User login
Need blood STAT? Call for a drone
BOSTON – While Amazon and other retailers are experimenting with drones to deliver toasters and toilet seats to your doorstep, drone-delivered platelets and fresh frozen plasma may be coming soon to a hospital near you, experts said at AABB 2018, the annual meeting of the group formerly known as the American Association of Blood Banks.
Using a system of completely autonomous delivery drones launched from a central location, U.S.-based Zipline International delivers blood products to treat postpartum hemorrhage, trauma, malaria, and other life-threatening conditions to patients in rural Rwanda, according to company spokesman Chris Kenney.
“In less than 2 years in Rwanda, we’ve made almost 10,000 deliveries – that’s almost 20,000 units of blood,” he said.
One-third of all deliveries are needed for urgent, life-saving interventions, he said.
The system, which delivers 30% of all blood products used in Rwanda outside the capital Kigali, has resulted in 100% availability of blood products when needed, a 98% reduction in waste (i.e., when unused blood products are discarded because of age), and a 175% increase in the use of platelets and fresh frozen plasma, Mr. Kenney said.
Setting up an airborne delivery network in the largely unregulated and uncrowded Rwandan airspace was a relatively simple process, however, compared with the myriad challenges of establishing a similar system for deliveries to urban medical centers in Boston, Chicago, New York, or Los Angeles, said Paul Eastvold, MD, chief medical officer at Vitalant, a nonprofit network of community blood banks headquartered in Spokane, Wash.
Dr. Eastvold, who is also a private pilot, described the regulatory hurdles that will need to be surmounted before blood-delivery drones are as common a sight as traffic helicopters are currently. He added, however, “I can guarantee you that in the future this is going to be an applicable technology to our industry in one way, shape, or another.”
Fast and cheap
Speed and cost are two of the most compelling arguments for blood banks to use drones. Mr. Kenney described the case of a 24-year-old Rwandan woman who had uncontrolled bleeding from complications following a cesarean section. The clinicians treating her opted to give her an immediate red blood cell transfusion, but she continued to bleed, and the hospital ran out of red blood cells in about 15 minutes.
They placed an order for more blood products – ordering can be done by text message or via WhatsApp, a free, cross-platform messaging and voiceover IP calling service – and over the course of 90 minutes Zipline was able to deliver, using multiple drone launches, 7 units of red blood cells, 4 units of plasma, and 2 units of platelets, all of which were transfused into the patient and allowed her condition to stabilize.
Deliveries that would take a minimum of 3 hours by road can be accomplished in about 15-25 minutes by air, Mr. Kenney said.
The drones – more formally known as “unmanned aerial vehicles” (UAVs) – fly a loop starting at the distribution center, find their target, descend to a height of about 10 meters and drop the package, which has a parachute attached. Packages can be delivered within a drop zone the size of two parking spaces, even in gale-force winds, Mr. Kenney said.
“The whole process is 100% autonomous. The aircraft knows where it’s going, it knows what conditions [are], it knows what its payload characteristics are and flies to the delivery point and drops its package,” he explained.
As drones return to the distribution center, they are snared from the air with a wire that catches a small tail hook on the fuselage.
Airborne deliveries are also significantly cheaper than ground-based services for local delivery, Dr. Eastvold noted. He cited a study showing that the cost of ground shipping from a local warehouse by carriers such as UPS or FedEx could be $6 or more, drones could be as cheap as 5 cents per mile with delivery within about 30 minutes, he said.
The fly in the ointment
Dr. Eastvold outlined the significant barriers to adoption of drone-based delivery systems in the United States, ranging from differences in state laws about when, where, and how drones can be used and who can operate them, to Federal Aviation Administration airspace restrictions and regulations.
For example, the FAA currently requires “line-of-sight” operation only for most drone operators, meaning that the operator must have visual contact with the drone at all times. The FAA will, however, grant waivers to individual operators for specified flying conditions on a case-by-case basis, if compelling need or extenuating circumstances can be satisfactorily explained.
In addition, federal regulations require commercial drone pilots to be 16 years old or older, be fluent in English, be in a physical and mental condition that would not interfere with safe operation of a drone, pass an aeronautical knowledge exam at an FAA-approved testing center, and undergo a Transportation Safety Administration background security screening.
Despite these challenges, at least one U.S. medical center, Johns Hopkins University, is testing the use of drones for blood delivery. In 2017, they demonstrated that a drone could successfully deliver human blood samples in temperature-controlled conditions across 161 miles of Arizona desert, in a flight lasting 3 hours.
Mr. Kenney said that his company is developing a second distribution center in Rwanda that will expand coverage to the entire country and is also working with the FAA, federal regulators, and the state of North Carolina to develop a drone-based blood delivery system in the United States.
BOSTON – While Amazon and other retailers are experimenting with drones to deliver toasters and toilet seats to your doorstep, drone-delivered platelets and fresh frozen plasma may be coming soon to a hospital near you, experts said at AABB 2018, the annual meeting of the group formerly known as the American Association of Blood Banks.
Using a system of completely autonomous delivery drones launched from a central location, U.S.-based Zipline International delivers blood products to treat postpartum hemorrhage, trauma, malaria, and other life-threatening conditions to patients in rural Rwanda, according to company spokesman Chris Kenney.
“In less than 2 years in Rwanda, we’ve made almost 10,000 deliveries – that’s almost 20,000 units of blood,” he said.
One-third of all deliveries are needed for urgent, life-saving interventions, he said.
The system, which delivers 30% of all blood products used in Rwanda outside the capital Kigali, has resulted in 100% availability of blood products when needed, a 98% reduction in waste (i.e., when unused blood products are discarded because of age), and a 175% increase in the use of platelets and fresh frozen plasma, Mr. Kenney said.
Setting up an airborne delivery network in the largely unregulated and uncrowded Rwandan airspace was a relatively simple process, however, compared with the myriad challenges of establishing a similar system for deliveries to urban medical centers in Boston, Chicago, New York, or Los Angeles, said Paul Eastvold, MD, chief medical officer at Vitalant, a nonprofit network of community blood banks headquartered in Spokane, Wash.
Dr. Eastvold, who is also a private pilot, described the regulatory hurdles that will need to be surmounted before blood-delivery drones are as common a sight as traffic helicopters are currently. He added, however, “I can guarantee you that in the future this is going to be an applicable technology to our industry in one way, shape, or another.”
Fast and cheap
Speed and cost are two of the most compelling arguments for blood banks to use drones. Mr. Kenney described the case of a 24-year-old Rwandan woman who had uncontrolled bleeding from complications following a cesarean section. The clinicians treating her opted to give her an immediate red blood cell transfusion, but she continued to bleed, and the hospital ran out of red blood cells in about 15 minutes.
They placed an order for more blood products – ordering can be done by text message or via WhatsApp, a free, cross-platform messaging and voiceover IP calling service – and over the course of 90 minutes Zipline was able to deliver, using multiple drone launches, 7 units of red blood cells, 4 units of plasma, and 2 units of platelets, all of which were transfused into the patient and allowed her condition to stabilize.
Deliveries that would take a minimum of 3 hours by road can be accomplished in about 15-25 minutes by air, Mr. Kenney said.
The drones – more formally known as “unmanned aerial vehicles” (UAVs) – fly a loop starting at the distribution center, find their target, descend to a height of about 10 meters and drop the package, which has a parachute attached. Packages can be delivered within a drop zone the size of two parking spaces, even in gale-force winds, Mr. Kenney said.
“The whole process is 100% autonomous. The aircraft knows where it’s going, it knows what conditions [are], it knows what its payload characteristics are and flies to the delivery point and drops its package,” he explained.
As drones return to the distribution center, they are snared from the air with a wire that catches a small tail hook on the fuselage.
Airborne deliveries are also significantly cheaper than ground-based services for local delivery, Dr. Eastvold noted. He cited a study showing that the cost of ground shipping from a local warehouse by carriers such as UPS or FedEx could be $6 or more, drones could be as cheap as 5 cents per mile with delivery within about 30 minutes, he said.
The fly in the ointment
Dr. Eastvold outlined the significant barriers to adoption of drone-based delivery systems in the United States, ranging from differences in state laws about when, where, and how drones can be used and who can operate them, to Federal Aviation Administration airspace restrictions and regulations.
For example, the FAA currently requires “line-of-sight” operation only for most drone operators, meaning that the operator must have visual contact with the drone at all times. The FAA will, however, grant waivers to individual operators for specified flying conditions on a case-by-case basis, if compelling need or extenuating circumstances can be satisfactorily explained.
In addition, federal regulations require commercial drone pilots to be 16 years old or older, be fluent in English, be in a physical and mental condition that would not interfere with safe operation of a drone, pass an aeronautical knowledge exam at an FAA-approved testing center, and undergo a Transportation Safety Administration background security screening.
Despite these challenges, at least one U.S. medical center, Johns Hopkins University, is testing the use of drones for blood delivery. In 2017, they demonstrated that a drone could successfully deliver human blood samples in temperature-controlled conditions across 161 miles of Arizona desert, in a flight lasting 3 hours.
Mr. Kenney said that his company is developing a second distribution center in Rwanda that will expand coverage to the entire country and is also working with the FAA, federal regulators, and the state of North Carolina to develop a drone-based blood delivery system in the United States.
BOSTON – While Amazon and other retailers are experimenting with drones to deliver toasters and toilet seats to your doorstep, drone-delivered platelets and fresh frozen plasma may be coming soon to a hospital near you, experts said at AABB 2018, the annual meeting of the group formerly known as the American Association of Blood Banks.
Using a system of completely autonomous delivery drones launched from a central location, U.S.-based Zipline International delivers blood products to treat postpartum hemorrhage, trauma, malaria, and other life-threatening conditions to patients in rural Rwanda, according to company spokesman Chris Kenney.
“In less than 2 years in Rwanda, we’ve made almost 10,000 deliveries – that’s almost 20,000 units of blood,” he said.
One-third of all deliveries are needed for urgent, life-saving interventions, he said.
The system, which delivers 30% of all blood products used in Rwanda outside the capital Kigali, has resulted in 100% availability of blood products when needed, a 98% reduction in waste (i.e., when unused blood products are discarded because of age), and a 175% increase in the use of platelets and fresh frozen plasma, Mr. Kenney said.
Setting up an airborne delivery network in the largely unregulated and uncrowded Rwandan airspace was a relatively simple process, however, compared with the myriad challenges of establishing a similar system for deliveries to urban medical centers in Boston, Chicago, New York, or Los Angeles, said Paul Eastvold, MD, chief medical officer at Vitalant, a nonprofit network of community blood banks headquartered in Spokane, Wash.
Dr. Eastvold, who is also a private pilot, described the regulatory hurdles that will need to be surmounted before blood-delivery drones are as common a sight as traffic helicopters are currently. He added, however, “I can guarantee you that in the future this is going to be an applicable technology to our industry in one way, shape, or another.”
Fast and cheap
Speed and cost are two of the most compelling arguments for blood banks to use drones. Mr. Kenney described the case of a 24-year-old Rwandan woman who had uncontrolled bleeding from complications following a cesarean section. The clinicians treating her opted to give her an immediate red blood cell transfusion, but she continued to bleed, and the hospital ran out of red blood cells in about 15 minutes.
They placed an order for more blood products – ordering can be done by text message or via WhatsApp, a free, cross-platform messaging and voiceover IP calling service – and over the course of 90 minutes Zipline was able to deliver, using multiple drone launches, 7 units of red blood cells, 4 units of plasma, and 2 units of platelets, all of which were transfused into the patient and allowed her condition to stabilize.
Deliveries that would take a minimum of 3 hours by road can be accomplished in about 15-25 minutes by air, Mr. Kenney said.
The drones – more formally known as “unmanned aerial vehicles” (UAVs) – fly a loop starting at the distribution center, find their target, descend to a height of about 10 meters and drop the package, which has a parachute attached. Packages can be delivered within a drop zone the size of two parking spaces, even in gale-force winds, Mr. Kenney said.
“The whole process is 100% autonomous. The aircraft knows where it’s going, it knows what conditions [are], it knows what its payload characteristics are and flies to the delivery point and drops its package,” he explained.
As drones return to the distribution center, they are snared from the air with a wire that catches a small tail hook on the fuselage.
Airborne deliveries are also significantly cheaper than ground-based services for local delivery, Dr. Eastvold noted. He cited a study showing that the cost of ground shipping from a local warehouse by carriers such as UPS or FedEx could be $6 or more, drones could be as cheap as 5 cents per mile with delivery within about 30 minutes, he said.
The fly in the ointment
Dr. Eastvold outlined the significant barriers to adoption of drone-based delivery systems in the United States, ranging from differences in state laws about when, where, and how drones can be used and who can operate them, to Federal Aviation Administration airspace restrictions and regulations.
For example, the FAA currently requires “line-of-sight” operation only for most drone operators, meaning that the operator must have visual contact with the drone at all times. The FAA will, however, grant waivers to individual operators for specified flying conditions on a case-by-case basis, if compelling need or extenuating circumstances can be satisfactorily explained.
In addition, federal regulations require commercial drone pilots to be 16 years old or older, be fluent in English, be in a physical and mental condition that would not interfere with safe operation of a drone, pass an aeronautical knowledge exam at an FAA-approved testing center, and undergo a Transportation Safety Administration background security screening.
Despite these challenges, at least one U.S. medical center, Johns Hopkins University, is testing the use of drones for blood delivery. In 2017, they demonstrated that a drone could successfully deliver human blood samples in temperature-controlled conditions across 161 miles of Arizona desert, in a flight lasting 3 hours.
Mr. Kenney said that his company is developing a second distribution center in Rwanda that will expand coverage to the entire country and is also working with the FAA, federal regulators, and the state of North Carolina to develop a drone-based blood delivery system in the United States.
AT AABB 2018
Tech-based cancer company raises access concerns
Oncologists are raising concerns about care access after the launch of a new company that links patients to cancer care options and clinical trials through mobile technology.
Driver, which began in September in the U.S. and China, is a global technology platform that allows patients to access treatment options across a broad network of cancer centers without leaving home. Cancer patients join the platform using a mobile app, through which Driver obtains the required consent to acquire medical records and tumor samples, and the company uses the information to recommend treatment options and clinical trials.
A separate app called Driver for Clinic enables oncologists who belong to Driver’s partner hospitals to manage their institution’s clinical trial information and quickly filter that information based on patients’ medical history to determine the patient’s eligibility for treatments.
Driver’s mission is to connect more patients to the best cancer treatments, regardless of location, said Will Polkinghorn, MD, Driver cofounder and CEO.
“Driver’s cofounders met at Harvard Medical School [in Boston] and saw firsthand the challenges of patients getting access to the latest, cutting-edge treatments available,” Dr. Polkinghorn said in an interview. “As doctors, [we] also witnessed how difficult it was for doctors to manage information in clinic and know about all the treatments that become available all around the world. Driver was created as a platform, with an app for the patient and an app for the doctor, to solve this broken marketplace.”
As part of the model, patients can review their recommended treatment options through video with an expert oncologist and select a hospital within Driver’s network for further evaluation. The company’s global network includes more than 30 leading U.S. cancer centers, including the Cleveland Clinic; multiple locations of the Mayo Clinic; the University of California, San Francisco; and Massachusetts General Hospital, Boston. The U.S. National Cancer Institute (NCI) and the Chinese National Cancer Center are founding members of Driver’s global network, according to the company.
Making more information and treatment options available is a positive for patients, said Walter Stadler, MD, chief of hematology/oncology and director of the genitourinary oncology program at the University of Chicago. However, he noted that the cost for patients to use Driver is prohibitive for many patients. Driver charges patients $3,000 up front and then a $20 monthly fee to use its service. Insurance does not subsidize the cost, nor does Driver help with travel or treatment costs, according to its website.
“It’s inequality of access,” Dr. Stadler said in an interview. “Many of us are very concerned that the clinical trials currently being conducted do not represent the general population well because they don’t represent patients with disparities … Here, we further exacerbate the problem by saying, ‘Okay, we’ll take the 5% of patients who can afford the service and expand their access, and the others, well, that’s not our problem.’ ”
Kashyap Patel, MD, secretary for the Community Oncology Alliance and CEO for the Carolina Blood and Cancer Care in Rock Hill, S.C., also sees positives and negatives about the business model. Using technology to link patients with care and clinical trials can help speed treatment and accelerate drug development, he said. But Driver’s network of large tertiary care centers in metropolitan areas poses challenges for rural cancer patients, he said.
“Access to clinical trials for patients residing in rural areas, as well as those getting their treatment in community based clinics, would not change,” Dr. Patel said in an interview. “Hence, challenges of social and demographic disparities and inequalities in clinical trial access and participation would be altered minimally. There is much greater need for such [platforms to include] community cancer clinics that would be more inclusive and encompass larger geographic areas where the majority of patients receive their care.”
Disadvantaged populations with limited access are not being overlooked by the company, according to Driver leaders. A branch of the company called Driver for All aims to increase access to optimal treatments for free through partnerships with local communities, Dr. Polkinghorn said. Driver for All has thus far partnered with Howard University Hospital in Washington to connect Howard patients to clinical trials at NCI. A partnership with Beijing Children’s Hospital and the Futang Research Center of Pediatric Development, meanwhile, is working to connect patients with rare-disease experts. Driver has funded 100% of the cost of these projects to date, according to its website.
Outside of Driver for All, Dr. Polkinghorn acknowledges that patients must bear the cost of Driver’s consumer products; however, the price should be viewed in context, he said.
“It’s important to remember that today, in order to be evaluated by 30 [plus] centers for treatment options, patients would need to fly to these centers, make appointments, and be seen by a doctor – this would require both time and resources for flights/hotels, which would cost much more than our sticker price,” he said. “So while $3,000 is a lot of money for some patients, Driver’s product is ultimately able to provide more visibility to options that simply would not be realistic today.”
James Gulley, MD, of the National Cancer Institute Center for Cancer Research, said any platform that can efficiently provide access to clinical trial options yields another source of information for patients to utilize in decision making with their health provider. Dr. Gulley, who heads the center’s genitourinary malignancies branch, declined to comment about access-to-care concerns with Driver’s model. He emphasized that patients who participate in NIH research studies are treated without charge.
“The key to finding better [cancer] treatment is to perform science-driven clinical trials,” Dr. Gulley said in an interview. “However, there are many barriers for enrollment in clinical trials. … As a government agency, NCI is open to partnering with any organization that seeks to improve access to clinical trials for cancer patients.”
NCI and Driver recently conducted a study to validate Driver’s platform; it showed that Driver’s technology successfully predicted the eligibility of patients in NCI Center for Cancer Research clinical trials. The study, presented at a recent American Society of Clinical Oncology meeting, evaluated Driver’s processing of 21 metastatic prostate cancer patients enrolled in a therapeutic NCI clinical trial within the last five years. Results showed Driver correctly predicted that 20 of the patients were “potentially eligible” for the trial in which they were enrolled, and that one was ineligible. Based on the study, a protocol is now in development for a new clinical study, which will seek to further determine the efficiency and accuracy of the clinical trial access program created by Driver, according to Dr. Gulley.
Charles Ryan, MD, director of the division of hematology, oncology, and transplantation for the University of Minnesota, Minneapolis, views Driver’s platform as a way to eliminate geographical barriers, which often keep patients from care, while at the same time enabling researchers to find the right patients for clinical trials.
“We need breakthrough technologies and opportunities for patients to be able to access the most successful and promising cancer treatments, regardless of where they live,” Dr. Ryan said in an interview. “Companies like Driver are attempting to bridge that gap by connecting patients to doctors at world class cancer institutes and direct them toward the best care for their particular condition.”
Driver’s model also allows researchers the opportunity to develop specific, unique treatment for less common cancers and remain optimistic that they can attract patients to receive such treatments as they are developed, Dr. Ryan said.
Dr. Stadler, however, worries that Driver may be giving patients the wrong perception that all it takes is a computer and medical records to determine their best treatment route.
“There’s a lot more subtlety to treatment decisions than most people would like to admit,” Dr. Stadler said. “It’s more than just a bunch of data from sophisticated laboratory tests and the written medical record. Obtaining objective information is the first step, but it’s far from the only step.”
Patients may have significant limitations in functional status that is apparent only during an in-person assessment, for example, he said. In other cases, family members may be essential in conveying information about a patient’s cognitive disabilities. Even when such information is documented, it is sometimes difficult to extract the full picture from the record alone, he said. Dr. Stadler is also bothered that the model requires physicians and hospitals to provide their skilled analyses to a for-profit company, which in turn, charges patients to review the information.
“This is our work,” he said. “I agree that patients should have the information, and I don’t mind sharing anything I have with patients, but now I’m going to share it with another business that essentially is competing with me in terms of providing guidance to patients.”
Oncologists are raising concerns about care access after the launch of a new company that links patients to cancer care options and clinical trials through mobile technology.
Driver, which began in September in the U.S. and China, is a global technology platform that allows patients to access treatment options across a broad network of cancer centers without leaving home. Cancer patients join the platform using a mobile app, through which Driver obtains the required consent to acquire medical records and tumor samples, and the company uses the information to recommend treatment options and clinical trials.
A separate app called Driver for Clinic enables oncologists who belong to Driver’s partner hospitals to manage their institution’s clinical trial information and quickly filter that information based on patients’ medical history to determine the patient’s eligibility for treatments.
Driver’s mission is to connect more patients to the best cancer treatments, regardless of location, said Will Polkinghorn, MD, Driver cofounder and CEO.
“Driver’s cofounders met at Harvard Medical School [in Boston] and saw firsthand the challenges of patients getting access to the latest, cutting-edge treatments available,” Dr. Polkinghorn said in an interview. “As doctors, [we] also witnessed how difficult it was for doctors to manage information in clinic and know about all the treatments that become available all around the world. Driver was created as a platform, with an app for the patient and an app for the doctor, to solve this broken marketplace.”
As part of the model, patients can review their recommended treatment options through video with an expert oncologist and select a hospital within Driver’s network for further evaluation. The company’s global network includes more than 30 leading U.S. cancer centers, including the Cleveland Clinic; multiple locations of the Mayo Clinic; the University of California, San Francisco; and Massachusetts General Hospital, Boston. The U.S. National Cancer Institute (NCI) and the Chinese National Cancer Center are founding members of Driver’s global network, according to the company.
Making more information and treatment options available is a positive for patients, said Walter Stadler, MD, chief of hematology/oncology and director of the genitourinary oncology program at the University of Chicago. However, he noted that the cost for patients to use Driver is prohibitive for many patients. Driver charges patients $3,000 up front and then a $20 monthly fee to use its service. Insurance does not subsidize the cost, nor does Driver help with travel or treatment costs, according to its website.
“It’s inequality of access,” Dr. Stadler said in an interview. “Many of us are very concerned that the clinical trials currently being conducted do not represent the general population well because they don’t represent patients with disparities … Here, we further exacerbate the problem by saying, ‘Okay, we’ll take the 5% of patients who can afford the service and expand their access, and the others, well, that’s not our problem.’ ”
Kashyap Patel, MD, secretary for the Community Oncology Alliance and CEO for the Carolina Blood and Cancer Care in Rock Hill, S.C., also sees positives and negatives about the business model. Using technology to link patients with care and clinical trials can help speed treatment and accelerate drug development, he said. But Driver’s network of large tertiary care centers in metropolitan areas poses challenges for rural cancer patients, he said.
“Access to clinical trials for patients residing in rural areas, as well as those getting their treatment in community based clinics, would not change,” Dr. Patel said in an interview. “Hence, challenges of social and demographic disparities and inequalities in clinical trial access and participation would be altered minimally. There is much greater need for such [platforms to include] community cancer clinics that would be more inclusive and encompass larger geographic areas where the majority of patients receive their care.”
Disadvantaged populations with limited access are not being overlooked by the company, according to Driver leaders. A branch of the company called Driver for All aims to increase access to optimal treatments for free through partnerships with local communities, Dr. Polkinghorn said. Driver for All has thus far partnered with Howard University Hospital in Washington to connect Howard patients to clinical trials at NCI. A partnership with Beijing Children’s Hospital and the Futang Research Center of Pediatric Development, meanwhile, is working to connect patients with rare-disease experts. Driver has funded 100% of the cost of these projects to date, according to its website.
Outside of Driver for All, Dr. Polkinghorn acknowledges that patients must bear the cost of Driver’s consumer products; however, the price should be viewed in context, he said.
“It’s important to remember that today, in order to be evaluated by 30 [plus] centers for treatment options, patients would need to fly to these centers, make appointments, and be seen by a doctor – this would require both time and resources for flights/hotels, which would cost much more than our sticker price,” he said. “So while $3,000 is a lot of money for some patients, Driver’s product is ultimately able to provide more visibility to options that simply would not be realistic today.”
James Gulley, MD, of the National Cancer Institute Center for Cancer Research, said any platform that can efficiently provide access to clinical trial options yields another source of information for patients to utilize in decision making with their health provider. Dr. Gulley, who heads the center’s genitourinary malignancies branch, declined to comment about access-to-care concerns with Driver’s model. He emphasized that patients who participate in NIH research studies are treated without charge.
“The key to finding better [cancer] treatment is to perform science-driven clinical trials,” Dr. Gulley said in an interview. “However, there are many barriers for enrollment in clinical trials. … As a government agency, NCI is open to partnering with any organization that seeks to improve access to clinical trials for cancer patients.”
NCI and Driver recently conducted a study to validate Driver’s platform; it showed that Driver’s technology successfully predicted the eligibility of patients in NCI Center for Cancer Research clinical trials. The study, presented at a recent American Society of Clinical Oncology meeting, evaluated Driver’s processing of 21 metastatic prostate cancer patients enrolled in a therapeutic NCI clinical trial within the last five years. Results showed Driver correctly predicted that 20 of the patients were “potentially eligible” for the trial in which they were enrolled, and that one was ineligible. Based on the study, a protocol is now in development for a new clinical study, which will seek to further determine the efficiency and accuracy of the clinical trial access program created by Driver, according to Dr. Gulley.
Charles Ryan, MD, director of the division of hematology, oncology, and transplantation for the University of Minnesota, Minneapolis, views Driver’s platform as a way to eliminate geographical barriers, which often keep patients from care, while at the same time enabling researchers to find the right patients for clinical trials.
“We need breakthrough technologies and opportunities for patients to be able to access the most successful and promising cancer treatments, regardless of where they live,” Dr. Ryan said in an interview. “Companies like Driver are attempting to bridge that gap by connecting patients to doctors at world class cancer institutes and direct them toward the best care for their particular condition.”
Driver’s model also allows researchers the opportunity to develop specific, unique treatment for less common cancers and remain optimistic that they can attract patients to receive such treatments as they are developed, Dr. Ryan said.
Dr. Stadler, however, worries that Driver may be giving patients the wrong perception that all it takes is a computer and medical records to determine their best treatment route.
“There’s a lot more subtlety to treatment decisions than most people would like to admit,” Dr. Stadler said. “It’s more than just a bunch of data from sophisticated laboratory tests and the written medical record. Obtaining objective information is the first step, but it’s far from the only step.”
Patients may have significant limitations in functional status that is apparent only during an in-person assessment, for example, he said. In other cases, family members may be essential in conveying information about a patient’s cognitive disabilities. Even when such information is documented, it is sometimes difficult to extract the full picture from the record alone, he said. Dr. Stadler is also bothered that the model requires physicians and hospitals to provide their skilled analyses to a for-profit company, which in turn, charges patients to review the information.
“This is our work,” he said. “I agree that patients should have the information, and I don’t mind sharing anything I have with patients, but now I’m going to share it with another business that essentially is competing with me in terms of providing guidance to patients.”
Oncologists are raising concerns about care access after the launch of a new company that links patients to cancer care options and clinical trials through mobile technology.
Driver, which began in September in the U.S. and China, is a global technology platform that allows patients to access treatment options across a broad network of cancer centers without leaving home. Cancer patients join the platform using a mobile app, through which Driver obtains the required consent to acquire medical records and tumor samples, and the company uses the information to recommend treatment options and clinical trials.
A separate app called Driver for Clinic enables oncologists who belong to Driver’s partner hospitals to manage their institution’s clinical trial information and quickly filter that information based on patients’ medical history to determine the patient’s eligibility for treatments.
Driver’s mission is to connect more patients to the best cancer treatments, regardless of location, said Will Polkinghorn, MD, Driver cofounder and CEO.
“Driver’s cofounders met at Harvard Medical School [in Boston] and saw firsthand the challenges of patients getting access to the latest, cutting-edge treatments available,” Dr. Polkinghorn said in an interview. “As doctors, [we] also witnessed how difficult it was for doctors to manage information in clinic and know about all the treatments that become available all around the world. Driver was created as a platform, with an app for the patient and an app for the doctor, to solve this broken marketplace.”
As part of the model, patients can review their recommended treatment options through video with an expert oncologist and select a hospital within Driver’s network for further evaluation. The company’s global network includes more than 30 leading U.S. cancer centers, including the Cleveland Clinic; multiple locations of the Mayo Clinic; the University of California, San Francisco; and Massachusetts General Hospital, Boston. The U.S. National Cancer Institute (NCI) and the Chinese National Cancer Center are founding members of Driver’s global network, according to the company.
Making more information and treatment options available is a positive for patients, said Walter Stadler, MD, chief of hematology/oncology and director of the genitourinary oncology program at the University of Chicago. However, he noted that the cost for patients to use Driver is prohibitive for many patients. Driver charges patients $3,000 up front and then a $20 monthly fee to use its service. Insurance does not subsidize the cost, nor does Driver help with travel or treatment costs, according to its website.
“It’s inequality of access,” Dr. Stadler said in an interview. “Many of us are very concerned that the clinical trials currently being conducted do not represent the general population well because they don’t represent patients with disparities … Here, we further exacerbate the problem by saying, ‘Okay, we’ll take the 5% of patients who can afford the service and expand their access, and the others, well, that’s not our problem.’ ”
Kashyap Patel, MD, secretary for the Community Oncology Alliance and CEO for the Carolina Blood and Cancer Care in Rock Hill, S.C., also sees positives and negatives about the business model. Using technology to link patients with care and clinical trials can help speed treatment and accelerate drug development, he said. But Driver’s network of large tertiary care centers in metropolitan areas poses challenges for rural cancer patients, he said.
“Access to clinical trials for patients residing in rural areas, as well as those getting their treatment in community based clinics, would not change,” Dr. Patel said in an interview. “Hence, challenges of social and demographic disparities and inequalities in clinical trial access and participation would be altered minimally. There is much greater need for such [platforms to include] community cancer clinics that would be more inclusive and encompass larger geographic areas where the majority of patients receive their care.”
Disadvantaged populations with limited access are not being overlooked by the company, according to Driver leaders. A branch of the company called Driver for All aims to increase access to optimal treatments for free through partnerships with local communities, Dr. Polkinghorn said. Driver for All has thus far partnered with Howard University Hospital in Washington to connect Howard patients to clinical trials at NCI. A partnership with Beijing Children’s Hospital and the Futang Research Center of Pediatric Development, meanwhile, is working to connect patients with rare-disease experts. Driver has funded 100% of the cost of these projects to date, according to its website.
Outside of Driver for All, Dr. Polkinghorn acknowledges that patients must bear the cost of Driver’s consumer products; however, the price should be viewed in context, he said.
“It’s important to remember that today, in order to be evaluated by 30 [plus] centers for treatment options, patients would need to fly to these centers, make appointments, and be seen by a doctor – this would require both time and resources for flights/hotels, which would cost much more than our sticker price,” he said. “So while $3,000 is a lot of money for some patients, Driver’s product is ultimately able to provide more visibility to options that simply would not be realistic today.”
James Gulley, MD, of the National Cancer Institute Center for Cancer Research, said any platform that can efficiently provide access to clinical trial options yields another source of information for patients to utilize in decision making with their health provider. Dr. Gulley, who heads the center’s genitourinary malignancies branch, declined to comment about access-to-care concerns with Driver’s model. He emphasized that patients who participate in NIH research studies are treated without charge.
“The key to finding better [cancer] treatment is to perform science-driven clinical trials,” Dr. Gulley said in an interview. “However, there are many barriers for enrollment in clinical trials. … As a government agency, NCI is open to partnering with any organization that seeks to improve access to clinical trials for cancer patients.”
NCI and Driver recently conducted a study to validate Driver’s platform; it showed that Driver’s technology successfully predicted the eligibility of patients in NCI Center for Cancer Research clinical trials. The study, presented at a recent American Society of Clinical Oncology meeting, evaluated Driver’s processing of 21 metastatic prostate cancer patients enrolled in a therapeutic NCI clinical trial within the last five years. Results showed Driver correctly predicted that 20 of the patients were “potentially eligible” for the trial in which they were enrolled, and that one was ineligible. Based on the study, a protocol is now in development for a new clinical study, which will seek to further determine the efficiency and accuracy of the clinical trial access program created by Driver, according to Dr. Gulley.
Charles Ryan, MD, director of the division of hematology, oncology, and transplantation for the University of Minnesota, Minneapolis, views Driver’s platform as a way to eliminate geographical barriers, which often keep patients from care, while at the same time enabling researchers to find the right patients for clinical trials.
“We need breakthrough technologies and opportunities for patients to be able to access the most successful and promising cancer treatments, regardless of where they live,” Dr. Ryan said in an interview. “Companies like Driver are attempting to bridge that gap by connecting patients to doctors at world class cancer institutes and direct them toward the best care for their particular condition.”
Driver’s model also allows researchers the opportunity to develop specific, unique treatment for less common cancers and remain optimistic that they can attract patients to receive such treatments as they are developed, Dr. Ryan said.
Dr. Stadler, however, worries that Driver may be giving patients the wrong perception that all it takes is a computer and medical records to determine their best treatment route.
“There’s a lot more subtlety to treatment decisions than most people would like to admit,” Dr. Stadler said. “It’s more than just a bunch of data from sophisticated laboratory tests and the written medical record. Obtaining objective information is the first step, but it’s far from the only step.”
Patients may have significant limitations in functional status that is apparent only during an in-person assessment, for example, he said. In other cases, family members may be essential in conveying information about a patient’s cognitive disabilities. Even when such information is documented, it is sometimes difficult to extract the full picture from the record alone, he said. Dr. Stadler is also bothered that the model requires physicians and hospitals to provide their skilled analyses to a for-profit company, which in turn, charges patients to review the information.
“This is our work,” he said. “I agree that patients should have the information, and I don’t mind sharing anything I have with patients, but now I’m going to share it with another business that essentially is competing with me in terms of providing guidance to patients.”
FDA approves DNA-based blood type test
The for use in transfusion.
It’s the second molecular test for blood compatibility but the first to report genotype in its results, according to an announcement from the agency.
The test is important because it evaluates patients – especially those who receive repeated blood transfusions for conditions such as sickle cell anemia – for non-ABO antigens, but it does so without using antisera, which is sometimes unavailable.
A study found comparable performance between the ID CORE XT, licensed serologic reagents, and DNA sequencing tests, according to the FDA.
The ID CORE XT test is marketed by Progenika Biopharma, a Grifols company.
More information can be found in the full FDA press announcement.
The for use in transfusion.
It’s the second molecular test for blood compatibility but the first to report genotype in its results, according to an announcement from the agency.
The test is important because it evaluates patients – especially those who receive repeated blood transfusions for conditions such as sickle cell anemia – for non-ABO antigens, but it does so without using antisera, which is sometimes unavailable.
A study found comparable performance between the ID CORE XT, licensed serologic reagents, and DNA sequencing tests, according to the FDA.
The ID CORE XT test is marketed by Progenika Biopharma, a Grifols company.
More information can be found in the full FDA press announcement.
The for use in transfusion.
It’s the second molecular test for blood compatibility but the first to report genotype in its results, according to an announcement from the agency.
The test is important because it evaluates patients – especially those who receive repeated blood transfusions for conditions such as sickle cell anemia – for non-ABO antigens, but it does so without using antisera, which is sometimes unavailable.
A study found comparable performance between the ID CORE XT, licensed serologic reagents, and DNA sequencing tests, according to the FDA.
The ID CORE XT test is marketed by Progenika Biopharma, a Grifols company.
More information can be found in the full FDA press announcement.
Acute Superior Mesenteric Venous Thrombosis in a Young Patient Without Risk Factors
In this case report, the authors address the diagnostic challenges of a young, healthy patient who presented to the ED with unrelenting abdominal pain.
Acute mesenteric ischemia (AMI) results when oxygen delivery to the mesenteric artery is compromised, and is a serious diagnosis that should be considered in patients of all ages to avoid significant morbidity and mortality. The majority of cases are due to arterial embolism, arterial thrombus, or intestinal hypoperfusion (non-occlusive). Acute mesenteric venous thrombosis (MVT) accounts for only 2% to 10% of AMI cases, and only 0.01% of emergency surgery admissions.1 A large systematic review showed a 44% mortality rate for MVT, in contrast to 66% to 89% for all other forms of AMI.2 The typical age range for MVT is reported between 45 and 60 years, with a slight male predominance.3 Dull, central abdominal pain is the most frequently reported symptom of MVT, although it is generally less impressive than the pain described in other forms of AMI.3Along with the hallmark of abdominal pain out of proportion to the examination, other gastrointestinal symptoms include weight loss and non-specific altered bowel function (constipation, diarrhea, abdominal distention, and bloating), which are present in half of all patients with MVT.1 Peritoneal signs and bloody stools portend poor outcomes, as they often occur with disease progression.4
Case
A 26-year-old man presented to the ED with periumbilical and lower abdominal pain for 1 week. The pain was described as constant and dull, worsened by movement and oral intake, and improved with lying flat. He described bloating and decreased volume of bowel movements. He denied nausea, vomiting, fever, colicky pain, blood in stool, testicular pain, urinary complaints, trauma, or any similar episodes in the past. The patient had no known medical conditions or surgical history, except for a remote history of alcohol dependence (in remission) and tobacco use. There was no personal or family history of coagulopathy. Of note, he was seen by his primary care physician a few days prior to his ED presentation and had been instructed to take acetaminophen, which did not provide relief.
The patient’s vital signs at presentation were: blood pressure, 122/70 mm Hg; heart rate, 93 beats/min; respiratory rate, 18 breaths/min; and temperature, 37.5°C (99.5°F). Oxygen saturation was 99% on room air. The physical examination was remarkable only for mild abdominal tenderness diffusely, greater in the lower and central abdomen than in the upper abdomen. The remainder of the physical examination was unremarkable.
Laboratory studies ordered included a complete blood count, comprehensive metabolic profile, lipase, and urinalysis. The patient did have a mild transaminitis (aspartate aminotransferase, 48 U/L; alanine aminotransferase, 84 U/L); the remainder of the studies were normal. A serum lactate, drawn after the 1 L of normal saline was administered intravenously (IV), was within normal limits (0.7 mmol/L). No prior laboratory studies were available for comparison.
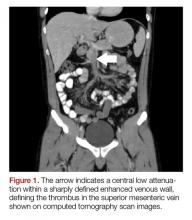
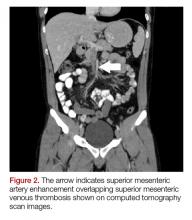
The patient’s continued abdominal pain and transaminitis prompted an ED bedside right upper quadrant ultrasound, which showed a small gallbladder polyp; no signs of gallbladder disease were present. The patient required three doses of morphine 4 mg IV without complete pain relief. Given the concern for pain out of proportion to physical examination, a computed tomography (CT) scan of the abdomen/pelvis with IV and oral contrast was ordered. The radiologist interpreted the scan as showing a superior mesenteric vein (SMV) thrombus extending into the splenic/portal vein confluence and the intrahepatic portal veins (Figures 1 and 2).
Ciprofloxacin and metronidazole were administered IV for antibiotic prophylaxis, and the patient was placed on bowel rest with advancement to regular diet as tolerated. Propranolol was given for variceal prophylaxis. The patient was discharged home the following day in stable condition. Although he still had mild abdominal tenderness, the vital signs and physical examination were within normal limits. The patient was placed on a 6-month course of rivaroxaban therapy. Coagulopathy testing was scheduled at a later date, since ongoing anticoagulation treatment could interfere with test results. Unfortunately, the patient did not attend follow-up appointments to obtain testing.
Discussion
Mesenteric venous thrombosis is seen predominantly in middle-aged patients presenting with vague symptoms, which makes this a challenging diagnosis to make in the acute care setting. Risk factors for MVT include recent injury (causing trauma to the vasculature), recent surgery (causing stagnant blood flow), inflammatory conditions, and hypercoagulable states.1 In this patient’s case, no risk factors were identified; although the majority of cases of MVT will have an identifiable risk factor.2 Still, 21% to 49% of cases of MVT are considered idiopathic.1,3It is possible that our patient had a prior undiagnosed pancreatitis associated with his history of alcoholism that contributed to his thrombosis. Pancreatitis and other inflammatory conditions, including diverticulitis or inflammatory bowel disease, are more commonly associated with thrombus formation in the large veins, as opposed to an undiagnosed hypercoagulable state, which would more likely affect distal venuoles, vasa recta, or venous arcades.1,5 The patient’s mild transaminitis was likely secondary to hepatic congestion from the venous thrombus extending to the splenic-portal vein confluence and intrahepatic portal vein. One study looked at patients with pancreatitis and found that 16.7% of their study population had an SMV thrombus, while 4.1% had a SMV thrombus with a concomitant portal vein thrombus.6
Although there are no pathognomonic laboratory findings of MVT, elevated lactate, leukocytosis, and elevated D-dimer levels may be helpful in supporting the diagnosis.7,8 A recent study found that elevated D-dimer levels may be a specific marker in the early recognition of acute SMV thrombosis, as well as predicting risk, outcomes, and treatment options.8 However, emergency physicians should maintain a high index of suspicion in patients with concerning features of the disease, since normal laboratory values, including lactate, do not reliably exclude the diagnosis.
Computed tomography scanning and CT angiography (CTA) are quite helpful in diagnosing MVT. Ultrasound of the upper abdomen may also play a role, noting dilated or thickened bowel wall with intraluminal air or echogenic material in the superior mesenteric vein or portal vein.9 Although magnetic resonance venography most reliably demonstrates thrombi, its lack of widespread availability makes CT with IV contrast the preferred initial study.3Computed tomography not only has high sensitivity, but also offers alternative diagnoses in the undifferentiated presentation.1One study found CT to be 100% sensitive in detecting any abnormality associated with MVT or bowel ischemia.10 Common CT findings of MVT include dilated and thickened bowel loops, mesenteric fat standing, ascites, a halo or target appearance of bowel, vessel filling defects from a thrombus, and pneumatosis intestinalis.11 The latter usually indicates transmural infarction, and can extend as portomesenteric vein gas.11 Of note, if the initial CT scan is non-diagnostic and a high clinical suspicion for mesenteric ischemia remains with no alternative diagnosis, CTA is the gold standard.3,7Expeditious diagnosis of MVT is imperative, given the potential complications of intestinal infarction, submucosal hemorrhage secondary to edema, and third spacing of the venous outflow into the bowel wall due to collateral vessels being unable to redirect blood flow in conjunction with complete venous occlusion.12Not all MVTs progress to infarction, given the extensive collateral circulation. Early diagnosis, however, is crucial for conservative management to be effective.9Acute MVT without signs of infarction necessitates anticoagulation therapy to decrease clot propagation and recurrence.1 In addition, prophylactic antibiotics to limit bacterial translocation, and bowel rest are advised.13,14 If the patient is unresponsive to anticoagulation, thrombolytic and endovascular therapies may be of benefit in select patients.15 Once intestinal ischemia or infarction develops, the prognosis is poor: mortality approaches 75% with infarction.1 If signs of bowel infarction are present, a laparotomy must be performed promptly, although in most cases, delayed patient presentation makes small bowel resection unavoidable.9 Further testing for hypercoagulability is recommended, particularly in isolated thrombosis, since long-term anticoagulation therapy may be necessary if a coagulopathy is discovered.1
Conclusion
Mesenteric venous thrombosis is atypical in a young, healthy patient. However, due to high mortality rates with disease progression, it is important to consider in any patient with unrelenting abdominal pain and vague gastrointestinal symptoms of uncertain cause, even in those without risk factors. Early detection and management of MVT before progression to mesenteric ischemia and infarction considerably lowers the mortality rate. Emergency physicians must be vigilant when treating a patient with abdominal pain out of proportion to physical examination, unrelenting pain despite analgesic medications, or repeat ED visits for the same abdominal complaints.
1. Harnik IG, Brandt LJ. Mesenteric venous thrombosis. Vasc Med. 2010;15(5):407-418. doi:10.1177/1358863x10379673.
2. Tilsed JV, Casamassima A, Kurihara H, et al. ESTES guidelines: acute mesenteric ischaemia. Eur J Trauma Emerg Surg. 2016;42(2):253-270. doi:10.1007/s00068-016-0634-0.
3. Tendler DA, Lamont JT, Grubel P. Mesenteric venous thrombosis in adults. UpToDate Web site. https://www.uptodate.com/contents/mesenteric-venous-thrombosis-in-adults. Accessed November 16, 2017.
4. Al-Zahrani HA, Lindsay T. Mesenteric ischemia. In: Hall JB, Schmidt GA, Kress JP, eds. Principles of Critical Care. 4th ed. New York, NY: McGraw Hill; 2015:1036-1044.
5. Kumar S, Sarr MG, Kamath PS. Mesenteric venous thrombosis. N Engl J Med. 2001;345(23):1683-1688. doi:10.1056/nejmra010076.
6. Al-Khazraji A, Hasan AQ, Patel I, Alkhawam H, Ghrair F, Lieber J. The role of abdominal computed tomography scan in acute pancreatitis. Pancreas. 2017;46(6):e52-e54. doi:10.1097/mpa.0000000000000837.
7. Bradbury MS, Kavanagh PV, Bechtold RE, et al. Mesenteric venous thrombosis: diagnosis and noninvasive imaging. Radiographics. 2002;22(3):527-541.
8. Yang S, Fan X, Ding W, et al. D-dimer as an early marker of severity in patients with acute superior mesenteric venous thrombosis. Medicine (Baltimore). 2014;93(29):e270. doi:10.1097/md.0000000000000270.
9. Matos C, Van Gansbeke D, Zalcman M, et al. Mesenteric vein thrombosis: early CT and US diagnosis and conservative management. Gastrointest Radiol. 1986;11(4):322-325.
10. Rhee RY, Gloviczki P, Mendonca CT, et al. Mesenteric venous thrombosis: still a lethal disease in the 1990s. J Vasc Surg. 1994;20(5):688-697.
11. Furukawa A, Kanasaki S, Kono N, et al. CT diagnosis of acute mesenteric ischemia from various causes. AJR Am J Roentgenol. 2009;192(2):408-416. doi:10.2214/ajr.08.1138.
12. Johnson CC, Baggenstoss AH. Mesenteric vascular occlusion; study of 99 cases of occlusion of veins. Proc Staff Meet Mayo Clin. 1949;24(25):628-636.13. Hmoud B, Singal AK, Kamath PS. Mesenteric venous thrombosis. J Clin Exp Hepatol. 2014;4(3):257-263. doi:10.1016/j.jceh.2014.03.052.
14. Schoots IG, Koffeman GI, Legemate DA, Levi M, van Gulik TM. Systematic review of survival after acute mesenteric ischaemia according to disease aetiology. Br J Surg. 2004;91(1):17-27.
15. Yang S, Fan X, Ding W, et al. Multidisciplinary stepwise management strategy for acute superior mesenteric venous thrombosis: an intestinal stroke center experience. Thromb Res. 2015;135(1):36-45. doi:10.1016/j.thromres.2014.10.018.
In this case report, the authors address the diagnostic challenges of a young, healthy patient who presented to the ED with unrelenting abdominal pain.
In this case report, the authors address the diagnostic challenges of a young, healthy patient who presented to the ED with unrelenting abdominal pain.
Acute mesenteric ischemia (AMI) results when oxygen delivery to the mesenteric artery is compromised, and is a serious diagnosis that should be considered in patients of all ages to avoid significant morbidity and mortality. The majority of cases are due to arterial embolism, arterial thrombus, or intestinal hypoperfusion (non-occlusive). Acute mesenteric venous thrombosis (MVT) accounts for only 2% to 10% of AMI cases, and only 0.01% of emergency surgery admissions.1 A large systematic review showed a 44% mortality rate for MVT, in contrast to 66% to 89% for all other forms of AMI.2 The typical age range for MVT is reported between 45 and 60 years, with a slight male predominance.3 Dull, central abdominal pain is the most frequently reported symptom of MVT, although it is generally less impressive than the pain described in other forms of AMI.3Along with the hallmark of abdominal pain out of proportion to the examination, other gastrointestinal symptoms include weight loss and non-specific altered bowel function (constipation, diarrhea, abdominal distention, and bloating), which are present in half of all patients with MVT.1 Peritoneal signs and bloody stools portend poor outcomes, as they often occur with disease progression.4
Case
A 26-year-old man presented to the ED with periumbilical and lower abdominal pain for 1 week. The pain was described as constant and dull, worsened by movement and oral intake, and improved with lying flat. He described bloating and decreased volume of bowel movements. He denied nausea, vomiting, fever, colicky pain, blood in stool, testicular pain, urinary complaints, trauma, or any similar episodes in the past. The patient had no known medical conditions or surgical history, except for a remote history of alcohol dependence (in remission) and tobacco use. There was no personal or family history of coagulopathy. Of note, he was seen by his primary care physician a few days prior to his ED presentation and had been instructed to take acetaminophen, which did not provide relief.
The patient’s vital signs at presentation were: blood pressure, 122/70 mm Hg; heart rate, 93 beats/min; respiratory rate, 18 breaths/min; and temperature, 37.5°C (99.5°F). Oxygen saturation was 99% on room air. The physical examination was remarkable only for mild abdominal tenderness diffusely, greater in the lower and central abdomen than in the upper abdomen. The remainder of the physical examination was unremarkable.
Laboratory studies ordered included a complete blood count, comprehensive metabolic profile, lipase, and urinalysis. The patient did have a mild transaminitis (aspartate aminotransferase, 48 U/L; alanine aminotransferase, 84 U/L); the remainder of the studies were normal. A serum lactate, drawn after the 1 L of normal saline was administered intravenously (IV), was within normal limits (0.7 mmol/L). No prior laboratory studies were available for comparison.
The patient’s continued abdominal pain and transaminitis prompted an ED bedside right upper quadrant ultrasound, which showed a small gallbladder polyp; no signs of gallbladder disease were present. The patient required three doses of morphine 4 mg IV without complete pain relief. Given the concern for pain out of proportion to physical examination, a computed tomography (CT) scan of the abdomen/pelvis with IV and oral contrast was ordered. The radiologist interpreted the scan as showing a superior mesenteric vein (SMV) thrombus extending into the splenic/portal vein confluence and the intrahepatic portal veins (Figures 1 and 2).
Ciprofloxacin and metronidazole were administered IV for antibiotic prophylaxis, and the patient was placed on bowel rest with advancement to regular diet as tolerated. Propranolol was given for variceal prophylaxis. The patient was discharged home the following day in stable condition. Although he still had mild abdominal tenderness, the vital signs and physical examination were within normal limits. The patient was placed on a 6-month course of rivaroxaban therapy. Coagulopathy testing was scheduled at a later date, since ongoing anticoagulation treatment could interfere with test results. Unfortunately, the patient did not attend follow-up appointments to obtain testing.
Discussion
Mesenteric venous thrombosis is seen predominantly in middle-aged patients presenting with vague symptoms, which makes this a challenging diagnosis to make in the acute care setting. Risk factors for MVT include recent injury (causing trauma to the vasculature), recent surgery (causing stagnant blood flow), inflammatory conditions, and hypercoagulable states.1 In this patient’s case, no risk factors were identified; although the majority of cases of MVT will have an identifiable risk factor.2 Still, 21% to 49% of cases of MVT are considered idiopathic.1,3It is possible that our patient had a prior undiagnosed pancreatitis associated with his history of alcoholism that contributed to his thrombosis. Pancreatitis and other inflammatory conditions, including diverticulitis or inflammatory bowel disease, are more commonly associated with thrombus formation in the large veins, as opposed to an undiagnosed hypercoagulable state, which would more likely affect distal venuoles, vasa recta, or venous arcades.1,5 The patient’s mild transaminitis was likely secondary to hepatic congestion from the venous thrombus extending to the splenic-portal vein confluence and intrahepatic portal vein. One study looked at patients with pancreatitis and found that 16.7% of their study population had an SMV thrombus, while 4.1% had a SMV thrombus with a concomitant portal vein thrombus.6
Although there are no pathognomonic laboratory findings of MVT, elevated lactate, leukocytosis, and elevated D-dimer levels may be helpful in supporting the diagnosis.7,8 A recent study found that elevated D-dimer levels may be a specific marker in the early recognition of acute SMV thrombosis, as well as predicting risk, outcomes, and treatment options.8 However, emergency physicians should maintain a high index of suspicion in patients with concerning features of the disease, since normal laboratory values, including lactate, do not reliably exclude the diagnosis.
Computed tomography scanning and CT angiography (CTA) are quite helpful in diagnosing MVT. Ultrasound of the upper abdomen may also play a role, noting dilated or thickened bowel wall with intraluminal air or echogenic material in the superior mesenteric vein or portal vein.9 Although magnetic resonance venography most reliably demonstrates thrombi, its lack of widespread availability makes CT with IV contrast the preferred initial study.3Computed tomography not only has high sensitivity, but also offers alternative diagnoses in the undifferentiated presentation.1One study found CT to be 100% sensitive in detecting any abnormality associated with MVT or bowel ischemia.10 Common CT findings of MVT include dilated and thickened bowel loops, mesenteric fat standing, ascites, a halo or target appearance of bowel, vessel filling defects from a thrombus, and pneumatosis intestinalis.11 The latter usually indicates transmural infarction, and can extend as portomesenteric vein gas.11 Of note, if the initial CT scan is non-diagnostic and a high clinical suspicion for mesenteric ischemia remains with no alternative diagnosis, CTA is the gold standard.3,7Expeditious diagnosis of MVT is imperative, given the potential complications of intestinal infarction, submucosal hemorrhage secondary to edema, and third spacing of the venous outflow into the bowel wall due to collateral vessels being unable to redirect blood flow in conjunction with complete venous occlusion.12Not all MVTs progress to infarction, given the extensive collateral circulation. Early diagnosis, however, is crucial for conservative management to be effective.9Acute MVT without signs of infarction necessitates anticoagulation therapy to decrease clot propagation and recurrence.1 In addition, prophylactic antibiotics to limit bacterial translocation, and bowel rest are advised.13,14 If the patient is unresponsive to anticoagulation, thrombolytic and endovascular therapies may be of benefit in select patients.15 Once intestinal ischemia or infarction develops, the prognosis is poor: mortality approaches 75% with infarction.1 If signs of bowel infarction are present, a laparotomy must be performed promptly, although in most cases, delayed patient presentation makes small bowel resection unavoidable.9 Further testing for hypercoagulability is recommended, particularly in isolated thrombosis, since long-term anticoagulation therapy may be necessary if a coagulopathy is discovered.1
Conclusion
Mesenteric venous thrombosis is atypical in a young, healthy patient. However, due to high mortality rates with disease progression, it is important to consider in any patient with unrelenting abdominal pain and vague gastrointestinal symptoms of uncertain cause, even in those without risk factors. Early detection and management of MVT before progression to mesenteric ischemia and infarction considerably lowers the mortality rate. Emergency physicians must be vigilant when treating a patient with abdominal pain out of proportion to physical examination, unrelenting pain despite analgesic medications, or repeat ED visits for the same abdominal complaints.
Acute mesenteric ischemia (AMI) results when oxygen delivery to the mesenteric artery is compromised, and is a serious diagnosis that should be considered in patients of all ages to avoid significant morbidity and mortality. The majority of cases are due to arterial embolism, arterial thrombus, or intestinal hypoperfusion (non-occlusive). Acute mesenteric venous thrombosis (MVT) accounts for only 2% to 10% of AMI cases, and only 0.01% of emergency surgery admissions.1 A large systematic review showed a 44% mortality rate for MVT, in contrast to 66% to 89% for all other forms of AMI.2 The typical age range for MVT is reported between 45 and 60 years, with a slight male predominance.3 Dull, central abdominal pain is the most frequently reported symptom of MVT, although it is generally less impressive than the pain described in other forms of AMI.3Along with the hallmark of abdominal pain out of proportion to the examination, other gastrointestinal symptoms include weight loss and non-specific altered bowel function (constipation, diarrhea, abdominal distention, and bloating), which are present in half of all patients with MVT.1 Peritoneal signs and bloody stools portend poor outcomes, as they often occur with disease progression.4
Case
A 26-year-old man presented to the ED with periumbilical and lower abdominal pain for 1 week. The pain was described as constant and dull, worsened by movement and oral intake, and improved with lying flat. He described bloating and decreased volume of bowel movements. He denied nausea, vomiting, fever, colicky pain, blood in stool, testicular pain, urinary complaints, trauma, or any similar episodes in the past. The patient had no known medical conditions or surgical history, except for a remote history of alcohol dependence (in remission) and tobacco use. There was no personal or family history of coagulopathy. Of note, he was seen by his primary care physician a few days prior to his ED presentation and had been instructed to take acetaminophen, which did not provide relief.
The patient’s vital signs at presentation were: blood pressure, 122/70 mm Hg; heart rate, 93 beats/min; respiratory rate, 18 breaths/min; and temperature, 37.5°C (99.5°F). Oxygen saturation was 99% on room air. The physical examination was remarkable only for mild abdominal tenderness diffusely, greater in the lower and central abdomen than in the upper abdomen. The remainder of the physical examination was unremarkable.
Laboratory studies ordered included a complete blood count, comprehensive metabolic profile, lipase, and urinalysis. The patient did have a mild transaminitis (aspartate aminotransferase, 48 U/L; alanine aminotransferase, 84 U/L); the remainder of the studies were normal. A serum lactate, drawn after the 1 L of normal saline was administered intravenously (IV), was within normal limits (0.7 mmol/L). No prior laboratory studies were available for comparison.
The patient’s continued abdominal pain and transaminitis prompted an ED bedside right upper quadrant ultrasound, which showed a small gallbladder polyp; no signs of gallbladder disease were present. The patient required three doses of morphine 4 mg IV without complete pain relief. Given the concern for pain out of proportion to physical examination, a computed tomography (CT) scan of the abdomen/pelvis with IV and oral contrast was ordered. The radiologist interpreted the scan as showing a superior mesenteric vein (SMV) thrombus extending into the splenic/portal vein confluence and the intrahepatic portal veins (Figures 1 and 2).
Ciprofloxacin and metronidazole were administered IV for antibiotic prophylaxis, and the patient was placed on bowel rest with advancement to regular diet as tolerated. Propranolol was given for variceal prophylaxis. The patient was discharged home the following day in stable condition. Although he still had mild abdominal tenderness, the vital signs and physical examination were within normal limits. The patient was placed on a 6-month course of rivaroxaban therapy. Coagulopathy testing was scheduled at a later date, since ongoing anticoagulation treatment could interfere with test results. Unfortunately, the patient did not attend follow-up appointments to obtain testing.
Discussion
Mesenteric venous thrombosis is seen predominantly in middle-aged patients presenting with vague symptoms, which makes this a challenging diagnosis to make in the acute care setting. Risk factors for MVT include recent injury (causing trauma to the vasculature), recent surgery (causing stagnant blood flow), inflammatory conditions, and hypercoagulable states.1 In this patient’s case, no risk factors were identified; although the majority of cases of MVT will have an identifiable risk factor.2 Still, 21% to 49% of cases of MVT are considered idiopathic.1,3It is possible that our patient had a prior undiagnosed pancreatitis associated with his history of alcoholism that contributed to his thrombosis. Pancreatitis and other inflammatory conditions, including diverticulitis or inflammatory bowel disease, are more commonly associated with thrombus formation in the large veins, as opposed to an undiagnosed hypercoagulable state, which would more likely affect distal venuoles, vasa recta, or venous arcades.1,5 The patient’s mild transaminitis was likely secondary to hepatic congestion from the venous thrombus extending to the splenic-portal vein confluence and intrahepatic portal vein. One study looked at patients with pancreatitis and found that 16.7% of their study population had an SMV thrombus, while 4.1% had a SMV thrombus with a concomitant portal vein thrombus.6
Although there are no pathognomonic laboratory findings of MVT, elevated lactate, leukocytosis, and elevated D-dimer levels may be helpful in supporting the diagnosis.7,8 A recent study found that elevated D-dimer levels may be a specific marker in the early recognition of acute SMV thrombosis, as well as predicting risk, outcomes, and treatment options.8 However, emergency physicians should maintain a high index of suspicion in patients with concerning features of the disease, since normal laboratory values, including lactate, do not reliably exclude the diagnosis.
Computed tomography scanning and CT angiography (CTA) are quite helpful in diagnosing MVT. Ultrasound of the upper abdomen may also play a role, noting dilated or thickened bowel wall with intraluminal air or echogenic material in the superior mesenteric vein or portal vein.9 Although magnetic resonance venography most reliably demonstrates thrombi, its lack of widespread availability makes CT with IV contrast the preferred initial study.3Computed tomography not only has high sensitivity, but also offers alternative diagnoses in the undifferentiated presentation.1One study found CT to be 100% sensitive in detecting any abnormality associated with MVT or bowel ischemia.10 Common CT findings of MVT include dilated and thickened bowel loops, mesenteric fat standing, ascites, a halo or target appearance of bowel, vessel filling defects from a thrombus, and pneumatosis intestinalis.11 The latter usually indicates transmural infarction, and can extend as portomesenteric vein gas.11 Of note, if the initial CT scan is non-diagnostic and a high clinical suspicion for mesenteric ischemia remains with no alternative diagnosis, CTA is the gold standard.3,7Expeditious diagnosis of MVT is imperative, given the potential complications of intestinal infarction, submucosal hemorrhage secondary to edema, and third spacing of the venous outflow into the bowel wall due to collateral vessels being unable to redirect blood flow in conjunction with complete venous occlusion.12Not all MVTs progress to infarction, given the extensive collateral circulation. Early diagnosis, however, is crucial for conservative management to be effective.9Acute MVT without signs of infarction necessitates anticoagulation therapy to decrease clot propagation and recurrence.1 In addition, prophylactic antibiotics to limit bacterial translocation, and bowel rest are advised.13,14 If the patient is unresponsive to anticoagulation, thrombolytic and endovascular therapies may be of benefit in select patients.15 Once intestinal ischemia or infarction develops, the prognosis is poor: mortality approaches 75% with infarction.1 If signs of bowel infarction are present, a laparotomy must be performed promptly, although in most cases, delayed patient presentation makes small bowel resection unavoidable.9 Further testing for hypercoagulability is recommended, particularly in isolated thrombosis, since long-term anticoagulation therapy may be necessary if a coagulopathy is discovered.1
Conclusion
Mesenteric venous thrombosis is atypical in a young, healthy patient. However, due to high mortality rates with disease progression, it is important to consider in any patient with unrelenting abdominal pain and vague gastrointestinal symptoms of uncertain cause, even in those without risk factors. Early detection and management of MVT before progression to mesenteric ischemia and infarction considerably lowers the mortality rate. Emergency physicians must be vigilant when treating a patient with abdominal pain out of proportion to physical examination, unrelenting pain despite analgesic medications, or repeat ED visits for the same abdominal complaints.
1. Harnik IG, Brandt LJ. Mesenteric venous thrombosis. Vasc Med. 2010;15(5):407-418. doi:10.1177/1358863x10379673.
2. Tilsed JV, Casamassima A, Kurihara H, et al. ESTES guidelines: acute mesenteric ischaemia. Eur J Trauma Emerg Surg. 2016;42(2):253-270. doi:10.1007/s00068-016-0634-0.
3. Tendler DA, Lamont JT, Grubel P. Mesenteric venous thrombosis in adults. UpToDate Web site. https://www.uptodate.com/contents/mesenteric-venous-thrombosis-in-adults. Accessed November 16, 2017.
4. Al-Zahrani HA, Lindsay T. Mesenteric ischemia. In: Hall JB, Schmidt GA, Kress JP, eds. Principles of Critical Care. 4th ed. New York, NY: McGraw Hill; 2015:1036-1044.
5. Kumar S, Sarr MG, Kamath PS. Mesenteric venous thrombosis. N Engl J Med. 2001;345(23):1683-1688. doi:10.1056/nejmra010076.
6. Al-Khazraji A, Hasan AQ, Patel I, Alkhawam H, Ghrair F, Lieber J. The role of abdominal computed tomography scan in acute pancreatitis. Pancreas. 2017;46(6):e52-e54. doi:10.1097/mpa.0000000000000837.
7. Bradbury MS, Kavanagh PV, Bechtold RE, et al. Mesenteric venous thrombosis: diagnosis and noninvasive imaging. Radiographics. 2002;22(3):527-541.
8. Yang S, Fan X, Ding W, et al. D-dimer as an early marker of severity in patients with acute superior mesenteric venous thrombosis. Medicine (Baltimore). 2014;93(29):e270. doi:10.1097/md.0000000000000270.
9. Matos C, Van Gansbeke D, Zalcman M, et al. Mesenteric vein thrombosis: early CT and US diagnosis and conservative management. Gastrointest Radiol. 1986;11(4):322-325.
10. Rhee RY, Gloviczki P, Mendonca CT, et al. Mesenteric venous thrombosis: still a lethal disease in the 1990s. J Vasc Surg. 1994;20(5):688-697.
11. Furukawa A, Kanasaki S, Kono N, et al. CT diagnosis of acute mesenteric ischemia from various causes. AJR Am J Roentgenol. 2009;192(2):408-416. doi:10.2214/ajr.08.1138.
12. Johnson CC, Baggenstoss AH. Mesenteric vascular occlusion; study of 99 cases of occlusion of veins. Proc Staff Meet Mayo Clin. 1949;24(25):628-636.13. Hmoud B, Singal AK, Kamath PS. Mesenteric venous thrombosis. J Clin Exp Hepatol. 2014;4(3):257-263. doi:10.1016/j.jceh.2014.03.052.
14. Schoots IG, Koffeman GI, Legemate DA, Levi M, van Gulik TM. Systematic review of survival after acute mesenteric ischaemia according to disease aetiology. Br J Surg. 2004;91(1):17-27.
15. Yang S, Fan X, Ding W, et al. Multidisciplinary stepwise management strategy for acute superior mesenteric venous thrombosis: an intestinal stroke center experience. Thromb Res. 2015;135(1):36-45. doi:10.1016/j.thromres.2014.10.018.
1. Harnik IG, Brandt LJ. Mesenteric venous thrombosis. Vasc Med. 2010;15(5):407-418. doi:10.1177/1358863x10379673.
2. Tilsed JV, Casamassima A, Kurihara H, et al. ESTES guidelines: acute mesenteric ischaemia. Eur J Trauma Emerg Surg. 2016;42(2):253-270. doi:10.1007/s00068-016-0634-0.
3. Tendler DA, Lamont JT, Grubel P. Mesenteric venous thrombosis in adults. UpToDate Web site. https://www.uptodate.com/contents/mesenteric-venous-thrombosis-in-adults. Accessed November 16, 2017.
4. Al-Zahrani HA, Lindsay T. Mesenteric ischemia. In: Hall JB, Schmidt GA, Kress JP, eds. Principles of Critical Care. 4th ed. New York, NY: McGraw Hill; 2015:1036-1044.
5. Kumar S, Sarr MG, Kamath PS. Mesenteric venous thrombosis. N Engl J Med. 2001;345(23):1683-1688. doi:10.1056/nejmra010076.
6. Al-Khazraji A, Hasan AQ, Patel I, Alkhawam H, Ghrair F, Lieber J. The role of abdominal computed tomography scan in acute pancreatitis. Pancreas. 2017;46(6):e52-e54. doi:10.1097/mpa.0000000000000837.
7. Bradbury MS, Kavanagh PV, Bechtold RE, et al. Mesenteric venous thrombosis: diagnosis and noninvasive imaging. Radiographics. 2002;22(3):527-541.
8. Yang S, Fan X, Ding W, et al. D-dimer as an early marker of severity in patients with acute superior mesenteric venous thrombosis. Medicine (Baltimore). 2014;93(29):e270. doi:10.1097/md.0000000000000270.
9. Matos C, Van Gansbeke D, Zalcman M, et al. Mesenteric vein thrombosis: early CT and US diagnosis and conservative management. Gastrointest Radiol. 1986;11(4):322-325.
10. Rhee RY, Gloviczki P, Mendonca CT, et al. Mesenteric venous thrombosis: still a lethal disease in the 1990s. J Vasc Surg. 1994;20(5):688-697.
11. Furukawa A, Kanasaki S, Kono N, et al. CT diagnosis of acute mesenteric ischemia from various causes. AJR Am J Roentgenol. 2009;192(2):408-416. doi:10.2214/ajr.08.1138.
12. Johnson CC, Baggenstoss AH. Mesenteric vascular occlusion; study of 99 cases of occlusion of veins. Proc Staff Meet Mayo Clin. 1949;24(25):628-636.13. Hmoud B, Singal AK, Kamath PS. Mesenteric venous thrombosis. J Clin Exp Hepatol. 2014;4(3):257-263. doi:10.1016/j.jceh.2014.03.052.
14. Schoots IG, Koffeman GI, Legemate DA, Levi M, van Gulik TM. Systematic review of survival after acute mesenteric ischaemia according to disease aetiology. Br J Surg. 2004;91(1):17-27.
15. Yang S, Fan X, Ding W, et al. Multidisciplinary stepwise management strategy for acute superior mesenteric venous thrombosis: an intestinal stroke center experience. Thromb Res. 2015;135(1):36-45. doi:10.1016/j.thromres.2014.10.018.
Crizanlizumab relieves sickle cell crises across subgroups
Crizanlizumab effectively reduced vaso-occlusive crises among patients with sickle cell disease (SCD) who have numerous crises, exhibit the HbSS genotype, and take concomitant hydroxyurea, according to investigators.
Across subgroups, crizanlizumab was safe and more effective than placebo at delaying time to first vaso-occlusive crisis (VOC) and eliminating crises, reported lead author Abdullah Kutlar, MD, of the Sickle Cell Center at the Medical College of Georgia, Augusta, and his colleagues.
The phase 2 SUSTAIN trial recently showed that crizanlizumab – a humanized, anti–P-selectin monoclonal antibody – reduced the frequency of VOCs by 45% and delayed time to first crisis by about 3 months (N Engl J Med. 2017;376:429-39).
Additionally, a subgroup analysis showed that there was a lower frequency of pain crises with crizanlizumab 5 mg/kg, compared with placebo, regardless of the number of prior VOCs, concomitant hydroxyurea use, or the SCD genotype.
The present post hoc analysis took a deeper look at these observations across the same subgroups; specifically, the investigators assessed elimination of VOCs, time to first crisis, and adverse events. They reported the findings in the American Journal of Hematology.
Crizanlizumab eliminated pain crises about seven times more frequently than did placebo in patients who had a high frequency of VOCs before the study (28.0% vs. 4.2%), and about twice as often in patients with the HbSS genotype (31.9% vs. 17.0%), and patients who were using concomitant hydroxyurea (33.3% vs. 17.5%).
Further analysis showed that crizanlizumab delayed time to first pain crisis across all subgroups, most dramatically in patients with the HbSS genotype (4.07 months for crizanlizumab vs. 1.12 months for placebo). Safety was comparable across subgroups.
“These findings provide supportive evidence that crizanlizumab provides a clinically meaningful treatment benefit when used alone or in combination with hydroxyurea for the prevention of VOCs,” the investigators wrote.
An ongoing phase 2 pharmacokinetic/pharmacodynamic study is evaluating a higher dose of crizanlizumab (7.5 mg/kg), and another trial seeks to evaluate pediatric doses of the drug.
The study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
SOURCE: Kutlar A et al. Am J Hematol. 2018 Oct 8. doi: 10.1002/ajh.25308.
Crizanlizumab effectively reduced vaso-occlusive crises among patients with sickle cell disease (SCD) who have numerous crises, exhibit the HbSS genotype, and take concomitant hydroxyurea, according to investigators.
Across subgroups, crizanlizumab was safe and more effective than placebo at delaying time to first vaso-occlusive crisis (VOC) and eliminating crises, reported lead author Abdullah Kutlar, MD, of the Sickle Cell Center at the Medical College of Georgia, Augusta, and his colleagues.
The phase 2 SUSTAIN trial recently showed that crizanlizumab – a humanized, anti–P-selectin monoclonal antibody – reduced the frequency of VOCs by 45% and delayed time to first crisis by about 3 months (N Engl J Med. 2017;376:429-39).
Additionally, a subgroup analysis showed that there was a lower frequency of pain crises with crizanlizumab 5 mg/kg, compared with placebo, regardless of the number of prior VOCs, concomitant hydroxyurea use, or the SCD genotype.
The present post hoc analysis took a deeper look at these observations across the same subgroups; specifically, the investigators assessed elimination of VOCs, time to first crisis, and adverse events. They reported the findings in the American Journal of Hematology.
Crizanlizumab eliminated pain crises about seven times more frequently than did placebo in patients who had a high frequency of VOCs before the study (28.0% vs. 4.2%), and about twice as often in patients with the HbSS genotype (31.9% vs. 17.0%), and patients who were using concomitant hydroxyurea (33.3% vs. 17.5%).
Further analysis showed that crizanlizumab delayed time to first pain crisis across all subgroups, most dramatically in patients with the HbSS genotype (4.07 months for crizanlizumab vs. 1.12 months for placebo). Safety was comparable across subgroups.
“These findings provide supportive evidence that crizanlizumab provides a clinically meaningful treatment benefit when used alone or in combination with hydroxyurea for the prevention of VOCs,” the investigators wrote.
An ongoing phase 2 pharmacokinetic/pharmacodynamic study is evaluating a higher dose of crizanlizumab (7.5 mg/kg), and another trial seeks to evaluate pediatric doses of the drug.
The study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
SOURCE: Kutlar A et al. Am J Hematol. 2018 Oct 8. doi: 10.1002/ajh.25308.
Crizanlizumab effectively reduced vaso-occlusive crises among patients with sickle cell disease (SCD) who have numerous crises, exhibit the HbSS genotype, and take concomitant hydroxyurea, according to investigators.
Across subgroups, crizanlizumab was safe and more effective than placebo at delaying time to first vaso-occlusive crisis (VOC) and eliminating crises, reported lead author Abdullah Kutlar, MD, of the Sickle Cell Center at the Medical College of Georgia, Augusta, and his colleagues.
The phase 2 SUSTAIN trial recently showed that crizanlizumab – a humanized, anti–P-selectin monoclonal antibody – reduced the frequency of VOCs by 45% and delayed time to first crisis by about 3 months (N Engl J Med. 2017;376:429-39).
Additionally, a subgroup analysis showed that there was a lower frequency of pain crises with crizanlizumab 5 mg/kg, compared with placebo, regardless of the number of prior VOCs, concomitant hydroxyurea use, or the SCD genotype.
The present post hoc analysis took a deeper look at these observations across the same subgroups; specifically, the investigators assessed elimination of VOCs, time to first crisis, and adverse events. They reported the findings in the American Journal of Hematology.
Crizanlizumab eliminated pain crises about seven times more frequently than did placebo in patients who had a high frequency of VOCs before the study (28.0% vs. 4.2%), and about twice as often in patients with the HbSS genotype (31.9% vs. 17.0%), and patients who were using concomitant hydroxyurea (33.3% vs. 17.5%).
Further analysis showed that crizanlizumab delayed time to first pain crisis across all subgroups, most dramatically in patients with the HbSS genotype (4.07 months for crizanlizumab vs. 1.12 months for placebo). Safety was comparable across subgroups.
“These findings provide supportive evidence that crizanlizumab provides a clinically meaningful treatment benefit when used alone or in combination with hydroxyurea for the prevention of VOCs,” the investigators wrote.
An ongoing phase 2 pharmacokinetic/pharmacodynamic study is evaluating a higher dose of crizanlizumab (7.5 mg/kg), and another trial seeks to evaluate pediatric doses of the drug.
The study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
SOURCE: Kutlar A et al. Am J Hematol. 2018 Oct 8. doi: 10.1002/ajh.25308.
FROM THE AMERICAN JOURNAL OF HEMATOLOGY
Key clinical point:
Major finding: Crizanlizumab eliminated vaso-occlusive crises about seven times more frequently than did placebo in patients with numerous crises (28.0% vs. 4.2%).
Study details: A post hoc analysis of 132 patients from the phase 2 SUSTAIN trial.
Disclosures: The study was sponsored by Novartis. The authors reported financial relationships with Novartis, Bluebird Bio, AstraZeneca, and others.
Source: Kutlar A et al. Am J Hematol. 2018 Oct 8. doi: 10.1002/ajh.25308.
Five “can’t miss” oncologic emergencies
SAN DIEGO – Acute promyelocytic leukemia is one of five “can’t miss” oncologic emergencies, Megan Boysen Osborn, MD, MHPE, told a standing-room-only crowd at the annual meeting of the American College of Emergency Physicians.
In our exclusive video interview, Dr. Osborn, vice chair of education and the residency program director in the department of emergency medicine at the University of California, Irvine, offered tips on how to recognize acute promyelocytic leukemia, leukostasis, neutropenic fever, tumor lysis syndrome, and disseminated intravascular coagulation.
“All patients with suspected leukemias should be admitted,” she said. “Time is of the essence.”
Dr. Osborn reported having no financial disclosures related to her presentation.
SAN DIEGO – Acute promyelocytic leukemia is one of five “can’t miss” oncologic emergencies, Megan Boysen Osborn, MD, MHPE, told a standing-room-only crowd at the annual meeting of the American College of Emergency Physicians.
In our exclusive video interview, Dr. Osborn, vice chair of education and the residency program director in the department of emergency medicine at the University of California, Irvine, offered tips on how to recognize acute promyelocytic leukemia, leukostasis, neutropenic fever, tumor lysis syndrome, and disseminated intravascular coagulation.
“All patients with suspected leukemias should be admitted,” she said. “Time is of the essence.”
Dr. Osborn reported having no financial disclosures related to her presentation.
SAN DIEGO – Acute promyelocytic leukemia is one of five “can’t miss” oncologic emergencies, Megan Boysen Osborn, MD, MHPE, told a standing-room-only crowd at the annual meeting of the American College of Emergency Physicians.
In our exclusive video interview, Dr. Osborn, vice chair of education and the residency program director in the department of emergency medicine at the University of California, Irvine, offered tips on how to recognize acute promyelocytic leukemia, leukostasis, neutropenic fever, tumor lysis syndrome, and disseminated intravascular coagulation.
“All patients with suspected leukemias should be admitted,” she said. “Time is of the essence.”
Dr. Osborn reported having no financial disclosures related to her presentation.
REPORTING FROM ACEP18
What’s the best VTE treatment for patients with cancer?
EVIDENCE SUMMARY
No head-to-head studies or umbrella meta-analyses assess all the main treatments for VTE against each other.
Long-term LMWH decreases VTE recurrence compared with VKA
Two meta-analyses of RCTs evaluating LMWH and VKA for long-term treatment (3-12 months) of confirmed VTE in patients with cancer found that LMWH didn’t change mortality, but reduced the rate of VTE recurrence compared with VKA (40% relative reduction).1,2 The comparison showed no differences in major or minor bleeding or thrombocytopenia between LMWH and VKA (TABLE1-5).
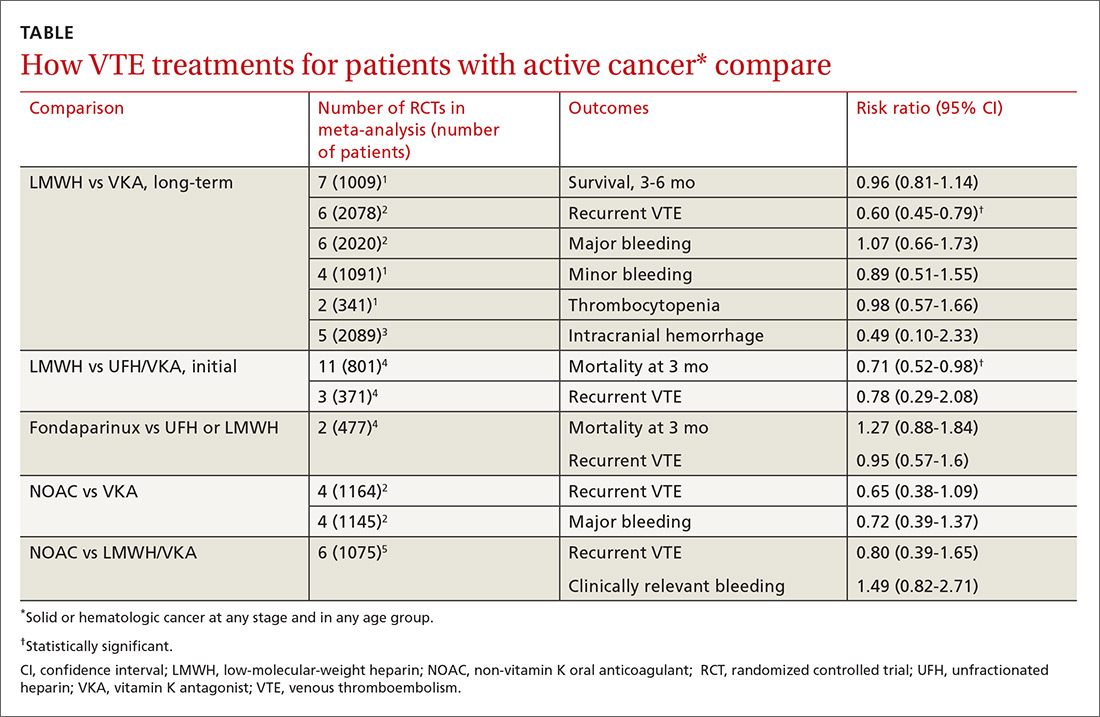
The studies included patients with any solid or hematologic cancer at any stage and from any age group, including children. Overall, the mean age of patients was in the mid 60s; approximately 50% were male when specified. Investigators rated the evidence quality as moderate for VTE, but low for the other outcomes.1
The most recent meta-analysis of the same RCTs comparing LMWH with VKA evaluated intracranial hemorrhage rates and found no difference.3
Initial therapy with LMWH: A look at mortality
A meta-analysis of RCTs that compared LMWH with UFH/VKA for initial treatment of confirmed VTE in adult cancer patients (any type or stage of cancer, mean ages not specified) found that LMWH reduced mortality by 30%, but didn’t affect VTE recurrence or major bleeding.4
The control groups received UFH for 5 to 10 days and then continued with VKA, whereas the experimental groups received different types of LMWH (reviparin, nadroparin, tinzaparin, enoxaparin) initially and for 3 months thereafter. Investigators rated all studies low quality because of imprecision and publication bias favoring LMWH.
Fondaparinux shows no advantage for initial therapy
The same meta-analysis compared initial treatment with fondaparinux and initial therapy with enoxaparin or UFH transitioning to warfarin.4 It found no differences in any outcomes at 3 months. Investigators rated both studies as low quality for recurrent VTE and moderate for mortality and bleeding.
Continue to: Non-vitamin K oral anticoagulants vs LMWH/VKA or VKA
Non-vitamin K oral anticoagulants vs LMWH/VKA or VKA: No differences
A meta-analysis of RCTs comparing NOACs (dabigatran, edoxaban, apixaban, rivaroxaban) with VKA for 6 months found no differences in recurrent VTE or major bleeding.2
A second meta-analysis of RCTs that compared NOACs (rivaroxaban, dabigatran, apixaban) with control (LMWH followed by VKA) in adult cancer patients (mean ages, 54-66 years; 50%-60% men) reported no difference in the composite outcome of recurrent VTE or VTE-related death nor clinically significant bleeding over 1 to 36 months (most RCTs ran 3-12 months).5 Separate comparisons for rivaroxaban and dabigatran found no difference in the composite outcome, and rivaroxaban also produced no difference in clinically-significant bleeding.
RECOMMENDATIONS
The 2016 CHEST guidelines recommend LMWH as first-line treatment for VTE in patients with cancer and indicate no preference between NOACs and VKA for second-line treatment.6
1. Akl EA, Kahale L, Barba M, et al. Anticoagulation for the long-term treatment of venous thromboembolism in patients with cancer. Cochrane Database Syst Rev. 2014;(7):CD006650.
2. Posch F, Königsbrügge O, Zielinski C, et al. Treatment of venous thromboembolism in patients with cancer: A network meta-analysis comparing efficacy and safety of anticoagulants. Thromb Res. 2015;136:582-589.
3. Rojas-Hernandez CM, Oo TH, García-Perdomo HA. Risk of intracranial hemorrhage associated with therapeutic anticoagulation for venous thromboembolism in cancer patients: a systematic review and meta-analysis. J Thromb Thrombolysis. 2017;43:233-240.
4. Akl EA, Kahale L, Neumann I, et al. Anticoagulation for the initial treatment of venous thromboembolism in patients with cancer. Cochrane Database Syst Rev. 2014;(6):CD006649.
5. Sardar P, Chatterjee S, Herzog E, et al. New oral anticoagulants in patients with cancer: current state of evidence. Am J Ther. 2015;22:460-468.
6. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149:315-352.
EVIDENCE SUMMARY
No head-to-head studies or umbrella meta-analyses assess all the main treatments for VTE against each other.
Long-term LMWH decreases VTE recurrence compared with VKA
Two meta-analyses of RCTs evaluating LMWH and VKA for long-term treatment (3-12 months) of confirmed VTE in patients with cancer found that LMWH didn’t change mortality, but reduced the rate of VTE recurrence compared with VKA (40% relative reduction).1,2 The comparison showed no differences in major or minor bleeding or thrombocytopenia between LMWH and VKA (TABLE1-5).

The studies included patients with any solid or hematologic cancer at any stage and from any age group, including children. Overall, the mean age of patients was in the mid 60s; approximately 50% were male when specified. Investigators rated the evidence quality as moderate for VTE, but low for the other outcomes.1
The most recent meta-analysis of the same RCTs comparing LMWH with VKA evaluated intracranial hemorrhage rates and found no difference.3
Initial therapy with LMWH: A look at mortality
A meta-analysis of RCTs that compared LMWH with UFH/VKA for initial treatment of confirmed VTE in adult cancer patients (any type or stage of cancer, mean ages not specified) found that LMWH reduced mortality by 30%, but didn’t affect VTE recurrence or major bleeding.4
The control groups received UFH for 5 to 10 days and then continued with VKA, whereas the experimental groups received different types of LMWH (reviparin, nadroparin, tinzaparin, enoxaparin) initially and for 3 months thereafter. Investigators rated all studies low quality because of imprecision and publication bias favoring LMWH.
Fondaparinux shows no advantage for initial therapy
The same meta-analysis compared initial treatment with fondaparinux and initial therapy with enoxaparin or UFH transitioning to warfarin.4 It found no differences in any outcomes at 3 months. Investigators rated both studies as low quality for recurrent VTE and moderate for mortality and bleeding.
Continue to: Non-vitamin K oral anticoagulants vs LMWH/VKA or VKA
Non-vitamin K oral anticoagulants vs LMWH/VKA or VKA: No differences
A meta-analysis of RCTs comparing NOACs (dabigatran, edoxaban, apixaban, rivaroxaban) with VKA for 6 months found no differences in recurrent VTE or major bleeding.2
A second meta-analysis of RCTs that compared NOACs (rivaroxaban, dabigatran, apixaban) with control (LMWH followed by VKA) in adult cancer patients (mean ages, 54-66 years; 50%-60% men) reported no difference in the composite outcome of recurrent VTE or VTE-related death nor clinically significant bleeding over 1 to 36 months (most RCTs ran 3-12 months).5 Separate comparisons for rivaroxaban and dabigatran found no difference in the composite outcome, and rivaroxaban also produced no difference in clinically-significant bleeding.
RECOMMENDATIONS
The 2016 CHEST guidelines recommend LMWH as first-line treatment for VTE in patients with cancer and indicate no preference between NOACs and VKA for second-line treatment.6
EVIDENCE SUMMARY
No head-to-head studies or umbrella meta-analyses assess all the main treatments for VTE against each other.
Long-term LMWH decreases VTE recurrence compared with VKA
Two meta-analyses of RCTs evaluating LMWH and VKA for long-term treatment (3-12 months) of confirmed VTE in patients with cancer found that LMWH didn’t change mortality, but reduced the rate of VTE recurrence compared with VKA (40% relative reduction).1,2 The comparison showed no differences in major or minor bleeding or thrombocytopenia between LMWH and VKA (TABLE1-5).

The studies included patients with any solid or hematologic cancer at any stage and from any age group, including children. Overall, the mean age of patients was in the mid 60s; approximately 50% were male when specified. Investigators rated the evidence quality as moderate for VTE, but low for the other outcomes.1
The most recent meta-analysis of the same RCTs comparing LMWH with VKA evaluated intracranial hemorrhage rates and found no difference.3
Initial therapy with LMWH: A look at mortality
A meta-analysis of RCTs that compared LMWH with UFH/VKA for initial treatment of confirmed VTE in adult cancer patients (any type or stage of cancer, mean ages not specified) found that LMWH reduced mortality by 30%, but didn’t affect VTE recurrence or major bleeding.4
The control groups received UFH for 5 to 10 days and then continued with VKA, whereas the experimental groups received different types of LMWH (reviparin, nadroparin, tinzaparin, enoxaparin) initially and for 3 months thereafter. Investigators rated all studies low quality because of imprecision and publication bias favoring LMWH.
Fondaparinux shows no advantage for initial therapy
The same meta-analysis compared initial treatment with fondaparinux and initial therapy with enoxaparin or UFH transitioning to warfarin.4 It found no differences in any outcomes at 3 months. Investigators rated both studies as low quality for recurrent VTE and moderate for mortality and bleeding.
Continue to: Non-vitamin K oral anticoagulants vs LMWH/VKA or VKA
Non-vitamin K oral anticoagulants vs LMWH/VKA or VKA: No differences
A meta-analysis of RCTs comparing NOACs (dabigatran, edoxaban, apixaban, rivaroxaban) with VKA for 6 months found no differences in recurrent VTE or major bleeding.2
A second meta-analysis of RCTs that compared NOACs (rivaroxaban, dabigatran, apixaban) with control (LMWH followed by VKA) in adult cancer patients (mean ages, 54-66 years; 50%-60% men) reported no difference in the composite outcome of recurrent VTE or VTE-related death nor clinically significant bleeding over 1 to 36 months (most RCTs ran 3-12 months).5 Separate comparisons for rivaroxaban and dabigatran found no difference in the composite outcome, and rivaroxaban also produced no difference in clinically-significant bleeding.
RECOMMENDATIONS
The 2016 CHEST guidelines recommend LMWH as first-line treatment for VTE in patients with cancer and indicate no preference between NOACs and VKA for second-line treatment.6
1. Akl EA, Kahale L, Barba M, et al. Anticoagulation for the long-term treatment of venous thromboembolism in patients with cancer. Cochrane Database Syst Rev. 2014;(7):CD006650.
2. Posch F, Königsbrügge O, Zielinski C, et al. Treatment of venous thromboembolism in patients with cancer: A network meta-analysis comparing efficacy and safety of anticoagulants. Thromb Res. 2015;136:582-589.
3. Rojas-Hernandez CM, Oo TH, García-Perdomo HA. Risk of intracranial hemorrhage associated with therapeutic anticoagulation for venous thromboembolism in cancer patients: a systematic review and meta-analysis. J Thromb Thrombolysis. 2017;43:233-240.
4. Akl EA, Kahale L, Neumann I, et al. Anticoagulation for the initial treatment of venous thromboembolism in patients with cancer. Cochrane Database Syst Rev. 2014;(6):CD006649.
5. Sardar P, Chatterjee S, Herzog E, et al. New oral anticoagulants in patients with cancer: current state of evidence. Am J Ther. 2015;22:460-468.
6. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149:315-352.
1. Akl EA, Kahale L, Barba M, et al. Anticoagulation for the long-term treatment of venous thromboembolism in patients with cancer. Cochrane Database Syst Rev. 2014;(7):CD006650.
2. Posch F, Königsbrügge O, Zielinski C, et al. Treatment of venous thromboembolism in patients with cancer: A network meta-analysis comparing efficacy and safety of anticoagulants. Thromb Res. 2015;136:582-589.
3. Rojas-Hernandez CM, Oo TH, García-Perdomo HA. Risk of intracranial hemorrhage associated with therapeutic anticoagulation for venous thromboembolism in cancer patients: a systematic review and meta-analysis. J Thromb Thrombolysis. 2017;43:233-240.
4. Akl EA, Kahale L, Neumann I, et al. Anticoagulation for the initial treatment of venous thromboembolism in patients with cancer. Cochrane Database Syst Rev. 2014;(6):CD006649.
5. Sardar P, Chatterjee S, Herzog E, et al. New oral anticoagulants in patients with cancer: current state of evidence. Am J Ther. 2015;22:460-468.
6. Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149:315-352.
EVIDENCE-BASED ANSWER:
No head-to-head studies directly compare all the main treatments for venous thromboembolism (VTE) in cancer patients. Long-term treatment (3-12 months) with low-molecular-weight heparin (LMWH) reduces recurrence of VTE by 40% compared with vitamin K antagonists (VKA), but doesn’t change rates of mortality, major or minor bleeding, or intracranial hemorrhage in patients with solid or hematologic cancer at any stage or in any age group. Initial treatment with LMWH reduces mortality by 30% compared with unfractionated heparin (UFH) for 5 to 10 days followed by warfarin, but doesn’t alter recurrent VTE or bleeding. Non-vitamin K oral anticoagulants (NOACs) have risks of recurrent VTE or VTE-related death (composite outcome) and clinically significant bleeding comparable to VKA or LMWH/VKA (strength of recommendation [SOR]: B, meta-analyses of randomized controlled trials [RCTs], mostly of low quality).
Strategies for caring for the well cancer survivor
Cancer survivors represent a rapidly increasing population. In 1971, there were 3 million cancer survivors; this number increased to 15.5 million in 2016 and will reach 20 million by 2026.1TABLE 11 shows the percentage of survivors by type of cancer. Cancer survivors tend to be older,* comprising nearly 1 of every 5 people older than 65 years.2
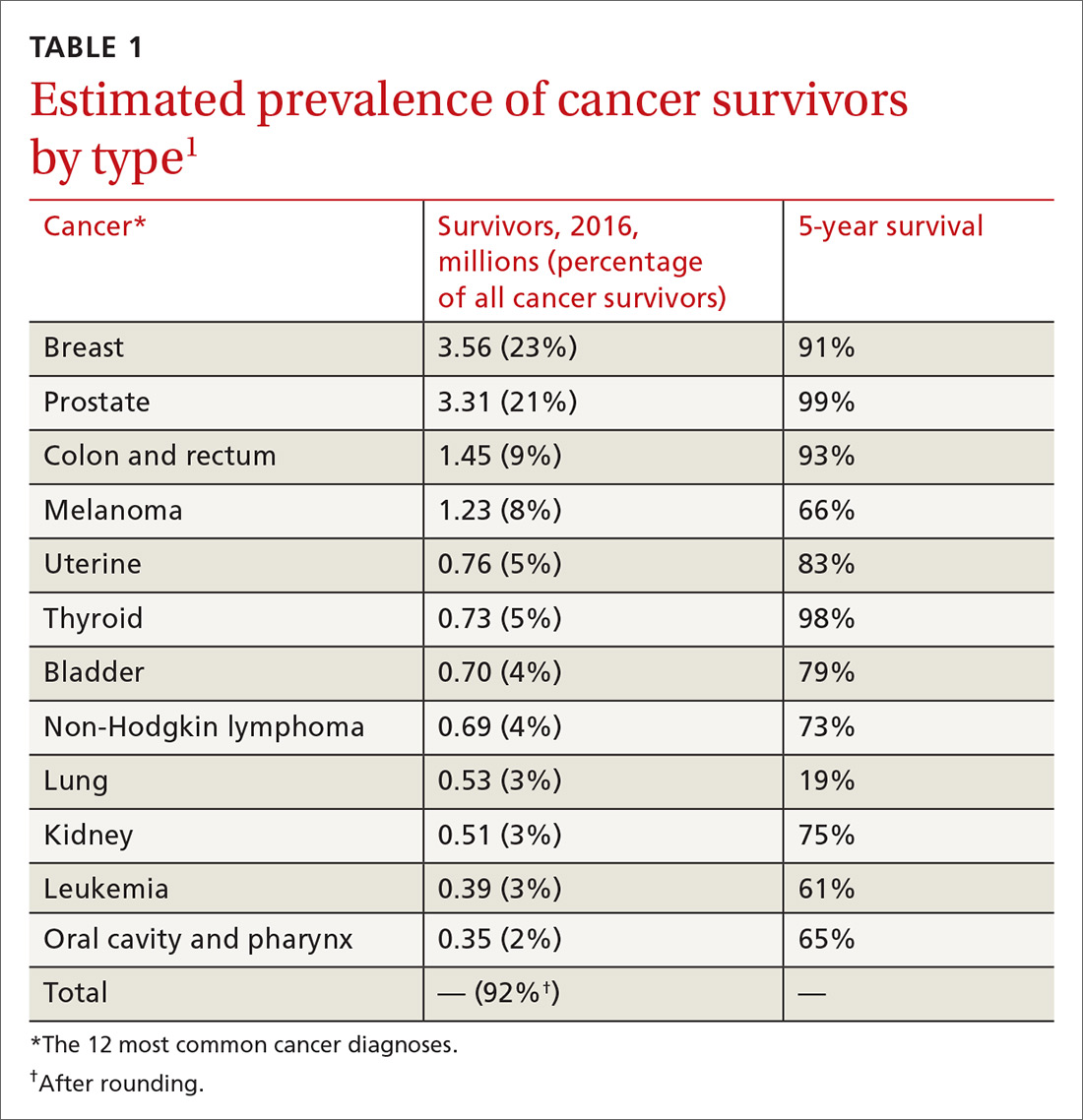
The Institute of Medicine (IOM) identified 3 key characteristics of cancer survivors3:
- Trajectories of survivorship are variable; many cancer patients have periods of relative health between episodes of their disease.
- Survivors require careful cancer monitoring; in addition to the risk that their primary cancer will recur, they have an elevated risk for another, second cancer.
- Both cancer and its treatments increase the risk of other medical and psychiatric problems.
Family physicians (FPs) have optimal skills for navigating the chronic risks and health concerns of the well cancer survivor. This article reviews the primary care management of the functional cancer survivor, focusing on the management of chronic conditions and preventive care.
Survivorship follows any of 6 paths
Cancer survivorship is increasing in importance as treatment has steadily reduced mortality. Six trajectories of cancer survivors have been identified1:
- living cancer-free after treatment with minimal effects
- living cancer-free but suffering serious treatment complications
- Suffering late recurrence
- Developing a second cancer
- Living with intermittent cancer recurrences
- Living with cancer continuously.
Only patients in the last 2 groups are likely to be managed primarily by oncologists.
Survivors look to their FPs for ongoing care
Cancer survivors routinely see their primary care physician after initial treatment. A study of 30,000 Canadian breast cancer survivors demonstrated that follow-up care was limited to an oncologist in only 2%; 84% saw a primary care provider and an oncologist; and 14% saw a primary care provider only.4 A study of colorectal cancer survivors showed that primary care visits increased in each of the 5 years after diagnosis, during which time oncology visits decreased steadily5; in that study, primary care physicians delivered more preventive care than oncologists did.5 Similar to what is done in other chronic conditions, the various effects of cancer are best managed as a whole.
The IOM recommends that cancer survivor care comprise 4 elements2:
- coordination between oncologist and primary care physician
- surveillance for recurrence or spread of existing cancer
- screening for new cancer
- intervention for the effects of cancer and treatment.
Continue to: The following discussion summarizes...
The following discussion summarizes evidence and recommendations for each element of the IOM recommendations for survivor care.
Implementing the 4 elements of cancer survivor care
1. Coordinate care through a unified survivorship care plan
The IOM has noted that the needs of cancer survivors are rarely met2; communication between oncology and primary care is often deficient during transition of care. The IOM has recommended that oncologists provide a survivorship care plan that details the cancer (ie, tumor characteristics), the type of treatment (ie, enrollment in a clinical trial; medical, surgical, or radiation), support services, and follow-up recommendations for the primary care provider. (Examples of elements of a survivorship care plan can be found at www.mskcc.org/hcp-education-training/survivorship/survivorship-care-plan6 and http://sma.org/southern-medical-journal/article/cancer-survivors-history-physical/7).
Regrettably, survivorship care plans have been rarely and poorly employed. Studies show that fewer than one-half of oncologists provide a plan, and that when they do, the plan often lacks recommended information.8,9 Survivorship care plans may soon become common practice, however; the Commission on Cancer of the American College of Surgeons has required their use in all certified cancer centers since 2015.10
2. Provide surveillance of existing cancer
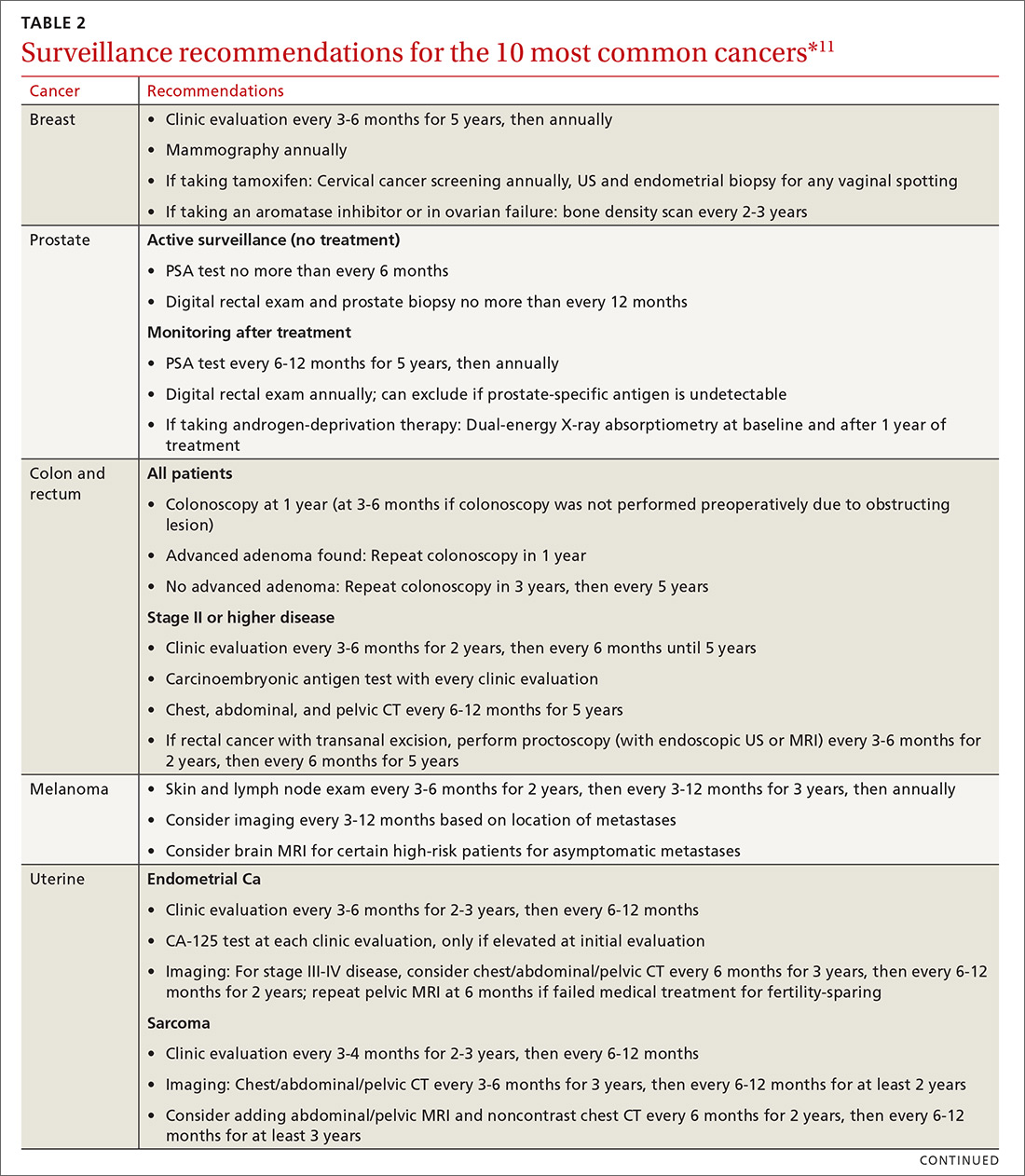
Cancer follow-up is challenging after the initial treatment phase. Although there are many conflicting guidelines for surveillance after cancer, guidelines of the National Comprehensive Cancer Network (NCCN) (summarized in TABLE 211 for the 10 most common cancers in survivors) are the ones generally accepted.12,13
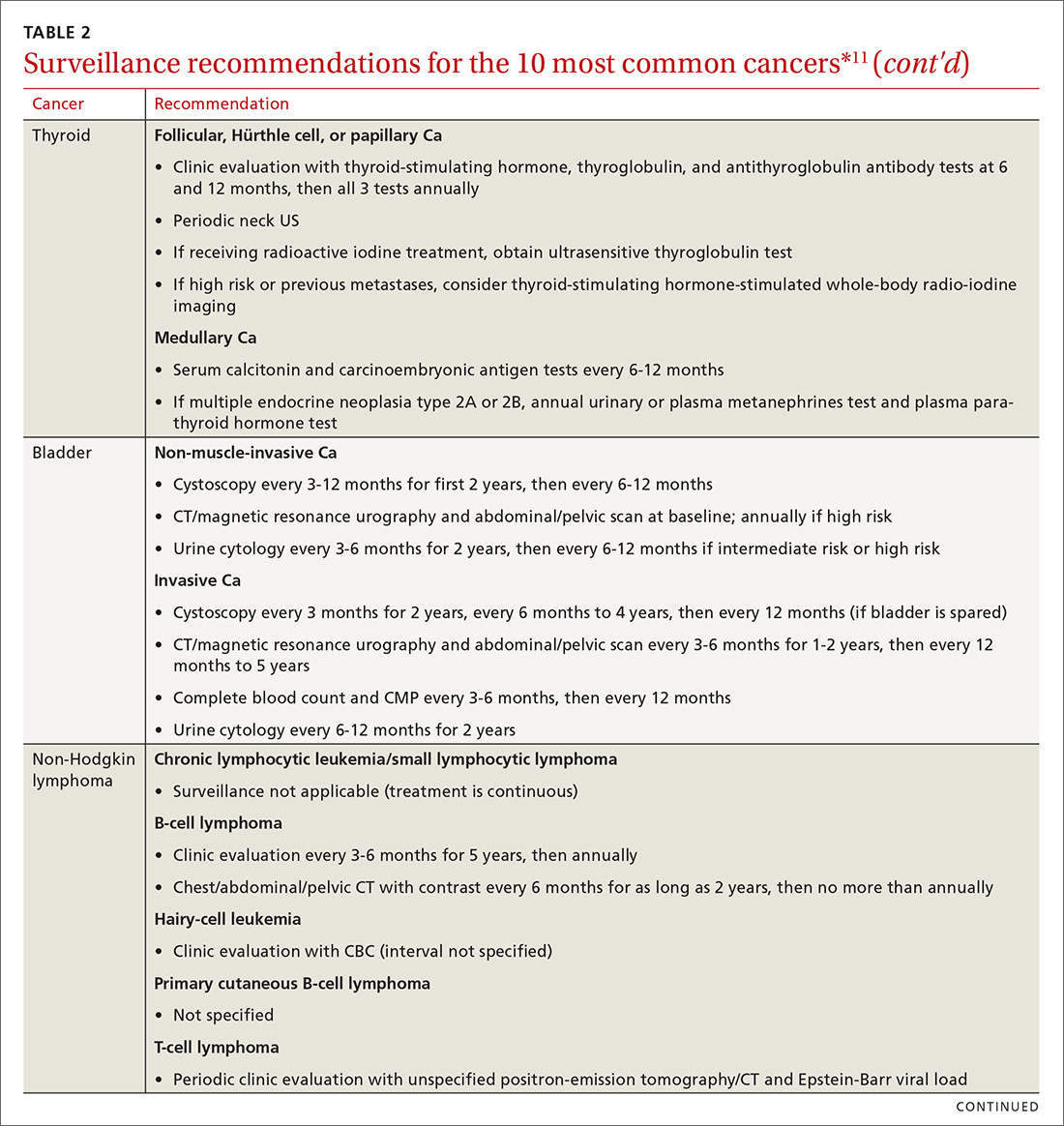
Although individual surveillance recommendations are based on limited evidence, studies confirm the importance of surveillance. A systematic review showed that surveillance mammography after breast cancer reduces breast cancer mortality by 36%.14 A study showed that bladder cancer recurrence diagnosed by surveillance instead of by symptoms led to a 35% increase in 5-year survival.15
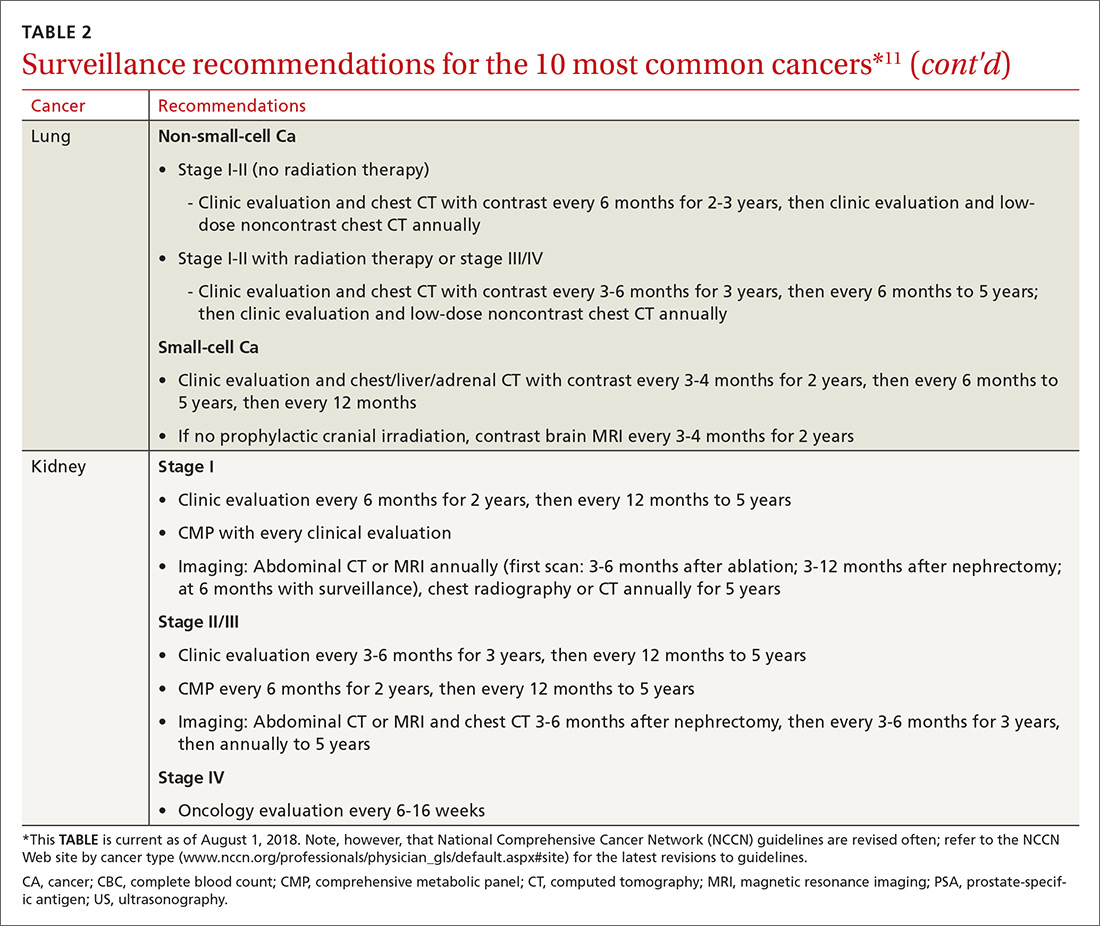
Continue to: Yet adherence to cancer surveillance...
Yet adherence to cancer surveillance recommendations is poor. A study of patients with colon cancer demonstrated that only 12% met all recommended surveillance guidelines.16 A study of patients with bladder cancer after radical cystectomy showed that only 9% met recommended surveillance more than 2 years after diagnosis.17 Those dismal statistics may be the result of provider oversight—not patient reluctance.
In the colon cancer study, for example, compliance with follow-up colonoscopy was 80% but compliance with carcinoembryonic antigen testing was only 22%.16 In the bladder cancer study, follow-up urine cytology was obtained in only 23% of patients, although 75% completed recommended imaging.17
Although surveillance remains the oncologist’s responsibility, visits to the FP provide an opportunity to review surveillance and order needed laboratory testing and other studies, including imaging.
3. Screen for new cancers
The risk of a second cancer is elevated for cancer survivors compared with the risk of a primary cancer in the healthy general population; some survivors have a lifetime risk of a second cancer as high as 36%.18 Risk varies by cancer type (TABLE 319). Some of this variation is due to the impact of smoking: Smoking-related cancers have the highest risk of second malignancy.19 Genetic predisposition to malignant transformation is also theorized to contribute to increased risk. Second malignancies are dangerous; 55% of patients die of the second cancer compared with only 13% of their initial cancer.19
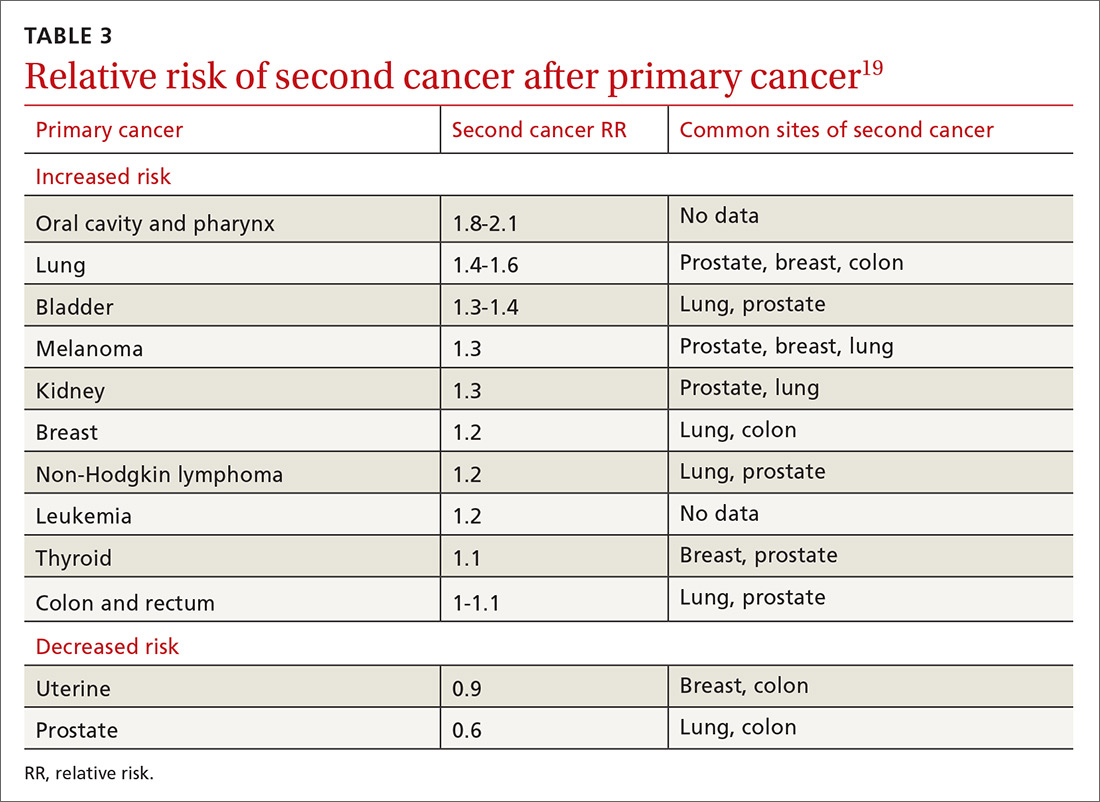
Studies show that cancer survivors display varying adherence with recommended screening for second cancers. In a study of Latina cancer survivors, depressive symptoms were associated with lower screening compliance.20 A study of survivors of hematologic cancer showed a low rate of cancer screening and high fear of cancer recurrence—suggesting avoidance due to fear.21 Other studies, however, show similar or increased compliance with screening in cancer survivors.22,23 A meta-analysis of 19 studies determined that, overall, cancer survivors receive 25% to 38% more recommended screening than the general population.24
Continue to: Few guidelines exist to guide FPs...
Few guidelines exist to guide FPs in adjusting screening for the cancer survivor. For women who received radiation therapy for a tumor in the chest, for example, the recommendation offered by several groups is to start breast cancer screening 8 to 10 years after treatment or by 30 years of age, and to consider combining magnetic resonance imaging and mammography.25 Recommendations for breast cancer screening do not account for a history of other gynecologic cancers unless genetic markers are present.25 On the other hand, the impact of a history of cancer on the risk of prostate cancer and on screening decisions has not been studied,26 and cervical cancer screening guidelines, which recommend that screening continue after 65 years of age for patients who are immunocompromised, do not address a history of other cancer.27
4. Manage the effects of both the cancer and the treatment
Medical issues faced by cancer survivors are familiar to FPs, but there are some specific recommendations regarding evaluation and treatment that stand in contrast to what would be considered for a healthy, or non-cancer, patient. For example, each chemotherapeutic agent has characteristic adverse effects; TABLE 47 lists the principal adverse effects of common agents and recommendations for testing when these problems develop. Common long-term problems in cancer survivors include fatigue, chronic pain, cognitive dysfunction, psychiatric illness, and cardiovascular disease. Although these symptoms and manifestations are common, the physician must be careful: New or changing symptoms could signal the spread or recurrence of disease. Fear of recurrence can lead patients to exaggerate or minimize symptoms.
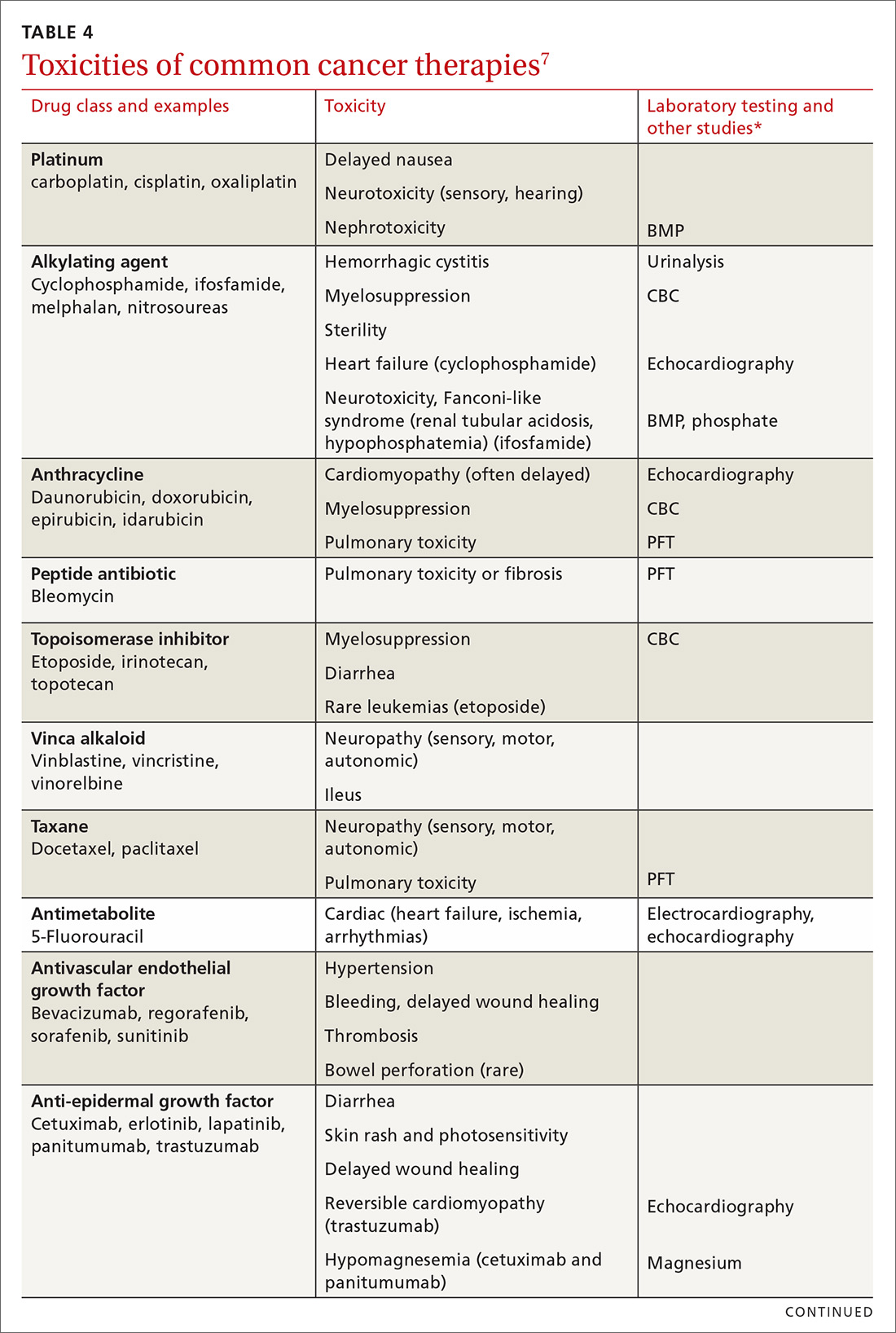
Fatigue is the most common symptom seen in cancer survivors during treatment and following remission.28 More than 40% of cancer survivors report significant fatigue.29 Although fatigue is concerning for cancer recurrence, other causes are common in cancer survivors. Both depression and anxiety commonly present with worsened fatigue.30 Sleep disturbances are common, even without a psychiatric diagnosis.31 Effects of treatment, including nausea, anemia, heart failure, and medication adverse effects can cause or worsen fatigue. Pain is associated with fatigue, but to a lesser extent than are depression, anxiety, and nausea.32
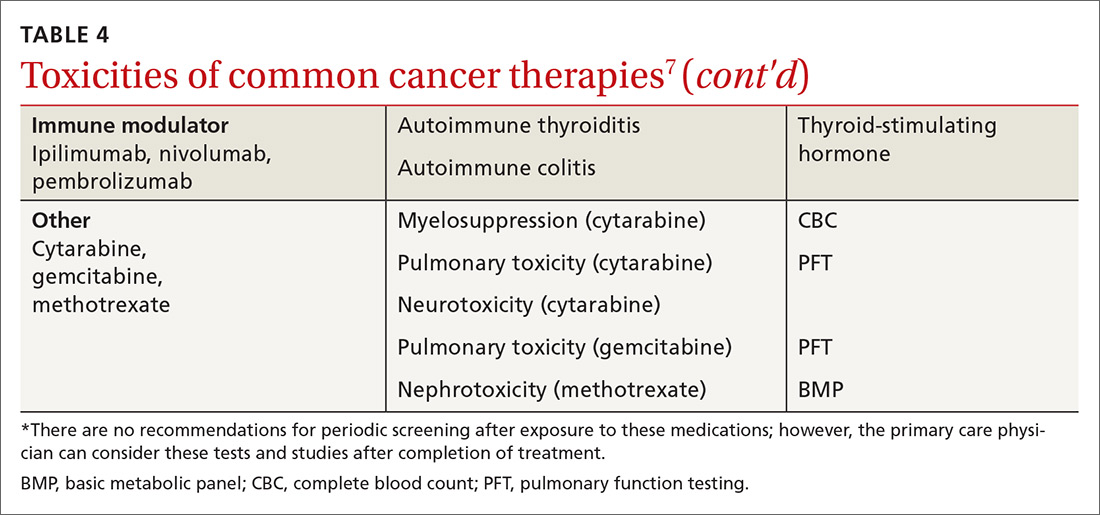
Pharmacotherapy of cancer-related fatigue is challenging. Psychostimulants have been most studied. A recent systematic review shows that methylphenidate produces mild or moderate improvement in fatigue, whereas modafanil has minimal effectiveness.33 Antidepressants have not been shown to relieve fatigue.33
A recent meta-analysis showed that nonpharmaceutical treatments for cancer-related fatigue are more effective than pharmacotherapy. In this review, both exercise and pharmacotherapy had a mild-to-moderate effect on fatigue.35 Exercise is best studied in this regard, and has shown the most consistent results.31
Continue to: Chronic pain
Chronic pain. Pain is common in cancer survivors: As many as 40% experience pain for years after initial therapy.36 Treatment of some cancers—eg, thoracotomy (80%), amputation (50%-80%), neck dissection (52%), and surgical management of breast cancer (63%)—increase the likelihood of chronic pain.37 Reports of pain in cancer survivors that should be considered red flags that might signal recurrence of cancer include new or worsening pain; pain worse at night or when recumbent; new neurologic symptoms; and general symptoms of systemic illness37 (TABLE 537).

Management of pain is best approached by its cause, with neurologic, rheumatologic (including myofascial pain and arthralgia), lymphatic, and genital causes most common.37 Across all types of pain, complete relief is unlikely; functional goals provide a more effective target.
For neuropathic cancer pain, duloxetine is the only medication with evidence of benefit; anticonvulsant and topical medications are recommended on the basis of the findings of studies of noncancer pain.38 There are few data on the value of treatments for cancer-related rheumatologic and lymphatic pain, although exercise has shown benefit in both types.38 For dyspareunia and sexual dysfunction (common after gynecologic and nongynecologic cancers), vaginal lubricants and pelvic-floor physiotherapy have shown benefit.39 There is significant overlap in psychiatric comorbidities, sleep, and pain, and addressing all of a patient’s problems can reduce pain and improve function.40
Opioids are often prescribed for pain in cancer survivors. Cancer survivors have a higher rate of opioid prescribing compared with that of non-cancer patients, even 10 years after diagnosis.41 Guidelines of the Centers for Disease Control and Prevention for using opioids to manage chronic pain specifically exclude cancer patients.42 Regrettably, there is no evidence that opioids have long-term efficacy in chronic pain; in fact, evidence is accumulating that chronic opioid therapy exacerbates chronic pain.43
Cognitive dysfunction is present in 17% to 75% of cancer survivors as memory disturbance, psychological disorder, sleep dysfunction, or impairment of executive functioning.44 Cognitive deficits appear to be secondary to both cancer and treatment modalities45; as many as one-third of patients have cognitive dysfunction prior to receiving chemotherapy.46
Continue to: Chemotherapies that are more likely...
Chemotherapies that are more likely to cause cognitive symptoms include methotrexate, 5-fluorouracil, cyclophosphamide, and hormone antagonists.47 More powerful regimens and repetitive chemotherapy regimens tend to cause more cognitive effects.47
Cognitive training interventions show evidence of likely benefit,44,48 leading to recommendations for self-treatment strategies, such as written lists, wordplay, crossword puzzles, jigsaw puzzles, playing a musical instrument, and new hobbies. Small studies suggest a benefit from cognitive behavioral therapy.44,49 A study of breast cancer survivors showed that yoga led to improvement in patient-reported cognitive dysfunction.50 Physical exercise yields cognitive benefit in healthy older adults and is supported by limited evidence in cancer survivors.51
There is no effective pharmacotherapy for cancer- and cancer chemotherapy-related cognitive dysfunction unless a treatable underlying cause is found.44 Symptoms tend to subside with time after completion of chemotherapy, which might be reassuring to patients and families.45
Psychiatric problems. The most common psychiatric issues in cancer survivors are anxiety and depression; the prevalence of anxiety is nearly double that of depression.52 Anxiety often presents as fear of a recurrence of cancer or a feeling of lack of control over present or future circumstances.53 Screening for anxiety and depression is recommended at each visit, using standardized screening questionnaires.54
A small study suggests that psychiatric treatment reduces the risk of early mortality.55 Small studies also suggest that mindfulness-based therapy and cognitive behavioral therapy delivered by telehealth offer benefit.56 A meta-analysis shows that exercise interventions improve depression and anxiety in breast cancer patients.57
Continue to: There are few studies of pharmacotherapy...
There are few studies of pharmacotherapy of anxiety or depression in cancer survivors56; it is known that cancer survivors are nearly twice as likely as the general population to be taking medical therapy for anxiety and depression.58 A Cochrane systematic review of 7 small studies showed uncertain improvement in depressive symptoms in patients with cancer from antidepressant medication; however, an earlier systematic review did show benefit.59,60
In a trial of patients without depression who were being treated for head and neck cancer, escitalopram, 20 mg/d, reduced the risk of subsequent depression compared with placebo.61 A study of 420 breast cancer survivors showed that 300 mg/d and 900 mg/d dosages of gabapentin were both superior to placebo, and nearly equivalent to each other, at reducing anxiety scores.62 In both studies, however, the evidence is nonetheless insufficient to make specific recommendations about these medications.
Cardiac risk
Among chemotherapeutic agents, anthracyclines, such as doxorubicin, cause the most rapid and striking myocyte damage. This damage is dose-dependent and nearly irreversible, with 98% of injury occurring within the first year of chemotherapy.64 More than one half of cancer patients taking an anthracycline have cardiac dysfunction on imaging; 5% will be in overt heart failure 10 to 20 years, or longer, after chemotherapy.63 Following monitoring at 1 year post-therapy, regular cardiac imaging is not recommended in the absence of symptoms.62
Because other cardiotoxic chemotherapeutic agents cause partially reversible damage, imaging is not recommended in the absence of symptoms in patients taking those agents.64
Continue to: Radiation therapy to the chest leads...
Radiation therapy to the chest leads to many cardiac complications, including cardiomyopathy, valvular disease, pericardial disease, and arrhythmias. Development of cardiomyopathy can be delayed 20 to 30 years after radiation; screening echocardiography is therefore recommended every 5 to 10 years after radiation therapy.65 Recent adjustments to the dosages and delivery of radiation therapy should reduce cardiac damage, but will require decades to validate.63
For patients at risk of cardiovascular disease prior to treatment of cancer, there is evidence to support preventive treatment with angiotensin II-receptor antagonists, beta-blockers, and statins to prevent cardiomyopathy.63 Treatment of diagnosed cardiomyopathy and heart failure follows standard guidelines, with significant emphasis on aerobic exercise and smoking cessation.63
Cancer survivorship care: Your critical role
Cancer survivors constitute a large population who frequent the practices of primary care physicians. Primary care visits provide an opportunity to monitor key elements of survivorship, including surveillance of the current cancer and screening for second cancers. Similar to what is seen with diabetes and coronary artery disease, cancer increases cardiac risk, which requires preventive care and chronic management. FPs are well placed to treat common issues in cancer survivors—issues that mirror concerns seen in the general population.
CORRESPONDENCE
Michael J. Arnold, MD, CDR, USN, Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Bethesda, MD 20814; michael.arnold@usuhs.edu.
ACKNOWLEDGEMENT
Kristian Sanchack, MD, and James Higgins, DO, assisted with the editing of the manuscript.
1. American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2016-2017. Atlanta, GA: American Cancer Society; 2016. www.cancer.org/research/cancer-facts-statistics/survivor-facts-figures.html. Accessed July 25, 2018.
2. Survivorship. NCCN Guidelines (version 1.2017). Fort Washington, PA: National Comprehensive Cancer Network; 2017. www.nccn.org/professionals/physician_gls/default.aspx#supportive. Accessed July 26, 2018.
3. Kendall C, Decker KM, Groome PA, et al. Use of physician services during the survivorship phase: a multi-province study of women diagnosed with breast cancer. Curr Oncolog. 2017;24:81-89.
4. Snyder CF, Earle CC, Herbert RJ, et al. Preventive care for colorectal cancer survivors: a 5-year longitudinal study. J Clin Oncol. 2008;26:1073-1079.
5. Hewitt M, Greenfield S, Stovall E (eds); Institute of Medicine and National Research Council. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington DC: The National Academies Press; 2006. www.nap.edu/read/11468/chapter/1. Accessed July 25, 2018.
6. Survivorship care plan. New York, NY: Memorial Sloan Kettering Cancer Center. www.mskcc.org/hcp-education-training/survivorship/survivorship-care-plan. Accessed August 11, 2018.
7. Fuentes AC, Lambird JE, George TJ, et al. Cancer survivor’s history and physical. South Med J. 2017;110:37-44. http://sma.org/southern-medical-journal/article/cancer-survivors-history-physical/. Accessed July 26, 2018.
8. Salz T, Oeffinger KC, McCabe MS, et al. Survivorship care plans in research and practice. CA Cancer J Clin. 2012;62:101-117.
9. Birken SA, Mayer DK, Weiner BJ. Survivorship care plans: prevalence and barriers to use. J Cancer Educ. 2013;28:290-296.
10. American College of Surgeons Commission on Cancer. Cancer program standards 2012: Ensuring patient-centered care. V1.2.1. www.facs.org/~/media/files/quality%20programs/cancer/coc/programstandards2012.ashx. Accessed July 25, 2018.
11. NCCN guidelines for treatment of cancer by site. NCCN Guidelines (version 1.2018). Fort Washington, PA: National Comprehensive Cancer Network; 2018. www.nccn.org/professionals/physician_gls/default.aspx#site. Accessed July 25, 2018.
12. Spronka I, Korevaar JC, Burgers JS, et al. Review of guidance on recurrence risk management for general practitioners in breast cancer, colorectal cancer and melanoma guidelines. Family Pract. 2017;34:154-160.
13. Merkow RP, Korenstein D, Yeahia R, et al. Quality of cancer surveillance clinical practice guidelines: specificity and consistency of recommendations. JAMA Intern Med. 2017;177:701-709.
14. Muradali D, Kennedy EB, Eisen A, et al. Breast screening for survivors of breast cancer: a systematic review. Prev Med. 2017;103:70-75.
15. Giannarini G, Kessler TM, Thoeny HC, et al. Do patients benefit from routine follow-up to detect recurrences after radical cystectomy and ileal orthotopic bladder substitution? Eur Urol. 2010;58:486-494.
16. Sisler JJ, Seo B, Katz A, et al. Concordance with ASCO guidelines for surveillance after colorectal cancer treatment: a population-based analysis. J Oncol Pract. 2012;8:e69-e79.
17. Ehdaie B. Atoria CL, Lowrance WT, et al. Adherence to surveillance guidelines after radical cystectomy: a population-based analysis. Urol Oncol. 2014;32:779-784.
18. Travis LB, Fosså SD, Schonfeld SJ, et al. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst. 2005;97:1354-1365.
19. Donin N, Filson C, Drakaki A, et al. Risk of second primary malignancies among cancer survivors in the United States, 1992 through 2008. Cancer. 2016;122:3075-3086.
20. Holder AE, Ramirez AG, Gallion K. Depressive symptoms in Latina breast cancer survivors: a barrier to cancer screening. Health Psycholog. 2014;33:242-248.
21. Dyer G, Larsen SR, Gilroy N, et al. Adherence to cancer screening guidelines in Australian survivors of allogenic blood and marrow transplantation (BMT). Cancer Med. 2016;5:1702-1716.
22. Mandelzweig L, Chetrit A, Amitai T, et al. Primary prevention and screening practices among long-term breast cancer survivors. Cancer Causes Control. 2017;28:657-666.
23. Bishop MM, Lee SJ, Beaumont JL, et al. The preventive health behaviors of long-term survivors of cancer and hematopoietic stem cell transplantation compared with matched controls. Biol Blood Marrow Transplant. 2010;16:207-214.
24. Uhlig A, Mei J, Baik I, et al. Screening utilization among cancer survivors: a meta-analysis. J Public Health (Oxf). 2018;40:129-137.
25. Hilal T, Rudy DW. Radiation-induced breast cancer: the question of early breast cancer screening in Hodgkin’s lymphoma survivors. Oxf Med Case Reports. 2016;2016:17-18.
26. Lin K, Croswell JM, Koenig H, et al. Prostate-specific antigen-based screening for prostate cancer: an evidence update for the U.S. Preventive Services Task Force [Internet]. Evidence Syntheses No. 90. AHRQ Publication No. 12-05160-EF-1. Rockville, MD: Agency for Healthcare Research and Quality (US); October 2011. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0032900/. Accessed July 25, 2018.
27. US Preventive Services Task Force. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
28. Hofman M, Ryan JL, Figueroa-Moseley CD, et al. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12 Suppl 1:4-10.
29. Jung JY, Lee JM, Kim MS, et al. Comparison of fatigue, depression, and anxiety as factors affecting posttreatment health-related quality of life in lung cancer survivors. Psychooncology. 2018;27:465-470.
30. Bower JE. Cancer-related fatigue--mechanisms, risk factors, and treatment. Nat Rev Clin Oncol. 2014;11:597-609.
31. Medysky ME, Temesi J, Culos-Reed SN, et al. Exercise, sleep and cancer-related fatigue: are they related? Neurophysiol Clin. 2017;47:111-122.
32. Oh HS, Sea WS. Systematic review and meta-analysis of the correlates of cancer-related fatigue. Worldviews Evid Based Nurs. 2011;8:191-201.
33. Qu D, Zhang Z, Yu X, et al. Psychotropic drugs for the management of cancer-related fatigue: a systematic review and meta-analysis. Eur J Cancer Care (Engl). 2016;25:970-979.
34. Escalante CP, Manzullo EF. Cancer-related fatigue: the approach and treatment. J Gen Intern Med. 2009;24(suppl 2):S412-S416.
35. Mustian KM, Alfano CM, Heckler C, et al. Comparison of pharmaceutical, psychological, and exercise treatments for cancer-related fatigue: a meta-analysis. JAMA Oncol. 2017;3:961-968.
36. Glare PA, Davies PS, Finlay E, et al. Pain in cancer survivors. J Clin Oncol. 2014;32:1739-1747.
37. Davies PS. Chronic pain management in the cancer survivor: tips for primary care providers. Nurse Pract. 2013;39:28-38.
38. Boland EG, Ahmedzai SH. Persistent pain in cancer survivors. Curr Opin Support Palliat Care. 2017;11:181-190.
39. Sears CS, Robinson JW, Walker LM. A comprehensive review of sexual health concerns after cancer treatment and the biopsychosocial treatment options available to female patients. Eur J Cancer Care (Engl). 2017;27:e12738.
40. Schou Bredal I, Smeby NA, Ottesen S, et al. Chronic pain in breast cancer survivors: comparison of psychological, surgical, and medical characteristics between survivors with and without pain. J Pain Symptom Manage. 2014;48:852-862.
41. Sutradhar R, Lokku A, Barbera L. Cancer survivorship and opioid prescribing rates: a population-based matched cohort study among individuals with and without a history of cancer. Cancer. 2017;123:4286-4293.
42. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain - United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
43. Davis MP, Mehta Z. Opioids and chronic pain: where is the balance? Curr Oncol Rep. 2016;18:71.
44. Von Ah D. Cognitive changes associated with cancer and cancer treatment: state of the science. Clin J Oncol Nurs. 2015;19:47-56.
45. Moore HC. An overview of chemotherapy-related cognitive dysfunction, or ‘chemobrain’. Oncology (Williston Park). 2014;28:797-804.
46. Asher A. Cognitive dysfunction among cancer survivors. Am J Phys Med Rehabil. 2011;90(suppl):S16-S26.
47. Joly F, Rigal O, Noal S, et al. Cognitive dysfunction and cancer: which consequences in terms of disease management? Psychooncology. 2011;20:1251-1258.
48. Attention, thinking or memory problems. American Society of Clinical Oncology Cancer.Net. April 2018. www.cancer.net/navigating-cancer-care/side-effects/attention-thinking-or-memory-problems. Accessed July 25, 2018.
49. Kucherer S, Ferguson RJ. Cognitive behavioral therapy for cancer-related cognitive dysfunction. Curr Opin Support Palliat Care. 2017;11:46-51.
50. Derry HM, Jaremka LM, Bennet JM, et al. Yoga and self-reported cognitive problems in breast cancer survivors: a randomized controlled trial. Psychooncology. 2015;24:958-966.
51. Treanor CJ, McMenamin UC, O’Neill RF, et al. Non-pharmacological interventions for cognitive impairment due to systemic cancer treatment. Cochrane Database Syst Rev. 2016 Aug 16;(8):CD011325.
52. Mitchell AJ, Ferguson DW, Gill J, et al. Depression and anxiety in long-term cancer survivors compared with spouses and healthy controls: a systematic review and meta-analysis. Lancet Oncol. 2013;14:721-732.
53. Inhestern L, Beierlein V, Bultmann JC, et al. Anxiety and depression in working-age cancer survivors: a register-based study. BMC Cancer. 2017;17:347.
54. Partridge AH, Jacobsen PB, Andersen BL. Challenges to standardizing the care for adult cancer survivors: highlighting ASCO’s fatigue and anxiety and depression guidelines. Am Soc Clin Oncol Educ Book. 2015:188-194.
55. Andersen BL, Yang HC, Farrar WB, et al. Psychologic intervention improves survival for breast cancer patients: a randomized clinical trial. Cancer. 2008;113:3450-3458.
56. Yi JC, Syrjala KL. Anxiety and depression in cancer survivors. Med Clin N Am. 2017;101:1099-1113.
57. Zhu G, Zhang X, Wang Y, et al. Effects of exercise intervention in breast cancer survivors: a meta-analysis of 33 randomized controlled trials. Onco Targets Ther. 2016;9:2153-2168.
58. Hawkins NA, Soman A, Lunsford N, et al. Use of medications for treating anxiety and depression in cancer survivors in the United States. J Clin Oncol. 2017;35:78-85.
59. Ostuzzi G, Matcham F, Dauchy S, et al. Antidepressants for the treatment of depression in people with cancer. Cochrane Database Syst Rev. 2015 June 1;(6):CD011006.
60. Laoutidis ZG, Mathiak K. Antidepressants in the treatment of depression/depressive symptoms in cancer patients: a systematic review and meta-analysis. BMC Psychiatry. 2013;13:140.
61. Lydiatt WM, Bessette D, Schmid KK, et al. Prevention of depression with escitalopram in patients undergoing treatment for head and neck cancer: randomized, double-blind, placebo-controlled clinical trial. JAMA Otolaryngol Head Neck Surg. 2013;139:678-686.
62. Lavigne JE, Heckler C, Mathews JL, et al. A randomized, controlled, double-blinded clinical trial of gabapentin 300 versus 900 mg versus placebo for anxiety symptoms in breast cancer survivors. Breast Cancer Res Treat. 2012;136:479-486.
63. Okwuosa TM, Anzevino S, Rao R. Cardiovascular disease in cancer survivors. Postgrad Med J. 2017;93:82-90.
64. Plana, JC, Galderisi M, Barac A, et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2014;27:911-939.
65. Lancellotti, P, Nkomo VT, Badano LP, et al. Expert consensus for multi-modality imaging evaluation of cardiovascular complications of radiotherapy in adults: a report from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:1013-1032.
Cancer survivors represent a rapidly increasing population. In 1971, there were 3 million cancer survivors; this number increased to 15.5 million in 2016 and will reach 20 million by 2026.1TABLE 11 shows the percentage of survivors by type of cancer. Cancer survivors tend to be older,* comprising nearly 1 of every 5 people older than 65 years.2

The Institute of Medicine (IOM) identified 3 key characteristics of cancer survivors3:
- Trajectories of survivorship are variable; many cancer patients have periods of relative health between episodes of their disease.
- Survivors require careful cancer monitoring; in addition to the risk that their primary cancer will recur, they have an elevated risk for another, second cancer.
- Both cancer and its treatments increase the risk of other medical and psychiatric problems.
Family physicians (FPs) have optimal skills for navigating the chronic risks and health concerns of the well cancer survivor. This article reviews the primary care management of the functional cancer survivor, focusing on the management of chronic conditions and preventive care.
Survivorship follows any of 6 paths
Cancer survivorship is increasing in importance as treatment has steadily reduced mortality. Six trajectories of cancer survivors have been identified1:
- living cancer-free after treatment with minimal effects
- living cancer-free but suffering serious treatment complications
- Suffering late recurrence
- Developing a second cancer
- Living with intermittent cancer recurrences
- Living with cancer continuously.
Only patients in the last 2 groups are likely to be managed primarily by oncologists.
Survivors look to their FPs for ongoing care
Cancer survivors routinely see their primary care physician after initial treatment. A study of 30,000 Canadian breast cancer survivors demonstrated that follow-up care was limited to an oncologist in only 2%; 84% saw a primary care provider and an oncologist; and 14% saw a primary care provider only.4 A study of colorectal cancer survivors showed that primary care visits increased in each of the 5 years after diagnosis, during which time oncology visits decreased steadily5; in that study, primary care physicians delivered more preventive care than oncologists did.5 Similar to what is done in other chronic conditions, the various effects of cancer are best managed as a whole.
The IOM recommends that cancer survivor care comprise 4 elements2:
- coordination between oncologist and primary care physician
- surveillance for recurrence or spread of existing cancer
- screening for new cancer
- intervention for the effects of cancer and treatment.
Continue to: The following discussion summarizes...
The following discussion summarizes evidence and recommendations for each element of the IOM recommendations for survivor care.
Implementing the 4 elements of cancer survivor care
1. Coordinate care through a unified survivorship care plan
The IOM has noted that the needs of cancer survivors are rarely met2; communication between oncology and primary care is often deficient during transition of care. The IOM has recommended that oncologists provide a survivorship care plan that details the cancer (ie, tumor characteristics), the type of treatment (ie, enrollment in a clinical trial; medical, surgical, or radiation), support services, and follow-up recommendations for the primary care provider. (Examples of elements of a survivorship care plan can be found at www.mskcc.org/hcp-education-training/survivorship/survivorship-care-plan6 and http://sma.org/southern-medical-journal/article/cancer-survivors-history-physical/7).
Regrettably, survivorship care plans have been rarely and poorly employed. Studies show that fewer than one-half of oncologists provide a plan, and that when they do, the plan often lacks recommended information.8,9 Survivorship care plans may soon become common practice, however; the Commission on Cancer of the American College of Surgeons has required their use in all certified cancer centers since 2015.10
2. Provide surveillance of existing cancer

Cancer follow-up is challenging after the initial treatment phase. Although there are many conflicting guidelines for surveillance after cancer, guidelines of the National Comprehensive Cancer Network (NCCN) (summarized in TABLE 211 for the 10 most common cancers in survivors) are the ones generally accepted.12,13

Although individual surveillance recommendations are based on limited evidence, studies confirm the importance of surveillance. A systematic review showed that surveillance mammography after breast cancer reduces breast cancer mortality by 36%.14 A study showed that bladder cancer recurrence diagnosed by surveillance instead of by symptoms led to a 35% increase in 5-year survival.15

Continue to: Yet adherence to cancer surveillance...
Yet adherence to cancer surveillance recommendations is poor. A study of patients with colon cancer demonstrated that only 12% met all recommended surveillance guidelines.16 A study of patients with bladder cancer after radical cystectomy showed that only 9% met recommended surveillance more than 2 years after diagnosis.17 Those dismal statistics may be the result of provider oversight—not patient reluctance.
In the colon cancer study, for example, compliance with follow-up colonoscopy was 80% but compliance with carcinoembryonic antigen testing was only 22%.16 In the bladder cancer study, follow-up urine cytology was obtained in only 23% of patients, although 75% completed recommended imaging.17
Although surveillance remains the oncologist’s responsibility, visits to the FP provide an opportunity to review surveillance and order needed laboratory testing and other studies, including imaging.
3. Screen for new cancers
The risk of a second cancer is elevated for cancer survivors compared with the risk of a primary cancer in the healthy general population; some survivors have a lifetime risk of a second cancer as high as 36%.18 Risk varies by cancer type (TABLE 319). Some of this variation is due to the impact of smoking: Smoking-related cancers have the highest risk of second malignancy.19 Genetic predisposition to malignant transformation is also theorized to contribute to increased risk. Second malignancies are dangerous; 55% of patients die of the second cancer compared with only 13% of their initial cancer.19

Studies show that cancer survivors display varying adherence with recommended screening for second cancers. In a study of Latina cancer survivors, depressive symptoms were associated with lower screening compliance.20 A study of survivors of hematologic cancer showed a low rate of cancer screening and high fear of cancer recurrence—suggesting avoidance due to fear.21 Other studies, however, show similar or increased compliance with screening in cancer survivors.22,23 A meta-analysis of 19 studies determined that, overall, cancer survivors receive 25% to 38% more recommended screening than the general population.24
Continue to: Few guidelines exist to guide FPs...
Few guidelines exist to guide FPs in adjusting screening for the cancer survivor. For women who received radiation therapy for a tumor in the chest, for example, the recommendation offered by several groups is to start breast cancer screening 8 to 10 years after treatment or by 30 years of age, and to consider combining magnetic resonance imaging and mammography.25 Recommendations for breast cancer screening do not account for a history of other gynecologic cancers unless genetic markers are present.25 On the other hand, the impact of a history of cancer on the risk of prostate cancer and on screening decisions has not been studied,26 and cervical cancer screening guidelines, which recommend that screening continue after 65 years of age for patients who are immunocompromised, do not address a history of other cancer.27
4. Manage the effects of both the cancer and the treatment
Medical issues faced by cancer survivors are familiar to FPs, but there are some specific recommendations regarding evaluation and treatment that stand in contrast to what would be considered for a healthy, or non-cancer, patient. For example, each chemotherapeutic agent has characteristic adverse effects; TABLE 47 lists the principal adverse effects of common agents and recommendations for testing when these problems develop. Common long-term problems in cancer survivors include fatigue, chronic pain, cognitive dysfunction, psychiatric illness, and cardiovascular disease. Although these symptoms and manifestations are common, the physician must be careful: New or changing symptoms could signal the spread or recurrence of disease. Fear of recurrence can lead patients to exaggerate or minimize symptoms.

Fatigue is the most common symptom seen in cancer survivors during treatment and following remission.28 More than 40% of cancer survivors report significant fatigue.29 Although fatigue is concerning for cancer recurrence, other causes are common in cancer survivors. Both depression and anxiety commonly present with worsened fatigue.30 Sleep disturbances are common, even without a psychiatric diagnosis.31 Effects of treatment, including nausea, anemia, heart failure, and medication adverse effects can cause or worsen fatigue. Pain is associated with fatigue, but to a lesser extent than are depression, anxiety, and nausea.32

Pharmacotherapy of cancer-related fatigue is challenging. Psychostimulants have been most studied. A recent systematic review shows that methylphenidate produces mild or moderate improvement in fatigue, whereas modafanil has minimal effectiveness.33 Antidepressants have not been shown to relieve fatigue.33
A recent meta-analysis showed that nonpharmaceutical treatments for cancer-related fatigue are more effective than pharmacotherapy. In this review, both exercise and pharmacotherapy had a mild-to-moderate effect on fatigue.35 Exercise is best studied in this regard, and has shown the most consistent results.31
Continue to: Chronic pain
Chronic pain. Pain is common in cancer survivors: As many as 40% experience pain for years after initial therapy.36 Treatment of some cancers—eg, thoracotomy (80%), amputation (50%-80%), neck dissection (52%), and surgical management of breast cancer (63%)—increase the likelihood of chronic pain.37 Reports of pain in cancer survivors that should be considered red flags that might signal recurrence of cancer include new or worsening pain; pain worse at night or when recumbent; new neurologic symptoms; and general symptoms of systemic illness37 (TABLE 537).

Management of pain is best approached by its cause, with neurologic, rheumatologic (including myofascial pain and arthralgia), lymphatic, and genital causes most common.37 Across all types of pain, complete relief is unlikely; functional goals provide a more effective target.
For neuropathic cancer pain, duloxetine is the only medication with evidence of benefit; anticonvulsant and topical medications are recommended on the basis of the findings of studies of noncancer pain.38 There are few data on the value of treatments for cancer-related rheumatologic and lymphatic pain, although exercise has shown benefit in both types.38 For dyspareunia and sexual dysfunction (common after gynecologic and nongynecologic cancers), vaginal lubricants and pelvic-floor physiotherapy have shown benefit.39 There is significant overlap in psychiatric comorbidities, sleep, and pain, and addressing all of a patient’s problems can reduce pain and improve function.40
Opioids are often prescribed for pain in cancer survivors. Cancer survivors have a higher rate of opioid prescribing compared with that of non-cancer patients, even 10 years after diagnosis.41 Guidelines of the Centers for Disease Control and Prevention for using opioids to manage chronic pain specifically exclude cancer patients.42 Regrettably, there is no evidence that opioids have long-term efficacy in chronic pain; in fact, evidence is accumulating that chronic opioid therapy exacerbates chronic pain.43
Cognitive dysfunction is present in 17% to 75% of cancer survivors as memory disturbance, psychological disorder, sleep dysfunction, or impairment of executive functioning.44 Cognitive deficits appear to be secondary to both cancer and treatment modalities45; as many as one-third of patients have cognitive dysfunction prior to receiving chemotherapy.46
Continue to: Chemotherapies that are more likely...
Chemotherapies that are more likely to cause cognitive symptoms include methotrexate, 5-fluorouracil, cyclophosphamide, and hormone antagonists.47 More powerful regimens and repetitive chemotherapy regimens tend to cause more cognitive effects.47
Cognitive training interventions show evidence of likely benefit,44,48 leading to recommendations for self-treatment strategies, such as written lists, wordplay, crossword puzzles, jigsaw puzzles, playing a musical instrument, and new hobbies. Small studies suggest a benefit from cognitive behavioral therapy.44,49 A study of breast cancer survivors showed that yoga led to improvement in patient-reported cognitive dysfunction.50 Physical exercise yields cognitive benefit in healthy older adults and is supported by limited evidence in cancer survivors.51
There is no effective pharmacotherapy for cancer- and cancer chemotherapy-related cognitive dysfunction unless a treatable underlying cause is found.44 Symptoms tend to subside with time after completion of chemotherapy, which might be reassuring to patients and families.45
Psychiatric problems. The most common psychiatric issues in cancer survivors are anxiety and depression; the prevalence of anxiety is nearly double that of depression.52 Anxiety often presents as fear of a recurrence of cancer or a feeling of lack of control over present or future circumstances.53 Screening for anxiety and depression is recommended at each visit, using standardized screening questionnaires.54
A small study suggests that psychiatric treatment reduces the risk of early mortality.55 Small studies also suggest that mindfulness-based therapy and cognitive behavioral therapy delivered by telehealth offer benefit.56 A meta-analysis shows that exercise interventions improve depression and anxiety in breast cancer patients.57
Continue to: There are few studies of pharmacotherapy...
There are few studies of pharmacotherapy of anxiety or depression in cancer survivors56; it is known that cancer survivors are nearly twice as likely as the general population to be taking medical therapy for anxiety and depression.58 A Cochrane systematic review of 7 small studies showed uncertain improvement in depressive symptoms in patients with cancer from antidepressant medication; however, an earlier systematic review did show benefit.59,60
In a trial of patients without depression who were being treated for head and neck cancer, escitalopram, 20 mg/d, reduced the risk of subsequent depression compared with placebo.61 A study of 420 breast cancer survivors showed that 300 mg/d and 900 mg/d dosages of gabapentin were both superior to placebo, and nearly equivalent to each other, at reducing anxiety scores.62 In both studies, however, the evidence is nonetheless insufficient to make specific recommendations about these medications.
Cardiac risk
Among chemotherapeutic agents, anthracyclines, such as doxorubicin, cause the most rapid and striking myocyte damage. This damage is dose-dependent and nearly irreversible, with 98% of injury occurring within the first year of chemotherapy.64 More than one half of cancer patients taking an anthracycline have cardiac dysfunction on imaging; 5% will be in overt heart failure 10 to 20 years, or longer, after chemotherapy.63 Following monitoring at 1 year post-therapy, regular cardiac imaging is not recommended in the absence of symptoms.62
Because other cardiotoxic chemotherapeutic agents cause partially reversible damage, imaging is not recommended in the absence of symptoms in patients taking those agents.64
Continue to: Radiation therapy to the chest leads...
Radiation therapy to the chest leads to many cardiac complications, including cardiomyopathy, valvular disease, pericardial disease, and arrhythmias. Development of cardiomyopathy can be delayed 20 to 30 years after radiation; screening echocardiography is therefore recommended every 5 to 10 years after radiation therapy.65 Recent adjustments to the dosages and delivery of radiation therapy should reduce cardiac damage, but will require decades to validate.63
For patients at risk of cardiovascular disease prior to treatment of cancer, there is evidence to support preventive treatment with angiotensin II-receptor antagonists, beta-blockers, and statins to prevent cardiomyopathy.63 Treatment of diagnosed cardiomyopathy and heart failure follows standard guidelines, with significant emphasis on aerobic exercise and smoking cessation.63
Cancer survivorship care: Your critical role
Cancer survivors constitute a large population who frequent the practices of primary care physicians. Primary care visits provide an opportunity to monitor key elements of survivorship, including surveillance of the current cancer and screening for second cancers. Similar to what is seen with diabetes and coronary artery disease, cancer increases cardiac risk, which requires preventive care and chronic management. FPs are well placed to treat common issues in cancer survivors—issues that mirror concerns seen in the general population.
CORRESPONDENCE
Michael J. Arnold, MD, CDR, USN, Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Bethesda, MD 20814; michael.arnold@usuhs.edu.
ACKNOWLEDGEMENT
Kristian Sanchack, MD, and James Higgins, DO, assisted with the editing of the manuscript.
Cancer survivors represent a rapidly increasing population. In 1971, there were 3 million cancer survivors; this number increased to 15.5 million in 2016 and will reach 20 million by 2026.1TABLE 11 shows the percentage of survivors by type of cancer. Cancer survivors tend to be older,* comprising nearly 1 of every 5 people older than 65 years.2

The Institute of Medicine (IOM) identified 3 key characteristics of cancer survivors3:
- Trajectories of survivorship are variable; many cancer patients have periods of relative health between episodes of their disease.
- Survivors require careful cancer monitoring; in addition to the risk that their primary cancer will recur, they have an elevated risk for another, second cancer.
- Both cancer and its treatments increase the risk of other medical and psychiatric problems.
Family physicians (FPs) have optimal skills for navigating the chronic risks and health concerns of the well cancer survivor. This article reviews the primary care management of the functional cancer survivor, focusing on the management of chronic conditions and preventive care.
Survivorship follows any of 6 paths
Cancer survivorship is increasing in importance as treatment has steadily reduced mortality. Six trajectories of cancer survivors have been identified1:
- living cancer-free after treatment with minimal effects
- living cancer-free but suffering serious treatment complications
- Suffering late recurrence
- Developing a second cancer
- Living with intermittent cancer recurrences
- Living with cancer continuously.
Only patients in the last 2 groups are likely to be managed primarily by oncologists.
Survivors look to their FPs for ongoing care
Cancer survivors routinely see their primary care physician after initial treatment. A study of 30,000 Canadian breast cancer survivors demonstrated that follow-up care was limited to an oncologist in only 2%; 84% saw a primary care provider and an oncologist; and 14% saw a primary care provider only.4 A study of colorectal cancer survivors showed that primary care visits increased in each of the 5 years after diagnosis, during which time oncology visits decreased steadily5; in that study, primary care physicians delivered more preventive care than oncologists did.5 Similar to what is done in other chronic conditions, the various effects of cancer are best managed as a whole.
The IOM recommends that cancer survivor care comprise 4 elements2:
- coordination between oncologist and primary care physician
- surveillance for recurrence or spread of existing cancer
- screening for new cancer
- intervention for the effects of cancer and treatment.
Continue to: The following discussion summarizes...
The following discussion summarizes evidence and recommendations for each element of the IOM recommendations for survivor care.
Implementing the 4 elements of cancer survivor care
1. Coordinate care through a unified survivorship care plan
The IOM has noted that the needs of cancer survivors are rarely met2; communication between oncology and primary care is often deficient during transition of care. The IOM has recommended that oncologists provide a survivorship care plan that details the cancer (ie, tumor characteristics), the type of treatment (ie, enrollment in a clinical trial; medical, surgical, or radiation), support services, and follow-up recommendations for the primary care provider. (Examples of elements of a survivorship care plan can be found at www.mskcc.org/hcp-education-training/survivorship/survivorship-care-plan6 and http://sma.org/southern-medical-journal/article/cancer-survivors-history-physical/7).
Regrettably, survivorship care plans have been rarely and poorly employed. Studies show that fewer than one-half of oncologists provide a plan, and that when they do, the plan often lacks recommended information.8,9 Survivorship care plans may soon become common practice, however; the Commission on Cancer of the American College of Surgeons has required their use in all certified cancer centers since 2015.10
2. Provide surveillance of existing cancer

Cancer follow-up is challenging after the initial treatment phase. Although there are many conflicting guidelines for surveillance after cancer, guidelines of the National Comprehensive Cancer Network (NCCN) (summarized in TABLE 211 for the 10 most common cancers in survivors) are the ones generally accepted.12,13

Although individual surveillance recommendations are based on limited evidence, studies confirm the importance of surveillance. A systematic review showed that surveillance mammography after breast cancer reduces breast cancer mortality by 36%.14 A study showed that bladder cancer recurrence diagnosed by surveillance instead of by symptoms led to a 35% increase in 5-year survival.15

Continue to: Yet adherence to cancer surveillance...
Yet adherence to cancer surveillance recommendations is poor. A study of patients with colon cancer demonstrated that only 12% met all recommended surveillance guidelines.16 A study of patients with bladder cancer after radical cystectomy showed that only 9% met recommended surveillance more than 2 years after diagnosis.17 Those dismal statistics may be the result of provider oversight—not patient reluctance.
In the colon cancer study, for example, compliance with follow-up colonoscopy was 80% but compliance with carcinoembryonic antigen testing was only 22%.16 In the bladder cancer study, follow-up urine cytology was obtained in only 23% of patients, although 75% completed recommended imaging.17
Although surveillance remains the oncologist’s responsibility, visits to the FP provide an opportunity to review surveillance and order needed laboratory testing and other studies, including imaging.
3. Screen for new cancers
The risk of a second cancer is elevated for cancer survivors compared with the risk of a primary cancer in the healthy general population; some survivors have a lifetime risk of a second cancer as high as 36%.18 Risk varies by cancer type (TABLE 319). Some of this variation is due to the impact of smoking: Smoking-related cancers have the highest risk of second malignancy.19 Genetic predisposition to malignant transformation is also theorized to contribute to increased risk. Second malignancies are dangerous; 55% of patients die of the second cancer compared with only 13% of their initial cancer.19

Studies show that cancer survivors display varying adherence with recommended screening for second cancers. In a study of Latina cancer survivors, depressive symptoms were associated with lower screening compliance.20 A study of survivors of hematologic cancer showed a low rate of cancer screening and high fear of cancer recurrence—suggesting avoidance due to fear.21 Other studies, however, show similar or increased compliance with screening in cancer survivors.22,23 A meta-analysis of 19 studies determined that, overall, cancer survivors receive 25% to 38% more recommended screening than the general population.24
Continue to: Few guidelines exist to guide FPs...
Few guidelines exist to guide FPs in adjusting screening for the cancer survivor. For women who received radiation therapy for a tumor in the chest, for example, the recommendation offered by several groups is to start breast cancer screening 8 to 10 years after treatment or by 30 years of age, and to consider combining magnetic resonance imaging and mammography.25 Recommendations for breast cancer screening do not account for a history of other gynecologic cancers unless genetic markers are present.25 On the other hand, the impact of a history of cancer on the risk of prostate cancer and on screening decisions has not been studied,26 and cervical cancer screening guidelines, which recommend that screening continue after 65 years of age for patients who are immunocompromised, do not address a history of other cancer.27
4. Manage the effects of both the cancer and the treatment
Medical issues faced by cancer survivors are familiar to FPs, but there are some specific recommendations regarding evaluation and treatment that stand in contrast to what would be considered for a healthy, or non-cancer, patient. For example, each chemotherapeutic agent has characteristic adverse effects; TABLE 47 lists the principal adverse effects of common agents and recommendations for testing when these problems develop. Common long-term problems in cancer survivors include fatigue, chronic pain, cognitive dysfunction, psychiatric illness, and cardiovascular disease. Although these symptoms and manifestations are common, the physician must be careful: New or changing symptoms could signal the spread or recurrence of disease. Fear of recurrence can lead patients to exaggerate or minimize symptoms.

Fatigue is the most common symptom seen in cancer survivors during treatment and following remission.28 More than 40% of cancer survivors report significant fatigue.29 Although fatigue is concerning for cancer recurrence, other causes are common in cancer survivors. Both depression and anxiety commonly present with worsened fatigue.30 Sleep disturbances are common, even without a psychiatric diagnosis.31 Effects of treatment, including nausea, anemia, heart failure, and medication adverse effects can cause or worsen fatigue. Pain is associated with fatigue, but to a lesser extent than are depression, anxiety, and nausea.32

Pharmacotherapy of cancer-related fatigue is challenging. Psychostimulants have been most studied. A recent systematic review shows that methylphenidate produces mild or moderate improvement in fatigue, whereas modafanil has minimal effectiveness.33 Antidepressants have not been shown to relieve fatigue.33
A recent meta-analysis showed that nonpharmaceutical treatments for cancer-related fatigue are more effective than pharmacotherapy. In this review, both exercise and pharmacotherapy had a mild-to-moderate effect on fatigue.35 Exercise is best studied in this regard, and has shown the most consistent results.31
Continue to: Chronic pain
Chronic pain. Pain is common in cancer survivors: As many as 40% experience pain for years after initial therapy.36 Treatment of some cancers—eg, thoracotomy (80%), amputation (50%-80%), neck dissection (52%), and surgical management of breast cancer (63%)—increase the likelihood of chronic pain.37 Reports of pain in cancer survivors that should be considered red flags that might signal recurrence of cancer include new or worsening pain; pain worse at night or when recumbent; new neurologic symptoms; and general symptoms of systemic illness37 (TABLE 537).

Management of pain is best approached by its cause, with neurologic, rheumatologic (including myofascial pain and arthralgia), lymphatic, and genital causes most common.37 Across all types of pain, complete relief is unlikely; functional goals provide a more effective target.
For neuropathic cancer pain, duloxetine is the only medication with evidence of benefit; anticonvulsant and topical medications are recommended on the basis of the findings of studies of noncancer pain.38 There are few data on the value of treatments for cancer-related rheumatologic and lymphatic pain, although exercise has shown benefit in both types.38 For dyspareunia and sexual dysfunction (common after gynecologic and nongynecologic cancers), vaginal lubricants and pelvic-floor physiotherapy have shown benefit.39 There is significant overlap in psychiatric comorbidities, sleep, and pain, and addressing all of a patient’s problems can reduce pain and improve function.40
Opioids are often prescribed for pain in cancer survivors. Cancer survivors have a higher rate of opioid prescribing compared with that of non-cancer patients, even 10 years after diagnosis.41 Guidelines of the Centers for Disease Control and Prevention for using opioids to manage chronic pain specifically exclude cancer patients.42 Regrettably, there is no evidence that opioids have long-term efficacy in chronic pain; in fact, evidence is accumulating that chronic opioid therapy exacerbates chronic pain.43
Cognitive dysfunction is present in 17% to 75% of cancer survivors as memory disturbance, psychological disorder, sleep dysfunction, or impairment of executive functioning.44 Cognitive deficits appear to be secondary to both cancer and treatment modalities45; as many as one-third of patients have cognitive dysfunction prior to receiving chemotherapy.46
Continue to: Chemotherapies that are more likely...
Chemotherapies that are more likely to cause cognitive symptoms include methotrexate, 5-fluorouracil, cyclophosphamide, and hormone antagonists.47 More powerful regimens and repetitive chemotherapy regimens tend to cause more cognitive effects.47
Cognitive training interventions show evidence of likely benefit,44,48 leading to recommendations for self-treatment strategies, such as written lists, wordplay, crossword puzzles, jigsaw puzzles, playing a musical instrument, and new hobbies. Small studies suggest a benefit from cognitive behavioral therapy.44,49 A study of breast cancer survivors showed that yoga led to improvement in patient-reported cognitive dysfunction.50 Physical exercise yields cognitive benefit in healthy older adults and is supported by limited evidence in cancer survivors.51
There is no effective pharmacotherapy for cancer- and cancer chemotherapy-related cognitive dysfunction unless a treatable underlying cause is found.44 Symptoms tend to subside with time after completion of chemotherapy, which might be reassuring to patients and families.45
Psychiatric problems. The most common psychiatric issues in cancer survivors are anxiety and depression; the prevalence of anxiety is nearly double that of depression.52 Anxiety often presents as fear of a recurrence of cancer or a feeling of lack of control over present or future circumstances.53 Screening for anxiety and depression is recommended at each visit, using standardized screening questionnaires.54
A small study suggests that psychiatric treatment reduces the risk of early mortality.55 Small studies also suggest that mindfulness-based therapy and cognitive behavioral therapy delivered by telehealth offer benefit.56 A meta-analysis shows that exercise interventions improve depression and anxiety in breast cancer patients.57
Continue to: There are few studies of pharmacotherapy...
There are few studies of pharmacotherapy of anxiety or depression in cancer survivors56; it is known that cancer survivors are nearly twice as likely as the general population to be taking medical therapy for anxiety and depression.58 A Cochrane systematic review of 7 small studies showed uncertain improvement in depressive symptoms in patients with cancer from antidepressant medication; however, an earlier systematic review did show benefit.59,60
In a trial of patients without depression who were being treated for head and neck cancer, escitalopram, 20 mg/d, reduced the risk of subsequent depression compared with placebo.61 A study of 420 breast cancer survivors showed that 300 mg/d and 900 mg/d dosages of gabapentin were both superior to placebo, and nearly equivalent to each other, at reducing anxiety scores.62 In both studies, however, the evidence is nonetheless insufficient to make specific recommendations about these medications.
Cardiac risk
Among chemotherapeutic agents, anthracyclines, such as doxorubicin, cause the most rapid and striking myocyte damage. This damage is dose-dependent and nearly irreversible, with 98% of injury occurring within the first year of chemotherapy.64 More than one half of cancer patients taking an anthracycline have cardiac dysfunction on imaging; 5% will be in overt heart failure 10 to 20 years, or longer, after chemotherapy.63 Following monitoring at 1 year post-therapy, regular cardiac imaging is not recommended in the absence of symptoms.62
Because other cardiotoxic chemotherapeutic agents cause partially reversible damage, imaging is not recommended in the absence of symptoms in patients taking those agents.64
Continue to: Radiation therapy to the chest leads...
Radiation therapy to the chest leads to many cardiac complications, including cardiomyopathy, valvular disease, pericardial disease, and arrhythmias. Development of cardiomyopathy can be delayed 20 to 30 years after radiation; screening echocardiography is therefore recommended every 5 to 10 years after radiation therapy.65 Recent adjustments to the dosages and delivery of radiation therapy should reduce cardiac damage, but will require decades to validate.63
For patients at risk of cardiovascular disease prior to treatment of cancer, there is evidence to support preventive treatment with angiotensin II-receptor antagonists, beta-blockers, and statins to prevent cardiomyopathy.63 Treatment of diagnosed cardiomyopathy and heart failure follows standard guidelines, with significant emphasis on aerobic exercise and smoking cessation.63
Cancer survivorship care: Your critical role
Cancer survivors constitute a large population who frequent the practices of primary care physicians. Primary care visits provide an opportunity to monitor key elements of survivorship, including surveillance of the current cancer and screening for second cancers. Similar to what is seen with diabetes and coronary artery disease, cancer increases cardiac risk, which requires preventive care and chronic management. FPs are well placed to treat common issues in cancer survivors—issues that mirror concerns seen in the general population.
CORRESPONDENCE
Michael J. Arnold, MD, CDR, USN, Uniformed Services University of the Health Sciences, 4301 Jones Bridge Road, Bethesda, MD 20814; michael.arnold@usuhs.edu.
ACKNOWLEDGEMENT
Kristian Sanchack, MD, and James Higgins, DO, assisted with the editing of the manuscript.
1. American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2016-2017. Atlanta, GA: American Cancer Society; 2016. www.cancer.org/research/cancer-facts-statistics/survivor-facts-figures.html. Accessed July 25, 2018.
2. Survivorship. NCCN Guidelines (version 1.2017). Fort Washington, PA: National Comprehensive Cancer Network; 2017. www.nccn.org/professionals/physician_gls/default.aspx#supportive. Accessed July 26, 2018.
3. Kendall C, Decker KM, Groome PA, et al. Use of physician services during the survivorship phase: a multi-province study of women diagnosed with breast cancer. Curr Oncolog. 2017;24:81-89.
4. Snyder CF, Earle CC, Herbert RJ, et al. Preventive care for colorectal cancer survivors: a 5-year longitudinal study. J Clin Oncol. 2008;26:1073-1079.
5. Hewitt M, Greenfield S, Stovall E (eds); Institute of Medicine and National Research Council. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington DC: The National Academies Press; 2006. www.nap.edu/read/11468/chapter/1. Accessed July 25, 2018.
6. Survivorship care plan. New York, NY: Memorial Sloan Kettering Cancer Center. www.mskcc.org/hcp-education-training/survivorship/survivorship-care-plan. Accessed August 11, 2018.
7. Fuentes AC, Lambird JE, George TJ, et al. Cancer survivor’s history and physical. South Med J. 2017;110:37-44. http://sma.org/southern-medical-journal/article/cancer-survivors-history-physical/. Accessed July 26, 2018.
8. Salz T, Oeffinger KC, McCabe MS, et al. Survivorship care plans in research and practice. CA Cancer J Clin. 2012;62:101-117.
9. Birken SA, Mayer DK, Weiner BJ. Survivorship care plans: prevalence and barriers to use. J Cancer Educ. 2013;28:290-296.
10. American College of Surgeons Commission on Cancer. Cancer program standards 2012: Ensuring patient-centered care. V1.2.1. www.facs.org/~/media/files/quality%20programs/cancer/coc/programstandards2012.ashx. Accessed July 25, 2018.
11. NCCN guidelines for treatment of cancer by site. NCCN Guidelines (version 1.2018). Fort Washington, PA: National Comprehensive Cancer Network; 2018. www.nccn.org/professionals/physician_gls/default.aspx#site. Accessed July 25, 2018.
12. Spronka I, Korevaar JC, Burgers JS, et al. Review of guidance on recurrence risk management for general practitioners in breast cancer, colorectal cancer and melanoma guidelines. Family Pract. 2017;34:154-160.
13. Merkow RP, Korenstein D, Yeahia R, et al. Quality of cancer surveillance clinical practice guidelines: specificity and consistency of recommendations. JAMA Intern Med. 2017;177:701-709.
14. Muradali D, Kennedy EB, Eisen A, et al. Breast screening for survivors of breast cancer: a systematic review. Prev Med. 2017;103:70-75.
15. Giannarini G, Kessler TM, Thoeny HC, et al. Do patients benefit from routine follow-up to detect recurrences after radical cystectomy and ileal orthotopic bladder substitution? Eur Urol. 2010;58:486-494.
16. Sisler JJ, Seo B, Katz A, et al. Concordance with ASCO guidelines for surveillance after colorectal cancer treatment: a population-based analysis. J Oncol Pract. 2012;8:e69-e79.
17. Ehdaie B. Atoria CL, Lowrance WT, et al. Adherence to surveillance guidelines after radical cystectomy: a population-based analysis. Urol Oncol. 2014;32:779-784.
18. Travis LB, Fosså SD, Schonfeld SJ, et al. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst. 2005;97:1354-1365.
19. Donin N, Filson C, Drakaki A, et al. Risk of second primary malignancies among cancer survivors in the United States, 1992 through 2008. Cancer. 2016;122:3075-3086.
20. Holder AE, Ramirez AG, Gallion K. Depressive symptoms in Latina breast cancer survivors: a barrier to cancer screening. Health Psycholog. 2014;33:242-248.
21. Dyer G, Larsen SR, Gilroy N, et al. Adherence to cancer screening guidelines in Australian survivors of allogenic blood and marrow transplantation (BMT). Cancer Med. 2016;5:1702-1716.
22. Mandelzweig L, Chetrit A, Amitai T, et al. Primary prevention and screening practices among long-term breast cancer survivors. Cancer Causes Control. 2017;28:657-666.
23. Bishop MM, Lee SJ, Beaumont JL, et al. The preventive health behaviors of long-term survivors of cancer and hematopoietic stem cell transplantation compared with matched controls. Biol Blood Marrow Transplant. 2010;16:207-214.
24. Uhlig A, Mei J, Baik I, et al. Screening utilization among cancer survivors: a meta-analysis. J Public Health (Oxf). 2018;40:129-137.
25. Hilal T, Rudy DW. Radiation-induced breast cancer: the question of early breast cancer screening in Hodgkin’s lymphoma survivors. Oxf Med Case Reports. 2016;2016:17-18.
26. Lin K, Croswell JM, Koenig H, et al. Prostate-specific antigen-based screening for prostate cancer: an evidence update for the U.S. Preventive Services Task Force [Internet]. Evidence Syntheses No. 90. AHRQ Publication No. 12-05160-EF-1. Rockville, MD: Agency for Healthcare Research and Quality (US); October 2011. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0032900/. Accessed July 25, 2018.
27. US Preventive Services Task Force. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
28. Hofman M, Ryan JL, Figueroa-Moseley CD, et al. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12 Suppl 1:4-10.
29. Jung JY, Lee JM, Kim MS, et al. Comparison of fatigue, depression, and anxiety as factors affecting posttreatment health-related quality of life in lung cancer survivors. Psychooncology. 2018;27:465-470.
30. Bower JE. Cancer-related fatigue--mechanisms, risk factors, and treatment. Nat Rev Clin Oncol. 2014;11:597-609.
31. Medysky ME, Temesi J, Culos-Reed SN, et al. Exercise, sleep and cancer-related fatigue: are they related? Neurophysiol Clin. 2017;47:111-122.
32. Oh HS, Sea WS. Systematic review and meta-analysis of the correlates of cancer-related fatigue. Worldviews Evid Based Nurs. 2011;8:191-201.
33. Qu D, Zhang Z, Yu X, et al. Psychotropic drugs for the management of cancer-related fatigue: a systematic review and meta-analysis. Eur J Cancer Care (Engl). 2016;25:970-979.
34. Escalante CP, Manzullo EF. Cancer-related fatigue: the approach and treatment. J Gen Intern Med. 2009;24(suppl 2):S412-S416.
35. Mustian KM, Alfano CM, Heckler C, et al. Comparison of pharmaceutical, psychological, and exercise treatments for cancer-related fatigue: a meta-analysis. JAMA Oncol. 2017;3:961-968.
36. Glare PA, Davies PS, Finlay E, et al. Pain in cancer survivors. J Clin Oncol. 2014;32:1739-1747.
37. Davies PS. Chronic pain management in the cancer survivor: tips for primary care providers. Nurse Pract. 2013;39:28-38.
38. Boland EG, Ahmedzai SH. Persistent pain in cancer survivors. Curr Opin Support Palliat Care. 2017;11:181-190.
39. Sears CS, Robinson JW, Walker LM. A comprehensive review of sexual health concerns after cancer treatment and the biopsychosocial treatment options available to female patients. Eur J Cancer Care (Engl). 2017;27:e12738.
40. Schou Bredal I, Smeby NA, Ottesen S, et al. Chronic pain in breast cancer survivors: comparison of psychological, surgical, and medical characteristics between survivors with and without pain. J Pain Symptom Manage. 2014;48:852-862.
41. Sutradhar R, Lokku A, Barbera L. Cancer survivorship and opioid prescribing rates: a population-based matched cohort study among individuals with and without a history of cancer. Cancer. 2017;123:4286-4293.
42. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain - United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
43. Davis MP, Mehta Z. Opioids and chronic pain: where is the balance? Curr Oncol Rep. 2016;18:71.
44. Von Ah D. Cognitive changes associated with cancer and cancer treatment: state of the science. Clin J Oncol Nurs. 2015;19:47-56.
45. Moore HC. An overview of chemotherapy-related cognitive dysfunction, or ‘chemobrain’. Oncology (Williston Park). 2014;28:797-804.
46. Asher A. Cognitive dysfunction among cancer survivors. Am J Phys Med Rehabil. 2011;90(suppl):S16-S26.
47. Joly F, Rigal O, Noal S, et al. Cognitive dysfunction and cancer: which consequences in terms of disease management? Psychooncology. 2011;20:1251-1258.
48. Attention, thinking or memory problems. American Society of Clinical Oncology Cancer.Net. April 2018. www.cancer.net/navigating-cancer-care/side-effects/attention-thinking-or-memory-problems. Accessed July 25, 2018.
49. Kucherer S, Ferguson RJ. Cognitive behavioral therapy for cancer-related cognitive dysfunction. Curr Opin Support Palliat Care. 2017;11:46-51.
50. Derry HM, Jaremka LM, Bennet JM, et al. Yoga and self-reported cognitive problems in breast cancer survivors: a randomized controlled trial. Psychooncology. 2015;24:958-966.
51. Treanor CJ, McMenamin UC, O’Neill RF, et al. Non-pharmacological interventions for cognitive impairment due to systemic cancer treatment. Cochrane Database Syst Rev. 2016 Aug 16;(8):CD011325.
52. Mitchell AJ, Ferguson DW, Gill J, et al. Depression and anxiety in long-term cancer survivors compared with spouses and healthy controls: a systematic review and meta-analysis. Lancet Oncol. 2013;14:721-732.
53. Inhestern L, Beierlein V, Bultmann JC, et al. Anxiety and depression in working-age cancer survivors: a register-based study. BMC Cancer. 2017;17:347.
54. Partridge AH, Jacobsen PB, Andersen BL. Challenges to standardizing the care for adult cancer survivors: highlighting ASCO’s fatigue and anxiety and depression guidelines. Am Soc Clin Oncol Educ Book. 2015:188-194.
55. Andersen BL, Yang HC, Farrar WB, et al. Psychologic intervention improves survival for breast cancer patients: a randomized clinical trial. Cancer. 2008;113:3450-3458.
56. Yi JC, Syrjala KL. Anxiety and depression in cancer survivors. Med Clin N Am. 2017;101:1099-1113.
57. Zhu G, Zhang X, Wang Y, et al. Effects of exercise intervention in breast cancer survivors: a meta-analysis of 33 randomized controlled trials. Onco Targets Ther. 2016;9:2153-2168.
58. Hawkins NA, Soman A, Lunsford N, et al. Use of medications for treating anxiety and depression in cancer survivors in the United States. J Clin Oncol. 2017;35:78-85.
59. Ostuzzi G, Matcham F, Dauchy S, et al. Antidepressants for the treatment of depression in people with cancer. Cochrane Database Syst Rev. 2015 June 1;(6):CD011006.
60. Laoutidis ZG, Mathiak K. Antidepressants in the treatment of depression/depressive symptoms in cancer patients: a systematic review and meta-analysis. BMC Psychiatry. 2013;13:140.
61. Lydiatt WM, Bessette D, Schmid KK, et al. Prevention of depression with escitalopram in patients undergoing treatment for head and neck cancer: randomized, double-blind, placebo-controlled clinical trial. JAMA Otolaryngol Head Neck Surg. 2013;139:678-686.
62. Lavigne JE, Heckler C, Mathews JL, et al. A randomized, controlled, double-blinded clinical trial of gabapentin 300 versus 900 mg versus placebo for anxiety symptoms in breast cancer survivors. Breast Cancer Res Treat. 2012;136:479-486.
63. Okwuosa TM, Anzevino S, Rao R. Cardiovascular disease in cancer survivors. Postgrad Med J. 2017;93:82-90.
64. Plana, JC, Galderisi M, Barac A, et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2014;27:911-939.
65. Lancellotti, P, Nkomo VT, Badano LP, et al. Expert consensus for multi-modality imaging evaluation of cardiovascular complications of radiotherapy in adults: a report from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:1013-1032.
1. American Cancer Society. Cancer Treatment & Survivorship Facts & Figures 2016-2017. Atlanta, GA: American Cancer Society; 2016. www.cancer.org/research/cancer-facts-statistics/survivor-facts-figures.html. Accessed July 25, 2018.
2. Survivorship. NCCN Guidelines (version 1.2017). Fort Washington, PA: National Comprehensive Cancer Network; 2017. www.nccn.org/professionals/physician_gls/default.aspx#supportive. Accessed July 26, 2018.
3. Kendall C, Decker KM, Groome PA, et al. Use of physician services during the survivorship phase: a multi-province study of women diagnosed with breast cancer. Curr Oncolog. 2017;24:81-89.
4. Snyder CF, Earle CC, Herbert RJ, et al. Preventive care for colorectal cancer survivors: a 5-year longitudinal study. J Clin Oncol. 2008;26:1073-1079.
5. Hewitt M, Greenfield S, Stovall E (eds); Institute of Medicine and National Research Council. From Cancer Patient to Cancer Survivor: Lost in Transition. Washington DC: The National Academies Press; 2006. www.nap.edu/read/11468/chapter/1. Accessed July 25, 2018.
6. Survivorship care plan. New York, NY: Memorial Sloan Kettering Cancer Center. www.mskcc.org/hcp-education-training/survivorship/survivorship-care-plan. Accessed August 11, 2018.
7. Fuentes AC, Lambird JE, George TJ, et al. Cancer survivor’s history and physical. South Med J. 2017;110:37-44. http://sma.org/southern-medical-journal/article/cancer-survivors-history-physical/. Accessed July 26, 2018.
8. Salz T, Oeffinger KC, McCabe MS, et al. Survivorship care plans in research and practice. CA Cancer J Clin. 2012;62:101-117.
9. Birken SA, Mayer DK, Weiner BJ. Survivorship care plans: prevalence and barriers to use. J Cancer Educ. 2013;28:290-296.
10. American College of Surgeons Commission on Cancer. Cancer program standards 2012: Ensuring patient-centered care. V1.2.1. www.facs.org/~/media/files/quality%20programs/cancer/coc/programstandards2012.ashx. Accessed July 25, 2018.
11. NCCN guidelines for treatment of cancer by site. NCCN Guidelines (version 1.2018). Fort Washington, PA: National Comprehensive Cancer Network; 2018. www.nccn.org/professionals/physician_gls/default.aspx#site. Accessed July 25, 2018.
12. Spronka I, Korevaar JC, Burgers JS, et al. Review of guidance on recurrence risk management for general practitioners in breast cancer, colorectal cancer and melanoma guidelines. Family Pract. 2017;34:154-160.
13. Merkow RP, Korenstein D, Yeahia R, et al. Quality of cancer surveillance clinical practice guidelines: specificity and consistency of recommendations. JAMA Intern Med. 2017;177:701-709.
14. Muradali D, Kennedy EB, Eisen A, et al. Breast screening for survivors of breast cancer: a systematic review. Prev Med. 2017;103:70-75.
15. Giannarini G, Kessler TM, Thoeny HC, et al. Do patients benefit from routine follow-up to detect recurrences after radical cystectomy and ileal orthotopic bladder substitution? Eur Urol. 2010;58:486-494.
16. Sisler JJ, Seo B, Katz A, et al. Concordance with ASCO guidelines for surveillance after colorectal cancer treatment: a population-based analysis. J Oncol Pract. 2012;8:e69-e79.
17. Ehdaie B. Atoria CL, Lowrance WT, et al. Adherence to surveillance guidelines after radical cystectomy: a population-based analysis. Urol Oncol. 2014;32:779-784.
18. Travis LB, Fosså SD, Schonfeld SJ, et al. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst. 2005;97:1354-1365.
19. Donin N, Filson C, Drakaki A, et al. Risk of second primary malignancies among cancer survivors in the United States, 1992 through 2008. Cancer. 2016;122:3075-3086.
20. Holder AE, Ramirez AG, Gallion K. Depressive symptoms in Latina breast cancer survivors: a barrier to cancer screening. Health Psycholog. 2014;33:242-248.
21. Dyer G, Larsen SR, Gilroy N, et al. Adherence to cancer screening guidelines in Australian survivors of allogenic blood and marrow transplantation (BMT). Cancer Med. 2016;5:1702-1716.
22. Mandelzweig L, Chetrit A, Amitai T, et al. Primary prevention and screening practices among long-term breast cancer survivors. Cancer Causes Control. 2017;28:657-666.
23. Bishop MM, Lee SJ, Beaumont JL, et al. The preventive health behaviors of long-term survivors of cancer and hematopoietic stem cell transplantation compared with matched controls. Biol Blood Marrow Transplant. 2010;16:207-214.
24. Uhlig A, Mei J, Baik I, et al. Screening utilization among cancer survivors: a meta-analysis. J Public Health (Oxf). 2018;40:129-137.
25. Hilal T, Rudy DW. Radiation-induced breast cancer: the question of early breast cancer screening in Hodgkin’s lymphoma survivors. Oxf Med Case Reports. 2016;2016:17-18.
26. Lin K, Croswell JM, Koenig H, et al. Prostate-specific antigen-based screening for prostate cancer: an evidence update for the U.S. Preventive Services Task Force [Internet]. Evidence Syntheses No. 90. AHRQ Publication No. 12-05160-EF-1. Rockville, MD: Agency for Healthcare Research and Quality (US); October 2011. www.ncbi.nlm.nih.gov/pubmedhealth/PMH0032900/. Accessed July 25, 2018.
27. US Preventive Services Task Force. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. JAMA. 2018;320:674-686.
28. Hofman M, Ryan JL, Figueroa-Moseley CD, et al. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12 Suppl 1:4-10.
29. Jung JY, Lee JM, Kim MS, et al. Comparison of fatigue, depression, and anxiety as factors affecting posttreatment health-related quality of life in lung cancer survivors. Psychooncology. 2018;27:465-470.
30. Bower JE. Cancer-related fatigue--mechanisms, risk factors, and treatment. Nat Rev Clin Oncol. 2014;11:597-609.
31. Medysky ME, Temesi J, Culos-Reed SN, et al. Exercise, sleep and cancer-related fatigue: are they related? Neurophysiol Clin. 2017;47:111-122.
32. Oh HS, Sea WS. Systematic review and meta-analysis of the correlates of cancer-related fatigue. Worldviews Evid Based Nurs. 2011;8:191-201.
33. Qu D, Zhang Z, Yu X, et al. Psychotropic drugs for the management of cancer-related fatigue: a systematic review and meta-analysis. Eur J Cancer Care (Engl). 2016;25:970-979.
34. Escalante CP, Manzullo EF. Cancer-related fatigue: the approach and treatment. J Gen Intern Med. 2009;24(suppl 2):S412-S416.
35. Mustian KM, Alfano CM, Heckler C, et al. Comparison of pharmaceutical, psychological, and exercise treatments for cancer-related fatigue: a meta-analysis. JAMA Oncol. 2017;3:961-968.
36. Glare PA, Davies PS, Finlay E, et al. Pain in cancer survivors. J Clin Oncol. 2014;32:1739-1747.
37. Davies PS. Chronic pain management in the cancer survivor: tips for primary care providers. Nurse Pract. 2013;39:28-38.
38. Boland EG, Ahmedzai SH. Persistent pain in cancer survivors. Curr Opin Support Palliat Care. 2017;11:181-190.
39. Sears CS, Robinson JW, Walker LM. A comprehensive review of sexual health concerns after cancer treatment and the biopsychosocial treatment options available to female patients. Eur J Cancer Care (Engl). 2017;27:e12738.
40. Schou Bredal I, Smeby NA, Ottesen S, et al. Chronic pain in breast cancer survivors: comparison of psychological, surgical, and medical characteristics between survivors with and without pain. J Pain Symptom Manage. 2014;48:852-862.
41. Sutradhar R, Lokku A, Barbera L. Cancer survivorship and opioid prescribing rates: a population-based matched cohort study among individuals with and without a history of cancer. Cancer. 2017;123:4286-4293.
42. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain - United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
43. Davis MP, Mehta Z. Opioids and chronic pain: where is the balance? Curr Oncol Rep. 2016;18:71.
44. Von Ah D. Cognitive changes associated with cancer and cancer treatment: state of the science. Clin J Oncol Nurs. 2015;19:47-56.
45. Moore HC. An overview of chemotherapy-related cognitive dysfunction, or ‘chemobrain’. Oncology (Williston Park). 2014;28:797-804.
46. Asher A. Cognitive dysfunction among cancer survivors. Am J Phys Med Rehabil. 2011;90(suppl):S16-S26.
47. Joly F, Rigal O, Noal S, et al. Cognitive dysfunction and cancer: which consequences in terms of disease management? Psychooncology. 2011;20:1251-1258.
48. Attention, thinking or memory problems. American Society of Clinical Oncology Cancer.Net. April 2018. www.cancer.net/navigating-cancer-care/side-effects/attention-thinking-or-memory-problems. Accessed July 25, 2018.
49. Kucherer S, Ferguson RJ. Cognitive behavioral therapy for cancer-related cognitive dysfunction. Curr Opin Support Palliat Care. 2017;11:46-51.
50. Derry HM, Jaremka LM, Bennet JM, et al. Yoga and self-reported cognitive problems in breast cancer survivors: a randomized controlled trial. Psychooncology. 2015;24:958-966.
51. Treanor CJ, McMenamin UC, O’Neill RF, et al. Non-pharmacological interventions for cognitive impairment due to systemic cancer treatment. Cochrane Database Syst Rev. 2016 Aug 16;(8):CD011325.
52. Mitchell AJ, Ferguson DW, Gill J, et al. Depression and anxiety in long-term cancer survivors compared with spouses and healthy controls: a systematic review and meta-analysis. Lancet Oncol. 2013;14:721-732.
53. Inhestern L, Beierlein V, Bultmann JC, et al. Anxiety and depression in working-age cancer survivors: a register-based study. BMC Cancer. 2017;17:347.
54. Partridge AH, Jacobsen PB, Andersen BL. Challenges to standardizing the care for adult cancer survivors: highlighting ASCO’s fatigue and anxiety and depression guidelines. Am Soc Clin Oncol Educ Book. 2015:188-194.
55. Andersen BL, Yang HC, Farrar WB, et al. Psychologic intervention improves survival for breast cancer patients: a randomized clinical trial. Cancer. 2008;113:3450-3458.
56. Yi JC, Syrjala KL. Anxiety and depression in cancer survivors. Med Clin N Am. 2017;101:1099-1113.
57. Zhu G, Zhang X, Wang Y, et al. Effects of exercise intervention in breast cancer survivors: a meta-analysis of 33 randomized controlled trials. Onco Targets Ther. 2016;9:2153-2168.
58. Hawkins NA, Soman A, Lunsford N, et al. Use of medications for treating anxiety and depression in cancer survivors in the United States. J Clin Oncol. 2017;35:78-85.
59. Ostuzzi G, Matcham F, Dauchy S, et al. Antidepressants for the treatment of depression in people with cancer. Cochrane Database Syst Rev. 2015 June 1;(6):CD011006.
60. Laoutidis ZG, Mathiak K. Antidepressants in the treatment of depression/depressive symptoms in cancer patients: a systematic review and meta-analysis. BMC Psychiatry. 2013;13:140.
61. Lydiatt WM, Bessette D, Schmid KK, et al. Prevention of depression with escitalopram in patients undergoing treatment for head and neck cancer: randomized, double-blind, placebo-controlled clinical trial. JAMA Otolaryngol Head Neck Surg. 2013;139:678-686.
62. Lavigne JE, Heckler C, Mathews JL, et al. A randomized, controlled, double-blinded clinical trial of gabapentin 300 versus 900 mg versus placebo for anxiety symptoms in breast cancer survivors. Breast Cancer Res Treat. 2012;136:479-486.
63. Okwuosa TM, Anzevino S, Rao R. Cardiovascular disease in cancer survivors. Postgrad Med J. 2017;93:82-90.
64. Plana, JC, Galderisi M, Barac A, et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2014;27:911-939.
65. Lancellotti, P, Nkomo VT, Badano LP, et al. Expert consensus for multi-modality imaging evaluation of cardiovascular complications of radiotherapy in adults: a report from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26:1013-1032.
PRACTICE RECOMMENDATIONS
› Provide normal age-related cancer screening for cancer survivors because of their high risk of a second cancer. B
› Strongly encourage lifestyle changes for cancer survivors, especially smoking cessation. B
› Recommend exercise, which alleviates pain, depression, anxiety, and (more effectively than any other intervention) fatigue, for cancer survivors. B
› Remain vigilant for the development in cancer survivors of cardiovascular disease, including heart failure, which can appear long after therapy. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
*Cancer survivor care in the pediatric patients, including application of a survivorship care plan (also discussed later in this article), is reviewed in “Partnering to optimize care of childhood cancer survivors,” The Journal of Family Practice, April 2017.
Antithrombotic strategy 1 year after stenting in AF patients leans toward oral anticoagulant alone
SAN DIEGO – In patients with atrial fibrillation and stable coronary artery disease, a randomized trial of oral anticoagulation alone versus an anticoagulant plus a single antiplatelet agent failed to establish noninferiority of the single-agent approach. The trial could not demonstrate its primary endpoint of all-cause death, myocardial infarction, stroke, or systemic embolism.
But a secondary endpoint that included major bleeding did demonstrate equivalence, leading the researchers to suggest that oral anticoagulation (OAC) alone may be sufficient in most patients.
“Combined OAC and single antiplatelet therapy is unlikely to provide net clinical benefit over OAC alone. Thus, OAC alone might be reasonable for AF [atrial fibrillation] patients beyond 1 year after coronary stenting,” Yukiko Nakano, MD, of Kyoto (Japan) University Graduate School of Medicine, said during a press conference at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation. The report was simultaneously published Sept. 24 in Circulation (doi: 10.1161/CIRCULATIONAHA.118.036768).
The results support the European Society of Cardiology practice guidelines, which recommend lifelong OAC without antiplatelet therapy. But physicians often continue to prescribe antiplatelet agents out of concern that stent thrombosis could occur if the therapy is stopped.
The study was stopped prematurely because of insufficient recruitment, which may have contributed to the failed primary endpoint. It’s a shortcoming that befalls many such studies, perhaps because cardiologists tend to be set in their ways when it comes to treatment of patients after a stent implant. “Cardiologists just think they know the answer, and they don’t want to expose their patients (to a clinical trial). They say, ‘I have my patients on whatever regimen. It seems to be working, and they’re not bleeding, so I don’t want to change it.’ This study suggests that we probably can stop one of the two (antiplatelet drugs) and get by with a single agent, and in this case they got by with no agent (in the monotherapy arm),” said C. Michael Gibson, MD, chief of clinical research in the division of cardiology at Beth Israel Deaconess Medical Center, Boston, who was a discussant at the press conference.
The study recruited 696 patients who were receiving OAC plus single antiplatelet therapy (SAPT) 1 year after receiving a stent. They were randomized 1:1 to continue combined therapy or to stop SAPT and then followed for a median of 2.5 years. A total of 74% of patients who received OAC alone were taking warfarin, while 26% were taking a direct oral anticoagulant. The SAPT group took aspirin or clopidogrel.
Overall, 15.7% of OAC patients experienced the primary endpoint, compared with 13.6% of the combined group (noninferiority P = .20). None of the individual components of the primary endpoint were statistically significantly different between the groups. International Society on Thrombosis and Haemostasis major bleeding and Thrombolysis in Myocardial Infarction major bleeding trended in favor of OAC alone. The secondary endpoint (primary endpoint plus major bleeding) achieved noninferiority, occurring in 19.5% of the OAC group and 19.4% of the combined therapy group (noninferiority P = .016; superiority P = .96).
Daiichi-Sankyo funded the trial. Dr. Nakano had no conflicts of interest. Dr. Gibson reported numerous financial ties to pharmaceutical companies, including Daiichi-Sankyo.
SAN DIEGO – In patients with atrial fibrillation and stable coronary artery disease, a randomized trial of oral anticoagulation alone versus an anticoagulant plus a single antiplatelet agent failed to establish noninferiority of the single-agent approach. The trial could not demonstrate its primary endpoint of all-cause death, myocardial infarction, stroke, or systemic embolism.
But a secondary endpoint that included major bleeding did demonstrate equivalence, leading the researchers to suggest that oral anticoagulation (OAC) alone may be sufficient in most patients.
“Combined OAC and single antiplatelet therapy is unlikely to provide net clinical benefit over OAC alone. Thus, OAC alone might be reasonable for AF [atrial fibrillation] patients beyond 1 year after coronary stenting,” Yukiko Nakano, MD, of Kyoto (Japan) University Graduate School of Medicine, said during a press conference at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation. The report was simultaneously published Sept. 24 in Circulation (doi: 10.1161/CIRCULATIONAHA.118.036768).
The results support the European Society of Cardiology practice guidelines, which recommend lifelong OAC without antiplatelet therapy. But physicians often continue to prescribe antiplatelet agents out of concern that stent thrombosis could occur if the therapy is stopped.
The study was stopped prematurely because of insufficient recruitment, which may have contributed to the failed primary endpoint. It’s a shortcoming that befalls many such studies, perhaps because cardiologists tend to be set in their ways when it comes to treatment of patients after a stent implant. “Cardiologists just think they know the answer, and they don’t want to expose their patients (to a clinical trial). They say, ‘I have my patients on whatever regimen. It seems to be working, and they’re not bleeding, so I don’t want to change it.’ This study suggests that we probably can stop one of the two (antiplatelet drugs) and get by with a single agent, and in this case they got by with no agent (in the monotherapy arm),” said C. Michael Gibson, MD, chief of clinical research in the division of cardiology at Beth Israel Deaconess Medical Center, Boston, who was a discussant at the press conference.
The study recruited 696 patients who were receiving OAC plus single antiplatelet therapy (SAPT) 1 year after receiving a stent. They were randomized 1:1 to continue combined therapy or to stop SAPT and then followed for a median of 2.5 years. A total of 74% of patients who received OAC alone were taking warfarin, while 26% were taking a direct oral anticoagulant. The SAPT group took aspirin or clopidogrel.
Overall, 15.7% of OAC patients experienced the primary endpoint, compared with 13.6% of the combined group (noninferiority P = .20). None of the individual components of the primary endpoint were statistically significantly different between the groups. International Society on Thrombosis and Haemostasis major bleeding and Thrombolysis in Myocardial Infarction major bleeding trended in favor of OAC alone. The secondary endpoint (primary endpoint plus major bleeding) achieved noninferiority, occurring in 19.5% of the OAC group and 19.4% of the combined therapy group (noninferiority P = .016; superiority P = .96).
Daiichi-Sankyo funded the trial. Dr. Nakano had no conflicts of interest. Dr. Gibson reported numerous financial ties to pharmaceutical companies, including Daiichi-Sankyo.
SAN DIEGO – In patients with atrial fibrillation and stable coronary artery disease, a randomized trial of oral anticoagulation alone versus an anticoagulant plus a single antiplatelet agent failed to establish noninferiority of the single-agent approach. The trial could not demonstrate its primary endpoint of all-cause death, myocardial infarction, stroke, or systemic embolism.
But a secondary endpoint that included major bleeding did demonstrate equivalence, leading the researchers to suggest that oral anticoagulation (OAC) alone may be sufficient in most patients.
“Combined OAC and single antiplatelet therapy is unlikely to provide net clinical benefit over OAC alone. Thus, OAC alone might be reasonable for AF [atrial fibrillation] patients beyond 1 year after coronary stenting,” Yukiko Nakano, MD, of Kyoto (Japan) University Graduate School of Medicine, said during a press conference at the Transcatheter Cardiovascular Therapeutics annual meeting, sponsored by the Cardiovascular Research Foundation. The report was simultaneously published Sept. 24 in Circulation (doi: 10.1161/CIRCULATIONAHA.118.036768).
The results support the European Society of Cardiology practice guidelines, which recommend lifelong OAC without antiplatelet therapy. But physicians often continue to prescribe antiplatelet agents out of concern that stent thrombosis could occur if the therapy is stopped.
The study was stopped prematurely because of insufficient recruitment, which may have contributed to the failed primary endpoint. It’s a shortcoming that befalls many such studies, perhaps because cardiologists tend to be set in their ways when it comes to treatment of patients after a stent implant. “Cardiologists just think they know the answer, and they don’t want to expose their patients (to a clinical trial). They say, ‘I have my patients on whatever regimen. It seems to be working, and they’re not bleeding, so I don’t want to change it.’ This study suggests that we probably can stop one of the two (antiplatelet drugs) and get by with a single agent, and in this case they got by with no agent (in the monotherapy arm),” said C. Michael Gibson, MD, chief of clinical research in the division of cardiology at Beth Israel Deaconess Medical Center, Boston, who was a discussant at the press conference.
The study recruited 696 patients who were receiving OAC plus single antiplatelet therapy (SAPT) 1 year after receiving a stent. They were randomized 1:1 to continue combined therapy or to stop SAPT and then followed for a median of 2.5 years. A total of 74% of patients who received OAC alone were taking warfarin, while 26% were taking a direct oral anticoagulant. The SAPT group took aspirin or clopidogrel.
Overall, 15.7% of OAC patients experienced the primary endpoint, compared with 13.6% of the combined group (noninferiority P = .20). None of the individual components of the primary endpoint were statistically significantly different between the groups. International Society on Thrombosis and Haemostasis major bleeding and Thrombolysis in Myocardial Infarction major bleeding trended in favor of OAC alone. The secondary endpoint (primary endpoint plus major bleeding) achieved noninferiority, occurring in 19.5% of the OAC group and 19.4% of the combined therapy group (noninferiority P = .016; superiority P = .96).
Daiichi-Sankyo funded the trial. Dr. Nakano had no conflicts of interest. Dr. Gibson reported numerous financial ties to pharmaceutical companies, including Daiichi-Sankyo.
REPORTING FROM TCT 2018
Key clinical point:
Major finding: A measure that included cardiac events plus major bleeding showed an oral anticoagulant alone was noninferior to an OAC plus antiplatelet therapy.
Study details: Randomized, controlled trial of 696 patients.
Disclosures: Daiichi-Sankyo funded the trial. Dr. Nakano had no conflicts of interest. Dr. Gibson reported numerous financial ties to pharmaceutical companies, including Daiichi-Sankyo.
Coagulopathy outbreak underscores danger of synthetic cannabinoids
Synthetic cannabinoids laced with superwarfarin were behind a recent outbreak of severe coagulopathy in Illinois.
In most cases, vitamin K replacement therapy alleviated symptoms, but four patients died after developing intracranial bleeding, said Amar H. Kelkar, MD, of the University of Illinois at Peoria.
Experts continue to look for how and why superwarfarin ended up in synthetic cannabinoids, whose street names include spice and K2, wrote Dr. Kelkar and his associates. Their report is in the New England Journal of Medicine.
Starting in March 2018, more than 150 patients across Illinois presented to hospitals with bleeding diathesis that involved persistent coagulopathy, the investigators explained. Early inquiries revealed that patients had consumed synthetic cannabinoids. Serum tests identified vitamin K antagonists, including brodifacoum, bromadiolone, and difenacoum. During arrests of suspected distributors, police confiscated synthetic cannabinoids that also tested positive for brodifacoum.
To help characterize the outbreak, the investigators reviewed admissions to Saint Francis Medical Center in Peoria, Ill., between March 28 and April 21, 2018. They identified 34 cases in which patients with vitamin K–dependent factor coagulopathy reported recent exposure to synthetic cannabinoids.
Fifteen of these patients underwent confirmatory anticoagulant testing, which universally confirmed superwarfarin poisoning. Brodifacoum was detected in all patients, difenacoum in five patients, bromadiolone in two patients, and warfarin in one patient.
Common presenting symptoms included gross hematuria (56% of patients) and abdominal pain (47%). Computed tomography identified renal abnormalities in 12 patients.
All patients received oral vitamin K1 (phytonadione). Red cell transfusions, fresh-frozen plasma infusions, and 4-factor prothrombin complex concentrate, or a combination of these treatments, were also used in some patients.
Among the four confirmed deaths in this outbreak, one occurred in a patient in this case series. The patient, a 37-year-old woman presenting to the emergency department with markedly reduced consciousness, was reported by her friends to have recently used synthetic cannabinoids and methamphetamine. She had no personal or family history of coagulopathy.
Computed tomography of the head without contrast material revealed severe acute intraparenchymal hemorrhage of the right basal nuclei and insula with intraventricular extension, a 10-mm left-sided midline shift, and herniation.
She met criteria for brain death 15 hours after hospital admission despite treatment with 10 mg of intravenous vitamin K1, four units of fresh frozen plasma, and 2,300 units of Kcentra.
Treating these patients after hospital discharge was difficult because of a lack of consensus guidelines and access to follow-up care, Dr. Kelkar and his associates noted. Some patients were quoted $24,000 to $34,000 per month for oral vitamin K1 therapy, which also made caring for them difficult and highlighted the need for confirmatory laboratory testing of suspected cases of superwarfarin poisoning.
Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
SOURCE: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
Treating patients who are exposed to synthetic cannabinoid and a superwarfarin such as brodifacoum “requires more than the usual ‘treat ’em and street ’em’ approach,” wrote Jean M. Connors, MD.
“Brodifacoum is a successful rodenticide because of its extremely long half-life (approximately 16-36 days in humans),” Dr. Connors noted.
The drug also is lipophilic, causing tissue sequestration. Once exposed, patients often develop coagulopathy lasting 9 months or longer, she said.
Compared with warfarin poisoning, brodifacoum therefore requires substantially higher-dose and longer-term vitamin K1 therapy. Among the patients in this case series, the maximum outpatient dose was 50 mg, three times daily, and one patient was prescribed 25 mg, twice daily for 270 days, Dr. Connors noted.
“[Dr. Kelkar and his associates] highlight the resources and coordination needed for dealing with a public health crisis that has a prolonged duration of effect,” she added. “Because the synthetic cannabinoid market is lucrative, new products with new toxicity profiles are likely to crop up.”
Dr. Connors is with Brigham and Women’s Hospital, Dana-Farber Cancer Institute, and Harvard Medical School, all in Boston. She reported personal fees from Bristol-Myers Squibb, Portola, Dova Pharmaceuticals, and Unum Therapeutics outside the submitted work. These comments are from her accompanying editorial (N Engl J Med. 2018;379:1275-7).
Treating patients who are exposed to synthetic cannabinoid and a superwarfarin such as brodifacoum “requires more than the usual ‘treat ’em and street ’em’ approach,” wrote Jean M. Connors, MD.
“Brodifacoum is a successful rodenticide because of its extremely long half-life (approximately 16-36 days in humans),” Dr. Connors noted.
The drug also is lipophilic, causing tissue sequestration. Once exposed, patients often develop coagulopathy lasting 9 months or longer, she said.
Compared with warfarin poisoning, brodifacoum therefore requires substantially higher-dose and longer-term vitamin K1 therapy. Among the patients in this case series, the maximum outpatient dose was 50 mg, three times daily, and one patient was prescribed 25 mg, twice daily for 270 days, Dr. Connors noted.
“[Dr. Kelkar and his associates] highlight the resources and coordination needed for dealing with a public health crisis that has a prolonged duration of effect,” she added. “Because the synthetic cannabinoid market is lucrative, new products with new toxicity profiles are likely to crop up.”
Dr. Connors is with Brigham and Women’s Hospital, Dana-Farber Cancer Institute, and Harvard Medical School, all in Boston. She reported personal fees from Bristol-Myers Squibb, Portola, Dova Pharmaceuticals, and Unum Therapeutics outside the submitted work. These comments are from her accompanying editorial (N Engl J Med. 2018;379:1275-7).
Treating patients who are exposed to synthetic cannabinoid and a superwarfarin such as brodifacoum “requires more than the usual ‘treat ’em and street ’em’ approach,” wrote Jean M. Connors, MD.
“Brodifacoum is a successful rodenticide because of its extremely long half-life (approximately 16-36 days in humans),” Dr. Connors noted.
The drug also is lipophilic, causing tissue sequestration. Once exposed, patients often develop coagulopathy lasting 9 months or longer, she said.
Compared with warfarin poisoning, brodifacoum therefore requires substantially higher-dose and longer-term vitamin K1 therapy. Among the patients in this case series, the maximum outpatient dose was 50 mg, three times daily, and one patient was prescribed 25 mg, twice daily for 270 days, Dr. Connors noted.
“[Dr. Kelkar and his associates] highlight the resources and coordination needed for dealing with a public health crisis that has a prolonged duration of effect,” she added. “Because the synthetic cannabinoid market is lucrative, new products with new toxicity profiles are likely to crop up.”
Dr. Connors is with Brigham and Women’s Hospital, Dana-Farber Cancer Institute, and Harvard Medical School, all in Boston. She reported personal fees from Bristol-Myers Squibb, Portola, Dova Pharmaceuticals, and Unum Therapeutics outside the submitted work. These comments are from her accompanying editorial (N Engl J Med. 2018;379:1275-7).
Synthetic cannabinoids laced with superwarfarin were behind a recent outbreak of severe coagulopathy in Illinois.
In most cases, vitamin K replacement therapy alleviated symptoms, but four patients died after developing intracranial bleeding, said Amar H. Kelkar, MD, of the University of Illinois at Peoria.
Experts continue to look for how and why superwarfarin ended up in synthetic cannabinoids, whose street names include spice and K2, wrote Dr. Kelkar and his associates. Their report is in the New England Journal of Medicine.
Starting in March 2018, more than 150 patients across Illinois presented to hospitals with bleeding diathesis that involved persistent coagulopathy, the investigators explained. Early inquiries revealed that patients had consumed synthetic cannabinoids. Serum tests identified vitamin K antagonists, including brodifacoum, bromadiolone, and difenacoum. During arrests of suspected distributors, police confiscated synthetic cannabinoids that also tested positive for brodifacoum.
To help characterize the outbreak, the investigators reviewed admissions to Saint Francis Medical Center in Peoria, Ill., between March 28 and April 21, 2018. They identified 34 cases in which patients with vitamin K–dependent factor coagulopathy reported recent exposure to synthetic cannabinoids.
Fifteen of these patients underwent confirmatory anticoagulant testing, which universally confirmed superwarfarin poisoning. Brodifacoum was detected in all patients, difenacoum in five patients, bromadiolone in two patients, and warfarin in one patient.
Common presenting symptoms included gross hematuria (56% of patients) and abdominal pain (47%). Computed tomography identified renal abnormalities in 12 patients.
All patients received oral vitamin K1 (phytonadione). Red cell transfusions, fresh-frozen plasma infusions, and 4-factor prothrombin complex concentrate, or a combination of these treatments, were also used in some patients.
Among the four confirmed deaths in this outbreak, one occurred in a patient in this case series. The patient, a 37-year-old woman presenting to the emergency department with markedly reduced consciousness, was reported by her friends to have recently used synthetic cannabinoids and methamphetamine. She had no personal or family history of coagulopathy.
Computed tomography of the head without contrast material revealed severe acute intraparenchymal hemorrhage of the right basal nuclei and insula with intraventricular extension, a 10-mm left-sided midline shift, and herniation.
She met criteria for brain death 15 hours after hospital admission despite treatment with 10 mg of intravenous vitamin K1, four units of fresh frozen plasma, and 2,300 units of Kcentra.
Treating these patients after hospital discharge was difficult because of a lack of consensus guidelines and access to follow-up care, Dr. Kelkar and his associates noted. Some patients were quoted $24,000 to $34,000 per month for oral vitamin K1 therapy, which also made caring for them difficult and highlighted the need for confirmatory laboratory testing of suspected cases of superwarfarin poisoning.
Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
SOURCE: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
Synthetic cannabinoids laced with superwarfarin were behind a recent outbreak of severe coagulopathy in Illinois.
In most cases, vitamin K replacement therapy alleviated symptoms, but four patients died after developing intracranial bleeding, said Amar H. Kelkar, MD, of the University of Illinois at Peoria.
Experts continue to look for how and why superwarfarin ended up in synthetic cannabinoids, whose street names include spice and K2, wrote Dr. Kelkar and his associates. Their report is in the New England Journal of Medicine.
Starting in March 2018, more than 150 patients across Illinois presented to hospitals with bleeding diathesis that involved persistent coagulopathy, the investigators explained. Early inquiries revealed that patients had consumed synthetic cannabinoids. Serum tests identified vitamin K antagonists, including brodifacoum, bromadiolone, and difenacoum. During arrests of suspected distributors, police confiscated synthetic cannabinoids that also tested positive for brodifacoum.
To help characterize the outbreak, the investigators reviewed admissions to Saint Francis Medical Center in Peoria, Ill., between March 28 and April 21, 2018. They identified 34 cases in which patients with vitamin K–dependent factor coagulopathy reported recent exposure to synthetic cannabinoids.
Fifteen of these patients underwent confirmatory anticoagulant testing, which universally confirmed superwarfarin poisoning. Brodifacoum was detected in all patients, difenacoum in five patients, bromadiolone in two patients, and warfarin in one patient.
Common presenting symptoms included gross hematuria (56% of patients) and abdominal pain (47%). Computed tomography identified renal abnormalities in 12 patients.
All patients received oral vitamin K1 (phytonadione). Red cell transfusions, fresh-frozen plasma infusions, and 4-factor prothrombin complex concentrate, or a combination of these treatments, were also used in some patients.
Among the four confirmed deaths in this outbreak, one occurred in a patient in this case series. The patient, a 37-year-old woman presenting to the emergency department with markedly reduced consciousness, was reported by her friends to have recently used synthetic cannabinoids and methamphetamine. She had no personal or family history of coagulopathy.
Computed tomography of the head without contrast material revealed severe acute intraparenchymal hemorrhage of the right basal nuclei and insula with intraventricular extension, a 10-mm left-sided midline shift, and herniation.
She met criteria for brain death 15 hours after hospital admission despite treatment with 10 mg of intravenous vitamin K1, four units of fresh frozen plasma, and 2,300 units of Kcentra.
Treating these patients after hospital discharge was difficult because of a lack of consensus guidelines and access to follow-up care, Dr. Kelkar and his associates noted. Some patients were quoted $24,000 to $34,000 per month for oral vitamin K1 therapy, which also made caring for them difficult and highlighted the need for confirmatory laboratory testing of suspected cases of superwarfarin poisoning.
Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
SOURCE: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point:
Major finding: There were more than 150 cases in Illinois with four deaths among patients who developed spontaneous intracranial bleeding.
Study details: A single-institution case series of 15 patients.
Disclosures: Dr. Kelkar reported having no conflicts of interest. Two coinvestigators reported relationships outside the submitted work with Shire, CSL Behring, HEMA Biologics, and other companies.
Source: Kelkar AH et al. N Engl J Med. 2018;379:1216-23.