User login
To the Editor:
Nephrogenic systemic fibrosis (NSF) is an emerging medical entity in patients with renal disease, which results in progressive cutaneous and systemic fibrosis. It is a rare disorder that has been recognized in patients with renal impairment since 2000.1 Patients with NSF demonstrate symmetric dermal and subcutaneous fibrosis evidenced by increasing skin induration on clinical examination. Nephrogenic systemic fibrosis most commonly involves the lower extremities, and after extending to the upper extremities and trunk, it sporadically involves the head and neck.
The clinical manifestation of NSF begins with edema, followed by marked dermal induration, sclerotic plaques, and joint contractures that can lead to considerable disability. Pathogenesis remains to be elucidated; it has been hypothesized that it could be related to gadolinium (Gd). Currently, there is no treatment of this unremitting disease.1-3 We report the case of a patient affected by NSF after administration of Gd for magnetic resonance angiography.
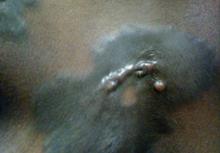
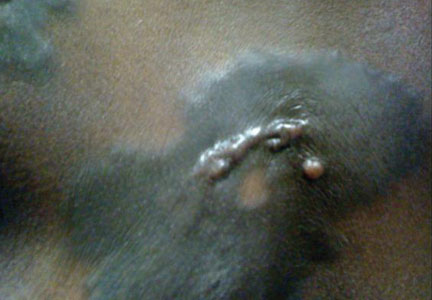
A 56-year-old woman was referred to the department of dermatology at the University of Maryland (Baltimore, Maryland) with persistent swelling of the lower legs, forearms, and trunk of 5 months’ duration. She had end-stage renal disease of nonspecific origin. Five months prior to presentation, she had magnetic resonance angiography, during which 10 mmol of Gd was administered. After, she developed a persistent rash and swelling of the lower legs. On presentation, physical examination revealed symmetric, shiny, pigmented papules and plaques on the forearms, buttocks, thighs, and legs, with no facial involvement (Figure).
A skin biopsy of thigh lesions showed a diffuse dermal proliferation of bland spindle cells associated with dermal fibrosis. A CD4+ cellular infiltrate showed extension into the subcutaneous tissue. Deposition of Gd also was noted in the skin. She was treated with corticosteroid therapy, and after 2 months she reported softening of the affected skin. On 6-month follow-up, her skin lesions did not progress and there was no evidence of systemic involvement. Additionally, renal function had improved.
Nephrogenic systemic fibrosis, also known as nephrogenic fibrosing dermopathy, was first described by Cowper et al1 in 2000. Since then, more than 215 cases have been reported in the literature. Clinically, it is characterized by acute onset of cutaneous hardening and thickening of the extremities and the trunk, often resulting in flexion contractures. There may be varying surface changes such as pigmentation, peau d’orange texture, and shiny sclerosis. Patients often experience unpleasant symptoms such as pain, pruritus, stiffness, and paresthesia. Systemic involvement has been documented in the heart, lung, tendons, muscle, testes, and lamina dura.1-4
Histologic findings of NSF are diffuse dermal fibrosis with increased cellular infiltrates comprised of bland spindled fibrocytes. These fibrocytes express CD34 and type I procollagen. Collagen bundles are thickened but retain clefting, and elastic fibers often are prominent. This fibrotic pattern typically extends to the subcutaneous fat septa, which are widened and collagenous. The epidermis generally is uninvolved. Other findings include dermal mucin deposition, calcification of collagen and vessels, increased CD68+ histiocytes, increased factor XIIIa and dendritic cells, and neoangiogenesis. Rarely, multinucleated giant cells and Miescher radial granulomas with lymphocytic aggregates mimicking erythema nodosum have been described.2-4
Dermatologic entities with similar clinical and histopathologic features, including scleroderma, scleromyxedema, lipodermatosclerosis, erythema nodosum, eosinophilic fasciitis, and spindle cell neoplasms, should be excluded.1-4 The exact pathogenetic mechanisms of NSF have yet to be determined, but there is strong evidence that Gd plays an important causative role.1 In fact, almost all patients with NSF have been exposed to Gd. Gadolinium has been documented in affected skin of patients with NSF and has been shown to induce NSF-like changes in rat models.
Other clinical factors that have been associated with NSF include erythropoietin, elevated serum calcium and phosphate levels, vascular injury or surgery, iron metabolic abnormalities, and metabolic acidosis. It is likely that many factors in the unique physiologic state of patients with renal failure contribute to the abnormal fibrotic reaction to Gd-containing contrast agents in NSF. Gadolinium is a member of a group of 15 elemental metals termed lanthanoids and has been used extensively worldwide in magnetic resonance imaging as a component of intravenously administered contrast agents. Currently, 6 such agents are approved for use in the United States: gadopentetate dimeglumine, gadoteridol, gadodiamide, gadoversetamide, gadobenate dimeglumine, and gadoxetate sodium. All are chelated Gd products, with the chelate serving to prevent toxicity from free Gd ions.
In patients with no renal function abnormalities, the biologic half-life of Gd-based magnetic resonance contrast agents (GBCAs) is 1.5 to 2.0 hours. However, in patients with abnormal kidney function, this half-life is inversely prolonged, proportional to the glomerular filtration rate.5-7 The link between GBCA administration and NSF is compelling, though other etiologic associations have been reported. Surgical or vascular procedures, history of a hypercoagulable state, erythropoietin administration, and immune suppression have been proposed as triggering factors in NSF. The proposed mechanisms responsible for fibrosis in NSF have centered on a collagen-producing cell in the peripheral blood termed the circulating fibrocyte. These cells express CD34 and CD45RO antigens and are capable of producing type I collagen.
Circulating fibrocytes traffic to areas of chronic antigenic stimulation promoting wound repair and fibrotic reactions. Some authors have proposed that materials deposited in the skin might serve as targets for circulating fibrocytes.8 Circulating fibrocytes also are known to produce inflammatory cytokines including IL-1 and chemokines such as platelet-derived growth factor, transforming growth factor b, and others capable of propagating fibrotic responses. Increased expression of transforming growth factor has been reported in dendritic cells in NSF lesions and Parsons et al9 postulated that transglutaminase-2 activation of this protein may be responsible for inciting fibrosis in NSF. Transglutaminases also are known to be directly activated by Gd.10,11
Transmetalation has been proposed as a possible operative phenomenon responsible for NSF. Several cations including zinc, copper, iron, and carbon are known to compete with Gd and may displace it from the ligand, with anions such as OHe, PO4 3e, and CO3 2e binding the resultant free Gd. Some GBCAs contain excess ligand to diminish potential free Gd concentrations. In fact, substantial elevations of serum calcium and phosphorus in patients with NSF have been noted in a large series of patients with NSF. Calciphylaxis, an often catastrophic condition arising in patients with renal failure, has been described in association with NSF, and sodium thiosulfate has been used with success in treating both conditions.10 In addition, Sanyal et al12 noted a substantially higher serum calcium in NSF cases compared with controls.
Gadolinium plays an important role in the pathology of NSF and is confirmed by the presence of Gd in skin biopsies.
1. Cowper SE, Robin HS, Steinberg SM, et al. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet. 2000;356:1000-1001.
2. Girardi M, Kay J, Elston DM, et al. Nephrogenic systemic fibrosis: clinicopathologiocal definition and workup recommendations [published online ahead of print July 2, 2011]. J Am Acad Dermatol. 2011;65:1095-1106.
3. Gupta A, Shamseddin MK, Khaira A. Pathomechanisms of nephrogenic systemic fibrosis: new insights [published online ahead of print July 25, 2011]. Clin Exp Dermatol. 2011;36:763-768.
4. Zou Z, Ma L. Nephrogenic systemic fibrosis: review of 408 biopsy-confirmed cases. Indian J Dermatol. 2011;56:65-73.
5. Pan D, Schmieder AH, Wickline SA, et al. Manganese-based MRI contrast agents: past, present and future. Tetrahedron. 2011;67:8431-8444.
6. Abu-Alfa AK. Nephrogenic systemic fibrosis and gadolinium-based contrast agents. Adv Chronic Kidney Dis. 2011;18:188-198.
7. Wang Y, Alkasab TK, Narin O, et al. Incidence of nephrogenic systemic fibrosis after adoption of restrictive gadolinium-based contrast agent guidelines [published online ahead of print May 17, 2011]. Radiology. 2011;260:105-111.
8. Ortonne N, Lipsker D, Chantrel F, et al. Presence of CD45RO+ CD34+ cells with collagen synthesis activity in nephrogenic fibrosing dermopathy: a new pathogenic hypothesis. Br J Dermatol. 2004;150:1050-1052.
9. Parsons AC, Yosipovitch G, Sheehan DJ, et al. Transglutaminases: the missing link in nephrogenic systemic fibrosis. Am J Dermatopathol. 2007;29:433-436.
10. Wahba IM, Simpson EL, White K. Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature [published online ahead of print August 16, 2007]. Am J Transplant. 2007;7:2425-2432.
11. Goveia M, Chan BP, Patel PR. Evaluating the role of recombinant erythropoietin in nephrogenic systemic fibrosis [published online ahead of print August 8, 2007]. J Am Acad Dermatol. 2007;57:725-727.
12. Sanyal S, Marckmann P, Scherer S, et al. Multiorgan gadolinium (Gd) deposition and fibrosis in a patient with nephrogenic systemic fibrosis–an autopsy-based review [published online ahead of print March 25, 2011]. Nephrol Dial Transplant. 2011;26:3616-3626.
To the Editor:
Nephrogenic systemic fibrosis (NSF) is an emerging medical entity in patients with renal disease, which results in progressive cutaneous and systemic fibrosis. It is a rare disorder that has been recognized in patients with renal impairment since 2000.1 Patients with NSF demonstrate symmetric dermal and subcutaneous fibrosis evidenced by increasing skin induration on clinical examination. Nephrogenic systemic fibrosis most commonly involves the lower extremities, and after extending to the upper extremities and trunk, it sporadically involves the head and neck.
The clinical manifestation of NSF begins with edema, followed by marked dermal induration, sclerotic plaques, and joint contractures that can lead to considerable disability. Pathogenesis remains to be elucidated; it has been hypothesized that it could be related to gadolinium (Gd). Currently, there is no treatment of this unremitting disease.1-3 We report the case of a patient affected by NSF after administration of Gd for magnetic resonance angiography.
A 56-year-old woman was referred to the department of dermatology at the University of Maryland (Baltimore, Maryland) with persistent swelling of the lower legs, forearms, and trunk of 5 months’ duration. She had end-stage renal disease of nonspecific origin. Five months prior to presentation, she had magnetic resonance angiography, during which 10 mmol of Gd was administered. After, she developed a persistent rash and swelling of the lower legs. On presentation, physical examination revealed symmetric, shiny, pigmented papules and plaques on the forearms, buttocks, thighs, and legs, with no facial involvement (Figure).
A skin biopsy of thigh lesions showed a diffuse dermal proliferation of bland spindle cells associated with dermal fibrosis. A CD4+ cellular infiltrate showed extension into the subcutaneous tissue. Deposition of Gd also was noted in the skin. She was treated with corticosteroid therapy, and after 2 months she reported softening of the affected skin. On 6-month follow-up, her skin lesions did not progress and there was no evidence of systemic involvement. Additionally, renal function had improved.
Nephrogenic systemic fibrosis, also known as nephrogenic fibrosing dermopathy, was first described by Cowper et al1 in 2000. Since then, more than 215 cases have been reported in the literature. Clinically, it is characterized by acute onset of cutaneous hardening and thickening of the extremities and the trunk, often resulting in flexion contractures. There may be varying surface changes such as pigmentation, peau d’orange texture, and shiny sclerosis. Patients often experience unpleasant symptoms such as pain, pruritus, stiffness, and paresthesia. Systemic involvement has been documented in the heart, lung, tendons, muscle, testes, and lamina dura.1-4
Histologic findings of NSF are diffuse dermal fibrosis with increased cellular infiltrates comprised of bland spindled fibrocytes. These fibrocytes express CD34 and type I procollagen. Collagen bundles are thickened but retain clefting, and elastic fibers often are prominent. This fibrotic pattern typically extends to the subcutaneous fat septa, which are widened and collagenous. The epidermis generally is uninvolved. Other findings include dermal mucin deposition, calcification of collagen and vessels, increased CD68+ histiocytes, increased factor XIIIa and dendritic cells, and neoangiogenesis. Rarely, multinucleated giant cells and Miescher radial granulomas with lymphocytic aggregates mimicking erythema nodosum have been described.2-4
Dermatologic entities with similar clinical and histopathologic features, including scleroderma, scleromyxedema, lipodermatosclerosis, erythema nodosum, eosinophilic fasciitis, and spindle cell neoplasms, should be excluded.1-4 The exact pathogenetic mechanisms of NSF have yet to be determined, but there is strong evidence that Gd plays an important causative role.1 In fact, almost all patients with NSF have been exposed to Gd. Gadolinium has been documented in affected skin of patients with NSF and has been shown to induce NSF-like changes in rat models.
Other clinical factors that have been associated with NSF include erythropoietin, elevated serum calcium and phosphate levels, vascular injury or surgery, iron metabolic abnormalities, and metabolic acidosis. It is likely that many factors in the unique physiologic state of patients with renal failure contribute to the abnormal fibrotic reaction to Gd-containing contrast agents in NSF. Gadolinium is a member of a group of 15 elemental metals termed lanthanoids and has been used extensively worldwide in magnetic resonance imaging as a component of intravenously administered contrast agents. Currently, 6 such agents are approved for use in the United States: gadopentetate dimeglumine, gadoteridol, gadodiamide, gadoversetamide, gadobenate dimeglumine, and gadoxetate sodium. All are chelated Gd products, with the chelate serving to prevent toxicity from free Gd ions.
In patients with no renal function abnormalities, the biologic half-life of Gd-based magnetic resonance contrast agents (GBCAs) is 1.5 to 2.0 hours. However, in patients with abnormal kidney function, this half-life is inversely prolonged, proportional to the glomerular filtration rate.5-7 The link between GBCA administration and NSF is compelling, though other etiologic associations have been reported. Surgical or vascular procedures, history of a hypercoagulable state, erythropoietin administration, and immune suppression have been proposed as triggering factors in NSF. The proposed mechanisms responsible for fibrosis in NSF have centered on a collagen-producing cell in the peripheral blood termed the circulating fibrocyte. These cells express CD34 and CD45RO antigens and are capable of producing type I collagen.
Circulating fibrocytes traffic to areas of chronic antigenic stimulation promoting wound repair and fibrotic reactions. Some authors have proposed that materials deposited in the skin might serve as targets for circulating fibrocytes.8 Circulating fibrocytes also are known to produce inflammatory cytokines including IL-1 and chemokines such as platelet-derived growth factor, transforming growth factor b, and others capable of propagating fibrotic responses. Increased expression of transforming growth factor has been reported in dendritic cells in NSF lesions and Parsons et al9 postulated that transglutaminase-2 activation of this protein may be responsible for inciting fibrosis in NSF. Transglutaminases also are known to be directly activated by Gd.10,11
Transmetalation has been proposed as a possible operative phenomenon responsible for NSF. Several cations including zinc, copper, iron, and carbon are known to compete with Gd and may displace it from the ligand, with anions such as OHe, PO4 3e, and CO3 2e binding the resultant free Gd. Some GBCAs contain excess ligand to diminish potential free Gd concentrations. In fact, substantial elevations of serum calcium and phosphorus in patients with NSF have been noted in a large series of patients with NSF. Calciphylaxis, an often catastrophic condition arising in patients with renal failure, has been described in association with NSF, and sodium thiosulfate has been used with success in treating both conditions.10 In addition, Sanyal et al12 noted a substantially higher serum calcium in NSF cases compared with controls.
Gadolinium plays an important role in the pathology of NSF and is confirmed by the presence of Gd in skin biopsies.
To the Editor:
Nephrogenic systemic fibrosis (NSF) is an emerging medical entity in patients with renal disease, which results in progressive cutaneous and systemic fibrosis. It is a rare disorder that has been recognized in patients with renal impairment since 2000.1 Patients with NSF demonstrate symmetric dermal and subcutaneous fibrosis evidenced by increasing skin induration on clinical examination. Nephrogenic systemic fibrosis most commonly involves the lower extremities, and after extending to the upper extremities and trunk, it sporadically involves the head and neck.
The clinical manifestation of NSF begins with edema, followed by marked dermal induration, sclerotic plaques, and joint contractures that can lead to considerable disability. Pathogenesis remains to be elucidated; it has been hypothesized that it could be related to gadolinium (Gd). Currently, there is no treatment of this unremitting disease.1-3 We report the case of a patient affected by NSF after administration of Gd for magnetic resonance angiography.
A 56-year-old woman was referred to the department of dermatology at the University of Maryland (Baltimore, Maryland) with persistent swelling of the lower legs, forearms, and trunk of 5 months’ duration. She had end-stage renal disease of nonspecific origin. Five months prior to presentation, she had magnetic resonance angiography, during which 10 mmol of Gd was administered. After, she developed a persistent rash and swelling of the lower legs. On presentation, physical examination revealed symmetric, shiny, pigmented papules and plaques on the forearms, buttocks, thighs, and legs, with no facial involvement (Figure).
A skin biopsy of thigh lesions showed a diffuse dermal proliferation of bland spindle cells associated with dermal fibrosis. A CD4+ cellular infiltrate showed extension into the subcutaneous tissue. Deposition of Gd also was noted in the skin. She was treated with corticosteroid therapy, and after 2 months she reported softening of the affected skin. On 6-month follow-up, her skin lesions did not progress and there was no evidence of systemic involvement. Additionally, renal function had improved.
Nephrogenic systemic fibrosis, also known as nephrogenic fibrosing dermopathy, was first described by Cowper et al1 in 2000. Since then, more than 215 cases have been reported in the literature. Clinically, it is characterized by acute onset of cutaneous hardening and thickening of the extremities and the trunk, often resulting in flexion contractures. There may be varying surface changes such as pigmentation, peau d’orange texture, and shiny sclerosis. Patients often experience unpleasant symptoms such as pain, pruritus, stiffness, and paresthesia. Systemic involvement has been documented in the heart, lung, tendons, muscle, testes, and lamina dura.1-4
Histologic findings of NSF are diffuse dermal fibrosis with increased cellular infiltrates comprised of bland spindled fibrocytes. These fibrocytes express CD34 and type I procollagen. Collagen bundles are thickened but retain clefting, and elastic fibers often are prominent. This fibrotic pattern typically extends to the subcutaneous fat septa, which are widened and collagenous. The epidermis generally is uninvolved. Other findings include dermal mucin deposition, calcification of collagen and vessels, increased CD68+ histiocytes, increased factor XIIIa and dendritic cells, and neoangiogenesis. Rarely, multinucleated giant cells and Miescher radial granulomas with lymphocytic aggregates mimicking erythema nodosum have been described.2-4
Dermatologic entities with similar clinical and histopathologic features, including scleroderma, scleromyxedema, lipodermatosclerosis, erythema nodosum, eosinophilic fasciitis, and spindle cell neoplasms, should be excluded.1-4 The exact pathogenetic mechanisms of NSF have yet to be determined, but there is strong evidence that Gd plays an important causative role.1 In fact, almost all patients with NSF have been exposed to Gd. Gadolinium has been documented in affected skin of patients with NSF and has been shown to induce NSF-like changes in rat models.
Other clinical factors that have been associated with NSF include erythropoietin, elevated serum calcium and phosphate levels, vascular injury or surgery, iron metabolic abnormalities, and metabolic acidosis. It is likely that many factors in the unique physiologic state of patients with renal failure contribute to the abnormal fibrotic reaction to Gd-containing contrast agents in NSF. Gadolinium is a member of a group of 15 elemental metals termed lanthanoids and has been used extensively worldwide in magnetic resonance imaging as a component of intravenously administered contrast agents. Currently, 6 such agents are approved for use in the United States: gadopentetate dimeglumine, gadoteridol, gadodiamide, gadoversetamide, gadobenate dimeglumine, and gadoxetate sodium. All are chelated Gd products, with the chelate serving to prevent toxicity from free Gd ions.
In patients with no renal function abnormalities, the biologic half-life of Gd-based magnetic resonance contrast agents (GBCAs) is 1.5 to 2.0 hours. However, in patients with abnormal kidney function, this half-life is inversely prolonged, proportional to the glomerular filtration rate.5-7 The link between GBCA administration and NSF is compelling, though other etiologic associations have been reported. Surgical or vascular procedures, history of a hypercoagulable state, erythropoietin administration, and immune suppression have been proposed as triggering factors in NSF. The proposed mechanisms responsible for fibrosis in NSF have centered on a collagen-producing cell in the peripheral blood termed the circulating fibrocyte. These cells express CD34 and CD45RO antigens and are capable of producing type I collagen.
Circulating fibrocytes traffic to areas of chronic antigenic stimulation promoting wound repair and fibrotic reactions. Some authors have proposed that materials deposited in the skin might serve as targets for circulating fibrocytes.8 Circulating fibrocytes also are known to produce inflammatory cytokines including IL-1 and chemokines such as platelet-derived growth factor, transforming growth factor b, and others capable of propagating fibrotic responses. Increased expression of transforming growth factor has been reported in dendritic cells in NSF lesions and Parsons et al9 postulated that transglutaminase-2 activation of this protein may be responsible for inciting fibrosis in NSF. Transglutaminases also are known to be directly activated by Gd.10,11
Transmetalation has been proposed as a possible operative phenomenon responsible for NSF. Several cations including zinc, copper, iron, and carbon are known to compete with Gd and may displace it from the ligand, with anions such as OHe, PO4 3e, and CO3 2e binding the resultant free Gd. Some GBCAs contain excess ligand to diminish potential free Gd concentrations. In fact, substantial elevations of serum calcium and phosphorus in patients with NSF have been noted in a large series of patients with NSF. Calciphylaxis, an often catastrophic condition arising in patients with renal failure, has been described in association with NSF, and sodium thiosulfate has been used with success in treating both conditions.10 In addition, Sanyal et al12 noted a substantially higher serum calcium in NSF cases compared with controls.
Gadolinium plays an important role in the pathology of NSF and is confirmed by the presence of Gd in skin biopsies.
1. Cowper SE, Robin HS, Steinberg SM, et al. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet. 2000;356:1000-1001.
2. Girardi M, Kay J, Elston DM, et al. Nephrogenic systemic fibrosis: clinicopathologiocal definition and workup recommendations [published online ahead of print July 2, 2011]. J Am Acad Dermatol. 2011;65:1095-1106.
3. Gupta A, Shamseddin MK, Khaira A. Pathomechanisms of nephrogenic systemic fibrosis: new insights [published online ahead of print July 25, 2011]. Clin Exp Dermatol. 2011;36:763-768.
4. Zou Z, Ma L. Nephrogenic systemic fibrosis: review of 408 biopsy-confirmed cases. Indian J Dermatol. 2011;56:65-73.
5. Pan D, Schmieder AH, Wickline SA, et al. Manganese-based MRI contrast agents: past, present and future. Tetrahedron. 2011;67:8431-8444.
6. Abu-Alfa AK. Nephrogenic systemic fibrosis and gadolinium-based contrast agents. Adv Chronic Kidney Dis. 2011;18:188-198.
7. Wang Y, Alkasab TK, Narin O, et al. Incidence of nephrogenic systemic fibrosis after adoption of restrictive gadolinium-based contrast agent guidelines [published online ahead of print May 17, 2011]. Radiology. 2011;260:105-111.
8. Ortonne N, Lipsker D, Chantrel F, et al. Presence of CD45RO+ CD34+ cells with collagen synthesis activity in nephrogenic fibrosing dermopathy: a new pathogenic hypothesis. Br J Dermatol. 2004;150:1050-1052.
9. Parsons AC, Yosipovitch G, Sheehan DJ, et al. Transglutaminases: the missing link in nephrogenic systemic fibrosis. Am J Dermatopathol. 2007;29:433-436.
10. Wahba IM, Simpson EL, White K. Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature [published online ahead of print August 16, 2007]. Am J Transplant. 2007;7:2425-2432.
11. Goveia M, Chan BP, Patel PR. Evaluating the role of recombinant erythropoietin in nephrogenic systemic fibrosis [published online ahead of print August 8, 2007]. J Am Acad Dermatol. 2007;57:725-727.
12. Sanyal S, Marckmann P, Scherer S, et al. Multiorgan gadolinium (Gd) deposition and fibrosis in a patient with nephrogenic systemic fibrosis–an autopsy-based review [published online ahead of print March 25, 2011]. Nephrol Dial Transplant. 2011;26:3616-3626.
1. Cowper SE, Robin HS, Steinberg SM, et al. Scleromyxoedema-like cutaneous diseases in renal-dialysis patients. Lancet. 2000;356:1000-1001.
2. Girardi M, Kay J, Elston DM, et al. Nephrogenic systemic fibrosis: clinicopathologiocal definition and workup recommendations [published online ahead of print July 2, 2011]. J Am Acad Dermatol. 2011;65:1095-1106.
3. Gupta A, Shamseddin MK, Khaira A. Pathomechanisms of nephrogenic systemic fibrosis: new insights [published online ahead of print July 25, 2011]. Clin Exp Dermatol. 2011;36:763-768.
4. Zou Z, Ma L. Nephrogenic systemic fibrosis: review of 408 biopsy-confirmed cases. Indian J Dermatol. 2011;56:65-73.
5. Pan D, Schmieder AH, Wickline SA, et al. Manganese-based MRI contrast agents: past, present and future. Tetrahedron. 2011;67:8431-8444.
6. Abu-Alfa AK. Nephrogenic systemic fibrosis and gadolinium-based contrast agents. Adv Chronic Kidney Dis. 2011;18:188-198.
7. Wang Y, Alkasab TK, Narin O, et al. Incidence of nephrogenic systemic fibrosis after adoption of restrictive gadolinium-based contrast agent guidelines [published online ahead of print May 17, 2011]. Radiology. 2011;260:105-111.
8. Ortonne N, Lipsker D, Chantrel F, et al. Presence of CD45RO+ CD34+ cells with collagen synthesis activity in nephrogenic fibrosing dermopathy: a new pathogenic hypothesis. Br J Dermatol. 2004;150:1050-1052.
9. Parsons AC, Yosipovitch G, Sheehan DJ, et al. Transglutaminases: the missing link in nephrogenic systemic fibrosis. Am J Dermatopathol. 2007;29:433-436.
10. Wahba IM, Simpson EL, White K. Gadolinium is not the only trigger for nephrogenic systemic fibrosis: insights from two cases and review of the recent literature [published online ahead of print August 16, 2007]. Am J Transplant. 2007;7:2425-2432.
11. Goveia M, Chan BP, Patel PR. Evaluating the role of recombinant erythropoietin in nephrogenic systemic fibrosis [published online ahead of print August 8, 2007]. J Am Acad Dermatol. 2007;57:725-727.
12. Sanyal S, Marckmann P, Scherer S, et al. Multiorgan gadolinium (Gd) deposition and fibrosis in a patient with nephrogenic systemic fibrosis–an autopsy-based review [published online ahead of print March 25, 2011]. Nephrol Dial Transplant. 2011;26:3616-3626.