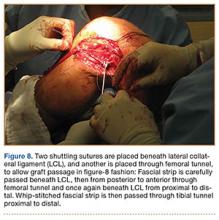
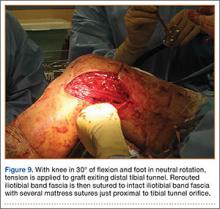
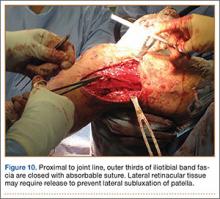
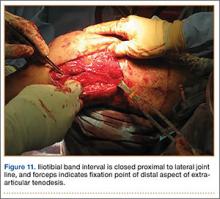
User login
Failure of Artelon Interposition Arthroplasty After Partial Trapeziectomy: A Case Report With Histologic and Immunohistochemical Analysis
Osteoarthritis (OA) of the first carpometacarpal (CMC) joint is a common disabling condition that mostly affects women over 45 years of age.1 Surgical intervention is usually indicated in advanced stage OA of the first CMC joint that has failed conservative treatment. Several surgical techniques have been described, including partial or total trapeziectomy, interposition arthroplasty with or without ligament reconstruction,2,3 metacarpal osteotomy,4 hematoma and distraction arthroplasty,5 total joint arthroplasty, arthrodesis, and suspensionplasty.6 However, no single surgical procedure has proved to be superior.7
The Artelon implant (Artelon, Nashville, Tennessee) is a T-shaped spacer composed of a biocompatible and biodegradable polycaprolactone-based polyurethane urea polymer. The developers of the implant first presented its use in CMC OA in 2005.8 The device, an endoprosthetic replacement for the CMC joint, was designed to work through 2 modes of action: stabilization of the CMC joint by augmentation of the joint capsule and by formation of a new articular surface at the trapeziometacarpal interface. The interposed biomaterial has been described as preventing bony impingement and allowing time for replacement with a newly formed articular surface as it undergoes slow and controlled degradation.8
We present a patient with recurrent CMC pain and disability 4 years after arthroscopic hemitrapeziectomy and Artelon interposition and discuss the associated histologic findings. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
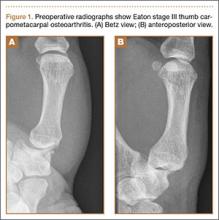
A 53-year-old man presented with painful disability of right thumb of several months’ duration. Clinical and radiographic evaluation supported the diagnosis of right thumb CMC joint Eaton stage III arthritis (Figures 1A, 1B). Surgical intervention was indicated after a failed course of conservative treatment, including splinting, nonsteroidal anti-inflammatory medications, activity modification, and corticosteroid injection. Preoperatively, the patient reported a visual analog scale (VAS) score of 8 with activity and 5 at rest, and a Disabilities of the Arm, Shoulder, and Hand (DASH) score of 72.5.
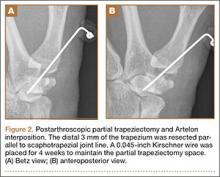
Arthroscopic débridement, hemitrapeziectomy, and interposition arthroplasty with the Artelon spacer were performed. Using standard thumb arthroscopy, 3 mm of the distal trapezium was excised and shaped parallel to scaphotrapezial joint. The wings of the standard-sized Artelon spacer were removed, and the central (articulating) portion was rolled into a tube and inserted through the 1R portal (directly radial to the abductor pollicis longus tendon) into the trapezial space. The Artelon spacer was unrolled within the joint to cover the remaining trapezium and was stabilized with the placement of a 0.045-inch Kirschner wire through the metacarpal, the spacer, and the remaining trapezium. The patient used a thumb spica splint for 4 weeks.
The postoperative radiographs showed a smooth and adequate hemitrapeziectomy with good alignment and implant position (Figures 2A, 2B). Four weeks after surgery, the Kirschner wire and cast were removed and physical therapy was initiated. The patient’s CMC pain gradually subsided. At the 3-month postoperative visit, the patient’s VAS score was 3 with activity and 1 at rest, with a DASH score of 28. His key pinch strength was 12 lb, compared with 20 lb on the contralateral side. At 6 months, the patient’s VAS score was 1 with activity and 0 at rest, with a DASH score of 12. His key pinch strength was 18 lb, compared with 22 lb on the contralateral side. At his 2-year postoperative visit, the patient was doing well with the exception of some mild residual pain when he opened tight jars. His VAS score was 1 with activity and 0 at rest, with a DASH score of 3. His key pinch strength was 20 lb, compared with 23 lb on the contralateral side. Radiographs showed good maintenance of the CMC space.
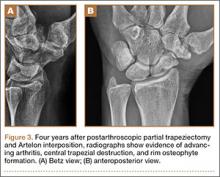
Four years postoperatively, the patient presented with worsening right CMC pain with decrease in pinch strength that interfered with his activities of daily living. His VAS score was 9 with activity and 6 at rest, with a DASH score of 70. On examination, pinch strength was 16 lb, compared with 22 lb on the contralateral side. Radiographs showed advancing arthritis with new osteophyte formation and irregular contour of distal trapezium (Figures 3A, 3B). The symptoms were refractory to conservative measures and continued to interfere with his activities of daily living. Revision surgical intervention was indicated and pursued in the form of an open CMC arthroplasty.
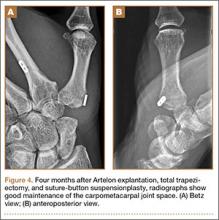
The intraoperative findings revealed degradation and disorganization of the Artelon implant within the central portion of the remaining distal trapezium. Rim osteophytes, especially along the ulnar aspect, were noted. Total trapeziectomy and débridement within the CMC space and suture-button suspensionplasty were performed.8 Slight degenerative changes of the distal scaphoid were also noted. The incision was irrigated, closed, and stabilized in a thumb spica splint (Figures 4A, 4B).
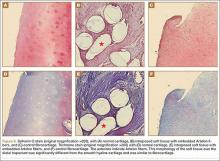
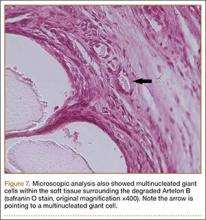
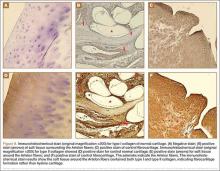
The harvested trapezium was immediately immersed in buffered formalin. The bone tissue was decalcified, dehydrated, embedded in paraffin, and sectioned in the coronal plane. The sections were stained with safranin O and trichrome, and light microscopic analysis was performed. Central erosion of distal trapezium without smooth resurfacing soft-tissue formation was noted grossly (Figure 5A) and microscopically (Figures 5B, 5C). The histologic morphology of the soft tissue over the distal trapezium was significantly different when compared with the smooth hyaline cartilage at the preserved trapezio-trapezoidal joint (Figures 6A-6F). Microscopic analysis also showed multinucleated giant cells within the soft tissue surrounding the degraded Artelon B (Figure 7).
Immunohistochemical analysis was performed to identify type I and type II collagen using the Histostain-Plus,3rd Gen IHC Detection Kit (Invitrogen Corporation, Camarillo, California) (Figures 8A-8F).9 The immunohistochemical stain was used to identify new hyaline cartilage formation that may have been induced by the Artelon as the resurfacing articulation. Hyaline cartilage contains mainly type II collagen, and collagen types VI, IX, X, XI, XII, and XIV all contribute to the mature matrix.10 Little type I collagen is found in hyaline cartilage. The results showed that the soft tissue over the distal trapezium with embedded Artelon fiber contained both type I and type II collagen. There was no visible hyaline cartilage formation induced by the Artelon. Both morphologic analysis and immunohistochemical staining revealed that the soft-tissue growth into the Artelon spacer on the distal trapezium consisted primarily of fibrocartilaginous tissue, which is composed mainly of type I collagen with some type II collagen.
Two weeks after total surgical excision of the Artelon implant, total trapeziectomy and suture-button suspensionplasty, the sutures were removed and physical therapy was initiated. Radiographs showed good alignment and position of thumb metacarpal with good maintenance of the implant and CMC space. Four months postoperatively, the patient reported that he was doing well without pain and without interference in his activities of daily living. On examination, the patient exhibited no pain with the CMC grind maneuver. Radial abduction of the right thumb was 85° and palmar abduction was 90° (compared with 100° and 90° of the left thumb), obtained by measuring the angle between thumb and index finger, respectively. Opposition was to the small finger metacarpophalangeal joint. Grip strength was 72 lb and pinch strength was 20 lb (compared with 70 lb and 24 lb, respectively, on the contralateral side).
Discussion
The use of Artelon as an endoprosthetic spacer to treat osteoarthritis in the CMC joint of the thumb appears to stabilize and resurface the joint while avoiding total trapeziectomy.8 Nilsson and colleagues8 presented a prospective study concluding that the Artelon CMC spacer provided better pinch strength when compared with a traditional abductor pollicis longus suspensionplasty procedure. This study also suggested incorporation of the device in the surface of the adjacent bone with no signs of foreign-body reaction. The synthetic material was shown to be safe and biocompatible in vitro and in animal studies.11-13
This case report describes the gross and histologic findings after continued pain led to explantation 4 years after arthroscopic partial trapeziectomy and insertion of the spacer. Intraoperative findings at this stage showed lack of incorporation of the Artelon material, central destruction of distal trapezium, and no evidence of smooth articular surface formation. Our histologic analysis showed only poorly organized fibrocartilage within the CMC space rather than a smooth articular surface. These histologic findings may correlate more with Jörheim and colleagues’14 matched cohort study, which showed that short-term outcomes after treatment with the Artelon implant were not clinically superior to those of tendon suspension-interposition arthroplasties. Multinucleated giant cells were also seen in our specimens. Choung and Tan15 presented a case report of foreign-body reaction to the Artelon spacer with histologic findings. The foreign body–type reactions associated with Artelon resulted in multinucleated giant cells in their specimens. Recently, several case reports have described similar foreign-body reactions.16 Nilsson and coauthors17 presented a randomized, controlled, multicenter study of 109 patients. They reported the Artelon CMC spacer did not result in superior results compared with tendon interposition arthroplasty. In a study by Gretzer and colleagues,18 the authors suggested that chronic inflammation may result from unstable Artelon fixation instead of the foreign-body reaction.
It is possible that the central erosion of the distal trapezium seen in our case may have resulted from chronic inflammation caused by foreign-body reaction and/or an unstably fixed spacer. The spacer was transfixed to the remaining trapezium in the CMC joint with a Kirschner wire followed by immobilization for 4 weeks. Poor soft-tissue integration of the Artelon spacer may have led to unintended motion and chronic inflammation, which may have also resulted in erosion between the Artelon spacer and the trapezium, leading to central destruction of the distal trapezium. Lastly, the byproducts formed by the degradation of the spacer may have resulted in erosion of the remaining trapezium.
Conclusion
The Artelon CMC spacer used in this patient provided comparable, but not superior, clinical results to other procedures. Histologically, the new articular surface in our patient was formed with rugged fibrocartilage instead of the expected smooth cartilaginous surface. The chronic inflammatory reaction may have resulted from foreign-body reaction, unstable implant fixation, or poor soft-tissue integration. This inflammatory reaction may have contributed to the patient’s recurrence of symptoms. These findings support recent clinical data that suggest the use of the Artelon spacer may not provide superior results to other surgical options for the treatment of CMC joint arthritis.
1. Dahaghin S, Bierma-Zeinstra SM, Ginai AZ, Pols HA, Hazes JM, Koes BW. Prevalence and pattern of radiographic hand osteoarthritis and association with pain and disability (the Rotterdam study). Ann Rheum Dis. 2005;64(5):682-687.
2. Eaton RG, Glickel SZ, Littler JW. Tendon interposition arthroplasty for degenerative arthritis of the trapeziometacarpal joint of the thumb. J Hand Surg. 1985;10(5):645-654.
3. Gibbons CE, Gosal HS, Choudri AH, Magnussen PA. Trapeziectomy for basal thumb joint osteoarthritis: 3- to 19-year follow-up. Int Orthop. 1999;23(4):216-218.
4. Gwynne-Jones DP, Penny ID, Sewell SA, Hughes TH. Basal thumb metacarpal osteotomy for trapeziometacarpal osteoarthritis. J Orthop Surg (Hong Kong). 2006;14(1):58-63.
5. Gray KV, Meals RA. Hematoma and distraction arthroplasty for thumb basal joint osteoarthritis: minimum 6.5-year follow-up evaluation. J Hand Surg Am. 2007;32(1):23-29.
6. Cox CA, Zlotolow DA, Yao J. Suture button suspensionplasty after arthroscopic hemitrapeziectomy for treatment of thumb carpometacarpal arthritis. Arthroscopy. 2010;26(10):1395-1403.
7. Vermeulen GM, Slijper H, Feitz R, Hovius SE, Moojen TM, Selles RW. Surgical management of primary thumb carpometacarpal osteoarthritis: a systematic review. J Hand Surg Am. 2011;36(1):157-169.
8. Nilsson A, Liljensten E, Bergström C, Sollerman C. Results from a degradable TMC joint Spacer (Artelon) compared with tendon arthroplasty. J Hand Surg Am. 2005;30(2):380-389.
9. Histostain®-Plus, 3rd Gen IHC Detection Kit [product information]. Invitrogen website. http://tools.invitrogen.com/content/sfs/manuals/859073_Rev1108.pdf. Revised November 2008. Accessed February 27, 2015.
10. Eyre D. Collagen of articular cartilage. Arthritis Res. 2002;4(1):30-35.
11. Gisselfält K, Edberg B, Flodin P. Synthesis and properties of degradable poly(urethane urea)s to be used for ligament reconstructions. Biomacromolecules. 2002;3(5):951-958.
12. Liljensten E, Gisselfält K, Edberg B, et al. Studies of polyurethane urea bands for ACL reconstruction. J Mater Sci Mater Med. 2002;13(4):351-359.
13. Gretzer C, Gisselfält K, Liljensten E, Rydén L, Thomsen P. Adhesion, apoptosis and cytokine release of human mononuclear cells cultured on degradable poly(urethane urea), polystyrene and titanium in vitro. Biomaterials. 2003;24(17):2843-2852.
14. Jörheim M, Isaxon I, Flondell M, Kalén P, Atroshi I. Short-term outcomes of trapeziometacarpal artelon implant compared with tendon suspension interposition arthroplasty for osteoarthritis: a matched cohort study. J Hand Surg Am. 2009;34(8):1381-1387.
15. Choung EW, Tan V. Foreign-body reaction to the Artelon CMC joint spacer: case report. J Hand Surg Am. 2008;33(9):1617-1620.
16. Robinson PM, Muir LT. Foreign body reaction associated with Artelon: report of three cases. J Hand Surg Am. 2011;36(1):116-120.
17. Nilsson A, Wiig M, Alnehill H, et al. The Artelon CMC spacer compared with tendon interposition arthroplasty. Acta Orthop. 2010;81(2):237-244.
18. Gretzer C, Emanuelsson L, Liljensten E, Thomsen P. The inflammatory cell influx and cytokines changes during transition from acute inflammation to fibrous repair around implanted materials. J Biomater Sci Polym Ed. 2006;17(6):669-687.
Osteoarthritis (OA) of the first carpometacarpal (CMC) joint is a common disabling condition that mostly affects women over 45 years of age.1 Surgical intervention is usually indicated in advanced stage OA of the first CMC joint that has failed conservative treatment. Several surgical techniques have been described, including partial or total trapeziectomy, interposition arthroplasty with or without ligament reconstruction,2,3 metacarpal osteotomy,4 hematoma and distraction arthroplasty,5 total joint arthroplasty, arthrodesis, and suspensionplasty.6 However, no single surgical procedure has proved to be superior.7
The Artelon implant (Artelon, Nashville, Tennessee) is a T-shaped spacer composed of a biocompatible and biodegradable polycaprolactone-based polyurethane urea polymer. The developers of the implant first presented its use in CMC OA in 2005.8 The device, an endoprosthetic replacement for the CMC joint, was designed to work through 2 modes of action: stabilization of the CMC joint by augmentation of the joint capsule and by formation of a new articular surface at the trapeziometacarpal interface. The interposed biomaterial has been described as preventing bony impingement and allowing time for replacement with a newly formed articular surface as it undergoes slow and controlled degradation.8
We present a patient with recurrent CMC pain and disability 4 years after arthroscopic hemitrapeziectomy and Artelon interposition and discuss the associated histologic findings. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 53-year-old man presented with painful disability of right thumb of several months’ duration. Clinical and radiographic evaluation supported the diagnosis of right thumb CMC joint Eaton stage III arthritis (Figures 1A, 1B). Surgical intervention was indicated after a failed course of conservative treatment, including splinting, nonsteroidal anti-inflammatory medications, activity modification, and corticosteroid injection. Preoperatively, the patient reported a visual analog scale (VAS) score of 8 with activity and 5 at rest, and a Disabilities of the Arm, Shoulder, and Hand (DASH) score of 72.5.
Arthroscopic débridement, hemitrapeziectomy, and interposition arthroplasty with the Artelon spacer were performed. Using standard thumb arthroscopy, 3 mm of the distal trapezium was excised and shaped parallel to scaphotrapezial joint. The wings of the standard-sized Artelon spacer were removed, and the central (articulating) portion was rolled into a tube and inserted through the 1R portal (directly radial to the abductor pollicis longus tendon) into the trapezial space. The Artelon spacer was unrolled within the joint to cover the remaining trapezium and was stabilized with the placement of a 0.045-inch Kirschner wire through the metacarpal, the spacer, and the remaining trapezium. The patient used a thumb spica splint for 4 weeks.
The postoperative radiographs showed a smooth and adequate hemitrapeziectomy with good alignment and implant position (Figures 2A, 2B). Four weeks after surgery, the Kirschner wire and cast were removed and physical therapy was initiated. The patient’s CMC pain gradually subsided. At the 3-month postoperative visit, the patient’s VAS score was 3 with activity and 1 at rest, with a DASH score of 28. His key pinch strength was 12 lb, compared with 20 lb on the contralateral side. At 6 months, the patient’s VAS score was 1 with activity and 0 at rest, with a DASH score of 12. His key pinch strength was 18 lb, compared with 22 lb on the contralateral side. At his 2-year postoperative visit, the patient was doing well with the exception of some mild residual pain when he opened tight jars. His VAS score was 1 with activity and 0 at rest, with a DASH score of 3. His key pinch strength was 20 lb, compared with 23 lb on the contralateral side. Radiographs showed good maintenance of the CMC space.
Four years postoperatively, the patient presented with worsening right CMC pain with decrease in pinch strength that interfered with his activities of daily living. His VAS score was 9 with activity and 6 at rest, with a DASH score of 70. On examination, pinch strength was 16 lb, compared with 22 lb on the contralateral side. Radiographs showed advancing arthritis with new osteophyte formation and irregular contour of distal trapezium (Figures 3A, 3B). The symptoms were refractory to conservative measures and continued to interfere with his activities of daily living. Revision surgical intervention was indicated and pursued in the form of an open CMC arthroplasty.
The intraoperative findings revealed degradation and disorganization of the Artelon implant within the central portion of the remaining distal trapezium. Rim osteophytes, especially along the ulnar aspect, were noted. Total trapeziectomy and débridement within the CMC space and suture-button suspensionplasty were performed.8 Slight degenerative changes of the distal scaphoid were also noted. The incision was irrigated, closed, and stabilized in a thumb spica splint (Figures 4A, 4B).
The harvested trapezium was immediately immersed in buffered formalin. The bone tissue was decalcified, dehydrated, embedded in paraffin, and sectioned in the coronal plane. The sections were stained with safranin O and trichrome, and light microscopic analysis was performed. Central erosion of distal trapezium without smooth resurfacing soft-tissue formation was noted grossly (Figure 5A) and microscopically (Figures 5B, 5C). The histologic morphology of the soft tissue over the distal trapezium was significantly different when compared with the smooth hyaline cartilage at the preserved trapezio-trapezoidal joint (Figures 6A-6F). Microscopic analysis also showed multinucleated giant cells within the soft tissue surrounding the degraded Artelon B (Figure 7).
Immunohistochemical analysis was performed to identify type I and type II collagen using the Histostain-Plus,3rd Gen IHC Detection Kit (Invitrogen Corporation, Camarillo, California) (Figures 8A-8F).9 The immunohistochemical stain was used to identify new hyaline cartilage formation that may have been induced by the Artelon as the resurfacing articulation. Hyaline cartilage contains mainly type II collagen, and collagen types VI, IX, X, XI, XII, and XIV all contribute to the mature matrix.10 Little type I collagen is found in hyaline cartilage. The results showed that the soft tissue over the distal trapezium with embedded Artelon fiber contained both type I and type II collagen. There was no visible hyaline cartilage formation induced by the Artelon. Both morphologic analysis and immunohistochemical staining revealed that the soft-tissue growth into the Artelon spacer on the distal trapezium consisted primarily of fibrocartilaginous tissue, which is composed mainly of type I collagen with some type II collagen.
Two weeks after total surgical excision of the Artelon implant, total trapeziectomy and suture-button suspensionplasty, the sutures were removed and physical therapy was initiated. Radiographs showed good alignment and position of thumb metacarpal with good maintenance of the implant and CMC space. Four months postoperatively, the patient reported that he was doing well without pain and without interference in his activities of daily living. On examination, the patient exhibited no pain with the CMC grind maneuver. Radial abduction of the right thumb was 85° and palmar abduction was 90° (compared with 100° and 90° of the left thumb), obtained by measuring the angle between thumb and index finger, respectively. Opposition was to the small finger metacarpophalangeal joint. Grip strength was 72 lb and pinch strength was 20 lb (compared with 70 lb and 24 lb, respectively, on the contralateral side).
Discussion
The use of Artelon as an endoprosthetic spacer to treat osteoarthritis in the CMC joint of the thumb appears to stabilize and resurface the joint while avoiding total trapeziectomy.8 Nilsson and colleagues8 presented a prospective study concluding that the Artelon CMC spacer provided better pinch strength when compared with a traditional abductor pollicis longus suspensionplasty procedure. This study also suggested incorporation of the device in the surface of the adjacent bone with no signs of foreign-body reaction. The synthetic material was shown to be safe and biocompatible in vitro and in animal studies.11-13
This case report describes the gross and histologic findings after continued pain led to explantation 4 years after arthroscopic partial trapeziectomy and insertion of the spacer. Intraoperative findings at this stage showed lack of incorporation of the Artelon material, central destruction of distal trapezium, and no evidence of smooth articular surface formation. Our histologic analysis showed only poorly organized fibrocartilage within the CMC space rather than a smooth articular surface. These histologic findings may correlate more with Jörheim and colleagues’14 matched cohort study, which showed that short-term outcomes after treatment with the Artelon implant were not clinically superior to those of tendon suspension-interposition arthroplasties. Multinucleated giant cells were also seen in our specimens. Choung and Tan15 presented a case report of foreign-body reaction to the Artelon spacer with histologic findings. The foreign body–type reactions associated with Artelon resulted in multinucleated giant cells in their specimens. Recently, several case reports have described similar foreign-body reactions.16 Nilsson and coauthors17 presented a randomized, controlled, multicenter study of 109 patients. They reported the Artelon CMC spacer did not result in superior results compared with tendon interposition arthroplasty. In a study by Gretzer and colleagues,18 the authors suggested that chronic inflammation may result from unstable Artelon fixation instead of the foreign-body reaction.
It is possible that the central erosion of the distal trapezium seen in our case may have resulted from chronic inflammation caused by foreign-body reaction and/or an unstably fixed spacer. The spacer was transfixed to the remaining trapezium in the CMC joint with a Kirschner wire followed by immobilization for 4 weeks. Poor soft-tissue integration of the Artelon spacer may have led to unintended motion and chronic inflammation, which may have also resulted in erosion between the Artelon spacer and the trapezium, leading to central destruction of the distal trapezium. Lastly, the byproducts formed by the degradation of the spacer may have resulted in erosion of the remaining trapezium.
Conclusion
The Artelon CMC spacer used in this patient provided comparable, but not superior, clinical results to other procedures. Histologically, the new articular surface in our patient was formed with rugged fibrocartilage instead of the expected smooth cartilaginous surface. The chronic inflammatory reaction may have resulted from foreign-body reaction, unstable implant fixation, or poor soft-tissue integration. This inflammatory reaction may have contributed to the patient’s recurrence of symptoms. These findings support recent clinical data that suggest the use of the Artelon spacer may not provide superior results to other surgical options for the treatment of CMC joint arthritis.
Osteoarthritis (OA) of the first carpometacarpal (CMC) joint is a common disabling condition that mostly affects women over 45 years of age.1 Surgical intervention is usually indicated in advanced stage OA of the first CMC joint that has failed conservative treatment. Several surgical techniques have been described, including partial or total trapeziectomy, interposition arthroplasty with or without ligament reconstruction,2,3 metacarpal osteotomy,4 hematoma and distraction arthroplasty,5 total joint arthroplasty, arthrodesis, and suspensionplasty.6 However, no single surgical procedure has proved to be superior.7
The Artelon implant (Artelon, Nashville, Tennessee) is a T-shaped spacer composed of a biocompatible and biodegradable polycaprolactone-based polyurethane urea polymer. The developers of the implant first presented its use in CMC OA in 2005.8 The device, an endoprosthetic replacement for the CMC joint, was designed to work through 2 modes of action: stabilization of the CMC joint by augmentation of the joint capsule and by formation of a new articular surface at the trapeziometacarpal interface. The interposed biomaterial has been described as preventing bony impingement and allowing time for replacement with a newly formed articular surface as it undergoes slow and controlled degradation.8
We present a patient with recurrent CMC pain and disability 4 years after arthroscopic hemitrapeziectomy and Artelon interposition and discuss the associated histologic findings. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
A 53-year-old man presented with painful disability of right thumb of several months’ duration. Clinical and radiographic evaluation supported the diagnosis of right thumb CMC joint Eaton stage III arthritis (Figures 1A, 1B). Surgical intervention was indicated after a failed course of conservative treatment, including splinting, nonsteroidal anti-inflammatory medications, activity modification, and corticosteroid injection. Preoperatively, the patient reported a visual analog scale (VAS) score of 8 with activity and 5 at rest, and a Disabilities of the Arm, Shoulder, and Hand (DASH) score of 72.5.
Arthroscopic débridement, hemitrapeziectomy, and interposition arthroplasty with the Artelon spacer were performed. Using standard thumb arthroscopy, 3 mm of the distal trapezium was excised and shaped parallel to scaphotrapezial joint. The wings of the standard-sized Artelon spacer were removed, and the central (articulating) portion was rolled into a tube and inserted through the 1R portal (directly radial to the abductor pollicis longus tendon) into the trapezial space. The Artelon spacer was unrolled within the joint to cover the remaining trapezium and was stabilized with the placement of a 0.045-inch Kirschner wire through the metacarpal, the spacer, and the remaining trapezium. The patient used a thumb spica splint for 4 weeks.
The postoperative radiographs showed a smooth and adequate hemitrapeziectomy with good alignment and implant position (Figures 2A, 2B). Four weeks after surgery, the Kirschner wire and cast were removed and physical therapy was initiated. The patient’s CMC pain gradually subsided. At the 3-month postoperative visit, the patient’s VAS score was 3 with activity and 1 at rest, with a DASH score of 28. His key pinch strength was 12 lb, compared with 20 lb on the contralateral side. At 6 months, the patient’s VAS score was 1 with activity and 0 at rest, with a DASH score of 12. His key pinch strength was 18 lb, compared with 22 lb on the contralateral side. At his 2-year postoperative visit, the patient was doing well with the exception of some mild residual pain when he opened tight jars. His VAS score was 1 with activity and 0 at rest, with a DASH score of 3. His key pinch strength was 20 lb, compared with 23 lb on the contralateral side. Radiographs showed good maintenance of the CMC space.
Four years postoperatively, the patient presented with worsening right CMC pain with decrease in pinch strength that interfered with his activities of daily living. His VAS score was 9 with activity and 6 at rest, with a DASH score of 70. On examination, pinch strength was 16 lb, compared with 22 lb on the contralateral side. Radiographs showed advancing arthritis with new osteophyte formation and irregular contour of distal trapezium (Figures 3A, 3B). The symptoms were refractory to conservative measures and continued to interfere with his activities of daily living. Revision surgical intervention was indicated and pursued in the form of an open CMC arthroplasty.
The intraoperative findings revealed degradation and disorganization of the Artelon implant within the central portion of the remaining distal trapezium. Rim osteophytes, especially along the ulnar aspect, were noted. Total trapeziectomy and débridement within the CMC space and suture-button suspensionplasty were performed.8 Slight degenerative changes of the distal scaphoid were also noted. The incision was irrigated, closed, and stabilized in a thumb spica splint (Figures 4A, 4B).
The harvested trapezium was immediately immersed in buffered formalin. The bone tissue was decalcified, dehydrated, embedded in paraffin, and sectioned in the coronal plane. The sections were stained with safranin O and trichrome, and light microscopic analysis was performed. Central erosion of distal trapezium without smooth resurfacing soft-tissue formation was noted grossly (Figure 5A) and microscopically (Figures 5B, 5C). The histologic morphology of the soft tissue over the distal trapezium was significantly different when compared with the smooth hyaline cartilage at the preserved trapezio-trapezoidal joint (Figures 6A-6F). Microscopic analysis also showed multinucleated giant cells within the soft tissue surrounding the degraded Artelon B (Figure 7).
Immunohistochemical analysis was performed to identify type I and type II collagen using the Histostain-Plus,3rd Gen IHC Detection Kit (Invitrogen Corporation, Camarillo, California) (Figures 8A-8F).9 The immunohistochemical stain was used to identify new hyaline cartilage formation that may have been induced by the Artelon as the resurfacing articulation. Hyaline cartilage contains mainly type II collagen, and collagen types VI, IX, X, XI, XII, and XIV all contribute to the mature matrix.10 Little type I collagen is found in hyaline cartilage. The results showed that the soft tissue over the distal trapezium with embedded Artelon fiber contained both type I and type II collagen. There was no visible hyaline cartilage formation induced by the Artelon. Both morphologic analysis and immunohistochemical staining revealed that the soft-tissue growth into the Artelon spacer on the distal trapezium consisted primarily of fibrocartilaginous tissue, which is composed mainly of type I collagen with some type II collagen.
Two weeks after total surgical excision of the Artelon implant, total trapeziectomy and suture-button suspensionplasty, the sutures were removed and physical therapy was initiated. Radiographs showed good alignment and position of thumb metacarpal with good maintenance of the implant and CMC space. Four months postoperatively, the patient reported that he was doing well without pain and without interference in his activities of daily living. On examination, the patient exhibited no pain with the CMC grind maneuver. Radial abduction of the right thumb was 85° and palmar abduction was 90° (compared with 100° and 90° of the left thumb), obtained by measuring the angle between thumb and index finger, respectively. Opposition was to the small finger metacarpophalangeal joint. Grip strength was 72 lb and pinch strength was 20 lb (compared with 70 lb and 24 lb, respectively, on the contralateral side).
Discussion
The use of Artelon as an endoprosthetic spacer to treat osteoarthritis in the CMC joint of the thumb appears to stabilize and resurface the joint while avoiding total trapeziectomy.8 Nilsson and colleagues8 presented a prospective study concluding that the Artelon CMC spacer provided better pinch strength when compared with a traditional abductor pollicis longus suspensionplasty procedure. This study also suggested incorporation of the device in the surface of the adjacent bone with no signs of foreign-body reaction. The synthetic material was shown to be safe and biocompatible in vitro and in animal studies.11-13
This case report describes the gross and histologic findings after continued pain led to explantation 4 years after arthroscopic partial trapeziectomy and insertion of the spacer. Intraoperative findings at this stage showed lack of incorporation of the Artelon material, central destruction of distal trapezium, and no evidence of smooth articular surface formation. Our histologic analysis showed only poorly organized fibrocartilage within the CMC space rather than a smooth articular surface. These histologic findings may correlate more with Jörheim and colleagues’14 matched cohort study, which showed that short-term outcomes after treatment with the Artelon implant were not clinically superior to those of tendon suspension-interposition arthroplasties. Multinucleated giant cells were also seen in our specimens. Choung and Tan15 presented a case report of foreign-body reaction to the Artelon spacer with histologic findings. The foreign body–type reactions associated with Artelon resulted in multinucleated giant cells in their specimens. Recently, several case reports have described similar foreign-body reactions.16 Nilsson and coauthors17 presented a randomized, controlled, multicenter study of 109 patients. They reported the Artelon CMC spacer did not result in superior results compared with tendon interposition arthroplasty. In a study by Gretzer and colleagues,18 the authors suggested that chronic inflammation may result from unstable Artelon fixation instead of the foreign-body reaction.
It is possible that the central erosion of the distal trapezium seen in our case may have resulted from chronic inflammation caused by foreign-body reaction and/or an unstably fixed spacer. The spacer was transfixed to the remaining trapezium in the CMC joint with a Kirschner wire followed by immobilization for 4 weeks. Poor soft-tissue integration of the Artelon spacer may have led to unintended motion and chronic inflammation, which may have also resulted in erosion between the Artelon spacer and the trapezium, leading to central destruction of the distal trapezium. Lastly, the byproducts formed by the degradation of the spacer may have resulted in erosion of the remaining trapezium.
Conclusion
The Artelon CMC spacer used in this patient provided comparable, but not superior, clinical results to other procedures. Histologically, the new articular surface in our patient was formed with rugged fibrocartilage instead of the expected smooth cartilaginous surface. The chronic inflammatory reaction may have resulted from foreign-body reaction, unstable implant fixation, or poor soft-tissue integration. This inflammatory reaction may have contributed to the patient’s recurrence of symptoms. These findings support recent clinical data that suggest the use of the Artelon spacer may not provide superior results to other surgical options for the treatment of CMC joint arthritis.
1. Dahaghin S, Bierma-Zeinstra SM, Ginai AZ, Pols HA, Hazes JM, Koes BW. Prevalence and pattern of radiographic hand osteoarthritis and association with pain and disability (the Rotterdam study). Ann Rheum Dis. 2005;64(5):682-687.
2. Eaton RG, Glickel SZ, Littler JW. Tendon interposition arthroplasty for degenerative arthritis of the trapeziometacarpal joint of the thumb. J Hand Surg. 1985;10(5):645-654.
3. Gibbons CE, Gosal HS, Choudri AH, Magnussen PA. Trapeziectomy for basal thumb joint osteoarthritis: 3- to 19-year follow-up. Int Orthop. 1999;23(4):216-218.
4. Gwynne-Jones DP, Penny ID, Sewell SA, Hughes TH. Basal thumb metacarpal osteotomy for trapeziometacarpal osteoarthritis. J Orthop Surg (Hong Kong). 2006;14(1):58-63.
5. Gray KV, Meals RA. Hematoma and distraction arthroplasty for thumb basal joint osteoarthritis: minimum 6.5-year follow-up evaluation. J Hand Surg Am. 2007;32(1):23-29.
6. Cox CA, Zlotolow DA, Yao J. Suture button suspensionplasty after arthroscopic hemitrapeziectomy for treatment of thumb carpometacarpal arthritis. Arthroscopy. 2010;26(10):1395-1403.
7. Vermeulen GM, Slijper H, Feitz R, Hovius SE, Moojen TM, Selles RW. Surgical management of primary thumb carpometacarpal osteoarthritis: a systematic review. J Hand Surg Am. 2011;36(1):157-169.
8. Nilsson A, Liljensten E, Bergström C, Sollerman C. Results from a degradable TMC joint Spacer (Artelon) compared with tendon arthroplasty. J Hand Surg Am. 2005;30(2):380-389.
9. Histostain®-Plus, 3rd Gen IHC Detection Kit [product information]. Invitrogen website. http://tools.invitrogen.com/content/sfs/manuals/859073_Rev1108.pdf. Revised November 2008. Accessed February 27, 2015.
10. Eyre D. Collagen of articular cartilage. Arthritis Res. 2002;4(1):30-35.
11. Gisselfält K, Edberg B, Flodin P. Synthesis and properties of degradable poly(urethane urea)s to be used for ligament reconstructions. Biomacromolecules. 2002;3(5):951-958.
12. Liljensten E, Gisselfält K, Edberg B, et al. Studies of polyurethane urea bands for ACL reconstruction. J Mater Sci Mater Med. 2002;13(4):351-359.
13. Gretzer C, Gisselfält K, Liljensten E, Rydén L, Thomsen P. Adhesion, apoptosis and cytokine release of human mononuclear cells cultured on degradable poly(urethane urea), polystyrene and titanium in vitro. Biomaterials. 2003;24(17):2843-2852.
14. Jörheim M, Isaxon I, Flondell M, Kalén P, Atroshi I. Short-term outcomes of trapeziometacarpal artelon implant compared with tendon suspension interposition arthroplasty for osteoarthritis: a matched cohort study. J Hand Surg Am. 2009;34(8):1381-1387.
15. Choung EW, Tan V. Foreign-body reaction to the Artelon CMC joint spacer: case report. J Hand Surg Am. 2008;33(9):1617-1620.
16. Robinson PM, Muir LT. Foreign body reaction associated with Artelon: report of three cases. J Hand Surg Am. 2011;36(1):116-120.
17. Nilsson A, Wiig M, Alnehill H, et al. The Artelon CMC spacer compared with tendon interposition arthroplasty. Acta Orthop. 2010;81(2):237-244.
18. Gretzer C, Emanuelsson L, Liljensten E, Thomsen P. The inflammatory cell influx and cytokines changes during transition from acute inflammation to fibrous repair around implanted materials. J Biomater Sci Polym Ed. 2006;17(6):669-687.
1. Dahaghin S, Bierma-Zeinstra SM, Ginai AZ, Pols HA, Hazes JM, Koes BW. Prevalence and pattern of radiographic hand osteoarthritis and association with pain and disability (the Rotterdam study). Ann Rheum Dis. 2005;64(5):682-687.
2. Eaton RG, Glickel SZ, Littler JW. Tendon interposition arthroplasty for degenerative arthritis of the trapeziometacarpal joint of the thumb. J Hand Surg. 1985;10(5):645-654.
3. Gibbons CE, Gosal HS, Choudri AH, Magnussen PA. Trapeziectomy for basal thumb joint osteoarthritis: 3- to 19-year follow-up. Int Orthop. 1999;23(4):216-218.
4. Gwynne-Jones DP, Penny ID, Sewell SA, Hughes TH. Basal thumb metacarpal osteotomy for trapeziometacarpal osteoarthritis. J Orthop Surg (Hong Kong). 2006;14(1):58-63.
5. Gray KV, Meals RA. Hematoma and distraction arthroplasty for thumb basal joint osteoarthritis: minimum 6.5-year follow-up evaluation. J Hand Surg Am. 2007;32(1):23-29.
6. Cox CA, Zlotolow DA, Yao J. Suture button suspensionplasty after arthroscopic hemitrapeziectomy for treatment of thumb carpometacarpal arthritis. Arthroscopy. 2010;26(10):1395-1403.
7. Vermeulen GM, Slijper H, Feitz R, Hovius SE, Moojen TM, Selles RW. Surgical management of primary thumb carpometacarpal osteoarthritis: a systematic review. J Hand Surg Am. 2011;36(1):157-169.
8. Nilsson A, Liljensten E, Bergström C, Sollerman C. Results from a degradable TMC joint Spacer (Artelon) compared with tendon arthroplasty. J Hand Surg Am. 2005;30(2):380-389.
9. Histostain®-Plus, 3rd Gen IHC Detection Kit [product information]. Invitrogen website. http://tools.invitrogen.com/content/sfs/manuals/859073_Rev1108.pdf. Revised November 2008. Accessed February 27, 2015.
10. Eyre D. Collagen of articular cartilage. Arthritis Res. 2002;4(1):30-35.
11. Gisselfält K, Edberg B, Flodin P. Synthesis and properties of degradable poly(urethane urea)s to be used for ligament reconstructions. Biomacromolecules. 2002;3(5):951-958.
12. Liljensten E, Gisselfält K, Edberg B, et al. Studies of polyurethane urea bands for ACL reconstruction. J Mater Sci Mater Med. 2002;13(4):351-359.
13. Gretzer C, Gisselfält K, Liljensten E, Rydén L, Thomsen P. Adhesion, apoptosis and cytokine release of human mononuclear cells cultured on degradable poly(urethane urea), polystyrene and titanium in vitro. Biomaterials. 2003;24(17):2843-2852.
14. Jörheim M, Isaxon I, Flondell M, Kalén P, Atroshi I. Short-term outcomes of trapeziometacarpal artelon implant compared with tendon suspension interposition arthroplasty for osteoarthritis: a matched cohort study. J Hand Surg Am. 2009;34(8):1381-1387.
15. Choung EW, Tan V. Foreign-body reaction to the Artelon CMC joint spacer: case report. J Hand Surg Am. 2008;33(9):1617-1620.
16. Robinson PM, Muir LT. Foreign body reaction associated with Artelon: report of three cases. J Hand Surg Am. 2011;36(1):116-120.
17. Nilsson A, Wiig M, Alnehill H, et al. The Artelon CMC spacer compared with tendon interposition arthroplasty. Acta Orthop. 2010;81(2):237-244.
18. Gretzer C, Emanuelsson L, Liljensten E, Thomsen P. The inflammatory cell influx and cytokines changes during transition from acute inflammation to fibrous repair around implanted materials. J Biomater Sci Polym Ed. 2006;17(6):669-687.
Massive Baker Cyst Resulting in Tibial Nerve Compression Neuropathy Secondary to Polyethylene Wear Disease
Symptomatic synovial cyst formation is a rare, late occurrence after total knee arthroplasty (TKA); these cysts are generally discovered by chance. If they enlarge, they can result in significant pain and disability. A few case reports have described the development of very large cysts that required revision knee surgery. In this patient, polyethylene wear disease after TKA resulted in a massive synovial cyst that extended into the posterior compartment of the leg, as well as a progressive peripheral neuropathy. Revision of a loose patella component and worn polyethylene liner with complete synovectomy, plus decompression of the cyst via needle aspiration, resulted in an excellent short-term outcome.
To the author’s knowledge, this is the first case report of peripheral neuropathy of the tibial nerve secondary to a massive Baker cyst after total knee replacement. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
The patient was a 65-year-old woman with a complex medical history and multiple left knee surgeries, including a high tibial osteotomy and subsequent cemented TKA performed in the mid-1990s. She presented to the orthopedic department at a university hospital with complaints of knee pain 13 years after TKA. Observation was recommended; however, she was lost to follow-up.
The patient presented to her primary care physician (PCP) 16 years after TKA with a very large, painful mass in the back of her left leg. An ultrasound showed a large Baker cyst, and the patient was sent to interventional radiology. A few months later, she had an ultrasound-guided aspiration into the left calf, which produced 300 mL of thick synovial fluid. A cell count was not performed, but bacterial cultures were negative. Immediately after the aspiration, the pain was relieved.
Approximately 3 months after the aspiration, she presented again to her PCP with re-accumulation of fluid in the back of her left leg and severe leg pain. She was referred to a different orthopedic surgeon who determined that the risk of surgery was too great given her complex medical history.
The woman’s PCP referred the woman to our office 6 months after the aspiration. On presentation, her pain was localized to the posterior left leg. She reported the pain level as a constant 9 out of 10 on the visual analog scale, despite ingesting high doses of narcotics, including oxycontin and morphine. Her physical examination was remarkable for an ill-defined large calf mass. The posterior compartment of her left leg was firm and severely tender, similar to the characteristic findings seen in acute compartment syndrome.
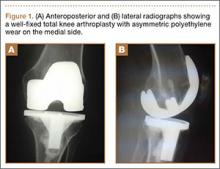
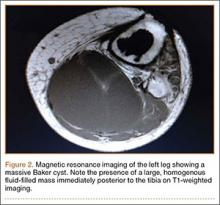
Radiographs showed evidence of asymmetric polyethylene wear on the medial side of the knee (Figures 1A, 1B). Serum labs were ordered to evaluate for infection. C-reactive protein was mildly elevated at 5.5 mg/L (normal range, 0-5 mg/L); however, the erythrocyte sedimentation rate was normal at 12 mm/h (normal range, 0-20 mm/h). Magnetic resonance imaging of the left lower extremity with intravenous contrast showed the presence of a very large Baker cyst contained within the posterior compartment of the knee and a smaller surrounding cyst adjacent to the popliteal neurovascular bundle (Figure 2).
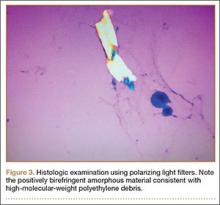
The Baker cyst was re-aspirated in the office. The automated synovial fluid cell count could not be performed because of high fluid viscosity. However, a manual review of the fluid specimen under light microscopy revealed proteinaceous, viscous tan-colored fluid containing no neutrophils and a few macrophages. Fluid cytology was also sent for review under polarizing light microscopy as described by Peterson and colleagues.1 Scattered fragments of polarizable foreign material were consistent with polyethylene debris (Figure 3).
The patient was counseled about the risks and benefits of surgery and was offered revision TKA with polyethylene liner exchange and synovectomy, only after complete cessation of smoking. She underwent serum nicotine monitoring to ensure tobacco cessation; however, she also reported the onset of a progressive sensory deficit over her left foot during this period. Although her medical history was remarkable for spinal stenosis, she noted a progressive decline in sensory function and new-onset paresthesia of her left foot.
An urgent consult to neurology was requested for nerve conduction studies. According to the electrodiagnostic study, the patient had a moderately severe left tibial neuropathy, likely at the popliteal fossa or distal to it. The nerve conduction study showed a chronic tibial nerve peripheral compressive mononeuropathy, and she was immediately scheduled for revision knee surgery with decompression of her Baker cyst to prevent further neurologic deficit.
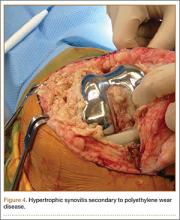
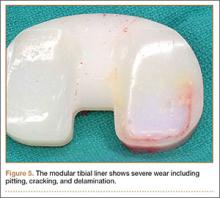
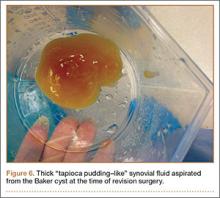
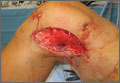
During surgery, the knee joint exhibited hypertrophic synovitis with a characteristic pale-yellowish discoloration secondary to significant polyethylene wear disease (Figure 4). The polyethylene liner was severely worn with pitting, cracking, and delamination (Figure 5). While the patellar component was grossly loose, the tibial and femoral components were stable. After a complete synovectomy, the loose patellar component and tibial polyethylene liner were replaced. Osteolytic areas within the tibia underwent curettage and allograft impaction grafting. Lastly, decompression of the ruptured Baker cyst was performed via a 16-gauge needle placed in the posterior compartment of the left leg. The calf was gently squeezed with a “milking” maneuver, which yielded approximately 200 mL of thick, mucoid yellowish-brown synovial fluid resembling tapioca pudding (Figure 6).
Postoperatively, all intraoperative cultures were negative, and the patient was followed closely at 1 week, 2 weeks, 6 weeks, and 3 months after the surgery. At her latest follow-up, the posterior leg compartment remained decompressed and her progressive sensory deficit had nearly resolved. Moreover, the left leg and posterior knee pain completely resolved.
Discussion
A leading cause of TKA failure is attributed to aseptic loosening from polyethylene wear disease.2 Implanted high-molecular-weight polyethylene (HMWPE) liners are known to undergo a variety of mechanical wear patterns within the knee. Observed patterns include pitting, scratching, burnishing, scratching, and delamination, which can all liberate numerous fine polyethylene particles.3 This wear debris induces macrophage phagocytosis that triggers an inflammatory reaction within the knee joint and can lead to synovitis, repeat effusions and, ultimately, to aseptic loosening.
Prior to 1996, polyethylene used in total knee replacement underwent a sterilization process in air. This oxygen-rich environment led to the development of free radical formation within the HMWPE. Ultimately, this had a detrimental effect on the polyethylene, leading to the formation of increased wear debris.4
Subsequently, orthopedic companies have changed their sterilization and manufacturing methods. Polyethylene components now undergo a variety of processes to eliminate or reduce oxidation, free-radical formation, and mechanical wear debris. Now, sterilization typically takes place in an inert atmospheric environment. Modern HMWPE implants often undergo higher irradiation to induce mechanical cross-linking, followed by either a re-annealing or remelting step. In other cases, manufacturers “dope” their polyethylene with vitamin E to quench free radicals within the material. While these steps have reduced the number of in vitro wear particles, the problem of wear debris, subsequent osteolysis, and aseptic loosening has not been eliminated.1-5
Polyethylene wear debris within the synovial fluid or tissue of failed TKAs can be identified with scanning electron microscopy or by light microscopy utilizing polarized light.1 In this particular case, wear debris was confirmed within the synovial tissue and in the fluid of the Baker cyst by microscopic analysis.
Formation of a popliteal or Baker cyst as a result of polyethylene wear disease is an infrequent but known complication of TKA. Reports have demonstrated variable success in cyst eradication when revision surgery is performed on knees with synovial cysts. Most of these reports indicate that cyst formation tends to occur as a late complication (7 or more years) after TKA.6-12
Treatment options may include skillful observation with close follow-up or revision surgery. Polyethylene exchange with synovectomy when feasible, as well as component revision with or without excision of the synovial cyst, are surgical options.
Niki and colleagues13 described a gigantic popliteal synovial cyst caused by wear particles after TKA. In this report, the surgeon performed a synovectomy and polyethylene liner exchange with retention of prosthetic components. At 12-month follow-up, the patient was reported to be doing well.
Mavrogenis and coauthors14 reported a wear debris–induced pseudotumor in the popliteal fossa and calf after TKA. In this case, in addition to the synovectomy, the surgeon removed all prosthetic components and used a semi-constrained implant to revise the knee. At 30-month follow-up, the patient reported having a painless knee.
While case reports have indicated that revision TKA for large, painful synovial cysts is a reasonable treatment option in carefully selected patients, there is a paucity of literature on this subject. Moreover, the present case appears to be the first literature report of a tibial nerve compressive neuropathy secondary to a synovial cyst after TKA.
Conclusion
In this report, polyethylene wear disease after TKA resulted in a massive synovial cyst extending into the posterior compartment of the leg. A progressive peripheral neuropathy confirmed by electromyography was also discovered. The patient underwent revision of a loose patellar component and worn polyethylene liner with complete synovectomy plus decompression of the cyst via needle aspiration. This resulted in an excellent short-term outcome with resolution of pain and significant improvement of the peripheral neuropathy 3 months after surgery.
1. Peterson C, Benjamin JB, Szivek JA, Anderson PL, Shriki J, Wong M. Polyethylene particle morphology in synovial fluid of failed knee arthroplasty. Clin Orthop. 1999;359:167-175.
2. Sadoghi P, Liebensteiner M, Agreiter M, Leithner A, Böhler N, Labek G. Revision surgery after total joint arthroplasty: a complication-based analysis using worldwide arthroplasty registers. J Arthroplasty. 2013;28(8):1329-1332.
3. Calonius O, Saikko V. Analysis of polyethylene particles produced in different wear conditions in vitro. Clin Orthop. 2002;399:219-230.
4. Edwards BT, Leach PB, Zura R, Corpe RS, Young TR. Presentation of gamma-irradiated-in-air polyethylene wear in the form of a synovial cyst. J Long Term Eff Med Implants. 2003;13(5):413-417.
5. Bosco J, Benjamin J, Wallace D. Quantitative and qualitative analysis of polyethylene wear particles in synovial fluid of patients with total arthroplasty. A preliminary report. Clin Orthop. 1994;309:11-19.
6. Moretti B, Patella V, Mouhsine E, Pesce V, Spinarelli A, Garofalo R. Multilobulated popliteal cyst after a failed total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2007;15(2):212-216.
7. Segura J, Palanca D, Bueno AL, Seral B, Castiella T, Seral F. Baker’s pseudocyst in the prosthetic knee affected with aggressive granulomatosis caused by polyethylene wear. Chir Organi Mov. 1996;81(4):421-426.
8. Ghanem G, Ghanem I, Dagher F. Popliteal cyst in a patient with total knee arthroplasty: a case report and review of the literature. J Med Liban. 2001;49(6):347-350.
9. Hsu WH, Hsu RW, Huang TJ, Lee KF. Dissecting popliteal cyst resulting from a fragmented, dislodged metal part of the patellar component after total knee arthroplasty. J Arthroplasty. 2002;17(6):792-797.
10. Chan YS, Wang CJ, Shin CH. Two-stage operation for treatment of a large dissecting popliteal cyst after failed total knee arthroplasty. J Arthroplasty. 2000;15(8):1068-1072.
11. Dirschl DR, Lachiewicz PF. Dissecting popliteal cyst as the presenting symptom of a malfunctioning total knee arthroplasty. Report of four cases. J Arthroplasty. 1992;7(1):37-41.
12. Akisue T, Kurosaka M, Matsui N, et al. Paratibial cyst associated with wear debris after total knee arthroplasty. J Arthroplasty. 2001;16(3):389-393.
13. Niki Y, Matsumoto H, Otani T, Yoshimine F, Inokuchi W, Morisue H. Gigantic popliteal synovial cyst caused by wear particles after total knee arthroplasty. J Arthroplasty. 2003;18(8):1071-1075.
14. Mavrogenis AF, Nomikos GN, Sakellariou VI, Karaliotas GI, Kontovazenitis P, Papagelopoulos PJ. Wear debris pseudotumor following total knee arthroplasty: a case report. J Med Case Rep. 2009;3:9304.
Symptomatic synovial cyst formation is a rare, late occurrence after total knee arthroplasty (TKA); these cysts are generally discovered by chance. If they enlarge, they can result in significant pain and disability. A few case reports have described the development of very large cysts that required revision knee surgery. In this patient, polyethylene wear disease after TKA resulted in a massive synovial cyst that extended into the posterior compartment of the leg, as well as a progressive peripheral neuropathy. Revision of a loose patella component and worn polyethylene liner with complete synovectomy, plus decompression of the cyst via needle aspiration, resulted in an excellent short-term outcome.
To the author’s knowledge, this is the first case report of peripheral neuropathy of the tibial nerve secondary to a massive Baker cyst after total knee replacement. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
The patient was a 65-year-old woman with a complex medical history and multiple left knee surgeries, including a high tibial osteotomy and subsequent cemented TKA performed in the mid-1990s. She presented to the orthopedic department at a university hospital with complaints of knee pain 13 years after TKA. Observation was recommended; however, she was lost to follow-up.
The patient presented to her primary care physician (PCP) 16 years after TKA with a very large, painful mass in the back of her left leg. An ultrasound showed a large Baker cyst, and the patient was sent to interventional radiology. A few months later, she had an ultrasound-guided aspiration into the left calf, which produced 300 mL of thick synovial fluid. A cell count was not performed, but bacterial cultures were negative. Immediately after the aspiration, the pain was relieved.
Approximately 3 months after the aspiration, she presented again to her PCP with re-accumulation of fluid in the back of her left leg and severe leg pain. She was referred to a different orthopedic surgeon who determined that the risk of surgery was too great given her complex medical history.
The woman’s PCP referred the woman to our office 6 months after the aspiration. On presentation, her pain was localized to the posterior left leg. She reported the pain level as a constant 9 out of 10 on the visual analog scale, despite ingesting high doses of narcotics, including oxycontin and morphine. Her physical examination was remarkable for an ill-defined large calf mass. The posterior compartment of her left leg was firm and severely tender, similar to the characteristic findings seen in acute compartment syndrome.
Radiographs showed evidence of asymmetric polyethylene wear on the medial side of the knee (Figures 1A, 1B). Serum labs were ordered to evaluate for infection. C-reactive protein was mildly elevated at 5.5 mg/L (normal range, 0-5 mg/L); however, the erythrocyte sedimentation rate was normal at 12 mm/h (normal range, 0-20 mm/h). Magnetic resonance imaging of the left lower extremity with intravenous contrast showed the presence of a very large Baker cyst contained within the posterior compartment of the knee and a smaller surrounding cyst adjacent to the popliteal neurovascular bundle (Figure 2).
The Baker cyst was re-aspirated in the office. The automated synovial fluid cell count could not be performed because of high fluid viscosity. However, a manual review of the fluid specimen under light microscopy revealed proteinaceous, viscous tan-colored fluid containing no neutrophils and a few macrophages. Fluid cytology was also sent for review under polarizing light microscopy as described by Peterson and colleagues.1 Scattered fragments of polarizable foreign material were consistent with polyethylene debris (Figure 3).
The patient was counseled about the risks and benefits of surgery and was offered revision TKA with polyethylene liner exchange and synovectomy, only after complete cessation of smoking. She underwent serum nicotine monitoring to ensure tobacco cessation; however, she also reported the onset of a progressive sensory deficit over her left foot during this period. Although her medical history was remarkable for spinal stenosis, she noted a progressive decline in sensory function and new-onset paresthesia of her left foot.
An urgent consult to neurology was requested for nerve conduction studies. According to the electrodiagnostic study, the patient had a moderately severe left tibial neuropathy, likely at the popliteal fossa or distal to it. The nerve conduction study showed a chronic tibial nerve peripheral compressive mononeuropathy, and she was immediately scheduled for revision knee surgery with decompression of her Baker cyst to prevent further neurologic deficit.
During surgery, the knee joint exhibited hypertrophic synovitis with a characteristic pale-yellowish discoloration secondary to significant polyethylene wear disease (Figure 4). The polyethylene liner was severely worn with pitting, cracking, and delamination (Figure 5). While the patellar component was grossly loose, the tibial and femoral components were stable. After a complete synovectomy, the loose patellar component and tibial polyethylene liner were replaced. Osteolytic areas within the tibia underwent curettage and allograft impaction grafting. Lastly, decompression of the ruptured Baker cyst was performed via a 16-gauge needle placed in the posterior compartment of the left leg. The calf was gently squeezed with a “milking” maneuver, which yielded approximately 200 mL of thick, mucoid yellowish-brown synovial fluid resembling tapioca pudding (Figure 6).
Postoperatively, all intraoperative cultures were negative, and the patient was followed closely at 1 week, 2 weeks, 6 weeks, and 3 months after the surgery. At her latest follow-up, the posterior leg compartment remained decompressed and her progressive sensory deficit had nearly resolved. Moreover, the left leg and posterior knee pain completely resolved.
Discussion
A leading cause of TKA failure is attributed to aseptic loosening from polyethylene wear disease.2 Implanted high-molecular-weight polyethylene (HMWPE) liners are known to undergo a variety of mechanical wear patterns within the knee. Observed patterns include pitting, scratching, burnishing, scratching, and delamination, which can all liberate numerous fine polyethylene particles.3 This wear debris induces macrophage phagocytosis that triggers an inflammatory reaction within the knee joint and can lead to synovitis, repeat effusions and, ultimately, to aseptic loosening.
Prior to 1996, polyethylene used in total knee replacement underwent a sterilization process in air. This oxygen-rich environment led to the development of free radical formation within the HMWPE. Ultimately, this had a detrimental effect on the polyethylene, leading to the formation of increased wear debris.4
Subsequently, orthopedic companies have changed their sterilization and manufacturing methods. Polyethylene components now undergo a variety of processes to eliminate or reduce oxidation, free-radical formation, and mechanical wear debris. Now, sterilization typically takes place in an inert atmospheric environment. Modern HMWPE implants often undergo higher irradiation to induce mechanical cross-linking, followed by either a re-annealing or remelting step. In other cases, manufacturers “dope” their polyethylene with vitamin E to quench free radicals within the material. While these steps have reduced the number of in vitro wear particles, the problem of wear debris, subsequent osteolysis, and aseptic loosening has not been eliminated.1-5
Polyethylene wear debris within the synovial fluid or tissue of failed TKAs can be identified with scanning electron microscopy or by light microscopy utilizing polarized light.1 In this particular case, wear debris was confirmed within the synovial tissue and in the fluid of the Baker cyst by microscopic analysis.
Formation of a popliteal or Baker cyst as a result of polyethylene wear disease is an infrequent but known complication of TKA. Reports have demonstrated variable success in cyst eradication when revision surgery is performed on knees with synovial cysts. Most of these reports indicate that cyst formation tends to occur as a late complication (7 or more years) after TKA.6-12
Treatment options may include skillful observation with close follow-up or revision surgery. Polyethylene exchange with synovectomy when feasible, as well as component revision with or without excision of the synovial cyst, are surgical options.
Niki and colleagues13 described a gigantic popliteal synovial cyst caused by wear particles after TKA. In this report, the surgeon performed a synovectomy and polyethylene liner exchange with retention of prosthetic components. At 12-month follow-up, the patient was reported to be doing well.
Mavrogenis and coauthors14 reported a wear debris–induced pseudotumor in the popliteal fossa and calf after TKA. In this case, in addition to the synovectomy, the surgeon removed all prosthetic components and used a semi-constrained implant to revise the knee. At 30-month follow-up, the patient reported having a painless knee.
While case reports have indicated that revision TKA for large, painful synovial cysts is a reasonable treatment option in carefully selected patients, there is a paucity of literature on this subject. Moreover, the present case appears to be the first literature report of a tibial nerve compressive neuropathy secondary to a synovial cyst after TKA.
Conclusion
In this report, polyethylene wear disease after TKA resulted in a massive synovial cyst extending into the posterior compartment of the leg. A progressive peripheral neuropathy confirmed by electromyography was also discovered. The patient underwent revision of a loose patellar component and worn polyethylene liner with complete synovectomy plus decompression of the cyst via needle aspiration. This resulted in an excellent short-term outcome with resolution of pain and significant improvement of the peripheral neuropathy 3 months after surgery.
Symptomatic synovial cyst formation is a rare, late occurrence after total knee arthroplasty (TKA); these cysts are generally discovered by chance. If they enlarge, they can result in significant pain and disability. A few case reports have described the development of very large cysts that required revision knee surgery. In this patient, polyethylene wear disease after TKA resulted in a massive synovial cyst that extended into the posterior compartment of the leg, as well as a progressive peripheral neuropathy. Revision of a loose patella component and worn polyethylene liner with complete synovectomy, plus decompression of the cyst via needle aspiration, resulted in an excellent short-term outcome.
To the author’s knowledge, this is the first case report of peripheral neuropathy of the tibial nerve secondary to a massive Baker cyst after total knee replacement. The patient provided written informed consent for print and electronic publication of this case report.
Case Report
The patient was a 65-year-old woman with a complex medical history and multiple left knee surgeries, including a high tibial osteotomy and subsequent cemented TKA performed in the mid-1990s. She presented to the orthopedic department at a university hospital with complaints of knee pain 13 years after TKA. Observation was recommended; however, she was lost to follow-up.
The patient presented to her primary care physician (PCP) 16 years after TKA with a very large, painful mass in the back of her left leg. An ultrasound showed a large Baker cyst, and the patient was sent to interventional radiology. A few months later, she had an ultrasound-guided aspiration into the left calf, which produced 300 mL of thick synovial fluid. A cell count was not performed, but bacterial cultures were negative. Immediately after the aspiration, the pain was relieved.
Approximately 3 months after the aspiration, she presented again to her PCP with re-accumulation of fluid in the back of her left leg and severe leg pain. She was referred to a different orthopedic surgeon who determined that the risk of surgery was too great given her complex medical history.
The woman’s PCP referred the woman to our office 6 months after the aspiration. On presentation, her pain was localized to the posterior left leg. She reported the pain level as a constant 9 out of 10 on the visual analog scale, despite ingesting high doses of narcotics, including oxycontin and morphine. Her physical examination was remarkable for an ill-defined large calf mass. The posterior compartment of her left leg was firm and severely tender, similar to the characteristic findings seen in acute compartment syndrome.
Radiographs showed evidence of asymmetric polyethylene wear on the medial side of the knee (Figures 1A, 1B). Serum labs were ordered to evaluate for infection. C-reactive protein was mildly elevated at 5.5 mg/L (normal range, 0-5 mg/L); however, the erythrocyte sedimentation rate was normal at 12 mm/h (normal range, 0-20 mm/h). Magnetic resonance imaging of the left lower extremity with intravenous contrast showed the presence of a very large Baker cyst contained within the posterior compartment of the knee and a smaller surrounding cyst adjacent to the popliteal neurovascular bundle (Figure 2).
The Baker cyst was re-aspirated in the office. The automated synovial fluid cell count could not be performed because of high fluid viscosity. However, a manual review of the fluid specimen under light microscopy revealed proteinaceous, viscous tan-colored fluid containing no neutrophils and a few macrophages. Fluid cytology was also sent for review under polarizing light microscopy as described by Peterson and colleagues.1 Scattered fragments of polarizable foreign material were consistent with polyethylene debris (Figure 3).
The patient was counseled about the risks and benefits of surgery and was offered revision TKA with polyethylene liner exchange and synovectomy, only after complete cessation of smoking. She underwent serum nicotine monitoring to ensure tobacco cessation; however, she also reported the onset of a progressive sensory deficit over her left foot during this period. Although her medical history was remarkable for spinal stenosis, she noted a progressive decline in sensory function and new-onset paresthesia of her left foot.
An urgent consult to neurology was requested for nerve conduction studies. According to the electrodiagnostic study, the patient had a moderately severe left tibial neuropathy, likely at the popliteal fossa or distal to it. The nerve conduction study showed a chronic tibial nerve peripheral compressive mononeuropathy, and she was immediately scheduled for revision knee surgery with decompression of her Baker cyst to prevent further neurologic deficit.
During surgery, the knee joint exhibited hypertrophic synovitis with a characteristic pale-yellowish discoloration secondary to significant polyethylene wear disease (Figure 4). The polyethylene liner was severely worn with pitting, cracking, and delamination (Figure 5). While the patellar component was grossly loose, the tibial and femoral components were stable. After a complete synovectomy, the loose patellar component and tibial polyethylene liner were replaced. Osteolytic areas within the tibia underwent curettage and allograft impaction grafting. Lastly, decompression of the ruptured Baker cyst was performed via a 16-gauge needle placed in the posterior compartment of the left leg. The calf was gently squeezed with a “milking” maneuver, which yielded approximately 200 mL of thick, mucoid yellowish-brown synovial fluid resembling tapioca pudding (Figure 6).
Postoperatively, all intraoperative cultures were negative, and the patient was followed closely at 1 week, 2 weeks, 6 weeks, and 3 months after the surgery. At her latest follow-up, the posterior leg compartment remained decompressed and her progressive sensory deficit had nearly resolved. Moreover, the left leg and posterior knee pain completely resolved.
Discussion
A leading cause of TKA failure is attributed to aseptic loosening from polyethylene wear disease.2 Implanted high-molecular-weight polyethylene (HMWPE) liners are known to undergo a variety of mechanical wear patterns within the knee. Observed patterns include pitting, scratching, burnishing, scratching, and delamination, which can all liberate numerous fine polyethylene particles.3 This wear debris induces macrophage phagocytosis that triggers an inflammatory reaction within the knee joint and can lead to synovitis, repeat effusions and, ultimately, to aseptic loosening.
Prior to 1996, polyethylene used in total knee replacement underwent a sterilization process in air. This oxygen-rich environment led to the development of free radical formation within the HMWPE. Ultimately, this had a detrimental effect on the polyethylene, leading to the formation of increased wear debris.4
Subsequently, orthopedic companies have changed their sterilization and manufacturing methods. Polyethylene components now undergo a variety of processes to eliminate or reduce oxidation, free-radical formation, and mechanical wear debris. Now, sterilization typically takes place in an inert atmospheric environment. Modern HMWPE implants often undergo higher irradiation to induce mechanical cross-linking, followed by either a re-annealing or remelting step. In other cases, manufacturers “dope” their polyethylene with vitamin E to quench free radicals within the material. While these steps have reduced the number of in vitro wear particles, the problem of wear debris, subsequent osteolysis, and aseptic loosening has not been eliminated.1-5
Polyethylene wear debris within the synovial fluid or tissue of failed TKAs can be identified with scanning electron microscopy or by light microscopy utilizing polarized light.1 In this particular case, wear debris was confirmed within the synovial tissue and in the fluid of the Baker cyst by microscopic analysis.
Formation of a popliteal or Baker cyst as a result of polyethylene wear disease is an infrequent but known complication of TKA. Reports have demonstrated variable success in cyst eradication when revision surgery is performed on knees with synovial cysts. Most of these reports indicate that cyst formation tends to occur as a late complication (7 or more years) after TKA.6-12
Treatment options may include skillful observation with close follow-up or revision surgery. Polyethylene exchange with synovectomy when feasible, as well as component revision with or without excision of the synovial cyst, are surgical options.
Niki and colleagues13 described a gigantic popliteal synovial cyst caused by wear particles after TKA. In this report, the surgeon performed a synovectomy and polyethylene liner exchange with retention of prosthetic components. At 12-month follow-up, the patient was reported to be doing well.
Mavrogenis and coauthors14 reported a wear debris–induced pseudotumor in the popliteal fossa and calf after TKA. In this case, in addition to the synovectomy, the surgeon removed all prosthetic components and used a semi-constrained implant to revise the knee. At 30-month follow-up, the patient reported having a painless knee.
While case reports have indicated that revision TKA for large, painful synovial cysts is a reasonable treatment option in carefully selected patients, there is a paucity of literature on this subject. Moreover, the present case appears to be the first literature report of a tibial nerve compressive neuropathy secondary to a synovial cyst after TKA.
Conclusion
In this report, polyethylene wear disease after TKA resulted in a massive synovial cyst extending into the posterior compartment of the leg. A progressive peripheral neuropathy confirmed by electromyography was also discovered. The patient underwent revision of a loose patellar component and worn polyethylene liner with complete synovectomy plus decompression of the cyst via needle aspiration. This resulted in an excellent short-term outcome with resolution of pain and significant improvement of the peripheral neuropathy 3 months after surgery.
1. Peterson C, Benjamin JB, Szivek JA, Anderson PL, Shriki J, Wong M. Polyethylene particle morphology in synovial fluid of failed knee arthroplasty. Clin Orthop. 1999;359:167-175.
2. Sadoghi P, Liebensteiner M, Agreiter M, Leithner A, Böhler N, Labek G. Revision surgery after total joint arthroplasty: a complication-based analysis using worldwide arthroplasty registers. J Arthroplasty. 2013;28(8):1329-1332.
3. Calonius O, Saikko V. Analysis of polyethylene particles produced in different wear conditions in vitro. Clin Orthop. 2002;399:219-230.
4. Edwards BT, Leach PB, Zura R, Corpe RS, Young TR. Presentation of gamma-irradiated-in-air polyethylene wear in the form of a synovial cyst. J Long Term Eff Med Implants. 2003;13(5):413-417.
5. Bosco J, Benjamin J, Wallace D. Quantitative and qualitative analysis of polyethylene wear particles in synovial fluid of patients with total arthroplasty. A preliminary report. Clin Orthop. 1994;309:11-19.
6. Moretti B, Patella V, Mouhsine E, Pesce V, Spinarelli A, Garofalo R. Multilobulated popliteal cyst after a failed total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2007;15(2):212-216.
7. Segura J, Palanca D, Bueno AL, Seral B, Castiella T, Seral F. Baker’s pseudocyst in the prosthetic knee affected with aggressive granulomatosis caused by polyethylene wear. Chir Organi Mov. 1996;81(4):421-426.
8. Ghanem G, Ghanem I, Dagher F. Popliteal cyst in a patient with total knee arthroplasty: a case report and review of the literature. J Med Liban. 2001;49(6):347-350.
9. Hsu WH, Hsu RW, Huang TJ, Lee KF. Dissecting popliteal cyst resulting from a fragmented, dislodged metal part of the patellar component after total knee arthroplasty. J Arthroplasty. 2002;17(6):792-797.
10. Chan YS, Wang CJ, Shin CH. Two-stage operation for treatment of a large dissecting popliteal cyst after failed total knee arthroplasty. J Arthroplasty. 2000;15(8):1068-1072.
11. Dirschl DR, Lachiewicz PF. Dissecting popliteal cyst as the presenting symptom of a malfunctioning total knee arthroplasty. Report of four cases. J Arthroplasty. 1992;7(1):37-41.
12. Akisue T, Kurosaka M, Matsui N, et al. Paratibial cyst associated with wear debris after total knee arthroplasty. J Arthroplasty. 2001;16(3):389-393.
13. Niki Y, Matsumoto H, Otani T, Yoshimine F, Inokuchi W, Morisue H. Gigantic popliteal synovial cyst caused by wear particles after total knee arthroplasty. J Arthroplasty. 2003;18(8):1071-1075.
14. Mavrogenis AF, Nomikos GN, Sakellariou VI, Karaliotas GI, Kontovazenitis P, Papagelopoulos PJ. Wear debris pseudotumor following total knee arthroplasty: a case report. J Med Case Rep. 2009;3:9304.
1. Peterson C, Benjamin JB, Szivek JA, Anderson PL, Shriki J, Wong M. Polyethylene particle morphology in synovial fluid of failed knee arthroplasty. Clin Orthop. 1999;359:167-175.
2. Sadoghi P, Liebensteiner M, Agreiter M, Leithner A, Böhler N, Labek G. Revision surgery after total joint arthroplasty: a complication-based analysis using worldwide arthroplasty registers. J Arthroplasty. 2013;28(8):1329-1332.
3. Calonius O, Saikko V. Analysis of polyethylene particles produced in different wear conditions in vitro. Clin Orthop. 2002;399:219-230.
4. Edwards BT, Leach PB, Zura R, Corpe RS, Young TR. Presentation of gamma-irradiated-in-air polyethylene wear in the form of a synovial cyst. J Long Term Eff Med Implants. 2003;13(5):413-417.
5. Bosco J, Benjamin J, Wallace D. Quantitative and qualitative analysis of polyethylene wear particles in synovial fluid of patients with total arthroplasty. A preliminary report. Clin Orthop. 1994;309:11-19.
6. Moretti B, Patella V, Mouhsine E, Pesce V, Spinarelli A, Garofalo R. Multilobulated popliteal cyst after a failed total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc. 2007;15(2):212-216.
7. Segura J, Palanca D, Bueno AL, Seral B, Castiella T, Seral F. Baker’s pseudocyst in the prosthetic knee affected with aggressive granulomatosis caused by polyethylene wear. Chir Organi Mov. 1996;81(4):421-426.
8. Ghanem G, Ghanem I, Dagher F. Popliteal cyst in a patient with total knee arthroplasty: a case report and review of the literature. J Med Liban. 2001;49(6):347-350.
9. Hsu WH, Hsu RW, Huang TJ, Lee KF. Dissecting popliteal cyst resulting from a fragmented, dislodged metal part of the patellar component after total knee arthroplasty. J Arthroplasty. 2002;17(6):792-797.
10. Chan YS, Wang CJ, Shin CH. Two-stage operation for treatment of a large dissecting popliteal cyst after failed total knee arthroplasty. J Arthroplasty. 2000;15(8):1068-1072.
11. Dirschl DR, Lachiewicz PF. Dissecting popliteal cyst as the presenting symptom of a malfunctioning total knee arthroplasty. Report of four cases. J Arthroplasty. 1992;7(1):37-41.
12. Akisue T, Kurosaka M, Matsui N, et al. Paratibial cyst associated with wear debris after total knee arthroplasty. J Arthroplasty. 2001;16(3):389-393.
13. Niki Y, Matsumoto H, Otani T, Yoshimine F, Inokuchi W, Morisue H. Gigantic popliteal synovial cyst caused by wear particles after total knee arthroplasty. J Arthroplasty. 2003;18(8):1071-1075.
14. Mavrogenis AF, Nomikos GN, Sakellariou VI, Karaliotas GI, Kontovazenitis P, Papagelopoulos PJ. Wear debris pseudotumor following total knee arthroplasty: a case report. J Med Case Rep. 2009;3:9304.
Lumbar Degenerative Disc Disease and Tibiotalar Joint Arthritis: A 710-Specimen Postmortem Study
Osteoarthritis is the most common joint disorder, resulting in significant morbidity and disability. The worldwide prevalence of osteoarthritis was estimated at more than 151 million people, according to data published in 2004.1 In the United States, almost 27 million adults age 25 years and older suffer from clinically apparent disease.2 The spine is one of the most commonly affected joints of arthritis, and idiopathic low back pain is the most frequent complaint in the adult population.3 In adults with low back pain, evidence of lumbar intervertebral disc degeneration is often found on radiography.4 In 1 study, evidence of disc degeneration was found in 90% of adults age 50 to 59 years.5
Degenerative spinal disease most commonly affects the lumbar spine due to its high degree of mobility and weight-loading.6,7 Clinical8,9 and experimental studies10 have suggested that the degenerative changes in the lumbar spine begin in the intervertebral discs. Degenerative disc disease (DDD) results from a continuum of dehydration, degradation, and remodeling of the intervertebral discs and neighboring vertebrae to accommodate the changes in physical loading.11-13 This results in disc-space narrowing, disc bulging and herniation, vertebral rim osteophyte formation, and endplate sclerosis.7,14 Symptomatic neural compression may occur, often manifested by localized lower back and extremity pain, as well as sensory loss and weakness of the lower extremities.15-17 Changes in posture and gait may result because of altered sensation, and the consequent abnormal force transmission may predispose joints to accelerated wear and arthrosis.15,18
Numerous studies have delineated the association between lumbar spinal disorders and lower extremity arthrosis. Of note, research has demonstrated that hip and/or knee pathology and gait alteration may promote low back pain and lumbar disc degeneration.19-21 Although spinal abnormalities, such as scoliosis, may predispose an individual to accelerated hip degeneration,20 no studies have investigated the relationship between lumbar DDD and ankle osteoarthritis.
Ankle arthritis differs from hip and knee arthritis demographically, occurring approximately 9 times less frequently.21 The ankle joint is subjected to more weight-bearing force per square centimeter and is more commonly injured than any other joint in the body.21 Trauma and/or abnormal ankle mechanics are the most common causes of degenerative ankle arthritis.22 Other potential causes include inflammatory arthropathies, neuropathic arthropathy, infection, and tumor. The purpose of this study was to determine if a relationship exists between ankle arthrosis and lumbar disc degeneration, and to delineate if one may promote the onset or progression of the other.
Materials and Methods
We randomly chose 710 cadaveric specimens from the Hamann-Todd Osteological Collection in Cleveland, Ohio. The Hamann-Todd Collection contains skeletal remains from more than 3000 individuals who died in Cleveland, Ohio between 1893 and 1938. The cohort for this study included 583 male and 127 female cadavers, ranging in age from 17 to 105 years at the time of death. Table 1 shows the breakdown of these specimens according to age group; of the 710 specimens, 306 were of African American ancestry, and 404 were Caucasian.
Lumbar DDD was graded at each lumbar spinal level by a single examiner using the Eubanks modification23 of the Kettler and Wilke classification of vertebral endplate osteophytosis24:
Grade 0: normal vertebral endplates;
Grade 1: mild arthrosis, with evidence of osteophytic reaction involving up to 50% of the vertebral endplates;
Grade 2: moderate arthrosis, with evidence of osteophytic reaction involving 50% to 100% of the vertebral endplates;
Grade 3: severe arthrosis, with evidence of osteophytic reaction involving 100% of the vertebral endplates. Osteophytes are hypertrophic and bridging the joint space (Figure 1);
Grade 4: complete ankylosis.
Tibiotalar joint osteoarthritis was evaluated by a single examiner using a modification of the Kellgren-Lawrence classification4 for knee osteoarthritis:
Grade 0: no discernable wear/osteophytes;
Grade 1: 1-mm osteophyte(s) and/or <25% surface wear;
Grade 2: 1- to 2-mm osteophyte(s) and/or 25% to 50% joint surface;
Grade 3: 2- to 3-mm osteophyte(s) and/or >50% joint surface (Figure 2);
Grade 4: multiple large osteophytes and/or definite bony end deformity.
Statistical analysis was performed on the compiled data using Stata software (StataCorp, College Station, Texas). Linear and logistic regression analyses correcting for confounding factors of age, sex, race, and height were performed using a standard P-value cutoff (P < .05) and 95% confidence interval to determine statistical significance.
Results
Patients were considered to have osteoarthritis of the tibiotalar joint if either of the extremities measured grade 1 or higher. Of the 710 specimens selected, 14 specimens did not have adequate bone available for bilateral tibiotalar joint measurement, either from extensive bone degradation or amputation. Of the remaining 696 specimens, 586 had some degree of tibiotalar osteoarthritis present (Table 2). Regression analysis showed a significant positive association between right- and left-ankle osteoarthritis (coefficient: 0.491, P < .01). Tibiotalar joint arthritis was classified as severe if either extremity had arthrosis of grade 3 or higher. Of the 586 specimens that had tibiotalar joint arthritis, only 16% (97 specimens) had severe tibiotalar joint arthritis.
Data regarding lumbar disc degeneration were available for 516 of the 710 specimens selected, 443 of which showed some disc degeneration. Disc degeneration was most prevalent and significant at the L4-L5 and L3-L4 intervertebral levels (Figures 3, 4). Of these 516 specimens, 30 had degeneration at 1 level, 47 specimens had degeneration at 2 levels, 29 specimens had degeneration at 3 levels, 52 had degeneration at 4 levels, and 285 specimens had degeneration at all 5 lumbar levels. The majority of specimens were found to have some degree of degeneration at all 5 lumbar spinal levels (Figure 5). Severe lumbar DDD was defined as grade 3 or higher osteoarthritis present in at least 1 of the 5 lumbar levels. Of the 516 specimens that showed some degree of disc degeneration, 152 were classified as severe. When stratified by number of spinal levels, only 30% of specimens were found to have evidence of severe arthrosis, the majority of which was located at only 1 lumbar segment (Figure 6).
Linear regression analysis of the data showed a statistically significant positive association between lumbar disc degeneration and tibiotalar osteoarthritis (coefficient: 0.844, P < .01), even when correcting for confounding factors, such as age, sex, and race (coefficient: 0.331, P < .01).
Additional analysis of the data demonstrated that tibiotalar joint arthritis remained significantly associated with lumbar DDD across each lumbar level: L1-L2 (coefficient: 0.269, P < .01), L2-L3 (coefficient: 0.283, P < .01), L3-L4 (coefficient: 0.299, P < .01), L4-L5 (coefficient: 0.240, P < .02), L5-S1 (coefficient: 0.167, P < .05).
The presence of 3 or more levels of lumbar DDD significantly increased the possibility of developing severe tibiotalar joint arthritis. Lumbar DDD that encompassed 3 levels showed the highest odds for development of severe tibiotalar joint arthritis with an odds ratio (OR) of 20.542 (Table 3).
When subjects were compared by decade, the mean grade of tibiotalar joint arthritis was significantly higher than lumbar DDD in specimens who died in their 20s and 30s. This difference was insignificant in the fourth decade, and thereafter the mean value of lumbar DDD surpassed that of tibiotalar joint arthritis (Figure 7).
In contrast, severe lumbar DDD was more prevalent than severe tibiotalar joint arthritis in individuals age 20 years or older (Figure 8). There were no specimens under age 20 years with severe lumbar DDD or severe tibiotalar joint arthritis.
Logistic regression showed that individuals with severe lumbar disc degeneration had significantly higher odds of developing severe ankle arthritis (OR: 1.93, P < .05). Similarly, individuals with severe tibiotalar joint arthritis were just as likely to develop severe lumbar DDD with an OR of 1.97 (P < .05).
Discussion
Multiple joint involvement in osteoarthritis is well established with a wide range of evidence linking lower extremity joint pathology and lumbar spinal disease. In 1983, Offierski and MacNab20 were the first to describe hip-spine syndrome. In the next year, a study by Sponseller and colleagues25 of pediatric patients after hip arthrodesis further substantiated the association between spine and extremity disease, and demonstrated a continued cause and effect relationship after surgery.
Lumbar spinal degeneration has also been correlated with knee osteoarthritis. Tsuji and colleagues26 reported that degenerative changes in spinal alignment result in increased thigh muscle tension and knee flexion. Furthermore, in their radiographic analysis of 682 individuals, Horvath and colleagues27 also showed that individuals with spinal degeneration had a higher prevalence of knee and hip osteoarthritis.
One might hypothesize from this evidence that lumbar spinal degeneration and ankle arthritis would also be interrelated, given their interconnected role in lower extremity force transmission. Surprisingly, the literature correlating lumbar degeneration and lower extremity osteoarthritis has overlooked this association and has focused solely on the hip and knee. To our knowledge, this study is the first to identify a statistically significant association between tibiotalar joint osteoarthritis and lumbar disc degeneration.
The literature supported analysis of our data. Miller and colleagues28 evaluated disc degeneration in 600 autopsy specimens using the Nachemson29 grading system. This system categorizes disc degeneration into 4 grades based on macroscopic appearance. Miller and colleagues28 reported evidence of degenerative changes as early as the second decade of life, primarily involving the L3–L4 and L4–L5 levels. Of note, the Nachemson29 classification system includes only evidence of marginal osteophytes in grade 4 disease, which was not identified by Miller and colleagues28 until the fourth decade. These results were similar to those in our study, in which the L3-L4 and L4-L5 intervertebral levels were most commonly affected. However, in our study, significant degenerative changes were found in the third decade of life.
In addition, the percentage of specimens with severe disc degeneration increased with each decade (Figure 8). A substantial amount of histologic evidence demonstrates the progression of disc degeneration with age. With increased age, there is a gradual decrease in the osmotic swelling of intervertebral discs30 and a 2-fold decrease in disc hydration between adolescence and the eighth decade.31 Furthermore, the nucleus pulposus undergoes progressive fibrosis,32,33 with a 5-fold decrease in the fixed-charge density of nucleus glycosaminoglycans,34 and a 2-fold increase in intervertebral disc creep while under compression after age 30 years.35
While analyzing our findings, we had difficulty in determining which pathologic condition debuts and, subsequently, affects the other. According to our results, the mean grade of tibiotalar joint arthritis was higher than that of DDD in specimens through the third and fourth decades of life (Figure 7). After the age of 50 years, the mean grade of DDD surpasses that of tibiotalar arthritis. This may be initially interpreted that development of tibiotalar joint arthritis precedes lumbar disc degeneration. Ankle osteoarthritis is relatively rare, and given that the vast majority of ankle osteoarthritis is secondary to trauma,22 we would expect to see a higher incidence of ankle osteoarthritis in a younger, more active cohort. In addition, given our finding that ankle arthritis is related to lumbar disc degeneration, one could speculate that tibiotalar arthritis at a young age predisposes an individual to developing lumbar degeneration later in life.
However, this conclusion is inherently flawed; closer examination of the data revealed that the mean grade of tibiotalar arthritis and DDD in the third and fourth decades is relatively low, between grade 0 and grade 1 (Figure 7). Therefore, it is difficult to arrive at a conclusion when comparing such small values. Second, we must remember that we are comparing an average value of disc degeneration across all lumbar levels. When a specimen has only 1 disc that is severely degenerated, this value is averaged across all 5 lumbar levels and, thus, the overall mean grade of arthrosis is significantly diminished.
In fact, data from previous studies concur with the second argument. Upper-level lumbar disc degeneration is relatively rare and the vast majority of patients with disc degeneration present with significant disease in only 1 or 2 discs.36,37 Analysis of the specimens in this study revealed bony evidence of disc degeneration present at all 5 lumbar levels in over half of the specimens examined (57%). However, the majority of specimens in this cohort exhibit only low-grade degeneration. When specimens were analyzed for severe arthrosis (grade 3 and higher), nearly half of the specimens were found to have severe disease involving only 1 intervertebral disc (Figure 6). Data from Miller and colleagues28 and the present study show that the upper lumbar levels were relatively spared; the L3-L4 and L4-L5 lumbar levels showed the highest prevalence and severity of degenerative change.
To address this issue, we evaluated the percentage of specimens per decade with severe arthrosis (grade 3 and higher) of at least 1 lumbar intervertebral disc and 1 tibiotalar joint. Severe lumbar disc degeneration was found to be more prevalent than severe ankle arthritis in individuals age 20 years or older (Figure 8). Therefore, we postulate that significant degenerative changes in the lumbar spine precede the development of severe ankle arthritis.
One can further speculate that sequelae from lumbar disc degeneration may lead to the development of tibiotalar arthritis, given our finding that severe lumbar degeneration predisposes an individual to the development of ankle arthritis. Because significant lumbar disc degeneration has long been known to result in both spinal nerve and cord compression, we hypothesize that this resultant neurocompression promotes altered gait and translation of atypical forces to the ankle and foot, thus predisposing to the onset and/or progression of osteoarthritis. In support of this hypothesis, Morag and colleagues15 demonstrated that neurologic compression produced an altered posture and gait because of lost motor function and afferent proprioceptive sensation. This form of neurologic compromise may exert atypical forces upon the foot and ankle, predisposing the joint to accelerated wear and primary arthrosis.
In addition, DDD involving 3 or more lumbar intervertebral levels was found to significantly increase the likelihood of the subject having severe tibiotalar joint arthritis. Provided that lumbar disc degeneration typically involves significant degeneration at 1 level, we assume that significant arthrosis at 3 or more levels correlates to an overall more severe DDD with a higher corresponding likelihood of neural compression. However, compression of peripheral lower extremity nerves has been shown to result in neuropathic arthropathy akin to the diabetic Charcot foot.38 This could be a possible mechanism of accelerated ankle arthritis, but this study did not examine soft-tissue disease nor take into account other medical comorbidities of each specimen, including genetic predispositions towards osteoarthritis.
It should be noted that the aforementioned causative relationship between lumbar disc degeneration and tibiotalar arthritis is speculative and cannot be demonstrated definitively by this investigation. We acknowledge limitations of this study and the need for further research of the possible causative mechanism(s) of accelerated ankle arthrosis secondary to lumbar spinal disease. Ideally, the questions posed by our report would be answered via a large prospective cohort study that utilized both serial imaging and autopsy analysis. Unfortunately, this form of study is logistically and financially difficult to perform.
This was a retrospective cadaveric study in which determination of arthrosis severity was based solely on bony evidence. Therefore, the role of soft-tissue disease in the pathogenesis of arthrosis of the lumbar spine and tibiotalar joint could not be assessed, nor could definitive associations to clinically symptomatic disease. We made the assumption that progression of bone degeneration in both the lumbar spine and tibiotalar joint corresponded equally to the associated soft-tissue changes. Given this assumption, we cannot definitively conclude that degeneration of the lumbar spine precedes that of the ankle, because the absence of magnetic resonance imaging or fresh autopsy specimens in our study misses the early degenerative changes in the discs that precede the bony alteration measured in our study. Furthermore, readers should note that since this study compared only bone morphology, no emphasis was placed on clinical manifestation of lumbar disc degeneration or tibiotalar joint arthritis. As mentioned earlier, radiologic evidence of disc degeneration was found in 90% of adults age 50 to 59 years, according to a study by Hult5; however, it is important to note that not all individuals studied were symptomatic clinically. Unfortunately, medical records were not available for the bony specimens, and clinical correlations could not be assessed during this investigation.
Furthermore, no special attention was given to other pathologic conditions observed during specimen measurement. The presence of diseases, such as osteoporosis, spondylolysis, or previous traumatic injury, may have had implications in the resultant joint degeneration. Finally, the evaluation of arthrosis was performed subjectively without measuring reliability. However, the present analysis includes a large sample, each joint type was reviewed by a single examiner, and used a classification system that was modeled on a validated grading system. Ideally, multiple individuals should have been used for each type of measurement, with subsequent analysis of intraobserver and interobserver reliability.
Conclusion
Based on our study of a large population of adult skeletal specimens, we ascertained that lumbar intervertebral disc degeneration and tibiotalar osteoarthritis are associated. The prevalence of severe lumbar disc degeneration was higher than that of tibiotalar joint arthritis in individuals age 20 years or older. This may suggest that gait changes from disc degeneration or neural compression in the lumbar spine may play a role in the development of ankle osteoarthritis. Additionally, subjects with severe disc degeneration were twice as likely to develop significant tibiotalar osteoarthritis. This must be considered in the differential when treating patients with degenerative changes of the lumbar spine and leg pain.
1. Mathers C, Fat DM, Boerma JT, for the World Health Organization. The Global Burden of Disease: 2004 Update. Geneva, Switzerland: World Health Organization, 2008.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35.
3. Kelsey JL, Githens PB, White AA, et al. An epidemiological study of lifting and twisting on the job and risk for acute prolapsed lumbar intervertebral disc. J Orthop Res. 1984;2(1):61-66.
4. Kellgren JH, Lawrence JS. Osteoarthrosis and disc degeneration in an urban population. Ann Rheum Dis. 1958;17(4):388-397.
5. Hult L. Cervical, dorsal and lumbar spinal syndromes; a field investigation of a non-selected material of 1200 workers in different occupations with special reference to disc degeneration and so-called muscular rheumatism. Acta Orthop Scand Suppl. 1954;17:65-73.
6. Hirsch C. The reaction of intervertebral discs to compression forces. J Bone Joint Surg Am. 1955;37(6):1188-1196.
7. Videman T, Nurminen M, Troup JD. Lumbar spinal pathology in cadaveric material in relation to history of back pain, occupation and physical loading. Spine. 1990;15(8):728-740.
8. Butler D, Trafimow JH, Andersson GB, McNeil TW, Huckman MS. Discs degenerate before facets. Spine. 1990;15(2):111-113.
9. Fujiwara A, Tamai K, Yamato M, et al. The relationship between facet joint osteoarthritis and disc degeneration of the lumbar spine: an MRI study. Eur Spine J. 1999;8(5):396-401.
10. Lipson SJ, Muir H. Experimental intervertebral disc degeneration: morphologic and proteoglycan changes over time. Arthritis Rheum. 1981;24(1):12-21.
11. Eisenstein S, Roberts S. The physiology of the disc and its clinical relevance. J Bone Joint Surg Br. 2003;85(5):633-636.
12. Hughes SP, Freemont AJ, Hukins DW, McGregor AH, Roberts S. The pathogenesis of degeneration of the intervertebral disc and emerging therapies in the management of back pain. J Bone Joint Surg Br. 2012;94(10):1298-1304.
13. Inoue N, Espinoza Orías AA. Biomechanics of intervertebral disk degeneration. Orthop Clin North Am. 2011;42(4):487-499.
14. Battié MC, Videman T. Lumbar disc degeneration: epidemiology and genetics. J Bone Joint Surg Am. 2006;88(suppl 2):3-9.
15. Morag E, Hurwitz DE, Andriacchi TP, Hickey M, Andersson GB. Abnormalities in muscle function during gait in relation to the level of lumbar disc herniation. Spine. 2000;25(7):829-833.
16. Oikawa Y, Ohtori S, Koshi T, et al. Lumbar disc degeneration induces persistent groin pain. Spine. 2012;37(2):114-118.
17. Porter RW. Spinal stenosis and neurogenic claudication. Spine. 1996;21(17):2046-2052.
18. Papadakis NC, Christakis DG, Tzagarakis GN, et al. Gait variability measurements in lumbar spinal stenosis patients: part A. Comparison with healthy subjects. Physiol Meas. 2009;30(11):1171-1186.
19. McGregor AH, Hukins DW. Lower limb involvement in spinal function and low back pain. J Back Musculoskelet Rehabil. 2009;22(4):219-222.
20. Offierski CM, MacNab I. Hip-spine syndrome. Spine. 1983;8(3):316-321.
21. Thomas RH, Daniels TR. Ankle arthritis. J Bone Joint Surg Am. 2003;85(5):923-936.
22. Valderrabano V, Horisberger M, Russell I, Dougall H, Hintermann B. Etiology of ankle osteoarthritis. Clin Orthop. 2009;467(7):1800-1806.
23. Eubanks JD, Lee MJ, Cassinelli E, Ahn NU. Does lumbar facet arthrosis precede disc degeneration? A postmortem study. Clin Orthop. 2007;464:184-189.
24. Friberg S, Hirsch C. Anatomical and clinical changes in lumbar disc degeneration. Acta Orthop Scand. 1949;19(2):222-242.
25. Sponseller PD, McBeath AA, Perpich M. Hip arthrodesis in young patients. A long-term follow-up study. J Bone Joint Surg Am. 1984;66(6):853-859.
26. Tsuji T, Matsuyama Y, Goto M, et al. Knee-spine syndrome: correlation between sacral inclination and patellofemoral joint pain. J Orthop Sci. 2002;7(5):519-523.
27. Horvath G, Koroknai G, Acs B, Than P, Illés T. Prevalence of low back pain and lumbar spine degenerative disorders. Questionnaire survey and clinical-radiological analysis of a representative Hungarian population. Int Orthop. 2010;34(8):1245-1249.
28. Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine. 1988;13(2):173-178.
29. Nachemson A. Lumbar intradiscal pressure: experimental studies on post-mortem material. Acta Orthop Scand Suppl. 1960;43:1-104.
30. Kraemer J. Pressure-dependent fluid shifts in the intervertebral disc. Orthop Clin North Am. 1977;8(1):211-216.
31. Urban JP, McMullin JF. Swelling pressure of the intervertebral disc: influence of proteoglycan and collagen contents. Biorheology. 1985;22(2):145-157.
32. Coventry MB, Ghromley RK, Kernohan JW. The intervertebral disc, its macroscopic anatomy and pathology: Part III. Pathologic changes in the intervertebral disc. J Bone Joint Surg Br. 1945;27:460-474.
33. Friberg S, Hirsch C. Anatomical and clinical changes in lumbar disc degeneration. Acta Orthop Scand. 1949;19(2):222-242.
34. Lyons G, Eisenstein SM, Sweet MB. Biochemical changes in intervertebral disc degeneration. Biochim Biophys Acta. 1981;673(4):443-453.
35. Koeller W, Muehlhaus S, Meier W, Hartmann F. Biomechanical properties of human intervertebral discs subjected to axial dynamic compression: influence of age and degeneration. J Biomech. 1986;19(10):807-816.
36. Bosacco SJ, Berman AT, Raisis LW, Zamarin RI. High lumbar herniations. Case reports. Orthopaedics. 1989;12(2):275-278.
37. Spangfort EV. The lumbar disc herniation. A computer-aided analysis of 2,504 operations. Acta Orthop Scand Suppl. 1972;142:1-95.
38. Gupta R. A short history of neuropathic arthropathy. Clin Orthop. 1993;296:43-49.
Osteoarthritis is the most common joint disorder, resulting in significant morbidity and disability. The worldwide prevalence of osteoarthritis was estimated at more than 151 million people, according to data published in 2004.1 In the United States, almost 27 million adults age 25 years and older suffer from clinically apparent disease.2 The spine is one of the most commonly affected joints of arthritis, and idiopathic low back pain is the most frequent complaint in the adult population.3 In adults with low back pain, evidence of lumbar intervertebral disc degeneration is often found on radiography.4 In 1 study, evidence of disc degeneration was found in 90% of adults age 50 to 59 years.5
Degenerative spinal disease most commonly affects the lumbar spine due to its high degree of mobility and weight-loading.6,7 Clinical8,9 and experimental studies10 have suggested that the degenerative changes in the lumbar spine begin in the intervertebral discs. Degenerative disc disease (DDD) results from a continuum of dehydration, degradation, and remodeling of the intervertebral discs and neighboring vertebrae to accommodate the changes in physical loading.11-13 This results in disc-space narrowing, disc bulging and herniation, vertebral rim osteophyte formation, and endplate sclerosis.7,14 Symptomatic neural compression may occur, often manifested by localized lower back and extremity pain, as well as sensory loss and weakness of the lower extremities.15-17 Changes in posture and gait may result because of altered sensation, and the consequent abnormal force transmission may predispose joints to accelerated wear and arthrosis.15,18
Numerous studies have delineated the association between lumbar spinal disorders and lower extremity arthrosis. Of note, research has demonstrated that hip and/or knee pathology and gait alteration may promote low back pain and lumbar disc degeneration.19-21 Although spinal abnormalities, such as scoliosis, may predispose an individual to accelerated hip degeneration,20 no studies have investigated the relationship between lumbar DDD and ankle osteoarthritis.
Ankle arthritis differs from hip and knee arthritis demographically, occurring approximately 9 times less frequently.21 The ankle joint is subjected to more weight-bearing force per square centimeter and is more commonly injured than any other joint in the body.21 Trauma and/or abnormal ankle mechanics are the most common causes of degenerative ankle arthritis.22 Other potential causes include inflammatory arthropathies, neuropathic arthropathy, infection, and tumor. The purpose of this study was to determine if a relationship exists between ankle arthrosis and lumbar disc degeneration, and to delineate if one may promote the onset or progression of the other.
Materials and Methods
We randomly chose 710 cadaveric specimens from the Hamann-Todd Osteological Collection in Cleveland, Ohio. The Hamann-Todd Collection contains skeletal remains from more than 3000 individuals who died in Cleveland, Ohio between 1893 and 1938. The cohort for this study included 583 male and 127 female cadavers, ranging in age from 17 to 105 years at the time of death. Table 1 shows the breakdown of these specimens according to age group; of the 710 specimens, 306 were of African American ancestry, and 404 were Caucasian.
Lumbar DDD was graded at each lumbar spinal level by a single examiner using the Eubanks modification23 of the Kettler and Wilke classification of vertebral endplate osteophytosis24:
Grade 0: normal vertebral endplates;
Grade 1: mild arthrosis, with evidence of osteophytic reaction involving up to 50% of the vertebral endplates;
Grade 2: moderate arthrosis, with evidence of osteophytic reaction involving 50% to 100% of the vertebral endplates;
Grade 3: severe arthrosis, with evidence of osteophytic reaction involving 100% of the vertebral endplates. Osteophytes are hypertrophic and bridging the joint space (Figure 1);
Grade 4: complete ankylosis.
Tibiotalar joint osteoarthritis was evaluated by a single examiner using a modification of the Kellgren-Lawrence classification4 for knee osteoarthritis:
Grade 0: no discernable wear/osteophytes;
Grade 1: 1-mm osteophyte(s) and/or <25% surface wear;
Grade 2: 1- to 2-mm osteophyte(s) and/or 25% to 50% joint surface;
Grade 3: 2- to 3-mm osteophyte(s) and/or >50% joint surface (Figure 2);
Grade 4: multiple large osteophytes and/or definite bony end deformity.
Statistical analysis was performed on the compiled data using Stata software (StataCorp, College Station, Texas). Linear and logistic regression analyses correcting for confounding factors of age, sex, race, and height were performed using a standard P-value cutoff (P < .05) and 95% confidence interval to determine statistical significance.
Results
Patients were considered to have osteoarthritis of the tibiotalar joint if either of the extremities measured grade 1 or higher. Of the 710 specimens selected, 14 specimens did not have adequate bone available for bilateral tibiotalar joint measurement, either from extensive bone degradation or amputation. Of the remaining 696 specimens, 586 had some degree of tibiotalar osteoarthritis present (Table 2). Regression analysis showed a significant positive association between right- and left-ankle osteoarthritis (coefficient: 0.491, P < .01). Tibiotalar joint arthritis was classified as severe if either extremity had arthrosis of grade 3 or higher. Of the 586 specimens that had tibiotalar joint arthritis, only 16% (97 specimens) had severe tibiotalar joint arthritis.
Data regarding lumbar disc degeneration were available for 516 of the 710 specimens selected, 443 of which showed some disc degeneration. Disc degeneration was most prevalent and significant at the L4-L5 and L3-L4 intervertebral levels (Figures 3, 4). Of these 516 specimens, 30 had degeneration at 1 level, 47 specimens had degeneration at 2 levels, 29 specimens had degeneration at 3 levels, 52 had degeneration at 4 levels, and 285 specimens had degeneration at all 5 lumbar levels. The majority of specimens were found to have some degree of degeneration at all 5 lumbar spinal levels (Figure 5). Severe lumbar DDD was defined as grade 3 or higher osteoarthritis present in at least 1 of the 5 lumbar levels. Of the 516 specimens that showed some degree of disc degeneration, 152 were classified as severe. When stratified by number of spinal levels, only 30% of specimens were found to have evidence of severe arthrosis, the majority of which was located at only 1 lumbar segment (Figure 6).
Linear regression analysis of the data showed a statistically significant positive association between lumbar disc degeneration and tibiotalar osteoarthritis (coefficient: 0.844, P < .01), even when correcting for confounding factors, such as age, sex, and race (coefficient: 0.331, P < .01).
Additional analysis of the data demonstrated that tibiotalar joint arthritis remained significantly associated with lumbar DDD across each lumbar level: L1-L2 (coefficient: 0.269, P < .01), L2-L3 (coefficient: 0.283, P < .01), L3-L4 (coefficient: 0.299, P < .01), L4-L5 (coefficient: 0.240, P < .02), L5-S1 (coefficient: 0.167, P < .05).
The presence of 3 or more levels of lumbar DDD significantly increased the possibility of developing severe tibiotalar joint arthritis. Lumbar DDD that encompassed 3 levels showed the highest odds for development of severe tibiotalar joint arthritis with an odds ratio (OR) of 20.542 (Table 3).
When subjects were compared by decade, the mean grade of tibiotalar joint arthritis was significantly higher than lumbar DDD in specimens who died in their 20s and 30s. This difference was insignificant in the fourth decade, and thereafter the mean value of lumbar DDD surpassed that of tibiotalar joint arthritis (Figure 7).
In contrast, severe lumbar DDD was more prevalent than severe tibiotalar joint arthritis in individuals age 20 years or older (Figure 8). There were no specimens under age 20 years with severe lumbar DDD or severe tibiotalar joint arthritis.
Logistic regression showed that individuals with severe lumbar disc degeneration had significantly higher odds of developing severe ankle arthritis (OR: 1.93, P < .05). Similarly, individuals with severe tibiotalar joint arthritis were just as likely to develop severe lumbar DDD with an OR of 1.97 (P < .05).
Discussion
Multiple joint involvement in osteoarthritis is well established with a wide range of evidence linking lower extremity joint pathology and lumbar spinal disease. In 1983, Offierski and MacNab20 were the first to describe hip-spine syndrome. In the next year, a study by Sponseller and colleagues25 of pediatric patients after hip arthrodesis further substantiated the association between spine and extremity disease, and demonstrated a continued cause and effect relationship after surgery.
Lumbar spinal degeneration has also been correlated with knee osteoarthritis. Tsuji and colleagues26 reported that degenerative changes in spinal alignment result in increased thigh muscle tension and knee flexion. Furthermore, in their radiographic analysis of 682 individuals, Horvath and colleagues27 also showed that individuals with spinal degeneration had a higher prevalence of knee and hip osteoarthritis.
One might hypothesize from this evidence that lumbar spinal degeneration and ankle arthritis would also be interrelated, given their interconnected role in lower extremity force transmission. Surprisingly, the literature correlating lumbar degeneration and lower extremity osteoarthritis has overlooked this association and has focused solely on the hip and knee. To our knowledge, this study is the first to identify a statistically significant association between tibiotalar joint osteoarthritis and lumbar disc degeneration.
The literature supported analysis of our data. Miller and colleagues28 evaluated disc degeneration in 600 autopsy specimens using the Nachemson29 grading system. This system categorizes disc degeneration into 4 grades based on macroscopic appearance. Miller and colleagues28 reported evidence of degenerative changes as early as the second decade of life, primarily involving the L3–L4 and L4–L5 levels. Of note, the Nachemson29 classification system includes only evidence of marginal osteophytes in grade 4 disease, which was not identified by Miller and colleagues28 until the fourth decade. These results were similar to those in our study, in which the L3-L4 and L4-L5 intervertebral levels were most commonly affected. However, in our study, significant degenerative changes were found in the third decade of life.
In addition, the percentage of specimens with severe disc degeneration increased with each decade (Figure 8). A substantial amount of histologic evidence demonstrates the progression of disc degeneration with age. With increased age, there is a gradual decrease in the osmotic swelling of intervertebral discs30 and a 2-fold decrease in disc hydration between adolescence and the eighth decade.31 Furthermore, the nucleus pulposus undergoes progressive fibrosis,32,33 with a 5-fold decrease in the fixed-charge density of nucleus glycosaminoglycans,34 and a 2-fold increase in intervertebral disc creep while under compression after age 30 years.35
While analyzing our findings, we had difficulty in determining which pathologic condition debuts and, subsequently, affects the other. According to our results, the mean grade of tibiotalar joint arthritis was higher than that of DDD in specimens through the third and fourth decades of life (Figure 7). After the age of 50 years, the mean grade of DDD surpasses that of tibiotalar arthritis. This may be initially interpreted that development of tibiotalar joint arthritis precedes lumbar disc degeneration. Ankle osteoarthritis is relatively rare, and given that the vast majority of ankle osteoarthritis is secondary to trauma,22 we would expect to see a higher incidence of ankle osteoarthritis in a younger, more active cohort. In addition, given our finding that ankle arthritis is related to lumbar disc degeneration, one could speculate that tibiotalar arthritis at a young age predisposes an individual to developing lumbar degeneration later in life.
However, this conclusion is inherently flawed; closer examination of the data revealed that the mean grade of tibiotalar arthritis and DDD in the third and fourth decades is relatively low, between grade 0 and grade 1 (Figure 7). Therefore, it is difficult to arrive at a conclusion when comparing such small values. Second, we must remember that we are comparing an average value of disc degeneration across all lumbar levels. When a specimen has only 1 disc that is severely degenerated, this value is averaged across all 5 lumbar levels and, thus, the overall mean grade of arthrosis is significantly diminished.
In fact, data from previous studies concur with the second argument. Upper-level lumbar disc degeneration is relatively rare and the vast majority of patients with disc degeneration present with significant disease in only 1 or 2 discs.36,37 Analysis of the specimens in this study revealed bony evidence of disc degeneration present at all 5 lumbar levels in over half of the specimens examined (57%). However, the majority of specimens in this cohort exhibit only low-grade degeneration. When specimens were analyzed for severe arthrosis (grade 3 and higher), nearly half of the specimens were found to have severe disease involving only 1 intervertebral disc (Figure 6). Data from Miller and colleagues28 and the present study show that the upper lumbar levels were relatively spared; the L3-L4 and L4-L5 lumbar levels showed the highest prevalence and severity of degenerative change.
To address this issue, we evaluated the percentage of specimens per decade with severe arthrosis (grade 3 and higher) of at least 1 lumbar intervertebral disc and 1 tibiotalar joint. Severe lumbar disc degeneration was found to be more prevalent than severe ankle arthritis in individuals age 20 years or older (Figure 8). Therefore, we postulate that significant degenerative changes in the lumbar spine precede the development of severe ankle arthritis.
One can further speculate that sequelae from lumbar disc degeneration may lead to the development of tibiotalar arthritis, given our finding that severe lumbar degeneration predisposes an individual to the development of ankle arthritis. Because significant lumbar disc degeneration has long been known to result in both spinal nerve and cord compression, we hypothesize that this resultant neurocompression promotes altered gait and translation of atypical forces to the ankle and foot, thus predisposing to the onset and/or progression of osteoarthritis. In support of this hypothesis, Morag and colleagues15 demonstrated that neurologic compression produced an altered posture and gait because of lost motor function and afferent proprioceptive sensation. This form of neurologic compromise may exert atypical forces upon the foot and ankle, predisposing the joint to accelerated wear and primary arthrosis.
In addition, DDD involving 3 or more lumbar intervertebral levels was found to significantly increase the likelihood of the subject having severe tibiotalar joint arthritis. Provided that lumbar disc degeneration typically involves significant degeneration at 1 level, we assume that significant arthrosis at 3 or more levels correlates to an overall more severe DDD with a higher corresponding likelihood of neural compression. However, compression of peripheral lower extremity nerves has been shown to result in neuropathic arthropathy akin to the diabetic Charcot foot.38 This could be a possible mechanism of accelerated ankle arthritis, but this study did not examine soft-tissue disease nor take into account other medical comorbidities of each specimen, including genetic predispositions towards osteoarthritis.
It should be noted that the aforementioned causative relationship between lumbar disc degeneration and tibiotalar arthritis is speculative and cannot be demonstrated definitively by this investigation. We acknowledge limitations of this study and the need for further research of the possible causative mechanism(s) of accelerated ankle arthrosis secondary to lumbar spinal disease. Ideally, the questions posed by our report would be answered via a large prospective cohort study that utilized both serial imaging and autopsy analysis. Unfortunately, this form of study is logistically and financially difficult to perform.
This was a retrospective cadaveric study in which determination of arthrosis severity was based solely on bony evidence. Therefore, the role of soft-tissue disease in the pathogenesis of arthrosis of the lumbar spine and tibiotalar joint could not be assessed, nor could definitive associations to clinically symptomatic disease. We made the assumption that progression of bone degeneration in both the lumbar spine and tibiotalar joint corresponded equally to the associated soft-tissue changes. Given this assumption, we cannot definitively conclude that degeneration of the lumbar spine precedes that of the ankle, because the absence of magnetic resonance imaging or fresh autopsy specimens in our study misses the early degenerative changes in the discs that precede the bony alteration measured in our study. Furthermore, readers should note that since this study compared only bone morphology, no emphasis was placed on clinical manifestation of lumbar disc degeneration or tibiotalar joint arthritis. As mentioned earlier, radiologic evidence of disc degeneration was found in 90% of adults age 50 to 59 years, according to a study by Hult5; however, it is important to note that not all individuals studied were symptomatic clinically. Unfortunately, medical records were not available for the bony specimens, and clinical correlations could not be assessed during this investigation.
Furthermore, no special attention was given to other pathologic conditions observed during specimen measurement. The presence of diseases, such as osteoporosis, spondylolysis, or previous traumatic injury, may have had implications in the resultant joint degeneration. Finally, the evaluation of arthrosis was performed subjectively without measuring reliability. However, the present analysis includes a large sample, each joint type was reviewed by a single examiner, and used a classification system that was modeled on a validated grading system. Ideally, multiple individuals should have been used for each type of measurement, with subsequent analysis of intraobserver and interobserver reliability.
Conclusion
Based on our study of a large population of adult skeletal specimens, we ascertained that lumbar intervertebral disc degeneration and tibiotalar osteoarthritis are associated. The prevalence of severe lumbar disc degeneration was higher than that of tibiotalar joint arthritis in individuals age 20 years or older. This may suggest that gait changes from disc degeneration or neural compression in the lumbar spine may play a role in the development of ankle osteoarthritis. Additionally, subjects with severe disc degeneration were twice as likely to develop significant tibiotalar osteoarthritis. This must be considered in the differential when treating patients with degenerative changes of the lumbar spine and leg pain.
Osteoarthritis is the most common joint disorder, resulting in significant morbidity and disability. The worldwide prevalence of osteoarthritis was estimated at more than 151 million people, according to data published in 2004.1 In the United States, almost 27 million adults age 25 years and older suffer from clinically apparent disease.2 The spine is one of the most commonly affected joints of arthritis, and idiopathic low back pain is the most frequent complaint in the adult population.3 In adults with low back pain, evidence of lumbar intervertebral disc degeneration is often found on radiography.4 In 1 study, evidence of disc degeneration was found in 90% of adults age 50 to 59 years.5
Degenerative spinal disease most commonly affects the lumbar spine due to its high degree of mobility and weight-loading.6,7 Clinical8,9 and experimental studies10 have suggested that the degenerative changes in the lumbar spine begin in the intervertebral discs. Degenerative disc disease (DDD) results from a continuum of dehydration, degradation, and remodeling of the intervertebral discs and neighboring vertebrae to accommodate the changes in physical loading.11-13 This results in disc-space narrowing, disc bulging and herniation, vertebral rim osteophyte formation, and endplate sclerosis.7,14 Symptomatic neural compression may occur, often manifested by localized lower back and extremity pain, as well as sensory loss and weakness of the lower extremities.15-17 Changes in posture and gait may result because of altered sensation, and the consequent abnormal force transmission may predispose joints to accelerated wear and arthrosis.15,18
Numerous studies have delineated the association between lumbar spinal disorders and lower extremity arthrosis. Of note, research has demonstrated that hip and/or knee pathology and gait alteration may promote low back pain and lumbar disc degeneration.19-21 Although spinal abnormalities, such as scoliosis, may predispose an individual to accelerated hip degeneration,20 no studies have investigated the relationship between lumbar DDD and ankle osteoarthritis.
Ankle arthritis differs from hip and knee arthritis demographically, occurring approximately 9 times less frequently.21 The ankle joint is subjected to more weight-bearing force per square centimeter and is more commonly injured than any other joint in the body.21 Trauma and/or abnormal ankle mechanics are the most common causes of degenerative ankle arthritis.22 Other potential causes include inflammatory arthropathies, neuropathic arthropathy, infection, and tumor. The purpose of this study was to determine if a relationship exists between ankle arthrosis and lumbar disc degeneration, and to delineate if one may promote the onset or progression of the other.
Materials and Methods
We randomly chose 710 cadaveric specimens from the Hamann-Todd Osteological Collection in Cleveland, Ohio. The Hamann-Todd Collection contains skeletal remains from more than 3000 individuals who died in Cleveland, Ohio between 1893 and 1938. The cohort for this study included 583 male and 127 female cadavers, ranging in age from 17 to 105 years at the time of death. Table 1 shows the breakdown of these specimens according to age group; of the 710 specimens, 306 were of African American ancestry, and 404 were Caucasian.
Lumbar DDD was graded at each lumbar spinal level by a single examiner using the Eubanks modification23 of the Kettler and Wilke classification of vertebral endplate osteophytosis24:
Grade 0: normal vertebral endplates;
Grade 1: mild arthrosis, with evidence of osteophytic reaction involving up to 50% of the vertebral endplates;
Grade 2: moderate arthrosis, with evidence of osteophytic reaction involving 50% to 100% of the vertebral endplates;
Grade 3: severe arthrosis, with evidence of osteophytic reaction involving 100% of the vertebral endplates. Osteophytes are hypertrophic and bridging the joint space (Figure 1);
Grade 4: complete ankylosis.
Tibiotalar joint osteoarthritis was evaluated by a single examiner using a modification of the Kellgren-Lawrence classification4 for knee osteoarthritis:
Grade 0: no discernable wear/osteophytes;
Grade 1: 1-mm osteophyte(s) and/or <25% surface wear;
Grade 2: 1- to 2-mm osteophyte(s) and/or 25% to 50% joint surface;
Grade 3: 2- to 3-mm osteophyte(s) and/or >50% joint surface (Figure 2);
Grade 4: multiple large osteophytes and/or definite bony end deformity.
Statistical analysis was performed on the compiled data using Stata software (StataCorp, College Station, Texas). Linear and logistic regression analyses correcting for confounding factors of age, sex, race, and height were performed using a standard P-value cutoff (P < .05) and 95% confidence interval to determine statistical significance.
Results
Patients were considered to have osteoarthritis of the tibiotalar joint if either of the extremities measured grade 1 or higher. Of the 710 specimens selected, 14 specimens did not have adequate bone available for bilateral tibiotalar joint measurement, either from extensive bone degradation or amputation. Of the remaining 696 specimens, 586 had some degree of tibiotalar osteoarthritis present (Table 2). Regression analysis showed a significant positive association between right- and left-ankle osteoarthritis (coefficient: 0.491, P < .01). Tibiotalar joint arthritis was classified as severe if either extremity had arthrosis of grade 3 or higher. Of the 586 specimens that had tibiotalar joint arthritis, only 16% (97 specimens) had severe tibiotalar joint arthritis.
Data regarding lumbar disc degeneration were available for 516 of the 710 specimens selected, 443 of which showed some disc degeneration. Disc degeneration was most prevalent and significant at the L4-L5 and L3-L4 intervertebral levels (Figures 3, 4). Of these 516 specimens, 30 had degeneration at 1 level, 47 specimens had degeneration at 2 levels, 29 specimens had degeneration at 3 levels, 52 had degeneration at 4 levels, and 285 specimens had degeneration at all 5 lumbar levels. The majority of specimens were found to have some degree of degeneration at all 5 lumbar spinal levels (Figure 5). Severe lumbar DDD was defined as grade 3 or higher osteoarthritis present in at least 1 of the 5 lumbar levels. Of the 516 specimens that showed some degree of disc degeneration, 152 were classified as severe. When stratified by number of spinal levels, only 30% of specimens were found to have evidence of severe arthrosis, the majority of which was located at only 1 lumbar segment (Figure 6).
Linear regression analysis of the data showed a statistically significant positive association between lumbar disc degeneration and tibiotalar osteoarthritis (coefficient: 0.844, P < .01), even when correcting for confounding factors, such as age, sex, and race (coefficient: 0.331, P < .01).
Additional analysis of the data demonstrated that tibiotalar joint arthritis remained significantly associated with lumbar DDD across each lumbar level: L1-L2 (coefficient: 0.269, P < .01), L2-L3 (coefficient: 0.283, P < .01), L3-L4 (coefficient: 0.299, P < .01), L4-L5 (coefficient: 0.240, P < .02), L5-S1 (coefficient: 0.167, P < .05).
The presence of 3 or more levels of lumbar DDD significantly increased the possibility of developing severe tibiotalar joint arthritis. Lumbar DDD that encompassed 3 levels showed the highest odds for development of severe tibiotalar joint arthritis with an odds ratio (OR) of 20.542 (Table 3).
When subjects were compared by decade, the mean grade of tibiotalar joint arthritis was significantly higher than lumbar DDD in specimens who died in their 20s and 30s. This difference was insignificant in the fourth decade, and thereafter the mean value of lumbar DDD surpassed that of tibiotalar joint arthritis (Figure 7).
In contrast, severe lumbar DDD was more prevalent than severe tibiotalar joint arthritis in individuals age 20 years or older (Figure 8). There were no specimens under age 20 years with severe lumbar DDD or severe tibiotalar joint arthritis.
Logistic regression showed that individuals with severe lumbar disc degeneration had significantly higher odds of developing severe ankle arthritis (OR: 1.93, P < .05). Similarly, individuals with severe tibiotalar joint arthritis were just as likely to develop severe lumbar DDD with an OR of 1.97 (P < .05).
Discussion
Multiple joint involvement in osteoarthritis is well established with a wide range of evidence linking lower extremity joint pathology and lumbar spinal disease. In 1983, Offierski and MacNab20 were the first to describe hip-spine syndrome. In the next year, a study by Sponseller and colleagues25 of pediatric patients after hip arthrodesis further substantiated the association between spine and extremity disease, and demonstrated a continued cause and effect relationship after surgery.
Lumbar spinal degeneration has also been correlated with knee osteoarthritis. Tsuji and colleagues26 reported that degenerative changes in spinal alignment result in increased thigh muscle tension and knee flexion. Furthermore, in their radiographic analysis of 682 individuals, Horvath and colleagues27 also showed that individuals with spinal degeneration had a higher prevalence of knee and hip osteoarthritis.
One might hypothesize from this evidence that lumbar spinal degeneration and ankle arthritis would also be interrelated, given their interconnected role in lower extremity force transmission. Surprisingly, the literature correlating lumbar degeneration and lower extremity osteoarthritis has overlooked this association and has focused solely on the hip and knee. To our knowledge, this study is the first to identify a statistically significant association between tibiotalar joint osteoarthritis and lumbar disc degeneration.
The literature supported analysis of our data. Miller and colleagues28 evaluated disc degeneration in 600 autopsy specimens using the Nachemson29 grading system. This system categorizes disc degeneration into 4 grades based on macroscopic appearance. Miller and colleagues28 reported evidence of degenerative changes as early as the second decade of life, primarily involving the L3–L4 and L4–L5 levels. Of note, the Nachemson29 classification system includes only evidence of marginal osteophytes in grade 4 disease, which was not identified by Miller and colleagues28 until the fourth decade. These results were similar to those in our study, in which the L3-L4 and L4-L5 intervertebral levels were most commonly affected. However, in our study, significant degenerative changes were found in the third decade of life.
In addition, the percentage of specimens with severe disc degeneration increased with each decade (Figure 8). A substantial amount of histologic evidence demonstrates the progression of disc degeneration with age. With increased age, there is a gradual decrease in the osmotic swelling of intervertebral discs30 and a 2-fold decrease in disc hydration between adolescence and the eighth decade.31 Furthermore, the nucleus pulposus undergoes progressive fibrosis,32,33 with a 5-fold decrease in the fixed-charge density of nucleus glycosaminoglycans,34 and a 2-fold increase in intervertebral disc creep while under compression after age 30 years.35
While analyzing our findings, we had difficulty in determining which pathologic condition debuts and, subsequently, affects the other. According to our results, the mean grade of tibiotalar joint arthritis was higher than that of DDD in specimens through the third and fourth decades of life (Figure 7). After the age of 50 years, the mean grade of DDD surpasses that of tibiotalar arthritis. This may be initially interpreted that development of tibiotalar joint arthritis precedes lumbar disc degeneration. Ankle osteoarthritis is relatively rare, and given that the vast majority of ankle osteoarthritis is secondary to trauma,22 we would expect to see a higher incidence of ankle osteoarthritis in a younger, more active cohort. In addition, given our finding that ankle arthritis is related to lumbar disc degeneration, one could speculate that tibiotalar arthritis at a young age predisposes an individual to developing lumbar degeneration later in life.
However, this conclusion is inherently flawed; closer examination of the data revealed that the mean grade of tibiotalar arthritis and DDD in the third and fourth decades is relatively low, between grade 0 and grade 1 (Figure 7). Therefore, it is difficult to arrive at a conclusion when comparing such small values. Second, we must remember that we are comparing an average value of disc degeneration across all lumbar levels. When a specimen has only 1 disc that is severely degenerated, this value is averaged across all 5 lumbar levels and, thus, the overall mean grade of arthrosis is significantly diminished.
In fact, data from previous studies concur with the second argument. Upper-level lumbar disc degeneration is relatively rare and the vast majority of patients with disc degeneration present with significant disease in only 1 or 2 discs.36,37 Analysis of the specimens in this study revealed bony evidence of disc degeneration present at all 5 lumbar levels in over half of the specimens examined (57%). However, the majority of specimens in this cohort exhibit only low-grade degeneration. When specimens were analyzed for severe arthrosis (grade 3 and higher), nearly half of the specimens were found to have severe disease involving only 1 intervertebral disc (Figure 6). Data from Miller and colleagues28 and the present study show that the upper lumbar levels were relatively spared; the L3-L4 and L4-L5 lumbar levels showed the highest prevalence and severity of degenerative change.
To address this issue, we evaluated the percentage of specimens per decade with severe arthrosis (grade 3 and higher) of at least 1 lumbar intervertebral disc and 1 tibiotalar joint. Severe lumbar disc degeneration was found to be more prevalent than severe ankle arthritis in individuals age 20 years or older (Figure 8). Therefore, we postulate that significant degenerative changes in the lumbar spine precede the development of severe ankle arthritis.
One can further speculate that sequelae from lumbar disc degeneration may lead to the development of tibiotalar arthritis, given our finding that severe lumbar degeneration predisposes an individual to the development of ankle arthritis. Because significant lumbar disc degeneration has long been known to result in both spinal nerve and cord compression, we hypothesize that this resultant neurocompression promotes altered gait and translation of atypical forces to the ankle and foot, thus predisposing to the onset and/or progression of osteoarthritis. In support of this hypothesis, Morag and colleagues15 demonstrated that neurologic compression produced an altered posture and gait because of lost motor function and afferent proprioceptive sensation. This form of neurologic compromise may exert atypical forces upon the foot and ankle, predisposing the joint to accelerated wear and primary arthrosis.
In addition, DDD involving 3 or more lumbar intervertebral levels was found to significantly increase the likelihood of the subject having severe tibiotalar joint arthritis. Provided that lumbar disc degeneration typically involves significant degeneration at 1 level, we assume that significant arthrosis at 3 or more levels correlates to an overall more severe DDD with a higher corresponding likelihood of neural compression. However, compression of peripheral lower extremity nerves has been shown to result in neuropathic arthropathy akin to the diabetic Charcot foot.38 This could be a possible mechanism of accelerated ankle arthritis, but this study did not examine soft-tissue disease nor take into account other medical comorbidities of each specimen, including genetic predispositions towards osteoarthritis.
It should be noted that the aforementioned causative relationship between lumbar disc degeneration and tibiotalar arthritis is speculative and cannot be demonstrated definitively by this investigation. We acknowledge limitations of this study and the need for further research of the possible causative mechanism(s) of accelerated ankle arthrosis secondary to lumbar spinal disease. Ideally, the questions posed by our report would be answered via a large prospective cohort study that utilized both serial imaging and autopsy analysis. Unfortunately, this form of study is logistically and financially difficult to perform.
This was a retrospective cadaveric study in which determination of arthrosis severity was based solely on bony evidence. Therefore, the role of soft-tissue disease in the pathogenesis of arthrosis of the lumbar spine and tibiotalar joint could not be assessed, nor could definitive associations to clinically symptomatic disease. We made the assumption that progression of bone degeneration in both the lumbar spine and tibiotalar joint corresponded equally to the associated soft-tissue changes. Given this assumption, we cannot definitively conclude that degeneration of the lumbar spine precedes that of the ankle, because the absence of magnetic resonance imaging or fresh autopsy specimens in our study misses the early degenerative changes in the discs that precede the bony alteration measured in our study. Furthermore, readers should note that since this study compared only bone morphology, no emphasis was placed on clinical manifestation of lumbar disc degeneration or tibiotalar joint arthritis. As mentioned earlier, radiologic evidence of disc degeneration was found in 90% of adults age 50 to 59 years, according to a study by Hult5; however, it is important to note that not all individuals studied were symptomatic clinically. Unfortunately, medical records were not available for the bony specimens, and clinical correlations could not be assessed during this investigation.
Furthermore, no special attention was given to other pathologic conditions observed during specimen measurement. The presence of diseases, such as osteoporosis, spondylolysis, or previous traumatic injury, may have had implications in the resultant joint degeneration. Finally, the evaluation of arthrosis was performed subjectively without measuring reliability. However, the present analysis includes a large sample, each joint type was reviewed by a single examiner, and used a classification system that was modeled on a validated grading system. Ideally, multiple individuals should have been used for each type of measurement, with subsequent analysis of intraobserver and interobserver reliability.
Conclusion
Based on our study of a large population of adult skeletal specimens, we ascertained that lumbar intervertebral disc degeneration and tibiotalar osteoarthritis are associated. The prevalence of severe lumbar disc degeneration was higher than that of tibiotalar joint arthritis in individuals age 20 years or older. This may suggest that gait changes from disc degeneration or neural compression in the lumbar spine may play a role in the development of ankle osteoarthritis. Additionally, subjects with severe disc degeneration were twice as likely to develop significant tibiotalar osteoarthritis. This must be considered in the differential when treating patients with degenerative changes of the lumbar spine and leg pain.
1. Mathers C, Fat DM, Boerma JT, for the World Health Organization. The Global Burden of Disease: 2004 Update. Geneva, Switzerland: World Health Organization, 2008.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35.
3. Kelsey JL, Githens PB, White AA, et al. An epidemiological study of lifting and twisting on the job and risk for acute prolapsed lumbar intervertebral disc. J Orthop Res. 1984;2(1):61-66.
4. Kellgren JH, Lawrence JS. Osteoarthrosis and disc degeneration in an urban population. Ann Rheum Dis. 1958;17(4):388-397.
5. Hult L. Cervical, dorsal and lumbar spinal syndromes; a field investigation of a non-selected material of 1200 workers in different occupations with special reference to disc degeneration and so-called muscular rheumatism. Acta Orthop Scand Suppl. 1954;17:65-73.
6. Hirsch C. The reaction of intervertebral discs to compression forces. J Bone Joint Surg Am. 1955;37(6):1188-1196.
7. Videman T, Nurminen M, Troup JD. Lumbar spinal pathology in cadaveric material in relation to history of back pain, occupation and physical loading. Spine. 1990;15(8):728-740.
8. Butler D, Trafimow JH, Andersson GB, McNeil TW, Huckman MS. Discs degenerate before facets. Spine. 1990;15(2):111-113.
9. Fujiwara A, Tamai K, Yamato M, et al. The relationship between facet joint osteoarthritis and disc degeneration of the lumbar spine: an MRI study. Eur Spine J. 1999;8(5):396-401.
10. Lipson SJ, Muir H. Experimental intervertebral disc degeneration: morphologic and proteoglycan changes over time. Arthritis Rheum. 1981;24(1):12-21.
11. Eisenstein S, Roberts S. The physiology of the disc and its clinical relevance. J Bone Joint Surg Br. 2003;85(5):633-636.
12. Hughes SP, Freemont AJ, Hukins DW, McGregor AH, Roberts S. The pathogenesis of degeneration of the intervertebral disc and emerging therapies in the management of back pain. J Bone Joint Surg Br. 2012;94(10):1298-1304.
13. Inoue N, Espinoza Orías AA. Biomechanics of intervertebral disk degeneration. Orthop Clin North Am. 2011;42(4):487-499.
14. Battié MC, Videman T. Lumbar disc degeneration: epidemiology and genetics. J Bone Joint Surg Am. 2006;88(suppl 2):3-9.
15. Morag E, Hurwitz DE, Andriacchi TP, Hickey M, Andersson GB. Abnormalities in muscle function during gait in relation to the level of lumbar disc herniation. Spine. 2000;25(7):829-833.
16. Oikawa Y, Ohtori S, Koshi T, et al. Lumbar disc degeneration induces persistent groin pain. Spine. 2012;37(2):114-118.
17. Porter RW. Spinal stenosis and neurogenic claudication. Spine. 1996;21(17):2046-2052.
18. Papadakis NC, Christakis DG, Tzagarakis GN, et al. Gait variability measurements in lumbar spinal stenosis patients: part A. Comparison with healthy subjects. Physiol Meas. 2009;30(11):1171-1186.
19. McGregor AH, Hukins DW. Lower limb involvement in spinal function and low back pain. J Back Musculoskelet Rehabil. 2009;22(4):219-222.
20. Offierski CM, MacNab I. Hip-spine syndrome. Spine. 1983;8(3):316-321.
21. Thomas RH, Daniels TR. Ankle arthritis. J Bone Joint Surg Am. 2003;85(5):923-936.
22. Valderrabano V, Horisberger M, Russell I, Dougall H, Hintermann B. Etiology of ankle osteoarthritis. Clin Orthop. 2009;467(7):1800-1806.
23. Eubanks JD, Lee MJ, Cassinelli E, Ahn NU. Does lumbar facet arthrosis precede disc degeneration? A postmortem study. Clin Orthop. 2007;464:184-189.
24. Friberg S, Hirsch C. Anatomical and clinical changes in lumbar disc degeneration. Acta Orthop Scand. 1949;19(2):222-242.
25. Sponseller PD, McBeath AA, Perpich M. Hip arthrodesis in young patients. A long-term follow-up study. J Bone Joint Surg Am. 1984;66(6):853-859.
26. Tsuji T, Matsuyama Y, Goto M, et al. Knee-spine syndrome: correlation between sacral inclination and patellofemoral joint pain. J Orthop Sci. 2002;7(5):519-523.
27. Horvath G, Koroknai G, Acs B, Than P, Illés T. Prevalence of low back pain and lumbar spine degenerative disorders. Questionnaire survey and clinical-radiological analysis of a representative Hungarian population. Int Orthop. 2010;34(8):1245-1249.
28. Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine. 1988;13(2):173-178.
29. Nachemson A. Lumbar intradiscal pressure: experimental studies on post-mortem material. Acta Orthop Scand Suppl. 1960;43:1-104.
30. Kraemer J. Pressure-dependent fluid shifts in the intervertebral disc. Orthop Clin North Am. 1977;8(1):211-216.
31. Urban JP, McMullin JF. Swelling pressure of the intervertebral disc: influence of proteoglycan and collagen contents. Biorheology. 1985;22(2):145-157.
32. Coventry MB, Ghromley RK, Kernohan JW. The intervertebral disc, its macroscopic anatomy and pathology: Part III. Pathologic changes in the intervertebral disc. J Bone Joint Surg Br. 1945;27:460-474.
33. Friberg S, Hirsch C. Anatomical and clinical changes in lumbar disc degeneration. Acta Orthop Scand. 1949;19(2):222-242.
34. Lyons G, Eisenstein SM, Sweet MB. Biochemical changes in intervertebral disc degeneration. Biochim Biophys Acta. 1981;673(4):443-453.
35. Koeller W, Muehlhaus S, Meier W, Hartmann F. Biomechanical properties of human intervertebral discs subjected to axial dynamic compression: influence of age and degeneration. J Biomech. 1986;19(10):807-816.
36. Bosacco SJ, Berman AT, Raisis LW, Zamarin RI. High lumbar herniations. Case reports. Orthopaedics. 1989;12(2):275-278.
37. Spangfort EV. The lumbar disc herniation. A computer-aided analysis of 2,504 operations. Acta Orthop Scand Suppl. 1972;142:1-95.
38. Gupta R. A short history of neuropathic arthropathy. Clin Orthop. 1993;296:43-49.
1. Mathers C, Fat DM, Boerma JT, for the World Health Organization. The Global Burden of Disease: 2004 Update. Geneva, Switzerland: World Health Organization, 2008.
2. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58(1):26-35.
3. Kelsey JL, Githens PB, White AA, et al. An epidemiological study of lifting and twisting on the job and risk for acute prolapsed lumbar intervertebral disc. J Orthop Res. 1984;2(1):61-66.
4. Kellgren JH, Lawrence JS. Osteoarthrosis and disc degeneration in an urban population. Ann Rheum Dis. 1958;17(4):388-397.
5. Hult L. Cervical, dorsal and lumbar spinal syndromes; a field investigation of a non-selected material of 1200 workers in different occupations with special reference to disc degeneration and so-called muscular rheumatism. Acta Orthop Scand Suppl. 1954;17:65-73.
6. Hirsch C. The reaction of intervertebral discs to compression forces. J Bone Joint Surg Am. 1955;37(6):1188-1196.
7. Videman T, Nurminen M, Troup JD. Lumbar spinal pathology in cadaveric material in relation to history of back pain, occupation and physical loading. Spine. 1990;15(8):728-740.
8. Butler D, Trafimow JH, Andersson GB, McNeil TW, Huckman MS. Discs degenerate before facets. Spine. 1990;15(2):111-113.
9. Fujiwara A, Tamai K, Yamato M, et al. The relationship between facet joint osteoarthritis and disc degeneration of the lumbar spine: an MRI study. Eur Spine J. 1999;8(5):396-401.
10. Lipson SJ, Muir H. Experimental intervertebral disc degeneration: morphologic and proteoglycan changes over time. Arthritis Rheum. 1981;24(1):12-21.
11. Eisenstein S, Roberts S. The physiology of the disc and its clinical relevance. J Bone Joint Surg Br. 2003;85(5):633-636.
12. Hughes SP, Freemont AJ, Hukins DW, McGregor AH, Roberts S. The pathogenesis of degeneration of the intervertebral disc and emerging therapies in the management of back pain. J Bone Joint Surg Br. 2012;94(10):1298-1304.
13. Inoue N, Espinoza Orías AA. Biomechanics of intervertebral disk degeneration. Orthop Clin North Am. 2011;42(4):487-499.
14. Battié MC, Videman T. Lumbar disc degeneration: epidemiology and genetics. J Bone Joint Surg Am. 2006;88(suppl 2):3-9.
15. Morag E, Hurwitz DE, Andriacchi TP, Hickey M, Andersson GB. Abnormalities in muscle function during gait in relation to the level of lumbar disc herniation. Spine. 2000;25(7):829-833.
16. Oikawa Y, Ohtori S, Koshi T, et al. Lumbar disc degeneration induces persistent groin pain. Spine. 2012;37(2):114-118.
17. Porter RW. Spinal stenosis and neurogenic claudication. Spine. 1996;21(17):2046-2052.
18. Papadakis NC, Christakis DG, Tzagarakis GN, et al. Gait variability measurements in lumbar spinal stenosis patients: part A. Comparison with healthy subjects. Physiol Meas. 2009;30(11):1171-1186.
19. McGregor AH, Hukins DW. Lower limb involvement in spinal function and low back pain. J Back Musculoskelet Rehabil. 2009;22(4):219-222.
20. Offierski CM, MacNab I. Hip-spine syndrome. Spine. 1983;8(3):316-321.
21. Thomas RH, Daniels TR. Ankle arthritis. J Bone Joint Surg Am. 2003;85(5):923-936.
22. Valderrabano V, Horisberger M, Russell I, Dougall H, Hintermann B. Etiology of ankle osteoarthritis. Clin Orthop. 2009;467(7):1800-1806.
23. Eubanks JD, Lee MJ, Cassinelli E, Ahn NU. Does lumbar facet arthrosis precede disc degeneration? A postmortem study. Clin Orthop. 2007;464:184-189.
24. Friberg S, Hirsch C. Anatomical and clinical changes in lumbar disc degeneration. Acta Orthop Scand. 1949;19(2):222-242.
25. Sponseller PD, McBeath AA, Perpich M. Hip arthrodesis in young patients. A long-term follow-up study. J Bone Joint Surg Am. 1984;66(6):853-859.
26. Tsuji T, Matsuyama Y, Goto M, et al. Knee-spine syndrome: correlation between sacral inclination and patellofemoral joint pain. J Orthop Sci. 2002;7(5):519-523.
27. Horvath G, Koroknai G, Acs B, Than P, Illés T. Prevalence of low back pain and lumbar spine degenerative disorders. Questionnaire survey and clinical-radiological analysis of a representative Hungarian population. Int Orthop. 2010;34(8):1245-1249.
28. Miller JA, Schmatz C, Schultz AB. Lumbar disc degeneration: correlation with age, sex, and spine level in 600 autopsy specimens. Spine. 1988;13(2):173-178.
29. Nachemson A. Lumbar intradiscal pressure: experimental studies on post-mortem material. Acta Orthop Scand Suppl. 1960;43:1-104.
30. Kraemer J. Pressure-dependent fluid shifts in the intervertebral disc. Orthop Clin North Am. 1977;8(1):211-216.
31. Urban JP, McMullin JF. Swelling pressure of the intervertebral disc: influence of proteoglycan and collagen contents. Biorheology. 1985;22(2):145-157.
32. Coventry MB, Ghromley RK, Kernohan JW. The intervertebral disc, its macroscopic anatomy and pathology: Part III. Pathologic changes in the intervertebral disc. J Bone Joint Surg Br. 1945;27:460-474.
33. Friberg S, Hirsch C. Anatomical and clinical changes in lumbar disc degeneration. Acta Orthop Scand. 1949;19(2):222-242.
34. Lyons G, Eisenstein SM, Sweet MB. Biochemical changes in intervertebral disc degeneration. Biochim Biophys Acta. 1981;673(4):443-453.
35. Koeller W, Muehlhaus S, Meier W, Hartmann F. Biomechanical properties of human intervertebral discs subjected to axial dynamic compression: influence of age and degeneration. J Biomech. 1986;19(10):807-816.
36. Bosacco SJ, Berman AT, Raisis LW, Zamarin RI. High lumbar herniations. Case reports. Orthopaedics. 1989;12(2):275-278.
37. Spangfort EV. The lumbar disc herniation. A computer-aided analysis of 2,504 operations. Acta Orthop Scand Suppl. 1972;142:1-95.
38. Gupta R. A short history of neuropathic arthropathy. Clin Orthop. 1993;296:43-49.
Revision Anterior Cruciate Ligament Reconstruction With Bone–Patellar Tendon–Bone Allograft and Extra-Articular Iliotibial Band Tenodesis
Primary anterior cruciate ligament (ACL) reconstruction has satisfactory outcomes in 75% to 97% of patients.1-3 Despite this high success rate, the number of revision ACL reconstructions has risen4 and is likely underreported.5 Recurrent instability occurs if the reconstructed ligament fails to provide adequate anterior and rotational knee stability. Causes of graft failure include repeat trauma, early return to high-demand activity, poor operative technique (including poor graft placement), failure to address concomitant pathology, and perioperative complications (eg, infection, stiffness).4 In addition, most patients who have revision ACL reconstruction received autograft tissue in the initial surgery, and allograft is thus not uncommon in revision ACL surgery. Allograft tissue has longer incorporation times6 and increased incidence of recurrent postoperative instability when compared with autograft tissue.7 Extra-articular tenodesis may thus be used to provide additional stability to the revision allograft tissue while it incorporates.
In this article, we describe our use of an extra-articular iliotibial band (ITB) tenodesis as an augmentative procedure in patients undergoing revision ACL reconstruction with bone–patellar tendon–bone (BPTB) allograft.
Surgical Technique
After induction of anesthesia and careful positioning, the patient is prepared and draped in the usual sterile fashion. Standard anteromedial, anterolateral, and superolateral outflow portals are established, and diagnostic arthroscopy is performed to inspect the cruciate ligaments, menisci, and articular cartilage (Figure 1). Peripheral meniscal tears should be repaired (Figure 2), and central or inner tears should be débrided to a stable rim. If meniscal repair is performed, sutures should be tied at the end of the case. Unstable articular cartilage defects should also be débrided. An 8- to 12-cm lateral hockey-stick incision is then made from the Gerdy tubercle to the inferior edge of the lateral femoral epicondyle in preparation for the ITB tenodesis (Figure 1). The lateral collateral ligament (LCL), the lateral head of the gastrocnemius, and the ITB are identified. The peroneal nerve should be significantly distal to the working field.
Remnants of the previous ACL graft are débrided, and, if necessary, a modified notchplasty is performed. A position for the new femoral tunnel is located and is confirmed with intraoperative fluoroscopy. This tunnel is established with compaction drill bits and dilated to the appropriate diameter through the anteromedial portal with the knee in 120° of flexion.
BPTB allograft is prepared first by cutting its central third to the desired diameter (Figure 3). The bone-plug ends are prepared with compaction pliers. Two 2.0-mm drill holes are made in each of the allograft bone plugs, and a No. 5 Ti-Cron suture (Covidien, New Haven, Connecticut) is placed through each of the holes. We typically use 2 sutures on each bone plug.
A tibial tunnel is then established with an ACL drill guide under arthroscopic visualization and intraoperative fluoroscopy for confirmation of correct pin placement. We use Kirschner wires (with parallel pin guides as needed), compaction drills, and dilators to create a well-positioned tunnel of the appropriate diameter. The allograft is then passed through the tibia and femur in retrograde fashion. We secure the femoral side with an AO (Arbeitsgemeinschaft für Osteosynthesefragen) 4.5-mm bicortical screw and washer. Our tibial fixation is secured after the ITB tenodesis. The knee is then cycled a dozen times.
In preparation for the ITB tenodesis, we lengthen our previously made incision by about 4 cm proximally along the posterior aspect of the ITB. The central portion of the ITB is then outlined at the Gerdy tubercle and split with a No. 10 blade. This generally leaves an approximately 12- to 14-mm strip of ITB centrally (Figure 4). This portion should be gently lifted from the underlying tissue attachments distally at the insertion on the Gerdy tubercle. The interval between the LCL and lateral capsule of the knee is identified, and a No. 2 Ti-Cron whip-stitch is thrown through the free end of the ITB graft (Figure 5). The anterior aspect of the femoral tunnel is at the distal aspect of the lateral femoral condyle, and the posterior aspect is at the juncture of the proximal LCL and the lateral head of the gastrocnemius. The cortices of these landmarks should be perforated with a drill, and a curved instrument should be used to create a bone tunnel at this location (Figure 6). The tibial tunnel is just posterior and distal to the Gerdy tubercle and should be created in similar fashion. The graft is then passed underneath the LCL (Figure 7), through the proximal tunnel that has been created on the lateral femoral condyle, and then back down through the LCL and back onto itself after exiting the tibial tunnel (Figure 8). With the knee at 30° of flexion, the ITB graft is tensioned and sutured down to intact ITB fascia just proximal to the tibial tunnel orifice (Figure 9). We check knee range of motion (ROM) and then perform a Lachman test to assess changes in knee stability. The pivot shift examination is omitted to avoid placing excessive stress on the tenodesis. The tibial side of the patellar tendon allograft is then tensioned and secured over an AO 4.5-mm bicortical screw with washer with the knee in full extension. The screw is then tightened at 30° of knee flexion.
The ITB fascia is closed to the lateral femoral epicondyle with a running heavy suture, and all incisions are then irrigated and closed (Figures 10, 11). Standard sterile surgical dressing, Cryo/Cuff (Aircast, Vista, California), and brace are applied with the knee locked at 20°. Patients are generally discharged home the same day and followed up in clinic 1 week after surgery.
Complications
The peroneal nerve must be identified and protected during the open lateral procedure. In addition, the need for the extra lateral incision poses a slightly higher risk for infection compared with the traditional arthroscopic revision ACL procedure. Last, the additional tunnels required for the tenodesis can increase the theoretical potential for distal femur fracture and ACL graft fixation failure on the femoral side.
Postoperative Management
The operative knee is kept in extension in a brace locked at 20° for week 1 after surgery. Isometric quadriceps exercises are started immediately after surgery. Flexion to 90° is allowed starting week 2 after surgery, when the patient begins supervised active/passive flexion and progressive ROM exercises. In most cases, full ROM should be achieved by 6 to 8 weeks after surgery. Patients are progressed in their weight-bearing status by about 25% of their body weight per week, and use of crutches should be discontinued by week 4 after surgery. The brace should be discontinued by week 6 after surgery, when use of stationary bicycle and closed chain exercises begin. The patient may begin jogging when the operative leg regains 80% of contralateral quadriceps strength via Cybex strength testing. Functional drills begin in month 6, but patients should be counseled against returning to sport any earlier than 9 months after surgery.
Discussion
Achieving a successful outcome in revision ACL surgery (vs primary ACL surgery) is a significant challenge. Any of numerous factors can make the revision surgery more challenging, including existing poorly placed tunnels, tunnel expansion, lack of ideal graft choice, loss of secondary stabilizers, and deviations of the weight-bearing axis. Therefore, outcomes of revision surgery tend to be more moderate than outcomes of primary procedures.4,8-12
Revision ACL reconstruction techniques are varied and can involve use of autograft or allograft tissue as well as extra-articular augmentation techniques. Diamantopoulos and colleagues8 reported the outcomes of revision ACL reconstruction using bone–tendon–bone, hamstring, or quadriceps autografts in 107 patients. The majority of patients had improved outcome measures (mean Lysholm score improved from 51.5 to 88.5) and side-to-side laxity measurements. However, only 36.4% returned to preinjury activity level. Similarly, Noyes and Barber-Westin9 reported the outcomes of revision ACL reconstruction using quadriceps tendon–patellar bone autograft in 21 patients. Although there was significant improvement in terms of symptoms and activity level, 4 of the 21 knees were graded abnormal or severely abnormal on the IKDC (International Knee Documentation Committee) ligament rating. In a systematic review, pooled results of revision ACL reconstructions reiterated the above results.10 Eight hundred sixty-three patients from 21 studies were included in the analysis, which found significantly worse subjective outcomes than for primary procedures and a dramatically higher failure rate for the re-reconstructed ACL.
Several authors have directly compared primary cohorts with revision cohorts. Ahn and colleagues11 compared the outcomes of 59 revision ACL reconstructions with those of 117 primary reconstructions at a single institution. Although statistical comparison of stability between primary and revision ACL reconstructions showed no difference, revision reconstructions fared more poorly in terms of quality of life and return to activity compared with primary reconstructions. In a large cohort study of the Danish registry, revisions were found to have worse subjective outcomes than primary reconstructions as well.12 The study also found that the rerupture risk was significantly higher (relative risk, 2.05) when allograft was used.
Given the inferior results of revision surgery, our technique is recommended to augment the stability of reconstructed knees in the setting of revision ACL reconstruction. Adding the extra-articular procedure may augment the revised graft and protect it from excessive stress.13 A cadaver study compared double-bundle ACL reconstruction with single-bundle hamstring reconstruction plus extra-articular lateral tenodesis and found improved internal rotation control at 30° of flexion in the latter.14 Using contralateral 4-strand hamstring autograft in combination with an extra-articular lateral augment can have encouraging outcomes. Ferretti and colleagues15 reported an average Lysholm score of 95 in 12 patients who underwent this revision procedure and good anterior-to-posterior stability in 11 of the 12 patients. Trojani and colleagues16 reported on a cohort of 163 patients who underwent ACL revision surgery over a 10-year period. The authors found that 80% of patients with a lateral extra-articular tenodesis performed to augment their revision reconstruction had a negative pivot shift at long-term follow-up—versus only 63% of patients who underwent isolated revision ACL reconstruction. This finding was statistically significant, but the authors did not find any differences in IKDC scores between groups. These results support the initial biomechanical findings of Engebretsen and colleagues,17 who found that adding a lateral tenodesis decreased the forces on the reconstructed graft by 15%.
Conclusion
This technique allows for protection of the intra-articular allograft ligament reconstruction with improved rotational control that may potentially allow for improved subjective outcomes and protect against graft failure. Given the common pitfalls with stability in revision ACL surgery with allograft, this lateral extra-articular procedure can be an important structural augmentation in this challenging clinical issue in knee surgery.
1. Bach BR Jr. Revision anterior cruciate ligament surgery. Arthroscopy. 2003;19(suppl 1):14-29.
2. Baer GS, Harner CD. Clinical outcomes of allograft versus autograft in anterior cruciate ligament reconstruction. Clin Sports Med. 2007;26(4):661-681.
3. Spindler KP, Kuhn JE, Freedman KB, Matthews CE, Dittus RS, Harrell FE Jr. Anterior cruciate ligament reconstruction autograft choice: bone–tendon–bone versus hamstring: does it really matter? A systematic review. Am J Sports Med. 2004;32(8):1986-1995.
4. Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39(1):199-217.
5. Gianotti SM, Marshall SW, Hume PA, Bunt L. Incidence of anterior cruciate ligament injury and other knee ligament injuries: a national population-based study. J Sci Med Sport. 2009;12(6):622-627.
6. Jackson DW, Grood ES, Goldstein JD, et al. A comparison of patellar tendon autograft and allograft used for anterior cruciate ligament reconstruction in the goat model. Am J Sports Med. 1993;21(2):176-185.
7. Mascarenhas R, Tranovich M, Karpie JC, Irrgang JJ, Fu FH, Harner CD. Patellar tendon anterior cruciate ligament reconstruction in the high-demand patient: evaluation of autograft versus allograft reconstruction. Arthroscopy. 2010;26(9 Suppl):S58-S66.
8. Diamantopoulos AP, Lorbach O, Paessler HH. Anterior cruciate ligament revision reconstruction: results in 107 patients. Am J Sports Med. 2008;36(5):851-860.
9. Noyes FR, Barber-Westin SD. Anterior cruciate ligament revision reconstruction: results using a quadriceps tendon–patellar bone autograft. Am J Sports Med. 2006;34(4):553-564.
10. Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531-536.
11. Ahn JH, Lee YS, Ha HC. Comparison of revision surgery with primary anterior cruciate ligament reconstruction and outcome of revision surgery between different graft materials. Am J Sports Med. 2008;36(10):1889-1895.
12. Lind M, Menhert F, Pedersen AB. Incidence and outcome after revision anterior cruciate ligament reconstruction: results from the Danish registry for knee ligament reconstructions. Am J Sports Med. 2012;40(7):1551-1557.
13. Ferretti A, Conteduca F, Monaco E, De Carli A, D’Arrigo C. Revision anterior cruciate ligament reconstruction with doubled semitendinosus and gracilis tendons and lateral extra-articular reconstruction. J Bone Joint Surg Am. 2006;88(11):2373-2379.
14. Monaco E, Labianca L, Conteduca F, De Carli A, Ferretti A. Double bundle or single bundle plus extraarticular tenodesis in ACL reconstruction? A CAOS study. Knee Surg Sports Traumatol Arthrosc. 2007;15(10):1168-1174.
15. Ferretti A, Monaco E, Caperna L, Palma T, Conteduca F. Revision ACL reconstruction using contralateral hamstrings. Knee Surg Sports Traumatol Arthrosc. 2013;21(3):690-695.
16. Trojani C, Beaufils P, Burdin G, et al. Revision ACL reconstruction: influence of a lateral tenodesis. Knee Surg Sports Traumatol Arthrosc. 2012;20(8):1565-1570.
17. Engebretsen L, Lew WD, Lewis JL, Hunter RE. The effect of an iliotibial tenodesis on intraarticular graft forces and knee joint motion. Am J Sports Med. 1990;18(2):169-176.
Primary anterior cruciate ligament (ACL) reconstruction has satisfactory outcomes in 75% to 97% of patients.1-3 Despite this high success rate, the number of revision ACL reconstructions has risen4 and is likely underreported.5 Recurrent instability occurs if the reconstructed ligament fails to provide adequate anterior and rotational knee stability. Causes of graft failure include repeat trauma, early return to high-demand activity, poor operative technique (including poor graft placement), failure to address concomitant pathology, and perioperative complications (eg, infection, stiffness).4 In addition, most patients who have revision ACL reconstruction received autograft tissue in the initial surgery, and allograft is thus not uncommon in revision ACL surgery. Allograft tissue has longer incorporation times6 and increased incidence of recurrent postoperative instability when compared with autograft tissue.7 Extra-articular tenodesis may thus be used to provide additional stability to the revision allograft tissue while it incorporates.
In this article, we describe our use of an extra-articular iliotibial band (ITB) tenodesis as an augmentative procedure in patients undergoing revision ACL reconstruction with bone–patellar tendon–bone (BPTB) allograft.
Surgical Technique
After induction of anesthesia and careful positioning, the patient is prepared and draped in the usual sterile fashion. Standard anteromedial, anterolateral, and superolateral outflow portals are established, and diagnostic arthroscopy is performed to inspect the cruciate ligaments, menisci, and articular cartilage (Figure 1). Peripheral meniscal tears should be repaired (Figure 2), and central or inner tears should be débrided to a stable rim. If meniscal repair is performed, sutures should be tied at the end of the case. Unstable articular cartilage defects should also be débrided. An 8- to 12-cm lateral hockey-stick incision is then made from the Gerdy tubercle to the inferior edge of the lateral femoral epicondyle in preparation for the ITB tenodesis (Figure 1). The lateral collateral ligament (LCL), the lateral head of the gastrocnemius, and the ITB are identified. The peroneal nerve should be significantly distal to the working field.
Remnants of the previous ACL graft are débrided, and, if necessary, a modified notchplasty is performed. A position for the new femoral tunnel is located and is confirmed with intraoperative fluoroscopy. This tunnel is established with compaction drill bits and dilated to the appropriate diameter through the anteromedial portal with the knee in 120° of flexion.
BPTB allograft is prepared first by cutting its central third to the desired diameter (Figure 3). The bone-plug ends are prepared with compaction pliers. Two 2.0-mm drill holes are made in each of the allograft bone plugs, and a No. 5 Ti-Cron suture (Covidien, New Haven, Connecticut) is placed through each of the holes. We typically use 2 sutures on each bone plug.
A tibial tunnel is then established with an ACL drill guide under arthroscopic visualization and intraoperative fluoroscopy for confirmation of correct pin placement. We use Kirschner wires (with parallel pin guides as needed), compaction drills, and dilators to create a well-positioned tunnel of the appropriate diameter. The allograft is then passed through the tibia and femur in retrograde fashion. We secure the femoral side with an AO (Arbeitsgemeinschaft für Osteosynthesefragen) 4.5-mm bicortical screw and washer. Our tibial fixation is secured after the ITB tenodesis. The knee is then cycled a dozen times.
In preparation for the ITB tenodesis, we lengthen our previously made incision by about 4 cm proximally along the posterior aspect of the ITB. The central portion of the ITB is then outlined at the Gerdy tubercle and split with a No. 10 blade. This generally leaves an approximately 12- to 14-mm strip of ITB centrally (Figure 4). This portion should be gently lifted from the underlying tissue attachments distally at the insertion on the Gerdy tubercle. The interval between the LCL and lateral capsule of the knee is identified, and a No. 2 Ti-Cron whip-stitch is thrown through the free end of the ITB graft (Figure 5). The anterior aspect of the femoral tunnel is at the distal aspect of the lateral femoral condyle, and the posterior aspect is at the juncture of the proximal LCL and the lateral head of the gastrocnemius. The cortices of these landmarks should be perforated with a drill, and a curved instrument should be used to create a bone tunnel at this location (Figure 6). The tibial tunnel is just posterior and distal to the Gerdy tubercle and should be created in similar fashion. The graft is then passed underneath the LCL (Figure 7), through the proximal tunnel that has been created on the lateral femoral condyle, and then back down through the LCL and back onto itself after exiting the tibial tunnel (Figure 8). With the knee at 30° of flexion, the ITB graft is tensioned and sutured down to intact ITB fascia just proximal to the tibial tunnel orifice (Figure 9). We check knee range of motion (ROM) and then perform a Lachman test to assess changes in knee stability. The pivot shift examination is omitted to avoid placing excessive stress on the tenodesis. The tibial side of the patellar tendon allograft is then tensioned and secured over an AO 4.5-mm bicortical screw with washer with the knee in full extension. The screw is then tightened at 30° of knee flexion.
The ITB fascia is closed to the lateral femoral epicondyle with a running heavy suture, and all incisions are then irrigated and closed (Figures 10, 11). Standard sterile surgical dressing, Cryo/Cuff (Aircast, Vista, California), and brace are applied with the knee locked at 20°. Patients are generally discharged home the same day and followed up in clinic 1 week after surgery.
Complications
The peroneal nerve must be identified and protected during the open lateral procedure. In addition, the need for the extra lateral incision poses a slightly higher risk for infection compared with the traditional arthroscopic revision ACL procedure. Last, the additional tunnels required for the tenodesis can increase the theoretical potential for distal femur fracture and ACL graft fixation failure on the femoral side.
Postoperative Management
The operative knee is kept in extension in a brace locked at 20° for week 1 after surgery. Isometric quadriceps exercises are started immediately after surgery. Flexion to 90° is allowed starting week 2 after surgery, when the patient begins supervised active/passive flexion and progressive ROM exercises. In most cases, full ROM should be achieved by 6 to 8 weeks after surgery. Patients are progressed in their weight-bearing status by about 25% of their body weight per week, and use of crutches should be discontinued by week 4 after surgery. The brace should be discontinued by week 6 after surgery, when use of stationary bicycle and closed chain exercises begin. The patient may begin jogging when the operative leg regains 80% of contralateral quadriceps strength via Cybex strength testing. Functional drills begin in month 6, but patients should be counseled against returning to sport any earlier than 9 months after surgery.
Discussion
Achieving a successful outcome in revision ACL surgery (vs primary ACL surgery) is a significant challenge. Any of numerous factors can make the revision surgery more challenging, including existing poorly placed tunnels, tunnel expansion, lack of ideal graft choice, loss of secondary stabilizers, and deviations of the weight-bearing axis. Therefore, outcomes of revision surgery tend to be more moderate than outcomes of primary procedures.4,8-12
Revision ACL reconstruction techniques are varied and can involve use of autograft or allograft tissue as well as extra-articular augmentation techniques. Diamantopoulos and colleagues8 reported the outcomes of revision ACL reconstruction using bone–tendon–bone, hamstring, or quadriceps autografts in 107 patients. The majority of patients had improved outcome measures (mean Lysholm score improved from 51.5 to 88.5) and side-to-side laxity measurements. However, only 36.4% returned to preinjury activity level. Similarly, Noyes and Barber-Westin9 reported the outcomes of revision ACL reconstruction using quadriceps tendon–patellar bone autograft in 21 patients. Although there was significant improvement in terms of symptoms and activity level, 4 of the 21 knees were graded abnormal or severely abnormal on the IKDC (International Knee Documentation Committee) ligament rating. In a systematic review, pooled results of revision ACL reconstructions reiterated the above results.10 Eight hundred sixty-three patients from 21 studies were included in the analysis, which found significantly worse subjective outcomes than for primary procedures and a dramatically higher failure rate for the re-reconstructed ACL.
Several authors have directly compared primary cohorts with revision cohorts. Ahn and colleagues11 compared the outcomes of 59 revision ACL reconstructions with those of 117 primary reconstructions at a single institution. Although statistical comparison of stability between primary and revision ACL reconstructions showed no difference, revision reconstructions fared more poorly in terms of quality of life and return to activity compared with primary reconstructions. In a large cohort study of the Danish registry, revisions were found to have worse subjective outcomes than primary reconstructions as well.12 The study also found that the rerupture risk was significantly higher (relative risk, 2.05) when allograft was used.
Given the inferior results of revision surgery, our technique is recommended to augment the stability of reconstructed knees in the setting of revision ACL reconstruction. Adding the extra-articular procedure may augment the revised graft and protect it from excessive stress.13 A cadaver study compared double-bundle ACL reconstruction with single-bundle hamstring reconstruction plus extra-articular lateral tenodesis and found improved internal rotation control at 30° of flexion in the latter.14 Using contralateral 4-strand hamstring autograft in combination with an extra-articular lateral augment can have encouraging outcomes. Ferretti and colleagues15 reported an average Lysholm score of 95 in 12 patients who underwent this revision procedure and good anterior-to-posterior stability in 11 of the 12 patients. Trojani and colleagues16 reported on a cohort of 163 patients who underwent ACL revision surgery over a 10-year period. The authors found that 80% of patients with a lateral extra-articular tenodesis performed to augment their revision reconstruction had a negative pivot shift at long-term follow-up—versus only 63% of patients who underwent isolated revision ACL reconstruction. This finding was statistically significant, but the authors did not find any differences in IKDC scores between groups. These results support the initial biomechanical findings of Engebretsen and colleagues,17 who found that adding a lateral tenodesis decreased the forces on the reconstructed graft by 15%.
Conclusion
This technique allows for protection of the intra-articular allograft ligament reconstruction with improved rotational control that may potentially allow for improved subjective outcomes and protect against graft failure. Given the common pitfalls with stability in revision ACL surgery with allograft, this lateral extra-articular procedure can be an important structural augmentation in this challenging clinical issue in knee surgery.
Primary anterior cruciate ligament (ACL) reconstruction has satisfactory outcomes in 75% to 97% of patients.1-3 Despite this high success rate, the number of revision ACL reconstructions has risen4 and is likely underreported.5 Recurrent instability occurs if the reconstructed ligament fails to provide adequate anterior and rotational knee stability. Causes of graft failure include repeat trauma, early return to high-demand activity, poor operative technique (including poor graft placement), failure to address concomitant pathology, and perioperative complications (eg, infection, stiffness).4 In addition, most patients who have revision ACL reconstruction received autograft tissue in the initial surgery, and allograft is thus not uncommon in revision ACL surgery. Allograft tissue has longer incorporation times6 and increased incidence of recurrent postoperative instability when compared with autograft tissue.7 Extra-articular tenodesis may thus be used to provide additional stability to the revision allograft tissue while it incorporates.
In this article, we describe our use of an extra-articular iliotibial band (ITB) tenodesis as an augmentative procedure in patients undergoing revision ACL reconstruction with bone–patellar tendon–bone (BPTB) allograft.
Surgical Technique
After induction of anesthesia and careful positioning, the patient is prepared and draped in the usual sterile fashion. Standard anteromedial, anterolateral, and superolateral outflow portals are established, and diagnostic arthroscopy is performed to inspect the cruciate ligaments, menisci, and articular cartilage (Figure 1). Peripheral meniscal tears should be repaired (Figure 2), and central or inner tears should be débrided to a stable rim. If meniscal repair is performed, sutures should be tied at the end of the case. Unstable articular cartilage defects should also be débrided. An 8- to 12-cm lateral hockey-stick incision is then made from the Gerdy tubercle to the inferior edge of the lateral femoral epicondyle in preparation for the ITB tenodesis (Figure 1). The lateral collateral ligament (LCL), the lateral head of the gastrocnemius, and the ITB are identified. The peroneal nerve should be significantly distal to the working field.
Remnants of the previous ACL graft are débrided, and, if necessary, a modified notchplasty is performed. A position for the new femoral tunnel is located and is confirmed with intraoperative fluoroscopy. This tunnel is established with compaction drill bits and dilated to the appropriate diameter through the anteromedial portal with the knee in 120° of flexion.
BPTB allograft is prepared first by cutting its central third to the desired diameter (Figure 3). The bone-plug ends are prepared with compaction pliers. Two 2.0-mm drill holes are made in each of the allograft bone plugs, and a No. 5 Ti-Cron suture (Covidien, New Haven, Connecticut) is placed through each of the holes. We typically use 2 sutures on each bone plug.
A tibial tunnel is then established with an ACL drill guide under arthroscopic visualization and intraoperative fluoroscopy for confirmation of correct pin placement. We use Kirschner wires (with parallel pin guides as needed), compaction drills, and dilators to create a well-positioned tunnel of the appropriate diameter. The allograft is then passed through the tibia and femur in retrograde fashion. We secure the femoral side with an AO (Arbeitsgemeinschaft für Osteosynthesefragen) 4.5-mm bicortical screw and washer. Our tibial fixation is secured after the ITB tenodesis. The knee is then cycled a dozen times.
In preparation for the ITB tenodesis, we lengthen our previously made incision by about 4 cm proximally along the posterior aspect of the ITB. The central portion of the ITB is then outlined at the Gerdy tubercle and split with a No. 10 blade. This generally leaves an approximately 12- to 14-mm strip of ITB centrally (Figure 4). This portion should be gently lifted from the underlying tissue attachments distally at the insertion on the Gerdy tubercle. The interval between the LCL and lateral capsule of the knee is identified, and a No. 2 Ti-Cron whip-stitch is thrown through the free end of the ITB graft (Figure 5). The anterior aspect of the femoral tunnel is at the distal aspect of the lateral femoral condyle, and the posterior aspect is at the juncture of the proximal LCL and the lateral head of the gastrocnemius. The cortices of these landmarks should be perforated with a drill, and a curved instrument should be used to create a bone tunnel at this location (Figure 6). The tibial tunnel is just posterior and distal to the Gerdy tubercle and should be created in similar fashion. The graft is then passed underneath the LCL (Figure 7), through the proximal tunnel that has been created on the lateral femoral condyle, and then back down through the LCL and back onto itself after exiting the tibial tunnel (Figure 8). With the knee at 30° of flexion, the ITB graft is tensioned and sutured down to intact ITB fascia just proximal to the tibial tunnel orifice (Figure 9). We check knee range of motion (ROM) and then perform a Lachman test to assess changes in knee stability. The pivot shift examination is omitted to avoid placing excessive stress on the tenodesis. The tibial side of the patellar tendon allograft is then tensioned and secured over an AO 4.5-mm bicortical screw with washer with the knee in full extension. The screw is then tightened at 30° of knee flexion.
The ITB fascia is closed to the lateral femoral epicondyle with a running heavy suture, and all incisions are then irrigated and closed (Figures 10, 11). Standard sterile surgical dressing, Cryo/Cuff (Aircast, Vista, California), and brace are applied with the knee locked at 20°. Patients are generally discharged home the same day and followed up in clinic 1 week after surgery.
Complications
The peroneal nerve must be identified and protected during the open lateral procedure. In addition, the need for the extra lateral incision poses a slightly higher risk for infection compared with the traditional arthroscopic revision ACL procedure. Last, the additional tunnels required for the tenodesis can increase the theoretical potential for distal femur fracture and ACL graft fixation failure on the femoral side.
Postoperative Management
The operative knee is kept in extension in a brace locked at 20° for week 1 after surgery. Isometric quadriceps exercises are started immediately after surgery. Flexion to 90° is allowed starting week 2 after surgery, when the patient begins supervised active/passive flexion and progressive ROM exercises. In most cases, full ROM should be achieved by 6 to 8 weeks after surgery. Patients are progressed in their weight-bearing status by about 25% of their body weight per week, and use of crutches should be discontinued by week 4 after surgery. The brace should be discontinued by week 6 after surgery, when use of stationary bicycle and closed chain exercises begin. The patient may begin jogging when the operative leg regains 80% of contralateral quadriceps strength via Cybex strength testing. Functional drills begin in month 6, but patients should be counseled against returning to sport any earlier than 9 months after surgery.
Discussion
Achieving a successful outcome in revision ACL surgery (vs primary ACL surgery) is a significant challenge. Any of numerous factors can make the revision surgery more challenging, including existing poorly placed tunnels, tunnel expansion, lack of ideal graft choice, loss of secondary stabilizers, and deviations of the weight-bearing axis. Therefore, outcomes of revision surgery tend to be more moderate than outcomes of primary procedures.4,8-12
Revision ACL reconstruction techniques are varied and can involve use of autograft or allograft tissue as well as extra-articular augmentation techniques. Diamantopoulos and colleagues8 reported the outcomes of revision ACL reconstruction using bone–tendon–bone, hamstring, or quadriceps autografts in 107 patients. The majority of patients had improved outcome measures (mean Lysholm score improved from 51.5 to 88.5) and side-to-side laxity measurements. However, only 36.4% returned to preinjury activity level. Similarly, Noyes and Barber-Westin9 reported the outcomes of revision ACL reconstruction using quadriceps tendon–patellar bone autograft in 21 patients. Although there was significant improvement in terms of symptoms and activity level, 4 of the 21 knees were graded abnormal or severely abnormal on the IKDC (International Knee Documentation Committee) ligament rating. In a systematic review, pooled results of revision ACL reconstructions reiterated the above results.10 Eight hundred sixty-three patients from 21 studies were included in the analysis, which found significantly worse subjective outcomes than for primary procedures and a dramatically higher failure rate for the re-reconstructed ACL.
Several authors have directly compared primary cohorts with revision cohorts. Ahn and colleagues11 compared the outcomes of 59 revision ACL reconstructions with those of 117 primary reconstructions at a single institution. Although statistical comparison of stability between primary and revision ACL reconstructions showed no difference, revision reconstructions fared more poorly in terms of quality of life and return to activity compared with primary reconstructions. In a large cohort study of the Danish registry, revisions were found to have worse subjective outcomes than primary reconstructions as well.12 The study also found that the rerupture risk was significantly higher (relative risk, 2.05) when allograft was used.
Given the inferior results of revision surgery, our technique is recommended to augment the stability of reconstructed knees in the setting of revision ACL reconstruction. Adding the extra-articular procedure may augment the revised graft and protect it from excessive stress.13 A cadaver study compared double-bundle ACL reconstruction with single-bundle hamstring reconstruction plus extra-articular lateral tenodesis and found improved internal rotation control at 30° of flexion in the latter.14 Using contralateral 4-strand hamstring autograft in combination with an extra-articular lateral augment can have encouraging outcomes. Ferretti and colleagues15 reported an average Lysholm score of 95 in 12 patients who underwent this revision procedure and good anterior-to-posterior stability in 11 of the 12 patients. Trojani and colleagues16 reported on a cohort of 163 patients who underwent ACL revision surgery over a 10-year period. The authors found that 80% of patients with a lateral extra-articular tenodesis performed to augment their revision reconstruction had a negative pivot shift at long-term follow-up—versus only 63% of patients who underwent isolated revision ACL reconstruction. This finding was statistically significant, but the authors did not find any differences in IKDC scores between groups. These results support the initial biomechanical findings of Engebretsen and colleagues,17 who found that adding a lateral tenodesis decreased the forces on the reconstructed graft by 15%.
Conclusion
This technique allows for protection of the intra-articular allograft ligament reconstruction with improved rotational control that may potentially allow for improved subjective outcomes and protect against graft failure. Given the common pitfalls with stability in revision ACL surgery with allograft, this lateral extra-articular procedure can be an important structural augmentation in this challenging clinical issue in knee surgery.
1. Bach BR Jr. Revision anterior cruciate ligament surgery. Arthroscopy. 2003;19(suppl 1):14-29.
2. Baer GS, Harner CD. Clinical outcomes of allograft versus autograft in anterior cruciate ligament reconstruction. Clin Sports Med. 2007;26(4):661-681.
3. Spindler KP, Kuhn JE, Freedman KB, Matthews CE, Dittus RS, Harrell FE Jr. Anterior cruciate ligament reconstruction autograft choice: bone–tendon–bone versus hamstring: does it really matter? A systematic review. Am J Sports Med. 2004;32(8):1986-1995.
4. Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39(1):199-217.
5. Gianotti SM, Marshall SW, Hume PA, Bunt L. Incidence of anterior cruciate ligament injury and other knee ligament injuries: a national population-based study. J Sci Med Sport. 2009;12(6):622-627.
6. Jackson DW, Grood ES, Goldstein JD, et al. A comparison of patellar tendon autograft and allograft used for anterior cruciate ligament reconstruction in the goat model. Am J Sports Med. 1993;21(2):176-185.
7. Mascarenhas R, Tranovich M, Karpie JC, Irrgang JJ, Fu FH, Harner CD. Patellar tendon anterior cruciate ligament reconstruction in the high-demand patient: evaluation of autograft versus allograft reconstruction. Arthroscopy. 2010;26(9 Suppl):S58-S66.
8. Diamantopoulos AP, Lorbach O, Paessler HH. Anterior cruciate ligament revision reconstruction: results in 107 patients. Am J Sports Med. 2008;36(5):851-860.
9. Noyes FR, Barber-Westin SD. Anterior cruciate ligament revision reconstruction: results using a quadriceps tendon–patellar bone autograft. Am J Sports Med. 2006;34(4):553-564.
10. Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531-536.
11. Ahn JH, Lee YS, Ha HC. Comparison of revision surgery with primary anterior cruciate ligament reconstruction and outcome of revision surgery between different graft materials. Am J Sports Med. 2008;36(10):1889-1895.
12. Lind M, Menhert F, Pedersen AB. Incidence and outcome after revision anterior cruciate ligament reconstruction: results from the Danish registry for knee ligament reconstructions. Am J Sports Med. 2012;40(7):1551-1557.
13. Ferretti A, Conteduca F, Monaco E, De Carli A, D’Arrigo C. Revision anterior cruciate ligament reconstruction with doubled semitendinosus and gracilis tendons and lateral extra-articular reconstruction. J Bone Joint Surg Am. 2006;88(11):2373-2379.
14. Monaco E, Labianca L, Conteduca F, De Carli A, Ferretti A. Double bundle or single bundle plus extraarticular tenodesis in ACL reconstruction? A CAOS study. Knee Surg Sports Traumatol Arthrosc. 2007;15(10):1168-1174.
15. Ferretti A, Monaco E, Caperna L, Palma T, Conteduca F. Revision ACL reconstruction using contralateral hamstrings. Knee Surg Sports Traumatol Arthrosc. 2013;21(3):690-695.
16. Trojani C, Beaufils P, Burdin G, et al. Revision ACL reconstruction: influence of a lateral tenodesis. Knee Surg Sports Traumatol Arthrosc. 2012;20(8):1565-1570.
17. Engebretsen L, Lew WD, Lewis JL, Hunter RE. The effect of an iliotibial tenodesis on intraarticular graft forces and knee joint motion. Am J Sports Med. 1990;18(2):169-176.
1. Bach BR Jr. Revision anterior cruciate ligament surgery. Arthroscopy. 2003;19(suppl 1):14-29.
2. Baer GS, Harner CD. Clinical outcomes of allograft versus autograft in anterior cruciate ligament reconstruction. Clin Sports Med. 2007;26(4):661-681.
3. Spindler KP, Kuhn JE, Freedman KB, Matthews CE, Dittus RS, Harrell FE Jr. Anterior cruciate ligament reconstruction autograft choice: bone–tendon–bone versus hamstring: does it really matter? A systematic review. Am J Sports Med. 2004;32(8):1986-1995.
4. Kamath GV, Redfern JC, Greis PE, Burks RT. Revision anterior cruciate ligament reconstruction. Am J Sports Med. 2011;39(1):199-217.
5. Gianotti SM, Marshall SW, Hume PA, Bunt L. Incidence of anterior cruciate ligament injury and other knee ligament injuries: a national population-based study. J Sci Med Sport. 2009;12(6):622-627.
6. Jackson DW, Grood ES, Goldstein JD, et al. A comparison of patellar tendon autograft and allograft used for anterior cruciate ligament reconstruction in the goat model. Am J Sports Med. 1993;21(2):176-185.
7. Mascarenhas R, Tranovich M, Karpie JC, Irrgang JJ, Fu FH, Harner CD. Patellar tendon anterior cruciate ligament reconstruction in the high-demand patient: evaluation of autograft versus allograft reconstruction. Arthroscopy. 2010;26(9 Suppl):S58-S66.
8. Diamantopoulos AP, Lorbach O, Paessler HH. Anterior cruciate ligament revision reconstruction: results in 107 patients. Am J Sports Med. 2008;36(5):851-860.
9. Noyes FR, Barber-Westin SD. Anterior cruciate ligament revision reconstruction: results using a quadriceps tendon–patellar bone autograft. Am J Sports Med. 2006;34(4):553-564.
10. Wright RW, Gill CS, Chen L, et al. Outcome of revision anterior cruciate ligament reconstruction: a systematic review. J Bone Joint Surg Am. 2012;94(6):531-536.
11. Ahn JH, Lee YS, Ha HC. Comparison of revision surgery with primary anterior cruciate ligament reconstruction and outcome of revision surgery between different graft materials. Am J Sports Med. 2008;36(10):1889-1895.
12. Lind M, Menhert F, Pedersen AB. Incidence and outcome after revision anterior cruciate ligament reconstruction: results from the Danish registry for knee ligament reconstructions. Am J Sports Med. 2012;40(7):1551-1557.
13. Ferretti A, Conteduca F, Monaco E, De Carli A, D’Arrigo C. Revision anterior cruciate ligament reconstruction with doubled semitendinosus and gracilis tendons and lateral extra-articular reconstruction. J Bone Joint Surg Am. 2006;88(11):2373-2379.
14. Monaco E, Labianca L, Conteduca F, De Carli A, Ferretti A. Double bundle or single bundle plus extraarticular tenodesis in ACL reconstruction? A CAOS study. Knee Surg Sports Traumatol Arthrosc. 2007;15(10):1168-1174.
15. Ferretti A, Monaco E, Caperna L, Palma T, Conteduca F. Revision ACL reconstruction using contralateral hamstrings. Knee Surg Sports Traumatol Arthrosc. 2013;21(3):690-695.
16. Trojani C, Beaufils P, Burdin G, et al. Revision ACL reconstruction: influence of a lateral tenodesis. Knee Surg Sports Traumatol Arthrosc. 2012;20(8):1565-1570.
17. Engebretsen L, Lew WD, Lewis JL, Hunter RE. The effect of an iliotibial tenodesis on intraarticular graft forces and knee joint motion. Am J Sports Med. 1990;18(2):169-176.
More Than One-Third of Division I College Athletes May Have Low Vitamin D Levels
LAS VEGAS—A new study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found that more than one-third of elite, Division I college athletes may have low levels of vitamin D, which aids the absorption of calcium. Male, black, and Hispanic athletes are at greatest risk, researchers reported.
“Although multiple studies have demonstrated a high prevalence of vitamin D insufficiency across various populations, there is a paucity of data regarding elite level athletes,” said orthopedic surgeon and lead study author Diego Villacis, MD, Administrative Chief Resident at the University of Southern California. “Recent studies also have demonstrated that vitamin D levels have a direct relationship with muscle power, force, velocity, and optimal bone mass.”
In this study, which appeared in the February 2014 online issue of Sports Health, researchers measured the serum 25-hydroxyvitamin D (serum 25) levels of 223 athletes (121 men and 102 women) between June 2012 and August 2012. The mean serum 25 level for the athletes, enrolled in a broad range of indoor and outdoor sports, was 40.1 ±14.9 ng/mL (≥32 ng/mL is considered normal; 20 to <32 ng/mL, insufficient; and <20 ng/mL, deficient). Overall, 66.4% of participants had sufficient vitamin D levels and 33.6% had insufficient or deficient levels.
Men were 2.8 times more likely to have an abnormal vitamin D level, according to the results, and athletes with darker skin tones also faced a “much higher risk” for insufficient vitamin D. Black athletes were 19.1 times more likely to have abnormal vitamin D levels compared to white athletes, and Hispanics, 6.1 times more likely.
“Our study demonstrated abnormal vitamin D levels in nearly one out of three elite NCAA Division I athletes tested,” said Dr. Villacis. “Although there is much more work to be done, our results open the possibility for improved performance and most importantly decreased risk of injury with correction of vitamin D levels. This may potentially be achieved simply and safely through modification of diet, sunlight exposure, and vitamin D supplementation.”
LAS VEGAS—A new study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found that more than one-third of elite, Division I college athletes may have low levels of vitamin D, which aids the absorption of calcium. Male, black, and Hispanic athletes are at greatest risk, researchers reported.
“Although multiple studies have demonstrated a high prevalence of vitamin D insufficiency across various populations, there is a paucity of data regarding elite level athletes,” said orthopedic surgeon and lead study author Diego Villacis, MD, Administrative Chief Resident at the University of Southern California. “Recent studies also have demonstrated that vitamin D levels have a direct relationship with muscle power, force, velocity, and optimal bone mass.”
In this study, which appeared in the February 2014 online issue of Sports Health, researchers measured the serum 25-hydroxyvitamin D (serum 25) levels of 223 athletes (121 men and 102 women) between June 2012 and August 2012. The mean serum 25 level for the athletes, enrolled in a broad range of indoor and outdoor sports, was 40.1 ±14.9 ng/mL (≥32 ng/mL is considered normal; 20 to <32 ng/mL, insufficient; and <20 ng/mL, deficient). Overall, 66.4% of participants had sufficient vitamin D levels and 33.6% had insufficient or deficient levels.
Men were 2.8 times more likely to have an abnormal vitamin D level, according to the results, and athletes with darker skin tones also faced a “much higher risk” for insufficient vitamin D. Black athletes were 19.1 times more likely to have abnormal vitamin D levels compared to white athletes, and Hispanics, 6.1 times more likely.
“Our study demonstrated abnormal vitamin D levels in nearly one out of three elite NCAA Division I athletes tested,” said Dr. Villacis. “Although there is much more work to be done, our results open the possibility for improved performance and most importantly decreased risk of injury with correction of vitamin D levels. This may potentially be achieved simply and safely through modification of diet, sunlight exposure, and vitamin D supplementation.”
LAS VEGAS—A new study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found that more than one-third of elite, Division I college athletes may have low levels of vitamin D, which aids the absorption of calcium. Male, black, and Hispanic athletes are at greatest risk, researchers reported.
“Although multiple studies have demonstrated a high prevalence of vitamin D insufficiency across various populations, there is a paucity of data regarding elite level athletes,” said orthopedic surgeon and lead study author Diego Villacis, MD, Administrative Chief Resident at the University of Southern California. “Recent studies also have demonstrated that vitamin D levels have a direct relationship with muscle power, force, velocity, and optimal bone mass.”
In this study, which appeared in the February 2014 online issue of Sports Health, researchers measured the serum 25-hydroxyvitamin D (serum 25) levels of 223 athletes (121 men and 102 women) between June 2012 and August 2012. The mean serum 25 level for the athletes, enrolled in a broad range of indoor and outdoor sports, was 40.1 ±14.9 ng/mL (≥32 ng/mL is considered normal; 20 to <32 ng/mL, insufficient; and <20 ng/mL, deficient). Overall, 66.4% of participants had sufficient vitamin D levels and 33.6% had insufficient or deficient levels.
Men were 2.8 times more likely to have an abnormal vitamin D level, according to the results, and athletes with darker skin tones also faced a “much higher risk” for insufficient vitamin D. Black athletes were 19.1 times more likely to have abnormal vitamin D levels compared to white athletes, and Hispanics, 6.1 times more likely.
“Our study demonstrated abnormal vitamin D levels in nearly one out of three elite NCAA Division I athletes tested,” said Dr. Villacis. “Although there is much more work to be done, our results open the possibility for improved performance and most importantly decreased risk of injury with correction of vitamin D levels. This may potentially be achieved simply and safely through modification of diet, sunlight exposure, and vitamin D supplementation.”
Women Fare Better Than Men Following Total Knee, Hip Replacement
LAS VEGAS—While women may have their first total joint replacement (TJR) at an older age, they are less likely to have complications related to their surgery or require revision surgery, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). The findings contradict the theory that TJR is underutilized in female patients because they have worse outcomes than men.
Researchers reviewed patient databases from an Ontario hospital for first-time primary total hip replacement (THR) and total knee replacement (TKR) patients between 2002 and 2009. There were 37,881 THR surgeries (53.8% female) and 59,564 TKR surgeries (60.5% female). Women who underwent THR were significantly older than males (70 years vs. 65 years); however, there was no difference in age between male and female patients undergoing TKR (median age 68 years for both). A greater proportion of female patients undergoing TJR were defined as frail (6.6% vs. 3.5% for THR; and, 6.7% vs. 4% for TKR).
Following surgery, men were:
• 15% more likely to return to the emergency department within 30 days of hospital discharge following either THR or TKR.
• 60% and 70% more likely to have an acute myocardial infarction within 3 months following THR and TKR, respectively.
• 50% more likely to require a revision arthroplasty within 2 years of TKR.
• 25% more likely to be readmitted to the hospital and 70% more likely to experience an infection or revision surgery within 2 years of TKR, compared to women.
“Despite the fact that women have a higher prevalence of advanced hip and knee arthritis, prior research indicates that North American women with arthritis are less likely to receive joint replacement than men,” said lead study author Bheeshma Ravi, MD, PhD, an orthopedic surgery resident at the University of Toronto. “One possible explanation is that women are less often offered or accept surgery because their risk of serious complications following surgery is greater than that of men.
“In this study, we found that while overall rates of serious complications were low for both groups, they were lower for women than for men for both hip and knee replacement, particularly the latter” said Dr. Ravi. “Thus, the previously documented sex difference utilization of TJR cannot be explained by differential risks of complications following surgery.”
LAS VEGAS—While women may have their first total joint replacement (TJR) at an older age, they are less likely to have complications related to their surgery or require revision surgery, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). The findings contradict the theory that TJR is underutilized in female patients because they have worse outcomes than men.
Researchers reviewed patient databases from an Ontario hospital for first-time primary total hip replacement (THR) and total knee replacement (TKR) patients between 2002 and 2009. There were 37,881 THR surgeries (53.8% female) and 59,564 TKR surgeries (60.5% female). Women who underwent THR were significantly older than males (70 years vs. 65 years); however, there was no difference in age between male and female patients undergoing TKR (median age 68 years for both). A greater proportion of female patients undergoing TJR were defined as frail (6.6% vs. 3.5% for THR; and, 6.7% vs. 4% for TKR).
Following surgery, men were:
• 15% more likely to return to the emergency department within 30 days of hospital discharge following either THR or TKR.
• 60% and 70% more likely to have an acute myocardial infarction within 3 months following THR and TKR, respectively.
• 50% more likely to require a revision arthroplasty within 2 years of TKR.
• 25% more likely to be readmitted to the hospital and 70% more likely to experience an infection or revision surgery within 2 years of TKR, compared to women.
“Despite the fact that women have a higher prevalence of advanced hip and knee arthritis, prior research indicates that North American women with arthritis are less likely to receive joint replacement than men,” said lead study author Bheeshma Ravi, MD, PhD, an orthopedic surgery resident at the University of Toronto. “One possible explanation is that women are less often offered or accept surgery because their risk of serious complications following surgery is greater than that of men.
“In this study, we found that while overall rates of serious complications were low for both groups, they were lower for women than for men for both hip and knee replacement, particularly the latter” said Dr. Ravi. “Thus, the previously documented sex difference utilization of TJR cannot be explained by differential risks of complications following surgery.”
LAS VEGAS—While women may have their first total joint replacement (TJR) at an older age, they are less likely to have complications related to their surgery or require revision surgery, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). The findings contradict the theory that TJR is underutilized in female patients because they have worse outcomes than men.
Researchers reviewed patient databases from an Ontario hospital for first-time primary total hip replacement (THR) and total knee replacement (TKR) patients between 2002 and 2009. There were 37,881 THR surgeries (53.8% female) and 59,564 TKR surgeries (60.5% female). Women who underwent THR were significantly older than males (70 years vs. 65 years); however, there was no difference in age between male and female patients undergoing TKR (median age 68 years for both). A greater proportion of female patients undergoing TJR were defined as frail (6.6% vs. 3.5% for THR; and, 6.7% vs. 4% for TKR).
Following surgery, men were:
• 15% more likely to return to the emergency department within 30 days of hospital discharge following either THR or TKR.
• 60% and 70% more likely to have an acute myocardial infarction within 3 months following THR and TKR, respectively.
• 50% more likely to require a revision arthroplasty within 2 years of TKR.
• 25% more likely to be readmitted to the hospital and 70% more likely to experience an infection or revision surgery within 2 years of TKR, compared to women.
“Despite the fact that women have a higher prevalence of advanced hip and knee arthritis, prior research indicates that North American women with arthritis are less likely to receive joint replacement than men,” said lead study author Bheeshma Ravi, MD, PhD, an orthopedic surgery resident at the University of Toronto. “One possible explanation is that women are less often offered or accept surgery because their risk of serious complications following surgery is greater than that of men.
“In this study, we found that while overall rates of serious complications were low for both groups, they were lower for women than for men for both hip and knee replacement, particularly the latter” said Dr. Ravi. “Thus, the previously documented sex difference utilization of TJR cannot be explained by differential risks of complications following surgery.”
Black, Hispanic Patients More Likely to Be Readmitted to the Hospital Within 30 Days Following Hip or Knee Replacement Surgery
LAS VEGAS—Black and Hispanic patients were 62% and 50%, respectively, more likely to be readmitted to the hospital within 30 days after total joint replacement (TJR) surgery compared to white patients, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). In addition, Medicaid patients were 40% more likely to be readmitted to the hospital than patients with private insurance. Poorer outcomes, due in part to patient comorbidities, may reflect limited access to primary care, insufficient patient-doctor communication, researchers suggest.
Disparities in the provision of health care services have long been documented, including that black patients utilize hip and total knee replacement at rates nearly 40% less than white patients, despite having comparable or higher rates of osteoarthritis.
In this study, researchers analyzed 5 years of data—demographic (including race/ethnicity), clinical, and billing—on nearly 53,000 patients admitted to Connecticut hospitals for TJR from 2008 to 2012. The average patient age was 67 years, and the vast majority of patients were white (87%), covered by Medicare (56.7%), and female (61%).
The overall 30-day readmission rate for patients was 5.2%. The most common reasons for readmission were postoperative infection (8%), infection and inflammatory reaction due to internal joint prosthesis (6%), hematoma complications during a procedure (3%), and dislocation of a prosthetic joint (3%). Among the other study findings:
• Readmission rates were 83.5 per thousand for black patients, 78.9 for Hispanic patients, and 53.3 for white patients.
• Longer length of hospital stay was significantly associated with increased odds of readmission.
• When controlling for comorbidities and type of insurance coverage, the readmission rate for Hispanic patients dropped 44%, and for black patients, 38%. Black patients remained significantly more likely than white patients to be readmitted following surgery, after controlling for comorbidities.
• Patients covered by Medicare were 30% more likely to be readmitted within 30 days following discharge compared to patients covered by private insurance, and Medicaid patients were 40% more likely.
Recent research using national data on Medicare suggests that community-based factors, such as availability of general practitioners in the area, may be as or more important than hospital factors in determining readmission rates, and that patients may have few options other than hospital care for both urgent and non-urgent conditions related to their surgery or other conditions.
“Using an all-payer database, our study shows that black patients who undergo total knee replacement may have poorer outcomes,” said lead study author and orthopedic surgeon Courtland Lewis, MD. “After controlling for two key variables implicated in race and ethnic disparities in hospital readmission—preoperative comorbidities and type of insurance coverage—black patients still have a 35% higher likelihood of all-cause, 30-day readmission compared to white patients.
“Our ongoing research in this area is focused on other factors, such as the patient’s connection to primary care and patient-provider communication, that may explain this troubling finding,” said Dr. Lewis.
LAS VEGAS—Black and Hispanic patients were 62% and 50%, respectively, more likely to be readmitted to the hospital within 30 days after total joint replacement (TJR) surgery compared to white patients, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). In addition, Medicaid patients were 40% more likely to be readmitted to the hospital than patients with private insurance. Poorer outcomes, due in part to patient comorbidities, may reflect limited access to primary care, insufficient patient-doctor communication, researchers suggest.
Disparities in the provision of health care services have long been documented, including that black patients utilize hip and total knee replacement at rates nearly 40% less than white patients, despite having comparable or higher rates of osteoarthritis.
In this study, researchers analyzed 5 years of data—demographic (including race/ethnicity), clinical, and billing—on nearly 53,000 patients admitted to Connecticut hospitals for TJR from 2008 to 2012. The average patient age was 67 years, and the vast majority of patients were white (87%), covered by Medicare (56.7%), and female (61%).
The overall 30-day readmission rate for patients was 5.2%. The most common reasons for readmission were postoperative infection (8%), infection and inflammatory reaction due to internal joint prosthesis (6%), hematoma complications during a procedure (3%), and dislocation of a prosthetic joint (3%). Among the other study findings:
• Readmission rates were 83.5 per thousand for black patients, 78.9 for Hispanic patients, and 53.3 for white patients.
• Longer length of hospital stay was significantly associated with increased odds of readmission.
• When controlling for comorbidities and type of insurance coverage, the readmission rate for Hispanic patients dropped 44%, and for black patients, 38%. Black patients remained significantly more likely than white patients to be readmitted following surgery, after controlling for comorbidities.
• Patients covered by Medicare were 30% more likely to be readmitted within 30 days following discharge compared to patients covered by private insurance, and Medicaid patients were 40% more likely.
Recent research using national data on Medicare suggests that community-based factors, such as availability of general practitioners in the area, may be as or more important than hospital factors in determining readmission rates, and that patients may have few options other than hospital care for both urgent and non-urgent conditions related to their surgery or other conditions.
“Using an all-payer database, our study shows that black patients who undergo total knee replacement may have poorer outcomes,” said lead study author and orthopedic surgeon Courtland Lewis, MD. “After controlling for two key variables implicated in race and ethnic disparities in hospital readmission—preoperative comorbidities and type of insurance coverage—black patients still have a 35% higher likelihood of all-cause, 30-day readmission compared to white patients.
“Our ongoing research in this area is focused on other factors, such as the patient’s connection to primary care and patient-provider communication, that may explain this troubling finding,” said Dr. Lewis.
LAS VEGAS—Black and Hispanic patients were 62% and 50%, respectively, more likely to be readmitted to the hospital within 30 days after total joint replacement (TJR) surgery compared to white patients, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). In addition, Medicaid patients were 40% more likely to be readmitted to the hospital than patients with private insurance. Poorer outcomes, due in part to patient comorbidities, may reflect limited access to primary care, insufficient patient-doctor communication, researchers suggest.
Disparities in the provision of health care services have long been documented, including that black patients utilize hip and total knee replacement at rates nearly 40% less than white patients, despite having comparable or higher rates of osteoarthritis.
In this study, researchers analyzed 5 years of data—demographic (including race/ethnicity), clinical, and billing—on nearly 53,000 patients admitted to Connecticut hospitals for TJR from 2008 to 2012. The average patient age was 67 years, and the vast majority of patients were white (87%), covered by Medicare (56.7%), and female (61%).
The overall 30-day readmission rate for patients was 5.2%. The most common reasons for readmission were postoperative infection (8%), infection and inflammatory reaction due to internal joint prosthesis (6%), hematoma complications during a procedure (3%), and dislocation of a prosthetic joint (3%). Among the other study findings:
• Readmission rates were 83.5 per thousand for black patients, 78.9 for Hispanic patients, and 53.3 for white patients.
• Longer length of hospital stay was significantly associated with increased odds of readmission.
• When controlling for comorbidities and type of insurance coverage, the readmission rate for Hispanic patients dropped 44%, and for black patients, 38%. Black patients remained significantly more likely than white patients to be readmitted following surgery, after controlling for comorbidities.
• Patients covered by Medicare were 30% more likely to be readmitted within 30 days following discharge compared to patients covered by private insurance, and Medicaid patients were 40% more likely.
Recent research using national data on Medicare suggests that community-based factors, such as availability of general practitioners in the area, may be as or more important than hospital factors in determining readmission rates, and that patients may have few options other than hospital care for both urgent and non-urgent conditions related to their surgery or other conditions.
“Using an all-payer database, our study shows that black patients who undergo total knee replacement may have poorer outcomes,” said lead study author and orthopedic surgeon Courtland Lewis, MD. “After controlling for two key variables implicated in race and ethnic disparities in hospital readmission—preoperative comorbidities and type of insurance coverage—black patients still have a 35% higher likelihood of all-cause, 30-day readmission compared to white patients.
“Our ongoing research in this area is focused on other factors, such as the patient’s connection to primary care and patient-provider communication, that may explain this troubling finding,” said Dr. Lewis.
Hip Replacements in Middle-Age Nearly Double From 2002-2011, Outpacing Growth in Elderly Population
LAS VEGAS—The number of total hip replacements (THRs) nearly doubled among middle-age patients between 2002 and 2011, primarily due to the expansion of the middle-age population in the United States, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). Continued growth in utilization of hip replacement surgery in patients ages 45 to 64 years, an increase in revision surgeries for this population as they age, and a nearly 30% decline in the number of surgeons who perform THR could have significant implications for future health care costs, THR demand, and access, researchers said.
The researchers used the Nationwide Inpatient Sample (NIS) to identify primary THRs performed between 2002 and 2011 in patients ages 45 to 64 years, as well as related hospital charges. Population data and projections were obtained from the US Census Bureau and surgeon workforce estimates from the AAOS.
In 2011, 42.3% of THRs were performed in patients ages 45 to 64 years compared to 33.9% in 2002. Utilization of THR in this age group increased 89.2% from 2002 to 2011, from approximately 68,000 THRs in 2002 to 128,000 THRs in 2011. The overall population increased 21.3%. In addition, the authors found that:
• Growth of THR utilization in the 45- to 64-year-old age group grew 2.4 times faster than it did in the Medicare-aged population (age > 65 years).
• A rise in the prevalence of obesity, a known risk factor for hip osteoarthritis, among middle-age Americans was not significantly associated with increased THR utilization.
• Mean hospital charges in the THR 45- to 64-year-old age group declined 5.7% from 2002 to 2011, and declined 2.5% in the Medicare population (age > 65 years).
• Mean physician reimbursement per THR, in 2011 US dollars, declined 26.2% over the same period.
• Concurrently, the number of physicians reporting that they performed THR surgeries declined 28.2%.
“The purpose of this study was to identify potential drivers of THR utilization in the middle-age patient segment,” said lead study author Alexander S. McLawhorn, MD, MBA, an orthopedic surgery resident at the Hospital for Special Surgery in New York City. “Our multivariable statistical model suggested that the observed growth was best explained by an expansion of the middle-age population in the US. This particular age group is projected to continue expanding, and as such the demand for THR in this active group of patients will likely continue to rise as well. Our results underscore concerns about consumption of premium-priced implants in younger patients and the future revision burden this trend implies in the face of a dwindling number of physicians who specialize in hip arthroplasty surgery.”
LAS VEGAS—The number of total hip replacements (THRs) nearly doubled among middle-age patients between 2002 and 2011, primarily due to the expansion of the middle-age population in the United States, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). Continued growth in utilization of hip replacement surgery in patients ages 45 to 64 years, an increase in revision surgeries for this population as they age, and a nearly 30% decline in the number of surgeons who perform THR could have significant implications for future health care costs, THR demand, and access, researchers said.
The researchers used the Nationwide Inpatient Sample (NIS) to identify primary THRs performed between 2002 and 2011 in patients ages 45 to 64 years, as well as related hospital charges. Population data and projections were obtained from the US Census Bureau and surgeon workforce estimates from the AAOS.
In 2011, 42.3% of THRs were performed in patients ages 45 to 64 years compared to 33.9% in 2002. Utilization of THR in this age group increased 89.2% from 2002 to 2011, from approximately 68,000 THRs in 2002 to 128,000 THRs in 2011. The overall population increased 21.3%. In addition, the authors found that:
• Growth of THR utilization in the 45- to 64-year-old age group grew 2.4 times faster than it did in the Medicare-aged population (age > 65 years).
• A rise in the prevalence of obesity, a known risk factor for hip osteoarthritis, among middle-age Americans was not significantly associated with increased THR utilization.
• Mean hospital charges in the THR 45- to 64-year-old age group declined 5.7% from 2002 to 2011, and declined 2.5% in the Medicare population (age > 65 years).
• Mean physician reimbursement per THR, in 2011 US dollars, declined 26.2% over the same period.
• Concurrently, the number of physicians reporting that they performed THR surgeries declined 28.2%.
“The purpose of this study was to identify potential drivers of THR utilization in the middle-age patient segment,” said lead study author Alexander S. McLawhorn, MD, MBA, an orthopedic surgery resident at the Hospital for Special Surgery in New York City. “Our multivariable statistical model suggested that the observed growth was best explained by an expansion of the middle-age population in the US. This particular age group is projected to continue expanding, and as such the demand for THR in this active group of patients will likely continue to rise as well. Our results underscore concerns about consumption of premium-priced implants in younger patients and the future revision burden this trend implies in the face of a dwindling number of physicians who specialize in hip arthroplasty surgery.”
LAS VEGAS—The number of total hip replacements (THRs) nearly doubled among middle-age patients between 2002 and 2011, primarily due to the expansion of the middle-age population in the United States, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). Continued growth in utilization of hip replacement surgery in patients ages 45 to 64 years, an increase in revision surgeries for this population as they age, and a nearly 30% decline in the number of surgeons who perform THR could have significant implications for future health care costs, THR demand, and access, researchers said.
The researchers used the Nationwide Inpatient Sample (NIS) to identify primary THRs performed between 2002 and 2011 in patients ages 45 to 64 years, as well as related hospital charges. Population data and projections were obtained from the US Census Bureau and surgeon workforce estimates from the AAOS.
In 2011, 42.3% of THRs were performed in patients ages 45 to 64 years compared to 33.9% in 2002. Utilization of THR in this age group increased 89.2% from 2002 to 2011, from approximately 68,000 THRs in 2002 to 128,000 THRs in 2011. The overall population increased 21.3%. In addition, the authors found that:
• Growth of THR utilization in the 45- to 64-year-old age group grew 2.4 times faster than it did in the Medicare-aged population (age > 65 years).
• A rise in the prevalence of obesity, a known risk factor for hip osteoarthritis, among middle-age Americans was not significantly associated with increased THR utilization.
• Mean hospital charges in the THR 45- to 64-year-old age group declined 5.7% from 2002 to 2011, and declined 2.5% in the Medicare population (age > 65 years).
• Mean physician reimbursement per THR, in 2011 US dollars, declined 26.2% over the same period.
• Concurrently, the number of physicians reporting that they performed THR surgeries declined 28.2%.
“The purpose of this study was to identify potential drivers of THR utilization in the middle-age patient segment,” said lead study author Alexander S. McLawhorn, MD, MBA, an orthopedic surgery resident at the Hospital for Special Surgery in New York City. “Our multivariable statistical model suggested that the observed growth was best explained by an expansion of the middle-age population in the US. This particular age group is projected to continue expanding, and as such the demand for THR in this active group of patients will likely continue to rise as well. Our results underscore concerns about consumption of premium-priced implants in younger patients and the future revision burden this trend implies in the face of a dwindling number of physicians who specialize in hip arthroplasty surgery.”
Study Identifies Low Back Pain Risk Factors
LAS VEGAS—Nicotine dependence, obesity, alcohol abuse, and depressive disorders are risk factors for low back pain, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). Monitoring and counseling at-risk patients may prevent and minimize pain and improve quality of life, researchers said.
According to the U.S. Centers for Disease Control and Prevention’s (CDC) 2012 National Health Survey, nearly one-third of American adults reported that they had experienced low back pain during the previous 3 months. Determining modifiable risk factors for low back pain could help avoid or diminish the financial and emotional costs of this condition.
Researchers reviewed electronic records of more than 26 million patients from 13 health care systems across the United States, including 1.2 million patients diagnosed with low back pain (approximately 4.54% of the patient records).
The review found that 19.3% of the patients diagnosed with a depressive disorder reported lower back pain, as did 16.75% of patients diagnosed as obese (BMI > 30kg/m²), 16.53% of the patients diagnosed with nicotine dependence, and 14.66% with reported alcohol abuse. Patients with nicotine dependence, obesity, depressive disorders, and alcohol abuse had statistically significant relative risks of 4.489, 6.007, 5.511, and 3.326 for low back pain, respectively, when compared to other patients.
“This study used an electronic health care database to identify modifiable risk factors—obesity, depressive disorders, alcohol and tobacco use—in patients with low back pain,” said lead study author and orthopedic surgeon Scott Shemory, MD. “The findings will allow physicians to better counsel and more closely follow their high-risk patients.”
LAS VEGAS—Nicotine dependence, obesity, alcohol abuse, and depressive disorders are risk factors for low back pain, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). Monitoring and counseling at-risk patients may prevent and minimize pain and improve quality of life, researchers said.
According to the U.S. Centers for Disease Control and Prevention’s (CDC) 2012 National Health Survey, nearly one-third of American adults reported that they had experienced low back pain during the previous 3 months. Determining modifiable risk factors for low back pain could help avoid or diminish the financial and emotional costs of this condition.
Researchers reviewed electronic records of more than 26 million patients from 13 health care systems across the United States, including 1.2 million patients diagnosed with low back pain (approximately 4.54% of the patient records).
The review found that 19.3% of the patients diagnosed with a depressive disorder reported lower back pain, as did 16.75% of patients diagnosed as obese (BMI > 30kg/m²), 16.53% of the patients diagnosed with nicotine dependence, and 14.66% with reported alcohol abuse. Patients with nicotine dependence, obesity, depressive disorders, and alcohol abuse had statistically significant relative risks of 4.489, 6.007, 5.511, and 3.326 for low back pain, respectively, when compared to other patients.
“This study used an electronic health care database to identify modifiable risk factors—obesity, depressive disorders, alcohol and tobacco use—in patients with low back pain,” said lead study author and orthopedic surgeon Scott Shemory, MD. “The findings will allow physicians to better counsel and more closely follow their high-risk patients.”
LAS VEGAS—Nicotine dependence, obesity, alcohol abuse, and depressive disorders are risk factors for low back pain, according to a study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS). Monitoring and counseling at-risk patients may prevent and minimize pain and improve quality of life, researchers said.
According to the U.S. Centers for Disease Control and Prevention’s (CDC) 2012 National Health Survey, nearly one-third of American adults reported that they had experienced low back pain during the previous 3 months. Determining modifiable risk factors for low back pain could help avoid or diminish the financial and emotional costs of this condition.
Researchers reviewed electronic records of more than 26 million patients from 13 health care systems across the United States, including 1.2 million patients diagnosed with low back pain (approximately 4.54% of the patient records).
The review found that 19.3% of the patients diagnosed with a depressive disorder reported lower back pain, as did 16.75% of patients diagnosed as obese (BMI > 30kg/m²), 16.53% of the patients diagnosed with nicotine dependence, and 14.66% with reported alcohol abuse. Patients with nicotine dependence, obesity, depressive disorders, and alcohol abuse had statistically significant relative risks of 4.489, 6.007, 5.511, and 3.326 for low back pain, respectively, when compared to other patients.
“This study used an electronic health care database to identify modifiable risk factors—obesity, depressive disorders, alcohol and tobacco use—in patients with low back pain,” said lead study author and orthopedic surgeon Scott Shemory, MD. “The findings will allow physicians to better counsel and more closely follow their high-risk patients.”
Majority of Parents, Caregivers of Youth Baseball Pitchers Unaware of Safe Pitching Practices Designed to Prevent Overuse Injuries
LAS VEGAS—A new study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found that 53% of the parents/caregivers of youth baseball pitchers are unaware of safe pitching practices designed to prevent overuse injuries—common tears or damage, most often to the elbow (ulnar collateral ligament) or shoulder—which can cause pain, lost play time and, if not treated appropriately, arthritis, deformity, and disability. Additionally, 64% of parents said their child had suffered upper extremity pain due to pitching.
Between 2% and 8% of youth pitchers will suffer an overuse injury from throwing too hard, too often, too young, and/or without appropriate rest once pain begins in the shoulder or elbow. One recent study found that approximately 38% of pitchers will miss at least 1 game because of arm pain, with 34% experiencing pain severe enough to warrant a doctor’s visit.
As the number of overuse injuries continues to rise in young baseball players, safe pitching guidelines—which focus on proper warm up exercises; maximum play time, and pitch counts; recommended rest periods; appropriate ages for learning various types of pitches; and not playing on multiple teams, year round or on consecutive days—are being integrated into play at many of the nation’s 200,000 youth baseball teams, ideally with a firm, cooperative commitment from coaches, parents/caregivers, and players.
“Ensuring the safety of all athletes through injury prevention is absolutely necessary, especially when it comes to youth competitors,” said University of Florida orthopedic surgeon and lead study author Andrew Waligora, MD.
In the study, researchers distributed and analyzed results from a 22-question survey completed by the parents of 60 youth baseball players. The content of the survey was based on recommendations provided by the USA Baseball and Medical Safety Advisory, Little League Baseball, and the American Sports Medicine Institute. Just over 60% of the respondents identified themselves as the child’s mother. Nearly 95% of the respondents were white, 41% were 41 to 50 years old, and 44% were the parent of a pitcher between the ages of 13 and 16 years.
Of the parents or caregivers surveyed, 53% said they were unaware of the existence of safe pitching guidelines, 54% stated that they did not actively participate in monitoring their child’s pitch count, and 20% were unaware of how many pitches their child threw in a typical game. Among the other findings:
• 49% of pitchers threw in more than one league at a time, and 25% pitched in a league more than 9 months out of the year.
• 16% of the parents or caregivers surveyed did not know what kind of pitches their children threw.
• 75% of parents or caregivers of 11- to 12-year-old players said their child threw “curveballs,” and 17%, “sliders.” Of those who were aware of their child throwing “breaking pitches”—curveballs, sliders, slurves, or screwballs (all more advanced pitches)—18% reported that they learned how to throw these pitches from a parent.
• 64% of parents or caregivers recalled their child having upper extremity pain as a direct result of pitching, 38% had to miss either a game or pitching because of discomfort, and 34% experienced pain concerning enough to be evaluated by a medical professional.
There were no significant correlations between the number of pitches typically thrown in a game, the child’s position when not pitching, number of months pitched during the year, who keeps track of pitch count, and arm pain.
“This research emphasizes the importance of caregiver participation in ensuring compliance with youth safe pitching practices,” said Dr. Waligora. “Despite the implementation and easy accessibility of safe pitching guidelines, a large portion of caregivers surveyed were unaware and/or noncompliant with these established recommendations. Given the results of this study, further measures need to be taken to improve both education and compliance. Injury prevention should be a multidisciplinary approach that includes informing coaches, parents, and youth pitchers about safe pitching practices. Future directions may include establishing outreach programs for the youth baseball community.”
More information on youth baseball safety is available at STOP Sports Injuries and the American Orthopaedic Society for Sports Medicine (AOSSM).
LAS VEGAS—A new study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found that 53% of the parents/caregivers of youth baseball pitchers are unaware of safe pitching practices designed to prevent overuse injuries—common tears or damage, most often to the elbow (ulnar collateral ligament) or shoulder—which can cause pain, lost play time and, if not treated appropriately, arthritis, deformity, and disability. Additionally, 64% of parents said their child had suffered upper extremity pain due to pitching.
Between 2% and 8% of youth pitchers will suffer an overuse injury from throwing too hard, too often, too young, and/or without appropriate rest once pain begins in the shoulder or elbow. One recent study found that approximately 38% of pitchers will miss at least 1 game because of arm pain, with 34% experiencing pain severe enough to warrant a doctor’s visit.
As the number of overuse injuries continues to rise in young baseball players, safe pitching guidelines—which focus on proper warm up exercises; maximum play time, and pitch counts; recommended rest periods; appropriate ages for learning various types of pitches; and not playing on multiple teams, year round or on consecutive days—are being integrated into play at many of the nation’s 200,000 youth baseball teams, ideally with a firm, cooperative commitment from coaches, parents/caregivers, and players.
“Ensuring the safety of all athletes through injury prevention is absolutely necessary, especially when it comes to youth competitors,” said University of Florida orthopedic surgeon and lead study author Andrew Waligora, MD.
In the study, researchers distributed and analyzed results from a 22-question survey completed by the parents of 60 youth baseball players. The content of the survey was based on recommendations provided by the USA Baseball and Medical Safety Advisory, Little League Baseball, and the American Sports Medicine Institute. Just over 60% of the respondents identified themselves as the child’s mother. Nearly 95% of the respondents were white, 41% were 41 to 50 years old, and 44% were the parent of a pitcher between the ages of 13 and 16 years.
Of the parents or caregivers surveyed, 53% said they were unaware of the existence of safe pitching guidelines, 54% stated that they did not actively participate in monitoring their child’s pitch count, and 20% were unaware of how many pitches their child threw in a typical game. Among the other findings:
• 49% of pitchers threw in more than one league at a time, and 25% pitched in a league more than 9 months out of the year.
• 16% of the parents or caregivers surveyed did not know what kind of pitches their children threw.
• 75% of parents or caregivers of 11- to 12-year-old players said their child threw “curveballs,” and 17%, “sliders.” Of those who were aware of their child throwing “breaking pitches”—curveballs, sliders, slurves, or screwballs (all more advanced pitches)—18% reported that they learned how to throw these pitches from a parent.
• 64% of parents or caregivers recalled their child having upper extremity pain as a direct result of pitching, 38% had to miss either a game or pitching because of discomfort, and 34% experienced pain concerning enough to be evaluated by a medical professional.
There were no significant correlations between the number of pitches typically thrown in a game, the child’s position when not pitching, number of months pitched during the year, who keeps track of pitch count, and arm pain.
“This research emphasizes the importance of caregiver participation in ensuring compliance with youth safe pitching practices,” said Dr. Waligora. “Despite the implementation and easy accessibility of safe pitching guidelines, a large portion of caregivers surveyed were unaware and/or noncompliant with these established recommendations. Given the results of this study, further measures need to be taken to improve both education and compliance. Injury prevention should be a multidisciplinary approach that includes informing coaches, parents, and youth pitchers about safe pitching practices. Future directions may include establishing outreach programs for the youth baseball community.”
More information on youth baseball safety is available at STOP Sports Injuries and the American Orthopaedic Society for Sports Medicine (AOSSM).
LAS VEGAS—A new study presented at the 2015 Annual Meeting of the American Academy of Orthopaedic Surgeons (AAOS) found that 53% of the parents/caregivers of youth baseball pitchers are unaware of safe pitching practices designed to prevent overuse injuries—common tears or damage, most often to the elbow (ulnar collateral ligament) or shoulder—which can cause pain, lost play time and, if not treated appropriately, arthritis, deformity, and disability. Additionally, 64% of parents said their child had suffered upper extremity pain due to pitching.
Between 2% and 8% of youth pitchers will suffer an overuse injury from throwing too hard, too often, too young, and/or without appropriate rest once pain begins in the shoulder or elbow. One recent study found that approximately 38% of pitchers will miss at least 1 game because of arm pain, with 34% experiencing pain severe enough to warrant a doctor’s visit.
As the number of overuse injuries continues to rise in young baseball players, safe pitching guidelines—which focus on proper warm up exercises; maximum play time, and pitch counts; recommended rest periods; appropriate ages for learning various types of pitches; and not playing on multiple teams, year round or on consecutive days—are being integrated into play at many of the nation’s 200,000 youth baseball teams, ideally with a firm, cooperative commitment from coaches, parents/caregivers, and players.
“Ensuring the safety of all athletes through injury prevention is absolutely necessary, especially when it comes to youth competitors,” said University of Florida orthopedic surgeon and lead study author Andrew Waligora, MD.
In the study, researchers distributed and analyzed results from a 22-question survey completed by the parents of 60 youth baseball players. The content of the survey was based on recommendations provided by the USA Baseball and Medical Safety Advisory, Little League Baseball, and the American Sports Medicine Institute. Just over 60% of the respondents identified themselves as the child’s mother. Nearly 95% of the respondents were white, 41% were 41 to 50 years old, and 44% were the parent of a pitcher between the ages of 13 and 16 years.
Of the parents or caregivers surveyed, 53% said they were unaware of the existence of safe pitching guidelines, 54% stated that they did not actively participate in monitoring their child’s pitch count, and 20% were unaware of how many pitches their child threw in a typical game. Among the other findings:
• 49% of pitchers threw in more than one league at a time, and 25% pitched in a league more than 9 months out of the year.
• 16% of the parents or caregivers surveyed did not know what kind of pitches their children threw.
• 75% of parents or caregivers of 11- to 12-year-old players said their child threw “curveballs,” and 17%, “sliders.” Of those who were aware of their child throwing “breaking pitches”—curveballs, sliders, slurves, or screwballs (all more advanced pitches)—18% reported that they learned how to throw these pitches from a parent.
• 64% of parents or caregivers recalled their child having upper extremity pain as a direct result of pitching, 38% had to miss either a game or pitching because of discomfort, and 34% experienced pain concerning enough to be evaluated by a medical professional.
There were no significant correlations between the number of pitches typically thrown in a game, the child’s position when not pitching, number of months pitched during the year, who keeps track of pitch count, and arm pain.
“This research emphasizes the importance of caregiver participation in ensuring compliance with youth safe pitching practices,” said Dr. Waligora. “Despite the implementation and easy accessibility of safe pitching guidelines, a large portion of caregivers surveyed were unaware and/or noncompliant with these established recommendations. Given the results of this study, further measures need to be taken to improve both education and compliance. Injury prevention should be a multidisciplinary approach that includes informing coaches, parents, and youth pitchers about safe pitching practices. Future directions may include establishing outreach programs for the youth baseball community.”
More information on youth baseball safety is available at STOP Sports Injuries and the American Orthopaedic Society for Sports Medicine (AOSSM).