User login
Richard Quinn is an award-winning journalist with 15 years’ experience. He has worked at the Asbury Park Press in New Jersey and The Virginian-Pilot in Norfolk, Va., and currently is managing editor for a leading commercial real estate publication. His freelance work has appeared in The Jewish State, The Hospitalist, The Rheumatologist, ACEP Now, and ENT Today. He lives in New Jersey with his wife and three cats.
How should Parkinson’s disease be managed perioperatively?
Case
A 67-year-old female with moderately advanced Parkinson’s disease (PD) had a mechanical fall in her home, which resulted in a humeral fracture. The fall occurred in the morning before she was able to take her medications and was related to her difficulty in initiating movements.
On her current regimen, her PD symptoms are controlled. She is able to perform daily living activities independently and ambulates without assistance. She also performs more complex tasks (e.g., cooking and managing her finances). She has not exhibited any symptoms consistent with dementia. She occasionally experiences dyspnea on exertion and dysphagia, but she has not been evaluated for these complaints. She takes carbidopa/levodopa (CD/LD) 25 mg/100 mg four times a day, amantadine 100 mg twice daily, and ropinirole 3 mg three times a day.
She is scheduled for open reduction internal fixation of her fracture; the orthopedic surgeon has requested a perioperative risk assessment and recommendations concerning her medications. How should PD be managed perioperatively?
Overview
Advances in surgical and anesthetic techniques, in combination with an aging population, have contributed to an increasing number of geriatric patients undergoing surgery. As many as 50% of Americans older than 65 will undergo a surgical procedure; hospitalists will comanage many of these patients in the perioperative period.1
Although cardiopulmonary disorders receive a great deal of attention with regard to perioperative risk assessment, other comorbid conditions also contribute to perioperative risk—namely, disorders specific to the elderly population. Parkinson’s disease is one such condition that deserves attention.
PD is a progressive, neurodegenerative condition associated with loss of dopaminergic neurons and the presence of Lewy bodies within the substantia nigra and other areas of the brain and peripheral autonomic nervous system.2 Cardinal clinical features include rigidity, bradykinesia, and resting tremor. A supportive feature is a consistent response to levodopa. Postural instability, cognitive impairment, and autonomic dysfunction usually occur later in the disease.3,4
As the population ages, Parkinson’s disease is becoming more prevalent, affecting approximately 1% of individuals older than 60.5 These patients pose a specific challenge to the hospitalist, not only because the multiorgan system manifestations of PD can raise surgical risk, but also due to the direct effects of dopaminergic medications used to treat PD, lack of a parenteral route for these medications in NPO patients, and the risks associated with abrupt withdrawal of these medications.
Although surgical risk in PD patients has received intermittent attention in surgical, anesthesia, and neurology literature, there is no broad consensus statement or treatment guideline for the perioperative approach.
Literature Review
A retrospective cohort of 51 PD patients undergoing various types of surgery revealed that PD patients have a longer hospital stay than matched cohorts.6 Pepper et al studied a cohort of 234 PD patients in the Veterans Administration population who were undergoing a variety of surgeries.7 They found that patients with PD had a longer acute hospital stay and had higher in-hospital mortality.7 The multisystem manifestations of PD might account for this global increase in perioperative risk.
The following are reviews of organ-system manifestations of PD and their relevance to the perioperative period.
Motor: The motor symptoms of PD place patients at increased risk for falls and might impair their ability to participate in rehabilitation. Mueller et al demonstrated that there was a significantly increased risk of postoperative falls and a higher need for inpatient rehabilitation due to motor difficulties in the PD cohort.6
Pulmonary: PD patients have increased risk of abnormal pulmonary function secondary to rigidity and akinesia. Increased airway resistance and decreased lung elastic recoil lead to obstructive lung disease.8,9 Rigidity of voluntary chest wall and upper airway muscles leads to a restrictive lung disease pattern.8,10 Furthermore, respiratory dyskinesia is a common side effect of levodopa, which can result in restrictive and dyskinetic ventilation.11 As a consequence of disordered respiratory mechanics (especially in combination with disordered swallow mechanics), PD patients are at increased risk of lower respiratory infections. In fact, pneumonia remains the leading cause of mortality among PD patients.11
Not surprisingly, several cohorts have suggested that PD patients undergoing surgical procedures are at higher risk for atelectasis, pneumonia, and postoperative respiratory failure.7,12 Postoperative VTE rates are not statistically different between PD patients and matched cohorts.
Gastrointestinal: Abnor-malities in muscles of the mouth, pharynx, and esophagus account for the dysphagia commonly noted in PD.13 Barium swallow tests are abnormal in 80% or more of PD patients.14 Dysphagia can lead to aspiration, as well as inadequate oral intake, resulting in pneumonia and malnutrition, respectively.15 Dysfunction of the myenteric plexus (evidenced by Lewy Body deposition) accounts for gastrointestinal dysmotility manifested as gastroparesis, ileus, and slow colonic transit, which results in constipation.16
PD patients in the postoperative period are at risk for swallowing difficulties, which increases the risk of aspiration and might delay initiation of oral medications. Gastroparesis threatens appropriate delivery of oral medications for adequate absorption. In addition, postoperative ileus and constipation can pose challenges.
Cardiovascular: Such cardiac sympathetic abnormalities as orthostatic hypotension, postprandial or exercise-induced hypotension, impaired heart rate variability, and dysrhythmias are common in PD.17 Pepper et al found a trend toward increased risk of hypotension and acute myocardial infarction (MI) in PD patients undergoing surgery.7
Genitourinary: Urinary complaints (e.g., nocturia, frequency, urgency, and urge incontinence) are common in PD patients.18 These clinical complaints correspond to involuntary detrusor contractions (detrusor hyperreflexia).19 Pepper et al found an increased risk of postoperative urinary tract infection in PD patients.7
Cognitive: A recognized feature of advanced PD is cognitive impairment. Studies estimate the prevalence of dementia in cohorts of PD patients is from 28% to 44%. PD with dementia has been associated with shortened survival, impaired quality of life, and increased caregiver distress.20 Pepper et al noted a trend toward increased incidence of postoperative delirium in their cohort of 234 PD patients undergoing surgery.7
Medication: Management of anti-Parkinsonian medications in the perioperative period poses unique challenges. These medications’ prodopaminergic effects can lead to hemodynamic compromise and are potentially arrhythmogenic. At the same time, abrupt withdrawal of these medications can lead to a potentially lethal condition called Parkinsonism-hyperpyrexia syndrome (PHS), which is clinically similar to neuroleptic malignant syndrome.21 PHS is characterized by very high fever, extreme muscle rigidity, autonomic instability, altered consciousness, and multiple severe systemic complications (e.g., acute renal failure, disseminated intravascular coagulation, autonomic failure, aspiration pneumonia, and infections). PHS occurs in up to 4% of PD patients; mortality is reported to be from 4% for treated to 20% for untreated episodes.22-24
As many as 30% of patients who survive a PHS episode have worsening of their PD symptoms and never return to their pre-PHS baseline. PHS prevention in hospitalized patients by uninterrupted administration of PD medications should be the goal. Early recognition and aggressive treatment is key to successful recovery.
Furthermore, even brief interruption of medications can lead to decompensation of Parkinsonian symptoms, which not only delays recovery from surgery, but also increases the risk for multisystem complications as discussed above.25 Traditional anti-Parkinsonian medications can only be delivered orally, presenting significant challenges for NPO patients, especially those undergoing enteric surgery requiring bowel rest.
Case reports describe various approaches to medication management in the perioperative period, but no single consensus statement (or treatment guideline) exists. The most common clinical scenarios are:
- Patient undergoes short surgery and is able to take oral medication immediately after the procedure (e.g., orthopedic, eye);
- Patient undergoes more lengthy surgery and will be able to take enteric medications perioperatively (e.g., longer orthopedic surgeries, genitourinary); and
- Patient undergoes lengthy procedure in which they will be required to adhere to bowel rest (e.g., bowel resection).
Depending on the category, the approach to medication management might differ.
Furuya et al describe a 70-year-old male with PD who had previously experienced perioperative complications associated with his medication management, including postoperative rigidity, dysphagia, and difficulty maintaining respiratory secretions.26 These symptoms were reversed with intravenous levodopa. However, the patient experienced hypotension and premature ventricular contractions as side effects of this therapy. This patient was scheduled to undergo hepatic lobectomy. Given his previous complications, Furuya et al provided enteral administration of CD/LD via nasogastric tube every two hours during the surgery, with placement of a duodenostomy for postoperative administration of CD/LD. The patient maintained hemodynamic stability throughout the perioperative period and emerged from anesthesia smoothly with no muscle rigidity or postoperative complications.26
Fujii et al described three cases of PD patients undergoing gastrointestinal surgery. They suggested that the dose of medication required to control symptoms should be minimized before surgery to avoid withdrawal symptoms. They also described the use of intravenous levodopa immediately, postoperatively, while the patient was unable to tolerate enteric medications, and suggested the use of prokinetics to prevent ileus and maximize drug absorption.27
Gálvez-Jiménez et al discuss the limitations of intravenous levodopa, including hemodynamic compromise, need for escalating doses, frequent adjustments to maintain effect, and large amount of fluids required. They propose the use of subcutaneous apomorphine, which is a potent D1/D2 dopamine agonist in conjunction with rectal domeperidone, a D2 antagonist with poor blood-brain-barrier penetration, to counteract the peripheral dopaminergic side effects. The main limitation to this approach is that domeperidone is not available in the U.S.28
Intravenous antihistamines and anticholinergics are readily available. However, they show limited efficacy in halting Parkinsonian symptoms and carry multiple side effects (e.g., confusion, delay in gastrointestinal recovery, and urinary retention).28
Various anesthesiologists agree with administering anti-PD medications immediately, preoperatively, and restarting medications as soon as possible postoperatively. However, they do not provide uniform treatment guidelines regarding specific medication management.29,30
Deep-brain-stimulation (DBS) management: DBS is an effective treatment for advanced PD. There are more than 60,000 patients around the world who have DBS for various conditions, mostly for PD. Therefore, it is increasingly likely that hospitalists will encounter hospitalized patients with advanced PD who are treated both pharmacologically and with DBS. It is important to recognize that stimulation, just like PD medications, cannot be stopped suddenly. If there is any concern of the DBS system malfunctioning (i.e., fracture of the hardware during a fall), the neurologist or neurosurgeon managing the DBS should be contacted immediately. Certain diagnostic tests (MRI) and treatment procedures (diathermy) are contraindicated, and if done inappropriately, can result in permanent brain damage.31,32,33
During surgeries requiring blood-vessel cauterization, DBS should be temporarily turned off. This can be done with the patient’s handheld device or, preferably, by a trained technician usually available through 24/7 technical support services provided by the manufacturer.
Summary of recommendations: There are no clear treatment guidelines regarding the optimal perioperative management of PD patients. The following measures are based on available data and are extensions of routine perioperative management; however, there is no evidence to demonstrate their efficacy in decreasing complication rates among patients with PD:
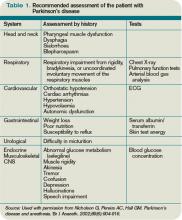
- A thorough preoperative history and physical examination should include Parkinsonian signs and symptoms, precise medication regimen with doses and timing of intake, effects of medication withdrawal or missed doses, type of surgery planned, and comorbid conditions;
- Depending on symptoms mentioned in the history, consider further testing for dysphagia (preoperative swallow evaluation) and dyspnea (preoperative pulmonary function tests);
- The major goal of medication management in the perioperative period is to continue administration of dopamine replacement therapy as close to the outpatient regimen as possible.
- Titrate down dose of anti-Parkinsonian medications to lowest possible dose prior to surgery if prolonged NPO status is anticipated;
- Ensure medications are administered immediately prior to surgery;
- For short, nonenteric procedures, resume outpatient medication doses and timing of administration as soon as possible postoperatively. For longer, nonenteric surgeries, consider placement of nasogastric tube for medication delivery during procedure and immediately postoperatively;
- If the major limitation of oral medication intake is dysphagia, the use of orally disintegrated formulation CD/LD (parcopa) is helpful;
- For longer enteric surgeries in which patient must be on bowel rest, recommend consultation with neurologist specialized in movement disorders to guide use of intravenous or subcutaneous agents. Transdermal delivery systems of the dopamine agonist rotigotine are in the process of being approved in the U.S. market and might be helpful for this purpose;
- Initiation of tube feeding, when co-administered with levodopa, might result in sudden changes in medication absorption and potential worsening of PD symptoms; feeding should be started slowly and preferably at night when the body’s dopamine requirements are lower;
- Consider use of promotility agents;
- If apomorphine or intravenous LD are not available, consider trial of intravenous anticholinergics or antihistamines, carefully observing for potential cognitive and behavioral side effects;
- Avoid such dopamine antagonists as droperidol, haloperidol, risperidone, metaclopramide, prochlorperazine, or promethazine, as these medications can worsen Parkinsonian symptoms; and
- If patient was on MAO-B inhibitors (selegiline, rasagiline) before surgery, be aware of multiple potential interactions with various medications that are commonly used in perioperative period, including anesthesia agents and certain analgesics, specifically meperidine. These interactions include serotonin syndrome, and can be life-threatening.34
- Psychiatric considerations: delirium precautions;
- Motor considerations: early PT/OT, early referral to inpatient rehabilitation; fall precautions;
- Pulmonary considerations: institute aggressive incentive spirometry, postural drainage, management of respiratory secretions, and breathing exercises; VTE prophylaxis;
- Gastrointestinal considerations: aspiration precautions and prompt speech therapy to evaluate for aspiration and to teach appropriate swallow techniques (chin tuck); institute aggressive bowel regimen; maximize fluids, electrolytes, and avoid narcotics to prevent precipitating or exacerbating ileus;
- Cardiovascular considerations: monitor orthostatic vital signs; fall precautions to avoid syncopal falls; and
- Genitourinary considerations: early urinary catheter removal; vigilance in monitoring for urinary tract infection.
Back to the Case
The patient underwent repair of her fracture, was extubated, and recovered from general anesthesia without incident. She was evaluated in the postanesthesia care unit, at which time she had a slight tremor and mild rigidity. She was immediately given a dose of her CD/LD, and her evening doses of amantadine and ropinirole were resumed. The patient had no significant flare of her Parkinsonian symptoms and did not exhibit any evidence of PHS.
A postoperative consultation was placed for speech therapy, physical therapy, and occupational therapy. She was given low-molecular-weight heparin for VTE prophylaxis and asked to use incentive spirometry. On postoperative day one, she complained of urinary frequency. A urinalysis was consistent with possible infection. She was discharged home on her previous medication regimen, in addition to antibiotics for cystitis.
If the procedure had not been emergent, the patient might have benefited from a preoperative swallow evaluation, given her dysphagia. Consultation with a speech therapist would have ensured that the patient was educated regarding aspiration precautions. Although this patient did not have difficulty with extubation or experience postoperative respiratory failure, abnormal preoperative pulmonary function tests might have prompted the anesthesiologists to consider alternative low-risk techniques (e.g., a local nerve block).
Bottom Line
Perioperative management of patients with Parkinson’s disease requires knowledge of the multisystem disease characteristics that raise perioperative risk and the effects of the medications used to treat PD. To date, no clear treatment guidelines exist for the optimal perioperative management of PD patients.
However, vigilance in detecting possible complications and instituting attentive perioperative care can aid a hospitalist consultant in improving overall care for these patients. TH
Dr. Patel is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Denver VA Medical Center and instructor of medicine at the University of Colorado Denver. Dr. Anderson is a hospitalist at the Denver VA Medical Center and assistant professor of medicine at the University of Colorado Denver. Dr. Klepitskaya is a neurologist and assistant professor of neurology at the University of Colorado Denver.
References
- Ersan T. Perioperative management of the geriatric patient. Web MD website. Available at: http://emedicine.medscape.com/article/285433-overview. Accessed May 10, 2010.
- Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211.
- Ward CD, Gibb WR. Research diagnostic criteria for Parkinson’s disease. Adv Neurol. 1990;53:245-249.
- Stern MB, Koller WC. Parkinsonian Syndromes. New York: Informa Healthcare Publishing; 1993.
- Nutt JG, Wooten GF. Clinical practice. Diagnosis and initial management of Parkinson’s disease. N Engl J Med. 2005;353(10):1021-1027.
- Mueller MC, Jüptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394(3):511-515.
- Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47(8):967-972.
- Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci. 1998;5(2):131-135.
- Neu HC, Connolly JJ Jr., Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Ann Rev Respir Dis. 1967;95(1):33-47.
- Chokroverty S. The spectrum of ventilatory disturbances in movement disorders. In: Chokroverty S, ed. Movement Disorders. Great Neck, New York: PMA Publishing Corp.; 1990:365-392.
- Shill H, Stacy M. Respiratory complications of Parkinson’s disease. Semin Respir Crit Care Med. 2002;23(3):261-265.
- Easdown LJ, Tessler KJ, Minuk J. Upper airway involvement in Parkinson’s disease resulting in postoperative respiratory failure. Can J Anaesth. 1995;42(4):344-347.
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2(2):107-116.
- Castell JA, Johnston BT, Colcher A, Li Q, Gideon RM, Castell DO. Manometric abnormalities of the oesophagus in patients with Parkinson’s disease. Neurogastroenterol Motil. 2001;13(4):361-364.
- Marcason W. What are the primary nutritional issues for a patient with Parkinson’s disease? J Am Diet Assoc. 2009;109(7):1316.
- Djaldetti R, Lev N, Melamed E. Lesions outside the CNS in Parkinson’s disease. Mov Disord. 2009;24(6):793-800.
- Orimo S, Takahashi A, Uchihara T, Mori F, Kakita A, Wakabayashi K, Takahashi H. Degeneration of cardiac sympathetic nerve begins in the early disease process of Parkinson’s disease. Brain Pathol. 2007;17(1):24-30.
- Lemack GE, Dewey RB Jr., Roehrborn CG, O’Suilleabhain PE, Zimmern PE. Questionnaire-based assessment of bladder dysfunction in patients with mild to moderate Parkinson’s disease. Urology. 2000;56(2):250-254.
- Singer C. Urinary dysfunction in Parkinson’s disease. Clin Neurosci. 1998;5(2):78-86.
- Leverenz JB, Quinn JF, Zabetian C, Zhang J, Montine KS, Montine TJ. Cognitive impairment and dementia in patients with Parkinson disease. Curr Top Med Chem. 2009;9(10):903-912.
- Factor SA, Santiago A. Parkinson-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht S, Fahn S, eds. Movement Disorder Emergencies: Diagnosis and Treatment. New York: Humana Press; 2005:29-40.
- Reed AP, Han DG: Intraoperative exacerbation of Parkinson’s disease. Anesth Analg. 1992;75(5):850-853.
- Harada T, Mitsuoka K, Kumagai R, et al. Clinical features of malignant syndrome in Parkinson’s disease and related neurological disorders. Parkinsonism Relat Disord. 2003;9(Suppl 1):S15-23.
- Onofrj M, Thomas A. Acute akinesia in Parkinson disease. Neurology. 2005;64(7):1162-1169.
- Serrano-Dueñas M. Neuroleptic malignant syndrome-like, or—dopaminergic malignant syndrome—due to levodopa therapy withdrawal. Clinical features in 11 patients. Parkinsonism Relat Disord. 2003;9(3):175-178.
- Furuya R, Hirai A, Andoh T, Kudoh I, Okumura F. Successful perioperative management of a patient with Parkinson’s disease by enteral levodopa administration under propofol anesthesia. Anesthesiology. 1998;89(1):261-263.
- Fujii T, Nakabayashi T, Hashimoto S, Kuwano H. Successful perioperative management of patients with Parkinson’s disease following gastrointestinal surgery: report of three cases. Surg Today. 2009;39(9):807-810.
- Gálvez-Jiménez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22(2):367-377.
- Kalenka A, Schwarz A. Anasthesia and Parkinson’s disease: how to manage with new therapies? Curr Opin Anesthesiol. 2009;22(3):419-424.
- Nicholson G, Pereira AC, Hall GM. Parkinson’s disease and anesthesia. Br J Anaesthesia. 2002;89(6):904-916.
- Rezai AR, Baker KB, Tkach JA, et al. Is magnetic resonance imaging safe for patients with neurostimulation systems used for deep brain stimulation? Neurosurgery. 2005:57(5):1056-1062.
- Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery. 2005:57(5):E1063.
- Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology. 2001;56 (10):1384-1386.
- Gordon PH, Frucht SJ. Neuroleptic malignant syndrome in advanced Parkinson’s disease. Mov Disord. 2001;16(5):960-962.
Case
A 67-year-old female with moderately advanced Parkinson’s disease (PD) had a mechanical fall in her home, which resulted in a humeral fracture. The fall occurred in the morning before she was able to take her medications and was related to her difficulty in initiating movements.
On her current regimen, her PD symptoms are controlled. She is able to perform daily living activities independently and ambulates without assistance. She also performs more complex tasks (e.g., cooking and managing her finances). She has not exhibited any symptoms consistent with dementia. She occasionally experiences dyspnea on exertion and dysphagia, but she has not been evaluated for these complaints. She takes carbidopa/levodopa (CD/LD) 25 mg/100 mg four times a day, amantadine 100 mg twice daily, and ropinirole 3 mg three times a day.
She is scheduled for open reduction internal fixation of her fracture; the orthopedic surgeon has requested a perioperative risk assessment and recommendations concerning her medications. How should PD be managed perioperatively?
Overview
Advances in surgical and anesthetic techniques, in combination with an aging population, have contributed to an increasing number of geriatric patients undergoing surgery. As many as 50% of Americans older than 65 will undergo a surgical procedure; hospitalists will comanage many of these patients in the perioperative period.1
Although cardiopulmonary disorders receive a great deal of attention with regard to perioperative risk assessment, other comorbid conditions also contribute to perioperative risk—namely, disorders specific to the elderly population. Parkinson’s disease is one such condition that deserves attention.
PD is a progressive, neurodegenerative condition associated with loss of dopaminergic neurons and the presence of Lewy bodies within the substantia nigra and other areas of the brain and peripheral autonomic nervous system.2 Cardinal clinical features include rigidity, bradykinesia, and resting tremor. A supportive feature is a consistent response to levodopa. Postural instability, cognitive impairment, and autonomic dysfunction usually occur later in the disease.3,4
As the population ages, Parkinson’s disease is becoming more prevalent, affecting approximately 1% of individuals older than 60.5 These patients pose a specific challenge to the hospitalist, not only because the multiorgan system manifestations of PD can raise surgical risk, but also due to the direct effects of dopaminergic medications used to treat PD, lack of a parenteral route for these medications in NPO patients, and the risks associated with abrupt withdrawal of these medications.
Although surgical risk in PD patients has received intermittent attention in surgical, anesthesia, and neurology literature, there is no broad consensus statement or treatment guideline for the perioperative approach.
Literature Review
A retrospective cohort of 51 PD patients undergoing various types of surgery revealed that PD patients have a longer hospital stay than matched cohorts.6 Pepper et al studied a cohort of 234 PD patients in the Veterans Administration population who were undergoing a variety of surgeries.7 They found that patients with PD had a longer acute hospital stay and had higher in-hospital mortality.7 The multisystem manifestations of PD might account for this global increase in perioperative risk.
The following are reviews of organ-system manifestations of PD and their relevance to the perioperative period.
Motor: The motor symptoms of PD place patients at increased risk for falls and might impair their ability to participate in rehabilitation. Mueller et al demonstrated that there was a significantly increased risk of postoperative falls and a higher need for inpatient rehabilitation due to motor difficulties in the PD cohort.6
Pulmonary: PD patients have increased risk of abnormal pulmonary function secondary to rigidity and akinesia. Increased airway resistance and decreased lung elastic recoil lead to obstructive lung disease.8,9 Rigidity of voluntary chest wall and upper airway muscles leads to a restrictive lung disease pattern.8,10 Furthermore, respiratory dyskinesia is a common side effect of levodopa, which can result in restrictive and dyskinetic ventilation.11 As a consequence of disordered respiratory mechanics (especially in combination with disordered swallow mechanics), PD patients are at increased risk of lower respiratory infections. In fact, pneumonia remains the leading cause of mortality among PD patients.11
Not surprisingly, several cohorts have suggested that PD patients undergoing surgical procedures are at higher risk for atelectasis, pneumonia, and postoperative respiratory failure.7,12 Postoperative VTE rates are not statistically different between PD patients and matched cohorts.
Gastrointestinal: Abnor-malities in muscles of the mouth, pharynx, and esophagus account for the dysphagia commonly noted in PD.13 Barium swallow tests are abnormal in 80% or more of PD patients.14 Dysphagia can lead to aspiration, as well as inadequate oral intake, resulting in pneumonia and malnutrition, respectively.15 Dysfunction of the myenteric plexus (evidenced by Lewy Body deposition) accounts for gastrointestinal dysmotility manifested as gastroparesis, ileus, and slow colonic transit, which results in constipation.16
PD patients in the postoperative period are at risk for swallowing difficulties, which increases the risk of aspiration and might delay initiation of oral medications. Gastroparesis threatens appropriate delivery of oral medications for adequate absorption. In addition, postoperative ileus and constipation can pose challenges.
Cardiovascular: Such cardiac sympathetic abnormalities as orthostatic hypotension, postprandial or exercise-induced hypotension, impaired heart rate variability, and dysrhythmias are common in PD.17 Pepper et al found a trend toward increased risk of hypotension and acute myocardial infarction (MI) in PD patients undergoing surgery.7
Genitourinary: Urinary complaints (e.g., nocturia, frequency, urgency, and urge incontinence) are common in PD patients.18 These clinical complaints correspond to involuntary detrusor contractions (detrusor hyperreflexia).19 Pepper et al found an increased risk of postoperative urinary tract infection in PD patients.7
Cognitive: A recognized feature of advanced PD is cognitive impairment. Studies estimate the prevalence of dementia in cohorts of PD patients is from 28% to 44%. PD with dementia has been associated with shortened survival, impaired quality of life, and increased caregiver distress.20 Pepper et al noted a trend toward increased incidence of postoperative delirium in their cohort of 234 PD patients undergoing surgery.7
Medication: Management of anti-Parkinsonian medications in the perioperative period poses unique challenges. These medications’ prodopaminergic effects can lead to hemodynamic compromise and are potentially arrhythmogenic. At the same time, abrupt withdrawal of these medications can lead to a potentially lethal condition called Parkinsonism-hyperpyrexia syndrome (PHS), which is clinically similar to neuroleptic malignant syndrome.21 PHS is characterized by very high fever, extreme muscle rigidity, autonomic instability, altered consciousness, and multiple severe systemic complications (e.g., acute renal failure, disseminated intravascular coagulation, autonomic failure, aspiration pneumonia, and infections). PHS occurs in up to 4% of PD patients; mortality is reported to be from 4% for treated to 20% for untreated episodes.22-24
As many as 30% of patients who survive a PHS episode have worsening of their PD symptoms and never return to their pre-PHS baseline. PHS prevention in hospitalized patients by uninterrupted administration of PD medications should be the goal. Early recognition and aggressive treatment is key to successful recovery.
Furthermore, even brief interruption of medications can lead to decompensation of Parkinsonian symptoms, which not only delays recovery from surgery, but also increases the risk for multisystem complications as discussed above.25 Traditional anti-Parkinsonian medications can only be delivered orally, presenting significant challenges for NPO patients, especially those undergoing enteric surgery requiring bowel rest.
Case reports describe various approaches to medication management in the perioperative period, but no single consensus statement (or treatment guideline) exists. The most common clinical scenarios are:
- Patient undergoes short surgery and is able to take oral medication immediately after the procedure (e.g., orthopedic, eye);
- Patient undergoes more lengthy surgery and will be able to take enteric medications perioperatively (e.g., longer orthopedic surgeries, genitourinary); and
- Patient undergoes lengthy procedure in which they will be required to adhere to bowel rest (e.g., bowel resection).
Depending on the category, the approach to medication management might differ.
Furuya et al describe a 70-year-old male with PD who had previously experienced perioperative complications associated with his medication management, including postoperative rigidity, dysphagia, and difficulty maintaining respiratory secretions.26 These symptoms were reversed with intravenous levodopa. However, the patient experienced hypotension and premature ventricular contractions as side effects of this therapy. This patient was scheduled to undergo hepatic lobectomy. Given his previous complications, Furuya et al provided enteral administration of CD/LD via nasogastric tube every two hours during the surgery, with placement of a duodenostomy for postoperative administration of CD/LD. The patient maintained hemodynamic stability throughout the perioperative period and emerged from anesthesia smoothly with no muscle rigidity or postoperative complications.26
Fujii et al described three cases of PD patients undergoing gastrointestinal surgery. They suggested that the dose of medication required to control symptoms should be minimized before surgery to avoid withdrawal symptoms. They also described the use of intravenous levodopa immediately, postoperatively, while the patient was unable to tolerate enteric medications, and suggested the use of prokinetics to prevent ileus and maximize drug absorption.27
Gálvez-Jiménez et al discuss the limitations of intravenous levodopa, including hemodynamic compromise, need for escalating doses, frequent adjustments to maintain effect, and large amount of fluids required. They propose the use of subcutaneous apomorphine, which is a potent D1/D2 dopamine agonist in conjunction with rectal domeperidone, a D2 antagonist with poor blood-brain-barrier penetration, to counteract the peripheral dopaminergic side effects. The main limitation to this approach is that domeperidone is not available in the U.S.28
Intravenous antihistamines and anticholinergics are readily available. However, they show limited efficacy in halting Parkinsonian symptoms and carry multiple side effects (e.g., confusion, delay in gastrointestinal recovery, and urinary retention).28
Various anesthesiologists agree with administering anti-PD medications immediately, preoperatively, and restarting medications as soon as possible postoperatively. However, they do not provide uniform treatment guidelines regarding specific medication management.29,30
Deep-brain-stimulation (DBS) management: DBS is an effective treatment for advanced PD. There are more than 60,000 patients around the world who have DBS for various conditions, mostly for PD. Therefore, it is increasingly likely that hospitalists will encounter hospitalized patients with advanced PD who are treated both pharmacologically and with DBS. It is important to recognize that stimulation, just like PD medications, cannot be stopped suddenly. If there is any concern of the DBS system malfunctioning (i.e., fracture of the hardware during a fall), the neurologist or neurosurgeon managing the DBS should be contacted immediately. Certain diagnostic tests (MRI) and treatment procedures (diathermy) are contraindicated, and if done inappropriately, can result in permanent brain damage.31,32,33
During surgeries requiring blood-vessel cauterization, DBS should be temporarily turned off. This can be done with the patient’s handheld device or, preferably, by a trained technician usually available through 24/7 technical support services provided by the manufacturer.
Summary of recommendations: There are no clear treatment guidelines regarding the optimal perioperative management of PD patients. The following measures are based on available data and are extensions of routine perioperative management; however, there is no evidence to demonstrate their efficacy in decreasing complication rates among patients with PD:
- A thorough preoperative history and physical examination should include Parkinsonian signs and symptoms, precise medication regimen with doses and timing of intake, effects of medication withdrawal or missed doses, type of surgery planned, and comorbid conditions;
- Depending on symptoms mentioned in the history, consider further testing for dysphagia (preoperative swallow evaluation) and dyspnea (preoperative pulmonary function tests);
- The major goal of medication management in the perioperative period is to continue administration of dopamine replacement therapy as close to the outpatient regimen as possible.
- Titrate down dose of anti-Parkinsonian medications to lowest possible dose prior to surgery if prolonged NPO status is anticipated;
- Ensure medications are administered immediately prior to surgery;
- For short, nonenteric procedures, resume outpatient medication doses and timing of administration as soon as possible postoperatively. For longer, nonenteric surgeries, consider placement of nasogastric tube for medication delivery during procedure and immediately postoperatively;
- If the major limitation of oral medication intake is dysphagia, the use of orally disintegrated formulation CD/LD (parcopa) is helpful;
- For longer enteric surgeries in which patient must be on bowel rest, recommend consultation with neurologist specialized in movement disorders to guide use of intravenous or subcutaneous agents. Transdermal delivery systems of the dopamine agonist rotigotine are in the process of being approved in the U.S. market and might be helpful for this purpose;
- Initiation of tube feeding, when co-administered with levodopa, might result in sudden changes in medication absorption and potential worsening of PD symptoms; feeding should be started slowly and preferably at night when the body’s dopamine requirements are lower;
- Consider use of promotility agents;
- If apomorphine or intravenous LD are not available, consider trial of intravenous anticholinergics or antihistamines, carefully observing for potential cognitive and behavioral side effects;
- Avoid such dopamine antagonists as droperidol, haloperidol, risperidone, metaclopramide, prochlorperazine, or promethazine, as these medications can worsen Parkinsonian symptoms; and
- If patient was on MAO-B inhibitors (selegiline, rasagiline) before surgery, be aware of multiple potential interactions with various medications that are commonly used in perioperative period, including anesthesia agents and certain analgesics, specifically meperidine. These interactions include serotonin syndrome, and can be life-threatening.34
- Psychiatric considerations: delirium precautions;
- Motor considerations: early PT/OT, early referral to inpatient rehabilitation; fall precautions;
- Pulmonary considerations: institute aggressive incentive spirometry, postural drainage, management of respiratory secretions, and breathing exercises; VTE prophylaxis;
- Gastrointestinal considerations: aspiration precautions and prompt speech therapy to evaluate for aspiration and to teach appropriate swallow techniques (chin tuck); institute aggressive bowel regimen; maximize fluids, electrolytes, and avoid narcotics to prevent precipitating or exacerbating ileus;
- Cardiovascular considerations: monitor orthostatic vital signs; fall precautions to avoid syncopal falls; and
- Genitourinary considerations: early urinary catheter removal; vigilance in monitoring for urinary tract infection.
Back to the Case
The patient underwent repair of her fracture, was extubated, and recovered from general anesthesia without incident. She was evaluated in the postanesthesia care unit, at which time she had a slight tremor and mild rigidity. She was immediately given a dose of her CD/LD, and her evening doses of amantadine and ropinirole were resumed. The patient had no significant flare of her Parkinsonian symptoms and did not exhibit any evidence of PHS.
A postoperative consultation was placed for speech therapy, physical therapy, and occupational therapy. She was given low-molecular-weight heparin for VTE prophylaxis and asked to use incentive spirometry. On postoperative day one, she complained of urinary frequency. A urinalysis was consistent with possible infection. She was discharged home on her previous medication regimen, in addition to antibiotics for cystitis.
If the procedure had not been emergent, the patient might have benefited from a preoperative swallow evaluation, given her dysphagia. Consultation with a speech therapist would have ensured that the patient was educated regarding aspiration precautions. Although this patient did not have difficulty with extubation or experience postoperative respiratory failure, abnormal preoperative pulmonary function tests might have prompted the anesthesiologists to consider alternative low-risk techniques (e.g., a local nerve block).
Bottom Line
Perioperative management of patients with Parkinson’s disease requires knowledge of the multisystem disease characteristics that raise perioperative risk and the effects of the medications used to treat PD. To date, no clear treatment guidelines exist for the optimal perioperative management of PD patients.
However, vigilance in detecting possible complications and instituting attentive perioperative care can aid a hospitalist consultant in improving overall care for these patients. TH
Dr. Patel is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Denver VA Medical Center and instructor of medicine at the University of Colorado Denver. Dr. Anderson is a hospitalist at the Denver VA Medical Center and assistant professor of medicine at the University of Colorado Denver. Dr. Klepitskaya is a neurologist and assistant professor of neurology at the University of Colorado Denver.
References
- Ersan T. Perioperative management of the geriatric patient. Web MD website. Available at: http://emedicine.medscape.com/article/285433-overview. Accessed May 10, 2010.
- Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211.
- Ward CD, Gibb WR. Research diagnostic criteria for Parkinson’s disease. Adv Neurol. 1990;53:245-249.
- Stern MB, Koller WC. Parkinsonian Syndromes. New York: Informa Healthcare Publishing; 1993.
- Nutt JG, Wooten GF. Clinical practice. Diagnosis and initial management of Parkinson’s disease. N Engl J Med. 2005;353(10):1021-1027.
- Mueller MC, Jüptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394(3):511-515.
- Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47(8):967-972.
- Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci. 1998;5(2):131-135.
- Neu HC, Connolly JJ Jr., Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Ann Rev Respir Dis. 1967;95(1):33-47.
- Chokroverty S. The spectrum of ventilatory disturbances in movement disorders. In: Chokroverty S, ed. Movement Disorders. Great Neck, New York: PMA Publishing Corp.; 1990:365-392.
- Shill H, Stacy M. Respiratory complications of Parkinson’s disease. Semin Respir Crit Care Med. 2002;23(3):261-265.
- Easdown LJ, Tessler KJ, Minuk J. Upper airway involvement in Parkinson’s disease resulting in postoperative respiratory failure. Can J Anaesth. 1995;42(4):344-347.
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2(2):107-116.
- Castell JA, Johnston BT, Colcher A, Li Q, Gideon RM, Castell DO. Manometric abnormalities of the oesophagus in patients with Parkinson’s disease. Neurogastroenterol Motil. 2001;13(4):361-364.
- Marcason W. What are the primary nutritional issues for a patient with Parkinson’s disease? J Am Diet Assoc. 2009;109(7):1316.
- Djaldetti R, Lev N, Melamed E. Lesions outside the CNS in Parkinson’s disease. Mov Disord. 2009;24(6):793-800.
- Orimo S, Takahashi A, Uchihara T, Mori F, Kakita A, Wakabayashi K, Takahashi H. Degeneration of cardiac sympathetic nerve begins in the early disease process of Parkinson’s disease. Brain Pathol. 2007;17(1):24-30.
- Lemack GE, Dewey RB Jr., Roehrborn CG, O’Suilleabhain PE, Zimmern PE. Questionnaire-based assessment of bladder dysfunction in patients with mild to moderate Parkinson’s disease. Urology. 2000;56(2):250-254.
- Singer C. Urinary dysfunction in Parkinson’s disease. Clin Neurosci. 1998;5(2):78-86.
- Leverenz JB, Quinn JF, Zabetian C, Zhang J, Montine KS, Montine TJ. Cognitive impairment and dementia in patients with Parkinson disease. Curr Top Med Chem. 2009;9(10):903-912.
- Factor SA, Santiago A. Parkinson-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht S, Fahn S, eds. Movement Disorder Emergencies: Diagnosis and Treatment. New York: Humana Press; 2005:29-40.
- Reed AP, Han DG: Intraoperative exacerbation of Parkinson’s disease. Anesth Analg. 1992;75(5):850-853.
- Harada T, Mitsuoka K, Kumagai R, et al. Clinical features of malignant syndrome in Parkinson’s disease and related neurological disorders. Parkinsonism Relat Disord. 2003;9(Suppl 1):S15-23.
- Onofrj M, Thomas A. Acute akinesia in Parkinson disease. Neurology. 2005;64(7):1162-1169.
- Serrano-Dueñas M. Neuroleptic malignant syndrome-like, or—dopaminergic malignant syndrome—due to levodopa therapy withdrawal. Clinical features in 11 patients. Parkinsonism Relat Disord. 2003;9(3):175-178.
- Furuya R, Hirai A, Andoh T, Kudoh I, Okumura F. Successful perioperative management of a patient with Parkinson’s disease by enteral levodopa administration under propofol anesthesia. Anesthesiology. 1998;89(1):261-263.
- Fujii T, Nakabayashi T, Hashimoto S, Kuwano H. Successful perioperative management of patients with Parkinson’s disease following gastrointestinal surgery: report of three cases. Surg Today. 2009;39(9):807-810.
- Gálvez-Jiménez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22(2):367-377.
- Kalenka A, Schwarz A. Anasthesia and Parkinson’s disease: how to manage with new therapies? Curr Opin Anesthesiol. 2009;22(3):419-424.
- Nicholson G, Pereira AC, Hall GM. Parkinson’s disease and anesthesia. Br J Anaesthesia. 2002;89(6):904-916.
- Rezai AR, Baker KB, Tkach JA, et al. Is magnetic resonance imaging safe for patients with neurostimulation systems used for deep brain stimulation? Neurosurgery. 2005:57(5):1056-1062.
- Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery. 2005:57(5):E1063.
- Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology. 2001;56 (10):1384-1386.
- Gordon PH, Frucht SJ. Neuroleptic malignant syndrome in advanced Parkinson’s disease. Mov Disord. 2001;16(5):960-962.
Case
A 67-year-old female with moderately advanced Parkinson’s disease (PD) had a mechanical fall in her home, which resulted in a humeral fracture. The fall occurred in the morning before she was able to take her medications and was related to her difficulty in initiating movements.
On her current regimen, her PD symptoms are controlled. She is able to perform daily living activities independently and ambulates without assistance. She also performs more complex tasks (e.g., cooking and managing her finances). She has not exhibited any symptoms consistent with dementia. She occasionally experiences dyspnea on exertion and dysphagia, but she has not been evaluated for these complaints. She takes carbidopa/levodopa (CD/LD) 25 mg/100 mg four times a day, amantadine 100 mg twice daily, and ropinirole 3 mg three times a day.
She is scheduled for open reduction internal fixation of her fracture; the orthopedic surgeon has requested a perioperative risk assessment and recommendations concerning her medications. How should PD be managed perioperatively?
Overview
Advances in surgical and anesthetic techniques, in combination with an aging population, have contributed to an increasing number of geriatric patients undergoing surgery. As many as 50% of Americans older than 65 will undergo a surgical procedure; hospitalists will comanage many of these patients in the perioperative period.1
Although cardiopulmonary disorders receive a great deal of attention with regard to perioperative risk assessment, other comorbid conditions also contribute to perioperative risk—namely, disorders specific to the elderly population. Parkinson’s disease is one such condition that deserves attention.
PD is a progressive, neurodegenerative condition associated with loss of dopaminergic neurons and the presence of Lewy bodies within the substantia nigra and other areas of the brain and peripheral autonomic nervous system.2 Cardinal clinical features include rigidity, bradykinesia, and resting tremor. A supportive feature is a consistent response to levodopa. Postural instability, cognitive impairment, and autonomic dysfunction usually occur later in the disease.3,4
As the population ages, Parkinson’s disease is becoming more prevalent, affecting approximately 1% of individuals older than 60.5 These patients pose a specific challenge to the hospitalist, not only because the multiorgan system manifestations of PD can raise surgical risk, but also due to the direct effects of dopaminergic medications used to treat PD, lack of a parenteral route for these medications in NPO patients, and the risks associated with abrupt withdrawal of these medications.
Although surgical risk in PD patients has received intermittent attention in surgical, anesthesia, and neurology literature, there is no broad consensus statement or treatment guideline for the perioperative approach.
Literature Review
A retrospective cohort of 51 PD patients undergoing various types of surgery revealed that PD patients have a longer hospital stay than matched cohorts.6 Pepper et al studied a cohort of 234 PD patients in the Veterans Administration population who were undergoing a variety of surgeries.7 They found that patients with PD had a longer acute hospital stay and had higher in-hospital mortality.7 The multisystem manifestations of PD might account for this global increase in perioperative risk.
The following are reviews of organ-system manifestations of PD and their relevance to the perioperative period.
Motor: The motor symptoms of PD place patients at increased risk for falls and might impair their ability to participate in rehabilitation. Mueller et al demonstrated that there was a significantly increased risk of postoperative falls and a higher need for inpatient rehabilitation due to motor difficulties in the PD cohort.6
Pulmonary: PD patients have increased risk of abnormal pulmonary function secondary to rigidity and akinesia. Increased airway resistance and decreased lung elastic recoil lead to obstructive lung disease.8,9 Rigidity of voluntary chest wall and upper airway muscles leads to a restrictive lung disease pattern.8,10 Furthermore, respiratory dyskinesia is a common side effect of levodopa, which can result in restrictive and dyskinetic ventilation.11 As a consequence of disordered respiratory mechanics (especially in combination with disordered swallow mechanics), PD patients are at increased risk of lower respiratory infections. In fact, pneumonia remains the leading cause of mortality among PD patients.11
Not surprisingly, several cohorts have suggested that PD patients undergoing surgical procedures are at higher risk for atelectasis, pneumonia, and postoperative respiratory failure.7,12 Postoperative VTE rates are not statistically different between PD patients and matched cohorts.
Gastrointestinal: Abnor-malities in muscles of the mouth, pharynx, and esophagus account for the dysphagia commonly noted in PD.13 Barium swallow tests are abnormal in 80% or more of PD patients.14 Dysphagia can lead to aspiration, as well as inadequate oral intake, resulting in pneumonia and malnutrition, respectively.15 Dysfunction of the myenteric plexus (evidenced by Lewy Body deposition) accounts for gastrointestinal dysmotility manifested as gastroparesis, ileus, and slow colonic transit, which results in constipation.16
PD patients in the postoperative period are at risk for swallowing difficulties, which increases the risk of aspiration and might delay initiation of oral medications. Gastroparesis threatens appropriate delivery of oral medications for adequate absorption. In addition, postoperative ileus and constipation can pose challenges.
Cardiovascular: Such cardiac sympathetic abnormalities as orthostatic hypotension, postprandial or exercise-induced hypotension, impaired heart rate variability, and dysrhythmias are common in PD.17 Pepper et al found a trend toward increased risk of hypotension and acute myocardial infarction (MI) in PD patients undergoing surgery.7
Genitourinary: Urinary complaints (e.g., nocturia, frequency, urgency, and urge incontinence) are common in PD patients.18 These clinical complaints correspond to involuntary detrusor contractions (detrusor hyperreflexia).19 Pepper et al found an increased risk of postoperative urinary tract infection in PD patients.7
Cognitive: A recognized feature of advanced PD is cognitive impairment. Studies estimate the prevalence of dementia in cohorts of PD patients is from 28% to 44%. PD with dementia has been associated with shortened survival, impaired quality of life, and increased caregiver distress.20 Pepper et al noted a trend toward increased incidence of postoperative delirium in their cohort of 234 PD patients undergoing surgery.7
Medication: Management of anti-Parkinsonian medications in the perioperative period poses unique challenges. These medications’ prodopaminergic effects can lead to hemodynamic compromise and are potentially arrhythmogenic. At the same time, abrupt withdrawal of these medications can lead to a potentially lethal condition called Parkinsonism-hyperpyrexia syndrome (PHS), which is clinically similar to neuroleptic malignant syndrome.21 PHS is characterized by very high fever, extreme muscle rigidity, autonomic instability, altered consciousness, and multiple severe systemic complications (e.g., acute renal failure, disseminated intravascular coagulation, autonomic failure, aspiration pneumonia, and infections). PHS occurs in up to 4% of PD patients; mortality is reported to be from 4% for treated to 20% for untreated episodes.22-24
As many as 30% of patients who survive a PHS episode have worsening of their PD symptoms and never return to their pre-PHS baseline. PHS prevention in hospitalized patients by uninterrupted administration of PD medications should be the goal. Early recognition and aggressive treatment is key to successful recovery.
Furthermore, even brief interruption of medications can lead to decompensation of Parkinsonian symptoms, which not only delays recovery from surgery, but also increases the risk for multisystem complications as discussed above.25 Traditional anti-Parkinsonian medications can only be delivered orally, presenting significant challenges for NPO patients, especially those undergoing enteric surgery requiring bowel rest.
Case reports describe various approaches to medication management in the perioperative period, but no single consensus statement (or treatment guideline) exists. The most common clinical scenarios are:
- Patient undergoes short surgery and is able to take oral medication immediately after the procedure (e.g., orthopedic, eye);
- Patient undergoes more lengthy surgery and will be able to take enteric medications perioperatively (e.g., longer orthopedic surgeries, genitourinary); and
- Patient undergoes lengthy procedure in which they will be required to adhere to bowel rest (e.g., bowel resection).
Depending on the category, the approach to medication management might differ.
Furuya et al describe a 70-year-old male with PD who had previously experienced perioperative complications associated with his medication management, including postoperative rigidity, dysphagia, and difficulty maintaining respiratory secretions.26 These symptoms were reversed with intravenous levodopa. However, the patient experienced hypotension and premature ventricular contractions as side effects of this therapy. This patient was scheduled to undergo hepatic lobectomy. Given his previous complications, Furuya et al provided enteral administration of CD/LD via nasogastric tube every two hours during the surgery, with placement of a duodenostomy for postoperative administration of CD/LD. The patient maintained hemodynamic stability throughout the perioperative period and emerged from anesthesia smoothly with no muscle rigidity or postoperative complications.26
Fujii et al described three cases of PD patients undergoing gastrointestinal surgery. They suggested that the dose of medication required to control symptoms should be minimized before surgery to avoid withdrawal symptoms. They also described the use of intravenous levodopa immediately, postoperatively, while the patient was unable to tolerate enteric medications, and suggested the use of prokinetics to prevent ileus and maximize drug absorption.27
Gálvez-Jiménez et al discuss the limitations of intravenous levodopa, including hemodynamic compromise, need for escalating doses, frequent adjustments to maintain effect, and large amount of fluids required. They propose the use of subcutaneous apomorphine, which is a potent D1/D2 dopamine agonist in conjunction with rectal domeperidone, a D2 antagonist with poor blood-brain-barrier penetration, to counteract the peripheral dopaminergic side effects. The main limitation to this approach is that domeperidone is not available in the U.S.28
Intravenous antihistamines and anticholinergics are readily available. However, they show limited efficacy in halting Parkinsonian symptoms and carry multiple side effects (e.g., confusion, delay in gastrointestinal recovery, and urinary retention).28
Various anesthesiologists agree with administering anti-PD medications immediately, preoperatively, and restarting medications as soon as possible postoperatively. However, they do not provide uniform treatment guidelines regarding specific medication management.29,30
Deep-brain-stimulation (DBS) management: DBS is an effective treatment for advanced PD. There are more than 60,000 patients around the world who have DBS for various conditions, mostly for PD. Therefore, it is increasingly likely that hospitalists will encounter hospitalized patients with advanced PD who are treated both pharmacologically and with DBS. It is important to recognize that stimulation, just like PD medications, cannot be stopped suddenly. If there is any concern of the DBS system malfunctioning (i.e., fracture of the hardware during a fall), the neurologist or neurosurgeon managing the DBS should be contacted immediately. Certain diagnostic tests (MRI) and treatment procedures (diathermy) are contraindicated, and if done inappropriately, can result in permanent brain damage.31,32,33
During surgeries requiring blood-vessel cauterization, DBS should be temporarily turned off. This can be done with the patient’s handheld device or, preferably, by a trained technician usually available through 24/7 technical support services provided by the manufacturer.
Summary of recommendations: There are no clear treatment guidelines regarding the optimal perioperative management of PD patients. The following measures are based on available data and are extensions of routine perioperative management; however, there is no evidence to demonstrate their efficacy in decreasing complication rates among patients with PD:
- A thorough preoperative history and physical examination should include Parkinsonian signs and symptoms, precise medication regimen with doses and timing of intake, effects of medication withdrawal or missed doses, type of surgery planned, and comorbid conditions;
- Depending on symptoms mentioned in the history, consider further testing for dysphagia (preoperative swallow evaluation) and dyspnea (preoperative pulmonary function tests);
- The major goal of medication management in the perioperative period is to continue administration of dopamine replacement therapy as close to the outpatient regimen as possible.
- Titrate down dose of anti-Parkinsonian medications to lowest possible dose prior to surgery if prolonged NPO status is anticipated;
- Ensure medications are administered immediately prior to surgery;
- For short, nonenteric procedures, resume outpatient medication doses and timing of administration as soon as possible postoperatively. For longer, nonenteric surgeries, consider placement of nasogastric tube for medication delivery during procedure and immediately postoperatively;
- If the major limitation of oral medication intake is dysphagia, the use of orally disintegrated formulation CD/LD (parcopa) is helpful;
- For longer enteric surgeries in which patient must be on bowel rest, recommend consultation with neurologist specialized in movement disorders to guide use of intravenous or subcutaneous agents. Transdermal delivery systems of the dopamine agonist rotigotine are in the process of being approved in the U.S. market and might be helpful for this purpose;
- Initiation of tube feeding, when co-administered with levodopa, might result in sudden changes in medication absorption and potential worsening of PD symptoms; feeding should be started slowly and preferably at night when the body’s dopamine requirements are lower;
- Consider use of promotility agents;
- If apomorphine or intravenous LD are not available, consider trial of intravenous anticholinergics or antihistamines, carefully observing for potential cognitive and behavioral side effects;
- Avoid such dopamine antagonists as droperidol, haloperidol, risperidone, metaclopramide, prochlorperazine, or promethazine, as these medications can worsen Parkinsonian symptoms; and
- If patient was on MAO-B inhibitors (selegiline, rasagiline) before surgery, be aware of multiple potential interactions with various medications that are commonly used in perioperative period, including anesthesia agents and certain analgesics, specifically meperidine. These interactions include serotonin syndrome, and can be life-threatening.34
- Psychiatric considerations: delirium precautions;
- Motor considerations: early PT/OT, early referral to inpatient rehabilitation; fall precautions;
- Pulmonary considerations: institute aggressive incentive spirometry, postural drainage, management of respiratory secretions, and breathing exercises; VTE prophylaxis;
- Gastrointestinal considerations: aspiration precautions and prompt speech therapy to evaluate for aspiration and to teach appropriate swallow techniques (chin tuck); institute aggressive bowel regimen; maximize fluids, electrolytes, and avoid narcotics to prevent precipitating or exacerbating ileus;
- Cardiovascular considerations: monitor orthostatic vital signs; fall precautions to avoid syncopal falls; and
- Genitourinary considerations: early urinary catheter removal; vigilance in monitoring for urinary tract infection.
Back to the Case
The patient underwent repair of her fracture, was extubated, and recovered from general anesthesia without incident. She was evaluated in the postanesthesia care unit, at which time she had a slight tremor and mild rigidity. She was immediately given a dose of her CD/LD, and her evening doses of amantadine and ropinirole were resumed. The patient had no significant flare of her Parkinsonian symptoms and did not exhibit any evidence of PHS.
A postoperative consultation was placed for speech therapy, physical therapy, and occupational therapy. She was given low-molecular-weight heparin for VTE prophylaxis and asked to use incentive spirometry. On postoperative day one, she complained of urinary frequency. A urinalysis was consistent with possible infection. She was discharged home on her previous medication regimen, in addition to antibiotics for cystitis.
If the procedure had not been emergent, the patient might have benefited from a preoperative swallow evaluation, given her dysphagia. Consultation with a speech therapist would have ensured that the patient was educated regarding aspiration precautions. Although this patient did not have difficulty with extubation or experience postoperative respiratory failure, abnormal preoperative pulmonary function tests might have prompted the anesthesiologists to consider alternative low-risk techniques (e.g., a local nerve block).
Bottom Line
Perioperative management of patients with Parkinson’s disease requires knowledge of the multisystem disease characteristics that raise perioperative risk and the effects of the medications used to treat PD. To date, no clear treatment guidelines exist for the optimal perioperative management of PD patients.
However, vigilance in detecting possible complications and instituting attentive perioperative care can aid a hospitalist consultant in improving overall care for these patients. TH
Dr. Patel is a medical resident at the University of Colorado Denver. Dr. Stickrath is a hospitalist at the Denver VA Medical Center and instructor of medicine at the University of Colorado Denver. Dr. Anderson is a hospitalist at the Denver VA Medical Center and assistant professor of medicine at the University of Colorado Denver. Dr. Klepitskaya is a neurologist and assistant professor of neurology at the University of Colorado Denver.
References
- Ersan T. Perioperative management of the geriatric patient. Web MD website. Available at: http://emedicine.medscape.com/article/285433-overview. Accessed May 10, 2010.
- Braak H, Del Tredici K, Rüb U, de Vos RA, Jansen Steur EN, Braak E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol Aging. 2003;24(2):197-211.
- Ward CD, Gibb WR. Research diagnostic criteria for Parkinson’s disease. Adv Neurol. 1990;53:245-249.
- Stern MB, Koller WC. Parkinsonian Syndromes. New York: Informa Healthcare Publishing; 1993.
- Nutt JG, Wooten GF. Clinical practice. Diagnosis and initial management of Parkinson’s disease. N Engl J Med. 2005;353(10):1021-1027.
- Mueller MC, Jüptner U, Wuellner U, et al. Parkinson’s disease influences the perioperative risk profile in surgery. Langenbecks Arch Surg. 2009;394(3):511-515.
- Pepper PV, Goldstein MK. Postoperative complications in Parkinson’s disease. J Am Geriatr Soc. 1999;47(8):967-972.
- Shill H, Stacy M. Respiratory function in Parkinson’s disease. Clin Neurosci. 1998;5(2):131-135.
- Neu HC, Connolly JJ Jr., Schwertley FW, Ladwig HA, Brody AW. Obstructive respiratory dysfunction in parkinsonian patients. Ann Rev Respir Dis. 1967;95(1):33-47.
- Chokroverty S. The spectrum of ventilatory disturbances in movement disorders. In: Chokroverty S, ed. Movement Disorders. Great Neck, New York: PMA Publishing Corp.; 1990:365-392.
- Shill H, Stacy M. Respiratory complications of Parkinson’s disease. Semin Respir Crit Care Med. 2002;23(3):261-265.
- Easdown LJ, Tessler KJ, Minuk J. Upper airway involvement in Parkinson’s disease resulting in postoperative respiratory failure. Can J Anaesth. 1995;42(4):344-347.
- Pfeiffer RF. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2003;2(2):107-116.
- Castell JA, Johnston BT, Colcher A, Li Q, Gideon RM, Castell DO. Manometric abnormalities of the oesophagus in patients with Parkinson’s disease. Neurogastroenterol Motil. 2001;13(4):361-364.
- Marcason W. What are the primary nutritional issues for a patient with Parkinson’s disease? J Am Diet Assoc. 2009;109(7):1316.
- Djaldetti R, Lev N, Melamed E. Lesions outside the CNS in Parkinson’s disease. Mov Disord. 2009;24(6):793-800.
- Orimo S, Takahashi A, Uchihara T, Mori F, Kakita A, Wakabayashi K, Takahashi H. Degeneration of cardiac sympathetic nerve begins in the early disease process of Parkinson’s disease. Brain Pathol. 2007;17(1):24-30.
- Lemack GE, Dewey RB Jr., Roehrborn CG, O’Suilleabhain PE, Zimmern PE. Questionnaire-based assessment of bladder dysfunction in patients with mild to moderate Parkinson’s disease. Urology. 2000;56(2):250-254.
- Singer C. Urinary dysfunction in Parkinson’s disease. Clin Neurosci. 1998;5(2):78-86.
- Leverenz JB, Quinn JF, Zabetian C, Zhang J, Montine KS, Montine TJ. Cognitive impairment and dementia in patients with Parkinson disease. Curr Top Med Chem. 2009;9(10):903-912.
- Factor SA, Santiago A. Parkinson-hyperpyrexia syndrome in Parkinson’s disease. In: Frucht S, Fahn S, eds. Movement Disorder Emergencies: Diagnosis and Treatment. New York: Humana Press; 2005:29-40.
- Reed AP, Han DG: Intraoperative exacerbation of Parkinson’s disease. Anesth Analg. 1992;75(5):850-853.
- Harada T, Mitsuoka K, Kumagai R, et al. Clinical features of malignant syndrome in Parkinson’s disease and related neurological disorders. Parkinsonism Relat Disord. 2003;9(Suppl 1):S15-23.
- Onofrj M, Thomas A. Acute akinesia in Parkinson disease. Neurology. 2005;64(7):1162-1169.
- Serrano-Dueñas M. Neuroleptic malignant syndrome-like, or—dopaminergic malignant syndrome—due to levodopa therapy withdrawal. Clinical features in 11 patients. Parkinsonism Relat Disord. 2003;9(3):175-178.
- Furuya R, Hirai A, Andoh T, Kudoh I, Okumura F. Successful perioperative management of a patient with Parkinson’s disease by enteral levodopa administration under propofol anesthesia. Anesthesiology. 1998;89(1):261-263.
- Fujii T, Nakabayashi T, Hashimoto S, Kuwano H. Successful perioperative management of patients with Parkinson’s disease following gastrointestinal surgery: report of three cases. Surg Today. 2009;39(9):807-810.
- Gálvez-Jiménez N, Lang AE. The perioperative management of Parkinson’s disease revisited. Neurol Clin. 2004;22(2):367-377.
- Kalenka A, Schwarz A. Anasthesia and Parkinson’s disease: how to manage with new therapies? Curr Opin Anesthesiol. 2009;22(3):419-424.
- Nicholson G, Pereira AC, Hall GM. Parkinson’s disease and anesthesia. Br J Anaesthesia. 2002;89(6):904-916.
- Rezai AR, Baker KB, Tkach JA, et al. Is magnetic resonance imaging safe for patients with neurostimulation systems used for deep brain stimulation? Neurosurgery. 2005:57(5):1056-1062.
- Henderson JM, Tkach J, Phillips M, Baker K, Shellock FG, Rezai AR. Permanent neurological deficit related to magnetic resonance imaging in a patient with implanted deep brain stimulation electrodes for Parkinson’s disease: case report. Neurosurgery. 2005:57(5):E1063.
- Nutt JG, Anderson VC, Peacock JH, Hammerstad JP, Burchiel KJ. DBS and diathermy interaction induces severe CNS damage. Neurology. 2001;56 (10):1384-1386.
- Gordon PH, Frucht SJ. Neuroleptic malignant syndrome in advanced Parkinson’s disease. Mov Disord. 2001;16(5):960-962.
ONLINE EXCLUSIVE: Audio interview with Ethan Fried, MD, MS
The Cost of Regulation
The impact of last summer’s new restrictions from the Accreditation Council for Graduate Medical Education (ACGME) on how many hospitalized patients a first-year resident can treat on an internal-medicine (IM) rotation was as immediate as it was evident at Monmouth Medical Center, a 527-bed teaching hospital in Long Branch, N.J. The institution had a class of eight rookie residents whose caseloads were cut from 12 to the new threshold of 10.
Physicians “had to find some other way of getting attention . . . for 16 patients,” says Sarah Wallach, MD, FACP, director of Monmouth’s IM residency program and vice chair of the department of medicine at the hospital. At Monmouth, the solution came in the form of a new hire—a nurse practitioner (NP)—to handle the overflow. The NP service is used predominantly for referral patients from primary-care physicians (PCPs), as opposed to independent hospital admissions.
But because the NP service does not provide 24-hour coverage, the hospital can get away with only one person in the position. To extend coverage all day long, Dr. Wallach estimates she would need to hire two or three additional NPs, plus another one or two administrative positions to provide relief on holidays and vacations. “You would need five people,” she says. “I can’t afford that.”
Few hospitals or HM groups can afford new hires in today’s world of Medicare reimbursement cuts, shrinking budgets, and—courtesy of the newest rules—restricting patient caps for residents. The latest rules took hold about a year ago, but hospitalists in both academic and community settings say the impact already is noticeable.
Many hospitals have had to craft solutions, which have included burdening academic hospitals with more clinical responsibilities, turning to private HM groups (HMGs) to assume the patients residents can no longer care for, or hiring nonphysician providers (NPPs) to pick up the slack. As Dr. Wallach pointedly notes, the latter two solutions cost money at a time when hospitals have less to go around.
Already, teaching hospitals have begun discussions about how the newest rules—and the future changes they presage—will change the playing field. Will a wave of academics flee their classroom (the teaching hospital), as nonteaching duties become an intrusion? Will teaching hospitals face financial pressure as they struggle to replace the low-cost labor force that residents represent?
Perhaps most importantly from a medical perspective, will graduate trainees be as prepared as their predecessors when they enter practice?
The answers will have a direct correlation to private HMGs, which are poised to see more patients in the wake of residency restrictions, particularly on overnight services. The cost of hospital care will increase for hospitals, putting more pressure on hospitalist groups that tout themselves to C-suites as engines for cost savings. Long-term implications, unfortunately, remain murky, as the newest rules have been in place for a relatively short time. Plus, ACGME is expected—at the end of this month, according to a recent memo to program directors—to announce more changes to residency guidelines.
“Hospitalists will always be involved in teaching—it will never go away,” says Julia Wright, MD, FHM, clinical professor of medicine and director of hospital medicine at the University of Wisconsin School of Medicine and Public Health in Madison and a member of Team Hospitalist. “But it will be a very different balance, a different kind of feel.”
The Past to the Future
To understand the concerns moving forward, it’s important to first look back. In July 2003, new ACGME rules went into place capping the workweeks of residents at 80 hours. Rules were put into place that regulated the number of patients that residents could be assigned, and those thresholds were further tightened on July 1, 2009. The most notable 2009 change: A first-year resident’s patient census must not exceed 10 patients. ACGME CEO Thomas J. Nasca, MD, MACP, sent a letter to program directors in early May announcing more changes to resident work hours. The letter indicates proposals will be announced by the end of this month, and public comment will follow. At the earliest, new rules changes would go into effect in 2011. “The board may adopt a modification to the duty-hours standard,” says Julie Jacob, a spokeswoman for Chicago-based ACGME. “Any proposed standards would get a public comment.”
Jacob declined further comment, but various hospitalists and academics say they wouldn’t be surprised if new rules reflect 2008 Institute of Medicine (IOM) recommendations.1 The IOM report called for a maximum resident shift length of 30 hours, with admission of patients for up to 16 hours, plus a five-hour uninterrupted sleep period between 10 p.m. and 8 a.m. It also suggested the remaining workweek hours be used for transitional and educational activities.
However those IOM recommendations are incorporated, one thing is clear: Any adoption of those standards will have a financial impact. In fact, a study published last year reported that annual labor costs from implementing the IOM standards was estimated to be $1.6 billion in 2006 dollars (see “The Cost of Progress,” p. 25).2
“Any replacement of a resident costs more than a resident, whether it’s an NP, a PA (physician assistant), an MD, or a DO,” says Kevin O’Leary, MD, MS, associate program director of the IM residency program at Northwestern University’s Feinberg School of Medicine in Chicago. “Everybody costs more.”
The Fate of Teachers
Some of the largest academic centers, including the Feinberg School, the University of Michigan, and the teaching service at St. Luke’s-Roosevelt Hospital in New York City, reduced patient caseloads ahead of the 2009 round of residency rule changes. Hospitalists and educators at those institutions say the proactive approach helped them adjust to the newest rules, which by some estimates reduce resident productivity by 20%.
But the changes shift the workload to academic hospitalists, many of whom forego higher-paying positions to pursue teaching and research. According to the latest SHM survey data, academic hospitalists make about $50,000 less per year than the average community hospitalist. But as clinical work intrudes further, as residents are unable to assume the patient care they once did, educators are put into positions of having to balance the educational portion of their job with patient care, says John Del Valle, MD, professor and residency program director in the department of internal medicine at the University of Michigan Health System in Ann Arbor.
“This is where difficult decisions have to be made,” Dr. Del Valle says. “This is not the blend of activities that traditional academics signed up for.”
Solutions to relieve current and impending pressure on teaching hospitalists have presented themselves in different ways. In Dr. Del Valle’s hospital, there is a split between the hospitalist service and the house staff, which is aimed at keeping up with the growth in IM admissions. That tally has climbed an average of 4% per year for the past five years, reaching some 18,000 admissions last year. To handle that workload, the nonresident service last year added three clinical full-time equivalents (FTEs) to bring its total to nearly 30 FTEs.
Dr. Del Valle notes his institution has been fortunate to be able to afford growth, thanks in large part to a payor mix with a relatively low percentage of charity care and high level of activity.
At Brigham and Women’s Hospital in Boston, the answer is a freestanding PA service that has been in place since 2005. Last summer, the program went to a 24-hour rotation to increase continuity for overnight services and to provide coverage on night shifts, an area most in the industry agree will be hit hardest by the resident caps. Physicians at Brigham’s, a teaching affiliate of Harvard Medical School, are now discussing an expansion of the PA service, or perhaps even an overhaul to a more cost-efficient solution, says Danielle Scheurer, MD, MSc, FHM, assistant professor of medicine at Harvard and director of Brigham’s general medicine service.
At Medical Center Hospital (MCH) in Odessa, Texas, the hospitalists were added to the ED call schedule once every five nights. The plan was under discussion before the new residency rules went into place; however, it was implemented to keep the IM residency program within the new limits, says Bruce Becker, MD, MCH’s chief medical officer.
And at St. Luke’s-Roosevelt Hospital, discussions are under way on how to best extend the nonteaching staff, says Ethan Fried, MD, MS, FACP, assistant professor of clinical medicine at Columbia University, vice chair for education in the department of medicine and director of graduate medical education at St. Luke’s-Roosevelt. “The adjustment has to come from the nonteaching side because the house staff at this point is saturated,” says Dr. Fried, president-elect of the Association of Program Directors in Internal Medicine (APDIM). “You can’t be cheap about acquiring your nonteaching staff.”
The Fate of Students
Perhaps paramount to the fears of how teaching hospitalists will react to current or future restrictions is the effect those limits have on the residents they safeguard. Some physicians think the new rules will produce crops of ill-prepared residents because they have been coddled with limited patient censuses. Other physicians argue that the new thresholds will actually better prepare physicians when HM groups are hiring residents for full-time positions.
Dr. Del Valle acknowledges there is as yet no rigorous data to show the impact of the current restrictions, but he agrees it’s a simple equation of patient-care mathematics. “You can’t [easily] replace 100-110 hours [of care per week],” he says.
Others say patient caps and rules to limit how much work residents do are in line with the purpose of medical training programs. “I’ve bought into the fact that these programs exist to train residents, not to provide clinical care,” Dr. O’Leary says. “I’ve drunk that Kool-Aid. … I think there’s more variation, person to person, than ‘my era vs. the current era.’ Like any new hospitalist that you hire, you need to give an orientation and give enough support to them so when they begin to see patients that they are not overwhelmed.”
Shaun Frost, MD, FACP, FHM, might be best described as halfway between those two extremes. A regional director for the eastern U.S. for Cogent Healthcare, he says duty-hour restrictions have had deleterious impacts but also create learning opportunities.
“The residency work-hour restrictions have inhibited our ability to train people to work as efficiently as trainees who were taught in the past,” says Dr. Frost, an SHM board member. “That doesn’t necessarily mean you can’t teach people to work more efficiently . . . but in the future, my hope is that residency training programs will recognize the deficit that exists in personal work efficiencies between their completion and their responsibilities as a hospitalist.”
To that end, Dr. Frost works with others to develop both structured curriculum and classroom didactics that help new hospitalists make up for gaps in preparation that weren’t addressed in residency. In some cases, that can be practice management and billing issues, but often, according to Dr. Frost, it is addressing personal workflow and bridging the “unnatural discontinuity” in patient care from residency to the real world.
“There is a cost to this investment for the future,” Dr. Frost adds. “If people don’t recognize the potential return on investment as being critical to the development of an educated workforce—an efficient and competent workforce—and thus critical to the retention of high-performing hospitalists, they are selling themselves, unfortunately, significantly short.”
Caught in the Middle
One man’s trash is another man’s treasure, the axiom tells us. Well, in healthcare circles, that could just as easily read: The woes of academic hospitalists are the wealth of community hospitalists.
The new rules “may result in more opportunities for hospitalists to provide needed clinical services,” Dr. Wright says.
The long-term implications, though, remain to be seen. While academic hospitalists say they have seen preliminary increases in care-delivery costs because of the latest rules changes, many say it’s too soon to tell just how high those costs might climb and what ripple effect might follow.
Some physicians, including Dr. Del Valle, note that while the 2009 changes and the expectation of more changes in 2011 are cause for attention, that doesn’t translate to cause for concern. In 2003, months before the 80-hour workweek rules were first put in place by ACGME, many of the same debates were already under way: How will the faculty of IM residency programs cope? How will institutions pay the bills while putting money aside for other physicians picking up the slack?
“This is a pendulum,” Dr. Del Valle says. “I think it will come back to a balanced place.”
Dr. Fried, who is more optimistic that the residency rules can have a positive, long-term effect, agrees. He says residency caps and limits should not be viewed as “things that limit education. We [should] look at them as things that ensure education continues while patient care continues.” TH
Richard Quinn is a freelance writer based in New Jersey.
References
- Institute of Medicine. Resident Duty Hours: Enhancing Sleep, Supervision, and Safety. Ulmer C, Wolman DM, Johns MM, eds. Washington, D.C.: The National Academies Press; 2008.
- Nuckols TK, Bhattacharya J, Wolman DM, Ulmer C, Escarce JJ. Cost implications of reduced work hours and workloads for resident physicians. N Engl J Med. 2009:360(21):2202-2215.
The impact of last summer’s new restrictions from the Accreditation Council for Graduate Medical Education (ACGME) on how many hospitalized patients a first-year resident can treat on an internal-medicine (IM) rotation was as immediate as it was evident at Monmouth Medical Center, a 527-bed teaching hospital in Long Branch, N.J. The institution had a class of eight rookie residents whose caseloads were cut from 12 to the new threshold of 10.
Physicians “had to find some other way of getting attention . . . for 16 patients,” says Sarah Wallach, MD, FACP, director of Monmouth’s IM residency program and vice chair of the department of medicine at the hospital. At Monmouth, the solution came in the form of a new hire—a nurse practitioner (NP)—to handle the overflow. The NP service is used predominantly for referral patients from primary-care physicians (PCPs), as opposed to independent hospital admissions.
But because the NP service does not provide 24-hour coverage, the hospital can get away with only one person in the position. To extend coverage all day long, Dr. Wallach estimates she would need to hire two or three additional NPs, plus another one or two administrative positions to provide relief on holidays and vacations. “You would need five people,” she says. “I can’t afford that.”
Few hospitals or HM groups can afford new hires in today’s world of Medicare reimbursement cuts, shrinking budgets, and—courtesy of the newest rules—restricting patient caps for residents. The latest rules took hold about a year ago, but hospitalists in both academic and community settings say the impact already is noticeable.
Many hospitals have had to craft solutions, which have included burdening academic hospitals with more clinical responsibilities, turning to private HM groups (HMGs) to assume the patients residents can no longer care for, or hiring nonphysician providers (NPPs) to pick up the slack. As Dr. Wallach pointedly notes, the latter two solutions cost money at a time when hospitals have less to go around.
Already, teaching hospitals have begun discussions about how the newest rules—and the future changes they presage—will change the playing field. Will a wave of academics flee their classroom (the teaching hospital), as nonteaching duties become an intrusion? Will teaching hospitals face financial pressure as they struggle to replace the low-cost labor force that residents represent?
Perhaps most importantly from a medical perspective, will graduate trainees be as prepared as their predecessors when they enter practice?
The answers will have a direct correlation to private HMGs, which are poised to see more patients in the wake of residency restrictions, particularly on overnight services. The cost of hospital care will increase for hospitals, putting more pressure on hospitalist groups that tout themselves to C-suites as engines for cost savings. Long-term implications, unfortunately, remain murky, as the newest rules have been in place for a relatively short time. Plus, ACGME is expected—at the end of this month, according to a recent memo to program directors—to announce more changes to residency guidelines.
“Hospitalists will always be involved in teaching—it will never go away,” says Julia Wright, MD, FHM, clinical professor of medicine and director of hospital medicine at the University of Wisconsin School of Medicine and Public Health in Madison and a member of Team Hospitalist. “But it will be a very different balance, a different kind of feel.”
The Past to the Future
To understand the concerns moving forward, it’s important to first look back. In July 2003, new ACGME rules went into place capping the workweeks of residents at 80 hours. Rules were put into place that regulated the number of patients that residents could be assigned, and those thresholds were further tightened on July 1, 2009. The most notable 2009 change: A first-year resident’s patient census must not exceed 10 patients. ACGME CEO Thomas J. Nasca, MD, MACP, sent a letter to program directors in early May announcing more changes to resident work hours. The letter indicates proposals will be announced by the end of this month, and public comment will follow. At the earliest, new rules changes would go into effect in 2011. “The board may adopt a modification to the duty-hours standard,” says Julie Jacob, a spokeswoman for Chicago-based ACGME. “Any proposed standards would get a public comment.”
Jacob declined further comment, but various hospitalists and academics say they wouldn’t be surprised if new rules reflect 2008 Institute of Medicine (IOM) recommendations.1 The IOM report called for a maximum resident shift length of 30 hours, with admission of patients for up to 16 hours, plus a five-hour uninterrupted sleep period between 10 p.m. and 8 a.m. It also suggested the remaining workweek hours be used for transitional and educational activities.
However those IOM recommendations are incorporated, one thing is clear: Any adoption of those standards will have a financial impact. In fact, a study published last year reported that annual labor costs from implementing the IOM standards was estimated to be $1.6 billion in 2006 dollars (see “The Cost of Progress,” p. 25).2
“Any replacement of a resident costs more than a resident, whether it’s an NP, a PA (physician assistant), an MD, or a DO,” says Kevin O’Leary, MD, MS, associate program director of the IM residency program at Northwestern University’s Feinberg School of Medicine in Chicago. “Everybody costs more.”
The Fate of Teachers
Some of the largest academic centers, including the Feinberg School, the University of Michigan, and the teaching service at St. Luke’s-Roosevelt Hospital in New York City, reduced patient caseloads ahead of the 2009 round of residency rule changes. Hospitalists and educators at those institutions say the proactive approach helped them adjust to the newest rules, which by some estimates reduce resident productivity by 20%.
But the changes shift the workload to academic hospitalists, many of whom forego higher-paying positions to pursue teaching and research. According to the latest SHM survey data, academic hospitalists make about $50,000 less per year than the average community hospitalist. But as clinical work intrudes further, as residents are unable to assume the patient care they once did, educators are put into positions of having to balance the educational portion of their job with patient care, says John Del Valle, MD, professor and residency program director in the department of internal medicine at the University of Michigan Health System in Ann Arbor.
“This is where difficult decisions have to be made,” Dr. Del Valle says. “This is not the blend of activities that traditional academics signed up for.”
Solutions to relieve current and impending pressure on teaching hospitalists have presented themselves in different ways. In Dr. Del Valle’s hospital, there is a split between the hospitalist service and the house staff, which is aimed at keeping up with the growth in IM admissions. That tally has climbed an average of 4% per year for the past five years, reaching some 18,000 admissions last year. To handle that workload, the nonresident service last year added three clinical full-time equivalents (FTEs) to bring its total to nearly 30 FTEs.
Dr. Del Valle notes his institution has been fortunate to be able to afford growth, thanks in large part to a payor mix with a relatively low percentage of charity care and high level of activity.
At Brigham and Women’s Hospital in Boston, the answer is a freestanding PA service that has been in place since 2005. Last summer, the program went to a 24-hour rotation to increase continuity for overnight services and to provide coverage on night shifts, an area most in the industry agree will be hit hardest by the resident caps. Physicians at Brigham’s, a teaching affiliate of Harvard Medical School, are now discussing an expansion of the PA service, or perhaps even an overhaul to a more cost-efficient solution, says Danielle Scheurer, MD, MSc, FHM, assistant professor of medicine at Harvard and director of Brigham’s general medicine service.
At Medical Center Hospital (MCH) in Odessa, Texas, the hospitalists were added to the ED call schedule once every five nights. The plan was under discussion before the new residency rules went into place; however, it was implemented to keep the IM residency program within the new limits, says Bruce Becker, MD, MCH’s chief medical officer.
And at St. Luke’s-Roosevelt Hospital, discussions are under way on how to best extend the nonteaching staff, says Ethan Fried, MD, MS, FACP, assistant professor of clinical medicine at Columbia University, vice chair for education in the department of medicine and director of graduate medical education at St. Luke’s-Roosevelt. “The adjustment has to come from the nonteaching side because the house staff at this point is saturated,” says Dr. Fried, president-elect of the Association of Program Directors in Internal Medicine (APDIM). “You can’t be cheap about acquiring your nonteaching staff.”
The Fate of Students
Perhaps paramount to the fears of how teaching hospitalists will react to current or future restrictions is the effect those limits have on the residents they safeguard. Some physicians think the new rules will produce crops of ill-prepared residents because they have been coddled with limited patient censuses. Other physicians argue that the new thresholds will actually better prepare physicians when HM groups are hiring residents for full-time positions.
Dr. Del Valle acknowledges there is as yet no rigorous data to show the impact of the current restrictions, but he agrees it’s a simple equation of patient-care mathematics. “You can’t [easily] replace 100-110 hours [of care per week],” he says.
Others say patient caps and rules to limit how much work residents do are in line with the purpose of medical training programs. “I’ve bought into the fact that these programs exist to train residents, not to provide clinical care,” Dr. O’Leary says. “I’ve drunk that Kool-Aid. … I think there’s more variation, person to person, than ‘my era vs. the current era.’ Like any new hospitalist that you hire, you need to give an orientation and give enough support to them so when they begin to see patients that they are not overwhelmed.”
Shaun Frost, MD, FACP, FHM, might be best described as halfway between those two extremes. A regional director for the eastern U.S. for Cogent Healthcare, he says duty-hour restrictions have had deleterious impacts but also create learning opportunities.
“The residency work-hour restrictions have inhibited our ability to train people to work as efficiently as trainees who were taught in the past,” says Dr. Frost, an SHM board member. “That doesn’t necessarily mean you can’t teach people to work more efficiently . . . but in the future, my hope is that residency training programs will recognize the deficit that exists in personal work efficiencies between their completion and their responsibilities as a hospitalist.”
To that end, Dr. Frost works with others to develop both structured curriculum and classroom didactics that help new hospitalists make up for gaps in preparation that weren’t addressed in residency. In some cases, that can be practice management and billing issues, but often, according to Dr. Frost, it is addressing personal workflow and bridging the “unnatural discontinuity” in patient care from residency to the real world.
“There is a cost to this investment for the future,” Dr. Frost adds. “If people don’t recognize the potential return on investment as being critical to the development of an educated workforce—an efficient and competent workforce—and thus critical to the retention of high-performing hospitalists, they are selling themselves, unfortunately, significantly short.”
Caught in the Middle
One man’s trash is another man’s treasure, the axiom tells us. Well, in healthcare circles, that could just as easily read: The woes of academic hospitalists are the wealth of community hospitalists.
The new rules “may result in more opportunities for hospitalists to provide needed clinical services,” Dr. Wright says.
The long-term implications, though, remain to be seen. While academic hospitalists say they have seen preliminary increases in care-delivery costs because of the latest rules changes, many say it’s too soon to tell just how high those costs might climb and what ripple effect might follow.
Some physicians, including Dr. Del Valle, note that while the 2009 changes and the expectation of more changes in 2011 are cause for attention, that doesn’t translate to cause for concern. In 2003, months before the 80-hour workweek rules were first put in place by ACGME, many of the same debates were already under way: How will the faculty of IM residency programs cope? How will institutions pay the bills while putting money aside for other physicians picking up the slack?
“This is a pendulum,” Dr. Del Valle says. “I think it will come back to a balanced place.”
Dr. Fried, who is more optimistic that the residency rules can have a positive, long-term effect, agrees. He says residency caps and limits should not be viewed as “things that limit education. We [should] look at them as things that ensure education continues while patient care continues.” TH
Richard Quinn is a freelance writer based in New Jersey.
References
- Institute of Medicine. Resident Duty Hours: Enhancing Sleep, Supervision, and Safety. Ulmer C, Wolman DM, Johns MM, eds. Washington, D.C.: The National Academies Press; 2008.
- Nuckols TK, Bhattacharya J, Wolman DM, Ulmer C, Escarce JJ. Cost implications of reduced work hours and workloads for resident physicians. N Engl J Med. 2009:360(21):2202-2215.
The impact of last summer’s new restrictions from the Accreditation Council for Graduate Medical Education (ACGME) on how many hospitalized patients a first-year resident can treat on an internal-medicine (IM) rotation was as immediate as it was evident at Monmouth Medical Center, a 527-bed teaching hospital in Long Branch, N.J. The institution had a class of eight rookie residents whose caseloads were cut from 12 to the new threshold of 10.
Physicians “had to find some other way of getting attention . . . for 16 patients,” says Sarah Wallach, MD, FACP, director of Monmouth’s IM residency program and vice chair of the department of medicine at the hospital. At Monmouth, the solution came in the form of a new hire—a nurse practitioner (NP)—to handle the overflow. The NP service is used predominantly for referral patients from primary-care physicians (PCPs), as opposed to independent hospital admissions.
But because the NP service does not provide 24-hour coverage, the hospital can get away with only one person in the position. To extend coverage all day long, Dr. Wallach estimates she would need to hire two or three additional NPs, plus another one or two administrative positions to provide relief on holidays and vacations. “You would need five people,” she says. “I can’t afford that.”
Few hospitals or HM groups can afford new hires in today’s world of Medicare reimbursement cuts, shrinking budgets, and—courtesy of the newest rules—restricting patient caps for residents. The latest rules took hold about a year ago, but hospitalists in both academic and community settings say the impact already is noticeable.
Many hospitals have had to craft solutions, which have included burdening academic hospitals with more clinical responsibilities, turning to private HM groups (HMGs) to assume the patients residents can no longer care for, or hiring nonphysician providers (NPPs) to pick up the slack. As Dr. Wallach pointedly notes, the latter two solutions cost money at a time when hospitals have less to go around.
Already, teaching hospitals have begun discussions about how the newest rules—and the future changes they presage—will change the playing field. Will a wave of academics flee their classroom (the teaching hospital), as nonteaching duties become an intrusion? Will teaching hospitals face financial pressure as they struggle to replace the low-cost labor force that residents represent?
Perhaps most importantly from a medical perspective, will graduate trainees be as prepared as their predecessors when they enter practice?
The answers will have a direct correlation to private HMGs, which are poised to see more patients in the wake of residency restrictions, particularly on overnight services. The cost of hospital care will increase for hospitals, putting more pressure on hospitalist groups that tout themselves to C-suites as engines for cost savings. Long-term implications, unfortunately, remain murky, as the newest rules have been in place for a relatively short time. Plus, ACGME is expected—at the end of this month, according to a recent memo to program directors—to announce more changes to residency guidelines.
“Hospitalists will always be involved in teaching—it will never go away,” says Julia Wright, MD, FHM, clinical professor of medicine and director of hospital medicine at the University of Wisconsin School of Medicine and Public Health in Madison and a member of Team Hospitalist. “But it will be a very different balance, a different kind of feel.”
The Past to the Future
To understand the concerns moving forward, it’s important to first look back. In July 2003, new ACGME rules went into place capping the workweeks of residents at 80 hours. Rules were put into place that regulated the number of patients that residents could be assigned, and those thresholds were further tightened on July 1, 2009. The most notable 2009 change: A first-year resident’s patient census must not exceed 10 patients. ACGME CEO Thomas J. Nasca, MD, MACP, sent a letter to program directors in early May announcing more changes to resident work hours. The letter indicates proposals will be announced by the end of this month, and public comment will follow. At the earliest, new rules changes would go into effect in 2011. “The board may adopt a modification to the duty-hours standard,” says Julie Jacob, a spokeswoman for Chicago-based ACGME. “Any proposed standards would get a public comment.”
Jacob declined further comment, but various hospitalists and academics say they wouldn’t be surprised if new rules reflect 2008 Institute of Medicine (IOM) recommendations.1 The IOM report called for a maximum resident shift length of 30 hours, with admission of patients for up to 16 hours, plus a five-hour uninterrupted sleep period between 10 p.m. and 8 a.m. It also suggested the remaining workweek hours be used for transitional and educational activities.
However those IOM recommendations are incorporated, one thing is clear: Any adoption of those standards will have a financial impact. In fact, a study published last year reported that annual labor costs from implementing the IOM standards was estimated to be $1.6 billion in 2006 dollars (see “The Cost of Progress,” p. 25).2
“Any replacement of a resident costs more than a resident, whether it’s an NP, a PA (physician assistant), an MD, or a DO,” says Kevin O’Leary, MD, MS, associate program director of the IM residency program at Northwestern University’s Feinberg School of Medicine in Chicago. “Everybody costs more.”
The Fate of Teachers
Some of the largest academic centers, including the Feinberg School, the University of Michigan, and the teaching service at St. Luke’s-Roosevelt Hospital in New York City, reduced patient caseloads ahead of the 2009 round of residency rule changes. Hospitalists and educators at those institutions say the proactive approach helped them adjust to the newest rules, which by some estimates reduce resident productivity by 20%.
But the changes shift the workload to academic hospitalists, many of whom forego higher-paying positions to pursue teaching and research. According to the latest SHM survey data, academic hospitalists make about $50,000 less per year than the average community hospitalist. But as clinical work intrudes further, as residents are unable to assume the patient care they once did, educators are put into positions of having to balance the educational portion of their job with patient care, says John Del Valle, MD, professor and residency program director in the department of internal medicine at the University of Michigan Health System in Ann Arbor.
“This is where difficult decisions have to be made,” Dr. Del Valle says. “This is not the blend of activities that traditional academics signed up for.”
Solutions to relieve current and impending pressure on teaching hospitalists have presented themselves in different ways. In Dr. Del Valle’s hospital, there is a split between the hospitalist service and the house staff, which is aimed at keeping up with the growth in IM admissions. That tally has climbed an average of 4% per year for the past five years, reaching some 18,000 admissions last year. To handle that workload, the nonresident service last year added three clinical full-time equivalents (FTEs) to bring its total to nearly 30 FTEs.
Dr. Del Valle notes his institution has been fortunate to be able to afford growth, thanks in large part to a payor mix with a relatively low percentage of charity care and high level of activity.
At Brigham and Women’s Hospital in Boston, the answer is a freestanding PA service that has been in place since 2005. Last summer, the program went to a 24-hour rotation to increase continuity for overnight services and to provide coverage on night shifts, an area most in the industry agree will be hit hardest by the resident caps. Physicians at Brigham’s, a teaching affiliate of Harvard Medical School, are now discussing an expansion of the PA service, or perhaps even an overhaul to a more cost-efficient solution, says Danielle Scheurer, MD, MSc, FHM, assistant professor of medicine at Harvard and director of Brigham’s general medicine service.
At Medical Center Hospital (MCH) in Odessa, Texas, the hospitalists were added to the ED call schedule once every five nights. The plan was under discussion before the new residency rules went into place; however, it was implemented to keep the IM residency program within the new limits, says Bruce Becker, MD, MCH’s chief medical officer.
And at St. Luke’s-Roosevelt Hospital, discussions are under way on how to best extend the nonteaching staff, says Ethan Fried, MD, MS, FACP, assistant professor of clinical medicine at Columbia University, vice chair for education in the department of medicine and director of graduate medical education at St. Luke’s-Roosevelt. “The adjustment has to come from the nonteaching side because the house staff at this point is saturated,” says Dr. Fried, president-elect of the Association of Program Directors in Internal Medicine (APDIM). “You can’t be cheap about acquiring your nonteaching staff.”
The Fate of Students
Perhaps paramount to the fears of how teaching hospitalists will react to current or future restrictions is the effect those limits have on the residents they safeguard. Some physicians think the new rules will produce crops of ill-prepared residents because they have been coddled with limited patient censuses. Other physicians argue that the new thresholds will actually better prepare physicians when HM groups are hiring residents for full-time positions.
Dr. Del Valle acknowledges there is as yet no rigorous data to show the impact of the current restrictions, but he agrees it’s a simple equation of patient-care mathematics. “You can’t [easily] replace 100-110 hours [of care per week],” he says.
Others say patient caps and rules to limit how much work residents do are in line with the purpose of medical training programs. “I’ve bought into the fact that these programs exist to train residents, not to provide clinical care,” Dr. O’Leary says. “I’ve drunk that Kool-Aid. … I think there’s more variation, person to person, than ‘my era vs. the current era.’ Like any new hospitalist that you hire, you need to give an orientation and give enough support to them so when they begin to see patients that they are not overwhelmed.”
Shaun Frost, MD, FACP, FHM, might be best described as halfway between those two extremes. A regional director for the eastern U.S. for Cogent Healthcare, he says duty-hour restrictions have had deleterious impacts but also create learning opportunities.
“The residency work-hour restrictions have inhibited our ability to train people to work as efficiently as trainees who were taught in the past,” says Dr. Frost, an SHM board member. “That doesn’t necessarily mean you can’t teach people to work more efficiently . . . but in the future, my hope is that residency training programs will recognize the deficit that exists in personal work efficiencies between their completion and their responsibilities as a hospitalist.”
To that end, Dr. Frost works with others to develop both structured curriculum and classroom didactics that help new hospitalists make up for gaps in preparation that weren’t addressed in residency. In some cases, that can be practice management and billing issues, but often, according to Dr. Frost, it is addressing personal workflow and bridging the “unnatural discontinuity” in patient care from residency to the real world.
“There is a cost to this investment for the future,” Dr. Frost adds. “If people don’t recognize the potential return on investment as being critical to the development of an educated workforce—an efficient and competent workforce—and thus critical to the retention of high-performing hospitalists, they are selling themselves, unfortunately, significantly short.”
Caught in the Middle
One man’s trash is another man’s treasure, the axiom tells us. Well, in healthcare circles, that could just as easily read: The woes of academic hospitalists are the wealth of community hospitalists.
The new rules “may result in more opportunities for hospitalists to provide needed clinical services,” Dr. Wright says.
The long-term implications, though, remain to be seen. While academic hospitalists say they have seen preliminary increases in care-delivery costs because of the latest rules changes, many say it’s too soon to tell just how high those costs might climb and what ripple effect might follow.
Some physicians, including Dr. Del Valle, note that while the 2009 changes and the expectation of more changes in 2011 are cause for attention, that doesn’t translate to cause for concern. In 2003, months before the 80-hour workweek rules were first put in place by ACGME, many of the same debates were already under way: How will the faculty of IM residency programs cope? How will institutions pay the bills while putting money aside for other physicians picking up the slack?
“This is a pendulum,” Dr. Del Valle says. “I think it will come back to a balanced place.”
Dr. Fried, who is more optimistic that the residency rules can have a positive, long-term effect, agrees. He says residency caps and limits should not be viewed as “things that limit education. We [should] look at them as things that ensure education continues while patient care continues.” TH
Richard Quinn is a freelance writer based in New Jersey.
References
- Institute of Medicine. Resident Duty Hours: Enhancing Sleep, Supervision, and Safety. Ulmer C, Wolman DM, Johns MM, eds. Washington, D.C.: The National Academies Press; 2008.
- Nuckols TK, Bhattacharya J, Wolman DM, Ulmer C, Escarce JJ. Cost implications of reduced work hours and workloads for resident physicians. N Engl J Med. 2009:360(21):2202-2215.
Two-Pronged Approach
An author of a recent study that found improved results for depressed alcohol-dependent patients when they were treated for both diagnoses says hospitalists are positioned to help push increased usage of the dual-medication approach.
“This could change the way people treat these patients,” says first author Helen Pettinati, PhD, professor of psychiatry and director of the Addiction Treatment & Medication Development Division at the University of Pennsylvania School of Medicine’s Treatment Research Center in Philadelphia.
The study in the American Journal of Psychiatry tracked 170 patients, with some treated with 14 weeks of sertraline (200 mg/day, n=40), naltrexone (100 mg/day, n=49), both drugs (n=42), or double placebo (n=39). All groups received weekly counseling as well. Patients treated with both medications produced a higher alcohol abstinence rate (53.7%) and demonstrated a longer delay before relapse to heavy drinking (median delay=98 days) than the naltrexone (abstinence rate: 21.3%; delay=29 days), sertraline (abstinence rate: 27.5%; delay=23 days), and placebo (abstinence rate: 23.1%; delay=26 days) groups. In addition, patients treated with both medications “reported fewer serious adverse events, and tended to not be depressed by the end of treatment,” the authors wrote.
Pettinati says it is uncommon for hospitalists and other physicians to consider treating hospitalized patients with medication for alcohol dependence. She hopes hospitalists see this study as a spotlight on that approach. In past years, Pettinati suggests, medication for alcohol dependence might have been viewed as unnecessary because patient lengths-of-stay (LOS) were longer. Today, HM leaders are pushing for ever-shorter LOS.
“Now when a person comes in,” Pettinati says, “you have to make an immediate decision what kind of medication you want to treat this person with.”
An author of a recent study that found improved results for depressed alcohol-dependent patients when they were treated for both diagnoses says hospitalists are positioned to help push increased usage of the dual-medication approach.
“This could change the way people treat these patients,” says first author Helen Pettinati, PhD, professor of psychiatry and director of the Addiction Treatment & Medication Development Division at the University of Pennsylvania School of Medicine’s Treatment Research Center in Philadelphia.
The study in the American Journal of Psychiatry tracked 170 patients, with some treated with 14 weeks of sertraline (200 mg/day, n=40), naltrexone (100 mg/day, n=49), both drugs (n=42), or double placebo (n=39). All groups received weekly counseling as well. Patients treated with both medications produced a higher alcohol abstinence rate (53.7%) and demonstrated a longer delay before relapse to heavy drinking (median delay=98 days) than the naltrexone (abstinence rate: 21.3%; delay=29 days), sertraline (abstinence rate: 27.5%; delay=23 days), and placebo (abstinence rate: 23.1%; delay=26 days) groups. In addition, patients treated with both medications “reported fewer serious adverse events, and tended to not be depressed by the end of treatment,” the authors wrote.
Pettinati says it is uncommon for hospitalists and other physicians to consider treating hospitalized patients with medication for alcohol dependence. She hopes hospitalists see this study as a spotlight on that approach. In past years, Pettinati suggests, medication for alcohol dependence might have been viewed as unnecessary because patient lengths-of-stay (LOS) were longer. Today, HM leaders are pushing for ever-shorter LOS.
“Now when a person comes in,” Pettinati says, “you have to make an immediate decision what kind of medication you want to treat this person with.”
An author of a recent study that found improved results for depressed alcohol-dependent patients when they were treated for both diagnoses says hospitalists are positioned to help push increased usage of the dual-medication approach.
“This could change the way people treat these patients,” says first author Helen Pettinati, PhD, professor of psychiatry and director of the Addiction Treatment & Medication Development Division at the University of Pennsylvania School of Medicine’s Treatment Research Center in Philadelphia.
The study in the American Journal of Psychiatry tracked 170 patients, with some treated with 14 weeks of sertraline (200 mg/day, n=40), naltrexone (100 mg/day, n=49), both drugs (n=42), or double placebo (n=39). All groups received weekly counseling as well. Patients treated with both medications produced a higher alcohol abstinence rate (53.7%) and demonstrated a longer delay before relapse to heavy drinking (median delay=98 days) than the naltrexone (abstinence rate: 21.3%; delay=29 days), sertraline (abstinence rate: 27.5%; delay=23 days), and placebo (abstinence rate: 23.1%; delay=26 days) groups. In addition, patients treated with both medications “reported fewer serious adverse events, and tended to not be depressed by the end of treatment,” the authors wrote.
Pettinati says it is uncommon for hospitalists and other physicians to consider treating hospitalized patients with medication for alcohol dependence. She hopes hospitalists see this study as a spotlight on that approach. In past years, Pettinati suggests, medication for alcohol dependence might have been viewed as unnecessary because patient lengths-of-stay (LOS) were longer. Today, HM leaders are pushing for ever-shorter LOS.
“Now when a person comes in,” Pettinati says, “you have to make an immediate decision what kind of medication you want to treat this person with.”
New Cost-Control Strategy
A recent study that associates lower 30-day readmission rates for heart failure patients who receive followups within one week might be a jumpstart to new incentives for fewer readmissions, says the report’s author.
“In a way, [hospitalists] are central to all this,” says Adrian F. Hernandez, MD, MHS, an assistant professor at Duke University School of Medicine and a cardiologist at Duke University Medical Center in Durham, N.C. “During the hospital stay, they are quarterbacking that patient’s care. They have a central responsibility to make sure that patient discharge is seamless.”
The study tracked 30,136 patients who were seen at 225 hospitals from January 2003 to December 2006. It reported that in the first 30 days after discharge, 6,428 patients (21.3 percent) were readmitted (JAMA. 2010;303(17):1716-1722).
At the hospital level, the median rate of early followup was 38.3 percent. According to the study, patients whose index admission was in a hospital in the lowest quartile of early followup had a 23.3% 30-day readmission rate. The rates of 30-day readmission were 20.5% among patients in the second quartile, 20.5% among patients in the third quartile, and 20.9% among patients in the fourth quartile.
Dr. Hernandez says the next step is for hospitals and their staffs to commit to more streamlined transitional-care techniques that include immediate followup with patients, be it via teleconferencing with doctors or phone calls with nonphysician providers (NPPs) or clinical pharmacists. He adds that incentivizing doctors to reduce readmissions is a logical next step to improving the discharge process.
“Now that 30-day readmissions are publicly reported and hospitals are being held accountable for that, they need to invest in systems that will enhance that transitional period,” Dr. Hernandez says.
A recent study that associates lower 30-day readmission rates for heart failure patients who receive followups within one week might be a jumpstart to new incentives for fewer readmissions, says the report’s author.
“In a way, [hospitalists] are central to all this,” says Adrian F. Hernandez, MD, MHS, an assistant professor at Duke University School of Medicine and a cardiologist at Duke University Medical Center in Durham, N.C. “During the hospital stay, they are quarterbacking that patient’s care. They have a central responsibility to make sure that patient discharge is seamless.”
The study tracked 30,136 patients who were seen at 225 hospitals from January 2003 to December 2006. It reported that in the first 30 days after discharge, 6,428 patients (21.3 percent) were readmitted (JAMA. 2010;303(17):1716-1722).
At the hospital level, the median rate of early followup was 38.3 percent. According to the study, patients whose index admission was in a hospital in the lowest quartile of early followup had a 23.3% 30-day readmission rate. The rates of 30-day readmission were 20.5% among patients in the second quartile, 20.5% among patients in the third quartile, and 20.9% among patients in the fourth quartile.
Dr. Hernandez says the next step is for hospitals and their staffs to commit to more streamlined transitional-care techniques that include immediate followup with patients, be it via teleconferencing with doctors or phone calls with nonphysician providers (NPPs) or clinical pharmacists. He adds that incentivizing doctors to reduce readmissions is a logical next step to improving the discharge process.
“Now that 30-day readmissions are publicly reported and hospitals are being held accountable for that, they need to invest in systems that will enhance that transitional period,” Dr. Hernandez says.
A recent study that associates lower 30-day readmission rates for heart failure patients who receive followups within one week might be a jumpstart to new incentives for fewer readmissions, says the report’s author.
“In a way, [hospitalists] are central to all this,” says Adrian F. Hernandez, MD, MHS, an assistant professor at Duke University School of Medicine and a cardiologist at Duke University Medical Center in Durham, N.C. “During the hospital stay, they are quarterbacking that patient’s care. They have a central responsibility to make sure that patient discharge is seamless.”
The study tracked 30,136 patients who were seen at 225 hospitals from January 2003 to December 2006. It reported that in the first 30 days after discharge, 6,428 patients (21.3 percent) were readmitted (JAMA. 2010;303(17):1716-1722).
At the hospital level, the median rate of early followup was 38.3 percent. According to the study, patients whose index admission was in a hospital in the lowest quartile of early followup had a 23.3% 30-day readmission rate. The rates of 30-day readmission were 20.5% among patients in the second quartile, 20.5% among patients in the third quartile, and 20.9% among patients in the fourth quartile.
Dr. Hernandez says the next step is for hospitals and their staffs to commit to more streamlined transitional-care techniques that include immediate followup with patients, be it via teleconferencing with doctors or phone calls with nonphysician providers (NPPs) or clinical pharmacists. He adds that incentivizing doctors to reduce readmissions is a logical next step to improving the discharge process.
“Now that 30-day readmissions are publicly reported and hospitals are being held accountable for that, they need to invest in systems that will enhance that transitional period,” Dr. Hernandez says.
Resident Refunds
The IRS has sided with medical residents and their employers who for years have argued that they should have always been eligible for the “student exemption”—but don’t count on any money just yet.
By mid-June, the IRS expects to have contacted hospitals, universities, and individual residents who filed Social Security and Medicare payroll tax refund claims as of April 1, 2005. The date is significant because it is when the IRS ruled that employees who work 40 hours or more at a school, college, or university are eligible for student exemptions.
The IRS’ administrative decision in early March affects taxes paid before 2005.
The IRS has only taken the first step and says instructions on how to further process claims will be forthcoming. For now, the federal agency says, “employers and individuals with pending claims do not need to take any action at this time.”
Still, Joseph Ming-Wah Li, MD, SFHM, assistant professor of medicine at Harvard Medical School and director of the hospital medicine division at Beth Israel Deaconess Medical Center in Boston, urges hospitalists to pay attention to the refund; over three years of residency, it could amount to several thousand dollars per physician.
“The government ruling recently was that residents should be treated more like students instead of employees,” says Dr. Li, SHM's president-elect.
Hospitals, medical schools, and residents themselves have been filing so-called “FICA refund claims” since the 1990s. A series of legal challenges led to opposing interpretations of tax codes, leading the IRS to suspend all claims until a ruling was made.
And while pending claims will now be processed, it is too late for new claims to be filed. However, residents who did not file individual claims in what the IRS calls a “timely fashion” should check with their residency institution to determine if a claim was filed on their behalf.
Read "Dr. Hospitalist's" take on this topic in this month's issue of The Hospitalist.
For more details, visit the IRS website.
The IRS has sided with medical residents and their employers who for years have argued that they should have always been eligible for the “student exemption”—but don’t count on any money just yet.
By mid-June, the IRS expects to have contacted hospitals, universities, and individual residents who filed Social Security and Medicare payroll tax refund claims as of April 1, 2005. The date is significant because it is when the IRS ruled that employees who work 40 hours or more at a school, college, or university are eligible for student exemptions.
The IRS’ administrative decision in early March affects taxes paid before 2005.
The IRS has only taken the first step and says instructions on how to further process claims will be forthcoming. For now, the federal agency says, “employers and individuals with pending claims do not need to take any action at this time.”
Still, Joseph Ming-Wah Li, MD, SFHM, assistant professor of medicine at Harvard Medical School and director of the hospital medicine division at Beth Israel Deaconess Medical Center in Boston, urges hospitalists to pay attention to the refund; over three years of residency, it could amount to several thousand dollars per physician.
“The government ruling recently was that residents should be treated more like students instead of employees,” says Dr. Li, SHM's president-elect.
Hospitals, medical schools, and residents themselves have been filing so-called “FICA refund claims” since the 1990s. A series of legal challenges led to opposing interpretations of tax codes, leading the IRS to suspend all claims until a ruling was made.
And while pending claims will now be processed, it is too late for new claims to be filed. However, residents who did not file individual claims in what the IRS calls a “timely fashion” should check with their residency institution to determine if a claim was filed on their behalf.
Read "Dr. Hospitalist's" take on this topic in this month's issue of The Hospitalist.
For more details, visit the IRS website.
The IRS has sided with medical residents and their employers who for years have argued that they should have always been eligible for the “student exemption”—but don’t count on any money just yet.
By mid-June, the IRS expects to have contacted hospitals, universities, and individual residents who filed Social Security and Medicare payroll tax refund claims as of April 1, 2005. The date is significant because it is when the IRS ruled that employees who work 40 hours or more at a school, college, or university are eligible for student exemptions.
The IRS’ administrative decision in early March affects taxes paid before 2005.
The IRS has only taken the first step and says instructions on how to further process claims will be forthcoming. For now, the federal agency says, “employers and individuals with pending claims do not need to take any action at this time.”
Still, Joseph Ming-Wah Li, MD, SFHM, assistant professor of medicine at Harvard Medical School and director of the hospital medicine division at Beth Israel Deaconess Medical Center in Boston, urges hospitalists to pay attention to the refund; over three years of residency, it could amount to several thousand dollars per physician.
“The government ruling recently was that residents should be treated more like students instead of employees,” says Dr. Li, SHM's president-elect.
Hospitals, medical schools, and residents themselves have been filing so-called “FICA refund claims” since the 1990s. A series of legal challenges led to opposing interpretations of tax codes, leading the IRS to suspend all claims until a ruling was made.
And while pending claims will now be processed, it is too late for new claims to be filed. However, residents who did not file individual claims in what the IRS calls a “timely fashion” should check with their residency institution to determine if a claim was filed on their behalf.
Read "Dr. Hospitalist's" take on this topic in this month's issue of The Hospitalist.
For more details, visit the IRS website.
Obama Confirms Partners’ Visitation, Decision Rights
HM leaders should pay close attention to the landmark decision by President Obama to extend visitation and critical-care decision rights to the partners of gay and lesbian patients, according to one hospitalist.
“The hospitalists would be the gatekeepers, in some ways, to make those calls,” says Heather Whelan, MD, an assistant professor and medical director at Mount Zion Medical Service, Division of Hospital Medicine, at the University of California at San Francisco. “I think hospitalists, group managers, and hospitals themselves are going to build this into policies and practices.”
Via a memo written in mid-April, the president mandated that all hospitals receiving federal funding must extend visitation rights to gay partners and “respect the patient’s choices about who may make critical heath-care decisions for them,” according to the Washington Post. Gay activists cheered the decisions; many conservative groups decried it.
An SHM spokesperson says the society “always expects that its hospitalists and their policies know and comply with federal, state and local regulations.”
Dr. Whelan notes that many hospitals in more progressive areas of the country—including San Francisco and New York City—already extend the rights that Obama has now codified. However, she says she has heard from physicians working elsewhere that the issue can crop up on who can be involved in critical-care meetings and decisions. The issue can be particularly sensitive in cases in which the patient is noncommunicative due to a stroke or other acute condition.
“This will standardize care,” Dr. Whelan says, adding, “It almost doesn’t matter how frequently it comes up. It’s a high-stakes event. One or two instances of it coming up are too many.”
HM leaders should pay close attention to the landmark decision by President Obama to extend visitation and critical-care decision rights to the partners of gay and lesbian patients, according to one hospitalist.
“The hospitalists would be the gatekeepers, in some ways, to make those calls,” says Heather Whelan, MD, an assistant professor and medical director at Mount Zion Medical Service, Division of Hospital Medicine, at the University of California at San Francisco. “I think hospitalists, group managers, and hospitals themselves are going to build this into policies and practices.”
Via a memo written in mid-April, the president mandated that all hospitals receiving federal funding must extend visitation rights to gay partners and “respect the patient’s choices about who may make critical heath-care decisions for them,” according to the Washington Post. Gay activists cheered the decisions; many conservative groups decried it.
An SHM spokesperson says the society “always expects that its hospitalists and their policies know and comply with federal, state and local regulations.”
Dr. Whelan notes that many hospitals in more progressive areas of the country—including San Francisco and New York City—already extend the rights that Obama has now codified. However, she says she has heard from physicians working elsewhere that the issue can crop up on who can be involved in critical-care meetings and decisions. The issue can be particularly sensitive in cases in which the patient is noncommunicative due to a stroke or other acute condition.
“This will standardize care,” Dr. Whelan says, adding, “It almost doesn’t matter how frequently it comes up. It’s a high-stakes event. One or two instances of it coming up are too many.”
HM leaders should pay close attention to the landmark decision by President Obama to extend visitation and critical-care decision rights to the partners of gay and lesbian patients, according to one hospitalist.
“The hospitalists would be the gatekeepers, in some ways, to make those calls,” says Heather Whelan, MD, an assistant professor and medical director at Mount Zion Medical Service, Division of Hospital Medicine, at the University of California at San Francisco. “I think hospitalists, group managers, and hospitals themselves are going to build this into policies and practices.”
Via a memo written in mid-April, the president mandated that all hospitals receiving federal funding must extend visitation rights to gay partners and “respect the patient’s choices about who may make critical heath-care decisions for them,” according to the Washington Post. Gay activists cheered the decisions; many conservative groups decried it.
An SHM spokesperson says the society “always expects that its hospitalists and their policies know and comply with federal, state and local regulations.”
Dr. Whelan notes that many hospitals in more progressive areas of the country—including San Francisco and New York City—already extend the rights that Obama has now codified. However, she says she has heard from physicians working elsewhere that the issue can crop up on who can be involved in critical-care meetings and decisions. The issue can be particularly sensitive in cases in which the patient is noncommunicative due to a stroke or other acute condition.
“This will standardize care,” Dr. Whelan says, adding, “It almost doesn’t matter how frequently it comes up. It’s a high-stakes event. One or two instances of it coming up are too many.”
ONLINE EXCLUSIVE: Audio interview with SHM President Jeff Wiese, MD, FACP, SFHM
Wachter’s World
NATIONAL HARBOR, Md.—Democrats and Republicans have trumpeted that unprecedented changes in the healthcare system are on the way, but the dean of HM cautions that significant change is still years away.
“The reform bill, to my mind, mostly kicked the hard decision for cost, quality, and safety down the road,” said Bob Wachter, MD, MHM, chief of the hospitalist division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco. “All of these issues have been raised, though.”
Dr. Wachter, former SHM president, author of the blog Wachter’s World (www.wachters world.com), and recently named the 10th-most-powerful physician executive in the nation by Modern Healthcare, used his annual HM10 address to paint a cautiously optimistic picture of HM playing a leading role in quality, safety, and innovation in the delivery of healthcare.
“It is a completely open question, whether we will be capable of snapping our fingers and creating a set of incentives or policy drivers that will allow the creation of the next Geisinger [Health System] without waiting 50 years,” Dr. Wachter said. “These cultures take a long time to develop. It’s not just about the [organizational] chart and the way money flows. You have to develop the culture of shared governance.”
In what has become a rite, Dr. Wachter gives the closing address at SHM’s annual meeting. This year’s title: “Use Your Words: Understanding the New Language of Healthcare Reform.” He focused most of his speech on finding the balance between high-quality and low-cost patient care, particularly when viewed through the prism of the “cost curve,” the economic principle that measures benefits against their cost.
Medical care on the “flat part of the curve” equates to tests, procedures, or other engagements that might have prophylactic value but little clinical benefit. From a purely clinical point of view, that is acceptable, but layering in a cost-benefit analysis adds a more objective way of deciding whether the care delivered is “worth the cost,” Dr. Wachter said.
—Bob Wachter, MD, MHM, chief of the hospitalist division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco
“The question is: Where do we want to live on this curve?” he added. “As you spend more money, you may be getting more benefit, but the incremental amount . . . pushes out past the flat part of curve.”
Dr. Wachter boiled his lesson down to two philosophies. In one, the practice of HM means a test, a procedure, or a consult is ordered because the benefits outweigh the risks. In the other, that same episodic treatment is ordered only if every less-expensive option has already been attempted. “These are absolutely fundamental tensions,” he admitted.
But not all that is reform must be contentious, he said. Take the renewed push toward “accountable-care organizations,” in which providers partner and share responsibility for both the quality and cost of healthcare for a specific population of beneficiaries. The healthcare reform bill contains incentives for such a structure, which Dr. Wachter views as the government’s latest attempt to improve care by controlling how much reimbursement physicians and their employers receive.
While other specialists might not be experienced with data-point discussions on cost savings with hospital administrators, HM leaders are all too familiar with the concept, as most have those discussions during annual hospital subsidy negotiations. Correspondingly, those who listened to Dr. Wachter’s advice agreed that there is ample opportunity to lead the charge for quality and safety improvement—and the likely savings to be associated with those changes.
“Let’s be patient for what’s coming around the corner,” said Daniel Dressler, MD, SFHM, director of hospital medicine at Emory University Hospital in Atlanta and an SHM board member. “But let’s not miss the boat.” HM2010
Richard Quinn is a freelance writer based in New Jersey.
NATIONAL HARBOR, Md.—Democrats and Republicans have trumpeted that unprecedented changes in the healthcare system are on the way, but the dean of HM cautions that significant change is still years away.
“The reform bill, to my mind, mostly kicked the hard decision for cost, quality, and safety down the road,” said Bob Wachter, MD, MHM, chief of the hospitalist division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco. “All of these issues have been raised, though.”
Dr. Wachter, former SHM president, author of the blog Wachter’s World (www.wachters world.com), and recently named the 10th-most-powerful physician executive in the nation by Modern Healthcare, used his annual HM10 address to paint a cautiously optimistic picture of HM playing a leading role in quality, safety, and innovation in the delivery of healthcare.
“It is a completely open question, whether we will be capable of snapping our fingers and creating a set of incentives or policy drivers that will allow the creation of the next Geisinger [Health System] without waiting 50 years,” Dr. Wachter said. “These cultures take a long time to develop. It’s not just about the [organizational] chart and the way money flows. You have to develop the culture of shared governance.”
In what has become a rite, Dr. Wachter gives the closing address at SHM’s annual meeting. This year’s title: “Use Your Words: Understanding the New Language of Healthcare Reform.” He focused most of his speech on finding the balance between high-quality and low-cost patient care, particularly when viewed through the prism of the “cost curve,” the economic principle that measures benefits against their cost.
Medical care on the “flat part of the curve” equates to tests, procedures, or other engagements that might have prophylactic value but little clinical benefit. From a purely clinical point of view, that is acceptable, but layering in a cost-benefit analysis adds a more objective way of deciding whether the care delivered is “worth the cost,” Dr. Wachter said.
—Bob Wachter, MD, MHM, chief of the hospitalist division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco
“The question is: Where do we want to live on this curve?” he added. “As you spend more money, you may be getting more benefit, but the incremental amount . . . pushes out past the flat part of curve.”
Dr. Wachter boiled his lesson down to two philosophies. In one, the practice of HM means a test, a procedure, or a consult is ordered because the benefits outweigh the risks. In the other, that same episodic treatment is ordered only if every less-expensive option has already been attempted. “These are absolutely fundamental tensions,” he admitted.
But not all that is reform must be contentious, he said. Take the renewed push toward “accountable-care organizations,” in which providers partner and share responsibility for both the quality and cost of healthcare for a specific population of beneficiaries. The healthcare reform bill contains incentives for such a structure, which Dr. Wachter views as the government’s latest attempt to improve care by controlling how much reimbursement physicians and their employers receive.
While other specialists might not be experienced with data-point discussions on cost savings with hospital administrators, HM leaders are all too familiar with the concept, as most have those discussions during annual hospital subsidy negotiations. Correspondingly, those who listened to Dr. Wachter’s advice agreed that there is ample opportunity to lead the charge for quality and safety improvement—and the likely savings to be associated with those changes.
“Let’s be patient for what’s coming around the corner,” said Daniel Dressler, MD, SFHM, director of hospital medicine at Emory University Hospital in Atlanta and an SHM board member. “But let’s not miss the boat.” HM2010
Richard Quinn is a freelance writer based in New Jersey.
NATIONAL HARBOR, Md.—Democrats and Republicans have trumpeted that unprecedented changes in the healthcare system are on the way, but the dean of HM cautions that significant change is still years away.
“The reform bill, to my mind, mostly kicked the hard decision for cost, quality, and safety down the road,” said Bob Wachter, MD, MHM, chief of the hospitalist division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco. “All of these issues have been raised, though.”
Dr. Wachter, former SHM president, author of the blog Wachter’s World (www.wachters world.com), and recently named the 10th-most-powerful physician executive in the nation by Modern Healthcare, used his annual HM10 address to paint a cautiously optimistic picture of HM playing a leading role in quality, safety, and innovation in the delivery of healthcare.
“It is a completely open question, whether we will be capable of snapping our fingers and creating a set of incentives or policy drivers that will allow the creation of the next Geisinger [Health System] without waiting 50 years,” Dr. Wachter said. “These cultures take a long time to develop. It’s not just about the [organizational] chart and the way money flows. You have to develop the culture of shared governance.”
In what has become a rite, Dr. Wachter gives the closing address at SHM’s annual meeting. This year’s title: “Use Your Words: Understanding the New Language of Healthcare Reform.” He focused most of his speech on finding the balance between high-quality and low-cost patient care, particularly when viewed through the prism of the “cost curve,” the economic principle that measures benefits against their cost.
Medical care on the “flat part of the curve” equates to tests, procedures, or other engagements that might have prophylactic value but little clinical benefit. From a purely clinical point of view, that is acceptable, but layering in a cost-benefit analysis adds a more objective way of deciding whether the care delivered is “worth the cost,” Dr. Wachter said.
—Bob Wachter, MD, MHM, chief of the hospitalist division, professor, and associate chair of the Department of Medicine at the University of California at San Francisco
“The question is: Where do we want to live on this curve?” he added. “As you spend more money, you may be getting more benefit, but the incremental amount . . . pushes out past the flat part of curve.”
Dr. Wachter boiled his lesson down to two philosophies. In one, the practice of HM means a test, a procedure, or a consult is ordered because the benefits outweigh the risks. In the other, that same episodic treatment is ordered only if every less-expensive option has already been attempted. “These are absolutely fundamental tensions,” he admitted.
But not all that is reform must be contentious, he said. Take the renewed push toward “accountable-care organizations,” in which providers partner and share responsibility for both the quality and cost of healthcare for a specific population of beneficiaries. The healthcare reform bill contains incentives for such a structure, which Dr. Wachter views as the government’s latest attempt to improve care by controlling how much reimbursement physicians and their employers receive.
While other specialists might not be experienced with data-point discussions on cost savings with hospital administrators, HM leaders are all too familiar with the concept, as most have those discussions during annual hospital subsidy negotiations. Correspondingly, those who listened to Dr. Wachter’s advice agreed that there is ample opportunity to lead the charge for quality and safety improvement—and the likely savings to be associated with those changes.
“Let’s be patient for what’s coming around the corner,” said Daniel Dressler, MD, SFHM, director of hospital medicine at Emory University Hospital in Atlanta and an SHM board member. “But let’s not miss the boat.” HM2010
Richard Quinn is a freelance writer based in New Jersey.
Special Interests
HOSPITALISTS FROM ALL PARTS OF THE COUNTRY—and a few other countries—discussed a wide swath of topics during a community-based HM special-interest forum at HM10. Issues that were discussed included unit-based rounding, changes to Medicare consult codes, strategies for avoiding “dumps,” and working with specialists.
Two established community hospitalists—SHM co-founders John Nelson, MD, MHM, and Winthrop Whitcomb, MD, MHM—moderated the one-hour session.
Much of the debate centered on defining a hospitalist’s role and relationships with others in the hospital. One hospitalist said he’d noticed significant changes in the 15 years since he began HM practice; however, some issues remain unresolved: Primary-care physicians (PCPs) still know the patients better, and medical specialists still want hospitalists to be their “interns.”
“We have two things to sell: your expertise and your availability. It’s up to your group to determine which one you want to sell,” said Tony Lin, MD, FHM, a hospitalist and chief of the Department of Internal Medicine at Kelsey-Seybold Clinic in Houston. “I don’t think you have to pick one. So I think you have to ask yourself: What does our group want to sell to the specialist? Sometimes you might have to turn them down to make that point.”
Dr. Lin also described a phenomena emerging in the Houston area: independent, one-physician HM groups taking root in community hospitals. “A lot of the surgeons are using them because they are willing to work as the interns and residents, the first people the nurses call at 2 a.m.,” he said. “There is a market for them.”
Dr. Nelson advised community hospitalists to avoid doing “the things that make you appear different from everyone else. Build social connections with specialists; call them by their first name; eat lunch in the cafeteria; and dress professionally.”
•
One community hospitalist spoke of an ethical situation she regularly encounters at her hospital, which contracts with multiple HM groups. Anna Rodriguez, MD, of Chesapeake Hospitalists in Chesapeake, Va., explained that her group’s issue is acutely ill patients who are assigned to one of the other HM group services—which, unlike Dr. Rodriguez’s group, are not responsible for codes or 24/7 patient coverage. So what happens when the “other” group’s patient has a sudden deterioration and the hospital staff calls us to run the rapid response? Dr. Rodriguez asked the group.
Dr. Whitcomb suggested Dr. Rodriguez’s group, which is not contracted to run the code, work to iron that situation out. “Then, that is your job and contractually recognized,” he said.
“We get into the exact same situation in our hospital. We created a hospital medicine section and … established expectations for who responds to codes,” said Dennis Kold, MD, medical director of the hospitalist service Tri-Health in Cincinnati. “If the patient is declining, we will respond to code, but we have it set up where the expectation is that the [attending] will be in to take care of the patient in one hour, or if the patient is admitted overnight to the ICU at 10 p.m., that the [admitting] will be in the ICU to take care of the patient within four hours.” Dr. Kold added that when the attending doesn’t show up in time that penalties are enforced (e.g., taken off the ER call schedule, restriction of hospital privileges).
“If you are not dealing with rapid response, then you are just hurting yourself,” added Edward Rosenfeld, MD, a hospitalist with Lehigh Valley Medical Associates in Allentown, Pa. “You need to do it; that’s your code prevention.”
•
Community hospitalists also discussed bundled payments and the recent changes in Medicare consult codes. “As a hospitalist service, I want to be involved in divvying up the money,” said Dan Allen, MD, a group director in Des Moines, Iowa. “I don’t know where it’s going, but I want to have a seat at the table.”
When asked by Dr. Nelson if they had noticed a significant change in reimbursement due to Medicare’s elimination of consultation codes, few in the room raised their hands. In fact, Dr. Nelson explained, “you can bill initial hospital care instead of initial hospital consult.”
“If done right, you might get paid better,” Dr. Rosenfeld added.
—Jason Carris
Health Information Technology on the Hospitalist Radar
Health information technology (HIT) isn’t for geeks anymore. A year after a mostly tech-savvy room discussed the basics of introducing more IT aspects to HM, nearly three dozen hospitalists clamored for SHM to take advocacy positions on everything from best practices to best vendors.
“SHM could help us all speak the language we need to speak,” said Tosha Wetterneck, MD, MS, a hospitalist with the University of Wisconsin Clinic in Madison. “Visibility, transparency—give us the words.”
Participants in last year’s group focused on the technical side of IT. This year’s attendees talked about the need for SHM to create portals for shared information, message boards to spur interinstitution conversations, and, perhaps, a weekend boot-camp-style course to introduce novices to basic IT information.
“SHM needs to take a stand now,” said Damascene Kurukulasuriya, MD, FACP, CMD, CCD, a hospitalist in perioperative medicine at the University of Missouri Health System in Columbia. “We need to be part of the solution.”
To that end, Kendall Rogers, MD, assistant professor at the University of New Mexico School of Medicine and chair of SHM’s IT Task Force, says the society is making progress. More hospitalists have been encouraged to sign up for the BioMedical Informatics course at the Marine Biology Laboratory in Woods Hole, Mass. The weeklong course is an introduction to the use of computer technologies and information science related to biomedicine and health science, according to the program’s Web site (www.courses.mbl.edu/mi/). The cost of travel, housing, and meals are fully paid for by the National Library of Medicine, making the fellowship even more appealing for cash-strapped hospitals and HM groups. “It’s a hidden program,” Dr. Rogers said.
Dr. Rogers and SHM CEO Larry Wellikson have toured the country meeting with top officials from the largest IT vendors, including Cerner and GE Healthcare. An IT committee has formed, with subcommittees dedicated to policy, quality, and leadership/education. But Bob Lineberger, MD, medical information officer at Durham Regional Hospital in North Carolina, says a nuanced message will take time.
“Our focus is just coming into focus,” Dr. Lineberger conceded. “We do need to come up with a position statement.”
—Richard Quinn
Education in HM: How to Grow Rock Stars and Champions
What skills does a hospitalist need to know to practice well that they didn’t learn in residency? That was the question new SHM President Jeff Weise, MD, SFHM, posed to about 20 hospitalists attending the special-interest forum on educational initiatives at HM10. Led by Dr. Wiese and SHM Education Committee co-chair Vikas Parekh, MD, FHM, the discussion focused on what SHM can do—or perhaps do better—in this capacity.
Dr. Parekh said hospitalists should be experts in quality-improvement (QI) and patient safety, and HM must incorporate that expertise into daily practice. However, he said, SHM’s largest educational focus is the new Focused Practice in Hospital Medicine pathway to American Board of Internal Medicine’s (ABIM) recertification.
In describing the future of this dynamic field, Dr. Weise raised concerns about managing the pipeline of approximately 2,500 new residents turning out each year and the potential for “losing intimacy” among SHM members—which he described as “the curse of being a champion.”
“IT is the only solution,” he added, “and identifying new and better ways of communicating.”
Competency-Based Train-ing (CBT) is critical to the development of new hospitalists, Dr. Wiese explained, as a supplement for what isn’t taught in residency. He posed a question: Should residencies last four or five years to incorporate additional training and career planning? “It’s an MBA paradigm of learning what we do,” he said. “What compels residents to join fellowship programs and earn $50K per year when they can start practicing and earning $150K?”
Educating the membership requires innovation and more than just bench-to-bedside research, Dr. Wiese added. Translational research and best-evidence practices will improve the field. “Five or 10 programs are rock stars,” he said, “but there are 377 that are terrible.”
Future SHM goals include a vision of having hospitalists hold 20% of all Internal Medicine Residency Program Director positions; developing best practices, not unfunded mandates; establishing protected academic time; and encouraging mentorship that positions hospitalists as heroes for the next generation.
An education committee sub-group has been tasked to focus on the recruitment of hospitalists and expose them to the best the society and field have to offer. HM10
—Phaedra Cress
HOSPITALISTS FROM ALL PARTS OF THE COUNTRY—and a few other countries—discussed a wide swath of topics during a community-based HM special-interest forum at HM10. Issues that were discussed included unit-based rounding, changes to Medicare consult codes, strategies for avoiding “dumps,” and working with specialists.
Two established community hospitalists—SHM co-founders John Nelson, MD, MHM, and Winthrop Whitcomb, MD, MHM—moderated the one-hour session.
Much of the debate centered on defining a hospitalist’s role and relationships with others in the hospital. One hospitalist said he’d noticed significant changes in the 15 years since he began HM practice; however, some issues remain unresolved: Primary-care physicians (PCPs) still know the patients better, and medical specialists still want hospitalists to be their “interns.”
“We have two things to sell: your expertise and your availability. It’s up to your group to determine which one you want to sell,” said Tony Lin, MD, FHM, a hospitalist and chief of the Department of Internal Medicine at Kelsey-Seybold Clinic in Houston. “I don’t think you have to pick one. So I think you have to ask yourself: What does our group want to sell to the specialist? Sometimes you might have to turn them down to make that point.”
Dr. Lin also described a phenomena emerging in the Houston area: independent, one-physician HM groups taking root in community hospitals. “A lot of the surgeons are using them because they are willing to work as the interns and residents, the first people the nurses call at 2 a.m.,” he said. “There is a market for them.”
Dr. Nelson advised community hospitalists to avoid doing “the things that make you appear different from everyone else. Build social connections with specialists; call them by their first name; eat lunch in the cafeteria; and dress professionally.”
•
One community hospitalist spoke of an ethical situation she regularly encounters at her hospital, which contracts with multiple HM groups. Anna Rodriguez, MD, of Chesapeake Hospitalists in Chesapeake, Va., explained that her group’s issue is acutely ill patients who are assigned to one of the other HM group services—which, unlike Dr. Rodriguez’s group, are not responsible for codes or 24/7 patient coverage. So what happens when the “other” group’s patient has a sudden deterioration and the hospital staff calls us to run the rapid response? Dr. Rodriguez asked the group.
Dr. Whitcomb suggested Dr. Rodriguez’s group, which is not contracted to run the code, work to iron that situation out. “Then, that is your job and contractually recognized,” he said.
“We get into the exact same situation in our hospital. We created a hospital medicine section and … established expectations for who responds to codes,” said Dennis Kold, MD, medical director of the hospitalist service Tri-Health in Cincinnati. “If the patient is declining, we will respond to code, but we have it set up where the expectation is that the [attending] will be in to take care of the patient in one hour, or if the patient is admitted overnight to the ICU at 10 p.m., that the [admitting] will be in the ICU to take care of the patient within four hours.” Dr. Kold added that when the attending doesn’t show up in time that penalties are enforced (e.g., taken off the ER call schedule, restriction of hospital privileges).
“If you are not dealing with rapid response, then you are just hurting yourself,” added Edward Rosenfeld, MD, a hospitalist with Lehigh Valley Medical Associates in Allentown, Pa. “You need to do it; that’s your code prevention.”
•
Community hospitalists also discussed bundled payments and the recent changes in Medicare consult codes. “As a hospitalist service, I want to be involved in divvying up the money,” said Dan Allen, MD, a group director in Des Moines, Iowa. “I don’t know where it’s going, but I want to have a seat at the table.”
When asked by Dr. Nelson if they had noticed a significant change in reimbursement due to Medicare’s elimination of consultation codes, few in the room raised their hands. In fact, Dr. Nelson explained, “you can bill initial hospital care instead of initial hospital consult.”
“If done right, you might get paid better,” Dr. Rosenfeld added.
—Jason Carris
Health Information Technology on the Hospitalist Radar
Health information technology (HIT) isn’t for geeks anymore. A year after a mostly tech-savvy room discussed the basics of introducing more IT aspects to HM, nearly three dozen hospitalists clamored for SHM to take advocacy positions on everything from best practices to best vendors.
“SHM could help us all speak the language we need to speak,” said Tosha Wetterneck, MD, MS, a hospitalist with the University of Wisconsin Clinic in Madison. “Visibility, transparency—give us the words.”
Participants in last year’s group focused on the technical side of IT. This year’s attendees talked about the need for SHM to create portals for shared information, message boards to spur interinstitution conversations, and, perhaps, a weekend boot-camp-style course to introduce novices to basic IT information.
“SHM needs to take a stand now,” said Damascene Kurukulasuriya, MD, FACP, CMD, CCD, a hospitalist in perioperative medicine at the University of Missouri Health System in Columbia. “We need to be part of the solution.”
To that end, Kendall Rogers, MD, assistant professor at the University of New Mexico School of Medicine and chair of SHM’s IT Task Force, says the society is making progress. More hospitalists have been encouraged to sign up for the BioMedical Informatics course at the Marine Biology Laboratory in Woods Hole, Mass. The weeklong course is an introduction to the use of computer technologies and information science related to biomedicine and health science, according to the program’s Web site (www.courses.mbl.edu/mi/). The cost of travel, housing, and meals are fully paid for by the National Library of Medicine, making the fellowship even more appealing for cash-strapped hospitals and HM groups. “It’s a hidden program,” Dr. Rogers said.
Dr. Rogers and SHM CEO Larry Wellikson have toured the country meeting with top officials from the largest IT vendors, including Cerner and GE Healthcare. An IT committee has formed, with subcommittees dedicated to policy, quality, and leadership/education. But Bob Lineberger, MD, medical information officer at Durham Regional Hospital in North Carolina, says a nuanced message will take time.
“Our focus is just coming into focus,” Dr. Lineberger conceded. “We do need to come up with a position statement.”
—Richard Quinn
Education in HM: How to Grow Rock Stars and Champions
What skills does a hospitalist need to know to practice well that they didn’t learn in residency? That was the question new SHM President Jeff Weise, MD, SFHM, posed to about 20 hospitalists attending the special-interest forum on educational initiatives at HM10. Led by Dr. Wiese and SHM Education Committee co-chair Vikas Parekh, MD, FHM, the discussion focused on what SHM can do—or perhaps do better—in this capacity.
Dr. Parekh said hospitalists should be experts in quality-improvement (QI) and patient safety, and HM must incorporate that expertise into daily practice. However, he said, SHM’s largest educational focus is the new Focused Practice in Hospital Medicine pathway to American Board of Internal Medicine’s (ABIM) recertification.
In describing the future of this dynamic field, Dr. Weise raised concerns about managing the pipeline of approximately 2,500 new residents turning out each year and the potential for “losing intimacy” among SHM members—which he described as “the curse of being a champion.”
“IT is the only solution,” he added, “and identifying new and better ways of communicating.”
Competency-Based Train-ing (CBT) is critical to the development of new hospitalists, Dr. Wiese explained, as a supplement for what isn’t taught in residency. He posed a question: Should residencies last four or five years to incorporate additional training and career planning? “It’s an MBA paradigm of learning what we do,” he said. “What compels residents to join fellowship programs and earn $50K per year when they can start practicing and earning $150K?”
Educating the membership requires innovation and more than just bench-to-bedside research, Dr. Wiese added. Translational research and best-evidence practices will improve the field. “Five or 10 programs are rock stars,” he said, “but there are 377 that are terrible.”
Future SHM goals include a vision of having hospitalists hold 20% of all Internal Medicine Residency Program Director positions; developing best practices, not unfunded mandates; establishing protected academic time; and encouraging mentorship that positions hospitalists as heroes for the next generation.
An education committee sub-group has been tasked to focus on the recruitment of hospitalists and expose them to the best the society and field have to offer. HM10
—Phaedra Cress
HOSPITALISTS FROM ALL PARTS OF THE COUNTRY—and a few other countries—discussed a wide swath of topics during a community-based HM special-interest forum at HM10. Issues that were discussed included unit-based rounding, changes to Medicare consult codes, strategies for avoiding “dumps,” and working with specialists.
Two established community hospitalists—SHM co-founders John Nelson, MD, MHM, and Winthrop Whitcomb, MD, MHM—moderated the one-hour session.
Much of the debate centered on defining a hospitalist’s role and relationships with others in the hospital. One hospitalist said he’d noticed significant changes in the 15 years since he began HM practice; however, some issues remain unresolved: Primary-care physicians (PCPs) still know the patients better, and medical specialists still want hospitalists to be their “interns.”
“We have two things to sell: your expertise and your availability. It’s up to your group to determine which one you want to sell,” said Tony Lin, MD, FHM, a hospitalist and chief of the Department of Internal Medicine at Kelsey-Seybold Clinic in Houston. “I don’t think you have to pick one. So I think you have to ask yourself: What does our group want to sell to the specialist? Sometimes you might have to turn them down to make that point.”
Dr. Lin also described a phenomena emerging in the Houston area: independent, one-physician HM groups taking root in community hospitals. “A lot of the surgeons are using them because they are willing to work as the interns and residents, the first people the nurses call at 2 a.m.,” he said. “There is a market for them.”
Dr. Nelson advised community hospitalists to avoid doing “the things that make you appear different from everyone else. Build social connections with specialists; call them by their first name; eat lunch in the cafeteria; and dress professionally.”
•
One community hospitalist spoke of an ethical situation she regularly encounters at her hospital, which contracts with multiple HM groups. Anna Rodriguez, MD, of Chesapeake Hospitalists in Chesapeake, Va., explained that her group’s issue is acutely ill patients who are assigned to one of the other HM group services—which, unlike Dr. Rodriguez’s group, are not responsible for codes or 24/7 patient coverage. So what happens when the “other” group’s patient has a sudden deterioration and the hospital staff calls us to run the rapid response? Dr. Rodriguez asked the group.
Dr. Whitcomb suggested Dr. Rodriguez’s group, which is not contracted to run the code, work to iron that situation out. “Then, that is your job and contractually recognized,” he said.
“We get into the exact same situation in our hospital. We created a hospital medicine section and … established expectations for who responds to codes,” said Dennis Kold, MD, medical director of the hospitalist service Tri-Health in Cincinnati. “If the patient is declining, we will respond to code, but we have it set up where the expectation is that the [attending] will be in to take care of the patient in one hour, or if the patient is admitted overnight to the ICU at 10 p.m., that the [admitting] will be in the ICU to take care of the patient within four hours.” Dr. Kold added that when the attending doesn’t show up in time that penalties are enforced (e.g., taken off the ER call schedule, restriction of hospital privileges).
“If you are not dealing with rapid response, then you are just hurting yourself,” added Edward Rosenfeld, MD, a hospitalist with Lehigh Valley Medical Associates in Allentown, Pa. “You need to do it; that’s your code prevention.”
•
Community hospitalists also discussed bundled payments and the recent changes in Medicare consult codes. “As a hospitalist service, I want to be involved in divvying up the money,” said Dan Allen, MD, a group director in Des Moines, Iowa. “I don’t know where it’s going, but I want to have a seat at the table.”
When asked by Dr. Nelson if they had noticed a significant change in reimbursement due to Medicare’s elimination of consultation codes, few in the room raised their hands. In fact, Dr. Nelson explained, “you can bill initial hospital care instead of initial hospital consult.”
“If done right, you might get paid better,” Dr. Rosenfeld added.
—Jason Carris
Health Information Technology on the Hospitalist Radar
Health information technology (HIT) isn’t for geeks anymore. A year after a mostly tech-savvy room discussed the basics of introducing more IT aspects to HM, nearly three dozen hospitalists clamored for SHM to take advocacy positions on everything from best practices to best vendors.
“SHM could help us all speak the language we need to speak,” said Tosha Wetterneck, MD, MS, a hospitalist with the University of Wisconsin Clinic in Madison. “Visibility, transparency—give us the words.”
Participants in last year’s group focused on the technical side of IT. This year’s attendees talked about the need for SHM to create portals for shared information, message boards to spur interinstitution conversations, and, perhaps, a weekend boot-camp-style course to introduce novices to basic IT information.
“SHM needs to take a stand now,” said Damascene Kurukulasuriya, MD, FACP, CMD, CCD, a hospitalist in perioperative medicine at the University of Missouri Health System in Columbia. “We need to be part of the solution.”
To that end, Kendall Rogers, MD, assistant professor at the University of New Mexico School of Medicine and chair of SHM’s IT Task Force, says the society is making progress. More hospitalists have been encouraged to sign up for the BioMedical Informatics course at the Marine Biology Laboratory in Woods Hole, Mass. The weeklong course is an introduction to the use of computer technologies and information science related to biomedicine and health science, according to the program’s Web site (www.courses.mbl.edu/mi/). The cost of travel, housing, and meals are fully paid for by the National Library of Medicine, making the fellowship even more appealing for cash-strapped hospitals and HM groups. “It’s a hidden program,” Dr. Rogers said.
Dr. Rogers and SHM CEO Larry Wellikson have toured the country meeting with top officials from the largest IT vendors, including Cerner and GE Healthcare. An IT committee has formed, with subcommittees dedicated to policy, quality, and leadership/education. But Bob Lineberger, MD, medical information officer at Durham Regional Hospital in North Carolina, says a nuanced message will take time.
“Our focus is just coming into focus,” Dr. Lineberger conceded. “We do need to come up with a position statement.”
—Richard Quinn
Education in HM: How to Grow Rock Stars and Champions
What skills does a hospitalist need to know to practice well that they didn’t learn in residency? That was the question new SHM President Jeff Weise, MD, SFHM, posed to about 20 hospitalists attending the special-interest forum on educational initiatives at HM10. Led by Dr. Wiese and SHM Education Committee co-chair Vikas Parekh, MD, FHM, the discussion focused on what SHM can do—or perhaps do better—in this capacity.
Dr. Parekh said hospitalists should be experts in quality-improvement (QI) and patient safety, and HM must incorporate that expertise into daily practice. However, he said, SHM’s largest educational focus is the new Focused Practice in Hospital Medicine pathway to American Board of Internal Medicine’s (ABIM) recertification.
In describing the future of this dynamic field, Dr. Weise raised concerns about managing the pipeline of approximately 2,500 new residents turning out each year and the potential for “losing intimacy” among SHM members—which he described as “the curse of being a champion.”
“IT is the only solution,” he added, “and identifying new and better ways of communicating.”
Competency-Based Train-ing (CBT) is critical to the development of new hospitalists, Dr. Wiese explained, as a supplement for what isn’t taught in residency. He posed a question: Should residencies last four or five years to incorporate additional training and career planning? “It’s an MBA paradigm of learning what we do,” he said. “What compels residents to join fellowship programs and earn $50K per year when they can start practicing and earning $150K?”
Educating the membership requires innovation and more than just bench-to-bedside research, Dr. Wiese added. Translational research and best-evidence practices will improve the field. “Five or 10 programs are rock stars,” he said, “but there are 377 that are terrible.”
Future SHM goals include a vision of having hospitalists hold 20% of all Internal Medicine Residency Program Director positions; developing best practices, not unfunded mandates; establishing protected academic time; and encouraging mentorship that positions hospitalists as heroes for the next generation.
An education committee sub-group has been tasked to focus on the recruitment of hospitalists and expose them to the best the society and field have to offer. HM10
—Phaedra Cress