User login
Transgender hormone therapy linked to blood pressure changes
Transgender people treated with gender-affirming hormone therapy show distinctive changes in blood pressure that begin soon after treatment initiation and do not subside over years of treatment, according to the largest and longest observational study to date to look at the issue.
“Many physicians may not be aware of the changes to blood pressure in trans patients who start hormone therapy,” senior author Michael S. Irwig, MD, director of transgender medicine at Beth Israel Deaconess Medical Center in Boston, told this news organization.
“The take-away message for physicians is to monitor blood pressure both before and after starting hormone therapy in transgender patients, as over a third of transgender individuals had stage 1 hypertension before starting hormone therapy, and many had their blood pressure increase after starting hormone therapy.”
Mean blood pressure increases in transgender males, decreases in females
In the study, published in Hypertension, Katherine Banks, MD, George Washington University, Washington, and colleagues, followed 470 transgender adult patients for up to 5 years.
The mean systolic blood pressure levels in transgender female patients (male at birth) significantly decreased compared with baseline within a few months of them starting gender-affirming hormone treatment.
Conversely, the systolic blood pressure levels in transgender males (females at birth) who were treated with testosterone increased over the same period.
There were no significant changes in the groups in terms of diastolic blood pressure, consistent with other studies.
“Our study is the first to describe the time course of the blood pressure effects of gender-affirming hormone therapy and to compare the rates of elevated blood pressure and stage 1 and stage 2 hypertension using blood pressure readings from gender-diverse individuals pre- and post–gender-affirming hormone therapy,” the authors note.
Gender-affirming hormone therapy – which has been prescribed to transgender patients for more than 25 years – typically involves a combination of estrogen and an anti-androgen for males transitioning to female, while the therapy for those transitioning to male generally only involves testosterone.
The therapy has previously been linked to various cardiac effects, with evidence showing transgender men have as much as a 5-times greater risk of heart attack versus cisgender women, the authors note.
Although the American Heart Association issued a 2020 Scientific Statement addressing the cardiovascular disease risk, evidence on the effects specifically on blood pressure in transgender patients has been inconsistent.
For the new study, Dr. Banks and colleagues enrolled 247 transgender females and 223 transgender males who were treated between 2007 and 2015 at two medical centers in Washington, D.C. Of the individuals, who had a mean age of 27.8, about 27% were non-White and 16% were Latinx.
They had blood pressure measurements taken at baseline and at follow-up clinical visits for up to 57 months following the initiation of gender-affirming hormone therapy.
Over the follow-up period, the transgender females had decreases in mean systolic blood pressure of 4.0 mm Hg within 2 to 4 months of starting hormone therapy (P < .0001) and mean declines of 6.0 mm Hg were further observed at 11 to 21 months compared with baseline.
In transgender males, the mean systolic blood pressure increased by 2.6 mm Hg at 2 to 4 months (P = .02), and by 2.9 mm Hg at 11 to 21 months after starting therapy.
Furthermore, “although the average increase in systolic blood pressure was 2.6 mm Hg in transgender men within 2 to 4 months, some patients had much higher increases,” Dr. Irwig noted.
As many as 40% of transgender men had stage 1 hypertension after 11 to 21 months of hormone therapy.
The blood pressure changes in transgender males and females were observed across all three racial ethnic groups of Whites, Blacks, and Latinx, and the changes remained consistent throughout the entire follow-up period of approximately 5 years while hormone therapy was continued.
In addition to the changes after therapy initiation, the researchers note that more than one-third of individuals in both groups had stage 1 hypertension even before starting hormone therapy.
The findings are a concern in light of “clear evidence linking hypertension and higher blood pressure with cardiovascular events such as stroke and heart attacks,” Dr. Irwig said.
Protective effects for transgender females?
Transgender females showed as much as a 47% decrease in the prevalence of stage 2 hypertension, from 19% to 10%, within 2 to 4 months of treatment with gender-affirming hormone therapy (P = .001), and the rate declined further to 8% at 11 to 21 months, suggesting a protective effect of the treatment.
“The rate of stage 2 hypertension did drop in transgender feminine individuals, which could be protective and lower their risk for cardiovascular events,” Dr. Irwig said.
“This was not a surprise, as lowering testosterone and the use of spironolactone can lower blood pressure,” he noted.
Exceptions in both groups
Of note, a sizable proportion of patients had blood pressure changes that were in fact the opposite of the patterns seen in the majority of their gender group.
Specifically, while 42% to 53% of the transgender females had systolic blood pressure readings of at least 5 mm Hg lower than their baseline readings, up to 32% had increases of at least 5 mm Hg compared to baseline readings.
Likewise, whereas 41% to 59% of transgender males had increases of at least 5 mm Hg compared with baseline, up to 35% had levels that were at least 5 mm Hg lower than baseline.
“It was a surprise that over a quarter of individuals had changes opposite to the mean changes,” Dr. Irwig said.
The differing blood pressure changes underscore that “more research is needed to determine which formulations of estrogen, testosterone, and antiandrogens are optimal regarding blood pressure and cardiovascular health, especially in older individuals,” the authors note.
Gender-affirming hormone therapy formulations differ
Various formulations for gender-affirming hormone regimens are available, including oral, transdermal, sublingual, and intramuscular preparations.
In the study, 77% to 91% of transgender males were on intramuscular testosterone injections, with the rest on transdermal formulations, and 92% of transgender female patients were started on oral estradiol, with mean doses generally increasing over time.
The study’s results are consistent with evidence from other studies, with 7 of 8 involving transgender males showing mean increases in systolic blood pressure ranging from 1 to 14 mm Hg.
Previous research supports cardiovascular risk
As reported by this news organization, other emerging research on cardiovascular risks to transgender people include a recent study showing more than 10% of transgender males were found to have hematocrit levels that could put them at risk for blood clots.
And further research on transgender youth also shows concerning elevations in lipids and other cardiovascular risks.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Transgender people treated with gender-affirming hormone therapy show distinctive changes in blood pressure that begin soon after treatment initiation and do not subside over years of treatment, according to the largest and longest observational study to date to look at the issue.
“Many physicians may not be aware of the changes to blood pressure in trans patients who start hormone therapy,” senior author Michael S. Irwig, MD, director of transgender medicine at Beth Israel Deaconess Medical Center in Boston, told this news organization.
“The take-away message for physicians is to monitor blood pressure both before and after starting hormone therapy in transgender patients, as over a third of transgender individuals had stage 1 hypertension before starting hormone therapy, and many had their blood pressure increase after starting hormone therapy.”
Mean blood pressure increases in transgender males, decreases in females
In the study, published in Hypertension, Katherine Banks, MD, George Washington University, Washington, and colleagues, followed 470 transgender adult patients for up to 5 years.
The mean systolic blood pressure levels in transgender female patients (male at birth) significantly decreased compared with baseline within a few months of them starting gender-affirming hormone treatment.
Conversely, the systolic blood pressure levels in transgender males (females at birth) who were treated with testosterone increased over the same period.
There were no significant changes in the groups in terms of diastolic blood pressure, consistent with other studies.
“Our study is the first to describe the time course of the blood pressure effects of gender-affirming hormone therapy and to compare the rates of elevated blood pressure and stage 1 and stage 2 hypertension using blood pressure readings from gender-diverse individuals pre- and post–gender-affirming hormone therapy,” the authors note.
Gender-affirming hormone therapy – which has been prescribed to transgender patients for more than 25 years – typically involves a combination of estrogen and an anti-androgen for males transitioning to female, while the therapy for those transitioning to male generally only involves testosterone.
The therapy has previously been linked to various cardiac effects, with evidence showing transgender men have as much as a 5-times greater risk of heart attack versus cisgender women, the authors note.
Although the American Heart Association issued a 2020 Scientific Statement addressing the cardiovascular disease risk, evidence on the effects specifically on blood pressure in transgender patients has been inconsistent.
For the new study, Dr. Banks and colleagues enrolled 247 transgender females and 223 transgender males who were treated between 2007 and 2015 at two medical centers in Washington, D.C. Of the individuals, who had a mean age of 27.8, about 27% were non-White and 16% were Latinx.
They had blood pressure measurements taken at baseline and at follow-up clinical visits for up to 57 months following the initiation of gender-affirming hormone therapy.
Over the follow-up period, the transgender females had decreases in mean systolic blood pressure of 4.0 mm Hg within 2 to 4 months of starting hormone therapy (P < .0001) and mean declines of 6.0 mm Hg were further observed at 11 to 21 months compared with baseline.
In transgender males, the mean systolic blood pressure increased by 2.6 mm Hg at 2 to 4 months (P = .02), and by 2.9 mm Hg at 11 to 21 months after starting therapy.
Furthermore, “although the average increase in systolic blood pressure was 2.6 mm Hg in transgender men within 2 to 4 months, some patients had much higher increases,” Dr. Irwig noted.
As many as 40% of transgender men had stage 1 hypertension after 11 to 21 months of hormone therapy.
The blood pressure changes in transgender males and females were observed across all three racial ethnic groups of Whites, Blacks, and Latinx, and the changes remained consistent throughout the entire follow-up period of approximately 5 years while hormone therapy was continued.
In addition to the changes after therapy initiation, the researchers note that more than one-third of individuals in both groups had stage 1 hypertension even before starting hormone therapy.
The findings are a concern in light of “clear evidence linking hypertension and higher blood pressure with cardiovascular events such as stroke and heart attacks,” Dr. Irwig said.
Protective effects for transgender females?
Transgender females showed as much as a 47% decrease in the prevalence of stage 2 hypertension, from 19% to 10%, within 2 to 4 months of treatment with gender-affirming hormone therapy (P = .001), and the rate declined further to 8% at 11 to 21 months, suggesting a protective effect of the treatment.
“The rate of stage 2 hypertension did drop in transgender feminine individuals, which could be protective and lower their risk for cardiovascular events,” Dr. Irwig said.
“This was not a surprise, as lowering testosterone and the use of spironolactone can lower blood pressure,” he noted.
Exceptions in both groups
Of note, a sizable proportion of patients had blood pressure changes that were in fact the opposite of the patterns seen in the majority of their gender group.
Specifically, while 42% to 53% of the transgender females had systolic blood pressure readings of at least 5 mm Hg lower than their baseline readings, up to 32% had increases of at least 5 mm Hg compared to baseline readings.
Likewise, whereas 41% to 59% of transgender males had increases of at least 5 mm Hg compared with baseline, up to 35% had levels that were at least 5 mm Hg lower than baseline.
“It was a surprise that over a quarter of individuals had changes opposite to the mean changes,” Dr. Irwig said.
The differing blood pressure changes underscore that “more research is needed to determine which formulations of estrogen, testosterone, and antiandrogens are optimal regarding blood pressure and cardiovascular health, especially in older individuals,” the authors note.
Gender-affirming hormone therapy formulations differ
Various formulations for gender-affirming hormone regimens are available, including oral, transdermal, sublingual, and intramuscular preparations.
In the study, 77% to 91% of transgender males were on intramuscular testosterone injections, with the rest on transdermal formulations, and 92% of transgender female patients were started on oral estradiol, with mean doses generally increasing over time.
The study’s results are consistent with evidence from other studies, with 7 of 8 involving transgender males showing mean increases in systolic blood pressure ranging from 1 to 14 mm Hg.
Previous research supports cardiovascular risk
As reported by this news organization, other emerging research on cardiovascular risks to transgender people include a recent study showing more than 10% of transgender males were found to have hematocrit levels that could put them at risk for blood clots.
And further research on transgender youth also shows concerning elevations in lipids and other cardiovascular risks.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Transgender people treated with gender-affirming hormone therapy show distinctive changes in blood pressure that begin soon after treatment initiation and do not subside over years of treatment, according to the largest and longest observational study to date to look at the issue.
“Many physicians may not be aware of the changes to blood pressure in trans patients who start hormone therapy,” senior author Michael S. Irwig, MD, director of transgender medicine at Beth Israel Deaconess Medical Center in Boston, told this news organization.
“The take-away message for physicians is to monitor blood pressure both before and after starting hormone therapy in transgender patients, as over a third of transgender individuals had stage 1 hypertension before starting hormone therapy, and many had their blood pressure increase after starting hormone therapy.”
Mean blood pressure increases in transgender males, decreases in females
In the study, published in Hypertension, Katherine Banks, MD, George Washington University, Washington, and colleagues, followed 470 transgender adult patients for up to 5 years.
The mean systolic blood pressure levels in transgender female patients (male at birth) significantly decreased compared with baseline within a few months of them starting gender-affirming hormone treatment.
Conversely, the systolic blood pressure levels in transgender males (females at birth) who were treated with testosterone increased over the same period.
There were no significant changes in the groups in terms of diastolic blood pressure, consistent with other studies.
“Our study is the first to describe the time course of the blood pressure effects of gender-affirming hormone therapy and to compare the rates of elevated blood pressure and stage 1 and stage 2 hypertension using blood pressure readings from gender-diverse individuals pre- and post–gender-affirming hormone therapy,” the authors note.
Gender-affirming hormone therapy – which has been prescribed to transgender patients for more than 25 years – typically involves a combination of estrogen and an anti-androgen for males transitioning to female, while the therapy for those transitioning to male generally only involves testosterone.
The therapy has previously been linked to various cardiac effects, with evidence showing transgender men have as much as a 5-times greater risk of heart attack versus cisgender women, the authors note.
Although the American Heart Association issued a 2020 Scientific Statement addressing the cardiovascular disease risk, evidence on the effects specifically on blood pressure in transgender patients has been inconsistent.
For the new study, Dr. Banks and colleagues enrolled 247 transgender females and 223 transgender males who were treated between 2007 and 2015 at two medical centers in Washington, D.C. Of the individuals, who had a mean age of 27.8, about 27% were non-White and 16% were Latinx.
They had blood pressure measurements taken at baseline and at follow-up clinical visits for up to 57 months following the initiation of gender-affirming hormone therapy.
Over the follow-up period, the transgender females had decreases in mean systolic blood pressure of 4.0 mm Hg within 2 to 4 months of starting hormone therapy (P < .0001) and mean declines of 6.0 mm Hg were further observed at 11 to 21 months compared with baseline.
In transgender males, the mean systolic blood pressure increased by 2.6 mm Hg at 2 to 4 months (P = .02), and by 2.9 mm Hg at 11 to 21 months after starting therapy.
Furthermore, “although the average increase in systolic blood pressure was 2.6 mm Hg in transgender men within 2 to 4 months, some patients had much higher increases,” Dr. Irwig noted.
As many as 40% of transgender men had stage 1 hypertension after 11 to 21 months of hormone therapy.
The blood pressure changes in transgender males and females were observed across all three racial ethnic groups of Whites, Blacks, and Latinx, and the changes remained consistent throughout the entire follow-up period of approximately 5 years while hormone therapy was continued.
In addition to the changes after therapy initiation, the researchers note that more than one-third of individuals in both groups had stage 1 hypertension even before starting hormone therapy.
The findings are a concern in light of “clear evidence linking hypertension and higher blood pressure with cardiovascular events such as stroke and heart attacks,” Dr. Irwig said.
Protective effects for transgender females?
Transgender females showed as much as a 47% decrease in the prevalence of stage 2 hypertension, from 19% to 10%, within 2 to 4 months of treatment with gender-affirming hormone therapy (P = .001), and the rate declined further to 8% at 11 to 21 months, suggesting a protective effect of the treatment.
“The rate of stage 2 hypertension did drop in transgender feminine individuals, which could be protective and lower their risk for cardiovascular events,” Dr. Irwig said.
“This was not a surprise, as lowering testosterone and the use of spironolactone can lower blood pressure,” he noted.
Exceptions in both groups
Of note, a sizable proportion of patients had blood pressure changes that were in fact the opposite of the patterns seen in the majority of their gender group.
Specifically, while 42% to 53% of the transgender females had systolic blood pressure readings of at least 5 mm Hg lower than their baseline readings, up to 32% had increases of at least 5 mm Hg compared to baseline readings.
Likewise, whereas 41% to 59% of transgender males had increases of at least 5 mm Hg compared with baseline, up to 35% had levels that were at least 5 mm Hg lower than baseline.
“It was a surprise that over a quarter of individuals had changes opposite to the mean changes,” Dr. Irwig said.
The differing blood pressure changes underscore that “more research is needed to determine which formulations of estrogen, testosterone, and antiandrogens are optimal regarding blood pressure and cardiovascular health, especially in older individuals,” the authors note.
Gender-affirming hormone therapy formulations differ
Various formulations for gender-affirming hormone regimens are available, including oral, transdermal, sublingual, and intramuscular preparations.
In the study, 77% to 91% of transgender males were on intramuscular testosterone injections, with the rest on transdermal formulations, and 92% of transgender female patients were started on oral estradiol, with mean doses generally increasing over time.
The study’s results are consistent with evidence from other studies, with 7 of 8 involving transgender males showing mean increases in systolic blood pressure ranging from 1 to 14 mm Hg.
Previous research supports cardiovascular risk
As reported by this news organization, other emerging research on cardiovascular risks to transgender people include a recent study showing more than 10% of transgender males were found to have hematocrit levels that could put them at risk for blood clots.
And further research on transgender youth also shows concerning elevations in lipids and other cardiovascular risks.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cardiologists can perform stroke thrombectomy to fill ‘unmet need’
Cardiologists experienced in cardiac interventions can competently perform stroke thrombectomy after a short period of training, with outcomes comparable to those achieved by neuroradiology centers, a new study suggests.
“Using interventional cardiologists in this way will help address the huge unmet need for stroke thrombectomy that currently exists,” senior author Petr Widimsky, MD, said in an interview.
Although this may be a feasible way forward in Europe, there is strong opposition to such a proposal from U.S. neurointerventionalists.
The study, published in the April 12 issue of JACC: Cardiovascular Interventions, describes the establishment of a stroke thrombectomy program in University Hospital Kralovske Vinohrady, a large tertiary hospital in Prague, Czech Republic.
The hospital did not have a neurointerventional program until 2012 when a joint program was started involving an experienced team of cardiologists, angiologists, and one interventional radiologist who trained the cardiologists on the thrombectomy procedure.
The current paper reports on the outcomes of the 333 patients with large vessel occlusion stroke treated under this program between October 2012 and December 2019.
The decision to perform catheter-based thrombectomy was made by a neurologist and was based on acute stroke clinical symptoms and CT angiographic findings.
Results show that functional clinical outcomes, assessed via the Modified Rankin Scale (mRS) score at 3 months, did not vary significantly across years 2012 to 2019, with a favorable outcome (mRS 0 to 2) achieved in 47.9% of patients.
Symptomatic intracerebral hemorrhage occurred in 19 patients (5.7%) and embolization in a new vascular territory occurred in 6 patients (1.8%), outcomes similar to those of neuroradiology centers.
The desired clinical results were achieved from the onset of the program, without any signs of a learning curve effect, they reported.
“These findings support the potential role of interventional cardiac cath labs in the treatment of acute stroke in regions where this therapy is not readily available due to the lack of neurointerventionalists,” the authors concluded.
“Our main message is that our results were excellent from the beginning,” Dr. Widimsky said. “When centers prepare properly, they can achieve excellent results from the beginning with cardiologists who are experienced in interventional procedures and who have spent sufficient time learning about the brain.”
The authors noted that despite thrombectomy being an extremely beneficial treatment for severe stroke, many eligible patients remain untreated, largely because of a lack of neurointerventionalists in many regions worldwide. They estimate that about 15% of all stroke patients are eligible for thrombectomy but only around 2% of stroke patients in Europe actually receive such treatment.
Dr. Widimsky, an interventional cardiologist, first thought of the idea of using cardiologists to perform stroke thrombectomies after a good friend and colleague suffered a severe stroke in 2010.
“This made us realize that our hospital needed to be more active in the stroke field,” he said. “We decided that we needed to start doing stroke interventions.”
But the major problem was the lack of neurointerventionalists.
“There are not enough neurointerventionalists in Europe. Interventional cardiologists can perform thousands of procedures every year whereas a neurointerventionalist will at best perform hundreds a year. It is quicker and simpler to train the cardiologist to do it,” Dr. Widimsky said.
They hired one neurointerventionalist to lead the program. “He was our tutor, he taught us his skills,” Dr. Widimsky said. “The cath lab is open 24/7, but if we only have one neurointerventionalist we cannot offer a 24/7 service for stroke thrombectomy. But if we merge with cardiology then we can,” he added.
Their hospital is a very busy center for myocardial infarction, percutaneous coronary intervention, and carotid stenting, he noted. “It is not difficult to make the step from that to stroke thrombectomy. Interventional cardiologists are used to performing carotid and coronary artery stenting. Stroke thrombectomy is a similar technique. The thrombectomy procedure is different from coronary angioplasty but it is not more difficult. Actually, I think coronary angioplasty can be more difficult.”
Dr. Widimsky explained that cardiologists need to learn about the brain anatomy and physiology and learn the stroke imaging techniques. “I spent 1 month in the U.S. learning stroke interventions working with simulators,” he said. “I think interventional cardiologists can learn what they need to know in about 6 months. I would recommend they should watch about 50 procedures and perform at least 25 under supervision.”
He said this model is the way forward and hopes it will become routine. Thrombectomy is “tremendously effective” in improving outcomes in severe strokes, with a number needed to treat (NNT) of just 2.6 to prevent long-term disability in one patient, he said, while other procedures can have NNTs of 50 or more.
“But millions of patients with acute severe stroke are not getting this life-changing treatment,” he added. “We must do everything we can to make this service available to as many patients as possible.”
Dr. Widimsky acknowledges that there has been opposition to this idea from the neurointerventionalist professional bodies but this has lessened recently, at least in Europe. And a program that allows interventionalists with experience in extracranial carotid and vertebral endovascular procedures to “fast-track” technical training has now been proposed.
“There is an enormous unmet need for stroke thrombectomy in Europe, with some countries needing to increase the number of procedures done by 10 or 20 times. These include the U.K., Sweden, Italy, Spain, and Portugal. This cannot be done without cardiology,” Dr. Widimsky said.
Editorial strongly supportive
An accompanying editorial strongly endorses the idea of using interdisciplinary teams to deliver high standard stroke care.
Marius Hornung, MD, and Horst Sievert, MD, from CardioVascular Center Frankfurt (Germany), point out that many experienced cardiologists are trained in performing carotid artery interventions and are therefore experienced in accessing the supra-aortic arteries.
“To be able to guarantee optimized stroke therapy as soon as possible, disputes over competence among the individual medical societies involved must be ended,” they wrote.
They advocate for the creation of interdisciplinary teams, with diagnostics, patient selection, and follow-up care remaining the core competencies and tasks of neurology; in addition, they call for appropriately trained and experienced physicians – regardless of their specialties – performing acute stroke interventions and endovascular thrombectomy.
“Such a network must be installed as soon as possible to fulfill the mantra ‘time is brain’ ... and not losing unnecessary time to patient transfer, or continuing to offer only the second-best therapy,” they concluded.
Opposition in the United States
Dr. Widimsky explained that this proposal may not be so applicable to the United States, where the need for more clinicians capable of performing stroke thrombectomies does not appear to be as critical, possibly because vascular neurosurgeons as well as neuroradiologists are qualified to undertake these procedures.
In an interview, J. Mocco, MD, director of the cerebrovascular center, department of neurological surgery, at Mount Sinai Health System, New York, confirmed that this was the case.
“There is no legitimate data to support the claim that there is a lack of an adequate workforce to provide stroke thrombectomy, at least in the U.S.,” he said, adding that, rather, the primary limitation to patient access is a lack of adequate systems of care. “We should learn from the trauma model, which is strongly evidence based, and provide emergency stroke care in a similarly regionalized manner.”
Dr. Mocco, vice president of the Society of NeuroInterventional Surgery, was not impressed with the current study.
“This paper is a retrospective, single-center, unadjudicated, nonindependent assessor case series and therefore, as the authors acknowledge in the limitations section of their paper, it is invalid to compare these data to the results from high-quality, prospective, core-lab, and independent assessor adjudicated randomized trials,” he said. “The supposition that this trial provides evidence that the reported model should be widely considered lacks scientific rigor.”
Furthermore, “the interventional cardiology literature is replete with data regarding the importance of technical expertise and content knowledge,” he added. “Why would that community now propose that such expertise and knowledge is not necessary for the brain?”
Dr. Mocco argues that the concept that interventional cardiologists should be fast-tracked to perform stroke interventions because they use similar tools, navigate blood vessels, and are comfortable working in critical situations, does not hold up.
“Liver surgeons and brain surgeons are both familiar with tissue manipulation, are used to operating in critical situations, and use cautery, scissors, and scalpels; but no one would argue that a brain surgeon should be fast-tracked to perform liver surgery, or vice versa.”
He added: “Stroke patients do not have the luxury of choosing the physician who provides their thrombectomy. We should do everything reasonable to ensure that our systems of care are organized so that these vulnerable patients are treated by physicians who have appropriate knowledge and expertise.”
This study was supported by the Charles University Research program. The authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cardiologists experienced in cardiac interventions can competently perform stroke thrombectomy after a short period of training, with outcomes comparable to those achieved by neuroradiology centers, a new study suggests.
“Using interventional cardiologists in this way will help address the huge unmet need for stroke thrombectomy that currently exists,” senior author Petr Widimsky, MD, said in an interview.
Although this may be a feasible way forward in Europe, there is strong opposition to such a proposal from U.S. neurointerventionalists.
The study, published in the April 12 issue of JACC: Cardiovascular Interventions, describes the establishment of a stroke thrombectomy program in University Hospital Kralovske Vinohrady, a large tertiary hospital in Prague, Czech Republic.
The hospital did not have a neurointerventional program until 2012 when a joint program was started involving an experienced team of cardiologists, angiologists, and one interventional radiologist who trained the cardiologists on the thrombectomy procedure.
The current paper reports on the outcomes of the 333 patients with large vessel occlusion stroke treated under this program between October 2012 and December 2019.
The decision to perform catheter-based thrombectomy was made by a neurologist and was based on acute stroke clinical symptoms and CT angiographic findings.
Results show that functional clinical outcomes, assessed via the Modified Rankin Scale (mRS) score at 3 months, did not vary significantly across years 2012 to 2019, with a favorable outcome (mRS 0 to 2) achieved in 47.9% of patients.
Symptomatic intracerebral hemorrhage occurred in 19 patients (5.7%) and embolization in a new vascular territory occurred in 6 patients (1.8%), outcomes similar to those of neuroradiology centers.
The desired clinical results were achieved from the onset of the program, without any signs of a learning curve effect, they reported.
“These findings support the potential role of interventional cardiac cath labs in the treatment of acute stroke in regions where this therapy is not readily available due to the lack of neurointerventionalists,” the authors concluded.
“Our main message is that our results were excellent from the beginning,” Dr. Widimsky said. “When centers prepare properly, they can achieve excellent results from the beginning with cardiologists who are experienced in interventional procedures and who have spent sufficient time learning about the brain.”
The authors noted that despite thrombectomy being an extremely beneficial treatment for severe stroke, many eligible patients remain untreated, largely because of a lack of neurointerventionalists in many regions worldwide. They estimate that about 15% of all stroke patients are eligible for thrombectomy but only around 2% of stroke patients in Europe actually receive such treatment.
Dr. Widimsky, an interventional cardiologist, first thought of the idea of using cardiologists to perform stroke thrombectomies after a good friend and colleague suffered a severe stroke in 2010.
“This made us realize that our hospital needed to be more active in the stroke field,” he said. “We decided that we needed to start doing stroke interventions.”
But the major problem was the lack of neurointerventionalists.
“There are not enough neurointerventionalists in Europe. Interventional cardiologists can perform thousands of procedures every year whereas a neurointerventionalist will at best perform hundreds a year. It is quicker and simpler to train the cardiologist to do it,” Dr. Widimsky said.
They hired one neurointerventionalist to lead the program. “He was our tutor, he taught us his skills,” Dr. Widimsky said. “The cath lab is open 24/7, but if we only have one neurointerventionalist we cannot offer a 24/7 service for stroke thrombectomy. But if we merge with cardiology then we can,” he added.
Their hospital is a very busy center for myocardial infarction, percutaneous coronary intervention, and carotid stenting, he noted. “It is not difficult to make the step from that to stroke thrombectomy. Interventional cardiologists are used to performing carotid and coronary artery stenting. Stroke thrombectomy is a similar technique. The thrombectomy procedure is different from coronary angioplasty but it is not more difficult. Actually, I think coronary angioplasty can be more difficult.”
Dr. Widimsky explained that cardiologists need to learn about the brain anatomy and physiology and learn the stroke imaging techniques. “I spent 1 month in the U.S. learning stroke interventions working with simulators,” he said. “I think interventional cardiologists can learn what they need to know in about 6 months. I would recommend they should watch about 50 procedures and perform at least 25 under supervision.”
He said this model is the way forward and hopes it will become routine. Thrombectomy is “tremendously effective” in improving outcomes in severe strokes, with a number needed to treat (NNT) of just 2.6 to prevent long-term disability in one patient, he said, while other procedures can have NNTs of 50 or more.
“But millions of patients with acute severe stroke are not getting this life-changing treatment,” he added. “We must do everything we can to make this service available to as many patients as possible.”
Dr. Widimsky acknowledges that there has been opposition to this idea from the neurointerventionalist professional bodies but this has lessened recently, at least in Europe. And a program that allows interventionalists with experience in extracranial carotid and vertebral endovascular procedures to “fast-track” technical training has now been proposed.
“There is an enormous unmet need for stroke thrombectomy in Europe, with some countries needing to increase the number of procedures done by 10 or 20 times. These include the U.K., Sweden, Italy, Spain, and Portugal. This cannot be done without cardiology,” Dr. Widimsky said.
Editorial strongly supportive
An accompanying editorial strongly endorses the idea of using interdisciplinary teams to deliver high standard stroke care.
Marius Hornung, MD, and Horst Sievert, MD, from CardioVascular Center Frankfurt (Germany), point out that many experienced cardiologists are trained in performing carotid artery interventions and are therefore experienced in accessing the supra-aortic arteries.
“To be able to guarantee optimized stroke therapy as soon as possible, disputes over competence among the individual medical societies involved must be ended,” they wrote.
They advocate for the creation of interdisciplinary teams, with diagnostics, patient selection, and follow-up care remaining the core competencies and tasks of neurology; in addition, they call for appropriately trained and experienced physicians – regardless of their specialties – performing acute stroke interventions and endovascular thrombectomy.
“Such a network must be installed as soon as possible to fulfill the mantra ‘time is brain’ ... and not losing unnecessary time to patient transfer, or continuing to offer only the second-best therapy,” they concluded.
Opposition in the United States
Dr. Widimsky explained that this proposal may not be so applicable to the United States, where the need for more clinicians capable of performing stroke thrombectomies does not appear to be as critical, possibly because vascular neurosurgeons as well as neuroradiologists are qualified to undertake these procedures.
In an interview, J. Mocco, MD, director of the cerebrovascular center, department of neurological surgery, at Mount Sinai Health System, New York, confirmed that this was the case.
“There is no legitimate data to support the claim that there is a lack of an adequate workforce to provide stroke thrombectomy, at least in the U.S.,” he said, adding that, rather, the primary limitation to patient access is a lack of adequate systems of care. “We should learn from the trauma model, which is strongly evidence based, and provide emergency stroke care in a similarly regionalized manner.”
Dr. Mocco, vice president of the Society of NeuroInterventional Surgery, was not impressed with the current study.
“This paper is a retrospective, single-center, unadjudicated, nonindependent assessor case series and therefore, as the authors acknowledge in the limitations section of their paper, it is invalid to compare these data to the results from high-quality, prospective, core-lab, and independent assessor adjudicated randomized trials,” he said. “The supposition that this trial provides evidence that the reported model should be widely considered lacks scientific rigor.”
Furthermore, “the interventional cardiology literature is replete with data regarding the importance of technical expertise and content knowledge,” he added. “Why would that community now propose that such expertise and knowledge is not necessary for the brain?”
Dr. Mocco argues that the concept that interventional cardiologists should be fast-tracked to perform stroke interventions because they use similar tools, navigate blood vessels, and are comfortable working in critical situations, does not hold up.
“Liver surgeons and brain surgeons are both familiar with tissue manipulation, are used to operating in critical situations, and use cautery, scissors, and scalpels; but no one would argue that a brain surgeon should be fast-tracked to perform liver surgery, or vice versa.”
He added: “Stroke patients do not have the luxury of choosing the physician who provides their thrombectomy. We should do everything reasonable to ensure that our systems of care are organized so that these vulnerable patients are treated by physicians who have appropriate knowledge and expertise.”
This study was supported by the Charles University Research program. The authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cardiologists experienced in cardiac interventions can competently perform stroke thrombectomy after a short period of training, with outcomes comparable to those achieved by neuroradiology centers, a new study suggests.
“Using interventional cardiologists in this way will help address the huge unmet need for stroke thrombectomy that currently exists,” senior author Petr Widimsky, MD, said in an interview.
Although this may be a feasible way forward in Europe, there is strong opposition to such a proposal from U.S. neurointerventionalists.
The study, published in the April 12 issue of JACC: Cardiovascular Interventions, describes the establishment of a stroke thrombectomy program in University Hospital Kralovske Vinohrady, a large tertiary hospital in Prague, Czech Republic.
The hospital did not have a neurointerventional program until 2012 when a joint program was started involving an experienced team of cardiologists, angiologists, and one interventional radiologist who trained the cardiologists on the thrombectomy procedure.
The current paper reports on the outcomes of the 333 patients with large vessel occlusion stroke treated under this program between October 2012 and December 2019.
The decision to perform catheter-based thrombectomy was made by a neurologist and was based on acute stroke clinical symptoms and CT angiographic findings.
Results show that functional clinical outcomes, assessed via the Modified Rankin Scale (mRS) score at 3 months, did not vary significantly across years 2012 to 2019, with a favorable outcome (mRS 0 to 2) achieved in 47.9% of patients.
Symptomatic intracerebral hemorrhage occurred in 19 patients (5.7%) and embolization in a new vascular territory occurred in 6 patients (1.8%), outcomes similar to those of neuroradiology centers.
The desired clinical results were achieved from the onset of the program, without any signs of a learning curve effect, they reported.
“These findings support the potential role of interventional cardiac cath labs in the treatment of acute stroke in regions where this therapy is not readily available due to the lack of neurointerventionalists,” the authors concluded.
“Our main message is that our results were excellent from the beginning,” Dr. Widimsky said. “When centers prepare properly, they can achieve excellent results from the beginning with cardiologists who are experienced in interventional procedures and who have spent sufficient time learning about the brain.”
The authors noted that despite thrombectomy being an extremely beneficial treatment for severe stroke, many eligible patients remain untreated, largely because of a lack of neurointerventionalists in many regions worldwide. They estimate that about 15% of all stroke patients are eligible for thrombectomy but only around 2% of stroke patients in Europe actually receive such treatment.
Dr. Widimsky, an interventional cardiologist, first thought of the idea of using cardiologists to perform stroke thrombectomies after a good friend and colleague suffered a severe stroke in 2010.
“This made us realize that our hospital needed to be more active in the stroke field,” he said. “We decided that we needed to start doing stroke interventions.”
But the major problem was the lack of neurointerventionalists.
“There are not enough neurointerventionalists in Europe. Interventional cardiologists can perform thousands of procedures every year whereas a neurointerventionalist will at best perform hundreds a year. It is quicker and simpler to train the cardiologist to do it,” Dr. Widimsky said.
They hired one neurointerventionalist to lead the program. “He was our tutor, he taught us his skills,” Dr. Widimsky said. “The cath lab is open 24/7, but if we only have one neurointerventionalist we cannot offer a 24/7 service for stroke thrombectomy. But if we merge with cardiology then we can,” he added.
Their hospital is a very busy center for myocardial infarction, percutaneous coronary intervention, and carotid stenting, he noted. “It is not difficult to make the step from that to stroke thrombectomy. Interventional cardiologists are used to performing carotid and coronary artery stenting. Stroke thrombectomy is a similar technique. The thrombectomy procedure is different from coronary angioplasty but it is not more difficult. Actually, I think coronary angioplasty can be more difficult.”
Dr. Widimsky explained that cardiologists need to learn about the brain anatomy and physiology and learn the stroke imaging techniques. “I spent 1 month in the U.S. learning stroke interventions working with simulators,” he said. “I think interventional cardiologists can learn what they need to know in about 6 months. I would recommend they should watch about 50 procedures and perform at least 25 under supervision.”
He said this model is the way forward and hopes it will become routine. Thrombectomy is “tremendously effective” in improving outcomes in severe strokes, with a number needed to treat (NNT) of just 2.6 to prevent long-term disability in one patient, he said, while other procedures can have NNTs of 50 or more.
“But millions of patients with acute severe stroke are not getting this life-changing treatment,” he added. “We must do everything we can to make this service available to as many patients as possible.”
Dr. Widimsky acknowledges that there has been opposition to this idea from the neurointerventionalist professional bodies but this has lessened recently, at least in Europe. And a program that allows interventionalists with experience in extracranial carotid and vertebral endovascular procedures to “fast-track” technical training has now been proposed.
“There is an enormous unmet need for stroke thrombectomy in Europe, with some countries needing to increase the number of procedures done by 10 or 20 times. These include the U.K., Sweden, Italy, Spain, and Portugal. This cannot be done without cardiology,” Dr. Widimsky said.
Editorial strongly supportive
An accompanying editorial strongly endorses the idea of using interdisciplinary teams to deliver high standard stroke care.
Marius Hornung, MD, and Horst Sievert, MD, from CardioVascular Center Frankfurt (Germany), point out that many experienced cardiologists are trained in performing carotid artery interventions and are therefore experienced in accessing the supra-aortic arteries.
“To be able to guarantee optimized stroke therapy as soon as possible, disputes over competence among the individual medical societies involved must be ended,” they wrote.
They advocate for the creation of interdisciplinary teams, with diagnostics, patient selection, and follow-up care remaining the core competencies and tasks of neurology; in addition, they call for appropriately trained and experienced physicians – regardless of their specialties – performing acute stroke interventions and endovascular thrombectomy.
“Such a network must be installed as soon as possible to fulfill the mantra ‘time is brain’ ... and not losing unnecessary time to patient transfer, or continuing to offer only the second-best therapy,” they concluded.
Opposition in the United States
Dr. Widimsky explained that this proposal may not be so applicable to the United States, where the need for more clinicians capable of performing stroke thrombectomies does not appear to be as critical, possibly because vascular neurosurgeons as well as neuroradiologists are qualified to undertake these procedures.
In an interview, J. Mocco, MD, director of the cerebrovascular center, department of neurological surgery, at Mount Sinai Health System, New York, confirmed that this was the case.
“There is no legitimate data to support the claim that there is a lack of an adequate workforce to provide stroke thrombectomy, at least in the U.S.,” he said, adding that, rather, the primary limitation to patient access is a lack of adequate systems of care. “We should learn from the trauma model, which is strongly evidence based, and provide emergency stroke care in a similarly regionalized manner.”
Dr. Mocco, vice president of the Society of NeuroInterventional Surgery, was not impressed with the current study.
“This paper is a retrospective, single-center, unadjudicated, nonindependent assessor case series and therefore, as the authors acknowledge in the limitations section of their paper, it is invalid to compare these data to the results from high-quality, prospective, core-lab, and independent assessor adjudicated randomized trials,” he said. “The supposition that this trial provides evidence that the reported model should be widely considered lacks scientific rigor.”
Furthermore, “the interventional cardiology literature is replete with data regarding the importance of technical expertise and content knowledge,” he added. “Why would that community now propose that such expertise and knowledge is not necessary for the brain?”
Dr. Mocco argues that the concept that interventional cardiologists should be fast-tracked to perform stroke interventions because they use similar tools, navigate blood vessels, and are comfortable working in critical situations, does not hold up.
“Liver surgeons and brain surgeons are both familiar with tissue manipulation, are used to operating in critical situations, and use cautery, scissors, and scalpels; but no one would argue that a brain surgeon should be fast-tracked to perform liver surgery, or vice versa.”
He added: “Stroke patients do not have the luxury of choosing the physician who provides their thrombectomy. We should do everything reasonable to ensure that our systems of care are organized so that these vulnerable patients are treated by physicians who have appropriate knowledge and expertise.”
This study was supported by the Charles University Research program. The authors and editorialists have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pneumonia risk soars in heart failure patients, especially HFpEF
Patients with heart failure get pneumonia at a rate almost three times greater than expected and, once they do get pneumonia, have about a fourfold greater risk of death, investigators for a retrospective analysis of 13,000 patients from two landmark randomized HF trials have found.
The investigators also found that HF patients with preserved ejection fraction (HFpEF) are at the highest risk of developing pneumonia. The findings underscore the importance of patients with HF getting a pneumonia vaccination, they found.
The analysis showed that 6.3% of patients in the PARADIGM-HF trial and 10.6% of those in the PARAGON-HF trial developed pneumonia, reported the study authors, led by John J.V. McMurray, MD, of the British Heart Foundation Cardiovascular Research Center at the University of Glasgow in Scotland (J Am Coll Cardiol. 2021;77:1961-73).
“The main reason for doing this study was the fact that many heart failure patients are not vaccinated, as they should be, against pneumonia – both pneumococcus and influenza vaccination,” Dr. McMurray said in an interview. “We wanted to document the frequency and consequences of pneumonia in patients with heart failure to help highlight this deficiency in care.”
Dr. McMurray said he believes this is the first study to document the incidence of pneumonia and pneumonia-related outcomes according to the two major ejection fraction phenotypes.
PARADIGM-HF and PARAGON-HF
The post hoc analysis consisted of 8,399 patients with HF with reduced ejection fraction (HFrEF) in PARADIGM-HF (Eur J Heart Fail. 2013 Sep;15[9]:1062-73) and 4,796 patients with HFpEF in PARAGON-HF (N Engl J Med. 2014 Sep 11;371[11]:993-1004). The analysis focused on the 528 and 510 patients in each study, respectively, who developed pneumonia. Those rates translated to an incidence rate of 29 per 1,000 patient-years (95% confidence interval, 27-31) in PARADIGM-HF and 39 per 1,000 patient-years (95% CI, 36-42) in PARAGON-HF.
After pneumonia, the risk of death in patients increased substantially. In PARADIGM-HF, the adjusted hazard ratio for the risk of death from any cause after pneumonia was 4.34 (95% CI, 3.73-5.05). In PARAGON-HF, it was 3.76 (95% CI, 3.09-4.58). HF patients who contracted pneumonia also tended to have HF longer than their counterparts who didn’t develop pneumonia, but the frequency of previous hospitalization for HF didn’t vary between the pneumonia and no-pneumonia groups.
Patients who developed pneumonia tended to be older (average age of 66.9 years vs. 64.6 years, P < .001) and male (83.9% vs. 77.8%, P < .001). The mean age of patients in PARADIGM-HF was almost a decade younger than those in PARAGON-HF, 64 vs. 73 years.
Pneumonia patients also had worse Kansas City Cardiomyopathy Questionnaire scores (76 vs. 80 on average), but no difference in New York Heart Association functional class. “In general, patients who developed pneumonia had more symptoms and signs and HF than those who did not develop pneumonia,” Dr. McMurray and colleagues wrote.
Pneumonia patients also had higher rates of chronic obstructive pulmonary disease (26% vs. 12%), diabetes (43% vs. 34%), and atrial fibrillation (46% vs. 36%).
Another reason for conducting the study, Dr. McMurray said, “was the prior findings in patients with coronary disease and acute myocardial infarction that the risk associated with an episode of pneumonia [e.g., in subsequent vascular events and deaths] persisted long after the acute event. We wanted to see if this was also the case for heart failure, and indeed it was.”
For example, the adjusted HR for cardiovascular death or hospitalization in the first month following an episode of pneumonia was 9.48 (range of 6.85-13.12, P < .001), leveling off to 1.59 after 3 months or more.
Vaccination crucial in HF patients
Dr. McMurray noted that this study emphasizes the importance of pneumonia vaccination for patients with HF. “Given that we have so few treatments to offer patients with HFpEF, this makes the potential value of vaccination in these patients all the greater,” he said.
The COVID-19 pandemic, Dr. McMurray said, is a “good reminder of the dangers of a respiratory infection and the importance of vaccination in these patients. COVID-19 has interesting parallels in being a systemic disease and one with postacute, persisting effects.”
The persistent risk for adverse cardiovascular events 3 months and later after pneumonia is a novel finding of the study, wrote Donna Mancini, MD, and Gregory Gibson, MD, in an invited commentary (J Am Coll Cardiol. 2021;77:1974-6). Both are with the Icahn School of Medicine at Mt. Sinai in New York. The post hoc study also “serves as an important reminder” of pneumonia risk in patients with HF, especially during the pandemic, they wrote.
“Although vaccination alone appears unlikely to be a panacea, it is a readily accessible tool for mitigating disease severity and improving outcomes,” Dr. Mancini and Dr. Gibson wrote. “After all, an ounce of prevention is worth a pound of cure.”
Novartis provided funding for the PARADIGM-HF and PARAGON-HF trials, and Dr. McMurray and coauthors disclosed financial relationships with Novartis. Dr. Mancini and Dr. Gibson have no relevant financial relationships to disclose.
Patients with heart failure get pneumonia at a rate almost three times greater than expected and, once they do get pneumonia, have about a fourfold greater risk of death, investigators for a retrospective analysis of 13,000 patients from two landmark randomized HF trials have found.
The investigators also found that HF patients with preserved ejection fraction (HFpEF) are at the highest risk of developing pneumonia. The findings underscore the importance of patients with HF getting a pneumonia vaccination, they found.
The analysis showed that 6.3% of patients in the PARADIGM-HF trial and 10.6% of those in the PARAGON-HF trial developed pneumonia, reported the study authors, led by John J.V. McMurray, MD, of the British Heart Foundation Cardiovascular Research Center at the University of Glasgow in Scotland (J Am Coll Cardiol. 2021;77:1961-73).
“The main reason for doing this study was the fact that many heart failure patients are not vaccinated, as they should be, against pneumonia – both pneumococcus and influenza vaccination,” Dr. McMurray said in an interview. “We wanted to document the frequency and consequences of pneumonia in patients with heart failure to help highlight this deficiency in care.”
Dr. McMurray said he believes this is the first study to document the incidence of pneumonia and pneumonia-related outcomes according to the two major ejection fraction phenotypes.
PARADIGM-HF and PARAGON-HF
The post hoc analysis consisted of 8,399 patients with HF with reduced ejection fraction (HFrEF) in PARADIGM-HF (Eur J Heart Fail. 2013 Sep;15[9]:1062-73) and 4,796 patients with HFpEF in PARAGON-HF (N Engl J Med. 2014 Sep 11;371[11]:993-1004). The analysis focused on the 528 and 510 patients in each study, respectively, who developed pneumonia. Those rates translated to an incidence rate of 29 per 1,000 patient-years (95% confidence interval, 27-31) in PARADIGM-HF and 39 per 1,000 patient-years (95% CI, 36-42) in PARAGON-HF.
After pneumonia, the risk of death in patients increased substantially. In PARADIGM-HF, the adjusted hazard ratio for the risk of death from any cause after pneumonia was 4.34 (95% CI, 3.73-5.05). In PARAGON-HF, it was 3.76 (95% CI, 3.09-4.58). HF patients who contracted pneumonia also tended to have HF longer than their counterparts who didn’t develop pneumonia, but the frequency of previous hospitalization for HF didn’t vary between the pneumonia and no-pneumonia groups.
Patients who developed pneumonia tended to be older (average age of 66.9 years vs. 64.6 years, P < .001) and male (83.9% vs. 77.8%, P < .001). The mean age of patients in PARADIGM-HF was almost a decade younger than those in PARAGON-HF, 64 vs. 73 years.
Pneumonia patients also had worse Kansas City Cardiomyopathy Questionnaire scores (76 vs. 80 on average), but no difference in New York Heart Association functional class. “In general, patients who developed pneumonia had more symptoms and signs and HF than those who did not develop pneumonia,” Dr. McMurray and colleagues wrote.
Pneumonia patients also had higher rates of chronic obstructive pulmonary disease (26% vs. 12%), diabetes (43% vs. 34%), and atrial fibrillation (46% vs. 36%).
Another reason for conducting the study, Dr. McMurray said, “was the prior findings in patients with coronary disease and acute myocardial infarction that the risk associated with an episode of pneumonia [e.g., in subsequent vascular events and deaths] persisted long after the acute event. We wanted to see if this was also the case for heart failure, and indeed it was.”
For example, the adjusted HR for cardiovascular death or hospitalization in the first month following an episode of pneumonia was 9.48 (range of 6.85-13.12, P < .001), leveling off to 1.59 after 3 months or more.
Vaccination crucial in HF patients
Dr. McMurray noted that this study emphasizes the importance of pneumonia vaccination for patients with HF. “Given that we have so few treatments to offer patients with HFpEF, this makes the potential value of vaccination in these patients all the greater,” he said.
The COVID-19 pandemic, Dr. McMurray said, is a “good reminder of the dangers of a respiratory infection and the importance of vaccination in these patients. COVID-19 has interesting parallels in being a systemic disease and one with postacute, persisting effects.”
The persistent risk for adverse cardiovascular events 3 months and later after pneumonia is a novel finding of the study, wrote Donna Mancini, MD, and Gregory Gibson, MD, in an invited commentary (J Am Coll Cardiol. 2021;77:1974-6). Both are with the Icahn School of Medicine at Mt. Sinai in New York. The post hoc study also “serves as an important reminder” of pneumonia risk in patients with HF, especially during the pandemic, they wrote.
“Although vaccination alone appears unlikely to be a panacea, it is a readily accessible tool for mitigating disease severity and improving outcomes,” Dr. Mancini and Dr. Gibson wrote. “After all, an ounce of prevention is worth a pound of cure.”
Novartis provided funding for the PARADIGM-HF and PARAGON-HF trials, and Dr. McMurray and coauthors disclosed financial relationships with Novartis. Dr. Mancini and Dr. Gibson have no relevant financial relationships to disclose.
Patients with heart failure get pneumonia at a rate almost three times greater than expected and, once they do get pneumonia, have about a fourfold greater risk of death, investigators for a retrospective analysis of 13,000 patients from two landmark randomized HF trials have found.
The investigators also found that HF patients with preserved ejection fraction (HFpEF) are at the highest risk of developing pneumonia. The findings underscore the importance of patients with HF getting a pneumonia vaccination, they found.
The analysis showed that 6.3% of patients in the PARADIGM-HF trial and 10.6% of those in the PARAGON-HF trial developed pneumonia, reported the study authors, led by John J.V. McMurray, MD, of the British Heart Foundation Cardiovascular Research Center at the University of Glasgow in Scotland (J Am Coll Cardiol. 2021;77:1961-73).
“The main reason for doing this study was the fact that many heart failure patients are not vaccinated, as they should be, against pneumonia – both pneumococcus and influenza vaccination,” Dr. McMurray said in an interview. “We wanted to document the frequency and consequences of pneumonia in patients with heart failure to help highlight this deficiency in care.”
Dr. McMurray said he believes this is the first study to document the incidence of pneumonia and pneumonia-related outcomes according to the two major ejection fraction phenotypes.
PARADIGM-HF and PARAGON-HF
The post hoc analysis consisted of 8,399 patients with HF with reduced ejection fraction (HFrEF) in PARADIGM-HF (Eur J Heart Fail. 2013 Sep;15[9]:1062-73) and 4,796 patients with HFpEF in PARAGON-HF (N Engl J Med. 2014 Sep 11;371[11]:993-1004). The analysis focused on the 528 and 510 patients in each study, respectively, who developed pneumonia. Those rates translated to an incidence rate of 29 per 1,000 patient-years (95% confidence interval, 27-31) in PARADIGM-HF and 39 per 1,000 patient-years (95% CI, 36-42) in PARAGON-HF.
After pneumonia, the risk of death in patients increased substantially. In PARADIGM-HF, the adjusted hazard ratio for the risk of death from any cause after pneumonia was 4.34 (95% CI, 3.73-5.05). In PARAGON-HF, it was 3.76 (95% CI, 3.09-4.58). HF patients who contracted pneumonia also tended to have HF longer than their counterparts who didn’t develop pneumonia, but the frequency of previous hospitalization for HF didn’t vary between the pneumonia and no-pneumonia groups.
Patients who developed pneumonia tended to be older (average age of 66.9 years vs. 64.6 years, P < .001) and male (83.9% vs. 77.8%, P < .001). The mean age of patients in PARADIGM-HF was almost a decade younger than those in PARAGON-HF, 64 vs. 73 years.
Pneumonia patients also had worse Kansas City Cardiomyopathy Questionnaire scores (76 vs. 80 on average), but no difference in New York Heart Association functional class. “In general, patients who developed pneumonia had more symptoms and signs and HF than those who did not develop pneumonia,” Dr. McMurray and colleagues wrote.
Pneumonia patients also had higher rates of chronic obstructive pulmonary disease (26% vs. 12%), diabetes (43% vs. 34%), and atrial fibrillation (46% vs. 36%).
Another reason for conducting the study, Dr. McMurray said, “was the prior findings in patients with coronary disease and acute myocardial infarction that the risk associated with an episode of pneumonia [e.g., in subsequent vascular events and deaths] persisted long after the acute event. We wanted to see if this was also the case for heart failure, and indeed it was.”
For example, the adjusted HR for cardiovascular death or hospitalization in the first month following an episode of pneumonia was 9.48 (range of 6.85-13.12, P < .001), leveling off to 1.59 after 3 months or more.
Vaccination crucial in HF patients
Dr. McMurray noted that this study emphasizes the importance of pneumonia vaccination for patients with HF. “Given that we have so few treatments to offer patients with HFpEF, this makes the potential value of vaccination in these patients all the greater,” he said.
The COVID-19 pandemic, Dr. McMurray said, is a “good reminder of the dangers of a respiratory infection and the importance of vaccination in these patients. COVID-19 has interesting parallels in being a systemic disease and one with postacute, persisting effects.”
The persistent risk for adverse cardiovascular events 3 months and later after pneumonia is a novel finding of the study, wrote Donna Mancini, MD, and Gregory Gibson, MD, in an invited commentary (J Am Coll Cardiol. 2021;77:1974-6). Both are with the Icahn School of Medicine at Mt. Sinai in New York. The post hoc study also “serves as an important reminder” of pneumonia risk in patients with HF, especially during the pandemic, they wrote.
“Although vaccination alone appears unlikely to be a panacea, it is a readily accessible tool for mitigating disease severity and improving outcomes,” Dr. Mancini and Dr. Gibson wrote. “After all, an ounce of prevention is worth a pound of cure.”
Novartis provided funding for the PARADIGM-HF and PARAGON-HF trials, and Dr. McMurray and coauthors disclosed financial relationships with Novartis. Dr. Mancini and Dr. Gibson have no relevant financial relationships to disclose.
FROM JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
How often should we check EKGs in patients starting antipsychotic medications?
Determining relative risk with available data
Case
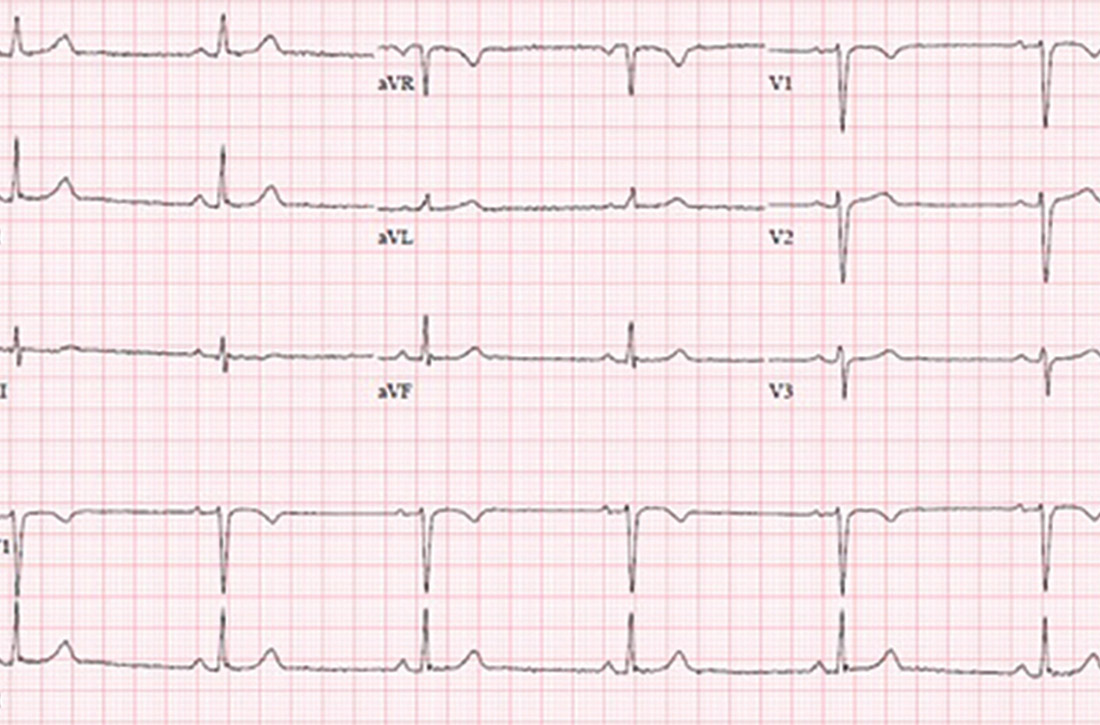
An 88-year-old woman with history of osteoporosis, hyperlipidemia, and a remote myocardial infarction presents to the ED with altered mental status and agitation. The patient is admitted to the medicine service for further management. Her current medications include a thiazide and a statin. Psychiatry is consulted and recommends administering intravenous haloperidol. A baseline EKG shows a corrected QT interval (QTc) of 486 milliseconds (ms). How often should subsequent EKGs be ordered?
Overview of issue
A prolonged QT interval can predispose a patient to dangerous arrhythmias such as Torsades de pointes (TdP), which results in sudden cardiac death in about 10% of cases.1,2 A prolonged QTc interval can be caused by cardiac, renal, or hepatic dysfunction; congenital Long QT Syndrome (LQTS)2; electrolyte abnormalities; or as a result of many drugs, including most antipsychotic medications such as quetiapine, olanzapine, risperidone and haloperidol.
To diminish risk of TdP while taking these medications, it is necessary to monitor the QTc interval. Before commencing a QT-prolonging medication, it is recommended to get a baseline EKG, then perform EKG monitoring after administering the medication.
According to American Heart Association guidelines, a prolonged QT interval is considered more than 460 ms in women or above 450 ms in men.3 If an abnormal rhythm and/or prolonged QTc is detected via EKG monitoring, then the drug dosage can be changed or an alternative therapy selected.4 However, there are no current guidelines recommending how often EKG monitoring should be performed after a QT-prolonging antipsychotic medication is administered on an inpatient medicine unit. Without guidelines, there is potential for health care providers to under- or over-order EKG monitoring, possibly putting patients at risk of TdP or wasting hospital resources, respectively.
Overview of the data
There are currently no universally accepted guidelines regarding inpatient EKG monitoring for patients started on QTc prolonging antipsychotic medications. A 2018 review of the literature surrounding assessment and management of patients on QTc prolonging medications was performed to analyze the available data and make recommendations; notably the evidence was limited as none of the studies were randomized controlled trials.
The authors recommend assessing the drug for QTc prolonging potential, and if possible, choosing alternative treatment in patients with baseline prolonged QTc. If the QT prolonging medication is the best or only option, then the next step is assessing the patient’s risk for QTc prolongation based on that person’s current condition and medical history.5 They recommend using the QTc prolonging risk point system developed by Tisdale and colleagues, which identified patient risk factors for elevated QTc intervals based on EKG findings in cardiac care units at a large tertiary hospital center.6
Based on the patient’s demographics, current condition, and medication list, the score can be used to stratify patients into low-, medium-, and high-risk categories (see Table 1). 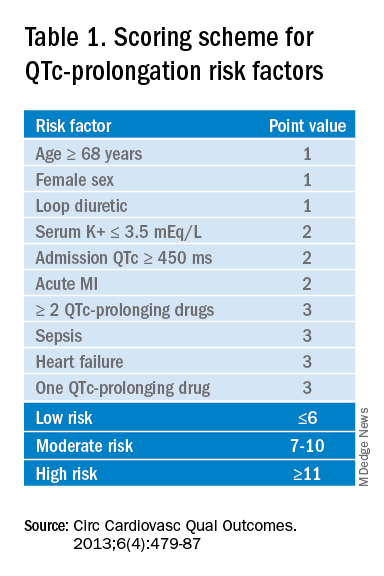
Risk factors include age over 68 years, female sex, prior MI, concurrent use of other QTc prolonging medications, and sepsis, all of which have differing ability to cause QTc prolongation and thus are weighted differentially. This scoring system is helpful in identifying high risk patients; however, the review does not include recommendations for management of these patients beyond removing the offending drug or monitoring EKGs more aggressively in higher risk patients once identified.6
Low-risk patients can be managed expectantly. If the baseline QTc is < 500 ms, then the provider may administer the medication, but should obtain follow-up EKG monitoring to ensure the QTc does not rise above 500 ms; if it does, a management change is necessitated. For moderate- to high-risk patients with a baseline QTc > 500 ms, they recommend not administering the medication and consulting a cardiologist. The review does not provide a recommendation on how often EKG monitoring should be performed after prescribing an antipsychotic medication in an inpatient setting.5
A 2018 review article explored patient risk factors for a prolonged QTc in the setting of prescribing potentially QTc prolonging antipsychotics.7 The authors reiterate that QTc prolonging risk factors are important considerations when prescribing antipsychotics that can lead to adverse events, though they note that much of the literature associating antipsychotics with negative outcomes consists of case reports in which patients had independent risk factors for development of TdP, such as preexisting ventricular arrhythmias.
In addition, the data regarding the risk of each individual antipsychotic agent are not comprehensive. Some medications that have been deemed “QTc prolonging” were identified as such in only a handful of cases where patients had confounding comorbid risk factors. This raises concern that some medications are being unduly stigmatized in situations where there is little chance of TdP. If there is no equivalent or alternate treatment available, this may lead to an antipsychotic medication being held unnecessarily, which may exacerbate the psychiatric illness.
The authors note that the trend toward ordering baseline EKGs in the inpatient setting following administration of a new antipsychotic may be partly attributed to the ready availability of EKG testing in hospitals. They recommend a baseline EKG to assess the patient’s risk. For most agents, they recommend no further EKG monitoring unless there is a change in patient risk factors. Follow-up EKGs should be done in patients with multiple or significant risk factors to assess their QTc stability. In patients with a QTc > 500 ms on a follow-up EKG, daily monitoring is encouraged alongside reassessment of the current treatment regimen.7
Overall, the current literature suggests that providers should know which antipsychotics carry a risk for QTc prolongation and what other treatment options are available. The risk of QTc prolongation for common antipsychotic agents is provided in Table 2.
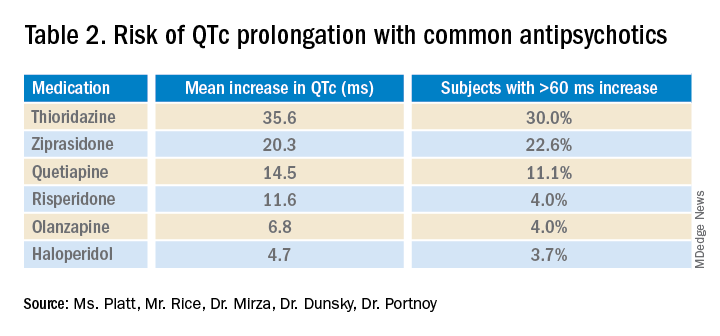
Providers should assess their patients’ risk factors for QTc prolongation and order a baseline EKG to help quantify the cardiac risk associated with prescribing the drug. In patients with many risk factors or complicated medication regimens, a follow-up EKG should be performed to assess the new QTc baseline. If the subsequent QTc is > 500 ms, then an alternative medication should be strongly considered. The majority of patients, however, will not have a clinically significant increase in their QTc, in which case there is no need for a change in medication and monitoring frequency can be deescalated.
Application of data to the case
Our 88-year-old patient has multiple risk factors for a prolonged QTc, and according to the Tisdale scoring system is at moderate risk (7-10 points). Her risk of developing TdP increases with the addition of IV haloperidol to her regimen.
Because of her increased risk, it is reasonable to consider alternative management. If she can cooperate with PO medications, then olanzapine could be given, which has a lesser effect on the QTc interval. If unable to take oral medications, she could be given haloperidol intramuscularly, which causes less QTc prolongation than the IV formulation. If an antipsychotic is administered, she should receive EKG monitoring.
Given the lack of evidence on the optimal monitoring strategy, a protocol should be utilized that balances the ability to capture a clinically meaningful increase in the QTc with appropriate stewardship of resources. Our practice is to initially monitor the EKG every 3 days in moderate- to high-risk patients with baseline QTc < 500 ms. If the QTc remains below 500 ms over three EKGs, then treatment may continue with EKG monitoring weekly while the patient is hospitalized. If the QTc rises above 500 ms, then a management change would be indicated (either dose reduction or a change of agents). If antipsychotic medications are continued, we check the EKG daily while the QTc is >500 ms until there are three unchanged EKGS, and then consider deescalating monitoring to every 3 days.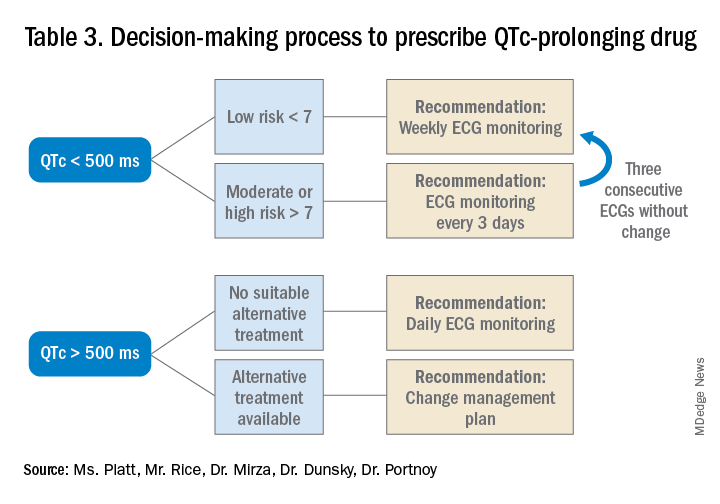
Bottom line
Prior to prescribing, perform a baseline EKG and assess the patient’s risk of QTc prolongation. If the patient is at increased risk, avoid prescribing QTc prolonging medications where alternatives exist. If a QTc prolonging medication is used in a patient with a moderate- to high-risk score, check an EKG every 3 days or daily if the QTc increases to > 500 ms.
Ms. Platt is a medical student at the Icahn School of Medicine at Mount Sinai in New York. Mr. Rice is a medical student at the Icahn School of Medicine. Dr. Mirza is assistant clinical professor of psychiatry at the Icahn School of Medicine. Dr. Dunsky is a cardiologist and assistant professor at the Icahn School of Medicine. Dr. Portnoy is a hospitalist and assistant professor at the Icahn School of Medicine.
References
1. Darpö B. Spectrum of drugs prolonging QT interval and the incidence of torsades de pointes. Eur Heart J Supplements. 2001;3(suppl_K):K70-K80. doi: 10.1016/S1520-765X(01)90009-4.
2. Schwartz PJ, Woosley RL. Predicting the unpredictable: Drug-induced QT prolongation and torsades de pointes. J Am Coll Cardiol. 2016;67(13):1639-50. doi: 10.1016/j.jacc.2015.12.063.
3. Rautaharju PM et al. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram. Part IV: The ST Segment, T and U Waves, and the QT Interval A Scientific Statement From the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol. 2009 Mar 17;53(11):982-91. doi: 10.1016/j.jacc.2008.12.014.
4. Drew BJ et al. Prevention of torsades de pointes in hospital settings: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047-60. doi: 10.1161/CIRCULATIONAHA.109.192704.
5. Zolezzi M, Cheung L. A literature-based algorithm for the assessment, management, and monitoring of drug-induced QTc prolongation in the psychiatric population. Neuropsychiatr Dis Treat. 2019;15:105-14. doi: 10.2147/NDT.S186474.
6. Tisdale JE et al. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013;6(4):479-87. doi: 10.1161/CIRCOUTCOMES.113.000152.
7. Beach SR et al. QT prolongation, torsades de pointes, and psychotropic medications: A 5-year update. Psychosomatics. 2018;59(2):105-22. doi: 10.1016/j.psym.2017.10.009.
Key points
- An increased QTc interval can lead to TdP, ventricular fibrillation and cardiac death.
- The relative risk of each antipsychotic medication should be determined based on available data and the Tisdale scoring system can provide a system to assess a patient’s risk of QTc prolongation.
- Low-risk patients with a baseline QTc <500 ms should receive a baseline EKG and inpatient EKG monitoring weekly while moderate- to high-risk patients should receive EKG monitoring every 3 days.
- A QTc > 500 ms suggests the need for a management change (drug discontinuation, dose reduction, or a switch to another agent). If the antipsychotic is absolutely necessary, perform daily EKG monitoring until there are three unchanged EKGs, and then consider deescalating monitoring to every 3 days.
Additional reading
Beach SR et al. QT Prolongation, torsades de pointes, and psychotropic medications: A 5-year update. Psychosomatics. 2018;59(2):105-22. doi: 10.1016/j.psym.2017.10.009.
Drew BJ et al. Prevention of torsades de pointes in hospital settings: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047-60. doi: 10.1161/CIRCULATIONAHA.109.192704.
Zolezzi M, Cheung L. A literature-based algorithm for the assessment, management, and monitoring of drug-induced QTc prolongation in the psychiatric population. Neuropsychiatr Dis Treat. 2019;15:105-14. doi: 10.2147/NDT.S186474.
Quiz
A 70-year-old male inpatient on furosemide with last known potassium level of 3.3 is going to be started on olanzapine. His baseline EKG has a QTc of 470 ms.
How often should he receive EKG monitoring?
A. Daily
B. Every 3 days
C. Weekly
D. Monthly
Answer (C): He is a low risk patient (6 points: over 70 yrs, loop diuretic, K+< 3.5, QTc > 450 ms), so he should receive weekly EKG monitoring.
Determining relative risk with available data
Determining relative risk with available data
Case
An 88-year-old woman with history of osteoporosis, hyperlipidemia, and a remote myocardial infarction presents to the ED with altered mental status and agitation. The patient is admitted to the medicine service for further management. Her current medications include a thiazide and a statin. Psychiatry is consulted and recommends administering intravenous haloperidol. A baseline EKG shows a corrected QT interval (QTc) of 486 milliseconds (ms). How often should subsequent EKGs be ordered?
Overview of issue
A prolonged QT interval can predispose a patient to dangerous arrhythmias such as Torsades de pointes (TdP), which results in sudden cardiac death in about 10% of cases.1,2 A prolonged QTc interval can be caused by cardiac, renal, or hepatic dysfunction; congenital Long QT Syndrome (LQTS)2; electrolyte abnormalities; or as a result of many drugs, including most antipsychotic medications such as quetiapine, olanzapine, risperidone and haloperidol.
To diminish risk of TdP while taking these medications, it is necessary to monitor the QTc interval. Before commencing a QT-prolonging medication, it is recommended to get a baseline EKG, then perform EKG monitoring after administering the medication.
According to American Heart Association guidelines, a prolonged QT interval is considered more than 460 ms in women or above 450 ms in men.3 If an abnormal rhythm and/or prolonged QTc is detected via EKG monitoring, then the drug dosage can be changed or an alternative therapy selected.4 However, there are no current guidelines recommending how often EKG monitoring should be performed after a QT-prolonging antipsychotic medication is administered on an inpatient medicine unit. Without guidelines, there is potential for health care providers to under- or over-order EKG monitoring, possibly putting patients at risk of TdP or wasting hospital resources, respectively.
Overview of the data
There are currently no universally accepted guidelines regarding inpatient EKG monitoring for patients started on QTc prolonging antipsychotic medications. A 2018 review of the literature surrounding assessment and management of patients on QTc prolonging medications was performed to analyze the available data and make recommendations; notably the evidence was limited as none of the studies were randomized controlled trials.
The authors recommend assessing the drug for QTc prolonging potential, and if possible, choosing alternative treatment in patients with baseline prolonged QTc. If the QT prolonging medication is the best or only option, then the next step is assessing the patient’s risk for QTc prolongation based on that person’s current condition and medical history.5 They recommend using the QTc prolonging risk point system developed by Tisdale and colleagues, which identified patient risk factors for elevated QTc intervals based on EKG findings in cardiac care units at a large tertiary hospital center.6
Based on the patient’s demographics, current condition, and medication list, the score can be used to stratify patients into low-, medium-, and high-risk categories (see Table 1). 
Risk factors include age over 68 years, female sex, prior MI, concurrent use of other QTc prolonging medications, and sepsis, all of which have differing ability to cause QTc prolongation and thus are weighted differentially. This scoring system is helpful in identifying high risk patients; however, the review does not include recommendations for management of these patients beyond removing the offending drug or monitoring EKGs more aggressively in higher risk patients once identified.6
Low-risk patients can be managed expectantly. If the baseline QTc is < 500 ms, then the provider may administer the medication, but should obtain follow-up EKG monitoring to ensure the QTc does not rise above 500 ms; if it does, a management change is necessitated. For moderate- to high-risk patients with a baseline QTc > 500 ms, they recommend not administering the medication and consulting a cardiologist. The review does not provide a recommendation on how often EKG monitoring should be performed after prescribing an antipsychotic medication in an inpatient setting.5
A 2018 review article explored patient risk factors for a prolonged QTc in the setting of prescribing potentially QTc prolonging antipsychotics.7 The authors reiterate that QTc prolonging risk factors are important considerations when prescribing antipsychotics that can lead to adverse events, though they note that much of the literature associating antipsychotics with negative outcomes consists of case reports in which patients had independent risk factors for development of TdP, such as preexisting ventricular arrhythmias.
In addition, the data regarding the risk of each individual antipsychotic agent are not comprehensive. Some medications that have been deemed “QTc prolonging” were identified as such in only a handful of cases where patients had confounding comorbid risk factors. This raises concern that some medications are being unduly stigmatized in situations where there is little chance of TdP. If there is no equivalent or alternate treatment available, this may lead to an antipsychotic medication being held unnecessarily, which may exacerbate the psychiatric illness.
The authors note that the trend toward ordering baseline EKGs in the inpatient setting following administration of a new antipsychotic may be partly attributed to the ready availability of EKG testing in hospitals. They recommend a baseline EKG to assess the patient’s risk. For most agents, they recommend no further EKG monitoring unless there is a change in patient risk factors. Follow-up EKGs should be done in patients with multiple or significant risk factors to assess their QTc stability. In patients with a QTc > 500 ms on a follow-up EKG, daily monitoring is encouraged alongside reassessment of the current treatment regimen.7
Overall, the current literature suggests that providers should know which antipsychotics carry a risk for QTc prolongation and what other treatment options are available. The risk of QTc prolongation for common antipsychotic agents is provided in Table 2.

Providers should assess their patients’ risk factors for QTc prolongation and order a baseline EKG to help quantify the cardiac risk associated with prescribing the drug. In patients with many risk factors or complicated medication regimens, a follow-up EKG should be performed to assess the new QTc baseline. If the subsequent QTc is > 500 ms, then an alternative medication should be strongly considered. The majority of patients, however, will not have a clinically significant increase in their QTc, in which case there is no need for a change in medication and monitoring frequency can be deescalated.
Application of data to the case
Our 88-year-old patient has multiple risk factors for a prolonged QTc, and according to the Tisdale scoring system is at moderate risk (7-10 points). Her risk of developing TdP increases with the addition of IV haloperidol to her regimen.
Because of her increased risk, it is reasonable to consider alternative management. If she can cooperate with PO medications, then olanzapine could be given, which has a lesser effect on the QTc interval. If unable to take oral medications, she could be given haloperidol intramuscularly, which causes less QTc prolongation than the IV formulation. If an antipsychotic is administered, she should receive EKG monitoring.
Given the lack of evidence on the optimal monitoring strategy, a protocol should be utilized that balances the ability to capture a clinically meaningful increase in the QTc with appropriate stewardship of resources. Our practice is to initially monitor the EKG every 3 days in moderate- to high-risk patients with baseline QTc < 500 ms. If the QTc remains below 500 ms over three EKGs, then treatment may continue with EKG monitoring weekly while the patient is hospitalized. If the QTc rises above 500 ms, then a management change would be indicated (either dose reduction or a change of agents). If antipsychotic medications are continued, we check the EKG daily while the QTc is >500 ms until there are three unchanged EKGS, and then consider deescalating monitoring to every 3 days.
Bottom line
Prior to prescribing, perform a baseline EKG and assess the patient’s risk of QTc prolongation. If the patient is at increased risk, avoid prescribing QTc prolonging medications where alternatives exist. If a QTc prolonging medication is used in a patient with a moderate- to high-risk score, check an EKG every 3 days or daily if the QTc increases to > 500 ms.
Ms. Platt is a medical student at the Icahn School of Medicine at Mount Sinai in New York. Mr. Rice is a medical student at the Icahn School of Medicine. Dr. Mirza is assistant clinical professor of psychiatry at the Icahn School of Medicine. Dr. Dunsky is a cardiologist and assistant professor at the Icahn School of Medicine. Dr. Portnoy is a hospitalist and assistant professor at the Icahn School of Medicine.
References
1. Darpö B. Spectrum of drugs prolonging QT interval and the incidence of torsades de pointes. Eur Heart J Supplements. 2001;3(suppl_K):K70-K80. doi: 10.1016/S1520-765X(01)90009-4.
2. Schwartz PJ, Woosley RL. Predicting the unpredictable: Drug-induced QT prolongation and torsades de pointes. J Am Coll Cardiol. 2016;67(13):1639-50. doi: 10.1016/j.jacc.2015.12.063.
3. Rautaharju PM et al. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram. Part IV: The ST Segment, T and U Waves, and the QT Interval A Scientific Statement From the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol. 2009 Mar 17;53(11):982-91. doi: 10.1016/j.jacc.2008.12.014.
4. Drew BJ et al. Prevention of torsades de pointes in hospital settings: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047-60. doi: 10.1161/CIRCULATIONAHA.109.192704.
5. Zolezzi M, Cheung L. A literature-based algorithm for the assessment, management, and monitoring of drug-induced QTc prolongation in the psychiatric population. Neuropsychiatr Dis Treat. 2019;15:105-14. doi: 10.2147/NDT.S186474.
6. Tisdale JE et al. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013;6(4):479-87. doi: 10.1161/CIRCOUTCOMES.113.000152.
7. Beach SR et al. QT prolongation, torsades de pointes, and psychotropic medications: A 5-year update. Psychosomatics. 2018;59(2):105-22. doi: 10.1016/j.psym.2017.10.009.
Key points
- An increased QTc interval can lead to TdP, ventricular fibrillation and cardiac death.
- The relative risk of each antipsychotic medication should be determined based on available data and the Tisdale scoring system can provide a system to assess a patient’s risk of QTc prolongation.
- Low-risk patients with a baseline QTc <500 ms should receive a baseline EKG and inpatient EKG monitoring weekly while moderate- to high-risk patients should receive EKG monitoring every 3 days.
- A QTc > 500 ms suggests the need for a management change (drug discontinuation, dose reduction, or a switch to another agent). If the antipsychotic is absolutely necessary, perform daily EKG monitoring until there are three unchanged EKGs, and then consider deescalating monitoring to every 3 days.
Additional reading
Beach SR et al. QT Prolongation, torsades de pointes, and psychotropic medications: A 5-year update. Psychosomatics. 2018;59(2):105-22. doi: 10.1016/j.psym.2017.10.009.
Drew BJ et al. Prevention of torsades de pointes in hospital settings: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047-60. doi: 10.1161/CIRCULATIONAHA.109.192704.
Zolezzi M, Cheung L. A literature-based algorithm for the assessment, management, and monitoring of drug-induced QTc prolongation in the psychiatric population. Neuropsychiatr Dis Treat. 2019;15:105-14. doi: 10.2147/NDT.S186474.
Quiz
A 70-year-old male inpatient on furosemide with last known potassium level of 3.3 is going to be started on olanzapine. His baseline EKG has a QTc of 470 ms.
How often should he receive EKG monitoring?
A. Daily
B. Every 3 days
C. Weekly
D. Monthly
Answer (C): He is a low risk patient (6 points: over 70 yrs, loop diuretic, K+< 3.5, QTc > 450 ms), so he should receive weekly EKG monitoring.
Case
An 88-year-old woman with history of osteoporosis, hyperlipidemia, and a remote myocardial infarction presents to the ED with altered mental status and agitation. The patient is admitted to the medicine service for further management. Her current medications include a thiazide and a statin. Psychiatry is consulted and recommends administering intravenous haloperidol. A baseline EKG shows a corrected QT interval (QTc) of 486 milliseconds (ms). How often should subsequent EKGs be ordered?
Overview of issue
A prolonged QT interval can predispose a patient to dangerous arrhythmias such as Torsades de pointes (TdP), which results in sudden cardiac death in about 10% of cases.1,2 A prolonged QTc interval can be caused by cardiac, renal, or hepatic dysfunction; congenital Long QT Syndrome (LQTS)2; electrolyte abnormalities; or as a result of many drugs, including most antipsychotic medications such as quetiapine, olanzapine, risperidone and haloperidol.
To diminish risk of TdP while taking these medications, it is necessary to monitor the QTc interval. Before commencing a QT-prolonging medication, it is recommended to get a baseline EKG, then perform EKG monitoring after administering the medication.
According to American Heart Association guidelines, a prolonged QT interval is considered more than 460 ms in women or above 450 ms in men.3 If an abnormal rhythm and/or prolonged QTc is detected via EKG monitoring, then the drug dosage can be changed or an alternative therapy selected.4 However, there are no current guidelines recommending how often EKG monitoring should be performed after a QT-prolonging antipsychotic medication is administered on an inpatient medicine unit. Without guidelines, there is potential for health care providers to under- or over-order EKG monitoring, possibly putting patients at risk of TdP or wasting hospital resources, respectively.
Overview of the data
There are currently no universally accepted guidelines regarding inpatient EKG monitoring for patients started on QTc prolonging antipsychotic medications. A 2018 review of the literature surrounding assessment and management of patients on QTc prolonging medications was performed to analyze the available data and make recommendations; notably the evidence was limited as none of the studies were randomized controlled trials.
The authors recommend assessing the drug for QTc prolonging potential, and if possible, choosing alternative treatment in patients with baseline prolonged QTc. If the QT prolonging medication is the best or only option, then the next step is assessing the patient’s risk for QTc prolongation based on that person’s current condition and medical history.5 They recommend using the QTc prolonging risk point system developed by Tisdale and colleagues, which identified patient risk factors for elevated QTc intervals based on EKG findings in cardiac care units at a large tertiary hospital center.6
Based on the patient’s demographics, current condition, and medication list, the score can be used to stratify patients into low-, medium-, and high-risk categories (see Table 1). 
Risk factors include age over 68 years, female sex, prior MI, concurrent use of other QTc prolonging medications, and sepsis, all of which have differing ability to cause QTc prolongation and thus are weighted differentially. This scoring system is helpful in identifying high risk patients; however, the review does not include recommendations for management of these patients beyond removing the offending drug or monitoring EKGs more aggressively in higher risk patients once identified.6
Low-risk patients can be managed expectantly. If the baseline QTc is < 500 ms, then the provider may administer the medication, but should obtain follow-up EKG monitoring to ensure the QTc does not rise above 500 ms; if it does, a management change is necessitated. For moderate- to high-risk patients with a baseline QTc > 500 ms, they recommend not administering the medication and consulting a cardiologist. The review does not provide a recommendation on how often EKG monitoring should be performed after prescribing an antipsychotic medication in an inpatient setting.5
A 2018 review article explored patient risk factors for a prolonged QTc in the setting of prescribing potentially QTc prolonging antipsychotics.7 The authors reiterate that QTc prolonging risk factors are important considerations when prescribing antipsychotics that can lead to adverse events, though they note that much of the literature associating antipsychotics with negative outcomes consists of case reports in which patients had independent risk factors for development of TdP, such as preexisting ventricular arrhythmias.
In addition, the data regarding the risk of each individual antipsychotic agent are not comprehensive. Some medications that have been deemed “QTc prolonging” were identified as such in only a handful of cases where patients had confounding comorbid risk factors. This raises concern that some medications are being unduly stigmatized in situations where there is little chance of TdP. If there is no equivalent or alternate treatment available, this may lead to an antipsychotic medication being held unnecessarily, which may exacerbate the psychiatric illness.
The authors note that the trend toward ordering baseline EKGs in the inpatient setting following administration of a new antipsychotic may be partly attributed to the ready availability of EKG testing in hospitals. They recommend a baseline EKG to assess the patient’s risk. For most agents, they recommend no further EKG monitoring unless there is a change in patient risk factors. Follow-up EKGs should be done in patients with multiple or significant risk factors to assess their QTc stability. In patients with a QTc > 500 ms on a follow-up EKG, daily monitoring is encouraged alongside reassessment of the current treatment regimen.7
Overall, the current literature suggests that providers should know which antipsychotics carry a risk for QTc prolongation and what other treatment options are available. The risk of QTc prolongation for common antipsychotic agents is provided in Table 2.

Providers should assess their patients’ risk factors for QTc prolongation and order a baseline EKG to help quantify the cardiac risk associated with prescribing the drug. In patients with many risk factors or complicated medication regimens, a follow-up EKG should be performed to assess the new QTc baseline. If the subsequent QTc is > 500 ms, then an alternative medication should be strongly considered. The majority of patients, however, will not have a clinically significant increase in their QTc, in which case there is no need for a change in medication and monitoring frequency can be deescalated.
Application of data to the case
Our 88-year-old patient has multiple risk factors for a prolonged QTc, and according to the Tisdale scoring system is at moderate risk (7-10 points). Her risk of developing TdP increases with the addition of IV haloperidol to her regimen.
Because of her increased risk, it is reasonable to consider alternative management. If she can cooperate with PO medications, then olanzapine could be given, which has a lesser effect on the QTc interval. If unable to take oral medications, she could be given haloperidol intramuscularly, which causes less QTc prolongation than the IV formulation. If an antipsychotic is administered, she should receive EKG monitoring.
Given the lack of evidence on the optimal monitoring strategy, a protocol should be utilized that balances the ability to capture a clinically meaningful increase in the QTc with appropriate stewardship of resources. Our practice is to initially monitor the EKG every 3 days in moderate- to high-risk patients with baseline QTc < 500 ms. If the QTc remains below 500 ms over three EKGs, then treatment may continue with EKG monitoring weekly while the patient is hospitalized. If the QTc rises above 500 ms, then a management change would be indicated (either dose reduction or a change of agents). If antipsychotic medications are continued, we check the EKG daily while the QTc is >500 ms until there are three unchanged EKGS, and then consider deescalating monitoring to every 3 days.
Bottom line
Prior to prescribing, perform a baseline EKG and assess the patient’s risk of QTc prolongation. If the patient is at increased risk, avoid prescribing QTc prolonging medications where alternatives exist. If a QTc prolonging medication is used in a patient with a moderate- to high-risk score, check an EKG every 3 days or daily if the QTc increases to > 500 ms.
Ms. Platt is a medical student at the Icahn School of Medicine at Mount Sinai in New York. Mr. Rice is a medical student at the Icahn School of Medicine. Dr. Mirza is assistant clinical professor of psychiatry at the Icahn School of Medicine. Dr. Dunsky is a cardiologist and assistant professor at the Icahn School of Medicine. Dr. Portnoy is a hospitalist and assistant professor at the Icahn School of Medicine.
References
1. Darpö B. Spectrum of drugs prolonging QT interval and the incidence of torsades de pointes. Eur Heart J Supplements. 2001;3(suppl_K):K70-K80. doi: 10.1016/S1520-765X(01)90009-4.
2. Schwartz PJ, Woosley RL. Predicting the unpredictable: Drug-induced QT prolongation and torsades de pointes. J Am Coll Cardiol. 2016;67(13):1639-50. doi: 10.1016/j.jacc.2015.12.063.
3. Rautaharju PM et al. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram. Part IV: The ST Segment, T and U Waves, and the QT Interval A Scientific Statement From the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol. 2009 Mar 17;53(11):982-91. doi: 10.1016/j.jacc.2008.12.014.
4. Drew BJ et al. Prevention of torsades de pointes in hospital settings: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047-60. doi: 10.1161/CIRCULATIONAHA.109.192704.
5. Zolezzi M, Cheung L. A literature-based algorithm for the assessment, management, and monitoring of drug-induced QTc prolongation in the psychiatric population. Neuropsychiatr Dis Treat. 2019;15:105-14. doi: 10.2147/NDT.S186474.
6. Tisdale JE et al. Development and validation of a risk score to predict QT interval prolongation in hospitalized patients. Circ Cardiovasc Qual Outcomes. 2013;6(4):479-87. doi: 10.1161/CIRCOUTCOMES.113.000152.
7. Beach SR et al. QT prolongation, torsades de pointes, and psychotropic medications: A 5-year update. Psychosomatics. 2018;59(2):105-22. doi: 10.1016/j.psym.2017.10.009.
Key points
- An increased QTc interval can lead to TdP, ventricular fibrillation and cardiac death.
- The relative risk of each antipsychotic medication should be determined based on available data and the Tisdale scoring system can provide a system to assess a patient’s risk of QTc prolongation.
- Low-risk patients with a baseline QTc <500 ms should receive a baseline EKG and inpatient EKG monitoring weekly while moderate- to high-risk patients should receive EKG monitoring every 3 days.
- A QTc > 500 ms suggests the need for a management change (drug discontinuation, dose reduction, or a switch to another agent). If the antipsychotic is absolutely necessary, perform daily EKG monitoring until there are three unchanged EKGs, and then consider deescalating monitoring to every 3 days.
Additional reading
Beach SR et al. QT Prolongation, torsades de pointes, and psychotropic medications: A 5-year update. Psychosomatics. 2018;59(2):105-22. doi: 10.1016/j.psym.2017.10.009.
Drew BJ et al. Prevention of torsades de pointes in hospital settings: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation. 2010;121(8):1047-60. doi: 10.1161/CIRCULATIONAHA.109.192704.
Zolezzi M, Cheung L. A literature-based algorithm for the assessment, management, and monitoring of drug-induced QTc prolongation in the psychiatric population. Neuropsychiatr Dis Treat. 2019;15:105-14. doi: 10.2147/NDT.S186474.
Quiz
A 70-year-old male inpatient on furosemide with last known potassium level of 3.3 is going to be started on olanzapine. His baseline EKG has a QTc of 470 ms.
How often should he receive EKG monitoring?
A. Daily
B. Every 3 days
C. Weekly
D. Monthly
Answer (C): He is a low risk patient (6 points: over 70 yrs, loop diuretic, K+< 3.5, QTc > 450 ms), so he should receive weekly EKG monitoring.
How some COVID-19 vaccines could cause rare blood clots
on April 14, 2021, after the CDC and Food and Drug Administration recommended that states hold off on using it pending a detailed review of six cases of the same kind of rare but serious event – a blood clot in the vessels that drain blood from the brain combined with a large drop in platelets, which increases the risk for bleeding.
This combination can lead to severe strokes that can lead to brain damage or death. Among the six cases reported, which came to light over the past 3 weeks, one person died, according to the CDC. All six were women and ranged in age from 18 to 48 years.
According to a report from the Vaccine Adverse Event Reporting System (VAERS), which is maintained by the Department of Health & Human Services, the woman who died was 45. She developed a gradually worsening headache about a week after receiving the Johnson & Johnson vaccine.
On March 17, the day she came to the hospital, she was dry heaving. Her headache had suddenly gotten much worse, and the left side of her body was weak, which are signs of a stroke. A CT scan revealed both bleeding in her brain and a clot in her cortical vein. She died the following day.
In addition to VAERS, which accepts reports from anyone, the CDC and FDA are monitoring at least eight other safety systems maintained by hospitals, research centers, long-term care facilities, and insurance companies for signs of trouble with the vaccines. VAERS data is searchable and open to the public. Most of these systems are not publicly available to protect patient privacy. It’s unclear which systems detected the six cases cited by federal regulators.
“These are very serious and potentially fatal problems occurring in a healthy young adult. It’s serious and we need to get to the bottom of it,” said Ed Belongia, MD, director of the Center for Clinical Epidemiology and Population Health at the Marshfield (Wis.) Clinic Research Institute. Dr. Belongia leads a research team that helps the CDC monitor vaccine safety and effectiveness.
“Safety is always the highest priority, and I think what we’ve seen here in the past 24 hours is our vaccine safety monitoring system is working,” he said.
Others agree. “I think what CDC and FDA have detected is a rare, but likely real adverse event associated with this vaccine,” said Paul Offit, MD, director of vaccine education at Children’s Hospital of Philadelphia.
Although much is still unknown about these events, they follow a similar pattern of blood clots reported with the AstraZeneca vaccine in Europe. That vaccine is now sold under the brand name Vaxzevria.
This has experts questioning whether all vaccines of this type may cause these rare clots.
“I think it’s likely a class effect,” said Dr. Offit, who was a member of the FDA advisory committee that reviewed clinical trial data on the J&J vaccine before it was authorized for use.
Adenovirus vaccines scrutinized
Both the Johnson & Johnson and Vaxzevria vaccines use an adenovirus to ferry genetic instructions for making the coronaviruses spike protein into our cells.
Adenoviruses are common, relatively simple viruses that normally cause mild cold or flu symptoms. The ones used in the vaccine are disabled so they can’t make us sick. They’re more like Trojan horses.
Once inside our cells, they release the DNA instructions they carry to make the spike protein of the new coronavirus. Those cells then crank out copies of the spike protein, which then get displayed on the outer surface of the cell membrane where they are recognized by the immune system.
The immune system then makes antibodies and other defenses against the spike so that, when the real coronavirus comes along, our bodies are ready to fight the infection.
There’s no question the vaccine works. In clinical trials, the Johnson & Johnson vaccine was 66% percent effective at preventing against moderate to severe COVID-19 infection, and none of the patients who got COVID-19 after vaccination had to be admitted to the hospital or died.
The idea behind using adenoviruses in vaccines isn’t a new one. In a kind of fight-fire-with-fire approach, the idea is to use a virus, which is good at infecting us, to fight a different kind of virus.
Researchers have been working on the concept for about 10 years, but the COVID-19 vaccines that use this technology are some of the first adenovirus-vector vaccines deployed in humans.
Only one other adenovirus vaccine, for Ebola, has been approved for use in humans. It was approved in Europe last year. Before the Johnson & Johnson vaccine, no other adenovirus vector has been available for use in humans in the United States.
There are six adenovirus-vector vaccines for COVID-19. In addition to AstraZeneca and Johnson & Johnson, there’s the Russian-developed vaccine Sputnik V, along with CanSino from China, and the Covishield vaccine in India.
Adenovirus vaccines are more stable than the mRNA vaccines. That makes them easier to store and transport.
But they have a significant downside, too. Because adenoviruses infect humans out in the world, we already make antibodies against them. So there’s always a danger that our immune systems might recognize and react to the vaccine, rendering it ineffective. For that reason, scientists try to carefully select the adenovirus vectors, or carriers, they use.
The two vaccines under investigation for blood clots are slightly different. The Johnson & Johnson vaccine uses the vector AD26, because most of the population lacks preexisting immunity to it. Vaxzevria uses an adenovirus that infects chimpanzees, called ChAdOx1.
Vaxzevria has been widely used in Europe but has not yet been authorized in the United States.
On April 7, the European Medicines Agency, Europe’s counterpart to the FDA, ruled that unusual blood clots with low blood platelets should be listed as rare side effects on the Vaxzevria vaccine.
The decision came after reviewing 62 cases of cerebral venous sinus thrombosis (CVST) linked to the vaccine and 25 cases of another rare type of clot, called a splanchnic vein thrombosis. Splanchnic veins drain blood from the major organs in the digestive system, including the stomach, liver, and intestines; 18 of those events were fatal.
The reports were culled from reporting in Europe and the United Kingdom, where around 25 million people have received the Vaxzevria vaccine, making these clots exceptionally rare, but serious.
So far, six cases of CVST have been reported in the United States, after more than 7 million doses of the Johnson & Johnson vaccines have been administered.
A key question for U.S. regulators will be the background rate for these types of rare combinations of clots and deplenished platelets. The background rate is the number of events that would be expected to occur naturally in a population of unvaccinated people. On a press call on April 13, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, was asked about the frequency of this dangerous combination. He said the combination of low platelets and clots was so rare that it was hard to pinpoint, but might be somewhere between 2 and 14 cases per million people over the course of a year.
The first Johnson & Johnson doses were given in early March. That means the six cases came to light within the first few weeks of use of the vaccine in the United States, a very short amount of time.
“These were six cases per million people for 2 weeks, which is the same thing as 25 million per year, so it’s clearly above the background rate,” Dr. Offit said.
Studies suggest possible mechanism
On April 9, the New England Journal of Medicine published a detailed evaluation of the 11 patients in Germany and Austria who developed the rare clots after their Vaxzevria vaccines.
The study detected rare antibodies to a signaling protein called platelet factor 4, which helps to coordinate clot formation.
These same type of antibodies form in some people given the blood thinning drug heparin. In those reactions, which are also exceptionally rare, the same type of syndrome develops, leading to large, devastating clots that consume circulating platelets.
It’s not yet clear whether people who develop reactions to the vaccines already have some platelet factor 4 antibodies before they are vaccinated, or whether the vaccines somehow spur the body to make these antibodies, which then launch a kind of autoimmune attack.
The researchers on the paper gave the syndrome a name, vaccine-induced thrombotic thrombocytopenia (VITT).
It’s also not clear why more cases seem to be in women than in men. Andrew Eisenberger, MD, an associate professor of hematology and oncology at Columbia University, New York, said the most common causes of cerebral venous sinus thrombosis have to do with conditions that raise estrogen levels, like pregnancy and hormonal contraception.
“Estrogen naturally leads to changes in several clotting proteins in the blood that may predispose to abnormal blood clotting in a few different sites in the body,” he said. “The clotting changes we are encountering with some of COVID-19 vaccines are likely to be synergistic with the effects of estrogen on the blood.”
No matter the cause, the CDC on April 13 alerted doctors to keep a high index of suspicion for VITT in patients who have received the Johnson & Johnson vaccination within the last 2 weeks. In those patients, the usual course of treatment with blood thinning drugs like heparin may be harmful.
Symptoms to watch for include severe headache or backache, new neurologic symptoms, severe abdominal pain, shortness of breath, leg swelling, tiny red spots on the skin, or easy bruising.
Grappling with evidence
The CDC’s Advisory Committee on Immunization Practices will meet today in an emergency session to review the cases and see if any changes are needed to use of the J&J vaccine in the United States.
Last week, for example, the United Kingdom restricted the use of the AstraZeneca vaccine in people aged younger than 30 years, saying the risks and benefits of vaccination are “more finely balanced” for this age group.
With cases of COVID-19 rising again in the United States, and the Johnson & Johnson vaccine currently the most convenient form of protection against the virus, the committee will have to weigh the risks of that infection against the risk of rare clots caused by vaccination.
They will also likely have to rule out whether any of the cases had COVID. At least one study has reported CVST clots in three patients with confirmed COVID infections. In Europe, COVID infection did not seem to play a role in the formation of the clots with low platelets.
Hilda Bastian, PhD, a clinical trials expert who cofounded the Cochrane Collaboration, said it won’t be an easy task. Much will depend on how certain the committee members feel they know about all the events linked to the vaccine.
“That’s the really, really hard issue from my point of view for them right this moment. Have we missed any? Or how many are we likely to have missed?” asked Dr. Bastian, who lives in Australia.
“In a country that size with that fragmented [of] a health care system, how sure can you be that you know them all? That’s going to be a really difficult situation for them to grapple with, the quality of information that they’ve got,” she said.
A version of this article first appeared on Medscape.com.
on April 14, 2021, after the CDC and Food and Drug Administration recommended that states hold off on using it pending a detailed review of six cases of the same kind of rare but serious event – a blood clot in the vessels that drain blood from the brain combined with a large drop in platelets, which increases the risk for bleeding.
This combination can lead to severe strokes that can lead to brain damage or death. Among the six cases reported, which came to light over the past 3 weeks, one person died, according to the CDC. All six were women and ranged in age from 18 to 48 years.
According to a report from the Vaccine Adverse Event Reporting System (VAERS), which is maintained by the Department of Health & Human Services, the woman who died was 45. She developed a gradually worsening headache about a week after receiving the Johnson & Johnson vaccine.
On March 17, the day she came to the hospital, she was dry heaving. Her headache had suddenly gotten much worse, and the left side of her body was weak, which are signs of a stroke. A CT scan revealed both bleeding in her brain and a clot in her cortical vein. She died the following day.
In addition to VAERS, which accepts reports from anyone, the CDC and FDA are monitoring at least eight other safety systems maintained by hospitals, research centers, long-term care facilities, and insurance companies for signs of trouble with the vaccines. VAERS data is searchable and open to the public. Most of these systems are not publicly available to protect patient privacy. It’s unclear which systems detected the six cases cited by federal regulators.
“These are very serious and potentially fatal problems occurring in a healthy young adult. It’s serious and we need to get to the bottom of it,” said Ed Belongia, MD, director of the Center for Clinical Epidemiology and Population Health at the Marshfield (Wis.) Clinic Research Institute. Dr. Belongia leads a research team that helps the CDC monitor vaccine safety and effectiveness.
“Safety is always the highest priority, and I think what we’ve seen here in the past 24 hours is our vaccine safety monitoring system is working,” he said.
Others agree. “I think what CDC and FDA have detected is a rare, but likely real adverse event associated with this vaccine,” said Paul Offit, MD, director of vaccine education at Children’s Hospital of Philadelphia.
Although much is still unknown about these events, they follow a similar pattern of blood clots reported with the AstraZeneca vaccine in Europe. That vaccine is now sold under the brand name Vaxzevria.
This has experts questioning whether all vaccines of this type may cause these rare clots.
“I think it’s likely a class effect,” said Dr. Offit, who was a member of the FDA advisory committee that reviewed clinical trial data on the J&J vaccine before it was authorized for use.
Adenovirus vaccines scrutinized
Both the Johnson & Johnson and Vaxzevria vaccines use an adenovirus to ferry genetic instructions for making the coronaviruses spike protein into our cells.
Adenoviruses are common, relatively simple viruses that normally cause mild cold or flu symptoms. The ones used in the vaccine are disabled so they can’t make us sick. They’re more like Trojan horses.
Once inside our cells, they release the DNA instructions they carry to make the spike protein of the new coronavirus. Those cells then crank out copies of the spike protein, which then get displayed on the outer surface of the cell membrane where they are recognized by the immune system.
The immune system then makes antibodies and other defenses against the spike so that, when the real coronavirus comes along, our bodies are ready to fight the infection.
There’s no question the vaccine works. In clinical trials, the Johnson & Johnson vaccine was 66% percent effective at preventing against moderate to severe COVID-19 infection, and none of the patients who got COVID-19 after vaccination had to be admitted to the hospital or died.
The idea behind using adenoviruses in vaccines isn’t a new one. In a kind of fight-fire-with-fire approach, the idea is to use a virus, which is good at infecting us, to fight a different kind of virus.
Researchers have been working on the concept for about 10 years, but the COVID-19 vaccines that use this technology are some of the first adenovirus-vector vaccines deployed in humans.
Only one other adenovirus vaccine, for Ebola, has been approved for use in humans. It was approved in Europe last year. Before the Johnson & Johnson vaccine, no other adenovirus vector has been available for use in humans in the United States.
There are six adenovirus-vector vaccines for COVID-19. In addition to AstraZeneca and Johnson & Johnson, there’s the Russian-developed vaccine Sputnik V, along with CanSino from China, and the Covishield vaccine in India.
Adenovirus vaccines are more stable than the mRNA vaccines. That makes them easier to store and transport.
But they have a significant downside, too. Because adenoviruses infect humans out in the world, we already make antibodies against them. So there’s always a danger that our immune systems might recognize and react to the vaccine, rendering it ineffective. For that reason, scientists try to carefully select the adenovirus vectors, or carriers, they use.
The two vaccines under investigation for blood clots are slightly different. The Johnson & Johnson vaccine uses the vector AD26, because most of the population lacks preexisting immunity to it. Vaxzevria uses an adenovirus that infects chimpanzees, called ChAdOx1.
Vaxzevria has been widely used in Europe but has not yet been authorized in the United States.
On April 7, the European Medicines Agency, Europe’s counterpart to the FDA, ruled that unusual blood clots with low blood platelets should be listed as rare side effects on the Vaxzevria vaccine.
The decision came after reviewing 62 cases of cerebral venous sinus thrombosis (CVST) linked to the vaccine and 25 cases of another rare type of clot, called a splanchnic vein thrombosis. Splanchnic veins drain blood from the major organs in the digestive system, including the stomach, liver, and intestines; 18 of those events were fatal.
The reports were culled from reporting in Europe and the United Kingdom, where around 25 million people have received the Vaxzevria vaccine, making these clots exceptionally rare, but serious.
So far, six cases of CVST have been reported in the United States, after more than 7 million doses of the Johnson & Johnson vaccines have been administered.
A key question for U.S. regulators will be the background rate for these types of rare combinations of clots and deplenished platelets. The background rate is the number of events that would be expected to occur naturally in a population of unvaccinated people. On a press call on April 13, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, was asked about the frequency of this dangerous combination. He said the combination of low platelets and clots was so rare that it was hard to pinpoint, but might be somewhere between 2 and 14 cases per million people over the course of a year.
The first Johnson & Johnson doses were given in early March. That means the six cases came to light within the first few weeks of use of the vaccine in the United States, a very short amount of time.
“These were six cases per million people for 2 weeks, which is the same thing as 25 million per year, so it’s clearly above the background rate,” Dr. Offit said.
Studies suggest possible mechanism
On April 9, the New England Journal of Medicine published a detailed evaluation of the 11 patients in Germany and Austria who developed the rare clots after their Vaxzevria vaccines.
The study detected rare antibodies to a signaling protein called platelet factor 4, which helps to coordinate clot formation.
These same type of antibodies form in some people given the blood thinning drug heparin. In those reactions, which are also exceptionally rare, the same type of syndrome develops, leading to large, devastating clots that consume circulating platelets.
It’s not yet clear whether people who develop reactions to the vaccines already have some platelet factor 4 antibodies before they are vaccinated, or whether the vaccines somehow spur the body to make these antibodies, which then launch a kind of autoimmune attack.
The researchers on the paper gave the syndrome a name, vaccine-induced thrombotic thrombocytopenia (VITT).
It’s also not clear why more cases seem to be in women than in men. Andrew Eisenberger, MD, an associate professor of hematology and oncology at Columbia University, New York, said the most common causes of cerebral venous sinus thrombosis have to do with conditions that raise estrogen levels, like pregnancy and hormonal contraception.
“Estrogen naturally leads to changes in several clotting proteins in the blood that may predispose to abnormal blood clotting in a few different sites in the body,” he said. “The clotting changes we are encountering with some of COVID-19 vaccines are likely to be synergistic with the effects of estrogen on the blood.”
No matter the cause, the CDC on April 13 alerted doctors to keep a high index of suspicion for VITT in patients who have received the Johnson & Johnson vaccination within the last 2 weeks. In those patients, the usual course of treatment with blood thinning drugs like heparin may be harmful.
Symptoms to watch for include severe headache or backache, new neurologic symptoms, severe abdominal pain, shortness of breath, leg swelling, tiny red spots on the skin, or easy bruising.
Grappling with evidence
The CDC’s Advisory Committee on Immunization Practices will meet today in an emergency session to review the cases and see if any changes are needed to use of the J&J vaccine in the United States.
Last week, for example, the United Kingdom restricted the use of the AstraZeneca vaccine in people aged younger than 30 years, saying the risks and benefits of vaccination are “more finely balanced” for this age group.
With cases of COVID-19 rising again in the United States, and the Johnson & Johnson vaccine currently the most convenient form of protection against the virus, the committee will have to weigh the risks of that infection against the risk of rare clots caused by vaccination.
They will also likely have to rule out whether any of the cases had COVID. At least one study has reported CVST clots in three patients with confirmed COVID infections. In Europe, COVID infection did not seem to play a role in the formation of the clots with low platelets.
Hilda Bastian, PhD, a clinical trials expert who cofounded the Cochrane Collaboration, said it won’t be an easy task. Much will depend on how certain the committee members feel they know about all the events linked to the vaccine.
“That’s the really, really hard issue from my point of view for them right this moment. Have we missed any? Or how many are we likely to have missed?” asked Dr. Bastian, who lives in Australia.
“In a country that size with that fragmented [of] a health care system, how sure can you be that you know them all? That’s going to be a really difficult situation for them to grapple with, the quality of information that they’ve got,” she said.
A version of this article first appeared on Medscape.com.
on April 14, 2021, after the CDC and Food and Drug Administration recommended that states hold off on using it pending a detailed review of six cases of the same kind of rare but serious event – a blood clot in the vessels that drain blood from the brain combined with a large drop in platelets, which increases the risk for bleeding.
This combination can lead to severe strokes that can lead to brain damage or death. Among the six cases reported, which came to light over the past 3 weeks, one person died, according to the CDC. All six were women and ranged in age from 18 to 48 years.
According to a report from the Vaccine Adverse Event Reporting System (VAERS), which is maintained by the Department of Health & Human Services, the woman who died was 45. She developed a gradually worsening headache about a week after receiving the Johnson & Johnson vaccine.
On March 17, the day she came to the hospital, she was dry heaving. Her headache had suddenly gotten much worse, and the left side of her body was weak, which are signs of a stroke. A CT scan revealed both bleeding in her brain and a clot in her cortical vein. She died the following day.
In addition to VAERS, which accepts reports from anyone, the CDC and FDA are monitoring at least eight other safety systems maintained by hospitals, research centers, long-term care facilities, and insurance companies for signs of trouble with the vaccines. VAERS data is searchable and open to the public. Most of these systems are not publicly available to protect patient privacy. It’s unclear which systems detected the six cases cited by federal regulators.
“These are very serious and potentially fatal problems occurring in a healthy young adult. It’s serious and we need to get to the bottom of it,” said Ed Belongia, MD, director of the Center for Clinical Epidemiology and Population Health at the Marshfield (Wis.) Clinic Research Institute. Dr. Belongia leads a research team that helps the CDC monitor vaccine safety and effectiveness.
“Safety is always the highest priority, and I think what we’ve seen here in the past 24 hours is our vaccine safety monitoring system is working,” he said.
Others agree. “I think what CDC and FDA have detected is a rare, but likely real adverse event associated with this vaccine,” said Paul Offit, MD, director of vaccine education at Children’s Hospital of Philadelphia.
Although much is still unknown about these events, they follow a similar pattern of blood clots reported with the AstraZeneca vaccine in Europe. That vaccine is now sold under the brand name Vaxzevria.
This has experts questioning whether all vaccines of this type may cause these rare clots.
“I think it’s likely a class effect,” said Dr. Offit, who was a member of the FDA advisory committee that reviewed clinical trial data on the J&J vaccine before it was authorized for use.
Adenovirus vaccines scrutinized
Both the Johnson & Johnson and Vaxzevria vaccines use an adenovirus to ferry genetic instructions for making the coronaviruses spike protein into our cells.
Adenoviruses are common, relatively simple viruses that normally cause mild cold or flu symptoms. The ones used in the vaccine are disabled so they can’t make us sick. They’re more like Trojan horses.
Once inside our cells, they release the DNA instructions they carry to make the spike protein of the new coronavirus. Those cells then crank out copies of the spike protein, which then get displayed on the outer surface of the cell membrane where they are recognized by the immune system.
The immune system then makes antibodies and other defenses against the spike so that, when the real coronavirus comes along, our bodies are ready to fight the infection.
There’s no question the vaccine works. In clinical trials, the Johnson & Johnson vaccine was 66% percent effective at preventing against moderate to severe COVID-19 infection, and none of the patients who got COVID-19 after vaccination had to be admitted to the hospital or died.
The idea behind using adenoviruses in vaccines isn’t a new one. In a kind of fight-fire-with-fire approach, the idea is to use a virus, which is good at infecting us, to fight a different kind of virus.
Researchers have been working on the concept for about 10 years, but the COVID-19 vaccines that use this technology are some of the first adenovirus-vector vaccines deployed in humans.
Only one other adenovirus vaccine, for Ebola, has been approved for use in humans. It was approved in Europe last year. Before the Johnson & Johnson vaccine, no other adenovirus vector has been available for use in humans in the United States.
There are six adenovirus-vector vaccines for COVID-19. In addition to AstraZeneca and Johnson & Johnson, there’s the Russian-developed vaccine Sputnik V, along with CanSino from China, and the Covishield vaccine in India.
Adenovirus vaccines are more stable than the mRNA vaccines. That makes them easier to store and transport.
But they have a significant downside, too. Because adenoviruses infect humans out in the world, we already make antibodies against them. So there’s always a danger that our immune systems might recognize and react to the vaccine, rendering it ineffective. For that reason, scientists try to carefully select the adenovirus vectors, or carriers, they use.
The two vaccines under investigation for blood clots are slightly different. The Johnson & Johnson vaccine uses the vector AD26, because most of the population lacks preexisting immunity to it. Vaxzevria uses an adenovirus that infects chimpanzees, called ChAdOx1.
Vaxzevria has been widely used in Europe but has not yet been authorized in the United States.
On April 7, the European Medicines Agency, Europe’s counterpart to the FDA, ruled that unusual blood clots with low blood platelets should be listed as rare side effects on the Vaxzevria vaccine.
The decision came after reviewing 62 cases of cerebral venous sinus thrombosis (CVST) linked to the vaccine and 25 cases of another rare type of clot, called a splanchnic vein thrombosis. Splanchnic veins drain blood from the major organs in the digestive system, including the stomach, liver, and intestines; 18 of those events were fatal.
The reports were culled from reporting in Europe and the United Kingdom, where around 25 million people have received the Vaxzevria vaccine, making these clots exceptionally rare, but serious.
So far, six cases of CVST have been reported in the United States, after more than 7 million doses of the Johnson & Johnson vaccines have been administered.
A key question for U.S. regulators will be the background rate for these types of rare combinations of clots and deplenished platelets. The background rate is the number of events that would be expected to occur naturally in a population of unvaccinated people. On a press call on April 13, Peter Marks, MD, PhD, director of the FDA’s Center for Biologics Evaluation and Research, was asked about the frequency of this dangerous combination. He said the combination of low platelets and clots was so rare that it was hard to pinpoint, but might be somewhere between 2 and 14 cases per million people over the course of a year.
The first Johnson & Johnson doses were given in early March. That means the six cases came to light within the first few weeks of use of the vaccine in the United States, a very short amount of time.
“These were six cases per million people for 2 weeks, which is the same thing as 25 million per year, so it’s clearly above the background rate,” Dr. Offit said.
Studies suggest possible mechanism
On April 9, the New England Journal of Medicine published a detailed evaluation of the 11 patients in Germany and Austria who developed the rare clots after their Vaxzevria vaccines.
The study detected rare antibodies to a signaling protein called platelet factor 4, which helps to coordinate clot formation.
These same type of antibodies form in some people given the blood thinning drug heparin. In those reactions, which are also exceptionally rare, the same type of syndrome develops, leading to large, devastating clots that consume circulating platelets.
It’s not yet clear whether people who develop reactions to the vaccines already have some platelet factor 4 antibodies before they are vaccinated, or whether the vaccines somehow spur the body to make these antibodies, which then launch a kind of autoimmune attack.
The researchers on the paper gave the syndrome a name, vaccine-induced thrombotic thrombocytopenia (VITT).
It’s also not clear why more cases seem to be in women than in men. Andrew Eisenberger, MD, an associate professor of hematology and oncology at Columbia University, New York, said the most common causes of cerebral venous sinus thrombosis have to do with conditions that raise estrogen levels, like pregnancy and hormonal contraception.
“Estrogen naturally leads to changes in several clotting proteins in the blood that may predispose to abnormal blood clotting in a few different sites in the body,” he said. “The clotting changes we are encountering with some of COVID-19 vaccines are likely to be synergistic with the effects of estrogen on the blood.”
No matter the cause, the CDC on April 13 alerted doctors to keep a high index of suspicion for VITT in patients who have received the Johnson & Johnson vaccination within the last 2 weeks. In those patients, the usual course of treatment with blood thinning drugs like heparin may be harmful.
Symptoms to watch for include severe headache or backache, new neurologic symptoms, severe abdominal pain, shortness of breath, leg swelling, tiny red spots on the skin, or easy bruising.
Grappling with evidence
The CDC’s Advisory Committee on Immunization Practices will meet today in an emergency session to review the cases and see if any changes are needed to use of the J&J vaccine in the United States.
Last week, for example, the United Kingdom restricted the use of the AstraZeneca vaccine in people aged younger than 30 years, saying the risks and benefits of vaccination are “more finely balanced” for this age group.
With cases of COVID-19 rising again in the United States, and the Johnson & Johnson vaccine currently the most convenient form of protection against the virus, the committee will have to weigh the risks of that infection against the risk of rare clots caused by vaccination.
They will also likely have to rule out whether any of the cases had COVID. At least one study has reported CVST clots in three patients with confirmed COVID infections. In Europe, COVID infection did not seem to play a role in the formation of the clots with low platelets.
Hilda Bastian, PhD, a clinical trials expert who cofounded the Cochrane Collaboration, said it won’t be an easy task. Much will depend on how certain the committee members feel they know about all the events linked to the vaccine.
“That’s the really, really hard issue from my point of view for them right this moment. Have we missed any? Or how many are we likely to have missed?” asked Dr. Bastian, who lives in Australia.
“In a country that size with that fragmented [of] a health care system, how sure can you be that you know them all? That’s going to be a really difficult situation for them to grapple with, the quality of information that they’ve got,” she said.
A version of this article first appeared on Medscape.com.
Arthritis drug may curb myocardial damage in acute STEMI
Early use of tocilizumab (Actemra) does not reduce myocardial infarct size but modestly increases myocardial salvage in patients with acute ST-segment elevation MI (STEMI), results of the ASSAIL-MI trial suggest.
“We’re among the first to show that you can actually affect the reperfusion injury through anti-inflammatory treatment – it’s sort of a new attack point for treatments in STEMI,” lead author Kaspar Broch, MD, PhD, Oslo University Hospital Rikshospitalet, said in an interview. “What we do now is reperfuse as soon as we can and then add drugs in order to prevent new events, but we don’t really attack the reperfusion injury that occurs when you perform PCI [percutaneous coronary intervention], which has been shown to actually account for some 50% of the final injury.”
The phase 2, proof-of-concept study was prompted by the team’s earlier work in non-STEMI patients, in which a single dose of the interleukin-6 receptor antagonist cut C-reactive protein (CRP) levels by more than 50% during hospitalization and reduced troponin T release after PCI.
For ASSAIL-MI, Dr. Broch and colleagues randomly assigned 199 patients presenting with acute STEMI within 6 hours of symptom onset to a single intravenous injection of 280 mg tocilizumab or placebo during PCI. Patients, study personnel, and caretakers were blinded to treatment. Data were available for 195 patients for the primary endpoint of myocardial salvage index.
As reported in the Journal of the American College of Cardiology, tocilizumab was associated with a higher adjusted myocardial salvage index on cardiac MRI 3-7 days after PCI than placebo (69.3% vs. 63.6%; P = .04).
The extent of microvascular obstruction was less with tocilizumab (0% vs. 4%; P = .03), as was the area under the curve of CRP during hospitalization (1.9 vs. 8.6 mg/L per hour; P < .001).
The final infarct size at 6 months was 21% lower in the tocilizumab group but the difference did not reach statistical significance (7.2% vs. 9.1% of left ventricular mass; P = .08).
There were no between-group differences in troponin T area under the curve during hospitalization (1,614 vs. 2,357 ng/L per hour; P = .13), N-terminal of the prohormone brain natriuretic peptide concentrations at 6 months (79 vs. 63 ng/L; P = .25), or baseline-adjusted left ventricular end-diastolic volume at 6 months (157 vs. 160 mL; P = .54).
Subgroup analyses suggested the positive effect of tocilizumab on myocardial salvage index is limited to patients presenting at least 3 hours after symptom onset versus 3 hours or less (P = .034), with a trend for greater benefit among men versus women (P = .053).
Dr. Broch noted that the absolute effect of tocilizumab on myocardial necrosis was smaller than anticipated when the trial was designed, which may explain the lack of significant reduction in infarct size.
“We were aiming for patients with larger infarctions than we actually ended up with, which is partly due to the strict inclusion criteria and the fact that, with modern treatments, patients don’t end up with large myocardial infarctions,” he said. “But if they had been larger, I think that 20% absolute reduction would have meant a lot in terms of clinical events.”
The study also used a very modest dose of tocilizumab, compared with that used for inflammatory diseases, to minimize a potential negative effect on myocardial healing, for instance, myocardial ruptures, Dr. Broch said. “I’m not sure whether you gain anything by giving a larger dose.”
Serious adverse events were similar in the tocilizumab and placebo groups (19 vs. 15; P = .57). There were no myocardial ruptures, and no patient died or developed heart failure. LDL cholesterol, triglycerides, and liver enzymes increased in the tocilizumab group but were similar at 3 and 6 months.
“IL-6 is a central cytokine involved in all stages of plaque growth, progression, and rupture,” Paul Ridker, MD, MPH, of the Brigham and Women’s Hospital in Boston, and a long-standing investigator in inflammation and atherothrombosis, said in an interview. “These preliminary data in STEMI, like the authors’ prior data in non-STEMI, are consistent with the idea that inhibiting IL-6 could have clinical benefit, a concept that will be taken into a major cardiovascular outcomes trial later this year.”
The cardiovascular outcomes trial, known as ZEUS, will test the novel IL-6 inhibitor ziltivekimab among more than 6,000 very-high-risk atherosclerosis patients who have moderate to severe chronic kidney disease and high sensitivity CRP greater than 2 mg/L, he noted.
Moving beyond IL-1b blockade as done in CANTOS to direct downstream inhibition of IL-6 represents a “logical next scientific step” in the development of anti-inflammatory therapies for acute ischemia and chronic atherosclerosis, Dr. Ridker, who led the CANTOS trial, noted in an accompanying editorial.
“Preventive cardiologists, however, need not wait until outcome trials are complete to use this evolving biological knowledge to their patient’s advantage,” he wrote. “As recently confirmed in the pages of the Journal, exercise, smoking cessation, and a healthy diet reduce both C-reactive protein and IL-6, and clearly have lifelong benefits. Our immediate task is thus to incorporate inflammation inhibition through lifestyle management into our daily practice.”
The study was supported by the South-Eastern Norway Regional Health Authority, Central Norway Regional Health Authority, and Roche, which provided the medicinal products and an unrestricted grant. Dr. Broch has disclosed no relevant financial relationships. Dr. Ridker has received investigator-initiated research grant support from Kowa, Novartis, Amarin, Pfizer, and the National Heart, Lung, and Blood Institute; and has served as a consultant to Novartis, Janssen, Agepha, Flame, Civi Biopharma, Inflazome, Corvidia, Novo Nordisk, SOCAR, IQVIA, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Early use of tocilizumab (Actemra) does not reduce myocardial infarct size but modestly increases myocardial salvage in patients with acute ST-segment elevation MI (STEMI), results of the ASSAIL-MI trial suggest.
“We’re among the first to show that you can actually affect the reperfusion injury through anti-inflammatory treatment – it’s sort of a new attack point for treatments in STEMI,” lead author Kaspar Broch, MD, PhD, Oslo University Hospital Rikshospitalet, said in an interview. “What we do now is reperfuse as soon as we can and then add drugs in order to prevent new events, but we don’t really attack the reperfusion injury that occurs when you perform PCI [percutaneous coronary intervention], which has been shown to actually account for some 50% of the final injury.”
The phase 2, proof-of-concept study was prompted by the team’s earlier work in non-STEMI patients, in which a single dose of the interleukin-6 receptor antagonist cut C-reactive protein (CRP) levels by more than 50% during hospitalization and reduced troponin T release after PCI.
For ASSAIL-MI, Dr. Broch and colleagues randomly assigned 199 patients presenting with acute STEMI within 6 hours of symptom onset to a single intravenous injection of 280 mg tocilizumab or placebo during PCI. Patients, study personnel, and caretakers were blinded to treatment. Data were available for 195 patients for the primary endpoint of myocardial salvage index.
As reported in the Journal of the American College of Cardiology, tocilizumab was associated with a higher adjusted myocardial salvage index on cardiac MRI 3-7 days after PCI than placebo (69.3% vs. 63.6%; P = .04).
The extent of microvascular obstruction was less with tocilizumab (0% vs. 4%; P = .03), as was the area under the curve of CRP during hospitalization (1.9 vs. 8.6 mg/L per hour; P < .001).
The final infarct size at 6 months was 21% lower in the tocilizumab group but the difference did not reach statistical significance (7.2% vs. 9.1% of left ventricular mass; P = .08).
There were no between-group differences in troponin T area under the curve during hospitalization (1,614 vs. 2,357 ng/L per hour; P = .13), N-terminal of the prohormone brain natriuretic peptide concentrations at 6 months (79 vs. 63 ng/L; P = .25), or baseline-adjusted left ventricular end-diastolic volume at 6 months (157 vs. 160 mL; P = .54).
Subgroup analyses suggested the positive effect of tocilizumab on myocardial salvage index is limited to patients presenting at least 3 hours after symptom onset versus 3 hours or less (P = .034), with a trend for greater benefit among men versus women (P = .053).
Dr. Broch noted that the absolute effect of tocilizumab on myocardial necrosis was smaller than anticipated when the trial was designed, which may explain the lack of significant reduction in infarct size.
“We were aiming for patients with larger infarctions than we actually ended up with, which is partly due to the strict inclusion criteria and the fact that, with modern treatments, patients don’t end up with large myocardial infarctions,” he said. “But if they had been larger, I think that 20% absolute reduction would have meant a lot in terms of clinical events.”
The study also used a very modest dose of tocilizumab, compared with that used for inflammatory diseases, to minimize a potential negative effect on myocardial healing, for instance, myocardial ruptures, Dr. Broch said. “I’m not sure whether you gain anything by giving a larger dose.”
Serious adverse events were similar in the tocilizumab and placebo groups (19 vs. 15; P = .57). There were no myocardial ruptures, and no patient died or developed heart failure. LDL cholesterol, triglycerides, and liver enzymes increased in the tocilizumab group but were similar at 3 and 6 months.
“IL-6 is a central cytokine involved in all stages of plaque growth, progression, and rupture,” Paul Ridker, MD, MPH, of the Brigham and Women’s Hospital in Boston, and a long-standing investigator in inflammation and atherothrombosis, said in an interview. “These preliminary data in STEMI, like the authors’ prior data in non-STEMI, are consistent with the idea that inhibiting IL-6 could have clinical benefit, a concept that will be taken into a major cardiovascular outcomes trial later this year.”
The cardiovascular outcomes trial, known as ZEUS, will test the novel IL-6 inhibitor ziltivekimab among more than 6,000 very-high-risk atherosclerosis patients who have moderate to severe chronic kidney disease and high sensitivity CRP greater than 2 mg/L, he noted.
Moving beyond IL-1b blockade as done in CANTOS to direct downstream inhibition of IL-6 represents a “logical next scientific step” in the development of anti-inflammatory therapies for acute ischemia and chronic atherosclerosis, Dr. Ridker, who led the CANTOS trial, noted in an accompanying editorial.
“Preventive cardiologists, however, need not wait until outcome trials are complete to use this evolving biological knowledge to their patient’s advantage,” he wrote. “As recently confirmed in the pages of the Journal, exercise, smoking cessation, and a healthy diet reduce both C-reactive protein and IL-6, and clearly have lifelong benefits. Our immediate task is thus to incorporate inflammation inhibition through lifestyle management into our daily practice.”
The study was supported by the South-Eastern Norway Regional Health Authority, Central Norway Regional Health Authority, and Roche, which provided the medicinal products and an unrestricted grant. Dr. Broch has disclosed no relevant financial relationships. Dr. Ridker has received investigator-initiated research grant support from Kowa, Novartis, Amarin, Pfizer, and the National Heart, Lung, and Blood Institute; and has served as a consultant to Novartis, Janssen, Agepha, Flame, Civi Biopharma, Inflazome, Corvidia, Novo Nordisk, SOCAR, IQVIA, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Early use of tocilizumab (Actemra) does not reduce myocardial infarct size but modestly increases myocardial salvage in patients with acute ST-segment elevation MI (STEMI), results of the ASSAIL-MI trial suggest.
“We’re among the first to show that you can actually affect the reperfusion injury through anti-inflammatory treatment – it’s sort of a new attack point for treatments in STEMI,” lead author Kaspar Broch, MD, PhD, Oslo University Hospital Rikshospitalet, said in an interview. “What we do now is reperfuse as soon as we can and then add drugs in order to prevent new events, but we don’t really attack the reperfusion injury that occurs when you perform PCI [percutaneous coronary intervention], which has been shown to actually account for some 50% of the final injury.”
The phase 2, proof-of-concept study was prompted by the team’s earlier work in non-STEMI patients, in which a single dose of the interleukin-6 receptor antagonist cut C-reactive protein (CRP) levels by more than 50% during hospitalization and reduced troponin T release after PCI.
For ASSAIL-MI, Dr. Broch and colleagues randomly assigned 199 patients presenting with acute STEMI within 6 hours of symptom onset to a single intravenous injection of 280 mg tocilizumab or placebo during PCI. Patients, study personnel, and caretakers were blinded to treatment. Data were available for 195 patients for the primary endpoint of myocardial salvage index.
As reported in the Journal of the American College of Cardiology, tocilizumab was associated with a higher adjusted myocardial salvage index on cardiac MRI 3-7 days after PCI than placebo (69.3% vs. 63.6%; P = .04).
The extent of microvascular obstruction was less with tocilizumab (0% vs. 4%; P = .03), as was the area under the curve of CRP during hospitalization (1.9 vs. 8.6 mg/L per hour; P < .001).
The final infarct size at 6 months was 21% lower in the tocilizumab group but the difference did not reach statistical significance (7.2% vs. 9.1% of left ventricular mass; P = .08).
There were no between-group differences in troponin T area under the curve during hospitalization (1,614 vs. 2,357 ng/L per hour; P = .13), N-terminal of the prohormone brain natriuretic peptide concentrations at 6 months (79 vs. 63 ng/L; P = .25), or baseline-adjusted left ventricular end-diastolic volume at 6 months (157 vs. 160 mL; P = .54).
Subgroup analyses suggested the positive effect of tocilizumab on myocardial salvage index is limited to patients presenting at least 3 hours after symptom onset versus 3 hours or less (P = .034), with a trend for greater benefit among men versus women (P = .053).
Dr. Broch noted that the absolute effect of tocilizumab on myocardial necrosis was smaller than anticipated when the trial was designed, which may explain the lack of significant reduction in infarct size.
“We were aiming for patients with larger infarctions than we actually ended up with, which is partly due to the strict inclusion criteria and the fact that, with modern treatments, patients don’t end up with large myocardial infarctions,” he said. “But if they had been larger, I think that 20% absolute reduction would have meant a lot in terms of clinical events.”
The study also used a very modest dose of tocilizumab, compared with that used for inflammatory diseases, to minimize a potential negative effect on myocardial healing, for instance, myocardial ruptures, Dr. Broch said. “I’m not sure whether you gain anything by giving a larger dose.”
Serious adverse events were similar in the tocilizumab and placebo groups (19 vs. 15; P = .57). There were no myocardial ruptures, and no patient died or developed heart failure. LDL cholesterol, triglycerides, and liver enzymes increased in the tocilizumab group but were similar at 3 and 6 months.
“IL-6 is a central cytokine involved in all stages of plaque growth, progression, and rupture,” Paul Ridker, MD, MPH, of the Brigham and Women’s Hospital in Boston, and a long-standing investigator in inflammation and atherothrombosis, said in an interview. “These preliminary data in STEMI, like the authors’ prior data in non-STEMI, are consistent with the idea that inhibiting IL-6 could have clinical benefit, a concept that will be taken into a major cardiovascular outcomes trial later this year.”
The cardiovascular outcomes trial, known as ZEUS, will test the novel IL-6 inhibitor ziltivekimab among more than 6,000 very-high-risk atherosclerosis patients who have moderate to severe chronic kidney disease and high sensitivity CRP greater than 2 mg/L, he noted.
Moving beyond IL-1b blockade as done in CANTOS to direct downstream inhibition of IL-6 represents a “logical next scientific step” in the development of anti-inflammatory therapies for acute ischemia and chronic atherosclerosis, Dr. Ridker, who led the CANTOS trial, noted in an accompanying editorial.
“Preventive cardiologists, however, need not wait until outcome trials are complete to use this evolving biological knowledge to their patient’s advantage,” he wrote. “As recently confirmed in the pages of the Journal, exercise, smoking cessation, and a healthy diet reduce both C-reactive protein and IL-6, and clearly have lifelong benefits. Our immediate task is thus to incorporate inflammation inhibition through lifestyle management into our daily practice.”
The study was supported by the South-Eastern Norway Regional Health Authority, Central Norway Regional Health Authority, and Roche, which provided the medicinal products and an unrestricted grant. Dr. Broch has disclosed no relevant financial relationships. Dr. Ridker has received investigator-initiated research grant support from Kowa, Novartis, Amarin, Pfizer, and the National Heart, Lung, and Blood Institute; and has served as a consultant to Novartis, Janssen, Agepha, Flame, Civi Biopharma, Inflazome, Corvidia, Novo Nordisk, SOCAR, IQVIA, and AstraZeneca.
A version of this article first appeared on Medscape.com.
Medtronic recall of almost 240,000 ICDs is class I, FDA says
The Food and Drug Administration has declared Medtronic’s recall of seven models of defibrillating cardiac rhythm devices, caused by a risk for premature battery depletion, as class I, which implies a potential risk for serious injury or death. A total of 444 complaints, but no deaths, have been reported in association with the 239,171 affected devices, the agency said in a statement on April 12, 2021.
Physicians were notified of the company’s recall in early February. It covered implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy–defibrillator (CRT-D) models Evera, Viva, Brava, Claria, Amplia, Compia, and Visia distributed from Aug. 31, 2012 to May 9, 2018.
The devices could be subject to “an unexpected and rapid decrease in battery life” because of a possible short circuit that could lead to a device-replacement alert “earlier than expected.” Some devices may experience full battery depletion “within as little as 1 day” after such an alert.
“If the user does not respond to the first warning, the device may stop functioning. The likelihood that this issue will occur is constant after approximately 3 years after device use,” the announcement said.
Medtronic recommends device replacement no more than 1 week after such an early warning for patients who are not pacing dependent or who have them for primary prevention, but right away for pacing-dependent patients.
A version of this article first appeared on Medscape.com
The Food and Drug Administration has declared Medtronic’s recall of seven models of defibrillating cardiac rhythm devices, caused by a risk for premature battery depletion, as class I, which implies a potential risk for serious injury or death. A total of 444 complaints, but no deaths, have been reported in association with the 239,171 affected devices, the agency said in a statement on April 12, 2021.
Physicians were notified of the company’s recall in early February. It covered implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy–defibrillator (CRT-D) models Evera, Viva, Brava, Claria, Amplia, Compia, and Visia distributed from Aug. 31, 2012 to May 9, 2018.
The devices could be subject to “an unexpected and rapid decrease in battery life” because of a possible short circuit that could lead to a device-replacement alert “earlier than expected.” Some devices may experience full battery depletion “within as little as 1 day” after such an alert.
“If the user does not respond to the first warning, the device may stop functioning. The likelihood that this issue will occur is constant after approximately 3 years after device use,” the announcement said.
Medtronic recommends device replacement no more than 1 week after such an early warning for patients who are not pacing dependent or who have them for primary prevention, but right away for pacing-dependent patients.
A version of this article first appeared on Medscape.com
The Food and Drug Administration has declared Medtronic’s recall of seven models of defibrillating cardiac rhythm devices, caused by a risk for premature battery depletion, as class I, which implies a potential risk for serious injury or death. A total of 444 complaints, but no deaths, have been reported in association with the 239,171 affected devices, the agency said in a statement on April 12, 2021.
Physicians were notified of the company’s recall in early February. It covered implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy–defibrillator (CRT-D) models Evera, Viva, Brava, Claria, Amplia, Compia, and Visia distributed from Aug. 31, 2012 to May 9, 2018.
The devices could be subject to “an unexpected and rapid decrease in battery life” because of a possible short circuit that could lead to a device-replacement alert “earlier than expected.” Some devices may experience full battery depletion “within as little as 1 day” after such an alert.
“If the user does not respond to the first warning, the device may stop functioning. The likelihood that this issue will occur is constant after approximately 3 years after device use,” the announcement said.
Medtronic recommends device replacement no more than 1 week after such an early warning for patients who are not pacing dependent or who have them for primary prevention, but right away for pacing-dependent patients.
A version of this article first appeared on Medscape.com
Remote cardio visits expand access for underserved during COVID
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Remote cardiology clinic visits during COVID-19 were used more often by certain traditionally underserved patient groups, but were also associated with less frequent testing and prescribing, new research shows.
“The COVID-19 pandemic has led to an unprecedented shift in ambulatory cardiovascular care from in-person to remote visits,” lead author Neal Yuan, MD, a cardiology fellow at the Smidt Heart Institute, Cedars-Sinai Medical Center, Los Angeles, said in an interview.
Their findings were published online April 5 in JAMA Network Open.
“We wanted to explore whether the transition to remote visits was associated with disparities in how patients accessed care, and also how this transition affected diagnostic test ordering and medication prescribing,” Dr. Yuan said.
The researchers used electronic health records data for all ambulatory cardiology visits at an urban, multisite health system in Los Angeles County during two periods: April 1 to Dec. 31, 2019, the pre-COVID era; and April 1 to Dec. 31, 2020, the COVID era.
The investigators compared patient characteristics and frequencies of medication ordering and cardiology-specific testing across four visit types: pre-COVID in person, used as reference; COVID-era in person; COVID-era video; and COVID-era telephone.
The study looked at 176,781 ambulatory cardiology visits. Of these visits, 87,182 were conducted in person in the pre-COVID period; 74,498 were conducted in person in the COVID era; 4,720 were COVID-era video visits; and 10,381 were COVID-era telephone visits.
In the study cohort, 79,572 patients (45.0%) were female, 127,080 patients (71.9%) were non-Hispanic White, and the mean age was 68.1 years (standard deviation, 17.0).
Patients accessing COVID-era remote visits were more likely to be Asian, Black, or Hispanic, to have private insurance, and to have cardiovascular comorbidities, such as hypertension and heart failure.
Also, patients whose visits were conducted by video were significantly younger than patients whose visits were conducted in person or by telephone (P < .001).
In addition, the study found that clinicians ordered fewer diagnostic tests, such as electrocardiograms and echocardiograms, and were less likely to order any medication, in the pre-COVID era than during the COVID era.
“If you don’t have a patient in front of you, it’s much more difficult to get a physical exam or obtain reliable vital signs,” said Dr. Yuan. Communication can sometimes be difficult, often because of technical issues, like a bad connection. “You might be more reticent to get testing or to prescribe medications if you don’t feel confident knowing what the patient’s vital signs are.”
In addition, he added, “a lot of medications used in the cardiology setting require monitoring patients’ kidney function and electrolytes, and if you can’t do that reliably, you might be more cautious about prescribing those types of medications.”
An eye-opening study
Cardiologist Nieca Goldberg, MD, medical director of the New York University Langone womens’ heart program and spokesperson for the American Heart Association, recounted her experience with telemedicine at the height of the pandemic in New York, when everything, including medical outpatient offices, had to close.
“We were experienced with telemedicine because we had started a virtual urgent care program well ahead of the pandemic,” she said. “We started using that to screen people with potential COVID symptoms so that they wouldn’t have to come into the hospital, the medical center, or to the offices and expose people. We learned that it was great to have the telemedicine option from the infectious disease standpoint, and I did visits like that for my own patient population.”
An equally if not more important finding from the study is the fact that telemedicine increased access to care among traditionally underserved demographics, she said.
“This is eye-opening, that you can actually improve access to care by doing telemedicine visits. It was really important to see that telemedicine has added benefit to the way we can see people in the health care system.”
Telemedicine visits had a positive impact at a time when people were isolated at home, Dr. Goldberg said.
“It was a way for them to connect with their doctor and in some ways it was more personal,” she added. “I actually got to meet some of my patients’ family members. It was like making a remote house call.”
Stable cardiology patients can take their blood pressure at home, weigh themselves, and take their own pulse to give an excellent set of vital signs that will indicate how they are doing, said Dr. Goldberg.
“During a remote visit, we can talk to the patient and notice whether or not they are short of breath or coughing, but we can’t listen to their heart or do an EKG or any of the traditional cardiac testing. Still, for someone who is not having symptoms and is able to reliably monitor their blood pressure and weight, a remote visit is sufficient to give you a good sense of how that patient is doing,” she said. “We can talk to them about their medications, any potential side effects, and we can use their blood pressure information to adjust their medications.”
Many patients are becoming more savvy about using tech gadgets and devices to monitor their health.
“Some of my patients were using Apple watches and the Kardia app to address their heart rate. Many had purchased inexpensive pulse oximeters to check their oxygen during the pandemic, and that also reads the pulse,” Dr. Goldberg said.
In-person visits were reserved for symptomatic cardiac patients, she explained.
“Initially during the pandemic, we did mostly telemedicine visits and we organized the office so that each cardiologist would come in 1 day a week to take care of symptomatic cardiac patients. In that way, we were able to socially distance – they provided us with [personal protective equipment]; at NYU there was no problem with that – and nobody waited in the waiting room. To this day, office issues are more efficient and people are not waiting in the waiting room,” she added. “Telemedicine improves access to health care in populations where such access is limited.”
Dr. Yuan’s research is supported by a grant from the National Institutes of Health. Dr. Goldberg reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
23-year-old woman • syncopal episode • sinus bradycardia • history of bipolar disorder • Dx?
THE CASE
A 23-year-old woman with past medical history of bipolar II disorder and a REM-specific seizure disorder that resolved at age 9 presented after a syncopal episode. The patient reported an initial sensation of lightheadedness while at work, which was followed by a syncopal episode with brief (1-2 min) loss of consciousness and a minor head injury.
She denied other prodromal symptoms including chest pain, shortness of breath, palpitations, and nausea. She also did not experience convulsions, urinary/bowel incontinence, or confusion upon regaining consciousness.
She denied previous syncopal episodes. However, she reported that, 2 weeks prior, there had been an event similar to that of her presenting complaint. During that episode, she experienced lightheadedness and a fall without loss of consciousness.
The patient had been prescribed a regimen of sertraline 100 mg/d and aripiprazole 10 mg/d to maintain mood stability. She had self-discontinued these medications about 8 months prior to presentation. A recent return of her depressive features had prompted a restart of this regimen 1 week before her first fall, without an initial taper upward.
While in the emergency department, she became bradycardic (heart rate, 38 beats/min) and hypotensive (blood pressure, 70/40 mm Hg). She subsequently became increasingly somnolent and had 1 episode of emesis. An electrocardiogram (EKG) revealed sinus bradycardia without other acute abnormalities (FIGURE).
Blood work including a basic metabolic panel, complete blood count, and cardiac enzymes were all within normal limits. Computed tomography of the head revealed no intracranial pathology. Her vitals were initially unresponsive to a fluid bolus but improved and stabilized after administration of intravenous atropine 0.5 mg.
Aripiprazole was held and sertraline was decreased to 75 mg on hospital Day 1, with close monitoring of her mood. Cardiology was consulted and followed the patient during her stay. The patient was monitored on telemetry for 3 days, exhibiting only sinus bradycardia with a stable heart rate of 45-55 beats/min. Systolic blood pressures were stable within 120 to 130 mm Hg. Transthoracic echocardiogram performed on hospital Day 2 was unremarkable, revealing a normal left ventricular ejection fraction of 65% and no wall motion abnormalities. She had no recurrence of the syncope or emesis.
Continue to: THE DIAGNOSIS
THE DIAGNOSIS
Given her benign cardiac work-up and symptom onset coinciding with the abrupt resumption of high doses of aripiprazole after an 8-month abstinence, the patient’s presentation was attributed to a rather uncommon adverse drug reaction to aripiprazole. This has only been described in a few case reports.
DISCUSSION
Aripiprazole (Abilify) is an atypical antipsychotic frequently used in the treatment of psychiatric conditions, including bipolar disorder and schizophrenia. While the specific therapeutic mechanism is unknown, it is believed that drug efficacy is related to partial agonism at dopamine D2, serotonin 5-HT1A, and serotonin 5-HT2A.1 As aripiprazole works on a variety of receptors involved in other physiologic processes, clinical adverse effects have been reported, most of which are associated with the adrenergic alpha1 receptors.1 These include cognitive impairment and seizures. Cardiovascular adverse effects of aripiprazole include orthostatic hypotension, cardiac arrhythmia, prolonged QT interval, and syncope.1-5
Selective serotonin reuptake inhibitors (SSRIs) such as sertraline (Zoloft) have also been shown to cause cardiac arrhythmia and syncope.6 Although sertraline may have contributed to the patient’s cardiac symptoms, it is more likely that the aripiprazole was the direct cause, as she remained asymptomatic while on a therapeutic dose of sertraline. Furthermore, aripiprazole is primarily metabolized though hepatic CYP2D6, which sertraline has been shown to inhibit.1,7 Therefore, the concomitant use of sertraline with no initial taper of either medication likely led to an increased effective dose of aripiprazole in our patient and subsequently to her presentation.
Few prior cases have identified aripiprazole as a cause of antipsychotic-associated bradycardic response.8 Based on the Adverse Drug Reaction Probability Scale, often referred to as the Naranjo Scale, we believe this to be a probable adverse response in our patient.9 Bradycardia followed a reasonable temporal sequence after aripiprazole use with a response previously described in the literature. Symptoms also improved after discontinuation of the drug and other etiologies of the bradycardia were ruled out.
Our patient was discharged with a 30-day cardiac event monitor and a scheduled appointment with Cardiology.
Continue to: THE TAKEAWAY
THE TAKEAWAY
As this case suggests, there may be an association between aripiprazole and symptomatic bradycardia. Therefore, family physicians should inquire about aripiprazole use in patients who present with cardiac symptoms and consider tapering this medication if other causes cannot be identified. Additionally, given the potential cardiac adverse effects of atypical antipsychotics, physicians may consider ordering baseline and follow-up EKGs to monitor for arrhythmias in patients prescribed aripiprazole. This may be especially prudent when an atypical antipsychotic is combined with an SSRI, as potential cardiac adverse effects may occur more frequently.
CORRESPONDENCE
Kyle Fletke, MD, Department of Family and Community Medicine, University of Maryland School of Medicine, 29 South Paca Street, Baltimore, MD 21201; kfletke@som.umaryland.edu
1. Abilify [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc; 2014.
2. Belemonte C, Ochoa D, Román M, et al. Evaluation of the relationship between pharmacokinetics and the safety of aripiprazole and its cardiovascular side effects in health volunteers. J Clin Psychopharmacol. 2016;36:608-614.
3. Torgovnic J, Sethi NK, Arsura E. Aripiprazole-induced orthostatic hypotension and cardiac arrhythmia. Psychiatry Clin Neurosci. 2008:62:485.
4. Pacher P, Kecskemeti V. Cardiovascular side effects of new antidepressants and antipsychotics: new drugs, old concerns? Curr Pharm Des. 2004;10:2463-2475.
5. Russo L, Rizzo A, Di Vincenzo A, et al. Aripiprazole overdose and transient 2:1 second degree atrioventricular block: only a coincidence? Curr Drug Saf. 2019;14:155-157.
6. Pacher P, Ungvari Z, Kecskemeti V, et al. Review of cardiovascular effects of fluoxetine, a selective serotonin reuptake inhibitor, compared to tricyclic antidepressants. Curr Med Chem. 1998;5:381-90.
7. Hemeryck A, Belpaire FM. Selective serotonin reuptake inhibitors and cytochrome P-450 mediated drug-drug interactions: an update. Curr Drub Metab. 2002;3:13-37.
8. Snarr BS, Phan SV, Garner A, et al. Symptomatic bradycardia with oral aripiprazole and oral ziprasidone. Ann Pharmacother. 2010;44:760-763.
9. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.
THE CASE
A 23-year-old woman with past medical history of bipolar II disorder and a REM-specific seizure disorder that resolved at age 9 presented after a syncopal episode. The patient reported an initial sensation of lightheadedness while at work, which was followed by a syncopal episode with brief (1-2 min) loss of consciousness and a minor head injury.
She denied other prodromal symptoms including chest pain, shortness of breath, palpitations, and nausea. She also did not experience convulsions, urinary/bowel incontinence, or confusion upon regaining consciousness.
She denied previous syncopal episodes. However, she reported that, 2 weeks prior, there had been an event similar to that of her presenting complaint. During that episode, she experienced lightheadedness and a fall without loss of consciousness.
The patient had been prescribed a regimen of sertraline 100 mg/d and aripiprazole 10 mg/d to maintain mood stability. She had self-discontinued these medications about 8 months prior to presentation. A recent return of her depressive features had prompted a restart of this regimen 1 week before her first fall, without an initial taper upward.
While in the emergency department, she became bradycardic (heart rate, 38 beats/min) and hypotensive (blood pressure, 70/40 mm Hg). She subsequently became increasingly somnolent and had 1 episode of emesis. An electrocardiogram (EKG) revealed sinus bradycardia without other acute abnormalities (FIGURE).

Blood work including a basic metabolic panel, complete blood count, and cardiac enzymes were all within normal limits. Computed tomography of the head revealed no intracranial pathology. Her vitals were initially unresponsive to a fluid bolus but improved and stabilized after administration of intravenous atropine 0.5 mg.
Aripiprazole was held and sertraline was decreased to 75 mg on hospital Day 1, with close monitoring of her mood. Cardiology was consulted and followed the patient during her stay. The patient was monitored on telemetry for 3 days, exhibiting only sinus bradycardia with a stable heart rate of 45-55 beats/min. Systolic blood pressures were stable within 120 to 130 mm Hg. Transthoracic echocardiogram performed on hospital Day 2 was unremarkable, revealing a normal left ventricular ejection fraction of 65% and no wall motion abnormalities. She had no recurrence of the syncope or emesis.
Continue to: THE DIAGNOSIS
THE DIAGNOSIS
Given her benign cardiac work-up and symptom onset coinciding with the abrupt resumption of high doses of aripiprazole after an 8-month abstinence, the patient’s presentation was attributed to a rather uncommon adverse drug reaction to aripiprazole. This has only been described in a few case reports.
DISCUSSION
Aripiprazole (Abilify) is an atypical antipsychotic frequently used in the treatment of psychiatric conditions, including bipolar disorder and schizophrenia. While the specific therapeutic mechanism is unknown, it is believed that drug efficacy is related to partial agonism at dopamine D2, serotonin 5-HT1A, and serotonin 5-HT2A.1 As aripiprazole works on a variety of receptors involved in other physiologic processes, clinical adverse effects have been reported, most of which are associated with the adrenergic alpha1 receptors.1 These include cognitive impairment and seizures. Cardiovascular adverse effects of aripiprazole include orthostatic hypotension, cardiac arrhythmia, prolonged QT interval, and syncope.1-5
Selective serotonin reuptake inhibitors (SSRIs) such as sertraline (Zoloft) have also been shown to cause cardiac arrhythmia and syncope.6 Although sertraline may have contributed to the patient’s cardiac symptoms, it is more likely that the aripiprazole was the direct cause, as she remained asymptomatic while on a therapeutic dose of sertraline. Furthermore, aripiprazole is primarily metabolized though hepatic CYP2D6, which sertraline has been shown to inhibit.1,7 Therefore, the concomitant use of sertraline with no initial taper of either medication likely led to an increased effective dose of aripiprazole in our patient and subsequently to her presentation.
Few prior cases have identified aripiprazole as a cause of antipsychotic-associated bradycardic response.8 Based on the Adverse Drug Reaction Probability Scale, often referred to as the Naranjo Scale, we believe this to be a probable adverse response in our patient.9 Bradycardia followed a reasonable temporal sequence after aripiprazole use with a response previously described in the literature. Symptoms also improved after discontinuation of the drug and other etiologies of the bradycardia were ruled out.
Our patient was discharged with a 30-day cardiac event monitor and a scheduled appointment with Cardiology.
Continue to: THE TAKEAWAY
THE TAKEAWAY
As this case suggests, there may be an association between aripiprazole and symptomatic bradycardia. Therefore, family physicians should inquire about aripiprazole use in patients who present with cardiac symptoms and consider tapering this medication if other causes cannot be identified. Additionally, given the potential cardiac adverse effects of atypical antipsychotics, physicians may consider ordering baseline and follow-up EKGs to monitor for arrhythmias in patients prescribed aripiprazole. This may be especially prudent when an atypical antipsychotic is combined with an SSRI, as potential cardiac adverse effects may occur more frequently.
CORRESPONDENCE
Kyle Fletke, MD, Department of Family and Community Medicine, University of Maryland School of Medicine, 29 South Paca Street, Baltimore, MD 21201; kfletke@som.umaryland.edu
THE CASE
A 23-year-old woman with past medical history of bipolar II disorder and a REM-specific seizure disorder that resolved at age 9 presented after a syncopal episode. The patient reported an initial sensation of lightheadedness while at work, which was followed by a syncopal episode with brief (1-2 min) loss of consciousness and a minor head injury.
She denied other prodromal symptoms including chest pain, shortness of breath, palpitations, and nausea. She also did not experience convulsions, urinary/bowel incontinence, or confusion upon regaining consciousness.
She denied previous syncopal episodes. However, she reported that, 2 weeks prior, there had been an event similar to that of her presenting complaint. During that episode, she experienced lightheadedness and a fall without loss of consciousness.
The patient had been prescribed a regimen of sertraline 100 mg/d and aripiprazole 10 mg/d to maintain mood stability. She had self-discontinued these medications about 8 months prior to presentation. A recent return of her depressive features had prompted a restart of this regimen 1 week before her first fall, without an initial taper upward.
While in the emergency department, she became bradycardic (heart rate, 38 beats/min) and hypotensive (blood pressure, 70/40 mm Hg). She subsequently became increasingly somnolent and had 1 episode of emesis. An electrocardiogram (EKG) revealed sinus bradycardia without other acute abnormalities (FIGURE).

Blood work including a basic metabolic panel, complete blood count, and cardiac enzymes were all within normal limits. Computed tomography of the head revealed no intracranial pathology. Her vitals were initially unresponsive to a fluid bolus but improved and stabilized after administration of intravenous atropine 0.5 mg.
Aripiprazole was held and sertraline was decreased to 75 mg on hospital Day 1, with close monitoring of her mood. Cardiology was consulted and followed the patient during her stay. The patient was monitored on telemetry for 3 days, exhibiting only sinus bradycardia with a stable heart rate of 45-55 beats/min. Systolic blood pressures were stable within 120 to 130 mm Hg. Transthoracic echocardiogram performed on hospital Day 2 was unremarkable, revealing a normal left ventricular ejection fraction of 65% and no wall motion abnormalities. She had no recurrence of the syncope or emesis.
Continue to: THE DIAGNOSIS
THE DIAGNOSIS
Given her benign cardiac work-up and symptom onset coinciding with the abrupt resumption of high doses of aripiprazole after an 8-month abstinence, the patient’s presentation was attributed to a rather uncommon adverse drug reaction to aripiprazole. This has only been described in a few case reports.
DISCUSSION
Aripiprazole (Abilify) is an atypical antipsychotic frequently used in the treatment of psychiatric conditions, including bipolar disorder and schizophrenia. While the specific therapeutic mechanism is unknown, it is believed that drug efficacy is related to partial agonism at dopamine D2, serotonin 5-HT1A, and serotonin 5-HT2A.1 As aripiprazole works on a variety of receptors involved in other physiologic processes, clinical adverse effects have been reported, most of which are associated with the adrenergic alpha1 receptors.1 These include cognitive impairment and seizures. Cardiovascular adverse effects of aripiprazole include orthostatic hypotension, cardiac arrhythmia, prolonged QT interval, and syncope.1-5
Selective serotonin reuptake inhibitors (SSRIs) such as sertraline (Zoloft) have also been shown to cause cardiac arrhythmia and syncope.6 Although sertraline may have contributed to the patient’s cardiac symptoms, it is more likely that the aripiprazole was the direct cause, as she remained asymptomatic while on a therapeutic dose of sertraline. Furthermore, aripiprazole is primarily metabolized though hepatic CYP2D6, which sertraline has been shown to inhibit.1,7 Therefore, the concomitant use of sertraline with no initial taper of either medication likely led to an increased effective dose of aripiprazole in our patient and subsequently to her presentation.
Few prior cases have identified aripiprazole as a cause of antipsychotic-associated bradycardic response.8 Based on the Adverse Drug Reaction Probability Scale, often referred to as the Naranjo Scale, we believe this to be a probable adverse response in our patient.9 Bradycardia followed a reasonable temporal sequence after aripiprazole use with a response previously described in the literature. Symptoms also improved after discontinuation of the drug and other etiologies of the bradycardia were ruled out.
Our patient was discharged with a 30-day cardiac event monitor and a scheduled appointment with Cardiology.
Continue to: THE TAKEAWAY
THE TAKEAWAY
As this case suggests, there may be an association between aripiprazole and symptomatic bradycardia. Therefore, family physicians should inquire about aripiprazole use in patients who present with cardiac symptoms and consider tapering this medication if other causes cannot be identified. Additionally, given the potential cardiac adverse effects of atypical antipsychotics, physicians may consider ordering baseline and follow-up EKGs to monitor for arrhythmias in patients prescribed aripiprazole. This may be especially prudent when an atypical antipsychotic is combined with an SSRI, as potential cardiac adverse effects may occur more frequently.
CORRESPONDENCE
Kyle Fletke, MD, Department of Family and Community Medicine, University of Maryland School of Medicine, 29 South Paca Street, Baltimore, MD 21201; kfletke@som.umaryland.edu
1. Abilify [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc; 2014.
2. Belemonte C, Ochoa D, Román M, et al. Evaluation of the relationship between pharmacokinetics and the safety of aripiprazole and its cardiovascular side effects in health volunteers. J Clin Psychopharmacol. 2016;36:608-614.
3. Torgovnic J, Sethi NK, Arsura E. Aripiprazole-induced orthostatic hypotension and cardiac arrhythmia. Psychiatry Clin Neurosci. 2008:62:485.
4. Pacher P, Kecskemeti V. Cardiovascular side effects of new antidepressants and antipsychotics: new drugs, old concerns? Curr Pharm Des. 2004;10:2463-2475.
5. Russo L, Rizzo A, Di Vincenzo A, et al. Aripiprazole overdose and transient 2:1 second degree atrioventricular block: only a coincidence? Curr Drug Saf. 2019;14:155-157.
6. Pacher P, Ungvari Z, Kecskemeti V, et al. Review of cardiovascular effects of fluoxetine, a selective serotonin reuptake inhibitor, compared to tricyclic antidepressants. Curr Med Chem. 1998;5:381-90.
7. Hemeryck A, Belpaire FM. Selective serotonin reuptake inhibitors and cytochrome P-450 mediated drug-drug interactions: an update. Curr Drub Metab. 2002;3:13-37.
8. Snarr BS, Phan SV, Garner A, et al. Symptomatic bradycardia with oral aripiprazole and oral ziprasidone. Ann Pharmacother. 2010;44:760-763.
9. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.
1. Abilify [package insert]. Rockville, MD: Otsuka America Pharmaceutical, Inc; 2014.
2. Belemonte C, Ochoa D, Román M, et al. Evaluation of the relationship between pharmacokinetics and the safety of aripiprazole and its cardiovascular side effects in health volunteers. J Clin Psychopharmacol. 2016;36:608-614.
3. Torgovnic J, Sethi NK, Arsura E. Aripiprazole-induced orthostatic hypotension and cardiac arrhythmia. Psychiatry Clin Neurosci. 2008:62:485.
4. Pacher P, Kecskemeti V. Cardiovascular side effects of new antidepressants and antipsychotics: new drugs, old concerns? Curr Pharm Des. 2004;10:2463-2475.
5. Russo L, Rizzo A, Di Vincenzo A, et al. Aripiprazole overdose and transient 2:1 second degree atrioventricular block: only a coincidence? Curr Drug Saf. 2019;14:155-157.
6. Pacher P, Ungvari Z, Kecskemeti V, et al. Review of cardiovascular effects of fluoxetine, a selective serotonin reuptake inhibitor, compared to tricyclic antidepressants. Curr Med Chem. 1998;5:381-90.
7. Hemeryck A, Belpaire FM. Selective serotonin reuptake inhibitors and cytochrome P-450 mediated drug-drug interactions: an update. Curr Drub Metab. 2002;3:13-37.
8. Snarr BS, Phan SV, Garner A, et al. Symptomatic bradycardia with oral aripiprazole and oral ziprasidone. Ann Pharmacother. 2010;44:760-763.
9. Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-245.
Novel antiplatelet drug: Hope for efficacy without bleeding?
A new antiplatelet drug with a completely novel mechanism of action may hold the promise of delivering the holy grail – reducing cardiac events without increasing bleeding. That is the hope behind the new class of drugs directed against the platelet collagen glycoprotein VI (GPVI) receptor.
A phase 2 trial with the first agent in this class, known as revacept (advanceCOR), showed no increase in bleeding with the product when added to standard dual-antiplatelet therapy for patients with stable ischemic heart disease undergoing elective percutaneous coronary intervention (PCI), despite the drug’s being used at a dose that has been shown to increase platelet inhibition.
Unfortunately, there was no reduction in the primary clinical efficacy endpoint, a myocardial injury surrogate, but the authors pointed out that the overall event rate was low, and they were hopeful that future trials in a higher-risk population will show efficacy.
The ISAR PLASTER study was published online on March 31 in JAMA Cardiology.
“This new drug is targeting the collagen in the extracellular matrix of atherosclerotic plaque rather than the platelets themselves. So, in theory, this agent should not cause an increase in bleeding,” study author Steffen Massberg, DrMed, said in an interview.
Dr. Massberg explained that revacept targets the binding site for platelets on collagen that is exposed on rupture of atherosclerotic plaques and is a major trigger of platelet activation.
“In contrast to aspirin and P2Y12 inhibitors, which target all platelets, revacept only binds to sites where there is ruptured plaque. But the platelets themselves otherwise have normal function, so regular coagulation processes should be unaffected,” he commented.
“While collagen also has a role in the coagulation process, it is more involved in atherosclerotic plaque rupture, and in animal studies, revacept was effective in preventing clot formation in large arteries but only had a small effect on bleeding,” Dr. Massberg added.
In the JAMA Cardiology article, the authors further elaborated that, when collagen is exposed during atherosclerotic plaque rupture, it binds platelet GPVI, the major platelet collagen receptor.
“Glycoprotein VI in turn mediates local platelet recruitment, activation, and aggregation. Glycoprotein VI is an attractive antiplatelet target because GPVI-mediated platelet response plays a central role during myocardial infarction and stroke but is less relevant in physiological hemostasis,” they wrote.
The researchers describe revacept as a dimeric, soluble fusion protein composed of the extracellular domain of the GPVI receptor and the human Fc-fragment. It competes with endogenous platelet GPVI for binding to exposed collagen fibers and inhibits collagen-mediated platelet adhesion and aggregation selectively at the site of plaque rupture.
In addition, revacept blocks binding of von Willebrand factor to collagen and inhibits von Willebrand factor–mediated platelet activation, they reported.
“As a lesion-directed drug, revacept does not interfere with the function of circulating platelets beyond the atherosclerotic lesion,” the authors said.
In animal studies and a phase 1 clinical trial, the drug was shown to inhibit atherothrombosis but to have little effect on systemic hemostasis or bleeding.
The current ISAR-PLASTER trial is the first study of the use of the agent for patients with coronary heart disease.
For the study, 334 patients with stable ischemic heart disease undergoing elective PCI were randomly assigned to receive a single intravenous infusion of revacept 160 mg, revacept 80 mg, or placebo prior to the start of PCI in addition to standard antithrombotic therapy.
The safety endpoint was bleeding of type 2-5, per Bleeding Academic Research Consortium (BARC) criteria, at 30 days.
Results showed no significant differences in the primary efficacy endpoint (the composite of death or myocardial injury, defined as an increase in high-sensitivity cardiac troponin T [hsTnT] to at least five times the upper limit of normal within 48 hours from randomization) between the revacept and placebo groups. The primary efficacy endpoint occurred in 24.4% of the revacept 160-mg group, 25.0% of the revacept 80-mg group, and 23.3% of the placebo group.
The high dose of revacept was associated with a small but significant reduction of high-concentration collagen-induced platelet aggregation, but adenosine 5-diphosphate–induced aggregation was not affected.
Revacept did not increase bleeding. Bleeding of BARC type 2 or higher at 30 days occurred in 5.0% of the 160-mg group, 5.9% of the 80-mg group, and 8.6% of the placebo group.
Dr. Massberg pointed out that one possible explanation for the lack of difference in the efficacy outcome was that the patients enrolled in the study were at low risk.
“The rate of major adverse cardiovascular events was very low (2.5% at 30 days), and this was a low-risk population undergoing elective PCI,” he commented.
The authors also pointed out that the five-times increase in hsTnT endpoint used in the current study has little prognostic impact.
In addition, Dr. Massberg noted that, in the stable situation, myocardial injury is mostly triggered by cholesterol embolism during PCI and side-branch occlusion due to distal plaque embolization, problems that are unlikely to respond to inhibition of GPVI-collagen interaction by revacept.
He suggested that better results may be achieved in patients with acute coronary syndrome (ACS). “In ACS patients, the myocardial injury is caused by ongoing thrombotic cascades, where the collagen-platelet interaction plays a much larger role, so in theory, this drug should show a greater effect in an ACS population.”
The researchers are now planning a larger phase 3 study in that group.
“I am still optimistic. I still believe it could work,” Dr. Massberg said. “The major aim for this study was safety and dosing. There was no difference in bleeding, so safety was supported,” he added.
The ISAR-PLASTER study was funded by the German Center for Cardiovascular Research, Deutsches Herzzentrum Munchen, the Federal Ministry of Education and Research, and advanceCOR (the manufacturer of revacept). One of the coauthors of the study is a cofounder of advanceCor.
A version of this article first appeared on Medscape.com.
A new antiplatelet drug with a completely novel mechanism of action may hold the promise of delivering the holy grail – reducing cardiac events without increasing bleeding. That is the hope behind the new class of drugs directed against the platelet collagen glycoprotein VI (GPVI) receptor.
A phase 2 trial with the first agent in this class, known as revacept (advanceCOR), showed no increase in bleeding with the product when added to standard dual-antiplatelet therapy for patients with stable ischemic heart disease undergoing elective percutaneous coronary intervention (PCI), despite the drug’s being used at a dose that has been shown to increase platelet inhibition.
Unfortunately, there was no reduction in the primary clinical efficacy endpoint, a myocardial injury surrogate, but the authors pointed out that the overall event rate was low, and they were hopeful that future trials in a higher-risk population will show efficacy.
The ISAR PLASTER study was published online on March 31 in JAMA Cardiology.
“This new drug is targeting the collagen in the extracellular matrix of atherosclerotic plaque rather than the platelets themselves. So, in theory, this agent should not cause an increase in bleeding,” study author Steffen Massberg, DrMed, said in an interview.
Dr. Massberg explained that revacept targets the binding site for platelets on collagen that is exposed on rupture of atherosclerotic plaques and is a major trigger of platelet activation.
“In contrast to aspirin and P2Y12 inhibitors, which target all platelets, revacept only binds to sites where there is ruptured plaque. But the platelets themselves otherwise have normal function, so regular coagulation processes should be unaffected,” he commented.
“While collagen also has a role in the coagulation process, it is more involved in atherosclerotic plaque rupture, and in animal studies, revacept was effective in preventing clot formation in large arteries but only had a small effect on bleeding,” Dr. Massberg added.
In the JAMA Cardiology article, the authors further elaborated that, when collagen is exposed during atherosclerotic plaque rupture, it binds platelet GPVI, the major platelet collagen receptor.
“Glycoprotein VI in turn mediates local platelet recruitment, activation, and aggregation. Glycoprotein VI is an attractive antiplatelet target because GPVI-mediated platelet response plays a central role during myocardial infarction and stroke but is less relevant in physiological hemostasis,” they wrote.
The researchers describe revacept as a dimeric, soluble fusion protein composed of the extracellular domain of the GPVI receptor and the human Fc-fragment. It competes with endogenous platelet GPVI for binding to exposed collagen fibers and inhibits collagen-mediated platelet adhesion and aggregation selectively at the site of plaque rupture.
In addition, revacept blocks binding of von Willebrand factor to collagen and inhibits von Willebrand factor–mediated platelet activation, they reported.
“As a lesion-directed drug, revacept does not interfere with the function of circulating platelets beyond the atherosclerotic lesion,” the authors said.
In animal studies and a phase 1 clinical trial, the drug was shown to inhibit atherothrombosis but to have little effect on systemic hemostasis or bleeding.
The current ISAR-PLASTER trial is the first study of the use of the agent for patients with coronary heart disease.
For the study, 334 patients with stable ischemic heart disease undergoing elective PCI were randomly assigned to receive a single intravenous infusion of revacept 160 mg, revacept 80 mg, or placebo prior to the start of PCI in addition to standard antithrombotic therapy.
The safety endpoint was bleeding of type 2-5, per Bleeding Academic Research Consortium (BARC) criteria, at 30 days.
Results showed no significant differences in the primary efficacy endpoint (the composite of death or myocardial injury, defined as an increase in high-sensitivity cardiac troponin T [hsTnT] to at least five times the upper limit of normal within 48 hours from randomization) between the revacept and placebo groups. The primary efficacy endpoint occurred in 24.4% of the revacept 160-mg group, 25.0% of the revacept 80-mg group, and 23.3% of the placebo group.
The high dose of revacept was associated with a small but significant reduction of high-concentration collagen-induced platelet aggregation, but adenosine 5-diphosphate–induced aggregation was not affected.
Revacept did not increase bleeding. Bleeding of BARC type 2 or higher at 30 days occurred in 5.0% of the 160-mg group, 5.9% of the 80-mg group, and 8.6% of the placebo group.
Dr. Massberg pointed out that one possible explanation for the lack of difference in the efficacy outcome was that the patients enrolled in the study were at low risk.
“The rate of major adverse cardiovascular events was very low (2.5% at 30 days), and this was a low-risk population undergoing elective PCI,” he commented.
The authors also pointed out that the five-times increase in hsTnT endpoint used in the current study has little prognostic impact.
In addition, Dr. Massberg noted that, in the stable situation, myocardial injury is mostly triggered by cholesterol embolism during PCI and side-branch occlusion due to distal plaque embolization, problems that are unlikely to respond to inhibition of GPVI-collagen interaction by revacept.
He suggested that better results may be achieved in patients with acute coronary syndrome (ACS). “In ACS patients, the myocardial injury is caused by ongoing thrombotic cascades, where the collagen-platelet interaction plays a much larger role, so in theory, this drug should show a greater effect in an ACS population.”
The researchers are now planning a larger phase 3 study in that group.
“I am still optimistic. I still believe it could work,” Dr. Massberg said. “The major aim for this study was safety and dosing. There was no difference in bleeding, so safety was supported,” he added.
The ISAR-PLASTER study was funded by the German Center for Cardiovascular Research, Deutsches Herzzentrum Munchen, the Federal Ministry of Education and Research, and advanceCOR (the manufacturer of revacept). One of the coauthors of the study is a cofounder of advanceCor.
A version of this article first appeared on Medscape.com.
A new antiplatelet drug with a completely novel mechanism of action may hold the promise of delivering the holy grail – reducing cardiac events without increasing bleeding. That is the hope behind the new class of drugs directed against the platelet collagen glycoprotein VI (GPVI) receptor.
A phase 2 trial with the first agent in this class, known as revacept (advanceCOR), showed no increase in bleeding with the product when added to standard dual-antiplatelet therapy for patients with stable ischemic heart disease undergoing elective percutaneous coronary intervention (PCI), despite the drug’s being used at a dose that has been shown to increase platelet inhibition.
Unfortunately, there was no reduction in the primary clinical efficacy endpoint, a myocardial injury surrogate, but the authors pointed out that the overall event rate was low, and they were hopeful that future trials in a higher-risk population will show efficacy.
The ISAR PLASTER study was published online on March 31 in JAMA Cardiology.
“This new drug is targeting the collagen in the extracellular matrix of atherosclerotic plaque rather than the platelets themselves. So, in theory, this agent should not cause an increase in bleeding,” study author Steffen Massberg, DrMed, said in an interview.
Dr. Massberg explained that revacept targets the binding site for platelets on collagen that is exposed on rupture of atherosclerotic plaques and is a major trigger of platelet activation.
“In contrast to aspirin and P2Y12 inhibitors, which target all platelets, revacept only binds to sites where there is ruptured plaque. But the platelets themselves otherwise have normal function, so regular coagulation processes should be unaffected,” he commented.
“While collagen also has a role in the coagulation process, it is more involved in atherosclerotic plaque rupture, and in animal studies, revacept was effective in preventing clot formation in large arteries but only had a small effect on bleeding,” Dr. Massberg added.
In the JAMA Cardiology article, the authors further elaborated that, when collagen is exposed during atherosclerotic plaque rupture, it binds platelet GPVI, the major platelet collagen receptor.
“Glycoprotein VI in turn mediates local platelet recruitment, activation, and aggregation. Glycoprotein VI is an attractive antiplatelet target because GPVI-mediated platelet response plays a central role during myocardial infarction and stroke but is less relevant in physiological hemostasis,” they wrote.
The researchers describe revacept as a dimeric, soluble fusion protein composed of the extracellular domain of the GPVI receptor and the human Fc-fragment. It competes with endogenous platelet GPVI for binding to exposed collagen fibers and inhibits collagen-mediated platelet adhesion and aggregation selectively at the site of plaque rupture.
In addition, revacept blocks binding of von Willebrand factor to collagen and inhibits von Willebrand factor–mediated platelet activation, they reported.
“As a lesion-directed drug, revacept does not interfere with the function of circulating platelets beyond the atherosclerotic lesion,” the authors said.
In animal studies and a phase 1 clinical trial, the drug was shown to inhibit atherothrombosis but to have little effect on systemic hemostasis or bleeding.
The current ISAR-PLASTER trial is the first study of the use of the agent for patients with coronary heart disease.
For the study, 334 patients with stable ischemic heart disease undergoing elective PCI were randomly assigned to receive a single intravenous infusion of revacept 160 mg, revacept 80 mg, or placebo prior to the start of PCI in addition to standard antithrombotic therapy.
The safety endpoint was bleeding of type 2-5, per Bleeding Academic Research Consortium (BARC) criteria, at 30 days.
Results showed no significant differences in the primary efficacy endpoint (the composite of death or myocardial injury, defined as an increase in high-sensitivity cardiac troponin T [hsTnT] to at least five times the upper limit of normal within 48 hours from randomization) between the revacept and placebo groups. The primary efficacy endpoint occurred in 24.4% of the revacept 160-mg group, 25.0% of the revacept 80-mg group, and 23.3% of the placebo group.
The high dose of revacept was associated with a small but significant reduction of high-concentration collagen-induced platelet aggregation, but adenosine 5-diphosphate–induced aggregation was not affected.
Revacept did not increase bleeding. Bleeding of BARC type 2 or higher at 30 days occurred in 5.0% of the 160-mg group, 5.9% of the 80-mg group, and 8.6% of the placebo group.
Dr. Massberg pointed out that one possible explanation for the lack of difference in the efficacy outcome was that the patients enrolled in the study were at low risk.
“The rate of major adverse cardiovascular events was very low (2.5% at 30 days), and this was a low-risk population undergoing elective PCI,” he commented.
The authors also pointed out that the five-times increase in hsTnT endpoint used in the current study has little prognostic impact.
In addition, Dr. Massberg noted that, in the stable situation, myocardial injury is mostly triggered by cholesterol embolism during PCI and side-branch occlusion due to distal plaque embolization, problems that are unlikely to respond to inhibition of GPVI-collagen interaction by revacept.
He suggested that better results may be achieved in patients with acute coronary syndrome (ACS). “In ACS patients, the myocardial injury is caused by ongoing thrombotic cascades, where the collagen-platelet interaction plays a much larger role, so in theory, this drug should show a greater effect in an ACS population.”
The researchers are now planning a larger phase 3 study in that group.
“I am still optimistic. I still believe it could work,” Dr. Massberg said. “The major aim for this study was safety and dosing. There was no difference in bleeding, so safety was supported,” he added.
The ISAR-PLASTER study was funded by the German Center for Cardiovascular Research, Deutsches Herzzentrum Munchen, the Federal Ministry of Education and Research, and advanceCOR (the manufacturer of revacept). One of the coauthors of the study is a cofounder of advanceCor.
A version of this article first appeared on Medscape.com.