User login
NIH Tackles Chronic Low Back Pain
Standardized research methods are needed to advance efforts toward reducing the costs and high burden of chronic low back pain, according to a multidisciplinary NIH Task Force report published online ahead of print May 30 in Spine.
The article introduces a set of proposed research standards to help in comparing the results of chronic low back pain studies. The Task Force co-chairs were Drs. Richard A. Deyo of Oregon Health and Science University, and Samuel F. Dworkin of the University of Washington in Seattle.
“Researchers use varied inclusion criteria, definitions, baseline assessments, and outcome measures, which impede comparisons and consensus,” the Task Force wrote.
To address this issue, the Task Force followed a structured approach to developing a set of standards for chronic low back pain research. Overriding issues included defining the problem of chronic low back pain, identifying the minimum dataset that should be collected in chronic low back pain research, assessing its impact on patients’ lives, and defining the best outcomes to evaluate treatment effectiveness.
The Task Force recommends that chronic low back pain be defined as back pain that lasts at least 3 months and causing pain on at least half of days over the past 6 months. Their recommended definition does not include ratings of pain severity.
In terms of impact, the Task Force recommends focusing on how back pain affects patients’ lives. They recommend a 9-item chronic low back pain “Impact Score” that incorporates ratings of pain intensity, interference with normal activities, and functional ability.
A key point for the Task Force was to define a minimum set of data to be gathered in any study of chronic low back pain. The recommended dataset included legal or workers compensation issues, previous treatments, and important contributing factors (eg, smoking, obesity, substance abuse, and widespread pain).
For outcomes, the Task Force concluded there was no set definition of what degree of improvement should be considered “clinically important.” Likewise, there was no consensus on the use of combined outcome measures, time frame for improvement, or adverse effects.
Finally, testing and developing new combined outcome measures was identified as an important aspect of future research, more specifically, other included approaches to predicting treatment results and studies to evaluate and improve the minimum dataset.
Overall, the recommendations stress the importance of getting a fuller picture of the patient’s medical history, even more so than the physical examination. On the other hand, the Task Force specified no standard laboratory or imaging tests, citing the lack of association with patient functioning or symptoms. Evaluations of depression, sleep disturbance, physical functioning, and catastrophic thinking were rated as important for all groups of patients with chronic low back pain.
The Task Force hopes that their recommended standards reflect the “complex, intertwined factors” affecting the development and clinical course of chronic low back pain. The NIH Pain Consortium has approved the recommendations and advises that investigators should incorporate them into NIH grant proposals. “As adopted by the NIH, these recommendations have the potential to standardize methods for identifying chronic low back pain research cases, describe research subjects, and compare published reports.” The Task Force added that recommendations should be subject to periodic validation and refinement in years ahead.
Suggested Reading
Deyo RA, Dworkin SF, Amtmann D, et al. NIH task force proposes standards for research on chronic low back pain. Spine. 2014 May 30 [Epub ahead of print].
Standardized research methods are needed to advance efforts toward reducing the costs and high burden of chronic low back pain, according to a multidisciplinary NIH Task Force report published online ahead of print May 30 in Spine.
The article introduces a set of proposed research standards to help in comparing the results of chronic low back pain studies. The Task Force co-chairs were Drs. Richard A. Deyo of Oregon Health and Science University, and Samuel F. Dworkin of the University of Washington in Seattle.
“Researchers use varied inclusion criteria, definitions, baseline assessments, and outcome measures, which impede comparisons and consensus,” the Task Force wrote.
To address this issue, the Task Force followed a structured approach to developing a set of standards for chronic low back pain research. Overriding issues included defining the problem of chronic low back pain, identifying the minimum dataset that should be collected in chronic low back pain research, assessing its impact on patients’ lives, and defining the best outcomes to evaluate treatment effectiveness.
The Task Force recommends that chronic low back pain be defined as back pain that lasts at least 3 months and causing pain on at least half of days over the past 6 months. Their recommended definition does not include ratings of pain severity.
In terms of impact, the Task Force recommends focusing on how back pain affects patients’ lives. They recommend a 9-item chronic low back pain “Impact Score” that incorporates ratings of pain intensity, interference with normal activities, and functional ability.
A key point for the Task Force was to define a minimum set of data to be gathered in any study of chronic low back pain. The recommended dataset included legal or workers compensation issues, previous treatments, and important contributing factors (eg, smoking, obesity, substance abuse, and widespread pain).
For outcomes, the Task Force concluded there was no set definition of what degree of improvement should be considered “clinically important.” Likewise, there was no consensus on the use of combined outcome measures, time frame for improvement, or adverse effects.
Finally, testing and developing new combined outcome measures was identified as an important aspect of future research, more specifically, other included approaches to predicting treatment results and studies to evaluate and improve the minimum dataset.
Overall, the recommendations stress the importance of getting a fuller picture of the patient’s medical history, even more so than the physical examination. On the other hand, the Task Force specified no standard laboratory or imaging tests, citing the lack of association with patient functioning or symptoms. Evaluations of depression, sleep disturbance, physical functioning, and catastrophic thinking were rated as important for all groups of patients with chronic low back pain.
The Task Force hopes that their recommended standards reflect the “complex, intertwined factors” affecting the development and clinical course of chronic low back pain. The NIH Pain Consortium has approved the recommendations and advises that investigators should incorporate them into NIH grant proposals. “As adopted by the NIH, these recommendations have the potential to standardize methods for identifying chronic low back pain research cases, describe research subjects, and compare published reports.” The Task Force added that recommendations should be subject to periodic validation and refinement in years ahead.
Standardized research methods are needed to advance efforts toward reducing the costs and high burden of chronic low back pain, according to a multidisciplinary NIH Task Force report published online ahead of print May 30 in Spine.
The article introduces a set of proposed research standards to help in comparing the results of chronic low back pain studies. The Task Force co-chairs were Drs. Richard A. Deyo of Oregon Health and Science University, and Samuel F. Dworkin of the University of Washington in Seattle.
“Researchers use varied inclusion criteria, definitions, baseline assessments, and outcome measures, which impede comparisons and consensus,” the Task Force wrote.
To address this issue, the Task Force followed a structured approach to developing a set of standards for chronic low back pain research. Overriding issues included defining the problem of chronic low back pain, identifying the minimum dataset that should be collected in chronic low back pain research, assessing its impact on patients’ lives, and defining the best outcomes to evaluate treatment effectiveness.
The Task Force recommends that chronic low back pain be defined as back pain that lasts at least 3 months and causing pain on at least half of days over the past 6 months. Their recommended definition does not include ratings of pain severity.
In terms of impact, the Task Force recommends focusing on how back pain affects patients’ lives. They recommend a 9-item chronic low back pain “Impact Score” that incorporates ratings of pain intensity, interference with normal activities, and functional ability.
A key point for the Task Force was to define a minimum set of data to be gathered in any study of chronic low back pain. The recommended dataset included legal or workers compensation issues, previous treatments, and important contributing factors (eg, smoking, obesity, substance abuse, and widespread pain).
For outcomes, the Task Force concluded there was no set definition of what degree of improvement should be considered “clinically important.” Likewise, there was no consensus on the use of combined outcome measures, time frame for improvement, or adverse effects.
Finally, testing and developing new combined outcome measures was identified as an important aspect of future research, more specifically, other included approaches to predicting treatment results and studies to evaluate and improve the minimum dataset.
Overall, the recommendations stress the importance of getting a fuller picture of the patient’s medical history, even more so than the physical examination. On the other hand, the Task Force specified no standard laboratory or imaging tests, citing the lack of association with patient functioning or symptoms. Evaluations of depression, sleep disturbance, physical functioning, and catastrophic thinking were rated as important for all groups of patients with chronic low back pain.
The Task Force hopes that their recommended standards reflect the “complex, intertwined factors” affecting the development and clinical course of chronic low back pain. The NIH Pain Consortium has approved the recommendations and advises that investigators should incorporate them into NIH grant proposals. “As adopted by the NIH, these recommendations have the potential to standardize methods for identifying chronic low back pain research cases, describe research subjects, and compare published reports.” The Task Force added that recommendations should be subject to periodic validation and refinement in years ahead.
Suggested Reading
Deyo RA, Dworkin SF, Amtmann D, et al. NIH task force proposes standards for research on chronic low back pain. Spine. 2014 May 30 [Epub ahead of print].
Suggested Reading
Deyo RA, Dworkin SF, Amtmann D, et al. NIH task force proposes standards for research on chronic low back pain. Spine. 2014 May 30 [Epub ahead of print].
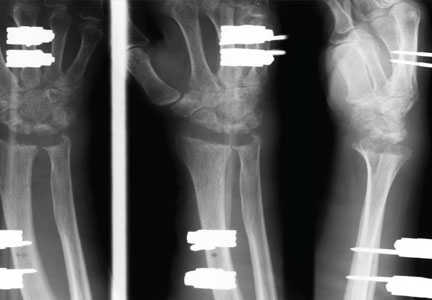
Bionic Arm Still in Development Stage
Within the past 13 years, roughly 2000 veterans who have returned from Afghanistan and Iraq have sustained injuries that required amputations. Of these injured veterans, 14% required upper extremity amputations. An article published in the June issue of the Journal of the American Academy of Orthopaedic Surgeons reviewed recent advancements in upper extremity bionics. Also reviewed were the challenges that linger in creating a prosthesis that meets or surpasses the abilities of the human hand and arm.
During the next 50 years, “I truly believe we will be able to make artificial arms that function better than many injured arms that doctors are saving today,” said article author Dr. Douglas T. Hutchinson, Associate Professor of Orthopedics at the University of Utah Medical School and Chief of Hand Surgery at Primary Children’s Medical Center, the Veterans Affairs Medical Center, and Shriners Intermountain Hospital.
Created more than 50 years ago, the myoelectric prosthesis continues to be the most commonly used upper extremity prostheses. This prosthesis allows residual muscles to act as natural batteries to create transcutaneous signals, to control the movements of the prosthetic hand and arm. However, the muscles used most often are the triceps and biceps, which do not inherently translate to the opening and closing of the hand. Another drawback is that sometimes the socket interface used to attach the prosthesis may interfere with the function of the residual joint, such as the elbow. Myoelectric prosthetics also do not look natural and are heavy, hot, and uncomfortable, and are not waterproof.
The current federal budget for prostheses research is $2.5 billion. The US Department of Defense Advanced Research Project (DARPA) already has invested more than $150 million for their Revolutionizing Prosthetics Program. The later program, which seeks to create an upper extremity prosthesis that can function as a normal hand and arm does, but with full sensory and motor functions.
In order for these prosthetic devices to be used effectively in a broad range for patients, adjustments still need to be made. For example, many have short-life batteries, along with being weighty and uncomfortable. Particularly challenging is the problem of accurately and efficiently sending brain signals through the peripheral nerves and muscles of the hands and arm, a feat that may warrant the creation and use of a reliable wireless device or direct wiring through an osseous-integrated implant. Current infection rates (nearly 45%) with osseous-integrated devices at the prosthesis-skin interface also pose an issue.
Suggested Reading
Hutchinson DT. The quest for the bionic arm. J Am Acad Orthop Surg. 2014;22(6):346-351.
Within the past 13 years, roughly 2000 veterans who have returned from Afghanistan and Iraq have sustained injuries that required amputations. Of these injured veterans, 14% required upper extremity amputations. An article published in the June issue of the Journal of the American Academy of Orthopaedic Surgeons reviewed recent advancements in upper extremity bionics. Also reviewed were the challenges that linger in creating a prosthesis that meets or surpasses the abilities of the human hand and arm.
During the next 50 years, “I truly believe we will be able to make artificial arms that function better than many injured arms that doctors are saving today,” said article author Dr. Douglas T. Hutchinson, Associate Professor of Orthopedics at the University of Utah Medical School and Chief of Hand Surgery at Primary Children’s Medical Center, the Veterans Affairs Medical Center, and Shriners Intermountain Hospital.
Created more than 50 years ago, the myoelectric prosthesis continues to be the most commonly used upper extremity prostheses. This prosthesis allows residual muscles to act as natural batteries to create transcutaneous signals, to control the movements of the prosthetic hand and arm. However, the muscles used most often are the triceps and biceps, which do not inherently translate to the opening and closing of the hand. Another drawback is that sometimes the socket interface used to attach the prosthesis may interfere with the function of the residual joint, such as the elbow. Myoelectric prosthetics also do not look natural and are heavy, hot, and uncomfortable, and are not waterproof.
The current federal budget for prostheses research is $2.5 billion. The US Department of Defense Advanced Research Project (DARPA) already has invested more than $150 million for their Revolutionizing Prosthetics Program. The later program, which seeks to create an upper extremity prosthesis that can function as a normal hand and arm does, but with full sensory and motor functions.
In order for these prosthetic devices to be used effectively in a broad range for patients, adjustments still need to be made. For example, many have short-life batteries, along with being weighty and uncomfortable. Particularly challenging is the problem of accurately and efficiently sending brain signals through the peripheral nerves and muscles of the hands and arm, a feat that may warrant the creation and use of a reliable wireless device or direct wiring through an osseous-integrated implant. Current infection rates (nearly 45%) with osseous-integrated devices at the prosthesis-skin interface also pose an issue.
Within the past 13 years, roughly 2000 veterans who have returned from Afghanistan and Iraq have sustained injuries that required amputations. Of these injured veterans, 14% required upper extremity amputations. An article published in the June issue of the Journal of the American Academy of Orthopaedic Surgeons reviewed recent advancements in upper extremity bionics. Also reviewed were the challenges that linger in creating a prosthesis that meets or surpasses the abilities of the human hand and arm.
During the next 50 years, “I truly believe we will be able to make artificial arms that function better than many injured arms that doctors are saving today,” said article author Dr. Douglas T. Hutchinson, Associate Professor of Orthopedics at the University of Utah Medical School and Chief of Hand Surgery at Primary Children’s Medical Center, the Veterans Affairs Medical Center, and Shriners Intermountain Hospital.
Created more than 50 years ago, the myoelectric prosthesis continues to be the most commonly used upper extremity prostheses. This prosthesis allows residual muscles to act as natural batteries to create transcutaneous signals, to control the movements of the prosthetic hand and arm. However, the muscles used most often are the triceps and biceps, which do not inherently translate to the opening and closing of the hand. Another drawback is that sometimes the socket interface used to attach the prosthesis may interfere with the function of the residual joint, such as the elbow. Myoelectric prosthetics also do not look natural and are heavy, hot, and uncomfortable, and are not waterproof.
The current federal budget for prostheses research is $2.5 billion. The US Department of Defense Advanced Research Project (DARPA) already has invested more than $150 million for their Revolutionizing Prosthetics Program. The later program, which seeks to create an upper extremity prosthesis that can function as a normal hand and arm does, but with full sensory and motor functions.
In order for these prosthetic devices to be used effectively in a broad range for patients, adjustments still need to be made. For example, many have short-life batteries, along with being weighty and uncomfortable. Particularly challenging is the problem of accurately and efficiently sending brain signals through the peripheral nerves and muscles of the hands and arm, a feat that may warrant the creation and use of a reliable wireless device or direct wiring through an osseous-integrated implant. Current infection rates (nearly 45%) with osseous-integrated devices at the prosthesis-skin interface also pose an issue.
Suggested Reading
Hutchinson DT. The quest for the bionic arm. J Am Acad Orthop Surg. 2014;22(6):346-351.
Suggested Reading
Hutchinson DT. The quest for the bionic arm. J Am Acad Orthop Surg. 2014;22(6):346-351.