User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Wegovy scores HFpEF benefits in people with obesity
AMSTERDAM – Adults with heart failure with preserved ejection fraction (HFpEF) but without diabetes showed significant improvements in their heart failure-related symptoms and physical limitations, exercise function, and weight loss when treated with a weight-reducing dose of semaglutide for 52 weeks, compared with placebo, in the randomized STEP-HFpEF trial.
The results, which also showed the treatment’s safety in these patients, “indicate that treatment with semaglutide is a valuable therapeutic approach in the management of patients with HFpEF and obesity,” Mikhail Kosiborod, MD, said at the annual congress of the European Society of Cardiology.
The findings establish semaglutide, a glucagonlike peptide–1 (GLP-1) receptor agonist, as a second class of medication with proven efficacy and safety for people with HFpEF, joining two agents also proven beneficial for people with HFpEF, dapagliflozin (Farxiga) and empagliflozin (Jardiance), both from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors.
When administered at the approved dose for weight loss of 2.4 mg, injected subcutaneously weekly for 52 weeks, semaglutide (Wegovy) produced an average 7.8-point incremental improvement in patients’ scores on the Kansas City Cardiomyopathy Questionnaire (KCCQ), a validated measure of symptoms and functional limitations, compared with controls who received placebo injections, as well as an average incremental weight loss from baseline, compared with placebo, of 10.7%. Both were significant effects, compared with placebo, and clinically meaningful benefits for the study’s two primary endpoints.
Simultaneously with Kosiborod’s report the results also appeared in a report posted online in the New England Journal of Medicine.
A ‘paradigm shift’ for medical weight loss in cardiology
The findings from this study with 529 randomized patients immediately propelled the weight loss formulation of semaglutide into the ranks of agents used to treat and prevent cardiovascular disease events. This evolution in the indications for semaglutide will be driven not only by the STEP-HFpEF results but also by findings from the SELECT trial, which tested the same semaglutide weight-loss dose in people with obesity, established cardiovascular disease, and had positive top-line results for prevention of major cardiovascular adverse events, according to a press release from Novo Nordisk on Aug. 8.
The STEP-HFpEF and SELECT results will trigger “a paradigm shift” for cardiologists, who will now need to consider prescribing a weight-loss medication to many of their patients, agents that until now were not part of the usual pharmacologic toolbox for cardiologists, said Dr. Kosiborod, a cardiologist and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo. This shift will require education to bring the clinical cardiology community on board, he added in an interview.
Given that semaglutide administered at this dose already has a Food and Drug Administration–approved indication for weight loss in people with obesity or overweight plus at least one comorbidity, clinicians could immediately start using the treatment in people with obesity and HFpEF, said Dr. Kosiborod and other cardiologists.
Weekly semaglutide injections “could be considered a treatment option right now” for people with obesity and HFpEF, Dr. Kosiborod said during a press briefing.
Other experts agreed, especially because the STEP-HFpEF results confirmed that weight loss treatment with semaglutide was safe in this population.
‘A terrific win for patients’
The new findings are “a terrific win and game changer for patients with HFpEF,” commented Gregg C. Fonarow, MD, professor and cochief of cardiology at the University of California, Los Angeles, who was not involved with the study.
“The magnitude of improvement in the patient-reported health status scores are large and impressive. These data support clinical use of this agent for individuals with HFpEF with a body mass index of 30 kg/m2, patients who already fall within existing indications,” Dr. Fonarow said in an interview.
“Given the improvements in clinical outcomes in the STEP-HFpEF and SELECT trials, cardiologists should be prescribing these medications to eligible patients without conditions,” he added. “The perception of [semaglutide] needs to shift and be viewed as a component of the comprehensive medical therapies provided to individuals with established cardiovascular disease or HFpEF who also have elevated body mass index to improve their clinical outcomes.”
Historically, cardiologists have had a concern that weight loss was potentially harmful in people with heart failure and that obesity was protective, a phenomenon known as the “obesity paradox,” but the STEP-HFpEF data help disprove that notion, commented Nancy K. Sweitzer, MD, PhD, a heart failure specialist and vice chair of clinical research in the department of medicine at Washington University in St. Louis, who also was not involved in the study.
No signal of an obesity paradox
“There’s been a concern in the heart failure community to use weight-loss strategies in people with heart failure because of this, but this evidence provides a lot of confidence that it’s safe to use this weight loss treatment. The results show that patients feel better and lose weight with no signal of harm,” Dr. Sweitzer said in an interview.
The “encouraging findings” for semaglutide in patients with HFpEF “potentially add a much needed extra option for these patients and provide another upstream treatment for patients with signs of this condition plus a high body mass index,” commented Yigal M. Pinto, MD, PhD, in an editorial that accompanied the published report.
“How these findings translate to hard end points remains to be established and will be important in determining the role of GLP-1 agonism,” wrote Dr. Pinto, a professor and heart failure specialist at Amsterdam University Medical Center.
But Dr. Kosiborod said that the improvement seen in the KCCQ score was itself an important benefit for patients. “Heart failure is defined clinically based on symptoms,” he noted, and results in prior studies documented that patients value improvements in symptoms and physical limitations even more than they value “hard endpoints” such as survival.
The new findings, which indicate that two different and expensive classes of medications are now standard of care for many people with HFpEF and obesity – the SGLT2 inhibitors and the GLP-1 receptor agonist semaglutide – also raise concerns over patient access and affordability, as many U.S. insurers have a history of requiring prior authorization, high copays, or coverage denials for these two medical classes.
But Dr. Sweitzer and Dr. Kosiborod both said that the insurance-coverage climate seems, in just the past couple of years or so, to have dramatically improved, although it’s still not ideal.
Prior authorization hoops have decreased
“We still have prior-authorization hoops to jump through, but I expect these will continue to decrease over time as evidence for clinical benefits [from weight loss] continues to accumulate,” said Dr. Sweitzer.
And “the SELECT data mean that cardiologists will need to become comfortable prescribing GLP-1 receptor agonists,” she added.
“It’s not okay for insurers to say we are not going to cover weight loss medications because it’s a cosmetic indication,” said Dr. Kosiborod. “Obesity appears to be very important in the pathogenesis and progression of heart failure, and if patients derive substantial benefit, they should have access to this treatment.”
The improvements in KCCQ score, as well as in several secondary and exploratory endpoints including a significant reduction in C-reactive protein (an indication of a potent anti-inflammatory effect), an average 20 m increase in 6-minute walk distance, a significant average drop in N-terminal pro-brain natriuretic peptide, and a drop in heart failure hospitalizations or urgent heart failure visits (although the trial was not powered to show differences in clinical events), “were the largest benefits in these outcomes we’ve seen,” compared with any other medical intervention in people with HFpEF, he noted.
“About 80% of U.S. patients with HFpEF have obesity or overweight,” Dr. Kosiborod noted. Using semaglutide on these patients “is an issue of access and insurance coverage. My hope is that these and other data will favorably change this.”
A related trial with a similar design, STEP-HFpEF DM, is still in progress and testing the same semaglutide treatment in adults with HFpEF, obesity, and type 2 diabetes, noted Dr. Kosiborod, who is also lead investigator for that study. He said those results will likely become available before the end of 2023.
The study was funded by Novo Nordisk, the company that markets semaglutide (Wegovy). Dr. Kosiborod has been a consultant and adviser to and has received honoraria from Novo Nordisk. He has also been a consultant to numerous other companies, received research grants from AstraZeneca, Boehringer Ingelheim, and Pfizer, honoraria from AstraZeneca, and is a stockholder in Artera Health and Saghmos Therapeutics. Dr. Fonarow has been a consultant to Abbott, Amgen, AstraZeneca, CHF Solutions, Cytokinetics, Edwards, Janssen, Medtronic, Merck, Novartis, and Regeneron. Dr. Sweitzer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AMSTERDAM – Adults with heart failure with preserved ejection fraction (HFpEF) but without diabetes showed significant improvements in their heart failure-related symptoms and physical limitations, exercise function, and weight loss when treated with a weight-reducing dose of semaglutide for 52 weeks, compared with placebo, in the randomized STEP-HFpEF trial.
The results, which also showed the treatment’s safety in these patients, “indicate that treatment with semaglutide is a valuable therapeutic approach in the management of patients with HFpEF and obesity,” Mikhail Kosiborod, MD, said at the annual congress of the European Society of Cardiology.
The findings establish semaglutide, a glucagonlike peptide–1 (GLP-1) receptor agonist, as a second class of medication with proven efficacy and safety for people with HFpEF, joining two agents also proven beneficial for people with HFpEF, dapagliflozin (Farxiga) and empagliflozin (Jardiance), both from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors.
When administered at the approved dose for weight loss of 2.4 mg, injected subcutaneously weekly for 52 weeks, semaglutide (Wegovy) produced an average 7.8-point incremental improvement in patients’ scores on the Kansas City Cardiomyopathy Questionnaire (KCCQ), a validated measure of symptoms and functional limitations, compared with controls who received placebo injections, as well as an average incremental weight loss from baseline, compared with placebo, of 10.7%. Both were significant effects, compared with placebo, and clinically meaningful benefits for the study’s two primary endpoints.
Simultaneously with Kosiborod’s report the results also appeared in a report posted online in the New England Journal of Medicine.
A ‘paradigm shift’ for medical weight loss in cardiology
The findings from this study with 529 randomized patients immediately propelled the weight loss formulation of semaglutide into the ranks of agents used to treat and prevent cardiovascular disease events. This evolution in the indications for semaglutide will be driven not only by the STEP-HFpEF results but also by findings from the SELECT trial, which tested the same semaglutide weight-loss dose in people with obesity, established cardiovascular disease, and had positive top-line results for prevention of major cardiovascular adverse events, according to a press release from Novo Nordisk on Aug. 8.
The STEP-HFpEF and SELECT results will trigger “a paradigm shift” for cardiologists, who will now need to consider prescribing a weight-loss medication to many of their patients, agents that until now were not part of the usual pharmacologic toolbox for cardiologists, said Dr. Kosiborod, a cardiologist and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo. This shift will require education to bring the clinical cardiology community on board, he added in an interview.
Given that semaglutide administered at this dose already has a Food and Drug Administration–approved indication for weight loss in people with obesity or overweight plus at least one comorbidity, clinicians could immediately start using the treatment in people with obesity and HFpEF, said Dr. Kosiborod and other cardiologists.
Weekly semaglutide injections “could be considered a treatment option right now” for people with obesity and HFpEF, Dr. Kosiborod said during a press briefing.
Other experts agreed, especially because the STEP-HFpEF results confirmed that weight loss treatment with semaglutide was safe in this population.
‘A terrific win for patients’
The new findings are “a terrific win and game changer for patients with HFpEF,” commented Gregg C. Fonarow, MD, professor and cochief of cardiology at the University of California, Los Angeles, who was not involved with the study.
“The magnitude of improvement in the patient-reported health status scores are large and impressive. These data support clinical use of this agent for individuals with HFpEF with a body mass index of 30 kg/m2, patients who already fall within existing indications,” Dr. Fonarow said in an interview.
“Given the improvements in clinical outcomes in the STEP-HFpEF and SELECT trials, cardiologists should be prescribing these medications to eligible patients without conditions,” he added. “The perception of [semaglutide] needs to shift and be viewed as a component of the comprehensive medical therapies provided to individuals with established cardiovascular disease or HFpEF who also have elevated body mass index to improve their clinical outcomes.”
Historically, cardiologists have had a concern that weight loss was potentially harmful in people with heart failure and that obesity was protective, a phenomenon known as the “obesity paradox,” but the STEP-HFpEF data help disprove that notion, commented Nancy K. Sweitzer, MD, PhD, a heart failure specialist and vice chair of clinical research in the department of medicine at Washington University in St. Louis, who also was not involved in the study.
No signal of an obesity paradox
“There’s been a concern in the heart failure community to use weight-loss strategies in people with heart failure because of this, but this evidence provides a lot of confidence that it’s safe to use this weight loss treatment. The results show that patients feel better and lose weight with no signal of harm,” Dr. Sweitzer said in an interview.
The “encouraging findings” for semaglutide in patients with HFpEF “potentially add a much needed extra option for these patients and provide another upstream treatment for patients with signs of this condition plus a high body mass index,” commented Yigal M. Pinto, MD, PhD, in an editorial that accompanied the published report.
“How these findings translate to hard end points remains to be established and will be important in determining the role of GLP-1 agonism,” wrote Dr. Pinto, a professor and heart failure specialist at Amsterdam University Medical Center.
But Dr. Kosiborod said that the improvement seen in the KCCQ score was itself an important benefit for patients. “Heart failure is defined clinically based on symptoms,” he noted, and results in prior studies documented that patients value improvements in symptoms and physical limitations even more than they value “hard endpoints” such as survival.
The new findings, which indicate that two different and expensive classes of medications are now standard of care for many people with HFpEF and obesity – the SGLT2 inhibitors and the GLP-1 receptor agonist semaglutide – also raise concerns over patient access and affordability, as many U.S. insurers have a history of requiring prior authorization, high copays, or coverage denials for these two medical classes.
But Dr. Sweitzer and Dr. Kosiborod both said that the insurance-coverage climate seems, in just the past couple of years or so, to have dramatically improved, although it’s still not ideal.
Prior authorization hoops have decreased
“We still have prior-authorization hoops to jump through, but I expect these will continue to decrease over time as evidence for clinical benefits [from weight loss] continues to accumulate,” said Dr. Sweitzer.
And “the SELECT data mean that cardiologists will need to become comfortable prescribing GLP-1 receptor agonists,” she added.
“It’s not okay for insurers to say we are not going to cover weight loss medications because it’s a cosmetic indication,” said Dr. Kosiborod. “Obesity appears to be very important in the pathogenesis and progression of heart failure, and if patients derive substantial benefit, they should have access to this treatment.”
The improvements in KCCQ score, as well as in several secondary and exploratory endpoints including a significant reduction in C-reactive protein (an indication of a potent anti-inflammatory effect), an average 20 m increase in 6-minute walk distance, a significant average drop in N-terminal pro-brain natriuretic peptide, and a drop in heart failure hospitalizations or urgent heart failure visits (although the trial was not powered to show differences in clinical events), “were the largest benefits in these outcomes we’ve seen,” compared with any other medical intervention in people with HFpEF, he noted.
“About 80% of U.S. patients with HFpEF have obesity or overweight,” Dr. Kosiborod noted. Using semaglutide on these patients “is an issue of access and insurance coverage. My hope is that these and other data will favorably change this.”
A related trial with a similar design, STEP-HFpEF DM, is still in progress and testing the same semaglutide treatment in adults with HFpEF, obesity, and type 2 diabetes, noted Dr. Kosiborod, who is also lead investigator for that study. He said those results will likely become available before the end of 2023.
The study was funded by Novo Nordisk, the company that markets semaglutide (Wegovy). Dr. Kosiborod has been a consultant and adviser to and has received honoraria from Novo Nordisk. He has also been a consultant to numerous other companies, received research grants from AstraZeneca, Boehringer Ingelheim, and Pfizer, honoraria from AstraZeneca, and is a stockholder in Artera Health and Saghmos Therapeutics. Dr. Fonarow has been a consultant to Abbott, Amgen, AstraZeneca, CHF Solutions, Cytokinetics, Edwards, Janssen, Medtronic, Merck, Novartis, and Regeneron. Dr. Sweitzer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AMSTERDAM – Adults with heart failure with preserved ejection fraction (HFpEF) but without diabetes showed significant improvements in their heart failure-related symptoms and physical limitations, exercise function, and weight loss when treated with a weight-reducing dose of semaglutide for 52 weeks, compared with placebo, in the randomized STEP-HFpEF trial.
The results, which also showed the treatment’s safety in these patients, “indicate that treatment with semaglutide is a valuable therapeutic approach in the management of patients with HFpEF and obesity,” Mikhail Kosiborod, MD, said at the annual congress of the European Society of Cardiology.
The findings establish semaglutide, a glucagonlike peptide–1 (GLP-1) receptor agonist, as a second class of medication with proven efficacy and safety for people with HFpEF, joining two agents also proven beneficial for people with HFpEF, dapagliflozin (Farxiga) and empagliflozin (Jardiance), both from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors.
When administered at the approved dose for weight loss of 2.4 mg, injected subcutaneously weekly for 52 weeks, semaglutide (Wegovy) produced an average 7.8-point incremental improvement in patients’ scores on the Kansas City Cardiomyopathy Questionnaire (KCCQ), a validated measure of symptoms and functional limitations, compared with controls who received placebo injections, as well as an average incremental weight loss from baseline, compared with placebo, of 10.7%. Both were significant effects, compared with placebo, and clinically meaningful benefits for the study’s two primary endpoints.
Simultaneously with Kosiborod’s report the results also appeared in a report posted online in the New England Journal of Medicine.
A ‘paradigm shift’ for medical weight loss in cardiology
The findings from this study with 529 randomized patients immediately propelled the weight loss formulation of semaglutide into the ranks of agents used to treat and prevent cardiovascular disease events. This evolution in the indications for semaglutide will be driven not only by the STEP-HFpEF results but also by findings from the SELECT trial, which tested the same semaglutide weight-loss dose in people with obesity, established cardiovascular disease, and had positive top-line results for prevention of major cardiovascular adverse events, according to a press release from Novo Nordisk on Aug. 8.
The STEP-HFpEF and SELECT results will trigger “a paradigm shift” for cardiologists, who will now need to consider prescribing a weight-loss medication to many of their patients, agents that until now were not part of the usual pharmacologic toolbox for cardiologists, said Dr. Kosiborod, a cardiologist and codirector of the Haverty Cardiometabolic Center of Excellence at Saint Luke’s Mid America Heart Institute in Kansas City, Mo. This shift will require education to bring the clinical cardiology community on board, he added in an interview.
Given that semaglutide administered at this dose already has a Food and Drug Administration–approved indication for weight loss in people with obesity or overweight plus at least one comorbidity, clinicians could immediately start using the treatment in people with obesity and HFpEF, said Dr. Kosiborod and other cardiologists.
Weekly semaglutide injections “could be considered a treatment option right now” for people with obesity and HFpEF, Dr. Kosiborod said during a press briefing.
Other experts agreed, especially because the STEP-HFpEF results confirmed that weight loss treatment with semaglutide was safe in this population.
‘A terrific win for patients’
The new findings are “a terrific win and game changer for patients with HFpEF,” commented Gregg C. Fonarow, MD, professor and cochief of cardiology at the University of California, Los Angeles, who was not involved with the study.
“The magnitude of improvement in the patient-reported health status scores are large and impressive. These data support clinical use of this agent for individuals with HFpEF with a body mass index of 30 kg/m2, patients who already fall within existing indications,” Dr. Fonarow said in an interview.
“Given the improvements in clinical outcomes in the STEP-HFpEF and SELECT trials, cardiologists should be prescribing these medications to eligible patients without conditions,” he added. “The perception of [semaglutide] needs to shift and be viewed as a component of the comprehensive medical therapies provided to individuals with established cardiovascular disease or HFpEF who also have elevated body mass index to improve their clinical outcomes.”
Historically, cardiologists have had a concern that weight loss was potentially harmful in people with heart failure and that obesity was protective, a phenomenon known as the “obesity paradox,” but the STEP-HFpEF data help disprove that notion, commented Nancy K. Sweitzer, MD, PhD, a heart failure specialist and vice chair of clinical research in the department of medicine at Washington University in St. Louis, who also was not involved in the study.
No signal of an obesity paradox
“There’s been a concern in the heart failure community to use weight-loss strategies in people with heart failure because of this, but this evidence provides a lot of confidence that it’s safe to use this weight loss treatment. The results show that patients feel better and lose weight with no signal of harm,” Dr. Sweitzer said in an interview.
The “encouraging findings” for semaglutide in patients with HFpEF “potentially add a much needed extra option for these patients and provide another upstream treatment for patients with signs of this condition plus a high body mass index,” commented Yigal M. Pinto, MD, PhD, in an editorial that accompanied the published report.
“How these findings translate to hard end points remains to be established and will be important in determining the role of GLP-1 agonism,” wrote Dr. Pinto, a professor and heart failure specialist at Amsterdam University Medical Center.
But Dr. Kosiborod said that the improvement seen in the KCCQ score was itself an important benefit for patients. “Heart failure is defined clinically based on symptoms,” he noted, and results in prior studies documented that patients value improvements in symptoms and physical limitations even more than they value “hard endpoints” such as survival.
The new findings, which indicate that two different and expensive classes of medications are now standard of care for many people with HFpEF and obesity – the SGLT2 inhibitors and the GLP-1 receptor agonist semaglutide – also raise concerns over patient access and affordability, as many U.S. insurers have a history of requiring prior authorization, high copays, or coverage denials for these two medical classes.
But Dr. Sweitzer and Dr. Kosiborod both said that the insurance-coverage climate seems, in just the past couple of years or so, to have dramatically improved, although it’s still not ideal.
Prior authorization hoops have decreased
“We still have prior-authorization hoops to jump through, but I expect these will continue to decrease over time as evidence for clinical benefits [from weight loss] continues to accumulate,” said Dr. Sweitzer.
And “the SELECT data mean that cardiologists will need to become comfortable prescribing GLP-1 receptor agonists,” she added.
“It’s not okay for insurers to say we are not going to cover weight loss medications because it’s a cosmetic indication,” said Dr. Kosiborod. “Obesity appears to be very important in the pathogenesis and progression of heart failure, and if patients derive substantial benefit, they should have access to this treatment.”
The improvements in KCCQ score, as well as in several secondary and exploratory endpoints including a significant reduction in C-reactive protein (an indication of a potent anti-inflammatory effect), an average 20 m increase in 6-minute walk distance, a significant average drop in N-terminal pro-brain natriuretic peptide, and a drop in heart failure hospitalizations or urgent heart failure visits (although the trial was not powered to show differences in clinical events), “were the largest benefits in these outcomes we’ve seen,” compared with any other medical intervention in people with HFpEF, he noted.
“About 80% of U.S. patients with HFpEF have obesity or overweight,” Dr. Kosiborod noted. Using semaglutide on these patients “is an issue of access and insurance coverage. My hope is that these and other data will favorably change this.”
A related trial with a similar design, STEP-HFpEF DM, is still in progress and testing the same semaglutide treatment in adults with HFpEF, obesity, and type 2 diabetes, noted Dr. Kosiborod, who is also lead investigator for that study. He said those results will likely become available before the end of 2023.
The study was funded by Novo Nordisk, the company that markets semaglutide (Wegovy). Dr. Kosiborod has been a consultant and adviser to and has received honoraria from Novo Nordisk. He has also been a consultant to numerous other companies, received research grants from AstraZeneca, Boehringer Ingelheim, and Pfizer, honoraria from AstraZeneca, and is a stockholder in Artera Health and Saghmos Therapeutics. Dr. Fonarow has been a consultant to Abbott, Amgen, AstraZeneca, CHF Solutions, Cytokinetics, Edwards, Janssen, Medtronic, Merck, Novartis, and Regeneron. Dr. Sweitzer reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
AT THE ESC CONGRESS 2023
It’s not ‘reckless’ to consider Ozempic
A stylish 40-something-year-old walks into my office looking mildly sheepish. She is a well-known actress who was recently panned by the paparazzi for having “too much cellulite” after they illegally photographed her playing with her child on a private beach.
Without a doubt, she will request semaglutide (Ozempic) before long, but first we will need to wade through the morass of social condemnation out there about Ozempic to assure her that she is being neither immoral nor reckless for considering it.
Ozempic is nothing new, people! Endocrinologists have been using this class of medication since Byetta hit the market in 2005. We have had 18 years to make informed risk-benefit analyses.
People are obsessed with the risk for pancreatitis. Any type of weight loss can cause gallstones, and this is what can trigger pancreatitis. Unless you’re the type of person who worries that your balanced Weight Watchers diet is going to cause pancreatitis, you should probably remove this risk from your calculations.
Glucagonlike peptide–1 (GLP-1) receptor agonists are naturally occurring gut hormones that reduce inflammatory cascades and clotting risk. We are not giving a dangerous treatment (e.g., fen-phen) that increases cardiovascular risk – quite the contrary, in fact.
Just because influencers are promoting a product doesn’t mean the product is inherently worthless. One of my patients accused me of prescribing a medication that is the “laughingstock of America.” Try telling that to the scores of cardiologists who send patients to my colleagues and me to start Ozempic to help lower their patients’ risk for stroke and heart attack. Or tell this to my patient who survived an episode of rapid atrial fibrillation and was told by his cardiologist that he definitely would have died if he had not lost 30 pounds from Ozempic in the preceding year.
Sometimes it seems like society has become more judgmental about Ozempic than about plastic surgery for weight loss. If we have to choose between liposuction (which doesn’t reduce visceral fat – the dangerous type of fat) or Ozempic, the latter clearly wins because of its real health benefits.
How does it make any sense to say that this medication should be reserved for patients who already have obesity and type 2 diabetes? Why should we penalize patients who have not yet reached those thresholds by denying access to preventive care? Don’t we constantly hear about how our health care system would be much more efficient if we focused on preventive care and not just treatment?
Some people claim that we have to limit access to this medication because of drug shortages. Thankfully, the United States responds to supply and demand economics and will quickly adjust.
I’ve had more patients than I can possibly number with severe binge eating disorders (resistant to years of therapy and medication) who finally developed healthy relationships with food while taking these types of medications. Mounjaro, I’m talking about you…
I always hear the argument that it is immoral to give these medications to patients with a history of restrictive eating patterns. Although every patient needs to be carefully evaluated, often these medications remove food as both the enemy and primary focus of every waking thought. They allow patients to refocus on other aspects of their lives – such as family, friends, hobbies, work – and regain a sense of purpose. If anyone wants to run a trial on this little hypothesis of mine, please reach out to me.
Okay, I agree you might get a little constipated (most often described by patients as the “rabbit pellet phenomenon”), but it’s a small price to pay, no? I’ll throw in a few prunes with the prescription.
Suffice it to say, I did give my 40-something-year-old patient the medication she desired, and she has a new lease on life (as well as better blood pressure and cholesterol).
A version of this article first appeared on Medscape.com.
A stylish 40-something-year-old walks into my office looking mildly sheepish. She is a well-known actress who was recently panned by the paparazzi for having “too much cellulite” after they illegally photographed her playing with her child on a private beach.
Without a doubt, she will request semaglutide (Ozempic) before long, but first we will need to wade through the morass of social condemnation out there about Ozempic to assure her that she is being neither immoral nor reckless for considering it.
Ozempic is nothing new, people! Endocrinologists have been using this class of medication since Byetta hit the market in 2005. We have had 18 years to make informed risk-benefit analyses.
People are obsessed with the risk for pancreatitis. Any type of weight loss can cause gallstones, and this is what can trigger pancreatitis. Unless you’re the type of person who worries that your balanced Weight Watchers diet is going to cause pancreatitis, you should probably remove this risk from your calculations.
Glucagonlike peptide–1 (GLP-1) receptor agonists are naturally occurring gut hormones that reduce inflammatory cascades and clotting risk. We are not giving a dangerous treatment (e.g., fen-phen) that increases cardiovascular risk – quite the contrary, in fact.
Just because influencers are promoting a product doesn’t mean the product is inherently worthless. One of my patients accused me of prescribing a medication that is the “laughingstock of America.” Try telling that to the scores of cardiologists who send patients to my colleagues and me to start Ozempic to help lower their patients’ risk for stroke and heart attack. Or tell this to my patient who survived an episode of rapid atrial fibrillation and was told by his cardiologist that he definitely would have died if he had not lost 30 pounds from Ozempic in the preceding year.
Sometimes it seems like society has become more judgmental about Ozempic than about plastic surgery for weight loss. If we have to choose between liposuction (which doesn’t reduce visceral fat – the dangerous type of fat) or Ozempic, the latter clearly wins because of its real health benefits.
How does it make any sense to say that this medication should be reserved for patients who already have obesity and type 2 diabetes? Why should we penalize patients who have not yet reached those thresholds by denying access to preventive care? Don’t we constantly hear about how our health care system would be much more efficient if we focused on preventive care and not just treatment?
Some people claim that we have to limit access to this medication because of drug shortages. Thankfully, the United States responds to supply and demand economics and will quickly adjust.
I’ve had more patients than I can possibly number with severe binge eating disorders (resistant to years of therapy and medication) who finally developed healthy relationships with food while taking these types of medications. Mounjaro, I’m talking about you…
I always hear the argument that it is immoral to give these medications to patients with a history of restrictive eating patterns. Although every patient needs to be carefully evaluated, often these medications remove food as both the enemy and primary focus of every waking thought. They allow patients to refocus on other aspects of their lives – such as family, friends, hobbies, work – and regain a sense of purpose. If anyone wants to run a trial on this little hypothesis of mine, please reach out to me.
Okay, I agree you might get a little constipated (most often described by patients as the “rabbit pellet phenomenon”), but it’s a small price to pay, no? I’ll throw in a few prunes with the prescription.
Suffice it to say, I did give my 40-something-year-old patient the medication she desired, and she has a new lease on life (as well as better blood pressure and cholesterol).
A version of this article first appeared on Medscape.com.
A stylish 40-something-year-old walks into my office looking mildly sheepish. She is a well-known actress who was recently panned by the paparazzi for having “too much cellulite” after they illegally photographed her playing with her child on a private beach.
Without a doubt, she will request semaglutide (Ozempic) before long, but first we will need to wade through the morass of social condemnation out there about Ozempic to assure her that she is being neither immoral nor reckless for considering it.
Ozempic is nothing new, people! Endocrinologists have been using this class of medication since Byetta hit the market in 2005. We have had 18 years to make informed risk-benefit analyses.
People are obsessed with the risk for pancreatitis. Any type of weight loss can cause gallstones, and this is what can trigger pancreatitis. Unless you’re the type of person who worries that your balanced Weight Watchers diet is going to cause pancreatitis, you should probably remove this risk from your calculations.
Glucagonlike peptide–1 (GLP-1) receptor agonists are naturally occurring gut hormones that reduce inflammatory cascades and clotting risk. We are not giving a dangerous treatment (e.g., fen-phen) that increases cardiovascular risk – quite the contrary, in fact.
Just because influencers are promoting a product doesn’t mean the product is inherently worthless. One of my patients accused me of prescribing a medication that is the “laughingstock of America.” Try telling that to the scores of cardiologists who send patients to my colleagues and me to start Ozempic to help lower their patients’ risk for stroke and heart attack. Or tell this to my patient who survived an episode of rapid atrial fibrillation and was told by his cardiologist that he definitely would have died if he had not lost 30 pounds from Ozempic in the preceding year.
Sometimes it seems like society has become more judgmental about Ozempic than about plastic surgery for weight loss. If we have to choose between liposuction (which doesn’t reduce visceral fat – the dangerous type of fat) or Ozempic, the latter clearly wins because of its real health benefits.
How does it make any sense to say that this medication should be reserved for patients who already have obesity and type 2 diabetes? Why should we penalize patients who have not yet reached those thresholds by denying access to preventive care? Don’t we constantly hear about how our health care system would be much more efficient if we focused on preventive care and not just treatment?
Some people claim that we have to limit access to this medication because of drug shortages. Thankfully, the United States responds to supply and demand economics and will quickly adjust.
I’ve had more patients than I can possibly number with severe binge eating disorders (resistant to years of therapy and medication) who finally developed healthy relationships with food while taking these types of medications. Mounjaro, I’m talking about you…
I always hear the argument that it is immoral to give these medications to patients with a history of restrictive eating patterns. Although every patient needs to be carefully evaluated, often these medications remove food as both the enemy and primary focus of every waking thought. They allow patients to refocus on other aspects of their lives – such as family, friends, hobbies, work – and regain a sense of purpose. If anyone wants to run a trial on this little hypothesis of mine, please reach out to me.
Okay, I agree you might get a little constipated (most often described by patients as the “rabbit pellet phenomenon”), but it’s a small price to pay, no? I’ll throw in a few prunes with the prescription.
Suffice it to say, I did give my 40-something-year-old patient the medication she desired, and she has a new lease on life (as well as better blood pressure and cholesterol).
A version of this article first appeared on Medscape.com.
Human frailty is a cash cow
Doctor, if you are caring for patients with diabetes, I sure hope you know more about it than I do. The longer I live, it seems, the less I understand.
In a free society, people can do what they want, and that’s great except when it isn’t. That’s why societies develop ethics and even public laws if ethics are not strong enough to protect us from ourselves and others.
Sugar, sugar
When I was growing up in small-town Alabama during the Depression and World War II, we called it sugar diabetes. Eat too much sugar, you got fat; your blood sugar went up, and you spilled sugar into your urine. Diabetes was fairly rare, and so was obesity. Doctors treated it by limiting the intake of sugar (and various sweet foods), along with attempting weight loss. If that didn’t do the trick, insulin injections.
From then until now, note these trends.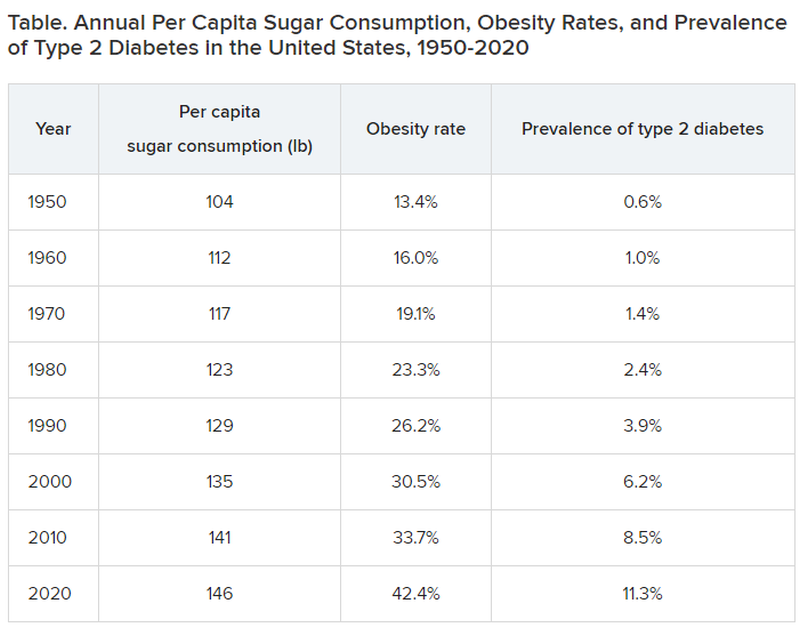
Type 2 diabetes was diagnosed even more infrequently before 1950:
- 1920: 0.2% of the population
- 1930: 0.3% of the population
- 1940: 0.4% of the population
In 2020, although 11.3% of the population was diagnosed with type 2 diabetes, the unknown undiagnosed proportion could be much higher.
Notice a correlation between sugar consumption and prevalence of diabetes? Of course, correlation is not causation, but at the same time, it sure as hell is not negation. Such concordance can be considered hypothesis generating. It may not be true causation, but it’s a good bet when 89% of people with diabetes have overweight or obesity.
What did the entire medical, public health, government, agriculture, nursing, food manufacturing, marketing, advertising, restaurant, and education constituencies do about this as it was happening? They observed, documented, gave lip service, and wrung their hands in public a bit. I do not believe that this is an organized active conspiracy; it would take too many players cooperating over too long a period of time. But it certainly may be a passive conspiracy, and primary care physicians and their patients are trapped.
The proper daily practice of medicine consists of one patient, one physician, one moment, and one decision. Let it be a shared decision, informed by the best evidence and taking cost into consideration. That encounter represents an opportunity, a responsibility, and a conundrum.
Individual health is subsumed under the collective health of the public. As such, a patient’s health is out of the control of both physician and patient; instead, patients are the beneficiaries or victims of the “marketplace.” Humans are frail and easily taken advantage of by the brilliant and highly motivated strategic planning and execution of Big Agriculture, Big Food, Big Pharma, Big Marketing, and Big Money-Driven Medicine and generally failed by Big Government, Big Public Health, Big Education, Big Psychology, and Big Religion.
Rethinking diabetes
Consider diabetes as one of many examples. What a terrific deal for capitalism. then it discovers (invents) long-term, very expensive, compelling treatments to slim us down, with no end in sight, and still without ever understanding the true nature of diabetes.
Gary Taubes’s great new book, “Rethinking Diabetes: What Science Reveals About Diet, Insulin, and Successful Treatments,” is being published by Alfred A. Knopf in early 2024.
It is 404 pages of (dense) text, with 401 numbered references and footnotes, a bibliography of 790 references, alphabetically arranged for easy cross-checking, and a 25-page index.
Remember Mr. Taubes’s earlier definitive historical treatises: “Good Calories, Bad Calories” (2007), “Why We Get Fat” (2010), “The Case Against Sugar” (2016), and “The Case for Keto” (2020)?
This new book is more like “Good Calories, Bad Calories”: long, dense, detailed, definitive, and of great historical reference value, including original research information from other countries in other languages. The author told me that the many early research reference sources were available only in German and that his use of generative artificial intelligence as an assistant researcher was of great value.
Nonphysician author Mr. Taubes uses his deep understanding of science and history to inform his long-honed talents of impartial investigative journalism as he attempts to understand and then explain why after all these years, the medical scientific community still does not have a sound consensus about the essence of diabetes, diet, insulin, and proper prevention and treatment at a level that is actually effective – amazing and so sad.
To signal these evolved and evolving conflicts, the book includes the following chapters:
- “Rise of the Carbohydrate-Rich and Very-Low-Carbohydrate Diets”
- “The Fear of Fat and High-Fat Diets”
- “Insulin and The End of Carbohydrate Restriction and Low Blood Sugar”
Yes, it is difficult. Imagine the bookend segments: “The Nature of Medical Knowledge” and “The Conflicts of Evidence-Based Medicine.” There is also a detailed discussion of good versus bad science spanning three long chapters.
If all that reads like a greatly confused mess to you then you’re beginning to understand. If you are a fan of an unbiased explication of the evolution of understanding the ins and outs of scientific history in richly documented detail, this is a book for you. It’s not a quick nor easy read. And don’t expect to discover whether the newest wonder drugs for weight loss and control of diabetes will be the long-term solution for people with obesity and diabetes worldwide.
Obesity and overweight are major risk factors for type 2 diabetes. About 90% of patients with diabetes have either overweight or obesity. Thus, the complications of these two conditions, which largely overlap, include atherosclerotic cardiovascular disease; myocardial infarction; stroke; hypertension; metabolic syndrome; lower-extremity gangrene; chronic kidney disease; retinopathy; glaucoma; cataracts; disabling osteoarthritis; breast, endometrial, colon, and other cancers; fatty liver; sleep apnea; and peripheral neuropathy. These diseases create a major lucrative business for a wide swathe of medical and surgical specialties, plus hospital, clinic, device, pharmaceutical, and food industries.
In summary, we’ve just been through 40 years of failure to recognize the sugar-elephant in the room and intervene with serious preventive efforts. Forty years of fleshing out both the populace and the American medical-industrial complex (AMIC). Talk about a sweet spot. The only successful long-term treatment of obesity (and with it, diabetes) is prevention. Don’t emphasize losing weight. Focus on preventing excessive weight gain, right now, for the population, beginning with yourselves. Otherwise, we continue openly to perpetuate a terrific deal for the AMIC, a travesty for everyone else. Time for some industrial grade penance and a course correction.
Meanwhile, here we are living out Big Pharma’s dream of a big populace, produced by the agriculture and food industries, enjoyed by capitalism after failures of education, medicine, and public health: a seemingly endless supply of people living with big complications who are ready for big (expensive, new) medications to fix the world’s big health problems.
Dr. Lundberg is editor in chief, Cancer Commons. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Doctor, if you are caring for patients with diabetes, I sure hope you know more about it than I do. The longer I live, it seems, the less I understand.
In a free society, people can do what they want, and that’s great except when it isn’t. That’s why societies develop ethics and even public laws if ethics are not strong enough to protect us from ourselves and others.
Sugar, sugar
When I was growing up in small-town Alabama during the Depression and World War II, we called it sugar diabetes. Eat too much sugar, you got fat; your blood sugar went up, and you spilled sugar into your urine. Diabetes was fairly rare, and so was obesity. Doctors treated it by limiting the intake of sugar (and various sweet foods), along with attempting weight loss. If that didn’t do the trick, insulin injections.
From then until now, note these trends.
Type 2 diabetes was diagnosed even more infrequently before 1950:
- 1920: 0.2% of the population
- 1930: 0.3% of the population
- 1940: 0.4% of the population
In 2020, although 11.3% of the population was diagnosed with type 2 diabetes, the unknown undiagnosed proportion could be much higher.
Notice a correlation between sugar consumption and prevalence of diabetes? Of course, correlation is not causation, but at the same time, it sure as hell is not negation. Such concordance can be considered hypothesis generating. It may not be true causation, but it’s a good bet when 89% of people with diabetes have overweight or obesity.
What did the entire medical, public health, government, agriculture, nursing, food manufacturing, marketing, advertising, restaurant, and education constituencies do about this as it was happening? They observed, documented, gave lip service, and wrung their hands in public a bit. I do not believe that this is an organized active conspiracy; it would take too many players cooperating over too long a period of time. But it certainly may be a passive conspiracy, and primary care physicians and their patients are trapped.
The proper daily practice of medicine consists of one patient, one physician, one moment, and one decision. Let it be a shared decision, informed by the best evidence and taking cost into consideration. That encounter represents an opportunity, a responsibility, and a conundrum.
Individual health is subsumed under the collective health of the public. As such, a patient’s health is out of the control of both physician and patient; instead, patients are the beneficiaries or victims of the “marketplace.” Humans are frail and easily taken advantage of by the brilliant and highly motivated strategic planning and execution of Big Agriculture, Big Food, Big Pharma, Big Marketing, and Big Money-Driven Medicine and generally failed by Big Government, Big Public Health, Big Education, Big Psychology, and Big Religion.
Rethinking diabetes
Consider diabetes as one of many examples. What a terrific deal for capitalism. then it discovers (invents) long-term, very expensive, compelling treatments to slim us down, with no end in sight, and still without ever understanding the true nature of diabetes.
Gary Taubes’s great new book, “Rethinking Diabetes: What Science Reveals About Diet, Insulin, and Successful Treatments,” is being published by Alfred A. Knopf in early 2024.
It is 404 pages of (dense) text, with 401 numbered references and footnotes, a bibliography of 790 references, alphabetically arranged for easy cross-checking, and a 25-page index.
Remember Mr. Taubes’s earlier definitive historical treatises: “Good Calories, Bad Calories” (2007), “Why We Get Fat” (2010), “The Case Against Sugar” (2016), and “The Case for Keto” (2020)?
This new book is more like “Good Calories, Bad Calories”: long, dense, detailed, definitive, and of great historical reference value, including original research information from other countries in other languages. The author told me that the many early research reference sources were available only in German and that his use of generative artificial intelligence as an assistant researcher was of great value.
Nonphysician author Mr. Taubes uses his deep understanding of science and history to inform his long-honed talents of impartial investigative journalism as he attempts to understand and then explain why after all these years, the medical scientific community still does not have a sound consensus about the essence of diabetes, diet, insulin, and proper prevention and treatment at a level that is actually effective – amazing and so sad.
To signal these evolved and evolving conflicts, the book includes the following chapters:
- “Rise of the Carbohydrate-Rich and Very-Low-Carbohydrate Diets”
- “The Fear of Fat and High-Fat Diets”
- “Insulin and The End of Carbohydrate Restriction and Low Blood Sugar”
Yes, it is difficult. Imagine the bookend segments: “The Nature of Medical Knowledge” and “The Conflicts of Evidence-Based Medicine.” There is also a detailed discussion of good versus bad science spanning three long chapters.
If all that reads like a greatly confused mess to you then you’re beginning to understand. If you are a fan of an unbiased explication of the evolution of understanding the ins and outs of scientific history in richly documented detail, this is a book for you. It’s not a quick nor easy read. And don’t expect to discover whether the newest wonder drugs for weight loss and control of diabetes will be the long-term solution for people with obesity and diabetes worldwide.
Obesity and overweight are major risk factors for type 2 diabetes. About 90% of patients with diabetes have either overweight or obesity. Thus, the complications of these two conditions, which largely overlap, include atherosclerotic cardiovascular disease; myocardial infarction; stroke; hypertension; metabolic syndrome; lower-extremity gangrene; chronic kidney disease; retinopathy; glaucoma; cataracts; disabling osteoarthritis; breast, endometrial, colon, and other cancers; fatty liver; sleep apnea; and peripheral neuropathy. These diseases create a major lucrative business for a wide swathe of medical and surgical specialties, plus hospital, clinic, device, pharmaceutical, and food industries.
In summary, we’ve just been through 40 years of failure to recognize the sugar-elephant in the room and intervene with serious preventive efforts. Forty years of fleshing out both the populace and the American medical-industrial complex (AMIC). Talk about a sweet spot. The only successful long-term treatment of obesity (and with it, diabetes) is prevention. Don’t emphasize losing weight. Focus on preventing excessive weight gain, right now, for the population, beginning with yourselves. Otherwise, we continue openly to perpetuate a terrific deal for the AMIC, a travesty for everyone else. Time for some industrial grade penance and a course correction.
Meanwhile, here we are living out Big Pharma’s dream of a big populace, produced by the agriculture and food industries, enjoyed by capitalism after failures of education, medicine, and public health: a seemingly endless supply of people living with big complications who are ready for big (expensive, new) medications to fix the world’s big health problems.
Dr. Lundberg is editor in chief, Cancer Commons. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Doctor, if you are caring for patients with diabetes, I sure hope you know more about it than I do. The longer I live, it seems, the less I understand.
In a free society, people can do what they want, and that’s great except when it isn’t. That’s why societies develop ethics and even public laws if ethics are not strong enough to protect us from ourselves and others.
Sugar, sugar
When I was growing up in small-town Alabama during the Depression and World War II, we called it sugar diabetes. Eat too much sugar, you got fat; your blood sugar went up, and you spilled sugar into your urine. Diabetes was fairly rare, and so was obesity. Doctors treated it by limiting the intake of sugar (and various sweet foods), along with attempting weight loss. If that didn’t do the trick, insulin injections.
From then until now, note these trends.
Type 2 diabetes was diagnosed even more infrequently before 1950:
- 1920: 0.2% of the population
- 1930: 0.3% of the population
- 1940: 0.4% of the population
In 2020, although 11.3% of the population was diagnosed with type 2 diabetes, the unknown undiagnosed proportion could be much higher.
Notice a correlation between sugar consumption and prevalence of diabetes? Of course, correlation is not causation, but at the same time, it sure as hell is not negation. Such concordance can be considered hypothesis generating. It may not be true causation, but it’s a good bet when 89% of people with diabetes have overweight or obesity.
What did the entire medical, public health, government, agriculture, nursing, food manufacturing, marketing, advertising, restaurant, and education constituencies do about this as it was happening? They observed, documented, gave lip service, and wrung their hands in public a bit. I do not believe that this is an organized active conspiracy; it would take too many players cooperating over too long a period of time. But it certainly may be a passive conspiracy, and primary care physicians and their patients are trapped.
The proper daily practice of medicine consists of one patient, one physician, one moment, and one decision. Let it be a shared decision, informed by the best evidence and taking cost into consideration. That encounter represents an opportunity, a responsibility, and a conundrum.
Individual health is subsumed under the collective health of the public. As such, a patient’s health is out of the control of both physician and patient; instead, patients are the beneficiaries or victims of the “marketplace.” Humans are frail and easily taken advantage of by the brilliant and highly motivated strategic planning and execution of Big Agriculture, Big Food, Big Pharma, Big Marketing, and Big Money-Driven Medicine and generally failed by Big Government, Big Public Health, Big Education, Big Psychology, and Big Religion.
Rethinking diabetes
Consider diabetes as one of many examples. What a terrific deal for capitalism. then it discovers (invents) long-term, very expensive, compelling treatments to slim us down, with no end in sight, and still without ever understanding the true nature of diabetes.
Gary Taubes’s great new book, “Rethinking Diabetes: What Science Reveals About Diet, Insulin, and Successful Treatments,” is being published by Alfred A. Knopf in early 2024.
It is 404 pages of (dense) text, with 401 numbered references and footnotes, a bibliography of 790 references, alphabetically arranged for easy cross-checking, and a 25-page index.
Remember Mr. Taubes’s earlier definitive historical treatises: “Good Calories, Bad Calories” (2007), “Why We Get Fat” (2010), “The Case Against Sugar” (2016), and “The Case for Keto” (2020)?
This new book is more like “Good Calories, Bad Calories”: long, dense, detailed, definitive, and of great historical reference value, including original research information from other countries in other languages. The author told me that the many early research reference sources were available only in German and that his use of generative artificial intelligence as an assistant researcher was of great value.
Nonphysician author Mr. Taubes uses his deep understanding of science and history to inform his long-honed talents of impartial investigative journalism as he attempts to understand and then explain why after all these years, the medical scientific community still does not have a sound consensus about the essence of diabetes, diet, insulin, and proper prevention and treatment at a level that is actually effective – amazing and so sad.
To signal these evolved and evolving conflicts, the book includes the following chapters:
- “Rise of the Carbohydrate-Rich and Very-Low-Carbohydrate Diets”
- “The Fear of Fat and High-Fat Diets”
- “Insulin and The End of Carbohydrate Restriction and Low Blood Sugar”
Yes, it is difficult. Imagine the bookend segments: “The Nature of Medical Knowledge” and “The Conflicts of Evidence-Based Medicine.” There is also a detailed discussion of good versus bad science spanning three long chapters.
If all that reads like a greatly confused mess to you then you’re beginning to understand. If you are a fan of an unbiased explication of the evolution of understanding the ins and outs of scientific history in richly documented detail, this is a book for you. It’s not a quick nor easy read. And don’t expect to discover whether the newest wonder drugs for weight loss and control of diabetes will be the long-term solution for people with obesity and diabetes worldwide.
Obesity and overweight are major risk factors for type 2 diabetes. About 90% of patients with diabetes have either overweight or obesity. Thus, the complications of these two conditions, which largely overlap, include atherosclerotic cardiovascular disease; myocardial infarction; stroke; hypertension; metabolic syndrome; lower-extremity gangrene; chronic kidney disease; retinopathy; glaucoma; cataracts; disabling osteoarthritis; breast, endometrial, colon, and other cancers; fatty liver; sleep apnea; and peripheral neuropathy. These diseases create a major lucrative business for a wide swathe of medical and surgical specialties, plus hospital, clinic, device, pharmaceutical, and food industries.
In summary, we’ve just been through 40 years of failure to recognize the sugar-elephant in the room and intervene with serious preventive efforts. Forty years of fleshing out both the populace and the American medical-industrial complex (AMIC). Talk about a sweet spot. The only successful long-term treatment of obesity (and with it, diabetes) is prevention. Don’t emphasize losing weight. Focus on preventing excessive weight gain, right now, for the population, beginning with yourselves. Otherwise, we continue openly to perpetuate a terrific deal for the AMIC, a travesty for everyone else. Time for some industrial grade penance and a course correction.
Meanwhile, here we are living out Big Pharma’s dream of a big populace, produced by the agriculture and food industries, enjoyed by capitalism after failures of education, medicine, and public health: a seemingly endless supply of people living with big complications who are ready for big (expensive, new) medications to fix the world’s big health problems.
Dr. Lundberg is editor in chief, Cancer Commons. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Long COVID leads to greater health risks, research finds
That is the finding of a new study from Washington University in St. Louis. The school distributed a press release about the study, which was published in the journal Nature Medicine.
“Some estimates show more than 90% of the U.S. population has been infected with COVID-19,” Ziyad Al-Aly, chief of research and development at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University, told the St. Louis Post–Dispatch. “Doctors need to realize that their patients could be at risk of these conditions, be it heart disease or lung problems or brain problems – they’re at risk.”
The researchers compared the health records for 138,000 patients who had been infected with those of 6 million who had not. They followed 80 health conditions associated with long COVID for 2 years. They used unnamed records from the VA.
“There was really nothing at all looking at what happens to people at two years after the infection,” Dr. Al-Aly said. “So we decided to take a look.”
Patients who hadn’t been hospitalized within 30 days of infection had a higher risk of death 6 months after recovery, and a higher risk of hospitalization within 18 months. They had higher risk of diabetes, fatigue, joint pain, and other problems compared with people who had not been infected.
“In the nonhospitalized group, risks remained elevated for several problems, for several organ systems,” Dr. Al-Aly said. “For the people who were hospitalized, the risk was ubiquitous across all organ systems. It really spans the gamut with respect to the organ systems that are affected.”
People who had been hospitalized had a 65% greater risk of illnesses after 2 years. Nonhospitalized patients had just a 35% greater risk.
A version of this article first appeared on WebMD.com.
That is the finding of a new study from Washington University in St. Louis. The school distributed a press release about the study, which was published in the journal Nature Medicine.
“Some estimates show more than 90% of the U.S. population has been infected with COVID-19,” Ziyad Al-Aly, chief of research and development at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University, told the St. Louis Post–Dispatch. “Doctors need to realize that their patients could be at risk of these conditions, be it heart disease or lung problems or brain problems – they’re at risk.”
The researchers compared the health records for 138,000 patients who had been infected with those of 6 million who had not. They followed 80 health conditions associated with long COVID for 2 years. They used unnamed records from the VA.
“There was really nothing at all looking at what happens to people at two years after the infection,” Dr. Al-Aly said. “So we decided to take a look.”
Patients who hadn’t been hospitalized within 30 days of infection had a higher risk of death 6 months after recovery, and a higher risk of hospitalization within 18 months. They had higher risk of diabetes, fatigue, joint pain, and other problems compared with people who had not been infected.
“In the nonhospitalized group, risks remained elevated for several problems, for several organ systems,” Dr. Al-Aly said. “For the people who were hospitalized, the risk was ubiquitous across all organ systems. It really spans the gamut with respect to the organ systems that are affected.”
People who had been hospitalized had a 65% greater risk of illnesses after 2 years. Nonhospitalized patients had just a 35% greater risk.
A version of this article first appeared on WebMD.com.
That is the finding of a new study from Washington University in St. Louis. The school distributed a press release about the study, which was published in the journal Nature Medicine.
“Some estimates show more than 90% of the U.S. population has been infected with COVID-19,” Ziyad Al-Aly, chief of research and development at Veterans Affairs St. Louis Health Care System and clinical epidemiologist at Washington University, told the St. Louis Post–Dispatch. “Doctors need to realize that their patients could be at risk of these conditions, be it heart disease or lung problems or brain problems – they’re at risk.”
The researchers compared the health records for 138,000 patients who had been infected with those of 6 million who had not. They followed 80 health conditions associated with long COVID for 2 years. They used unnamed records from the VA.
“There was really nothing at all looking at what happens to people at two years after the infection,” Dr. Al-Aly said. “So we decided to take a look.”
Patients who hadn’t been hospitalized within 30 days of infection had a higher risk of death 6 months after recovery, and a higher risk of hospitalization within 18 months. They had higher risk of diabetes, fatigue, joint pain, and other problems compared with people who had not been infected.
“In the nonhospitalized group, risks remained elevated for several problems, for several organ systems,” Dr. Al-Aly said. “For the people who were hospitalized, the risk was ubiquitous across all organ systems. It really spans the gamut with respect to the organ systems that are affected.”
People who had been hospitalized had a 65% greater risk of illnesses after 2 years. Nonhospitalized patients had just a 35% greater risk.
A version of this article first appeared on WebMD.com.
FROM NATURE MEDICINE
Triglyceride puzzle: Do TG metabolites better predict risk?
Triglyceride levels are a measure of cardiovascular risk and a target for therapy, but a focus on TG levels as a bad guy in CV risk assessments may be missing the mark, a population-based cohort study suggests.
The analysis, based on 30,000 participants in the Copenhagen General Population Study, saw sharply increased risks for all-cause mortality, CV mortality, and cancer mortality over 10 years among those with robust TG metabolism.
Those significant risks, gauged by concentrations of two molecules considered markers of TG metabolic rate, were independent of body mass index (BMI) and a range of other TG-linked risk factors, including plasma TG levels themselves.
All-cause mortality jumped 31% for plasma levels of glycerol in the highest versus lowest quartiles and rose 18% for highest-quartile levels of beta-hydroxybutyrate. In parallel, CV mortality climbed 37% for glycerol and 18% for beta-hydroxybutyrate in the study, published in the European Heart Journal.
The findings “implicate triglyceride metabolic rate as a risk factor for mortality not explained by high plasma triglycerides or high BMI,” the report states. The study, it continues, may be the first to link increased mortality to more active TG metabolism – according to levels of the two biomarkers – in the general population.
The results were “really, really surprising,” senior author Børge G. Nordestgaard, MD, DMSc, said in an interview. They are “completely novel” and “may make people think differently” about TG and mortality risk.
Given their unexpected findings, the group conducted further analyses for evidence that the metabolite-mortality associations weren’t independent. “We tried to stratify them away, but they stayed,” said Dr. Nordestgaard, of the University of Copenhagen.
In a weight-stratified analysis, for example, findings were similar in people with normal weight and with overweight and who were obese, Dr. Nordestgaard observed. “Even in the ones with normal weight by World Health Organization criteria, we saw the same and maybe even stronger relationships” between TG metabolism and mortality.
The study authors were is careful to note the retrospective cohort study’s limitations, but its findings “at most support an association, not causation,” Michael Miller, MD, Hospital of the University of Pennsylvania, Philadelphia, observed in an interview. Therefore, it can’t answer “whether and to what extent glycerol and/or beta-hydroxybutyrate independently contribute to mortality beyond triglyceride levels per se.”
Assessing levels of the two biomarkers “was an interesting way to indirectly assess whole-body TG metabolism,” but they were not fasting levels, said Dr. Miller, who wasn’t part of the study.
Also, the analysis doesn’t account for heparinization and other factors “that artificially raise glycerol levels” and suffers in other ways “from the inherent limitations of residual confounding,” said Dr. Miller, who is also chief of medicine at Corporal Michael J Crescenz VA Medical Center, Philadelphia.
The analysis tracked 30,000 men and women, participants in the much larger Copenhagen General Population Study cohort, for a median of 10.7 years. During that time, 9,897 of them died.
Plasma levels of glycerol and beta-hydroxybutyrate, the study authors noted, were measured using high-throughput nuclear magnetic resonance spectroscopy.
Glycerol levels greater than 80 mcmol/L represented the highest quartile and those less than 52 mcmol/L the lowest quartile. The corresponding beta-hydroxybutyrate quartiles were greater than 154 mcmol/L and less than 91 mcmol/L, respectively.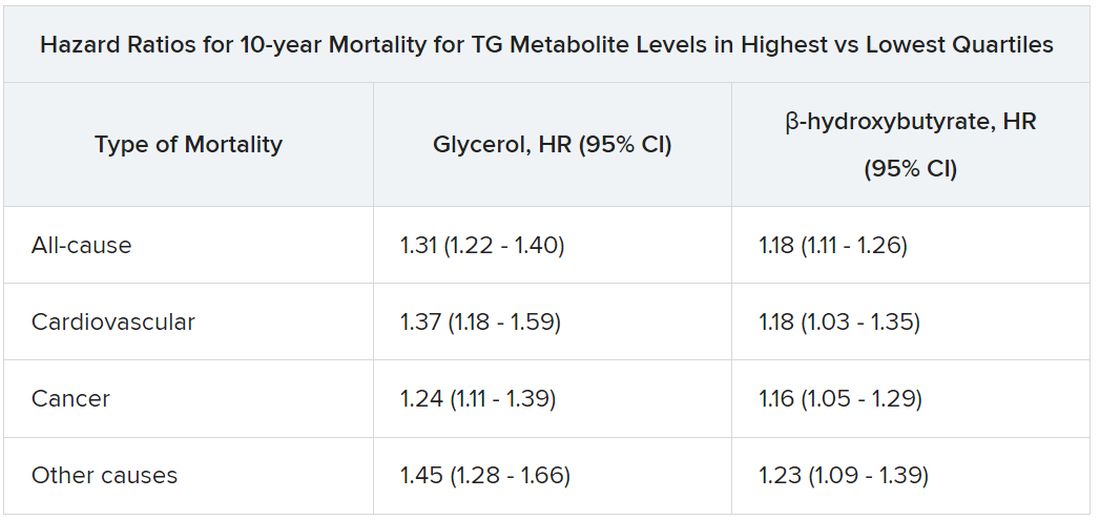
Mortality risks were independent not only of BMI and TG levels but also of age, greater waist circumference, many other standard CV risk factors, chronic obstructive pulmonary disease, diabetes, insulin use, and CV comorbidities and medications.
Dr. Nordestgaard, who also stressed that the findings are only hypothesis generating, speculated that glycerol and beta-hydroxybutyrate could potentially serve as biomarkers for predicting risk or guiding therapy and, indeed, might be amenable to risk-factor modification. “But I have absolutely no data to support that.”
The study was funded by the Independent Research Fund, and by Johan Boserup and Lise Boserups Grant. Dr. Nordestgaard reported consulting for or giving talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amgen, Kowa, Denka, Amarin, Novartis, Novo Nordisk, Esperion, and Silence Therapeutics. The other authors reported no conflicts. Dr. Miller disclosed serving as a scientific adviser for Amarin and 89bio.
A version of this article first appeared on Medscape.com.
Triglyceride levels are a measure of cardiovascular risk and a target for therapy, but a focus on TG levels as a bad guy in CV risk assessments may be missing the mark, a population-based cohort study suggests.
The analysis, based on 30,000 participants in the Copenhagen General Population Study, saw sharply increased risks for all-cause mortality, CV mortality, and cancer mortality over 10 years among those with robust TG metabolism.
Those significant risks, gauged by concentrations of two molecules considered markers of TG metabolic rate, were independent of body mass index (BMI) and a range of other TG-linked risk factors, including plasma TG levels themselves.
All-cause mortality jumped 31% for plasma levels of glycerol in the highest versus lowest quartiles and rose 18% for highest-quartile levels of beta-hydroxybutyrate. In parallel, CV mortality climbed 37% for glycerol and 18% for beta-hydroxybutyrate in the study, published in the European Heart Journal.
The findings “implicate triglyceride metabolic rate as a risk factor for mortality not explained by high plasma triglycerides or high BMI,” the report states. The study, it continues, may be the first to link increased mortality to more active TG metabolism – according to levels of the two biomarkers – in the general population.
The results were “really, really surprising,” senior author Børge G. Nordestgaard, MD, DMSc, said in an interview. They are “completely novel” and “may make people think differently” about TG and mortality risk.
Given their unexpected findings, the group conducted further analyses for evidence that the metabolite-mortality associations weren’t independent. “We tried to stratify them away, but they stayed,” said Dr. Nordestgaard, of the University of Copenhagen.
In a weight-stratified analysis, for example, findings were similar in people with normal weight and with overweight and who were obese, Dr. Nordestgaard observed. “Even in the ones with normal weight by World Health Organization criteria, we saw the same and maybe even stronger relationships” between TG metabolism and mortality.
The study authors were is careful to note the retrospective cohort study’s limitations, but its findings “at most support an association, not causation,” Michael Miller, MD, Hospital of the University of Pennsylvania, Philadelphia, observed in an interview. Therefore, it can’t answer “whether and to what extent glycerol and/or beta-hydroxybutyrate independently contribute to mortality beyond triglyceride levels per se.”
Assessing levels of the two biomarkers “was an interesting way to indirectly assess whole-body TG metabolism,” but they were not fasting levels, said Dr. Miller, who wasn’t part of the study.
Also, the analysis doesn’t account for heparinization and other factors “that artificially raise glycerol levels” and suffers in other ways “from the inherent limitations of residual confounding,” said Dr. Miller, who is also chief of medicine at Corporal Michael J Crescenz VA Medical Center, Philadelphia.
The analysis tracked 30,000 men and women, participants in the much larger Copenhagen General Population Study cohort, for a median of 10.7 years. During that time, 9,897 of them died.
Plasma levels of glycerol and beta-hydroxybutyrate, the study authors noted, were measured using high-throughput nuclear magnetic resonance spectroscopy.
Glycerol levels greater than 80 mcmol/L represented the highest quartile and those less than 52 mcmol/L the lowest quartile. The corresponding beta-hydroxybutyrate quartiles were greater than 154 mcmol/L and less than 91 mcmol/L, respectively.
Mortality risks were independent not only of BMI and TG levels but also of age, greater waist circumference, many other standard CV risk factors, chronic obstructive pulmonary disease, diabetes, insulin use, and CV comorbidities and medications.
Dr. Nordestgaard, who also stressed that the findings are only hypothesis generating, speculated that glycerol and beta-hydroxybutyrate could potentially serve as biomarkers for predicting risk or guiding therapy and, indeed, might be amenable to risk-factor modification. “But I have absolutely no data to support that.”
The study was funded by the Independent Research Fund, and by Johan Boserup and Lise Boserups Grant. Dr. Nordestgaard reported consulting for or giving talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amgen, Kowa, Denka, Amarin, Novartis, Novo Nordisk, Esperion, and Silence Therapeutics. The other authors reported no conflicts. Dr. Miller disclosed serving as a scientific adviser for Amarin and 89bio.
A version of this article first appeared on Medscape.com.
Triglyceride levels are a measure of cardiovascular risk and a target for therapy, but a focus on TG levels as a bad guy in CV risk assessments may be missing the mark, a population-based cohort study suggests.
The analysis, based on 30,000 participants in the Copenhagen General Population Study, saw sharply increased risks for all-cause mortality, CV mortality, and cancer mortality over 10 years among those with robust TG metabolism.
Those significant risks, gauged by concentrations of two molecules considered markers of TG metabolic rate, were independent of body mass index (BMI) and a range of other TG-linked risk factors, including plasma TG levels themselves.
All-cause mortality jumped 31% for plasma levels of glycerol in the highest versus lowest quartiles and rose 18% for highest-quartile levels of beta-hydroxybutyrate. In parallel, CV mortality climbed 37% for glycerol and 18% for beta-hydroxybutyrate in the study, published in the European Heart Journal.
The findings “implicate triglyceride metabolic rate as a risk factor for mortality not explained by high plasma triglycerides or high BMI,” the report states. The study, it continues, may be the first to link increased mortality to more active TG metabolism – according to levels of the two biomarkers – in the general population.
The results were “really, really surprising,” senior author Børge G. Nordestgaard, MD, DMSc, said in an interview. They are “completely novel” and “may make people think differently” about TG and mortality risk.
Given their unexpected findings, the group conducted further analyses for evidence that the metabolite-mortality associations weren’t independent. “We tried to stratify them away, but they stayed,” said Dr. Nordestgaard, of the University of Copenhagen.
In a weight-stratified analysis, for example, findings were similar in people with normal weight and with overweight and who were obese, Dr. Nordestgaard observed. “Even in the ones with normal weight by World Health Organization criteria, we saw the same and maybe even stronger relationships” between TG metabolism and mortality.
The study authors were is careful to note the retrospective cohort study’s limitations, but its findings “at most support an association, not causation,” Michael Miller, MD, Hospital of the University of Pennsylvania, Philadelphia, observed in an interview. Therefore, it can’t answer “whether and to what extent glycerol and/or beta-hydroxybutyrate independently contribute to mortality beyond triglyceride levels per se.”
Assessing levels of the two biomarkers “was an interesting way to indirectly assess whole-body TG metabolism,” but they were not fasting levels, said Dr. Miller, who wasn’t part of the study.
Also, the analysis doesn’t account for heparinization and other factors “that artificially raise glycerol levels” and suffers in other ways “from the inherent limitations of residual confounding,” said Dr. Miller, who is also chief of medicine at Corporal Michael J Crescenz VA Medical Center, Philadelphia.
The analysis tracked 30,000 men and women, participants in the much larger Copenhagen General Population Study cohort, for a median of 10.7 years. During that time, 9,897 of them died.
Plasma levels of glycerol and beta-hydroxybutyrate, the study authors noted, were measured using high-throughput nuclear magnetic resonance spectroscopy.
Glycerol levels greater than 80 mcmol/L represented the highest quartile and those less than 52 mcmol/L the lowest quartile. The corresponding beta-hydroxybutyrate quartiles were greater than 154 mcmol/L and less than 91 mcmol/L, respectively.
Mortality risks were independent not only of BMI and TG levels but also of age, greater waist circumference, many other standard CV risk factors, chronic obstructive pulmonary disease, diabetes, insulin use, and CV comorbidities and medications.
Dr. Nordestgaard, who also stressed that the findings are only hypothesis generating, speculated that glycerol and beta-hydroxybutyrate could potentially serve as biomarkers for predicting risk or guiding therapy and, indeed, might be amenable to risk-factor modification. “But I have absolutely no data to support that.”
The study was funded by the Independent Research Fund, and by Johan Boserup and Lise Boserups Grant. Dr. Nordestgaard reported consulting for or giving talks sponsored by AstraZeneca, Sanofi, Regeneron, Akcea, Amgen, Kowa, Denka, Amarin, Novartis, Novo Nordisk, Esperion, and Silence Therapeutics. The other authors reported no conflicts. Dr. Miller disclosed serving as a scientific adviser for Amarin and 89bio.
A version of this article first appeared on Medscape.com.
FROM THE EUROPEAN HEART JOURNAL
FDA approves new tubeless insulin pump
The product received CE Mark in Europe in 2018 and is now available in 19 markets worldwide. It offers users a choice of bolusing directly from the pump or from a handheld remote-control device. The pump can be detached and reattached without wasting insulin.
The remote-control device also incorporates blood glucose monitoring and bolus advice, although it currently does not integrate with continuous glucose monitoring (CGM) devices.
A Roche spokesperson said in an interview, “For future product generations, we are exploring possibilities to integrate CGM data into the system. Already today, the diabetes manager allows users to manually enter a glucose value that can be used to calculate a bolus. To do so, people with diabetes could use their CGM device of choice in conjunction with the Accu-Chek Solo micropump system.”
Roche will provide an update on next steps for further developments and time lines for launch “in due course,” according to a company statement.
A version of this article appeared on Medscape.com.
The product received CE Mark in Europe in 2018 and is now available in 19 markets worldwide. It offers users a choice of bolusing directly from the pump or from a handheld remote-control device. The pump can be detached and reattached without wasting insulin.
The remote-control device also incorporates blood glucose monitoring and bolus advice, although it currently does not integrate with continuous glucose monitoring (CGM) devices.
A Roche spokesperson said in an interview, “For future product generations, we are exploring possibilities to integrate CGM data into the system. Already today, the diabetes manager allows users to manually enter a glucose value that can be used to calculate a bolus. To do so, people with diabetes could use their CGM device of choice in conjunction with the Accu-Chek Solo micropump system.”
Roche will provide an update on next steps for further developments and time lines for launch “in due course,” according to a company statement.
A version of this article appeared on Medscape.com.
The product received CE Mark in Europe in 2018 and is now available in 19 markets worldwide. It offers users a choice of bolusing directly from the pump or from a handheld remote-control device. The pump can be detached and reattached without wasting insulin.
The remote-control device also incorporates blood glucose monitoring and bolus advice, although it currently does not integrate with continuous glucose monitoring (CGM) devices.
A Roche spokesperson said in an interview, “For future product generations, we are exploring possibilities to integrate CGM data into the system. Already today, the diabetes manager allows users to manually enter a glucose value that can be used to calculate a bolus. To do so, people with diabetes could use their CGM device of choice in conjunction with the Accu-Chek Solo micropump system.”
Roche will provide an update on next steps for further developments and time lines for launch “in due course,” according to a company statement.
A version of this article appeared on Medscape.com.
Can caffeine improve thyroid function?
“Although the causal relationship between caffeine intake and thyroid function requires further verification, as an easily obtainable and widely consumed dietary ingredient, caffeine is a potential candidate for improving thyroid health in people with metabolic disorders,” reported the authors in the study, published in Nutritional Journal.
Caffeine intake, within established healthy ranges, showed a nonlinear association with thyroid levels.
Moderate caffeine intake has been associated with reducing the risk of metabolic disorders in addition to showing some mental health benefits. However, research on its effects on thyroid hormone, which importantly plays a key role in systemic metabolism and neurologic development, is lacking.
To investigate the effects, Yu Zhou, of the Department of Rehabilitation Medicine, School of Health, Fujian Medical University, Fuzhou, China, and colleagues evaluated data from the National Health and Nutrition Examination Survey (NHANES) III 2007-2012 study involving 2,582 participants for whom data were available regarding medical conditions, dietary intake, thyroid function, and demographic background.
The participants were divided into three subgroups based on sex, age, body mass index, hyperglycemia, hypertension, and cardio-cerebral vascular disease (CVD).
Group 1 (n = 208) was the most metabolically unhealthy. Patients in that group had the highest BMI and were of oldest age. In addition, that group had higher rates of hypertension, hyperglycemia, and CVD, but, notably, it had the lowest level of caffeine consumption.
In group 2 (n = 543), all participants were current smokers, and 90.4% had a habit of drinking alcohol. That group also had the highest percentage of men.
Group 3 (n = 1,183) was the most metabolically healthy, with more women, younger age, and lowest BMI. No participants in that group had hyperglycemia, hypertension, or CVD.
Group 1, the most metabolically unhealthy, had the highest serum thyroid-stimulating hormone (TSH) levels. Of note, while participants with thyroid diseases were initially excluded from the analysis, higher TSH levels are predictive of subclinical hypothyroidism or progression to overt hypothyroidism.
Overall, there was no association between caffeine and TSH levels.
However, a subgroup analysis of the groups showed that in group 1, caffeine intake correlated with TSH nonlinearly (P = .0019), with minimal average consumption of caffeine (< 9.97 mg/d). There was an association with slightly higher TSH levels (P = .035) after adjustment for age, sex, race, drink, disease state, micronutrients, and macronutrients.
However, in higher, moderate amounts of caffeine consumption (9.97 – 264.97 mg/d), there was an inverse association, with lower TSH (P = .001).
There was no association between daily caffeine consumption of more than 264.97 mg and TSH levels.
For context, a typical 8-ounce cup of coffee generally contains 80-100 mg of caffeine, and the Food and Drug Administration indicates that 400 mg/d of caffeine is safe for healthy adults.
Group 2 consumed the highest amount of caffeine. Notably, that group had the lowest serum TSH levels of the three groups. There were no significant associations between caffeine consumption and TSH levels in group 2 or group 3.
There were no significant associations between caffeine consumption and levels of serum FT4 or FT3, also linked to thyroid dysfunction, in any of the groups.
The findings show that “caffeine consumption was correlated with serum TSH nonlinearly, and when taken in moderate amounts (9.97-264.97 mg/d), caffeine demonstrated a positive correlation with serum TSH levels in patients with metabolic disorders,” the authors concluded.
Mechanisms?
Caffeine is believed to modulate pituitary hormone secretion, which has been shown to influence the hypothalamic-pituitary-adrenal axis. The authors speculated that caffeine could potentially affect thyroid activity by affecting pituitary function.
“However, the effects of transient and chronic caffeine administration on human thyroid function need to be verified further, and the related mechanisms remain unclear,” they noted.
Commenting on the study, Maik Pietzner, PhD, of the Berlin Institute of Health, noted that an important limitation of the study is that various patient groups were excluded, including those with abnormal TSH levels.
“What makes me wonder is the high number of exclusions and the focus on very specific groups of people. This almost certainly introduces bias, e.g., what is specific to people not reporting coffee consumption,” Dr. Pietzner said.
Furthermore, “we already know that patients with poor metabolic health do also have slight variations in thyroid hormone levels and also have different dietary patterns,” he explained.
“So reverse confounding might occur in which the poor metabolic health is associated with both poor thyroid hormone levels and coffee consumption,” Dr. Pietzner said.
He also noted the “somewhat odd” finding that the group with the highest metabolic disorders had the lowest coffee consumption, yet the highest TSH levels.
“My guess would be that this might also be a chance finding, given that the distribution of TSH values is very skewed, which can have a strong effect in linear regression models,” Dr. Pietzner said.
In general, “the evidence generated by the study is rather weak, but there is good evidence that higher coffee consumption is linked to better metabolic health, although the exact mechanisms is not known, if indeed causal,” Dr. Pietzner added. “Prospective studies are needed to evaluate whether higher coffee consumption indeed lowers the risk for thyroid disease.”
A version of this article first appeared on Medscape.com.
“Although the causal relationship between caffeine intake and thyroid function requires further verification, as an easily obtainable and widely consumed dietary ingredient, caffeine is a potential candidate for improving thyroid health in people with metabolic disorders,” reported the authors in the study, published in Nutritional Journal.
Caffeine intake, within established healthy ranges, showed a nonlinear association with thyroid levels.
Moderate caffeine intake has been associated with reducing the risk of metabolic disorders in addition to showing some mental health benefits. However, research on its effects on thyroid hormone, which importantly plays a key role in systemic metabolism and neurologic development, is lacking.
To investigate the effects, Yu Zhou, of the Department of Rehabilitation Medicine, School of Health, Fujian Medical University, Fuzhou, China, and colleagues evaluated data from the National Health and Nutrition Examination Survey (NHANES) III 2007-2012 study involving 2,582 participants for whom data were available regarding medical conditions, dietary intake, thyroid function, and demographic background.
The participants were divided into three subgroups based on sex, age, body mass index, hyperglycemia, hypertension, and cardio-cerebral vascular disease (CVD).
Group 1 (n = 208) was the most metabolically unhealthy. Patients in that group had the highest BMI and were of oldest age. In addition, that group had higher rates of hypertension, hyperglycemia, and CVD, but, notably, it had the lowest level of caffeine consumption.
In group 2 (n = 543), all participants were current smokers, and 90.4% had a habit of drinking alcohol. That group also had the highest percentage of men.
Group 3 (n = 1,183) was the most metabolically healthy, with more women, younger age, and lowest BMI. No participants in that group had hyperglycemia, hypertension, or CVD.
Group 1, the most metabolically unhealthy, had the highest serum thyroid-stimulating hormone (TSH) levels. Of note, while participants with thyroid diseases were initially excluded from the analysis, higher TSH levels are predictive of subclinical hypothyroidism or progression to overt hypothyroidism.
Overall, there was no association between caffeine and TSH levels.
However, a subgroup analysis of the groups showed that in group 1, caffeine intake correlated with TSH nonlinearly (P = .0019), with minimal average consumption of caffeine (< 9.97 mg/d). There was an association with slightly higher TSH levels (P = .035) after adjustment for age, sex, race, drink, disease state, micronutrients, and macronutrients.
However, in higher, moderate amounts of caffeine consumption (9.97 – 264.97 mg/d), there was an inverse association, with lower TSH (P = .001).
There was no association between daily caffeine consumption of more than 264.97 mg and TSH levels.
For context, a typical 8-ounce cup of coffee generally contains 80-100 mg of caffeine, and the Food and Drug Administration indicates that 400 mg/d of caffeine is safe for healthy adults.
Group 2 consumed the highest amount of caffeine. Notably, that group had the lowest serum TSH levels of the three groups. There were no significant associations between caffeine consumption and TSH levels in group 2 or group 3.
There were no significant associations between caffeine consumption and levels of serum FT4 or FT3, also linked to thyroid dysfunction, in any of the groups.
The findings show that “caffeine consumption was correlated with serum TSH nonlinearly, and when taken in moderate amounts (9.97-264.97 mg/d), caffeine demonstrated a positive correlation with serum TSH levels in patients with metabolic disorders,” the authors concluded.
Mechanisms?
Caffeine is believed to modulate pituitary hormone secretion, which has been shown to influence the hypothalamic-pituitary-adrenal axis. The authors speculated that caffeine could potentially affect thyroid activity by affecting pituitary function.
“However, the effects of transient and chronic caffeine administration on human thyroid function need to be verified further, and the related mechanisms remain unclear,” they noted.
Commenting on the study, Maik Pietzner, PhD, of the Berlin Institute of Health, noted that an important limitation of the study is that various patient groups were excluded, including those with abnormal TSH levels.
“What makes me wonder is the high number of exclusions and the focus on very specific groups of people. This almost certainly introduces bias, e.g., what is specific to people not reporting coffee consumption,” Dr. Pietzner said.
Furthermore, “we already know that patients with poor metabolic health do also have slight variations in thyroid hormone levels and also have different dietary patterns,” he explained.
“So reverse confounding might occur in which the poor metabolic health is associated with both poor thyroid hormone levels and coffee consumption,” Dr. Pietzner said.
He also noted the “somewhat odd” finding that the group with the highest metabolic disorders had the lowest coffee consumption, yet the highest TSH levels.
“My guess would be that this might also be a chance finding, given that the distribution of TSH values is very skewed, which can have a strong effect in linear regression models,” Dr. Pietzner said.
In general, “the evidence generated by the study is rather weak, but there is good evidence that higher coffee consumption is linked to better metabolic health, although the exact mechanisms is not known, if indeed causal,” Dr. Pietzner added. “Prospective studies are needed to evaluate whether higher coffee consumption indeed lowers the risk for thyroid disease.”
A version of this article first appeared on Medscape.com.
“Although the causal relationship between caffeine intake and thyroid function requires further verification, as an easily obtainable and widely consumed dietary ingredient, caffeine is a potential candidate for improving thyroid health in people with metabolic disorders,” reported the authors in the study, published in Nutritional Journal.
Caffeine intake, within established healthy ranges, showed a nonlinear association with thyroid levels.
Moderate caffeine intake has been associated with reducing the risk of metabolic disorders in addition to showing some mental health benefits. However, research on its effects on thyroid hormone, which importantly plays a key role in systemic metabolism and neurologic development, is lacking.
To investigate the effects, Yu Zhou, of the Department of Rehabilitation Medicine, School of Health, Fujian Medical University, Fuzhou, China, and colleagues evaluated data from the National Health and Nutrition Examination Survey (NHANES) III 2007-2012 study involving 2,582 participants for whom data were available regarding medical conditions, dietary intake, thyroid function, and demographic background.
The participants were divided into three subgroups based on sex, age, body mass index, hyperglycemia, hypertension, and cardio-cerebral vascular disease (CVD).
Group 1 (n = 208) was the most metabolically unhealthy. Patients in that group had the highest BMI and were of oldest age. In addition, that group had higher rates of hypertension, hyperglycemia, and CVD, but, notably, it had the lowest level of caffeine consumption.
In group 2 (n = 543), all participants were current smokers, and 90.4% had a habit of drinking alcohol. That group also had the highest percentage of men.
Group 3 (n = 1,183) was the most metabolically healthy, with more women, younger age, and lowest BMI. No participants in that group had hyperglycemia, hypertension, or CVD.
Group 1, the most metabolically unhealthy, had the highest serum thyroid-stimulating hormone (TSH) levels. Of note, while participants with thyroid diseases were initially excluded from the analysis, higher TSH levels are predictive of subclinical hypothyroidism or progression to overt hypothyroidism.
Overall, there was no association between caffeine and TSH levels.
However, a subgroup analysis of the groups showed that in group 1, caffeine intake correlated with TSH nonlinearly (P = .0019), with minimal average consumption of caffeine (< 9.97 mg/d). There was an association with slightly higher TSH levels (P = .035) after adjustment for age, sex, race, drink, disease state, micronutrients, and macronutrients.
However, in higher, moderate amounts of caffeine consumption (9.97 – 264.97 mg/d), there was an inverse association, with lower TSH (P = .001).
There was no association between daily caffeine consumption of more than 264.97 mg and TSH levels.
For context, a typical 8-ounce cup of coffee generally contains 80-100 mg of caffeine, and the Food and Drug Administration indicates that 400 mg/d of caffeine is safe for healthy adults.
Group 2 consumed the highest amount of caffeine. Notably, that group had the lowest serum TSH levels of the three groups. There were no significant associations between caffeine consumption and TSH levels in group 2 or group 3.
There were no significant associations between caffeine consumption and levels of serum FT4 or FT3, also linked to thyroid dysfunction, in any of the groups.
The findings show that “caffeine consumption was correlated with serum TSH nonlinearly, and when taken in moderate amounts (9.97-264.97 mg/d), caffeine demonstrated a positive correlation with serum TSH levels in patients with metabolic disorders,” the authors concluded.
Mechanisms?
Caffeine is believed to modulate pituitary hormone secretion, which has been shown to influence the hypothalamic-pituitary-adrenal axis. The authors speculated that caffeine could potentially affect thyroid activity by affecting pituitary function.
“However, the effects of transient and chronic caffeine administration on human thyroid function need to be verified further, and the related mechanisms remain unclear,” they noted.
Commenting on the study, Maik Pietzner, PhD, of the Berlin Institute of Health, noted that an important limitation of the study is that various patient groups were excluded, including those with abnormal TSH levels.
“What makes me wonder is the high number of exclusions and the focus on very specific groups of people. This almost certainly introduces bias, e.g., what is specific to people not reporting coffee consumption,” Dr. Pietzner said.
Furthermore, “we already know that patients with poor metabolic health do also have slight variations in thyroid hormone levels and also have different dietary patterns,” he explained.
“So reverse confounding might occur in which the poor metabolic health is associated with both poor thyroid hormone levels and coffee consumption,” Dr. Pietzner said.
He also noted the “somewhat odd” finding that the group with the highest metabolic disorders had the lowest coffee consumption, yet the highest TSH levels.
“My guess would be that this might also be a chance finding, given that the distribution of TSH values is very skewed, which can have a strong effect in linear regression models,” Dr. Pietzner said.
In general, “the evidence generated by the study is rather weak, but there is good evidence that higher coffee consumption is linked to better metabolic health, although the exact mechanisms is not known, if indeed causal,” Dr. Pietzner added. “Prospective studies are needed to evaluate whether higher coffee consumption indeed lowers the risk for thyroid disease.”
A version of this article first appeared on Medscape.com.
FROM NUTRITIONAL JOURNAL
Type 1 diabetes management improves as technology advances
Significant reductions in hemoglobin A1c have occurred over time among adults with type 1 diabetes as their use of diabetes technology has increased, yet there is still room for improvement, new data suggest.
The new findings are from a study involving patients at the Barbara Davis Center for Diabetes Adult Clinic between Jan. 1, 2014, and Dec. 31, 2021. They show that as technology use has increased, A1c levels have dropped in parallel. Moreover, progression from use of stand-alone continuous glucose monitors (CGMs) to automated insulin delivery systems (AIDs), which comprise insulin pumps and connected CGMs, furthered that progress.
The findings “are in agreement with American Diabetes Association standards of care, and recent international consensus recommending CGM and AID for most people with type 1 diabetes, and early initiation of diabetes technology from the onset of type 1 diabetes,” write Kagan E. Karakus, MD, of the University of Colorado’s Barbara Davis Center, Aurora, and colleagues in the article, which was published online in Diabetes Care.
“It’s very rewarding to us. We can see clearly that the uptake is going up and the A1c is dropping,” lead author Viral N. Shah, MD, of the Barbara Davis Center, told this news organization.
On the flip side, A1c levels rose significantly over the study period among nonusers of technology. “We cannot rule out provider bias for not prescribing diabetes technology among those with higher A1c or from disadvantaged socioeconomic backgrounds,” Dr. Karakus and colleagues write.
Also of note, even with use of the most advanced AID systems available during the study period, just under half of patients were still not achieving A1c levels below 7%. “The technology helps, but it’s not perfect,” Dr. Shah observed.
This study is the first to examine the relationship of A1c with technology use over time, in contrast to prior cross-sectional studies. “The intention here was to look at the landscape over a decade,” Dr. Shah said.
As overall use of technology use rose, A1c levels fell
The analysis included data for 4,174 unique patients (mean number of patients, 1,988/yr); 15,903 clinic visits were included over the 8-year study period. Technology use was defined as CGM use without an AID system or with an AID system.
Over the study period, diabetes technology use increased from 26.9% to 82.7% of the clinic population (P < .001). At the same time, the overall proportion patients who achieved the A1c goal of less than 7% increased from 32.3% to 41.7%, while the mean A1c level dropped from 7.7% to 7.5% (P < .001).
But among the technology nonusers, A1c rose from 7.85% in 2014 to 8.4% in 2021 (P < .001).
Regardless of diabetes technology use, White patients (about 80% of the total study population) had significantly lower A1c than non-White patients (7.5% vs. 7.7% for technology users [P = .02]; 8.0% vs. 8.3% for nontechnology users [P < .001]).
The non-White group was too small to enable the researchers to break down the data by technology type. Nonetheless, Dr. Shah said, “As a clinician, I can say that the penetration of diabetes technology in non-White populations remains low. These are also the people more vulnerable for socioeconomic and psychosocial reasons.”
The A1c increase among technology nonusers may be a result of a statistical artifact, as the number of those individuals was much lower in 2021 than in 2014. It’s possible that those remaining individuals have exceedingly high A1c levels, bringing the average up. “It’s still not good, though,” Dr. Shah said.
The more technology, the lower the A1c
Over the study period, the proportion of stand-alone CGM users rose from 26.9% to 44.1%, while use of AIDs rose from 0% in 2014 and 2015 to 38.6% in 2021. The latter group included patients who used first-generation Medtronic 670G and 770G devices and second-generation Tandem t:slim X2 with Control-IQ devices.
Between 2017 and 2021, AIDs users had significantly lower A1c levels than nontechnology users: 7.4% vs. 8.1% in 2017, and 7.3% vs. 8.4% in 2021 (P < .001 for every year). CGM users also had significantly lower A1c levels than nonusers at all time points (P < .001 per year).
The proportions achieving an A1c less than 7% differed significantly across users of CGMs, AIDs, and no technology (P < .01 for all years). In 2021, the percentage of people who achieved an A1c less than 7% were 50.9% with AIDs and 44.1% for CGMs vs, just 15.2% with no technology.
Work to be done: Why aren’t more achieving < 7% with AIDs?
Asked why only slightly more than half of patients who used AIDs achieved A1c levels below 7%, Dr. Shah listed three possibilities:
First, the 7% goal doesn’t apply to everyone with type 1 diabetes, including those with multiple comorbidities or with short life expectancy, for whom the recommended goal is 7.5%-8.0% to prevent hypoglycemia. “We didn’t separate out patients by A1c goals. If we add that, the number might go up,” Dr. Shah said.
Second, AID technology is continually improving, but it’s not perfect. Users still must enter carbohydrate counts and signal the devices for exercise, which can lead to errors. “It’s a wonderful technology for overnight control, but still, during the daytime, there are so many factors with the user interface and how much a person is engaged with the technology,” Dr. Shah explained.
Third, he said, “Unfortunately, obesity is increasing in type 1 diabetes, and insulin doses are increasing. Higher BMI [body mass index] and more insulin resistance can mean higher A1c. I really think for many patients, we probably will need an adjunct therapy, such as an SGLT2 [sodium-glucose cotransporter-2] inhibitor or a GLP-1 [glucagonlike peptide-1] agonist, even though they’re not approved in type 1 diabetes, for both glycemic and metabolic control including weight. I think that’s another missing piece.”
He also pointed out, “If someone has an A1c of 7.5%, I don’t expect a huge change. But if they’re at 10%, a drop to 8% is a huge change.”
Overall, Dr. Shah said, the news from the study is good. “In the past, only 30% were achieving an A1c less than 7%. Now we’re 20% above that. ... It’s a glass half full.”
Dr. Karakus has disclosed no relevant financial relationships. Dr. Shah has received, through the University of Colorado, research support from Novo Nordisk, Insulet, Tandem Diabetes, and Dexcom, and honoraria from Medscape, Lifescan, Novo Nordisk, and DKSH Singapore for advisory board attendance and from Insulet and Dexcom for speaking engagements.
A version of this article first appeared on Medscape.com.
Significant reductions in hemoglobin A1c have occurred over time among adults with type 1 diabetes as their use of diabetes technology has increased, yet there is still room for improvement, new data suggest.
The new findings are from a study involving patients at the Barbara Davis Center for Diabetes Adult Clinic between Jan. 1, 2014, and Dec. 31, 2021. They show that as technology use has increased, A1c levels have dropped in parallel. Moreover, progression from use of stand-alone continuous glucose monitors (CGMs) to automated insulin delivery systems (AIDs), which comprise insulin pumps and connected CGMs, furthered that progress.
The findings “are in agreement with American Diabetes Association standards of care, and recent international consensus recommending CGM and AID for most people with type 1 diabetes, and early initiation of diabetes technology from the onset of type 1 diabetes,” write Kagan E. Karakus, MD, of the University of Colorado’s Barbara Davis Center, Aurora, and colleagues in the article, which was published online in Diabetes Care.
“It’s very rewarding to us. We can see clearly that the uptake is going up and the A1c is dropping,” lead author Viral N. Shah, MD, of the Barbara Davis Center, told this news organization.
On the flip side, A1c levels rose significantly over the study period among nonusers of technology. “We cannot rule out provider bias for not prescribing diabetes technology among those with higher A1c or from disadvantaged socioeconomic backgrounds,” Dr. Karakus and colleagues write.
Also of note, even with use of the most advanced AID systems available during the study period, just under half of patients were still not achieving A1c levels below 7%. “The technology helps, but it’s not perfect,” Dr. Shah observed.
This study is the first to examine the relationship of A1c with technology use over time, in contrast to prior cross-sectional studies. “The intention here was to look at the landscape over a decade,” Dr. Shah said.
As overall use of technology use rose, A1c levels fell
The analysis included data for 4,174 unique patients (mean number of patients, 1,988/yr); 15,903 clinic visits were included over the 8-year study period. Technology use was defined as CGM use without an AID system or with an AID system.
Over the study period, diabetes technology use increased from 26.9% to 82.7% of the clinic population (P < .001). At the same time, the overall proportion patients who achieved the A1c goal of less than 7% increased from 32.3% to 41.7%, while the mean A1c level dropped from 7.7% to 7.5% (P < .001).
But among the technology nonusers, A1c rose from 7.85% in 2014 to 8.4% in 2021 (P < .001).
Regardless of diabetes technology use, White patients (about 80% of the total study population) had significantly lower A1c than non-White patients (7.5% vs. 7.7% for technology users [P = .02]; 8.0% vs. 8.3% for nontechnology users [P < .001]).
The non-White group was too small to enable the researchers to break down the data by technology type. Nonetheless, Dr. Shah said, “As a clinician, I can say that the penetration of diabetes technology in non-White populations remains low. These are also the people more vulnerable for socioeconomic and psychosocial reasons.”
The A1c increase among technology nonusers may be a result of a statistical artifact, as the number of those individuals was much lower in 2021 than in 2014. It’s possible that those remaining individuals have exceedingly high A1c levels, bringing the average up. “It’s still not good, though,” Dr. Shah said.
The more technology, the lower the A1c
Over the study period, the proportion of stand-alone CGM users rose from 26.9% to 44.1%, while use of AIDs rose from 0% in 2014 and 2015 to 38.6% in 2021. The latter group included patients who used first-generation Medtronic 670G and 770G devices and second-generation Tandem t:slim X2 with Control-IQ devices.
Between 2017 and 2021, AIDs users had significantly lower A1c levels than nontechnology users: 7.4% vs. 8.1% in 2017, and 7.3% vs. 8.4% in 2021 (P < .001 for every year). CGM users also had significantly lower A1c levels than nonusers at all time points (P < .001 per year).
The proportions achieving an A1c less than 7% differed significantly across users of CGMs, AIDs, and no technology (P < .01 for all years). In 2021, the percentage of people who achieved an A1c less than 7% were 50.9% with AIDs and 44.1% for CGMs vs, just 15.2% with no technology.
Work to be done: Why aren’t more achieving < 7% with AIDs?
Asked why only slightly more than half of patients who used AIDs achieved A1c levels below 7%, Dr. Shah listed three possibilities:
First, the 7% goal doesn’t apply to everyone with type 1 diabetes, including those with multiple comorbidities or with short life expectancy, for whom the recommended goal is 7.5%-8.0% to prevent hypoglycemia. “We didn’t separate out patients by A1c goals. If we add that, the number might go up,” Dr. Shah said.
Second, AID technology is continually improving, but it’s not perfect. Users still must enter carbohydrate counts and signal the devices for exercise, which can lead to errors. “It’s a wonderful technology for overnight control, but still, during the daytime, there are so many factors with the user interface and how much a person is engaged with the technology,” Dr. Shah explained.
Third, he said, “Unfortunately, obesity is increasing in type 1 diabetes, and insulin doses are increasing. Higher BMI [body mass index] and more insulin resistance can mean higher A1c. I really think for many patients, we probably will need an adjunct therapy, such as an SGLT2 [sodium-glucose cotransporter-2] inhibitor or a GLP-1 [glucagonlike peptide-1] agonist, even though they’re not approved in type 1 diabetes, for both glycemic and metabolic control including weight. I think that’s another missing piece.”
He also pointed out, “If someone has an A1c of 7.5%, I don’t expect a huge change. But if they’re at 10%, a drop to 8% is a huge change.”
Overall, Dr. Shah said, the news from the study is good. “In the past, only 30% were achieving an A1c less than 7%. Now we’re 20% above that. ... It’s a glass half full.”
Dr. Karakus has disclosed no relevant financial relationships. Dr. Shah has received, through the University of Colorado, research support from Novo Nordisk, Insulet, Tandem Diabetes, and Dexcom, and honoraria from Medscape, Lifescan, Novo Nordisk, and DKSH Singapore for advisory board attendance and from Insulet and Dexcom for speaking engagements.
A version of this article first appeared on Medscape.com.
Significant reductions in hemoglobin A1c have occurred over time among adults with type 1 diabetes as their use of diabetes technology has increased, yet there is still room for improvement, new data suggest.
The new findings are from a study involving patients at the Barbara Davis Center for Diabetes Adult Clinic between Jan. 1, 2014, and Dec. 31, 2021. They show that as technology use has increased, A1c levels have dropped in parallel. Moreover, progression from use of stand-alone continuous glucose monitors (CGMs) to automated insulin delivery systems (AIDs), which comprise insulin pumps and connected CGMs, furthered that progress.
The findings “are in agreement with American Diabetes Association standards of care, and recent international consensus recommending CGM and AID for most people with type 1 diabetes, and early initiation of diabetes technology from the onset of type 1 diabetes,” write Kagan E. Karakus, MD, of the University of Colorado’s Barbara Davis Center, Aurora, and colleagues in the article, which was published online in Diabetes Care.
“It’s very rewarding to us. We can see clearly that the uptake is going up and the A1c is dropping,” lead author Viral N. Shah, MD, of the Barbara Davis Center, told this news organization.
On the flip side, A1c levels rose significantly over the study period among nonusers of technology. “We cannot rule out provider bias for not prescribing diabetes technology among those with higher A1c or from disadvantaged socioeconomic backgrounds,” Dr. Karakus and colleagues write.
Also of note, even with use of the most advanced AID systems available during the study period, just under half of patients were still not achieving A1c levels below 7%. “The technology helps, but it’s not perfect,” Dr. Shah observed.
This study is the first to examine the relationship of A1c with technology use over time, in contrast to prior cross-sectional studies. “The intention here was to look at the landscape over a decade,” Dr. Shah said.
As overall use of technology use rose, A1c levels fell
The analysis included data for 4,174 unique patients (mean number of patients, 1,988/yr); 15,903 clinic visits were included over the 8-year study period. Technology use was defined as CGM use without an AID system or with an AID system.
Over the study period, diabetes technology use increased from 26.9% to 82.7% of the clinic population (P < .001). At the same time, the overall proportion patients who achieved the A1c goal of less than 7% increased from 32.3% to 41.7%, while the mean A1c level dropped from 7.7% to 7.5% (P < .001).
But among the technology nonusers, A1c rose from 7.85% in 2014 to 8.4% in 2021 (P < .001).
Regardless of diabetes technology use, White patients (about 80% of the total study population) had significantly lower A1c than non-White patients (7.5% vs. 7.7% for technology users [P = .02]; 8.0% vs. 8.3% for nontechnology users [P < .001]).
The non-White group was too small to enable the researchers to break down the data by technology type. Nonetheless, Dr. Shah said, “As a clinician, I can say that the penetration of diabetes technology in non-White populations remains low. These are also the people more vulnerable for socioeconomic and psychosocial reasons.”
The A1c increase among technology nonusers may be a result of a statistical artifact, as the number of those individuals was much lower in 2021 than in 2014. It’s possible that those remaining individuals have exceedingly high A1c levels, bringing the average up. “It’s still not good, though,” Dr. Shah said.
The more technology, the lower the A1c
Over the study period, the proportion of stand-alone CGM users rose from 26.9% to 44.1%, while use of AIDs rose from 0% in 2014 and 2015 to 38.6% in 2021. The latter group included patients who used first-generation Medtronic 670G and 770G devices and second-generation Tandem t:slim X2 with Control-IQ devices.
Between 2017 and 2021, AIDs users had significantly lower A1c levels than nontechnology users: 7.4% vs. 8.1% in 2017, and 7.3% vs. 8.4% in 2021 (P < .001 for every year). CGM users also had significantly lower A1c levels than nonusers at all time points (P < .001 per year).
The proportions achieving an A1c less than 7% differed significantly across users of CGMs, AIDs, and no technology (P < .01 for all years). In 2021, the percentage of people who achieved an A1c less than 7% were 50.9% with AIDs and 44.1% for CGMs vs, just 15.2% with no technology.
Work to be done: Why aren’t more achieving < 7% with AIDs?
Asked why only slightly more than half of patients who used AIDs achieved A1c levels below 7%, Dr. Shah listed three possibilities:
First, the 7% goal doesn’t apply to everyone with type 1 diabetes, including those with multiple comorbidities or with short life expectancy, for whom the recommended goal is 7.5%-8.0% to prevent hypoglycemia. “We didn’t separate out patients by A1c goals. If we add that, the number might go up,” Dr. Shah said.
Second, AID technology is continually improving, but it’s not perfect. Users still must enter carbohydrate counts and signal the devices for exercise, which can lead to errors. “It’s a wonderful technology for overnight control, but still, during the daytime, there are so many factors with the user interface and how much a person is engaged with the technology,” Dr. Shah explained.
Third, he said, “Unfortunately, obesity is increasing in type 1 diabetes, and insulin doses are increasing. Higher BMI [body mass index] and more insulin resistance can mean higher A1c. I really think for many patients, we probably will need an adjunct therapy, such as an SGLT2 [sodium-glucose cotransporter-2] inhibitor or a GLP-1 [glucagonlike peptide-1] agonist, even though they’re not approved in type 1 diabetes, for both glycemic and metabolic control including weight. I think that’s another missing piece.”
He also pointed out, “If someone has an A1c of 7.5%, I don’t expect a huge change. But if they’re at 10%, a drop to 8% is a huge change.”
Overall, Dr. Shah said, the news from the study is good. “In the past, only 30% were achieving an A1c less than 7%. Now we’re 20% above that. ... It’s a glass half full.”
Dr. Karakus has disclosed no relevant financial relationships. Dr. Shah has received, through the University of Colorado, research support from Novo Nordisk, Insulet, Tandem Diabetes, and Dexcom, and honoraria from Medscape, Lifescan, Novo Nordisk, and DKSH Singapore for advisory board attendance and from Insulet and Dexcom for speaking engagements.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE
Artificial sweeteners no help for weight loss: Review
It also shows evidence that these products are not beneficial for controlling excess weight.
Francisco Gómez-Delgado, MD, PhD, and Pablo Pérez-Martínez, MD, PhD, are members of the Spanish Society of Arteriosclerosis and of the Spanish Society of Internal Medicine. They have coordinated an updated review of the leading scientific evidence surrounding artificial sweeteners: evidence showing that far from positively affecting our health, they have “negative effects for the cardiometabolic system.”
The paper, published in Current Opinion in Cardiology, delves into the consumption of these sweeteners and their negative influence on the development of obesity and of several of the most important cardiometabolic risk factors (hypertension, dyslipidemia, and diabetes).
Globalization and the increase in consumption of ultraprocessed foods have led to a need for greater knowledge on the health impacts of certain nutrients such as artificial sweeteners (nutritive and nonnutritive). This review aims to analyze their role and their effect on cardiometabolic and cardiovascular disease risk.
Cardiovascular risk
The detrimental effects of a high-calorie, high-sugar diet have been well established. For this reason, health authorities recommend limiting sugar consumption. The recommendation has led the food industry to develop different artificial sweeteners with specific properties, such as flavor and stability (nutritive artificial sweeteners), and others aimed at limiting sugar in the diet (nonnutritive artificial sweeteners). Recent evidence explores the influence of these two types of artificial sweeteners on cardiovascular disease risk through risk factors such as obesity and type 2 diabetes, among others.
Initially, the consumption of artificial sweeteners was presented as an alternative for reducing calorie intake in the diet as an option for people with excess weight and obesity. However, as this paper explains, the consumption of these artificial sweeteners favors weight gain because of neuroendocrine mechanisms related to satiety that are abnormally activated when artificial sweeteners are consumed.
Weight gain
On the other hand, evidence shows that consuming artificial sweeteners does not encourage weight loss. “Quite the contrary,” Dr. Pérez-Martínez, scientific director at the Maimonides Biomedical Research Institute and internist at the University Hospital Reina Sofia, both in Córdoba, told this news organization. “There is evidence showing weight gain resulting from the effect that artificial sweetener consumption has at the neurohormonal level by altering the mechanisms involved in regulating the feeling of satiety.”
However, on the basis of current evidence, sugar cannot be claimed to be less harmful. “What we do know is that in both cases, we should reduce or remove them from our diets and replace them with other healthier alternatives for weight management, such as eating plant-based products or being physically active.”
Confronting ignorance
Nonetheless, these recommendations are conditional, “because the weight of the evidence is not extremely high, since there have not been a whole lot of studies. All nutritional studies must be viewed with caution,” Manuel Anguita, MD, PhD, said in an interview. Dr. Anguita is department head of clinical cardiology at the University Hospital Reina Sofia in Córdoba and past president of the Spanish Society of Cardiology.
“It’s something that should be included within the medical record when you’re assessing cardiovascular risk. In addition to identifying patients who use artificial sweeteners, it’s especially important to emphasize that it’s not an appropriate recommendation for weight management.” Healthier measures include moderate exercise and the Mediterranean diet.
Explaining why this research is valuable, he said, “It’s generally useful because there’s ignorance not only in the population but among physicians as well [about] these negative effects of sweeteners.”
Diabetes and metabolic syndrome
Artificial sweeteners cause significant disruptions in the endocrine system, leading our metabolism to function abnormally. The review revealed that consuming artificial sweeteners raises the risk for type 2 diabetes by between 18% and 24% and raises the risk for metabolic syndrome by up to 44%.
Dr. Gómez-Delgado, an internal medicine specialist at the University Hospital of Jaen in Spain and first author of the study, discussed the deleterious effects of sweeteners on metabolism. “On one hand, neurohormonal disorders impact appetite, and the feeling of satiety is abnormally delayed.” On the other hand, “they induce excessive insulin secretion in the pancreas,” which in the long run, encourages metabolic disorders that lead to diabetes. Ultimately, this process produces what we know as “dysbiosis, since our microbiota is unable to process these artificial sweeteners.” Dysbiosis triggers specific pathophysiologic processes that negatively affect cardiometabolic and cardiovascular systems.
No differences
Regarding the type of sweetener, Dr. Gómez-Delgado noted that currently available studies assess the consumption of special dietary products that, in most cases, include various types of artificial sweeteners. “So, it’s not possible to define specific differences between them as to how they impact our health.” Additional studies are needed to confirm this effect at the cardiometabolic level and to analyze the different types of artificial sweeteners individually.
“There’s enough evidence to confirm that consuming artificial sweeteners negatively interferes with our metabolism – especially glucose metabolism – and increases the risk of developing diabetes,” said Dr. Gómez-Delgado.
High-sodium drinks
When it comes to the influence of artificial sweeteners on hypertension, “there is no single explanation. The World Health Organization already discussed this issue 4-5 years ago, not only due to their carcinogenic risk, but also due to this cardiovascular risk in terms of a lack of control of obesity, diabetes, and hypertension,” said Dr. Anguita.
Another important point “is that this is not in reference to the sweeteners themselves, but to soft drinks containing those components, which is where we have more studies,” he added. There are two factors explaining this increase in hypertension, which poses a problem at the population level, with medium- to long-term follow-up. “The sugary beverages that we mentioned have a higher sodium content. That is, the sweeteners add this element, which is a factor that’s directly linked to the increase in blood pressure levels.” Another factor that can influence blood pressure is “the increase in insulin secretion that has been described as resulting from sweeteners. In the medium and long term, this is associated with increased blood pressure levels.”
Cardiovascular risk factor?
Are artificial sweeteners considered to be a new cardiovascular risk factor? “What they really do is increase the incidence of the other classic risk factors,” including obesity, said Dr. Anguita. It has been shown that artificial sweeteners don’t reduce obesity when used continuously. Nonetheless, “there is still not enough evidence to view it in the same light as the classic risk factors,” added Dr. Anguita. However, it is a factor that can clearly worsen the control of the other factors. Therefore, “it’s appropriate to sound an alarm and explain that it’s not the best way to lose weight; there are many other healthier choices.”
“We need more robust evidence to take a clear position on the use of this type of sweetener and its detrimental effect on health. Meanwhile, it would be ideal to limit their consumption or even avoid adding artificial sweeteners to coffee or teas,” added Dr. Pérez-Martínez.
Regulate consumption
Dr. Pérez-Martínez mentioned that the measures proposed to regulate the consumption of artificial sweeteners and to modify the current legislation must involve “minimizing the consumption of these special dietary products as much as possible and even avoiding adding these artificial sweeteners to the foods that we consume; for example, to coffee and tea.” On the other hand, “we must provide consumers with information that is as clear and simple as possible regarding the composition of the food they consume and how it impacts their health.”
However, “we need more evidence to be able to take a clear position on what type of sweeteners we can consume in our diet and also to what extent we should limit their presence in the foods we consume,” said Dr. Pérez-Martínez.
Last, “most of the evidence is from short-term observational studies that assess frequencies and patterns of consumption of foods containing these artificial sweeteners.” Of course, “we need studies that specifically analyze their effects at the metabolic level as well as longer-term studies where the nutritional follow-up of participants is more accurate and rigorous, especially when it comes to the consumption of this type of food,” concluded Dr. Gómez-Delgado.
This article was translated from the Medscape Spanish Edition. A version appeared on Medscape.com.
It also shows evidence that these products are not beneficial for controlling excess weight.
Francisco Gómez-Delgado, MD, PhD, and Pablo Pérez-Martínez, MD, PhD, are members of the Spanish Society of Arteriosclerosis and of the Spanish Society of Internal Medicine. They have coordinated an updated review of the leading scientific evidence surrounding artificial sweeteners: evidence showing that far from positively affecting our health, they have “negative effects for the cardiometabolic system.”
The paper, published in Current Opinion in Cardiology, delves into the consumption of these sweeteners and their negative influence on the development of obesity and of several of the most important cardiometabolic risk factors (hypertension, dyslipidemia, and diabetes).
Globalization and the increase in consumption of ultraprocessed foods have led to a need for greater knowledge on the health impacts of certain nutrients such as artificial sweeteners (nutritive and nonnutritive). This review aims to analyze their role and their effect on cardiometabolic and cardiovascular disease risk.
Cardiovascular risk
The detrimental effects of a high-calorie, high-sugar diet have been well established. For this reason, health authorities recommend limiting sugar consumption. The recommendation has led the food industry to develop different artificial sweeteners with specific properties, such as flavor and stability (nutritive artificial sweeteners), and others aimed at limiting sugar in the diet (nonnutritive artificial sweeteners). Recent evidence explores the influence of these two types of artificial sweeteners on cardiovascular disease risk through risk factors such as obesity and type 2 diabetes, among others.
Initially, the consumption of artificial sweeteners was presented as an alternative for reducing calorie intake in the diet as an option for people with excess weight and obesity. However, as this paper explains, the consumption of these artificial sweeteners favors weight gain because of neuroendocrine mechanisms related to satiety that are abnormally activated when artificial sweeteners are consumed.
Weight gain
On the other hand, evidence shows that consuming artificial sweeteners does not encourage weight loss. “Quite the contrary,” Dr. Pérez-Martínez, scientific director at the Maimonides Biomedical Research Institute and internist at the University Hospital Reina Sofia, both in Córdoba, told this news organization. “There is evidence showing weight gain resulting from the effect that artificial sweetener consumption has at the neurohormonal level by altering the mechanisms involved in regulating the feeling of satiety.”
However, on the basis of current evidence, sugar cannot be claimed to be less harmful. “What we do know is that in both cases, we should reduce or remove them from our diets and replace them with other healthier alternatives for weight management, such as eating plant-based products or being physically active.”
Confronting ignorance
Nonetheless, these recommendations are conditional, “because the weight of the evidence is not extremely high, since there have not been a whole lot of studies. All nutritional studies must be viewed with caution,” Manuel Anguita, MD, PhD, said in an interview. Dr. Anguita is department head of clinical cardiology at the University Hospital Reina Sofia in Córdoba and past president of the Spanish Society of Cardiology.
“It’s something that should be included within the medical record when you’re assessing cardiovascular risk. In addition to identifying patients who use artificial sweeteners, it’s especially important to emphasize that it’s not an appropriate recommendation for weight management.” Healthier measures include moderate exercise and the Mediterranean diet.
Explaining why this research is valuable, he said, “It’s generally useful because there’s ignorance not only in the population but among physicians as well [about] these negative effects of sweeteners.”
Diabetes and metabolic syndrome
Artificial sweeteners cause significant disruptions in the endocrine system, leading our metabolism to function abnormally. The review revealed that consuming artificial sweeteners raises the risk for type 2 diabetes by between 18% and 24% and raises the risk for metabolic syndrome by up to 44%.
Dr. Gómez-Delgado, an internal medicine specialist at the University Hospital of Jaen in Spain and first author of the study, discussed the deleterious effects of sweeteners on metabolism. “On one hand, neurohormonal disorders impact appetite, and the feeling of satiety is abnormally delayed.” On the other hand, “they induce excessive insulin secretion in the pancreas,” which in the long run, encourages metabolic disorders that lead to diabetes. Ultimately, this process produces what we know as “dysbiosis, since our microbiota is unable to process these artificial sweeteners.” Dysbiosis triggers specific pathophysiologic processes that negatively affect cardiometabolic and cardiovascular systems.
No differences
Regarding the type of sweetener, Dr. Gómez-Delgado noted that currently available studies assess the consumption of special dietary products that, in most cases, include various types of artificial sweeteners. “So, it’s not possible to define specific differences between them as to how they impact our health.” Additional studies are needed to confirm this effect at the cardiometabolic level and to analyze the different types of artificial sweeteners individually.
“There’s enough evidence to confirm that consuming artificial sweeteners negatively interferes with our metabolism – especially glucose metabolism – and increases the risk of developing diabetes,” said Dr. Gómez-Delgado.
High-sodium drinks
When it comes to the influence of artificial sweeteners on hypertension, “there is no single explanation. The World Health Organization already discussed this issue 4-5 years ago, not only due to their carcinogenic risk, but also due to this cardiovascular risk in terms of a lack of control of obesity, diabetes, and hypertension,” said Dr. Anguita.
Another important point “is that this is not in reference to the sweeteners themselves, but to soft drinks containing those components, which is where we have more studies,” he added. There are two factors explaining this increase in hypertension, which poses a problem at the population level, with medium- to long-term follow-up. “The sugary beverages that we mentioned have a higher sodium content. That is, the sweeteners add this element, which is a factor that’s directly linked to the increase in blood pressure levels.” Another factor that can influence blood pressure is “the increase in insulin secretion that has been described as resulting from sweeteners. In the medium and long term, this is associated with increased blood pressure levels.”
Cardiovascular risk factor?
Are artificial sweeteners considered to be a new cardiovascular risk factor? “What they really do is increase the incidence of the other classic risk factors,” including obesity, said Dr. Anguita. It has been shown that artificial sweeteners don’t reduce obesity when used continuously. Nonetheless, “there is still not enough evidence to view it in the same light as the classic risk factors,” added Dr. Anguita. However, it is a factor that can clearly worsen the control of the other factors. Therefore, “it’s appropriate to sound an alarm and explain that it’s not the best way to lose weight; there are many other healthier choices.”
“We need more robust evidence to take a clear position on the use of this type of sweetener and its detrimental effect on health. Meanwhile, it would be ideal to limit their consumption or even avoid adding artificial sweeteners to coffee or teas,” added Dr. Pérez-Martínez.
Regulate consumption
Dr. Pérez-Martínez mentioned that the measures proposed to regulate the consumption of artificial sweeteners and to modify the current legislation must involve “minimizing the consumption of these special dietary products as much as possible and even avoiding adding these artificial sweeteners to the foods that we consume; for example, to coffee and tea.” On the other hand, “we must provide consumers with information that is as clear and simple as possible regarding the composition of the food they consume and how it impacts their health.”
However, “we need more evidence to be able to take a clear position on what type of sweeteners we can consume in our diet and also to what extent we should limit their presence in the foods we consume,” said Dr. Pérez-Martínez.
Last, “most of the evidence is from short-term observational studies that assess frequencies and patterns of consumption of foods containing these artificial sweeteners.” Of course, “we need studies that specifically analyze their effects at the metabolic level as well as longer-term studies where the nutritional follow-up of participants is more accurate and rigorous, especially when it comes to the consumption of this type of food,” concluded Dr. Gómez-Delgado.
This article was translated from the Medscape Spanish Edition. A version appeared on Medscape.com.
It also shows evidence that these products are not beneficial for controlling excess weight.
Francisco Gómez-Delgado, MD, PhD, and Pablo Pérez-Martínez, MD, PhD, are members of the Spanish Society of Arteriosclerosis and of the Spanish Society of Internal Medicine. They have coordinated an updated review of the leading scientific evidence surrounding artificial sweeteners: evidence showing that far from positively affecting our health, they have “negative effects for the cardiometabolic system.”
The paper, published in Current Opinion in Cardiology, delves into the consumption of these sweeteners and their negative influence on the development of obesity and of several of the most important cardiometabolic risk factors (hypertension, dyslipidemia, and diabetes).
Globalization and the increase in consumption of ultraprocessed foods have led to a need for greater knowledge on the health impacts of certain nutrients such as artificial sweeteners (nutritive and nonnutritive). This review aims to analyze their role and their effect on cardiometabolic and cardiovascular disease risk.
Cardiovascular risk
The detrimental effects of a high-calorie, high-sugar diet have been well established. For this reason, health authorities recommend limiting sugar consumption. The recommendation has led the food industry to develop different artificial sweeteners with specific properties, such as flavor and stability (nutritive artificial sweeteners), and others aimed at limiting sugar in the diet (nonnutritive artificial sweeteners). Recent evidence explores the influence of these two types of artificial sweeteners on cardiovascular disease risk through risk factors such as obesity and type 2 diabetes, among others.
Initially, the consumption of artificial sweeteners was presented as an alternative for reducing calorie intake in the diet as an option for people with excess weight and obesity. However, as this paper explains, the consumption of these artificial sweeteners favors weight gain because of neuroendocrine mechanisms related to satiety that are abnormally activated when artificial sweeteners are consumed.
Weight gain
On the other hand, evidence shows that consuming artificial sweeteners does not encourage weight loss. “Quite the contrary,” Dr. Pérez-Martínez, scientific director at the Maimonides Biomedical Research Institute and internist at the University Hospital Reina Sofia, both in Córdoba, told this news organization. “There is evidence showing weight gain resulting from the effect that artificial sweetener consumption has at the neurohormonal level by altering the mechanisms involved in regulating the feeling of satiety.”
However, on the basis of current evidence, sugar cannot be claimed to be less harmful. “What we do know is that in both cases, we should reduce or remove them from our diets and replace them with other healthier alternatives for weight management, such as eating plant-based products or being physically active.”
Confronting ignorance
Nonetheless, these recommendations are conditional, “because the weight of the evidence is not extremely high, since there have not been a whole lot of studies. All nutritional studies must be viewed with caution,” Manuel Anguita, MD, PhD, said in an interview. Dr. Anguita is department head of clinical cardiology at the University Hospital Reina Sofia in Córdoba and past president of the Spanish Society of Cardiology.
“It’s something that should be included within the medical record when you’re assessing cardiovascular risk. In addition to identifying patients who use artificial sweeteners, it’s especially important to emphasize that it’s not an appropriate recommendation for weight management.” Healthier measures include moderate exercise and the Mediterranean diet.
Explaining why this research is valuable, he said, “It’s generally useful because there’s ignorance not only in the population but among physicians as well [about] these negative effects of sweeteners.”
Diabetes and metabolic syndrome
Artificial sweeteners cause significant disruptions in the endocrine system, leading our metabolism to function abnormally. The review revealed that consuming artificial sweeteners raises the risk for type 2 diabetes by between 18% and 24% and raises the risk for metabolic syndrome by up to 44%.
Dr. Gómez-Delgado, an internal medicine specialist at the University Hospital of Jaen in Spain and first author of the study, discussed the deleterious effects of sweeteners on metabolism. “On one hand, neurohormonal disorders impact appetite, and the feeling of satiety is abnormally delayed.” On the other hand, “they induce excessive insulin secretion in the pancreas,” which in the long run, encourages metabolic disorders that lead to diabetes. Ultimately, this process produces what we know as “dysbiosis, since our microbiota is unable to process these artificial sweeteners.” Dysbiosis triggers specific pathophysiologic processes that negatively affect cardiometabolic and cardiovascular systems.
No differences
Regarding the type of sweetener, Dr. Gómez-Delgado noted that currently available studies assess the consumption of special dietary products that, in most cases, include various types of artificial sweeteners. “So, it’s not possible to define specific differences between them as to how they impact our health.” Additional studies are needed to confirm this effect at the cardiometabolic level and to analyze the different types of artificial sweeteners individually.
“There’s enough evidence to confirm that consuming artificial sweeteners negatively interferes with our metabolism – especially glucose metabolism – and increases the risk of developing diabetes,” said Dr. Gómez-Delgado.
High-sodium drinks
When it comes to the influence of artificial sweeteners on hypertension, “there is no single explanation. The World Health Organization already discussed this issue 4-5 years ago, not only due to their carcinogenic risk, but also due to this cardiovascular risk in terms of a lack of control of obesity, diabetes, and hypertension,” said Dr. Anguita.
Another important point “is that this is not in reference to the sweeteners themselves, but to soft drinks containing those components, which is where we have more studies,” he added. There are two factors explaining this increase in hypertension, which poses a problem at the population level, with medium- to long-term follow-up. “The sugary beverages that we mentioned have a higher sodium content. That is, the sweeteners add this element, which is a factor that’s directly linked to the increase in blood pressure levels.” Another factor that can influence blood pressure is “the increase in insulin secretion that has been described as resulting from sweeteners. In the medium and long term, this is associated with increased blood pressure levels.”
Cardiovascular risk factor?
Are artificial sweeteners considered to be a new cardiovascular risk factor? “What they really do is increase the incidence of the other classic risk factors,” including obesity, said Dr. Anguita. It has been shown that artificial sweeteners don’t reduce obesity when used continuously. Nonetheless, “there is still not enough evidence to view it in the same light as the classic risk factors,” added Dr. Anguita. However, it is a factor that can clearly worsen the control of the other factors. Therefore, “it’s appropriate to sound an alarm and explain that it’s not the best way to lose weight; there are many other healthier choices.”
“We need more robust evidence to take a clear position on the use of this type of sweetener and its detrimental effect on health. Meanwhile, it would be ideal to limit their consumption or even avoid adding artificial sweeteners to coffee or teas,” added Dr. Pérez-Martínez.
Regulate consumption
Dr. Pérez-Martínez mentioned that the measures proposed to regulate the consumption of artificial sweeteners and to modify the current legislation must involve “minimizing the consumption of these special dietary products as much as possible and even avoiding adding these artificial sweeteners to the foods that we consume; for example, to coffee and tea.” On the other hand, “we must provide consumers with information that is as clear and simple as possible regarding the composition of the food they consume and how it impacts their health.”
However, “we need more evidence to be able to take a clear position on what type of sweeteners we can consume in our diet and also to what extent we should limit their presence in the foods we consume,” said Dr. Pérez-Martínez.
Last, “most of the evidence is from short-term observational studies that assess frequencies and patterns of consumption of foods containing these artificial sweeteners.” Of course, “we need studies that specifically analyze their effects at the metabolic level as well as longer-term studies where the nutritional follow-up of participants is more accurate and rigorous, especially when it comes to the consumption of this type of food,” concluded Dr. Gómez-Delgado.
This article was translated from the Medscape Spanish Edition. A version appeared on Medscape.com.
FROM CURRENT OPINION IN CARDIOLOGY
Dementia diagnosis a good time to reduce polypharmacy
Physicians may be missing opportunities to reduce harmful polypharmacy in elderly patients with newly diagnosed dementia, investigators for a large study of Medicare beneficiaries reported.
They found that those with an incident dementia diagnosis were somewhat more likely to initiate central nervous system–active medications and slightly more likely to discontinue cardiometabolic and anticholinergic medications, compared with controls.
According to the authors, time of diagnosis can be a potential inflexion point for deprescribing long-term medications with high safety risks, limited likelihood of benefit, or possible association with impaired cognition.
“Understanding the chronology of medication changes following a first dementia diagnosis may identify targets for deprescribing interventions to reduce preventable medication-related harms, said Timothy S. Anderson, MD, MAS, of the division of general medicine at Beth Israel Deaconess Medical Center, Boston, and colleagues in JAMA Internal Medicine.
“Our results provide a baseline to inform efforts to rethink the clinical approach to medication use at the time of a new dementia diagnosis.”
Hundreds of thousands of Americans are diagnosed annually with Alzheimer’s and related dementias, the authors pointed out, and the majority have multiple other chronic conditions. Worsening cognitive impairment may alter the risk-benefit balance of medications taken for these conditions.
Matched cohort study
The sample consisted of adults 67 years or older enrolled in traditional Medicare and Medicare Part D. Patients with an initial incident dementia diagnosis between January 2012 and December 2018 were matched with controls (as of last doctor’s office visit) based on demographics, geographic location, and baseline medication count. Data were analyzed from 2021 to June 2023.
The study included 266,675 adults with incident dementia and 266,675 controls. In both groups, 65.1% were 80 years or older (mean age, 82.2) and 67.8% were female. At baseline, patients with incident dementia were more likely than controls to use CNS-active medications (54.32% vs. 48.39%) and anticholinergic medications (17.79% vs. 15.96%) and less likely to use most cardiometabolic medications (for example, antidiabetics, 31.19% vs. 36.45%).
Immediately following the index diagnosis, the dementia cohort had greater increases in the mean number of medications used: 0.41 vs. –0.06 (95% confidence interval, 0.27-0.66) and in the proportion using CNS-active medications (absolute change, 3.44% vs. 0.79%; 95% CI, 0.85%-4.45%). The rise was because of an increased use of antipsychotics, antidepressants, and antiepileptics.
The affected cohort showed a modestly greater decline in anticholinergic medications: quarterly change in use: −0.53% vs. −0.21% (95% CI, −0.55% to −0.08%); and in most cardiometabolic medications: for example, quarterly change in antihypertensive use: –0.84% vs. –0.40% (95% CI, –0.64% to –0.25%). Still, a year post diagnosis, 75.2% of dementia patients were using five or more medications, for a 2.8% increase.
The drug classes with the steepest rate of discontinuation – such as lipid-lowering and antihypertensive medications – had low risks for adverse drug events, while higher-risk classes – such as insulins and antiplatelet and anticoagulant agents – had smaller or no reductions in use.
While the findings point to opportunities to reduce polypharmacy by deprescribing long-term medications of dubious benefit, interventions to reduce polypharmacy and inappropriate medications have been modestly successful for patients without dementia, the authors said. But the recent OPTIMIZE trial, an educational effort aimed at primary care clinicians and patients with cognitive impairment, reduced neither polypharmacy nor potentially inappropriate medications.
Luke D. Kim, MD, a geriatrician at the Cleveland Clinic in Ohio, agreed that seniors with dementia can benefit from reassessment of their pharmacologic therapies. “Older adults in general are more prone to have side effects from medications as their renal and hepatic clearance and metabolism are different and lower than those of younger individuals. But they tend to take multiple medications owing to more comorbidities,” said Dr. Kim, who was not involved in the study. “While all older adults need to be more careful about medication management, those with dementia need an even more careful approach as they have diminished cognitive reserve and risk more potential harm from medications.”
The authors noted that since decision-making models aligned with patient priorities for older adults without dementia led to reductions in overall medication use, that may be a path forward in populations with dementia.
The study was supported by grants from the National Institute on Aging, National Institutes of Health. The authors had no competing interests to disclose. Dr. Kim disclosed no competing interests relevant to his comments.
Physicians may be missing opportunities to reduce harmful polypharmacy in elderly patients with newly diagnosed dementia, investigators for a large study of Medicare beneficiaries reported.
They found that those with an incident dementia diagnosis were somewhat more likely to initiate central nervous system–active medications and slightly more likely to discontinue cardiometabolic and anticholinergic medications, compared with controls.
According to the authors, time of diagnosis can be a potential inflexion point for deprescribing long-term medications with high safety risks, limited likelihood of benefit, or possible association with impaired cognition.
“Understanding the chronology of medication changes following a first dementia diagnosis may identify targets for deprescribing interventions to reduce preventable medication-related harms, said Timothy S. Anderson, MD, MAS, of the division of general medicine at Beth Israel Deaconess Medical Center, Boston, and colleagues in JAMA Internal Medicine.
“Our results provide a baseline to inform efforts to rethink the clinical approach to medication use at the time of a new dementia diagnosis.”
Hundreds of thousands of Americans are diagnosed annually with Alzheimer’s and related dementias, the authors pointed out, and the majority have multiple other chronic conditions. Worsening cognitive impairment may alter the risk-benefit balance of medications taken for these conditions.
Matched cohort study
The sample consisted of adults 67 years or older enrolled in traditional Medicare and Medicare Part D. Patients with an initial incident dementia diagnosis between January 2012 and December 2018 were matched with controls (as of last doctor’s office visit) based on demographics, geographic location, and baseline medication count. Data were analyzed from 2021 to June 2023.
The study included 266,675 adults with incident dementia and 266,675 controls. In both groups, 65.1% were 80 years or older (mean age, 82.2) and 67.8% were female. At baseline, patients with incident dementia were more likely than controls to use CNS-active medications (54.32% vs. 48.39%) and anticholinergic medications (17.79% vs. 15.96%) and less likely to use most cardiometabolic medications (for example, antidiabetics, 31.19% vs. 36.45%).
Immediately following the index diagnosis, the dementia cohort had greater increases in the mean number of medications used: 0.41 vs. –0.06 (95% confidence interval, 0.27-0.66) and in the proportion using CNS-active medications (absolute change, 3.44% vs. 0.79%; 95% CI, 0.85%-4.45%). The rise was because of an increased use of antipsychotics, antidepressants, and antiepileptics.
The affected cohort showed a modestly greater decline in anticholinergic medications: quarterly change in use: −0.53% vs. −0.21% (95% CI, −0.55% to −0.08%); and in most cardiometabolic medications: for example, quarterly change in antihypertensive use: –0.84% vs. –0.40% (95% CI, –0.64% to –0.25%). Still, a year post diagnosis, 75.2% of dementia patients were using five or more medications, for a 2.8% increase.
The drug classes with the steepest rate of discontinuation – such as lipid-lowering and antihypertensive medications – had low risks for adverse drug events, while higher-risk classes – such as insulins and antiplatelet and anticoagulant agents – had smaller or no reductions in use.
While the findings point to opportunities to reduce polypharmacy by deprescribing long-term medications of dubious benefit, interventions to reduce polypharmacy and inappropriate medications have been modestly successful for patients without dementia, the authors said. But the recent OPTIMIZE trial, an educational effort aimed at primary care clinicians and patients with cognitive impairment, reduced neither polypharmacy nor potentially inappropriate medications.
Luke D. Kim, MD, a geriatrician at the Cleveland Clinic in Ohio, agreed that seniors with dementia can benefit from reassessment of their pharmacologic therapies. “Older adults in general are more prone to have side effects from medications as their renal and hepatic clearance and metabolism are different and lower than those of younger individuals. But they tend to take multiple medications owing to more comorbidities,” said Dr. Kim, who was not involved in the study. “While all older adults need to be more careful about medication management, those with dementia need an even more careful approach as they have diminished cognitive reserve and risk more potential harm from medications.”
The authors noted that since decision-making models aligned with patient priorities for older adults without dementia led to reductions in overall medication use, that may be a path forward in populations with dementia.
The study was supported by grants from the National Institute on Aging, National Institutes of Health. The authors had no competing interests to disclose. Dr. Kim disclosed no competing interests relevant to his comments.
Physicians may be missing opportunities to reduce harmful polypharmacy in elderly patients with newly diagnosed dementia, investigators for a large study of Medicare beneficiaries reported.
They found that those with an incident dementia diagnosis were somewhat more likely to initiate central nervous system–active medications and slightly more likely to discontinue cardiometabolic and anticholinergic medications, compared with controls.
According to the authors, time of diagnosis can be a potential inflexion point for deprescribing long-term medications with high safety risks, limited likelihood of benefit, or possible association with impaired cognition.
“Understanding the chronology of medication changes following a first dementia diagnosis may identify targets for deprescribing interventions to reduce preventable medication-related harms, said Timothy S. Anderson, MD, MAS, of the division of general medicine at Beth Israel Deaconess Medical Center, Boston, and colleagues in JAMA Internal Medicine.
“Our results provide a baseline to inform efforts to rethink the clinical approach to medication use at the time of a new dementia diagnosis.”
Hundreds of thousands of Americans are diagnosed annually with Alzheimer’s and related dementias, the authors pointed out, and the majority have multiple other chronic conditions. Worsening cognitive impairment may alter the risk-benefit balance of medications taken for these conditions.
Matched cohort study
The sample consisted of adults 67 years or older enrolled in traditional Medicare and Medicare Part D. Patients with an initial incident dementia diagnosis between January 2012 and December 2018 were matched with controls (as of last doctor’s office visit) based on demographics, geographic location, and baseline medication count. Data were analyzed from 2021 to June 2023.
The study included 266,675 adults with incident dementia and 266,675 controls. In both groups, 65.1% were 80 years or older (mean age, 82.2) and 67.8% were female. At baseline, patients with incident dementia were more likely than controls to use CNS-active medications (54.32% vs. 48.39%) and anticholinergic medications (17.79% vs. 15.96%) and less likely to use most cardiometabolic medications (for example, antidiabetics, 31.19% vs. 36.45%).
Immediately following the index diagnosis, the dementia cohort had greater increases in the mean number of medications used: 0.41 vs. –0.06 (95% confidence interval, 0.27-0.66) and in the proportion using CNS-active medications (absolute change, 3.44% vs. 0.79%; 95% CI, 0.85%-4.45%). The rise was because of an increased use of antipsychotics, antidepressants, and antiepileptics.
The affected cohort showed a modestly greater decline in anticholinergic medications: quarterly change in use: −0.53% vs. −0.21% (95% CI, −0.55% to −0.08%); and in most cardiometabolic medications: for example, quarterly change in antihypertensive use: –0.84% vs. –0.40% (95% CI, –0.64% to –0.25%). Still, a year post diagnosis, 75.2% of dementia patients were using five or more medications, for a 2.8% increase.
The drug classes with the steepest rate of discontinuation – such as lipid-lowering and antihypertensive medications – had low risks for adverse drug events, while higher-risk classes – such as insulins and antiplatelet and anticoagulant agents – had smaller or no reductions in use.
While the findings point to opportunities to reduce polypharmacy by deprescribing long-term medications of dubious benefit, interventions to reduce polypharmacy and inappropriate medications have been modestly successful for patients without dementia, the authors said. But the recent OPTIMIZE trial, an educational effort aimed at primary care clinicians and patients with cognitive impairment, reduced neither polypharmacy nor potentially inappropriate medications.
Luke D. Kim, MD, a geriatrician at the Cleveland Clinic in Ohio, agreed that seniors with dementia can benefit from reassessment of their pharmacologic therapies. “Older adults in general are more prone to have side effects from medications as their renal and hepatic clearance and metabolism are different and lower than those of younger individuals. But they tend to take multiple medications owing to more comorbidities,” said Dr. Kim, who was not involved in the study. “While all older adults need to be more careful about medication management, those with dementia need an even more careful approach as they have diminished cognitive reserve and risk more potential harm from medications.”
The authors noted that since decision-making models aligned with patient priorities for older adults without dementia led to reductions in overall medication use, that may be a path forward in populations with dementia.
The study was supported by grants from the National Institute on Aging, National Institutes of Health. The authors had no competing interests to disclose. Dr. Kim disclosed no competing interests relevant to his comments.
FROM JAMA INTERNAL MEDICINE






