User login
Panel Calls for More Safety Data for Hepatitis B Vaccine
SILVER SPRING, MD. – Despite evidence of effectiveness and enthusiasm about its potential, more safety data in thousands of people are needed before a two-dose hepatitis B vaccine can be approved for use in adults, according to the majority of a Food and Drug Administration advisory panel.
At a meeting on Nov. 15, the FDA’s Vaccines and Related Biological Products Advisory Committee voted 13 to 1 that the immunogenicity data on the Heplisav vaccine were adequate to support its effectiveness in preventing hepatitis B infection in adults aged 18-70 years. The proposed indication for the vaccine, which combines hepatitis B surface antigen (HBsAg) with a novel adjuvant to enhance the immune response, is for the active immunization against all known subtypes of the hepatitis B virus in adults aged 18-70 years.
But the panel voted 8 to 5, with one abstention, that the available data were not adequate to support the safety of the vaccine, citing the need for more data because the adjuvant, a Toll-like receptor 9 agonist, is not included in any available vaccine. Almost 4,000 people received the vaccine in two phase III studies. The manufacturer, Dynavax Technologies, also has proposed a postmarketing safety study that will enroll up to 30,000 recipients of the vaccine in a managed care organization. Panelists voting no on the safety question said that more data from a more ethnically diverse population than those enrolled in the studies would be needed in as many as 10,000 patients before approval.
While a hepatitis B vaccine that is more immunogenic in populations that do not respond as well to the hepatitis B vaccines would be beneficial, "I don’t think the safety data is sufficiently large to support a recommendation for use in the general adult population given that this vaccine contains a new adjuvant," said one of the panelists, Dr. Melinda Wharton, deputy director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention.
The two Heplisav doses are administered intramuscularly 1 month apart, compared with the 0, 1, and 6 month schedule for the two currently approved hepatitis B vaccines, Engerix-B and Recombivax HB.
The two phase III noninferiority studies of healthy adults aged 18-70 years compared immune response to vaccination with Heplisav (administered at 0 and 1 months, with a saline placebo administered at 6 months) in 3,778 adults, and with Engerix-B (administered at 0, 1, and 6 months) in 1,089 people. The primary immunogenicity end point was the seroprotection rate (SPR) – an anti-HBsAg level of 10 mIU/mL or greater, recognized as conferring protection against hepatitis B virus infection. In both studies, the SPR results for Heplisav met the noninferiority criteria for the studies.
In the two studies, the SPRs were higher among those who received Heplisav: 95% and 90% at 3 months (8 weeks after the last active dose), compared with 81.1% (4 weeks after the last dose) and 70.5% (8 weeks after the last dose), respectively, of those who received Engerix-B.
The most common adverse event associated with the vaccine was injection-site reaction in both groups. Rates of severe adverse events were lower among those who received Heplisav, and rates of autoimmune events and autoantibody conversions were similar in the two groups, according to Dynavax.
However, thyroid-related adverse events, which could be representative of autoimmune events, were reported in a higher proportion of people who received Heplisav. Cases of serious events, although rare, included one of Wegener’s granulomatosis and one of Guillain-Barré syndrome. Autoimmune diseases are relatively rare in the general population, and a large sample size of patients was necessary to accurately evaluate the associated risk, according to the FDA reviewer.
An increased risk of autoimmune reactions is a theoretical risk with adjuvants.
The FDA’s deadline for making a decision on the approval is Feb. 24, 2013, according to Dynavax. If approved, the company plans to market the vaccine as Heplisav. The vaccine also is under review in Europe.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting, although a panelist may be given a waiver for conflict of interest, but none were granted at this meeting.
SILVER SPRING, MD. – Despite evidence of effectiveness and enthusiasm about its potential, more safety data in thousands of people are needed before a two-dose hepatitis B vaccine can be approved for use in adults, according to the majority of a Food and Drug Administration advisory panel.
At a meeting on Nov. 15, the FDA’s Vaccines and Related Biological Products Advisory Committee voted 13 to 1 that the immunogenicity data on the Heplisav vaccine were adequate to support its effectiveness in preventing hepatitis B infection in adults aged 18-70 years. The proposed indication for the vaccine, which combines hepatitis B surface antigen (HBsAg) with a novel adjuvant to enhance the immune response, is for the active immunization against all known subtypes of the hepatitis B virus in adults aged 18-70 years.
But the panel voted 8 to 5, with one abstention, that the available data were not adequate to support the safety of the vaccine, citing the need for more data because the adjuvant, a Toll-like receptor 9 agonist, is not included in any available vaccine. Almost 4,000 people received the vaccine in two phase III studies. The manufacturer, Dynavax Technologies, also has proposed a postmarketing safety study that will enroll up to 30,000 recipients of the vaccine in a managed care organization. Panelists voting no on the safety question said that more data from a more ethnically diverse population than those enrolled in the studies would be needed in as many as 10,000 patients before approval.
While a hepatitis B vaccine that is more immunogenic in populations that do not respond as well to the hepatitis B vaccines would be beneficial, "I don’t think the safety data is sufficiently large to support a recommendation for use in the general adult population given that this vaccine contains a new adjuvant," said one of the panelists, Dr. Melinda Wharton, deputy director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention.
The two Heplisav doses are administered intramuscularly 1 month apart, compared with the 0, 1, and 6 month schedule for the two currently approved hepatitis B vaccines, Engerix-B and Recombivax HB.
The two phase III noninferiority studies of healthy adults aged 18-70 years compared immune response to vaccination with Heplisav (administered at 0 and 1 months, with a saline placebo administered at 6 months) in 3,778 adults, and with Engerix-B (administered at 0, 1, and 6 months) in 1,089 people. The primary immunogenicity end point was the seroprotection rate (SPR) – an anti-HBsAg level of 10 mIU/mL or greater, recognized as conferring protection against hepatitis B virus infection. In both studies, the SPR results for Heplisav met the noninferiority criteria for the studies.
In the two studies, the SPRs were higher among those who received Heplisav: 95% and 90% at 3 months (8 weeks after the last active dose), compared with 81.1% (4 weeks after the last dose) and 70.5% (8 weeks after the last dose), respectively, of those who received Engerix-B.
The most common adverse event associated with the vaccine was injection-site reaction in both groups. Rates of severe adverse events were lower among those who received Heplisav, and rates of autoimmune events and autoantibody conversions were similar in the two groups, according to Dynavax.
However, thyroid-related adverse events, which could be representative of autoimmune events, were reported in a higher proportion of people who received Heplisav. Cases of serious events, although rare, included one of Wegener’s granulomatosis and one of Guillain-Barré syndrome. Autoimmune diseases are relatively rare in the general population, and a large sample size of patients was necessary to accurately evaluate the associated risk, according to the FDA reviewer.
An increased risk of autoimmune reactions is a theoretical risk with adjuvants.
The FDA’s deadline for making a decision on the approval is Feb. 24, 2013, according to Dynavax. If approved, the company plans to market the vaccine as Heplisav. The vaccine also is under review in Europe.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting, although a panelist may be given a waiver for conflict of interest, but none were granted at this meeting.
SILVER SPRING, MD. – Despite evidence of effectiveness and enthusiasm about its potential, more safety data in thousands of people are needed before a two-dose hepatitis B vaccine can be approved for use in adults, according to the majority of a Food and Drug Administration advisory panel.
At a meeting on Nov. 15, the FDA’s Vaccines and Related Biological Products Advisory Committee voted 13 to 1 that the immunogenicity data on the Heplisav vaccine were adequate to support its effectiveness in preventing hepatitis B infection in adults aged 18-70 years. The proposed indication for the vaccine, which combines hepatitis B surface antigen (HBsAg) with a novel adjuvant to enhance the immune response, is for the active immunization against all known subtypes of the hepatitis B virus in adults aged 18-70 years.
But the panel voted 8 to 5, with one abstention, that the available data were not adequate to support the safety of the vaccine, citing the need for more data because the adjuvant, a Toll-like receptor 9 agonist, is not included in any available vaccine. Almost 4,000 people received the vaccine in two phase III studies. The manufacturer, Dynavax Technologies, also has proposed a postmarketing safety study that will enroll up to 30,000 recipients of the vaccine in a managed care organization. Panelists voting no on the safety question said that more data from a more ethnically diverse population than those enrolled in the studies would be needed in as many as 10,000 patients before approval.
While a hepatitis B vaccine that is more immunogenic in populations that do not respond as well to the hepatitis B vaccines would be beneficial, "I don’t think the safety data is sufficiently large to support a recommendation for use in the general adult population given that this vaccine contains a new adjuvant," said one of the panelists, Dr. Melinda Wharton, deputy director of the National Center for Immunization and Respiratory Diseases at the Centers for Disease Control and Prevention.
The two Heplisav doses are administered intramuscularly 1 month apart, compared with the 0, 1, and 6 month schedule for the two currently approved hepatitis B vaccines, Engerix-B and Recombivax HB.
The two phase III noninferiority studies of healthy adults aged 18-70 years compared immune response to vaccination with Heplisav (administered at 0 and 1 months, with a saline placebo administered at 6 months) in 3,778 adults, and with Engerix-B (administered at 0, 1, and 6 months) in 1,089 people. The primary immunogenicity end point was the seroprotection rate (SPR) – an anti-HBsAg level of 10 mIU/mL or greater, recognized as conferring protection against hepatitis B virus infection. In both studies, the SPR results for Heplisav met the noninferiority criteria for the studies.
In the two studies, the SPRs were higher among those who received Heplisav: 95% and 90% at 3 months (8 weeks after the last active dose), compared with 81.1% (4 weeks after the last dose) and 70.5% (8 weeks after the last dose), respectively, of those who received Engerix-B.
The most common adverse event associated with the vaccine was injection-site reaction in both groups. Rates of severe adverse events were lower among those who received Heplisav, and rates of autoimmune events and autoantibody conversions were similar in the two groups, according to Dynavax.
However, thyroid-related adverse events, which could be representative of autoimmune events, were reported in a higher proportion of people who received Heplisav. Cases of serious events, although rare, included one of Wegener’s granulomatosis and one of Guillain-Barré syndrome. Autoimmune diseases are relatively rare in the general population, and a large sample size of patients was necessary to accurately evaluate the associated risk, according to the FDA reviewer.
An increased risk of autoimmune reactions is a theoretical risk with adjuvants.
The FDA’s deadline for making a decision on the approval is Feb. 24, 2013, according to Dynavax. If approved, the company plans to market the vaccine as Heplisav. The vaccine also is under review in Europe.
The FDA usually follows the recommendations of its advisory panels, which are not binding. Panelists have been cleared of potential conflicts of interest related to the topic of the meeting, although a panelist may be given a waiver for conflict of interest, but none were granted at this meeting.
AT A MEETING OF THE FDA'S VACCINES AND RELATED BIOLOGICAL PRODUCTS ADVISORY COMMITTEE
Excessive Acetaminophen Dosing Seen Among Inpatients
Four percent of hospitalized adolescents and adults at two academic tertiary care medical centers received supratherapeutic doses of acetaminophen during their stays, according to a report published online Nov. 12 in Archives of Internal Medicine.
"These were not isolated events but often were successive and overlapping." Nearly half of the exposed patients received 5 g or more of acetaminophen per day, and 40% received excessive dosing for 3 or more days, said Dr. Li Zhou of Partners HealthCare System, Wellesley, Mass., and her associates.
Such overmedicating puts patients at unnecessary risk for hepatotoxicity, acute liver failure, and even death. In this study, even inpatients at specific risk for liver damage didn’t escape excessive dosing with acetaminophen: 22% of the elderly and 18% of patients with existing chronic liver disease were given acetaminophen in amounts exceeding their recommended limit of 3 g/day, said Dr. Zhou, who is also at Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues.
The risk factors most strongly associated with supratherapeutic dosing were receiving recurring scheduled dosing (that is, standing orders) as opposed to as-needed or one-time-only administration, which carried a hazard ratio of 16.6; receiving multiple products containing acetaminophen, which carried a hazard ratio of 2.4 for each additional product; and receiving products containing 500 mg of acetaminophen, which carried an HR of 1.9.
The investigators used the hospitals’ electronic medication administration record systems to assess acetaminophen exposure in the inpatient setting – a topic that has received little attention from researchers before now, they wrote. The study population comprised the 14,411 inpatients aged 12 and older who received any acetaminophen during their stays in a 3-month period. That accounts for about 61% of the entire patient population hospitalized at the two centers during the study period.
The average age of the study subjects was 55 years (range, 12-110 years), and a little more than one-third were older than 65. Approximately 75 had chronic liver disease.
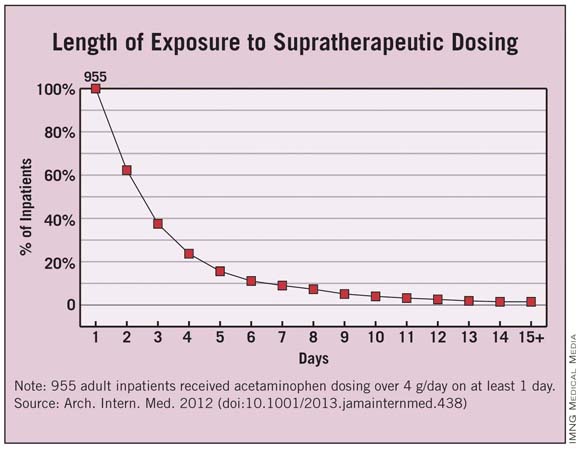
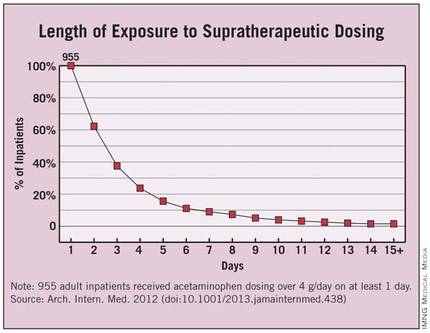
Overall, 955 patients were given acetaminophen in doses exceeding the 4-g/day limit. This represents 4% of the entire hospitalized population and 6.6% of the cohort of patients given any acetaminophen, the researchers said (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.438]).
Each patient who received supratherapeutic dosing had a mean of five such incidents during the course of a mean of 3 days. Instances of such overmedicating ranged as high as 48 separate occasions over the course of 30 days.
Approximately 40% of the 955 patients received supratherapeutic dosing for 3 or more days, and 4% received it for 10 or more days.
"Our findings show that despite policies and procedures to monitor and control patients’ acetaminophen exposure, the incidence of supratherapeutic acetaminophen dosing in hospitalized patients remains high," Dr. Zhou and her associates said.
Previous studies have shown that many physicians and nurses aren’t aware of the maximal recommended daily dosing of acetaminophen, and that many have some difficulty identifying which products contain the agent. Therefore, increased training is warranted "to help clinicians identify acetaminophen-containing products and monitor closely the daily dose of acetaminophen," the investigators said.
A reassessment of long-accepted dosing regimens may also be warranted, "especially the scheduled use of products containing 500 mg of acetaminophen," they added.
The study was funded by the Partners-Siemens Research Council. The investigators reported no relevant financial conflicts.
Dr. Li Zhou and her colleagues have identified a serious threat to patient safety and a cause for great concern, said Dr. Walter H. Ettinger.
"On an annualized basis, more than 3,800 patients at these two hospitals alone were put at unnecessary risk. If the estimates in this manuscript are generalizable, hundreds of thousands of patients nationwide may be receiving toxic doses of acetaminophen while in the hospital," he noted.
The study is important, "but it is incomplete and illustrates the obstacles we must overcome in using [health information technology] to drive performance. The authors identified a major flaw in the way in which a common drug is used in the hospital setting, and in a rapid-learning, high-performance health system, the results would be rapidly disseminated, together with a definitive solution for preventing excessive dosing in the future. The authors suggested educating health care providers about the risks of acetaminophen use and changing hospital policies to reduce risk factors for excessive dosing as a means of prevention. But excessive dosing of acetaminophen should be a ‘never event.’ The best way to prevent excessive dosing is to engineer a process and imbed it in the [electronic health record] such that it creates a hard stop that prevents ordering and administering more than 4 g/day of acetaminophen."
Dr. Ettinger is at the University of Massachusetts, Worcester, and at Accretive Health, Chicago, a health care technology and services firm. He reported no other relevant financial conflicts. These remarks were taken from his invited commentary accompanying Dr. Zhou’s report (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.607]).
Dr. Li Zhou and her colleagues have identified a serious threat to patient safety and a cause for great concern, said Dr. Walter H. Ettinger.
"On an annualized basis, more than 3,800 patients at these two hospitals alone were put at unnecessary risk. If the estimates in this manuscript are generalizable, hundreds of thousands of patients nationwide may be receiving toxic doses of acetaminophen while in the hospital," he noted.
The study is important, "but it is incomplete and illustrates the obstacles we must overcome in using [health information technology] to drive performance. The authors identified a major flaw in the way in which a common drug is used in the hospital setting, and in a rapid-learning, high-performance health system, the results would be rapidly disseminated, together with a definitive solution for preventing excessive dosing in the future. The authors suggested educating health care providers about the risks of acetaminophen use and changing hospital policies to reduce risk factors for excessive dosing as a means of prevention. But excessive dosing of acetaminophen should be a ‘never event.’ The best way to prevent excessive dosing is to engineer a process and imbed it in the [electronic health record] such that it creates a hard stop that prevents ordering and administering more than 4 g/day of acetaminophen."
Dr. Ettinger is at the University of Massachusetts, Worcester, and at Accretive Health, Chicago, a health care technology and services firm. He reported no other relevant financial conflicts. These remarks were taken from his invited commentary accompanying Dr. Zhou’s report (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.607]).
Dr. Li Zhou and her colleagues have identified a serious threat to patient safety and a cause for great concern, said Dr. Walter H. Ettinger.
"On an annualized basis, more than 3,800 patients at these two hospitals alone were put at unnecessary risk. If the estimates in this manuscript are generalizable, hundreds of thousands of patients nationwide may be receiving toxic doses of acetaminophen while in the hospital," he noted.
The study is important, "but it is incomplete and illustrates the obstacles we must overcome in using [health information technology] to drive performance. The authors identified a major flaw in the way in which a common drug is used in the hospital setting, and in a rapid-learning, high-performance health system, the results would be rapidly disseminated, together with a definitive solution for preventing excessive dosing in the future. The authors suggested educating health care providers about the risks of acetaminophen use and changing hospital policies to reduce risk factors for excessive dosing as a means of prevention. But excessive dosing of acetaminophen should be a ‘never event.’ The best way to prevent excessive dosing is to engineer a process and imbed it in the [electronic health record] such that it creates a hard stop that prevents ordering and administering more than 4 g/day of acetaminophen."
Dr. Ettinger is at the University of Massachusetts, Worcester, and at Accretive Health, Chicago, a health care technology and services firm. He reported no other relevant financial conflicts. These remarks were taken from his invited commentary accompanying Dr. Zhou’s report (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.607]).
Four percent of hospitalized adolescents and adults at two academic tertiary care medical centers received supratherapeutic doses of acetaminophen during their stays, according to a report published online Nov. 12 in Archives of Internal Medicine.
"These were not isolated events but often were successive and overlapping." Nearly half of the exposed patients received 5 g or more of acetaminophen per day, and 40% received excessive dosing for 3 or more days, said Dr. Li Zhou of Partners HealthCare System, Wellesley, Mass., and her associates.

Such overmedicating puts patients at unnecessary risk for hepatotoxicity, acute liver failure, and even death. In this study, even inpatients at specific risk for liver damage didn’t escape excessive dosing with acetaminophen: 22% of the elderly and 18% of patients with existing chronic liver disease were given acetaminophen in amounts exceeding their recommended limit of 3 g/day, said Dr. Zhou, who is also at Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues.
The risk factors most strongly associated with supratherapeutic dosing were receiving recurring scheduled dosing (that is, standing orders) as opposed to as-needed or one-time-only administration, which carried a hazard ratio of 16.6; receiving multiple products containing acetaminophen, which carried a hazard ratio of 2.4 for each additional product; and receiving products containing 500 mg of acetaminophen, which carried an HR of 1.9.
The investigators used the hospitals’ electronic medication administration record systems to assess acetaminophen exposure in the inpatient setting – a topic that has received little attention from researchers before now, they wrote. The study population comprised the 14,411 inpatients aged 12 and older who received any acetaminophen during their stays in a 3-month period. That accounts for about 61% of the entire patient population hospitalized at the two centers during the study period.
The average age of the study subjects was 55 years (range, 12-110 years), and a little more than one-third were older than 65. Approximately 75 had chronic liver disease.
Overall, 955 patients were given acetaminophen in doses exceeding the 4-g/day limit. This represents 4% of the entire hospitalized population and 6.6% of the cohort of patients given any acetaminophen, the researchers said (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.438]).
Each patient who received supratherapeutic dosing had a mean of five such incidents during the course of a mean of 3 days. Instances of such overmedicating ranged as high as 48 separate occasions over the course of 30 days.
Approximately 40% of the 955 patients received supratherapeutic dosing for 3 or more days, and 4% received it for 10 or more days.
"Our findings show that despite policies and procedures to monitor and control patients’ acetaminophen exposure, the incidence of supratherapeutic acetaminophen dosing in hospitalized patients remains high," Dr. Zhou and her associates said.
Previous studies have shown that many physicians and nurses aren’t aware of the maximal recommended daily dosing of acetaminophen, and that many have some difficulty identifying which products contain the agent. Therefore, increased training is warranted "to help clinicians identify acetaminophen-containing products and monitor closely the daily dose of acetaminophen," the investigators said.
A reassessment of long-accepted dosing regimens may also be warranted, "especially the scheduled use of products containing 500 mg of acetaminophen," they added.
The study was funded by the Partners-Siemens Research Council. The investigators reported no relevant financial conflicts.
Four percent of hospitalized adolescents and adults at two academic tertiary care medical centers received supratherapeutic doses of acetaminophen during their stays, according to a report published online Nov. 12 in Archives of Internal Medicine.
"These were not isolated events but often were successive and overlapping." Nearly half of the exposed patients received 5 g or more of acetaminophen per day, and 40% received excessive dosing for 3 or more days, said Dr. Li Zhou of Partners HealthCare System, Wellesley, Mass., and her associates.

Such overmedicating puts patients at unnecessary risk for hepatotoxicity, acute liver failure, and even death. In this study, even inpatients at specific risk for liver damage didn’t escape excessive dosing with acetaminophen: 22% of the elderly and 18% of patients with existing chronic liver disease were given acetaminophen in amounts exceeding their recommended limit of 3 g/day, said Dr. Zhou, who is also at Brigham and Women’s Hospital and Harvard Medical School, Boston, and her colleagues.
The risk factors most strongly associated with supratherapeutic dosing were receiving recurring scheduled dosing (that is, standing orders) as opposed to as-needed or one-time-only administration, which carried a hazard ratio of 16.6; receiving multiple products containing acetaminophen, which carried a hazard ratio of 2.4 for each additional product; and receiving products containing 500 mg of acetaminophen, which carried an HR of 1.9.
The investigators used the hospitals’ electronic medication administration record systems to assess acetaminophen exposure in the inpatient setting – a topic that has received little attention from researchers before now, they wrote. The study population comprised the 14,411 inpatients aged 12 and older who received any acetaminophen during their stays in a 3-month period. That accounts for about 61% of the entire patient population hospitalized at the two centers during the study period.
The average age of the study subjects was 55 years (range, 12-110 years), and a little more than one-third were older than 65. Approximately 75 had chronic liver disease.
Overall, 955 patients were given acetaminophen in doses exceeding the 4-g/day limit. This represents 4% of the entire hospitalized population and 6.6% of the cohort of patients given any acetaminophen, the researchers said (Arch. Intern. Med. 2012 [doi:10.1001/2013.jamainternmed.438]).
Each patient who received supratherapeutic dosing had a mean of five such incidents during the course of a mean of 3 days. Instances of such overmedicating ranged as high as 48 separate occasions over the course of 30 days.
Approximately 40% of the 955 patients received supratherapeutic dosing for 3 or more days, and 4% received it for 10 or more days.
"Our findings show that despite policies and procedures to monitor and control patients’ acetaminophen exposure, the incidence of supratherapeutic acetaminophen dosing in hospitalized patients remains high," Dr. Zhou and her associates said.
Previous studies have shown that many physicians and nurses aren’t aware of the maximal recommended daily dosing of acetaminophen, and that many have some difficulty identifying which products contain the agent. Therefore, increased training is warranted "to help clinicians identify acetaminophen-containing products and monitor closely the daily dose of acetaminophen," the investigators said.
A reassessment of long-accepted dosing regimens may also be warranted, "especially the scheduled use of products containing 500 mg of acetaminophen," they added.
The study was funded by the Partners-Siemens Research Council. The investigators reported no relevant financial conflicts.
FROM ARCHIVES OF INTERNAL MEDICINE
Major Finding: Four percent of all adolescent and adult inpatients at two hospitals received supratherapeutic doses of acetaminophen during their stays, including 22% of the elderly and 18% of patients with chronic liver disease.
Data Source: Data are based on acetaminophen exposure determined from electronic medication administration records for 14,411 inpatients aged 12 and older at two urban academic tertiary care hospitals during a 3-month period.
Disclosures: The study was funded by the Partners-Siemens Research Council. The authors reported no relevant financial conflicts.
Benefit of Young Liver Donors Scrutinized in Study
BOSTON – Younger age of deceased liver donors appeared to improve patient and graft survival when donors and recipients were both of younger age, but not when younger donor livers went to older recipients, according to a secondary analysis of the Organ Procurement and Transplantation Network national database from 2002 to 2011.
"Matching younger recipients with younger donors appears to be the best strategy in order to maximize life years after liver transplantation. However, before this can be considered for allocation policy, much more work needs to be done," including analyses specific to United Network for Organ Sharing regions, Model for End-Stage Liver Disease groups, and modeling of the potential life-years saved with a change in allocation policy, said lead investigator Dr. Neehar D. Parikh of the Comprehensive Transplant Center at Northwestern University, Chicago.
The risk of recipient death is known to increase with advancing donor age, beginning at ages older than 40 years and especially with donors older than 60 years. Also, advancing recipient age has been shown, with some controversy, to affect outcomes negatively, Dr. Parikh said at the annual meeting of the American Association for the Study of Liver Diseases.
Dr. Parikh and his colleagues’ study of the Organ Procurement and Transplantation Network database analyzed 26,849 cases of liver transplantation in which the donor was younger than the recipient, 782 cases in which they were the same age, and 12,107 cases in which the donor was older than the recipient. They also looked at outcomes of patients aged 18-40 years and those 60 years or older, based on the donor age.
As would be expected, recipients had progressively younger mean ages as the donor age increased: 56 years for recipients with younger donors, 52 years for same-age donors, and 48 years for older donors. Hepatitis C virus seropositivity was lowest in recipients who had an older donor (41%), followed by same-age recipient-donor pairs (51%), and recipients who had a younger donor (50%).
In Kaplan-Meier analyses, patient survival at 1 year was significantly worse for those who received a liver from a donor with an older age (86%) or the same age (86%) than from a younger donor (87.4%). Similar results were seen at 3 years between recipients of livers from older (74.5%), same age (73.5%), and younger donors (77.9%).
Older donor age increased the risk for patient death by 14% and graft failure by 13%, in comparison with younger donor age in an adjusted analysis.
For every 10-year increase in patient age for younger donors, the risk of patient mortality increased by 24% and graft failure by 17%. For every 10 years the donor was younger than the recipient, however, there was a 27% decline in the risk of patient mortality and 30% decrease in the risk of graft failure. A small but significance beneficial interaction was found between young donor age and young recipient age on both outcomes.
In contrast, for every 10 years the donor was older than the recipient, the risk of patient mortality increased 9% and the risk of graft failure increased by 22%, and there was no significant interaction between recipient and donor age.
When compared against donor and recipient pairs who were both aged 18-40 years, the risk of patient mortality and graft failure increased significantly for all other combinations of donor and recipient pairs: recipients aged 18-40 years and donors aged 60 years or older (hazard ratios of 1.76 and 1.49, respectively), recipients aged 60 years older and donors aged 18-40 years (HRs of 1.67 and 1.83), and recipients and donors both aged 60 years or older (HRs of 2.12 and 1.87). These results were consistent at both 1 and 3 years.
The overall results "suggest that there is something intrinsic about being older or having an older graft that cannot be remediated by graft or patient age factors," Dr. Parikh said.
The results might be explained in livers from older donors by impaired liver regeneration and increased susceptibility to ischemic reperfusion injury, while older recipients might have decreased circulating stem cells and more competing risks that are not adequately risk adjusted in the available data in older recipients, he said.
One audience member noted that there is still a "huge survival advantage" in all patient populations that receive younger donor livers and that allocating organs based on age is a slippery slope.
Dr. Parikh and his colleagues had no relevant financial disclosures.
BOSTON – Younger age of deceased liver donors appeared to improve patient and graft survival when donors and recipients were both of younger age, but not when younger donor livers went to older recipients, according to a secondary analysis of the Organ Procurement and Transplantation Network national database from 2002 to 2011.
"Matching younger recipients with younger donors appears to be the best strategy in order to maximize life years after liver transplantation. However, before this can be considered for allocation policy, much more work needs to be done," including analyses specific to United Network for Organ Sharing regions, Model for End-Stage Liver Disease groups, and modeling of the potential life-years saved with a change in allocation policy, said lead investigator Dr. Neehar D. Parikh of the Comprehensive Transplant Center at Northwestern University, Chicago.
The risk of recipient death is known to increase with advancing donor age, beginning at ages older than 40 years and especially with donors older than 60 years. Also, advancing recipient age has been shown, with some controversy, to affect outcomes negatively, Dr. Parikh said at the annual meeting of the American Association for the Study of Liver Diseases.
Dr. Parikh and his colleagues’ study of the Organ Procurement and Transplantation Network database analyzed 26,849 cases of liver transplantation in which the donor was younger than the recipient, 782 cases in which they were the same age, and 12,107 cases in which the donor was older than the recipient. They also looked at outcomes of patients aged 18-40 years and those 60 years or older, based on the donor age.
As would be expected, recipients had progressively younger mean ages as the donor age increased: 56 years for recipients with younger donors, 52 years for same-age donors, and 48 years for older donors. Hepatitis C virus seropositivity was lowest in recipients who had an older donor (41%), followed by same-age recipient-donor pairs (51%), and recipients who had a younger donor (50%).
In Kaplan-Meier analyses, patient survival at 1 year was significantly worse for those who received a liver from a donor with an older age (86%) or the same age (86%) than from a younger donor (87.4%). Similar results were seen at 3 years between recipients of livers from older (74.5%), same age (73.5%), and younger donors (77.9%).
Older donor age increased the risk for patient death by 14% and graft failure by 13%, in comparison with younger donor age in an adjusted analysis.
For every 10-year increase in patient age for younger donors, the risk of patient mortality increased by 24% and graft failure by 17%. For every 10 years the donor was younger than the recipient, however, there was a 27% decline in the risk of patient mortality and 30% decrease in the risk of graft failure. A small but significance beneficial interaction was found between young donor age and young recipient age on both outcomes.
In contrast, for every 10 years the donor was older than the recipient, the risk of patient mortality increased 9% and the risk of graft failure increased by 22%, and there was no significant interaction between recipient and donor age.
When compared against donor and recipient pairs who were both aged 18-40 years, the risk of patient mortality and graft failure increased significantly for all other combinations of donor and recipient pairs: recipients aged 18-40 years and donors aged 60 years or older (hazard ratios of 1.76 and 1.49, respectively), recipients aged 60 years older and donors aged 18-40 years (HRs of 1.67 and 1.83), and recipients and donors both aged 60 years or older (HRs of 2.12 and 1.87). These results were consistent at both 1 and 3 years.
The overall results "suggest that there is something intrinsic about being older or having an older graft that cannot be remediated by graft or patient age factors," Dr. Parikh said.
The results might be explained in livers from older donors by impaired liver regeneration and increased susceptibility to ischemic reperfusion injury, while older recipients might have decreased circulating stem cells and more competing risks that are not adequately risk adjusted in the available data in older recipients, he said.
One audience member noted that there is still a "huge survival advantage" in all patient populations that receive younger donor livers and that allocating organs based on age is a slippery slope.
Dr. Parikh and his colleagues had no relevant financial disclosures.
BOSTON – Younger age of deceased liver donors appeared to improve patient and graft survival when donors and recipients were both of younger age, but not when younger donor livers went to older recipients, according to a secondary analysis of the Organ Procurement and Transplantation Network national database from 2002 to 2011.
"Matching younger recipients with younger donors appears to be the best strategy in order to maximize life years after liver transplantation. However, before this can be considered for allocation policy, much more work needs to be done," including analyses specific to United Network for Organ Sharing regions, Model for End-Stage Liver Disease groups, and modeling of the potential life-years saved with a change in allocation policy, said lead investigator Dr. Neehar D. Parikh of the Comprehensive Transplant Center at Northwestern University, Chicago.
The risk of recipient death is known to increase with advancing donor age, beginning at ages older than 40 years and especially with donors older than 60 years. Also, advancing recipient age has been shown, with some controversy, to affect outcomes negatively, Dr. Parikh said at the annual meeting of the American Association for the Study of Liver Diseases.
Dr. Parikh and his colleagues’ study of the Organ Procurement and Transplantation Network database analyzed 26,849 cases of liver transplantation in which the donor was younger than the recipient, 782 cases in which they were the same age, and 12,107 cases in which the donor was older than the recipient. They also looked at outcomes of patients aged 18-40 years and those 60 years or older, based on the donor age.
As would be expected, recipients had progressively younger mean ages as the donor age increased: 56 years for recipients with younger donors, 52 years for same-age donors, and 48 years for older donors. Hepatitis C virus seropositivity was lowest in recipients who had an older donor (41%), followed by same-age recipient-donor pairs (51%), and recipients who had a younger donor (50%).
In Kaplan-Meier analyses, patient survival at 1 year was significantly worse for those who received a liver from a donor with an older age (86%) or the same age (86%) than from a younger donor (87.4%). Similar results were seen at 3 years between recipients of livers from older (74.5%), same age (73.5%), and younger donors (77.9%).
Older donor age increased the risk for patient death by 14% and graft failure by 13%, in comparison with younger donor age in an adjusted analysis.
For every 10-year increase in patient age for younger donors, the risk of patient mortality increased by 24% and graft failure by 17%. For every 10 years the donor was younger than the recipient, however, there was a 27% decline in the risk of patient mortality and 30% decrease in the risk of graft failure. A small but significance beneficial interaction was found between young donor age and young recipient age on both outcomes.
In contrast, for every 10 years the donor was older than the recipient, the risk of patient mortality increased 9% and the risk of graft failure increased by 22%, and there was no significant interaction between recipient and donor age.
When compared against donor and recipient pairs who were both aged 18-40 years, the risk of patient mortality and graft failure increased significantly for all other combinations of donor and recipient pairs: recipients aged 18-40 years and donors aged 60 years or older (hazard ratios of 1.76 and 1.49, respectively), recipients aged 60 years older and donors aged 18-40 years (HRs of 1.67 and 1.83), and recipients and donors both aged 60 years or older (HRs of 2.12 and 1.87). These results were consistent at both 1 and 3 years.
The overall results "suggest that there is something intrinsic about being older or having an older graft that cannot be remediated by graft or patient age factors," Dr. Parikh said.
The results might be explained in livers from older donors by impaired liver regeneration and increased susceptibility to ischemic reperfusion injury, while older recipients might have decreased circulating stem cells and more competing risks that are not adequately risk adjusted in the available data in older recipients, he said.
One audience member noted that there is still a "huge survival advantage" in all patient populations that receive younger donor livers and that allocating organs based on age is a slippery slope.
Dr. Parikh and his colleagues had no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE AMERICAN ASSOCIATION FOR THE STUDY OF LIVER DISEASES
Major Finding: For every 10-year increase in patient age for younger donors, the risk of patient mortality increased by 24% and graft failure by 17%; for every 10 years the donor was younger than the recipient, there was a 27% decline in the risk of patient mortality and a 30% decrease in the risk of graft failure.
Data Source: This was a secondary analysis of the Organ Procurement and Transplantation Network national database from 2002 to 2011 for adult cadaveric liver transplantation.
Disclosures: Dr. Parikh and his colleagues had no relevant financial disclosures.
Acetaminophen Liver Failure Least Likely to Get Transplant
BOSTON – Acetaminophen toxicity is the most frequent cause of acute liver failure in the United States and its usually milder presentation, along with comorbid psychosocial issues, explain why fewer patients with the etiology undergo liver transplantation, compared with other causes, according to a 10-year retrospective study of the National Institute of Diabetes and Digestive and Kidney Diseases Acute Liver Failure database.
Dr. K. Rajender Reddy and his colleagues in the National Institutes of Health Acute Liver Failure Study Group set out to determine factors that could help clinicians to improve their selection of acute liver failure patients for liver transplantation listing and their decision to proceed with transplantation.
At the annual meeting of the American Association for the Study of Liver Diseases, Dr. Reddy reported that of 1,144 patients who had at least 1 year of follow-up in the database, 491 (43%) had an acetaminophen etiology for acute liver failure, but only 26% of all patients listed for transplantation had an acetaminophen etiology and just 14% of all transplants occurred in patients with acetaminophen toxicity as the underlying cause.
In comparison, patients with other etiologies had higher listing and transplantation rates, with the highest being for autoimmune hepatitis (62% of all patients listed and 50% of all transplanted).
Overall during 2000-2010, 697 patients were not listed and not transplanted (group A), 177 were listed and not transplanted (group B), and 270 were listed and transplanted (group C). After 2 years of follow-up, patient survival was highest in group C (82%), followed by group A (47%) and group B (41%). Survival at 21 days followed the same pattern (C, 89%; A, 58%; B, 45%). Patients in groups A and B with acetaminophen etiology had greater 21-day survival than did those without an acetaminophen etiology.
Patients in group A had significantly higher rates of positive toxicology screens than did groups B and C (43% vs. 35% and 19%, respectively) and history of illicit drug use (10% vs. 4% and 2%).
Comparisons between patients who died in group B and all patients in group C could not find significant risk factors to explain the higher mortality in group B, including longer wait list times and variations in survival and transplant rates by geographic location, although greater coma severity may have contributed to the higher mortality, said Dr. Reddy, professor of medicine in the division of gastroenterology at the Hospital of the University of Pennsylvania, Philadelphia.
Dr. Reddy said that he had no relevant financial disclosures. The study was funded by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases to the National Institutes of Health Acute Liver Failure Study Group.
BOSTON – Acetaminophen toxicity is the most frequent cause of acute liver failure in the United States and its usually milder presentation, along with comorbid psychosocial issues, explain why fewer patients with the etiology undergo liver transplantation, compared with other causes, according to a 10-year retrospective study of the National Institute of Diabetes and Digestive and Kidney Diseases Acute Liver Failure database.
Dr. K. Rajender Reddy and his colleagues in the National Institutes of Health Acute Liver Failure Study Group set out to determine factors that could help clinicians to improve their selection of acute liver failure patients for liver transplantation listing and their decision to proceed with transplantation.
At the annual meeting of the American Association for the Study of Liver Diseases, Dr. Reddy reported that of 1,144 patients who had at least 1 year of follow-up in the database, 491 (43%) had an acetaminophen etiology for acute liver failure, but only 26% of all patients listed for transplantation had an acetaminophen etiology and just 14% of all transplants occurred in patients with acetaminophen toxicity as the underlying cause.
In comparison, patients with other etiologies had higher listing and transplantation rates, with the highest being for autoimmune hepatitis (62% of all patients listed and 50% of all transplanted).
Overall during 2000-2010, 697 patients were not listed and not transplanted (group A), 177 were listed and not transplanted (group B), and 270 were listed and transplanted (group C). After 2 years of follow-up, patient survival was highest in group C (82%), followed by group A (47%) and group B (41%). Survival at 21 days followed the same pattern (C, 89%; A, 58%; B, 45%). Patients in groups A and B with acetaminophen etiology had greater 21-day survival than did those without an acetaminophen etiology.
Patients in group A had significantly higher rates of positive toxicology screens than did groups B and C (43% vs. 35% and 19%, respectively) and history of illicit drug use (10% vs. 4% and 2%).
Comparisons between patients who died in group B and all patients in group C could not find significant risk factors to explain the higher mortality in group B, including longer wait list times and variations in survival and transplant rates by geographic location, although greater coma severity may have contributed to the higher mortality, said Dr. Reddy, professor of medicine in the division of gastroenterology at the Hospital of the University of Pennsylvania, Philadelphia.
Dr. Reddy said that he had no relevant financial disclosures. The study was funded by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases to the National Institutes of Health Acute Liver Failure Study Group.
BOSTON – Acetaminophen toxicity is the most frequent cause of acute liver failure in the United States and its usually milder presentation, along with comorbid psychosocial issues, explain why fewer patients with the etiology undergo liver transplantation, compared with other causes, according to a 10-year retrospective study of the National Institute of Diabetes and Digestive and Kidney Diseases Acute Liver Failure database.
Dr. K. Rajender Reddy and his colleagues in the National Institutes of Health Acute Liver Failure Study Group set out to determine factors that could help clinicians to improve their selection of acute liver failure patients for liver transplantation listing and their decision to proceed with transplantation.
At the annual meeting of the American Association for the Study of Liver Diseases, Dr. Reddy reported that of 1,144 patients who had at least 1 year of follow-up in the database, 491 (43%) had an acetaminophen etiology for acute liver failure, but only 26% of all patients listed for transplantation had an acetaminophen etiology and just 14% of all transplants occurred in patients with acetaminophen toxicity as the underlying cause.
In comparison, patients with other etiologies had higher listing and transplantation rates, with the highest being for autoimmune hepatitis (62% of all patients listed and 50% of all transplanted).
Overall during 2000-2010, 697 patients were not listed and not transplanted (group A), 177 were listed and not transplanted (group B), and 270 were listed and transplanted (group C). After 2 years of follow-up, patient survival was highest in group C (82%), followed by group A (47%) and group B (41%). Survival at 21 days followed the same pattern (C, 89%; A, 58%; B, 45%). Patients in groups A and B with acetaminophen etiology had greater 21-day survival than did those without an acetaminophen etiology.
Patients in group A had significantly higher rates of positive toxicology screens than did groups B and C (43% vs. 35% and 19%, respectively) and history of illicit drug use (10% vs. 4% and 2%).
Comparisons between patients who died in group B and all patients in group C could not find significant risk factors to explain the higher mortality in group B, including longer wait list times and variations in survival and transplant rates by geographic location, although greater coma severity may have contributed to the higher mortality, said Dr. Reddy, professor of medicine in the division of gastroenterology at the Hospital of the University of Pennsylvania, Philadelphia.
Dr. Reddy said that he had no relevant financial disclosures. The study was funded by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases to the National Institutes of Health Acute Liver Failure Study Group.
AT THE ANNUAL MEETING OF THE AMERICAN ASSOCIATION FOR THE STUDY OF LIVER DISEASES
Major Finding: A total of 43% in the study had an acetaminophen etiology for acute liver failure, but only 26% of all patients listed for transplantation had an acetaminophen etiology and just 14% of all transplants occurred in patients with acetaminophen toxicity as the underlying cause.
Data Source: This was a 10-year retrospective study of the National Institute of Diabetes and Digestive and Kidney Diseases Acute Liver Failure database.
Disclosures: Dr. Reddy said he had no relevant financial disclosures. The study was funded by a grant from the National Institute of Diabetes and Digestive and Kidney Diseases to the National Institutes of Health Acute Liver Failure Study Group.
Fatty Liver Disease Cited for Rise in Hepatocellular Carcinoma
BOSTON – Nonalcoholic fatty liver disease without cirrhosis appears to be a significant contributor to the rise in the incidence of hepatocellular carcinoma in the past two decades, according to a study linking population-based data from the National Cancer Institute with Medicare enrollment and claim files during 1993-2007.
"Our data suggest a unique underlying pathophysiology for development of HCC [hepatocellular carcinoma] in noncirrhotic NAFLD [nonalcoholic fatty liver disease]," Dr. Rubayat Rahman said at the annual meeting of the American Association for the Study of Liver Diseases.
The finding may help to explain the rise in the incidence of the malignancy, which has had no clear explanation, added Dr. Rahman of the division of gastroenterology and hepatology at the University of Missouri, Columbia.
Of 17,895 HCC cases in the linked Surveillance, Epidemiology and End Results (SEER)-Medicare database, 2,863 (16%) had only NAFLD without any other risk factors or etiologies for HCC. The linked database covers 30% of the U.S. Medicare population. SEER itself contains data from 18 cancer registries covering 28% of the U.S. population, and 93% of the individuals in the database who are at least 65 years are matched to a Medicare enrollment file.
At 16%, NAFLD was third most common risk factor for HCC after infection (44%) and alcoholic diseases (19%) – 21% were other causes – but it was the second most common risk factor after infection in Asians and Pacific islanders, said Dr. Rahman.
Cirrhosis was not present in 36% of the NAFLD-related HCC cases; of those, 18% had only steatosis and not nonalcoholic steatohepatitis (NASH) or other adverse changes in pathology. Patients without cirrhosis more often had early-stage disease (stage I or II) than did those with cirrhosis (62% vs. 44%), as well as favorable grades (I or II, 76% vs. 56%).
Although the annual percentage change of NAFLD-related HCC with cirrhosis has increased since 1993, 1999 marked a point when the annual growth of NAFLD-related cases of HCC without cirrhosis outpaced those with cirrhosis. The average number of cases per year of NAFLD-related HCC without cirrhosis grew significantly from 51 in 1993-2000 to 88 in 2001-2007, compared with no change in cases with cirrhosis.
Three components of the metabolic syndrome – body-mass index greater than 30 kg/m2, type 2 diabetes mellitus, and dyslipidemia – occurred in significantly greater proportions of patients with NAFLD-related HCC without cirrhosis than in those with cirrhosis. The odds of developing HCC for each of those three components among noncirrhotic NAFLD patients was higher than for those with cirrhotic NAFLD, although the overall odds of developing HCC were higher among patients with cirrhotic NAFLD (odds ratio, 16.5) than in those with noncirrhotic NAFLD (OR, 3.1).
One audience member expressed concern about determining the presence of cirrhosis or NASH in the data without a systematic assessment of histopathology performed in a centralized way, but Dr. Rahman noted that the Medicare-matched SEER data have CPT procedural codes and ICD-9 diagnostic codes for diagnoses made through liver biopsy. Another attendee also suggested caution in calling NAFLD a risk factor for cancer alone because the people in the database have a higher rate of cancer, and the database does not include the peak of NASH at age 55 years.
None of the study investigators had relevant financial disclosures.
BOSTON – Nonalcoholic fatty liver disease without cirrhosis appears to be a significant contributor to the rise in the incidence of hepatocellular carcinoma in the past two decades, according to a study linking population-based data from the National Cancer Institute with Medicare enrollment and claim files during 1993-2007.
"Our data suggest a unique underlying pathophysiology for development of HCC [hepatocellular carcinoma] in noncirrhotic NAFLD [nonalcoholic fatty liver disease]," Dr. Rubayat Rahman said at the annual meeting of the American Association for the Study of Liver Diseases.
The finding may help to explain the rise in the incidence of the malignancy, which has had no clear explanation, added Dr. Rahman of the division of gastroenterology and hepatology at the University of Missouri, Columbia.
Of 17,895 HCC cases in the linked Surveillance, Epidemiology and End Results (SEER)-Medicare database, 2,863 (16%) had only NAFLD without any other risk factors or etiologies for HCC. The linked database covers 30% of the U.S. Medicare population. SEER itself contains data from 18 cancer registries covering 28% of the U.S. population, and 93% of the individuals in the database who are at least 65 years are matched to a Medicare enrollment file.
At 16%, NAFLD was third most common risk factor for HCC after infection (44%) and alcoholic diseases (19%) – 21% were other causes – but it was the second most common risk factor after infection in Asians and Pacific islanders, said Dr. Rahman.
Cirrhosis was not present in 36% of the NAFLD-related HCC cases; of those, 18% had only steatosis and not nonalcoholic steatohepatitis (NASH) or other adverse changes in pathology. Patients without cirrhosis more often had early-stage disease (stage I or II) than did those with cirrhosis (62% vs. 44%), as well as favorable grades (I or II, 76% vs. 56%).
Although the annual percentage change of NAFLD-related HCC with cirrhosis has increased since 1993, 1999 marked a point when the annual growth of NAFLD-related cases of HCC without cirrhosis outpaced those with cirrhosis. The average number of cases per year of NAFLD-related HCC without cirrhosis grew significantly from 51 in 1993-2000 to 88 in 2001-2007, compared with no change in cases with cirrhosis.
Three components of the metabolic syndrome – body-mass index greater than 30 kg/m2, type 2 diabetes mellitus, and dyslipidemia – occurred in significantly greater proportions of patients with NAFLD-related HCC without cirrhosis than in those with cirrhosis. The odds of developing HCC for each of those three components among noncirrhotic NAFLD patients was higher than for those with cirrhotic NAFLD, although the overall odds of developing HCC were higher among patients with cirrhotic NAFLD (odds ratio, 16.5) than in those with noncirrhotic NAFLD (OR, 3.1).
One audience member expressed concern about determining the presence of cirrhosis or NASH in the data without a systematic assessment of histopathology performed in a centralized way, but Dr. Rahman noted that the Medicare-matched SEER data have CPT procedural codes and ICD-9 diagnostic codes for diagnoses made through liver biopsy. Another attendee also suggested caution in calling NAFLD a risk factor for cancer alone because the people in the database have a higher rate of cancer, and the database does not include the peak of NASH at age 55 years.
None of the study investigators had relevant financial disclosures.
BOSTON – Nonalcoholic fatty liver disease without cirrhosis appears to be a significant contributor to the rise in the incidence of hepatocellular carcinoma in the past two decades, according to a study linking population-based data from the National Cancer Institute with Medicare enrollment and claim files during 1993-2007.
"Our data suggest a unique underlying pathophysiology for development of HCC [hepatocellular carcinoma] in noncirrhotic NAFLD [nonalcoholic fatty liver disease]," Dr. Rubayat Rahman said at the annual meeting of the American Association for the Study of Liver Diseases.
The finding may help to explain the rise in the incidence of the malignancy, which has had no clear explanation, added Dr. Rahman of the division of gastroenterology and hepatology at the University of Missouri, Columbia.
Of 17,895 HCC cases in the linked Surveillance, Epidemiology and End Results (SEER)-Medicare database, 2,863 (16%) had only NAFLD without any other risk factors or etiologies for HCC. The linked database covers 30% of the U.S. Medicare population. SEER itself contains data from 18 cancer registries covering 28% of the U.S. population, and 93% of the individuals in the database who are at least 65 years are matched to a Medicare enrollment file.
At 16%, NAFLD was third most common risk factor for HCC after infection (44%) and alcoholic diseases (19%) – 21% were other causes – but it was the second most common risk factor after infection in Asians and Pacific islanders, said Dr. Rahman.
Cirrhosis was not present in 36% of the NAFLD-related HCC cases; of those, 18% had only steatosis and not nonalcoholic steatohepatitis (NASH) or other adverse changes in pathology. Patients without cirrhosis more often had early-stage disease (stage I or II) than did those with cirrhosis (62% vs. 44%), as well as favorable grades (I or II, 76% vs. 56%).
Although the annual percentage change of NAFLD-related HCC with cirrhosis has increased since 1993, 1999 marked a point when the annual growth of NAFLD-related cases of HCC without cirrhosis outpaced those with cirrhosis. The average number of cases per year of NAFLD-related HCC without cirrhosis grew significantly from 51 in 1993-2000 to 88 in 2001-2007, compared with no change in cases with cirrhosis.
Three components of the metabolic syndrome – body-mass index greater than 30 kg/m2, type 2 diabetes mellitus, and dyslipidemia – occurred in significantly greater proportions of patients with NAFLD-related HCC without cirrhosis than in those with cirrhosis. The odds of developing HCC for each of those three components among noncirrhotic NAFLD patients was higher than for those with cirrhotic NAFLD, although the overall odds of developing HCC were higher among patients with cirrhotic NAFLD (odds ratio, 16.5) than in those with noncirrhotic NAFLD (OR, 3.1).
One audience member expressed concern about determining the presence of cirrhosis or NASH in the data without a systematic assessment of histopathology performed in a centralized way, but Dr. Rahman noted that the Medicare-matched SEER data have CPT procedural codes and ICD-9 diagnostic codes for diagnoses made through liver biopsy. Another attendee also suggested caution in calling NAFLD a risk factor for cancer alone because the people in the database have a higher rate of cancer, and the database does not include the peak of NASH at age 55 years.
None of the study investigators had relevant financial disclosures.
AT THE ANNUAL MEETING OF THE AMERICAN ASSOCIATION FOR THE STUDY OF LIVER DISEASES
Major Finding: The average number of cases per year of NAFLD-related hepatocellular carcinoma without cirrhosis grew significantly from 51 in 1993-2000 to 88 in 2001-2007, compared with no change in cases with cirrhosis.
Data Source: This population-based study used data from the SEER-Medicare database from 1993 to 2007.
Disclosures: None of the study investigators had relevant financial disclosures.
Acetaminophen: Effect on Drinkers' ALT Levels Appears Safe
SAN DIEGO – Giving acetaminophen to patients who reported consuming ethanol did not adversely affect markers of liver damage in a meta-analysis of randomized, controlled trials.
"One of the questions we often get asked is the role of acetaminophen in patients with liver disease," according to Dr. Kennon J. Heard, who is an emergency medicine physician at the University of Colorado and director of the Medical Toxicology Fellowship at the Rocky Mountain Poison and Drug Center, Denver.
The findings of the meta-analysis suggest that "acetaminophen is safe in alcoholics," Dr. Heard said at the annual meeting of the Society of Hospital Medicine.
The meta-analysis included five trials involving 901 subjects (including patients who reported drinking ethanol) who were randomized to receive acetaminophen or placebo.
Dr. Heard and his associates looked at daily ALT measurements out to a mean of 4 days, a time period for which most of the studies had data.
They also looked for any evidence of liver injury or dysfunction, hepatotoxicity, or death.
The alanine aminotransferase (ALT) levels changed by a mean of 0.04 IU/L after starting acetaminophen or placebo, "less than a tenth of a point in ALT," reported Dr. Heard.
"Essentially, in this group of patients who consume alcohol, if you give them acetaminophen for 4 days, you don’t see any change in their ALT," according to Dr. Heard.
The study is to be published in the journal Pharmacotherapy.
When acetaminophen consumption continued beyond 4 days, ALT levels increased in most patients who consumed alcohol but also increased in 60% of nondrinkers.
"The changes in the alcoholics look exactly like the changes in the nonalcoholics," he said.
The median increase in ALT was between 10-20 IU/L.
Among patients who drink alcohol, the highest ALT level in the acetaminophen group was 312 IU/L, "which is pretty impressive until you see that in the placebo group, somebody went up 288" IU/L, he said.
The biggest increase in ALT was in a healthy nondrinking patient on acetaminophen, whose ALT increased by 638 IU/L.
Most importantly, none of the 551 people who received acetaminophen in those trials developed an increase in International Normalized Ratio, bilirubin level, or symptomatic liver injury, Dr. Heard and his associates found.
Dr. Heard said that he and his associates are now in the process of finishing a separate study that appears to confirm that these are asymptomatic, self-limiting elevations in ALT that will go away even if people stay on acetaminophen.
Such information is valuable, he said. "It is worth knowing that if you have someone who has an ALT elevation while taking acetaminophen, it may be the cause, and it is reasonable to stop the acetaminophen and see if their ALT elevations go away rather than do an extensive work-up for hepatitis," Dr. Heard said.
SAN DIEGO – Giving acetaminophen to patients who reported consuming ethanol did not adversely affect markers of liver damage in a meta-analysis of randomized, controlled trials.
"One of the questions we often get asked is the role of acetaminophen in patients with liver disease," according to Dr. Kennon J. Heard, who is an emergency medicine physician at the University of Colorado and director of the Medical Toxicology Fellowship at the Rocky Mountain Poison and Drug Center, Denver.
The findings of the meta-analysis suggest that "acetaminophen is safe in alcoholics," Dr. Heard said at the annual meeting of the Society of Hospital Medicine.
The meta-analysis included five trials involving 901 subjects (including patients who reported drinking ethanol) who were randomized to receive acetaminophen or placebo.
Dr. Heard and his associates looked at daily ALT measurements out to a mean of 4 days, a time period for which most of the studies had data.
They also looked for any evidence of liver injury or dysfunction, hepatotoxicity, or death.
The alanine aminotransferase (ALT) levels changed by a mean of 0.04 IU/L after starting acetaminophen or placebo, "less than a tenth of a point in ALT," reported Dr. Heard.
"Essentially, in this group of patients who consume alcohol, if you give them acetaminophen for 4 days, you don’t see any change in their ALT," according to Dr. Heard.
The study is to be published in the journal Pharmacotherapy.
When acetaminophen consumption continued beyond 4 days, ALT levels increased in most patients who consumed alcohol but also increased in 60% of nondrinkers.
"The changes in the alcoholics look exactly like the changes in the nonalcoholics," he said.
The median increase in ALT was between 10-20 IU/L.
Among patients who drink alcohol, the highest ALT level in the acetaminophen group was 312 IU/L, "which is pretty impressive until you see that in the placebo group, somebody went up 288" IU/L, he said.
The biggest increase in ALT was in a healthy nondrinking patient on acetaminophen, whose ALT increased by 638 IU/L.
Most importantly, none of the 551 people who received acetaminophen in those trials developed an increase in International Normalized Ratio, bilirubin level, or symptomatic liver injury, Dr. Heard and his associates found.
Dr. Heard said that he and his associates are now in the process of finishing a separate study that appears to confirm that these are asymptomatic, self-limiting elevations in ALT that will go away even if people stay on acetaminophen.
Such information is valuable, he said. "It is worth knowing that if you have someone who has an ALT elevation while taking acetaminophen, it may be the cause, and it is reasonable to stop the acetaminophen and see if their ALT elevations go away rather than do an extensive work-up for hepatitis," Dr. Heard said.
SAN DIEGO – Giving acetaminophen to patients who reported consuming ethanol did not adversely affect markers of liver damage in a meta-analysis of randomized, controlled trials.
"One of the questions we often get asked is the role of acetaminophen in patients with liver disease," according to Dr. Kennon J. Heard, who is an emergency medicine physician at the University of Colorado and director of the Medical Toxicology Fellowship at the Rocky Mountain Poison and Drug Center, Denver.
The findings of the meta-analysis suggest that "acetaminophen is safe in alcoholics," Dr. Heard said at the annual meeting of the Society of Hospital Medicine.
The meta-analysis included five trials involving 901 subjects (including patients who reported drinking ethanol) who were randomized to receive acetaminophen or placebo.
Dr. Heard and his associates looked at daily ALT measurements out to a mean of 4 days, a time period for which most of the studies had data.
They also looked for any evidence of liver injury or dysfunction, hepatotoxicity, or death.
The alanine aminotransferase (ALT) levels changed by a mean of 0.04 IU/L after starting acetaminophen or placebo, "less than a tenth of a point in ALT," reported Dr. Heard.
"Essentially, in this group of patients who consume alcohol, if you give them acetaminophen for 4 days, you don’t see any change in their ALT," according to Dr. Heard.
The study is to be published in the journal Pharmacotherapy.
When acetaminophen consumption continued beyond 4 days, ALT levels increased in most patients who consumed alcohol but also increased in 60% of nondrinkers.
"The changes in the alcoholics look exactly like the changes in the nonalcoholics," he said.
The median increase in ALT was between 10-20 IU/L.
Among patients who drink alcohol, the highest ALT level in the acetaminophen group was 312 IU/L, "which is pretty impressive until you see that in the placebo group, somebody went up 288" IU/L, he said.
The biggest increase in ALT was in a healthy nondrinking patient on acetaminophen, whose ALT increased by 638 IU/L.
Most importantly, none of the 551 people who received acetaminophen in those trials developed an increase in International Normalized Ratio, bilirubin level, or symptomatic liver injury, Dr. Heard and his associates found.
Dr. Heard said that he and his associates are now in the process of finishing a separate study that appears to confirm that these are asymptomatic, self-limiting elevations in ALT that will go away even if people stay on acetaminophen.
Such information is valuable, he said. "It is worth knowing that if you have someone who has an ALT elevation while taking acetaminophen, it may be the cause, and it is reasonable to stop the acetaminophen and see if their ALT elevations go away rather than do an extensive work-up for hepatitis," Dr. Heard said.
Major Finding: ALT levels changed by a median of 0.04 IU/L in alcohol drinkers and nondrinkers after taking acetaminophen for 4 days, with no significant difference between subjects on acetaminophen or placebo.
Data Source: The meta-analysis of five randomized, controlled trials included 901 subjects.
Disclosures: Dr. Heard has been a consultant or received research grants from Cadence Pharmaceuticals, McNeil Consumer Healthcare, and Cumberland Pharmaceuticals.
Analysis Details the GI Disease Burden in U.S.
Clostridium difficile contributes mightily to the overall burden of gastrointestinal disease in the United States and was associated with a 237% increase in hospitalizations in the last decade.
Researchers who examined the latest data on the nationwide toll of GI and liver disease also found a 314% rise in hospitalizations related to morbid obesity and a continuing national health burden exacted by reflux symptoms, Barrett’s esophagus, and colorectal cancer.
Video from the American Gastroenterological Association (http://www.youtube.com/amergastroassn)
"We compiled the most recently available statistics on GI symptoms, quality of life, outpatient diagnoses, hospitalizations, costs, mortality, and endoscopic utilization from a variety of publicly and privately held databases," Dr. Anne F. Peery of the University of North Carolina, Chapel Hill, and her colleagues reported in the November issue of Gastroenterology (doi:10.1053/j.gastro.2012.08.002).
"Payers, policy makers, clinicians, and others interested in resource utilization may use these statistics to better understand evolving disease trends, and the best way to meet the challenge of these diseases."
The findings are based on data for 2009, the most recent year for which complete information was available, from the National Ambulatory Medical Care Survey, sponsored by the U.S. Centers for Disease Control and Prevention; the United States National Health and Wellness Survey, sponsored by the private company Kantar Health; the Nationwide Inpatient Sample, sponsored by the Agency for Healthcare Research and Quality; the Surveillance, Epidemiology, and End Results database of the National Cancer Institute; the National Vital Statistics System, sponsored by the National Center for Health Statistics and the CDC; and the Thomson Reuters MarketScan’s databases of commercial, Medicare, and Medicaid records.
Among the findings:
• C. difficile hospitalizations have increased 237% since 2000 and were associated with 4% in-hospital mortality. Now the ninth leading GI cause of mortality, with an absolute increase of 230% in the number of C. difficile–related deaths since 2002, the infection also markedly impairs quality of life and the capacity for work and other activities.
• Hospitalizations related to obesity remained relatively stable since 2000, but those associated with morbid obesity rose by 314%, and many were likely caused by the marked increase in bariatric surgery.
• Gastroesophageal reflux remains the most common GI-associated diagnosis in primary care, accounting for 9 million outpatient visits in 2009, and the most common GI-associated discharge diagnosis, with 4.4 million such diagnoses in 2009. Obesity was associated with 1.7 million discharge diagnoses and constipation with 1 million.
• Barrett’s esophagus accounted for almost half a million outpatient visits in 2009, when an estimated 3.3 million Americans had this diagnosis. Given that endoscopic surveillance is recommended every 3-5 years, Barrett’s contributes substantially to resource utilization.
• Colorectal cancer, with an estimated 147,000 patients diagnosed in 2008, accounts for more than half of all GI cancer diagnoses and continues to be the primary cause of GI-associated mortality. Pancreatic and hepatobiliary cancers are the next most frequently diagnosed GI cancers.
• Of the approximately 2.5 million deaths in the United States in 2009, 10% were attributed to an underlying GI cause. Chronic liver disease and cirrhosis are the 12th leading causes of death in the country.
• The total outpatient cost for GI endoscopy in 2009 was estimated to be $32.4 billion, which is higher than previously published estimates. An estimated 6.9 million upper endoscopies, 11.5 million lower endoscopies, and 228,000 biliary endoscopies were performed in the United States in 2009.
• Chronic liver disease and viral hepatitis were associated with 6% mortality and cost an estimated $1.8 billion per year in inpatient cost.
• Hospitalizations for nonalcoholic fatty liver disease increased 97% since 2000.
This study was supported in part by the National Institutes of Health. No financial conflicts of interest were reported.
Digestive (GI and liver) diseases constitute a substantial and growing burden in the United States. As detailed in the report by Dr. Peery and colleagues, there were over 46 million outpatient encounters associated with the top 20 digestive disease diagnoses in 2009, with approximately 10% of deaths nationwide with an underlying digestive disease cause. The observed increased prevalence of hospitalizations for many diagnoses (e.g., a 14% increase with principal discharge diagnosis of chronic liver disease with viral hepatitis) and procedures (e.g., a 17% increase in lower GI endoscopies among commercially insured patients) between 2000 and 2009 is expected given population growth and aging. In addition, dramatic increases in hospitalizations associated with C. difficile and morbid obesity were also noted.
The report provides crucial information for diverse constituencies, including healthcare planners, clinicians and researchers. However, as acknowledged by the authors, there are some important caveats with respect to coverage or quality for some data sources to bear in mind when interpreting these results. This is particularly relevant for non-alcoholic fatty liver disease, which is likely underestimated because of well-known problems in diagnostic code specificity and use.
Several factors suggest that the prevalence and costs of digestive diseases will increase substantially during the next decade. These include: an aging population with the number of people aged 65 years or older projected to be greater than 54 million by 2020;the estimated tens of millions of individuals with newly available healthcare coverage as of 2014 as part of the Affordable Care Act; continued increases in obesity rates; and the recent CDC recommendation that all baby-boomers be screened for hepatitis C. This report will facilitate timely planning and also serve as benchmark to help measure the impact of these forces on the scope and burden of digestive diseases and their clinical management.
DONNA L. WHITE, PH.D., MPH, is an investigator in the Clinical Epidemiology and Outcomes Program in the Houston VA Health Services Research and Development Center of Excellence at the Michael E. DeBakey VA Medical Center, Houston. She also is an assistant professor in the department of medicine at Baylor College of Medicine, Houston.
Digestive (GI and liver) diseases constitute a substantial and growing burden in the United States. As detailed in the report by Dr. Peery and colleagues, there were over 46 million outpatient encounters associated with the top 20 digestive disease diagnoses in 2009, with approximately 10% of deaths nationwide with an underlying digestive disease cause. The observed increased prevalence of hospitalizations for many diagnoses (e.g., a 14% increase with principal discharge diagnosis of chronic liver disease with viral hepatitis) and procedures (e.g., a 17% increase in lower GI endoscopies among commercially insured patients) between 2000 and 2009 is expected given population growth and aging. In addition, dramatic increases in hospitalizations associated with C. difficile and morbid obesity were also noted.
The report provides crucial information for diverse constituencies, including healthcare planners, clinicians and researchers. However, as acknowledged by the authors, there are some important caveats with respect to coverage or quality for some data sources to bear in mind when interpreting these results. This is particularly relevant for non-alcoholic fatty liver disease, which is likely underestimated because of well-known problems in diagnostic code specificity and use.
Several factors suggest that the prevalence and costs of digestive diseases will increase substantially during the next decade. These include: an aging population with the number of people aged 65 years or older projected to be greater than 54 million by 2020;the estimated tens of millions of individuals with newly available healthcare coverage as of 2014 as part of the Affordable Care Act; continued increases in obesity rates; and the recent CDC recommendation that all baby-boomers be screened for hepatitis C. This report will facilitate timely planning and also serve as benchmark to help measure the impact of these forces on the scope and burden of digestive diseases and their clinical management.
DONNA L. WHITE, PH.D., MPH, is an investigator in the Clinical Epidemiology and Outcomes Program in the Houston VA Health Services Research and Development Center of Excellence at the Michael E. DeBakey VA Medical Center, Houston. She also is an assistant professor in the department of medicine at Baylor College of Medicine, Houston.
Digestive (GI and liver) diseases constitute a substantial and growing burden in the United States. As detailed in the report by Dr. Peery and colleagues, there were over 46 million outpatient encounters associated with the top 20 digestive disease diagnoses in 2009, with approximately 10% of deaths nationwide with an underlying digestive disease cause. The observed increased prevalence of hospitalizations for many diagnoses (e.g., a 14% increase with principal discharge diagnosis of chronic liver disease with viral hepatitis) and procedures (e.g., a 17% increase in lower GI endoscopies among commercially insured patients) between 2000 and 2009 is expected given population growth and aging. In addition, dramatic increases in hospitalizations associated with C. difficile and morbid obesity were also noted.
The report provides crucial information for diverse constituencies, including healthcare planners, clinicians and researchers. However, as acknowledged by the authors, there are some important caveats with respect to coverage or quality for some data sources to bear in mind when interpreting these results. This is particularly relevant for non-alcoholic fatty liver disease, which is likely underestimated because of well-known problems in diagnostic code specificity and use.
Several factors suggest that the prevalence and costs of digestive diseases will increase substantially during the next decade. These include: an aging population with the number of people aged 65 years or older projected to be greater than 54 million by 2020;the estimated tens of millions of individuals with newly available healthcare coverage as of 2014 as part of the Affordable Care Act; continued increases in obesity rates; and the recent CDC recommendation that all baby-boomers be screened for hepatitis C. This report will facilitate timely planning and also serve as benchmark to help measure the impact of these forces on the scope and burden of digestive diseases and their clinical management.
DONNA L. WHITE, PH.D., MPH, is an investigator in the Clinical Epidemiology and Outcomes Program in the Houston VA Health Services Research and Development Center of Excellence at the Michael E. DeBakey VA Medical Center, Houston. She also is an assistant professor in the department of medicine at Baylor College of Medicine, Houston.
Clostridium difficile contributes mightily to the overall burden of gastrointestinal disease in the United States and was associated with a 237% increase in hospitalizations in the last decade.
Researchers who examined the latest data on the nationwide toll of GI and liver disease also found a 314% rise in hospitalizations related to morbid obesity and a continuing national health burden exacted by reflux symptoms, Barrett’s esophagus, and colorectal cancer.
Video from the American Gastroenterological Association (http://www.youtube.com/amergastroassn)
"We compiled the most recently available statistics on GI symptoms, quality of life, outpatient diagnoses, hospitalizations, costs, mortality, and endoscopic utilization from a variety of publicly and privately held databases," Dr. Anne F. Peery of the University of North Carolina, Chapel Hill, and her colleagues reported in the November issue of Gastroenterology (doi:10.1053/j.gastro.2012.08.002).
"Payers, policy makers, clinicians, and others interested in resource utilization may use these statistics to better understand evolving disease trends, and the best way to meet the challenge of these diseases."
The findings are based on data for 2009, the most recent year for which complete information was available, from the National Ambulatory Medical Care Survey, sponsored by the U.S. Centers for Disease Control and Prevention; the United States National Health and Wellness Survey, sponsored by the private company Kantar Health; the Nationwide Inpatient Sample, sponsored by the Agency for Healthcare Research and Quality; the Surveillance, Epidemiology, and End Results database of the National Cancer Institute; the National Vital Statistics System, sponsored by the National Center for Health Statistics and the CDC; and the Thomson Reuters MarketScan’s databases of commercial, Medicare, and Medicaid records.
Among the findings:
• C. difficile hospitalizations have increased 237% since 2000 and were associated with 4% in-hospital mortality. Now the ninth leading GI cause of mortality, with an absolute increase of 230% in the number of C. difficile–related deaths since 2002, the infection also markedly impairs quality of life and the capacity for work and other activities.
• Hospitalizations related to obesity remained relatively stable since 2000, but those associated with morbid obesity rose by 314%, and many were likely caused by the marked increase in bariatric surgery.
• Gastroesophageal reflux remains the most common GI-associated diagnosis in primary care, accounting for 9 million outpatient visits in 2009, and the most common GI-associated discharge diagnosis, with 4.4 million such diagnoses in 2009. Obesity was associated with 1.7 million discharge diagnoses and constipation with 1 million.
• Barrett’s esophagus accounted for almost half a million outpatient visits in 2009, when an estimated 3.3 million Americans had this diagnosis. Given that endoscopic surveillance is recommended every 3-5 years, Barrett’s contributes substantially to resource utilization.
• Colorectal cancer, with an estimated 147,000 patients diagnosed in 2008, accounts for more than half of all GI cancer diagnoses and continues to be the primary cause of GI-associated mortality. Pancreatic and hepatobiliary cancers are the next most frequently diagnosed GI cancers.
• Of the approximately 2.5 million deaths in the United States in 2009, 10% were attributed to an underlying GI cause. Chronic liver disease and cirrhosis are the 12th leading causes of death in the country.
• The total outpatient cost for GI endoscopy in 2009 was estimated to be $32.4 billion, which is higher than previously published estimates. An estimated 6.9 million upper endoscopies, 11.5 million lower endoscopies, and 228,000 biliary endoscopies were performed in the United States in 2009.
• Chronic liver disease and viral hepatitis were associated with 6% mortality and cost an estimated $1.8 billion per year in inpatient cost.
• Hospitalizations for nonalcoholic fatty liver disease increased 97% since 2000.
This study was supported in part by the National Institutes of Health. No financial conflicts of interest were reported.
Clostridium difficile contributes mightily to the overall burden of gastrointestinal disease in the United States and was associated with a 237% increase in hospitalizations in the last decade.
Researchers who examined the latest data on the nationwide toll of GI and liver disease also found a 314% rise in hospitalizations related to morbid obesity and a continuing national health burden exacted by reflux symptoms, Barrett’s esophagus, and colorectal cancer.
Video from the American Gastroenterological Association (http://www.youtube.com/amergastroassn)
"We compiled the most recently available statistics on GI symptoms, quality of life, outpatient diagnoses, hospitalizations, costs, mortality, and endoscopic utilization from a variety of publicly and privately held databases," Dr. Anne F. Peery of the University of North Carolina, Chapel Hill, and her colleagues reported in the November issue of Gastroenterology (doi:10.1053/j.gastro.2012.08.002).
"Payers, policy makers, clinicians, and others interested in resource utilization may use these statistics to better understand evolving disease trends, and the best way to meet the challenge of these diseases."
The findings are based on data for 2009, the most recent year for which complete information was available, from the National Ambulatory Medical Care Survey, sponsored by the U.S. Centers for Disease Control and Prevention; the United States National Health and Wellness Survey, sponsored by the private company Kantar Health; the Nationwide Inpatient Sample, sponsored by the Agency for Healthcare Research and Quality; the Surveillance, Epidemiology, and End Results database of the National Cancer Institute; the National Vital Statistics System, sponsored by the National Center for Health Statistics and the CDC; and the Thomson Reuters MarketScan’s databases of commercial, Medicare, and Medicaid records.
Among the findings:
• C. difficile hospitalizations have increased 237% since 2000 and were associated with 4% in-hospital mortality. Now the ninth leading GI cause of mortality, with an absolute increase of 230% in the number of C. difficile–related deaths since 2002, the infection also markedly impairs quality of life and the capacity for work and other activities.
• Hospitalizations related to obesity remained relatively stable since 2000, but those associated with morbid obesity rose by 314%, and many were likely caused by the marked increase in bariatric surgery.
• Gastroesophageal reflux remains the most common GI-associated diagnosis in primary care, accounting for 9 million outpatient visits in 2009, and the most common GI-associated discharge diagnosis, with 4.4 million such diagnoses in 2009. Obesity was associated with 1.7 million discharge diagnoses and constipation with 1 million.
• Barrett’s esophagus accounted for almost half a million outpatient visits in 2009, when an estimated 3.3 million Americans had this diagnosis. Given that endoscopic surveillance is recommended every 3-5 years, Barrett’s contributes substantially to resource utilization.
• Colorectal cancer, with an estimated 147,000 patients diagnosed in 2008, accounts for more than half of all GI cancer diagnoses and continues to be the primary cause of GI-associated mortality. Pancreatic and hepatobiliary cancers are the next most frequently diagnosed GI cancers.
• Of the approximately 2.5 million deaths in the United States in 2009, 10% were attributed to an underlying GI cause. Chronic liver disease and cirrhosis are the 12th leading causes of death in the country.
• The total outpatient cost for GI endoscopy in 2009 was estimated to be $32.4 billion, which is higher than previously published estimates. An estimated 6.9 million upper endoscopies, 11.5 million lower endoscopies, and 228,000 biliary endoscopies were performed in the United States in 2009.
• Chronic liver disease and viral hepatitis were associated with 6% mortality and cost an estimated $1.8 billion per year in inpatient cost.
• Hospitalizations for nonalcoholic fatty liver disease increased 97% since 2000.
This study was supported in part by the National Institutes of Health. No financial conflicts of interest were reported.
FROM GASTROENTEROLOGY
Liver Candidates Decline Many Organ Offers
Eighty-four percent of candidates on the wait-list for liver transplant who either died or were removed from the list before they were able to undergo transplantation declined at least one offer of a donor liver, Dr. Jennifer Cindy Lai of the University of California, San Francisco, and her colleagues reported in the November issue of Gastroenterology.
Even more surprising, most of these candidates declined "not just one or two but a median of six liver offers during their time on the wait-list."
The "declined" donor organs were then successfully transplanted into lower-priority recipients.
These findings suggest that mortality among wait-listed patients "is not simply a result of not having the opportunity for transplantation, as many of us assume. Rather, wait-list mortality appears to result from opportunities for transplantation that were declined," Dr. Lai and her associates wrote.
The reasons that so many viable donor livers were initially declined are not yet clear. General, somewhat vague reasons were listed but not fully explained in the records the researchers analyzed for this study, which they obtained from the United Network for Organ Sharing/Organ Procurement Transplantation Network database.
The investigators assessed organ offers to 33,389 liver transplant candidates aged 18 years and older who were wait-listed across the United States between 2005 and 2010.
The reasons that proffered organs were declined, as listed in the medical records, fit into six broad categories: unfavorable donor age or quality of organ; unfavorable donor organ size/weight; other unfavorable donor factors, such as ABO blood transfusion incompatibility, "social history," "positive serologic tests," or "organ anatomical damage or defect"; unreadiness of the recipient, usually because he or she was ill, unavailable, refused the organ, or required multiple organ transplants at the same time; problems with the transplant program itself, such as a "heavy workload" or unavailability of a surgeon or operating room at the recipient’s medical center, failure to respond to the offer in a timely way, or excessive distance to ship the organ.
A total of 20% of the study population (6,737 patients) died or were removed from the wait-list because they became too sick before they could undergo transplantation. A total of 5,680 (84%) of those patients had been offered one or more donor livers before they died or were taken off the list.
Offers of donor livers were declined most often (68%) because of "unfavorable donor age or quality of organ," whereas 9% were declined because of unfavorable organ size, 15% because of "other donor factors," 4% because the recipient wasn’t ready, and 4% because of transplant program or miscellaneous other factors.
However, the dominant use of the "donor quality or age" refusal code in the database almost certainly "does not accurately or fully capture the true refusal reason," Dr. Lai and her associates said.
Even livers judged to be of high quality according to standard criteria were declined because of supposed "unfavorable donor age or quality of organ." But the investigators found no difference in the risk of graft failure between such high-quality livers that were declined and other high-quality livers that were accepted on the first offer.
Other reasons must be playing an important role in this high rate refusal, but "the nuances of these refusals cannot be determined" without more individualized data, they said.
Dr. Lai and her colleagues suggested that to cut down on refusals of apparently viable organs, the transplant community should "reduce the stigma associated with non–ideal livers, and set realistic expectations for wait-listed candidates" so that they’re less likely to pass up a suitable donation while assuming that a better offer will come along.
Patients also should be educated about the unpredictability of death or of sudden worsening of liver disease while on the wait-list. They should be advised that there is a survival benefit associated with the transplantation of any graft, compared with continuing on the wait-list.
In addition, the current regulatory environment focuses on transplant centers’ outcomes, which may influence some centers to discourage the acceptance of less than optimal donor organs. "This may be especially relevant for low-volume transplant centers, for whom even a small number of poor outcomes ... may make a relatively large difference in the centers’ perceived performance," the researchers wrote.
Finally, wait-list candidates should be encouraged to complete their transplant work-ups as expeditiously as possible to avoid having to refuse a donor offer simply because they have not yet undergone the necessary cardiac testing or cancer screening.
This study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases and the University of California, San Francisco. No financial conflicts of interest were reported.
Eighty-four percent of candidates on the wait-list for liver transplant who either died or were removed from the list before they were able to undergo transplantation declined at least one offer of a donor liver, Dr. Jennifer Cindy Lai of the University of California, San Francisco, and her colleagues reported in the November issue of Gastroenterology.
Even more surprising, most of these candidates declined "not just one or two but a median of six liver offers during their time on the wait-list."
The "declined" donor organs were then successfully transplanted into lower-priority recipients.
These findings suggest that mortality among wait-listed patients "is not simply a result of not having the opportunity for transplantation, as many of us assume. Rather, wait-list mortality appears to result from opportunities for transplantation that were declined," Dr. Lai and her associates wrote.
The reasons that so many viable donor livers were initially declined are not yet clear. General, somewhat vague reasons were listed but not fully explained in the records the researchers analyzed for this study, which they obtained from the United Network for Organ Sharing/Organ Procurement Transplantation Network database.
The investigators assessed organ offers to 33,389 liver transplant candidates aged 18 years and older who were wait-listed across the United States between 2005 and 2010.
The reasons that proffered organs were declined, as listed in the medical records, fit into six broad categories: unfavorable donor age or quality of organ; unfavorable donor organ size/weight; other unfavorable donor factors, such as ABO blood transfusion incompatibility, "social history," "positive serologic tests," or "organ anatomical damage or defect"; unreadiness of the recipient, usually because he or she was ill, unavailable, refused the organ, or required multiple organ transplants at the same time; problems with the transplant program itself, such as a "heavy workload" or unavailability of a surgeon or operating room at the recipient’s medical center, failure to respond to the offer in a timely way, or excessive distance to ship the organ.
A total of 20% of the study population (6,737 patients) died or were removed from the wait-list because they became too sick before they could undergo transplantation. A total of 5,680 (84%) of those patients had been offered one or more donor livers before they died or were taken off the list.
Offers of donor livers were declined most often (68%) because of "unfavorable donor age or quality of organ," whereas 9% were declined because of unfavorable organ size, 15% because of "other donor factors," 4% because the recipient wasn’t ready, and 4% because of transplant program or miscellaneous other factors.
However, the dominant use of the "donor quality or age" refusal code in the database almost certainly "does not accurately or fully capture the true refusal reason," Dr. Lai and her associates said.
Even livers judged to be of high quality according to standard criteria were declined because of supposed "unfavorable donor age or quality of organ." But the investigators found no difference in the risk of graft failure between such high-quality livers that were declined and other high-quality livers that were accepted on the first offer.
Other reasons must be playing an important role in this high rate refusal, but "the nuances of these refusals cannot be determined" without more individualized data, they said.
Dr. Lai and her colleagues suggested that to cut down on refusals of apparently viable organs, the transplant community should "reduce the stigma associated with non–ideal livers, and set realistic expectations for wait-listed candidates" so that they’re less likely to pass up a suitable donation while assuming that a better offer will come along.
Patients also should be educated about the unpredictability of death or of sudden worsening of liver disease while on the wait-list. They should be advised that there is a survival benefit associated with the transplantation of any graft, compared with continuing on the wait-list.
In addition, the current regulatory environment focuses on transplant centers’ outcomes, which may influence some centers to discourage the acceptance of less than optimal donor organs. "This may be especially relevant for low-volume transplant centers, for whom even a small number of poor outcomes ... may make a relatively large difference in the centers’ perceived performance," the researchers wrote.
Finally, wait-list candidates should be encouraged to complete their transplant work-ups as expeditiously as possible to avoid having to refuse a donor offer simply because they have not yet undergone the necessary cardiac testing or cancer screening.
This study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases and the University of California, San Francisco. No financial conflicts of interest were reported.
Eighty-four percent of candidates on the wait-list for liver transplant who either died or were removed from the list before they were able to undergo transplantation declined at least one offer of a donor liver, Dr. Jennifer Cindy Lai of the University of California, San Francisco, and her colleagues reported in the November issue of Gastroenterology.
Even more surprising, most of these candidates declined "not just one or two but a median of six liver offers during their time on the wait-list."
The "declined" donor organs were then successfully transplanted into lower-priority recipients.
These findings suggest that mortality among wait-listed patients "is not simply a result of not having the opportunity for transplantation, as many of us assume. Rather, wait-list mortality appears to result from opportunities for transplantation that were declined," Dr. Lai and her associates wrote.
The reasons that so many viable donor livers were initially declined are not yet clear. General, somewhat vague reasons were listed but not fully explained in the records the researchers analyzed for this study, which they obtained from the United Network for Organ Sharing/Organ Procurement Transplantation Network database.
The investigators assessed organ offers to 33,389 liver transplant candidates aged 18 years and older who were wait-listed across the United States between 2005 and 2010.
The reasons that proffered organs were declined, as listed in the medical records, fit into six broad categories: unfavorable donor age or quality of organ; unfavorable donor organ size/weight; other unfavorable donor factors, such as ABO blood transfusion incompatibility, "social history," "positive serologic tests," or "organ anatomical damage or defect"; unreadiness of the recipient, usually because he or she was ill, unavailable, refused the organ, or required multiple organ transplants at the same time; problems with the transplant program itself, such as a "heavy workload" or unavailability of a surgeon or operating room at the recipient’s medical center, failure to respond to the offer in a timely way, or excessive distance to ship the organ.
A total of 20% of the study population (6,737 patients) died or were removed from the wait-list because they became too sick before they could undergo transplantation. A total of 5,680 (84%) of those patients had been offered one or more donor livers before they died or were taken off the list.
Offers of donor livers were declined most often (68%) because of "unfavorable donor age or quality of organ," whereas 9% were declined because of unfavorable organ size, 15% because of "other donor factors," 4% because the recipient wasn’t ready, and 4% because of transplant program or miscellaneous other factors.
However, the dominant use of the "donor quality or age" refusal code in the database almost certainly "does not accurately or fully capture the true refusal reason," Dr. Lai and her associates said.
Even livers judged to be of high quality according to standard criteria were declined because of supposed "unfavorable donor age or quality of organ." But the investigators found no difference in the risk of graft failure between such high-quality livers that were declined and other high-quality livers that were accepted on the first offer.
Other reasons must be playing an important role in this high rate refusal, but "the nuances of these refusals cannot be determined" without more individualized data, they said.
Dr. Lai and her colleagues suggested that to cut down on refusals of apparently viable organs, the transplant community should "reduce the stigma associated with non–ideal livers, and set realistic expectations for wait-listed candidates" so that they’re less likely to pass up a suitable donation while assuming that a better offer will come along.
Patients also should be educated about the unpredictability of death or of sudden worsening of liver disease while on the wait-list. They should be advised that there is a survival benefit associated with the transplantation of any graft, compared with continuing on the wait-list.
In addition, the current regulatory environment focuses on transplant centers’ outcomes, which may influence some centers to discourage the acceptance of less than optimal donor organs. "This may be especially relevant for low-volume transplant centers, for whom even a small number of poor outcomes ... may make a relatively large difference in the centers’ perceived performance," the researchers wrote.
Finally, wait-list candidates should be encouraged to complete their transplant work-ups as expeditiously as possible to avoid having to refuse a donor offer simply because they have not yet undergone the necessary cardiac testing or cancer screening.
This study was supported by the National Institute of Diabetes and Digestive and Kidney Diseases and the University of California, San Francisco. No financial conflicts of interest were reported.
FROM GASTROENTEROLOGY
Pretreatment Care Predicts HCV Outcomes
Patients with hepatitis C infections are more likely to initiate appropriate antiviral therapy and achieve a sustained virologic response if they receive high quality health care, Dr. Fasiha Kanwal of the Michael E. DeBakey Veterans Affairs Medical Center, Houston, and her colleagues reported in the November issue of Clinical Gastroenterology and Hepatology.
In their study of nearly 35,000 adults with HCV, the odds of initiating antiviral therapy were threefold higher in patients who received optimal care from the moment HCV infection was diagnosed than in those who did not. And among patients who initiated antiviral therapy, the quality of care they received before that therapy even began strongly predicted whether they would complete antiviral therapy and achieve a sustained virologic response, the investigators said.
The study showed, however, that only 11% of patients received all of the appropriate initial care.
Process-of-care measures are frequently used to assess the quality of care for HCV, but until now no study has assessed whether these measures actually correlate with better outcomes. To address this issue, Dr. Kanwal and her associates "evaluated the relationship between adherence to a broad set of process-based measures in HCV and 3 subsequent HCV-specific endpoints: receipt of antiviral treatment, completion of antiviral treatment, and the clinical outcome associated with improved survival: sustained virologic response."
The investigators used data from the VA registry on HCV clinical care, which covers patient demographics, lab tests, pharmacy information, and data on inpatient and outpatient care for approximately 300,000 patients across the country. For this study, they included data on 34,749 adults.
The mean subject age was 53 years, and 97% were men. Approximately half the study population was white and 26% was black; ethnicity was not reported for the others.
The researchers assessed seven process-of-care measures: confirmation of HCV viremia, evaluation by HCV specialists, HCV genotype testing, liver biopsy for those found to have genotype 1 HCV, and the ruling out of liver diseases related to hepatitis B, autoimmunity, or iron overload.
They also assessed seven process-of-care measures related to the prevention and management of comorbid conditions: HIV testing; hepatitis A and B serology testing, hepatitis A and B vaccination if serology results proved negative; treatment of depression; and treatment of substance abuse disorder.
Finally, they assessed six process-of-care measures related to monitoring of antiviral therapy’s effects: testing of viral load before antivirals were initiated and again at weeks 12, 24, and 48; reduction of ribavirin dose if anemia developed during treatment; and avoidance of prescribing growth-stimulating factors for leukopenia during antiviral therapy.
Overall, only 11% of the study subjects received all of the appropriate initial care, and 8% received all the appropriate care related to prevention and management of comorbid conditions. Moreover, of the study subjects who received antiviral therapy, just 37% received all the appropriate monitoring of treatment effects Dr. Kanwal and her associates said.
In patients who received optimal care before a definitive diagnosis was made, the odds of receiving antiviral therapy were 3.2 times higher than in patients who did not receive optimal care before diagnosis, they reported.
Similarly, patients who received optimal preventive and comorbid-condition care showed rates of antiviral therapy that were 36% higher than those of patients who received suboptimal preventive and comorbid-condition care.
The strong association between fulfillment of these process-of-care measures and appropriate antiviral therapy remained robust in a series of sensitivity analyses, which means it’s likely that meeting process-of-care goals directly leads to better HCV outcomes, Dr. Kanwal and her colleagues said.
The investigators could not, however, rule out the possibility that meeting these goals is simply a marker of more compliant patients, which in turn produces better outcomes.
The study findings imply that the effectiveness of the two new direct-acting antiviral agents that recently became available for HCV may hinge on the quality of care patients are receiving before they even start taking these drugs, rather than simply on the effectiveness of the drugs alone, Dr. Kanwal and her associates said.
This study was supported by the U.S. Department of Veterans Affairs Health Services Research and Development Service. No financial conflicts of interest were reported.
Financial reimbursement in medicine has long been driven by volume rather than quality. This incentive structure is changing, and in the near future practitioners will experience increased scrutiny of the quality of care we provide.

|
|
Quality can be divided into structure (having the proper equipment to clean endoscopes), process (testing for latent tuberculosis before beginning anti–tumor necrosis factor therapy), and outcomes (perforation rate during colonoscopy). The latter is, of course, the most important, but it is also the most difficult to measure because of low event rates and inadequate risk adjustment. Therefore, most quality measures are based on processes of care. For quality measurement to yield any true value to the patient, however, it is important that these processes are clearly linked to better patient outcomes.
Hepatitis C, which affects 1.3% of Americans, has recently been a target disease for measuring and improving quality of care. Dr. Kanwal and her colleagues found that patients receiving optimum process-related quality care were more likely to undergo antiviral therapy. Such patients also were more likely to complete treatment once started and to achieve sustained virologic response if treatment was completed. Since these findings persisted despite adjustment for numerous potential confounders such as comorbidities, it appears that higher quality of care (as measured by processes) may truly lead to better patient outcomes.
What does this mean for practitioners? Particularly in the era of triple therapy, hepatitis C virus (HCV) infection cannot be managed like any other disease. Protocols and tracking systems need to be developed to ensure that quality measures are met. Many practices find it helpful to funnel HCV patients to a single person for case management, such as a nurse or midlevel provider. Hopefully, these efforts, in conjunction with newer antivirals, will soon eradicate hepatitis C altogether.
Michael Volk, M.D., is assistant professor of hepatology at the University of Michigan, Ann Arbor.
Financial reimbursement in medicine has long been driven by volume rather than quality. This incentive structure is changing, and in the near future practitioners will experience increased scrutiny of the quality of care we provide.

|
|
Quality can be divided into structure (having the proper equipment to clean endoscopes), process (testing for latent tuberculosis before beginning anti–tumor necrosis factor therapy), and outcomes (perforation rate during colonoscopy). The latter is, of course, the most important, but it is also the most difficult to measure because of low event rates and inadequate risk adjustment. Therefore, most quality measures are based on processes of care. For quality measurement to yield any true value to the patient, however, it is important that these processes are clearly linked to better patient outcomes.
Hepatitis C, which affects 1.3% of Americans, has recently been a target disease for measuring and improving quality of care. Dr. Kanwal and her colleagues found that patients receiving optimum process-related quality care were more likely to undergo antiviral therapy. Such patients also were more likely to complete treatment once started and to achieve sustained virologic response if treatment was completed. Since these findings persisted despite adjustment for numerous potential confounders such as comorbidities, it appears that higher quality of care (as measured by processes) may truly lead to better patient outcomes.
What does this mean for practitioners? Particularly in the era of triple therapy, hepatitis C virus (HCV) infection cannot be managed like any other disease. Protocols and tracking systems need to be developed to ensure that quality measures are met. Many practices find it helpful to funnel HCV patients to a single person for case management, such as a nurse or midlevel provider. Hopefully, these efforts, in conjunction with newer antivirals, will soon eradicate hepatitis C altogether.
Michael Volk, M.D., is assistant professor of hepatology at the University of Michigan, Ann Arbor.
Financial reimbursement in medicine has long been driven by volume rather than quality. This incentive structure is changing, and in the near future practitioners will experience increased scrutiny of the quality of care we provide.

|
|
Quality can be divided into structure (having the proper equipment to clean endoscopes), process (testing for latent tuberculosis before beginning anti–tumor necrosis factor therapy), and outcomes (perforation rate during colonoscopy). The latter is, of course, the most important, but it is also the most difficult to measure because of low event rates and inadequate risk adjustment. Therefore, most quality measures are based on processes of care. For quality measurement to yield any true value to the patient, however, it is important that these processes are clearly linked to better patient outcomes.
Hepatitis C, which affects 1.3% of Americans, has recently been a target disease for measuring and improving quality of care. Dr. Kanwal and her colleagues found that patients receiving optimum process-related quality care were more likely to undergo antiviral therapy. Such patients also were more likely to complete treatment once started and to achieve sustained virologic response if treatment was completed. Since these findings persisted despite adjustment for numerous potential confounders such as comorbidities, it appears that higher quality of care (as measured by processes) may truly lead to better patient outcomes.
What does this mean for practitioners? Particularly in the era of triple therapy, hepatitis C virus (HCV) infection cannot be managed like any other disease. Protocols and tracking systems need to be developed to ensure that quality measures are met. Many practices find it helpful to funnel HCV patients to a single person for case management, such as a nurse or midlevel provider. Hopefully, these efforts, in conjunction with newer antivirals, will soon eradicate hepatitis C altogether.
Michael Volk, M.D., is assistant professor of hepatology at the University of Michigan, Ann Arbor.
Patients with hepatitis C infections are more likely to initiate appropriate antiviral therapy and achieve a sustained virologic response if they receive high quality health care, Dr. Fasiha Kanwal of the Michael E. DeBakey Veterans Affairs Medical Center, Houston, and her colleagues reported in the November issue of Clinical Gastroenterology and Hepatology.
In their study of nearly 35,000 adults with HCV, the odds of initiating antiviral therapy were threefold higher in patients who received optimal care from the moment HCV infection was diagnosed than in those who did not. And among patients who initiated antiviral therapy, the quality of care they received before that therapy even began strongly predicted whether they would complete antiviral therapy and achieve a sustained virologic response, the investigators said.
The study showed, however, that only 11% of patients received all of the appropriate initial care.
Process-of-care measures are frequently used to assess the quality of care for HCV, but until now no study has assessed whether these measures actually correlate with better outcomes. To address this issue, Dr. Kanwal and her associates "evaluated the relationship between adherence to a broad set of process-based measures in HCV and 3 subsequent HCV-specific endpoints: receipt of antiviral treatment, completion of antiviral treatment, and the clinical outcome associated with improved survival: sustained virologic response."
The investigators used data from the VA registry on HCV clinical care, which covers patient demographics, lab tests, pharmacy information, and data on inpatient and outpatient care for approximately 300,000 patients across the country. For this study, they included data on 34,749 adults.
The mean subject age was 53 years, and 97% were men. Approximately half the study population was white and 26% was black; ethnicity was not reported for the others.
The researchers assessed seven process-of-care measures: confirmation of HCV viremia, evaluation by HCV specialists, HCV genotype testing, liver biopsy for those found to have genotype 1 HCV, and the ruling out of liver diseases related to hepatitis B, autoimmunity, or iron overload.
They also assessed seven process-of-care measures related to the prevention and management of comorbid conditions: HIV testing; hepatitis A and B serology testing, hepatitis A and B vaccination if serology results proved negative; treatment of depression; and treatment of substance abuse disorder.
Finally, they assessed six process-of-care measures related to monitoring of antiviral therapy’s effects: testing of viral load before antivirals were initiated and again at weeks 12, 24, and 48; reduction of ribavirin dose if anemia developed during treatment; and avoidance of prescribing growth-stimulating factors for leukopenia during antiviral therapy.
Overall, only 11% of the study subjects received all of the appropriate initial care, and 8% received all the appropriate care related to prevention and management of comorbid conditions. Moreover, of the study subjects who received antiviral therapy, just 37% received all the appropriate monitoring of treatment effects Dr. Kanwal and her associates said.
In patients who received optimal care before a definitive diagnosis was made, the odds of receiving antiviral therapy were 3.2 times higher than in patients who did not receive optimal care before diagnosis, they reported.
Similarly, patients who received optimal preventive and comorbid-condition care showed rates of antiviral therapy that were 36% higher than those of patients who received suboptimal preventive and comorbid-condition care.
The strong association between fulfillment of these process-of-care measures and appropriate antiviral therapy remained robust in a series of sensitivity analyses, which means it’s likely that meeting process-of-care goals directly leads to better HCV outcomes, Dr. Kanwal and her colleagues said.
The investigators could not, however, rule out the possibility that meeting these goals is simply a marker of more compliant patients, which in turn produces better outcomes.
The study findings imply that the effectiveness of the two new direct-acting antiviral agents that recently became available for HCV may hinge on the quality of care patients are receiving before they even start taking these drugs, rather than simply on the effectiveness of the drugs alone, Dr. Kanwal and her associates said.
This study was supported by the U.S. Department of Veterans Affairs Health Services Research and Development Service. No financial conflicts of interest were reported.
Patients with hepatitis C infections are more likely to initiate appropriate antiviral therapy and achieve a sustained virologic response if they receive high quality health care, Dr. Fasiha Kanwal of the Michael E. DeBakey Veterans Affairs Medical Center, Houston, and her colleagues reported in the November issue of Clinical Gastroenterology and Hepatology.
In their study of nearly 35,000 adults with HCV, the odds of initiating antiviral therapy were threefold higher in patients who received optimal care from the moment HCV infection was diagnosed than in those who did not. And among patients who initiated antiviral therapy, the quality of care they received before that therapy even began strongly predicted whether they would complete antiviral therapy and achieve a sustained virologic response, the investigators said.
The study showed, however, that only 11% of patients received all of the appropriate initial care.
Process-of-care measures are frequently used to assess the quality of care for HCV, but until now no study has assessed whether these measures actually correlate with better outcomes. To address this issue, Dr. Kanwal and her associates "evaluated the relationship between adherence to a broad set of process-based measures in HCV and 3 subsequent HCV-specific endpoints: receipt of antiviral treatment, completion of antiviral treatment, and the clinical outcome associated with improved survival: sustained virologic response."
The investigators used data from the VA registry on HCV clinical care, which covers patient demographics, lab tests, pharmacy information, and data on inpatient and outpatient care for approximately 300,000 patients across the country. For this study, they included data on 34,749 adults.
The mean subject age was 53 years, and 97% were men. Approximately half the study population was white and 26% was black; ethnicity was not reported for the others.
The researchers assessed seven process-of-care measures: confirmation of HCV viremia, evaluation by HCV specialists, HCV genotype testing, liver biopsy for those found to have genotype 1 HCV, and the ruling out of liver diseases related to hepatitis B, autoimmunity, or iron overload.
They also assessed seven process-of-care measures related to the prevention and management of comorbid conditions: HIV testing; hepatitis A and B serology testing, hepatitis A and B vaccination if serology results proved negative; treatment of depression; and treatment of substance abuse disorder.
Finally, they assessed six process-of-care measures related to monitoring of antiviral therapy’s effects: testing of viral load before antivirals were initiated and again at weeks 12, 24, and 48; reduction of ribavirin dose if anemia developed during treatment; and avoidance of prescribing growth-stimulating factors for leukopenia during antiviral therapy.
Overall, only 11% of the study subjects received all of the appropriate initial care, and 8% received all the appropriate care related to prevention and management of comorbid conditions. Moreover, of the study subjects who received antiviral therapy, just 37% received all the appropriate monitoring of treatment effects Dr. Kanwal and her associates said.
In patients who received optimal care before a definitive diagnosis was made, the odds of receiving antiviral therapy were 3.2 times higher than in patients who did not receive optimal care before diagnosis, they reported.
Similarly, patients who received optimal preventive and comorbid-condition care showed rates of antiviral therapy that were 36% higher than those of patients who received suboptimal preventive and comorbid-condition care.
The strong association between fulfillment of these process-of-care measures and appropriate antiviral therapy remained robust in a series of sensitivity analyses, which means it’s likely that meeting process-of-care goals directly leads to better HCV outcomes, Dr. Kanwal and her colleagues said.
The investigators could not, however, rule out the possibility that meeting these goals is simply a marker of more compliant patients, which in turn produces better outcomes.
The study findings imply that the effectiveness of the two new direct-acting antiviral agents that recently became available for HCV may hinge on the quality of care patients are receiving before they even start taking these drugs, rather than simply on the effectiveness of the drugs alone, Dr. Kanwal and her associates said.
This study was supported by the U.S. Department of Veterans Affairs Health Services Research and Development Service. No financial conflicts of interest were reported.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Less Liver Cancer in HCV Patients Given Antiviral Therapy
Antiviral therapy may reduce the risk of hepatocellular carcinoma in patients with hepatitis C infections, Dr. Nina Kimer reported in the Oct. 22 issue of BMJ Open, published online.
Moreover, the effect seems to persist regardless of whether sustained virologic response is achieved.
Dr. Kimer, of Copenhagen University Hospital, Hvidovre, Denmark, and colleagues conducted a meta-analysis of trials looking at hepatitis C-related cirrhosis or fibrosis (BMJ Open 2012;2:e001313 [doi:10.1136/bmjopen-2012-001313]).
The primary analysis focused on randomized controlled trials, although prospective cohort studies with control groups were included in sensitivity analyses.
The authors searched the Cochrane Library, PubMed, EMBASE, and Web of Science databases, as well as reference lists from relevant papers, conference proceedings, and the World Health Organization Trial Search Portal.
Studies looking at HIV and chronic hepatitis B were excluded, for a total of eight randomized trials and five prospective cohort studies.
The duration of therapy (which included pegylated interferon in two trials, interferon plus ribavirin in one trial, and interferon monotherapy in the remaining trials) varied from 1 to 5 years. Follow-up ranged from 2 to 8.7 years.
Overall, the authors found that 81 of 1,156 patients who received antiviral therapy and 129 of 1,074 control patients who received no therapy developed hepatocellular carcinoma.
The relative risk for antiviral therapy, compared with no treatment, was 0.53 (95% confidence interval, 0.34-0.81).
"The corresponding number needed to treat to prevent one case of [hepatocellular carcinoma] was eight patients," added the researchers.
And while the effect of antivirals was more pronounced among patients with a virological response (RR 0.15; 95% CI, 0.05-0.45), there was nevertheless a clear reduction in risk for nonresponders to antivirals, with a relative risk of 0.57, compared with controls (95% CI 0.37-0.85).
"Although the intervention was more beneficial among sustained virological responders than nonresponders, there was a clear effect in both patient groups. ... Antiviral therapy may have beneficial effects on the risk of developing HCC that are unrelated to the virological response."
On the other hand, there was no reported effect of antiviral therapy on all-cause mortality, liver-related mortality, or liver-related morbidity in this population.
Dr. Kimer conceded several limitations to this analysis. "Only two of the included trials evaluated pegylated interferon, which is the current standard treatment for chronic hepatitis C."
Moreover, the duration of treatment in several included studies was "relatively long, which may increase the proportion of responders."
Finally, Dr. Kimer added that the researchers were unable to perform subgroup analysis to determine which treatment duration or dose is best.
"Based on the duration of follow-up and the lack of clear evidence concerning morbidity or mortality, we cannot exclude that interferon delays rather than prevents carcinogenesis," concluded the investigators.
Nevertheless, the "protection from HCC might be even better among patients in current antiviral therapy since the proportion of virological responders continues to increase with ongoing improvements in therapy," they wrote.
"Additional randomized trials with longer follow-up are still warranted to determine whether this is the case," they noted.
The authors disclosed having no outside funding and no competing interests related to this study.
sustained virologic response, hepatitis C-related cirrhosis, fibrosis,
Antiviral therapy may reduce the risk of hepatocellular carcinoma in patients with hepatitis C infections, Dr. Nina Kimer reported in the Oct. 22 issue of BMJ Open, published online.
Moreover, the effect seems to persist regardless of whether sustained virologic response is achieved.
Dr. Kimer, of Copenhagen University Hospital, Hvidovre, Denmark, and colleagues conducted a meta-analysis of trials looking at hepatitis C-related cirrhosis or fibrosis (BMJ Open 2012;2:e001313 [doi:10.1136/bmjopen-2012-001313]).
The primary analysis focused on randomized controlled trials, although prospective cohort studies with control groups were included in sensitivity analyses.
The authors searched the Cochrane Library, PubMed, EMBASE, and Web of Science databases, as well as reference lists from relevant papers, conference proceedings, and the World Health Organization Trial Search Portal.
Studies looking at HIV and chronic hepatitis B were excluded, for a total of eight randomized trials and five prospective cohort studies.
The duration of therapy (which included pegylated interferon in two trials, interferon plus ribavirin in one trial, and interferon monotherapy in the remaining trials) varied from 1 to 5 years. Follow-up ranged from 2 to 8.7 years.
Overall, the authors found that 81 of 1,156 patients who received antiviral therapy and 129 of 1,074 control patients who received no therapy developed hepatocellular carcinoma.
The relative risk for antiviral therapy, compared with no treatment, was 0.53 (95% confidence interval, 0.34-0.81).
"The corresponding number needed to treat to prevent one case of [hepatocellular carcinoma] was eight patients," added the researchers.
And while the effect of antivirals was more pronounced among patients with a virological response (RR 0.15; 95% CI, 0.05-0.45), there was nevertheless a clear reduction in risk for nonresponders to antivirals, with a relative risk of 0.57, compared with controls (95% CI 0.37-0.85).
"Although the intervention was more beneficial among sustained virological responders than nonresponders, there was a clear effect in both patient groups. ... Antiviral therapy may have beneficial effects on the risk of developing HCC that are unrelated to the virological response."
On the other hand, there was no reported effect of antiviral therapy on all-cause mortality, liver-related mortality, or liver-related morbidity in this population.
Dr. Kimer conceded several limitations to this analysis. "Only two of the included trials evaluated pegylated interferon, which is the current standard treatment for chronic hepatitis C."
Moreover, the duration of treatment in several included studies was "relatively long, which may increase the proportion of responders."
Finally, Dr. Kimer added that the researchers were unable to perform subgroup analysis to determine which treatment duration or dose is best.
"Based on the duration of follow-up and the lack of clear evidence concerning morbidity or mortality, we cannot exclude that interferon delays rather than prevents carcinogenesis," concluded the investigators.
Nevertheless, the "protection from HCC might be even better among patients in current antiviral therapy since the proportion of virological responders continues to increase with ongoing improvements in therapy," they wrote.
"Additional randomized trials with longer follow-up are still warranted to determine whether this is the case," they noted.
The authors disclosed having no outside funding and no competing interests related to this study.
Antiviral therapy may reduce the risk of hepatocellular carcinoma in patients with hepatitis C infections, Dr. Nina Kimer reported in the Oct. 22 issue of BMJ Open, published online.
Moreover, the effect seems to persist regardless of whether sustained virologic response is achieved.
Dr. Kimer, of Copenhagen University Hospital, Hvidovre, Denmark, and colleagues conducted a meta-analysis of trials looking at hepatitis C-related cirrhosis or fibrosis (BMJ Open 2012;2:e001313 [doi:10.1136/bmjopen-2012-001313]).
The primary analysis focused on randomized controlled trials, although prospective cohort studies with control groups were included in sensitivity analyses.
The authors searched the Cochrane Library, PubMed, EMBASE, and Web of Science databases, as well as reference lists from relevant papers, conference proceedings, and the World Health Organization Trial Search Portal.
Studies looking at HIV and chronic hepatitis B were excluded, for a total of eight randomized trials and five prospective cohort studies.
The duration of therapy (which included pegylated interferon in two trials, interferon plus ribavirin in one trial, and interferon monotherapy in the remaining trials) varied from 1 to 5 years. Follow-up ranged from 2 to 8.7 years.
Overall, the authors found that 81 of 1,156 patients who received antiviral therapy and 129 of 1,074 control patients who received no therapy developed hepatocellular carcinoma.
The relative risk for antiviral therapy, compared with no treatment, was 0.53 (95% confidence interval, 0.34-0.81).
"The corresponding number needed to treat to prevent one case of [hepatocellular carcinoma] was eight patients," added the researchers.
And while the effect of antivirals was more pronounced among patients with a virological response (RR 0.15; 95% CI, 0.05-0.45), there was nevertheless a clear reduction in risk for nonresponders to antivirals, with a relative risk of 0.57, compared with controls (95% CI 0.37-0.85).
"Although the intervention was more beneficial among sustained virological responders than nonresponders, there was a clear effect in both patient groups. ... Antiviral therapy may have beneficial effects on the risk of developing HCC that are unrelated to the virological response."
On the other hand, there was no reported effect of antiviral therapy on all-cause mortality, liver-related mortality, or liver-related morbidity in this population.
Dr. Kimer conceded several limitations to this analysis. "Only two of the included trials evaluated pegylated interferon, which is the current standard treatment for chronic hepatitis C."
Moreover, the duration of treatment in several included studies was "relatively long, which may increase the proportion of responders."
Finally, Dr. Kimer added that the researchers were unable to perform subgroup analysis to determine which treatment duration or dose is best.
"Based on the duration of follow-up and the lack of clear evidence concerning morbidity or mortality, we cannot exclude that interferon delays rather than prevents carcinogenesis," concluded the investigators.
Nevertheless, the "protection from HCC might be even better among patients in current antiviral therapy since the proportion of virological responders continues to increase with ongoing improvements in therapy," they wrote.
"Additional randomized trials with longer follow-up are still warranted to determine whether this is the case," they noted.
The authors disclosed having no outside funding and no competing interests related to this study.
sustained virologic response, hepatitis C-related cirrhosis, fibrosis,
sustained virologic response, hepatitis C-related cirrhosis, fibrosis,
FROM BMJ OPEN
Major Finding: Antiviral therapy for hepatitis C carried a relative risk of 0.53 for developing hepatocellular carcinoma, compared with no treatment (95% confidence interval, 0.34-0.81).
Data Source: A meta-analysis of randomized controlled trials.
Disclosures: The authors disclosed having no outside funding and no competing interests related to this study.