 12 months.
A response was defined as at least a 50% improvement in the baseline Composite Assessment of Index Lesion Severity score.
In the intent-to-treat population, 59% (76/130) of patients who received Ledaga achieved a clinical response, compared to 48% (62/130) of patients treated with the compounded control. The rate of complete response was 14% (n=18) and 12% (n=15), respectively.
Patients who were treated for at least 6 months were included in the efficacy-evaluable population. In this population, 77% (69/90) of patients who received Ledaga achieved a clinical response, compared to 59% (56/95) of patients treated with the compounded control. The rate of complete response was 19% (n=17) and 15% (n=14), respectively.
Reductions in mean lesion severity were seen as early as 4 weeks into the study, with further reductions observed with continuing therapy. The time to first confirmed response favored Ledaga.
The most frequent adverse reactions reported with Ledaga were skin-related—dermatitis (55%; eg, skin irritation, erythema, rash, urticaria, skin-burning sensation, skin pain), pruritus (20%), skin infections (12%), skin ulceration and blistering (6%), and skin hyperpigmentation (6%).
No systemic absorption of chlormethine was detected with treatment. ![]()
Pathogenesis of breast-implant-associated ALCL
SAN FRANCISCO—A small study suggests an abnormal immune response characterized by the production of interleukin-13 (IL-13) underlies the pathogenesis of breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL).
The immune response appears to be directed toward bacterial antigens on the surface of the breast implants.
Marshall E. Kadin, MD, of Roger Williams Medical Center in Providence, Rhode Island, presented these findings at the 9th Annual T-cell Lymphoma Forum.
Dr Kadin noted that BIA-ALCL is a rare type of CD30+ T-cell ALCL that has been reported in more than 200 women worldwide.
Although a cause-and-effect relationship between breast implants and BIA-ALCL has been suggested, the underlying pathogenesis of this malignancy is unclear.
A bacterial biofilm containing gram-negative bacilli has been detected in breast implants from patients with BIA-ALCL.
Therefore, Dr Kadin and his colleagues hypothesized that an immune response toward the bacterial antigens may mediate the pathogenesis of BIA-ALCL.
The researchers studied 13 clinical samples of breast implant capsules and regional lymph nodes from 4 patients with BIA-ALCL, 7 patients with systemic ALCL, and 1 patient with peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS).
Immunohistochemistry was used to determine the presence of IL-13, IL-4, GATA3, and immunoglobulin E (IgE) in these samples.
All clinical samples of anaplastic cells from breast implant capsules tested positive for the presence of IL-13 (13/13). GATA3 was expressed in most anaplastic cell samples (12/13), and IL-4 expression was found in some anaplastic cell samples (6/13). IL-13 and GATA3 expression were observed in some intra-capsular small lymphocytes.
While IL-13 was also detected in BIA-ALCL cell lines, it was not found in 4 of the 7 systemic ALCL cases or the PTCL-NOS case.
Dr Kadin said the lack of IL-13 receptor expression in BIA-ALCL cell lines suggests that IL-13 is not an autocrine growth factor for BIA-ALCL, and its expression is most likely associated with an allergic immune response.
IL-13 is known to induce immunoglobulin class switching in plasma cells to produce IgE. H&E and Giemsa staining of the BIA-ALCL tumor tissue and involved regional lymph nodes revealed IgE and eosinophils on the surface of mast cells and follicular dendritic cells.
Taken together, these data point to an allergic reaction in breast implant capsules of BIA-ALCL.
Dr Kadin was hopeful that these findings could be extrapolated to prevent BIA-ALCL by identifying individuals at higher risk for developing the disease.
The next step for this research is to decipher the role of bacterial antigens in mediating the immune response and whether women who develop BIA-ALCL have a significant increase in other atopic conditions. ![]()
SAN FRANCISCO—A small study suggests an abnormal immune response characterized by the production of interleukin-13 (IL-13) underlies the pathogenesis of breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL).
The immune response appears to be directed toward bacterial antigens on the surface of the breast implants.
Marshall E. Kadin, MD, of Roger Williams Medical Center in Providence, Rhode Island, presented these findings at the 9th Annual T-cell Lymphoma Forum.
Dr Kadin noted that BIA-ALCL is a rare type of CD30+ T-cell ALCL that has been reported in more than 200 women worldwide.
Although a cause-and-effect relationship between breast implants and BIA-ALCL has been suggested, the underlying pathogenesis of this malignancy is unclear.
A bacterial biofilm containing gram-negative bacilli has been detected in breast implants from patients with BIA-ALCL.
Therefore, Dr Kadin and his colleagues hypothesized that an immune response toward the bacterial antigens may mediate the pathogenesis of BIA-ALCL.
The researchers studied 13 clinical samples of breast implant capsules and regional lymph nodes from 4 patients with BIA-ALCL, 7 patients with systemic ALCL, and 1 patient with peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS).
Immunohistochemistry was used to determine the presence of IL-13, IL-4, GATA3, and immunoglobulin E (IgE) in these samples.
All clinical samples of anaplastic cells from breast implant capsules tested positive for the presence of IL-13 (13/13). GATA3 was expressed in most anaplastic cell samples (12/13), and IL-4 expression was found in some anaplastic cell samples (6/13). IL-13 and GATA3 expression were observed in some intra-capsular small lymphocytes.
While IL-13 was also detected in BIA-ALCL cell lines, it was not found in 4 of the 7 systemic ALCL cases or the PTCL-NOS case.
Dr Kadin said the lack of IL-13 receptor expression in BIA-ALCL cell lines suggests that IL-13 is not an autocrine growth factor for BIA-ALCL, and its expression is most likely associated with an allergic immune response.
IL-13 is known to induce immunoglobulin class switching in plasma cells to produce IgE. H&E and Giemsa staining of the BIA-ALCL tumor tissue and involved regional lymph nodes revealed IgE and eosinophils on the surface of mast cells and follicular dendritic cells.
Taken together, these data point to an allergic reaction in breast implant capsules of BIA-ALCL.
Dr Kadin was hopeful that these findings could be extrapolated to prevent BIA-ALCL by identifying individuals at higher risk for developing the disease.
The next step for this research is to decipher the role of bacterial antigens in mediating the immune response and whether women who develop BIA-ALCL have a significant increase in other atopic conditions. ![]()
SAN FRANCISCO—A small study suggests an abnormal immune response characterized by the production of interleukin-13 (IL-13) underlies the pathogenesis of breast implant-associated anaplastic large-cell lymphoma (BIA-ALCL).
The immune response appears to be directed toward bacterial antigens on the surface of the breast implants.
Marshall E. Kadin, MD, of Roger Williams Medical Center in Providence, Rhode Island, presented these findings at the 9th Annual T-cell Lymphoma Forum.
Dr Kadin noted that BIA-ALCL is a rare type of CD30+ T-cell ALCL that has been reported in more than 200 women worldwide.
Although a cause-and-effect relationship between breast implants and BIA-ALCL has been suggested, the underlying pathogenesis of this malignancy is unclear.
A bacterial biofilm containing gram-negative bacilli has been detected in breast implants from patients with BIA-ALCL.
Therefore, Dr Kadin and his colleagues hypothesized that an immune response toward the bacterial antigens may mediate the pathogenesis of BIA-ALCL.
The researchers studied 13 clinical samples of breast implant capsules and regional lymph nodes from 4 patients with BIA-ALCL, 7 patients with systemic ALCL, and 1 patient with peripheral T-cell lymphoma-not otherwise specified (PTCL-NOS).
Immunohistochemistry was used to determine the presence of IL-13, IL-4, GATA3, and immunoglobulin E (IgE) in these samples.
All clinical samples of anaplastic cells from breast implant capsules tested positive for the presence of IL-13 (13/13). GATA3 was expressed in most anaplastic cell samples (12/13), and IL-4 expression was found in some anaplastic cell samples (6/13). IL-13 and GATA3 expression were observed in some intra-capsular small lymphocytes.
While IL-13 was also detected in BIA-ALCL cell lines, it was not found in 4 of the 7 systemic ALCL cases or the PTCL-NOS case.
Dr Kadin said the lack of IL-13 receptor expression in BIA-ALCL cell lines suggests that IL-13 is not an autocrine growth factor for BIA-ALCL, and its expression is most likely associated with an allergic immune response.
IL-13 is known to induce immunoglobulin class switching in plasma cells to produce IgE. H&E and Giemsa staining of the BIA-ALCL tumor tissue and involved regional lymph nodes revealed IgE and eosinophils on the surface of mast cells and follicular dendritic cells.
Taken together, these data point to an allergic reaction in breast implant capsules of BIA-ALCL.
Dr Kadin was hopeful that these findings could be extrapolated to prevent BIA-ALCL by identifying individuals at higher risk for developing the disease.
The next step for this research is to decipher the role of bacterial antigens in mediating the immune response and whether women who develop BIA-ALCL have a significant increase in other atopic conditions. ![]()
Use of Fluorodeoxyglucose-Positron Emission Tomography in the Diagnosis of Intravascular Diffuse Large B-Cell Lymphoma
Patient 1
A white man aged 67 years, with diastolic heart failure and chronic obstructive pulmonary disease, presented to the emergency department (ED) with shortness of breath. The initial laboratory results were significant for a newly elevated creatinine level of 2.06 mg/dL and a brain natriuretic peptide level of 648 pg/mL.
Imaging studies included a chest radiograph, a ventilation/perfusion scan, and an echocardiogram, as well as a right heart catheterization. All were nondiagnostic.
The patient's shortness of breath persisted despite treatment with diuretics, antibiotics, and steroids. Further laboratory workup revealed an elevated lactate dehydrogenase (LDH) level of 1,338 IU/L. A bone marrow biopsy performed because of concern about malignancy was unremarkable. Flow cytometry of the bone marrow aspirate did not reveal clonal B- or T-cell populations. Immunohistochemical staining was not performed. During this hospitalization for shortness of breath, the patient's mental staus began to decline, and his oxygen requirements increased. The patient was intubated but expired 48 hours after mechanical ventilation was initiated.
Patient 2
A white woman aged 67 years presented to the ED with generalized weakness, fatigue, and nausea. The patient’s medical history was significant for a diagnosis of stage IIIa ovarian cancer. She was treated with surgical resection and completed 6 cycles of adjuvant carboplatin and paclitaxel 3 months prior to this presentation. She had good response to treatment with normalization of CA-125.
After completion of chemotherapy, the patient was found to have persistent anemia and thrombocytopenia. Admission laboratory results were significant for a hemoglobin level of 8.4 g/dL, a platelet count of 20,000/μL, and an LDH level of 1,220 IU/L.
Chest, abdomen, and pelvis CT scans showed mesenteric adenopathy and splenomegaly (Figures 3A, 3B, and 3C) compared with prior imaging. Bone marrow biopsy revealed large lymphoid cells with scant cytoplasm and irregular nuclei, primarily within blood vessels and sinusoids consistent with IVLBCL (Figure 4). Flow cytometry of the bone marrow specimen showed an abnormal B-cell population with expression CD20, CD19, FMC-7, and dim κ light chain restriction. The cells were negative for CD5 and CD10. Immunohistochemical staining was positive for CD20, CD79a, PAX5, BCL-2 , and MUM1.
The patient was treated with 4 cycles of cyclophosphamide, doxorubicin, vincristine, prednisone, and rituximab, plus intrathecal methotrexate. The chemotherapy dose was reduced in the final cycle because of neuropathy in the hands and feet. The patient had undergone autologous stem-cell transplantation to allow high-dose chemotherapy. She was doing well more than 5 months after her transplant without evidence of recurrent disease.
Patient 3
A white man aged 76 years presented to the ED with cutaneous nodules, weight loss, fatigue, fevers, and epigastric pain. The patient’s medical history was significant for asymptomatic lymphoplasmacytic lymphoma diagnosed 2 months earlier, which had not required treatment. Laboratory results on admission revealed transaminitis, mild anemia with a hemoglobin level of 11 g/dL, and LDH level of 497 IU/L.
Chest, abdomen, and pelvis CT scans showed a 1.7cm hepatic lesion and mesenteric adenopathy. A bone marrow biopsy was unchanged from prior studies and showed minimal involvement (5%) of marrow space by low grade B-cell lymphoma.
Fluorodeoxyglucose-positron emission tomography (FDG-PET) scans showed multiple areas of uptake in the neck, chest, abdomen, and pelvis (Figures 5 and 6). No increased uptake in the subcutaneous nodules was noted on examination. Laparoscopic biopsy of FDG-avid mesenteric nodes showed clusters of atypical large lymphoid cells resulting in distention of the vascular lumina, resulting in the diagnosis of IVLBCL (Figure 7).
Immunohistochemical stains showed that the intravascular lymphocytes were strongly positive for CD20 and BCL-2 and negative for CD5 and CD10. Flow cytometry on the sample was limited by a low cell count and could not be assessed for clonality. The patient completed 6 cycles of rituximab as well as intrathecal methotrexate. Restaging studies showed a complete remission.
Two months later, the patient developed a skin nodule on the right shoulder. A repeat FDG-PET scan showed increased uptake, and fine-needle biopsy confirmed recurrent disease. The patient is undergoing treatment with ifosfamide, carboplatin, etoposide, and rituximab, as well as workup for autologous stem-cell transplant.
Discussion
Intravascular large B-cell lymphoma, a subtype of diffuse large B-cell lymphoma, is unique because it is primarily extranodal and typically without significant tumor burden.1-4 Standard imaging modalities, therefore, are often nonspecific and do not aid clinicians in establishing a diagnosis. Fluorodeoxyglucose-positron emission tomography has a known role in the assessment of diffuse large B-cell lymphoma, both at time of diagnosis and in monitoring response to treatment.5 However, the use of FDGPET in the diagnosis and management of IVLBCL has not been clearly established.
In a review of the literature, 26 English-language case reports and small case series reporting individual centers’ experience with the use of this imaging modality in the diagnosis of IVLBCL were identified. Two cases were eliminated from review because they did not discuss the use of FDG-PET in relationship to diagnosis. Of the remaining 24 cases, 21 underwent initial imaging with 1 or more of the following imaging modalities: CT, magnetic resonance, ultrasound, bone scan, and gallium scintigraphy, all of which were nonspecific and did not lead to a definitive diagnosis.3,6-25 Each of the 21 cases was followed up by FDG-PET; in 19, the FDG-PET scan was positive and resulted in a diagnosis of IVLBCL. In 2 cases, the FDG-PET scan was nonrevealing and was not considered helpful in diagnosis.11,18 In 3 of the 21 cases, the FDG-PET scan was the primary imaging modality.6,14,25
In this review, all 3 patients had initial imaging with CT scans of anatomic locations that were largely unrevealing, although later histologic examination showed them to be locations of active disease either by biopsy or on autopsy. One patient who underwent early FDG-PET was found to have increased uptake in the mesenteric lymph nodes, which were later biopsied, as well as uptake in the bilateral adrenal glands, lungs, and bone.
Several characteristic FDG-PET findings that have been described in the literature have been identified in patients with IVLBCL, including diffuse accumulation in bilateral lung fields, accumulation in the renal cortex or adrenal glands, diffuse bony involvement, and hypometabolism in the brain.7,10,12,13,17,23,25 These findings show that organs with the richest blood supply, specifically the lungs and kidneys, often are affected. The brain, an obligate glucose metabolizer, would be expected to have high uptake; however, with tumor thrombi occluding small intracranial vessels, micro infarcts ensue and are evidenced by areas of low uptake on FDG-PET scans in patients with IVLBCL.7 These characteristic patterns seen on FDG-PET scans can help to support a diagnosis of IVLBCL when clinical suspicion is high. Further, clinicians may be able to use imaging results to guide an appropriate site for biopsy to confirm diagnosis.
Conclusion
Intravascular large B-cell lymphoma remains a diagnostic challenge for clinicians. Prognosis is generally poor and likely related to frequent delays in diagnosis.1 Clinicians continue to work toward improving their ability to diagnose this disease in its early stages. New diagnostic algorithms and the use of random skin biopsies have shown some promise in improving diagnostic efficiency.26-28 Based on the authors’ experience and review of the literature, FDG-PET may be another promising tool to aid early diagnosis. Characteristic FDG-PET findings have been well described and may help to support the diagnosis of IVLBCL and guide an appropriate biopsy site when clinical suspicion for IVLBCL exists.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
Patient 1
A white man aged 67 years, with diastolic heart failure and chronic obstructive pulmonary disease, presented to the emergency department (ED) with shortness of breath. The initial laboratory results were significant for a newly elevated creatinine level of 2.06 mg/dL and a brain natriuretic peptide level of 648 pg/mL.
Imaging studies included a chest radiograph, a ventilation/perfusion scan, and an echocardiogram, as well as a right heart catheterization. All were nondiagnostic.
The patient's shortness of breath persisted despite treatment with diuretics, antibiotics, and steroids. Further laboratory workup revealed an elevated lactate dehydrogenase (LDH) level of 1,338 IU/L. A bone marrow biopsy performed because of concern about malignancy was unremarkable. Flow cytometry of the bone marrow aspirate did not reveal clonal B- or T-cell populations. Immunohistochemical staining was not performed. During this hospitalization for shortness of breath, the patient's mental staus began to decline, and his oxygen requirements increased. The patient was intubated but expired 48 hours after mechanical ventilation was initiated.
Patient 2
A white woman aged 67 years presented to the ED with generalized weakness, fatigue, and nausea. The patient’s medical history was significant for a diagnosis of stage IIIa ovarian cancer. She was treated with surgical resection and completed 6 cycles of adjuvant carboplatin and paclitaxel 3 months prior to this presentation. She had good response to treatment with normalization of CA-125.
After completion of chemotherapy, the patient was found to have persistent anemia and thrombocytopenia. Admission laboratory results were significant for a hemoglobin level of 8.4 g/dL, a platelet count of 20,000/μL, and an LDH level of 1,220 IU/L.
Chest, abdomen, and pelvis CT scans showed mesenteric adenopathy and splenomegaly (Figures 3A, 3B, and 3C) compared with prior imaging. Bone marrow biopsy revealed large lymphoid cells with scant cytoplasm and irregular nuclei, primarily within blood vessels and sinusoids consistent with IVLBCL (Figure 4). Flow cytometry of the bone marrow specimen showed an abnormal B-cell population with expression CD20, CD19, FMC-7, and dim κ light chain restriction. The cells were negative for CD5 and CD10. Immunohistochemical staining was positive for CD20, CD79a, PAX5, BCL-2 , and MUM1.
The patient was treated with 4 cycles of cyclophosphamide, doxorubicin, vincristine, prednisone, and rituximab, plus intrathecal methotrexate. The chemotherapy dose was reduced in the final cycle because of neuropathy in the hands and feet. The patient had undergone autologous stem-cell transplantation to allow high-dose chemotherapy. She was doing well more than 5 months after her transplant without evidence of recurrent disease.
Patient 3
A white man aged 76 years presented to the ED with cutaneous nodules, weight loss, fatigue, fevers, and epigastric pain. The patient’s medical history was significant for asymptomatic lymphoplasmacytic lymphoma diagnosed 2 months earlier, which had not required treatment. Laboratory results on admission revealed transaminitis, mild anemia with a hemoglobin level of 11 g/dL, and LDH level of 497 IU/L.
Chest, abdomen, and pelvis CT scans showed a 1.7cm hepatic lesion and mesenteric adenopathy. A bone marrow biopsy was unchanged from prior studies and showed minimal involvement (5%) of marrow space by low grade B-cell lymphoma.
Fluorodeoxyglucose-positron emission tomography (FDG-PET) scans showed multiple areas of uptake in the neck, chest, abdomen, and pelvis (Figures 5 and 6). No increased uptake in the subcutaneous nodules was noted on examination. Laparoscopic biopsy of FDG-avid mesenteric nodes showed clusters of atypical large lymphoid cells resulting in distention of the vascular lumina, resulting in the diagnosis of IVLBCL (Figure 7).
Immunohistochemical stains showed that the intravascular lymphocytes were strongly positive for CD20 and BCL-2 and negative for CD5 and CD10. Flow cytometry on the sample was limited by a low cell count and could not be assessed for clonality. The patient completed 6 cycles of rituximab as well as intrathecal methotrexate. Restaging studies showed a complete remission.
Two months later, the patient developed a skin nodule on the right shoulder. A repeat FDG-PET scan showed increased uptake, and fine-needle biopsy confirmed recurrent disease. The patient is undergoing treatment with ifosfamide, carboplatin, etoposide, and rituximab, as well as workup for autologous stem-cell transplant.
Discussion
Intravascular large B-cell lymphoma, a subtype of diffuse large B-cell lymphoma, is unique because it is primarily extranodal and typically without significant tumor burden.1-4 Standard imaging modalities, therefore, are often nonspecific and do not aid clinicians in establishing a diagnosis. Fluorodeoxyglucose-positron emission tomography has a known role in the assessment of diffuse large B-cell lymphoma, both at time of diagnosis and in monitoring response to treatment.5 However, the use of FDGPET in the diagnosis and management of IVLBCL has not been clearly established.
In a review of the literature, 26 English-language case reports and small case series reporting individual centers’ experience with the use of this imaging modality in the diagnosis of IVLBCL were identified. Two cases were eliminated from review because they did not discuss the use of FDG-PET in relationship to diagnosis. Of the remaining 24 cases, 21 underwent initial imaging with 1 or more of the following imaging modalities: CT, magnetic resonance, ultrasound, bone scan, and gallium scintigraphy, all of which were nonspecific and did not lead to a definitive diagnosis.3,6-25 Each of the 21 cases was followed up by FDG-PET; in 19, the FDG-PET scan was positive and resulted in a diagnosis of IVLBCL. In 2 cases, the FDG-PET scan was nonrevealing and was not considered helpful in diagnosis.11,18 In 3 of the 21 cases, the FDG-PET scan was the primary imaging modality.6,14,25
In this review, all 3 patients had initial imaging with CT scans of anatomic locations that were largely unrevealing, although later histologic examination showed them to be locations of active disease either by biopsy or on autopsy. One patient who underwent early FDG-PET was found to have increased uptake in the mesenteric lymph nodes, which were later biopsied, as well as uptake in the bilateral adrenal glands, lungs, and bone.
Several characteristic FDG-PET findings that have been described in the literature have been identified in patients with IVLBCL, including diffuse accumulation in bilateral lung fields, accumulation in the renal cortex or adrenal glands, diffuse bony involvement, and hypometabolism in the brain.7,10,12,13,17,23,25 These findings show that organs with the richest blood supply, specifically the lungs and kidneys, often are affected. The brain, an obligate glucose metabolizer, would be expected to have high uptake; however, with tumor thrombi occluding small intracranial vessels, micro infarcts ensue and are evidenced by areas of low uptake on FDG-PET scans in patients with IVLBCL.7 These characteristic patterns seen on FDG-PET scans can help to support a diagnosis of IVLBCL when clinical suspicion is high. Further, clinicians may be able to use imaging results to guide an appropriate site for biopsy to confirm diagnosis.
Conclusion
Intravascular large B-cell lymphoma remains a diagnostic challenge for clinicians. Prognosis is generally poor and likely related to frequent delays in diagnosis.1 Clinicians continue to work toward improving their ability to diagnose this disease in its early stages. New diagnostic algorithms and the use of random skin biopsies have shown some promise in improving diagnostic efficiency.26-28 Based on the authors’ experience and review of the literature, FDG-PET may be another promising tool to aid early diagnosis. Characteristic FDG-PET findings have been well described and may help to support the diagnosis of IVLBCL and guide an appropriate biopsy site when clinical suspicion for IVLBCL exists.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.
Click here to read the digital edition.
Patient 1
A white man aged 67 years, with diastolic heart failure and chronic obstructive pulmonary disease, presented to the emergency department (ED) with shortness of breath. The initial laboratory results were significant for a newly elevated creatinine level of 2.06 mg/dL and a brain natriuretic peptide level of 648 pg/mL.
Imaging studies included a chest radiograph, a ventilation/perfusion scan, and an echocardiogram, as well as a right heart catheterization. All were nondiagnostic.
The patient's shortness of breath persisted despite treatment with diuretics, antibiotics, and steroids. Further laboratory workup revealed an elevated lactate dehydrogenase (LDH) level of 1,338 IU/L. A bone marrow biopsy performed because of concern about malignancy was unremarkable. Flow cytometry of the bone marrow aspirate did not reveal clonal B- or T-cell populations. Immunohistochemical staining was not performed. During this hospitalization for shortness of breath, the patient's mental staus began to decline, and his oxygen requirements increased. The patient was intubated but expired 48 hours after mechanical ventilation was initiated.
Patient 2
A white woman aged 67 years presented to the ED with generalized weakness, fatigue, and nausea. The patient’s medical history was significant for a diagnosis of stage IIIa ovarian cancer. She was treated with surgical resection and completed 6 cycles of adjuvant carboplatin and paclitaxel 3 months prior to this presentation. She had good response to treatment with normalization of CA-125.
After completion of chemotherapy, the patient was found to have persistent anemia and thrombocytopenia. Admission laboratory results were significant for a hemoglobin level of 8.4 g/dL, a platelet count of 20,000/μL, and an LDH level of 1,220 IU/L.
Chest, abdomen, and pelvis CT scans showed mesenteric adenopathy and splenomegaly (Figures 3A, 3B, and 3C) compared with prior imaging. Bone marrow biopsy revealed large lymphoid cells with scant cytoplasm and irregular nuclei, primarily within blood vessels and sinusoids consistent with IVLBCL (Figure 4). Flow cytometry of the bone marrow specimen showed an abnormal B-cell population with expression CD20, CD19, FMC-7, and dim κ light chain restriction. The cells were negative for CD5 and CD10. Immunohistochemical staining was positive for CD20, CD79a, PAX5, BCL-2 , and MUM1.
The patient was treated with 4 cycles of cyclophosphamide, doxorubicin, vincristine, prednisone, and rituximab, plus intrathecal methotrexate. The chemotherapy dose was reduced in the final cycle because of neuropathy in the hands and feet. The patient had undergone autologous stem-cell transplantation to allow high-dose chemotherapy. She was doing well more than 5 months after her transplant without evidence of recurrent disease.
Patient 3
A white man aged 76 years presented to the ED with cutaneous nodules, weight loss, fatigue, fevers, and epigastric pain. The patient’s medical history was significant for asymptomatic lymphoplasmacytic lymphoma diagnosed 2 months earlier, which had not required treatment. Laboratory results on admission revealed transaminitis, mild anemia with a hemoglobin level of 11 g/dL, and LDH level of 497 IU/L.
Chest, abdomen, and pelvis CT scans showed a 1.7cm hepatic lesion and mesenteric adenopathy. A bone marrow biopsy was unchanged from prior studies and showed minimal involvement (5%) of marrow space by low grade B-cell lymphoma.
Fluorodeoxyglucose-positron emission tomography (FDG-PET) scans showed multiple areas of uptake in the neck, chest, abdomen, and pelvis (Figures 5 and 6). No increased uptake in the subcutaneous nodules was noted on examination. Laparoscopic biopsy of FDG-avid mesenteric nodes showed clusters of atypical large lymphoid cells resulting in distention of the vascular lumina, resulting in the diagnosis of IVLBCL (Figure 7).
Immunohistochemical stains showed that the intravascular lymphocytes were strongly positive for CD20 and BCL-2 and negative for CD5 and CD10. Flow cytometry on the sample was limited by a low cell count and could not be assessed for clonality. The patient completed 6 cycles of rituximab as well as intrathecal methotrexate. Restaging studies showed a complete remission.
Two months later, the patient developed a skin nodule on the right shoulder. A repeat FDG-PET scan showed increased uptake, and fine-needle biopsy confirmed recurrent disease. The patient is undergoing treatment with ifosfamide, carboplatin, etoposide, and rituximab, as well as workup for autologous stem-cell transplant.
Discussion
Intravascular large B-cell lymphoma, a subtype of diffuse large B-cell lymphoma, is unique because it is primarily extranodal and typically without significant tumor burden.1-4 Standard imaging modalities, therefore, are often nonspecific and do not aid clinicians in establishing a diagnosis. Fluorodeoxyglucose-positron emission tomography has a known role in the assessment of diffuse large B-cell lymphoma, both at time of diagnosis and in monitoring response to treatment.5 However, the use of FDGPET in the diagnosis and management of IVLBCL has not been clearly established.
In a review of the literature, 26 English-language case reports and small case series reporting individual centers’ experience with the use of this imaging modality in the diagnosis of IVLBCL were identified. Two cases were eliminated from review because they did not discuss the use of FDG-PET in relationship to diagnosis. Of the remaining 24 cases, 21 underwent initial imaging with 1 or more of the following imaging modalities: CT, magnetic resonance, ultrasound, bone scan, and gallium scintigraphy, all of which were nonspecific and did not lead to a definitive diagnosis.3,6-25 Each of the 21 cases was followed up by FDG-PET; in 19, the FDG-PET scan was positive and resulted in a diagnosis of IVLBCL. In 2 cases, the FDG-PET scan was nonrevealing and was not considered helpful in diagnosis.11,18 In 3 of the 21 cases, the FDG-PET scan was the primary imaging modality.6,14,25
In this review, all 3 patients had initial imaging with CT scans of anatomic locations that were largely unrevealing, although later histologic examination showed them to be locations of active disease either by biopsy or on autopsy. One patient who underwent early FDG-PET was found to have increased uptake in the mesenteric lymph nodes, which were later biopsied, as well as uptake in the bilateral adrenal glands, lungs, and bone.
Several characteristic FDG-PET findings that have been described in the literature have been identified in patients with IVLBCL, including diffuse accumulation in bilateral lung fields, accumulation in the renal cortex or adrenal glands, diffuse bony involvement, and hypometabolism in the brain.7,10,12,13,17,23,25 These findings show that organs with the richest blood supply, specifically the lungs and kidneys, often are affected. The brain, an obligate glucose metabolizer, would be expected to have high uptake; however, with tumor thrombi occluding small intracranial vessels, micro infarcts ensue and are evidenced by areas of low uptake on FDG-PET scans in patients with IVLBCL.7 These characteristic patterns seen on FDG-PET scans can help to support a diagnosis of IVLBCL when clinical suspicion is high. Further, clinicians may be able to use imaging results to guide an appropriate site for biopsy to confirm diagnosis.
Conclusion
Intravascular large B-cell lymphoma remains a diagnostic challenge for clinicians. Prognosis is generally poor and likely related to frequent delays in diagnosis.1 Clinicians continue to work toward improving their ability to diagnose this disease in its early stages. New diagnostic algorithms and the use of random skin biopsies have shown some promise in improving diagnostic efficiency.26-28 Based on the authors’ experience and review of the literature, FDG-PET may be another promising tool to aid early diagnosis. Characteristic FDG-PET findings have been well described and may help to support the diagnosis of IVLBCL and guide an appropriate biopsy site when clinical suspicion for IVLBCL exists.
Author disclosures
The authors report no actual or potential conflicts of interest with regard to this article.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect those of Federal Practitioner, Frontline Medical Communications Inc., the U.S. Government, or any of its agencies. This article may discuss unlabeled or investigational use of certain drugs. Please review complete prescribing information for specific drugs or drug combinations—including indications, contraindications, warnings, and adverse effects—before administering pharmacologic therapy to patients.