User login
Potential Utility of Liposome Bupivacaine in Orthopedic Surgery
Approximately 5.5 million patients undergo orthopedic surgery in the United States each year, and more than 1 million of the procedures are total knee arthroplasty (TKA) or total hip arthroplasty.1 From its 2010 level, demand for joint arthroplasty is expected to double by 2020 and quadruple by 2030.2
About half the patients who have major joint arthroplasty experience severe postsurgical pain.3 Because postsurgical pain may persist for days or weeks, and inadequate treatment is associated with negative outcomes, achieving effective postsurgical analgesia is an important consideration.4-7 Complications of inadequate postsurgical pain management include thromboembolic or pulmonary complications, development of chronic pain, and decrements in health-related quality of life.4,8
In patients who have orthopedic surgery, the inability to adequately control postsurgical pain has been associated with increased hospital length of stay (LOS), delayed time to ambulation, and reduced capacity for exercise.9-12 A recent study involving 4709 patients who had hip or knee arthroplasty found that postsurgical pain relief was the second most highly correlated factor with respect to overall patient satisfaction (how well surgery met patient expectations was the most highly correlated factor),13 suggesting that postsurgical analgesia should be a focus of surgical practice.
A prolonged-release liposomal formulation of the local anesthetic bupivacaine is now available. Bupivacaine liposome injectable suspension (Exparel; Pacira Pharmaceuticals, Inc., Parsippany, New Jersey) is indicated for administration into the surgical site to produce postsurgical analgesia.14 In this article, we review evidence from clinical studies regarding the potential contribution of liposome bupivacaine to improving postsurgical pain management when used as part of a multimodal analgesic regimen in patients undergoing orthopedic surgery.
Postsurgical Pain Management in Orthopedic Surgery
Frequently Used Modalities
Analgesic modalities commonly used for perioperative pain management include central (eg, epidural),4,10,15,16 central regional (eg, neuraxial),4 peripheral regional (eg, peripheral nerve blocks, local/regional surgical site infiltration, intra-articular administration),4,10,15,17-25 and intravenous (IV) patient-controlled analgesia.4,10,25 These pharmacologic interventions may be augmented by nonpharmacologic modalities (eg, transcutaneous electrical nerve stimulation).26
Pharmacologic treatment options for perioperative pain management include opioids, local anesthetics, clonidine, ketamine, nonsteroidal anti-inflammatory drugs, acetaminophen, and calcium-channel blockers.4,26-28 In TKA, “drug cocktails” (eg, combinations of ropivacaine, ketorolac, epinephrine, and clonidine) for regional or intra-articular injection can also provide effective immediate postsurgical analgesia.25 Although opioids are the most commonly used analgesics for management of orthopedic perioperative pain,25 their use is often associated with adverse effects (AEs), including constipation or ileus, nausea, sedation, dizziness, pruritus, urinary retention, and respiratory depression.6
Multimodal Analgesic Regimens for Postsurgical Pain Management
Current American Society of Anesthesiologists guidelines endorse use of multimodal analgesia, whenever possible, to provide effective management of acute perioperative pain.4 Multimodal analgesia involves applying 2 or more agents with different mechanisms of action to achieve a synergistic effect, which allows each agent to be reduced in dose4,28 and thereby may limit the risk and severity of dose-related AEs.4,25,28
Multimodal analgesia aims to reduce the risk for opioid-related AEs (ORAEs) and the impact of opioids on postsurgical milestones (eg, ambulation, discharge) and may reduce opioid consumption, with attendant reductions in ORAE risk.29,30 Health economics studies have shown that postsurgical ORAEs are associated with increased hospital costs and LOS.6 In a study using a national hospital database, development of an ORAE (vs no ORAE) in postsurgical patients was associated with mean increases of about $4700 in hospital costs and 3.3 days in LOS.7 Reducing postsurgical opioid use may also help reduce the risk for opioid abuse, addiction, and diversion.31-33
One approach to reducing opioid use involves continuous or intermittent administration of local anesthetics by elastomeric pumps to extend duration of postsurgical analgesia.34-36 However, use of elastomeric pumps has been associated with risk for AEs, including tissue necrosis, sloughing, wound infection, and chondrolysis.37-40 In addition, AEs related to “dose dumping” (accidental delivery of excessive doses) have been reported.40-44 Key issues that may negatively affect rehabilitation after orthopedic surgery include consistency and accuracy of analgesic delivery and the potential for motor block–induced muscle weakness, which may lead to falls and constrain ambulation.45-47
Liposome Bupivacaine
Description
Drug Delivery Technology. Liposome bupivacaine incorporates DepoFoam drug delivery technology (Pacira Pharmaceuticals, Inc.) to facilitate prolonged release of bupivacaine. This technology is based on creation of multivesicular liposome particles (diameter, 10-30 µm) with multiple aqueous chambers.30,48 After administration into the surgical site, bupivacaine diffuses from chambers in the liposomal particles over time, providing analgesia and reduced opioid requirements for up to 72 hours.29,30
Indication, Mechanism of Action, Pharmacokinetics, and Dose/Administration. Liposome bupivacaine is indicated for single-dose administration into the surgical site to produce postsurgical analgesia in patients at least 18 years old.14 Like other local anesthetics, liposome bupivacaine is thought to exert its pharmacologic effects by interacting with voltage-gated Na+ channels on neural membranes to raise the threshold for electrical excitability, to slow nerve impulse propagation, and to reduce the rate of rise of the action potential.14,49
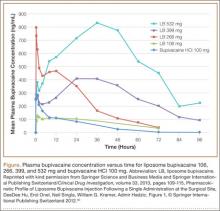
Liposome bupivacaine has dose-proportional pharmacokinetics.50 Presence of a small amount of extra-liposomal bupivacaine in the formulation leads to a bimodal pharmacokinetic profile, with an initial peak serum concentration about 1 hour after administration, followed by a second peak within 12 to 36 hours (Figure).50
Maximum amount of liposome bupivacaine approved for single administration is 266 mg (packaged as 20 mL of a 1.3% solution). However, product labeling includes safety data associated with doses of 532 mg or less.14 The appropriate volume to be used should be based on the amount required to cover the surgical area. Liposome bupivacaine may be expanded with preservative-free normal (0.9%) sterile saline to a total volume of 300 mL: 20 mL liposome bupivacaine plus 280 mL or less diluent, with final concentration of 0.89 mg/mL (1:14 by volume).14
A 25-gauge or larger bore needle should be used to slowly inject liposome bupivacaine into soft tissues of the surgical site, with frequent aspiration to check for blood to minimize risk for intravascular injection.14 Total volume used and fraction injected in specific regions of the surgical site depend on the procedure. For example, a TKA study used 266 mg diluted to a total volume of 60 mL, with 8 mL infiltrated to the area around the medial capsule, 8 mL around the lateral capsule, 12 mL around the posterior capsule, 8 mL around the peripatellar area, 12 mL into the capsulotomy incision, and 12 mL into the subcutaneous tissue on each side of the incision.51
Efficacy
Multiple Surgical Settings. The efficacy of liposome bupivacaine, either alone or as a component of a multimodal analgesic regimen, has been evaluated in a series of 10 phase 2 and 3 studies (8 active-controlled, 2 placebo-controlled) involving 823 patients undergoing TKA, bunionectomy, hemorrhoidectomy, inguinal hernia repair, or mammoplasty.52 Patients received a single liposome bupivacaine dose ranging from 66 to 532 mg.52
Combined analyses of efficacy data from these studies found that liposome bupivacaine–based multimodal analgesic regimens produced postsurgical analgesia for up to 72 hours, increased time to first use of opioid rescue medication after surgery, and reduced total amount of postsurgical opioid consumption versus placebo.52
Compared with standard of care, liposome bupivacaine has been shown to provide effective analgesia in open-label studies in patients undergoing open colectomy,53 laparoscopic colectomy,54 and ileostomy reversal,55,56 as reflected in assessments of postsurgical opioid consumption, LOS, and hospital costs. It has also been studied when administered by infiltration into the transversus abdominis plane (TAP) in patients having laparoscopic prostatectomy and open abdominal hernia repair.57,58
Orthopedic Surgery. In a phase 2 randomized, double-blind, dose-ranging study, TKA patients (N = 138) received bupivacaine HCl 150 mg or liposome bupivacaine 133, 266, 399, or 532 mg administered by local infiltration into the capsulotomy incision and on either side of the incision before wound closure.51 Postsurgical rescue analgesia was available to all patients. Cumulative pain intensity scores with activity (primary efficacy measure) were not statistically different between liposome bupivacaine groups and the bupivacaine HCl group through postoperative day 4. Mean scores in the liposome bupivacaine 266-, 399-, and 532-mg groups were numerically lower than for those treated with bupivacaine HCl on postoperative days 2 to 5, with all doses of liposome bupivacaine having a statistically significant lower pain score at rest on day 5. There were no statistically significant differences across treatment groups with respect to total amount of postsurgical opioids used.
In a phase 3 randomized, double-blind study of TKA patients (N = 245), liposome bupivacaine 532 mg administered into the surgical site was compared with bupivacaine HCl 200 mg for postsurgical analgesia.52 Rescue analgesia was available to all patients. No statistically significant between-group differences were found with respect to postsurgical cumulative pain scores through 72 hours (primary efficacy endpoint).
In a single-center retrospective TKA study, postsurgical outcomes in a patient cohort that received intraoperative periarticular infiltration with liposome bupivacaine 266 mg (n = 65) were compared with a cohort that received infiltration with a combination of ropivacaine 400 mg, morphine 5 mg, and epinephrine 0.4 mg (n = 85).59 Patient-reported postsurgical pain scores were similar in the 2 treatment groups during the first 24 hours after surgery and at discharge. Mean (SD) pain scores during hospitalization after the first 24 hours until discharge were significantly (P = .04) higher in the liposome bupivacaine group, 4.9 (1.4), than in the periarticular group, 4.4 (1.6). There was no significant difference between the 2 treatment groups in postsurgical opioid use. The study demonstrated no advantage to using liposome bupivacaine injections with respect to pain relief, but it was a retrospective review in which pain scores were obtained from electronic medical records. It is essential that liposome bupivacaine be compared with intra-articular injections in well-designed randomized trials.
Another single-center, matched-cohort TKA study (N = 200) compared a liposome bupivacaine regimen with femoral nerve block.60 Compared with patients who received femoral nerve block, patients who received liposome bupivacaine reported lower pain intensity scores after surgery and had shorter LOS, reduced costs, and improved knee flexion at follow-up.60
Results from 2 other studies were presented at the 2014 meeting of the American Academy of Orthopaedic Surgeons (AAOS). One was a single-center, matched-cohort TKA study (N = 72) comparing infiltration of a single dose of liposome bupivacaine into the surgical site with continuous femoral nerve block.61 The 2 treatment groups had similar mean postsurgical pain intensity scores on a 0-to-10 visual analog scale, 1.8 for liposome bupivacaine and 2.3 for continuous nerve block (P = NS), but total amount of postsurgical opioids (hydrocodone-equivalent milligrams) was significantly (P < .0001) less in the liposome bupivacaine group (82 vs 177 mg).
The other study presented at the AAOS meeting was a larger, prospective case–control study comparing outcomes between 1000 patients who had total joint arthroplasty (TJA) with liposome bupivacaine and 1000 control patients who had TJA without liposome bupivacaine.62 For the control and liposome bupivacaine cohorts, respectively, mean postsurgical pain intensity scores were 2.41 and 1.98 (P < .0001), mean LOS was 2.83 days and 2.66 days (P < .02), and incidence of falls was 1.0% and 0.2% (P = .02). Average per-patient costs were $1246 lower in the liposome bupivacaine cohort.
A pivotal phase 3 placebo-controlled study compared liposome bupivacaine 106 mg with placebo in patients undergoing bunionectomy (N = 193).5 Rescue medication was available to all patients. Cumulative pain scores were significantly (P = .0005) lower in the liposome bupivacaine group (125) than in the placebo group (146) through 24 hours after surgery (primary efficacy measure) and significantly (P = .0229) lower (197 vs 220) through 36 hours. Median time to first use of rescue opioids was delayed in favor of the liposome bupivacaine group (7.2 vs 4.3 hours; P < .0001). Mean total number of opioid tablets used within 24 hours after surgery was also significantly lower (3.8 vs 4.7; P = .008), and a larger percentage of patients in the liposome bupivacaine group avoided opioid use altogether through 24 hours (7% vs 1%; P = .04).
Efficacy data for liposome bupivacaine appear promising for relief of pain after joint arthroplasty and other orthopedic procedures but have their limitations. First, no randomized trials have compared liposome bupivacaine with locally injected pain medications (intra-articular injections in TKA or hip arthroplasty). As these injections are quite common now, such analyses are essential. Second, cost-effectiveness studies are needed for orthopedic procedures. Third, most of the published studies were sponsored by the manufacturer of liposome bupivacaine—a situation that raises questions about potential bias. Non-industry-sponsored randomized trials assessing efficacy, safety, and cost-effectiveness are needed.
Safety
Local anesthetics, including liposome bupivacaine, have the potential for central nervous system (CNS) or cardiac toxicity resulting from excessive systemic absorption or inadvertent IV administration.63 However, reported serious CNS or cardiac-related AEs are rare.63,64
AE Profile. Safety data from 10 phase 2 and 3 studies involving 823 patients who received liposome bupivacaine were evaluated.65 Of these patients, 545 received a dose of 266 mg or less (maximum dose approved by the US Food and Drug Administration [FDA]). Liposome bupivacaine was generally well tolerated. Reported AE incidence was 62% (liposome bupivacaine), 75% (bupivacaine HCl), and 43% (placebo). More than 90% of reported AEs were mild or moderate. The most frequently reported AEs were nausea, constipation, and vomiting (liposome bupivacaine, bupivacaine HCl) and nausea, dizziness, and vomiting (placebo).
Serious AEs were reported in 22 (2.7%) of the 823 patients in the liposome bupivacaine group, 24 (5.4%) of the 446 in the bupivacaine HCl group, and 2 (1.1%) of the 190 in the placebo group.65 None of the serious AEs in the liposome bupivacaine and placebo groups were considered treatment-related. Six patients in the bupivacaine HCl group had treatment-related serious AEs (hypoglycemia, arthrofibrosis, hemarthrosis, joint swelling, scar, knee arthroplasty).
Cardiac Safety. Possible cardiac effects associated with liposome bupivacaine were evaluated with data from studies conducted during the clinical development program.66 One hundred thirty-eight patients participated in the phase 2 safety and efficacy study in TKA. In these patients, a consistent change in mean heart rate (range, +12.2 to +16.5 beats per minute) was found across all liposome bupivacaine doses and with bupivacaine HCl. No clinically relevant changes from baseline in mean electrocardiographic parameters, including QTcF interval (QT interval adjusted using Fridericia’s correction formula), were found. In another analysis,67 liposome bupivacaine administered in a single subcutaneous dose (266, 399, 532, or 665 mg) to healthy volunteers did not prolong (vs placebo) QTc interval.
Wound Healing. The potential effects of liposome bupivacaine on wound healing were evaluated with results from 10 phase 2 and 3 studies.68 The assessments, which varied across studies, included clinicians’ overall satisfaction with patient wound healing, wound status assessment (categories included erythema, drainage, edema, and induration), and wound scarring (categories included pigmentation, height, pliability, and vascularity). Clinician-assessed scores reflected high satisfaction with wound healing overall. There were few statistically significant differences in wound status assessments between liposome bupivacaine and the comparators and no statistically significant differences in scarring between liposome bupivacaine and bupivacaine HCl.
The potential of liposome bupivacaine to have adverse intra-articular effects was assessed with drainage samples from patients (n = 23) who had TKA and received liposome bupivacaine (133, 266, 399, or 532 mg) or bupivacaine HCl (150 mg) by wound infiltration near the intra-articular space.51,65 Only small amounts of bupivacaine were present in drainage fluid collected for 12 hours after liposome bupivacaine administration, comparable to bupivacaine HCl administration.65 Currently, the product is not approved for intra-articular use.
Compatibility With Diluents, Other Medications, and Implant Materials
Liposome bupivacaine may be expanded up to a ratio of 1:14 by volume (to a final total volume of 300 mL or a concentration of 0.89 mg/mL) using preservative-free normal (0.9%) sterile saline for injection.14 It has also been shown in vitro to be compatible with lactated Ringer solution as a diluent.69
Liposome bupivacaine should not be admixed with other medications before administration.14 No formal drug–drug interaction studies have been conducted with liposome bupivacaine, but it has been shown in vitro to be compatible with epinephrine solutions, with certain anti-infective medications (eg, bacitracin, gentamicin, cefazolin, cefuroxime), with certain analgesics (eg, ketorolac, morphine), with an antihypertensive medication (clonidine), with an antihemorrhagic medication (tranexamic acid), and with certain corticosteroids (eg, methylprednisolone, triamcinolone acetonide). These medications may be coadministered in the same location as liposome bupivacaine.69
Topical antiseptics (eg, povidone iodine) may be used in surgical procedures involving liposome bupivacaine as long as they are not directly mixed with liposome bupivacaine and are allowed to dry before it is administered. If a topical antiseptic is used for wound irrigation, the wound should be rinsed clear before liposome bupivacaine administration.14,69
Liposome bupivacaine may be coadministered into the same surgical site immediately after bupivacaine HCl as long as the dose ratio of liposome bupivacaine to bupivacaine HCl is 2:1 or higher. Because of the prolonged-release pharmacokinetic profile of liposome bupivacaine and the potential for increased bupivacaine exposure, bupivacaine HCl should not be administered within 96 hours after administration of liposome bupivacaine.14,69
In vitro coincubation studies of liposome bupivacaine and other local anesthetics, including ropivacaine, lidocaine, and mepivacaine, have found rapid release of free bupivacaine from the liposome matrix. Therefore, after giving any of these other local anesthetics, surgeons should wait at least 20 minutes before administering liposome bupivacaine into the same area.14,69
In vitro studies have shown that liposome bupivacaine is compatible with a wide range of commonly used implant materials, including polypropylene, expanded polytetrafluoroethylene, stainless steel, titanium, and smooth- and textured-type silicone.69
Investigational Use and Ongoing Studies
A phase 2 randomized, double-masked, dose-escalating/deescalating study was conducted to evaluate the efficacy, safety, and pharmacokinetics of liposome bupivacaine (155, 199, or 310 mg) in comparison with bupivacaine HCl 125 mg for ankle nerve block in patients undergoing bunionectomy (N = 58).70 The study medication was injected into 3 sites to reach the posterior tibial, sural, deep peroneal, superficial peroneal, and saphenous nerves. Pharmacokinetic exposure was higher for liposome bupivacaine than for bupivacaine HCl, as reflected by a significantly greater area under the curve, lower Cmax (maximum serum concentration), and longer mean half-life. Mean pain intensity scores were lower in the bupivacaine HCl group than in each liposome bupivacaine group the first 12 hours after surgery. However, the liposome bupivacaine 310-mg group had similar or lower scores than the bupivacaine HCl group from 12 to 96 hours after surgery. The most common AEs in the liposome bupivacaine group were gastrointestinal and not treatment-related.70
The efficacy and safety of liposome bupivacaine, administered as a femoral nerve block for postsurgical analgesia, were assessed in a phase 2/3 manufacturer-sponsored, placebo-controlled, multicenter, randomized, double-blind 2-part study (NCT01683071)71 in 280 TKA patients.71,72 Part 2 of the study, comparing liposome bupivacaine 266 mg (n = 116) and placebo (n = 116), met its primary endpoint, demonstrating statistical significance in favor of liposome bupivacaine for cumulative pain scores over 72 hours (P < .0001), with decreased opioid use (P < .05) and a safety profile similar to that of placebo.72
Other ongoing investigator-sponsored studies in orthopedic populations include comparisons of liposome bupivacaine and bupivacaine HCl for ultrasound-guided periarticular hip infiltration in hip arthroplasty (NTC01917191),73 as femoral nerve block in TKA (NCT01977339),74 and as interscalene brachial plexus block in arthroscopic shoulder surgery (NCT01977352).75 The primary efficacy outcome measure in these studies was postsurgical opioid use.73-75
Health Economics
A series of phase 4 health economics studies was conducted for gastrointestinal surgeries, including open colectomy, laparoscopic colectomy, and ileostomy reversal.53-56,76 These studies, of similar design, showed that a liposome bupivacaine–based multimodal analgesic regimen was associated with reduced opioid use, shorter hospital LOS, and lower hospitalization costs in comparison with a traditional opioid-based regimen.53-56 Although pooled analysis of these studies showed a cost savings of more than $2000 per patient and an LOS decrease of 1.4 days,76 all were conducted in the gastrointestinal surgery setting. Studies are needed to fully assess the economic benefits associated with liposome bupivacaine in the orthopedic surgery setting.
Conclusion
Liposome bupivacaine represents a potentially important contributor to multimodal analgesic regimens used to manage postsurgical pain. Liposome bupivacaine has demonstrated efficacy in providing prolonged postsurgical analgesia and reducing postsurgical opioid use in most surgical settings studied. Additional data from health economics studies in gastrointestinal surgery suggest liposome bupivacaine–based multimodal analgesic regimens may also contribute to reductions in hospital LOS and hospitalization costs. Non-industry-sponsored trials are needed to answer these crucial questions in orthopedic surgery settings. Nevertheless, data on the safety and efficacy of liposome bupivacaine for postsurgical analgesia continue to accumulate, and liposome bupivacaine appears to be a feasible therapeutic option for managing postsurgical pain in orthopedic surgery.
1. Centers for Disease Control and Prevention. Number of all-listed procedures for discharges from short-stay hospitals, by procedure category and age: United States, 2010. http://www.cdc.gov/nchs/data/nhds/4procedures/2010pro4_numberprocedureage.pdf. Accessed January 30, 2015.
2. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.
3. Bonica JJ. Postoperative pain. In: Bonica JJ, ed. The Management of Pain. Malvern, PA: Lea & Febiger; 1990:461-480.
4. Apfelbaum JL, Ashburn MA, Connis RT, et al; American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012;116(2):248-273.
5. Golf M, Daniels SE, Onel E. A phase 3, randomized, placebo-controlled trial of DepoFoam® bupivacaine (extended-release bupivacaine local analgesic) in bunionectomy. Adv Ther. 2011;28(9):776-788.
6. Oderda G. Challenges in the management of acute postsurgical pain. Pharmacotherapy. 2012;32(9 pt 2):6S-11S.
7. Oderda GM, Gan TJ, Johnson BH, Robinson SB. Effect of opioid-related adverse events on outcomes in selected surgical patients. J Pain Palliat Care Pharmacother. 2013;27(1):62-70.
8. Wu CL, Naqibuddin M, Rowlingson AJ, Lietman SA, Jermyn RM, Fleisher LA. The effect of pain on health-related quality of life in the immediate postoperative period. Anesth Analg. 2003;97(4):1078-1085.
9. Morrison RS, Magaziner J, McLaughlin MA, et al. The impact of post-operative pain on outcomes following hip fracture. Pain. 2003;103(3):303-311.
10. Capdevila X, Barthelet Y, Biboulet P, Ryckwaert Y, Rubenovitch J, d’Athis F. Effects of perioperative analgesic technique on the surgical outcome and duration of rehabilitation after major knee surgery. Anesthesiology. 1999;91(1):8-15.
11. Capdevila X, Dadure C, Bringuier S, et al. Effect of patient-controlled perineural analgesia on rehabilitation and pain after ambulatory orthopedic surgery: a multicenter randomized trial. Anesthesiology. 2006;105(3):566-573.
12. Ranawat AS, Ranawat CS. Pain management and accelerated rehabilitation for total hip and total knee arthroplasty. J Arthroplasty. 2007;22(7 Suppl 3):12-15.
13. Hamilton DF, Lane JV, Gaston P, et al. What determines patient satisfaction with surgery? A prospective cohort study of 4709 patients following total joint replacement. BMJ Open. 2013;3(4):e002525.
14. Exparel [prescribing information]. Parsippany, NJ: Pacira Pharmaceuticals, Inc.; 2014.
15. DeWeese FT, Akbari Z, Carline E. Pain control after knee arthroplasty: intraarticular versus epidural anesthesia. Clin Orthop Relat Res. 2001;392(11):226-231.
16. Pati AB, Perme D, Trail M, Henry PK, Bryan WJ. Rehabilitation parameters in total knee replacement patients undergoing epidural vs. conventional analgesia. J Orthop Sports Phys Ther. 1994;19(2):88-92.
17. Browne C, Copp S, Reden L, Pulido P, Colwell C Jr. Bupivacaine bolus injection versus placebo for pain management following total knee arthroplasty. J Arthroplasty. 2004;19(3):377-380.
18. Nechleba J, Rogers V, Cortina G, Cooney T. Continuous intra-articular infusion of bupivacaine for postoperative pain following total knee arthroplasty. J Knee Surg. 2005;18(3):197-202.
19. Campbell A, McCormick M, McKinlay K, Scott NB. Epidural vs. lumbar plexus infusions following total knee arthroplasty: randomized controlled trial. Eur J Anaesthesiol. 2008;25(6):502-507.
20. Serpell MG, Millar FA, Thomson MF. Comparison of lumbar plexus block versus conventional opioid analgesia after total knee replacement. Anaesthesia. 1991;46(4):275-277.
21. Lareau JM, Robbins CE, Talmo CT, Mehio AK, Puri L, Bono JV. Complications of femoral nerve blockade in total knee arthroplasty and strategies to reduce patient risk. J Arthroplasty. 2012;27(4):564-568.
22. Charous MT, Madison SJ, Suresh PJ, et al. Continuous femoral nerve blocks: varying local anesthetic delivery method (bolus versus basal) to minimize quadriceps motor block while maintaining sensory block. Anesthesiology. 2011;115(4):774-781.
23. Gottschalk A, Burmeister MA, Radtke P, et al. Continuous wound infiltration with ropivacaine reduces pain and analgesic requirement after shoulder surgery. Anesth Analg. 2003;97(4):1086-1091.
24. Kerr DR, Kohan L. Local infiltration analgesia: a technique for the control of acute postoperative pain following knee and hip surgery: a case study of 325 patients. Acta Orthop. 2008;79(2):174-183.
25. Dalury DF, Lieberman JR, MacDonald SJ. Current and innovative pain management techniques in total knee arthroplasty. J Bone Joint Surg Am. 2011;93(20):1938-1943.
26. White PF. The role of non-opioid analgesic techniques in the management of pain after ambulatory surgery. Anesth Analg. 2002;94(3):577-585.
27. Dahl JB, Kehlet H. Non-steroidal anti-inflammatory drugs: rationale for use in severe postoperative pain. Br J Anaesth. 1991;66(6):703-712.
28. Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048-1056.
29. Candiotti K. Liposomal bupivacaine: an innovative nonopioid local analgesic for the management of postsurgical pain. Pharmacotherapy. 2012;32(9 Pt 2):19S-26S.
30. Bergese SD, Onel E, Portillo J. Evaluation of DepoFoam® bupivacaine for the treatment of postsurgical pain. Pain Manag. 2011;1(6):539-547.
31. Joranson DE, Ryan KM, Gilson AM, Dahl JL. Trends in medical use and abuse of opioid analgesics. JAMA. 2000;283(13):1710-1714.
32. Kuehn BM. Opioid prescriptions soar: increase in legitimate use as well as abuse. JAMA. 2007;297(3):249-251.
33. Compton WM, Volkow ND. Major increases in opioid analgesic abuse in the United States: concerns and strategies. Drug Alcohol Depend. 2006;81(2):103-107.
34. Ilfeld BM, Morey TE, Enneking FK. Delivery rate accuracy of portable, bolus-capable infusion pumps used for patient-controlled continuous regional analgesia. Reg Anesth Pain Med. 2003;28(1):17-23.
35. Ganapathy S, Amendola A, Lichfield R, Fowler PJ, Ling E. Elastomeric pumps for ambulatory patient controlled regional analgesia. Can J Anaesth. 2000;47(9):897-902.
36. Bray DA Jr, Nguyen J, Craig J, Cohen BE, Collins DR Jr. Efficacy of a local anesthetic pain pump in abdominoplasty. Plast Reconstr Surg. 2007;119(3):1054-1059.
37. Brown SL, Morrison AE. Local anesthetic infusion pump systems adverse events reported to the Food and Drug Administration. Anesthesiology. 2004;100(5):1305-1307.
38. Noyes FR, Fleckenstein CM, Barber-Westin SD. The development of postoperative knee chondrolysis after intra-articular pain pump infusion of an anesthetic medication: a series of twenty-one cases. J Bone Joint Surg Am. 2012;94(16):1448-1457.
39. Rapley JH, Beavis RC, Barber FA. Glenohumeral chondrolysis after shoulder arthroscopy associated with continuous bupivacaine infusion. Arthroscopy. 2009;25(12):1367-1373.
40. Institute for Safe Medication Practices. Process for handling elastomeric pain relief balls (ON-Q PainBuster and others) requires safety improvements. ISMP Medication Safety Alert. http://www.ismp.org/Newsletters/acutecare/articles/20090716.asp. Accessed January 30, 2015.
41. Pepin JL, Dasta JF, New M. Ensuring safe and economical use of elastomeric infusion devices. Am J Health Syst Pharm. 2011;68(24):2330-2331.
42. Birrer KL, Anderson RL, Liu-DeRyke X, Patel KR. Measures to improve safety of an elastomeric infusion system for pain management. Am J Health Syst Pharm. 2011;68(13):1251-1255.
43. Ilfeld BM, Enneking FK. Continuous peripheral nerve blocks at home: a review. Anesth Analg. 2005;100(6):1822-1833.
44. US Food and Drug Administration. Medical device recalls: I-Flow ON-Q Pump with ONDEMAND Bolus Button. http://www.fda.gov/MedicalDevices/Safety/ListofRecalls/ucm317826.htm. Accessed July 15, 2014.
45. Ilfeld BM, Morey TE, Enneking FK. Portable infusion pumps used for continuous regional analgesia: delivery rate accuracy and consistency. Reg Anesth Pain Med. 2003;28(5):424-432.
46. Ganapathy S. Wound/intra-articular infiltration or peripheral nerve blocks for orthopedic joint surgery: efficacy and safety issues. Curr Opin Anaesthesiol. 2012;25(5):615-620.
47. Ilfeld BM, Duke KB, Donohue MC. The association between lower extremity continuous peripheral nerve blocks and patient falls after knee and hip arthroplasty. Anesth Analg. 2010;111(6):1552-1554.
48. Angst MS, Drover DR. Pharmacology of drugs formulated with DepoFoam™: a sustained release drug delivery system for parenteral administration using multivesicular liposome technology. Clin Pharmacokinet. 2006;45(12):1153-1176.
49. Catterall WA, Mackie K. Local anesthetics. In: Gutstein HB, Akil H, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York, NY: McGraw-Hill; 2011:565-582.
50. Hu D, Onel E, Singla N, Kramer WG, Hadzic A. Pharmacokinetic profile of liposome bupivacaine injection following a single administration at the surgical site. Clin Drug Investig. 2013;33(2):109-115.
51. Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536.
52. Bergese SD, Ramamoorthy S, Patou G, Bramlett K, Gorfine SR, Candiotti KA. Efficacy profile of liposome bupivacaine, a novel formulation of bupivacaine for postsurgical analgesia. J Pain Res. 2012;5:107-116.
53. Cohen SM. Extended pain relief trial utilizing infiltration of Exparel®, a long-acting multivesicular liposome formulation of bupivacaine: a phase IV health economic trial in adult patients undergoing open colectomy. J Pain Res. 2012;5:567-572.
54. Candiotti KA, Sands LR, Lee E, et al. Liposome bupivacaine for postsurgical analgesia in adult patients undergoing laparoscopic colectomy: results from prospective phase IV sequential cohort studies assessing health economic outcomes. Curr Ther Res. 2014;76:1-6.
55. Marcet JE, Nfonsam VN, Larach S. An extended paIn relief trial utilizing the infiltration of a long-acting Multivesicular liPosome foRmulation Of bupiVacaine, EXPAREL (IMPROVE): a Phase IV health economic trial in adult patients undergoing ileostomy reversal. J Pain Res. 2013;6:549-555.
56. Vogel JD. Liposome bupivacaine (EXPAREL®) for extended pain relief in patients undergoing ileostomy reversal at a single institution with a fast-track discharge protocol: an IMPROVE phase IV health economics trial. J Pain Res. 2013;6:605-610.
57. Sternlicht A, Shapiro M, Robelen G, Vellayappan U, Tuerk IA. Initial findings using EXPAREL® (bupivacaine liposome injectable suspension) via infiltration into the transversus abdominis plane (TAP) for postsurgical analgesia in robotic prostatectomy (RP). Abstract presented at: Annual Fall Pain Meeting and Workshops of the American Society of Regional Anesthesia and Pain Medicine; November 15-18, 2012; Miami Beach, FL.
58. Feierman DE, Kronenfeld M, Gupta PM, Younger N, Logvinskiy E. Evaluation of Exparel® use via infiltration into the transversus abdominis plane for prolonged postoperative analgesia in subjects undergoing open abdominal hernia repair. Poster presented at: Annual Meeting of the International Anesthesia Research Society; May 4-7, 2013; San Diego, CA.
59. Bagsby DT, Ireland PH, Meneghini RM. Liposomal bupivacaine versus traditional periarticular injection for pain control after total knee arthroplasty. J Arthroplasty. 2014;29(8):1687-1690.
60. Broome B, Backlund I. Rapid recovery pain pathway for total knee arthroplasty results in improved pain management, decreased length of stay, and significant cost savings. Poster presented at: Annual Orthopedic and Spine Summit; September 18-20, 2013; San Antonio, TX.
61. Emerson RH, Barrington JW. Comparison of infiltration with long-acting bupivacaine to a femoral nerve catheter for total knee replacement. Abstract presented at: Annual Meeting of the American Academy of Orthopaedic Surgeons; March 11-15, 2014; New Orleans, LA. Abstract P124.
62. Barrington JW. Emerging data in the use of liposome bupivacaine: comparative review in 2,000 TJA patients. Oral presentation presented at: Annual Meeting of the American Academy of Orthopaedic Surgeons; March 11-15, 2014; New Orleans, LA.
63. Neal JM, Bernards CM, Butterworth JF, et al. ASRA practice advisory on local anesthetic systemic toxicity. Reg Anesth Pain Med. 2010;35(2):152-161.
64. D’Angelo R. Are the new local anesthetics worth their cost? Acta Anaesthesiol Scand. 2000;44(6):639-641.
65. Viscusi ER, Sinatra R, Onel E, Ramamoorthy SL. The safety of liposome bupivacaine, a novel local analgesic formulation. Clin J Pain. 2014;30(2):102-110.
66. Bergese SD, Onel E, Morren M, Morganroth J. Bupivacaine extended-release liposome injection exhibits a favorable cardiac safety profile. Reg Anesth Pain Med. 2012;37(2):145-151.
67. Naseem A, Harada T, Wang D, et al. Bupivacaine extended release liposome injection does not prolong QTc interval in a thorough QT/QTc study in healthy volunteers. J Clin Pharmacol. 2012;52(9):1441-1447.
68. Baxter R, Bramlett K, Onel E, Daniels S. Impact of local administration of liposome bupivacaine for postsurgical analgesia on wound healing: a review of data from ten prospective, controlled clinical studies. Clin Ther. 2013;35(3):312-320.
69. Kharitonov V. A review of the compatibility of liposome bupivacaine with other drug products and commonly used implant materials. Postgrad Med. 2014;126(1):129-138.
70. Ilfeld BM. Liposome bupivacaine in peripheral nerve blocks and epidural injections to manage postoperative pain. Expert Opin Pharmacother. 2013;14(17):2421-2431.
71. Femoral nerve block with liposome bupivacaine for postsurgical analgesia following total knee arthroplasty [NCT01683071]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01683071?term=NCT01683071%5C&rank=1. Accessed January 30, 2015.
72. Minkowitz H, Matthews A, Puckett C, Melson T. Liposome bupivacaine in femoral nerve block: initial results from a phase 2/3 pivotal study. Poster presented at: Annual Meeting of the American Society of Regional Anesthesia and Pain Medicine; April 3-6, 2014; Chicago, IL.
73. Ultrasound guided local infiltration analgesia for hip arthroscopy [NCT01907191]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01907191?term=NCT01907191&rank=1. Accessed January 30, 2015.
74. Efficacy of single injection femoral nerve block with liposomal bupivacaine for total knee arthroplasty [NCT01977339]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01977339?term=NCT01977339&rank=1. Accessed January 30, 2015.
75. Efficacy of interscalene brachial plexus block with liposomal bupivacaine for arthroscopic shoulder surgery [NCT01977352]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01977352?term=NCT01977352&rank=1. Accessed January 30, 2015.
76. Cohen SM, Vogel JD, Marcet JE, Candiotti K. Liposome bupivacaine for improvement in economic outcomes and opioid burden in GI surgery: IMPROVE study pooled analysis. J Pain Res. 2014;7:359-366.
Approximately 5.5 million patients undergo orthopedic surgery in the United States each year, and more than 1 million of the procedures are total knee arthroplasty (TKA) or total hip arthroplasty.1 From its 2010 level, demand for joint arthroplasty is expected to double by 2020 and quadruple by 2030.2
About half the patients who have major joint arthroplasty experience severe postsurgical pain.3 Because postsurgical pain may persist for days or weeks, and inadequate treatment is associated with negative outcomes, achieving effective postsurgical analgesia is an important consideration.4-7 Complications of inadequate postsurgical pain management include thromboembolic or pulmonary complications, development of chronic pain, and decrements in health-related quality of life.4,8
In patients who have orthopedic surgery, the inability to adequately control postsurgical pain has been associated with increased hospital length of stay (LOS), delayed time to ambulation, and reduced capacity for exercise.9-12 A recent study involving 4709 patients who had hip or knee arthroplasty found that postsurgical pain relief was the second most highly correlated factor with respect to overall patient satisfaction (how well surgery met patient expectations was the most highly correlated factor),13 suggesting that postsurgical analgesia should be a focus of surgical practice.
A prolonged-release liposomal formulation of the local anesthetic bupivacaine is now available. Bupivacaine liposome injectable suspension (Exparel; Pacira Pharmaceuticals, Inc., Parsippany, New Jersey) is indicated for administration into the surgical site to produce postsurgical analgesia.14 In this article, we review evidence from clinical studies regarding the potential contribution of liposome bupivacaine to improving postsurgical pain management when used as part of a multimodal analgesic regimen in patients undergoing orthopedic surgery.
Postsurgical Pain Management in Orthopedic Surgery
Frequently Used Modalities
Analgesic modalities commonly used for perioperative pain management include central (eg, epidural),4,10,15,16 central regional (eg, neuraxial),4 peripheral regional (eg, peripheral nerve blocks, local/regional surgical site infiltration, intra-articular administration),4,10,15,17-25 and intravenous (IV) patient-controlled analgesia.4,10,25 These pharmacologic interventions may be augmented by nonpharmacologic modalities (eg, transcutaneous electrical nerve stimulation).26
Pharmacologic treatment options for perioperative pain management include opioids, local anesthetics, clonidine, ketamine, nonsteroidal anti-inflammatory drugs, acetaminophen, and calcium-channel blockers.4,26-28 In TKA, “drug cocktails” (eg, combinations of ropivacaine, ketorolac, epinephrine, and clonidine) for regional or intra-articular injection can also provide effective immediate postsurgical analgesia.25 Although opioids are the most commonly used analgesics for management of orthopedic perioperative pain,25 their use is often associated with adverse effects (AEs), including constipation or ileus, nausea, sedation, dizziness, pruritus, urinary retention, and respiratory depression.6
Multimodal Analgesic Regimens for Postsurgical Pain Management
Current American Society of Anesthesiologists guidelines endorse use of multimodal analgesia, whenever possible, to provide effective management of acute perioperative pain.4 Multimodal analgesia involves applying 2 or more agents with different mechanisms of action to achieve a synergistic effect, which allows each agent to be reduced in dose4,28 and thereby may limit the risk and severity of dose-related AEs.4,25,28
Multimodal analgesia aims to reduce the risk for opioid-related AEs (ORAEs) and the impact of opioids on postsurgical milestones (eg, ambulation, discharge) and may reduce opioid consumption, with attendant reductions in ORAE risk.29,30 Health economics studies have shown that postsurgical ORAEs are associated with increased hospital costs and LOS.6 In a study using a national hospital database, development of an ORAE (vs no ORAE) in postsurgical patients was associated with mean increases of about $4700 in hospital costs and 3.3 days in LOS.7 Reducing postsurgical opioid use may also help reduce the risk for opioid abuse, addiction, and diversion.31-33
One approach to reducing opioid use involves continuous or intermittent administration of local anesthetics by elastomeric pumps to extend duration of postsurgical analgesia.34-36 However, use of elastomeric pumps has been associated with risk for AEs, including tissue necrosis, sloughing, wound infection, and chondrolysis.37-40 In addition, AEs related to “dose dumping” (accidental delivery of excessive doses) have been reported.40-44 Key issues that may negatively affect rehabilitation after orthopedic surgery include consistency and accuracy of analgesic delivery and the potential for motor block–induced muscle weakness, which may lead to falls and constrain ambulation.45-47
Liposome Bupivacaine
Description
Drug Delivery Technology. Liposome bupivacaine incorporates DepoFoam drug delivery technology (Pacira Pharmaceuticals, Inc.) to facilitate prolonged release of bupivacaine. This technology is based on creation of multivesicular liposome particles (diameter, 10-30 µm) with multiple aqueous chambers.30,48 After administration into the surgical site, bupivacaine diffuses from chambers in the liposomal particles over time, providing analgesia and reduced opioid requirements for up to 72 hours.29,30
Indication, Mechanism of Action, Pharmacokinetics, and Dose/Administration. Liposome bupivacaine is indicated for single-dose administration into the surgical site to produce postsurgical analgesia in patients at least 18 years old.14 Like other local anesthetics, liposome bupivacaine is thought to exert its pharmacologic effects by interacting with voltage-gated Na+ channels on neural membranes to raise the threshold for electrical excitability, to slow nerve impulse propagation, and to reduce the rate of rise of the action potential.14,49
Liposome bupivacaine has dose-proportional pharmacokinetics.50 Presence of a small amount of extra-liposomal bupivacaine in the formulation leads to a bimodal pharmacokinetic profile, with an initial peak serum concentration about 1 hour after administration, followed by a second peak within 12 to 36 hours (Figure).50
Maximum amount of liposome bupivacaine approved for single administration is 266 mg (packaged as 20 mL of a 1.3% solution). However, product labeling includes safety data associated with doses of 532 mg or less.14 The appropriate volume to be used should be based on the amount required to cover the surgical area. Liposome bupivacaine may be expanded with preservative-free normal (0.9%) sterile saline to a total volume of 300 mL: 20 mL liposome bupivacaine plus 280 mL or less diluent, with final concentration of 0.89 mg/mL (1:14 by volume).14
A 25-gauge or larger bore needle should be used to slowly inject liposome bupivacaine into soft tissues of the surgical site, with frequent aspiration to check for blood to minimize risk for intravascular injection.14 Total volume used and fraction injected in specific regions of the surgical site depend on the procedure. For example, a TKA study used 266 mg diluted to a total volume of 60 mL, with 8 mL infiltrated to the area around the medial capsule, 8 mL around the lateral capsule, 12 mL around the posterior capsule, 8 mL around the peripatellar area, 12 mL into the capsulotomy incision, and 12 mL into the subcutaneous tissue on each side of the incision.51
Efficacy
Multiple Surgical Settings. The efficacy of liposome bupivacaine, either alone or as a component of a multimodal analgesic regimen, has been evaluated in a series of 10 phase 2 and 3 studies (8 active-controlled, 2 placebo-controlled) involving 823 patients undergoing TKA, bunionectomy, hemorrhoidectomy, inguinal hernia repair, or mammoplasty.52 Patients received a single liposome bupivacaine dose ranging from 66 to 532 mg.52
Combined analyses of efficacy data from these studies found that liposome bupivacaine–based multimodal analgesic regimens produced postsurgical analgesia for up to 72 hours, increased time to first use of opioid rescue medication after surgery, and reduced total amount of postsurgical opioid consumption versus placebo.52
Compared with standard of care, liposome bupivacaine has been shown to provide effective analgesia in open-label studies in patients undergoing open colectomy,53 laparoscopic colectomy,54 and ileostomy reversal,55,56 as reflected in assessments of postsurgical opioid consumption, LOS, and hospital costs. It has also been studied when administered by infiltration into the transversus abdominis plane (TAP) in patients having laparoscopic prostatectomy and open abdominal hernia repair.57,58
Orthopedic Surgery. In a phase 2 randomized, double-blind, dose-ranging study, TKA patients (N = 138) received bupivacaine HCl 150 mg or liposome bupivacaine 133, 266, 399, or 532 mg administered by local infiltration into the capsulotomy incision and on either side of the incision before wound closure.51 Postsurgical rescue analgesia was available to all patients. Cumulative pain intensity scores with activity (primary efficacy measure) were not statistically different between liposome bupivacaine groups and the bupivacaine HCl group through postoperative day 4. Mean scores in the liposome bupivacaine 266-, 399-, and 532-mg groups were numerically lower than for those treated with bupivacaine HCl on postoperative days 2 to 5, with all doses of liposome bupivacaine having a statistically significant lower pain score at rest on day 5. There were no statistically significant differences across treatment groups with respect to total amount of postsurgical opioids used.
In a phase 3 randomized, double-blind study of TKA patients (N = 245), liposome bupivacaine 532 mg administered into the surgical site was compared with bupivacaine HCl 200 mg for postsurgical analgesia.52 Rescue analgesia was available to all patients. No statistically significant between-group differences were found with respect to postsurgical cumulative pain scores through 72 hours (primary efficacy endpoint).
In a single-center retrospective TKA study, postsurgical outcomes in a patient cohort that received intraoperative periarticular infiltration with liposome bupivacaine 266 mg (n = 65) were compared with a cohort that received infiltration with a combination of ropivacaine 400 mg, morphine 5 mg, and epinephrine 0.4 mg (n = 85).59 Patient-reported postsurgical pain scores were similar in the 2 treatment groups during the first 24 hours after surgery and at discharge. Mean (SD) pain scores during hospitalization after the first 24 hours until discharge were significantly (P = .04) higher in the liposome bupivacaine group, 4.9 (1.4), than in the periarticular group, 4.4 (1.6). There was no significant difference between the 2 treatment groups in postsurgical opioid use. The study demonstrated no advantage to using liposome bupivacaine injections with respect to pain relief, but it was a retrospective review in which pain scores were obtained from electronic medical records. It is essential that liposome bupivacaine be compared with intra-articular injections in well-designed randomized trials.
Another single-center, matched-cohort TKA study (N = 200) compared a liposome bupivacaine regimen with femoral nerve block.60 Compared with patients who received femoral nerve block, patients who received liposome bupivacaine reported lower pain intensity scores after surgery and had shorter LOS, reduced costs, and improved knee flexion at follow-up.60
Results from 2 other studies were presented at the 2014 meeting of the American Academy of Orthopaedic Surgeons (AAOS). One was a single-center, matched-cohort TKA study (N = 72) comparing infiltration of a single dose of liposome bupivacaine into the surgical site with continuous femoral nerve block.61 The 2 treatment groups had similar mean postsurgical pain intensity scores on a 0-to-10 visual analog scale, 1.8 for liposome bupivacaine and 2.3 for continuous nerve block (P = NS), but total amount of postsurgical opioids (hydrocodone-equivalent milligrams) was significantly (P < .0001) less in the liposome bupivacaine group (82 vs 177 mg).
The other study presented at the AAOS meeting was a larger, prospective case–control study comparing outcomes between 1000 patients who had total joint arthroplasty (TJA) with liposome bupivacaine and 1000 control patients who had TJA without liposome bupivacaine.62 For the control and liposome bupivacaine cohorts, respectively, mean postsurgical pain intensity scores were 2.41 and 1.98 (P < .0001), mean LOS was 2.83 days and 2.66 days (P < .02), and incidence of falls was 1.0% and 0.2% (P = .02). Average per-patient costs were $1246 lower in the liposome bupivacaine cohort.
A pivotal phase 3 placebo-controlled study compared liposome bupivacaine 106 mg with placebo in patients undergoing bunionectomy (N = 193).5 Rescue medication was available to all patients. Cumulative pain scores were significantly (P = .0005) lower in the liposome bupivacaine group (125) than in the placebo group (146) through 24 hours after surgery (primary efficacy measure) and significantly (P = .0229) lower (197 vs 220) through 36 hours. Median time to first use of rescue opioids was delayed in favor of the liposome bupivacaine group (7.2 vs 4.3 hours; P < .0001). Mean total number of opioid tablets used within 24 hours after surgery was also significantly lower (3.8 vs 4.7; P = .008), and a larger percentage of patients in the liposome bupivacaine group avoided opioid use altogether through 24 hours (7% vs 1%; P = .04).
Efficacy data for liposome bupivacaine appear promising for relief of pain after joint arthroplasty and other orthopedic procedures but have their limitations. First, no randomized trials have compared liposome bupivacaine with locally injected pain medications (intra-articular injections in TKA or hip arthroplasty). As these injections are quite common now, such analyses are essential. Second, cost-effectiveness studies are needed for orthopedic procedures. Third, most of the published studies were sponsored by the manufacturer of liposome bupivacaine—a situation that raises questions about potential bias. Non-industry-sponsored randomized trials assessing efficacy, safety, and cost-effectiveness are needed.
Safety
Local anesthetics, including liposome bupivacaine, have the potential for central nervous system (CNS) or cardiac toxicity resulting from excessive systemic absorption or inadvertent IV administration.63 However, reported serious CNS or cardiac-related AEs are rare.63,64
AE Profile. Safety data from 10 phase 2 and 3 studies involving 823 patients who received liposome bupivacaine were evaluated.65 Of these patients, 545 received a dose of 266 mg or less (maximum dose approved by the US Food and Drug Administration [FDA]). Liposome bupivacaine was generally well tolerated. Reported AE incidence was 62% (liposome bupivacaine), 75% (bupivacaine HCl), and 43% (placebo). More than 90% of reported AEs were mild or moderate. The most frequently reported AEs were nausea, constipation, and vomiting (liposome bupivacaine, bupivacaine HCl) and nausea, dizziness, and vomiting (placebo).
Serious AEs were reported in 22 (2.7%) of the 823 patients in the liposome bupivacaine group, 24 (5.4%) of the 446 in the bupivacaine HCl group, and 2 (1.1%) of the 190 in the placebo group.65 None of the serious AEs in the liposome bupivacaine and placebo groups were considered treatment-related. Six patients in the bupivacaine HCl group had treatment-related serious AEs (hypoglycemia, arthrofibrosis, hemarthrosis, joint swelling, scar, knee arthroplasty).
Cardiac Safety. Possible cardiac effects associated with liposome bupivacaine were evaluated with data from studies conducted during the clinical development program.66 One hundred thirty-eight patients participated in the phase 2 safety and efficacy study in TKA. In these patients, a consistent change in mean heart rate (range, +12.2 to +16.5 beats per minute) was found across all liposome bupivacaine doses and with bupivacaine HCl. No clinically relevant changes from baseline in mean electrocardiographic parameters, including QTcF interval (QT interval adjusted using Fridericia’s correction formula), were found. In another analysis,67 liposome bupivacaine administered in a single subcutaneous dose (266, 399, 532, or 665 mg) to healthy volunteers did not prolong (vs placebo) QTc interval.
Wound Healing. The potential effects of liposome bupivacaine on wound healing were evaluated with results from 10 phase 2 and 3 studies.68 The assessments, which varied across studies, included clinicians’ overall satisfaction with patient wound healing, wound status assessment (categories included erythema, drainage, edema, and induration), and wound scarring (categories included pigmentation, height, pliability, and vascularity). Clinician-assessed scores reflected high satisfaction with wound healing overall. There were few statistically significant differences in wound status assessments between liposome bupivacaine and the comparators and no statistically significant differences in scarring between liposome bupivacaine and bupivacaine HCl.
The potential of liposome bupivacaine to have adverse intra-articular effects was assessed with drainage samples from patients (n = 23) who had TKA and received liposome bupivacaine (133, 266, 399, or 532 mg) or bupivacaine HCl (150 mg) by wound infiltration near the intra-articular space.51,65 Only small amounts of bupivacaine were present in drainage fluid collected for 12 hours after liposome bupivacaine administration, comparable to bupivacaine HCl administration.65 Currently, the product is not approved for intra-articular use.
Compatibility With Diluents, Other Medications, and Implant Materials
Liposome bupivacaine may be expanded up to a ratio of 1:14 by volume (to a final total volume of 300 mL or a concentration of 0.89 mg/mL) using preservative-free normal (0.9%) sterile saline for injection.14 It has also been shown in vitro to be compatible with lactated Ringer solution as a diluent.69
Liposome bupivacaine should not be admixed with other medications before administration.14 No formal drug–drug interaction studies have been conducted with liposome bupivacaine, but it has been shown in vitro to be compatible with epinephrine solutions, with certain anti-infective medications (eg, bacitracin, gentamicin, cefazolin, cefuroxime), with certain analgesics (eg, ketorolac, morphine), with an antihypertensive medication (clonidine), with an antihemorrhagic medication (tranexamic acid), and with certain corticosteroids (eg, methylprednisolone, triamcinolone acetonide). These medications may be coadministered in the same location as liposome bupivacaine.69
Topical antiseptics (eg, povidone iodine) may be used in surgical procedures involving liposome bupivacaine as long as they are not directly mixed with liposome bupivacaine and are allowed to dry before it is administered. If a topical antiseptic is used for wound irrigation, the wound should be rinsed clear before liposome bupivacaine administration.14,69
Liposome bupivacaine may be coadministered into the same surgical site immediately after bupivacaine HCl as long as the dose ratio of liposome bupivacaine to bupivacaine HCl is 2:1 or higher. Because of the prolonged-release pharmacokinetic profile of liposome bupivacaine and the potential for increased bupivacaine exposure, bupivacaine HCl should not be administered within 96 hours after administration of liposome bupivacaine.14,69
In vitro coincubation studies of liposome bupivacaine and other local anesthetics, including ropivacaine, lidocaine, and mepivacaine, have found rapid release of free bupivacaine from the liposome matrix. Therefore, after giving any of these other local anesthetics, surgeons should wait at least 20 minutes before administering liposome bupivacaine into the same area.14,69
In vitro studies have shown that liposome bupivacaine is compatible with a wide range of commonly used implant materials, including polypropylene, expanded polytetrafluoroethylene, stainless steel, titanium, and smooth- and textured-type silicone.69
Investigational Use and Ongoing Studies
A phase 2 randomized, double-masked, dose-escalating/deescalating study was conducted to evaluate the efficacy, safety, and pharmacokinetics of liposome bupivacaine (155, 199, or 310 mg) in comparison with bupivacaine HCl 125 mg for ankle nerve block in patients undergoing bunionectomy (N = 58).70 The study medication was injected into 3 sites to reach the posterior tibial, sural, deep peroneal, superficial peroneal, and saphenous nerves. Pharmacokinetic exposure was higher for liposome bupivacaine than for bupivacaine HCl, as reflected by a significantly greater area under the curve, lower Cmax (maximum serum concentration), and longer mean half-life. Mean pain intensity scores were lower in the bupivacaine HCl group than in each liposome bupivacaine group the first 12 hours after surgery. However, the liposome bupivacaine 310-mg group had similar or lower scores than the bupivacaine HCl group from 12 to 96 hours after surgery. The most common AEs in the liposome bupivacaine group were gastrointestinal and not treatment-related.70
The efficacy and safety of liposome bupivacaine, administered as a femoral nerve block for postsurgical analgesia, were assessed in a phase 2/3 manufacturer-sponsored, placebo-controlled, multicenter, randomized, double-blind 2-part study (NCT01683071)71 in 280 TKA patients.71,72 Part 2 of the study, comparing liposome bupivacaine 266 mg (n = 116) and placebo (n = 116), met its primary endpoint, demonstrating statistical significance in favor of liposome bupivacaine for cumulative pain scores over 72 hours (P < .0001), with decreased opioid use (P < .05) and a safety profile similar to that of placebo.72
Other ongoing investigator-sponsored studies in orthopedic populations include comparisons of liposome bupivacaine and bupivacaine HCl for ultrasound-guided periarticular hip infiltration in hip arthroplasty (NTC01917191),73 as femoral nerve block in TKA (NCT01977339),74 and as interscalene brachial plexus block in arthroscopic shoulder surgery (NCT01977352).75 The primary efficacy outcome measure in these studies was postsurgical opioid use.73-75
Health Economics
A series of phase 4 health economics studies was conducted for gastrointestinal surgeries, including open colectomy, laparoscopic colectomy, and ileostomy reversal.53-56,76 These studies, of similar design, showed that a liposome bupivacaine–based multimodal analgesic regimen was associated with reduced opioid use, shorter hospital LOS, and lower hospitalization costs in comparison with a traditional opioid-based regimen.53-56 Although pooled analysis of these studies showed a cost savings of more than $2000 per patient and an LOS decrease of 1.4 days,76 all were conducted in the gastrointestinal surgery setting. Studies are needed to fully assess the economic benefits associated with liposome bupivacaine in the orthopedic surgery setting.
Conclusion
Liposome bupivacaine represents a potentially important contributor to multimodal analgesic regimens used to manage postsurgical pain. Liposome bupivacaine has demonstrated efficacy in providing prolonged postsurgical analgesia and reducing postsurgical opioid use in most surgical settings studied. Additional data from health economics studies in gastrointestinal surgery suggest liposome bupivacaine–based multimodal analgesic regimens may also contribute to reductions in hospital LOS and hospitalization costs. Non-industry-sponsored trials are needed to answer these crucial questions in orthopedic surgery settings. Nevertheless, data on the safety and efficacy of liposome bupivacaine for postsurgical analgesia continue to accumulate, and liposome bupivacaine appears to be a feasible therapeutic option for managing postsurgical pain in orthopedic surgery.
Approximately 5.5 million patients undergo orthopedic surgery in the United States each year, and more than 1 million of the procedures are total knee arthroplasty (TKA) or total hip arthroplasty.1 From its 2010 level, demand for joint arthroplasty is expected to double by 2020 and quadruple by 2030.2
About half the patients who have major joint arthroplasty experience severe postsurgical pain.3 Because postsurgical pain may persist for days or weeks, and inadequate treatment is associated with negative outcomes, achieving effective postsurgical analgesia is an important consideration.4-7 Complications of inadequate postsurgical pain management include thromboembolic or pulmonary complications, development of chronic pain, and decrements in health-related quality of life.4,8
In patients who have orthopedic surgery, the inability to adequately control postsurgical pain has been associated with increased hospital length of stay (LOS), delayed time to ambulation, and reduced capacity for exercise.9-12 A recent study involving 4709 patients who had hip or knee arthroplasty found that postsurgical pain relief was the second most highly correlated factor with respect to overall patient satisfaction (how well surgery met patient expectations was the most highly correlated factor),13 suggesting that postsurgical analgesia should be a focus of surgical practice.
A prolonged-release liposomal formulation of the local anesthetic bupivacaine is now available. Bupivacaine liposome injectable suspension (Exparel; Pacira Pharmaceuticals, Inc., Parsippany, New Jersey) is indicated for administration into the surgical site to produce postsurgical analgesia.14 In this article, we review evidence from clinical studies regarding the potential contribution of liposome bupivacaine to improving postsurgical pain management when used as part of a multimodal analgesic regimen in patients undergoing orthopedic surgery.
Postsurgical Pain Management in Orthopedic Surgery
Frequently Used Modalities
Analgesic modalities commonly used for perioperative pain management include central (eg, epidural),4,10,15,16 central regional (eg, neuraxial),4 peripheral regional (eg, peripheral nerve blocks, local/regional surgical site infiltration, intra-articular administration),4,10,15,17-25 and intravenous (IV) patient-controlled analgesia.4,10,25 These pharmacologic interventions may be augmented by nonpharmacologic modalities (eg, transcutaneous electrical nerve stimulation).26
Pharmacologic treatment options for perioperative pain management include opioids, local anesthetics, clonidine, ketamine, nonsteroidal anti-inflammatory drugs, acetaminophen, and calcium-channel blockers.4,26-28 In TKA, “drug cocktails” (eg, combinations of ropivacaine, ketorolac, epinephrine, and clonidine) for regional or intra-articular injection can also provide effective immediate postsurgical analgesia.25 Although opioids are the most commonly used analgesics for management of orthopedic perioperative pain,25 their use is often associated with adverse effects (AEs), including constipation or ileus, nausea, sedation, dizziness, pruritus, urinary retention, and respiratory depression.6
Multimodal Analgesic Regimens for Postsurgical Pain Management
Current American Society of Anesthesiologists guidelines endorse use of multimodal analgesia, whenever possible, to provide effective management of acute perioperative pain.4 Multimodal analgesia involves applying 2 or more agents with different mechanisms of action to achieve a synergistic effect, which allows each agent to be reduced in dose4,28 and thereby may limit the risk and severity of dose-related AEs.4,25,28
Multimodal analgesia aims to reduce the risk for opioid-related AEs (ORAEs) and the impact of opioids on postsurgical milestones (eg, ambulation, discharge) and may reduce opioid consumption, with attendant reductions in ORAE risk.29,30 Health economics studies have shown that postsurgical ORAEs are associated with increased hospital costs and LOS.6 In a study using a national hospital database, development of an ORAE (vs no ORAE) in postsurgical patients was associated with mean increases of about $4700 in hospital costs and 3.3 days in LOS.7 Reducing postsurgical opioid use may also help reduce the risk for opioid abuse, addiction, and diversion.31-33
One approach to reducing opioid use involves continuous or intermittent administration of local anesthetics by elastomeric pumps to extend duration of postsurgical analgesia.34-36 However, use of elastomeric pumps has been associated with risk for AEs, including tissue necrosis, sloughing, wound infection, and chondrolysis.37-40 In addition, AEs related to “dose dumping” (accidental delivery of excessive doses) have been reported.40-44 Key issues that may negatively affect rehabilitation after orthopedic surgery include consistency and accuracy of analgesic delivery and the potential for motor block–induced muscle weakness, which may lead to falls and constrain ambulation.45-47
Liposome Bupivacaine
Description
Drug Delivery Technology. Liposome bupivacaine incorporates DepoFoam drug delivery technology (Pacira Pharmaceuticals, Inc.) to facilitate prolonged release of bupivacaine. This technology is based on creation of multivesicular liposome particles (diameter, 10-30 µm) with multiple aqueous chambers.30,48 After administration into the surgical site, bupivacaine diffuses from chambers in the liposomal particles over time, providing analgesia and reduced opioid requirements for up to 72 hours.29,30
Indication, Mechanism of Action, Pharmacokinetics, and Dose/Administration. Liposome bupivacaine is indicated for single-dose administration into the surgical site to produce postsurgical analgesia in patients at least 18 years old.14 Like other local anesthetics, liposome bupivacaine is thought to exert its pharmacologic effects by interacting with voltage-gated Na+ channels on neural membranes to raise the threshold for electrical excitability, to slow nerve impulse propagation, and to reduce the rate of rise of the action potential.14,49
Liposome bupivacaine has dose-proportional pharmacokinetics.50 Presence of a small amount of extra-liposomal bupivacaine in the formulation leads to a bimodal pharmacokinetic profile, with an initial peak serum concentration about 1 hour after administration, followed by a second peak within 12 to 36 hours (Figure).50
Maximum amount of liposome bupivacaine approved for single administration is 266 mg (packaged as 20 mL of a 1.3% solution). However, product labeling includes safety data associated with doses of 532 mg or less.14 The appropriate volume to be used should be based on the amount required to cover the surgical area. Liposome bupivacaine may be expanded with preservative-free normal (0.9%) sterile saline to a total volume of 300 mL: 20 mL liposome bupivacaine plus 280 mL or less diluent, with final concentration of 0.89 mg/mL (1:14 by volume).14
A 25-gauge or larger bore needle should be used to slowly inject liposome bupivacaine into soft tissues of the surgical site, with frequent aspiration to check for blood to minimize risk for intravascular injection.14 Total volume used and fraction injected in specific regions of the surgical site depend on the procedure. For example, a TKA study used 266 mg diluted to a total volume of 60 mL, with 8 mL infiltrated to the area around the medial capsule, 8 mL around the lateral capsule, 12 mL around the posterior capsule, 8 mL around the peripatellar area, 12 mL into the capsulotomy incision, and 12 mL into the subcutaneous tissue on each side of the incision.51
Efficacy
Multiple Surgical Settings. The efficacy of liposome bupivacaine, either alone or as a component of a multimodal analgesic regimen, has been evaluated in a series of 10 phase 2 and 3 studies (8 active-controlled, 2 placebo-controlled) involving 823 patients undergoing TKA, bunionectomy, hemorrhoidectomy, inguinal hernia repair, or mammoplasty.52 Patients received a single liposome bupivacaine dose ranging from 66 to 532 mg.52
Combined analyses of efficacy data from these studies found that liposome bupivacaine–based multimodal analgesic regimens produced postsurgical analgesia for up to 72 hours, increased time to first use of opioid rescue medication after surgery, and reduced total amount of postsurgical opioid consumption versus placebo.52
Compared with standard of care, liposome bupivacaine has been shown to provide effective analgesia in open-label studies in patients undergoing open colectomy,53 laparoscopic colectomy,54 and ileostomy reversal,55,56 as reflected in assessments of postsurgical opioid consumption, LOS, and hospital costs. It has also been studied when administered by infiltration into the transversus abdominis plane (TAP) in patients having laparoscopic prostatectomy and open abdominal hernia repair.57,58
Orthopedic Surgery. In a phase 2 randomized, double-blind, dose-ranging study, TKA patients (N = 138) received bupivacaine HCl 150 mg or liposome bupivacaine 133, 266, 399, or 532 mg administered by local infiltration into the capsulotomy incision and on either side of the incision before wound closure.51 Postsurgical rescue analgesia was available to all patients. Cumulative pain intensity scores with activity (primary efficacy measure) were not statistically different between liposome bupivacaine groups and the bupivacaine HCl group through postoperative day 4. Mean scores in the liposome bupivacaine 266-, 399-, and 532-mg groups were numerically lower than for those treated with bupivacaine HCl on postoperative days 2 to 5, with all doses of liposome bupivacaine having a statistically significant lower pain score at rest on day 5. There were no statistically significant differences across treatment groups with respect to total amount of postsurgical opioids used.
In a phase 3 randomized, double-blind study of TKA patients (N = 245), liposome bupivacaine 532 mg administered into the surgical site was compared with bupivacaine HCl 200 mg for postsurgical analgesia.52 Rescue analgesia was available to all patients. No statistically significant between-group differences were found with respect to postsurgical cumulative pain scores through 72 hours (primary efficacy endpoint).
In a single-center retrospective TKA study, postsurgical outcomes in a patient cohort that received intraoperative periarticular infiltration with liposome bupivacaine 266 mg (n = 65) were compared with a cohort that received infiltration with a combination of ropivacaine 400 mg, morphine 5 mg, and epinephrine 0.4 mg (n = 85).59 Patient-reported postsurgical pain scores were similar in the 2 treatment groups during the first 24 hours after surgery and at discharge. Mean (SD) pain scores during hospitalization after the first 24 hours until discharge were significantly (P = .04) higher in the liposome bupivacaine group, 4.9 (1.4), than in the periarticular group, 4.4 (1.6). There was no significant difference between the 2 treatment groups in postsurgical opioid use. The study demonstrated no advantage to using liposome bupivacaine injections with respect to pain relief, but it was a retrospective review in which pain scores were obtained from electronic medical records. It is essential that liposome bupivacaine be compared with intra-articular injections in well-designed randomized trials.
Another single-center, matched-cohort TKA study (N = 200) compared a liposome bupivacaine regimen with femoral nerve block.60 Compared with patients who received femoral nerve block, patients who received liposome bupivacaine reported lower pain intensity scores after surgery and had shorter LOS, reduced costs, and improved knee flexion at follow-up.60
Results from 2 other studies were presented at the 2014 meeting of the American Academy of Orthopaedic Surgeons (AAOS). One was a single-center, matched-cohort TKA study (N = 72) comparing infiltration of a single dose of liposome bupivacaine into the surgical site with continuous femoral nerve block.61 The 2 treatment groups had similar mean postsurgical pain intensity scores on a 0-to-10 visual analog scale, 1.8 for liposome bupivacaine and 2.3 for continuous nerve block (P = NS), but total amount of postsurgical opioids (hydrocodone-equivalent milligrams) was significantly (P < .0001) less in the liposome bupivacaine group (82 vs 177 mg).
The other study presented at the AAOS meeting was a larger, prospective case–control study comparing outcomes between 1000 patients who had total joint arthroplasty (TJA) with liposome bupivacaine and 1000 control patients who had TJA without liposome bupivacaine.62 For the control and liposome bupivacaine cohorts, respectively, mean postsurgical pain intensity scores were 2.41 and 1.98 (P < .0001), mean LOS was 2.83 days and 2.66 days (P < .02), and incidence of falls was 1.0% and 0.2% (P = .02). Average per-patient costs were $1246 lower in the liposome bupivacaine cohort.
A pivotal phase 3 placebo-controlled study compared liposome bupivacaine 106 mg with placebo in patients undergoing bunionectomy (N = 193).5 Rescue medication was available to all patients. Cumulative pain scores were significantly (P = .0005) lower in the liposome bupivacaine group (125) than in the placebo group (146) through 24 hours after surgery (primary efficacy measure) and significantly (P = .0229) lower (197 vs 220) through 36 hours. Median time to first use of rescue opioids was delayed in favor of the liposome bupivacaine group (7.2 vs 4.3 hours; P < .0001). Mean total number of opioid tablets used within 24 hours after surgery was also significantly lower (3.8 vs 4.7; P = .008), and a larger percentage of patients in the liposome bupivacaine group avoided opioid use altogether through 24 hours (7% vs 1%; P = .04).
Efficacy data for liposome bupivacaine appear promising for relief of pain after joint arthroplasty and other orthopedic procedures but have their limitations. First, no randomized trials have compared liposome bupivacaine with locally injected pain medications (intra-articular injections in TKA or hip arthroplasty). As these injections are quite common now, such analyses are essential. Second, cost-effectiveness studies are needed for orthopedic procedures. Third, most of the published studies were sponsored by the manufacturer of liposome bupivacaine—a situation that raises questions about potential bias. Non-industry-sponsored randomized trials assessing efficacy, safety, and cost-effectiveness are needed.
Safety
Local anesthetics, including liposome bupivacaine, have the potential for central nervous system (CNS) or cardiac toxicity resulting from excessive systemic absorption or inadvertent IV administration.63 However, reported serious CNS or cardiac-related AEs are rare.63,64
AE Profile. Safety data from 10 phase 2 and 3 studies involving 823 patients who received liposome bupivacaine were evaluated.65 Of these patients, 545 received a dose of 266 mg or less (maximum dose approved by the US Food and Drug Administration [FDA]). Liposome bupivacaine was generally well tolerated. Reported AE incidence was 62% (liposome bupivacaine), 75% (bupivacaine HCl), and 43% (placebo). More than 90% of reported AEs were mild or moderate. The most frequently reported AEs were nausea, constipation, and vomiting (liposome bupivacaine, bupivacaine HCl) and nausea, dizziness, and vomiting (placebo).
Serious AEs were reported in 22 (2.7%) of the 823 patients in the liposome bupivacaine group, 24 (5.4%) of the 446 in the bupivacaine HCl group, and 2 (1.1%) of the 190 in the placebo group.65 None of the serious AEs in the liposome bupivacaine and placebo groups were considered treatment-related. Six patients in the bupivacaine HCl group had treatment-related serious AEs (hypoglycemia, arthrofibrosis, hemarthrosis, joint swelling, scar, knee arthroplasty).
Cardiac Safety. Possible cardiac effects associated with liposome bupivacaine were evaluated with data from studies conducted during the clinical development program.66 One hundred thirty-eight patients participated in the phase 2 safety and efficacy study in TKA. In these patients, a consistent change in mean heart rate (range, +12.2 to +16.5 beats per minute) was found across all liposome bupivacaine doses and with bupivacaine HCl. No clinically relevant changes from baseline in mean electrocardiographic parameters, including QTcF interval (QT interval adjusted using Fridericia’s correction formula), were found. In another analysis,67 liposome bupivacaine administered in a single subcutaneous dose (266, 399, 532, or 665 mg) to healthy volunteers did not prolong (vs placebo) QTc interval.
Wound Healing. The potential effects of liposome bupivacaine on wound healing were evaluated with results from 10 phase 2 and 3 studies.68 The assessments, which varied across studies, included clinicians’ overall satisfaction with patient wound healing, wound status assessment (categories included erythema, drainage, edema, and induration), and wound scarring (categories included pigmentation, height, pliability, and vascularity). Clinician-assessed scores reflected high satisfaction with wound healing overall. There were few statistically significant differences in wound status assessments between liposome bupivacaine and the comparators and no statistically significant differences in scarring between liposome bupivacaine and bupivacaine HCl.
The potential of liposome bupivacaine to have adverse intra-articular effects was assessed with drainage samples from patients (n = 23) who had TKA and received liposome bupivacaine (133, 266, 399, or 532 mg) or bupivacaine HCl (150 mg) by wound infiltration near the intra-articular space.51,65 Only small amounts of bupivacaine were present in drainage fluid collected for 12 hours after liposome bupivacaine administration, comparable to bupivacaine HCl administration.65 Currently, the product is not approved for intra-articular use.
Compatibility With Diluents, Other Medications, and Implant Materials
Liposome bupivacaine may be expanded up to a ratio of 1:14 by volume (to a final total volume of 300 mL or a concentration of 0.89 mg/mL) using preservative-free normal (0.9%) sterile saline for injection.14 It has also been shown in vitro to be compatible with lactated Ringer solution as a diluent.69
Liposome bupivacaine should not be admixed with other medications before administration.14 No formal drug–drug interaction studies have been conducted with liposome bupivacaine, but it has been shown in vitro to be compatible with epinephrine solutions, with certain anti-infective medications (eg, bacitracin, gentamicin, cefazolin, cefuroxime), with certain analgesics (eg, ketorolac, morphine), with an antihypertensive medication (clonidine), with an antihemorrhagic medication (tranexamic acid), and with certain corticosteroids (eg, methylprednisolone, triamcinolone acetonide). These medications may be coadministered in the same location as liposome bupivacaine.69
Topical antiseptics (eg, povidone iodine) may be used in surgical procedures involving liposome bupivacaine as long as they are not directly mixed with liposome bupivacaine and are allowed to dry before it is administered. If a topical antiseptic is used for wound irrigation, the wound should be rinsed clear before liposome bupivacaine administration.14,69
Liposome bupivacaine may be coadministered into the same surgical site immediately after bupivacaine HCl as long as the dose ratio of liposome bupivacaine to bupivacaine HCl is 2:1 or higher. Because of the prolonged-release pharmacokinetic profile of liposome bupivacaine and the potential for increased bupivacaine exposure, bupivacaine HCl should not be administered within 96 hours after administration of liposome bupivacaine.14,69
In vitro coincubation studies of liposome bupivacaine and other local anesthetics, including ropivacaine, lidocaine, and mepivacaine, have found rapid release of free bupivacaine from the liposome matrix. Therefore, after giving any of these other local anesthetics, surgeons should wait at least 20 minutes before administering liposome bupivacaine into the same area.14,69
In vitro studies have shown that liposome bupivacaine is compatible with a wide range of commonly used implant materials, including polypropylene, expanded polytetrafluoroethylene, stainless steel, titanium, and smooth- and textured-type silicone.69
Investigational Use and Ongoing Studies
A phase 2 randomized, double-masked, dose-escalating/deescalating study was conducted to evaluate the efficacy, safety, and pharmacokinetics of liposome bupivacaine (155, 199, or 310 mg) in comparison with bupivacaine HCl 125 mg for ankle nerve block in patients undergoing bunionectomy (N = 58).70 The study medication was injected into 3 sites to reach the posterior tibial, sural, deep peroneal, superficial peroneal, and saphenous nerves. Pharmacokinetic exposure was higher for liposome bupivacaine than for bupivacaine HCl, as reflected by a significantly greater area under the curve, lower Cmax (maximum serum concentration), and longer mean half-life. Mean pain intensity scores were lower in the bupivacaine HCl group than in each liposome bupivacaine group the first 12 hours after surgery. However, the liposome bupivacaine 310-mg group had similar or lower scores than the bupivacaine HCl group from 12 to 96 hours after surgery. The most common AEs in the liposome bupivacaine group were gastrointestinal and not treatment-related.70
The efficacy and safety of liposome bupivacaine, administered as a femoral nerve block for postsurgical analgesia, were assessed in a phase 2/3 manufacturer-sponsored, placebo-controlled, multicenter, randomized, double-blind 2-part study (NCT01683071)71 in 280 TKA patients.71,72 Part 2 of the study, comparing liposome bupivacaine 266 mg (n = 116) and placebo (n = 116), met its primary endpoint, demonstrating statistical significance in favor of liposome bupivacaine for cumulative pain scores over 72 hours (P < .0001), with decreased opioid use (P < .05) and a safety profile similar to that of placebo.72
Other ongoing investigator-sponsored studies in orthopedic populations include comparisons of liposome bupivacaine and bupivacaine HCl for ultrasound-guided periarticular hip infiltration in hip arthroplasty (NTC01917191),73 as femoral nerve block in TKA (NCT01977339),74 and as interscalene brachial plexus block in arthroscopic shoulder surgery (NCT01977352).75 The primary efficacy outcome measure in these studies was postsurgical opioid use.73-75
Health Economics
A series of phase 4 health economics studies was conducted for gastrointestinal surgeries, including open colectomy, laparoscopic colectomy, and ileostomy reversal.53-56,76 These studies, of similar design, showed that a liposome bupivacaine–based multimodal analgesic regimen was associated with reduced opioid use, shorter hospital LOS, and lower hospitalization costs in comparison with a traditional opioid-based regimen.53-56 Although pooled analysis of these studies showed a cost savings of more than $2000 per patient and an LOS decrease of 1.4 days,76 all were conducted in the gastrointestinal surgery setting. Studies are needed to fully assess the economic benefits associated with liposome bupivacaine in the orthopedic surgery setting.
Conclusion
Liposome bupivacaine represents a potentially important contributor to multimodal analgesic regimens used to manage postsurgical pain. Liposome bupivacaine has demonstrated efficacy in providing prolonged postsurgical analgesia and reducing postsurgical opioid use in most surgical settings studied. Additional data from health economics studies in gastrointestinal surgery suggest liposome bupivacaine–based multimodal analgesic regimens may also contribute to reductions in hospital LOS and hospitalization costs. Non-industry-sponsored trials are needed to answer these crucial questions in orthopedic surgery settings. Nevertheless, data on the safety and efficacy of liposome bupivacaine for postsurgical analgesia continue to accumulate, and liposome bupivacaine appears to be a feasible therapeutic option for managing postsurgical pain in orthopedic surgery.
1. Centers for Disease Control and Prevention. Number of all-listed procedures for discharges from short-stay hospitals, by procedure category and age: United States, 2010. http://www.cdc.gov/nchs/data/nhds/4procedures/2010pro4_numberprocedureage.pdf. Accessed January 30, 2015.
2. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.
3. Bonica JJ. Postoperative pain. In: Bonica JJ, ed. The Management of Pain. Malvern, PA: Lea & Febiger; 1990:461-480.
4. Apfelbaum JL, Ashburn MA, Connis RT, et al; American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012;116(2):248-273.
5. Golf M, Daniels SE, Onel E. A phase 3, randomized, placebo-controlled trial of DepoFoam® bupivacaine (extended-release bupivacaine local analgesic) in bunionectomy. Adv Ther. 2011;28(9):776-788.
6. Oderda G. Challenges in the management of acute postsurgical pain. Pharmacotherapy. 2012;32(9 pt 2):6S-11S.
7. Oderda GM, Gan TJ, Johnson BH, Robinson SB. Effect of opioid-related adverse events on outcomes in selected surgical patients. J Pain Palliat Care Pharmacother. 2013;27(1):62-70.
8. Wu CL, Naqibuddin M, Rowlingson AJ, Lietman SA, Jermyn RM, Fleisher LA. The effect of pain on health-related quality of life in the immediate postoperative period. Anesth Analg. 2003;97(4):1078-1085.
9. Morrison RS, Magaziner J, McLaughlin MA, et al. The impact of post-operative pain on outcomes following hip fracture. Pain. 2003;103(3):303-311.
10. Capdevila X, Barthelet Y, Biboulet P, Ryckwaert Y, Rubenovitch J, d’Athis F. Effects of perioperative analgesic technique on the surgical outcome and duration of rehabilitation after major knee surgery. Anesthesiology. 1999;91(1):8-15.
11. Capdevila X, Dadure C, Bringuier S, et al. Effect of patient-controlled perineural analgesia on rehabilitation and pain after ambulatory orthopedic surgery: a multicenter randomized trial. Anesthesiology. 2006;105(3):566-573.
12. Ranawat AS, Ranawat CS. Pain management and accelerated rehabilitation for total hip and total knee arthroplasty. J Arthroplasty. 2007;22(7 Suppl 3):12-15.
13. Hamilton DF, Lane JV, Gaston P, et al. What determines patient satisfaction with surgery? A prospective cohort study of 4709 patients following total joint replacement. BMJ Open. 2013;3(4):e002525.
14. Exparel [prescribing information]. Parsippany, NJ: Pacira Pharmaceuticals, Inc.; 2014.
15. DeWeese FT, Akbari Z, Carline E. Pain control after knee arthroplasty: intraarticular versus epidural anesthesia. Clin Orthop Relat Res. 2001;392(11):226-231.
16. Pati AB, Perme D, Trail M, Henry PK, Bryan WJ. Rehabilitation parameters in total knee replacement patients undergoing epidural vs. conventional analgesia. J Orthop Sports Phys Ther. 1994;19(2):88-92.
17. Browne C, Copp S, Reden L, Pulido P, Colwell C Jr. Bupivacaine bolus injection versus placebo for pain management following total knee arthroplasty. J Arthroplasty. 2004;19(3):377-380.
18. Nechleba J, Rogers V, Cortina G, Cooney T. Continuous intra-articular infusion of bupivacaine for postoperative pain following total knee arthroplasty. J Knee Surg. 2005;18(3):197-202.
19. Campbell A, McCormick M, McKinlay K, Scott NB. Epidural vs. lumbar plexus infusions following total knee arthroplasty: randomized controlled trial. Eur J Anaesthesiol. 2008;25(6):502-507.
20. Serpell MG, Millar FA, Thomson MF. Comparison of lumbar plexus block versus conventional opioid analgesia after total knee replacement. Anaesthesia. 1991;46(4):275-277.
21. Lareau JM, Robbins CE, Talmo CT, Mehio AK, Puri L, Bono JV. Complications of femoral nerve blockade in total knee arthroplasty and strategies to reduce patient risk. J Arthroplasty. 2012;27(4):564-568.
22. Charous MT, Madison SJ, Suresh PJ, et al. Continuous femoral nerve blocks: varying local anesthetic delivery method (bolus versus basal) to minimize quadriceps motor block while maintaining sensory block. Anesthesiology. 2011;115(4):774-781.
23. Gottschalk A, Burmeister MA, Radtke P, et al. Continuous wound infiltration with ropivacaine reduces pain and analgesic requirement after shoulder surgery. Anesth Analg. 2003;97(4):1086-1091.
24. Kerr DR, Kohan L. Local infiltration analgesia: a technique for the control of acute postoperative pain following knee and hip surgery: a case study of 325 patients. Acta Orthop. 2008;79(2):174-183.
25. Dalury DF, Lieberman JR, MacDonald SJ. Current and innovative pain management techniques in total knee arthroplasty. J Bone Joint Surg Am. 2011;93(20):1938-1943.
26. White PF. The role of non-opioid analgesic techniques in the management of pain after ambulatory surgery. Anesth Analg. 2002;94(3):577-585.
27. Dahl JB, Kehlet H. Non-steroidal anti-inflammatory drugs: rationale for use in severe postoperative pain. Br J Anaesth. 1991;66(6):703-712.
28. Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048-1056.
29. Candiotti K. Liposomal bupivacaine: an innovative nonopioid local analgesic for the management of postsurgical pain. Pharmacotherapy. 2012;32(9 Pt 2):19S-26S.
30. Bergese SD, Onel E, Portillo J. Evaluation of DepoFoam® bupivacaine for the treatment of postsurgical pain. Pain Manag. 2011;1(6):539-547.
31. Joranson DE, Ryan KM, Gilson AM, Dahl JL. Trends in medical use and abuse of opioid analgesics. JAMA. 2000;283(13):1710-1714.
32. Kuehn BM. Opioid prescriptions soar: increase in legitimate use as well as abuse. JAMA. 2007;297(3):249-251.
33. Compton WM, Volkow ND. Major increases in opioid analgesic abuse in the United States: concerns and strategies. Drug Alcohol Depend. 2006;81(2):103-107.
34. Ilfeld BM, Morey TE, Enneking FK. Delivery rate accuracy of portable, bolus-capable infusion pumps used for patient-controlled continuous regional analgesia. Reg Anesth Pain Med. 2003;28(1):17-23.
35. Ganapathy S, Amendola A, Lichfield R, Fowler PJ, Ling E. Elastomeric pumps for ambulatory patient controlled regional analgesia. Can J Anaesth. 2000;47(9):897-902.
36. Bray DA Jr, Nguyen J, Craig J, Cohen BE, Collins DR Jr. Efficacy of a local anesthetic pain pump in abdominoplasty. Plast Reconstr Surg. 2007;119(3):1054-1059.
37. Brown SL, Morrison AE. Local anesthetic infusion pump systems adverse events reported to the Food and Drug Administration. Anesthesiology. 2004;100(5):1305-1307.
38. Noyes FR, Fleckenstein CM, Barber-Westin SD. The development of postoperative knee chondrolysis after intra-articular pain pump infusion of an anesthetic medication: a series of twenty-one cases. J Bone Joint Surg Am. 2012;94(16):1448-1457.
39. Rapley JH, Beavis RC, Barber FA. Glenohumeral chondrolysis after shoulder arthroscopy associated with continuous bupivacaine infusion. Arthroscopy. 2009;25(12):1367-1373.
40. Institute for Safe Medication Practices. Process for handling elastomeric pain relief balls (ON-Q PainBuster and others) requires safety improvements. ISMP Medication Safety Alert. http://www.ismp.org/Newsletters/acutecare/articles/20090716.asp. Accessed January 30, 2015.
41. Pepin JL, Dasta JF, New M. Ensuring safe and economical use of elastomeric infusion devices. Am J Health Syst Pharm. 2011;68(24):2330-2331.
42. Birrer KL, Anderson RL, Liu-DeRyke X, Patel KR. Measures to improve safety of an elastomeric infusion system for pain management. Am J Health Syst Pharm. 2011;68(13):1251-1255.
43. Ilfeld BM, Enneking FK. Continuous peripheral nerve blocks at home: a review. Anesth Analg. 2005;100(6):1822-1833.
44. US Food and Drug Administration. Medical device recalls: I-Flow ON-Q Pump with ONDEMAND Bolus Button. http://www.fda.gov/MedicalDevices/Safety/ListofRecalls/ucm317826.htm. Accessed July 15, 2014.
45. Ilfeld BM, Morey TE, Enneking FK. Portable infusion pumps used for continuous regional analgesia: delivery rate accuracy and consistency. Reg Anesth Pain Med. 2003;28(5):424-432.
46. Ganapathy S. Wound/intra-articular infiltration or peripheral nerve blocks for orthopedic joint surgery: efficacy and safety issues. Curr Opin Anaesthesiol. 2012;25(5):615-620.
47. Ilfeld BM, Duke KB, Donohue MC. The association between lower extremity continuous peripheral nerve blocks and patient falls after knee and hip arthroplasty. Anesth Analg. 2010;111(6):1552-1554.
48. Angst MS, Drover DR. Pharmacology of drugs formulated with DepoFoam™: a sustained release drug delivery system for parenteral administration using multivesicular liposome technology. Clin Pharmacokinet. 2006;45(12):1153-1176.
49. Catterall WA, Mackie K. Local anesthetics. In: Gutstein HB, Akil H, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York, NY: McGraw-Hill; 2011:565-582.
50. Hu D, Onel E, Singla N, Kramer WG, Hadzic A. Pharmacokinetic profile of liposome bupivacaine injection following a single administration at the surgical site. Clin Drug Investig. 2013;33(2):109-115.
51. Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536.
52. Bergese SD, Ramamoorthy S, Patou G, Bramlett K, Gorfine SR, Candiotti KA. Efficacy profile of liposome bupivacaine, a novel formulation of bupivacaine for postsurgical analgesia. J Pain Res. 2012;5:107-116.
53. Cohen SM. Extended pain relief trial utilizing infiltration of Exparel®, a long-acting multivesicular liposome formulation of bupivacaine: a phase IV health economic trial in adult patients undergoing open colectomy. J Pain Res. 2012;5:567-572.
54. Candiotti KA, Sands LR, Lee E, et al. Liposome bupivacaine for postsurgical analgesia in adult patients undergoing laparoscopic colectomy: results from prospective phase IV sequential cohort studies assessing health economic outcomes. Curr Ther Res. 2014;76:1-6.
55. Marcet JE, Nfonsam VN, Larach S. An extended paIn relief trial utilizing the infiltration of a long-acting Multivesicular liPosome foRmulation Of bupiVacaine, EXPAREL (IMPROVE): a Phase IV health economic trial in adult patients undergoing ileostomy reversal. J Pain Res. 2013;6:549-555.
56. Vogel JD. Liposome bupivacaine (EXPAREL®) for extended pain relief in patients undergoing ileostomy reversal at a single institution with a fast-track discharge protocol: an IMPROVE phase IV health economics trial. J Pain Res. 2013;6:605-610.
57. Sternlicht A, Shapiro M, Robelen G, Vellayappan U, Tuerk IA. Initial findings using EXPAREL® (bupivacaine liposome injectable suspension) via infiltration into the transversus abdominis plane (TAP) for postsurgical analgesia in robotic prostatectomy (RP). Abstract presented at: Annual Fall Pain Meeting and Workshops of the American Society of Regional Anesthesia and Pain Medicine; November 15-18, 2012; Miami Beach, FL.
58. Feierman DE, Kronenfeld M, Gupta PM, Younger N, Logvinskiy E. Evaluation of Exparel® use via infiltration into the transversus abdominis plane for prolonged postoperative analgesia in subjects undergoing open abdominal hernia repair. Poster presented at: Annual Meeting of the International Anesthesia Research Society; May 4-7, 2013; San Diego, CA.
59. Bagsby DT, Ireland PH, Meneghini RM. Liposomal bupivacaine versus traditional periarticular injection for pain control after total knee arthroplasty. J Arthroplasty. 2014;29(8):1687-1690.
60. Broome B, Backlund I. Rapid recovery pain pathway for total knee arthroplasty results in improved pain management, decreased length of stay, and significant cost savings. Poster presented at: Annual Orthopedic and Spine Summit; September 18-20, 2013; San Antonio, TX.
61. Emerson RH, Barrington JW. Comparison of infiltration with long-acting bupivacaine to a femoral nerve catheter for total knee replacement. Abstract presented at: Annual Meeting of the American Academy of Orthopaedic Surgeons; March 11-15, 2014; New Orleans, LA. Abstract P124.
62. Barrington JW. Emerging data in the use of liposome bupivacaine: comparative review in 2,000 TJA patients. Oral presentation presented at: Annual Meeting of the American Academy of Orthopaedic Surgeons; March 11-15, 2014; New Orleans, LA.
63. Neal JM, Bernards CM, Butterworth JF, et al. ASRA practice advisory on local anesthetic systemic toxicity. Reg Anesth Pain Med. 2010;35(2):152-161.
64. D’Angelo R. Are the new local anesthetics worth their cost? Acta Anaesthesiol Scand. 2000;44(6):639-641.
65. Viscusi ER, Sinatra R, Onel E, Ramamoorthy SL. The safety of liposome bupivacaine, a novel local analgesic formulation. Clin J Pain. 2014;30(2):102-110.
66. Bergese SD, Onel E, Morren M, Morganroth J. Bupivacaine extended-release liposome injection exhibits a favorable cardiac safety profile. Reg Anesth Pain Med. 2012;37(2):145-151.
67. Naseem A, Harada T, Wang D, et al. Bupivacaine extended release liposome injection does not prolong QTc interval in a thorough QT/QTc study in healthy volunteers. J Clin Pharmacol. 2012;52(9):1441-1447.
68. Baxter R, Bramlett K, Onel E, Daniels S. Impact of local administration of liposome bupivacaine for postsurgical analgesia on wound healing: a review of data from ten prospective, controlled clinical studies. Clin Ther. 2013;35(3):312-320.
69. Kharitonov V. A review of the compatibility of liposome bupivacaine with other drug products and commonly used implant materials. Postgrad Med. 2014;126(1):129-138.
70. Ilfeld BM. Liposome bupivacaine in peripheral nerve blocks and epidural injections to manage postoperative pain. Expert Opin Pharmacother. 2013;14(17):2421-2431.
71. Femoral nerve block with liposome bupivacaine for postsurgical analgesia following total knee arthroplasty [NCT01683071]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01683071?term=NCT01683071%5C&rank=1. Accessed January 30, 2015.
72. Minkowitz H, Matthews A, Puckett C, Melson T. Liposome bupivacaine in femoral nerve block: initial results from a phase 2/3 pivotal study. Poster presented at: Annual Meeting of the American Society of Regional Anesthesia and Pain Medicine; April 3-6, 2014; Chicago, IL.
73. Ultrasound guided local infiltration analgesia for hip arthroscopy [NCT01907191]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01907191?term=NCT01907191&rank=1. Accessed January 30, 2015.
74. Efficacy of single injection femoral nerve block with liposomal bupivacaine for total knee arthroplasty [NCT01977339]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01977339?term=NCT01977339&rank=1. Accessed January 30, 2015.
75. Efficacy of interscalene brachial plexus block with liposomal bupivacaine for arthroscopic shoulder surgery [NCT01977352]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01977352?term=NCT01977352&rank=1. Accessed January 30, 2015.
76. Cohen SM, Vogel JD, Marcet JE, Candiotti K. Liposome bupivacaine for improvement in economic outcomes and opioid burden in GI surgery: IMPROVE study pooled analysis. J Pain Res. 2014;7:359-366.
1. Centers for Disease Control and Prevention. Number of all-listed procedures for discharges from short-stay hospitals, by procedure category and age: United States, 2010. http://www.cdc.gov/nchs/data/nhds/4procedures/2010pro4_numberprocedureage.pdf. Accessed January 30, 2015.
2. Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007;89(4):780-785.
3. Bonica JJ. Postoperative pain. In: Bonica JJ, ed. The Management of Pain. Malvern, PA: Lea & Febiger; 1990:461-480.
4. Apfelbaum JL, Ashburn MA, Connis RT, et al; American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012;116(2):248-273.
5. Golf M, Daniels SE, Onel E. A phase 3, randomized, placebo-controlled trial of DepoFoam® bupivacaine (extended-release bupivacaine local analgesic) in bunionectomy. Adv Ther. 2011;28(9):776-788.
6. Oderda G. Challenges in the management of acute postsurgical pain. Pharmacotherapy. 2012;32(9 pt 2):6S-11S.
7. Oderda GM, Gan TJ, Johnson BH, Robinson SB. Effect of opioid-related adverse events on outcomes in selected surgical patients. J Pain Palliat Care Pharmacother. 2013;27(1):62-70.
8. Wu CL, Naqibuddin M, Rowlingson AJ, Lietman SA, Jermyn RM, Fleisher LA. The effect of pain on health-related quality of life in the immediate postoperative period. Anesth Analg. 2003;97(4):1078-1085.
9. Morrison RS, Magaziner J, McLaughlin MA, et al. The impact of post-operative pain on outcomes following hip fracture. Pain. 2003;103(3):303-311.
10. Capdevila X, Barthelet Y, Biboulet P, Ryckwaert Y, Rubenovitch J, d’Athis F. Effects of perioperative analgesic technique on the surgical outcome and duration of rehabilitation after major knee surgery. Anesthesiology. 1999;91(1):8-15.
11. Capdevila X, Dadure C, Bringuier S, et al. Effect of patient-controlled perineural analgesia on rehabilitation and pain after ambulatory orthopedic surgery: a multicenter randomized trial. Anesthesiology. 2006;105(3):566-573.
12. Ranawat AS, Ranawat CS. Pain management and accelerated rehabilitation for total hip and total knee arthroplasty. J Arthroplasty. 2007;22(7 Suppl 3):12-15.
13. Hamilton DF, Lane JV, Gaston P, et al. What determines patient satisfaction with surgery? A prospective cohort study of 4709 patients following total joint replacement. BMJ Open. 2013;3(4):e002525.
14. Exparel [prescribing information]. Parsippany, NJ: Pacira Pharmaceuticals, Inc.; 2014.
15. DeWeese FT, Akbari Z, Carline E. Pain control after knee arthroplasty: intraarticular versus epidural anesthesia. Clin Orthop Relat Res. 2001;392(11):226-231.
16. Pati AB, Perme D, Trail M, Henry PK, Bryan WJ. Rehabilitation parameters in total knee replacement patients undergoing epidural vs. conventional analgesia. J Orthop Sports Phys Ther. 1994;19(2):88-92.
17. Browne C, Copp S, Reden L, Pulido P, Colwell C Jr. Bupivacaine bolus injection versus placebo for pain management following total knee arthroplasty. J Arthroplasty. 2004;19(3):377-380.
18. Nechleba J, Rogers V, Cortina G, Cooney T. Continuous intra-articular infusion of bupivacaine for postoperative pain following total knee arthroplasty. J Knee Surg. 2005;18(3):197-202.
19. Campbell A, McCormick M, McKinlay K, Scott NB. Epidural vs. lumbar plexus infusions following total knee arthroplasty: randomized controlled trial. Eur J Anaesthesiol. 2008;25(6):502-507.
20. Serpell MG, Millar FA, Thomson MF. Comparison of lumbar plexus block versus conventional opioid analgesia after total knee replacement. Anaesthesia. 1991;46(4):275-277.
21. Lareau JM, Robbins CE, Talmo CT, Mehio AK, Puri L, Bono JV. Complications of femoral nerve blockade in total knee arthroplasty and strategies to reduce patient risk. J Arthroplasty. 2012;27(4):564-568.
22. Charous MT, Madison SJ, Suresh PJ, et al. Continuous femoral nerve blocks: varying local anesthetic delivery method (bolus versus basal) to minimize quadriceps motor block while maintaining sensory block. Anesthesiology. 2011;115(4):774-781.
23. Gottschalk A, Burmeister MA, Radtke P, et al. Continuous wound infiltration with ropivacaine reduces pain and analgesic requirement after shoulder surgery. Anesth Analg. 2003;97(4):1086-1091.
24. Kerr DR, Kohan L. Local infiltration analgesia: a technique for the control of acute postoperative pain following knee and hip surgery: a case study of 325 patients. Acta Orthop. 2008;79(2):174-183.
25. Dalury DF, Lieberman JR, MacDonald SJ. Current and innovative pain management techniques in total knee arthroplasty. J Bone Joint Surg Am. 2011;93(20):1938-1943.
26. White PF. The role of non-opioid analgesic techniques in the management of pain after ambulatory surgery. Anesth Analg. 2002;94(3):577-585.
27. Dahl JB, Kehlet H. Non-steroidal anti-inflammatory drugs: rationale for use in severe postoperative pain. Br J Anaesth. 1991;66(6):703-712.
28. Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. 1993;77(5):1048-1056.
29. Candiotti K. Liposomal bupivacaine: an innovative nonopioid local analgesic for the management of postsurgical pain. Pharmacotherapy. 2012;32(9 Pt 2):19S-26S.
30. Bergese SD, Onel E, Portillo J. Evaluation of DepoFoam® bupivacaine for the treatment of postsurgical pain. Pain Manag. 2011;1(6):539-547.
31. Joranson DE, Ryan KM, Gilson AM, Dahl JL. Trends in medical use and abuse of opioid analgesics. JAMA. 2000;283(13):1710-1714.
32. Kuehn BM. Opioid prescriptions soar: increase in legitimate use as well as abuse. JAMA. 2007;297(3):249-251.
33. Compton WM, Volkow ND. Major increases in opioid analgesic abuse in the United States: concerns and strategies. Drug Alcohol Depend. 2006;81(2):103-107.
34. Ilfeld BM, Morey TE, Enneking FK. Delivery rate accuracy of portable, bolus-capable infusion pumps used for patient-controlled continuous regional analgesia. Reg Anesth Pain Med. 2003;28(1):17-23.
35. Ganapathy S, Amendola A, Lichfield R, Fowler PJ, Ling E. Elastomeric pumps for ambulatory patient controlled regional analgesia. Can J Anaesth. 2000;47(9):897-902.
36. Bray DA Jr, Nguyen J, Craig J, Cohen BE, Collins DR Jr. Efficacy of a local anesthetic pain pump in abdominoplasty. Plast Reconstr Surg. 2007;119(3):1054-1059.
37. Brown SL, Morrison AE. Local anesthetic infusion pump systems adverse events reported to the Food and Drug Administration. Anesthesiology. 2004;100(5):1305-1307.
38. Noyes FR, Fleckenstein CM, Barber-Westin SD. The development of postoperative knee chondrolysis after intra-articular pain pump infusion of an anesthetic medication: a series of twenty-one cases. J Bone Joint Surg Am. 2012;94(16):1448-1457.
39. Rapley JH, Beavis RC, Barber FA. Glenohumeral chondrolysis after shoulder arthroscopy associated with continuous bupivacaine infusion. Arthroscopy. 2009;25(12):1367-1373.
40. Institute for Safe Medication Practices. Process for handling elastomeric pain relief balls (ON-Q PainBuster and others) requires safety improvements. ISMP Medication Safety Alert. http://www.ismp.org/Newsletters/acutecare/articles/20090716.asp. Accessed January 30, 2015.
41. Pepin JL, Dasta JF, New M. Ensuring safe and economical use of elastomeric infusion devices. Am J Health Syst Pharm. 2011;68(24):2330-2331.
42. Birrer KL, Anderson RL, Liu-DeRyke X, Patel KR. Measures to improve safety of an elastomeric infusion system for pain management. Am J Health Syst Pharm. 2011;68(13):1251-1255.
43. Ilfeld BM, Enneking FK. Continuous peripheral nerve blocks at home: a review. Anesth Analg. 2005;100(6):1822-1833.
44. US Food and Drug Administration. Medical device recalls: I-Flow ON-Q Pump with ONDEMAND Bolus Button. http://www.fda.gov/MedicalDevices/Safety/ListofRecalls/ucm317826.htm. Accessed July 15, 2014.
45. Ilfeld BM, Morey TE, Enneking FK. Portable infusion pumps used for continuous regional analgesia: delivery rate accuracy and consistency. Reg Anesth Pain Med. 2003;28(5):424-432.
46. Ganapathy S. Wound/intra-articular infiltration or peripheral nerve blocks for orthopedic joint surgery: efficacy and safety issues. Curr Opin Anaesthesiol. 2012;25(5):615-620.
47. Ilfeld BM, Duke KB, Donohue MC. The association between lower extremity continuous peripheral nerve blocks and patient falls after knee and hip arthroplasty. Anesth Analg. 2010;111(6):1552-1554.
48. Angst MS, Drover DR. Pharmacology of drugs formulated with DepoFoam™: a sustained release drug delivery system for parenteral administration using multivesicular liposome technology. Clin Pharmacokinet. 2006;45(12):1153-1176.
49. Catterall WA, Mackie K. Local anesthetics. In: Gutstein HB, Akil H, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 12th ed. New York, NY: McGraw-Hill; 2011:565-582.
50. Hu D, Onel E, Singla N, Kramer WG, Hadzic A. Pharmacokinetic profile of liposome bupivacaine injection following a single administration at the surgical site. Clin Drug Investig. 2013;33(2):109-115.
51. Bramlett K, Onel E, Viscusi ER, Jones K. A randomized, double-blind, dose-ranging study comparing wound infiltration of DepoFoam bupivacaine, an extended-release liposomal bupivacaine, to bupivacaine HCl for postsurgical analgesia in total knee arthroplasty. Knee. 2012;19(5):530-536.
52. Bergese SD, Ramamoorthy S, Patou G, Bramlett K, Gorfine SR, Candiotti KA. Efficacy profile of liposome bupivacaine, a novel formulation of bupivacaine for postsurgical analgesia. J Pain Res. 2012;5:107-116.
53. Cohen SM. Extended pain relief trial utilizing infiltration of Exparel®, a long-acting multivesicular liposome formulation of bupivacaine: a phase IV health economic trial in adult patients undergoing open colectomy. J Pain Res. 2012;5:567-572.
54. Candiotti KA, Sands LR, Lee E, et al. Liposome bupivacaine for postsurgical analgesia in adult patients undergoing laparoscopic colectomy: results from prospective phase IV sequential cohort studies assessing health economic outcomes. Curr Ther Res. 2014;76:1-6.
55. Marcet JE, Nfonsam VN, Larach S. An extended paIn relief trial utilizing the infiltration of a long-acting Multivesicular liPosome foRmulation Of bupiVacaine, EXPAREL (IMPROVE): a Phase IV health economic trial in adult patients undergoing ileostomy reversal. J Pain Res. 2013;6:549-555.
56. Vogel JD. Liposome bupivacaine (EXPAREL®) for extended pain relief in patients undergoing ileostomy reversal at a single institution with a fast-track discharge protocol: an IMPROVE phase IV health economics trial. J Pain Res. 2013;6:605-610.
57. Sternlicht A, Shapiro M, Robelen G, Vellayappan U, Tuerk IA. Initial findings using EXPAREL® (bupivacaine liposome injectable suspension) via infiltration into the transversus abdominis plane (TAP) for postsurgical analgesia in robotic prostatectomy (RP). Abstract presented at: Annual Fall Pain Meeting and Workshops of the American Society of Regional Anesthesia and Pain Medicine; November 15-18, 2012; Miami Beach, FL.
58. Feierman DE, Kronenfeld M, Gupta PM, Younger N, Logvinskiy E. Evaluation of Exparel® use via infiltration into the transversus abdominis plane for prolonged postoperative analgesia in subjects undergoing open abdominal hernia repair. Poster presented at: Annual Meeting of the International Anesthesia Research Society; May 4-7, 2013; San Diego, CA.
59. Bagsby DT, Ireland PH, Meneghini RM. Liposomal bupivacaine versus traditional periarticular injection for pain control after total knee arthroplasty. J Arthroplasty. 2014;29(8):1687-1690.
60. Broome B, Backlund I. Rapid recovery pain pathway for total knee arthroplasty results in improved pain management, decreased length of stay, and significant cost savings. Poster presented at: Annual Orthopedic and Spine Summit; September 18-20, 2013; San Antonio, TX.
61. Emerson RH, Barrington JW. Comparison of infiltration with long-acting bupivacaine to a femoral nerve catheter for total knee replacement. Abstract presented at: Annual Meeting of the American Academy of Orthopaedic Surgeons; March 11-15, 2014; New Orleans, LA. Abstract P124.
62. Barrington JW. Emerging data in the use of liposome bupivacaine: comparative review in 2,000 TJA patients. Oral presentation presented at: Annual Meeting of the American Academy of Orthopaedic Surgeons; March 11-15, 2014; New Orleans, LA.
63. Neal JM, Bernards CM, Butterworth JF, et al. ASRA practice advisory on local anesthetic systemic toxicity. Reg Anesth Pain Med. 2010;35(2):152-161.
64. D’Angelo R. Are the new local anesthetics worth their cost? Acta Anaesthesiol Scand. 2000;44(6):639-641.
65. Viscusi ER, Sinatra R, Onel E, Ramamoorthy SL. The safety of liposome bupivacaine, a novel local analgesic formulation. Clin J Pain. 2014;30(2):102-110.
66. Bergese SD, Onel E, Morren M, Morganroth J. Bupivacaine extended-release liposome injection exhibits a favorable cardiac safety profile. Reg Anesth Pain Med. 2012;37(2):145-151.
67. Naseem A, Harada T, Wang D, et al. Bupivacaine extended release liposome injection does not prolong QTc interval in a thorough QT/QTc study in healthy volunteers. J Clin Pharmacol. 2012;52(9):1441-1447.
68. Baxter R, Bramlett K, Onel E, Daniels S. Impact of local administration of liposome bupivacaine for postsurgical analgesia on wound healing: a review of data from ten prospective, controlled clinical studies. Clin Ther. 2013;35(3):312-320.
69. Kharitonov V. A review of the compatibility of liposome bupivacaine with other drug products and commonly used implant materials. Postgrad Med. 2014;126(1):129-138.
70. Ilfeld BM. Liposome bupivacaine in peripheral nerve blocks and epidural injections to manage postoperative pain. Expert Opin Pharmacother. 2013;14(17):2421-2431.
71. Femoral nerve block with liposome bupivacaine for postsurgical analgesia following total knee arthroplasty [NCT01683071]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01683071?term=NCT01683071%5C&rank=1. Accessed January 30, 2015.
72. Minkowitz H, Matthews A, Puckett C, Melson T. Liposome bupivacaine in femoral nerve block: initial results from a phase 2/3 pivotal study. Poster presented at: Annual Meeting of the American Society of Regional Anesthesia and Pain Medicine; April 3-6, 2014; Chicago, IL.
73. Ultrasound guided local infiltration analgesia for hip arthroscopy [NCT01907191]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01907191?term=NCT01907191&rank=1. Accessed January 30, 2015.
74. Efficacy of single injection femoral nerve block with liposomal bupivacaine for total knee arthroplasty [NCT01977339]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01977339?term=NCT01977339&rank=1. Accessed January 30, 2015.
75. Efficacy of interscalene brachial plexus block with liposomal bupivacaine for arthroscopic shoulder surgery [NCT01977352]. ClinicalTrials.gov website. http://clinicaltrials.gov/ct2/show/NCT01977352?term=NCT01977352&rank=1. Accessed January 30, 2015.
76. Cohen SM, Vogel JD, Marcet JE, Candiotti K. Liposome bupivacaine for improvement in economic outcomes and opioid burden in GI surgery: IMPROVE study pooled analysis. J Pain Res. 2014;7:359-366.
Use of Cross-Leg Flap for Wound Complications Resulting From Open Pilon Fracture
Soft-tissue complications are a known problem in the treatment of pilon fractures of the distal end of the tibia. These fractures typically occur as the result of a high-energy mechanism, and axial load and shear forces often lead to a severe soft-tissue injury. In many cases, these injuries may require additional procedures to provide adequate soft-tissue coverage. These procedures can include use of either a rotational muscle flap or a free flap transfer. In some cases, however, these flaps are not possible secondary to vascular compromise.
In this article, we report the case of a pilon fracture combined with severe soft-tissue injury and vascular compromise of the leg. A cross-leg fasciocutaneous flap was performed as a salvage procedure for coverage of the soft-tissue defect. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 23-year-old man sustained a left grade III open pilon fracture after a fall off a cherry picker. He was initially treated with irrigation and débridement of the open anteromedial wound, wound closure, application of external fixation, and open reduction and internal fixation (ORIF) of the concomitant comminuted fibular fracture. Operative fixation of the pilon was performed 3 weeks after injury, once skin and soft tissues were in acceptable condition (Figure 1). Skin closure was performed with 2-0 Vicryl sutures (Ethicon, Inc, Somerville, New Jersey) followed by 3-0 nylon skin sutures and No. 2 nylon retention sutures to reduce tension at the incision.
On postoperative day 17, the patient was found to have skin necrosis with exposed hardware over the medial laceration that had resulted from the open fracture (Figure 2). The wound measured 7×6 cm. The plastic surgery team was consulted, and a soft-tissue flap was recommended. Preoperative computed tomography angiogram (Figure 3) revealed 1 vessel runoff in the leg, constituting the peroneal artery, and a conventional angiogram confirmed this finding (Figure 4). Despite these findings, the patient was taken to the operating room 4 weeks after initial injury to try to find a vessel compatible with anastomosis. Intraoperative wound exploration confirmed no patent blood supply for local soft-tissue flap coverage. Therefore, the wound was irrigated and débrided, and a vacuum-assisted closure (VAC) dressing was applied despite exposed hardware and bone. A decision was then made to attempt a cross-leg flap as a salvage procedure, and VAC dressing therapy was continued for several weeks to prepare the recipient site (Figure 5).
Seven weeks after injury, the patient was taken to the operating room by the orthopedic surgery and plastic surgery teams. After débridement, a fasciocutaneous flap was raised from the middle third of the contralateral leg (Figure 6) based on a posterior tibial artery perforator. The flap, which measured 7×7 cm (sufficient to cover the defect), was raised from lateral to medial from the posterior aspect of the leg with the pedicle located on the medial aspect of the right leg. Flap placement was facilitated by flexing the left knee to 80°. The flap was sutured into place with 4-0 Vicryl deep sutures followed by 4-0 nylon and superficial sutures in an interrupted fashion (Figure 7). Rigid external fixation was then applied to both extremities, bridging them together in optimal position (Figure 8). This construct included 2 short bars that would elevate the patient’s heels off the bed to reduce the chance of heel decubiti. Although including the feet in the external fixator construct may help prevent equinus contracture, we splinted the ankles in neutral position immediately after surgery so that we could begin early range-of-motion (ROM) exercises of the ankles to prevent stiffness. Ankle ROM exercises were started once the flap incorporated, 3 weeks after placement of the external fixator. Lacking medical insurance coverage, the patient could not be admitted to a rehabilitation facility or receive home care. He lived independently and had no help at home, so he had to remain hospitalized after placement of the external fixator. While hospitalized, the surgical site was treated with frequent dressing changes, including use of bacitracin and nonadherent dressing.
After flap coverage and 4 weeks of bed rest, a base clamping test confirmed the flap was incorporated into the recipient bed. The patient was then returned to the operating room for removal of the external fixator and skin grafting of the donor site. After surgery, he was started on physical therapy, including exercises for bilateral hip, knee, and ankle ROM and strengthening of the lower extremities. Four months after initial injury, the fracture was healed, based on bone consolidation, seen on radiographs, that is consistent with other pilon fractures treated at our institution. Six months after external fixator removal, the patient was able to ambulate independently with minimal discomfort (Figure 9). Passive and active ankle ROM was 20° of dorsiflexion and 25° of plantarflexion, compared with 25° of dorsiflexion and 45° of plantarflexion on the contralateral extremity. Subtalar motion had some stiffness with a 10° arc, compared with a 25° arc on the contralateral extremity. On simple manual testing, the patient had 5/5 motor strength with dorsiflexion, plantarflexion, inversion, and eversion. He returned to full duty as a landscaper about 1 year after initial injury and had no recurrence of wound complications or infection.
Discussion
Fractures of the distal tibia are commonly known as pilon or plafond fractures. They represent up to 10% of all tibial fractures. The injury consists of an intra-articular fracture of the tibiotalar joint with varying degrees of proximal extension into the tibial metaphysis. The etiology is an axial load on the tibia with or without a rotational force.1 Treatment is challenging. The literature includes many reports of wound and soft-tissue complications after ORIF. In 1969, Rüedi and Allgöwer2 published recommendations that have become the standard for treatment of pilon fractures. Twelve percent of the 84 fractures included in their study were associated with wound complications. In 2004, Sirkin and colleagues3 suggested that wound problems associated with ORIF of pilon fractures may be caused by attempts at immediate fixation through swollen soft tissue. They postulated that staging the procedure and waiting for decreased soft-tissue swelling may reduce the incidence of wound complications. In their series, only 2.9% of closed pilon fractures and only 9.1% of open fractures had any wound complications, and none of their patients required skin grafts, rotation flaps, or free tissue transfers.
However, soft-tissue complications still remain a significant threat in the treatment of pilon fracture, and cases that require additional procedures for soft-tissue coverage are common. In some cases, wound necrosis may lead to below-knee amputation.4 There are several coverage options, including local rotational flaps using the soleus muscle5,6 as well as free flaps using the latissimus dorsi, gracilis, or rectus abdominis muscles.7 These options require a sufficient blood supply to the region.
Many high-energy pilon fractures may be associated with vascular injury, and therefore flap survival may be compromised. We have reported such a case in the present article. Our patient’s preoperative angiogram indicated he had 1-vessel runoff to the distal leg—a situation incompatible with free tissue transfer. It is not clear whether this finding is secondary to trauma to the leg or is caused by an anatomical anomaly. Nevertheless, the poor vascularity posed a challenge to providing soft-tissue coverage. Cross-finger8 and cross-foot9 flaps have been described in upper and lower extremity injuries. In 2006, Zhao and colleagues10 reported on 5 patients with tibia and/or hardware exposure after operative fixation of tibia fractures. These patients had poor local soft tissue around the wound and therefore underwent cross-leg flap for coverage. It is not clear where the soft-tissue defects were located and whether any studies were performed to assess the local blood flow.
From our patient’s case, we learned that multiple factors should be considered when assessing such high-energy injuries. First, respecting the soft tissues is of paramount importance. Our initial management on presentation consisted of irrigation and débridement of the wound, fixation of the fibula, and application of an external fixator to allow for soft-tissue healing before definitive fixation of the pilon. Although ultimately the patient required soft-tissue coverage, soft-tissue healing and viability are important in preventing unnecessary soft-tissue procedures, and therefore we would not have handled our initial treatment differently.
Patient selection is also important. The ideal candidate for a cross-leg flap is a young, healthy person who is compliant and has a strong support system to help with activities of daily living. Unfortunately, because of financial issues and lack of home support, our patient remained hospitalized during his treatment course. For a patient who has support, it is possible to be discharged either home or to a rehabilitation facility once flap viability has been confirmed after surgery.
Another consideration is type of immobilization. Immobilization options include casting, use of Kirschner wires (K-wires), and use of rigid external fixation. For cross-leg flaps, external fixation is superior to casting and K-wires, as it provides a more rigid construct and easier access to the flap for serial evaluation. Further, it is easier for the patient to maintain personal hygiene, and it can provide heel rises to avoid pressure ulcers.
Conclusion
To our knowledge, there have been no reports of using a cross-leg flap for wound complications in high-energy pilon fractures. As already mentioned, many of these fractures may be associated with severe soft-tissue injury and may need flap coverage. A cross-leg flap with external fixation of both legs provides a limb salvage option with satisfactory patient outcomes.
1. McCann PA, Jackson M, Mitchell ST, Atkins RM. Complications of definitive open reduction and internal fixation of pilon fractures of the distal tibia. Int Orthop. 2011;35(3):413-418.
2. Rüedi TP, Allgöwer M. Fractures of the lower end of the tibia into the ankle joint. Injury. 1969;1:92-99.
3. Sirkin M, Sanders R, DiPasquale T, Herscovici D Jr. A staged protocol for soft tissue management in the treatment of complex pilon fractures. J Orthop Trauma. 2004;18(8 suppl):S32-S38.
4. Boraiah S, Kemp TJ, Erwteman A, Lucas PA, Asprinio DE. Outcome following open reduction and internal fixation of open pilon fractures. J Bone Joint Surg Am. 2010;92(2):346-352.
5. Cheng C, Li X, Abudu S. Repairing postoperative soft tissue defects of tibia and ankle open fractures with muscle flap pedicled with medial half of soleus [in Chinese]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2009;23(12):1440-1442.
6. Yunus A, Yusuf A, Chen G. Repair of soft tissue defect by reverse soleus muscle flap after pilon fracture fixation [in Chinese]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2007;21(9):925-927.
7. Conroy J, Agarwal M, Giannoudis PV, Matthews SJ. Early internal fixation and soft tissue cover of severe open tibial pilon fractures. Int Orthop. 2003;27(6):343-347.
8. Megerle K, Palm-Bröking K, Germann G. The cross-finger flap [in German]. Oper Orthop Traumatol. 2008;20(2):97-102.
9. Largey A, Faline A, Hebrard W, Hamoui M, Canovas F. Management of massive traumatic compound defects of the foot. Orthop Traumatol Surg Res. 2009;95(4):301-304.
10. Zhao L, Wan L, Wang S. Clinical studies on maintenance of cross-leg position through internal fixation with Kirschner wire after cross-leg flap procedure. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2006;20(12):1211-1213.
Soft-tissue complications are a known problem in the treatment of pilon fractures of the distal end of the tibia. These fractures typically occur as the result of a high-energy mechanism, and axial load and shear forces often lead to a severe soft-tissue injury. In many cases, these injuries may require additional procedures to provide adequate soft-tissue coverage. These procedures can include use of either a rotational muscle flap or a free flap transfer. In some cases, however, these flaps are not possible secondary to vascular compromise.
In this article, we report the case of a pilon fracture combined with severe soft-tissue injury and vascular compromise of the leg. A cross-leg fasciocutaneous flap was performed as a salvage procedure for coverage of the soft-tissue defect. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 23-year-old man sustained a left grade III open pilon fracture after a fall off a cherry picker. He was initially treated with irrigation and débridement of the open anteromedial wound, wound closure, application of external fixation, and open reduction and internal fixation (ORIF) of the concomitant comminuted fibular fracture. Operative fixation of the pilon was performed 3 weeks after injury, once skin and soft tissues were in acceptable condition (Figure 1). Skin closure was performed with 2-0 Vicryl sutures (Ethicon, Inc, Somerville, New Jersey) followed by 3-0 nylon skin sutures and No. 2 nylon retention sutures to reduce tension at the incision.
On postoperative day 17, the patient was found to have skin necrosis with exposed hardware over the medial laceration that had resulted from the open fracture (Figure 2). The wound measured 7×6 cm. The plastic surgery team was consulted, and a soft-tissue flap was recommended. Preoperative computed tomography angiogram (Figure 3) revealed 1 vessel runoff in the leg, constituting the peroneal artery, and a conventional angiogram confirmed this finding (Figure 4). Despite these findings, the patient was taken to the operating room 4 weeks after initial injury to try to find a vessel compatible with anastomosis. Intraoperative wound exploration confirmed no patent blood supply for local soft-tissue flap coverage. Therefore, the wound was irrigated and débrided, and a vacuum-assisted closure (VAC) dressing was applied despite exposed hardware and bone. A decision was then made to attempt a cross-leg flap as a salvage procedure, and VAC dressing therapy was continued for several weeks to prepare the recipient site (Figure 5).
Seven weeks after injury, the patient was taken to the operating room by the orthopedic surgery and plastic surgery teams. After débridement, a fasciocutaneous flap was raised from the middle third of the contralateral leg (Figure 6) based on a posterior tibial artery perforator. The flap, which measured 7×7 cm (sufficient to cover the defect), was raised from lateral to medial from the posterior aspect of the leg with the pedicle located on the medial aspect of the right leg. Flap placement was facilitated by flexing the left knee to 80°. The flap was sutured into place with 4-0 Vicryl deep sutures followed by 4-0 nylon and superficial sutures in an interrupted fashion (Figure 7). Rigid external fixation was then applied to both extremities, bridging them together in optimal position (Figure 8). This construct included 2 short bars that would elevate the patient’s heels off the bed to reduce the chance of heel decubiti. Although including the feet in the external fixator construct may help prevent equinus contracture, we splinted the ankles in neutral position immediately after surgery so that we could begin early range-of-motion (ROM) exercises of the ankles to prevent stiffness. Ankle ROM exercises were started once the flap incorporated, 3 weeks after placement of the external fixator. Lacking medical insurance coverage, the patient could not be admitted to a rehabilitation facility or receive home care. He lived independently and had no help at home, so he had to remain hospitalized after placement of the external fixator. While hospitalized, the surgical site was treated with frequent dressing changes, including use of bacitracin and nonadherent dressing.
After flap coverage and 4 weeks of bed rest, a base clamping test confirmed the flap was incorporated into the recipient bed. The patient was then returned to the operating room for removal of the external fixator and skin grafting of the donor site. After surgery, he was started on physical therapy, including exercises for bilateral hip, knee, and ankle ROM and strengthening of the lower extremities. Four months after initial injury, the fracture was healed, based on bone consolidation, seen on radiographs, that is consistent with other pilon fractures treated at our institution. Six months after external fixator removal, the patient was able to ambulate independently with minimal discomfort (Figure 9). Passive and active ankle ROM was 20° of dorsiflexion and 25° of plantarflexion, compared with 25° of dorsiflexion and 45° of plantarflexion on the contralateral extremity. Subtalar motion had some stiffness with a 10° arc, compared with a 25° arc on the contralateral extremity. On simple manual testing, the patient had 5/5 motor strength with dorsiflexion, plantarflexion, inversion, and eversion. He returned to full duty as a landscaper about 1 year after initial injury and had no recurrence of wound complications or infection.
Discussion
Fractures of the distal tibia are commonly known as pilon or plafond fractures. They represent up to 10% of all tibial fractures. The injury consists of an intra-articular fracture of the tibiotalar joint with varying degrees of proximal extension into the tibial metaphysis. The etiology is an axial load on the tibia with or without a rotational force.1 Treatment is challenging. The literature includes many reports of wound and soft-tissue complications after ORIF. In 1969, Rüedi and Allgöwer2 published recommendations that have become the standard for treatment of pilon fractures. Twelve percent of the 84 fractures included in their study were associated with wound complications. In 2004, Sirkin and colleagues3 suggested that wound problems associated with ORIF of pilon fractures may be caused by attempts at immediate fixation through swollen soft tissue. They postulated that staging the procedure and waiting for decreased soft-tissue swelling may reduce the incidence of wound complications. In their series, only 2.9% of closed pilon fractures and only 9.1% of open fractures had any wound complications, and none of their patients required skin grafts, rotation flaps, or free tissue transfers.
However, soft-tissue complications still remain a significant threat in the treatment of pilon fracture, and cases that require additional procedures for soft-tissue coverage are common. In some cases, wound necrosis may lead to below-knee amputation.4 There are several coverage options, including local rotational flaps using the soleus muscle5,6 as well as free flaps using the latissimus dorsi, gracilis, or rectus abdominis muscles.7 These options require a sufficient blood supply to the region.
Many high-energy pilon fractures may be associated with vascular injury, and therefore flap survival may be compromised. We have reported such a case in the present article. Our patient’s preoperative angiogram indicated he had 1-vessel runoff to the distal leg—a situation incompatible with free tissue transfer. It is not clear whether this finding is secondary to trauma to the leg or is caused by an anatomical anomaly. Nevertheless, the poor vascularity posed a challenge to providing soft-tissue coverage. Cross-finger8 and cross-foot9 flaps have been described in upper and lower extremity injuries. In 2006, Zhao and colleagues10 reported on 5 patients with tibia and/or hardware exposure after operative fixation of tibia fractures. These patients had poor local soft tissue around the wound and therefore underwent cross-leg flap for coverage. It is not clear where the soft-tissue defects were located and whether any studies were performed to assess the local blood flow.
From our patient’s case, we learned that multiple factors should be considered when assessing such high-energy injuries. First, respecting the soft tissues is of paramount importance. Our initial management on presentation consisted of irrigation and débridement of the wound, fixation of the fibula, and application of an external fixator to allow for soft-tissue healing before definitive fixation of the pilon. Although ultimately the patient required soft-tissue coverage, soft-tissue healing and viability are important in preventing unnecessary soft-tissue procedures, and therefore we would not have handled our initial treatment differently.
Patient selection is also important. The ideal candidate for a cross-leg flap is a young, healthy person who is compliant and has a strong support system to help with activities of daily living. Unfortunately, because of financial issues and lack of home support, our patient remained hospitalized during his treatment course. For a patient who has support, it is possible to be discharged either home or to a rehabilitation facility once flap viability has been confirmed after surgery.
Another consideration is type of immobilization. Immobilization options include casting, use of Kirschner wires (K-wires), and use of rigid external fixation. For cross-leg flaps, external fixation is superior to casting and K-wires, as it provides a more rigid construct and easier access to the flap for serial evaluation. Further, it is easier for the patient to maintain personal hygiene, and it can provide heel rises to avoid pressure ulcers.
Conclusion
To our knowledge, there have been no reports of using a cross-leg flap for wound complications in high-energy pilon fractures. As already mentioned, many of these fractures may be associated with severe soft-tissue injury and may need flap coverage. A cross-leg flap with external fixation of both legs provides a limb salvage option with satisfactory patient outcomes.
Soft-tissue complications are a known problem in the treatment of pilon fractures of the distal end of the tibia. These fractures typically occur as the result of a high-energy mechanism, and axial load and shear forces often lead to a severe soft-tissue injury. In many cases, these injuries may require additional procedures to provide adequate soft-tissue coverage. These procedures can include use of either a rotational muscle flap or a free flap transfer. In some cases, however, these flaps are not possible secondary to vascular compromise.
In this article, we report the case of a pilon fracture combined with severe soft-tissue injury and vascular compromise of the leg. A cross-leg fasciocutaneous flap was performed as a salvage procedure for coverage of the soft-tissue defect. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 23-year-old man sustained a left grade III open pilon fracture after a fall off a cherry picker. He was initially treated with irrigation and débridement of the open anteromedial wound, wound closure, application of external fixation, and open reduction and internal fixation (ORIF) of the concomitant comminuted fibular fracture. Operative fixation of the pilon was performed 3 weeks after injury, once skin and soft tissues were in acceptable condition (Figure 1). Skin closure was performed with 2-0 Vicryl sutures (Ethicon, Inc, Somerville, New Jersey) followed by 3-0 nylon skin sutures and No. 2 nylon retention sutures to reduce tension at the incision.
On postoperative day 17, the patient was found to have skin necrosis with exposed hardware over the medial laceration that had resulted from the open fracture (Figure 2). The wound measured 7×6 cm. The plastic surgery team was consulted, and a soft-tissue flap was recommended. Preoperative computed tomography angiogram (Figure 3) revealed 1 vessel runoff in the leg, constituting the peroneal artery, and a conventional angiogram confirmed this finding (Figure 4). Despite these findings, the patient was taken to the operating room 4 weeks after initial injury to try to find a vessel compatible with anastomosis. Intraoperative wound exploration confirmed no patent blood supply for local soft-tissue flap coverage. Therefore, the wound was irrigated and débrided, and a vacuum-assisted closure (VAC) dressing was applied despite exposed hardware and bone. A decision was then made to attempt a cross-leg flap as a salvage procedure, and VAC dressing therapy was continued for several weeks to prepare the recipient site (Figure 5).
Seven weeks after injury, the patient was taken to the operating room by the orthopedic surgery and plastic surgery teams. After débridement, a fasciocutaneous flap was raised from the middle third of the contralateral leg (Figure 6) based on a posterior tibial artery perforator. The flap, which measured 7×7 cm (sufficient to cover the defect), was raised from lateral to medial from the posterior aspect of the leg with the pedicle located on the medial aspect of the right leg. Flap placement was facilitated by flexing the left knee to 80°. The flap was sutured into place with 4-0 Vicryl deep sutures followed by 4-0 nylon and superficial sutures in an interrupted fashion (Figure 7). Rigid external fixation was then applied to both extremities, bridging them together in optimal position (Figure 8). This construct included 2 short bars that would elevate the patient’s heels off the bed to reduce the chance of heel decubiti. Although including the feet in the external fixator construct may help prevent equinus contracture, we splinted the ankles in neutral position immediately after surgery so that we could begin early range-of-motion (ROM) exercises of the ankles to prevent stiffness. Ankle ROM exercises were started once the flap incorporated, 3 weeks after placement of the external fixator. Lacking medical insurance coverage, the patient could not be admitted to a rehabilitation facility or receive home care. He lived independently and had no help at home, so he had to remain hospitalized after placement of the external fixator. While hospitalized, the surgical site was treated with frequent dressing changes, including use of bacitracin and nonadherent dressing.
After flap coverage and 4 weeks of bed rest, a base clamping test confirmed the flap was incorporated into the recipient bed. The patient was then returned to the operating room for removal of the external fixator and skin grafting of the donor site. After surgery, he was started on physical therapy, including exercises for bilateral hip, knee, and ankle ROM and strengthening of the lower extremities. Four months after initial injury, the fracture was healed, based on bone consolidation, seen on radiographs, that is consistent with other pilon fractures treated at our institution. Six months after external fixator removal, the patient was able to ambulate independently with minimal discomfort (Figure 9). Passive and active ankle ROM was 20° of dorsiflexion and 25° of plantarflexion, compared with 25° of dorsiflexion and 45° of plantarflexion on the contralateral extremity. Subtalar motion had some stiffness with a 10° arc, compared with a 25° arc on the contralateral extremity. On simple manual testing, the patient had 5/5 motor strength with dorsiflexion, plantarflexion, inversion, and eversion. He returned to full duty as a landscaper about 1 year after initial injury and had no recurrence of wound complications or infection.
Discussion
Fractures of the distal tibia are commonly known as pilon or plafond fractures. They represent up to 10% of all tibial fractures. The injury consists of an intra-articular fracture of the tibiotalar joint with varying degrees of proximal extension into the tibial metaphysis. The etiology is an axial load on the tibia with or without a rotational force.1 Treatment is challenging. The literature includes many reports of wound and soft-tissue complications after ORIF. In 1969, Rüedi and Allgöwer2 published recommendations that have become the standard for treatment of pilon fractures. Twelve percent of the 84 fractures included in their study were associated with wound complications. In 2004, Sirkin and colleagues3 suggested that wound problems associated with ORIF of pilon fractures may be caused by attempts at immediate fixation through swollen soft tissue. They postulated that staging the procedure and waiting for decreased soft-tissue swelling may reduce the incidence of wound complications. In their series, only 2.9% of closed pilon fractures and only 9.1% of open fractures had any wound complications, and none of their patients required skin grafts, rotation flaps, or free tissue transfers.
However, soft-tissue complications still remain a significant threat in the treatment of pilon fracture, and cases that require additional procedures for soft-tissue coverage are common. In some cases, wound necrosis may lead to below-knee amputation.4 There are several coverage options, including local rotational flaps using the soleus muscle5,6 as well as free flaps using the latissimus dorsi, gracilis, or rectus abdominis muscles.7 These options require a sufficient blood supply to the region.
Many high-energy pilon fractures may be associated with vascular injury, and therefore flap survival may be compromised. We have reported such a case in the present article. Our patient’s preoperative angiogram indicated he had 1-vessel runoff to the distal leg—a situation incompatible with free tissue transfer. It is not clear whether this finding is secondary to trauma to the leg or is caused by an anatomical anomaly. Nevertheless, the poor vascularity posed a challenge to providing soft-tissue coverage. Cross-finger8 and cross-foot9 flaps have been described in upper and lower extremity injuries. In 2006, Zhao and colleagues10 reported on 5 patients with tibia and/or hardware exposure after operative fixation of tibia fractures. These patients had poor local soft tissue around the wound and therefore underwent cross-leg flap for coverage. It is not clear where the soft-tissue defects were located and whether any studies were performed to assess the local blood flow.
From our patient’s case, we learned that multiple factors should be considered when assessing such high-energy injuries. First, respecting the soft tissues is of paramount importance. Our initial management on presentation consisted of irrigation and débridement of the wound, fixation of the fibula, and application of an external fixator to allow for soft-tissue healing before definitive fixation of the pilon. Although ultimately the patient required soft-tissue coverage, soft-tissue healing and viability are important in preventing unnecessary soft-tissue procedures, and therefore we would not have handled our initial treatment differently.
Patient selection is also important. The ideal candidate for a cross-leg flap is a young, healthy person who is compliant and has a strong support system to help with activities of daily living. Unfortunately, because of financial issues and lack of home support, our patient remained hospitalized during his treatment course. For a patient who has support, it is possible to be discharged either home or to a rehabilitation facility once flap viability has been confirmed after surgery.
Another consideration is type of immobilization. Immobilization options include casting, use of Kirschner wires (K-wires), and use of rigid external fixation. For cross-leg flaps, external fixation is superior to casting and K-wires, as it provides a more rigid construct and easier access to the flap for serial evaluation. Further, it is easier for the patient to maintain personal hygiene, and it can provide heel rises to avoid pressure ulcers.
Conclusion
To our knowledge, there have been no reports of using a cross-leg flap for wound complications in high-energy pilon fractures. As already mentioned, many of these fractures may be associated with severe soft-tissue injury and may need flap coverage. A cross-leg flap with external fixation of both legs provides a limb salvage option with satisfactory patient outcomes.
1. McCann PA, Jackson M, Mitchell ST, Atkins RM. Complications of definitive open reduction and internal fixation of pilon fractures of the distal tibia. Int Orthop. 2011;35(3):413-418.
2. Rüedi TP, Allgöwer M. Fractures of the lower end of the tibia into the ankle joint. Injury. 1969;1:92-99.
3. Sirkin M, Sanders R, DiPasquale T, Herscovici D Jr. A staged protocol for soft tissue management in the treatment of complex pilon fractures. J Orthop Trauma. 2004;18(8 suppl):S32-S38.
4. Boraiah S, Kemp TJ, Erwteman A, Lucas PA, Asprinio DE. Outcome following open reduction and internal fixation of open pilon fractures. J Bone Joint Surg Am. 2010;92(2):346-352.
5. Cheng C, Li X, Abudu S. Repairing postoperative soft tissue defects of tibia and ankle open fractures with muscle flap pedicled with medial half of soleus [in Chinese]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2009;23(12):1440-1442.
6. Yunus A, Yusuf A, Chen G. Repair of soft tissue defect by reverse soleus muscle flap after pilon fracture fixation [in Chinese]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2007;21(9):925-927.
7. Conroy J, Agarwal M, Giannoudis PV, Matthews SJ. Early internal fixation and soft tissue cover of severe open tibial pilon fractures. Int Orthop. 2003;27(6):343-347.
8. Megerle K, Palm-Bröking K, Germann G. The cross-finger flap [in German]. Oper Orthop Traumatol. 2008;20(2):97-102.
9. Largey A, Faline A, Hebrard W, Hamoui M, Canovas F. Management of massive traumatic compound defects of the foot. Orthop Traumatol Surg Res. 2009;95(4):301-304.
10. Zhao L, Wan L, Wang S. Clinical studies on maintenance of cross-leg position through internal fixation with Kirschner wire after cross-leg flap procedure. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2006;20(12):1211-1213.
1. McCann PA, Jackson M, Mitchell ST, Atkins RM. Complications of definitive open reduction and internal fixation of pilon fractures of the distal tibia. Int Orthop. 2011;35(3):413-418.
2. Rüedi TP, Allgöwer M. Fractures of the lower end of the tibia into the ankle joint. Injury. 1969;1:92-99.
3. Sirkin M, Sanders R, DiPasquale T, Herscovici D Jr. A staged protocol for soft tissue management in the treatment of complex pilon fractures. J Orthop Trauma. 2004;18(8 suppl):S32-S38.
4. Boraiah S, Kemp TJ, Erwteman A, Lucas PA, Asprinio DE. Outcome following open reduction and internal fixation of open pilon fractures. J Bone Joint Surg Am. 2010;92(2):346-352.
5. Cheng C, Li X, Abudu S. Repairing postoperative soft tissue defects of tibia and ankle open fractures with muscle flap pedicled with medial half of soleus [in Chinese]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2009;23(12):1440-1442.
6. Yunus A, Yusuf A, Chen G. Repair of soft tissue defect by reverse soleus muscle flap after pilon fracture fixation [in Chinese]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2007;21(9):925-927.
7. Conroy J, Agarwal M, Giannoudis PV, Matthews SJ. Early internal fixation and soft tissue cover of severe open tibial pilon fractures. Int Orthop. 2003;27(6):343-347.
8. Megerle K, Palm-Bröking K, Germann G. The cross-finger flap [in German]. Oper Orthop Traumatol. 2008;20(2):97-102.
9. Largey A, Faline A, Hebrard W, Hamoui M, Canovas F. Management of massive traumatic compound defects of the foot. Orthop Traumatol Surg Res. 2009;95(4):301-304.
10. Zhao L, Wan L, Wang S. Clinical studies on maintenance of cross-leg position through internal fixation with Kirschner wire after cross-leg flap procedure. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2006;20(12):1211-1213.
MOVES Study Touts Benefits of Glucosamine/Chondroitin for Knee Osteoarthritis
The combination of chondroitin sulfate plus glucosamine hydrochloride has comparable efficacy to the anti-inflammatory drug celecoxib after 6 months of treatment in severe osteoarthritis, according to a recent clinical trial published online ahead of print January 14 in Annals of the Rheumatic Diseases. Specifically, it was seen that the combination of these two drugs caused a clinically relevant reduction in pain, functional disability, stiffness, swelling, and joint effusion.
MOVES (Multicentre Osteoarthritis InterVEntion Trial with Sysadoa) is a multicenter, randomized, parallel-group, double-blind controlled clinical trial that enrolled 606 patients from at 42 centers in France, Germany, Poland, and Spain. The study was sponsored by Bioibérica, suppliers of the chondroitin sulfate used in Cosamin DS. The trial was developed as an extension of the GAIT (Glucosamine/Chondroitin Arthritis Intervention Trial) study, which showed that the combination of 1,500 mg per day of glucosamine hydrochloride and 1,200 mg per day of chondroitin sulfate was effective in a moderate to severe subgroup of patients.
Patients with primary knee osteoarthritis and moderate to severe pain were randomized to receive 1,200 mg of chondroitin sulfate (400 mg/d tid) plus 1,500 mg of glucosamine hydrochloride (500 mg/d tid) or celecoxib (200 mg) every day for 6 months.
After 6 months, patients experienced the following:
• A reduction in pain by 50.1%.
• A reduction in swelling by 53%.
• A reduction in stiffness by 46.9%.
• A reduction in functional disability by 45.5%.
• A reduction in joint effusion by 56%.
“This study confirms the efficacy of the combination of pharmaceutical-grade chondroitin sulfate and glucosamine [hydrochloride] in the long term and suggests that, considering its excellent safety profile, it may be a good alternative for patients with cardiovascular or gastrointestinal problems, for whom chronic treatment with NSAIDs cannot be recommended,” said lead author Marc C. Hochberg, MD, MPH, a Professor at the University of Maryland School of Medicine in Baltimore.
Suggested Reading
Hochberg MC, Martel-Pelletier J, Monfort J, et al. Combined chondroitin sulfate and glucosamine for painful knee osteoarthritis: a multicentre, randomised, double-blind, non-inferiority trial versus celecoxib. Ann Rheum Dis. 2015 Jan 14. [Epub ahead of print]
The combination of chondroitin sulfate plus glucosamine hydrochloride has comparable efficacy to the anti-inflammatory drug celecoxib after 6 months of treatment in severe osteoarthritis, according to a recent clinical trial published online ahead of print January 14 in Annals of the Rheumatic Diseases. Specifically, it was seen that the combination of these two drugs caused a clinically relevant reduction in pain, functional disability, stiffness, swelling, and joint effusion.
MOVES (Multicentre Osteoarthritis InterVEntion Trial with Sysadoa) is a multicenter, randomized, parallel-group, double-blind controlled clinical trial that enrolled 606 patients from at 42 centers in France, Germany, Poland, and Spain. The study was sponsored by Bioibérica, suppliers of the chondroitin sulfate used in Cosamin DS. The trial was developed as an extension of the GAIT (Glucosamine/Chondroitin Arthritis Intervention Trial) study, which showed that the combination of 1,500 mg per day of glucosamine hydrochloride and 1,200 mg per day of chondroitin sulfate was effective in a moderate to severe subgroup of patients.
Patients with primary knee osteoarthritis and moderate to severe pain were randomized to receive 1,200 mg of chondroitin sulfate (400 mg/d tid) plus 1,500 mg of glucosamine hydrochloride (500 mg/d tid) or celecoxib (200 mg) every day for 6 months.
After 6 months, patients experienced the following:
• A reduction in pain by 50.1%.
• A reduction in swelling by 53%.
• A reduction in stiffness by 46.9%.
• A reduction in functional disability by 45.5%.
• A reduction in joint effusion by 56%.
“This study confirms the efficacy of the combination of pharmaceutical-grade chondroitin sulfate and glucosamine [hydrochloride] in the long term and suggests that, considering its excellent safety profile, it may be a good alternative for patients with cardiovascular or gastrointestinal problems, for whom chronic treatment with NSAIDs cannot be recommended,” said lead author Marc C. Hochberg, MD, MPH, a Professor at the University of Maryland School of Medicine in Baltimore.
The combination of chondroitin sulfate plus glucosamine hydrochloride has comparable efficacy to the anti-inflammatory drug celecoxib after 6 months of treatment in severe osteoarthritis, according to a recent clinical trial published online ahead of print January 14 in Annals of the Rheumatic Diseases. Specifically, it was seen that the combination of these two drugs caused a clinically relevant reduction in pain, functional disability, stiffness, swelling, and joint effusion.
MOVES (Multicentre Osteoarthritis InterVEntion Trial with Sysadoa) is a multicenter, randomized, parallel-group, double-blind controlled clinical trial that enrolled 606 patients from at 42 centers in France, Germany, Poland, and Spain. The study was sponsored by Bioibérica, suppliers of the chondroitin sulfate used in Cosamin DS. The trial was developed as an extension of the GAIT (Glucosamine/Chondroitin Arthritis Intervention Trial) study, which showed that the combination of 1,500 mg per day of glucosamine hydrochloride and 1,200 mg per day of chondroitin sulfate was effective in a moderate to severe subgroup of patients.
Patients with primary knee osteoarthritis and moderate to severe pain were randomized to receive 1,200 mg of chondroitin sulfate (400 mg/d tid) plus 1,500 mg of glucosamine hydrochloride (500 mg/d tid) or celecoxib (200 mg) every day for 6 months.
After 6 months, patients experienced the following:
• A reduction in pain by 50.1%.
• A reduction in swelling by 53%.
• A reduction in stiffness by 46.9%.
• A reduction in functional disability by 45.5%.
• A reduction in joint effusion by 56%.
“This study confirms the efficacy of the combination of pharmaceutical-grade chondroitin sulfate and glucosamine [hydrochloride] in the long term and suggests that, considering its excellent safety profile, it may be a good alternative for patients with cardiovascular or gastrointestinal problems, for whom chronic treatment with NSAIDs cannot be recommended,” said lead author Marc C. Hochberg, MD, MPH, a Professor at the University of Maryland School of Medicine in Baltimore.
Suggested Reading
Hochberg MC, Martel-Pelletier J, Monfort J, et al. Combined chondroitin sulfate and glucosamine for painful knee osteoarthritis: a multicentre, randomised, double-blind, non-inferiority trial versus celecoxib. Ann Rheum Dis. 2015 Jan 14. [Epub ahead of print]
Suggested Reading
Hochberg MC, Martel-Pelletier J, Monfort J, et al. Combined chondroitin sulfate and glucosamine for painful knee osteoarthritis: a multicentre, randomised, double-blind, non-inferiority trial versus celecoxib. Ann Rheum Dis. 2015 Jan 14. [Epub ahead of print]
Fluorescent Probe May Detect Early to Moderate Osteoarthritis
A fluorescent probe may make it easier to diagnose and monitor osteoarthritis, according to a study published in the February issue of Arthritis & Rheumatology. Researchers found that a fluorescent probe tracked the development of osteoarthritis in male mice, brightening as the disease progressed. Their study is the first to demonstrate that near-infared fluorescence can be used to detect osteoarthritis changes over time.
“Patients are frequently in pain by the time osteoarthritis is diagnosed. The imaging tests most frequently used, x-rays, do not indicate the level of pain or allow us to directly see the amount of cartilage loss, which is a challenge for physicians and patients,” said lead author Averi A. Leahy, BA, an MD/PhD student in the medical scientist training program at Tufts University School of Medicine (TUSM) and the Sackler School of Graduate Biomedical Sciences at Tufts in Boston.
For this study, the right knees of 54 mice were affected by injury-induced osteoarthritis and served as the experimental group. The healthy left knees of the mice served as the control group.
Over a 2-month period, the researchers took images of each knee every 2 weeks to determine if the fluorescent probe emitted a signal. The signal became brighter in the injured right knee, at every examined time point, through the early to moderate stages of osteoarthritis. The probe emitted a lower signal in the healthy left knee, and did not increase significantly over time.
According to the researchers, the fluorescent probe made it easy to see the activities that lead to cartilage breakdown in the initial and moderate stages of osteoarthritis, which is necessary for early detection and adequate monitoring of the disease.
Senior author Li Zeng, PhD, an Associate Professor in the Department of Integrative Physiology and Pathobiology at TUSM and member of the cellular, molecular, and developmental biology program faculty at the Sackler School reported that the next step is to monitor the fluorescent probe over a longer period of time to determine whether the same results are produced during the late stages of osteoarthritis.
Suggested Reading
Leahy AA, Esfahani SA, Foote AT, et al. Analysis of the trajectory of osteoarthritis development in a mouse model by serial near-infrared fluorescence imaging of matrix metalloproteinase activities. Arthritis Rheumatol. 2015;67(2):442-453.
A fluorescent probe may make it easier to diagnose and monitor osteoarthritis, according to a study published in the February issue of Arthritis & Rheumatology. Researchers found that a fluorescent probe tracked the development of osteoarthritis in male mice, brightening as the disease progressed. Their study is the first to demonstrate that near-infared fluorescence can be used to detect osteoarthritis changes over time.
“Patients are frequently in pain by the time osteoarthritis is diagnosed. The imaging tests most frequently used, x-rays, do not indicate the level of pain or allow us to directly see the amount of cartilage loss, which is a challenge for physicians and patients,” said lead author Averi A. Leahy, BA, an MD/PhD student in the medical scientist training program at Tufts University School of Medicine (TUSM) and the Sackler School of Graduate Biomedical Sciences at Tufts in Boston.
For this study, the right knees of 54 mice were affected by injury-induced osteoarthritis and served as the experimental group. The healthy left knees of the mice served as the control group.
Over a 2-month period, the researchers took images of each knee every 2 weeks to determine if the fluorescent probe emitted a signal. The signal became brighter in the injured right knee, at every examined time point, through the early to moderate stages of osteoarthritis. The probe emitted a lower signal in the healthy left knee, and did not increase significantly over time.
According to the researchers, the fluorescent probe made it easy to see the activities that lead to cartilage breakdown in the initial and moderate stages of osteoarthritis, which is necessary for early detection and adequate monitoring of the disease.
Senior author Li Zeng, PhD, an Associate Professor in the Department of Integrative Physiology and Pathobiology at TUSM and member of the cellular, molecular, and developmental biology program faculty at the Sackler School reported that the next step is to monitor the fluorescent probe over a longer period of time to determine whether the same results are produced during the late stages of osteoarthritis.
A fluorescent probe may make it easier to diagnose and monitor osteoarthritis, according to a study published in the February issue of Arthritis & Rheumatology. Researchers found that a fluorescent probe tracked the development of osteoarthritis in male mice, brightening as the disease progressed. Their study is the first to demonstrate that near-infared fluorescence can be used to detect osteoarthritis changes over time.
“Patients are frequently in pain by the time osteoarthritis is diagnosed. The imaging tests most frequently used, x-rays, do not indicate the level of pain or allow us to directly see the amount of cartilage loss, which is a challenge for physicians and patients,” said lead author Averi A. Leahy, BA, an MD/PhD student in the medical scientist training program at Tufts University School of Medicine (TUSM) and the Sackler School of Graduate Biomedical Sciences at Tufts in Boston.
For this study, the right knees of 54 mice were affected by injury-induced osteoarthritis and served as the experimental group. The healthy left knees of the mice served as the control group.
Over a 2-month period, the researchers took images of each knee every 2 weeks to determine if the fluorescent probe emitted a signal. The signal became brighter in the injured right knee, at every examined time point, through the early to moderate stages of osteoarthritis. The probe emitted a lower signal in the healthy left knee, and did not increase significantly over time.
According to the researchers, the fluorescent probe made it easy to see the activities that lead to cartilage breakdown in the initial and moderate stages of osteoarthritis, which is necessary for early detection and adequate monitoring of the disease.
Senior author Li Zeng, PhD, an Associate Professor in the Department of Integrative Physiology and Pathobiology at TUSM and member of the cellular, molecular, and developmental biology program faculty at the Sackler School reported that the next step is to monitor the fluorescent probe over a longer period of time to determine whether the same results are produced during the late stages of osteoarthritis.
Suggested Reading
Leahy AA, Esfahani SA, Foote AT, et al. Analysis of the trajectory of osteoarthritis development in a mouse model by serial near-infrared fluorescence imaging of matrix metalloproteinase activities. Arthritis Rheumatol. 2015;67(2):442-453.
Suggested Reading
Leahy AA, Esfahani SA, Foote AT, et al. Analysis of the trajectory of osteoarthritis development in a mouse model by serial near-infrared fluorescence imaging of matrix metalloproteinase activities. Arthritis Rheumatol. 2015;67(2):442-453.
Low Back Pain Triggered by Fatigue, Distractions, and Awkward Positions
New research reveals the physical and psychosocial factors that significantly increase the risk of low back pain onset. The trial results, which were published online ahead of print February 9 in Arthritis Care & Research, show that being engaged in manual tasks involving awkward positions will increase the risk of low back pain by 8 times. People who are distracted during activities or fatigued also significantly increase their risk of acute low back pain.
“Understanding which risk factors contribute to back pain and controlling exposure to these risks is an important first step in prevention,” said Manuela Ferreira, PhD, Associate Professor at the George Institute for Global Health and Sydney Medical School at the University of Sydney in New South Wales, Australia. “Our study is the first to examine brief exposure to a range of modifiable triggers for an acute episode of low back pain.”
For this case-crossover study, Dr. Ferreira and colleagues recruited 999 participants from 300 primary care clinics in Sydney, Australia, who had an acute low back pain episode between October 2011 and November 2012. Participants were asked to report exposure to 12 physical or psychosocial factors in the 96 hours prior to the onset of back pain.
The study found that:
• The risk of a new episode of low back pain significantly increased due to a range of triggers, from an odds ratio of 2.7 for moderate to vigorous physical activity to 25.0 for distraction during an activity.
• Back pain risk was highest between 7:00 am and at noontime.
• Age moderated the effect of exposure to heavy loads, with an odds ratio for individuals age 20, 40, or 60 at 13.6, 6.0, and 2.7, respectively.
“Understanding which modifiable risk factors lead to low back pain is an important step toward controlling a condition that affects so many worldwide,” said Dr. Ferreira. “Our findings enhance knowledge of low back pain triggers and will assist the development of new prevention programs that can reduce suffering from this potentially disabling condition.”
Suggested Reading
Steffens D, Ferreira ML, Latimer J, et al. What triggers an episode of acute low back pain? a case-crossover study. Arthritis Care Res. 2015 Feb 9. [Epub ahead of print]
New research reveals the physical and psychosocial factors that significantly increase the risk of low back pain onset. The trial results, which were published online ahead of print February 9 in Arthritis Care & Research, show that being engaged in manual tasks involving awkward positions will increase the risk of low back pain by 8 times. People who are distracted during activities or fatigued also significantly increase their risk of acute low back pain.
“Understanding which risk factors contribute to back pain and controlling exposure to these risks is an important first step in prevention,” said Manuela Ferreira, PhD, Associate Professor at the George Institute for Global Health and Sydney Medical School at the University of Sydney in New South Wales, Australia. “Our study is the first to examine brief exposure to a range of modifiable triggers for an acute episode of low back pain.”
For this case-crossover study, Dr. Ferreira and colleagues recruited 999 participants from 300 primary care clinics in Sydney, Australia, who had an acute low back pain episode between October 2011 and November 2012. Participants were asked to report exposure to 12 physical or psychosocial factors in the 96 hours prior to the onset of back pain.
The study found that:
• The risk of a new episode of low back pain significantly increased due to a range of triggers, from an odds ratio of 2.7 for moderate to vigorous physical activity to 25.0 for distraction during an activity.
• Back pain risk was highest between 7:00 am and at noontime.
• Age moderated the effect of exposure to heavy loads, with an odds ratio for individuals age 20, 40, or 60 at 13.6, 6.0, and 2.7, respectively.
“Understanding which modifiable risk factors lead to low back pain is an important step toward controlling a condition that affects so many worldwide,” said Dr. Ferreira. “Our findings enhance knowledge of low back pain triggers and will assist the development of new prevention programs that can reduce suffering from this potentially disabling condition.”
New research reveals the physical and psychosocial factors that significantly increase the risk of low back pain onset. The trial results, which were published online ahead of print February 9 in Arthritis Care & Research, show that being engaged in manual tasks involving awkward positions will increase the risk of low back pain by 8 times. People who are distracted during activities or fatigued also significantly increase their risk of acute low back pain.
“Understanding which risk factors contribute to back pain and controlling exposure to these risks is an important first step in prevention,” said Manuela Ferreira, PhD, Associate Professor at the George Institute for Global Health and Sydney Medical School at the University of Sydney in New South Wales, Australia. “Our study is the first to examine brief exposure to a range of modifiable triggers for an acute episode of low back pain.”
For this case-crossover study, Dr. Ferreira and colleagues recruited 999 participants from 300 primary care clinics in Sydney, Australia, who had an acute low back pain episode between October 2011 and November 2012. Participants were asked to report exposure to 12 physical or psychosocial factors in the 96 hours prior to the onset of back pain.
The study found that:
• The risk of a new episode of low back pain significantly increased due to a range of triggers, from an odds ratio of 2.7 for moderate to vigorous physical activity to 25.0 for distraction during an activity.
• Back pain risk was highest between 7:00 am and at noontime.
• Age moderated the effect of exposure to heavy loads, with an odds ratio for individuals age 20, 40, or 60 at 13.6, 6.0, and 2.7, respectively.
“Understanding which modifiable risk factors lead to low back pain is an important step toward controlling a condition that affects so many worldwide,” said Dr. Ferreira. “Our findings enhance knowledge of low back pain triggers and will assist the development of new prevention programs that can reduce suffering from this potentially disabling condition.”
Suggested Reading
Steffens D, Ferreira ML, Latimer J, et al. What triggers an episode of acute low back pain? a case-crossover study. Arthritis Care Res. 2015 Feb 9. [Epub ahead of print]
Suggested Reading
Steffens D, Ferreira ML, Latimer J, et al. What triggers an episode of acute low back pain? a case-crossover study. Arthritis Care Res. 2015 Feb 9. [Epub ahead of print]
Mycobacterium bovis Infection of Total Knee Arthroplasty After Bacillus Calmette-Guérin Therapy for Bladder Cancer
Intravesicular instillation of bacillus Calmette-Guérin (BCG), an attenuated form of Mycobacterium bovis, is the most effective treatment for superficial bladder cancer.1,2 Minor local reactions to this treatment, such as cystitis and hematuria, are common, but more severe systemic complications3,4 have also been documented, including sepsis, pneumonitis, granulomatous hepatitis, vertebral osteomyelitis,5,6 and rarely, total joint infection.7-11
We present a case of M bovis infection of a total knee arthroplasty (TKA) after BCG immunotherapy for bladder cancer that was successfully treated with antitubercular chemotherapy and retention of implants. We include a review of the literature addressing this rare mode of infection. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 66-year-old man presented with a chief complaint of progressive left knee stiffness over several months. Five years earlier, he underwent uncemented left TKA. His knee was functioning well with active range of motion from 0° to 126°, and he had returned to strenuous cycling. One year after his TKA and 4 years prior to the onset of stiffness, he had been diagnosed with superficial transitional cell carcinoma of the bladder. His treatment included intravesicular BCG therapy weekly for 6 weeks followed by semi-annual maintenance therapy.
Initial examination upon presentation with left knee stiffness showed a significant effusion and diminished range of motion but little discomfort. The patient denied fever, chills, night sweats, and weight loss. Radiographs were normal with good component positioning and normal-appearing bone-implant interfaces (Figures A, B). Erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and white blood cell count (WBC) were within normal limits, and aspirate of the knee revealed no organisms. Based on these findings, the presumptive diagnosis was an adverse reaction to polyethylene wear. Because of persistent stiffness, the patient underwent an examination under anesthesia, arthroscopy, and major synovectomy with biopsy. Intraoperative findings included normal polyethylene but a marked hypertrophic synovitis and abnormal, semi-turbid fluid. The fluid WBC count was 5.35×109/L but no organisms were isolated initially. Histologic samples showed chronic inflammation with patches of acute inflammation. Approximately 6 weeks after surgery, cultures became positive for acid-fast bacillus, which was identified as M bovis.
Maintenance BCG therapy was discontinued, and antitubercular chemotherapy was initiated, consisting of 12 months of rifampin 600 mg daily and isoniazid 300 mg daily. Because symptoms significantly improved after arthroscopic incision and drainage and synovectomy, the TKA implants were maintained and symptoms closely monitored. Subsequent cultures and biopsies remained negative, and the patient continued to do well clinically with no residual stiffness.
At 7½-year follow-up, there is no clinical evidence of infection, and the patient continues to enjoy a high level of function with no pain and no recurrent stiffness. He has returned to cycling, logging more than 40,000 miles. However, a recurrence of bladder cancer is being treated with mitomycin C and gemcitabine, alternative to BCG.
Discussion
Mycobacterial infection in total joint arthroplasty (TJA) is uncommon;12M bovis infection of joint arthroplasty after intravesicular BCG therapy is exceedingly rare. Joint infection is thought to be the result of dissemination of BCG throughout the bloodstream.13
A review of the literature of BCG infection of TJA after intravesicular therapy for bladder cancer revealed only 5 case reports (Table). The average age on presentation was 77 years, and all patients were men, with 4 total hip arthroplasties (THAs) and 1 TKA. The average time from index procedure to initial presentation was 7.8 years, and the average time from cancer diagnosis to initial presentation was 20 months. Patients received an average of 8.6 consecutive weeks of BCG treatments, and maintenance therapy was not noted in any of the published reports. The average duration of antitubercular therapy was 13 months, and it comprised either 2- or 3-agent therapy. All reported cases were treated with removal of primary implants in either a 1- or 2-stage fashion. To our knowledge, this is only the second case of BCG infection of TKA reported in the literature and the first report of successful treatment with retention of primary implants.
There are several possible explanations for the success of a more conservative treatment approach in our patient. First, this TKA was uncemented. Second, BCG is an attenuated form of M bovis, which is itself a relatively less virulent species than M tuberculosis. Finally, mycobacterial species do not produce the biofilm that is seen in other bacterial arthroplasty infections, which typically necessitate removal of implants in cases of chronic infection.14
This case was unique because the patient lacked signs of infectious symptoms, there were normal inflammatory markers, and arthroscopy was necessary to aid in the diagnosis. The definitive diagnosis in this case was significantly delayed to attain a positive M bovis culture. Definitive treatment was provided by arthroscopy, implant salvage, and antitubercular chemotherapy only. The standard of care for an infected modular TKA normally involves revision of the polyethylene tibial insert with irrigation and débridement, or removal of components and insertion of new implants in a 1- or 2-stage procedure. Despite the unusual algorithm to reach a definitive diagnosis of an infected joint arthroplasty in this case, we do not recommend arthroscopic biopsy, washout, and antimicrobial therapy as definitive treatment for infected joint arthroplasty, and we continue to support the removal of infected components in a staged manner.
Conclusion
Joint replacement patients with bladder cancer represent a relatively small cohort. Based on current demographics and the increasing demand for joint arthroplasty, it is likely that this unique subset of patients will grow. No current standard of care exists for the treatment of these patients. One preventative measure is to consider alternative types of chemotherapy for bladder cancer treatment, such as mitomycin. Another potential solution would be administration of prophylactic doses of antitubercular agents concomitantly with intravesicular BCG, which would allow for the local effects of BCG immunotherapy while controlling the potential for systemic dissemination. The optimal dose range to achieve this dual effect is not known and is an area for research.
It is important for both arthroplasty surgeons and urologists to be aware of this potential complication in order to appropriately counsel this unique subset of patients. Our case report is the first to demonstrate that a successful outcome can be obtained with retention of primary components. Through research and continued data acquisition, a more concrete standard of care can be established. Until then, we recommend a collaborative approach between informed parties to devise a patient-specific plan of care.
1. Herr HW, Morales A. History of bacillus Calmette-Guérin and bladder cancer: an immunotherapy success story. J Urol. 2008;179(1):53-56.
2. Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-Guérin in the treatment of superficial bladder tumors. J Urol. 1976;116(2):180-183.
3. Lamm DL. Complications of bacillus Calmette-Guérin immunotherapy. Urol Clin North Am. 1992;19(3):565-572.
4. Lamm DL, van der Meijden PM, Morales A, et al. Incidence and treatment of complications of bacillus Calmette-Guérin intravesical therapy in superficial bladder cancer. J Urol. 1992;147(3):596-600.
5. Rozenblit A, Wasserman E, Marin ML, Veith FJ, Cynamon J, Rosenblit G. Infected aortic aneurysm and vertebral osteomyelitis after intravesical bacillus Calmette-Guérin therapy. AJR Am J Roentgenol. 1996;167(3):711-713.
6. Aljada IS, Crane JK, Corriere N, Wagle DG, Amsterdam D. Mycobacterium bovis BCG causing vertebral osteomyelitis (Pott’s disease) following intravesical BCG therapy. J Clin Microbiol. 1999;37(6):2106-2108.
7. Chazerain P, Desplaces N, Mamoudy P, Leonard P, Ziza JM. Prosthetic total knee infection with a bacillus Calmette-Guerin (BCG) strain after BCG therapy for bladder cancer. J Rheum. 1993;20(12):2171-2172.
8. Guerra CE, Betts RF, O’Keefe RJ, Shilling JW. Mycobacterium bovis osteomyelitis involving a hip arthroplasty after intravesicular bacille Calmette-Guérin for bladder cancer. Clin Infect Dis. 1998;27(3):639-640.
9. Segal A, Krauss ES. Infected total hip arthroplasty after intravesical bacillus Calmette-Guérin therapy. J Arthroplasty. 2007;22(5):759-762.
10. Reigstad O, Siewers P. A total hip replacement infected with mycobacterium bovis after intravesicular treatment with Bacille Calmette-Guérin for bladder cancer. J Bone Joint Surg Br. 2008;90(2):225-227.
11. Gomez E, Chiang T, Louie T, Ponnapalli M, Eng R, Huang DB. Prosthetic joint infection due to Mycobacterium bovis after intravesical instillation of Bacillus Calmette-Guerin (BCG). International J Microbiol. 2009;2009:527208. doi: 10.1155/2009/527208. Epub 2009 Dec 16.
12. Buchholz HW, Elson RA, Engelbrecht E, Lodenkämper H, Röttger J, Siegel A. Management of deep infection of total hip replacement. J Bone Joint Surg Br. 1981;63(3):342-353.
13. Xerri B, Chrétien Y, Le Parc JM. Reactive polyarthritis induced by intravesical BCG therapy for carcinoma of the bladder. Eur J Med. 1993;2(8):503-505.
14. Ha KY, Chung YG, Ryoo SJ. Adherence and biofilm formation of Staphylococcus epidermidis and Mycobacterium tuberculosis on various spinal implants. Spine (Phila Pa 1976). 2005;30(1):38-43.
Intravesicular instillation of bacillus Calmette-Guérin (BCG), an attenuated form of Mycobacterium bovis, is the most effective treatment for superficial bladder cancer.1,2 Minor local reactions to this treatment, such as cystitis and hematuria, are common, but more severe systemic complications3,4 have also been documented, including sepsis, pneumonitis, granulomatous hepatitis, vertebral osteomyelitis,5,6 and rarely, total joint infection.7-11
We present a case of M bovis infection of a total knee arthroplasty (TKA) after BCG immunotherapy for bladder cancer that was successfully treated with antitubercular chemotherapy and retention of implants. We include a review of the literature addressing this rare mode of infection. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 66-year-old man presented with a chief complaint of progressive left knee stiffness over several months. Five years earlier, he underwent uncemented left TKA. His knee was functioning well with active range of motion from 0° to 126°, and he had returned to strenuous cycling. One year after his TKA and 4 years prior to the onset of stiffness, he had been diagnosed with superficial transitional cell carcinoma of the bladder. His treatment included intravesicular BCG therapy weekly for 6 weeks followed by semi-annual maintenance therapy.
Initial examination upon presentation with left knee stiffness showed a significant effusion and diminished range of motion but little discomfort. The patient denied fever, chills, night sweats, and weight loss. Radiographs were normal with good component positioning and normal-appearing bone-implant interfaces (Figures A, B). Erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and white blood cell count (WBC) were within normal limits, and aspirate of the knee revealed no organisms. Based on these findings, the presumptive diagnosis was an adverse reaction to polyethylene wear. Because of persistent stiffness, the patient underwent an examination under anesthesia, arthroscopy, and major synovectomy with biopsy. Intraoperative findings included normal polyethylene but a marked hypertrophic synovitis and abnormal, semi-turbid fluid. The fluid WBC count was 5.35×109/L but no organisms were isolated initially. Histologic samples showed chronic inflammation with patches of acute inflammation. Approximately 6 weeks after surgery, cultures became positive for acid-fast bacillus, which was identified as M bovis.
Maintenance BCG therapy was discontinued, and antitubercular chemotherapy was initiated, consisting of 12 months of rifampin 600 mg daily and isoniazid 300 mg daily. Because symptoms significantly improved after arthroscopic incision and drainage and synovectomy, the TKA implants were maintained and symptoms closely monitored. Subsequent cultures and biopsies remained negative, and the patient continued to do well clinically with no residual stiffness.
At 7½-year follow-up, there is no clinical evidence of infection, and the patient continues to enjoy a high level of function with no pain and no recurrent stiffness. He has returned to cycling, logging more than 40,000 miles. However, a recurrence of bladder cancer is being treated with mitomycin C and gemcitabine, alternative to BCG.
Discussion
Mycobacterial infection in total joint arthroplasty (TJA) is uncommon;12M bovis infection of joint arthroplasty after intravesicular BCG therapy is exceedingly rare. Joint infection is thought to be the result of dissemination of BCG throughout the bloodstream.13
A review of the literature of BCG infection of TJA after intravesicular therapy for bladder cancer revealed only 5 case reports (Table). The average age on presentation was 77 years, and all patients were men, with 4 total hip arthroplasties (THAs) and 1 TKA. The average time from index procedure to initial presentation was 7.8 years, and the average time from cancer diagnosis to initial presentation was 20 months. Patients received an average of 8.6 consecutive weeks of BCG treatments, and maintenance therapy was not noted in any of the published reports. The average duration of antitubercular therapy was 13 months, and it comprised either 2- or 3-agent therapy. All reported cases were treated with removal of primary implants in either a 1- or 2-stage fashion. To our knowledge, this is only the second case of BCG infection of TKA reported in the literature and the first report of successful treatment with retention of primary implants.
There are several possible explanations for the success of a more conservative treatment approach in our patient. First, this TKA was uncemented. Second, BCG is an attenuated form of M bovis, which is itself a relatively less virulent species than M tuberculosis. Finally, mycobacterial species do not produce the biofilm that is seen in other bacterial arthroplasty infections, which typically necessitate removal of implants in cases of chronic infection.14
This case was unique because the patient lacked signs of infectious symptoms, there were normal inflammatory markers, and arthroscopy was necessary to aid in the diagnosis. The definitive diagnosis in this case was significantly delayed to attain a positive M bovis culture. Definitive treatment was provided by arthroscopy, implant salvage, and antitubercular chemotherapy only. The standard of care for an infected modular TKA normally involves revision of the polyethylene tibial insert with irrigation and débridement, or removal of components and insertion of new implants in a 1- or 2-stage procedure. Despite the unusual algorithm to reach a definitive diagnosis of an infected joint arthroplasty in this case, we do not recommend arthroscopic biopsy, washout, and antimicrobial therapy as definitive treatment for infected joint arthroplasty, and we continue to support the removal of infected components in a staged manner.
Conclusion
Joint replacement patients with bladder cancer represent a relatively small cohort. Based on current demographics and the increasing demand for joint arthroplasty, it is likely that this unique subset of patients will grow. No current standard of care exists for the treatment of these patients. One preventative measure is to consider alternative types of chemotherapy for bladder cancer treatment, such as mitomycin. Another potential solution would be administration of prophylactic doses of antitubercular agents concomitantly with intravesicular BCG, which would allow for the local effects of BCG immunotherapy while controlling the potential for systemic dissemination. The optimal dose range to achieve this dual effect is not known and is an area for research.
It is important for both arthroplasty surgeons and urologists to be aware of this potential complication in order to appropriately counsel this unique subset of patients. Our case report is the first to demonstrate that a successful outcome can be obtained with retention of primary components. Through research and continued data acquisition, a more concrete standard of care can be established. Until then, we recommend a collaborative approach between informed parties to devise a patient-specific plan of care.
Intravesicular instillation of bacillus Calmette-Guérin (BCG), an attenuated form of Mycobacterium bovis, is the most effective treatment for superficial bladder cancer.1,2 Minor local reactions to this treatment, such as cystitis and hematuria, are common, but more severe systemic complications3,4 have also been documented, including sepsis, pneumonitis, granulomatous hepatitis, vertebral osteomyelitis,5,6 and rarely, total joint infection.7-11
We present a case of M bovis infection of a total knee arthroplasty (TKA) after BCG immunotherapy for bladder cancer that was successfully treated with antitubercular chemotherapy and retention of implants. We include a review of the literature addressing this rare mode of infection. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 66-year-old man presented with a chief complaint of progressive left knee stiffness over several months. Five years earlier, he underwent uncemented left TKA. His knee was functioning well with active range of motion from 0° to 126°, and he had returned to strenuous cycling. One year after his TKA and 4 years prior to the onset of stiffness, he had been diagnosed with superficial transitional cell carcinoma of the bladder. His treatment included intravesicular BCG therapy weekly for 6 weeks followed by semi-annual maintenance therapy.
Initial examination upon presentation with left knee stiffness showed a significant effusion and diminished range of motion but little discomfort. The patient denied fever, chills, night sweats, and weight loss. Radiographs were normal with good component positioning and normal-appearing bone-implant interfaces (Figures A, B). Erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and white blood cell count (WBC) were within normal limits, and aspirate of the knee revealed no organisms. Based on these findings, the presumptive diagnosis was an adverse reaction to polyethylene wear. Because of persistent stiffness, the patient underwent an examination under anesthesia, arthroscopy, and major synovectomy with biopsy. Intraoperative findings included normal polyethylene but a marked hypertrophic synovitis and abnormal, semi-turbid fluid. The fluid WBC count was 5.35×109/L but no organisms were isolated initially. Histologic samples showed chronic inflammation with patches of acute inflammation. Approximately 6 weeks after surgery, cultures became positive for acid-fast bacillus, which was identified as M bovis.
Maintenance BCG therapy was discontinued, and antitubercular chemotherapy was initiated, consisting of 12 months of rifampin 600 mg daily and isoniazid 300 mg daily. Because symptoms significantly improved after arthroscopic incision and drainage and synovectomy, the TKA implants were maintained and symptoms closely monitored. Subsequent cultures and biopsies remained negative, and the patient continued to do well clinically with no residual stiffness.
At 7½-year follow-up, there is no clinical evidence of infection, and the patient continues to enjoy a high level of function with no pain and no recurrent stiffness. He has returned to cycling, logging more than 40,000 miles. However, a recurrence of bladder cancer is being treated with mitomycin C and gemcitabine, alternative to BCG.
Discussion
Mycobacterial infection in total joint arthroplasty (TJA) is uncommon;12M bovis infection of joint arthroplasty after intravesicular BCG therapy is exceedingly rare. Joint infection is thought to be the result of dissemination of BCG throughout the bloodstream.13
A review of the literature of BCG infection of TJA after intravesicular therapy for bladder cancer revealed only 5 case reports (Table). The average age on presentation was 77 years, and all patients were men, with 4 total hip arthroplasties (THAs) and 1 TKA. The average time from index procedure to initial presentation was 7.8 years, and the average time from cancer diagnosis to initial presentation was 20 months. Patients received an average of 8.6 consecutive weeks of BCG treatments, and maintenance therapy was not noted in any of the published reports. The average duration of antitubercular therapy was 13 months, and it comprised either 2- or 3-agent therapy. All reported cases were treated with removal of primary implants in either a 1- or 2-stage fashion. To our knowledge, this is only the second case of BCG infection of TKA reported in the literature and the first report of successful treatment with retention of primary implants.
There are several possible explanations for the success of a more conservative treatment approach in our patient. First, this TKA was uncemented. Second, BCG is an attenuated form of M bovis, which is itself a relatively less virulent species than M tuberculosis. Finally, mycobacterial species do not produce the biofilm that is seen in other bacterial arthroplasty infections, which typically necessitate removal of implants in cases of chronic infection.14
This case was unique because the patient lacked signs of infectious symptoms, there were normal inflammatory markers, and arthroscopy was necessary to aid in the diagnosis. The definitive diagnosis in this case was significantly delayed to attain a positive M bovis culture. Definitive treatment was provided by arthroscopy, implant salvage, and antitubercular chemotherapy only. The standard of care for an infected modular TKA normally involves revision of the polyethylene tibial insert with irrigation and débridement, or removal of components and insertion of new implants in a 1- or 2-stage procedure. Despite the unusual algorithm to reach a definitive diagnosis of an infected joint arthroplasty in this case, we do not recommend arthroscopic biopsy, washout, and antimicrobial therapy as definitive treatment for infected joint arthroplasty, and we continue to support the removal of infected components in a staged manner.
Conclusion
Joint replacement patients with bladder cancer represent a relatively small cohort. Based on current demographics and the increasing demand for joint arthroplasty, it is likely that this unique subset of patients will grow. No current standard of care exists for the treatment of these patients. One preventative measure is to consider alternative types of chemotherapy for bladder cancer treatment, such as mitomycin. Another potential solution would be administration of prophylactic doses of antitubercular agents concomitantly with intravesicular BCG, which would allow for the local effects of BCG immunotherapy while controlling the potential for systemic dissemination. The optimal dose range to achieve this dual effect is not known and is an area for research.
It is important for both arthroplasty surgeons and urologists to be aware of this potential complication in order to appropriately counsel this unique subset of patients. Our case report is the first to demonstrate that a successful outcome can be obtained with retention of primary components. Through research and continued data acquisition, a more concrete standard of care can be established. Until then, we recommend a collaborative approach between informed parties to devise a patient-specific plan of care.
1. Herr HW, Morales A. History of bacillus Calmette-Guérin and bladder cancer: an immunotherapy success story. J Urol. 2008;179(1):53-56.
2. Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-Guérin in the treatment of superficial bladder tumors. J Urol. 1976;116(2):180-183.
3. Lamm DL. Complications of bacillus Calmette-Guérin immunotherapy. Urol Clin North Am. 1992;19(3):565-572.
4. Lamm DL, van der Meijden PM, Morales A, et al. Incidence and treatment of complications of bacillus Calmette-Guérin intravesical therapy in superficial bladder cancer. J Urol. 1992;147(3):596-600.
5. Rozenblit A, Wasserman E, Marin ML, Veith FJ, Cynamon J, Rosenblit G. Infected aortic aneurysm and vertebral osteomyelitis after intravesical bacillus Calmette-Guérin therapy. AJR Am J Roentgenol. 1996;167(3):711-713.
6. Aljada IS, Crane JK, Corriere N, Wagle DG, Amsterdam D. Mycobacterium bovis BCG causing vertebral osteomyelitis (Pott’s disease) following intravesical BCG therapy. J Clin Microbiol. 1999;37(6):2106-2108.
7. Chazerain P, Desplaces N, Mamoudy P, Leonard P, Ziza JM. Prosthetic total knee infection with a bacillus Calmette-Guerin (BCG) strain after BCG therapy for bladder cancer. J Rheum. 1993;20(12):2171-2172.
8. Guerra CE, Betts RF, O’Keefe RJ, Shilling JW. Mycobacterium bovis osteomyelitis involving a hip arthroplasty after intravesicular bacille Calmette-Guérin for bladder cancer. Clin Infect Dis. 1998;27(3):639-640.
9. Segal A, Krauss ES. Infected total hip arthroplasty after intravesical bacillus Calmette-Guérin therapy. J Arthroplasty. 2007;22(5):759-762.
10. Reigstad O, Siewers P. A total hip replacement infected with mycobacterium bovis after intravesicular treatment with Bacille Calmette-Guérin for bladder cancer. J Bone Joint Surg Br. 2008;90(2):225-227.
11. Gomez E, Chiang T, Louie T, Ponnapalli M, Eng R, Huang DB. Prosthetic joint infection due to Mycobacterium bovis after intravesical instillation of Bacillus Calmette-Guerin (BCG). International J Microbiol. 2009;2009:527208. doi: 10.1155/2009/527208. Epub 2009 Dec 16.
12. Buchholz HW, Elson RA, Engelbrecht E, Lodenkämper H, Röttger J, Siegel A. Management of deep infection of total hip replacement. J Bone Joint Surg Br. 1981;63(3):342-353.
13. Xerri B, Chrétien Y, Le Parc JM. Reactive polyarthritis induced by intravesical BCG therapy for carcinoma of the bladder. Eur J Med. 1993;2(8):503-505.
14. Ha KY, Chung YG, Ryoo SJ. Adherence and biofilm formation of Staphylococcus epidermidis and Mycobacterium tuberculosis on various spinal implants. Spine (Phila Pa 1976). 2005;30(1):38-43.
1. Herr HW, Morales A. History of bacillus Calmette-Guérin and bladder cancer: an immunotherapy success story. J Urol. 2008;179(1):53-56.
2. Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-Guérin in the treatment of superficial bladder tumors. J Urol. 1976;116(2):180-183.
3. Lamm DL. Complications of bacillus Calmette-Guérin immunotherapy. Urol Clin North Am. 1992;19(3):565-572.
4. Lamm DL, van der Meijden PM, Morales A, et al. Incidence and treatment of complications of bacillus Calmette-Guérin intravesical therapy in superficial bladder cancer. J Urol. 1992;147(3):596-600.
5. Rozenblit A, Wasserman E, Marin ML, Veith FJ, Cynamon J, Rosenblit G. Infected aortic aneurysm and vertebral osteomyelitis after intravesical bacillus Calmette-Guérin therapy. AJR Am J Roentgenol. 1996;167(3):711-713.
6. Aljada IS, Crane JK, Corriere N, Wagle DG, Amsterdam D. Mycobacterium bovis BCG causing vertebral osteomyelitis (Pott’s disease) following intravesical BCG therapy. J Clin Microbiol. 1999;37(6):2106-2108.
7. Chazerain P, Desplaces N, Mamoudy P, Leonard P, Ziza JM. Prosthetic total knee infection with a bacillus Calmette-Guerin (BCG) strain after BCG therapy for bladder cancer. J Rheum. 1993;20(12):2171-2172.
8. Guerra CE, Betts RF, O’Keefe RJ, Shilling JW. Mycobacterium bovis osteomyelitis involving a hip arthroplasty after intravesicular bacille Calmette-Guérin for bladder cancer. Clin Infect Dis. 1998;27(3):639-640.
9. Segal A, Krauss ES. Infected total hip arthroplasty after intravesical bacillus Calmette-Guérin therapy. J Arthroplasty. 2007;22(5):759-762.
10. Reigstad O, Siewers P. A total hip replacement infected with mycobacterium bovis after intravesicular treatment with Bacille Calmette-Guérin for bladder cancer. J Bone Joint Surg Br. 2008;90(2):225-227.
11. Gomez E, Chiang T, Louie T, Ponnapalli M, Eng R, Huang DB. Prosthetic joint infection due to Mycobacterium bovis after intravesical instillation of Bacillus Calmette-Guerin (BCG). International J Microbiol. 2009;2009:527208. doi: 10.1155/2009/527208. Epub 2009 Dec 16.
12. Buchholz HW, Elson RA, Engelbrecht E, Lodenkämper H, Röttger J, Siegel A. Management of deep infection of total hip replacement. J Bone Joint Surg Br. 1981;63(3):342-353.
13. Xerri B, Chrétien Y, Le Parc JM. Reactive polyarthritis induced by intravesical BCG therapy for carcinoma of the bladder. Eur J Med. 1993;2(8):503-505.
14. Ha KY, Chung YG, Ryoo SJ. Adherence and biofilm formation of Staphylococcus epidermidis and Mycobacterium tuberculosis on various spinal implants. Spine (Phila Pa 1976). 2005;30(1):38-43.
Failure of Total Hip Arthroplasty Secondary to Infection Caused by Brucella abortus and the Risk of Transmission to Operative Staff
Brucellosis is a zoonotic disease transmitted to humans through contact with animal hosts or animal products. Infection of total knee or hip arthroplasty by Brucella species is a rare complication with only 18 cases reported in the English literature.1-12 We describe a case of an infected total hip replacement, its treatment, and 2-year follow-up and review the available literature. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 67-year-old Spanish-speaking woman, a native of Mexico, presented with a painful right total hip arthroplasty (THA) 2 years after implantation in Chihuahua, Mexico. The patient reported 1 year of increasing thigh pain with recent onset of start-up pain, and also mild groin pain. The patient reported an uneventful postoperative course without wound drainage and denied any history of fevers, chills, or night sweats after the procedure. Preoperative notes and radiographs were unavailable for review. Radiographic evaluation showed a hybrid construct with a well-fixed–appearing, uncemented acetabular component but a failed cemented femoral stem (Figures 1A, 1B). Although we discussed revision surgery, the patient elected not to proceed with surgery or to undergo evaluation to rule out infection. Nine months later, she returned with worsening pain and requested revision surgery; radiographs showed progressive bone loss around the cement mantle (Figures 2A, 2B).
Hematologic evaluation showed an erythrocyte sedimentation rate (ESR) of 54 mm/h (normal, 0-27 mm/h) and C-reactive protein (CRP) level of 0.24 mg/L (normal, <0.8). An aspiration of the hip with fluoroscopic guidance produced a small sample (0.2 mL) of yellow synovial fluid. There was not enough fluid for cell count, but fluid culture was negative.
The patient was taken to the operating room for revision THA. Because of concern about progressive bone loss and elevated infectious indices, the administration of antibiotics was delayed until we obtained sufficient deep-tissue specimens. Before opening the capsule, we introduced a syringe into the joint and aspirated 10 mL of cloudy yellow synovial fluid that was sent for cell count. Additional findings at surgery included a grossly loose stem with a fragmented cement mantle surrounded by poor bone stock with anterior cortical bone loss and a loose acetabular component with pockets of cavitary bone loss. Frozen section showed up to 5 nucleated cells per high power field, and the cell count showed 1480 nucleated cells/µL (50% polymorphonuclear cells). The equivocal intraoperative findings (cell count and frozen section) and the loose femoral and acetabular components with significant bone loss were sufficiently concerning that we removed the components and placed a cement spacer rather than proceed with revision arthroplasty (Figures 3A, 3B). The surgeon, first assistant, and scrub technician wore body exhaust suits. We performed irrigation of the wound bed with pulse lavage.
Intraoperative cultures (synovial fluid, joint capsule synovium, and femur pseudocapsule) were positive after 8 days and growing B abortus. Infectious disease consultants prescribed rifampin 300 mg twice daily and doxycycline 100 mg twice daily for 5 months. Follow-up ESR and CRP returned to normal range. A preoperative aspiration of the hip was negative as well. The patient returned to the operating room at 6 months for re-implantation using uncemented components; synovial fluid and tissue cultures taken at this time were negative. Two years after re-implantation, the patient is doing well without evidence of infection (Figures 4A, 4B). Additional follow-up will be required to monitor for infection and implant survival. Additional history taken from the patient after the culture results revealed that her development of hip pain was preceded by a febrile illness consistent with brucellosis.
Because of the nature of the procedure (irrigation and débridement using pulse lavage), we were concerned about aerosolization of Brucella bacteria and possible transmission to all staff present during the procedure. After consulting with the New Mexico Department of Health (NMDOH) and the Centers for Disease Control and Prevention (CDC), all surgical, anesthesia, and support personnel present in the operative suite and staff who cleaned the room after the procedure were treated prophylactically (rifampin 600 mg daily, doxycycline 100 mg twice daily for 3 weeks) to prevent development of brucellosis.13 All 15 operating room personnel who were exposed elected to proceed with antibiotic prophylaxis. In addition to prophylactic antibiotics, serial serologic testing for anti-Brucella antibodies was conducted at baseline and 2, 4, 6, and 24 weeks postexposure to monitor for the development of Brucella infection. There were no conversions to positive antibody status. No personnel complained of symptoms that would indicate development of brucellosis. At the recommendation of NMDOH and CDC, all staff in the operating room during and immediately after the re-implantation procedure wore properly fitting N-95 disposable respiratory masks (3M, St. Paul, Minnesota) to guard against the potential risk of further exposure.
Discussion
Brucellosis is a zoonotic disease transmitted to humans through contact with animal hosts. Transmission can occur via breaks in the skin in direct contact, through the ingestion of unpasteurized dairy products or raw meat, or through ingestion of aerosolized bacteria. Transmission via aerosolization has been described during medical procedures.
Brucella is endemic in India, Middle Eastern and Mediterranean countries, Central Asia, and South America. Brucella species are gram-negative coccobacilli that are capable of surviving within phagocytic cells, making antibiotic treatment difficult. Brucellosis is a febrile illness that occurs after a 1- to 3-week incubation period and is often accompanied by headache, arthralgias, and hepatosplenomegaly. Osteoarticular infection is the most common complication, occurring in 10% to 85% of cases and usually involves the sacroiliac joint and the large joints of the lower extremity. Spondylitis, bursitis, tenosynovitis, endocarditis, colitis, meningitis, and osteomyelitis have also been described.7,14-17
As mentioned previously, 18 cases of infected THAs and total knee arthroplasties (TKAs) in 16 patients were identified in the English literature: 9 THAs and 9 TKAs.1-12 With the exception of 1 case reported in Texas, all others were from the Middle East or the Mediterranean region. In these patients, symptom onset occurred from 2 months to 14 years from the time of the index surgery, and symptom duration ranged from 1 month to 2 years prior to presentation. The exposure was not reported in 2 cases, but the remaining patients either ingested unpasteurized dairy products or worked closely with livestock. Laboratory evaluation revealed elevated ESR or CRP in 8 cases. In 7 cases, no laboratory results were reported, although 1 had a draining sinus. In 1 case, the ESR was normal, but a bone scan was positive. Joint aspiration yielded Brucella species in 8 cases, was negative in 3, and not reported in 5 cases (one aspirate yielded Acinetobacter baumanii). Only 3 cases reported a time-to-culture positivity (1 “prolonged” and 2 took 7 days).
Eight cases presented with loose components, while 1 case was not reported, and the remaining were presumed to be well-fixed. In cases that were identified as loose, 5 underwent a 2-stage revision and 2 underwent a 1-stage revision (in one of the 1-stage revisions, the infection was identified only after the revision from intra-operative cultures). Of those with well-fixed components, 7 patients with 9 infected joints (including the case where no preoperative description of the components was reported) were treated with oral antibiotics only (range, 6 weeks to 26 months) and 1 with irrigation and débridement and oral antibiotics. Among those treated only with antibiotics, there were 2 failures (2 joints) leading to revision surgery. The other 5 cases were reportedly doing well between 8 months and 5 years after treatment. There were no reports of transmission to hospital or laboratory personnel in any of these cases nor were there reports of precautions to limit exposure for operating room staff or hospital personnel.
Failure of TKA or THA secondary to periprosthetic infection by Brucella species is rare, and this represents only the second reported case in the United States.4 This case highlights several important principles. Maintaining a high level of suspicion for infection in cases of failed joint arthroplasty is important. In addition, as more international travel occurs and patients are seen from areas where Brucella is endemic, the possibility of this infectious etiology should be considered. Based on reported cases, patients will usually have elevated ESR or CRP; all (except 2 cases in which no exposure was reported) had known exposure to unpasteurized dairy products or livestock. Joint aspiration yielded Brucella species in 8 cases, was negative in 3, and not reported in 5 cases (1 aspirate yielded Acinetobacter baumanii). In this case, ESR and CRP were elevated, and infection was suspected but joint aspiration was negative. The initial aspiration was cultured for 5 days and previous data, as well as that presented here, suggest that prolonged culture may provide diagnostic value.18 The patient had resided in an endemic area and had exposure to unpasteurized dairy products, but Brucella infection was not considered and, therefore, no precautions were taken.
Of the reported cases, only 1 met major criteria for periprosthetic joint infection (draining sinus) while 10 of the remaining 15 cases were positive for minor criteria of periprosthetic joint infection (elevated ESR or CRP, or positive culture from joint aspiration).19 Unfortunately, the available case reports did not detail the extent to which preoperative periprosthetic joint infection could be established based on minor criteria for periprosthetic joint infection (elevated joint synovial white blood cell count or neutrophil percentage, intra-articular purulence, or elevated neutrophil count on periprosthetic tissue histologic analysis).19
Periprosthetic joint infection by Brucella species is so rare that specific recommendations for this infectious etiology based on 18 reported cases would be overreaching. However, Brucella should be considered when evaluating a potentially infected joint replacement where the possibility of exposure exists (eg, travel to or previous residence in endemic areas, close contact with livestock, or ingestion of unpasteurized dairy products in endemic regions), with the potential for transmission to operating room and hospital personnel also considered. If there is concern about Brucella involvement, tissue and fluid specimens should be labeled so that laboratory personnel can take appropriate precautions. Brucella can be cultured using routine techniques on standard, nonselective media, but the culture time-to-growth may be prolonged. Culture plates should be held for 14 days before reporting no growth of Brucella if it is suspected; the New Mexico Department of Health Microbiology Laboratory holds routine cultures for 1 week after a report of no growth. Thus, a suspicion of Brucella should be communicated in order for culture time to be adjusted if the holding of culture plates after an initial report of no growth is not standard practice. If operative intervention is planned and brucellosis is known, personnel should be notified of the possibility of exposure and appropriate measures taken (ie, wearing N-95 respiratory masks during the procedure and considering other methods of irrigation less likely to aerosolize particulates). It is not known if preoperative antibiotic therapy can sufficiently lower the bacterial load to make aerosolization less likely. If brucellosis is suspected but not identified preoperatively, wearing N-95 respiratory masks should be considered during any open procedures.
Conclusion
In cases of Brucella infection and loose components, 1- or 2-stage revision with appropriate antibiotic therapy is indicated. (There is not enough data to recommend either 1- or 2-stage revision.) Several reports comment on the ability to treat periprosthetic joint infection in the setting of well-fixed components with antibiotic therapy alone. While this appears to have been successful in 7 of 9 infected joints reported in the literature, length of follow-up ranged from 8 months to 5 years, with no report of length of follow-up in some cases. Antibiotic therapy duration ranged from 6 weeks to 26 months, and the antibiotic treatment involved combination therapy with multiple agents reported but, most commonly, doxycycline, rifampin, and streptomycin. With 2 of 9 (22%) joints failing antibiotic therapy alone and those reported to be successful having relatively short-term follow-up, this treatment strategy should be approached with caution.
1. Agarwal S, Kadhi SK, Rooney RJ. Brucellosis complicating bilateral total knee arthroplasty. Clin Orthop. 1991;267:179-181.
2. Cairó M, Calbo E, Gomez L, et al. Foreign-body osteoarticular infection by Brucella melitensis: A report of three cases. J Bone Joint Surg Am. 2006; 88(1):202-204.
3. Erdogan H, Cakmak G, Erdogan A, Arslan H. Brucella melitensis infection in total knee arthroplasty: a case report. Knee Surg Sports Traumatol Arthrosc. 2010;18(7):908-910.
4. Jones RE, Berryhill WH, Smith J, Hofman A, Rogers D. Secondary infection of a total hip replacement with Brucella abortus. Orthopedics. 1983; 6(2):184-186.
5. Kasim RA, Araj GF, Afeiche NE, Tabbarah ZA. Brucella infection in total hip replacement: case report and review of the literature. Scand J Infect Dis. 2004;36(1):65-67.
6. Malizos KN, Makris CA, Soucacos PN. Total knee arthroplasties infected by Brucella melitensis: a case report. Am J Orthop. 1997;26(4):283-285.
7. Ortega-Andreu M, Rodriguez-Merchan EC, Aguera-Gavalda M. Brucellosis as a cause of septic loosening of total hip arthroplasty. J Arthroplasty. 2002;17(3):384-387.
8. Orti A, Alcala R, Navarro V, et al. Brucellar arthritis in a total knee replacement. Eur J Clin Microbiol Infect Dis. 1997;16(11):843-845.
9. Ruiz-Iban MA, Crespo P, Diaz-Peletier R, Rozado AM, Lopez-Pardo A. Total hip arthroplasty infected by Brucella: a report of two cases. J Orthop Surg (Hong Kong). 2006;14(1):99-103.
10. Tassinari E, Di Motta D, Giardina F, Traina F, Fine MD, Toni A. Brucella infection in total knee arthroplasty. Case report and revision of the literature. Chir Organi Mov. 2008;92(1):55-59.
11. Tena D, Romanillos O, Rodriguez-Zapata M, et al. Prosthetic hip infection due to Brucella melitensis: case report and literature review. Diagn Microbiol Infect Dis. 2007;58(4):481-485.
12. Weil Y, Mattan Y, Liebergall M, Rahav G. Brucella prosthetic joint infection: a report of 3 cases and a review of the literature. Clin Infect Dis. 2003;36(7):e81-e86.
13. Brucellosis. Centers for Disease Control and Prevention website. http://www.cdc.gov/nczved/divisions/dfbmd/diseases/brucellosis/recommendations.html. Updated November 12, 2012. Accessed December 22, 2014.
14. Franco MP, Mulder M, Gilman RH, Smits HL. Human brucellosis. Lancet Infect Dis. 2007;7(12):775-786.
15. Khateeb MI, Araj GF, Majeed SA, Lulu AR. Brucella arthritis: a study of 96 cases in Kuwait. Ann Rheum Dis. 1990;49(12):994-998.
16. Luna-Martinez JE, Mejía-Terán C. Brucellosis in Mexico: current status and trends. Vet Microbiol. 2002;90(1-4):19-30.
17. Pappas G, Papadimitriou P, Akritidis N, Christou L, Tsianos EV. The new global map of human brucellosis. Lancet Infect Dis. 2006;6(2):91-99.
18. Schafer P, Fink B, Margull A, Berger I, Frommelt L. Prolonged bacterial culture to identify late periprosthetic joint infection: a promising strategy. Clin Infect Disease. 2008;47(11):1403-1409.
19. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop. 2011;469(11):2992-2994.
Brucellosis is a zoonotic disease transmitted to humans through contact with animal hosts or animal products. Infection of total knee or hip arthroplasty by Brucella species is a rare complication with only 18 cases reported in the English literature.1-12 We describe a case of an infected total hip replacement, its treatment, and 2-year follow-up and review the available literature. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 67-year-old Spanish-speaking woman, a native of Mexico, presented with a painful right total hip arthroplasty (THA) 2 years after implantation in Chihuahua, Mexico. The patient reported 1 year of increasing thigh pain with recent onset of start-up pain, and also mild groin pain. The patient reported an uneventful postoperative course without wound drainage and denied any history of fevers, chills, or night sweats after the procedure. Preoperative notes and radiographs were unavailable for review. Radiographic evaluation showed a hybrid construct with a well-fixed–appearing, uncemented acetabular component but a failed cemented femoral stem (Figures 1A, 1B). Although we discussed revision surgery, the patient elected not to proceed with surgery or to undergo evaluation to rule out infection. Nine months later, she returned with worsening pain and requested revision surgery; radiographs showed progressive bone loss around the cement mantle (Figures 2A, 2B).
Hematologic evaluation showed an erythrocyte sedimentation rate (ESR) of 54 mm/h (normal, 0-27 mm/h) and C-reactive protein (CRP) level of 0.24 mg/L (normal, <0.8). An aspiration of the hip with fluoroscopic guidance produced a small sample (0.2 mL) of yellow synovial fluid. There was not enough fluid for cell count, but fluid culture was negative.
The patient was taken to the operating room for revision THA. Because of concern about progressive bone loss and elevated infectious indices, the administration of antibiotics was delayed until we obtained sufficient deep-tissue specimens. Before opening the capsule, we introduced a syringe into the joint and aspirated 10 mL of cloudy yellow synovial fluid that was sent for cell count. Additional findings at surgery included a grossly loose stem with a fragmented cement mantle surrounded by poor bone stock with anterior cortical bone loss and a loose acetabular component with pockets of cavitary bone loss. Frozen section showed up to 5 nucleated cells per high power field, and the cell count showed 1480 nucleated cells/µL (50% polymorphonuclear cells). The equivocal intraoperative findings (cell count and frozen section) and the loose femoral and acetabular components with significant bone loss were sufficiently concerning that we removed the components and placed a cement spacer rather than proceed with revision arthroplasty (Figures 3A, 3B). The surgeon, first assistant, and scrub technician wore body exhaust suits. We performed irrigation of the wound bed with pulse lavage.
Intraoperative cultures (synovial fluid, joint capsule synovium, and femur pseudocapsule) were positive after 8 days and growing B abortus. Infectious disease consultants prescribed rifampin 300 mg twice daily and doxycycline 100 mg twice daily for 5 months. Follow-up ESR and CRP returned to normal range. A preoperative aspiration of the hip was negative as well. The patient returned to the operating room at 6 months for re-implantation using uncemented components; synovial fluid and tissue cultures taken at this time were negative. Two years after re-implantation, the patient is doing well without evidence of infection (Figures 4A, 4B). Additional follow-up will be required to monitor for infection and implant survival. Additional history taken from the patient after the culture results revealed that her development of hip pain was preceded by a febrile illness consistent with brucellosis.
Because of the nature of the procedure (irrigation and débridement using pulse lavage), we were concerned about aerosolization of Brucella bacteria and possible transmission to all staff present during the procedure. After consulting with the New Mexico Department of Health (NMDOH) and the Centers for Disease Control and Prevention (CDC), all surgical, anesthesia, and support personnel present in the operative suite and staff who cleaned the room after the procedure were treated prophylactically (rifampin 600 mg daily, doxycycline 100 mg twice daily for 3 weeks) to prevent development of brucellosis.13 All 15 operating room personnel who were exposed elected to proceed with antibiotic prophylaxis. In addition to prophylactic antibiotics, serial serologic testing for anti-Brucella antibodies was conducted at baseline and 2, 4, 6, and 24 weeks postexposure to monitor for the development of Brucella infection. There were no conversions to positive antibody status. No personnel complained of symptoms that would indicate development of brucellosis. At the recommendation of NMDOH and CDC, all staff in the operating room during and immediately after the re-implantation procedure wore properly fitting N-95 disposable respiratory masks (3M, St. Paul, Minnesota) to guard against the potential risk of further exposure.
Discussion
Brucellosis is a zoonotic disease transmitted to humans through contact with animal hosts. Transmission can occur via breaks in the skin in direct contact, through the ingestion of unpasteurized dairy products or raw meat, or through ingestion of aerosolized bacteria. Transmission via aerosolization has been described during medical procedures.
Brucella is endemic in India, Middle Eastern and Mediterranean countries, Central Asia, and South America. Brucella species are gram-negative coccobacilli that are capable of surviving within phagocytic cells, making antibiotic treatment difficult. Brucellosis is a febrile illness that occurs after a 1- to 3-week incubation period and is often accompanied by headache, arthralgias, and hepatosplenomegaly. Osteoarticular infection is the most common complication, occurring in 10% to 85% of cases and usually involves the sacroiliac joint and the large joints of the lower extremity. Spondylitis, bursitis, tenosynovitis, endocarditis, colitis, meningitis, and osteomyelitis have also been described.7,14-17
As mentioned previously, 18 cases of infected THAs and total knee arthroplasties (TKAs) in 16 patients were identified in the English literature: 9 THAs and 9 TKAs.1-12 With the exception of 1 case reported in Texas, all others were from the Middle East or the Mediterranean region. In these patients, symptom onset occurred from 2 months to 14 years from the time of the index surgery, and symptom duration ranged from 1 month to 2 years prior to presentation. The exposure was not reported in 2 cases, but the remaining patients either ingested unpasteurized dairy products or worked closely with livestock. Laboratory evaluation revealed elevated ESR or CRP in 8 cases. In 7 cases, no laboratory results were reported, although 1 had a draining sinus. In 1 case, the ESR was normal, but a bone scan was positive. Joint aspiration yielded Brucella species in 8 cases, was negative in 3, and not reported in 5 cases (one aspirate yielded Acinetobacter baumanii). Only 3 cases reported a time-to-culture positivity (1 “prolonged” and 2 took 7 days).
Eight cases presented with loose components, while 1 case was not reported, and the remaining were presumed to be well-fixed. In cases that were identified as loose, 5 underwent a 2-stage revision and 2 underwent a 1-stage revision (in one of the 1-stage revisions, the infection was identified only after the revision from intra-operative cultures). Of those with well-fixed components, 7 patients with 9 infected joints (including the case where no preoperative description of the components was reported) were treated with oral antibiotics only (range, 6 weeks to 26 months) and 1 with irrigation and débridement and oral antibiotics. Among those treated only with antibiotics, there were 2 failures (2 joints) leading to revision surgery. The other 5 cases were reportedly doing well between 8 months and 5 years after treatment. There were no reports of transmission to hospital or laboratory personnel in any of these cases nor were there reports of precautions to limit exposure for operating room staff or hospital personnel.
Failure of TKA or THA secondary to periprosthetic infection by Brucella species is rare, and this represents only the second reported case in the United States.4 This case highlights several important principles. Maintaining a high level of suspicion for infection in cases of failed joint arthroplasty is important. In addition, as more international travel occurs and patients are seen from areas where Brucella is endemic, the possibility of this infectious etiology should be considered. Based on reported cases, patients will usually have elevated ESR or CRP; all (except 2 cases in which no exposure was reported) had known exposure to unpasteurized dairy products or livestock. Joint aspiration yielded Brucella species in 8 cases, was negative in 3, and not reported in 5 cases (1 aspirate yielded Acinetobacter baumanii). In this case, ESR and CRP were elevated, and infection was suspected but joint aspiration was negative. The initial aspiration was cultured for 5 days and previous data, as well as that presented here, suggest that prolonged culture may provide diagnostic value.18 The patient had resided in an endemic area and had exposure to unpasteurized dairy products, but Brucella infection was not considered and, therefore, no precautions were taken.
Of the reported cases, only 1 met major criteria for periprosthetic joint infection (draining sinus) while 10 of the remaining 15 cases were positive for minor criteria of periprosthetic joint infection (elevated ESR or CRP, or positive culture from joint aspiration).19 Unfortunately, the available case reports did not detail the extent to which preoperative periprosthetic joint infection could be established based on minor criteria for periprosthetic joint infection (elevated joint synovial white blood cell count or neutrophil percentage, intra-articular purulence, or elevated neutrophil count on periprosthetic tissue histologic analysis).19
Periprosthetic joint infection by Brucella species is so rare that specific recommendations for this infectious etiology based on 18 reported cases would be overreaching. However, Brucella should be considered when evaluating a potentially infected joint replacement where the possibility of exposure exists (eg, travel to or previous residence in endemic areas, close contact with livestock, or ingestion of unpasteurized dairy products in endemic regions), with the potential for transmission to operating room and hospital personnel also considered. If there is concern about Brucella involvement, tissue and fluid specimens should be labeled so that laboratory personnel can take appropriate precautions. Brucella can be cultured using routine techniques on standard, nonselective media, but the culture time-to-growth may be prolonged. Culture plates should be held for 14 days before reporting no growth of Brucella if it is suspected; the New Mexico Department of Health Microbiology Laboratory holds routine cultures for 1 week after a report of no growth. Thus, a suspicion of Brucella should be communicated in order for culture time to be adjusted if the holding of culture plates after an initial report of no growth is not standard practice. If operative intervention is planned and brucellosis is known, personnel should be notified of the possibility of exposure and appropriate measures taken (ie, wearing N-95 respiratory masks during the procedure and considering other methods of irrigation less likely to aerosolize particulates). It is not known if preoperative antibiotic therapy can sufficiently lower the bacterial load to make aerosolization less likely. If brucellosis is suspected but not identified preoperatively, wearing N-95 respiratory masks should be considered during any open procedures.
Conclusion
In cases of Brucella infection and loose components, 1- or 2-stage revision with appropriate antibiotic therapy is indicated. (There is not enough data to recommend either 1- or 2-stage revision.) Several reports comment on the ability to treat periprosthetic joint infection in the setting of well-fixed components with antibiotic therapy alone. While this appears to have been successful in 7 of 9 infected joints reported in the literature, length of follow-up ranged from 8 months to 5 years, with no report of length of follow-up in some cases. Antibiotic therapy duration ranged from 6 weeks to 26 months, and the antibiotic treatment involved combination therapy with multiple agents reported but, most commonly, doxycycline, rifampin, and streptomycin. With 2 of 9 (22%) joints failing antibiotic therapy alone and those reported to be successful having relatively short-term follow-up, this treatment strategy should be approached with caution.
Brucellosis is a zoonotic disease transmitted to humans through contact with animal hosts or animal products. Infection of total knee or hip arthroplasty by Brucella species is a rare complication with only 18 cases reported in the English literature.1-12 We describe a case of an infected total hip replacement, its treatment, and 2-year follow-up and review the available literature. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 67-year-old Spanish-speaking woman, a native of Mexico, presented with a painful right total hip arthroplasty (THA) 2 years after implantation in Chihuahua, Mexico. The patient reported 1 year of increasing thigh pain with recent onset of start-up pain, and also mild groin pain. The patient reported an uneventful postoperative course without wound drainage and denied any history of fevers, chills, or night sweats after the procedure. Preoperative notes and radiographs were unavailable for review. Radiographic evaluation showed a hybrid construct with a well-fixed–appearing, uncemented acetabular component but a failed cemented femoral stem (Figures 1A, 1B). Although we discussed revision surgery, the patient elected not to proceed with surgery or to undergo evaluation to rule out infection. Nine months later, she returned with worsening pain and requested revision surgery; radiographs showed progressive bone loss around the cement mantle (Figures 2A, 2B).
Hematologic evaluation showed an erythrocyte sedimentation rate (ESR) of 54 mm/h (normal, 0-27 mm/h) and C-reactive protein (CRP) level of 0.24 mg/L (normal, <0.8). An aspiration of the hip with fluoroscopic guidance produced a small sample (0.2 mL) of yellow synovial fluid. There was not enough fluid for cell count, but fluid culture was negative.
The patient was taken to the operating room for revision THA. Because of concern about progressive bone loss and elevated infectious indices, the administration of antibiotics was delayed until we obtained sufficient deep-tissue specimens. Before opening the capsule, we introduced a syringe into the joint and aspirated 10 mL of cloudy yellow synovial fluid that was sent for cell count. Additional findings at surgery included a grossly loose stem with a fragmented cement mantle surrounded by poor bone stock with anterior cortical bone loss and a loose acetabular component with pockets of cavitary bone loss. Frozen section showed up to 5 nucleated cells per high power field, and the cell count showed 1480 nucleated cells/µL (50% polymorphonuclear cells). The equivocal intraoperative findings (cell count and frozen section) and the loose femoral and acetabular components with significant bone loss were sufficiently concerning that we removed the components and placed a cement spacer rather than proceed with revision arthroplasty (Figures 3A, 3B). The surgeon, first assistant, and scrub technician wore body exhaust suits. We performed irrigation of the wound bed with pulse lavage.
Intraoperative cultures (synovial fluid, joint capsule synovium, and femur pseudocapsule) were positive after 8 days and growing B abortus. Infectious disease consultants prescribed rifampin 300 mg twice daily and doxycycline 100 mg twice daily for 5 months. Follow-up ESR and CRP returned to normal range. A preoperative aspiration of the hip was negative as well. The patient returned to the operating room at 6 months for re-implantation using uncemented components; synovial fluid and tissue cultures taken at this time were negative. Two years after re-implantation, the patient is doing well without evidence of infection (Figures 4A, 4B). Additional follow-up will be required to monitor for infection and implant survival. Additional history taken from the patient after the culture results revealed that her development of hip pain was preceded by a febrile illness consistent with brucellosis.
Because of the nature of the procedure (irrigation and débridement using pulse lavage), we were concerned about aerosolization of Brucella bacteria and possible transmission to all staff present during the procedure. After consulting with the New Mexico Department of Health (NMDOH) and the Centers for Disease Control and Prevention (CDC), all surgical, anesthesia, and support personnel present in the operative suite and staff who cleaned the room after the procedure were treated prophylactically (rifampin 600 mg daily, doxycycline 100 mg twice daily for 3 weeks) to prevent development of brucellosis.13 All 15 operating room personnel who were exposed elected to proceed with antibiotic prophylaxis. In addition to prophylactic antibiotics, serial serologic testing for anti-Brucella antibodies was conducted at baseline and 2, 4, 6, and 24 weeks postexposure to monitor for the development of Brucella infection. There were no conversions to positive antibody status. No personnel complained of symptoms that would indicate development of brucellosis. At the recommendation of NMDOH and CDC, all staff in the operating room during and immediately after the re-implantation procedure wore properly fitting N-95 disposable respiratory masks (3M, St. Paul, Minnesota) to guard against the potential risk of further exposure.
Discussion
Brucellosis is a zoonotic disease transmitted to humans through contact with animal hosts. Transmission can occur via breaks in the skin in direct contact, through the ingestion of unpasteurized dairy products or raw meat, or through ingestion of aerosolized bacteria. Transmission via aerosolization has been described during medical procedures.
Brucella is endemic in India, Middle Eastern and Mediterranean countries, Central Asia, and South America. Brucella species are gram-negative coccobacilli that are capable of surviving within phagocytic cells, making antibiotic treatment difficult. Brucellosis is a febrile illness that occurs after a 1- to 3-week incubation period and is often accompanied by headache, arthralgias, and hepatosplenomegaly. Osteoarticular infection is the most common complication, occurring in 10% to 85% of cases and usually involves the sacroiliac joint and the large joints of the lower extremity. Spondylitis, bursitis, tenosynovitis, endocarditis, colitis, meningitis, and osteomyelitis have also been described.7,14-17
As mentioned previously, 18 cases of infected THAs and total knee arthroplasties (TKAs) in 16 patients were identified in the English literature: 9 THAs and 9 TKAs.1-12 With the exception of 1 case reported in Texas, all others were from the Middle East or the Mediterranean region. In these patients, symptom onset occurred from 2 months to 14 years from the time of the index surgery, and symptom duration ranged from 1 month to 2 years prior to presentation. The exposure was not reported in 2 cases, but the remaining patients either ingested unpasteurized dairy products or worked closely with livestock. Laboratory evaluation revealed elevated ESR or CRP in 8 cases. In 7 cases, no laboratory results were reported, although 1 had a draining sinus. In 1 case, the ESR was normal, but a bone scan was positive. Joint aspiration yielded Brucella species in 8 cases, was negative in 3, and not reported in 5 cases (one aspirate yielded Acinetobacter baumanii). Only 3 cases reported a time-to-culture positivity (1 “prolonged” and 2 took 7 days).
Eight cases presented with loose components, while 1 case was not reported, and the remaining were presumed to be well-fixed. In cases that were identified as loose, 5 underwent a 2-stage revision and 2 underwent a 1-stage revision (in one of the 1-stage revisions, the infection was identified only after the revision from intra-operative cultures). Of those with well-fixed components, 7 patients with 9 infected joints (including the case where no preoperative description of the components was reported) were treated with oral antibiotics only (range, 6 weeks to 26 months) and 1 with irrigation and débridement and oral antibiotics. Among those treated only with antibiotics, there were 2 failures (2 joints) leading to revision surgery. The other 5 cases were reportedly doing well between 8 months and 5 years after treatment. There were no reports of transmission to hospital or laboratory personnel in any of these cases nor were there reports of precautions to limit exposure for operating room staff or hospital personnel.
Failure of TKA or THA secondary to periprosthetic infection by Brucella species is rare, and this represents only the second reported case in the United States.4 This case highlights several important principles. Maintaining a high level of suspicion for infection in cases of failed joint arthroplasty is important. In addition, as more international travel occurs and patients are seen from areas where Brucella is endemic, the possibility of this infectious etiology should be considered. Based on reported cases, patients will usually have elevated ESR or CRP; all (except 2 cases in which no exposure was reported) had known exposure to unpasteurized dairy products or livestock. Joint aspiration yielded Brucella species in 8 cases, was negative in 3, and not reported in 5 cases (1 aspirate yielded Acinetobacter baumanii). In this case, ESR and CRP were elevated, and infection was suspected but joint aspiration was negative. The initial aspiration was cultured for 5 days and previous data, as well as that presented here, suggest that prolonged culture may provide diagnostic value.18 The patient had resided in an endemic area and had exposure to unpasteurized dairy products, but Brucella infection was not considered and, therefore, no precautions were taken.
Of the reported cases, only 1 met major criteria for periprosthetic joint infection (draining sinus) while 10 of the remaining 15 cases were positive for minor criteria of periprosthetic joint infection (elevated ESR or CRP, or positive culture from joint aspiration).19 Unfortunately, the available case reports did not detail the extent to which preoperative periprosthetic joint infection could be established based on minor criteria for periprosthetic joint infection (elevated joint synovial white blood cell count or neutrophil percentage, intra-articular purulence, or elevated neutrophil count on periprosthetic tissue histologic analysis).19
Periprosthetic joint infection by Brucella species is so rare that specific recommendations for this infectious etiology based on 18 reported cases would be overreaching. However, Brucella should be considered when evaluating a potentially infected joint replacement where the possibility of exposure exists (eg, travel to or previous residence in endemic areas, close contact with livestock, or ingestion of unpasteurized dairy products in endemic regions), with the potential for transmission to operating room and hospital personnel also considered. If there is concern about Brucella involvement, tissue and fluid specimens should be labeled so that laboratory personnel can take appropriate precautions. Brucella can be cultured using routine techniques on standard, nonselective media, but the culture time-to-growth may be prolonged. Culture plates should be held for 14 days before reporting no growth of Brucella if it is suspected; the New Mexico Department of Health Microbiology Laboratory holds routine cultures for 1 week after a report of no growth. Thus, a suspicion of Brucella should be communicated in order for culture time to be adjusted if the holding of culture plates after an initial report of no growth is not standard practice. If operative intervention is planned and brucellosis is known, personnel should be notified of the possibility of exposure and appropriate measures taken (ie, wearing N-95 respiratory masks during the procedure and considering other methods of irrigation less likely to aerosolize particulates). It is not known if preoperative antibiotic therapy can sufficiently lower the bacterial load to make aerosolization less likely. If brucellosis is suspected but not identified preoperatively, wearing N-95 respiratory masks should be considered during any open procedures.
Conclusion
In cases of Brucella infection and loose components, 1- or 2-stage revision with appropriate antibiotic therapy is indicated. (There is not enough data to recommend either 1- or 2-stage revision.) Several reports comment on the ability to treat periprosthetic joint infection in the setting of well-fixed components with antibiotic therapy alone. While this appears to have been successful in 7 of 9 infected joints reported in the literature, length of follow-up ranged from 8 months to 5 years, with no report of length of follow-up in some cases. Antibiotic therapy duration ranged from 6 weeks to 26 months, and the antibiotic treatment involved combination therapy with multiple agents reported but, most commonly, doxycycline, rifampin, and streptomycin. With 2 of 9 (22%) joints failing antibiotic therapy alone and those reported to be successful having relatively short-term follow-up, this treatment strategy should be approached with caution.
1. Agarwal S, Kadhi SK, Rooney RJ. Brucellosis complicating bilateral total knee arthroplasty. Clin Orthop. 1991;267:179-181.
2. Cairó M, Calbo E, Gomez L, et al. Foreign-body osteoarticular infection by Brucella melitensis: A report of three cases. J Bone Joint Surg Am. 2006; 88(1):202-204.
3. Erdogan H, Cakmak G, Erdogan A, Arslan H. Brucella melitensis infection in total knee arthroplasty: a case report. Knee Surg Sports Traumatol Arthrosc. 2010;18(7):908-910.
4. Jones RE, Berryhill WH, Smith J, Hofman A, Rogers D. Secondary infection of a total hip replacement with Brucella abortus. Orthopedics. 1983; 6(2):184-186.
5. Kasim RA, Araj GF, Afeiche NE, Tabbarah ZA. Brucella infection in total hip replacement: case report and review of the literature. Scand J Infect Dis. 2004;36(1):65-67.
6. Malizos KN, Makris CA, Soucacos PN. Total knee arthroplasties infected by Brucella melitensis: a case report. Am J Orthop. 1997;26(4):283-285.
7. Ortega-Andreu M, Rodriguez-Merchan EC, Aguera-Gavalda M. Brucellosis as a cause of septic loosening of total hip arthroplasty. J Arthroplasty. 2002;17(3):384-387.
8. Orti A, Alcala R, Navarro V, et al. Brucellar arthritis in a total knee replacement. Eur J Clin Microbiol Infect Dis. 1997;16(11):843-845.
9. Ruiz-Iban MA, Crespo P, Diaz-Peletier R, Rozado AM, Lopez-Pardo A. Total hip arthroplasty infected by Brucella: a report of two cases. J Orthop Surg (Hong Kong). 2006;14(1):99-103.
10. Tassinari E, Di Motta D, Giardina F, Traina F, Fine MD, Toni A. Brucella infection in total knee arthroplasty. Case report and revision of the literature. Chir Organi Mov. 2008;92(1):55-59.
11. Tena D, Romanillos O, Rodriguez-Zapata M, et al. Prosthetic hip infection due to Brucella melitensis: case report and literature review. Diagn Microbiol Infect Dis. 2007;58(4):481-485.
12. Weil Y, Mattan Y, Liebergall M, Rahav G. Brucella prosthetic joint infection: a report of 3 cases and a review of the literature. Clin Infect Dis. 2003;36(7):e81-e86.
13. Brucellosis. Centers for Disease Control and Prevention website. http://www.cdc.gov/nczved/divisions/dfbmd/diseases/brucellosis/recommendations.html. Updated November 12, 2012. Accessed December 22, 2014.
14. Franco MP, Mulder M, Gilman RH, Smits HL. Human brucellosis. Lancet Infect Dis. 2007;7(12):775-786.
15. Khateeb MI, Araj GF, Majeed SA, Lulu AR. Brucella arthritis: a study of 96 cases in Kuwait. Ann Rheum Dis. 1990;49(12):994-998.
16. Luna-Martinez JE, Mejía-Terán C. Brucellosis in Mexico: current status and trends. Vet Microbiol. 2002;90(1-4):19-30.
17. Pappas G, Papadimitriou P, Akritidis N, Christou L, Tsianos EV. The new global map of human brucellosis. Lancet Infect Dis. 2006;6(2):91-99.
18. Schafer P, Fink B, Margull A, Berger I, Frommelt L. Prolonged bacterial culture to identify late periprosthetic joint infection: a promising strategy. Clin Infect Disease. 2008;47(11):1403-1409.
19. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop. 2011;469(11):2992-2994.
1. Agarwal S, Kadhi SK, Rooney RJ. Brucellosis complicating bilateral total knee arthroplasty. Clin Orthop. 1991;267:179-181.
2. Cairó M, Calbo E, Gomez L, et al. Foreign-body osteoarticular infection by Brucella melitensis: A report of three cases. J Bone Joint Surg Am. 2006; 88(1):202-204.
3. Erdogan H, Cakmak G, Erdogan A, Arslan H. Brucella melitensis infection in total knee arthroplasty: a case report. Knee Surg Sports Traumatol Arthrosc. 2010;18(7):908-910.
4. Jones RE, Berryhill WH, Smith J, Hofman A, Rogers D. Secondary infection of a total hip replacement with Brucella abortus. Orthopedics. 1983; 6(2):184-186.
5. Kasim RA, Araj GF, Afeiche NE, Tabbarah ZA. Brucella infection in total hip replacement: case report and review of the literature. Scand J Infect Dis. 2004;36(1):65-67.
6. Malizos KN, Makris CA, Soucacos PN. Total knee arthroplasties infected by Brucella melitensis: a case report. Am J Orthop. 1997;26(4):283-285.
7. Ortega-Andreu M, Rodriguez-Merchan EC, Aguera-Gavalda M. Brucellosis as a cause of septic loosening of total hip arthroplasty. J Arthroplasty. 2002;17(3):384-387.
8. Orti A, Alcala R, Navarro V, et al. Brucellar arthritis in a total knee replacement. Eur J Clin Microbiol Infect Dis. 1997;16(11):843-845.
9. Ruiz-Iban MA, Crespo P, Diaz-Peletier R, Rozado AM, Lopez-Pardo A. Total hip arthroplasty infected by Brucella: a report of two cases. J Orthop Surg (Hong Kong). 2006;14(1):99-103.
10. Tassinari E, Di Motta D, Giardina F, Traina F, Fine MD, Toni A. Brucella infection in total knee arthroplasty. Case report and revision of the literature. Chir Organi Mov. 2008;92(1):55-59.
11. Tena D, Romanillos O, Rodriguez-Zapata M, et al. Prosthetic hip infection due to Brucella melitensis: case report and literature review. Diagn Microbiol Infect Dis. 2007;58(4):481-485.
12. Weil Y, Mattan Y, Liebergall M, Rahav G. Brucella prosthetic joint infection: a report of 3 cases and a review of the literature. Clin Infect Dis. 2003;36(7):e81-e86.
13. Brucellosis. Centers for Disease Control and Prevention website. http://www.cdc.gov/nczved/divisions/dfbmd/diseases/brucellosis/recommendations.html. Updated November 12, 2012. Accessed December 22, 2014.
14. Franco MP, Mulder M, Gilman RH, Smits HL. Human brucellosis. Lancet Infect Dis. 2007;7(12):775-786.
15. Khateeb MI, Araj GF, Majeed SA, Lulu AR. Brucella arthritis: a study of 96 cases in Kuwait. Ann Rheum Dis. 1990;49(12):994-998.
16. Luna-Martinez JE, Mejía-Terán C. Brucellosis in Mexico: current status and trends. Vet Microbiol. 2002;90(1-4):19-30.
17. Pappas G, Papadimitriou P, Akritidis N, Christou L, Tsianos EV. The new global map of human brucellosis. Lancet Infect Dis. 2006;6(2):91-99.
18. Schafer P, Fink B, Margull A, Berger I, Frommelt L. Prolonged bacterial culture to identify late periprosthetic joint infection: a promising strategy. Clin Infect Disease. 2008;47(11):1403-1409.
19. Parvizi J, Zmistowski B, Berbari EF, et al. New definition for periprosthetic joint infection: from the Workgroup of the Musculoskeletal Infection Society. Clin Orthop. 2011;469(11):2992-2994.
Dilute Betadine Lavage Reduces Implant-Related Bacterial Burden in a Rabbit Knee Prosthetic Infection Model
Surgical site infection after arthroplasty causes substantial morbidity and potential mortality. Prosthetic joint infection (PJI) ranges from simple superficial wound infection and cellulitis to deep subfascial infection that involves the prosthesis. Consistent use of prophylactic antibiotics has reduced postoperative hip and knee arthroplasty infections to rates of 0.25% to 2%.1-4 Treatment of a patient with PJI commonly includes hospitalization, long-term intravenously administered antibiotics, resection arthroplasty, and staged reimplantation. The estimated cost of interventions reaches tens of millions of dollars annually in the United States and does not include the costs of psychosocial effects on patients and their families.5,6
Betadine (povidone-iodine) is a widely used antiseptic for skin and mucous membrane wounds and has been shown to be effective for the prevention of PJI.7 Dilute Betadine solution has been proposed as an aid in treatment of PJI.8 At a minimum concentration of 5%, cytotoxicity has been observed in chicken tibia osteoblasts.9 A balance of the bactericidal and cytotoxic activities of Betadine, while maintaining its efficacy against resistant organisms, such as methicillin-resistant Staphylococcus aureus (MRSA), is optimized at dilutions between 0.5% and 4%.10-14 We hypothesized that a dilute Betadine lavage of 3.5% would achieve a significant decrease in bacterial counts compared with an isolated saline lavage in an in vivo knee PJI model.
Materials and Methods
Animal Protocol
All surgical procedures were conducted according to the protocol approved by our institutional animal care and use committee. Using a power analysis and data obtained at our institution, we determined that 12 was the minimum number of animals needed to reach significance set at P < .05 and assuming a 50% decrease in colony-forming units (CFU) (SigmaStat Version 2.03; Aspire Software International, Ashburn, Virginia). Eight New Zealand White rabbits were used in our study; because significance was reached early, 12 were not needed. The average weight of the rabbits was 3.5 kg (weight range, 3.2-4.1 kg). All rabbits completed 1 week of acclimation before surgery.
Bacteria Preparation
A broth culture of methicillin-sensitive S aureus (MSSA) (ATCC 25923) was prepared 1 day before surgery. The bacteria were suspended in 5 mL of Trypticase Soy Broth (Becton Dickinson & Co, Franklin Lakes, New Jersey) and incubated at 37°C in a shaking incubator for 16 hours. The next day, the culture was centrifuged and irrigated twice with normal saline to remove the broth and prevent further growth. The bacteria were reconstituted in normal saline, and the concentration was standardized using a turbidity meter (LaMotte 2020e; LaMotte Co, Chestertown, Maryland), which correlated with 106 CFU/100 µl plated on trypticase soy agar plates with 10% sheep blood (Fisher Scientific, Pittsburgh, Pennsylvania).
Surgical and Postoperative Procedures
Our procedure was based on the New Zealand White rabbit knee PJI model.15 General anesthesia was induced with ketamine and xylazine, and maintained with isoflurane inhalation via a nose cone mask. Rabbits were positioned supine, and bilateral knees were shaved, prepped, and draped in a sterile fashion.
A 2-cm longitudinal incision was made over the lateral knee, and arthrotomy was performed, exposing the lateral collateral ligament attachment at the lateral femoral condyle. Using a 4-mm drill bit, a defect was drilled obliquely into the lateral femoral condyle, anterior to the lateral collateral ligament attachment. This produced a defect in the non-weight-bearing, nonarticulating portion of the knee. A fully threaded 4×14-mm stainless steel screw (Synthes, West Chester, Pennsylvania) with a U-shaped ultrahigh-molecular-weight polyethylene washer (Synthes) was inserted into the defect. The joint capsule was closed with a running 3-0 Vicryl suture (Ethicon, Somerville, New Jersey). The knee joint was inoculated with 100 µL of the S aureus preparation using a 22-gauge needle. The skin was closed with a 4-0 Biosyn suture (Ethicon). The procedure was repeated on the contralateral knee (Figures 1A, 1B).
Seven days after the initial surgery, the rabbits were returned to the operating room and were anesthetized, positioned, and prepped for surgery as detailed above. Ceftriaxone (20 mg/kg of body weight) was intravenously administered to all rabbits for the treatment procedure. For each rabbit, a control knee and an experimental knee were randomly assigned. A longitudinal incision was made, exposing the previously placed implants. The screw was loosened slightly to remove the U-shaped polyethylene washer. Each knee then underwent lavage 2 times, for 90 seconds each time, with 3.5% dilute Betadine solution (experimental knee) or with normal saline (control knee). Because Pseudomonas contamination has been reported with povidone-iodine taken from unsterilized bottles,16,17 packets of sterilized povidone-iodine (Aplicare; Clorox, Oakland, California) were used. After the irrigation was complete, a new sterile polyethylene washer was placed and the screw was tightened. The wound closure was repeated as detailed above.
Postoperative analgesia was provided based on a standard institutional animal care and use committee protocol. Rabbits were permitted full cage activity and nutrition ad libitum. Wound healing, body weight, and signs of distress were monitored daily.
Outcome Measures
Seven days after surgery, the rabbits were euthanized with administration of phenobarbital (100 mg/kg of body weight). Arterial blood samples were obtained from the auricular vein to ensure that the rabbits were not systemically infected. Using a sterile technique, the screw, polyethylene washer, lateral femoral condyle bone from the defect, and joint capsule were cultured. Harvested bone and soft tissues were weighed and immediately homogenized (PowerGen Model 35 Handheld Homogenizer; Thermo Fisher Scientific, Inc, Waltham, Massachusetts). Implants were sonicated (UBATH-Y; World Precision Instruments, Inc, Sarasota, Florida) in cold saline to obtain a sensitive culture.18
Bacterial quantification was determined by using trypticase soy agar plates after 24 hours of growth. Final CFU were calculated after serial dilutions and were standardized per gram of biopsied tissues.19 Members of the team were blinded to the treatment type.
Statistical Analysis
Statistical differences in mean bacterial burden were calculated independently for lateral condyle bone, joint capsule, polyethylene, and screws by conducting a Student t test.
Results
Treatment effect was higher than expected, and the study was terminated after 8 animals completed the protocol. All 8 rabbits tolerated the procedures well and were appropriately monitored during the postoperative period. No animals had signs of systemic infection or positive blood culture. All local cultures for screw, polyethylene washer, lateral femoral condyle defect, and joint capsule were positive.
Statistically significant decreases were shown in the bacterial burden of the Betadine-irrigated screws and the Betadine-irrigated polyethylene washers compared with the saline-irrigated controls. Betadine-irrigated screws grew an average of 7.16 × 101 CFU of S aureus/g, whereas screws from control knees grew an average of 1.45 × 103 CFU/g (P = .0003) (Figure 2). Betadine-treated washers grew an average of 1.28 × 103 CFU/g compared with 1.62 × 104 CFU/g for control washers (P =. 04) (Figure 3).
A trend toward decreased bacterial counts was shown in Betadine-treated soft tissues compared with saline-treated soft tissues, but the difference did not reach statistical significance (P = .9). Biopsied joint capsule from knees treated with Betadine grew an average of 2.84 × 104 CFU/g compared with an average of 3.16 × 104 CFU/g in control-rabbit knees (Figure 4). Cultured lateral condyle from Betadine-treated knees had an average bacterial load of 3.22 × 104 CFU/g compared with an average bacterial load of 1 × 105 CFU/g in control knees (Figure 4).
Discussion
Knees irrigated with Betadine showed a significant (P = .0003) decrease in metal implant–related S aureus bacterial counts by 20-fold and a significant (P < .05) decrease in polyethylene implant–related counts by more than 10-fold. This arthroplasty model used Betadine lavage as a treatment adjunct with intravenously administered antibiotics and polyethylene exchange. Our 1-week interval after the index procedure classifies the infection as an acute postoperative arthroplasty infection (occurring less than 4 weeks postoperatively).
The gold standard treatment for these infections is irrigation and débridement with component retention.18 The success rate has been reported to be as high as 71%20 but was closer to 44% in a study by Fridkin and colleagues,21 especially with more virulent bacteria. Staphylococcal species, higher American Society of Anesthesiologists scores, and frank pus around the prosthesis were markers of débridement failure in a recent study by Azzam and colleagues.18
The majority of postoperative joint arthroplasty infections are caused by S aureus, and the incidence of MRSA bacteria continues to rise.22 Community-acquired MRSA is increasing at an alarming rate and is now the predominant organism in skin and soft-tissue infections.23 Organism resistance also occurs at a cellular level by the formation of a glycocalyx layer, or biofilm. This layer assists in changing the phenotypic properties of the organism and decreases the efficacy of antibiotics.24 The self-produced layer of extracellular matrices, deoxyribonucleic acid, and polysaccharides attaches to inert material, preventing phagocytic action by neutrophils. In addition to antibacterial activity, povidone-iodine has antibiofilm activity against Staphylococcal species.25 The active ingredient targets the gene that produces biofilm. This correlates to our study in which the largest decrease in bacterial counts was noted on the implants.
The use of Betadine lavage has shown some promise in vivo as well. A prospective randomized controlled trial26 used 3.5% Betadine irrigation to prevent spine infection. No infections occurred in the Betadine group compared with a deep-infection rate of 2.9% in the control group. Brown and colleagues8 reviewed 1862 hip and knee arthroplasty cases before the use of Betadine lavage and 688 cases after the use of Betadine lavage and found a decrease in infection rate, from 0.97% to 0.15%. S aureus caused 13 of the 18 infections in the control group. These studies8,26 used Betadine lavage for prophylaxis and prevention of deep spine and arthroplasty infection. Betadine lavage as a treatment adjunct for acute arthroplasty infection has not been studied clinically. It has the potential to increase isolated incision and débridement success and to improve component survivorship.
Our arthroplasty model mimics an intra-articular environment and accounts for an implant–polyethylene interface.15 Limitations of our study include the use of MSSA as opposed to MRSA. However, povidone-iodine has the same effects on both MSSA and MRSA.12 We also treated our postoperative infection with 1 dose of antibiotics and not a course, although it should be noted that the single dose of ceftriaxone allowed us to isolate the independent effect of the Betadine lavage. A baseline level of infection severity could have been measured with cultures obtained at the time of irrigation and débridement. Also, a decrease in CFU does not directly correlate to a clinically significant outcome, such as a defined surgical site infection requiring intervention. Nevertheless, it is noteworthy that the decrease in bacterial counts on the stainless steel screws and polyethylene washers were maintained 1 week after the Betadine lavage.
Conclusion
Dilute Betadine lavage is a simple and inexpensive adjunct for the treatment of acute postoperative arthroplasty infection and may increase the rate of component retention. Additionally, the bactericidal and antibiofilm activities of Betadine may improve the effectiveness of systemic antibiotics. Further clinical investigation is warranted.
1. Wilson MG, Kelley K, Thornhill TS. Infection as a complication of total knee-replacement arthroplasty. Risk factors and treatment in sixty-seven cases. J Bone Joint Surg Am. 1990;72(6):878-883.
2. Ridgeway S, Wilson J, Charlet A, Kafatos G, Pearson A, Coello R. Infection of the surgical site after arthroplasty of the hip. J Bone Joint Surg Br. 2005;87(6):844-850.
3. Mahomed NN, Barrett JA, Katz JN, et al. Rates and outcomes of primary and revision total hip replacement in the United States medicare population. J Bone Joint Surg Am. 2003;85(1):27-32.
4. Mahomed NN, Barrett J, Katz JN, Baron JA, Wright J, Losina E. Epidemiology of total knee replacement in the United States Medicare population. J Bone Joint Surg Am. 2005;87(6):1222-1228.
5. Parvizi J, Zmistowski B, Adeli B. Periprosthetic joint infection: treatment options. Orthopedics. 2010;33(9):659.
6. Poultsides LA, Liaropoulos LL, Malizos KN. The socioeconomic impact of musculoskeletal infections. J Bone Joint Surg Am. 2010;92(11):e13.
7. Chundamala J, Wright JG. The efficacy and risks of using povidone-iodine irrigation to prevent surgical site infection: an evidence-based review. Can J Surg. 2007;50(6):473-481.
8. Brown NM, Cipriano CA, Moric M, Sporer SM, Della Valle CJ. Dilute betadine lavage before closure for the prevention of acute postoperative deep periprosthetic joint infection. J Arthroplasty. 2012;27(1):27-30.
9. Kaysinger KK, Nicholson NC, Ramp WK, Kellam JF. Toxic effects of wound irrigation solutions on cultured tibiae and osteoblasts. J Orthop Trauma. 1995;9(4):303-311.
10. Haley CE, Marling-Cason M, Smith JW, Luby JP, Mackowiak PA. Bactericidal activity of antiseptics against methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 1985;21(6):991-992.
11. Lacey RW, Catto A. Action of povidone-iodine against methicillin-sensitive and -resistant cultures of Staphylococcus aureus. Postgrad Med J. 1993;69(3 suppl):S78-S83.
12. McLure AR, Gordon J. In-vitro evaluation of povidone-iodine and chlorhexidine against methicillin-resistant Staphylococcus aureus. J Hosp Infect. 1992;21(4):291-299.
13. Suzuki J, Komatsuzawa H, Kozai K, Nagasaka N. In vitro susceptibility of Staphylococcus aureus including MRSA to four disinfectants. ASDC J Dent Child. 1997;64(4):260-263.
14. Yasuda T, Yoshimura S, Katsuno Y, et al. Comparison of bactericidal activities of various disinfectants against methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus. Postgrad Med J. 1993;69(3 suppl):S66-S69.
15. Craig MR, Poelstra KA, Sherrell JC, Kwon MS, Belzile EL, Brown TE. A novel total knee arthroplasty infection model in rabbits. J Orthop Res. 2005;23(5):1100-1104.
16. Hartman MB, Fehring TK, Jordan L, Norton HJ. Periprosthetic knee sepsis. The role of irrigation and debridement. Clin Orthop. 1991;273:113-118.
17. Mont MA, Waldman B, Banerjee C, Pacheco IH, Hungerford DS. Multiple irrigation, debridement, and retention of components in infected total knee arthroplasty. J Arthroplasty. 1997;12(4):426-433.
18. Azzam KA, Seeley M, Ghanem E, Austin MS, Purtill JJ, Parvizi J. Irrigation and debridement in the management of prosthetic joint infection: traditional indications revisited. J Arthroplasty. 2010;25(7):1022-1027.
19. Stall AC, Becker E, Ludwig SC, Gelb D, Poelstra KA. Reduction of postoperative spinal implant infection using gentamicin microspheres. Spine (Phila Pa 1976). 2009;34(5):479-483.
20. Hota B, Ellenbogen C, Hayden MK, Aroutcheva A, Rice TW, Weinstein RA. Community-associated methicillin-resistant Staphylococcus aureus skin and soft tissue infections at a public hospital: do public housing and incarceration amplify transmission? Arch Intern Med. 2007;167(10):1026-1033.
21. Fridkin SK, Hageman JC, Morrison M, et al, Active Bacterial Core Surveillance Program of the Emerging Infections Program Network. Methicillin-resistant Staphylococcus aureus disease in three communities. N Engl J Med. 2005;352(14):1436-1444.
22. Hosman AH, van der Mei HC, Bulstra SK, Busscher HJ, Neut D. Metal-on-metal bearings in total hip arthroplasties: influence of cobalt and chromium ions on bacterial growth and biofilm formation. J Biomed Mater Res A. 2009;88(3):711-716.
23. Oduwole KO, Glynn AA, Molony DC, et al. Anti-biofilm activity of sub-inhibitory povidone-iodine concentrations against Staphylococcus epidermidis and Staphylococcus aureus. J Orthop Res. 2010;28(9):1252-1256.
24. Cheng MT, Chang MC, Wang ST, Yu WK, Liu CL, Chen TH. Efficacy of dilute betadine solution irrigation in the prevention of postoperative infection of spinal surgery. Spine (Phila Pa 1976). 2005;30(15):1689-1693.
25. Anderson RL, Vess RW, Panlilio AL, Favero MS. Prolonged survival of Pseudomonas cepacia in commercially manufactured povidone-iodine. Appl Environ Microbiol. 1990;56(11):3598-3600.
26. Panlilio AL, Beck-Sague CM, Siegel JD, et al. Infections and pseudoinfections due to povidone-iodine solution contaminated with Pseudomonas cepacia. Clin Infect Dis. 1992;14(5):1078-1083.
Surgical site infection after arthroplasty causes substantial morbidity and potential mortality. Prosthetic joint infection (PJI) ranges from simple superficial wound infection and cellulitis to deep subfascial infection that involves the prosthesis. Consistent use of prophylactic antibiotics has reduced postoperative hip and knee arthroplasty infections to rates of 0.25% to 2%.1-4 Treatment of a patient with PJI commonly includes hospitalization, long-term intravenously administered antibiotics, resection arthroplasty, and staged reimplantation. The estimated cost of interventions reaches tens of millions of dollars annually in the United States and does not include the costs of psychosocial effects on patients and their families.5,6
Betadine (povidone-iodine) is a widely used antiseptic for skin and mucous membrane wounds and has been shown to be effective for the prevention of PJI.7 Dilute Betadine solution has been proposed as an aid in treatment of PJI.8 At a minimum concentration of 5%, cytotoxicity has been observed in chicken tibia osteoblasts.9 A balance of the bactericidal and cytotoxic activities of Betadine, while maintaining its efficacy against resistant organisms, such as methicillin-resistant Staphylococcus aureus (MRSA), is optimized at dilutions between 0.5% and 4%.10-14 We hypothesized that a dilute Betadine lavage of 3.5% would achieve a significant decrease in bacterial counts compared with an isolated saline lavage in an in vivo knee PJI model.
Materials and Methods
Animal Protocol
All surgical procedures were conducted according to the protocol approved by our institutional animal care and use committee. Using a power analysis and data obtained at our institution, we determined that 12 was the minimum number of animals needed to reach significance set at P < .05 and assuming a 50% decrease in colony-forming units (CFU) (SigmaStat Version 2.03; Aspire Software International, Ashburn, Virginia). Eight New Zealand White rabbits were used in our study; because significance was reached early, 12 were not needed. The average weight of the rabbits was 3.5 kg (weight range, 3.2-4.1 kg). All rabbits completed 1 week of acclimation before surgery.
Bacteria Preparation
A broth culture of methicillin-sensitive S aureus (MSSA) (ATCC 25923) was prepared 1 day before surgery. The bacteria were suspended in 5 mL of Trypticase Soy Broth (Becton Dickinson & Co, Franklin Lakes, New Jersey) and incubated at 37°C in a shaking incubator for 16 hours. The next day, the culture was centrifuged and irrigated twice with normal saline to remove the broth and prevent further growth. The bacteria were reconstituted in normal saline, and the concentration was standardized using a turbidity meter (LaMotte 2020e; LaMotte Co, Chestertown, Maryland), which correlated with 106 CFU/100 µl plated on trypticase soy agar plates with 10% sheep blood (Fisher Scientific, Pittsburgh, Pennsylvania).
Surgical and Postoperative Procedures
Our procedure was based on the New Zealand White rabbit knee PJI model.15 General anesthesia was induced with ketamine and xylazine, and maintained with isoflurane inhalation via a nose cone mask. Rabbits were positioned supine, and bilateral knees were shaved, prepped, and draped in a sterile fashion.
A 2-cm longitudinal incision was made over the lateral knee, and arthrotomy was performed, exposing the lateral collateral ligament attachment at the lateral femoral condyle. Using a 4-mm drill bit, a defect was drilled obliquely into the lateral femoral condyle, anterior to the lateral collateral ligament attachment. This produced a defect in the non-weight-bearing, nonarticulating portion of the knee. A fully threaded 4×14-mm stainless steel screw (Synthes, West Chester, Pennsylvania) with a U-shaped ultrahigh-molecular-weight polyethylene washer (Synthes) was inserted into the defect. The joint capsule was closed with a running 3-0 Vicryl suture (Ethicon, Somerville, New Jersey). The knee joint was inoculated with 100 µL of the S aureus preparation using a 22-gauge needle. The skin was closed with a 4-0 Biosyn suture (Ethicon). The procedure was repeated on the contralateral knee (Figures 1A, 1B).
Seven days after the initial surgery, the rabbits were returned to the operating room and were anesthetized, positioned, and prepped for surgery as detailed above. Ceftriaxone (20 mg/kg of body weight) was intravenously administered to all rabbits for the treatment procedure. For each rabbit, a control knee and an experimental knee were randomly assigned. A longitudinal incision was made, exposing the previously placed implants. The screw was loosened slightly to remove the U-shaped polyethylene washer. Each knee then underwent lavage 2 times, for 90 seconds each time, with 3.5% dilute Betadine solution (experimental knee) or with normal saline (control knee). Because Pseudomonas contamination has been reported with povidone-iodine taken from unsterilized bottles,16,17 packets of sterilized povidone-iodine (Aplicare; Clorox, Oakland, California) were used. After the irrigation was complete, a new sterile polyethylene washer was placed and the screw was tightened. The wound closure was repeated as detailed above.
Postoperative analgesia was provided based on a standard institutional animal care and use committee protocol. Rabbits were permitted full cage activity and nutrition ad libitum. Wound healing, body weight, and signs of distress were monitored daily.
Outcome Measures
Seven days after surgery, the rabbits were euthanized with administration of phenobarbital (100 mg/kg of body weight). Arterial blood samples were obtained from the auricular vein to ensure that the rabbits were not systemically infected. Using a sterile technique, the screw, polyethylene washer, lateral femoral condyle bone from the defect, and joint capsule were cultured. Harvested bone and soft tissues were weighed and immediately homogenized (PowerGen Model 35 Handheld Homogenizer; Thermo Fisher Scientific, Inc, Waltham, Massachusetts). Implants were sonicated (UBATH-Y; World Precision Instruments, Inc, Sarasota, Florida) in cold saline to obtain a sensitive culture.18
Bacterial quantification was determined by using trypticase soy agar plates after 24 hours of growth. Final CFU were calculated after serial dilutions and were standardized per gram of biopsied tissues.19 Members of the team were blinded to the treatment type.
Statistical Analysis
Statistical differences in mean bacterial burden were calculated independently for lateral condyle bone, joint capsule, polyethylene, and screws by conducting a Student t test.
Results
Treatment effect was higher than expected, and the study was terminated after 8 animals completed the protocol. All 8 rabbits tolerated the procedures well and were appropriately monitored during the postoperative period. No animals had signs of systemic infection or positive blood culture. All local cultures for screw, polyethylene washer, lateral femoral condyle defect, and joint capsule were positive.
Statistically significant decreases were shown in the bacterial burden of the Betadine-irrigated screws and the Betadine-irrigated polyethylene washers compared with the saline-irrigated controls. Betadine-irrigated screws grew an average of 7.16 × 101 CFU of S aureus/g, whereas screws from control knees grew an average of 1.45 × 103 CFU/g (P = .0003) (Figure 2). Betadine-treated washers grew an average of 1.28 × 103 CFU/g compared with 1.62 × 104 CFU/g for control washers (P =. 04) (Figure 3).
A trend toward decreased bacterial counts was shown in Betadine-treated soft tissues compared with saline-treated soft tissues, but the difference did not reach statistical significance (P = .9). Biopsied joint capsule from knees treated with Betadine grew an average of 2.84 × 104 CFU/g compared with an average of 3.16 × 104 CFU/g in control-rabbit knees (Figure 4). Cultured lateral condyle from Betadine-treated knees had an average bacterial load of 3.22 × 104 CFU/g compared with an average bacterial load of 1 × 105 CFU/g in control knees (Figure 4).
Discussion
Knees irrigated with Betadine showed a significant (P = .0003) decrease in metal implant–related S aureus bacterial counts by 20-fold and a significant (P < .05) decrease in polyethylene implant–related counts by more than 10-fold. This arthroplasty model used Betadine lavage as a treatment adjunct with intravenously administered antibiotics and polyethylene exchange. Our 1-week interval after the index procedure classifies the infection as an acute postoperative arthroplasty infection (occurring less than 4 weeks postoperatively).
The gold standard treatment for these infections is irrigation and débridement with component retention.18 The success rate has been reported to be as high as 71%20 but was closer to 44% in a study by Fridkin and colleagues,21 especially with more virulent bacteria. Staphylococcal species, higher American Society of Anesthesiologists scores, and frank pus around the prosthesis were markers of débridement failure in a recent study by Azzam and colleagues.18
The majority of postoperative joint arthroplasty infections are caused by S aureus, and the incidence of MRSA bacteria continues to rise.22 Community-acquired MRSA is increasing at an alarming rate and is now the predominant organism in skin and soft-tissue infections.23 Organism resistance also occurs at a cellular level by the formation of a glycocalyx layer, or biofilm. This layer assists in changing the phenotypic properties of the organism and decreases the efficacy of antibiotics.24 The self-produced layer of extracellular matrices, deoxyribonucleic acid, and polysaccharides attaches to inert material, preventing phagocytic action by neutrophils. In addition to antibacterial activity, povidone-iodine has antibiofilm activity against Staphylococcal species.25 The active ingredient targets the gene that produces biofilm. This correlates to our study in which the largest decrease in bacterial counts was noted on the implants.
The use of Betadine lavage has shown some promise in vivo as well. A prospective randomized controlled trial26 used 3.5% Betadine irrigation to prevent spine infection. No infections occurred in the Betadine group compared with a deep-infection rate of 2.9% in the control group. Brown and colleagues8 reviewed 1862 hip and knee arthroplasty cases before the use of Betadine lavage and 688 cases after the use of Betadine lavage and found a decrease in infection rate, from 0.97% to 0.15%. S aureus caused 13 of the 18 infections in the control group. These studies8,26 used Betadine lavage for prophylaxis and prevention of deep spine and arthroplasty infection. Betadine lavage as a treatment adjunct for acute arthroplasty infection has not been studied clinically. It has the potential to increase isolated incision and débridement success and to improve component survivorship.
Our arthroplasty model mimics an intra-articular environment and accounts for an implant–polyethylene interface.15 Limitations of our study include the use of MSSA as opposed to MRSA. However, povidone-iodine has the same effects on both MSSA and MRSA.12 We also treated our postoperative infection with 1 dose of antibiotics and not a course, although it should be noted that the single dose of ceftriaxone allowed us to isolate the independent effect of the Betadine lavage. A baseline level of infection severity could have been measured with cultures obtained at the time of irrigation and débridement. Also, a decrease in CFU does not directly correlate to a clinically significant outcome, such as a defined surgical site infection requiring intervention. Nevertheless, it is noteworthy that the decrease in bacterial counts on the stainless steel screws and polyethylene washers were maintained 1 week after the Betadine lavage.
Conclusion
Dilute Betadine lavage is a simple and inexpensive adjunct for the treatment of acute postoperative arthroplasty infection and may increase the rate of component retention. Additionally, the bactericidal and antibiofilm activities of Betadine may improve the effectiveness of systemic antibiotics. Further clinical investigation is warranted.
Surgical site infection after arthroplasty causes substantial morbidity and potential mortality. Prosthetic joint infection (PJI) ranges from simple superficial wound infection and cellulitis to deep subfascial infection that involves the prosthesis. Consistent use of prophylactic antibiotics has reduced postoperative hip and knee arthroplasty infections to rates of 0.25% to 2%.1-4 Treatment of a patient with PJI commonly includes hospitalization, long-term intravenously administered antibiotics, resection arthroplasty, and staged reimplantation. The estimated cost of interventions reaches tens of millions of dollars annually in the United States and does not include the costs of psychosocial effects on patients and their families.5,6
Betadine (povidone-iodine) is a widely used antiseptic for skin and mucous membrane wounds and has been shown to be effective for the prevention of PJI.7 Dilute Betadine solution has been proposed as an aid in treatment of PJI.8 At a minimum concentration of 5%, cytotoxicity has been observed in chicken tibia osteoblasts.9 A balance of the bactericidal and cytotoxic activities of Betadine, while maintaining its efficacy against resistant organisms, such as methicillin-resistant Staphylococcus aureus (MRSA), is optimized at dilutions between 0.5% and 4%.10-14 We hypothesized that a dilute Betadine lavage of 3.5% would achieve a significant decrease in bacterial counts compared with an isolated saline lavage in an in vivo knee PJI model.
Materials and Methods
Animal Protocol
All surgical procedures were conducted according to the protocol approved by our institutional animal care and use committee. Using a power analysis and data obtained at our institution, we determined that 12 was the minimum number of animals needed to reach significance set at P < .05 and assuming a 50% decrease in colony-forming units (CFU) (SigmaStat Version 2.03; Aspire Software International, Ashburn, Virginia). Eight New Zealand White rabbits were used in our study; because significance was reached early, 12 were not needed. The average weight of the rabbits was 3.5 kg (weight range, 3.2-4.1 kg). All rabbits completed 1 week of acclimation before surgery.
Bacteria Preparation
A broth culture of methicillin-sensitive S aureus (MSSA) (ATCC 25923) was prepared 1 day before surgery. The bacteria were suspended in 5 mL of Trypticase Soy Broth (Becton Dickinson & Co, Franklin Lakes, New Jersey) and incubated at 37°C in a shaking incubator for 16 hours. The next day, the culture was centrifuged and irrigated twice with normal saline to remove the broth and prevent further growth. The bacteria were reconstituted in normal saline, and the concentration was standardized using a turbidity meter (LaMotte 2020e; LaMotte Co, Chestertown, Maryland), which correlated with 106 CFU/100 µl plated on trypticase soy agar plates with 10% sheep blood (Fisher Scientific, Pittsburgh, Pennsylvania).
Surgical and Postoperative Procedures
Our procedure was based on the New Zealand White rabbit knee PJI model.15 General anesthesia was induced with ketamine and xylazine, and maintained with isoflurane inhalation via a nose cone mask. Rabbits were positioned supine, and bilateral knees were shaved, prepped, and draped in a sterile fashion.
A 2-cm longitudinal incision was made over the lateral knee, and arthrotomy was performed, exposing the lateral collateral ligament attachment at the lateral femoral condyle. Using a 4-mm drill bit, a defect was drilled obliquely into the lateral femoral condyle, anterior to the lateral collateral ligament attachment. This produced a defect in the non-weight-bearing, nonarticulating portion of the knee. A fully threaded 4×14-mm stainless steel screw (Synthes, West Chester, Pennsylvania) with a U-shaped ultrahigh-molecular-weight polyethylene washer (Synthes) was inserted into the defect. The joint capsule was closed with a running 3-0 Vicryl suture (Ethicon, Somerville, New Jersey). The knee joint was inoculated with 100 µL of the S aureus preparation using a 22-gauge needle. The skin was closed with a 4-0 Biosyn suture (Ethicon). The procedure was repeated on the contralateral knee (Figures 1A, 1B).
Seven days after the initial surgery, the rabbits were returned to the operating room and were anesthetized, positioned, and prepped for surgery as detailed above. Ceftriaxone (20 mg/kg of body weight) was intravenously administered to all rabbits for the treatment procedure. For each rabbit, a control knee and an experimental knee were randomly assigned. A longitudinal incision was made, exposing the previously placed implants. The screw was loosened slightly to remove the U-shaped polyethylene washer. Each knee then underwent lavage 2 times, for 90 seconds each time, with 3.5% dilute Betadine solution (experimental knee) or with normal saline (control knee). Because Pseudomonas contamination has been reported with povidone-iodine taken from unsterilized bottles,16,17 packets of sterilized povidone-iodine (Aplicare; Clorox, Oakland, California) were used. After the irrigation was complete, a new sterile polyethylene washer was placed and the screw was tightened. The wound closure was repeated as detailed above.
Postoperative analgesia was provided based on a standard institutional animal care and use committee protocol. Rabbits were permitted full cage activity and nutrition ad libitum. Wound healing, body weight, and signs of distress were monitored daily.
Outcome Measures
Seven days after surgery, the rabbits were euthanized with administration of phenobarbital (100 mg/kg of body weight). Arterial blood samples were obtained from the auricular vein to ensure that the rabbits were not systemically infected. Using a sterile technique, the screw, polyethylene washer, lateral femoral condyle bone from the defect, and joint capsule were cultured. Harvested bone and soft tissues were weighed and immediately homogenized (PowerGen Model 35 Handheld Homogenizer; Thermo Fisher Scientific, Inc, Waltham, Massachusetts). Implants were sonicated (UBATH-Y; World Precision Instruments, Inc, Sarasota, Florida) in cold saline to obtain a sensitive culture.18
Bacterial quantification was determined by using trypticase soy agar plates after 24 hours of growth. Final CFU were calculated after serial dilutions and were standardized per gram of biopsied tissues.19 Members of the team were blinded to the treatment type.
Statistical Analysis
Statistical differences in mean bacterial burden were calculated independently for lateral condyle bone, joint capsule, polyethylene, and screws by conducting a Student t test.
Results
Treatment effect was higher than expected, and the study was terminated after 8 animals completed the protocol. All 8 rabbits tolerated the procedures well and were appropriately monitored during the postoperative period. No animals had signs of systemic infection or positive blood culture. All local cultures for screw, polyethylene washer, lateral femoral condyle defect, and joint capsule were positive.
Statistically significant decreases were shown in the bacterial burden of the Betadine-irrigated screws and the Betadine-irrigated polyethylene washers compared with the saline-irrigated controls. Betadine-irrigated screws grew an average of 7.16 × 101 CFU of S aureus/g, whereas screws from control knees grew an average of 1.45 × 103 CFU/g (P = .0003) (Figure 2). Betadine-treated washers grew an average of 1.28 × 103 CFU/g compared with 1.62 × 104 CFU/g for control washers (P =. 04) (Figure 3).
A trend toward decreased bacterial counts was shown in Betadine-treated soft tissues compared with saline-treated soft tissues, but the difference did not reach statistical significance (P = .9). Biopsied joint capsule from knees treated with Betadine grew an average of 2.84 × 104 CFU/g compared with an average of 3.16 × 104 CFU/g in control-rabbit knees (Figure 4). Cultured lateral condyle from Betadine-treated knees had an average bacterial load of 3.22 × 104 CFU/g compared with an average bacterial load of 1 × 105 CFU/g in control knees (Figure 4).
Discussion
Knees irrigated with Betadine showed a significant (P = .0003) decrease in metal implant–related S aureus bacterial counts by 20-fold and a significant (P < .05) decrease in polyethylene implant–related counts by more than 10-fold. This arthroplasty model used Betadine lavage as a treatment adjunct with intravenously administered antibiotics and polyethylene exchange. Our 1-week interval after the index procedure classifies the infection as an acute postoperative arthroplasty infection (occurring less than 4 weeks postoperatively).
The gold standard treatment for these infections is irrigation and débridement with component retention.18 The success rate has been reported to be as high as 71%20 but was closer to 44% in a study by Fridkin and colleagues,21 especially with more virulent bacteria. Staphylococcal species, higher American Society of Anesthesiologists scores, and frank pus around the prosthesis were markers of débridement failure in a recent study by Azzam and colleagues.18
The majority of postoperative joint arthroplasty infections are caused by S aureus, and the incidence of MRSA bacteria continues to rise.22 Community-acquired MRSA is increasing at an alarming rate and is now the predominant organism in skin and soft-tissue infections.23 Organism resistance also occurs at a cellular level by the formation of a glycocalyx layer, or biofilm. This layer assists in changing the phenotypic properties of the organism and decreases the efficacy of antibiotics.24 The self-produced layer of extracellular matrices, deoxyribonucleic acid, and polysaccharides attaches to inert material, preventing phagocytic action by neutrophils. In addition to antibacterial activity, povidone-iodine has antibiofilm activity against Staphylococcal species.25 The active ingredient targets the gene that produces biofilm. This correlates to our study in which the largest decrease in bacterial counts was noted on the implants.
The use of Betadine lavage has shown some promise in vivo as well. A prospective randomized controlled trial26 used 3.5% Betadine irrigation to prevent spine infection. No infections occurred in the Betadine group compared with a deep-infection rate of 2.9% in the control group. Brown and colleagues8 reviewed 1862 hip and knee arthroplasty cases before the use of Betadine lavage and 688 cases after the use of Betadine lavage and found a decrease in infection rate, from 0.97% to 0.15%. S aureus caused 13 of the 18 infections in the control group. These studies8,26 used Betadine lavage for prophylaxis and prevention of deep spine and arthroplasty infection. Betadine lavage as a treatment adjunct for acute arthroplasty infection has not been studied clinically. It has the potential to increase isolated incision and débridement success and to improve component survivorship.
Our arthroplasty model mimics an intra-articular environment and accounts for an implant–polyethylene interface.15 Limitations of our study include the use of MSSA as opposed to MRSA. However, povidone-iodine has the same effects on both MSSA and MRSA.12 We also treated our postoperative infection with 1 dose of antibiotics and not a course, although it should be noted that the single dose of ceftriaxone allowed us to isolate the independent effect of the Betadine lavage. A baseline level of infection severity could have been measured with cultures obtained at the time of irrigation and débridement. Also, a decrease in CFU does not directly correlate to a clinically significant outcome, such as a defined surgical site infection requiring intervention. Nevertheless, it is noteworthy that the decrease in bacterial counts on the stainless steel screws and polyethylene washers were maintained 1 week after the Betadine lavage.
Conclusion
Dilute Betadine lavage is a simple and inexpensive adjunct for the treatment of acute postoperative arthroplasty infection and may increase the rate of component retention. Additionally, the bactericidal and antibiofilm activities of Betadine may improve the effectiveness of systemic antibiotics. Further clinical investigation is warranted.
1. Wilson MG, Kelley K, Thornhill TS. Infection as a complication of total knee-replacement arthroplasty. Risk factors and treatment in sixty-seven cases. J Bone Joint Surg Am. 1990;72(6):878-883.
2. Ridgeway S, Wilson J, Charlet A, Kafatos G, Pearson A, Coello R. Infection of the surgical site after arthroplasty of the hip. J Bone Joint Surg Br. 2005;87(6):844-850.
3. Mahomed NN, Barrett JA, Katz JN, et al. Rates and outcomes of primary and revision total hip replacement in the United States medicare population. J Bone Joint Surg Am. 2003;85(1):27-32.
4. Mahomed NN, Barrett J, Katz JN, Baron JA, Wright J, Losina E. Epidemiology of total knee replacement in the United States Medicare population. J Bone Joint Surg Am. 2005;87(6):1222-1228.
5. Parvizi J, Zmistowski B, Adeli B. Periprosthetic joint infection: treatment options. Orthopedics. 2010;33(9):659.
6. Poultsides LA, Liaropoulos LL, Malizos KN. The socioeconomic impact of musculoskeletal infections. J Bone Joint Surg Am. 2010;92(11):e13.
7. Chundamala J, Wright JG. The efficacy and risks of using povidone-iodine irrigation to prevent surgical site infection: an evidence-based review. Can J Surg. 2007;50(6):473-481.
8. Brown NM, Cipriano CA, Moric M, Sporer SM, Della Valle CJ. Dilute betadine lavage before closure for the prevention of acute postoperative deep periprosthetic joint infection. J Arthroplasty. 2012;27(1):27-30.
9. Kaysinger KK, Nicholson NC, Ramp WK, Kellam JF. Toxic effects of wound irrigation solutions on cultured tibiae and osteoblasts. J Orthop Trauma. 1995;9(4):303-311.
10. Haley CE, Marling-Cason M, Smith JW, Luby JP, Mackowiak PA. Bactericidal activity of antiseptics against methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 1985;21(6):991-992.
11. Lacey RW, Catto A. Action of povidone-iodine against methicillin-sensitive and -resistant cultures of Staphylococcus aureus. Postgrad Med J. 1993;69(3 suppl):S78-S83.
12. McLure AR, Gordon J. In-vitro evaluation of povidone-iodine and chlorhexidine against methicillin-resistant Staphylococcus aureus. J Hosp Infect. 1992;21(4):291-299.
13. Suzuki J, Komatsuzawa H, Kozai K, Nagasaka N. In vitro susceptibility of Staphylococcus aureus including MRSA to four disinfectants. ASDC J Dent Child. 1997;64(4):260-263.
14. Yasuda T, Yoshimura S, Katsuno Y, et al. Comparison of bactericidal activities of various disinfectants against methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus. Postgrad Med J. 1993;69(3 suppl):S66-S69.
15. Craig MR, Poelstra KA, Sherrell JC, Kwon MS, Belzile EL, Brown TE. A novel total knee arthroplasty infection model in rabbits. J Orthop Res. 2005;23(5):1100-1104.
16. Hartman MB, Fehring TK, Jordan L, Norton HJ. Periprosthetic knee sepsis. The role of irrigation and debridement. Clin Orthop. 1991;273:113-118.
17. Mont MA, Waldman B, Banerjee C, Pacheco IH, Hungerford DS. Multiple irrigation, debridement, and retention of components in infected total knee arthroplasty. J Arthroplasty. 1997;12(4):426-433.
18. Azzam KA, Seeley M, Ghanem E, Austin MS, Purtill JJ, Parvizi J. Irrigation and debridement in the management of prosthetic joint infection: traditional indications revisited. J Arthroplasty. 2010;25(7):1022-1027.
19. Stall AC, Becker E, Ludwig SC, Gelb D, Poelstra KA. Reduction of postoperative spinal implant infection using gentamicin microspheres. Spine (Phila Pa 1976). 2009;34(5):479-483.
20. Hota B, Ellenbogen C, Hayden MK, Aroutcheva A, Rice TW, Weinstein RA. Community-associated methicillin-resistant Staphylococcus aureus skin and soft tissue infections at a public hospital: do public housing and incarceration amplify transmission? Arch Intern Med. 2007;167(10):1026-1033.
21. Fridkin SK, Hageman JC, Morrison M, et al, Active Bacterial Core Surveillance Program of the Emerging Infections Program Network. Methicillin-resistant Staphylococcus aureus disease in three communities. N Engl J Med. 2005;352(14):1436-1444.
22. Hosman AH, van der Mei HC, Bulstra SK, Busscher HJ, Neut D. Metal-on-metal bearings in total hip arthroplasties: influence of cobalt and chromium ions on bacterial growth and biofilm formation. J Biomed Mater Res A. 2009;88(3):711-716.
23. Oduwole KO, Glynn AA, Molony DC, et al. Anti-biofilm activity of sub-inhibitory povidone-iodine concentrations against Staphylococcus epidermidis and Staphylococcus aureus. J Orthop Res. 2010;28(9):1252-1256.
24. Cheng MT, Chang MC, Wang ST, Yu WK, Liu CL, Chen TH. Efficacy of dilute betadine solution irrigation in the prevention of postoperative infection of spinal surgery. Spine (Phila Pa 1976). 2005;30(15):1689-1693.
25. Anderson RL, Vess RW, Panlilio AL, Favero MS. Prolonged survival of Pseudomonas cepacia in commercially manufactured povidone-iodine. Appl Environ Microbiol. 1990;56(11):3598-3600.
26. Panlilio AL, Beck-Sague CM, Siegel JD, et al. Infections and pseudoinfections due to povidone-iodine solution contaminated with Pseudomonas cepacia. Clin Infect Dis. 1992;14(5):1078-1083.
1. Wilson MG, Kelley K, Thornhill TS. Infection as a complication of total knee-replacement arthroplasty. Risk factors and treatment in sixty-seven cases. J Bone Joint Surg Am. 1990;72(6):878-883.
2. Ridgeway S, Wilson J, Charlet A, Kafatos G, Pearson A, Coello R. Infection of the surgical site after arthroplasty of the hip. J Bone Joint Surg Br. 2005;87(6):844-850.
3. Mahomed NN, Barrett JA, Katz JN, et al. Rates and outcomes of primary and revision total hip replacement in the United States medicare population. J Bone Joint Surg Am. 2003;85(1):27-32.
4. Mahomed NN, Barrett J, Katz JN, Baron JA, Wright J, Losina E. Epidemiology of total knee replacement in the United States Medicare population. J Bone Joint Surg Am. 2005;87(6):1222-1228.
5. Parvizi J, Zmistowski B, Adeli B. Periprosthetic joint infection: treatment options. Orthopedics. 2010;33(9):659.
6. Poultsides LA, Liaropoulos LL, Malizos KN. The socioeconomic impact of musculoskeletal infections. J Bone Joint Surg Am. 2010;92(11):e13.
7. Chundamala J, Wright JG. The efficacy and risks of using povidone-iodine irrigation to prevent surgical site infection: an evidence-based review. Can J Surg. 2007;50(6):473-481.
8. Brown NM, Cipriano CA, Moric M, Sporer SM, Della Valle CJ. Dilute betadine lavage before closure for the prevention of acute postoperative deep periprosthetic joint infection. J Arthroplasty. 2012;27(1):27-30.
9. Kaysinger KK, Nicholson NC, Ramp WK, Kellam JF. Toxic effects of wound irrigation solutions on cultured tibiae and osteoblasts. J Orthop Trauma. 1995;9(4):303-311.
10. Haley CE, Marling-Cason M, Smith JW, Luby JP, Mackowiak PA. Bactericidal activity of antiseptics against methicillin-resistant Staphylococcus aureus. J Clin Microbiol. 1985;21(6):991-992.
11. Lacey RW, Catto A. Action of povidone-iodine against methicillin-sensitive and -resistant cultures of Staphylococcus aureus. Postgrad Med J. 1993;69(3 suppl):S78-S83.
12. McLure AR, Gordon J. In-vitro evaluation of povidone-iodine and chlorhexidine against methicillin-resistant Staphylococcus aureus. J Hosp Infect. 1992;21(4):291-299.
13. Suzuki J, Komatsuzawa H, Kozai K, Nagasaka N. In vitro susceptibility of Staphylococcus aureus including MRSA to four disinfectants. ASDC J Dent Child. 1997;64(4):260-263.
14. Yasuda T, Yoshimura S, Katsuno Y, et al. Comparison of bactericidal activities of various disinfectants against methicillin-sensitive Staphylococcus aureus and methicillin-resistant Staphylococcus aureus. Postgrad Med J. 1993;69(3 suppl):S66-S69.
15. Craig MR, Poelstra KA, Sherrell JC, Kwon MS, Belzile EL, Brown TE. A novel total knee arthroplasty infection model in rabbits. J Orthop Res. 2005;23(5):1100-1104.
16. Hartman MB, Fehring TK, Jordan L, Norton HJ. Periprosthetic knee sepsis. The role of irrigation and debridement. Clin Orthop. 1991;273:113-118.
17. Mont MA, Waldman B, Banerjee C, Pacheco IH, Hungerford DS. Multiple irrigation, debridement, and retention of components in infected total knee arthroplasty. J Arthroplasty. 1997;12(4):426-433.
18. Azzam KA, Seeley M, Ghanem E, Austin MS, Purtill JJ, Parvizi J. Irrigation and debridement in the management of prosthetic joint infection: traditional indications revisited. J Arthroplasty. 2010;25(7):1022-1027.
19. Stall AC, Becker E, Ludwig SC, Gelb D, Poelstra KA. Reduction of postoperative spinal implant infection using gentamicin microspheres. Spine (Phila Pa 1976). 2009;34(5):479-483.
20. Hota B, Ellenbogen C, Hayden MK, Aroutcheva A, Rice TW, Weinstein RA. Community-associated methicillin-resistant Staphylococcus aureus skin and soft tissue infections at a public hospital: do public housing and incarceration amplify transmission? Arch Intern Med. 2007;167(10):1026-1033.
21. Fridkin SK, Hageman JC, Morrison M, et al, Active Bacterial Core Surveillance Program of the Emerging Infections Program Network. Methicillin-resistant Staphylococcus aureus disease in three communities. N Engl J Med. 2005;352(14):1436-1444.
22. Hosman AH, van der Mei HC, Bulstra SK, Busscher HJ, Neut D. Metal-on-metal bearings in total hip arthroplasties: influence of cobalt and chromium ions on bacterial growth and biofilm formation. J Biomed Mater Res A. 2009;88(3):711-716.
23. Oduwole KO, Glynn AA, Molony DC, et al. Anti-biofilm activity of sub-inhibitory povidone-iodine concentrations against Staphylococcus epidermidis and Staphylococcus aureus. J Orthop Res. 2010;28(9):1252-1256.
24. Cheng MT, Chang MC, Wang ST, Yu WK, Liu CL, Chen TH. Efficacy of dilute betadine solution irrigation in the prevention of postoperative infection of spinal surgery. Spine (Phila Pa 1976). 2005;30(15):1689-1693.
25. Anderson RL, Vess RW, Panlilio AL, Favero MS. Prolonged survival of Pseudomonas cepacia in commercially manufactured povidone-iodine. Appl Environ Microbiol. 1990;56(11):3598-3600.
26. Panlilio AL, Beck-Sague CM, Siegel JD, et al. Infections and pseudoinfections due to povidone-iodine solution contaminated with Pseudomonas cepacia. Clin Infect Dis. 1992;14(5):1078-1083.
Meniscus Regenerated With 3D-Printed Implant in Animal Study
Researchers have honed in on a way to replace the meniscus, using a personalized 3D-printed implant in sheep, according to a study published online December 10, 2014, in Science Translational Medicine. The therapy, successfully tested in sheep, could provide the first effective and long-lasting repair of damaged menisci.
“At present, there’s little that orthopedists can do to regenerate a torn knee meniscus,” said lead study author Jeremy Mao, DDS, PhD, the Edwin S. Robinson Professor of Dentistry in Orthopedic Surgery at Columbia University Medical Center in New York. “In contrast, we’re jumpstarting the process within the body, using factors that promote endogenous stem cells for tissue regeneration.”
This process was tested on 11 sheep. Knees of sheep were chosen because of their close resemblance to the human knee. In this study, the sheep were randomized to have part of their knee meniscus replaced with a protein-infused 3D scaffold or a 3D scaffold without protein.
Dr. Mao’s approach starts with MRI scans of the intact meniscus in the undamaged knee, which is then converted into a 3D image. Data from the image are then used to drive a 3D printer, which produces a scaffold in the exact shape of the meniscus, down to a resolution of 10 microns. The scaffold is infused with connective growth factor (CTGF) and transforming growth factor β3 (TGFβ3). Dr. Mao’s team found that sequential delivery of these two proteins attracts existing stem cells from the body and induces them to form meniscal tissue.
After three months, treated animals were walking normally. In a postmortem analysis, the researchers found that the regenerated meniscus in the treatment group had structural and mechanical properties very similar to those of natural meniscus. In sheep, the meniscus regenerates at a rate of about 4 to 6 weeks. Eventually, the scaffold dissolves and is eliminated by the body.
Suggested Reading
Lee CH, Rodeo SA, Fortier LA, et al. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci Transl Med. 2014 Dec 10;6(266):266ra171.
Researchers have honed in on a way to replace the meniscus, using a personalized 3D-printed implant in sheep, according to a study published online December 10, 2014, in Science Translational Medicine. The therapy, successfully tested in sheep, could provide the first effective and long-lasting repair of damaged menisci.
“At present, there’s little that orthopedists can do to regenerate a torn knee meniscus,” said lead study author Jeremy Mao, DDS, PhD, the Edwin S. Robinson Professor of Dentistry in Orthopedic Surgery at Columbia University Medical Center in New York. “In contrast, we’re jumpstarting the process within the body, using factors that promote endogenous stem cells for tissue regeneration.”
This process was tested on 11 sheep. Knees of sheep were chosen because of their close resemblance to the human knee. In this study, the sheep were randomized to have part of their knee meniscus replaced with a protein-infused 3D scaffold or a 3D scaffold without protein.
Dr. Mao’s approach starts with MRI scans of the intact meniscus in the undamaged knee, which is then converted into a 3D image. Data from the image are then used to drive a 3D printer, which produces a scaffold in the exact shape of the meniscus, down to a resolution of 10 microns. The scaffold is infused with connective growth factor (CTGF) and transforming growth factor β3 (TGFβ3). Dr. Mao’s team found that sequential delivery of these two proteins attracts existing stem cells from the body and induces them to form meniscal tissue.
After three months, treated animals were walking normally. In a postmortem analysis, the researchers found that the regenerated meniscus in the treatment group had structural and mechanical properties very similar to those of natural meniscus. In sheep, the meniscus regenerates at a rate of about 4 to 6 weeks. Eventually, the scaffold dissolves and is eliminated by the body.
Researchers have honed in on a way to replace the meniscus, using a personalized 3D-printed implant in sheep, according to a study published online December 10, 2014, in Science Translational Medicine. The therapy, successfully tested in sheep, could provide the first effective and long-lasting repair of damaged menisci.
“At present, there’s little that orthopedists can do to regenerate a torn knee meniscus,” said lead study author Jeremy Mao, DDS, PhD, the Edwin S. Robinson Professor of Dentistry in Orthopedic Surgery at Columbia University Medical Center in New York. “In contrast, we’re jumpstarting the process within the body, using factors that promote endogenous stem cells for tissue regeneration.”
This process was tested on 11 sheep. Knees of sheep were chosen because of their close resemblance to the human knee. In this study, the sheep were randomized to have part of their knee meniscus replaced with a protein-infused 3D scaffold or a 3D scaffold without protein.
Dr. Mao’s approach starts with MRI scans of the intact meniscus in the undamaged knee, which is then converted into a 3D image. Data from the image are then used to drive a 3D printer, which produces a scaffold in the exact shape of the meniscus, down to a resolution of 10 microns. The scaffold is infused with connective growth factor (CTGF) and transforming growth factor β3 (TGFβ3). Dr. Mao’s team found that sequential delivery of these two proteins attracts existing stem cells from the body and induces them to form meniscal tissue.
After three months, treated animals were walking normally. In a postmortem analysis, the researchers found that the regenerated meniscus in the treatment group had structural and mechanical properties very similar to those of natural meniscus. In sheep, the meniscus regenerates at a rate of about 4 to 6 weeks. Eventually, the scaffold dissolves and is eliminated by the body.
Suggested Reading
Lee CH, Rodeo SA, Fortier LA, et al. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci Transl Med. 2014 Dec 10;6(266):266ra171.
Suggested Reading
Lee CH, Rodeo SA, Fortier LA, et al. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci Transl Med. 2014 Dec 10;6(266):266ra171.
An Additional Option for Pain Control Following Knee Replacement Surgery
DALLAS—Injecting long-acting liposomal bupivacaine into the tissue surrounding the knee during knee replacement surgery may provide a faster receovery and higher patient satisfaction, according to a report presented at the recent American Association for Hip and Knee Surgeons meeting.
“The pain scores for this injection technique averaged about 3/10, which is similar to the pain scores seen with our traditional method,” said Jason Davis, MD, a Henry Ford West Bloomfield Hospital joint replacement surgeon and senior study author. “Patients had pain relief for up to 2 days after surgery and better knee function compared with the traditional method.”
In the study, 216 patients were evaluated for pain control the first 2 days after surgery from October 2012 to September 2013. Half of the patients received the traditional pain control method with continuous femoral nerve blockade, in which common numbing medicine is injected into the groin area, blunting the main nerve down the front of the knee—a method that calls for a pain pump to extend pain control for two days but causes some leg weakness.
“Pain control came at the price of weakness and made patients somewhat tentative when walking during their hospital stay,” stated Dr. Davis.
The other half of patients received the liposomal bupivacaine injection at the site of the surgery. With this method, Dr. Davis says many patients were able to walk comfortably within hours after surgery.
Dr. Davis said that the injection around the knee itself “optimizes pain control early on” without the side effects of the traditional technique. “Function-wise, it was a lot easier for patients to move around more confidently,” Dr. Davis stated.
“In the past decade, we’ve made major advancements in pain control for knee replacement surgery. This option is a promising, viable one for our patients,” Dr. Davis said.
DALLAS—Injecting long-acting liposomal bupivacaine into the tissue surrounding the knee during knee replacement surgery may provide a faster receovery and higher patient satisfaction, according to a report presented at the recent American Association for Hip and Knee Surgeons meeting.
“The pain scores for this injection technique averaged about 3/10, which is similar to the pain scores seen with our traditional method,” said Jason Davis, MD, a Henry Ford West Bloomfield Hospital joint replacement surgeon and senior study author. “Patients had pain relief for up to 2 days after surgery and better knee function compared with the traditional method.”
In the study, 216 patients were evaluated for pain control the first 2 days after surgery from October 2012 to September 2013. Half of the patients received the traditional pain control method with continuous femoral nerve blockade, in which common numbing medicine is injected into the groin area, blunting the main nerve down the front of the knee—a method that calls for a pain pump to extend pain control for two days but causes some leg weakness.
“Pain control came at the price of weakness and made patients somewhat tentative when walking during their hospital stay,” stated Dr. Davis.
The other half of patients received the liposomal bupivacaine injection at the site of the surgery. With this method, Dr. Davis says many patients were able to walk comfortably within hours after surgery.
Dr. Davis said that the injection around the knee itself “optimizes pain control early on” without the side effects of the traditional technique. “Function-wise, it was a lot easier for patients to move around more confidently,” Dr. Davis stated.
“In the past decade, we’ve made major advancements in pain control for knee replacement surgery. This option is a promising, viable one for our patients,” Dr. Davis said.
DALLAS—Injecting long-acting liposomal bupivacaine into the tissue surrounding the knee during knee replacement surgery may provide a faster receovery and higher patient satisfaction, according to a report presented at the recent American Association for Hip and Knee Surgeons meeting.
“The pain scores for this injection technique averaged about 3/10, which is similar to the pain scores seen with our traditional method,” said Jason Davis, MD, a Henry Ford West Bloomfield Hospital joint replacement surgeon and senior study author. “Patients had pain relief for up to 2 days after surgery and better knee function compared with the traditional method.”
In the study, 216 patients were evaluated for pain control the first 2 days after surgery from October 2012 to September 2013. Half of the patients received the traditional pain control method with continuous femoral nerve blockade, in which common numbing medicine is injected into the groin area, blunting the main nerve down the front of the knee—a method that calls for a pain pump to extend pain control for two days but causes some leg weakness.
“Pain control came at the price of weakness and made patients somewhat tentative when walking during their hospital stay,” stated Dr. Davis.
The other half of patients received the liposomal bupivacaine injection at the site of the surgery. With this method, Dr. Davis says many patients were able to walk comfortably within hours after surgery.
Dr. Davis said that the injection around the knee itself “optimizes pain control early on” without the side effects of the traditional technique. “Function-wise, it was a lot easier for patients to move around more confidently,” Dr. Davis stated.
“In the past decade, we’ve made major advancements in pain control for knee replacement surgery. This option is a promising, viable one for our patients,” Dr. Davis said.