User login
Therapeutic management of NAFLD
Nonalcoholic fatty liver disease (NAFLD) is defined by the presence of hepatic steatosis detected on either imaging or histology in the absence of secondary causes of fatty liver (e.g., excessive alcohol consumption) or other chronic liver diseases.1 For practical NAFLD diagnosis purposes, excessive alcohol intake can be defined as an active or recent history of more than 21 standard drinks per week in men and more than14 standard drinks per week in women. For the sake of terminology, NAFLD is characterized by fatty liver infiltration, affecting at least 5% of hepatocytes, with no evidence of hepatocyte injury, whereas nonalcoholic steatohepatitis (NASH) is defined as the presence of necroinflammation with or without fibrosis in a background of fatty liver.1
Natural history
NASH and the degree of fibrosis are the two most important determinants of the natural history of NAFLD. NASH can evolve into fibrosis and cirrhosis, whereas advanced fibrosis and cirrhosis (stages 3 or 4 of fibrosis) significantly increase the risk of liver-related decompensation and mortality. NAFLD, per se, has been associated with an increased risk of overall mortality, compared with that of the general population.2 The three most common causes of mortality for patients with NAFLD are cardiovascular diseases (CVD), extrahepatic malignancies, and liver-related deaths. Mortality and liver-related events, including hepatic decompensation and hepatocellular carcinoma (HCC), may significantly increase in a dose-dependent manner with increasing fibrosis stages, and stages 3 or 4 of fibrosis may display the highest rates of all-cause mortality and liver-related events.3,4 It is important to note, however, that almost 15% of HCCs occur in patients with NAFLD who do not have cirrhosis.5 The presence of commonly associated comorbidities such as obesity, insulin resistance or diabetes, dyslipidemia, hypothyroidism, polycystic ovary syndrome, and sleep apnea may contribute to an increased risk of NASH and advanced fibrosis and, therefore, an accelerated clinical course of NAFLD.
Nonpharmacological interventions
Lifestyle modification
Lifestyle modification to achieve weight loss remains a first-line intervention in patients with NAFLD. Weight loss achieved either by hypocaloric diet alone or in conjunction with increased physical activity can be beneficial for all patients with NAFLD. The benefits extend not only to those who are overweight and obese but also to those within normal body weight (lean NAFLD).1,6,7 Weight loss of approximately 3%-5% is necessary to improve hepatic steatosis, but a greater weight loss (7%-10%) is required to improve other histopathological features like necroinflammatory lesions and fibrosis.8-10 Individuals with higher BMI and/or type 2 diabetes (T2D) will require a larger weight reduction to achieve a similar benefit on NAFLD-related features.7,8 Weight loss via lifestyle changes can also decrease hepatic venous pressure gradient (HVPG), with greater declines reported among those with more than 10% weight loss.11
Weight loss can be achieved through a variety of modalities, but long-term maintenance of lost weight is much more challenging. A combination of a hypocaloric diet with a caloric deficit of 500-1,000 kcal/d, alongside moderate-intensity exercise and intensive on-site behavioral treatment, will likely increase the possibility of a sustained weight loss over time.1,12 A growing body of scientific evidence indicates that a healthy diet that includes a reduction of high-glycemic-index foods and refined carbohydrates; increased consumption of monounsaturated fatty acids, omega-3 fatty acids, and fibers; and high intakes of olive oil, nuts, vegetables, fruits, legumes, whole grains, and fish can have beneficial effects on NAFLD and its severity.13-16 Adherence to these healthy dietary patterns has been associated with a marked reduction in CVD morbidity and mortality and is, thus, a strategic lifestyle recommendation for patients with NAFLD in whom the leading cause of morbidity and death is CVD.1,3
Exercise alone in adults with NAFLD may reduce hepatic steatosis, but its ability to improve inflammation and fibrosis has not been proven in well-designed RCTs.17,18 Physical activity and exercise have been shown to curb both the development and the progression of NAFLD, and beneficial effects could be achieved independent of weight loss.17,19,20 Most importantly, moderate-to-vigorous physical activity is likely associated with lower all-cause and cardiovascular mortality in patients with NAFLD.21
Heavy alcohol intake should be avoided by patients with NAFLD or NASH, and those with cirrhotic NASH should avoid any alcohol consumption given the risk of HCC and hepatic decompensation.1,4,22 Limiting light-to-moderate alcohol intake among patients without cirrhosis is still under debate.1 People with NAFLD may be advised to drink an equivalent of two to three 8-oz cups of regular brewed coffee daily as it has shown certain antifibrotic effects in NAFLD patients.23
Bariatric surgery
Bariatric surgery is an attractive therapeutic option for eligible obese patients with NAFLD. Bariatric surgery has the potential for inducing great weight loss and, therefore, reverses not only the steatosis, inflammation, and fibrosis among NAFLD individuals but also important comorbid conditions like T2D. A recent systematic review and meta-analysis examining data on the effects of bariatric surgery on histologic features of NAFLD from 32 cohort studies (no RCTs included) showed that bariatric surgery was associated with significant improvements in steatosis (66%), lobular inflammation (50%), ballooning degeneration (76%), and fibrosis (40%), and the benefits were significantly higher in those who underwent Roux-en-Y gastric bypass (RYGB). Of note, worsening of liver histology, including fibrosis, could be seen in up to 12% of patients who underwent bariatric surgery.24 The postsurgical weight regained after RYGB could explain partly the lack of fibrosis improvement or even worsening of fibrosis, although further research is needed to clarify these controversial findings.
RYGB and sleeve gastrectomy (SG) are the most commonly performed bariatric surgeries worldwide. Patients who undergo RYGB achieve higher weight loss when compared with those treated with SG.25 Among all bariatric procedures, RYGB could result in a higher proportion of complete resolution of NAFLD than SG, although evidence is inconclusive on fibrosis improvement rates.24,26 Most recently, a single-center RCT has compared the effects of RYGB vs. SG on liver fat content and fibrosis in patients with severe obesity and T2D.27 Data showed that both surgical procedures were highly and equally effective in reducing fatty liver content (quantified by magnetic resonance imaging), with an almost complete resolution of the fatty liver at 1 year of both surgical interventions. The beneficial effects of both GB and SG on fibrosis (assessed by enhanced liver test [ELF]) were less evident with no substantial difference between the two groups. Importantly, 69% of participants had an increase in their ELF scores during the study, despite the majority of participants achieving significant reductions in their body weights and better glycemic control at the end of the study. These findings might be considered with caution as several factors, such as the duration of the study (only 1 year) and lack of a liver biopsy to confirm fibrosis changes over time, could be influencing the study results.
Among all NAFLD phenotypes, those with cirrhosis and, most importantly, hepatic decompensation appear to be at increased risk of perioperative mortality and inpatient hospital stays than those without cirrhosis.28-29 Bariatric surgery is an absolute contraindication in patients with decompensated cirrhosis (Child B and Child C). Among compensated -Child A- cirrhotics, those with portal hypertension are at increased risk of morbidity and perioperative mortality.30 A recent analysis of National Inpatient Sample data suggested that the rates of complications in those with cirrhosis have decreased with time, which could be due to a better selection process and the use of more restrictive bariatric surgery in those with cirrhosis. Low volume centers (defined as less than 50 procedures per year) and nonrestrictive bariatric surgery were associated with a higher mortality rate. These data may suggest that patients with cirrhosis should undergo bariatric surgery only in high-volume centers after a multidisciplinary evaluation.31 Bariatric endoscopy is emerging as a new treatment for obesity, but the long-term durability of its effects remains to be determined.
A recent retrospective cohort study, including 1,158 adult patients with biopsy-proven NASH, has investigated the benefits of bariatric surgery on the occurrence of major adverse liver and cardiovascular outcomes in 650 patients who underwent bariatric surgery, compared with 508 patients who received nonsurgical usual care. This study showed that bariatric surgery was associated with 88% lower risk of progression of fatty liver to cirrhosis, liver cancer, or liver-related death, and 70% lower risk of serious CVD events during a follow-up period of 10 years.32 Within 1 year after surgery, 0.6% of patients died from surgical complications. The potential benefits of bariatric surgery in patients with NAFLD must be balanced against surgical risk, especially in eligible obese individuals with established cirrhosis. Data from a retrospective cohort study have shown that bariatric surgery in obese cirrhotic patients does not seem to associate with excessive mortality, compared with noncirrhotic obese patients.33 More data on immediate complication rates and long-term outcomes in patients with NAFLD by type of bariatric surgery is also required.
NAFLD as a standalone is not an indication for bariatric surgery. However, it could be considered in NAFLD patients who have a BMI of 40 kg/m2 or more without coexisting comorbidities or with a BMI of 35 kg/m2 or more and one or more severe obesity-related comorbidities, including T2D, hypertension, hyperlipidemia, or obstructive sleep apnea. Bariatric surgery must always be offered in centers with an experienced bariatric surgery program.1
Management of comorbidities
Given the multiple comorbidities associated with NAFLD and the potential to influence its severity, a comprehensive and multidisciplinary approach is needed to ameliorate not only the progression of liver disease but also those complications related to metabolic syndrome, hyperlipidemia, hypertension, diabetes, and other related conditions. Of note, all patients with NAFLD should receive aggressive management of comorbidities regardless of the severity of NAFLD. Ideally, a multidisciplinary team – including a primary care provider, an endocrinologist for patients with T2D, and a gastroenterologist/hepatologist – is needed to successfully manage patients with NAFLD.
It is well recognized that individuals with biopsy-proven NAFLD are at a higher risk of coronary heart disease, stroke, congestive heart failure, and death resulting from CVD when compared with the non-NAFLD population, and excess in CVD morbidity and mortality is evident across all stages of NAFLD and increases with worsening disease severity.34 The strong association between CVD and NAFLD has important clinical implications that may influence the decision to initiate treatment for primary prevention, including lipid-lowering, antihypertensive, or antiplatelet therapies.35 Statins are widely used to reduce LDL cholesterol and have been proven to be safe in NAFLD, including for those with elevated liver enzymes and even in compensated cirrhosis, in several studies conducted during the last 15 years.36 Statins are characterized by anti-inflammatory, anti-oxidative, antifibrotic, and plaque-stabilizing effects, whereby they may improve vascular and hepatic function among patients with NAFLD and reduce cardiovascular risk.37 Statin use for the treatment of NAFLD is still controversial and off-label and is not specifically recommended to treat NASH, but positive results have been shown for reductions in liver enzymes.1 A recent meta-analysis of 13 studies showed that continued use of statin in cirrhosis was associated with a 46% and 44% risk reduction in hepatic decompensation and mortality, respectively.38
The Food and Drug Administration has approved omega-3 (n-3) fatty acid agents and fibrates for the treatment of very high triglycerides (500 mg/dL or higher); however, no specific indications exist to treat NAFLD.1 Fenofibrate is related to mild aminotransferase elevations and, in some cases, severe liver injury, so caution must be paid, especially within 2 days of taking the drug.39-40
NAFLD phenotypes that need liver pharmacotherapy
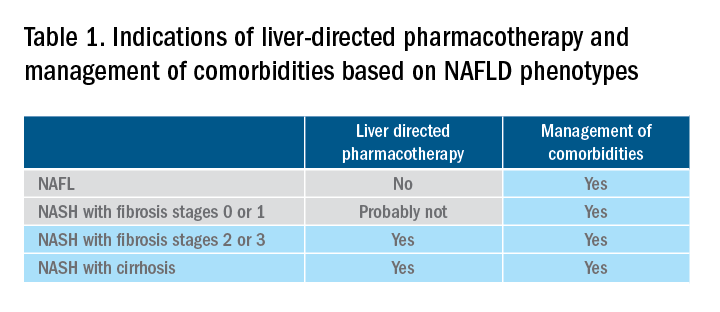
There are still no FDA-approved drugs or biological treatments for NASH. Pharmacological interventions aiming primarily at improving liver disease should generally be limited to those with biopsy-proven NASH and clinically significant fibrosis (fibrosis stages of 2 or greater).1,4 For FDA approval, medications used for treating NAFLD with fibrosis need to meet one of the following endpoint criteria: resolution of NASH without worsening of fibrosis, improvement in fibrosis without worsening of NASH, or both. In addition to those criteria, a new medication might improve the metabolic profile and have a tolerable safety profile. Table 1 displays those NAFLD phenotypes that will likely benefit from liver-directed therapy.
Obeticholic acid as an experimental therapy for NASH
A planned month-18 interim analysis of a multicentre, phase III RCT examined the efficacy and safety of obeticholic acid (OCA), a farnesoid X receptor agonist, in patients with NASH and stages 1-3 of fibrosis. The primary endpoint (fibrosis reduction 1 stage or more with no worsening of NASH) was met by 12% of patients in the placebo group, 18% of patients receiving OCA 10 mg (P = .045), and 23% of those receiving OCA 25 mg (P = .0002). An alternative primary endpoint of NASH resolution with no worsening of fibrosis was not met. OCA 25 mg led to the highest rates of pruritus and hyperlipidemia, compared with OCA 10 mg.42 These side effects seem to be related to the activation of the farnesoid X receptor.43
Currently available but off label medications
Vitamin E, an antioxidant, administered at a daily dose of 800 IU/day improves steatosis, inflammation, and ballooning, but not fibrosis in nondiabetic adults with biopsy-proven NASH.44 Vitamin E for 96 weeks was associated with a significantly higher rate of improvement in NASH (43% vs. 19%, P less than .01), compared with placebo.44 In the Treatment of Nonalcoholic Fatty Liver Disease in Children trial (TONIC), which examined vitamin E (800 IU/day) or metformin (500 mg twice daily) against placebo in children with biopsy-proven NAFLD, resolution of NASH was significantly greater in children treated with vitamin E than in children treated with placebo (58% vs. 28%, P less than .01). Metformin did not significantly improve the NASH resolution rates, compared with placebo (41% vs. 28%, P = .23). Vitamin E could be recommended for nondiabetic adults or children if lifestyle modifications do not produce the expected results as a result of noncompliance or ineffectiveness. Since continued use of vitamin E has been suggested to be associated with a very small increase in the risk for prostate cancer (an absolute increase of 1.6 per 1,000 person-years of vitamin E use) in men, risks and benefits should be discussed with each patient before starting therapy. A meta-analysis of nine placebo-controlled trials including roughly 119,000 patients reported that vitamin E supplementation increases the risk of hemorrhagic stroke by 20% while reducing ischemic stroke by 10%. It was estimated that vitamin E supplementation would prevent one ischemic stroke per 476 treated patients while inducing one hemorrhagic stroke for every 1,250 patients. It is noteworthy that the combination of vitamin E with anticoagulant and/or antiplatelet therapy was not examined in this trial, so we could not determine how combination therapy might affect the risk of ischemic or hemorrhagic stroke.45
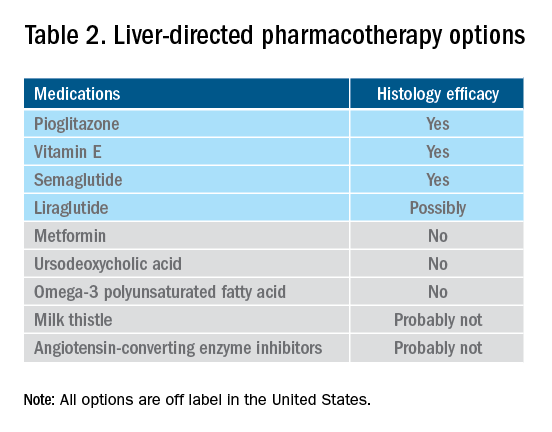
Thiazolidinediones drugs have been reported to be effective in improving NAFLD in many human studies. Evidence from RCTs suggests that pioglitazone could significantly improve glucose metabolism, alanine aminotransferase, and liver histology – such as hepatic steatosis, lobular inflammation, and ballooning degeneration – among patients with or without T2D. However, the beneficial effects on improving fibrosis remain to be verified.1,46 Because of safety concerns, the risk/benefit balance of using pioglitazone to treat NASH should be discussed with each patient.47-48 Pioglitazone has been associated with long-term risk of bladder cancer,49 congestive heart failure,50 and bone fractures.51 Data from the Pioglitazone, Vitamin E, or Placebo for Nonalcoholic Steatohepatitis (PIVENS) trial showed that pioglitazone was significantly associated with weight gain but with no other serious adverse events. However, this study was not powered to test any safety-related hypotheses.44
Glucagon-like peptide 1 analogs have been reported to induce weight loss and reduce insulin resistance, which may lead to improvements in NAFLD. Phase II RCTs of glucagon-like peptide 1 receptor agonists (liraglutide and semaglutide) for the treatment of biopsy-proven NASH showed significant improvements in serum liver enzymes, steatosis, and inflammation, as well as NASH resolution without worsening liver fibrosis, although no direct benefit was observed in reversing fibrosis.52-53 One of these studies explores the efficacy and safety of different doses of daily subcutaneous semaglutide vs. placebo on the rates of resolution of NASH with no worsening of fibrosis. The highest dose (0.4 mg) showed the greatest difference (59% vs. 17%, P less than .01), compared with the placebo arm. However, there was no difference in improvement in fibrosis stage between the two groups (43% in the 0.4-mg group vs. 33% in the placebo group, P = .48).53 Gastrointestinal adverse events were common in the semaglutide arm.
“Spontaneous” NASH resolution and fibrosis improvement are commonly seen in participants assigned to placebo arms in clinical trials. A recent meta-analysis of 43 RCTs including 2649 placebo-treated patients showed a pooled estimate of NASH resolution without worsening of fibrosis and 1 stage reduction or more in fibrosis of 12% and 19%, respectively. Relevant factors involved in “spontaneous” NASH improvement are unknown but could be related to changes in BMI resulting from lifestyle changes, race and ethnicity, age, and, likely, NAFLD-related genetic variations, although more data is needed to better understand the histologic response in placebo-treated patients.54
Semaglutide injections (2.4 mg once weekly) or (2.0 mg once weekly) have been recently approved by the FDA for chronic weight management in adults with obesity or overweight with at least one weight-related condition or glucose control of T2D, respectively. Of note, the semaglutide dose used in the NASH trial is not currently available for the treatment of patients who are overweight/obese or have T2D, but the beneficial effects on body weight reductions and glucose control are similar overall to the effects seen with currently available doses for management of obesity or diabetes. One may consider using semaglutide in patients who are overweight/obese or have T2D with NASH, but in the senior author’s experience, it has been quite challenging to receive the payer’s approval, as its use is not specifically approved to treat liver disease.1
How to follow patients with NAFLD in the clinic
Once a diagnosis of NAFLD is made, the use of noninvasive testing may aid to identify which patients are at high risk of fibrosis. Easy to use clinical tools, such as the NAFLD Fibrosis Score and the Fib-4 index, and liver stiffness measurements using vibration-controlled transient elastography (FibroScan) or magnetic resonance elastography (MRE) are clinically useful noninvasive tools for identifying patients with NAFLD who have a higher likelihood of progressing to advanced fibrosis.1,55 The use of either NAFLD Fibrosis Score (less than -1.455) or Fib-4 index (less than 1.30) low cutoffs may be particularly useful to rule out advanced fibrosis. People with a NAFLD Fibrosis Score (greater than –1.455) or Fib-4 index (greater than 1.30) should undergo liver stiffness measurement (LSM) via FibroScan. Those with an LSM of 8 kPa or higher should be referred to specialized care, where a decision to perform a liver biopsy and initiate monitoring and therapy will be taken. MRE is the most accurate noninvasive method for the estimation of liver fibrosis. When MRE is available, it can be a diagnostic alternative to accurately rule in and rule out patients with advanced fibrosis. This technique can be preferred in clinical trials, but it is rarely used in clinical practice because it is expensive and not easily available. Reassessment by noninvasive scores at 1-3 years’ follow-up will be considered for those with an LSM less than 8 kPa. Patients with NASH cirrhosis should be screened for both gastroesophageal varix and HCC according to the American Association for the Study of Liver Diseases guidelines.56-57
Dr. Vilar-Gomez is assistant professor in the division of gastroenterology and hepatology at Indiana University, Indianapolis. Dr. Chalasani is vice president for academic affairs at Indiana University Health, Indianapolis, and the David W. Crabb Professor of Gastroenterology and Hepatology and an adjunct professor of anatomy, cell biology, and physiology in the division of gastroenterology and hepatology at Indiana University. Dr. Vilar-Gomez reports no financial conflicts of interest. Dr. Chalasani serves as a paid consultant to AbbVie, Boehringer-Ingelheim, Altimmune, Madrigal, Lilly, Zydus, and Galectin. He receives research support from Galectin and DSM.
References
1. Chalasani N et al. Hepatology 2018;67:328-57.
2. Söderberg C et al. Hepatology 2010;51:595-602.
3. Sanyal AJ et al. N Engl J Med 2021;385:1559-69.
4. Vilar-Gomez E et al. Gastroenterology 2018;155:443-57.e17.
5. Younossi ZM et al. Hepatology 2016;64:73-84.
6. EASL-EASD-EASO. J Hepatol 2016;64:1388-402.
7. Wong VW et al. J Hepatol 2018; 69:1349-56.
8. Vilar-Gomez E et al. Gastroenterology 2015;149:367-78.e5; quiz e14-5.
9. Promrat K et al. Hepatology 2010;51:121-9.
10. Wong VW et al. J Hepatol 2013;59:536-42.
11. Berzigotti A et al. Hepatology 2017;65:1293-1305.
12. Sacks FM et al. N Engl J Med 2009;360:859-73.
13. Vilar-Gomez E et al. Hepatology 2022 Jun;75(6):1491-1506.
14. Zelber-Sagi S et al. Liver Int 2017;37:936-49.
15. Hassani Zadeh S et al. J Gastroenterol Hepatol 2021;36:1470-8.
16. Yaskolka Meir A et al. Gut 2021;70:2085-95.
17. Sung KC et al. J Hepatol 2016;65:791-7.
18. Orci LA et al. Clin Gastroenterol Hepatol 2016;14:1398-411.
19. Ryu S et al. J Hepatol 2015;63:1229-37.
20. Kim D et al. Hepatology 2020;72:1556-68.
21. Kim D et al. Clin Gastroenterol Hepatol 2021;19:1240-7.e5.
22. Ascha MS et al. Hepatology 2010;51:1972-8.
23. Bambha K et al. Liver Int 2014;34:1250-8.
24. Lee Y et al. Clin Gastroenterol Hepatol 2019;17:1040-60.e11.
25. Grönroos S et al. JAMA Surg 2021;156:137-46.
26. Fakhry TK et al. Surg Obes Relat Dis 2019;15:502-11.
27. Seeberg KA et al. Ann Intern Med 2022;175:74-83.
28. Bower G et al. Obes Surg 2015;25:2280-9.
29. Jan A et al. Obes Surg 2015;25:1518-26.
30. Hanipah ZN et al. Obes Surg 2018;28:3431-8.
31. Are VS et al. Am J Gastroenterol 2020;115:1849-56.
32. Aminian A et al. JAMA 2021;326:2031-42.
33. Vuppalanchi R et al. Ann Surg 2022;275:e174-80.
34. Simon TG et al. Gut 2021. doi: 10.1136/gutjnl-2021-325724.
35. Lonardo A et al. J Hepatol 2018;68:335-52.
36. Chalasani N et al. Gastroenterology 2004;126:1287-92.
37. Pastori D et al. Dig Liver Dis 2015;47:4-11.
38. Kim RG et al. Clin Gastroenterol Hepatol 2017;15:1521-30.e8.
39. Ahmad J et al. Dig Dis Sci 2017;62:3596-604.
40. Chalasani NP et al. Am J Gastroenterol 2021;116(5):878-98.
41. Rinella ME et al. Hepatology 2019;70:1424-36.
42. Younossi ZM et al. Lancet 2019;394:2184-96.
43. Ratziu V. Clin Liver Dis (Hoboken) 2021;17:398-400.
44. Sanyal AJ et al. N Engl J Med 2010;341:1675-85.
45. Schürks M et al. BMJ 2010;341:c5702.
46. Cusi K et al. Ann Intern Med 2016;165:305-15.
47. Lewis JD et al. JAMA 2015;314:265-77.
48. Billington EO et al. Diabetologia 2015;58:2238-46.
49. Lewis JD et al. Diabetes Care 2011;34:916-22.
50. Erdmann E et al. Diabetes Care 2007;30:2773-8.
51. Viscoli CM et al. J Clin Endocrinol Metab 2017;102:914-22.
52. Armstong MJ et al. Lancet 2016;387:679-90.
53. Newsome PN et al. N Engl J Med 2021;384:1113-24.
54. Ng CH et al. Hepatology 2022;75:1647-61.
55. Kanwal F et al. Gastroenterology 2021;161:1030-1042.e8.
56. Garcia-Tsao G et al. Hepatology 2017;65:310-35.
57. Heimbach JK et al. Hepatology 2018;67:358-80.
Nonalcoholic fatty liver disease (NAFLD) is defined by the presence of hepatic steatosis detected on either imaging or histology in the absence of secondary causes of fatty liver (e.g., excessive alcohol consumption) or other chronic liver diseases.1 For practical NAFLD diagnosis purposes, excessive alcohol intake can be defined as an active or recent history of more than 21 standard drinks per week in men and more than14 standard drinks per week in women. For the sake of terminology, NAFLD is characterized by fatty liver infiltration, affecting at least 5% of hepatocytes, with no evidence of hepatocyte injury, whereas nonalcoholic steatohepatitis (NASH) is defined as the presence of necroinflammation with or without fibrosis in a background of fatty liver.1
Natural history
NASH and the degree of fibrosis are the two most important determinants of the natural history of NAFLD. NASH can evolve into fibrosis and cirrhosis, whereas advanced fibrosis and cirrhosis (stages 3 or 4 of fibrosis) significantly increase the risk of liver-related decompensation and mortality. NAFLD, per se, has been associated with an increased risk of overall mortality, compared with that of the general population.2 The three most common causes of mortality for patients with NAFLD are cardiovascular diseases (CVD), extrahepatic malignancies, and liver-related deaths. Mortality and liver-related events, including hepatic decompensation and hepatocellular carcinoma (HCC), may significantly increase in a dose-dependent manner with increasing fibrosis stages, and stages 3 or 4 of fibrosis may display the highest rates of all-cause mortality and liver-related events.3,4 It is important to note, however, that almost 15% of HCCs occur in patients with NAFLD who do not have cirrhosis.5 The presence of commonly associated comorbidities such as obesity, insulin resistance or diabetes, dyslipidemia, hypothyroidism, polycystic ovary syndrome, and sleep apnea may contribute to an increased risk of NASH and advanced fibrosis and, therefore, an accelerated clinical course of NAFLD.
Nonpharmacological interventions
Lifestyle modification
Lifestyle modification to achieve weight loss remains a first-line intervention in patients with NAFLD. Weight loss achieved either by hypocaloric diet alone or in conjunction with increased physical activity can be beneficial for all patients with NAFLD. The benefits extend not only to those who are overweight and obese but also to those within normal body weight (lean NAFLD).1,6,7 Weight loss of approximately 3%-5% is necessary to improve hepatic steatosis, but a greater weight loss (7%-10%) is required to improve other histopathological features like necroinflammatory lesions and fibrosis.8-10 Individuals with higher BMI and/or type 2 diabetes (T2D) will require a larger weight reduction to achieve a similar benefit on NAFLD-related features.7,8 Weight loss via lifestyle changes can also decrease hepatic venous pressure gradient (HVPG), with greater declines reported among those with more than 10% weight loss.11
Weight loss can be achieved through a variety of modalities, but long-term maintenance of lost weight is much more challenging. A combination of a hypocaloric diet with a caloric deficit of 500-1,000 kcal/d, alongside moderate-intensity exercise and intensive on-site behavioral treatment, will likely increase the possibility of a sustained weight loss over time.1,12 A growing body of scientific evidence indicates that a healthy diet that includes a reduction of high-glycemic-index foods and refined carbohydrates; increased consumption of monounsaturated fatty acids, omega-3 fatty acids, and fibers; and high intakes of olive oil, nuts, vegetables, fruits, legumes, whole grains, and fish can have beneficial effects on NAFLD and its severity.13-16 Adherence to these healthy dietary patterns has been associated with a marked reduction in CVD morbidity and mortality and is, thus, a strategic lifestyle recommendation for patients with NAFLD in whom the leading cause of morbidity and death is CVD.1,3
Exercise alone in adults with NAFLD may reduce hepatic steatosis, but its ability to improve inflammation and fibrosis has not been proven in well-designed RCTs.17,18 Physical activity and exercise have been shown to curb both the development and the progression of NAFLD, and beneficial effects could be achieved independent of weight loss.17,19,20 Most importantly, moderate-to-vigorous physical activity is likely associated with lower all-cause and cardiovascular mortality in patients with NAFLD.21
Heavy alcohol intake should be avoided by patients with NAFLD or NASH, and those with cirrhotic NASH should avoid any alcohol consumption given the risk of HCC and hepatic decompensation.1,4,22 Limiting light-to-moderate alcohol intake among patients without cirrhosis is still under debate.1 People with NAFLD may be advised to drink an equivalent of two to three 8-oz cups of regular brewed coffee daily as it has shown certain antifibrotic effects in NAFLD patients.23
Bariatric surgery
Bariatric surgery is an attractive therapeutic option for eligible obese patients with NAFLD. Bariatric surgery has the potential for inducing great weight loss and, therefore, reverses not only the steatosis, inflammation, and fibrosis among NAFLD individuals but also important comorbid conditions like T2D. A recent systematic review and meta-analysis examining data on the effects of bariatric surgery on histologic features of NAFLD from 32 cohort studies (no RCTs included) showed that bariatric surgery was associated with significant improvements in steatosis (66%), lobular inflammation (50%), ballooning degeneration (76%), and fibrosis (40%), and the benefits were significantly higher in those who underwent Roux-en-Y gastric bypass (RYGB). Of note, worsening of liver histology, including fibrosis, could be seen in up to 12% of patients who underwent bariatric surgery.24 The postsurgical weight regained after RYGB could explain partly the lack of fibrosis improvement or even worsening of fibrosis, although further research is needed to clarify these controversial findings.
RYGB and sleeve gastrectomy (SG) are the most commonly performed bariatric surgeries worldwide. Patients who undergo RYGB achieve higher weight loss when compared with those treated with SG.25 Among all bariatric procedures, RYGB could result in a higher proportion of complete resolution of NAFLD than SG, although evidence is inconclusive on fibrosis improvement rates.24,26 Most recently, a single-center RCT has compared the effects of RYGB vs. SG on liver fat content and fibrosis in patients with severe obesity and T2D.27 Data showed that both surgical procedures were highly and equally effective in reducing fatty liver content (quantified by magnetic resonance imaging), with an almost complete resolution of the fatty liver at 1 year of both surgical interventions. The beneficial effects of both GB and SG on fibrosis (assessed by enhanced liver test [ELF]) were less evident with no substantial difference between the two groups. Importantly, 69% of participants had an increase in their ELF scores during the study, despite the majority of participants achieving significant reductions in their body weights and better glycemic control at the end of the study. These findings might be considered with caution as several factors, such as the duration of the study (only 1 year) and lack of a liver biopsy to confirm fibrosis changes over time, could be influencing the study results.
Among all NAFLD phenotypes, those with cirrhosis and, most importantly, hepatic decompensation appear to be at increased risk of perioperative mortality and inpatient hospital stays than those without cirrhosis.28-29 Bariatric surgery is an absolute contraindication in patients with decompensated cirrhosis (Child B and Child C). Among compensated -Child A- cirrhotics, those with portal hypertension are at increased risk of morbidity and perioperative mortality.30 A recent analysis of National Inpatient Sample data suggested that the rates of complications in those with cirrhosis have decreased with time, which could be due to a better selection process and the use of more restrictive bariatric surgery in those with cirrhosis. Low volume centers (defined as less than 50 procedures per year) and nonrestrictive bariatric surgery were associated with a higher mortality rate. These data may suggest that patients with cirrhosis should undergo bariatric surgery only in high-volume centers after a multidisciplinary evaluation.31 Bariatric endoscopy is emerging as a new treatment for obesity, but the long-term durability of its effects remains to be determined.
A recent retrospective cohort study, including 1,158 adult patients with biopsy-proven NASH, has investigated the benefits of bariatric surgery on the occurrence of major adverse liver and cardiovascular outcomes in 650 patients who underwent bariatric surgery, compared with 508 patients who received nonsurgical usual care. This study showed that bariatric surgery was associated with 88% lower risk of progression of fatty liver to cirrhosis, liver cancer, or liver-related death, and 70% lower risk of serious CVD events during a follow-up period of 10 years.32 Within 1 year after surgery, 0.6% of patients died from surgical complications. The potential benefits of bariatric surgery in patients with NAFLD must be balanced against surgical risk, especially in eligible obese individuals with established cirrhosis. Data from a retrospective cohort study have shown that bariatric surgery in obese cirrhotic patients does not seem to associate with excessive mortality, compared with noncirrhotic obese patients.33 More data on immediate complication rates and long-term outcomes in patients with NAFLD by type of bariatric surgery is also required.
NAFLD as a standalone is not an indication for bariatric surgery. However, it could be considered in NAFLD patients who have a BMI of 40 kg/m2 or more without coexisting comorbidities or with a BMI of 35 kg/m2 or more and one or more severe obesity-related comorbidities, including T2D, hypertension, hyperlipidemia, or obstructive sleep apnea. Bariatric surgery must always be offered in centers with an experienced bariatric surgery program.1
Management of comorbidities
Given the multiple comorbidities associated with NAFLD and the potential to influence its severity, a comprehensive and multidisciplinary approach is needed to ameliorate not only the progression of liver disease but also those complications related to metabolic syndrome, hyperlipidemia, hypertension, diabetes, and other related conditions. Of note, all patients with NAFLD should receive aggressive management of comorbidities regardless of the severity of NAFLD. Ideally, a multidisciplinary team – including a primary care provider, an endocrinologist for patients with T2D, and a gastroenterologist/hepatologist – is needed to successfully manage patients with NAFLD.
It is well recognized that individuals with biopsy-proven NAFLD are at a higher risk of coronary heart disease, stroke, congestive heart failure, and death resulting from CVD when compared with the non-NAFLD population, and excess in CVD morbidity and mortality is evident across all stages of NAFLD and increases with worsening disease severity.34 The strong association between CVD and NAFLD has important clinical implications that may influence the decision to initiate treatment for primary prevention, including lipid-lowering, antihypertensive, or antiplatelet therapies.35 Statins are widely used to reduce LDL cholesterol and have been proven to be safe in NAFLD, including for those with elevated liver enzymes and even in compensated cirrhosis, in several studies conducted during the last 15 years.36 Statins are characterized by anti-inflammatory, anti-oxidative, antifibrotic, and plaque-stabilizing effects, whereby they may improve vascular and hepatic function among patients with NAFLD and reduce cardiovascular risk.37 Statin use for the treatment of NAFLD is still controversial and off-label and is not specifically recommended to treat NASH, but positive results have been shown for reductions in liver enzymes.1 A recent meta-analysis of 13 studies showed that continued use of statin in cirrhosis was associated with a 46% and 44% risk reduction in hepatic decompensation and mortality, respectively.38
The Food and Drug Administration has approved omega-3 (n-3) fatty acid agents and fibrates for the treatment of very high triglycerides (500 mg/dL or higher); however, no specific indications exist to treat NAFLD.1 Fenofibrate is related to mild aminotransferase elevations and, in some cases, severe liver injury, so caution must be paid, especially within 2 days of taking the drug.39-40
NAFLD phenotypes that need liver pharmacotherapy
There are still no FDA-approved drugs or biological treatments for NASH. Pharmacological interventions aiming primarily at improving liver disease should generally be limited to those with biopsy-proven NASH and clinically significant fibrosis (fibrosis stages of 2 or greater).1,4 For FDA approval, medications used for treating NAFLD with fibrosis need to meet one of the following endpoint criteria: resolution of NASH without worsening of fibrosis, improvement in fibrosis without worsening of NASH, or both. In addition to those criteria, a new medication might improve the metabolic profile and have a tolerable safety profile. Table 1 displays those NAFLD phenotypes that will likely benefit from liver-directed therapy.

Obeticholic acid as an experimental therapy for NASH
A planned month-18 interim analysis of a multicentre, phase III RCT examined the efficacy and safety of obeticholic acid (OCA), a farnesoid X receptor agonist, in patients with NASH and stages 1-3 of fibrosis. The primary endpoint (fibrosis reduction 1 stage or more with no worsening of NASH) was met by 12% of patients in the placebo group, 18% of patients receiving OCA 10 mg (P = .045), and 23% of those receiving OCA 25 mg (P = .0002). An alternative primary endpoint of NASH resolution with no worsening of fibrosis was not met. OCA 25 mg led to the highest rates of pruritus and hyperlipidemia, compared with OCA 10 mg.42 These side effects seem to be related to the activation of the farnesoid X receptor.43
Currently available but off label medications
Vitamin E, an antioxidant, administered at a daily dose of 800 IU/day improves steatosis, inflammation, and ballooning, but not fibrosis in nondiabetic adults with biopsy-proven NASH.44 Vitamin E for 96 weeks was associated with a significantly higher rate of improvement in NASH (43% vs. 19%, P less than .01), compared with placebo.44 In the Treatment of Nonalcoholic Fatty Liver Disease in Children trial (TONIC), which examined vitamin E (800 IU/day) or metformin (500 mg twice daily) against placebo in children with biopsy-proven NAFLD, resolution of NASH was significantly greater in children treated with vitamin E than in children treated with placebo (58% vs. 28%, P less than .01). Metformin did not significantly improve the NASH resolution rates, compared with placebo (41% vs. 28%, P = .23). Vitamin E could be recommended for nondiabetic adults or children if lifestyle modifications do not produce the expected results as a result of noncompliance or ineffectiveness. Since continued use of vitamin E has been suggested to be associated with a very small increase in the risk for prostate cancer (an absolute increase of 1.6 per 1,000 person-years of vitamin E use) in men, risks and benefits should be discussed with each patient before starting therapy. A meta-analysis of nine placebo-controlled trials including roughly 119,000 patients reported that vitamin E supplementation increases the risk of hemorrhagic stroke by 20% while reducing ischemic stroke by 10%. It was estimated that vitamin E supplementation would prevent one ischemic stroke per 476 treated patients while inducing one hemorrhagic stroke for every 1,250 patients. It is noteworthy that the combination of vitamin E with anticoagulant and/or antiplatelet therapy was not examined in this trial, so we could not determine how combination therapy might affect the risk of ischemic or hemorrhagic stroke.45

Thiazolidinediones drugs have been reported to be effective in improving NAFLD in many human studies. Evidence from RCTs suggests that pioglitazone could significantly improve glucose metabolism, alanine aminotransferase, and liver histology – such as hepatic steatosis, lobular inflammation, and ballooning degeneration – among patients with or without T2D. However, the beneficial effects on improving fibrosis remain to be verified.1,46 Because of safety concerns, the risk/benefit balance of using pioglitazone to treat NASH should be discussed with each patient.47-48 Pioglitazone has been associated with long-term risk of bladder cancer,49 congestive heart failure,50 and bone fractures.51 Data from the Pioglitazone, Vitamin E, or Placebo for Nonalcoholic Steatohepatitis (PIVENS) trial showed that pioglitazone was significantly associated with weight gain but with no other serious adverse events. However, this study was not powered to test any safety-related hypotheses.44
Glucagon-like peptide 1 analogs have been reported to induce weight loss and reduce insulin resistance, which may lead to improvements in NAFLD. Phase II RCTs of glucagon-like peptide 1 receptor agonists (liraglutide and semaglutide) for the treatment of biopsy-proven NASH showed significant improvements in serum liver enzymes, steatosis, and inflammation, as well as NASH resolution without worsening liver fibrosis, although no direct benefit was observed in reversing fibrosis.52-53 One of these studies explores the efficacy and safety of different doses of daily subcutaneous semaglutide vs. placebo on the rates of resolution of NASH with no worsening of fibrosis. The highest dose (0.4 mg) showed the greatest difference (59% vs. 17%, P less than .01), compared with the placebo arm. However, there was no difference in improvement in fibrosis stage between the two groups (43% in the 0.4-mg group vs. 33% in the placebo group, P = .48).53 Gastrointestinal adverse events were common in the semaglutide arm.
“Spontaneous” NASH resolution and fibrosis improvement are commonly seen in participants assigned to placebo arms in clinical trials. A recent meta-analysis of 43 RCTs including 2649 placebo-treated patients showed a pooled estimate of NASH resolution without worsening of fibrosis and 1 stage reduction or more in fibrosis of 12% and 19%, respectively. Relevant factors involved in “spontaneous” NASH improvement are unknown but could be related to changes in BMI resulting from lifestyle changes, race and ethnicity, age, and, likely, NAFLD-related genetic variations, although more data is needed to better understand the histologic response in placebo-treated patients.54
Semaglutide injections (2.4 mg once weekly) or (2.0 mg once weekly) have been recently approved by the FDA for chronic weight management in adults with obesity or overweight with at least one weight-related condition or glucose control of T2D, respectively. Of note, the semaglutide dose used in the NASH trial is not currently available for the treatment of patients who are overweight/obese or have T2D, but the beneficial effects on body weight reductions and glucose control are similar overall to the effects seen with currently available doses for management of obesity or diabetes. One may consider using semaglutide in patients who are overweight/obese or have T2D with NASH, but in the senior author’s experience, it has been quite challenging to receive the payer’s approval, as its use is not specifically approved to treat liver disease.1
How to follow patients with NAFLD in the clinic
Once a diagnosis of NAFLD is made, the use of noninvasive testing may aid to identify which patients are at high risk of fibrosis. Easy to use clinical tools, such as the NAFLD Fibrosis Score and the Fib-4 index, and liver stiffness measurements using vibration-controlled transient elastography (FibroScan) or magnetic resonance elastography (MRE) are clinically useful noninvasive tools for identifying patients with NAFLD who have a higher likelihood of progressing to advanced fibrosis.1,55 The use of either NAFLD Fibrosis Score (less than -1.455) or Fib-4 index (less than 1.30) low cutoffs may be particularly useful to rule out advanced fibrosis. People with a NAFLD Fibrosis Score (greater than –1.455) or Fib-4 index (greater than 1.30) should undergo liver stiffness measurement (LSM) via FibroScan. Those with an LSM of 8 kPa or higher should be referred to specialized care, where a decision to perform a liver biopsy and initiate monitoring and therapy will be taken. MRE is the most accurate noninvasive method for the estimation of liver fibrosis. When MRE is available, it can be a diagnostic alternative to accurately rule in and rule out patients with advanced fibrosis. This technique can be preferred in clinical trials, but it is rarely used in clinical practice because it is expensive and not easily available. Reassessment by noninvasive scores at 1-3 years’ follow-up will be considered for those with an LSM less than 8 kPa. Patients with NASH cirrhosis should be screened for both gastroesophageal varix and HCC according to the American Association for the Study of Liver Diseases guidelines.56-57
Dr. Vilar-Gomez is assistant professor in the division of gastroenterology and hepatology at Indiana University, Indianapolis. Dr. Chalasani is vice president for academic affairs at Indiana University Health, Indianapolis, and the David W. Crabb Professor of Gastroenterology and Hepatology and an adjunct professor of anatomy, cell biology, and physiology in the division of gastroenterology and hepatology at Indiana University. Dr. Vilar-Gomez reports no financial conflicts of interest. Dr. Chalasani serves as a paid consultant to AbbVie, Boehringer-Ingelheim, Altimmune, Madrigal, Lilly, Zydus, and Galectin. He receives research support from Galectin and DSM.
References
1. Chalasani N et al. Hepatology 2018;67:328-57.
2. Söderberg C et al. Hepatology 2010;51:595-602.
3. Sanyal AJ et al. N Engl J Med 2021;385:1559-69.
4. Vilar-Gomez E et al. Gastroenterology 2018;155:443-57.e17.
5. Younossi ZM et al. Hepatology 2016;64:73-84.
6. EASL-EASD-EASO. J Hepatol 2016;64:1388-402.
7. Wong VW et al. J Hepatol 2018; 69:1349-56.
8. Vilar-Gomez E et al. Gastroenterology 2015;149:367-78.e5; quiz e14-5.
9. Promrat K et al. Hepatology 2010;51:121-9.
10. Wong VW et al. J Hepatol 2013;59:536-42.
11. Berzigotti A et al. Hepatology 2017;65:1293-1305.
12. Sacks FM et al. N Engl J Med 2009;360:859-73.
13. Vilar-Gomez E et al. Hepatology 2022 Jun;75(6):1491-1506.
14. Zelber-Sagi S et al. Liver Int 2017;37:936-49.
15. Hassani Zadeh S et al. J Gastroenterol Hepatol 2021;36:1470-8.
16. Yaskolka Meir A et al. Gut 2021;70:2085-95.
17. Sung KC et al. J Hepatol 2016;65:791-7.
18. Orci LA et al. Clin Gastroenterol Hepatol 2016;14:1398-411.
19. Ryu S et al. J Hepatol 2015;63:1229-37.
20. Kim D et al. Hepatology 2020;72:1556-68.
21. Kim D et al. Clin Gastroenterol Hepatol 2021;19:1240-7.e5.
22. Ascha MS et al. Hepatology 2010;51:1972-8.
23. Bambha K et al. Liver Int 2014;34:1250-8.
24. Lee Y et al. Clin Gastroenterol Hepatol 2019;17:1040-60.e11.
25. Grönroos S et al. JAMA Surg 2021;156:137-46.
26. Fakhry TK et al. Surg Obes Relat Dis 2019;15:502-11.
27. Seeberg KA et al. Ann Intern Med 2022;175:74-83.
28. Bower G et al. Obes Surg 2015;25:2280-9.
29. Jan A et al. Obes Surg 2015;25:1518-26.
30. Hanipah ZN et al. Obes Surg 2018;28:3431-8.
31. Are VS et al. Am J Gastroenterol 2020;115:1849-56.
32. Aminian A et al. JAMA 2021;326:2031-42.
33. Vuppalanchi R et al. Ann Surg 2022;275:e174-80.
34. Simon TG et al. Gut 2021. doi: 10.1136/gutjnl-2021-325724.
35. Lonardo A et al. J Hepatol 2018;68:335-52.
36. Chalasani N et al. Gastroenterology 2004;126:1287-92.
37. Pastori D et al. Dig Liver Dis 2015;47:4-11.
38. Kim RG et al. Clin Gastroenterol Hepatol 2017;15:1521-30.e8.
39. Ahmad J et al. Dig Dis Sci 2017;62:3596-604.
40. Chalasani NP et al. Am J Gastroenterol 2021;116(5):878-98.
41. Rinella ME et al. Hepatology 2019;70:1424-36.
42. Younossi ZM et al. Lancet 2019;394:2184-96.
43. Ratziu V. Clin Liver Dis (Hoboken) 2021;17:398-400.
44. Sanyal AJ et al. N Engl J Med 2010;341:1675-85.
45. Schürks M et al. BMJ 2010;341:c5702.
46. Cusi K et al. Ann Intern Med 2016;165:305-15.
47. Lewis JD et al. JAMA 2015;314:265-77.
48. Billington EO et al. Diabetologia 2015;58:2238-46.
49. Lewis JD et al. Diabetes Care 2011;34:916-22.
50. Erdmann E et al. Diabetes Care 2007;30:2773-8.
51. Viscoli CM et al. J Clin Endocrinol Metab 2017;102:914-22.
52. Armstong MJ et al. Lancet 2016;387:679-90.
53. Newsome PN et al. N Engl J Med 2021;384:1113-24.
54. Ng CH et al. Hepatology 2022;75:1647-61.
55. Kanwal F et al. Gastroenterology 2021;161:1030-1042.e8.
56. Garcia-Tsao G et al. Hepatology 2017;65:310-35.
57. Heimbach JK et al. Hepatology 2018;67:358-80.
Nonalcoholic fatty liver disease (NAFLD) is defined by the presence of hepatic steatosis detected on either imaging or histology in the absence of secondary causes of fatty liver (e.g., excessive alcohol consumption) or other chronic liver diseases.1 For practical NAFLD diagnosis purposes, excessive alcohol intake can be defined as an active or recent history of more than 21 standard drinks per week in men and more than14 standard drinks per week in women. For the sake of terminology, NAFLD is characterized by fatty liver infiltration, affecting at least 5% of hepatocytes, with no evidence of hepatocyte injury, whereas nonalcoholic steatohepatitis (NASH) is defined as the presence of necroinflammation with or without fibrosis in a background of fatty liver.1
Natural history
NASH and the degree of fibrosis are the two most important determinants of the natural history of NAFLD. NASH can evolve into fibrosis and cirrhosis, whereas advanced fibrosis and cirrhosis (stages 3 or 4 of fibrosis) significantly increase the risk of liver-related decompensation and mortality. NAFLD, per se, has been associated with an increased risk of overall mortality, compared with that of the general population.2 The three most common causes of mortality for patients with NAFLD are cardiovascular diseases (CVD), extrahepatic malignancies, and liver-related deaths. Mortality and liver-related events, including hepatic decompensation and hepatocellular carcinoma (HCC), may significantly increase in a dose-dependent manner with increasing fibrosis stages, and stages 3 or 4 of fibrosis may display the highest rates of all-cause mortality and liver-related events.3,4 It is important to note, however, that almost 15% of HCCs occur in patients with NAFLD who do not have cirrhosis.5 The presence of commonly associated comorbidities such as obesity, insulin resistance or diabetes, dyslipidemia, hypothyroidism, polycystic ovary syndrome, and sleep apnea may contribute to an increased risk of NASH and advanced fibrosis and, therefore, an accelerated clinical course of NAFLD.
Nonpharmacological interventions
Lifestyle modification
Lifestyle modification to achieve weight loss remains a first-line intervention in patients with NAFLD. Weight loss achieved either by hypocaloric diet alone or in conjunction with increased physical activity can be beneficial for all patients with NAFLD. The benefits extend not only to those who are overweight and obese but also to those within normal body weight (lean NAFLD).1,6,7 Weight loss of approximately 3%-5% is necessary to improve hepatic steatosis, but a greater weight loss (7%-10%) is required to improve other histopathological features like necroinflammatory lesions and fibrosis.8-10 Individuals with higher BMI and/or type 2 diabetes (T2D) will require a larger weight reduction to achieve a similar benefit on NAFLD-related features.7,8 Weight loss via lifestyle changes can also decrease hepatic venous pressure gradient (HVPG), with greater declines reported among those with more than 10% weight loss.11
Weight loss can be achieved through a variety of modalities, but long-term maintenance of lost weight is much more challenging. A combination of a hypocaloric diet with a caloric deficit of 500-1,000 kcal/d, alongside moderate-intensity exercise and intensive on-site behavioral treatment, will likely increase the possibility of a sustained weight loss over time.1,12 A growing body of scientific evidence indicates that a healthy diet that includes a reduction of high-glycemic-index foods and refined carbohydrates; increased consumption of monounsaturated fatty acids, omega-3 fatty acids, and fibers; and high intakes of olive oil, nuts, vegetables, fruits, legumes, whole grains, and fish can have beneficial effects on NAFLD and its severity.13-16 Adherence to these healthy dietary patterns has been associated with a marked reduction in CVD morbidity and mortality and is, thus, a strategic lifestyle recommendation for patients with NAFLD in whom the leading cause of morbidity and death is CVD.1,3
Exercise alone in adults with NAFLD may reduce hepatic steatosis, but its ability to improve inflammation and fibrosis has not been proven in well-designed RCTs.17,18 Physical activity and exercise have been shown to curb both the development and the progression of NAFLD, and beneficial effects could be achieved independent of weight loss.17,19,20 Most importantly, moderate-to-vigorous physical activity is likely associated with lower all-cause and cardiovascular mortality in patients with NAFLD.21
Heavy alcohol intake should be avoided by patients with NAFLD or NASH, and those with cirrhotic NASH should avoid any alcohol consumption given the risk of HCC and hepatic decompensation.1,4,22 Limiting light-to-moderate alcohol intake among patients without cirrhosis is still under debate.1 People with NAFLD may be advised to drink an equivalent of two to three 8-oz cups of regular brewed coffee daily as it has shown certain antifibrotic effects in NAFLD patients.23
Bariatric surgery
Bariatric surgery is an attractive therapeutic option for eligible obese patients with NAFLD. Bariatric surgery has the potential for inducing great weight loss and, therefore, reverses not only the steatosis, inflammation, and fibrosis among NAFLD individuals but also important comorbid conditions like T2D. A recent systematic review and meta-analysis examining data on the effects of bariatric surgery on histologic features of NAFLD from 32 cohort studies (no RCTs included) showed that bariatric surgery was associated with significant improvements in steatosis (66%), lobular inflammation (50%), ballooning degeneration (76%), and fibrosis (40%), and the benefits were significantly higher in those who underwent Roux-en-Y gastric bypass (RYGB). Of note, worsening of liver histology, including fibrosis, could be seen in up to 12% of patients who underwent bariatric surgery.24 The postsurgical weight regained after RYGB could explain partly the lack of fibrosis improvement or even worsening of fibrosis, although further research is needed to clarify these controversial findings.
RYGB and sleeve gastrectomy (SG) are the most commonly performed bariatric surgeries worldwide. Patients who undergo RYGB achieve higher weight loss when compared with those treated with SG.25 Among all bariatric procedures, RYGB could result in a higher proportion of complete resolution of NAFLD than SG, although evidence is inconclusive on fibrosis improvement rates.24,26 Most recently, a single-center RCT has compared the effects of RYGB vs. SG on liver fat content and fibrosis in patients with severe obesity and T2D.27 Data showed that both surgical procedures were highly and equally effective in reducing fatty liver content (quantified by magnetic resonance imaging), with an almost complete resolution of the fatty liver at 1 year of both surgical interventions. The beneficial effects of both GB and SG on fibrosis (assessed by enhanced liver test [ELF]) were less evident with no substantial difference between the two groups. Importantly, 69% of participants had an increase in their ELF scores during the study, despite the majority of participants achieving significant reductions in their body weights and better glycemic control at the end of the study. These findings might be considered with caution as several factors, such as the duration of the study (only 1 year) and lack of a liver biopsy to confirm fibrosis changes over time, could be influencing the study results.
Among all NAFLD phenotypes, those with cirrhosis and, most importantly, hepatic decompensation appear to be at increased risk of perioperative mortality and inpatient hospital stays than those without cirrhosis.28-29 Bariatric surgery is an absolute contraindication in patients with decompensated cirrhosis (Child B and Child C). Among compensated -Child A- cirrhotics, those with portal hypertension are at increased risk of morbidity and perioperative mortality.30 A recent analysis of National Inpatient Sample data suggested that the rates of complications in those with cirrhosis have decreased with time, which could be due to a better selection process and the use of more restrictive bariatric surgery in those with cirrhosis. Low volume centers (defined as less than 50 procedures per year) and nonrestrictive bariatric surgery were associated with a higher mortality rate. These data may suggest that patients with cirrhosis should undergo bariatric surgery only in high-volume centers after a multidisciplinary evaluation.31 Bariatric endoscopy is emerging as a new treatment for obesity, but the long-term durability of its effects remains to be determined.
A recent retrospective cohort study, including 1,158 adult patients with biopsy-proven NASH, has investigated the benefits of bariatric surgery on the occurrence of major adverse liver and cardiovascular outcomes in 650 patients who underwent bariatric surgery, compared with 508 patients who received nonsurgical usual care. This study showed that bariatric surgery was associated with 88% lower risk of progression of fatty liver to cirrhosis, liver cancer, or liver-related death, and 70% lower risk of serious CVD events during a follow-up period of 10 years.32 Within 1 year after surgery, 0.6% of patients died from surgical complications. The potential benefits of bariatric surgery in patients with NAFLD must be balanced against surgical risk, especially in eligible obese individuals with established cirrhosis. Data from a retrospective cohort study have shown that bariatric surgery in obese cirrhotic patients does not seem to associate with excessive mortality, compared with noncirrhotic obese patients.33 More data on immediate complication rates and long-term outcomes in patients with NAFLD by type of bariatric surgery is also required.
NAFLD as a standalone is not an indication for bariatric surgery. However, it could be considered in NAFLD patients who have a BMI of 40 kg/m2 or more without coexisting comorbidities or with a BMI of 35 kg/m2 or more and one or more severe obesity-related comorbidities, including T2D, hypertension, hyperlipidemia, or obstructive sleep apnea. Bariatric surgery must always be offered in centers with an experienced bariatric surgery program.1
Management of comorbidities
Given the multiple comorbidities associated with NAFLD and the potential to influence its severity, a comprehensive and multidisciplinary approach is needed to ameliorate not only the progression of liver disease but also those complications related to metabolic syndrome, hyperlipidemia, hypertension, diabetes, and other related conditions. Of note, all patients with NAFLD should receive aggressive management of comorbidities regardless of the severity of NAFLD. Ideally, a multidisciplinary team – including a primary care provider, an endocrinologist for patients with T2D, and a gastroenterologist/hepatologist – is needed to successfully manage patients with NAFLD.
It is well recognized that individuals with biopsy-proven NAFLD are at a higher risk of coronary heart disease, stroke, congestive heart failure, and death resulting from CVD when compared with the non-NAFLD population, and excess in CVD morbidity and mortality is evident across all stages of NAFLD and increases with worsening disease severity.34 The strong association between CVD and NAFLD has important clinical implications that may influence the decision to initiate treatment for primary prevention, including lipid-lowering, antihypertensive, or antiplatelet therapies.35 Statins are widely used to reduce LDL cholesterol and have been proven to be safe in NAFLD, including for those with elevated liver enzymes and even in compensated cirrhosis, in several studies conducted during the last 15 years.36 Statins are characterized by anti-inflammatory, anti-oxidative, antifibrotic, and plaque-stabilizing effects, whereby they may improve vascular and hepatic function among patients with NAFLD and reduce cardiovascular risk.37 Statin use for the treatment of NAFLD is still controversial and off-label and is not specifically recommended to treat NASH, but positive results have been shown for reductions in liver enzymes.1 A recent meta-analysis of 13 studies showed that continued use of statin in cirrhosis was associated with a 46% and 44% risk reduction in hepatic decompensation and mortality, respectively.38
The Food and Drug Administration has approved omega-3 (n-3) fatty acid agents and fibrates for the treatment of very high triglycerides (500 mg/dL or higher); however, no specific indications exist to treat NAFLD.1 Fenofibrate is related to mild aminotransferase elevations and, in some cases, severe liver injury, so caution must be paid, especially within 2 days of taking the drug.39-40
NAFLD phenotypes that need liver pharmacotherapy
There are still no FDA-approved drugs or biological treatments for NASH. Pharmacological interventions aiming primarily at improving liver disease should generally be limited to those with biopsy-proven NASH and clinically significant fibrosis (fibrosis stages of 2 or greater).1,4 For FDA approval, medications used for treating NAFLD with fibrosis need to meet one of the following endpoint criteria: resolution of NASH without worsening of fibrosis, improvement in fibrosis without worsening of NASH, or both. In addition to those criteria, a new medication might improve the metabolic profile and have a tolerable safety profile. Table 1 displays those NAFLD phenotypes that will likely benefit from liver-directed therapy.

Obeticholic acid as an experimental therapy for NASH
A planned month-18 interim analysis of a multicentre, phase III RCT examined the efficacy and safety of obeticholic acid (OCA), a farnesoid X receptor agonist, in patients with NASH and stages 1-3 of fibrosis. The primary endpoint (fibrosis reduction 1 stage or more with no worsening of NASH) was met by 12% of patients in the placebo group, 18% of patients receiving OCA 10 mg (P = .045), and 23% of those receiving OCA 25 mg (P = .0002). An alternative primary endpoint of NASH resolution with no worsening of fibrosis was not met. OCA 25 mg led to the highest rates of pruritus and hyperlipidemia, compared with OCA 10 mg.42 These side effects seem to be related to the activation of the farnesoid X receptor.43
Currently available but off label medications
Vitamin E, an antioxidant, administered at a daily dose of 800 IU/day improves steatosis, inflammation, and ballooning, but not fibrosis in nondiabetic adults with biopsy-proven NASH.44 Vitamin E for 96 weeks was associated with a significantly higher rate of improvement in NASH (43% vs. 19%, P less than .01), compared with placebo.44 In the Treatment of Nonalcoholic Fatty Liver Disease in Children trial (TONIC), which examined vitamin E (800 IU/day) or metformin (500 mg twice daily) against placebo in children with biopsy-proven NAFLD, resolution of NASH was significantly greater in children treated with vitamin E than in children treated with placebo (58% vs. 28%, P less than .01). Metformin did not significantly improve the NASH resolution rates, compared with placebo (41% vs. 28%, P = .23). Vitamin E could be recommended for nondiabetic adults or children if lifestyle modifications do not produce the expected results as a result of noncompliance or ineffectiveness. Since continued use of vitamin E has been suggested to be associated with a very small increase in the risk for prostate cancer (an absolute increase of 1.6 per 1,000 person-years of vitamin E use) in men, risks and benefits should be discussed with each patient before starting therapy. A meta-analysis of nine placebo-controlled trials including roughly 119,000 patients reported that vitamin E supplementation increases the risk of hemorrhagic stroke by 20% while reducing ischemic stroke by 10%. It was estimated that vitamin E supplementation would prevent one ischemic stroke per 476 treated patients while inducing one hemorrhagic stroke for every 1,250 patients. It is noteworthy that the combination of vitamin E with anticoagulant and/or antiplatelet therapy was not examined in this trial, so we could not determine how combination therapy might affect the risk of ischemic or hemorrhagic stroke.45

Thiazolidinediones drugs have been reported to be effective in improving NAFLD in many human studies. Evidence from RCTs suggests that pioglitazone could significantly improve glucose metabolism, alanine aminotransferase, and liver histology – such as hepatic steatosis, lobular inflammation, and ballooning degeneration – among patients with or without T2D. However, the beneficial effects on improving fibrosis remain to be verified.1,46 Because of safety concerns, the risk/benefit balance of using pioglitazone to treat NASH should be discussed with each patient.47-48 Pioglitazone has been associated with long-term risk of bladder cancer,49 congestive heart failure,50 and bone fractures.51 Data from the Pioglitazone, Vitamin E, or Placebo for Nonalcoholic Steatohepatitis (PIVENS) trial showed that pioglitazone was significantly associated with weight gain but with no other serious adverse events. However, this study was not powered to test any safety-related hypotheses.44
Glucagon-like peptide 1 analogs have been reported to induce weight loss and reduce insulin resistance, which may lead to improvements in NAFLD. Phase II RCTs of glucagon-like peptide 1 receptor agonists (liraglutide and semaglutide) for the treatment of biopsy-proven NASH showed significant improvements in serum liver enzymes, steatosis, and inflammation, as well as NASH resolution without worsening liver fibrosis, although no direct benefit was observed in reversing fibrosis.52-53 One of these studies explores the efficacy and safety of different doses of daily subcutaneous semaglutide vs. placebo on the rates of resolution of NASH with no worsening of fibrosis. The highest dose (0.4 mg) showed the greatest difference (59% vs. 17%, P less than .01), compared with the placebo arm. However, there was no difference in improvement in fibrosis stage between the two groups (43% in the 0.4-mg group vs. 33% in the placebo group, P = .48).53 Gastrointestinal adverse events were common in the semaglutide arm.
“Spontaneous” NASH resolution and fibrosis improvement are commonly seen in participants assigned to placebo arms in clinical trials. A recent meta-analysis of 43 RCTs including 2649 placebo-treated patients showed a pooled estimate of NASH resolution without worsening of fibrosis and 1 stage reduction or more in fibrosis of 12% and 19%, respectively. Relevant factors involved in “spontaneous” NASH improvement are unknown but could be related to changes in BMI resulting from lifestyle changes, race and ethnicity, age, and, likely, NAFLD-related genetic variations, although more data is needed to better understand the histologic response in placebo-treated patients.54
Semaglutide injections (2.4 mg once weekly) or (2.0 mg once weekly) have been recently approved by the FDA for chronic weight management in adults with obesity or overweight with at least one weight-related condition or glucose control of T2D, respectively. Of note, the semaglutide dose used in the NASH trial is not currently available for the treatment of patients who are overweight/obese or have T2D, but the beneficial effects on body weight reductions and glucose control are similar overall to the effects seen with currently available doses for management of obesity or diabetes. One may consider using semaglutide in patients who are overweight/obese or have T2D with NASH, but in the senior author’s experience, it has been quite challenging to receive the payer’s approval, as its use is not specifically approved to treat liver disease.1
How to follow patients with NAFLD in the clinic
Once a diagnosis of NAFLD is made, the use of noninvasive testing may aid to identify which patients are at high risk of fibrosis. Easy to use clinical tools, such as the NAFLD Fibrosis Score and the Fib-4 index, and liver stiffness measurements using vibration-controlled transient elastography (FibroScan) or magnetic resonance elastography (MRE) are clinically useful noninvasive tools for identifying patients with NAFLD who have a higher likelihood of progressing to advanced fibrosis.1,55 The use of either NAFLD Fibrosis Score (less than -1.455) or Fib-4 index (less than 1.30) low cutoffs may be particularly useful to rule out advanced fibrosis. People with a NAFLD Fibrosis Score (greater than –1.455) or Fib-4 index (greater than 1.30) should undergo liver stiffness measurement (LSM) via FibroScan. Those with an LSM of 8 kPa or higher should be referred to specialized care, where a decision to perform a liver biopsy and initiate monitoring and therapy will be taken. MRE is the most accurate noninvasive method for the estimation of liver fibrosis. When MRE is available, it can be a diagnostic alternative to accurately rule in and rule out patients with advanced fibrosis. This technique can be preferred in clinical trials, but it is rarely used in clinical practice because it is expensive and not easily available. Reassessment by noninvasive scores at 1-3 years’ follow-up will be considered for those with an LSM less than 8 kPa. Patients with NASH cirrhosis should be screened for both gastroesophageal varix and HCC according to the American Association for the Study of Liver Diseases guidelines.56-57
Dr. Vilar-Gomez is assistant professor in the division of gastroenterology and hepatology at Indiana University, Indianapolis. Dr. Chalasani is vice president for academic affairs at Indiana University Health, Indianapolis, and the David W. Crabb Professor of Gastroenterology and Hepatology and an adjunct professor of anatomy, cell biology, and physiology in the division of gastroenterology and hepatology at Indiana University. Dr. Vilar-Gomez reports no financial conflicts of interest. Dr. Chalasani serves as a paid consultant to AbbVie, Boehringer-Ingelheim, Altimmune, Madrigal, Lilly, Zydus, and Galectin. He receives research support from Galectin and DSM.
References
1. Chalasani N et al. Hepatology 2018;67:328-57.
2. Söderberg C et al. Hepatology 2010;51:595-602.
3. Sanyal AJ et al. N Engl J Med 2021;385:1559-69.
4. Vilar-Gomez E et al. Gastroenterology 2018;155:443-57.e17.
5. Younossi ZM et al. Hepatology 2016;64:73-84.
6. EASL-EASD-EASO. J Hepatol 2016;64:1388-402.
7. Wong VW et al. J Hepatol 2018; 69:1349-56.
8. Vilar-Gomez E et al. Gastroenterology 2015;149:367-78.e5; quiz e14-5.
9. Promrat K et al. Hepatology 2010;51:121-9.
10. Wong VW et al. J Hepatol 2013;59:536-42.
11. Berzigotti A et al. Hepatology 2017;65:1293-1305.
12. Sacks FM et al. N Engl J Med 2009;360:859-73.
13. Vilar-Gomez E et al. Hepatology 2022 Jun;75(6):1491-1506.
14. Zelber-Sagi S et al. Liver Int 2017;37:936-49.
15. Hassani Zadeh S et al. J Gastroenterol Hepatol 2021;36:1470-8.
16. Yaskolka Meir A et al. Gut 2021;70:2085-95.
17. Sung KC et al. J Hepatol 2016;65:791-7.
18. Orci LA et al. Clin Gastroenterol Hepatol 2016;14:1398-411.
19. Ryu S et al. J Hepatol 2015;63:1229-37.
20. Kim D et al. Hepatology 2020;72:1556-68.
21. Kim D et al. Clin Gastroenterol Hepatol 2021;19:1240-7.e5.
22. Ascha MS et al. Hepatology 2010;51:1972-8.
23. Bambha K et al. Liver Int 2014;34:1250-8.
24. Lee Y et al. Clin Gastroenterol Hepatol 2019;17:1040-60.e11.
25. Grönroos S et al. JAMA Surg 2021;156:137-46.
26. Fakhry TK et al. Surg Obes Relat Dis 2019;15:502-11.
27. Seeberg KA et al. Ann Intern Med 2022;175:74-83.
28. Bower G et al. Obes Surg 2015;25:2280-9.
29. Jan A et al. Obes Surg 2015;25:1518-26.
30. Hanipah ZN et al. Obes Surg 2018;28:3431-8.
31. Are VS et al. Am J Gastroenterol 2020;115:1849-56.
32. Aminian A et al. JAMA 2021;326:2031-42.
33. Vuppalanchi R et al. Ann Surg 2022;275:e174-80.
34. Simon TG et al. Gut 2021. doi: 10.1136/gutjnl-2021-325724.
35. Lonardo A et al. J Hepatol 2018;68:335-52.
36. Chalasani N et al. Gastroenterology 2004;126:1287-92.
37. Pastori D et al. Dig Liver Dis 2015;47:4-11.
38. Kim RG et al. Clin Gastroenterol Hepatol 2017;15:1521-30.e8.
39. Ahmad J et al. Dig Dis Sci 2017;62:3596-604.
40. Chalasani NP et al. Am J Gastroenterol 2021;116(5):878-98.
41. Rinella ME et al. Hepatology 2019;70:1424-36.
42. Younossi ZM et al. Lancet 2019;394:2184-96.
43. Ratziu V. Clin Liver Dis (Hoboken) 2021;17:398-400.
44. Sanyal AJ et al. N Engl J Med 2010;341:1675-85.
45. Schürks M et al. BMJ 2010;341:c5702.
46. Cusi K et al. Ann Intern Med 2016;165:305-15.
47. Lewis JD et al. JAMA 2015;314:265-77.
48. Billington EO et al. Diabetologia 2015;58:2238-46.
49. Lewis JD et al. Diabetes Care 2011;34:916-22.
50. Erdmann E et al. Diabetes Care 2007;30:2773-8.
51. Viscoli CM et al. J Clin Endocrinol Metab 2017;102:914-22.
52. Armstong MJ et al. Lancet 2016;387:679-90.
53. Newsome PN et al. N Engl J Med 2021;384:1113-24.
54. Ng CH et al. Hepatology 2022;75:1647-61.
55. Kanwal F et al. Gastroenterology 2021;161:1030-1042.e8.
56. Garcia-Tsao G et al. Hepatology 2017;65:310-35.
57. Heimbach JK et al. Hepatology 2018;67:358-80.
Novel liquid biopsy may identify NASH, fibrosis
A novel liquid biopsy test, which uses two circulating proteins, appears to be effective for diagnosing two major liver conditions, according to a new study published in Gut.
The test could allow clinicians to determine the staging of both liver fibrosis and nonalcoholic steatohepatitis, wrote the researchers led by Giulia Angelini, PhD, a postdoctoral fellow focused on nonalcoholic fatty liver disease pathophysiology at the Catholic University of the Sacred Heart in Rome.
“The diagnosis of nonalcoholic steatohepatitis (NASH) currently relies on invasive liver biopsy,” they wrote. “There is, therefore, an urgent need to find noninvasive biomarkers for NASH diagnosis, disease progression, and intervention response monitoring.”
The research team sought to identify a biomarker and algorithm able to predict the presence and severity of nonalcoholic steatohepatitis (NASH) or liver fibrosis. The study evaluated two proteins found in circulating monocytes, which are a type of white blood cell: PLIN2 as a predictor of histological NASH and RAB14 levels as a predictor of liver fibrosis.
The multicenter study included 250 patients, with 100 subjects in the discovery cohort from the Bariatric Surgery Versus Nonalcoholic Steatohepatitis trial, or BRAVES, and 150 subjects in the validation cohort from the Liquid Biopsy for NASH and Liver Fibrosis trial, or LIBRA. The patients had histologically proven nonalcoholic fatty liver disease or NASH with or without fibrosis.
After careful molecular analysis, the research team used neural network classifiers to predict the presence of NASH and NASH stages. The analysis for the prediction of the presence of NASH produced an accuracy of 93% in the discovery cohort and 92% in the validation cohort. Sensitivity and specificity were 95% and 90% in the discovery group and 88% and 100% in the validation group, respectively. The research team also used a neural network analysis to predict the stages of NASH, which showed an accuracy of 85% in the discovery group and 85.2% in the validation cohort.
RAB14 was used to predict liver fibrosis with a logistic model that included waist circumference, age, plasma glucose, high-density lipoprotein, and alanine aminotransferase. In the discovery group, accuracy was 99.2%, sensitivity was 100%, and specificity was 95.8%. In the validation group, accuracy was 97.6%, sensitivity was 99%, and specificity was 89.6%.
When RAB14 was used as the only variable in the model, the accuracy, sensitivity, and specificity in the discovery cohort were 86.4%, 96%, and 45.8%, respectively. In the validation cohort, they were 92.4%, 96.9%, and 34.5%, respectively. In both cohorts, half of the subjects without fibrosis were erroneously predicted as having fibrosis, but the diagnosis of fibrosis was correctly predicted in nearly all subjects.
A limitation of the study is that only White subjects were enrolled, which limits the generalizability to other racial/ethnic groups, the investigators wrote, although they don’t expect differences would be seen in other groups.
“PLIN2 and RAB14 may permit diagnosis of NASH and/or liver fibrosis with a simple blood test,” they wrote. “Our biomarkers can be used in community and population studies permitting to investigate the real prevalence of NASH and liver fibrosis. Moreover, since it requires only blood sampling, they are potentially valuable tools for population-based and prevention studies in children.”
A step forward
“Obesity is a silent pandemic with an expected prevalence rate that will exceed 50% globally by 2030, of which 25% of the adults have fatty liver and approximately 6.5% with NASH, a progressive form of fatty liver,” said Kalyan Ram Bhamidimarri, MD, chief of hepatology and associate professor of clinical medicine at the University of Miami, who was not involved in the research. “Liver biopsy is the current clinical standard to diagnose NASH, but relying on an invasive procedure like liver biopsy that is fraught with several risks in a consistently growing volume of individuals with obesity is unsustainable.
“So, there is an unmet need to diagnose NASH without invasive procedures such as liver biopsy,” he said. He pointed out that many of the alternatives to liver biopsy, such as liver stiffness measurements and scoring systems, pose their own difficulties.
On the other hand, he noted that “blood-based tests that correlate well with liver biopsy, the so-called wet biomarkers or liquid liver biopsy, are easier to perform, accessed widely, and could be tested frequently to assess efficacy of therapies.”
The study was funded by Elucidating Pathways of Steatohepatitis (EPOS Horizon 2020), Stratification of Obese Phenotypes to Optimize Future Obesity Therapy (SOPHIA IMI), Metadeq Inc., and support from the Transcampus Initiative. The study authors declared various competing interests, including some who serve as an advisor or stock option holder for Metadeq Limited. Dr. Bhamidimarri reported no relevant conflicts of interest.
Help your patients understand their risks for NASH by sharing AGA patient education at www.gastro.org/NASH.
A novel liquid biopsy test, which uses two circulating proteins, appears to be effective for diagnosing two major liver conditions, according to a new study published in Gut.
The test could allow clinicians to determine the staging of both liver fibrosis and nonalcoholic steatohepatitis, wrote the researchers led by Giulia Angelini, PhD, a postdoctoral fellow focused on nonalcoholic fatty liver disease pathophysiology at the Catholic University of the Sacred Heart in Rome.
“The diagnosis of nonalcoholic steatohepatitis (NASH) currently relies on invasive liver biopsy,” they wrote. “There is, therefore, an urgent need to find noninvasive biomarkers for NASH diagnosis, disease progression, and intervention response monitoring.”
The research team sought to identify a biomarker and algorithm able to predict the presence and severity of nonalcoholic steatohepatitis (NASH) or liver fibrosis. The study evaluated two proteins found in circulating monocytes, which are a type of white blood cell: PLIN2 as a predictor of histological NASH and RAB14 levels as a predictor of liver fibrosis.
The multicenter study included 250 patients, with 100 subjects in the discovery cohort from the Bariatric Surgery Versus Nonalcoholic Steatohepatitis trial, or BRAVES, and 150 subjects in the validation cohort from the Liquid Biopsy for NASH and Liver Fibrosis trial, or LIBRA. The patients had histologically proven nonalcoholic fatty liver disease or NASH with or without fibrosis.
After careful molecular analysis, the research team used neural network classifiers to predict the presence of NASH and NASH stages. The analysis for the prediction of the presence of NASH produced an accuracy of 93% in the discovery cohort and 92% in the validation cohort. Sensitivity and specificity were 95% and 90% in the discovery group and 88% and 100% in the validation group, respectively. The research team also used a neural network analysis to predict the stages of NASH, which showed an accuracy of 85% in the discovery group and 85.2% in the validation cohort.
RAB14 was used to predict liver fibrosis with a logistic model that included waist circumference, age, plasma glucose, high-density lipoprotein, and alanine aminotransferase. In the discovery group, accuracy was 99.2%, sensitivity was 100%, and specificity was 95.8%. In the validation group, accuracy was 97.6%, sensitivity was 99%, and specificity was 89.6%.
When RAB14 was used as the only variable in the model, the accuracy, sensitivity, and specificity in the discovery cohort were 86.4%, 96%, and 45.8%, respectively. In the validation cohort, they were 92.4%, 96.9%, and 34.5%, respectively. In both cohorts, half of the subjects without fibrosis were erroneously predicted as having fibrosis, but the diagnosis of fibrosis was correctly predicted in nearly all subjects.
A limitation of the study is that only White subjects were enrolled, which limits the generalizability to other racial/ethnic groups, the investigators wrote, although they don’t expect differences would be seen in other groups.
“PLIN2 and RAB14 may permit diagnosis of NASH and/or liver fibrosis with a simple blood test,” they wrote. “Our biomarkers can be used in community and population studies permitting to investigate the real prevalence of NASH and liver fibrosis. Moreover, since it requires only blood sampling, they are potentially valuable tools for population-based and prevention studies in children.”
A step forward
“Obesity is a silent pandemic with an expected prevalence rate that will exceed 50% globally by 2030, of which 25% of the adults have fatty liver and approximately 6.5% with NASH, a progressive form of fatty liver,” said Kalyan Ram Bhamidimarri, MD, chief of hepatology and associate professor of clinical medicine at the University of Miami, who was not involved in the research. “Liver biopsy is the current clinical standard to diagnose NASH, but relying on an invasive procedure like liver biopsy that is fraught with several risks in a consistently growing volume of individuals with obesity is unsustainable.
“So, there is an unmet need to diagnose NASH without invasive procedures such as liver biopsy,” he said. He pointed out that many of the alternatives to liver biopsy, such as liver stiffness measurements and scoring systems, pose their own difficulties.
On the other hand, he noted that “blood-based tests that correlate well with liver biopsy, the so-called wet biomarkers or liquid liver biopsy, are easier to perform, accessed widely, and could be tested frequently to assess efficacy of therapies.”
The study was funded by Elucidating Pathways of Steatohepatitis (EPOS Horizon 2020), Stratification of Obese Phenotypes to Optimize Future Obesity Therapy (SOPHIA IMI), Metadeq Inc., and support from the Transcampus Initiative. The study authors declared various competing interests, including some who serve as an advisor or stock option holder for Metadeq Limited. Dr. Bhamidimarri reported no relevant conflicts of interest.
Help your patients understand their risks for NASH by sharing AGA patient education at www.gastro.org/NASH.
A novel liquid biopsy test, which uses two circulating proteins, appears to be effective for diagnosing two major liver conditions, according to a new study published in Gut.
The test could allow clinicians to determine the staging of both liver fibrosis and nonalcoholic steatohepatitis, wrote the researchers led by Giulia Angelini, PhD, a postdoctoral fellow focused on nonalcoholic fatty liver disease pathophysiology at the Catholic University of the Sacred Heart in Rome.
“The diagnosis of nonalcoholic steatohepatitis (NASH) currently relies on invasive liver biopsy,” they wrote. “There is, therefore, an urgent need to find noninvasive biomarkers for NASH diagnosis, disease progression, and intervention response monitoring.”
The research team sought to identify a biomarker and algorithm able to predict the presence and severity of nonalcoholic steatohepatitis (NASH) or liver fibrosis. The study evaluated two proteins found in circulating monocytes, which are a type of white blood cell: PLIN2 as a predictor of histological NASH and RAB14 levels as a predictor of liver fibrosis.
The multicenter study included 250 patients, with 100 subjects in the discovery cohort from the Bariatric Surgery Versus Nonalcoholic Steatohepatitis trial, or BRAVES, and 150 subjects in the validation cohort from the Liquid Biopsy for NASH and Liver Fibrosis trial, or LIBRA. The patients had histologically proven nonalcoholic fatty liver disease or NASH with or without fibrosis.
After careful molecular analysis, the research team used neural network classifiers to predict the presence of NASH and NASH stages. The analysis for the prediction of the presence of NASH produced an accuracy of 93% in the discovery cohort and 92% in the validation cohort. Sensitivity and specificity were 95% and 90% in the discovery group and 88% and 100% in the validation group, respectively. The research team also used a neural network analysis to predict the stages of NASH, which showed an accuracy of 85% in the discovery group and 85.2% in the validation cohort.
RAB14 was used to predict liver fibrosis with a logistic model that included waist circumference, age, plasma glucose, high-density lipoprotein, and alanine aminotransferase. In the discovery group, accuracy was 99.2%, sensitivity was 100%, and specificity was 95.8%. In the validation group, accuracy was 97.6%, sensitivity was 99%, and specificity was 89.6%.
When RAB14 was used as the only variable in the model, the accuracy, sensitivity, and specificity in the discovery cohort were 86.4%, 96%, and 45.8%, respectively. In the validation cohort, they were 92.4%, 96.9%, and 34.5%, respectively. In both cohorts, half of the subjects without fibrosis were erroneously predicted as having fibrosis, but the diagnosis of fibrosis was correctly predicted in nearly all subjects.
A limitation of the study is that only White subjects were enrolled, which limits the generalizability to other racial/ethnic groups, the investigators wrote, although they don’t expect differences would be seen in other groups.
“PLIN2 and RAB14 may permit diagnosis of NASH and/or liver fibrosis with a simple blood test,” they wrote. “Our biomarkers can be used in community and population studies permitting to investigate the real prevalence of NASH and liver fibrosis. Moreover, since it requires only blood sampling, they are potentially valuable tools for population-based and prevention studies in children.”
A step forward
“Obesity is a silent pandemic with an expected prevalence rate that will exceed 50% globally by 2030, of which 25% of the adults have fatty liver and approximately 6.5% with NASH, a progressive form of fatty liver,” said Kalyan Ram Bhamidimarri, MD, chief of hepatology and associate professor of clinical medicine at the University of Miami, who was not involved in the research. “Liver biopsy is the current clinical standard to diagnose NASH, but relying on an invasive procedure like liver biopsy that is fraught with several risks in a consistently growing volume of individuals with obesity is unsustainable.
“So, there is an unmet need to diagnose NASH without invasive procedures such as liver biopsy,” he said. He pointed out that many of the alternatives to liver biopsy, such as liver stiffness measurements and scoring systems, pose their own difficulties.
On the other hand, he noted that “blood-based tests that correlate well with liver biopsy, the so-called wet biomarkers or liquid liver biopsy, are easier to perform, accessed widely, and could be tested frequently to assess efficacy of therapies.”
The study was funded by Elucidating Pathways of Steatohepatitis (EPOS Horizon 2020), Stratification of Obese Phenotypes to Optimize Future Obesity Therapy (SOPHIA IMI), Metadeq Inc., and support from the Transcampus Initiative. The study authors declared various competing interests, including some who serve as an advisor or stock option holder for Metadeq Limited. Dr. Bhamidimarri reported no relevant conflicts of interest.
Help your patients understand their risks for NASH by sharing AGA patient education at www.gastro.org/NASH.
FROM GUT
Alcohol-related cirrhosis associated with higher risk of fractures, death
Patients with alcohol-related cirrhosis have a higher fracture rate and a higher post-fracture mortality rate, compared with the general population, according to a large new study from Sweden.
Alcohol-related cirrhosis was associated with an almost fourfold increased fracture rate, and the post-fracture mortality rates were higher at both 30 days and 1 year later.
“Half of all fractures were presumably associated with osteoporosis,” write the study authors, who are gastroenterologists and epidemiologists at the Karolinska Institute, Stockholm. “This suggests that existing pharmacotherapy for osteoporosis may reduce the fracture risk in patients with alcohol-related cirrhosis and possibly also reduce mortality rates.”
But, the authors continue, “our data indicate that osteoporosis may not be the only explanatory factor for this increased fracture risk. Removing modifiable risk factors such as smoking, heavy alcohol use, or malnutrition may further reduce the risk of fractures.”
The study was published online in Clinical Gastroenterology and Hepatology.
Analyzing risks
The association between liver cirrhosis and fractures appears strongest in patients with alcohol-related cirrhosis, the most common cause of cirrhosis in many countries, including Sweden, the authors write.
Previous studies have examined mostly relative risk or hip fractures. The authors aimed to determine not only the relative risk but also the absolute risk, which “can better inform clinicians and policymakers of the actual size of the problem,” they write.
In a nationwide population-based cohort study, they analyzed data from the Swedish National Patient Registry between 1969 and 2016, which included 25,090 patients with alcohol-related cirrhosis. Patients were matched for sex, age, and municipality with 239,458 controls from the Swedish Total Population Registry. They calculated the cumulative incidence of fractures and accounted for competing risks, such as death or liver transportation.
Overall, 48,635 fractures occurred during 3.4 million person-years of follow-up, including 3,659 (14.6%) among patients and 44,976 (18.8%) among controls.
Patients with alcohol-related cirrhosis had a 3.8-times higher fracture rate, with 38.7 fractures per 1,000 person-years, compared with 13.3 in controls. Alcohol-related cirrhosis was also associated with a 1.9-times higher fracture rate than nonalcoholic cirrhosis and a 1.3-times higher fracture rate than noncirrhotic alcohol-related liver disease.
The cumulative incidence of fractures was elevated for patients with alcohol-related cirrhosis in the first 19 years of follow-up, with a 5-year risk at nearly 10%, compared with 4.5% for controls, and a 10-year risk of 13.5%, compared with 8.7% for controls.
Among those with a fracture, the median time to death was 2.8 years in patients with alcohol-related cirrhosis and 3.5 years in controls.
Patients with alcohol-related cirrhosis had a 1.6-times higher post-fracture mortality rate at 30 days, as well as a 1.8-times higher post-fracture mortality rate after one year.
“Falls and fractures kill patients with cirrhosis. Data like these are crucial to spread awareness and represent a call to arms,” Elliot Tapper, MD, an assistant professor of gastroenterology at the University of Michigan, Ann Arbor, told this news organization.
Dr. Tapper, who wasn’t involved with this study, researches the health outcomes of patients with cirrhosis. His previous studies have found that falls, injuries, and death are common in patients with cirrhosis, which could be predicted with an algorithm based on a prior history of falls, blood sodium level, mobility, and quality of life.
“The data emphasize that a fall and fracture herald a time of increased risk,” he said. “Research is needed to develop interventions that prevent falls and help patients remain more resilient when they happen.”
Promoting bone health
Osteoporosis was the most common presumed mechanism in both patients with alcohol-related cirrhosis (49.4%) and controls (52.2%), while high-energy trauma from motor vehicle crashes or heights preceded 10.9% of fractures in patients and 13.5% in controls.
The Karolinska Institute study found that patients with alcohol-related cirrhosis had a 4.4-times higher rate of osteoporotic fracture than controls, which remained 3.6-times higher when using a stricter definition of osteoporotic fracture (a diagnosis of osteoporosis before, at, or within 3 months from the date of a fracture of the vertebrae, pelvis, proximal humerus, distal forearm, or hip).
Patients with osteoporosis at baseline had a 2.5-times higher incidence of fractures than controls with baseline osteoporosis. The absolute risk of fractures in patients with alcohol-related cirrhosis and osteoporosis was higher than for controls with osteoporosis during the first 3 years after a cirrhosis diagnosis.
In addition, the post-fracture mortality rate in those with osteoporosis was more than double in patients with cirrhosis in the first 30 days after a fracture and more than tripled after one year.
“Bone health isn’t necessarily prioritized for our patients, even though it is linked to higher mortality and disability,” Arpan Patel, MD, PhD, a hepatologist at the West Los Angeles VA Medical Center and assistant professor at the David Geffen School of Medicine at the University of California, Los Angeles, told this news organization.
Dr. Patel, who wasn’t involved with this study, has researched the associations between osteoporotic fracture risk, hospitalization, and death in patients with cirrhosis.
“Current guidelines support assessing post-liver transplant patients for bone density but do not currently advocate for doing so in patients with cirrhosis or alcohol-associated liver disease, who are a much larger at-risk population,” Dr. Patel said.
“The current paper supports the idea that we should consider the broad ramifications of alcohol use on bone health for our patients and suggests that there should be greater efforts to screen for and manage osteoporosis and osteopenia in our patients earlier,” he added.
The researchers were supported by grants from Region Stockholm and the Syskonen Svensson Foundation, though the funders had no role in the conduct of the study. The study authors reported no other disclosures or conflicts of interest. Dr. Tapper and Dr. Patel report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients with alcohol-related cirrhosis have a higher fracture rate and a higher post-fracture mortality rate, compared with the general population, according to a large new study from Sweden.
Alcohol-related cirrhosis was associated with an almost fourfold increased fracture rate, and the post-fracture mortality rates were higher at both 30 days and 1 year later.
“Half of all fractures were presumably associated with osteoporosis,” write the study authors, who are gastroenterologists and epidemiologists at the Karolinska Institute, Stockholm. “This suggests that existing pharmacotherapy for osteoporosis may reduce the fracture risk in patients with alcohol-related cirrhosis and possibly also reduce mortality rates.”
But, the authors continue, “our data indicate that osteoporosis may not be the only explanatory factor for this increased fracture risk. Removing modifiable risk factors such as smoking, heavy alcohol use, or malnutrition may further reduce the risk of fractures.”
The study was published online in Clinical Gastroenterology and Hepatology.
Analyzing risks
The association between liver cirrhosis and fractures appears strongest in patients with alcohol-related cirrhosis, the most common cause of cirrhosis in many countries, including Sweden, the authors write.
Previous studies have examined mostly relative risk or hip fractures. The authors aimed to determine not only the relative risk but also the absolute risk, which “can better inform clinicians and policymakers of the actual size of the problem,” they write.
In a nationwide population-based cohort study, they analyzed data from the Swedish National Patient Registry between 1969 and 2016, which included 25,090 patients with alcohol-related cirrhosis. Patients were matched for sex, age, and municipality with 239,458 controls from the Swedish Total Population Registry. They calculated the cumulative incidence of fractures and accounted for competing risks, such as death or liver transportation.
Overall, 48,635 fractures occurred during 3.4 million person-years of follow-up, including 3,659 (14.6%) among patients and 44,976 (18.8%) among controls.
Patients with alcohol-related cirrhosis had a 3.8-times higher fracture rate, with 38.7 fractures per 1,000 person-years, compared with 13.3 in controls. Alcohol-related cirrhosis was also associated with a 1.9-times higher fracture rate than nonalcoholic cirrhosis and a 1.3-times higher fracture rate than noncirrhotic alcohol-related liver disease.
The cumulative incidence of fractures was elevated for patients with alcohol-related cirrhosis in the first 19 years of follow-up, with a 5-year risk at nearly 10%, compared with 4.5% for controls, and a 10-year risk of 13.5%, compared with 8.7% for controls.
Among those with a fracture, the median time to death was 2.8 years in patients with alcohol-related cirrhosis and 3.5 years in controls.
Patients with alcohol-related cirrhosis had a 1.6-times higher post-fracture mortality rate at 30 days, as well as a 1.8-times higher post-fracture mortality rate after one year.
“Falls and fractures kill patients with cirrhosis. Data like these are crucial to spread awareness and represent a call to arms,” Elliot Tapper, MD, an assistant professor of gastroenterology at the University of Michigan, Ann Arbor, told this news organization.
Dr. Tapper, who wasn’t involved with this study, researches the health outcomes of patients with cirrhosis. His previous studies have found that falls, injuries, and death are common in patients with cirrhosis, which could be predicted with an algorithm based on a prior history of falls, blood sodium level, mobility, and quality of life.
“The data emphasize that a fall and fracture herald a time of increased risk,” he said. “Research is needed to develop interventions that prevent falls and help patients remain more resilient when they happen.”
Promoting bone health
Osteoporosis was the most common presumed mechanism in both patients with alcohol-related cirrhosis (49.4%) and controls (52.2%), while high-energy trauma from motor vehicle crashes or heights preceded 10.9% of fractures in patients and 13.5% in controls.
The Karolinska Institute study found that patients with alcohol-related cirrhosis had a 4.4-times higher rate of osteoporotic fracture than controls, which remained 3.6-times higher when using a stricter definition of osteoporotic fracture (a diagnosis of osteoporosis before, at, or within 3 months from the date of a fracture of the vertebrae, pelvis, proximal humerus, distal forearm, or hip).
Patients with osteoporosis at baseline had a 2.5-times higher incidence of fractures than controls with baseline osteoporosis. The absolute risk of fractures in patients with alcohol-related cirrhosis and osteoporosis was higher than for controls with osteoporosis during the first 3 years after a cirrhosis diagnosis.
In addition, the post-fracture mortality rate in those with osteoporosis was more than double in patients with cirrhosis in the first 30 days after a fracture and more than tripled after one year.
“Bone health isn’t necessarily prioritized for our patients, even though it is linked to higher mortality and disability,” Arpan Patel, MD, PhD, a hepatologist at the West Los Angeles VA Medical Center and assistant professor at the David Geffen School of Medicine at the University of California, Los Angeles, told this news organization.
Dr. Patel, who wasn’t involved with this study, has researched the associations between osteoporotic fracture risk, hospitalization, and death in patients with cirrhosis.
“Current guidelines support assessing post-liver transplant patients for bone density but do not currently advocate for doing so in patients with cirrhosis or alcohol-associated liver disease, who are a much larger at-risk population,” Dr. Patel said.
“The current paper supports the idea that we should consider the broad ramifications of alcohol use on bone health for our patients and suggests that there should be greater efforts to screen for and manage osteoporosis and osteopenia in our patients earlier,” he added.
The researchers were supported by grants from Region Stockholm and the Syskonen Svensson Foundation, though the funders had no role in the conduct of the study. The study authors reported no other disclosures or conflicts of interest. Dr. Tapper and Dr. Patel report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients with alcohol-related cirrhosis have a higher fracture rate and a higher post-fracture mortality rate, compared with the general population, according to a large new study from Sweden.
Alcohol-related cirrhosis was associated with an almost fourfold increased fracture rate, and the post-fracture mortality rates were higher at both 30 days and 1 year later.
“Half of all fractures were presumably associated with osteoporosis,” write the study authors, who are gastroenterologists and epidemiologists at the Karolinska Institute, Stockholm. “This suggests that existing pharmacotherapy for osteoporosis may reduce the fracture risk in patients with alcohol-related cirrhosis and possibly also reduce mortality rates.”
But, the authors continue, “our data indicate that osteoporosis may not be the only explanatory factor for this increased fracture risk. Removing modifiable risk factors such as smoking, heavy alcohol use, or malnutrition may further reduce the risk of fractures.”
The study was published online in Clinical Gastroenterology and Hepatology.
Analyzing risks
The association between liver cirrhosis and fractures appears strongest in patients with alcohol-related cirrhosis, the most common cause of cirrhosis in many countries, including Sweden, the authors write.
Previous studies have examined mostly relative risk or hip fractures. The authors aimed to determine not only the relative risk but also the absolute risk, which “can better inform clinicians and policymakers of the actual size of the problem,” they write.
In a nationwide population-based cohort study, they analyzed data from the Swedish National Patient Registry between 1969 and 2016, which included 25,090 patients with alcohol-related cirrhosis. Patients were matched for sex, age, and municipality with 239,458 controls from the Swedish Total Population Registry. They calculated the cumulative incidence of fractures and accounted for competing risks, such as death or liver transportation.
Overall, 48,635 fractures occurred during 3.4 million person-years of follow-up, including 3,659 (14.6%) among patients and 44,976 (18.8%) among controls.
Patients with alcohol-related cirrhosis had a 3.8-times higher fracture rate, with 38.7 fractures per 1,000 person-years, compared with 13.3 in controls. Alcohol-related cirrhosis was also associated with a 1.9-times higher fracture rate than nonalcoholic cirrhosis and a 1.3-times higher fracture rate than noncirrhotic alcohol-related liver disease.
The cumulative incidence of fractures was elevated for patients with alcohol-related cirrhosis in the first 19 years of follow-up, with a 5-year risk at nearly 10%, compared with 4.5% for controls, and a 10-year risk of 13.5%, compared with 8.7% for controls.
Among those with a fracture, the median time to death was 2.8 years in patients with alcohol-related cirrhosis and 3.5 years in controls.
Patients with alcohol-related cirrhosis had a 1.6-times higher post-fracture mortality rate at 30 days, as well as a 1.8-times higher post-fracture mortality rate after one year.
“Falls and fractures kill patients with cirrhosis. Data like these are crucial to spread awareness and represent a call to arms,” Elliot Tapper, MD, an assistant professor of gastroenterology at the University of Michigan, Ann Arbor, told this news organization.
Dr. Tapper, who wasn’t involved with this study, researches the health outcomes of patients with cirrhosis. His previous studies have found that falls, injuries, and death are common in patients with cirrhosis, which could be predicted with an algorithm based on a prior history of falls, blood sodium level, mobility, and quality of life.
“The data emphasize that a fall and fracture herald a time of increased risk,” he said. “Research is needed to develop interventions that prevent falls and help patients remain more resilient when they happen.”
Promoting bone health
Osteoporosis was the most common presumed mechanism in both patients with alcohol-related cirrhosis (49.4%) and controls (52.2%), while high-energy trauma from motor vehicle crashes or heights preceded 10.9% of fractures in patients and 13.5% in controls.
The Karolinska Institute study found that patients with alcohol-related cirrhosis had a 4.4-times higher rate of osteoporotic fracture than controls, which remained 3.6-times higher when using a stricter definition of osteoporotic fracture (a diagnosis of osteoporosis before, at, or within 3 months from the date of a fracture of the vertebrae, pelvis, proximal humerus, distal forearm, or hip).
Patients with osteoporosis at baseline had a 2.5-times higher incidence of fractures than controls with baseline osteoporosis. The absolute risk of fractures in patients with alcohol-related cirrhosis and osteoporosis was higher than for controls with osteoporosis during the first 3 years after a cirrhosis diagnosis.
In addition, the post-fracture mortality rate in those with osteoporosis was more than double in patients with cirrhosis in the first 30 days after a fracture and more than tripled after one year.
“Bone health isn’t necessarily prioritized for our patients, even though it is linked to higher mortality and disability,” Arpan Patel, MD, PhD, a hepatologist at the West Los Angeles VA Medical Center and assistant professor at the David Geffen School of Medicine at the University of California, Los Angeles, told this news organization.
Dr. Patel, who wasn’t involved with this study, has researched the associations between osteoporotic fracture risk, hospitalization, and death in patients with cirrhosis.
“Current guidelines support assessing post-liver transplant patients for bone density but do not currently advocate for doing so in patients with cirrhosis or alcohol-associated liver disease, who are a much larger at-risk population,” Dr. Patel said.
“The current paper supports the idea that we should consider the broad ramifications of alcohol use on bone health for our patients and suggests that there should be greater efforts to screen for and manage osteoporosis and osteopenia in our patients earlier,” he added.
The researchers were supported by grants from Region Stockholm and the Syskonen Svensson Foundation, though the funders had no role in the conduct of the study. The study authors reported no other disclosures or conflicts of interest. Dr. Tapper and Dr. Patel report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
New update focuses on NAFLD in lean people
Ongoing follow-up and lifestyle interventions are needed in lean patients with nonalcoholic fatty liver disease (NAFLD), suggests a panel of experts in a recent review.
They also urge screening for NAFLD in individuals who are older than 40 years with type 2 diabetes, even if they are not overweight.
NAFLD is a leading cause of chronic liver disease that affects more than 25% of the United States and worldwide populations, note lead author Michelle T. Long, MD, Boston Medical Center, Boston University, and colleagues.
They add that around one-quarter of those affected have nonalcoholic steatohepatitis, which is associated with significant morbidity and mortality due to complications of liver cirrhosis, hepatic decompensation, and hepatocellular carcinoma.
Although NAFLD occurs primarily in individuals with obesity or type 2 diabetes, between 7%-20% have a lean body habitus, they write.
There are differences in rates of disease progression, associated conditions, and diagnostic and management approaches between lean and non-lean patients, the authors note, but there is limited guidance on the appropriate clinical evaluation of the former group.
The American Gastroenterological Association therefore commissioned an expert review to provide best practice advice on key clinical issues relating to the diagnosis, risk stratification, and treatment of NAFLD in lean individuals.
Their review was published online in Gastroenterology.
Evidence-based approaches
The 15 best practice advice statements covered a wide range of clinical areas, first defining lean as a body mass index (BMI) less than 25 in non-Asian persons and less than 23 in Asian persons.
The authors go on to stipulate, for example, that lean individuals in the general population should not be screened for NAFLD but that screening should be considered for individuals older than 40 years with type 2 diabetes.
More broadly, they write that the condition should be considered in lean individuals with metabolic diseases, such as type 2 diabetes, dyslipidemia, and hypertension, as well as elevated values on liver biochemical tests or incidentally noted hepatic steatosis.
After other causes of liver diseases are ruled out, the authors note that clinicians should consider liver biopsy as the reference test if uncertainties remain about liver injury causes and/or liver fibrosis staging.
They also write that the NAFLD fibrosis score and Fibrosis-4 score, along with imaging techniques, may be used as alternatives to biopsy for staging and during follow-up.
The authors, who provide a diagnosis and management algorithm to aid clinicians, suggest that lean patients with NAFLD follow lifestyle interventions, such as exercise, diet modification, and avoidance of fructose- and sugar-sweetened drinks, to achieve weight loss of 3%-5%.
Vitamin E may be considered, they continue, in patients with biopsy-confirmed nonalcoholic steatohepatitis but without type 2 diabetes or cirrhosis. Additionally, oral pioglitazone may be considered in lean persons with biopsy-confirmed nonalcoholic steatohepatitis without cirrhosis.
In contrast, they write that the role of glucagonlike peptide 1 agonists and sodium-glucose cotransporter 2 inhibitors requires further investigation.
The advice also says that lean patients with NAFLD should be routinely evaluated for comorbid conditions, such as type 2 diabetes, dyslipidemia, and hypertension, and risk-stratified for hepatic fibrosis to identify those with advanced fibrosis or cirrhosis.
For lean patients with NAFLD and clinical markers compatible with liver cirrhosis, twice-yearly surveillance for hepatocellular carcinoma is also advised.
Fatty liver disease in lean people with metabolic conditions
Approached for comment, Liyun Yuan, MD, PhD, assistant professor of clinical medicine, University of Southern California, Los Angeles, said it is very important to have uniform guidelines for general practitioners and other specialties on NAFLD in lean individuals.
Dr. Yuan, who was not involved in the review, told this news organization that it is crucial to raise awareness of NAFLD, just like awareness of breast cancer screening among women of a certain age was increased, so that individuals are screened for metabolic conditions regardless of whether they have obesity or overweight.
Zobair Younossi, MD, MPH, professor of medicine, Virginia Commonwealth University, Inova Campus, Falls Church, Va., added that there is a lack of awareness that NAFLD occurs in lean individuals, especially in those who have diabetes.
He said in an interview that although it is accurate to define individuals as being lean in terms of their BMI, the best way is to look not only at BMI but also at waist circumference.
Dr. Younossi said that he and his colleagues have shown that when BMI is combined with waist circumference, the prediction of mortality risk in NAFLD is affected, such that lean individuals with an obese waist circumference have a higher risk for all-cause mortality.
Dr. Long is supported in part by the National Institute of Diabetes and Digestive and Kidney Diseases, Doris Duke Charitable Foundation, Gilead Sciences Research Scholars Award, Boston University School of Medicine Department of Medicine Career Investment Award, and Boston University Clinical Translational Science Institute. Dr. Long declares relationships with Novo Nordisk, Echosens Corporation, and Gilead Sciences. Dr. Yuan declares relationships with Genfit, Intercept, and Gilead Sciences. Dr. Younossi declares no relevant relationships.
A version of this article first appeared on Medscape.com.
*This article was updated on July 27, 2022.
Ongoing follow-up and lifestyle interventions are needed in lean patients with nonalcoholic fatty liver disease (NAFLD), suggests a panel of experts in a recent review.
They also urge screening for NAFLD in individuals who are older than 40 years with type 2 diabetes, even if they are not overweight.
NAFLD is a leading cause of chronic liver disease that affects more than 25% of the United States and worldwide populations, note lead author Michelle T. Long, MD, Boston Medical Center, Boston University, and colleagues.
They add that around one-quarter of those affected have nonalcoholic steatohepatitis, which is associated with significant morbidity and mortality due to complications of liver cirrhosis, hepatic decompensation, and hepatocellular carcinoma.
Although NAFLD occurs primarily in individuals with obesity or type 2 diabetes, between 7%-20% have a lean body habitus, they write.
There are differences in rates of disease progression, associated conditions, and diagnostic and management approaches between lean and non-lean patients, the authors note, but there is limited guidance on the appropriate clinical evaluation of the former group.
The American Gastroenterological Association therefore commissioned an expert review to provide best practice advice on key clinical issues relating to the diagnosis, risk stratification, and treatment of NAFLD in lean individuals.
Their review was published online in Gastroenterology.
Evidence-based approaches
The 15 best practice advice statements covered a wide range of clinical areas, first defining lean as a body mass index (BMI) less than 25 in non-Asian persons and less than 23 in Asian persons.
The authors go on to stipulate, for example, that lean individuals in the general population should not be screened for NAFLD but that screening should be considered for individuals older than 40 years with type 2 diabetes.
More broadly, they write that the condition should be considered in lean individuals with metabolic diseases, such as type 2 diabetes, dyslipidemia, and hypertension, as well as elevated values on liver biochemical tests or incidentally noted hepatic steatosis.
After other causes of liver diseases are ruled out, the authors note that clinicians should consider liver biopsy as the reference test if uncertainties remain about liver injury causes and/or liver fibrosis staging.
They also write that the NAFLD fibrosis score and Fibrosis-4 score, along with imaging techniques, may be used as alternatives to biopsy for staging and during follow-up.
The authors, who provide a diagnosis and management algorithm to aid clinicians, suggest that lean patients with NAFLD follow lifestyle interventions, such as exercise, diet modification, and avoidance of fructose- and sugar-sweetened drinks, to achieve weight loss of 3%-5%.
Vitamin E may be considered, they continue, in patients with biopsy-confirmed nonalcoholic steatohepatitis but without type 2 diabetes or cirrhosis. Additionally, oral pioglitazone may be considered in lean persons with biopsy-confirmed nonalcoholic steatohepatitis without cirrhosis.
In contrast, they write that the role of glucagonlike peptide 1 agonists and sodium-glucose cotransporter 2 inhibitors requires further investigation.
The advice also says that lean patients with NAFLD should be routinely evaluated for comorbid conditions, such as type 2 diabetes, dyslipidemia, and hypertension, and risk-stratified for hepatic fibrosis to identify those with advanced fibrosis or cirrhosis.
For lean patients with NAFLD and clinical markers compatible with liver cirrhosis, twice-yearly surveillance for hepatocellular carcinoma is also advised.
Fatty liver disease in lean people with metabolic conditions
Approached for comment, Liyun Yuan, MD, PhD, assistant professor of clinical medicine, University of Southern California, Los Angeles, said it is very important to have uniform guidelines for general practitioners and other specialties on NAFLD in lean individuals.
Dr. Yuan, who was not involved in the review, told this news organization that it is crucial to raise awareness of NAFLD, just like awareness of breast cancer screening among women of a certain age was increased, so that individuals are screened for metabolic conditions regardless of whether they have obesity or overweight.
Zobair Younossi, MD, MPH, professor of medicine, Virginia Commonwealth University, Inova Campus, Falls Church, Va., added that there is a lack of awareness that NAFLD occurs in lean individuals, especially in those who have diabetes.
He said in an interview that although it is accurate to define individuals as being lean in terms of their BMI, the best way is to look not only at BMI but also at waist circumference.
Dr. Younossi said that he and his colleagues have shown that when BMI is combined with waist circumference, the prediction of mortality risk in NAFLD is affected, such that lean individuals with an obese waist circumference have a higher risk for all-cause mortality.
Dr. Long is supported in part by the National Institute of Diabetes and Digestive and Kidney Diseases, Doris Duke Charitable Foundation, Gilead Sciences Research Scholars Award, Boston University School of Medicine Department of Medicine Career Investment Award, and Boston University Clinical Translational Science Institute. Dr. Long declares relationships with Novo Nordisk, Echosens Corporation, and Gilead Sciences. Dr. Yuan declares relationships with Genfit, Intercept, and Gilead Sciences. Dr. Younossi declares no relevant relationships.
A version of this article first appeared on Medscape.com.
*This article was updated on July 27, 2022.
Ongoing follow-up and lifestyle interventions are needed in lean patients with nonalcoholic fatty liver disease (NAFLD), suggests a panel of experts in a recent review.
They also urge screening for NAFLD in individuals who are older than 40 years with type 2 diabetes, even if they are not overweight.
NAFLD is a leading cause of chronic liver disease that affects more than 25% of the United States and worldwide populations, note lead author Michelle T. Long, MD, Boston Medical Center, Boston University, and colleagues.
They add that around one-quarter of those affected have nonalcoholic steatohepatitis, which is associated with significant morbidity and mortality due to complications of liver cirrhosis, hepatic decompensation, and hepatocellular carcinoma.
Although NAFLD occurs primarily in individuals with obesity or type 2 diabetes, between 7%-20% have a lean body habitus, they write.
There are differences in rates of disease progression, associated conditions, and diagnostic and management approaches between lean and non-lean patients, the authors note, but there is limited guidance on the appropriate clinical evaluation of the former group.
The American Gastroenterological Association therefore commissioned an expert review to provide best practice advice on key clinical issues relating to the diagnosis, risk stratification, and treatment of NAFLD in lean individuals.
Their review was published online in Gastroenterology.
Evidence-based approaches
The 15 best practice advice statements covered a wide range of clinical areas, first defining lean as a body mass index (BMI) less than 25 in non-Asian persons and less than 23 in Asian persons.
The authors go on to stipulate, for example, that lean individuals in the general population should not be screened for NAFLD but that screening should be considered for individuals older than 40 years with type 2 diabetes.
More broadly, they write that the condition should be considered in lean individuals with metabolic diseases, such as type 2 diabetes, dyslipidemia, and hypertension, as well as elevated values on liver biochemical tests or incidentally noted hepatic steatosis.
After other causes of liver diseases are ruled out, the authors note that clinicians should consider liver biopsy as the reference test if uncertainties remain about liver injury causes and/or liver fibrosis staging.
They also write that the NAFLD fibrosis score and Fibrosis-4 score, along with imaging techniques, may be used as alternatives to biopsy for staging and during follow-up.
The authors, who provide a diagnosis and management algorithm to aid clinicians, suggest that lean patients with NAFLD follow lifestyle interventions, such as exercise, diet modification, and avoidance of fructose- and sugar-sweetened drinks, to achieve weight loss of 3%-5%.
Vitamin E may be considered, they continue, in patients with biopsy-confirmed nonalcoholic steatohepatitis but without type 2 diabetes or cirrhosis. Additionally, oral pioglitazone may be considered in lean persons with biopsy-confirmed nonalcoholic steatohepatitis without cirrhosis.
In contrast, they write that the role of glucagonlike peptide 1 agonists and sodium-glucose cotransporter 2 inhibitors requires further investigation.
The advice also says that lean patients with NAFLD should be routinely evaluated for comorbid conditions, such as type 2 diabetes, dyslipidemia, and hypertension, and risk-stratified for hepatic fibrosis to identify those with advanced fibrosis or cirrhosis.
For lean patients with NAFLD and clinical markers compatible with liver cirrhosis, twice-yearly surveillance for hepatocellular carcinoma is also advised.
Fatty liver disease in lean people with metabolic conditions
Approached for comment, Liyun Yuan, MD, PhD, assistant professor of clinical medicine, University of Southern California, Los Angeles, said it is very important to have uniform guidelines for general practitioners and other specialties on NAFLD in lean individuals.
Dr. Yuan, who was not involved in the review, told this news organization that it is crucial to raise awareness of NAFLD, just like awareness of breast cancer screening among women of a certain age was increased, so that individuals are screened for metabolic conditions regardless of whether they have obesity or overweight.
Zobair Younossi, MD, MPH, professor of medicine, Virginia Commonwealth University, Inova Campus, Falls Church, Va., added that there is a lack of awareness that NAFLD occurs in lean individuals, especially in those who have diabetes.
He said in an interview that although it is accurate to define individuals as being lean in terms of their BMI, the best way is to look not only at BMI but also at waist circumference.
Dr. Younossi said that he and his colleagues have shown that when BMI is combined with waist circumference, the prediction of mortality risk in NAFLD is affected, such that lean individuals with an obese waist circumference have a higher risk for all-cause mortality.
Dr. Long is supported in part by the National Institute of Diabetes and Digestive and Kidney Diseases, Doris Duke Charitable Foundation, Gilead Sciences Research Scholars Award, Boston University School of Medicine Department of Medicine Career Investment Award, and Boston University Clinical Translational Science Institute. Dr. Long declares relationships with Novo Nordisk, Echosens Corporation, and Gilead Sciences. Dr. Yuan declares relationships with Genfit, Intercept, and Gilead Sciences. Dr. Younossi declares no relevant relationships.
A version of this article first appeared on Medscape.com.
*This article was updated on July 27, 2022.
FROM GASTROENTEROLOGY
‘Alarming’ global rise in NAFLD
The global prevalence of fatty liver disease not caused by alcohol is considerably higher than previously estimated and is continuing to increase at an alarming rate, report researchers from Canada.
Their analysis suggests nearly one-third of the global general adult population has nonalcoholic fatty liver disease (NAFLD), with men much more likely to have the disease than women.
“Greater awareness of NAFLD and the development of cost-effective risk stratification strategies are needed to address the growing burden NAFLD,” wrote Abdel-Aziz Shaheen, MBBCh, MSc, and colleagues with the University of Calgary (Alta.).
The study was published online in Lancet Gastroenterology and Hepatology.
NAFLD is the most common liver disease worldwide and a leading cause of liver-related illness and death. Yet, high-quality reports on the epidemiology of NAFLD at a global level are scarce and temporal trends of the NAFLD burden, including by gender, have not been described, until now.
Using MEDLINE, EMBASE, Scopus, and Web of Science, the Calgary team identified reports on NAFLD incidence and prevalence in study populations representative of the general adult population published between the date of database inception to May 25, 2021.
In total, 72 publications, with a sample population of more than 1 million adults from 17 countries, were included in the prevalence analysis, and 16 publications, with a sample population of nearly 382,000 individuals from five countries, were included in the incidence analysis.
By their estimates, the overall global prevalence of NAFLD is 32.4%, with prevalence increasing steadily and significantly over time, from 25.5% in or before 2005 to 37.8% in 2016 or later. The overall prevalence is significantly higher in men than in women (39.7% vs. 25.6%).
These figures contrast with recent meta-analyses and systematic reviews that put the global prevalence of NAFLD at between 25.2% and 29.8%. However, these studies had “considerable” limitations with “potentially biased inferences,” Dr. Shaheen and colleagues noted.
By region, their data put the prevalence of NAFLD at 31.6% in Asia, 32.6% in Europe, 47.8% in North America, and 56.8% in Africa.
Dr. Shaheen and colleagues estimate the overall incidence of NAFLD to be 46.9 cases per 1,000 person-years, with a higher incidence in men than women (70.8 vs. 29.6 cases per 1000 person-years), in line with the gender differences in prevalence.
They caution that there was “considerable” heterogeneity between studies in both NAFLD prevalence and incidence (I2 = 99.9%) and few “high-quality” studies.
Despite these limitations, Dr. Shaheen and colleagues said the rise in NAFLD prevalence “should drive enhanced awareness of NAFLD at the level of primary care physicians, public health specialists, and health policy makers to encourage the development of more effective preventive policies.”
Funding for the study was provided by the Canadian Institutes of Health. Dr. Shaheen has received research grants from Gilead and Intercept, and honoraria from SCOPE Canada.
A version of this article first appeared on Medscape.com.
The global prevalence of fatty liver disease not caused by alcohol is considerably higher than previously estimated and is continuing to increase at an alarming rate, report researchers from Canada.
Their analysis suggests nearly one-third of the global general adult population has nonalcoholic fatty liver disease (NAFLD), with men much more likely to have the disease than women.
“Greater awareness of NAFLD and the development of cost-effective risk stratification strategies are needed to address the growing burden NAFLD,” wrote Abdel-Aziz Shaheen, MBBCh, MSc, and colleagues with the University of Calgary (Alta.).
The study was published online in Lancet Gastroenterology and Hepatology.
NAFLD is the most common liver disease worldwide and a leading cause of liver-related illness and death. Yet, high-quality reports on the epidemiology of NAFLD at a global level are scarce and temporal trends of the NAFLD burden, including by gender, have not been described, until now.
Using MEDLINE, EMBASE, Scopus, and Web of Science, the Calgary team identified reports on NAFLD incidence and prevalence in study populations representative of the general adult population published between the date of database inception to May 25, 2021.
In total, 72 publications, with a sample population of more than 1 million adults from 17 countries, were included in the prevalence analysis, and 16 publications, with a sample population of nearly 382,000 individuals from five countries, were included in the incidence analysis.
By their estimates, the overall global prevalence of NAFLD is 32.4%, with prevalence increasing steadily and significantly over time, from 25.5% in or before 2005 to 37.8% in 2016 or later. The overall prevalence is significantly higher in men than in women (39.7% vs. 25.6%).
These figures contrast with recent meta-analyses and systematic reviews that put the global prevalence of NAFLD at between 25.2% and 29.8%. However, these studies had “considerable” limitations with “potentially biased inferences,” Dr. Shaheen and colleagues noted.
By region, their data put the prevalence of NAFLD at 31.6% in Asia, 32.6% in Europe, 47.8% in North America, and 56.8% in Africa.
Dr. Shaheen and colleagues estimate the overall incidence of NAFLD to be 46.9 cases per 1,000 person-years, with a higher incidence in men than women (70.8 vs. 29.6 cases per 1000 person-years), in line with the gender differences in prevalence.
They caution that there was “considerable” heterogeneity between studies in both NAFLD prevalence and incidence (I2 = 99.9%) and few “high-quality” studies.
Despite these limitations, Dr. Shaheen and colleagues said the rise in NAFLD prevalence “should drive enhanced awareness of NAFLD at the level of primary care physicians, public health specialists, and health policy makers to encourage the development of more effective preventive policies.”
Funding for the study was provided by the Canadian Institutes of Health. Dr. Shaheen has received research grants from Gilead and Intercept, and honoraria from SCOPE Canada.
A version of this article first appeared on Medscape.com.
The global prevalence of fatty liver disease not caused by alcohol is considerably higher than previously estimated and is continuing to increase at an alarming rate, report researchers from Canada.
Their analysis suggests nearly one-third of the global general adult population has nonalcoholic fatty liver disease (NAFLD), with men much more likely to have the disease than women.
“Greater awareness of NAFLD and the development of cost-effective risk stratification strategies are needed to address the growing burden NAFLD,” wrote Abdel-Aziz Shaheen, MBBCh, MSc, and colleagues with the University of Calgary (Alta.).
The study was published online in Lancet Gastroenterology and Hepatology.
NAFLD is the most common liver disease worldwide and a leading cause of liver-related illness and death. Yet, high-quality reports on the epidemiology of NAFLD at a global level are scarce and temporal trends of the NAFLD burden, including by gender, have not been described, until now.
Using MEDLINE, EMBASE, Scopus, and Web of Science, the Calgary team identified reports on NAFLD incidence and prevalence in study populations representative of the general adult population published between the date of database inception to May 25, 2021.
In total, 72 publications, with a sample population of more than 1 million adults from 17 countries, were included in the prevalence analysis, and 16 publications, with a sample population of nearly 382,000 individuals from five countries, were included in the incidence analysis.
By their estimates, the overall global prevalence of NAFLD is 32.4%, with prevalence increasing steadily and significantly over time, from 25.5% in or before 2005 to 37.8% in 2016 or later. The overall prevalence is significantly higher in men than in women (39.7% vs. 25.6%).
These figures contrast with recent meta-analyses and systematic reviews that put the global prevalence of NAFLD at between 25.2% and 29.8%. However, these studies had “considerable” limitations with “potentially biased inferences,” Dr. Shaheen and colleagues noted.
By region, their data put the prevalence of NAFLD at 31.6% in Asia, 32.6% in Europe, 47.8% in North America, and 56.8% in Africa.
Dr. Shaheen and colleagues estimate the overall incidence of NAFLD to be 46.9 cases per 1,000 person-years, with a higher incidence in men than women (70.8 vs. 29.6 cases per 1000 person-years), in line with the gender differences in prevalence.
They caution that there was “considerable” heterogeneity between studies in both NAFLD prevalence and incidence (I2 = 99.9%) and few “high-quality” studies.
Despite these limitations, Dr. Shaheen and colleagues said the rise in NAFLD prevalence “should drive enhanced awareness of NAFLD at the level of primary care physicians, public health specialists, and health policy makers to encourage the development of more effective preventive policies.”
Funding for the study was provided by the Canadian Institutes of Health. Dr. Shaheen has received research grants from Gilead and Intercept, and honoraria from SCOPE Canada.
A version of this article first appeared on Medscape.com.
FROM LANCET GASTROENTEROLOGY AND HEPATOLOGY
Prior decompensation in alcohol-associated hepatitis not an ‘absolute contraindication’ for early liver transplant
Past decompensation in alcohol-associated hepatitis may be linked with worse survival following liver transplantation, but it’s not all bad news, according to a retrospective study.
Traditionally, patients with alcoholic liver disease were asked to be alcohol free for 6 months before consideration for a liver transplantation. In recent years, there’s been a loosening of that policy, with physicians considering “early” liver transplantation (early LT) instead of waiting 6 months. “It became obvious that a lot of patients do resume alcohol use after transplant, and most of them don’t appear to suffer too much in the way of adverse consequences,” said Paul Martin, MD, chief of hepatology at the University of Miami, who was not involved in the current research.
In 2011, a study confirmed that suspicion, finding that 6-month survival was 77% among carefully selected patients with alcohol-associated hepatitis for whom the 6-month sobriety requirement was waived; 6-month survival in those who did not receive a transplant was 22%. The selection criteria included the presence of supportive family members, the absence of severe coexisting conditions, and a commitment to abstaining from alcohol.
However, authors of the current study, published in the American Journal of Gastroenterology sought nuance: The appropriateness of prior decompensation as exclusion criteria in published studies is unknown, so the researchers compared outcomes of patients with prior versus first-time liver decompensation in alcohol-associated hepatitis.
Not all bad news
The study included 241 patients from six sites who consecutively received early LT between 2007 and 2020. Among these, 210 were identified as having a first-time liver decompensation event and 31 as having had a prior history of liver decompensation, defined as being diagnosed with ascites, hepatic encephalopathy, variceal bleeding, or jaundice.
There was no significant difference in median age, Model for End-Stage Liver Disease (MELD) scores, or post–liver transplant follow-up time between those with first-time liver decompensation or a prior history. The unadjusted 1-year survival rate was 93% in the first decompensation group (95% confidence interval, 89%-96%) and 86% in the prior decompensation group (95% CI, 66%-94%). The unadjusted 3-year survival rates were 85% (95% CI, 79%-90%) and 78% (95% CI, 57%-89%), respectively.
Importantly, the researchers found an association between prior decompensation and higher adjusted post–liver transplantation mortality (adjusted hazard ratio, 2.72; 95% CI, 1.61-4.59) and harmful alcohol use (aHR, 1.77; 95% CI, 1.07-2.92).
However, the researchers noted that these patients, who had MELD scores of 39 and previous decompensation, were at exceptionally high risk of short-term mortality, but still had 1- and 3-year survival rates above 85% and 75%, respectively, with early LT. “While longer follow-up is desirable as graft failure related to alcohol is most apparent after 5-years post LT, these results suggest that prior decompensation alone should not be considered an absolute contraindication to early LT.”
Limitations of the study included its retrospective data and small sample size for patients with prior decompensation.
“These findings validate the value of the ‘first decompensation’ criteria in published experiences regarding early LT for [alcoholic hepatitis],” the investigators concluded. “Further larger and prospective studies with longer-term follow-up will be needed to assess ways to optimally select patients in this cohort who may benefit most from early LT, and ways to manage patients at highest risk for worse outcomes post LT.”
A note of caution for early LT
About half of all liver mortality is attributable to alcoholic-associated liver disease. Corticosteroids can improve short-term survival, but there are no medications proven to increase long-term survival. That leaves liver transplant as the sole alternative for patients who don’t respond to corticosteroids.
“Programs in North America have liberalized their acceptance criteria for patients with alcoholic liver disease, and that’s resulted in large numbers of patients being transplanted who have less than 6 months abstinence. And overall, the results seem good, but I think this paper strikes an appropriate note of caution. In essence, if a patient had at least one prior episode of liver failure related to alcoholic excess and had recovered from that, and continued to drink and got into trouble again, [and then] presented for consideration for liver transplantation, the fact that they resumed alcohol use after prior episodes of decompensation suggests that they may be less-than-ideal candidates [for liver transplantation],” said Dr. Martin.
He pointed out important caveats to the study, including its retrospective nature and its inclusion of a relatively small number of patients with a history of liver decompensation. But it reinforces what physicians generally know, which is that some patients with severe alcohol use disorder also have liver failure, and they tend to fare worse than others after a liver transplant.
Still, physicians also face a conundrum because there are increasing numbers of younger patients who won’t survive if they don’t get a liver transplant. “The challenge is picking out patients who are going to be good candidates from a purely medical point of view, but have a low likelihood of resuming alcohol use after transplantation [which could injure] the new liver,” said Dr. Martin. The new study has the potential to provide some additional guidance in patient selection.
The study authors disclosed no relevant conflicts of interest. Dr. Martin has no relevant financial disclosures.
Past decompensation in alcohol-associated hepatitis may be linked with worse survival following liver transplantation, but it’s not all bad news, according to a retrospective study.
Traditionally, patients with alcoholic liver disease were asked to be alcohol free for 6 months before consideration for a liver transplantation. In recent years, there’s been a loosening of that policy, with physicians considering “early” liver transplantation (early LT) instead of waiting 6 months. “It became obvious that a lot of patients do resume alcohol use after transplant, and most of them don’t appear to suffer too much in the way of adverse consequences,” said Paul Martin, MD, chief of hepatology at the University of Miami, who was not involved in the current research.
In 2011, a study confirmed that suspicion, finding that 6-month survival was 77% among carefully selected patients with alcohol-associated hepatitis for whom the 6-month sobriety requirement was waived; 6-month survival in those who did not receive a transplant was 22%. The selection criteria included the presence of supportive family members, the absence of severe coexisting conditions, and a commitment to abstaining from alcohol.
However, authors of the current study, published in the American Journal of Gastroenterology sought nuance: The appropriateness of prior decompensation as exclusion criteria in published studies is unknown, so the researchers compared outcomes of patients with prior versus first-time liver decompensation in alcohol-associated hepatitis.
Not all bad news
The study included 241 patients from six sites who consecutively received early LT between 2007 and 2020. Among these, 210 were identified as having a first-time liver decompensation event and 31 as having had a prior history of liver decompensation, defined as being diagnosed with ascites, hepatic encephalopathy, variceal bleeding, or jaundice.
There was no significant difference in median age, Model for End-Stage Liver Disease (MELD) scores, or post–liver transplant follow-up time between those with first-time liver decompensation or a prior history. The unadjusted 1-year survival rate was 93% in the first decompensation group (95% confidence interval, 89%-96%) and 86% in the prior decompensation group (95% CI, 66%-94%). The unadjusted 3-year survival rates were 85% (95% CI, 79%-90%) and 78% (95% CI, 57%-89%), respectively.
Importantly, the researchers found an association between prior decompensation and higher adjusted post–liver transplantation mortality (adjusted hazard ratio, 2.72; 95% CI, 1.61-4.59) and harmful alcohol use (aHR, 1.77; 95% CI, 1.07-2.92).
However, the researchers noted that these patients, who had MELD scores of 39 and previous decompensation, were at exceptionally high risk of short-term mortality, but still had 1- and 3-year survival rates above 85% and 75%, respectively, with early LT. “While longer follow-up is desirable as graft failure related to alcohol is most apparent after 5-years post LT, these results suggest that prior decompensation alone should not be considered an absolute contraindication to early LT.”
Limitations of the study included its retrospective data and small sample size for patients with prior decompensation.
“These findings validate the value of the ‘first decompensation’ criteria in published experiences regarding early LT for [alcoholic hepatitis],” the investigators concluded. “Further larger and prospective studies with longer-term follow-up will be needed to assess ways to optimally select patients in this cohort who may benefit most from early LT, and ways to manage patients at highest risk for worse outcomes post LT.”
A note of caution for early LT
About half of all liver mortality is attributable to alcoholic-associated liver disease. Corticosteroids can improve short-term survival, but there are no medications proven to increase long-term survival. That leaves liver transplant as the sole alternative for patients who don’t respond to corticosteroids.
“Programs in North America have liberalized their acceptance criteria for patients with alcoholic liver disease, and that’s resulted in large numbers of patients being transplanted who have less than 6 months abstinence. And overall, the results seem good, but I think this paper strikes an appropriate note of caution. In essence, if a patient had at least one prior episode of liver failure related to alcoholic excess and had recovered from that, and continued to drink and got into trouble again, [and then] presented for consideration for liver transplantation, the fact that they resumed alcohol use after prior episodes of decompensation suggests that they may be less-than-ideal candidates [for liver transplantation],” said Dr. Martin.
He pointed out important caveats to the study, including its retrospective nature and its inclusion of a relatively small number of patients with a history of liver decompensation. But it reinforces what physicians generally know, which is that some patients with severe alcohol use disorder also have liver failure, and they tend to fare worse than others after a liver transplant.
Still, physicians also face a conundrum because there are increasing numbers of younger patients who won’t survive if they don’t get a liver transplant. “The challenge is picking out patients who are going to be good candidates from a purely medical point of view, but have a low likelihood of resuming alcohol use after transplantation [which could injure] the new liver,” said Dr. Martin. The new study has the potential to provide some additional guidance in patient selection.
The study authors disclosed no relevant conflicts of interest. Dr. Martin has no relevant financial disclosures.
Past decompensation in alcohol-associated hepatitis may be linked with worse survival following liver transplantation, but it’s not all bad news, according to a retrospective study.
Traditionally, patients with alcoholic liver disease were asked to be alcohol free for 6 months before consideration for a liver transplantation. In recent years, there’s been a loosening of that policy, with physicians considering “early” liver transplantation (early LT) instead of waiting 6 months. “It became obvious that a lot of patients do resume alcohol use after transplant, and most of them don’t appear to suffer too much in the way of adverse consequences,” said Paul Martin, MD, chief of hepatology at the University of Miami, who was not involved in the current research.
In 2011, a study confirmed that suspicion, finding that 6-month survival was 77% among carefully selected patients with alcohol-associated hepatitis for whom the 6-month sobriety requirement was waived; 6-month survival in those who did not receive a transplant was 22%. The selection criteria included the presence of supportive family members, the absence of severe coexisting conditions, and a commitment to abstaining from alcohol.
However, authors of the current study, published in the American Journal of Gastroenterology sought nuance: The appropriateness of prior decompensation as exclusion criteria in published studies is unknown, so the researchers compared outcomes of patients with prior versus first-time liver decompensation in alcohol-associated hepatitis.
Not all bad news
The study included 241 patients from six sites who consecutively received early LT between 2007 and 2020. Among these, 210 were identified as having a first-time liver decompensation event and 31 as having had a prior history of liver decompensation, defined as being diagnosed with ascites, hepatic encephalopathy, variceal bleeding, or jaundice.
There was no significant difference in median age, Model for End-Stage Liver Disease (MELD) scores, or post–liver transplant follow-up time between those with first-time liver decompensation or a prior history. The unadjusted 1-year survival rate was 93% in the first decompensation group (95% confidence interval, 89%-96%) and 86% in the prior decompensation group (95% CI, 66%-94%). The unadjusted 3-year survival rates were 85% (95% CI, 79%-90%) and 78% (95% CI, 57%-89%), respectively.
Importantly, the researchers found an association between prior decompensation and higher adjusted post–liver transplantation mortality (adjusted hazard ratio, 2.72; 95% CI, 1.61-4.59) and harmful alcohol use (aHR, 1.77; 95% CI, 1.07-2.92).
However, the researchers noted that these patients, who had MELD scores of 39 and previous decompensation, were at exceptionally high risk of short-term mortality, but still had 1- and 3-year survival rates above 85% and 75%, respectively, with early LT. “While longer follow-up is desirable as graft failure related to alcohol is most apparent after 5-years post LT, these results suggest that prior decompensation alone should not be considered an absolute contraindication to early LT.”
Limitations of the study included its retrospective data and small sample size for patients with prior decompensation.
“These findings validate the value of the ‘first decompensation’ criteria in published experiences regarding early LT for [alcoholic hepatitis],” the investigators concluded. “Further larger and prospective studies with longer-term follow-up will be needed to assess ways to optimally select patients in this cohort who may benefit most from early LT, and ways to manage patients at highest risk for worse outcomes post LT.”
A note of caution for early LT
About half of all liver mortality is attributable to alcoholic-associated liver disease. Corticosteroids can improve short-term survival, but there are no medications proven to increase long-term survival. That leaves liver transplant as the sole alternative for patients who don’t respond to corticosteroids.
“Programs in North America have liberalized their acceptance criteria for patients with alcoholic liver disease, and that’s resulted in large numbers of patients being transplanted who have less than 6 months abstinence. And overall, the results seem good, but I think this paper strikes an appropriate note of caution. In essence, if a patient had at least one prior episode of liver failure related to alcoholic excess and had recovered from that, and continued to drink and got into trouble again, [and then] presented for consideration for liver transplantation, the fact that they resumed alcohol use after prior episodes of decompensation suggests that they may be less-than-ideal candidates [for liver transplantation],” said Dr. Martin.
He pointed out important caveats to the study, including its retrospective nature and its inclusion of a relatively small number of patients with a history of liver decompensation. But it reinforces what physicians generally know, which is that some patients with severe alcohol use disorder also have liver failure, and they tend to fare worse than others after a liver transplant.
Still, physicians also face a conundrum because there are increasing numbers of younger patients who won’t survive if they don’t get a liver transplant. “The challenge is picking out patients who are going to be good candidates from a purely medical point of view, but have a low likelihood of resuming alcohol use after transplantation [which could injure] the new liver,” said Dr. Martin. The new study has the potential to provide some additional guidance in patient selection.
The study authors disclosed no relevant conflicts of interest. Dr. Martin has no relevant financial disclosures.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Link between pediatric hepatitis and adenovirus 41 still unclear
While two new studies reiterate a possible relationship between adenovirus 41 and acute hepatitis of unknown cause in children, whether these infections are significant or merely bystanders remains unclear.
In both studies – one conducted in Alabama and the other conducted in the United Kingdom – researchers found that 90% of children with acute hepatitis of unknown cause tested positive for adenovirus 41. The virus subtype is not an uncommon infection, but it usually causes gastroenteritis in children.
“Across the world, adenovirus continues to be a common signal” in these pediatric hepatitis cases, said Helena Gutierrez, MD, the medical director of the Pediatric Liver Transplant Program at the University of Alabama at Birmingham, in an interview. She led one of the studies. More data are necessary to understand what role this virus may play in these cases, she said.
In November, the Alabama Department of Public Health and the U.S. Centers for Disease Control and Prevention began investigating a cluster of severe pediatric hepatitis cases at the Children’s of Alabama hospital in Birmingham. These children also tested positive for adenovirus. In April, the United Kingdom announced they were investigating similar cases, and the CDC expanded their search nationally. As of July 8, 1,010 cases in 35 countries have been reported to the World Health Organization. There are 263 confirmed cases in the United Kingdom and 332 cases under investigation by the CDC in the United States, according to the most recent counts.
The two studies, both published in the New England Journal of Medicine, provide additional clinical data on a number of these mysterious hepatitis cases. Dr. Gutierrez’s study looked at nine children admitted for hepatitis of unknown origin between October 1 and February 28. Patients had a median age of 2 years 11 months and two required liver transplants, and there were no deaths.
Eight out of nine patients (89%) tested positive for adenovirus, and all five of the samples that were of sufficient quality for gene sequencing tested positive for adenovirus 41. None of the six liver biopsies performed found signs of adenovirus infection, but the liver tissue samples of three patients tested positive for adenovirus via PCR.
The second study involved 44 children referred to a liver transplantation center in the United Kingdom between January 1 and April 11, 2022. The median age for patients was 4 years. Six children required liver transplants, and there were no deaths. Of the 30 patients who underwent molecular adenovirus testing, 27 (90%) were positive for adenovirus 41. Liver samples of nine children (3 from biopsies and 6 from explanted livers) all tested negative for adenovirus antibodies.
In both studies, however, the median adenovirus viral load of patients needing a transplant was much higher than the viral loads in children who did not require liver transplants.
Although most of the clinical features and test results of these cases suggest that adenovirus may be involved, the negative results in histology are “intriguing,” Chayarani Kelgeri, MD, a consultant pediatric hepatologist at the Birmingham Women’s and Children’s Hospital, U.K., said in an email. She is the lead author of the U.K. study. “Whether this is because the liver injury we see is an aftermath of the viral infection, the mechanism of injury is immune mediated, and if other cofactors are involved is being explored,” she added. “Further investigations being undertaken by UK Health Security Agency will add to our understanding of this illness.”
Although there is a high adenovirus positivity rate amongst these cases, there is not enough evidence yet to say adenovirus 41 is a new cause of pediatric hepatitis in previously healthy children, said Saul Karpen, MD, PhD, the division chief of pediatric gastroenterology, hepatology, and nutrition at Emory University School of Medicine, Atlanta. He wrote an editorial accompanying the two NEJM studies.
The CDC has not yet found an increase in pediatric hepatitis cases, according to a recent analysis, though the United Kingdom has found an uptick in cases this year, he told this news organization. Also, the cases highlighted in both articles showed no histological evidence of adenovirus in liver biopsies. “That’s completely opposite of what we generally see in adenoviral hepatitis that can be quite severe,” he said, adding that in general, there are detectable viral particles and antigens in affected livers.
“These two important reports indicate to those inside and outside the field of pediatric hepatology that registries and clinical studies of acute hepatitis in children are sorely needed,” Dr. Karpen writes in the editorial; “It is likely that with greater attention to collecting data on cases and biospecimens from children with acute hepatitis, we will be able to determine whether this one virus, human adenovirus 41, is of relevance to this important and serious condition in children.”
Dr. Gutierrez, Dr. Kelgeri, and Dr. Karpen report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While two new studies reiterate a possible relationship between adenovirus 41 and acute hepatitis of unknown cause in children, whether these infections are significant or merely bystanders remains unclear.
In both studies – one conducted in Alabama and the other conducted in the United Kingdom – researchers found that 90% of children with acute hepatitis of unknown cause tested positive for adenovirus 41. The virus subtype is not an uncommon infection, but it usually causes gastroenteritis in children.
“Across the world, adenovirus continues to be a common signal” in these pediatric hepatitis cases, said Helena Gutierrez, MD, the medical director of the Pediatric Liver Transplant Program at the University of Alabama at Birmingham, in an interview. She led one of the studies. More data are necessary to understand what role this virus may play in these cases, she said.
In November, the Alabama Department of Public Health and the U.S. Centers for Disease Control and Prevention began investigating a cluster of severe pediatric hepatitis cases at the Children’s of Alabama hospital in Birmingham. These children also tested positive for adenovirus. In April, the United Kingdom announced they were investigating similar cases, and the CDC expanded their search nationally. As of July 8, 1,010 cases in 35 countries have been reported to the World Health Organization. There are 263 confirmed cases in the United Kingdom and 332 cases under investigation by the CDC in the United States, according to the most recent counts.
The two studies, both published in the New England Journal of Medicine, provide additional clinical data on a number of these mysterious hepatitis cases. Dr. Gutierrez’s study looked at nine children admitted for hepatitis of unknown origin between October 1 and February 28. Patients had a median age of 2 years 11 months and two required liver transplants, and there were no deaths.
Eight out of nine patients (89%) tested positive for adenovirus, and all five of the samples that were of sufficient quality for gene sequencing tested positive for adenovirus 41. None of the six liver biopsies performed found signs of adenovirus infection, but the liver tissue samples of three patients tested positive for adenovirus via PCR.
The second study involved 44 children referred to a liver transplantation center in the United Kingdom between January 1 and April 11, 2022. The median age for patients was 4 years. Six children required liver transplants, and there were no deaths. Of the 30 patients who underwent molecular adenovirus testing, 27 (90%) were positive for adenovirus 41. Liver samples of nine children (3 from biopsies and 6 from explanted livers) all tested negative for adenovirus antibodies.
In both studies, however, the median adenovirus viral load of patients needing a transplant was much higher than the viral loads in children who did not require liver transplants.
Although most of the clinical features and test results of these cases suggest that adenovirus may be involved, the negative results in histology are “intriguing,” Chayarani Kelgeri, MD, a consultant pediatric hepatologist at the Birmingham Women’s and Children’s Hospital, U.K., said in an email. She is the lead author of the U.K. study. “Whether this is because the liver injury we see is an aftermath of the viral infection, the mechanism of injury is immune mediated, and if other cofactors are involved is being explored,” she added. “Further investigations being undertaken by UK Health Security Agency will add to our understanding of this illness.”
Although there is a high adenovirus positivity rate amongst these cases, there is not enough evidence yet to say adenovirus 41 is a new cause of pediatric hepatitis in previously healthy children, said Saul Karpen, MD, PhD, the division chief of pediatric gastroenterology, hepatology, and nutrition at Emory University School of Medicine, Atlanta. He wrote an editorial accompanying the two NEJM studies.
The CDC has not yet found an increase in pediatric hepatitis cases, according to a recent analysis, though the United Kingdom has found an uptick in cases this year, he told this news organization. Also, the cases highlighted in both articles showed no histological evidence of adenovirus in liver biopsies. “That’s completely opposite of what we generally see in adenoviral hepatitis that can be quite severe,” he said, adding that in general, there are detectable viral particles and antigens in affected livers.
“These two important reports indicate to those inside and outside the field of pediatric hepatology that registries and clinical studies of acute hepatitis in children are sorely needed,” Dr. Karpen writes in the editorial; “It is likely that with greater attention to collecting data on cases and biospecimens from children with acute hepatitis, we will be able to determine whether this one virus, human adenovirus 41, is of relevance to this important and serious condition in children.”
Dr. Gutierrez, Dr. Kelgeri, and Dr. Karpen report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
While two new studies reiterate a possible relationship between adenovirus 41 and acute hepatitis of unknown cause in children, whether these infections are significant or merely bystanders remains unclear.
In both studies – one conducted in Alabama and the other conducted in the United Kingdom – researchers found that 90% of children with acute hepatitis of unknown cause tested positive for adenovirus 41. The virus subtype is not an uncommon infection, but it usually causes gastroenteritis in children.
“Across the world, adenovirus continues to be a common signal” in these pediatric hepatitis cases, said Helena Gutierrez, MD, the medical director of the Pediatric Liver Transplant Program at the University of Alabama at Birmingham, in an interview. She led one of the studies. More data are necessary to understand what role this virus may play in these cases, she said.
In November, the Alabama Department of Public Health and the U.S. Centers for Disease Control and Prevention began investigating a cluster of severe pediatric hepatitis cases at the Children’s of Alabama hospital in Birmingham. These children also tested positive for adenovirus. In April, the United Kingdom announced they were investigating similar cases, and the CDC expanded their search nationally. As of July 8, 1,010 cases in 35 countries have been reported to the World Health Organization. There are 263 confirmed cases in the United Kingdom and 332 cases under investigation by the CDC in the United States, according to the most recent counts.
The two studies, both published in the New England Journal of Medicine, provide additional clinical data on a number of these mysterious hepatitis cases. Dr. Gutierrez’s study looked at nine children admitted for hepatitis of unknown origin between October 1 and February 28. Patients had a median age of 2 years 11 months and two required liver transplants, and there were no deaths.
Eight out of nine patients (89%) tested positive for adenovirus, and all five of the samples that were of sufficient quality for gene sequencing tested positive for adenovirus 41. None of the six liver biopsies performed found signs of adenovirus infection, but the liver tissue samples of three patients tested positive for adenovirus via PCR.
The second study involved 44 children referred to a liver transplantation center in the United Kingdom between January 1 and April 11, 2022. The median age for patients was 4 years. Six children required liver transplants, and there were no deaths. Of the 30 patients who underwent molecular adenovirus testing, 27 (90%) were positive for adenovirus 41. Liver samples of nine children (3 from biopsies and 6 from explanted livers) all tested negative for adenovirus antibodies.
In both studies, however, the median adenovirus viral load of patients needing a transplant was much higher than the viral loads in children who did not require liver transplants.
Although most of the clinical features and test results of these cases suggest that adenovirus may be involved, the negative results in histology are “intriguing,” Chayarani Kelgeri, MD, a consultant pediatric hepatologist at the Birmingham Women’s and Children’s Hospital, U.K., said in an email. She is the lead author of the U.K. study. “Whether this is because the liver injury we see is an aftermath of the viral infection, the mechanism of injury is immune mediated, and if other cofactors are involved is being explored,” she added. “Further investigations being undertaken by UK Health Security Agency will add to our understanding of this illness.”
Although there is a high adenovirus positivity rate amongst these cases, there is not enough evidence yet to say adenovirus 41 is a new cause of pediatric hepatitis in previously healthy children, said Saul Karpen, MD, PhD, the division chief of pediatric gastroenterology, hepatology, and nutrition at Emory University School of Medicine, Atlanta. He wrote an editorial accompanying the two NEJM studies.
The CDC has not yet found an increase in pediatric hepatitis cases, according to a recent analysis, though the United Kingdom has found an uptick in cases this year, he told this news organization. Also, the cases highlighted in both articles showed no histological evidence of adenovirus in liver biopsies. “That’s completely opposite of what we generally see in adenoviral hepatitis that can be quite severe,” he said, adding that in general, there are detectable viral particles and antigens in affected livers.
“These two important reports indicate to those inside and outside the field of pediatric hepatology that registries and clinical studies of acute hepatitis in children are sorely needed,” Dr. Karpen writes in the editorial; “It is likely that with greater attention to collecting data on cases and biospecimens from children with acute hepatitis, we will be able to determine whether this one virus, human adenovirus 41, is of relevance to this important and serious condition in children.”
Dr. Gutierrez, Dr. Kelgeri, and Dr. Karpen report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Two genetic intestinal diseases linked
Two genes that have been linked separately to rare intestinal diseases appear to share a functional relationship. The genes have independently been linked to osteo-oto-hepato-enteric (O2HE) syndrome and microvillus inclusion disease (MVID), which are characterized by congenital diarrhea and, in some patients, intrahepatic cholestasis.
It appears that one gene, UNC45A, is directly responsible for the proper function of the protein encoded by the other gene, called MYO5B, according to investigators, who published their findings in Cellular and Molecular Gastroenterology and Hepatology. UNC45A is a chaperone protein that helps proteins fold properly. It has been linked to O2HE patients experiencing congenital diarrhea and intrahepatic cholestasis. The mutation has been identified in four patients from three different families with O2HE, which can also present with sensorineural hearing loss and bone fragility. Cellular analyses have shown that the mutation leads to reduction in protein expression by 70%-90%.
Intestinal symptoms similar to those in O2HE have also been described in diseases caused by mutations in genes that encode the myosin motor proteins that are involved in cellular protein trafficking. This group of disorders includes MVID. The researchers hypothesized that the UNC45A mutation in O2HE might lead to similar symptoms as MVID and others through the altered protein’s failure to assist in the folding of myosin proteins, although to date only the myosin IIa protein has been shown to be a target of UNC45A.
To investigate the possibility, they examined in more detail the relationship between UNC45A and intestinal symptoms. There are various known mutations in myosin proteins. Some have been linked to deafness, but these do not appear to contribute to intestinal symptoms since patients with myelin-related inherited deafness don’t typically have diarrhea. Bone fragility, also sometimes caused by myosin mutations, also appears to be unrelated to intestinal symptoms.
Previous experiments in yeast suggest that the related gene UNC45 may serve as a chaperone for type V myosin: Loss of a yeast version of UNC45 caused a type V myosin called MYO4P to be mislocalized in yeast. In zebrafish, reduction in intestinal levels of the UNC45A gene or the fish’s version of MYO5B interfered with development of intestinal folds.
The researchers used CRISPR-Cas9 gene editing and site-directed mutagenesis in intestinal epithelial and liver cell lines to investigate the relationships between UNC45A and MYO5B mutants. UNC45A depletion or introduction of the UNC45A mutation found in patients led to lower MYO5B expression. Within epithelial cells, loss of UNC45A led to changes in MYO5B–linked processes that are known to play a role in MVID pathogenesis. These included alteration of microvilli development and interference with the location of rat sarcoma–associated binding protein (RAB) 11A–positive recycling endosomes. When normal UNC45A was reintroduced to these cells, MYO5B expression returned. Reintroduction of either UNC45A or MYO5B repaired the alterations to recycling endosome position and microvilli development.
Loss of UNC45A did not appear to affect transcription of the MYO5B gen, which suggests a suggesting a functional interaction between the two at a protein level.
UNC45A has been shown to destabilize microtubules. Exposure of a kidney epithelial cell line to the microtubule-stabilizing drug taxol also led to displacement of RAB11A-positve recycling endosomes, though the specific changes were different than what is seen in MYO5B mutants. The researchers were unable to validate the findings in tissue derived from O2HE patients because of insufficient material, but they maintain that the cell lines used have proven to be highly predictive for the cellular characteristics of MVID.
Overall, the study suggests that reductions in MYO5B and subsequent changes to the cellular processes that depend on it may underlie the intestinal symptoms in O2HE.
The researchers noted that O2HE patients have different phenotypes. Of the four patients they studied, three had severe chronic diarrhea and required parenteral nutrition. One patient later had the diarrhea resolve and her sister did not have diarrhea at all. This heterogeneity in severity and duration of clinical symptoms may be driven by differences in the molecular effects of patient-specific mutations. The two siblings had mutations in a different region of the UNC45A gene than the other two participants.
“Taken together, this study revealed a functional relationship between UNC45A and MYO5B protein expression, thereby connecting two rare congenital diseases with overlapping intestinal symptoms at the molecular level,” the authors wrote.
The authors reported that they had no conflicts of interest.
This article was updated 7/13/22.
Congenital diarrheas and enteropathies (CoDEs) are rare monogenic disorders caused by genes important for intestinal epithelial function. The increasing availability of exome sequencing in clinical practice has accelerated the discovery of new genes associated with these disorders over the past few years. Several CoDE disorders revolve around defects in trafficking of vesicles in epithelial cells. One of these is microvillus inclusion disease which is caused by loss-of-function variants in the gene MYO5B, which encodes an important epithelial motor protein. This study by Li and colleagues reveals that a recently discovered novel CoDE gene and protein, UNC45A, is functionally linked to MYO5B and that loss of UNC45A in cells causes a very similar cellular phenotype to MYO5B-deficient cells.
Jay Thiagarajah, MD, PhD, attending in the division of gastroenterology, hepatology and nutrition and codirector of the congenital enteropathy program at Boston Children’s Hospital, as well as assistant professor in pediatrics at Harvard Medical School, also in Boston. Dr. Thiagarajah stated he had no relevant conflicts to disclose.
Congenital diarrheas and enteropathies (CoDEs) are rare monogenic disorders caused by genes important for intestinal epithelial function. The increasing availability of exome sequencing in clinical practice has accelerated the discovery of new genes associated with these disorders over the past few years. Several CoDE disorders revolve around defects in trafficking of vesicles in epithelial cells. One of these is microvillus inclusion disease which is caused by loss-of-function variants in the gene MYO5B, which encodes an important epithelial motor protein. This study by Li and colleagues reveals that a recently discovered novel CoDE gene and protein, UNC45A, is functionally linked to MYO5B and that loss of UNC45A in cells causes a very similar cellular phenotype to MYO5B-deficient cells.
Jay Thiagarajah, MD, PhD, attending in the division of gastroenterology, hepatology and nutrition and codirector of the congenital enteropathy program at Boston Children’s Hospital, as well as assistant professor in pediatrics at Harvard Medical School, also in Boston. Dr. Thiagarajah stated he had no relevant conflicts to disclose.
Congenital diarrheas and enteropathies (CoDEs) are rare monogenic disorders caused by genes important for intestinal epithelial function. The increasing availability of exome sequencing in clinical practice has accelerated the discovery of new genes associated with these disorders over the past few years. Several CoDE disorders revolve around defects in trafficking of vesicles in epithelial cells. One of these is microvillus inclusion disease which is caused by loss-of-function variants in the gene MYO5B, which encodes an important epithelial motor protein. This study by Li and colleagues reveals that a recently discovered novel CoDE gene and protein, UNC45A, is functionally linked to MYO5B and that loss of UNC45A in cells causes a very similar cellular phenotype to MYO5B-deficient cells.
Jay Thiagarajah, MD, PhD, attending in the division of gastroenterology, hepatology and nutrition and codirector of the congenital enteropathy program at Boston Children’s Hospital, as well as assistant professor in pediatrics at Harvard Medical School, also in Boston. Dr. Thiagarajah stated he had no relevant conflicts to disclose.
Two genes that have been linked separately to rare intestinal diseases appear to share a functional relationship. The genes have independently been linked to osteo-oto-hepato-enteric (O2HE) syndrome and microvillus inclusion disease (MVID), which are characterized by congenital diarrhea and, in some patients, intrahepatic cholestasis.
It appears that one gene, UNC45A, is directly responsible for the proper function of the protein encoded by the other gene, called MYO5B, according to investigators, who published their findings in Cellular and Molecular Gastroenterology and Hepatology. UNC45A is a chaperone protein that helps proteins fold properly. It has been linked to O2HE patients experiencing congenital diarrhea and intrahepatic cholestasis. The mutation has been identified in four patients from three different families with O2HE, which can also present with sensorineural hearing loss and bone fragility. Cellular analyses have shown that the mutation leads to reduction in protein expression by 70%-90%.
Intestinal symptoms similar to those in O2HE have also been described in diseases caused by mutations in genes that encode the myosin motor proteins that are involved in cellular protein trafficking. This group of disorders includes MVID. The researchers hypothesized that the UNC45A mutation in O2HE might lead to similar symptoms as MVID and others through the altered protein’s failure to assist in the folding of myosin proteins, although to date only the myosin IIa protein has been shown to be a target of UNC45A.
To investigate the possibility, they examined in more detail the relationship between UNC45A and intestinal symptoms. There are various known mutations in myosin proteins. Some have been linked to deafness, but these do not appear to contribute to intestinal symptoms since patients with myelin-related inherited deafness don’t typically have diarrhea. Bone fragility, also sometimes caused by myosin mutations, also appears to be unrelated to intestinal symptoms.
Previous experiments in yeast suggest that the related gene UNC45 may serve as a chaperone for type V myosin: Loss of a yeast version of UNC45 caused a type V myosin called MYO4P to be mislocalized in yeast. In zebrafish, reduction in intestinal levels of the UNC45A gene or the fish’s version of MYO5B interfered with development of intestinal folds.
The researchers used CRISPR-Cas9 gene editing and site-directed mutagenesis in intestinal epithelial and liver cell lines to investigate the relationships between UNC45A and MYO5B mutants. UNC45A depletion or introduction of the UNC45A mutation found in patients led to lower MYO5B expression. Within epithelial cells, loss of UNC45A led to changes in MYO5B–linked processes that are known to play a role in MVID pathogenesis. These included alteration of microvilli development and interference with the location of rat sarcoma–associated binding protein (RAB) 11A–positive recycling endosomes. When normal UNC45A was reintroduced to these cells, MYO5B expression returned. Reintroduction of either UNC45A or MYO5B repaired the alterations to recycling endosome position and microvilli development.
Loss of UNC45A did not appear to affect transcription of the MYO5B gen, which suggests a suggesting a functional interaction between the two at a protein level.
UNC45A has been shown to destabilize microtubules. Exposure of a kidney epithelial cell line to the microtubule-stabilizing drug taxol also led to displacement of RAB11A-positve recycling endosomes, though the specific changes were different than what is seen in MYO5B mutants. The researchers were unable to validate the findings in tissue derived from O2HE patients because of insufficient material, but they maintain that the cell lines used have proven to be highly predictive for the cellular characteristics of MVID.
Overall, the study suggests that reductions in MYO5B and subsequent changes to the cellular processes that depend on it may underlie the intestinal symptoms in O2HE.
The researchers noted that O2HE patients have different phenotypes. Of the four patients they studied, three had severe chronic diarrhea and required parenteral nutrition. One patient later had the diarrhea resolve and her sister did not have diarrhea at all. This heterogeneity in severity and duration of clinical symptoms may be driven by differences in the molecular effects of patient-specific mutations. The two siblings had mutations in a different region of the UNC45A gene than the other two participants.
“Taken together, this study revealed a functional relationship between UNC45A and MYO5B protein expression, thereby connecting two rare congenital diseases with overlapping intestinal symptoms at the molecular level,” the authors wrote.
The authors reported that they had no conflicts of interest.
This article was updated 7/13/22.
Two genes that have been linked separately to rare intestinal diseases appear to share a functional relationship. The genes have independently been linked to osteo-oto-hepato-enteric (O2HE) syndrome and microvillus inclusion disease (MVID), which are characterized by congenital diarrhea and, in some patients, intrahepatic cholestasis.
It appears that one gene, UNC45A, is directly responsible for the proper function of the protein encoded by the other gene, called MYO5B, according to investigators, who published their findings in Cellular and Molecular Gastroenterology and Hepatology. UNC45A is a chaperone protein that helps proteins fold properly. It has been linked to O2HE patients experiencing congenital diarrhea and intrahepatic cholestasis. The mutation has been identified in four patients from three different families with O2HE, which can also present with sensorineural hearing loss and bone fragility. Cellular analyses have shown that the mutation leads to reduction in protein expression by 70%-90%.
Intestinal symptoms similar to those in O2HE have also been described in diseases caused by mutations in genes that encode the myosin motor proteins that are involved in cellular protein trafficking. This group of disorders includes MVID. The researchers hypothesized that the UNC45A mutation in O2HE might lead to similar symptoms as MVID and others through the altered protein’s failure to assist in the folding of myosin proteins, although to date only the myosin IIa protein has been shown to be a target of UNC45A.
To investigate the possibility, they examined in more detail the relationship between UNC45A and intestinal symptoms. There are various known mutations in myosin proteins. Some have been linked to deafness, but these do not appear to contribute to intestinal symptoms since patients with myelin-related inherited deafness don’t typically have diarrhea. Bone fragility, also sometimes caused by myosin mutations, also appears to be unrelated to intestinal symptoms.
Previous experiments in yeast suggest that the related gene UNC45 may serve as a chaperone for type V myosin: Loss of a yeast version of UNC45 caused a type V myosin called MYO4P to be mislocalized in yeast. In zebrafish, reduction in intestinal levels of the UNC45A gene or the fish’s version of MYO5B interfered with development of intestinal folds.
The researchers used CRISPR-Cas9 gene editing and site-directed mutagenesis in intestinal epithelial and liver cell lines to investigate the relationships between UNC45A and MYO5B mutants. UNC45A depletion or introduction of the UNC45A mutation found in patients led to lower MYO5B expression. Within epithelial cells, loss of UNC45A led to changes in MYO5B–linked processes that are known to play a role in MVID pathogenesis. These included alteration of microvilli development and interference with the location of rat sarcoma–associated binding protein (RAB) 11A–positive recycling endosomes. When normal UNC45A was reintroduced to these cells, MYO5B expression returned. Reintroduction of either UNC45A or MYO5B repaired the alterations to recycling endosome position and microvilli development.
Loss of UNC45A did not appear to affect transcription of the MYO5B gen, which suggests a suggesting a functional interaction between the two at a protein level.
UNC45A has been shown to destabilize microtubules. Exposure of a kidney epithelial cell line to the microtubule-stabilizing drug taxol also led to displacement of RAB11A-positve recycling endosomes, though the specific changes were different than what is seen in MYO5B mutants. The researchers were unable to validate the findings in tissue derived from O2HE patients because of insufficient material, but they maintain that the cell lines used have proven to be highly predictive for the cellular characteristics of MVID.
Overall, the study suggests that reductions in MYO5B and subsequent changes to the cellular processes that depend on it may underlie the intestinal symptoms in O2HE.
The researchers noted that O2HE patients have different phenotypes. Of the four patients they studied, three had severe chronic diarrhea and required parenteral nutrition. One patient later had the diarrhea resolve and her sister did not have diarrhea at all. This heterogeneity in severity and duration of clinical symptoms may be driven by differences in the molecular effects of patient-specific mutations. The two siblings had mutations in a different region of the UNC45A gene than the other two participants.
“Taken together, this study revealed a functional relationship between UNC45A and MYO5B protein expression, thereby connecting two rare congenital diseases with overlapping intestinal symptoms at the molecular level,” the authors wrote.
The authors reported that they had no conflicts of interest.
This article was updated 7/13/22.
FROM CELLULAR AND MOLECULAR GASTROENTEROLOGY AND HEPATOLOGY
Finding HBV ‘cure’ may mean going ‘back to the drawing board’
LONDON – Achieving a functional cure for hepatitis B virus (HBV) is not going to be easily achieved with the drugs that are currently in development, according to a presentation at the annual International Liver Congress sponsored by the European Association for the Study of the Liver.
“Intriguing results have been presented at ILC 2022 that must be carefully interpreted,” said Jean-Michel Pawlotsky, MD, PhD, of Henri Mondor Hospital in Créteil, France, during the viral hepatitis highlights session on the closing day of the meeting.
“New HBV drug development looks more complicated than initially expected and its goals and strategies need to be redefined and refocused,” he added
“This is really something that came from the discussions we had during the sessions but also in the corridors,” Dr. Pawlotsky added. “We know it’s going to be difficult; we have to reset, restart – not from zero, but from not much – and revise our strategy,” he suggested.
There are many new drugs under investigation for HBV, Dr. Pawlotsky said, noting that the number of studies being presented at the meeting was reminiscent of the flurry of activity before a functional cure for hepatitis C had been found. “It’s good to see that this is happening again for HBV,” he said.
Indeed, there are many new direct-acting antiviral agents, immunomodulatory, or other approaches being tested, and some of the more advanced studies are “teaching us a few things and probably raising more questions than getting answers,” Dr. Pawlotsky said.
The B-CLEAR study
One these studies is the phase 2b B-CLEAR study presented during the late-breaker session. This study involved bepirovirsen, an antisense oligonucleotide, and tested its efficacy and safety in patients with chronic hepatitis B virus infection who were either on or off stable nucleos(t)ide analogue (NA/NUC) therapy.
A similar proportion (28% and 29%, respectively) of patients achieved an hepatitis B surface antigen (HBsAg) level below the lower limit of quantification at the end of 24 weeks treatment. However, the effect on HBsAg varied according to the treatment arm, with changes to the dosing or switching to placebo indicating that the effect might wane when the treatment is stopped or if the dose is reduced.
“Interestingly, ALT elevations were observed in association with most HBsAg declines,” Dr. Pawlotsky pointed out. “I think we still have to determine whether this is good flare/bad flare, good sign/bad sign, of what is going to happen afterward.”
The REEF studies
Another approach highlighted was the combination of the silencing or small interfering RNA (siRNA) JNJ-3989 with the capsid assembly modulator (CAM) JNJ-6379 in the phase 2 REEF-1 and REEF-2 studies.
REEF-1, conducted in patients who were either hepatitis B e antigen (HBeAg) positive or negative who were not treated with NA/NUC or were NA/NUC suppressed, showed a dose-dependent, but variable effect among individual patients as might be expected at the end of 48 weeks’ treatment. This was sustained at week 72, which was 24 weeks’ follow-up after stopping treatment.
However, pointed out Dr. Pawlotsky “I think the most important part of this is that if you add a CAM on top of the siRNA, you do not improve the effect on HBsAg levels.”
Then there is the REEF-2 study, testing the same combination but in only patients who were NA suppressed or HBeAg negative alongside standard NA/NUC therapy. As well as being the first novel combination treatment trial to report, this was essentially a stopping trial, Kosh Agarwal, BMedSci (Hons), MBBS, MD, one of the study’s investigators explained separately at a media briefing.
Patients (n = 130) were treated for 48 weeks, then all treatment – including NA/NUC – was discontinued, with 48 weeks of follow-up after discontinuation, said Dr. Agarwal, who is a consultant hepatologist based at the Institute of Liver Studies at King’s College Hospital, London. He presented data from the first 24 week period after treatment had ended.
At the end of treatment, the combination had resulted in a mean reduction in HBsAg of 1.89 log10 IU/mL versus a reduction of 0.06 for the NA/NUC-only group, which acted as the control group in this trial. But “no patient in this study lost their surface antigen, i.e., were cured of their hepatitis B in the active arm or in the control arm,” Dr. Agarwal said.
“We didn’t achieve a cure, but a significant proportion were in a ‘controlled’ viral stage,” said Dr. Agarwal. Indeed, during his presentation of the findings, he showed that HBsAg inhibition was maintained in the majority (72%) of patients after stopping the combination.
While the trial’s primary endpoint wasn’t met, “it’s a really important study,” said Dr. Agarwal. “This [study] was fulfilled and delivered in the COVID era, so a lot of patients were looked after very carefully by sites in Europe,” he observed.
Further follow-up from the trial is expected, and Dr. Agarwal said that the subsequent discussion will “take us back to the drawing board to think about whether we need better antiviral treatments or whether we need to think about different combinations, and whether actually stopping treatment with every treatment is the right strategy to take.”
Both Dr. Agarwal and Dr. Pawlotsky flagged up the case of one patient in the trial who had been in the control arm and had experienced severe HBV reactivation that required a liver transplant.
“This patient is a warning signal,” Dr. Pawlotsky suggested in his talk. “When we think about NUC stopping, we have to think about the potential benefit in terms of HbsAg loss but also the potential risks.”
While Dr. Agarwal had noted that it highlights that “careful design of retreatment criteria is important in studies assessing the NA/NUC-stopping concept”.
Monoclonal antibody shows promise
Other combinations could involve an siRNA and an immunomodulatory agent and, during the poster sessions at the meeting, Dr. Agarwal also presented data from an ongoing phase 1 study with a novel, neutralizing monoclonal antibody called VIR-3434.
This monoclonal antibody is novel because it is thought to have several modes of action, first by binding to HBV and affecting its entry into liver cells, then by presenting the virus to T cells and stimulating a ‘vaccinal’ or immune effect, and then by helping the with the clearance of HBsAg and delivery of the virus to dendritic cells.
In the study, single doses of VIR-3434 were found to be well tolerated and to produce rapid reductions in HBsAg, with the highest dose used (300 mg) producing the greatest and most durable effect up to week 8.
VIR-3434 is also being tested in combination with other drugs in the phase 2 MARCH trial. One of these combinations is VIR-3434 together with an investigational siRNA dubbed VIR-2218. Preclinical work presented at ILC 2022 suggests that this combination appears to be capable of reducing HBsAg to a greater extent than using either agent alone.
Rethinking the strategy to get to a cure
Of course, VIR-3434 is one of several immunomodulatory compounds in development. There are therapeutic vaccines, drugs targeting the innate immune response, other monoclonal antibodies, T-cell receptors, checkpoint inhibitors and PD-L1 inhibitors. Then there are other compounds such as entry inhibitors, apoptosis inducers, and farnesoid X receptor agonists.
“I finish this meeting with more questions than answers,” Dr. Pawlotsky said. “What is the right target to enhance specific anti-HBV immunity? Does in vivo induction of immune responses translate into any beneficial effect on HBV infection? Will therapeutic vaccines every work in a viral infection?”
Moreover, he asked, “how can we avoid the side effect of enhancing multiple and complex nonspecific immune responses? Are treatment-induced flares good flares or bad flares? All of these are questions that are really unanswered and that we’ll have to get answers to in the near future.”
The B-CLEAR study was sponsored by GlaxoSmithKline. The REEF-2 study was sponsored by Janssen Research & Development. The VIR-3434 studies were funded by Vir Biotechnology. Dr. Pawlotsky has received grant and research support, acted as a consultant, adviser, or speaker, and participated in advisory boards for multiple pharmaceutical and biotechnology companies. This news organization was unable to verify Dr. Agarwal’s ties to Vir Biotechnology, but he presented one of the posters on VIR-3434 at the meeting and has been involved in the phase 1 study that was reported.
LONDON – Achieving a functional cure for hepatitis B virus (HBV) is not going to be easily achieved with the drugs that are currently in development, according to a presentation at the annual International Liver Congress sponsored by the European Association for the Study of the Liver.
“Intriguing results have been presented at ILC 2022 that must be carefully interpreted,” said Jean-Michel Pawlotsky, MD, PhD, of Henri Mondor Hospital in Créteil, France, during the viral hepatitis highlights session on the closing day of the meeting.
“New HBV drug development looks more complicated than initially expected and its goals and strategies need to be redefined and refocused,” he added
“This is really something that came from the discussions we had during the sessions but also in the corridors,” Dr. Pawlotsky added. “We know it’s going to be difficult; we have to reset, restart – not from zero, but from not much – and revise our strategy,” he suggested.
There are many new drugs under investigation for HBV, Dr. Pawlotsky said, noting that the number of studies being presented at the meeting was reminiscent of the flurry of activity before a functional cure for hepatitis C had been found. “It’s good to see that this is happening again for HBV,” he said.
Indeed, there are many new direct-acting antiviral agents, immunomodulatory, or other approaches being tested, and some of the more advanced studies are “teaching us a few things and probably raising more questions than getting answers,” Dr. Pawlotsky said.
The B-CLEAR study
One these studies is the phase 2b B-CLEAR study presented during the late-breaker session. This study involved bepirovirsen, an antisense oligonucleotide, and tested its efficacy and safety in patients with chronic hepatitis B virus infection who were either on or off stable nucleos(t)ide analogue (NA/NUC) therapy.
A similar proportion (28% and 29%, respectively) of patients achieved an hepatitis B surface antigen (HBsAg) level below the lower limit of quantification at the end of 24 weeks treatment. However, the effect on HBsAg varied according to the treatment arm, with changes to the dosing or switching to placebo indicating that the effect might wane when the treatment is stopped or if the dose is reduced.
“Interestingly, ALT elevations were observed in association with most HBsAg declines,” Dr. Pawlotsky pointed out. “I think we still have to determine whether this is good flare/bad flare, good sign/bad sign, of what is going to happen afterward.”
The REEF studies
Another approach highlighted was the combination of the silencing or small interfering RNA (siRNA) JNJ-3989 with the capsid assembly modulator (CAM) JNJ-6379 in the phase 2 REEF-1 and REEF-2 studies.
REEF-1, conducted in patients who were either hepatitis B e antigen (HBeAg) positive or negative who were not treated with NA/NUC or were NA/NUC suppressed, showed a dose-dependent, but variable effect among individual patients as might be expected at the end of 48 weeks’ treatment. This was sustained at week 72, which was 24 weeks’ follow-up after stopping treatment.
However, pointed out Dr. Pawlotsky “I think the most important part of this is that if you add a CAM on top of the siRNA, you do not improve the effect on HBsAg levels.”
Then there is the REEF-2 study, testing the same combination but in only patients who were NA suppressed or HBeAg negative alongside standard NA/NUC therapy. As well as being the first novel combination treatment trial to report, this was essentially a stopping trial, Kosh Agarwal, BMedSci (Hons), MBBS, MD, one of the study’s investigators explained separately at a media briefing.
Patients (n = 130) were treated for 48 weeks, then all treatment – including NA/NUC – was discontinued, with 48 weeks of follow-up after discontinuation, said Dr. Agarwal, who is a consultant hepatologist based at the Institute of Liver Studies at King’s College Hospital, London. He presented data from the first 24 week period after treatment had ended.
At the end of treatment, the combination had resulted in a mean reduction in HBsAg of 1.89 log10 IU/mL versus a reduction of 0.06 for the NA/NUC-only group, which acted as the control group in this trial. But “no patient in this study lost their surface antigen, i.e., were cured of their hepatitis B in the active arm or in the control arm,” Dr. Agarwal said.
“We didn’t achieve a cure, but a significant proportion were in a ‘controlled’ viral stage,” said Dr. Agarwal. Indeed, during his presentation of the findings, he showed that HBsAg inhibition was maintained in the majority (72%) of patients after stopping the combination.
While the trial’s primary endpoint wasn’t met, “it’s a really important study,” said Dr. Agarwal. “This [study] was fulfilled and delivered in the COVID era, so a lot of patients were looked after very carefully by sites in Europe,” he observed.
Further follow-up from the trial is expected, and Dr. Agarwal said that the subsequent discussion will “take us back to the drawing board to think about whether we need better antiviral treatments or whether we need to think about different combinations, and whether actually stopping treatment with every treatment is the right strategy to take.”
Both Dr. Agarwal and Dr. Pawlotsky flagged up the case of one patient in the trial who had been in the control arm and had experienced severe HBV reactivation that required a liver transplant.
“This patient is a warning signal,” Dr. Pawlotsky suggested in his talk. “When we think about NUC stopping, we have to think about the potential benefit in terms of HbsAg loss but also the potential risks.”
While Dr. Agarwal had noted that it highlights that “careful design of retreatment criteria is important in studies assessing the NA/NUC-stopping concept”.
Monoclonal antibody shows promise
Other combinations could involve an siRNA and an immunomodulatory agent and, during the poster sessions at the meeting, Dr. Agarwal also presented data from an ongoing phase 1 study with a novel, neutralizing monoclonal antibody called VIR-3434.
This monoclonal antibody is novel because it is thought to have several modes of action, first by binding to HBV and affecting its entry into liver cells, then by presenting the virus to T cells and stimulating a ‘vaccinal’ or immune effect, and then by helping the with the clearance of HBsAg and delivery of the virus to dendritic cells.
In the study, single doses of VIR-3434 were found to be well tolerated and to produce rapid reductions in HBsAg, with the highest dose used (300 mg) producing the greatest and most durable effect up to week 8.
VIR-3434 is also being tested in combination with other drugs in the phase 2 MARCH trial. One of these combinations is VIR-3434 together with an investigational siRNA dubbed VIR-2218. Preclinical work presented at ILC 2022 suggests that this combination appears to be capable of reducing HBsAg to a greater extent than using either agent alone.
Rethinking the strategy to get to a cure
Of course, VIR-3434 is one of several immunomodulatory compounds in development. There are therapeutic vaccines, drugs targeting the innate immune response, other monoclonal antibodies, T-cell receptors, checkpoint inhibitors and PD-L1 inhibitors. Then there are other compounds such as entry inhibitors, apoptosis inducers, and farnesoid X receptor agonists.
“I finish this meeting with more questions than answers,” Dr. Pawlotsky said. “What is the right target to enhance specific anti-HBV immunity? Does in vivo induction of immune responses translate into any beneficial effect on HBV infection? Will therapeutic vaccines every work in a viral infection?”
Moreover, he asked, “how can we avoid the side effect of enhancing multiple and complex nonspecific immune responses? Are treatment-induced flares good flares or bad flares? All of these are questions that are really unanswered and that we’ll have to get answers to in the near future.”
The B-CLEAR study was sponsored by GlaxoSmithKline. The REEF-2 study was sponsored by Janssen Research & Development. The VIR-3434 studies were funded by Vir Biotechnology. Dr. Pawlotsky has received grant and research support, acted as a consultant, adviser, or speaker, and participated in advisory boards for multiple pharmaceutical and biotechnology companies. This news organization was unable to verify Dr. Agarwal’s ties to Vir Biotechnology, but he presented one of the posters on VIR-3434 at the meeting and has been involved in the phase 1 study that was reported.
LONDON – Achieving a functional cure for hepatitis B virus (HBV) is not going to be easily achieved with the drugs that are currently in development, according to a presentation at the annual International Liver Congress sponsored by the European Association for the Study of the Liver.
“Intriguing results have been presented at ILC 2022 that must be carefully interpreted,” said Jean-Michel Pawlotsky, MD, PhD, of Henri Mondor Hospital in Créteil, France, during the viral hepatitis highlights session on the closing day of the meeting.
“New HBV drug development looks more complicated than initially expected and its goals and strategies need to be redefined and refocused,” he added
“This is really something that came from the discussions we had during the sessions but also in the corridors,” Dr. Pawlotsky added. “We know it’s going to be difficult; we have to reset, restart – not from zero, but from not much – and revise our strategy,” he suggested.
There are many new drugs under investigation for HBV, Dr. Pawlotsky said, noting that the number of studies being presented at the meeting was reminiscent of the flurry of activity before a functional cure for hepatitis C had been found. “It’s good to see that this is happening again for HBV,” he said.
Indeed, there are many new direct-acting antiviral agents, immunomodulatory, or other approaches being tested, and some of the more advanced studies are “teaching us a few things and probably raising more questions than getting answers,” Dr. Pawlotsky said.
The B-CLEAR study
One these studies is the phase 2b B-CLEAR study presented during the late-breaker session. This study involved bepirovirsen, an antisense oligonucleotide, and tested its efficacy and safety in patients with chronic hepatitis B virus infection who were either on or off stable nucleos(t)ide analogue (NA/NUC) therapy.
A similar proportion (28% and 29%, respectively) of patients achieved an hepatitis B surface antigen (HBsAg) level below the lower limit of quantification at the end of 24 weeks treatment. However, the effect on HBsAg varied according to the treatment arm, with changes to the dosing or switching to placebo indicating that the effect might wane when the treatment is stopped or if the dose is reduced.
“Interestingly, ALT elevations were observed in association with most HBsAg declines,” Dr. Pawlotsky pointed out. “I think we still have to determine whether this is good flare/bad flare, good sign/bad sign, of what is going to happen afterward.”
The REEF studies
Another approach highlighted was the combination of the silencing or small interfering RNA (siRNA) JNJ-3989 with the capsid assembly modulator (CAM) JNJ-6379 in the phase 2 REEF-1 and REEF-2 studies.
REEF-1, conducted in patients who were either hepatitis B e antigen (HBeAg) positive or negative who were not treated with NA/NUC or were NA/NUC suppressed, showed a dose-dependent, but variable effect among individual patients as might be expected at the end of 48 weeks’ treatment. This was sustained at week 72, which was 24 weeks’ follow-up after stopping treatment.
However, pointed out Dr. Pawlotsky “I think the most important part of this is that if you add a CAM on top of the siRNA, you do not improve the effect on HBsAg levels.”
Then there is the REEF-2 study, testing the same combination but in only patients who were NA suppressed or HBeAg negative alongside standard NA/NUC therapy. As well as being the first novel combination treatment trial to report, this was essentially a stopping trial, Kosh Agarwal, BMedSci (Hons), MBBS, MD, one of the study’s investigators explained separately at a media briefing.
Patients (n = 130) were treated for 48 weeks, then all treatment – including NA/NUC – was discontinued, with 48 weeks of follow-up after discontinuation, said Dr. Agarwal, who is a consultant hepatologist based at the Institute of Liver Studies at King’s College Hospital, London. He presented data from the first 24 week period after treatment had ended.
At the end of treatment, the combination had resulted in a mean reduction in HBsAg of 1.89 log10 IU/mL versus a reduction of 0.06 for the NA/NUC-only group, which acted as the control group in this trial. But “no patient in this study lost their surface antigen, i.e., were cured of their hepatitis B in the active arm or in the control arm,” Dr. Agarwal said.
“We didn’t achieve a cure, but a significant proportion were in a ‘controlled’ viral stage,” said Dr. Agarwal. Indeed, during his presentation of the findings, he showed that HBsAg inhibition was maintained in the majority (72%) of patients after stopping the combination.
While the trial’s primary endpoint wasn’t met, “it’s a really important study,” said Dr. Agarwal. “This [study] was fulfilled and delivered in the COVID era, so a lot of patients were looked after very carefully by sites in Europe,” he observed.
Further follow-up from the trial is expected, and Dr. Agarwal said that the subsequent discussion will “take us back to the drawing board to think about whether we need better antiviral treatments or whether we need to think about different combinations, and whether actually stopping treatment with every treatment is the right strategy to take.”
Both Dr. Agarwal and Dr. Pawlotsky flagged up the case of one patient in the trial who had been in the control arm and had experienced severe HBV reactivation that required a liver transplant.
“This patient is a warning signal,” Dr. Pawlotsky suggested in his talk. “When we think about NUC stopping, we have to think about the potential benefit in terms of HbsAg loss but also the potential risks.”
While Dr. Agarwal had noted that it highlights that “careful design of retreatment criteria is important in studies assessing the NA/NUC-stopping concept”.
Monoclonal antibody shows promise
Other combinations could involve an siRNA and an immunomodulatory agent and, during the poster sessions at the meeting, Dr. Agarwal also presented data from an ongoing phase 1 study with a novel, neutralizing monoclonal antibody called VIR-3434.
This monoclonal antibody is novel because it is thought to have several modes of action, first by binding to HBV and affecting its entry into liver cells, then by presenting the virus to T cells and stimulating a ‘vaccinal’ or immune effect, and then by helping the with the clearance of HBsAg and delivery of the virus to dendritic cells.
In the study, single doses of VIR-3434 were found to be well tolerated and to produce rapid reductions in HBsAg, with the highest dose used (300 mg) producing the greatest and most durable effect up to week 8.
VIR-3434 is also being tested in combination with other drugs in the phase 2 MARCH trial. One of these combinations is VIR-3434 together with an investigational siRNA dubbed VIR-2218. Preclinical work presented at ILC 2022 suggests that this combination appears to be capable of reducing HBsAg to a greater extent than using either agent alone.
Rethinking the strategy to get to a cure
Of course, VIR-3434 is one of several immunomodulatory compounds in development. There are therapeutic vaccines, drugs targeting the innate immune response, other monoclonal antibodies, T-cell receptors, checkpoint inhibitors and PD-L1 inhibitors. Then there are other compounds such as entry inhibitors, apoptosis inducers, and farnesoid X receptor agonists.
“I finish this meeting with more questions than answers,” Dr. Pawlotsky said. “What is the right target to enhance specific anti-HBV immunity? Does in vivo induction of immune responses translate into any beneficial effect on HBV infection? Will therapeutic vaccines every work in a viral infection?”
Moreover, he asked, “how can we avoid the side effect of enhancing multiple and complex nonspecific immune responses? Are treatment-induced flares good flares or bad flares? All of these are questions that are really unanswered and that we’ll have to get answers to in the near future.”
The B-CLEAR study was sponsored by GlaxoSmithKline. The REEF-2 study was sponsored by Janssen Research & Development. The VIR-3434 studies were funded by Vir Biotechnology. Dr. Pawlotsky has received grant and research support, acted as a consultant, adviser, or speaker, and participated in advisory boards for multiple pharmaceutical and biotechnology companies. This news organization was unable to verify Dr. Agarwal’s ties to Vir Biotechnology, but he presented one of the posters on VIR-3434 at the meeting and has been involved in the phase 1 study that was reported.
AT ILC 2022
Statins in NAFLD: Taking a closer look at benefits
LONDON – Substantial reductions in liver fat and fibrosis can be achieved with statin therapy, according to research presented at the annual International Liver Congress sponsored by the European Association for the Study of the Liver.
Statins are thought to have multiple beneficial actions in people with fatty liver disease, but there has been little insight into how they may be exerting such effects.
Now, data from the Rotterdam Study and others suggest that statins may be reducing the formation of lipid droplets as well as influencing the expression of important inflammatory genes.
The results “require further confirmation,” the team behind the work said, which was done at Erasmus Medical Center in Rotterdam, the Netherlands, in collaboration with researchers at The First Affiliated Hospital of Wenzhou (China) Medical University.
“Statins are inversely associated with multiple components of the NAFLD [nonalcoholic fatty liver disease] spectrum,” said Ibrahim Ayada, a PhD student in the department of gastroenterology and hepatology at Erasmus MC.
“Statins can inhibit lipid synthesis in organoids and statins also exhibit healthy inflammatory effects, which might contribute to the hepatoprotective effects that we observe in our population studies,” Mr. Ayada said.
A rising problem that needs addressing
Together NAFLD and NASH constitute a significant and increasing health burden, Mr. Ayada observed, noting that there were an estimated 64 million people in the United States and 52 million people in Europe, at least, with the rise mirroring the obesity pandemic.
“The number of patients visiting outpatient clinics has nearly doubled within a study period of 5 years,” he said.
“There is no pharmacologic therapy,” he reminded his audience, observing that fatty liver disease was a major indication for liver transplantation.
Statins are a long-standing staple of cardiovascular disease management and are known to have pleiotropic effects, Mr. Ayada explained. Their use in NAFLD and non-alcoholic steatohepatitis (NASH) has been purported but is supported by inconclusive evidence.
Indeed, a prior Cochrane review performed in 2013 found only two studies that were eligible for analysis and had “high risk of bias and a small numbers of participants,” according to the review’s authors.
Examining the connection
To look at the possible benefits of statins in people with NASH and examine how these effects might be occurring, Mr. Ayada and collaborators first took data from the Rotterdam Study, a large population-based prospective cohort that has been collecting data on its participants since the early 90s.
Data on over 4,500 participants were examined and of these, just over 1,000 had NAFLD. Statin versus no statin use was found to be associated with around a 30% reduction in fatty liver disease, with an odds ratio or 0.72 for NAFLD.
Then, looking only at a subset of patients with biopsy-proven NAFLD, statin use was associated with a 45% reduction in NASH (OR, 0.55) and a 24% reduction in fibrosis, although only the NASH reduction was significant (P = .031). The purpose of this cohort is to look at potential biomarkers and all participants had donated blood, urine, and stool samples; all were of Chinese descent, Mr. Ayada said.
“We then pooled our results with existing evidence in a meta-analysis,” said Mr. Ayada, including 16 studies. While results showed an overall inverse association, only the findings for a reduction in NAFLD and fibrosis were significant; the relationship between statins and NASH was not significant.
Investigating mechanistic effects
Then, for the second part of their work, Mr. Ayada and associates looked at potential mechanistic effects of statins.
“We did part two because we knew part one was going to be cross-sectional and we could only show the association and not causality, so we tried to shed some light on possible pathways,” he said.
To do this they used a novel model of liver organoids developed to study fatty liver disease and test potential therapeutics. In this model human liver organoids are exposed to sodium lactate, sodium pyruvate, and octanoic acid, which induce the formation of lipid droplets. Exposing the organoids to statins – simvastatin and lovastatin were used in the experiments – resulted in a reduced number of the induced lipid droplets.
“Although all concentrations of statins significantly inhibited the lipid size versus the control, the major effect was quite modest,” observed Mr. Ayada. The effect was most noticeable at the highest dose used (10 micromolar), and what they think might be happening is that the statins are clearing the smaller droplets first, leaving the larger ones behind.
Next, they looked at the effect of statin treatment on inflammatory gene expression in liver-derived monocytes. These will turn into macrophages and play a key role in chronic inflammation, Mr. Ayada explained. Initial results suggest that several proinflammatory cytokines such as interleukin-1 beta, IL-6, and IL-8 may be downplayed by statin therapy.
An anti-inflammatory effect of statins was also reported in unrelated poster presentations at the congress. While researchers Seul Ki Han and associated from South Korea showed an anti-inflammatory effect of a combination of simvastatin and ezetimibe (SAT083), a Dutch team found that atorvastatin reduced the infiltration of hepatic macrophages, neutrophils, and monocytes, as well as lowering levels of proinflammatory cytokines (SAT033).
Statins for NASH – a missed opportunity?
“As far as I am aware there is no robust evidence from large, randomized trials to suggest statins lessen chances of NAFLD, or improve its surrogate markers such as ALT or GGT [gamma-glutamyltransferase] levels,” Naveed Sattar, PhD, FRCP, FRCPath, FRSE, FMedSci, commented in an interview.
“The Rotterdam study is merely cross-sectional and cannot answer the question of causality,” added Dr. Sattar, who is professor of metabolic medicine and Honorary Consultant in Cardiovascular & Medical Science at the University of Glasgow. “It may be people who have less NAFLD are more likely to be prescribed statins, perhaps because doctors are wary of prescribing statins to those with slightly deranged liver tests,” he qualified.
Moreover, said Dr. Sattar, “prior evidence shows statins are underused in people with heart disease but who have NAFLD, which represents a missed opportunity to prevent heart disease.
“If statins had positive effects for preventing conversion of NAFLD to NASH or lessening fibrosis, I believe we would have known that by now.”
As for use of statins in future treatments of fatty liver disease, Dr. Sattar said: “I would not pin my hopes on statins to improve liver health, but doctors need to remember statins are safe in people with NAFLD or NASH and they should not be withheld in those who have existing cardiovascular disease or at elevated risk.”
The study received no commercial funding. Mr. Ayada and Dr. Sattar had no relevant conflicts of interest.
LONDON – Substantial reductions in liver fat and fibrosis can be achieved with statin therapy, according to research presented at the annual International Liver Congress sponsored by the European Association for the Study of the Liver.
Statins are thought to have multiple beneficial actions in people with fatty liver disease, but there has been little insight into how they may be exerting such effects.
Now, data from the Rotterdam Study and others suggest that statins may be reducing the formation of lipid droplets as well as influencing the expression of important inflammatory genes.
The results “require further confirmation,” the team behind the work said, which was done at Erasmus Medical Center in Rotterdam, the Netherlands, in collaboration with researchers at The First Affiliated Hospital of Wenzhou (China) Medical University.
“Statins are inversely associated with multiple components of the NAFLD [nonalcoholic fatty liver disease] spectrum,” said Ibrahim Ayada, a PhD student in the department of gastroenterology and hepatology at Erasmus MC.
“Statins can inhibit lipid synthesis in organoids and statins also exhibit healthy inflammatory effects, which might contribute to the hepatoprotective effects that we observe in our population studies,” Mr. Ayada said.
A rising problem that needs addressing
Together NAFLD and NASH constitute a significant and increasing health burden, Mr. Ayada observed, noting that there were an estimated 64 million people in the United States and 52 million people in Europe, at least, with the rise mirroring the obesity pandemic.
“The number of patients visiting outpatient clinics has nearly doubled within a study period of 5 years,” he said.
“There is no pharmacologic therapy,” he reminded his audience, observing that fatty liver disease was a major indication for liver transplantation.
Statins are a long-standing staple of cardiovascular disease management and are known to have pleiotropic effects, Mr. Ayada explained. Their use in NAFLD and non-alcoholic steatohepatitis (NASH) has been purported but is supported by inconclusive evidence.
Indeed, a prior Cochrane review performed in 2013 found only two studies that were eligible for analysis and had “high risk of bias and a small numbers of participants,” according to the review’s authors.
Examining the connection
To look at the possible benefits of statins in people with NASH and examine how these effects might be occurring, Mr. Ayada and collaborators first took data from the Rotterdam Study, a large population-based prospective cohort that has been collecting data on its participants since the early 90s.
Data on over 4,500 participants were examined and of these, just over 1,000 had NAFLD. Statin versus no statin use was found to be associated with around a 30% reduction in fatty liver disease, with an odds ratio or 0.72 for NAFLD.
Then, looking only at a subset of patients with biopsy-proven NAFLD, statin use was associated with a 45% reduction in NASH (OR, 0.55) and a 24% reduction in fibrosis, although only the NASH reduction was significant (P = .031). The purpose of this cohort is to look at potential biomarkers and all participants had donated blood, urine, and stool samples; all were of Chinese descent, Mr. Ayada said.
“We then pooled our results with existing evidence in a meta-analysis,” said Mr. Ayada, including 16 studies. While results showed an overall inverse association, only the findings for a reduction in NAFLD and fibrosis were significant; the relationship between statins and NASH was not significant.
Investigating mechanistic effects
Then, for the second part of their work, Mr. Ayada and associates looked at potential mechanistic effects of statins.
“We did part two because we knew part one was going to be cross-sectional and we could only show the association and not causality, so we tried to shed some light on possible pathways,” he said.
To do this they used a novel model of liver organoids developed to study fatty liver disease and test potential therapeutics. In this model human liver organoids are exposed to sodium lactate, sodium pyruvate, and octanoic acid, which induce the formation of lipid droplets. Exposing the organoids to statins – simvastatin and lovastatin were used in the experiments – resulted in a reduced number of the induced lipid droplets.
“Although all concentrations of statins significantly inhibited the lipid size versus the control, the major effect was quite modest,” observed Mr. Ayada. The effect was most noticeable at the highest dose used (10 micromolar), and what they think might be happening is that the statins are clearing the smaller droplets first, leaving the larger ones behind.
Next, they looked at the effect of statin treatment on inflammatory gene expression in liver-derived monocytes. These will turn into macrophages and play a key role in chronic inflammation, Mr. Ayada explained. Initial results suggest that several proinflammatory cytokines such as interleukin-1 beta, IL-6, and IL-8 may be downplayed by statin therapy.
An anti-inflammatory effect of statins was also reported in unrelated poster presentations at the congress. While researchers Seul Ki Han and associated from South Korea showed an anti-inflammatory effect of a combination of simvastatin and ezetimibe (SAT083), a Dutch team found that atorvastatin reduced the infiltration of hepatic macrophages, neutrophils, and monocytes, as well as lowering levels of proinflammatory cytokines (SAT033).
Statins for NASH – a missed opportunity?
“As far as I am aware there is no robust evidence from large, randomized trials to suggest statins lessen chances of NAFLD, or improve its surrogate markers such as ALT or GGT [gamma-glutamyltransferase] levels,” Naveed Sattar, PhD, FRCP, FRCPath, FRSE, FMedSci, commented in an interview.
“The Rotterdam study is merely cross-sectional and cannot answer the question of causality,” added Dr. Sattar, who is professor of metabolic medicine and Honorary Consultant in Cardiovascular & Medical Science at the University of Glasgow. “It may be people who have less NAFLD are more likely to be prescribed statins, perhaps because doctors are wary of prescribing statins to those with slightly deranged liver tests,” he qualified.
Moreover, said Dr. Sattar, “prior evidence shows statins are underused in people with heart disease but who have NAFLD, which represents a missed opportunity to prevent heart disease.
“If statins had positive effects for preventing conversion of NAFLD to NASH or lessening fibrosis, I believe we would have known that by now.”
As for use of statins in future treatments of fatty liver disease, Dr. Sattar said: “I would not pin my hopes on statins to improve liver health, but doctors need to remember statins are safe in people with NAFLD or NASH and they should not be withheld in those who have existing cardiovascular disease or at elevated risk.”
The study received no commercial funding. Mr. Ayada and Dr. Sattar had no relevant conflicts of interest.
LONDON – Substantial reductions in liver fat and fibrosis can be achieved with statin therapy, according to research presented at the annual International Liver Congress sponsored by the European Association for the Study of the Liver.
Statins are thought to have multiple beneficial actions in people with fatty liver disease, but there has been little insight into how they may be exerting such effects.
Now, data from the Rotterdam Study and others suggest that statins may be reducing the formation of lipid droplets as well as influencing the expression of important inflammatory genes.
The results “require further confirmation,” the team behind the work said, which was done at Erasmus Medical Center in Rotterdam, the Netherlands, in collaboration with researchers at The First Affiliated Hospital of Wenzhou (China) Medical University.
“Statins are inversely associated with multiple components of the NAFLD [nonalcoholic fatty liver disease] spectrum,” said Ibrahim Ayada, a PhD student in the department of gastroenterology and hepatology at Erasmus MC.
“Statins can inhibit lipid synthesis in organoids and statins also exhibit healthy inflammatory effects, which might contribute to the hepatoprotective effects that we observe in our population studies,” Mr. Ayada said.
A rising problem that needs addressing
Together NAFLD and NASH constitute a significant and increasing health burden, Mr. Ayada observed, noting that there were an estimated 64 million people in the United States and 52 million people in Europe, at least, with the rise mirroring the obesity pandemic.
“The number of patients visiting outpatient clinics has nearly doubled within a study period of 5 years,” he said.
“There is no pharmacologic therapy,” he reminded his audience, observing that fatty liver disease was a major indication for liver transplantation.
Statins are a long-standing staple of cardiovascular disease management and are known to have pleiotropic effects, Mr. Ayada explained. Their use in NAFLD and non-alcoholic steatohepatitis (NASH) has been purported but is supported by inconclusive evidence.
Indeed, a prior Cochrane review performed in 2013 found only two studies that were eligible for analysis and had “high risk of bias and a small numbers of participants,” according to the review’s authors.
Examining the connection
To look at the possible benefits of statins in people with NASH and examine how these effects might be occurring, Mr. Ayada and collaborators first took data from the Rotterdam Study, a large population-based prospective cohort that has been collecting data on its participants since the early 90s.
Data on over 4,500 participants were examined and of these, just over 1,000 had NAFLD. Statin versus no statin use was found to be associated with around a 30% reduction in fatty liver disease, with an odds ratio or 0.72 for NAFLD.
Then, looking only at a subset of patients with biopsy-proven NAFLD, statin use was associated with a 45% reduction in NASH (OR, 0.55) and a 24% reduction in fibrosis, although only the NASH reduction was significant (P = .031). The purpose of this cohort is to look at potential biomarkers and all participants had donated blood, urine, and stool samples; all were of Chinese descent, Mr. Ayada said.
“We then pooled our results with existing evidence in a meta-analysis,” said Mr. Ayada, including 16 studies. While results showed an overall inverse association, only the findings for a reduction in NAFLD and fibrosis were significant; the relationship between statins and NASH was not significant.
Investigating mechanistic effects
Then, for the second part of their work, Mr. Ayada and associates looked at potential mechanistic effects of statins.
“We did part two because we knew part one was going to be cross-sectional and we could only show the association and not causality, so we tried to shed some light on possible pathways,” he said.
To do this they used a novel model of liver organoids developed to study fatty liver disease and test potential therapeutics. In this model human liver organoids are exposed to sodium lactate, sodium pyruvate, and octanoic acid, which induce the formation of lipid droplets. Exposing the organoids to statins – simvastatin and lovastatin were used in the experiments – resulted in a reduced number of the induced lipid droplets.
“Although all concentrations of statins significantly inhibited the lipid size versus the control, the major effect was quite modest,” observed Mr. Ayada. The effect was most noticeable at the highest dose used (10 micromolar), and what they think might be happening is that the statins are clearing the smaller droplets first, leaving the larger ones behind.
Next, they looked at the effect of statin treatment on inflammatory gene expression in liver-derived monocytes. These will turn into macrophages and play a key role in chronic inflammation, Mr. Ayada explained. Initial results suggest that several proinflammatory cytokines such as interleukin-1 beta, IL-6, and IL-8 may be downplayed by statin therapy.
An anti-inflammatory effect of statins was also reported in unrelated poster presentations at the congress. While researchers Seul Ki Han and associated from South Korea showed an anti-inflammatory effect of a combination of simvastatin and ezetimibe (SAT083), a Dutch team found that atorvastatin reduced the infiltration of hepatic macrophages, neutrophils, and monocytes, as well as lowering levels of proinflammatory cytokines (SAT033).
Statins for NASH – a missed opportunity?
“As far as I am aware there is no robust evidence from large, randomized trials to suggest statins lessen chances of NAFLD, or improve its surrogate markers such as ALT or GGT [gamma-glutamyltransferase] levels,” Naveed Sattar, PhD, FRCP, FRCPath, FRSE, FMedSci, commented in an interview.
“The Rotterdam study is merely cross-sectional and cannot answer the question of causality,” added Dr. Sattar, who is professor of metabolic medicine and Honorary Consultant in Cardiovascular & Medical Science at the University of Glasgow. “It may be people who have less NAFLD are more likely to be prescribed statins, perhaps because doctors are wary of prescribing statins to those with slightly deranged liver tests,” he qualified.
Moreover, said Dr. Sattar, “prior evidence shows statins are underused in people with heart disease but who have NAFLD, which represents a missed opportunity to prevent heart disease.
“If statins had positive effects for preventing conversion of NAFLD to NASH or lessening fibrosis, I believe we would have known that by now.”
As for use of statins in future treatments of fatty liver disease, Dr. Sattar said: “I would not pin my hopes on statins to improve liver health, but doctors need to remember statins are safe in people with NAFLD or NASH and they should not be withheld in those who have existing cardiovascular disease or at elevated risk.”
The study received no commercial funding. Mr. Ayada and Dr. Sattar had no relevant conflicts of interest.
AT ILC 2022