User login
Survey of MS patients reveals numerous pregnancy-related concerns
DALLAS – When it comes to family planning and pregnancy-related decisions such as breastfeeding and medication management, patients with multiple sclerosis (MS) receive a wide variety of advice, guidance, and engagement from their health care providers, results from a single-center survey demonstrated.
“We want our patients to feel comfortable when they come to us in their 20s or 30s and they get diagnosed, they’re scared, and it’s all new to them,” one of the study authors, Casey E. Engel said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “We want them to know that family planning is something to consider and that they can proceed with having a family with our help and guidance.”
In an effort to collect patient-experience data around family planning, pregnancy, and breastfeeding post-MS diagnosis, Ms. Engel and senior author Myla D. Goldman, MD, mailed a survey to 1,000 women with confirmed MS diagnosis who had received care at the University of Virginia Medical Center in Charlottesville. The researchers reported findings from 173 respondents, of whom 70% were receiving specialty care for MS. Most of the survey participants (137) did not become pregnant following their diagnosis, while 36 did.
Of the 137 respondents who did not become pregnant following diagnosis, 22 (16%) indicated that their decision was driven by MS-related concerns, including MS worsening with pregnancy (64%), ability to care for child secondary to MS (46%), lack of knowledge about options for pregnancy and MS (18%), passing MS onto child (18%), and stopping disease-modifying therapy (DMT) to attempt pregnancy (9%).
Of the 36 women who had a pregnancy following diagnosis, 20% reported postpartum depression or anxiety, higher than the national average of 10%-15%. In addition, 79% reported not being on DMT at the time of conception, 9% were on either glatiramer acetate injection or interferon beta-1a at time of conception, and 3% were on fingolimod (Gilenya) at time of conception. The majority reported receiving inconsistent advice about when to discontinue DMT before attempting pregnancy (a range from 0 to 6 months).
“It’s also noteworthy that 20% took a year to achieve pregnancy,” said Dr. Goldman, a neurologist who directs the university’s MS clinic. “If these women stop [their DMT] 6 months in advance and they take a year to achieve pregnancy, that’s 18 months without therapeutic coverage. That’s a concern to bring to light.”
Breastfeeding was reported in 71% of mothers in postdiagnosis pregnancy with a range between 1 week and 10 months, driven in part by variable guidelines regarding DMT reinitiation. In the meantime, respondents who did not breastfeed made this decision due to fear of relapse, glucocorticoids, or desire to reinitiate medication.
“Though our study was limited by low survey response, we hope that our work may highlight the difficulty our patients face and foster discussions within the MS community around these issues to improve the individual patient experience,” the researchers wrote in their poster.
Ms. Engel worked on the study while an undergraduate at the University of Virginia. The study was supported by the ziMS Foundation.
SOURCE: Engel CE et al. ACTRIMS Forum 2019, Poster 307.
DALLAS – When it comes to family planning and pregnancy-related decisions such as breastfeeding and medication management, patients with multiple sclerosis (MS) receive a wide variety of advice, guidance, and engagement from their health care providers, results from a single-center survey demonstrated.
“We want our patients to feel comfortable when they come to us in their 20s or 30s and they get diagnosed, they’re scared, and it’s all new to them,” one of the study authors, Casey E. Engel said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “We want them to know that family planning is something to consider and that they can proceed with having a family with our help and guidance.”
In an effort to collect patient-experience data around family planning, pregnancy, and breastfeeding post-MS diagnosis, Ms. Engel and senior author Myla D. Goldman, MD, mailed a survey to 1,000 women with confirmed MS diagnosis who had received care at the University of Virginia Medical Center in Charlottesville. The researchers reported findings from 173 respondents, of whom 70% were receiving specialty care for MS. Most of the survey participants (137) did not become pregnant following their diagnosis, while 36 did.
Of the 137 respondents who did not become pregnant following diagnosis, 22 (16%) indicated that their decision was driven by MS-related concerns, including MS worsening with pregnancy (64%), ability to care for child secondary to MS (46%), lack of knowledge about options for pregnancy and MS (18%), passing MS onto child (18%), and stopping disease-modifying therapy (DMT) to attempt pregnancy (9%).
Of the 36 women who had a pregnancy following diagnosis, 20% reported postpartum depression or anxiety, higher than the national average of 10%-15%. In addition, 79% reported not being on DMT at the time of conception, 9% were on either glatiramer acetate injection or interferon beta-1a at time of conception, and 3% were on fingolimod (Gilenya) at time of conception. The majority reported receiving inconsistent advice about when to discontinue DMT before attempting pregnancy (a range from 0 to 6 months).
“It’s also noteworthy that 20% took a year to achieve pregnancy,” said Dr. Goldman, a neurologist who directs the university’s MS clinic. “If these women stop [their DMT] 6 months in advance and they take a year to achieve pregnancy, that’s 18 months without therapeutic coverage. That’s a concern to bring to light.”
Breastfeeding was reported in 71% of mothers in postdiagnosis pregnancy with a range between 1 week and 10 months, driven in part by variable guidelines regarding DMT reinitiation. In the meantime, respondents who did not breastfeed made this decision due to fear of relapse, glucocorticoids, or desire to reinitiate medication.
“Though our study was limited by low survey response, we hope that our work may highlight the difficulty our patients face and foster discussions within the MS community around these issues to improve the individual patient experience,” the researchers wrote in their poster.
Ms. Engel worked on the study while an undergraduate at the University of Virginia. The study was supported by the ziMS Foundation.
SOURCE: Engel CE et al. ACTRIMS Forum 2019, Poster 307.
DALLAS – When it comes to family planning and pregnancy-related decisions such as breastfeeding and medication management, patients with multiple sclerosis (MS) receive a wide variety of advice, guidance, and engagement from their health care providers, results from a single-center survey demonstrated.
“We want our patients to feel comfortable when they come to us in their 20s or 30s and they get diagnosed, they’re scared, and it’s all new to them,” one of the study authors, Casey E. Engel said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “We want them to know that family planning is something to consider and that they can proceed with having a family with our help and guidance.”
In an effort to collect patient-experience data around family planning, pregnancy, and breastfeeding post-MS diagnosis, Ms. Engel and senior author Myla D. Goldman, MD, mailed a survey to 1,000 women with confirmed MS diagnosis who had received care at the University of Virginia Medical Center in Charlottesville. The researchers reported findings from 173 respondents, of whom 70% were receiving specialty care for MS. Most of the survey participants (137) did not become pregnant following their diagnosis, while 36 did.
Of the 137 respondents who did not become pregnant following diagnosis, 22 (16%) indicated that their decision was driven by MS-related concerns, including MS worsening with pregnancy (64%), ability to care for child secondary to MS (46%), lack of knowledge about options for pregnancy and MS (18%), passing MS onto child (18%), and stopping disease-modifying therapy (DMT) to attempt pregnancy (9%).
Of the 36 women who had a pregnancy following diagnosis, 20% reported postpartum depression or anxiety, higher than the national average of 10%-15%. In addition, 79% reported not being on DMT at the time of conception, 9% were on either glatiramer acetate injection or interferon beta-1a at time of conception, and 3% were on fingolimod (Gilenya) at time of conception. The majority reported receiving inconsistent advice about when to discontinue DMT before attempting pregnancy (a range from 0 to 6 months).
“It’s also noteworthy that 20% took a year to achieve pregnancy,” said Dr. Goldman, a neurologist who directs the university’s MS clinic. “If these women stop [their DMT] 6 months in advance and they take a year to achieve pregnancy, that’s 18 months without therapeutic coverage. That’s a concern to bring to light.”
Breastfeeding was reported in 71% of mothers in postdiagnosis pregnancy with a range between 1 week and 10 months, driven in part by variable guidelines regarding DMT reinitiation. In the meantime, respondents who did not breastfeed made this decision due to fear of relapse, glucocorticoids, or desire to reinitiate medication.
“Though our study was limited by low survey response, we hope that our work may highlight the difficulty our patients face and foster discussions within the MS community around these issues to improve the individual patient experience,” the researchers wrote in their poster.
Ms. Engel worked on the study while an undergraduate at the University of Virginia. The study was supported by the ziMS Foundation.
SOURCE: Engel CE et al. ACTRIMS Forum 2019, Poster 307.
REPORTING FROM ACTRIMS FORUM 2019
Teriflunomide transmission can occur in female partners of men taking the drug
DALLAS – Low or undetectable levels of teriflunomide (Aubagio) occur in women who are sexually active with men taking the drug for relapsing multiple sclerosis, results from a small study demonstrated.
“One of the issues with this particular drug is that it carries a strong pregnancy warning because in animal studies the drug has been teratogenic,” Joseph B. Guarnaccia, MD, said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “The other issue is that it remains detectable in the body for some time. The issue of females taking this drug and conception are well known. There are strong warnings that, if a woman wants to become pregnant, the drug should be removed quickly from the system. But if their male partner is on the drug, does that pose a risk to their female partner? That question has never been addressed in a human study.”
The Food and Drug Administration prescribing information recommends that men wishing to father a child should discontinue use of teriflunomide and undergo the accelerated elimination procedure. It also recommends that female partners wishing to become pregnant should discontinue the drug and undergo an accelerated elimination procedure to verify that the plasma teriflunomide concentration is less than 0.020 mcg/mL.
In an effort to test the risk of female exposure to potentially teratogenic levels of teriflunomide through sexual intercourse, Dr. Guarnaccia, a neurologist with the Multiple Sclerosis Treatment Center at Griffin Hospital in Derby, Conn., and his colleagues recruited 10 couples and compared serum levels of teriflunomide in men with relapsing multiple sclerosis with those of their female partners. Enrollment criteria for men included a diagnosis of relapsing multiple sclerosis, age between 18 and 55 years, treatment with teriflunomide for at least 2 months prior to study entry, and frequency of sexual intercourse with their female partners at least twice a month. Pregnancy was excluded in females, and couples could not use barrier or withdrawal methods of contraception. The couples completed a brief questionnaire and underwent a one-time blood draw for teriflunomide levels either at the investigator’s office or at a LabCorp facility.
The mean age of study participants was 47 years and the mean frequency of intercourse was seven episodes per month. The mean teriflunomide concentration in men was 42.30 mcg/mL (ranged from 10.07 to 142.84 mcg/mL). Six women had teriflunomide below detection levels (0.020 mcg/mL). However, four women had detectable levels that averaged 0.045 mcg/mL (ranging from 0.022 to 0.077 mcg/mL).
“This small study demonstrates that low or undetectable levels of teriflunomide occur in females who are sexually active with males taking teriflunomide for relapsing multiple sclerosis,” the researchers wrote in their poster. They found that women who had low detectable levels of teriflunomide, compared with women with undetectable levels, did not engage in more frequent sexual intercourse nor were their levels associated with higher levels of teriflunomide in their male partners.
“Indeed, one might have expected a positive correlation between serum levels of teriflunomide in females and the frequency or concentration of inoculation in semen from their partners,” the researchers wrote. “While semen levels of teriflunomide were not measured in this study, it might be assumed that serum and semen concentrations of small molecules like teriflunomide are similar.”
The study was supported by a investigator-sponsored research grant from Sanofi-Genzyme. Dr. Guarnaccia reported that he has received speaking honoraria and educational grants from Sanofi-Genzyme, Biogen, Teva, Acorda Therapeutics, Bayer, EMD Serono, and Genentech.
SOURCE: Guarnaccia JB et al. ACTRIMS Forum 2019, Poster 115.
DALLAS – Low or undetectable levels of teriflunomide (Aubagio) occur in women who are sexually active with men taking the drug for relapsing multiple sclerosis, results from a small study demonstrated.
“One of the issues with this particular drug is that it carries a strong pregnancy warning because in animal studies the drug has been teratogenic,” Joseph B. Guarnaccia, MD, said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “The other issue is that it remains detectable in the body for some time. The issue of females taking this drug and conception are well known. There are strong warnings that, if a woman wants to become pregnant, the drug should be removed quickly from the system. But if their male partner is on the drug, does that pose a risk to their female partner? That question has never been addressed in a human study.”
The Food and Drug Administration prescribing information recommends that men wishing to father a child should discontinue use of teriflunomide and undergo the accelerated elimination procedure. It also recommends that female partners wishing to become pregnant should discontinue the drug and undergo an accelerated elimination procedure to verify that the plasma teriflunomide concentration is less than 0.020 mcg/mL.
In an effort to test the risk of female exposure to potentially teratogenic levels of teriflunomide through sexual intercourse, Dr. Guarnaccia, a neurologist with the Multiple Sclerosis Treatment Center at Griffin Hospital in Derby, Conn., and his colleagues recruited 10 couples and compared serum levels of teriflunomide in men with relapsing multiple sclerosis with those of their female partners. Enrollment criteria for men included a diagnosis of relapsing multiple sclerosis, age between 18 and 55 years, treatment with teriflunomide for at least 2 months prior to study entry, and frequency of sexual intercourse with their female partners at least twice a month. Pregnancy was excluded in females, and couples could not use barrier or withdrawal methods of contraception. The couples completed a brief questionnaire and underwent a one-time blood draw for teriflunomide levels either at the investigator’s office or at a LabCorp facility.
The mean age of study participants was 47 years and the mean frequency of intercourse was seven episodes per month. The mean teriflunomide concentration in men was 42.30 mcg/mL (ranged from 10.07 to 142.84 mcg/mL). Six women had teriflunomide below detection levels (0.020 mcg/mL). However, four women had detectable levels that averaged 0.045 mcg/mL (ranging from 0.022 to 0.077 mcg/mL).
“This small study demonstrates that low or undetectable levels of teriflunomide occur in females who are sexually active with males taking teriflunomide for relapsing multiple sclerosis,” the researchers wrote in their poster. They found that women who had low detectable levels of teriflunomide, compared with women with undetectable levels, did not engage in more frequent sexual intercourse nor were their levels associated with higher levels of teriflunomide in their male partners.
“Indeed, one might have expected a positive correlation between serum levels of teriflunomide in females and the frequency or concentration of inoculation in semen from their partners,” the researchers wrote. “While semen levels of teriflunomide were not measured in this study, it might be assumed that serum and semen concentrations of small molecules like teriflunomide are similar.”
The study was supported by a investigator-sponsored research grant from Sanofi-Genzyme. Dr. Guarnaccia reported that he has received speaking honoraria and educational grants from Sanofi-Genzyme, Biogen, Teva, Acorda Therapeutics, Bayer, EMD Serono, and Genentech.
SOURCE: Guarnaccia JB et al. ACTRIMS Forum 2019, Poster 115.
DALLAS – Low or undetectable levels of teriflunomide (Aubagio) occur in women who are sexually active with men taking the drug for relapsing multiple sclerosis, results from a small study demonstrated.
“One of the issues with this particular drug is that it carries a strong pregnancy warning because in animal studies the drug has been teratogenic,” Joseph B. Guarnaccia, MD, said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “The other issue is that it remains detectable in the body for some time. The issue of females taking this drug and conception are well known. There are strong warnings that, if a woman wants to become pregnant, the drug should be removed quickly from the system. But if their male partner is on the drug, does that pose a risk to their female partner? That question has never been addressed in a human study.”
The Food and Drug Administration prescribing information recommends that men wishing to father a child should discontinue use of teriflunomide and undergo the accelerated elimination procedure. It also recommends that female partners wishing to become pregnant should discontinue the drug and undergo an accelerated elimination procedure to verify that the plasma teriflunomide concentration is less than 0.020 mcg/mL.
In an effort to test the risk of female exposure to potentially teratogenic levels of teriflunomide through sexual intercourse, Dr. Guarnaccia, a neurologist with the Multiple Sclerosis Treatment Center at Griffin Hospital in Derby, Conn., and his colleagues recruited 10 couples and compared serum levels of teriflunomide in men with relapsing multiple sclerosis with those of their female partners. Enrollment criteria for men included a diagnosis of relapsing multiple sclerosis, age between 18 and 55 years, treatment with teriflunomide for at least 2 months prior to study entry, and frequency of sexual intercourse with their female partners at least twice a month. Pregnancy was excluded in females, and couples could not use barrier or withdrawal methods of contraception. The couples completed a brief questionnaire and underwent a one-time blood draw for teriflunomide levels either at the investigator’s office or at a LabCorp facility.
The mean age of study participants was 47 years and the mean frequency of intercourse was seven episodes per month. The mean teriflunomide concentration in men was 42.30 mcg/mL (ranged from 10.07 to 142.84 mcg/mL). Six women had teriflunomide below detection levels (0.020 mcg/mL). However, four women had detectable levels that averaged 0.045 mcg/mL (ranging from 0.022 to 0.077 mcg/mL).
“This small study demonstrates that low or undetectable levels of teriflunomide occur in females who are sexually active with males taking teriflunomide for relapsing multiple sclerosis,” the researchers wrote in their poster. They found that women who had low detectable levels of teriflunomide, compared with women with undetectable levels, did not engage in more frequent sexual intercourse nor were their levels associated with higher levels of teriflunomide in their male partners.
“Indeed, one might have expected a positive correlation between serum levels of teriflunomide in females and the frequency or concentration of inoculation in semen from their partners,” the researchers wrote. “While semen levels of teriflunomide were not measured in this study, it might be assumed that serum and semen concentrations of small molecules like teriflunomide are similar.”
The study was supported by a investigator-sponsored research grant from Sanofi-Genzyme. Dr. Guarnaccia reported that he has received speaking honoraria and educational grants from Sanofi-Genzyme, Biogen, Teva, Acorda Therapeutics, Bayer, EMD Serono, and Genentech.
SOURCE: Guarnaccia JB et al. ACTRIMS Forum 2019, Poster 115.
REPORTING FROM ACTRIMS FORUM 2019
Neurologists grappling with patients who embrace ‘stem cell tourism’
DALLAS – Stem cell tourism – the unethical practice of offering unproven cellular preparations to patients for a variety of conditions – is increasingly sought by patients with incurable conditions such as multiple sclerosis and amyotrophic lateral sclerosis, results from a novel survey suggest.
In fact, most academic neurologists have been approached by patients with incurable conditions who ask them about stem cell therapy, while about two-thirds have had at least one patient who has undergone stem cell therapy.
“It’s really scary,” Wijdan Rai, MBBS, the study’s first author, said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “This is a more prevalent issue than we think, and the complication rates are higher than we think.”
According to the study’s senior author, Jaime Imitola, MD, who directs the Progressive Multiple Sclerosis Multidisciplinary Clinic and Translational Research Program at the Ohio State University Wexner Medical Center, Columbus, the results “call for the creation of a nationwide registry where neurologists can document adverse reactions to stem cell procedures and further support dedicated patient and neurologist education as we have proposed before” (See Semin Neurol. 2018; 38[2]:176-81 and JAMA Neurol. 2015;72[11]:1342-5).
In an effort to understand the experiences and attitudes of academic neurologists regarding stem cell tourism and patient-reported complications, the researchers developed a 25-question survey disseminated via Synapse, a web tool from the American Academy of Neurology. Respondents were asked about demographic information, frequency of patient questioning, perception of physician competence, patient complications and experiences, and attitudes toward increased physician education.
Dr. Rai, who is a senior neurology resident at the medical center, presented findings from 204 neurologist respondents, of whom 31% identified themselves as MS specialists. Nearly all respondents (91%) said they have been approached by patients with incurable conditions seeking information about stem cells (37% of whom had diagnosis of MS). In addition, 65% have had at least one patient that has undergone “stem cell therapy,” and 73% said it would be “helpful” or “very helpful” to have an evidence-based patient education tool on the topic. “Patients most often wanted general information,” Dr. Rai said. “However, 50% requested permission to undergo a stem cell procedure, and 31% approached their neurologist after the procedure.”
Survey respondents reported that 33% of the stem cell interventions were performed in the United States and 22% abroad, while 37% reported both in the U.S. and abroad. Patients underwent the procedures in China, Germany, the Bahamas, Mexico, Russia, and Costa Rica. Three-quarters of respondents (75%) indicated no patient experiencing complications from the stem cell interventions. However, 25% reported patients experiencing a variety of complications from the procedures, including strokes, meningoencephalitis, quadriparesis, MS deterioration, sepsis, hepatitis C, seizures, meningitis from intrathecal cell injections, infections, and spinal cord tumors. “At least three respondents had a patient who died as a direct complication from stem cell therapy,” Dr. Rai said.
In their poster, the researchers recommended a “multipronged approach to improve education of MS patients from exploitation and engaging multiple stakeholders in the field, including MS academic societies, licensing boards, and legislative bodies. Specifically, we call for creation of evidence-based education for both neurologists and patients, including physical resources that neurologists can use when discussing stem cell interventions with patients and videos on proper counseling during these visits.”
Colleagues from OSU’s Laboratory for Neural Stem Cells and Functional Neurogenetics contributed to this work. The researchers reported having no financial disclosures.
SOURCE: Rai W et al. ACTRIMS Forum 2019, Poster 237.
DALLAS – Stem cell tourism – the unethical practice of offering unproven cellular preparations to patients for a variety of conditions – is increasingly sought by patients with incurable conditions such as multiple sclerosis and amyotrophic lateral sclerosis, results from a novel survey suggest.
In fact, most academic neurologists have been approached by patients with incurable conditions who ask them about stem cell therapy, while about two-thirds have had at least one patient who has undergone stem cell therapy.
“It’s really scary,” Wijdan Rai, MBBS, the study’s first author, said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “This is a more prevalent issue than we think, and the complication rates are higher than we think.”
According to the study’s senior author, Jaime Imitola, MD, who directs the Progressive Multiple Sclerosis Multidisciplinary Clinic and Translational Research Program at the Ohio State University Wexner Medical Center, Columbus, the results “call for the creation of a nationwide registry where neurologists can document adverse reactions to stem cell procedures and further support dedicated patient and neurologist education as we have proposed before” (See Semin Neurol. 2018; 38[2]:176-81 and JAMA Neurol. 2015;72[11]:1342-5).
In an effort to understand the experiences and attitudes of academic neurologists regarding stem cell tourism and patient-reported complications, the researchers developed a 25-question survey disseminated via Synapse, a web tool from the American Academy of Neurology. Respondents were asked about demographic information, frequency of patient questioning, perception of physician competence, patient complications and experiences, and attitudes toward increased physician education.
Dr. Rai, who is a senior neurology resident at the medical center, presented findings from 204 neurologist respondents, of whom 31% identified themselves as MS specialists. Nearly all respondents (91%) said they have been approached by patients with incurable conditions seeking information about stem cells (37% of whom had diagnosis of MS). In addition, 65% have had at least one patient that has undergone “stem cell therapy,” and 73% said it would be “helpful” or “very helpful” to have an evidence-based patient education tool on the topic. “Patients most often wanted general information,” Dr. Rai said. “However, 50% requested permission to undergo a stem cell procedure, and 31% approached their neurologist after the procedure.”
Survey respondents reported that 33% of the stem cell interventions were performed in the United States and 22% abroad, while 37% reported both in the U.S. and abroad. Patients underwent the procedures in China, Germany, the Bahamas, Mexico, Russia, and Costa Rica. Three-quarters of respondents (75%) indicated no patient experiencing complications from the stem cell interventions. However, 25% reported patients experiencing a variety of complications from the procedures, including strokes, meningoencephalitis, quadriparesis, MS deterioration, sepsis, hepatitis C, seizures, meningitis from intrathecal cell injections, infections, and spinal cord tumors. “At least three respondents had a patient who died as a direct complication from stem cell therapy,” Dr. Rai said.
In their poster, the researchers recommended a “multipronged approach to improve education of MS patients from exploitation and engaging multiple stakeholders in the field, including MS academic societies, licensing boards, and legislative bodies. Specifically, we call for creation of evidence-based education for both neurologists and patients, including physical resources that neurologists can use when discussing stem cell interventions with patients and videos on proper counseling during these visits.”
Colleagues from OSU’s Laboratory for Neural Stem Cells and Functional Neurogenetics contributed to this work. The researchers reported having no financial disclosures.
SOURCE: Rai W et al. ACTRIMS Forum 2019, Poster 237.
DALLAS – Stem cell tourism – the unethical practice of offering unproven cellular preparations to patients for a variety of conditions – is increasingly sought by patients with incurable conditions such as multiple sclerosis and amyotrophic lateral sclerosis, results from a novel survey suggest.
In fact, most academic neurologists have been approached by patients with incurable conditions who ask them about stem cell therapy, while about two-thirds have had at least one patient who has undergone stem cell therapy.
“It’s really scary,” Wijdan Rai, MBBS, the study’s first author, said in an interview at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis. “This is a more prevalent issue than we think, and the complication rates are higher than we think.”
According to the study’s senior author, Jaime Imitola, MD, who directs the Progressive Multiple Sclerosis Multidisciplinary Clinic and Translational Research Program at the Ohio State University Wexner Medical Center, Columbus, the results “call for the creation of a nationwide registry where neurologists can document adverse reactions to stem cell procedures and further support dedicated patient and neurologist education as we have proposed before” (See Semin Neurol. 2018; 38[2]:176-81 and JAMA Neurol. 2015;72[11]:1342-5).
In an effort to understand the experiences and attitudes of academic neurologists regarding stem cell tourism and patient-reported complications, the researchers developed a 25-question survey disseminated via Synapse, a web tool from the American Academy of Neurology. Respondents were asked about demographic information, frequency of patient questioning, perception of physician competence, patient complications and experiences, and attitudes toward increased physician education.
Dr. Rai, who is a senior neurology resident at the medical center, presented findings from 204 neurologist respondents, of whom 31% identified themselves as MS specialists. Nearly all respondents (91%) said they have been approached by patients with incurable conditions seeking information about stem cells (37% of whom had diagnosis of MS). In addition, 65% have had at least one patient that has undergone “stem cell therapy,” and 73% said it would be “helpful” or “very helpful” to have an evidence-based patient education tool on the topic. “Patients most often wanted general information,” Dr. Rai said. “However, 50% requested permission to undergo a stem cell procedure, and 31% approached their neurologist after the procedure.”
Survey respondents reported that 33% of the stem cell interventions were performed in the United States and 22% abroad, while 37% reported both in the U.S. and abroad. Patients underwent the procedures in China, Germany, the Bahamas, Mexico, Russia, and Costa Rica. Three-quarters of respondents (75%) indicated no patient experiencing complications from the stem cell interventions. However, 25% reported patients experiencing a variety of complications from the procedures, including strokes, meningoencephalitis, quadriparesis, MS deterioration, sepsis, hepatitis C, seizures, meningitis from intrathecal cell injections, infections, and spinal cord tumors. “At least three respondents had a patient who died as a direct complication from stem cell therapy,” Dr. Rai said.
In their poster, the researchers recommended a “multipronged approach to improve education of MS patients from exploitation and engaging multiple stakeholders in the field, including MS academic societies, licensing boards, and legislative bodies. Specifically, we call for creation of evidence-based education for both neurologists and patients, including physical resources that neurologists can use when discussing stem cell interventions with patients and videos on proper counseling during these visits.”
Colleagues from OSU’s Laboratory for Neural Stem Cells and Functional Neurogenetics contributed to this work. The researchers reported having no financial disclosures.
SOURCE: Rai W et al. ACTRIMS Forum 2019, Poster 237.
REPORTING FROM ACTRIMS FORUM 2019
Evaluations for possible MS often turn up one of its many mimics
DALLAS – Of 95 patients referred to two multiple sclerosis (MS) centers for a possible diagnosis of MS, 74% did not have MS, according to a study presented at ACTRIMS Forum 2019. A majority had clinical syndromes or imaging findings that are atypical for MS, which “underscores the importance of familiarity with typical MS clinical and imaging findings in avoiding misdiagnosis,” said Marwa Kaisey, MD, and her research colleagues. Dr. Kaisey is a neurologist at Cedars-Sinai Medical Center in Los Angeles.
Physicians often refer patients to academic MS centers to determine whether patients have MS or one of its many mimics. To study the characteristics and final diagnoses of patients referred to MS centers for evaluation of possible MS, the investigators reviewed electronic medical records and MRI from all new patient evaluations at the Cedars-Sinai Medical Center and University of California, Los Angeles MS clinics between July 2016 and June 2017. The researchers excluded patients referred with a previously established diagnosis of MS.
There were 366 new patients evaluated, including 236 patients with previously established MS diagnoses and 35 patients whose evaluations were not related to MS. Of the 95 patients referred for a question of MS diagnosis, 60% had clinical syndromes that were atypical for MS, 22% had normal neurologic exams, and a third had pain or sensory changes that were not localizable to the CNS.
Sixty-seven percent had MRI that was atypical for MS, and nearly half of the patients without MS had nonspecific MRI changes. “Often, these MRI changes alone prompted referral for an MS evaluation,” Dr. Kaisey and colleagues reported. “This suggests that novel, specific imaging tools may increase diagnostic confidence in the clinical setting.”
In all, the referred patients received 28 diagnoses other than MS, most commonly migraine (10 patients), anxiety or conversion disorder (9), postinfectious or idiopathic transverse myelitis (8), compression myelopathy or spondylopathy (8), and peripheral neuropathy or radiculopathy (7).
The researchers did not have any relevant disclosures.
jremaly@mdedge.com
SOURCE: Kaisey M et al. ACTRIMS Forum 2019, Abstract 90.
DALLAS – Of 95 patients referred to two multiple sclerosis (MS) centers for a possible diagnosis of MS, 74% did not have MS, according to a study presented at ACTRIMS Forum 2019. A majority had clinical syndromes or imaging findings that are atypical for MS, which “underscores the importance of familiarity with typical MS clinical and imaging findings in avoiding misdiagnosis,” said Marwa Kaisey, MD, and her research colleagues. Dr. Kaisey is a neurologist at Cedars-Sinai Medical Center in Los Angeles.
Physicians often refer patients to academic MS centers to determine whether patients have MS or one of its many mimics. To study the characteristics and final diagnoses of patients referred to MS centers for evaluation of possible MS, the investigators reviewed electronic medical records and MRI from all new patient evaluations at the Cedars-Sinai Medical Center and University of California, Los Angeles MS clinics between July 2016 and June 2017. The researchers excluded patients referred with a previously established diagnosis of MS.
There were 366 new patients evaluated, including 236 patients with previously established MS diagnoses and 35 patients whose evaluations were not related to MS. Of the 95 patients referred for a question of MS diagnosis, 60% had clinical syndromes that were atypical for MS, 22% had normal neurologic exams, and a third had pain or sensory changes that were not localizable to the CNS.
Sixty-seven percent had MRI that was atypical for MS, and nearly half of the patients without MS had nonspecific MRI changes. “Often, these MRI changes alone prompted referral for an MS evaluation,” Dr. Kaisey and colleagues reported. “This suggests that novel, specific imaging tools may increase diagnostic confidence in the clinical setting.”
In all, the referred patients received 28 diagnoses other than MS, most commonly migraine (10 patients), anxiety or conversion disorder (9), postinfectious or idiopathic transverse myelitis (8), compression myelopathy or spondylopathy (8), and peripheral neuropathy or radiculopathy (7).
The researchers did not have any relevant disclosures.
jremaly@mdedge.com
SOURCE: Kaisey M et al. ACTRIMS Forum 2019, Abstract 90.
DALLAS – Of 95 patients referred to two multiple sclerosis (MS) centers for a possible diagnosis of MS, 74% did not have MS, according to a study presented at ACTRIMS Forum 2019. A majority had clinical syndromes or imaging findings that are atypical for MS, which “underscores the importance of familiarity with typical MS clinical and imaging findings in avoiding misdiagnosis,” said Marwa Kaisey, MD, and her research colleagues. Dr. Kaisey is a neurologist at Cedars-Sinai Medical Center in Los Angeles.
Physicians often refer patients to academic MS centers to determine whether patients have MS or one of its many mimics. To study the characteristics and final diagnoses of patients referred to MS centers for evaluation of possible MS, the investigators reviewed electronic medical records and MRI from all new patient evaluations at the Cedars-Sinai Medical Center and University of California, Los Angeles MS clinics between July 2016 and June 2017. The researchers excluded patients referred with a previously established diagnosis of MS.
There were 366 new patients evaluated, including 236 patients with previously established MS diagnoses and 35 patients whose evaluations were not related to MS. Of the 95 patients referred for a question of MS diagnosis, 60% had clinical syndromes that were atypical for MS, 22% had normal neurologic exams, and a third had pain or sensory changes that were not localizable to the CNS.
Sixty-seven percent had MRI that was atypical for MS, and nearly half of the patients without MS had nonspecific MRI changes. “Often, these MRI changes alone prompted referral for an MS evaluation,” Dr. Kaisey and colleagues reported. “This suggests that novel, specific imaging tools may increase diagnostic confidence in the clinical setting.”
In all, the referred patients received 28 diagnoses other than MS, most commonly migraine (10 patients), anxiety or conversion disorder (9), postinfectious or idiopathic transverse myelitis (8), compression myelopathy or spondylopathy (8), and peripheral neuropathy or radiculopathy (7).
The researchers did not have any relevant disclosures.
jremaly@mdedge.com
SOURCE: Kaisey M et al. ACTRIMS Forum 2019, Abstract 90.
REPORTING FROM ACTRIMS FORUM 2019
MS prevalence estimates reach highest point to date
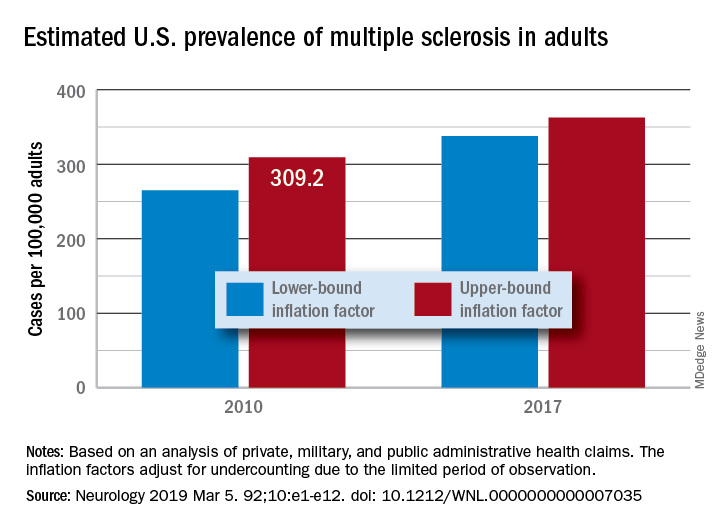
“Our findings suggest that there has been a steady rise in the prevalence of MS over the past 5 decades, that the prevalence of MS remains higher for women than men, and that a north-south geographic gradient still persists,” wrote lead author Mitchell T. Wallin, MD, of Georgetown University, Washington, and his coauthors. The study was published in Neurology.
To determine adult cases of MS, Dr. Wallin and colleagues applied a validated algorithm to private, military, and public AHC datasets. Data from the 2010 U.S. Census were also used to standardize age and sex. In total, 125 million people over 18 years of age were captured in the study, nearly 45% of the U.S. population.
After adjustment, the 2010 prevalence for MS cumulated over 10 years was 309.2 per 100,000 adults (95% confidence interval, 308.1-310.1). This represented a total of 727,344 people with MS. The female to male ratio was 2.8, with a prevalence of 450.1 per 100,000 (95% CI, 448.1-451.6) for women versus a prevalence of 159.7 (95% CI, 158.7-160.6) for men. The age group with the highest estimated prevalence was 55-64 years old, and the prevalence in northern regions of the United States was statistically significantly higher than in southern regions.
The limitations of this study included not including children, the Indian Health Service, the U.S. prison system, or undocumented U.S. residents in the prevalence estimates. However, the authors did note that “these segments of the population are relatively small or, in the case of children, would contribute few cases.” In addition, they were unable to acquire more than 3 years of data for all insurance pools because of high costs.
The study was funded by a grant from the National Multiple Sclerosis Society. The authors reported numerous disclosures, including receiving consulting fees, researching funding, and grant support from various government agencies, foundations, and pharmaceutical companies.
SOURCE: Wallin MT et al. Neurology. 2019 Feb 15. doi: 10.1212/WNL.0000000000007035.

“Our findings suggest that there has been a steady rise in the prevalence of MS over the past 5 decades, that the prevalence of MS remains higher for women than men, and that a north-south geographic gradient still persists,” wrote lead author Mitchell T. Wallin, MD, of Georgetown University, Washington, and his coauthors. The study was published in Neurology.
To determine adult cases of MS, Dr. Wallin and colleagues applied a validated algorithm to private, military, and public AHC datasets. Data from the 2010 U.S. Census were also used to standardize age and sex. In total, 125 million people over 18 years of age were captured in the study, nearly 45% of the U.S. population.
After adjustment, the 2010 prevalence for MS cumulated over 10 years was 309.2 per 100,000 adults (95% confidence interval, 308.1-310.1). This represented a total of 727,344 people with MS. The female to male ratio was 2.8, with a prevalence of 450.1 per 100,000 (95% CI, 448.1-451.6) for women versus a prevalence of 159.7 (95% CI, 158.7-160.6) for men. The age group with the highest estimated prevalence was 55-64 years old, and the prevalence in northern regions of the United States was statistically significantly higher than in southern regions.
The limitations of this study included not including children, the Indian Health Service, the U.S. prison system, or undocumented U.S. residents in the prevalence estimates. However, the authors did note that “these segments of the population are relatively small or, in the case of children, would contribute few cases.” In addition, they were unable to acquire more than 3 years of data for all insurance pools because of high costs.
The study was funded by a grant from the National Multiple Sclerosis Society. The authors reported numerous disclosures, including receiving consulting fees, researching funding, and grant support from various government agencies, foundations, and pharmaceutical companies.
SOURCE: Wallin MT et al. Neurology. 2019 Feb 15. doi: 10.1212/WNL.0000000000007035.

“Our findings suggest that there has been a steady rise in the prevalence of MS over the past 5 decades, that the prevalence of MS remains higher for women than men, and that a north-south geographic gradient still persists,” wrote lead author Mitchell T. Wallin, MD, of Georgetown University, Washington, and his coauthors. The study was published in Neurology.
To determine adult cases of MS, Dr. Wallin and colleagues applied a validated algorithm to private, military, and public AHC datasets. Data from the 2010 U.S. Census were also used to standardize age and sex. In total, 125 million people over 18 years of age were captured in the study, nearly 45% of the U.S. population.
After adjustment, the 2010 prevalence for MS cumulated over 10 years was 309.2 per 100,000 adults (95% confidence interval, 308.1-310.1). This represented a total of 727,344 people with MS. The female to male ratio was 2.8, with a prevalence of 450.1 per 100,000 (95% CI, 448.1-451.6) for women versus a prevalence of 159.7 (95% CI, 158.7-160.6) for men. The age group with the highest estimated prevalence was 55-64 years old, and the prevalence in northern regions of the United States was statistically significantly higher than in southern regions.
The limitations of this study included not including children, the Indian Health Service, the U.S. prison system, or undocumented U.S. residents in the prevalence estimates. However, the authors did note that “these segments of the population are relatively small or, in the case of children, would contribute few cases.” In addition, they were unable to acquire more than 3 years of data for all insurance pools because of high costs.
The study was funded by a grant from the National Multiple Sclerosis Society. The authors reported numerous disclosures, including receiving consulting fees, researching funding, and grant support from various government agencies, foundations, and pharmaceutical companies.
SOURCE: Wallin MT et al. Neurology. 2019 Feb 15. doi: 10.1212/WNL.0000000000007035.
FROM NEUROLOGY
What happens when RRMS patients discontinue their DMT?
DALLAS – results from a single-center study showed.
In addition, being over the age of 45 years was associated with a better disease course after treatment discontinuation.
“Being clinically and radiologically stable for more than 2 years can be a potential milestone to regard the discontinuation of DMT [disease-modifying therapy] as a reasonable option in a subset of patients, especially patients who are nondisabled,” lead study author Hajime Yano, MD, said in an interview at the Americas Committee for Treatment and Research in Multiple Sclerosis.
According to Dr. Yano, a research fellow at the Ann Romney Center for Neurologic Diseases and Partners Multiple Sclerosis Center in Boston, relapsing remitting multiple sclerosis (RRMS) patients without relapse for long periods on treatment may consider discontinuing DMT, but there is limited information regarding the impact of discontinuation, especially in terms of MRI activity.
In an effort to investigate the impact of DMT discontinuation on clinical and radiologic outcomes in RRMS patients, he and his colleagues identified 70 patients from the Comprehensive Longitudinal Investigation of Multiple Sclerosis at the Brigham and Women’s Hospital (CLIMB) study, which was initiated in 2000 and has enrolled more than 2,400 patients cared for at the Partners Multiple Sclerosis Center. Relapse date, symptoms, and Expanded Disability Status Scale (EDSS) were evaluated at 6-month intervals for each patient during the time of clinic visits by the treating neurologist. Additionally, brain MRIs were performed annually.
Next, the researchers matched the patients with 70 patients who remained on DMT identified by age, sex, treatment, treatment duration, disease duration, and EDSS. They used univariate and multivariable Cox proportional hazard models to test the differences between DMT discontinuation status with time to clinical relapse, MRI event, disability progression, and any inflammatory event (either clinical relapse or MRI event).
The mean age of patients was 45 years, 87% were female, their mean disease duration was about 13 years, and they had been receiving treatment for a mean of about 6 years. In adjusted analyses, the 70 pairs of patients who discontinued DMT and patients who continued DMT had similar outcomes in time to clinical relapse (hazard ratio, 0.93; P = .84), MRI event (HR, 1.01; P = .98), disability progression (HR, 1.33; P = .43), and any inflammatory event (HR, 0.93; P = .85). In a subgroup analysis, which compared the impact of DMT discontinuation between patients over the age of 45 years and those aged 45 years and younger, the researchers observed a statistically significant difference in effect of discontinuation on time to clinical relapse (P = .032), time to MRI event (P = .013), and time to any inflammatory event (P = .0005), all favoring patients over the age of 45 years.
“This finding makes sense since age has been reported as one of the factors that negatively impacts on the inflammatory activity in patients with RRMS,” Dr. Yano said. “However, our study is the first study [to find] that the impact of discontinuing DMT on RRMS patient prognosis may differ based on the age at the discontinuation. In short, stopping DMT at a younger age has a statistically significant higher risk on inflammatory activities, compared to [stopping DMT at an] older age.”
He acknowledged certain limitations of the study, including its small sample size and single-center design. However, Dr. Yano said that a key strength of the analysis was the inclusion of MRI activity prior to DMT as the definition of stable state, “which is an integral piece of information when physicians and patients consider DMT discontinuation in a ‘real world’ clinical setting. We also used MRI activity as an outcome measure, which is lacking in prior discontinuation studies.”
Dr. Yano reported that he has received a research grant from Yoshida Scholarship Foundation in Japan. His coauthors reported having numerous financial ties to industry.
SOURCE: Yano H et al. ACTRIMS Forum 2019, Poster 061.
DALLAS – results from a single-center study showed.
In addition, being over the age of 45 years was associated with a better disease course after treatment discontinuation.
“Being clinically and radiologically stable for more than 2 years can be a potential milestone to regard the discontinuation of DMT [disease-modifying therapy] as a reasonable option in a subset of patients, especially patients who are nondisabled,” lead study author Hajime Yano, MD, said in an interview at the Americas Committee for Treatment and Research in Multiple Sclerosis.
According to Dr. Yano, a research fellow at the Ann Romney Center for Neurologic Diseases and Partners Multiple Sclerosis Center in Boston, relapsing remitting multiple sclerosis (RRMS) patients without relapse for long periods on treatment may consider discontinuing DMT, but there is limited information regarding the impact of discontinuation, especially in terms of MRI activity.
In an effort to investigate the impact of DMT discontinuation on clinical and radiologic outcomes in RRMS patients, he and his colleagues identified 70 patients from the Comprehensive Longitudinal Investigation of Multiple Sclerosis at the Brigham and Women’s Hospital (CLIMB) study, which was initiated in 2000 and has enrolled more than 2,400 patients cared for at the Partners Multiple Sclerosis Center. Relapse date, symptoms, and Expanded Disability Status Scale (EDSS) were evaluated at 6-month intervals for each patient during the time of clinic visits by the treating neurologist. Additionally, brain MRIs were performed annually.
Next, the researchers matched the patients with 70 patients who remained on DMT identified by age, sex, treatment, treatment duration, disease duration, and EDSS. They used univariate and multivariable Cox proportional hazard models to test the differences between DMT discontinuation status with time to clinical relapse, MRI event, disability progression, and any inflammatory event (either clinical relapse or MRI event).
The mean age of patients was 45 years, 87% were female, their mean disease duration was about 13 years, and they had been receiving treatment for a mean of about 6 years. In adjusted analyses, the 70 pairs of patients who discontinued DMT and patients who continued DMT had similar outcomes in time to clinical relapse (hazard ratio, 0.93; P = .84), MRI event (HR, 1.01; P = .98), disability progression (HR, 1.33; P = .43), and any inflammatory event (HR, 0.93; P = .85). In a subgroup analysis, which compared the impact of DMT discontinuation between patients over the age of 45 years and those aged 45 years and younger, the researchers observed a statistically significant difference in effect of discontinuation on time to clinical relapse (P = .032), time to MRI event (P = .013), and time to any inflammatory event (P = .0005), all favoring patients over the age of 45 years.
“This finding makes sense since age has been reported as one of the factors that negatively impacts on the inflammatory activity in patients with RRMS,” Dr. Yano said. “However, our study is the first study [to find] that the impact of discontinuing DMT on RRMS patient prognosis may differ based on the age at the discontinuation. In short, stopping DMT at a younger age has a statistically significant higher risk on inflammatory activities, compared to [stopping DMT at an] older age.”
He acknowledged certain limitations of the study, including its small sample size and single-center design. However, Dr. Yano said that a key strength of the analysis was the inclusion of MRI activity prior to DMT as the definition of stable state, “which is an integral piece of information when physicians and patients consider DMT discontinuation in a ‘real world’ clinical setting. We also used MRI activity as an outcome measure, which is lacking in prior discontinuation studies.”
Dr. Yano reported that he has received a research grant from Yoshida Scholarship Foundation in Japan. His coauthors reported having numerous financial ties to industry.
SOURCE: Yano H et al. ACTRIMS Forum 2019, Poster 061.
DALLAS – results from a single-center study showed.
In addition, being over the age of 45 years was associated with a better disease course after treatment discontinuation.
“Being clinically and radiologically stable for more than 2 years can be a potential milestone to regard the discontinuation of DMT [disease-modifying therapy] as a reasonable option in a subset of patients, especially patients who are nondisabled,” lead study author Hajime Yano, MD, said in an interview at the Americas Committee for Treatment and Research in Multiple Sclerosis.
According to Dr. Yano, a research fellow at the Ann Romney Center for Neurologic Diseases and Partners Multiple Sclerosis Center in Boston, relapsing remitting multiple sclerosis (RRMS) patients without relapse for long periods on treatment may consider discontinuing DMT, but there is limited information regarding the impact of discontinuation, especially in terms of MRI activity.
In an effort to investigate the impact of DMT discontinuation on clinical and radiologic outcomes in RRMS patients, he and his colleagues identified 70 patients from the Comprehensive Longitudinal Investigation of Multiple Sclerosis at the Brigham and Women’s Hospital (CLIMB) study, which was initiated in 2000 and has enrolled more than 2,400 patients cared for at the Partners Multiple Sclerosis Center. Relapse date, symptoms, and Expanded Disability Status Scale (EDSS) were evaluated at 6-month intervals for each patient during the time of clinic visits by the treating neurologist. Additionally, brain MRIs were performed annually.
Next, the researchers matched the patients with 70 patients who remained on DMT identified by age, sex, treatment, treatment duration, disease duration, and EDSS. They used univariate and multivariable Cox proportional hazard models to test the differences between DMT discontinuation status with time to clinical relapse, MRI event, disability progression, and any inflammatory event (either clinical relapse or MRI event).
The mean age of patients was 45 years, 87% were female, their mean disease duration was about 13 years, and they had been receiving treatment for a mean of about 6 years. In adjusted analyses, the 70 pairs of patients who discontinued DMT and patients who continued DMT had similar outcomes in time to clinical relapse (hazard ratio, 0.93; P = .84), MRI event (HR, 1.01; P = .98), disability progression (HR, 1.33; P = .43), and any inflammatory event (HR, 0.93; P = .85). In a subgroup analysis, which compared the impact of DMT discontinuation between patients over the age of 45 years and those aged 45 years and younger, the researchers observed a statistically significant difference in effect of discontinuation on time to clinical relapse (P = .032), time to MRI event (P = .013), and time to any inflammatory event (P = .0005), all favoring patients over the age of 45 years.
“This finding makes sense since age has been reported as one of the factors that negatively impacts on the inflammatory activity in patients with RRMS,” Dr. Yano said. “However, our study is the first study [to find] that the impact of discontinuing DMT on RRMS patient prognosis may differ based on the age at the discontinuation. In short, stopping DMT at a younger age has a statistically significant higher risk on inflammatory activities, compared to [stopping DMT at an] older age.”
He acknowledged certain limitations of the study, including its small sample size and single-center design. However, Dr. Yano said that a key strength of the analysis was the inclusion of MRI activity prior to DMT as the definition of stable state, “which is an integral piece of information when physicians and patients consider DMT discontinuation in a ‘real world’ clinical setting. We also used MRI activity as an outcome measure, which is lacking in prior discontinuation studies.”
Dr. Yano reported that he has received a research grant from Yoshida Scholarship Foundation in Japan. His coauthors reported having numerous financial ties to industry.
SOURCE: Yano H et al. ACTRIMS Forum 2019, Poster 061.
REPORTING FROM ACTRIMS FORUM 2019
Key clinical point: Patients who discontinued disease-modifying therapy after a period of disease inactivity had a similar time to next event, compared with patients who remained on treatment.
Major finding: Compared with patients aged 45 years and younger, older patients who discontinued disease-modifying therapy had significantly favorable disease course in terms of time to clinical relapse (P = .032), time to MRI event (P = .013), and time to any inflammatory event (P = .0005).
Study details: A single-center study of 140 patients with relapsing remitting multiple sclerosis.
Disclosures: Dr. Yano reported that he has received a research grant from the Yoshida Scholarship Foundation in Japan. His coauthors reported having numerous financial ties to industry.Source: Yano H et al. ACTRIMS Forum 2019, Poster 061.
Can technology automate assessments of patients with MS in the clinic?
DALLAS – according to research described at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
An analysis of data collected using these methods found that patient-reported outcomes and MRI measures correlate with neuroperformance test results, said Laura Baldassari, MD, a clinical neuroimmunology fellow at the Mellen Center for Multiple Sclerosis at the Cleveland Clinic. Such assessments “could potentially enable us to better tune in to disability worsening and treatment response in our patients.”
The Multiple Sclerosis Performance Test (MSPT) collects patient-reported outcomes and tests patients’ processing speed, contrast sensitivity, manual dexterity, and walking speed. The MSPT is designed for supervised or independent administration with an assistant and has been “incorporated into routine clinical care at the Mellen Center,” Dr. Baldassari said. Before seeing their provider, patients complete the MSPT with a biomedical assistant, which usually takes 30-40 minutes. The data are scored instantly and “integrated into the electronic medical record for use during the clinical encounter.”
Dr. Baldassari and her research colleagues analyzed associations between the neuroperformance metrics, patient-reported outcome measures, and quantitative MRI metrics. The analysis included 976 patients who completed the MSPT between December 2015 and December 2017 and had an MRI within 3 months of a clinical encounter. T2 lesion volume, normalized whole brain volume or whole brain fraction, thalamic volume, and cross-sectional upper cervical spinal cord area at the level of C2 on MRI were calculated using a fully automated method.
Patient-reported outcomes included Quality of Life in Neurological Disorders (Neuro-QoL) upper and lower extremity function, Patient-Reported Outcomes Measurement Information System (PROMIS) physical, and Patient Determined Disease Steps (PDDS).
The researchers used Spearman correlation coefficients to examine the relationships between each neuroperformance test, patient-reported outcome, and MRI measure. Linear regression models determined which clinical demographic, patient-reported outcome, or MRI characteristic predicted neuroperformance test results.
Patients had a mean age of about 48 years, and the population was predominantly female and white with relapsing remitting MS.
“There were significant correlations between all neuroperformance tests and all patient-reported outcomes except for the contrast sensitivity test and PROMIS physical,” Dr. Baldassari said. “The processing speed test was most strongly correlated with the PDDS as well as the Neuro-QoL lower extremity. The contrast sensitivity test was correlated with Neuro-QoL lower extremity as well.” The manual dexterity test correlated with PDDS and Neuro-QoL upper and lower extremity and the walking speed test correlated with PDDS and Neuro-QoL lower extremity.
“With worsening self-reported functions, these neuroperformance test results demonstrated impairment as well,” she said.
The neuroperformance tests and all MRI metrics had significant, moderate correlations. “The strongest correlations here are between the processing speed test and whole brain fraction and T2 lesion volume; contrast sensitivity and T2 lesion volume, whole brain fraction, and thalamic volume; manual dexterity test and T2 lesion volume and whole brain fraction; and walking speed test and whole brain fraction and cord area,” she said.
“The strongest predictors of each neuroperformance test varied, which highlights the unique complementary contribution of each patient-reported outcome measure and MRI metric to the complex domains of disability in MS,” Dr. Baldassari said.
Comprehensive, quantitative MS assessments may lead to detailed patient profiles, which could support more precise clinical care and observational studies. In future studies, the researchers plan to examine how these measures relate over time.
The MSPT was developed by the Cleveland Clinic in partnership with Biogen. Dr. Baldassari reported receiving funding through the National Multiple Sclerosis Society and personal fees for serving on a scientific advisory board for Teva. His coauthors’ disclosures included the contribution of intellectual property to the MSPT, for which they could receive royalties.
SOURCE: Baldassari L et al. ACTRIMS Forum 2019, Abstract 32.
DALLAS – according to research described at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
An analysis of data collected using these methods found that patient-reported outcomes and MRI measures correlate with neuroperformance test results, said Laura Baldassari, MD, a clinical neuroimmunology fellow at the Mellen Center for Multiple Sclerosis at the Cleveland Clinic. Such assessments “could potentially enable us to better tune in to disability worsening and treatment response in our patients.”
The Multiple Sclerosis Performance Test (MSPT) collects patient-reported outcomes and tests patients’ processing speed, contrast sensitivity, manual dexterity, and walking speed. The MSPT is designed for supervised or independent administration with an assistant and has been “incorporated into routine clinical care at the Mellen Center,” Dr. Baldassari said. Before seeing their provider, patients complete the MSPT with a biomedical assistant, which usually takes 30-40 minutes. The data are scored instantly and “integrated into the electronic medical record for use during the clinical encounter.”
Dr. Baldassari and her research colleagues analyzed associations between the neuroperformance metrics, patient-reported outcome measures, and quantitative MRI metrics. The analysis included 976 patients who completed the MSPT between December 2015 and December 2017 and had an MRI within 3 months of a clinical encounter. T2 lesion volume, normalized whole brain volume or whole brain fraction, thalamic volume, and cross-sectional upper cervical spinal cord area at the level of C2 on MRI were calculated using a fully automated method.
Patient-reported outcomes included Quality of Life in Neurological Disorders (Neuro-QoL) upper and lower extremity function, Patient-Reported Outcomes Measurement Information System (PROMIS) physical, and Patient Determined Disease Steps (PDDS).
The researchers used Spearman correlation coefficients to examine the relationships between each neuroperformance test, patient-reported outcome, and MRI measure. Linear regression models determined which clinical demographic, patient-reported outcome, or MRI characteristic predicted neuroperformance test results.
Patients had a mean age of about 48 years, and the population was predominantly female and white with relapsing remitting MS.
“There were significant correlations between all neuroperformance tests and all patient-reported outcomes except for the contrast sensitivity test and PROMIS physical,” Dr. Baldassari said. “The processing speed test was most strongly correlated with the PDDS as well as the Neuro-QoL lower extremity. The contrast sensitivity test was correlated with Neuro-QoL lower extremity as well.” The manual dexterity test correlated with PDDS and Neuro-QoL upper and lower extremity and the walking speed test correlated with PDDS and Neuro-QoL lower extremity.
“With worsening self-reported functions, these neuroperformance test results demonstrated impairment as well,” she said.
The neuroperformance tests and all MRI metrics had significant, moderate correlations. “The strongest correlations here are between the processing speed test and whole brain fraction and T2 lesion volume; contrast sensitivity and T2 lesion volume, whole brain fraction, and thalamic volume; manual dexterity test and T2 lesion volume and whole brain fraction; and walking speed test and whole brain fraction and cord area,” she said.
“The strongest predictors of each neuroperformance test varied, which highlights the unique complementary contribution of each patient-reported outcome measure and MRI metric to the complex domains of disability in MS,” Dr. Baldassari said.
Comprehensive, quantitative MS assessments may lead to detailed patient profiles, which could support more precise clinical care and observational studies. In future studies, the researchers plan to examine how these measures relate over time.
The MSPT was developed by the Cleveland Clinic in partnership with Biogen. Dr. Baldassari reported receiving funding through the National Multiple Sclerosis Society and personal fees for serving on a scientific advisory board for Teva. His coauthors’ disclosures included the contribution of intellectual property to the MSPT, for which they could receive royalties.
SOURCE: Baldassari L et al. ACTRIMS Forum 2019, Abstract 32.
DALLAS – according to research described at the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis.
An analysis of data collected using these methods found that patient-reported outcomes and MRI measures correlate with neuroperformance test results, said Laura Baldassari, MD, a clinical neuroimmunology fellow at the Mellen Center for Multiple Sclerosis at the Cleveland Clinic. Such assessments “could potentially enable us to better tune in to disability worsening and treatment response in our patients.”
The Multiple Sclerosis Performance Test (MSPT) collects patient-reported outcomes and tests patients’ processing speed, contrast sensitivity, manual dexterity, and walking speed. The MSPT is designed for supervised or independent administration with an assistant and has been “incorporated into routine clinical care at the Mellen Center,” Dr. Baldassari said. Before seeing their provider, patients complete the MSPT with a biomedical assistant, which usually takes 30-40 minutes. The data are scored instantly and “integrated into the electronic medical record for use during the clinical encounter.”
Dr. Baldassari and her research colleagues analyzed associations between the neuroperformance metrics, patient-reported outcome measures, and quantitative MRI metrics. The analysis included 976 patients who completed the MSPT between December 2015 and December 2017 and had an MRI within 3 months of a clinical encounter. T2 lesion volume, normalized whole brain volume or whole brain fraction, thalamic volume, and cross-sectional upper cervical spinal cord area at the level of C2 on MRI were calculated using a fully automated method.
Patient-reported outcomes included Quality of Life in Neurological Disorders (Neuro-QoL) upper and lower extremity function, Patient-Reported Outcomes Measurement Information System (PROMIS) physical, and Patient Determined Disease Steps (PDDS).
The researchers used Spearman correlation coefficients to examine the relationships between each neuroperformance test, patient-reported outcome, and MRI measure. Linear regression models determined which clinical demographic, patient-reported outcome, or MRI characteristic predicted neuroperformance test results.
Patients had a mean age of about 48 years, and the population was predominantly female and white with relapsing remitting MS.
“There were significant correlations between all neuroperformance tests and all patient-reported outcomes except for the contrast sensitivity test and PROMIS physical,” Dr. Baldassari said. “The processing speed test was most strongly correlated with the PDDS as well as the Neuro-QoL lower extremity. The contrast sensitivity test was correlated with Neuro-QoL lower extremity as well.” The manual dexterity test correlated with PDDS and Neuro-QoL upper and lower extremity and the walking speed test correlated with PDDS and Neuro-QoL lower extremity.
“With worsening self-reported functions, these neuroperformance test results demonstrated impairment as well,” she said.
The neuroperformance tests and all MRI metrics had significant, moderate correlations. “The strongest correlations here are between the processing speed test and whole brain fraction and T2 lesion volume; contrast sensitivity and T2 lesion volume, whole brain fraction, and thalamic volume; manual dexterity test and T2 lesion volume and whole brain fraction; and walking speed test and whole brain fraction and cord area,” she said.
“The strongest predictors of each neuroperformance test varied, which highlights the unique complementary contribution of each patient-reported outcome measure and MRI metric to the complex domains of disability in MS,” Dr. Baldassari said.
Comprehensive, quantitative MS assessments may lead to detailed patient profiles, which could support more precise clinical care and observational studies. In future studies, the researchers plan to examine how these measures relate over time.
The MSPT was developed by the Cleveland Clinic in partnership with Biogen. Dr. Baldassari reported receiving funding through the National Multiple Sclerosis Society and personal fees for serving on a scientific advisory board for Teva. His coauthors’ disclosures included the contribution of intellectual property to the MSPT, for which they could receive royalties.
SOURCE: Baldassari L et al. ACTRIMS Forum 2019, Abstract 32.
REPORTING FROM ACTRIMS FORUM 2019
Don’t forget social determinants of health in minority MS patients
DALLAS – The way Lilyana Amezcua, MD, sees it, clinicians should view race and ethnicity as health disparities when assessing individuals with multiple sclerosis.
Whites are predominately affected with MS, “but we have seen changing demographics,” said Dr. Amezcua, of the University of Southern California MS Comprehensive Care and Research Group. “Why are African Americans now at higher risk ... and why do African Americans appear to have more severe disease? Is it a biological difference ... or is it because of poor access” to health care?
At the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis, Dr. Amezcua delivered a presentation entitled “Effect of Race and Ethnicity on MS Presentation and Disease Course.” She called on researchers in the field “to not just take race and ethnicity as any small variable. We need to be cognizant and use the correct methodology, depending on what [question] we want to answer. We need to better define how we ascertain race, how we ascertain ethnicity.”
Dr. Amezcua, who is also the MS fellowship program director at the Keck School of Medicine, disclosed that she receives funding from the National MS Society, the National Institutes of Health, the California Community Foundation, and Biogen.
DALLAS – The way Lilyana Amezcua, MD, sees it, clinicians should view race and ethnicity as health disparities when assessing individuals with multiple sclerosis.
Whites are predominately affected with MS, “but we have seen changing demographics,” said Dr. Amezcua, of the University of Southern California MS Comprehensive Care and Research Group. “Why are African Americans now at higher risk ... and why do African Americans appear to have more severe disease? Is it a biological difference ... or is it because of poor access” to health care?
At the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis, Dr. Amezcua delivered a presentation entitled “Effect of Race and Ethnicity on MS Presentation and Disease Course.” She called on researchers in the field “to not just take race and ethnicity as any small variable. We need to be cognizant and use the correct methodology, depending on what [question] we want to answer. We need to better define how we ascertain race, how we ascertain ethnicity.”
Dr. Amezcua, who is also the MS fellowship program director at the Keck School of Medicine, disclosed that she receives funding from the National MS Society, the National Institutes of Health, the California Community Foundation, and Biogen.
DALLAS – The way Lilyana Amezcua, MD, sees it, clinicians should view race and ethnicity as health disparities when assessing individuals with multiple sclerosis.
Whites are predominately affected with MS, “but we have seen changing demographics,” said Dr. Amezcua, of the University of Southern California MS Comprehensive Care and Research Group. “Why are African Americans now at higher risk ... and why do African Americans appear to have more severe disease? Is it a biological difference ... or is it because of poor access” to health care?
At the meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis, Dr. Amezcua delivered a presentation entitled “Effect of Race and Ethnicity on MS Presentation and Disease Course.” She called on researchers in the field “to not just take race and ethnicity as any small variable. We need to be cognizant and use the correct methodology, depending on what [question] we want to answer. We need to better define how we ascertain race, how we ascertain ethnicity.”
Dr. Amezcua, who is also the MS fellowship program director at the Keck School of Medicine, disclosed that she receives funding from the National MS Society, the National Institutes of Health, the California Community Foundation, and Biogen.
REPORTING FROM ACTRIMS FORUM 2019
Ublituximab depletes B cells in phase 2 trial
DALLAS – (MS), according to phase 2 trial results presented at ACTRIMS Forum 2019. Patients treated with the investigational therapy had reduced MRI activity and relapse rates during the 48-week trial, and the treatment was well tolerated, researchers said.
The monoclonal antibody targets a unique epitope on the CD20 antigen and is glycoengineered for enhanced B-cell targeting through antibody-dependent cellular cytotoxicity, said presenting author Edward Fox, MD, PhD, director of the MS Clinic of Central Texas, Round Rock. Ublituximab’s potency “may offer a benefit over currently available anti-CD20s in terms of lower doses and shorter infusion times,” Dr. Fox and his research colleagues said.
To assess the optimal dose, infusion time, safety, and tolerability of ublituximab in relapsing MS, investigators conducted a phase 2, multicenter study. The trial included 48 patients with relapsing MS; 65% were female. Patients’ average age was 40 years and average disease duration was 7.7 years. The researchers included patients with one or more confirmed relapse in the past year, two relapses in the past 2 years, or at least one active gadolinium-enhancing T1 lesion. The primary endpoint was the percentage of patients with at least a 95% reduction in peripheral CD19+ B cells within 2 weeks after the second infusion on day 15.
For their first infusions, patients received 150 mg of ublituximab over an infusion time of 1, 2, 3, or 4 hours. On day 15, patients received 450 mg or 600 mg of ublituximab over an infusion time of 1, 1.5, or 3 hours. At week 24, patients received 450 mg or 650 mg of ublituximab infused over 1 hour or 1.5 hours.
All patients met the primary endpoint of greater than 95% B-cell depletion between baseline and week 4. Median B-cell depletion was 99% at week 4, and this effect was maintained at weeks 24 and 48.
The researchers detected no T1 gadolinium-enhancing lesions at week 24 or week 48, and total T2 lesion volume decreased by 10.6% between baseline and week 48.
The most frequent adverse events were infusion-related reactions, which occurred in 48% of patients and were more common with the first infusion, particularly when the infusion time was less than 4 hours. All of the infusion-related reactions were grade 1 or 2. One grade 3 serious adverse event of fatigue was considered possibly related to ublituximab. No patients withdrew from the study because of drug-related adverse events. At week 48, 93% of the patients were relapse free, 7% had 24-week confirmed disability progression, and 17% had confirmed disability improvement.
TG Therapeutics, the company developing ublituximab, is evaluating the therapy in phase 3 trials known as ULTIMATE I and 2. The phase 3 trials are using the 450-mg dose with a first dose of 150 mg delivered over 4 hours.
Dr. Fox has disclosed research support from TG Therapeutics and other pharmaceutical companies and working as a consultant and speaker for TG Therapeutics and other companies.
SOURCE: Fox E et al. ACTRIMS Forum 2019, Abstract 66.
DALLAS – (MS), according to phase 2 trial results presented at ACTRIMS Forum 2019. Patients treated with the investigational therapy had reduced MRI activity and relapse rates during the 48-week trial, and the treatment was well tolerated, researchers said.
The monoclonal antibody targets a unique epitope on the CD20 antigen and is glycoengineered for enhanced B-cell targeting through antibody-dependent cellular cytotoxicity, said presenting author Edward Fox, MD, PhD, director of the MS Clinic of Central Texas, Round Rock. Ublituximab’s potency “may offer a benefit over currently available anti-CD20s in terms of lower doses and shorter infusion times,” Dr. Fox and his research colleagues said.
To assess the optimal dose, infusion time, safety, and tolerability of ublituximab in relapsing MS, investigators conducted a phase 2, multicenter study. The trial included 48 patients with relapsing MS; 65% were female. Patients’ average age was 40 years and average disease duration was 7.7 years. The researchers included patients with one or more confirmed relapse in the past year, two relapses in the past 2 years, or at least one active gadolinium-enhancing T1 lesion. The primary endpoint was the percentage of patients with at least a 95% reduction in peripheral CD19+ B cells within 2 weeks after the second infusion on day 15.
For their first infusions, patients received 150 mg of ublituximab over an infusion time of 1, 2, 3, or 4 hours. On day 15, patients received 450 mg or 600 mg of ublituximab over an infusion time of 1, 1.5, or 3 hours. At week 24, patients received 450 mg or 650 mg of ublituximab infused over 1 hour or 1.5 hours.
All patients met the primary endpoint of greater than 95% B-cell depletion between baseline and week 4. Median B-cell depletion was 99% at week 4, and this effect was maintained at weeks 24 and 48.
The researchers detected no T1 gadolinium-enhancing lesions at week 24 or week 48, and total T2 lesion volume decreased by 10.6% between baseline and week 48.
The most frequent adverse events were infusion-related reactions, which occurred in 48% of patients and were more common with the first infusion, particularly when the infusion time was less than 4 hours. All of the infusion-related reactions were grade 1 or 2. One grade 3 serious adverse event of fatigue was considered possibly related to ublituximab. No patients withdrew from the study because of drug-related adverse events. At week 48, 93% of the patients were relapse free, 7% had 24-week confirmed disability progression, and 17% had confirmed disability improvement.
TG Therapeutics, the company developing ublituximab, is evaluating the therapy in phase 3 trials known as ULTIMATE I and 2. The phase 3 trials are using the 450-mg dose with a first dose of 150 mg delivered over 4 hours.
Dr. Fox has disclosed research support from TG Therapeutics and other pharmaceutical companies and working as a consultant and speaker for TG Therapeutics and other companies.
SOURCE: Fox E et al. ACTRIMS Forum 2019, Abstract 66.
DALLAS – (MS), according to phase 2 trial results presented at ACTRIMS Forum 2019. Patients treated with the investigational therapy had reduced MRI activity and relapse rates during the 48-week trial, and the treatment was well tolerated, researchers said.
The monoclonal antibody targets a unique epitope on the CD20 antigen and is glycoengineered for enhanced B-cell targeting through antibody-dependent cellular cytotoxicity, said presenting author Edward Fox, MD, PhD, director of the MS Clinic of Central Texas, Round Rock. Ublituximab’s potency “may offer a benefit over currently available anti-CD20s in terms of lower doses and shorter infusion times,” Dr. Fox and his research colleagues said.
To assess the optimal dose, infusion time, safety, and tolerability of ublituximab in relapsing MS, investigators conducted a phase 2, multicenter study. The trial included 48 patients with relapsing MS; 65% were female. Patients’ average age was 40 years and average disease duration was 7.7 years. The researchers included patients with one or more confirmed relapse in the past year, two relapses in the past 2 years, or at least one active gadolinium-enhancing T1 lesion. The primary endpoint was the percentage of patients with at least a 95% reduction in peripheral CD19+ B cells within 2 weeks after the second infusion on day 15.
For their first infusions, patients received 150 mg of ublituximab over an infusion time of 1, 2, 3, or 4 hours. On day 15, patients received 450 mg or 600 mg of ublituximab over an infusion time of 1, 1.5, or 3 hours. At week 24, patients received 450 mg or 650 mg of ublituximab infused over 1 hour or 1.5 hours.
All patients met the primary endpoint of greater than 95% B-cell depletion between baseline and week 4. Median B-cell depletion was 99% at week 4, and this effect was maintained at weeks 24 and 48.
The researchers detected no T1 gadolinium-enhancing lesions at week 24 or week 48, and total T2 lesion volume decreased by 10.6% between baseline and week 48.
The most frequent adverse events were infusion-related reactions, which occurred in 48% of patients and were more common with the first infusion, particularly when the infusion time was less than 4 hours. All of the infusion-related reactions were grade 1 or 2. One grade 3 serious adverse event of fatigue was considered possibly related to ublituximab. No patients withdrew from the study because of drug-related adverse events. At week 48, 93% of the patients were relapse free, 7% had 24-week confirmed disability progression, and 17% had confirmed disability improvement.
TG Therapeutics, the company developing ublituximab, is evaluating the therapy in phase 3 trials known as ULTIMATE I and 2. The phase 3 trials are using the 450-mg dose with a first dose of 150 mg delivered over 4 hours.
Dr. Fox has disclosed research support from TG Therapeutics and other pharmaceutical companies and working as a consultant and speaker for TG Therapeutics and other companies.
SOURCE: Fox E et al. ACTRIMS Forum 2019, Abstract 66.
REPORTING FROM ACTRIMS FORUM 2019
CSF biomarker clusters correlate with MS severity
DALLAS – Patients with multiple sclerosis (MS) have elevated levels of specific clusters of cerebrospinal fluid (CSF) biomarkers related to astrocytes and microglia that correlated with disease severity in a blinded analysis of more than 1,000 proteins from the CSF of more than 400 patients with neuroimmunologic disease and healthy volunteers.
Previous studies have indicated that aberrant activation of astrocytes and microglia underlies disability progression in older patients with MS, but researchers have lacked biomarkers of these processes in living subjects. In a presentation at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis, Ruturaj R. Masvekar, PhD, described developing biomarkers of CNS cell–specific processes and examining how they relate to MS disability progression. Dr. Masvekar, a researcher at the National Institute of Allergy and Infectious Diseases, and his coinvestigators used a modified DNA aptamer assay to measure proteins in the CSF of 431 patients with neuroimmunologic diseases and healthy volunteers, followed by variable cluster analysis and in vitro modeling to define 64 clusters of CSF biomarkers that relate to CNS cell types.
The study included 42 healthy donors, 20 patients with clinically isolated syndrome, 57 patients with noninflammatory neurologic disorders, 127 patients with relapsing-remitting MS, 72 patients with secondary progressive MS, and 113 patients with primary progressive MS. In a training cohort of 217 participants, the researchers assessed how biomarkers differed between the diagnostic categories. The researchers then validated the results in an independent cohort of 214 participants.
One astrocyte-related cluster (MMP7, SERPINA3, GZMA, and CLIC1) and one microglia-related cluster (DSG2 and TNFRSF25) was significantly elevated in all MS subgroups, compared with healthy controls and patients with noninflammatory neurologic disorders.
In addition, these clusters “significantly correlated with clinical measures of disability, CNS tissue destruction, and MS severity,” Dr. Masvekar said.
The microglial cluster was significantly elevated in all MS subgroups, whereas neuronal endothelial, astrocytic, and oligodendroglial biomarker clusters were elevated only in patients with progressive MS.
“Microglial activation is present in all stages of MS, while toxic astrogliosis increases with MS duration, concomitantly with neuronal and oligodendroglial degeneration,” Dr. Masvekar said. “Microglial activation and toxic astrogliosis likely partake in CNS tissue destruction and enhance MS severity.”
This study, which was recently published in Multiple Sclerosis and Related Disorders (2019 Feb;28:34-43), was supported by the intramural research program at NIAID.
jremaly@mdedge.com
SOURCE: Masvekar RR et al. ACTRIMS Forum 2019, Abstract 281.
DALLAS – Patients with multiple sclerosis (MS) have elevated levels of specific clusters of cerebrospinal fluid (CSF) biomarkers related to astrocytes and microglia that correlated with disease severity in a blinded analysis of more than 1,000 proteins from the CSF of more than 400 patients with neuroimmunologic disease and healthy volunteers.
Previous studies have indicated that aberrant activation of astrocytes and microglia underlies disability progression in older patients with MS, but researchers have lacked biomarkers of these processes in living subjects. In a presentation at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis, Ruturaj R. Masvekar, PhD, described developing biomarkers of CNS cell–specific processes and examining how they relate to MS disability progression. Dr. Masvekar, a researcher at the National Institute of Allergy and Infectious Diseases, and his coinvestigators used a modified DNA aptamer assay to measure proteins in the CSF of 431 patients with neuroimmunologic diseases and healthy volunteers, followed by variable cluster analysis and in vitro modeling to define 64 clusters of CSF biomarkers that relate to CNS cell types.
The study included 42 healthy donors, 20 patients with clinically isolated syndrome, 57 patients with noninflammatory neurologic disorders, 127 patients with relapsing-remitting MS, 72 patients with secondary progressive MS, and 113 patients with primary progressive MS. In a training cohort of 217 participants, the researchers assessed how biomarkers differed between the diagnostic categories. The researchers then validated the results in an independent cohort of 214 participants.
One astrocyte-related cluster (MMP7, SERPINA3, GZMA, and CLIC1) and one microglia-related cluster (DSG2 and TNFRSF25) was significantly elevated in all MS subgroups, compared with healthy controls and patients with noninflammatory neurologic disorders.
In addition, these clusters “significantly correlated with clinical measures of disability, CNS tissue destruction, and MS severity,” Dr. Masvekar said.
The microglial cluster was significantly elevated in all MS subgroups, whereas neuronal endothelial, astrocytic, and oligodendroglial biomarker clusters were elevated only in patients with progressive MS.
“Microglial activation is present in all stages of MS, while toxic astrogliosis increases with MS duration, concomitantly with neuronal and oligodendroglial degeneration,” Dr. Masvekar said. “Microglial activation and toxic astrogliosis likely partake in CNS tissue destruction and enhance MS severity.”
This study, which was recently published in Multiple Sclerosis and Related Disorders (2019 Feb;28:34-43), was supported by the intramural research program at NIAID.
jremaly@mdedge.com
SOURCE: Masvekar RR et al. ACTRIMS Forum 2019, Abstract 281.
DALLAS – Patients with multiple sclerosis (MS) have elevated levels of specific clusters of cerebrospinal fluid (CSF) biomarkers related to astrocytes and microglia that correlated with disease severity in a blinded analysis of more than 1,000 proteins from the CSF of more than 400 patients with neuroimmunologic disease and healthy volunteers.
Previous studies have indicated that aberrant activation of astrocytes and microglia underlies disability progression in older patients with MS, but researchers have lacked biomarkers of these processes in living subjects. In a presentation at a meeting held by the Americas Committee for Treatment and Research in Multiple Sclerosis, Ruturaj R. Masvekar, PhD, described developing biomarkers of CNS cell–specific processes and examining how they relate to MS disability progression. Dr. Masvekar, a researcher at the National Institute of Allergy and Infectious Diseases, and his coinvestigators used a modified DNA aptamer assay to measure proteins in the CSF of 431 patients with neuroimmunologic diseases and healthy volunteers, followed by variable cluster analysis and in vitro modeling to define 64 clusters of CSF biomarkers that relate to CNS cell types.
The study included 42 healthy donors, 20 patients with clinically isolated syndrome, 57 patients with noninflammatory neurologic disorders, 127 patients with relapsing-remitting MS, 72 patients with secondary progressive MS, and 113 patients with primary progressive MS. In a training cohort of 217 participants, the researchers assessed how biomarkers differed between the diagnostic categories. The researchers then validated the results in an independent cohort of 214 participants.
One astrocyte-related cluster (MMP7, SERPINA3, GZMA, and CLIC1) and one microglia-related cluster (DSG2 and TNFRSF25) was significantly elevated in all MS subgroups, compared with healthy controls and patients with noninflammatory neurologic disorders.
In addition, these clusters “significantly correlated with clinical measures of disability, CNS tissue destruction, and MS severity,” Dr. Masvekar said.
The microglial cluster was significantly elevated in all MS subgroups, whereas neuronal endothelial, astrocytic, and oligodendroglial biomarker clusters were elevated only in patients with progressive MS.
“Microglial activation is present in all stages of MS, while toxic astrogliosis increases with MS duration, concomitantly with neuronal and oligodendroglial degeneration,” Dr. Masvekar said. “Microglial activation and toxic astrogliosis likely partake in CNS tissue destruction and enhance MS severity.”
This study, which was recently published in Multiple Sclerosis and Related Disorders (2019 Feb;28:34-43), was supported by the intramural research program at NIAID.
jremaly@mdedge.com
SOURCE: Masvekar RR et al. ACTRIMS Forum 2019, Abstract 281.
REPORTING FROM ACTRIMS FORUM 2019














