User login
A novel proprietary James Del Rosso, MD, reported at MedscapeLive’s annual Las Vegas Dermatology Seminar, held virtually this year.
Sol-Gel Technologies, the Israeli company developing the fixed-dose cream, called Twyneo, has applied to the Food and Drug Administration for marketing approval.
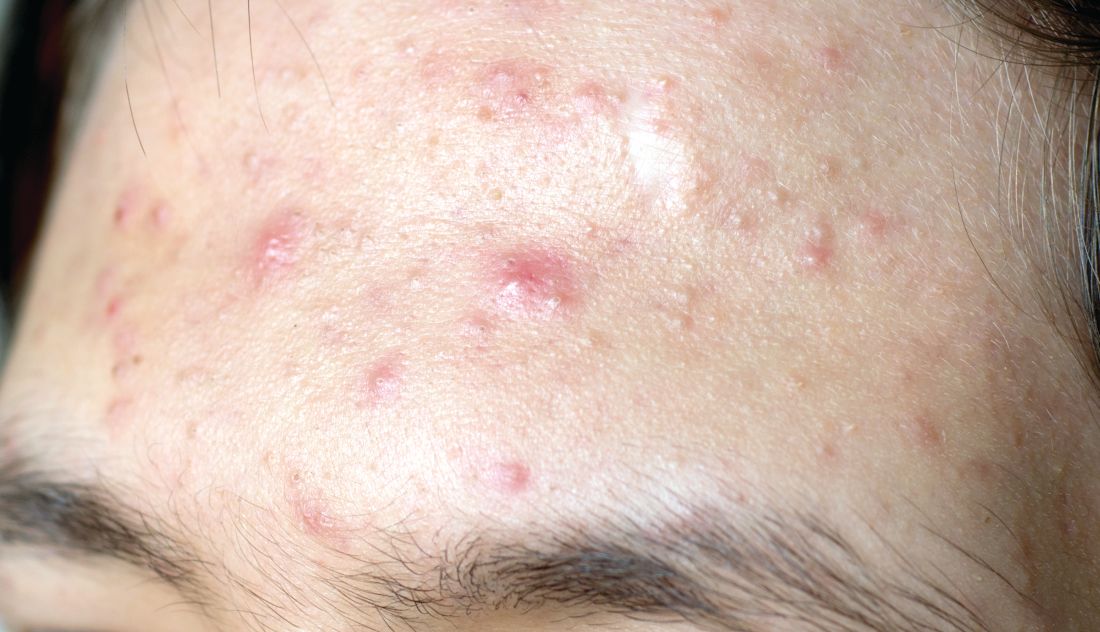
The product combines two workhorse topical agents for the treatment of acne, which are ordinarily incompatible, since benzoyl peroxide degrades tretinoin and reduces its effectiveness. The company’s silica-based microencapsulation technology overcomes that obstacle, explained Dr. Del Rosso, a dermatologist at JDR Research in Las Vegas.
The two identical phase 3, randomized, double-blind, vehicle-controlled clinical trials included a total of 858 patients ages 9 years and older with moderate to severe acne enrolled at 63 U.S. sites. Participants were randomized 2:1 to once-daily application of Twyneo or its vehicle cream for 12 weeks.
In one trial, the coprimary endpoint of at least a two-grade reduction and clear or almost clear skin at week 12 on a 5-point Investigator Global Assessment (IGA) scale was achieved in 38.5% of patients on Twyneo and 11.5% of controls. In the other trial, the IGA success rates were 25.4% and 14.7%. In both trials, the between-group difference was statistically significant.
The other coprimary endpoints were the absolute change from baseline in inflammatory and noninflammatory lesion counts. Inflammatory lesions were reduced by 21.6% and 16.2% in the active treatment arms of the two trials, compared with 14.8% and 14.1% reductions in the control groups. Noninflammatory lesion counts fell by 29.7% and 24.2% in patients on active treatment, versus 19.8% and 17.4% reductions in controls. The between-group differences were statistically significant.
Skin tolerability of Twyneo was “very good” and similar to vehicle, according to Dr. Del Rosso.
He reported receiving research funding from Sol-Gel, the studies’ sponsor.
MedscapeLive and this news organization are owned by the same parent company.
bjancin@mdedge.com
A novel proprietary James Del Rosso, MD, reported at MedscapeLive’s annual Las Vegas Dermatology Seminar, held virtually this year.
Sol-Gel Technologies, the Israeli company developing the fixed-dose cream, called Twyneo, has applied to the Food and Drug Administration for marketing approval.
The product combines two workhorse topical agents for the treatment of acne, which are ordinarily incompatible, since benzoyl peroxide degrades tretinoin and reduces its effectiveness. The company’s silica-based microencapsulation technology overcomes that obstacle, explained Dr. Del Rosso, a dermatologist at JDR Research in Las Vegas.
The two identical phase 3, randomized, double-blind, vehicle-controlled clinical trials included a total of 858 patients ages 9 years and older with moderate to severe acne enrolled at 63 U.S. sites. Participants were randomized 2:1 to once-daily application of Twyneo or its vehicle cream for 12 weeks.
In one trial, the coprimary endpoint of at least a two-grade reduction and clear or almost clear skin at week 12 on a 5-point Investigator Global Assessment (IGA) scale was achieved in 38.5% of patients on Twyneo and 11.5% of controls. In the other trial, the IGA success rates were 25.4% and 14.7%. In both trials, the between-group difference was statistically significant.
The other coprimary endpoints were the absolute change from baseline in inflammatory and noninflammatory lesion counts. Inflammatory lesions were reduced by 21.6% and 16.2% in the active treatment arms of the two trials, compared with 14.8% and 14.1% reductions in the control groups. Noninflammatory lesion counts fell by 29.7% and 24.2% in patients on active treatment, versus 19.8% and 17.4% reductions in controls. The between-group differences were statistically significant.
Skin tolerability of Twyneo was “very good” and similar to vehicle, according to Dr. Del Rosso.
He reported receiving research funding from Sol-Gel, the studies’ sponsor.
MedscapeLive and this news organization are owned by the same parent company.
bjancin@mdedge.com
A novel proprietary James Del Rosso, MD, reported at MedscapeLive’s annual Las Vegas Dermatology Seminar, held virtually this year.
Sol-Gel Technologies, the Israeli company developing the fixed-dose cream, called Twyneo, has applied to the Food and Drug Administration for marketing approval.
The product combines two workhorse topical agents for the treatment of acne, which are ordinarily incompatible, since benzoyl peroxide degrades tretinoin and reduces its effectiveness. The company’s silica-based microencapsulation technology overcomes that obstacle, explained Dr. Del Rosso, a dermatologist at JDR Research in Las Vegas.
The two identical phase 3, randomized, double-blind, vehicle-controlled clinical trials included a total of 858 patients ages 9 years and older with moderate to severe acne enrolled at 63 U.S. sites. Participants were randomized 2:1 to once-daily application of Twyneo or its vehicle cream for 12 weeks.
In one trial, the coprimary endpoint of at least a two-grade reduction and clear or almost clear skin at week 12 on a 5-point Investigator Global Assessment (IGA) scale was achieved in 38.5% of patients on Twyneo and 11.5% of controls. In the other trial, the IGA success rates were 25.4% and 14.7%. In both trials, the between-group difference was statistically significant.
The other coprimary endpoints were the absolute change from baseline in inflammatory and noninflammatory lesion counts. Inflammatory lesions were reduced by 21.6% and 16.2% in the active treatment arms of the two trials, compared with 14.8% and 14.1% reductions in the control groups. Noninflammatory lesion counts fell by 29.7% and 24.2% in patients on active treatment, versus 19.8% and 17.4% reductions in controls. The between-group differences were statistically significant.
Skin tolerability of Twyneo was “very good” and similar to vehicle, according to Dr. Del Rosso.
He reported receiving research funding from Sol-Gel, the studies’ sponsor.
MedscapeLive and this news organization are owned by the same parent company.
bjancin@mdedge.com
FROM MEDSCAPELIVE LAS VEGAS DERMATOLOGY SEMINAR