User login
ESC heart failure guideline to integrate bounty of new meds
Today there are so many evidence-based drug therapies for heart failure with reduced ejection fraction (HFrEF) that physicians treating HF patients almost don’t know what to do them.
It’s an exciting new age that way, but to many vexingly unclear how best to merge the shiny new options with mainstay regimens based on time-honored renin-angiotensin system (RAS) inhibitors and beta-blockers.
To impart some clarity, the authors of a new HF guideline document recently took center stage at the Heart Failure Association of the European Society of Cardiology (ESC-HFA) annual meeting to preview their updated recommendations, with novel twists based on recent major trials, for the new age of HF pharmacotherapeutics.
The guideline committee considered the evidence base that existed “up until the end of March of this year,” Theresa A. McDonagh, MD, King’s College London, said during the presentation. The document “is now finalized, it’s with the publishers, and it will be presented in full with simultaneous publication at the ESC meeting” that starts August 27.
It describes a game plan, already followed by some clinicians in practice without official guidance, for initiating drugs from each of four classes in virtually all patients with HFrEF.
New indicated drugs, new perspective for HFrEF
Three of the drug categories are old acquaintances. Among them are the RAS inhibitors, which include angiotensin-receptor/neprilysin inhibitors, beta-blockers, and the mineralocorticoid receptor antagonists. The latter drugs are gaining new respect after having been underplayed in HF prescribing despite longstanding evidence of efficacy.
Completing the quartet of first-line HFrEF drug classes is a recent arrival to the HF arena, the sodium-glucose cotransporter 2 inhibitors.
“We now have new data and a simplified treatment algorithm for heart failure with reduced ejection fraction based on the early administration of the four major classes of drugs,” said Marco Metra, MD, University of Brescia (Italy), previewing the medical-therapy portions of the new guideline at the ESC-HFA sessions, which launched virtually and live in Florence, Italy, on July 29.
The new game plan offers a simple answer to a once-common but complex question: How and in what order are the different drug classes initiated in patients with HFrEF? In the new document, the stated goal is to get them all on board expeditiously and safely, by any means possible.
The guideline writers did not specify a sequence, preferring to leave that decision to physicians, said Dr. Metra, who stated only two guiding principles. The first is to consider the patient’s unique circumstances. The order in which the drugs are introduced might vary, depending on, for example, whether the patient has low or high blood pressure or renal dysfunction.
Second, “it is very important that we try to give all four classes of drugs to the patient in the shortest time possible, because this saves lives,” he said.
That there is no recommendation on sequencing the drugs has led some to the wrong interpretation that all should be started at once, observed coauthor Javed Butler, MD, MPH, University of Mississippi, Jackson, as a panelist during the presentation. Far from it, he said. “The doctor with the patient in front of you can make the best decision. The idea here is to get all the therapies on as soon as possible, as safely as possible.”
“The order in which they are introduced is not really important,” agreed Vijay Chopra, MD, Max Super Specialty Hospital Saket, New Delhi, another coauthor on the panel. “The important thing is that at least some dose of all the four drugs needs to be introduced in the first 4-6 weeks, and then up-titrated.”
Other medical therapy can be more tailored, Dr. Metra noted, such as loop diuretics for patients with congestion, iron for those with iron deficiency, and other drugs depending on whether there is, for example, atrial fibrillation or coronary disease.
Adoption of emerging definitions
The document adopts the emerging characterization of HFrEF by a left ventricular ejection fraction (LVEF) up to 40%.
And it will leverage an expanding evidence base for medication in a segment of patients once said to have HF with preserved ejection fraction (HFpEF), who had therefore lacked specific, guideline-directed medical therapies. Now, patients with an LVEF of 41%-49% will be said to have HF with mildly reduced ejection fraction (HFmrEF), a tweak to the recently introduced HF with “mid-range” LVEF that is designed to assert its nature as something to treat. The new document’s HFmrEF recommendations come with various class and level-of-evidence ratings.
That leaves HFpEF to be characterized by an LVEF of 50% in combination with structural or functional abnormalities associated with LV diastolic dysfunction or raised LV filling pressures, including raised natriuretic peptide levels.
The definitions are consistent with those proposed internationally by the ESC-HFA, the Heart Failure Society of America, and other groups in a statement published in March.
Expanded HFrEF med landscape
Since the 2016 ESC guideline on HF therapy, Dr. McDonagh said, “there’s been no substantial change in the evidence for many of the classical drugs that we use in heart failure. However, we had a lot of new and exciting evidence to consider,” especially in support of the SGLT2 inhibitors as one of the core medications in HFrEF.
The new data came from two controlled trials in particular. In DAPA-HF, patients with HFrEF who were initially without diabetes and who went on dapagliflozin (Farxiga, AstraZeneca) showed a 27% drop in cardiovascular (CV) death or worsening-HF events over a median of 18 months.
“That was followed up with very concordant results with empagliflozin [Jardiance, Boehringer Ingelheim/Eli Lilly] in HFrEF in the EMPEROR-Reduced trial,” Dr. McDonagh said. In that trial, comparable patients who took empagliflozin showed a 25% drop in a primary endpoint similar to that in DAPA-HF over the median 16-month follow-up.
Other HFrEF recommendations are for selected patients. They include ivabradine, already in the guidelines, for patients in sinus rhythm with an elevated resting heart rate who can’t take beta-blockers for whatever reason. But, Dr. McDonagh noted, “we had some new classes of drugs to consider as well.”
In particular, the oral soluble guanylate-cyclase receptor stimulator vericiguat (Verquvo) emerged about a year ago from the VICTORIA trial as a modest success for patients with HFrEF and a previous HF hospitalization. In the trial with more than 5,000 patients, treatment with vericiguat atop standard drug and device therapy was followed by a significant 10% drop in risk for CV death or HF hospitalization.
Available now or likely to be available in the United States, the European Union, Japan, and other countries, vericiguat is recommended in the new guideline for VICTORIA-like patients who don’t adequately respond to other indicated medications.
Little for HFpEF as newly defined
“Almost nothing is new” in the guidelines for HFpEF, Dr. Metra said. The document recommends screening for and treatment of any underlying disorder and comorbidities, plus diuretics for any congestion. “That’s what we have to date.”
But that evidence base might soon change. The new HFpEF recommendations could possibly be up-staged at the ESC sessions by the August 27 scheduled presentation of EMPEROR-Preserved, a randomized test of empagliflozin in HFpEF and – it could be said – HFmrEF. The trial entered patients with chronic HF and an LVEF greater than 40%.
Eli Lilly and Boehringer Ingelheim offered the world a peek at the results, which suggest the SGLT2 inhibitor had a positive impact on the primary endpoint of CV death or HF hospitalization. They announced the cursory top-line outcomes in early July as part of its regulatory obligations, noting that the trial had “met” its primary endpoint.
But many unknowns remain, including the degree of benefit and whether it varied among subgroups, and especially whether outcomes were different for HFmrEF than for HFpEF.
Upgrades for familiar agents
Still, HFmrEF gets noteworthy attention in the document. “For the first time, we have recommendations for these patients,” Dr. Metra said. “We already knew that diuretics are indicated for the treatment of congestion. But now, ACE inhibitors, ARBs, beta-blockers, mineralocorticoid antagonists, as well as sacubitril/valsartan, may be considered to improve outcomes in these patients.” Their upgrades in the new guidelines were based on review of trials in the CHARM program and of TOPCAT and PARAGON-HF, among others, he said.

The new document also includes “treatment algorithms based on phenotypes”; that is, comorbidities and less common HF precipitants. For example, “assessment of iron status is now mandated in all patients with heart failure,” Dr. Metra said.
AFFIRM-HF is the key trial in this arena, with its more than 1,100 iron-deficient patients with LVEF less than 50% who had been recently hospitalized for HF. A year of treatment with ferric carboxymaltose (Ferinject/Injectafer, Vifor) led to a 26% drop in risk for HF hospitalization, but without affecting mortality.
For those who are iron deficient, Dr. Metra said, “ferric carboxymaltose intravenously should be considered not only in patients with low ejection fraction and outpatients, but also in patients recently hospitalized for acute heart failure.”
The SGLT2 inhibitors are recommended in HFrEF patients with type 2 diabetes. And treatment with tafamidis (Vyndaqel, Pfizer) in patients with genetic or wild-type transthyretin cardiac amyloidosis gets a class I recommendation based on survival gains seen in the ATTR-ACT trial.
Also recommended is a full CV assessment for patients with cancer who are on cardiotoxic agents or otherwise might be at risk for chemotherapy cardiotoxicity. “Beta-blockers and ACE inhibitors should be considered in those who develop left ventricular systolic dysfunction after anticancer therapy,” Dr. Metra said.
The ongoing pandemic made its mark on the document’s genesis, as it has with most everything else. “For better or worse, we were a ‘COVID guideline,’ ” Dr. McDonagh said. The writing committee consisted of “a large task force of 31 individuals, including two patients,” and there were “only two face-to-face meetings prior to the first wave of COVID hitting Europe.”
The committee voted on each of the recommendations, “and we had to have agreement of more than 75% of the task force to assign a class of recommendation or level of evidence,” she said. “I think we did the best we could in the circumstances. We had the benefit of many discussions over Zoom, and I think at the end of the day we have achieved a consensus.”
With such a large body of participants and the 75% threshold for agreement, “you end up with perhaps a conservative guideline. But that’s not a bad thing for clinical practice, for guidelines to be conservative,” Dr. McDonagh said. “They’re mainly concerned with looking at evidence and safety.”
A version of this article first appeared on Medscape.com.
Today there are so many evidence-based drug therapies for heart failure with reduced ejection fraction (HFrEF) that physicians treating HF patients almost don’t know what to do them.
It’s an exciting new age that way, but to many vexingly unclear how best to merge the shiny new options with mainstay regimens based on time-honored renin-angiotensin system (RAS) inhibitors and beta-blockers.
To impart some clarity, the authors of a new HF guideline document recently took center stage at the Heart Failure Association of the European Society of Cardiology (ESC-HFA) annual meeting to preview their updated recommendations, with novel twists based on recent major trials, for the new age of HF pharmacotherapeutics.
The guideline committee considered the evidence base that existed “up until the end of March of this year,” Theresa A. McDonagh, MD, King’s College London, said during the presentation. The document “is now finalized, it’s with the publishers, and it will be presented in full with simultaneous publication at the ESC meeting” that starts August 27.
It describes a game plan, already followed by some clinicians in practice without official guidance, for initiating drugs from each of four classes in virtually all patients with HFrEF.
New indicated drugs, new perspective for HFrEF
Three of the drug categories are old acquaintances. Among them are the RAS inhibitors, which include angiotensin-receptor/neprilysin inhibitors, beta-blockers, and the mineralocorticoid receptor antagonists. The latter drugs are gaining new respect after having been underplayed in HF prescribing despite longstanding evidence of efficacy.
Completing the quartet of first-line HFrEF drug classes is a recent arrival to the HF arena, the sodium-glucose cotransporter 2 inhibitors.
“We now have new data and a simplified treatment algorithm for heart failure with reduced ejection fraction based on the early administration of the four major classes of drugs,” said Marco Metra, MD, University of Brescia (Italy), previewing the medical-therapy portions of the new guideline at the ESC-HFA sessions, which launched virtually and live in Florence, Italy, on July 29.
The new game plan offers a simple answer to a once-common but complex question: How and in what order are the different drug classes initiated in patients with HFrEF? In the new document, the stated goal is to get them all on board expeditiously and safely, by any means possible.
The guideline writers did not specify a sequence, preferring to leave that decision to physicians, said Dr. Metra, who stated only two guiding principles. The first is to consider the patient’s unique circumstances. The order in which the drugs are introduced might vary, depending on, for example, whether the patient has low or high blood pressure or renal dysfunction.
Second, “it is very important that we try to give all four classes of drugs to the patient in the shortest time possible, because this saves lives,” he said.
That there is no recommendation on sequencing the drugs has led some to the wrong interpretation that all should be started at once, observed coauthor Javed Butler, MD, MPH, University of Mississippi, Jackson, as a panelist during the presentation. Far from it, he said. “The doctor with the patient in front of you can make the best decision. The idea here is to get all the therapies on as soon as possible, as safely as possible.”
“The order in which they are introduced is not really important,” agreed Vijay Chopra, MD, Max Super Specialty Hospital Saket, New Delhi, another coauthor on the panel. “The important thing is that at least some dose of all the four drugs needs to be introduced in the first 4-6 weeks, and then up-titrated.”
Other medical therapy can be more tailored, Dr. Metra noted, such as loop diuretics for patients with congestion, iron for those with iron deficiency, and other drugs depending on whether there is, for example, atrial fibrillation or coronary disease.
Adoption of emerging definitions
The document adopts the emerging characterization of HFrEF by a left ventricular ejection fraction (LVEF) up to 40%.
And it will leverage an expanding evidence base for medication in a segment of patients once said to have HF with preserved ejection fraction (HFpEF), who had therefore lacked specific, guideline-directed medical therapies. Now, patients with an LVEF of 41%-49% will be said to have HF with mildly reduced ejection fraction (HFmrEF), a tweak to the recently introduced HF with “mid-range” LVEF that is designed to assert its nature as something to treat. The new document’s HFmrEF recommendations come with various class and level-of-evidence ratings.
That leaves HFpEF to be characterized by an LVEF of 50% in combination with structural or functional abnormalities associated with LV diastolic dysfunction or raised LV filling pressures, including raised natriuretic peptide levels.
The definitions are consistent with those proposed internationally by the ESC-HFA, the Heart Failure Society of America, and other groups in a statement published in March.
Expanded HFrEF med landscape
Since the 2016 ESC guideline on HF therapy, Dr. McDonagh said, “there’s been no substantial change in the evidence for many of the classical drugs that we use in heart failure. However, we had a lot of new and exciting evidence to consider,” especially in support of the SGLT2 inhibitors as one of the core medications in HFrEF.
The new data came from two controlled trials in particular. In DAPA-HF, patients with HFrEF who were initially without diabetes and who went on dapagliflozin (Farxiga, AstraZeneca) showed a 27% drop in cardiovascular (CV) death or worsening-HF events over a median of 18 months.
“That was followed up with very concordant results with empagliflozin [Jardiance, Boehringer Ingelheim/Eli Lilly] in HFrEF in the EMPEROR-Reduced trial,” Dr. McDonagh said. In that trial, comparable patients who took empagliflozin showed a 25% drop in a primary endpoint similar to that in DAPA-HF over the median 16-month follow-up.
Other HFrEF recommendations are for selected patients. They include ivabradine, already in the guidelines, for patients in sinus rhythm with an elevated resting heart rate who can’t take beta-blockers for whatever reason. But, Dr. McDonagh noted, “we had some new classes of drugs to consider as well.”
In particular, the oral soluble guanylate-cyclase receptor stimulator vericiguat (Verquvo) emerged about a year ago from the VICTORIA trial as a modest success for patients with HFrEF and a previous HF hospitalization. In the trial with more than 5,000 patients, treatment with vericiguat atop standard drug and device therapy was followed by a significant 10% drop in risk for CV death or HF hospitalization.
Available now or likely to be available in the United States, the European Union, Japan, and other countries, vericiguat is recommended in the new guideline for VICTORIA-like patients who don’t adequately respond to other indicated medications.
Little for HFpEF as newly defined
“Almost nothing is new” in the guidelines for HFpEF, Dr. Metra said. The document recommends screening for and treatment of any underlying disorder and comorbidities, plus diuretics for any congestion. “That’s what we have to date.”
But that evidence base might soon change. The new HFpEF recommendations could possibly be up-staged at the ESC sessions by the August 27 scheduled presentation of EMPEROR-Preserved, a randomized test of empagliflozin in HFpEF and – it could be said – HFmrEF. The trial entered patients with chronic HF and an LVEF greater than 40%.
Eli Lilly and Boehringer Ingelheim offered the world a peek at the results, which suggest the SGLT2 inhibitor had a positive impact on the primary endpoint of CV death or HF hospitalization. They announced the cursory top-line outcomes in early July as part of its regulatory obligations, noting that the trial had “met” its primary endpoint.
But many unknowns remain, including the degree of benefit and whether it varied among subgroups, and especially whether outcomes were different for HFmrEF than for HFpEF.
Upgrades for familiar agents
Still, HFmrEF gets noteworthy attention in the document. “For the first time, we have recommendations for these patients,” Dr. Metra said. “We already knew that diuretics are indicated for the treatment of congestion. But now, ACE inhibitors, ARBs, beta-blockers, mineralocorticoid antagonists, as well as sacubitril/valsartan, may be considered to improve outcomes in these patients.” Their upgrades in the new guidelines were based on review of trials in the CHARM program and of TOPCAT and PARAGON-HF, among others, he said.

The new document also includes “treatment algorithms based on phenotypes”; that is, comorbidities and less common HF precipitants. For example, “assessment of iron status is now mandated in all patients with heart failure,” Dr. Metra said.
AFFIRM-HF is the key trial in this arena, with its more than 1,100 iron-deficient patients with LVEF less than 50% who had been recently hospitalized for HF. A year of treatment with ferric carboxymaltose (Ferinject/Injectafer, Vifor) led to a 26% drop in risk for HF hospitalization, but without affecting mortality.
For those who are iron deficient, Dr. Metra said, “ferric carboxymaltose intravenously should be considered not only in patients with low ejection fraction and outpatients, but also in patients recently hospitalized for acute heart failure.”
The SGLT2 inhibitors are recommended in HFrEF patients with type 2 diabetes. And treatment with tafamidis (Vyndaqel, Pfizer) in patients with genetic or wild-type transthyretin cardiac amyloidosis gets a class I recommendation based on survival gains seen in the ATTR-ACT trial.
Also recommended is a full CV assessment for patients with cancer who are on cardiotoxic agents or otherwise might be at risk for chemotherapy cardiotoxicity. “Beta-blockers and ACE inhibitors should be considered in those who develop left ventricular systolic dysfunction after anticancer therapy,” Dr. Metra said.
The ongoing pandemic made its mark on the document’s genesis, as it has with most everything else. “For better or worse, we were a ‘COVID guideline,’ ” Dr. McDonagh said. The writing committee consisted of “a large task force of 31 individuals, including two patients,” and there were “only two face-to-face meetings prior to the first wave of COVID hitting Europe.”
The committee voted on each of the recommendations, “and we had to have agreement of more than 75% of the task force to assign a class of recommendation or level of evidence,” she said. “I think we did the best we could in the circumstances. We had the benefit of many discussions over Zoom, and I think at the end of the day we have achieved a consensus.”
With such a large body of participants and the 75% threshold for agreement, “you end up with perhaps a conservative guideline. But that’s not a bad thing for clinical practice, for guidelines to be conservative,” Dr. McDonagh said. “They’re mainly concerned with looking at evidence and safety.”
A version of this article first appeared on Medscape.com.
Today there are so many evidence-based drug therapies for heart failure with reduced ejection fraction (HFrEF) that physicians treating HF patients almost don’t know what to do them.
It’s an exciting new age that way, but to many vexingly unclear how best to merge the shiny new options with mainstay regimens based on time-honored renin-angiotensin system (RAS) inhibitors and beta-blockers.
To impart some clarity, the authors of a new HF guideline document recently took center stage at the Heart Failure Association of the European Society of Cardiology (ESC-HFA) annual meeting to preview their updated recommendations, with novel twists based on recent major trials, for the new age of HF pharmacotherapeutics.
The guideline committee considered the evidence base that existed “up until the end of March of this year,” Theresa A. McDonagh, MD, King’s College London, said during the presentation. The document “is now finalized, it’s with the publishers, and it will be presented in full with simultaneous publication at the ESC meeting” that starts August 27.
It describes a game plan, already followed by some clinicians in practice without official guidance, for initiating drugs from each of four classes in virtually all patients with HFrEF.
New indicated drugs, new perspective for HFrEF
Three of the drug categories are old acquaintances. Among them are the RAS inhibitors, which include angiotensin-receptor/neprilysin inhibitors, beta-blockers, and the mineralocorticoid receptor antagonists. The latter drugs are gaining new respect after having been underplayed in HF prescribing despite longstanding evidence of efficacy.
Completing the quartet of first-line HFrEF drug classes is a recent arrival to the HF arena, the sodium-glucose cotransporter 2 inhibitors.
“We now have new data and a simplified treatment algorithm for heart failure with reduced ejection fraction based on the early administration of the four major classes of drugs,” said Marco Metra, MD, University of Brescia (Italy), previewing the medical-therapy portions of the new guideline at the ESC-HFA sessions, which launched virtually and live in Florence, Italy, on July 29.
The new game plan offers a simple answer to a once-common but complex question: How and in what order are the different drug classes initiated in patients with HFrEF? In the new document, the stated goal is to get them all on board expeditiously and safely, by any means possible.
The guideline writers did not specify a sequence, preferring to leave that decision to physicians, said Dr. Metra, who stated only two guiding principles. The first is to consider the patient’s unique circumstances. The order in which the drugs are introduced might vary, depending on, for example, whether the patient has low or high blood pressure or renal dysfunction.
Second, “it is very important that we try to give all four classes of drugs to the patient in the shortest time possible, because this saves lives,” he said.
That there is no recommendation on sequencing the drugs has led some to the wrong interpretation that all should be started at once, observed coauthor Javed Butler, MD, MPH, University of Mississippi, Jackson, as a panelist during the presentation. Far from it, he said. “The doctor with the patient in front of you can make the best decision. The idea here is to get all the therapies on as soon as possible, as safely as possible.”
“The order in which they are introduced is not really important,” agreed Vijay Chopra, MD, Max Super Specialty Hospital Saket, New Delhi, another coauthor on the panel. “The important thing is that at least some dose of all the four drugs needs to be introduced in the first 4-6 weeks, and then up-titrated.”
Other medical therapy can be more tailored, Dr. Metra noted, such as loop diuretics for patients with congestion, iron for those with iron deficiency, and other drugs depending on whether there is, for example, atrial fibrillation or coronary disease.
Adoption of emerging definitions
The document adopts the emerging characterization of HFrEF by a left ventricular ejection fraction (LVEF) up to 40%.
And it will leverage an expanding evidence base for medication in a segment of patients once said to have HF with preserved ejection fraction (HFpEF), who had therefore lacked specific, guideline-directed medical therapies. Now, patients with an LVEF of 41%-49% will be said to have HF with mildly reduced ejection fraction (HFmrEF), a tweak to the recently introduced HF with “mid-range” LVEF that is designed to assert its nature as something to treat. The new document’s HFmrEF recommendations come with various class and level-of-evidence ratings.
That leaves HFpEF to be characterized by an LVEF of 50% in combination with structural or functional abnormalities associated with LV diastolic dysfunction or raised LV filling pressures, including raised natriuretic peptide levels.
The definitions are consistent with those proposed internationally by the ESC-HFA, the Heart Failure Society of America, and other groups in a statement published in March.
Expanded HFrEF med landscape
Since the 2016 ESC guideline on HF therapy, Dr. McDonagh said, “there’s been no substantial change in the evidence for many of the classical drugs that we use in heart failure. However, we had a lot of new and exciting evidence to consider,” especially in support of the SGLT2 inhibitors as one of the core medications in HFrEF.
The new data came from two controlled trials in particular. In DAPA-HF, patients with HFrEF who were initially without diabetes and who went on dapagliflozin (Farxiga, AstraZeneca) showed a 27% drop in cardiovascular (CV) death or worsening-HF events over a median of 18 months.
“That was followed up with very concordant results with empagliflozin [Jardiance, Boehringer Ingelheim/Eli Lilly] in HFrEF in the EMPEROR-Reduced trial,” Dr. McDonagh said. In that trial, comparable patients who took empagliflozin showed a 25% drop in a primary endpoint similar to that in DAPA-HF over the median 16-month follow-up.
Other HFrEF recommendations are for selected patients. They include ivabradine, already in the guidelines, for patients in sinus rhythm with an elevated resting heart rate who can’t take beta-blockers for whatever reason. But, Dr. McDonagh noted, “we had some new classes of drugs to consider as well.”
In particular, the oral soluble guanylate-cyclase receptor stimulator vericiguat (Verquvo) emerged about a year ago from the VICTORIA trial as a modest success for patients with HFrEF and a previous HF hospitalization. In the trial with more than 5,000 patients, treatment with vericiguat atop standard drug and device therapy was followed by a significant 10% drop in risk for CV death or HF hospitalization.
Available now or likely to be available in the United States, the European Union, Japan, and other countries, vericiguat is recommended in the new guideline for VICTORIA-like patients who don’t adequately respond to other indicated medications.
Little for HFpEF as newly defined
“Almost nothing is new” in the guidelines for HFpEF, Dr. Metra said. The document recommends screening for and treatment of any underlying disorder and comorbidities, plus diuretics for any congestion. “That’s what we have to date.”
But that evidence base might soon change. The new HFpEF recommendations could possibly be up-staged at the ESC sessions by the August 27 scheduled presentation of EMPEROR-Preserved, a randomized test of empagliflozin in HFpEF and – it could be said – HFmrEF. The trial entered patients with chronic HF and an LVEF greater than 40%.
Eli Lilly and Boehringer Ingelheim offered the world a peek at the results, which suggest the SGLT2 inhibitor had a positive impact on the primary endpoint of CV death or HF hospitalization. They announced the cursory top-line outcomes in early July as part of its regulatory obligations, noting that the trial had “met” its primary endpoint.
But many unknowns remain, including the degree of benefit and whether it varied among subgroups, and especially whether outcomes were different for HFmrEF than for HFpEF.
Upgrades for familiar agents
Still, HFmrEF gets noteworthy attention in the document. “For the first time, we have recommendations for these patients,” Dr. Metra said. “We already knew that diuretics are indicated for the treatment of congestion. But now, ACE inhibitors, ARBs, beta-blockers, mineralocorticoid antagonists, as well as sacubitril/valsartan, may be considered to improve outcomes in these patients.” Their upgrades in the new guidelines were based on review of trials in the CHARM program and of TOPCAT and PARAGON-HF, among others, he said.

The new document also includes “treatment algorithms based on phenotypes”; that is, comorbidities and less common HF precipitants. For example, “assessment of iron status is now mandated in all patients with heart failure,” Dr. Metra said.
AFFIRM-HF is the key trial in this arena, with its more than 1,100 iron-deficient patients with LVEF less than 50% who had been recently hospitalized for HF. A year of treatment with ferric carboxymaltose (Ferinject/Injectafer, Vifor) led to a 26% drop in risk for HF hospitalization, but without affecting mortality.
For those who are iron deficient, Dr. Metra said, “ferric carboxymaltose intravenously should be considered not only in patients with low ejection fraction and outpatients, but also in patients recently hospitalized for acute heart failure.”
The SGLT2 inhibitors are recommended in HFrEF patients with type 2 diabetes. And treatment with tafamidis (Vyndaqel, Pfizer) in patients with genetic or wild-type transthyretin cardiac amyloidosis gets a class I recommendation based on survival gains seen in the ATTR-ACT trial.
Also recommended is a full CV assessment for patients with cancer who are on cardiotoxic agents or otherwise might be at risk for chemotherapy cardiotoxicity. “Beta-blockers and ACE inhibitors should be considered in those who develop left ventricular systolic dysfunction after anticancer therapy,” Dr. Metra said.
The ongoing pandemic made its mark on the document’s genesis, as it has with most everything else. “For better or worse, we were a ‘COVID guideline,’ ” Dr. McDonagh said. The writing committee consisted of “a large task force of 31 individuals, including two patients,” and there were “only two face-to-face meetings prior to the first wave of COVID hitting Europe.”
The committee voted on each of the recommendations, “and we had to have agreement of more than 75% of the task force to assign a class of recommendation or level of evidence,” she said. “I think we did the best we could in the circumstances. We had the benefit of many discussions over Zoom, and I think at the end of the day we have achieved a consensus.”
With such a large body of participants and the 75% threshold for agreement, “you end up with perhaps a conservative guideline. But that’s not a bad thing for clinical practice, for guidelines to be conservative,” Dr. McDonagh said. “They’re mainly concerned with looking at evidence and safety.”
A version of this article first appeared on Medscape.com.
ACC issues decision pathway for hypertriglyceridemia management
A new decision pathway for the management of hypertriglyceridemia, prompted by a large and growing body of evidence that elevated triglycerides to a targetable risk factor for atherosclerotic cardiovascular disease (ASCVD), has been issued by the American College of Cardiology.
According to the chairman of the writing committee, Salim S. Virani, MD, PhD, the recommendations amplify and update more than alter the hypertriglyceridemia treatment recommendations in the 2018 joint multisociety blood cholesterol guidelines issued in 2018.
This decision pathway, however, is focused on triglycerides alone.
“The previous guidelines included a section on strategies for addressing hypertriglyceridemia to reduce ASCVD risk, but this new decision pathway builds on the recommendations with more details and with additional information,” explained Dr. Virani, professor of medicine in the section of cardiovascular research, Baylor College of Medicine, Houston.
Within this newly published document, the definitions of hypertriglyceridemia and rationale for treatment are followed by detailed algorithms for four specific patient groups with varying triglyceride levels:
- Adults with ASCVD.
- Adults at least 40 years of age with diabetes but no ASCVD.
- Adults at least 20 years of age with no ASCVD or diabetes.
- Adults at least 20 years of age with severe hypertriglyceridemia.
“In the design of these algorithms, we made an active effort to make them suitable for use by primary care physicians as well as specialists,” said Dr. Virani. Despite “lots of boxes and arrows,” the flowcharts for each of these patient groups permit clinicians to follow the decision pathway without having to reread the text.
The common emphasis in all four algorithms is to begin by evaluating patients for secondary causes of hypertriglyceridemia, such as multifactorial chylomicronemia syndrome and other diseases associated with elevated triglycerides. The next steps, also common to all algorithms, are to optimize diet and lifestyle changes that lower triglycerides, optimize glycemic control, and optimize statin therapy.
“Although commonly recognized for their impact on LDL-C, statins also provide a 10%-30% dose-dependent reduction in triglycerides in patients with elevated levels,” the guidelines state. Statins are considered a fundamental step to secondary prevention of ASCVD regardless of triglyceride levels.
Once treatable causes or contributors to hypertriglyceridemia have been addressed, lifestyle interventions and statin therapy have been optimized, pharmacologic therapy directed specifically at control of hypertriglyceridemia “can be considered” in those at highest risk of ASCVD events, but Dr. Virani explained that this is never an early or first step in control of elevated triglycerides.
“The entire working group agreed that lifestyle interventions should be highlighted front and center before considering any other intervention,” Dr. Virani explained.
Pharmacologic therapy for hypertriglyceridemia is not ignored. Prescription omega-3 fatty acid products are preferred over nonprescription dietary supplements, which may vary in quality and purity. But these products, rather than a standalone solution, are best applied within the context of efforts to improve diet, lower body weight, and increase physical activity.
Several trials have associated ethyl ester and carboxylic acid preparations with meaningful reductions in triglycerides, but these drugs, including icosapent ethyl (IPE), are not without adverse events. In the pivotal REDUCE-IT trial, IPE was linked with an increased risk of atrial fibrillation relative to placebo.
IPE is “the best option” and the only therapy with an indication for reduction in ASCVD risk, according to Dr. Virani, but he explained that safety concerns led the authors of the new decision pathway to employ cautious language in regard to its use, suggesting that it be “considered” in high-risk patients after other methods of lowering triglycerides have been optimized.
In the algorithm for the four different risk groups, the decision pathways follow stratifications for different levels of hypertriglyceridemia (defined under fasting and nonfasting conditions) and also for specific levels of LDL cholesterol. ASCVD risk assessment is also a factor in determining the next steps along the decision pathway.
According to Michael Davidson, MD, director of the lipid clinic at the University of Chicago, the emphasis on lifestyle changes for hypertriglyceridemia and the prudent language in regard to pharmacologic therapy is appropriate.
“In light of the controversies regarding the REDUCE-IT trial, the writing committee has done a nice job with providing useful guidance regarding the utilization of icosapent ethyl in higher risk patients,” Dr. Davidson said.
Calling the ACC decision pathway “a welcome enhancement of the 2018 ACC/AHA cholesterol guidelines,” Dr. Davidson praised the way in which the limitations of the evidence regarding pharmacologic therapies were explained.
“Most importantly, this decision pathway helps clinicians appreciate that hypertriglyceridemia can be best managed with lifestyle changes and addressing potential secondary causes,” Dr. Davidson said.
Dr. Virani reports no potential conflicts of interest. Dr. Davidson reports financial relationships with multiple pharmaceutical companies including those making or pursuing therapies for control of hypertriglyceridemia.
A new decision pathway for the management of hypertriglyceridemia, prompted by a large and growing body of evidence that elevated triglycerides to a targetable risk factor for atherosclerotic cardiovascular disease (ASCVD), has been issued by the American College of Cardiology.
According to the chairman of the writing committee, Salim S. Virani, MD, PhD, the recommendations amplify and update more than alter the hypertriglyceridemia treatment recommendations in the 2018 joint multisociety blood cholesterol guidelines issued in 2018.
This decision pathway, however, is focused on triglycerides alone.
“The previous guidelines included a section on strategies for addressing hypertriglyceridemia to reduce ASCVD risk, but this new decision pathway builds on the recommendations with more details and with additional information,” explained Dr. Virani, professor of medicine in the section of cardiovascular research, Baylor College of Medicine, Houston.
Within this newly published document, the definitions of hypertriglyceridemia and rationale for treatment are followed by detailed algorithms for four specific patient groups with varying triglyceride levels:
- Adults with ASCVD.
- Adults at least 40 years of age with diabetes but no ASCVD.
- Adults at least 20 years of age with no ASCVD or diabetes.
- Adults at least 20 years of age with severe hypertriglyceridemia.
“In the design of these algorithms, we made an active effort to make them suitable for use by primary care physicians as well as specialists,” said Dr. Virani. Despite “lots of boxes and arrows,” the flowcharts for each of these patient groups permit clinicians to follow the decision pathway without having to reread the text.
The common emphasis in all four algorithms is to begin by evaluating patients for secondary causes of hypertriglyceridemia, such as multifactorial chylomicronemia syndrome and other diseases associated with elevated triglycerides. The next steps, also common to all algorithms, are to optimize diet and lifestyle changes that lower triglycerides, optimize glycemic control, and optimize statin therapy.
“Although commonly recognized for their impact on LDL-C, statins also provide a 10%-30% dose-dependent reduction in triglycerides in patients with elevated levels,” the guidelines state. Statins are considered a fundamental step to secondary prevention of ASCVD regardless of triglyceride levels.
Once treatable causes or contributors to hypertriglyceridemia have been addressed, lifestyle interventions and statin therapy have been optimized, pharmacologic therapy directed specifically at control of hypertriglyceridemia “can be considered” in those at highest risk of ASCVD events, but Dr. Virani explained that this is never an early or first step in control of elevated triglycerides.
“The entire working group agreed that lifestyle interventions should be highlighted front and center before considering any other intervention,” Dr. Virani explained.
Pharmacologic therapy for hypertriglyceridemia is not ignored. Prescription omega-3 fatty acid products are preferred over nonprescription dietary supplements, which may vary in quality and purity. But these products, rather than a standalone solution, are best applied within the context of efforts to improve diet, lower body weight, and increase physical activity.
Several trials have associated ethyl ester and carboxylic acid preparations with meaningful reductions in triglycerides, but these drugs, including icosapent ethyl (IPE), are not without adverse events. In the pivotal REDUCE-IT trial, IPE was linked with an increased risk of atrial fibrillation relative to placebo.
IPE is “the best option” and the only therapy with an indication for reduction in ASCVD risk, according to Dr. Virani, but he explained that safety concerns led the authors of the new decision pathway to employ cautious language in regard to its use, suggesting that it be “considered” in high-risk patients after other methods of lowering triglycerides have been optimized.
In the algorithm for the four different risk groups, the decision pathways follow stratifications for different levels of hypertriglyceridemia (defined under fasting and nonfasting conditions) and also for specific levels of LDL cholesterol. ASCVD risk assessment is also a factor in determining the next steps along the decision pathway.
According to Michael Davidson, MD, director of the lipid clinic at the University of Chicago, the emphasis on lifestyle changes for hypertriglyceridemia and the prudent language in regard to pharmacologic therapy is appropriate.
“In light of the controversies regarding the REDUCE-IT trial, the writing committee has done a nice job with providing useful guidance regarding the utilization of icosapent ethyl in higher risk patients,” Dr. Davidson said.
Calling the ACC decision pathway “a welcome enhancement of the 2018 ACC/AHA cholesterol guidelines,” Dr. Davidson praised the way in which the limitations of the evidence regarding pharmacologic therapies were explained.
“Most importantly, this decision pathway helps clinicians appreciate that hypertriglyceridemia can be best managed with lifestyle changes and addressing potential secondary causes,” Dr. Davidson said.
Dr. Virani reports no potential conflicts of interest. Dr. Davidson reports financial relationships with multiple pharmaceutical companies including those making or pursuing therapies for control of hypertriglyceridemia.
A new decision pathway for the management of hypertriglyceridemia, prompted by a large and growing body of evidence that elevated triglycerides to a targetable risk factor for atherosclerotic cardiovascular disease (ASCVD), has been issued by the American College of Cardiology.
According to the chairman of the writing committee, Salim S. Virani, MD, PhD, the recommendations amplify and update more than alter the hypertriglyceridemia treatment recommendations in the 2018 joint multisociety blood cholesterol guidelines issued in 2018.
This decision pathway, however, is focused on triglycerides alone.
“The previous guidelines included a section on strategies for addressing hypertriglyceridemia to reduce ASCVD risk, but this new decision pathway builds on the recommendations with more details and with additional information,” explained Dr. Virani, professor of medicine in the section of cardiovascular research, Baylor College of Medicine, Houston.
Within this newly published document, the definitions of hypertriglyceridemia and rationale for treatment are followed by detailed algorithms for four specific patient groups with varying triglyceride levels:
- Adults with ASCVD.
- Adults at least 40 years of age with diabetes but no ASCVD.
- Adults at least 20 years of age with no ASCVD or diabetes.
- Adults at least 20 years of age with severe hypertriglyceridemia.
“In the design of these algorithms, we made an active effort to make them suitable for use by primary care physicians as well as specialists,” said Dr. Virani. Despite “lots of boxes and arrows,” the flowcharts for each of these patient groups permit clinicians to follow the decision pathway without having to reread the text.
The common emphasis in all four algorithms is to begin by evaluating patients for secondary causes of hypertriglyceridemia, such as multifactorial chylomicronemia syndrome and other diseases associated with elevated triglycerides. The next steps, also common to all algorithms, are to optimize diet and lifestyle changes that lower triglycerides, optimize glycemic control, and optimize statin therapy.
“Although commonly recognized for their impact on LDL-C, statins also provide a 10%-30% dose-dependent reduction in triglycerides in patients with elevated levels,” the guidelines state. Statins are considered a fundamental step to secondary prevention of ASCVD regardless of triglyceride levels.
Once treatable causes or contributors to hypertriglyceridemia have been addressed, lifestyle interventions and statin therapy have been optimized, pharmacologic therapy directed specifically at control of hypertriglyceridemia “can be considered” in those at highest risk of ASCVD events, but Dr. Virani explained that this is never an early or first step in control of elevated triglycerides.
“The entire working group agreed that lifestyle interventions should be highlighted front and center before considering any other intervention,” Dr. Virani explained.
Pharmacologic therapy for hypertriglyceridemia is not ignored. Prescription omega-3 fatty acid products are preferred over nonprescription dietary supplements, which may vary in quality and purity. But these products, rather than a standalone solution, are best applied within the context of efforts to improve diet, lower body weight, and increase physical activity.
Several trials have associated ethyl ester and carboxylic acid preparations with meaningful reductions in triglycerides, but these drugs, including icosapent ethyl (IPE), are not without adverse events. In the pivotal REDUCE-IT trial, IPE was linked with an increased risk of atrial fibrillation relative to placebo.
IPE is “the best option” and the only therapy with an indication for reduction in ASCVD risk, according to Dr. Virani, but he explained that safety concerns led the authors of the new decision pathway to employ cautious language in regard to its use, suggesting that it be “considered” in high-risk patients after other methods of lowering triglycerides have been optimized.
In the algorithm for the four different risk groups, the decision pathways follow stratifications for different levels of hypertriglyceridemia (defined under fasting and nonfasting conditions) and also for specific levels of LDL cholesterol. ASCVD risk assessment is also a factor in determining the next steps along the decision pathway.
According to Michael Davidson, MD, director of the lipid clinic at the University of Chicago, the emphasis on lifestyle changes for hypertriglyceridemia and the prudent language in regard to pharmacologic therapy is appropriate.
“In light of the controversies regarding the REDUCE-IT trial, the writing committee has done a nice job with providing useful guidance regarding the utilization of icosapent ethyl in higher risk patients,” Dr. Davidson said.
Calling the ACC decision pathway “a welcome enhancement of the 2018 ACC/AHA cholesterol guidelines,” Dr. Davidson praised the way in which the limitations of the evidence regarding pharmacologic therapies were explained.
“Most importantly, this decision pathway helps clinicians appreciate that hypertriglyceridemia can be best managed with lifestyle changes and addressing potential secondary causes,” Dr. Davidson said.
Dr. Virani reports no potential conflicts of interest. Dr. Davidson reports financial relationships with multiple pharmaceutical companies including those making or pursuing therapies for control of hypertriglyceridemia.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
ARBs equal ACE inhibitors for hypertension, and better tolerated
In the largest comparison of angiotensin receptor blockers (ARBs) and ACE inhibitors to date, a study of nearly 2.3 million patients starting the drugs as monotherapy shows no significant differences between the two in the long-term prevention of hypertension-related cardiovascular events.
However, side effects were notably lower with ARBs.
“This is a very large, well-executed observational study that confirms that ARBs appear to have fewer side effects than ACE inhibitors, and no unexpected ARB side effects were detected,” senior author George Hripcsak, MD, professor and chair of biomedical informatics at Columbia University, New York, told this news organization.
“Despite being equally guideline-recommended first-line therapies for hypertension, these results support preferentially starting ARBs rather than ACE inhibitors when initiating treatment for hypertension for physicians and patients considering renin-angiotensin system (RAS) inhibition,” the authors added in the study, published online July 26, 2021, in the journal Hypertension.
They noted that both drug classes have been on the market a long time, with proven efficacy in hypertension and “a wide availability of inexpensive generic forms.”
They also stressed that their findings only apply to patients with hypertension for whom a RAS inhibitor would be the best choice of therapy.
Commenting on the research, George Bakris, MD, of the American Heart Association’s Comprehensive Hypertension Center at the University of Chicago, said the findings were consistent with his experience in prescribing as well as researching the two drug classes.
“I have been in practice for over 30 years and studied both classes, including head-to-head prospective trials to assess blood pressure, and found in many cases better blood pressure lowering by some ARBs and always better tolerability,” he told this news organization. “I think this study confirms and extends my thoughts between the two classes of blood pressure–lowering agents.”
Head-to-head comparisons of ACE inhibitors and ARBs limited to date
ACE inhibitors and ARBs each have extensive evidence supporting their roles as first-line medications in the treatment of hypertension, and each have the strongest recommendations in international guidelines.
However, ACE inhibitors are prescribed more commonly than ARBs as the first-line drug for lowering blood pressure, and head-to-head comparisons of the two are limited, with conflicting results.
For the study, Dr. Hripcsak and colleagues evaluated data on almost 3 million patients starting monotherapy with an ACE inhibitor or ARB for the first time between 1996 and 2018 in the United States, Germany, and South Korea, who had no history of heart disease or stroke.
They identified a total of 2,297,881 patients initiating ACE inhibitors and 673,938 starting ARBs. Among new users of ACE inhibitors, most received lisinopril (80%), followed by ramipril and enalapril, while most patients prescribed ARBs received losartan (45%), followed by valsartan and olmesartan.
With follow-up times ranging from about 4 months to more than 18 months, the data show no statistically significant differences between ACE inhibitors versus ARBs in the primary outcomes of acute myocardial infarction (hazard ratio, 1.11), heart failure (HR, 1.03), stroke (HR, 1.07), or composite cardiovascular events (HR, 1.06).
For secondary and safety outcomes, including an analysis of 51 possible side effects, ACE inhibitors, compared with ARBs, were associated with a significantly higher risk of angioedema (HR, 3.31; P < .01), cough (HR, 1.32; P < .01), acute pancreatitis (HR, 1.32; P = .02), gastrointestinal bleeding (HR, 1.18; P = .04), and abnormal weight loss (HR, 1.18; P = .04).
While the link between ACE inhibitors and pancreatitis has been previously reported, the association with GI bleeding may be a novel finding, with no prior studies comparing those effects in the two drug classes, the authors noted.
Despite most patients taking just a couple of drugs in either class, Dr. Hripcsak said, “we don’t expect that other drugs from those classes will have fewer differences. It is possible, of course, but that is not our expectation.”
Results only applicable to those starting therapy with RAS inhibitors
First author RuiJun Chen, MD, added that, importantly, the results may not apply to patients switching therapies or adding on therapy, “such as for the patient whose hypertension is not effectively controlled with one drug and requires the addition of a second medication,” he said in an interview.
“Also, the suggestion of preferentially prescribing ARBs only applies to those patients and providers intending to control blood pressure through RAS inhibition,” said Dr. Chen, an assistant professor in translational data science and informatics at Geisinger Medical Center in Danville, Pa., who was a National Library of Medicine postdoctoral fellow at Columbia University at the time of the study.
Hence, he stressed the results do not extend to other classes of recommended first-line blood pressure medications.
“Essentially, since this is an ACE inhibitor versus ARB study, we would not claim that ARBs are preferred over all other types of hypertension medications which were not studied here,” the researchers emphasize.
In addition to ARBs and ACE inhibitors, other medications recommended by the AHA/American College of Cardiology in the 2017 “Guideline for the Prevention, Detection, Evaluation and Management of High Blood Pressure in Adults” for the primary treatment of hypertension include thiazide diuretics and calcium channel blockers.
The study received support from the National Library of Medicine and the National Institute of Allergy and Infectious Diseases of the National Institutes of Health; the National Science Foundation; and the Ministries of Health & Welfare and of Trade, Industry & Energy of the Republic of Korea. Dr. Hripcsak reported receiving grants from the National Library of Medicine during the study and grants from Janssen Research outside the submitted work. Dr. Bakris reported being a consultant for Merck, KBP Biosciences, and Ionis.
A version of this article first appeared on Medscape.com.
In the largest comparison of angiotensin receptor blockers (ARBs) and ACE inhibitors to date, a study of nearly 2.3 million patients starting the drugs as monotherapy shows no significant differences between the two in the long-term prevention of hypertension-related cardiovascular events.
However, side effects were notably lower with ARBs.
“This is a very large, well-executed observational study that confirms that ARBs appear to have fewer side effects than ACE inhibitors, and no unexpected ARB side effects were detected,” senior author George Hripcsak, MD, professor and chair of biomedical informatics at Columbia University, New York, told this news organization.
“Despite being equally guideline-recommended first-line therapies for hypertension, these results support preferentially starting ARBs rather than ACE inhibitors when initiating treatment for hypertension for physicians and patients considering renin-angiotensin system (RAS) inhibition,” the authors added in the study, published online July 26, 2021, in the journal Hypertension.
They noted that both drug classes have been on the market a long time, with proven efficacy in hypertension and “a wide availability of inexpensive generic forms.”
They also stressed that their findings only apply to patients with hypertension for whom a RAS inhibitor would be the best choice of therapy.
Commenting on the research, George Bakris, MD, of the American Heart Association’s Comprehensive Hypertension Center at the University of Chicago, said the findings were consistent with his experience in prescribing as well as researching the two drug classes.
“I have been in practice for over 30 years and studied both classes, including head-to-head prospective trials to assess blood pressure, and found in many cases better blood pressure lowering by some ARBs and always better tolerability,” he told this news organization. “I think this study confirms and extends my thoughts between the two classes of blood pressure–lowering agents.”
Head-to-head comparisons of ACE inhibitors and ARBs limited to date
ACE inhibitors and ARBs each have extensive evidence supporting their roles as first-line medications in the treatment of hypertension, and each have the strongest recommendations in international guidelines.
However, ACE inhibitors are prescribed more commonly than ARBs as the first-line drug for lowering blood pressure, and head-to-head comparisons of the two are limited, with conflicting results.
For the study, Dr. Hripcsak and colleagues evaluated data on almost 3 million patients starting monotherapy with an ACE inhibitor or ARB for the first time between 1996 and 2018 in the United States, Germany, and South Korea, who had no history of heart disease or stroke.
They identified a total of 2,297,881 patients initiating ACE inhibitors and 673,938 starting ARBs. Among new users of ACE inhibitors, most received lisinopril (80%), followed by ramipril and enalapril, while most patients prescribed ARBs received losartan (45%), followed by valsartan and olmesartan.
With follow-up times ranging from about 4 months to more than 18 months, the data show no statistically significant differences between ACE inhibitors versus ARBs in the primary outcomes of acute myocardial infarction (hazard ratio, 1.11), heart failure (HR, 1.03), stroke (HR, 1.07), or composite cardiovascular events (HR, 1.06).
For secondary and safety outcomes, including an analysis of 51 possible side effects, ACE inhibitors, compared with ARBs, were associated with a significantly higher risk of angioedema (HR, 3.31; P < .01), cough (HR, 1.32; P < .01), acute pancreatitis (HR, 1.32; P = .02), gastrointestinal bleeding (HR, 1.18; P = .04), and abnormal weight loss (HR, 1.18; P = .04).
While the link between ACE inhibitors and pancreatitis has been previously reported, the association with GI bleeding may be a novel finding, with no prior studies comparing those effects in the two drug classes, the authors noted.
Despite most patients taking just a couple of drugs in either class, Dr. Hripcsak said, “we don’t expect that other drugs from those classes will have fewer differences. It is possible, of course, but that is not our expectation.”
Results only applicable to those starting therapy with RAS inhibitors
First author RuiJun Chen, MD, added that, importantly, the results may not apply to patients switching therapies or adding on therapy, “such as for the patient whose hypertension is not effectively controlled with one drug and requires the addition of a second medication,” he said in an interview.
“Also, the suggestion of preferentially prescribing ARBs only applies to those patients and providers intending to control blood pressure through RAS inhibition,” said Dr. Chen, an assistant professor in translational data science and informatics at Geisinger Medical Center in Danville, Pa., who was a National Library of Medicine postdoctoral fellow at Columbia University at the time of the study.
Hence, he stressed the results do not extend to other classes of recommended first-line blood pressure medications.
“Essentially, since this is an ACE inhibitor versus ARB study, we would not claim that ARBs are preferred over all other types of hypertension medications which were not studied here,” the researchers emphasize.
In addition to ARBs and ACE inhibitors, other medications recommended by the AHA/American College of Cardiology in the 2017 “Guideline for the Prevention, Detection, Evaluation and Management of High Blood Pressure in Adults” for the primary treatment of hypertension include thiazide diuretics and calcium channel blockers.
The study received support from the National Library of Medicine and the National Institute of Allergy and Infectious Diseases of the National Institutes of Health; the National Science Foundation; and the Ministries of Health & Welfare and of Trade, Industry & Energy of the Republic of Korea. Dr. Hripcsak reported receiving grants from the National Library of Medicine during the study and grants from Janssen Research outside the submitted work. Dr. Bakris reported being a consultant for Merck, KBP Biosciences, and Ionis.
A version of this article first appeared on Medscape.com.
In the largest comparison of angiotensin receptor blockers (ARBs) and ACE inhibitors to date, a study of nearly 2.3 million patients starting the drugs as monotherapy shows no significant differences between the two in the long-term prevention of hypertension-related cardiovascular events.
However, side effects were notably lower with ARBs.
“This is a very large, well-executed observational study that confirms that ARBs appear to have fewer side effects than ACE inhibitors, and no unexpected ARB side effects were detected,” senior author George Hripcsak, MD, professor and chair of biomedical informatics at Columbia University, New York, told this news organization.
“Despite being equally guideline-recommended first-line therapies for hypertension, these results support preferentially starting ARBs rather than ACE inhibitors when initiating treatment for hypertension for physicians and patients considering renin-angiotensin system (RAS) inhibition,” the authors added in the study, published online July 26, 2021, in the journal Hypertension.
They noted that both drug classes have been on the market a long time, with proven efficacy in hypertension and “a wide availability of inexpensive generic forms.”
They also stressed that their findings only apply to patients with hypertension for whom a RAS inhibitor would be the best choice of therapy.
Commenting on the research, George Bakris, MD, of the American Heart Association’s Comprehensive Hypertension Center at the University of Chicago, said the findings were consistent with his experience in prescribing as well as researching the two drug classes.
“I have been in practice for over 30 years and studied both classes, including head-to-head prospective trials to assess blood pressure, and found in many cases better blood pressure lowering by some ARBs and always better tolerability,” he told this news organization. “I think this study confirms and extends my thoughts between the two classes of blood pressure–lowering agents.”
Head-to-head comparisons of ACE inhibitors and ARBs limited to date
ACE inhibitors and ARBs each have extensive evidence supporting their roles as first-line medications in the treatment of hypertension, and each have the strongest recommendations in international guidelines.
However, ACE inhibitors are prescribed more commonly than ARBs as the first-line drug for lowering blood pressure, and head-to-head comparisons of the two are limited, with conflicting results.
For the study, Dr. Hripcsak and colleagues evaluated data on almost 3 million patients starting monotherapy with an ACE inhibitor or ARB for the first time between 1996 and 2018 in the United States, Germany, and South Korea, who had no history of heart disease or stroke.
They identified a total of 2,297,881 patients initiating ACE inhibitors and 673,938 starting ARBs. Among new users of ACE inhibitors, most received lisinopril (80%), followed by ramipril and enalapril, while most patients prescribed ARBs received losartan (45%), followed by valsartan and olmesartan.
With follow-up times ranging from about 4 months to more than 18 months, the data show no statistically significant differences between ACE inhibitors versus ARBs in the primary outcomes of acute myocardial infarction (hazard ratio, 1.11), heart failure (HR, 1.03), stroke (HR, 1.07), or composite cardiovascular events (HR, 1.06).
For secondary and safety outcomes, including an analysis of 51 possible side effects, ACE inhibitors, compared with ARBs, were associated with a significantly higher risk of angioedema (HR, 3.31; P < .01), cough (HR, 1.32; P < .01), acute pancreatitis (HR, 1.32; P = .02), gastrointestinal bleeding (HR, 1.18; P = .04), and abnormal weight loss (HR, 1.18; P = .04).
While the link between ACE inhibitors and pancreatitis has been previously reported, the association with GI bleeding may be a novel finding, with no prior studies comparing those effects in the two drug classes, the authors noted.
Despite most patients taking just a couple of drugs in either class, Dr. Hripcsak said, “we don’t expect that other drugs from those classes will have fewer differences. It is possible, of course, but that is not our expectation.”
Results only applicable to those starting therapy with RAS inhibitors
First author RuiJun Chen, MD, added that, importantly, the results may not apply to patients switching therapies or adding on therapy, “such as for the patient whose hypertension is not effectively controlled with one drug and requires the addition of a second medication,” he said in an interview.
“Also, the suggestion of preferentially prescribing ARBs only applies to those patients and providers intending to control blood pressure through RAS inhibition,” said Dr. Chen, an assistant professor in translational data science and informatics at Geisinger Medical Center in Danville, Pa., who was a National Library of Medicine postdoctoral fellow at Columbia University at the time of the study.
Hence, he stressed the results do not extend to other classes of recommended first-line blood pressure medications.
“Essentially, since this is an ACE inhibitor versus ARB study, we would not claim that ARBs are preferred over all other types of hypertension medications which were not studied here,” the researchers emphasize.
In addition to ARBs and ACE inhibitors, other medications recommended by the AHA/American College of Cardiology in the 2017 “Guideline for the Prevention, Detection, Evaluation and Management of High Blood Pressure in Adults” for the primary treatment of hypertension include thiazide diuretics and calcium channel blockers.
The study received support from the National Library of Medicine and the National Institute of Allergy and Infectious Diseases of the National Institutes of Health; the National Science Foundation; and the Ministries of Health & Welfare and of Trade, Industry & Energy of the Republic of Korea. Dr. Hripcsak reported receiving grants from the National Library of Medicine during the study and grants from Janssen Research outside the submitted work. Dr. Bakris reported being a consultant for Merck, KBP Biosciences, and Ionis.
A version of this article first appeared on Medscape.com.
In sickness and in health: Spouses can share risk for cardiac events
A study from Japan suggests that a history of cardiovascular events in a spouse may elevate risk for future CV events in the other partner, with one caveat: Men in the cohort study were at increased risk if their wives had such a history, but the association was only one way. The risk of events didn’t go up for women with husbands who had previously experienced a CV event.
The results highlight the need for clinicians to screen and possibly intervene with a primary CV prevention strategy “not only first-degree relatives but also spouses with a history of cardiovascular disease,” which is not currently part of the primary prevention guidelines, Hiroyuki Ohbe, MD, University of Tokyo, told this news organization.
In their study published online July 9 in Circulation: Cardiovascular Quality and Outcomes, Dr. Ohbe and Hideo Yasunaga, MD, PhD, of the same institution, assessed the risk of subsequent CV events in adults with a spouse who had experienced a stroke of any kind or had clinical ischemic heart disease such as angina or myocardial infarction.
Johanna Contreras, MD, director of heart failure at Mount Sinai Health System in New York, is not surprised by the finding that a wife’s CV history is linked to the CV risk in the husband.
“I see this often in my practice. When you live with someone, you also behave in a similar way as the other person,” Dr. Contreras told this news organization. “For example, couples who live together are likely to both exercise and have a healthy diet and not smoke.”
And most notably, she said, “the women are usually the ones who drive the healthy behaviors in the family; they watch what the family eats, where they eat, when they eat, and the men tend to allow the women to guide this behavior.”
Dr. Ohbe and Dr. Yasunaga agree, proposing that different results for men and women in the analysis may be because of the dependence of working-aged men on their wives for major aspects of lifestyle, such as diet and exercise. Moreover, they write, increased psychological and physical stress from taking care of a spouse with CV disease may also play a role, as caregivers often neglect their own health.
The team identified 13,759 adults in a large administrative database with no history of CV disease whose spouse had such a history at their first health checkup; they were the exposure group. The team matched each of them with up to four individuals (n = 55,027) who had no CV disease history and spouses without CV disease at their first health checkup; they were the nonexposure group.
The mean observation period was 7.9 years from the first health checkup, at which the subjects’ mean age was 56 years. During the follow-up, more people in the exposure group than the nonexposure group had a history of CV events, 0.6% versus 0.4%.
In the overall cohort, the hazard ratio for future severe CV events – heart failure hospitalization or MI – in those with spouses with a history of CV disease was 1.48 (95% confidence interval, 1.15-1.90).
When stratified by sex, men whose wives had CV disease showed a significantly increased risk of a future severe CV event (HR, 1.68; 95% CI, 1.22-2.32). But women with husbands with CV disease did not (HR, 1.22; 95% CI, 0.82-1.83).
The results of all four sensitivity analyses were similar to those of the primary analysis, both in the overall cohort and in the cohorts stratified by sex. The investigators performed multivariate survival analyses: one that excluded people whose partners had died, one that included death by any cause as an outcome, and one with propensity score matching.
Further studies are needed to confirm their observations and test whether a primary prevention strategy targeted at married couples could reduce CV events, note Dr. Ohbe and Dr. Yasunaga.
The findings have implications for everyday clinical practice, Dr. Contreras said. “When I see a patient who is married and has had a heart attack, I will insist on seeing the partner as well, and I will counsel them on working together to change their lifestyle,” she said in an interview.
“Often when you have that discussion with the couple after one has a heart attack, they quit smoking together, they go the gym together, and they get healthier together,” she said. “That’s now a very important conversation we have before they leave the hospital.”
The study was supported by grants from the Japan Ministry of Health, Ministry of Labour and Welfare, and the Ministry of Education, Culture, Sports, Science and Technology. Dr. Ohbe, Dr. Yasunaga, and Dr. Contreras have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A study from Japan suggests that a history of cardiovascular events in a spouse may elevate risk for future CV events in the other partner, with one caveat: Men in the cohort study were at increased risk if their wives had such a history, but the association was only one way. The risk of events didn’t go up for women with husbands who had previously experienced a CV event.
The results highlight the need for clinicians to screen and possibly intervene with a primary CV prevention strategy “not only first-degree relatives but also spouses with a history of cardiovascular disease,” which is not currently part of the primary prevention guidelines, Hiroyuki Ohbe, MD, University of Tokyo, told this news organization.
In their study published online July 9 in Circulation: Cardiovascular Quality and Outcomes, Dr. Ohbe and Hideo Yasunaga, MD, PhD, of the same institution, assessed the risk of subsequent CV events in adults with a spouse who had experienced a stroke of any kind or had clinical ischemic heart disease such as angina or myocardial infarction.
Johanna Contreras, MD, director of heart failure at Mount Sinai Health System in New York, is not surprised by the finding that a wife’s CV history is linked to the CV risk in the husband.
“I see this often in my practice. When you live with someone, you also behave in a similar way as the other person,” Dr. Contreras told this news organization. “For example, couples who live together are likely to both exercise and have a healthy diet and not smoke.”
And most notably, she said, “the women are usually the ones who drive the healthy behaviors in the family; they watch what the family eats, where they eat, when they eat, and the men tend to allow the women to guide this behavior.”
Dr. Ohbe and Dr. Yasunaga agree, proposing that different results for men and women in the analysis may be because of the dependence of working-aged men on their wives for major aspects of lifestyle, such as diet and exercise. Moreover, they write, increased psychological and physical stress from taking care of a spouse with CV disease may also play a role, as caregivers often neglect their own health.
The team identified 13,759 adults in a large administrative database with no history of CV disease whose spouse had such a history at their first health checkup; they were the exposure group. The team matched each of them with up to four individuals (n = 55,027) who had no CV disease history and spouses without CV disease at their first health checkup; they were the nonexposure group.
The mean observation period was 7.9 years from the first health checkup, at which the subjects’ mean age was 56 years. During the follow-up, more people in the exposure group than the nonexposure group had a history of CV events, 0.6% versus 0.4%.
In the overall cohort, the hazard ratio for future severe CV events – heart failure hospitalization or MI – in those with spouses with a history of CV disease was 1.48 (95% confidence interval, 1.15-1.90).
When stratified by sex, men whose wives had CV disease showed a significantly increased risk of a future severe CV event (HR, 1.68; 95% CI, 1.22-2.32). But women with husbands with CV disease did not (HR, 1.22; 95% CI, 0.82-1.83).
The results of all four sensitivity analyses were similar to those of the primary analysis, both in the overall cohort and in the cohorts stratified by sex. The investigators performed multivariate survival analyses: one that excluded people whose partners had died, one that included death by any cause as an outcome, and one with propensity score matching.
Further studies are needed to confirm their observations and test whether a primary prevention strategy targeted at married couples could reduce CV events, note Dr. Ohbe and Dr. Yasunaga.
The findings have implications for everyday clinical practice, Dr. Contreras said. “When I see a patient who is married and has had a heart attack, I will insist on seeing the partner as well, and I will counsel them on working together to change their lifestyle,” she said in an interview.
“Often when you have that discussion with the couple after one has a heart attack, they quit smoking together, they go the gym together, and they get healthier together,” she said. “That’s now a very important conversation we have before they leave the hospital.”
The study was supported by grants from the Japan Ministry of Health, Ministry of Labour and Welfare, and the Ministry of Education, Culture, Sports, Science and Technology. Dr. Ohbe, Dr. Yasunaga, and Dr. Contreras have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A study from Japan suggests that a history of cardiovascular events in a spouse may elevate risk for future CV events in the other partner, with one caveat: Men in the cohort study were at increased risk if their wives had such a history, but the association was only one way. The risk of events didn’t go up for women with husbands who had previously experienced a CV event.
The results highlight the need for clinicians to screen and possibly intervene with a primary CV prevention strategy “not only first-degree relatives but also spouses with a history of cardiovascular disease,” which is not currently part of the primary prevention guidelines, Hiroyuki Ohbe, MD, University of Tokyo, told this news organization.
In their study published online July 9 in Circulation: Cardiovascular Quality and Outcomes, Dr. Ohbe and Hideo Yasunaga, MD, PhD, of the same institution, assessed the risk of subsequent CV events in adults with a spouse who had experienced a stroke of any kind or had clinical ischemic heart disease such as angina or myocardial infarction.
Johanna Contreras, MD, director of heart failure at Mount Sinai Health System in New York, is not surprised by the finding that a wife’s CV history is linked to the CV risk in the husband.
“I see this often in my practice. When you live with someone, you also behave in a similar way as the other person,” Dr. Contreras told this news organization. “For example, couples who live together are likely to both exercise and have a healthy diet and not smoke.”
And most notably, she said, “the women are usually the ones who drive the healthy behaviors in the family; they watch what the family eats, where they eat, when they eat, and the men tend to allow the women to guide this behavior.”
Dr. Ohbe and Dr. Yasunaga agree, proposing that different results for men and women in the analysis may be because of the dependence of working-aged men on their wives for major aspects of lifestyle, such as diet and exercise. Moreover, they write, increased psychological and physical stress from taking care of a spouse with CV disease may also play a role, as caregivers often neglect their own health.
The team identified 13,759 adults in a large administrative database with no history of CV disease whose spouse had such a history at their first health checkup; they were the exposure group. The team matched each of them with up to four individuals (n = 55,027) who had no CV disease history and spouses without CV disease at their first health checkup; they were the nonexposure group.
The mean observation period was 7.9 years from the first health checkup, at which the subjects’ mean age was 56 years. During the follow-up, more people in the exposure group than the nonexposure group had a history of CV events, 0.6% versus 0.4%.
In the overall cohort, the hazard ratio for future severe CV events – heart failure hospitalization or MI – in those with spouses with a history of CV disease was 1.48 (95% confidence interval, 1.15-1.90).
When stratified by sex, men whose wives had CV disease showed a significantly increased risk of a future severe CV event (HR, 1.68; 95% CI, 1.22-2.32). But women with husbands with CV disease did not (HR, 1.22; 95% CI, 0.82-1.83).
The results of all four sensitivity analyses were similar to those of the primary analysis, both in the overall cohort and in the cohorts stratified by sex. The investigators performed multivariate survival analyses: one that excluded people whose partners had died, one that included death by any cause as an outcome, and one with propensity score matching.
Further studies are needed to confirm their observations and test whether a primary prevention strategy targeted at married couples could reduce CV events, note Dr. Ohbe and Dr. Yasunaga.
The findings have implications for everyday clinical practice, Dr. Contreras said. “When I see a patient who is married and has had a heart attack, I will insist on seeing the partner as well, and I will counsel them on working together to change their lifestyle,” she said in an interview.
“Often when you have that discussion with the couple after one has a heart attack, they quit smoking together, they go the gym together, and they get healthier together,” she said. “That’s now a very important conversation we have before they leave the hospital.”
The study was supported by grants from the Japan Ministry of Health, Ministry of Labour and Welfare, and the Ministry of Education, Culture, Sports, Science and Technology. Dr. Ohbe, Dr. Yasunaga, and Dr. Contreras have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Direct oral anticoagulants: Competition brought no cost relief
Medicare Part D spending for oral anticoagulants has risen by almost 1,600% since 2011, while the number of users has increased by just 95%, according to a new study.
In 2011, the year after the first direct oral anticoagulant (DOACs) was approved, Medicare Part D spent $0.44 billion on all oral anticoagulants. By 2019, when there a total of four DOACs on the market, spending was $7.38 billion, an increase of 1,577%, Aaron Troy, MD, MPH, and Timothy S. Anderson, MD, MAS, said in JAMA Health Forum.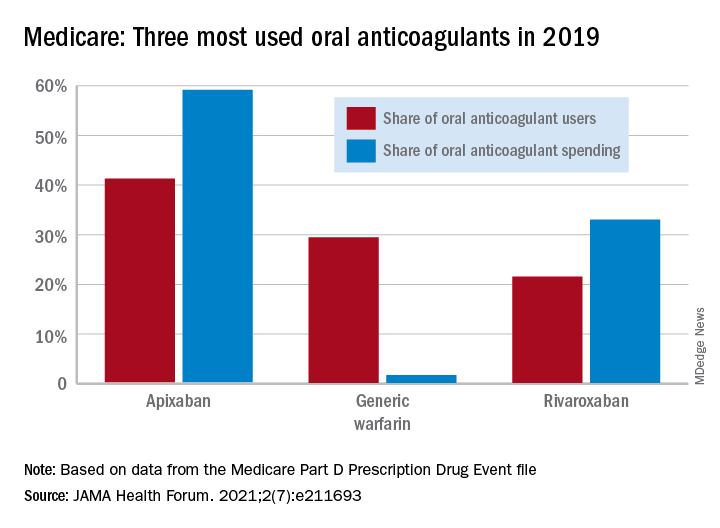
Over that same time, the number of beneficiaries using oral anticoagulants went from 2.68 million to 5.24 million, they said, based on data from the Medicare Part D Prescription Drug Event file.
“While higher prices for novel therapeutics like DOACs, which offer clear benefits, such as decreased drug-drug interactions and improved persistence, may partly reflect value and help drive innovation, the patterns and effects of spending on novel medications still merit attention,” they noted.
One pattern of use looked like this: 0.2 million Medicare beneficiaries took DOACs in 2011,compared with 3.5 million in 2019, while the number of warfarin users dropped from 2.48 million to 1.74 million, the investigators reported.
As for spending over the study period, the cost to treat one beneficiary with atrial fibrillation increased by 9.3% each year for apixaban (a DOAC that was the most popular oral anticoagulant in 2019), decreased 27.6% per year for generic warfarin, and increased 9.5% per year for rivaroxaban, said Dr. Troy and Dr. Anderson of Beth Israel Deaconess Medical Center, Boston.
Rising Part D enrollment had an effect on spending growth, as did increased use of oral anticoagulants in general. The introduction of competing DOACs, however, “did not substantially curb annual spending increases, suggesting a lack of price competition, which is consistent with trends observed in other therapeutic categories,” they wrote.
Dr. Anderson has received research grants from the National Institute on Aging and the American College of Cardiology outside of this study and honoraria from Alosa Health. No other disclosures were reported.
Medicare Part D spending for oral anticoagulants has risen by almost 1,600% since 2011, while the number of users has increased by just 95%, according to a new study.
In 2011, the year after the first direct oral anticoagulant (DOACs) was approved, Medicare Part D spent $0.44 billion on all oral anticoagulants. By 2019, when there a total of four DOACs on the market, spending was $7.38 billion, an increase of 1,577%, Aaron Troy, MD, MPH, and Timothy S. Anderson, MD, MAS, said in JAMA Health Forum.
Over that same time, the number of beneficiaries using oral anticoagulants went from 2.68 million to 5.24 million, they said, based on data from the Medicare Part D Prescription Drug Event file.
“While higher prices for novel therapeutics like DOACs, which offer clear benefits, such as decreased drug-drug interactions and improved persistence, may partly reflect value and help drive innovation, the patterns and effects of spending on novel medications still merit attention,” they noted.
One pattern of use looked like this: 0.2 million Medicare beneficiaries took DOACs in 2011,compared with 3.5 million in 2019, while the number of warfarin users dropped from 2.48 million to 1.74 million, the investigators reported.
As for spending over the study period, the cost to treat one beneficiary with atrial fibrillation increased by 9.3% each year for apixaban (a DOAC that was the most popular oral anticoagulant in 2019), decreased 27.6% per year for generic warfarin, and increased 9.5% per year for rivaroxaban, said Dr. Troy and Dr. Anderson of Beth Israel Deaconess Medical Center, Boston.
Rising Part D enrollment had an effect on spending growth, as did increased use of oral anticoagulants in general. The introduction of competing DOACs, however, “did not substantially curb annual spending increases, suggesting a lack of price competition, which is consistent with trends observed in other therapeutic categories,” they wrote.
Dr. Anderson has received research grants from the National Institute on Aging and the American College of Cardiology outside of this study and honoraria from Alosa Health. No other disclosures were reported.
Medicare Part D spending for oral anticoagulants has risen by almost 1,600% since 2011, while the number of users has increased by just 95%, according to a new study.
In 2011, the year after the first direct oral anticoagulant (DOACs) was approved, Medicare Part D spent $0.44 billion on all oral anticoagulants. By 2019, when there a total of four DOACs on the market, spending was $7.38 billion, an increase of 1,577%, Aaron Troy, MD, MPH, and Timothy S. Anderson, MD, MAS, said in JAMA Health Forum.
Over that same time, the number of beneficiaries using oral anticoagulants went from 2.68 million to 5.24 million, they said, based on data from the Medicare Part D Prescription Drug Event file.
“While higher prices for novel therapeutics like DOACs, which offer clear benefits, such as decreased drug-drug interactions and improved persistence, may partly reflect value and help drive innovation, the patterns and effects of spending on novel medications still merit attention,” they noted.
One pattern of use looked like this: 0.2 million Medicare beneficiaries took DOACs in 2011,compared with 3.5 million in 2019, while the number of warfarin users dropped from 2.48 million to 1.74 million, the investigators reported.
As for spending over the study period, the cost to treat one beneficiary with atrial fibrillation increased by 9.3% each year for apixaban (a DOAC that was the most popular oral anticoagulant in 2019), decreased 27.6% per year for generic warfarin, and increased 9.5% per year for rivaroxaban, said Dr. Troy and Dr. Anderson of Beth Israel Deaconess Medical Center, Boston.
Rising Part D enrollment had an effect on spending growth, as did increased use of oral anticoagulants in general. The introduction of competing DOACs, however, “did not substantially curb annual spending increases, suggesting a lack of price competition, which is consistent with trends observed in other therapeutic categories,” they wrote.
Dr. Anderson has received research grants from the National Institute on Aging and the American College of Cardiology outside of this study and honoraria from Alosa Health. No other disclosures were reported.
FROM JAMA HEALTH FORUM
More on GRADE: Cognitive deficits linked to CV risk factors in T2D
In type 2 diabetes (T2D), a greater degree of hyperlipidemia and hypertension, although not hyperglycemia, was associated with measurable cognitive impairment even among patients with only a 4-year mean disease duration, according to a substudy of the GRADE trial.
The association of these cardiovascular (CV) risk factors with impairments in cognition has been reported before, but the findings are notable because the mean duration of T2D was short in a relatively healthy study population, reported a multicenter team of investigators.
The relative impairments in cognitive function “may not be clinically significant given the very small size of the differences,” conceded the authors of this study, led by José A. Luchsinger, MD, but they are consistent with previous reports of the same association in older patients with a longer duration of diabetes. In other words, the data suggest the risk of cognitive loss from CV risk factors in T2D patients begins early.
“A potential explanation for the small differences, compared with those previously reported, is that the GRADE cohort is relatively young with a healthier cardiovascular profile and shorter diabetes duration compared with other studies,” reported the investigators, whose results were published online July 20, 2021, in Diabetes Care.
99% complete cognitive assessments
In the GRADE (Glycemia Reduction Approaches in Diabetes: Comparative Effectiveness) trial, 5,018 (99.4%) of the 5,047 enrolled patients completed a battery of cognitive assessments at baseline. Patients were excluded from this study if they had any major CV event in the previous year, if they had T2D for more than 10 years, if they had significant renal impairment, and if they had any history of stage 3 or greater heart failure. Their mean age was 56.7 years.
By cross-sectional analysis, cognitive evaluations, including the Digit Symbol Substitution Test (DSST) and the Spanish English Verbal Learning Test, were evaluated in relation to baseline LDL cholesterol levels, systolic and diastolic blood pressure, hemoglobin A1c, and statin use.
Unlike previous studies in T2D patients, no relationship was observed between cognitive function and A1c level at baseline. However, LDL cholesterol greater than 100 mg/dL was associated with cognitive impairment as measured with the DSST after adjustment for age, sex, education, and general health. The mean difference relative to LDL cholesterol below 70 mg/dL was only 1.8 points, but this was highly significant (P < .001).
Similarly, significant but modest cognitive impairment on DSST score after adjustment for variables were seen for those with a systolic BP between 120 mg and <140 mg relative to either <120 mm Hg or at least 140 mm Hg (P = .014). The same was seen for diastolic BPs of 80 to <90 when compared with either <80 mm Hg or to 90 mm Hg or higher (P = .01).
For those taking statins versus no statins at baseline, there was a 1.4-point mean advantage in DSST score after adjusting for variables (P < .001).
Modest cognitive impairments recorded
Again, the absolute mean differences in the DSST cognitive scores, despite their statistical significance, were modest, according to the authors. In general, the mean difference was rarely greater than 2.0 points and often 1.0 point or less. The authors acknowledged that these changes are of an uncertain clinical significance, but they considered the findings consistent with the association of CV risk factors with cognitive deficits in older T2DM patients or T2DM patients with longer duration of disease.
One difference between this GRADE substudy and previous studies was the lack of an association between cognitive impairment and hyperglycemia. In the ACCORD trial for example, increased levels of blood glycemia were associated with lower performance on numerous tests of cognitive function.
In the Diabetes Control and Complications Trial (DCCT), poorer glycemic control was related to poorer performance on tests of executive function.
Both of those studies also linked hypertension and hyperlipidemia with cognitive deficits, but given that patients in ACCORD had T2DM of substantially longer duration and those in DCCT were older, “it seems reasonable to speculate that, in patients with diabetes duration of less than 10 years, the association between hyperglycemia and cognitive performance may not yet be evident,” the GRADE authors reported.
GRADE trial compares drugs in four classes
The GRADE trial was conducted to compare four classes of T2D therapies for long-term glycemic control as expressed by A1c control over time. The results of the trial, presented recently at the 2021 annual scientific sessions of the American Diabetes Association, found that insulin glargine and the glucagonlike peptide–1 receptor agonist liraglutide performed best on the primary endpoint of maintaining A1c below 7.0%. Both performed significantly better than the sulfonylurea glimepiride and the dipeptidyl peptidase–4 inhibitor sitagliptin.
This substudy of baseline cognitive function in the relatively large GRADE trial provided a unique opportunity to evaluate the impact of CV risk factors in patients with T2D of relatively short duration.
While the data support the adverse impact of inadequately controlled modifiable risk factors on cognitive function in T2D patients, David R. Matthews, DPhil, BM, BCh, emeritus professor of diabetes medicine at the University of Oxford (England), noted that the association was weak and advised a cautious interpretation.
“The effect size is very small indeed. The data are found as a subset of multiple testing,” he said in an interview. He suggested the associations might be the result of “data farming,” and he emphasized that the relationships between these risk factors and cognitive deficits are associations that do not imply causation.
Nevertheless, and despite their unclear clinical implications, Dr. Matthews said that these data might still have a message.
“It is another reminder that for many reasons we all need to be alert to the need for lowering hyperlipidemia and hypertension to normal levels – the benefits may not just be limited to cardiovascular outcome,” Dr. Matthews stated.
The lead author of the study, Dr. Luchsinger, also cautioned against overinterpreting the data.
While the data show that “lipid and blood pressure control within recommended guidelines are associated with marginally better cognitive function in patients with type 2 diabetes of less than 5 years duration on average,” he added that “the study is limited by its cross-sectional nature.”
He indicated that further analysis will be helpful in assessing the implications.
“Longitudinal analyses of the same group of individuals will be conducted next year,” noted Dr. Luchsinger, associate professor of medicine and epidemiology, Columbia University Medical Center, New York.
Dr. Luchsinger reported financial relationships with vTv therapeutics. Dr. Matthews reported no potential conflicts of interest.
In type 2 diabetes (T2D), a greater degree of hyperlipidemia and hypertension, although not hyperglycemia, was associated with measurable cognitive impairment even among patients with only a 4-year mean disease duration, according to a substudy of the GRADE trial.
The association of these cardiovascular (CV) risk factors with impairments in cognition has been reported before, but the findings are notable because the mean duration of T2D was short in a relatively healthy study population, reported a multicenter team of investigators.
The relative impairments in cognitive function “may not be clinically significant given the very small size of the differences,” conceded the authors of this study, led by José A. Luchsinger, MD, but they are consistent with previous reports of the same association in older patients with a longer duration of diabetes. In other words, the data suggest the risk of cognitive loss from CV risk factors in T2D patients begins early.
“A potential explanation for the small differences, compared with those previously reported, is that the GRADE cohort is relatively young with a healthier cardiovascular profile and shorter diabetes duration compared with other studies,” reported the investigators, whose results were published online July 20, 2021, in Diabetes Care.
99% complete cognitive assessments
In the GRADE (Glycemia Reduction Approaches in Diabetes: Comparative Effectiveness) trial, 5,018 (99.4%) of the 5,047 enrolled patients completed a battery of cognitive assessments at baseline. Patients were excluded from this study if they had any major CV event in the previous year, if they had T2D for more than 10 years, if they had significant renal impairment, and if they had any history of stage 3 or greater heart failure. Their mean age was 56.7 years.
By cross-sectional analysis, cognitive evaluations, including the Digit Symbol Substitution Test (DSST) and the Spanish English Verbal Learning Test, were evaluated in relation to baseline LDL cholesterol levels, systolic and diastolic blood pressure, hemoglobin A1c, and statin use.
Unlike previous studies in T2D patients, no relationship was observed between cognitive function and A1c level at baseline. However, LDL cholesterol greater than 100 mg/dL was associated with cognitive impairment as measured with the DSST after adjustment for age, sex, education, and general health. The mean difference relative to LDL cholesterol below 70 mg/dL was only 1.8 points, but this was highly significant (P < .001).
Similarly, significant but modest cognitive impairment on DSST score after adjustment for variables were seen for those with a systolic BP between 120 mg and <140 mg relative to either <120 mm Hg or at least 140 mm Hg (P = .014). The same was seen for diastolic BPs of 80 to <90 when compared with either <80 mm Hg or to 90 mm Hg or higher (P = .01).
For those taking statins versus no statins at baseline, there was a 1.4-point mean advantage in DSST score after adjusting for variables (P < .001).
Modest cognitive impairments recorded
Again, the absolute mean differences in the DSST cognitive scores, despite their statistical significance, were modest, according to the authors. In general, the mean difference was rarely greater than 2.0 points and often 1.0 point or less. The authors acknowledged that these changes are of an uncertain clinical significance, but they considered the findings consistent with the association of CV risk factors with cognitive deficits in older T2DM patients or T2DM patients with longer duration of disease.
One difference between this GRADE substudy and previous studies was the lack of an association between cognitive impairment and hyperglycemia. In the ACCORD trial for example, increased levels of blood glycemia were associated with lower performance on numerous tests of cognitive function.
In the Diabetes Control and Complications Trial (DCCT), poorer glycemic control was related to poorer performance on tests of executive function.
Both of those studies also linked hypertension and hyperlipidemia with cognitive deficits, but given that patients in ACCORD had T2DM of substantially longer duration and those in DCCT were older, “it seems reasonable to speculate that, in patients with diabetes duration of less than 10 years, the association between hyperglycemia and cognitive performance may not yet be evident,” the GRADE authors reported.
GRADE trial compares drugs in four classes
The GRADE trial was conducted to compare four classes of T2D therapies for long-term glycemic control as expressed by A1c control over time. The results of the trial, presented recently at the 2021 annual scientific sessions of the American Diabetes Association, found that insulin glargine and the glucagonlike peptide–1 receptor agonist liraglutide performed best on the primary endpoint of maintaining A1c below 7.0%. Both performed significantly better than the sulfonylurea glimepiride and the dipeptidyl peptidase–4 inhibitor sitagliptin.
This substudy of baseline cognitive function in the relatively large GRADE trial provided a unique opportunity to evaluate the impact of CV risk factors in patients with T2D of relatively short duration.
While the data support the adverse impact of inadequately controlled modifiable risk factors on cognitive function in T2D patients, David R. Matthews, DPhil, BM, BCh, emeritus professor of diabetes medicine at the University of Oxford (England), noted that the association was weak and advised a cautious interpretation.
“The effect size is very small indeed. The data are found as a subset of multiple testing,” he said in an interview. He suggested the associations might be the result of “data farming,” and he emphasized that the relationships between these risk factors and cognitive deficits are associations that do not imply causation.
Nevertheless, and despite their unclear clinical implications, Dr. Matthews said that these data might still have a message.
“It is another reminder that for many reasons we all need to be alert to the need for lowering hyperlipidemia and hypertension to normal levels – the benefits may not just be limited to cardiovascular outcome,” Dr. Matthews stated.
The lead author of the study, Dr. Luchsinger, also cautioned against overinterpreting the data.
While the data show that “lipid and blood pressure control within recommended guidelines are associated with marginally better cognitive function in patients with type 2 diabetes of less than 5 years duration on average,” he added that “the study is limited by its cross-sectional nature.”
He indicated that further analysis will be helpful in assessing the implications.
“Longitudinal analyses of the same group of individuals will be conducted next year,” noted Dr. Luchsinger, associate professor of medicine and epidemiology, Columbia University Medical Center, New York.
Dr. Luchsinger reported financial relationships with vTv therapeutics. Dr. Matthews reported no potential conflicts of interest.
In type 2 diabetes (T2D), a greater degree of hyperlipidemia and hypertension, although not hyperglycemia, was associated with measurable cognitive impairment even among patients with only a 4-year mean disease duration, according to a substudy of the GRADE trial.
The association of these cardiovascular (CV) risk factors with impairments in cognition has been reported before, but the findings are notable because the mean duration of T2D was short in a relatively healthy study population, reported a multicenter team of investigators.
The relative impairments in cognitive function “may not be clinically significant given the very small size of the differences,” conceded the authors of this study, led by José A. Luchsinger, MD, but they are consistent with previous reports of the same association in older patients with a longer duration of diabetes. In other words, the data suggest the risk of cognitive loss from CV risk factors in T2D patients begins early.
“A potential explanation for the small differences, compared with those previously reported, is that the GRADE cohort is relatively young with a healthier cardiovascular profile and shorter diabetes duration compared with other studies,” reported the investigators, whose results were published online July 20, 2021, in Diabetes Care.
99% complete cognitive assessments
In the GRADE (Glycemia Reduction Approaches in Diabetes: Comparative Effectiveness) trial, 5,018 (99.4%) of the 5,047 enrolled patients completed a battery of cognitive assessments at baseline. Patients were excluded from this study if they had any major CV event in the previous year, if they had T2D for more than 10 years, if they had significant renal impairment, and if they had any history of stage 3 or greater heart failure. Their mean age was 56.7 years.
By cross-sectional analysis, cognitive evaluations, including the Digit Symbol Substitution Test (DSST) and the Spanish English Verbal Learning Test, were evaluated in relation to baseline LDL cholesterol levels, systolic and diastolic blood pressure, hemoglobin A1c, and statin use.
Unlike previous studies in T2D patients, no relationship was observed between cognitive function and A1c level at baseline. However, LDL cholesterol greater than 100 mg/dL was associated with cognitive impairment as measured with the DSST after adjustment for age, sex, education, and general health. The mean difference relative to LDL cholesterol below 70 mg/dL was only 1.8 points, but this was highly significant (P < .001).
Similarly, significant but modest cognitive impairment on DSST score after adjustment for variables were seen for those with a systolic BP between 120 mg and <140 mg relative to either <120 mm Hg or at least 140 mm Hg (P = .014). The same was seen for diastolic BPs of 80 to <90 when compared with either <80 mm Hg or to 90 mm Hg or higher (P = .01).
For those taking statins versus no statins at baseline, there was a 1.4-point mean advantage in DSST score after adjusting for variables (P < .001).
Modest cognitive impairments recorded
Again, the absolute mean differences in the DSST cognitive scores, despite their statistical significance, were modest, according to the authors. In general, the mean difference was rarely greater than 2.0 points and often 1.0 point or less. The authors acknowledged that these changes are of an uncertain clinical significance, but they considered the findings consistent with the association of CV risk factors with cognitive deficits in older T2DM patients or T2DM patients with longer duration of disease.
One difference between this GRADE substudy and previous studies was the lack of an association between cognitive impairment and hyperglycemia. In the ACCORD trial for example, increased levels of blood glycemia were associated with lower performance on numerous tests of cognitive function.
In the Diabetes Control and Complications Trial (DCCT), poorer glycemic control was related to poorer performance on tests of executive function.
Both of those studies also linked hypertension and hyperlipidemia with cognitive deficits, but given that patients in ACCORD had T2DM of substantially longer duration and those in DCCT were older, “it seems reasonable to speculate that, in patients with diabetes duration of less than 10 years, the association between hyperglycemia and cognitive performance may not yet be evident,” the GRADE authors reported.
GRADE trial compares drugs in four classes
The GRADE trial was conducted to compare four classes of T2D therapies for long-term glycemic control as expressed by A1c control over time. The results of the trial, presented recently at the 2021 annual scientific sessions of the American Diabetes Association, found that insulin glargine and the glucagonlike peptide–1 receptor agonist liraglutide performed best on the primary endpoint of maintaining A1c below 7.0%. Both performed significantly better than the sulfonylurea glimepiride and the dipeptidyl peptidase–4 inhibitor sitagliptin.
This substudy of baseline cognitive function in the relatively large GRADE trial provided a unique opportunity to evaluate the impact of CV risk factors in patients with T2D of relatively short duration.
While the data support the adverse impact of inadequately controlled modifiable risk factors on cognitive function in T2D patients, David R. Matthews, DPhil, BM, BCh, emeritus professor of diabetes medicine at the University of Oxford (England), noted that the association was weak and advised a cautious interpretation.
“The effect size is very small indeed. The data are found as a subset of multiple testing,” he said in an interview. He suggested the associations might be the result of “data farming,” and he emphasized that the relationships between these risk factors and cognitive deficits are associations that do not imply causation.
Nevertheless, and despite their unclear clinical implications, Dr. Matthews said that these data might still have a message.
“It is another reminder that for many reasons we all need to be alert to the need for lowering hyperlipidemia and hypertension to normal levels – the benefits may not just be limited to cardiovascular outcome,” Dr. Matthews stated.
The lead author of the study, Dr. Luchsinger, also cautioned against overinterpreting the data.
While the data show that “lipid and blood pressure control within recommended guidelines are associated with marginally better cognitive function in patients with type 2 diabetes of less than 5 years duration on average,” he added that “the study is limited by its cross-sectional nature.”
He indicated that further analysis will be helpful in assessing the implications.
“Longitudinal analyses of the same group of individuals will be conducted next year,” noted Dr. Luchsinger, associate professor of medicine and epidemiology, Columbia University Medical Center, New York.
Dr. Luchsinger reported financial relationships with vTv therapeutics. Dr. Matthews reported no potential conflicts of interest.
FROM DIABETES CARE
Diabetes duration linked to increasing heart failure risk
In a multivariable analysis the rate of incident heart failure increased steadily and significantly as diabetes duration increased. Among the 168 study subjects (2% of the total study group) who had diabetes for at least 15 years, the subsequent incidence of heart failure was nearly threefold higher than among the 4,802 subjects (49%) who never had diabetes or prediabetes, reported Justin B. Echouffo-Tcheugui, MD, PhD, and coauthors in an article published in JACC Heart Failure.
People with prediabetes (32% of the study population) had a significant but modest increased rate of incident heart failure that was 16% higher than in control subjects who never developed diabetes. People with diabetes for durations of 0-4.9 years, 5.0-9.9 years, or 10-14.9 years, had steadily increasing relative incident heart failure rates of 29%, 97%, and 210%, respectively, compared with controls, reported Dr. Echouffo-Tcheugui, an endocrinologist at Johns Hopkins Medicine in Baltimore.
Similar rates of HFrEF and HFpEF
Among all 1,841 people in the dataset with diabetes for any length of time each additional 5 years of the disorder linked with a significant, relative 17% increase in the rate of incident heart failure. Incidence of heart failure rose even more sharply with added duration among those with a hemoglobin A1c of 7% or greater, compared with those with better glycemic control. And the rate of incident heart failure with reduced ejection fraction (HFrEF) roughly matched the rate of incident heart failure with preserved ejection fraction (HFpEF).
The study dataset included 9,734 adults enrolled into the Atherosclerosis Risk in Communities (ARIC) study, and during a median follow-up of 22.5 years they had nearly 2,000 episodes of either hospitalization or death secondary to incident heart failure. This included 617 (31%) events involving HFpEF, 495 events (25%) involving HFrEF, and 876 unclassified heart failure events.
The cohort averaged 63 years of age; 58% were women, 23% were Black, and 77% were White (the study design excluded people with other racial and ethnic backgrounds). The study design also excluded people with a history of heart failure or coronary artery disease, as well as those diagnosed with diabetes prior to age 18 resulting in a study group that presumably mostly had type 2 diabetes when diabetes was present. The report provided no data on the specific numbers of patients with type 1 or type 2 diabetes.
“It’s not surprising that a longer duration of diabetes is associated with heart failure, but the etiology remains problematic,” commented Robert H. Eckel, MD, an endocrinologist at the University of Colorado at Denver, Aurora. “The impact of diabetes on incident heart failure is not well know, particularly duration of diabetes,” although disorders often found in patients with diabetes, such as hypertension and diabetic cardiomyopathy, likely have roles in causing heart failure, he said.
Diabetes duration may signal need for an SGLT2 inhibitor
“With emerging novel treatments like the SGLT2 [sodium-glucose cotransporter 2] inhibitors for preventing heart failure hospitalizations and deaths in patients with type 2 diabetes, this is a timely analysis,” Dr. Eckel said in an interview.
“There is no question that with increased duration of type 2 diabetes” the need for an agent from the SGLT2-inhibitor class increases. Although, because of the proven protection these drugs give against heart failure events and progression of chronic kidney disease, treatment with this drug class should start early in patients with type 2 diabetes, he added.
Dr. Echouffo-Tcheugui and his coauthors agreed, citing two important clinical take-aways from their findings:
First, interventions that delay the onset of diabetes may potentially reduce incident heart failure; second, patients with diabetes might benefit from cardioprotective treatments such as SGLT2 inhibitors, the report said.
“Our observations suggest the potential prognostic relevance of diabetes duration in assessing heart failure,” the authors wrote. Integrating diabetes duration into heart failure risk estimation in people with diabetes “could help refine the selection of high-risk individuals who may derive the greatest absolute benefit from aggressive cardioprotective therapies such as SGLT2 inhibitors.”
The analysis also identified several other demographic and clinical factors that influenced the relative effect of diabetes duration. Longer duration was linked with higher rates of incident heart failure in women compared with men, in Blacks compared with Whites, in people younger than 65 compared with older people, in people with an A1c of 7% or higher, and in those with a body mass index of 30 kg/m2 or greater.
The ARIC study and the analyses run by Dr. Echouffo-Tcheugui and his coauthors received no commercial funding. Dr. Echouffo-Tcheugui and Dr. Eckel had no relevant disclosures.
In a multivariable analysis the rate of incident heart failure increased steadily and significantly as diabetes duration increased. Among the 168 study subjects (2% of the total study group) who had diabetes for at least 15 years, the subsequent incidence of heart failure was nearly threefold higher than among the 4,802 subjects (49%) who never had diabetes or prediabetes, reported Justin B. Echouffo-Tcheugui, MD, PhD, and coauthors in an article published in JACC Heart Failure.
People with prediabetes (32% of the study population) had a significant but modest increased rate of incident heart failure that was 16% higher than in control subjects who never developed diabetes. People with diabetes for durations of 0-4.9 years, 5.0-9.9 years, or 10-14.9 years, had steadily increasing relative incident heart failure rates of 29%, 97%, and 210%, respectively, compared with controls, reported Dr. Echouffo-Tcheugui, an endocrinologist at Johns Hopkins Medicine in Baltimore.
Similar rates of HFrEF and HFpEF
Among all 1,841 people in the dataset with diabetes for any length of time each additional 5 years of the disorder linked with a significant, relative 17% increase in the rate of incident heart failure. Incidence of heart failure rose even more sharply with added duration among those with a hemoglobin A1c of 7% or greater, compared with those with better glycemic control. And the rate of incident heart failure with reduced ejection fraction (HFrEF) roughly matched the rate of incident heart failure with preserved ejection fraction (HFpEF).
The study dataset included 9,734 adults enrolled into the Atherosclerosis Risk in Communities (ARIC) study, and during a median follow-up of 22.5 years they had nearly 2,000 episodes of either hospitalization or death secondary to incident heart failure. This included 617 (31%) events involving HFpEF, 495 events (25%) involving HFrEF, and 876 unclassified heart failure events.
The cohort averaged 63 years of age; 58% were women, 23% were Black, and 77% were White (the study design excluded people with other racial and ethnic backgrounds). The study design also excluded people with a history of heart failure or coronary artery disease, as well as those diagnosed with diabetes prior to age 18 resulting in a study group that presumably mostly had type 2 diabetes when diabetes was present. The report provided no data on the specific numbers of patients with type 1 or type 2 diabetes.
“It’s not surprising that a longer duration of diabetes is associated with heart failure, but the etiology remains problematic,” commented Robert H. Eckel, MD, an endocrinologist at the University of Colorado at Denver, Aurora. “The impact of diabetes on incident heart failure is not well know, particularly duration of diabetes,” although disorders often found in patients with diabetes, such as hypertension and diabetic cardiomyopathy, likely have roles in causing heart failure, he said.
Diabetes duration may signal need for an SGLT2 inhibitor
“With emerging novel treatments like the SGLT2 [sodium-glucose cotransporter 2] inhibitors for preventing heart failure hospitalizations and deaths in patients with type 2 diabetes, this is a timely analysis,” Dr. Eckel said in an interview.
“There is no question that with increased duration of type 2 diabetes” the need for an agent from the SGLT2-inhibitor class increases. Although, because of the proven protection these drugs give against heart failure events and progression of chronic kidney disease, treatment with this drug class should start early in patients with type 2 diabetes, he added.
Dr. Echouffo-Tcheugui and his coauthors agreed, citing two important clinical take-aways from their findings:
First, interventions that delay the onset of diabetes may potentially reduce incident heart failure; second, patients with diabetes might benefit from cardioprotective treatments such as SGLT2 inhibitors, the report said.
“Our observations suggest the potential prognostic relevance of diabetes duration in assessing heart failure,” the authors wrote. Integrating diabetes duration into heart failure risk estimation in people with diabetes “could help refine the selection of high-risk individuals who may derive the greatest absolute benefit from aggressive cardioprotective therapies such as SGLT2 inhibitors.”
The analysis also identified several other demographic and clinical factors that influenced the relative effect of diabetes duration. Longer duration was linked with higher rates of incident heart failure in women compared with men, in Blacks compared with Whites, in people younger than 65 compared with older people, in people with an A1c of 7% or higher, and in those with a body mass index of 30 kg/m2 or greater.
The ARIC study and the analyses run by Dr. Echouffo-Tcheugui and his coauthors received no commercial funding. Dr. Echouffo-Tcheugui and Dr. Eckel had no relevant disclosures.
In a multivariable analysis the rate of incident heart failure increased steadily and significantly as diabetes duration increased. Among the 168 study subjects (2% of the total study group) who had diabetes for at least 15 years, the subsequent incidence of heart failure was nearly threefold higher than among the 4,802 subjects (49%) who never had diabetes or prediabetes, reported Justin B. Echouffo-Tcheugui, MD, PhD, and coauthors in an article published in JACC Heart Failure.
People with prediabetes (32% of the study population) had a significant but modest increased rate of incident heart failure that was 16% higher than in control subjects who never developed diabetes. People with diabetes for durations of 0-4.9 years, 5.0-9.9 years, or 10-14.9 years, had steadily increasing relative incident heart failure rates of 29%, 97%, and 210%, respectively, compared with controls, reported Dr. Echouffo-Tcheugui, an endocrinologist at Johns Hopkins Medicine in Baltimore.
Similar rates of HFrEF and HFpEF
Among all 1,841 people in the dataset with diabetes for any length of time each additional 5 years of the disorder linked with a significant, relative 17% increase in the rate of incident heart failure. Incidence of heart failure rose even more sharply with added duration among those with a hemoglobin A1c of 7% or greater, compared with those with better glycemic control. And the rate of incident heart failure with reduced ejection fraction (HFrEF) roughly matched the rate of incident heart failure with preserved ejection fraction (HFpEF).
The study dataset included 9,734 adults enrolled into the Atherosclerosis Risk in Communities (ARIC) study, and during a median follow-up of 22.5 years they had nearly 2,000 episodes of either hospitalization or death secondary to incident heart failure. This included 617 (31%) events involving HFpEF, 495 events (25%) involving HFrEF, and 876 unclassified heart failure events.
The cohort averaged 63 years of age; 58% were women, 23% were Black, and 77% were White (the study design excluded people with other racial and ethnic backgrounds). The study design also excluded people with a history of heart failure or coronary artery disease, as well as those diagnosed with diabetes prior to age 18 resulting in a study group that presumably mostly had type 2 diabetes when diabetes was present. The report provided no data on the specific numbers of patients with type 1 or type 2 diabetes.
“It’s not surprising that a longer duration of diabetes is associated with heart failure, but the etiology remains problematic,” commented Robert H. Eckel, MD, an endocrinologist at the University of Colorado at Denver, Aurora. “The impact of diabetes on incident heart failure is not well know, particularly duration of diabetes,” although disorders often found in patients with diabetes, such as hypertension and diabetic cardiomyopathy, likely have roles in causing heart failure, he said.
Diabetes duration may signal need for an SGLT2 inhibitor
“With emerging novel treatments like the SGLT2 [sodium-glucose cotransporter 2] inhibitors for preventing heart failure hospitalizations and deaths in patients with type 2 diabetes, this is a timely analysis,” Dr. Eckel said in an interview.
“There is no question that with increased duration of type 2 diabetes” the need for an agent from the SGLT2-inhibitor class increases. Although, because of the proven protection these drugs give against heart failure events and progression of chronic kidney disease, treatment with this drug class should start early in patients with type 2 diabetes, he added.
Dr. Echouffo-Tcheugui and his coauthors agreed, citing two important clinical take-aways from their findings:
First, interventions that delay the onset of diabetes may potentially reduce incident heart failure; second, patients with diabetes might benefit from cardioprotective treatments such as SGLT2 inhibitors, the report said.
“Our observations suggest the potential prognostic relevance of diabetes duration in assessing heart failure,” the authors wrote. Integrating diabetes duration into heart failure risk estimation in people with diabetes “could help refine the selection of high-risk individuals who may derive the greatest absolute benefit from aggressive cardioprotective therapies such as SGLT2 inhibitors.”
The analysis also identified several other demographic and clinical factors that influenced the relative effect of diabetes duration. Longer duration was linked with higher rates of incident heart failure in women compared with men, in Blacks compared with Whites, in people younger than 65 compared with older people, in people with an A1c of 7% or higher, and in those with a body mass index of 30 kg/m2 or greater.
The ARIC study and the analyses run by Dr. Echouffo-Tcheugui and his coauthors received no commercial funding. Dr. Echouffo-Tcheugui and Dr. Eckel had no relevant disclosures.
FROM JACC HEART FAILURE
This is not the time to modify a HTN regimen
ILLUSTRATIVE CASE
A 67-year-old man with hypertension that is well controlled on hydrochlorothiazide 25 mg po daily was admitted to the family medicine inpatient service for community-acquired pneumonia requiring antibiotic therapy and oxygen support. Despite improvement in his overall condition, his blood pressure was consistently > 160/90 mm Hg during his hospitalization. He was treated with lisinopril 10 mg po daily in addition to his home medications, which helped achieve recommended blood pressure goals.
Prior to discharge, his blood pressure was noted to be 108/62 mm Hg. He asks if it is necessary to continue this new blood pressure medicine, as his home blood pressure readings had been within the goal set by his primary care physician. Should you continue this new antihypertensive agent at discharge?
Outpatient antihypertensive medication regimens are commonly intensified at hospital discharge in response to transient short-term elevations in blood pressure during inpatient encounters for noncardiac conditions.1,2 This is typically a reflexive response during a hospitalization, despite the unknown long-term, patient-oriented clinical outcomes. These short-term, in-hospital blood pressure elevations may be due to numerous temporary causes, such as stress/anxiety, a pain response, agitation, a medication adverse effect, or volume overload.3
The transition from inpatient to outpatient care is a high-risk period, especially for older adults, as functional status is generally worse at hospital discharge than prehospitalization baseline.4 To compound this problem, adverse drug reactions are a common cause of hospitalization for older adults. Changing blood pressure medications in response to acute physiologic changes during illness may contribute to patient harm. Although observational studies of adverse drug reactions related to blood pressure medications are numerous, researchers have only evaluated adverse drug reactions pertaining to hospital admissions.5-8 This study sought to evaluate the clinical outcomes associated with intensification of antihypertensive regimens at discharge among older adults.
STUDY SUMMARY
Increased risk of readmission, adverse events after intensification at discharge
This retrospective cohort study, which was conducted across multiple
Antihypertensive medication changes at discharge were evaluated using information pulled from VHA pharmacies, combined with clinical data merged from VHA and Medicare claims. Intensification was defined as either adding a new blood pressure medication or a dose increase of more than 20% on a previously prescribed antihypertensive medication. Patients were excluded if they were discharged with a secondary diagnosis that required modifications to a blood pressure medication (such as atrial fibrillation, acute coronary syndrome, or stroke), were hospitalized in the previous 30 days, were admitted from a skilled nursing facility, or received more than 20% of their care (including filling prescriptions) outside the VHA system.
Primary outcomes included hospital readmission or SAEs (falls, syncope, hypotension, serious electrolyte abnormalities, or acute kidney injury) within 30 days or having a cardiovascular event within 1 year of hospital discharge. Secondary outcomes included the change in systolic blood pressure (SBP) within 1 year after discharge. Propensity score matching was used as a balancing factor to create a matched-pairs cohort to compare those receiving blood pressure medication intensification at hospital discharge with those who did not.
Continue to: Intensification of the blood pressure...
Intensification of the blood pressure regimen at hospital discharge was associated with an increased risk in 30-day hospital readmission (hazard ratio [HR] = 1.23; 95% CI, 1.07–1.42; number needed to harm [NNH] = 27) and SAEs (HR = 1.41; 95% CI, 1.06–1.88; NNH = 63). There was no associated reduction in cardiovascular events (HR = 1.18; 95% CI, 0.99–1.40) or change in mean SBP within 1 year after hospital discharge in those who received intensification vs those who did not (mean BP, 134.7 vs 134.4 mm Hg; difference-in-differences estimate = 0.2 mm Hg; 95% CI, −2.0 to 2.4 mm Hg).
WHAT’S NEW
First study on outcomes related to HTN med changes at hospital discharge
This well-designed, retrospective cohort study provides important clinical data to help guide inpatient blood pressure management decisions for patients with noncardiac conditions. No clinical trials up to that time had assessed patient-oriented outcomes when antihypertensive medication regimens were intensified at hospital discharge.
CAVEATS
Study population: Primarily older men with noncardiac conditions
Selected populations benefit from intensive blood pressure control based on specific risk factors and medical conditions. In patients at high risk for cardiovascular disease, without a history of stroke or diabetes, intensive blood pressure control (SBP < 120 mm Hg) improves cardiovascular outcomes and overall survival compared with standard therapy (SBP < 140 mm Hg).9 This retrospective cohort study involved mainly elderly male patients with noncardiac conditions. The study also excluded patients with a secondary diagnosis requiring modifications to an antihypertensive regimen, such as atrial fibrillation, acute coronary syndrome, or cerebrovascular accident. Thus, the findings may not be applicable to these patient populations.
CHALLENGES TO IMPLEMENTATION
Clinicians will need to address individual needs
Physicians have to balance various antihypertensive management strategies, as competing medical specialty society guidelines recommend differing targets for optimal blood pressure control. Given the concern for medicolegal liability and potential harms of therapeutic inertia, inpatient physicians must consider whether hospitalization is the best time to alter medications for long-term outpatient blood pressure control. Finally, the decision to leave blood pressure management to outpatient physicians assumes the patient has a continuity relationship with a primary care medical home.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
2. Harris CM, Sridharan A, Landis R, et al. What happens to the medication regimens of older adults during and after an acute hospitalization? J Patient Saf. 2013;9:150-153.
3. Aung WM, Menon SV, Materson BJ. Management of hypertension in hospitalized patients. Hosp Pract (1995). 2015;43:101-106.
4. Covinsky KE, Palmer RM, Fortinsky RH, et al. Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc. 2003;51:451-458.
5. Omer HMRB, Hodson J, Pontefract SK, et al. Inpatient falls in older adults: a cohort study of antihypertensive prescribing pre- and post-fall. BMC Geriatr. 2018;18:58.
6. Alhawassi TM, Krass I, Pont LG. Antihypertensive-related adverse drug reactions among older hospitalized adults. Int J Clin Pharm. 2018;40:428-435.
7. Passarelli MCG, Jacob-Filho W, Figueras A. Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging. 2005;22:767-777.
8. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887-1898.
9. SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-2116. Published correction appears in N Engl J Med. 2017;377:2506.
ILLUSTRATIVE CASE
A 67-year-old man with hypertension that is well controlled on hydrochlorothiazide 25 mg po daily was admitted to the family medicine inpatient service for community-acquired pneumonia requiring antibiotic therapy and oxygen support. Despite improvement in his overall condition, his blood pressure was consistently > 160/90 mm Hg during his hospitalization. He was treated with lisinopril 10 mg po daily in addition to his home medications, which helped achieve recommended blood pressure goals.
Prior to discharge, his blood pressure was noted to be 108/62 mm Hg. He asks if it is necessary to continue this new blood pressure medicine, as his home blood pressure readings had been within the goal set by his primary care physician. Should you continue this new antihypertensive agent at discharge?
Outpatient antihypertensive medication regimens are commonly intensified at hospital discharge in response to transient short-term elevations in blood pressure during inpatient encounters for noncardiac conditions.1,2 This is typically a reflexive response during a hospitalization, despite the unknown long-term, patient-oriented clinical outcomes. These short-term, in-hospital blood pressure elevations may be due to numerous temporary causes, such as stress/anxiety, a pain response, agitation, a medication adverse effect, or volume overload.3
The transition from inpatient to outpatient care is a high-risk period, especially for older adults, as functional status is generally worse at hospital discharge than prehospitalization baseline.4 To compound this problem, adverse drug reactions are a common cause of hospitalization for older adults. Changing blood pressure medications in response to acute physiologic changes during illness may contribute to patient harm. Although observational studies of adverse drug reactions related to blood pressure medications are numerous, researchers have only evaluated adverse drug reactions pertaining to hospital admissions.5-8 This study sought to evaluate the clinical outcomes associated with intensification of antihypertensive regimens at discharge among older adults.
STUDY SUMMARY
Increased risk of readmission, adverse events after intensification at discharge
This retrospective cohort study, which was conducted across multiple
Antihypertensive medication changes at discharge were evaluated using information pulled from VHA pharmacies, combined with clinical data merged from VHA and Medicare claims. Intensification was defined as either adding a new blood pressure medication or a dose increase of more than 20% on a previously prescribed antihypertensive medication. Patients were excluded if they were discharged with a secondary diagnosis that required modifications to a blood pressure medication (such as atrial fibrillation, acute coronary syndrome, or stroke), were hospitalized in the previous 30 days, were admitted from a skilled nursing facility, or received more than 20% of their care (including filling prescriptions) outside the VHA system.
Primary outcomes included hospital readmission or SAEs (falls, syncope, hypotension, serious electrolyte abnormalities, or acute kidney injury) within 30 days or having a cardiovascular event within 1 year of hospital discharge. Secondary outcomes included the change in systolic blood pressure (SBP) within 1 year after discharge. Propensity score matching was used as a balancing factor to create a matched-pairs cohort to compare those receiving blood pressure medication intensification at hospital discharge with those who did not.
Continue to: Intensification of the blood pressure...
Intensification of the blood pressure regimen at hospital discharge was associated with an increased risk in 30-day hospital readmission (hazard ratio [HR] = 1.23; 95% CI, 1.07–1.42; number needed to harm [NNH] = 27) and SAEs (HR = 1.41; 95% CI, 1.06–1.88; NNH = 63). There was no associated reduction in cardiovascular events (HR = 1.18; 95% CI, 0.99–1.40) or change in mean SBP within 1 year after hospital discharge in those who received intensification vs those who did not (mean BP, 134.7 vs 134.4 mm Hg; difference-in-differences estimate = 0.2 mm Hg; 95% CI, −2.0 to 2.4 mm Hg).
WHAT’S NEW
First study on outcomes related to HTN med changes at hospital discharge
This well-designed, retrospective cohort study provides important clinical data to help guide inpatient blood pressure management decisions for patients with noncardiac conditions. No clinical trials up to that time had assessed patient-oriented outcomes when antihypertensive medication regimens were intensified at hospital discharge.
CAVEATS
Study population: Primarily older men with noncardiac conditions
Selected populations benefit from intensive blood pressure control based on specific risk factors and medical conditions. In patients at high risk for cardiovascular disease, without a history of stroke or diabetes, intensive blood pressure control (SBP < 120 mm Hg) improves cardiovascular outcomes and overall survival compared with standard therapy (SBP < 140 mm Hg).9 This retrospective cohort study involved mainly elderly male patients with noncardiac conditions. The study also excluded patients with a secondary diagnosis requiring modifications to an antihypertensive regimen, such as atrial fibrillation, acute coronary syndrome, or cerebrovascular accident. Thus, the findings may not be applicable to these patient populations.
CHALLENGES TO IMPLEMENTATION
Clinicians will need to address individual needs
Physicians have to balance various antihypertensive management strategies, as competing medical specialty society guidelines recommend differing targets for optimal blood pressure control. Given the concern for medicolegal liability and potential harms of therapeutic inertia, inpatient physicians must consider whether hospitalization is the best time to alter medications for long-term outpatient blood pressure control. Finally, the decision to leave blood pressure management to outpatient physicians assumes the patient has a continuity relationship with a primary care medical home.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 67-year-old man with hypertension that is well controlled on hydrochlorothiazide 25 mg po daily was admitted to the family medicine inpatient service for community-acquired pneumonia requiring antibiotic therapy and oxygen support. Despite improvement in his overall condition, his blood pressure was consistently > 160/90 mm Hg during his hospitalization. He was treated with lisinopril 10 mg po daily in addition to his home medications, which helped achieve recommended blood pressure goals.
Prior to discharge, his blood pressure was noted to be 108/62 mm Hg. He asks if it is necessary to continue this new blood pressure medicine, as his home blood pressure readings had been within the goal set by his primary care physician. Should you continue this new antihypertensive agent at discharge?
Outpatient antihypertensive medication regimens are commonly intensified at hospital discharge in response to transient short-term elevations in blood pressure during inpatient encounters for noncardiac conditions.1,2 This is typically a reflexive response during a hospitalization, despite the unknown long-term, patient-oriented clinical outcomes. These short-term, in-hospital blood pressure elevations may be due to numerous temporary causes, such as stress/anxiety, a pain response, agitation, a medication adverse effect, or volume overload.3
The transition from inpatient to outpatient care is a high-risk period, especially for older adults, as functional status is generally worse at hospital discharge than prehospitalization baseline.4 To compound this problem, adverse drug reactions are a common cause of hospitalization for older adults. Changing blood pressure medications in response to acute physiologic changes during illness may contribute to patient harm. Although observational studies of adverse drug reactions related to blood pressure medications are numerous, researchers have only evaluated adverse drug reactions pertaining to hospital admissions.5-8 This study sought to evaluate the clinical outcomes associated with intensification of antihypertensive regimens at discharge among older adults.
STUDY SUMMARY
Increased risk of readmission, adverse events after intensification at discharge
This retrospective cohort study, which was conducted across multiple
Antihypertensive medication changes at discharge were evaluated using information pulled from VHA pharmacies, combined with clinical data merged from VHA and Medicare claims. Intensification was defined as either adding a new blood pressure medication or a dose increase of more than 20% on a previously prescribed antihypertensive medication. Patients were excluded if they were discharged with a secondary diagnosis that required modifications to a blood pressure medication (such as atrial fibrillation, acute coronary syndrome, or stroke), were hospitalized in the previous 30 days, were admitted from a skilled nursing facility, or received more than 20% of their care (including filling prescriptions) outside the VHA system.
Primary outcomes included hospital readmission or SAEs (falls, syncope, hypotension, serious electrolyte abnormalities, or acute kidney injury) within 30 days or having a cardiovascular event within 1 year of hospital discharge. Secondary outcomes included the change in systolic blood pressure (SBP) within 1 year after discharge. Propensity score matching was used as a balancing factor to create a matched-pairs cohort to compare those receiving blood pressure medication intensification at hospital discharge with those who did not.
Continue to: Intensification of the blood pressure...
Intensification of the blood pressure regimen at hospital discharge was associated with an increased risk in 30-day hospital readmission (hazard ratio [HR] = 1.23; 95% CI, 1.07–1.42; number needed to harm [NNH] = 27) and SAEs (HR = 1.41; 95% CI, 1.06–1.88; NNH = 63). There was no associated reduction in cardiovascular events (HR = 1.18; 95% CI, 0.99–1.40) or change in mean SBP within 1 year after hospital discharge in those who received intensification vs those who did not (mean BP, 134.7 vs 134.4 mm Hg; difference-in-differences estimate = 0.2 mm Hg; 95% CI, −2.0 to 2.4 mm Hg).
WHAT’S NEW
First study on outcomes related to HTN med changes at hospital discharge
This well-designed, retrospective cohort study provides important clinical data to help guide inpatient blood pressure management decisions for patients with noncardiac conditions. No clinical trials up to that time had assessed patient-oriented outcomes when antihypertensive medication regimens were intensified at hospital discharge.
CAVEATS
Study population: Primarily older men with noncardiac conditions
Selected populations benefit from intensive blood pressure control based on specific risk factors and medical conditions. In patients at high risk for cardiovascular disease, without a history of stroke or diabetes, intensive blood pressure control (SBP < 120 mm Hg) improves cardiovascular outcomes and overall survival compared with standard therapy (SBP < 140 mm Hg).9 This retrospective cohort study involved mainly elderly male patients with noncardiac conditions. The study also excluded patients with a secondary diagnosis requiring modifications to an antihypertensive regimen, such as atrial fibrillation, acute coronary syndrome, or cerebrovascular accident. Thus, the findings may not be applicable to these patient populations.
CHALLENGES TO IMPLEMENTATION
Clinicians will need to address individual needs
Physicians have to balance various antihypertensive management strategies, as competing medical specialty society guidelines recommend differing targets for optimal blood pressure control. Given the concern for medicolegal liability and potential harms of therapeutic inertia, inpatient physicians must consider whether hospitalization is the best time to alter medications for long-term outpatient blood pressure control. Finally, the decision to leave blood pressure management to outpatient physicians assumes the patient has a continuity relationship with a primary care medical home.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
2. Harris CM, Sridharan A, Landis R, et al. What happens to the medication regimens of older adults during and after an acute hospitalization? J Patient Saf. 2013;9:150-153.
3. Aung WM, Menon SV, Materson BJ. Management of hypertension in hospitalized patients. Hosp Pract (1995). 2015;43:101-106.
4. Covinsky KE, Palmer RM, Fortinsky RH, et al. Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc. 2003;51:451-458.
5. Omer HMRB, Hodson J, Pontefract SK, et al. Inpatient falls in older adults: a cohort study of antihypertensive prescribing pre- and post-fall. BMC Geriatr. 2018;18:58.
6. Alhawassi TM, Krass I, Pont LG. Antihypertensive-related adverse drug reactions among older hospitalized adults. Int J Clin Pharm. 2018;40:428-435.
7. Passarelli MCG, Jacob-Filho W, Figueras A. Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging. 2005;22:767-777.
8. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887-1898.
9. SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-2116. Published correction appears in N Engl J Med. 2017;377:2506.
1. Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
2. Harris CM, Sridharan A, Landis R, et al. What happens to the medication regimens of older adults during and after an acute hospitalization? J Patient Saf. 2013;9:150-153.
3. Aung WM, Menon SV, Materson BJ. Management of hypertension in hospitalized patients. Hosp Pract (1995). 2015;43:101-106.
4. Covinsky KE, Palmer RM, Fortinsky RH, et al. Loss of independence in activities of daily living in older adults hospitalized with medical illnesses: increased vulnerability with age. J Am Geriatr Soc. 2003;51:451-458.
5. Omer HMRB, Hodson J, Pontefract SK, et al. Inpatient falls in older adults: a cohort study of antihypertensive prescribing pre- and post-fall. BMC Geriatr. 2018;18:58.
6. Alhawassi TM, Krass I, Pont LG. Antihypertensive-related adverse drug reactions among older hospitalized adults. Int J Clin Pharm. 2018;40:428-435.
7. Passarelli MCG, Jacob-Filho W, Figueras A. Adverse drug reactions in an elderly hospitalised population: inappropriate prescription is a leading cause. Drugs Aging. 2005;22:767-777.
8. Beckett NS, Peters R, Fletcher AE, et al; HYVET Study Group. Treatment of hypertension in patients 80 years of age or older. N Engl J Med. 2008;358:1887-1898.
9. SPRINT Research Group; Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood-pressure control. N Engl J Med. 2015;373:2103-2116. Published correction appears in N Engl J Med. 2017;377:2506.
PRACTICE CHANGER
Avoid intensifying antihypertensive medication regimens at hospital discharge in older adults; making such changes increases the risk of serious adverse events (SAEs) and hospital readmission within 30 days without reducing the risk of serious cardiovascular events at 1 year post discharge.
STRENGTH OF RECOMMENDATION
B: Based on a large retrospective cohort study evaluating patient-oriented outcomes.1
Anderson TS, Jing B, Auerbach A, et al. Clinical outcomes after intensifying antihypertensive medication regimens among older adults at hospital discharge. JAMA Intern Med. 2019;179:1528-1536.
Dissolving pacemaker impressive in early research
A fully implantable, bioresorbable pacemaker has been developed that’s capable of sustaining heart rhythms in animal and human donor hearts before disappearing over 5-7 weeks.
Temporary pacing devices are frequently used after cardiac surgery but rely on bulky external generators and transcutaneous pacing leads that run the risk of becoming infected or dislodged and can damage the heart when removed if they’re enveloped in fibrotic tissue.
The experimental device is thin, powered without leads or batteries, and made of water-soluble, biocompatible materials, thereby bypassing many of the disadvantages of conventional temporary pacing devices, according to John A. Rogers, PhD, who led the device’s development and directs the Querrey Simpson Institute for Bioelectronics at Northwestern University in Chicago.
“The total material load on the body is very minimal,” he said in an interview. “The amount of silicon and magnesium in a multivitamin tablet is about 3,000 times more than the amount of those materials in our electronics. So you can think of them as a very tiny vitamin pill, in a sense, but configured with electronic functionality.”
Dr. Rogers and his team have a reputation for innovation in bioelectronic medicine, having recently constructed transient wireless devices to accelerate neuroregeneration associated with damaged peripheral nerves, to monitor critically ill neonates, and to detect early signs and symptoms associated with COVID-19.
Shortly after Dr. Rogers joined Northwestern, Rishi Arora, MD, a cardiac electrophysiologist and professor of medicine at Northwestern, reached out to discuss how they could leverage wireless electronics for patients needing temporary pacing.
“It was a natural marriage,” Dr. Arora said in an interview. “Part of the reason to go into the heart was because the cardiology group here at Northwestern, especially on the electrophysiology side, has been very involved in translational research, and John also had a very strong collaboration before he came here with Igor Efimov, [PhD, of George Washington University, Washington], a giant in the field in terms of heart rhythm research.”
Dr. Arora noted that the incidence of temporary pacing after cardiac surgery is at least 10% but can reach 20%. Current devices work well in most patients, but temporary pacing with epicardial wires can cause complications and, typically, work well only for a few days after cardiac surgery. Clinically, though, several patients need postoperative pacing support for 1-2 weeks.
“So if something like this were available where you could tack it onto the surface and forget it for a week or 10 days or 2 weeks, you’d be doing those 20% of patients a huge service,” he said.
Bioresorbable scaffold déjà vu?
The philosophy of “leave nothing behind” is nothing new in cardiology, with bioresorbable vascular scaffolds (BVS) gaining initial support as a potential solution to neoatherosclerosis and late-stent thrombosis in permanent metal stents. Failure to show advantages, and safety concerns such as in-scaffold thrombosis, however, led Abbott to stop global sales of the first approved BVS and Boston Scientific to halt its BVS program in 2017.
The wireless pacemaker, however, is an electrical device, not a mechanical one, observed Dr. Rogers. “The fact that it’s not in the bloodstream greatly lowers risks and, as I mentioned before, everything is super thin, low-mass quantities of materials. So, I guess there’s a relationship there, but it’s different in a couple of very important ways.”
As Dr. Rogers, Dr. Arora, Dr. Efimov, and colleagues recently reported in Nature Biotechnology, the electronic part of the pacemaker contains three layers: A loop antenna with a bilayer tungsten-coated magnesium inductive coil, a radiofrequency PIN diode based on a monocrystalline silicon nanomembrane, and a poly (lactide-co-glycolide) (PLGA) dielectric interlayer.
The electronic components rest between two encapsulation layers of PLGA to isolate the active materials from the surrounding biofluids during implantation, and connect to a pair of flexible extension electrodes that deliver the electrical stimuli to a contact pad sutured onto the heart. The entire system is about 16 mm in width and 15 mm in length, and weighs in at about 0.3 g.
The pacemaker receives power and control commands through a wireless inductive power transfer – the same technology used in implanted medical devices, smartphones, and radio-frequency identification tags – between the receiver coil in the device and a wand-shaped, external transmission coil placed on top of or within a few inches of the heart.
“Right now we’re almost at 15 inches, which I think is a very respectable distance for this particular piece of hardware, and clinically very doable,” observed Dr. Arora.
Competing considerations
Testing thus far shows effective ventricular capture across a range of frequencies in mouse and rabbit hearts and successful pacing and activation of human cardiac tissue.
In vivo tests in dogs also suggest that the system can “achieve the power necessary for operation of bioresorbable pacemakers in adult human patients,” the authors say.
Electrodes placed on the dogs’ legs showed a change in ECG signals from a narrow QRS complex (consistent with a normal rate sinus rhythm of 350-400 bpm) to a widened QRS complex with a shortened R-R interval (consistent with a paced rhythm of 400-450 bpm) – indicating successful ventricular capture.
The device successfully paced the dogs through postoperative day 4 but couldn’t provide enough energy to capture the ventricular myocardium on day 5 and failed to pace the heart on day 6, even when transmitting voltages were increased from 1 Vpp to more than 10 Vpp.
Dr. Rogers pointed out that a transient device of theirs that uses very thin films of silica provides stable intracranial pressure monitoring for traumatic brain injury recovery for 3 weeks before dissolving. The problem with the polymers used as encapsulating layers in the pacemaker is that even if they haven’t completely dissolved, there’s a finite rate of water permeation through the film.
“It turns out that’s what’s become the limiting factor, rather than the chemistry of bioresorption,” he said. “So, what we’re seeing with these devices beginning to degrade electrically in terms of performance around 5-6 days is due to that water permeation.”
Although it is not part of the current study, there’s no reason thin silica layers couldn’t be incorporated into the pacemaker to make it less water permeable, Dr. Rogers said. Still, this will have to be weighed against the competing consideration of stable operating life.
The researchers specifically chose materials that would naturally bioresorb via hydrolysis and metabolic action in the body. PLGA degrades into glycolic and lactic acid, the tungsten-coated magnesium inductive coil into Wox and Mg(OH)2, and the silicon nanomembrane radiofrequency PIN diode into Si(OH)4.
CT imaging in rat models shows the device is enveloped in fibrotic tissue and completely decouples from the heart at 4 weeks, while images of explanted devices suggest the pacemaker largely dissolves within 3 weeks and the remaining residues disappear after 12 weeks.
The researchers have started an investigational device exemption process to allow the device to be used in clinical trials, and they plan to dig deeper into the potential for fragments to form at various stages of resorption, which some imaging suggests may occur.
“Because these devices are made out of pure materials and they’re in a heterogeneous environment, both mechanically and biomechanically, the devices don’t resorb in a perfectly uniform way and, as a result, at the tail end of the process you can end up with small fragments that eventually bioresorb, but before they’re gone, they are potentially mobile within the body cavity,” Dr. Rogers said.
“We feel that because the devices aren’t in the bloodstream, the risk associated with those fragments is probably manageable but at the same time, these are the sorts of details that must be thoroughly addressed before trials in humans,” he said, adding that one solution, if needed, would be to encapsulate the entire device in a thin bioresorbable hydrogel as a containment vehicle.
Dr. Arora said they hope the pacemaker “will make patients’ lives a lot easier in the postoperative setting but, even there, I think one must remember current pacing technology in this setting is actually very good. So there’s a word of caution not to get ahead of ourselves.”
Looking forward, the excitement of this approach is not only in the immediate postop setting but in the transvenous setting, he said. “If we can get to the point where we can actually do this transvenously, that opens up a huge window of opportunity because there we’re talking about post-TAVR [transcatheter aortic valve replacement], post–myocardial infarction, etc.”
Currently, temporary transvenous pacing can be quite unreliable because of a high risk of dislodgement and infection – much higher than for surgical pacing wires, he noted.
“In terms of translatability to larger numbers of patients, the value would be huge. But again, a lot needs to be done before we can get there. But if it can get to that point, then I think you have a real therapy that could potentially be transformative,” Dr. Arora said.
Dr. Rogers reported support from the Leducq Foundation projects RHYTHM and ROI-HL121270. Dr. Arora has disclosed no relevant financial relationships. Coauthor disclosures are listed in the original article.
A version of this article first appeared on Medscape.com.
A fully implantable, bioresorbable pacemaker has been developed that’s capable of sustaining heart rhythms in animal and human donor hearts before disappearing over 5-7 weeks.
Temporary pacing devices are frequently used after cardiac surgery but rely on bulky external generators and transcutaneous pacing leads that run the risk of becoming infected or dislodged and can damage the heart when removed if they’re enveloped in fibrotic tissue.
The experimental device is thin, powered without leads or batteries, and made of water-soluble, biocompatible materials, thereby bypassing many of the disadvantages of conventional temporary pacing devices, according to John A. Rogers, PhD, who led the device’s development and directs the Querrey Simpson Institute for Bioelectronics at Northwestern University in Chicago.
“The total material load on the body is very minimal,” he said in an interview. “The amount of silicon and magnesium in a multivitamin tablet is about 3,000 times more than the amount of those materials in our electronics. So you can think of them as a very tiny vitamin pill, in a sense, but configured with electronic functionality.”
Dr. Rogers and his team have a reputation for innovation in bioelectronic medicine, having recently constructed transient wireless devices to accelerate neuroregeneration associated with damaged peripheral nerves, to monitor critically ill neonates, and to detect early signs and symptoms associated with COVID-19.
Shortly after Dr. Rogers joined Northwestern, Rishi Arora, MD, a cardiac electrophysiologist and professor of medicine at Northwestern, reached out to discuss how they could leverage wireless electronics for patients needing temporary pacing.
“It was a natural marriage,” Dr. Arora said in an interview. “Part of the reason to go into the heart was because the cardiology group here at Northwestern, especially on the electrophysiology side, has been very involved in translational research, and John also had a very strong collaboration before he came here with Igor Efimov, [PhD, of George Washington University, Washington], a giant in the field in terms of heart rhythm research.”
Dr. Arora noted that the incidence of temporary pacing after cardiac surgery is at least 10% but can reach 20%. Current devices work well in most patients, but temporary pacing with epicardial wires can cause complications and, typically, work well only for a few days after cardiac surgery. Clinically, though, several patients need postoperative pacing support for 1-2 weeks.
“So if something like this were available where you could tack it onto the surface and forget it for a week or 10 days or 2 weeks, you’d be doing those 20% of patients a huge service,” he said.
Bioresorbable scaffold déjà vu?
The philosophy of “leave nothing behind” is nothing new in cardiology, with bioresorbable vascular scaffolds (BVS) gaining initial support as a potential solution to neoatherosclerosis and late-stent thrombosis in permanent metal stents. Failure to show advantages, and safety concerns such as in-scaffold thrombosis, however, led Abbott to stop global sales of the first approved BVS and Boston Scientific to halt its BVS program in 2017.
The wireless pacemaker, however, is an electrical device, not a mechanical one, observed Dr. Rogers. “The fact that it’s not in the bloodstream greatly lowers risks and, as I mentioned before, everything is super thin, low-mass quantities of materials. So, I guess there’s a relationship there, but it’s different in a couple of very important ways.”
As Dr. Rogers, Dr. Arora, Dr. Efimov, and colleagues recently reported in Nature Biotechnology, the electronic part of the pacemaker contains three layers: A loop antenna with a bilayer tungsten-coated magnesium inductive coil, a radiofrequency PIN diode based on a monocrystalline silicon nanomembrane, and a poly (lactide-co-glycolide) (PLGA) dielectric interlayer.
The electronic components rest between two encapsulation layers of PLGA to isolate the active materials from the surrounding biofluids during implantation, and connect to a pair of flexible extension electrodes that deliver the electrical stimuli to a contact pad sutured onto the heart. The entire system is about 16 mm in width and 15 mm in length, and weighs in at about 0.3 g.
The pacemaker receives power and control commands through a wireless inductive power transfer – the same technology used in implanted medical devices, smartphones, and radio-frequency identification tags – between the receiver coil in the device and a wand-shaped, external transmission coil placed on top of or within a few inches of the heart.
“Right now we’re almost at 15 inches, which I think is a very respectable distance for this particular piece of hardware, and clinically very doable,” observed Dr. Arora.
Competing considerations
Testing thus far shows effective ventricular capture across a range of frequencies in mouse and rabbit hearts and successful pacing and activation of human cardiac tissue.
In vivo tests in dogs also suggest that the system can “achieve the power necessary for operation of bioresorbable pacemakers in adult human patients,” the authors say.
Electrodes placed on the dogs’ legs showed a change in ECG signals from a narrow QRS complex (consistent with a normal rate sinus rhythm of 350-400 bpm) to a widened QRS complex with a shortened R-R interval (consistent with a paced rhythm of 400-450 bpm) – indicating successful ventricular capture.
The device successfully paced the dogs through postoperative day 4 but couldn’t provide enough energy to capture the ventricular myocardium on day 5 and failed to pace the heart on day 6, even when transmitting voltages were increased from 1 Vpp to more than 10 Vpp.
Dr. Rogers pointed out that a transient device of theirs that uses very thin films of silica provides stable intracranial pressure monitoring for traumatic brain injury recovery for 3 weeks before dissolving. The problem with the polymers used as encapsulating layers in the pacemaker is that even if they haven’t completely dissolved, there’s a finite rate of water permeation through the film.
“It turns out that’s what’s become the limiting factor, rather than the chemistry of bioresorption,” he said. “So, what we’re seeing with these devices beginning to degrade electrically in terms of performance around 5-6 days is due to that water permeation.”
Although it is not part of the current study, there’s no reason thin silica layers couldn’t be incorporated into the pacemaker to make it less water permeable, Dr. Rogers said. Still, this will have to be weighed against the competing consideration of stable operating life.
The researchers specifically chose materials that would naturally bioresorb via hydrolysis and metabolic action in the body. PLGA degrades into glycolic and lactic acid, the tungsten-coated magnesium inductive coil into Wox and Mg(OH)2, and the silicon nanomembrane radiofrequency PIN diode into Si(OH)4.
CT imaging in rat models shows the device is enveloped in fibrotic tissue and completely decouples from the heart at 4 weeks, while images of explanted devices suggest the pacemaker largely dissolves within 3 weeks and the remaining residues disappear after 12 weeks.
The researchers have started an investigational device exemption process to allow the device to be used in clinical trials, and they plan to dig deeper into the potential for fragments to form at various stages of resorption, which some imaging suggests may occur.
“Because these devices are made out of pure materials and they’re in a heterogeneous environment, both mechanically and biomechanically, the devices don’t resorb in a perfectly uniform way and, as a result, at the tail end of the process you can end up with small fragments that eventually bioresorb, but before they’re gone, they are potentially mobile within the body cavity,” Dr. Rogers said.
“We feel that because the devices aren’t in the bloodstream, the risk associated with those fragments is probably manageable but at the same time, these are the sorts of details that must be thoroughly addressed before trials in humans,” he said, adding that one solution, if needed, would be to encapsulate the entire device in a thin bioresorbable hydrogel as a containment vehicle.
Dr. Arora said they hope the pacemaker “will make patients’ lives a lot easier in the postoperative setting but, even there, I think one must remember current pacing technology in this setting is actually very good. So there’s a word of caution not to get ahead of ourselves.”
Looking forward, the excitement of this approach is not only in the immediate postop setting but in the transvenous setting, he said. “If we can get to the point where we can actually do this transvenously, that opens up a huge window of opportunity because there we’re talking about post-TAVR [transcatheter aortic valve replacement], post–myocardial infarction, etc.”
Currently, temporary transvenous pacing can be quite unreliable because of a high risk of dislodgement and infection – much higher than for surgical pacing wires, he noted.
“In terms of translatability to larger numbers of patients, the value would be huge. But again, a lot needs to be done before we can get there. But if it can get to that point, then I think you have a real therapy that could potentially be transformative,” Dr. Arora said.
Dr. Rogers reported support from the Leducq Foundation projects RHYTHM and ROI-HL121270. Dr. Arora has disclosed no relevant financial relationships. Coauthor disclosures are listed in the original article.
A version of this article first appeared on Medscape.com.
A fully implantable, bioresorbable pacemaker has been developed that’s capable of sustaining heart rhythms in animal and human donor hearts before disappearing over 5-7 weeks.
Temporary pacing devices are frequently used after cardiac surgery but rely on bulky external generators and transcutaneous pacing leads that run the risk of becoming infected or dislodged and can damage the heart when removed if they’re enveloped in fibrotic tissue.
The experimental device is thin, powered without leads or batteries, and made of water-soluble, biocompatible materials, thereby bypassing many of the disadvantages of conventional temporary pacing devices, according to John A. Rogers, PhD, who led the device’s development and directs the Querrey Simpson Institute for Bioelectronics at Northwestern University in Chicago.
“The total material load on the body is very minimal,” he said in an interview. “The amount of silicon and magnesium in a multivitamin tablet is about 3,000 times more than the amount of those materials in our electronics. So you can think of them as a very tiny vitamin pill, in a sense, but configured with electronic functionality.”
Dr. Rogers and his team have a reputation for innovation in bioelectronic medicine, having recently constructed transient wireless devices to accelerate neuroregeneration associated with damaged peripheral nerves, to monitor critically ill neonates, and to detect early signs and symptoms associated with COVID-19.
Shortly after Dr. Rogers joined Northwestern, Rishi Arora, MD, a cardiac electrophysiologist and professor of medicine at Northwestern, reached out to discuss how they could leverage wireless electronics for patients needing temporary pacing.
“It was a natural marriage,” Dr. Arora said in an interview. “Part of the reason to go into the heart was because the cardiology group here at Northwestern, especially on the electrophysiology side, has been very involved in translational research, and John also had a very strong collaboration before he came here with Igor Efimov, [PhD, of George Washington University, Washington], a giant in the field in terms of heart rhythm research.”
Dr. Arora noted that the incidence of temporary pacing after cardiac surgery is at least 10% but can reach 20%. Current devices work well in most patients, but temporary pacing with epicardial wires can cause complications and, typically, work well only for a few days after cardiac surgery. Clinically, though, several patients need postoperative pacing support for 1-2 weeks.
“So if something like this were available where you could tack it onto the surface and forget it for a week or 10 days or 2 weeks, you’d be doing those 20% of patients a huge service,” he said.
Bioresorbable scaffold déjà vu?
The philosophy of “leave nothing behind” is nothing new in cardiology, with bioresorbable vascular scaffolds (BVS) gaining initial support as a potential solution to neoatherosclerosis and late-stent thrombosis in permanent metal stents. Failure to show advantages, and safety concerns such as in-scaffold thrombosis, however, led Abbott to stop global sales of the first approved BVS and Boston Scientific to halt its BVS program in 2017.
The wireless pacemaker, however, is an electrical device, not a mechanical one, observed Dr. Rogers. “The fact that it’s not in the bloodstream greatly lowers risks and, as I mentioned before, everything is super thin, low-mass quantities of materials. So, I guess there’s a relationship there, but it’s different in a couple of very important ways.”
As Dr. Rogers, Dr. Arora, Dr. Efimov, and colleagues recently reported in Nature Biotechnology, the electronic part of the pacemaker contains three layers: A loop antenna with a bilayer tungsten-coated magnesium inductive coil, a radiofrequency PIN diode based on a monocrystalline silicon nanomembrane, and a poly (lactide-co-glycolide) (PLGA) dielectric interlayer.
The electronic components rest between two encapsulation layers of PLGA to isolate the active materials from the surrounding biofluids during implantation, and connect to a pair of flexible extension electrodes that deliver the electrical stimuli to a contact pad sutured onto the heart. The entire system is about 16 mm in width and 15 mm in length, and weighs in at about 0.3 g.
The pacemaker receives power and control commands through a wireless inductive power transfer – the same technology used in implanted medical devices, smartphones, and radio-frequency identification tags – between the receiver coil in the device and a wand-shaped, external transmission coil placed on top of or within a few inches of the heart.
“Right now we’re almost at 15 inches, which I think is a very respectable distance for this particular piece of hardware, and clinically very doable,” observed Dr. Arora.
Competing considerations
Testing thus far shows effective ventricular capture across a range of frequencies in mouse and rabbit hearts and successful pacing and activation of human cardiac tissue.
In vivo tests in dogs also suggest that the system can “achieve the power necessary for operation of bioresorbable pacemakers in adult human patients,” the authors say.
Electrodes placed on the dogs’ legs showed a change in ECG signals from a narrow QRS complex (consistent with a normal rate sinus rhythm of 350-400 bpm) to a widened QRS complex with a shortened R-R interval (consistent with a paced rhythm of 400-450 bpm) – indicating successful ventricular capture.
The device successfully paced the dogs through postoperative day 4 but couldn’t provide enough energy to capture the ventricular myocardium on day 5 and failed to pace the heart on day 6, even when transmitting voltages were increased from 1 Vpp to more than 10 Vpp.
Dr. Rogers pointed out that a transient device of theirs that uses very thin films of silica provides stable intracranial pressure monitoring for traumatic brain injury recovery for 3 weeks before dissolving. The problem with the polymers used as encapsulating layers in the pacemaker is that even if they haven’t completely dissolved, there’s a finite rate of water permeation through the film.
“It turns out that’s what’s become the limiting factor, rather than the chemistry of bioresorption,” he said. “So, what we’re seeing with these devices beginning to degrade electrically in terms of performance around 5-6 days is due to that water permeation.”
Although it is not part of the current study, there’s no reason thin silica layers couldn’t be incorporated into the pacemaker to make it less water permeable, Dr. Rogers said. Still, this will have to be weighed against the competing consideration of stable operating life.
The researchers specifically chose materials that would naturally bioresorb via hydrolysis and metabolic action in the body. PLGA degrades into glycolic and lactic acid, the tungsten-coated magnesium inductive coil into Wox and Mg(OH)2, and the silicon nanomembrane radiofrequency PIN diode into Si(OH)4.
CT imaging in rat models shows the device is enveloped in fibrotic tissue and completely decouples from the heart at 4 weeks, while images of explanted devices suggest the pacemaker largely dissolves within 3 weeks and the remaining residues disappear after 12 weeks.
The researchers have started an investigational device exemption process to allow the device to be used in clinical trials, and they plan to dig deeper into the potential for fragments to form at various stages of resorption, which some imaging suggests may occur.
“Because these devices are made out of pure materials and they’re in a heterogeneous environment, both mechanically and biomechanically, the devices don’t resorb in a perfectly uniform way and, as a result, at the tail end of the process you can end up with small fragments that eventually bioresorb, but before they’re gone, they are potentially mobile within the body cavity,” Dr. Rogers said.
“We feel that because the devices aren’t in the bloodstream, the risk associated with those fragments is probably manageable but at the same time, these are the sorts of details that must be thoroughly addressed before trials in humans,” he said, adding that one solution, if needed, would be to encapsulate the entire device in a thin bioresorbable hydrogel as a containment vehicle.
Dr. Arora said they hope the pacemaker “will make patients’ lives a lot easier in the postoperative setting but, even there, I think one must remember current pacing technology in this setting is actually very good. So there’s a word of caution not to get ahead of ourselves.”
Looking forward, the excitement of this approach is not only in the immediate postop setting but in the transvenous setting, he said. “If we can get to the point where we can actually do this transvenously, that opens up a huge window of opportunity because there we’re talking about post-TAVR [transcatheter aortic valve replacement], post–myocardial infarction, etc.”
Currently, temporary transvenous pacing can be quite unreliable because of a high risk of dislodgement and infection – much higher than for surgical pacing wires, he noted.
“In terms of translatability to larger numbers of patients, the value would be huge. But again, a lot needs to be done before we can get there. But if it can get to that point, then I think you have a real therapy that could potentially be transformative,” Dr. Arora said.
Dr. Rogers reported support from the Leducq Foundation projects RHYTHM and ROI-HL121270. Dr. Arora has disclosed no relevant financial relationships. Coauthor disclosures are listed in the original article.
A version of this article first appeared on Medscape.com.
Younger adults with HIV have higher CVD risk but low ASCVD scores
People age 40 and younger living with HIV have a higher risk for heart disease than even their over-40 peers living with HIV – and that risk was 54% higher than the general public.
And this was among people without traditional heart disease risks, such as smoking and obesity.
“What’s surprising is that not only do we see that, yes, they do have increased risk, but this is after controlling for all of that – which means the mechanism underlying this risk,” said Tiffany Gooden, MPH and a PhD candidate at the University of Birmingham, England, who presented the data at the 11th International AIDS Society Conference on HIV Science (IAS 2021).
“If we’re using a non–HIV-validated assessment tool, you should always know that there could be a risk that you are under-recognizing,” she added.
Right now, there’s not a lot to aid clinicians in ferreting out this increased risk. Traditional cardiovascular risk assessment tools, like Framingham risk scores and atherosclerotic cardiovascular disease risk score from the American College of Cardiology, have been found to overlook the real risk of cardiovascular disease in people living with HIV. Plus, most guidelines, including those from the British HIV Medical Association and the American College of Cardiology, primarily focus screening on people 40 or older.
Ms. Gooden’s study drew data from The Health Improvement Network (THIN) database, which combines data from 800 primary care practices in the United Kingdom. Looking at data between January 2000 and January 2020, the investigators compared each person living with HIV with four peers not living with HIV, matched for age, gender, and practice. In total, 9,233 people living with HIV and 35,721 people without HIV were included in the analysis. Median age of participants was 41 years in people living with HIV and 40.4 years in people without HIV. About 35% of participants in both arms were women, and a greater proportion of participants living with HIV were Black, accounting for 22.5% of people living with HIV, versus 3.8% of the general population. Fewer people living with HIV were overweight or obese compared to people without HIV.
Researchers then tracked participants over time to identify the incidence of heart attack, stroke, peripheral vascular disease, and heart failure, as well as common risk factors for heart problems, such as high blood pressure, type 2 diabetes, chronic kidney disease, atrial fibrillation, and use of a lipid-lowering drug such as a statin.
The investigators then sectioned the data on heart disease risk by decade – 2000-2009 and 2010-2019 – in order to separate the potential impact of antiretroviral treatment (ART) drugs, from early combinations that have been associated with cardiovascular disease, to current drugs that are less likely to have that effect.
Overall risk for any kind of cardiovascular disease was 54% higher among people living with HIV of any age, compared to their age- and risk-matched peers. And when they broke the data down by age, they found that people younger than 40 had nearly twice the risk for any heart disease as their HIV-negative peers, which was a numerically higher risk than for people older than 40 – though not significantly so.
People living with HIV also had a 49% increased risk for stroke and a 59% increased risk for ischemic heart disease but no increased risk for peripheral vascular disease, heart failure, or heart attack. But the confidence intervals here were wide, “which may indicate lack of power and therefore not be conclusive,” Ms. Gooden said.
People living with HIV also had a 37% increased risk for hypertension, were 96% more likely to be prescribed lipid-lowering drugs, 2.4-times more likely to have chronic kidney disease, and 2.68-times more likely to experience all-cause mortality. The study couldn’t account for the type of HIV medications people living with the virus used, their viral load, or their CD4 counts – all of which have been found in previous studies to contribute to heart disease in people with HIV.
“That was the biggest limitation of our study,” Ms. Gooden said in an interview. “The fact that the risk of cardiovascular disease remains the same in the [first decade] and the later decade goes to show that even if antiretroviral therapies contributed to that … now or 20 years ago, it’s still not the entire reason for the risk.”
Steven Grinspoon, MD, of Harvard Medical School, Boston, is the lead author on the REPRIEVE trial, now testing statins as a treatment for people like those in this study. He told this news organization that this large analysis had one of the youngest cohorts of people living with HIV he’d seen to explore these issues. Additionally, it backs up what the team recently reported in the Journal of the American Medical Association – that plaque was present in 49% of 755 people living with HIV, despite having risk scores for cardiovascular disease in the normal range. This was as true for people younger than 40 as those older than 40.
For primary care clinicians, the message is that even relatively young people with HIV should be counseled early and often about amending traditional risk factors, while we wait for the results of REPRIEVE to say whether statins improve outcomes for people living with HIV, Dr. Grinspoon said in an interview.
“Sometimes physicians and primary care providers say, ‘Well I’ll focus my hypertension efforts on older people, who are closer to having heart attacks,’” Dr. Grinspoon said. “But this data suggests we should pay attention even in young people … and pay particular attention to women who wouldn’t have traditional risk scores that were very high at all, largely because they are women.”
The study was funded by Merck. Ms. Gooden has disclosed no relevant financial relationships. Dr. Grinspoon reports receiving personal and consulting fees from Theratechnologies and ViiV Healthcare.
A version of this article first appeared on Medscape.com.
People age 40 and younger living with HIV have a higher risk for heart disease than even their over-40 peers living with HIV – and that risk was 54% higher than the general public.
And this was among people without traditional heart disease risks, such as smoking and obesity.
“What’s surprising is that not only do we see that, yes, they do have increased risk, but this is after controlling for all of that – which means the mechanism underlying this risk,” said Tiffany Gooden, MPH and a PhD candidate at the University of Birmingham, England, who presented the data at the 11th International AIDS Society Conference on HIV Science (IAS 2021).
“If we’re using a non–HIV-validated assessment tool, you should always know that there could be a risk that you are under-recognizing,” she added.
Right now, there’s not a lot to aid clinicians in ferreting out this increased risk. Traditional cardiovascular risk assessment tools, like Framingham risk scores and atherosclerotic cardiovascular disease risk score from the American College of Cardiology, have been found to overlook the real risk of cardiovascular disease in people living with HIV. Plus, most guidelines, including those from the British HIV Medical Association and the American College of Cardiology, primarily focus screening on people 40 or older.
Ms. Gooden’s study drew data from The Health Improvement Network (THIN) database, which combines data from 800 primary care practices in the United Kingdom. Looking at data between January 2000 and January 2020, the investigators compared each person living with HIV with four peers not living with HIV, matched for age, gender, and practice. In total, 9,233 people living with HIV and 35,721 people without HIV were included in the analysis. Median age of participants was 41 years in people living with HIV and 40.4 years in people without HIV. About 35% of participants in both arms were women, and a greater proportion of participants living with HIV were Black, accounting for 22.5% of people living with HIV, versus 3.8% of the general population. Fewer people living with HIV were overweight or obese compared to people without HIV.
Researchers then tracked participants over time to identify the incidence of heart attack, stroke, peripheral vascular disease, and heart failure, as well as common risk factors for heart problems, such as high blood pressure, type 2 diabetes, chronic kidney disease, atrial fibrillation, and use of a lipid-lowering drug such as a statin.
The investigators then sectioned the data on heart disease risk by decade – 2000-2009 and 2010-2019 – in order to separate the potential impact of antiretroviral treatment (ART) drugs, from early combinations that have been associated with cardiovascular disease, to current drugs that are less likely to have that effect.
Overall risk for any kind of cardiovascular disease was 54% higher among people living with HIV of any age, compared to their age- and risk-matched peers. And when they broke the data down by age, they found that people younger than 40 had nearly twice the risk for any heart disease as their HIV-negative peers, which was a numerically higher risk than for people older than 40 – though not significantly so.
People living with HIV also had a 49% increased risk for stroke and a 59% increased risk for ischemic heart disease but no increased risk for peripheral vascular disease, heart failure, or heart attack. But the confidence intervals here were wide, “which may indicate lack of power and therefore not be conclusive,” Ms. Gooden said.
People living with HIV also had a 37% increased risk for hypertension, were 96% more likely to be prescribed lipid-lowering drugs, 2.4-times more likely to have chronic kidney disease, and 2.68-times more likely to experience all-cause mortality. The study couldn’t account for the type of HIV medications people living with the virus used, their viral load, or their CD4 counts – all of which have been found in previous studies to contribute to heart disease in people with HIV.
“That was the biggest limitation of our study,” Ms. Gooden said in an interview. “The fact that the risk of cardiovascular disease remains the same in the [first decade] and the later decade goes to show that even if antiretroviral therapies contributed to that … now or 20 years ago, it’s still not the entire reason for the risk.”
Steven Grinspoon, MD, of Harvard Medical School, Boston, is the lead author on the REPRIEVE trial, now testing statins as a treatment for people like those in this study. He told this news organization that this large analysis had one of the youngest cohorts of people living with HIV he’d seen to explore these issues. Additionally, it backs up what the team recently reported in the Journal of the American Medical Association – that plaque was present in 49% of 755 people living with HIV, despite having risk scores for cardiovascular disease in the normal range. This was as true for people younger than 40 as those older than 40.
For primary care clinicians, the message is that even relatively young people with HIV should be counseled early and often about amending traditional risk factors, while we wait for the results of REPRIEVE to say whether statins improve outcomes for people living with HIV, Dr. Grinspoon said in an interview.
“Sometimes physicians and primary care providers say, ‘Well I’ll focus my hypertension efforts on older people, who are closer to having heart attacks,’” Dr. Grinspoon said. “But this data suggests we should pay attention even in young people … and pay particular attention to women who wouldn’t have traditional risk scores that were very high at all, largely because they are women.”
The study was funded by Merck. Ms. Gooden has disclosed no relevant financial relationships. Dr. Grinspoon reports receiving personal and consulting fees from Theratechnologies and ViiV Healthcare.
A version of this article first appeared on Medscape.com.
People age 40 and younger living with HIV have a higher risk for heart disease than even their over-40 peers living with HIV – and that risk was 54% higher than the general public.
And this was among people without traditional heart disease risks, such as smoking and obesity.
“What’s surprising is that not only do we see that, yes, they do have increased risk, but this is after controlling for all of that – which means the mechanism underlying this risk,” said Tiffany Gooden, MPH and a PhD candidate at the University of Birmingham, England, who presented the data at the 11th International AIDS Society Conference on HIV Science (IAS 2021).
“If we’re using a non–HIV-validated assessment tool, you should always know that there could be a risk that you are under-recognizing,” she added.
Right now, there’s not a lot to aid clinicians in ferreting out this increased risk. Traditional cardiovascular risk assessment tools, like Framingham risk scores and atherosclerotic cardiovascular disease risk score from the American College of Cardiology, have been found to overlook the real risk of cardiovascular disease in people living with HIV. Plus, most guidelines, including those from the British HIV Medical Association and the American College of Cardiology, primarily focus screening on people 40 or older.
Ms. Gooden’s study drew data from The Health Improvement Network (THIN) database, which combines data from 800 primary care practices in the United Kingdom. Looking at data between January 2000 and January 2020, the investigators compared each person living with HIV with four peers not living with HIV, matched for age, gender, and practice. In total, 9,233 people living with HIV and 35,721 people without HIV were included in the analysis. Median age of participants was 41 years in people living with HIV and 40.4 years in people without HIV. About 35% of participants in both arms were women, and a greater proportion of participants living with HIV were Black, accounting for 22.5% of people living with HIV, versus 3.8% of the general population. Fewer people living with HIV were overweight or obese compared to people without HIV.
Researchers then tracked participants over time to identify the incidence of heart attack, stroke, peripheral vascular disease, and heart failure, as well as common risk factors for heart problems, such as high blood pressure, type 2 diabetes, chronic kidney disease, atrial fibrillation, and use of a lipid-lowering drug such as a statin.
The investigators then sectioned the data on heart disease risk by decade – 2000-2009 and 2010-2019 – in order to separate the potential impact of antiretroviral treatment (ART) drugs, from early combinations that have been associated with cardiovascular disease, to current drugs that are less likely to have that effect.
Overall risk for any kind of cardiovascular disease was 54% higher among people living with HIV of any age, compared to their age- and risk-matched peers. And when they broke the data down by age, they found that people younger than 40 had nearly twice the risk for any heart disease as their HIV-negative peers, which was a numerically higher risk than for people older than 40 – though not significantly so.
People living with HIV also had a 49% increased risk for stroke and a 59% increased risk for ischemic heart disease but no increased risk for peripheral vascular disease, heart failure, or heart attack. But the confidence intervals here were wide, “which may indicate lack of power and therefore not be conclusive,” Ms. Gooden said.
People living with HIV also had a 37% increased risk for hypertension, were 96% more likely to be prescribed lipid-lowering drugs, 2.4-times more likely to have chronic kidney disease, and 2.68-times more likely to experience all-cause mortality. The study couldn’t account for the type of HIV medications people living with the virus used, their viral load, or their CD4 counts – all of which have been found in previous studies to contribute to heart disease in people with HIV.
“That was the biggest limitation of our study,” Ms. Gooden said in an interview. “The fact that the risk of cardiovascular disease remains the same in the [first decade] and the later decade goes to show that even if antiretroviral therapies contributed to that … now or 20 years ago, it’s still not the entire reason for the risk.”
Steven Grinspoon, MD, of Harvard Medical School, Boston, is the lead author on the REPRIEVE trial, now testing statins as a treatment for people like those in this study. He told this news organization that this large analysis had one of the youngest cohorts of people living with HIV he’d seen to explore these issues. Additionally, it backs up what the team recently reported in the Journal of the American Medical Association – that plaque was present in 49% of 755 people living with HIV, despite having risk scores for cardiovascular disease in the normal range. This was as true for people younger than 40 as those older than 40.
For primary care clinicians, the message is that even relatively young people with HIV should be counseled early and often about amending traditional risk factors, while we wait for the results of REPRIEVE to say whether statins improve outcomes for people living with HIV, Dr. Grinspoon said in an interview.
“Sometimes physicians and primary care providers say, ‘Well I’ll focus my hypertension efforts on older people, who are closer to having heart attacks,’” Dr. Grinspoon said. “But this data suggests we should pay attention even in young people … and pay particular attention to women who wouldn’t have traditional risk scores that were very high at all, largely because they are women.”
The study was funded by Merck. Ms. Gooden has disclosed no relevant financial relationships. Dr. Grinspoon reports receiving personal and consulting fees from Theratechnologies and ViiV Healthcare.
A version of this article first appeared on Medscape.com.