User login
CECCY: Carvedilol didn’t curb cardiotoxicity in breast cancer patients
ORLANDO – Anthracycline chemotherapy was associated with a cardiotoxicity incidence of roughly 14% of breast cancer patients regardless of treatment with carvedilol, based on data from a randomized trial of 200 patients.
“Cardio-oncology has been neglected,” Monica Samuel Avila, MD, of Hospital das Clínicas da Faculdade de Medicina da Universidade in São Paulo, Brazil, said in a video interview at the annual meeting of the American College of Cardiology. “We have seen improvement of survival in patients with cancer, but with that comes complications related to treatment. I think that the interactions between cardiologists and oncologists are increasing in a more important way,” she said.
In the Carvedilol for Prevention of Chemotherapy-Induced Cardiotoxicity (CECCY) Trial, Dr. Avila and colleagues evaluated primary prevention of cardiotoxicity in women with normal hearts who were undergoing chemotherapy for breast cancer.
Patients in the treatment group received a median carvedilol dose of 18.4 mg/day. The primary endpoint of cardiotoxicity, defined as a decrease in left ventricular ejection fraction (LVEF) of at least 10% at 6 months, occurred in 15% of carvedilol patients and 14% placebo patients, a nonsignificant difference. No significant differences occurred in diastolic dysfunction or in B-type natriuretic peptide (BNP) levels at 6 weeks, 12 weeks, or 24 weeks between the groups.
However, carvedilol patients showed significantly reduced troponin 1 levels compared with placebo, which suggests protection against myocardial injury, Dr. Avila said.
“In short follow up, we can see cardiotoxicity appearing, and we know we have to treat it promptly to prevent cardiac events,” she said.
Dr. Avila and colleagues identified 200 women older than 18 years with HER2-negative breast cancer tumor status and normal left ventricular ejection fraction. The patients were undergoing chemotherapy with 240 mg/m2 of anthracycline and were randomized to treatment with carvedilol or a placebo. Baseline characteristics were similar between the two groups.
Adverse effects were not significantly different between the groups, and the most common events in each group included dizziness, dry mouth, symptomatic hypertension, stomachache, and nausea. Although the results suggest that carvedilol can reduce the risk of myocardial injury, more research is needed to address the question of the increase in troponin without change in the LVEF, Dr. Avila noted. The study is ongoing and the research team intends to follow the low-risk patient population for a total of 2 years. “For high-risk patients, I am already giving carvedilol,” she said. “We believe if we find a difference in LVEF or clinical events, we could encourage cardiologists to give carvedilol in a low-risk population,” she said.
“This study highlights that there is no safe dose of anthracycline,” commented Bonnie Ky, MD of the University of Pennsylvania, Philadelphia, at a press briefing. She emphasized the value of carvedilol for a high-risk population, and stressed the importance of following long-term changes in heart injury markers after 1-2 years for low-risk patients.
Dr. Avila had no financial conflicts to disclose. Dr. Ky disclosed relationships with multiple companies including Bioinvent and Bristol Myers.
The findings were published simultaneously in the Journal of the American College of Cardiology.
SOURCE: Avila, M. ACC 18
ORLANDO – Anthracycline chemotherapy was associated with a cardiotoxicity incidence of roughly 14% of breast cancer patients regardless of treatment with carvedilol, based on data from a randomized trial of 200 patients.
“Cardio-oncology has been neglected,” Monica Samuel Avila, MD, of Hospital das Clínicas da Faculdade de Medicina da Universidade in São Paulo, Brazil, said in a video interview at the annual meeting of the American College of Cardiology. “We have seen improvement of survival in patients with cancer, but with that comes complications related to treatment. I think that the interactions between cardiologists and oncologists are increasing in a more important way,” she said.
In the Carvedilol for Prevention of Chemotherapy-Induced Cardiotoxicity (CECCY) Trial, Dr. Avila and colleagues evaluated primary prevention of cardiotoxicity in women with normal hearts who were undergoing chemotherapy for breast cancer.
Patients in the treatment group received a median carvedilol dose of 18.4 mg/day. The primary endpoint of cardiotoxicity, defined as a decrease in left ventricular ejection fraction (LVEF) of at least 10% at 6 months, occurred in 15% of carvedilol patients and 14% placebo patients, a nonsignificant difference. No significant differences occurred in diastolic dysfunction or in B-type natriuretic peptide (BNP) levels at 6 weeks, 12 weeks, or 24 weeks between the groups.
However, carvedilol patients showed significantly reduced troponin 1 levels compared with placebo, which suggests protection against myocardial injury, Dr. Avila said.
“In short follow up, we can see cardiotoxicity appearing, and we know we have to treat it promptly to prevent cardiac events,” she said.
Dr. Avila and colleagues identified 200 women older than 18 years with HER2-negative breast cancer tumor status and normal left ventricular ejection fraction. The patients were undergoing chemotherapy with 240 mg/m2 of anthracycline and were randomized to treatment with carvedilol or a placebo. Baseline characteristics were similar between the two groups.
Adverse effects were not significantly different between the groups, and the most common events in each group included dizziness, dry mouth, symptomatic hypertension, stomachache, and nausea. Although the results suggest that carvedilol can reduce the risk of myocardial injury, more research is needed to address the question of the increase in troponin without change in the LVEF, Dr. Avila noted. The study is ongoing and the research team intends to follow the low-risk patient population for a total of 2 years. “For high-risk patients, I am already giving carvedilol,” she said. “We believe if we find a difference in LVEF or clinical events, we could encourage cardiologists to give carvedilol in a low-risk population,” she said.
“This study highlights that there is no safe dose of anthracycline,” commented Bonnie Ky, MD of the University of Pennsylvania, Philadelphia, at a press briefing. She emphasized the value of carvedilol for a high-risk population, and stressed the importance of following long-term changes in heart injury markers after 1-2 years for low-risk patients.
Dr. Avila had no financial conflicts to disclose. Dr. Ky disclosed relationships with multiple companies including Bioinvent and Bristol Myers.
The findings were published simultaneously in the Journal of the American College of Cardiology.
SOURCE: Avila, M. ACC 18
ORLANDO – Anthracycline chemotherapy was associated with a cardiotoxicity incidence of roughly 14% of breast cancer patients regardless of treatment with carvedilol, based on data from a randomized trial of 200 patients.
“Cardio-oncology has been neglected,” Monica Samuel Avila, MD, of Hospital das Clínicas da Faculdade de Medicina da Universidade in São Paulo, Brazil, said in a video interview at the annual meeting of the American College of Cardiology. “We have seen improvement of survival in patients with cancer, but with that comes complications related to treatment. I think that the interactions between cardiologists and oncologists are increasing in a more important way,” she said.
In the Carvedilol for Prevention of Chemotherapy-Induced Cardiotoxicity (CECCY) Trial, Dr. Avila and colleagues evaluated primary prevention of cardiotoxicity in women with normal hearts who were undergoing chemotherapy for breast cancer.
Patients in the treatment group received a median carvedilol dose of 18.4 mg/day. The primary endpoint of cardiotoxicity, defined as a decrease in left ventricular ejection fraction (LVEF) of at least 10% at 6 months, occurred in 15% of carvedilol patients and 14% placebo patients, a nonsignificant difference. No significant differences occurred in diastolic dysfunction or in B-type natriuretic peptide (BNP) levels at 6 weeks, 12 weeks, or 24 weeks between the groups.
However, carvedilol patients showed significantly reduced troponin 1 levels compared with placebo, which suggests protection against myocardial injury, Dr. Avila said.
“In short follow up, we can see cardiotoxicity appearing, and we know we have to treat it promptly to prevent cardiac events,” she said.
Dr. Avila and colleagues identified 200 women older than 18 years with HER2-negative breast cancer tumor status and normal left ventricular ejection fraction. The patients were undergoing chemotherapy with 240 mg/m2 of anthracycline and were randomized to treatment with carvedilol or a placebo. Baseline characteristics were similar between the two groups.
Adverse effects were not significantly different between the groups, and the most common events in each group included dizziness, dry mouth, symptomatic hypertension, stomachache, and nausea. Although the results suggest that carvedilol can reduce the risk of myocardial injury, more research is needed to address the question of the increase in troponin without change in the LVEF, Dr. Avila noted. The study is ongoing and the research team intends to follow the low-risk patient population for a total of 2 years. “For high-risk patients, I am already giving carvedilol,” she said. “We believe if we find a difference in LVEF or clinical events, we could encourage cardiologists to give carvedilol in a low-risk population,” she said.
“This study highlights that there is no safe dose of anthracycline,” commented Bonnie Ky, MD of the University of Pennsylvania, Philadelphia, at a press briefing. She emphasized the value of carvedilol for a high-risk population, and stressed the importance of following long-term changes in heart injury markers after 1-2 years for low-risk patients.
Dr. Avila had no financial conflicts to disclose. Dr. Ky disclosed relationships with multiple companies including Bioinvent and Bristol Myers.
The findings were published simultaneously in the Journal of the American College of Cardiology.
SOURCE: Avila, M. ACC 18
REPORTING FROM ACC 18
Key clinical point:
Major finding: Cardiotoxicity was roughly 14% in breast cancer patients treated with anthracycline whether they received carvedilol or placebo.
Study details: CECCY was a randomized, placebo-controlled trial of 200 patients with HER2-negative breast cancer tumor status.
Disclosures: Dr. Avila had no financial conflicts to disclose.
Source: Avila M. ACC 2018.
Wearable defibrillator cuts mortality in post-MI patients
ORLANDO – Wearable cardioverter defibrillator vests failed to significantly cut the rate of arrhythmic death in at-risk post-MI patients but succeeded in significantly dropping total mortality during a median of 84 days of use in the first randomized trial of nonimplanted defibrillators in such patients.
But despite this overall mortality benefit, the 1,524 patients randomized to the WCD group failed to show a significant improvement in the rate of sudden and ventricular tachycardia death, the primary endpoint for the study, said Dr. Olgin, chief of cardiology at the University of California, San Francisco. Total mortality was a secondary endpoint in the study. Based on the total mortality benefit observed and the “totality of evidence” from prior, uncontrolled observational studies, Dr. Olgin concluded that it is now “reasonable” to protect post-MI patients with ejection fractions of 35% or less during the first 40-90 days following an MI when patients can then be assessed for receiving an implantable cardioverter defibrillator.
That would be an upgrade from the current American College of Cardiology/American Heart Association guidelines on managing ventricular arrhythmias and preventing sudden cardiac death, issued in 2017, that classified WCDs as a class IIb recommendations – “may be reasonable” – for post-MI patients with a reduced left ventricular ejection fraction (Circulation. 2017 Oct 30;doi:10.1161/CIR.0000000000000549).
WCDs are currently approved for routine prescribing by U.S. physicians, but their use is very variable in post-MI patients. Just before Dr. Olgin delivered his report at the meeting, a poll of the several thousand meeting attendees who heard his talk showed that roughly a third reported routinely prescribing WCDs, with the other two thirds saying they did not.
Several electrophysiologists who heard the report agreed that further research needs to better tease out which post-MI patients get the most benefit from this treatment.
The patients enrolled in the study “were not a sick population; they had a low event rate,” commented Sana M. Al-Khatib, MD, professor of medicine at Duke University in Durham, N.C. and chair of the panel that wrote the 2017 ventricular arrhythmia guidelines. She suggested testing the efficacy of WCDs in post-MI patients with lower ejection fractions or those with a greater history of heart disease prior to their index MI. Nearly half of the patients enrolled in the study had New York Heart Association class I symptoms, indicating that they had mild heart disease, she noted in an interview. Another issue left unresolved by the results Dr. Olgin reported was how much of the mortality benefit was attributable to the shocks delivered by the tested WCDs and how much derived from the arrhythmia monitoring that the WCDs provided.
Another way to better target WCDs to post-MI patients who could derive the most benefit might be to focus on patients with frequent premature ventricular contractions and nonsustained ventricular tachycardia, suggested Dhanunjaya Lakkireddy, MD, professor of medicine and director of the Center for Excellence in AF and Complex Arrhythmias at the University of Kansas Medical Center in Kansas City. But Dr. Lakkireddy acknowledged that currently left ventricular ejection fraction is the primary surrogate marker cardiologists rely on to identify post-MI patients who are at increased risk for ventricular arrhythmia.
Dr. Olgin countered that the total mortality rate seen among the control, usual care patients in his study, 4.9% during the median 84 day follow-up, closely matched the 5% rate reported in prior trials of at-risk patients who received implantable cardioverter defibrillators.
During follow-up, total mortality occurred in 3.1% of the patients randomized to WCD use and 4.9% among the control patients.
The results also showed that 19% of the patients randomized to the WCD arm failed to ever use the device, and that over the course of follow-up the usage rate fell below 50%. Patients who used the device generally wore it as directed, however, with an average 22 hours a day of use at the start of treatment that subsequently dipped to 21 hours a day near the end of the 90-day treatment period, Dr. Olgin reported.
The most likely explanation for the disparity between the significant effect on total mortality and the insignificant effect on arrhythmia mortality is misclassification of some deaths. “Any small number of misclassified sudden deaths would have dramatically reduced our power to see a difference” in arrhythmia deaths, Dr. Olgin noted.
VEST was sponsored by Zoll, the company that markets the tested device. Dr. Olgin has no personal disclosures. Dr. Al-Khatib and Dr. Lakkireddy had no disclosures. Dr. Wilber is a consultant to Biosense Webster and Medtronic.
mzoler@frontlinemedcom.com
SOURCE: Olgin J et al. ACC 18.
ORLANDO – Wearable cardioverter defibrillator vests failed to significantly cut the rate of arrhythmic death in at-risk post-MI patients but succeeded in significantly dropping total mortality during a median of 84 days of use in the first randomized trial of nonimplanted defibrillators in such patients.
But despite this overall mortality benefit, the 1,524 patients randomized to the WCD group failed to show a significant improvement in the rate of sudden and ventricular tachycardia death, the primary endpoint for the study, said Dr. Olgin, chief of cardiology at the University of California, San Francisco. Total mortality was a secondary endpoint in the study. Based on the total mortality benefit observed and the “totality of evidence” from prior, uncontrolled observational studies, Dr. Olgin concluded that it is now “reasonable” to protect post-MI patients with ejection fractions of 35% or less during the first 40-90 days following an MI when patients can then be assessed for receiving an implantable cardioverter defibrillator.
That would be an upgrade from the current American College of Cardiology/American Heart Association guidelines on managing ventricular arrhythmias and preventing sudden cardiac death, issued in 2017, that classified WCDs as a class IIb recommendations – “may be reasonable” – for post-MI patients with a reduced left ventricular ejection fraction (Circulation. 2017 Oct 30;doi:10.1161/CIR.0000000000000549).
WCDs are currently approved for routine prescribing by U.S. physicians, but their use is very variable in post-MI patients. Just before Dr. Olgin delivered his report at the meeting, a poll of the several thousand meeting attendees who heard his talk showed that roughly a third reported routinely prescribing WCDs, with the other two thirds saying they did not.
Several electrophysiologists who heard the report agreed that further research needs to better tease out which post-MI patients get the most benefit from this treatment.
The patients enrolled in the study “were not a sick population; they had a low event rate,” commented Sana M. Al-Khatib, MD, professor of medicine at Duke University in Durham, N.C. and chair of the panel that wrote the 2017 ventricular arrhythmia guidelines. She suggested testing the efficacy of WCDs in post-MI patients with lower ejection fractions or those with a greater history of heart disease prior to their index MI. Nearly half of the patients enrolled in the study had New York Heart Association class I symptoms, indicating that they had mild heart disease, she noted in an interview. Another issue left unresolved by the results Dr. Olgin reported was how much of the mortality benefit was attributable to the shocks delivered by the tested WCDs and how much derived from the arrhythmia monitoring that the WCDs provided.
Another way to better target WCDs to post-MI patients who could derive the most benefit might be to focus on patients with frequent premature ventricular contractions and nonsustained ventricular tachycardia, suggested Dhanunjaya Lakkireddy, MD, professor of medicine and director of the Center for Excellence in AF and Complex Arrhythmias at the University of Kansas Medical Center in Kansas City. But Dr. Lakkireddy acknowledged that currently left ventricular ejection fraction is the primary surrogate marker cardiologists rely on to identify post-MI patients who are at increased risk for ventricular arrhythmia.
Dr. Olgin countered that the total mortality rate seen among the control, usual care patients in his study, 4.9% during the median 84 day follow-up, closely matched the 5% rate reported in prior trials of at-risk patients who received implantable cardioverter defibrillators.
During follow-up, total mortality occurred in 3.1% of the patients randomized to WCD use and 4.9% among the control patients.
The results also showed that 19% of the patients randomized to the WCD arm failed to ever use the device, and that over the course of follow-up the usage rate fell below 50%. Patients who used the device generally wore it as directed, however, with an average 22 hours a day of use at the start of treatment that subsequently dipped to 21 hours a day near the end of the 90-day treatment period, Dr. Olgin reported.
The most likely explanation for the disparity between the significant effect on total mortality and the insignificant effect on arrhythmia mortality is misclassification of some deaths. “Any small number of misclassified sudden deaths would have dramatically reduced our power to see a difference” in arrhythmia deaths, Dr. Olgin noted.
VEST was sponsored by Zoll, the company that markets the tested device. Dr. Olgin has no personal disclosures. Dr. Al-Khatib and Dr. Lakkireddy had no disclosures. Dr. Wilber is a consultant to Biosense Webster and Medtronic.
mzoler@frontlinemedcom.com
SOURCE: Olgin J et al. ACC 18.
ORLANDO – Wearable cardioverter defibrillator vests failed to significantly cut the rate of arrhythmic death in at-risk post-MI patients but succeeded in significantly dropping total mortality during a median of 84 days of use in the first randomized trial of nonimplanted defibrillators in such patients.
But despite this overall mortality benefit, the 1,524 patients randomized to the WCD group failed to show a significant improvement in the rate of sudden and ventricular tachycardia death, the primary endpoint for the study, said Dr. Olgin, chief of cardiology at the University of California, San Francisco. Total mortality was a secondary endpoint in the study. Based on the total mortality benefit observed and the “totality of evidence” from prior, uncontrolled observational studies, Dr. Olgin concluded that it is now “reasonable” to protect post-MI patients with ejection fractions of 35% or less during the first 40-90 days following an MI when patients can then be assessed for receiving an implantable cardioverter defibrillator.
That would be an upgrade from the current American College of Cardiology/American Heart Association guidelines on managing ventricular arrhythmias and preventing sudden cardiac death, issued in 2017, that classified WCDs as a class IIb recommendations – “may be reasonable” – for post-MI patients with a reduced left ventricular ejection fraction (Circulation. 2017 Oct 30;doi:10.1161/CIR.0000000000000549).
WCDs are currently approved for routine prescribing by U.S. physicians, but their use is very variable in post-MI patients. Just before Dr. Olgin delivered his report at the meeting, a poll of the several thousand meeting attendees who heard his talk showed that roughly a third reported routinely prescribing WCDs, with the other two thirds saying they did not.
Several electrophysiologists who heard the report agreed that further research needs to better tease out which post-MI patients get the most benefit from this treatment.
The patients enrolled in the study “were not a sick population; they had a low event rate,” commented Sana M. Al-Khatib, MD, professor of medicine at Duke University in Durham, N.C. and chair of the panel that wrote the 2017 ventricular arrhythmia guidelines. She suggested testing the efficacy of WCDs in post-MI patients with lower ejection fractions or those with a greater history of heart disease prior to their index MI. Nearly half of the patients enrolled in the study had New York Heart Association class I symptoms, indicating that they had mild heart disease, she noted in an interview. Another issue left unresolved by the results Dr. Olgin reported was how much of the mortality benefit was attributable to the shocks delivered by the tested WCDs and how much derived from the arrhythmia monitoring that the WCDs provided.
Another way to better target WCDs to post-MI patients who could derive the most benefit might be to focus on patients with frequent premature ventricular contractions and nonsustained ventricular tachycardia, suggested Dhanunjaya Lakkireddy, MD, professor of medicine and director of the Center for Excellence in AF and Complex Arrhythmias at the University of Kansas Medical Center in Kansas City. But Dr. Lakkireddy acknowledged that currently left ventricular ejection fraction is the primary surrogate marker cardiologists rely on to identify post-MI patients who are at increased risk for ventricular arrhythmia.
Dr. Olgin countered that the total mortality rate seen among the control, usual care patients in his study, 4.9% during the median 84 day follow-up, closely matched the 5% rate reported in prior trials of at-risk patients who received implantable cardioverter defibrillators.
During follow-up, total mortality occurred in 3.1% of the patients randomized to WCD use and 4.9% among the control patients.
The results also showed that 19% of the patients randomized to the WCD arm failed to ever use the device, and that over the course of follow-up the usage rate fell below 50%. Patients who used the device generally wore it as directed, however, with an average 22 hours a day of use at the start of treatment that subsequently dipped to 21 hours a day near the end of the 90-day treatment period, Dr. Olgin reported.
The most likely explanation for the disparity between the significant effect on total mortality and the insignificant effect on arrhythmia mortality is misclassification of some deaths. “Any small number of misclassified sudden deaths would have dramatically reduced our power to see a difference” in arrhythmia deaths, Dr. Olgin noted.
VEST was sponsored by Zoll, the company that markets the tested device. Dr. Olgin has no personal disclosures. Dr. Al-Khatib and Dr. Lakkireddy had no disclosures. Dr. Wilber is a consultant to Biosense Webster and Medtronic.
mzoler@frontlinemedcom.com
SOURCE: Olgin J et al. ACC 18.
REPORTING FROM ACC18
Key clinical point: The first RCT of a wearable defibrillator in post-MI patients showed reduced total mortality.
Major finding: Total mortality was 3.2% in patients treated with a wearable cardioverter defibrillator and 4.9% in controls after a median of 84 days.
Study details: VEST, a multicenter, randomized trial with 2,302 patients.
Disclosures: VEST was sponsored by Zoll, the company that markets the tested device. Dr. Olgin has no personal disclosures.
Source: Olgin J. ACC 18.
VEST: Closer tailoring might boost wearable cardioverter defibrillator’s benefit
ORLANDO – , the findings failed to show a statistically significant reduction in sudden death or death from ventricular tachycardia. This suggests better targeting of the device is needed, commented Dhanunjaya Lakkireddy, MD, in a video interview at the annual meeting of the American College of Cardiology.
The Vest Prevention of Early Sudden Death Trial (VEST) randomized 2,302 patients within the first 7 days following an acute MI who also had a left ventricular ejection fraction of 35% or less to either 90 days of treatment with a wearable cardioverter defibrillator (WCD) or usual care. After a median of 84 days, the results showed no statistically significant reduction from WCD use in the primary endpoint of sudden death or death from ventricular tachyarrhythmias, but a statistically significant reduction in the secondary endpoint of all-cause death: 3.1% in the patients randomized to WCD use and 4.9% among the control patients, reported Jeffrey Olgin, MD, chief of cardiology at the University of California, San Francisco.
The results suggest that a more robust benefit might occur in post-MI, low ejection fraction patients who undergo additional selection based on having frequent premature ventricular contractions and nonsustained ventricular tachycardia, suggested Dr. Lakkireddy, professor of medicine and director of the Center for Excellence in AF and Complex Arrhythmias at the University of Kansas Medical Center in Kansas City.
VEST was sponsored by Zoll, a company that markets a WCD. Dr. Lakkireddy had no relevant disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Source: Olgin J and Lakkireddy D ACC 18.
ORLANDO – , the findings failed to show a statistically significant reduction in sudden death or death from ventricular tachycardia. This suggests better targeting of the device is needed, commented Dhanunjaya Lakkireddy, MD, in a video interview at the annual meeting of the American College of Cardiology.
The Vest Prevention of Early Sudden Death Trial (VEST) randomized 2,302 patients within the first 7 days following an acute MI who also had a left ventricular ejection fraction of 35% or less to either 90 days of treatment with a wearable cardioverter defibrillator (WCD) or usual care. After a median of 84 days, the results showed no statistically significant reduction from WCD use in the primary endpoint of sudden death or death from ventricular tachyarrhythmias, but a statistically significant reduction in the secondary endpoint of all-cause death: 3.1% in the patients randomized to WCD use and 4.9% among the control patients, reported Jeffrey Olgin, MD, chief of cardiology at the University of California, San Francisco.
The results suggest that a more robust benefit might occur in post-MI, low ejection fraction patients who undergo additional selection based on having frequent premature ventricular contractions and nonsustained ventricular tachycardia, suggested Dr. Lakkireddy, professor of medicine and director of the Center for Excellence in AF and Complex Arrhythmias at the University of Kansas Medical Center in Kansas City.
VEST was sponsored by Zoll, a company that markets a WCD. Dr. Lakkireddy had no relevant disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Source: Olgin J and Lakkireddy D ACC 18.
ORLANDO – , the findings failed to show a statistically significant reduction in sudden death or death from ventricular tachycardia. This suggests better targeting of the device is needed, commented Dhanunjaya Lakkireddy, MD, in a video interview at the annual meeting of the American College of Cardiology.
The Vest Prevention of Early Sudden Death Trial (VEST) randomized 2,302 patients within the first 7 days following an acute MI who also had a left ventricular ejection fraction of 35% or less to either 90 days of treatment with a wearable cardioverter defibrillator (WCD) or usual care. After a median of 84 days, the results showed no statistically significant reduction from WCD use in the primary endpoint of sudden death or death from ventricular tachyarrhythmias, but a statistically significant reduction in the secondary endpoint of all-cause death: 3.1% in the patients randomized to WCD use and 4.9% among the control patients, reported Jeffrey Olgin, MD, chief of cardiology at the University of California, San Francisco.
The results suggest that a more robust benefit might occur in post-MI, low ejection fraction patients who undergo additional selection based on having frequent premature ventricular contractions and nonsustained ventricular tachycardia, suggested Dr. Lakkireddy, professor of medicine and director of the Center for Excellence in AF and Complex Arrhythmias at the University of Kansas Medical Center in Kansas City.
VEST was sponsored by Zoll, a company that markets a WCD. Dr. Lakkireddy had no relevant disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Source: Olgin J and Lakkireddy D ACC 18.
EXPERT ANALYSIS FROM ACC 18
Postmenopausal women: Walk farther and faster to reduce heart failure risk
Brisk walking for at least 40 minutes two or three times a week reduced the risk of heart failure by approximately 25% in postmenopausal women, according to data from more that 89,000 participants in the Women’s Health Initiative.
The benefits of walking are well understood, said Somwail Rasla, MD, of Saint Vincent Hospital in Worcester, Mass., but he and his colleagues focused for the first time on how the speed, frequency, and duration of walking affected health in older women who may be less likely to visit a gym or engage in a formal exercise program.
The researchers followed the women, aged 50-79 years, for approximately 10 years.
Overall, the risk of heart failure was 20%-25% less for women who walked at least twice a week than it was for women who walked less frequently. In addition, women who walked for at least 40 minutes per walk had a 21%-25% lower heart failure risk than did those who walked less than 40 minutes per walk.
Pace mattered as well, Dr. Rasla pointed out. Women walking at an average pace and a fast pace had, respectively, 26% and 38% lower heart failure risk, compared with women who walked at a casual pace.
The researchers measured the women’s energy expenditure using the Metabolic Equivalent of Task (MET), a value calculated using the women’s self-reports of their walking frequency, duration, and speed. The results were similar across different age groups, ethnicities, and baseline body weight, which suggests the findings can be generalized to apply to most women. “I think we could give the same advice [about walking] to women up to age 79,” said Dr. Rasla.
The findings were limited by the use of self-reports, Dr. Rasla noted. However, the results suggest that walking can be a valuable and accessible form of exercise for older women, he said.
The Women’s Health Initiative is sponsored by the National Institutes of Health. The investigators reported no relevant conflicts of interest.
SOURCE: Rasla S et al. ACC 18, Poster 1315M-03.
Brisk walking for at least 40 minutes two or three times a week reduced the risk of heart failure by approximately 25% in postmenopausal women, according to data from more that 89,000 participants in the Women’s Health Initiative.
The benefits of walking are well understood, said Somwail Rasla, MD, of Saint Vincent Hospital in Worcester, Mass., but he and his colleagues focused for the first time on how the speed, frequency, and duration of walking affected health in older women who may be less likely to visit a gym or engage in a formal exercise program.
The researchers followed the women, aged 50-79 years, for approximately 10 years.
Overall, the risk of heart failure was 20%-25% less for women who walked at least twice a week than it was for women who walked less frequently. In addition, women who walked for at least 40 minutes per walk had a 21%-25% lower heart failure risk than did those who walked less than 40 minutes per walk.
Pace mattered as well, Dr. Rasla pointed out. Women walking at an average pace and a fast pace had, respectively, 26% and 38% lower heart failure risk, compared with women who walked at a casual pace.
The researchers measured the women’s energy expenditure using the Metabolic Equivalent of Task (MET), a value calculated using the women’s self-reports of their walking frequency, duration, and speed. The results were similar across different age groups, ethnicities, and baseline body weight, which suggests the findings can be generalized to apply to most women. “I think we could give the same advice [about walking] to women up to age 79,” said Dr. Rasla.
The findings were limited by the use of self-reports, Dr. Rasla noted. However, the results suggest that walking can be a valuable and accessible form of exercise for older women, he said.
The Women’s Health Initiative is sponsored by the National Institutes of Health. The investigators reported no relevant conflicts of interest.
SOURCE: Rasla S et al. ACC 18, Poster 1315M-03.
Brisk walking for at least 40 minutes two or three times a week reduced the risk of heart failure by approximately 25% in postmenopausal women, according to data from more that 89,000 participants in the Women’s Health Initiative.
The benefits of walking are well understood, said Somwail Rasla, MD, of Saint Vincent Hospital in Worcester, Mass., but he and his colleagues focused for the first time on how the speed, frequency, and duration of walking affected health in older women who may be less likely to visit a gym or engage in a formal exercise program.
The researchers followed the women, aged 50-79 years, for approximately 10 years.
Overall, the risk of heart failure was 20%-25% less for women who walked at least twice a week than it was for women who walked less frequently. In addition, women who walked for at least 40 minutes per walk had a 21%-25% lower heart failure risk than did those who walked less than 40 minutes per walk.
Pace mattered as well, Dr. Rasla pointed out. Women walking at an average pace and a fast pace had, respectively, 26% and 38% lower heart failure risk, compared with women who walked at a casual pace.
The researchers measured the women’s energy expenditure using the Metabolic Equivalent of Task (MET), a value calculated using the women’s self-reports of their walking frequency, duration, and speed. The results were similar across different age groups, ethnicities, and baseline body weight, which suggests the findings can be generalized to apply to most women. “I think we could give the same advice [about walking] to women up to age 79,” said Dr. Rasla.
The findings were limited by the use of self-reports, Dr. Rasla noted. However, the results suggest that walking can be a valuable and accessible form of exercise for older women, he said.
The Women’s Health Initiative is sponsored by the National Institutes of Health. The investigators reported no relevant conflicts of interest.
SOURCE: Rasla S et al. ACC 18, Poster 1315M-03.
FROM ACC18
Key clinical point: Urge older female patients to walk briskly at least twice a week.
Major finding: Patients with a fast pace had a 38% lower risk of heart failure.
Study details: A long-term, national observational study of 89,270 women.
Disclosures: The Women’s Health Initiative is sponsored by the National Institutes of Health. The investigators reported no relevant conflicts of interest.
Source: Rasla S et al. ACC 18, Poster 1315M-03.
ACC Day 2: Late-Breaking Clinical Trial highlights
Of the late-breaking clinical trials to be presented on Sunday, March 11, at the annual meeting of the American College of Cardiology in Orlando, the meeting’s vice chair, Andrew Kates, MD, highlighted two in a press briefing: ARTEMIS and MOMENTUM 3.
ARTEMIS
The investigators of this unique trial sought to determine whether reducing patients’ cost burden for their medication would improve adherence and, in turn, improve patient outcomes.
Specifically, ARTEMIS (Affordability and Real-World Antiplatelet Treatment Effectiveness After Myocardial Infarction Study) evaluated whether eliminating copayments for patients after acute MI could optimize antiplatelet therapy selection and long-term adherence, as well as patient outcomes and overall cost of care following acute MI.
Dr. Kates, professor of medicine, Washington University, St. Louis, said ARTEMIS would go far in answering the question, “If costs are lowered, are guidelines followed?”
Tracy Wang, MD, of Duke University, Durham, N.C., will present the results on Sunday in a joint Late-Breaking Clinical Trial session with JAMA, being held at 8 a.m.-9 a.m. in the Main Tent (Hall C).
MOMENTUM 3
Last year, HeartMate 3, a fully magnetically levitated centrifugal-flow circulatory pump, showed better outcomes at 6 months than did HEARTMATE 2, an axial-flow pump, in the primary results of MOMENTUM 3 (Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy With HeartMate 3).
Most impressive was the complete elimination of pump thrombosis in the patients with the smaller HeartMate 3 device. On Sunday, the 2-year results will reveal whether longer-term use of the novel technology will carry similar benefits, Dr. Kates said.
MOMENTUM 3 will be presented at the third Late-Breaking Clinical Trials session, at 10:45-11:45 a.m., in the Main Tent (Hall C) by Mandeep R. Mehra, MD, professor of medicine at Harvard Medical School and medical director of the Heart and Vascular Center of Brigham and Women’s Hospital, both in Boston.
Of the late-breaking clinical trials to be presented on Sunday, March 11, at the annual meeting of the American College of Cardiology in Orlando, the meeting’s vice chair, Andrew Kates, MD, highlighted two in a press briefing: ARTEMIS and MOMENTUM 3.
ARTEMIS
The investigators of this unique trial sought to determine whether reducing patients’ cost burden for their medication would improve adherence and, in turn, improve patient outcomes.
Specifically, ARTEMIS (Affordability and Real-World Antiplatelet Treatment Effectiveness After Myocardial Infarction Study) evaluated whether eliminating copayments for patients after acute MI could optimize antiplatelet therapy selection and long-term adherence, as well as patient outcomes and overall cost of care following acute MI.
Dr. Kates, professor of medicine, Washington University, St. Louis, said ARTEMIS would go far in answering the question, “If costs are lowered, are guidelines followed?”
Tracy Wang, MD, of Duke University, Durham, N.C., will present the results on Sunday in a joint Late-Breaking Clinical Trial session with JAMA, being held at 8 a.m.-9 a.m. in the Main Tent (Hall C).
MOMENTUM 3
Last year, HeartMate 3, a fully magnetically levitated centrifugal-flow circulatory pump, showed better outcomes at 6 months than did HEARTMATE 2, an axial-flow pump, in the primary results of MOMENTUM 3 (Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy With HeartMate 3).
Most impressive was the complete elimination of pump thrombosis in the patients with the smaller HeartMate 3 device. On Sunday, the 2-year results will reveal whether longer-term use of the novel technology will carry similar benefits, Dr. Kates said.
MOMENTUM 3 will be presented at the third Late-Breaking Clinical Trials session, at 10:45-11:45 a.m., in the Main Tent (Hall C) by Mandeep R. Mehra, MD, professor of medicine at Harvard Medical School and medical director of the Heart and Vascular Center of Brigham and Women’s Hospital, both in Boston.
Of the late-breaking clinical trials to be presented on Sunday, March 11, at the annual meeting of the American College of Cardiology in Orlando, the meeting’s vice chair, Andrew Kates, MD, highlighted two in a press briefing: ARTEMIS and MOMENTUM 3.
ARTEMIS
The investigators of this unique trial sought to determine whether reducing patients’ cost burden for their medication would improve adherence and, in turn, improve patient outcomes.
Specifically, ARTEMIS (Affordability and Real-World Antiplatelet Treatment Effectiveness After Myocardial Infarction Study) evaluated whether eliminating copayments for patients after acute MI could optimize antiplatelet therapy selection and long-term adherence, as well as patient outcomes and overall cost of care following acute MI.
Dr. Kates, professor of medicine, Washington University, St. Louis, said ARTEMIS would go far in answering the question, “If costs are lowered, are guidelines followed?”
Tracy Wang, MD, of Duke University, Durham, N.C., will present the results on Sunday in a joint Late-Breaking Clinical Trial session with JAMA, being held at 8 a.m.-9 a.m. in the Main Tent (Hall C).
MOMENTUM 3
Last year, HeartMate 3, a fully magnetically levitated centrifugal-flow circulatory pump, showed better outcomes at 6 months than did HEARTMATE 2, an axial-flow pump, in the primary results of MOMENTUM 3 (Multicenter Study of MagLev Technology in Patients Undergoing Mechanical Circulatory Support Therapy With HeartMate 3).
Most impressive was the complete elimination of pump thrombosis in the patients with the smaller HeartMate 3 device. On Sunday, the 2-year results will reveal whether longer-term use of the novel technology will carry similar benefits, Dr. Kates said.
MOMENTUM 3 will be presented at the third Late-Breaking Clinical Trials session, at 10:45-11:45 a.m., in the Main Tent (Hall C) by Mandeep R. Mehra, MD, professor of medicine at Harvard Medical School and medical director of the Heart and Vascular Center of Brigham and Women’s Hospital, both in Boston.
Mild cognitive impairment rises in heart patients with comorbidities
ANAHEIM, CALIF. – Across the spectrum of cardiovascular disease, the more comorbid conditions a patient has, the higher the likelihood of mild cognitive impairment, Jocasta Ball, PhD, reported at the American Heart Association scientific sessions.
Indeed, her cross-sectional analysis of baseline data on 2,161 participants in five randomized controlled trials of nurse-led chronic disease management in cardiovascular disease (CVD) showed that for every 1-unit increase in the age-adjusted Charlson Comorbidity Index, the likelihood of mild cognitive impairment (MCI) jumped by 19%.
This novel observation has important clinical implications: “MCI is becoming increasingly recognized as exerting a powerful and negative impact on the risk, management, and prognosis of CVD patients,” explained Dr. Ball of the Baker Heart and Diabetes Institute in Melbourne. “Because MCI undermines a patient’s ability to comply with medical treatment and adds to patient complexity, it is critical [to identify] higher-risk individuals who require closer surveillance and improved early intervention.”
She added that the findings open up a whole new field of research aimed at developing new interventions to help patients with CVD and MCI stay on track with their heart disease treatment program.
The 2,161 subjects, mean age 70 years and two-thirds male, ranged across the full spectrum of cardiovascular disease, from mild to severe. All were screened for MCI by completing the Montreal Cognitive Assessment, or MoCA. A MoCA score below 26 out of a possible 30 is defined as MCI.
Forty-seven percent of subjects had MCI. They were older, with a mean age of 73 years versus 67 years; were more likely to have a history of stroke, by a margin of 20% versus 12%; had a 52% prevalence of atrial fibrillation versus 44%; and had a 50% prevalence of heart failure versus 39% in subjects with normal cognition. In addition, 48% of the MCI group screened positive for depressive symptoms versus 37% of those without MCI, and 28% of patients with MCI had type 2 diabetes, compared with 22% of those without MCI. Renal disease was also significantly more prevalent in the MCI group, by a margin of 21% versus 14%.
In a multivariate regression analysis, the strongest predictors of MCI in patients across the spectrum of CVD were current smoking, with a 2.5-fold increased risk compared with that of nonsmokers, and atrial fibrillation, with a 1.3-fold increased risk.
Dr. Ball reported having no financial conflicts regarding her study.
SOURCE: Ball J. et al. AHA 2017, Abstract 16240.
ANAHEIM, CALIF. – Across the spectrum of cardiovascular disease, the more comorbid conditions a patient has, the higher the likelihood of mild cognitive impairment, Jocasta Ball, PhD, reported at the American Heart Association scientific sessions.
Indeed, her cross-sectional analysis of baseline data on 2,161 participants in five randomized controlled trials of nurse-led chronic disease management in cardiovascular disease (CVD) showed that for every 1-unit increase in the age-adjusted Charlson Comorbidity Index, the likelihood of mild cognitive impairment (MCI) jumped by 19%.
This novel observation has important clinical implications: “MCI is becoming increasingly recognized as exerting a powerful and negative impact on the risk, management, and prognosis of CVD patients,” explained Dr. Ball of the Baker Heart and Diabetes Institute in Melbourne. “Because MCI undermines a patient’s ability to comply with medical treatment and adds to patient complexity, it is critical [to identify] higher-risk individuals who require closer surveillance and improved early intervention.”
She added that the findings open up a whole new field of research aimed at developing new interventions to help patients with CVD and MCI stay on track with their heart disease treatment program.
The 2,161 subjects, mean age 70 years and two-thirds male, ranged across the full spectrum of cardiovascular disease, from mild to severe. All were screened for MCI by completing the Montreal Cognitive Assessment, or MoCA. A MoCA score below 26 out of a possible 30 is defined as MCI.
Forty-seven percent of subjects had MCI. They were older, with a mean age of 73 years versus 67 years; were more likely to have a history of stroke, by a margin of 20% versus 12%; had a 52% prevalence of atrial fibrillation versus 44%; and had a 50% prevalence of heart failure versus 39% in subjects with normal cognition. In addition, 48% of the MCI group screened positive for depressive symptoms versus 37% of those without MCI, and 28% of patients with MCI had type 2 diabetes, compared with 22% of those without MCI. Renal disease was also significantly more prevalent in the MCI group, by a margin of 21% versus 14%.
In a multivariate regression analysis, the strongest predictors of MCI in patients across the spectrum of CVD were current smoking, with a 2.5-fold increased risk compared with that of nonsmokers, and atrial fibrillation, with a 1.3-fold increased risk.
Dr. Ball reported having no financial conflicts regarding her study.
SOURCE: Ball J. et al. AHA 2017, Abstract 16240.
ANAHEIM, CALIF. – Across the spectrum of cardiovascular disease, the more comorbid conditions a patient has, the higher the likelihood of mild cognitive impairment, Jocasta Ball, PhD, reported at the American Heart Association scientific sessions.
Indeed, her cross-sectional analysis of baseline data on 2,161 participants in five randomized controlled trials of nurse-led chronic disease management in cardiovascular disease (CVD) showed that for every 1-unit increase in the age-adjusted Charlson Comorbidity Index, the likelihood of mild cognitive impairment (MCI) jumped by 19%.
This novel observation has important clinical implications: “MCI is becoming increasingly recognized as exerting a powerful and negative impact on the risk, management, and prognosis of CVD patients,” explained Dr. Ball of the Baker Heart and Diabetes Institute in Melbourne. “Because MCI undermines a patient’s ability to comply with medical treatment and adds to patient complexity, it is critical [to identify] higher-risk individuals who require closer surveillance and improved early intervention.”
She added that the findings open up a whole new field of research aimed at developing new interventions to help patients with CVD and MCI stay on track with their heart disease treatment program.
The 2,161 subjects, mean age 70 years and two-thirds male, ranged across the full spectrum of cardiovascular disease, from mild to severe. All were screened for MCI by completing the Montreal Cognitive Assessment, or MoCA. A MoCA score below 26 out of a possible 30 is defined as MCI.
Forty-seven percent of subjects had MCI. They were older, with a mean age of 73 years versus 67 years; were more likely to have a history of stroke, by a margin of 20% versus 12%; had a 52% prevalence of atrial fibrillation versus 44%; and had a 50% prevalence of heart failure versus 39% in subjects with normal cognition. In addition, 48% of the MCI group screened positive for depressive symptoms versus 37% of those without MCI, and 28% of patients with MCI had type 2 diabetes, compared with 22% of those without MCI. Renal disease was also significantly more prevalent in the MCI group, by a margin of 21% versus 14%.
In a multivariate regression analysis, the strongest predictors of MCI in patients across the spectrum of CVD were current smoking, with a 2.5-fold increased risk compared with that of nonsmokers, and atrial fibrillation, with a 1.3-fold increased risk.
Dr. Ball reported having no financial conflicts regarding her study.
SOURCE: Ball J. et al. AHA 2017, Abstract 16240.
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: The more comorbid conditions a patient with cardiovascular disease has, the greater the likelihood of mild cognitive impairment becomes.
Major finding: For each 1-unit increase in the Charlson Comorbidity Index, the likelihood of prevalent mild cognitive impairment rose by 19%.
Study details: This cross-sectional study assessed the association between mild cognitive impairment and Charlson Comorbidity Index score in 2,161 patients with cardiovascular disease of varied degrees of severity.
Disclosures: The presenter reported having no financial conflicts regarding her study.
Source: Ball J et al. AHA 2017, Abstract 16240
200 cardiovascular drugs now in development
, according to Pharmaceutical Research and Manufacturers of America.
One of those 42 treatments is “a nonviral gene therapy that targets a tissue repair and regeneration pathway in the body,” PhRMA noted. A total of 200 medicines, including those for lipid disorders and hypertension, were either in clinical trials or undergoing regulatory review as of Jan. 24, 2018, the group reported. Some treatments are being considered for more than one condition.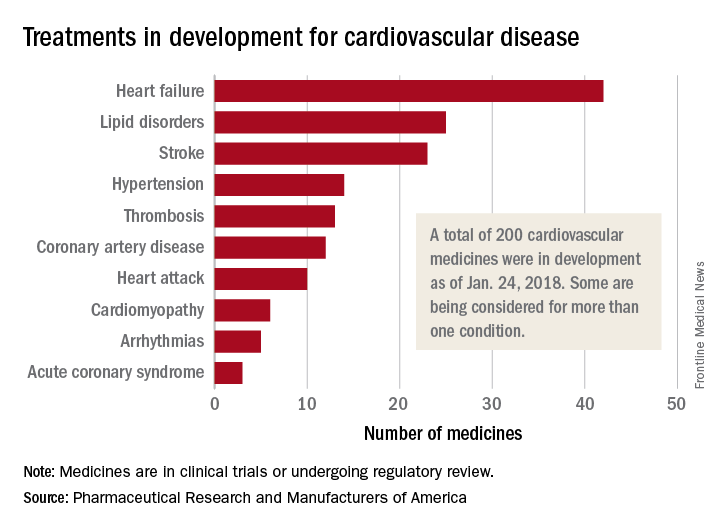
, according to Pharmaceutical Research and Manufacturers of America.
One of those 42 treatments is “a nonviral gene therapy that targets a tissue repair and regeneration pathway in the body,” PhRMA noted. A total of 200 medicines, including those for lipid disorders and hypertension, were either in clinical trials or undergoing regulatory review as of Jan. 24, 2018, the group reported. Some treatments are being considered for more than one condition.
, according to Pharmaceutical Research and Manufacturers of America.
One of those 42 treatments is “a nonviral gene therapy that targets a tissue repair and regeneration pathway in the body,” PhRMA noted. A total of 200 medicines, including those for lipid disorders and hypertension, were either in clinical trials or undergoing regulatory review as of Jan. 24, 2018, the group reported. Some treatments are being considered for more than one condition.
Abruptio placenta brings increased cardiovascular risk – and soon
ANAHEIM, CALIF. – Women who experience placental abruption are at significantly increased risk for multiple forms of cardiovascular disease beginning within the first few years after their pregnancy complication, according to a study of more than 1.6 million California women.
While gestational hypertension, preeclampsia, and fetal growth restriction have previously all been shown to be associated with increased risk of incident cardiovascular disease, this huge California study provides the first strong epidemiologic evidence that placental abruption is as well. Prior studies looking at the issue have been underpowered, Michael J. Healey, MD, said at the American Heart Association scientific sessions.
“Our hypothesis is that there might be some type of shared mechanism, probably involving microvascular dysfunction, that explains the relationships we see between these pregnancy complications and increased near-term risk of cardiovascular disease,” he explained in an interview.
Dr. Healy, a hospitalist attached to the heart failure service at the University of California, San Francisco, presented a retrospective study of a multiethnic cohort comprising 1,614,950 parous women aged 15-50 years who participated in the California Healthcare Cost and Utility Project during 2005-2009. Placental abruption occurred in 15,057 of them at a mean age of 29.2 years.
During a median 4.9 years of follow-up, women who experienced abruptio placenta were at 6% increased risk for heart failure, 11% greater risk for MI, 8% increased risk for hypertensive urgency, and 2% greater risk for myocardial infarction with no obstructive atherosclerosis (MINOCA) in an age- and race-adjusted analysis. All of these were statistically significant differences.
Of note, however, in a multivariate analysis fully adjusted for standard cardiovascular risk factors, as well as hypercoagulability, preterm birth, grand multiparity, and insurance status, placental abruption was independently associated with a 2.14-fold risk of MINOCA, but it was no longer linked to significantly increased risks of the other cardiovascular events.
The implication is that the increased risk of these other forms of cardiovascular disease is mediated through the women’s increased prevalence of the traditional cardiovascular risk factors, whereas a novel mechanism – most likely microvascular dysfunction – underlies the association between placental abruption and MINOCA, according to Dr. Healy.
He plans to extend this research by taking a look at the relationship between placental abruption and the various subtypes of MINOCA, including coronary dissection, vasospasm, thrombophilia disorders, and stress cardiomyopathy, in order to examine whether the increased risk posed by placental abruption is concentrated in certain forms of MINOCA. Data on MINOCA subtypes were recorded as part of the California project.
He reported having no financial conflicts of interest regarding his study.
ANAHEIM, CALIF. – Women who experience placental abruption are at significantly increased risk for multiple forms of cardiovascular disease beginning within the first few years after their pregnancy complication, according to a study of more than 1.6 million California women.
While gestational hypertension, preeclampsia, and fetal growth restriction have previously all been shown to be associated with increased risk of incident cardiovascular disease, this huge California study provides the first strong epidemiologic evidence that placental abruption is as well. Prior studies looking at the issue have been underpowered, Michael J. Healey, MD, said at the American Heart Association scientific sessions.
“Our hypothesis is that there might be some type of shared mechanism, probably involving microvascular dysfunction, that explains the relationships we see between these pregnancy complications and increased near-term risk of cardiovascular disease,” he explained in an interview.
Dr. Healy, a hospitalist attached to the heart failure service at the University of California, San Francisco, presented a retrospective study of a multiethnic cohort comprising 1,614,950 parous women aged 15-50 years who participated in the California Healthcare Cost and Utility Project during 2005-2009. Placental abruption occurred in 15,057 of them at a mean age of 29.2 years.
During a median 4.9 years of follow-up, women who experienced abruptio placenta were at 6% increased risk for heart failure, 11% greater risk for MI, 8% increased risk for hypertensive urgency, and 2% greater risk for myocardial infarction with no obstructive atherosclerosis (MINOCA) in an age- and race-adjusted analysis. All of these were statistically significant differences.
Of note, however, in a multivariate analysis fully adjusted for standard cardiovascular risk factors, as well as hypercoagulability, preterm birth, grand multiparity, and insurance status, placental abruption was independently associated with a 2.14-fold risk of MINOCA, but it was no longer linked to significantly increased risks of the other cardiovascular events.
The implication is that the increased risk of these other forms of cardiovascular disease is mediated through the women’s increased prevalence of the traditional cardiovascular risk factors, whereas a novel mechanism – most likely microvascular dysfunction – underlies the association between placental abruption and MINOCA, according to Dr. Healy.
He plans to extend this research by taking a look at the relationship between placental abruption and the various subtypes of MINOCA, including coronary dissection, vasospasm, thrombophilia disorders, and stress cardiomyopathy, in order to examine whether the increased risk posed by placental abruption is concentrated in certain forms of MINOCA. Data on MINOCA subtypes were recorded as part of the California project.
He reported having no financial conflicts of interest regarding his study.
ANAHEIM, CALIF. – Women who experience placental abruption are at significantly increased risk for multiple forms of cardiovascular disease beginning within the first few years after their pregnancy complication, according to a study of more than 1.6 million California women.
While gestational hypertension, preeclampsia, and fetal growth restriction have previously all been shown to be associated with increased risk of incident cardiovascular disease, this huge California study provides the first strong epidemiologic evidence that placental abruption is as well. Prior studies looking at the issue have been underpowered, Michael J. Healey, MD, said at the American Heart Association scientific sessions.
“Our hypothesis is that there might be some type of shared mechanism, probably involving microvascular dysfunction, that explains the relationships we see between these pregnancy complications and increased near-term risk of cardiovascular disease,” he explained in an interview.
Dr. Healy, a hospitalist attached to the heart failure service at the University of California, San Francisco, presented a retrospective study of a multiethnic cohort comprising 1,614,950 parous women aged 15-50 years who participated in the California Healthcare Cost and Utility Project during 2005-2009. Placental abruption occurred in 15,057 of them at a mean age of 29.2 years.
During a median 4.9 years of follow-up, women who experienced abruptio placenta were at 6% increased risk for heart failure, 11% greater risk for MI, 8% increased risk for hypertensive urgency, and 2% greater risk for myocardial infarction with no obstructive atherosclerosis (MINOCA) in an age- and race-adjusted analysis. All of these were statistically significant differences.
Of note, however, in a multivariate analysis fully adjusted for standard cardiovascular risk factors, as well as hypercoagulability, preterm birth, grand multiparity, and insurance status, placental abruption was independently associated with a 2.14-fold risk of MINOCA, but it was no longer linked to significantly increased risks of the other cardiovascular events.
The implication is that the increased risk of these other forms of cardiovascular disease is mediated through the women’s increased prevalence of the traditional cardiovascular risk factors, whereas a novel mechanism – most likely microvascular dysfunction – underlies the association between placental abruption and MINOCA, according to Dr. Healy.
He plans to extend this research by taking a look at the relationship between placental abruption and the various subtypes of MINOCA, including coronary dissection, vasospasm, thrombophilia disorders, and stress cardiomyopathy, in order to examine whether the increased risk posed by placental abruption is concentrated in certain forms of MINOCA. Data on MINOCA subtypes were recorded as part of the California project.
He reported having no financial conflicts of interest regarding his study.
REPORTING FROM THE AHA SCIENTIFIC SESSIONS
Key clinical point: Placental abruption is associated with increased risk of maternal cardiovascular events within a few years after delivery.
Major finding: Placental abruption was independently associated with a 2.14-fold increased risk of myocardial infarction with no obstructive atherosclerosis during a median 4.9 years of follow-up.
Study details: This was a retrospective study of more than 1.6 million parous women enrolled in the California Healthcare Cost and Utilization Project, including 15,057 with placental abruption.
Disclosures: The study presenter reported having no financial conflicts of interest.
Overweight and obese individuals face greater cardiovascular morbidity
.
In a report published online Feb. 28 in JAMA Cardiology, researchers presented an analysis of pooled data from 190,672 participants and 3.2 million person-years of follow-up in 10 prospective cohort studies, including the Framingham Heart Study, the Multi-Ethnic Study of Atherosclerosis, and the Atherosclerosis Risk in Communities Study.
Incident cardiovascular events occurred in 37% of overweight middle-aged men and 28% of overweight middle-aged women. In obese middle-aged men and women, those figures were 47% and 39%, respectively, and in the morbidly obese they were 65% and 48%. By comparison, incident cardiovascular events occurred in 32% of middle-aged men of normal BMI, and 22% of women.
Across all the studies, there were 7,136 fatal or nonfatal myocardial infarctions, 3,733 fatal or nonfatal strokes, 4,614 diagnoses of heart failure, and 13,457 cardiovascular disease events during 856,532 person-years of follow-up in middle-aged adults.
After adjustment for age, ethnicity, and smoking status, the competing hazard ratios for experiencing a cardiovascular disease event compared to a noncardiovascular disease death were greater in the higher-BMI categories, and greatest among morbidly obese middle-aged men and women, largely because of a greater proportion of coronary heart disease and heart failure events.
“In addition, greater all-cause mortality in higher-BMI categories occurred at the expense of a greater proportion of deaths from cardiovascular causes in middle-aged men and women who are overweight and obese,” wrote Sadiya S. Khan, MD, MSc, of Northwestern University, Chicago, and her coauthors.
The research suggested that for each increasing unit of BMI in middle-aged men and women, the adjusted competing hazard ratios of incident cardiovascular disease events increased by a significant 5%.
The study found that middle-aged men in the normal and overweight BMI group enjoyed more years free from cardiovascular disease than did obese middle-aged men. In middle-aged women, those who were in the normal BMI range had significantly more years lived free of cardiovascular disease than did overweight or obese women.
The incidence of cardiovascular disease was significantly delayed by an average of 7.5 years in middle-aged men of normal BMI and 7.1 years in middle-aged women of normal BMI, compared with those with morbid obesity.
In terms of longevity, men and women with normal BMI lived on average 5.6 years and 2 years longer, respectively, than did men and women with morbid obesity.
“The results of this study build on prior research from the Cardiovascular Disease Lifetime Risk Pooling Project highlighting marked differences in lifetime risks of CVD and further highlight the importance of consideration of BMI as a risk factor for diminished healthy longevity and greater overall CVD morbidity and mortality,” the authors wrote.
The study was intended to address recent controversy over the health implications of overweight, with some evidence suggesting that overweight individuals have all-cause mortality similar to or lower than that of normal-weight groups.
“While we do observe evidence of the well-described overweight and obesity paradox, in which heavier individuals appear to live longer on average after diagnosis of CVD compared with individuals with normal BMI, our data when following up individuals prior to the onset of CVD indicate that this occurs because of a trend toward earlier onset of disease in individuals who are overweight and obese,” they wrote.
The study did not account for change in BMI over the course of follow-up, nor did it use data on fat distribution or the degree of visceral adiposity, the researchers noted.
“Additional important outcomes of obesity-related morbidity, such as atrial fibrillation, sleep-disordered breathing, and chronic liver disease, were not ascertained routinely in our cohort studies, and we likely underestimated the overall comorbidity burden of excess weight.”
The National Heart, Lung, and Blood Institute supported the study. No conflicts of interest were declared.
SOURCE: Khan SS et al. JAMA Cardiol. 2018 Feb 28. doi: 10.1001/jamacardio.2018.0022.
.
In a report published online Feb. 28 in JAMA Cardiology, researchers presented an analysis of pooled data from 190,672 participants and 3.2 million person-years of follow-up in 10 prospective cohort studies, including the Framingham Heart Study, the Multi-Ethnic Study of Atherosclerosis, and the Atherosclerosis Risk in Communities Study.
Incident cardiovascular events occurred in 37% of overweight middle-aged men and 28% of overweight middle-aged women. In obese middle-aged men and women, those figures were 47% and 39%, respectively, and in the morbidly obese they were 65% and 48%. By comparison, incident cardiovascular events occurred in 32% of middle-aged men of normal BMI, and 22% of women.
Across all the studies, there were 7,136 fatal or nonfatal myocardial infarctions, 3,733 fatal or nonfatal strokes, 4,614 diagnoses of heart failure, and 13,457 cardiovascular disease events during 856,532 person-years of follow-up in middle-aged adults.
After adjustment for age, ethnicity, and smoking status, the competing hazard ratios for experiencing a cardiovascular disease event compared to a noncardiovascular disease death were greater in the higher-BMI categories, and greatest among morbidly obese middle-aged men and women, largely because of a greater proportion of coronary heart disease and heart failure events.
“In addition, greater all-cause mortality in higher-BMI categories occurred at the expense of a greater proportion of deaths from cardiovascular causes in middle-aged men and women who are overweight and obese,” wrote Sadiya S. Khan, MD, MSc, of Northwestern University, Chicago, and her coauthors.
The research suggested that for each increasing unit of BMI in middle-aged men and women, the adjusted competing hazard ratios of incident cardiovascular disease events increased by a significant 5%.
The study found that middle-aged men in the normal and overweight BMI group enjoyed more years free from cardiovascular disease than did obese middle-aged men. In middle-aged women, those who were in the normal BMI range had significantly more years lived free of cardiovascular disease than did overweight or obese women.
The incidence of cardiovascular disease was significantly delayed by an average of 7.5 years in middle-aged men of normal BMI and 7.1 years in middle-aged women of normal BMI, compared with those with morbid obesity.
In terms of longevity, men and women with normal BMI lived on average 5.6 years and 2 years longer, respectively, than did men and women with morbid obesity.
“The results of this study build on prior research from the Cardiovascular Disease Lifetime Risk Pooling Project highlighting marked differences in lifetime risks of CVD and further highlight the importance of consideration of BMI as a risk factor for diminished healthy longevity and greater overall CVD morbidity and mortality,” the authors wrote.
The study was intended to address recent controversy over the health implications of overweight, with some evidence suggesting that overweight individuals have all-cause mortality similar to or lower than that of normal-weight groups.
“While we do observe evidence of the well-described overweight and obesity paradox, in which heavier individuals appear to live longer on average after diagnosis of CVD compared with individuals with normal BMI, our data when following up individuals prior to the onset of CVD indicate that this occurs because of a trend toward earlier onset of disease in individuals who are overweight and obese,” they wrote.
The study did not account for change in BMI over the course of follow-up, nor did it use data on fat distribution or the degree of visceral adiposity, the researchers noted.
“Additional important outcomes of obesity-related morbidity, such as atrial fibrillation, sleep-disordered breathing, and chronic liver disease, were not ascertained routinely in our cohort studies, and we likely underestimated the overall comorbidity burden of excess weight.”
The National Heart, Lung, and Blood Institute supported the study. No conflicts of interest were declared.
SOURCE: Khan SS et al. JAMA Cardiol. 2018 Feb 28. doi: 10.1001/jamacardio.2018.0022.
.
In a report published online Feb. 28 in JAMA Cardiology, researchers presented an analysis of pooled data from 190,672 participants and 3.2 million person-years of follow-up in 10 prospective cohort studies, including the Framingham Heart Study, the Multi-Ethnic Study of Atherosclerosis, and the Atherosclerosis Risk in Communities Study.
Incident cardiovascular events occurred in 37% of overweight middle-aged men and 28% of overweight middle-aged women. In obese middle-aged men and women, those figures were 47% and 39%, respectively, and in the morbidly obese they were 65% and 48%. By comparison, incident cardiovascular events occurred in 32% of middle-aged men of normal BMI, and 22% of women.
Across all the studies, there were 7,136 fatal or nonfatal myocardial infarctions, 3,733 fatal or nonfatal strokes, 4,614 diagnoses of heart failure, and 13,457 cardiovascular disease events during 856,532 person-years of follow-up in middle-aged adults.
After adjustment for age, ethnicity, and smoking status, the competing hazard ratios for experiencing a cardiovascular disease event compared to a noncardiovascular disease death were greater in the higher-BMI categories, and greatest among morbidly obese middle-aged men and women, largely because of a greater proportion of coronary heart disease and heart failure events.
“In addition, greater all-cause mortality in higher-BMI categories occurred at the expense of a greater proportion of deaths from cardiovascular causes in middle-aged men and women who are overweight and obese,” wrote Sadiya S. Khan, MD, MSc, of Northwestern University, Chicago, and her coauthors.
The research suggested that for each increasing unit of BMI in middle-aged men and women, the adjusted competing hazard ratios of incident cardiovascular disease events increased by a significant 5%.
The study found that middle-aged men in the normal and overweight BMI group enjoyed more years free from cardiovascular disease than did obese middle-aged men. In middle-aged women, those who were in the normal BMI range had significantly more years lived free of cardiovascular disease than did overweight or obese women.
The incidence of cardiovascular disease was significantly delayed by an average of 7.5 years in middle-aged men of normal BMI and 7.1 years in middle-aged women of normal BMI, compared with those with morbid obesity.
In terms of longevity, men and women with normal BMI lived on average 5.6 years and 2 years longer, respectively, than did men and women with morbid obesity.
“The results of this study build on prior research from the Cardiovascular Disease Lifetime Risk Pooling Project highlighting marked differences in lifetime risks of CVD and further highlight the importance of consideration of BMI as a risk factor for diminished healthy longevity and greater overall CVD morbidity and mortality,” the authors wrote.
The study was intended to address recent controversy over the health implications of overweight, with some evidence suggesting that overweight individuals have all-cause mortality similar to or lower than that of normal-weight groups.
“While we do observe evidence of the well-described overweight and obesity paradox, in which heavier individuals appear to live longer on average after diagnosis of CVD compared with individuals with normal BMI, our data when following up individuals prior to the onset of CVD indicate that this occurs because of a trend toward earlier onset of disease in individuals who are overweight and obese,” they wrote.
The study did not account for change in BMI over the course of follow-up, nor did it use data on fat distribution or the degree of visceral adiposity, the researchers noted.
“Additional important outcomes of obesity-related morbidity, such as atrial fibrillation, sleep-disordered breathing, and chronic liver disease, were not ascertained routinely in our cohort studies, and we likely underestimated the overall comorbidity burden of excess weight.”
The National Heart, Lung, and Blood Institute supported the study. No conflicts of interest were declared.
SOURCE: Khan SS et al. JAMA Cardiol. 2018 Feb 28. doi: 10.1001/jamacardio.2018.0022.
FROM JAMA CARDIOLOGY
Key clinical point: Obese individuals have shorter life spans and spend significantly more time dealing with the burden of cardiovascular morbidity than do normal-weight individuals.
Major finding: Overweight and obese middle-aged individuals have a significantly higher incidence of cardiovascular events and mortality compared with normal-weight middle-aged individuals.
Data source: Analysis of pooled data from 190,672 participants and 3.2 million person-years of follow-up in 10 prospective cohort studies.
Disclosures: The National Heart, Lung, and Blood Institute supported the study. No conflicts of interest were declared.
Source: Khan SS et al. JAMA Cardiol. 2018 Feb 28. doi: 10.1001/jamacardio.2018.0022
Viremic suppression linked to decreased MACE rate in patients with HCV-cirrhosis
Hepatitis C viremic suppression was associated with a lower rate of cardiovascular events in patients with compensated HCV-related cirrhosis, compared with control patients who did not achieve a sustained virological response. In addition, predictive factors for major adverse cardiovascular events (MACEs) in compensated HCV-related cirrhosis were Asian ethnic origin, hypertension, smoking, and low serum albumin, according to a report in American Heart Journal.
A total of 878 patients with HCV-related cirrhosis were enrolled at 35 French centers. Upon enrollment, all patients received HCV treatment and were followed for MACEs, including stroke, myocardial infarction, ischemic heart disease, heart failure, peripheral arterial disease, cardiac arrest, and cardiovascular-related death, according to Patrice Cacoub, MD, of Sorbonne Universités, Paris, and his colleagues.
Five-year survival for patients presenting with a MACE was 60% vs. 88% in patients who did not have an event.
“[Our] results strengthen the systemic nature of HCV infection, a chronic disease in which cardiovascular risk must be carefully assessed. The decreased rate of MACEs after [sustained virological response] in this population should be taken into account to enable wider access to new [direct-acting antivirals]. Further studies are warranted to evaluate whether a similar benefit can be obtained in less severe patients, such as noncirrhotic HCV-infected patients,” the researchers concluded.
The authors reported having no conflicts of interest.
SOURCE: Cacoub, P et al. Am Heart J. 2018;198:4-17.
Hepatitis C viremic suppression was associated with a lower rate of cardiovascular events in patients with compensated HCV-related cirrhosis, compared with control patients who did not achieve a sustained virological response. In addition, predictive factors for major adverse cardiovascular events (MACEs) in compensated HCV-related cirrhosis were Asian ethnic origin, hypertension, smoking, and low serum albumin, according to a report in American Heart Journal.
A total of 878 patients with HCV-related cirrhosis were enrolled at 35 French centers. Upon enrollment, all patients received HCV treatment and were followed for MACEs, including stroke, myocardial infarction, ischemic heart disease, heart failure, peripheral arterial disease, cardiac arrest, and cardiovascular-related death, according to Patrice Cacoub, MD, of Sorbonne Universités, Paris, and his colleagues.
Five-year survival for patients presenting with a MACE was 60% vs. 88% in patients who did not have an event.
“[Our] results strengthen the systemic nature of HCV infection, a chronic disease in which cardiovascular risk must be carefully assessed. The decreased rate of MACEs after [sustained virological response] in this population should be taken into account to enable wider access to new [direct-acting antivirals]. Further studies are warranted to evaluate whether a similar benefit can be obtained in less severe patients, such as noncirrhotic HCV-infected patients,” the researchers concluded.
The authors reported having no conflicts of interest.
SOURCE: Cacoub, P et al. Am Heart J. 2018;198:4-17.
Hepatitis C viremic suppression was associated with a lower rate of cardiovascular events in patients with compensated HCV-related cirrhosis, compared with control patients who did not achieve a sustained virological response. In addition, predictive factors for major adverse cardiovascular events (MACEs) in compensated HCV-related cirrhosis were Asian ethnic origin, hypertension, smoking, and low serum albumin, according to a report in American Heart Journal.
A total of 878 patients with HCV-related cirrhosis were enrolled at 35 French centers. Upon enrollment, all patients received HCV treatment and were followed for MACEs, including stroke, myocardial infarction, ischemic heart disease, heart failure, peripheral arterial disease, cardiac arrest, and cardiovascular-related death, according to Patrice Cacoub, MD, of Sorbonne Universités, Paris, and his colleagues.
Five-year survival for patients presenting with a MACE was 60% vs. 88% in patients who did not have an event.
“[Our] results strengthen the systemic nature of HCV infection, a chronic disease in which cardiovascular risk must be carefully assessed. The decreased rate of MACEs after [sustained virological response] in this population should be taken into account to enable wider access to new [direct-acting antivirals]. Further studies are warranted to evaluate whether a similar benefit can be obtained in less severe patients, such as noncirrhotic HCV-infected patients,” the researchers concluded.
The authors reported having no conflicts of interest.
SOURCE: Cacoub, P et al. Am Heart J. 2018;198:4-17.
FROM AMERICAN HEART JOURNAL
Key clinical point: Predictive factors for MACE in compensated HCV-related cirrhosis were Asian ethnic origin, hypertension, smoking, and low serum albumin.
Major finding: Achieving viremic suppression was associated with a lower rate of cardiovascular events in patients with compensated HCV-related cirrhosis.
Study details: A study at 35 French centers of 878 patients with HCV-related cirrhosis.
Disclosures: The authors reported having no conflicts of interest.
Source: Cacoub, P et al. Am Heart J. 2018;198:4-17.