User login
Quitting smoking after MI has huge benefits in young adults
Young adult smokers who stop smoking in the first year after an initial myocardial infarction are far less likely to die over the next 10 years than their peers who continue to smoke. Yet nearly two-thirds keep smoking after the event, according to new data from the Partners YOUNG-MI Registry.
“Smoking is one of the most common risk factors for developing an MI at a young age. ... This reinforces the need to have more young individuals avoid, or quit, the use of tobacco,” Ron Blankstein, MD, Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview.
Yet, the finding that 62% of young adults continue to smoke 1 year after MI points to an “enormous need for better smoking cessation efforts following a heart attack,” he said.
“Powerful” message for clinicians
“This study joins an incredibly powerful body of evidence that says if you quit smoking, you’re going to live longer,” said Michael Fiore, MD, MPH, MBA, director of the University of Wisconsin Center for Tobacco Research and Intervention, Madison, who wasn’t involved in the study.
“As physicians, there is nothing we can do that will have a greater impact for our patients than quitting smoking. The study is a powerful call for clinicians to intervene with their patients that smoke – both if you have an MI or if you don’t,” Dr. Fiore told this news organization.
The study involved 2,072 individuals 50 years or younger (median age, 45 years; 81% male) who were hospitalized for an initial MI at two large academic medical centers in Boston. Of these, 33.9% were never-smokers, 13.6% were former smokers, and 52.5% were smokers at the time of their MI.
During a median follow-up of 10.2 years, those who quit smoking had a significantly lower rate of death from any cause (unadjusted hazard ratio, 0.35; 95% confidence interval, 0.19-0.63; P < .001) and a cardiovascular cause (HR, 0.29; 95% CI, 0.11-0.79; P = .02), relative to those who continued to smoke.
The results remained statistically significant in a propensity-matched analysis for both all-cause (HR, 0.30; 95% CI, 0.16-0.56; P < .001) and CV mortality (HR, 0.19; 95% CI, 0.06-0.56; P = .003).
“Although patients who quit smoking were similar to those who continued to smoke with respect to their baseline characteristics, smoking cessation was associated with an approximate 70%-80% reduction in all-cause and CV mortality,” the authors note in their article, published online July 8 in JAMA Network Open.
They say it’s also noteworthy that long-term death rates of never-smokers and former smokers who quit before the MI were nearly identical.
‘A failure of our health care system’
The bottom line, said Dr. Blankstein, is that it is “never too late to quit, and those who experience an MI should do so right away. Our health care system must help promote such efforts, as there is immense room for improvement.”
Dr. Fiore said: “When I see an article like this, it just reminds me that, if you’re really thinking about staying healthy, there is nothing better you can do to improve the quality and longevity of your life than quitting smoking.”
The observation that many patients continue to smoke after MI is a “failure of our health care system, and it’s an individual failure in that these individuals are not able to overcome their powerful nicotine dependence. It’s an unfortunate occurrence that’s resulting in unnecessary deaths,” said Dr. Fiore.
There is no “magic bullet” to overcome nicotine addiction, but there are approved treatments that can “substantially boost quit rates,” he noted.
The two most effective smoking-cessation treatments are varenicline (Chantix) and combination nicotine replacement therapy, a patch combined ideally with nicotine mini lozenges, particularly when combined with some brief counseling, said Fiore.
He encourages cardiologists to get their patients to commit to quitting and then link them to resources such as 1-800-QUIT-NOW or SmokeFree.gov.
Funding for the study was provided by grants from the National Heart, Lung, and Blood Institute. Dr. Blankstein reported receiving research support from Amgen and Astellas. Dr. Fiore had no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Young adult smokers who stop smoking in the first year after an initial myocardial infarction are far less likely to die over the next 10 years than their peers who continue to smoke. Yet nearly two-thirds keep smoking after the event, according to new data from the Partners YOUNG-MI Registry.
“Smoking is one of the most common risk factors for developing an MI at a young age. ... This reinforces the need to have more young individuals avoid, or quit, the use of tobacco,” Ron Blankstein, MD, Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview.
Yet, the finding that 62% of young adults continue to smoke 1 year after MI points to an “enormous need for better smoking cessation efforts following a heart attack,” he said.
“Powerful” message for clinicians
“This study joins an incredibly powerful body of evidence that says if you quit smoking, you’re going to live longer,” said Michael Fiore, MD, MPH, MBA, director of the University of Wisconsin Center for Tobacco Research and Intervention, Madison, who wasn’t involved in the study.
“As physicians, there is nothing we can do that will have a greater impact for our patients than quitting smoking. The study is a powerful call for clinicians to intervene with their patients that smoke – both if you have an MI or if you don’t,” Dr. Fiore told this news organization.
The study involved 2,072 individuals 50 years or younger (median age, 45 years; 81% male) who were hospitalized for an initial MI at two large academic medical centers in Boston. Of these, 33.9% were never-smokers, 13.6% were former smokers, and 52.5% were smokers at the time of their MI.
During a median follow-up of 10.2 years, those who quit smoking had a significantly lower rate of death from any cause (unadjusted hazard ratio, 0.35; 95% confidence interval, 0.19-0.63; P < .001) and a cardiovascular cause (HR, 0.29; 95% CI, 0.11-0.79; P = .02), relative to those who continued to smoke.
The results remained statistically significant in a propensity-matched analysis for both all-cause (HR, 0.30; 95% CI, 0.16-0.56; P < .001) and CV mortality (HR, 0.19; 95% CI, 0.06-0.56; P = .003).
“Although patients who quit smoking were similar to those who continued to smoke with respect to their baseline characteristics, smoking cessation was associated with an approximate 70%-80% reduction in all-cause and CV mortality,” the authors note in their article, published online July 8 in JAMA Network Open.
They say it’s also noteworthy that long-term death rates of never-smokers and former smokers who quit before the MI were nearly identical.
‘A failure of our health care system’
The bottom line, said Dr. Blankstein, is that it is “never too late to quit, and those who experience an MI should do so right away. Our health care system must help promote such efforts, as there is immense room for improvement.”
Dr. Fiore said: “When I see an article like this, it just reminds me that, if you’re really thinking about staying healthy, there is nothing better you can do to improve the quality and longevity of your life than quitting smoking.”
The observation that many patients continue to smoke after MI is a “failure of our health care system, and it’s an individual failure in that these individuals are not able to overcome their powerful nicotine dependence. It’s an unfortunate occurrence that’s resulting in unnecessary deaths,” said Dr. Fiore.
There is no “magic bullet” to overcome nicotine addiction, but there are approved treatments that can “substantially boost quit rates,” he noted.
The two most effective smoking-cessation treatments are varenicline (Chantix) and combination nicotine replacement therapy, a patch combined ideally with nicotine mini lozenges, particularly when combined with some brief counseling, said Fiore.
He encourages cardiologists to get their patients to commit to quitting and then link them to resources such as 1-800-QUIT-NOW or SmokeFree.gov.
Funding for the study was provided by grants from the National Heart, Lung, and Blood Institute. Dr. Blankstein reported receiving research support from Amgen and Astellas. Dr. Fiore had no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Young adult smokers who stop smoking in the first year after an initial myocardial infarction are far less likely to die over the next 10 years than their peers who continue to smoke. Yet nearly two-thirds keep smoking after the event, according to new data from the Partners YOUNG-MI Registry.
“Smoking is one of the most common risk factors for developing an MI at a young age. ... This reinforces the need to have more young individuals avoid, or quit, the use of tobacco,” Ron Blankstein, MD, Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview.
Yet, the finding that 62% of young adults continue to smoke 1 year after MI points to an “enormous need for better smoking cessation efforts following a heart attack,” he said.
“Powerful” message for clinicians
“This study joins an incredibly powerful body of evidence that says if you quit smoking, you’re going to live longer,” said Michael Fiore, MD, MPH, MBA, director of the University of Wisconsin Center for Tobacco Research and Intervention, Madison, who wasn’t involved in the study.
“As physicians, there is nothing we can do that will have a greater impact for our patients than quitting smoking. The study is a powerful call for clinicians to intervene with their patients that smoke – both if you have an MI or if you don’t,” Dr. Fiore told this news organization.
The study involved 2,072 individuals 50 years or younger (median age, 45 years; 81% male) who were hospitalized for an initial MI at two large academic medical centers in Boston. Of these, 33.9% were never-smokers, 13.6% were former smokers, and 52.5% were smokers at the time of their MI.
During a median follow-up of 10.2 years, those who quit smoking had a significantly lower rate of death from any cause (unadjusted hazard ratio, 0.35; 95% confidence interval, 0.19-0.63; P < .001) and a cardiovascular cause (HR, 0.29; 95% CI, 0.11-0.79; P = .02), relative to those who continued to smoke.
The results remained statistically significant in a propensity-matched analysis for both all-cause (HR, 0.30; 95% CI, 0.16-0.56; P < .001) and CV mortality (HR, 0.19; 95% CI, 0.06-0.56; P = .003).
“Although patients who quit smoking were similar to those who continued to smoke with respect to their baseline characteristics, smoking cessation was associated with an approximate 70%-80% reduction in all-cause and CV mortality,” the authors note in their article, published online July 8 in JAMA Network Open.
They say it’s also noteworthy that long-term death rates of never-smokers and former smokers who quit before the MI were nearly identical.
‘A failure of our health care system’
The bottom line, said Dr. Blankstein, is that it is “never too late to quit, and those who experience an MI should do so right away. Our health care system must help promote such efforts, as there is immense room for improvement.”
Dr. Fiore said: “When I see an article like this, it just reminds me that, if you’re really thinking about staying healthy, there is nothing better you can do to improve the quality and longevity of your life than quitting smoking.”
The observation that many patients continue to smoke after MI is a “failure of our health care system, and it’s an individual failure in that these individuals are not able to overcome their powerful nicotine dependence. It’s an unfortunate occurrence that’s resulting in unnecessary deaths,” said Dr. Fiore.
There is no “magic bullet” to overcome nicotine addiction, but there are approved treatments that can “substantially boost quit rates,” he noted.
The two most effective smoking-cessation treatments are varenicline (Chantix) and combination nicotine replacement therapy, a patch combined ideally with nicotine mini lozenges, particularly when combined with some brief counseling, said Fiore.
He encourages cardiologists to get their patients to commit to quitting and then link them to resources such as 1-800-QUIT-NOW or SmokeFree.gov.
Funding for the study was provided by grants from the National Heart, Lung, and Blood Institute. Dr. Blankstein reported receiving research support from Amgen and Astellas. Dr. Fiore had no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Used together, troponin and coronary calcium improve CV risk assessment
If either high sensitivity cardiac troponin (hs-cTnT) or coronary artery calcium (CAC) are elevated, the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) climbs substantially, which suggests these biomarkers yield more prognostic information when they are used together, according to a cohort study with a median 15 years of follow-up.
Among those with a double negative result, meaning hs-cTnT was less than the limit of detection (<3 ng/L) and the CAC score was zero, only 2.8% developed ASCVD within 10 years, but the rates climbed to 4.6% if hs-cTnT was detectable and to 9.8% if the CAC score exceeded zero even when the other biomarker was negative.
“The increased risk for ASCVD among those with discordant results indicate that their prognostic information is complementary, favoring their conjoined use for risk prediction,” reported a multicenter team of investigators led by Allan S. Jaffe, MD, professor of laboratory medicine and pathology, Mayo Clinic, Rochester, Minn.
The study was performed with data from 6,749 participants in the Multi-Ethnic Study of Atherosclerosis (MESA), which is a longitudinal, community-based study funded by the National Heart, Lung, and Blood Institute. Over the course of long-term follow-up in a patient population that was about half female, 39% non-Hispanic white, 28% Black, 22% Hispanic American, and 12% Asian, ASCVD events were evaluated in relation to both biomarkers measured at baseline.
At baseline, both biomarkers were negative in 22%, both positive in 40%, and discordant in 38%.
After a median follow-up of 15 years, when 1,002 ASCVD events had occurred, the crude rate of ASCVD was 2.8 per 1,000 person-years in the double-negative group. When compared with this, the adjusted hazard ratio for ASCVD among those with double positive biomarkers was 3.5 (P < .00001). Increased risk was also highly significant if just hs-cTnT was positive (HR, 1.59; P = .003) or if just CAC was positive (HR, 2.74; P < .00001).
The added value of using both biomarkers to identify individuals at very low risk of ASCVD makes sense, according to the authors of an accompanying editorial. Written by a team led by John W. McEvoy, MB, BCh, National University of Ireland, Galway, the editorial explained why the information is complementary.
“CAC indicates subclinical atherosclerosis, whereas hs-cTnT indicates myocardial ischemia or damage, not just from coronary stenosis but also due to other conditions like hypertensive heart and left ventricular hypertrophy,” the authors stated.
Although they maintained that adding N-terminal pro-brain natriuretic peptide, which could be drawn from the same blood sample as hs-cTnT, might prove to be an even better but still simple strategy to identify low-risk patients, they praised the concept of combining biomarkers.
“If one’s wish is to identify truly low-risk individuals, then it appears that it takes two negative ASCVD biomarkers to make that wish come true,” the authors of the editorial concluded.
Relative to alternative methods of ASCVD risk assessment, measurement of these biomarkers might be useful for sparing patients from interventions, such as lipid lowering with statin therapy, being considered on the basis of conventional risk factors alone.
Dr. Jaffe said in an interview that he considers the two-biomarker assessment to be a useful tool in the low-risk population that he studied, but he does not consider this strategy as a substitute for other methods, such as those outline in the 2019 ACC/AHA guidelines that address the entire spectrum of risk, although work is planned to see if this approach can be extended to this broader group.*
“The data we have presented now is a good start and suggests that these two objective measures can identify those who are at very low risk and avoid adding individuals who may not be at as low risk if only one of the two tests is used,” Dr. Jaffe explained.
“Given there are now techniques to measure coronary calcium from any chest CT study, and that high sensitivity cardiac troponin is a relatively inexpensive test, putting them together should really help risk stratify patients,” he added.
When asked whether this approach will eventually replace conventional methods of ASCVD risk assessment, such as those proposed in the 2019 American College of Cardiology/American Heart Association guidelines for the primary prevention of cardiovascular disease (Circulation. 2019;140:e596-e646), he said maybe.
“The answer is that we will probe that question in our ongoing studies using continuous data in an attempt to evaluate how to use this approach to risk stratify larger numbers of individuals,” Dr. Jaffe replied.
The senior investigator, Dr. Jaffe, has consulting relationships with many pharmaceutical companies. The editorial authors had no relevant disclosures.
SOURCE: Sandoval Y et al. J Am Coll Cardiol. 2020;76:357-370.
*Correction, 7/27/20: An earlier version of this article mischaracterized Dr. Jaffe's statement.
If either high sensitivity cardiac troponin (hs-cTnT) or coronary artery calcium (CAC) are elevated, the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) climbs substantially, which suggests these biomarkers yield more prognostic information when they are used together, according to a cohort study with a median 15 years of follow-up.
Among those with a double negative result, meaning hs-cTnT was less than the limit of detection (<3 ng/L) and the CAC score was zero, only 2.8% developed ASCVD within 10 years, but the rates climbed to 4.6% if hs-cTnT was detectable and to 9.8% if the CAC score exceeded zero even when the other biomarker was negative.
“The increased risk for ASCVD among those with discordant results indicate that their prognostic information is complementary, favoring their conjoined use for risk prediction,” reported a multicenter team of investigators led by Allan S. Jaffe, MD, professor of laboratory medicine and pathology, Mayo Clinic, Rochester, Minn.
The study was performed with data from 6,749 participants in the Multi-Ethnic Study of Atherosclerosis (MESA), which is a longitudinal, community-based study funded by the National Heart, Lung, and Blood Institute. Over the course of long-term follow-up in a patient population that was about half female, 39% non-Hispanic white, 28% Black, 22% Hispanic American, and 12% Asian, ASCVD events were evaluated in relation to both biomarkers measured at baseline.
At baseline, both biomarkers were negative in 22%, both positive in 40%, and discordant in 38%.
After a median follow-up of 15 years, when 1,002 ASCVD events had occurred, the crude rate of ASCVD was 2.8 per 1,000 person-years in the double-negative group. When compared with this, the adjusted hazard ratio for ASCVD among those with double positive biomarkers was 3.5 (P < .00001). Increased risk was also highly significant if just hs-cTnT was positive (HR, 1.59; P = .003) or if just CAC was positive (HR, 2.74; P < .00001).
The added value of using both biomarkers to identify individuals at very low risk of ASCVD makes sense, according to the authors of an accompanying editorial. Written by a team led by John W. McEvoy, MB, BCh, National University of Ireland, Galway, the editorial explained why the information is complementary.
“CAC indicates subclinical atherosclerosis, whereas hs-cTnT indicates myocardial ischemia or damage, not just from coronary stenosis but also due to other conditions like hypertensive heart and left ventricular hypertrophy,” the authors stated.
Although they maintained that adding N-terminal pro-brain natriuretic peptide, which could be drawn from the same blood sample as hs-cTnT, might prove to be an even better but still simple strategy to identify low-risk patients, they praised the concept of combining biomarkers.
“If one’s wish is to identify truly low-risk individuals, then it appears that it takes two negative ASCVD biomarkers to make that wish come true,” the authors of the editorial concluded.
Relative to alternative methods of ASCVD risk assessment, measurement of these biomarkers might be useful for sparing patients from interventions, such as lipid lowering with statin therapy, being considered on the basis of conventional risk factors alone.
Dr. Jaffe said in an interview that he considers the two-biomarker assessment to be a useful tool in the low-risk population that he studied, but he does not consider this strategy as a substitute for other methods, such as those outline in the 2019 ACC/AHA guidelines that address the entire spectrum of risk, although work is planned to see if this approach can be extended to this broader group.*
“The data we have presented now is a good start and suggests that these two objective measures can identify those who are at very low risk and avoid adding individuals who may not be at as low risk if only one of the two tests is used,” Dr. Jaffe explained.
“Given there are now techniques to measure coronary calcium from any chest CT study, and that high sensitivity cardiac troponin is a relatively inexpensive test, putting them together should really help risk stratify patients,” he added.
When asked whether this approach will eventually replace conventional methods of ASCVD risk assessment, such as those proposed in the 2019 American College of Cardiology/American Heart Association guidelines for the primary prevention of cardiovascular disease (Circulation. 2019;140:e596-e646), he said maybe.
“The answer is that we will probe that question in our ongoing studies using continuous data in an attempt to evaluate how to use this approach to risk stratify larger numbers of individuals,” Dr. Jaffe replied.
The senior investigator, Dr. Jaffe, has consulting relationships with many pharmaceutical companies. The editorial authors had no relevant disclosures.
SOURCE: Sandoval Y et al. J Am Coll Cardiol. 2020;76:357-370.
*Correction, 7/27/20: An earlier version of this article mischaracterized Dr. Jaffe's statement.
If either high sensitivity cardiac troponin (hs-cTnT) or coronary artery calcium (CAC) are elevated, the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) climbs substantially, which suggests these biomarkers yield more prognostic information when they are used together, according to a cohort study with a median 15 years of follow-up.
Among those with a double negative result, meaning hs-cTnT was less than the limit of detection (<3 ng/L) and the CAC score was zero, only 2.8% developed ASCVD within 10 years, but the rates climbed to 4.6% if hs-cTnT was detectable and to 9.8% if the CAC score exceeded zero even when the other biomarker was negative.
“The increased risk for ASCVD among those with discordant results indicate that their prognostic information is complementary, favoring their conjoined use for risk prediction,” reported a multicenter team of investigators led by Allan S. Jaffe, MD, professor of laboratory medicine and pathology, Mayo Clinic, Rochester, Minn.
The study was performed with data from 6,749 participants in the Multi-Ethnic Study of Atherosclerosis (MESA), which is a longitudinal, community-based study funded by the National Heart, Lung, and Blood Institute. Over the course of long-term follow-up in a patient population that was about half female, 39% non-Hispanic white, 28% Black, 22% Hispanic American, and 12% Asian, ASCVD events were evaluated in relation to both biomarkers measured at baseline.
At baseline, both biomarkers were negative in 22%, both positive in 40%, and discordant in 38%.
After a median follow-up of 15 years, when 1,002 ASCVD events had occurred, the crude rate of ASCVD was 2.8 per 1,000 person-years in the double-negative group. When compared with this, the adjusted hazard ratio for ASCVD among those with double positive biomarkers was 3.5 (P < .00001). Increased risk was also highly significant if just hs-cTnT was positive (HR, 1.59; P = .003) or if just CAC was positive (HR, 2.74; P < .00001).
The added value of using both biomarkers to identify individuals at very low risk of ASCVD makes sense, according to the authors of an accompanying editorial. Written by a team led by John W. McEvoy, MB, BCh, National University of Ireland, Galway, the editorial explained why the information is complementary.
“CAC indicates subclinical atherosclerosis, whereas hs-cTnT indicates myocardial ischemia or damage, not just from coronary stenosis but also due to other conditions like hypertensive heart and left ventricular hypertrophy,” the authors stated.
Although they maintained that adding N-terminal pro-brain natriuretic peptide, which could be drawn from the same blood sample as hs-cTnT, might prove to be an even better but still simple strategy to identify low-risk patients, they praised the concept of combining biomarkers.
“If one’s wish is to identify truly low-risk individuals, then it appears that it takes two negative ASCVD biomarkers to make that wish come true,” the authors of the editorial concluded.
Relative to alternative methods of ASCVD risk assessment, measurement of these biomarkers might be useful for sparing patients from interventions, such as lipid lowering with statin therapy, being considered on the basis of conventional risk factors alone.
Dr. Jaffe said in an interview that he considers the two-biomarker assessment to be a useful tool in the low-risk population that he studied, but he does not consider this strategy as a substitute for other methods, such as those outline in the 2019 ACC/AHA guidelines that address the entire spectrum of risk, although work is planned to see if this approach can be extended to this broader group.*
“The data we have presented now is a good start and suggests that these two objective measures can identify those who are at very low risk and avoid adding individuals who may not be at as low risk if only one of the two tests is used,” Dr. Jaffe explained.
“Given there are now techniques to measure coronary calcium from any chest CT study, and that high sensitivity cardiac troponin is a relatively inexpensive test, putting them together should really help risk stratify patients,” he added.
When asked whether this approach will eventually replace conventional methods of ASCVD risk assessment, such as those proposed in the 2019 American College of Cardiology/American Heart Association guidelines for the primary prevention of cardiovascular disease (Circulation. 2019;140:e596-e646), he said maybe.
“The answer is that we will probe that question in our ongoing studies using continuous data in an attempt to evaluate how to use this approach to risk stratify larger numbers of individuals,” Dr. Jaffe replied.
The senior investigator, Dr. Jaffe, has consulting relationships with many pharmaceutical companies. The editorial authors had no relevant disclosures.
SOURCE: Sandoval Y et al. J Am Coll Cardiol. 2020;76:357-370.
*Correction, 7/27/20: An earlier version of this article mischaracterized Dr. Jaffe's statement.
FROM JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Radial artery beats saphenous vein grafting 10 years after CABG
With a median follow-up of 10 years after coronary artery bypass grafting (CABG), patients who received a radial artery graft rather than a saphenous vein graft as a second conduit were less likely to experience death, MI, or repeat revascularization, according to pooled data from five randomized trials.
The same result from the same set of data was produced after a median of 5 years, but the longer follow-up provides a more compelling case for the superiority of the radial artery graft, according to the authors of this meta-analysis, led by Mario F.L. Gaudino, MD, professor of cardiothoracic surgery at Weill Cornell Medicine, New York.
For the primary composite endpoint of death, MI, or repeat revascularization, the favorable hazard ratio at 5 years corresponded to a 33% risk reduction (HR, 0.67; P = .01), according to the previously published results (Gaudino M et al. N Engl J Med. 2018;378:2069-77).
The new data at 10 years show about the same risk reduction for the primary endpoint, but with more robust statistical significance (HR, 0.73; P < .001).
More importantly, because of the greater number of events by 10 years, the advantage of radial artery graft for the secondary composite outcome of death or MI has now reached statistical significance (HR, 0.77; P = .01).
In addition, there was a 27% reduction in risk of all-cause mortality (HR, 0.73; 95% confidence interval, 0.57-0.93) at 10 years associated with the radial artery graft. But this was not a prespecified endpoint, and so this is a hypothesis-generating post hoc finding.
The data was drawn from five randomized trials with a total of 1,036 patients. When used as an additional conduit to an internal thoracic artery in CABG, radial artery grafts relative to saphenous vein grafts were associated with a lower but nonsignificant risk of adverse outcomes in all five trials.
The advantage of radial artery grafts in the meta-analysis at 5 and now 10 years supports a series of observational studies that have also claimed better results with radial artery grafts.
The analysis was published July 14 in JAMA with essentially the same outcomes reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation in March.
However, a editorial that accompanied this meta-analysis in JAMA raised fundamental questions about revascularization.
“Intuitively, high-severity coronary lesions with significant ischemic burden, poor collateralization, and significant myocardium at risk may benefit from a durable revascularization option,” observed the editorial coauthors, Steven E. Nissen, MD, and Faisal G. Bakaeen, MD, both of the Cleveland Clinic. However, they cautioned that there is no definitive evidence that “any revascularization procedure reduces cardiovascular morbidity or mortality in patients with anatomically and physiologically stable coronary artery disease.”
They called the 10-year outcomes from the meta-analysis “the best available long-term data on the potential value of using the radial artery as a bypass conduit,” but warned that no randomized trial has confirmed that two or more conduits are superior to a single internal thoracic artery in CABG to for preventing death and major adverse cardiovascular events.
Such a trial, called ROMA, is now underway (Eur J Cardiothorac Surg. 2017;52:1031-40), but results are not expected until 2025.
In the meantime, placement of second conduits remains common in CABG procedures, about 400,000 of which are performed each year in the United States. According to Dr. Gaudino, there are indications and contraindications for second conduits, but radial artery should be the preferred standard when these are considered.
“Our data indicate that the radial artery graft should be used to complement the left internal thoracic artery in all patients who meet the indications for radial artery grafts,” he explained in an interview.
“Unfortunately, at the moment radial artery grafts are used in less than 10% of CABG cases in the U.S.,” he reported. “Hopefully, our data will lead to a larger use of this conduit by the surgical community.”
Dr. Gaudino, the principal investigator, reported no potential conflicts of interest relevant to this study.
SOURCE: Gaudino MFL et al. JAMA. 2020;324:179-87.
With a median follow-up of 10 years after coronary artery bypass grafting (CABG), patients who received a radial artery graft rather than a saphenous vein graft as a second conduit were less likely to experience death, MI, or repeat revascularization, according to pooled data from five randomized trials.
The same result from the same set of data was produced after a median of 5 years, but the longer follow-up provides a more compelling case for the superiority of the radial artery graft, according to the authors of this meta-analysis, led by Mario F.L. Gaudino, MD, professor of cardiothoracic surgery at Weill Cornell Medicine, New York.
For the primary composite endpoint of death, MI, or repeat revascularization, the favorable hazard ratio at 5 years corresponded to a 33% risk reduction (HR, 0.67; P = .01), according to the previously published results (Gaudino M et al. N Engl J Med. 2018;378:2069-77).
The new data at 10 years show about the same risk reduction for the primary endpoint, but with more robust statistical significance (HR, 0.73; P < .001).
More importantly, because of the greater number of events by 10 years, the advantage of radial artery graft for the secondary composite outcome of death or MI has now reached statistical significance (HR, 0.77; P = .01).
In addition, there was a 27% reduction in risk of all-cause mortality (HR, 0.73; 95% confidence interval, 0.57-0.93) at 10 years associated with the radial artery graft. But this was not a prespecified endpoint, and so this is a hypothesis-generating post hoc finding.
The data was drawn from five randomized trials with a total of 1,036 patients. When used as an additional conduit to an internal thoracic artery in CABG, radial artery grafts relative to saphenous vein grafts were associated with a lower but nonsignificant risk of adverse outcomes in all five trials.
The advantage of radial artery grafts in the meta-analysis at 5 and now 10 years supports a series of observational studies that have also claimed better results with radial artery grafts.
The analysis was published July 14 in JAMA with essentially the same outcomes reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation in March.
However, a editorial that accompanied this meta-analysis in JAMA raised fundamental questions about revascularization.
“Intuitively, high-severity coronary lesions with significant ischemic burden, poor collateralization, and significant myocardium at risk may benefit from a durable revascularization option,” observed the editorial coauthors, Steven E. Nissen, MD, and Faisal G. Bakaeen, MD, both of the Cleveland Clinic. However, they cautioned that there is no definitive evidence that “any revascularization procedure reduces cardiovascular morbidity or mortality in patients with anatomically and physiologically stable coronary artery disease.”
They called the 10-year outcomes from the meta-analysis “the best available long-term data on the potential value of using the radial artery as a bypass conduit,” but warned that no randomized trial has confirmed that two or more conduits are superior to a single internal thoracic artery in CABG to for preventing death and major adverse cardiovascular events.
Such a trial, called ROMA, is now underway (Eur J Cardiothorac Surg. 2017;52:1031-40), but results are not expected until 2025.
In the meantime, placement of second conduits remains common in CABG procedures, about 400,000 of which are performed each year in the United States. According to Dr. Gaudino, there are indications and contraindications for second conduits, but radial artery should be the preferred standard when these are considered.
“Our data indicate that the radial artery graft should be used to complement the left internal thoracic artery in all patients who meet the indications for radial artery grafts,” he explained in an interview.
“Unfortunately, at the moment radial artery grafts are used in less than 10% of CABG cases in the U.S.,” he reported. “Hopefully, our data will lead to a larger use of this conduit by the surgical community.”
Dr. Gaudino, the principal investigator, reported no potential conflicts of interest relevant to this study.
SOURCE: Gaudino MFL et al. JAMA. 2020;324:179-87.
With a median follow-up of 10 years after coronary artery bypass grafting (CABG), patients who received a radial artery graft rather than a saphenous vein graft as a second conduit were less likely to experience death, MI, or repeat revascularization, according to pooled data from five randomized trials.
The same result from the same set of data was produced after a median of 5 years, but the longer follow-up provides a more compelling case for the superiority of the radial artery graft, according to the authors of this meta-analysis, led by Mario F.L. Gaudino, MD, professor of cardiothoracic surgery at Weill Cornell Medicine, New York.
For the primary composite endpoint of death, MI, or repeat revascularization, the favorable hazard ratio at 5 years corresponded to a 33% risk reduction (HR, 0.67; P = .01), according to the previously published results (Gaudino M et al. N Engl J Med. 2018;378:2069-77).
The new data at 10 years show about the same risk reduction for the primary endpoint, but with more robust statistical significance (HR, 0.73; P < .001).
More importantly, because of the greater number of events by 10 years, the advantage of radial artery graft for the secondary composite outcome of death or MI has now reached statistical significance (HR, 0.77; P = .01).
In addition, there was a 27% reduction in risk of all-cause mortality (HR, 0.73; 95% confidence interval, 0.57-0.93) at 10 years associated with the radial artery graft. But this was not a prespecified endpoint, and so this is a hypothesis-generating post hoc finding.
The data was drawn from five randomized trials with a total of 1,036 patients. When used as an additional conduit to an internal thoracic artery in CABG, radial artery grafts relative to saphenous vein grafts were associated with a lower but nonsignificant risk of adverse outcomes in all five trials.
The advantage of radial artery grafts in the meta-analysis at 5 and now 10 years supports a series of observational studies that have also claimed better results with radial artery grafts.
The analysis was published July 14 in JAMA with essentially the same outcomes reported at the joint scientific sessions of the American College of Cardiology and the World Heart Federation in March.
However, a editorial that accompanied this meta-analysis in JAMA raised fundamental questions about revascularization.
“Intuitively, high-severity coronary lesions with significant ischemic burden, poor collateralization, and significant myocardium at risk may benefit from a durable revascularization option,” observed the editorial coauthors, Steven E. Nissen, MD, and Faisal G. Bakaeen, MD, both of the Cleveland Clinic. However, they cautioned that there is no definitive evidence that “any revascularization procedure reduces cardiovascular morbidity or mortality in patients with anatomically and physiologically stable coronary artery disease.”
They called the 10-year outcomes from the meta-analysis “the best available long-term data on the potential value of using the radial artery as a bypass conduit,” but warned that no randomized trial has confirmed that two or more conduits are superior to a single internal thoracic artery in CABG to for preventing death and major adverse cardiovascular events.
Such a trial, called ROMA, is now underway (Eur J Cardiothorac Surg. 2017;52:1031-40), but results are not expected until 2025.
In the meantime, placement of second conduits remains common in CABG procedures, about 400,000 of which are performed each year in the United States. According to Dr. Gaudino, there are indications and contraindications for second conduits, but radial artery should be the preferred standard when these are considered.
“Our data indicate that the radial artery graft should be used to complement the left internal thoracic artery in all patients who meet the indications for radial artery grafts,” he explained in an interview.
“Unfortunately, at the moment radial artery grafts are used in less than 10% of CABG cases in the U.S.,” he reported. “Hopefully, our data will lead to a larger use of this conduit by the surgical community.”
Dr. Gaudino, the principal investigator, reported no potential conflicts of interest relevant to this study.
SOURCE: Gaudino MFL et al. JAMA. 2020;324:179-87.
FROM JAMA
Two-stent technique shown superior for complex coronary bifurcations
A systematic two-stent approach to complex coronary bifurcation lesions led to significantly improved clinical outcomes at 1 year, compared with the long-popular provisional stenting technique, in the first randomized trial to prospectively validate a standardized definition of what constitutes a complex bifurcation.
Since the double-kissing (DK) crush technique was employed in 78% of the systematic two-stent procedures, and the two-stent approach provided superior outcomes, it’s reasonable to infer that the DK crush is the preferred technique in patients with truly complex coronary bifurcation lesions (CBLs), Shao-Liang Chen, MD, reported at the virtual annual meeting of the European Association of Percutaneous Cardiovascular Interventions.
He presented the results of the DEFINITION II trial, a multinational trial in which 653 patients at 49 medical centers who fulfilled the criteria for complex CBLs were randomized to a systematic two-stent approach or provisional stenting, with a second stent deployed by interventionalists as needed. Dr. Chen, director of the cardiology department and deputy president of Nanjing (China) Medical University, and coworkers had previously published their standardized criteria for CBLs (JACC Cardiovasc Interv. 2014 Nov;7[11]:1266-76), which they developed by analysis of a large bifurcation cohort; however, until the DEFINITION II trial, the criteria had never been used in a prospective randomized trial.
According to the standardized definition developed by Dr. Chen and associates, complex coronary bifurcation lesions must meet one major and two minor criteria.
Major criteria:
- A side branch lesion length of at least 10 mm with a diameter stenosis of 70% or more for distal left main bifurcation lesions.
- For non–left main bifurcation lesions, a side branch diameter stenosis of at least 90% along with a side branch lesion length of at least 10 mm.
Minor criteria:
- Moderate to severe calcification multiple lesions
- Bifurcation angle of <45 degrees or >70 degrees
- Thrombus-containing lesions
- Main vessel residual diameter <2.5 mm
- Main vessel lesion length of at least 25 mm
Interventionalists were strongly encouraged to utilize the DK crush or culotte stenting techniques in patients randomized to the systematic two-stent approach. In contrast, in the provisional stenting group, where 23% of patients received a second stent, that stent was placed using the T and small protrusion technique 64% of the time.
The primary endpoint was the target lesion failure rate at 1-year of follow-up. Target lesion failure was a composite comprising cardiac death, target vessel MI, and clinically driven target vessel revascularization. The rate was 6.1% in the systematic two-stent group and 11.4% with provisional stenting, for a highly significant 48% relative risk reduction. The difference was driven largely by the systematic two-stent group’s lower rates of target vessel MI – 3.0% versus 7.1% with provisional stenting – and target lesion revascularization, with rates of 2.4% and 5.5%, respectively.
“The underlying mechanisms for the increased target vessel MI rate after the provisional stenting technique are unclear, and further study is urgently warranted,” Dr. Chen said.
There were no significant between-group differences in all-cause mortality or cardiac death, although both endpoints were numerically less frequent in the two-stent group.
The primary safety outcome was the 12-month rate of definite or probable stent thrombosis. This occurred in 1.2% of the systematic two-stent group and 2.5% of the provisional stent patients, a nonsignificant difference.
Discussant Davide Capodanno, MD, PhD, declared the DEFINITE II trial to be “another success for this DK crush technique everyone is talking about recently.”
He noted that, in a recent meta-analysis of 21 randomized, controlled trials including 5,711 patients with bifurcation lesions treated using five different percutaneous coronary intervention techniques, DK crush stood out from the pack. Particularly impressive was the finding that the target lesion revascularization rate in patients treated using the DK crush technique was 64% lower than with provisional stenting (JACC Cardiovasc Interv. 2020 Jun 22;13[12]:1432-44).
Dr. Capodanno said that, although the DEFINITE II results were strongly positive in favor of the systematic two-stent approach and DK crush technique, he’s not convinced of the generalizability of the study results.
“These investigators are very expert in this technique. They invented it. They’ve been using it for 10 years. So of course you may expect excellent results when you have masters of this technique,” observed Dr. Capodanno, a cardiologist at the University of Catania (Italy).
Independent replication of the DEFINITE II findings is needed. Fortunately, two ongoing randomized trials are addressing the issue of how to best treat bifurcation lesions. The EBC-MAIN trial is comparing the provisional approach with the systematic two-stent strategy in patients with left main bifurcation lesions; the study will include the DK crush as well as culotte and TAP PCI techniques, with a primary endpoint consisting of the 12-month rate of death, MI, and target lesion revascularization. And the BBK-3 trial will compare systematic two-stent strategies pitting the culotte against the DK crush, with the primary endpoint being the 9-month rate of angiographic restenosis by quantitative coronary angiography.
“After these trials are complete, we’ll probably know much more about the tailoring of bifurcation techniques for particular patients,” according to Dr. Capodanno.
Simultaneous with Dr. Chen’s presentation, the results of the DEFINITION II trial were published online (Eur Heart J. 2020 Jun 26.doi: 10.1093/eurheartj/ehaa543).
Dr. Chen and Dr. Capodanno reported having no financial conflicts of interest regarding the study, which was funded mainly by the National Science Foundation of China.
A systematic two-stent approach to complex coronary bifurcation lesions led to significantly improved clinical outcomes at 1 year, compared with the long-popular provisional stenting technique, in the first randomized trial to prospectively validate a standardized definition of what constitutes a complex bifurcation.
Since the double-kissing (DK) crush technique was employed in 78% of the systematic two-stent procedures, and the two-stent approach provided superior outcomes, it’s reasonable to infer that the DK crush is the preferred technique in patients with truly complex coronary bifurcation lesions (CBLs), Shao-Liang Chen, MD, reported at the virtual annual meeting of the European Association of Percutaneous Cardiovascular Interventions.
He presented the results of the DEFINITION II trial, a multinational trial in which 653 patients at 49 medical centers who fulfilled the criteria for complex CBLs were randomized to a systematic two-stent approach or provisional stenting, with a second stent deployed by interventionalists as needed. Dr. Chen, director of the cardiology department and deputy president of Nanjing (China) Medical University, and coworkers had previously published their standardized criteria for CBLs (JACC Cardiovasc Interv. 2014 Nov;7[11]:1266-76), which they developed by analysis of a large bifurcation cohort; however, until the DEFINITION II trial, the criteria had never been used in a prospective randomized trial.
According to the standardized definition developed by Dr. Chen and associates, complex coronary bifurcation lesions must meet one major and two minor criteria.
Major criteria:
- A side branch lesion length of at least 10 mm with a diameter stenosis of 70% or more for distal left main bifurcation lesions.
- For non–left main bifurcation lesions, a side branch diameter stenosis of at least 90% along with a side branch lesion length of at least 10 mm.
Minor criteria:
- Moderate to severe calcification multiple lesions
- Bifurcation angle of <45 degrees or >70 degrees
- Thrombus-containing lesions
- Main vessel residual diameter <2.5 mm
- Main vessel lesion length of at least 25 mm
Interventionalists were strongly encouraged to utilize the DK crush or culotte stenting techniques in patients randomized to the systematic two-stent approach. In contrast, in the provisional stenting group, where 23% of patients received a second stent, that stent was placed using the T and small protrusion technique 64% of the time.
The primary endpoint was the target lesion failure rate at 1-year of follow-up. Target lesion failure was a composite comprising cardiac death, target vessel MI, and clinically driven target vessel revascularization. The rate was 6.1% in the systematic two-stent group and 11.4% with provisional stenting, for a highly significant 48% relative risk reduction. The difference was driven largely by the systematic two-stent group’s lower rates of target vessel MI – 3.0% versus 7.1% with provisional stenting – and target lesion revascularization, with rates of 2.4% and 5.5%, respectively.
“The underlying mechanisms for the increased target vessel MI rate after the provisional stenting technique are unclear, and further study is urgently warranted,” Dr. Chen said.
There were no significant between-group differences in all-cause mortality or cardiac death, although both endpoints were numerically less frequent in the two-stent group.
The primary safety outcome was the 12-month rate of definite or probable stent thrombosis. This occurred in 1.2% of the systematic two-stent group and 2.5% of the provisional stent patients, a nonsignificant difference.
Discussant Davide Capodanno, MD, PhD, declared the DEFINITE II trial to be “another success for this DK crush technique everyone is talking about recently.”
He noted that, in a recent meta-analysis of 21 randomized, controlled trials including 5,711 patients with bifurcation lesions treated using five different percutaneous coronary intervention techniques, DK crush stood out from the pack. Particularly impressive was the finding that the target lesion revascularization rate in patients treated using the DK crush technique was 64% lower than with provisional stenting (JACC Cardiovasc Interv. 2020 Jun 22;13[12]:1432-44).
Dr. Capodanno said that, although the DEFINITE II results were strongly positive in favor of the systematic two-stent approach and DK crush technique, he’s not convinced of the generalizability of the study results.
“These investigators are very expert in this technique. They invented it. They’ve been using it for 10 years. So of course you may expect excellent results when you have masters of this technique,” observed Dr. Capodanno, a cardiologist at the University of Catania (Italy).
Independent replication of the DEFINITE II findings is needed. Fortunately, two ongoing randomized trials are addressing the issue of how to best treat bifurcation lesions. The EBC-MAIN trial is comparing the provisional approach with the systematic two-stent strategy in patients with left main bifurcation lesions; the study will include the DK crush as well as culotte and TAP PCI techniques, with a primary endpoint consisting of the 12-month rate of death, MI, and target lesion revascularization. And the BBK-3 trial will compare systematic two-stent strategies pitting the culotte against the DK crush, with the primary endpoint being the 9-month rate of angiographic restenosis by quantitative coronary angiography.
“After these trials are complete, we’ll probably know much more about the tailoring of bifurcation techniques for particular patients,” according to Dr. Capodanno.
Simultaneous with Dr. Chen’s presentation, the results of the DEFINITION II trial were published online (Eur Heart J. 2020 Jun 26.doi: 10.1093/eurheartj/ehaa543).
Dr. Chen and Dr. Capodanno reported having no financial conflicts of interest regarding the study, which was funded mainly by the National Science Foundation of China.
A systematic two-stent approach to complex coronary bifurcation lesions led to significantly improved clinical outcomes at 1 year, compared with the long-popular provisional stenting technique, in the first randomized trial to prospectively validate a standardized definition of what constitutes a complex bifurcation.
Since the double-kissing (DK) crush technique was employed in 78% of the systematic two-stent procedures, and the two-stent approach provided superior outcomes, it’s reasonable to infer that the DK crush is the preferred technique in patients with truly complex coronary bifurcation lesions (CBLs), Shao-Liang Chen, MD, reported at the virtual annual meeting of the European Association of Percutaneous Cardiovascular Interventions.
He presented the results of the DEFINITION II trial, a multinational trial in which 653 patients at 49 medical centers who fulfilled the criteria for complex CBLs were randomized to a systematic two-stent approach or provisional stenting, with a second stent deployed by interventionalists as needed. Dr. Chen, director of the cardiology department and deputy president of Nanjing (China) Medical University, and coworkers had previously published their standardized criteria for CBLs (JACC Cardiovasc Interv. 2014 Nov;7[11]:1266-76), which they developed by analysis of a large bifurcation cohort; however, until the DEFINITION II trial, the criteria had never been used in a prospective randomized trial.
According to the standardized definition developed by Dr. Chen and associates, complex coronary bifurcation lesions must meet one major and two minor criteria.
Major criteria:
- A side branch lesion length of at least 10 mm with a diameter stenosis of 70% or more for distal left main bifurcation lesions.
- For non–left main bifurcation lesions, a side branch diameter stenosis of at least 90% along with a side branch lesion length of at least 10 mm.
Minor criteria:
- Moderate to severe calcification multiple lesions
- Bifurcation angle of <45 degrees or >70 degrees
- Thrombus-containing lesions
- Main vessel residual diameter <2.5 mm
- Main vessel lesion length of at least 25 mm
Interventionalists were strongly encouraged to utilize the DK crush or culotte stenting techniques in patients randomized to the systematic two-stent approach. In contrast, in the provisional stenting group, where 23% of patients received a second stent, that stent was placed using the T and small protrusion technique 64% of the time.
The primary endpoint was the target lesion failure rate at 1-year of follow-up. Target lesion failure was a composite comprising cardiac death, target vessel MI, and clinically driven target vessel revascularization. The rate was 6.1% in the systematic two-stent group and 11.4% with provisional stenting, for a highly significant 48% relative risk reduction. The difference was driven largely by the systematic two-stent group’s lower rates of target vessel MI – 3.0% versus 7.1% with provisional stenting – and target lesion revascularization, with rates of 2.4% and 5.5%, respectively.
“The underlying mechanisms for the increased target vessel MI rate after the provisional stenting technique are unclear, and further study is urgently warranted,” Dr. Chen said.
There were no significant between-group differences in all-cause mortality or cardiac death, although both endpoints were numerically less frequent in the two-stent group.
The primary safety outcome was the 12-month rate of definite or probable stent thrombosis. This occurred in 1.2% of the systematic two-stent group and 2.5% of the provisional stent patients, a nonsignificant difference.
Discussant Davide Capodanno, MD, PhD, declared the DEFINITE II trial to be “another success for this DK crush technique everyone is talking about recently.”
He noted that, in a recent meta-analysis of 21 randomized, controlled trials including 5,711 patients with bifurcation lesions treated using five different percutaneous coronary intervention techniques, DK crush stood out from the pack. Particularly impressive was the finding that the target lesion revascularization rate in patients treated using the DK crush technique was 64% lower than with provisional stenting (JACC Cardiovasc Interv. 2020 Jun 22;13[12]:1432-44).
Dr. Capodanno said that, although the DEFINITE II results were strongly positive in favor of the systematic two-stent approach and DK crush technique, he’s not convinced of the generalizability of the study results.
“These investigators are very expert in this technique. They invented it. They’ve been using it for 10 years. So of course you may expect excellent results when you have masters of this technique,” observed Dr. Capodanno, a cardiologist at the University of Catania (Italy).
Independent replication of the DEFINITE II findings is needed. Fortunately, two ongoing randomized trials are addressing the issue of how to best treat bifurcation lesions. The EBC-MAIN trial is comparing the provisional approach with the systematic two-stent strategy in patients with left main bifurcation lesions; the study will include the DK crush as well as culotte and TAP PCI techniques, with a primary endpoint consisting of the 12-month rate of death, MI, and target lesion revascularization. And the BBK-3 trial will compare systematic two-stent strategies pitting the culotte against the DK crush, with the primary endpoint being the 9-month rate of angiographic restenosis by quantitative coronary angiography.
“After these trials are complete, we’ll probably know much more about the tailoring of bifurcation techniques for particular patients,” according to Dr. Capodanno.
Simultaneous with Dr. Chen’s presentation, the results of the DEFINITION II trial were published online (Eur Heart J. 2020 Jun 26.doi: 10.1093/eurheartj/ehaa543).
Dr. Chen and Dr. Capodanno reported having no financial conflicts of interest regarding the study, which was funded mainly by the National Science Foundation of China.
FROM EUROPCR 2020
Zoledronic acid fails to impact abdominal aortic calcification
A single yearly dose of zoledronic acid had no impact on the progression of abdominal aortic calcification in postmenopausal women with osteoporosis, based on data from 502 women.
Although bisphosphonates have been shown to reduce the formation and progression of vascular calcification in animal studies, the impact on aortic calcification in humans has not been studied, wrote Guoqi Cai, PhD, of the University of Tasmania, Australia, and colleagues.
In a post hoc analysis published in Osteoporosis International, the researchers reviewed data from the HORIZON Pivotal Fracture trial of women with osteoporosis.
The study population included 234 postmenopausal women with osteoporosis who received an annual infusion of 5 mg zoledronic acid (ZA) and 268 who received a placebo. The mean age of the women was 72.5 years. Overall, abdominal aortic calcification (AAC) was present in 292 women (58%) at baseline, defined as an AAC score greater than 0, and AAC scores were similar between the intervention and placebo groups.
Over 3 years, AAC progressed similarly between the ZA and placebo groups (29% and 31%, respectively). Progression was defined as an increase in AAC score, which was measured by comparing spinal x-rays at baseline and after 3 years. In a subgroup analysis, progression of AAC was similar between the ZA and placebo groups with and without baseline AAC.
“The lack of effect on the progression of vascular calcification with zoledronic acid treatment in this study does not rule out a potential role of bisphosphonates in reducing cardiovascular mortality mediated through other mechanisms,” the researchers noted.
No correlation appeared between change in AAC score and change in bone mineral density at the total hip and femoral neck during the study period in any of the groups.
The study findings were limited by several factors including the post hoc analysis, potential lack of sensitivity of the AAC-8 scale in measuring small AAC changes, and homogenous study population, the researchers noted.
However, the study is the first to examine the impact of zoledronic acid on aortic calcification in humans, and was strengthened by the randomized design, the researchers said. Although other studies on the impact of bisphosphonates on vascular calcification have been inconsistent, the “finding that zoledronic acid was not protective against vascular calcification agrees with previous trials of nitrogen-containing bisphosphonates conducted in postmenopausal women with osteoporosis,” as well as chronic kidney disease patients and renal transplant patients, they said.
“Thus, our findings do not support the use of zoledronic acid for the treatment of vascular calcification,” they concluded.
The study was supported by Novartis. Dr. Cai had no financial conflicts to disclose.
SOURCE: Cai G. et al. Osteoporosis Int. 2020 May 2. doi: 10.1007/s00198-020-05430-z.
A single yearly dose of zoledronic acid had no impact on the progression of abdominal aortic calcification in postmenopausal women with osteoporosis, based on data from 502 women.
Although bisphosphonates have been shown to reduce the formation and progression of vascular calcification in animal studies, the impact on aortic calcification in humans has not been studied, wrote Guoqi Cai, PhD, of the University of Tasmania, Australia, and colleagues.
In a post hoc analysis published in Osteoporosis International, the researchers reviewed data from the HORIZON Pivotal Fracture trial of women with osteoporosis.
The study population included 234 postmenopausal women with osteoporosis who received an annual infusion of 5 mg zoledronic acid (ZA) and 268 who received a placebo. The mean age of the women was 72.5 years. Overall, abdominal aortic calcification (AAC) was present in 292 women (58%) at baseline, defined as an AAC score greater than 0, and AAC scores were similar between the intervention and placebo groups.
Over 3 years, AAC progressed similarly between the ZA and placebo groups (29% and 31%, respectively). Progression was defined as an increase in AAC score, which was measured by comparing spinal x-rays at baseline and after 3 years. In a subgroup analysis, progression of AAC was similar between the ZA and placebo groups with and without baseline AAC.
“The lack of effect on the progression of vascular calcification with zoledronic acid treatment in this study does not rule out a potential role of bisphosphonates in reducing cardiovascular mortality mediated through other mechanisms,” the researchers noted.
No correlation appeared between change in AAC score and change in bone mineral density at the total hip and femoral neck during the study period in any of the groups.
The study findings were limited by several factors including the post hoc analysis, potential lack of sensitivity of the AAC-8 scale in measuring small AAC changes, and homogenous study population, the researchers noted.
However, the study is the first to examine the impact of zoledronic acid on aortic calcification in humans, and was strengthened by the randomized design, the researchers said. Although other studies on the impact of bisphosphonates on vascular calcification have been inconsistent, the “finding that zoledronic acid was not protective against vascular calcification agrees with previous trials of nitrogen-containing bisphosphonates conducted in postmenopausal women with osteoporosis,” as well as chronic kidney disease patients and renal transplant patients, they said.
“Thus, our findings do not support the use of zoledronic acid for the treatment of vascular calcification,” they concluded.
The study was supported by Novartis. Dr. Cai had no financial conflicts to disclose.
SOURCE: Cai G. et al. Osteoporosis Int. 2020 May 2. doi: 10.1007/s00198-020-05430-z.
A single yearly dose of zoledronic acid had no impact on the progression of abdominal aortic calcification in postmenopausal women with osteoporosis, based on data from 502 women.
Although bisphosphonates have been shown to reduce the formation and progression of vascular calcification in animal studies, the impact on aortic calcification in humans has not been studied, wrote Guoqi Cai, PhD, of the University of Tasmania, Australia, and colleagues.
In a post hoc analysis published in Osteoporosis International, the researchers reviewed data from the HORIZON Pivotal Fracture trial of women with osteoporosis.
The study population included 234 postmenopausal women with osteoporosis who received an annual infusion of 5 mg zoledronic acid (ZA) and 268 who received a placebo. The mean age of the women was 72.5 years. Overall, abdominal aortic calcification (AAC) was present in 292 women (58%) at baseline, defined as an AAC score greater than 0, and AAC scores were similar between the intervention and placebo groups.
Over 3 years, AAC progressed similarly between the ZA and placebo groups (29% and 31%, respectively). Progression was defined as an increase in AAC score, which was measured by comparing spinal x-rays at baseline and after 3 years. In a subgroup analysis, progression of AAC was similar between the ZA and placebo groups with and without baseline AAC.
“The lack of effect on the progression of vascular calcification with zoledronic acid treatment in this study does not rule out a potential role of bisphosphonates in reducing cardiovascular mortality mediated through other mechanisms,” the researchers noted.
No correlation appeared between change in AAC score and change in bone mineral density at the total hip and femoral neck during the study period in any of the groups.
The study findings were limited by several factors including the post hoc analysis, potential lack of sensitivity of the AAC-8 scale in measuring small AAC changes, and homogenous study population, the researchers noted.
However, the study is the first to examine the impact of zoledronic acid on aortic calcification in humans, and was strengthened by the randomized design, the researchers said. Although other studies on the impact of bisphosphonates on vascular calcification have been inconsistent, the “finding that zoledronic acid was not protective against vascular calcification agrees with previous trials of nitrogen-containing bisphosphonates conducted in postmenopausal women with osteoporosis,” as well as chronic kidney disease patients and renal transplant patients, they said.
“Thus, our findings do not support the use of zoledronic acid for the treatment of vascular calcification,” they concluded.
The study was supported by Novartis. Dr. Cai had no financial conflicts to disclose.
SOURCE: Cai G. et al. Osteoporosis Int. 2020 May 2. doi: 10.1007/s00198-020-05430-z.
FROM OSTEOPOROSIS INTERNATIONAL
U.S. adults reach Healthy People 2020 cholesterol goal
Good news: High cholesterol is down in the United States. More good news: Low HDL cholesterol is down in the United States.
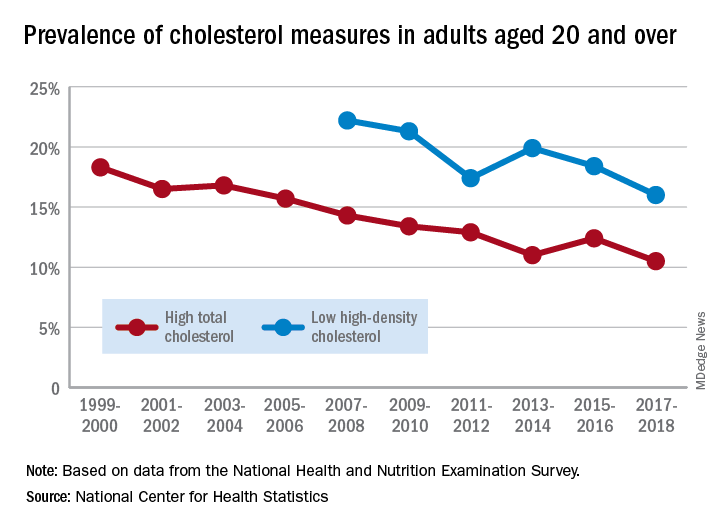
The prevalence of high total cholesterol in adults aged 20 years and older dropped from 18.3% in 1999-2000 to 10.5% in 2017-2018. And starting in 2007-2008, the prevalence of low HDL cholesterol declined from 22.2% to 16.0% in 2017-2018, the National Center for Health Statistics reported.
HDL cholesterol data before 2007 were not presented because of changes in laboratories and methods, but both trends are significant, and the decline in high total cholesterol means that the Healthy People 2020 goal of dropping prevalence to 13.5% has been met, said Margaret D. Carroll, MSPH, and Cheryl D. Fryar, MSPH, of the NCHS.
The demographic details, however, show some disparities hidden by the broader measures. The prevalence of low HDL cholesterol for women in 2015-2018 was 8.5%, but for men it was 26.6%, the NCHS investigators said.
And that Healthy People 2020 goal for total cholesterol? Age makes a difference: 7.5% of adults aged 20-39 years had high total cholesterol in 2015-2018, as did 11.4% of those aged 60 years and older, but those aged 40-59 years had a significantly higher prevalence of 15.7%, they reported.
Race/ethnicity was also a factor. Prevalence of low HDL was similar for white (16.6%) and Asian (15.8%) adults in 2015-2018, but black adults’ low HDL prevalence was significantly lower (11.9%) and Hispanics’ was significantly higher (21.9%), the researchers said.
The analysis was based on data from the National Health and Nutrition Examination Survey. The investigators defined high total cholesterol as a level of 240 mg/dL or more, and low HDL cholesterol as less than 40 mg/dL. LDL cholesterol was not included in the analysis.
Good news: High cholesterol is down in the United States. More good news: Low HDL cholesterol is down in the United States.

The prevalence of high total cholesterol in adults aged 20 years and older dropped from 18.3% in 1999-2000 to 10.5% in 2017-2018. And starting in 2007-2008, the prevalence of low HDL cholesterol declined from 22.2% to 16.0% in 2017-2018, the National Center for Health Statistics reported.
HDL cholesterol data before 2007 were not presented because of changes in laboratories and methods, but both trends are significant, and the decline in high total cholesterol means that the Healthy People 2020 goal of dropping prevalence to 13.5% has been met, said Margaret D. Carroll, MSPH, and Cheryl D. Fryar, MSPH, of the NCHS.
The demographic details, however, show some disparities hidden by the broader measures. The prevalence of low HDL cholesterol for women in 2015-2018 was 8.5%, but for men it was 26.6%, the NCHS investigators said.
And that Healthy People 2020 goal for total cholesterol? Age makes a difference: 7.5% of adults aged 20-39 years had high total cholesterol in 2015-2018, as did 11.4% of those aged 60 years and older, but those aged 40-59 years had a significantly higher prevalence of 15.7%, they reported.
Race/ethnicity was also a factor. Prevalence of low HDL was similar for white (16.6%) and Asian (15.8%) adults in 2015-2018, but black adults’ low HDL prevalence was significantly lower (11.9%) and Hispanics’ was significantly higher (21.9%), the researchers said.
The analysis was based on data from the National Health and Nutrition Examination Survey. The investigators defined high total cholesterol as a level of 240 mg/dL or more, and low HDL cholesterol as less than 40 mg/dL. LDL cholesterol was not included in the analysis.
Good news: High cholesterol is down in the United States. More good news: Low HDL cholesterol is down in the United States.

The prevalence of high total cholesterol in adults aged 20 years and older dropped from 18.3% in 1999-2000 to 10.5% in 2017-2018. And starting in 2007-2008, the prevalence of low HDL cholesterol declined from 22.2% to 16.0% in 2017-2018, the National Center for Health Statistics reported.
HDL cholesterol data before 2007 were not presented because of changes in laboratories and methods, but both trends are significant, and the decline in high total cholesterol means that the Healthy People 2020 goal of dropping prevalence to 13.5% has been met, said Margaret D. Carroll, MSPH, and Cheryl D. Fryar, MSPH, of the NCHS.
The demographic details, however, show some disparities hidden by the broader measures. The prevalence of low HDL cholesterol for women in 2015-2018 was 8.5%, but for men it was 26.6%, the NCHS investigators said.
And that Healthy People 2020 goal for total cholesterol? Age makes a difference: 7.5% of adults aged 20-39 years had high total cholesterol in 2015-2018, as did 11.4% of those aged 60 years and older, but those aged 40-59 years had a significantly higher prevalence of 15.7%, they reported.
Race/ethnicity was also a factor. Prevalence of low HDL was similar for white (16.6%) and Asian (15.8%) adults in 2015-2018, but black adults’ low HDL prevalence was significantly lower (11.9%) and Hispanics’ was significantly higher (21.9%), the researchers said.
The analysis was based on data from the National Health and Nutrition Examination Survey. The investigators defined high total cholesterol as a level of 240 mg/dL or more, and low HDL cholesterol as less than 40 mg/dL. LDL cholesterol was not included in the analysis.
CAC scoring pinpoints stenoses in asymptomatic diabetes patients
For diabetes patients with no cardiovascular symptoms despite certain risk factors, incorporating coronary calcium scoring into a silent myocardial ischemia screening algorithm may be an effective and cost-conscious strategy that avoids missed coronary stenoses suitable for revascularization, results of a recent study suggest.
Zero patients in need of revascularization were missed in a risk stratification model in which screening for silent myocardial ischemia (SMI) was done only for patients with peripheral artery disease, severe nephropathy, or a high coronary artery calcium (CAC) score, according to investigator Paul Valensi, MD.
In practical terms, that means stress myocardial scintigraphy to detect SMI could be reserved for patients with evidence of target organ damage or a CAC score of 100 or higher, according to Dr. Valensi, head of the department of endocrinology, diabetology, and nutrition at Jean Verdier Hospital in Bondy, France.
“The strategy appears to be a good compromise, and the most cost effective strategy,” Dr. Valensi said in a presentation of the results at the virtual annual scientific sessions of the American Diabetes Association.
Utility of CAC scoring in diabetes
This algorithm proposed by Dr. Valenti and colleagues is a “reasonable” approach to guide risk stratification in asymptomatic diabetes patients, said Matthew J. Budoff, MD, professor of medicine and director of cardiac CT at Harbor-UCLA Medical Center in Torrance, Calif.
“Calcium scoring could certainly help you identify those patients (at increased risk) as a first-line test, because if their calcium score is zero, their chance of having obstructive disease is probably either zero or very close to zero,” Dr. Budoff said in an interview.
Using CAC scores to assess cardiovascular risk in asymptomatic adults with diabetes was supported by 2010 guidelines from the American College of Cardiology and the American Heart Association, Dr. Budoff said, while 2019 guidelines from the European Society of Cardiology (ESC) describe CAC score combined with CT as a potential risk modifier in the evaluation of certain asymptomatic patients with diabetes.
“We are starting to see that we might be able to understand diabetes better and the cardiovascular implications by understanding how much plaque (patients) have at the time that we see them,” Dr. Budoff said in a presentation on use of CAC scans he gave earlier at the virtual ADA meeting.
In the interview, Dr. Budoff also noted that CAC scores may be particularly useful for guiding use of statins, PCSK9 (proprotein convertase subtilisin kexin 9) inhibitors, or other treatments in patients with diabetes: “There are a lot of therapies that we can apply, if we knew somebody was at higher risk, that would potentially help them avoid a heart attack, stroke, or cardiovascular death,” he said.
CAC scoring and coronary artery stenoses
Although about 20% of patients with type 2 diabetes have SMI, screening for it is “debated,” according to Dr. Valensi.
The recent ESC guidelines state that while routine screening for coronary artery disease in asymptomatic diabetics is not recommended, stress testing or coronary angiography “may be indicated” in asymptomatic diabetics in the very-high cardiovascular risk category.
That position is based on a lack of benefit seen with a broad screening strategy, the guidelines say, possibly due in part to low event rates in randomized controlled trials that have studied the approach.
Using CAC scoring could change the equation by helping to identify a greater proportion of type 2 diabetics with SMI, according to Dr. Valensi.
“The role of the CAC score in the strategy of detection of SMI needs to be defined, and this role may depend on the a priori cardiovascular risk,” he said.
Dr. Valensi and colleagues accordingly tested several different approaches to selecting asymptomatic diabetic patients for SMI screening to see how they would perform in finding patients with coronary stenoses eligible for revascularization.
Their study included 416 diabetes patients with diabetes at very high cardiovascular risk but with no cardiac history or symptoms. A total of 40 patients (9.6%) had SMI, including 15 patients in which coronary stenoses were found; of those, 11 (73.5%) underwent a revascularization procedure.
They found that, by performing myocardial scintigraphy only in those patients with peripheral artery disease or severe nephropathy, they would have missed 6 patients with coronary stenosis suitable for revascularization among the 275 patients who did not meet those target organ damage criteria.
By contrast, zero patients would have been missed by performing myocardial scintigraphy in patients who either met those target organ damage criteria, or who had an elevated CAC score.
“We suggest screening for SMI, using stress myocardial CT scanning and coronary stenosis screening, only the patients with peripheral artery disease or severe nephropathy or with a high CAC score over 100 Agatston units,” said Dr. Valensi.
Dr. Valensi reported disclosures related to Merck Sharp Dohme, Novo Nordisk, Pierre Fabre, Eli Lilly, Bristol-Myers Squibb, AstraZeneca, Daiichi-Sankyo, and others. Coauthors provided no disclosures related to the research. Dr. Budoff reported that he has served as a paid consultant to GE.
SOURCE: Berkane N et al. ADA 2020. Abstract 8-OR.
For diabetes patients with no cardiovascular symptoms despite certain risk factors, incorporating coronary calcium scoring into a silent myocardial ischemia screening algorithm may be an effective and cost-conscious strategy that avoids missed coronary stenoses suitable for revascularization, results of a recent study suggest.
Zero patients in need of revascularization were missed in a risk stratification model in which screening for silent myocardial ischemia (SMI) was done only for patients with peripheral artery disease, severe nephropathy, or a high coronary artery calcium (CAC) score, according to investigator Paul Valensi, MD.
In practical terms, that means stress myocardial scintigraphy to detect SMI could be reserved for patients with evidence of target organ damage or a CAC score of 100 or higher, according to Dr. Valensi, head of the department of endocrinology, diabetology, and nutrition at Jean Verdier Hospital in Bondy, France.
“The strategy appears to be a good compromise, and the most cost effective strategy,” Dr. Valensi said in a presentation of the results at the virtual annual scientific sessions of the American Diabetes Association.
Utility of CAC scoring in diabetes
This algorithm proposed by Dr. Valenti and colleagues is a “reasonable” approach to guide risk stratification in asymptomatic diabetes patients, said Matthew J. Budoff, MD, professor of medicine and director of cardiac CT at Harbor-UCLA Medical Center in Torrance, Calif.
“Calcium scoring could certainly help you identify those patients (at increased risk) as a first-line test, because if their calcium score is zero, their chance of having obstructive disease is probably either zero or very close to zero,” Dr. Budoff said in an interview.
Using CAC scores to assess cardiovascular risk in asymptomatic adults with diabetes was supported by 2010 guidelines from the American College of Cardiology and the American Heart Association, Dr. Budoff said, while 2019 guidelines from the European Society of Cardiology (ESC) describe CAC score combined with CT as a potential risk modifier in the evaluation of certain asymptomatic patients with diabetes.
“We are starting to see that we might be able to understand diabetes better and the cardiovascular implications by understanding how much plaque (patients) have at the time that we see them,” Dr. Budoff said in a presentation on use of CAC scans he gave earlier at the virtual ADA meeting.
In the interview, Dr. Budoff also noted that CAC scores may be particularly useful for guiding use of statins, PCSK9 (proprotein convertase subtilisin kexin 9) inhibitors, or other treatments in patients with diabetes: “There are a lot of therapies that we can apply, if we knew somebody was at higher risk, that would potentially help them avoid a heart attack, stroke, or cardiovascular death,” he said.
CAC scoring and coronary artery stenoses
Although about 20% of patients with type 2 diabetes have SMI, screening for it is “debated,” according to Dr. Valensi.
The recent ESC guidelines state that while routine screening for coronary artery disease in asymptomatic diabetics is not recommended, stress testing or coronary angiography “may be indicated” in asymptomatic diabetics in the very-high cardiovascular risk category.
That position is based on a lack of benefit seen with a broad screening strategy, the guidelines say, possibly due in part to low event rates in randomized controlled trials that have studied the approach.
Using CAC scoring could change the equation by helping to identify a greater proportion of type 2 diabetics with SMI, according to Dr. Valensi.
“The role of the CAC score in the strategy of detection of SMI needs to be defined, and this role may depend on the a priori cardiovascular risk,” he said.
Dr. Valensi and colleagues accordingly tested several different approaches to selecting asymptomatic diabetic patients for SMI screening to see how they would perform in finding patients with coronary stenoses eligible for revascularization.
Their study included 416 diabetes patients with diabetes at very high cardiovascular risk but with no cardiac history or symptoms. A total of 40 patients (9.6%) had SMI, including 15 patients in which coronary stenoses were found; of those, 11 (73.5%) underwent a revascularization procedure.
They found that, by performing myocardial scintigraphy only in those patients with peripheral artery disease or severe nephropathy, they would have missed 6 patients with coronary stenosis suitable for revascularization among the 275 patients who did not meet those target organ damage criteria.
By contrast, zero patients would have been missed by performing myocardial scintigraphy in patients who either met those target organ damage criteria, or who had an elevated CAC score.
“We suggest screening for SMI, using stress myocardial CT scanning and coronary stenosis screening, only the patients with peripheral artery disease or severe nephropathy or with a high CAC score over 100 Agatston units,” said Dr. Valensi.
Dr. Valensi reported disclosures related to Merck Sharp Dohme, Novo Nordisk, Pierre Fabre, Eli Lilly, Bristol-Myers Squibb, AstraZeneca, Daiichi-Sankyo, and others. Coauthors provided no disclosures related to the research. Dr. Budoff reported that he has served as a paid consultant to GE.
SOURCE: Berkane N et al. ADA 2020. Abstract 8-OR.
For diabetes patients with no cardiovascular symptoms despite certain risk factors, incorporating coronary calcium scoring into a silent myocardial ischemia screening algorithm may be an effective and cost-conscious strategy that avoids missed coronary stenoses suitable for revascularization, results of a recent study suggest.
Zero patients in need of revascularization were missed in a risk stratification model in which screening for silent myocardial ischemia (SMI) was done only for patients with peripheral artery disease, severe nephropathy, or a high coronary artery calcium (CAC) score, according to investigator Paul Valensi, MD.
In practical terms, that means stress myocardial scintigraphy to detect SMI could be reserved for patients with evidence of target organ damage or a CAC score of 100 or higher, according to Dr. Valensi, head of the department of endocrinology, diabetology, and nutrition at Jean Verdier Hospital in Bondy, France.
“The strategy appears to be a good compromise, and the most cost effective strategy,” Dr. Valensi said in a presentation of the results at the virtual annual scientific sessions of the American Diabetes Association.
Utility of CAC scoring in diabetes
This algorithm proposed by Dr. Valenti and colleagues is a “reasonable” approach to guide risk stratification in asymptomatic diabetes patients, said Matthew J. Budoff, MD, professor of medicine and director of cardiac CT at Harbor-UCLA Medical Center in Torrance, Calif.
“Calcium scoring could certainly help you identify those patients (at increased risk) as a first-line test, because if their calcium score is zero, their chance of having obstructive disease is probably either zero or very close to zero,” Dr. Budoff said in an interview.
Using CAC scores to assess cardiovascular risk in asymptomatic adults with diabetes was supported by 2010 guidelines from the American College of Cardiology and the American Heart Association, Dr. Budoff said, while 2019 guidelines from the European Society of Cardiology (ESC) describe CAC score combined with CT as a potential risk modifier in the evaluation of certain asymptomatic patients with diabetes.
“We are starting to see that we might be able to understand diabetes better and the cardiovascular implications by understanding how much plaque (patients) have at the time that we see them,” Dr. Budoff said in a presentation on use of CAC scans he gave earlier at the virtual ADA meeting.
In the interview, Dr. Budoff also noted that CAC scores may be particularly useful for guiding use of statins, PCSK9 (proprotein convertase subtilisin kexin 9) inhibitors, or other treatments in patients with diabetes: “There are a lot of therapies that we can apply, if we knew somebody was at higher risk, that would potentially help them avoid a heart attack, stroke, or cardiovascular death,” he said.
CAC scoring and coronary artery stenoses
Although about 20% of patients with type 2 diabetes have SMI, screening for it is “debated,” according to Dr. Valensi.
The recent ESC guidelines state that while routine screening for coronary artery disease in asymptomatic diabetics is not recommended, stress testing or coronary angiography “may be indicated” in asymptomatic diabetics in the very-high cardiovascular risk category.
That position is based on a lack of benefit seen with a broad screening strategy, the guidelines say, possibly due in part to low event rates in randomized controlled trials that have studied the approach.
Using CAC scoring could change the equation by helping to identify a greater proportion of type 2 diabetics with SMI, according to Dr. Valensi.
“The role of the CAC score in the strategy of detection of SMI needs to be defined, and this role may depend on the a priori cardiovascular risk,” he said.
Dr. Valensi and colleagues accordingly tested several different approaches to selecting asymptomatic diabetic patients for SMI screening to see how they would perform in finding patients with coronary stenoses eligible for revascularization.
Their study included 416 diabetes patients with diabetes at very high cardiovascular risk but with no cardiac history or symptoms. A total of 40 patients (9.6%) had SMI, including 15 patients in which coronary stenoses were found; of those, 11 (73.5%) underwent a revascularization procedure.
They found that, by performing myocardial scintigraphy only in those patients with peripheral artery disease or severe nephropathy, they would have missed 6 patients with coronary stenosis suitable for revascularization among the 275 patients who did not meet those target organ damage criteria.
By contrast, zero patients would have been missed by performing myocardial scintigraphy in patients who either met those target organ damage criteria, or who had an elevated CAC score.
“We suggest screening for SMI, using stress myocardial CT scanning and coronary stenosis screening, only the patients with peripheral artery disease or severe nephropathy or with a high CAC score over 100 Agatston units,” said Dr. Valensi.
Dr. Valensi reported disclosures related to Merck Sharp Dohme, Novo Nordisk, Pierre Fabre, Eli Lilly, Bristol-Myers Squibb, AstraZeneca, Daiichi-Sankyo, and others. Coauthors provided no disclosures related to the research. Dr. Budoff reported that he has served as a paid consultant to GE.
SOURCE: Berkane N et al. ADA 2020. Abstract 8-OR.
FROM ADA 2020
RA raises cardiac risk even without CAD
In patients with rheumatoid arthritis (RA), strategies to prevent cardiovascular events, such as treating hypertension, encouraging patients to stop smoking, and reinforcing statin therapy, may be especially important, regardless of whether they have a history of coronary artery disease because their risk for adverse cardiovascular outcomes is significantly greater than for patients who have neither RA nor coronary artery disease (CAD), a large population-based study from Denmark suggests.
“Among patients with RA, risk stratification by presence or absence of documented CAD may allow for screening and personalized treatment strategies,” wrote Brian B. Løgstrup, MD, PhD, DMSc, of Aarhus (Denmark) University Hospital, and his colleagues.
The study, published in Annals of the Rheumatic Diseases, analyzed 125,331 patients with and without CAD in the Western Denmark Heart Registry who had coronary angiography from 2003 through 2016. The cohort included 671 RA patients with no confirmed CAD and 1,061 RA patients who had CAD.
The study makes a significant contribution to the literature in reporting on the additive risk of RA and CAD, said Christie M. Bartels, MD, associate professor in the division of rheumatology at the University of Wisconsin, Madison. “Even among patients with both conditions [RA and CVD], they were less likely to get statin therapy,” she said, noting that the 82.6% of study patients with both CAD and RA were on statins vs. 86.5% of those with CAD alone, while the former had significantly higher rates of hypertension – 64.3% vs. 58.8%. “We’re doing a less effective job on secondary prevention,” she said. The anti-inflammatory properties of statins can also have an additive benefit in RA, she noted.
“This study shows that the rheumatologist can play a role in reinforcing the importance of primary and secondary cardiovascular disease prevention – meaning hypertension control, counseling patients to stop smoking and following up on statin therapy in RA,” Dr. Bartels added.
The study presents two novel findings, Dr. Løgstrup and colleagues noted:
- That RA confers a statistically significant, “but numerically marginally,” heightened risk of cardiovascular events other than stroke.
- Among patients with CAD, RA confers an increased risk of cardiac and all-cause death as well as MI and major adverse cardiovascular events (MACE).
“These finding indicate that RA may have a potential impact for precipitating cardiovascular events beyond CAD and, even more importantly, that RA seems to exacerbate the clinical risk of cardiovascular events in the presence of CAD,” Dr. Løgstrup and colleagues wrote.
The study found that patients with neither RA nor CAD had the lowest 10-year rates of MI (2.7%), ischemic stroke (2.9%), all-cause death (21.6%), cardiac death (2.3%), and MACE (7.3%).
By comparison, those with RA but no CAD had 10-year rates of 3.8% for MI, 5.5% for stroke, 35.6% for all-cause death, 3% for cardiac death, and 11.5% for MACE. Rates for those outcomes for people with CAD but no RA were 9.9% for MI, 4.6% for stroke, 33.3% for all-cause death, 7% for cardiac death, and 19.1% for MACE.
For patients with both RA and CAD, 10-year rates were 12.2% for MI, 4.4% for stroke, 49% for all-cause death, 10.9% for cardiac death, and 24.3% for MACE.
The researchers also performed a risk adjustment analysis based on potential confounding variables across the different groups, such as age, gender, comorbidities including diabetes and hypertension, active smoking status, and anticoagulant, antiplatelet, and statin therapy. The adjusted analysis revealed that patients with RA alone had a 63% greater risk of MI, 68% greater risk for stroke, 42% greater risk for all-cause death, 25% greater risk for cardiac death, and 60% greater risk for MACE than did people who had neither RA nor CAD.
For people with both RA and CAD, the adjusted risks were significantly higher when compared to people with neither: more than four times greater for MI and MACE, 55% greater for stroke, almost double for all-cause death, and 3.7 times greater for cardiac death. People with CAD but no RA also had higher adjusted risk rates compared to people with neither, but had variable rates when compared to people with RA but no CAD, and significantly lower adjusted rates compared to people with both.
The nature of CAD was also a factor, Dr. Løgstrup and colleagues noted. “We found more non-obstructive CAD but no increased incidence of one-vessel, two-vessel, and three-vessel disease in patients with RA than in patients without RA,” they wrote. That’s in line with other published studies (Semin Arthritis Rheum. 2010;40[3]:215–21 and J Rheumatol. 2007;34[5]:937–42), but counter to a study that found increased plaque burden and higher rates of multivessel disease among people with RA (Ann Rheum Dis. 2014;73:1797–804). Differences in methodology, vessel disease definitions, and study population may explain these deviations.
The study authors did not declare any outside source of funding or any competing interests.
Dr. Bartels disclosed receiving institutional grant funding through Pfizer.
SOURCE: Løgstrup BB et al. Ann Rheum Dis. 2020 May 29. doi: 10.1136/annrheumdis-2020-217154.
In patients with rheumatoid arthritis (RA), strategies to prevent cardiovascular events, such as treating hypertension, encouraging patients to stop smoking, and reinforcing statin therapy, may be especially important, regardless of whether they have a history of coronary artery disease because their risk for adverse cardiovascular outcomes is significantly greater than for patients who have neither RA nor coronary artery disease (CAD), a large population-based study from Denmark suggests.
“Among patients with RA, risk stratification by presence or absence of documented CAD may allow for screening and personalized treatment strategies,” wrote Brian B. Løgstrup, MD, PhD, DMSc, of Aarhus (Denmark) University Hospital, and his colleagues.
The study, published in Annals of the Rheumatic Diseases, analyzed 125,331 patients with and without CAD in the Western Denmark Heart Registry who had coronary angiography from 2003 through 2016. The cohort included 671 RA patients with no confirmed CAD and 1,061 RA patients who had CAD.
The study makes a significant contribution to the literature in reporting on the additive risk of RA and CAD, said Christie M. Bartels, MD, associate professor in the division of rheumatology at the University of Wisconsin, Madison. “Even among patients with both conditions [RA and CVD], they were less likely to get statin therapy,” she said, noting that the 82.6% of study patients with both CAD and RA were on statins vs. 86.5% of those with CAD alone, while the former had significantly higher rates of hypertension – 64.3% vs. 58.8%. “We’re doing a less effective job on secondary prevention,” she said. The anti-inflammatory properties of statins can also have an additive benefit in RA, she noted.
“This study shows that the rheumatologist can play a role in reinforcing the importance of primary and secondary cardiovascular disease prevention – meaning hypertension control, counseling patients to stop smoking and following up on statin therapy in RA,” Dr. Bartels added.
The study presents two novel findings, Dr. Løgstrup and colleagues noted:
- That RA confers a statistically significant, “but numerically marginally,” heightened risk of cardiovascular events other than stroke.
- Among patients with CAD, RA confers an increased risk of cardiac and all-cause death as well as MI and major adverse cardiovascular events (MACE).
“These finding indicate that RA may have a potential impact for precipitating cardiovascular events beyond CAD and, even more importantly, that RA seems to exacerbate the clinical risk of cardiovascular events in the presence of CAD,” Dr. Løgstrup and colleagues wrote.
The study found that patients with neither RA nor CAD had the lowest 10-year rates of MI (2.7%), ischemic stroke (2.9%), all-cause death (21.6%), cardiac death (2.3%), and MACE (7.3%).
By comparison, those with RA but no CAD had 10-year rates of 3.8% for MI, 5.5% for stroke, 35.6% for all-cause death, 3% for cardiac death, and 11.5% for MACE. Rates for those outcomes for people with CAD but no RA were 9.9% for MI, 4.6% for stroke, 33.3% for all-cause death, 7% for cardiac death, and 19.1% for MACE.
For patients with both RA and CAD, 10-year rates were 12.2% for MI, 4.4% for stroke, 49% for all-cause death, 10.9% for cardiac death, and 24.3% for MACE.
The researchers also performed a risk adjustment analysis based on potential confounding variables across the different groups, such as age, gender, comorbidities including diabetes and hypertension, active smoking status, and anticoagulant, antiplatelet, and statin therapy. The adjusted analysis revealed that patients with RA alone had a 63% greater risk of MI, 68% greater risk for stroke, 42% greater risk for all-cause death, 25% greater risk for cardiac death, and 60% greater risk for MACE than did people who had neither RA nor CAD.
For people with both RA and CAD, the adjusted risks were significantly higher when compared to people with neither: more than four times greater for MI and MACE, 55% greater for stroke, almost double for all-cause death, and 3.7 times greater for cardiac death. People with CAD but no RA also had higher adjusted risk rates compared to people with neither, but had variable rates when compared to people with RA but no CAD, and significantly lower adjusted rates compared to people with both.
The nature of CAD was also a factor, Dr. Løgstrup and colleagues noted. “We found more non-obstructive CAD but no increased incidence of one-vessel, two-vessel, and three-vessel disease in patients with RA than in patients without RA,” they wrote. That’s in line with other published studies (Semin Arthritis Rheum. 2010;40[3]:215–21 and J Rheumatol. 2007;34[5]:937–42), but counter to a study that found increased plaque burden and higher rates of multivessel disease among people with RA (Ann Rheum Dis. 2014;73:1797–804). Differences in methodology, vessel disease definitions, and study population may explain these deviations.
The study authors did not declare any outside source of funding or any competing interests.
Dr. Bartels disclosed receiving institutional grant funding through Pfizer.
SOURCE: Løgstrup BB et al. Ann Rheum Dis. 2020 May 29. doi: 10.1136/annrheumdis-2020-217154.
In patients with rheumatoid arthritis (RA), strategies to prevent cardiovascular events, such as treating hypertension, encouraging patients to stop smoking, and reinforcing statin therapy, may be especially important, regardless of whether they have a history of coronary artery disease because their risk for adverse cardiovascular outcomes is significantly greater than for patients who have neither RA nor coronary artery disease (CAD), a large population-based study from Denmark suggests.
“Among patients with RA, risk stratification by presence or absence of documented CAD may allow for screening and personalized treatment strategies,” wrote Brian B. Løgstrup, MD, PhD, DMSc, of Aarhus (Denmark) University Hospital, and his colleagues.
The study, published in Annals of the Rheumatic Diseases, analyzed 125,331 patients with and without CAD in the Western Denmark Heart Registry who had coronary angiography from 2003 through 2016. The cohort included 671 RA patients with no confirmed CAD and 1,061 RA patients who had CAD.
The study makes a significant contribution to the literature in reporting on the additive risk of RA and CAD, said Christie M. Bartels, MD, associate professor in the division of rheumatology at the University of Wisconsin, Madison. “Even among patients with both conditions [RA and CVD], they were less likely to get statin therapy,” she said, noting that the 82.6% of study patients with both CAD and RA were on statins vs. 86.5% of those with CAD alone, while the former had significantly higher rates of hypertension – 64.3% vs. 58.8%. “We’re doing a less effective job on secondary prevention,” she said. The anti-inflammatory properties of statins can also have an additive benefit in RA, she noted.
“This study shows that the rheumatologist can play a role in reinforcing the importance of primary and secondary cardiovascular disease prevention – meaning hypertension control, counseling patients to stop smoking and following up on statin therapy in RA,” Dr. Bartels added.
The study presents two novel findings, Dr. Løgstrup and colleagues noted:
- That RA confers a statistically significant, “but numerically marginally,” heightened risk of cardiovascular events other than stroke.
- Among patients with CAD, RA confers an increased risk of cardiac and all-cause death as well as MI and major adverse cardiovascular events (MACE).
“These finding indicate that RA may have a potential impact for precipitating cardiovascular events beyond CAD and, even more importantly, that RA seems to exacerbate the clinical risk of cardiovascular events in the presence of CAD,” Dr. Løgstrup and colleagues wrote.
The study found that patients with neither RA nor CAD had the lowest 10-year rates of MI (2.7%), ischemic stroke (2.9%), all-cause death (21.6%), cardiac death (2.3%), and MACE (7.3%).
By comparison, those with RA but no CAD had 10-year rates of 3.8% for MI, 5.5% for stroke, 35.6% for all-cause death, 3% for cardiac death, and 11.5% for MACE. Rates for those outcomes for people with CAD but no RA were 9.9% for MI, 4.6% for stroke, 33.3% for all-cause death, 7% for cardiac death, and 19.1% for MACE.
For patients with both RA and CAD, 10-year rates were 12.2% for MI, 4.4% for stroke, 49% for all-cause death, 10.9% for cardiac death, and 24.3% for MACE.
The researchers also performed a risk adjustment analysis based on potential confounding variables across the different groups, such as age, gender, comorbidities including diabetes and hypertension, active smoking status, and anticoagulant, antiplatelet, and statin therapy. The adjusted analysis revealed that patients with RA alone had a 63% greater risk of MI, 68% greater risk for stroke, 42% greater risk for all-cause death, 25% greater risk for cardiac death, and 60% greater risk for MACE than did people who had neither RA nor CAD.
For people with both RA and CAD, the adjusted risks were significantly higher when compared to people with neither: more than four times greater for MI and MACE, 55% greater for stroke, almost double for all-cause death, and 3.7 times greater for cardiac death. People with CAD but no RA also had higher adjusted risk rates compared to people with neither, but had variable rates when compared to people with RA but no CAD, and significantly lower adjusted rates compared to people with both.
The nature of CAD was also a factor, Dr. Løgstrup and colleagues noted. “We found more non-obstructive CAD but no increased incidence of one-vessel, two-vessel, and three-vessel disease in patients with RA than in patients without RA,” they wrote. That’s in line with other published studies (Semin Arthritis Rheum. 2010;40[3]:215–21 and J Rheumatol. 2007;34[5]:937–42), but counter to a study that found increased plaque burden and higher rates of multivessel disease among people with RA (Ann Rheum Dis. 2014;73:1797–804). Differences in methodology, vessel disease definitions, and study population may explain these deviations.
The study authors did not declare any outside source of funding or any competing interests.
Dr. Bartels disclosed receiving institutional grant funding through Pfizer.
SOURCE: Løgstrup BB et al. Ann Rheum Dis. 2020 May 29. doi: 10.1136/annrheumdis-2020-217154.
FROM ANNALS OF THE RHEUMATIC DISEASES
Ticagrelor gets FDA nod for DAPT in high-risk patients with CAD
The Food and Drug Administration has approved ticagrelor (Brilinta/Brilique, AstraZeneca) for use with aspirin to cut the risk for a first myocardial infarction or stroke in high-risk patients with coronary artery disease (CAD) but no history of MI or stroke, AstraZeneca announced today.
The new indication is based on the results of THEMIS (Effect of Ticagrelor on Health Outcomes in Diabetes Mellitus Patients Intervention Study), in which such patients with both CAD and type 2 diabetes on dual-antiplatelet therapy that included ticagrelor 60 mg twice-daily showed a 10% drop in risk for major adverse cardiac events, compared with aspirin alone over about 3 years. The absolute difference was 0.8% in the 42-country trial with more than 19,000 patients.
Patients falling under the new indication do not need to have diabetes, although THEMIS had entered patients with diabetes and CAD, the latter defined as a 50% or greater narrowing of a coronary artery or a history of coronary revascularization but without a history of MI or stroke.
The trial showed a significant reduction in the rate of the primary efficacy end point (P = .04), a composite of cardiovascular death, MI, and stroke. But the risk of TIMI (Thrombolysis in Myocardial Infarction) bleeding classification major bleeding was more than doubled in the ticagrelor group (P < .001) and the risk for intracranial hemorrhage went up 71% (P = .005). Net clinical benefit didn't differ significantly between the groups in an exploratory analysis.
The benefit of dual-antiplatelet therapy with ticagrelor for the primary efficacy end point was even more pronounced in a prespecified THEMIS subanalysis of more than 11,000 patients with a history of percutaneous coronary intervention. In this group, the risk for intracerebral hemorrhage didn't differ significantly between the groups, and the net clinical benefit favored ticagrelor by a significant 15%.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has approved ticagrelor (Brilinta/Brilique, AstraZeneca) for use with aspirin to cut the risk for a first myocardial infarction or stroke in high-risk patients with coronary artery disease (CAD) but no history of MI or stroke, AstraZeneca announced today.
The new indication is based on the results of THEMIS (Effect of Ticagrelor on Health Outcomes in Diabetes Mellitus Patients Intervention Study), in which such patients with both CAD and type 2 diabetes on dual-antiplatelet therapy that included ticagrelor 60 mg twice-daily showed a 10% drop in risk for major adverse cardiac events, compared with aspirin alone over about 3 years. The absolute difference was 0.8% in the 42-country trial with more than 19,000 patients.
Patients falling under the new indication do not need to have diabetes, although THEMIS had entered patients with diabetes and CAD, the latter defined as a 50% or greater narrowing of a coronary artery or a history of coronary revascularization but without a history of MI or stroke.
The trial showed a significant reduction in the rate of the primary efficacy end point (P = .04), a composite of cardiovascular death, MI, and stroke. But the risk of TIMI (Thrombolysis in Myocardial Infarction) bleeding classification major bleeding was more than doubled in the ticagrelor group (P < .001) and the risk for intracranial hemorrhage went up 71% (P = .005). Net clinical benefit didn't differ significantly between the groups in an exploratory analysis.
The benefit of dual-antiplatelet therapy with ticagrelor for the primary efficacy end point was even more pronounced in a prespecified THEMIS subanalysis of more than 11,000 patients with a history of percutaneous coronary intervention. In this group, the risk for intracerebral hemorrhage didn't differ significantly between the groups, and the net clinical benefit favored ticagrelor by a significant 15%.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has approved ticagrelor (Brilinta/Brilique, AstraZeneca) for use with aspirin to cut the risk for a first myocardial infarction or stroke in high-risk patients with coronary artery disease (CAD) but no history of MI or stroke, AstraZeneca announced today.
The new indication is based on the results of THEMIS (Effect of Ticagrelor on Health Outcomes in Diabetes Mellitus Patients Intervention Study), in which such patients with both CAD and type 2 diabetes on dual-antiplatelet therapy that included ticagrelor 60 mg twice-daily showed a 10% drop in risk for major adverse cardiac events, compared with aspirin alone over about 3 years. The absolute difference was 0.8% in the 42-country trial with more than 19,000 patients.
Patients falling under the new indication do not need to have diabetes, although THEMIS had entered patients with diabetes and CAD, the latter defined as a 50% or greater narrowing of a coronary artery or a history of coronary revascularization but without a history of MI or stroke.
The trial showed a significant reduction in the rate of the primary efficacy end point (P = .04), a composite of cardiovascular death, MI, and stroke. But the risk of TIMI (Thrombolysis in Myocardial Infarction) bleeding classification major bleeding was more than doubled in the ticagrelor group (P < .001) and the risk for intracranial hemorrhage went up 71% (P = .005). Net clinical benefit didn't differ significantly between the groups in an exploratory analysis.
The benefit of dual-antiplatelet therapy with ticagrelor for the primary efficacy end point was even more pronounced in a prespecified THEMIS subanalysis of more than 11,000 patients with a history of percutaneous coronary intervention. In this group, the risk for intracerebral hemorrhage didn't differ significantly between the groups, and the net clinical benefit favored ticagrelor by a significant 15%.
A version of this article originally appeared on Medscape.com.
Latest from ISCHEMIA: Worse outcomes in patients with intermediate left main disease on CCTA
Patients in the landmark ISCHEMIA trial with intermediate left main disease had a greater extent of coronary artery disease on invasive angiography, indicating greater atherosclerotic burden. They also had worse prognosis with a higher risk of cardiovascular events.
“Many times, we are looking at results as to whether patients have left main disease or not,” Sripal Bangalore, MD, said during the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions. “Here, we are showing that it’s not black and white; there are shades of gray. If a patient has intermediate left main disease, the prognosis is worse. That’s very important information we need to convey to our referrals also, because many times they may just look at the bottom line and say, ‘there is no left main disease.’ But here, we’re seeing that even having intermediate left main disease has significantly worse prognosis. We need to take that seriously.”
Prior studies show that patients with significant left main disease (LMD; defined as 50% or greater stenosis on coronary CT angiography [CCTA]) have a high risk of cardiovascular events and guidelines recommend revascularization to improve survival, said Dr. Bangalore, an interventional cardiologist at New York University Langone Health. However, the impact of intermediate LMD (defined as 25%-49% stenosis on CCTA) on outcomes is unclear.
Members of the ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) research group randomized 5,179 participants to an initial invasive or conservative strategy. The main results showed that immediate revascularization in patients with stable ischemic heart disease provided no reduction in cardiovascular endpoints through 4 years of follow-up, compared with initial optimal medical therapy alone.
‘Discordance’ revealed in imaging modalities
For the current analysis, named the ISCHEMIA Intermediate LM Substudy, those who underwent coronary CCTA comprise the LMD substudy cohort. The objective was to evaluate clinical and quality of life outcomes in patients with and without intermediate left main disease on coronary CT and to evaluate the impact of treatment strategy on those outcomes across subgroups.
At baseline, these patients were categorized into those with and without intermediate LMD as determined by a core lab. Patients with LMD of 50% or greater, those with prior coronary artery bypass graft surgery, and those with nonevaluable or missing data on LM stenosis were excluded.
Among the 3,913 ISCHEMIA participants who underwent CCTA, 3,699 satisfied the inclusion criteria. Of these patients, 962 (26%) had intermediate LMD and 2,737 (74%) did not.
The researchers observed no significant differences in baseline characteristics between patients with and without LMD. However, patients with intermediate LMD tended to be older, and a greater proportion had hypertension and diabetes. Stress test characteristics were also similar between patients with and without LMD. However, patients with intermediate LMD tended toward a greater severity of severe ischemia.
This was also true for anatomic disease on CCTA. A higher proportion of patients with intermediate LMD had triple-vessel disease (61%-62%, compared with 36%-40% along those without intermediate LMD). In addition, a higher proportion of patients with intermediate LMD had stenosis in the proximal left anterior artery descending (LAD) artery (65% vs. 39% among those without intermediate LMD).
On analysis limited to 1,846 patients who underwent invasive angiography treatment in the main ISCHEMIA trial, 7% of those who were categorized into the intermediate LMD group were found to have LMD disease of 50% or greater, compared with 1.4% of patients who were categorized as not having intermediate LMD. “This goes to show this discordance between the two modalities [CCTA and coronary angiography], and I think we have to be careful,” said Dr. Bangalore, who also directs NYU Langone’s Cardiac Catheterization Laboratory. “There may be patients with left main disease, even if the CCTA says it’s not at 25%-29% [stenosis].”
The researchers found that, among patients who underwent invasive angiography, a greater proportion of those who were categorized into the LMD group had proximal LAD disease (43% vs. 33% among those who were categorized into the nonintermediate LMD group), triple-vessel disease (47% vs. 35%), a greater extent of coronary artery disease as denoted by a higher SYNTAX score (21 vs. 15), and a higher proportion underwent coronary artery bypass graft surgery (32% vs. 18%).
Intermediate LMD linked to worse outcomes
After the researchers adjusted for baseline differences between the two groups in overall substudy cohort, they found that intermediate LMD severity was an independent predictor of the primary composite endpoint of cardiovascular death, MI, hospitalization for unstable angina, heart failure, and resuscitated cardiac arrest (hazard ratio, 1.31; P = .0123); cardiovascular death/MI/stroke (HR, 1.30; P = .0143); procedural primary MI (HR, 1.64; P = .0487); heart failure (HR, 2.06; P = .0239); and stroke (HR, 1.82, P = .0362).
“We then looked to see if there is a treatment difference, a treatment effect based on whether patients had intermediate LMD,” Dr. Bangalore said. “Most of the P values were not significant. The results are very consistent with what we saw in the main analysis: not a significant difference between invasive and conservative strategy. We do see some differences, though. An invasive strategy was associated with a significantly higher risk of procedural MI [2.9% vs. 1.5%], but a significantly lower risk of nonprocedural MI [–6.4% vs. –2%].”
Dr. Bangalore added that there was significant benefit of the invasive strategy in reducing angina and improving quality of life based on the Seattle Angina Questionnaire-7. “This result was durable up to 48 months of follow-up, whether the patient had intermediate left main disease or not. These results were dependent on baseline angina status. The benefit of invasive strategy was mainly in patients who had daily, weekly, and monthly angina, and no benefit in patients with no angina; there was no interaction based on intermediate left main status.”
Dr. Bangalore emphasized that the original ISCHEMIA trial excluded patients with severe left main disease by design. “But patients with intermediate left main disease in ISCHEMIA tended to have a greater extent of coronary artery disease, indicating greater atherosclerotic burden. I don’t think that’s any surprise. They had a worse prognosis with higher risk of cardiovascular events but similar quality of life, including angina-specific quality of life.”
The key clinical message, he said, is that patients with intermediate LMD face an increased risk of cardiovascular events. “I think we have to be aggressive in trying to reduce their risk with medical therapy, etc.,” he said. “If they are symptomatic, ISCHEMIA tells us that patients have two options. They can choose an invasive strategy, because clearly there is a benefit. You have a significant benefit at making you feel better and potentially reducing the risk of spontaneous MI over a period of time. Or, you can try medical therapy first. If you do see some left main disease, it’s showing the general burden of atherosclerosis disease in those patients. I think that’s the critical message, that we have to be very aggressive with these patients.”
A call for more imaging studies
An invited panelist, Timothy D. Henry, MD, said that the results of the ISCHEMIA substudy should stimulate further research. “With an intermediate lesion, clearly the interventional group did better, and it wasn’t symptom related,” said Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research and Education at the Christ Hospital in Cincinnati. “So even if you do medical therapy, you’re not going to really find it out. In my mind, this should stimulate us to do more imaging of the left main that are moderate lesions, and follow this up as an independent study. I think this is a really important finding.”
ISCHEMIA was supported by grants from the National Heart, Lung, and Blood Institute. Dr. Bangalore disclosed that he is a member of the advisory board and/or a board member for Meril, SMT, Pfizer, Amgen, Biotronik, and Abbott. He also is a consultant for Reata Pharmaceuticals.
SOURCE: Bangalore S et al. SCAI 2020, Abstract 11656.
Patients in the landmark ISCHEMIA trial with intermediate left main disease had a greater extent of coronary artery disease on invasive angiography, indicating greater atherosclerotic burden. They also had worse prognosis with a higher risk of cardiovascular events.
“Many times, we are looking at results as to whether patients have left main disease or not,” Sripal Bangalore, MD, said during the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions. “Here, we are showing that it’s not black and white; there are shades of gray. If a patient has intermediate left main disease, the prognosis is worse. That’s very important information we need to convey to our referrals also, because many times they may just look at the bottom line and say, ‘there is no left main disease.’ But here, we’re seeing that even having intermediate left main disease has significantly worse prognosis. We need to take that seriously.”
Prior studies show that patients with significant left main disease (LMD; defined as 50% or greater stenosis on coronary CT angiography [CCTA]) have a high risk of cardiovascular events and guidelines recommend revascularization to improve survival, said Dr. Bangalore, an interventional cardiologist at New York University Langone Health. However, the impact of intermediate LMD (defined as 25%-49% stenosis on CCTA) on outcomes is unclear.
Members of the ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) research group randomized 5,179 participants to an initial invasive or conservative strategy. The main results showed that immediate revascularization in patients with stable ischemic heart disease provided no reduction in cardiovascular endpoints through 4 years of follow-up, compared with initial optimal medical therapy alone.
‘Discordance’ revealed in imaging modalities
For the current analysis, named the ISCHEMIA Intermediate LM Substudy, those who underwent coronary CCTA comprise the LMD substudy cohort. The objective was to evaluate clinical and quality of life outcomes in patients with and without intermediate left main disease on coronary CT and to evaluate the impact of treatment strategy on those outcomes across subgroups.
At baseline, these patients were categorized into those with and without intermediate LMD as determined by a core lab. Patients with LMD of 50% or greater, those with prior coronary artery bypass graft surgery, and those with nonevaluable or missing data on LM stenosis were excluded.
Among the 3,913 ISCHEMIA participants who underwent CCTA, 3,699 satisfied the inclusion criteria. Of these patients, 962 (26%) had intermediate LMD and 2,737 (74%) did not.
The researchers observed no significant differences in baseline characteristics between patients with and without LMD. However, patients with intermediate LMD tended to be older, and a greater proportion had hypertension and diabetes. Stress test characteristics were also similar between patients with and without LMD. However, patients with intermediate LMD tended toward a greater severity of severe ischemia.
This was also true for anatomic disease on CCTA. A higher proportion of patients with intermediate LMD had triple-vessel disease (61%-62%, compared with 36%-40% along those without intermediate LMD). In addition, a higher proportion of patients with intermediate LMD had stenosis in the proximal left anterior artery descending (LAD) artery (65% vs. 39% among those without intermediate LMD).
On analysis limited to 1,846 patients who underwent invasive angiography treatment in the main ISCHEMIA trial, 7% of those who were categorized into the intermediate LMD group were found to have LMD disease of 50% or greater, compared with 1.4% of patients who were categorized as not having intermediate LMD. “This goes to show this discordance between the two modalities [CCTA and coronary angiography], and I think we have to be careful,” said Dr. Bangalore, who also directs NYU Langone’s Cardiac Catheterization Laboratory. “There may be patients with left main disease, even if the CCTA says it’s not at 25%-29% [stenosis].”
The researchers found that, among patients who underwent invasive angiography, a greater proportion of those who were categorized into the LMD group had proximal LAD disease (43% vs. 33% among those who were categorized into the nonintermediate LMD group), triple-vessel disease (47% vs. 35%), a greater extent of coronary artery disease as denoted by a higher SYNTAX score (21 vs. 15), and a higher proportion underwent coronary artery bypass graft surgery (32% vs. 18%).
Intermediate LMD linked to worse outcomes
After the researchers adjusted for baseline differences between the two groups in overall substudy cohort, they found that intermediate LMD severity was an independent predictor of the primary composite endpoint of cardiovascular death, MI, hospitalization for unstable angina, heart failure, and resuscitated cardiac arrest (hazard ratio, 1.31; P = .0123); cardiovascular death/MI/stroke (HR, 1.30; P = .0143); procedural primary MI (HR, 1.64; P = .0487); heart failure (HR, 2.06; P = .0239); and stroke (HR, 1.82, P = .0362).
“We then looked to see if there is a treatment difference, a treatment effect based on whether patients had intermediate LMD,” Dr. Bangalore said. “Most of the P values were not significant. The results are very consistent with what we saw in the main analysis: not a significant difference between invasive and conservative strategy. We do see some differences, though. An invasive strategy was associated with a significantly higher risk of procedural MI [2.9% vs. 1.5%], but a significantly lower risk of nonprocedural MI [–6.4% vs. –2%].”
Dr. Bangalore added that there was significant benefit of the invasive strategy in reducing angina and improving quality of life based on the Seattle Angina Questionnaire-7. “This result was durable up to 48 months of follow-up, whether the patient had intermediate left main disease or not. These results were dependent on baseline angina status. The benefit of invasive strategy was mainly in patients who had daily, weekly, and monthly angina, and no benefit in patients with no angina; there was no interaction based on intermediate left main status.”
Dr. Bangalore emphasized that the original ISCHEMIA trial excluded patients with severe left main disease by design. “But patients with intermediate left main disease in ISCHEMIA tended to have a greater extent of coronary artery disease, indicating greater atherosclerotic burden. I don’t think that’s any surprise. They had a worse prognosis with higher risk of cardiovascular events but similar quality of life, including angina-specific quality of life.”
The key clinical message, he said, is that patients with intermediate LMD face an increased risk of cardiovascular events. “I think we have to be aggressive in trying to reduce their risk with medical therapy, etc.,” he said. “If they are symptomatic, ISCHEMIA tells us that patients have two options. They can choose an invasive strategy, because clearly there is a benefit. You have a significant benefit at making you feel better and potentially reducing the risk of spontaneous MI over a period of time. Or, you can try medical therapy first. If you do see some left main disease, it’s showing the general burden of atherosclerosis disease in those patients. I think that’s the critical message, that we have to be very aggressive with these patients.”
A call for more imaging studies
An invited panelist, Timothy D. Henry, MD, said that the results of the ISCHEMIA substudy should stimulate further research. “With an intermediate lesion, clearly the interventional group did better, and it wasn’t symptom related,” said Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research and Education at the Christ Hospital in Cincinnati. “So even if you do medical therapy, you’re not going to really find it out. In my mind, this should stimulate us to do more imaging of the left main that are moderate lesions, and follow this up as an independent study. I think this is a really important finding.”
ISCHEMIA was supported by grants from the National Heart, Lung, and Blood Institute. Dr. Bangalore disclosed that he is a member of the advisory board and/or a board member for Meril, SMT, Pfizer, Amgen, Biotronik, and Abbott. He also is a consultant for Reata Pharmaceuticals.
SOURCE: Bangalore S et al. SCAI 2020, Abstract 11656.
Patients in the landmark ISCHEMIA trial with intermediate left main disease had a greater extent of coronary artery disease on invasive angiography, indicating greater atherosclerotic burden. They also had worse prognosis with a higher risk of cardiovascular events.
“Many times, we are looking at results as to whether patients have left main disease or not,” Sripal Bangalore, MD, said during the Society for Cardiovascular Angiography & Interventions virtual annual scientific sessions. “Here, we are showing that it’s not black and white; there are shades of gray. If a patient has intermediate left main disease, the prognosis is worse. That’s very important information we need to convey to our referrals also, because many times they may just look at the bottom line and say, ‘there is no left main disease.’ But here, we’re seeing that even having intermediate left main disease has significantly worse prognosis. We need to take that seriously.”
Prior studies show that patients with significant left main disease (LMD; defined as 50% or greater stenosis on coronary CT angiography [CCTA]) have a high risk of cardiovascular events and guidelines recommend revascularization to improve survival, said Dr. Bangalore, an interventional cardiologist at New York University Langone Health. However, the impact of intermediate LMD (defined as 25%-49% stenosis on CCTA) on outcomes is unclear.
Members of the ISCHEMIA (International Study of Comparative Health Effectiveness with Medical and Invasive Approaches) research group randomized 5,179 participants to an initial invasive or conservative strategy. The main results showed that immediate revascularization in patients with stable ischemic heart disease provided no reduction in cardiovascular endpoints through 4 years of follow-up, compared with initial optimal medical therapy alone.
‘Discordance’ revealed in imaging modalities
For the current analysis, named the ISCHEMIA Intermediate LM Substudy, those who underwent coronary CCTA comprise the LMD substudy cohort. The objective was to evaluate clinical and quality of life outcomes in patients with and without intermediate left main disease on coronary CT and to evaluate the impact of treatment strategy on those outcomes across subgroups.
At baseline, these patients were categorized into those with and without intermediate LMD as determined by a core lab. Patients with LMD of 50% or greater, those with prior coronary artery bypass graft surgery, and those with nonevaluable or missing data on LM stenosis were excluded.
Among the 3,913 ISCHEMIA participants who underwent CCTA, 3,699 satisfied the inclusion criteria. Of these patients, 962 (26%) had intermediate LMD and 2,737 (74%) did not.
The researchers observed no significant differences in baseline characteristics between patients with and without LMD. However, patients with intermediate LMD tended to be older, and a greater proportion had hypertension and diabetes. Stress test characteristics were also similar between patients with and without LMD. However, patients with intermediate LMD tended toward a greater severity of severe ischemia.
This was also true for anatomic disease on CCTA. A higher proportion of patients with intermediate LMD had triple-vessel disease (61%-62%, compared with 36%-40% along those without intermediate LMD). In addition, a higher proportion of patients with intermediate LMD had stenosis in the proximal left anterior artery descending (LAD) artery (65% vs. 39% among those without intermediate LMD).
On analysis limited to 1,846 patients who underwent invasive angiography treatment in the main ISCHEMIA trial, 7% of those who were categorized into the intermediate LMD group were found to have LMD disease of 50% or greater, compared with 1.4% of patients who were categorized as not having intermediate LMD. “This goes to show this discordance between the two modalities [CCTA and coronary angiography], and I think we have to be careful,” said Dr. Bangalore, who also directs NYU Langone’s Cardiac Catheterization Laboratory. “There may be patients with left main disease, even if the CCTA says it’s not at 25%-29% [stenosis].”
The researchers found that, among patients who underwent invasive angiography, a greater proportion of those who were categorized into the LMD group had proximal LAD disease (43% vs. 33% among those who were categorized into the nonintermediate LMD group), triple-vessel disease (47% vs. 35%), a greater extent of coronary artery disease as denoted by a higher SYNTAX score (21 vs. 15), and a higher proportion underwent coronary artery bypass graft surgery (32% vs. 18%).
Intermediate LMD linked to worse outcomes
After the researchers adjusted for baseline differences between the two groups in overall substudy cohort, they found that intermediate LMD severity was an independent predictor of the primary composite endpoint of cardiovascular death, MI, hospitalization for unstable angina, heart failure, and resuscitated cardiac arrest (hazard ratio, 1.31; P = .0123); cardiovascular death/MI/stroke (HR, 1.30; P = .0143); procedural primary MI (HR, 1.64; P = .0487); heart failure (HR, 2.06; P = .0239); and stroke (HR, 1.82, P = .0362).
“We then looked to see if there is a treatment difference, a treatment effect based on whether patients had intermediate LMD,” Dr. Bangalore said. “Most of the P values were not significant. The results are very consistent with what we saw in the main analysis: not a significant difference between invasive and conservative strategy. We do see some differences, though. An invasive strategy was associated with a significantly higher risk of procedural MI [2.9% vs. 1.5%], but a significantly lower risk of nonprocedural MI [–6.4% vs. –2%].”
Dr. Bangalore added that there was significant benefit of the invasive strategy in reducing angina and improving quality of life based on the Seattle Angina Questionnaire-7. “This result was durable up to 48 months of follow-up, whether the patient had intermediate left main disease or not. These results were dependent on baseline angina status. The benefit of invasive strategy was mainly in patients who had daily, weekly, and monthly angina, and no benefit in patients with no angina; there was no interaction based on intermediate left main status.”
Dr. Bangalore emphasized that the original ISCHEMIA trial excluded patients with severe left main disease by design. “But patients with intermediate left main disease in ISCHEMIA tended to have a greater extent of coronary artery disease, indicating greater atherosclerotic burden. I don’t think that’s any surprise. They had a worse prognosis with higher risk of cardiovascular events but similar quality of life, including angina-specific quality of life.”
The key clinical message, he said, is that patients with intermediate LMD face an increased risk of cardiovascular events. “I think we have to be aggressive in trying to reduce their risk with medical therapy, etc.,” he said. “If they are symptomatic, ISCHEMIA tells us that patients have two options. They can choose an invasive strategy, because clearly there is a benefit. You have a significant benefit at making you feel better and potentially reducing the risk of spontaneous MI over a period of time. Or, you can try medical therapy first. If you do see some left main disease, it’s showing the general burden of atherosclerosis disease in those patients. I think that’s the critical message, that we have to be very aggressive with these patients.”
A call for more imaging studies
An invited panelist, Timothy D. Henry, MD, said that the results of the ISCHEMIA substudy should stimulate further research. “With an intermediate lesion, clearly the interventional group did better, and it wasn’t symptom related,” said Dr. Henry, medical director of the Carl and Edyth Lindner Center for Research and Education at the Christ Hospital in Cincinnati. “So even if you do medical therapy, you’re not going to really find it out. In my mind, this should stimulate us to do more imaging of the left main that are moderate lesions, and follow this up as an independent study. I think this is a really important finding.”
ISCHEMIA was supported by grants from the National Heart, Lung, and Blood Institute. Dr. Bangalore disclosed that he is a member of the advisory board and/or a board member for Meril, SMT, Pfizer, Amgen, Biotronik, and Abbott. He also is a consultant for Reata Pharmaceuticals.
SOURCE: Bangalore S et al. SCAI 2020, Abstract 11656.
FROM SCAI 2020














