User login
CABG safe 3 days after stopping ticagrelor: RAPID CABG
Patients with acute coronary syndromes who have been taking the antiplatelet medication, ticagrelor, and who need coronary artery bypass surgery (CABG) may be able to safely have the procedure earlier than typically recommended, a new randomized trial suggests.
The RAPID CABG trial found that early surgery 2-3 days after ticagrelor cessation was noninferior in incurring severe or massive perioperative bleeding, compared with waiting 5-7 days. There was also no significant difference in TIMI CABG or Bleeding Academic Research Consortium (BARC) type 4 or 5 bleeding.
Patients in the delayed group had a numerically higher number of ischemic events requiring earlier surgery and had a longer hospital stay.
The study was presented at the American Heart Association scientific sessions.
“RAPID CABG is the first and only randomized controlled trial evaluating the safety of early surgery in patients taking ticagrelor,” said lead investigator Derek So, MD.
Dr. So, a cardiologist at the University of Ottawa Heart Institute and a professor at the University of Ottawa, explained that ticagrelor is a first-line antiplatelet agent for patients with acute coronary syndromes (ACS), but around 10% of patients presenting with ACS require CABG surgery.
A major concern among patients requiring bypass surgery is perioperative bleeding, and it has been shown that patients undergoing urgent bypass within 24 hours of the last dose of ticagrelor have increased mortality. Accordingly, guidelines suggest a waiting period for patients not requiring urgent bypass surgery, Dr. So noted.
Current North American guidelines suggest a waiting period of at least 5 days after stopping ticagrelor before bypass surgery. In contrast, the updated European and Japanese guidelines suggest a waiting period of 3 days.
Dr. So noted that all of the guidelines are based on cohort studies and pharmacodynamic studies, with no randomized evidence. Pharmacodynamic studies have shown that at 48 hours after the last dose of ticagrelor, the level of platelet inhibition drops to the same levels seen with long-term treatment with clopidogrel, a weaker antiplatelet drug, and after 120 hours (5 days) the effect has completely worn off.
Dr. So concluded that these new results from the RAPID CABG trial “may influence future iterations of North American guidelines with reduced waiting prior to bypass surgery” for patients receiving ticagrelor, and “they could also strengthen the level of evidence in European and Asian guidelines.”
Designated discussant of the RAPID CABG trial, Roxana Mehran, MD, professor of medicine at the Icahn School of Medicine at Mount Sinai, New York, said this was a “very important study,” being the only randomized trial to look at this issue to date.
Dr. Mehran noted that the results showed a similar number of major life-threatening bleeding events in the early and delayed groups and met the noninferiority endpoint, but she pointed out that the trial had a small sample size and a small number of events. “Therefore, larger trials are needed to verify these important and encouraging results.”
However, she concluded that these results should be considered in decisions about the timing of bypass surgery in patients receiving ticagrelor. “I will be changing my practice and sending patients earlier based on this data,” she said.
RAPID CABG
RAPID CABG was a physician-initiated multicenter randomized study evaluating the safety of early surgery at 2-3 days after ticagrelor cessation, compared with a delay of 5-7 days among patients presenting with ACS who required nonemergency CABG surgery.
The study enrolled 143 patients with ACS who were receiving ticagrelor and needed CABG surgery. Patients with stenting for culprit lesions, those requiring urgent surgery (less than 24 hours after presentation), and those requiring valve surgery were excluded.
Three patients declined surgery, and several others underwent surgery outside the assigned time window, so the results were based on the per protocol analysis of patients who actually had CABG in the assigned time window: 65 patients in the early CABG group and 58 in the delayed group.
The mean time from last ticagrelor dose to surgery was 3 days in the early group and 6 days in the delayed group.
Platelet reactivity on the VerifyNow test showed more residual antiplatelet activity in the early group, with P2Y12 reaction unit (PRU) levels of 200 (vs. 251 in the delayed group). This test measures the extent of platelet aggregation in the presence of P2Y12-inhibitor drugs, with lower PRU levels showing stronger antiplatelet effects.
The primary outcome of the study was severe or massive bleeding by Universal Definition of Perioperative Bleeding (UDPB) class 3 or 4. This is defined as a blood transfusions of more than 5 units of red blood cells or plasma within 24 hours of surgical closure, chest tube drainage of over 1,000 mL in the first 12 hours, and reoperation for bleeding.
Results showed that 4.6% of the early-surgery group had a primary outcome bleeding event, compared with 5.2% of the delayed surgery group, meeting the criteria for noninferiority (P = .0253 for noninferiority).
Individual components of the primary endpoint showed three class 3 (severe) bleeding events in both groups and no class 4 (massive) bleeding events in either group.
In terms of other bleeding outcomes, TIMI CABG bleeding occurred in two patients (3.1%) in the early-surgery group vs. no patients in the delayed group; BARC 4 bleeding occurred in two patients (3.1%) in the early group versus none in the delayed group, and there were no BARC 5 bleeding events in either group.
In the intention-to-treat analysis, ischemic events before surgery occurred in six patients (8.7%) in the delayed group (one myocardial infarction, four cases of recurrent ischemia, and one ventricular tachycardia) versus none in the early group.
Cumulative 6-month ischemic events occurred in nine patients (13.0%) in the delayed group vs. four patients (5.6%) in the early group, the difference being driven by nonfatal MI and recurrent ischemia.
There were no cardiovascular deaths in either group and one all-cause death in both groups.
Patients undergoing early surgery also had a shorter hospitalization, with a median length of stay of 9 days versus 12 days in the delayed group.
Larger trial needed
Commenting on the RAPID CABG study at an AHA press conference, Joanna Chikwe, MD, chair of the cardiac surgery department at Cedars-Sinai Medical Center, Los Angeles, said the results were in line with her practice.
“These results confirm what I already think is safe,” she said. “I’m comfortable going within 48 hours. But we individualize our approach, so it was helpful that the study investigators included platelet reactivity data. The interesting thing for me in this study was the number of adverse events in patients who waited longer.”
Dr. Chikwe said her top-line message was that “Surgery looked incredibly safe; there was amazingly low mortality. And if a patient has an indication for surgery, waiting does not serve you well.”
However, she also cautioned that the trial was somewhat underpowered, with a small number of events that drove the primary outcome, leading to some uncertainty on the results.
“The RAPID trial was helpful, and although it confirms my practice, I think physicians may want to see a larger-powered trial to be convincingly compelled that they should change their practice,” Dr. Chikwe noted.
She added that clinical trials in cardiac surgery are driven by inherent challenges. “Cardiac surgery is not very common, and it is hard to recruit patients into these trials, so you are generally tied to a small number of patients, and you therefore have to be extremely thoughtful about the study design. It is almost a given that you will need to use surrogate endpoints, and the choice of the surrogate endpoint can determine which way the trial goes.”
The RAPID CABG study was funded by the Canadian Institutes of Health Research. Dr. So reports research support, consultancy, or speaker’s fees from AggreDyne, Roche Diagnostics, Fujimori Kogyo, and AstraZeneca Canada. Dr. Mehran reports that her institution has received significant trial funding from AstraZeneca (the manufacturer of ticagrelor).
A version of this article first appeared on Medscape.com.
Patients with acute coronary syndromes who have been taking the antiplatelet medication, ticagrelor, and who need coronary artery bypass surgery (CABG) may be able to safely have the procedure earlier than typically recommended, a new randomized trial suggests.
The RAPID CABG trial found that early surgery 2-3 days after ticagrelor cessation was noninferior in incurring severe or massive perioperative bleeding, compared with waiting 5-7 days. There was also no significant difference in TIMI CABG or Bleeding Academic Research Consortium (BARC) type 4 or 5 bleeding.
Patients in the delayed group had a numerically higher number of ischemic events requiring earlier surgery and had a longer hospital stay.
The study was presented at the American Heart Association scientific sessions.
“RAPID CABG is the first and only randomized controlled trial evaluating the safety of early surgery in patients taking ticagrelor,” said lead investigator Derek So, MD.
Dr. So, a cardiologist at the University of Ottawa Heart Institute and a professor at the University of Ottawa, explained that ticagrelor is a first-line antiplatelet agent for patients with acute coronary syndromes (ACS), but around 10% of patients presenting with ACS require CABG surgery.
A major concern among patients requiring bypass surgery is perioperative bleeding, and it has been shown that patients undergoing urgent bypass within 24 hours of the last dose of ticagrelor have increased mortality. Accordingly, guidelines suggest a waiting period for patients not requiring urgent bypass surgery, Dr. So noted.
Current North American guidelines suggest a waiting period of at least 5 days after stopping ticagrelor before bypass surgery. In contrast, the updated European and Japanese guidelines suggest a waiting period of 3 days.
Dr. So noted that all of the guidelines are based on cohort studies and pharmacodynamic studies, with no randomized evidence. Pharmacodynamic studies have shown that at 48 hours after the last dose of ticagrelor, the level of platelet inhibition drops to the same levels seen with long-term treatment with clopidogrel, a weaker antiplatelet drug, and after 120 hours (5 days) the effect has completely worn off.
Dr. So concluded that these new results from the RAPID CABG trial “may influence future iterations of North American guidelines with reduced waiting prior to bypass surgery” for patients receiving ticagrelor, and “they could also strengthen the level of evidence in European and Asian guidelines.”
Designated discussant of the RAPID CABG trial, Roxana Mehran, MD, professor of medicine at the Icahn School of Medicine at Mount Sinai, New York, said this was a “very important study,” being the only randomized trial to look at this issue to date.
Dr. Mehran noted that the results showed a similar number of major life-threatening bleeding events in the early and delayed groups and met the noninferiority endpoint, but she pointed out that the trial had a small sample size and a small number of events. “Therefore, larger trials are needed to verify these important and encouraging results.”
However, she concluded that these results should be considered in decisions about the timing of bypass surgery in patients receiving ticagrelor. “I will be changing my practice and sending patients earlier based on this data,” she said.
RAPID CABG
RAPID CABG was a physician-initiated multicenter randomized study evaluating the safety of early surgery at 2-3 days after ticagrelor cessation, compared with a delay of 5-7 days among patients presenting with ACS who required nonemergency CABG surgery.
The study enrolled 143 patients with ACS who were receiving ticagrelor and needed CABG surgery. Patients with stenting for culprit lesions, those requiring urgent surgery (less than 24 hours after presentation), and those requiring valve surgery were excluded.
Three patients declined surgery, and several others underwent surgery outside the assigned time window, so the results were based on the per protocol analysis of patients who actually had CABG in the assigned time window: 65 patients in the early CABG group and 58 in the delayed group.
The mean time from last ticagrelor dose to surgery was 3 days in the early group and 6 days in the delayed group.
Platelet reactivity on the VerifyNow test showed more residual antiplatelet activity in the early group, with P2Y12 reaction unit (PRU) levels of 200 (vs. 251 in the delayed group). This test measures the extent of platelet aggregation in the presence of P2Y12-inhibitor drugs, with lower PRU levels showing stronger antiplatelet effects.
The primary outcome of the study was severe or massive bleeding by Universal Definition of Perioperative Bleeding (UDPB) class 3 or 4. This is defined as a blood transfusions of more than 5 units of red blood cells or plasma within 24 hours of surgical closure, chest tube drainage of over 1,000 mL in the first 12 hours, and reoperation for bleeding.
Results showed that 4.6% of the early-surgery group had a primary outcome bleeding event, compared with 5.2% of the delayed surgery group, meeting the criteria for noninferiority (P = .0253 for noninferiority).
Individual components of the primary endpoint showed three class 3 (severe) bleeding events in both groups and no class 4 (massive) bleeding events in either group.
In terms of other bleeding outcomes, TIMI CABG bleeding occurred in two patients (3.1%) in the early-surgery group vs. no patients in the delayed group; BARC 4 bleeding occurred in two patients (3.1%) in the early group versus none in the delayed group, and there were no BARC 5 bleeding events in either group.
In the intention-to-treat analysis, ischemic events before surgery occurred in six patients (8.7%) in the delayed group (one myocardial infarction, four cases of recurrent ischemia, and one ventricular tachycardia) versus none in the early group.
Cumulative 6-month ischemic events occurred in nine patients (13.0%) in the delayed group vs. four patients (5.6%) in the early group, the difference being driven by nonfatal MI and recurrent ischemia.
There were no cardiovascular deaths in either group and one all-cause death in both groups.
Patients undergoing early surgery also had a shorter hospitalization, with a median length of stay of 9 days versus 12 days in the delayed group.
Larger trial needed
Commenting on the RAPID CABG study at an AHA press conference, Joanna Chikwe, MD, chair of the cardiac surgery department at Cedars-Sinai Medical Center, Los Angeles, said the results were in line with her practice.
“These results confirm what I already think is safe,” she said. “I’m comfortable going within 48 hours. But we individualize our approach, so it was helpful that the study investigators included platelet reactivity data. The interesting thing for me in this study was the number of adverse events in patients who waited longer.”
Dr. Chikwe said her top-line message was that “Surgery looked incredibly safe; there was amazingly low mortality. And if a patient has an indication for surgery, waiting does not serve you well.”
However, she also cautioned that the trial was somewhat underpowered, with a small number of events that drove the primary outcome, leading to some uncertainty on the results.
“The RAPID trial was helpful, and although it confirms my practice, I think physicians may want to see a larger-powered trial to be convincingly compelled that they should change their practice,” Dr. Chikwe noted.
She added that clinical trials in cardiac surgery are driven by inherent challenges. “Cardiac surgery is not very common, and it is hard to recruit patients into these trials, so you are generally tied to a small number of patients, and you therefore have to be extremely thoughtful about the study design. It is almost a given that you will need to use surrogate endpoints, and the choice of the surrogate endpoint can determine which way the trial goes.”
The RAPID CABG study was funded by the Canadian Institutes of Health Research. Dr. So reports research support, consultancy, or speaker’s fees from AggreDyne, Roche Diagnostics, Fujimori Kogyo, and AstraZeneca Canada. Dr. Mehran reports that her institution has received significant trial funding from AstraZeneca (the manufacturer of ticagrelor).
A version of this article first appeared on Medscape.com.
Patients with acute coronary syndromes who have been taking the antiplatelet medication, ticagrelor, and who need coronary artery bypass surgery (CABG) may be able to safely have the procedure earlier than typically recommended, a new randomized trial suggests.
The RAPID CABG trial found that early surgery 2-3 days after ticagrelor cessation was noninferior in incurring severe or massive perioperative bleeding, compared with waiting 5-7 days. There was also no significant difference in TIMI CABG or Bleeding Academic Research Consortium (BARC) type 4 or 5 bleeding.
Patients in the delayed group had a numerically higher number of ischemic events requiring earlier surgery and had a longer hospital stay.
The study was presented at the American Heart Association scientific sessions.
“RAPID CABG is the first and only randomized controlled trial evaluating the safety of early surgery in patients taking ticagrelor,” said lead investigator Derek So, MD.
Dr. So, a cardiologist at the University of Ottawa Heart Institute and a professor at the University of Ottawa, explained that ticagrelor is a first-line antiplatelet agent for patients with acute coronary syndromes (ACS), but around 10% of patients presenting with ACS require CABG surgery.
A major concern among patients requiring bypass surgery is perioperative bleeding, and it has been shown that patients undergoing urgent bypass within 24 hours of the last dose of ticagrelor have increased mortality. Accordingly, guidelines suggest a waiting period for patients not requiring urgent bypass surgery, Dr. So noted.
Current North American guidelines suggest a waiting period of at least 5 days after stopping ticagrelor before bypass surgery. In contrast, the updated European and Japanese guidelines suggest a waiting period of 3 days.
Dr. So noted that all of the guidelines are based on cohort studies and pharmacodynamic studies, with no randomized evidence. Pharmacodynamic studies have shown that at 48 hours after the last dose of ticagrelor, the level of platelet inhibition drops to the same levels seen with long-term treatment with clopidogrel, a weaker antiplatelet drug, and after 120 hours (5 days) the effect has completely worn off.
Dr. So concluded that these new results from the RAPID CABG trial “may influence future iterations of North American guidelines with reduced waiting prior to bypass surgery” for patients receiving ticagrelor, and “they could also strengthen the level of evidence in European and Asian guidelines.”
Designated discussant of the RAPID CABG trial, Roxana Mehran, MD, professor of medicine at the Icahn School of Medicine at Mount Sinai, New York, said this was a “very important study,” being the only randomized trial to look at this issue to date.
Dr. Mehran noted that the results showed a similar number of major life-threatening bleeding events in the early and delayed groups and met the noninferiority endpoint, but she pointed out that the trial had a small sample size and a small number of events. “Therefore, larger trials are needed to verify these important and encouraging results.”
However, she concluded that these results should be considered in decisions about the timing of bypass surgery in patients receiving ticagrelor. “I will be changing my practice and sending patients earlier based on this data,” she said.
RAPID CABG
RAPID CABG was a physician-initiated multicenter randomized study evaluating the safety of early surgery at 2-3 days after ticagrelor cessation, compared with a delay of 5-7 days among patients presenting with ACS who required nonemergency CABG surgery.
The study enrolled 143 patients with ACS who were receiving ticagrelor and needed CABG surgery. Patients with stenting for culprit lesions, those requiring urgent surgery (less than 24 hours after presentation), and those requiring valve surgery were excluded.
Three patients declined surgery, and several others underwent surgery outside the assigned time window, so the results were based on the per protocol analysis of patients who actually had CABG in the assigned time window: 65 patients in the early CABG group and 58 in the delayed group.
The mean time from last ticagrelor dose to surgery was 3 days in the early group and 6 days in the delayed group.
Platelet reactivity on the VerifyNow test showed more residual antiplatelet activity in the early group, with P2Y12 reaction unit (PRU) levels of 200 (vs. 251 in the delayed group). This test measures the extent of platelet aggregation in the presence of P2Y12-inhibitor drugs, with lower PRU levels showing stronger antiplatelet effects.
The primary outcome of the study was severe or massive bleeding by Universal Definition of Perioperative Bleeding (UDPB) class 3 or 4. This is defined as a blood transfusions of more than 5 units of red blood cells or plasma within 24 hours of surgical closure, chest tube drainage of over 1,000 mL in the first 12 hours, and reoperation for bleeding.
Results showed that 4.6% of the early-surgery group had a primary outcome bleeding event, compared with 5.2% of the delayed surgery group, meeting the criteria for noninferiority (P = .0253 for noninferiority).
Individual components of the primary endpoint showed three class 3 (severe) bleeding events in both groups and no class 4 (massive) bleeding events in either group.
In terms of other bleeding outcomes, TIMI CABG bleeding occurred in two patients (3.1%) in the early-surgery group vs. no patients in the delayed group; BARC 4 bleeding occurred in two patients (3.1%) in the early group versus none in the delayed group, and there were no BARC 5 bleeding events in either group.
In the intention-to-treat analysis, ischemic events before surgery occurred in six patients (8.7%) in the delayed group (one myocardial infarction, four cases of recurrent ischemia, and one ventricular tachycardia) versus none in the early group.
Cumulative 6-month ischemic events occurred in nine patients (13.0%) in the delayed group vs. four patients (5.6%) in the early group, the difference being driven by nonfatal MI and recurrent ischemia.
There were no cardiovascular deaths in either group and one all-cause death in both groups.
Patients undergoing early surgery also had a shorter hospitalization, with a median length of stay of 9 days versus 12 days in the delayed group.
Larger trial needed
Commenting on the RAPID CABG study at an AHA press conference, Joanna Chikwe, MD, chair of the cardiac surgery department at Cedars-Sinai Medical Center, Los Angeles, said the results were in line with her practice.
“These results confirm what I already think is safe,” she said. “I’m comfortable going within 48 hours. But we individualize our approach, so it was helpful that the study investigators included platelet reactivity data. The interesting thing for me in this study was the number of adverse events in patients who waited longer.”
Dr. Chikwe said her top-line message was that “Surgery looked incredibly safe; there was amazingly low mortality. And if a patient has an indication for surgery, waiting does not serve you well.”
However, she also cautioned that the trial was somewhat underpowered, with a small number of events that drove the primary outcome, leading to some uncertainty on the results.
“The RAPID trial was helpful, and although it confirms my practice, I think physicians may want to see a larger-powered trial to be convincingly compelled that they should change their practice,” Dr. Chikwe noted.
She added that clinical trials in cardiac surgery are driven by inherent challenges. “Cardiac surgery is not very common, and it is hard to recruit patients into these trials, so you are generally tied to a small number of patients, and you therefore have to be extremely thoughtful about the study design. It is almost a given that you will need to use surrogate endpoints, and the choice of the surrogate endpoint can determine which way the trial goes.”
The RAPID CABG study was funded by the Canadian Institutes of Health Research. Dr. So reports research support, consultancy, or speaker’s fees from AggreDyne, Roche Diagnostics, Fujimori Kogyo, and AstraZeneca Canada. Dr. Mehran reports that her institution has received significant trial funding from AstraZeneca (the manufacturer of ticagrelor).
A version of this article first appeared on Medscape.com.
FROM AHA 2021
Concomitant tricuspid-mitral surgery beneficial but with a trade-off
Tricuspid valve repair at the time of mitral valve surgery reduces tricuspid regurgitation progression, but at the cost of more than a fivefold increase in permanent pacemakers, results of a new Cardiothoracic Surgical Trials Network study show.
The results were presented during the opening late-breaking science session at the American Heart Association scientific sessions and published simultaneously in the New England Journal of Medicine.
Tricuspid regurgitation (TR) is common among patients undergoing mitral valve surgery, and there’s broad agreement to intervene when a patient has severe TR. There’s uncertainty, however, about the management of moderate or less TR during mitral valve surgery, which is reflected in current guidelines on the basis of observational data, explained coprimary investigator James Gammie, MD, codirector and surgical director of the Johns Hopkins Heart and Vascular Institute, Baltimore. As a result, rates of concomitant tricuspid-mitral surgery range from 5% to 75% at various centers.
To help fill the gap, Dr. Gammie and colleagues screened 5,208 patients at 29 centers in the United States, Canada, and Germany undergoing surgery for degenerative mitral regurgitation, and randomly assigned 401 patients (75% male) to mitral valve surgery alone or with tricuspid annuloplasty.
Patients had either moderate TR (37%) or less than moderate TR with a dilated tricuspid annulus of at least 40 mm or at least 21 mm/m2 indexed for body surface area. Importantly, there was a uniform surgical approach using undersized (26-30 mm) rigid nonplanar annuloplasty rings to repair the tricuspid valve, he said.
The study’s primary outcome of treatment failure at 2 years was defined as the composite of death, reoperation for TR, or progression of TR from baseline by 2 grades or severe TR.
The primary endpoint occurred in 10.2% of patients who underwent mitral valve surgery alone and 3.9% who underwent concomitant tricuspid annuloplasty (relative risk, 0.37; 95% confidence interval, 0.16-0.86; P = .02).
The endpoint was driven exclusively by less TR progression in the annuloplasty group, with no TR reoperations in either group, observed Dr. Gammie. At 2 years, just 0.6% of the annuloplasty group had severe TR, compared with 5.6% of the surgery-alone group.
The rate of permanent pacemaker implantations, however, jumped from 2.5% with surgery alone to 14.1% with concomitant tricuspid annuloplasty (rate ratio, 5.75; 95% CI, 2.27-14.60). More than half of pacemakers were placed during the first 2 days after surgery.
There was no between-group difference in 2-year rates of all-cause mortality, major adverse cardiac and cerebrovascular events, readmission, quality of life, or functional status.
Less than moderate TR
In a post hoc analysis stratified by baseline TR severity, treatment failure was significantly less common with surgery plus tricuspid annuloplasty among patients with moderate TR (4.5% vs. 18.1%) but not among those with less than moderate TR and tricuspid annular dilation (3.4% vs. 6.1%).
Although the trial was not powered for the subgroup analysis, “these results call into question the idea that less than moderate TR with annular dilation should be an indication for tricuspid valve repair,” Dr. Gammie told this news organization.
“I did not repair the tricuspid valve in the setting of less than moderate TR before the trial, and my practice won’t change; but it will be based on much better evidence,” he added. “Of course, long-term data from our trial will be of great interest.”
Discussant Joseph Woo, MD, chair of surgery at Stanford (Calif.) University, congratulated the authors on a “landmark trial” that addresses a highly relevant problem without a clear-cut indication.
In the 2020 AHA/American College of Cardiology heart valve disease guideline, tricuspid valve surgery is a class I recommendation when there’s severe TR (stages C and D) and left-sided valve surgery but a class IIa recommendation in patients with progressive TR (stage B) with an annular dilation of at least 40 mm.
“The interesting findings in this study include that moderate TR was only 37% of the enrolled patients, and only 97% of the patients with degenerative MR received a mitral valve repair,” Dr. Woo said. “This level of mitral valve repair is perhaps lower than what we might expect at these centers and lower, certainly, than what the AHA/ACC guidelines recommend for surgery on asymptomatic severe mitral regurgitation.”
Panelist Roxanna Mehran, MD, of Icahn School of Medicine at Mount Sinai in New York said, “What I was struck by is that we, as clinicians, believe that if you fix the mitral valve, maybe the tricuspid regurgitation will improve. And it seems like that is not what’s happening, and I think that’s a big takeaway.”
Session comoderator Joanna Chikwe, MD, head of cardiac surgery at Cedars-Sinai Medical Center, Los Angeles, said, “I think we can all agree that severe tricuspid regurgitation is a disaster for patients, and I think the fact the trial is designed for an additional 5 years’ follow-up will hopefully give us some insights into the clinical impact of severe tricuspid regurgitation.”
For now, “a back of the envelope calculation suggests that, for every 20 patients with moderate tricuspid regurgitation who we repair the tricuspid valve in, we would prevent severe tricuspid valve regurgitation in 1 at the price of pacemakers in 2,” she said.
Dr. Chikwe said in an interview that “transcatheter tricuspid repair is increasingly helping these patients, but if you could avoid it with a technique that doesn’t cause incremental harm beyond, perhaps, the need for pacemakers, then this is helpful data that supports that approach.”
The pacemaker burden is not negligible, she said, but also not surprising to surgeons. “If you look at national practice of mitral-tricuspid surgery, it’s about 15% after that, and it’s simply because the conduction tissue is so close to the tricuspid annulus.”
Pacemaker implantation rates, like those for concomitant tricuspid-mitral surgery, are also highly variable, and in some single-center series only around 2%, Dr. Chikwe said. “So that suggests there are technical approaches that can minimize the pacemaker rate [such as] being extremely careful to avoid suture placement around the area of the conduction tissues.”
For some the trade-off between reduced TR progression and the risk of a permanent pacemaker is worth it. “But the fact that the trial didn’t show a difference in survival, a difference in symptoms or quality of life, might suggest that patients you anticipated were high risk for surgery or didn’t have a longer projected survival aren’t going to benefit from what is quite an aggressive surgical approach,” Dr. Chikwe said.
In an accompanying editorial, Dr. Chikwe and Mario Gaudino, MD, of Weill Cornell Medicine, New York, also point out that the “very dynamic nature of tricuspid regurgitation and wide variability in assessing tricuspid annular dilatation are additional compelling reasons to leave lesser regurgitation alone.”
Julia Grapsa, MD, PhD, Kings College and tricuspid service lead at Guys and St. Thomas NHS, London, also pointed to the need for longer-term follow-up but said increased use of imaging markers is also needed to help pinpoint TR progression in these patients. “For the moment, the results should remind imagers and clinicians to refer patients earlier.”
“As a valvular heart physician, I see more and more patients coming in with significant severe tricuspid regurgitation post–mitral valve surgery and because of the time that’s passed, there’s dysfunction of the right heart, the left heart, and it’s very hard to suggest an operation because they’re at high risk,” she said. “So we’re discussing with these patients whether to do an intervention or medical management.”
“Now, with this study, and the pending longer follow-up by the authors, I’m optimistic that the class II recommendation will be class I in order to help our patients treat tricuspid regurgitation earlier than late,” said Dr. Grapsa, who is also editor-in-chief of JACC: Case Reports.
The study was funded by the National Heart, Lung, and Blood Institute and the German Center for Cardiovascular Research. Dr. Gammie reports a consultant/stockholder relationship with Edwards Lifesciences. Dr. Grapsa reports no conflicts of interest. Dr. Chikwe reports that as coprincipal investigator/study director of NCT 05051033 (an NHLBI-sponsored Cardiothoracic Surgical Trials Network trial), she collaborates with several of the study authors.
A version of this article first appeared on Medscape.com.
Tricuspid valve repair at the time of mitral valve surgery reduces tricuspid regurgitation progression, but at the cost of more than a fivefold increase in permanent pacemakers, results of a new Cardiothoracic Surgical Trials Network study show.
The results were presented during the opening late-breaking science session at the American Heart Association scientific sessions and published simultaneously in the New England Journal of Medicine.
Tricuspid regurgitation (TR) is common among patients undergoing mitral valve surgery, and there’s broad agreement to intervene when a patient has severe TR. There’s uncertainty, however, about the management of moderate or less TR during mitral valve surgery, which is reflected in current guidelines on the basis of observational data, explained coprimary investigator James Gammie, MD, codirector and surgical director of the Johns Hopkins Heart and Vascular Institute, Baltimore. As a result, rates of concomitant tricuspid-mitral surgery range from 5% to 75% at various centers.
To help fill the gap, Dr. Gammie and colleagues screened 5,208 patients at 29 centers in the United States, Canada, and Germany undergoing surgery for degenerative mitral regurgitation, and randomly assigned 401 patients (75% male) to mitral valve surgery alone or with tricuspid annuloplasty.
Patients had either moderate TR (37%) or less than moderate TR with a dilated tricuspid annulus of at least 40 mm or at least 21 mm/m2 indexed for body surface area. Importantly, there was a uniform surgical approach using undersized (26-30 mm) rigid nonplanar annuloplasty rings to repair the tricuspid valve, he said.
The study’s primary outcome of treatment failure at 2 years was defined as the composite of death, reoperation for TR, or progression of TR from baseline by 2 grades or severe TR.
The primary endpoint occurred in 10.2% of patients who underwent mitral valve surgery alone and 3.9% who underwent concomitant tricuspid annuloplasty (relative risk, 0.37; 95% confidence interval, 0.16-0.86; P = .02).
The endpoint was driven exclusively by less TR progression in the annuloplasty group, with no TR reoperations in either group, observed Dr. Gammie. At 2 years, just 0.6% of the annuloplasty group had severe TR, compared with 5.6% of the surgery-alone group.
The rate of permanent pacemaker implantations, however, jumped from 2.5% with surgery alone to 14.1% with concomitant tricuspid annuloplasty (rate ratio, 5.75; 95% CI, 2.27-14.60). More than half of pacemakers were placed during the first 2 days after surgery.
There was no between-group difference in 2-year rates of all-cause mortality, major adverse cardiac and cerebrovascular events, readmission, quality of life, or functional status.
Less than moderate TR
In a post hoc analysis stratified by baseline TR severity, treatment failure was significantly less common with surgery plus tricuspid annuloplasty among patients with moderate TR (4.5% vs. 18.1%) but not among those with less than moderate TR and tricuspid annular dilation (3.4% vs. 6.1%).
Although the trial was not powered for the subgroup analysis, “these results call into question the idea that less than moderate TR with annular dilation should be an indication for tricuspid valve repair,” Dr. Gammie told this news organization.
“I did not repair the tricuspid valve in the setting of less than moderate TR before the trial, and my practice won’t change; but it will be based on much better evidence,” he added. “Of course, long-term data from our trial will be of great interest.”
Discussant Joseph Woo, MD, chair of surgery at Stanford (Calif.) University, congratulated the authors on a “landmark trial” that addresses a highly relevant problem without a clear-cut indication.
In the 2020 AHA/American College of Cardiology heart valve disease guideline, tricuspid valve surgery is a class I recommendation when there’s severe TR (stages C and D) and left-sided valve surgery but a class IIa recommendation in patients with progressive TR (stage B) with an annular dilation of at least 40 mm.
“The interesting findings in this study include that moderate TR was only 37% of the enrolled patients, and only 97% of the patients with degenerative MR received a mitral valve repair,” Dr. Woo said. “This level of mitral valve repair is perhaps lower than what we might expect at these centers and lower, certainly, than what the AHA/ACC guidelines recommend for surgery on asymptomatic severe mitral regurgitation.”
Panelist Roxanna Mehran, MD, of Icahn School of Medicine at Mount Sinai in New York said, “What I was struck by is that we, as clinicians, believe that if you fix the mitral valve, maybe the tricuspid regurgitation will improve. And it seems like that is not what’s happening, and I think that’s a big takeaway.”
Session comoderator Joanna Chikwe, MD, head of cardiac surgery at Cedars-Sinai Medical Center, Los Angeles, said, “I think we can all agree that severe tricuspid regurgitation is a disaster for patients, and I think the fact the trial is designed for an additional 5 years’ follow-up will hopefully give us some insights into the clinical impact of severe tricuspid regurgitation.”
For now, “a back of the envelope calculation suggests that, for every 20 patients with moderate tricuspid regurgitation who we repair the tricuspid valve in, we would prevent severe tricuspid valve regurgitation in 1 at the price of pacemakers in 2,” she said.
Dr. Chikwe said in an interview that “transcatheter tricuspid repair is increasingly helping these patients, but if you could avoid it with a technique that doesn’t cause incremental harm beyond, perhaps, the need for pacemakers, then this is helpful data that supports that approach.”
The pacemaker burden is not negligible, she said, but also not surprising to surgeons. “If you look at national practice of mitral-tricuspid surgery, it’s about 15% after that, and it’s simply because the conduction tissue is so close to the tricuspid annulus.”
Pacemaker implantation rates, like those for concomitant tricuspid-mitral surgery, are also highly variable, and in some single-center series only around 2%, Dr. Chikwe said. “So that suggests there are technical approaches that can minimize the pacemaker rate [such as] being extremely careful to avoid suture placement around the area of the conduction tissues.”
For some the trade-off between reduced TR progression and the risk of a permanent pacemaker is worth it. “But the fact that the trial didn’t show a difference in survival, a difference in symptoms or quality of life, might suggest that patients you anticipated were high risk for surgery or didn’t have a longer projected survival aren’t going to benefit from what is quite an aggressive surgical approach,” Dr. Chikwe said.
In an accompanying editorial, Dr. Chikwe and Mario Gaudino, MD, of Weill Cornell Medicine, New York, also point out that the “very dynamic nature of tricuspid regurgitation and wide variability in assessing tricuspid annular dilatation are additional compelling reasons to leave lesser regurgitation alone.”
Julia Grapsa, MD, PhD, Kings College and tricuspid service lead at Guys and St. Thomas NHS, London, also pointed to the need for longer-term follow-up but said increased use of imaging markers is also needed to help pinpoint TR progression in these patients. “For the moment, the results should remind imagers and clinicians to refer patients earlier.”
“As a valvular heart physician, I see more and more patients coming in with significant severe tricuspid regurgitation post–mitral valve surgery and because of the time that’s passed, there’s dysfunction of the right heart, the left heart, and it’s very hard to suggest an operation because they’re at high risk,” she said. “So we’re discussing with these patients whether to do an intervention or medical management.”
“Now, with this study, and the pending longer follow-up by the authors, I’m optimistic that the class II recommendation will be class I in order to help our patients treat tricuspid regurgitation earlier than late,” said Dr. Grapsa, who is also editor-in-chief of JACC: Case Reports.
The study was funded by the National Heart, Lung, and Blood Institute and the German Center for Cardiovascular Research. Dr. Gammie reports a consultant/stockholder relationship with Edwards Lifesciences. Dr. Grapsa reports no conflicts of interest. Dr. Chikwe reports that as coprincipal investigator/study director of NCT 05051033 (an NHLBI-sponsored Cardiothoracic Surgical Trials Network trial), she collaborates with several of the study authors.
A version of this article first appeared on Medscape.com.
Tricuspid valve repair at the time of mitral valve surgery reduces tricuspid regurgitation progression, but at the cost of more than a fivefold increase in permanent pacemakers, results of a new Cardiothoracic Surgical Trials Network study show.
The results were presented during the opening late-breaking science session at the American Heart Association scientific sessions and published simultaneously in the New England Journal of Medicine.
Tricuspid regurgitation (TR) is common among patients undergoing mitral valve surgery, and there’s broad agreement to intervene when a patient has severe TR. There’s uncertainty, however, about the management of moderate or less TR during mitral valve surgery, which is reflected in current guidelines on the basis of observational data, explained coprimary investigator James Gammie, MD, codirector and surgical director of the Johns Hopkins Heart and Vascular Institute, Baltimore. As a result, rates of concomitant tricuspid-mitral surgery range from 5% to 75% at various centers.
To help fill the gap, Dr. Gammie and colleagues screened 5,208 patients at 29 centers in the United States, Canada, and Germany undergoing surgery for degenerative mitral regurgitation, and randomly assigned 401 patients (75% male) to mitral valve surgery alone or with tricuspid annuloplasty.
Patients had either moderate TR (37%) or less than moderate TR with a dilated tricuspid annulus of at least 40 mm or at least 21 mm/m2 indexed for body surface area. Importantly, there was a uniform surgical approach using undersized (26-30 mm) rigid nonplanar annuloplasty rings to repair the tricuspid valve, he said.
The study’s primary outcome of treatment failure at 2 years was defined as the composite of death, reoperation for TR, or progression of TR from baseline by 2 grades or severe TR.
The primary endpoint occurred in 10.2% of patients who underwent mitral valve surgery alone and 3.9% who underwent concomitant tricuspid annuloplasty (relative risk, 0.37; 95% confidence interval, 0.16-0.86; P = .02).
The endpoint was driven exclusively by less TR progression in the annuloplasty group, with no TR reoperations in either group, observed Dr. Gammie. At 2 years, just 0.6% of the annuloplasty group had severe TR, compared with 5.6% of the surgery-alone group.
The rate of permanent pacemaker implantations, however, jumped from 2.5% with surgery alone to 14.1% with concomitant tricuspid annuloplasty (rate ratio, 5.75; 95% CI, 2.27-14.60). More than half of pacemakers were placed during the first 2 days after surgery.
There was no between-group difference in 2-year rates of all-cause mortality, major adverse cardiac and cerebrovascular events, readmission, quality of life, or functional status.
Less than moderate TR
In a post hoc analysis stratified by baseline TR severity, treatment failure was significantly less common with surgery plus tricuspid annuloplasty among patients with moderate TR (4.5% vs. 18.1%) but not among those with less than moderate TR and tricuspid annular dilation (3.4% vs. 6.1%).
Although the trial was not powered for the subgroup analysis, “these results call into question the idea that less than moderate TR with annular dilation should be an indication for tricuspid valve repair,” Dr. Gammie told this news organization.
“I did not repair the tricuspid valve in the setting of less than moderate TR before the trial, and my practice won’t change; but it will be based on much better evidence,” he added. “Of course, long-term data from our trial will be of great interest.”
Discussant Joseph Woo, MD, chair of surgery at Stanford (Calif.) University, congratulated the authors on a “landmark trial” that addresses a highly relevant problem without a clear-cut indication.
In the 2020 AHA/American College of Cardiology heart valve disease guideline, tricuspid valve surgery is a class I recommendation when there’s severe TR (stages C and D) and left-sided valve surgery but a class IIa recommendation in patients with progressive TR (stage B) with an annular dilation of at least 40 mm.
“The interesting findings in this study include that moderate TR was only 37% of the enrolled patients, and only 97% of the patients with degenerative MR received a mitral valve repair,” Dr. Woo said. “This level of mitral valve repair is perhaps lower than what we might expect at these centers and lower, certainly, than what the AHA/ACC guidelines recommend for surgery on asymptomatic severe mitral regurgitation.”
Panelist Roxanna Mehran, MD, of Icahn School of Medicine at Mount Sinai in New York said, “What I was struck by is that we, as clinicians, believe that if you fix the mitral valve, maybe the tricuspid regurgitation will improve. And it seems like that is not what’s happening, and I think that’s a big takeaway.”
Session comoderator Joanna Chikwe, MD, head of cardiac surgery at Cedars-Sinai Medical Center, Los Angeles, said, “I think we can all agree that severe tricuspid regurgitation is a disaster for patients, and I think the fact the trial is designed for an additional 5 years’ follow-up will hopefully give us some insights into the clinical impact of severe tricuspid regurgitation.”
For now, “a back of the envelope calculation suggests that, for every 20 patients with moderate tricuspid regurgitation who we repair the tricuspid valve in, we would prevent severe tricuspid valve regurgitation in 1 at the price of pacemakers in 2,” she said.
Dr. Chikwe said in an interview that “transcatheter tricuspid repair is increasingly helping these patients, but if you could avoid it with a technique that doesn’t cause incremental harm beyond, perhaps, the need for pacemakers, then this is helpful data that supports that approach.”
The pacemaker burden is not negligible, she said, but also not surprising to surgeons. “If you look at national practice of mitral-tricuspid surgery, it’s about 15% after that, and it’s simply because the conduction tissue is so close to the tricuspid annulus.”
Pacemaker implantation rates, like those for concomitant tricuspid-mitral surgery, are also highly variable, and in some single-center series only around 2%, Dr. Chikwe said. “So that suggests there are technical approaches that can minimize the pacemaker rate [such as] being extremely careful to avoid suture placement around the area of the conduction tissues.”
For some the trade-off between reduced TR progression and the risk of a permanent pacemaker is worth it. “But the fact that the trial didn’t show a difference in survival, a difference in symptoms or quality of life, might suggest that patients you anticipated were high risk for surgery or didn’t have a longer projected survival aren’t going to benefit from what is quite an aggressive surgical approach,” Dr. Chikwe said.
In an accompanying editorial, Dr. Chikwe and Mario Gaudino, MD, of Weill Cornell Medicine, New York, also point out that the “very dynamic nature of tricuspid regurgitation and wide variability in assessing tricuspid annular dilatation are additional compelling reasons to leave lesser regurgitation alone.”
Julia Grapsa, MD, PhD, Kings College and tricuspid service lead at Guys and St. Thomas NHS, London, also pointed to the need for longer-term follow-up but said increased use of imaging markers is also needed to help pinpoint TR progression in these patients. “For the moment, the results should remind imagers and clinicians to refer patients earlier.”
“As a valvular heart physician, I see more and more patients coming in with significant severe tricuspid regurgitation post–mitral valve surgery and because of the time that’s passed, there’s dysfunction of the right heart, the left heart, and it’s very hard to suggest an operation because they’re at high risk,” she said. “So we’re discussing with these patients whether to do an intervention or medical management.”
“Now, with this study, and the pending longer follow-up by the authors, I’m optimistic that the class II recommendation will be class I in order to help our patients treat tricuspid regurgitation earlier than late,” said Dr. Grapsa, who is also editor-in-chief of JACC: Case Reports.
The study was funded by the National Heart, Lung, and Blood Institute and the German Center for Cardiovascular Research. Dr. Gammie reports a consultant/stockholder relationship with Edwards Lifesciences. Dr. Grapsa reports no conflicts of interest. Dr. Chikwe reports that as coprincipal investigator/study director of NCT 05051033 (an NHLBI-sponsored Cardiothoracic Surgical Trials Network trial), she collaborates with several of the study authors.
A version of this article first appeared on Medscape.com.
FROM AHA 2021
Stroke thrombectomy alone fails noninferiority to bridging tPA
In the prospective, multicenter trial, the rate of good functional outcome was 57% for patients who underwent direct thrombectomy and 65% among patients who received IV thrombolysis before undergoing thrombectomy. This result failed to demonstrate noninferiority of direct mechanical thrombectomy compared to combination therapy, the researchers conclude.
“Good outcome was high in both treatment arms, with the point estimate in favor of the bridging cohort,” said lead investigator Urs Fischer, MD, co-chair of the stroke center at Inselspital, Bern University Hospital, Switzerland, during his presentation. “Postinterventional reperfusion was very high in both treatment arms and higher in patients with bridging thrombolysis, compared to direct mechanical thrombectomy.”
The findings were presented at the 13th World Stroke Congress (WSC) 2021.
Two views of thrombolysis
The value of bridging thrombolysis for patients who undergo mechanical thrombectomy is a matter of debate. One argument is that, for patients with large-vessel occlusion, IV thrombolysis may improve reperfusion before and after thrombectomy and yield better clinical outcomes. The opposing argument is that bridging thrombolysis may increase the risk for distal emboli, delay mechanical thrombectomy, and increase the rate of hemorrhage.
The researchers conducted the SWIFT DIRECT trial to investigate this question. They enrolled patients with acute ischemic stroke due to occlusion of the internal carotid artery or the M1 segment of the middle cerebral artery.
The trial was conducted at 48 sites in seven European countries and Canada. The investigators randomly assigned patients to receive IV alteplase (0.9 mg/kg) plus mechanical thrombectomy with the Solitaire device or to receive direct mechanical thrombectomy with the same device. Treatment was open label, but the assessment of endpoints was blinded.
Investigators assigned 423 patients to treatment, and 408 were included in the full analysis set. Of this group, 201 participants received direct mechanical thrombectomy, and 207 received IV thrombolysis plus thrombectomy. There were three crossovers in each treatment arm.
The primary outcome was functional independence, defined as a Modified Rankin Scale (mRS) score of 0-2, at 90 days. Secondary outcomes included mortality at 90 days, mRS shift, change in National Institutes of Health Stroke Scale (NIHSS) score at 24 hours, successful reperfusion, and symptomatic and asymptomatic intracranial hemorrhage (ICH).
Noninferiority not demonstrated
At baseline, patient characteristics were well balanced between the treatment groups. The median age of the patients was 72 years, and about 50% of participants were women. The median NIHSS score was 17 in both arms.
Approximately 57% of patients who underwent direct thrombectomy and 65% of those who received IV thrombolysis plus thrombectomy were functionally independent at 90 days, the primary outcome.
In addition, the researchers found no difference in mRS shift, mortality at 90 days, or change in NIHSS score at 24 hours. Postinterventional reperfusion was very high in both arms and was higher in patients who received IV tissue plasminogen activator, compared with those who received direct mechanical thrombectomy, said Dr. Fischer.
The rate of successful postinterventional reperfusion, however, was higher among patients who received thrombolysis than among those who underwent direct thrombectomy. The rate of symptomatic ICH was 1.5% in the direct thrombectomy group and 4.9% in the thrombolysis-plus-thrombectomy group.
New endpoints needed?
The investigators used noninferiority margins of 12%. “This question about the noninferiority margins, that’s a very tricky and difficult one in randomized clinical trials,” said Dr. Fischer. The investigators defined their margin using the 2015 HERMES data because no trials had yet compared direct mechanical thrombectomy and bridging thrombolysis at the time.
The researchers are performing a pooled analysis of all the trials that compared bridging thrombolysis with direct mechanical thrombectomy. “We are therefore looking at several margins, and I think this is the way we should look at these noninferiority margins,” said Dr. Fischer. “There’s not a clear-cut level which you can define.”
Enrollment in the trial was well balanced with respect to gender, which is not always the case in stroke studies, said Kevin Sheth, MD, professor of neurology and neurosurgery at Yale School of Medicine, New Haven, Conn., who commented on the study for this news organization.
The findings indicate that the likelihood of there being a difference between groups on this question is low, said Dr. Sheth. Both groups had large-vessel occlusion, both received thrombectomy, and both achieved reperfusion. But the higher rate of successful reperfusion in the bridging cohort was not reflected in any of the clinical endpoints that the investigators examined.
Observing a difference in this context will require very large trials or different endpoints that are more responsive to the intervention, said Dr. Sheth. “This is going to be a challenge for not just this but for any neuroprotection trial in the future,” he said.
The study was supported by Medtronic. Dr. Fischer has served as a consultant for Medtronic, Stryker, and CSL Behring. Dr. Sheth has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the prospective, multicenter trial, the rate of good functional outcome was 57% for patients who underwent direct thrombectomy and 65% among patients who received IV thrombolysis before undergoing thrombectomy. This result failed to demonstrate noninferiority of direct mechanical thrombectomy compared to combination therapy, the researchers conclude.
“Good outcome was high in both treatment arms, with the point estimate in favor of the bridging cohort,” said lead investigator Urs Fischer, MD, co-chair of the stroke center at Inselspital, Bern University Hospital, Switzerland, during his presentation. “Postinterventional reperfusion was very high in both treatment arms and higher in patients with bridging thrombolysis, compared to direct mechanical thrombectomy.”
The findings were presented at the 13th World Stroke Congress (WSC) 2021.
Two views of thrombolysis
The value of bridging thrombolysis for patients who undergo mechanical thrombectomy is a matter of debate. One argument is that, for patients with large-vessel occlusion, IV thrombolysis may improve reperfusion before and after thrombectomy and yield better clinical outcomes. The opposing argument is that bridging thrombolysis may increase the risk for distal emboli, delay mechanical thrombectomy, and increase the rate of hemorrhage.
The researchers conducted the SWIFT DIRECT trial to investigate this question. They enrolled patients with acute ischemic stroke due to occlusion of the internal carotid artery or the M1 segment of the middle cerebral artery.
The trial was conducted at 48 sites in seven European countries and Canada. The investigators randomly assigned patients to receive IV alteplase (0.9 mg/kg) plus mechanical thrombectomy with the Solitaire device or to receive direct mechanical thrombectomy with the same device. Treatment was open label, but the assessment of endpoints was blinded.
Investigators assigned 423 patients to treatment, and 408 were included in the full analysis set. Of this group, 201 participants received direct mechanical thrombectomy, and 207 received IV thrombolysis plus thrombectomy. There were three crossovers in each treatment arm.
The primary outcome was functional independence, defined as a Modified Rankin Scale (mRS) score of 0-2, at 90 days. Secondary outcomes included mortality at 90 days, mRS shift, change in National Institutes of Health Stroke Scale (NIHSS) score at 24 hours, successful reperfusion, and symptomatic and asymptomatic intracranial hemorrhage (ICH).
Noninferiority not demonstrated
At baseline, patient characteristics were well balanced between the treatment groups. The median age of the patients was 72 years, and about 50% of participants were women. The median NIHSS score was 17 in both arms.
Approximately 57% of patients who underwent direct thrombectomy and 65% of those who received IV thrombolysis plus thrombectomy were functionally independent at 90 days, the primary outcome.
In addition, the researchers found no difference in mRS shift, mortality at 90 days, or change in NIHSS score at 24 hours. Postinterventional reperfusion was very high in both arms and was higher in patients who received IV tissue plasminogen activator, compared with those who received direct mechanical thrombectomy, said Dr. Fischer.
The rate of successful postinterventional reperfusion, however, was higher among patients who received thrombolysis than among those who underwent direct thrombectomy. The rate of symptomatic ICH was 1.5% in the direct thrombectomy group and 4.9% in the thrombolysis-plus-thrombectomy group.
New endpoints needed?
The investigators used noninferiority margins of 12%. “This question about the noninferiority margins, that’s a very tricky and difficult one in randomized clinical trials,” said Dr. Fischer. The investigators defined their margin using the 2015 HERMES data because no trials had yet compared direct mechanical thrombectomy and bridging thrombolysis at the time.
The researchers are performing a pooled analysis of all the trials that compared bridging thrombolysis with direct mechanical thrombectomy. “We are therefore looking at several margins, and I think this is the way we should look at these noninferiority margins,” said Dr. Fischer. “There’s not a clear-cut level which you can define.”
Enrollment in the trial was well balanced with respect to gender, which is not always the case in stroke studies, said Kevin Sheth, MD, professor of neurology and neurosurgery at Yale School of Medicine, New Haven, Conn., who commented on the study for this news organization.
The findings indicate that the likelihood of there being a difference between groups on this question is low, said Dr. Sheth. Both groups had large-vessel occlusion, both received thrombectomy, and both achieved reperfusion. But the higher rate of successful reperfusion in the bridging cohort was not reflected in any of the clinical endpoints that the investigators examined.
Observing a difference in this context will require very large trials or different endpoints that are more responsive to the intervention, said Dr. Sheth. “This is going to be a challenge for not just this but for any neuroprotection trial in the future,” he said.
The study was supported by Medtronic. Dr. Fischer has served as a consultant for Medtronic, Stryker, and CSL Behring. Dr. Sheth has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In the prospective, multicenter trial, the rate of good functional outcome was 57% for patients who underwent direct thrombectomy and 65% among patients who received IV thrombolysis before undergoing thrombectomy. This result failed to demonstrate noninferiority of direct mechanical thrombectomy compared to combination therapy, the researchers conclude.
“Good outcome was high in both treatment arms, with the point estimate in favor of the bridging cohort,” said lead investigator Urs Fischer, MD, co-chair of the stroke center at Inselspital, Bern University Hospital, Switzerland, during his presentation. “Postinterventional reperfusion was very high in both treatment arms and higher in patients with bridging thrombolysis, compared to direct mechanical thrombectomy.”
The findings were presented at the 13th World Stroke Congress (WSC) 2021.
Two views of thrombolysis
The value of bridging thrombolysis for patients who undergo mechanical thrombectomy is a matter of debate. One argument is that, for patients with large-vessel occlusion, IV thrombolysis may improve reperfusion before and after thrombectomy and yield better clinical outcomes. The opposing argument is that bridging thrombolysis may increase the risk for distal emboli, delay mechanical thrombectomy, and increase the rate of hemorrhage.
The researchers conducted the SWIFT DIRECT trial to investigate this question. They enrolled patients with acute ischemic stroke due to occlusion of the internal carotid artery or the M1 segment of the middle cerebral artery.
The trial was conducted at 48 sites in seven European countries and Canada. The investigators randomly assigned patients to receive IV alteplase (0.9 mg/kg) plus mechanical thrombectomy with the Solitaire device or to receive direct mechanical thrombectomy with the same device. Treatment was open label, but the assessment of endpoints was blinded.
Investigators assigned 423 patients to treatment, and 408 were included in the full analysis set. Of this group, 201 participants received direct mechanical thrombectomy, and 207 received IV thrombolysis plus thrombectomy. There were three crossovers in each treatment arm.
The primary outcome was functional independence, defined as a Modified Rankin Scale (mRS) score of 0-2, at 90 days. Secondary outcomes included mortality at 90 days, mRS shift, change in National Institutes of Health Stroke Scale (NIHSS) score at 24 hours, successful reperfusion, and symptomatic and asymptomatic intracranial hemorrhage (ICH).
Noninferiority not demonstrated
At baseline, patient characteristics were well balanced between the treatment groups. The median age of the patients was 72 years, and about 50% of participants were women. The median NIHSS score was 17 in both arms.
Approximately 57% of patients who underwent direct thrombectomy and 65% of those who received IV thrombolysis plus thrombectomy were functionally independent at 90 days, the primary outcome.
In addition, the researchers found no difference in mRS shift, mortality at 90 days, or change in NIHSS score at 24 hours. Postinterventional reperfusion was very high in both arms and was higher in patients who received IV tissue plasminogen activator, compared with those who received direct mechanical thrombectomy, said Dr. Fischer.
The rate of successful postinterventional reperfusion, however, was higher among patients who received thrombolysis than among those who underwent direct thrombectomy. The rate of symptomatic ICH was 1.5% in the direct thrombectomy group and 4.9% in the thrombolysis-plus-thrombectomy group.
New endpoints needed?
The investigators used noninferiority margins of 12%. “This question about the noninferiority margins, that’s a very tricky and difficult one in randomized clinical trials,” said Dr. Fischer. The investigators defined their margin using the 2015 HERMES data because no trials had yet compared direct mechanical thrombectomy and bridging thrombolysis at the time.
The researchers are performing a pooled analysis of all the trials that compared bridging thrombolysis with direct mechanical thrombectomy. “We are therefore looking at several margins, and I think this is the way we should look at these noninferiority margins,” said Dr. Fischer. “There’s not a clear-cut level which you can define.”
Enrollment in the trial was well balanced with respect to gender, which is not always the case in stroke studies, said Kevin Sheth, MD, professor of neurology and neurosurgery at Yale School of Medicine, New Haven, Conn., who commented on the study for this news organization.
The findings indicate that the likelihood of there being a difference between groups on this question is low, said Dr. Sheth. Both groups had large-vessel occlusion, both received thrombectomy, and both achieved reperfusion. But the higher rate of successful reperfusion in the bridging cohort was not reflected in any of the clinical endpoints that the investigators examined.
Observing a difference in this context will require very large trials or different endpoints that are more responsive to the intervention, said Dr. Sheth. “This is going to be a challenge for not just this but for any neuroprotection trial in the future,” he said.
The study was supported by Medtronic. Dr. Fischer has served as a consultant for Medtronic, Stryker, and CSL Behring. Dr. Sheth has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM WSC 2021
Direct comparison shows differing strengths for left atrial closure devices
On the basis of outcomes, there was no clear winner from a trial that directly compared two modern devices used in patients undergoing percutaneous left atrial appendage (LAA) closure.
But the devices were not interchangeable for rates of complications or leaks, according to results of the open-label SWISS APERO trial, which compared the Amplatzer Amulet to the Watchman FLX device at eight participating centers in Europe.
At 45 days, the overall rates of leaks and the clinical outcomes in the two randomized groups were not significantly different, but there were differences in secondary endpoints, such as rates of peridevice leak (PDL), which were lower in the Amulet device group, and procedural complications, which were higher, Roberto Galea, MD, reported at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando.
LAA closure devices were developed as an alternative to oral anticoagulation in patients with nonvalvular atrial fibrillation. Although a similar comparison of LAA closure devices, called Amulet IDE, was recently published, that trial compared Amulet to Watchman 2.5, an earlier generation device.
Started later, SWISS APERO was also a planned comparison of Amulet and the Watchman 2.5, but the comparison switched to the Watchman FLX, when it was released in March of 2019.
First randomized comparison with Watchman FLX
“This is the first multicenter randomized controlled trial to include the Watchman FLX,” said Dr. Galea, a clinical investigator in the department of cardiology, Bern (Switzerland) University Hospital. He noted that Watchman FLX included some adjustments in design with the potential to reduce leak rates.
After preprocedural transesophageal echocardiography confirmed that patients had suitable anatomy to receive either device, the 221 patients who qualified for SWISS APERO were randomized. The primary endpoint was a composite of a justified crossover to a device other than the one to which they were assigned or residual patency detected by coronary computed tomography angiography (CCTA) at 45 days.
The primary endpoint was reached by 67.6% of patients randomized to the Amulet device and 70% of those randomized to Watchman Flex, a statistically nonsignificant difference (P = .71).
Because only one patient in the Amulet group and none in the Watchman group had a justified crossover to a nonrandomized device, most of the differences in the 45-day CCTA involved patency, defined as LAA density of at least 100 Hounsfield units. While the proportion of patients with leaks was similar, the types of leaks, which were stratified by underlying leak mechanism into PDL, mixed leaks (including incomplete side sealing), intradevice leaks, and leaks of unclear origin, were different.
Peridevice leaks twofold greater with Watchman
Those randomized to the Watchman device were more than twice as likely to have PDL (27.5% vs. 13.7%; P = .02), although no visible leak exceeded 5 mm in size. They were also more likely to have mixed leaks (14% vs. 3.8%; P = .01) and patency with no visible leak (21.0% vs. 9.5%; P = .02). There were also more device-related thrombi in the Watchman group even though the difference did not reach statistical significance (9.9% vs. 3.7%; P = .08).
Intradevice leaks (44.8% vs. 23.0%; P = .001) were the only type of patency significantly more common among patients randomized to Amulet, but the difference was relatively large. In addition, procedural complications of any type (32.4% vs. 19.1%; P = .023) were higher in the Amulet group. Most of these involved non–clinically relevant pericardial effusions, Dr. Galea said at the meeting, sponsored by the Cardiovascular Research Foundation.
The proportion of patients with adverse outcomes by 45 days was similar, but the types of complications differed. Of the six deaths, two occurred in the Amulet group as a result of periprocedural complications (one stemming from an air embolism and the other from a series of events following pericardial effusion). Three of the four deaths in the Watchman group were due to fatal bleeding. The fourth was a sudden death that occurred 30 days after the procedure.
Amulet IDE trial generates similar data
The much larger Amulet IDE trial, which compared Amulet to the Watchman 2.5 device, produced generally similar results. Again, the proportion of patients reaching the composite primary endpoints was similar.
The primary safety endpoint, which included death and major bleeding within 12 months of randomization, occurred in 14.5% and 14.7% of the Amulet and Watchman patients, respectively (P < .001 for noninferiority). The primary efficacy endpoint, which included stroke or systemic embolism within 18 months of randomization, occurred in 2.8% of patients in both groups.
As in SWISS APERO, the 1,878-patient Amulet IDE trial showed that the devices are similarly effective and safe but not necessarily interchangeable. Ultimately, the rate of LAA occlusion was higher for Amulet than the older generation Watchman (98.9% vs. 96.8%; P = .003) but procedural complication occurred more frequently among those randomized to the Amulet device (4.5% vs. 2.5%).
“The closure mechanisms are not the same, which might explain why we see differences in some secondary outcomes even when they perform similarly on the primary outcomes,” said Dhanunjaya R. Lakkireddy, MD, executive medical director, Kansas City (Kansas) Heart Rhythm Institute.
The lead investigator of the Amulet IDE trial, Dr. Lakkireddy was referring to both the AMULET IDE and the SWISS APERO study when he said that the currently available data do not allow one device to be considered superior to the other. He did suggest that differences between devices might still be considered meaningful in specific clinical situations or to specific clinicians.
Without studies to show objective differences, Dr. Lakkireddy suggested that training and experience is probably the most important variable in achieving treatment goals. “Operator comfort is certainly important,” he said.
Dr. Galea reports no significant financial relationships. The investigator-initiated study received funding from Abbott, the manufacturer of the Amulet device. Dr. Lakkireddy has financial relationships with Abbott, AltaThera, Medtronic, Biotronik, and Boston Scientific, which makes the Watchman device.
On the basis of outcomes, there was no clear winner from a trial that directly compared two modern devices used in patients undergoing percutaneous left atrial appendage (LAA) closure.
But the devices were not interchangeable for rates of complications or leaks, according to results of the open-label SWISS APERO trial, which compared the Amplatzer Amulet to the Watchman FLX device at eight participating centers in Europe.
At 45 days, the overall rates of leaks and the clinical outcomes in the two randomized groups were not significantly different, but there were differences in secondary endpoints, such as rates of peridevice leak (PDL), which were lower in the Amulet device group, and procedural complications, which were higher, Roberto Galea, MD, reported at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando.
LAA closure devices were developed as an alternative to oral anticoagulation in patients with nonvalvular atrial fibrillation. Although a similar comparison of LAA closure devices, called Amulet IDE, was recently published, that trial compared Amulet to Watchman 2.5, an earlier generation device.
Started later, SWISS APERO was also a planned comparison of Amulet and the Watchman 2.5, but the comparison switched to the Watchman FLX, when it was released in March of 2019.
First randomized comparison with Watchman FLX
“This is the first multicenter randomized controlled trial to include the Watchman FLX,” said Dr. Galea, a clinical investigator in the department of cardiology, Bern (Switzerland) University Hospital. He noted that Watchman FLX included some adjustments in design with the potential to reduce leak rates.
After preprocedural transesophageal echocardiography confirmed that patients had suitable anatomy to receive either device, the 221 patients who qualified for SWISS APERO were randomized. The primary endpoint was a composite of a justified crossover to a device other than the one to which they were assigned or residual patency detected by coronary computed tomography angiography (CCTA) at 45 days.
The primary endpoint was reached by 67.6% of patients randomized to the Amulet device and 70% of those randomized to Watchman Flex, a statistically nonsignificant difference (P = .71).
Because only one patient in the Amulet group and none in the Watchman group had a justified crossover to a nonrandomized device, most of the differences in the 45-day CCTA involved patency, defined as LAA density of at least 100 Hounsfield units. While the proportion of patients with leaks was similar, the types of leaks, which were stratified by underlying leak mechanism into PDL, mixed leaks (including incomplete side sealing), intradevice leaks, and leaks of unclear origin, were different.
Peridevice leaks twofold greater with Watchman
Those randomized to the Watchman device were more than twice as likely to have PDL (27.5% vs. 13.7%; P = .02), although no visible leak exceeded 5 mm in size. They were also more likely to have mixed leaks (14% vs. 3.8%; P = .01) and patency with no visible leak (21.0% vs. 9.5%; P = .02). There were also more device-related thrombi in the Watchman group even though the difference did not reach statistical significance (9.9% vs. 3.7%; P = .08).
Intradevice leaks (44.8% vs. 23.0%; P = .001) were the only type of patency significantly more common among patients randomized to Amulet, but the difference was relatively large. In addition, procedural complications of any type (32.4% vs. 19.1%; P = .023) were higher in the Amulet group. Most of these involved non–clinically relevant pericardial effusions, Dr. Galea said at the meeting, sponsored by the Cardiovascular Research Foundation.
The proportion of patients with adverse outcomes by 45 days was similar, but the types of complications differed. Of the six deaths, two occurred in the Amulet group as a result of periprocedural complications (one stemming from an air embolism and the other from a series of events following pericardial effusion). Three of the four deaths in the Watchman group were due to fatal bleeding. The fourth was a sudden death that occurred 30 days after the procedure.
Amulet IDE trial generates similar data
The much larger Amulet IDE trial, which compared Amulet to the Watchman 2.5 device, produced generally similar results. Again, the proportion of patients reaching the composite primary endpoints was similar.
The primary safety endpoint, which included death and major bleeding within 12 months of randomization, occurred in 14.5% and 14.7% of the Amulet and Watchman patients, respectively (P < .001 for noninferiority). The primary efficacy endpoint, which included stroke or systemic embolism within 18 months of randomization, occurred in 2.8% of patients in both groups.
As in SWISS APERO, the 1,878-patient Amulet IDE trial showed that the devices are similarly effective and safe but not necessarily interchangeable. Ultimately, the rate of LAA occlusion was higher for Amulet than the older generation Watchman (98.9% vs. 96.8%; P = .003) but procedural complication occurred more frequently among those randomized to the Amulet device (4.5% vs. 2.5%).
“The closure mechanisms are not the same, which might explain why we see differences in some secondary outcomes even when they perform similarly on the primary outcomes,” said Dhanunjaya R. Lakkireddy, MD, executive medical director, Kansas City (Kansas) Heart Rhythm Institute.
The lead investigator of the Amulet IDE trial, Dr. Lakkireddy was referring to both the AMULET IDE and the SWISS APERO study when he said that the currently available data do not allow one device to be considered superior to the other. He did suggest that differences between devices might still be considered meaningful in specific clinical situations or to specific clinicians.
Without studies to show objective differences, Dr. Lakkireddy suggested that training and experience is probably the most important variable in achieving treatment goals. “Operator comfort is certainly important,” he said.
Dr. Galea reports no significant financial relationships. The investigator-initiated study received funding from Abbott, the manufacturer of the Amulet device. Dr. Lakkireddy has financial relationships with Abbott, AltaThera, Medtronic, Biotronik, and Boston Scientific, which makes the Watchman device.
On the basis of outcomes, there was no clear winner from a trial that directly compared two modern devices used in patients undergoing percutaneous left atrial appendage (LAA) closure.
But the devices were not interchangeable for rates of complications or leaks, according to results of the open-label SWISS APERO trial, which compared the Amplatzer Amulet to the Watchman FLX device at eight participating centers in Europe.
At 45 days, the overall rates of leaks and the clinical outcomes in the two randomized groups were not significantly different, but there were differences in secondary endpoints, such as rates of peridevice leak (PDL), which were lower in the Amulet device group, and procedural complications, which were higher, Roberto Galea, MD, reported at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando.
LAA closure devices were developed as an alternative to oral anticoagulation in patients with nonvalvular atrial fibrillation. Although a similar comparison of LAA closure devices, called Amulet IDE, was recently published, that trial compared Amulet to Watchman 2.5, an earlier generation device.
Started later, SWISS APERO was also a planned comparison of Amulet and the Watchman 2.5, but the comparison switched to the Watchman FLX, when it was released in March of 2019.
First randomized comparison with Watchman FLX
“This is the first multicenter randomized controlled trial to include the Watchman FLX,” said Dr. Galea, a clinical investigator in the department of cardiology, Bern (Switzerland) University Hospital. He noted that Watchman FLX included some adjustments in design with the potential to reduce leak rates.
After preprocedural transesophageal echocardiography confirmed that patients had suitable anatomy to receive either device, the 221 patients who qualified for SWISS APERO were randomized. The primary endpoint was a composite of a justified crossover to a device other than the one to which they were assigned or residual patency detected by coronary computed tomography angiography (CCTA) at 45 days.
The primary endpoint was reached by 67.6% of patients randomized to the Amulet device and 70% of those randomized to Watchman Flex, a statistically nonsignificant difference (P = .71).
Because only one patient in the Amulet group and none in the Watchman group had a justified crossover to a nonrandomized device, most of the differences in the 45-day CCTA involved patency, defined as LAA density of at least 100 Hounsfield units. While the proportion of patients with leaks was similar, the types of leaks, which were stratified by underlying leak mechanism into PDL, mixed leaks (including incomplete side sealing), intradevice leaks, and leaks of unclear origin, were different.
Peridevice leaks twofold greater with Watchman
Those randomized to the Watchman device were more than twice as likely to have PDL (27.5% vs. 13.7%; P = .02), although no visible leak exceeded 5 mm in size. They were also more likely to have mixed leaks (14% vs. 3.8%; P = .01) and patency with no visible leak (21.0% vs. 9.5%; P = .02). There were also more device-related thrombi in the Watchman group even though the difference did not reach statistical significance (9.9% vs. 3.7%; P = .08).
Intradevice leaks (44.8% vs. 23.0%; P = .001) were the only type of patency significantly more common among patients randomized to Amulet, but the difference was relatively large. In addition, procedural complications of any type (32.4% vs. 19.1%; P = .023) were higher in the Amulet group. Most of these involved non–clinically relevant pericardial effusions, Dr. Galea said at the meeting, sponsored by the Cardiovascular Research Foundation.
The proportion of patients with adverse outcomes by 45 days was similar, but the types of complications differed. Of the six deaths, two occurred in the Amulet group as a result of periprocedural complications (one stemming from an air embolism and the other from a series of events following pericardial effusion). Three of the four deaths in the Watchman group were due to fatal bleeding. The fourth was a sudden death that occurred 30 days after the procedure.
Amulet IDE trial generates similar data
The much larger Amulet IDE trial, which compared Amulet to the Watchman 2.5 device, produced generally similar results. Again, the proportion of patients reaching the composite primary endpoints was similar.
The primary safety endpoint, which included death and major bleeding within 12 months of randomization, occurred in 14.5% and 14.7% of the Amulet and Watchman patients, respectively (P < .001 for noninferiority). The primary efficacy endpoint, which included stroke or systemic embolism within 18 months of randomization, occurred in 2.8% of patients in both groups.
As in SWISS APERO, the 1,878-patient Amulet IDE trial showed that the devices are similarly effective and safe but not necessarily interchangeable. Ultimately, the rate of LAA occlusion was higher for Amulet than the older generation Watchman (98.9% vs. 96.8%; P = .003) but procedural complication occurred more frequently among those randomized to the Amulet device (4.5% vs. 2.5%).
“The closure mechanisms are not the same, which might explain why we see differences in some secondary outcomes even when they perform similarly on the primary outcomes,” said Dhanunjaya R. Lakkireddy, MD, executive medical director, Kansas City (Kansas) Heart Rhythm Institute.
The lead investigator of the Amulet IDE trial, Dr. Lakkireddy was referring to both the AMULET IDE and the SWISS APERO study when he said that the currently available data do not allow one device to be considered superior to the other. He did suggest that differences between devices might still be considered meaningful in specific clinical situations or to specific clinicians.
Without studies to show objective differences, Dr. Lakkireddy suggested that training and experience is probably the most important variable in achieving treatment goals. “Operator comfort is certainly important,” he said.
Dr. Galea reports no significant financial relationships. The investigator-initiated study received funding from Abbott, the manufacturer of the Amulet device. Dr. Lakkireddy has financial relationships with Abbott, AltaThera, Medtronic, Biotronik, and Boston Scientific, which makes the Watchman device.
FROM TCT 2021
Treating young adults with high LDL may be cost-effective
Treating elevated low-density lipoprotein cholesterol (LDL-C) in adults younger than 40 with statins is highly cost-effective in men, and intermediately cost-effective in women, a new report suggests.
In a simulated model based on data from the U.S. National Health and Nutrition Examination Survey (NHANES), lipid lowering with statins or lifestyle interventions in this age group would prevent or reduce the risk of atherosclerotic cardiovascular disease (ASCVD) and improve quality of life in later years.
The findings were published online Nov. 8 in the Journal of the American College of Cardiology.
“My group does epidemiologic analyses with cohort studies as well as health economic analyses like this one, and if you have long-term longitudinal observation, you see that the early exposures are important for what happens later,” senior author Andrew E. Moran, MD, Columbia University Irving Medical Center, New York, told this news organization.
“But when it comes to treatment studies that a lot of the treatment guidelines are based on, those are usually short-term, and they usually enroll older people. We saw the gap in the evidence that this paper tries to fill,” Dr. Moran said.
His group used a computer simulation model to synthesize evidence from observational cohort studies and clinical trials of statin treatment, as well as health services data on the costs of medicines and treatments.
Combining information from these sources, the investigators made their best estimates of the potential health benefits and costs of treating high cholesterol earlier in life, compared with standard care, which was statin treatment at age 40, or if LDL-C was 190 mg/dL or greater.
Lipid lowering incremental to standard care with moderate-intensity statins or intensive lifestyle interventions was simulated starting when young adult LDL-C was either ≥160 mg/dL or ≥130 mg/dL.
They found that approximately 27% of young adults who are free of ASCVD have LDL-C ≥130 mg/dL, and 9% have LDL-C of ≥160 mg/dL.
Their model projected that treating adults younger than 40 with statins or lifestyle interventions would prevent lifetime ASCVD events and increase quality-adjusted life years (QALYs) compared with standard care, which would begin treatment at age 40.
Incremental cost-effectiveness ratios (ICERs) were $31,000/QALY for statin treatment in young adult men with LDL-C ≥130 mg/dL, and $106,000/QALY for statin treatment in young women with LDL-C ≥130 mg/dL.
Intensive lifestyle intervention was more costly and less effective than statin therapy.
“We are straining to find these young adults with very high cholesterol,” Dr. Moran noted. “A lot of young adults don’t even see a doctor. This is an argument for engaging them in their health care and getting them involved in some basic screening. Atherosclerosis is a long-term process that starts in childhood for a lot of people.”
More innovative approaches may be needed, because the traditional health care system is not doing a good job of reaching young adults, he added. “Many of them may not have adequate health insurance. They need health care in nontraditional ways; convenience is really important for them. Perhaps part of the solution here is to think about ways of reaching this particular group that is not engaged with health care generally.”
Time to relax the age 40 threshold
The U.S. Preventive Services Task Force and the American College of Cardiology/American Heart Association should emphasize lifetime risk of elevated cholesterol, Paul A. Heidenreich, MD, MS, Stanford University School of Medicine, California, and colleagues write in an accompanying editorial.
“In addition to calculating 10-year risk, we should calculate years of life lost (or QALYs lost) from unhealthy LDL-C levels, and both lifestyle and pharmacologic treatment should be considered to treat high LDL-C in adults regardless of age. We also need to communicate that the mantra ‘lower is better’ applies not only to a single measurement but to lifetime exposure to LDL-C,” the editorialists write.
“I think treatment should be earlier than age 40,” Dr. Heidenreich said in an interview.
“Part of the reason that 40 was chosen as a threshold was because everyone looked at 10-year, or even 20-year risk, and thought there was no reason to worry until you get older. It’s interesting that we never accepted that with high blood pressure. But more and more, we are learning that it is a lifelong process,” he said.
“Statins are getting less and less expensive, and their safety is more and more established with every decade that goes by. I definitely agree with this paper that it would actually make sense to be starting much earlier for those with elevated CVD risk from their high cholesterol.”
The study was supported by the U.S. National Heart, Lung, and Blood Institute (NHLBI), the Medical Research Council, Swindon, U.K. Dr. Moran and Dr. Heidenreich have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Treating elevated low-density lipoprotein cholesterol (LDL-C) in adults younger than 40 with statins is highly cost-effective in men, and intermediately cost-effective in women, a new report suggests.
In a simulated model based on data from the U.S. National Health and Nutrition Examination Survey (NHANES), lipid lowering with statins or lifestyle interventions in this age group would prevent or reduce the risk of atherosclerotic cardiovascular disease (ASCVD) and improve quality of life in later years.
The findings were published online Nov. 8 in the Journal of the American College of Cardiology.
“My group does epidemiologic analyses with cohort studies as well as health economic analyses like this one, and if you have long-term longitudinal observation, you see that the early exposures are important for what happens later,” senior author Andrew E. Moran, MD, Columbia University Irving Medical Center, New York, told this news organization.
“But when it comes to treatment studies that a lot of the treatment guidelines are based on, those are usually short-term, and they usually enroll older people. We saw the gap in the evidence that this paper tries to fill,” Dr. Moran said.
His group used a computer simulation model to synthesize evidence from observational cohort studies and clinical trials of statin treatment, as well as health services data on the costs of medicines and treatments.
Combining information from these sources, the investigators made their best estimates of the potential health benefits and costs of treating high cholesterol earlier in life, compared with standard care, which was statin treatment at age 40, or if LDL-C was 190 mg/dL or greater.
Lipid lowering incremental to standard care with moderate-intensity statins or intensive lifestyle interventions was simulated starting when young adult LDL-C was either ≥160 mg/dL or ≥130 mg/dL.
They found that approximately 27% of young adults who are free of ASCVD have LDL-C ≥130 mg/dL, and 9% have LDL-C of ≥160 mg/dL.
Their model projected that treating adults younger than 40 with statins or lifestyle interventions would prevent lifetime ASCVD events and increase quality-adjusted life years (QALYs) compared with standard care, which would begin treatment at age 40.
Incremental cost-effectiveness ratios (ICERs) were $31,000/QALY for statin treatment in young adult men with LDL-C ≥130 mg/dL, and $106,000/QALY for statin treatment in young women with LDL-C ≥130 mg/dL.
Intensive lifestyle intervention was more costly and less effective than statin therapy.
“We are straining to find these young adults with very high cholesterol,” Dr. Moran noted. “A lot of young adults don’t even see a doctor. This is an argument for engaging them in their health care and getting them involved in some basic screening. Atherosclerosis is a long-term process that starts in childhood for a lot of people.”
More innovative approaches may be needed, because the traditional health care system is not doing a good job of reaching young adults, he added. “Many of them may not have adequate health insurance. They need health care in nontraditional ways; convenience is really important for them. Perhaps part of the solution here is to think about ways of reaching this particular group that is not engaged with health care generally.”
Time to relax the age 40 threshold
The U.S. Preventive Services Task Force and the American College of Cardiology/American Heart Association should emphasize lifetime risk of elevated cholesterol, Paul A. Heidenreich, MD, MS, Stanford University School of Medicine, California, and colleagues write in an accompanying editorial.
“In addition to calculating 10-year risk, we should calculate years of life lost (or QALYs lost) from unhealthy LDL-C levels, and both lifestyle and pharmacologic treatment should be considered to treat high LDL-C in adults regardless of age. We also need to communicate that the mantra ‘lower is better’ applies not only to a single measurement but to lifetime exposure to LDL-C,” the editorialists write.
“I think treatment should be earlier than age 40,” Dr. Heidenreich said in an interview.
“Part of the reason that 40 was chosen as a threshold was because everyone looked at 10-year, or even 20-year risk, and thought there was no reason to worry until you get older. It’s interesting that we never accepted that with high blood pressure. But more and more, we are learning that it is a lifelong process,” he said.
“Statins are getting less and less expensive, and their safety is more and more established with every decade that goes by. I definitely agree with this paper that it would actually make sense to be starting much earlier for those with elevated CVD risk from their high cholesterol.”
The study was supported by the U.S. National Heart, Lung, and Blood Institute (NHLBI), the Medical Research Council, Swindon, U.K. Dr. Moran and Dr. Heidenreich have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Treating elevated low-density lipoprotein cholesterol (LDL-C) in adults younger than 40 with statins is highly cost-effective in men, and intermediately cost-effective in women, a new report suggests.
In a simulated model based on data from the U.S. National Health and Nutrition Examination Survey (NHANES), lipid lowering with statins or lifestyle interventions in this age group would prevent or reduce the risk of atherosclerotic cardiovascular disease (ASCVD) and improve quality of life in later years.
The findings were published online Nov. 8 in the Journal of the American College of Cardiology.
“My group does epidemiologic analyses with cohort studies as well as health economic analyses like this one, and if you have long-term longitudinal observation, you see that the early exposures are important for what happens later,” senior author Andrew E. Moran, MD, Columbia University Irving Medical Center, New York, told this news organization.
“But when it comes to treatment studies that a lot of the treatment guidelines are based on, those are usually short-term, and they usually enroll older people. We saw the gap in the evidence that this paper tries to fill,” Dr. Moran said.
His group used a computer simulation model to synthesize evidence from observational cohort studies and clinical trials of statin treatment, as well as health services data on the costs of medicines and treatments.
Combining information from these sources, the investigators made their best estimates of the potential health benefits and costs of treating high cholesterol earlier in life, compared with standard care, which was statin treatment at age 40, or if LDL-C was 190 mg/dL or greater.
Lipid lowering incremental to standard care with moderate-intensity statins or intensive lifestyle interventions was simulated starting when young adult LDL-C was either ≥160 mg/dL or ≥130 mg/dL.
They found that approximately 27% of young adults who are free of ASCVD have LDL-C ≥130 mg/dL, and 9% have LDL-C of ≥160 mg/dL.
Their model projected that treating adults younger than 40 with statins or lifestyle interventions would prevent lifetime ASCVD events and increase quality-adjusted life years (QALYs) compared with standard care, which would begin treatment at age 40.
Incremental cost-effectiveness ratios (ICERs) were $31,000/QALY for statin treatment in young adult men with LDL-C ≥130 mg/dL, and $106,000/QALY for statin treatment in young women with LDL-C ≥130 mg/dL.
Intensive lifestyle intervention was more costly and less effective than statin therapy.
“We are straining to find these young adults with very high cholesterol,” Dr. Moran noted. “A lot of young adults don’t even see a doctor. This is an argument for engaging them in their health care and getting them involved in some basic screening. Atherosclerosis is a long-term process that starts in childhood for a lot of people.”
More innovative approaches may be needed, because the traditional health care system is not doing a good job of reaching young adults, he added. “Many of them may not have adequate health insurance. They need health care in nontraditional ways; convenience is really important for them. Perhaps part of the solution here is to think about ways of reaching this particular group that is not engaged with health care generally.”
Time to relax the age 40 threshold
The U.S. Preventive Services Task Force and the American College of Cardiology/American Heart Association should emphasize lifetime risk of elevated cholesterol, Paul A. Heidenreich, MD, MS, Stanford University School of Medicine, California, and colleagues write in an accompanying editorial.
“In addition to calculating 10-year risk, we should calculate years of life lost (or QALYs lost) from unhealthy LDL-C levels, and both lifestyle and pharmacologic treatment should be considered to treat high LDL-C in adults regardless of age. We also need to communicate that the mantra ‘lower is better’ applies not only to a single measurement but to lifetime exposure to LDL-C,” the editorialists write.
“I think treatment should be earlier than age 40,” Dr. Heidenreich said in an interview.
“Part of the reason that 40 was chosen as a threshold was because everyone looked at 10-year, or even 20-year risk, and thought there was no reason to worry until you get older. It’s interesting that we never accepted that with high blood pressure. But more and more, we are learning that it is a lifelong process,” he said.
“Statins are getting less and less expensive, and their safety is more and more established with every decade that goes by. I definitely agree with this paper that it would actually make sense to be starting much earlier for those with elevated CVD risk from their high cholesterol.”
The study was supported by the U.S. National Heart, Lung, and Blood Institute (NHLBI), the Medical Research Council, Swindon, U.K. Dr. Moran and Dr. Heidenreich have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AHA 2021 puts scientific dialogue, health equity center stage
Virtual platforms democratized scientific meetings during the COVID-19 pandemic but, as any meeting-goer will tell you, it’s the questions from the floor and the back-and-forth of an expert panel that often reveal the importance of and/or problems with a presentation. It’s the scrutiny that makes the science resonate, especially in this postfactual era.
The all-virtual American Heart Association Scientific Sessions 2021 is looking to recreate the engagement of an in-person meeting by offering more live interactive events. They range from seven late-breaking science (LBS) sessions to Saturday’s fireside chat on the Pfizer and Moderna COVID-19 vaccines and Monday’s dive into the controversial new AHA/American College of Cardiology Chest Pain guidelines.
To help digest the latest science, attendees will be able to have their questions answered in real-time via Slido, meet with the trialists, and hear live commentary from key opinion leaders after the live events. A networking function will also allow attendees and exhibitors to chat or meet virtually.
“In this day and age, many people pretty quickly can get access to the science but it’s what I call the IC sort of phenomenon – the presentation of the information, the context of the information, putting it into how I’m going to use it in my practice, and then the critical appraisal – that’s what most people want at the Scientific Sessions,” program committee chair Manesh R. Patel, MD, of Duke University School of Medicine, said in an interview. “We’re all craving ways in which we can interact with one another to put things in context.”
Plans for a hybrid in-person meeting in Boston were scuttled in September because of the Delta variant surge, but the theme of the meeting remained: “One World. Together for Science.” Attendees will be able to access more than 500 live and on-demand sessions including 117 oral abstracts, 286 poster sessions, 59 moderated digital posters, and over a dozen sessions focused on strategies to promote health equity.
“Last year there was a Presidential Session and a statement on structural racism, so we wanted to take the next step and say, What are the ways in which people are starting to interact and do things to make a difference?” explained Dr. Patel. “So, this year, you’ll see different versions of that from the Main Event session, which has some case vignettes and a panel discussion, to other health equity sessions that describe not just COVID care, but blood pressure care, maternal-fetal medicine, and congenital kids. Wherever we can, we’ve tried to infuse it throughout the sessions and will continue to.”
Late-breaking science
The LBS sessions kick off at 9:30 a.m. ET Saturday with AVATAR, a randomized trial of aortic valve replacement vs. watchful waiting in severe aortic stenosis proved asymptomatic through exercise testing.
“The findings of that trial, depending on what they are, could certainly impact clinical practice because it’s a very common scenario in which we have elderly patients with aortic valve stenosis that might be severe but they may not be symptomatic,” he said.
It’s followed by a randomized trial from the Cardiothoracic Surgical Trials Network, examining whether tricuspid repair at the time of mitral valve surgery leads to beneficial outcomes. “I think it’s a pretty important study,” Dr. Patel said, “because it’ll again affect how we think about our clinical practice.”
Rounding out the LBS.01 session is RAPID CABG, comparing early vs. delayed coronary bypass graft surgery (CABG) in patients with acute coronary syndromes on ticagrelor, and the pivotal U.S. VEST trial of an external support device already approved in Europe for saphenous vein grafts during CABG.
Saturday’s LBS.02 at 3:00 p.m. ET is devoted to hypertension and looks at how the COVID-19 pandemic affected blood pressure control. There’s also a study of remotely delivered hypertension and lipid management in 10,000 patients across the Partners Healthcare System and a cluster randomized trial of a village doctor–led blood pressure intervention in rural China.
Sunday’s LBS.03 at 8:00 a.m. ET is focused on atrial arrhythmias, starting with the CRAVE trial examining the effect of caffeine consumption on cardiac ectopy burden in 108 patients using an N-of-1 design and 2-day blocks on and off caffeine. “There’s an ability to identify a dose response that you get arrhythmias when you increase the amount of coffee you drink vs. not in an individual, so I think that will be likely discussed a lot and worth paying attention to,” Dr. Patel said.
The session also includes GIRAF, a comparison of cognitive outcomes with dabigatran (Pradaxa) vs. warfarin (Coumadin) in nonvalvular atrial fibrillation (AF); PALACS, a randomized trial examining whether left-sided pericardiotomy prevents AF after cardiac surgery; and AMAZE, which study sponsor AtriCure revealed missed its primary efficacy endpoint of freedom from AF with the LARIAT suture delivery device for left atrial appendage closure plus pulmonary vein isolation.
LBS.04 at 3:30 p.m. ET Sunday takes on digital health, with results from the nonrandomized Fitbit Heart Study on AF notifications from 450,000 participants wearing a single-lead ECG patch. “A lot of technologies claim that they can detect things, and we should ask that people go through the rigorous evaluation to see if they in fact do. So, in that respect, I think it›s an important step,” observed Dr. Patel.
Also on tap is I-STOP-AFib, another N-of-1 study using mobile apps and the AliveCor device to identify individual AF triggers; and REVeAL-HF, a 4,000-patient study examining whether electronic alerts that provide clinicians with prognostic information on their heart failure (HF) patients will reduce mortality and 30-day HF hospitalizations.
LBS.05 at 5:00 p.m. ET provides new information from EMPEROR-Preserved in HF with preserved ejection fraction and main results from EMPULSE, also using the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in 530 patients hospitalized for acute HF.
The session also features CHIEF-HF, a randomized trial leveraging mobile technologies to test whether 12 weeks of another SGLT2 inhibitor, canagliflozin (Invokana), is superior to placebo for improving HF symptoms; and DREAM-HF, a comparison of transendocardial delivery of allogeneic mesenchymal precursor cells vs. a sham comparator in chronic HF as a result of left ventricular systolic dysfunction.
Monday’s LBS.06 at 8:00 a.m. ET details the safety and cholesterol-lowering efficacy of MK-0616, an investigational oral PCSK9 inhibitor. “It’s just a phase 2 [trial], but there’s interest in an oral PCSK9 inhibitor, given that the current ones are subcutaneous,” Dr. Patel said.
Results will also be presented from PREPARE-IT 2, which tested icosapent ethyl vs. placebo in outpatients with COVID-19. In the recently reported PREPARE-IT 1, a loading dose of icosapent ethyl failed to reduce the risk of hospitalization with SARS-CoV-2 infection among at-risk individuals.
LBS.07 at 11:00 a.m. Monday completes the late-breakers with new results from ASCEND, this time examining the effect of aspirin on dementia and cognitive impairment in patients with diabetes.
Next up is a look at the effectiveness of P2Y12 inhibitors in hospitalized patients with COVID-19 in the adaptive ACTIV-4a trial, followed by results of the pivotal phase 3 REVERSE-IT trial of bentracimab, a recombinant human monoclonal antibody antigen fragment designed to reverse the antiplatelet activity of ticagrelor in the event of major bleeding or when urgent surgery is needed.
Closing out the session is AXIOMATIC-TKR, a double-blind comparison of the safety and efficacy of the investigational oral factor XI anticoagulant JNJ-70033093 vs. subcutaneous enoxaparin (Lovenox) in elective total knee replacement.
For those searching for more AHA-related science online, the Resuscitation Science Symposium (ReSS) will run from this Friday through Sunday and the Quality of Care and Outcomes Research (QCOR) Scientific Sessions will take the stage next Monday, Nov. 15.
A version of this article first appeared on Medscape.com.
Virtual platforms democratized scientific meetings during the COVID-19 pandemic but, as any meeting-goer will tell you, it’s the questions from the floor and the back-and-forth of an expert panel that often reveal the importance of and/or problems with a presentation. It’s the scrutiny that makes the science resonate, especially in this postfactual era.
The all-virtual American Heart Association Scientific Sessions 2021 is looking to recreate the engagement of an in-person meeting by offering more live interactive events. They range from seven late-breaking science (LBS) sessions to Saturday’s fireside chat on the Pfizer and Moderna COVID-19 vaccines and Monday’s dive into the controversial new AHA/American College of Cardiology Chest Pain guidelines.
To help digest the latest science, attendees will be able to have their questions answered in real-time via Slido, meet with the trialists, and hear live commentary from key opinion leaders after the live events. A networking function will also allow attendees and exhibitors to chat or meet virtually.
“In this day and age, many people pretty quickly can get access to the science but it’s what I call the IC sort of phenomenon – the presentation of the information, the context of the information, putting it into how I’m going to use it in my practice, and then the critical appraisal – that’s what most people want at the Scientific Sessions,” program committee chair Manesh R. Patel, MD, of Duke University School of Medicine, said in an interview. “We’re all craving ways in which we can interact with one another to put things in context.”
Plans for a hybrid in-person meeting in Boston were scuttled in September because of the Delta variant surge, but the theme of the meeting remained: “One World. Together for Science.” Attendees will be able to access more than 500 live and on-demand sessions including 117 oral abstracts, 286 poster sessions, 59 moderated digital posters, and over a dozen sessions focused on strategies to promote health equity.
“Last year there was a Presidential Session and a statement on structural racism, so we wanted to take the next step and say, What are the ways in which people are starting to interact and do things to make a difference?” explained Dr. Patel. “So, this year, you’ll see different versions of that from the Main Event session, which has some case vignettes and a panel discussion, to other health equity sessions that describe not just COVID care, but blood pressure care, maternal-fetal medicine, and congenital kids. Wherever we can, we’ve tried to infuse it throughout the sessions and will continue to.”
Late-breaking science
The LBS sessions kick off at 9:30 a.m. ET Saturday with AVATAR, a randomized trial of aortic valve replacement vs. watchful waiting in severe aortic stenosis proved asymptomatic through exercise testing.
“The findings of that trial, depending on what they are, could certainly impact clinical practice because it’s a very common scenario in which we have elderly patients with aortic valve stenosis that might be severe but they may not be symptomatic,” he said.
It’s followed by a randomized trial from the Cardiothoracic Surgical Trials Network, examining whether tricuspid repair at the time of mitral valve surgery leads to beneficial outcomes. “I think it’s a pretty important study,” Dr. Patel said, “because it’ll again affect how we think about our clinical practice.”
Rounding out the LBS.01 session is RAPID CABG, comparing early vs. delayed coronary bypass graft surgery (CABG) in patients with acute coronary syndromes on ticagrelor, and the pivotal U.S. VEST trial of an external support device already approved in Europe for saphenous vein grafts during CABG.
Saturday’s LBS.02 at 3:00 p.m. ET is devoted to hypertension and looks at how the COVID-19 pandemic affected blood pressure control. There’s also a study of remotely delivered hypertension and lipid management in 10,000 patients across the Partners Healthcare System and a cluster randomized trial of a village doctor–led blood pressure intervention in rural China.
Sunday’s LBS.03 at 8:00 a.m. ET is focused on atrial arrhythmias, starting with the CRAVE trial examining the effect of caffeine consumption on cardiac ectopy burden in 108 patients using an N-of-1 design and 2-day blocks on and off caffeine. “There’s an ability to identify a dose response that you get arrhythmias when you increase the amount of coffee you drink vs. not in an individual, so I think that will be likely discussed a lot and worth paying attention to,” Dr. Patel said.
The session also includes GIRAF, a comparison of cognitive outcomes with dabigatran (Pradaxa) vs. warfarin (Coumadin) in nonvalvular atrial fibrillation (AF); PALACS, a randomized trial examining whether left-sided pericardiotomy prevents AF after cardiac surgery; and AMAZE, which study sponsor AtriCure revealed missed its primary efficacy endpoint of freedom from AF with the LARIAT suture delivery device for left atrial appendage closure plus pulmonary vein isolation.
LBS.04 at 3:30 p.m. ET Sunday takes on digital health, with results from the nonrandomized Fitbit Heart Study on AF notifications from 450,000 participants wearing a single-lead ECG patch. “A lot of technologies claim that they can detect things, and we should ask that people go through the rigorous evaluation to see if they in fact do. So, in that respect, I think it›s an important step,” observed Dr. Patel.
Also on tap is I-STOP-AFib, another N-of-1 study using mobile apps and the AliveCor device to identify individual AF triggers; and REVeAL-HF, a 4,000-patient study examining whether electronic alerts that provide clinicians with prognostic information on their heart failure (HF) patients will reduce mortality and 30-day HF hospitalizations.
LBS.05 at 5:00 p.m. ET provides new information from EMPEROR-Preserved in HF with preserved ejection fraction and main results from EMPULSE, also using the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in 530 patients hospitalized for acute HF.
The session also features CHIEF-HF, a randomized trial leveraging mobile technologies to test whether 12 weeks of another SGLT2 inhibitor, canagliflozin (Invokana), is superior to placebo for improving HF symptoms; and DREAM-HF, a comparison of transendocardial delivery of allogeneic mesenchymal precursor cells vs. a sham comparator in chronic HF as a result of left ventricular systolic dysfunction.
Monday’s LBS.06 at 8:00 a.m. ET details the safety and cholesterol-lowering efficacy of MK-0616, an investigational oral PCSK9 inhibitor. “It’s just a phase 2 [trial], but there’s interest in an oral PCSK9 inhibitor, given that the current ones are subcutaneous,” Dr. Patel said.
Results will also be presented from PREPARE-IT 2, which tested icosapent ethyl vs. placebo in outpatients with COVID-19. In the recently reported PREPARE-IT 1, a loading dose of icosapent ethyl failed to reduce the risk of hospitalization with SARS-CoV-2 infection among at-risk individuals.
LBS.07 at 11:00 a.m. Monday completes the late-breakers with new results from ASCEND, this time examining the effect of aspirin on dementia and cognitive impairment in patients with diabetes.
Next up is a look at the effectiveness of P2Y12 inhibitors in hospitalized patients with COVID-19 in the adaptive ACTIV-4a trial, followed by results of the pivotal phase 3 REVERSE-IT trial of bentracimab, a recombinant human monoclonal antibody antigen fragment designed to reverse the antiplatelet activity of ticagrelor in the event of major bleeding or when urgent surgery is needed.
Closing out the session is AXIOMATIC-TKR, a double-blind comparison of the safety and efficacy of the investigational oral factor XI anticoagulant JNJ-70033093 vs. subcutaneous enoxaparin (Lovenox) in elective total knee replacement.
For those searching for more AHA-related science online, the Resuscitation Science Symposium (ReSS) will run from this Friday through Sunday and the Quality of Care and Outcomes Research (QCOR) Scientific Sessions will take the stage next Monday, Nov. 15.
A version of this article first appeared on Medscape.com.
Virtual platforms democratized scientific meetings during the COVID-19 pandemic but, as any meeting-goer will tell you, it’s the questions from the floor and the back-and-forth of an expert panel that often reveal the importance of and/or problems with a presentation. It’s the scrutiny that makes the science resonate, especially in this postfactual era.
The all-virtual American Heart Association Scientific Sessions 2021 is looking to recreate the engagement of an in-person meeting by offering more live interactive events. They range from seven late-breaking science (LBS) sessions to Saturday’s fireside chat on the Pfizer and Moderna COVID-19 vaccines and Monday’s dive into the controversial new AHA/American College of Cardiology Chest Pain guidelines.
To help digest the latest science, attendees will be able to have their questions answered in real-time via Slido, meet with the trialists, and hear live commentary from key opinion leaders after the live events. A networking function will also allow attendees and exhibitors to chat or meet virtually.
“In this day and age, many people pretty quickly can get access to the science but it’s what I call the IC sort of phenomenon – the presentation of the information, the context of the information, putting it into how I’m going to use it in my practice, and then the critical appraisal – that’s what most people want at the Scientific Sessions,” program committee chair Manesh R. Patel, MD, of Duke University School of Medicine, said in an interview. “We’re all craving ways in which we can interact with one another to put things in context.”
Plans for a hybrid in-person meeting in Boston were scuttled in September because of the Delta variant surge, but the theme of the meeting remained: “One World. Together for Science.” Attendees will be able to access more than 500 live and on-demand sessions including 117 oral abstracts, 286 poster sessions, 59 moderated digital posters, and over a dozen sessions focused on strategies to promote health equity.
“Last year there was a Presidential Session and a statement on structural racism, so we wanted to take the next step and say, What are the ways in which people are starting to interact and do things to make a difference?” explained Dr. Patel. “So, this year, you’ll see different versions of that from the Main Event session, which has some case vignettes and a panel discussion, to other health equity sessions that describe not just COVID care, but blood pressure care, maternal-fetal medicine, and congenital kids. Wherever we can, we’ve tried to infuse it throughout the sessions and will continue to.”
Late-breaking science
The LBS sessions kick off at 9:30 a.m. ET Saturday with AVATAR, a randomized trial of aortic valve replacement vs. watchful waiting in severe aortic stenosis proved asymptomatic through exercise testing.
“The findings of that trial, depending on what they are, could certainly impact clinical practice because it’s a very common scenario in which we have elderly patients with aortic valve stenosis that might be severe but they may not be symptomatic,” he said.
It’s followed by a randomized trial from the Cardiothoracic Surgical Trials Network, examining whether tricuspid repair at the time of mitral valve surgery leads to beneficial outcomes. “I think it’s a pretty important study,” Dr. Patel said, “because it’ll again affect how we think about our clinical practice.”
Rounding out the LBS.01 session is RAPID CABG, comparing early vs. delayed coronary bypass graft surgery (CABG) in patients with acute coronary syndromes on ticagrelor, and the pivotal U.S. VEST trial of an external support device already approved in Europe for saphenous vein grafts during CABG.
Saturday’s LBS.02 at 3:00 p.m. ET is devoted to hypertension and looks at how the COVID-19 pandemic affected blood pressure control. There’s also a study of remotely delivered hypertension and lipid management in 10,000 patients across the Partners Healthcare System and a cluster randomized trial of a village doctor–led blood pressure intervention in rural China.
Sunday’s LBS.03 at 8:00 a.m. ET is focused on atrial arrhythmias, starting with the CRAVE trial examining the effect of caffeine consumption on cardiac ectopy burden in 108 patients using an N-of-1 design and 2-day blocks on and off caffeine. “There’s an ability to identify a dose response that you get arrhythmias when you increase the amount of coffee you drink vs. not in an individual, so I think that will be likely discussed a lot and worth paying attention to,” Dr. Patel said.
The session also includes GIRAF, a comparison of cognitive outcomes with dabigatran (Pradaxa) vs. warfarin (Coumadin) in nonvalvular atrial fibrillation (AF); PALACS, a randomized trial examining whether left-sided pericardiotomy prevents AF after cardiac surgery; and AMAZE, which study sponsor AtriCure revealed missed its primary efficacy endpoint of freedom from AF with the LARIAT suture delivery device for left atrial appendage closure plus pulmonary vein isolation.
LBS.04 at 3:30 p.m. ET Sunday takes on digital health, with results from the nonrandomized Fitbit Heart Study on AF notifications from 450,000 participants wearing a single-lead ECG patch. “A lot of technologies claim that they can detect things, and we should ask that people go through the rigorous evaluation to see if they in fact do. So, in that respect, I think it›s an important step,” observed Dr. Patel.
Also on tap is I-STOP-AFib, another N-of-1 study using mobile apps and the AliveCor device to identify individual AF triggers; and REVeAL-HF, a 4,000-patient study examining whether electronic alerts that provide clinicians with prognostic information on their heart failure (HF) patients will reduce mortality and 30-day HF hospitalizations.
LBS.05 at 5:00 p.m. ET provides new information from EMPEROR-Preserved in HF with preserved ejection fraction and main results from EMPULSE, also using the sodium-glucose cotransporter 2 (SGLT2) inhibitor empagliflozin (Jardiance) in 530 patients hospitalized for acute HF.
The session also features CHIEF-HF, a randomized trial leveraging mobile technologies to test whether 12 weeks of another SGLT2 inhibitor, canagliflozin (Invokana), is superior to placebo for improving HF symptoms; and DREAM-HF, a comparison of transendocardial delivery of allogeneic mesenchymal precursor cells vs. a sham comparator in chronic HF as a result of left ventricular systolic dysfunction.
Monday’s LBS.06 at 8:00 a.m. ET details the safety and cholesterol-lowering efficacy of MK-0616, an investigational oral PCSK9 inhibitor. “It’s just a phase 2 [trial], but there’s interest in an oral PCSK9 inhibitor, given that the current ones are subcutaneous,” Dr. Patel said.
Results will also be presented from PREPARE-IT 2, which tested icosapent ethyl vs. placebo in outpatients with COVID-19. In the recently reported PREPARE-IT 1, a loading dose of icosapent ethyl failed to reduce the risk of hospitalization with SARS-CoV-2 infection among at-risk individuals.
LBS.07 at 11:00 a.m. Monday completes the late-breakers with new results from ASCEND, this time examining the effect of aspirin on dementia and cognitive impairment in patients with diabetes.
Next up is a look at the effectiveness of P2Y12 inhibitors in hospitalized patients with COVID-19 in the adaptive ACTIV-4a trial, followed by results of the pivotal phase 3 REVERSE-IT trial of bentracimab, a recombinant human monoclonal antibody antigen fragment designed to reverse the antiplatelet activity of ticagrelor in the event of major bleeding or when urgent surgery is needed.
Closing out the session is AXIOMATIC-TKR, a double-blind comparison of the safety and efficacy of the investigational oral factor XI anticoagulant JNJ-70033093 vs. subcutaneous enoxaparin (Lovenox) in elective total knee replacement.
For those searching for more AHA-related science online, the Resuscitation Science Symposium (ReSS) will run from this Friday through Sunday and the Quality of Care and Outcomes Research (QCOR) Scientific Sessions will take the stage next Monday, Nov. 15.
A version of this article first appeared on Medscape.com.
FROM AHA 2021
Long QT and Cardiac Arrest After Symptomatic Improvement of Pulmonary Edema
A case of extreme QT prolongation induced following symptomatic resolution of acute pulmonary edema is both relatively unknown and poorly understood.
Abnormalities in the T-wave morphology of an electrocardiogram (ECG) are classically attributed to ischemic cardiac disease. However, these changes can be seen in a variety of other etiologies, including noncardiac pathology, which should be considered whenever reviewing an ECG: central nervous system disease, including stroke and subarachnoid hemorrhage; hypothermia; pulmonary disease, such as pulmonary embolism or chronic obstructive pulmonary disease; myopericarditis; drug effects; and electrolyte abnormalities.
Prolongation of the QT interval, on the other hand, can be precipitated by medications, metabolic derangements, or genetic phenotypes. The QT interval is measured from the beginning of the QRS complex to the termination of the T wave and represents the total time for ventricular depolarization and repolarization. The QT interval must be corrected based on the patient’s heart rate, known as the QTc. As the QTc interval lengthens, there is increased risk of R-on-T phenomena, which may result in Torsades de Pointes (TdP). Typical features of TdP include an antecedent prolonged QTc, cyclic polymorphic ventricular tachycardia on the surface ECG, and either a short-lived spontaneously terminating course or degeneration into ventricular fibrillation (VF) and sudden cardiac death.1 These dysrhythmias become more likely as the QTc interval exceeds 500 msec.2
The combination of new-onset global T-wave inversions with prolongation of the QT interval has been reported in only a few limited conditions. Some known causes of these QT T changes include cardiac ischemia, status epilepticus, pheochromocytoma, and acute cocaine intoxication.3 One uncommon and rarely reported cause of extreme QT prolongation and T-wave inversion is acute pulmonary edema. The ECG findings are not present on initial patient presentation; rather the dynamic changes occur after resolution of the pulmonary symptoms. Despite significant ECG changes, all prior reported cases describe ECG normalization without significant morbidity.4,5 We report a case of extreme QT prolongation following acute pulmonary edema that resulted in cardiac arrest secondary to VF.
Case Presentation
A 72-year-old male with medical history of combined systolic and diastolic heart failure, ischemic cardiomyopathy, coronary artery disease, cerebral vascular accident, hypertension, hyperlipidemia, type 2 diabetes mellitus, and tobacco dependence presented to the emergency department (ED) by emergency medical services after awaking with acute onset of dyspnea and diaphoresis. On arrival at the ED, the patient was noted to be in respiratory distress (ie, unable to speak single words) and was extremely diaphoretic. His initial vital signs included blood pressure, 186/113 mm Hg, heart rate, 104 beats per minute, respiratory rate, 40 breaths per minute, and temperature, 36.4 °C. The patient was quickly placed on bilevel positive airway pressure and given sublingual nitroglycerin followed by transdermal nitroglycerin with a single dose of 40 mg IV furosemide, which improved his respiratory status. A chest X-ray was consistent with pulmonary edema, and his brain natriuretic peptide was 1654 pg/mL. An ECG demonstrated new T-wave inversions, and his troponin increased from 0.04 to 0.24 ng/mL during his ED stay (Figure 1). He was started on a heparin infusion and admitted to the hospital for hypertensive emergency with presumed acute decompensated heart failure and non-ST-elevated myocardial infarction.
Throughout the patient’s first night, the troponin level started to down-trend after peaking at 0.24 ng/mL, and his oxygen requirements decreased allowing transition to nasal cannula. However, his repeat ECGs demonstrated significant T-wave abnormalities, new premature ventricular contractions, bradycardia, and a prolonging QTc interval to 703 msec (Figure 2). At this time, the patient’s electrolytes were normal, specifically a potassium level of 4.4 mEq/L, calcium 8.8 mg/dL, magnesium 2.0 mg/dL, and phosphorus 2.6 mg/dL. Given the worsening ECG changes, a computed tomography scan of his head was ordered to rule out intracranial pathology. While in the scanner, the patient went into pulseless VF, prompting defibrillation with 200 J. In addition, he was given 75 mg IV lidocaine, 2 g IV magnesium, and 1 ampule of both calcium chloride and sodium bicarbonate. With treatment, he had return of spontaneous circulation and was taken promptly to cardiac catheterization. The catheterization showed no significant obstructive coronary artery disease, and no interventions were performed. The patient was transferred to the cardiac intensive care unit for continued care.
During his course in the intensive care unit, the patient’s potassium and magnesium levels were maintained at high-normal levels. The patient was started on a dobutamine infusion to increase his heart rate and attempt to decrease his QTc. The patient also underwent cardiac magnetic resonance imaging (MRI) to evaluate for possible myocarditis, which showed no evidence of acute inflammation. Echocardiogram demonstrated an ejection fraction of 40% and global hypokinesis but no specific regional abnormalities and no change from prior echocardiogram performed 1 year earlier. Over the course of 3 days, his ECG normalized and his QTc shortened to 477 msec. Genetic testing was performed and did not reveal any mutations associated with long QT syndrome. Ultimately, an automated internal cardiac defibrillator (AICD) was placed, and the patient was discharged home.
Over the 2 years since his initial event, the patient has not experienced recurrent VF and his AICD has not fired. The patient continues to have ED presentations for heart-failure symptoms, though he has been stable from an electrophysiologic standpoint and his QTc remains less than 500 msec.
Discussion
Prolongation of the QT interval as a result of deep, global T-wave inversions after resolution of acute pulmonary edema has been minimally reported.4,5 This phenomenon has been described in the cardiology literature but has not been discussed in the emergency medicine literature and bears consideration in this case.4,5 As noted, an extensive evaluation did not reveal another cause of QTc prolongation. The patient had normal electrolytes and temperature, his neurologic examination and computed tomography were not remarkable. The patient had no obstructive coronary artery disease on catheterization, no evidence of acute myocarditis on cardiac MRI, no prescribed medications associated with QT prolongation, and no evidence of genetic mutations associated with QT prolongation on testing. The minimal troponin elevation was felt to represent a type II myocardial infarction related to ischemia due to supply-demand mismatch rather than acute plaque rupture.
Littmann published a case series of 9 cases of delayed onset T-wave inversion and extreme QTc prolongation in the 24 to 48 hours following treatment and symptomatic improvement in acute pulmonary edema.4 In each of his patients, an ischemic cardiac insult was ruled out as the etiology of the pulmonary edema by laboratory assessment, echocardiography, and left heart catheterization.All of the patients in this case series recovered without incident and with normalization of the QTc interval.4 Similarly, in our patient, significant QT T changes occurred approximately 22 hours after presentation and with resolution of symptoms of pulmonary edema. Pascale and colleagues also published a series of 3 patients developing similar ECG patterns following a hypertensive crisis with resolution of ECG findings and without any morbidity.5 In contrast, our patient experienced significant morbidity secondary to the extreme QTc prolongation.
Conclusions
We believe this is the first reported case of excessive prolongation of the QTc with VF arrest secondary to resolution of acute pulmonary edema. The pattern observed in our patient follows the patterns outlined in the previous case series—patients present with acute pulmonary edema and hypertensive crisis but develop significant ECG abnormalities about 24 hours after the resolution of the high catecholamine state. Our patient did have a history of prior cardiac insult, given the QTc changes developed acutely, with frequent premature ventricular contractions, and the cardiac arrest occurred at maximal QTc prolongation, yet after resolution of the high catecholamine state, the treatment team felt there was likely an uncaptured and short-lived episode of TdP that degenerated into VF. This theory is further supported by the lack of recurrent VF episodes, confirmed by AICD interrogation, after normalization of the QTc in our patient.
1. Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am. 2001;85(2):321-341. doi:10.1016/s0025-7125(05)70318-7
2. Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE. Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal. 2012;2012:212178. doi:10.1100/2012/212178
3. Miller MA, Elmariah S, Fischer A. Giant T-wave inversions and extreme QT prolongation. Circ Arrhythm Electrophysiol. 2009;2(6):e42-e43. doi:10.1161/CIRCEP.108.825729
4. Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: a report of nine cases. J Am Coll Cardiol. 1999;34(4):1106-1110. doi:10.1016/s0735-1097(99)00311-3
5. Pascale P, Quartenoud B, Stauffer JC. Isolated large inverted T wave in pulmonary edema due to hypertensive crisis: a novel electrocardiographic phenomenon mimicking ischemia?. Clin Res Cardiol. 2007;96(5):288-294. doi:10.1007/s00392-007-0504-1
A case of extreme QT prolongation induced following symptomatic resolution of acute pulmonary edema is both relatively unknown and poorly understood.
A case of extreme QT prolongation induced following symptomatic resolution of acute pulmonary edema is both relatively unknown and poorly understood.
Abnormalities in the T-wave morphology of an electrocardiogram (ECG) are classically attributed to ischemic cardiac disease. However, these changes can be seen in a variety of other etiologies, including noncardiac pathology, which should be considered whenever reviewing an ECG: central nervous system disease, including stroke and subarachnoid hemorrhage; hypothermia; pulmonary disease, such as pulmonary embolism or chronic obstructive pulmonary disease; myopericarditis; drug effects; and electrolyte abnormalities.
Prolongation of the QT interval, on the other hand, can be precipitated by medications, metabolic derangements, or genetic phenotypes. The QT interval is measured from the beginning of the QRS complex to the termination of the T wave and represents the total time for ventricular depolarization and repolarization. The QT interval must be corrected based on the patient’s heart rate, known as the QTc. As the QTc interval lengthens, there is increased risk of R-on-T phenomena, which may result in Torsades de Pointes (TdP). Typical features of TdP include an antecedent prolonged QTc, cyclic polymorphic ventricular tachycardia on the surface ECG, and either a short-lived spontaneously terminating course or degeneration into ventricular fibrillation (VF) and sudden cardiac death.1 These dysrhythmias become more likely as the QTc interval exceeds 500 msec.2
The combination of new-onset global T-wave inversions with prolongation of the QT interval has been reported in only a few limited conditions. Some known causes of these QT T changes include cardiac ischemia, status epilepticus, pheochromocytoma, and acute cocaine intoxication.3 One uncommon and rarely reported cause of extreme QT prolongation and T-wave inversion is acute pulmonary edema. The ECG findings are not present on initial patient presentation; rather the dynamic changes occur after resolution of the pulmonary symptoms. Despite significant ECG changes, all prior reported cases describe ECG normalization without significant morbidity.4,5 We report a case of extreme QT prolongation following acute pulmonary edema that resulted in cardiac arrest secondary to VF.
Case Presentation
A 72-year-old male with medical history of combined systolic and diastolic heart failure, ischemic cardiomyopathy, coronary artery disease, cerebral vascular accident, hypertension, hyperlipidemia, type 2 diabetes mellitus, and tobacco dependence presented to the emergency department (ED) by emergency medical services after awaking with acute onset of dyspnea and diaphoresis. On arrival at the ED, the patient was noted to be in respiratory distress (ie, unable to speak single words) and was extremely diaphoretic. His initial vital signs included blood pressure, 186/113 mm Hg, heart rate, 104 beats per minute, respiratory rate, 40 breaths per minute, and temperature, 36.4 °C. The patient was quickly placed on bilevel positive airway pressure and given sublingual nitroglycerin followed by transdermal nitroglycerin with a single dose of 40 mg IV furosemide, which improved his respiratory status. A chest X-ray was consistent with pulmonary edema, and his brain natriuretic peptide was 1654 pg/mL. An ECG demonstrated new T-wave inversions, and his troponin increased from 0.04 to 0.24 ng/mL during his ED stay (Figure 1). He was started on a heparin infusion and admitted to the hospital for hypertensive emergency with presumed acute decompensated heart failure and non-ST-elevated myocardial infarction.
Throughout the patient’s first night, the troponin level started to down-trend after peaking at 0.24 ng/mL, and his oxygen requirements decreased allowing transition to nasal cannula. However, his repeat ECGs demonstrated significant T-wave abnormalities, new premature ventricular contractions, bradycardia, and a prolonging QTc interval to 703 msec (Figure 2). At this time, the patient’s electrolytes were normal, specifically a potassium level of 4.4 mEq/L, calcium 8.8 mg/dL, magnesium 2.0 mg/dL, and phosphorus 2.6 mg/dL. Given the worsening ECG changes, a computed tomography scan of his head was ordered to rule out intracranial pathology. While in the scanner, the patient went into pulseless VF, prompting defibrillation with 200 J. In addition, he was given 75 mg IV lidocaine, 2 g IV magnesium, and 1 ampule of both calcium chloride and sodium bicarbonate. With treatment, he had return of spontaneous circulation and was taken promptly to cardiac catheterization. The catheterization showed no significant obstructive coronary artery disease, and no interventions were performed. The patient was transferred to the cardiac intensive care unit for continued care.
During his course in the intensive care unit, the patient’s potassium and magnesium levels were maintained at high-normal levels. The patient was started on a dobutamine infusion to increase his heart rate and attempt to decrease his QTc. The patient also underwent cardiac magnetic resonance imaging (MRI) to evaluate for possible myocarditis, which showed no evidence of acute inflammation. Echocardiogram demonstrated an ejection fraction of 40% and global hypokinesis but no specific regional abnormalities and no change from prior echocardiogram performed 1 year earlier. Over the course of 3 days, his ECG normalized and his QTc shortened to 477 msec. Genetic testing was performed and did not reveal any mutations associated with long QT syndrome. Ultimately, an automated internal cardiac defibrillator (AICD) was placed, and the patient was discharged home.
Over the 2 years since his initial event, the patient has not experienced recurrent VF and his AICD has not fired. The patient continues to have ED presentations for heart-failure symptoms, though he has been stable from an electrophysiologic standpoint and his QTc remains less than 500 msec.
Discussion
Prolongation of the QT interval as a result of deep, global T-wave inversions after resolution of acute pulmonary edema has been minimally reported.4,5 This phenomenon has been described in the cardiology literature but has not been discussed in the emergency medicine literature and bears consideration in this case.4,5 As noted, an extensive evaluation did not reveal another cause of QTc prolongation. The patient had normal electrolytes and temperature, his neurologic examination and computed tomography were not remarkable. The patient had no obstructive coronary artery disease on catheterization, no evidence of acute myocarditis on cardiac MRI, no prescribed medications associated with QT prolongation, and no evidence of genetic mutations associated with QT prolongation on testing. The minimal troponin elevation was felt to represent a type II myocardial infarction related to ischemia due to supply-demand mismatch rather than acute plaque rupture.
Littmann published a case series of 9 cases of delayed onset T-wave inversion and extreme QTc prolongation in the 24 to 48 hours following treatment and symptomatic improvement in acute pulmonary edema.4 In each of his patients, an ischemic cardiac insult was ruled out as the etiology of the pulmonary edema by laboratory assessment, echocardiography, and left heart catheterization.All of the patients in this case series recovered without incident and with normalization of the QTc interval.4 Similarly, in our patient, significant QT T changes occurred approximately 22 hours after presentation and with resolution of symptoms of pulmonary edema. Pascale and colleagues also published a series of 3 patients developing similar ECG patterns following a hypertensive crisis with resolution of ECG findings and without any morbidity.5 In contrast, our patient experienced significant morbidity secondary to the extreme QTc prolongation.
Conclusions
We believe this is the first reported case of excessive prolongation of the QTc with VF arrest secondary to resolution of acute pulmonary edema. The pattern observed in our patient follows the patterns outlined in the previous case series—patients present with acute pulmonary edema and hypertensive crisis but develop significant ECG abnormalities about 24 hours after the resolution of the high catecholamine state. Our patient did have a history of prior cardiac insult, given the QTc changes developed acutely, with frequent premature ventricular contractions, and the cardiac arrest occurred at maximal QTc prolongation, yet after resolution of the high catecholamine state, the treatment team felt there was likely an uncaptured and short-lived episode of TdP that degenerated into VF. This theory is further supported by the lack of recurrent VF episodes, confirmed by AICD interrogation, after normalization of the QTc in our patient.
Abnormalities in the T-wave morphology of an electrocardiogram (ECG) are classically attributed to ischemic cardiac disease. However, these changes can be seen in a variety of other etiologies, including noncardiac pathology, which should be considered whenever reviewing an ECG: central nervous system disease, including stroke and subarachnoid hemorrhage; hypothermia; pulmonary disease, such as pulmonary embolism or chronic obstructive pulmonary disease; myopericarditis; drug effects; and electrolyte abnormalities.
Prolongation of the QT interval, on the other hand, can be precipitated by medications, metabolic derangements, or genetic phenotypes. The QT interval is measured from the beginning of the QRS complex to the termination of the T wave and represents the total time for ventricular depolarization and repolarization. The QT interval must be corrected based on the patient’s heart rate, known as the QTc. As the QTc interval lengthens, there is increased risk of R-on-T phenomena, which may result in Torsades de Pointes (TdP). Typical features of TdP include an antecedent prolonged QTc, cyclic polymorphic ventricular tachycardia on the surface ECG, and either a short-lived spontaneously terminating course or degeneration into ventricular fibrillation (VF) and sudden cardiac death.1 These dysrhythmias become more likely as the QTc interval exceeds 500 msec.2
The combination of new-onset global T-wave inversions with prolongation of the QT interval has been reported in only a few limited conditions. Some known causes of these QT T changes include cardiac ischemia, status epilepticus, pheochromocytoma, and acute cocaine intoxication.3 One uncommon and rarely reported cause of extreme QT prolongation and T-wave inversion is acute pulmonary edema. The ECG findings are not present on initial patient presentation; rather the dynamic changes occur after resolution of the pulmonary symptoms. Despite significant ECG changes, all prior reported cases describe ECG normalization without significant morbidity.4,5 We report a case of extreme QT prolongation following acute pulmonary edema that resulted in cardiac arrest secondary to VF.
Case Presentation
A 72-year-old male with medical history of combined systolic and diastolic heart failure, ischemic cardiomyopathy, coronary artery disease, cerebral vascular accident, hypertension, hyperlipidemia, type 2 diabetes mellitus, and tobacco dependence presented to the emergency department (ED) by emergency medical services after awaking with acute onset of dyspnea and diaphoresis. On arrival at the ED, the patient was noted to be in respiratory distress (ie, unable to speak single words) and was extremely diaphoretic. His initial vital signs included blood pressure, 186/113 mm Hg, heart rate, 104 beats per minute, respiratory rate, 40 breaths per minute, and temperature, 36.4 °C. The patient was quickly placed on bilevel positive airway pressure and given sublingual nitroglycerin followed by transdermal nitroglycerin with a single dose of 40 mg IV furosemide, which improved his respiratory status. A chest X-ray was consistent with pulmonary edema, and his brain natriuretic peptide was 1654 pg/mL. An ECG demonstrated new T-wave inversions, and his troponin increased from 0.04 to 0.24 ng/mL during his ED stay (Figure 1). He was started on a heparin infusion and admitted to the hospital for hypertensive emergency with presumed acute decompensated heart failure and non-ST-elevated myocardial infarction.
Throughout the patient’s first night, the troponin level started to down-trend after peaking at 0.24 ng/mL, and his oxygen requirements decreased allowing transition to nasal cannula. However, his repeat ECGs demonstrated significant T-wave abnormalities, new premature ventricular contractions, bradycardia, and a prolonging QTc interval to 703 msec (Figure 2). At this time, the patient’s electrolytes were normal, specifically a potassium level of 4.4 mEq/L, calcium 8.8 mg/dL, magnesium 2.0 mg/dL, and phosphorus 2.6 mg/dL. Given the worsening ECG changes, a computed tomography scan of his head was ordered to rule out intracranial pathology. While in the scanner, the patient went into pulseless VF, prompting defibrillation with 200 J. In addition, he was given 75 mg IV lidocaine, 2 g IV magnesium, and 1 ampule of both calcium chloride and sodium bicarbonate. With treatment, he had return of spontaneous circulation and was taken promptly to cardiac catheterization. The catheterization showed no significant obstructive coronary artery disease, and no interventions were performed. The patient was transferred to the cardiac intensive care unit for continued care.
During his course in the intensive care unit, the patient’s potassium and magnesium levels were maintained at high-normal levels. The patient was started on a dobutamine infusion to increase his heart rate and attempt to decrease his QTc. The patient also underwent cardiac magnetic resonance imaging (MRI) to evaluate for possible myocarditis, which showed no evidence of acute inflammation. Echocardiogram demonstrated an ejection fraction of 40% and global hypokinesis but no specific regional abnormalities and no change from prior echocardiogram performed 1 year earlier. Over the course of 3 days, his ECG normalized and his QTc shortened to 477 msec. Genetic testing was performed and did not reveal any mutations associated with long QT syndrome. Ultimately, an automated internal cardiac defibrillator (AICD) was placed, and the patient was discharged home.
Over the 2 years since his initial event, the patient has not experienced recurrent VF and his AICD has not fired. The patient continues to have ED presentations for heart-failure symptoms, though he has been stable from an electrophysiologic standpoint and his QTc remains less than 500 msec.
Discussion
Prolongation of the QT interval as a result of deep, global T-wave inversions after resolution of acute pulmonary edema has been minimally reported.4,5 This phenomenon has been described in the cardiology literature but has not been discussed in the emergency medicine literature and bears consideration in this case.4,5 As noted, an extensive evaluation did not reveal another cause of QTc prolongation. The patient had normal electrolytes and temperature, his neurologic examination and computed tomography were not remarkable. The patient had no obstructive coronary artery disease on catheterization, no evidence of acute myocarditis on cardiac MRI, no prescribed medications associated with QT prolongation, and no evidence of genetic mutations associated with QT prolongation on testing. The minimal troponin elevation was felt to represent a type II myocardial infarction related to ischemia due to supply-demand mismatch rather than acute plaque rupture.
Littmann published a case series of 9 cases of delayed onset T-wave inversion and extreme QTc prolongation in the 24 to 48 hours following treatment and symptomatic improvement in acute pulmonary edema.4 In each of his patients, an ischemic cardiac insult was ruled out as the etiology of the pulmonary edema by laboratory assessment, echocardiography, and left heart catheterization.All of the patients in this case series recovered without incident and with normalization of the QTc interval.4 Similarly, in our patient, significant QT T changes occurred approximately 22 hours after presentation and with resolution of symptoms of pulmonary edema. Pascale and colleagues also published a series of 3 patients developing similar ECG patterns following a hypertensive crisis with resolution of ECG findings and without any morbidity.5 In contrast, our patient experienced significant morbidity secondary to the extreme QTc prolongation.
Conclusions
We believe this is the first reported case of excessive prolongation of the QTc with VF arrest secondary to resolution of acute pulmonary edema. The pattern observed in our patient follows the patterns outlined in the previous case series—patients present with acute pulmonary edema and hypertensive crisis but develop significant ECG abnormalities about 24 hours after the resolution of the high catecholamine state. Our patient did have a history of prior cardiac insult, given the QTc changes developed acutely, with frequent premature ventricular contractions, and the cardiac arrest occurred at maximal QTc prolongation, yet after resolution of the high catecholamine state, the treatment team felt there was likely an uncaptured and short-lived episode of TdP that degenerated into VF. This theory is further supported by the lack of recurrent VF episodes, confirmed by AICD interrogation, after normalization of the QTc in our patient.
1. Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am. 2001;85(2):321-341. doi:10.1016/s0025-7125(05)70318-7
2. Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE. Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal. 2012;2012:212178. doi:10.1100/2012/212178
3. Miller MA, Elmariah S, Fischer A. Giant T-wave inversions and extreme QT prolongation. Circ Arrhythm Electrophysiol. 2009;2(6):e42-e43. doi:10.1161/CIRCEP.108.825729
4. Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: a report of nine cases. J Am Coll Cardiol. 1999;34(4):1106-1110. doi:10.1016/s0735-1097(99)00311-3
5. Pascale P, Quartenoud B, Stauffer JC. Isolated large inverted T wave in pulmonary edema due to hypertensive crisis: a novel electrocardiographic phenomenon mimicking ischemia?. Clin Res Cardiol. 2007;96(5):288-294. doi:10.1007/s00392-007-0504-1
1. Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am. 2001;85(2):321-341. doi:10.1016/s0025-7125(05)70318-7
2. Kallergis EM, Goudis CA, Simantirakis EN, Kochiadakis GE, Vardas PE. Mechanisms, risk factors, and management of acquired long QT syndrome: a comprehensive review. ScientificWorldJournal. 2012;2012:212178. doi:10.1100/2012/212178
3. Miller MA, Elmariah S, Fischer A. Giant T-wave inversions and extreme QT prolongation. Circ Arrhythm Electrophysiol. 2009;2(6):e42-e43. doi:10.1161/CIRCEP.108.825729
4. Littmann L. Large T wave inversion and QT prolongation associated with pulmonary edema: a report of nine cases. J Am Coll Cardiol. 1999;34(4):1106-1110. doi:10.1016/s0735-1097(99)00311-3
5. Pascale P, Quartenoud B, Stauffer JC. Isolated large inverted T wave in pulmonary edema due to hypertensive crisis: a novel electrocardiographic phenomenon mimicking ischemia?. Clin Res Cardiol. 2007;96(5):288-294. doi:10.1007/s00392-007-0504-1
At 5 years, iFR found as effective and safe as FFR for guiding PCI intervention
The rate of major adverse cardiac events (MACE) over 5 years is similar whether revascularization is guided by instantaneous wave-free ratio (iFR) or fractional flow reserve (FFR), according to long-term results of the iFR-SWEDEHEART study.
“The results are about the same as reported at 12 months. There were no significant differences in any outcome we evaluated,” according to Matthias Götberg, MD, PhD.
When the initial results of the noninferiority iFR-SWEDEHEART trial were published after 1 year of follow-up, the primary MACE endpoint of death from any-cause nonfatal myocardial infarction, or unplanned revascularization, was met by 6.7% and 6.1% of those randomized to iFR or FFR, respectively.
These outcomes were not significantly different and placed iFR well within the predefined boundaries of noninferiority (P = .007).
In this new and final follow-up of iFR-SWEDEHEART, which evaluated the same 2,019 patients who were alive at 1 year (none were lost to follow-up), the MACE endpoint was met by 21.5% and 19.9% of those managed with iFR and FFR, respectively. The hazard ratio (1.09) had a wide 95% confidence interval (0.90-1.31) that did not approach statistical significance.
No differences seen across outcomes
When broken down into the MACE components, there were no differences between iFR and FFR, respectively, for all-cause death (9.4% vs. 7.9%), MI (5.8% vs. 5.7%) or unplanned revascularization (11.6% vs. 11.3%).
Across predefined subgroups, such as those defined by age, gender, stable versus unstable angina, and presence of risk factors such as diabetes, hypertension, hyperlipidemia, and smoking, there were also no significant differences in outcome.
At the time iFR-SWEDEHART was initiated, FFR had already been accepted as more effective than angiographic assessment to identify lesion ischemia and the need for percutaneous intervention (PCI). The iFR-SWEDEHEART trial tested iFR, a relatively new technology at the time, as a noninferior alternative. Unlike FFR, which requires adenosine to dilate the vessel, adding cost and patient discomfort, iFR measures the resting pressure gradient across the coronary lesion, and it is generally easier to perform.
“The advantage of iFR is that it provides an instantaneous lesion assessment without the need for adenosine,” Dr. Götberg explained in presenting the results at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando.
When the procedural results were compared in the published study at 1 year, it was noted that the mean number of lesions evaluated per patient was higher (1.55 vs. 1.43; P = .002), but the proportion of lesions found functionally significant was lower (29.2% vs. 36.8%; P < .0001) among those randomized to iFR than in the FFR group.
While most other procedural characteristics, such as PCI access route, fluoroscopy time, and contrast use did not differ significantly, fewer stents were placed in patients managed with iFR (1.58 vs. 1.73; P = .048), and a reduction in the average procedural time of a few minutes approached significance (P = .09).
Patient discomfort is greater with FFR
Patient discomfort measured during the procedure did differ, according to Dr. Götberg, an interventional cardiologist at Skåne University Hospital, Lund, Sweden.
Only about 30% in the FFR group reported no discomfort. Most of the others reported mild or moderate discomfort, but nearly 10% characterized the discomfort as severe. In the iFR group, more than 95% reported no discomfort. All of the remaining patients reported discomfort level as mild.
Because differences in MACE would be most likely to occur in the first year after revascularization, the similarity of the 1- and 5-year results were expected, according to Dr. Götberg. However, a 5-year follow-up was considered prudent given the relatively limited experience with iFR when the study was designed. This technique is now well established and widely used.
The study supports the premise that quicker and easier-to-obtain results with iFR are obtained without sacrificing greater relative risk of failing to identify a vulnerable lesion, according to Dr. Götberg.
Nevertheless, iFR and FFR “are not an exact match,” according to Jennifer A. Rymer, MD, an interventional cardiologist and assistant professor of medicine at Duke University, Durham, N.C. Although she called this trial an “excellent” demonstration of comparable utility in distinguishing lesions that do not require intervention from those that do, she implied that some clinicians might still prefer FFR for other reasons.
For example, FFR provides information about coronary flow reserve and microvascular resistance that are relevant to the underlying pathophysiology in a diseased vessel, according to Shmuel Banai, MD, head of interventional cardiology, Tel Aviv Medical Center. Recognizing that this information is not as readily generated by iFR, he is among those who plan to continue to use FFR despite these results.
However, for those who are now routinely performing iFR for the purposes of guiding revascularization, “these data are reassuring,” said David Kandzari, MD, director of interventional cardiology, Piedmont Hart Institute, Atlanta. The 5-year data essentially eliminate the likelihood that iFR relative to FFR increases the risk of missing functionally significant lesions for revascularization procedures.
Dr. Götberg reports financial relationships with Abbott, Boston Scientific, Medtronic, and Phillips Healthcare. Dr. Rymer reports no potential financial conflicts of interest. Dr. Banai has a financial relationship with Neovasc. Dr. Kandzari reports financial relationships with Ablative Solutions and Medtronic.
The rate of major adverse cardiac events (MACE) over 5 years is similar whether revascularization is guided by instantaneous wave-free ratio (iFR) or fractional flow reserve (FFR), according to long-term results of the iFR-SWEDEHEART study.
“The results are about the same as reported at 12 months. There were no significant differences in any outcome we evaluated,” according to Matthias Götberg, MD, PhD.
When the initial results of the noninferiority iFR-SWEDEHEART trial were published after 1 year of follow-up, the primary MACE endpoint of death from any-cause nonfatal myocardial infarction, or unplanned revascularization, was met by 6.7% and 6.1% of those randomized to iFR or FFR, respectively.
These outcomes were not significantly different and placed iFR well within the predefined boundaries of noninferiority (P = .007).
In this new and final follow-up of iFR-SWEDEHEART, which evaluated the same 2,019 patients who were alive at 1 year (none were lost to follow-up), the MACE endpoint was met by 21.5% and 19.9% of those managed with iFR and FFR, respectively. The hazard ratio (1.09) had a wide 95% confidence interval (0.90-1.31) that did not approach statistical significance.
No differences seen across outcomes
When broken down into the MACE components, there were no differences between iFR and FFR, respectively, for all-cause death (9.4% vs. 7.9%), MI (5.8% vs. 5.7%) or unplanned revascularization (11.6% vs. 11.3%).
Across predefined subgroups, such as those defined by age, gender, stable versus unstable angina, and presence of risk factors such as diabetes, hypertension, hyperlipidemia, and smoking, there were also no significant differences in outcome.
At the time iFR-SWEDEHART was initiated, FFR had already been accepted as more effective than angiographic assessment to identify lesion ischemia and the need for percutaneous intervention (PCI). The iFR-SWEDEHEART trial tested iFR, a relatively new technology at the time, as a noninferior alternative. Unlike FFR, which requires adenosine to dilate the vessel, adding cost and patient discomfort, iFR measures the resting pressure gradient across the coronary lesion, and it is generally easier to perform.
“The advantage of iFR is that it provides an instantaneous lesion assessment without the need for adenosine,” Dr. Götberg explained in presenting the results at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando.
When the procedural results were compared in the published study at 1 year, it was noted that the mean number of lesions evaluated per patient was higher (1.55 vs. 1.43; P = .002), but the proportion of lesions found functionally significant was lower (29.2% vs. 36.8%; P < .0001) among those randomized to iFR than in the FFR group.
While most other procedural characteristics, such as PCI access route, fluoroscopy time, and contrast use did not differ significantly, fewer stents were placed in patients managed with iFR (1.58 vs. 1.73; P = .048), and a reduction in the average procedural time of a few minutes approached significance (P = .09).
Patient discomfort is greater with FFR
Patient discomfort measured during the procedure did differ, according to Dr. Götberg, an interventional cardiologist at Skåne University Hospital, Lund, Sweden.
Only about 30% in the FFR group reported no discomfort. Most of the others reported mild or moderate discomfort, but nearly 10% characterized the discomfort as severe. In the iFR group, more than 95% reported no discomfort. All of the remaining patients reported discomfort level as mild.
Because differences in MACE would be most likely to occur in the first year after revascularization, the similarity of the 1- and 5-year results were expected, according to Dr. Götberg. However, a 5-year follow-up was considered prudent given the relatively limited experience with iFR when the study was designed. This technique is now well established and widely used.
The study supports the premise that quicker and easier-to-obtain results with iFR are obtained without sacrificing greater relative risk of failing to identify a vulnerable lesion, according to Dr. Götberg.
Nevertheless, iFR and FFR “are not an exact match,” according to Jennifer A. Rymer, MD, an interventional cardiologist and assistant professor of medicine at Duke University, Durham, N.C. Although she called this trial an “excellent” demonstration of comparable utility in distinguishing lesions that do not require intervention from those that do, she implied that some clinicians might still prefer FFR for other reasons.
For example, FFR provides information about coronary flow reserve and microvascular resistance that are relevant to the underlying pathophysiology in a diseased vessel, according to Shmuel Banai, MD, head of interventional cardiology, Tel Aviv Medical Center. Recognizing that this information is not as readily generated by iFR, he is among those who plan to continue to use FFR despite these results.
However, for those who are now routinely performing iFR for the purposes of guiding revascularization, “these data are reassuring,” said David Kandzari, MD, director of interventional cardiology, Piedmont Hart Institute, Atlanta. The 5-year data essentially eliminate the likelihood that iFR relative to FFR increases the risk of missing functionally significant lesions for revascularization procedures.
Dr. Götberg reports financial relationships with Abbott, Boston Scientific, Medtronic, and Phillips Healthcare. Dr. Rymer reports no potential financial conflicts of interest. Dr. Banai has a financial relationship with Neovasc. Dr. Kandzari reports financial relationships with Ablative Solutions and Medtronic.
The rate of major adverse cardiac events (MACE) over 5 years is similar whether revascularization is guided by instantaneous wave-free ratio (iFR) or fractional flow reserve (FFR), according to long-term results of the iFR-SWEDEHEART study.
“The results are about the same as reported at 12 months. There were no significant differences in any outcome we evaluated,” according to Matthias Götberg, MD, PhD.
When the initial results of the noninferiority iFR-SWEDEHEART trial were published after 1 year of follow-up, the primary MACE endpoint of death from any-cause nonfatal myocardial infarction, or unplanned revascularization, was met by 6.7% and 6.1% of those randomized to iFR or FFR, respectively.
These outcomes were not significantly different and placed iFR well within the predefined boundaries of noninferiority (P = .007).
In this new and final follow-up of iFR-SWEDEHEART, which evaluated the same 2,019 patients who were alive at 1 year (none were lost to follow-up), the MACE endpoint was met by 21.5% and 19.9% of those managed with iFR and FFR, respectively. The hazard ratio (1.09) had a wide 95% confidence interval (0.90-1.31) that did not approach statistical significance.
No differences seen across outcomes
When broken down into the MACE components, there were no differences between iFR and FFR, respectively, for all-cause death (9.4% vs. 7.9%), MI (5.8% vs. 5.7%) or unplanned revascularization (11.6% vs. 11.3%).
Across predefined subgroups, such as those defined by age, gender, stable versus unstable angina, and presence of risk factors such as diabetes, hypertension, hyperlipidemia, and smoking, there were also no significant differences in outcome.
At the time iFR-SWEDEHART was initiated, FFR had already been accepted as more effective than angiographic assessment to identify lesion ischemia and the need for percutaneous intervention (PCI). The iFR-SWEDEHEART trial tested iFR, a relatively new technology at the time, as a noninferior alternative. Unlike FFR, which requires adenosine to dilate the vessel, adding cost and patient discomfort, iFR measures the resting pressure gradient across the coronary lesion, and it is generally easier to perform.
“The advantage of iFR is that it provides an instantaneous lesion assessment without the need for adenosine,” Dr. Götberg explained in presenting the results at the Transcatheter Cardiovascular Therapeutics annual meeting, held virtually and live in Orlando.
When the procedural results were compared in the published study at 1 year, it was noted that the mean number of lesions evaluated per patient was higher (1.55 vs. 1.43; P = .002), but the proportion of lesions found functionally significant was lower (29.2% vs. 36.8%; P < .0001) among those randomized to iFR than in the FFR group.
While most other procedural characteristics, such as PCI access route, fluoroscopy time, and contrast use did not differ significantly, fewer stents were placed in patients managed with iFR (1.58 vs. 1.73; P = .048), and a reduction in the average procedural time of a few minutes approached significance (P = .09).
Patient discomfort is greater with FFR
Patient discomfort measured during the procedure did differ, according to Dr. Götberg, an interventional cardiologist at Skåne University Hospital, Lund, Sweden.
Only about 30% in the FFR group reported no discomfort. Most of the others reported mild or moderate discomfort, but nearly 10% characterized the discomfort as severe. In the iFR group, more than 95% reported no discomfort. All of the remaining patients reported discomfort level as mild.
Because differences in MACE would be most likely to occur in the first year after revascularization, the similarity of the 1- and 5-year results were expected, according to Dr. Götberg. However, a 5-year follow-up was considered prudent given the relatively limited experience with iFR when the study was designed. This technique is now well established and widely used.
The study supports the premise that quicker and easier-to-obtain results with iFR are obtained without sacrificing greater relative risk of failing to identify a vulnerable lesion, according to Dr. Götberg.
Nevertheless, iFR and FFR “are not an exact match,” according to Jennifer A. Rymer, MD, an interventional cardiologist and assistant professor of medicine at Duke University, Durham, N.C. Although she called this trial an “excellent” demonstration of comparable utility in distinguishing lesions that do not require intervention from those that do, she implied that some clinicians might still prefer FFR for other reasons.
For example, FFR provides information about coronary flow reserve and microvascular resistance that are relevant to the underlying pathophysiology in a diseased vessel, according to Shmuel Banai, MD, head of interventional cardiology, Tel Aviv Medical Center. Recognizing that this information is not as readily generated by iFR, he is among those who plan to continue to use FFR despite these results.
However, for those who are now routinely performing iFR for the purposes of guiding revascularization, “these data are reassuring,” said David Kandzari, MD, director of interventional cardiology, Piedmont Hart Institute, Atlanta. The 5-year data essentially eliminate the likelihood that iFR relative to FFR increases the risk of missing functionally significant lesions for revascularization procedures.
Dr. Götberg reports financial relationships with Abbott, Boston Scientific, Medtronic, and Phillips Healthcare. Dr. Rymer reports no potential financial conflicts of interest. Dr. Banai has a financial relationship with Neovasc. Dr. Kandzari reports financial relationships with Ablative Solutions and Medtronic.
FROM TCT 2021
Statins’ effects on CVD outweigh risk for diabetes in RA
The use of statins by patients with rheumatoid arthritis appears to provide an overall net benefit on cardiovascular disease outcomes that outweighs the risk of type 2 diabetes mellitus (T2DM) seen with the drugs in the general population, according to evidence from a cohort study of more than 16,000 people in the United Kingdom that was presented at the virtual annual meeting of the American College of Rheumatology.
“Our study emphasizes that RA patients should be assessed for statin initiation to improve CVD risk,” lead study author Gulsen Ozen, MD, a third-year resident at the University of Nebraska, Omaha, said in an interview. Because the risk of T2DM with statin use is no worse in patients with RA than in the general population, statin initiation “is actually a great opportunity to address the risk factors for T2DM such as activity and exercise, obesity and weight loss, and [use of glucocorticoids], which have other important health effects,” she said.
“Also, importantly, even if [patients] develop T2DM, statins still work on CVD and mortality outcomes as in patients without diabetes,” Dr. Ozen added. “Given all, the benefits of statins way outweigh the hazards.”
Dr. Ozen said this was the first large cohort study to evaluate CVD mortality and T2DM risks with statins in patients with RA, a claim with which rheumatologist Elena Myasoedova, MD, PhD, of the Mayo Clinic in Rochester, Minn., concurred.
Dr. Myasoedova, professor of rheumatology and epidemiology at Mayo, said in an interview that the study was “methodologically rigorous” using time-conditional propensity score (TCPS) matching and a prevalent new-user design, “thus addressing the immortal time bias” found in the design of studies in which patients enter a cohort but do not start a treatment before developing the outcome of interest and are assigned to the untreated group or when the period of delay from when patients enter the cohort to when they are treated is excluded from the analysis. An earlier study from the same authors did not use TCPS matching, she said.
“The study findings suggest that patients with RA can benefit from statin use in terms of CVD outcomes and mortality but physicians should use vigilance regarding increased T2DM risk and discuss this possibility with patients,” Dr. Myasoedova said. “Identifying patients who are at higher risk of developing T2DM after statin initiation would be important to personalize the approach to statin therapy.”
Study details
The study accessed records from the U.K. Clinical Practice Research Datalink and linked Hospital Episode Statistics and Office of National Statistics databases. It analyzed adult patients with RA who were diagnosed during 1989-2018 in two cohorts: One for CVD and all-cause mortality, consisting of 1,768 statin initiators and 3,528 TCPS-matched nonusers; and a T2DM cohort with 3,608 statin initiators and 7,208 TCPS-matched nonusers.
In the entire cohort, statin use was associated with a 32% reduction in CV events (composite endpoint of the nonfatal or fatal MI, stroke, hospitalized heart failure, or CVD mortality), a 54% reduction in all-cause mortality, and a 33% increase in risk for T2DM, Dr. Ozen said. Results were similar in both sexes, although CV event reduction with statins in men did not reach statistical significance, likely because of a smaller sample size, she said.
Patients with and without a history of CVD had a similar reduction in CV events and all-cause mortality, and risk for T2DM increased with statins, but the latter reached statistical significance only in patients without a history of CVD, Dr. Ozen said.
Patients with RA who are at risk for T2DM and who are taking statins require blood glucose monitoring, which is typically done in patients with RA on disease-modifying antirheumatic drugs, and hemoglobin A1c testing when glucose levels are impaired, she said. “Any concerns for T2DM would be also communicated by the primary care providers of the patients to initiate further assessment and management,” she said.
But Dr. Ozen noted that confusion exists among primary care physicians and rheumatologists about who’s responsible for prescribing statins in these patients. “I would like to remind you that instead of assigning this role to a certain specialty, just good communication could improve this care gap of statin underutilization in RA,” she said. “Also, for rheumatologists, given that all-cause mortality reduction with statins was as high as CV event reduction, statins may be reducing other causes of mortality through improving disease activity.”
Bristol-Myers Squibb provided funding for the study. Dr. Ozen and Dr. Myasoedova have no relevant disclosures.
The use of statins by patients with rheumatoid arthritis appears to provide an overall net benefit on cardiovascular disease outcomes that outweighs the risk of type 2 diabetes mellitus (T2DM) seen with the drugs in the general population, according to evidence from a cohort study of more than 16,000 people in the United Kingdom that was presented at the virtual annual meeting of the American College of Rheumatology.
“Our study emphasizes that RA patients should be assessed for statin initiation to improve CVD risk,” lead study author Gulsen Ozen, MD, a third-year resident at the University of Nebraska, Omaha, said in an interview. Because the risk of T2DM with statin use is no worse in patients with RA than in the general population, statin initiation “is actually a great opportunity to address the risk factors for T2DM such as activity and exercise, obesity and weight loss, and [use of glucocorticoids], which have other important health effects,” she said.
“Also, importantly, even if [patients] develop T2DM, statins still work on CVD and mortality outcomes as in patients without diabetes,” Dr. Ozen added. “Given all, the benefits of statins way outweigh the hazards.”
Dr. Ozen said this was the first large cohort study to evaluate CVD mortality and T2DM risks with statins in patients with RA, a claim with which rheumatologist Elena Myasoedova, MD, PhD, of the Mayo Clinic in Rochester, Minn., concurred.
Dr. Myasoedova, professor of rheumatology and epidemiology at Mayo, said in an interview that the study was “methodologically rigorous” using time-conditional propensity score (TCPS) matching and a prevalent new-user design, “thus addressing the immortal time bias” found in the design of studies in which patients enter a cohort but do not start a treatment before developing the outcome of interest and are assigned to the untreated group or when the period of delay from when patients enter the cohort to when they are treated is excluded from the analysis. An earlier study from the same authors did not use TCPS matching, she said.
“The study findings suggest that patients with RA can benefit from statin use in terms of CVD outcomes and mortality but physicians should use vigilance regarding increased T2DM risk and discuss this possibility with patients,” Dr. Myasoedova said. “Identifying patients who are at higher risk of developing T2DM after statin initiation would be important to personalize the approach to statin therapy.”
Study details
The study accessed records from the U.K. Clinical Practice Research Datalink and linked Hospital Episode Statistics and Office of National Statistics databases. It analyzed adult patients with RA who were diagnosed during 1989-2018 in two cohorts: One for CVD and all-cause mortality, consisting of 1,768 statin initiators and 3,528 TCPS-matched nonusers; and a T2DM cohort with 3,608 statin initiators and 7,208 TCPS-matched nonusers.
In the entire cohort, statin use was associated with a 32% reduction in CV events (composite endpoint of the nonfatal or fatal MI, stroke, hospitalized heart failure, or CVD mortality), a 54% reduction in all-cause mortality, and a 33% increase in risk for T2DM, Dr. Ozen said. Results were similar in both sexes, although CV event reduction with statins in men did not reach statistical significance, likely because of a smaller sample size, she said.
Patients with and without a history of CVD had a similar reduction in CV events and all-cause mortality, and risk for T2DM increased with statins, but the latter reached statistical significance only in patients without a history of CVD, Dr. Ozen said.
Patients with RA who are at risk for T2DM and who are taking statins require blood glucose monitoring, which is typically done in patients with RA on disease-modifying antirheumatic drugs, and hemoglobin A1c testing when glucose levels are impaired, she said. “Any concerns for T2DM would be also communicated by the primary care providers of the patients to initiate further assessment and management,” she said.
But Dr. Ozen noted that confusion exists among primary care physicians and rheumatologists about who’s responsible for prescribing statins in these patients. “I would like to remind you that instead of assigning this role to a certain specialty, just good communication could improve this care gap of statin underutilization in RA,” she said. “Also, for rheumatologists, given that all-cause mortality reduction with statins was as high as CV event reduction, statins may be reducing other causes of mortality through improving disease activity.”
Bristol-Myers Squibb provided funding for the study. Dr. Ozen and Dr. Myasoedova have no relevant disclosures.
The use of statins by patients with rheumatoid arthritis appears to provide an overall net benefit on cardiovascular disease outcomes that outweighs the risk of type 2 diabetes mellitus (T2DM) seen with the drugs in the general population, according to evidence from a cohort study of more than 16,000 people in the United Kingdom that was presented at the virtual annual meeting of the American College of Rheumatology.
“Our study emphasizes that RA patients should be assessed for statin initiation to improve CVD risk,” lead study author Gulsen Ozen, MD, a third-year resident at the University of Nebraska, Omaha, said in an interview. Because the risk of T2DM with statin use is no worse in patients with RA than in the general population, statin initiation “is actually a great opportunity to address the risk factors for T2DM such as activity and exercise, obesity and weight loss, and [use of glucocorticoids], which have other important health effects,” she said.
“Also, importantly, even if [patients] develop T2DM, statins still work on CVD and mortality outcomes as in patients without diabetes,” Dr. Ozen added. “Given all, the benefits of statins way outweigh the hazards.”
Dr. Ozen said this was the first large cohort study to evaluate CVD mortality and T2DM risks with statins in patients with RA, a claim with which rheumatologist Elena Myasoedova, MD, PhD, of the Mayo Clinic in Rochester, Minn., concurred.
Dr. Myasoedova, professor of rheumatology and epidemiology at Mayo, said in an interview that the study was “methodologically rigorous” using time-conditional propensity score (TCPS) matching and a prevalent new-user design, “thus addressing the immortal time bias” found in the design of studies in which patients enter a cohort but do not start a treatment before developing the outcome of interest and are assigned to the untreated group or when the period of delay from when patients enter the cohort to when they are treated is excluded from the analysis. An earlier study from the same authors did not use TCPS matching, she said.
“The study findings suggest that patients with RA can benefit from statin use in terms of CVD outcomes and mortality but physicians should use vigilance regarding increased T2DM risk and discuss this possibility with patients,” Dr. Myasoedova said. “Identifying patients who are at higher risk of developing T2DM after statin initiation would be important to personalize the approach to statin therapy.”
Study details
The study accessed records from the U.K. Clinical Practice Research Datalink and linked Hospital Episode Statistics and Office of National Statistics databases. It analyzed adult patients with RA who were diagnosed during 1989-2018 in two cohorts: One for CVD and all-cause mortality, consisting of 1,768 statin initiators and 3,528 TCPS-matched nonusers; and a T2DM cohort with 3,608 statin initiators and 7,208 TCPS-matched nonusers.
In the entire cohort, statin use was associated with a 32% reduction in CV events (composite endpoint of the nonfatal or fatal MI, stroke, hospitalized heart failure, or CVD mortality), a 54% reduction in all-cause mortality, and a 33% increase in risk for T2DM, Dr. Ozen said. Results were similar in both sexes, although CV event reduction with statins in men did not reach statistical significance, likely because of a smaller sample size, she said.
Patients with and without a history of CVD had a similar reduction in CV events and all-cause mortality, and risk for T2DM increased with statins, but the latter reached statistical significance only in patients without a history of CVD, Dr. Ozen said.
Patients with RA who are at risk for T2DM and who are taking statins require blood glucose monitoring, which is typically done in patients with RA on disease-modifying antirheumatic drugs, and hemoglobin A1c testing when glucose levels are impaired, she said. “Any concerns for T2DM would be also communicated by the primary care providers of the patients to initiate further assessment and management,” she said.
But Dr. Ozen noted that confusion exists among primary care physicians and rheumatologists about who’s responsible for prescribing statins in these patients. “I would like to remind you that instead of assigning this role to a certain specialty, just good communication could improve this care gap of statin underutilization in RA,” she said. “Also, for rheumatologists, given that all-cause mortality reduction with statins was as high as CV event reduction, statins may be reducing other causes of mortality through improving disease activity.”
Bristol-Myers Squibb provided funding for the study. Dr. Ozen and Dr. Myasoedova have no relevant disclosures.
FROM ACR 2021
Validated scoring system identifies low-risk syncope patients
ILLUSTRATIVE CASE
A 30-year-old woman presented to the ED after she “passed out” while standing at a concert. She lost consciousness for 10 seconds. After she revived, her friends drove her to the ED. She is healthy, with no chronic medical conditions, no medication use, and no drug or alcohol use. Should she be admitted to the hospital for observation?
Syncope, a transient loss of consciousness followed by spontaneous complete recovery, accounts for 1% of ED visits.2 Approximately 10% of patients presenting to the ED will have a serious underlying condition identified and among 3% to 5% of these patients with syncope, the serious condition will be identified only after they leave the ED.1 Most patients have a benign course, but more than half of all patients presenting to the ED with syncope will be hospitalized, costing $2.4 billion annually.2
Because of the high hospitalization rate of patients with syncope, a practical and accurate tool to risk-stratify patients is vital. Other tools, such as the San Francisco Syncope Rule, Short-Term Prognosis of Syncope, and Risk Stratification of Syncope in the Emergency Department, lack validation or are excessively complex, with extensive lab work or testing.3
The CSRS was previously derived from a large, multisite consecutive cohort, and was internally validated and reported according to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis guideline statement.4 Patients are assigned points based on clinical findings, test results, and the diagnosis given in the ED (TABLE4). The scoring system is used to stratify patients as very low (−3, −2), low (−1, 0), medium (1, 2, 3), high (4, 5), or very high (≥6) risk.4
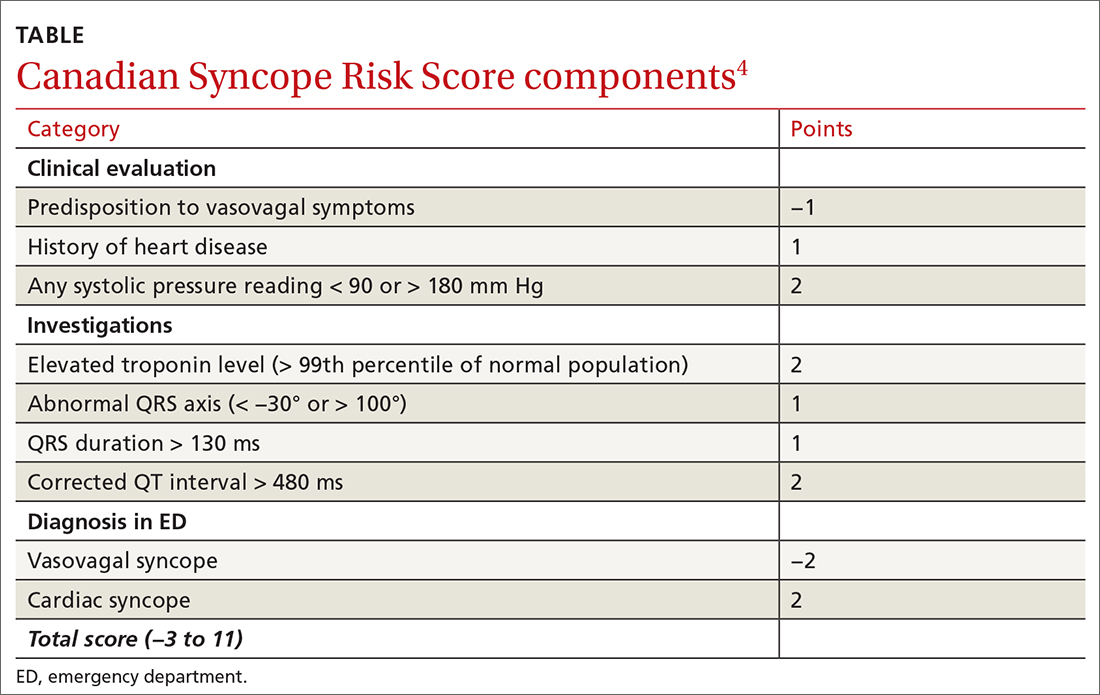
STUDY SUMMARY
Less than 1% of very low– and low-risk patients had serious 30-day outcomes
This multisite Canadian prospective validation cohort study enrolled patients age ≥ 16 years who presented to the ED within 24 hours of syncope. Both discharged and hospitalized patients were included.1
Patients were excluded if they had loss of consciousness for > 5 minutes, mental status changes at presentation, history of current or previous seizure, or head trauma
ED physicians confirmed patient eligibility, obtained verbal consent, and completed the data collection form. In addition, research assistants sought to identify eligible patients who were not previously enrolled by reviewing all ED visits during the study period.
Continue to: To examine 30-day outcomes...
To examine 30-day outcomes, researchers reviewed all available patient medical record
A total of 4131 patients made up the validation cohort. A serious condition was identified during the initial ED visit in 160 patients (3.9%), who were excluded from the study, and 152 patients (3.7%) were lost to follow-up. Of the 3819 patients included in the final analysis, troponin was not measured in 1566 patients (41%), and an electrocardiogram was not obtained in 114 patients (3%). A serious outcome within 30 days was experienced by 139 patients (3.6%; 95% CI, 3.1%-4.3%). There was good correlation to the model-predicted serious outcome probability of 3.2% (95% CI, 2.7%-3.8%).1
Three of 1631 (0.2%) patients classified as very low risk and 9 of 1254 (0.7%) low-risk patients experienced a serious outcome, and no patients died. In the group classified as medium risk, 55 of 687 (8%) patients experienced a serious outcome, and there was 1 death. In the high-risk group, 32 of 167 (19.2%) patients experienced a serious outcome, and there were 5 deaths. In the group classified as very high risk, 40 of 78 (51.3%) patients experienced a serious outcome, and there were 7 deaths. The CSRS was able to identify very low– or low-risk patients (score of −1 or better) with a sensitivity of 97.8% (95% CI, 93.8%-99.6%) and a specificity of 44.3% (95% CI, 42.7%-45.9%).1
WHAT’S NEW
This scoring system offers a validated method to risk-stratify ED patients
Previous recommendations from the American College of Cardiology/American Heart Associationsuggested determining disposition of ED patients by using clinical judgment based on a list of risk factors such as age, chronic conditions, and medications. However, there was no scoring system.3 This new scoring system allows physicians to send home very low– and low-risk patients with reassurance that the likelihood of a serious outcome is less than 1%. High-risk and very high–risk patients should be admitted to the hospital for further evaluation. Most moderate-risk patients (8% risk of serious outcome but 0.1% risk of death) can also be discharged after providers have a risk/benefit discussion, including precautions for signs of arrhythmia or need for urgent return to the hospital.
CAVEATS
The study does not translate to all clinical settings
Because this study was done in EDs, the scoring system cannot necessarily be applied to urgent care or outpatient settings. However, 41% of the patients in the study did not have troponin testing performed. Therefore, physicians could consider using the scoring system in settings where this lab test is not immediately available.
Continue to: This scoring system was also only...
This scoring system was also only validated with adult patients presenting within 24 hours of their syncopal episode. It is unknown how it may predict the outcomes of patients who present > 24 hours after syncope.
CHALLENGES TO IMPLEMENTATION
Clinicians may not be awareof the CSRS scoring system
The main challenge to implementation is practitioner awareness of the CSRS scoring system and how to use it appropriately, as there are several different syncopal scoring systems that may already be in use. Additionally, depending on the electronic health record used, the CSRS scoring system may not be embedded. Using and documenting scores may also be a challenge.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Thiruganasambandamoorthy V, Sivilotti MLA, Le Sage N, et al. Multicenter emergency department validation of the Canadian Syncope Risk Score. JAMA Intern Med. 2020;180:737-744. doi:10.1001/jamainternmed.2020.0288
2. Probst MA, Kanzaria HK, Gbedemah M, et al. National trends in resource utilization associated with ED visits for syncope. Am J Emerg Med. 2015;33:998-1001. doi:10.1016/j.ajem.2015.04.030
3. Shen WK, Sheldon RS, Benditt DG, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70:620-663. doi:10.1016/j.jacc.2017.03.002
4. Thiruganasambandamoorthy V, Kwong K, Wells GA, et al. Development of the Canadian Syncope Risk Score to predict serious adverse events after emergency department assessment of syncope. CMAJ. 2016;188:E289-E298. doi:10.1503/cmaj.151469
ILLUSTRATIVE CASE
A 30-year-old woman presented to the ED after she “passed out” while standing at a concert. She lost consciousness for 10 seconds. After she revived, her friends drove her to the ED. She is healthy, with no chronic medical conditions, no medication use, and no drug or alcohol use. Should she be admitted to the hospital for observation?
Syncope, a transient loss of consciousness followed by spontaneous complete recovery, accounts for 1% of ED visits.2 Approximately 10% of patients presenting to the ED will have a serious underlying condition identified and among 3% to 5% of these patients with syncope, the serious condition will be identified only after they leave the ED.1 Most patients have a benign course, but more than half of all patients presenting to the ED with syncope will be hospitalized, costing $2.4 billion annually.2
Because of the high hospitalization rate of patients with syncope, a practical and accurate tool to risk-stratify patients is vital. Other tools, such as the San Francisco Syncope Rule, Short-Term Prognosis of Syncope, and Risk Stratification of Syncope in the Emergency Department, lack validation or are excessively complex, with extensive lab work or testing.3
The CSRS was previously derived from a large, multisite consecutive cohort, and was internally validated and reported according to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis guideline statement.4 Patients are assigned points based on clinical findings, test results, and the diagnosis given in the ED (TABLE4). The scoring system is used to stratify patients as very low (−3, −2), low (−1, 0), medium (1, 2, 3), high (4, 5), or very high (≥6) risk.4

STUDY SUMMARY
Less than 1% of very low– and low-risk patients had serious 30-day outcomes
This multisite Canadian prospective validation cohort study enrolled patients age ≥ 16 years who presented to the ED within 24 hours of syncope. Both discharged and hospitalized patients were included.1
Patients were excluded if they had loss of consciousness for > 5 minutes, mental status changes at presentation, history of current or previous seizure, or head trauma
ED physicians confirmed patient eligibility, obtained verbal consent, and completed the data collection form. In addition, research assistants sought to identify eligible patients who were not previously enrolled by reviewing all ED visits during the study period.
Continue to: To examine 30-day outcomes...
To examine 30-day outcomes, researchers reviewed all available patient medical record
A total of 4131 patients made up the validation cohort. A serious condition was identified during the initial ED visit in 160 patients (3.9%), who were excluded from the study, and 152 patients (3.7%) were lost to follow-up. Of the 3819 patients included in the final analysis, troponin was not measured in 1566 patients (41%), and an electrocardiogram was not obtained in 114 patients (3%). A serious outcome within 30 days was experienced by 139 patients (3.6%; 95% CI, 3.1%-4.3%). There was good correlation to the model-predicted serious outcome probability of 3.2% (95% CI, 2.7%-3.8%).1
Three of 1631 (0.2%) patients classified as very low risk and 9 of 1254 (0.7%) low-risk patients experienced a serious outcome, and no patients died. In the group classified as medium risk, 55 of 687 (8%) patients experienced a serious outcome, and there was 1 death. In the high-risk group, 32 of 167 (19.2%) patients experienced a serious outcome, and there were 5 deaths. In the group classified as very high risk, 40 of 78 (51.3%) patients experienced a serious outcome, and there were 7 deaths. The CSRS was able to identify very low– or low-risk patients (score of −1 or better) with a sensitivity of 97.8% (95% CI, 93.8%-99.6%) and a specificity of 44.3% (95% CI, 42.7%-45.9%).1
WHAT’S NEW
This scoring system offers a validated method to risk-stratify ED patients
Previous recommendations from the American College of Cardiology/American Heart Associationsuggested determining disposition of ED patients by using clinical judgment based on a list of risk factors such as age, chronic conditions, and medications. However, there was no scoring system.3 This new scoring system allows physicians to send home very low– and low-risk patients with reassurance that the likelihood of a serious outcome is less than 1%. High-risk and very high–risk patients should be admitted to the hospital for further evaluation. Most moderate-risk patients (8% risk of serious outcome but 0.1% risk of death) can also be discharged after providers have a risk/benefit discussion, including precautions for signs of arrhythmia or need for urgent return to the hospital.
CAVEATS
The study does not translate to all clinical settings
Because this study was done in EDs, the scoring system cannot necessarily be applied to urgent care or outpatient settings. However, 41% of the patients in the study did not have troponin testing performed. Therefore, physicians could consider using the scoring system in settings where this lab test is not immediately available.
Continue to: This scoring system was also only...
This scoring system was also only validated with adult patients presenting within 24 hours of their syncopal episode. It is unknown how it may predict the outcomes of patients who present > 24 hours after syncope.
CHALLENGES TO IMPLEMENTATION
Clinicians may not be awareof the CSRS scoring system
The main challenge to implementation is practitioner awareness of the CSRS scoring system and how to use it appropriately, as there are several different syncopal scoring systems that may already be in use. Additionally, depending on the electronic health record used, the CSRS scoring system may not be embedded. Using and documenting scores may also be a challenge.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 30-year-old woman presented to the ED after she “passed out” while standing at a concert. She lost consciousness for 10 seconds. After she revived, her friends drove her to the ED. She is healthy, with no chronic medical conditions, no medication use, and no drug or alcohol use. Should she be admitted to the hospital for observation?
Syncope, a transient loss of consciousness followed by spontaneous complete recovery, accounts for 1% of ED visits.2 Approximately 10% of patients presenting to the ED will have a serious underlying condition identified and among 3% to 5% of these patients with syncope, the serious condition will be identified only after they leave the ED.1 Most patients have a benign course, but more than half of all patients presenting to the ED with syncope will be hospitalized, costing $2.4 billion annually.2
Because of the high hospitalization rate of patients with syncope, a practical and accurate tool to risk-stratify patients is vital. Other tools, such as the San Francisco Syncope Rule, Short-Term Prognosis of Syncope, and Risk Stratification of Syncope in the Emergency Department, lack validation or are excessively complex, with extensive lab work or testing.3
The CSRS was previously derived from a large, multisite consecutive cohort, and was internally validated and reported according to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis guideline statement.4 Patients are assigned points based on clinical findings, test results, and the diagnosis given in the ED (TABLE4). The scoring system is used to stratify patients as very low (−3, −2), low (−1, 0), medium (1, 2, 3), high (4, 5), or very high (≥6) risk.4

STUDY SUMMARY
Less than 1% of very low– and low-risk patients had serious 30-day outcomes
This multisite Canadian prospective validation cohort study enrolled patients age ≥ 16 years who presented to the ED within 24 hours of syncope. Both discharged and hospitalized patients were included.1
Patients were excluded if they had loss of consciousness for > 5 minutes, mental status changes at presentation, history of current or previous seizure, or head trauma
ED physicians confirmed patient eligibility, obtained verbal consent, and completed the data collection form. In addition, research assistants sought to identify eligible patients who were not previously enrolled by reviewing all ED visits during the study period.
Continue to: To examine 30-day outcomes...
To examine 30-day outcomes, researchers reviewed all available patient medical record
A total of 4131 patients made up the validation cohort. A serious condition was identified during the initial ED visit in 160 patients (3.9%), who were excluded from the study, and 152 patients (3.7%) were lost to follow-up. Of the 3819 patients included in the final analysis, troponin was not measured in 1566 patients (41%), and an electrocardiogram was not obtained in 114 patients (3%). A serious outcome within 30 days was experienced by 139 patients (3.6%; 95% CI, 3.1%-4.3%). There was good correlation to the model-predicted serious outcome probability of 3.2% (95% CI, 2.7%-3.8%).1
Three of 1631 (0.2%) patients classified as very low risk and 9 of 1254 (0.7%) low-risk patients experienced a serious outcome, and no patients died. In the group classified as medium risk, 55 of 687 (8%) patients experienced a serious outcome, and there was 1 death. In the high-risk group, 32 of 167 (19.2%) patients experienced a serious outcome, and there were 5 deaths. In the group classified as very high risk, 40 of 78 (51.3%) patients experienced a serious outcome, and there were 7 deaths. The CSRS was able to identify very low– or low-risk patients (score of −1 or better) with a sensitivity of 97.8% (95% CI, 93.8%-99.6%) and a specificity of 44.3% (95% CI, 42.7%-45.9%).1
WHAT’S NEW
This scoring system offers a validated method to risk-stratify ED patients
Previous recommendations from the American College of Cardiology/American Heart Associationsuggested determining disposition of ED patients by using clinical judgment based on a list of risk factors such as age, chronic conditions, and medications. However, there was no scoring system.3 This new scoring system allows physicians to send home very low– and low-risk patients with reassurance that the likelihood of a serious outcome is less than 1%. High-risk and very high–risk patients should be admitted to the hospital for further evaluation. Most moderate-risk patients (8% risk of serious outcome but 0.1% risk of death) can also be discharged after providers have a risk/benefit discussion, including precautions for signs of arrhythmia or need for urgent return to the hospital.
CAVEATS
The study does not translate to all clinical settings
Because this study was done in EDs, the scoring system cannot necessarily be applied to urgent care or outpatient settings. However, 41% of the patients in the study did not have troponin testing performed. Therefore, physicians could consider using the scoring system in settings where this lab test is not immediately available.
Continue to: This scoring system was also only...
This scoring system was also only validated with adult patients presenting within 24 hours of their syncopal episode. It is unknown how it may predict the outcomes of patients who present > 24 hours after syncope.
CHALLENGES TO IMPLEMENTATION
Clinicians may not be awareof the CSRS scoring system
The main challenge to implementation is practitioner awareness of the CSRS scoring system and how to use it appropriately, as there are several different syncopal scoring systems that may already be in use. Additionally, depending on the electronic health record used, the CSRS scoring system may not be embedded. Using and documenting scores may also be a challenge.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
1. Thiruganasambandamoorthy V, Sivilotti MLA, Le Sage N, et al. Multicenter emergency department validation of the Canadian Syncope Risk Score. JAMA Intern Med. 2020;180:737-744. doi:10.1001/jamainternmed.2020.0288
2. Probst MA, Kanzaria HK, Gbedemah M, et al. National trends in resource utilization associated with ED visits for syncope. Am J Emerg Med. 2015;33:998-1001. doi:10.1016/j.ajem.2015.04.030
3. Shen WK, Sheldon RS, Benditt DG, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70:620-663. doi:10.1016/j.jacc.2017.03.002
4. Thiruganasambandamoorthy V, Kwong K, Wells GA, et al. Development of the Canadian Syncope Risk Score to predict serious adverse events after emergency department assessment of syncope. CMAJ. 2016;188:E289-E298. doi:10.1503/cmaj.151469
1. Thiruganasambandamoorthy V, Sivilotti MLA, Le Sage N, et al. Multicenter emergency department validation of the Canadian Syncope Risk Score. JAMA Intern Med. 2020;180:737-744. doi:10.1001/jamainternmed.2020.0288
2. Probst MA, Kanzaria HK, Gbedemah M, et al. National trends in resource utilization associated with ED visits for syncope. Am J Emerg Med. 2015;33:998-1001. doi:10.1016/j.ajem.2015.04.030
3. Shen WK, Sheldon RS, Benditt DG, et al. 2017 ACC/AHA/HRS guideline for the evaluation and management of patients with syncope: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2017;70:620-663. doi:10.1016/j.jacc.2017.03.002
4. Thiruganasambandamoorthy V, Kwong K, Wells GA, et al. Development of the Canadian Syncope Risk Score to predict serious adverse events after emergency department assessment of syncope. CMAJ. 2016;188:E289-E298. doi:10.1503/cmaj.151469
PRACTICE CHANGER
Physicians should use the Canadian Syncope Risk Score (CSRS) to identify and send home very low– and low-risk patients from the emergency department (ED) after a syncopal episode.
STRENGTH OF RECOMMENDATION
A: Validated clinical decision rule based on a prospective cohort study1
Thiruganasambandamoorthy V, Sivilotti MLA, Le Sage N, et al. Multicenter emergency department validation of the Canadian Syncope Risk Score. JAMA Intern Med. 2020;180:737-744. doi:10.1001/jamainternmed.2020.0288