User login
NSAID use in diabetes may worsen risk for first HF hospitalization
suggests a prospective, controlled study.
Certain subgroups may account for much of the excess risk, the results suggest, including the very elderly, patients with uncontrolled diabetes, those prescribed an NSAID for the first time, and patients already taking both a renin-angiotensin system inhibitor (RASi) and a diuretic.
Such patients with a firm indication for NSAIDs potentially could “be the ones benefiting most from closer follow-up, reduced dosage, or other mitigation strategies,” Anders Holt, MD, said in an interview.
Dr. Holt, of Copenhagen University Hospital and Herlev-Gentofte Hospital in Hellerup, Denmark, is lead author on the analysis of Danish registry data published in the Journal of the American College of Cardiology. He presented essentially the same results in preliminary form at the 2022 annual congress of the European Society of Cardiology.
HF hospitalizations linked to NSAIDs, the published report notes, are often attributed to symptoms from temporary fluid overload, often without worsening cardiac function, that stem from the drugs’ renal effects.
“One could speculate,” Dr. Holt said, that such HF events might be less severe and even associated with better outcomes, compared with other forms of heart failure.
But the current analysis provides a hint to the contrary, he observed. The 5-year mortality was similar for patients with HF linked to NSAIDs and those with other forms of HF, “which could suggest that NSAID-associated heart failure is more than transient fluid overload.”
The drugs may promote HF through direct effects on the heart by any of several proposed mechanisms, including “induction of arrhythmias and heart fibrosis, vasoconstriction, subclinical inflammation, and blood pressure elevation,” Dr. Holt said.
The current study doesn’t determine whether NSAID-associated HF stems from transient fluid overload or direct cardiac effects, but it’s “most likely both.”
In other limitations, the analysis is unable to “reliably explore” whether promotion of HF is an NSAID class effect, a “clinically relevant” point given the drugs’ varying effects on cardiovascular risk, states an accompanying editorial. Nor was it able to determine whether the drugs exert a dose-response effect on HF risk, noted Hassan Khan, MD, PhD, Norton Healthcare, Louisville, Ky., and Setor K. Kunutsor, MD, PhD, University of Leicester (England).
Still, “given the well-established relationship between the use of NSAIDs and increased HF, these findings are not unexpected because type 2 diabetes is also a major risk factor for HF.”
But it may be “premature to issue guideline recommendations based on a single observational study,” the editorialists wrote. “Further robust clinical trial evidence is needed to replicate these results and investigate the relationship of the type and dose of NSAIDs with HF risk. However, it should be realized that short-term or long-term use of NSAIDs may be detrimental to cardiovascular health.”
The analysis covered 23,308 patients from throughout Denmark with a type 2 diabetes diagnosis and no HF history who experienced a first HF hospitalization; their age averaged 76 years and 39% were women.
They served as their own controls; their NSAID exposures at two 28-day periods preceding the HF event, the one immediately before and the other preceding it by 56 days, were compared as the index and control periods, respectively.
Exposure to NSAIDs was defined as obtaining a prescription for celecoxib, diclofenac, ibuprofen, or naproxen, “as these are NSAIDs used primarily in Denmark,” the report states.
The odds ratios for HF hospitalization associated with NSAID exposure within 28 days preceding the event were 1.43 (95% confidence interval, 1.27-1.63) overall, 1.41 (95% CI, 1.16-1.71) for an NSAID given on top of both RASi and diuretics, 1.68 (95% CI, 1.00-2.88) for patients with elevated hemoglobin A1c, 1.78 (95% CI, 1.39-2.28) for those 80 or older, and 2.71 (95% CI, 1.78-4.23) for those with prior NSAID use.
That NSAID use and diabetes are each associated with increased risk for HF is well established, Dr. Holt observed. Yet the drugs had been prescribed to 16% of patients in the study.
“One of the more surprising findings, to me, was the quite substantial use of prescribed NSAIDs in a population of patients with diabetes, a patient group with a well-established cardiovascular risk,” he said.
“This patient group is only growing, so emphasis on the possible associations between even short-term NSAID use and incident heart failure is probably timely and perhaps needed.”
Dr. Holt and the study were supported by grants from Ib Mogens Kristiansens Almene Fond, Helsefonden, Snedkermester Sophus Jacobsen og hustru Astrid Jacobsen Fond, Marie og M.B. Richters Fond, and the Dagmar Marshalls Fond. Dr. Khan and Dr. Kunutsor reported no relevant relationships.
A version of this article first appeared on Medscape.com.
suggests a prospective, controlled study.
Certain subgroups may account for much of the excess risk, the results suggest, including the very elderly, patients with uncontrolled diabetes, those prescribed an NSAID for the first time, and patients already taking both a renin-angiotensin system inhibitor (RASi) and a diuretic.
Such patients with a firm indication for NSAIDs potentially could “be the ones benefiting most from closer follow-up, reduced dosage, or other mitigation strategies,” Anders Holt, MD, said in an interview.
Dr. Holt, of Copenhagen University Hospital and Herlev-Gentofte Hospital in Hellerup, Denmark, is lead author on the analysis of Danish registry data published in the Journal of the American College of Cardiology. He presented essentially the same results in preliminary form at the 2022 annual congress of the European Society of Cardiology.
HF hospitalizations linked to NSAIDs, the published report notes, are often attributed to symptoms from temporary fluid overload, often without worsening cardiac function, that stem from the drugs’ renal effects.
“One could speculate,” Dr. Holt said, that such HF events might be less severe and even associated with better outcomes, compared with other forms of heart failure.
But the current analysis provides a hint to the contrary, he observed. The 5-year mortality was similar for patients with HF linked to NSAIDs and those with other forms of HF, “which could suggest that NSAID-associated heart failure is more than transient fluid overload.”
The drugs may promote HF through direct effects on the heart by any of several proposed mechanisms, including “induction of arrhythmias and heart fibrosis, vasoconstriction, subclinical inflammation, and blood pressure elevation,” Dr. Holt said.
The current study doesn’t determine whether NSAID-associated HF stems from transient fluid overload or direct cardiac effects, but it’s “most likely both.”
In other limitations, the analysis is unable to “reliably explore” whether promotion of HF is an NSAID class effect, a “clinically relevant” point given the drugs’ varying effects on cardiovascular risk, states an accompanying editorial. Nor was it able to determine whether the drugs exert a dose-response effect on HF risk, noted Hassan Khan, MD, PhD, Norton Healthcare, Louisville, Ky., and Setor K. Kunutsor, MD, PhD, University of Leicester (England).
Still, “given the well-established relationship between the use of NSAIDs and increased HF, these findings are not unexpected because type 2 diabetes is also a major risk factor for HF.”
But it may be “premature to issue guideline recommendations based on a single observational study,” the editorialists wrote. “Further robust clinical trial evidence is needed to replicate these results and investigate the relationship of the type and dose of NSAIDs with HF risk. However, it should be realized that short-term or long-term use of NSAIDs may be detrimental to cardiovascular health.”
The analysis covered 23,308 patients from throughout Denmark with a type 2 diabetes diagnosis and no HF history who experienced a first HF hospitalization; their age averaged 76 years and 39% were women.
They served as their own controls; their NSAID exposures at two 28-day periods preceding the HF event, the one immediately before and the other preceding it by 56 days, were compared as the index and control periods, respectively.
Exposure to NSAIDs was defined as obtaining a prescription for celecoxib, diclofenac, ibuprofen, or naproxen, “as these are NSAIDs used primarily in Denmark,” the report states.
The odds ratios for HF hospitalization associated with NSAID exposure within 28 days preceding the event were 1.43 (95% confidence interval, 1.27-1.63) overall, 1.41 (95% CI, 1.16-1.71) for an NSAID given on top of both RASi and diuretics, 1.68 (95% CI, 1.00-2.88) for patients with elevated hemoglobin A1c, 1.78 (95% CI, 1.39-2.28) for those 80 or older, and 2.71 (95% CI, 1.78-4.23) for those with prior NSAID use.
That NSAID use and diabetes are each associated with increased risk for HF is well established, Dr. Holt observed. Yet the drugs had been prescribed to 16% of patients in the study.
“One of the more surprising findings, to me, was the quite substantial use of prescribed NSAIDs in a population of patients with diabetes, a patient group with a well-established cardiovascular risk,” he said.
“This patient group is only growing, so emphasis on the possible associations between even short-term NSAID use and incident heart failure is probably timely and perhaps needed.”
Dr. Holt and the study were supported by grants from Ib Mogens Kristiansens Almene Fond, Helsefonden, Snedkermester Sophus Jacobsen og hustru Astrid Jacobsen Fond, Marie og M.B. Richters Fond, and the Dagmar Marshalls Fond. Dr. Khan and Dr. Kunutsor reported no relevant relationships.
A version of this article first appeared on Medscape.com.
suggests a prospective, controlled study.
Certain subgroups may account for much of the excess risk, the results suggest, including the very elderly, patients with uncontrolled diabetes, those prescribed an NSAID for the first time, and patients already taking both a renin-angiotensin system inhibitor (RASi) and a diuretic.
Such patients with a firm indication for NSAIDs potentially could “be the ones benefiting most from closer follow-up, reduced dosage, or other mitigation strategies,” Anders Holt, MD, said in an interview.
Dr. Holt, of Copenhagen University Hospital and Herlev-Gentofte Hospital in Hellerup, Denmark, is lead author on the analysis of Danish registry data published in the Journal of the American College of Cardiology. He presented essentially the same results in preliminary form at the 2022 annual congress of the European Society of Cardiology.
HF hospitalizations linked to NSAIDs, the published report notes, are often attributed to symptoms from temporary fluid overload, often without worsening cardiac function, that stem from the drugs’ renal effects.
“One could speculate,” Dr. Holt said, that such HF events might be less severe and even associated with better outcomes, compared with other forms of heart failure.
But the current analysis provides a hint to the contrary, he observed. The 5-year mortality was similar for patients with HF linked to NSAIDs and those with other forms of HF, “which could suggest that NSAID-associated heart failure is more than transient fluid overload.”
The drugs may promote HF through direct effects on the heart by any of several proposed mechanisms, including “induction of arrhythmias and heart fibrosis, vasoconstriction, subclinical inflammation, and blood pressure elevation,” Dr. Holt said.
The current study doesn’t determine whether NSAID-associated HF stems from transient fluid overload or direct cardiac effects, but it’s “most likely both.”
In other limitations, the analysis is unable to “reliably explore” whether promotion of HF is an NSAID class effect, a “clinically relevant” point given the drugs’ varying effects on cardiovascular risk, states an accompanying editorial. Nor was it able to determine whether the drugs exert a dose-response effect on HF risk, noted Hassan Khan, MD, PhD, Norton Healthcare, Louisville, Ky., and Setor K. Kunutsor, MD, PhD, University of Leicester (England).
Still, “given the well-established relationship between the use of NSAIDs and increased HF, these findings are not unexpected because type 2 diabetes is also a major risk factor for HF.”
But it may be “premature to issue guideline recommendations based on a single observational study,” the editorialists wrote. “Further robust clinical trial evidence is needed to replicate these results and investigate the relationship of the type and dose of NSAIDs with HF risk. However, it should be realized that short-term or long-term use of NSAIDs may be detrimental to cardiovascular health.”
The analysis covered 23,308 patients from throughout Denmark with a type 2 diabetes diagnosis and no HF history who experienced a first HF hospitalization; their age averaged 76 years and 39% were women.
They served as their own controls; their NSAID exposures at two 28-day periods preceding the HF event, the one immediately before and the other preceding it by 56 days, were compared as the index and control periods, respectively.
Exposure to NSAIDs was defined as obtaining a prescription for celecoxib, diclofenac, ibuprofen, or naproxen, “as these are NSAIDs used primarily in Denmark,” the report states.
The odds ratios for HF hospitalization associated with NSAID exposure within 28 days preceding the event were 1.43 (95% confidence interval, 1.27-1.63) overall, 1.41 (95% CI, 1.16-1.71) for an NSAID given on top of both RASi and diuretics, 1.68 (95% CI, 1.00-2.88) for patients with elevated hemoglobin A1c, 1.78 (95% CI, 1.39-2.28) for those 80 or older, and 2.71 (95% CI, 1.78-4.23) for those with prior NSAID use.
That NSAID use and diabetes are each associated with increased risk for HF is well established, Dr. Holt observed. Yet the drugs had been prescribed to 16% of patients in the study.
“One of the more surprising findings, to me, was the quite substantial use of prescribed NSAIDs in a population of patients with diabetes, a patient group with a well-established cardiovascular risk,” he said.
“This patient group is only growing, so emphasis on the possible associations between even short-term NSAID use and incident heart failure is probably timely and perhaps needed.”
Dr. Holt and the study were supported by grants from Ib Mogens Kristiansens Almene Fond, Helsefonden, Snedkermester Sophus Jacobsen og hustru Astrid Jacobsen Fond, Marie og M.B. Richters Fond, and the Dagmar Marshalls Fond. Dr. Khan and Dr. Kunutsor reported no relevant relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Study offers dozens of reasons to cut sugar
A new compilation of nearly all research to date on the health impacts of sugar offers dozens of reasons to cut back.
The studies accounted for decades of research on the topic, stretching back to the beginning of the largest electronic databases for scientific papers.
The result is a list that cites the world’s most common health problems like heart disease, diabetes, obesity, high blood pressure, heart attack, high cholesterol, cancer, and depression. The findings were published in the BMJ. Researchers looked at studies that evaluated the impacts of consuming free sugars, which means any food that contains processed or naturally occurring sugars like table sugar, honey, or maple syrup. Sugar found in whole fruits and vegetables and in milk is not free sugar.
U.S. dietary guidelines recommend getting no more than 10% of daily calories from added sugars. For a typical 2,000-calorie-per-day diet, that equals no more than 200 calories, or about 12 teaspoons. The CDC reports that the average person consumes 17 teaspoons per day, with the largest sources being sugar-sweetened beverages, desserts, and snacks. (For context: one 12-ounce can of soda contains the equivalent of 9 teaspoons of sugar, according to beverage maker Coca-Cola.)
The new analysis also found links between sugary beverage consumption and other diet and lifestyle characteristics that may contribute to health problems.
“People who consumed sugar-sweetened beverages more frequently were likely to ingest more total and saturated fat, carbohydrate, and sodium, and less fruit, fiber, dairy products, and whole grain foods,” the authors wrote. “This dietary pattern was also associated with more frequent smoking and drinking, lower physical activity levels, and more time spent watching television. Therefore, the role of these confounding factors should be taken into consideration when explaining the association between sugar consumption and burden of disease.”
Recommendations for limiting sugar consumption are in place worldwide, the authors noted. They concluded that more needs to be done given the known health dangers of sugar.
“To change sugar consumption patterns, especially for children and adolescents, a combination of widespread public health education and policies worldwide is urgently needed,” they said.
A version of this article first appeared on WebMD.com.
A new compilation of nearly all research to date on the health impacts of sugar offers dozens of reasons to cut back.
The studies accounted for decades of research on the topic, stretching back to the beginning of the largest electronic databases for scientific papers.
The result is a list that cites the world’s most common health problems like heart disease, diabetes, obesity, high blood pressure, heart attack, high cholesterol, cancer, and depression. The findings were published in the BMJ. Researchers looked at studies that evaluated the impacts of consuming free sugars, which means any food that contains processed or naturally occurring sugars like table sugar, honey, or maple syrup. Sugar found in whole fruits and vegetables and in milk is not free sugar.
U.S. dietary guidelines recommend getting no more than 10% of daily calories from added sugars. For a typical 2,000-calorie-per-day diet, that equals no more than 200 calories, or about 12 teaspoons. The CDC reports that the average person consumes 17 teaspoons per day, with the largest sources being sugar-sweetened beverages, desserts, and snacks. (For context: one 12-ounce can of soda contains the equivalent of 9 teaspoons of sugar, according to beverage maker Coca-Cola.)
The new analysis also found links between sugary beverage consumption and other diet and lifestyle characteristics that may contribute to health problems.
“People who consumed sugar-sweetened beverages more frequently were likely to ingest more total and saturated fat, carbohydrate, and sodium, and less fruit, fiber, dairy products, and whole grain foods,” the authors wrote. “This dietary pattern was also associated with more frequent smoking and drinking, lower physical activity levels, and more time spent watching television. Therefore, the role of these confounding factors should be taken into consideration when explaining the association between sugar consumption and burden of disease.”
Recommendations for limiting sugar consumption are in place worldwide, the authors noted. They concluded that more needs to be done given the known health dangers of sugar.
“To change sugar consumption patterns, especially for children and adolescents, a combination of widespread public health education and policies worldwide is urgently needed,” they said.
A version of this article first appeared on WebMD.com.
A new compilation of nearly all research to date on the health impacts of sugar offers dozens of reasons to cut back.
The studies accounted for decades of research on the topic, stretching back to the beginning of the largest electronic databases for scientific papers.
The result is a list that cites the world’s most common health problems like heart disease, diabetes, obesity, high blood pressure, heart attack, high cholesterol, cancer, and depression. The findings were published in the BMJ. Researchers looked at studies that evaluated the impacts of consuming free sugars, which means any food that contains processed or naturally occurring sugars like table sugar, honey, or maple syrup. Sugar found in whole fruits and vegetables and in milk is not free sugar.
U.S. dietary guidelines recommend getting no more than 10% of daily calories from added sugars. For a typical 2,000-calorie-per-day diet, that equals no more than 200 calories, or about 12 teaspoons. The CDC reports that the average person consumes 17 teaspoons per day, with the largest sources being sugar-sweetened beverages, desserts, and snacks. (For context: one 12-ounce can of soda contains the equivalent of 9 teaspoons of sugar, according to beverage maker Coca-Cola.)
The new analysis also found links between sugary beverage consumption and other diet and lifestyle characteristics that may contribute to health problems.
“People who consumed sugar-sweetened beverages more frequently were likely to ingest more total and saturated fat, carbohydrate, and sodium, and less fruit, fiber, dairy products, and whole grain foods,” the authors wrote. “This dietary pattern was also associated with more frequent smoking and drinking, lower physical activity levels, and more time spent watching television. Therefore, the role of these confounding factors should be taken into consideration when explaining the association between sugar consumption and burden of disease.”
Recommendations for limiting sugar consumption are in place worldwide, the authors noted. They concluded that more needs to be done given the known health dangers of sugar.
“To change sugar consumption patterns, especially for children and adolescents, a combination of widespread public health education and policies worldwide is urgently needed,” they said.
A version of this article first appeared on WebMD.com.
FROM THE BMJ
Antiphospholipid antibodies linked to future CV events
The presence of antiphospholipid antibodies is associated with an increased risk for future cardiovascular events, according to a new study.
The findings point to possible new approaches to risk stratification and the potential for new therapeutic targets in heart disease.
“In this study of the general population, we found that two antiphospholipid antibodies were associated with an increased risk of having a serious cardiovascular event over a follow-up of 8 years,” coauthor Jason Knight, MD, University of Michigan, Ann Arbor, said in an interview.
“If confirmed in further studies, these findings could be used to identify a subgroup of patients who need more careful monitoring and more aggressive risk-factor modification, and if the increased risk linked to these antibodies is high enough, it may also justify preemptive treatments such as the anticoagulants that are routinely used in antiphospholipid syndrome,” Dr. Knight said.
“The long-term vision is that we may identify some people in the general population who would benefit from treating the immune system for the prevention and treatment of cardiovascular disease instead of, or in addition to, using typical cardiovascular medications,” he added.
The study was published online in JAMA Network Open.
Individuals with autoimmune and inflammatory diseases have a greater risk for cardiovascular events than expected based on traditional cardiovascular risk factors, with mechanisms proposed to explain this risk including inflammation-mediated disruption of vascular integrity and activation of platelets and coagulation pathways, the authors explained. However, the role of autoantibodies remains unclear.
They noted that antiphospholipid antibodies can activate endothelial cells, platelets, and neutrophils, and some patients with persistently circulating antiphospholipid antibodies can develop antiphospholipid syndrome – an acquired thromboinflammatory disease characterized by arterial, venous, and microvascular thrombotic events and obstetric complications.
Cross-sectional studies have shown that antiphospholipid antibodies are acutely present in up to 17.4% of patients with stroke or transient ischemic attack, and small cohort studies have suggested that such antibodies may be present in 1%-12% of seemingly healthy individuals. However, the impact of sex, race, and ethnicity on the prevalence of antiphospholipid antibodies and their association with atherosclerotic cardiovascular disease is not known.
The researchers conducted the current study to look at the association between antiphospholipid antibodies and future risk for atherosclerotic cardiovascular events.
They analyzed data from 2,427 participants in the population-based Dallas Heart Study who had no history of atherosclerotic cardiovascular disease or autoimmune diseases requiring immunosuppressive medications at the time of blood sampling at study entry in 2007-2009.
Eight different types of antiphospholipid antibodies were measured, and data on cardiovascular events over the next 8 years was recorded.
Results showed that 14.5% of the cohort tested positive for one of these antiphospholipid antibodies at the start of the study, with approximately one-third of those detected at a moderate or high titer.
The researchers also found that the IgA isotypes of two antiphospholipid antibodies – anticardiolipin and anti-beta-2 glycoprotein – were associated with future atherosclerotic cardiovascular events.
After adjustment for other known risk factors, individuals testing positive for the IgA isotype of anticardiolipin had an almost five times increased risk (hazard ratio, 4.92) of the primary endpoint (myocardial infarction, stroke, coronary revascularization, or cardiovascular death); while those testing positive for anti–beta2-glycoprotein had an almost three times increased risk (HR, 2.91).
Furthermore, there was what appeared to be a dose effect. People with the highest levels of these antibodies also had the highest risk for cardiovascular events, with up to an almost 10-fold increased risk with the higher level of anticardiolipin.
Dr. Knight said that more research into the IgA isotypes of these antiphospholipid antibodies is needed.
“Most of the mechanistic work in the antiphospholipid syndrome field has focused on IgG antiphospholipid antibodies. While we commonly find these IgA antibodies in patients with APS, the extent to which they contribute to disease has not been firmly established,” he said. “The fact that IgA was the primary hit in our unbiased screen suggests that there is more to the story and we need to better understand the implications of having these antibodies in circulation, and what specific problems they may be causing.”
Noting that antiphospholipid antibodies can form transiently after certain situations, such as infections, Dr. Knight said that further studies were needed with repeat blood testing to detect the chronic presence of the antibodies.
He added that information of venous thromboses was not available in this study and “perhaps some of the other antibodies might have stood out if we were able to analyze for different outcomes.”
This study was supported by a Pfizer Aspire Award. Dr. Knight reported receiving research funding and consulting fees from Jazz Pharmaceuticals outside the submitted work.
A version of this article first appeared on Medscape.com.
The presence of antiphospholipid antibodies is associated with an increased risk for future cardiovascular events, according to a new study.
The findings point to possible new approaches to risk stratification and the potential for new therapeutic targets in heart disease.
“In this study of the general population, we found that two antiphospholipid antibodies were associated with an increased risk of having a serious cardiovascular event over a follow-up of 8 years,” coauthor Jason Knight, MD, University of Michigan, Ann Arbor, said in an interview.
“If confirmed in further studies, these findings could be used to identify a subgroup of patients who need more careful monitoring and more aggressive risk-factor modification, and if the increased risk linked to these antibodies is high enough, it may also justify preemptive treatments such as the anticoagulants that are routinely used in antiphospholipid syndrome,” Dr. Knight said.
“The long-term vision is that we may identify some people in the general population who would benefit from treating the immune system for the prevention and treatment of cardiovascular disease instead of, or in addition to, using typical cardiovascular medications,” he added.
The study was published online in JAMA Network Open.
Individuals with autoimmune and inflammatory diseases have a greater risk for cardiovascular events than expected based on traditional cardiovascular risk factors, with mechanisms proposed to explain this risk including inflammation-mediated disruption of vascular integrity and activation of platelets and coagulation pathways, the authors explained. However, the role of autoantibodies remains unclear.
They noted that antiphospholipid antibodies can activate endothelial cells, platelets, and neutrophils, and some patients with persistently circulating antiphospholipid antibodies can develop antiphospholipid syndrome – an acquired thromboinflammatory disease characterized by arterial, venous, and microvascular thrombotic events and obstetric complications.
Cross-sectional studies have shown that antiphospholipid antibodies are acutely present in up to 17.4% of patients with stroke or transient ischemic attack, and small cohort studies have suggested that such antibodies may be present in 1%-12% of seemingly healthy individuals. However, the impact of sex, race, and ethnicity on the prevalence of antiphospholipid antibodies and their association with atherosclerotic cardiovascular disease is not known.
The researchers conducted the current study to look at the association between antiphospholipid antibodies and future risk for atherosclerotic cardiovascular events.
They analyzed data from 2,427 participants in the population-based Dallas Heart Study who had no history of atherosclerotic cardiovascular disease or autoimmune diseases requiring immunosuppressive medications at the time of blood sampling at study entry in 2007-2009.
Eight different types of antiphospholipid antibodies were measured, and data on cardiovascular events over the next 8 years was recorded.
Results showed that 14.5% of the cohort tested positive for one of these antiphospholipid antibodies at the start of the study, with approximately one-third of those detected at a moderate or high titer.
The researchers also found that the IgA isotypes of two antiphospholipid antibodies – anticardiolipin and anti-beta-2 glycoprotein – were associated with future atherosclerotic cardiovascular events.
After adjustment for other known risk factors, individuals testing positive for the IgA isotype of anticardiolipin had an almost five times increased risk (hazard ratio, 4.92) of the primary endpoint (myocardial infarction, stroke, coronary revascularization, or cardiovascular death); while those testing positive for anti–beta2-glycoprotein had an almost three times increased risk (HR, 2.91).
Furthermore, there was what appeared to be a dose effect. People with the highest levels of these antibodies also had the highest risk for cardiovascular events, with up to an almost 10-fold increased risk with the higher level of anticardiolipin.
Dr. Knight said that more research into the IgA isotypes of these antiphospholipid antibodies is needed.
“Most of the mechanistic work in the antiphospholipid syndrome field has focused on IgG antiphospholipid antibodies. While we commonly find these IgA antibodies in patients with APS, the extent to which they contribute to disease has not been firmly established,” he said. “The fact that IgA was the primary hit in our unbiased screen suggests that there is more to the story and we need to better understand the implications of having these antibodies in circulation, and what specific problems they may be causing.”
Noting that antiphospholipid antibodies can form transiently after certain situations, such as infections, Dr. Knight said that further studies were needed with repeat blood testing to detect the chronic presence of the antibodies.
He added that information of venous thromboses was not available in this study and “perhaps some of the other antibodies might have stood out if we were able to analyze for different outcomes.”
This study was supported by a Pfizer Aspire Award. Dr. Knight reported receiving research funding and consulting fees from Jazz Pharmaceuticals outside the submitted work.
A version of this article first appeared on Medscape.com.
The presence of antiphospholipid antibodies is associated with an increased risk for future cardiovascular events, according to a new study.
The findings point to possible new approaches to risk stratification and the potential for new therapeutic targets in heart disease.
“In this study of the general population, we found that two antiphospholipid antibodies were associated with an increased risk of having a serious cardiovascular event over a follow-up of 8 years,” coauthor Jason Knight, MD, University of Michigan, Ann Arbor, said in an interview.
“If confirmed in further studies, these findings could be used to identify a subgroup of patients who need more careful monitoring and more aggressive risk-factor modification, and if the increased risk linked to these antibodies is high enough, it may also justify preemptive treatments such as the anticoagulants that are routinely used in antiphospholipid syndrome,” Dr. Knight said.
“The long-term vision is that we may identify some people in the general population who would benefit from treating the immune system for the prevention and treatment of cardiovascular disease instead of, or in addition to, using typical cardiovascular medications,” he added.
The study was published online in JAMA Network Open.
Individuals with autoimmune and inflammatory diseases have a greater risk for cardiovascular events than expected based on traditional cardiovascular risk factors, with mechanisms proposed to explain this risk including inflammation-mediated disruption of vascular integrity and activation of platelets and coagulation pathways, the authors explained. However, the role of autoantibodies remains unclear.
They noted that antiphospholipid antibodies can activate endothelial cells, platelets, and neutrophils, and some patients with persistently circulating antiphospholipid antibodies can develop antiphospholipid syndrome – an acquired thromboinflammatory disease characterized by arterial, venous, and microvascular thrombotic events and obstetric complications.
Cross-sectional studies have shown that antiphospholipid antibodies are acutely present in up to 17.4% of patients with stroke or transient ischemic attack, and small cohort studies have suggested that such antibodies may be present in 1%-12% of seemingly healthy individuals. However, the impact of sex, race, and ethnicity on the prevalence of antiphospholipid antibodies and their association with atherosclerotic cardiovascular disease is not known.
The researchers conducted the current study to look at the association between antiphospholipid antibodies and future risk for atherosclerotic cardiovascular events.
They analyzed data from 2,427 participants in the population-based Dallas Heart Study who had no history of atherosclerotic cardiovascular disease or autoimmune diseases requiring immunosuppressive medications at the time of blood sampling at study entry in 2007-2009.
Eight different types of antiphospholipid antibodies were measured, and data on cardiovascular events over the next 8 years was recorded.
Results showed that 14.5% of the cohort tested positive for one of these antiphospholipid antibodies at the start of the study, with approximately one-third of those detected at a moderate or high titer.
The researchers also found that the IgA isotypes of two antiphospholipid antibodies – anticardiolipin and anti-beta-2 glycoprotein – were associated with future atherosclerotic cardiovascular events.
After adjustment for other known risk factors, individuals testing positive for the IgA isotype of anticardiolipin had an almost five times increased risk (hazard ratio, 4.92) of the primary endpoint (myocardial infarction, stroke, coronary revascularization, or cardiovascular death); while those testing positive for anti–beta2-glycoprotein had an almost three times increased risk (HR, 2.91).
Furthermore, there was what appeared to be a dose effect. People with the highest levels of these antibodies also had the highest risk for cardiovascular events, with up to an almost 10-fold increased risk with the higher level of anticardiolipin.
Dr. Knight said that more research into the IgA isotypes of these antiphospholipid antibodies is needed.
“Most of the mechanistic work in the antiphospholipid syndrome field has focused on IgG antiphospholipid antibodies. While we commonly find these IgA antibodies in patients with APS, the extent to which they contribute to disease has not been firmly established,” he said. “The fact that IgA was the primary hit in our unbiased screen suggests that there is more to the story and we need to better understand the implications of having these antibodies in circulation, and what specific problems they may be causing.”
Noting that antiphospholipid antibodies can form transiently after certain situations, such as infections, Dr. Knight said that further studies were needed with repeat blood testing to detect the chronic presence of the antibodies.
He added that information of venous thromboses was not available in this study and “perhaps some of the other antibodies might have stood out if we were able to analyze for different outcomes.”
This study was supported by a Pfizer Aspire Award. Dr. Knight reported receiving research funding and consulting fees from Jazz Pharmaceuticals outside the submitted work.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
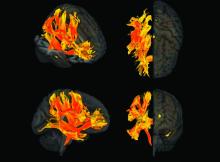
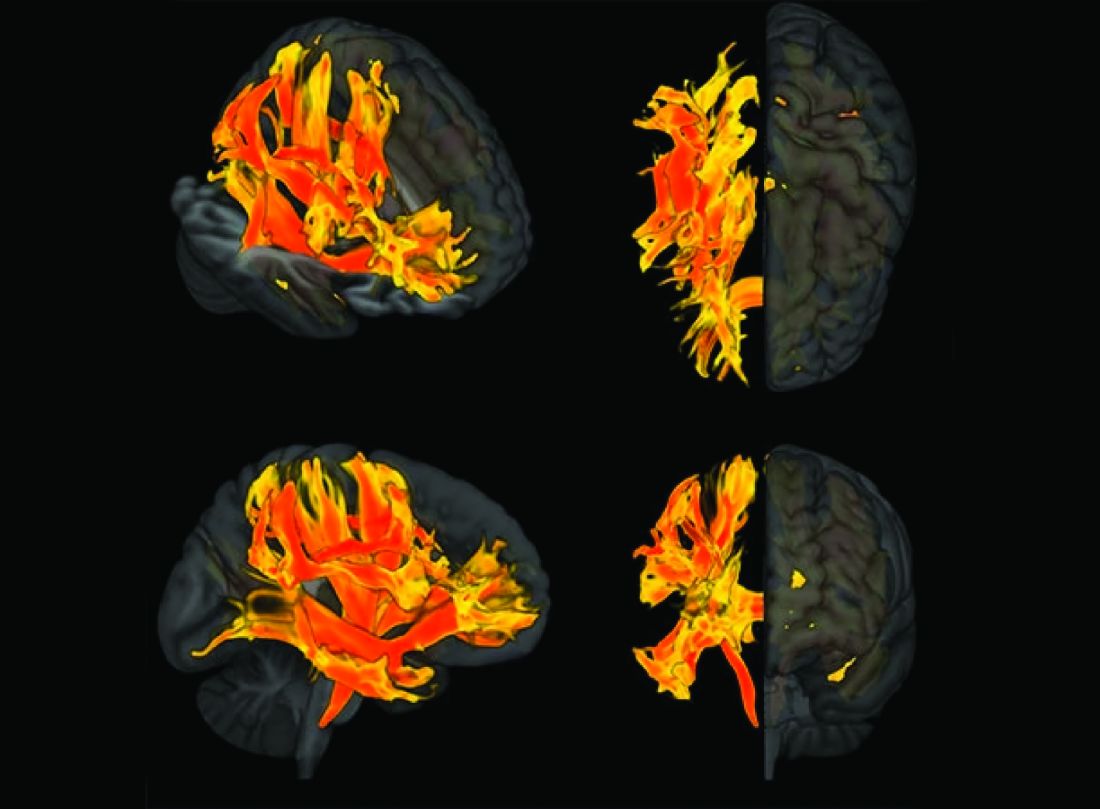
Spherical heart may predict cardiomyopathy, AFib
A round heart, or left ventricle sphericity, predicted cardiomyopathy and atrial fibrillation (AFib) in a deep learning analysis of MRI images from close to 39,000 participants in the UK Biobank, a new study shows.
An increase of 1 standard deviation in the sphericity index (short axis length/long axis length) was associated with a 47% increased incidence of cardiomyopathy and a 20% increased incidence of AFib, independent of clinical factors and traditional MRI measures.
Furthermore, a genetic analysis suggested a shared architecture between sphericity and nonischemic cardiomyopathy, pointing to NICM as a possible causal factor for left ventricle sphericity among individuals with normal LV size and function.
“Physicians have known the heart gets rounder after heart attacks and as we get older,” David Ouyang, MD, a cardiologist in the Smidt Heart Institute at Cedars-Sinai Medical Center, Los Angeles, and a researcher in the division of artificial intelligence in medicine, said in an interview. “We wanted to see if this sphericity is prognostic of future disease even in healthy individuals.”
Although it is too early to recommend heart shape assessment in healthy asymptomatic people, he said, “physicians should be extra careful and think about treatments when they notice a patient’s heart is particularly round.”
The study was published online March 29 in the journal Med.
Sphericity index key
The investigators hypothesized that there is variation in LV sphericity within the spectrum of normal LV chamber size and systolic function, and that such variation might be a marker of cardiac risk with genetic influences.
To test this hypothesis, they used automated deep-learning segmentation of cardiac MRI data to estimate and analyze the sphericity index in a cohort of 38,897 individuals participating in the UK Biobank.
After adjustment for age at MRI and sex, an increased sphericity index was associated with an increased risk for cardiomyopathy (hazard ratio, 1.57), AFib (HR, 1.35), and heart failure (HR, 1.37).
No significant association was seen with cardiac arrest.
The team then stratified the cohort into quintiles and compared the top 20%, middle 60%, and bottom 20%. The relationship between the sphericity index and risk extended across the distribution; individuals with higher than median sphericity had increased disease incidence, and those with lower than median sphericity had decreased incidence.
Overall, a single standard deviation in the sphericity index was associated with increased risk of cardiomyopathy (HR, 1.47) and of AFib (HR, 1.20), independent of clinical factors and usual MRI measurements.
In a minimally adjusted model, the sphericity index was a predictor of incident cardiomyopathy, AFib, and heart failure.
Adjustment for clinical factors partially attenuated the heart failure association; additional adjustment for MRI measurements fully attenuated that association and partially attenuated the association with AFib.
However, in all adjusted models, the association with cardiomyopathy showed little attenuation.
Furthermore, the team identified four loci associated with sphericity at genomewide significance – PLN, ANGPT1, PDZRN3, and HLA DR/DQ – and Mendelian randomization supported NICM as a cause of LV sphericity.
Looking ahead
“While conventional imaging metrics have significant diagnostic and prognostic value, some of these measurements have been adopted out of convenience or tradition,” the authors noted. “By representing a specific multidimensional remodeling phenotype, sphericity has emerged as a distinct morphologic trait with features not adequately captured by conventional measurements.
“We expect that the search space of potential imaging measurements is vast, and we have only begun to scratch at the surface of disease associations.”
Indeed, Dr. Ouyang said his group is “trying to evaluate the sphericity in echocardiograms or heart ultrasounds, which are more common and cheaper than MRI.”
“The main caveat is translating the information directly to patient care,” Richard C. Becker, MD, director and physician-in-chief of the University of Cincinnati Heart, Lung, and Vascular Institute, said in an interview. “Near-term yield could include using the spherical calculation in routine MRI of the heart, and based on the findings, following patients more closely if there is an abnormal shape. Or performing an MRI and targeted gene testing if there is a family history of cardiomyopathy or [of] an abnormal shape of the heart.”
“Validation of the findings and large-scale evaluation of the genes identified, and how they interact with patient and environmental factors, will be very important,” he added.
Nevertheless, “the study was well done and may serve as a foundation for future research,” Dr. Becker said. “The investigators used several powerful tools, including MRI, genomics, and [artificial intelligence] to draw their conclusions. This is precisely the way that ‘big data’ should be used – in a complementary fashion.”
The study authors and Dr. Becker reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A round heart, or left ventricle sphericity, predicted cardiomyopathy and atrial fibrillation (AFib) in a deep learning analysis of MRI images from close to 39,000 participants in the UK Biobank, a new study shows.
An increase of 1 standard deviation in the sphericity index (short axis length/long axis length) was associated with a 47% increased incidence of cardiomyopathy and a 20% increased incidence of AFib, independent of clinical factors and traditional MRI measures.
Furthermore, a genetic analysis suggested a shared architecture between sphericity and nonischemic cardiomyopathy, pointing to NICM as a possible causal factor for left ventricle sphericity among individuals with normal LV size and function.
“Physicians have known the heart gets rounder after heart attacks and as we get older,” David Ouyang, MD, a cardiologist in the Smidt Heart Institute at Cedars-Sinai Medical Center, Los Angeles, and a researcher in the division of artificial intelligence in medicine, said in an interview. “We wanted to see if this sphericity is prognostic of future disease even in healthy individuals.”
Although it is too early to recommend heart shape assessment in healthy asymptomatic people, he said, “physicians should be extra careful and think about treatments when they notice a patient’s heart is particularly round.”
The study was published online March 29 in the journal Med.
Sphericity index key
The investigators hypothesized that there is variation in LV sphericity within the spectrum of normal LV chamber size and systolic function, and that such variation might be a marker of cardiac risk with genetic influences.
To test this hypothesis, they used automated deep-learning segmentation of cardiac MRI data to estimate and analyze the sphericity index in a cohort of 38,897 individuals participating in the UK Biobank.
After adjustment for age at MRI and sex, an increased sphericity index was associated with an increased risk for cardiomyopathy (hazard ratio, 1.57), AFib (HR, 1.35), and heart failure (HR, 1.37).
No significant association was seen with cardiac arrest.
The team then stratified the cohort into quintiles and compared the top 20%, middle 60%, and bottom 20%. The relationship between the sphericity index and risk extended across the distribution; individuals with higher than median sphericity had increased disease incidence, and those with lower than median sphericity had decreased incidence.
Overall, a single standard deviation in the sphericity index was associated with increased risk of cardiomyopathy (HR, 1.47) and of AFib (HR, 1.20), independent of clinical factors and usual MRI measurements.
In a minimally adjusted model, the sphericity index was a predictor of incident cardiomyopathy, AFib, and heart failure.
Adjustment for clinical factors partially attenuated the heart failure association; additional adjustment for MRI measurements fully attenuated that association and partially attenuated the association with AFib.
However, in all adjusted models, the association with cardiomyopathy showed little attenuation.
Furthermore, the team identified four loci associated with sphericity at genomewide significance – PLN, ANGPT1, PDZRN3, and HLA DR/DQ – and Mendelian randomization supported NICM as a cause of LV sphericity.
Looking ahead
“While conventional imaging metrics have significant diagnostic and prognostic value, some of these measurements have been adopted out of convenience or tradition,” the authors noted. “By representing a specific multidimensional remodeling phenotype, sphericity has emerged as a distinct morphologic trait with features not adequately captured by conventional measurements.
“We expect that the search space of potential imaging measurements is vast, and we have only begun to scratch at the surface of disease associations.”
Indeed, Dr. Ouyang said his group is “trying to evaluate the sphericity in echocardiograms or heart ultrasounds, which are more common and cheaper than MRI.”
“The main caveat is translating the information directly to patient care,” Richard C. Becker, MD, director and physician-in-chief of the University of Cincinnati Heart, Lung, and Vascular Institute, said in an interview. “Near-term yield could include using the spherical calculation in routine MRI of the heart, and based on the findings, following patients more closely if there is an abnormal shape. Or performing an MRI and targeted gene testing if there is a family history of cardiomyopathy or [of] an abnormal shape of the heart.”
“Validation of the findings and large-scale evaluation of the genes identified, and how they interact with patient and environmental factors, will be very important,” he added.
Nevertheless, “the study was well done and may serve as a foundation for future research,” Dr. Becker said. “The investigators used several powerful tools, including MRI, genomics, and [artificial intelligence] to draw their conclusions. This is precisely the way that ‘big data’ should be used – in a complementary fashion.”
The study authors and Dr. Becker reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A round heart, or left ventricle sphericity, predicted cardiomyopathy and atrial fibrillation (AFib) in a deep learning analysis of MRI images from close to 39,000 participants in the UK Biobank, a new study shows.
An increase of 1 standard deviation in the sphericity index (short axis length/long axis length) was associated with a 47% increased incidence of cardiomyopathy and a 20% increased incidence of AFib, independent of clinical factors and traditional MRI measures.
Furthermore, a genetic analysis suggested a shared architecture between sphericity and nonischemic cardiomyopathy, pointing to NICM as a possible causal factor for left ventricle sphericity among individuals with normal LV size and function.
“Physicians have known the heart gets rounder after heart attacks and as we get older,” David Ouyang, MD, a cardiologist in the Smidt Heart Institute at Cedars-Sinai Medical Center, Los Angeles, and a researcher in the division of artificial intelligence in medicine, said in an interview. “We wanted to see if this sphericity is prognostic of future disease even in healthy individuals.”
Although it is too early to recommend heart shape assessment in healthy asymptomatic people, he said, “physicians should be extra careful and think about treatments when they notice a patient’s heart is particularly round.”
The study was published online March 29 in the journal Med.
Sphericity index key
The investigators hypothesized that there is variation in LV sphericity within the spectrum of normal LV chamber size and systolic function, and that such variation might be a marker of cardiac risk with genetic influences.
To test this hypothesis, they used automated deep-learning segmentation of cardiac MRI data to estimate and analyze the sphericity index in a cohort of 38,897 individuals participating in the UK Biobank.
After adjustment for age at MRI and sex, an increased sphericity index was associated with an increased risk for cardiomyopathy (hazard ratio, 1.57), AFib (HR, 1.35), and heart failure (HR, 1.37).
No significant association was seen with cardiac arrest.
The team then stratified the cohort into quintiles and compared the top 20%, middle 60%, and bottom 20%. The relationship between the sphericity index and risk extended across the distribution; individuals with higher than median sphericity had increased disease incidence, and those with lower than median sphericity had decreased incidence.
Overall, a single standard deviation in the sphericity index was associated with increased risk of cardiomyopathy (HR, 1.47) and of AFib (HR, 1.20), independent of clinical factors and usual MRI measurements.
In a minimally adjusted model, the sphericity index was a predictor of incident cardiomyopathy, AFib, and heart failure.
Adjustment for clinical factors partially attenuated the heart failure association; additional adjustment for MRI measurements fully attenuated that association and partially attenuated the association with AFib.
However, in all adjusted models, the association with cardiomyopathy showed little attenuation.
Furthermore, the team identified four loci associated with sphericity at genomewide significance – PLN, ANGPT1, PDZRN3, and HLA DR/DQ – and Mendelian randomization supported NICM as a cause of LV sphericity.
Looking ahead
“While conventional imaging metrics have significant diagnostic and prognostic value, some of these measurements have been adopted out of convenience or tradition,” the authors noted. “By representing a specific multidimensional remodeling phenotype, sphericity has emerged as a distinct morphologic trait with features not adequately captured by conventional measurements.
“We expect that the search space of potential imaging measurements is vast, and we have only begun to scratch at the surface of disease associations.”
Indeed, Dr. Ouyang said his group is “trying to evaluate the sphericity in echocardiograms or heart ultrasounds, which are more common and cheaper than MRI.”
“The main caveat is translating the information directly to patient care,” Richard C. Becker, MD, director and physician-in-chief of the University of Cincinnati Heart, Lung, and Vascular Institute, said in an interview. “Near-term yield could include using the spherical calculation in routine MRI of the heart, and based on the findings, following patients more closely if there is an abnormal shape. Or performing an MRI and targeted gene testing if there is a family history of cardiomyopathy or [of] an abnormal shape of the heart.”
“Validation of the findings and large-scale evaluation of the genes identified, and how they interact with patient and environmental factors, will be very important,” he added.
Nevertheless, “the study was well done and may serve as a foundation for future research,” Dr. Becker said. “The investigators used several powerful tools, including MRI, genomics, and [artificial intelligence] to draw their conclusions. This is precisely the way that ‘big data’ should be used – in a complementary fashion.”
The study authors and Dr. Becker reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM MED
Statins don’t worsen muscle injury from moderately intense exercise
People who are physically active and on statins may have one less potential concern about the drugs. Despite their reputation for causing muscle injury, a study suggests statins won’t worsen the toll that sustained, moderately intensive exercise already takes on patients’ muscles.
Statin therapy in this prospective, controlled study wasn’t seen to aggravate normal muscle fatigue or pain from sustained exercise or adversely affect enzymes or other biomarkers associated with muscle injury.
The findings come from 100 individuals, of whom about two-thirds were on statins, participating in a public, 4-day, long-distance walking event held annually in the Netherlands. Results were published in the Journal of the American College of Cardiology with Neeltje A.E. Allard, MD, Radboud University Medical Center, Nijmegen, the Netherlands, as lead author.
For all of statins’ common use in adults with cardiovascular (CV) risk factors, the drugs are often blamed for causing excessive muscle pain or injury as a side effect. Yet there is a predominance of evidence to the contrary based on meta-analyses and clinical trials, suggesting that the drugs are taking the rap for many entirely unrelated muscle symptoms.
The new findings, from people ranging widely in fitness levels, suggest that “exercise of moderate intensity is feasible and safe” in statin users, that the drugs won’t exacerbate normal muscle symptoms from exercise, Dr. Allard told this news organization.
And that exercise doesn’t have to be on an unusual scale. Regular exercise in statin users can simply be consistent with broader guidelines, say 30 minutes of walking per day, she noted.
The study has such broad applicability, Dr. Allard said, because participants represented the spectrum of the thousands who signed up for the walking event, who varied in age, level of physical fitness, and number of CV risk factors. They included CV patients, the physically fit, “recreational walkers who didn’t really exercise regularly,” and “habitual nonexercisers.”
It enrolled three groups of participants in the Four Days Marches in Nijmegen, which in a typical year attracts tens of thousands of participants who walk up to 30 km, 40 km, or 50 km per day for 4 consecutive days.
They included 35 statin users who walked the event despite muscle symptoms, 34 on statins but without such symptoms, and 31 non–statin-using controls. Their mean ages ranged from 65 to 68 years.
Statin users were overwhelmingly on simvastatin or atorvastatin. The average statin therapy durations were 60 months and 96 months for those with and without symptoms, respectively.
Assessments were performed several days before the event, at baseline, and after the end of walking on days 1, 2, and 3.
Scores for muscle pain on the Brief Pain Inventory were higher at baseline for the symptomatic-on-statins group (P < .001) compared with the other two groups, and went up (P < .001) similarly across the three groups during each of the 3 days, the report notes. Fatigue scores on the Brief Fatigue Inventory followed the same pattern.
All biomarkers of muscle injury or stress were at comparable levels at baseline in the three groups and went up similarly (P < .001) with no significant differences at the end of day 3. Biomarkers included lactate dehydrogenase, creatine kinase, myoglobin, cardiac troponin I, and N-terminal pro-brain natriuretic peptide.
Statin-related reductions in levels of coenzyme Q 10 (CoQ10) have been thought to exacerbate muscle injury, the authors note. But levels of CoQ10 weren’t significantly different across the three groups at any point in the study, and they did not show any significant associations with measures of muscle injury, symptoms, or fatigue.
Patients with statin-associated muscle symptoms (SAMS) often limit physical activity because of muscle pain or weakness, but also “concerns that exercise will exacerbate muscle injury,” an accompanying editorial notes. “Therefore, exercise, a foundation of improving and maintaining cardiometabolic health, is often avoided or limited.”
But the current study, writes Robert S. Rosenson, MD, of Mount Sinai Heart, New York, indeed suggests that “many patients who develop SAMS may engage in a moderately intensive walking program without concern for worsened muscle biomarkers or performance.”
The exercise didn’t seem to improve muscle function in symptomatic statin users, compared with the other groups over the study’s very short follow-up, Dr. Rosenson observes. But “it remains uncertain from this study whether sustained exercise in SAMS patients will effectuate improved metabolic biomarkers or exercise capacity in the long term.”
Dr. Allard is supported by a grant from the Radboud Institute for Health Sciences; the other authors have disclosed no relevant financial relationships. Dr. Rosenson disclosed receiving research funding to his institution from Amgen, Arrowhead, Lilly, Novartis, and Regeneron; consulting fees from Amgen, Arrowhead, Lilly, Lipigon, Novartis, CRISPR Therapeutics, Precision BioSciences, Verve, Ultragenyx Pharmaceutical, and Regeneron; speaking fees from Amgen, Kowa, and Regeneron; and royalties from Wolters Kluwer (UpToDate); and that he holds stock in MediMergent.
A version of this article first appeared on Medscape.com.
People who are physically active and on statins may have one less potential concern about the drugs. Despite their reputation for causing muscle injury, a study suggests statins won’t worsen the toll that sustained, moderately intensive exercise already takes on patients’ muscles.
Statin therapy in this prospective, controlled study wasn’t seen to aggravate normal muscle fatigue or pain from sustained exercise or adversely affect enzymes or other biomarkers associated with muscle injury.
The findings come from 100 individuals, of whom about two-thirds were on statins, participating in a public, 4-day, long-distance walking event held annually in the Netherlands. Results were published in the Journal of the American College of Cardiology with Neeltje A.E. Allard, MD, Radboud University Medical Center, Nijmegen, the Netherlands, as lead author.
For all of statins’ common use in adults with cardiovascular (CV) risk factors, the drugs are often blamed for causing excessive muscle pain or injury as a side effect. Yet there is a predominance of evidence to the contrary based on meta-analyses and clinical trials, suggesting that the drugs are taking the rap for many entirely unrelated muscle symptoms.
The new findings, from people ranging widely in fitness levels, suggest that “exercise of moderate intensity is feasible and safe” in statin users, that the drugs won’t exacerbate normal muscle symptoms from exercise, Dr. Allard told this news organization.
And that exercise doesn’t have to be on an unusual scale. Regular exercise in statin users can simply be consistent with broader guidelines, say 30 minutes of walking per day, she noted.
The study has such broad applicability, Dr. Allard said, because participants represented the spectrum of the thousands who signed up for the walking event, who varied in age, level of physical fitness, and number of CV risk factors. They included CV patients, the physically fit, “recreational walkers who didn’t really exercise regularly,” and “habitual nonexercisers.”
It enrolled three groups of participants in the Four Days Marches in Nijmegen, which in a typical year attracts tens of thousands of participants who walk up to 30 km, 40 km, or 50 km per day for 4 consecutive days.
They included 35 statin users who walked the event despite muscle symptoms, 34 on statins but without such symptoms, and 31 non–statin-using controls. Their mean ages ranged from 65 to 68 years.
Statin users were overwhelmingly on simvastatin or atorvastatin. The average statin therapy durations were 60 months and 96 months for those with and without symptoms, respectively.
Assessments were performed several days before the event, at baseline, and after the end of walking on days 1, 2, and 3.
Scores for muscle pain on the Brief Pain Inventory were higher at baseline for the symptomatic-on-statins group (P < .001) compared with the other two groups, and went up (P < .001) similarly across the three groups during each of the 3 days, the report notes. Fatigue scores on the Brief Fatigue Inventory followed the same pattern.
All biomarkers of muscle injury or stress were at comparable levels at baseline in the three groups and went up similarly (P < .001) with no significant differences at the end of day 3. Biomarkers included lactate dehydrogenase, creatine kinase, myoglobin, cardiac troponin I, and N-terminal pro-brain natriuretic peptide.
Statin-related reductions in levels of coenzyme Q 10 (CoQ10) have been thought to exacerbate muscle injury, the authors note. But levels of CoQ10 weren’t significantly different across the three groups at any point in the study, and they did not show any significant associations with measures of muscle injury, symptoms, or fatigue.
Patients with statin-associated muscle symptoms (SAMS) often limit physical activity because of muscle pain or weakness, but also “concerns that exercise will exacerbate muscle injury,” an accompanying editorial notes. “Therefore, exercise, a foundation of improving and maintaining cardiometabolic health, is often avoided or limited.”
But the current study, writes Robert S. Rosenson, MD, of Mount Sinai Heart, New York, indeed suggests that “many patients who develop SAMS may engage in a moderately intensive walking program without concern for worsened muscle biomarkers or performance.”
The exercise didn’t seem to improve muscle function in symptomatic statin users, compared with the other groups over the study’s very short follow-up, Dr. Rosenson observes. But “it remains uncertain from this study whether sustained exercise in SAMS patients will effectuate improved metabolic biomarkers or exercise capacity in the long term.”
Dr. Allard is supported by a grant from the Radboud Institute for Health Sciences; the other authors have disclosed no relevant financial relationships. Dr. Rosenson disclosed receiving research funding to his institution from Amgen, Arrowhead, Lilly, Novartis, and Regeneron; consulting fees from Amgen, Arrowhead, Lilly, Lipigon, Novartis, CRISPR Therapeutics, Precision BioSciences, Verve, Ultragenyx Pharmaceutical, and Regeneron; speaking fees from Amgen, Kowa, and Regeneron; and royalties from Wolters Kluwer (UpToDate); and that he holds stock in MediMergent.
A version of this article first appeared on Medscape.com.
People who are physically active and on statins may have one less potential concern about the drugs. Despite their reputation for causing muscle injury, a study suggests statins won’t worsen the toll that sustained, moderately intensive exercise already takes on patients’ muscles.
Statin therapy in this prospective, controlled study wasn’t seen to aggravate normal muscle fatigue or pain from sustained exercise or adversely affect enzymes or other biomarkers associated with muscle injury.
The findings come from 100 individuals, of whom about two-thirds were on statins, participating in a public, 4-day, long-distance walking event held annually in the Netherlands. Results were published in the Journal of the American College of Cardiology with Neeltje A.E. Allard, MD, Radboud University Medical Center, Nijmegen, the Netherlands, as lead author.
For all of statins’ common use in adults with cardiovascular (CV) risk factors, the drugs are often blamed for causing excessive muscle pain or injury as a side effect. Yet there is a predominance of evidence to the contrary based on meta-analyses and clinical trials, suggesting that the drugs are taking the rap for many entirely unrelated muscle symptoms.
The new findings, from people ranging widely in fitness levels, suggest that “exercise of moderate intensity is feasible and safe” in statin users, that the drugs won’t exacerbate normal muscle symptoms from exercise, Dr. Allard told this news organization.
And that exercise doesn’t have to be on an unusual scale. Regular exercise in statin users can simply be consistent with broader guidelines, say 30 minutes of walking per day, she noted.
The study has such broad applicability, Dr. Allard said, because participants represented the spectrum of the thousands who signed up for the walking event, who varied in age, level of physical fitness, and number of CV risk factors. They included CV patients, the physically fit, “recreational walkers who didn’t really exercise regularly,” and “habitual nonexercisers.”
It enrolled three groups of participants in the Four Days Marches in Nijmegen, which in a typical year attracts tens of thousands of participants who walk up to 30 km, 40 km, or 50 km per day for 4 consecutive days.
They included 35 statin users who walked the event despite muscle symptoms, 34 on statins but without such symptoms, and 31 non–statin-using controls. Their mean ages ranged from 65 to 68 years.
Statin users were overwhelmingly on simvastatin or atorvastatin. The average statin therapy durations were 60 months and 96 months for those with and without symptoms, respectively.
Assessments were performed several days before the event, at baseline, and after the end of walking on days 1, 2, and 3.
Scores for muscle pain on the Brief Pain Inventory were higher at baseline for the symptomatic-on-statins group (P < .001) compared with the other two groups, and went up (P < .001) similarly across the three groups during each of the 3 days, the report notes. Fatigue scores on the Brief Fatigue Inventory followed the same pattern.
All biomarkers of muscle injury or stress were at comparable levels at baseline in the three groups and went up similarly (P < .001) with no significant differences at the end of day 3. Biomarkers included lactate dehydrogenase, creatine kinase, myoglobin, cardiac troponin I, and N-terminal pro-brain natriuretic peptide.
Statin-related reductions in levels of coenzyme Q 10 (CoQ10) have been thought to exacerbate muscle injury, the authors note. But levels of CoQ10 weren’t significantly different across the three groups at any point in the study, and they did not show any significant associations with measures of muscle injury, symptoms, or fatigue.
Patients with statin-associated muscle symptoms (SAMS) often limit physical activity because of muscle pain or weakness, but also “concerns that exercise will exacerbate muscle injury,” an accompanying editorial notes. “Therefore, exercise, a foundation of improving and maintaining cardiometabolic health, is often avoided or limited.”
But the current study, writes Robert S. Rosenson, MD, of Mount Sinai Heart, New York, indeed suggests that “many patients who develop SAMS may engage in a moderately intensive walking program without concern for worsened muscle biomarkers or performance.”
The exercise didn’t seem to improve muscle function in symptomatic statin users, compared with the other groups over the study’s very short follow-up, Dr. Rosenson observes. But “it remains uncertain from this study whether sustained exercise in SAMS patients will effectuate improved metabolic biomarkers or exercise capacity in the long term.”
Dr. Allard is supported by a grant from the Radboud Institute for Health Sciences; the other authors have disclosed no relevant financial relationships. Dr. Rosenson disclosed receiving research funding to his institution from Amgen, Arrowhead, Lilly, Novartis, and Regeneron; consulting fees from Amgen, Arrowhead, Lilly, Lipigon, Novartis, CRISPR Therapeutics, Precision BioSciences, Verve, Ultragenyx Pharmaceutical, and Regeneron; speaking fees from Amgen, Kowa, and Regeneron; and royalties from Wolters Kluwer (UpToDate); and that he holds stock in MediMergent.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
High salt intake linked to atherosclerosis even with normal BP
A high salt intake is an important risk factor for atherosclerosis, even in the absence of hypertension, a large study from Sweden concludes.
The study, including more than 10,000 individuals between the ages of 50 and 64 years from the Swedish Cardiopulmonary bioImage Study, showed a significant link between dietary salt intake and the risk for atherosclerotic lesions in the coronary and carotid arteries, even in participants with normal blood pressure and without known cardiovascular disease.
The finding suggests that salt could be a damaging factor in its own right before the development of hypertension, the authors write. The results were published online in European Heart Journal Open.
It has been known for a long time that salt is linked to hypertension, but the role that salt plays in atherosclerosis has not been examined, first author Jonas Wuopio, MD, Karolinska Institutet, Huddinge, and Clinical Research Center, Falun, Uppsala University, both in Sweden, told this news organization.
“Hardly anyone looks at changes in the arteries’ calcification, the atherosclerotic plaques and the association with salt intake,” Dr. Wuopio said. “We had this exclusive data from our cohort, so we wanted to use it to close this knowledge gap.”
The analysis included 10,788 adults aged 50-64 years, (average age, 58 years; 52% women) who underwent a coronary computed tomography angiography (CCTA) scan. The estimated 24-hour sodium excretion was used to measure sodium intake.
CCTA was used to obtain 3-D images of the coronary arteries to measure the degree of coronary artery calcium as well as detect stenosis in the coronary arteries. Participants also had an ultrasound of the carotid arteries.
After adjusting for age, sex, and study site (the study was done at Uppsala and Malmö, Sweden), the researchers found that rising salt consumption was linked with increasing atherosclerosis in a linear fashion in both the coronary and carotid arteries.
Each 1,000 mg rise in sodium excretion was associated with a 9% increased occurrence of carotid plaque (odds ratio, 1.09; P < .001; confidence interval, 1.06-1.12), a higher coronary artery calcium score (OR, 1.16; P < .001; CI, 1.12-1.19), and a 17% increased occurrence of coronary artery stenosis (OR, 1.17; P < .001; CI, 1.13-1.20).
The association was abolished, though, after adjusting for blood pressure, they note. Their “interpretation is that the increase in blood pressure from sodium intake, even below the level that currently defines arterial hypertension, is an important factor that mediates the interplay between salt intake and the atherosclerotic process,” they write. “As we observed an association in individuals with normal blood pressure, one possible explanation for these findings is that the detrimental pathological processes begin already prior to the development of hypertension,” they note, although they caution that no causal relationships can be gleaned from this cross-sectional study.
They also reported no sign of a “J-curve”; participants with the lowest levels of sodium excretion had the lowest occurrence of both coronary and carotid atherosclerosis, which contradicts findings in some studies that found very low sodium linked to increased cardiovascular disease–related events.
“There have been some controversies among researchers regarding very low intake, where some say very low salt intake can increase the risk of cardiovascular disease, but we could not find this in this study,” Dr. Wuopio said.
“Our study is confirming that excess salt is not a good thing, but the fact that it is linked to atherosclerosis, even in the absence of hypertension, was a bit of a surprise,” he said.
“I will be telling my patients to follow the advice given by the World Health Organization and other medical societies, to limit your intake of salt to approximately 1 teaspoon, even if your blood pressure is normal.”
Time to scrutinize salt’s role in atherosclerosis
In an accompanying editorial, Maciej Banach, MD, Medical University of Lodz, and Stanislaw Surma, MD, Faculty of Medical Sciences in Katowice, both in Poland, write that excessive dietary salt intake is a well-documented cardiovascular risk factor, and that the association is explained in most studies by increased blood pressure.
“We should look more extensively on the role of dietary salt, as it affects many pathological mechanisms, by which, especially with the coexistence of other risk factors, atherosclerosis may progress very fast,” they write.
“The results of the study shed new light on the direct relationship between excessive dietary salt intake and the risk of ASCVD [atherosclerotic cardiovascular disease], indicating that salt intake might be a risk factor for atherosclerosis even prior to the development of hypertension,” they conclude.
Confirmatory and novel
“Nobody questions the fact that high blood pressure is a powerful risk factor for atherosclerotic disease, but not all studies have suggested that, at least at significantly higher levels of sodium intake, that high salt intake tracks with risk for atherosclerotic disease,” Alon Gitig, MD, assistant professor and director of cardiology, Mount Sinai Doctors-Westchester, Yonkers, New York, told this news organization.
Most of the studies of salt intake in the diet are based on patient self-reports via food frequency questionnaires, which can give a general idea of salt intake, but are often not totally accurate, Dr. Gitig said.
“Here, they measured sodium in the urine and estimated the 24-hour salt intake from that, which is slightly novel,” he said.
Everybody knows that high blood pressure is associated with future cardiovascular disease risk, but what many don’t realize is that that risk starts to increase slightly but significantly above a blood pressure that is already in the range of 115 mm Hg/75 mm Hg, he said.
“The lower you can get your blood pressure down, to around 115-120, the lower your risk for cardiovascular disease,” Dr. Gitig said.
It is possible for most people to lower blood pressure through attention to diet, restricting sodium, performing cardio and weight training exercises, and maintaining a healthy weight, he said.
An example of a cardiovascular health diet is the Dietary Approaches to Stop Hypertension (DASH) diet.
“The DASH diet, consisting of 9 servings of fruits and vegetables a day with few refined carbs, flour and sugar, has been shown in a randomized trial to dramatically reduce blood pressure. There are two reasons for that. One is that the fruits and vegetables have many phytonutrients that are good for arteries. The other is that a large proportion of U.S. adults have insulin resistance, which leads to high blood pressure.
“The more fruits and vegetables and healthy animal products, and less sugar and flour, the more you are going to improve your insulin resistance, so you can bring your blood pressure down that way,” Dr. Gitig said.
The study was funded by the Swedish Heart-Lung Foundation, the Knut and Alice Wallenberg Foundation, the Swedish Research Council and Vinnova (Sweden’s Innovation agency), the University of Gothenburg and Sahlgrenska University Hospital, the Karolinska Institutet and Stockholm County Council, the Linköping University and University Hospital, the Lund University and Skane University Hospital, the Umea University and University Hospital, and the Uppsala University and University Hospital. Dr. Wuopio and Dr. Gitig report no relevant financial relationships. Dr. Banach reports financial relationships with Adamed, Amgen, Daichii Sankyo, Esperion, KrKa, NewAmsterdam, Polpharma, Novartis, Pfizer, Sanofi, Teva, Viatris, and CMDO at Longevity Group (LU). Dr. Surma reports a financial relationship with Sanofi and Novartis.
A version of this article first appeared on Medscape.com.
A high salt intake is an important risk factor for atherosclerosis, even in the absence of hypertension, a large study from Sweden concludes.
The study, including more than 10,000 individuals between the ages of 50 and 64 years from the Swedish Cardiopulmonary bioImage Study, showed a significant link between dietary salt intake and the risk for atherosclerotic lesions in the coronary and carotid arteries, even in participants with normal blood pressure and without known cardiovascular disease.
The finding suggests that salt could be a damaging factor in its own right before the development of hypertension, the authors write. The results were published online in European Heart Journal Open.
It has been known for a long time that salt is linked to hypertension, but the role that salt plays in atherosclerosis has not been examined, first author Jonas Wuopio, MD, Karolinska Institutet, Huddinge, and Clinical Research Center, Falun, Uppsala University, both in Sweden, told this news organization.
“Hardly anyone looks at changes in the arteries’ calcification, the atherosclerotic plaques and the association with salt intake,” Dr. Wuopio said. “We had this exclusive data from our cohort, so we wanted to use it to close this knowledge gap.”
The analysis included 10,788 adults aged 50-64 years, (average age, 58 years; 52% women) who underwent a coronary computed tomography angiography (CCTA) scan. The estimated 24-hour sodium excretion was used to measure sodium intake.
CCTA was used to obtain 3-D images of the coronary arteries to measure the degree of coronary artery calcium as well as detect stenosis in the coronary arteries. Participants also had an ultrasound of the carotid arteries.
After adjusting for age, sex, and study site (the study was done at Uppsala and Malmö, Sweden), the researchers found that rising salt consumption was linked with increasing atherosclerosis in a linear fashion in both the coronary and carotid arteries.
Each 1,000 mg rise in sodium excretion was associated with a 9% increased occurrence of carotid plaque (odds ratio, 1.09; P < .001; confidence interval, 1.06-1.12), a higher coronary artery calcium score (OR, 1.16; P < .001; CI, 1.12-1.19), and a 17% increased occurrence of coronary artery stenosis (OR, 1.17; P < .001; CI, 1.13-1.20).
The association was abolished, though, after adjusting for blood pressure, they note. Their “interpretation is that the increase in blood pressure from sodium intake, even below the level that currently defines arterial hypertension, is an important factor that mediates the interplay between salt intake and the atherosclerotic process,” they write. “As we observed an association in individuals with normal blood pressure, one possible explanation for these findings is that the detrimental pathological processes begin already prior to the development of hypertension,” they note, although they caution that no causal relationships can be gleaned from this cross-sectional study.
They also reported no sign of a “J-curve”; participants with the lowest levels of sodium excretion had the lowest occurrence of both coronary and carotid atherosclerosis, which contradicts findings in some studies that found very low sodium linked to increased cardiovascular disease–related events.
“There have been some controversies among researchers regarding very low intake, where some say very low salt intake can increase the risk of cardiovascular disease, but we could not find this in this study,” Dr. Wuopio said.
“Our study is confirming that excess salt is not a good thing, but the fact that it is linked to atherosclerosis, even in the absence of hypertension, was a bit of a surprise,” he said.
“I will be telling my patients to follow the advice given by the World Health Organization and other medical societies, to limit your intake of salt to approximately 1 teaspoon, even if your blood pressure is normal.”
Time to scrutinize salt’s role in atherosclerosis
In an accompanying editorial, Maciej Banach, MD, Medical University of Lodz, and Stanislaw Surma, MD, Faculty of Medical Sciences in Katowice, both in Poland, write that excessive dietary salt intake is a well-documented cardiovascular risk factor, and that the association is explained in most studies by increased blood pressure.
“We should look more extensively on the role of dietary salt, as it affects many pathological mechanisms, by which, especially with the coexistence of other risk factors, atherosclerosis may progress very fast,” they write.
“The results of the study shed new light on the direct relationship between excessive dietary salt intake and the risk of ASCVD [atherosclerotic cardiovascular disease], indicating that salt intake might be a risk factor for atherosclerosis even prior to the development of hypertension,” they conclude.
Confirmatory and novel
“Nobody questions the fact that high blood pressure is a powerful risk factor for atherosclerotic disease, but not all studies have suggested that, at least at significantly higher levels of sodium intake, that high salt intake tracks with risk for atherosclerotic disease,” Alon Gitig, MD, assistant professor and director of cardiology, Mount Sinai Doctors-Westchester, Yonkers, New York, told this news organization.
Most of the studies of salt intake in the diet are based on patient self-reports via food frequency questionnaires, which can give a general idea of salt intake, but are often not totally accurate, Dr. Gitig said.
“Here, they measured sodium in the urine and estimated the 24-hour salt intake from that, which is slightly novel,” he said.
Everybody knows that high blood pressure is associated with future cardiovascular disease risk, but what many don’t realize is that that risk starts to increase slightly but significantly above a blood pressure that is already in the range of 115 mm Hg/75 mm Hg, he said.
“The lower you can get your blood pressure down, to around 115-120, the lower your risk for cardiovascular disease,” Dr. Gitig said.
It is possible for most people to lower blood pressure through attention to diet, restricting sodium, performing cardio and weight training exercises, and maintaining a healthy weight, he said.
An example of a cardiovascular health diet is the Dietary Approaches to Stop Hypertension (DASH) diet.
“The DASH diet, consisting of 9 servings of fruits and vegetables a day with few refined carbs, flour and sugar, has been shown in a randomized trial to dramatically reduce blood pressure. There are two reasons for that. One is that the fruits and vegetables have many phytonutrients that are good for arteries. The other is that a large proportion of U.S. adults have insulin resistance, which leads to high blood pressure.
“The more fruits and vegetables and healthy animal products, and less sugar and flour, the more you are going to improve your insulin resistance, so you can bring your blood pressure down that way,” Dr. Gitig said.
The study was funded by the Swedish Heart-Lung Foundation, the Knut and Alice Wallenberg Foundation, the Swedish Research Council and Vinnova (Sweden’s Innovation agency), the University of Gothenburg and Sahlgrenska University Hospital, the Karolinska Institutet and Stockholm County Council, the Linköping University and University Hospital, the Lund University and Skane University Hospital, the Umea University and University Hospital, and the Uppsala University and University Hospital. Dr. Wuopio and Dr. Gitig report no relevant financial relationships. Dr. Banach reports financial relationships with Adamed, Amgen, Daichii Sankyo, Esperion, KrKa, NewAmsterdam, Polpharma, Novartis, Pfizer, Sanofi, Teva, Viatris, and CMDO at Longevity Group (LU). Dr. Surma reports a financial relationship with Sanofi and Novartis.
A version of this article first appeared on Medscape.com.
A high salt intake is an important risk factor for atherosclerosis, even in the absence of hypertension, a large study from Sweden concludes.
The study, including more than 10,000 individuals between the ages of 50 and 64 years from the Swedish Cardiopulmonary bioImage Study, showed a significant link between dietary salt intake and the risk for atherosclerotic lesions in the coronary and carotid arteries, even in participants with normal blood pressure and without known cardiovascular disease.
The finding suggests that salt could be a damaging factor in its own right before the development of hypertension, the authors write. The results were published online in European Heart Journal Open.
It has been known for a long time that salt is linked to hypertension, but the role that salt plays in atherosclerosis has not been examined, first author Jonas Wuopio, MD, Karolinska Institutet, Huddinge, and Clinical Research Center, Falun, Uppsala University, both in Sweden, told this news organization.
“Hardly anyone looks at changes in the arteries’ calcification, the atherosclerotic plaques and the association with salt intake,” Dr. Wuopio said. “We had this exclusive data from our cohort, so we wanted to use it to close this knowledge gap.”
The analysis included 10,788 adults aged 50-64 years, (average age, 58 years; 52% women) who underwent a coronary computed tomography angiography (CCTA) scan. The estimated 24-hour sodium excretion was used to measure sodium intake.
CCTA was used to obtain 3-D images of the coronary arteries to measure the degree of coronary artery calcium as well as detect stenosis in the coronary arteries. Participants also had an ultrasound of the carotid arteries.
After adjusting for age, sex, and study site (the study was done at Uppsala and Malmö, Sweden), the researchers found that rising salt consumption was linked with increasing atherosclerosis in a linear fashion in both the coronary and carotid arteries.
Each 1,000 mg rise in sodium excretion was associated with a 9% increased occurrence of carotid plaque (odds ratio, 1.09; P < .001; confidence interval, 1.06-1.12), a higher coronary artery calcium score (OR, 1.16; P < .001; CI, 1.12-1.19), and a 17% increased occurrence of coronary artery stenosis (OR, 1.17; P < .001; CI, 1.13-1.20).
The association was abolished, though, after adjusting for blood pressure, they note. Their “interpretation is that the increase in blood pressure from sodium intake, even below the level that currently defines arterial hypertension, is an important factor that mediates the interplay between salt intake and the atherosclerotic process,” they write. “As we observed an association in individuals with normal blood pressure, one possible explanation for these findings is that the detrimental pathological processes begin already prior to the development of hypertension,” they note, although they caution that no causal relationships can be gleaned from this cross-sectional study.
They also reported no sign of a “J-curve”; participants with the lowest levels of sodium excretion had the lowest occurrence of both coronary and carotid atherosclerosis, which contradicts findings in some studies that found very low sodium linked to increased cardiovascular disease–related events.
“There have been some controversies among researchers regarding very low intake, where some say very low salt intake can increase the risk of cardiovascular disease, but we could not find this in this study,” Dr. Wuopio said.
“Our study is confirming that excess salt is not a good thing, but the fact that it is linked to atherosclerosis, even in the absence of hypertension, was a bit of a surprise,” he said.
“I will be telling my patients to follow the advice given by the World Health Organization and other medical societies, to limit your intake of salt to approximately 1 teaspoon, even if your blood pressure is normal.”
Time to scrutinize salt’s role in atherosclerosis
In an accompanying editorial, Maciej Banach, MD, Medical University of Lodz, and Stanislaw Surma, MD, Faculty of Medical Sciences in Katowice, both in Poland, write that excessive dietary salt intake is a well-documented cardiovascular risk factor, and that the association is explained in most studies by increased blood pressure.
“We should look more extensively on the role of dietary salt, as it affects many pathological mechanisms, by which, especially with the coexistence of other risk factors, atherosclerosis may progress very fast,” they write.
“The results of the study shed new light on the direct relationship between excessive dietary salt intake and the risk of ASCVD [atherosclerotic cardiovascular disease], indicating that salt intake might be a risk factor for atherosclerosis even prior to the development of hypertension,” they conclude.
Confirmatory and novel
“Nobody questions the fact that high blood pressure is a powerful risk factor for atherosclerotic disease, but not all studies have suggested that, at least at significantly higher levels of sodium intake, that high salt intake tracks with risk for atherosclerotic disease,” Alon Gitig, MD, assistant professor and director of cardiology, Mount Sinai Doctors-Westchester, Yonkers, New York, told this news organization.
Most of the studies of salt intake in the diet are based on patient self-reports via food frequency questionnaires, which can give a general idea of salt intake, but are often not totally accurate, Dr. Gitig said.
“Here, they measured sodium in the urine and estimated the 24-hour salt intake from that, which is slightly novel,” he said.
Everybody knows that high blood pressure is associated with future cardiovascular disease risk, but what many don’t realize is that that risk starts to increase slightly but significantly above a blood pressure that is already in the range of 115 mm Hg/75 mm Hg, he said.
“The lower you can get your blood pressure down, to around 115-120, the lower your risk for cardiovascular disease,” Dr. Gitig said.
It is possible for most people to lower blood pressure through attention to diet, restricting sodium, performing cardio and weight training exercises, and maintaining a healthy weight, he said.
An example of a cardiovascular health diet is the Dietary Approaches to Stop Hypertension (DASH) diet.
“The DASH diet, consisting of 9 servings of fruits and vegetables a day with few refined carbs, flour and sugar, has been shown in a randomized trial to dramatically reduce blood pressure. There are two reasons for that. One is that the fruits and vegetables have many phytonutrients that are good for arteries. The other is that a large proportion of U.S. adults have insulin resistance, which leads to high blood pressure.
“The more fruits and vegetables and healthy animal products, and less sugar and flour, the more you are going to improve your insulin resistance, so you can bring your blood pressure down that way,” Dr. Gitig said.
The study was funded by the Swedish Heart-Lung Foundation, the Knut and Alice Wallenberg Foundation, the Swedish Research Council and Vinnova (Sweden’s Innovation agency), the University of Gothenburg and Sahlgrenska University Hospital, the Karolinska Institutet and Stockholm County Council, the Linköping University and University Hospital, the Lund University and Skane University Hospital, the Umea University and University Hospital, and the Uppsala University and University Hospital. Dr. Wuopio and Dr. Gitig report no relevant financial relationships. Dr. Banach reports financial relationships with Adamed, Amgen, Daichii Sankyo, Esperion, KrKa, NewAmsterdam, Polpharma, Novartis, Pfizer, Sanofi, Teva, Viatris, and CMDO at Longevity Group (LU). Dr. Surma reports a financial relationship with Sanofi and Novartis.
A version of this article first appeared on Medscape.com.
New update on left atrial appendage closure recommendations
An updated consensus statement on transcatheter left atrial appendage closure (LAAC) has put a newfound focus on patient selection for the procedure, specifically recommending that the procedure is appropriate for patients with nonvalvular atrial fibrillation who have risk for thromboembolism, aren’t well suited for direct oral anticoagulants (DOACs) and have a good chance of living for at least another year.
The statement, published online in the Journal of the Society for Cardiovascular Angiography & Interventions, also makes recommendations for how much experience operators should have, how many procedures they should perform to keep their skills up, and when and how to use imaging and prescribe DOACs, among other suggestions.
The statement represents the first updated guidance for LAAC since 2015. “Since then this field has really expanded and evolved,” writing group chair Jacqueline Saw, MD, said in an interview. “For instance, the indications are more matured and specific, and the procedural technical steps have matured. Imaging has also advanced, there’s more understanding about postprocedural care and there are also new devices that have been approved.”
Dr. Saw, an interventional cardiologist at Vancouver General Hospital and St. Paul’s Hospital, and a professor at the University of British Columbia in Vancouver, called the statement “a piece that puts everything together.”
“This document really summarizes the whole practice for doing transcatheter procedures,” she added, “so it’s all-in-one document in terms of recommendation of who we do the procedure for, how we should do it, how we should image and guide the procedure, and what complications to look out for and how to manage patients post procedure, be it with antithrombotic therapy and/or device surveillance.”
13 recommendations
In all, the statement carries 13 recommendations for LAAC. The Society for Cardiovascular Angiography & Interventions and the Heart Rhythm Society commissioned the writing group. The American College of Cardiology and Society of Cardiovascular Computed Tomography have endorsed the statement. The following are among the recommendations:
- Transcatheter LAAC is appropriate for patients with nonvalvular atrial fibrillation with high thromboembolic risk but for whom long-term oral anticoagulation may be contraindicated and who have at least 1 year’s life expectancy.
- Operators should have performed at least 50 prior left-sided ablations or structural procedures and at least 25 transseptal punctures (TSPs). Interventional-imaging physicians should have experience in guiding 25 or more TSPs before supporting LAAC procedures independently.
- To maintain skills, operators should do 25 or more TSPs and at least 12 LAACs over each 2-year period.
- On-site cardiovascular surgery backup should be available for new programs and for operators early in their learning curve.
- Baseline imaging with transesophageal echocardiography (TEE) or cardiac computed tomography should be performed before LAAC.
- Intraprocedural imaging guidance with TEE or intracardiac echocardiography.
- Follow labeling of each specific LAAC device for technical aspects of the procedure.
- Familiarity with avoiding, recognizing, and managing LAAC complications.
- Predischarge 2-dimensional TEE to rule out pericardial effusion and device embolization.
- Anticoagulation for device-related thrombus.
- Make all efforts to minimize peridevice leaks during implantation because their clinical impact and management isn’t well understood.
- Antithrombotic therapy with warfarin, DOAC, or dual-antiplatelet therapy after LAAC based on the studied regimen and instructions for each specific device, tailored to the bleeding risks for each patient.
- TEE or cardiac computed tomography at 45-90 days after LAAC for device surveillance to assess for peridevice leak and device-related thrombus.
The statement also includes precautionary recommendations. It advises against using routine closure of LAAC-associated iatrogenic atrial septal defects and states that combined procedures with LAAC, such as structural interventions and pulmonary vein isolation, should be avoided because randomized controlled trial data are pending.
“These recommendations are based upon data from updated publications and randomized trial data as well as large registries, including the National Cardiovascular Data Registry, so I think this is a very practical statement that puts all these pieces together for any budding interventionalist doing this procedure and even experienced operations,” Dr. Saw said.
Authors of an accompanying editorial agreed that the “rigorous standards” set out in the statement will help maintain “a high level of procedural safety in the setting of rapid expansion.”
The editorialists, Faisal M. Merchant, MD, of Emory University, Atlanta, and Mohamad Alkhouli, MD, professor of medicine at Mayo Clinic School of Medicine, Rochester, Minn., point out that the incidence of pericardial effusion has decreased from more than 5% in the pivotal Watchman trials to less than 1.5% in the most recent report from the National Cardiovascular Data Registry, which shows that more than 100,000 procedures have been performed in the United States.
But most important as the field moves forward, they stress, is patient selection. The recommendation of limiting patients to those with a life expectancy of 1 year “is a tacit recognition of the fact that the benefits of LAAC take time to accrue, and many older and frail patients are unlikely to derive meaningful benefit.”
Dr. Merchant and Dr. Alkhouli also note that there remains a conundrum in patient selection that remains from the original LAAC trials, which enrolled patients who were eligible for anticoagulation. “Somewhat paradoxically, after its approval, LAAC is mostly prescribed to patients who are not felt to be good anticoagulation candidates.” This leaves physicians “in the precarious position of extrapolating data to patients who were excluded from the original clinical trials.”
Therefore, the consensus statement “is right to put patient selection front and center in its recommendations, but as the field of LAAC comes of age, better evidence to support patient selection will be the real sign of maturity.”
Dr. Saw said she envisions another update over the next 2 years or so as ongoing clinical trials comparing DOAC and LAAC, namely the CHAMPION-AF and OPTION trials, report results.
Dr. Saw and Dr. Merchant, reported no conflicts of interest. Dr. Alkhouli has financial ties to Boston Scientific, Abbott, and Philips.
An updated consensus statement on transcatheter left atrial appendage closure (LAAC) has put a newfound focus on patient selection for the procedure, specifically recommending that the procedure is appropriate for patients with nonvalvular atrial fibrillation who have risk for thromboembolism, aren’t well suited for direct oral anticoagulants (DOACs) and have a good chance of living for at least another year.
The statement, published online in the Journal of the Society for Cardiovascular Angiography & Interventions, also makes recommendations for how much experience operators should have, how many procedures they should perform to keep their skills up, and when and how to use imaging and prescribe DOACs, among other suggestions.
The statement represents the first updated guidance for LAAC since 2015. “Since then this field has really expanded and evolved,” writing group chair Jacqueline Saw, MD, said in an interview. “For instance, the indications are more matured and specific, and the procedural technical steps have matured. Imaging has also advanced, there’s more understanding about postprocedural care and there are also new devices that have been approved.”
Dr. Saw, an interventional cardiologist at Vancouver General Hospital and St. Paul’s Hospital, and a professor at the University of British Columbia in Vancouver, called the statement “a piece that puts everything together.”
“This document really summarizes the whole practice for doing transcatheter procedures,” she added, “so it’s all-in-one document in terms of recommendation of who we do the procedure for, how we should do it, how we should image and guide the procedure, and what complications to look out for and how to manage patients post procedure, be it with antithrombotic therapy and/or device surveillance.”
13 recommendations
In all, the statement carries 13 recommendations for LAAC. The Society for Cardiovascular Angiography & Interventions and the Heart Rhythm Society commissioned the writing group. The American College of Cardiology and Society of Cardiovascular Computed Tomography have endorsed the statement. The following are among the recommendations:
- Transcatheter LAAC is appropriate for patients with nonvalvular atrial fibrillation with high thromboembolic risk but for whom long-term oral anticoagulation may be contraindicated and who have at least 1 year’s life expectancy.
- Operators should have performed at least 50 prior left-sided ablations or structural procedures and at least 25 transseptal punctures (TSPs). Interventional-imaging physicians should have experience in guiding 25 or more TSPs before supporting LAAC procedures independently.
- To maintain skills, operators should do 25 or more TSPs and at least 12 LAACs over each 2-year period.
- On-site cardiovascular surgery backup should be available for new programs and for operators early in their learning curve.
- Baseline imaging with transesophageal echocardiography (TEE) or cardiac computed tomography should be performed before LAAC.
- Intraprocedural imaging guidance with TEE or intracardiac echocardiography.
- Follow labeling of each specific LAAC device for technical aspects of the procedure.
- Familiarity with avoiding, recognizing, and managing LAAC complications.
- Predischarge 2-dimensional TEE to rule out pericardial effusion and device embolization.
- Anticoagulation for device-related thrombus.
- Make all efforts to minimize peridevice leaks during implantation because their clinical impact and management isn’t well understood.
- Antithrombotic therapy with warfarin, DOAC, or dual-antiplatelet therapy after LAAC based on the studied regimen and instructions for each specific device, tailored to the bleeding risks for each patient.
- TEE or cardiac computed tomography at 45-90 days after LAAC for device surveillance to assess for peridevice leak and device-related thrombus.
The statement also includes precautionary recommendations. It advises against using routine closure of LAAC-associated iatrogenic atrial septal defects and states that combined procedures with LAAC, such as structural interventions and pulmonary vein isolation, should be avoided because randomized controlled trial data are pending.
“These recommendations are based upon data from updated publications and randomized trial data as well as large registries, including the National Cardiovascular Data Registry, so I think this is a very practical statement that puts all these pieces together for any budding interventionalist doing this procedure and even experienced operations,” Dr. Saw said.
Authors of an accompanying editorial agreed that the “rigorous standards” set out in the statement will help maintain “a high level of procedural safety in the setting of rapid expansion.”
The editorialists, Faisal M. Merchant, MD, of Emory University, Atlanta, and Mohamad Alkhouli, MD, professor of medicine at Mayo Clinic School of Medicine, Rochester, Minn., point out that the incidence of pericardial effusion has decreased from more than 5% in the pivotal Watchman trials to less than 1.5% in the most recent report from the National Cardiovascular Data Registry, which shows that more than 100,000 procedures have been performed in the United States.
But most important as the field moves forward, they stress, is patient selection. The recommendation of limiting patients to those with a life expectancy of 1 year “is a tacit recognition of the fact that the benefits of LAAC take time to accrue, and many older and frail patients are unlikely to derive meaningful benefit.”
Dr. Merchant and Dr. Alkhouli also note that there remains a conundrum in patient selection that remains from the original LAAC trials, which enrolled patients who were eligible for anticoagulation. “Somewhat paradoxically, after its approval, LAAC is mostly prescribed to patients who are not felt to be good anticoagulation candidates.” This leaves physicians “in the precarious position of extrapolating data to patients who were excluded from the original clinical trials.”
Therefore, the consensus statement “is right to put patient selection front and center in its recommendations, but as the field of LAAC comes of age, better evidence to support patient selection will be the real sign of maturity.”
Dr. Saw said she envisions another update over the next 2 years or so as ongoing clinical trials comparing DOAC and LAAC, namely the CHAMPION-AF and OPTION trials, report results.
Dr. Saw and Dr. Merchant, reported no conflicts of interest. Dr. Alkhouli has financial ties to Boston Scientific, Abbott, and Philips.
An updated consensus statement on transcatheter left atrial appendage closure (LAAC) has put a newfound focus on patient selection for the procedure, specifically recommending that the procedure is appropriate for patients with nonvalvular atrial fibrillation who have risk for thromboembolism, aren’t well suited for direct oral anticoagulants (DOACs) and have a good chance of living for at least another year.
The statement, published online in the Journal of the Society for Cardiovascular Angiography & Interventions, also makes recommendations for how much experience operators should have, how many procedures they should perform to keep their skills up, and when and how to use imaging and prescribe DOACs, among other suggestions.
The statement represents the first updated guidance for LAAC since 2015. “Since then this field has really expanded and evolved,” writing group chair Jacqueline Saw, MD, said in an interview. “For instance, the indications are more matured and specific, and the procedural technical steps have matured. Imaging has also advanced, there’s more understanding about postprocedural care and there are also new devices that have been approved.”
Dr. Saw, an interventional cardiologist at Vancouver General Hospital and St. Paul’s Hospital, and a professor at the University of British Columbia in Vancouver, called the statement “a piece that puts everything together.”
“This document really summarizes the whole practice for doing transcatheter procedures,” she added, “so it’s all-in-one document in terms of recommendation of who we do the procedure for, how we should do it, how we should image and guide the procedure, and what complications to look out for and how to manage patients post procedure, be it with antithrombotic therapy and/or device surveillance.”
13 recommendations
In all, the statement carries 13 recommendations for LAAC. The Society for Cardiovascular Angiography & Interventions and the Heart Rhythm Society commissioned the writing group. The American College of Cardiology and Society of Cardiovascular Computed Tomography have endorsed the statement. The following are among the recommendations:
- Transcatheter LAAC is appropriate for patients with nonvalvular atrial fibrillation with high thromboembolic risk but for whom long-term oral anticoagulation may be contraindicated and who have at least 1 year’s life expectancy.
- Operators should have performed at least 50 prior left-sided ablations or structural procedures and at least 25 transseptal punctures (TSPs). Interventional-imaging physicians should have experience in guiding 25 or more TSPs before supporting LAAC procedures independently.
- To maintain skills, operators should do 25 or more TSPs and at least 12 LAACs over each 2-year period.
- On-site cardiovascular surgery backup should be available for new programs and for operators early in their learning curve.
- Baseline imaging with transesophageal echocardiography (TEE) or cardiac computed tomography should be performed before LAAC.
- Intraprocedural imaging guidance with TEE or intracardiac echocardiography.
- Follow labeling of each specific LAAC device for technical aspects of the procedure.
- Familiarity with avoiding, recognizing, and managing LAAC complications.
- Predischarge 2-dimensional TEE to rule out pericardial effusion and device embolization.
- Anticoagulation for device-related thrombus.
- Make all efforts to minimize peridevice leaks during implantation because their clinical impact and management isn’t well understood.
- Antithrombotic therapy with warfarin, DOAC, or dual-antiplatelet therapy after LAAC based on the studied regimen and instructions for each specific device, tailored to the bleeding risks for each patient.
- TEE or cardiac computed tomography at 45-90 days after LAAC for device surveillance to assess for peridevice leak and device-related thrombus.
The statement also includes precautionary recommendations. It advises against using routine closure of LAAC-associated iatrogenic atrial septal defects and states that combined procedures with LAAC, such as structural interventions and pulmonary vein isolation, should be avoided because randomized controlled trial data are pending.
“These recommendations are based upon data from updated publications and randomized trial data as well as large registries, including the National Cardiovascular Data Registry, so I think this is a very practical statement that puts all these pieces together for any budding interventionalist doing this procedure and even experienced operations,” Dr. Saw said.
Authors of an accompanying editorial agreed that the “rigorous standards” set out in the statement will help maintain “a high level of procedural safety in the setting of rapid expansion.”
The editorialists, Faisal M. Merchant, MD, of Emory University, Atlanta, and Mohamad Alkhouli, MD, professor of medicine at Mayo Clinic School of Medicine, Rochester, Minn., point out that the incidence of pericardial effusion has decreased from more than 5% in the pivotal Watchman trials to less than 1.5% in the most recent report from the National Cardiovascular Data Registry, which shows that more than 100,000 procedures have been performed in the United States.
But most important as the field moves forward, they stress, is patient selection. The recommendation of limiting patients to those with a life expectancy of 1 year “is a tacit recognition of the fact that the benefits of LAAC take time to accrue, and many older and frail patients are unlikely to derive meaningful benefit.”
Dr. Merchant and Dr. Alkhouli also note that there remains a conundrum in patient selection that remains from the original LAAC trials, which enrolled patients who were eligible for anticoagulation. “Somewhat paradoxically, after its approval, LAAC is mostly prescribed to patients who are not felt to be good anticoagulation candidates.” This leaves physicians “in the precarious position of extrapolating data to patients who were excluded from the original clinical trials.”
Therefore, the consensus statement “is right to put patient selection front and center in its recommendations, but as the field of LAAC comes of age, better evidence to support patient selection will be the real sign of maturity.”
Dr. Saw said she envisions another update over the next 2 years or so as ongoing clinical trials comparing DOAC and LAAC, namely the CHAMPION-AF and OPTION trials, report results.
Dr. Saw and Dr. Merchant, reported no conflicts of interest. Dr. Alkhouli has financial ties to Boston Scientific, Abbott, and Philips.
FROM THE JOURNAL OF THE SOCIETY FOR CARDIOVASCULAR ANGIOGRAPHY & INTERVENTIONS
New AHA statement on pediatric primary hypertension issued
the American Heart Association said in a new scientific statement.
“Children can have secondary hypertension that is caused by an underlying condition such as chronic kidney disease, endocrine disorders, cardiac anomalies, and some syndromes. However, primary hypertension is now recognized as the most common type of hypertension in childhood,” Bonita Falkner, MD, chair of the writing group and emeritus professor of medicine and pediatrics, Thomas Jefferson University, Philadelphia, said in an interview.
And hypertensive children are “highly likely” to become hypertensive adults and to have measurable target organ injury, particularly left ventricular hypertrophy and vascular stiffening, the writing group noted.
The AHA statement on primary pediatric hypertension was published online in Hypertension.
Primary or essential hypertension occurs in up to 5% of children and adolescents in the United States and other countries.
The American Academy of Pediatrics (AAP), European Society of Hypertension and Hypertension Canada all define hypertension as repeated BP readings at or above the 95th percentile for children, but the thresholds differ by age.
The AAP adopts 130/80 mm Hg starting at age 13 years; the European Society of Hypertension adopts 140/90 mm Hg starting at age 16 years; and Hypertension Canada adopts 120/80 mm Hg for those aged 6-11 years and 130/85 mm Hg for those aged 12-17 years.
Adolescents entering adulthood with a BP < 120/80 mm Hg is an optimal goal, the writing group advised.
They recommend that health care professionals be trained on evidence-based methods to obtain accurate and reliable BP values with either auscultatory or oscillometric methods.
When the initial BP measurement is abnormal, repeat measurement by auscultation is recommended, within the same visit if possible, and then within weeks if the screening BP is hypertensive, or months if the screening BP is elevated.
Because BP levels are variable, even within a single visit, “best practice” is to obtain up to three BP measurements and to record the average of the latter two measurements unless the first measurement is normal, the writing group said. Further confirmation of diagnosis of hypertension can be obtained with 24-hour ambulatory BP monitoring (ABPM).
“Primary hypertension in youth is difficult to recognize in asymptomatic, otherwise healthy youth. There is now evidence that children and adolescents with primary hypertension may also have cardiac and vascular injury due to the hypertension,” Dr. Falkner told this news organization.
“If not identified and treated, the condition can progress to hypertension in young adulthood with heightened risk of premature cardiovascular events,” Dr. Falkner said.
The writing group said “primordial prevention” is an important public health goal because a population with lower BP will have fewer comorbidities related to hypertension and CVD.
Modifiable risk factors for primary hypertension in childhood include obesity, physical inactivity and poor diet/nutrition, disturbed sleep patterns, and environmental stress.
A healthy lifestyle in childhood – including eating healthy food, encouraging physical activity that leads to improved physical fitness and healthy sleep, and avoiding the development of obesity – may help mitigate the risk of hypertension in childhood, the writing group noted.
Looking ahead, they said efforts to improve recognition and diagnosis of high BP in children, as well as clinical trials to evaluate medical treatment and recommend public health initiatives, are all vital to combat rising rates of primary hypertension in children.
This scientific statement was prepared by the volunteer writing group on behalf of the American Heart Association’s Council on Hypertension, the Council on Lifelong Congenital Heart Disease and Heart Health in the Young, the Council on Kidney in Cardiovascular Disease, the Council on Lifestyle and Cardiometabolic Health, and the Council on Cardiovascular and Stroke Nursing.
A version of this article first appeared on Medscape.com.
the American Heart Association said in a new scientific statement.
“Children can have secondary hypertension that is caused by an underlying condition such as chronic kidney disease, endocrine disorders, cardiac anomalies, and some syndromes. However, primary hypertension is now recognized as the most common type of hypertension in childhood,” Bonita Falkner, MD, chair of the writing group and emeritus professor of medicine and pediatrics, Thomas Jefferson University, Philadelphia, said in an interview.
And hypertensive children are “highly likely” to become hypertensive adults and to have measurable target organ injury, particularly left ventricular hypertrophy and vascular stiffening, the writing group noted.
The AHA statement on primary pediatric hypertension was published online in Hypertension.
Primary or essential hypertension occurs in up to 5% of children and adolescents in the United States and other countries.
The American Academy of Pediatrics (AAP), European Society of Hypertension and Hypertension Canada all define hypertension as repeated BP readings at or above the 95th percentile for children, but the thresholds differ by age.
The AAP adopts 130/80 mm Hg starting at age 13 years; the European Society of Hypertension adopts 140/90 mm Hg starting at age 16 years; and Hypertension Canada adopts 120/80 mm Hg for those aged 6-11 years and 130/85 mm Hg for those aged 12-17 years.
Adolescents entering adulthood with a BP < 120/80 mm Hg is an optimal goal, the writing group advised.
They recommend that health care professionals be trained on evidence-based methods to obtain accurate and reliable BP values with either auscultatory or oscillometric methods.
When the initial BP measurement is abnormal, repeat measurement by auscultation is recommended, within the same visit if possible, and then within weeks if the screening BP is hypertensive, or months if the screening BP is elevated.
Because BP levels are variable, even within a single visit, “best practice” is to obtain up to three BP measurements and to record the average of the latter two measurements unless the first measurement is normal, the writing group said. Further confirmation of diagnosis of hypertension can be obtained with 24-hour ambulatory BP monitoring (ABPM).
“Primary hypertension in youth is difficult to recognize in asymptomatic, otherwise healthy youth. There is now evidence that children and adolescents with primary hypertension may also have cardiac and vascular injury due to the hypertension,” Dr. Falkner told this news organization.
“If not identified and treated, the condition can progress to hypertension in young adulthood with heightened risk of premature cardiovascular events,” Dr. Falkner said.
The writing group said “primordial prevention” is an important public health goal because a population with lower BP will have fewer comorbidities related to hypertension and CVD.
Modifiable risk factors for primary hypertension in childhood include obesity, physical inactivity and poor diet/nutrition, disturbed sleep patterns, and environmental stress.
A healthy lifestyle in childhood – including eating healthy food, encouraging physical activity that leads to improved physical fitness and healthy sleep, and avoiding the development of obesity – may help mitigate the risk of hypertension in childhood, the writing group noted.
Looking ahead, they said efforts to improve recognition and diagnosis of high BP in children, as well as clinical trials to evaluate medical treatment and recommend public health initiatives, are all vital to combat rising rates of primary hypertension in children.
This scientific statement was prepared by the volunteer writing group on behalf of the American Heart Association’s Council on Hypertension, the Council on Lifelong Congenital Heart Disease and Heart Health in the Young, the Council on Kidney in Cardiovascular Disease, the Council on Lifestyle and Cardiometabolic Health, and the Council on Cardiovascular and Stroke Nursing.
A version of this article first appeared on Medscape.com.
the American Heart Association said in a new scientific statement.
“Children can have secondary hypertension that is caused by an underlying condition such as chronic kidney disease, endocrine disorders, cardiac anomalies, and some syndromes. However, primary hypertension is now recognized as the most common type of hypertension in childhood,” Bonita Falkner, MD, chair of the writing group and emeritus professor of medicine and pediatrics, Thomas Jefferson University, Philadelphia, said in an interview.
And hypertensive children are “highly likely” to become hypertensive adults and to have measurable target organ injury, particularly left ventricular hypertrophy and vascular stiffening, the writing group noted.
The AHA statement on primary pediatric hypertension was published online in Hypertension.
Primary or essential hypertension occurs in up to 5% of children and adolescents in the United States and other countries.
The American Academy of Pediatrics (AAP), European Society of Hypertension and Hypertension Canada all define hypertension as repeated BP readings at or above the 95th percentile for children, but the thresholds differ by age.
The AAP adopts 130/80 mm Hg starting at age 13 years; the European Society of Hypertension adopts 140/90 mm Hg starting at age 16 years; and Hypertension Canada adopts 120/80 mm Hg for those aged 6-11 years and 130/85 mm Hg for those aged 12-17 years.
Adolescents entering adulthood with a BP < 120/80 mm Hg is an optimal goal, the writing group advised.
They recommend that health care professionals be trained on evidence-based methods to obtain accurate and reliable BP values with either auscultatory or oscillometric methods.
When the initial BP measurement is abnormal, repeat measurement by auscultation is recommended, within the same visit if possible, and then within weeks if the screening BP is hypertensive, or months if the screening BP is elevated.
Because BP levels are variable, even within a single visit, “best practice” is to obtain up to three BP measurements and to record the average of the latter two measurements unless the first measurement is normal, the writing group said. Further confirmation of diagnosis of hypertension can be obtained with 24-hour ambulatory BP monitoring (ABPM).
“Primary hypertension in youth is difficult to recognize in asymptomatic, otherwise healthy youth. There is now evidence that children and adolescents with primary hypertension may also have cardiac and vascular injury due to the hypertension,” Dr. Falkner told this news organization.
“If not identified and treated, the condition can progress to hypertension in young adulthood with heightened risk of premature cardiovascular events,” Dr. Falkner said.
The writing group said “primordial prevention” is an important public health goal because a population with lower BP will have fewer comorbidities related to hypertension and CVD.
Modifiable risk factors for primary hypertension in childhood include obesity, physical inactivity and poor diet/nutrition, disturbed sleep patterns, and environmental stress.
A healthy lifestyle in childhood – including eating healthy food, encouraging physical activity that leads to improved physical fitness and healthy sleep, and avoiding the development of obesity – may help mitigate the risk of hypertension in childhood, the writing group noted.
Looking ahead, they said efforts to improve recognition and diagnosis of high BP in children, as well as clinical trials to evaluate medical treatment and recommend public health initiatives, are all vital to combat rising rates of primary hypertension in children.
This scientific statement was prepared by the volunteer writing group on behalf of the American Heart Association’s Council on Hypertension, the Council on Lifelong Congenital Heart Disease and Heart Health in the Young, the Council on Kidney in Cardiovascular Disease, the Council on Lifestyle and Cardiometabolic Health, and the Council on Cardiovascular and Stroke Nursing.
A version of this article first appeared on Medscape.com.
FROM HYPERTENSION
Specific brain damage links hypertension to cognitive impairment
Researchers have identified specific regions of the brain that appear to be damaged by high blood pressure. The finding may explain the link between hypertension and cognitive impairment.
They used genetic information from genome-wide association studies (GWASs) and MRI scans of the brain to study the relationship between hypertension, changes in brain structures, and cognitive impairment. Using Mendelian randomization techniques, they identified nine brain structures related to cognitive impairment that are affected by blood pressure.
“We knew before that raised blood pressure was related to changes in the brain, but our research has narrowed down the changes to those that appear to be potentially causally related to cognitive impairment,” senior author Tomasz Guzik, professor of cardiovascular medicine, at the University of Edinburgh and of the Jagiellonian University, Krakow, Poland, told this news organization.
“Our study confirms a potentially causal relationship between raised blood pressure and cognitive impairment, emphasizing the importance of preventing and treating hypertension,” Prof. Guzik noted.
“But it also identifies the brain culprits of this relationship,” he added.
In the future, it may be possible to assess these nine brain structures in people with high blood pressure to identify those at increased risk of developing cognitive impairment, he said. “These patients may need more intensive care for their blood pressure. We can also investigate these brain structures for potential signaling pathways and molecular changes to see if we can find new targets for treatment to prevent cognitive impairment.”
For this report, the investigators married together different research datasets to identify brain structures potentially responsible for the effects of blood pressure on cognitive function, using results from previous GWASs and observational data from 39,000 people in the UK Biobank registry for whom brain MRI data were available.
First, they mapped brain structures potentially influenced by blood pressure in midlife using MRI scans from people in the UK Biobank registry. Then they examined the relationship between blood pressure and cognitive function in the UK Biobank registry. Next, of the brain structures affected by blood pressure, they identified those that are causally linked to cognitive impairment.
This was possible thanks to genetic markers coding for increased blood pressure, brain structure imaging phenotypes, and those coding for cognitive impairment that could be used in Mendelian randomization studies.
“We looked at 3935 brain magnetic resonance imaging–derived phenotypes in the brain and cognitive function defined by fluid intelligence score to identify genetically predicted causal relationships,” Prof. Guzik said.
They identified 200 brain structures that were causally affected by systolic blood pressure. Of these, nine were also causally related to cognitive impairment. The results were validated in a second prospective cohort of patients with hypertension.
“Some of these structures, including putamen and the white matter regions spanning between the anterior corona radiata, anterior thalamic radiation, and anterior limb of the internal capsule, may represent the target brain regions at which systolic blood pressure acts on cognitive function,” the authors comment.
In an accompanying editorial, Ernesto Schiffrin, MD, and James Engert, PhD, McGill University, Montreal, say that further mechanistic studies of the effects of blood pressure on cognitive function are required to determine precise causal pathways and the roles of relevant brain regions.
“Eventually, biomarkers could be developed to inform antihypertensive trials. Whether clinical trials targeting the specific brain structures will be feasible or if specific antihypertensives could be found that target specific structures remains to be demonstrated,” they write.
“Thus, these new studies could lead to an understanding of the signaling pathways that explain how these structures relate vascular damage to cognitive impairment in hypertension, and contribute to the development of novel interventions to more successfully address the scourge of cognitive decline and dementia in the future,” the editorialists conclude.
The study was funded by the European Research Council, the British Heart Foundation, and the Italian Ministry of Health.
A version of this article first appeared on Medscape.com.
Researchers have identified specific regions of the brain that appear to be damaged by high blood pressure. The finding may explain the link between hypertension and cognitive impairment.
They used genetic information from genome-wide association studies (GWASs) and MRI scans of the brain to study the relationship between hypertension, changes in brain structures, and cognitive impairment. Using Mendelian randomization techniques, they identified nine brain structures related to cognitive impairment that are affected by blood pressure.
“We knew before that raised blood pressure was related to changes in the brain, but our research has narrowed down the changes to those that appear to be potentially causally related to cognitive impairment,” senior author Tomasz Guzik, professor of cardiovascular medicine, at the University of Edinburgh and of the Jagiellonian University, Krakow, Poland, told this news organization.
“Our study confirms a potentially causal relationship between raised blood pressure and cognitive impairment, emphasizing the importance of preventing and treating hypertension,” Prof. Guzik noted.
“But it also identifies the brain culprits of this relationship,” he added.
In the future, it may be possible to assess these nine brain structures in people with high blood pressure to identify those at increased risk of developing cognitive impairment, he said. “These patients may need more intensive care for their blood pressure. We can also investigate these brain structures for potential signaling pathways and molecular changes to see if we can find new targets for treatment to prevent cognitive impairment.”
For this report, the investigators married together different research datasets to identify brain structures potentially responsible for the effects of blood pressure on cognitive function, using results from previous GWASs and observational data from 39,000 people in the UK Biobank registry for whom brain MRI data were available.
First, they mapped brain structures potentially influenced by blood pressure in midlife using MRI scans from people in the UK Biobank registry. Then they examined the relationship between blood pressure and cognitive function in the UK Biobank registry. Next, of the brain structures affected by blood pressure, they identified those that are causally linked to cognitive impairment.
This was possible thanks to genetic markers coding for increased blood pressure, brain structure imaging phenotypes, and those coding for cognitive impairment that could be used in Mendelian randomization studies.
“We looked at 3935 brain magnetic resonance imaging–derived phenotypes in the brain and cognitive function defined by fluid intelligence score to identify genetically predicted causal relationships,” Prof. Guzik said.
They identified 200 brain structures that were causally affected by systolic blood pressure. Of these, nine were also causally related to cognitive impairment. The results were validated in a second prospective cohort of patients with hypertension.
“Some of these structures, including putamen and the white matter regions spanning between the anterior corona radiata, anterior thalamic radiation, and anterior limb of the internal capsule, may represent the target brain regions at which systolic blood pressure acts on cognitive function,” the authors comment.
In an accompanying editorial, Ernesto Schiffrin, MD, and James Engert, PhD, McGill University, Montreal, say that further mechanistic studies of the effects of blood pressure on cognitive function are required to determine precise causal pathways and the roles of relevant brain regions.
“Eventually, biomarkers could be developed to inform antihypertensive trials. Whether clinical trials targeting the specific brain structures will be feasible or if specific antihypertensives could be found that target specific structures remains to be demonstrated,” they write.
“Thus, these new studies could lead to an understanding of the signaling pathways that explain how these structures relate vascular damage to cognitive impairment in hypertension, and contribute to the development of novel interventions to more successfully address the scourge of cognitive decline and dementia in the future,” the editorialists conclude.
The study was funded by the European Research Council, the British Heart Foundation, and the Italian Ministry of Health.
A version of this article first appeared on Medscape.com.
Researchers have identified specific regions of the brain that appear to be damaged by high blood pressure. The finding may explain the link between hypertension and cognitive impairment.
They used genetic information from genome-wide association studies (GWASs) and MRI scans of the brain to study the relationship between hypertension, changes in brain structures, and cognitive impairment. Using Mendelian randomization techniques, they identified nine brain structures related to cognitive impairment that are affected by blood pressure.
“We knew before that raised blood pressure was related to changes in the brain, but our research has narrowed down the changes to those that appear to be potentially causally related to cognitive impairment,” senior author Tomasz Guzik, professor of cardiovascular medicine, at the University of Edinburgh and of the Jagiellonian University, Krakow, Poland, told this news organization.
“Our study confirms a potentially causal relationship between raised blood pressure and cognitive impairment, emphasizing the importance of preventing and treating hypertension,” Prof. Guzik noted.
“But it also identifies the brain culprits of this relationship,” he added.
In the future, it may be possible to assess these nine brain structures in people with high blood pressure to identify those at increased risk of developing cognitive impairment, he said. “These patients may need more intensive care for their blood pressure. We can also investigate these brain structures for potential signaling pathways and molecular changes to see if we can find new targets for treatment to prevent cognitive impairment.”
For this report, the investigators married together different research datasets to identify brain structures potentially responsible for the effects of blood pressure on cognitive function, using results from previous GWASs and observational data from 39,000 people in the UK Biobank registry for whom brain MRI data were available.
First, they mapped brain structures potentially influenced by blood pressure in midlife using MRI scans from people in the UK Biobank registry. Then they examined the relationship between blood pressure and cognitive function in the UK Biobank registry. Next, of the brain structures affected by blood pressure, they identified those that are causally linked to cognitive impairment.
This was possible thanks to genetic markers coding for increased blood pressure, brain structure imaging phenotypes, and those coding for cognitive impairment that could be used in Mendelian randomization studies.
“We looked at 3935 brain magnetic resonance imaging–derived phenotypes in the brain and cognitive function defined by fluid intelligence score to identify genetically predicted causal relationships,” Prof. Guzik said.
They identified 200 brain structures that were causally affected by systolic blood pressure. Of these, nine were also causally related to cognitive impairment. The results were validated in a second prospective cohort of patients with hypertension.
“Some of these structures, including putamen and the white matter regions spanning between the anterior corona radiata, anterior thalamic radiation, and anterior limb of the internal capsule, may represent the target brain regions at which systolic blood pressure acts on cognitive function,” the authors comment.
In an accompanying editorial, Ernesto Schiffrin, MD, and James Engert, PhD, McGill University, Montreal, say that further mechanistic studies of the effects of blood pressure on cognitive function are required to determine precise causal pathways and the roles of relevant brain regions.
“Eventually, biomarkers could be developed to inform antihypertensive trials. Whether clinical trials targeting the specific brain structures will be feasible or if specific antihypertensives could be found that target specific structures remains to be demonstrated,” they write.
“Thus, these new studies could lead to an understanding of the signaling pathways that explain how these structures relate vascular damage to cognitive impairment in hypertension, and contribute to the development of novel interventions to more successfully address the scourge of cognitive decline and dementia in the future,” the editorialists conclude.
The study was funded by the European Research Council, the British Heart Foundation, and the Italian Ministry of Health.
A version of this article first appeared on Medscape.com.
AHA, ACC push supervised exercise training for HFpEF
A statement released by the American Heart Association and the American College of Cardiology advocates use of supervised exercise training in patients with heart failure with preserved ejection fraction (HFpEF), as well as coverage for these services by third-party payers.
The authors hope to boost the stature of supervised exercise training (SET) in HFpEF among practitioners and show Medicare and insurers that it deserves reimbursement. Currently, they noted, clinicians tend to recognize exercise as therapy more in HF with reduced ejection fraction (HFrEF). And Medicare covers exercise training within broader cardiac rehabilitation programs for patients with HFrEF but not HFpEF.
Yet exercise has been broadly effective in HFpEF clinical trials, as outlined in the document. And there are good mechanistic reasons to believe that patients with the disorder can gain as much or more from SET than those with HFrEF.
“The signals for improvement from exercise training, in symptoms and objective measures of exercise capacity, are considerably larger for HFpEF than for HFrEF,” Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C., said in an interview.
So, it’s a bit of a paradox that clinicians don’t prescribe it as often in HFpEF, probably because of the lack of reimbursement but also from less “awareness” and understanding of the disease itself, he proposed.
Dr. Kitzman is senior author on the statement sponsored by the AHA and the ACC. It was published in the societies’ flagship journals Circulation and the Journal of the American College of Cardiology. The statement was also endorsed by the Heart Failure Society of America, the American Association of Cardiovascular and Pulmonary Rehabilitation, and the American Association of Heart Failure Nurses.
Carefully chosen words
The statement makes its case in HFpEF specifically for SET rather than cardiac rehabilitation, the latter typically a comprehensive program that goes beyond exercise, Dr. Kitzman noted. And SET is closer to the exercise interventions used in the supportive HFpEF trials.
“Also, Medicare in recent years has approved something called ‘supervised exercise training’ for other disorders, such as peripheral artery disease.” So, the document specifies SET “to be fully aligned with the evidence base,” he said, as well as “align it with a type of treatment that Medicare has a precedent for approving for other disorders.”
Data and physiologic basis
Core features of the AHA/ACC statement is its review of HFpEF exercise physiology, survey of randomized trials supporting SET in the disease, and characterization of exercise as an especially suitable pleiotropic therapy.
Increasingly, “HFpEF is now accepted as a systemic disorder that affects and impacts all organs,” Dr. Kitzman observed. “With a systemic multiorgan disorder, it would make sense that a broad treatment like exercise might be just the right thing. We think that’s the reason that its benefits are really quite large in magnitude.”
The document notes that exercise seems “potentially well suited for the treatment of both the cardiac and, in particular, the extracardiac abnormalities that contribute to exercise intolerance in HFpEF.”
Its effects in the disorder are “anti-inflammatory, rheological, lipid lowering, antihypertensive, positive inotropic, positive lusitropic, negative chronotropic, vasodilation, diuretic, weight-reducing, hypoglycemic, hypnotic, and antidepressive,” the statement notes. It achieves them via multiple pathways involving the heart, lungs, vasculature and, notably, the skeletal muscles.
“It’s been widely overlooked that at least 50% of low exercise capacity and symptoms in HFpEF are due to skeletal muscle dysfunction,” said Dr. Kitzman, an authority on exercise physiology in heart failure.
“But we’ve spent about 95% of our attention trying to modify and understand the cardiac component.” Skeletal muscles, he said, “are not an innocent bystander. They’re part of the problem. And that’s why we should really spend more time focusing on them.”
Dr. Kitzman disclosed receiving consulting fees from Bayer, Medtronic, Corvia Medical, Boehringer Ingelheim, Keyto, Rivus, NovoNordisk, AstraZeneca, and Pfizer; holding stock in Gilead; and receiving grants to his institution from Bayer, Novo Nordisk, AstraZeneca, Rivus, and Pfizer.
A version of this article first appeared on Medscape.com.
A statement released by the American Heart Association and the American College of Cardiology advocates use of supervised exercise training in patients with heart failure with preserved ejection fraction (HFpEF), as well as coverage for these services by third-party payers.
The authors hope to boost the stature of supervised exercise training (SET) in HFpEF among practitioners and show Medicare and insurers that it deserves reimbursement. Currently, they noted, clinicians tend to recognize exercise as therapy more in HF with reduced ejection fraction (HFrEF). And Medicare covers exercise training within broader cardiac rehabilitation programs for patients with HFrEF but not HFpEF.
Yet exercise has been broadly effective in HFpEF clinical trials, as outlined in the document. And there are good mechanistic reasons to believe that patients with the disorder can gain as much or more from SET than those with HFrEF.
“The signals for improvement from exercise training, in symptoms and objective measures of exercise capacity, are considerably larger for HFpEF than for HFrEF,” Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C., said in an interview.
So, it’s a bit of a paradox that clinicians don’t prescribe it as often in HFpEF, probably because of the lack of reimbursement but also from less “awareness” and understanding of the disease itself, he proposed.
Dr. Kitzman is senior author on the statement sponsored by the AHA and the ACC. It was published in the societies’ flagship journals Circulation and the Journal of the American College of Cardiology. The statement was also endorsed by the Heart Failure Society of America, the American Association of Cardiovascular and Pulmonary Rehabilitation, and the American Association of Heart Failure Nurses.
Carefully chosen words
The statement makes its case in HFpEF specifically for SET rather than cardiac rehabilitation, the latter typically a comprehensive program that goes beyond exercise, Dr. Kitzman noted. And SET is closer to the exercise interventions used in the supportive HFpEF trials.
“Also, Medicare in recent years has approved something called ‘supervised exercise training’ for other disorders, such as peripheral artery disease.” So, the document specifies SET “to be fully aligned with the evidence base,” he said, as well as “align it with a type of treatment that Medicare has a precedent for approving for other disorders.”
Data and physiologic basis
Core features of the AHA/ACC statement is its review of HFpEF exercise physiology, survey of randomized trials supporting SET in the disease, and characterization of exercise as an especially suitable pleiotropic therapy.
Increasingly, “HFpEF is now accepted as a systemic disorder that affects and impacts all organs,” Dr. Kitzman observed. “With a systemic multiorgan disorder, it would make sense that a broad treatment like exercise might be just the right thing. We think that’s the reason that its benefits are really quite large in magnitude.”
The document notes that exercise seems “potentially well suited for the treatment of both the cardiac and, in particular, the extracardiac abnormalities that contribute to exercise intolerance in HFpEF.”
Its effects in the disorder are “anti-inflammatory, rheological, lipid lowering, antihypertensive, positive inotropic, positive lusitropic, negative chronotropic, vasodilation, diuretic, weight-reducing, hypoglycemic, hypnotic, and antidepressive,” the statement notes. It achieves them via multiple pathways involving the heart, lungs, vasculature and, notably, the skeletal muscles.
“It’s been widely overlooked that at least 50% of low exercise capacity and symptoms in HFpEF are due to skeletal muscle dysfunction,” said Dr. Kitzman, an authority on exercise physiology in heart failure.
“But we’ve spent about 95% of our attention trying to modify and understand the cardiac component.” Skeletal muscles, he said, “are not an innocent bystander. They’re part of the problem. And that’s why we should really spend more time focusing on them.”
Dr. Kitzman disclosed receiving consulting fees from Bayer, Medtronic, Corvia Medical, Boehringer Ingelheim, Keyto, Rivus, NovoNordisk, AstraZeneca, and Pfizer; holding stock in Gilead; and receiving grants to his institution from Bayer, Novo Nordisk, AstraZeneca, Rivus, and Pfizer.
A version of this article first appeared on Medscape.com.
A statement released by the American Heart Association and the American College of Cardiology advocates use of supervised exercise training in patients with heart failure with preserved ejection fraction (HFpEF), as well as coverage for these services by third-party payers.
The authors hope to boost the stature of supervised exercise training (SET) in HFpEF among practitioners and show Medicare and insurers that it deserves reimbursement. Currently, they noted, clinicians tend to recognize exercise as therapy more in HF with reduced ejection fraction (HFrEF). And Medicare covers exercise training within broader cardiac rehabilitation programs for patients with HFrEF but not HFpEF.
Yet exercise has been broadly effective in HFpEF clinical trials, as outlined in the document. And there are good mechanistic reasons to believe that patients with the disorder can gain as much or more from SET than those with HFrEF.
“The signals for improvement from exercise training, in symptoms and objective measures of exercise capacity, are considerably larger for HFpEF than for HFrEF,” Dalane W. Kitzman, MD, Wake Forest University, Winston-Salem, N.C., said in an interview.
So, it’s a bit of a paradox that clinicians don’t prescribe it as often in HFpEF, probably because of the lack of reimbursement but also from less “awareness” and understanding of the disease itself, he proposed.
Dr. Kitzman is senior author on the statement sponsored by the AHA and the ACC. It was published in the societies’ flagship journals Circulation and the Journal of the American College of Cardiology. The statement was also endorsed by the Heart Failure Society of America, the American Association of Cardiovascular and Pulmonary Rehabilitation, and the American Association of Heart Failure Nurses.
Carefully chosen words
The statement makes its case in HFpEF specifically for SET rather than cardiac rehabilitation, the latter typically a comprehensive program that goes beyond exercise, Dr. Kitzman noted. And SET is closer to the exercise interventions used in the supportive HFpEF trials.
“Also, Medicare in recent years has approved something called ‘supervised exercise training’ for other disorders, such as peripheral artery disease.” So, the document specifies SET “to be fully aligned with the evidence base,” he said, as well as “align it with a type of treatment that Medicare has a precedent for approving for other disorders.”
Data and physiologic basis
Core features of the AHA/ACC statement is its review of HFpEF exercise physiology, survey of randomized trials supporting SET in the disease, and characterization of exercise as an especially suitable pleiotropic therapy.
Increasingly, “HFpEF is now accepted as a systemic disorder that affects and impacts all organs,” Dr. Kitzman observed. “With a systemic multiorgan disorder, it would make sense that a broad treatment like exercise might be just the right thing. We think that’s the reason that its benefits are really quite large in magnitude.”
The document notes that exercise seems “potentially well suited for the treatment of both the cardiac and, in particular, the extracardiac abnormalities that contribute to exercise intolerance in HFpEF.”
Its effects in the disorder are “anti-inflammatory, rheological, lipid lowering, antihypertensive, positive inotropic, positive lusitropic, negative chronotropic, vasodilation, diuretic, weight-reducing, hypoglycemic, hypnotic, and antidepressive,” the statement notes. It achieves them via multiple pathways involving the heart, lungs, vasculature and, notably, the skeletal muscles.
“It’s been widely overlooked that at least 50% of low exercise capacity and symptoms in HFpEF are due to skeletal muscle dysfunction,” said Dr. Kitzman, an authority on exercise physiology in heart failure.
“But we’ve spent about 95% of our attention trying to modify and understand the cardiac component.” Skeletal muscles, he said, “are not an innocent bystander. They’re part of the problem. And that’s why we should really spend more time focusing on them.”
Dr. Kitzman disclosed receiving consulting fees from Bayer, Medtronic, Corvia Medical, Boehringer Ingelheim, Keyto, Rivus, NovoNordisk, AstraZeneca, and Pfizer; holding stock in Gilead; and receiving grants to his institution from Bayer, Novo Nordisk, AstraZeneca, Rivus, and Pfizer.
A version of this article first appeared on Medscape.com.