User login
Noninvasive positive pressure ventilation (NIPPV)—delivered via a tight-fitting mask rather than via an endotracheal tube or tracheostomy—is one of the most important advances in the management of acute respiratory failure to emerge in the past 2 decades. It is now recommended as the first choice for ventilatory support in selected patients, such as those with exacerbations of chronic obstructive pulmonary disease (COPD) or with cardiogenic pulmonary edema.1–3 In fact, some authors suggest that using NIPPV in more than 20% of COPD patients is a characteristic of respiratory care departments that are “avid for change”4—change being a good thing.
However, NIPPV has not been universally accepted, with wide variations in its utilization. In a 2006 survey, it was being used in only 33% of patients with COPD or congestive heart failure, for which it might be indicated. 5 Some potential reasons for the low rate are that physicians do not know about it, respiratory therapists are not sufficiently trained in it, and hospitals lack the equipment to do it.5
Our goal in this review is to familiarize the reader with how NIPPV has evolved and with its indications and contraindications in specific acute care conditions.
FROM A VACUUM CLEANER TO THE INTENSIVE CARE UNIT
NIPPV appears to have been first tried in 1870 by Chaussier, who used a bag and face mask to resuscitate neonates.6
In 1936, Poulton and Oxon7 described their “pulmonary plus pressure machine,” which used a vacuum cleaner blower and a mask to increase the alveolar pressure and thus counteract the increased intrapulmonary pressure in patients with heart failure, pulmonary edema, Cheyne-Stokes breathing, and asthma.
In the 1940s, intermittent positive pressure breathing devices were developed for use in high-altitude aviation. Motley, Werko, and Cournand8,9 subsequently used these devices to treat acute respiratory failure in pneumonia, pulmonary edema, near-drowning, Guillain-Barré syndrome, and acute severe asthma.
Although NIPPV was shown to be effective for acute conditions, invasive ventilation became preferred, particularly as blood gas analysis and ventilator technologies simultaneously matured, spurred at least in part by the polio epidemics of the 1950s.10
NIPPV reemerged in the 1980s for use in chronic conditions. First, continuous positive airway pressure (CPAP) came into use for obstructive sleep apnea,11 followed by noninvasive positive-pressure volume ventilation in neuromuscular diseases.12 Bilevel positive pressure devices (ie, with separate inspiratory and expiratory pressures) soon followed, again initially for obstructive sleep apnea13 and then for diverse neuromuscular diseases.14
NIPPV is now a mainstream therapy for diverse conditions in acute and chronic care.3 One reason we now use it in acute conditions is to avoid the complications associated with intubation.
Some clinicians initially resisted using NIPPV, concerned that it demanded too much of the nurses’ time15 and was costly.16 However, in a 1997 study in patients with COPD and acute respiratory failure, Nava et al17 found that NIPPV was no more expensive and no more demanding of staff resources than invasive mechanical ventilation in the first 48 hours of ventilation. Further, after the first few days of ventilation, NIPPV put fewer time demands on physicians and nurses than did invasive mechanical ventilation.
THREE MODES: CPAP, PRESSURE-LIMITED, VOLUME-LIMITED
The term “noninvasive ventilation” generally encompasses various forms of positive pressure ventilation. However, negative pressure ventilation, in the form of diaphragm pacing, may regain a foothold in the devices used for respiratory support.18 We therefore favor the term “NIPPV” in this review.
NIPPV IN ACUTE RESPIRATORY FAILURE
The main reasons to use NIPPV instead of invasive ventilation in acute care are to avoid the complications of invasive ventilation, to improve outcomes (eg, reduce mortality rates, decrease hospital length of stay), and to decrease the cost of care.
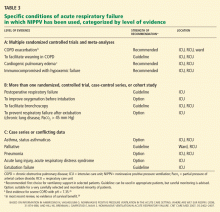
NIPPV is the standard of care for acute exacerbations of COPD
In a meta-analysis of eight randomized controlled trials,24 the specific advantages of NIPPV compared with usual care in acute exacerbations of COPD included:
- A lower risk of treatment failure, defined as death, need for intubation, or inability to tolerate the treatment (relative risk [RR] 0.51, number needed to treat [NNT] to prevent one treatment failure = 5)
- A lower risk of intubation (RR 0.43, NNT = 5)
- A lower mortality rate (RR 0.41, NNT = 8)
- A lower risk of complications (RR 0.32, NNT = 3)
- A shorter hospital length of stay (by about 3 days).
Mechanisms by which NIPPV may impart these benefits include reducing the work of breathing, unloading the respiratory muscles, lessening diaphragmatic pressure swings, reducing the respiratory rate, eliminating diaphragmatic work, and counteracting the threshold loading effects of auto-positive end-expiratory pressure (auto-PEEP).24–26
Also, if a patient with COPD is intubated, NIPPV seems to help after the tube is removed, preventing postextubation respiratory failure and facilitating weaning from invasive ventilation.27 These topics are discussed below.
A Cochrane systematic review24 concluded that NIPPV should be tried early in the course of respiratory failure, before severe acidosis develops. The patients in the studies in this review all had partial pressure of arterial carbon dioxide (Paco2) levels greater than 45 mm Hg.
In patients with severe respiratory acidosis (pH < 7.25), NIPPV failure rates are greater than 50%. However, trying NIPPV may still be justified, even in the presence of hypercapnic encephalopathy, as long as no other indications for invasive support and facilities for prompt endotracheal intubation are available. 1
However, in another systematic review,26 in patients with mild COPD exacerbations (pH > 7.35), NIPPV was no more effective than standard medical therapy in preventing acute respiratory failure, preventing death, or reducing length of hospitalization. Moreover, nearly 50% of the patients could not tolerate NIPPV.
Rapid improvement in cardiogenic pulmonary edema, but possibly no lower mortality rate
The Three Interventions in Cardiogenic Pulmonary Oedema (3CPO) trial,28 with 1,156 patients, was the largest randomized trial to compare NIPPV and standard oxygen therapy for acute pulmonary edema. It found that NIPPV (either CPAP or noninvasive intermittent positive pressure ventilation) was significantly better than standard oxygen therapy (through a variable-delivery oxygen mask with a reservoir) in the first hour of treatment in terms of the dyspnea score, heart rate, acidosis, and hypercapnia. However, there were no significant differences between groups in the 7- or 30-day mortality rates, the rates of intubation, rates of admission to the critical care unit, or in the mean length of hospital stay.
In contrast, several smaller randomized trials and meta-analyses showed lower intubation and mortality rates with NIPPV.29,30 Factors that may account for those differences include a much lower intubation rate in the 3CPO trial (2.9% overall, compared with 20% with conventional therapy in other trials), a higher mortality rate in the 3CPO trial, and methodologic differences (eg, patients for whom standard therapy failed in the 3CPO trial received rescue NIPPV).
If NIPPV is beneficial in cardiogenic pulmonary edema, the mechanisms are probably its favorable hemodynamic effects and its positive end-expiratory pressure (PEEP) effect on flooded alveoli. Specifically, positive intrathoracic pressure can be expected to reduce both preload and afterload, with improvement in the cardiac index and reduced work of breathing. 31,32
Notwithstanding the possible lack of impact of NIPPV on death or intubation rates in this setting, the intervention rapidly improves dyspnea and respiratory and metabolic abnormalities and should be considered for treatment of cardiogenic pulmonary edema associated with severe respiratory distress. A subgroup in which the NIPPV may reduce intubation rates is those with hypercapnia.33 A concern that NIPPV may increase the rate of myocardial infarction34 was not confirmed in the 3CPO trial.28 Interestingly, there were no differences in outcomes between CPAP and noninvasive intermittent positive pressure ventilation in this setting.28,34,35
Immunocompromised patients with acute respiratory failure
A particular challenge of NIPPV in immunocompromised patients, particularly compared with its use in COPD exacerbation or cardiogenic pulmonary edema, is that the underlying pathophysiology of respiratory dysfunction in immunocompromised patients may not be readily reversible. Therefore, its application in this group may need to follow clearly defined indications.
In one trial,20 inclusion criteria were:
- Immune suppression (due to neutropenia after chemotherapy or bone marrow transplantation, immunosuppressive drugs for organ transplantation, corticosteroids, cytotoxic therapy for nonmalignant conditions, or the acquired immunodeficiency syndrome)
- Persistent pulmonary infiltrates
- Fever (temperature > 38.3°C; 100.9°F)
- A respiratory rate greater than 30 breaths per minute
- Severe dyspnea at rest
- Early hypoxemic acute respiratory failure, defined as a ratio of the partial pressure of arterial oxygen to the fraction of inspired oxygen (Pao2/Fio2 ratio) less than 200 while on oxygen.
Compared with patients who received conventional treatment, fewer of those randomized to additional intermittent noninvasive ventilation had to be intubated (46% vs 77%, P = .03), suffered serious complications (50% vs 81%, P = .02), or died in the intensive care unit (38% vs 69%, P = .03) or in the hospital (50% vs 81%, P = .02).
Similarly, in a randomized trial in 40 patients with acute respiratory failure after solid organ transplantation, more patients in the NIPPV group than in the control group had an improvement in the Pao2/Fio2 ratio within the first hour (70% vs 25%, P = .004) or a sustained improvement in the Pao2/Fio2 ratio (60% vs 25%, P = .03); fewer of them needed endotracheal intubation (20% vs 70%, P = .002); fewer of them died of complications (20% vs 50%, P = .05); they had a shorter length of stay in the intensive care unit (mean 5.5 vs 9 days, P = .03); and fewer of them died in the intensive care unit (20% vs 50%, P = .05). There was, however, no difference in the overall hospital mortality rate.36
MAY NOT HELP AFTER EXTUBATION, EXCEPT IN SPECIFIC CASES
NIPPV has been used to treat respiratory failure after extubation,22,37 to prevent acute respiratory failure after failure of weaning,38–41 and to support breathing in patients who failed a trial of spontaneous breathing.42–45
Unfortunately, the evidence for using NIPPV in respiratory failure after extubation, including unplanned extubation, appears to be unfavorable, except possibly in patients with chronic pulmonary disease (particularly COPD and possibly obesity) and hypercapnia. An international consensus report stated that NIPPV should be considered in patients with hypercapnic respiratory insufficiency, especially those with COPD, to shorten the duration of intubation, but that it should not be routinely used in extubation respiratory failure.46
Treatment of respiratory failure after extubation
Two recent randomized controlled trials compared NIPPV and standard care in patients who met the criteria for readiness for extubation but who developed respiratory failure after mechanical ventilation was discontinued. 22,37 Those two studies showed a longer time to reintubation for patients randomized to NIPPV but no differences in the rate of reintubation between the two groups and no difference in the lengths of stay in the intensive care unit.
Of greater concern, one study showed a higher rate of death in the intensive care unit in the NIPPV group than in the standard therapy group (25% vs 14%, respectively).22 This finding suggests that NIPPV delayed necessary reintubation in patients developing respiratory failure after extubation, with a consequent risk of fatal complications.
Prevention of respiratory failure after extubation
Other studies used NIPPV to prevent respiratory failure after extubation rather than wait to apply it after respiratory failure developed.38–41
Nava et al,40 in a trial in patients successfully weaned but considered to be at risk of reintubation, found that fewer of those randomized to NIPPV had to be reintubated than those who received standard care (8% vs 24%), and 10% fewer of them died in the intensive care unit. Risk factors for reintubation (and therefore eligibility criteria for this trial) included a Paco2 higher than 45 mm Hg, more than one consecutive failure of weaning, chronic heart failure, other comorbidity, weak cough, or stridor.
Extubated patients are a heterogeneous group, so if some subgroups benefit from a transition to NIPPV after extubation, it will be important to identify them. For instance, a subgroup analysis of a study by Ferrer et al38 indicated the survival benefit of NIPPV after extubation was limited to patients with chronic respiratory disorders and hypercapnia during a trial of spontaneous breathing.
In a subsequent successful test of this hypothesis, a randomized trial showed that the early use of noninvasive ventilation in patients with hypercapnia after a trial of spontaneous breathing and with chronic respiratory disorders (COPD, chronic bronchitis, bronchiectasis, obesity-hypoventilation, sequelae of tuberculosis, chest wall deformity, or chronic persistent asthma) reduced the risk of respiratory failure after extubation and the risk of death within the first 90 days.39
Others in which this approach may be helpful are obese patients who have high Paco2 levels. Compared with historical controls, 62 patients with a body mass index greater than 35 kg/m2 who received NIPPV in the 48 hours after extubation had a lower rate of respiratory failure, shorter lengths of stay in the intensive care unit and hospital, and, in the subgroup with hypercapnia, a lower hospital mortality rate.41
NIPPV to facilitate weaning
In several studies, mechanically ventilated patients who had failed a trial of spontaneous breathing were randomized to undergo either accelerated weaning, extubation, and NIPPV or conventional weaning with pressure support via mechanical ventilation.42–46 Most patients developed hypercapnia during the spontaneous breathing trials, and most of the patients had COPD.
A meta-analysis47 of the randomized trials of this approach concluded that, compared with continued invasive ventilation, NIPPV decreased the risk of death (relative risk 0.41) and of ventilator-associated pneumonia (relative risk 0.28) and reduced the total duration of mechanical ventilation by a weighted mean difference of 7.33 days. The benefits appeared to be most significant in patients with COPD.
NIPPV IN ASTHMA AND STATUS ASTHMATICUS
Noninvasive ventilation is an attractive alternative to intubation for patients with status asthmaticus, given the challenges and conflicting demands of maintaining ventilation despite severe airway obstruction.
In a 1996 prospective study of 17 episodes of asthma associated with acute respiratory failure, Meduri et al48 showed that NIPPV could progressively improve the pH and the Paco2 over 12 to 24 hours and reduce the respiratory rate.
In a subsequent controlled trial, Soroksky et al49 randomized 30 patients presenting to an emergency room with a severe asthma attack to NIPPV with conventional therapy vs conventional therapy only. The study group had a significantly greater increase in the forced expiratory volume in 1 second compared with the control group (54% vs 29%, respectively) and a lower hospitalization rate (18% vs 63%).
Another randomized trial of NIPPV, in patients with status asthmaticus presenting to an emergency room, was prematurely terminated due to a physician treatment bias that favored NIPPV.50 The preliminary results of that study showed a 7.3% higher intubation rate in the control group than in the NIPPV group, along with trends toward a lower intubation rate, a shorter length of hospital stay, and lower hospital charges in the NIPPV group.
Despite these initial favorable results, a Cochrane review concluded that the use of NIPPV in patients with status asthmaticus is controversial.51 NIPPV can be tried in selected patients such as those with mild to moderate respiratory distress (respiratory rate greater than 25 breaths per minute, use of accessory muscles to breathe, difficulty speaking), an arterial pH of 7.25 to 7.35, and a Paco2 of 45 to 55 mm Hg.52 Patients with impending respiratory failure or the inability to protect the airway should probably not be considered for NIPPV.52
IN ACUTE LUNG INJURY AND ACUTE RESPIRATORY DISTRESS SYNDROME
The most challenging application of NIPPV may be in patients with acute lung injury and the acute respiratory distress syndrome.
Initial trials of NIPPV in this setting have been disappointing, and a meta-analysis of the topic concluded that NIPPV was unlikely to have any significant benefit.53 An earlier study that used CPAP in patients with acute respiratory failure predominantly due to acute lung injury showed early physiologic improvements but no reduction in the need for intubation, no improvement in outcomes, and a higher rate of adverse events, including cardiac arrest, in those randomized to CPAP.54
A subsequent observational cohort specifically identified shock, metabolic acidosis, and severe hypoxemia as predictors of NIPPV failure.55
A more recent prospective study demonstrated that NIPPV improved gas exchange and obviated intubation in 54% of patients, with a consequent reduction in ventilator-associated pneumonia and a lower rate of death in the intensive care unit.56 A Simplified Acute Physiology Score (SAPS) II greater than 34 and a Pao2/Fio2 ratio less than 175 after 1 hour of NIPPV were identified as predicting that NIPPV would fail.56
MISCELLANEOUS APPLICATIONS
The more widespread use of NIPPV has encouraged its use in other acute situations, including during procedures such as percutaneous endoscopic gastrostomy (PEG)57,58 or bronchoscopy,59,60 for palliative use in patients listed as “do-not-intubate,”61–63 and for oxygenation before intubation.64
NIPPV during PEG tube insertion
NIPPV during PEG tube placement is particularly useful for patients with neuromuscular diseases who are at a combined risk of aspiration, poor oral intake, and respiratory failure during procedures. The experience with patients with amyotrophic lateral sclerosis58 and Duchenne muscular dystrophy57 indicates that even patients at high risk of respiratory failure during procedures can be successfully managed with NIPPV. The most recent practice parameters for patients with amyotrophic lateral sclerosis propose that patients with dysphagia may be exposed to less risk if the PEG procedure is performed when the forced vital capacity is greater than 50% of predicted.65
In randomized trials of CPAP59 or pressure-support NIPPV60 in high-risk hypoxemic patients who needed diagnostic bronchoscopy, patients in the intervention groups fared better than those who received oxygen alone, with better oxygenation during and after the procedure and a lower risk of postprocedure respiratory failure. Improved hemodynamics with a lower mean heart rate and a stable mean arterial pressure were also reported in one of those studies.60
Palliative use in ‘do-not-intubate’ patients
In patients who decline intubation, NIPPV appears to be most effective in reversing acute respiratory failure and improving mortality rates in those with COPD or with cardiogenic pulmonary edema.61,62 Controversy surrounding the use of NIPPV in “do-not-intubate” patients, particularly as a potentially uncomfortable life support technique, has been addressed by a task force of the Society of Critical Care Medicine, which recommends that it be applied only after careful discussion of goals of care and parameters of treatment with patients and their families.63
Oxygenation before intubation
In a prospective randomized study of oxygenation before rapid-sequence intubation via either a nonrebreather bag-valve mask or NIPPV, the NIPPV group had a higher oxygen saturation rate before, during, and after the intubation procedure.64
Acknowledgment: The authors wish to thank Jodith Janes of the Cleveland Clinic Alumni Library for her help with reference citations and with locating articles.
- Ambrosino N, Vagheggini G. Noninvasive positive pressure ventilation in the acute care setting: where are we? Eur Respir J 2008; 31:874–886.
- Hill NS, Brennan J, Garpestad E, Nava S. Noninvasive ventilation in acute respiratory failure. Crit Care Med 2007; 35:2402–2407.
- Nava S, Navalesi P, Conti G. Time of non-invasive ventilation. Intensive Care Med 2006; 32:361–370.
- Stoller JK, Kester L, Roberts VT, et al; An analysis of features of respiratory therapy departments that are avid for change. Respir Care 2008; 53:871–884.
- Maheshwari V, Paioli D, Rothaar R, Hill NS. Utilization of noninvasive ventilation in acute care hospitals: a regional survey. Chest 2006; 129:1226–1233.
- Obladen M. History of neonatal resuscitation. Part 1: Artificial ventilation. Neonatology 2008; 94:144–149.
- Poulton EP, Oxon DM. Left-sided heart failure with pulmonary oedema: its treatment with the “pulmonary plus” pressure machine. Lancet 1936; 228:981–983.
- Motley HL, Werko L. Observations on the clinical use of intermittent positive pressure. J Aviat Med 1947; 18:417–435.
- Cournand A, Motley HL. Physiological studies of the effects of intermittent positive pressure breathing on cardiac output in man. Am J Physiol 1948; 152:162–174.
- Severinghaus JW, Astrup P, Murray JF. Blood gas analysis and critical care medicine. Am J Respir Crit Care Med 1998; 157:S114–S122.
- Sullivan CE, Berthon-Jones M, Issa FG. Remission of severe obesity-hypoventilation syndrome after short-term treatment during sleep with nasal continuous positive airway pressure. Am Rev Respir Dis 1983; 128:177–181.
- Ellis ER, Bye PT, Bruderer JW, Sullivan CE. Treatment of respiratory failure during sleep in patients with neuromuscular disease. Positive-pressure ventilation through a nose mask. Am Rev Respir Dis 1987; 135:148–152.
- Sanders MH, Kern N. Obstructive sleep apnea treated by independently adjusted inspiratory and expiratory positive airway pressures via nasal mask. Physiologic and clinical implications. Chest 1990; 98:317–324.
- Bach JR. Mechanical exsufflation, noninvasive ventilation, and new strategies for pulmonary rehabilitation and sleep disordered breathing. Bull N Y Acad Med 1992; 68:321–340.
- Chevrolet JC, Jolliet P, Abajo B, Toussi A, Louis M. Nasal positive pressure ventilation in patients with acute respiratory failure. Difficult and time-consuming procedure for nurses. Chest 1991; 100:775–782.
- Criner GJ, Kreimer DT, Tomaselli M, Pierson W, Evans D. Financial implications of noninvasive positive pressure ventilation (NPPV). Chest 1995; 108:475–481.
- Nava S, Evangelisti I, Rampulla C, Compagnoni ML, Fracchia C, Rubini F. Human and financial costs of noninvasive mechanical ventilation in patients affected by COPD and acute respiratory failure. Chest 1997; 111:1631–1638.
- DiMarco AF, Onders RP, Ignagni A, Kowalski KE, Mortimer JT. Phrenic nerve pacing via intramuscular diaphragm electrodes in tetraplegic subjects. Chest 2005; 127:671–678.
- Antonelli M, Conti G. Noninvasive positive pressure ventilation as treatment for acute respiratory failure in critically ill patients. Crit Care 2000; 4:15–22.
- Hilbert G, Gruson D, Vargas F, et al. Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. N Engl J Med 2001; 344:481–487.
- L’Her E, Deye N, Lellouche F, et al. Physiologic effects of noninvasive ventilation during acute lung injury. Am J Respir Crit Care Med 2005; 172:1112–1118.
- Esteban A, Frutos-Vivar F, Ferguson ND, et al. Noninvasive positive-pressure ventilation for respiratory failure after extubation. N Engl J Med 2004; 350:2452–2460.
- Hill NS. Noninvasive positive pressure ventilation for respiratory failure caused by exacerbations of chronic obstructive pulmonary disease: a standard of care? Crit Care 2003; 7:400–401.
- Lightowler JV, Wedzicha JA, Elliott MW, Ram FS. Non-invasive positive pressure ventilation to treat respiratory failure resulting from exacerbations of chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. BMJ 2003; 326:185–187.
- Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995; 333:817–822.
- Keenan SP, Sinuff T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic obstructive pulmonary disease benefit from noninvasive positive-pressure ventilation? A systematic review of the literature. Ann Intern Med 2003; 138:861–870.
- Epstein SK. Noninvasive ventilation to shorten the duration of mechanical ventilation. Respir Care 2009; 54:198–208.
- Gray A, Goodacre S, Newby DE, Masson M, Sampson F, Nicholl J; 3CPO Trialists. Noninvasive ventilation in acute cardiogenic pulmonary edema. N Engl J Med 2008; 359:142–151.
- Collins SP, Mielniczuk LM, Whittingham HA, Boseley ME, Schramm DR, Storrow AB. The use of noninvasive ventilation in emergency department patients with acute cardiogenic pulmonary edema: a systematic review. Ann Emerg Med 2006; 48:260–269.
- Masip J, Roque M, Sánchez B, Fernández R, Subirana M, Expósito JA. Noninvasive ventilation in acute cardiogenic pulmonary edema: systematic review and meta-analysis. JAMA 2005; 294:3124–3130.
- Baratz DM, Westbrook PR, Shah PK, Mohsenifar Z. Effect of nasal continuous positive airway pressure on cardiac output and oxygen delivery in patients with congestive heart failure. Chest 1992; 102:1397–1401.
- Naughton MT, Rahman MA, Hara K, Floras JS, Bradley TD. Effect of continuous positive airway pressure on intrathoracic and left ventricular transmural pressures in patients with congestive heart failure. Circulation 1995; 91:1725–1731.
- Nava S, Carbone G, DiBattista N, et al. Noninvasive ventilation in cardiogenic pulmonary edema: a multicenter randomized trial. Am J Respir Crit Care Med 2003; 168:1432–1437.
- Mehta S, Jay GD, Woolard RH, et al. Randomized, prospective trial of bilevel versus continuous positive airway pressure in acute pulmonary edema. Crit Care Med 1997; 25:620–628.
- Ho KM, Wong K. A comparison of continuous and bi-level positive airway pressure non-invasive ventilation in patients with acute cardiogenic pulmonary oedema: a meta-analysis. Crit Care 2006; 10:R49.
- Antonelli M, Conti G, Bufi M, et al. Noninvasive ventilation for treatment of acute respiratory failure in patients undergoing solid organ transplantation: a randomized trial. JAMA 2000; 283:235–241.
- Keenan SP, Powers C, McCormack DG, Block G. Noninvasive positive-pressure ventilation for postextubation respiratory distress: a randomized controlled trial. JAMA 2002; 287:3238–3244.
- Ferrer M, Valencia M, Nicolas JM, Bernadich O, Badia JR, Torres A. Early noninvasive ventilation averts extubation failure in patients at risk: a randomized trial. Am J Respir Crit Care Med 2006; 173:164–170.
- Ferrer M, Sellarés J, Valencia M, et al. Non-invasive ventilation after extubation in hypercapnic patients with chronic respiratory disorders: randomised controlled trial. Lancet 2009; 374:1082–1088.
- Nava S, Gregoretti C, Fanfulla F, et al. Noninvasive ventilation to prevent respiratory failure after extubation in high-risk patients. Crit Care Med 2005; 33:2465–2470.
- El-Solh AA, Aquilina A, Pineda L, Dhanvantri V, Grant B, Bouquin P. Noninvasive ventilation for prevention of post-extubation respiratory failure in obese patients. Eur Respir J 2006; 28:588–595.
- Ferrer M, Esquinas A, Arancibia F, et al. Noninvasive ventilation during persistent weaning failure: a randomized controlled trial. Am J Respir Crit Care Med 2003; 168:70–76.
- Girault C, Daudenthun I, Chevron V, Tamion F, Leroy J, Bonmarchand G. Noninvasive ventilation as a systematic extubation and weaning technique in acute-on-chronic respiratory failure: a prospective, randomized controlled study. Am J Respir Crit Care Med 1999; 160:86–92.
- Nava S, Ambrosino N, Clini E, et al. Noninvasive mechanical ventilation in the weaning of patients with respiratory failure due to chronic obstructive pulmonary disease. A randomized, controlled trial. Ann Intern Med 1998; 128:721–728.
- Trevisan CE, Vieira SR; Research Group in Mechanical Ventilation Weaning. Noninvasive mechanical ventilation may be useful in treating patients who fail weaning from invasive mechanical ventilation: a randomized clinical trial. Crit Care 2008; 12:R51.
- Boles JM, Bion J, Connors A, et al. Weaning from mechanical ventilation. Eur Respir J 2007; 29:1033–1056.
- Burns KE, Adhikari NK, Meade MO. A meta-analysis of noninvasive weaning to facilitate liberation from mechanical ventilation. Can J Anaesth 2006; 53:305–315.
- Meduri GU, Cook TR, Turner RE, Cohen M, Leeper KV. Noninvasive positive pressure ventilation in status asthmaticus. Chest 1996; 110:767–774.
- Soroksky A, Stav D, Shpirer I. A pilot prospective, randomized, placebo-controlled trial of bilevel positive airway pressure in acute asthmatic attack. Chest 2003; 123:1018–1025.
- Holley MT, Morrissey TK, Seaberg DC, Afessa B, Wears RL. Ethical dilemmas in a randomized trial of asthma treatment: can Bayesian statistical analysis explain the results? Acad Emerg Med 2001; 8:1128–1135.
- Ram FS, Wellington S, Rowe BH, Wedzicha JA. Non-invasive positive pressure ventilation for treatment of respiratory failure due to severe acute exacerbations of asthma. Cochrane Database Syst Rev 2005;CD004360.
- Medoff BD. Invasive and noninvasive ventilation in patients with asthma. Respir Care 2008; 53:740–748.
- Agarwal R, Reddy C, Aggarwal AN, Gupta D. Is there a role for noninvasive ventilation in acute respiratory distress syndrome? A meta-analysis. Respir Med 2006; 100:2235–2238.
- Delclaux C, L’Her E, Alberti C, et al. Treatment of acute hypoxemic nonhypercapnic respiratory insufficiency with continuous positive airway pressure delivered by a face mask: a randomized controlled trial. JAMA 2000; 284:2352–2360.
- Rana S, Jenad H, Gay PC, Buck CF, Hubmayr RD, Gajic O. Failure of non-invasive ventilation in patients with acute lung injury: observational cohort study. Crit Care 2006; 10:R79.
- Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med 2007; 35:18–25.
- Birnkrant DJ, Ferguson RD, Martin JE, Gordon GJ. Noninvasive ventilation during gastrostomy tube placement in patients with severe Duchenne muscular dystrophy: case reports and review of the literature. Pediatr Pulmonol 2006; 41:188–193.
- Boitano LJ, Jordan T, Benditt JO. Noninvasive ventilation allows gastrostomy tube placement in patients with advanced ALS. Neurology 2001; 56:413–414.
- Maitre B, Jaber S, Maggiore SM, et al. Continuous positive airway pressure during fiberoptic bronchoscopy in hypoxemic patients. A randomized double-blind study using a new device. Am J Respir Crit Care Med 2000; 162:1063–1067.
- Antonelli M, Conti G, Rocco M, et al. Noninvasive positive-pressure ventilation vs conventional oxygen supplementation in hypoxemic patients undergoing diagnostic bronchoscopy. Chest 2002; 121:1149–1154.
- Levy M, Tanios MA, Nelson D, et al. Outcomes of patients with do-not-intubate orders treated with noninvasive ventilation. Crit Care Med 2004; 32:2002–2007.
- Schettino G, Altobelli N, Kacmarek RM. Noninvasive positive pressure ventilation reverses acute respiratory failure in select “do-not-intubate” patients. Crit Care Med 2005; 33:1976–1982.
- Curtis JR, Cook DJ, Sinuff T, et al; Society of Critical Care Medicine Palliative Noninvasive Positive Ventilation Task Force. Noninvasive positive pressure ventilation in critical and palliative care settings: understanding the goals of therapy. Crit Care Med 2007; 35:932–939.
- Baillard C, Fosse JP, Sebbane M, et al. Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med 2006; 174:171–177.
- Miller RG, Jackson CE, Kasarskis EJ, et al; Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter update: the care of the patient with amyotrophic lateral sclerosis: drug, nutritional, and respiratory therapies (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2009; 73:1218–1226.
Noninvasive positive pressure ventilation (NIPPV)—delivered via a tight-fitting mask rather than via an endotracheal tube or tracheostomy—is one of the most important advances in the management of acute respiratory failure to emerge in the past 2 decades. It is now recommended as the first choice for ventilatory support in selected patients, such as those with exacerbations of chronic obstructive pulmonary disease (COPD) or with cardiogenic pulmonary edema.1–3 In fact, some authors suggest that using NIPPV in more than 20% of COPD patients is a characteristic of respiratory care departments that are “avid for change”4—change being a good thing.
However, NIPPV has not been universally accepted, with wide variations in its utilization. In a 2006 survey, it was being used in only 33% of patients with COPD or congestive heart failure, for which it might be indicated. 5 Some potential reasons for the low rate are that physicians do not know about it, respiratory therapists are not sufficiently trained in it, and hospitals lack the equipment to do it.5
Our goal in this review is to familiarize the reader with how NIPPV has evolved and with its indications and contraindications in specific acute care conditions.
FROM A VACUUM CLEANER TO THE INTENSIVE CARE UNIT
NIPPV appears to have been first tried in 1870 by Chaussier, who used a bag and face mask to resuscitate neonates.6
In 1936, Poulton and Oxon7 described their “pulmonary plus pressure machine,” which used a vacuum cleaner blower and a mask to increase the alveolar pressure and thus counteract the increased intrapulmonary pressure in patients with heart failure, pulmonary edema, Cheyne-Stokes breathing, and asthma.
In the 1940s, intermittent positive pressure breathing devices were developed for use in high-altitude aviation. Motley, Werko, and Cournand8,9 subsequently used these devices to treat acute respiratory failure in pneumonia, pulmonary edema, near-drowning, Guillain-Barré syndrome, and acute severe asthma.
Although NIPPV was shown to be effective for acute conditions, invasive ventilation became preferred, particularly as blood gas analysis and ventilator technologies simultaneously matured, spurred at least in part by the polio epidemics of the 1950s.10
NIPPV reemerged in the 1980s for use in chronic conditions. First, continuous positive airway pressure (CPAP) came into use for obstructive sleep apnea,11 followed by noninvasive positive-pressure volume ventilation in neuromuscular diseases.12 Bilevel positive pressure devices (ie, with separate inspiratory and expiratory pressures) soon followed, again initially for obstructive sleep apnea13 and then for diverse neuromuscular diseases.14
NIPPV is now a mainstream therapy for diverse conditions in acute and chronic care.3 One reason we now use it in acute conditions is to avoid the complications associated with intubation.
Some clinicians initially resisted using NIPPV, concerned that it demanded too much of the nurses’ time15 and was costly.16 However, in a 1997 study in patients with COPD and acute respiratory failure, Nava et al17 found that NIPPV was no more expensive and no more demanding of staff resources than invasive mechanical ventilation in the first 48 hours of ventilation. Further, after the first few days of ventilation, NIPPV put fewer time demands on physicians and nurses than did invasive mechanical ventilation.
THREE MODES: CPAP, PRESSURE-LIMITED, VOLUME-LIMITED
The term “noninvasive ventilation” generally encompasses various forms of positive pressure ventilation. However, negative pressure ventilation, in the form of diaphragm pacing, may regain a foothold in the devices used for respiratory support.18 We therefore favor the term “NIPPV” in this review.
NIPPV IN ACUTE RESPIRATORY FAILURE
The main reasons to use NIPPV instead of invasive ventilation in acute care are to avoid the complications of invasive ventilation, to improve outcomes (eg, reduce mortality rates, decrease hospital length of stay), and to decrease the cost of care.
NIPPV is the standard of care for acute exacerbations of COPD
In a meta-analysis of eight randomized controlled trials,24 the specific advantages of NIPPV compared with usual care in acute exacerbations of COPD included:
- A lower risk of treatment failure, defined as death, need for intubation, or inability to tolerate the treatment (relative risk [RR] 0.51, number needed to treat [NNT] to prevent one treatment failure = 5)
- A lower risk of intubation (RR 0.43, NNT = 5)
- A lower mortality rate (RR 0.41, NNT = 8)
- A lower risk of complications (RR 0.32, NNT = 3)
- A shorter hospital length of stay (by about 3 days).
Mechanisms by which NIPPV may impart these benefits include reducing the work of breathing, unloading the respiratory muscles, lessening diaphragmatic pressure swings, reducing the respiratory rate, eliminating diaphragmatic work, and counteracting the threshold loading effects of auto-positive end-expiratory pressure (auto-PEEP).24–26
Also, if a patient with COPD is intubated, NIPPV seems to help after the tube is removed, preventing postextubation respiratory failure and facilitating weaning from invasive ventilation.27 These topics are discussed below.
A Cochrane systematic review24 concluded that NIPPV should be tried early in the course of respiratory failure, before severe acidosis develops. The patients in the studies in this review all had partial pressure of arterial carbon dioxide (Paco2) levels greater than 45 mm Hg.
In patients with severe respiratory acidosis (pH < 7.25), NIPPV failure rates are greater than 50%. However, trying NIPPV may still be justified, even in the presence of hypercapnic encephalopathy, as long as no other indications for invasive support and facilities for prompt endotracheal intubation are available. 1
However, in another systematic review,26 in patients with mild COPD exacerbations (pH > 7.35), NIPPV was no more effective than standard medical therapy in preventing acute respiratory failure, preventing death, or reducing length of hospitalization. Moreover, nearly 50% of the patients could not tolerate NIPPV.
Rapid improvement in cardiogenic pulmonary edema, but possibly no lower mortality rate
The Three Interventions in Cardiogenic Pulmonary Oedema (3CPO) trial,28 with 1,156 patients, was the largest randomized trial to compare NIPPV and standard oxygen therapy for acute pulmonary edema. It found that NIPPV (either CPAP or noninvasive intermittent positive pressure ventilation) was significantly better than standard oxygen therapy (through a variable-delivery oxygen mask with a reservoir) in the first hour of treatment in terms of the dyspnea score, heart rate, acidosis, and hypercapnia. However, there were no significant differences between groups in the 7- or 30-day mortality rates, the rates of intubation, rates of admission to the critical care unit, or in the mean length of hospital stay.
In contrast, several smaller randomized trials and meta-analyses showed lower intubation and mortality rates with NIPPV.29,30 Factors that may account for those differences include a much lower intubation rate in the 3CPO trial (2.9% overall, compared with 20% with conventional therapy in other trials), a higher mortality rate in the 3CPO trial, and methodologic differences (eg, patients for whom standard therapy failed in the 3CPO trial received rescue NIPPV).
If NIPPV is beneficial in cardiogenic pulmonary edema, the mechanisms are probably its favorable hemodynamic effects and its positive end-expiratory pressure (PEEP) effect on flooded alveoli. Specifically, positive intrathoracic pressure can be expected to reduce both preload and afterload, with improvement in the cardiac index and reduced work of breathing. 31,32
Notwithstanding the possible lack of impact of NIPPV on death or intubation rates in this setting, the intervention rapidly improves dyspnea and respiratory and metabolic abnormalities and should be considered for treatment of cardiogenic pulmonary edema associated with severe respiratory distress. A subgroup in which the NIPPV may reduce intubation rates is those with hypercapnia.33 A concern that NIPPV may increase the rate of myocardial infarction34 was not confirmed in the 3CPO trial.28 Interestingly, there were no differences in outcomes between CPAP and noninvasive intermittent positive pressure ventilation in this setting.28,34,35
Immunocompromised patients with acute respiratory failure
A particular challenge of NIPPV in immunocompromised patients, particularly compared with its use in COPD exacerbation or cardiogenic pulmonary edema, is that the underlying pathophysiology of respiratory dysfunction in immunocompromised patients may not be readily reversible. Therefore, its application in this group may need to follow clearly defined indications.
In one trial,20 inclusion criteria were:
- Immune suppression (due to neutropenia after chemotherapy or bone marrow transplantation, immunosuppressive drugs for organ transplantation, corticosteroids, cytotoxic therapy for nonmalignant conditions, or the acquired immunodeficiency syndrome)
- Persistent pulmonary infiltrates
- Fever (temperature > 38.3°C; 100.9°F)
- A respiratory rate greater than 30 breaths per minute
- Severe dyspnea at rest
- Early hypoxemic acute respiratory failure, defined as a ratio of the partial pressure of arterial oxygen to the fraction of inspired oxygen (Pao2/Fio2 ratio) less than 200 while on oxygen.
Compared with patients who received conventional treatment, fewer of those randomized to additional intermittent noninvasive ventilation had to be intubated (46% vs 77%, P = .03), suffered serious complications (50% vs 81%, P = .02), or died in the intensive care unit (38% vs 69%, P = .03) or in the hospital (50% vs 81%, P = .02).
Similarly, in a randomized trial in 40 patients with acute respiratory failure after solid organ transplantation, more patients in the NIPPV group than in the control group had an improvement in the Pao2/Fio2 ratio within the first hour (70% vs 25%, P = .004) or a sustained improvement in the Pao2/Fio2 ratio (60% vs 25%, P = .03); fewer of them needed endotracheal intubation (20% vs 70%, P = .002); fewer of them died of complications (20% vs 50%, P = .05); they had a shorter length of stay in the intensive care unit (mean 5.5 vs 9 days, P = .03); and fewer of them died in the intensive care unit (20% vs 50%, P = .05). There was, however, no difference in the overall hospital mortality rate.36
MAY NOT HELP AFTER EXTUBATION, EXCEPT IN SPECIFIC CASES
NIPPV has been used to treat respiratory failure after extubation,22,37 to prevent acute respiratory failure after failure of weaning,38–41 and to support breathing in patients who failed a trial of spontaneous breathing.42–45
Unfortunately, the evidence for using NIPPV in respiratory failure after extubation, including unplanned extubation, appears to be unfavorable, except possibly in patients with chronic pulmonary disease (particularly COPD and possibly obesity) and hypercapnia. An international consensus report stated that NIPPV should be considered in patients with hypercapnic respiratory insufficiency, especially those with COPD, to shorten the duration of intubation, but that it should not be routinely used in extubation respiratory failure.46
Treatment of respiratory failure after extubation
Two recent randomized controlled trials compared NIPPV and standard care in patients who met the criteria for readiness for extubation but who developed respiratory failure after mechanical ventilation was discontinued. 22,37 Those two studies showed a longer time to reintubation for patients randomized to NIPPV but no differences in the rate of reintubation between the two groups and no difference in the lengths of stay in the intensive care unit.
Of greater concern, one study showed a higher rate of death in the intensive care unit in the NIPPV group than in the standard therapy group (25% vs 14%, respectively).22 This finding suggests that NIPPV delayed necessary reintubation in patients developing respiratory failure after extubation, with a consequent risk of fatal complications.
Prevention of respiratory failure after extubation
Other studies used NIPPV to prevent respiratory failure after extubation rather than wait to apply it after respiratory failure developed.38–41
Nava et al,40 in a trial in patients successfully weaned but considered to be at risk of reintubation, found that fewer of those randomized to NIPPV had to be reintubated than those who received standard care (8% vs 24%), and 10% fewer of them died in the intensive care unit. Risk factors for reintubation (and therefore eligibility criteria for this trial) included a Paco2 higher than 45 mm Hg, more than one consecutive failure of weaning, chronic heart failure, other comorbidity, weak cough, or stridor.
Extubated patients are a heterogeneous group, so if some subgroups benefit from a transition to NIPPV after extubation, it will be important to identify them. For instance, a subgroup analysis of a study by Ferrer et al38 indicated the survival benefit of NIPPV after extubation was limited to patients with chronic respiratory disorders and hypercapnia during a trial of spontaneous breathing.
In a subsequent successful test of this hypothesis, a randomized trial showed that the early use of noninvasive ventilation in patients with hypercapnia after a trial of spontaneous breathing and with chronic respiratory disorders (COPD, chronic bronchitis, bronchiectasis, obesity-hypoventilation, sequelae of tuberculosis, chest wall deformity, or chronic persistent asthma) reduced the risk of respiratory failure after extubation and the risk of death within the first 90 days.39
Others in which this approach may be helpful are obese patients who have high Paco2 levels. Compared with historical controls, 62 patients with a body mass index greater than 35 kg/m2 who received NIPPV in the 48 hours after extubation had a lower rate of respiratory failure, shorter lengths of stay in the intensive care unit and hospital, and, in the subgroup with hypercapnia, a lower hospital mortality rate.41
NIPPV to facilitate weaning
In several studies, mechanically ventilated patients who had failed a trial of spontaneous breathing were randomized to undergo either accelerated weaning, extubation, and NIPPV or conventional weaning with pressure support via mechanical ventilation.42–46 Most patients developed hypercapnia during the spontaneous breathing trials, and most of the patients had COPD.
A meta-analysis47 of the randomized trials of this approach concluded that, compared with continued invasive ventilation, NIPPV decreased the risk of death (relative risk 0.41) and of ventilator-associated pneumonia (relative risk 0.28) and reduced the total duration of mechanical ventilation by a weighted mean difference of 7.33 days. The benefits appeared to be most significant in patients with COPD.
NIPPV IN ASTHMA AND STATUS ASTHMATICUS
Noninvasive ventilation is an attractive alternative to intubation for patients with status asthmaticus, given the challenges and conflicting demands of maintaining ventilation despite severe airway obstruction.
In a 1996 prospective study of 17 episodes of asthma associated with acute respiratory failure, Meduri et al48 showed that NIPPV could progressively improve the pH and the Paco2 over 12 to 24 hours and reduce the respiratory rate.
In a subsequent controlled trial, Soroksky et al49 randomized 30 patients presenting to an emergency room with a severe asthma attack to NIPPV with conventional therapy vs conventional therapy only. The study group had a significantly greater increase in the forced expiratory volume in 1 second compared with the control group (54% vs 29%, respectively) and a lower hospitalization rate (18% vs 63%).
Another randomized trial of NIPPV, in patients with status asthmaticus presenting to an emergency room, was prematurely terminated due to a physician treatment bias that favored NIPPV.50 The preliminary results of that study showed a 7.3% higher intubation rate in the control group than in the NIPPV group, along with trends toward a lower intubation rate, a shorter length of hospital stay, and lower hospital charges in the NIPPV group.
Despite these initial favorable results, a Cochrane review concluded that the use of NIPPV in patients with status asthmaticus is controversial.51 NIPPV can be tried in selected patients such as those with mild to moderate respiratory distress (respiratory rate greater than 25 breaths per minute, use of accessory muscles to breathe, difficulty speaking), an arterial pH of 7.25 to 7.35, and a Paco2 of 45 to 55 mm Hg.52 Patients with impending respiratory failure or the inability to protect the airway should probably not be considered for NIPPV.52
IN ACUTE LUNG INJURY AND ACUTE RESPIRATORY DISTRESS SYNDROME
The most challenging application of NIPPV may be in patients with acute lung injury and the acute respiratory distress syndrome.
Initial trials of NIPPV in this setting have been disappointing, and a meta-analysis of the topic concluded that NIPPV was unlikely to have any significant benefit.53 An earlier study that used CPAP in patients with acute respiratory failure predominantly due to acute lung injury showed early physiologic improvements but no reduction in the need for intubation, no improvement in outcomes, and a higher rate of adverse events, including cardiac arrest, in those randomized to CPAP.54
A subsequent observational cohort specifically identified shock, metabolic acidosis, and severe hypoxemia as predictors of NIPPV failure.55
A more recent prospective study demonstrated that NIPPV improved gas exchange and obviated intubation in 54% of patients, with a consequent reduction in ventilator-associated pneumonia and a lower rate of death in the intensive care unit.56 A Simplified Acute Physiology Score (SAPS) II greater than 34 and a Pao2/Fio2 ratio less than 175 after 1 hour of NIPPV were identified as predicting that NIPPV would fail.56
MISCELLANEOUS APPLICATIONS
The more widespread use of NIPPV has encouraged its use in other acute situations, including during procedures such as percutaneous endoscopic gastrostomy (PEG)57,58 or bronchoscopy,59,60 for palliative use in patients listed as “do-not-intubate,”61–63 and for oxygenation before intubation.64
NIPPV during PEG tube insertion
NIPPV during PEG tube placement is particularly useful for patients with neuromuscular diseases who are at a combined risk of aspiration, poor oral intake, and respiratory failure during procedures. The experience with patients with amyotrophic lateral sclerosis58 and Duchenne muscular dystrophy57 indicates that even patients at high risk of respiratory failure during procedures can be successfully managed with NIPPV. The most recent practice parameters for patients with amyotrophic lateral sclerosis propose that patients with dysphagia may be exposed to less risk if the PEG procedure is performed when the forced vital capacity is greater than 50% of predicted.65
In randomized trials of CPAP59 or pressure-support NIPPV60 in high-risk hypoxemic patients who needed diagnostic bronchoscopy, patients in the intervention groups fared better than those who received oxygen alone, with better oxygenation during and after the procedure and a lower risk of postprocedure respiratory failure. Improved hemodynamics with a lower mean heart rate and a stable mean arterial pressure were also reported in one of those studies.60
Palliative use in ‘do-not-intubate’ patients
In patients who decline intubation, NIPPV appears to be most effective in reversing acute respiratory failure and improving mortality rates in those with COPD or with cardiogenic pulmonary edema.61,62 Controversy surrounding the use of NIPPV in “do-not-intubate” patients, particularly as a potentially uncomfortable life support technique, has been addressed by a task force of the Society of Critical Care Medicine, which recommends that it be applied only after careful discussion of goals of care and parameters of treatment with patients and their families.63
Oxygenation before intubation
In a prospective randomized study of oxygenation before rapid-sequence intubation via either a nonrebreather bag-valve mask or NIPPV, the NIPPV group had a higher oxygen saturation rate before, during, and after the intubation procedure.64
Acknowledgment: The authors wish to thank Jodith Janes of the Cleveland Clinic Alumni Library for her help with reference citations and with locating articles.
Noninvasive positive pressure ventilation (NIPPV)—delivered via a tight-fitting mask rather than via an endotracheal tube or tracheostomy—is one of the most important advances in the management of acute respiratory failure to emerge in the past 2 decades. It is now recommended as the first choice for ventilatory support in selected patients, such as those with exacerbations of chronic obstructive pulmonary disease (COPD) or with cardiogenic pulmonary edema.1–3 In fact, some authors suggest that using NIPPV in more than 20% of COPD patients is a characteristic of respiratory care departments that are “avid for change”4—change being a good thing.
However, NIPPV has not been universally accepted, with wide variations in its utilization. In a 2006 survey, it was being used in only 33% of patients with COPD or congestive heart failure, for which it might be indicated. 5 Some potential reasons for the low rate are that physicians do not know about it, respiratory therapists are not sufficiently trained in it, and hospitals lack the equipment to do it.5
Our goal in this review is to familiarize the reader with how NIPPV has evolved and with its indications and contraindications in specific acute care conditions.
FROM A VACUUM CLEANER TO THE INTENSIVE CARE UNIT
NIPPV appears to have been first tried in 1870 by Chaussier, who used a bag and face mask to resuscitate neonates.6
In 1936, Poulton and Oxon7 described their “pulmonary plus pressure machine,” which used a vacuum cleaner blower and a mask to increase the alveolar pressure and thus counteract the increased intrapulmonary pressure in patients with heart failure, pulmonary edema, Cheyne-Stokes breathing, and asthma.
In the 1940s, intermittent positive pressure breathing devices were developed for use in high-altitude aviation. Motley, Werko, and Cournand8,9 subsequently used these devices to treat acute respiratory failure in pneumonia, pulmonary edema, near-drowning, Guillain-Barré syndrome, and acute severe asthma.
Although NIPPV was shown to be effective for acute conditions, invasive ventilation became preferred, particularly as blood gas analysis and ventilator technologies simultaneously matured, spurred at least in part by the polio epidemics of the 1950s.10
NIPPV reemerged in the 1980s for use in chronic conditions. First, continuous positive airway pressure (CPAP) came into use for obstructive sleep apnea,11 followed by noninvasive positive-pressure volume ventilation in neuromuscular diseases.12 Bilevel positive pressure devices (ie, with separate inspiratory and expiratory pressures) soon followed, again initially for obstructive sleep apnea13 and then for diverse neuromuscular diseases.14
NIPPV is now a mainstream therapy for diverse conditions in acute and chronic care.3 One reason we now use it in acute conditions is to avoid the complications associated with intubation.
Some clinicians initially resisted using NIPPV, concerned that it demanded too much of the nurses’ time15 and was costly.16 However, in a 1997 study in patients with COPD and acute respiratory failure, Nava et al17 found that NIPPV was no more expensive and no more demanding of staff resources than invasive mechanical ventilation in the first 48 hours of ventilation. Further, after the first few days of ventilation, NIPPV put fewer time demands on physicians and nurses than did invasive mechanical ventilation.
THREE MODES: CPAP, PRESSURE-LIMITED, VOLUME-LIMITED
The term “noninvasive ventilation” generally encompasses various forms of positive pressure ventilation. However, negative pressure ventilation, in the form of diaphragm pacing, may regain a foothold in the devices used for respiratory support.18 We therefore favor the term “NIPPV” in this review.
NIPPV IN ACUTE RESPIRATORY FAILURE
The main reasons to use NIPPV instead of invasive ventilation in acute care are to avoid the complications of invasive ventilation, to improve outcomes (eg, reduce mortality rates, decrease hospital length of stay), and to decrease the cost of care.
NIPPV is the standard of care for acute exacerbations of COPD
In a meta-analysis of eight randomized controlled trials,24 the specific advantages of NIPPV compared with usual care in acute exacerbations of COPD included:
- A lower risk of treatment failure, defined as death, need for intubation, or inability to tolerate the treatment (relative risk [RR] 0.51, number needed to treat [NNT] to prevent one treatment failure = 5)
- A lower risk of intubation (RR 0.43, NNT = 5)
- A lower mortality rate (RR 0.41, NNT = 8)
- A lower risk of complications (RR 0.32, NNT = 3)
- A shorter hospital length of stay (by about 3 days).
Mechanisms by which NIPPV may impart these benefits include reducing the work of breathing, unloading the respiratory muscles, lessening diaphragmatic pressure swings, reducing the respiratory rate, eliminating diaphragmatic work, and counteracting the threshold loading effects of auto-positive end-expiratory pressure (auto-PEEP).24–26
Also, if a patient with COPD is intubated, NIPPV seems to help after the tube is removed, preventing postextubation respiratory failure and facilitating weaning from invasive ventilation.27 These topics are discussed below.
A Cochrane systematic review24 concluded that NIPPV should be tried early in the course of respiratory failure, before severe acidosis develops. The patients in the studies in this review all had partial pressure of arterial carbon dioxide (Paco2) levels greater than 45 mm Hg.
In patients with severe respiratory acidosis (pH < 7.25), NIPPV failure rates are greater than 50%. However, trying NIPPV may still be justified, even in the presence of hypercapnic encephalopathy, as long as no other indications for invasive support and facilities for prompt endotracheal intubation are available. 1
However, in another systematic review,26 in patients with mild COPD exacerbations (pH > 7.35), NIPPV was no more effective than standard medical therapy in preventing acute respiratory failure, preventing death, or reducing length of hospitalization. Moreover, nearly 50% of the patients could not tolerate NIPPV.
Rapid improvement in cardiogenic pulmonary edema, but possibly no lower mortality rate
The Three Interventions in Cardiogenic Pulmonary Oedema (3CPO) trial,28 with 1,156 patients, was the largest randomized trial to compare NIPPV and standard oxygen therapy for acute pulmonary edema. It found that NIPPV (either CPAP or noninvasive intermittent positive pressure ventilation) was significantly better than standard oxygen therapy (through a variable-delivery oxygen mask with a reservoir) in the first hour of treatment in terms of the dyspnea score, heart rate, acidosis, and hypercapnia. However, there were no significant differences between groups in the 7- or 30-day mortality rates, the rates of intubation, rates of admission to the critical care unit, or in the mean length of hospital stay.
In contrast, several smaller randomized trials and meta-analyses showed lower intubation and mortality rates with NIPPV.29,30 Factors that may account for those differences include a much lower intubation rate in the 3CPO trial (2.9% overall, compared with 20% with conventional therapy in other trials), a higher mortality rate in the 3CPO trial, and methodologic differences (eg, patients for whom standard therapy failed in the 3CPO trial received rescue NIPPV).
If NIPPV is beneficial in cardiogenic pulmonary edema, the mechanisms are probably its favorable hemodynamic effects and its positive end-expiratory pressure (PEEP) effect on flooded alveoli. Specifically, positive intrathoracic pressure can be expected to reduce both preload and afterload, with improvement in the cardiac index and reduced work of breathing. 31,32
Notwithstanding the possible lack of impact of NIPPV on death or intubation rates in this setting, the intervention rapidly improves dyspnea and respiratory and metabolic abnormalities and should be considered for treatment of cardiogenic pulmonary edema associated with severe respiratory distress. A subgroup in which the NIPPV may reduce intubation rates is those with hypercapnia.33 A concern that NIPPV may increase the rate of myocardial infarction34 was not confirmed in the 3CPO trial.28 Interestingly, there were no differences in outcomes between CPAP and noninvasive intermittent positive pressure ventilation in this setting.28,34,35
Immunocompromised patients with acute respiratory failure
A particular challenge of NIPPV in immunocompromised patients, particularly compared with its use in COPD exacerbation or cardiogenic pulmonary edema, is that the underlying pathophysiology of respiratory dysfunction in immunocompromised patients may not be readily reversible. Therefore, its application in this group may need to follow clearly defined indications.
In one trial,20 inclusion criteria were:
- Immune suppression (due to neutropenia after chemotherapy or bone marrow transplantation, immunosuppressive drugs for organ transplantation, corticosteroids, cytotoxic therapy for nonmalignant conditions, or the acquired immunodeficiency syndrome)
- Persistent pulmonary infiltrates
- Fever (temperature > 38.3°C; 100.9°F)
- A respiratory rate greater than 30 breaths per minute
- Severe dyspnea at rest
- Early hypoxemic acute respiratory failure, defined as a ratio of the partial pressure of arterial oxygen to the fraction of inspired oxygen (Pao2/Fio2 ratio) less than 200 while on oxygen.
Compared with patients who received conventional treatment, fewer of those randomized to additional intermittent noninvasive ventilation had to be intubated (46% vs 77%, P = .03), suffered serious complications (50% vs 81%, P = .02), or died in the intensive care unit (38% vs 69%, P = .03) or in the hospital (50% vs 81%, P = .02).
Similarly, in a randomized trial in 40 patients with acute respiratory failure after solid organ transplantation, more patients in the NIPPV group than in the control group had an improvement in the Pao2/Fio2 ratio within the first hour (70% vs 25%, P = .004) or a sustained improvement in the Pao2/Fio2 ratio (60% vs 25%, P = .03); fewer of them needed endotracheal intubation (20% vs 70%, P = .002); fewer of them died of complications (20% vs 50%, P = .05); they had a shorter length of stay in the intensive care unit (mean 5.5 vs 9 days, P = .03); and fewer of them died in the intensive care unit (20% vs 50%, P = .05). There was, however, no difference in the overall hospital mortality rate.36
MAY NOT HELP AFTER EXTUBATION, EXCEPT IN SPECIFIC CASES
NIPPV has been used to treat respiratory failure after extubation,22,37 to prevent acute respiratory failure after failure of weaning,38–41 and to support breathing in patients who failed a trial of spontaneous breathing.42–45
Unfortunately, the evidence for using NIPPV in respiratory failure after extubation, including unplanned extubation, appears to be unfavorable, except possibly in patients with chronic pulmonary disease (particularly COPD and possibly obesity) and hypercapnia. An international consensus report stated that NIPPV should be considered in patients with hypercapnic respiratory insufficiency, especially those with COPD, to shorten the duration of intubation, but that it should not be routinely used in extubation respiratory failure.46
Treatment of respiratory failure after extubation
Two recent randomized controlled trials compared NIPPV and standard care in patients who met the criteria for readiness for extubation but who developed respiratory failure after mechanical ventilation was discontinued. 22,37 Those two studies showed a longer time to reintubation for patients randomized to NIPPV but no differences in the rate of reintubation between the two groups and no difference in the lengths of stay in the intensive care unit.
Of greater concern, one study showed a higher rate of death in the intensive care unit in the NIPPV group than in the standard therapy group (25% vs 14%, respectively).22 This finding suggests that NIPPV delayed necessary reintubation in patients developing respiratory failure after extubation, with a consequent risk of fatal complications.
Prevention of respiratory failure after extubation
Other studies used NIPPV to prevent respiratory failure after extubation rather than wait to apply it after respiratory failure developed.38–41
Nava et al,40 in a trial in patients successfully weaned but considered to be at risk of reintubation, found that fewer of those randomized to NIPPV had to be reintubated than those who received standard care (8% vs 24%), and 10% fewer of them died in the intensive care unit. Risk factors for reintubation (and therefore eligibility criteria for this trial) included a Paco2 higher than 45 mm Hg, more than one consecutive failure of weaning, chronic heart failure, other comorbidity, weak cough, or stridor.
Extubated patients are a heterogeneous group, so if some subgroups benefit from a transition to NIPPV after extubation, it will be important to identify them. For instance, a subgroup analysis of a study by Ferrer et al38 indicated the survival benefit of NIPPV after extubation was limited to patients with chronic respiratory disorders and hypercapnia during a trial of spontaneous breathing.
In a subsequent successful test of this hypothesis, a randomized trial showed that the early use of noninvasive ventilation in patients with hypercapnia after a trial of spontaneous breathing and with chronic respiratory disorders (COPD, chronic bronchitis, bronchiectasis, obesity-hypoventilation, sequelae of tuberculosis, chest wall deformity, or chronic persistent asthma) reduced the risk of respiratory failure after extubation and the risk of death within the first 90 days.39
Others in which this approach may be helpful are obese patients who have high Paco2 levels. Compared with historical controls, 62 patients with a body mass index greater than 35 kg/m2 who received NIPPV in the 48 hours after extubation had a lower rate of respiratory failure, shorter lengths of stay in the intensive care unit and hospital, and, in the subgroup with hypercapnia, a lower hospital mortality rate.41
NIPPV to facilitate weaning
In several studies, mechanically ventilated patients who had failed a trial of spontaneous breathing were randomized to undergo either accelerated weaning, extubation, and NIPPV or conventional weaning with pressure support via mechanical ventilation.42–46 Most patients developed hypercapnia during the spontaneous breathing trials, and most of the patients had COPD.
A meta-analysis47 of the randomized trials of this approach concluded that, compared with continued invasive ventilation, NIPPV decreased the risk of death (relative risk 0.41) and of ventilator-associated pneumonia (relative risk 0.28) and reduced the total duration of mechanical ventilation by a weighted mean difference of 7.33 days. The benefits appeared to be most significant in patients with COPD.
NIPPV IN ASTHMA AND STATUS ASTHMATICUS
Noninvasive ventilation is an attractive alternative to intubation for patients with status asthmaticus, given the challenges and conflicting demands of maintaining ventilation despite severe airway obstruction.
In a 1996 prospective study of 17 episodes of asthma associated with acute respiratory failure, Meduri et al48 showed that NIPPV could progressively improve the pH and the Paco2 over 12 to 24 hours and reduce the respiratory rate.
In a subsequent controlled trial, Soroksky et al49 randomized 30 patients presenting to an emergency room with a severe asthma attack to NIPPV with conventional therapy vs conventional therapy only. The study group had a significantly greater increase in the forced expiratory volume in 1 second compared with the control group (54% vs 29%, respectively) and a lower hospitalization rate (18% vs 63%).
Another randomized trial of NIPPV, in patients with status asthmaticus presenting to an emergency room, was prematurely terminated due to a physician treatment bias that favored NIPPV.50 The preliminary results of that study showed a 7.3% higher intubation rate in the control group than in the NIPPV group, along with trends toward a lower intubation rate, a shorter length of hospital stay, and lower hospital charges in the NIPPV group.
Despite these initial favorable results, a Cochrane review concluded that the use of NIPPV in patients with status asthmaticus is controversial.51 NIPPV can be tried in selected patients such as those with mild to moderate respiratory distress (respiratory rate greater than 25 breaths per minute, use of accessory muscles to breathe, difficulty speaking), an arterial pH of 7.25 to 7.35, and a Paco2 of 45 to 55 mm Hg.52 Patients with impending respiratory failure or the inability to protect the airway should probably not be considered for NIPPV.52
IN ACUTE LUNG INJURY AND ACUTE RESPIRATORY DISTRESS SYNDROME
The most challenging application of NIPPV may be in patients with acute lung injury and the acute respiratory distress syndrome.
Initial trials of NIPPV in this setting have been disappointing, and a meta-analysis of the topic concluded that NIPPV was unlikely to have any significant benefit.53 An earlier study that used CPAP in patients with acute respiratory failure predominantly due to acute lung injury showed early physiologic improvements but no reduction in the need for intubation, no improvement in outcomes, and a higher rate of adverse events, including cardiac arrest, in those randomized to CPAP.54
A subsequent observational cohort specifically identified shock, metabolic acidosis, and severe hypoxemia as predictors of NIPPV failure.55
A more recent prospective study demonstrated that NIPPV improved gas exchange and obviated intubation in 54% of patients, with a consequent reduction in ventilator-associated pneumonia and a lower rate of death in the intensive care unit.56 A Simplified Acute Physiology Score (SAPS) II greater than 34 and a Pao2/Fio2 ratio less than 175 after 1 hour of NIPPV were identified as predicting that NIPPV would fail.56
MISCELLANEOUS APPLICATIONS
The more widespread use of NIPPV has encouraged its use in other acute situations, including during procedures such as percutaneous endoscopic gastrostomy (PEG)57,58 or bronchoscopy,59,60 for palliative use in patients listed as “do-not-intubate,”61–63 and for oxygenation before intubation.64
NIPPV during PEG tube insertion
NIPPV during PEG tube placement is particularly useful for patients with neuromuscular diseases who are at a combined risk of aspiration, poor oral intake, and respiratory failure during procedures. The experience with patients with amyotrophic lateral sclerosis58 and Duchenne muscular dystrophy57 indicates that even patients at high risk of respiratory failure during procedures can be successfully managed with NIPPV. The most recent practice parameters for patients with amyotrophic lateral sclerosis propose that patients with dysphagia may be exposed to less risk if the PEG procedure is performed when the forced vital capacity is greater than 50% of predicted.65
In randomized trials of CPAP59 or pressure-support NIPPV60 in high-risk hypoxemic patients who needed diagnostic bronchoscopy, patients in the intervention groups fared better than those who received oxygen alone, with better oxygenation during and after the procedure and a lower risk of postprocedure respiratory failure. Improved hemodynamics with a lower mean heart rate and a stable mean arterial pressure were also reported in one of those studies.60
Palliative use in ‘do-not-intubate’ patients
In patients who decline intubation, NIPPV appears to be most effective in reversing acute respiratory failure and improving mortality rates in those with COPD or with cardiogenic pulmonary edema.61,62 Controversy surrounding the use of NIPPV in “do-not-intubate” patients, particularly as a potentially uncomfortable life support technique, has been addressed by a task force of the Society of Critical Care Medicine, which recommends that it be applied only after careful discussion of goals of care and parameters of treatment with patients and their families.63
Oxygenation before intubation
In a prospective randomized study of oxygenation before rapid-sequence intubation via either a nonrebreather bag-valve mask or NIPPV, the NIPPV group had a higher oxygen saturation rate before, during, and after the intubation procedure.64
Acknowledgment: The authors wish to thank Jodith Janes of the Cleveland Clinic Alumni Library for her help with reference citations and with locating articles.
- Ambrosino N, Vagheggini G. Noninvasive positive pressure ventilation in the acute care setting: where are we? Eur Respir J 2008; 31:874–886.
- Hill NS, Brennan J, Garpestad E, Nava S. Noninvasive ventilation in acute respiratory failure. Crit Care Med 2007; 35:2402–2407.
- Nava S, Navalesi P, Conti G. Time of non-invasive ventilation. Intensive Care Med 2006; 32:361–370.
- Stoller JK, Kester L, Roberts VT, et al; An analysis of features of respiratory therapy departments that are avid for change. Respir Care 2008; 53:871–884.
- Maheshwari V, Paioli D, Rothaar R, Hill NS. Utilization of noninvasive ventilation in acute care hospitals: a regional survey. Chest 2006; 129:1226–1233.
- Obladen M. History of neonatal resuscitation. Part 1: Artificial ventilation. Neonatology 2008; 94:144–149.
- Poulton EP, Oxon DM. Left-sided heart failure with pulmonary oedema: its treatment with the “pulmonary plus” pressure machine. Lancet 1936; 228:981–983.
- Motley HL, Werko L. Observations on the clinical use of intermittent positive pressure. J Aviat Med 1947; 18:417–435.
- Cournand A, Motley HL. Physiological studies of the effects of intermittent positive pressure breathing on cardiac output in man. Am J Physiol 1948; 152:162–174.
- Severinghaus JW, Astrup P, Murray JF. Blood gas analysis and critical care medicine. Am J Respir Crit Care Med 1998; 157:S114–S122.
- Sullivan CE, Berthon-Jones M, Issa FG. Remission of severe obesity-hypoventilation syndrome after short-term treatment during sleep with nasal continuous positive airway pressure. Am Rev Respir Dis 1983; 128:177–181.
- Ellis ER, Bye PT, Bruderer JW, Sullivan CE. Treatment of respiratory failure during sleep in patients with neuromuscular disease. Positive-pressure ventilation through a nose mask. Am Rev Respir Dis 1987; 135:148–152.
- Sanders MH, Kern N. Obstructive sleep apnea treated by independently adjusted inspiratory and expiratory positive airway pressures via nasal mask. Physiologic and clinical implications. Chest 1990; 98:317–324.
- Bach JR. Mechanical exsufflation, noninvasive ventilation, and new strategies for pulmonary rehabilitation and sleep disordered breathing. Bull N Y Acad Med 1992; 68:321–340.
- Chevrolet JC, Jolliet P, Abajo B, Toussi A, Louis M. Nasal positive pressure ventilation in patients with acute respiratory failure. Difficult and time-consuming procedure for nurses. Chest 1991; 100:775–782.
- Criner GJ, Kreimer DT, Tomaselli M, Pierson W, Evans D. Financial implications of noninvasive positive pressure ventilation (NPPV). Chest 1995; 108:475–481.
- Nava S, Evangelisti I, Rampulla C, Compagnoni ML, Fracchia C, Rubini F. Human and financial costs of noninvasive mechanical ventilation in patients affected by COPD and acute respiratory failure. Chest 1997; 111:1631–1638.
- DiMarco AF, Onders RP, Ignagni A, Kowalski KE, Mortimer JT. Phrenic nerve pacing via intramuscular diaphragm electrodes in tetraplegic subjects. Chest 2005; 127:671–678.
- Antonelli M, Conti G. Noninvasive positive pressure ventilation as treatment for acute respiratory failure in critically ill patients. Crit Care 2000; 4:15–22.
- Hilbert G, Gruson D, Vargas F, et al. Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. N Engl J Med 2001; 344:481–487.
- L’Her E, Deye N, Lellouche F, et al. Physiologic effects of noninvasive ventilation during acute lung injury. Am J Respir Crit Care Med 2005; 172:1112–1118.
- Esteban A, Frutos-Vivar F, Ferguson ND, et al. Noninvasive positive-pressure ventilation for respiratory failure after extubation. N Engl J Med 2004; 350:2452–2460.
- Hill NS. Noninvasive positive pressure ventilation for respiratory failure caused by exacerbations of chronic obstructive pulmonary disease: a standard of care? Crit Care 2003; 7:400–401.
- Lightowler JV, Wedzicha JA, Elliott MW, Ram FS. Non-invasive positive pressure ventilation to treat respiratory failure resulting from exacerbations of chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. BMJ 2003; 326:185–187.
- Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995; 333:817–822.
- Keenan SP, Sinuff T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic obstructive pulmonary disease benefit from noninvasive positive-pressure ventilation? A systematic review of the literature. Ann Intern Med 2003; 138:861–870.
- Epstein SK. Noninvasive ventilation to shorten the duration of mechanical ventilation. Respir Care 2009; 54:198–208.
- Gray A, Goodacre S, Newby DE, Masson M, Sampson F, Nicholl J; 3CPO Trialists. Noninvasive ventilation in acute cardiogenic pulmonary edema. N Engl J Med 2008; 359:142–151.
- Collins SP, Mielniczuk LM, Whittingham HA, Boseley ME, Schramm DR, Storrow AB. The use of noninvasive ventilation in emergency department patients with acute cardiogenic pulmonary edema: a systematic review. Ann Emerg Med 2006; 48:260–269.
- Masip J, Roque M, Sánchez B, Fernández R, Subirana M, Expósito JA. Noninvasive ventilation in acute cardiogenic pulmonary edema: systematic review and meta-analysis. JAMA 2005; 294:3124–3130.
- Baratz DM, Westbrook PR, Shah PK, Mohsenifar Z. Effect of nasal continuous positive airway pressure on cardiac output and oxygen delivery in patients with congestive heart failure. Chest 1992; 102:1397–1401.
- Naughton MT, Rahman MA, Hara K, Floras JS, Bradley TD. Effect of continuous positive airway pressure on intrathoracic and left ventricular transmural pressures in patients with congestive heart failure. Circulation 1995; 91:1725–1731.
- Nava S, Carbone G, DiBattista N, et al. Noninvasive ventilation in cardiogenic pulmonary edema: a multicenter randomized trial. Am J Respir Crit Care Med 2003; 168:1432–1437.
- Mehta S, Jay GD, Woolard RH, et al. Randomized, prospective trial of bilevel versus continuous positive airway pressure in acute pulmonary edema. Crit Care Med 1997; 25:620–628.
- Ho KM, Wong K. A comparison of continuous and bi-level positive airway pressure non-invasive ventilation in patients with acute cardiogenic pulmonary oedema: a meta-analysis. Crit Care 2006; 10:R49.
- Antonelli M, Conti G, Bufi M, et al. Noninvasive ventilation for treatment of acute respiratory failure in patients undergoing solid organ transplantation: a randomized trial. JAMA 2000; 283:235–241.
- Keenan SP, Powers C, McCormack DG, Block G. Noninvasive positive-pressure ventilation for postextubation respiratory distress: a randomized controlled trial. JAMA 2002; 287:3238–3244.
- Ferrer M, Valencia M, Nicolas JM, Bernadich O, Badia JR, Torres A. Early noninvasive ventilation averts extubation failure in patients at risk: a randomized trial. Am J Respir Crit Care Med 2006; 173:164–170.
- Ferrer M, Sellarés J, Valencia M, et al. Non-invasive ventilation after extubation in hypercapnic patients with chronic respiratory disorders: randomised controlled trial. Lancet 2009; 374:1082–1088.
- Nava S, Gregoretti C, Fanfulla F, et al. Noninvasive ventilation to prevent respiratory failure after extubation in high-risk patients. Crit Care Med 2005; 33:2465–2470.
- El-Solh AA, Aquilina A, Pineda L, Dhanvantri V, Grant B, Bouquin P. Noninvasive ventilation for prevention of post-extubation respiratory failure in obese patients. Eur Respir J 2006; 28:588–595.
- Ferrer M, Esquinas A, Arancibia F, et al. Noninvasive ventilation during persistent weaning failure: a randomized controlled trial. Am J Respir Crit Care Med 2003; 168:70–76.
- Girault C, Daudenthun I, Chevron V, Tamion F, Leroy J, Bonmarchand G. Noninvasive ventilation as a systematic extubation and weaning technique in acute-on-chronic respiratory failure: a prospective, randomized controlled study. Am J Respir Crit Care Med 1999; 160:86–92.
- Nava S, Ambrosino N, Clini E, et al. Noninvasive mechanical ventilation in the weaning of patients with respiratory failure due to chronic obstructive pulmonary disease. A randomized, controlled trial. Ann Intern Med 1998; 128:721–728.
- Trevisan CE, Vieira SR; Research Group in Mechanical Ventilation Weaning. Noninvasive mechanical ventilation may be useful in treating patients who fail weaning from invasive mechanical ventilation: a randomized clinical trial. Crit Care 2008; 12:R51.
- Boles JM, Bion J, Connors A, et al. Weaning from mechanical ventilation. Eur Respir J 2007; 29:1033–1056.
- Burns KE, Adhikari NK, Meade MO. A meta-analysis of noninvasive weaning to facilitate liberation from mechanical ventilation. Can J Anaesth 2006; 53:305–315.
- Meduri GU, Cook TR, Turner RE, Cohen M, Leeper KV. Noninvasive positive pressure ventilation in status asthmaticus. Chest 1996; 110:767–774.
- Soroksky A, Stav D, Shpirer I. A pilot prospective, randomized, placebo-controlled trial of bilevel positive airway pressure in acute asthmatic attack. Chest 2003; 123:1018–1025.
- Holley MT, Morrissey TK, Seaberg DC, Afessa B, Wears RL. Ethical dilemmas in a randomized trial of asthma treatment: can Bayesian statistical analysis explain the results? Acad Emerg Med 2001; 8:1128–1135.
- Ram FS, Wellington S, Rowe BH, Wedzicha JA. Non-invasive positive pressure ventilation for treatment of respiratory failure due to severe acute exacerbations of asthma. Cochrane Database Syst Rev 2005;CD004360.
- Medoff BD. Invasive and noninvasive ventilation in patients with asthma. Respir Care 2008; 53:740–748.
- Agarwal R, Reddy C, Aggarwal AN, Gupta D. Is there a role for noninvasive ventilation in acute respiratory distress syndrome? A meta-analysis. Respir Med 2006; 100:2235–2238.
- Delclaux C, L’Her E, Alberti C, et al. Treatment of acute hypoxemic nonhypercapnic respiratory insufficiency with continuous positive airway pressure delivered by a face mask: a randomized controlled trial. JAMA 2000; 284:2352–2360.
- Rana S, Jenad H, Gay PC, Buck CF, Hubmayr RD, Gajic O. Failure of non-invasive ventilation in patients with acute lung injury: observational cohort study. Crit Care 2006; 10:R79.
- Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med 2007; 35:18–25.
- Birnkrant DJ, Ferguson RD, Martin JE, Gordon GJ. Noninvasive ventilation during gastrostomy tube placement in patients with severe Duchenne muscular dystrophy: case reports and review of the literature. Pediatr Pulmonol 2006; 41:188–193.
- Boitano LJ, Jordan T, Benditt JO. Noninvasive ventilation allows gastrostomy tube placement in patients with advanced ALS. Neurology 2001; 56:413–414.
- Maitre B, Jaber S, Maggiore SM, et al. Continuous positive airway pressure during fiberoptic bronchoscopy in hypoxemic patients. A randomized double-blind study using a new device. Am J Respir Crit Care Med 2000; 162:1063–1067.
- Antonelli M, Conti G, Rocco M, et al. Noninvasive positive-pressure ventilation vs conventional oxygen supplementation in hypoxemic patients undergoing diagnostic bronchoscopy. Chest 2002; 121:1149–1154.
- Levy M, Tanios MA, Nelson D, et al. Outcomes of patients with do-not-intubate orders treated with noninvasive ventilation. Crit Care Med 2004; 32:2002–2007.
- Schettino G, Altobelli N, Kacmarek RM. Noninvasive positive pressure ventilation reverses acute respiratory failure in select “do-not-intubate” patients. Crit Care Med 2005; 33:1976–1982.
- Curtis JR, Cook DJ, Sinuff T, et al; Society of Critical Care Medicine Palliative Noninvasive Positive Ventilation Task Force. Noninvasive positive pressure ventilation in critical and palliative care settings: understanding the goals of therapy. Crit Care Med 2007; 35:932–939.
- Baillard C, Fosse JP, Sebbane M, et al. Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med 2006; 174:171–177.
- Miller RG, Jackson CE, Kasarskis EJ, et al; Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter update: the care of the patient with amyotrophic lateral sclerosis: drug, nutritional, and respiratory therapies (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2009; 73:1218–1226.
- Ambrosino N, Vagheggini G. Noninvasive positive pressure ventilation in the acute care setting: where are we? Eur Respir J 2008; 31:874–886.
- Hill NS, Brennan J, Garpestad E, Nava S. Noninvasive ventilation in acute respiratory failure. Crit Care Med 2007; 35:2402–2407.
- Nava S, Navalesi P, Conti G. Time of non-invasive ventilation. Intensive Care Med 2006; 32:361–370.
- Stoller JK, Kester L, Roberts VT, et al; An analysis of features of respiratory therapy departments that are avid for change. Respir Care 2008; 53:871–884.
- Maheshwari V, Paioli D, Rothaar R, Hill NS. Utilization of noninvasive ventilation in acute care hospitals: a regional survey. Chest 2006; 129:1226–1233.
- Obladen M. History of neonatal resuscitation. Part 1: Artificial ventilation. Neonatology 2008; 94:144–149.
- Poulton EP, Oxon DM. Left-sided heart failure with pulmonary oedema: its treatment with the “pulmonary plus” pressure machine. Lancet 1936; 228:981–983.
- Motley HL, Werko L. Observations on the clinical use of intermittent positive pressure. J Aviat Med 1947; 18:417–435.
- Cournand A, Motley HL. Physiological studies of the effects of intermittent positive pressure breathing on cardiac output in man. Am J Physiol 1948; 152:162–174.
- Severinghaus JW, Astrup P, Murray JF. Blood gas analysis and critical care medicine. Am J Respir Crit Care Med 1998; 157:S114–S122.
- Sullivan CE, Berthon-Jones M, Issa FG. Remission of severe obesity-hypoventilation syndrome after short-term treatment during sleep with nasal continuous positive airway pressure. Am Rev Respir Dis 1983; 128:177–181.
- Ellis ER, Bye PT, Bruderer JW, Sullivan CE. Treatment of respiratory failure during sleep in patients with neuromuscular disease. Positive-pressure ventilation through a nose mask. Am Rev Respir Dis 1987; 135:148–152.
- Sanders MH, Kern N. Obstructive sleep apnea treated by independently adjusted inspiratory and expiratory positive airway pressures via nasal mask. Physiologic and clinical implications. Chest 1990; 98:317–324.
- Bach JR. Mechanical exsufflation, noninvasive ventilation, and new strategies for pulmonary rehabilitation and sleep disordered breathing. Bull N Y Acad Med 1992; 68:321–340.
- Chevrolet JC, Jolliet P, Abajo B, Toussi A, Louis M. Nasal positive pressure ventilation in patients with acute respiratory failure. Difficult and time-consuming procedure for nurses. Chest 1991; 100:775–782.
- Criner GJ, Kreimer DT, Tomaselli M, Pierson W, Evans D. Financial implications of noninvasive positive pressure ventilation (NPPV). Chest 1995; 108:475–481.
- Nava S, Evangelisti I, Rampulla C, Compagnoni ML, Fracchia C, Rubini F. Human and financial costs of noninvasive mechanical ventilation in patients affected by COPD and acute respiratory failure. Chest 1997; 111:1631–1638.
- DiMarco AF, Onders RP, Ignagni A, Kowalski KE, Mortimer JT. Phrenic nerve pacing via intramuscular diaphragm electrodes in tetraplegic subjects. Chest 2005; 127:671–678.
- Antonelli M, Conti G. Noninvasive positive pressure ventilation as treatment for acute respiratory failure in critically ill patients. Crit Care 2000; 4:15–22.
- Hilbert G, Gruson D, Vargas F, et al. Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. N Engl J Med 2001; 344:481–487.
- L’Her E, Deye N, Lellouche F, et al. Physiologic effects of noninvasive ventilation during acute lung injury. Am J Respir Crit Care Med 2005; 172:1112–1118.
- Esteban A, Frutos-Vivar F, Ferguson ND, et al. Noninvasive positive-pressure ventilation for respiratory failure after extubation. N Engl J Med 2004; 350:2452–2460.
- Hill NS. Noninvasive positive pressure ventilation for respiratory failure caused by exacerbations of chronic obstructive pulmonary disease: a standard of care? Crit Care 2003; 7:400–401.
- Lightowler JV, Wedzicha JA, Elliott MW, Ram FS. Non-invasive positive pressure ventilation to treat respiratory failure resulting from exacerbations of chronic obstructive pulmonary disease: Cochrane systematic review and meta-analysis. BMJ 2003; 326:185–187.
- Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995; 333:817–822.
- Keenan SP, Sinuff T, Cook DJ, Hill NS. Which patients with acute exacerbation of chronic obstructive pulmonary disease benefit from noninvasive positive-pressure ventilation? A systematic review of the literature. Ann Intern Med 2003; 138:861–870.
- Epstein SK. Noninvasive ventilation to shorten the duration of mechanical ventilation. Respir Care 2009; 54:198–208.
- Gray A, Goodacre S, Newby DE, Masson M, Sampson F, Nicholl J; 3CPO Trialists. Noninvasive ventilation in acute cardiogenic pulmonary edema. N Engl J Med 2008; 359:142–151.
- Collins SP, Mielniczuk LM, Whittingham HA, Boseley ME, Schramm DR, Storrow AB. The use of noninvasive ventilation in emergency department patients with acute cardiogenic pulmonary edema: a systematic review. Ann Emerg Med 2006; 48:260–269.
- Masip J, Roque M, Sánchez B, Fernández R, Subirana M, Expósito JA. Noninvasive ventilation in acute cardiogenic pulmonary edema: systematic review and meta-analysis. JAMA 2005; 294:3124–3130.
- Baratz DM, Westbrook PR, Shah PK, Mohsenifar Z. Effect of nasal continuous positive airway pressure on cardiac output and oxygen delivery in patients with congestive heart failure. Chest 1992; 102:1397–1401.
- Naughton MT, Rahman MA, Hara K, Floras JS, Bradley TD. Effect of continuous positive airway pressure on intrathoracic and left ventricular transmural pressures in patients with congestive heart failure. Circulation 1995; 91:1725–1731.
- Nava S, Carbone G, DiBattista N, et al. Noninvasive ventilation in cardiogenic pulmonary edema: a multicenter randomized trial. Am J Respir Crit Care Med 2003; 168:1432–1437.
- Mehta S, Jay GD, Woolard RH, et al. Randomized, prospective trial of bilevel versus continuous positive airway pressure in acute pulmonary edema. Crit Care Med 1997; 25:620–628.
- Ho KM, Wong K. A comparison of continuous and bi-level positive airway pressure non-invasive ventilation in patients with acute cardiogenic pulmonary oedema: a meta-analysis. Crit Care 2006; 10:R49.
- Antonelli M, Conti G, Bufi M, et al. Noninvasive ventilation for treatment of acute respiratory failure in patients undergoing solid organ transplantation: a randomized trial. JAMA 2000; 283:235–241.
- Keenan SP, Powers C, McCormack DG, Block G. Noninvasive positive-pressure ventilation for postextubation respiratory distress: a randomized controlled trial. JAMA 2002; 287:3238–3244.
- Ferrer M, Valencia M, Nicolas JM, Bernadich O, Badia JR, Torres A. Early noninvasive ventilation averts extubation failure in patients at risk: a randomized trial. Am J Respir Crit Care Med 2006; 173:164–170.
- Ferrer M, Sellarés J, Valencia M, et al. Non-invasive ventilation after extubation in hypercapnic patients with chronic respiratory disorders: randomised controlled trial. Lancet 2009; 374:1082–1088.
- Nava S, Gregoretti C, Fanfulla F, et al. Noninvasive ventilation to prevent respiratory failure after extubation in high-risk patients. Crit Care Med 2005; 33:2465–2470.
- El-Solh AA, Aquilina A, Pineda L, Dhanvantri V, Grant B, Bouquin P. Noninvasive ventilation for prevention of post-extubation respiratory failure in obese patients. Eur Respir J 2006; 28:588–595.
- Ferrer M, Esquinas A, Arancibia F, et al. Noninvasive ventilation during persistent weaning failure: a randomized controlled trial. Am J Respir Crit Care Med 2003; 168:70–76.
- Girault C, Daudenthun I, Chevron V, Tamion F, Leroy J, Bonmarchand G. Noninvasive ventilation as a systematic extubation and weaning technique in acute-on-chronic respiratory failure: a prospective, randomized controlled study. Am J Respir Crit Care Med 1999; 160:86–92.
- Nava S, Ambrosino N, Clini E, et al. Noninvasive mechanical ventilation in the weaning of patients with respiratory failure due to chronic obstructive pulmonary disease. A randomized, controlled trial. Ann Intern Med 1998; 128:721–728.
- Trevisan CE, Vieira SR; Research Group in Mechanical Ventilation Weaning. Noninvasive mechanical ventilation may be useful in treating patients who fail weaning from invasive mechanical ventilation: a randomized clinical trial. Crit Care 2008; 12:R51.
- Boles JM, Bion J, Connors A, et al. Weaning from mechanical ventilation. Eur Respir J 2007; 29:1033–1056.
- Burns KE, Adhikari NK, Meade MO. A meta-analysis of noninvasive weaning to facilitate liberation from mechanical ventilation. Can J Anaesth 2006; 53:305–315.
- Meduri GU, Cook TR, Turner RE, Cohen M, Leeper KV. Noninvasive positive pressure ventilation in status asthmaticus. Chest 1996; 110:767–774.
- Soroksky A, Stav D, Shpirer I. A pilot prospective, randomized, placebo-controlled trial of bilevel positive airway pressure in acute asthmatic attack. Chest 2003; 123:1018–1025.
- Holley MT, Morrissey TK, Seaberg DC, Afessa B, Wears RL. Ethical dilemmas in a randomized trial of asthma treatment: can Bayesian statistical analysis explain the results? Acad Emerg Med 2001; 8:1128–1135.
- Ram FS, Wellington S, Rowe BH, Wedzicha JA. Non-invasive positive pressure ventilation for treatment of respiratory failure due to severe acute exacerbations of asthma. Cochrane Database Syst Rev 2005;CD004360.
- Medoff BD. Invasive and noninvasive ventilation in patients with asthma. Respir Care 2008; 53:740–748.
- Agarwal R, Reddy C, Aggarwal AN, Gupta D. Is there a role for noninvasive ventilation in acute respiratory distress syndrome? A meta-analysis. Respir Med 2006; 100:2235–2238.
- Delclaux C, L’Her E, Alberti C, et al. Treatment of acute hypoxemic nonhypercapnic respiratory insufficiency with continuous positive airway pressure delivered by a face mask: a randomized controlled trial. JAMA 2000; 284:2352–2360.
- Rana S, Jenad H, Gay PC, Buck CF, Hubmayr RD, Gajic O. Failure of non-invasive ventilation in patients with acute lung injury: observational cohort study. Crit Care 2006; 10:R79.
- Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med 2007; 35:18–25.
- Birnkrant DJ, Ferguson RD, Martin JE, Gordon GJ. Noninvasive ventilation during gastrostomy tube placement in patients with severe Duchenne muscular dystrophy: case reports and review of the literature. Pediatr Pulmonol 2006; 41:188–193.
- Boitano LJ, Jordan T, Benditt JO. Noninvasive ventilation allows gastrostomy tube placement in patients with advanced ALS. Neurology 2001; 56:413–414.
- Maitre B, Jaber S, Maggiore SM, et al. Continuous positive airway pressure during fiberoptic bronchoscopy in hypoxemic patients. A randomized double-blind study using a new device. Am J Respir Crit Care Med 2000; 162:1063–1067.
- Antonelli M, Conti G, Rocco M, et al. Noninvasive positive-pressure ventilation vs conventional oxygen supplementation in hypoxemic patients undergoing diagnostic bronchoscopy. Chest 2002; 121:1149–1154.
- Levy M, Tanios MA, Nelson D, et al. Outcomes of patients with do-not-intubate orders treated with noninvasive ventilation. Crit Care Med 2004; 32:2002–2007.
- Schettino G, Altobelli N, Kacmarek RM. Noninvasive positive pressure ventilation reverses acute respiratory failure in select “do-not-intubate” patients. Crit Care Med 2005; 33:1976–1982.
- Curtis JR, Cook DJ, Sinuff T, et al; Society of Critical Care Medicine Palliative Noninvasive Positive Ventilation Task Force. Noninvasive positive pressure ventilation in critical and palliative care settings: understanding the goals of therapy. Crit Care Med 2007; 35:932–939.
- Baillard C, Fosse JP, Sebbane M, et al. Noninvasive ventilation improves preoxygenation before intubation of hypoxic patients. Am J Respir Crit Care Med 2006; 174:171–177.
- Miller RG, Jackson CE, Kasarskis EJ, et al; Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter update: the care of the patient with amyotrophic lateral sclerosis: drug, nutritional, and respiratory therapies (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2009; 73:1218–1226.
KEY POINTS
- The advantages of NIPPV over invasive ventilation are that it preserves normal physiologic functions such as coughing, swallowing, feeding, and speech and avoids the risks of tracheal and laryngeal injury and respiratory tract infections.
- The best level of evidence for the efficacy of NIPPV is in acute hypercarbic or hypoxemic respiratory failure during exacerbations of chronic obstructive pulmonary disease, in cardiogenic pulmonary edema, and in immunocompromised patients.
- NIPPV should not be applied indiscriminately for lessestablished indications (such as in unconscious patients, respiratory failure after extubation, acute lung injury, or acute respiratory distress syndrome), in severe hypoxemia or acidemia, or after failure to improve dyspnea or gas exchange. The use of NIPPV in these situations may delay a necessary intubation and increase the risks of such a delay, including death.