User login
Vascular lesions hurt cognition more than amyloid
Vascular brain injury had a greater influence on cognitive function than did beta-amyloid deposition, at least in the earliest phases of cognitive impairment, in a study examining the relative contributions of the two abnormalities in 61 elderly participants.
The findings indicate that brain infarctions should be considered in studies of mild cognitive impairment (MCI), and that reducing cerebrovascular disease may be important in preventing MCI, said Natalie L. Marchant, Ph.D., of the Helen Wills Neuroscience Institute at the University of California, Berkeley, and her associates. They reported their findings online Feb. 11 in JAMA Neurology.
Both areas of infarction and areas of beta-amyloid deposition are frequently found on imaging studies of the brain in elderly subjects, and oftentimes both are present in the same individual, the investigators said. MRI revealed at least one infarct in 56% of the participants. PET imaging with the beta-amyloid binding ligand Pittsburgh compound B (PiB) found early beta-amyloid deposition in 48%. Overall, 41% had both an infarct and tested positive for PiB while 56% had no infarct but tested positive for PiB.
The study subjects had a range of cognitive functioning based on assessments made with the Mini-Mental State Examination, the Geriatric Depression Scale, the Clinical Dementia Rating, and a battery of neuropsychological tests of executive function, verbal memory, and nonverbal memory. Overall, 30 subjects were clinically normal, 24 showed MCI (7 with amnestic, 13 with nonamnestic, and 4 with other MCI), and 7 had dementia. The subjects were participating in an ongoing multicenter study, the Aging Brain Project.
Comparisons of the subjects’ cognitive function based on neuropsychological testing with the imaging results showed that vascular brain injury strongly correlated with cognitive impairment for verbal memory and executive function, but not nonverbal memory, the investigators said (JAMA Neurol. 2013 Feb. 11 [doi:1001/2013.jamaneurol.405]).
In contrast, beta-amyloid deposition did not correlate with cognitive impairment. The lack of an association in this sample might be because such deposition is an initiating event, and the "downstream" consequences such as neuronal death and synaptic loss have not yet occurred in these subjects, Dr. Marchant and her associates said.
In contrast, they suggested that "vascular brain injury represents end-stage pathologic consequences of the vascular pathogenic process."
An infarct in the subcortical gray matter was the only significant predictor of verbal memory in a multivariate logistic regression equation that accounted for 17% of the patient sample’s variance in verbal memory. The equation included white matter hyperintensity volume, infarct location in the subcortical gray matter, PiB status, and age, sex, and other demographic variables.
Only age and educational level were significant predictors of nonverbal memory in a multivariate logistic regression equation that accounted for 48% of the patient sample’s variance in nonverbal memory.
In a multivariate logistic regression equation that explained 43% of the variance in executive function, an infarct location in the cortical gray matter and educational level were the only significant predictors.
Beta-amyloid deposition also did not correlate with vascular brain injury. It may be that both of these abnormalities are prevalent in the aging brain, often occur concomitantly, and affect cognitive function in different ways, but do not bear any causal relationship to each other, the investigators said.
This study was supported by grants from the National Institutes of Health. Dr. Marchant reported no financial conflicts of interest, but one of her associates reported ties to numerous industry sources.
Vascular brain injury had a greater influence on cognitive function than did beta-amyloid deposition, at least in the earliest phases of cognitive impairment, in a study examining the relative contributions of the two abnormalities in 61 elderly participants.
The findings indicate that brain infarctions should be considered in studies of mild cognitive impairment (MCI), and that reducing cerebrovascular disease may be important in preventing MCI, said Natalie L. Marchant, Ph.D., of the Helen Wills Neuroscience Institute at the University of California, Berkeley, and her associates. They reported their findings online Feb. 11 in JAMA Neurology.
Both areas of infarction and areas of beta-amyloid deposition are frequently found on imaging studies of the brain in elderly subjects, and oftentimes both are present in the same individual, the investigators said. MRI revealed at least one infarct in 56% of the participants. PET imaging with the beta-amyloid binding ligand Pittsburgh compound B (PiB) found early beta-amyloid deposition in 48%. Overall, 41% had both an infarct and tested positive for PiB while 56% had no infarct but tested positive for PiB.
The study subjects had a range of cognitive functioning based on assessments made with the Mini-Mental State Examination, the Geriatric Depression Scale, the Clinical Dementia Rating, and a battery of neuropsychological tests of executive function, verbal memory, and nonverbal memory. Overall, 30 subjects were clinically normal, 24 showed MCI (7 with amnestic, 13 with nonamnestic, and 4 with other MCI), and 7 had dementia. The subjects were participating in an ongoing multicenter study, the Aging Brain Project.
Comparisons of the subjects’ cognitive function based on neuropsychological testing with the imaging results showed that vascular brain injury strongly correlated with cognitive impairment for verbal memory and executive function, but not nonverbal memory, the investigators said (JAMA Neurol. 2013 Feb. 11 [doi:1001/2013.jamaneurol.405]).
In contrast, beta-amyloid deposition did not correlate with cognitive impairment. The lack of an association in this sample might be because such deposition is an initiating event, and the "downstream" consequences such as neuronal death and synaptic loss have not yet occurred in these subjects, Dr. Marchant and her associates said.
In contrast, they suggested that "vascular brain injury represents end-stage pathologic consequences of the vascular pathogenic process."
An infarct in the subcortical gray matter was the only significant predictor of verbal memory in a multivariate logistic regression equation that accounted for 17% of the patient sample’s variance in verbal memory. The equation included white matter hyperintensity volume, infarct location in the subcortical gray matter, PiB status, and age, sex, and other demographic variables.
Only age and educational level were significant predictors of nonverbal memory in a multivariate logistic regression equation that accounted for 48% of the patient sample’s variance in nonverbal memory.
In a multivariate logistic regression equation that explained 43% of the variance in executive function, an infarct location in the cortical gray matter and educational level were the only significant predictors.
Beta-amyloid deposition also did not correlate with vascular brain injury. It may be that both of these abnormalities are prevalent in the aging brain, often occur concomitantly, and affect cognitive function in different ways, but do not bear any causal relationship to each other, the investigators said.
This study was supported by grants from the National Institutes of Health. Dr. Marchant reported no financial conflicts of interest, but one of her associates reported ties to numerous industry sources.
Vascular brain injury had a greater influence on cognitive function than did beta-amyloid deposition, at least in the earliest phases of cognitive impairment, in a study examining the relative contributions of the two abnormalities in 61 elderly participants.
The findings indicate that brain infarctions should be considered in studies of mild cognitive impairment (MCI), and that reducing cerebrovascular disease may be important in preventing MCI, said Natalie L. Marchant, Ph.D., of the Helen Wills Neuroscience Institute at the University of California, Berkeley, and her associates. They reported their findings online Feb. 11 in JAMA Neurology.
Both areas of infarction and areas of beta-amyloid deposition are frequently found on imaging studies of the brain in elderly subjects, and oftentimes both are present in the same individual, the investigators said. MRI revealed at least one infarct in 56% of the participants. PET imaging with the beta-amyloid binding ligand Pittsburgh compound B (PiB) found early beta-amyloid deposition in 48%. Overall, 41% had both an infarct and tested positive for PiB while 56% had no infarct but tested positive for PiB.
The study subjects had a range of cognitive functioning based on assessments made with the Mini-Mental State Examination, the Geriatric Depression Scale, the Clinical Dementia Rating, and a battery of neuropsychological tests of executive function, verbal memory, and nonverbal memory. Overall, 30 subjects were clinically normal, 24 showed MCI (7 with amnestic, 13 with nonamnestic, and 4 with other MCI), and 7 had dementia. The subjects were participating in an ongoing multicenter study, the Aging Brain Project.
Comparisons of the subjects’ cognitive function based on neuropsychological testing with the imaging results showed that vascular brain injury strongly correlated with cognitive impairment for verbal memory and executive function, but not nonverbal memory, the investigators said (JAMA Neurol. 2013 Feb. 11 [doi:1001/2013.jamaneurol.405]).
In contrast, beta-amyloid deposition did not correlate with cognitive impairment. The lack of an association in this sample might be because such deposition is an initiating event, and the "downstream" consequences such as neuronal death and synaptic loss have not yet occurred in these subjects, Dr. Marchant and her associates said.
In contrast, they suggested that "vascular brain injury represents end-stage pathologic consequences of the vascular pathogenic process."
An infarct in the subcortical gray matter was the only significant predictor of verbal memory in a multivariate logistic regression equation that accounted for 17% of the patient sample’s variance in verbal memory. The equation included white matter hyperintensity volume, infarct location in the subcortical gray matter, PiB status, and age, sex, and other demographic variables.
Only age and educational level were significant predictors of nonverbal memory in a multivariate logistic regression equation that accounted for 48% of the patient sample’s variance in nonverbal memory.
In a multivariate logistic regression equation that explained 43% of the variance in executive function, an infarct location in the cortical gray matter and educational level were the only significant predictors.
Beta-amyloid deposition also did not correlate with vascular brain injury. It may be that both of these abnormalities are prevalent in the aging brain, often occur concomitantly, and affect cognitive function in different ways, but do not bear any causal relationship to each other, the investigators said.
This study was supported by grants from the National Institutes of Health. Dr. Marchant reported no financial conflicts of interest, but one of her associates reported ties to numerous industry sources.
FROM JAMA NEUROLOGY
Major Finding: An infarct in the subcortical gray matter was the only significant predictor of verbal memory in a logistic regression equation that accounted for 17% of variance in verbal memory, and an infarct in cortical gray matter was a significant predictor (along with educational level) in a logistic regression equation that accounted for 43% of the variance in executive function. Neither infarct nor PiB status were significant predictors of nonverbal memory.
Data Source: A cross-sectional study of MRI and PET brain imaging in 61 elderly subjects who had normal cognitive function, mild cognitive impairment, or dementia.
Disclosures: This study was supported by grants from the National Institutes of Health. Dr. Marchant reported no financial conflicts of interest, but one of her associates reported ties to numerous industry sources.
Screen asymptomatic siblings of CRC patients
Asymptomatic siblings of Chinese colorectal cancer patients are at threefold higher risk for advanced colorectal neoplasms and at twofold higher risk for any colorectal adenoma, compared with siblings of healthy Chinese adults, Dr. Siew C. Ng and her colleagues reported in the March issue of Gastroenterology (doi:10.1053/j.gastro.2012.11.011).
Given these findings from a large prospective cross-sectional study, colorectal screening, with the removal of any premalignant lesions that are found, is warranted in this high-risk group, said Dr. Ng of the Prince of Wales Hospital and the Chinese University of Hong Kong and her associates.
Source: American Gastroenterological Association
Current guidelines recommend earlier and more frequent screening of close relatives of patients who have colorectal cancer, but what to expect on these screenings is unclear because "data from well-conducted prospective studies are lacking," they said.
Dr. Ng and her colleagues compared the prevalence of advanced neoplasms in such siblings against the prevalence in siblings of patients who underwent colorectal screening but had normal results. "The use of such a control group avoids a biased estimate of the association with family history, and removes the acquired or environmental component to this association," the investigators wrote.
All the screenings used a conventional white-light colonocope without high definition and were performed with patients under conscious sedation with intravenous midazolam and pethidine.
During a period of 10 years, 374 siblings (mean age, 53 years) of CRC patients aged 40-70 years participated in the study, as did 374 age- and sex-matched control subjects. The quality of bowel preparation was similar between the two groups.
All three study endoscopists were experienced, and they had comparable rates of adenoma detection.
The primary outcome was the prevalence of advanced neoplasms, defined as cancers or adenomas at least 10 mm in diameter that had high-grade dysplasia or villous/tubovillous histologic traits. This prevalence was approximately three times higher in the siblings of CRC patients (7.5%) as in the siblings of healthy controls (2.9%), with an odds ratio of 3.07.
Adenocarcinomas were detected in six siblings of CRC patients, but in none of the control subjects. These included two stage I cancers, two stage II cancers, and two stage III cancers.
Similarly, the prevalence of large adenomas was approximately three times as high in siblings of CRC patients (5.9%) as in controls (2.1%).
Siblings of CRC patients also had a higher prevalence of smaller adenomas (31%) than did control subjects (18.2%).
When the data were analyzed by lesion location, the siblings of CRC patients had a higher prevalence of every type: distal adenomas (13.1% vs. 8.3%), proximal adenomas (12.0% vs. 6.2%), and synchronous adenomas (5.9% vs. 2.7%).
The prevalence of hyperplastic polyps was comparable between the two groups (27.3% and 21.4%).
When the data were analyzed by subject age, siblings of CRC patients had a higher prevalence of all colorectal adenomas whether they were younger than 50 years (21.0% vs. 9.8%), 50-60 years old (34.4% vs. 23.9%), or older than 60 years (41.0% vs. 20.5%).
The study findings remained robust in two further sensitivity analyses.
Among the siblings of CRC patients, the risk of detecting an advanced adenoma was higher if the affected sibling’s cancer was located in the distal colon than if it was located in the proximal colon. This risk also was higher if the affected sibling was a woman than a man; however, this finding must be interpreted with caution because the number of subjects in these subgroups was small.
These study results can be used to "provide a background against which screening strategies can be formulated." More important, the strong, significantly increased risk of advanced neoplasms means that siblings of CRC patients should be screened carefully, Dr. Ng and her associates said.
Asymptomatic siblings of Chinese colorectal cancer patients are at threefold higher risk for advanced colorectal neoplasms and at twofold higher risk for any colorectal adenoma, compared with siblings of healthy Chinese adults, Dr. Siew C. Ng and her colleagues reported in the March issue of Gastroenterology (doi:10.1053/j.gastro.2012.11.011).
Given these findings from a large prospective cross-sectional study, colorectal screening, with the removal of any premalignant lesions that are found, is warranted in this high-risk group, said Dr. Ng of the Prince of Wales Hospital and the Chinese University of Hong Kong and her associates.
Source: American Gastroenterological Association
Current guidelines recommend earlier and more frequent screening of close relatives of patients who have colorectal cancer, but what to expect on these screenings is unclear because "data from well-conducted prospective studies are lacking," they said.
Dr. Ng and her colleagues compared the prevalence of advanced neoplasms in such siblings against the prevalence in siblings of patients who underwent colorectal screening but had normal results. "The use of such a control group avoids a biased estimate of the association with family history, and removes the acquired or environmental component to this association," the investigators wrote.
All the screenings used a conventional white-light colonocope without high definition and were performed with patients under conscious sedation with intravenous midazolam and pethidine.
During a period of 10 years, 374 siblings (mean age, 53 years) of CRC patients aged 40-70 years participated in the study, as did 374 age- and sex-matched control subjects. The quality of bowel preparation was similar between the two groups.
All three study endoscopists were experienced, and they had comparable rates of adenoma detection.
The primary outcome was the prevalence of advanced neoplasms, defined as cancers or adenomas at least 10 mm in diameter that had high-grade dysplasia or villous/tubovillous histologic traits. This prevalence was approximately three times higher in the siblings of CRC patients (7.5%) as in the siblings of healthy controls (2.9%), with an odds ratio of 3.07.
Adenocarcinomas were detected in six siblings of CRC patients, but in none of the control subjects. These included two stage I cancers, two stage II cancers, and two stage III cancers.
Similarly, the prevalence of large adenomas was approximately three times as high in siblings of CRC patients (5.9%) as in controls (2.1%).
Siblings of CRC patients also had a higher prevalence of smaller adenomas (31%) than did control subjects (18.2%).
When the data were analyzed by lesion location, the siblings of CRC patients had a higher prevalence of every type: distal adenomas (13.1% vs. 8.3%), proximal adenomas (12.0% vs. 6.2%), and synchronous adenomas (5.9% vs. 2.7%).
The prevalence of hyperplastic polyps was comparable between the two groups (27.3% and 21.4%).
When the data were analyzed by subject age, siblings of CRC patients had a higher prevalence of all colorectal adenomas whether they were younger than 50 years (21.0% vs. 9.8%), 50-60 years old (34.4% vs. 23.9%), or older than 60 years (41.0% vs. 20.5%).
The study findings remained robust in two further sensitivity analyses.
Among the siblings of CRC patients, the risk of detecting an advanced adenoma was higher if the affected sibling’s cancer was located in the distal colon than if it was located in the proximal colon. This risk also was higher if the affected sibling was a woman than a man; however, this finding must be interpreted with caution because the number of subjects in these subgroups was small.
These study results can be used to "provide a background against which screening strategies can be formulated." More important, the strong, significantly increased risk of advanced neoplasms means that siblings of CRC patients should be screened carefully, Dr. Ng and her associates said.
Asymptomatic siblings of Chinese colorectal cancer patients are at threefold higher risk for advanced colorectal neoplasms and at twofold higher risk for any colorectal adenoma, compared with siblings of healthy Chinese adults, Dr. Siew C. Ng and her colleagues reported in the March issue of Gastroenterology (doi:10.1053/j.gastro.2012.11.011).
Given these findings from a large prospective cross-sectional study, colorectal screening, with the removal of any premalignant lesions that are found, is warranted in this high-risk group, said Dr. Ng of the Prince of Wales Hospital and the Chinese University of Hong Kong and her associates.
Source: American Gastroenterological Association
Current guidelines recommend earlier and more frequent screening of close relatives of patients who have colorectal cancer, but what to expect on these screenings is unclear because "data from well-conducted prospective studies are lacking," they said.
Dr. Ng and her colleagues compared the prevalence of advanced neoplasms in such siblings against the prevalence in siblings of patients who underwent colorectal screening but had normal results. "The use of such a control group avoids a biased estimate of the association with family history, and removes the acquired or environmental component to this association," the investigators wrote.
All the screenings used a conventional white-light colonocope without high definition and were performed with patients under conscious sedation with intravenous midazolam and pethidine.
During a period of 10 years, 374 siblings (mean age, 53 years) of CRC patients aged 40-70 years participated in the study, as did 374 age- and sex-matched control subjects. The quality of bowel preparation was similar between the two groups.
All three study endoscopists were experienced, and they had comparable rates of adenoma detection.
The primary outcome was the prevalence of advanced neoplasms, defined as cancers or adenomas at least 10 mm in diameter that had high-grade dysplasia or villous/tubovillous histologic traits. This prevalence was approximately three times higher in the siblings of CRC patients (7.5%) as in the siblings of healthy controls (2.9%), with an odds ratio of 3.07.
Adenocarcinomas were detected in six siblings of CRC patients, but in none of the control subjects. These included two stage I cancers, two stage II cancers, and two stage III cancers.
Similarly, the prevalence of large adenomas was approximately three times as high in siblings of CRC patients (5.9%) as in controls (2.1%).
Siblings of CRC patients also had a higher prevalence of smaller adenomas (31%) than did control subjects (18.2%).
When the data were analyzed by lesion location, the siblings of CRC patients had a higher prevalence of every type: distal adenomas (13.1% vs. 8.3%), proximal adenomas (12.0% vs. 6.2%), and synchronous adenomas (5.9% vs. 2.7%).
The prevalence of hyperplastic polyps was comparable between the two groups (27.3% and 21.4%).
When the data were analyzed by subject age, siblings of CRC patients had a higher prevalence of all colorectal adenomas whether they were younger than 50 years (21.0% vs. 9.8%), 50-60 years old (34.4% vs. 23.9%), or older than 60 years (41.0% vs. 20.5%).
The study findings remained robust in two further sensitivity analyses.
Among the siblings of CRC patients, the risk of detecting an advanced adenoma was higher if the affected sibling’s cancer was located in the distal colon than if it was located in the proximal colon. This risk also was higher if the affected sibling was a woman than a man; however, this finding must be interpreted with caution because the number of subjects in these subgroups was small.
These study results can be used to "provide a background against which screening strategies can be formulated." More important, the strong, significantly increased risk of advanced neoplasms means that siblings of CRC patients should be screened carefully, Dr. Ng and her associates said.
GASTROENTEROLOGY
Major Finding: The prevalence of advanced colorectal neoplasms was approximately three times higher among siblings of colorectal cancer patients (7.5%) as among siblings of adults who had normal colonoscopy results (2.9%).
Data Source: A prospective cross-sectional study of advanced colorectal neoplasms in 374 siblings of Chinese colorectal cancer patients and 374 age- and sex-matched siblings of Chinese volunteers who had negative colonoscopy results.
Disclosures: This study was supported by the Hong Kong Society of Gastroenterology and the Chinese University of Hong Kong. No financial conflicts of interest were reported.
Folic acid supplements have no effect on cancer rates
Taking folic acid supplements neither increases nor decreases the incidence of cancer during the treatment period, according to a meta-analysis of 13 large randomized controlled trials that examined this issue, which was reported online Jan. 25 in the Lancet.
Folic acid fortification of flour to prevent neural tube defects has been mandatory in several countries beginning in the late 1990s, and since then further supplementation has been touted as a possible cancer preventive. But several other countries do not allow folic acid fortification and discourage supplementation, partly because of fear that it could promote cancer incidence or progression.
The investigators reviewed all the randomized trials published in the medical literature through 2010 that assessed the effects of folic acid supplements. Trials were included in the meta-analysis if they contained at least 500 subjects, assessed cancer incidence as an outcome, and randomized subjects to either supplements or placebo for at least 1 year. The researchers included 13 trials with 49,621 subjects in their meta-analysis.Neither the hope that folic acid supplements might prevent cancer nor the fear that they might promote it were borne out by this meta-analysis, said Dr. Stein Emil Vollset of the Norwegian Institute of Public Health and the University of Bergen (Norway), and his associates.
Two-thirds of the participants were men, and the mean age at baseline was 64 years. Daily doses of folic acid ranged from 0.5 mg to 5 mg in 12 of the studies, and another study assessed a high dose of 40 mg daily. The duration of treatment was 1.8-7.4 years (mean, 5.2 years).
Taking the supplements was associated with a quadrupling of plasma levels of folate and a 25% reduction in plasma homocysteine levels. In the single trial of high-dose folic acid, supplements raised plasma levels of folate 100-fold but produced little further effect on plasma homocysteine levels.
Folic acid supplementation had no significant effect on the incidence of cancer overall. The rate of all types of cancer was 7.7% in subjects who took the supplements and 7.3% in subjects who took placebo, a nonsignificant difference, Dr. Vollset and his colleagues reported (Lancet 2013 [doi: 10.1016/S0140-6736(12)62001-7]).
The overall cancer incidence did not change with increasing duration of treatment or increasing dose of the supplements, nor did it vary by patient age, sex, baseline folate levels, baseline homocysteine levels, or the use of flour fortification in the country of residence.
Even in the trial of high-dose supplements that raised plasma levels of folate 100-fold, there was no significant difference in overall cancer incidence between the supplement group (65 cancers) and the placebo group (72 cancers).
Folic acid supplementation also had no effect on the incidence of site-specific cancers, including colorectal, lung, breast, and prostate cancers. Thus, these trials "provide no significant evidence of short-term effects of folic acid supplementation on overall cancer incidence or on the incidence of any particular type of cancer," the researchers said.
Neither this meta-analysis nor the randomized controlled trials it was based on addressed whether there are longer-term beneficial or harmful effects of folic acid supplementation, they added.
This study was supported by the British Heart Foundation, the UK Medical Research Council, Cancer Research UK, and the UK Food Standards Agency.
Taking folic acid supplements neither increases nor decreases the incidence of cancer during the treatment period, according to a meta-analysis of 13 large randomized controlled trials that examined this issue, which was reported online Jan. 25 in the Lancet.
Folic acid fortification of flour to prevent neural tube defects has been mandatory in several countries beginning in the late 1990s, and since then further supplementation has been touted as a possible cancer preventive. But several other countries do not allow folic acid fortification and discourage supplementation, partly because of fear that it could promote cancer incidence or progression.
The investigators reviewed all the randomized trials published in the medical literature through 2010 that assessed the effects of folic acid supplements. Trials were included in the meta-analysis if they contained at least 500 subjects, assessed cancer incidence as an outcome, and randomized subjects to either supplements or placebo for at least 1 year. The researchers included 13 trials with 49,621 subjects in their meta-analysis.Neither the hope that folic acid supplements might prevent cancer nor the fear that they might promote it were borne out by this meta-analysis, said Dr. Stein Emil Vollset of the Norwegian Institute of Public Health and the University of Bergen (Norway), and his associates.
Two-thirds of the participants were men, and the mean age at baseline was 64 years. Daily doses of folic acid ranged from 0.5 mg to 5 mg in 12 of the studies, and another study assessed a high dose of 40 mg daily. The duration of treatment was 1.8-7.4 years (mean, 5.2 years).
Taking the supplements was associated with a quadrupling of plasma levels of folate and a 25% reduction in plasma homocysteine levels. In the single trial of high-dose folic acid, supplements raised plasma levels of folate 100-fold but produced little further effect on plasma homocysteine levels.
Folic acid supplementation had no significant effect on the incidence of cancer overall. The rate of all types of cancer was 7.7% in subjects who took the supplements and 7.3% in subjects who took placebo, a nonsignificant difference, Dr. Vollset and his colleagues reported (Lancet 2013 [doi: 10.1016/S0140-6736(12)62001-7]).
The overall cancer incidence did not change with increasing duration of treatment or increasing dose of the supplements, nor did it vary by patient age, sex, baseline folate levels, baseline homocysteine levels, or the use of flour fortification in the country of residence.
Even in the trial of high-dose supplements that raised plasma levels of folate 100-fold, there was no significant difference in overall cancer incidence between the supplement group (65 cancers) and the placebo group (72 cancers).
Folic acid supplementation also had no effect on the incidence of site-specific cancers, including colorectal, lung, breast, and prostate cancers. Thus, these trials "provide no significant evidence of short-term effects of folic acid supplementation on overall cancer incidence or on the incidence of any particular type of cancer," the researchers said.
Neither this meta-analysis nor the randomized controlled trials it was based on addressed whether there are longer-term beneficial or harmful effects of folic acid supplementation, they added.
This study was supported by the British Heart Foundation, the UK Medical Research Council, Cancer Research UK, and the UK Food Standards Agency.
Taking folic acid supplements neither increases nor decreases the incidence of cancer during the treatment period, according to a meta-analysis of 13 large randomized controlled trials that examined this issue, which was reported online Jan. 25 in the Lancet.
Folic acid fortification of flour to prevent neural tube defects has been mandatory in several countries beginning in the late 1990s, and since then further supplementation has been touted as a possible cancer preventive. But several other countries do not allow folic acid fortification and discourage supplementation, partly because of fear that it could promote cancer incidence or progression.
The investigators reviewed all the randomized trials published in the medical literature through 2010 that assessed the effects of folic acid supplements. Trials were included in the meta-analysis if they contained at least 500 subjects, assessed cancer incidence as an outcome, and randomized subjects to either supplements or placebo for at least 1 year. The researchers included 13 trials with 49,621 subjects in their meta-analysis.Neither the hope that folic acid supplements might prevent cancer nor the fear that they might promote it were borne out by this meta-analysis, said Dr. Stein Emil Vollset of the Norwegian Institute of Public Health and the University of Bergen (Norway), and his associates.
Two-thirds of the participants were men, and the mean age at baseline was 64 years. Daily doses of folic acid ranged from 0.5 mg to 5 mg in 12 of the studies, and another study assessed a high dose of 40 mg daily. The duration of treatment was 1.8-7.4 years (mean, 5.2 years).
Taking the supplements was associated with a quadrupling of plasma levels of folate and a 25% reduction in plasma homocysteine levels. In the single trial of high-dose folic acid, supplements raised plasma levels of folate 100-fold but produced little further effect on plasma homocysteine levels.
Folic acid supplementation had no significant effect on the incidence of cancer overall. The rate of all types of cancer was 7.7% in subjects who took the supplements and 7.3% in subjects who took placebo, a nonsignificant difference, Dr. Vollset and his colleagues reported (Lancet 2013 [doi: 10.1016/S0140-6736(12)62001-7]).
The overall cancer incidence did not change with increasing duration of treatment or increasing dose of the supplements, nor did it vary by patient age, sex, baseline folate levels, baseline homocysteine levels, or the use of flour fortification in the country of residence.
Even in the trial of high-dose supplements that raised plasma levels of folate 100-fold, there was no significant difference in overall cancer incidence between the supplement group (65 cancers) and the placebo group (72 cancers).
Folic acid supplementation also had no effect on the incidence of site-specific cancers, including colorectal, lung, breast, and prostate cancers. Thus, these trials "provide no significant evidence of short-term effects of folic acid supplementation on overall cancer incidence or on the incidence of any particular type of cancer," the researchers said.
Neither this meta-analysis nor the randomized controlled trials it was based on addressed whether there are longer-term beneficial or harmful effects of folic acid supplementation, they added.
This study was supported by the British Heart Foundation, the UK Medical Research Council, Cancer Research UK, and the UK Food Standards Agency.
FROM THE LANCET
Major finding: Taking folic acid supplements had no significant effect on overall cancer incidence (7.7% vs. 7.3% with placebo) or on the incidence of any specific type of cancer.
Data source: A meta-analysis of 13 clinical trials (49,621 total subjects) that included at least 500 subjects each, randomly assigned subjects to receive folic acid supplements or placebo for at least 1 year, and assessed cancer incidence as an outcome.
Disclosures: This study was supported by the British Heart Foundation, the UK Medical Research Council, Cancer Research UK, and the UK Food Standards Agency.
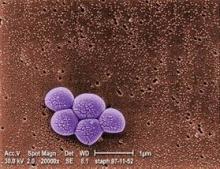
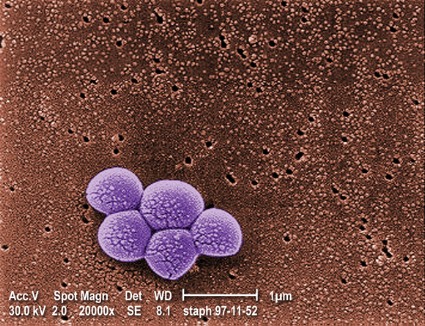
Daily chlorhexidine bath cuts bloodstream, MRSA infections in ICUs
For patients in intensive care units and bone marrow transplantation units, daily bathing with chlorhexidine-impregnated washcloths significantly reduced the incidence of bloodstream infections and decreased the acquisition of multidrug-resistant organisms in a randomized controlled trial reported online Feb. 7 in the New England Journal of Medicine.
The multicenter study, which included nine medical, coronary care, surgical, cardiac surgery, and bone marrow transplantation units in several geographic regions of the United States, confirms the results of previous single-center studies and demonstrates that the benefit of daily chlorhexidine bathing is widely generalizable, said Dr. Michael W. Climo of the Hunter Holmes McGuire Veterans Affairs Medical Center, Richmond, Va., and his associates.
Each hospital unit participating in the trial was randomized to perform daily bathing of patients using either washcloths impregnated with 2% chlorhexidine gluconate (intervention) or washcloths with no impregnated antimicrobial (control) for 6 months, followed by a crossover period in which patients were bathed daily with the alternate product for another 6 months. A total of 7,727 patients were included in the study.
Nurses were trained in the manufacturer’s instructions for bathing, which involved using the cloths in a sequential order to rinse all body surfaces except the face (to avoid getting chlorhexidine in the patient’s eyes and mouth). Implementing this cost-effective strategy was "relatively straightforward" because it did not require much of a change from current routine patient-bathing practices, the investigators said.
All the hospital units performed active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococcus (VRE) throughout the study.
There were 119 hospital-acquired bloodstream infections during the intervention and 165 during the control period, representing a 28% reduction with chlorhexidine bathing. The rate of primary bloodstream infections was 31% lower and the rate of central catheter–associated bloodstream infections was 53% lower with the intervention, Dr. Climo and his associates reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1113849]).
This benefit was greatest among patients who had longer lengths of stay in the hospital units.
One "unanticipated" finding was that rates of fungal infection in particular were dramatically reduced with chlorhexidine bathing. The incidence of primary bloodstream infection caused by fungi was 53% lower and the incidence of central catheter–associated fungal bloodstream infection was 90% lower during the intervention period than during the control period.
This antifungal effect has not been reported previously. "If our results are confirmed, topical use of chlorhexidine could be added to strategies to prevent fungal infection," the researchers noted.
The rate of MRSA or VRE acquisition was 23% lower during the intervention period (5.10 cases per 1,000 patient-days) than during the control period (6.60 cases per 1,000 patient-days). However, reductions in the incidence of MRSA and VRE bacteremia did not reach statistical significance, most likely because the overall number of cases was so low, Dr. Climo and his colleagues said.
No serious adverse effects were observed. The incidence of skin reactions was higher with the control washcloths (3.4%) than with the chlorhexidine washcloths (2.0%), and none of the skin reactions were considered to be related to the bathing intervention.
It was reassuring that none of the MRSA or VRE isolates that were detected in this study showed resistance to chlorhexidine. However, the development of resistance to biocides and disinfectants among nosocomial organisms remains "a substantial concern" and should continue to be monitored, they added.
This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
For patients in intensive care units and bone marrow transplantation units, daily bathing with chlorhexidine-impregnated washcloths significantly reduced the incidence of bloodstream infections and decreased the acquisition of multidrug-resistant organisms in a randomized controlled trial reported online Feb. 7 in the New England Journal of Medicine.
The multicenter study, which included nine medical, coronary care, surgical, cardiac surgery, and bone marrow transplantation units in several geographic regions of the United States, confirms the results of previous single-center studies and demonstrates that the benefit of daily chlorhexidine bathing is widely generalizable, said Dr. Michael W. Climo of the Hunter Holmes McGuire Veterans Affairs Medical Center, Richmond, Va., and his associates.
Each hospital unit participating in the trial was randomized to perform daily bathing of patients using either washcloths impregnated with 2% chlorhexidine gluconate (intervention) or washcloths with no impregnated antimicrobial (control) for 6 months, followed by a crossover period in which patients were bathed daily with the alternate product for another 6 months. A total of 7,727 patients were included in the study.
Nurses were trained in the manufacturer’s instructions for bathing, which involved using the cloths in a sequential order to rinse all body surfaces except the face (to avoid getting chlorhexidine in the patient’s eyes and mouth). Implementing this cost-effective strategy was "relatively straightforward" because it did not require much of a change from current routine patient-bathing practices, the investigators said.
All the hospital units performed active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococcus (VRE) throughout the study.
There were 119 hospital-acquired bloodstream infections during the intervention and 165 during the control period, representing a 28% reduction with chlorhexidine bathing. The rate of primary bloodstream infections was 31% lower and the rate of central catheter–associated bloodstream infections was 53% lower with the intervention, Dr. Climo and his associates reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1113849]).
This benefit was greatest among patients who had longer lengths of stay in the hospital units.
One "unanticipated" finding was that rates of fungal infection in particular were dramatically reduced with chlorhexidine bathing. The incidence of primary bloodstream infection caused by fungi was 53% lower and the incidence of central catheter–associated fungal bloodstream infection was 90% lower during the intervention period than during the control period.
This antifungal effect has not been reported previously. "If our results are confirmed, topical use of chlorhexidine could be added to strategies to prevent fungal infection," the researchers noted.
The rate of MRSA or VRE acquisition was 23% lower during the intervention period (5.10 cases per 1,000 patient-days) than during the control period (6.60 cases per 1,000 patient-days). However, reductions in the incidence of MRSA and VRE bacteremia did not reach statistical significance, most likely because the overall number of cases was so low, Dr. Climo and his colleagues said.
No serious adverse effects were observed. The incidence of skin reactions was higher with the control washcloths (3.4%) than with the chlorhexidine washcloths (2.0%), and none of the skin reactions were considered to be related to the bathing intervention.
It was reassuring that none of the MRSA or VRE isolates that were detected in this study showed resistance to chlorhexidine. However, the development of resistance to biocides and disinfectants among nosocomial organisms remains "a substantial concern" and should continue to be monitored, they added.
This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
For patients in intensive care units and bone marrow transplantation units, daily bathing with chlorhexidine-impregnated washcloths significantly reduced the incidence of bloodstream infections and decreased the acquisition of multidrug-resistant organisms in a randomized controlled trial reported online Feb. 7 in the New England Journal of Medicine.
The multicenter study, which included nine medical, coronary care, surgical, cardiac surgery, and bone marrow transplantation units in several geographic regions of the United States, confirms the results of previous single-center studies and demonstrates that the benefit of daily chlorhexidine bathing is widely generalizable, said Dr. Michael W. Climo of the Hunter Holmes McGuire Veterans Affairs Medical Center, Richmond, Va., and his associates.
Each hospital unit participating in the trial was randomized to perform daily bathing of patients using either washcloths impregnated with 2% chlorhexidine gluconate (intervention) or washcloths with no impregnated antimicrobial (control) for 6 months, followed by a crossover period in which patients were bathed daily with the alternate product for another 6 months. A total of 7,727 patients were included in the study.
Nurses were trained in the manufacturer’s instructions for bathing, which involved using the cloths in a sequential order to rinse all body surfaces except the face (to avoid getting chlorhexidine in the patient’s eyes and mouth). Implementing this cost-effective strategy was "relatively straightforward" because it did not require much of a change from current routine patient-bathing practices, the investigators said.
All the hospital units performed active surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococcus (VRE) throughout the study.
There were 119 hospital-acquired bloodstream infections during the intervention and 165 during the control period, representing a 28% reduction with chlorhexidine bathing. The rate of primary bloodstream infections was 31% lower and the rate of central catheter–associated bloodstream infections was 53% lower with the intervention, Dr. Climo and his associates reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1113849]).
This benefit was greatest among patients who had longer lengths of stay in the hospital units.
One "unanticipated" finding was that rates of fungal infection in particular were dramatically reduced with chlorhexidine bathing. The incidence of primary bloodstream infection caused by fungi was 53% lower and the incidence of central catheter–associated fungal bloodstream infection was 90% lower during the intervention period than during the control period.
This antifungal effect has not been reported previously. "If our results are confirmed, topical use of chlorhexidine could be added to strategies to prevent fungal infection," the researchers noted.
The rate of MRSA or VRE acquisition was 23% lower during the intervention period (5.10 cases per 1,000 patient-days) than during the control period (6.60 cases per 1,000 patient-days). However, reductions in the incidence of MRSA and VRE bacteremia did not reach statistical significance, most likely because the overall number of cases was so low, Dr. Climo and his colleagues said.
No serious adverse effects were observed. The incidence of skin reactions was higher with the control washcloths (3.4%) than with the chlorhexidine washcloths (2.0%), and none of the skin reactions were considered to be related to the bathing intervention.
It was reassuring that none of the MRSA or VRE isolates that were detected in this study showed resistance to chlorhexidine. However, the development of resistance to biocides and disinfectants among nosocomial organisms remains "a substantial concern" and should continue to be monitored, they added.
This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Bathing patients daily with chlorhexidine-impregnated washcloths reduced the rate of bloodstream infections by 28%, the rate of central catheter–associated infections by 53%, the rate of fungal bloodstream infections by 53%, and the rate of MRSA or VRE acquisition by 23%.
Data Source: A multicenter, randomized controlled trial assessing the ability of chlorhexidine bathing of 7,727 intensive care patients to reduce bloodstream infections and the acquisition of resistant organisms.
Disclosures: This study was supported by the Centers for Disease Control and Prevention and Sage Products. Sage Products supplied the washcloths, provided technical and educational support, and participated in weekly teleconferences with the research group but was not involved in the study design, data analysis, or reporting of the study results. Dr. Climo and his associates reported ties to Sage Products, Centene, and other companies.
MAGELLAN trial results support rivaroxaban for DVT prophylaxis
Nearly 2 years after their presentation at a national meeting, the results of MAGELLAN, comparing rivaroxaban to enoxaparin for thromboprophylaxis in critically ill hospitalized patients, have been published online Feb. 6 in the New England Journal of Medicine.
In that large, multicenter trial, rivaroxaban was noninferior to enoxaparin when used for the standard 10-day duration, and superior when used for an extended 35-day duration, for thromboprophylaxis.
However, the rate of clinically relevant bleeding and the incidence of major bleeding events were significantly higher with rivaroxaban than with enoxaparin. This adverse effect apparently balanced out the reduction in venous thromboembolism, as a prespecified analysis of net clinical benefit and harm did not show either one at day 10 or day 35, reported Dr. Alexander T. Cohen and his associates in MAGELLAN (Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin). The results were first presented by Dr. Cohen, of King's College Hospital, London, at the annual meeting of the American College of Cardiology in spring 2011.
In the 2.5-year study, 8,101 patients at 556 sites in 52 countries were randomized to receive rivaroxaban (4,050 subjects) or enoxaparin (4,051 subjects). Hundreds of patients in both study groups were excluded from the final analyses for various reasons, usually because they could not be assessed for venous thromboembolism using ultrasonography and other imaging techniques in a timely fashion, as required by the study protocol.
At 10 days' follow-up, 2.7% of patients in both groups had a primary outcome event - asymptomatic proximal deep vein thrombosis (DVT), symptomatic nonfatal pulmonary embolism (PE), or thromboembolism-related death - so rivaroxaban met the criteria for noninferiority to enoxaparin.
At the 35-day follow-up, 4.4% of subjects who received extended-duration rivaroxaban and 5.7% of those who received enoxaparin plus placebo had a primary outcome event, so rivaroxaban met the criteria for superiority to enoxaparin, the investigators reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1111096]).
Rivaroxaban (Xarelto)has several indications: for stroke prevention in nonvalvular atrial fibrillation, for the treatment of DVT and PE, for reducing the risk of recurrence of DVT and PE, and for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. It is not indicated for the condition studied in MAGELLAN, namely prevention of VTE in acutely medically ill patients.
This study was funded by Bayer HealthCare and Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen and his associates reported numerous ties to industry sources.
Nearly 2 years after their presentation at a national meeting, the results of MAGELLAN, comparing rivaroxaban to enoxaparin for thromboprophylaxis in critically ill hospitalized patients, have been published online Feb. 6 in the New England Journal of Medicine.
In that large, multicenter trial, rivaroxaban was noninferior to enoxaparin when used for the standard 10-day duration, and superior when used for an extended 35-day duration, for thromboprophylaxis.
However, the rate of clinically relevant bleeding and the incidence of major bleeding events were significantly higher with rivaroxaban than with enoxaparin. This adverse effect apparently balanced out the reduction in venous thromboembolism, as a prespecified analysis of net clinical benefit and harm did not show either one at day 10 or day 35, reported Dr. Alexander T. Cohen and his associates in MAGELLAN (Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin). The results were first presented by Dr. Cohen, of King's College Hospital, London, at the annual meeting of the American College of Cardiology in spring 2011.
In the 2.5-year study, 8,101 patients at 556 sites in 52 countries were randomized to receive rivaroxaban (4,050 subjects) or enoxaparin (4,051 subjects). Hundreds of patients in both study groups were excluded from the final analyses for various reasons, usually because they could not be assessed for venous thromboembolism using ultrasonography and other imaging techniques in a timely fashion, as required by the study protocol.
At 10 days' follow-up, 2.7% of patients in both groups had a primary outcome event - asymptomatic proximal deep vein thrombosis (DVT), symptomatic nonfatal pulmonary embolism (PE), or thromboembolism-related death - so rivaroxaban met the criteria for noninferiority to enoxaparin.
At the 35-day follow-up, 4.4% of subjects who received extended-duration rivaroxaban and 5.7% of those who received enoxaparin plus placebo had a primary outcome event, so rivaroxaban met the criteria for superiority to enoxaparin, the investigators reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1111096]).
Rivaroxaban (Xarelto)has several indications: for stroke prevention in nonvalvular atrial fibrillation, for the treatment of DVT and PE, for reducing the risk of recurrence of DVT and PE, and for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. It is not indicated for the condition studied in MAGELLAN, namely prevention of VTE in acutely medically ill patients.
This study was funded by Bayer HealthCare and Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen and his associates reported numerous ties to industry sources.
Nearly 2 years after their presentation at a national meeting, the results of MAGELLAN, comparing rivaroxaban to enoxaparin for thromboprophylaxis in critically ill hospitalized patients, have been published online Feb. 6 in the New England Journal of Medicine.
In that large, multicenter trial, rivaroxaban was noninferior to enoxaparin when used for the standard 10-day duration, and superior when used for an extended 35-day duration, for thromboprophylaxis.
However, the rate of clinically relevant bleeding and the incidence of major bleeding events were significantly higher with rivaroxaban than with enoxaparin. This adverse effect apparently balanced out the reduction in venous thromboembolism, as a prespecified analysis of net clinical benefit and harm did not show either one at day 10 or day 35, reported Dr. Alexander T. Cohen and his associates in MAGELLAN (Multicenter, Randomized, Parallel Group Efficacy and Safety Study for the Prevention of Venous Thromboembolism in Hospitalized Acutely Ill Medical Patients Comparing Rivaroxaban with Enoxaparin). The results were first presented by Dr. Cohen, of King's College Hospital, London, at the annual meeting of the American College of Cardiology in spring 2011.
In the 2.5-year study, 8,101 patients at 556 sites in 52 countries were randomized to receive rivaroxaban (4,050 subjects) or enoxaparin (4,051 subjects). Hundreds of patients in both study groups were excluded from the final analyses for various reasons, usually because they could not be assessed for venous thromboembolism using ultrasonography and other imaging techniques in a timely fashion, as required by the study protocol.
At 10 days' follow-up, 2.7% of patients in both groups had a primary outcome event - asymptomatic proximal deep vein thrombosis (DVT), symptomatic nonfatal pulmonary embolism (PE), or thromboembolism-related death - so rivaroxaban met the criteria for noninferiority to enoxaparin.
At the 35-day follow-up, 4.4% of subjects who received extended-duration rivaroxaban and 5.7% of those who received enoxaparin plus placebo had a primary outcome event, so rivaroxaban met the criteria for superiority to enoxaparin, the investigators reported (N. Engl. J. Med. 2013 Feb. 6 [doi: 10.1056/NEJMoa1111096]).
Rivaroxaban (Xarelto)has several indications: for stroke prevention in nonvalvular atrial fibrillation, for the treatment of DVT and PE, for reducing the risk of recurrence of DVT and PE, and for the prophylaxis of DVT in patients undergoing knee or hip replacement surgery. It is not indicated for the condition studied in MAGELLAN, namely prevention of VTE in acutely medically ill patients.
This study was funded by Bayer HealthCare and Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen and his associates reported numerous ties to industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: At 10-day follow-up, 2.7% of both study groups
had a thromboembolic event; at 35-day follow-up, 4.4% of subjects who received
rivaroxaban and 5.7% of those who received enoxaparin had a thromboembolic
event.
Data Source: A randomized, double-blind trial in 5,931 inpatients
with acute medical illness comparing rivaroxaban with enoxaparin for
thromboprophylaxis.
Disclosures: This study was funded by Bayer HealthCare and
Janssen, sponsors that also collected and analyzed the study data. Dr. Cohen
and his associates reported numerous ties to industry sources.
Ramipril boosts walking times in PAD with claudication
The ACE inhibitor ramipril substantially improves pain-free walking time and maximum walking time in patients who have peripheral artery disease with intermittent claudication, according to a report in the Feb. 6 issue of JAMA.
In what the investigators described as the first adequately powered randomized controlled trial to demonstrate ramipril’s benefit in this patient population, the magnitude of improvement in walking performance exceeded that reported for all other drug therapies.
The study subjects reported concomitant improvements in their ability to perform daily activities and their health-related quality of life, said Anna A. Ahimastos, Ph.D., of Baker IDI Heart and Diabetes Institute, Melbourne, and her associates.
The only two drugs currently approved in the United States for this indication, pentoxifylline and cilostazol, are reported to increase walking distance by 15% and 25%, respectively. In this trial, ramipril increased pain-free and maximum walking times by 77% and 123%.
Dr. Ahimastos and colleagues enrolled 212 patients with PAD and intermittent claudication in the double-blind trial, randomly assigning 106 to 6 months of ramipril (10 mg/d) and 106 to matching placebo. The mean age of the subjects was 65.5 years, and all were followed at three medical centers in Australia.
All the study subjects were receiving usual care according to their symptoms and risk profiles, which included antiplatelet and lipid-lowering agents. Roughly one-fourth of each group had diabetes, and the cohort included patients with aortoiliac as well as infrainguinal disease.
At 6-month follow-up, ramipril extended the pain-free walking time by a mean of 75 seconds and the maximum walking time by a mean of 255 seconds, compared with placebo. This corresponds to "a clinically significant increase in uphill walking distance of 184 meters," the investigators said (JAMA 2013;309:453-60).
The drug was particularly effective in patients who had femoropopliteal disease, increasing maximal walking time to 286 seconds, compared with patients who had aortoiliac disease, whose maximal walking time improved by 127 seconds.
These increases were independent of the small changes in blood pressure and ankle-brachial index associated with the drug. They exceed the improvements reported in the literature for other conventional PAD drug therapies including pentoxifylline, cilostazol, dipyridamole, ticlopidine, beraprost, iloprost, naftidrofuryl, and statins, Dr. Ahimastos and her associates said.
Patients in the ramipril group also showed improvements on the Walking Impairment Questionnaire in median walking distance, walking speed, and stair-climbing ability, compared with those in the placebo group. Similarly, the ramipril group showed improvement in the physical component of the Short Form–36, a measure of health-related quality of life, relative to the placebo group.
Ramipril did not significantly change any of the laboratory factors measured in the safety monitoring portion of the study. The only adverse events were transient dizziness, which affected 8.5% of the ramipril group and 2.8% of the placebo group, and persistent cough, which prompted seven patients in the ramipril group to withdraw from the study.
It is not yet known how ramipril improves functional capacity in PAD patients with intermittent claudication. Laboratory and animal studies suggest that the drug may increase peripheral blood flow and promote structural and functional changes in skeletal muscle, the researchers said.
This study was limited in that most of the subjects were whites of European descent, and the investigators had to select a population of patients with stable PAD "in whom we could ethically administer either a placebo or an ACE inhibitor" for 6 months. The cohort was thus "restricted to the lower end of the blood pressure spectrum and excluded patients with other major comorbid conditions," so the findings may not be generalizable to patients with higher blood pressure or to ethnically diverse populations, they added.
Anna A. Ahimastos, Ph.D.,
The ACE inhibitor ramipril substantially improves pain-free walking time and maximum walking time in patients who have peripheral artery disease with intermittent claudication, according to a report in the Feb. 6 issue of JAMA.
In what the investigators described as the first adequately powered randomized controlled trial to demonstrate ramipril’s benefit in this patient population, the magnitude of improvement in walking performance exceeded that reported for all other drug therapies.
The study subjects reported concomitant improvements in their ability to perform daily activities and their health-related quality of life, said Anna A. Ahimastos, Ph.D., of Baker IDI Heart and Diabetes Institute, Melbourne, and her associates.
The only two drugs currently approved in the United States for this indication, pentoxifylline and cilostazol, are reported to increase walking distance by 15% and 25%, respectively. In this trial, ramipril increased pain-free and maximum walking times by 77% and 123%.
Dr. Ahimastos and colleagues enrolled 212 patients with PAD and intermittent claudication in the double-blind trial, randomly assigning 106 to 6 months of ramipril (10 mg/d) and 106 to matching placebo. The mean age of the subjects was 65.5 years, and all were followed at three medical centers in Australia.
All the study subjects were receiving usual care according to their symptoms and risk profiles, which included antiplatelet and lipid-lowering agents. Roughly one-fourth of each group had diabetes, and the cohort included patients with aortoiliac as well as infrainguinal disease.
At 6-month follow-up, ramipril extended the pain-free walking time by a mean of 75 seconds and the maximum walking time by a mean of 255 seconds, compared with placebo. This corresponds to "a clinically significant increase in uphill walking distance of 184 meters," the investigators said (JAMA 2013;309:453-60).
The drug was particularly effective in patients who had femoropopliteal disease, increasing maximal walking time to 286 seconds, compared with patients who had aortoiliac disease, whose maximal walking time improved by 127 seconds.
These increases were independent of the small changes in blood pressure and ankle-brachial index associated with the drug. They exceed the improvements reported in the literature for other conventional PAD drug therapies including pentoxifylline, cilostazol, dipyridamole, ticlopidine, beraprost, iloprost, naftidrofuryl, and statins, Dr. Ahimastos and her associates said.
Patients in the ramipril group also showed improvements on the Walking Impairment Questionnaire in median walking distance, walking speed, and stair-climbing ability, compared with those in the placebo group. Similarly, the ramipril group showed improvement in the physical component of the Short Form–36, a measure of health-related quality of life, relative to the placebo group.
Ramipril did not significantly change any of the laboratory factors measured in the safety monitoring portion of the study. The only adverse events were transient dizziness, which affected 8.5% of the ramipril group and 2.8% of the placebo group, and persistent cough, which prompted seven patients in the ramipril group to withdraw from the study.
It is not yet known how ramipril improves functional capacity in PAD patients with intermittent claudication. Laboratory and animal studies suggest that the drug may increase peripheral blood flow and promote structural and functional changes in skeletal muscle, the researchers said.
This study was limited in that most of the subjects were whites of European descent, and the investigators had to select a population of patients with stable PAD "in whom we could ethically administer either a placebo or an ACE inhibitor" for 6 months. The cohort was thus "restricted to the lower end of the blood pressure spectrum and excluded patients with other major comorbid conditions," so the findings may not be generalizable to patients with higher blood pressure or to ethnically diverse populations, they added.
The ACE inhibitor ramipril substantially improves pain-free walking time and maximum walking time in patients who have peripheral artery disease with intermittent claudication, according to a report in the Feb. 6 issue of JAMA.
In what the investigators described as the first adequately powered randomized controlled trial to demonstrate ramipril’s benefit in this patient population, the magnitude of improvement in walking performance exceeded that reported for all other drug therapies.
The study subjects reported concomitant improvements in their ability to perform daily activities and their health-related quality of life, said Anna A. Ahimastos, Ph.D., of Baker IDI Heart and Diabetes Institute, Melbourne, and her associates.
The only two drugs currently approved in the United States for this indication, pentoxifylline and cilostazol, are reported to increase walking distance by 15% and 25%, respectively. In this trial, ramipril increased pain-free and maximum walking times by 77% and 123%.
Dr. Ahimastos and colleagues enrolled 212 patients with PAD and intermittent claudication in the double-blind trial, randomly assigning 106 to 6 months of ramipril (10 mg/d) and 106 to matching placebo. The mean age of the subjects was 65.5 years, and all were followed at three medical centers in Australia.
All the study subjects were receiving usual care according to their symptoms and risk profiles, which included antiplatelet and lipid-lowering agents. Roughly one-fourth of each group had diabetes, and the cohort included patients with aortoiliac as well as infrainguinal disease.
At 6-month follow-up, ramipril extended the pain-free walking time by a mean of 75 seconds and the maximum walking time by a mean of 255 seconds, compared with placebo. This corresponds to "a clinically significant increase in uphill walking distance of 184 meters," the investigators said (JAMA 2013;309:453-60).
The drug was particularly effective in patients who had femoropopliteal disease, increasing maximal walking time to 286 seconds, compared with patients who had aortoiliac disease, whose maximal walking time improved by 127 seconds.
These increases were independent of the small changes in blood pressure and ankle-brachial index associated with the drug. They exceed the improvements reported in the literature for other conventional PAD drug therapies including pentoxifylline, cilostazol, dipyridamole, ticlopidine, beraprost, iloprost, naftidrofuryl, and statins, Dr. Ahimastos and her associates said.
Patients in the ramipril group also showed improvements on the Walking Impairment Questionnaire in median walking distance, walking speed, and stair-climbing ability, compared with those in the placebo group. Similarly, the ramipril group showed improvement in the physical component of the Short Form–36, a measure of health-related quality of life, relative to the placebo group.
Ramipril did not significantly change any of the laboratory factors measured in the safety monitoring portion of the study. The only adverse events were transient dizziness, which affected 8.5% of the ramipril group and 2.8% of the placebo group, and persistent cough, which prompted seven patients in the ramipril group to withdraw from the study.
It is not yet known how ramipril improves functional capacity in PAD patients with intermittent claudication. Laboratory and animal studies suggest that the drug may increase peripheral blood flow and promote structural and functional changes in skeletal muscle, the researchers said.
This study was limited in that most of the subjects were whites of European descent, and the investigators had to select a population of patients with stable PAD "in whom we could ethically administer either a placebo or an ACE inhibitor" for 6 months. The cohort was thus "restricted to the lower end of the blood pressure spectrum and excluded patients with other major comorbid conditions," so the findings may not be generalizable to patients with higher blood pressure or to ethnically diverse populations, they added.
Anna A. Ahimastos, Ph.D.,
Anna A. Ahimastos, Ph.D.,
FROM JAMA
Major Finding: Ramipril extended pain-free walking time by 77% and maximal walking time by 123%, compared with placebo.
Data Source: A 6-month, double-blind, randomized controlled trial in 212 patients with PAD and intermittent claudication.
Disclosures: The study was supported by the Australian national and state agencies. Dr. Ahimastos’s associates reported ties to Sanofi-Aventis, maker of Ramace, and Novartis.
Calcium supplements raise CVD mortality only in men
Taking calcium supplements appears to raise the risk of death from cardiovascular disease among men but not women, according to a report published online Feb. 4 in JAMA Internal Medicine.
In contrast, the intake of dietary calcium has no apparent effect on cardiovascular disease (CVD) mortality in either sex, said Qian Xiao, Ph.D., of the division of cancer epidemiology and genetics, National Cancer Institute, and her associates.
These findings from a large prospective study that followed 388,229 adults for 12 years are not definitive but provide further support for the controversial idea that supplemental calcium may have adverse effects on cardiovascular health. However, calcium also "is widely involved in many aspects of human physiology" and is known to benefit some aspects of cardiovascular health, such as lowering blood pressure and improving lipid profiles, so further study is warranted.
"Given the extensive use of calcium supplements in the population, it is of great importance to assess the effect of supplemental calcium use beyond bone health," Dr. Xiao and her colleagues wrote.
The investigators examined this issue using data from the National Institutes of Health–AARP Diet and Health Study, which assessed adults who were aged 50-71 years in 1995 and resided in California, Florida, Louisiana, New Jersey, North Carolina, Pennsylvania, Atlanta, and Detroit. For this study, the investigators followed 219,059 men and 169,170 women participants, correlating their calcium intake at baseline with cumulative mortality from heart disease, cerebrovascular disease, and other forms of CVD.
A total of 23% of the men and 56% of the women took calcium supplements, and 56% of the men and 58% of the women took multivitamins containing calcium. During follow-up there were 7,904 CVD deaths in men and 3,874 in women.
The use of calcium supplements correlated with a significantly elevated risk of total CVD mortality, heart disease mortality, and cerebrovascular disease mortality in men. In addition, men taking the highest quintile of calcium supplementation showed an increased risk of CVD death, which was mainly driven by heart disease death, Dr. Xiao and her associates said (JAMA Intern. Med. 2013 [doi:10.1001/jamainternmed.2013.3283]).
In women, null associations were seen between calcium supplements and all of these outcomes.
Dietary calcium showed no relationship with any mortality outcome in men or women.
These findings remained robust in a sensitivity analysis that excluded data on all deaths that occurred during the first 2 years of follow-up.
In analyses that categorized subjects by age, smoking status, BMI, hypertension, hypercholesterolemia, magnesium intake, and alcohol consumption, the positive association between calcium supplements and increased mortality persisted in most of these subgroups among men. It remained null among women except in three notable subgroups: In women who were former smokers, who did not have hypertension, and who had hypercholesterolemia, calcium supplements were linked to an increase in CVD mortality.
One possible mechanism by which calcium supplements could exert an adverse effect on CVD is the deposition of calcium phosphate in cardiovascular structures, in the form of vascular calcification. Increased blood coagulation and arterial stiffness also have been linked to high serum calcium levels, the investigators noted.
The significant interaction by sex was an "intriguing" finding that also deserves further investigation. "It may be reasonable to assume that, on average, male users started taking calcium supplements at an older age. Therefore women were more likely to have achieved calcium balance and stable calcium levels long before the study, and the effect of calcium supplements became less profound," the investigators wrote.
This study was supported by the National Institutes of Health, the National Cancer Institute, and the National Institute on Aging. No financial conflicts of interest were reported.
This large, prospective, well-conducted observational study adds to the mounting evidence that calcium supplements may elevate CVD mortality, said Susanna C. Larsson, Ph.D.
"A safe alternative to calcium supplements is to consume calcium-rich foods, such as low-fat dairy foods, beans, and green leafy vegetables, which contain not only calcium but also a cocktail of essential minerals and vitamins," she wrote. This would have additional health benefits, such as improving glycemic control in patients with diabetes, Dr. Larsson added.
Dr. Larsson is in the division of nutritional epidemiology, Institute of Environmental Medicine, at Karolinska Institutet, Stockholm. She reported no financial conflicts of interest. These remarks were taken from her invited commentary accompanying Dr. Xiao’s report (JAMA Intern. Med. 2013 [doi:10.1001/jamainternmed.2013.3769]).
This large, prospective, well-conducted observational study adds to the mounting evidence that calcium supplements may elevate CVD mortality, said Susanna C. Larsson, Ph.D.
"A safe alternative to calcium supplements is to consume calcium-rich foods, such as low-fat dairy foods, beans, and green leafy vegetables, which contain not only calcium but also a cocktail of essential minerals and vitamins," she wrote. This would have additional health benefits, such as improving glycemic control in patients with diabetes, Dr. Larsson added.
Dr. Larsson is in the division of nutritional epidemiology, Institute of Environmental Medicine, at Karolinska Institutet, Stockholm. She reported no financial conflicts of interest. These remarks were taken from her invited commentary accompanying Dr. Xiao’s report (JAMA Intern. Med. 2013 [doi:10.1001/jamainternmed.2013.3769]).
This large, prospective, well-conducted observational study adds to the mounting evidence that calcium supplements may elevate CVD mortality, said Susanna C. Larsson, Ph.D.
"A safe alternative to calcium supplements is to consume calcium-rich foods, such as low-fat dairy foods, beans, and green leafy vegetables, which contain not only calcium but also a cocktail of essential minerals and vitamins," she wrote. This would have additional health benefits, such as improving glycemic control in patients with diabetes, Dr. Larsson added.
Dr. Larsson is in the division of nutritional epidemiology, Institute of Environmental Medicine, at Karolinska Institutet, Stockholm. She reported no financial conflicts of interest. These remarks were taken from her invited commentary accompanying Dr. Xiao’s report (JAMA Intern. Med. 2013 [doi:10.1001/jamainternmed.2013.3769]).
Taking calcium supplements appears to raise the risk of death from cardiovascular disease among men but not women, according to a report published online Feb. 4 in JAMA Internal Medicine.
In contrast, the intake of dietary calcium has no apparent effect on cardiovascular disease (CVD) mortality in either sex, said Qian Xiao, Ph.D., of the division of cancer epidemiology and genetics, National Cancer Institute, and her associates.
These findings from a large prospective study that followed 388,229 adults for 12 years are not definitive but provide further support for the controversial idea that supplemental calcium may have adverse effects on cardiovascular health. However, calcium also "is widely involved in many aspects of human physiology" and is known to benefit some aspects of cardiovascular health, such as lowering blood pressure and improving lipid profiles, so further study is warranted.
"Given the extensive use of calcium supplements in the population, it is of great importance to assess the effect of supplemental calcium use beyond bone health," Dr. Xiao and her colleagues wrote.
The investigators examined this issue using data from the National Institutes of Health–AARP Diet and Health Study, which assessed adults who were aged 50-71 years in 1995 and resided in California, Florida, Louisiana, New Jersey, North Carolina, Pennsylvania, Atlanta, and Detroit. For this study, the investigators followed 219,059 men and 169,170 women participants, correlating their calcium intake at baseline with cumulative mortality from heart disease, cerebrovascular disease, and other forms of CVD.
A total of 23% of the men and 56% of the women took calcium supplements, and 56% of the men and 58% of the women took multivitamins containing calcium. During follow-up there were 7,904 CVD deaths in men and 3,874 in women.
The use of calcium supplements correlated with a significantly elevated risk of total CVD mortality, heart disease mortality, and cerebrovascular disease mortality in men. In addition, men taking the highest quintile of calcium supplementation showed an increased risk of CVD death, which was mainly driven by heart disease death, Dr. Xiao and her associates said (JAMA Intern. Med. 2013 [doi:10.1001/jamainternmed.2013.3283]).
In women, null associations were seen between calcium supplements and all of these outcomes.
Dietary calcium showed no relationship with any mortality outcome in men or women.
These findings remained robust in a sensitivity analysis that excluded data on all deaths that occurred during the first 2 years of follow-up.
In analyses that categorized subjects by age, smoking status, BMI, hypertension, hypercholesterolemia, magnesium intake, and alcohol consumption, the positive association between calcium supplements and increased mortality persisted in most of these subgroups among men. It remained null among women except in three notable subgroups: In women who were former smokers, who did not have hypertension, and who had hypercholesterolemia, calcium supplements were linked to an increase in CVD mortality.
One possible mechanism by which calcium supplements could exert an adverse effect on CVD is the deposition of calcium phosphate in cardiovascular structures, in the form of vascular calcification. Increased blood coagulation and arterial stiffness also have been linked to high serum calcium levels, the investigators noted.
The significant interaction by sex was an "intriguing" finding that also deserves further investigation. "It may be reasonable to assume that, on average, male users started taking calcium supplements at an older age. Therefore women were more likely to have achieved calcium balance and stable calcium levels long before the study, and the effect of calcium supplements became less profound," the investigators wrote.
This study was supported by the National Institutes of Health, the National Cancer Institute, and the National Institute on Aging. No financial conflicts of interest were reported.
Taking calcium supplements appears to raise the risk of death from cardiovascular disease among men but not women, according to a report published online Feb. 4 in JAMA Internal Medicine.
In contrast, the intake of dietary calcium has no apparent effect on cardiovascular disease (CVD) mortality in either sex, said Qian Xiao, Ph.D., of the division of cancer epidemiology and genetics, National Cancer Institute, and her associates.
These findings from a large prospective study that followed 388,229 adults for 12 years are not definitive but provide further support for the controversial idea that supplemental calcium may have adverse effects on cardiovascular health. However, calcium also "is widely involved in many aspects of human physiology" and is known to benefit some aspects of cardiovascular health, such as lowering blood pressure and improving lipid profiles, so further study is warranted.
"Given the extensive use of calcium supplements in the population, it is of great importance to assess the effect of supplemental calcium use beyond bone health," Dr. Xiao and her colleagues wrote.
The investigators examined this issue using data from the National Institutes of Health–AARP Diet and Health Study, which assessed adults who were aged 50-71 years in 1995 and resided in California, Florida, Louisiana, New Jersey, North Carolina, Pennsylvania, Atlanta, and Detroit. For this study, the investigators followed 219,059 men and 169,170 women participants, correlating their calcium intake at baseline with cumulative mortality from heart disease, cerebrovascular disease, and other forms of CVD.
A total of 23% of the men and 56% of the women took calcium supplements, and 56% of the men and 58% of the women took multivitamins containing calcium. During follow-up there were 7,904 CVD deaths in men and 3,874 in women.
The use of calcium supplements correlated with a significantly elevated risk of total CVD mortality, heart disease mortality, and cerebrovascular disease mortality in men. In addition, men taking the highest quintile of calcium supplementation showed an increased risk of CVD death, which was mainly driven by heart disease death, Dr. Xiao and her associates said (JAMA Intern. Med. 2013 [doi:10.1001/jamainternmed.2013.3283]).
In women, null associations were seen between calcium supplements and all of these outcomes.
Dietary calcium showed no relationship with any mortality outcome in men or women.
These findings remained robust in a sensitivity analysis that excluded data on all deaths that occurred during the first 2 years of follow-up.
In analyses that categorized subjects by age, smoking status, BMI, hypertension, hypercholesterolemia, magnesium intake, and alcohol consumption, the positive association between calcium supplements and increased mortality persisted in most of these subgroups among men. It remained null among women except in three notable subgroups: In women who were former smokers, who did not have hypertension, and who had hypercholesterolemia, calcium supplements were linked to an increase in CVD mortality.
One possible mechanism by which calcium supplements could exert an adverse effect on CVD is the deposition of calcium phosphate in cardiovascular structures, in the form of vascular calcification. Increased blood coagulation and arterial stiffness also have been linked to high serum calcium levels, the investigators noted.
The significant interaction by sex was an "intriguing" finding that also deserves further investigation. "It may be reasonable to assume that, on average, male users started taking calcium supplements at an older age. Therefore women were more likely to have achieved calcium balance and stable calcium levels long before the study, and the effect of calcium supplements became less profound," the investigators wrote.
This study was supported by the National Institutes of Health, the National Cancer Institute, and the National Institute on Aging. No financial conflicts of interest were reported.
From JAMA Internal Medicine
Major Finding: The use of calcium supplements correlated with a significantly elevated risk of total CVD mortality, heart disease mortality, and cerebrovascular disease mortality in men, but the association was null in women.
Data Source: A large, prospective follow-up study of CVD mortality in 388,229 men and women aged 50-71 years at baseline.
Disclosures: This study was supported by the National Institutes of Health, the National Cancer Institute, and the National Institute on Aging. No financial conflicts of interest were reported.
Prostatectomy vs. radiotherapy: 15-year functional outcomes are same
Fifteen years after treatment for localized prostate cancer, men who underwent radical prostatectomy reported no differences in urinary, bowel, or sexual function compared with men who underwent external-beam radiation, according to a report published online Jan. 30 in the New England Journal of Medicine.
Using data from the Prostate Cancer Outcomes Study (PCOS), a population-based cohort of men across the United States diagnosed as having prostate cancer in the mid-1990s, researchers tracked the long-term disease-specific outcomes of 1,655 men who underwent radical prostatectomy (70.3% of patients) or radiotherapy (29.7%), with or without androgen-deprivation therapy.
Source: American Gastroenterological Association (www.youtube.com/AmerGastroAssn)
"Since the median life expectancy after treatment for prostate cancer is 13.8 years, a careful evaluation of long-term functional outcomes is critical to an understanding of the comprehensive experience of men living with a diagnosis of prostate cancer," said Dr. Matthew J. Resnick of the department of urologic surgery and the Center for Surgical Quality and Outcomes Research, Vanderbilt University, Nashville, Tenn., and his associates.
Even though men who had radical prostatectomy were more likely to be bothered by such difficulties as urinary incontinence, bowel urgency, and erectile dysfunction at 2 years and 5 years after treatment, those differences disappeared by the 15-year mark. At that time, the prevalence of ED was "nearly universal," affecting 87% of men in the prostatectomy group and 94% of those in the radiotherapy group; yet only 43% and 38%, respectively, reported that this bothered them.
"Long-term follow-up reveals consistent functional declines after 5 years. It remains unknown whether this continued decline is due to prostate cancer and its treatment, the normal aging process, or a combination of factors," the investigators said (N. Engl. J. Med. 2013 [doi10.1056/NEJMoa1209978]).
This study was funded by the National Cancer Institute. Dr. Resnick was supported by the VA National Quality Scholars Program and the T. J. Martell Foundation. Dr. Resnick reported ties to Dendreon and Bayer Healthcare, and his associates reported ties to Ferring and Johnson & Johnson.
Fifteen years after treatment for localized prostate cancer, men who underwent radical prostatectomy reported no differences in urinary, bowel, or sexual function compared with men who underwent external-beam radiation, according to a report published online Jan. 30 in the New England Journal of Medicine.
Using data from the Prostate Cancer Outcomes Study (PCOS), a population-based cohort of men across the United States diagnosed as having prostate cancer in the mid-1990s, researchers tracked the long-term disease-specific outcomes of 1,655 men who underwent radical prostatectomy (70.3% of patients) or radiotherapy (29.7%), with or without androgen-deprivation therapy.
Source: American Gastroenterological Association (www.youtube.com/AmerGastroAssn)
"Since the median life expectancy after treatment for prostate cancer is 13.8 years, a careful evaluation of long-term functional outcomes is critical to an understanding of the comprehensive experience of men living with a diagnosis of prostate cancer," said Dr. Matthew J. Resnick of the department of urologic surgery and the Center for Surgical Quality and Outcomes Research, Vanderbilt University, Nashville, Tenn., and his associates.
Even though men who had radical prostatectomy were more likely to be bothered by such difficulties as urinary incontinence, bowel urgency, and erectile dysfunction at 2 years and 5 years after treatment, those differences disappeared by the 15-year mark. At that time, the prevalence of ED was "nearly universal," affecting 87% of men in the prostatectomy group and 94% of those in the radiotherapy group; yet only 43% and 38%, respectively, reported that this bothered them.
"Long-term follow-up reveals consistent functional declines after 5 years. It remains unknown whether this continued decline is due to prostate cancer and its treatment, the normal aging process, or a combination of factors," the investigators said (N. Engl. J. Med. 2013 [doi10.1056/NEJMoa1209978]).
This study was funded by the National Cancer Institute. Dr. Resnick was supported by the VA National Quality Scholars Program and the T. J. Martell Foundation. Dr. Resnick reported ties to Dendreon and Bayer Healthcare, and his associates reported ties to Ferring and Johnson & Johnson.
Fifteen years after treatment for localized prostate cancer, men who underwent radical prostatectomy reported no differences in urinary, bowel, or sexual function compared with men who underwent external-beam radiation, according to a report published online Jan. 30 in the New England Journal of Medicine.
Using data from the Prostate Cancer Outcomes Study (PCOS), a population-based cohort of men across the United States diagnosed as having prostate cancer in the mid-1990s, researchers tracked the long-term disease-specific outcomes of 1,655 men who underwent radical prostatectomy (70.3% of patients) or radiotherapy (29.7%), with or without androgen-deprivation therapy.
Source: American Gastroenterological Association (www.youtube.com/AmerGastroAssn)
"Since the median life expectancy after treatment for prostate cancer is 13.8 years, a careful evaluation of long-term functional outcomes is critical to an understanding of the comprehensive experience of men living with a diagnosis of prostate cancer," said Dr. Matthew J. Resnick of the department of urologic surgery and the Center for Surgical Quality and Outcomes Research, Vanderbilt University, Nashville, Tenn., and his associates.
Even though men who had radical prostatectomy were more likely to be bothered by such difficulties as urinary incontinence, bowel urgency, and erectile dysfunction at 2 years and 5 years after treatment, those differences disappeared by the 15-year mark. At that time, the prevalence of ED was "nearly universal," affecting 87% of men in the prostatectomy group and 94% of those in the radiotherapy group; yet only 43% and 38%, respectively, reported that this bothered them.
"Long-term follow-up reveals consistent functional declines after 5 years. It remains unknown whether this continued decline is due to prostate cancer and its treatment, the normal aging process, or a combination of factors," the investigators said (N. Engl. J. Med. 2013 [doi10.1056/NEJMoa1209978]).
This study was funded by the National Cancer Institute. Dr. Resnick was supported by the VA National Quality Scholars Program and the T. J. Martell Foundation. Dr. Resnick reported ties to Dendreon and Bayer Healthcare, and his associates reported ties to Ferring and Johnson & Johnson.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: At 15-year follow-up, there were no significant differences in urinary, bowel, or sexual function between 1,164 men who had undergone radical prostatectomy and 491 who had undergone radiotherapy for localized prostate cancer.
Data Source: Long-term follow-up of 1,655 men participating in the Prostate Cancer Outcomes Study, a population-based prospective study of men in six states who were diagnosed as having localized prostate cancer in the mid-1990s.
Disclosures: This study was funded by the National Cancer Institute. Dr. Resnick was supported by the VA National Quality Scholars Program and the T. J. Martell Foundation. Dr. Resnick reported ties to Dendreon and Bayer Healthcare, and his associates reported ties to Ferring and Johnson & Johnson.
Cardiac disease doubles cognitive impairment risk in women
Among women aged 70-89 years, a history of cardiac disease was strongly associated with an increased risk of developing nonamnestic mild cognitive impairment in a large population-based study published online Jan. 28 in JAMA Neurology.
This association was not found among elderly men who participated in the study, said Dr. Rosebud O. Roberts of the department of Health Sciences Research and the division of epidemiology, Mayo Clinic, Rochester, Minn., and her associates.
A link between cardiovascular disease and mild cognitive impairment (MCI) and dementia has been reported in several previous studies, and association is stronger among women than men, the investigators said.
Dr. Roberts and her colleagues analyzed data from the Mayo Clinic Study of Aging, which assessed risk factors for MCI in residents of Olmsted County, Minn., who were aged 70-89 years at baseline in 2004. These study subjects underwent physical examinations and extensive cognitive testing every 15 months, and at each follow-up visit, they were diagnosed as having normal cognition, MCI, or dementia by a team of clinicians including a physician, a nurse, and a neuropsychologist.
For this study, Dr. Roberts and her associates assessed a random sample of 2,719 participants who were followed for a median of 4 years.
Cardiac disease was defined as a diagnosis of atrial fibrillation, coronary heart disease, or congestive heart failure. The physical and neuropsychological evaluations included assessment using the Clinical Dementia Rating Scale, Functional Activities Questionnaire, Short Test of Mental Status, and nine tests examining memory, executive function, language, and visuospatial skills.
A total of 1,450 of these subjects had at least one follow-up assessment; 348 of them developed incident MCI and 18 developed incident dementia. About 66% of those with MCI had amnestic MCI, 27% had nonamnestic MCI, and the remaining 7% had MCI of unknown subtype because they were followed up by telephone interview only.
In the study cohort as a whole, the incidence of MCI was higher among subjects with cardiac disease (73.2/1,000 person-years) than in those without it (62.1/1,000 person-years).
When the data were analyzed by type of MCI, cardiac disease was found to have no correlation with amnestic MCI but was linked to a doubling of the rate of nonamnestic MCI. Subjects with cardiac disease had a rate of nonamnestic MCI of 23.4/1,000 person-years, compared with 12.2/1,000 person-years for subjects without cardiac disease.
When the data were analyzed by gender, cardiac disease was found to have no correlation with nonamnestic MCI among men: The incidence of this form of cognitive impairment was similar between men with cardiac disease (22.3/1,000 person-years) and men without cardiac disease (18.9/1,000 person-years).
However, the rate of nonamnestic MCI was significantly higher among women with cardiac disease (24.3/1,000 person-years) than in those without (7.4/1,000 person-years), the investigators said (JAMA Neurol. 2013 Jan. 28 [doi:10.1001/jamaneurol.2013.607]).
This association in women remained robust in further analyses that adjusted for a history of stroke, age, education, and APOE genotype status.
For women with atrial fibrillation, the risk of nonamnestic MCI had a hazard ratio of 2.25. That risk had an HR of 2.05 for women with coronary artery disease and an HR of 2.82 for women with heart failure.
These findings suggest that cardiac disease is an independent risk factor for nonamnestic MCI in elderly women and support the hypothesis that such MCI has a vascular etiology, Dr. Roberts and her associates said.
Several possible mechanisms may contribute to the association between cardiac disease and nonamnestic MCI. Cardiac sympathetic nerve dysfunction and obstructive sleep apnea both have been linked to cardiac disease and could contribute to dementia as well. And cardiac disease could precipitate cerebral disease through the release of microemboli that travel to the brain or through hypoperfusion of the brain from impaired cardiac function, they added.
This study was limited in that the population of Olmsted County is predominantly of northern European extraction, so the findings may not be applicable to other ethnic groups.
Among women aged 70-89 years, a history of cardiac disease was strongly associated with an increased risk of developing nonamnestic mild cognitive impairment in a large population-based study published online Jan. 28 in JAMA Neurology.
This association was not found among elderly men who participated in the study, said Dr. Rosebud O. Roberts of the department of Health Sciences Research and the division of epidemiology, Mayo Clinic, Rochester, Minn., and her associates.
A link between cardiovascular disease and mild cognitive impairment (MCI) and dementia has been reported in several previous studies, and association is stronger among women than men, the investigators said.
Dr. Roberts and her colleagues analyzed data from the Mayo Clinic Study of Aging, which assessed risk factors for MCI in residents of Olmsted County, Minn., who were aged 70-89 years at baseline in 2004. These study subjects underwent physical examinations and extensive cognitive testing every 15 months, and at each follow-up visit, they were diagnosed as having normal cognition, MCI, or dementia by a team of clinicians including a physician, a nurse, and a neuropsychologist.
For this study, Dr. Roberts and her associates assessed a random sample of 2,719 participants who were followed for a median of 4 years.
Cardiac disease was defined as a diagnosis of atrial fibrillation, coronary heart disease, or congestive heart failure. The physical and neuropsychological evaluations included assessment using the Clinical Dementia Rating Scale, Functional Activities Questionnaire, Short Test of Mental Status, and nine tests examining memory, executive function, language, and visuospatial skills.
A total of 1,450 of these subjects had at least one follow-up assessment; 348 of them developed incident MCI and 18 developed incident dementia. About 66% of those with MCI had amnestic MCI, 27% had nonamnestic MCI, and the remaining 7% had MCI of unknown subtype because they were followed up by telephone interview only.
In the study cohort as a whole, the incidence of MCI was higher among subjects with cardiac disease (73.2/1,000 person-years) than in those without it (62.1/1,000 person-years).
When the data were analyzed by type of MCI, cardiac disease was found to have no correlation with amnestic MCI but was linked to a doubling of the rate of nonamnestic MCI. Subjects with cardiac disease had a rate of nonamnestic MCI of 23.4/1,000 person-years, compared with 12.2/1,000 person-years for subjects without cardiac disease.
When the data were analyzed by gender, cardiac disease was found to have no correlation with nonamnestic MCI among men: The incidence of this form of cognitive impairment was similar between men with cardiac disease (22.3/1,000 person-years) and men without cardiac disease (18.9/1,000 person-years).
However, the rate of nonamnestic MCI was significantly higher among women with cardiac disease (24.3/1,000 person-years) than in those without (7.4/1,000 person-years), the investigators said (JAMA Neurol. 2013 Jan. 28 [doi:10.1001/jamaneurol.2013.607]).
This association in women remained robust in further analyses that adjusted for a history of stroke, age, education, and APOE genotype status.
For women with atrial fibrillation, the risk of nonamnestic MCI had a hazard ratio of 2.25. That risk had an HR of 2.05 for women with coronary artery disease and an HR of 2.82 for women with heart failure.
These findings suggest that cardiac disease is an independent risk factor for nonamnestic MCI in elderly women and support the hypothesis that such MCI has a vascular etiology, Dr. Roberts and her associates said.
Several possible mechanisms may contribute to the association between cardiac disease and nonamnestic MCI. Cardiac sympathetic nerve dysfunction and obstructive sleep apnea both have been linked to cardiac disease and could contribute to dementia as well. And cardiac disease could precipitate cerebral disease through the release of microemboli that travel to the brain or through hypoperfusion of the brain from impaired cardiac function, they added.
This study was limited in that the population of Olmsted County is predominantly of northern European extraction, so the findings may not be applicable to other ethnic groups.
Among women aged 70-89 years, a history of cardiac disease was strongly associated with an increased risk of developing nonamnestic mild cognitive impairment in a large population-based study published online Jan. 28 in JAMA Neurology.
This association was not found among elderly men who participated in the study, said Dr. Rosebud O. Roberts of the department of Health Sciences Research and the division of epidemiology, Mayo Clinic, Rochester, Minn., and her associates.
A link between cardiovascular disease and mild cognitive impairment (MCI) and dementia has been reported in several previous studies, and association is stronger among women than men, the investigators said.
Dr. Roberts and her colleagues analyzed data from the Mayo Clinic Study of Aging, which assessed risk factors for MCI in residents of Olmsted County, Minn., who were aged 70-89 years at baseline in 2004. These study subjects underwent physical examinations and extensive cognitive testing every 15 months, and at each follow-up visit, they were diagnosed as having normal cognition, MCI, or dementia by a team of clinicians including a physician, a nurse, and a neuropsychologist.
For this study, Dr. Roberts and her associates assessed a random sample of 2,719 participants who were followed for a median of 4 years.
Cardiac disease was defined as a diagnosis of atrial fibrillation, coronary heart disease, or congestive heart failure. The physical and neuropsychological evaluations included assessment using the Clinical Dementia Rating Scale, Functional Activities Questionnaire, Short Test of Mental Status, and nine tests examining memory, executive function, language, and visuospatial skills.
A total of 1,450 of these subjects had at least one follow-up assessment; 348 of them developed incident MCI and 18 developed incident dementia. About 66% of those with MCI had amnestic MCI, 27% had nonamnestic MCI, and the remaining 7% had MCI of unknown subtype because they were followed up by telephone interview only.
In the study cohort as a whole, the incidence of MCI was higher among subjects with cardiac disease (73.2/1,000 person-years) than in those without it (62.1/1,000 person-years).
When the data were analyzed by type of MCI, cardiac disease was found to have no correlation with amnestic MCI but was linked to a doubling of the rate of nonamnestic MCI. Subjects with cardiac disease had a rate of nonamnestic MCI of 23.4/1,000 person-years, compared with 12.2/1,000 person-years for subjects without cardiac disease.
When the data were analyzed by gender, cardiac disease was found to have no correlation with nonamnestic MCI among men: The incidence of this form of cognitive impairment was similar between men with cardiac disease (22.3/1,000 person-years) and men without cardiac disease (18.9/1,000 person-years).
However, the rate of nonamnestic MCI was significantly higher among women with cardiac disease (24.3/1,000 person-years) than in those without (7.4/1,000 person-years), the investigators said (JAMA Neurol. 2013 Jan. 28 [doi:10.1001/jamaneurol.2013.607]).
This association in women remained robust in further analyses that adjusted for a history of stroke, age, education, and APOE genotype status.
For women with atrial fibrillation, the risk of nonamnestic MCI had a hazard ratio of 2.25. That risk had an HR of 2.05 for women with coronary artery disease and an HR of 2.82 for women with heart failure.
These findings suggest that cardiac disease is an independent risk factor for nonamnestic MCI in elderly women and support the hypothesis that such MCI has a vascular etiology, Dr. Roberts and her associates said.
Several possible mechanisms may contribute to the association between cardiac disease and nonamnestic MCI. Cardiac sympathetic nerve dysfunction and obstructive sleep apnea both have been linked to cardiac disease and could contribute to dementia as well. And cardiac disease could precipitate cerebral disease through the release of microemboli that travel to the brain or through hypoperfusion of the brain from impaired cardiac function, they added.
This study was limited in that the population of Olmsted County is predominantly of northern European extraction, so the findings may not be applicable to other ethnic groups.
FROM JAMA NEUROLOGY
Major Finding: The incidence of nonamnestic MCI was significantly higher among women with cardiac disease (24.3/1,000 person-years) than in women without cardiac disease (7.4/1,000 person-years).
Data Source: A prospective, population-based cohort study involving 1,450 men and women aged 70-89 years who were followed for the development of cognitive impairment every 15 months for a median of 4 years.
Disclosures: This study was supported by the National Institutes of Health, the Robert H. and Clarice Smith and Abigail van Buren Alzheimer's Disease Research Program, and the National Institute on Aging. Dr. Roberts and her associates reported ties to Lilly, Elan, Pfizer, Janssen Alzheimer Immunotherapy, GE Healthcare, Novartis, Abbott, and Baxter.
High-frequency oscillatory ventilation may worsen ARDS
High-frequency oscillatory ventilation doesn’t improve and may actually worsen moderate to severe acute respiratory distress syndrome in adults, compared with standard ventilation, according to two reports published online Jan. 23 in the New England Journal of Medicine.
In two large, separate, randomized controlled trials comparing the two ventilation strategies, 1-month mortality in critically ill adults with ARDS who received high-frequency oscillatory ventilation (HFOV) was either higher or not significantly different from that in patients who received standard low tidal volume and high positive end–expiratory pressure ventilation.
In one study, which was terminated early because of the large discrepancy in short-term mortality, HFOV also was associated with higher mean airway pressures and significantly greater need for sedatives, neuromuscular blockers, and vasoactive drugs.
Both trials call into question the current widespread use of HFOV early in the course of ARDS when patients don’t show an adequate response to conventional mechanical ventilation, the two research groups noted.
HFOV, which delivers very small tidal volumes at very high rates, is thought to minimize the lung damage done by ventilation’s repeated forced opening and collapsing of lung structures. Many clinicians now use it earlier in the course of ARDS, even though there are other approaches for improving oxygenation, based solely on the results of animal studies and small trials that used outdated ventilation methods as a control. The commercial availability of HFOV equipment has accelerated this trend.
In the absence of good evidence of HFOV’s effectiveness, experts in Canada and the United Kingdom called for rigorous randomized controlled trials.
Dr. Niall D. Ferguson and his associates in the Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) trial compared the two strategies in patients aged 16-85 years who had moderate to severe ARDS and were treated at 39 intensive care units in Canada, Saudi Arabia, the United States, Chile, and India.
The OSCILLATE steering committee terminated the trial early, after only 548 subjects had been randomized, because three consecutive interim analyses showed that 1-month mortality was consistently higher with HFOV. "The effect size was sufficiently large that we concluded that even if early HFOV did not increase mortality, it would be very unlikely to decrease mortality," wrote Dr. Ferguson of the division of critical care medicine and the departments of medicine and physiology, University of Toronto, and his colleagues.
At that time, 1-month mortality was 47% for HFOV (129 of 275 patients), compared with 35% (96 of 273 patients) in the control group (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215554]).
This result remained robust in three further analyses of the data that controlled for numerous variables. It also persisted in several subgroup analyses, regardless of the subjects’ baseline severity of hypoxemia, the subjects’ BMI, the use or nonuse of vasopressors, or the level of experience with ventilation at each medical center.
In addition, the use of vasopressors, which had been equivalent between the two study groups at baseline at approximately 63%, increased in the HFOV group to 73% within 4 hours of beginning the procedure and to 78% the next day. In contrast, vasopressors were used in only 62% and 58%, respectively, in the control group.
The use of neuromuscular blockers followed the same pattern, increasing in the HFOV group from 27% at baseline to 46% at 4 hours and 46% at 24 hours but remaining more steady in the control group at 29%, 31%, and 26%, respectively, they reported.
Cumulatively, vasoactive agents were used in 91% and neuromuscular blockers in 83% of the HFOV group, compared with 84% and 68%, respectively, in the control group. Vasoactive agents were required for an average of 2 days longer in the HFOV group, and neuromuscular blockers were required for an average of 1 day longer, compared with the control group.
Sedatives and opioids (primarily midazolam and fentanyl) were given for the same duration to the two study groups, but doses were higher in the HFOV group.
"Our results raise serious concerns about the early use of HFOV for the management of ARDS in adults. The results of this study increase the uncertainty about possible benefits of HFOV even when applied in patients with life-threatening refractory hypoxemia," Dr. Ferguson and his associates said.
In the Oscillation in ARDS (OSCAR) study, commissioned by the U.K.’s National Institute for Health Research, two ventilation techniques were compared in 795 patients in general ICUs at 12 university hospitals, 4 university-affiliated hospitals, and 13 district general hospitals in England, Wales, and Scotland.
As this was a pragmatic study meant to reflect real-world practice, HFOV was compared against whatever conventional ventilation was used in local practice, and the ICUs were "encouraged" to use the recommended combinations of positive end-expiratory pressure and fraction of inspired oxygen values. Consequently, management of the control group varied greatly from one ICU to the next, according to the researchers, led by Dr. Duncan Young of John Radcliffe Hospital and the University of Oxford, both in Oxford.
The primary outcome, 30-day all-cause mortality, occurred in 41.7% of the HFOV group (166 of 398 patients) and in 41.1% of the control group (163 of 397 patients), a nonsignificant difference. These rates remained largely unchanged in further analyses that adjusted for several variables, Dr. Young and his colleagues said (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215716]).
The duration of ICU stay was 17.6 days with HFOV and 16.1 days with conventional ventilation, and the duration of hospital stay was 33.1 days and 33.9 days, respectively.
"Since data collection is ongoing, we cannot yet report the longer-term outcomes (including survival and health-related quality of life)," they added.
In conclusion, "we were unable to find any benefit or harm from the use of HFOV in adult patients with ARDS. We recommend that this mode of ventilation not be used for routine care," Dr. Young and his associates said.
The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson’s associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
CareFusion provided the SensorMedics HFO ventilator and technical support for the OSCILLATE trial but had no role in study design, data collection or analysis, or manuscript preparation. Inspiration Healthcare supplied the ventilators for the OSCAR study but had no role in the study design, data acquisition or analysis, or manuscript preparation.
Both the OSCILLATE and the OSCAR trial raise concerns about the widespread and routine use of HFOV, said Dr. Atul Malhotra and Dr. Jeffrey M. Drazen.
Dr. Jeffrey M. Drazen |
"However, one could argue that it is not HFOV itself but the HFOV protocols studied in these trials that were ineffective, and perhaps worse, than usual care," they wrote. Questions remain concerning this method’s usefulness after more aggressive volume resuscitation, less aggressive use of sedation and paralysis, and selection of patients who have homgeneous, recruitable lung.
"Perhaps patients with ARDS will require individualized therapy that takes into consideration their body habitus, the cause of their disease, and the mechanisms leading to lung injury ... but for now clinicians should be cautious about applying HFOV routinely in patients with ARDS," they said.
Dr. Malhotra and Dr. Drazen are with the divisions of sleep medicine and pulmonary and critical care medicine at Brigham and Women’s Hospital, Boston. Dr. Malhotra reported ties to Philips Respironics, Pfizer, Apnex, and Apnicure. These remarks were taken from their editorial accompanying the two reports (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMe1300103]).
Both the OSCILLATE and the OSCAR trial raise concerns about the widespread and routine use of HFOV, said Dr. Atul Malhotra and Dr. Jeffrey M. Drazen.
Dr. Jeffrey M. Drazen |
"However, one could argue that it is not HFOV itself but the HFOV protocols studied in these trials that were ineffective, and perhaps worse, than usual care," they wrote. Questions remain concerning this method’s usefulness after more aggressive volume resuscitation, less aggressive use of sedation and paralysis, and selection of patients who have homgeneous, recruitable lung.
"Perhaps patients with ARDS will require individualized therapy that takes into consideration their body habitus, the cause of their disease, and the mechanisms leading to lung injury ... but for now clinicians should be cautious about applying HFOV routinely in patients with ARDS," they said.
Dr. Malhotra and Dr. Drazen are with the divisions of sleep medicine and pulmonary and critical care medicine at Brigham and Women’s Hospital, Boston. Dr. Malhotra reported ties to Philips Respironics, Pfizer, Apnex, and Apnicure. These remarks were taken from their editorial accompanying the two reports (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMe1300103]).
Both the OSCILLATE and the OSCAR trial raise concerns about the widespread and routine use of HFOV, said Dr. Atul Malhotra and Dr. Jeffrey M. Drazen.
Dr. Jeffrey M. Drazen |
"However, one could argue that it is not HFOV itself but the HFOV protocols studied in these trials that were ineffective, and perhaps worse, than usual care," they wrote. Questions remain concerning this method’s usefulness after more aggressive volume resuscitation, less aggressive use of sedation and paralysis, and selection of patients who have homgeneous, recruitable lung.
"Perhaps patients with ARDS will require individualized therapy that takes into consideration their body habitus, the cause of their disease, and the mechanisms leading to lung injury ... but for now clinicians should be cautious about applying HFOV routinely in patients with ARDS," they said.
Dr. Malhotra and Dr. Drazen are with the divisions of sleep medicine and pulmonary and critical care medicine at Brigham and Women’s Hospital, Boston. Dr. Malhotra reported ties to Philips Respironics, Pfizer, Apnex, and Apnicure. These remarks were taken from their editorial accompanying the two reports (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMe1300103]).
High-frequency oscillatory ventilation doesn’t improve and may actually worsen moderate to severe acute respiratory distress syndrome in adults, compared with standard ventilation, according to two reports published online Jan. 23 in the New England Journal of Medicine.
In two large, separate, randomized controlled trials comparing the two ventilation strategies, 1-month mortality in critically ill adults with ARDS who received high-frequency oscillatory ventilation (HFOV) was either higher or not significantly different from that in patients who received standard low tidal volume and high positive end–expiratory pressure ventilation.
In one study, which was terminated early because of the large discrepancy in short-term mortality, HFOV also was associated with higher mean airway pressures and significantly greater need for sedatives, neuromuscular blockers, and vasoactive drugs.
Both trials call into question the current widespread use of HFOV early in the course of ARDS when patients don’t show an adequate response to conventional mechanical ventilation, the two research groups noted.
HFOV, which delivers very small tidal volumes at very high rates, is thought to minimize the lung damage done by ventilation’s repeated forced opening and collapsing of lung structures. Many clinicians now use it earlier in the course of ARDS, even though there are other approaches for improving oxygenation, based solely on the results of animal studies and small trials that used outdated ventilation methods as a control. The commercial availability of HFOV equipment has accelerated this trend.
In the absence of good evidence of HFOV’s effectiveness, experts in Canada and the United Kingdom called for rigorous randomized controlled trials.
Dr. Niall D. Ferguson and his associates in the Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) trial compared the two strategies in patients aged 16-85 years who had moderate to severe ARDS and were treated at 39 intensive care units in Canada, Saudi Arabia, the United States, Chile, and India.
The OSCILLATE steering committee terminated the trial early, after only 548 subjects had been randomized, because three consecutive interim analyses showed that 1-month mortality was consistently higher with HFOV. "The effect size was sufficiently large that we concluded that even if early HFOV did not increase mortality, it would be very unlikely to decrease mortality," wrote Dr. Ferguson of the division of critical care medicine and the departments of medicine and physiology, University of Toronto, and his colleagues.
At that time, 1-month mortality was 47% for HFOV (129 of 275 patients), compared with 35% (96 of 273 patients) in the control group (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215554]).
This result remained robust in three further analyses of the data that controlled for numerous variables. It also persisted in several subgroup analyses, regardless of the subjects’ baseline severity of hypoxemia, the subjects’ BMI, the use or nonuse of vasopressors, or the level of experience with ventilation at each medical center.
In addition, the use of vasopressors, which had been equivalent between the two study groups at baseline at approximately 63%, increased in the HFOV group to 73% within 4 hours of beginning the procedure and to 78% the next day. In contrast, vasopressors were used in only 62% and 58%, respectively, in the control group.
The use of neuromuscular blockers followed the same pattern, increasing in the HFOV group from 27% at baseline to 46% at 4 hours and 46% at 24 hours but remaining more steady in the control group at 29%, 31%, and 26%, respectively, they reported.
Cumulatively, vasoactive agents were used in 91% and neuromuscular blockers in 83% of the HFOV group, compared with 84% and 68%, respectively, in the control group. Vasoactive agents were required for an average of 2 days longer in the HFOV group, and neuromuscular blockers were required for an average of 1 day longer, compared with the control group.
Sedatives and opioids (primarily midazolam and fentanyl) were given for the same duration to the two study groups, but doses were higher in the HFOV group.
"Our results raise serious concerns about the early use of HFOV for the management of ARDS in adults. The results of this study increase the uncertainty about possible benefits of HFOV even when applied in patients with life-threatening refractory hypoxemia," Dr. Ferguson and his associates said.
In the Oscillation in ARDS (OSCAR) study, commissioned by the U.K.’s National Institute for Health Research, two ventilation techniques were compared in 795 patients in general ICUs at 12 university hospitals, 4 university-affiliated hospitals, and 13 district general hospitals in England, Wales, and Scotland.
As this was a pragmatic study meant to reflect real-world practice, HFOV was compared against whatever conventional ventilation was used in local practice, and the ICUs were "encouraged" to use the recommended combinations of positive end-expiratory pressure and fraction of inspired oxygen values. Consequently, management of the control group varied greatly from one ICU to the next, according to the researchers, led by Dr. Duncan Young of John Radcliffe Hospital and the University of Oxford, both in Oxford.
The primary outcome, 30-day all-cause mortality, occurred in 41.7% of the HFOV group (166 of 398 patients) and in 41.1% of the control group (163 of 397 patients), a nonsignificant difference. These rates remained largely unchanged in further analyses that adjusted for several variables, Dr. Young and his colleagues said (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215716]).
The duration of ICU stay was 17.6 days with HFOV and 16.1 days with conventional ventilation, and the duration of hospital stay was 33.1 days and 33.9 days, respectively.
"Since data collection is ongoing, we cannot yet report the longer-term outcomes (including survival and health-related quality of life)," they added.
In conclusion, "we were unable to find any benefit or harm from the use of HFOV in adult patients with ARDS. We recommend that this mode of ventilation not be used for routine care," Dr. Young and his associates said.
The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson’s associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
CareFusion provided the SensorMedics HFO ventilator and technical support for the OSCILLATE trial but had no role in study design, data collection or analysis, or manuscript preparation. Inspiration Healthcare supplied the ventilators for the OSCAR study but had no role in the study design, data acquisition or analysis, or manuscript preparation.
High-frequency oscillatory ventilation doesn’t improve and may actually worsen moderate to severe acute respiratory distress syndrome in adults, compared with standard ventilation, according to two reports published online Jan. 23 in the New England Journal of Medicine.
In two large, separate, randomized controlled trials comparing the two ventilation strategies, 1-month mortality in critically ill adults with ARDS who received high-frequency oscillatory ventilation (HFOV) was either higher or not significantly different from that in patients who received standard low tidal volume and high positive end–expiratory pressure ventilation.
In one study, which was terminated early because of the large discrepancy in short-term mortality, HFOV also was associated with higher mean airway pressures and significantly greater need for sedatives, neuromuscular blockers, and vasoactive drugs.
Both trials call into question the current widespread use of HFOV early in the course of ARDS when patients don’t show an adequate response to conventional mechanical ventilation, the two research groups noted.
HFOV, which delivers very small tidal volumes at very high rates, is thought to minimize the lung damage done by ventilation’s repeated forced opening and collapsing of lung structures. Many clinicians now use it earlier in the course of ARDS, even though there are other approaches for improving oxygenation, based solely on the results of animal studies and small trials that used outdated ventilation methods as a control. The commercial availability of HFOV equipment has accelerated this trend.
In the absence of good evidence of HFOV’s effectiveness, experts in Canada and the United Kingdom called for rigorous randomized controlled trials.
Dr. Niall D. Ferguson and his associates in the Oscillation for Acute Respiratory Distress Syndrome Treated Early (OSCILLATE) trial compared the two strategies in patients aged 16-85 years who had moderate to severe ARDS and were treated at 39 intensive care units in Canada, Saudi Arabia, the United States, Chile, and India.
The OSCILLATE steering committee terminated the trial early, after only 548 subjects had been randomized, because three consecutive interim analyses showed that 1-month mortality was consistently higher with HFOV. "The effect size was sufficiently large that we concluded that even if early HFOV did not increase mortality, it would be very unlikely to decrease mortality," wrote Dr. Ferguson of the division of critical care medicine and the departments of medicine and physiology, University of Toronto, and his colleagues.
At that time, 1-month mortality was 47% for HFOV (129 of 275 patients), compared with 35% (96 of 273 patients) in the control group (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215554]).
This result remained robust in three further analyses of the data that controlled for numerous variables. It also persisted in several subgroup analyses, regardless of the subjects’ baseline severity of hypoxemia, the subjects’ BMI, the use or nonuse of vasopressors, or the level of experience with ventilation at each medical center.
In addition, the use of vasopressors, which had been equivalent between the two study groups at baseline at approximately 63%, increased in the HFOV group to 73% within 4 hours of beginning the procedure and to 78% the next day. In contrast, vasopressors were used in only 62% and 58%, respectively, in the control group.
The use of neuromuscular blockers followed the same pattern, increasing in the HFOV group from 27% at baseline to 46% at 4 hours and 46% at 24 hours but remaining more steady in the control group at 29%, 31%, and 26%, respectively, they reported.
Cumulatively, vasoactive agents were used in 91% and neuromuscular blockers in 83% of the HFOV group, compared with 84% and 68%, respectively, in the control group. Vasoactive agents were required for an average of 2 days longer in the HFOV group, and neuromuscular blockers were required for an average of 1 day longer, compared with the control group.
Sedatives and opioids (primarily midazolam and fentanyl) were given for the same duration to the two study groups, but doses were higher in the HFOV group.
"Our results raise serious concerns about the early use of HFOV for the management of ARDS in adults. The results of this study increase the uncertainty about possible benefits of HFOV even when applied in patients with life-threatening refractory hypoxemia," Dr. Ferguson and his associates said.
In the Oscillation in ARDS (OSCAR) study, commissioned by the U.K.’s National Institute for Health Research, two ventilation techniques were compared in 795 patients in general ICUs at 12 university hospitals, 4 university-affiliated hospitals, and 13 district general hospitals in England, Wales, and Scotland.
As this was a pragmatic study meant to reflect real-world practice, HFOV was compared against whatever conventional ventilation was used in local practice, and the ICUs were "encouraged" to use the recommended combinations of positive end-expiratory pressure and fraction of inspired oxygen values. Consequently, management of the control group varied greatly from one ICU to the next, according to the researchers, led by Dr. Duncan Young of John Radcliffe Hospital and the University of Oxford, both in Oxford.
The primary outcome, 30-day all-cause mortality, occurred in 41.7% of the HFOV group (166 of 398 patients) and in 41.1% of the control group (163 of 397 patients), a nonsignificant difference. These rates remained largely unchanged in further analyses that adjusted for several variables, Dr. Young and his colleagues said (N. Engl. J. Med. 2013 Jan. 23 [doi:10.1056/NEJMoa1215716]).
The duration of ICU stay was 17.6 days with HFOV and 16.1 days with conventional ventilation, and the duration of hospital stay was 33.1 days and 33.9 days, respectively.
"Since data collection is ongoing, we cannot yet report the longer-term outcomes (including survival and health-related quality of life)," they added.
In conclusion, "we were unable to find any benefit or harm from the use of HFOV in adult patients with ARDS. We recommend that this mode of ventilation not be used for routine care," Dr. Young and his associates said.
The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson’s associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.
CareFusion provided the SensorMedics HFO ventilator and technical support for the OSCILLATE trial but had no role in study design, data collection or analysis, or manuscript preparation. Inspiration Healthcare supplied the ventilators for the OSCAR study but had no role in the study design, data acquisition or analysis, or manuscript preparation.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: 1-month mortality was significantly higher with HFOV (47%) than with control ventilation (35%) in the OSCILLATE study; no significant difference was seen between groups in the OSCAR study (41.7% vs 41.1%).
Data Source: Two randomized controlled trials comparing HFOV against either low tidal volume, high positive end–expiratory pressure ventilation or conventional ventilation among 548 ARDS patients in Canada, Saudi Arabia, the United States, Chile, and India and among 795 ARDS patients in England, Wales, and Scotland.
Disclosures: The OSCILLATE study was supported by the Canadian Institutes of Health Research, the King Abdullah International Medical Research Center, and Fonds de Recherche de Quebec. Dr. Ferguson's associates reported ties to numerous industry sources. The OSCAR study was supported by the National Institute for Health Research Health Technology Assessment Programme. Dr. Young and his associates reported no relevant financial conflicts of interest.