User login
Dr. Duff reports no financial relationships relevant to this article.
Four important developments have marked the past year in infectious disease:
- A promising vaccine against cytomegalovirus (CMV) was tested in women of reproductive age
- Extended-spectrum antibiotic prophylaxis proved to be effective in reducing the incidence of wound infection following cesarean delivery
- Investigators developed a simple but effective method to prevent wound complications following repair of a third- or fourth-degree perineal laceration
- The incidence of severe Clostridium difficile-associated diarrhea crept upward, emerging as a threat to pregnant women.
CMV vaccine makes an auspicious debut—but isn’t ready for practice
Pass RF, Zhang C, Evans A, et al. Vaccine prevention of maternal cytomegalovirus infection. N Engl J Med. 2009; 360:1191–1199 [Classification of evidence – Level I].
This Phase-2, randomized, double-blind, placebo-controlled trial of a new cytomegalovirus (CMV) vaccine in women found an overall efficacy rate of 50% (95% confidence interval, 7% to 73%), with no unusually serious reactions among women who were vaccinated. This efficacy rate is disappointing, but it isn’t entirely surprising; even the immune response resulting from natural infection is not fully protective against reactivated infection or recurrent infection with a different strain of virus. Nor is natural immunity completely effective in preventing severe fetal injury in recurrent infection.1
Virus poses greatest risk to pregnant women and their fetuses
CMV is the most common perinatally transmitted infection, affecting 0.6% to 0.7% of infants ( FIGURE 1 ). The greatest risk of fetal injury occurs when the mother develops primary infection during pregnancy, which raises her infant’s risk of infection to 40% to 50%. Of infants delivered to mothers with primary infection, approximately 10% to 15% will be acutely symptomatic at birth.
Clinical manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, intracranial calcification, chorioretinitis, hearing impairment, hepatitis, and thrombocytopenia.
Because the morbidity and mortality associated with these conditions are alarmingly high, development of a safe, effective vaccine against CMV would be most welcome.2,3
Recurrent or reactivated maternal CMV infection poses a much lower risk to the fetus. Infected infants are rarely symptomatic at birth. Clinical manifestations of infection typically occur later in childhood and include hearing and visual deficits, dental anomalies, and learning or behavioral disorders.2,3
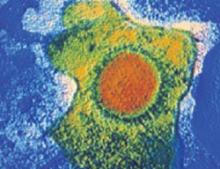
FIGURE 1 Cytomegalovirus
Cytomegalovirus is a member of the herpesvirus family. It is shed intermittently in bodily fluids, without detectable signs and symptoms.
Details of the trial
Women were eligible for the study if they were seronegative for CMV antibody, in good health, 14 to 40 years old, and not pregnant or lactating. Participants received three doses of vaccine or placebo at 0, 1, and 6 months. (The vaccine was composed of CMV envelope glycoprotein B with MF59 adjuvant.) The women were then tested for CMV infection every 3 months for as long as 42 months, using an assay for IgG antibodies directed against viral proteins other than glycoprotein B. Infection was confirmed by viral culture or immuno blotting. The primary endpoint was time until detection of CMV infection.
The vaccine was given to 234 patients, and 230 received placebo. Eighteen infections occurred in the vaccine group, compared with 31 in the placebo group. Vaccinated patients were more likely to remain uninfected during follow-up (p=.02).
One of 81 infants (1%) born to mothers in the vaccinated group had congenital CMV infection, compared with three of 97 (3%) infants born to mothers in the placebo group (p=.41). One infant in the placebo group had severe infection that was evident at birth. The other three infants were asymptomatic at birth and free of sequelae 3 to 5 years later.
The most promising preventive remains experimental
No drug is uniformly effective in treating maternal CMV infection and preventing congenital infection. The most promising intervention for prevention of congenital CMV infection is administration of hyperimmune anti-CMV antibody to the mother. A recent report by Nigro and colleagues4 found this agent to be of great value for both treating and preventing congenital CMV. However, because of limitations in the design of this study, administration of hyperimmune globulin still must be regarded as experimental.3
Key questions remain unanswered
The goal of a large-scale vaccination program is to ensure that women enter reproductive age with preexisting immunity to infection. In that light, the study by Pass and colleagues is only partially encouraging. Despite vaccination, 18 infections occurred, and the follow-up period was relatively short. We do not yet know whether the protective effect of the vaccine will be of extended duration. Moreover, one vaccinated mother delivered an infant who had congenital CMV infection.
Until additional trials of the CMV vaccine are reported, we must focus on helping patients prevent acquisition of infection during pregnancy. Preventive measures include:
- safe sex practices
- use of CMV-negative blood for transfusion to pregnant women and their fetuses
- strict hand-washing procedures for mothers when changing diapers and caring for young children.
Extended-spectrum antibiotics reduce the rate of postcesarean wound infection
Tita ATN, Owen J, Stamm AM, Grimes A, Hauth JC, Andrews WW. Impact of extended-spectrum antibiotic prophylaxis on incidence of postcesarean surgical wound infection. Am J Obstet Gynecol. 2008;199:303e.1–303e.3 [Classification of evidence – Level II].
This prospective study describes surveillance for postcesarean wound infection during three different periods at the University of Alabama:
- 1992–1996, during which patients undergoing cesarean delivery routinely received prophylaxis with a first- or second-generation cephalosporin. Overall incidence of wound infection: 3.1%
- 1997–1999, during which patients were randomized to standard prophylaxis with cefazolin or to cefazolin plus either intravenous (IV) doxycycline or oral azithromycin. Overall incidence of wound infection: 2.4%
- 2001–2006, during which patients routinely received IV cefazolin plus IV azithromycin. Overall incidence of wound infection: 1.3%.
In each time period, the prophylactic antibiotics were administered after the infant’s umbilical cord was clamped. The p value for test of trend was highly significant (p<.002). The same significant trend was noted when superficial and deep wound infections were examined separately.
This evidence is a “practice changer”
For almost 20 years, the standard of practice has been to routinely administer prophylactic antibiotics to all women having cesarean delivery. Essentially, every published study has demonstrated a highly significant reduction in the frequency of postcesarean endometritis when patients received prophylaxis. Multiple studies also confirmed that a more limited-spectrum cephalosporin was as effective as an extended-spectrum agent in reducing the frequency of endometritis.5
Many of these earlier reports were unable to demonstrate a consistently beneficial effect of prophylaxis on the incidence of postoperative wound infection. That is why the present study is of such interest and importance. Tita and colleagues previously demonstrated an improved effect of extended-spectrum prophylaxis on the incidence of postcesarean endometritis.6 Now they have confirmed that this method of prophylaxis is also effective in lowering the rate of surgical wound infection.
Wound infections are more troublesome than endometritis
Wound infections—either incisional abscess or cellulitis—are even more likely than post-cesarean endometritis to prolong a patient’s postoperative stay and create the potential for severe morbidity, such as fascial dehiscence and necrotizing fasciitis. With the increasing prevalence of obesity in the US population, wound infections are likely to become even more frequent.
These infections typically are caused by aerobic streptococci and staphylococci from the skin, combined with coliform organisms and anaerobes from the pelvic flora. Incisional abscesses require surgical drainage; cellulitis usually will respond to a change in antibiotic therapy that specifically targets streptococci and staphylococci, along with the coliforms and anaerobes.
I strongly recommend routine prophylaxis with IV cefazolin (1 g) plus azithromycin (500 mg) in all women having cesarean delivery. Moreover, in view of several recent investigations that evaluated the timing of antibiotic administration (immediately preoperative versus after the umbilical cord is clamped), I recommend that extended-spectrum prophylaxis be given before the start of surgery.7
Duggal N, Mercado C, Daniels K, Bujor A, Caughey AB, El-Sayed YY. Antibiotic prophylaxis for prevention of postpartum perineal wound complications: a randomized control trial. Obstet Gynecol. 2008;111:1268–1273 [Classification of evidence – Level I].
Take note of this prospective, randomized, placebo-controlled trial of prophylactic antibiotics in women who sustained a third-or fourth-degree perineal laceration during vaginal delivery: It is the first, and only, well-designed trial of antibiotic prophylaxis for prevention of complications after repair of a major perineal laceration. Among patients in the study, 8% who received antibiotics developed a wound complication, compared with 24% of patients who received placebo, a statistically and clinically significant difference.
Details of the study
Eighty-three women received placebo, and 64 received a single IV dose of either cefotetan (1 g) or cefoxitin (1 g) before their perineal laceration was repaired. Patients who were allergic to penicillin received clindamycin (900 mg). The primary endpoints of the study were gross disruption of the wound or purulent drainage from the wound site 2 weeks after delivery.
Forty patients (27%) did not return for their post-partum appointment. Of the remaining patients, four of 49 (8%) who received antibiotics developed a wound complication, compared with 14 of 58 (24%) of those who received placebo (p=.037).
Sequelae of major perineal laceration can be severe
Major perineal laceration occurs in approximately 2% to 20% of vaginal deliveries in the United States. The principal risk factors for third- and fourth-degree lacerations are nulliparity, midline episiotomy, and operative vaginal delivery, especially forceps extraction. Both types of laceration may lead to serious morbidity, such as prolonged pain, fecal incontinence, and perineal infection, including necrotizing fasciitis. These infections typically are polymicrobial, mixed aerobic–anaerobic. Moreover, fourth-degree lacerations may result in rectovaginal fistula if they are not repaired properly. This complication causes considerable debility and major social inconvenience for the patient.
Although the sample size was relatively small, this study clearly demonstrated that a single dose of extended-spectrum cephalosporin (cefotetan or cefoxitin) was highly effective in reducing the rate of perineal infection and perineal wound disruption. Whether a more limited-spectrum agent such as cefazolin would be as effective is not certain.
I strongly recommend routine antibiotic prophylaxis for any patient who sustains a third- or fourth-degree perineal laceration.
If the patient has a mild allergy to penicillin (morbilliform rash), I would administer cefotetan because it is less expensive than cefoxitin. If the patient has a severe reaction to penicillin (urticaria, anaphylaxis), I would administer both clindamycin and gentamicin in order to ensure adequate coverage of the multiple organisms likely to cause soft-tissue infection of the genital tract.
Clindamycin alone covers only aerobic gram-positive cocci and offers no protection against the coliform organisms that are so prevalent in perineal wound infection.8
Diarrhea linked to Clostridium difficile emerges as a potent threat to pregnant women
Rouphael NG, O’Donnell JA, Bhatnagar J, et al. Clostridium difficile-associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198:635.e1–635.e6 [Classification of evidence – Level III].
This report describes 10 cases of severe Clostridium difficile-associated diarrhea (CDAD) in pregnant women during 2005 and 2006. CDAD usually affects elderly debilitated patients in hospitals and nursing homes. This report is of great concern because the affected patients were otherwise healthy, young, pregnant women. The observations are even more alarming because the mortality rate in this small series was 30% for both mothers and babies.
Details of the series
The women developed signs of severe CDAD 3 to 60 days after receiving antibiotics; the median was 5 days. CDAD was considered severe if the patient required hospitalization, ICU admission, or colectomy, or if she died. These cases occurred in California, Georgia, Oklahoma, and Pennsylvania and were reported to the Centers for Disease Control and Prevention (CDC).
Six women became ill before delivery, and four developed symptoms postpartum. The most common manifestations of infection were diarrhea, abdominal pain and distention, and fever. The peripheral white blood cell count was in the range of 11–72 × 103/μL. In nine patients, the diagnosis was confirmed by a positive test for toxin A/B; seven of these patients also had visible pseudomembranes in the colon. One patient had a positive stool sample for C difficile ( FIGURE 2 ).
Six patients required admission to the ICU. Six developed toxic megacolon, and five required subtotal colectomy. Three had sepsis; three had acute renal failure; two had disseminated intravascular coagulation. Three patients died, and three had stillbirths. Two patients relapsed following treatment.
One patient had no treatment and died. The others received either metronidazole or vancomycin or a combination of the two. One of the patients who died received metronidazole, vancomycin, and cholestyramine.
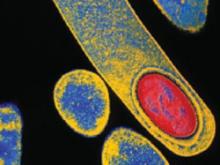
FIGURE 2 Clostridium difficile
C difficile is a spore-forming, gram-positive anaerobic bacillus that is a common cause of antibiotic-associated diarrhea.
An epidemic strain appears
The incidence of C difficile infection in acute care US hospitals has increased to 84 for every 100,000 patients in recent years, about three times the rate of 31 for every 100,000 that was reported in 1996, as the authors note. Many of the most severe cases of CDAD are caused by a new epidemic strain of bacteria, termed North American Pulsed Field type 1 (NAP1) and PCR ribotype 027.
This new strain is characterized by three key virulence factors:
- increased production of toxins A and B
- resistance to fluoroquinolones
- production of binary toxin.
Toxins A and B bind to the surface of intestinal epithelial cells, stimulate tissue injury and inflammation, and, ultimately, lead to cell death. Binary toxin appears to act synergistically with toxins A and B to cause severe colitis.9
Metronidazole is no longer the treatment of choice for severe CDAD
Before 2000, treatment of CDAD with vancomycin or metronidazole was 97% to 98% effective.9 In recent years, however, a failure rate as high as 26% has been reported among patients who are treated with metronidazole.10 One prospective, randomized clinical trial demonstrated that, in patients who had severe CDAD, vancomycin, 125 mg four times daily, was superior to metronidazole, 250 mg four times daily (97% success rate vs 76%; p=.02).11 The efficacy of the two drugs was comparable in treating milder cases of CDAD (98% for vancomycin, 90% for metronidazole; p=.36).
The clinical implications of this case series and the reports cited above are clear: When we administer broad-spectrum antibiotics to our patients, we must be ever watchful for signs of toxicity. If the patient develops diarrhea, the offending drug should be discontinued. If the diarrhea does not promptly resolve, tests to isolate C difficile in the stool and identify toxins unique to this organism are advised.
In addition, anoscopy or sigmoidoscopy should be performed to assess the patient for pseudomembranes.
If mild CDAD is confirmed, the patient may be treated with vancomycin or metronidazole. If severe CDAD is identified, vancomycin should be administered, and the patient should be transferred to the ICU for close monitoring and supportive care.
1. Dekker CL, Arvin AM. One step closer to a CMV vaccine. N Engl J Med. 2009;360:1250-1252.
2. Duff P. Immunotherapy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1402-1404.
3. Nigro G, Adler SP, LaTorre RL, Best AM. Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1350-1362.
4. Duff P, Barth WH, Jr, Post MD. Case records of the Massachusetts General Hospital. Case 4-2009: A 39-year-old pregnant woman with fever after a trip to Africa. N Engl J Med. 2009;360:508-516.
5. Duff P. Prophylactic antibiotics for cesarean delivery: a simple cost-effective strategy for prevention of postoperative morbidity. Am J Obstet Gynecol. 1987;157(4 Pt 1):794-798.
6. Tita AT, Hauth JC, Grimes A, Owen J, Stamm AM, Andrews WW. Decreasing incidence of postcesarean endometritis with extended-spectrum antibiotic prophylaxis. Obstet Gynecol. 2008;111:51-56.
7. Tita ATN, Rouse DJ, Blackwell S, Saade GR, Spong CY, Andrews WW. Emerging concepts in antibiotic prophylaxis for cesarean delivery: a systematic review. Obstet Gynecol. 2009;113:675-682.
8. Duff P. Antibiotic selection in obstetrics: making cost-effective choices. Clin Obstet Gynecol. 2002;45:59-72.
9. Kelly CP, LaMont JT. Clostridium difficile—more difficult than ever. N Engl J Med. 2008;359:1932-1940.
10. Pepin J, Alary ME, Valiquette L, et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40:1591-1597.
11. Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302-307.
Dr. Duff reports no financial relationships relevant to this article.
Four important developments have marked the past year in infectious disease:
- A promising vaccine against cytomegalovirus (CMV) was tested in women of reproductive age
- Extended-spectrum antibiotic prophylaxis proved to be effective in reducing the incidence of wound infection following cesarean delivery
- Investigators developed a simple but effective method to prevent wound complications following repair of a third- or fourth-degree perineal laceration
- The incidence of severe Clostridium difficile-associated diarrhea crept upward, emerging as a threat to pregnant women.
CMV vaccine makes an auspicious debut—but isn’t ready for practice
Pass RF, Zhang C, Evans A, et al. Vaccine prevention of maternal cytomegalovirus infection. N Engl J Med. 2009; 360:1191–1199 [Classification of evidence – Level I].
This Phase-2, randomized, double-blind, placebo-controlled trial of a new cytomegalovirus (CMV) vaccine in women found an overall efficacy rate of 50% (95% confidence interval, 7% to 73%), with no unusually serious reactions among women who were vaccinated. This efficacy rate is disappointing, but it isn’t entirely surprising; even the immune response resulting from natural infection is not fully protective against reactivated infection or recurrent infection with a different strain of virus. Nor is natural immunity completely effective in preventing severe fetal injury in recurrent infection.1
Virus poses greatest risk to pregnant women and their fetuses
CMV is the most common perinatally transmitted infection, affecting 0.6% to 0.7% of infants ( FIGURE 1 ). The greatest risk of fetal injury occurs when the mother develops primary infection during pregnancy, which raises her infant’s risk of infection to 40% to 50%. Of infants delivered to mothers with primary infection, approximately 10% to 15% will be acutely symptomatic at birth.
Clinical manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, intracranial calcification, chorioretinitis, hearing impairment, hepatitis, and thrombocytopenia.
Because the morbidity and mortality associated with these conditions are alarmingly high, development of a safe, effective vaccine against CMV would be most welcome.2,3
Recurrent or reactivated maternal CMV infection poses a much lower risk to the fetus. Infected infants are rarely symptomatic at birth. Clinical manifestations of infection typically occur later in childhood and include hearing and visual deficits, dental anomalies, and learning or behavioral disorders.2,3
FIGURE 1 Cytomegalovirus
Cytomegalovirus is a member of the herpesvirus family. It is shed intermittently in bodily fluids, without detectable signs and symptoms.
Details of the trial
Women were eligible for the study if they were seronegative for CMV antibody, in good health, 14 to 40 years old, and not pregnant or lactating. Participants received three doses of vaccine or placebo at 0, 1, and 6 months. (The vaccine was composed of CMV envelope glycoprotein B with MF59 adjuvant.) The women were then tested for CMV infection every 3 months for as long as 42 months, using an assay for IgG antibodies directed against viral proteins other than glycoprotein B. Infection was confirmed by viral culture or immuno blotting. The primary endpoint was time until detection of CMV infection.
The vaccine was given to 234 patients, and 230 received placebo. Eighteen infections occurred in the vaccine group, compared with 31 in the placebo group. Vaccinated patients were more likely to remain uninfected during follow-up (p=.02).
One of 81 infants (1%) born to mothers in the vaccinated group had congenital CMV infection, compared with three of 97 (3%) infants born to mothers in the placebo group (p=.41). One infant in the placebo group had severe infection that was evident at birth. The other three infants were asymptomatic at birth and free of sequelae 3 to 5 years later.
The most promising preventive remains experimental
No drug is uniformly effective in treating maternal CMV infection and preventing congenital infection. The most promising intervention for prevention of congenital CMV infection is administration of hyperimmune anti-CMV antibody to the mother. A recent report by Nigro and colleagues4 found this agent to be of great value for both treating and preventing congenital CMV. However, because of limitations in the design of this study, administration of hyperimmune globulin still must be regarded as experimental.3
Key questions remain unanswered
The goal of a large-scale vaccination program is to ensure that women enter reproductive age with preexisting immunity to infection. In that light, the study by Pass and colleagues is only partially encouraging. Despite vaccination, 18 infections occurred, and the follow-up period was relatively short. We do not yet know whether the protective effect of the vaccine will be of extended duration. Moreover, one vaccinated mother delivered an infant who had congenital CMV infection.
Until additional trials of the CMV vaccine are reported, we must focus on helping patients prevent acquisition of infection during pregnancy. Preventive measures include:
- safe sex practices
- use of CMV-negative blood for transfusion to pregnant women and their fetuses
- strict hand-washing procedures for mothers when changing diapers and caring for young children.
Extended-spectrum antibiotics reduce the rate of postcesarean wound infection
Tita ATN, Owen J, Stamm AM, Grimes A, Hauth JC, Andrews WW. Impact of extended-spectrum antibiotic prophylaxis on incidence of postcesarean surgical wound infection. Am J Obstet Gynecol. 2008;199:303e.1–303e.3 [Classification of evidence – Level II].
This prospective study describes surveillance for postcesarean wound infection during three different periods at the University of Alabama:
- 1992–1996, during which patients undergoing cesarean delivery routinely received prophylaxis with a first- or second-generation cephalosporin. Overall incidence of wound infection: 3.1%
- 1997–1999, during which patients were randomized to standard prophylaxis with cefazolin or to cefazolin plus either intravenous (IV) doxycycline or oral azithromycin. Overall incidence of wound infection: 2.4%
- 2001–2006, during which patients routinely received IV cefazolin plus IV azithromycin. Overall incidence of wound infection: 1.3%.
In each time period, the prophylactic antibiotics were administered after the infant’s umbilical cord was clamped. The p value for test of trend was highly significant (p<.002). The same significant trend was noted when superficial and deep wound infections were examined separately.
This evidence is a “practice changer”
For almost 20 years, the standard of practice has been to routinely administer prophylactic antibiotics to all women having cesarean delivery. Essentially, every published study has demonstrated a highly significant reduction in the frequency of postcesarean endometritis when patients received prophylaxis. Multiple studies also confirmed that a more limited-spectrum cephalosporin was as effective as an extended-spectrum agent in reducing the frequency of endometritis.5
Many of these earlier reports were unable to demonstrate a consistently beneficial effect of prophylaxis on the incidence of postoperative wound infection. That is why the present study is of such interest and importance. Tita and colleagues previously demonstrated an improved effect of extended-spectrum prophylaxis on the incidence of postcesarean endometritis.6 Now they have confirmed that this method of prophylaxis is also effective in lowering the rate of surgical wound infection.
Wound infections are more troublesome than endometritis
Wound infections—either incisional abscess or cellulitis—are even more likely than post-cesarean endometritis to prolong a patient’s postoperative stay and create the potential for severe morbidity, such as fascial dehiscence and necrotizing fasciitis. With the increasing prevalence of obesity in the US population, wound infections are likely to become even more frequent.
These infections typically are caused by aerobic streptococci and staphylococci from the skin, combined with coliform organisms and anaerobes from the pelvic flora. Incisional abscesses require surgical drainage; cellulitis usually will respond to a change in antibiotic therapy that specifically targets streptococci and staphylococci, along with the coliforms and anaerobes.
I strongly recommend routine prophylaxis with IV cefazolin (1 g) plus azithromycin (500 mg) in all women having cesarean delivery. Moreover, in view of several recent investigations that evaluated the timing of antibiotic administration (immediately preoperative versus after the umbilical cord is clamped), I recommend that extended-spectrum prophylaxis be given before the start of surgery.7
Duggal N, Mercado C, Daniels K, Bujor A, Caughey AB, El-Sayed YY. Antibiotic prophylaxis for prevention of postpartum perineal wound complications: a randomized control trial. Obstet Gynecol. 2008;111:1268–1273 [Classification of evidence – Level I].
Take note of this prospective, randomized, placebo-controlled trial of prophylactic antibiotics in women who sustained a third-or fourth-degree perineal laceration during vaginal delivery: It is the first, and only, well-designed trial of antibiotic prophylaxis for prevention of complications after repair of a major perineal laceration. Among patients in the study, 8% who received antibiotics developed a wound complication, compared with 24% of patients who received placebo, a statistically and clinically significant difference.
Details of the study
Eighty-three women received placebo, and 64 received a single IV dose of either cefotetan (1 g) or cefoxitin (1 g) before their perineal laceration was repaired. Patients who were allergic to penicillin received clindamycin (900 mg). The primary endpoints of the study were gross disruption of the wound or purulent drainage from the wound site 2 weeks after delivery.
Forty patients (27%) did not return for their post-partum appointment. Of the remaining patients, four of 49 (8%) who received antibiotics developed a wound complication, compared with 14 of 58 (24%) of those who received placebo (p=.037).
Sequelae of major perineal laceration can be severe
Major perineal laceration occurs in approximately 2% to 20% of vaginal deliveries in the United States. The principal risk factors for third- and fourth-degree lacerations are nulliparity, midline episiotomy, and operative vaginal delivery, especially forceps extraction. Both types of laceration may lead to serious morbidity, such as prolonged pain, fecal incontinence, and perineal infection, including necrotizing fasciitis. These infections typically are polymicrobial, mixed aerobic–anaerobic. Moreover, fourth-degree lacerations may result in rectovaginal fistula if they are not repaired properly. This complication causes considerable debility and major social inconvenience for the patient.
Although the sample size was relatively small, this study clearly demonstrated that a single dose of extended-spectrum cephalosporin (cefotetan or cefoxitin) was highly effective in reducing the rate of perineal infection and perineal wound disruption. Whether a more limited-spectrum agent such as cefazolin would be as effective is not certain.
I strongly recommend routine antibiotic prophylaxis for any patient who sustains a third- or fourth-degree perineal laceration.
If the patient has a mild allergy to penicillin (morbilliform rash), I would administer cefotetan because it is less expensive than cefoxitin. If the patient has a severe reaction to penicillin (urticaria, anaphylaxis), I would administer both clindamycin and gentamicin in order to ensure adequate coverage of the multiple organisms likely to cause soft-tissue infection of the genital tract.
Clindamycin alone covers only aerobic gram-positive cocci and offers no protection against the coliform organisms that are so prevalent in perineal wound infection.8
Diarrhea linked to Clostridium difficile emerges as a potent threat to pregnant women
Rouphael NG, O’Donnell JA, Bhatnagar J, et al. Clostridium difficile-associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198:635.e1–635.e6 [Classification of evidence – Level III].
This report describes 10 cases of severe Clostridium difficile-associated diarrhea (CDAD) in pregnant women during 2005 and 2006. CDAD usually affects elderly debilitated patients in hospitals and nursing homes. This report is of great concern because the affected patients were otherwise healthy, young, pregnant women. The observations are even more alarming because the mortality rate in this small series was 30% for both mothers and babies.
Details of the series
The women developed signs of severe CDAD 3 to 60 days after receiving antibiotics; the median was 5 days. CDAD was considered severe if the patient required hospitalization, ICU admission, or colectomy, or if she died. These cases occurred in California, Georgia, Oklahoma, and Pennsylvania and were reported to the Centers for Disease Control and Prevention (CDC).
Six women became ill before delivery, and four developed symptoms postpartum. The most common manifestations of infection were diarrhea, abdominal pain and distention, and fever. The peripheral white blood cell count was in the range of 11–72 × 103/μL. In nine patients, the diagnosis was confirmed by a positive test for toxin A/B; seven of these patients also had visible pseudomembranes in the colon. One patient had a positive stool sample for C difficile ( FIGURE 2 ).
Six patients required admission to the ICU. Six developed toxic megacolon, and five required subtotal colectomy. Three had sepsis; three had acute renal failure; two had disseminated intravascular coagulation. Three patients died, and three had stillbirths. Two patients relapsed following treatment.
One patient had no treatment and died. The others received either metronidazole or vancomycin or a combination of the two. One of the patients who died received metronidazole, vancomycin, and cholestyramine.
FIGURE 2 Clostridium difficile
C difficile is a spore-forming, gram-positive anaerobic bacillus that is a common cause of antibiotic-associated diarrhea.
An epidemic strain appears
The incidence of C difficile infection in acute care US hospitals has increased to 84 for every 100,000 patients in recent years, about three times the rate of 31 for every 100,000 that was reported in 1996, as the authors note. Many of the most severe cases of CDAD are caused by a new epidemic strain of bacteria, termed North American Pulsed Field type 1 (NAP1) and PCR ribotype 027.
This new strain is characterized by three key virulence factors:
- increased production of toxins A and B
- resistance to fluoroquinolones
- production of binary toxin.
Toxins A and B bind to the surface of intestinal epithelial cells, stimulate tissue injury and inflammation, and, ultimately, lead to cell death. Binary toxin appears to act synergistically with toxins A and B to cause severe colitis.9
Metronidazole is no longer the treatment of choice for severe CDAD
Before 2000, treatment of CDAD with vancomycin or metronidazole was 97% to 98% effective.9 In recent years, however, a failure rate as high as 26% has been reported among patients who are treated with metronidazole.10 One prospective, randomized clinical trial demonstrated that, in patients who had severe CDAD, vancomycin, 125 mg four times daily, was superior to metronidazole, 250 mg four times daily (97% success rate vs 76%; p=.02).11 The efficacy of the two drugs was comparable in treating milder cases of CDAD (98% for vancomycin, 90% for metronidazole; p=.36).
The clinical implications of this case series and the reports cited above are clear: When we administer broad-spectrum antibiotics to our patients, we must be ever watchful for signs of toxicity. If the patient develops diarrhea, the offending drug should be discontinued. If the diarrhea does not promptly resolve, tests to isolate C difficile in the stool and identify toxins unique to this organism are advised.
In addition, anoscopy or sigmoidoscopy should be performed to assess the patient for pseudomembranes.
If mild CDAD is confirmed, the patient may be treated with vancomycin or metronidazole. If severe CDAD is identified, vancomycin should be administered, and the patient should be transferred to the ICU for close monitoring and supportive care.
Dr. Duff reports no financial relationships relevant to this article.
Four important developments have marked the past year in infectious disease:
- A promising vaccine against cytomegalovirus (CMV) was tested in women of reproductive age
- Extended-spectrum antibiotic prophylaxis proved to be effective in reducing the incidence of wound infection following cesarean delivery
- Investigators developed a simple but effective method to prevent wound complications following repair of a third- or fourth-degree perineal laceration
- The incidence of severe Clostridium difficile-associated diarrhea crept upward, emerging as a threat to pregnant women.
CMV vaccine makes an auspicious debut—but isn’t ready for practice
Pass RF, Zhang C, Evans A, et al. Vaccine prevention of maternal cytomegalovirus infection. N Engl J Med. 2009; 360:1191–1199 [Classification of evidence – Level I].
This Phase-2, randomized, double-blind, placebo-controlled trial of a new cytomegalovirus (CMV) vaccine in women found an overall efficacy rate of 50% (95% confidence interval, 7% to 73%), with no unusually serious reactions among women who were vaccinated. This efficacy rate is disappointing, but it isn’t entirely surprising; even the immune response resulting from natural infection is not fully protective against reactivated infection or recurrent infection with a different strain of virus. Nor is natural immunity completely effective in preventing severe fetal injury in recurrent infection.1
Virus poses greatest risk to pregnant women and their fetuses
CMV is the most common perinatally transmitted infection, affecting 0.6% to 0.7% of infants ( FIGURE 1 ). The greatest risk of fetal injury occurs when the mother develops primary infection during pregnancy, which raises her infant’s risk of infection to 40% to 50%. Of infants delivered to mothers with primary infection, approximately 10% to 15% will be acutely symptomatic at birth.
Clinical manifestations of severe congenital CMV infection include growth restriction, microcephaly, ventriculomegaly, intracranial calcification, chorioretinitis, hearing impairment, hepatitis, and thrombocytopenia.
Because the morbidity and mortality associated with these conditions are alarmingly high, development of a safe, effective vaccine against CMV would be most welcome.2,3
Recurrent or reactivated maternal CMV infection poses a much lower risk to the fetus. Infected infants are rarely symptomatic at birth. Clinical manifestations of infection typically occur later in childhood and include hearing and visual deficits, dental anomalies, and learning or behavioral disorders.2,3
FIGURE 1 Cytomegalovirus
Cytomegalovirus is a member of the herpesvirus family. It is shed intermittently in bodily fluids, without detectable signs and symptoms.
Details of the trial
Women were eligible for the study if they were seronegative for CMV antibody, in good health, 14 to 40 years old, and not pregnant or lactating. Participants received three doses of vaccine or placebo at 0, 1, and 6 months. (The vaccine was composed of CMV envelope glycoprotein B with MF59 adjuvant.) The women were then tested for CMV infection every 3 months for as long as 42 months, using an assay for IgG antibodies directed against viral proteins other than glycoprotein B. Infection was confirmed by viral culture or immuno blotting. The primary endpoint was time until detection of CMV infection.
The vaccine was given to 234 patients, and 230 received placebo. Eighteen infections occurred in the vaccine group, compared with 31 in the placebo group. Vaccinated patients were more likely to remain uninfected during follow-up (p=.02).
One of 81 infants (1%) born to mothers in the vaccinated group had congenital CMV infection, compared with three of 97 (3%) infants born to mothers in the placebo group (p=.41). One infant in the placebo group had severe infection that was evident at birth. The other three infants were asymptomatic at birth and free of sequelae 3 to 5 years later.
The most promising preventive remains experimental
No drug is uniformly effective in treating maternal CMV infection and preventing congenital infection. The most promising intervention for prevention of congenital CMV infection is administration of hyperimmune anti-CMV antibody to the mother. A recent report by Nigro and colleagues4 found this agent to be of great value for both treating and preventing congenital CMV. However, because of limitations in the design of this study, administration of hyperimmune globulin still must be regarded as experimental.3
Key questions remain unanswered
The goal of a large-scale vaccination program is to ensure that women enter reproductive age with preexisting immunity to infection. In that light, the study by Pass and colleagues is only partially encouraging. Despite vaccination, 18 infections occurred, and the follow-up period was relatively short. We do not yet know whether the protective effect of the vaccine will be of extended duration. Moreover, one vaccinated mother delivered an infant who had congenital CMV infection.
Until additional trials of the CMV vaccine are reported, we must focus on helping patients prevent acquisition of infection during pregnancy. Preventive measures include:
- safe sex practices
- use of CMV-negative blood for transfusion to pregnant women and their fetuses
- strict hand-washing procedures for mothers when changing diapers and caring for young children.
Extended-spectrum antibiotics reduce the rate of postcesarean wound infection
Tita ATN, Owen J, Stamm AM, Grimes A, Hauth JC, Andrews WW. Impact of extended-spectrum antibiotic prophylaxis on incidence of postcesarean surgical wound infection. Am J Obstet Gynecol. 2008;199:303e.1–303e.3 [Classification of evidence – Level II].
This prospective study describes surveillance for postcesarean wound infection during three different periods at the University of Alabama:
- 1992–1996, during which patients undergoing cesarean delivery routinely received prophylaxis with a first- or second-generation cephalosporin. Overall incidence of wound infection: 3.1%
- 1997–1999, during which patients were randomized to standard prophylaxis with cefazolin or to cefazolin plus either intravenous (IV) doxycycline or oral azithromycin. Overall incidence of wound infection: 2.4%
- 2001–2006, during which patients routinely received IV cefazolin plus IV azithromycin. Overall incidence of wound infection: 1.3%.
In each time period, the prophylactic antibiotics were administered after the infant’s umbilical cord was clamped. The p value for test of trend was highly significant (p<.002). The same significant trend was noted when superficial and deep wound infections were examined separately.
This evidence is a “practice changer”
For almost 20 years, the standard of practice has been to routinely administer prophylactic antibiotics to all women having cesarean delivery. Essentially, every published study has demonstrated a highly significant reduction in the frequency of postcesarean endometritis when patients received prophylaxis. Multiple studies also confirmed that a more limited-spectrum cephalosporin was as effective as an extended-spectrum agent in reducing the frequency of endometritis.5
Many of these earlier reports were unable to demonstrate a consistently beneficial effect of prophylaxis on the incidence of postoperative wound infection. That is why the present study is of such interest and importance. Tita and colleagues previously demonstrated an improved effect of extended-spectrum prophylaxis on the incidence of postcesarean endometritis.6 Now they have confirmed that this method of prophylaxis is also effective in lowering the rate of surgical wound infection.
Wound infections are more troublesome than endometritis
Wound infections—either incisional abscess or cellulitis—are even more likely than post-cesarean endometritis to prolong a patient’s postoperative stay and create the potential for severe morbidity, such as fascial dehiscence and necrotizing fasciitis. With the increasing prevalence of obesity in the US population, wound infections are likely to become even more frequent.
These infections typically are caused by aerobic streptococci and staphylococci from the skin, combined with coliform organisms and anaerobes from the pelvic flora. Incisional abscesses require surgical drainage; cellulitis usually will respond to a change in antibiotic therapy that specifically targets streptococci and staphylococci, along with the coliforms and anaerobes.
I strongly recommend routine prophylaxis with IV cefazolin (1 g) plus azithromycin (500 mg) in all women having cesarean delivery. Moreover, in view of several recent investigations that evaluated the timing of antibiotic administration (immediately preoperative versus after the umbilical cord is clamped), I recommend that extended-spectrum prophylaxis be given before the start of surgery.7
Duggal N, Mercado C, Daniels K, Bujor A, Caughey AB, El-Sayed YY. Antibiotic prophylaxis for prevention of postpartum perineal wound complications: a randomized control trial. Obstet Gynecol. 2008;111:1268–1273 [Classification of evidence – Level I].
Take note of this prospective, randomized, placebo-controlled trial of prophylactic antibiotics in women who sustained a third-or fourth-degree perineal laceration during vaginal delivery: It is the first, and only, well-designed trial of antibiotic prophylaxis for prevention of complications after repair of a major perineal laceration. Among patients in the study, 8% who received antibiotics developed a wound complication, compared with 24% of patients who received placebo, a statistically and clinically significant difference.
Details of the study
Eighty-three women received placebo, and 64 received a single IV dose of either cefotetan (1 g) or cefoxitin (1 g) before their perineal laceration was repaired. Patients who were allergic to penicillin received clindamycin (900 mg). The primary endpoints of the study were gross disruption of the wound or purulent drainage from the wound site 2 weeks after delivery.
Forty patients (27%) did not return for their post-partum appointment. Of the remaining patients, four of 49 (8%) who received antibiotics developed a wound complication, compared with 14 of 58 (24%) of those who received placebo (p=.037).
Sequelae of major perineal laceration can be severe
Major perineal laceration occurs in approximately 2% to 20% of vaginal deliveries in the United States. The principal risk factors for third- and fourth-degree lacerations are nulliparity, midline episiotomy, and operative vaginal delivery, especially forceps extraction. Both types of laceration may lead to serious morbidity, such as prolonged pain, fecal incontinence, and perineal infection, including necrotizing fasciitis. These infections typically are polymicrobial, mixed aerobic–anaerobic. Moreover, fourth-degree lacerations may result in rectovaginal fistula if they are not repaired properly. This complication causes considerable debility and major social inconvenience for the patient.
Although the sample size was relatively small, this study clearly demonstrated that a single dose of extended-spectrum cephalosporin (cefotetan or cefoxitin) was highly effective in reducing the rate of perineal infection and perineal wound disruption. Whether a more limited-spectrum agent such as cefazolin would be as effective is not certain.
I strongly recommend routine antibiotic prophylaxis for any patient who sustains a third- or fourth-degree perineal laceration.
If the patient has a mild allergy to penicillin (morbilliform rash), I would administer cefotetan because it is less expensive than cefoxitin. If the patient has a severe reaction to penicillin (urticaria, anaphylaxis), I would administer both clindamycin and gentamicin in order to ensure adequate coverage of the multiple organisms likely to cause soft-tissue infection of the genital tract.
Clindamycin alone covers only aerobic gram-positive cocci and offers no protection against the coliform organisms that are so prevalent in perineal wound infection.8
Diarrhea linked to Clostridium difficile emerges as a potent threat to pregnant women
Rouphael NG, O’Donnell JA, Bhatnagar J, et al. Clostridium difficile-associated diarrhea: an emerging threat to pregnant women. Am J Obstet Gynecol. 2008;198:635.e1–635.e6 [Classification of evidence – Level III].
This report describes 10 cases of severe Clostridium difficile-associated diarrhea (CDAD) in pregnant women during 2005 and 2006. CDAD usually affects elderly debilitated patients in hospitals and nursing homes. This report is of great concern because the affected patients were otherwise healthy, young, pregnant women. The observations are even more alarming because the mortality rate in this small series was 30% for both mothers and babies.
Details of the series
The women developed signs of severe CDAD 3 to 60 days after receiving antibiotics; the median was 5 days. CDAD was considered severe if the patient required hospitalization, ICU admission, or colectomy, or if she died. These cases occurred in California, Georgia, Oklahoma, and Pennsylvania and were reported to the Centers for Disease Control and Prevention (CDC).
Six women became ill before delivery, and four developed symptoms postpartum. The most common manifestations of infection were diarrhea, abdominal pain and distention, and fever. The peripheral white blood cell count was in the range of 11–72 × 103/μL. In nine patients, the diagnosis was confirmed by a positive test for toxin A/B; seven of these patients also had visible pseudomembranes in the colon. One patient had a positive stool sample for C difficile ( FIGURE 2 ).
Six patients required admission to the ICU. Six developed toxic megacolon, and five required subtotal colectomy. Three had sepsis; three had acute renal failure; two had disseminated intravascular coagulation. Three patients died, and three had stillbirths. Two patients relapsed following treatment.
One patient had no treatment and died. The others received either metronidazole or vancomycin or a combination of the two. One of the patients who died received metronidazole, vancomycin, and cholestyramine.
FIGURE 2 Clostridium difficile
C difficile is a spore-forming, gram-positive anaerobic bacillus that is a common cause of antibiotic-associated diarrhea.
An epidemic strain appears
The incidence of C difficile infection in acute care US hospitals has increased to 84 for every 100,000 patients in recent years, about three times the rate of 31 for every 100,000 that was reported in 1996, as the authors note. Many of the most severe cases of CDAD are caused by a new epidemic strain of bacteria, termed North American Pulsed Field type 1 (NAP1) and PCR ribotype 027.
This new strain is characterized by three key virulence factors:
- increased production of toxins A and B
- resistance to fluoroquinolones
- production of binary toxin.
Toxins A and B bind to the surface of intestinal epithelial cells, stimulate tissue injury and inflammation, and, ultimately, lead to cell death. Binary toxin appears to act synergistically with toxins A and B to cause severe colitis.9
Metronidazole is no longer the treatment of choice for severe CDAD
Before 2000, treatment of CDAD with vancomycin or metronidazole was 97% to 98% effective.9 In recent years, however, a failure rate as high as 26% has been reported among patients who are treated with metronidazole.10 One prospective, randomized clinical trial demonstrated that, in patients who had severe CDAD, vancomycin, 125 mg four times daily, was superior to metronidazole, 250 mg four times daily (97% success rate vs 76%; p=.02).11 The efficacy of the two drugs was comparable in treating milder cases of CDAD (98% for vancomycin, 90% for metronidazole; p=.36).
The clinical implications of this case series and the reports cited above are clear: When we administer broad-spectrum antibiotics to our patients, we must be ever watchful for signs of toxicity. If the patient develops diarrhea, the offending drug should be discontinued. If the diarrhea does not promptly resolve, tests to isolate C difficile in the stool and identify toxins unique to this organism are advised.
In addition, anoscopy or sigmoidoscopy should be performed to assess the patient for pseudomembranes.
If mild CDAD is confirmed, the patient may be treated with vancomycin or metronidazole. If severe CDAD is identified, vancomycin should be administered, and the patient should be transferred to the ICU for close monitoring and supportive care.
1. Dekker CL, Arvin AM. One step closer to a CMV vaccine. N Engl J Med. 2009;360:1250-1252.
2. Duff P. Immunotherapy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1402-1404.
3. Nigro G, Adler SP, LaTorre RL, Best AM. Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1350-1362.
4. Duff P, Barth WH, Jr, Post MD. Case records of the Massachusetts General Hospital. Case 4-2009: A 39-year-old pregnant woman with fever after a trip to Africa. N Engl J Med. 2009;360:508-516.
5. Duff P. Prophylactic antibiotics for cesarean delivery: a simple cost-effective strategy for prevention of postoperative morbidity. Am J Obstet Gynecol. 1987;157(4 Pt 1):794-798.
6. Tita AT, Hauth JC, Grimes A, Owen J, Stamm AM, Andrews WW. Decreasing incidence of postcesarean endometritis with extended-spectrum antibiotic prophylaxis. Obstet Gynecol. 2008;111:51-56.
7. Tita ATN, Rouse DJ, Blackwell S, Saade GR, Spong CY, Andrews WW. Emerging concepts in antibiotic prophylaxis for cesarean delivery: a systematic review. Obstet Gynecol. 2009;113:675-682.
8. Duff P. Antibiotic selection in obstetrics: making cost-effective choices. Clin Obstet Gynecol. 2002;45:59-72.
9. Kelly CP, LaMont JT. Clostridium difficile—more difficult than ever. N Engl J Med. 2008;359:1932-1940.
10. Pepin J, Alary ME, Valiquette L, et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40:1591-1597.
11. Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302-307.
1. Dekker CL, Arvin AM. One step closer to a CMV vaccine. N Engl J Med. 2009;360:1250-1252.
2. Duff P. Immunotherapy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1402-1404.
3. Nigro G, Adler SP, LaTorre RL, Best AM. Congenital Cytomegalovirus Collaborating Group. Passive immunization during pregnancy for congenital cytomegalovirus infection. N Engl J Med. 2005;353:1350-1362.
4. Duff P, Barth WH, Jr, Post MD. Case records of the Massachusetts General Hospital. Case 4-2009: A 39-year-old pregnant woman with fever after a trip to Africa. N Engl J Med. 2009;360:508-516.
5. Duff P. Prophylactic antibiotics for cesarean delivery: a simple cost-effective strategy for prevention of postoperative morbidity. Am J Obstet Gynecol. 1987;157(4 Pt 1):794-798.
6. Tita AT, Hauth JC, Grimes A, Owen J, Stamm AM, Andrews WW. Decreasing incidence of postcesarean endometritis with extended-spectrum antibiotic prophylaxis. Obstet Gynecol. 2008;111:51-56.
7. Tita ATN, Rouse DJ, Blackwell S, Saade GR, Spong CY, Andrews WW. Emerging concepts in antibiotic prophylaxis for cesarean delivery: a systematic review. Obstet Gynecol. 2009;113:675-682.
8. Duff P. Antibiotic selection in obstetrics: making cost-effective choices. Clin Obstet Gynecol. 2002;45:59-72.
9. Kelly CP, LaMont JT. Clostridium difficile—more difficult than ever. N Engl J Med. 2008;359:1932-1940.
10. Pepin J, Alary ME, Valiquette L, et al. Increasing risk of relapse after treatment of Clostridium difficile colitis in Quebec, Canada. Clin Infect Dis. 2005;40:1591-1597.
11. Zar FA, Bakkanagari SR, Moorthi KM, Davis MB. A comparison of vancomycin and metronidazole for the treatment of Clostridium difficile-associated diarrhea, stratified by disease severity. Clin Infect Dis. 2007;45:302-307.