User login
Kidney Transplantation: Who is Eligible?
For many years, the medical community speculated about the possibility of organ transplantation. The first successful transplant of any kind involving humans was a corneal transplant in 1905.1
It wasn’t until 1954 that the first successful organ transplant, a kidney transplant between identical twins, occurred.2 Several new concepts emerged: organ rejection plays a major role in the failure or success of a transplant; and donors and recipients must be matched based on blood group.
Today, about 169,000 people in the US live with a donated kidney. Each year, some 10,500 cadaveric organs are transplanted, and 6,400 donors are living donors.3 The National Kidney Foundation’s recent 10-year initiative, End the Wait!,4 seeks to close the gap between the more than 50,000 people on the transplant waiting list3 and the number of available donor organs.
Since many patients live for years with their transplanted organs, the primary care clinician is likely to see transplant recipients in a family practice or internal medicine setting. While each patient has unique needs, there are commonalities among them.
Renal Consult welcomes any additional comments or questions regarding care of the renal patient. Please address them to editor@clinicianreviews.com.
Jane S. Davis, CRNP, DNP
Q: I have a 70-year-old male patient who is losing kidney function. He asked me about transplantation, but I really don’t know whether he is eligible to get on the list. Who is eligible? Is there an age limit? Are patients with chronic illnesses (hepatitis B, hepatitis C, HIV) eligible? How long is the list? Where can I find these answers?
There are no specific guidelines regarding eligibility or age restrictions for kidney transplantation in the United States. Most transplant centers look at patients older than 65 a little more carefully than younger patients—they have to be in good health apart from their renal disease. Some centers will not transplant patients older than 70, while others transplant patients who are 80 or older.15 The best thing to do is to refer the patient to the local center or call and find out. Again, the Organ Procurement and Transplantation Network,7 which lists transplant centers and contact information, can be accessed at optn.transplant.hrsa.gov/mem bers/search.asp
Chronic illnesses are not automatic rule-outs for the most part. Very few centers transplant HIV-positive patients, but this does occur, especially in major cities with a large population of persons with HIV (eg, Washington, DC; San Francisco, New York City, Cincinnati). An infectious disease specialist must follow these patients after transplantation and adjust their HAART (highly active antiretroviral therapy) medications to compensate for both the decreased renal function and anti-rejection medications. Hepatitis B and C patients are often accepted as long as liver biopsy shows no cirrhosis and the viral load is low or manageable. If the patient is found to have cirrhosis or decompensation, a combined liver-kidney transplant can be planned, although the success rate of this procedure is low.16,17
Patients with certain types of hepatitis C may be eligible to receive a kidney from a donor with hepatitis C18 in order to shorten the wait time and make use of a kidney that cannot be transplanted into a person not infected with hepatitis C.
Transplant waiting lists vary by region across the country. There is a centralized electronic list managed by UNOS, on which eligible recipients are placed once they have been approved by the transplant center, following the medical work-up and acceptance by the transplant committee at each center. This is referred to as “being listed” or “on the list.” Patients begin to accrue waiting time as soon as they are added, and this list is precise to the second! There is a list for each blood type, with its own set of waiting times in each region.20 Average waiting times, by blood type (ie, ABO), can be searched at www.ustransplant.org/Calcula tors/KidneyWaitTime.aspx
When a donor organ becomes available and has been evaluated by the procurement team, the donor’s information is entered into the system and the computer generates a list of eligible candidates, based on a variety of factors. This is called a “match-run.”
Waiting time is the most important factor, but consideration is given to patients younger than 18, those who have previously donated an organ, and those with high antibody levels (ie, panel-reactive antibodies, or PRAs). Patients in the latter group may find it more difficult to locate a compatible donor, as these patients have been sensitized as a result of prior transplantation, pregnancy, or blood transfusions. It is very rare for a patient to be a perfect match (0 mismatch), but should the right organ become available, the matched patient receives priority consideration.
Besides the ABO match, human leukocyte antigen (HLA) matching of six main HLA antibodies is done. Within each of these six antibodies (HLA-A, B, C, DP, DR, DQ), subgroup matching is also done because some HLA subgroups are more highly correlated with rejection than others.21,22 A more complete explanation of organ matching and allocation can be found on the “Transplant Living” Web site: www.transplantliving.org/beforethetransplant/allocation/matchingorgans.aspx.
Patients should be encouraged to access “Transplant Living” (www.transplantliving.org) and UNOS for information and links. Additional information about transplantation, eligibility, performance statistics, policies, procedures, and other questions and answers, for both clinicians and patients, can be found on the Organ Procurement and Transplantation Network Web site (optn.transplant.hrsa.gov).
Annette Needham, MSN, ARNP, NP-C, CNN-NP, CCTC, Florida Hospital Transplant Center, Orlando
References
1. Armitage WJ, Tullo AB, Larkin DFP. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222-1223.
2. Kidney transplantation: past, present, and future. www.stanford.edu/dept/HPS/transplant/html/history.html. Accessed September 16, 2011.
3. United States Renal Data System. Atlas. www .usrds.org/atlas.htm. Accessed September 16, 2011.
4. National Kidney Foundation. End the wait! www.kidney.org/news/end_the_wait/index.cfm. Accessed September 16, 2011.
5. National Kidney Foundation. Kidney transplant (2011). www.kidney.org/atoz/content/kidneytransnewlease.cfm. Accessed September 16, 2011.
6. United States Renal Data Systems. Presentations and posters (2000-2011). www.usrds.org/presentations.htm. Accessed September 16, 2011.
7. Organ Procurement and Transplantation Network. Members: member directory. optn.transplant.hrsa.gov/members/search.asp. Accessed September 16, 2011.
8. Jensen P, Møller B, Hansen S. Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens. J Am Acad Dermatol. 2000;42(2 pt 1):307.
9. Wong G, Chapman JR, Craig JC. Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol. 2008;3 suppl 2:S87-S100.
10. Parker A, Bowles K, Bradley JA, et al; Haemato-oncology subgroup of the British Committee for Standards in Haematology and the British Transplantation Society. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant patients. Br J Haematol. 2010;149(5):675-692.
11. Transplant Living. After the transplant (2011). www.transplantliving.org/afterthetransplant/default.aspx. Accessed September 16, 2011.
12. United Network for Organ Sharing. www.unos.org. Accessed September 16, 2011.
13. Kidney Disease Improving Global Outcomes. Managing your adult patients who have a kidney transplant (2010). www.kidney.org/professionals/tools/pdf/02-50-4079_ABB_ManagingTransRecip Bk_PC.pdf. Accessed September 16, 2011.
14. Abbud-Filho M, Adams P, Alberu J, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation. 2007; (Suppl 8):83:1-22.
15. Heldal K, Hartmann A, Leivestad T, et al. Risk variables associated with the outcome of kidney recipients >70 years of age in the new millennium. Nephrol Dial Transplant. 2011;26(8):2706-2711.
16. Chava SP, Singh B, Stangou A, et al. Simultaneous combined liver and kidney transplantation: a single center experience. Clin Transplant. 2010; 24(3):E62-E68.
17. Ruiz R, Kunitake H, Wilkinson AH, et al. Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg. 2006;141 (8):735-741.
18. Veroux P, Veroux M, Puliatti C, et al. Kidney transplantation from hepatitis C virus-positive donors into hepatitis C virus-positive recipients: a safe way to expand the donor pool? Transplant Proc. 2005;37(6):2571-2573.
19. United States Renal Data Systems, Annual Data Reports. National Kidney and Urologic Disease Information Clearinghouse. Figure 6ii. Transplant (kidney only) wait list and wait times. www.usrds.org/2010/pdf/v2_07.pdf. Accessed September 16, 2011.
20. Arbor Research Collaborative for Health. Kidney waiting time calculator. www.ustransplant.org/Calculators/KidneyWaitTime.aspx. Accessed September 16, 2011.
21. Karakayali FY, Ozdemir H, Kivrakdal S, et al. Recurrent glomerular diseases after renal transplantation. Transplant Proc. 2006;38(2):470-472.
22. Nojima M, Ichikawa Y, Ihara H, et al. Significant effect of HLA-DRB1 matching on acute rejection of kidney transplants within 3 months. Transplant Proc. 2001;33(1-2):1182-1184.
For many years, the medical community speculated about the possibility of organ transplantation. The first successful transplant of any kind involving humans was a corneal transplant in 1905.1
It wasn’t until 1954 that the first successful organ transplant, a kidney transplant between identical twins, occurred.2 Several new concepts emerged: organ rejection plays a major role in the failure or success of a transplant; and donors and recipients must be matched based on blood group.
Today, about 169,000 people in the US live with a donated kidney. Each year, some 10,500 cadaveric organs are transplanted, and 6,400 donors are living donors.3 The National Kidney Foundation’s recent 10-year initiative, End the Wait!,4 seeks to close the gap between the more than 50,000 people on the transplant waiting list3 and the number of available donor organs.
Since many patients live for years with their transplanted organs, the primary care clinician is likely to see transplant recipients in a family practice or internal medicine setting. While each patient has unique needs, there are commonalities among them.
Renal Consult welcomes any additional comments or questions regarding care of the renal patient. Please address them to editor@clinicianreviews.com.
Jane S. Davis, CRNP, DNP
Q: I have a 70-year-old male patient who is losing kidney function. He asked me about transplantation, but I really don’t know whether he is eligible to get on the list. Who is eligible? Is there an age limit? Are patients with chronic illnesses (hepatitis B, hepatitis C, HIV) eligible? How long is the list? Where can I find these answers?
There are no specific guidelines regarding eligibility or age restrictions for kidney transplantation in the United States. Most transplant centers look at patients older than 65 a little more carefully than younger patients—they have to be in good health apart from their renal disease. Some centers will not transplant patients older than 70, while others transplant patients who are 80 or older.15 The best thing to do is to refer the patient to the local center or call and find out. Again, the Organ Procurement and Transplantation Network,7 which lists transplant centers and contact information, can be accessed at optn.transplant.hrsa.gov/mem bers/search.asp
Chronic illnesses are not automatic rule-outs for the most part. Very few centers transplant HIV-positive patients, but this does occur, especially in major cities with a large population of persons with HIV (eg, Washington, DC; San Francisco, New York City, Cincinnati). An infectious disease specialist must follow these patients after transplantation and adjust their HAART (highly active antiretroviral therapy) medications to compensate for both the decreased renal function and anti-rejection medications. Hepatitis B and C patients are often accepted as long as liver biopsy shows no cirrhosis and the viral load is low or manageable. If the patient is found to have cirrhosis or decompensation, a combined liver-kidney transplant can be planned, although the success rate of this procedure is low.16,17
Patients with certain types of hepatitis C may be eligible to receive a kidney from a donor with hepatitis C18 in order to shorten the wait time and make use of a kidney that cannot be transplanted into a person not infected with hepatitis C.
Transplant waiting lists vary by region across the country. There is a centralized electronic list managed by UNOS, on which eligible recipients are placed once they have been approved by the transplant center, following the medical work-up and acceptance by the transplant committee at each center. This is referred to as “being listed” or “on the list.” Patients begin to accrue waiting time as soon as they are added, and this list is precise to the second! There is a list for each blood type, with its own set of waiting times in each region.20 Average waiting times, by blood type (ie, ABO), can be searched at www.ustransplant.org/Calcula tors/KidneyWaitTime.aspx
When a donor organ becomes available and has been evaluated by the procurement team, the donor’s information is entered into the system and the computer generates a list of eligible candidates, based on a variety of factors. This is called a “match-run.”
Waiting time is the most important factor, but consideration is given to patients younger than 18, those who have previously donated an organ, and those with high antibody levels (ie, panel-reactive antibodies, or PRAs). Patients in the latter group may find it more difficult to locate a compatible donor, as these patients have been sensitized as a result of prior transplantation, pregnancy, or blood transfusions. It is very rare for a patient to be a perfect match (0 mismatch), but should the right organ become available, the matched patient receives priority consideration.
Besides the ABO match, human leukocyte antigen (HLA) matching of six main HLA antibodies is done. Within each of these six antibodies (HLA-A, B, C, DP, DR, DQ), subgroup matching is also done because some HLA subgroups are more highly correlated with rejection than others.21,22 A more complete explanation of organ matching and allocation can be found on the “Transplant Living” Web site: www.transplantliving.org/beforethetransplant/allocation/matchingorgans.aspx.
Patients should be encouraged to access “Transplant Living” (www.transplantliving.org) and UNOS for information and links. Additional information about transplantation, eligibility, performance statistics, policies, procedures, and other questions and answers, for both clinicians and patients, can be found on the Organ Procurement and Transplantation Network Web site (optn.transplant.hrsa.gov).
Annette Needham, MSN, ARNP, NP-C, CNN-NP, CCTC, Florida Hospital Transplant Center, Orlando
References
1. Armitage WJ, Tullo AB, Larkin DFP. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222-1223.
2. Kidney transplantation: past, present, and future. www.stanford.edu/dept/HPS/transplant/html/history.html. Accessed September 16, 2011.
3. United States Renal Data System. Atlas. www .usrds.org/atlas.htm. Accessed September 16, 2011.
4. National Kidney Foundation. End the wait! www.kidney.org/news/end_the_wait/index.cfm. Accessed September 16, 2011.
5. National Kidney Foundation. Kidney transplant (2011). www.kidney.org/atoz/content/kidneytransnewlease.cfm. Accessed September 16, 2011.
6. United States Renal Data Systems. Presentations and posters (2000-2011). www.usrds.org/presentations.htm. Accessed September 16, 2011.
7. Organ Procurement and Transplantation Network. Members: member directory. optn.transplant.hrsa.gov/members/search.asp. Accessed September 16, 2011.
8. Jensen P, Møller B, Hansen S. Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens. J Am Acad Dermatol. 2000;42(2 pt 1):307.
9. Wong G, Chapman JR, Craig JC. Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol. 2008;3 suppl 2:S87-S100.
10. Parker A, Bowles K, Bradley JA, et al; Haemato-oncology subgroup of the British Committee for Standards in Haematology and the British Transplantation Society. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant patients. Br J Haematol. 2010;149(5):675-692.
11. Transplant Living. After the transplant (2011). www.transplantliving.org/afterthetransplant/default.aspx. Accessed September 16, 2011.
12. United Network for Organ Sharing. www.unos.org. Accessed September 16, 2011.
13. Kidney Disease Improving Global Outcomes. Managing your adult patients who have a kidney transplant (2010). www.kidney.org/professionals/tools/pdf/02-50-4079_ABB_ManagingTransRecip Bk_PC.pdf. Accessed September 16, 2011.
14. Abbud-Filho M, Adams P, Alberu J, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation. 2007; (Suppl 8):83:1-22.
15. Heldal K, Hartmann A, Leivestad T, et al. Risk variables associated with the outcome of kidney recipients >70 years of age in the new millennium. Nephrol Dial Transplant. 2011;26(8):2706-2711.
16. Chava SP, Singh B, Stangou A, et al. Simultaneous combined liver and kidney transplantation: a single center experience. Clin Transplant. 2010; 24(3):E62-E68.
17. Ruiz R, Kunitake H, Wilkinson AH, et al. Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg. 2006;141 (8):735-741.
18. Veroux P, Veroux M, Puliatti C, et al. Kidney transplantation from hepatitis C virus-positive donors into hepatitis C virus-positive recipients: a safe way to expand the donor pool? Transplant Proc. 2005;37(6):2571-2573.
19. United States Renal Data Systems, Annual Data Reports. National Kidney and Urologic Disease Information Clearinghouse. Figure 6ii. Transplant (kidney only) wait list and wait times. www.usrds.org/2010/pdf/v2_07.pdf. Accessed September 16, 2011.
20. Arbor Research Collaborative for Health. Kidney waiting time calculator. www.ustransplant.org/Calculators/KidneyWaitTime.aspx. Accessed September 16, 2011.
21. Karakayali FY, Ozdemir H, Kivrakdal S, et al. Recurrent glomerular diseases after renal transplantation. Transplant Proc. 2006;38(2):470-472.
22. Nojima M, Ichikawa Y, Ihara H, et al. Significant effect of HLA-DRB1 matching on acute rejection of kidney transplants within 3 months. Transplant Proc. 2001;33(1-2):1182-1184.
For many years, the medical community speculated about the possibility of organ transplantation. The first successful transplant of any kind involving humans was a corneal transplant in 1905.1
It wasn’t until 1954 that the first successful organ transplant, a kidney transplant between identical twins, occurred.2 Several new concepts emerged: organ rejection plays a major role in the failure or success of a transplant; and donors and recipients must be matched based on blood group.
Today, about 169,000 people in the US live with a donated kidney. Each year, some 10,500 cadaveric organs are transplanted, and 6,400 donors are living donors.3 The National Kidney Foundation’s recent 10-year initiative, End the Wait!,4 seeks to close the gap between the more than 50,000 people on the transplant waiting list3 and the number of available donor organs.
Since many patients live for years with their transplanted organs, the primary care clinician is likely to see transplant recipients in a family practice or internal medicine setting. While each patient has unique needs, there are commonalities among them.
Renal Consult welcomes any additional comments or questions regarding care of the renal patient. Please address them to editor@clinicianreviews.com.
Jane S. Davis, CRNP, DNP
Q: I have a 70-year-old male patient who is losing kidney function. He asked me about transplantation, but I really don’t know whether he is eligible to get on the list. Who is eligible? Is there an age limit? Are patients with chronic illnesses (hepatitis B, hepatitis C, HIV) eligible? How long is the list? Where can I find these answers?
There are no specific guidelines regarding eligibility or age restrictions for kidney transplantation in the United States. Most transplant centers look at patients older than 65 a little more carefully than younger patients—they have to be in good health apart from their renal disease. Some centers will not transplant patients older than 70, while others transplant patients who are 80 or older.15 The best thing to do is to refer the patient to the local center or call and find out. Again, the Organ Procurement and Transplantation Network,7 which lists transplant centers and contact information, can be accessed at optn.transplant.hrsa.gov/mem bers/search.asp
Chronic illnesses are not automatic rule-outs for the most part. Very few centers transplant HIV-positive patients, but this does occur, especially in major cities with a large population of persons with HIV (eg, Washington, DC; San Francisco, New York City, Cincinnati). An infectious disease specialist must follow these patients after transplantation and adjust their HAART (highly active antiretroviral therapy) medications to compensate for both the decreased renal function and anti-rejection medications. Hepatitis B and C patients are often accepted as long as liver biopsy shows no cirrhosis and the viral load is low or manageable. If the patient is found to have cirrhosis or decompensation, a combined liver-kidney transplant can be planned, although the success rate of this procedure is low.16,17
Patients with certain types of hepatitis C may be eligible to receive a kidney from a donor with hepatitis C18 in order to shorten the wait time and make use of a kidney that cannot be transplanted into a person not infected with hepatitis C.
Transplant waiting lists vary by region across the country. There is a centralized electronic list managed by UNOS, on which eligible recipients are placed once they have been approved by the transplant center, following the medical work-up and acceptance by the transplant committee at each center. This is referred to as “being listed” or “on the list.” Patients begin to accrue waiting time as soon as they are added, and this list is precise to the second! There is a list for each blood type, with its own set of waiting times in each region.20 Average waiting times, by blood type (ie, ABO), can be searched at www.ustransplant.org/Calcula tors/KidneyWaitTime.aspx
When a donor organ becomes available and has been evaluated by the procurement team, the donor’s information is entered into the system and the computer generates a list of eligible candidates, based on a variety of factors. This is called a “match-run.”
Waiting time is the most important factor, but consideration is given to patients younger than 18, those who have previously donated an organ, and those with high antibody levels (ie, panel-reactive antibodies, or PRAs). Patients in the latter group may find it more difficult to locate a compatible donor, as these patients have been sensitized as a result of prior transplantation, pregnancy, or blood transfusions. It is very rare for a patient to be a perfect match (0 mismatch), but should the right organ become available, the matched patient receives priority consideration.
Besides the ABO match, human leukocyte antigen (HLA) matching of six main HLA antibodies is done. Within each of these six antibodies (HLA-A, B, C, DP, DR, DQ), subgroup matching is also done because some HLA subgroups are more highly correlated with rejection than others.21,22 A more complete explanation of organ matching and allocation can be found on the “Transplant Living” Web site: www.transplantliving.org/beforethetransplant/allocation/matchingorgans.aspx.
Patients should be encouraged to access “Transplant Living” (www.transplantliving.org) and UNOS for information and links. Additional information about transplantation, eligibility, performance statistics, policies, procedures, and other questions and answers, for both clinicians and patients, can be found on the Organ Procurement and Transplantation Network Web site (optn.transplant.hrsa.gov).
Annette Needham, MSN, ARNP, NP-C, CNN-NP, CCTC, Florida Hospital Transplant Center, Orlando
References
1. Armitage WJ, Tullo AB, Larkin DFP. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222-1223.
2. Kidney transplantation: past, present, and future. www.stanford.edu/dept/HPS/transplant/html/history.html. Accessed September 16, 2011.
3. United States Renal Data System. Atlas. www .usrds.org/atlas.htm. Accessed September 16, 2011.
4. National Kidney Foundation. End the wait! www.kidney.org/news/end_the_wait/index.cfm. Accessed September 16, 2011.
5. National Kidney Foundation. Kidney transplant (2011). www.kidney.org/atoz/content/kidneytransnewlease.cfm. Accessed September 16, 2011.
6. United States Renal Data Systems. Presentations and posters (2000-2011). www.usrds.org/presentations.htm. Accessed September 16, 2011.
7. Organ Procurement and Transplantation Network. Members: member directory. optn.transplant.hrsa.gov/members/search.asp. Accessed September 16, 2011.
8. Jensen P, Møller B, Hansen S. Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens. J Am Acad Dermatol. 2000;42(2 pt 1):307.
9. Wong G, Chapman JR, Craig JC. Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol. 2008;3 suppl 2:S87-S100.
10. Parker A, Bowles K, Bradley JA, et al; Haemato-oncology subgroup of the British Committee for Standards in Haematology and the British Transplantation Society. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant patients. Br J Haematol. 2010;149(5):675-692.
11. Transplant Living. After the transplant (2011). www.transplantliving.org/afterthetransplant/default.aspx. Accessed September 16, 2011.
12. United Network for Organ Sharing. www.unos.org. Accessed September 16, 2011.
13. Kidney Disease Improving Global Outcomes. Managing your adult patients who have a kidney transplant (2010). www.kidney.org/professionals/tools/pdf/02-50-4079_ABB_ManagingTransRecip Bk_PC.pdf. Accessed September 16, 2011.
14. Abbud-Filho M, Adams P, Alberu J, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation. 2007; (Suppl 8):83:1-22.
15. Heldal K, Hartmann A, Leivestad T, et al. Risk variables associated with the outcome of kidney recipients >70 years of age in the new millennium. Nephrol Dial Transplant. 2011;26(8):2706-2711.
16. Chava SP, Singh B, Stangou A, et al. Simultaneous combined liver and kidney transplantation: a single center experience. Clin Transplant. 2010; 24(3):E62-E68.
17. Ruiz R, Kunitake H, Wilkinson AH, et al. Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg. 2006;141 (8):735-741.
18. Veroux P, Veroux M, Puliatti C, et al. Kidney transplantation from hepatitis C virus-positive donors into hepatitis C virus-positive recipients: a safe way to expand the donor pool? Transplant Proc. 2005;37(6):2571-2573.
19. United States Renal Data Systems, Annual Data Reports. National Kidney and Urologic Disease Information Clearinghouse. Figure 6ii. Transplant (kidney only) wait list and wait times. www.usrds.org/2010/pdf/v2_07.pdf. Accessed September 16, 2011.
20. Arbor Research Collaborative for Health. Kidney waiting time calculator. www.ustransplant.org/Calculators/KidneyWaitTime.aspx. Accessed September 16, 2011.
21. Karakayali FY, Ozdemir H, Kivrakdal S, et al. Recurrent glomerular diseases after renal transplantation. Transplant Proc. 2006;38(2):470-472.
22. Nojima M, Ichikawa Y, Ihara H, et al. Significant effect of HLA-DRB1 matching on acute rejection of kidney transplants within 3 months. Transplant Proc. 2001;33(1-2):1182-1184.
Kidney Transplantation: Posttransplant Preventive Care
For many years, the medical community speculated about the possibility of organ transplantation. The first successful transplant of any kind involving humans was a corneal transplant in 1905.1
It wasn’t until 1954 that the first successful organ transplant, a kidney transplant between identical twins, occurred.2 Several new concepts emerged: organ rejection plays a major role in the failure or success of a transplant; and donors and recipients must be matched based on blood group.
Today, about 169,000 people in the US live with a donated kidney. Each year, some 10,500 cadaveric organs are transplanted, and 6,400 donors are living donors.3 The National Kidney Foundation’s recent 10-year initiative, End the Wait!,4 seeks to close the gap between the more than 50,000 people on the transplant waiting list3 and the number of available donor organs.
Since many patients live for years with their transplanted organs, the primary care clinician is likely to see transplant recipients in a family practice or internal medicine setting. While each patient has unique needs, there are commonalities among them.
Renal Consult welcomes any additional comments or questions regarding care of the renal patient. Please address them to editor@clinicianreviews.com.
Jane S. Davis, CRNP, DNP
Q: I am in primary care and have a kidney transplant patient that I see annually for her Pap test and pelvic exam. Is there anything in particular that I am supposed to look for? I feel out of my comfort zone.
As with most people, preventive care is vital and posttransplant patients are no different. However, there are a few “special circumstances” to keep in mind.
Besides ascertaining that posttransplant patients are taking their medications every day, determine whether they have recently had a generic substituted for their regular anti-rejection meds. Many transplant medications have generic equivalents now; while we want changes made only with the approval of a transplant center, it is legal for a pharmacy to substitute a generic without notifying the transplant nephrologist. We have seen rejection, toxicities, or changes in creatinine levels due to substitution of generics—or even substitution from one generic equivalent to another. These medications have a small effective window and have to be closely monitored whenever different manufacturers are used.
In addition, some patients will stop taking their immunosuppressive drug, either because they “feel better” and don’t believe they need it anymore, or because they can no longer afford it. Medicare will only pay for 36 months of these medications, and patients often halve the dose or stop taking the medication altogether when the cost becomes too high.5
There is a very useful Web site on transplant medications from the United States Renal Data System.6 The site, which also offers a wealth of information on chronic kidney disease (CKD), is www.usrds.org/presentations.htm
Dosing for any medication is based on the patient’s glomerular filtration rate (GFR). Your transplant patients have been taught their baseline creatinine level, but some do forget. Even after transplant (whether of a kidney, a pancreas, a liver, lungs, or a heart), the immunosuppressive medications will affect the GFR, and the patient is a CKD patient.
If a patient’s creatinine level is 1.9 mg/dL (normal range, 0.6 to 1.2), but it has varied between 1.8 and 2.0 ever since the transplant and they are not having any other issues, this is “normal” for them and no cause for alarm. On the other hand, if the creatinine level is 1.9 mg/dL and the patient reports that it is always 1.2, they need immediate referral. If the patient is new to the area, you can find a local transplant center on the Organ Procurement and Transplantation Network directory7: optn.transplant.hrsa.gov/mem bers/search.asp
Screening for infections and malignancies is another important aspect of posttransplant care. I advise all patients to see a dermatologist at least once annually, as the risk for skin cancer is increased sevenfold in a transplant patient, compared with the general population.8 Annual Pap test, pelvic exam, and mammogram are important for female posttransplant patients, as is annual prostate-specific antigen testing for male posttransplant patients older than 45 with a life expectancy of at least 10 years.9
During the physical exam, the clinician should always check for lymphadenopathy or any other “lumps and bumps,” as posttransplant lymphoproliferative disorder is also a risk associated with long-term immunosuppression.10 A wonderful online resource for patients and providers, “Transplant Living,”11 has an excellent section on posttransplant care: www.transplantliving.org/af terthetransplant/default.aspx. This Web site is managed by the United Network of Organ Sharing12 (UNOS; www.unos.org), the organization that manages organ transplantation and donation under contract with the federal government.
Routine vaccinations are recommended—especially pneumococcal vaccine and an annual flu shot. Diphtheria-pertussis-tetanus, hepatitis A, hepatitis B, inactivated polio, and typhoid are also acceptable vaccines for a transplant patient. Vaccines that are contraindicated after transplantation include varicella, bacillus Calmette-Guérin, smallpox, intranasal influenza, live oral typhoid, measles, mumps, rubella, oral polio, live Japanese B encephalitis, and yellow fever.13,14
References
1. Armitage WJ, Tullo AB, Larkin DFP. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222-1223.
2. Kidney transplantation: past, present, and future. www.stanford.edu/dept/HPS/transplant/html/history.html. Accessed September 16, 2011.
3. United States Renal Data System. Atlas. www .usrds.org/atlas.htm. Accessed September 16, 2011.
4. National Kidney Foundation. End the wait! www.kidney.org/news/end_the_wait/index.cfm. Accessed September 16, 2011.
5. National Kidney Foundation. Kidney transplant (2011). www.kidney.org/atoz/content/kidneytransnewlease.cfm. Accessed September 16, 2011.
6. United States Renal Data Systems. Presentations and posters (2000-2011). www.usrds.org/presentations.htm. Accessed September 16, 2011.
7. Organ Procurement and Transplantation Network. Members: member directory. optn.transplant.hrsa.gov/members/search.asp. Accessed September 16, 2011.
8. Jensen P, Møller B, Hansen S. Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens. J Am Acad Dermatol. 2000;42(2 pt 1):307.
9. Wong G, Chapman JR, Craig JC. Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol. 2008;3 suppl 2:S87-S100.
10. Parker A, Bowles K, Bradley JA, et al; Haemato-oncology subgroup of the British Committee for Standards in Haematology and the British Transplantation Society. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant patients. Br J Haematol. 2010;149(5):675-692.
11. Transplant Living. After the transplant (2011). www.transplantliving.org/afterthetransplant/default.aspx. Accessed September 16, 2011.
12. United Network for Organ Sharing. www.unos.org. Accessed September 16, 2011.
13. Kidney Disease Improving Global Outcomes. Managing your adult patients who have a kidney transplant (2010). www.kidney.org/professionals/tools/pdf/02-50-4079_ABB_ManagingTransRecip Bk_PC.pdf. Accessed September 16, 2011.
14. Abbud-Filho M, Adams P, Alberu J, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation. 2007; (Suppl 8):83:1-22.
15. Heldal K, Hartmann A, Leivestad T, et al. Risk variables associated with the outcome of kidney recipients >70 years of age in the new millennium. Nephrol Dial Transplant. 2011;26(8):2706-2711.
16. Chava SP, Singh B, Stangou A, et al. Simultaneous combined liver and kidney transplantation: a single center experience. Clin Transplant. 2010; 24(3):E62-E68.
17. Ruiz R, Kunitake H, Wilkinson AH, et al. Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg. 2006;141 (8):735-741.
18. Veroux P, Veroux M, Puliatti C, et al. Kidney transplantation from hepatitis C virus-positive donors into hepatitis C virus-positive recipients: a safe way to expand the donor pool? Transplant Proc. 2005;37(6):2571-2573.
19. United States Renal Data Systems, Annual Data Reports. National Kidney and Urologic Disease Information Clearinghouse. Figure 6ii. Transplant (kidney only) wait list and wait times. www.usrds.org/2010/pdf/v2_07.pdf. Accessed September 16, 2011.
20. Arbor Research Collaborative for Health. Kidney waiting time calculator. www.ustransplant.org/Calculators/KidneyWaitTime.aspx. Accessed September 16, 2011.
21. Karakayali FY, Ozdemir H, Kivrakdal S, et al. Recurrent glomerular diseases after renal transplantation. Transplant Proc. 2006;38(2):470-472.
22. Nojima M, Ichikawa Y, Ihara H, et al. Significant effect of HLA-DRB1 matching on acute rejection of kidney transplants within 3 months. Transplant Proc. 2001;33(1-2):1182-1184.
For many years, the medical community speculated about the possibility of organ transplantation. The first successful transplant of any kind involving humans was a corneal transplant in 1905.1
It wasn’t until 1954 that the first successful organ transplant, a kidney transplant between identical twins, occurred.2 Several new concepts emerged: organ rejection plays a major role in the failure or success of a transplant; and donors and recipients must be matched based on blood group.
Today, about 169,000 people in the US live with a donated kidney. Each year, some 10,500 cadaveric organs are transplanted, and 6,400 donors are living donors.3 The National Kidney Foundation’s recent 10-year initiative, End the Wait!,4 seeks to close the gap between the more than 50,000 people on the transplant waiting list3 and the number of available donor organs.
Since many patients live for years with their transplanted organs, the primary care clinician is likely to see transplant recipients in a family practice or internal medicine setting. While each patient has unique needs, there are commonalities among them.
Renal Consult welcomes any additional comments or questions regarding care of the renal patient. Please address them to editor@clinicianreviews.com.
Jane S. Davis, CRNP, DNP
Q: I am in primary care and have a kidney transplant patient that I see annually for her Pap test and pelvic exam. Is there anything in particular that I am supposed to look for? I feel out of my comfort zone.
As with most people, preventive care is vital and posttransplant patients are no different. However, there are a few “special circumstances” to keep in mind.
Besides ascertaining that posttransplant patients are taking their medications every day, determine whether they have recently had a generic substituted for their regular anti-rejection meds. Many transplant medications have generic equivalents now; while we want changes made only with the approval of a transplant center, it is legal for a pharmacy to substitute a generic without notifying the transplant nephrologist. We have seen rejection, toxicities, or changes in creatinine levels due to substitution of generics—or even substitution from one generic equivalent to another. These medications have a small effective window and have to be closely monitored whenever different manufacturers are used.
In addition, some patients will stop taking their immunosuppressive drug, either because they “feel better” and don’t believe they need it anymore, or because they can no longer afford it. Medicare will only pay for 36 months of these medications, and patients often halve the dose or stop taking the medication altogether when the cost becomes too high.5
There is a very useful Web site on transplant medications from the United States Renal Data System.6 The site, which also offers a wealth of information on chronic kidney disease (CKD), is www.usrds.org/presentations.htm
Dosing for any medication is based on the patient’s glomerular filtration rate (GFR). Your transplant patients have been taught their baseline creatinine level, but some do forget. Even after transplant (whether of a kidney, a pancreas, a liver, lungs, or a heart), the immunosuppressive medications will affect the GFR, and the patient is a CKD patient.
If a patient’s creatinine level is 1.9 mg/dL (normal range, 0.6 to 1.2), but it has varied between 1.8 and 2.0 ever since the transplant and they are not having any other issues, this is “normal” for them and no cause for alarm. On the other hand, if the creatinine level is 1.9 mg/dL and the patient reports that it is always 1.2, they need immediate referral. If the patient is new to the area, you can find a local transplant center on the Organ Procurement and Transplantation Network directory7: optn.transplant.hrsa.gov/mem bers/search.asp
Screening for infections and malignancies is another important aspect of posttransplant care. I advise all patients to see a dermatologist at least once annually, as the risk for skin cancer is increased sevenfold in a transplant patient, compared with the general population.8 Annual Pap test, pelvic exam, and mammogram are important for female posttransplant patients, as is annual prostate-specific antigen testing for male posttransplant patients older than 45 with a life expectancy of at least 10 years.9
During the physical exam, the clinician should always check for lymphadenopathy or any other “lumps and bumps,” as posttransplant lymphoproliferative disorder is also a risk associated with long-term immunosuppression.10 A wonderful online resource for patients and providers, “Transplant Living,”11 has an excellent section on posttransplant care: www.transplantliving.org/af terthetransplant/default.aspx. This Web site is managed by the United Network of Organ Sharing12 (UNOS; www.unos.org), the organization that manages organ transplantation and donation under contract with the federal government.
Routine vaccinations are recommended—especially pneumococcal vaccine and an annual flu shot. Diphtheria-pertussis-tetanus, hepatitis A, hepatitis B, inactivated polio, and typhoid are also acceptable vaccines for a transplant patient. Vaccines that are contraindicated after transplantation include varicella, bacillus Calmette-Guérin, smallpox, intranasal influenza, live oral typhoid, measles, mumps, rubella, oral polio, live Japanese B encephalitis, and yellow fever.13,14
References
1. Armitage WJ, Tullo AB, Larkin DFP. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222-1223.
2. Kidney transplantation: past, present, and future. www.stanford.edu/dept/HPS/transplant/html/history.html. Accessed September 16, 2011.
3. United States Renal Data System. Atlas. www .usrds.org/atlas.htm. Accessed September 16, 2011.
4. National Kidney Foundation. End the wait! www.kidney.org/news/end_the_wait/index.cfm. Accessed September 16, 2011.
5. National Kidney Foundation. Kidney transplant (2011). www.kidney.org/atoz/content/kidneytransnewlease.cfm. Accessed September 16, 2011.
6. United States Renal Data Systems. Presentations and posters (2000-2011). www.usrds.org/presentations.htm. Accessed September 16, 2011.
7. Organ Procurement and Transplantation Network. Members: member directory. optn.transplant.hrsa.gov/members/search.asp. Accessed September 16, 2011.
8. Jensen P, Møller B, Hansen S. Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens. J Am Acad Dermatol. 2000;42(2 pt 1):307.
9. Wong G, Chapman JR, Craig JC. Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol. 2008;3 suppl 2:S87-S100.
10. Parker A, Bowles K, Bradley JA, et al; Haemato-oncology subgroup of the British Committee for Standards in Haematology and the British Transplantation Society. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant patients. Br J Haematol. 2010;149(5):675-692.
11. Transplant Living. After the transplant (2011). www.transplantliving.org/afterthetransplant/default.aspx. Accessed September 16, 2011.
12. United Network for Organ Sharing. www.unos.org. Accessed September 16, 2011.
13. Kidney Disease Improving Global Outcomes. Managing your adult patients who have a kidney transplant (2010). www.kidney.org/professionals/tools/pdf/02-50-4079_ABB_ManagingTransRecip Bk_PC.pdf. Accessed September 16, 2011.
14. Abbud-Filho M, Adams P, Alberu J, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation. 2007; (Suppl 8):83:1-22.
15. Heldal K, Hartmann A, Leivestad T, et al. Risk variables associated with the outcome of kidney recipients >70 years of age in the new millennium. Nephrol Dial Transplant. 2011;26(8):2706-2711.
16. Chava SP, Singh B, Stangou A, et al. Simultaneous combined liver and kidney transplantation: a single center experience. Clin Transplant. 2010; 24(3):E62-E68.
17. Ruiz R, Kunitake H, Wilkinson AH, et al. Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg. 2006;141 (8):735-741.
18. Veroux P, Veroux M, Puliatti C, et al. Kidney transplantation from hepatitis C virus-positive donors into hepatitis C virus-positive recipients: a safe way to expand the donor pool? Transplant Proc. 2005;37(6):2571-2573.
19. United States Renal Data Systems, Annual Data Reports. National Kidney and Urologic Disease Information Clearinghouse. Figure 6ii. Transplant (kidney only) wait list and wait times. www.usrds.org/2010/pdf/v2_07.pdf. Accessed September 16, 2011.
20. Arbor Research Collaborative for Health. Kidney waiting time calculator. www.ustransplant.org/Calculators/KidneyWaitTime.aspx. Accessed September 16, 2011.
21. Karakayali FY, Ozdemir H, Kivrakdal S, et al. Recurrent glomerular diseases after renal transplantation. Transplant Proc. 2006;38(2):470-472.
22. Nojima M, Ichikawa Y, Ihara H, et al. Significant effect of HLA-DRB1 matching on acute rejection of kidney transplants within 3 months. Transplant Proc. 2001;33(1-2):1182-1184.
For many years, the medical community speculated about the possibility of organ transplantation. The first successful transplant of any kind involving humans was a corneal transplant in 1905.1
It wasn’t until 1954 that the first successful organ transplant, a kidney transplant between identical twins, occurred.2 Several new concepts emerged: organ rejection plays a major role in the failure or success of a transplant; and donors and recipients must be matched based on blood group.
Today, about 169,000 people in the US live with a donated kidney. Each year, some 10,500 cadaveric organs are transplanted, and 6,400 donors are living donors.3 The National Kidney Foundation’s recent 10-year initiative, End the Wait!,4 seeks to close the gap between the more than 50,000 people on the transplant waiting list3 and the number of available donor organs.
Since many patients live for years with their transplanted organs, the primary care clinician is likely to see transplant recipients in a family practice or internal medicine setting. While each patient has unique needs, there are commonalities among them.
Renal Consult welcomes any additional comments or questions regarding care of the renal patient. Please address them to editor@clinicianreviews.com.
Jane S. Davis, CRNP, DNP
Q: I am in primary care and have a kidney transplant patient that I see annually for her Pap test and pelvic exam. Is there anything in particular that I am supposed to look for? I feel out of my comfort zone.
As with most people, preventive care is vital and posttransplant patients are no different. However, there are a few “special circumstances” to keep in mind.
Besides ascertaining that posttransplant patients are taking their medications every day, determine whether they have recently had a generic substituted for their regular anti-rejection meds. Many transplant medications have generic equivalents now; while we want changes made only with the approval of a transplant center, it is legal for a pharmacy to substitute a generic without notifying the transplant nephrologist. We have seen rejection, toxicities, or changes in creatinine levels due to substitution of generics—or even substitution from one generic equivalent to another. These medications have a small effective window and have to be closely monitored whenever different manufacturers are used.
In addition, some patients will stop taking their immunosuppressive drug, either because they “feel better” and don’t believe they need it anymore, or because they can no longer afford it. Medicare will only pay for 36 months of these medications, and patients often halve the dose or stop taking the medication altogether when the cost becomes too high.5
There is a very useful Web site on transplant medications from the United States Renal Data System.6 The site, which also offers a wealth of information on chronic kidney disease (CKD), is www.usrds.org/presentations.htm
Dosing for any medication is based on the patient’s glomerular filtration rate (GFR). Your transplant patients have been taught their baseline creatinine level, but some do forget. Even after transplant (whether of a kidney, a pancreas, a liver, lungs, or a heart), the immunosuppressive medications will affect the GFR, and the patient is a CKD patient.
If a patient’s creatinine level is 1.9 mg/dL (normal range, 0.6 to 1.2), but it has varied between 1.8 and 2.0 ever since the transplant and they are not having any other issues, this is “normal” for them and no cause for alarm. On the other hand, if the creatinine level is 1.9 mg/dL and the patient reports that it is always 1.2, they need immediate referral. If the patient is new to the area, you can find a local transplant center on the Organ Procurement and Transplantation Network directory7: optn.transplant.hrsa.gov/mem bers/search.asp
Screening for infections and malignancies is another important aspect of posttransplant care. I advise all patients to see a dermatologist at least once annually, as the risk for skin cancer is increased sevenfold in a transplant patient, compared with the general population.8 Annual Pap test, pelvic exam, and mammogram are important for female posttransplant patients, as is annual prostate-specific antigen testing for male posttransplant patients older than 45 with a life expectancy of at least 10 years.9
During the physical exam, the clinician should always check for lymphadenopathy or any other “lumps and bumps,” as posttransplant lymphoproliferative disorder is also a risk associated with long-term immunosuppression.10 A wonderful online resource for patients and providers, “Transplant Living,”11 has an excellent section on posttransplant care: www.transplantliving.org/af terthetransplant/default.aspx. This Web site is managed by the United Network of Organ Sharing12 (UNOS; www.unos.org), the organization that manages organ transplantation and donation under contract with the federal government.
Routine vaccinations are recommended—especially pneumococcal vaccine and an annual flu shot. Diphtheria-pertussis-tetanus, hepatitis A, hepatitis B, inactivated polio, and typhoid are also acceptable vaccines for a transplant patient. Vaccines that are contraindicated after transplantation include varicella, bacillus Calmette-Guérin, smallpox, intranasal influenza, live oral typhoid, measles, mumps, rubella, oral polio, live Japanese B encephalitis, and yellow fever.13,14
References
1. Armitage WJ, Tullo AB, Larkin DFP. The first successful full-thickness corneal transplant: a commentary on Eduard Zirm’s landmark paper of 1906. Br J Ophthalmol. 2006;90(10):1222-1223.
2. Kidney transplantation: past, present, and future. www.stanford.edu/dept/HPS/transplant/html/history.html. Accessed September 16, 2011.
3. United States Renal Data System. Atlas. www .usrds.org/atlas.htm. Accessed September 16, 2011.
4. National Kidney Foundation. End the wait! www.kidney.org/news/end_the_wait/index.cfm. Accessed September 16, 2011.
5. National Kidney Foundation. Kidney transplant (2011). www.kidney.org/atoz/content/kidneytransnewlease.cfm. Accessed September 16, 2011.
6. United States Renal Data Systems. Presentations and posters (2000-2011). www.usrds.org/presentations.htm. Accessed September 16, 2011.
7. Organ Procurement and Transplantation Network. Members: member directory. optn.transplant.hrsa.gov/members/search.asp. Accessed September 16, 2011.
8. Jensen P, Møller B, Hansen S. Skin cancer in kidney and heart transplant recipients and different long-term immunosuppressive therapy regimens. J Am Acad Dermatol. 2000;42(2 pt 1):307.
9. Wong G, Chapman JR, Craig JC. Cancer screening in renal transplant recipients: what is the evidence? Clin J Am Soc Nephrol. 2008;3 suppl 2:S87-S100.
10. Parker A, Bowles K, Bradley JA, et al; Haemato-oncology subgroup of the British Committee for Standards in Haematology and the British Transplantation Society. Diagnosis of post-transplant lymphoproliferative disorder in solid organ transplant patients. Br J Haematol. 2010;149(5):675-692.
11. Transplant Living. After the transplant (2011). www.transplantliving.org/afterthetransplant/default.aspx. Accessed September 16, 2011.
12. United Network for Organ Sharing. www.unos.org. Accessed September 16, 2011.
13. Kidney Disease Improving Global Outcomes. Managing your adult patients who have a kidney transplant (2010). www.kidney.org/professionals/tools/pdf/02-50-4079_ABB_ManagingTransRecip Bk_PC.pdf. Accessed September 16, 2011.
14. Abbud-Filho M, Adams P, Alberu J, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation. 2007; (Suppl 8):83:1-22.
15. Heldal K, Hartmann A, Leivestad T, et al. Risk variables associated with the outcome of kidney recipients >70 years of age in the new millennium. Nephrol Dial Transplant. 2011;26(8):2706-2711.
16. Chava SP, Singh B, Stangou A, et al. Simultaneous combined liver and kidney transplantation: a single center experience. Clin Transplant. 2010; 24(3):E62-E68.
17. Ruiz R, Kunitake H, Wilkinson AH, et al. Long-term analysis of combined liver and kidney transplantation at a single center. Arch Surg. 2006;141 (8):735-741.
18. Veroux P, Veroux M, Puliatti C, et al. Kidney transplantation from hepatitis C virus-positive donors into hepatitis C virus-positive recipients: a safe way to expand the donor pool? Transplant Proc. 2005;37(6):2571-2573.
19. United States Renal Data Systems, Annual Data Reports. National Kidney and Urologic Disease Information Clearinghouse. Figure 6ii. Transplant (kidney only) wait list and wait times. www.usrds.org/2010/pdf/v2_07.pdf. Accessed September 16, 2011.
20. Arbor Research Collaborative for Health. Kidney waiting time calculator. www.ustransplant.org/Calculators/KidneyWaitTime.aspx. Accessed September 16, 2011.
21. Karakayali FY, Ozdemir H, Kivrakdal S, et al. Recurrent glomerular diseases after renal transplantation. Transplant Proc. 2006;38(2):470-472.
22. Nojima M, Ichikawa Y, Ihara H, et al. Significant effect of HLA-DRB1 matching on acute rejection of kidney transplants within 3 months. Transplant Proc. 2001;33(1-2):1182-1184.
Autosomal Dominant Polycystic Kidney Disease
Twice as common as autism and half as well-known,1 autosomal polycystic kidney disease (ADPKD) occurs in one in 400 to one in 1,000 people.2 It is an inherited progressive genetic disorder that causes hypertension and decreased renal function and, over time, can lead to kidney failure. Two polycstin genes that code for ADPKD, PKD1 and PKD2, were identified in 1994 and 1996, respectively.3,4 Awareness and understanding of the genes responsible for ADPKD have increased clinicians’ ability to identify at-risk patients and to slow or alter the course of the disease.
Case Presentation
A 45-year-old black man presents to your office with severe, nonradiating back pain and new-onset hypertension. Regarding the pain, he stated, “I turned around to see who kicked me, but no one was there.” When the pain began, he went to see the nurse at the school where he is employed, and she found that his blood pressure was high at 162/90 mm Hg. Although the patient’s back pain is resolving, he is very concerned about his blood pressure, since he has never had a high reading before.
He is the baseball coach and physical education teacher at the local high school and is in excellent physical condition as a result of his professional interaction with teenagers every day. He does not smoke or use any illicit drugs but does admit to occasional alcohol consumption. His medical history is significant only for occasional broken fingers and twisted ankles, all occurring while he was engaged in sports.
His family history includes one brother without medical problems, a brother and a sister with hypertension, a sister with diabetes and obesity, and a brother with a congenital abnormality that required a living donor kidney transplant at age 17 (the father served as donor). No family-wide workup has ever been done because no one practitioner has ever made a connection among these conditions and considered a diagnosis of ADPKD.
The patient’s blood pressure in the office is 172/92 mm Hg while sitting and 166/88 mm Hg while standing. He is somewhat sore with a localized spasm in the lumbar-sacral area but no radiation of pain. The patient has trouble touching his toes but reports that he can never touch his toes. His straight leg lift is negative. The rest of his physical exam is noncontributory.
What should be the next step in this patient’s workup?
PATHOPHYSIOLOGY
ADPKD is a progressive expansion of numerous fluid-filled cysts that result in massive enlargement of the kidneys.5 Less than 5% of all nephrons become cystic; however, the average volume of a polycystic kidney is 1,000 mL (normal, 300 mL), that is, the volume of a standard-sized pineapple. Even with this significant enlargement, a decline in the glomerular filtration rate (GFR) is not usually seen initially. Each cyst is derived from a single hyperproliferative epithelial cell. Increased cellular proliferation, followed by fluid secretion and alterations in the extracellular matrix, cause an outpouching from the parent nephron, which eventually detaches from the parent nephron and continues to enlarge and autonomously secrete fluid.6,7
PKD1 and PKD2 are two genes responsible for ADPKD that have been isolated so far. Since there are families carrying neither the PKD1 nor the PKD2 gene that still have an inherited type of ADPKD, there is suspicion that at least one more PKD gene, not yet isolated, exists.8 It is also possible that other genetic or environmental factors may be at play.9,10
In 1994, the PKD1 gene was isolated on chromosome 16,3 and it was found to code for polycystin 1. A lack of polycystin 1 causes an abnormality in the Na+/K(+)-ATPase pumps, leading to abnormal sodium reabsorption.11 How and why this happens is not quite clear. However, the hypertension that is a key objective finding in patients with ADPKD is thought to result from this pump abnormality.
PKD2 is found on the long arm of chromosome 4 and codes for polycystin 2.4 Polycystin 2 is an amino acid that is responsible for voltage-activated cellular calcium channels,5 again explaining the hypertension so commonly seen in the course of ADPKD. ADPKD-associated hypertension may be present as early as the teenage years.12
EPIDEMIOLOGY
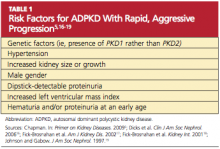
More than 85% of ADPKD cases are associated with PKD1, and this form is called polycystic kidney disease 1 (PKD 1), the more aggressive form of the disease.13,14 PKD 2 (the form associated with the gene PKD2), though less common, is also likely to progress to end-stage renal disease (ESRD), but at a later age (median age of 74 years, compared with 54 in patients with PKD 1).14 ADPKD accounts for about 5% of cases of ESRD in North America,9 but for most patients, presentation and decreased renal function do not occur until the 40s.15 However, patients with the risk factors listed in Table 15,16-19 are likely to experience a more rapid and aggressive form of the disease.
Even with the same germline mutation in a family with this inherited disease, the severity of ADPKD among family members is quite variable; this is true even in the case of twins.9,10,20 Since the age and symptoms at presentation can vary so greatly, a uniform method of identifying patients with ADPKD, along with staging, was needed. Most patients do not undergo genetic testing (ie, DNA linkage or gene-based direct sequencing9) for a diagnosis of ADPKD or to differentiate between the PKD 1 and PKD 2 disease forms unless they are participating in a research study. Diagnostic criteria were needed that were applicable for any type of ADPKD.
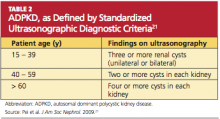
In 2009, the University of Toronto’s Division of Nephrology convened experts in the fields of nephrology and radiology to reach a consensus on standardized ultrasonographic diagnostic criteria.21 They formulated definitions based on a study of 948 individuals who were at risk for either PKD 1 or PKD 2 (see Table 221). The specificity and sensitivity of the resulting criteria range from 82% to 100%, making it possible to standardize the care and classification of renal patients worldwide.
Since family members with the same genotypes can experience very divergent disease manifestations, the two-hit hypothesis has been developed.22 In simple terms, it proposes that after the germline mutation (PKD1 or PKD2), there is a second somatic mutation that leads to progressive cyst formation; when the number and size of cysts increase, the patient starts to experience symptoms of ADPKD.22
Age at presentation can be quite variable, as can the presenting symptoms. Most patients with PKD 1 present in their 50s, with 54 being the average age in US patients.14 The most common presenting symptom is flank or back pain.2,5 The pain is due to the massive enlargement of the kidneys, causing a stretching of the kidney capsule and leading to a chronic, dull and persistent pain in the low back. Severe pain, sharp and cutting, occurs when one of the cysts hemorrhages; to some patients, the pain resembles a quick, powerful “kick in the back.” Hematuria can occur following cyst hemorrhage; depending on the location of the cyst that burst within the kidney (ie, how close it is to the collecting system) and how large it is, the amount and color of the hematuria can be impressive.
ADPKD is more common in men than women, and cyst rupture can be precipitated by trauma or lifting heavy objects. Cyst hemorrhage can turn the urine bright red, which is especially frightening to the male patient. Hematuria is often the key presenting symptom in patients who will be diagnosed with ADPKD-induced hypertension.
Besides hematuria, other common manifestations of ADPKD include:
• Hypertension (60% of affected patients, which increases to 100% by the time ESRD develops)
• Extrarenal cysts (100% of affected patients)
• Urinary tract infections
• Nephrolithiasis (20% of affected patients)
• Proteinuria, occasionally (18% of affected patients).2,5,23
Among these manifestations, those most commonly attributed to a diagnosis of ADPKD are hypertension, kidney stones, and urinary tract or kidney infections. Since isolated proteinuria is unusual in patients with ADPKD, it is recommended that another cause of kidney disease be explored in patients with this presentation.24
Extrarenal manifestations of cyst development are common, eventually occurring in all patients as they age. Hepatic cysts are universal in patients with ADPKD by age 30, although hepatic function is preserved. There may be a mild elevation in the alkaline phosphatase level in patients with ADPKD, resulting from the presence of hepatic cysts. Cysts may also be found in the pancreas, spleen, thyroid, and epididymis.5,25 Some patients may complain of dyspnea, pain, early satiety, or lower extremity edema as a result of enlarged cyst.
The Case Patient
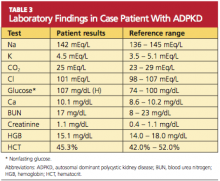
Because you recently attended a lecture about ADPKD, you are aware that flank pain in men with hypertension is indicative of ADPKD until proven otherwise. Believing that this patient’s hypertension is renal in origin, you order an abdominal ultrasound. You also order a comprehensive metabolic panel and a complete blood count. The patient’s GFR is measured at 89 mL/min (indicative of stage 2 kidney disease). Other results are shown in Table 3.
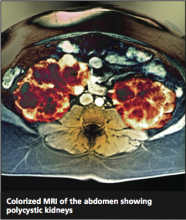
The very broad differential includes essential hypertension, hypertension resulting from intake of “power drinks” or salt in an athlete, illicit use of medications (including steroids), herniated disc leading to transient hypertension, and urinary tract infection or sexually transmitted disease. All of this is moot when the ultrasound shows both kidneys measuring greater than 15 cm, with four distinct cysts on the right kidney and three distinct cysts on the left.
You explain to the patient that ADPKD is a genetic disease and that he and his siblings each had a certain chance of inheriting it. Although different presentations may occur (“congenital” polycystic kidney disease, hypertension, or obesity), they all must undergo ultrasonographic screening for ADPKD. You add that although ADPKD is a genetic disease, it can also be diagnosed in different members of the same family at different ages.
TREATMENT
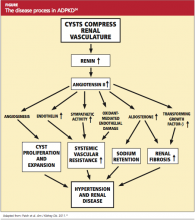
The goal of treatment for the patient with ADPKD is to slow cyst development and the natural course of the disease. If this can be achieved, the need for dialysis or kidney transplantation may be postponed for a number of years. Because cyst growth causes an elevation in renin and activates the angiotensin II renin system26 (see figure,24), an ACE inhibitor is the most effective treatment to lower blood pressure and thus slow the progression of ADPKD. Most patients with ADPKD are started on an ACE inhibitor at an early age to slow the rate of disease progression.27,28 Several studies are under way to determine the best antihypertensive medication and the optimal age for initiating treatment.29,30
Lipid screening and treatment for dyslipidemia are important23 because ADPKD can lead to a reduction in kidney function, resulting in chronic kidney disease (CKD). CKD is considered a coronary heart disease risk equivalent, and most professionals will treat the patient with ADPKD for hyperlipidemia.23,31 While there are no data showing that statin use will reduce the incidence of ESRD or delay the need for dialysis or kidney transplantation in patients with ADPKD, the beneficial effects of good renal blood flow and endothelial function have been noted.32,33
One of the most common and significant complications in ADPKD is intracranial hemorrhage resulting from a ruptured cerebral aneurysm. In the younger adult, the incidence of cerebral aneurysm is 4%, but incidence increases to 10% in patients older than 65.34-36 Family clusters of aneurysms have been reported.37 If an intracranial aneurysm is found in the family history, the risk of an aneurysm in another family member increases to 22%.38
Since rupture of an intracranial hemorrhage is associated with a 30-day mortality rate of 50% and 80% morbidity,5,38 standard of care for patients with ADPKD includes CT or magnetic resonance angiographic (MRA) screening in the asymptomatic patient with a positive family history.34,38 If an aneurysm is found, the lifetime chance of rupture is 50%, although the risk is greater in the case of an aneurysm larger than 10 mm.5
As in all patients with kidney disease, left ventricular hypertrophy is common among patients with ADPKD.23,28,39
The Case Patient
The patient is started on an ACE inhibitor, scheduled for fasting lipid screening, and referred to a nephrology practice for disease management. As research and investigation of possible treatment options for ADPKD are ongoing, the patient may benefit from participating in a new research protocol.
Because the patient’s family has no history of cerebral aneurysm, CT/MRA screening is not required. A discussion of the pros and cons of genetic testing for the entire family, including nieces and nephews, is initiated. The patient and his family are referred to a genetic counselor to decide whether the benefit of early treatment for hypertension outweighs the risk of carrying a diagnosis of ADPKD for his younger relatives, who may later seek health insurance coverage.
NATURAL PROGRESSION OF ADPKD
Hypertension and cyst formation will continue as the patient ages. The natural progression of ADPKD is to renal failure with renal replacement therapy (dialysis or organ transplantation) as treatment options. If the progression of ADPKD can be slowed through pharmacotherapy, the patient may live for many years without needing dialysis. This ideal can be accomplished only through aggressive hypertension control, which should be started in the teenage years.23,30,31
Suggestions to increase fluid consumption and to limit the use of NSAIDs, contrast dye, and MRI with gadolinium are appropriate. It is rare for hypertension to be diagnosed before some organ damage has already occurred.12 Often the patient’s renal function, as determined by measuring the GFR, remains stable until the patient reaches his or her 40s.40 However, kidney damage often begins before any detectable change in GFR. Once the GFR does start to decline, the average decrease is 4.4 to 5.9 mL/min/1.73m2 each year.41
MANAGEMENT CONSIDERATIONS
For ESRD Organ Transplantation
Kidney transplantation—the only curative treatment for ADPKD—can be offered to patients once the GFR falls below 20 mL/min. However, the patient with ADPKD can experience kidney enlargement to such an extent that introducing a third kidney into the limited abdominal space becomes technically difficult. Although nephrectomy is avoided whenever possible, there are cases in which there is no alternative.42
In addition to space concerns, recurrent urinary tract infections, chronic pain, renal cell carcinoma, chronic hematuria, or chronic cyst infections can necessitate a nephrectomy.43,44 A laparoscopic approach with decompression of cysts or removal of only one kidney is preferred.43,45 If removal of both kidneys is required before a transplant, the patient must be maintained on dialysis until after transplantation. Since the transplant waiting list can exceed seven years in some areas, most patients arrange for a willing live donor before agreeing to a bilateral nephrectomy.46,47
Dialysis
Either peritoneal dialysis (PD) or hemodialysis (HD) can be offered to patients with severe ADPKD. Depending on the size of the native kidneys and the history of previous abdominal surgery, PD often offers a better chance of survival in these patients, particularly compared with patients who have ESRD associated with other causes.48
For management of the patient with ADPKD who receives PD, it can be difficult to differentiate between the pain of a cyst and the pain of a peritoneal infection. Generally, cyst rupture is accompanied by hematuria; and peritonitis, by cloudy fluid.5 Management provided by an experienced nephrologist and PD nurse is vital.
In ADPKD patients who undergo HD, too, survival is better than in patients who have ESRD with other causes49,50; five-year survival can be as high as 10% to 15%.51 This is likely due to the lower incidence of coronary artery disease in the ADPKD population, compared with patients who have ESRD associated with other chronic diseases.49
FUTURE TRENDS AND ONGOING TRIALS
HALT PKD29,30 is an NIH-funded, double-blind study to determine whether adding an angiotensin receptor blocker (ARB) to standard ACE inhibitor therapy results in a more significant decrease in the progression of renal cysts. The rationale for this is that the ARB is expected to block the renin-angiotensin-aldosterone system in the kidney. Use of ACE inhibitor monotherapy versus ARB/ACE inhibitor therapy is being compared in two study arms: patients between ages 15 and 49 with a GFR of 60 mL/min or greater; and patients between ages 18 and 64 with a GFR of 25 to 60 mL/min.29 To date, preliminary results indicate no benefit in adding the second medication.49
The TEMPO Trial52 is a multicenter, double-blind study looking at the effect of tolvaptan on renal cyst growth. Tolvaptan is a potent vasopressin receptor antagonist, and in vitro evidence has shown that intracellular cyclic adenosine monophosphate (cAMP) plays a large role in the development of cysts in patients with ADPKD. If it is possible to block the cAMP that is causing cyst growth, progression of ADPKD should slow.53,54 Only short-term effects of tolvaptan use are currently known.55
High Water Intake to Slow Progression of Polycystic Kidney Disease56 is an open-label, nonrandomized trial in which patients drink a minimum of
3 L of water. Previously, a small study showed that an increase in fluid intake partially suppresses the urine osmolality and the serum antidiuretic hormone (ADH) levels.57 According to this theory, increasing water intake to greater than 3 L/d may result in complete suppression of ADH and cAMP. This is a small study (n = 20),56 since patients with ADPKD are likely to have urinary concentrating defects, and hyponatremia is a concern is in these patients.58
Sirolimus and ADPKD59 is an open-label randomized study to see whether sirolimus (also known as rapamycin) can reduce cyst growth. Originally, it was noted that posttransplant ADPKD patients underwent a regression of both liver and kidney cysts when they were taking sirolimus, and a preliminary crossover study was done.60 However, preliminary results at 18 months showed no difference in renal growth or cyst growth but did show kidney damage as determined by an increase of proteinuria in the treatment group.59 The study is still in progress.
Somatostatin in Polycystic Kidney61 is a long-term (three-year) study following patients who agreed to participate in a randomized, double-blind protocol; in it, an intramuscular injection of either an octreotide (ie, somatastatin) or placebo was administered every four weeks for one year in an effort to reduce the size of kidney and liver cysts.62 At one year, the quality of life in the treatment group was rated better, as measured by pain reduction and improved physical activity. Cyst growth in the treatment group was smaller for both the kidney and liver. However, the GFR decreased to the same degree in both groups.62
CONCLUSION
ADPKD is a common, often overlooked genetic disease that is a cause of hypertension. ADPKD’s presenting symptoms of flank pain, back pain, and/or hematuria often bring the patient to the provider, but a high index of suspicion must be maintained to diagnose these patients at an early age. Due to the variable presentation even within affected families, many patients do not realize that their family carries the PKD gene.
While genetic testing is available, ultrasound is a quick, relatively inexpensive, and easy method to screen for this diagnosis. The progression of ADPKD to ESRD, requiring dialysis or organ transplantation, can be slowed with early and aggressive treatment of hypertension. As with all patients affected by renal impairment, suggestions for patients with ADPKD to avoid use of NSAIDs, contrast dye, and gadolinium-enhanced MRI are appropriate. The primary care PA or NP is in an appropriate position to see to this.
REFERENCES
1. Newschaffer CJ, Falb MD, Gurney JG. National autism prevalence trends from United States special education data. Pediatrics. 2005;115 (3):e277-e282.
2. Torres VE, Harris PC, Pirson Y. Autosomal dominant polycystic kidney disease. Lancet. 2007;369(9569):1287-1301.
3. The polycystic kidney disease 1 gene encodes a 14 kb transcript and lies within a duplicated region on chromosome 16: European Polycystic Kidney Disease Consortium. Cell. 1994;77(6):881-894.
4. Mochizuki T, Wu G, Hayashi T, et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science. 1996; 272(5266):1339-1342.
5. Chapman AB. Polycystic and other cystic kidney diseases. In: Greenberg A, ed. Primer on Kidney Diseases: Expert Consult. 5th ed. National Kidney Foundation; 2009:345-353.
6. Fischer E, Legue E, Doyen A, et al. Defective planar cell polarity in polycystic kidney disease. Nat Genet. 2006;38(1):21-23.
7. Murcia NS, Sweeney WE Jr, Avner ED. New insights into the molecular pathophysiology of polycystic kidney disease. Kidney Int. 1999;55 (4):1187-1197.
8. Paterson AD, Pei Y. Is there a third gene for autosomal dominant polycystic kidney disease? Kidney Int. 1998;54(5):1759-1761.
9. Pei Y. Practical genetics for autosomal dominant polycystic kidney disease. Nephron Clin Pract. 2011;118(1):c19-c30.
10. Tan YC, Blumenfeld J, Rennert H. Autosomal dominant polycystic kidney disease: genetics, mutations and microRNAs. Biochim Biophys Acta. 2011 Mar 16 [Epub ahead of print].
11. Avner ED, Sweeney WE Jr, Nelson WJ. Abnormal sodium pump distribution during renal tubulogenesis in congenital murine polycystic kidney disease. Proc Natl Acad Sci U S A. 1992;89(16):7447-7451.
12. Torra R, Badenas C, Darnell A, et al. Linkage, clinical features, and prognosis of autosomal dominant polycystic kidney disease types 1 and 2. J Am Soc Nephrol. 1996;7(10):2142-2151.
13. Davies F, Coles GA, Harper PS, et al. Polycystic kidney disease re-evaluated: a population-based study. Q J Med. 1991;79(290):477-485.
14. Hateboer N, v Dijk MA, Bogdanova N, et al; European PKD1-PKD2 Study Group. Comparison of phenotypes of polycystic kidney disease types 1 and 2. Lancet. 1999;353(9147):103-107.
15. Peters DJ, Breuning MH. Autosomal dominant polycystic kidney disease: modification of disease progression. Lancet. 2001;358(9291):1439-1444.
16. Dicks E, Ravani P, Langman D, et al. Incident renal events and risk factors in autosomal dominant polycystic kidney disease: a population- and family-based cohort followed for 22 years. Clin J Am Soc Nephrol. 2006;1(4):710-717.
17. Fick-Brosnahan GM, Belz MM, McFann KK, etc. Relationship between renal volume growth and renal function in autosomal dominant polycystic kidney disease: a longitudinal study. Am J Kidney Dis. 2002;39(6):1127-1134.
18. Fick-Brosnahan GM, Tran ZV, Johnson AM, et al. Progression of autosomal-dominant polycystic kidney disease in children. Kidney Int. 2001;59(5):1654-1662.
19. Johnson AM, Gabow PA. Identification of patients with autosomal dominant polycystic kidney disease at highest risk for end-stage renal disease. J Am Soc Nephrol. 1997;8(10): 1560-1567.
20. Risk D. Autosomal dominant polycystic kidney disease. Presented at: National Kidney Foundation, Spring Clinical Meetings; April 28, 2011; Las Vegas, NV.
21. Pei Y, Obaji J, Dupuis A, et al. Unified criteria for ultrasonographic diagnosis of ADPKD. J Am Soc Nephrol. 2009;20(1):205-212.
22. Watnick T, Germino GG. Molecular basis of autosomal dominant polycystic kidney disease. Semin Nephrol. 1999;19(4):327-343.
23. Ecder T, Schrier RW. Cardiovascular abnormalities in autosomal-dominant polycystic kidney disease. Nat Rev Nephrol. 2009;5(4):221-228.
24. Patch C, Charlton J, Roderick PJ, Gulliford MC. Use of antihypertensive medications and mortality of patients with autosomal dominant polycystic kidney disease: a population-based study. Am J Kidney Dis. 2011;57(6):856-862.
25. Pirson Y. Extrarenal manifestations of autosomal dominant polycystic kidney disease. Adv Chronic Kidney Dis. 2010;17(2):173-180.
26. Chapman AB, Johnson A, Gabow PA, Schrier RW. The renin-angiotensin-aldosterone system and autosomal dominant polycystic kidney disease. N Engl J Med. 1990;323(16):1091-1096.
27. Jafar TH, Stark PC, Schmid CH, et al. The effect of angiotensin-converting-enzyme inhibitors on progression of advanced polycystic kidney disease. Kidney Int. 2005;67(1):265-271.
28. Schrier RW. Renal volume, renin-angiotensin-aldosterone system, hypertension, and left ventricular hypertrophy in patients with autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 2009;20(9):1888-1893.
29. National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), NIH, sponsor. HALT PKD (Halt Progression of Polycystic Kidney Disease): Efficacy of Aggressive Renin-Angiotensin-Aldosterone Axis Blockade in Preventing/Slowing Renal Function Decline in ADPKD. www2.niddk.nih.gov/NR/rdonlyres/175578F6-62B4-429A-9BBF-96CCEC2FFB3A/0/KUHHALT PKDPROTOCOL9107.pdf. Accessed July 22, 2011.
30. Chapman AB, Torres VE, Perrone RD, et al. The HALT polycystic kidney disease trials: design and implementation. Clin J Am Soc Nephrol. 2010;5(1):102-109.
31. Taylor M, Johnson AM, Tison M, et al. Earlier diagnosis of autosomal dominant polycystic kidney disease: importance of family history and implications for cardiovascular and renal complications. Am J Kidney Dis. 2005;46(3):415-423.
32. Namli S, Oflaz H, Turgut F, et al. Improvement of endothelial dysfunction with simvastatin in patients with autosomal dominant polycystic kidney disease. Ren Fail. 2007;29(1):55-59.
33. Bremmer MS, Jacobs SC. Renal artery embolization for the symptomatic treatment of adult polycystic kidney disease. Nat Clin Pract Nephrol. 2008;4(5):236-237.
34. Chapman AB, Rubinstein D, Hughes R, et al. Intracranial aneurysms in autosomal dominant polycystic kidney disease. N Engl J Med. 1992; 327(13):916-920.
35. Schievink WI, Torres VE, Piepgras DG, Wiebers DO. Saccular intracranial aneurysms in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 1992;3(1):88-95.
36. Fick GM, Gabow PA. Hereditary and acquired cystic disease of the kidney. Kidney Int. 1994;46(4):951-964.
37. Watson ML. Complications of polycystic kidney disease. Kidney Int. 1997;51(1):353-365.
38. Huston J 3rd, Torres VE, Sulivan PP, et al. Value of magnetic resonance angiography for the detection of intracranial aneurysms in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 1993;3(12):1871-1877.
39. Longenecker JC, Coresh J, Powe NR, et al. Traditional cardiovascular disease risk factors in dialysis patients compared with the general population: the CHOICE Study. J Am Soc Nephrol. 2002;13(7):1918-1927.
40. Meijer E, Rook M, Tent H, et al. Early renal abnormalities in autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol. 2010; 5(6):1091-1098.
41. Torres VE, Harris PC. Autosomal dominant polycystic kidney disease: the last 3 years. Kidney Int. 2009;76(2):149-168.
42. Tabibi A, Simforoosh N, Abadpour P, et al. Concomitant nephrectomy of massively enlarged kidneys and renal transplantation in autosomal dominant polycystic kidney disease. Transplant Proc. 2005;37(7):2939-2940.
43. Dunn MD, Portis AJ, Elbahnasy AM, et al. Laparoscopic nephrectomy in patients with end-stage renal disease and autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2000;35(4):720-725.
44. Sulikowski T, Tejchman K, Zietek Z, et al. Experience with autosomal dominant polycystic kidney disease in patients before and after renal transplantation: a 7-year observation. Transplant Proc. 2009;41(1):177-180.
45. Desai MR, Nandkishore SK, Ganpule A, Thimmegowda M. Pretransplant laparoscopic nephrectomy in adult polycystic kidney disease: a single centre experience. BJU Int. 2008;101 (1):94-97.
46. Glassman DT, Nipkow L, Bartlett ST, Jacobs SC. Bilateral nephrectomy with concomitant renal graft transplantation for autosomal dominant polycystic kidney disease. J Urol. 2000;164 (3 pt 1):661-664.
47. Fuller TF, Brennan TV, Feng S, et al. End stage polycystic kidney disease: indications and timing of native nephrectomy relative to kidney transplantation. J Urol. 2005;174(6):2284-2288.
48. Abbott KC, Agodoa LY. Polycystic kidney disease at end-stage renal disease in the United States: patient characteristics and survival. Clin Nephrol. 2002;57(3):208-214.
49. Perrone RD, Ruthazer R, Terrin NC. Survival after end-stage renal disease in autosomal dominant polycystic kidney disease: contribution of extrarenal complications to mortality. Am J Kidney Dis. 2001;38(4):777-784.
50. Batista PB, Lopes AA, Costa FA. Association between attributed cause of end-stage renal disease and risk of death in Brazilian patients receiving renal replacement therapy. Ren Fail. 2005;27(6):651-656.
51. Pirson Y, Christophe JL, Goffin E. Outcome of renal replacement therapy in autosomal dominant polycystic kidney disease. Nephrol Dial Transplant. 1996;11 suppl 6:24-28.
52. Torres VE, Meijer E, Bae KT, et al. Rationale and design of the TEMPO (Tolvaptan Efficacy and Safety in Management of Autosomal Dominant Polycystic Kidney Disease and its Outcomes) 3-4 Study. Am J Kidney Dis. 2011;57(5):692-699.
53. Calvet JP. Strategies to inhibit cyst formation in ADPKD. Clin J Am Soc Nephrol. 2008;3 (4):1205-1211.
54. Grantham JJ. Lillian Jean Kaplan International Prize for advancement in the understanding of polycystic kidney disease. Understanding polycystic kidney disease: a systems biology approach. Kidney Int. 2003;64(4):1157-1162.
55. Irazabal MV, Torres VE, Hogan MC, et al. Short-term effects of tolvaptan on renal function and volume in patients with Autosomal Dominant Polycystic Kidney Disease. Kidney Int. 2011 May 4 [Epub ahead of print].
56. New York University, sponsor. High Water Intake to Slow Progression of Polycystic Kidney Disease. http://clinicaltrials.gov/ct2/show/NCT00784030. Accessed July 22, 2011.
57. Wang CJ, Creed C, Winklhofer FT, Grantham JJ. Water prescription in autosomal dominant polycystic kidney disease: a pilot study. Clin J Am Soc Nephrol. 2011;6(1):192-197.
58. Grampsas SA, Chandhoke Ps, Fan J, et al. Anatomic and metabolic risk factors for nephrolithiasis in patients with autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2000;36(1):53-57.
59. Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney growth in autosomal dominant polycystic kidney disease. N Engl J Med. 2010; 363(9):820-829.
60. Perico N, Antiga L, Caroli A, et al. Sirolimus therapy to halt the progression of ADPKD. J Am Soc Nephrol. 2010;21(6):1031-1040.
61. Mario Negri Institute for Pharmacological Research, sponsor. Somatostatin in Polycystic Kidney: a Long-term Three Year Follow up Study. http://clinicaltrials.gov/ct2/show/NCT00309283. Accessed July 22, 2011.
62. Hogan MC, Masyuk TV, Page LJ, et al. Randomized clinical trial of long-acting somatostatin for autosomal dominant polycystic kidney and liver disease. J Am Soc Nephrol. 2010; 21(6):1052-1061.
Twice as common as autism and half as well-known,1 autosomal polycystic kidney disease (ADPKD) occurs in one in 400 to one in 1,000 people.2 It is an inherited progressive genetic disorder that causes hypertension and decreased renal function and, over time, can lead to kidney failure. Two polycstin genes that code for ADPKD, PKD1 and PKD2, were identified in 1994 and 1996, respectively.3,4 Awareness and understanding of the genes responsible for ADPKD have increased clinicians’ ability to identify at-risk patients and to slow or alter the course of the disease.
Case Presentation
A 45-year-old black man presents to your office with severe, nonradiating back pain and new-onset hypertension. Regarding the pain, he stated, “I turned around to see who kicked me, but no one was there.” When the pain began, he went to see the nurse at the school where he is employed, and she found that his blood pressure was high at 162/90 mm Hg. Although the patient’s back pain is resolving, he is very concerned about his blood pressure, since he has never had a high reading before.
He is the baseball coach and physical education teacher at the local high school and is in excellent physical condition as a result of his professional interaction with teenagers every day. He does not smoke or use any illicit drugs but does admit to occasional alcohol consumption. His medical history is significant only for occasional broken fingers and twisted ankles, all occurring while he was engaged in sports.
His family history includes one brother without medical problems, a brother and a sister with hypertension, a sister with diabetes and obesity, and a brother with a congenital abnormality that required a living donor kidney transplant at age 17 (the father served as donor). No family-wide workup has ever been done because no one practitioner has ever made a connection among these conditions and considered a diagnosis of ADPKD.
The patient’s blood pressure in the office is 172/92 mm Hg while sitting and 166/88 mm Hg while standing. He is somewhat sore with a localized spasm in the lumbar-sacral area but no radiation of pain. The patient has trouble touching his toes but reports that he can never touch his toes. His straight leg lift is negative. The rest of his physical exam is noncontributory.
What should be the next step in this patient’s workup?
PATHOPHYSIOLOGY
ADPKD is a progressive expansion of numerous fluid-filled cysts that result in massive enlargement of the kidneys.5 Less than 5% of all nephrons become cystic; however, the average volume of a polycystic kidney is 1,000 mL (normal, 300 mL), that is, the volume of a standard-sized pineapple. Even with this significant enlargement, a decline in the glomerular filtration rate (GFR) is not usually seen initially. Each cyst is derived from a single hyperproliferative epithelial cell. Increased cellular proliferation, followed by fluid secretion and alterations in the extracellular matrix, cause an outpouching from the parent nephron, which eventually detaches from the parent nephron and continues to enlarge and autonomously secrete fluid.6,7
PKD1 and PKD2 are two genes responsible for ADPKD that have been isolated so far. Since there are families carrying neither the PKD1 nor the PKD2 gene that still have an inherited type of ADPKD, there is suspicion that at least one more PKD gene, not yet isolated, exists.8 It is also possible that other genetic or environmental factors may be at play.9,10
In 1994, the PKD1 gene was isolated on chromosome 16,3 and it was found to code for polycystin 1. A lack of polycystin 1 causes an abnormality in the Na+/K(+)-ATPase pumps, leading to abnormal sodium reabsorption.11 How and why this happens is not quite clear. However, the hypertension that is a key objective finding in patients with ADPKD is thought to result from this pump abnormality.
PKD2 is found on the long arm of chromosome 4 and codes for polycystin 2.4 Polycystin 2 is an amino acid that is responsible for voltage-activated cellular calcium channels,5 again explaining the hypertension so commonly seen in the course of ADPKD. ADPKD-associated hypertension may be present as early as the teenage years.12
EPIDEMIOLOGY
More than 85% of ADPKD cases are associated with PKD1, and this form is called polycystic kidney disease 1 (PKD 1), the more aggressive form of the disease.13,14 PKD 2 (the form associated with the gene PKD2), though less common, is also likely to progress to end-stage renal disease (ESRD), but at a later age (median age of 74 years, compared with 54 in patients with PKD 1).14 ADPKD accounts for about 5% of cases of ESRD in North America,9 but for most patients, presentation and decreased renal function do not occur until the 40s.15 However, patients with the risk factors listed in Table 15,16-19 are likely to experience a more rapid and aggressive form of the disease.
Even with the same germline mutation in a family with this inherited disease, the severity of ADPKD among family members is quite variable; this is true even in the case of twins.9,10,20 Since the age and symptoms at presentation can vary so greatly, a uniform method of identifying patients with ADPKD, along with staging, was needed. Most patients do not undergo genetic testing (ie, DNA linkage or gene-based direct sequencing9) for a diagnosis of ADPKD or to differentiate between the PKD 1 and PKD 2 disease forms unless they are participating in a research study. Diagnostic criteria were needed that were applicable for any type of ADPKD.
In 2009, the University of Toronto’s Division of Nephrology convened experts in the fields of nephrology and radiology to reach a consensus on standardized ultrasonographic diagnostic criteria.21 They formulated definitions based on a study of 948 individuals who were at risk for either PKD 1 or PKD 2 (see Table 221). The specificity and sensitivity of the resulting criteria range from 82% to 100%, making it possible to standardize the care and classification of renal patients worldwide.
Since family members with the same genotypes can experience very divergent disease manifestations, the two-hit hypothesis has been developed.22 In simple terms, it proposes that after the germline mutation (PKD1 or PKD2), there is a second somatic mutation that leads to progressive cyst formation; when the number and size of cysts increase, the patient starts to experience symptoms of ADPKD.22
Age at presentation can be quite variable, as can the presenting symptoms. Most patients with PKD 1 present in their 50s, with 54 being the average age in US patients.14 The most common presenting symptom is flank or back pain.2,5 The pain is due to the massive enlargement of the kidneys, causing a stretching of the kidney capsule and leading to a chronic, dull and persistent pain in the low back. Severe pain, sharp and cutting, occurs when one of the cysts hemorrhages; to some patients, the pain resembles a quick, powerful “kick in the back.” Hematuria can occur following cyst hemorrhage; depending on the location of the cyst that burst within the kidney (ie, how close it is to the collecting system) and how large it is, the amount and color of the hematuria can be impressive.
ADPKD is more common in men than women, and cyst rupture can be precipitated by trauma or lifting heavy objects. Cyst hemorrhage can turn the urine bright red, which is especially frightening to the male patient. Hematuria is often the key presenting symptom in patients who will be diagnosed with ADPKD-induced hypertension.
Besides hematuria, other common manifestations of ADPKD include:
• Hypertension (60% of affected patients, which increases to 100% by the time ESRD develops)
• Extrarenal cysts (100% of affected patients)
• Urinary tract infections
• Nephrolithiasis (20% of affected patients)
• Proteinuria, occasionally (18% of affected patients).2,5,23
Among these manifestations, those most commonly attributed to a diagnosis of ADPKD are hypertension, kidney stones, and urinary tract or kidney infections. Since isolated proteinuria is unusual in patients with ADPKD, it is recommended that another cause of kidney disease be explored in patients with this presentation.24
Extrarenal manifestations of cyst development are common, eventually occurring in all patients as they age. Hepatic cysts are universal in patients with ADPKD by age 30, although hepatic function is preserved. There may be a mild elevation in the alkaline phosphatase level in patients with ADPKD, resulting from the presence of hepatic cysts. Cysts may also be found in the pancreas, spleen, thyroid, and epididymis.5,25 Some patients may complain of dyspnea, pain, early satiety, or lower extremity edema as a result of enlarged cyst.
The Case Patient
Because you recently attended a lecture about ADPKD, you are aware that flank pain in men with hypertension is indicative of ADPKD until proven otherwise. Believing that this patient’s hypertension is renal in origin, you order an abdominal ultrasound. You also order a comprehensive metabolic panel and a complete blood count. The patient’s GFR is measured at 89 mL/min (indicative of stage 2 kidney disease). Other results are shown in Table 3.
The very broad differential includes essential hypertension, hypertension resulting from intake of “power drinks” or salt in an athlete, illicit use of medications (including steroids), herniated disc leading to transient hypertension, and urinary tract infection or sexually transmitted disease. All of this is moot when the ultrasound shows both kidneys measuring greater than 15 cm, with four distinct cysts on the right kidney and three distinct cysts on the left.
You explain to the patient that ADPKD is a genetic disease and that he and his siblings each had a certain chance of inheriting it. Although different presentations may occur (“congenital” polycystic kidney disease, hypertension, or obesity), they all must undergo ultrasonographic screening for ADPKD. You add that although ADPKD is a genetic disease, it can also be diagnosed in different members of the same family at different ages.
TREATMENT
The goal of treatment for the patient with ADPKD is to slow cyst development and the natural course of the disease. If this can be achieved, the need for dialysis or kidney transplantation may be postponed for a number of years. Because cyst growth causes an elevation in renin and activates the angiotensin II renin system26 (see figure,24), an ACE inhibitor is the most effective treatment to lower blood pressure and thus slow the progression of ADPKD. Most patients with ADPKD are started on an ACE inhibitor at an early age to slow the rate of disease progression.27,28 Several studies are under way to determine the best antihypertensive medication and the optimal age for initiating treatment.29,30
Lipid screening and treatment for dyslipidemia are important23 because ADPKD can lead to a reduction in kidney function, resulting in chronic kidney disease (CKD). CKD is considered a coronary heart disease risk equivalent, and most professionals will treat the patient with ADPKD for hyperlipidemia.23,31 While there are no data showing that statin use will reduce the incidence of ESRD or delay the need for dialysis or kidney transplantation in patients with ADPKD, the beneficial effects of good renal blood flow and endothelial function have been noted.32,33
One of the most common and significant complications in ADPKD is intracranial hemorrhage resulting from a ruptured cerebral aneurysm. In the younger adult, the incidence of cerebral aneurysm is 4%, but incidence increases to 10% in patients older than 65.34-36 Family clusters of aneurysms have been reported.37 If an intracranial aneurysm is found in the family history, the risk of an aneurysm in another family member increases to 22%.38
Since rupture of an intracranial hemorrhage is associated with a 30-day mortality rate of 50% and 80% morbidity,5,38 standard of care for patients with ADPKD includes CT or magnetic resonance angiographic (MRA) screening in the asymptomatic patient with a positive family history.34,38 If an aneurysm is found, the lifetime chance of rupture is 50%, although the risk is greater in the case of an aneurysm larger than 10 mm.5
As in all patients with kidney disease, left ventricular hypertrophy is common among patients with ADPKD.23,28,39
The Case Patient
The patient is started on an ACE inhibitor, scheduled for fasting lipid screening, and referred to a nephrology practice for disease management. As research and investigation of possible treatment options for ADPKD are ongoing, the patient may benefit from participating in a new research protocol.
Because the patient’s family has no history of cerebral aneurysm, CT/MRA screening is not required. A discussion of the pros and cons of genetic testing for the entire family, including nieces and nephews, is initiated. The patient and his family are referred to a genetic counselor to decide whether the benefit of early treatment for hypertension outweighs the risk of carrying a diagnosis of ADPKD for his younger relatives, who may later seek health insurance coverage.
NATURAL PROGRESSION OF ADPKD
Hypertension and cyst formation will continue as the patient ages. The natural progression of ADPKD is to renal failure with renal replacement therapy (dialysis or organ transplantation) as treatment options. If the progression of ADPKD can be slowed through pharmacotherapy, the patient may live for many years without needing dialysis. This ideal can be accomplished only through aggressive hypertension control, which should be started in the teenage years.23,30,31
Suggestions to increase fluid consumption and to limit the use of NSAIDs, contrast dye, and MRI with gadolinium are appropriate. It is rare for hypertension to be diagnosed before some organ damage has already occurred.12 Often the patient’s renal function, as determined by measuring the GFR, remains stable until the patient reaches his or her 40s.40 However, kidney damage often begins before any detectable change in GFR. Once the GFR does start to decline, the average decrease is 4.4 to 5.9 mL/min/1.73m2 each year.41
MANAGEMENT CONSIDERATIONS
For ESRD Organ Transplantation
Kidney transplantation—the only curative treatment for ADPKD—can be offered to patients once the GFR falls below 20 mL/min. However, the patient with ADPKD can experience kidney enlargement to such an extent that introducing a third kidney into the limited abdominal space becomes technically difficult. Although nephrectomy is avoided whenever possible, there are cases in which there is no alternative.42
In addition to space concerns, recurrent urinary tract infections, chronic pain, renal cell carcinoma, chronic hematuria, or chronic cyst infections can necessitate a nephrectomy.43,44 A laparoscopic approach with decompression of cysts or removal of only one kidney is preferred.43,45 If removal of both kidneys is required before a transplant, the patient must be maintained on dialysis until after transplantation. Since the transplant waiting list can exceed seven years in some areas, most patients arrange for a willing live donor before agreeing to a bilateral nephrectomy.46,47
Dialysis
Either peritoneal dialysis (PD) or hemodialysis (HD) can be offered to patients with severe ADPKD. Depending on the size of the native kidneys and the history of previous abdominal surgery, PD often offers a better chance of survival in these patients, particularly compared with patients who have ESRD associated with other causes.48
For management of the patient with ADPKD who receives PD, it can be difficult to differentiate between the pain of a cyst and the pain of a peritoneal infection. Generally, cyst rupture is accompanied by hematuria; and peritonitis, by cloudy fluid.5 Management provided by an experienced nephrologist and PD nurse is vital.
In ADPKD patients who undergo HD, too, survival is better than in patients who have ESRD with other causes49,50; five-year survival can be as high as 10% to 15%.51 This is likely due to the lower incidence of coronary artery disease in the ADPKD population, compared with patients who have ESRD associated with other chronic diseases.49
FUTURE TRENDS AND ONGOING TRIALS
HALT PKD29,30 is an NIH-funded, double-blind study to determine whether adding an angiotensin receptor blocker (ARB) to standard ACE inhibitor therapy results in a more significant decrease in the progression of renal cysts. The rationale for this is that the ARB is expected to block the renin-angiotensin-aldosterone system in the kidney. Use of ACE inhibitor monotherapy versus ARB/ACE inhibitor therapy is being compared in two study arms: patients between ages 15 and 49 with a GFR of 60 mL/min or greater; and patients between ages 18 and 64 with a GFR of 25 to 60 mL/min.29 To date, preliminary results indicate no benefit in adding the second medication.49
The TEMPO Trial52 is a multicenter, double-blind study looking at the effect of tolvaptan on renal cyst growth. Tolvaptan is a potent vasopressin receptor antagonist, and in vitro evidence has shown that intracellular cyclic adenosine monophosphate (cAMP) plays a large role in the development of cysts in patients with ADPKD. If it is possible to block the cAMP that is causing cyst growth, progression of ADPKD should slow.53,54 Only short-term effects of tolvaptan use are currently known.55
High Water Intake to Slow Progression of Polycystic Kidney Disease56 is an open-label, nonrandomized trial in which patients drink a minimum of
3 L of water. Previously, a small study showed that an increase in fluid intake partially suppresses the urine osmolality and the serum antidiuretic hormone (ADH) levels.57 According to this theory, increasing water intake to greater than 3 L/d may result in complete suppression of ADH and cAMP. This is a small study (n = 20),56 since patients with ADPKD are likely to have urinary concentrating defects, and hyponatremia is a concern is in these patients.58
Sirolimus and ADPKD59 is an open-label randomized study to see whether sirolimus (also known as rapamycin) can reduce cyst growth. Originally, it was noted that posttransplant ADPKD patients underwent a regression of both liver and kidney cysts when they were taking sirolimus, and a preliminary crossover study was done.60 However, preliminary results at 18 months showed no difference in renal growth or cyst growth but did show kidney damage as determined by an increase of proteinuria in the treatment group.59 The study is still in progress.
Somatostatin in Polycystic Kidney61 is a long-term (three-year) study following patients who agreed to participate in a randomized, double-blind protocol; in it, an intramuscular injection of either an octreotide (ie, somatastatin) or placebo was administered every four weeks for one year in an effort to reduce the size of kidney and liver cysts.62 At one year, the quality of life in the treatment group was rated better, as measured by pain reduction and improved physical activity. Cyst growth in the treatment group was smaller for both the kidney and liver. However, the GFR decreased to the same degree in both groups.62
CONCLUSION
ADPKD is a common, often overlooked genetic disease that is a cause of hypertension. ADPKD’s presenting symptoms of flank pain, back pain, and/or hematuria often bring the patient to the provider, but a high index of suspicion must be maintained to diagnose these patients at an early age. Due to the variable presentation even within affected families, many patients do not realize that their family carries the PKD gene.
While genetic testing is available, ultrasound is a quick, relatively inexpensive, and easy method to screen for this diagnosis. The progression of ADPKD to ESRD, requiring dialysis or organ transplantation, can be slowed with early and aggressive treatment of hypertension. As with all patients affected by renal impairment, suggestions for patients with ADPKD to avoid use of NSAIDs, contrast dye, and gadolinium-enhanced MRI are appropriate. The primary care PA or NP is in an appropriate position to see to this.
REFERENCES
1. Newschaffer CJ, Falb MD, Gurney JG. National autism prevalence trends from United States special education data. Pediatrics. 2005;115 (3):e277-e282.
2. Torres VE, Harris PC, Pirson Y. Autosomal dominant polycystic kidney disease. Lancet. 2007;369(9569):1287-1301.
3. The polycystic kidney disease 1 gene encodes a 14 kb transcript and lies within a duplicated region on chromosome 16: European Polycystic Kidney Disease Consortium. Cell. 1994;77(6):881-894.
4. Mochizuki T, Wu G, Hayashi T, et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science. 1996; 272(5266):1339-1342.
5. Chapman AB. Polycystic and other cystic kidney diseases. In: Greenberg A, ed. Primer on Kidney Diseases: Expert Consult. 5th ed. National Kidney Foundation; 2009:345-353.
6. Fischer E, Legue E, Doyen A, et al. Defective planar cell polarity in polycystic kidney disease. Nat Genet. 2006;38(1):21-23.
7. Murcia NS, Sweeney WE Jr, Avner ED. New insights into the molecular pathophysiology of polycystic kidney disease. Kidney Int. 1999;55 (4):1187-1197.
8. Paterson AD, Pei Y. Is there a third gene for autosomal dominant polycystic kidney disease? Kidney Int. 1998;54(5):1759-1761.
9. Pei Y. Practical genetics for autosomal dominant polycystic kidney disease. Nephron Clin Pract. 2011;118(1):c19-c30.
10. Tan YC, Blumenfeld J, Rennert H. Autosomal dominant polycystic kidney disease: genetics, mutations and microRNAs. Biochim Biophys Acta. 2011 Mar 16 [Epub ahead of print].
11. Avner ED, Sweeney WE Jr, Nelson WJ. Abnormal sodium pump distribution during renal tubulogenesis in congenital murine polycystic kidney disease. Proc Natl Acad Sci U S A. 1992;89(16):7447-7451.
12. Torra R, Badenas C, Darnell A, et al. Linkage, clinical features, and prognosis of autosomal dominant polycystic kidney disease types 1 and 2. J Am Soc Nephrol. 1996;7(10):2142-2151.
13. Davies F, Coles GA, Harper PS, et al. Polycystic kidney disease re-evaluated: a population-based study. Q J Med. 1991;79(290):477-485.
14. Hateboer N, v Dijk MA, Bogdanova N, et al; European PKD1-PKD2 Study Group. Comparison of phenotypes of polycystic kidney disease types 1 and 2. Lancet. 1999;353(9147):103-107.
15. Peters DJ, Breuning MH. Autosomal dominant polycystic kidney disease: modification of disease progression. Lancet. 2001;358(9291):1439-1444.
16. Dicks E, Ravani P, Langman D, et al. Incident renal events and risk factors in autosomal dominant polycystic kidney disease: a population- and family-based cohort followed for 22 years. Clin J Am Soc Nephrol. 2006;1(4):710-717.
17. Fick-Brosnahan GM, Belz MM, McFann KK, etc. Relationship between renal volume growth and renal function in autosomal dominant polycystic kidney disease: a longitudinal study. Am J Kidney Dis. 2002;39(6):1127-1134.
18. Fick-Brosnahan GM, Tran ZV, Johnson AM, et al. Progression of autosomal-dominant polycystic kidney disease in children. Kidney Int. 2001;59(5):1654-1662.
19. Johnson AM, Gabow PA. Identification of patients with autosomal dominant polycystic kidney disease at highest risk for end-stage renal disease. J Am Soc Nephrol. 1997;8(10): 1560-1567.
20. Risk D. Autosomal dominant polycystic kidney disease. Presented at: National Kidney Foundation, Spring Clinical Meetings; April 28, 2011; Las Vegas, NV.
21. Pei Y, Obaji J, Dupuis A, et al. Unified criteria for ultrasonographic diagnosis of ADPKD. J Am Soc Nephrol. 2009;20(1):205-212.
22. Watnick T, Germino GG. Molecular basis of autosomal dominant polycystic kidney disease. Semin Nephrol. 1999;19(4):327-343.
23. Ecder T, Schrier RW. Cardiovascular abnormalities in autosomal-dominant polycystic kidney disease. Nat Rev Nephrol. 2009;5(4):221-228.
24. Patch C, Charlton J, Roderick PJ, Gulliford MC. Use of antihypertensive medications and mortality of patients with autosomal dominant polycystic kidney disease: a population-based study. Am J Kidney Dis. 2011;57(6):856-862.
25. Pirson Y. Extrarenal manifestations of autosomal dominant polycystic kidney disease. Adv Chronic Kidney Dis. 2010;17(2):173-180.
26. Chapman AB, Johnson A, Gabow PA, Schrier RW. The renin-angiotensin-aldosterone system and autosomal dominant polycystic kidney disease. N Engl J Med. 1990;323(16):1091-1096.
27. Jafar TH, Stark PC, Schmid CH, et al. The effect of angiotensin-converting-enzyme inhibitors on progression of advanced polycystic kidney disease. Kidney Int. 2005;67(1):265-271.
28. Schrier RW. Renal volume, renin-angiotensin-aldosterone system, hypertension, and left ventricular hypertrophy in patients with autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 2009;20(9):1888-1893.
29. National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), NIH, sponsor. HALT PKD (Halt Progression of Polycystic Kidney Disease): Efficacy of Aggressive Renin-Angiotensin-Aldosterone Axis Blockade in Preventing/Slowing Renal Function Decline in ADPKD. www2.niddk.nih.gov/NR/rdonlyres/175578F6-62B4-429A-9BBF-96CCEC2FFB3A/0/KUHHALT PKDPROTOCOL9107.pdf. Accessed July 22, 2011.
30. Chapman AB, Torres VE, Perrone RD, et al. The HALT polycystic kidney disease trials: design and implementation. Clin J Am Soc Nephrol. 2010;5(1):102-109.
31. Taylor M, Johnson AM, Tison M, et al. Earlier diagnosis of autosomal dominant polycystic kidney disease: importance of family history and implications for cardiovascular and renal complications. Am J Kidney Dis. 2005;46(3):415-423.
32. Namli S, Oflaz H, Turgut F, et al. Improvement of endothelial dysfunction with simvastatin in patients with autosomal dominant polycystic kidney disease. Ren Fail. 2007;29(1):55-59.
33. Bremmer MS, Jacobs SC. Renal artery embolization for the symptomatic treatment of adult polycystic kidney disease. Nat Clin Pract Nephrol. 2008;4(5):236-237.
34. Chapman AB, Rubinstein D, Hughes R, et al. Intracranial aneurysms in autosomal dominant polycystic kidney disease. N Engl J Med. 1992; 327(13):916-920.
35. Schievink WI, Torres VE, Piepgras DG, Wiebers DO. Saccular intracranial aneurysms in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 1992;3(1):88-95.
36. Fick GM, Gabow PA. Hereditary and acquired cystic disease of the kidney. Kidney Int. 1994;46(4):951-964.
37. Watson ML. Complications of polycystic kidney disease. Kidney Int. 1997;51(1):353-365.
38. Huston J 3rd, Torres VE, Sulivan PP, et al. Value of magnetic resonance angiography for the detection of intracranial aneurysms in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 1993;3(12):1871-1877.
39. Longenecker JC, Coresh J, Powe NR, et al. Traditional cardiovascular disease risk factors in dialysis patients compared with the general population: the CHOICE Study. J Am Soc Nephrol. 2002;13(7):1918-1927.
40. Meijer E, Rook M, Tent H, et al. Early renal abnormalities in autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol. 2010; 5(6):1091-1098.
41. Torres VE, Harris PC. Autosomal dominant polycystic kidney disease: the last 3 years. Kidney Int. 2009;76(2):149-168.
42. Tabibi A, Simforoosh N, Abadpour P, et al. Concomitant nephrectomy of massively enlarged kidneys and renal transplantation in autosomal dominant polycystic kidney disease. Transplant Proc. 2005;37(7):2939-2940.
43. Dunn MD, Portis AJ, Elbahnasy AM, et al. Laparoscopic nephrectomy in patients with end-stage renal disease and autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2000;35(4):720-725.
44. Sulikowski T, Tejchman K, Zietek Z, et al. Experience with autosomal dominant polycystic kidney disease in patients before and after renal transplantation: a 7-year observation. Transplant Proc. 2009;41(1):177-180.
45. Desai MR, Nandkishore SK, Ganpule A, Thimmegowda M. Pretransplant laparoscopic nephrectomy in adult polycystic kidney disease: a single centre experience. BJU Int. 2008;101 (1):94-97.
46. Glassman DT, Nipkow L, Bartlett ST, Jacobs SC. Bilateral nephrectomy with concomitant renal graft transplantation for autosomal dominant polycystic kidney disease. J Urol. 2000;164 (3 pt 1):661-664.
47. Fuller TF, Brennan TV, Feng S, et al. End stage polycystic kidney disease: indications and timing of native nephrectomy relative to kidney transplantation. J Urol. 2005;174(6):2284-2288.
48. Abbott KC, Agodoa LY. Polycystic kidney disease at end-stage renal disease in the United States: patient characteristics and survival. Clin Nephrol. 2002;57(3):208-214.
49. Perrone RD, Ruthazer R, Terrin NC. Survival after end-stage renal disease in autosomal dominant polycystic kidney disease: contribution of extrarenal complications to mortality. Am J Kidney Dis. 2001;38(4):777-784.
50. Batista PB, Lopes AA, Costa FA. Association between attributed cause of end-stage renal disease and risk of death in Brazilian patients receiving renal replacement therapy. Ren Fail. 2005;27(6):651-656.
51. Pirson Y, Christophe JL, Goffin E. Outcome of renal replacement therapy in autosomal dominant polycystic kidney disease. Nephrol Dial Transplant. 1996;11 suppl 6:24-28.
52. Torres VE, Meijer E, Bae KT, et al. Rationale and design of the TEMPO (Tolvaptan Efficacy and Safety in Management of Autosomal Dominant Polycystic Kidney Disease and its Outcomes) 3-4 Study. Am J Kidney Dis. 2011;57(5):692-699.
53. Calvet JP. Strategies to inhibit cyst formation in ADPKD. Clin J Am Soc Nephrol. 2008;3 (4):1205-1211.
54. Grantham JJ. Lillian Jean Kaplan International Prize for advancement in the understanding of polycystic kidney disease. Understanding polycystic kidney disease: a systems biology approach. Kidney Int. 2003;64(4):1157-1162.
55. Irazabal MV, Torres VE, Hogan MC, et al. Short-term effects of tolvaptan on renal function and volume in patients with Autosomal Dominant Polycystic Kidney Disease. Kidney Int. 2011 May 4 [Epub ahead of print].
56. New York University, sponsor. High Water Intake to Slow Progression of Polycystic Kidney Disease. http://clinicaltrials.gov/ct2/show/NCT00784030. Accessed July 22, 2011.
57. Wang CJ, Creed C, Winklhofer FT, Grantham JJ. Water prescription in autosomal dominant polycystic kidney disease: a pilot study. Clin J Am Soc Nephrol. 2011;6(1):192-197.
58. Grampsas SA, Chandhoke Ps, Fan J, et al. Anatomic and metabolic risk factors for nephrolithiasis in patients with autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2000;36(1):53-57.
59. Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney growth in autosomal dominant polycystic kidney disease. N Engl J Med. 2010; 363(9):820-829.
60. Perico N, Antiga L, Caroli A, et al. Sirolimus therapy to halt the progression of ADPKD. J Am Soc Nephrol. 2010;21(6):1031-1040.
61. Mario Negri Institute for Pharmacological Research, sponsor. Somatostatin in Polycystic Kidney: a Long-term Three Year Follow up Study. http://clinicaltrials.gov/ct2/show/NCT00309283. Accessed July 22, 2011.
62. Hogan MC, Masyuk TV, Page LJ, et al. Randomized clinical trial of long-acting somatostatin for autosomal dominant polycystic kidney and liver disease. J Am Soc Nephrol. 2010; 21(6):1052-1061.
Twice as common as autism and half as well-known,1 autosomal polycystic kidney disease (ADPKD) occurs in one in 400 to one in 1,000 people.2 It is an inherited progressive genetic disorder that causes hypertension and decreased renal function and, over time, can lead to kidney failure. Two polycstin genes that code for ADPKD, PKD1 and PKD2, were identified in 1994 and 1996, respectively.3,4 Awareness and understanding of the genes responsible for ADPKD have increased clinicians’ ability to identify at-risk patients and to slow or alter the course of the disease.
Case Presentation
A 45-year-old black man presents to your office with severe, nonradiating back pain and new-onset hypertension. Regarding the pain, he stated, “I turned around to see who kicked me, but no one was there.” When the pain began, he went to see the nurse at the school where he is employed, and she found that his blood pressure was high at 162/90 mm Hg. Although the patient’s back pain is resolving, he is very concerned about his blood pressure, since he has never had a high reading before.
He is the baseball coach and physical education teacher at the local high school and is in excellent physical condition as a result of his professional interaction with teenagers every day. He does not smoke or use any illicit drugs but does admit to occasional alcohol consumption. His medical history is significant only for occasional broken fingers and twisted ankles, all occurring while he was engaged in sports.
His family history includes one brother without medical problems, a brother and a sister with hypertension, a sister with diabetes and obesity, and a brother with a congenital abnormality that required a living donor kidney transplant at age 17 (the father served as donor). No family-wide workup has ever been done because no one practitioner has ever made a connection among these conditions and considered a diagnosis of ADPKD.
The patient’s blood pressure in the office is 172/92 mm Hg while sitting and 166/88 mm Hg while standing. He is somewhat sore with a localized spasm in the lumbar-sacral area but no radiation of pain. The patient has trouble touching his toes but reports that he can never touch his toes. His straight leg lift is negative. The rest of his physical exam is noncontributory.
What should be the next step in this patient’s workup?
PATHOPHYSIOLOGY
ADPKD is a progressive expansion of numerous fluid-filled cysts that result in massive enlargement of the kidneys.5 Less than 5% of all nephrons become cystic; however, the average volume of a polycystic kidney is 1,000 mL (normal, 300 mL), that is, the volume of a standard-sized pineapple. Even with this significant enlargement, a decline in the glomerular filtration rate (GFR) is not usually seen initially. Each cyst is derived from a single hyperproliferative epithelial cell. Increased cellular proliferation, followed by fluid secretion and alterations in the extracellular matrix, cause an outpouching from the parent nephron, which eventually detaches from the parent nephron and continues to enlarge and autonomously secrete fluid.6,7
PKD1 and PKD2 are two genes responsible for ADPKD that have been isolated so far. Since there are families carrying neither the PKD1 nor the PKD2 gene that still have an inherited type of ADPKD, there is suspicion that at least one more PKD gene, not yet isolated, exists.8 It is also possible that other genetic or environmental factors may be at play.9,10
In 1994, the PKD1 gene was isolated on chromosome 16,3 and it was found to code for polycystin 1. A lack of polycystin 1 causes an abnormality in the Na+/K(+)-ATPase pumps, leading to abnormal sodium reabsorption.11 How and why this happens is not quite clear. However, the hypertension that is a key objective finding in patients with ADPKD is thought to result from this pump abnormality.
PKD2 is found on the long arm of chromosome 4 and codes for polycystin 2.4 Polycystin 2 is an amino acid that is responsible for voltage-activated cellular calcium channels,5 again explaining the hypertension so commonly seen in the course of ADPKD. ADPKD-associated hypertension may be present as early as the teenage years.12
EPIDEMIOLOGY
More than 85% of ADPKD cases are associated with PKD1, and this form is called polycystic kidney disease 1 (PKD 1), the more aggressive form of the disease.13,14 PKD 2 (the form associated with the gene PKD2), though less common, is also likely to progress to end-stage renal disease (ESRD), but at a later age (median age of 74 years, compared with 54 in patients with PKD 1).14 ADPKD accounts for about 5% of cases of ESRD in North America,9 but for most patients, presentation and decreased renal function do not occur until the 40s.15 However, patients with the risk factors listed in Table 15,16-19 are likely to experience a more rapid and aggressive form of the disease.
Even with the same germline mutation in a family with this inherited disease, the severity of ADPKD among family members is quite variable; this is true even in the case of twins.9,10,20 Since the age and symptoms at presentation can vary so greatly, a uniform method of identifying patients with ADPKD, along with staging, was needed. Most patients do not undergo genetic testing (ie, DNA linkage or gene-based direct sequencing9) for a diagnosis of ADPKD or to differentiate between the PKD 1 and PKD 2 disease forms unless they are participating in a research study. Diagnostic criteria were needed that were applicable for any type of ADPKD.
In 2009, the University of Toronto’s Division of Nephrology convened experts in the fields of nephrology and radiology to reach a consensus on standardized ultrasonographic diagnostic criteria.21 They formulated definitions based on a study of 948 individuals who were at risk for either PKD 1 or PKD 2 (see Table 221). The specificity and sensitivity of the resulting criteria range from 82% to 100%, making it possible to standardize the care and classification of renal patients worldwide.
Since family members with the same genotypes can experience very divergent disease manifestations, the two-hit hypothesis has been developed.22 In simple terms, it proposes that after the germline mutation (PKD1 or PKD2), there is a second somatic mutation that leads to progressive cyst formation; when the number and size of cysts increase, the patient starts to experience symptoms of ADPKD.22
Age at presentation can be quite variable, as can the presenting symptoms. Most patients with PKD 1 present in their 50s, with 54 being the average age in US patients.14 The most common presenting symptom is flank or back pain.2,5 The pain is due to the massive enlargement of the kidneys, causing a stretching of the kidney capsule and leading to a chronic, dull and persistent pain in the low back. Severe pain, sharp and cutting, occurs when one of the cysts hemorrhages; to some patients, the pain resembles a quick, powerful “kick in the back.” Hematuria can occur following cyst hemorrhage; depending on the location of the cyst that burst within the kidney (ie, how close it is to the collecting system) and how large it is, the amount and color of the hematuria can be impressive.
ADPKD is more common in men than women, and cyst rupture can be precipitated by trauma or lifting heavy objects. Cyst hemorrhage can turn the urine bright red, which is especially frightening to the male patient. Hematuria is often the key presenting symptom in patients who will be diagnosed with ADPKD-induced hypertension.
Besides hematuria, other common manifestations of ADPKD include:
• Hypertension (60% of affected patients, which increases to 100% by the time ESRD develops)
• Extrarenal cysts (100% of affected patients)
• Urinary tract infections
• Nephrolithiasis (20% of affected patients)
• Proteinuria, occasionally (18% of affected patients).2,5,23
Among these manifestations, those most commonly attributed to a diagnosis of ADPKD are hypertension, kidney stones, and urinary tract or kidney infections. Since isolated proteinuria is unusual in patients with ADPKD, it is recommended that another cause of kidney disease be explored in patients with this presentation.24
Extrarenal manifestations of cyst development are common, eventually occurring in all patients as they age. Hepatic cysts are universal in patients with ADPKD by age 30, although hepatic function is preserved. There may be a mild elevation in the alkaline phosphatase level in patients with ADPKD, resulting from the presence of hepatic cysts. Cysts may also be found in the pancreas, spleen, thyroid, and epididymis.5,25 Some patients may complain of dyspnea, pain, early satiety, or lower extremity edema as a result of enlarged cyst.
The Case Patient
Because you recently attended a lecture about ADPKD, you are aware that flank pain in men with hypertension is indicative of ADPKD until proven otherwise. Believing that this patient’s hypertension is renal in origin, you order an abdominal ultrasound. You also order a comprehensive metabolic panel and a complete blood count. The patient’s GFR is measured at 89 mL/min (indicative of stage 2 kidney disease). Other results are shown in Table 3.
The very broad differential includes essential hypertension, hypertension resulting from intake of “power drinks” or salt in an athlete, illicit use of medications (including steroids), herniated disc leading to transient hypertension, and urinary tract infection or sexually transmitted disease. All of this is moot when the ultrasound shows both kidneys measuring greater than 15 cm, with four distinct cysts on the right kidney and three distinct cysts on the left.
You explain to the patient that ADPKD is a genetic disease and that he and his siblings each had a certain chance of inheriting it. Although different presentations may occur (“congenital” polycystic kidney disease, hypertension, or obesity), they all must undergo ultrasonographic screening for ADPKD. You add that although ADPKD is a genetic disease, it can also be diagnosed in different members of the same family at different ages.
TREATMENT
The goal of treatment for the patient with ADPKD is to slow cyst development and the natural course of the disease. If this can be achieved, the need for dialysis or kidney transplantation may be postponed for a number of years. Because cyst growth causes an elevation in renin and activates the angiotensin II renin system26 (see figure,24), an ACE inhibitor is the most effective treatment to lower blood pressure and thus slow the progression of ADPKD. Most patients with ADPKD are started on an ACE inhibitor at an early age to slow the rate of disease progression.27,28 Several studies are under way to determine the best antihypertensive medication and the optimal age for initiating treatment.29,30
Lipid screening and treatment for dyslipidemia are important23 because ADPKD can lead to a reduction in kidney function, resulting in chronic kidney disease (CKD). CKD is considered a coronary heart disease risk equivalent, and most professionals will treat the patient with ADPKD for hyperlipidemia.23,31 While there are no data showing that statin use will reduce the incidence of ESRD or delay the need for dialysis or kidney transplantation in patients with ADPKD, the beneficial effects of good renal blood flow and endothelial function have been noted.32,33
One of the most common and significant complications in ADPKD is intracranial hemorrhage resulting from a ruptured cerebral aneurysm. In the younger adult, the incidence of cerebral aneurysm is 4%, but incidence increases to 10% in patients older than 65.34-36 Family clusters of aneurysms have been reported.37 If an intracranial aneurysm is found in the family history, the risk of an aneurysm in another family member increases to 22%.38
Since rupture of an intracranial hemorrhage is associated with a 30-day mortality rate of 50% and 80% morbidity,5,38 standard of care for patients with ADPKD includes CT or magnetic resonance angiographic (MRA) screening in the asymptomatic patient with a positive family history.34,38 If an aneurysm is found, the lifetime chance of rupture is 50%, although the risk is greater in the case of an aneurysm larger than 10 mm.5
As in all patients with kidney disease, left ventricular hypertrophy is common among patients with ADPKD.23,28,39
The Case Patient
The patient is started on an ACE inhibitor, scheduled for fasting lipid screening, and referred to a nephrology practice for disease management. As research and investigation of possible treatment options for ADPKD are ongoing, the patient may benefit from participating in a new research protocol.
Because the patient’s family has no history of cerebral aneurysm, CT/MRA screening is not required. A discussion of the pros and cons of genetic testing for the entire family, including nieces and nephews, is initiated. The patient and his family are referred to a genetic counselor to decide whether the benefit of early treatment for hypertension outweighs the risk of carrying a diagnosis of ADPKD for his younger relatives, who may later seek health insurance coverage.
NATURAL PROGRESSION OF ADPKD
Hypertension and cyst formation will continue as the patient ages. The natural progression of ADPKD is to renal failure with renal replacement therapy (dialysis or organ transplantation) as treatment options. If the progression of ADPKD can be slowed through pharmacotherapy, the patient may live for many years without needing dialysis. This ideal can be accomplished only through aggressive hypertension control, which should be started in the teenage years.23,30,31
Suggestions to increase fluid consumption and to limit the use of NSAIDs, contrast dye, and MRI with gadolinium are appropriate. It is rare for hypertension to be diagnosed before some organ damage has already occurred.12 Often the patient’s renal function, as determined by measuring the GFR, remains stable until the patient reaches his or her 40s.40 However, kidney damage often begins before any detectable change in GFR. Once the GFR does start to decline, the average decrease is 4.4 to 5.9 mL/min/1.73m2 each year.41
MANAGEMENT CONSIDERATIONS
For ESRD Organ Transplantation
Kidney transplantation—the only curative treatment for ADPKD—can be offered to patients once the GFR falls below 20 mL/min. However, the patient with ADPKD can experience kidney enlargement to such an extent that introducing a third kidney into the limited abdominal space becomes technically difficult. Although nephrectomy is avoided whenever possible, there are cases in which there is no alternative.42
In addition to space concerns, recurrent urinary tract infections, chronic pain, renal cell carcinoma, chronic hematuria, or chronic cyst infections can necessitate a nephrectomy.43,44 A laparoscopic approach with decompression of cysts or removal of only one kidney is preferred.43,45 If removal of both kidneys is required before a transplant, the patient must be maintained on dialysis until after transplantation. Since the transplant waiting list can exceed seven years in some areas, most patients arrange for a willing live donor before agreeing to a bilateral nephrectomy.46,47
Dialysis
Either peritoneal dialysis (PD) or hemodialysis (HD) can be offered to patients with severe ADPKD. Depending on the size of the native kidneys and the history of previous abdominal surgery, PD often offers a better chance of survival in these patients, particularly compared with patients who have ESRD associated with other causes.48
For management of the patient with ADPKD who receives PD, it can be difficult to differentiate between the pain of a cyst and the pain of a peritoneal infection. Generally, cyst rupture is accompanied by hematuria; and peritonitis, by cloudy fluid.5 Management provided by an experienced nephrologist and PD nurse is vital.
In ADPKD patients who undergo HD, too, survival is better than in patients who have ESRD with other causes49,50; five-year survival can be as high as 10% to 15%.51 This is likely due to the lower incidence of coronary artery disease in the ADPKD population, compared with patients who have ESRD associated with other chronic diseases.49
FUTURE TRENDS AND ONGOING TRIALS
HALT PKD29,30 is an NIH-funded, double-blind study to determine whether adding an angiotensin receptor blocker (ARB) to standard ACE inhibitor therapy results in a more significant decrease in the progression of renal cysts. The rationale for this is that the ARB is expected to block the renin-angiotensin-aldosterone system in the kidney. Use of ACE inhibitor monotherapy versus ARB/ACE inhibitor therapy is being compared in two study arms: patients between ages 15 and 49 with a GFR of 60 mL/min or greater; and patients between ages 18 and 64 with a GFR of 25 to 60 mL/min.29 To date, preliminary results indicate no benefit in adding the second medication.49
The TEMPO Trial52 is a multicenter, double-blind study looking at the effect of tolvaptan on renal cyst growth. Tolvaptan is a potent vasopressin receptor antagonist, and in vitro evidence has shown that intracellular cyclic adenosine monophosphate (cAMP) plays a large role in the development of cysts in patients with ADPKD. If it is possible to block the cAMP that is causing cyst growth, progression of ADPKD should slow.53,54 Only short-term effects of tolvaptan use are currently known.55
High Water Intake to Slow Progression of Polycystic Kidney Disease56 is an open-label, nonrandomized trial in which patients drink a minimum of
3 L of water. Previously, a small study showed that an increase in fluid intake partially suppresses the urine osmolality and the serum antidiuretic hormone (ADH) levels.57 According to this theory, increasing water intake to greater than 3 L/d may result in complete suppression of ADH and cAMP. This is a small study (n = 20),56 since patients with ADPKD are likely to have urinary concentrating defects, and hyponatremia is a concern is in these patients.58
Sirolimus and ADPKD59 is an open-label randomized study to see whether sirolimus (also known as rapamycin) can reduce cyst growth. Originally, it was noted that posttransplant ADPKD patients underwent a regression of both liver and kidney cysts when they were taking sirolimus, and a preliminary crossover study was done.60 However, preliminary results at 18 months showed no difference in renal growth or cyst growth but did show kidney damage as determined by an increase of proteinuria in the treatment group.59 The study is still in progress.
Somatostatin in Polycystic Kidney61 is a long-term (three-year) study following patients who agreed to participate in a randomized, double-blind protocol; in it, an intramuscular injection of either an octreotide (ie, somatastatin) or placebo was administered every four weeks for one year in an effort to reduce the size of kidney and liver cysts.62 At one year, the quality of life in the treatment group was rated better, as measured by pain reduction and improved physical activity. Cyst growth in the treatment group was smaller for both the kidney and liver. However, the GFR decreased to the same degree in both groups.62
CONCLUSION
ADPKD is a common, often overlooked genetic disease that is a cause of hypertension. ADPKD’s presenting symptoms of flank pain, back pain, and/or hematuria often bring the patient to the provider, but a high index of suspicion must be maintained to diagnose these patients at an early age. Due to the variable presentation even within affected families, many patients do not realize that their family carries the PKD gene.
While genetic testing is available, ultrasound is a quick, relatively inexpensive, and easy method to screen for this diagnosis. The progression of ADPKD to ESRD, requiring dialysis or organ transplantation, can be slowed with early and aggressive treatment of hypertension. As with all patients affected by renal impairment, suggestions for patients with ADPKD to avoid use of NSAIDs, contrast dye, and gadolinium-enhanced MRI are appropriate. The primary care PA or NP is in an appropriate position to see to this.
REFERENCES
1. Newschaffer CJ, Falb MD, Gurney JG. National autism prevalence trends from United States special education data. Pediatrics. 2005;115 (3):e277-e282.
2. Torres VE, Harris PC, Pirson Y. Autosomal dominant polycystic kidney disease. Lancet. 2007;369(9569):1287-1301.
3. The polycystic kidney disease 1 gene encodes a 14 kb transcript and lies within a duplicated region on chromosome 16: European Polycystic Kidney Disease Consortium. Cell. 1994;77(6):881-894.
4. Mochizuki T, Wu G, Hayashi T, et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science. 1996; 272(5266):1339-1342.
5. Chapman AB. Polycystic and other cystic kidney diseases. In: Greenberg A, ed. Primer on Kidney Diseases: Expert Consult. 5th ed. National Kidney Foundation; 2009:345-353.
6. Fischer E, Legue E, Doyen A, et al. Defective planar cell polarity in polycystic kidney disease. Nat Genet. 2006;38(1):21-23.
7. Murcia NS, Sweeney WE Jr, Avner ED. New insights into the molecular pathophysiology of polycystic kidney disease. Kidney Int. 1999;55 (4):1187-1197.
8. Paterson AD, Pei Y. Is there a third gene for autosomal dominant polycystic kidney disease? Kidney Int. 1998;54(5):1759-1761.
9. Pei Y. Practical genetics for autosomal dominant polycystic kidney disease. Nephron Clin Pract. 2011;118(1):c19-c30.
10. Tan YC, Blumenfeld J, Rennert H. Autosomal dominant polycystic kidney disease: genetics, mutations and microRNAs. Biochim Biophys Acta. 2011 Mar 16 [Epub ahead of print].
11. Avner ED, Sweeney WE Jr, Nelson WJ. Abnormal sodium pump distribution during renal tubulogenesis in congenital murine polycystic kidney disease. Proc Natl Acad Sci U S A. 1992;89(16):7447-7451.
12. Torra R, Badenas C, Darnell A, et al. Linkage, clinical features, and prognosis of autosomal dominant polycystic kidney disease types 1 and 2. J Am Soc Nephrol. 1996;7(10):2142-2151.
13. Davies F, Coles GA, Harper PS, et al. Polycystic kidney disease re-evaluated: a population-based study. Q J Med. 1991;79(290):477-485.
14. Hateboer N, v Dijk MA, Bogdanova N, et al; European PKD1-PKD2 Study Group. Comparison of phenotypes of polycystic kidney disease types 1 and 2. Lancet. 1999;353(9147):103-107.
15. Peters DJ, Breuning MH. Autosomal dominant polycystic kidney disease: modification of disease progression. Lancet. 2001;358(9291):1439-1444.
16. Dicks E, Ravani P, Langman D, et al. Incident renal events and risk factors in autosomal dominant polycystic kidney disease: a population- and family-based cohort followed for 22 years. Clin J Am Soc Nephrol. 2006;1(4):710-717.
17. Fick-Brosnahan GM, Belz MM, McFann KK, etc. Relationship between renal volume growth and renal function in autosomal dominant polycystic kidney disease: a longitudinal study. Am J Kidney Dis. 2002;39(6):1127-1134.
18. Fick-Brosnahan GM, Tran ZV, Johnson AM, et al. Progression of autosomal-dominant polycystic kidney disease in children. Kidney Int. 2001;59(5):1654-1662.
19. Johnson AM, Gabow PA. Identification of patients with autosomal dominant polycystic kidney disease at highest risk for end-stage renal disease. J Am Soc Nephrol. 1997;8(10): 1560-1567.
20. Risk D. Autosomal dominant polycystic kidney disease. Presented at: National Kidney Foundation, Spring Clinical Meetings; April 28, 2011; Las Vegas, NV.
21. Pei Y, Obaji J, Dupuis A, et al. Unified criteria for ultrasonographic diagnosis of ADPKD. J Am Soc Nephrol. 2009;20(1):205-212.
22. Watnick T, Germino GG. Molecular basis of autosomal dominant polycystic kidney disease. Semin Nephrol. 1999;19(4):327-343.
23. Ecder T, Schrier RW. Cardiovascular abnormalities in autosomal-dominant polycystic kidney disease. Nat Rev Nephrol. 2009;5(4):221-228.
24. Patch C, Charlton J, Roderick PJ, Gulliford MC. Use of antihypertensive medications and mortality of patients with autosomal dominant polycystic kidney disease: a population-based study. Am J Kidney Dis. 2011;57(6):856-862.
25. Pirson Y. Extrarenal manifestations of autosomal dominant polycystic kidney disease. Adv Chronic Kidney Dis. 2010;17(2):173-180.
26. Chapman AB, Johnson A, Gabow PA, Schrier RW. The renin-angiotensin-aldosterone system and autosomal dominant polycystic kidney disease. N Engl J Med. 1990;323(16):1091-1096.
27. Jafar TH, Stark PC, Schmid CH, et al. The effect of angiotensin-converting-enzyme inhibitors on progression of advanced polycystic kidney disease. Kidney Int. 2005;67(1):265-271.
28. Schrier RW. Renal volume, renin-angiotensin-aldosterone system, hypertension, and left ventricular hypertrophy in patients with autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 2009;20(9):1888-1893.
29. National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), NIH, sponsor. HALT PKD (Halt Progression of Polycystic Kidney Disease): Efficacy of Aggressive Renin-Angiotensin-Aldosterone Axis Blockade in Preventing/Slowing Renal Function Decline in ADPKD. www2.niddk.nih.gov/NR/rdonlyres/175578F6-62B4-429A-9BBF-96CCEC2FFB3A/0/KUHHALT PKDPROTOCOL9107.pdf. Accessed July 22, 2011.
30. Chapman AB, Torres VE, Perrone RD, et al. The HALT polycystic kidney disease trials: design and implementation. Clin J Am Soc Nephrol. 2010;5(1):102-109.
31. Taylor M, Johnson AM, Tison M, et al. Earlier diagnosis of autosomal dominant polycystic kidney disease: importance of family history and implications for cardiovascular and renal complications. Am J Kidney Dis. 2005;46(3):415-423.
32. Namli S, Oflaz H, Turgut F, et al. Improvement of endothelial dysfunction with simvastatin in patients with autosomal dominant polycystic kidney disease. Ren Fail. 2007;29(1):55-59.
33. Bremmer MS, Jacobs SC. Renal artery embolization for the symptomatic treatment of adult polycystic kidney disease. Nat Clin Pract Nephrol. 2008;4(5):236-237.
34. Chapman AB, Rubinstein D, Hughes R, et al. Intracranial aneurysms in autosomal dominant polycystic kidney disease. N Engl J Med. 1992; 327(13):916-920.
35. Schievink WI, Torres VE, Piepgras DG, Wiebers DO. Saccular intracranial aneurysms in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 1992;3(1):88-95.
36. Fick GM, Gabow PA. Hereditary and acquired cystic disease of the kidney. Kidney Int. 1994;46(4):951-964.
37. Watson ML. Complications of polycystic kidney disease. Kidney Int. 1997;51(1):353-365.
38. Huston J 3rd, Torres VE, Sulivan PP, et al. Value of magnetic resonance angiography for the detection of intracranial aneurysms in autosomal dominant polycystic kidney disease. J Am Soc Nephrol. 1993;3(12):1871-1877.
39. Longenecker JC, Coresh J, Powe NR, et al. Traditional cardiovascular disease risk factors in dialysis patients compared with the general population: the CHOICE Study. J Am Soc Nephrol. 2002;13(7):1918-1927.
40. Meijer E, Rook M, Tent H, et al. Early renal abnormalities in autosomal dominant polycystic kidney disease. Clin J Am Soc Nephrol. 2010; 5(6):1091-1098.
41. Torres VE, Harris PC. Autosomal dominant polycystic kidney disease: the last 3 years. Kidney Int. 2009;76(2):149-168.
42. Tabibi A, Simforoosh N, Abadpour P, et al. Concomitant nephrectomy of massively enlarged kidneys and renal transplantation in autosomal dominant polycystic kidney disease. Transplant Proc. 2005;37(7):2939-2940.
43. Dunn MD, Portis AJ, Elbahnasy AM, et al. Laparoscopic nephrectomy in patients with end-stage renal disease and autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2000;35(4):720-725.
44. Sulikowski T, Tejchman K, Zietek Z, et al. Experience with autosomal dominant polycystic kidney disease in patients before and after renal transplantation: a 7-year observation. Transplant Proc. 2009;41(1):177-180.
45. Desai MR, Nandkishore SK, Ganpule A, Thimmegowda M. Pretransplant laparoscopic nephrectomy in adult polycystic kidney disease: a single centre experience. BJU Int. 2008;101 (1):94-97.
46. Glassman DT, Nipkow L, Bartlett ST, Jacobs SC. Bilateral nephrectomy with concomitant renal graft transplantation for autosomal dominant polycystic kidney disease. J Urol. 2000;164 (3 pt 1):661-664.
47. Fuller TF, Brennan TV, Feng S, et al. End stage polycystic kidney disease: indications and timing of native nephrectomy relative to kidney transplantation. J Urol. 2005;174(6):2284-2288.
48. Abbott KC, Agodoa LY. Polycystic kidney disease at end-stage renal disease in the United States: patient characteristics and survival. Clin Nephrol. 2002;57(3):208-214.
49. Perrone RD, Ruthazer R, Terrin NC. Survival after end-stage renal disease in autosomal dominant polycystic kidney disease: contribution of extrarenal complications to mortality. Am J Kidney Dis. 2001;38(4):777-784.
50. Batista PB, Lopes AA, Costa FA. Association between attributed cause of end-stage renal disease and risk of death in Brazilian patients receiving renal replacement therapy. Ren Fail. 2005;27(6):651-656.
51. Pirson Y, Christophe JL, Goffin E. Outcome of renal replacement therapy in autosomal dominant polycystic kidney disease. Nephrol Dial Transplant. 1996;11 suppl 6:24-28.
52. Torres VE, Meijer E, Bae KT, et al. Rationale and design of the TEMPO (Tolvaptan Efficacy and Safety in Management of Autosomal Dominant Polycystic Kidney Disease and its Outcomes) 3-4 Study. Am J Kidney Dis. 2011;57(5):692-699.
53. Calvet JP. Strategies to inhibit cyst formation in ADPKD. Clin J Am Soc Nephrol. 2008;3 (4):1205-1211.
54. Grantham JJ. Lillian Jean Kaplan International Prize for advancement in the understanding of polycystic kidney disease. Understanding polycystic kidney disease: a systems biology approach. Kidney Int. 2003;64(4):1157-1162.
55. Irazabal MV, Torres VE, Hogan MC, et al. Short-term effects of tolvaptan on renal function and volume in patients with Autosomal Dominant Polycystic Kidney Disease. Kidney Int. 2011 May 4 [Epub ahead of print].
56. New York University, sponsor. High Water Intake to Slow Progression of Polycystic Kidney Disease. http://clinicaltrials.gov/ct2/show/NCT00784030. Accessed July 22, 2011.
57. Wang CJ, Creed C, Winklhofer FT, Grantham JJ. Water prescription in autosomal dominant polycystic kidney disease: a pilot study. Clin J Am Soc Nephrol. 2011;6(1):192-197.
58. Grampsas SA, Chandhoke Ps, Fan J, et al. Anatomic and metabolic risk factors for nephrolithiasis in patients with autosomal dominant polycystic kidney disease. Am J Kidney Dis. 2000;36(1):53-57.
59. Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney growth in autosomal dominant polycystic kidney disease. N Engl J Med. 2010; 363(9):820-829.
60. Perico N, Antiga L, Caroli A, et al. Sirolimus therapy to halt the progression of ADPKD. J Am Soc Nephrol. 2010;21(6):1031-1040.
61. Mario Negri Institute for Pharmacological Research, sponsor. Somatostatin in Polycystic Kidney: a Long-term Three Year Follow up Study. http://clinicaltrials.gov/ct2/show/NCT00309283. Accessed July 22, 2011.
62. Hogan MC, Masyuk TV, Page LJ, et al. Randomized clinical trial of long-acting somatostatin for autosomal dominant polycystic kidney and liver disease. J Am Soc Nephrol. 2010; 21(6):1052-1061.
Medications and the Renal Patient: TMP-SMX in Kidney Donor
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I was treating a patient for an uncomplicated urinary tract infection with trimethoprim–sulfamethoxazole (TMP-SMX). The pharmacist called and said the patient could not have any sulfa medications because she was going to donate a kidney to her brother. How do you explain this, and what should be my next step?
There is limited available evidence to support an answer to this question. Instead, I will provide a brief report of the available evidence, followed by the opinions of five experts in kidney transplantation.
TMP-SMX is a combination of two antimicrobial agents that act synergistically against a variety of bacteria.9 TMP decreases urinary excretion of potassium, leading to hyperkalemia—especially in persons with kidney disease or in those also taking other drugs that cause hyperkalemia.10 This scenario is unlikely, because the transplant team would have assessed the potential donor’s kidney function. When dosing the drug, clinicians should consider renal function and adjust the dose in those with a creatinine clearance less than or equal to 30 mL/min. Nephrotoxicity is uncommon in patients who take this drug; however, TMP is known to decrease tubular secretion of creatinine and may interfere with certain creatinine assays, leading to an artificial rise in serum creatinine. This is not reflective of a true reduction in the GFR and often is mild and reversible with discontinuation of the medication.
Querying experienced transplant professionals (two nephrologists, two transplant coordinators, and one doctoral-prepared pharmacist) yielded similar results. They all agreed that the only plausible reason to withhold TMP-SMX from this potential kidney donor was the risk of a transient rise in creatinine due to impaired secretion associated with TMP use. Many transplant teams recommend avoiding any medications that may affect the kidneys. So considering the lack of available evidence, I would recommend that you consult with the transplant team where your patient’s brother is receiving care before you prescribe TMP-SMX.
Debra Hain, PhD, APRN, GNP-BC
Florida Atlantic University, Boca Raton; Cleveland Clinic Florida; Department of Nephrology, Weston
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I was treating a patient for an uncomplicated urinary tract infection with trimethoprim–sulfamethoxazole (TMP-SMX). The pharmacist called and said the patient could not have any sulfa medications because she was going to donate a kidney to her brother. How do you explain this, and what should be my next step?
There is limited available evidence to support an answer to this question. Instead, I will provide a brief report of the available evidence, followed by the opinions of five experts in kidney transplantation.
TMP-SMX is a combination of two antimicrobial agents that act synergistically against a variety of bacteria.9 TMP decreases urinary excretion of potassium, leading to hyperkalemia—especially in persons with kidney disease or in those also taking other drugs that cause hyperkalemia.10 This scenario is unlikely, because the transplant team would have assessed the potential donor’s kidney function. When dosing the drug, clinicians should consider renal function and adjust the dose in those with a creatinine clearance less than or equal to 30 mL/min. Nephrotoxicity is uncommon in patients who take this drug; however, TMP is known to decrease tubular secretion of creatinine and may interfere with certain creatinine assays, leading to an artificial rise in serum creatinine. This is not reflective of a true reduction in the GFR and often is mild and reversible with discontinuation of the medication.
Querying experienced transplant professionals (two nephrologists, two transplant coordinators, and one doctoral-prepared pharmacist) yielded similar results. They all agreed that the only plausible reason to withhold TMP-SMX from this potential kidney donor was the risk of a transient rise in creatinine due to impaired secretion associated with TMP use. Many transplant teams recommend avoiding any medications that may affect the kidneys. So considering the lack of available evidence, I would recommend that you consult with the transplant team where your patient’s brother is receiving care before you prescribe TMP-SMX.
Debra Hain, PhD, APRN, GNP-BC
Florida Atlantic University, Boca Raton; Cleveland Clinic Florida; Department of Nephrology, Weston
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I was treating a patient for an uncomplicated urinary tract infection with trimethoprim–sulfamethoxazole (TMP-SMX). The pharmacist called and said the patient could not have any sulfa medications because she was going to donate a kidney to her brother. How do you explain this, and what should be my next step?
There is limited available evidence to support an answer to this question. Instead, I will provide a brief report of the available evidence, followed by the opinions of five experts in kidney transplantation.
TMP-SMX is a combination of two antimicrobial agents that act synergistically against a variety of bacteria.9 TMP decreases urinary excretion of potassium, leading to hyperkalemia—especially in persons with kidney disease or in those also taking other drugs that cause hyperkalemia.10 This scenario is unlikely, because the transplant team would have assessed the potential donor’s kidney function. When dosing the drug, clinicians should consider renal function and adjust the dose in those with a creatinine clearance less than or equal to 30 mL/min. Nephrotoxicity is uncommon in patients who take this drug; however, TMP is known to decrease tubular secretion of creatinine and may interfere with certain creatinine assays, leading to an artificial rise in serum creatinine. This is not reflective of a true reduction in the GFR and often is mild and reversible with discontinuation of the medication.
Querying experienced transplant professionals (two nephrologists, two transplant coordinators, and one doctoral-prepared pharmacist) yielded similar results. They all agreed that the only plausible reason to withhold TMP-SMX from this potential kidney donor was the risk of a transient rise in creatinine due to impaired secretion associated with TMP use. Many transplant teams recommend avoiding any medications that may affect the kidneys. So considering the lack of available evidence, I would recommend that you consult with the transplant team where your patient’s brother is receiving care before you prescribe TMP-SMX.
Debra Hain, PhD, APRN, GNP-BC
Florida Atlantic University, Boca Raton; Cleveland Clinic Florida; Department of Nephrology, Weston
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
Medications and the Renal Patient: CKD and Thiazide Diuretics
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I have a patient with CKD stage 4 (GFR, 30 mL/min/1.73m2). My supervising physician said to take him off his hydrochlorothiazide (50 mg qd) and start furosemide. Why do I do this, and at what dose do I start?
Thiazide diuretics work by blocking approximately 40% of sodium chloride reabsorption in the distal convoluting tubule of the nephron. This process increases the fractional excretion of sodium and provides a natriuresis with reduced blood pressure (BP). Thiazides also have a second mode of action in lowering BP by reducing peripheral vascular resistance.5
It is generally thought that thiazides are ineffective in patients with more advanced CKD because of more proximal sodium reabsorption in the nephron. This results in less sodium being delivered to the distal tubule and therefore less thiazide diuretic action in the distal tubule.5
The National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (K/DOQI)6 recommends that hypertension in CKD be treated with combination therapy: renin-angiotensin system blockade (ACE inhibitor/angiotensin receptor blocker) and thiazide diuretics in patients with CKD stages 1 to 3; and loop diuretics in those with CKD stages 4 and 5. No large study has evaluated thiazide diuretic use in patients with CKD stages 4 and 5. In one small study (n = 7) in which thiazides were compared with loop diuretics, there was similar BP control between the groups, but patients in the thiazide group had superior urinary fractional sodium excretion.5
There is new concern that thiazides may partially contribute to the increased risk for metabolic syndrome or diabetes. Reungjui et al7 have used animal models to demonstrate that the hypokalemic and hyperuricemic effects of thiazides are the culprits for this risk. They recommend managing the potassium and uric acid levels to manage or prevent this risk.
When a change is made from thiazides to loop diuretics, K/DOQI recommends starting furosemide at 40 mg/d and titrating upward as needed for BP and edema control. In my experience, counseling patients to follow a low-sodium diet (the American Heart Association recommends restricting sodium intake to 1500 mg/d) allows for a lower dose of furosemide (20 mg) to be effective.8
Elizabeth Evans, DNP
Renal Medicine Associates, Albuquerque, NM
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I have a patient with CKD stage 4 (GFR, 30 mL/min/1.73m2). My supervising physician said to take him off his hydrochlorothiazide (50 mg qd) and start furosemide. Why do I do this, and at what dose do I start?
Thiazide diuretics work by blocking approximately 40% of sodium chloride reabsorption in the distal convoluting tubule of the nephron. This process increases the fractional excretion of sodium and provides a natriuresis with reduced blood pressure (BP). Thiazides also have a second mode of action in lowering BP by reducing peripheral vascular resistance.5
It is generally thought that thiazides are ineffective in patients with more advanced CKD because of more proximal sodium reabsorption in the nephron. This results in less sodium being delivered to the distal tubule and therefore less thiazide diuretic action in the distal tubule.5
The National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (K/DOQI)6 recommends that hypertension in CKD be treated with combination therapy: renin-angiotensin system blockade (ACE inhibitor/angiotensin receptor blocker) and thiazide diuretics in patients with CKD stages 1 to 3; and loop diuretics in those with CKD stages 4 and 5. No large study has evaluated thiazide diuretic use in patients with CKD stages 4 and 5. In one small study (n = 7) in which thiazides were compared with loop diuretics, there was similar BP control between the groups, but patients in the thiazide group had superior urinary fractional sodium excretion.5
There is new concern that thiazides may partially contribute to the increased risk for metabolic syndrome or diabetes. Reungjui et al7 have used animal models to demonstrate that the hypokalemic and hyperuricemic effects of thiazides are the culprits for this risk. They recommend managing the potassium and uric acid levels to manage or prevent this risk.
When a change is made from thiazides to loop diuretics, K/DOQI recommends starting furosemide at 40 mg/d and titrating upward as needed for BP and edema control. In my experience, counseling patients to follow a low-sodium diet (the American Heart Association recommends restricting sodium intake to 1500 mg/d) allows for a lower dose of furosemide (20 mg) to be effective.8
Elizabeth Evans, DNP
Renal Medicine Associates, Albuquerque, NM
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I have a patient with CKD stage 4 (GFR, 30 mL/min/1.73m2). My supervising physician said to take him off his hydrochlorothiazide (50 mg qd) and start furosemide. Why do I do this, and at what dose do I start?
Thiazide diuretics work by blocking approximately 40% of sodium chloride reabsorption in the distal convoluting tubule of the nephron. This process increases the fractional excretion of sodium and provides a natriuresis with reduced blood pressure (BP). Thiazides also have a second mode of action in lowering BP by reducing peripheral vascular resistance.5
It is generally thought that thiazides are ineffective in patients with more advanced CKD because of more proximal sodium reabsorption in the nephron. This results in less sodium being delivered to the distal tubule and therefore less thiazide diuretic action in the distal tubule.5
The National Kidney Foundation’s Kidney Disease Outcomes Quality Initiative (K/DOQI)6 recommends that hypertension in CKD be treated with combination therapy: renin-angiotensin system blockade (ACE inhibitor/angiotensin receptor blocker) and thiazide diuretics in patients with CKD stages 1 to 3; and loop diuretics in those with CKD stages 4 and 5. No large study has evaluated thiazide diuretic use in patients with CKD stages 4 and 5. In one small study (n = 7) in which thiazides were compared with loop diuretics, there was similar BP control between the groups, but patients in the thiazide group had superior urinary fractional sodium excretion.5
There is new concern that thiazides may partially contribute to the increased risk for metabolic syndrome or diabetes. Reungjui et al7 have used animal models to demonstrate that the hypokalemic and hyperuricemic effects of thiazides are the culprits for this risk. They recommend managing the potassium and uric acid levels to manage or prevent this risk.
When a change is made from thiazides to loop diuretics, K/DOQI recommends starting furosemide at 40 mg/d and titrating upward as needed for BP and edema control. In my experience, counseling patients to follow a low-sodium diet (the American Heart Association recommends restricting sodium intake to 1500 mg/d) allows for a lower dose of furosemide (20 mg) to be effective.8
Elizabeth Evans, DNP
Renal Medicine Associates, Albuquerque, NM
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
Medications and the Renal Patient: NSAIDs and Acute Kidney Injury
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: In my orthopedic practice, we have a woman we are treating conservatively for low back pain (NSAIDs, muscle relaxants, and physical therapy). Her primary care provider told her that she cannot take the NSAIDs because of her kidney disease (she has chronic kidney disease [CKD] stage 3). Is there a safe dose of NSAIDs that she can use, or do I need to start narcotics? I would rather not do that!
Unfortunately, all NSAIDs increase the risk for acute kidney injury (AKI) and may exacerbate progression to chronic renal failure, particularly when large doses are taken chronically.3,4 Increased incidence of renal injury with NSAIDs has been seen in patients with existing CKD, hypertension, diabetes, and frequent hospitalizations.3 Most renal damage associated with NSAIDs is related to inhibition of prostaglandin synthesis (discussed below), but NSAIDs can also cause other types of kidney injury, such as interstitial nephritis, analgesic nephropathy, and membranous nephropathy. NSAID use is also associated with hyperkalemia, hyponatremia, and edema (sodium retention).
The primary mechanism of NSAID nephrotoxicity is inhibition of prostaglandin synthesis in the setting of decreased renal perfusion.3 Prostaglandins induce vasodilation of the afferent arterioles to maintain renal perfusion (and consequently GFR). If renal perfusion or effective fluid volume decreases (as in dehydration, diuretic therapy, heart failure, and cirrhosis), renal perfusion is maintained by increasing prostaglandin synthesis. NSAIDs block prostaglandin synthesis, therefore blunting this protective mechanism. Blocking prostaglandin with NSAIDs when renal perfusion is decreased can cause an AKI.
AKI can occur in patients with or without CKD, and it is associated with increased morbidity and mortality. AKI due to NSAID use may progress to end-stage renal disease if the NSAID is not stopped promptly. When NSAIDs are discontinued, renal function most often stabilizes, but residual renal insufficiency is likely to be permanent. In some patients, even after discontinuing the NSAID, the AKI progresses to more advanced kidney disease.
All NSAIDs carry the same risk for acute and chronic kidney disease, with little evidence to suggest that some are safer than others. Higher doses are more likely to cause renal damage, but dosing to prevent renal damage has not been defined. Indomethacin and ketorolac have most frequently been associated with AKI. If appropriate for the patient, acetaminophen 650 mg taken three times daily (scheduled, not “as needed”) can provide pain relief with less risk for kidney injury.
Additionally, narcotic medications are often necessary when treating severe pain in patients with high risk for NSAID-associated kidney injury.
Catherine Wells, DNP, ACNP, CNN-NP
University of Mississippi Health Care, Division of Nephrology, Jackson
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: In my orthopedic practice, we have a woman we are treating conservatively for low back pain (NSAIDs, muscle relaxants, and physical therapy). Her primary care provider told her that she cannot take the NSAIDs because of her kidney disease (she has chronic kidney disease [CKD] stage 3). Is there a safe dose of NSAIDs that she can use, or do I need to start narcotics? I would rather not do that!
Unfortunately, all NSAIDs increase the risk for acute kidney injury (AKI) and may exacerbate progression to chronic renal failure, particularly when large doses are taken chronically.3,4 Increased incidence of renal injury with NSAIDs has been seen in patients with existing CKD, hypertension, diabetes, and frequent hospitalizations.3 Most renal damage associated with NSAIDs is related to inhibition of prostaglandin synthesis (discussed below), but NSAIDs can also cause other types of kidney injury, such as interstitial nephritis, analgesic nephropathy, and membranous nephropathy. NSAID use is also associated with hyperkalemia, hyponatremia, and edema (sodium retention).
The primary mechanism of NSAID nephrotoxicity is inhibition of prostaglandin synthesis in the setting of decreased renal perfusion.3 Prostaglandins induce vasodilation of the afferent arterioles to maintain renal perfusion (and consequently GFR). If renal perfusion or effective fluid volume decreases (as in dehydration, diuretic therapy, heart failure, and cirrhosis), renal perfusion is maintained by increasing prostaglandin synthesis. NSAIDs block prostaglandin synthesis, therefore blunting this protective mechanism. Blocking prostaglandin with NSAIDs when renal perfusion is decreased can cause an AKI.
AKI can occur in patients with or without CKD, and it is associated with increased morbidity and mortality. AKI due to NSAID use may progress to end-stage renal disease if the NSAID is not stopped promptly. When NSAIDs are discontinued, renal function most often stabilizes, but residual renal insufficiency is likely to be permanent. In some patients, even after discontinuing the NSAID, the AKI progresses to more advanced kidney disease.
All NSAIDs carry the same risk for acute and chronic kidney disease, with little evidence to suggest that some are safer than others. Higher doses are more likely to cause renal damage, but dosing to prevent renal damage has not been defined. Indomethacin and ketorolac have most frequently been associated with AKI. If appropriate for the patient, acetaminophen 650 mg taken three times daily (scheduled, not “as needed”) can provide pain relief with less risk for kidney injury.
Additionally, narcotic medications are often necessary when treating severe pain in patients with high risk for NSAID-associated kidney injury.
Catherine Wells, DNP, ACNP, CNN-NP
University of Mississippi Health Care, Division of Nephrology, Jackson
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: In my orthopedic practice, we have a woman we are treating conservatively for low back pain (NSAIDs, muscle relaxants, and physical therapy). Her primary care provider told her that she cannot take the NSAIDs because of her kidney disease (she has chronic kidney disease [CKD] stage 3). Is there a safe dose of NSAIDs that she can use, or do I need to start narcotics? I would rather not do that!
Unfortunately, all NSAIDs increase the risk for acute kidney injury (AKI) and may exacerbate progression to chronic renal failure, particularly when large doses are taken chronically.3,4 Increased incidence of renal injury with NSAIDs has been seen in patients with existing CKD, hypertension, diabetes, and frequent hospitalizations.3 Most renal damage associated with NSAIDs is related to inhibition of prostaglandin synthesis (discussed below), but NSAIDs can also cause other types of kidney injury, such as interstitial nephritis, analgesic nephropathy, and membranous nephropathy. NSAID use is also associated with hyperkalemia, hyponatremia, and edema (sodium retention).
The primary mechanism of NSAID nephrotoxicity is inhibition of prostaglandin synthesis in the setting of decreased renal perfusion.3 Prostaglandins induce vasodilation of the afferent arterioles to maintain renal perfusion (and consequently GFR). If renal perfusion or effective fluid volume decreases (as in dehydration, diuretic therapy, heart failure, and cirrhosis), renal perfusion is maintained by increasing prostaglandin synthesis. NSAIDs block prostaglandin synthesis, therefore blunting this protective mechanism. Blocking prostaglandin with NSAIDs when renal perfusion is decreased can cause an AKI.
AKI can occur in patients with or without CKD, and it is associated with increased morbidity and mortality. AKI due to NSAID use may progress to end-stage renal disease if the NSAID is not stopped promptly. When NSAIDs are discontinued, renal function most often stabilizes, but residual renal insufficiency is likely to be permanent. In some patients, even after discontinuing the NSAID, the AKI progresses to more advanced kidney disease.
All NSAIDs carry the same risk for acute and chronic kidney disease, with little evidence to suggest that some are safer than others. Higher doses are more likely to cause renal damage, but dosing to prevent renal damage has not been defined. Indomethacin and ketorolac have most frequently been associated with AKI. If appropriate for the patient, acetaminophen 650 mg taken three times daily (scheduled, not “as needed”) can provide pain relief with less risk for kidney injury.
Additionally, narcotic medications are often necessary when treating severe pain in patients with high risk for NSAID-associated kidney injury.
Catherine Wells, DNP, ACNP, CNN-NP
University of Mississippi Health Care, Division of Nephrology, Jackson
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
Medications and the Renal Patient: Dialysis and BP
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I work in a cardiology practice. We received a note from the dialysis center telling us that one of our patients is hypotensive (systole < 100 mm Hg) during his dialysis treatment. His BP is usually 140/86 mm Hg in the office. Why the difference?
When considering BP values within this population, it is important to keep in mind that BP in dialysis patients can vary widely, with lower values in the period immediately following dialysis, then slowly increasing as patients’ fluid levels rise.
There are a few reasons why hypotension typically occurs during treatment. Taking sedating medication just before arriving for dialysis can dramatically lower BP during dialysis and should generally be avoided; advise the patient to take the medication after dialysis or at night instead.11 Many antihypertensive drugs that are removed by dialysis are often prescribed to be taken at night.
Another common reason for hypotension during dialysis is large-volume fluid removal. Patients are advised to limit fluids between treatments to avoid fluid overload, thereby limiting the volume of removal needed. Incorrect dry weight calculations can also cause hypotension during dialysis; if a patient gains weight that is not fluid related and attempts are made to dialyze the patient to the dry weight, hypotension can occur.11 The patient who sees another practitioner right before dialysis may appear volume-overloaded—or immediately after dialysis, may appear volume-depleted; neither impression is correct. Also, a 2- to 4-kg weight gain between dialysis treatments is acceptable.
It has been learned through observational research that hemodialysis patients tend to have higher mortality rates with a predialysis systolic BP (SBP) below 110 mm Hg, a postdialysis SBP greater than 180 mm Hg, or a postdialysis diastolic BP exceeding 110 mm Hg.12 According to the National Kidney Foundation’s K/DOQI practice guidelines,13 a predialysis BP of 140/90 mm Hg and a postdialysis BP of 130/80 mm Hg are reasonable targets. However, as with all guidelines, goals must be individualized to fit the patient’s age, comorbidities, and symptoms.14 This is a delicate balance, and safe management requires ongoing communication between providers.
Of interest, researchers for the Dialysis Outcomes and Practice Patterns Study suggested that patients with a predialysis SBP of 110 to 130 mm Hg had a higher risk for mortality than those with an SBP of 130 to 140 mm Hg. The same study showed an increased risk for death in patients with predialysis SBP greater than 160 mm Hg.14
Kristina Unterseher, CNN-NP
Idaho Nephrology Associates, Boise
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I work in a cardiology practice. We received a note from the dialysis center telling us that one of our patients is hypotensive (systole < 100 mm Hg) during his dialysis treatment. His BP is usually 140/86 mm Hg in the office. Why the difference?
When considering BP values within this population, it is important to keep in mind that BP in dialysis patients can vary widely, with lower values in the period immediately following dialysis, then slowly increasing as patients’ fluid levels rise.
There are a few reasons why hypotension typically occurs during treatment. Taking sedating medication just before arriving for dialysis can dramatically lower BP during dialysis and should generally be avoided; advise the patient to take the medication after dialysis or at night instead.11 Many antihypertensive drugs that are removed by dialysis are often prescribed to be taken at night.
Another common reason for hypotension during dialysis is large-volume fluid removal. Patients are advised to limit fluids between treatments to avoid fluid overload, thereby limiting the volume of removal needed. Incorrect dry weight calculations can also cause hypotension during dialysis; if a patient gains weight that is not fluid related and attempts are made to dialyze the patient to the dry weight, hypotension can occur.11 The patient who sees another practitioner right before dialysis may appear volume-overloaded—or immediately after dialysis, may appear volume-depleted; neither impression is correct. Also, a 2- to 4-kg weight gain between dialysis treatments is acceptable.
It has been learned through observational research that hemodialysis patients tend to have higher mortality rates with a predialysis systolic BP (SBP) below 110 mm Hg, a postdialysis SBP greater than 180 mm Hg, or a postdialysis diastolic BP exceeding 110 mm Hg.12 According to the National Kidney Foundation’s K/DOQI practice guidelines,13 a predialysis BP of 140/90 mm Hg and a postdialysis BP of 130/80 mm Hg are reasonable targets. However, as with all guidelines, goals must be individualized to fit the patient’s age, comorbidities, and symptoms.14 This is a delicate balance, and safe management requires ongoing communication between providers.
Of interest, researchers for the Dialysis Outcomes and Practice Patterns Study suggested that patients with a predialysis SBP of 110 to 130 mm Hg had a higher risk for mortality than those with an SBP of 130 to 140 mm Hg. The same study showed an increased risk for death in patients with predialysis SBP greater than 160 mm Hg.14
Kristina Unterseher, CNN-NP
Idaho Nephrology Associates, Boise
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.
When prescribing medications for patients, it is always advisable to know their estimated glomerular filtration rate (eGFR). The creatinine and blood urea nitrogen (BUN) by themselves are not always good indicators of renal function. If you have doubts, any reliable pharmacy source can guide you to dosing adjustments. Most medications do not require adjustments for eGFR greater than 60 mL/min/1.73m2.
Patients with an eGFR of less than 60 should never be prescribed NSAIDs, and extreme caution is advised with use of aminoglycosides and contrast dyes.
With medications such as ACE inhibitors, which can affect renal function (particularly levels of creatinine and potassium), eGFR should be monitored initially and within two weeks of each dosing adjustment. Other commonly prescribed drugs requiring dosing adjustment in patients with eGFR below 60 include gabapentin, metoclopramide, and ranitidine.1,2
As always, inquire about your patient’s use of complementary and alternative therapies, including herbal remedies, as these often are contraindicated in this population.
Jane S. Davis, CRNP, DNP
Q: I work in a cardiology practice. We received a note from the dialysis center telling us that one of our patients is hypotensive (systole < 100 mm Hg) during his dialysis treatment. His BP is usually 140/86 mm Hg in the office. Why the difference?
When considering BP values within this population, it is important to keep in mind that BP in dialysis patients can vary widely, with lower values in the period immediately following dialysis, then slowly increasing as patients’ fluid levels rise.
There are a few reasons why hypotension typically occurs during treatment. Taking sedating medication just before arriving for dialysis can dramatically lower BP during dialysis and should generally be avoided; advise the patient to take the medication after dialysis or at night instead.11 Many antihypertensive drugs that are removed by dialysis are often prescribed to be taken at night.
Another common reason for hypotension during dialysis is large-volume fluid removal. Patients are advised to limit fluids between treatments to avoid fluid overload, thereby limiting the volume of removal needed. Incorrect dry weight calculations can also cause hypotension during dialysis; if a patient gains weight that is not fluid related and attempts are made to dialyze the patient to the dry weight, hypotension can occur.11 The patient who sees another practitioner right before dialysis may appear volume-overloaded—or immediately after dialysis, may appear volume-depleted; neither impression is correct. Also, a 2- to 4-kg weight gain between dialysis treatments is acceptable.
It has been learned through observational research that hemodialysis patients tend to have higher mortality rates with a predialysis systolic BP (SBP) below 110 mm Hg, a postdialysis SBP greater than 180 mm Hg, or a postdialysis diastolic BP exceeding 110 mm Hg.12 According to the National Kidney Foundation’s K/DOQI practice guidelines,13 a predialysis BP of 140/90 mm Hg and a postdialysis BP of 130/80 mm Hg are reasonable targets. However, as with all guidelines, goals must be individualized to fit the patient’s age, comorbidities, and symptoms.14 This is a delicate balance, and safe management requires ongoing communication between providers.
Of interest, researchers for the Dialysis Outcomes and Practice Patterns Study suggested that patients with a predialysis SBP of 110 to 130 mm Hg had a higher risk for mortality than those with an SBP of 130 to 140 mm Hg. The same study showed an increased risk for death in patients with predialysis SBP greater than 160 mm Hg.14
Kristina Unterseher, CNN-NP
Idaho Nephrology Associates, Boise
REFERENCES
1. Gabardi S, Abramson S. Drug dosing in chronic kidney disease. Med Clin North Am. 2005;89(3):649-687.
2. Munar MY, Singh H. Drug dosing adjustments in patients with chronic kidney disease. Am Fam Physician. 2007;75(10):1487-1496.
3. Huerta C, Castellsague J, Varas-Lorenzo C, Garcia Rodriguez LA. Nonsteroidal anti-inflammatory drugs and risk of ARF in the general population. Am J Kidney Dis. 2005;45(3): 531-539.
4. Schneider V, Lévesque LE, Zhang B, et al. Association of selective and conventional nonsteroidal antiinflammatory drugs with acute renal failure: a population-based, nested case-control analysis. Am J Epidemiol. 2006; 164(9):881-889.
5. Loyd J, Wright P. Are thiazide diuretics an effective treatment for hypertension in patients with chronic kidney disease? J Okla State Med Assoc. 2008;101(5):84-85.
6. Kidney Disease Outcomes Quality Initiative (K/DOQI). K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis. 2004;43(5 suppl 1):S1-S290.
7. Reungjui S, Pratipanawatr T, Johnson RJ, Nakagawa T. Do thiazides worsen metabolic syndrome and renal disease? The pivotal roles for hyperuricemia and hypokalemia. Curr Opin Nephrol Hypertens. 2008;17(5):470-476.
8. Lichtenstein AH, Appel LJ, Brands M, et al. Diet and lifestyle recommendations revision 2006: a scientific statement from the American Heart Association Nutrition Committee. Circulation. 2006;114(1):82-96.
9. Pharmacokinetics. In: Golan DE, Tashjian AH, Armstrong EJ, Armstrong AW, eds. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. 2nd ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2007:31-48.
10. Masters PA, O’Bryan TA, Zurlo J, et al. Trimethoprim-sulfamethoxazole revisited. Arch Intern Med. 2003;163(4):402-410.
11. Singapuri MS, Lea JP. Management of hypertension in the end-stage renal disease patient. J Clin Outcomes Manage. 2010;17(2):87-95.
12. Port FK, Hulbert-Shearon TE, Wolfe RA, et al. Predialysis blood pressure and mortality risk in a national sample of maintenance hemodialysis patients. Am J Kidney Dis. 1999;33(3): 507-517.
13. K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis. 2005;45(4 suppl 3):S1–S153.
14. Schieszer J. BP guidelines may be inappropriate for HD patients. Renal Urol News. 2010 May 21. www.renalandurologynews.com/bp-guidelines-may-be-inappropriate-for-hd-patients/article/170707. Accessed May 19, 2011.